User login
Cannabidiol (CBD) for schizophrenia: Promise or pipe dream?
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.
Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.

Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
Over the past few decades, it has become increasingly clear that cannabis use can increase the risk of developing a psychotic disorder and worsen the course of existing schizophrenia in a dose-dependent fashion.1-3 Beyond psychosis, although many patients with mental illness use cannabis for recreational purposes or as purported “self-medication,” currently available evidence suggests that marijuana is more likely to represent a harm than a benefit for psychiatric disorders4 (Box4-8). Our current state of knowledge therefore suggests that psychiatrists should caution their patients against using cannabis and prioritize interventions to reduce or discontinue use, especially among those with psychotic disorders.
Box
Data from California in 2006—a decade after the state’s legalization of “medical marijuana”—revealed that 23% of patients in a sample enrolled in medical marijuana clinics were receiving cannabis to treat a mental disorder.5 That was a striking statistic given the dearth of evidence to support a benefit of cannabis for psychiatric conditions at the time, leaving clinicians who provided the necessary recommendations to obtain medical marijuana largely unable to give informed consent about the risks and benefits, much less recommendations about specific products, routes of administration, or dosing. In 2019, we know considerably more about the interaction between cannabinoids and mental health, but research findings thus far warrant more caution than enthusiasm, with one recent review concluding that “whenever an association is observed between cannabis use and psychiatric disorders, the relationship is generally an adverse one.”4
Some critics have argued that the medical marijuana industry represents little more than a front for recreational use. In California and other states that have legalized recreational use, that claim has been rendered all but moot, although the public remains curious about the potential health benefits of cannabinoids and will likely continue to look to clinicians for advice. For those seeking guidance from evidence-based research, the existing state of knowledge can seem like a “Wild West” of anecdotal subjective reports, biased opinions, and uncontrolled clinical studies. Cannabis remains a Schedule I drug at the federal level, and quality clinical research has been limited to a relatively modest number of randomized controlled trials (RCTs), mostly involving FDA-approved cannabinoids rather than smoked cannabis. Randomized controlled trials that have involved smoked marijuana have generally involved low-potency delta-9-tetrahydrocannabinol (THC) cannabis that may not reflect the same therapeutic and adverse effects of the increasingly high potency cannabis now available on the street and in dispensaries.
In psychiatry, a few RCTs are underway exploring cannabis as a viable treatment for mental disorders (eg, posttraumatic stress disorder), but none have yet been completed or published. At best, retrospective studies to date have failed to support a consistent benefit of cannabis for any psychiatric disorder and at worst increasingly suggest a negative impact on psychotic, mood, and anxiety disorders.4,6 Meanwhile, synthetic cannabinoid receptor agonists (eg, “Spice” products) have come to represent a clear public health risk, with both medical and psychiatric toxicity.7
A more cautiously optimistic case for the therapeutic potential of cannabinoids in psychiatry could be made for cannabidiol (CBD), which may possess anxiolytic, antipsychotic, and neuroprotective properties.8 Based on its purported health benefits, it is possible that CBD may even gain widespread popularity as a food supplement. Because a pharmaceutically-manufactured form of CBD was recently FDA-approved for the treatment of seizures associated with Lennox-Gastaut syndrome and Dravet syndrome, off-label prescribing of CBD for psychiatric disorders can be anticipated. While there is not yet sufficient evidence about risks and benefits to justify CBD being recommended broadly in psychiatry, that same informational vacuum has not stopped eager patients from seeking approval for cannabis, and some physicians from providing it.
Despite that conclusion, because cannabis is classified as a Schedule I drug by the US Drug Enforcement Agency, clinical research investigating the risks and benefits of cannabis has been limited. It therefore remains possible that cannabis, or individual cannabinoids such as cannabidiol (CBD), may yet find a therapeutic niche in psychiatry. This article reviews evidence on CBD for the treatment of schizophrenia.
Cannabinergic drugs as potential antipsychotics
Although the bulk of evidence indicates a harmful effect of cannabis in individuals with or at risk for psychosis, there have been a few published cases of schizophrenia improving with dronabinol, an FDA-approved, synthetic form of delta-9-tetrahydrocannabinol (THC).9,10 THC is the constituent of cannabis that produces euphoric effects. These provocative findings have not been replicated in controlled clinical trials, but suggest at least the theoretical possibility of idiosyncratic benefits from THC for some individuals within the psychotic spectrum.
Still, given that most available evidence supports that THC has a harmful effect on psychosis and psychosis risk, researchers have instead performed randomized controlled trials (RCTs) to investigate a possible therapeutic role for medications that oppose the agonist effects of THC at cannabinoid type 1 (CB1) receptors. To date, 2 RCTs comparing rimonabant, a CB1 inverse agonist, with placebo (PLB) in patients with schizophrenia have failed to demonstrate any benefit for psychotic symptoms or cognitive deficits.11,12 A third trial examining rimonabant for people diagnosed with schizophrenia who were overweight found significant benefits for anxiety and depressive symptoms, but none for positive symptoms or the primary outcome of weight loss.13 While these results are discouraging, the role of THC in precipitating psychosis suggests that novel agents opposing the actions of THC on the cannabinoid system could have antipsychotic properties.14
Cannabidiol: An antipsychotic medication?
In contrast to THC, CBD has minimal euphorigenic properties and has recently been heralded in the popular press as a “miracle drug” with benefits for medical and psychiatric disorders alike.15 It has even been speculated that it could become a popular food supplement.16 In 2018, the FDA gave full approval to a pharmaceutically manufactured form of CBD (brand name: Epidiolex) as a novel treatment for 2 rare and severe forms of pediatric epilepsy, Lennox-Gastaut syndrome and Dravet syndrome,17 based on RCTs supporting its efficacy for these often refractory and life-threatening conditions.18-20
In psychiatry, there have not yet been enough robust clinical studies to support broad therapeutic claims for CBD as a treatment for any mental disorder.21 However, there is growing evidence that CBD has potential as an antipsychotic medication. In 1995, the first case report was published describing the efficacy of CBD, 1,500 mg/d, as standalone therapy in a single individual with schizophrenia.22 In 2006, the same research group followed up with a case series in which only 1 out of 3 patients with treatment-refractory schizophrenia improved with flexible dosing of CBD to a maximum dose of 1,280 mg/d.23
There have been 3 published RCTs exploring the efficacy of CBD in schizophrenia (Table24-26). The first study, published in 2012, included 39 adults with schizophrenia who were randomized to 800 mg/d of CBD or amisulpride (AMS), a second-generation antipsychotic that is popular in Europe but is not available in the United States.24 Over 4 weeks of randomized treatment, CBD resulted in as much improvement in overall symptoms and positive symptoms as AMS, and improvement of negative symptoms was significantly greater with CBD. Compared with patients treated with antipsychotic medication, patients who were treated with CBD had fewer extrapyramidal symptoms, less weight gain, and less prolactin elevation. This initial trial suggests that CBD might be as efficacious in schizophrenia as antipsychotic medication, without its burdensome adverse effects. However, this is the only RCT of CBD monotherapy published to date.

Continue to: Two other recently published RCTs...
Two other recently published RCTs compared CBD with PLB as add-on therapy to antipsychotics. McGuire et al25 compared CBD, 1,000 mg/d, to PLB over 6 weeks in 88 patients with schizophrenia. Positive symptom improvement was statistically greater with CBD than with PLB, although the magnitude of clinical change was modest (using the Positive and Negative Syndrome Scale [PANSS] positive symptom subscale: −3.2 points for CBD vs −1.7 points for PLB). Changes in PANSS total score and subscales for general and negative symptoms were not significantly different between treatment groups. There was also no significant difference in overall change in neurocognitive symptoms, although post-hoc analysis revealed significantly greater improvement in motor speed for patients treated with CBD. More than twice the number of patients treated with CBD were rated as “much improved” by the Clinical Global Impressions scale compared with patients treated with PLB, but this was not a statistically significant finding, and most patients experienced only “minimal” or “no improvement.” In terms of adverse events, there were no significant differences between patients in the CBD and PLB groups. Although this study is technically “positive” for CBD and suggests minimal adverse effects, it is not clear whether the statistically significant positive symptom improvements (+1.5 PANSS points for CBD over PLB) were clinically significant.
The most recently published placebo-controlled RCT of CBD as add-on therapy to antipsychotic medication included 36 patients with schizophrenia treated over 6 weeks.26 In this study, there was no benefit of CBD, 600 mg/d, on any PANSS score outcome (total, general, positive, or negative symptoms). For the primary outcome of the Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery, there were no significant drug × time effects, and post-hoc analyses showed that only patients treated with PLB improved with time. Sedation was more common among patients treated with CBD compared with PLB.
Making sense of the data
There have been mixed results from the few case reports and 3 RCTs of patients with schizophrenia who were treated with CBD. How can we resolve these disparate findings? A few possible interpretations of the data that warrant clarification through additional research include:
Dosing. In the first case report with positive results, CBD was dosed at 1,500 mg/d,22 whereas in the subsequent case series with mixed results, the maximum allowable dose of CBD was 1,280 mg/d.23 Likewise, in the RCTs, positive results were found when CBD was dosed at 800 to 1,000 mg/d,24,25 but not at 600 mg/d.26 The efficacy of CBD for schizophrenia might depend on higher doses.
Treatment resistance. In the second case series in which only 1 out of 3 patients responded to treatment with CBD,23 the patients had demonstrated previous nonresponse to at least 2 first-generation antipsychotics (FGAs) and risperidone, 6 mg/d. In the RCTs, all patients were antipsychotic-responsive.24-26 Cannabidiol may not be as effective for patients with treatment-refractory schizophrenia as it is for patients with schizophrenia who respond to antipsychotics.
Continue to: Clinical stability
Clinical stability. Within the RCTs, the greatest response was observed in the study that enrolled patients who were hospitalized with acute symptoms of schizophrenia.23 In the 2 studies that found either modest or no benefit with CBD, the patients had been stabilized on antipsychotic medications prior to randomization. Cannabidiol may offer limited benefit as add-on therapy to patients who have already responded to antipsychotic treatment, where there is “less room” for additional improvement.
Monotherapy. Both the case reports22,23 and the RCT with the most robust positive findings24 involved treatment with CBD as monotherapy. For some patients with schizophrenia, CBD might be effective as standalone therapy as an alternative to antipsychotics that is better tolerated. Adding CBD to antipsychotic therapy might be redundant and therefore less effective.
Answering questions about CBD
Cannabidiol is becoming increasingly popular for its purported health benefits. The mixed results of the few studies published on CBD for schizophrenia place clinicians in a difficult position when attempting to answer questions about how cannabinoids might fit into treatment of patients with psychosis. Consider the following:
Is cannabis helpful for patients with schizophrenia? No. Aside from the few case reports suggesting that FDA-approved THC (dronabinol) can improve symptoms in some patients,9,10 most of the evidence from anecdotal reports and both experimental and observational studies indicate that cannabis, THC, and synthetic cannabinoids have a harmful effect in patients with or at risk for psychosis.1-3
If you are considering recommending some form of cannabis to patients with schizophrenia, what kind should you recommend? Recommending or encouraging cannabis use for patients with psychosis is ill-advised. Although certain types of cannabis might contain more THC (eg, Cannabis indica vs Cannabis sativa) or variable amounts of CBD, in general the amount of CBD in whole leaf cannabis is minimal, with the ratio of THC to CBD increasingly significantly over the past decade.3,27 Most forms of cannabis should therefore be avoided by individuals with or at risk for psychotic disorders.
Continue to: What about CBD oil and other CBD products sold in dispensaries?
What about CBD oil and other CBD products sold in dispensaries? Cannabidiol is increasingly available in various forms based on its ability to be designated as a legal hemp product (containing <0.3% THC) at the federal level or as a cannabinoid in states where cannabis is legal. However, several studies have now shown that cannabis products sold online or in dispensaries are often labeled inaccurately, with both under- and over-reporting of THC and CBD content.28-30 Some CBD products have been found to have almost no CBD at all.29,30 The unreliability of product labeling makes it difficult to predict the effects of CBD products that are not subject to FDA purity standards for medications or dietary supplements. It also raises questions about the sources of CBD and the reliability of dosing in the studies discussed above.
Why might CBD work as an antipsychotic? Although CBD has minimal affinity for cannabinoid receptors, it appears to act as a partial agonist of dopamine D2 receptors and an agonist at 5-HT1A receptors, with overall effects that decrease mesolimbic dopamine activity.31,32 In addition, CBD increases the availability of the endogenous cannabinoid anandamide, which may have antipsychotic properties.14,33
Now that the FDA has approved CBD manufactured by a pharmaceutical company, should it be prescribed “off-label” for patients with schizophrenia? This is the “million dollar question,” with insufficient evidence to provide a clear answer. It should now be possible to prescribe FDA-approved CBD for off-label purposes, including the treatment of schizophrenia and other psychiatric disorders. No doubt, some clinicians are already doing so. This will predictably yield more anecdotal evidence about efficacy and adverse effects in the future, but there is not yet adequate evidence to support an FDA indication for CBD in schizophrenia. Additional studies of CBD for schizophrenia are ongoing.
Bottom Line
Cannabidiol (CBD) is becoming increasingly popular based on its purported health benefits, but the evidence supporting a therapeutic role in psychiatry is preliminary at best. Although CBD is now available by prescription as an FDA-approved drug for the treatment of 2 rare forms of epilepsy, its benefits in patients with schizophrenia are uncertain based on mixed results in clinical trials.
Related Resources
- Clinicaltrials.gov. Studies of “cannabidiol” and “schizophrenia.” U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/results?cond=Schizophrenia&term=cannabidiol.
- Grinspoon P. Cannabidiol (CBD) – what we know and what we don’t. Harvard Health Blog. https://www.health.harvard.edu/blog/cannabidiol-cbd-what-we-know-and-what-wedont-2018082414476. Published August 24, 2018.
Drug Brand Names
Cannabidiol • Epidiolex
Dronabinol • Marinol
Risperidone • Risperdal
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
1. Pierre JM. Cannabis, synthetic cannabinoids, and psychosis risk: what the evidence says. Current Psychiatry. 2011;10(9):49-58.
2. Radhakrishan R, Wilkinson ST, D’Souza DC. Gone to pot – a review of the association between cannabis and psychosis. Front Psychiatry. 2014;5:54.
3. Pierre JM. Risks of increasingly potent cannabis: joint effects of potency and frequency. Current Psychiatry. 2016;16(2):14-20.
4. Hanna RC, Perez JM, Ghose S. Cannabis and development of dual diagnoses: a literature review. Am J Drug Alcohol Abuse. 2017;43(4):442-255.
5. Nunberg H, Kilmer B, Pacula RL, et al. An analysis of applicants presenting to a medical marijuana specialty practice in California. J Drug Policy Anal. 2011;4(1):1.
6. Wilkinson ST, Radhakrishnan, D’Souza DC. A systematic review of the evidence for medical marijuana in psychiatric indications. J Clin Psychiatry. 2016;77(8):1050-1064.
7. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: Update 2015. Subst Abus. 2016;38(3):344-366.
8. Crippa JA, Guimarães FS, Campos A, et al. Translational investigation of the therapeutic potential of cannabidiol (CBD): toward a new age. Front Immunol. 2018;9:2009.
9. Schwarz G, Karajgi B. Improvement in refractory psychosis with dronabinol: four case reports. J Clin Psychiatry. 2010;71(11):1552-1553.
10. Schwarz G, Karajgi B, McCarthy R. Synthetic delta-9-tetrahydrocannabinol (dronabinol) can improve the symptoms of schizophrenia. J Clin Psychopharmacol. 2009;29(3):255-258.
11. Meltzer HY, Arvanitis L, Bauer D, et al. Placebo-controlled evaluation of four novel compounds for the treatment of schizophrenia and schizoaffective disorder. Am J Psychiatry. 2004;161(6):975-984.
12. Boggs DL, Kelly DL, McMahon RP, et al. Rimonabant for neurocognition in schizophrenia: a 16-week double blind placebo controlled trial. Schizophr Res. 2012;134(2-3):207-210.
13. Kelly DL, Gorelick DA, Conley RR, et al. Effects of cannabinoid-1 receptor antagonist rimonabant on psychiatric symptoms in overweight people with schizophrenia: a randomized, double-blind, pilot study. J Clin Psychopharmacol. 2011;31(1):86-91.
14. Leweke FM, Mueller JK, Lange B, et al. Therapeutic potential of cannabinoids in psychosis. Biol Psychiatry. 2016;79(7):604-612.
15. Halperin A. What is CBD? The ‘miracle’ cannabis compound that doesn’t get you high. The Guardian. https://www.theguardian.com/society/2018/may/28/what-is-cbd-cannabidiol-cannabis-medical-uses. Published May 28, 2018. Accessed April 3, 2019.
16. Pierre J. Coca, cola, and cannabis: psychoactive drugs as beverages. Psychology Today (blog) Psych Unseen. https://www.psychologytoday.com/us/blog/psych-unseen/201810/coca-cola-and-cannabis-psychoactive-drugs-beverages. Published October 1, 2018. Accessed April 3, 2019.
17. U.S. Food and Drug Administration. FDA approves first drug comprised of an active ingredient derived from marijuana to treat rare, severe forms of epilepsy. FDA News Release. https://www.fda.gov/newsevents/newsroom/pressannouncements/ucm611046.htm. Published June 25, 2018. Accessed April 3, 2019.
18. Devinsky O, Cross JH, Laux L, et al. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N Engl J Med. 2017;376:2011-2020.
19. Thiele EA, March ED, French JA, et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet. 2018;391(10125):1085-1096.
20. Devinsky O, Patel AD, Cross JH, et al. Effect of cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N Engl J Med. 2018;378:1888-1897.
21. Khoury JM, Neves MCLD, Rogue MAV, et al. Is there a role of cannabidiol in psychiatry? World J Biol Psychiatry. 2017:1-16.
22. Zuardi AW, Morais SL, Guimares FS, et al. Antipsychotic effect of cannabidiol. J Clin Psychiatry. 1995;56(10):485-486.
23. Zuardi AW, Hallak JEC, Dursun SM. Cannabidiol monotherapy for treatment-resistant schizophrenia. J Psychopharmacol. 2006;20(5):683-686.
24. Leweke FM, Piomelli D, Pahlisch F, et al. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl Psychiatry. 2012;2:e94. doi: 10.1038/tp.2012.15.
25. McGuire P, Robson P, Cubala WJ, et al. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: a multicenter randomized controlled trial. Am J Psychiatry. 2018;175(3):225-231.
26. Boggs DL, Surti I, Gupta A, et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacol. 2018;235(7):1923-1932.
27. ElSohly MA, Mehmedic Z, Foster S, et al. Changes in cannabis potency over the last 2 decades (1995-2014): analysis of current data in the United States. Biol Psychiatry. 2016; 79(7):613-619.
28. Vandrey R, Raber JC, Raber ME, et al. Cannabinoid dose and label accuracy in edible medical cannabis products. JAMA. 2015;313(24):2491-2492.
29. Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, et al. Consistency of label claims of internet-purchased hemp oil and cannabis products as determined using IMS and LC-MS: a marketplace study. J Reg Sci. 2016;3:1-6.
30. Bonn-Miller MO, Loflin MJE, Thomas BF, et al. Labeling accuracy of cannabidiol extracts sold online. JAMA. 2017;318(17):1708-1709.
31. Seeman P. Cannabidiol is a partial agonist at dopamine D2High receptors, predicting its antipsychotic clinical dose. Transl Psychiatry. 2016;6(10):e920. doi: 10.1038/tp.2016.195.
32. Renard J, Norris C, Rushlow W, et al. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;157-165.
33. Gururajan A, Malone DT. Does cannabidiol have a role in the treatment of schizophrenia? Schizophr Res. 2016;176(2-3):281-290.
Management of Early Pulmonary Complications After Hematopoietic Stem Cell Transplantation
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
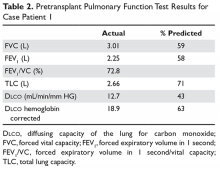
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
Hematopoietic stem cell transplantation (HSCT) is widely used in the economically developed world to treat a variety of hematologic malignancies as well as nonmalignant diseases and solid tumors. An estimated 17,900 HSCTs were performed in 2011, and survival rates continue to increase.1 Pulmonary complications post HSCT are common, with rates ranging from 40% to 60%, and are associated with increased morbidity and mortality.2
Clinical diagnosis of pulmonary complications in the HSCT population has been aided by a previously well-defined chronology of the most common diseases.3 Historically, early pulmonary complications were defined as pulmonary complications occurring within 100 days of HSCT (corresponding to the acute graft-versus-host disease [GVHD] period). Late pulmonary complications are those that occur thereafter. This timeline, however, is now more variable given the increasing indications for HSCT, the use of reduced-intensity conditioning strategies, and varied individual immune reconstitution. This article discusses the management of early post-HSCT pulmonary complications; late post-HSCT pulmonary complications will be discussed in a separate follow-up article.
Transplant Basics
The development of pulmonary complications is affected by many factors associated with the transplant. Autologous transplantation involves the collection of a patient’s own stem cells, appropriate storage and processing, and re-implantation after induction therapy. During induction therapy, the patient undergoes high-dose chemotherapy or radiation therapy that ablates the bone marrow. The stem cells are then transfused back into the patient to repopulate the bone marrow. Allogeneic transplants involve the collection of stem cells from a donor. Donors are matched as closely as possible to the recipient’s histocompatibility antigen (HLA) haplotypes to prevent graft failure and rejection. The donor can be related or unrelated to the recipient. If there is not a possibility of a related match (from a sibling), then a national search is undertaken to look for a match through the National Marrow Donor Program. There are fewer transplant reactions and occurrences of GVHD if the major HLAs of the donor and recipient match. Table 1 reviews basic definitions pertaining to HSCT.
How the cells for transplantation are obtained is also an important factor in the rate of complications. There are 3 main sources: peripheral blood, bone marrow, and umbilical cord. Peripheral stem cell harvesting involves exposing the donor to granulocyte-colony stimulating factor (gCSF), which increases peripheral circulation of stem cells. These cells are then collected and infused into the recipient after the recipient has completed an induction regimen involving chemotherapy and/or radiation, depending on the protocol. This procedure is called peripheral blood stem cell transplant (PBSCT). Stem cells can also be directly harvested from bone marrow cells, which are collected from repeated aspiration of bone marrow from the posterior iliac crest.4 This technique is most common in children, whereas in adults peripheral blood stem cells are the most common source. Overall mortality does not differ based on the source of the stem cells. It is postulated that GVHD may be more common in patients undergoing PBSCT, but the graft failure rate may be lower.5
The third option is umbilical cord blood (UCB) as the source of stem cells. This involves the collection of umbilical cord blood that is prepared and frozen after birth. It has a smaller volume of cells, and although fewer cells are needed when using UCB, 2 separate donors may be required for a single adult recipient. The engraftment of the stem cells is slower and infections in the post-transplant period are more common. Prior reports indicate GVHD rates may be lower.4 While the use of UCB is not common in adults, the incidence has doubled over the past decade, increasing from 3% to 6%.
The conditioning regimen can influence pulmonary complications. Traditionally, an ablative transplant involves high-dose chemotherapy or radiation to eradicate the recipient’s bone marrow. This regimen can lead to many complications, especially in the immediate post-transplant period. In the past 10 years, there has been increasing interest in non-myeloablative, or reduced-intensity, conditioning transplants.6 These “mini transplants” involve smaller doses of chemotherapy or radiation, which do not totally eradicate the bone marrow; after the transplant a degree of chimerism develops where the donor and recipient stem cells coexist. The medications in the preparative regimen also should be considered because they can affect pulmonary complications after transplant. Certain chemotherapeutic agents such as carmustine, bleomycin, and many others can lead to acute and chronic presentations of pulmonary diseases such as hypersensitivity pneumonitis, pulmonary fibrosis, acute respiratory distress syndrome, and abnormal pulmonary function testing.
After the HSCT, GVHD can develop in more than 50% of allogeneic recipients.3 The incidence of GVHD has been reported to be increasing over the past 12 years.It is divided into acute GVHD (which traditionally happens in the first 100 days after transplant) and chronic GVHD (after day 100). This calendar-day–based system has been augmented based on a 2006 National Institutes of Health working group report emphasizing the importance of organ-specific features of chronic GVHD in the clinical presentation of GVHD.7 Histologic changes in chronic organ GVHD tend to include more fibrotic features, whereas in acute GVHD more inflammatory changes are seen. The NIH working group report also stressed the importance of obtaining a biopsy specimen for histopathologic review and interdisciplinary collaboration to arrive at a consensus diagnosis, and noted the limitations of using histologic changes as the sole determinant of a “gold standard” diagnosis.7 GVHD can directly predispose patients to pulmonary GVHD and indirectly predispose them to infectious complications because the mainstay of therapy for GVHD is increased immunosuppression.
Pretransplant Evaluation
Case Patient 1
A 56-year-old man is diagnosed with acute myeloid leukemia (AML) after presenting with signs and symptoms consistent with pancytopenia. He has a past medical history of chronic sinus congestion, arthritis, depression, chronic pain, and carpal tunnel surgery. He is employed as an oilfield worker and has a 40-pack-year smoking history, but he recently cut back to half a pack per day. He is being evaluated for allogeneic transplant with his brother as the donor and the planned conditioning regimen is total body irradiation (TBI), thiotepa, cyclophosphamide, and antithymocyte globulin with T-cell depletion. Routine pretransplant pulmonary function testing (PFT) reveals a restrictive pattern and he is sent for pretransplant pulmonary evaluation.
Physical exam reveals a chronically ill appearing man. He is afebrile, the respiratory rate is 16 breaths/min, blood pressure is 145/88 mm Hg, heart rate is 92 beats/min, and oxygen saturation is 95%. He is in no distress. Auscultation of the chest reveals slightly diminished breath sounds bilaterally but is clear and without wheezes, rhonchi, or rales. Heart exam shows regular rate and rhythm without murmurs, rubs, or gallops. Extremities reveal no edema or rashes. Otherwise, the remainder of the exam is normal. The patient’s PFT results are shown in Table 2.
- What aspects of this patient’s history put him at risk for pulmonary complications after transplantation?
Risk Factors for Pulmonary Complications
Predicting who is at risk for pulmonary complications is difficult. Complications are generally divided into infectious and noninfectious categories. Regardless of category, allogeneic HSCT recipients are at increased risk compared with autologous recipients, but even in autologous transplants, more than 25% of patients will develop pulmonary complications in the first year.8 Prior to transplant, patients undergo full PFT. Early on, many studies attempted to show relationships between various factors and post-transplant pulmonary complications. Factors that were implicated were forced expiratory volume in 1 second (FEV1), diffusing capacity of the lung for carbon monoxide (D
Another sometimes overlooked risk before transplantation is restrictive lung disease. One study showed a twofold increase in respiratory failure and mortality if there was pretransplant restriction based on TLC < 80%.16
An interesting study by one group in pretransplant evaluation found decreased muscle strength by maximal inspiratory muscle strength (PImax), maximal expiratory muscle strength (PEmax), dominant hand grip strength, and 6-minute walk test (6MWT) distance prior to allogeneic transplant, but did not find a relationship between these variables and mortality.17 While this study had a small sample size, these findings likely deserve continued investigation.18
- What methods are used to calculate risk for complications?
Risk Scoring Systems
Several pretransplantation risk scores have been developed. In a study that looked at more than 2500 allogeneic transplants, Parimon et al showed that risk of mortality and respiratory failure could be estimated prior to transplant using a scoring system—the Lung Function Score (LFS)—that combines the FEV1 and D
The Pretransplantation Assessment of Mortality score, initially developed in 2006, predicts mortality within the first 2 years after HSCT based on 8 clinical factors: disease risk, age at transplant, donor type, conditioning regimen, and markers of organ function (percentage of predicted FEV1, percentage of predicted D
- What other preoperative testing or interventions should be considered in this patient?
Since there is a high risk of infectious complications after transplant, the question of whether pretransplantation patients should undergo screening imaging may arise. There is no evidence that routine chest computed tomography (CT) reduces the risk of infectious complications after transplantation.26 An area that may be insufficiently addressed in the pretransplantation evaluation is smoking cessation counseling.27 Studies have shown an elevated risk of mortality in smokers.28-30 Others have found a higher incidence of respiratory failure but not an increased mortality.31 Overall, with the good rates of smoking cessation that can be accomplished, smokers should be counseled to quit before transplantation.
In summary, patients should undergo full PFTs prior to transplantation to help stratify risk for pulmonary complications and mortality and to establish a clinical baseline. The LFS (using FEV1 and D
Case Patient 1 Conclusion
The patient undergoes transplantation due to his lack of other treatment options. Evaluation prior to transplant, however, shows that he is at high risk for pulmonary complications. He has a LFS of 7 prior to transplant (using the D
Early Infectious Pulmonary Complications
Case Patient 2
A 27-year-old man with a medical history significant for AML and allogeneic HSCT presents with cough productive of a small amount of clear to white sputum, dyspnea on exertion, and fevers for 1 week. He also has mild nausea and a decrease in appetite. He underwent HSCT 2.5 months prior to admission, which was a matched unrelated bone marrow transplant with TBI and cyclophosphamide conditioning. His past medical history is significant only for exercise-induced asthma for which he takes a rescue inhaler infrequently prior to transplantation. His pretransplant PFTs showed normal spirometry with an FEV1 of 106% of predicted and D
Physical exam is notable for fever of 101.0°F, heart rate 80 beats/min, respiratory rate 16 breaths/ min, and blood pressure 142/78 mm Hg; an admission oxygen saturation is 93% on room air. Lungs show bibasilar crackles and the remainder of the exam is normal. Laboratory testing shows a white blood cell count of 2400 cells/μL, hemoglobin 7.6 g/dL, and platelet count 66 × 103/μL. Creatinine is 1.0 mg/dL. Chest radiograph shows ill-defined bilateral lower-lobe infiltrates. CT scans are shown in the Figure.
- For which infectious complications is this patient most at risk?
Pneumonia
A prospective trial in the HSCT population reported a pneumonia incidence rate of 68%, and pneumonia is more common in allogeneic HSCT with prolonged immunosuppressive therapy.32 Development of pneumonia within 100 days of transplant directly correlates with nonrelapsed mortality.33 Early detection is key, and bronchoscopy within the first 5 days of symptoms has been shown to change therapy in approximately 40% of cases but has not been shown to affect mortality.34 The clinical presentation of pneumonia in the HSCT population can be variable because of the presence of neutropenia and profound immunosuppression. Traditionally accepted diagnostic criteria of fevers, sputum production, and new infiltrates should be used with caution, and an appropriately high index of suspicion should be maintained. Progression to respiratory failure, regardless of causative organism of infection, portends a poor prognosis, with mortality rates estimated at 70% to 90%.35,36 Several transplant-specific factors may affect early infections. For instance, UCB transplants have been found to have a higher incidence of invasive aspergillosis and cytomegalovirus (CMV) infections but without higher mortality attributed to the infections.37
Bacterial Pneumonia
Bacterial pneumonia accounts for 20% to 50% of pneumonia cases in HSCT recipients.38 Gram-negative organisms, specifically Pseudomonas aeruginosa and Escherichia coli, were reported to be the most common pathologic bacteria in recent prospective trials, whereas previous retrospective trials showed that common community-acquired organisms were the most common cause of pneumonia in HSCT recipients.32,39 This underscores the importance of being aware of the clinical prevalence of microorganisms and local antibiograms, along with associated institutional susceptibility profiles. Initiation of immediate empiric broad-spectrum antibiotics is essential when bacterial pneumonia is suspected.
Viral Pneumonia
The prevalence of viral pneumonia in stem cell transplant recipients is estimated at 28%,32 with most cases being caused by community viral pathogens such as rhinovirus, respiratory syncytial virus (RSV), influenza A and B, and parainfluenza.39 The prevention, prophylaxis, and early treatment of viral pneumonias, specifically CMV infection, have decreased the mortality associated with early pneumonia after HSCT. Co-infection with bacterial organisms must be considered and has been associated with increased mortality in the intensive care unit setting.40
Supportive treatment with rhinovirus infection is sufficient as the disease is usually self-limited in immunocompromised patients. In contrast, infection with RSV in the lower respiratory tract is associated with increased mortality in prior reports, and recent studies suggest that further exploration of prophylaxis strategies is warranted.41 Treatment with ribavirin remains the backbone of therapy, but drug toxicity continues to limit its use. The addition of immunomodulators such as RSV immune globulin or palivizumab to ribavirin remains controversial, but a retrospective review suggests that early treatment may prevent progression to lower respiratory tract infection and lead to improved mortality.42 Infection with influenza A/B must be considered during influenza season. Treatment with oseltamivir may shorten the duration of disease when influenza A/B or parainfluenza are detected. Reactivation of latent herpes simplex virus during the pre-engraftment phase should also be considered. Treatment is similar to that in nonimmunocompromised hosts. When CMV pneumonia is suspected, careful history regarding compliance with prophylactic antivirals and CMV status of both the recipient and donor are key. A presumptive diagnosis can be made with the presence of appropriate clinical scenario, supportive radiographic images showing areas of ground-glass opacification or consolidation, and positive CMV polymerase chain reaction (PCR) assay. Visualization of inclusion bodies on lung biopsy tissue remains the gold standard for diagnosis. Treatment consists of CMV immunoglobulin and ganciclovir.
Fungal Pneumonia
Early fungal pneumonias have been associated with increased mortality in the HSCT population.43 Clinical suspicion should remain high and compliance with antifungal prophylaxis should be questioned thoroughly. Invasive aspergillosis (IA) remains the most common fungal infection. A bimodal distribution of onset of infection peaking on day 16 and again on day 96 has been described in the literature.44 Patients often present with classic pneumonia symptoms, but these may be accompanied by hemoptysis. Proven IA diagnosis requires visualization of fungal forms from biopsy or needle aspiration or a positive culture obtained in a sterile fashion.45 Most clinical data comes from experience with probable and possible diagnosis of IA. Bronchoalveolar lavage with testing with Aspergillus galactomannan assay has been shown to be clinically useful in establishing the clinical diagnosis in the HSCT population.46 Classic air-crescent findings on chest CT are helpful in establishing a possible diagnosis, but retrospective analysis reveals CT findings such as focal infiltrates and pulmonary nodular patterns are more common.47 First-line treatment with voriconazole has been shown to decrease short-term mortality attributable to IA but has not had an effect on long-term, all-cause mortality.48 Surgical resection is reserved for patients with refractory disease or patients presenting with massive hemoptysis.
Mucormycosis is an emerging disease with ever increasing prevalence in the HSCT population, reflecting the improved prophylaxis and treatment of IA. Initial clinical presentation is similar to IA, most commonly affecting the lung, although craniofacial involvement is classic for mucormycosis, especially in HSCT patients with diabetes.49Mucor infections can present with massive hemoptysis due to tissue invasion and disregard for tissue and fascial planes. Diagnosis of mucormycosis is associated with as much as a six-fold increase in risk for death. Diagnosis requires identification of the organism by examination or culture and biopsy is often necessary.50,51 Amphotericin B remains first-line therapy as mucormycosis is resistant to azole antifungals, with higher doses recommended for cerebral involvement.52
Candida pulmonary infections during the early HSCT period are becoming increasingly rare due to widespread use of fluconazole prophylaxis and early treatment of mucosal involvement during neutropenia. Endemic fungal infections such as blastomycosis, coccidioidomycosis, and histoplasmosis should be considered in patients inhabiting specific geographic areas or with recent travel to these areas.
- What test should be performed to evaluate for infectious causes of pneumonia?
Role of Flexible Fiberoptic Bronchoscopy
The utility of flexible fiberoptic bronchoscopy (FOB) in immune-compromised patients for the evaluation of pulmonary infiltrates is a frequently debated topic. Current studies suggest a diagnosis can be made in approximately 80% of cases in the immune-compromised population.32,53 Noninvasive testing such as urine and serum antigens, sputum cultures, Aspergillus galactomannan assays, viral nasal swabs, and PCR studies often lead to a diagnosis in appropriate clinical scenarios. Conservative management would dictate the use of noninvasive testing whenever possible, and randomized controlled trials have shown noninvasive testing to be noninferior to FOB in preventing need for mechanical ventilation, with no difference in overall mortality.54 FOB has been shown to be most useful in establishing a diagnosis when an infectious etiology is suspected.55 In multivariate analysis, a delay in the identification of the etiology of pulmonary infiltrate was associated with increased mortality.56 Additionally, early FOB was found to be superior to late FOB in revealing a diagnosis. 32,57 Despite its ability to detect the cause of pulmonary disease, direct antibiotic therapy, and possibly change therapy, FOB with diagnostic maneuvers has not been shown to affect mortality.58 In a large case series, FOB with bronchoalveolar lavage (BAL) revealed a diagnosis in approximately 30% to 50% of cases. The addition of transbronchial biopsy did not improve diagnostic utility.58 More recent studies have confirmed that the addition of transbronchial biopsy does not add to diagnostic yield and is associated with increased adverse events.59 The appropriate use of advanced techniques such as endobronchial ultrasound–guided transbronchial needle aspirations, endobronchial biopsy, and CT-guided navigational bronchoscopy has not been established and should be considered on a case-by-case basis. In summary, routine early BAL is the diagnostic test of choice, especially when infectious pulmonary complications are suspected.
Contraindications for FOB in this population mirror those in the general population. These include acute severe hypoxemic respiratory failure, myocardial ischemia or acute coronary syndrome within 2 weeks of procedure, severe thrombocytopenia, and inability to provide or obtain informed consent from patient or health care power of attorney. Coagulopathy and thrombocytopenia are common comorbid conditions in the HSCT population. A platelet count of < 20 × 103/µL has generally been used as a cut-off for routine FOB with BAL.60 Risks of the procedures should be discussed clearly with the patient, but simple FOB for airway evaluation and BAL is generally well tolerated even under these conditions.
Early Nonifectious Pulmonary Complications
Case Patient 2 Continued
Bronchoscopy with BAL performed the day after admission is unremarkable and stains and cultures are negative for viral, bacterial, and fungal organisms. The patient is initially started on broad-spectrum antibiotics, but his oxygenation continues to worsen to the point that he is placed on noninvasive positive pressure ventilation. He is started empirically on amphotericin B and eventually is intubated. VATS lung biopsy is ultimately performed and pathology is consistent with diffuse alveolar damage.
- Based on these biopsy findings, what is the diagnosis?
Based on the pathology consistent with diffuse alveolar damage, a diagnosis of idiopathic pneumonia syndrome (IPS) is made.
- What noninfectious pulmonary complications occur in the early post-transplant period?
The overall incidence of noninfectious pulmonary complications after HSCT is generally estimated at 20% to 30%.32 Acute pulmonary edema is a common very early noninfectious pulmonary complication and clinically the most straightforward to treat. Three distinct clinical syndromes—peri-engraftment respiratory distress syndrome (PERDS), diffuse alveolar hemorrhage (DAH), and IPS—comprise the remainder of the pertinent early noninfectious complications. Clinical presentation differs based upon the disease entity. Recent studies have evaluated the role of angiotensin-converting enzyme polymorphisms as a predictive marker for risk of developing early noninfectious pulmonary complications.61
Peri-Engraftment Respiratory Distress Syndrome
PERDS is a clinical syndrome comprising the cardinal features of erythematous rash and fever along with noncardiogenic pulmonary infiltrates and hypoxemia that occur in the peri-engraftment period, defined as recovery of absolute neutrophil count to > 500/μL on 2 consecutive days.62 PERDS occurs in the autologous HSCT population and may be a clinical correlate to early GVHD in the allogeneic HSCT population. It is hypothesized that the pathophysiology underlying PERDS is an autoimmune-related capillary leak caused by pro-inflammatory cytokine release.63 Treatment remains anecdotal and currently consists of supportive care and high-dose corticosteroids. Some have favored limiting the use of gCSF given its role in stimulating rapid white blood cell recovery.33 Prognosis is favorable, but progression to fulminant respiratory failure requiring mechanical ventilation portends a poor prognosis.
Diffuse Alveolar Hemorrhage
DAH is clinical syndrome consisting of diffuse alveolar infiltrates on pulmonary imaging combined with progressively bloodier return per aliquot during BAL in 3 different subsegments or more than 20% hemosiderin-laden macrophages on BAL fluid evaluation. Classically, DAH is defined in the absence of pulmonary infection or cardiac dysfunction. The pathophysiology is thought to be related to inflammation of pulmonary vasculature within the alveolar walls leading to alveolitis. Although no prospective trials exist, early use of high-dose corticosteroid therapy is thought to improve outcomes;64,65 a recent study, however, showed low-dose steroids may be associated with the lowest mortality.66 Mortality is directly linked to the presence of superimposed infection, need for mechanical ventilation, late onset, and development of multiorgan failure.67
Idiopathic Pneumonia Syndrome
IPS is a complex clinical syndrome whose pathology is felt to stem from a variety of possible lung insults such as direct myeloablative drug toxicity, occult pulmonary infection, or cytokine-driven inflammation. The ATS published an article further subcategorizing IPS as different clinical entities based upon whether the primary insult involves the vascular endothelium, interstitial tissue, and airway tissue, truly idiopathic, or unclassified.68 In clinical practice, IPS is defined as widespread alveolar injury in the absence of evidence of renal failure, heart failure, and excessive fluid resuscitation. In addition, negative testing for a variety of bacterial, viral, and fungal causes is also necessary.69 Clinical syndromes included within the IPS definition are ARDS, acute interstitial pneumonia, DAH, cryptogenic organizing pneumonia, and BOS.70 Risk factors for developing IPS include TBI, older age of recipient, acute GVHD, and underlying diagnosis of AML or myelodysplastic syndrome.12 In addition, it has been shown that risk for developing IPS is lower in patients undergoing allogeneic HSCT who receive non-myeloablative conditioning regimens.71 The pathologic finding in IPS is diffuse alveolar damage. A 2006 study in which investigators reviewed BAL samples from patients with IPS found that 3% of the patients had PCR evidence of human metapneumovirus infection, and a study in 2015 found PCR evidence of infection in 53% of BAL samples from patients diagnosed with IPS.72,73 This fuels the debate on whether IPS is truly an infection-driven process where the source of infection, pulmonary or otherwise, simply escapes detection. Various surfactant proteins, which play a role in decreasing surface tension within the alveolar interface and function as mediators within the innate immunity of the lung, have been studied in regard to development of IPS. Small retrospective studies have shown a trend toward lower pre-transplant serum protein surfactant D and the development of IPS.74
The diagnosis of IPS does not require pathologic diagnosis in most circumstances. The correct clinical findings in association with a negative infectious workup lead to a presumptive diagnosis of IPS. The extent of the infectious workup that must be completed to adequately rule out infection is often a difficult clinical question. Recent recommendations include BAL fluid evaluation for routine bacterial cultures, appropriate viral culture, and consideration of PCR testing to evaluate for Mycoplasma, Chlamydia, and Aspergillus antigens.75 Transbronchial biopsy continues to appear in recommendations, but is not routinely performed and should be completed as the patient’s clinical status permits.8,68 Table 3 reviews basic features of early noninfectious pulmonary complications.
Treatment of IPS centers around moderate to high doses of corticosteroids. Based on IPS experimental modes, tumor necrosis factor (TNF)-α has been implicated as an important mediator. Unfortunately, several studies evaluating etanercept have produced conflicting results, and this agent’s clinical effects on morbidity and mortality remain in question.76
- What treatment should be offered to the patient with diffuse alveolar damage on biopsy?
Treatment consists of supportive care and empiric broad-spectrum antibiotics with consideration of high-dose corticosteroids. Based upon early studies in murine models implicating TNF, pilot studies were performed evaluating etanercept as a possible safe and effective addition to high-dose systemic corticosteroids.77 Although these results were promising, data from a truncated randomized control clinical trial failed to show improvement in patient response in the adult population.76 More recent data from the same author suggests that pediatric populations with IPS are, however, responsive to etanercept and high-dose corticosteroid therapy.78 When IPS develops as a late complication, treatment with high-dose corticosteroids (2 mg/kg/day) and etanercept (0.4 mg/kg twice weekly) has been shown to improve 2-year survival.79
Case Patient 2 Conclusion
The patient is started on steroids and makes a speedy recovery. He is successfully extubated 5 days later.
Conclusion
Careful pretransplant evaluation, including a full set of pulmonary function tests, can help predict a patient’s risk for pulmonary complications after transplant, allowing risk factor modification strategies to be implemented prior to transplant, including smoking cessation. It also helps identify patients at high risk for complications who will require closer monitoring after transplantation. Early posttransplant complications include infectious and noninfectious entities. Bacterial, viral, and fungal pneumonias are in the differential of infectious pneumonia, and bronchoscopy can be helpful in establishing a diagnosis. A common, important noninfectious cause of early pulmonary complications is IPS, which is treated with steroids and sometimes anti-TNF therapy.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
1. Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA 2010;303:1617–24.
2. Kotloff RM, Ahya VN, Crawford SW. Pulmonary complications of solid organ and hematopoietic stem cell transplantation. Am J Respir Crit Care Med 2004;170:22–48.
4. Copelan EA. Hematopoietic stem-cell transplantation. N Engl J Med 2006;354:1813–26.
5. Anasetti C, Logan BR, Lee SJ, et al. Peripheral-blood stem cells versus bone marrow from unrelated donors. N Engl J Med 2012;367:1487–96.
6. Giralt S, Ballen K, Rizzo D, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant 2009;15:367–9.
7. Shulman HM, Kleiner D, Lee SJ, et al. Histopathologic diagnosis of chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: II. Pathology Working Group Report. Biol Blood Marrow Transplant 2006;12:31–47.
8. Afessa B, Abdulai RM, Kremers WK, et al. Risk factors and outcome of pulmonary complications after autologous hematopoietic stem cell transplant. Chest 2012;141:442–50.
9. Bolwell BJ. Are predictive factors clinically useful in bone marrow transplantation? Bone Marrow Transplant 2003;32:853–61.
10. Carlson K, Backlund L, Smedmyr B, et al. Pulmonary function and complications subsequent to autologous bone marrow transplantation. Bone Marrow Transplant 1994;14:805–11.
11. Clark JG, Schwartz DA, Flournoy N, et al. Risk factors for airflow obstruction in recipients of bone marrow transplants. Ann Intern Med 1987;107:648–56.
12. Crawford SW, Fisher L. Predictive value of pulmonary function tests before marrow transplantation. Chest 1992; 101:1257–64.
13. Ghalie R, Szidon JP, Thompson L, et al. Evaluation of pulmonary complications after bone marrow transplantation: the role of pretransplant pulmonary function tests. Bone Marrow Transplant 1992;10:359–65.
14. Ho VT, Weller E, Lee SJ, et al. Prognostic factors for early severe pulmonary complications after hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2001;7:223–9.
15. Horak DA, Schmidt GM, Zaia JA, et al. Pretransplant pulmonary function predicts cytomegalovirus-associated interstitial pneumonia following bone marrow transplantation. Chest 1992;102:1484–90.
16. Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, et al. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transplant 2010;16:199–206.
17. White AC, Terrin N, Miller KB, Ryan HF. Impaired respiratory and skeletal muscle strength in patients prior to hematopoietic stem-cell transplantation. Chest 2005;128145–52.
18. Afessa B. Pretransplant pulmonary evaluation of the blood and marrow transplant recipient. Chest 2005;128:8–10.
19. Parimon T, Madtes DK, Au DH, et al. Pretransplant lung function, respiratory failure, and mortality after stem cell transplantation. Am J Respir Crit Care Med 2005;172:384–90.
20. Pavletic SZ, Martin P, Lee SJ, et al. Measuring therapeutic response in chronic graft-versus-host disease: National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease: IV. Response Criteria Working Group report. Biol Blood Marrow Transplant 2006;12:252–66.
21. Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med 2006;144:407–14.
22. Au BK, Gooley TA, Armand P, et al. Reevaluation of the pretransplant assessment of mortality score after allogeneic hematopoietic transplantation. Biol Blood Marrow Transplant 2015;21:848–54.
23. Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005;106:2912–9.
24. Chien JW, Sullivan KM. Carbon monoxide diffusion capacity: how low can you go for hematopoietic cell transplantation eligibility? Biol Blood Marrow Transplant 2009;15: 447–53.
25. Coffey DG, Pollyea DA, Myint H, et al. Adjusting DLCO for Hb and its effects on the Hematopoietic Cell Transplantation-specific Comorbidity Index. Bone Marrow Transplant 2013;48:1253–6.
26. Kasow KA, Krueger J, Srivastava DK, et al. Clinical utility of computed tomography screening of chest, abdomen, and sinuses before hematopoietic stem cell transplantation: the St. Jude experience. Biol Blood Marrow Transplant 2009;15:490–5.
27. Hamadani M, Craig M, Awan FT, Devine SM. How we approach patient evaluation for hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45: 1259–68.
28. Savani BN, Montero A, Wu C, et al. Prediction and prevention of transplant-related mortality from pulmonary causes after total body irradiation and allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:223–30.
29. Ehlers SL, Gastineau DA, Patten CA, et al. The impact of smoking on outcomes among patients undergoing hematopoietic SCT for the treatment of acute leukemia. Bone Marrow Transplant 2011;46:285–90.
30. Marks DI, Ballen K, Logan BR, et al. The effect of smoking on allogeneic transplant outcomes. Biol Blood Marrow Transplant 2009;15:1277–87.
31. Tran BT, Halperin A, Chien JW. Cigarette smoking and outcomes after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2011;17:1004–11.
32. Lucena CM, Torres A, Rovira M, et al. Pulmonary complications in hematopoietic SCT: a prospective study. Bone Marrow Transplant 2014;49:1293–9.
33. Chi AK, Soubani AO, White AC, Miller KB. An update on pulmonary complications of hematopoietic stem cell transplantation. Chest 2013;144:1913–22.
34. Dunagan DP, Baker AM, Hurd DD, Haponik EF. Bronchoscopic evaluation of pulmonary infiltrates following bone marrow transplantation. Chest 1997;111:135–41.
35. Naeem N, Reed MD, Creger RJ, et al. Transfer of the hematopoietic stem cell transplant patient to the intensive care unit: does it really matter? Bone Marrow Transplant 2006;37:119–33.
36. Afessa B, Tefferi A, Hoagland HC, et al. Outcome of recipients of bone marrow transplants who require intensive care unit support. Mayo Clin Proc 1992;67:117–22.
37. Parody R, Martino R, de la Camara R, et al. Fungal and viral infections after allogeneic hematopoietic transplantation from unrelated donors in adults: improving outcomes over time. Bone Marrow Transplant 2015;50:274–81.
38. Orasch C, Weisser M, Mertz D, et al. Comparison of infectious complications during induction/consolidation chemotherapy versus allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:521–6.
39. Aguilar-Guisado M, Jimenez-Jambrina M, Espigado I, et al. Pneumonia in allogeneic stem cell transplantation recipients: a multicenter prospective study. Clin Transplant 2011;25:E629–38.
40. Palacios G, Hornig M, Cisterna D, et al. Streptococcus pneumoniae coinfection is correlated with the severity of H1N1 pandemic influenza. PLoS One 2009;4:e8540.
41. Hynicka LM, Ensor CR. Prophylaxis and treatment of respiratory syncytial virus in adult immunocompromised patients. Ann Pharmacother 2012;46:558–66.
42. Shah JN, Chemaly RF. Management of RSV infections in adult recipients of hematopoietic stem cell transplantation. Blood 2011;2755–63.
43. Marr KA, Bowden RA. Fungal infections in patients undergoing blood and marrow transplantation. Transpl Infect Dis 1999;1:237–46.
44. Wald A, Leisenring W, van Burik JA, Bowden RA. Epidemiology of Aspergillus infections in a large cohort of patients undergoing bone marrow transplantation. J Infect Dis 1997;175:1459–66.
45. Ascioglu S, Rex JH, de Pauw B, et al. Defining opportunistic invasive fungal infections in immunocompromised patients with cancer and hematopoietic stem cell transplants: an international consensus. Clin Infect Dis 2002;34:7–14.
46. Fisher CE, Stevens AM, Leisenring W, et al. Independent contribution of bronchoalveolar lavage and serum galactomannan in the diagnosis of invasive pulmonary aspergillosis. Transpl Infect Dis 2014;16:505–10.
47. Kojima R, Tateishi U, Kami M, et al. Chest computed tomography of late invasive aspergillosis after allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2005;11:506–11.
48. Salmeron G, Porcher R, Bergeron A, et al. Persistent poor long-term prognosis of allogeneic hematopoietic stem cell transplant recipients surviving invasive aspergillosis. Haematologica 2012;97:1357–63.
49. McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982;92(10 Pt 1):1140.
50. Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012;54 Suppl 1:S55–60.
51. Klingspor L, Saaedi B, Ljungman P, Szakos A. Epidemiology and outcomes of patients with invasive mould infections: a retrospective observational study from a single centre (2005-2009). Mycoses 2015;58:470–7.
52. Danion F, Aguilar C, Catherinot E, et al. Mucormycosis: new developments in a persistently devastating infection. Semin Respir Crit Care Med 2015;36:692–70.
53. Rano A, Agusti C, Jimenez P, et al. Pulmonary infiltrates in non-HIV immunocompromised patients: a diagnostic approach using non-invasive and bronchoscopic procedures. Thorax 2001;56:379–87.
54. Azoulay E, Mokart D, Rabbat A, et al. Diagnostic bronchoscopy in hematology and oncology patients with acute respiratory failure: prospective multicenter data. Crit Care Med 2008;36:100–7.
55. Jain P, Sandur S, Meli Y, et al. Role of flexible bronchoscopy in immunocompromised patients with lung infiltrates. Chest 2004;125:712–22.
56. Rano A, Agusti C, Benito N, et al. Prognostic factors of non-HIV immunocompromised patients with pulmonary infiltrates. Chest 2002;122:253–61.
57. Shannon VR, Andersson BS, Lei X, et al. Utility of early versus late fiberoptic bronchoscopy in the evaluation of new pulmonary infiltrates following hematopoietic stem cell transplantation. Bone Marrow Transplant 2010;45:647–55.
58. Patel NR, Lee PS, Kim JH, et al. The influence of diagnostic bronchoscopy on clinical outcomes comparing adult autologous and allogeneic bone marrow transplant patients. Chest 2005;127:1388–96.
59. Chellapandian D, Lehrnbecher T, Phillips B, et al. Bronchoalveolar lavage and lung biopsy in patients with cancer and hematopoietic stem-cell transplantation recipients: a systematic review and meta-analysis. J Clin Oncol 2015;33:501–9.
60. Carr IM, Koegelenberg CF, von Groote-Bidlingmaier F, et al. Blood loss during flexible bronchoscopy: a prospective observational study. Respiration 2012;84:312–8.
61. Miyamoto M, Onizuka M, Machida S, et al. ACE deletion polymorphism is associated with a high risk of non-infectious pulmonary complications after stem cell transplantation. Int J Hematol 2014;99:175–83.
62. Capizzi SA, Kumar S, Huneke NE, et al. Peri-engraftment respiratory distress syndrome during autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:1299–303.
63. Spitzer TR. Engraftment syndrome following hematopoietic stem cell transplantation. Bone Marrow Transplant 2001;27:893–8.
64. Wanko SO, Broadwater G, Folz RJ, Chao NJ. Diffuse alveolar hemorrhage: retrospective review of clinical outcome in allogeneic transplant recipients treated with aminocaproic acid. Biol Blood Marrow Transplant 2006;12:949–53.
65. Metcalf JP, Rennard SI, Reed EC, et al. Corticosteroids as adjunctive therapy for diffuse alveolar hemorrhage associated with bone marrow transplantation. University of Nebraska Medical Center Bone Marrow Transplant Group. Am J Med 1994;96:327–34.
66. Rathi NK, Tanner AR, Dinh A, et al. Low-, medium- and high-dose steroids with or without aminocaproic acid in adult hematopoietic SCT patients with diffuse alveolar hemorrhage. Bone Marrow Transplant 2015;50:420–6.
67. Afessa B, Tefferi A, Litzow MR, Peters SG. Outcome of diffuse alveolar hemorrhage in hematopoietic stem cell transplant recipients. Am J Respir Crit Care Med 2002;166:1364–8.
68. Panoskaltsis-Mortari A, Griese M, Madtes DK, et al. An official American Thoracic Society research statement: noninfectious lung injury after hematopoietic stem cell transplantation: idiopathic pneumonia syndrome. Am J Respir Crit Care Med 2011;183:1262–79.
69. Clark JG, Hansen JA, Hertz MI, Pet al. NHLBI workshop summary. Idiopathic pneumonia syndrome after bone marrow transplantation. Am Rev Resp Dis 1993;147:1601–6.
70. Vande Vusse LK, Madtes DK. Early onset noninfectious pulmonary syndromes after hematopoietic cell transplantation. Clin Chest Med 2017;38:233–48.
71. Fukuda T, Hackman RC, Guthrie KA, et al. Risks and outcomes of idiopathic pneumonia syndrome after nonmyeloablative and conventional conditioning regimens for allogeneic hematopoietic stem cell transplantation. Blood 2003;102:2777–85.
72. Englund JA, Boeckh M, Kuypers J, et al. Brief communication: fatal human metapneumovirus infection in stem-cell transplant recipients. Ann Intern Med 2006;144:344–9.
73. Seo S, Renaud C, Kuypers JM, et al. Idiopathic pneumonia syndrome after hematopoietic cell transplantation: evidence of occult infectious etiologies. Blood 2015;125:3789–97.
74. Nakane T, Nakamae H, Kamoi H, et al. Prognostic value of serum surfactant protein D level prior to transplant for the development of bronchiolitis obliterans syndrome and idiopathic pneumonia syndrome following allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008;42:43–9.
75. Gilbert CR, Lerner A, Baram M, Awsare BK. Utility of flexible bronchoscopy in the evaluation of pulmonary infiltrates in the hematopoietic stem cell transplant population—a single center fourteen year experience. Arch Bronconeumol 2013;49:189–95.
76. Yanik GA, Horowitz MM, Weisdorf DJ, et al. Randomized, double-blind, placebo-controlled trial of soluble tumor necrosis factor receptor: enbrel (etanercept) for the treatment of idiopathic pneumonia syndrome after allogeneic stem cell transplantation: blood and marrow transplant clinical trials network protocol. Biol Blood Marrow Transplant 2014;20:858–64.
77. Levine JE, Paczesny S, Mineishi S, et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008;111:2470–5.
78. Yanik GA, Grupp SA, Pulsipher MA, et al. TNF-receptor inhibitor therapy for the treatment of children with idiopathic pneumonia syndrome. A joint Pediatric Blood and Marrow Transplant Consortium and Children’s Oncology Group Study (ASCT0521). Biol Blood Marrow Transplant 2015;21:67–73.
79. Thompson J, Yin Z, D’Souza A, et al. Etanercept and corticosteroid therapy for the treatment of late-onset idiopathic pneumonia syndrome. Biol Blood Marrow Transplant J 2017; 23:1955–60.
A Primary Care Provider’s Guide to Cataract Surgery in the Very Elderly
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
Cataract surgery is the most commonly performed surgical procedure in the US, including within the Veterans Health Administration (VHA).1,2 As the risk of surgical complications has decreased with improved techniques and instrumentation, the threshold for performing surgery has lowered.3 A substantial number of patients do not develop clinically significant cataracts until they are “very elderly,” defined as aged ≥ 85 years by the World Health Organization and National Institute of Aging.4
Should the general approach to cataract evaluation and surgery differ in this subset of patients? Advanced age is associated with a variety of systemic and ocular comorbidities that theoretically increase the risk of cataract surgery and reduce the potential visual benefit it might yield. However, the impact of age on the outcomes of cataract surgery differs even among the very elderly. There are no universally acknowledged guidelines that address the perioperative evaluation and management of cataracts in the very elderly, whose systemic and ocular health have greater variability than those of their younger counterparts. For very elderly patients who are found to have visually significant cataracts by their ophthalmologists, input from the primary care provider (PCP), who has insight into a patient’s health and well-being, is vital for formulating a management plan. Herein, we provide a framework for PCPs to assist very elderly patients and their ophthalmologists in making an informed decision regarding cataract surgery and in planning for perioperative care.
Cataract Surgery
Cataract surgeons recommend surgical extraction when there is a clinically significant lens opacity that imposes functional impairment, such as inability to read, perform near work, watch television, or drive.4 The standard of care for a clinically significant cataract is surgical removal of the crystalline lens and replacement with an artificial intraocular lens (IOL). At times, the onset of vision loss from a cataract is insidious such that patients may not be aware of their declining vision or the deterioration in quality of life (QOL) that it causes.
Despite the higher burden of ocular comorbidity (eg, age-related macular degeneration, glaucoma) relative to their younger counterparts, most very elderly patients obtain functionally important improvement in their vision, QOL, and cognitive function after surgery.5-16 Cataract surgery can also reduce the risk of dementia and the risk of falls and hip fractures.6,9,12-14,16-18 Ophthalmic complications of cataract surgery in the very elderly include posterior capsule tear (< 1%-9%), vitreous loss (< 1%-8%), zonular rupture (2%-5%), and retained lens fragments (≤ 1%).5,8-11,17,19-21 There is no evidence from well-controlled studies that suggests that very elderly cataract surgery patients are at higher risk of ocular complications relative to that of their younger counterparts.22
Surgery Alternatives
In some very elderly patients, cataract surgery may not be the best option, and PCPs can aid in establishing an alternative plan. Such patients include those with a limited life expectancy, incapacitating anxiety over surgery, or those in whom the potential for visual improvement is marginal because of ocular or systemic comorbidities—eg, vision-limiting glaucoma or age-related macular degeneration, history of stroke to the visual pathway, or restriction to bed. Alternatives to cataract surgery in these instances include changing environmental conditions to improve visual function, such as enhanced lighting and contrast, and/or use of low-vision aids (referring patients to low-vision professionals often improves QOL).23 Low-vision specialists also have a variety of nonvisual aids that can expand functional capabilities: large-print and talking versions of reading materials, telephones, remote controls, clocks, scales, calculators, and glucose monitors; glare-free lights for stairs, floors, and counters; and specialty glasses that use light-emitting diode screens and live video streams to magnify sight.23-25
Medical Evaluation
For patients who decide to proceed with surgery, it can be helpful to have a medical evaluation by their PCPs to minimize potential complications during surgery. The very elderly may be at increased risk of intraoperative transient hypertension, restlessness, and electrocardiogram abnormalities.5,7,17 Systemic comorbidities that become more prevalent with age, such as diabetes mellitus (DM), hypertension, heart disease, chronic obstructive pulmonary disease, and dementia, may adversely impact the risk of sedation and/or general anesthesia. In the VHA, providers also must be aware of combat-related disorders that can confound cataract surgery, such as posttraumatic stress disorder (PTSD), anxiety, and claustrophobia.26,27
Anesthesia in cataract surgery ranges from topical to general, and the selection largely rests on patient physical and psychological comfort and cooperation. Often, intracameral (inside the anterior eye) anesthetic is used with topical anesthesia to provide additional comfort.27 Patients who have high levels of anxiety about surgery may not tolerate topical anesthesia alone.28 In these cases, retrobulbar anesthesia may be performed to block all sensation and motility of the eye. IV sedation is performed prior to the retrobulbar injection to calm patients. Although cataract surgery is typically performed with topical or retrobulbar anesthesia (reducing the potential for systemic complications), there are cases in which general anesthesia may be considered.27 Very elderly patients may become confused or disoriented in the operating room (OR), leading to surgical complications and less than optimal outcomes.5 A higher rate of intraoperative “restlessness,” which occurred in patients who had comorbid dementia, and transient hypertension were found in a study on cataract surgery in the very elderly, but well-controlled studies are lacking.5 Dementia can impose problems with intraoperative cooperation, which is vital for successful surgery in patients who undergo topical or local anesthesia. If these potential problems are thought likely preoperatively, light sedation or general anesthesia—in conjunction with input from the patient’s PCP—are options to minimize disruptive behavior in the OR.
Additional features of the VHA population may influence the selection of anesthesia. The VHA has an important educational mission, and retrobulbar anesthesia may be preferred to minimize unpredictable intraoperative behavior in cases where resident surgeons are performing surgery under attending supervision.27,29,30 The prevalence of PTSD among veterans also may impact the selection of anesthesia. Patients with PTSD have displayed greater levels of anxiety and more discomfort, requiring more sedation and longer surgical times compared with that of a control group.28 Ophthalmic comorbidities prevalent among the predominantly older male population in the VHA include the use of α-1 antagonist prostate medications, such as tamsulosin and terazosin. These medications are associated with intraoperative floppy iris syndrome, which can increase case difficulty and prolong operative time.29
Surgery Preparation
Cataract surgery induces minimal physiologic stress since most surgeries are performed under local or topical anesthesia. Unless the preoperative medical history or physical examination detects an active or unattended medical condition that needs to be addressed, preoperative laboratory testing is generally not required.31-33 Current general guidelines for preoperative testing for cataract surgery exist but do not address specific issues facing very elderly patients. The American Academy of Ophthalmology advises against preoperative medical tests for eye surgery unless there are medical indications: an electrocardiogram for patients with a history of heart disease, a blood glucose test for those with DM, and a potassium test for patients who are on diuretics.31 The direct correlation of age with these comorbidities may translate into higher rates of preoperative testing among very elderly patients. In the VHA, 45% of ophthalmology services studied routinely performed preoperative electrocardiography, chemical analysis, and complete blood counts prior to performing cataract surgery.27 Patients who live with chronic bacterial colonization from indwelling catheters, ostomies, or bed sores need to be given instructions for proper hygienic practices to minimize risks of postoperative infection.34
Some patients undergoing cataract surgery may not be candidates for topical or local anesthesia alone. Sedation is often used to reduce anxiety and discomfort of surgery, but very elderly patients have narrower margins of therapeutic safety because of advanced aged or medical comorbidities. Since patients need to follow basic commands in the OR for ideal surgical execution, general anesthesia may need to be considered for those with dementia, deafness, anxiety attacks, or language barriers.
Postsurgical Care
Although cataract surgery is a less invasive procedure than it was in the past, full postoperative recovery regularly spans a month. During this time, proper healing relies on the regular administration of eye drops and a refrain from heavy lifting, straining, and eye rubbing. Very elderly patients may need varying degrees of assistance with postsurgical care. For example, adherence to the regimen of eye drops can be complicated by decreased dexterity from arthritis and difficulty remembering the administration schedule in some patients. Reliable transportation also is an important factor as patients are routinely scheduled for postoperative visits at the 1- day, 1-week, and 1-month mark. PCPs can assist in ensuring patients have prearranged assistance for eye care and transportation to and from appointments. Additionally, very elderly patients with a history of constipation may benefit from stool softeners and/or laxatives to help prevent straining.
Conclusion
The limited literature on clinical outcomes of cataract surgery in the very elderly indicates that most have successful surgery and improved postoperative QOL.22 Much of the benefits derived from cataract surgery in the very elderly can be ascribed to thoughtful preoperative evaluation and planning with the PCP.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
1. US Census Bureau. An aging nation: the older population in the United States. https://www.census.gov/library/publications/2014/demo/p25-1140.html Published May 2014. Accessed March 18, 2019.
2. VA Office of Inspector General. Healthcare inspection: evaluation of cataract surgeries and outcomes in veterans health administration facilities. Report No. 11-02487-158. https://www.va.gov/oig/pubs/vaoig-11-02487-158.pdf. Published March 28, 2013. Accessed March 11, 2019.
3. Lee CM, Afshari NA. The global state of cataract blindness. Curr Opin Ophthalmol. 2017;28(1):98-103.
4. American Academy of Ophthalmology. Cataract in the adult eye preferred practice pattern—2016. https://www.aao.org/preferred-practice-pattern/cataract-in-adult-eye-ppp-2016. Published October 2016. Accessed March 19, 2019.
5. Mutoh T, Isome S, Matsumoto Y, Chikuda M. Cataract surgery in patients older than 90 years of age. Can J Ophthalmol. 2012;47(2):140-144.
6. Monestam E, Wachmeister L. Impact of cataract surgery on the visual ability of the very old. Am J Ophthalmol. 2004;137(1):145-155.
7. Lai FH, Lok JY, Chow PP, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
8. Michalska-Malecka K, Nowak M, Gos´ciniewicz P, et al. Results of cataract surgery in the very elderly population. Clin Interv Aging. 2013;8:1041-1046.
9. Syam PP, Eleftheriadis H, Casswell AG, Brittain GP, McLeod BK, Liu CS. Clinical outcome following cataract surgery in very elderly patients. Eye (Lond). 2004;18(1):59-62.
10. Rosen E, Rubowitz A, Assia EI. Visual outcome following cataract extraction in patients aged 90 years and older. Eye (Lond). 2009;23(5):1120-1124.
11. Mehmet B, Abuzer G. Results of cataract surgery in the very elderly population. J Optom. 2009;2(3):138-141.
12. To KG, Meuleners L, Bulsara M, et al. A longitudinal cohort study of the impact of first- and both-eye cataract surgery on falls and other injuries in Vietnam. Clin Interv Aging. 2014;9:743-751.
13. Song E, Sun H, Xu Y, Ma Y, Zhu H, Pan CW. Age-related cataract, cataract surgery and subsequent mortality: a systematic review and meta-analysis. PLoS One. 2014;9(11):e112054.
14. Brannan S, Dewar C, Sen J, Clarke D, Marshall T, Murray PI. A prospective study of the rate of falls before and after cataract surgery. Br J Ophthalmol. 2003;87(5):560-562.
15. Jefferis JM, Mosimann UP, Clarke MP. Cataract and cognitive impairment: a review of the literature. Br J Ophthalmol. 2011;95(1):17-23.
16. Yu WK, Chen YT, Wang SJ, Kuo SC, Shia BC, Liu CJ. Cataract surgery is associated with a reduced risk of dementia: a nationwide population-based cohort study. Eur J Neurol. 2015;22(10):1370-1377, e1379-1380.
17. Tseng VL, Greenberg PB, Wu WC, et al. Cataract surgery complications in nonagenarians. Ophthalmology. 2011;118(7):1229-1235.
18. Jefferis JM, Clarke MP, Taylor JP. Effect of cataract surgery on cognition, mood, and visual hallucinations in older adults. J Cataract Refract Surg. 2015;41(6):1241-1247.
19. Celebi AR. The relationship between age and the intraoperative complication rate during phacoemulsification surgery. Aging Clin Exp Res. 2014;26(2):177-181.
20. Berler DK. Intraoperative complications during cataract surgery in the very old. Trans Am Ophthalmol Soc. 2000;98:127-130; discussion 130-132.
21. Lai FHP, Lok JYC, Chow PPC, Young AL. Clinical outcomes of cataract surgery in very elderly adults. J Am Geriatr Soc. 2014;62(1):165-170.
22. Li E, Margo CE, Greenberg PB. Cataract surgery outcomes in the very elderly. J Cataract Refract Surg. 2018;44(9):1144-1149.
23. Young JS. Age-related eye diseases and recommendations for low-vision AIDS. Home Healthc Now. 2015;33(1):10-17; quiz 18-19.
24. Virgili G, Acosta R, Grover LL, Bentley SA, Giacomelli G. Reading aids for adults with low vision. Cochrane Database Syst Rev. 2013;(10):CD003303.
25. Young JS. Age-related eye diseases: a review of current treatment and recommendations for low-vision aids. Home Healthc Nurse. 2008;26(8):464-471; quiz 472-473.
26. Thomas MM, Harpaz-Rotem I, Tsai J, Southwick SM, Pietrzak RH. Mental and physical health conditions in US combat veterans: results from the National Health and Resilience in Veterans study. Prim Care Companion CNS Disord. 2017;19(3):17m02118.
27. Havnaer AG, Greenberg PB, Cockerham GC, Clark MA, Chomsky A. Cataract surgery practices in the United States Veterans Health Administration. J Cataract Refract Surg. 2017;43(4):543-551.
28. Rapoport Y, Wayman LL, Chomsky AS. The effect of post-traumatic-stress-disorder on intra-operative analgesia in a veteran population during cataract procedures carried out using retrobulbar or topical anesthesia: a retrospective study. BMC Ophthalmol. 2017;17(1):85.
29. Payal AR, Gonzalez-Gonzalez LA, Chen X, et al. Outcomes of cataract surgery with residents as primary surgeons in the Veterans Affairs Healthcare System. J Cataract Refract Surg. 2016;42(3):370-384.
30. US Department of Veterans Affairs. Mission of the office of academic affiliations. https://www.va.gov/oaa/oaa_mission.asp. Updated November 30, 2018. Accessed March 18, 2019.
31. American Academy of Ophthalmology. Choosing wisely: five things ophthalmologists and patients should question. https://www.aao.org/choosing-wisely. Published February 2013. Accessed March 18, 2019.
32. Martin SK, Cifu AS. Routine preoperative laboratory tests for elective surgery. JAMA. 2017;318(6):567-568.
33. Schein OD, Katz J, Bass EB, et al; Study of Medical Testing for Cataract Surgery. The value of routine preoperative medical testing before cataract surgery. N Engl J Med. 2000;342(3):168-175.
34. Margo CE. Asymptomatic bacteriuria and acute-onset endophthalmitis after cataract surgery. Can J Ophthalmol. 2015;50(4):e51-52.
35. Fukui K, Fujioka M, Yamasaki K, Yamakawa S, Matsuo H, Noguchi M. Risk factors for postoperative complications among the elderly after plastic surgery procedures performed under general anesthesia. Plast Surg Int. 2018:7053839.
Management of Castration-Resistant Prostate Cancer
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.
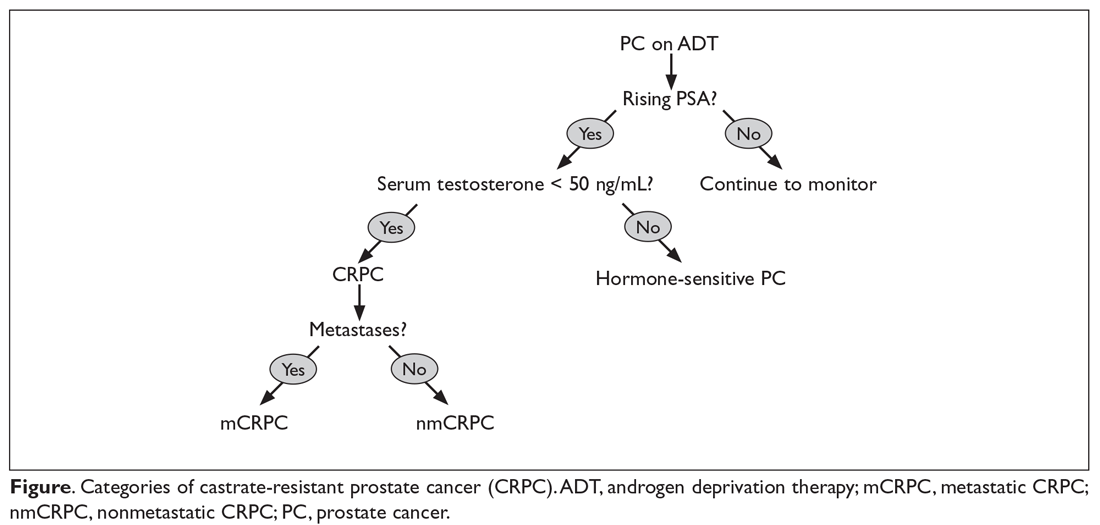
Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.
Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).
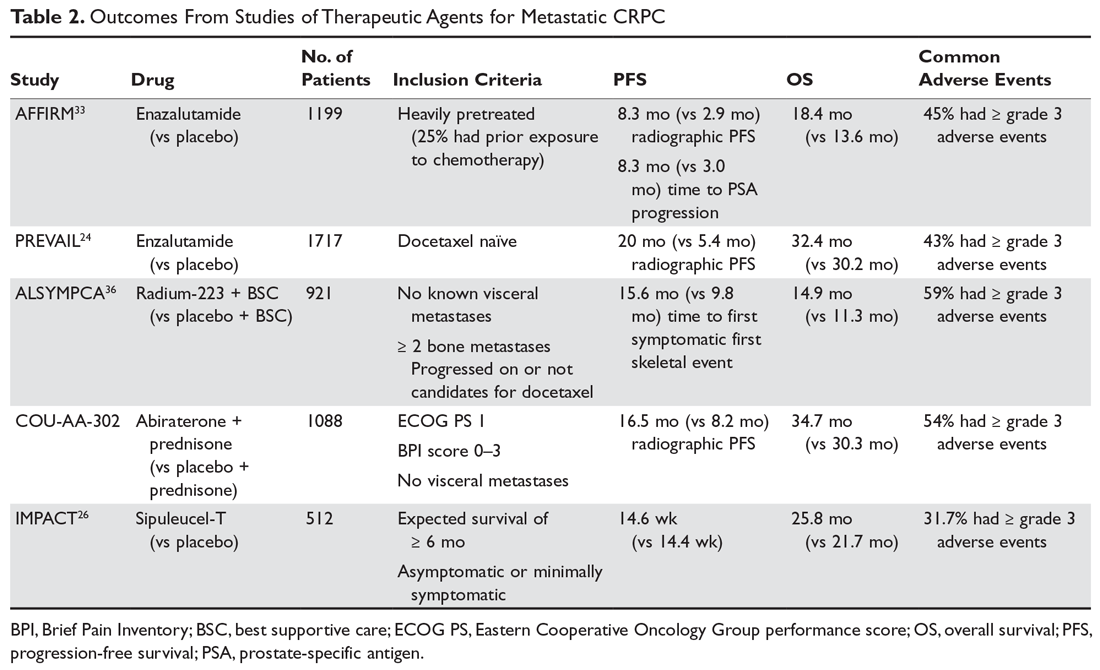
Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.

Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.

Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).

Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
Prostate cancer is the most common malignancy in men, with an estimated 165,000 new prostate cancer diagnoses and 29,000 prostate cancer deaths occurring in the United States in 2018.1 Due to the widespread use of screening prostate-specific antigen (PSA), prostate cancer has been mainly diagnosed when the tumor is confined to the prostate. Despite definitive treatment of localized prostate cancer, some men develop systemic disease, either biochemical failure, as defined by rising PSA level, or metastatic disease.1 Several factors have been demonstrated to predict risk of relapse, including higher pretreatment PSA, higher Gleason score, and a greater anatomic extent of disease.2 In addition, the incidence of de novo metastatic prostate cancer was recently noted to be increasing. This may be due to changes in the United States Preventive Services Task Force prostate cancer screening guidelines in 2012, which recommended against screening for prostate cancer in men of any age. The updated 2018 guidelines recommend a discussion of the risks versus benefits of screening for prostate cancer for all men aged 55 to 69 years,recommend against screening for men older than 70 years, and do not have recommendations for high-risk subgroups.3
Androgen deprivation therapy (ADT) has been the cornerstone of therapy since 1941 for men with hormone-sensitive systemic disease, both in biochemically relapsed and metastatic disease.4,5 While more than 90% of patients respond to initial ADT, castration resistance is inevitable in some men.6,7 Prostate cancer will become castration-resistant typically after 18 to 24 months of ADT, with the majority of patients developing castration-resistant prostate cancer (CRPC) within 5 years of initiation of ADT.8
Pathogenesis
CRPC (previously called androgen independent prostate cancer) is defined as progression of disease despite serum total testosterone levels less than 50 ng/dL. CRPC is characterized by a rising PSA level and/or radiographic progression. One mechanism of castration resistance is genetic modification of the androgen receptor (AR), including increased expression of the wild-type AR.9 Alternatively, mutations of the steroid-binding domain may play a role in the development of castration resistance by allowing the AR to become activated by non-androgen steroid hormones or even paradoxically by antiandrogens. Studies suggest, however, that AR mutations may be seen in only 10% of prostate cancers that have developed castration resistance.10 The AR-V7 splice variant of the AR lacks an androgen binding site altogether, and may play an important role in castration resistance. In one study, the presence of this splice variant in circulating prostate cancer tumor cells predicted resistance to enzalutamide and abiraterone as well as poor outcomes.11 Intratumoral androgen synthesis also may play a role in the development of CRPC.12,13
CRPC can be broadly categorized into 2 categories, metastatic (mCRPC) and nonmetastatic (nmCRPC; Figure). The exact proportion of patients entering CRPC at a nonmetastatic stage (M0) is largely unknown.14 In one study of patients at the time of diagnosis of CRPC, ≥ 84% of patients were shown to have metastases.8 In this article, we review key aspects of management of CRPC, including selection of first- and second-line therapy, and briefly discuss upcoming clinical trials.

Treatment of Nonmetastatic CRPC (M0 Disease)
Early identification of M0 CRPC is important because patients with nonmetastatic CRPC are at risk for metastasis, as demonstrated by Smith and colleagues.15 In this study that evaluated data from patients with nmCRPC in the placebo group (n = 331) of a randomized controlled trial, at 2 years 46% had developed ≥ 1 bone metastasis, 20% had died, and the median bone metastasis-free survival (MFS) was 25 months.15
Rapid PSA doubling time (PSADT) is linked to shorter time to metastasis in this group of patients. Patients with a PSADT of < 10 months had a risk for bone metastasis 12 times greater and a risk for death 4 times greater than patients with a PSADT of ≥ 10 months.16 Accordingly, observation should be reserved for those patients with a PSADT of ≥ 10 months.
Options for secondary hormonal therapy in those with a PSADT of ≤ 10 months include a first-generation antiandrogen (bicalutamide, flutamide, nilutamide), ketoconazole with hydrocortisone, and more recently second-generation antiandrogens (apalutamide or enzalutamide).
Bicalutamide competitively inhibits dihydrotestosterone and testosterone binding to the AR and is generally well-tolerated; it is given in conjunction with a GnRH agonist/castration.17 The use of other first-generation antiandrogens is limited mainly due to their toxicity profile. When compared to flutamide in a randomized, double-blinded control study, bicalutamide had significantly improved time to treatment failure.18 Due to promiscuous binding to AR, withdrawal of first-generation antiandrogen therapy has been associated with a biochemical response in a small proportion of patients, with response typically seen after 5 to 7 half-lives of the drug have elapsed.19
Although traditionally used as an antifungal agent, ketoconazole also inhibits androgen synthesis in the adrenal glands and acts as a direct cytotoxin to cancer cells.20 Ketoconazole (with hydrocortisone) has been considered as a treatment option, typically at the time of antiandrogen withdrawal. However, ketoconazole offers no survival benefit, and with the approval of abiraterone in M1 CRPC, its use has declined significantly.21 Additionally, ketoconazole poses a risk for severe hepatotoxicity and QT prolongation, and has significant interactions with numerous drugs, thereby limiting its use. Given the typically short duration of response to first-generation antiandrogens, the second-generation antiandrogens were developed and are associated with a significantly greater progression-free survival (PFS) in M0 CRPC.22,23
The second-generation antiandrogens enzalutamide and apalutamide not only competitively bind to the AR, inhibiting formation of the androgen/AR complex, but they also inhibit androgen/AR complex nuclear translocation and binding to nuclear DNA. In the PROSPER trial, enzalutamide significantly increased radiographic PFS and improved quality of life compared to placebo in chemotherapy-naive patients (Table 1).24 Apalutamide significantly increased MFS as well as PFS and time to PSA progression compared to placebo in the phase 3 SPARTAN trial.25 Apalutamide is generally well tolerated, with hypertension and rash being the most common severe adverse effects. Apalutamide also has less potential for central nervous system toxicities than enzalutamide. The recent approval of these agents is likely to change responses to subsequent treatments, especially in the metastatic setting.

Treatment of Metastatic CRPC (M1 Disease)
As with M0 CRPC, ADT should be continued in patients with mCRPC to maintain castration levels of testosterone while initiating additional treatments. Several drugs for the treatment of mCRPC have been approved by the US Food and Drug Administration (FDA) since 2010, including abiraterone with prednisone (or methylprednisolone), enzalutamide (but not apalutamide), radium-223, sipuleucel-T, and cabazitaxel (Table 2).

Given the availability of numerous treatment options for men with mCRPC, the sequencing of treatments should be based on careful consideration of the efficacy and adverse effect profiles of each drug as well as the anatomic and molecular characteristics of the cancer, comorbidities, and patient preference. If there is no evidence of visceral disease and the patient has an Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1 with an estimated life expectancy of greater than 6 months and is minimally symptomatic, then treatment with either oral targeted agents or immunotherapy with sipuleucel-T is considered appropriate.
Sipuleucel-T is an autologous dendritic cell vaccination designed to enhance T-cell–mediated response to prostatic acid phosphatase (PAP). The treatments are prepared from leukapheresed host mononuclear cells that are then exposed to PAP fused to granulocyte-macrophage colony-stimulating factor. The activated dendritic cells are then infused back into the host once every 2 weeks for a total of 3 treatments. The main side effects of this treatment include chills, fever, and headache, but it is generally well-tolerated and has demonstrated a survival benefit.26
Both enzalutamide and abiraterone (abiraterone given with physiologic-dose steroid replacement) confer a survival benefit in chemotherapy-naive patients with M1 CRPC. Per the PREVAIL study, enzalutamide (when compared to placebo) offers a median improvement in overall survival (OS) by about 2 months and in radiographic PFS by about 14.6 months.24 Abiraterone blocks the synthesis of androgens via inhibition of CYP17 in the testes and adrenal glands. Abiraterone also confers an overall survival advantage for patients with M1 CRPC who are chemotherapy-naïve, with an estimated 25% decrease in the risk of death (hazard ratio, 0.75, P = 0.009) when compared to prednisone.27
In patients with symptomatic M1 CRPC who have visceral disease or rapidly progressive disease and who are candidates for chemotherapy, docetaxel is frequently used and is given concurrently with steroids. Docetaxel has been given for up to 10 cycles in clinical trials (assuming no progression of disease or dose-limiting toxicities were observed), and at least 6 cycles of treatment are recommended. When compared to mitoxantrone plus prednisone in the TAX 327 phase 3 trial, docetaxel plus prednisone offered a significant OS benefit of about 3 months (19.2 months versus 16.3 months).28 For patients who are not candidates for docetaxel (eg, due to preexisting peripheral neuropathy), cabazitaxel should be considered. OS is similar for mCRPC with docetaxel versus cabazitaxel when given in the first-line setting.29 Additionally, cabazitaxel dosed at 20 mg/m2 is noninferior to cabazitaxel dosed at 25 mg/m2, and the lower dose is associated with lower rates of peripheral neuropathy.30 Cabazitaxel should also be considered for mCRPC that has progressed following treatment with docetaxel, with improved OS and PFS when compared to treatment with mitoxantrone and prednisone in this setting, as shown in the TROPIC study.31 Mitoxantrone given with prednisone has been shown to improve quality of life, but it is associated with significant cardiac toxicity. Additionally, mitoxantrone does not improve disease-free survival or OS in chemotherapy-naive patients32 or in patients who have progressed on docetaxel, and therefore should not be given to patients prior to a taxane chemotherapy unless the patient absolutely cannot tolerate docetaxel or cabazitaxel.
Once a patient’s prostate cancer progresses following treatment with a taxane, the sequence in which to administer subsequent therapies should involve careful consideration of previous treatments and duration of response to each of these treatments. Both enzalutamide and abiraterone are FDA-approved for use following treatment with chemotherapy. Per the AFFIRM trial, heavily pre-treated patients (including those who have received docetaxel) have a median 5-month OS benefit with enzalutamide compared to placebo.33 Another study of M1 CRPC patients who had previously received docetaxel demonstrated an OS benefit with abiraterone (versus placebo),34 but this regimen has limited benefit in patients who have previously received both docetaxel and enzalutamide.35 A rechallenge with docetaxel therapy also can be considered if the patient’s disease responded to docetaxel in the metastatic hormone-sensitive setting.
If the patient’s metastases are limited to bone (ie, no visceral disease), then radiotherapy with radium-223 should be considered. Radium-223 is an alpha-emitting calcium-mimetic radioactive compound that tracks to bone to delay the onset of symptoms from bone metastases.36 Radium-223 also confers a median OS benefit of about 3 months.37 However, this treatment is often limited by preexisting cytopenias.
Diethylstilbestrol (1 mg daily) competes with androgens for AR binding and is also cytotoxic to androgen-sensitive and insensitive prostate cancer cells. While its efficacy is similar to bicalutamide in terms of PSA response rate and median response duration, diethylstilbestrol is associated with significantly more cardiovascular toxicity, including stroke, pulmonary embolism, and heart failure, and its use is therefore limited.38 The glucocorticoids—prednisone (5 mg orally twice daily), dexamethasone (0.5 mg daily), and hydrocortisone (40 mg daily)—inhibit pituitary synthesis of adrenocorticotropic hormone, resulting in decreased adrenal androgen synthesis. Data suggest that among the glucocorticoids, dexamethasone monotherapy may produce superior response rates compared to prednisone monotherapy.39 While the glucocorticoids do produce a PSA response, prolong time to disease progression, and can provide symptomatic relief (eg, from bone pain), they have not been shown to confer a survival benefit and therefore are not commonly used as monotherapy.
Future of CRPC Treatment
Patients with CRPC should be considered for clinical trials when available. These patients’ tumors should be assessed with next-generation sequencing for analysis of microsatellite instability (MSI) or mismatch repair (MMR) as well as the presence of other potentially targetable mutations, as this information may bring into consideration additional investigational as well as FDA-approved treatment options. As of May 2017, immunotherapy with pembrolizumab is approved for patients whose prostate cancer is deficient in MMR or has a high MSI burden based on a study of 12 solid tumor types (including prostate cancer) with deficient MMR.40 Additionally, for patients whose tumor has ≥ 1% programmed death ligand 1 (PD-L1) expression, pembrolizumab has a 17% overall response rate and confers stability of disease in 35%, with a median response duration of 13.5 months.41 Cabozantinib is a mesenchymal epithelial transition (MET) kinase and vascular endothelial growth factor receptor (VEGF-R) inhibitor. When used in heavily pre-treated patients with mCRPC, it showed a radiographic PFS benefit but no survival benefit over prednisone monotherapy.42 One study showed that for patients whose mCRPC had a homozygous deletion and/or a deleterious mutation in the homologous recombination repair genes BRCA1/2, ATM, and CHEK2 or the Fanconi anemia genes, the response rate to the poly ADP ribose polymerase (PARP) inhibitor olaparib was 88%, with a 100% response rate in those with BRCA2 mutations.43 Furthermore, mutations in these DNA repair genes predict increased sensitivity to platinum-based chemotherapy.
Supportive Care
Zoledronic acid or denosumab are FDA approved for men with CRPC and bone metastasis based on the ability of these agents to delay skeletal-related events, including pathologic fracture and spinal cord compression.46 Bisphosphonates, however, do not decrease the incidence of bone metastases.47 And while denosumab does delay the time to first bone metastasis in nmCRPC (particularly in patients with a PSADT of ≤ 6 months), it does not improve OS.48 Other supportive measures include exercise and nutrition. Moderate aerobic exercise for 150 minutes in addition to 2 or 3 strength training sessions per week is recommended by the American College of Sport Medicine to combat cancer-related fatigue.49 There are currently no dietary changes that are routinely recommended to improve the outcome of prostate cancer, but a study noted a shorter biochemical failure–free survival in men with prostate cancer who were obese and consumed a diet high in saturated fat.50
Conclusion
Prostate cancer affects more men in the United States than any other cancer. Once a patient is started on hormone therapy, in all likelihood their prostate cancer will become castration-resistant. Once prostate cancer has developed hormone resistance, there are a host of further treatment options available, including further hormone therapy, chemotherapy, immunotherapy, radiation therapy, bone-targeting agents, and clinical trials. Determining the appropriate sequence in which to use these therapies requires knowledge of the natural history of CRPC, the indications for changing therapies, the mechanism of action and adverse event profile of each treatment, and the optimal time to enroll in a clinical trial.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
1. Pound CR, Partin AW, Epstein JI, Walsh PC. Prostate-specific antigen after anatomic radical retropubic prostatectomy. Patterns of recurrence and cancer control. Urol Clin North Am. 1997;24:395-406.
2. Caire AA, Sun L, Ode O, et al. Delayed prostate-specific antigen recurrence after radical prostatectomy: how to identify and what are their clinical outcomes? Urology. 2009;74:643-647.
3. US Preventive Services Task Force, Grossman DC, Curry SJ, Owens DK, et al. Screening for prostate cancer: US Preventive Services Task Force recommendation statement. JAMA. 2018;319:1901-1913.
4. Huggins C, Hodges CV. Studies on prostatic cancer. I: The effects of castration, of estrogen, and of androgen injection on serum phosphatases in metastatic carcinoma of the prostate. Cancer Res. 1941;1:293-297.
5. Huggins C, Stevens RE Jr, Hodges CV. Studies on prostatic cancer. II: The effects of castration on advanced carcinoma of the prostate gland. Arch Surg. 1941;43:209-223.
6. Pomerantz M, Kantoff P. Clinical progression to castration recurrent prostate cancer. In: Tindall DJ, James M, eds. Androgen Action in Prostate Cancer. New York: Springer; 2009:57-72.
7. Kirby M, Hirst C, Crawford ED. Characterising the castration-resistant prostate cancer population: a systematic review. Int J Clin Pract. 2011;65:1180-1192.
8. Sharifi N, Dahut WL, Steinberg SM, et al. A retrospective study of the time to clinical endpoints for advanced prostate cancer. BJU Int. 2005;96:985-989.
9. Chen CD, Welsbie DS, Tran C, et al. Molecular determinants of resistance to antiandrogen therapy. Nat Med. 2004;10:33-39.
10. Taplin ME, Rajeshkumar B, Halabi S, et al. Androgen receptor mutations in androgen-independent prostate cancer: Cancer and Leukemia Group B Study 9663. J Clin Oncol. 2003;21:2673-2678.
11. Antonarakis ES, Lu C, Luber B, et al. Clinical significance of androgen receptor splice variant-7 (AR-V7) mRNA detection in circulating tumor cells of men with metastatic castration-resistant prostate cancer treated with first- and second-line abiraterone and enzalutamide. J Clin Oncol. 2017;35:2149-2156.
12. Kahn B, Collazo J, Kyprianou N. Androgen receptor as a driver of therapeutic resistance in advanced prostate cancer. Int J Biol Sci. 2014;10:588-595.
13. Logothetis CJ, Gallick GE, Maity SN, et al. Molecular classification of prostate cancer progression: foundation for marker-driven treatment of prostate cancer. Cancer Discov. 2013;3:849-861
14. Tombal B. Non-metastatic CRPC and asymptomatic metastatic CRPC: which treatment for which patient? Ann Oncol. 2012;23(suppl 10):251-258
15. Smith MR, Cook R, Lee KA, et al. Disease and host characteristics as predictors of time to first bone metastasis and death in men with progressive castration-resistant non-metastatic prostate cancer. Cancer. 2011;117:2077-2085.
16. Metwalli AR, Rosner IL, Cullen J, et al. Elevated alkaline phosphatase velocity strongly predicts overall survival and the risk of bone metastases in castrate-resistant prostate cancer. Urol Oncol. 2014;32:761-768
17. Akaza H, Yamaguchi A, Matsuda T, et al. Superior anti-tumor efficacy of bicalutamide 80mg in combination with luteinizing hormone-releasing hormone (LHRH) against versus LHRH agonist monotherapy as first line treatment for advanced prostate cancer: Interim results of a randomized study in Japanese patients. J Clin Oncol. 2004;34:20-28
18. Schellhammer P, Patterson AL, Sharifi R, et al. A controlled trial of bicalutamide versus flutamide, each in combination with luteinizing hormone-releasing hormone analogue therapy, in patients with advanced prostate cancer. Urology. 1995;45(5):745-752.
19. Sartor AO, Tangen CM, Hussain MH, et al. Antiandrogen withdrawal in castrate-refractory prostate cancer: a Southwest Oncology Group Trial (SWOG 9426). Cancer. 2008;112:2393-2400.
20. Eichenberger T, Trachtenberg J, Toor P, et al. Ketoconazole: a possible direct cytotoxic effect on prostate carcinoma cells. J Urol. 1989;141:190-191.
21. Small EJ, Halabi S, Dawson NA, et al. Antiandrogen withdrawal alone or in combination with ketoconazole in androgen-independent prostate cancer patients: a phase III trial (CALGB 9583). J Clin Oncol. 2004;22:1025-1033.
22. Penson DF, Armstrong AJ, Concepcion R, et al. Enzalutamide versus bicalutamide in castration resistant prostate cancer: the STRIVE trial. J Clin Oncol. 2016;34:2098-2106.
23. Shore ND, Chowdhury S, Villers A, et al. Efficacy and safety of enzalutamide versus bicalutamide for patients with metastatic prostate cancer (TERRAIN): a randomized, double-blind, phase 2 study. Lancet Oncol. 2016;199:147-154.
24. Beer TM, Armstrong AJ, Rathkopf D, et al. Enzalutamide in men with chemotherapy-naïve castration-resistant prostate cancer: extended analysis of the phase 3 PREVAIL study. Eur Urol. 2017;71:151-154.
25. Smith MR, Saad F, Chowdhury S; SPARTAN Investigators, et al. Apalutamide treatment and metastasis-free survival in prostate cancer. N Engl J Med. 2018;378:1408-1418.
26. Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411-422.
27. Ryan CJ, Smith MR, de Bono JS, et al. Randomized phase 3 trial of abiraterone acetate in men with metastatic castration-resistant prostate cancer and no prior chemotherapy. N Engl J Med. 2013;368:138-148
28. Berthold DR, Pond GR, Soban F, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer: updated survival in the TAX 327 study. J Clin Oncol. 2008;26:242-245.
29. Oudard S, Fizazi K, Sengelov L, et al. Cabazitaxel versus docetaxel as first-line therapy for patients with metastatic castration-resistant prostate cancer: a randomized phase III trial—FIRSTANA. J Clin Oncol. 2017;35:3189-3197.
30. Eisenberger M, Hardy-Bessard A-C, Kim CS, et al. Phase III study comparing a reduced dose of cabazitaxel (20 mg/m2) and the currently approved dose (25 mg/m2) in postdocetaxel patients with metastatic castration-resistant prostate cancer—PROSELICA. J Clin Oncol. 2017;35:3198-3206.
31. de Bono JS, Oudard S, Ozguroglu M, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progressing after docetaxel treatment: a randomised open-label trial. Lancet. 2010;376(9747):P1147-1154.
32. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med 2004;351:1502-1512.
33. Fizazi K, Scher HI, Miller K, et al. Effect of enzalutamide on time to first skeletal-related event, pain, and quality of life in men with castration-resistant prostate cancer: results from the randomised, phase 3 AFFIRM trial. Lancet Oncol. 2014;15:1147-1156.
34. de Bono JS, Logothetis CJ, Molina A, et al. Abiraterone and increased survival in metastatic prostate cancer. N Engl J Med. 2011;364:1995-2005.
35. Loriot Y, Bianchini D, Ileana E, et al. Antitumor activity of abiraterone acetate against metastatic castration-resistant prostate cancer progressing after docetaxel and enzalutamide. Ann Oncol. 2013;24:1807-1812.
36. Sartor O, Coleman R, Nilsson S, et al. Effect of radium-223 dichloride on symptomatic skeletal events in patients with castration-resistant prostate cancer and bone metastases: results from a phase 3, double blind, randomized trial. Lancet Oncol. 2014;15:738-746.
37. Parker C, Nilsson S, Heinrich D, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013; 369:213-223.
38. Turo R, Smolski M, Esler R, et al. Diethylstilboestrol for the treatment of prostate cancer: past, present and future. Scand J Urol. 2014;48:4-14.
39. Venkitaraman R, Lorente D, Murthy V. A randomized phase 2 trial of dexamethasone versus prednisolone in castration-resistant prostate cancer. Eur Urol. 2015 67:673-679.
40. Le DT, Durham JN, Smith KN, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. 2017;357(6349):409-413.
41. Hansen AR, Massard C, Ott PA, et al. Pembrolizumab for advanced prostate adenocarcinoma: findings of the KEYNOTE-028 study. Ann Oncol. 2018;29:1807-1813.
42. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
43. Mateo J, Carreira S, Sandhu S, et al. DNA-Repair defects and olaparib in metastatic prostate cancer. N Engl J Med. 2015;373:1697-1708.
44. Kentepozidis N, Soultati A, Giassas S, et al. Paclitaxel in combination with carboplatin as salvage treatment in patients with castration-resistant prostate cancer: a Hellenic oncology research group multicenter phase II study. Cancer Chemother Pharmacol. 2012;70:161-168.
45. Smith M, De Bono J, Sternberg C, et al. Phase III study of cabozantinib in previously treated metastatic castration-resistant prostate cancer: COMET-1. J Clin Oncol. 2016;34:3005-3013.
46. Saad F, Gleason DM, Murray R, et al. A randomized, placebo-controlled trial of zoledronic acid in patients with hormone-refractory metastatic prostate carcinoma. J Natl Cancer Inst. 2002;94:1458-1468.
47. Wirth M, Tammela T, Cicalese V, et al. Prevention of bone metastases in patients with high-risk nonmetastatic prostate cancer treated with zoledronic acid: efficacy and safety results of the Zometa European Study (ZEUS). Eur Urol. 2015;67:482-491.
48. Smith MR, Saad F, Coleman R, et al. Denosumab and bone-metastases-free survival in men with castration-resistant prostate cancer: results of a phase 3, randomized, placebo-controlled trial. Lancet. 2012;379(9810):39-46.
49. Schmitz KH, Courneya KS, Matthews C, et al. American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Med Sci Sports Exerc. 2010;42:1409-1426.
50. Strom SS, Yamamura Y, Flores-Sandoval FN, et al. Prostate cancer in Mexican-Americans: identification of risk factors. Prostate. 2008;68:563-570.
Topical Natural Products in Managing Dermatologic Conditions: Observations and Recommendations
Patients seek healthy skin that conveys overall health and well-being. Cosmeceuticals claim to therapeutically affect the structure and function of the skin, and it is rational to hold them to scientific standards that substantiate efficacy claims.1 Notably, it is increasingly important to consider nature-based products in helping patients and consumers to achieve healthier skin. Despite the availability of sophisticated efficacy testing, explanations of the underlying physiologic and pharmacologic principles of nature-based products lag behind those of conventional formulations. In many instances, simple form and function information cannot adequately support their desired use and expected benefits. In addition, cosmetic regulations do not even permit structure-function claims that are allowed for dietary supplements.
Physicians whose patients want recommendations for nature-based products often do not know where to turn for definitive product and use information. Unlike prescription medications or even beauty-from-within dietary supplement products, natural cosmetics and cosmeceuticals are barred from communicating scientific evidence and experience of use to form proper opinions for recommendations. Without the benefit of full product labeling, physicians are left to mine sparse, confusing, and often contradictory literature in an effort to self-educate. Here, we share our experiences with patients, our operating knowledge base, and our recommendations for investigation to improve the available information and ensure practicing physicians have the information they need to appropriately recommend nature-based products.
General Observations Pertaining to Patients and Nature-Based Products
Ethnic and cultural customs and traditions have accepted and employed nature-based products for skin health for millennia (eTables 1–3).2-20 African and the derived Caribbean cultures frequently use shea butter, black soap, or coconut oil. East Asian ethnobotanical practices include the use of ginseng, green tea, almond, and angelica root in skin care. Indian culture employs Ayurvedic medicine principles that include herbal remedies comprised of ground chickpeas, rice, turmeric, neem, ashwagandha, moringa, and kutki. These cultural traditions continue into modern times, and patients regularly use these products. Modern social trends that focus on a healthy lifestyle also create demand for nature-based products for skin health. In our opinion, the current growing interest in nature-based products implies continued growth in their use as patients become more familiar and comfortable with them.



For beauty and skin health, a new trend has evolved in which the first source of advice is rarely a dermatologist. Social media, nonphysician influencers, and pseudoscience have created an authority previously reserved for dermatologists among patients and consumers. Bloggers and social media influencers, posting their individual real-world experiences, shape the perceptions of consumers and patients.21,22 Nonphysician influencers leverage their celebrity to provide guidance and advice on beauty and cosmetic tips.23 Much of the evidence supporting cosmetic and especially nature-based products for skin care and health often is believed to be less rigorous and of lower quality than that typically supporting physician recommendations.24-26
Nature-Based Products in Skin Health and Dermatologic Conditions
Patients turn to nature-based products for skin care and health for many reasons. The simplest reason is that they grew up with such products and continue their use. Many patients find nature-based products themselves, have favorable experiences, and seek advice on their efficacy and safety for continued use. Patients also use these products as part of a holistic approach to health in which diet and exercise coincide with the idea of ministering to the whole self instead of preventing or treating an illness. These nature-based treatment options fit their natural lifestyles. Patients sometimes express concerns about synthetic products that lead them to seek out nature-based products. Chemicals and preservatives (eg, parabens, sunscreens, nanoparticles) may evoke concerns about negative health consequences, which can be a cause of great anxiety to patients.
Nature-based products, when recommended by physicians, can fulfill important roles. As healthier alternatives, they can address health concerns in the belief that plant-based ingredients may be more compatible with overall health than synthetic ingredients. This compatibility may have resulted from the human species coevolving with plant species containing therapeutic utility, leading to the development of specific receptors for many natural products, such as digoxin from foxglove (Digitalis purpurea), opioids from poppies (Papaver somniferum), and cannabinoids (Cannabis sativa and hybrids). Natural products can become alternatives to synthetic products or adjuncts to prescription medications. Often, inclusion of nature-based products into a treatment plan enables patients to feel that they are a more integral part of the care team treating their conditions. By virtue of physician recommendations, patients may have expectations on product efficacy being as robust as prescription products with the safety profile of plant-based products. Patients should be advised to accept a realistic view of the efficacy and tolerability profiles. In the end, patients consider physician recommendations based on the assumption that they are credible and derived from experience and knowledge.
Physician Perceptions of Nature-Based Products
Physicians recommend nature-based products based on several factors. Central to the recommendation is an understanding, through appropriate documentation, that the product will be reasonably efficacious. Critical to this point, physicians must understand what ingredients are in nature-based products, their concentrations or amounts, and why they are present. However, our experience with nature-based products suggests that many of these factors are not met. Limited or unclear information on the efficacy of nature-based products fails to satisfy a physician’s need for adequate information to support recommendations. Although natural ingredients are listed on product labels, their intended benefit and efficacy characteristics often are unclear or poorly stated, in some cases resulting from improper labeling and in other cases due to claim restrictions imposed on cosmetics. In addition, insufficient details on formulation, such as type and percentages of oils, antioxidants, and vitamins, hinder the physician’s ability to identify and explain mechanisms that bring benefit to the patient. Universal benchmarks do not exist for amounts or concentrations of ingredients that are required for a stated benefit.27 Currently, no standards exist for assurances that product quality, control, and efficacy are consistently reproducible. For example, angel dusting is a practice that discloses that an active ingredient is present, yet these ingredients may be present in quantities that are insufficient to provide measurable benefit. Sourcing of ingredients also can be concerning, as they may not always meet manufacturer, physician, or patient expectations for characterization or efficacy.28,29 Dry testing, which is when a manufacturer contracts a laboratory to certify their ingredients without performing assays, has been increasingly reported in lay and botanical literature over the last few years.30
It is unknown if many nature-based products clinically exhibit their stated efficacy. Empirical evidence or well-conducted clinical studies on which to base recommendations of these products are limited. Individual natural ingredients, however, do have some supporting evidence of efficacy: shea butter moisturizes31; coconut oil exhibits anti-inflammatory properties32,33; and vinegar, yogurt, and diluted tea tree oil exhibit antibacterial properties in postprocedure care and fungal infections, and as adjuvants to prescription antibiotics in atopic dermatitis, acne, and rosacea.34-41 Honey also has been shown to improve wound healing and is even available as a medical device for wounds.42,43 Although nature-based products are an interesting alternative to synthetic products, they require a fulsome understanding of characteristics and efficacy properties to support physician recommendations.
Physician Recommendations
Physicians must be educated to understand when and how to recommend nature-based products. Although we recommend increased product information to guide physicians, current laws, including the Federal Food, Drug, and Cosmetic Act and the Fair Packaging and Labeling Act, are satisfactory from a regulatory standpoint.44 Here, we discuss the information physicians could use to support an informed recommendation of nature-based products.
A clear specific explanation of natural ingredient sources, their intended efficacy, and rigorous scientific clinical evidence supporting their use should be given. Manufacturers are needed to document and report the structure and function of natural ingredients, leading to a common understanding by practicing dermatologists.45 For this reason, manufacturers must provide nonambiguous and standardized methods and measures to demonstrate the mechanism of ingredient efficacy and the limits of safety and tolerability.
We recommend that manufacturers provide standardized transparency into the composition of nature-based formulations, including amounts and concentrations of ingredients; geographic sources; parts of plants used; and if extracted, what agent(s) this standard is based on (eg, hypericin in Saint-John’s-wort or kavalactones in kava kava). Most natural products contain an aqueous phase and therefore will likely require preservatives such as synthetic parabens or alcohols to avoid degradation. Unnecessary ingredients, including fragrances, fillers, and support chemicals, should be absent since inert agents may exhibit biologic effects, obscuring the boundary between active and inert. A clear explanation of the origins of these nature-based ingredients and the concentration, purity, and activity assessment should be provided. In the context of an authoritative review with standardized measures, labels that provide the common name, plant name, part used, how it was obtained, concentrations and/or amounts, and standardized activity measures can be helpful to the recommending physician, who will then know the efficacy patients should expect from the ingredients. They also can assess the expected tolerability based on the concentrations and their own experience managing a particular disorder, tempered by the patient’s experiences with prior therapies. Transparent and standardized labeling describing the formulation, quantities of ingredients, and intended activity will help inform expectations of efficacy.
We recommend clear preclinical and clinical demonstrations of the efficacy and benefits that are claimed by nature-based formulations. Properly designed placebo- or active-controlled, blinded, randomized studies with standardized measures and end points are recommended to determine efficacy and safety. These demonstrations of efficacy can provide physicians with credible evidence on which to base their recommendations and guide the use of products for the patient’s best experience. Given sufficient involvement from manufacturers and publication of the information in peer-reviewed journals, the relative benefits for each nature-based product can be cataloged as a resource for physicians.
Conclusion
Patients turn to nature-based products for many reasons. They have high expectations but also harbor concerns as to the efficacy of these products for skin and health care. Physicians seek to recommend nature-based products for these patients but often find themselves disadvantaged by limited published evidence and insufficient labeling information on composition and efficacy, which should support recommendations for use. To remedy this situation, we suggest research to allow a clear explanation of the activity of natural ingredients, clear demonstrations of the efficacy of nature-based formulas using clinical standardized measures and end points, and clear education and disclosure of ingredients contained within nature-based products.
Acknowledgments—Burt’s Bees (Durham, North Carolina) provided funding for editorial support by Medical Dynamics, Inc (New York, New York).
- Levin J, Momin SB. How much do we really know about our favorite cosmeceutical ingredients? J Clin Aesthet Dermatol. 2010;3:22-41.
- Ajala EO, Aberuagba F, Olaniyan AM, et al. Optimization of solvent extraction of shea butter (Vitellaria paradoxa) using response surface methodology and its characterization. J Food Sci Technol. 2016;53:730-738.
- Lin A, Nabatian A, Halverstam CP. Discovering black soap: a survey on the attitudes and practices of black soap users. J Clin Aesthet Dermatol. 2017;10:18-22.
- Lin TK, Zhong L, Santiago JL. Anti-inflammatory and skin barrier repair effects of topical application of some plant oils. Int J Mol Sci. 2017;19. pii:E70. doi:10.3390/ijms19010070.
- Dua K, Sheshala R, Ling TY, et al. Anti-inflammatory, antibacterial and analgesic potential of cocos nucifera linn.: a review. Antiinflamm Antiallergy Agents Med Chem. 2013;12:158-164.
- Hyun TK, Jang KI. Are berries useless by-products of ginseng? recent research on the potential health benefits of ginseng berry. EXCLI J. 2017;16:780-784.
- Truong VL, Bak MJ, Lee C, et al. Hair regenerative mechanisms of red ginseng oil and its major components in the testosterone-induced delay of anagen entry in C57BL/6 mice. Molecules. 2017;22. pii:E1505. doi:10.3390/molecules22091505.
- Hussain M, Habib Ur R, Akhtar L. Therapeutic benefits of green tea extract on various parameters in non-alcoholic fatty liver disease patients. Pak J Med Sci. 2017;33:931-936.
- Yi M, Fu J, Zhou L, et al. The effect of almond consumption on elements of endurance exercise performance in trained athletes. J Int Soc Sports Nutr. 2014;11:18.
- Sowndhararajan K, Deepa P, Kim M, et al. A review of the composition of the essential oils and biological activities of angelica species. Sci Pharm. 2017;85. pii:E33. doi:10.3390/scipharm85030033.
- Mahjour M, Khoushabi A, Noras M, et al. Effectiveness of Cicer arietinum in cutaneous problems: viewpoint of Avicenna and Razi. Curr Drug Discov Technol. 2018;15:243-250.
- Kanlayavattanakul M, Laurits N, Chaikul P. Jasmine rice panicle: a safe and efficient natural ingredient for skin aging treatments. J Ethnopharmacol. 2016;193:607-616.
- Aggarwal BB, Yuan W, Li S, et al. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
- Mohanty C, Sahoo SK. Curcumin and its topical formulations for wound healing applications. Drug Discov Today. 2017;22:1582-1592.
- Gupta SC, Prasad S, Tyagi AK, et al. Neem (Azadirachta indica): an Indian traditional panacea with modern molecular basis. Phytomedicine. 2017;34:14-20.
- Choudhary D, Bhattacharyya S, Bose S. Efficacy and safety of ashwagandha (Withania somnifera (L.) Dunal) root extract in improving memory and cognitive functions. J Diet Suppl. 2017;14:599-612.
- Halder B, Singh S, Thakur SS. Withania somnifera root extract has potent cytotoxic effect against human malignant melanoma cells. PLoS One. 2015;10:E0137498.
- Nadeem M, Imran M. Promising features of Moringa oleifera oil: recent updates and perspectives. Lipids Health Dis. 2016;15:212.
- Sultan P, Jan A, Pervaiz Q. Phytochemical studies for quantitative estimation of iridoid glycosides in Picrorhiza kurroa Royle. Bot Stud. 2016;57:7.
- Gianfaldoni S, Wollina U, Tirant M, et al. Herbal compounds for the treatment of vitiligo: a review. Open Access Maced J Med Sci. 2018;6:203-207.
- Diamantoglou M, Platz J, Vienken J. Cellulose carbamates and derivatives as hemocompatible membrane materials for hemodialysis. Artif Organs. 1999;23:15-22.
- Respiratory syncytial virus (RSV). Centers for Disease Control and Prevention website. http://www.cdc.gov/rsv/research/us-surveillance.html. Updated June 26, 2018. Accessed February 1, 2019.
- Dembo G, Park SB, Kharasch ED. Central nervous system concentrations of cyclooxygenase-2 inhibitors in humans. Anesthesiology. 2005;102:409-415.
- Fong P. CFTR-SLC26 transporter interactions in epithelia. Biophys Rev. 2012;4:107-116.
- Liu Z. How cosmeceuticals companies get away with pseudoscience. Pacific Standard website. https://psmag.com/environment/cosmetic-companies-get-away-pseudoscience-placebo-week-92455. Published October 15, 2014. Accessed February 1, 2019.
- Beyerstein BL. Alternative medicine and common errors of reasoning. Acad Med. 2001;76:230-237.
- Topical antimicrobial drug products for over-the-counter human use. US Food and Drug Administration website. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=333.310. Accessed February 1, 2019.
- Natural personal care. Natural Products Association website. https://www.npanational.org/certifications/natural-seal/natural-seal-personal-care/. Accessed March 27, 2019.
- Natural Cosmetics Standard. GFaW Web site. https://gfaw.eu/en/ncs-for-all-who-love-nature-and-cosmetics/ncs-information-for-consumer/. Accessed February 1, 2019.
- Brown PN, Betz JM, Jasch F. How to qualify an analytical laboratory for analysis of herbal dietary ingredients and avoid using a “dry lab”: a review of issues related to using a contract analytical laboratory by industry, academia, and regulatory agencies. HerbalGram. 2013:52-59.
- Oh MJ, Cho YH, Cha SY, et al. Novel phytoceramides containing fatty acids of diverse chain lengths are better than a single C18-ceramide N-stearoyl phytosphingosine to improve the physiological properties of human stratum corneum. Clin Cosmet Investig Dermatol. 2017;10:363-371.
- Famurewa AC, Aja PM, Maduagwuna EK, et al. Antioxidant and anti-inflammatory effects of virgin coconut oil supplementation abrogate acute chemotherapy oxidative nephrotoxicity induced by anticancer drug methotrexate in rats. Biomed Pharmacother. 2017;96:905-911.
- Intahphuak S, Khonsung P, Panthong A. Anti-inflammatory, analgesic, and antipyretic activities of virgin coconut oil. Pharm Biol. 2010;48:151-157.
- McKenna PJ, Lehr GS, Leist P, et al. Antiseptic effectiveness with fibroblast preservation. Ann Plast Surg. 1991;27:265-268.
- Brockow K, Grabenhorst P, Abeck D, et al. Effect of gentian violet, corticosteroid and tar preparations in Staphylococcus aureus-colonized atopic eczema. Dermatology. 1999;199:231-236.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
- Misner BD. A novel aromatic oil compound inhibits microbial overgrowth on feet: a case study. J Int Soc Sports Nutr. 2007;4:3.
- D’Auria FD, Laino L, Strippoli V, et al. In vitro activity of tea tree oil against Candida albicans mycelial conversion and other pathogenic fungi. J Chemother. 2001;13:377-383.
- Fuchs-Tarlovsky V, Marquez-Barba MF, Sriram K. Probiotics in dermatologic practice. Nutrition. 2016;32:289-295.
- Bowe W, Patel NB, Logan AC. Acne vulgaris, probiotics and the gut-brain-skin axis: from anecdote to translational medicine. Benef Microbes. 2014;5:185-199.
- Baquerizo Nole KL, Yim E, Keri JE. Probiotics and prebiotics in dermatology. J Am Acad Dermatol. 2014;71:814-821.
- Saikaly SK, Khachemoune A. Honey and wound healing: an update. Am J Clin Dermatol. 2017;18:237-251.
- Aziz Z, Abdul Rasool Hassan B. The effects of honey compared to silver sulfadiazine for the treatment of burns: a systematic review of randomized controlled trials. Burns. 2017;43:50-57.
- FDA authority over cosmetics: how cosmetics are not FDA-approved, but are FDA-regulated. US Food and Drug AdministrationWeb site. https://www.fda.gov/cosmetics/guidanceregulation/lawsregulations/ucm074162.htm. Updated July 24, 2018. Accessed February 1, 2019.
- Wohlrab J. Topical preparations and their use in dermatology. J Dtsch Dermatol Ges. 2016;4:1061-1070
Patients seek healthy skin that conveys overall health and well-being. Cosmeceuticals claim to therapeutically affect the structure and function of the skin, and it is rational to hold them to scientific standards that substantiate efficacy claims.1 Notably, it is increasingly important to consider nature-based products in helping patients and consumers to achieve healthier skin. Despite the availability of sophisticated efficacy testing, explanations of the underlying physiologic and pharmacologic principles of nature-based products lag behind those of conventional formulations. In many instances, simple form and function information cannot adequately support their desired use and expected benefits. In addition, cosmetic regulations do not even permit structure-function claims that are allowed for dietary supplements.
Physicians whose patients want recommendations for nature-based products often do not know where to turn for definitive product and use information. Unlike prescription medications or even beauty-from-within dietary supplement products, natural cosmetics and cosmeceuticals are barred from communicating scientific evidence and experience of use to form proper opinions for recommendations. Without the benefit of full product labeling, physicians are left to mine sparse, confusing, and often contradictory literature in an effort to self-educate. Here, we share our experiences with patients, our operating knowledge base, and our recommendations for investigation to improve the available information and ensure practicing physicians have the information they need to appropriately recommend nature-based products.
General Observations Pertaining to Patients and Nature-Based Products
Ethnic and cultural customs and traditions have accepted and employed nature-based products for skin health for millennia (eTables 1–3).2-20 African and the derived Caribbean cultures frequently use shea butter, black soap, or coconut oil. East Asian ethnobotanical practices include the use of ginseng, green tea, almond, and angelica root in skin care. Indian culture employs Ayurvedic medicine principles that include herbal remedies comprised of ground chickpeas, rice, turmeric, neem, ashwagandha, moringa, and kutki. These cultural traditions continue into modern times, and patients regularly use these products. Modern social trends that focus on a healthy lifestyle also create demand for nature-based products for skin health. In our opinion, the current growing interest in nature-based products implies continued growth in their use as patients become more familiar and comfortable with them.



For beauty and skin health, a new trend has evolved in which the first source of advice is rarely a dermatologist. Social media, nonphysician influencers, and pseudoscience have created an authority previously reserved for dermatologists among patients and consumers. Bloggers and social media influencers, posting their individual real-world experiences, shape the perceptions of consumers and patients.21,22 Nonphysician influencers leverage their celebrity to provide guidance and advice on beauty and cosmetic tips.23 Much of the evidence supporting cosmetic and especially nature-based products for skin care and health often is believed to be less rigorous and of lower quality than that typically supporting physician recommendations.24-26
Nature-Based Products in Skin Health and Dermatologic Conditions
Patients turn to nature-based products for skin care and health for many reasons. The simplest reason is that they grew up with such products and continue their use. Many patients find nature-based products themselves, have favorable experiences, and seek advice on their efficacy and safety for continued use. Patients also use these products as part of a holistic approach to health in which diet and exercise coincide with the idea of ministering to the whole self instead of preventing or treating an illness. These nature-based treatment options fit their natural lifestyles. Patients sometimes express concerns about synthetic products that lead them to seek out nature-based products. Chemicals and preservatives (eg, parabens, sunscreens, nanoparticles) may evoke concerns about negative health consequences, which can be a cause of great anxiety to patients.
Nature-based products, when recommended by physicians, can fulfill important roles. As healthier alternatives, they can address health concerns in the belief that plant-based ingredients may be more compatible with overall health than synthetic ingredients. This compatibility may have resulted from the human species coevolving with plant species containing therapeutic utility, leading to the development of specific receptors for many natural products, such as digoxin from foxglove (Digitalis purpurea), opioids from poppies (Papaver somniferum), and cannabinoids (Cannabis sativa and hybrids). Natural products can become alternatives to synthetic products or adjuncts to prescription medications. Often, inclusion of nature-based products into a treatment plan enables patients to feel that they are a more integral part of the care team treating their conditions. By virtue of physician recommendations, patients may have expectations on product efficacy being as robust as prescription products with the safety profile of plant-based products. Patients should be advised to accept a realistic view of the efficacy and tolerability profiles. In the end, patients consider physician recommendations based on the assumption that they are credible and derived from experience and knowledge.
Physician Perceptions of Nature-Based Products
Physicians recommend nature-based products based on several factors. Central to the recommendation is an understanding, through appropriate documentation, that the product will be reasonably efficacious. Critical to this point, physicians must understand what ingredients are in nature-based products, their concentrations or amounts, and why they are present. However, our experience with nature-based products suggests that many of these factors are not met. Limited or unclear information on the efficacy of nature-based products fails to satisfy a physician’s need for adequate information to support recommendations. Although natural ingredients are listed on product labels, their intended benefit and efficacy characteristics often are unclear or poorly stated, in some cases resulting from improper labeling and in other cases due to claim restrictions imposed on cosmetics. In addition, insufficient details on formulation, such as type and percentages of oils, antioxidants, and vitamins, hinder the physician’s ability to identify and explain mechanisms that bring benefit to the patient. Universal benchmarks do not exist for amounts or concentrations of ingredients that are required for a stated benefit.27 Currently, no standards exist for assurances that product quality, control, and efficacy are consistently reproducible. For example, angel dusting is a practice that discloses that an active ingredient is present, yet these ingredients may be present in quantities that are insufficient to provide measurable benefit. Sourcing of ingredients also can be concerning, as they may not always meet manufacturer, physician, or patient expectations for characterization or efficacy.28,29 Dry testing, which is when a manufacturer contracts a laboratory to certify their ingredients without performing assays, has been increasingly reported in lay and botanical literature over the last few years.30
It is unknown if many nature-based products clinically exhibit their stated efficacy. Empirical evidence or well-conducted clinical studies on which to base recommendations of these products are limited. Individual natural ingredients, however, do have some supporting evidence of efficacy: shea butter moisturizes31; coconut oil exhibits anti-inflammatory properties32,33; and vinegar, yogurt, and diluted tea tree oil exhibit antibacterial properties in postprocedure care and fungal infections, and as adjuvants to prescription antibiotics in atopic dermatitis, acne, and rosacea.34-41 Honey also has been shown to improve wound healing and is even available as a medical device for wounds.42,43 Although nature-based products are an interesting alternative to synthetic products, they require a fulsome understanding of characteristics and efficacy properties to support physician recommendations.
Physician Recommendations
Physicians must be educated to understand when and how to recommend nature-based products. Although we recommend increased product information to guide physicians, current laws, including the Federal Food, Drug, and Cosmetic Act and the Fair Packaging and Labeling Act, are satisfactory from a regulatory standpoint.44 Here, we discuss the information physicians could use to support an informed recommendation of nature-based products.
A clear specific explanation of natural ingredient sources, their intended efficacy, and rigorous scientific clinical evidence supporting their use should be given. Manufacturers are needed to document and report the structure and function of natural ingredients, leading to a common understanding by practicing dermatologists.45 For this reason, manufacturers must provide nonambiguous and standardized methods and measures to demonstrate the mechanism of ingredient efficacy and the limits of safety and tolerability.
We recommend that manufacturers provide standardized transparency into the composition of nature-based formulations, including amounts and concentrations of ingredients; geographic sources; parts of plants used; and if extracted, what agent(s) this standard is based on (eg, hypericin in Saint-John’s-wort or kavalactones in kava kava). Most natural products contain an aqueous phase and therefore will likely require preservatives such as synthetic parabens or alcohols to avoid degradation. Unnecessary ingredients, including fragrances, fillers, and support chemicals, should be absent since inert agents may exhibit biologic effects, obscuring the boundary between active and inert. A clear explanation of the origins of these nature-based ingredients and the concentration, purity, and activity assessment should be provided. In the context of an authoritative review with standardized measures, labels that provide the common name, plant name, part used, how it was obtained, concentrations and/or amounts, and standardized activity measures can be helpful to the recommending physician, who will then know the efficacy patients should expect from the ingredients. They also can assess the expected tolerability based on the concentrations and their own experience managing a particular disorder, tempered by the patient’s experiences with prior therapies. Transparent and standardized labeling describing the formulation, quantities of ingredients, and intended activity will help inform expectations of efficacy.
We recommend clear preclinical and clinical demonstrations of the efficacy and benefits that are claimed by nature-based formulations. Properly designed placebo- or active-controlled, blinded, randomized studies with standardized measures and end points are recommended to determine efficacy and safety. These demonstrations of efficacy can provide physicians with credible evidence on which to base their recommendations and guide the use of products for the patient’s best experience. Given sufficient involvement from manufacturers and publication of the information in peer-reviewed journals, the relative benefits for each nature-based product can be cataloged as a resource for physicians.
Conclusion
Patients turn to nature-based products for many reasons. They have high expectations but also harbor concerns as to the efficacy of these products for skin and health care. Physicians seek to recommend nature-based products for these patients but often find themselves disadvantaged by limited published evidence and insufficient labeling information on composition and efficacy, which should support recommendations for use. To remedy this situation, we suggest research to allow a clear explanation of the activity of natural ingredients, clear demonstrations of the efficacy of nature-based formulas using clinical standardized measures and end points, and clear education and disclosure of ingredients contained within nature-based products.
Acknowledgments—Burt’s Bees (Durham, North Carolina) provided funding for editorial support by Medical Dynamics, Inc (New York, New York).
Patients seek healthy skin that conveys overall health and well-being. Cosmeceuticals claim to therapeutically affect the structure and function of the skin, and it is rational to hold them to scientific standards that substantiate efficacy claims.1 Notably, it is increasingly important to consider nature-based products in helping patients and consumers to achieve healthier skin. Despite the availability of sophisticated efficacy testing, explanations of the underlying physiologic and pharmacologic principles of nature-based products lag behind those of conventional formulations. In many instances, simple form and function information cannot adequately support their desired use and expected benefits. In addition, cosmetic regulations do not even permit structure-function claims that are allowed for dietary supplements.
Physicians whose patients want recommendations for nature-based products often do not know where to turn for definitive product and use information. Unlike prescription medications or even beauty-from-within dietary supplement products, natural cosmetics and cosmeceuticals are barred from communicating scientific evidence and experience of use to form proper opinions for recommendations. Without the benefit of full product labeling, physicians are left to mine sparse, confusing, and often contradictory literature in an effort to self-educate. Here, we share our experiences with patients, our operating knowledge base, and our recommendations for investigation to improve the available information and ensure practicing physicians have the information they need to appropriately recommend nature-based products.
General Observations Pertaining to Patients and Nature-Based Products
Ethnic and cultural customs and traditions have accepted and employed nature-based products for skin health for millennia (eTables 1–3).2-20 African and the derived Caribbean cultures frequently use shea butter, black soap, or coconut oil. East Asian ethnobotanical practices include the use of ginseng, green tea, almond, and angelica root in skin care. Indian culture employs Ayurvedic medicine principles that include herbal remedies comprised of ground chickpeas, rice, turmeric, neem, ashwagandha, moringa, and kutki. These cultural traditions continue into modern times, and patients regularly use these products. Modern social trends that focus on a healthy lifestyle also create demand for nature-based products for skin health. In our opinion, the current growing interest in nature-based products implies continued growth in their use as patients become more familiar and comfortable with them.



For beauty and skin health, a new trend has evolved in which the first source of advice is rarely a dermatologist. Social media, nonphysician influencers, and pseudoscience have created an authority previously reserved for dermatologists among patients and consumers. Bloggers and social media influencers, posting their individual real-world experiences, shape the perceptions of consumers and patients.21,22 Nonphysician influencers leverage their celebrity to provide guidance and advice on beauty and cosmetic tips.23 Much of the evidence supporting cosmetic and especially nature-based products for skin care and health often is believed to be less rigorous and of lower quality than that typically supporting physician recommendations.24-26
Nature-Based Products in Skin Health and Dermatologic Conditions
Patients turn to nature-based products for skin care and health for many reasons. The simplest reason is that they grew up with such products and continue their use. Many patients find nature-based products themselves, have favorable experiences, and seek advice on their efficacy and safety for continued use. Patients also use these products as part of a holistic approach to health in which diet and exercise coincide with the idea of ministering to the whole self instead of preventing or treating an illness. These nature-based treatment options fit their natural lifestyles. Patients sometimes express concerns about synthetic products that lead them to seek out nature-based products. Chemicals and preservatives (eg, parabens, sunscreens, nanoparticles) may evoke concerns about negative health consequences, which can be a cause of great anxiety to patients.
Nature-based products, when recommended by physicians, can fulfill important roles. As healthier alternatives, they can address health concerns in the belief that plant-based ingredients may be more compatible with overall health than synthetic ingredients. This compatibility may have resulted from the human species coevolving with plant species containing therapeutic utility, leading to the development of specific receptors for many natural products, such as digoxin from foxglove (Digitalis purpurea), opioids from poppies (Papaver somniferum), and cannabinoids (Cannabis sativa and hybrids). Natural products can become alternatives to synthetic products or adjuncts to prescription medications. Often, inclusion of nature-based products into a treatment plan enables patients to feel that they are a more integral part of the care team treating their conditions. By virtue of physician recommendations, patients may have expectations on product efficacy being as robust as prescription products with the safety profile of plant-based products. Patients should be advised to accept a realistic view of the efficacy and tolerability profiles. In the end, patients consider physician recommendations based on the assumption that they are credible and derived from experience and knowledge.
Physician Perceptions of Nature-Based Products
Physicians recommend nature-based products based on several factors. Central to the recommendation is an understanding, through appropriate documentation, that the product will be reasonably efficacious. Critical to this point, physicians must understand what ingredients are in nature-based products, their concentrations or amounts, and why they are present. However, our experience with nature-based products suggests that many of these factors are not met. Limited or unclear information on the efficacy of nature-based products fails to satisfy a physician’s need for adequate information to support recommendations. Although natural ingredients are listed on product labels, their intended benefit and efficacy characteristics often are unclear or poorly stated, in some cases resulting from improper labeling and in other cases due to claim restrictions imposed on cosmetics. In addition, insufficient details on formulation, such as type and percentages of oils, antioxidants, and vitamins, hinder the physician’s ability to identify and explain mechanisms that bring benefit to the patient. Universal benchmarks do not exist for amounts or concentrations of ingredients that are required for a stated benefit.27 Currently, no standards exist for assurances that product quality, control, and efficacy are consistently reproducible. For example, angel dusting is a practice that discloses that an active ingredient is present, yet these ingredients may be present in quantities that are insufficient to provide measurable benefit. Sourcing of ingredients also can be concerning, as they may not always meet manufacturer, physician, or patient expectations for characterization or efficacy.28,29 Dry testing, which is when a manufacturer contracts a laboratory to certify their ingredients without performing assays, has been increasingly reported in lay and botanical literature over the last few years.30
It is unknown if many nature-based products clinically exhibit their stated efficacy. Empirical evidence or well-conducted clinical studies on which to base recommendations of these products are limited. Individual natural ingredients, however, do have some supporting evidence of efficacy: shea butter moisturizes31; coconut oil exhibits anti-inflammatory properties32,33; and vinegar, yogurt, and diluted tea tree oil exhibit antibacterial properties in postprocedure care and fungal infections, and as adjuvants to prescription antibiotics in atopic dermatitis, acne, and rosacea.34-41 Honey also has been shown to improve wound healing and is even available as a medical device for wounds.42,43 Although nature-based products are an interesting alternative to synthetic products, they require a fulsome understanding of characteristics and efficacy properties to support physician recommendations.
Physician Recommendations
Physicians must be educated to understand when and how to recommend nature-based products. Although we recommend increased product information to guide physicians, current laws, including the Federal Food, Drug, and Cosmetic Act and the Fair Packaging and Labeling Act, are satisfactory from a regulatory standpoint.44 Here, we discuss the information physicians could use to support an informed recommendation of nature-based products.
A clear specific explanation of natural ingredient sources, their intended efficacy, and rigorous scientific clinical evidence supporting their use should be given. Manufacturers are needed to document and report the structure and function of natural ingredients, leading to a common understanding by practicing dermatologists.45 For this reason, manufacturers must provide nonambiguous and standardized methods and measures to demonstrate the mechanism of ingredient efficacy and the limits of safety and tolerability.
We recommend that manufacturers provide standardized transparency into the composition of nature-based formulations, including amounts and concentrations of ingredients; geographic sources; parts of plants used; and if extracted, what agent(s) this standard is based on (eg, hypericin in Saint-John’s-wort or kavalactones in kava kava). Most natural products contain an aqueous phase and therefore will likely require preservatives such as synthetic parabens or alcohols to avoid degradation. Unnecessary ingredients, including fragrances, fillers, and support chemicals, should be absent since inert agents may exhibit biologic effects, obscuring the boundary between active and inert. A clear explanation of the origins of these nature-based ingredients and the concentration, purity, and activity assessment should be provided. In the context of an authoritative review with standardized measures, labels that provide the common name, plant name, part used, how it was obtained, concentrations and/or amounts, and standardized activity measures can be helpful to the recommending physician, who will then know the efficacy patients should expect from the ingredients. They also can assess the expected tolerability based on the concentrations and their own experience managing a particular disorder, tempered by the patient’s experiences with prior therapies. Transparent and standardized labeling describing the formulation, quantities of ingredients, and intended activity will help inform expectations of efficacy.
We recommend clear preclinical and clinical demonstrations of the efficacy and benefits that are claimed by nature-based formulations. Properly designed placebo- or active-controlled, blinded, randomized studies with standardized measures and end points are recommended to determine efficacy and safety. These demonstrations of efficacy can provide physicians with credible evidence on which to base their recommendations and guide the use of products for the patient’s best experience. Given sufficient involvement from manufacturers and publication of the information in peer-reviewed journals, the relative benefits for each nature-based product can be cataloged as a resource for physicians.
Conclusion
Patients turn to nature-based products for many reasons. They have high expectations but also harbor concerns as to the efficacy of these products for skin and health care. Physicians seek to recommend nature-based products for these patients but often find themselves disadvantaged by limited published evidence and insufficient labeling information on composition and efficacy, which should support recommendations for use. To remedy this situation, we suggest research to allow a clear explanation of the activity of natural ingredients, clear demonstrations of the efficacy of nature-based formulas using clinical standardized measures and end points, and clear education and disclosure of ingredients contained within nature-based products.
Acknowledgments—Burt’s Bees (Durham, North Carolina) provided funding for editorial support by Medical Dynamics, Inc (New York, New York).
- Levin J, Momin SB. How much do we really know about our favorite cosmeceutical ingredients? J Clin Aesthet Dermatol. 2010;3:22-41.
- Ajala EO, Aberuagba F, Olaniyan AM, et al. Optimization of solvent extraction of shea butter (Vitellaria paradoxa) using response surface methodology and its characterization. J Food Sci Technol. 2016;53:730-738.
- Lin A, Nabatian A, Halverstam CP. Discovering black soap: a survey on the attitudes and practices of black soap users. J Clin Aesthet Dermatol. 2017;10:18-22.
- Lin TK, Zhong L, Santiago JL. Anti-inflammatory and skin barrier repair effects of topical application of some plant oils. Int J Mol Sci. 2017;19. pii:E70. doi:10.3390/ijms19010070.
- Dua K, Sheshala R, Ling TY, et al. Anti-inflammatory, antibacterial and analgesic potential of cocos nucifera linn.: a review. Antiinflamm Antiallergy Agents Med Chem. 2013;12:158-164.
- Hyun TK, Jang KI. Are berries useless by-products of ginseng? recent research on the potential health benefits of ginseng berry. EXCLI J. 2017;16:780-784.
- Truong VL, Bak MJ, Lee C, et al. Hair regenerative mechanisms of red ginseng oil and its major components in the testosterone-induced delay of anagen entry in C57BL/6 mice. Molecules. 2017;22. pii:E1505. doi:10.3390/molecules22091505.
- Hussain M, Habib Ur R, Akhtar L. Therapeutic benefits of green tea extract on various parameters in non-alcoholic fatty liver disease patients. Pak J Med Sci. 2017;33:931-936.
- Yi M, Fu J, Zhou L, et al. The effect of almond consumption on elements of endurance exercise performance in trained athletes. J Int Soc Sports Nutr. 2014;11:18.
- Sowndhararajan K, Deepa P, Kim M, et al. A review of the composition of the essential oils and biological activities of angelica species. Sci Pharm. 2017;85. pii:E33. doi:10.3390/scipharm85030033.
- Mahjour M, Khoushabi A, Noras M, et al. Effectiveness of Cicer arietinum in cutaneous problems: viewpoint of Avicenna and Razi. Curr Drug Discov Technol. 2018;15:243-250.
- Kanlayavattanakul M, Laurits N, Chaikul P. Jasmine rice panicle: a safe and efficient natural ingredient for skin aging treatments. J Ethnopharmacol. 2016;193:607-616.
- Aggarwal BB, Yuan W, Li S, et al. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
- Mohanty C, Sahoo SK. Curcumin and its topical formulations for wound healing applications. Drug Discov Today. 2017;22:1582-1592.
- Gupta SC, Prasad S, Tyagi AK, et al. Neem (Azadirachta indica): an Indian traditional panacea with modern molecular basis. Phytomedicine. 2017;34:14-20.
- Choudhary D, Bhattacharyya S, Bose S. Efficacy and safety of ashwagandha (Withania somnifera (L.) Dunal) root extract in improving memory and cognitive functions. J Diet Suppl. 2017;14:599-612.
- Halder B, Singh S, Thakur SS. Withania somnifera root extract has potent cytotoxic effect against human malignant melanoma cells. PLoS One. 2015;10:E0137498.
- Nadeem M, Imran M. Promising features of Moringa oleifera oil: recent updates and perspectives. Lipids Health Dis. 2016;15:212.
- Sultan P, Jan A, Pervaiz Q. Phytochemical studies for quantitative estimation of iridoid glycosides in Picrorhiza kurroa Royle. Bot Stud. 2016;57:7.
- Gianfaldoni S, Wollina U, Tirant M, et al. Herbal compounds for the treatment of vitiligo: a review. Open Access Maced J Med Sci. 2018;6:203-207.
- Diamantoglou M, Platz J, Vienken J. Cellulose carbamates and derivatives as hemocompatible membrane materials for hemodialysis. Artif Organs. 1999;23:15-22.
- Respiratory syncytial virus (RSV). Centers for Disease Control and Prevention website. http://www.cdc.gov/rsv/research/us-surveillance.html. Updated June 26, 2018. Accessed February 1, 2019.
- Dembo G, Park SB, Kharasch ED. Central nervous system concentrations of cyclooxygenase-2 inhibitors in humans. Anesthesiology. 2005;102:409-415.
- Fong P. CFTR-SLC26 transporter interactions in epithelia. Biophys Rev. 2012;4:107-116.
- Liu Z. How cosmeceuticals companies get away with pseudoscience. Pacific Standard website. https://psmag.com/environment/cosmetic-companies-get-away-pseudoscience-placebo-week-92455. Published October 15, 2014. Accessed February 1, 2019.
- Beyerstein BL. Alternative medicine and common errors of reasoning. Acad Med. 2001;76:230-237.
- Topical antimicrobial drug products for over-the-counter human use. US Food and Drug Administration website. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=333.310. Accessed February 1, 2019.
- Natural personal care. Natural Products Association website. https://www.npanational.org/certifications/natural-seal/natural-seal-personal-care/. Accessed March 27, 2019.
- Natural Cosmetics Standard. GFaW Web site. https://gfaw.eu/en/ncs-for-all-who-love-nature-and-cosmetics/ncs-information-for-consumer/. Accessed February 1, 2019.
- Brown PN, Betz JM, Jasch F. How to qualify an analytical laboratory for analysis of herbal dietary ingredients and avoid using a “dry lab”: a review of issues related to using a contract analytical laboratory by industry, academia, and regulatory agencies. HerbalGram. 2013:52-59.
- Oh MJ, Cho YH, Cha SY, et al. Novel phytoceramides containing fatty acids of diverse chain lengths are better than a single C18-ceramide N-stearoyl phytosphingosine to improve the physiological properties of human stratum corneum. Clin Cosmet Investig Dermatol. 2017;10:363-371.
- Famurewa AC, Aja PM, Maduagwuna EK, et al. Antioxidant and anti-inflammatory effects of virgin coconut oil supplementation abrogate acute chemotherapy oxidative nephrotoxicity induced by anticancer drug methotrexate in rats. Biomed Pharmacother. 2017;96:905-911.
- Intahphuak S, Khonsung P, Panthong A. Anti-inflammatory, analgesic, and antipyretic activities of virgin coconut oil. Pharm Biol. 2010;48:151-157.
- McKenna PJ, Lehr GS, Leist P, et al. Antiseptic effectiveness with fibroblast preservation. Ann Plast Surg. 1991;27:265-268.
- Brockow K, Grabenhorst P, Abeck D, et al. Effect of gentian violet, corticosteroid and tar preparations in Staphylococcus aureus-colonized atopic eczema. Dermatology. 1999;199:231-236.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
- Misner BD. A novel aromatic oil compound inhibits microbial overgrowth on feet: a case study. J Int Soc Sports Nutr. 2007;4:3.
- D’Auria FD, Laino L, Strippoli V, et al. In vitro activity of tea tree oil against Candida albicans mycelial conversion and other pathogenic fungi. J Chemother. 2001;13:377-383.
- Fuchs-Tarlovsky V, Marquez-Barba MF, Sriram K. Probiotics in dermatologic practice. Nutrition. 2016;32:289-295.
- Bowe W, Patel NB, Logan AC. Acne vulgaris, probiotics and the gut-brain-skin axis: from anecdote to translational medicine. Benef Microbes. 2014;5:185-199.
- Baquerizo Nole KL, Yim E, Keri JE. Probiotics and prebiotics in dermatology. J Am Acad Dermatol. 2014;71:814-821.
- Saikaly SK, Khachemoune A. Honey and wound healing: an update. Am J Clin Dermatol. 2017;18:237-251.
- Aziz Z, Abdul Rasool Hassan B. The effects of honey compared to silver sulfadiazine for the treatment of burns: a systematic review of randomized controlled trials. Burns. 2017;43:50-57.
- FDA authority over cosmetics: how cosmetics are not FDA-approved, but are FDA-regulated. US Food and Drug AdministrationWeb site. https://www.fda.gov/cosmetics/guidanceregulation/lawsregulations/ucm074162.htm. Updated July 24, 2018. Accessed February 1, 2019.
- Wohlrab J. Topical preparations and their use in dermatology. J Dtsch Dermatol Ges. 2016;4:1061-1070
- Levin J, Momin SB. How much do we really know about our favorite cosmeceutical ingredients? J Clin Aesthet Dermatol. 2010;3:22-41.
- Ajala EO, Aberuagba F, Olaniyan AM, et al. Optimization of solvent extraction of shea butter (Vitellaria paradoxa) using response surface methodology and its characterization. J Food Sci Technol. 2016;53:730-738.
- Lin A, Nabatian A, Halverstam CP. Discovering black soap: a survey on the attitudes and practices of black soap users. J Clin Aesthet Dermatol. 2017;10:18-22.
- Lin TK, Zhong L, Santiago JL. Anti-inflammatory and skin barrier repair effects of topical application of some plant oils. Int J Mol Sci. 2017;19. pii:E70. doi:10.3390/ijms19010070.
- Dua K, Sheshala R, Ling TY, et al. Anti-inflammatory, antibacterial and analgesic potential of cocos nucifera linn.: a review. Antiinflamm Antiallergy Agents Med Chem. 2013;12:158-164.
- Hyun TK, Jang KI. Are berries useless by-products of ginseng? recent research on the potential health benefits of ginseng berry. EXCLI J. 2017;16:780-784.
- Truong VL, Bak MJ, Lee C, et al. Hair regenerative mechanisms of red ginseng oil and its major components in the testosterone-induced delay of anagen entry in C57BL/6 mice. Molecules. 2017;22. pii:E1505. doi:10.3390/molecules22091505.
- Hussain M, Habib Ur R, Akhtar L. Therapeutic benefits of green tea extract on various parameters in non-alcoholic fatty liver disease patients. Pak J Med Sci. 2017;33:931-936.
- Yi M, Fu J, Zhou L, et al. The effect of almond consumption on elements of endurance exercise performance in trained athletes. J Int Soc Sports Nutr. 2014;11:18.
- Sowndhararajan K, Deepa P, Kim M, et al. A review of the composition of the essential oils and biological activities of angelica species. Sci Pharm. 2017;85. pii:E33. doi:10.3390/scipharm85030033.
- Mahjour M, Khoushabi A, Noras M, et al. Effectiveness of Cicer arietinum in cutaneous problems: viewpoint of Avicenna and Razi. Curr Drug Discov Technol. 2018;15:243-250.
- Kanlayavattanakul M, Laurits N, Chaikul P. Jasmine rice panicle: a safe and efficient natural ingredient for skin aging treatments. J Ethnopharmacol. 2016;193:607-616.
- Aggarwal BB, Yuan W, Li S, et al. Curcumin-free turmeric exhibits anti-inflammatory and anticancer activities: identification of novel components of turmeric. Mol Nutr Food Res. 2013;57:1529-1542.
- Mohanty C, Sahoo SK. Curcumin and its topical formulations for wound healing applications. Drug Discov Today. 2017;22:1582-1592.
- Gupta SC, Prasad S, Tyagi AK, et al. Neem (Azadirachta indica): an Indian traditional panacea with modern molecular basis. Phytomedicine. 2017;34:14-20.
- Choudhary D, Bhattacharyya S, Bose S. Efficacy and safety of ashwagandha (Withania somnifera (L.) Dunal) root extract in improving memory and cognitive functions. J Diet Suppl. 2017;14:599-612.
- Halder B, Singh S, Thakur SS. Withania somnifera root extract has potent cytotoxic effect against human malignant melanoma cells. PLoS One. 2015;10:E0137498.
- Nadeem M, Imran M. Promising features of Moringa oleifera oil: recent updates and perspectives. Lipids Health Dis. 2016;15:212.
- Sultan P, Jan A, Pervaiz Q. Phytochemical studies for quantitative estimation of iridoid glycosides in Picrorhiza kurroa Royle. Bot Stud. 2016;57:7.
- Gianfaldoni S, Wollina U, Tirant M, et al. Herbal compounds for the treatment of vitiligo: a review. Open Access Maced J Med Sci. 2018;6:203-207.
- Diamantoglou M, Platz J, Vienken J. Cellulose carbamates and derivatives as hemocompatible membrane materials for hemodialysis. Artif Organs. 1999;23:15-22.
- Respiratory syncytial virus (RSV). Centers for Disease Control and Prevention website. http://www.cdc.gov/rsv/research/us-surveillance.html. Updated June 26, 2018. Accessed February 1, 2019.
- Dembo G, Park SB, Kharasch ED. Central nervous system concentrations of cyclooxygenase-2 inhibitors in humans. Anesthesiology. 2005;102:409-415.
- Fong P. CFTR-SLC26 transporter interactions in epithelia. Biophys Rev. 2012;4:107-116.
- Liu Z. How cosmeceuticals companies get away with pseudoscience. Pacific Standard website. https://psmag.com/environment/cosmetic-companies-get-away-pseudoscience-placebo-week-92455. Published October 15, 2014. Accessed February 1, 2019.
- Beyerstein BL. Alternative medicine and common errors of reasoning. Acad Med. 2001;76:230-237.
- Topical antimicrobial drug products for over-the-counter human use. US Food and Drug Administration website. https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=333.310. Accessed February 1, 2019.
- Natural personal care. Natural Products Association website. https://www.npanational.org/certifications/natural-seal/natural-seal-personal-care/. Accessed March 27, 2019.
- Natural Cosmetics Standard. GFaW Web site. https://gfaw.eu/en/ncs-for-all-who-love-nature-and-cosmetics/ncs-information-for-consumer/. Accessed February 1, 2019.
- Brown PN, Betz JM, Jasch F. How to qualify an analytical laboratory for analysis of herbal dietary ingredients and avoid using a “dry lab”: a review of issues related to using a contract analytical laboratory by industry, academia, and regulatory agencies. HerbalGram. 2013:52-59.
- Oh MJ, Cho YH, Cha SY, et al. Novel phytoceramides containing fatty acids of diverse chain lengths are better than a single C18-ceramide N-stearoyl phytosphingosine to improve the physiological properties of human stratum corneum. Clin Cosmet Investig Dermatol. 2017;10:363-371.
- Famurewa AC, Aja PM, Maduagwuna EK, et al. Antioxidant and anti-inflammatory effects of virgin coconut oil supplementation abrogate acute chemotherapy oxidative nephrotoxicity induced by anticancer drug methotrexate in rats. Biomed Pharmacother. 2017;96:905-911.
- Intahphuak S, Khonsung P, Panthong A. Anti-inflammatory, analgesic, and antipyretic activities of virgin coconut oil. Pharm Biol. 2010;48:151-157.
- McKenna PJ, Lehr GS, Leist P, et al. Antiseptic effectiveness with fibroblast preservation. Ann Plast Surg. 1991;27:265-268.
- Brockow K, Grabenhorst P, Abeck D, et al. Effect of gentian violet, corticosteroid and tar preparations in Staphylococcus aureus-colonized atopic eczema. Dermatology. 1999;199:231-236.
- Larson D, Jacob SE. Tea tree oil. Dermatitis. 2012;23:48-49.
- Misner BD. A novel aromatic oil compound inhibits microbial overgrowth on feet: a case study. J Int Soc Sports Nutr. 2007;4:3.
- D’Auria FD, Laino L, Strippoli V, et al. In vitro activity of tea tree oil against Candida albicans mycelial conversion and other pathogenic fungi. J Chemother. 2001;13:377-383.
- Fuchs-Tarlovsky V, Marquez-Barba MF, Sriram K. Probiotics in dermatologic practice. Nutrition. 2016;32:289-295.
- Bowe W, Patel NB, Logan AC. Acne vulgaris, probiotics and the gut-brain-skin axis: from anecdote to translational medicine. Benef Microbes. 2014;5:185-199.
- Baquerizo Nole KL, Yim E, Keri JE. Probiotics and prebiotics in dermatology. J Am Acad Dermatol. 2014;71:814-821.
- Saikaly SK, Khachemoune A. Honey and wound healing: an update. Am J Clin Dermatol. 2017;18:237-251.
- Aziz Z, Abdul Rasool Hassan B. The effects of honey compared to silver sulfadiazine for the treatment of burns: a systematic review of randomized controlled trials. Burns. 2017;43:50-57.
- FDA authority over cosmetics: how cosmetics are not FDA-approved, but are FDA-regulated. US Food and Drug AdministrationWeb site. https://www.fda.gov/cosmetics/guidanceregulation/lawsregulations/ucm074162.htm. Updated July 24, 2018. Accessed February 1, 2019.
- Wohlrab J. Topical preparations and their use in dermatology. J Dtsch Dermatol Ges. 2016;4:1061-1070
Practice Points
- Patients are increasingly interested in and asking for nature-based products and formulations to manage dermatologic conditions.
- Physicians can satisfy patient interests with nature-based formulations that are as beneficial or more so than synthetic formulations because of the physiologic activity of the ingredients within these formulations.
- Physicians should have resources available to them that adequately educate on nature-based ingredients and how to recommend them.
Aspirin for primary prevention: USPSTF recommendations for CVD and colorectal cancer
Which patients are likely to benefit from using aspirin for primary prevention? In this article, we review the evidence to date, summarized for primary care settings in guidelines issued by the US Preventive Services Task Force (USPSTF). We supplement this summary with a rundown of the risks associated with aspirin use. And then we wrap up by identifying a clinical decision tool that is available to help make personalized decisions in a busy clinic setting, where determining an individual’s potential cardiovascular benefits and bleeding risk can be challenging.
The “roadmap” from the guidelines. In 2014, after performing a review of the literature, the US Food and Drug Administration recommended against the routine use of aspirin for primary prevention of cardiovascular disease (CVD).1 In 2016, the USPSTF published 4 separate systematic reviews along with a decision analysis using a microsimulation model, which informed their position statement on aspirin for primary prevention.2-6 These USPSTF reviews and recommendations incorporated both CVD and colorectal cancer (CRC) benefits with the bleeding risks from aspirin. Generally, for individuals 50 to 59 years old, the benefits are deemed to outweigh the harms; shared decision making is advised with those 60 to 69 years of age. For patients younger than 50 or 70 and older, evidence is inconclusive.
The benefits of primary prevention with aspirin
Cardiovascular disease
The Antithrombotic Trialists’ (ATT) Collaboration was one of the first meta-analyses that addressed the benefit-to-harm balance and called into question the routine use of aspirin for primary prevention.7 The USPSTF systematic review included the studies from the ATT Collaboration as well as trials performed after its publication, bringing the total number of eligible randomized controlled trials reviewed to 11.2
The benefit of aspirin for primary prevention of nonfatal myocardial infarction (MI) has been shown in multiple randomized controlled trials. The USPSTF systematic review showed a statistically significant relative risk reduction of 17% in patients taking low-dose aspirin (≤ 100 mg; relative risk [RR] = 0.83; confidence interval [95% CI], 0.74-0.94), although the heterogeneity of the studies was high. The same low dose of aspirin showed a statistically significant reduction in nonfatal stroke (RR = 0.86; 95% CI, 0.76-0.98), although the same benefit was not observed when all doses of aspirin were included. Cardiovascular disease mortality and all-cause mortality were not statistically different for patients taking low-dose aspirin when compared with placebo (RR = 0.97; 95% CI, 0.85-1.10 for CVD mortality; RR = 0.95; 95% CI, 0.89-1.01 for all-cause mortality).2
One study of more than 14,000 older (≥ 60 years) Japanese patients showed a statistically significant reduction in nonfatal MI (hazard ratio [HR] = 0.53; 95% CI, 0.31-0.91, P = .02) and nonfatal strokes (HR = 0.57; 95% CI, 0.32-0.99; P = .04). The study was stopped early because at 5 years of follow-up there was no statistically significant difference in a composite primary outcome, which included death from cardiovascular causes, nonfatal MI, and nonfatal stroke (HR = 0.94; 95% CI, 0.77-1.15; P = .54).8
Several recent landmark studies have called into question the benefit of aspirin for cardiovascular primary prevention, especially in obese individuals, patients with diabetes, and the elderly. A meta-analysis of 10 trials showed that the effectiveness of aspirin doses between 75 mg and 100 mg for primary prevention decreased as weight increased; patients weighing 70 kg or more received no benefit.9 The ASCEND (A Study of Cardiovascular Events in Diabetes) trial included more than 15,000 patients with diabetes but no cardiovascular disease. Patients randomized to receive the low-dose aspirin did have fewer serious vascular events (incidence rate ratio [IRR] = 0.88; 95% CI, 0.79-0.97; P = .01), but they also had high risk of major bleeding events (IRR = 1.29; 95% CI, 1.09-1.52; P = .003).10 The ASPREE (Aspirin in Reducing Events in the Elderly) trial included more than 19,000 patients ages 70 years and older with no cardiovascular disease and compared low-dose aspirin to placebo. There was no statistically significant cardiovascular benefit, although there was an increase of major hemorrhage (HR = 1.38; 95% CI, 1.18-1.62; P < .001).11 The ARRIVE (A Randomized Trial of Induction Versus Expectant Management) trial included 12,546 moderate atherosclerotic CVD (ASCVD) risk patients. Although a per-protocol analysis showed a decrease in rates of fatal and nonfatal MI (HR = 0.53; 95% CI, 0.36-0.79; P = .0014), the more reliable intention-to-treat analysis showed no improvement for any outcomes.12
[polldaddy:10286821]
Colorectal cancer
The literature base on prevention of cancer has been growing rapidly. However, the deluge of findings over the past 2 decades of trials and analyses has also introduced ambiguity and, often, conflicting results. The first journal article suggesting aspirin for primary prevention of cancer, published in 1988, was a case-control study wherein a population with CRC was matched to controls to look for potential protective factors.13 The most notable finding was the CRC risk reduction for those taking aspirin or aspirin-containing medications. Since then numerous studies and analyses have explored aspirin’s potential in primary prevention of many types of cancer, with overall unclear findings as denoted in the 2016 USPSTF systemic reviews and recommendations.
Continue to: One major limiting factor...
One major limiting factor is that most data come from CVD prevention trials, and only a limited number of trials have focused specifically on cancer prevention. For the USPSTF, these data showed no statistically significant risk reduction in overall cancer mortality (RR = 0.96; 95% CI, 0.87-1.06) or in total cancer incidence (RR = 0.98; 95% CI, 0.93-1.04).4 Other ongoing trials may yield more definitive data.14
The particular interest in CRC was due to it being the first cancer found to be preventable with aspirin therapy. The USPSTF, while acknowledging the homogeneous nature of supporting studies, noted that their significant number and resulting evidence made CRC the only cancer warranting evaluation. Population studies have now shown more benefit than the few randomized control trials. The Women’s Health Study and the Physicians’ Health Study were both limited by their duration. But such studies conducted over a longer period revealed notable benefits in the second decade of use, with a statistically significant lower CRC incidence (RR = 0.60; 95% CI, 0.47-0.76). Additionally, CRC mortality at 20 years was decreased in patients taking aspirin regularly (RR = 0.67; 95% CI, 0.52-0.86).4 Multiple studies are in progress to better establish aspirin’s CRC benefit.
While not directly applicable to the general population, use of aspirin for patients with Lynch syndrome to prevent CRC has strong supporting evidence.15 Beyond CRC, there is nascent evidence from limited observational studies that aspirin may have a preventive effect on melanoma and ovarian and pancreatic cancers.16-18 Further studies or compilations of data would be needed to draw more significant conclusions on other types of cancers. Larger studies would prove more difficult to do, given the smaller incidences of these cancers.
Interestingly, a recent study showed that for individuals 70 years and older, aspirin might increase the risk for all-cause mortality, primarily due to increased cancer mortality across all types.19 Although this result was unexpected, caution should be used when prescribing aspirin particularly for patients 70 or older with active cancer.
A look at the harms associated with aspirin use
Aspirin has long been known to cause clinically significant bleeding. Aspirin inhibits platelet-derived cyclooxygenase-1 (COX-1), a potent vasoconstrictor, and thereby decreases platelet aggregation, reducing thromboembolic potential and prolonging bleeding time. These effects can confer health benefits but also carry the potential for risks. A decision to initiate aspirin therapy for primary prevention relies on an understanding of the benefit-to-harm balance.
Continue to: Initial aspirin studies...
Initial aspirin studies did not show a statistically significant increase in bleeding, likely due to too few events and inadequate powering. Subsequent meta-analyses from multiple evaluations have consistently shown bleeding to be a risk.3,7 The risk for bleeding with aspirin has also been examined in multiple cohort studies, which has helped elucidate the risk in greater detail.
Gastrointestinal bleeding
Epidemiologic data show that among patients who do not use nonsteroidal anti-inflammatory drugs (NSAIDs), the rate of upper gastrointestinal (GI) complications is 1 case per 1000 patient-years.20 Multiple studies have consistently shown that aspirin use increases the rate of significant upper GI bleeding over baseline risk (odds ratio [OR] = 1.54-1.58).3,21,22 Interestingly, these increases seem not to be influenced by other factors, such as comorbidities that increase the risk for ASCVD. Analysis of cancer prevention studies showed similar epidemiologic trends, with aspirin use exceeding a baseline bleeding risk of 0.7 cases of upper GI complications per 1000 patient-years (
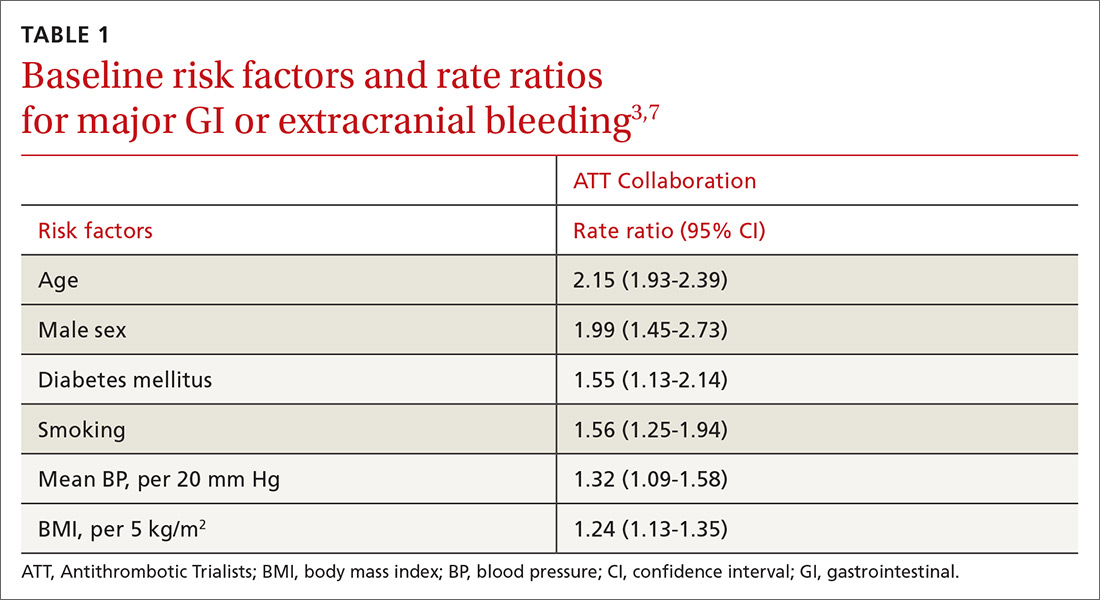
Other risk factors. Evaluation of risk factors for bleeding primarily comes from 2 studies.3,7 Most data concern the impact of individual factors on significant GI bleeding, with fewer data available for evaluating risk for intracerebral hemorrhage (ICH). Initial analysis of individual prospective studies showed little or no correlation between risk for bleeding and such factors as gender, age, or history of hypertension or ASCVD.21 Subsequent analysis of meta-data and large cohorts did show statistically significant impact on rates of bleeding across several factors (TABLE 13,7).
Of note is a large heterogeneous cohort study conducted in Spain. Data showed significant increases in baseline risk for GI bleeding in older men with a history of GI bleeding and NSAID use. The absolute risk for GI bleed in this group was potentially as high as 150 cases per 1000 patient-years, well above the risk level assumed for the average patient.24 A seemingly small OR of 1.5 could dramatically increase the absolute risk for bleeding in such patients, and it suggests that a generalized risk for bleeding probably shouldn’t be applied to all patients. Individuals may be better served by a baseline risk calculation reflecting multiple factors.
Intracerebral hemorrhage
Due to the comparatively uncommon nature of ICH, fewer data are available to support definitive conclusions about its increased risk with aspirin use. Aspirin use appeared to increase the risk for ICH with ratios between 1.27 and 1.32 in meta-analyses (measured as an OR or as an RR),3,7,21 with an IRR of 1.54 in a cohort study.22 The only statistically significant factors suspected to increase the risk of ICH at baseline were smoking (RR = 2.18) and mean BP > 20 mm Hg over normal (OR = 2.18). Age, gender, and diabetes all showed a nonsignificant trend toward risk increase.7
Continue to: Risk based on dose and formulation
Risk based on dose and formulation
The effect of aspirin dose and formulation on bleeding risk is uncertain. Some studies have shown an increased risk for bleeding with daily doses of aspirin ≥ 300 mg, while others have shown no significant increase in rates for bleeding with differing doses.21,25 Enteric coating does appear to lower the rates of gastric mucosal injury, although there are few data on the effect toward reducing clinically significant bleeding.26 Currently, several prospective studies are underway to help clarify the evidence.27
Putting it all together
For the general population, the evidence shows that the benefits and harms of aspirin for primary prevention are relatively even. The USPSTF guidelines are the first to recommend aspirin for both CVD and cancer prevention while taking into account the bleeding risk. According to the findings of the USPSTF, the balance of benefits and harms of aspirin use is contingent on 4 factors: age, baseline CVD risk, risk for bleeding, and preferences about taking aspirin.6 The complete recommendations from the USPSTF, along with other leading organizations, are outlined in TABLE 2.6,28-31
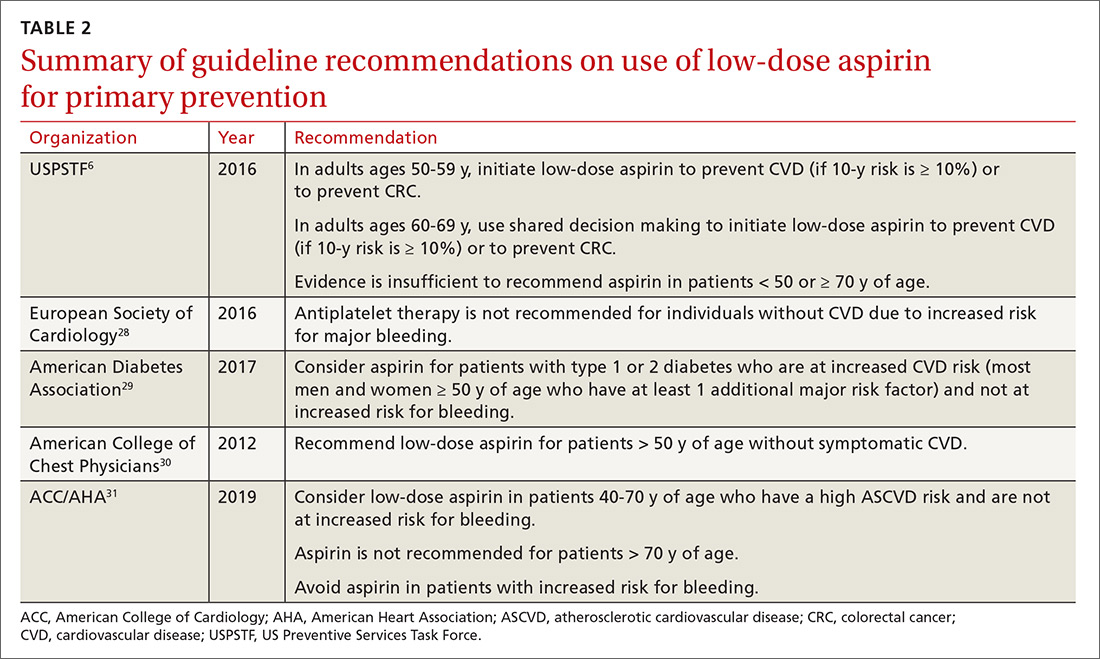
Applying the evidence and varying guidelines in practice can feel daunting. Some practical tools have been developed to help clinicians understand patients’ bleeding risk and potential benefits with aspirin use. One such tool is highlighted below. Others are also available, and each has its own strengths and weaknesses.
Aspirin-Guide (www.aspiringuide.com) is a Web-based clinical decision support tool with an associated mobile application. It uses internal calculators (including the pooled cohort calculator prepared jointly by the American College of Cardiology and the American Heart Association) to assess CVD risk as well as bleeding risk. This tool gives clinicians patient-specific numbers-needed-to-treat and numbers-needed-to-harm when considering starting aspirin for primary prevention. It gives specific recommendations for aspirin use based on the data entered, and it also gives providers information to help guide shared decision-making with patients.32 Unfortunately, this decision support tool and others do not take into account the data from the most recent trials, so they should be used with caution.
CORRESPONDENCE
LCDR Dustin K. Smith, DO, Naval Branch Clinic Diego Garcia, PSC 466, Box 301, FPO, AP 96595; [email protected].
1. FDA. Use of aspirin for primary prevention of heart attack and stroke. https://www.fda.gov/Drugs/ResourcesForYou/Consumers/ucm390574.htm. Accessed March 22, 2019.
2. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:804-813.
3. Whitlock EP, Burda BU, Williams SB, et al. Bleeding risks with aspirin use for primary prevention in adults: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:826-835.
4. Chubak J, Whitlock EP, Williams SB, et al. Aspirin for the prevention of cancer incidence and mortality: systematic evidence reviews for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:814-825.
5. Dehmer SP, Maciosek MV, Flottemesch TJ, et al. Aspirin for the primary prevention of cardiovascular disease and colorectal cancer: a decision analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:777-786.
6. Bibbins-Domingo K. Aspirin use for the primary prevention of cardiovascular disease and colorectal cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2016;164:836-845.
7. Baigent C, Blackwell L, Colins R, et al; Antithrombotic Trialists (ATT) Collaboration. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participation data from randomised trials. Lancet. 2009:373:1849-1860.
8. Ikeda Y, Shimada K, Teramoto T, et al. Low-dose aspirin for primary prevention of cardiovascular events in Japanese patients 60 years or older with atherosclerotic risk factors: a randomized clinical trial. JAMA. 2014;312:2510-2520.
9. Rothwell PM, Cook NR, Gaziano JM, et al. Effects of aspirin on risks of vascular events and cancer according to bodyweight and dose: analysis of individual patient data from randomised trials. Lancet. 2018;392:387-399.
10. Bowman L, Mafham M, Wallendszus K, et al; ASCEND Study Collaborative Group. Effects of aspirin for primary prevention in persons with diabetes mellitus. N Engl J Med. 2018;379:1529-1539.
11. McNeil JJ, Wolfe R, Woods RL, et al. Effect of aspirin on cardiovascular events and bleeding in the healthy elderly. N Engl J Med. 2018;379:1509-1518.
12. Gaziano JM, Brotons C, Coppolecchia R, et al. Use of aspirin to reduce risk of initial vascular events in patients at moderate risk of cardiovascular disease (ARRIVE): a randomised, double-blind, placebo-controlled trial. Lancet. 2018;392:1036-1046.
13. Kune GA, Kune S, Watson LF. Colorectal cancer risk, chronic illness, operations, and medications: case control results from Melbourne Colorectal Cancer Study. Cancer Res. 1988;48:4399-4404.
14. Sutcliffe P, Connock M, Gurung T, et al. Aspirin for prophylactic use in the primary prevention of cardiovascular disease and cancer: a systematic review and overview of reviews. Health Technol Assess. 2013;17:1-253.
15. Burn J, Gerdes AM, Macrae F, et al. Long-term effect of aspirin on cancer risk in carriers of hereditary colorectal cancer: an analysis from the CAPP2 randomised controlled trial. Lancet. 2011;378:2081-2087.
16. Gamba CA, Swetter SM, Stefanick ML, et al. Aspirin is associated with lower melanoma risk among postmenopausal Caucasian women: the Women’s Health Initiative. Cancer. 2013;119:1562-1569.
17. Trabert B, Ness RB, Lo-Ciganic WH, et al. Aspirin, nonaspirin nonsteroidal anti-inflammatory drug, and acetaminophen use and risk of invasive epithelial ovarian cancer: a pooled analysis in the Ovarian Cancer Association Consortium. J Natl Cancer Inst. 2014;106:djt431.
18. Risch H, Lu L, Streicher SA, et al. Aspirin use and reduced risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2016;26:68-74.
19. McNeil JJ, Nelson MR, Woods RL, et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-1528.
20. Hernández-Díaz S, Rodríguez LA. Incidence of serious upper gastrointestinal bleeding/perforation in the general population: review of epidemiologic studies. J Clin Epidemiol. 2002;55:157-163.
21. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Evidence Synthesis no 131. Rockville, MD: Agency for Healthcare Research and Quality; 2015. https://www.ncbi.nlm.nih.gov/books/NBK321623/. Accessed March 22, 2019.
22. De Berardis G, Lucisano G, D’Ettorre A, et al. Association of aspirin use with major bleeding in patients with and without diabetes. JAMA. 2012;307:2286-2294.
23. Thorat MA, Cuzick J. Prophylactic use of aspirin: systematic review of harms and approaches to mitigation in the general population. Eur J Epidemiol. 2015;30:5-18.
24. Hernández-Díaz S, García Rodríguez LA. Cardioprotective aspirin users and their excess risk of upper gastrointestinal complications. BMC Med. 2006;4:22.
25. Huang ES, Strate LL, Ho WW, et al. Long term use of aspirin and the risk of gastrointestinal bleeding. Am J Med. 2011:124;426-433.
26. Walker J, Robinson J, Stewart J, et al. Does enteric-coated aspirin result in a lower incidence of gastrointestinal complications compared to normal aspirin? Interact Cardiovasc Thorac Surg. 2007:6;519-522.
27. NIH. Aspirin dosing: a patient-centric trial assessing benefits and long-term effectiveness (ADAPTABLE). https://clinicaltrials.gov/ct2/show/NCT02697916. Accessed March 22, 2019.
28. Piepoli MF, Hoes AW, Agewall S, et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice: the Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice. Eur Heart J. 2016;37:2315-2381.
29. ADA. Standards of medical care in diabetes – 2017. Diabetes Care. 2017;40(suppl 1). http://care.diabetesjournals.org/content/diacare/suppl/2016/12/15/40.Supplement_1.DC1/DC_40_S1_final.pdf. Accessed March 22, 2019.
30. Vandvik PO, Lincoff AM, Gore JM, et al. Primary and secondary prevention of cardiovascular disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e637S-e668S.
31. Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease. J Am Col Cardiol. 2019. doi: https://doi.org/10.1016/j.jacc.2019.03.010. Accessed March 22, 2019.
32. Mora S, Manson JE. Aspirin for primary prevention of atherosclerotic cardiovascular disease: advances in diagnosis and treatment. JAMA Intern Med. 2016;176:1195-1204.
Which patients are likely to benefit from using aspirin for primary prevention? In this article, we review the evidence to date, summarized for primary care settings in guidelines issued by the US Preventive Services Task Force (USPSTF). We supplement this summary with a rundown of the risks associated with aspirin use. And then we wrap up by identifying a clinical decision tool that is available to help make personalized decisions in a busy clinic setting, where determining an individual’s potential cardiovascular benefits and bleeding risk can be challenging.
The “roadmap” from the guidelines. In 2014, after performing a review of the literature, the US Food and Drug Administration recommended against the routine use of aspirin for primary prevention of cardiovascular disease (CVD).1 In 2016, the USPSTF published 4 separate systematic reviews along with a decision analysis using a microsimulation model, which informed their position statement on aspirin for primary prevention.2-6 These USPSTF reviews and recommendations incorporated both CVD and colorectal cancer (CRC) benefits with the bleeding risks from aspirin. Generally, for individuals 50 to 59 years old, the benefits are deemed to outweigh the harms; shared decision making is advised with those 60 to 69 years of age. For patients younger than 50 or 70 and older, evidence is inconclusive.
The benefits of primary prevention with aspirin
Cardiovascular disease
The Antithrombotic Trialists’ (ATT) Collaboration was one of the first meta-analyses that addressed the benefit-to-harm balance and called into question the routine use of aspirin for primary prevention.7 The USPSTF systematic review included the studies from the ATT Collaboration as well as trials performed after its publication, bringing the total number of eligible randomized controlled trials reviewed to 11.2
The benefit of aspirin for primary prevention of nonfatal myocardial infarction (MI) has been shown in multiple randomized controlled trials. The USPSTF systematic review showed a statistically significant relative risk reduction of 17% in patients taking low-dose aspirin (≤ 100 mg; relative risk [RR] = 0.83; confidence interval [95% CI], 0.74-0.94), although the heterogeneity of the studies was high. The same low dose of aspirin showed a statistically significant reduction in nonfatal stroke (RR = 0.86; 95% CI, 0.76-0.98), although the same benefit was not observed when all doses of aspirin were included. Cardiovascular disease mortality and all-cause mortality were not statistically different for patients taking low-dose aspirin when compared with placebo (RR = 0.97; 95% CI, 0.85-1.10 for CVD mortality; RR = 0.95; 95% CI, 0.89-1.01 for all-cause mortality).2
One study of more than 14,000 older (≥ 60 years) Japanese patients showed a statistically significant reduction in nonfatal MI (hazard ratio [HR] = 0.53; 95% CI, 0.31-0.91, P = .02) and nonfatal strokes (HR = 0.57; 95% CI, 0.32-0.99; P = .04). The study was stopped early because at 5 years of follow-up there was no statistically significant difference in a composite primary outcome, which included death from cardiovascular causes, nonfatal MI, and nonfatal stroke (HR = 0.94; 95% CI, 0.77-1.15; P = .54).8
Several recent landmark studies have called into question the benefit of aspirin for cardiovascular primary prevention, especially in obese individuals, patients with diabetes, and the elderly. A meta-analysis of 10 trials showed that the effectiveness of aspirin doses between 75 mg and 100 mg for primary prevention decreased as weight increased; patients weighing 70 kg or more received no benefit.9 The ASCEND (A Study of Cardiovascular Events in Diabetes) trial included more than 15,000 patients with diabetes but no cardiovascular disease. Patients randomized to receive the low-dose aspirin did have fewer serious vascular events (incidence rate ratio [IRR] = 0.88; 95% CI, 0.79-0.97; P = .01), but they also had high risk of major bleeding events (IRR = 1.29; 95% CI, 1.09-1.52; P = .003).10 The ASPREE (Aspirin in Reducing Events in the Elderly) trial included more than 19,000 patients ages 70 years and older with no cardiovascular disease and compared low-dose aspirin to placebo. There was no statistically significant cardiovascular benefit, although there was an increase of major hemorrhage (HR = 1.38; 95% CI, 1.18-1.62; P < .001).11 The ARRIVE (A Randomized Trial of Induction Versus Expectant Management) trial included 12,546 moderate atherosclerotic CVD (ASCVD) risk patients. Although a per-protocol analysis showed a decrease in rates of fatal and nonfatal MI (HR = 0.53; 95% CI, 0.36-0.79; P = .0014), the more reliable intention-to-treat analysis showed no improvement for any outcomes.12
[polldaddy:10286821]
Colorectal cancer
The literature base on prevention of cancer has been growing rapidly. However, the deluge of findings over the past 2 decades of trials and analyses has also introduced ambiguity and, often, conflicting results. The first journal article suggesting aspirin for primary prevention of cancer, published in 1988, was a case-control study wherein a population with CRC was matched to controls to look for potential protective factors.13 The most notable finding was the CRC risk reduction for those taking aspirin or aspirin-containing medications. Since then numerous studies and analyses have explored aspirin’s potential in primary prevention of many types of cancer, with overall unclear findings as denoted in the 2016 USPSTF systemic reviews and recommendations.
Continue to: One major limiting factor...
One major limiting factor is that most data come from CVD prevention trials, and only a limited number of trials have focused specifically on cancer prevention. For the USPSTF, these data showed no statistically significant risk reduction in overall cancer mortality (RR = 0.96; 95% CI, 0.87-1.06) or in total cancer incidence (RR = 0.98; 95% CI, 0.93-1.04).4 Other ongoing trials may yield more definitive data.14
The particular interest in CRC was due to it being the first cancer found to be preventable with aspirin therapy. The USPSTF, while acknowledging the homogeneous nature of supporting studies, noted that their significant number and resulting evidence made CRC the only cancer warranting evaluation. Population studies have now shown more benefit than the few randomized control trials. The Women’s Health Study and the Physicians’ Health Study were both limited by their duration. But such studies conducted over a longer period revealed notable benefits in the second decade of use, with a statistically significant lower CRC incidence (RR = 0.60; 95% CI, 0.47-0.76). Additionally, CRC mortality at 20 years was decreased in patients taking aspirin regularly (RR = 0.67; 95% CI, 0.52-0.86).4 Multiple studies are in progress to better establish aspirin’s CRC benefit.
While not directly applicable to the general population, use of aspirin for patients with Lynch syndrome to prevent CRC has strong supporting evidence.15 Beyond CRC, there is nascent evidence from limited observational studies that aspirin may have a preventive effect on melanoma and ovarian and pancreatic cancers.16-18 Further studies or compilations of data would be needed to draw more significant conclusions on other types of cancers. Larger studies would prove more difficult to do, given the smaller incidences of these cancers.
Interestingly, a recent study showed that for individuals 70 years and older, aspirin might increase the risk for all-cause mortality, primarily due to increased cancer mortality across all types.19 Although this result was unexpected, caution should be used when prescribing aspirin particularly for patients 70 or older with active cancer.
A look at the harms associated with aspirin use
Aspirin has long been known to cause clinically significant bleeding. Aspirin inhibits platelet-derived cyclooxygenase-1 (COX-1), a potent vasoconstrictor, and thereby decreases platelet aggregation, reducing thromboembolic potential and prolonging bleeding time. These effects can confer health benefits but also carry the potential for risks. A decision to initiate aspirin therapy for primary prevention relies on an understanding of the benefit-to-harm balance.
Continue to: Initial aspirin studies...
Initial aspirin studies did not show a statistically significant increase in bleeding, likely due to too few events and inadequate powering. Subsequent meta-analyses from multiple evaluations have consistently shown bleeding to be a risk.3,7 The risk for bleeding with aspirin has also been examined in multiple cohort studies, which has helped elucidate the risk in greater detail.
Gastrointestinal bleeding
Epidemiologic data show that among patients who do not use nonsteroidal anti-inflammatory drugs (NSAIDs), the rate of upper gastrointestinal (GI) complications is 1 case per 1000 patient-years.20 Multiple studies have consistently shown that aspirin use increases the rate of significant upper GI bleeding over baseline risk (odds ratio [OR] = 1.54-1.58).3,21,22 Interestingly, these increases seem not to be influenced by other factors, such as comorbidities that increase the risk for ASCVD. Analysis of cancer prevention studies showed similar epidemiologic trends, with aspirin use exceeding a baseline bleeding risk of 0.7 cases of upper GI complications per 1000 patient-years (

Other risk factors. Evaluation of risk factors for bleeding primarily comes from 2 studies.3,7 Most data concern the impact of individual factors on significant GI bleeding, with fewer data available for evaluating risk for intracerebral hemorrhage (ICH). Initial analysis of individual prospective studies showed little or no correlation between risk for bleeding and such factors as gender, age, or history of hypertension or ASCVD.21 Subsequent analysis of meta-data and large cohorts did show statistically significant impact on rates of bleeding across several factors (TABLE 13,7).
Of note is a large heterogeneous cohort study conducted in Spain. Data showed significant increases in baseline risk for GI bleeding in older men with a history of GI bleeding and NSAID use. The absolute risk for GI bleed in this group was potentially as high as 150 cases per 1000 patient-years, well above the risk level assumed for the average patient.24 A seemingly small OR of 1.5 could dramatically increase the absolute risk for bleeding in such patients, and it suggests that a generalized risk for bleeding probably shouldn’t be applied to all patients. Individuals may be better served by a baseline risk calculation reflecting multiple factors.
Intracerebral hemorrhage
Due to the comparatively uncommon nature of ICH, fewer data are available to support definitive conclusions about its increased risk with aspirin use. Aspirin use appeared to increase the risk for ICH with ratios between 1.27 and 1.32 in meta-analyses (measured as an OR or as an RR),3,7,21 with an IRR of 1.54 in a cohort study.22 The only statistically significant factors suspected to increase the risk of ICH at baseline were smoking (RR = 2.18) and mean BP > 20 mm Hg over normal (OR = 2.18). Age, gender, and diabetes all showed a nonsignificant trend toward risk increase.7
Continue to: Risk based on dose and formulation
Risk based on dose and formulation
The effect of aspirin dose and formulation on bleeding risk is uncertain. Some studies have shown an increased risk for bleeding with daily doses of aspirin ≥ 300 mg, while others have shown no significant increase in rates for bleeding with differing doses.21,25 Enteric coating does appear to lower the rates of gastric mucosal injury, although there are few data on the effect toward reducing clinically significant bleeding.26 Currently, several prospective studies are underway to help clarify the evidence.27
Putting it all together
For the general population, the evidence shows that the benefits and harms of aspirin for primary prevention are relatively even. The USPSTF guidelines are the first to recommend aspirin for both CVD and cancer prevention while taking into account the bleeding risk. According to the findings of the USPSTF, the balance of benefits and harms of aspirin use is contingent on 4 factors: age, baseline CVD risk, risk for bleeding, and preferences about taking aspirin.6 The complete recommendations from the USPSTF, along with other leading organizations, are outlined in TABLE 2.6,28-31

Applying the evidence and varying guidelines in practice can feel daunting. Some practical tools have been developed to help clinicians understand patients’ bleeding risk and potential benefits with aspirin use. One such tool is highlighted below. Others are also available, and each has its own strengths and weaknesses.
Aspirin-Guide (www.aspiringuide.com) is a Web-based clinical decision support tool with an associated mobile application. It uses internal calculators (including the pooled cohort calculator prepared jointly by the American College of Cardiology and the American Heart Association) to assess CVD risk as well as bleeding risk. This tool gives clinicians patient-specific numbers-needed-to-treat and numbers-needed-to-harm when considering starting aspirin for primary prevention. It gives specific recommendations for aspirin use based on the data entered, and it also gives providers information to help guide shared decision-making with patients.32 Unfortunately, this decision support tool and others do not take into account the data from the most recent trials, so they should be used with caution.
CORRESPONDENCE
LCDR Dustin K. Smith, DO, Naval Branch Clinic Diego Garcia, PSC 466, Box 301, FPO, AP 96595; [email protected].
Which patients are likely to benefit from using aspirin for primary prevention? In this article, we review the evidence to date, summarized for primary care settings in guidelines issued by the US Preventive Services Task Force (USPSTF). We supplement this summary with a rundown of the risks associated with aspirin use. And then we wrap up by identifying a clinical decision tool that is available to help make personalized decisions in a busy clinic setting, where determining an individual’s potential cardiovascular benefits and bleeding risk can be challenging.
The “roadmap” from the guidelines. In 2014, after performing a review of the literature, the US Food and Drug Administration recommended against the routine use of aspirin for primary prevention of cardiovascular disease (CVD).1 In 2016, the USPSTF published 4 separate systematic reviews along with a decision analysis using a microsimulation model, which informed their position statement on aspirin for primary prevention.2-6 These USPSTF reviews and recommendations incorporated both CVD and colorectal cancer (CRC) benefits with the bleeding risks from aspirin. Generally, for individuals 50 to 59 years old, the benefits are deemed to outweigh the harms; shared decision making is advised with those 60 to 69 years of age. For patients younger than 50 or 70 and older, evidence is inconclusive.
The benefits of primary prevention with aspirin
Cardiovascular disease
The Antithrombotic Trialists’ (ATT) Collaboration was one of the first meta-analyses that addressed the benefit-to-harm balance and called into question the routine use of aspirin for primary prevention.7 The USPSTF systematic review included the studies from the ATT Collaboration as well as trials performed after its publication, bringing the total number of eligible randomized controlled trials reviewed to 11.2
The benefit of aspirin for primary prevention of nonfatal myocardial infarction (MI) has been shown in multiple randomized controlled trials. The USPSTF systematic review showed a statistically significant relative risk reduction of 17% in patients taking low-dose aspirin (≤ 100 mg; relative risk [RR] = 0.83; confidence interval [95% CI], 0.74-0.94), although the heterogeneity of the studies was high. The same low dose of aspirin showed a statistically significant reduction in nonfatal stroke (RR = 0.86; 95% CI, 0.76-0.98), although the same benefit was not observed when all doses of aspirin were included. Cardiovascular disease mortality and all-cause mortality were not statistically different for patients taking low-dose aspirin when compared with placebo (RR = 0.97; 95% CI, 0.85-1.10 for CVD mortality; RR = 0.95; 95% CI, 0.89-1.01 for all-cause mortality).2
One study of more than 14,000 older (≥ 60 years) Japanese patients showed a statistically significant reduction in nonfatal MI (hazard ratio [HR] = 0.53; 95% CI, 0.31-0.91, P = .02) and nonfatal strokes (HR = 0.57; 95% CI, 0.32-0.99; P = .04). The study was stopped early because at 5 years of follow-up there was no statistically significant difference in a composite primary outcome, which included death from cardiovascular causes, nonfatal MI, and nonfatal stroke (HR = 0.94; 95% CI, 0.77-1.15; P = .54).8
Several recent landmark studies have called into question the benefit of aspirin for cardiovascular primary prevention, especially in obese individuals, patients with diabetes, and the elderly. A meta-analysis of 10 trials showed that the effectiveness of aspirin doses between 75 mg and 100 mg for primary prevention decreased as weight increased; patients weighing 70 kg or more received no benefit.9 The ASCEND (A Study of Cardiovascular Events in Diabetes) trial included more than 15,000 patients with diabetes but no cardiovascular disease. Patients randomized to receive the low-dose aspirin did have fewer serious vascular events (incidence rate ratio [IRR] = 0.88; 95% CI, 0.79-0.97; P = .01), but they also had high risk of major bleeding events (IRR = 1.29; 95% CI, 1.09-1.52; P = .003).10 The ASPREE (Aspirin in Reducing Events in the Elderly) trial included more than 19,000 patients ages 70 years and older with no cardiovascular disease and compared low-dose aspirin to placebo. There was no statistically significant cardiovascular benefit, although there was an increase of major hemorrhage (HR = 1.38; 95% CI, 1.18-1.62; P < .001).11 The ARRIVE (A Randomized Trial of Induction Versus Expectant Management) trial included 12,546 moderate atherosclerotic CVD (ASCVD) risk patients. Although a per-protocol analysis showed a decrease in rates of fatal and nonfatal MI (HR = 0.53; 95% CI, 0.36-0.79; P = .0014), the more reliable intention-to-treat analysis showed no improvement for any outcomes.12
[polldaddy:10286821]
Colorectal cancer
The literature base on prevention of cancer has been growing rapidly. However, the deluge of findings over the past 2 decades of trials and analyses has also introduced ambiguity and, often, conflicting results. The first journal article suggesting aspirin for primary prevention of cancer, published in 1988, was a case-control study wherein a population with CRC was matched to controls to look for potential protective factors.13 The most notable finding was the CRC risk reduction for those taking aspirin or aspirin-containing medications. Since then numerous studies and analyses have explored aspirin’s potential in primary prevention of many types of cancer, with overall unclear findings as denoted in the 2016 USPSTF systemic reviews and recommendations.
Continue to: One major limiting factor...
One major limiting factor is that most data come from CVD prevention trials, and only a limited number of trials have focused specifically on cancer prevention. For the USPSTF, these data showed no statistically significant risk reduction in overall cancer mortality (RR = 0.96; 95% CI, 0.87-1.06) or in total cancer incidence (RR = 0.98; 95% CI, 0.93-1.04).4 Other ongoing trials may yield more definitive data.14
The particular interest in CRC was due to it being the first cancer found to be preventable with aspirin therapy. The USPSTF, while acknowledging the homogeneous nature of supporting studies, noted that their significant number and resulting evidence made CRC the only cancer warranting evaluation. Population studies have now shown more benefit than the few randomized control trials. The Women’s Health Study and the Physicians’ Health Study were both limited by their duration. But such studies conducted over a longer period revealed notable benefits in the second decade of use, with a statistically significant lower CRC incidence (RR = 0.60; 95% CI, 0.47-0.76). Additionally, CRC mortality at 20 years was decreased in patients taking aspirin regularly (RR = 0.67; 95% CI, 0.52-0.86).4 Multiple studies are in progress to better establish aspirin’s CRC benefit.
While not directly applicable to the general population, use of aspirin for patients with Lynch syndrome to prevent CRC has strong supporting evidence.15 Beyond CRC, there is nascent evidence from limited observational studies that aspirin may have a preventive effect on melanoma and ovarian and pancreatic cancers.16-18 Further studies or compilations of data would be needed to draw more significant conclusions on other types of cancers. Larger studies would prove more difficult to do, given the smaller incidences of these cancers.
Interestingly, a recent study showed that for individuals 70 years and older, aspirin might increase the risk for all-cause mortality, primarily due to increased cancer mortality across all types.19 Although this result was unexpected, caution should be used when prescribing aspirin particularly for patients 70 or older with active cancer.
A look at the harms associated with aspirin use
Aspirin has long been known to cause clinically significant bleeding. Aspirin inhibits platelet-derived cyclooxygenase-1 (COX-1), a potent vasoconstrictor, and thereby decreases platelet aggregation, reducing thromboembolic potential and prolonging bleeding time. These effects can confer health benefits but also carry the potential for risks. A decision to initiate aspirin therapy for primary prevention relies on an understanding of the benefit-to-harm balance.
Continue to: Initial aspirin studies...
Initial aspirin studies did not show a statistically significant increase in bleeding, likely due to too few events and inadequate powering. Subsequent meta-analyses from multiple evaluations have consistently shown bleeding to be a risk.3,7 The risk for bleeding with aspirin has also been examined in multiple cohort studies, which has helped elucidate the risk in greater detail.
Gastrointestinal bleeding
Epidemiologic data show that among patients who do not use nonsteroidal anti-inflammatory drugs (NSAIDs), the rate of upper gastrointestinal (GI) complications is 1 case per 1000 patient-years.20 Multiple studies have consistently shown that aspirin use increases the rate of significant upper GI bleeding over baseline risk (odds ratio [OR] = 1.54-1.58).3,21,22 Interestingly, these increases seem not to be influenced by other factors, such as comorbidities that increase the risk for ASCVD. Analysis of cancer prevention studies showed similar epidemiologic trends, with aspirin use exceeding a baseline bleeding risk of 0.7 cases of upper GI complications per 1000 patient-years (

Other risk factors. Evaluation of risk factors for bleeding primarily comes from 2 studies.3,7 Most data concern the impact of individual factors on significant GI bleeding, with fewer data available for evaluating risk for intracerebral hemorrhage (ICH). Initial analysis of individual prospective studies showed little or no correlation between risk for bleeding and such factors as gender, age, or history of hypertension or ASCVD.21 Subsequent analysis of meta-data and large cohorts did show statistically significant impact on rates of bleeding across several factors (TABLE 13,7).
Of note is a large heterogeneous cohort study conducted in Spain. Data showed significant increases in baseline risk for GI bleeding in older men with a history of GI bleeding and NSAID use. The absolute risk for GI bleed in this group was potentially as high as 150 cases per 1000 patient-years, well above the risk level assumed for the average patient.24 A seemingly small OR of 1.5 could dramatically increase the absolute risk for bleeding in such patients, and it suggests that a generalized risk for bleeding probably shouldn’t be applied to all patients. Individuals may be better served by a baseline risk calculation reflecting multiple factors.
Intracerebral hemorrhage
Due to the comparatively uncommon nature of ICH, fewer data are available to support definitive conclusions about its increased risk with aspirin use. Aspirin use appeared to increase the risk for ICH with ratios between 1.27 and 1.32 in meta-analyses (measured as an OR or as an RR),3,7,21 with an IRR of 1.54 in a cohort study.22 The only statistically significant factors suspected to increase the risk of ICH at baseline were smoking (RR = 2.18) and mean BP > 20 mm Hg over normal (OR = 2.18). Age, gender, and diabetes all showed a nonsignificant trend toward risk increase.7
Continue to: Risk based on dose and formulation
Risk based on dose and formulation
The effect of aspirin dose and formulation on bleeding risk is uncertain. Some studies have shown an increased risk for bleeding with daily doses of aspirin ≥ 300 mg, while others have shown no significant increase in rates for bleeding with differing doses.21,25 Enteric coating does appear to lower the rates of gastric mucosal injury, although there are few data on the effect toward reducing clinically significant bleeding.26 Currently, several prospective studies are underway to help clarify the evidence.27
Putting it all together
For the general population, the evidence shows that the benefits and harms of aspirin for primary prevention are relatively even. The USPSTF guidelines are the first to recommend aspirin for both CVD and cancer prevention while taking into account the bleeding risk. According to the findings of the USPSTF, the balance of benefits and harms of aspirin use is contingent on 4 factors: age, baseline CVD risk, risk for bleeding, and preferences about taking aspirin.6 The complete recommendations from the USPSTF, along with other leading organizations, are outlined in TABLE 2.6,28-31

Applying the evidence and varying guidelines in practice can feel daunting. Some practical tools have been developed to help clinicians understand patients’ bleeding risk and potential benefits with aspirin use. One such tool is highlighted below. Others are also available, and each has its own strengths and weaknesses.
Aspirin-Guide (www.aspiringuide.com) is a Web-based clinical decision support tool with an associated mobile application. It uses internal calculators (including the pooled cohort calculator prepared jointly by the American College of Cardiology and the American Heart Association) to assess CVD risk as well as bleeding risk. This tool gives clinicians patient-specific numbers-needed-to-treat and numbers-needed-to-harm when considering starting aspirin for primary prevention. It gives specific recommendations for aspirin use based on the data entered, and it also gives providers information to help guide shared decision-making with patients.32 Unfortunately, this decision support tool and others do not take into account the data from the most recent trials, so they should be used with caution.
CORRESPONDENCE
LCDR Dustin K. Smith, DO, Naval Branch Clinic Diego Garcia, PSC 466, Box 301, FPO, AP 96595; [email protected].
1. FDA. Use of aspirin for primary prevention of heart attack and stroke. https://www.fda.gov/Drugs/ResourcesForYou/Consumers/ucm390574.htm. Accessed March 22, 2019.
2. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:804-813.
3. Whitlock EP, Burda BU, Williams SB, et al. Bleeding risks with aspirin use for primary prevention in adults: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:826-835.
4. Chubak J, Whitlock EP, Williams SB, et al. Aspirin for the prevention of cancer incidence and mortality: systematic evidence reviews for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:814-825.
5. Dehmer SP, Maciosek MV, Flottemesch TJ, et al. Aspirin for the primary prevention of cardiovascular disease and colorectal cancer: a decision analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:777-786.
6. Bibbins-Domingo K. Aspirin use for the primary prevention of cardiovascular disease and colorectal cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2016;164:836-845.
7. Baigent C, Blackwell L, Colins R, et al; Antithrombotic Trialists (ATT) Collaboration. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participation data from randomised trials. Lancet. 2009:373:1849-1860.
8. Ikeda Y, Shimada K, Teramoto T, et al. Low-dose aspirin for primary prevention of cardiovascular events in Japanese patients 60 years or older with atherosclerotic risk factors: a randomized clinical trial. JAMA. 2014;312:2510-2520.
9. Rothwell PM, Cook NR, Gaziano JM, et al. Effects of aspirin on risks of vascular events and cancer according to bodyweight and dose: analysis of individual patient data from randomised trials. Lancet. 2018;392:387-399.
10. Bowman L, Mafham M, Wallendszus K, et al; ASCEND Study Collaborative Group. Effects of aspirin for primary prevention in persons with diabetes mellitus. N Engl J Med. 2018;379:1529-1539.
11. McNeil JJ, Wolfe R, Woods RL, et al. Effect of aspirin on cardiovascular events and bleeding in the healthy elderly. N Engl J Med. 2018;379:1509-1518.
12. Gaziano JM, Brotons C, Coppolecchia R, et al. Use of aspirin to reduce risk of initial vascular events in patients at moderate risk of cardiovascular disease (ARRIVE): a randomised, double-blind, placebo-controlled trial. Lancet. 2018;392:1036-1046.
13. Kune GA, Kune S, Watson LF. Colorectal cancer risk, chronic illness, operations, and medications: case control results from Melbourne Colorectal Cancer Study. Cancer Res. 1988;48:4399-4404.
14. Sutcliffe P, Connock M, Gurung T, et al. Aspirin for prophylactic use in the primary prevention of cardiovascular disease and cancer: a systematic review and overview of reviews. Health Technol Assess. 2013;17:1-253.
15. Burn J, Gerdes AM, Macrae F, et al. Long-term effect of aspirin on cancer risk in carriers of hereditary colorectal cancer: an analysis from the CAPP2 randomised controlled trial. Lancet. 2011;378:2081-2087.
16. Gamba CA, Swetter SM, Stefanick ML, et al. Aspirin is associated with lower melanoma risk among postmenopausal Caucasian women: the Women’s Health Initiative. Cancer. 2013;119:1562-1569.
17. Trabert B, Ness RB, Lo-Ciganic WH, et al. Aspirin, nonaspirin nonsteroidal anti-inflammatory drug, and acetaminophen use and risk of invasive epithelial ovarian cancer: a pooled analysis in the Ovarian Cancer Association Consortium. J Natl Cancer Inst. 2014;106:djt431.
18. Risch H, Lu L, Streicher SA, et al. Aspirin use and reduced risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2016;26:68-74.
19. McNeil JJ, Nelson MR, Woods RL, et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-1528.
20. Hernández-Díaz S, Rodríguez LA. Incidence of serious upper gastrointestinal bleeding/perforation in the general population: review of epidemiologic studies. J Clin Epidemiol. 2002;55:157-163.
21. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Evidence Synthesis no 131. Rockville, MD: Agency for Healthcare Research and Quality; 2015. https://www.ncbi.nlm.nih.gov/books/NBK321623/. Accessed March 22, 2019.
22. De Berardis G, Lucisano G, D’Ettorre A, et al. Association of aspirin use with major bleeding in patients with and without diabetes. JAMA. 2012;307:2286-2294.
23. Thorat MA, Cuzick J. Prophylactic use of aspirin: systematic review of harms and approaches to mitigation in the general population. Eur J Epidemiol. 2015;30:5-18.
24. Hernández-Díaz S, García Rodríguez LA. Cardioprotective aspirin users and their excess risk of upper gastrointestinal complications. BMC Med. 2006;4:22.
25. Huang ES, Strate LL, Ho WW, et al. Long term use of aspirin and the risk of gastrointestinal bleeding. Am J Med. 2011:124;426-433.
26. Walker J, Robinson J, Stewart J, et al. Does enteric-coated aspirin result in a lower incidence of gastrointestinal complications compared to normal aspirin? Interact Cardiovasc Thorac Surg. 2007:6;519-522.
27. NIH. Aspirin dosing: a patient-centric trial assessing benefits and long-term effectiveness (ADAPTABLE). https://clinicaltrials.gov/ct2/show/NCT02697916. Accessed March 22, 2019.
28. Piepoli MF, Hoes AW, Agewall S, et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice: the Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice. Eur Heart J. 2016;37:2315-2381.
29. ADA. Standards of medical care in diabetes – 2017. Diabetes Care. 2017;40(suppl 1). http://care.diabetesjournals.org/content/diacare/suppl/2016/12/15/40.Supplement_1.DC1/DC_40_S1_final.pdf. Accessed March 22, 2019.
30. Vandvik PO, Lincoff AM, Gore JM, et al. Primary and secondary prevention of cardiovascular disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e637S-e668S.
31. Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease. J Am Col Cardiol. 2019. doi: https://doi.org/10.1016/j.jacc.2019.03.010. Accessed March 22, 2019.
32. Mora S, Manson JE. Aspirin for primary prevention of atherosclerotic cardiovascular disease: advances in diagnosis and treatment. JAMA Intern Med. 2016;176:1195-1204.
1. FDA. Use of aspirin for primary prevention of heart attack and stroke. https://www.fda.gov/Drugs/ResourcesForYou/Consumers/ucm390574.htm. Accessed March 22, 2019.
2. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:804-813.
3. Whitlock EP, Burda BU, Williams SB, et al. Bleeding risks with aspirin use for primary prevention in adults: a systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:826-835.
4. Chubak J, Whitlock EP, Williams SB, et al. Aspirin for the prevention of cancer incidence and mortality: systematic evidence reviews for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:814-825.
5. Dehmer SP, Maciosek MV, Flottemesch TJ, et al. Aspirin for the primary prevention of cardiovascular disease and colorectal cancer: a decision analysis for the U.S. Preventive Services Task Force. Ann Intern Med. 2016;164:777-786.
6. Bibbins-Domingo K. Aspirin use for the primary prevention of cardiovascular disease and colorectal cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann Intern Med. 2016;164:836-845.
7. Baigent C, Blackwell L, Colins R, et al; Antithrombotic Trialists (ATT) Collaboration. Aspirin in the primary and secondary prevention of vascular disease: collaborative meta-analysis of individual participation data from randomised trials. Lancet. 2009:373:1849-1860.
8. Ikeda Y, Shimada K, Teramoto T, et al. Low-dose aspirin for primary prevention of cardiovascular events in Japanese patients 60 years or older with atherosclerotic risk factors: a randomized clinical trial. JAMA. 2014;312:2510-2520.
9. Rothwell PM, Cook NR, Gaziano JM, et al. Effects of aspirin on risks of vascular events and cancer according to bodyweight and dose: analysis of individual patient data from randomised trials. Lancet. 2018;392:387-399.
10. Bowman L, Mafham M, Wallendszus K, et al; ASCEND Study Collaborative Group. Effects of aspirin for primary prevention in persons with diabetes mellitus. N Engl J Med. 2018;379:1529-1539.
11. McNeil JJ, Wolfe R, Woods RL, et al. Effect of aspirin on cardiovascular events and bleeding in the healthy elderly. N Engl J Med. 2018;379:1509-1518.
12. Gaziano JM, Brotons C, Coppolecchia R, et al. Use of aspirin to reduce risk of initial vascular events in patients at moderate risk of cardiovascular disease (ARRIVE): a randomised, double-blind, placebo-controlled trial. Lancet. 2018;392:1036-1046.
13. Kune GA, Kune S, Watson LF. Colorectal cancer risk, chronic illness, operations, and medications: case control results from Melbourne Colorectal Cancer Study. Cancer Res. 1988;48:4399-4404.
14. Sutcliffe P, Connock M, Gurung T, et al. Aspirin for prophylactic use in the primary prevention of cardiovascular disease and cancer: a systematic review and overview of reviews. Health Technol Assess. 2013;17:1-253.
15. Burn J, Gerdes AM, Macrae F, et al. Long-term effect of aspirin on cancer risk in carriers of hereditary colorectal cancer: an analysis from the CAPP2 randomised controlled trial. Lancet. 2011;378:2081-2087.
16. Gamba CA, Swetter SM, Stefanick ML, et al. Aspirin is associated with lower melanoma risk among postmenopausal Caucasian women: the Women’s Health Initiative. Cancer. 2013;119:1562-1569.
17. Trabert B, Ness RB, Lo-Ciganic WH, et al. Aspirin, nonaspirin nonsteroidal anti-inflammatory drug, and acetaminophen use and risk of invasive epithelial ovarian cancer: a pooled analysis in the Ovarian Cancer Association Consortium. J Natl Cancer Inst. 2014;106:djt431.
18. Risch H, Lu L, Streicher SA, et al. Aspirin use and reduced risk of pancreatic cancer. Cancer Epidemiol Biomarkers Prev. 2016;26:68-74.
19. McNeil JJ, Nelson MR, Woods RL, et al. Effect of aspirin on all-cause mortality in the healthy elderly. N Engl J Med. 2018;379:1519-1528.
20. Hernández-Díaz S, Rodríguez LA. Incidence of serious upper gastrointestinal bleeding/perforation in the general population: review of epidemiologic studies. J Clin Epidemiol. 2002;55:157-163.
21. Guirguis-Blake JM, Evans CV, Senger CA, et al. Aspirin for the primary prevention of cardiovascular events: a systematic evidence review for the U.S. Preventive Services Task Force. Evidence Synthesis no 131. Rockville, MD: Agency for Healthcare Research and Quality; 2015. https://www.ncbi.nlm.nih.gov/books/NBK321623/. Accessed March 22, 2019.
22. De Berardis G, Lucisano G, D’Ettorre A, et al. Association of aspirin use with major bleeding in patients with and without diabetes. JAMA. 2012;307:2286-2294.
23. Thorat MA, Cuzick J. Prophylactic use of aspirin: systematic review of harms and approaches to mitigation in the general population. Eur J Epidemiol. 2015;30:5-18.
24. Hernández-Díaz S, García Rodríguez LA. Cardioprotective aspirin users and their excess risk of upper gastrointestinal complications. BMC Med. 2006;4:22.
25. Huang ES, Strate LL, Ho WW, et al. Long term use of aspirin and the risk of gastrointestinal bleeding. Am J Med. 2011:124;426-433.
26. Walker J, Robinson J, Stewart J, et al. Does enteric-coated aspirin result in a lower incidence of gastrointestinal complications compared to normal aspirin? Interact Cardiovasc Thorac Surg. 2007:6;519-522.
27. NIH. Aspirin dosing: a patient-centric trial assessing benefits and long-term effectiveness (ADAPTABLE). https://clinicaltrials.gov/ct2/show/NCT02697916. Accessed March 22, 2019.
28. Piepoli MF, Hoes AW, Agewall S, et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice: the Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice. Eur Heart J. 2016;37:2315-2381.
29. ADA. Standards of medical care in diabetes – 2017. Diabetes Care. 2017;40(suppl 1). http://care.diabetesjournals.org/content/diacare/suppl/2016/12/15/40.Supplement_1.DC1/DC_40_S1_final.pdf. Accessed March 22, 2019.
30. Vandvik PO, Lincoff AM, Gore JM, et al. Primary and secondary prevention of cardiovascular disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(suppl):e637S-e668S.
31. Arnett DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA Guideline on the Primary Prevention of Cardiovascular Disease. J Am Col Cardiol. 2019. doi: https://doi.org/10.1016/j.jacc.2019.03.010. Accessed March 22, 2019.
32. Mora S, Manson JE. Aspirin for primary prevention of atherosclerotic cardiovascular disease: advances in diagnosis and treatment. JAMA Intern Med. 2016;176:1195-1204.
PRACTICE RECOMMENDATIONS
› Consider aspirin for patients 50 to 59 years of age who have a 10-year cardiovascular disease (CVD) risk of ≥ 10% and low bleeding risk. C
› Discuss prophylactic aspirin (using a shared decision-making model) with patients 60 to 69 years of age who have a 10-year CVD risk of ≥ 10% and low bleeding risk. C
› Avoid using aspirin for primary prevention in patients ≥ 70 years of age. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Anterior, apical, posterior: Vaginal anatomy for the gynecologic surgeon
CASE 1 Defining anatomic structures to assure surgical precision
A 44-year-old woman is scheduled for a vaginal hysterectomy and bilateral salpingectomy for abnormal uterine bleeding. In your academic practice, a resident routinely operates with you and is accompanied by a medical student. As this is your first case with each learner, you review the steps of the procedure along with pertinent anatomy. During this discussion, numerous anatomic terms are used to describe anterior cul-de-sac entry, including pubocervical fascia, vesicouterine fold, and vesicovaginal space. Which of these terms, if any, are correct? Is there a preferred term that should be used to teach future learners so we can all “speak” the same language?
What’s in a name?
ObGyns must thoroughly understand pelvic anatomy, since much of our patient care relates to structures in that region. We also must understand the terminology that most appropriately describes each pelvic structure so that we can communicate effectively with colleagues and other providers. The case described above lists several terms that are commonly found in gynecologic textbooks and surgical atlases to describe dissection for vaginal hysterectomy. Lack of a standardized vocabulary, however, often confuses teachers and learners alike, and it highlights the importance of having a universal language to ensure the safe, effective performance of surgical procedures.1
At first glance, it may seem that anatomic terms are inherently descriptive of the structure they represent; for example, the terms uterus and vagina seem rather obvious. However, many anatomic terms convey ambiguity. Which muscles, for example, constitute the levator ani: pubococcygeus, pubovisceral, pubovisceralis, puboperinealis, puboanalis, pubovaginalis, puborectalis, puborectal, iliococcygeus, ischiococcygeus? Do any of these terms redundantly describe the same structure, or does each term refer to an independent structure?
Standard terminology is essential
Anatomists long have recognized the need for standardized terminology to facilitate clear communication. To provide historical background, the term anatomy is derived from the Greek word for “dissection” or “to cut open.”2 Records on the scientific study of human anatomy date back thousands of years.
A brief review of current standardized terminology can be traced back to 1895, with the publication of Basle Terminologia Anatomica.3 That work was intended to provide a consolidated reference with clear direction regarding which anatomic terms should be used. It was updated several times during the ensuing century and was later published as Nomina Anatomica.
In 1990, an international committee was formed with representatives from many anatomical organizations, again with the intention of providing standardized anatomic terminology. Those efforts resulted in the publication of Terminologia Anatomica: International Anatomical Terminology, commonly referred to as TA, in 1998. TA continues to be the referent standard for human anatomic terminology; it was most recently updated in 2011.4
CASE 2 Conveying details of mesh erosion
A 52-year-old woman presents to the general gynecology clinic with a 10-year history of pelvic pain and dyspareunia after undergoing vaginal mesh surgery for prolapse and urinary incontinence. On examination, there is a visible ridge of mesh extending from the left side of the midurethra along the anterior and lateral vagina for a length of 1.5 cm. There also is a palpable tight band on the right vaginal wall near the ischial spine that reproduces her pain and causes spasm of the levator ani. You believe the patient needs a urogynecology referral for complications of vaginal mesh. How do you best describe your findings to your urogynecology colleague?
Continue to: Pelvic anatomy from the SGS perspective...
Pelvic anatomy from the SGS perspective
The Society of Gynecologic Surgeons (SGS) recognized the importance of standardizing terminology specific to the pelvis. The SGS Pelvic Anatomy Group thus was organized in 2016. The Pelvic Anatomy Group’s purpose is to help educate physicians about pelvic anatomy, with the overarching goal of compiling instructional materials, primarily from dissections (surgical or cadaveric), and radiologic imaging for all pelvic structures. Throughout the discussions on this initiative, it became clear that standardized terms needed to be established and used for pelvic structures.
While TA is an excellent reference work, it does not include all of the clinically relevant structures for gynecologic surgeons. As physicians, surgeons, and women’s health care providers, we read about and discuss pelvic anatomy structures in medical textbooks, medical literature, and clinical settings that are not necessarily included in TA. In addition, advances in information technology have facilitated the creation of clinically oriented computer-based anatomy programs and expanded the number and availability of electronic publications on surgical and clinical anatomy.5 As a result, there is a need not only to standardize nomenclature but also to continually revise and update terminology and integrate new terms, both from an anatomic and a clinical perspective.
The Pelvic Anatomy Group developed a novel approach to anatomic terminology. We decided to review the medical literature, identify the terms used, adjudicate the terms with current TA terms, and provide consensus for the terms and structures in the pelvis. Because of the volume of literature available and the existing number of terms, we divided the pelvis into 4 regions—anterior, apical, posterior, and vulvar—to improve the feasibility of reviewing the medical literature for the entire female pelvis.
Our process for tackling terminology
Our literature review started with the anterior compartment. (For complete details, see our prior publication.3) Modeled on a systematic review, we searched the MEDLINE database for terms related to the anterior pelvis, screened all associated abstracts, and then extracted terms from appropriate papers. We also identified several book chapters from various disciplines (anatomy, gynecology, urology, and radiology) to ensure wide representation of disciplines. We then extracted all terms pertinent to the anterior pelvis.
We organized the terms, with terms that referred to the same anatomic structure grouped together. Whenever possible, we used TA terms as the preferred terms. In this process, however, we identified several clinically relevant terms that were not included in TA: pelvic sidewall, pelvic bones, anterior compartment, pubourethral ligament, vaginal sulcus, and levator hiatus, among others. The new terms were then proposed and agreed on by members of the SGS Pelvic Anatomy Group and accepted by SGS members. We currently are completing a similar process for the apical pelvis, posterior pelvis, and vulvar regions.
TA code numbers pinpoint the nomenclature
As we move forward, we suggest that physicians use TA or other approved terms for patient and research communication. Such use will help standardize anatomic terms and also will improve communication between providers and education for learners.
Continue to: TA includes approved options...
TA includes approved options in English and Latin and lists a unique identification number for each term (shown in parentheses in the examples that follow). For instance, to answer the question posed earlier, the levator ani (A04.5.04.002) is comprised of the pubococcygeus (A04.5.04.003), puborectalis (A04.5.04.007), and iliococcygeus (A04.5.04.008) muscles (FIGURE 1).The terms pubovisceral and pubovisceralis are used synonymously in the literature with pubococcygeus (A04.5.04.003).3 The additional terms puboperinealis (A04.5.04.004), pubovaginalis (A04.5.04.005), and puboanalis (A04.5.04.006) are subcomponents of the pubococcygeus (A04.5.04.003), and this relationship is indicated in TA by indentation formatting.4 Finally, the ischiococcygeus (A04.5.04.011) muscle is not considered part of the levator ani (A04.5.04.002).
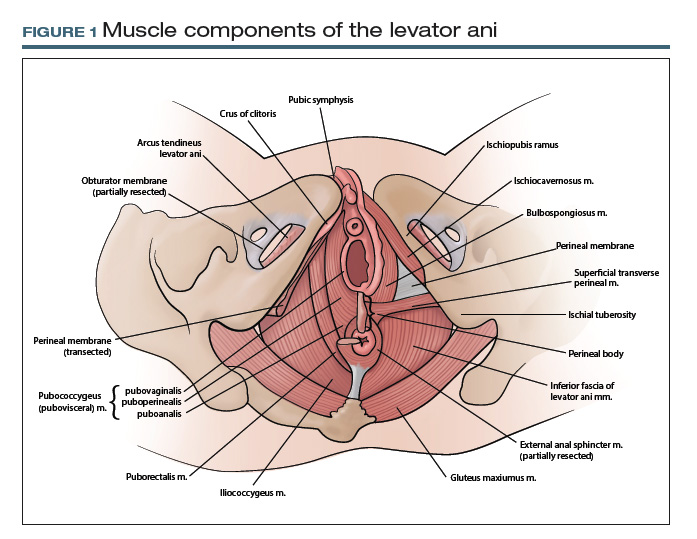
Revisiting the mesh erosion case: Reporting your findings
After reviewing the recommended terminology for the anterior pelvis,3,4 you might draft a report as follows: “A mesh erosion was visualized in anterior vaginal wall (A09.1.04.006) at the level of the mid-urethra extending into ‘anterior and lateral vaginal sulci’ (proposed term). In addition, there is a painful tight band in the ‘lateral vaginal wall’ (proposed term) near the ischial spine (A02.5.01.205). Palpation of this band reproduces the patient’s pain and causes secondary spasm of the levator ani (A04.5.04.002).” Certainly, TA identification numbers would not be expected to be included in medical communication; they are included here for reference.
From your description, your urogynecology colleague has a better understanding of the location of your patient’s vaginal mesh and requests her operative report from an outside facility. In the operative report, the surgeon described “placement of mesh into the vagina, dissection through the rectal spaces, and anchoring of the mesh into the levator/pelvic muscles, the cervix, and lastly to the paraurethral ligaments,” and “passage of trocars through the cave of Retzius at the level of the midurethra” (FIGURE 2).
Based on this description, the urogynecologist ascertains that the mesh is located in the anterior vaginal wall (A09.1.04.006), with passage of anchoring arms through the bilateral sacrospinous ligaments (A03.6.03.007) and retropubic space (A10.1.01.003). Exposed mesh is visible, extending from the midurethra to the “anterior and lateral vaginal sulci” (proposed term).
This case clearly demonstrates the importance of communication between providers for patient care, since understanding the patient’s anatomy and the location of the vaginal mesh is important for planning surgical excision of the exposed mesh.
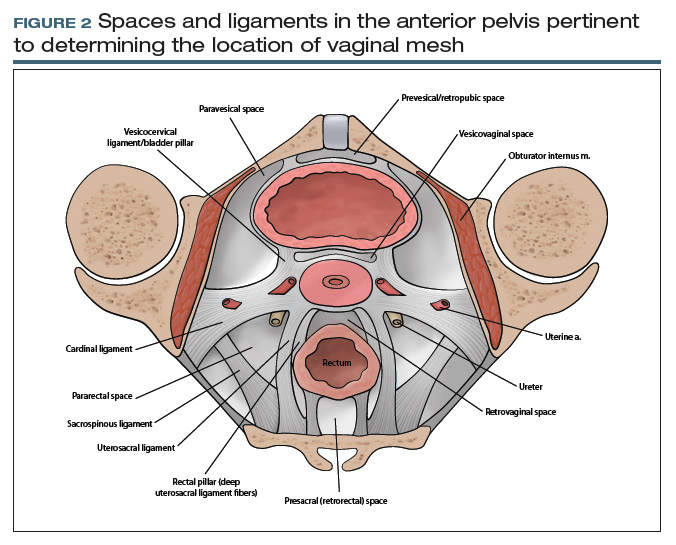
Additional initiatives
Outlining standardized terminology is just the first step toward improving the anatomic “language” used among providers. Ongoing efforts from the SGS Pelvic Anatomy Group include a special imaging group’s review of imaging modalities (ultrasonography, magnetic resonance imaging, computerized tomography) to improve standardization on reporting clinical anatomy. In addition, SGS has developed a group to create educational content related to the structures identified by the terminology group from cadaveric or surgical dissections. Educational materials will be compiled to help physicians and learners expand their anatomic understanding and improve their communication.
Further details of the Pelvic Anatomy Group’s efforts can be found on the SGS website at https://www.sgsonline.org.
- American Association of Clinical Anatomists, Educational Affairs Committee. The clinical anatomy of several invasive procedures. Clin Anat. 1999;12:43-54.
- Venes D, ed. Taber's Cyclopedic Medical Dictionary. 23rd ed. Philadelphia, PA: FA Davis Company; 2017.
- Jeppson PC, Balgobin S, Washington BB, et al; for the Society of Gynecologic Surgeons Pelvic Anatomy Group. Recommended standardized terminology of the anterior female pelvis based on a structured medical literature review. Am J Obstet Gynecol. 2018;219:26-39.
- Federative Committee on Anatomical Terminologies (FCAT). Terminologia Anatomica: International Anatomical Terminology. 2nd ed. Stuttgart, Germany: Thieme; 2011.
- Rosse C. Terminologia Anatomica: considered from the perspective of next-generation knowledge sources. Clin Anat. 2001;14:120-133.
CASE 1 Defining anatomic structures to assure surgical precision
A 44-year-old woman is scheduled for a vaginal hysterectomy and bilateral salpingectomy for abnormal uterine bleeding. In your academic practice, a resident routinely operates with you and is accompanied by a medical student. As this is your first case with each learner, you review the steps of the procedure along with pertinent anatomy. During this discussion, numerous anatomic terms are used to describe anterior cul-de-sac entry, including pubocervical fascia, vesicouterine fold, and vesicovaginal space. Which of these terms, if any, are correct? Is there a preferred term that should be used to teach future learners so we can all “speak” the same language?
What’s in a name?
ObGyns must thoroughly understand pelvic anatomy, since much of our patient care relates to structures in that region. We also must understand the terminology that most appropriately describes each pelvic structure so that we can communicate effectively with colleagues and other providers. The case described above lists several terms that are commonly found in gynecologic textbooks and surgical atlases to describe dissection for vaginal hysterectomy. Lack of a standardized vocabulary, however, often confuses teachers and learners alike, and it highlights the importance of having a universal language to ensure the safe, effective performance of surgical procedures.1
At first glance, it may seem that anatomic terms are inherently descriptive of the structure they represent; for example, the terms uterus and vagina seem rather obvious. However, many anatomic terms convey ambiguity. Which muscles, for example, constitute the levator ani: pubococcygeus, pubovisceral, pubovisceralis, puboperinealis, puboanalis, pubovaginalis, puborectalis, puborectal, iliococcygeus, ischiococcygeus? Do any of these terms redundantly describe the same structure, or does each term refer to an independent structure?
Standard terminology is essential
Anatomists long have recognized the need for standardized terminology to facilitate clear communication. To provide historical background, the term anatomy is derived from the Greek word for “dissection” or “to cut open.”2 Records on the scientific study of human anatomy date back thousands of years.
A brief review of current standardized terminology can be traced back to 1895, with the publication of Basle Terminologia Anatomica.3 That work was intended to provide a consolidated reference with clear direction regarding which anatomic terms should be used. It was updated several times during the ensuing century and was later published as Nomina Anatomica.
In 1990, an international committee was formed with representatives from many anatomical organizations, again with the intention of providing standardized anatomic terminology. Those efforts resulted in the publication of Terminologia Anatomica: International Anatomical Terminology, commonly referred to as TA, in 1998. TA continues to be the referent standard for human anatomic terminology; it was most recently updated in 2011.4
CASE 2 Conveying details of mesh erosion
A 52-year-old woman presents to the general gynecology clinic with a 10-year history of pelvic pain and dyspareunia after undergoing vaginal mesh surgery for prolapse and urinary incontinence. On examination, there is a visible ridge of mesh extending from the left side of the midurethra along the anterior and lateral vagina for a length of 1.5 cm. There also is a palpable tight band on the right vaginal wall near the ischial spine that reproduces her pain and causes spasm of the levator ani. You believe the patient needs a urogynecology referral for complications of vaginal mesh. How do you best describe your findings to your urogynecology colleague?
Continue to: Pelvic anatomy from the SGS perspective...
Pelvic anatomy from the SGS perspective
The Society of Gynecologic Surgeons (SGS) recognized the importance of standardizing terminology specific to the pelvis. The SGS Pelvic Anatomy Group thus was organized in 2016. The Pelvic Anatomy Group’s purpose is to help educate physicians about pelvic anatomy, with the overarching goal of compiling instructional materials, primarily from dissections (surgical or cadaveric), and radiologic imaging for all pelvic structures. Throughout the discussions on this initiative, it became clear that standardized terms needed to be established and used for pelvic structures.
While TA is an excellent reference work, it does not include all of the clinically relevant structures for gynecologic surgeons. As physicians, surgeons, and women’s health care providers, we read about and discuss pelvic anatomy structures in medical textbooks, medical literature, and clinical settings that are not necessarily included in TA. In addition, advances in information technology have facilitated the creation of clinically oriented computer-based anatomy programs and expanded the number and availability of electronic publications on surgical and clinical anatomy.5 As a result, there is a need not only to standardize nomenclature but also to continually revise and update terminology and integrate new terms, both from an anatomic and a clinical perspective.
The Pelvic Anatomy Group developed a novel approach to anatomic terminology. We decided to review the medical literature, identify the terms used, adjudicate the terms with current TA terms, and provide consensus for the terms and structures in the pelvis. Because of the volume of literature available and the existing number of terms, we divided the pelvis into 4 regions—anterior, apical, posterior, and vulvar—to improve the feasibility of reviewing the medical literature for the entire female pelvis.
Our process for tackling terminology
Our literature review started with the anterior compartment. (For complete details, see our prior publication.3) Modeled on a systematic review, we searched the MEDLINE database for terms related to the anterior pelvis, screened all associated abstracts, and then extracted terms from appropriate papers. We also identified several book chapters from various disciplines (anatomy, gynecology, urology, and radiology) to ensure wide representation of disciplines. We then extracted all terms pertinent to the anterior pelvis.
We organized the terms, with terms that referred to the same anatomic structure grouped together. Whenever possible, we used TA terms as the preferred terms. In this process, however, we identified several clinically relevant terms that were not included in TA: pelvic sidewall, pelvic bones, anterior compartment, pubourethral ligament, vaginal sulcus, and levator hiatus, among others. The new terms were then proposed and agreed on by members of the SGS Pelvic Anatomy Group and accepted by SGS members. We currently are completing a similar process for the apical pelvis, posterior pelvis, and vulvar regions.
TA code numbers pinpoint the nomenclature
As we move forward, we suggest that physicians use TA or other approved terms for patient and research communication. Such use will help standardize anatomic terms and also will improve communication between providers and education for learners.
Continue to: TA includes approved options...
TA includes approved options in English and Latin and lists a unique identification number for each term (shown in parentheses in the examples that follow). For instance, to answer the question posed earlier, the levator ani (A04.5.04.002) is comprised of the pubococcygeus (A04.5.04.003), puborectalis (A04.5.04.007), and iliococcygeus (A04.5.04.008) muscles (FIGURE 1).The terms pubovisceral and pubovisceralis are used synonymously in the literature with pubococcygeus (A04.5.04.003).3 The additional terms puboperinealis (A04.5.04.004), pubovaginalis (A04.5.04.005), and puboanalis (A04.5.04.006) are subcomponents of the pubococcygeus (A04.5.04.003), and this relationship is indicated in TA by indentation formatting.4 Finally, the ischiococcygeus (A04.5.04.011) muscle is not considered part of the levator ani (A04.5.04.002).

Revisiting the mesh erosion case: Reporting your findings
After reviewing the recommended terminology for the anterior pelvis,3,4 you might draft a report as follows: “A mesh erosion was visualized in anterior vaginal wall (A09.1.04.006) at the level of the mid-urethra extending into ‘anterior and lateral vaginal sulci’ (proposed term). In addition, there is a painful tight band in the ‘lateral vaginal wall’ (proposed term) near the ischial spine (A02.5.01.205). Palpation of this band reproduces the patient’s pain and causes secondary spasm of the levator ani (A04.5.04.002).” Certainly, TA identification numbers would not be expected to be included in medical communication; they are included here for reference.
From your description, your urogynecology colleague has a better understanding of the location of your patient’s vaginal mesh and requests her operative report from an outside facility. In the operative report, the surgeon described “placement of mesh into the vagina, dissection through the rectal spaces, and anchoring of the mesh into the levator/pelvic muscles, the cervix, and lastly to the paraurethral ligaments,” and “passage of trocars through the cave of Retzius at the level of the midurethra” (FIGURE 2).
Based on this description, the urogynecologist ascertains that the mesh is located in the anterior vaginal wall (A09.1.04.006), with passage of anchoring arms through the bilateral sacrospinous ligaments (A03.6.03.007) and retropubic space (A10.1.01.003). Exposed mesh is visible, extending from the midurethra to the “anterior and lateral vaginal sulci” (proposed term).
This case clearly demonstrates the importance of communication between providers for patient care, since understanding the patient’s anatomy and the location of the vaginal mesh is important for planning surgical excision of the exposed mesh.

Additional initiatives
Outlining standardized terminology is just the first step toward improving the anatomic “language” used among providers. Ongoing efforts from the SGS Pelvic Anatomy Group include a special imaging group’s review of imaging modalities (ultrasonography, magnetic resonance imaging, computerized tomography) to improve standardization on reporting clinical anatomy. In addition, SGS has developed a group to create educational content related to the structures identified by the terminology group from cadaveric or surgical dissections. Educational materials will be compiled to help physicians and learners expand their anatomic understanding and improve their communication.
Further details of the Pelvic Anatomy Group’s efforts can be found on the SGS website at https://www.sgsonline.org.
CASE 1 Defining anatomic structures to assure surgical precision
A 44-year-old woman is scheduled for a vaginal hysterectomy and bilateral salpingectomy for abnormal uterine bleeding. In your academic practice, a resident routinely operates with you and is accompanied by a medical student. As this is your first case with each learner, you review the steps of the procedure along with pertinent anatomy. During this discussion, numerous anatomic terms are used to describe anterior cul-de-sac entry, including pubocervical fascia, vesicouterine fold, and vesicovaginal space. Which of these terms, if any, are correct? Is there a preferred term that should be used to teach future learners so we can all “speak” the same language?
What’s in a name?
ObGyns must thoroughly understand pelvic anatomy, since much of our patient care relates to structures in that region. We also must understand the terminology that most appropriately describes each pelvic structure so that we can communicate effectively with colleagues and other providers. The case described above lists several terms that are commonly found in gynecologic textbooks and surgical atlases to describe dissection for vaginal hysterectomy. Lack of a standardized vocabulary, however, often confuses teachers and learners alike, and it highlights the importance of having a universal language to ensure the safe, effective performance of surgical procedures.1
At first glance, it may seem that anatomic terms are inherently descriptive of the structure they represent; for example, the terms uterus and vagina seem rather obvious. However, many anatomic terms convey ambiguity. Which muscles, for example, constitute the levator ani: pubococcygeus, pubovisceral, pubovisceralis, puboperinealis, puboanalis, pubovaginalis, puborectalis, puborectal, iliococcygeus, ischiococcygeus? Do any of these terms redundantly describe the same structure, or does each term refer to an independent structure?
Standard terminology is essential
Anatomists long have recognized the need for standardized terminology to facilitate clear communication. To provide historical background, the term anatomy is derived from the Greek word for “dissection” or “to cut open.”2 Records on the scientific study of human anatomy date back thousands of years.
A brief review of current standardized terminology can be traced back to 1895, with the publication of Basle Terminologia Anatomica.3 That work was intended to provide a consolidated reference with clear direction regarding which anatomic terms should be used. It was updated several times during the ensuing century and was later published as Nomina Anatomica.
In 1990, an international committee was formed with representatives from many anatomical organizations, again with the intention of providing standardized anatomic terminology. Those efforts resulted in the publication of Terminologia Anatomica: International Anatomical Terminology, commonly referred to as TA, in 1998. TA continues to be the referent standard for human anatomic terminology; it was most recently updated in 2011.4
CASE 2 Conveying details of mesh erosion
A 52-year-old woman presents to the general gynecology clinic with a 10-year history of pelvic pain and dyspareunia after undergoing vaginal mesh surgery for prolapse and urinary incontinence. On examination, there is a visible ridge of mesh extending from the left side of the midurethra along the anterior and lateral vagina for a length of 1.5 cm. There also is a palpable tight band on the right vaginal wall near the ischial spine that reproduces her pain and causes spasm of the levator ani. You believe the patient needs a urogynecology referral for complications of vaginal mesh. How do you best describe your findings to your urogynecology colleague?
Continue to: Pelvic anatomy from the SGS perspective...
Pelvic anatomy from the SGS perspective
The Society of Gynecologic Surgeons (SGS) recognized the importance of standardizing terminology specific to the pelvis. The SGS Pelvic Anatomy Group thus was organized in 2016. The Pelvic Anatomy Group’s purpose is to help educate physicians about pelvic anatomy, with the overarching goal of compiling instructional materials, primarily from dissections (surgical or cadaveric), and radiologic imaging for all pelvic structures. Throughout the discussions on this initiative, it became clear that standardized terms needed to be established and used for pelvic structures.
While TA is an excellent reference work, it does not include all of the clinically relevant structures for gynecologic surgeons. As physicians, surgeons, and women’s health care providers, we read about and discuss pelvic anatomy structures in medical textbooks, medical literature, and clinical settings that are not necessarily included in TA. In addition, advances in information technology have facilitated the creation of clinically oriented computer-based anatomy programs and expanded the number and availability of electronic publications on surgical and clinical anatomy.5 As a result, there is a need not only to standardize nomenclature but also to continually revise and update terminology and integrate new terms, both from an anatomic and a clinical perspective.
The Pelvic Anatomy Group developed a novel approach to anatomic terminology. We decided to review the medical literature, identify the terms used, adjudicate the terms with current TA terms, and provide consensus for the terms and structures in the pelvis. Because of the volume of literature available and the existing number of terms, we divided the pelvis into 4 regions—anterior, apical, posterior, and vulvar—to improve the feasibility of reviewing the medical literature for the entire female pelvis.
Our process for tackling terminology
Our literature review started with the anterior compartment. (For complete details, see our prior publication.3) Modeled on a systematic review, we searched the MEDLINE database for terms related to the anterior pelvis, screened all associated abstracts, and then extracted terms from appropriate papers. We also identified several book chapters from various disciplines (anatomy, gynecology, urology, and radiology) to ensure wide representation of disciplines. We then extracted all terms pertinent to the anterior pelvis.
We organized the terms, with terms that referred to the same anatomic structure grouped together. Whenever possible, we used TA terms as the preferred terms. In this process, however, we identified several clinically relevant terms that were not included in TA: pelvic sidewall, pelvic bones, anterior compartment, pubourethral ligament, vaginal sulcus, and levator hiatus, among others. The new terms were then proposed and agreed on by members of the SGS Pelvic Anatomy Group and accepted by SGS members. We currently are completing a similar process for the apical pelvis, posterior pelvis, and vulvar regions.
TA code numbers pinpoint the nomenclature
As we move forward, we suggest that physicians use TA or other approved terms for patient and research communication. Such use will help standardize anatomic terms and also will improve communication between providers and education for learners.
Continue to: TA includes approved options...
TA includes approved options in English and Latin and lists a unique identification number for each term (shown in parentheses in the examples that follow). For instance, to answer the question posed earlier, the levator ani (A04.5.04.002) is comprised of the pubococcygeus (A04.5.04.003), puborectalis (A04.5.04.007), and iliococcygeus (A04.5.04.008) muscles (FIGURE 1).The terms pubovisceral and pubovisceralis are used synonymously in the literature with pubococcygeus (A04.5.04.003).3 The additional terms puboperinealis (A04.5.04.004), pubovaginalis (A04.5.04.005), and puboanalis (A04.5.04.006) are subcomponents of the pubococcygeus (A04.5.04.003), and this relationship is indicated in TA by indentation formatting.4 Finally, the ischiococcygeus (A04.5.04.011) muscle is not considered part of the levator ani (A04.5.04.002).

Revisiting the mesh erosion case: Reporting your findings
After reviewing the recommended terminology for the anterior pelvis,3,4 you might draft a report as follows: “A mesh erosion was visualized in anterior vaginal wall (A09.1.04.006) at the level of the mid-urethra extending into ‘anterior and lateral vaginal sulci’ (proposed term). In addition, there is a painful tight band in the ‘lateral vaginal wall’ (proposed term) near the ischial spine (A02.5.01.205). Palpation of this band reproduces the patient’s pain and causes secondary spasm of the levator ani (A04.5.04.002).” Certainly, TA identification numbers would not be expected to be included in medical communication; they are included here for reference.
From your description, your urogynecology colleague has a better understanding of the location of your patient’s vaginal mesh and requests her operative report from an outside facility. In the operative report, the surgeon described “placement of mesh into the vagina, dissection through the rectal spaces, and anchoring of the mesh into the levator/pelvic muscles, the cervix, and lastly to the paraurethral ligaments,” and “passage of trocars through the cave of Retzius at the level of the midurethra” (FIGURE 2).
Based on this description, the urogynecologist ascertains that the mesh is located in the anterior vaginal wall (A09.1.04.006), with passage of anchoring arms through the bilateral sacrospinous ligaments (A03.6.03.007) and retropubic space (A10.1.01.003). Exposed mesh is visible, extending from the midurethra to the “anterior and lateral vaginal sulci” (proposed term).
This case clearly demonstrates the importance of communication between providers for patient care, since understanding the patient’s anatomy and the location of the vaginal mesh is important for planning surgical excision of the exposed mesh.

Additional initiatives
Outlining standardized terminology is just the first step toward improving the anatomic “language” used among providers. Ongoing efforts from the SGS Pelvic Anatomy Group include a special imaging group’s review of imaging modalities (ultrasonography, magnetic resonance imaging, computerized tomography) to improve standardization on reporting clinical anatomy. In addition, SGS has developed a group to create educational content related to the structures identified by the terminology group from cadaveric or surgical dissections. Educational materials will be compiled to help physicians and learners expand their anatomic understanding and improve their communication.
Further details of the Pelvic Anatomy Group’s efforts can be found on the SGS website at https://www.sgsonline.org.
- American Association of Clinical Anatomists, Educational Affairs Committee. The clinical anatomy of several invasive procedures. Clin Anat. 1999;12:43-54.
- Venes D, ed. Taber's Cyclopedic Medical Dictionary. 23rd ed. Philadelphia, PA: FA Davis Company; 2017.
- Jeppson PC, Balgobin S, Washington BB, et al; for the Society of Gynecologic Surgeons Pelvic Anatomy Group. Recommended standardized terminology of the anterior female pelvis based on a structured medical literature review. Am J Obstet Gynecol. 2018;219:26-39.
- Federative Committee on Anatomical Terminologies (FCAT). Terminologia Anatomica: International Anatomical Terminology. 2nd ed. Stuttgart, Germany: Thieme; 2011.
- Rosse C. Terminologia Anatomica: considered from the perspective of next-generation knowledge sources. Clin Anat. 2001;14:120-133.
- American Association of Clinical Anatomists, Educational Affairs Committee. The clinical anatomy of several invasive procedures. Clin Anat. 1999;12:43-54.
- Venes D, ed. Taber's Cyclopedic Medical Dictionary. 23rd ed. Philadelphia, PA: FA Davis Company; 2017.
- Jeppson PC, Balgobin S, Washington BB, et al; for the Society of Gynecologic Surgeons Pelvic Anatomy Group. Recommended standardized terminology of the anterior female pelvis based on a structured medical literature review. Am J Obstet Gynecol. 2018;219:26-39.
- Federative Committee on Anatomical Terminologies (FCAT). Terminologia Anatomica: International Anatomical Terminology. 2nd ed. Stuttgart, Germany: Thieme; 2011.
- Rosse C. Terminologia Anatomica: considered from the perspective of next-generation knowledge sources. Clin Anat. 2001;14:120-133.
Beyond enhanced recovery after surgery
Our specialty is focusing now more intently on perioperative optimization, commonly referred to as enhanced recovery after surgery (ERAS), a concept championed first and most visibly by colorectal surgeons in the 1990s.1 Both academic and nonacademic practices are challenging long-held beliefs about perioperative management.
The 3 tenets of ERAS
In multiple surgical specialties, proper implementation of 3 tenets—early feeding, perioperative euvolemia, and multimodal pain control—reduces the length of hospital stay, improves patient satisfaction, reduces complications, lowers health care costs, and most importantly hastens patient recovery.
1 Early feeding
Just as athletes hydrate and carbohydrate load prior to a competition, patients benefit if fluids and calories are not withheld in anticipation of a physiologically stressful surgical procedure. Similarly, modest benefit is associated with carbohydrate loading as a liquid supplement 2 hours before surgery.2 The American Society of Anesthesiologists guidelines state that while solid foods should not be consumed after midnight before surgery, clear liquids safely may be withheld for only 2 hours prior to anesthesia induction, and systematic reviews have failed to show harm.3,4 All patients, including those undergoing colonic resections, are allowed to eat a general diet as tolerated the evening before surgery, supplemented with caloric-dense nutritional supplements.
2 Multimodal pain control
Postsurgical pain is a top patient concern. Pain control is critical for rapid recovery; it helps avoid upregulation of the sympathetic axis and permits ambulation and resumption of normal activities. Although opioids relieve pain, they should not be considered a primary pain control approach.
Responding to the opioid epidemic, in 2015 the Centers for Disease Control and Prevention identified opioid overdose prevention as one of the top 5 public health challenges; notably, approximately 6% of patients will experience new, persistent opioid use following surgery.5 Optimal pain management therefore should provide effective pain relief while minimizing opioid use.
Preemptive oral acetaminophen, gabapentin, and celecoxib should be used routinely prior to incision; nonsteroidal anti-inflammatory drugs should be scheduled postoperatively. Even after a complex cytoreductive laparotomy, pain may be controlled with oral rather than intravenous (IV) medications in most patients, with opioid requirements averaging just 2 to 4 tablets of oxycodone in the first 48 hours after surgery, in our experience. The most critical need for pain medications occurs in the first 48 hours after surgery, which highlights the importance of local or regional analgesia. In one investigation, implementation of multimodal pain management that included incisional injection of liposomal bupivacaine reduced patient-controlled analgesia use to less than 5% after laparotomy.6 The need for opioids more than a week postoperatively is uncommon even after a laparotomy.
3 Perioperative euvolemia
Maintaining euvolemia is a central and underrecognized tenet of enhanced recovery pathways, and it facilitates the other 2 tenets of early feeding and optimal pain control. Overhydrated patients have more pain and prolonged recovery of bowel function. Unfortunately, euvolemia is the most difficult ERAS component to implement, requiring seamless communication between all members of the surgical team.
Continue to: Fluid therapy...
Fluid therapy should be respected as a pharmacologic agent with both benefits and risks. Recognizing that a single liter of lactated Ringer’s solution contains the sodium load of more than 30 bags of potato chips (and normal saline contains far more), one can imagine the impact of 10 L of solution on peripheral and bowel edema and on overall recovery. Importantly, euvolemia must be initiated during surgery. A meta-analysis of nearly 1,000 randomly assigned patients showed that benefits were limited when euvolemia was initiated in the postoperative period.7
When it comes to maintaining euvolemia, particular care must be taken to avoid erring toward hyperadherence. No difference in hospital length of stay, complications, or ileus was observed when patients were randomly assigned to goal-directed fluid therapy or standard practice.8 However, differences in the volume of fluid administered were relatively small, and while there was evidence of underhydration (likely responsible for acute kidney injury), there was no evidence of overhydration. For example, 4 L of fluid is likely superior to 15 L, but it may not be clinically different from 4.5 L. A threshold of fluid restriction is likely to be reached; that is, additional benefit is not achieved and, instead, detrimental effects may occur.
Rather than a specific directive, a more clinically relevant goal may be to replace insensible fluid losses and to maintain perfusion and blood pressure with the lowest volume possible. Note that estimation of fluid requirements is vastly simplified by omitting mechanical bowel preparation. Postoperatively, permissive oliguria (20 mL/h) is allowed since reduced urine output is a normal response to surgery (as a result of inappropriate secretion of antidiuretic hormone) and does not necessitate administration of a fluid bolus. Above all, anesthesiologists should acknowledge that fluid administration’s effects on a patient extend past the postanesthesia care unit, and the entire surgical team should be invested in the patient’s long-term recovery.
Our experience with ERAS
In 2011, Mayo Clinic was the first institution to implement enhanced recovery on a large scale in gynecologic surgery. We have subsequently made multiple pathway modifications in the spirit of continuous improvement (FIGURE 1).
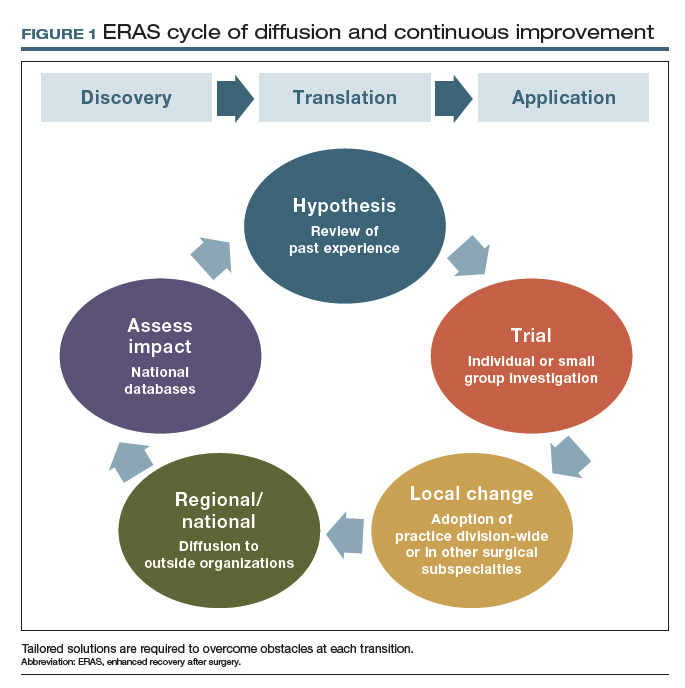
For patients with ovarian cancer requiring extended procedures for cytoreduction via laparotomy (such as colon resection, splenectomy, diaphragm resection), enhanced recovery reduced the median hospital stay by 3 days, patient-controlled IV analgesia use by 88%, and postoperative opioid requirements by 90%.9,10
At 48 hours after surgery, 40% of our patients require no opioids or tramadol, and epidurals are not utilized because of their effects on ambulation and the potential for hypotension. These reductions were met with stable to improved pain scores, a 60% decrease in nausea, and a 50% reduction in adynamic ileus.9,10
Our initial efforts reduced 30-day costs of care by more than $850,000 in just 6 months, with savings of more than $7,600 for each patient undergoing a complex cytoreduction. Furthermore, these improvements allowed consolidation of our inpatient unit with those of other surgical specialties, serving higher volumes of patients within a smaller inpatient footprint. This contraction of inpatient services has accounted for an additional $1.1 million in savings every year since implementation (FIGURE 2).9,10
Our group is not alone in realizing these benefits, and interest has intensified as demonstrated by the fact that the ERAS Society guidelines are among the all-time most downloaded articles in Gynecologic Oncology.11,12 Although our research to demonstrate safety has focused on women undergoing complex oncologic operations, ERAS nevertheless hastens recovery, improves patient satisfaction, and adds value for all patients undergoing gynecologic surgery.
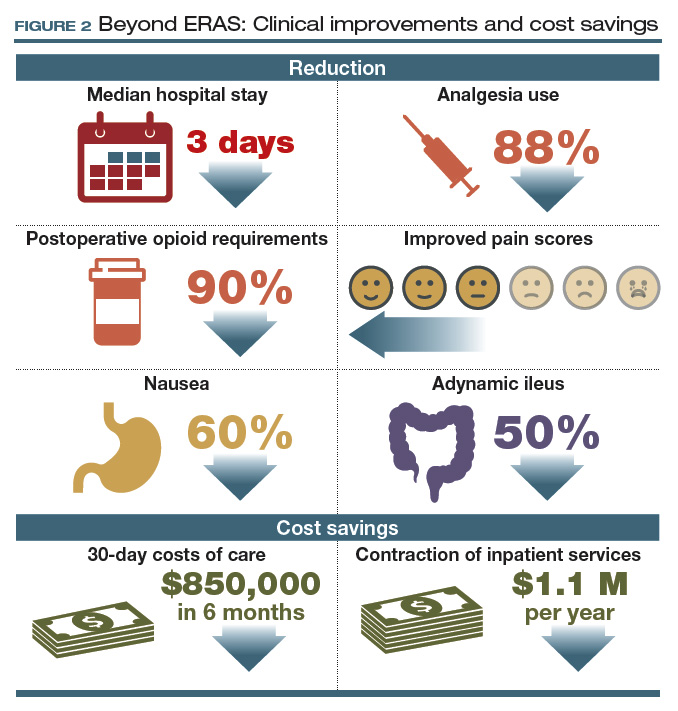
Continue to: Collateral improvements to practice...
Collateral improvements to practice
Clinical optimization using evidence-based practices such as enhanced recovery pathways can result in immediate patient benefit. Affecting such profound clinical improvements is energizing and creates a unique opportunity to transform the culture of the entire health care team. Irrespective of our provider roles (surgeon, anesthesiologist, nurse) or areas of interest (practice, research, education, leadership), we are united by a common purpose: to improve the human condition.13 Reaffirming this common purpose, through the collective effort involved in establishing a standardized enhanced recovery pathway, has allowed our practice and those of others to move beyond enhanced recovery and improve other areas of practice.
Other positive effects. The long-term collateral impact of this culture change at our institution is arguably more important than enhanced recovery itself. Examples of downstream impact include14,15:
- 80% reduction in surgical site infection
- 50% reduction in anastomotic leaks
- 60% reduction in blood utilization for patients undergoing surgery for ovarian cancer.
Team-based pragmatic strategies. Additionally, our willingness to make decisions as a division rather than as individuals facilitated universal implementation of sentinel lymph node biopsy for patients with endometrial cancer and standardized imaging, testing, and surgical decision making for patients with ovarian and endometrial cancer.
The interventions associated with these improvements were not tested in a randomized fashion; however, rather than await perfect data, we made informed decisions based on imperfect data together with a commitment to continuous data review. We find this to be an effective strategy if our goal is to ensure that tomorrow’s outcomes will be better than yesterday’s. In this way, pragmatic trials can be extremely effective in rural settings and tertiary centers.
Barriers to innovation
The widely reported benefits of enhanced recovery beg the question, Why has enhanced recovery not been adopted universally as standard of care? The answer is multifaceted and highlights long-standing shortcomings in our health care system.
Most importantly, our health care system lacks a robust interface to link discovery of new techniques, treatments, and workflows to clinical practice. Perhaps the best example of this is the adoption of minimally invasive surgery (MIS) for endometrial cancer. Ten years have passed since randomized trials showed MIS has equivalent oncologic outcomes and superior recovery compared to laparotomy, yet in the United States less than 50% of women with endometrial cancer benefit.16,17
However, even surgeons who are knowledgeable about recent innovations and genuinely wish to promote improvements may face near-insurmountable skepticism. Blind faith in our abilities and outcomes, overprotection of autonomy, close-mindedness, and satisfaction with the status quo are common responses to innovation and are the enemies of change. Resistance often comes from good intentions, but our desire to avoid complications may result in actions that could just as accurately be labeled superstitious as conservative. These observations suggest that developing methods to incorporate evidence-based practice into routine clinical use is the rate-limiting step in improving surgical quality.
The national Improving Surgical Care and Recovery program is available to specifically aid with ERAS implementation. A collaboration between the Agency for Healthcare Research and Quality (AHRQ) and the American College of Surgeons, the program aims to diffuse enhanced recovery to 750 service lines in 4 surgical subspecialties, including gynecologic surgery, over the next 5 years. (Note: The author is the content expert for the gynecology portion of this program.) The program’s larger aim is to measurably improve patient outcomes, reduce health care utilization, and improve patient experience through the use of an adaptation to AHRQ’s Comprehensive Unit-based Safety Program (CUSP). The backbone for this program is the recent systematic review to establish best practices for gynecologic surgery.1 Free to all participants, the program includes resources such as webinars and coaching calls to assist with the inevitable barriers to ERAS implementation. For more information and to enroll, visit https://www.ahrq.gov/professionals/quality-patient-safety/hais/tools/enhanced-recovery /index.html. An important aspect of the program is a registry for tracking outcomes and identifying areas for improvement. For members who currently participate in the National Surgical Quality Improvement Program, clinical data are automatically uploaded into the database. Programs such as Improving Surgical Care and Recovery may be the most reliable way to facilitate diffusion of best practices and take collective responsibility for not only “my outcomes” but also for “our outcomes” as a national community of gynecologic surgeons.
Reference
1. Kalogera E, Nelson G, Liu J, et al. Surgical technical evidence review for gynecologic surgery conducted for the Agency for Healthcare Research and Quality Safety Program for Improving Surgical Care and Recovery. Am J Obstet Gynecol. 2018;219:563.e1-563.e19.
Principles essential to change
Various methodologies have been described to manage change and facilitate implementation of new workflows and practices. Irrespective of the method used, including the more formal discipline of implementation science, at least 4 principles must be followed:
1. Teamwork. Mutual trust, mutual respect, and a sense of common purpose are minimum requirements for any successful initiative. Standardization is difficult or impossible without these elements. Thus, establishing a healthy team is the first step in implementing change.
2. Stakeholder analysis. Feedback from surgeons, nurses, residents, fellows, anesthesiologists, pharmacists, nurse anesthetists, and administrators is necessary to obtain diverse perspectives, facilitate engagement, and promote collaborative management. Negativity and resistance are common reactions to change, and it is particularly important to include those who are most skeptical in the stakeholder analysis to mitigate sabotage.
3. Concrete metrics. Success is possible only if defined a priori by specific and achievable goals. Counterbalances also are important to ensure that interventions do not have unintended consequences. Once a goal is met (for example, reduced hospital length of stay or costs), relevant metrics should be monitored after project completion for a minimum of 3 years to avoid regression to the pre-project state.
4. Leadership. The project champion responsible for the initiative must objectively facilitate all of the above and ensure excellent communication between stakeholders to nurture long-term engagement. Despite best efforts, if a minority of the group rejects compromise, this creates an opportunity to compare outcomes between those who do and do not accept the proposed change. Progress realized by early adopters may convince resistors to conform at a later time. Alternatively, the project champion also must have the insight to recognize when a proposed change is impossible at that point in time with that particular group. For example, our own initial attempts to implement enhanced recovery stalled in 2008, but they were successful 3 years later in a different environment.
Continue to: Although a discussion of leadership styles...
Although a discussion of leadership styles is beyond the scope of this article, in our experience, the most successful model is one of servant leadership that is team oriented rather than star dominated. Rather than being led by a single surgeon, each of the 4 quality improvement projects reviewed above (ERAS, and reductions in anastomotic leak, surgical site infection, and blood transfusion) that grew from enhanced recovery included trainees and was led by a different champion, encouraging teamwork and promoting career development. Such a model also supports the Accreditation Council for Graduate Medical Education’s emphasis on quality improvement education.
- Nygren J, Thacker J, Carli F, et al; Enhanced Recovery After Surgery (ERAS) Society, for Perioperative Care; European Society for Clinical Nutrition and Metabolism (ESPEN); International Association for Surgical Metabolism and Nutrition (IASMEN). Guidelines for perioperative care in elective rectal/pelvic surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations. World J Surg. 2013;37:285-305.
- American Society of Anesthesiologists Committee. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: an updated report by the American Society of Anesthesiologists Committee on Standards and Practice Parameters. Anesthesiology. 2011;114: 495-511.
- Brady M, Kinn S, Stuart P. Preoperative fasting for adults to prevent perioperative complications. Cochrane Database Syst Rev. 2003;(4):CD004423.
- Nygren J, Soop M, Thorell A, et al. Preoperative oral carbohydrate administration reduces postoperative insulin resistance. Clin Nutr. 1998;17:65-71.
- Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152:e170504.
- Kalogera, E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Rahbari NN, Zimmermann JB, Schmidt T, et al. Meta-analysis of standard, restrictive and supplemental fluid administration in colorectal surgery. Br J Surg. 2009;96:331-341.
- Myles PS, Bellomo R, Corcoran T, et al; Australian and New Zealand College of Anaesthetists Clinical Trials Network and the Australian and New Zealand Intensive Care Society Clinical Trials Group. Restrictive versus liberal fluid therapy for major abdominal surgery. N Engl J Med. 2018;378:2263-2274.
- Kalogera E, Bakkum-Gamez JN, Jankowski CJ, et al. Enhanced recovery in gynecologic surgery. Obstet Gynecol. 2013;122(2 pt 1):319-328.
- Kalogera E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Nelson G, Altman AD, Nick A, et al. Guidelines for pre- and intra-operative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part I. Gynecol Oncol. 2016;140:313-322.
- Nelson G, Altman AD, Nick A, et al. Guidelines for postoperative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part II. Gynecol Oncol. 2016;140:323-332.
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481.
- Johnson MP, Kim SJ, Langstraat CL, et al. Using bundled interventions to reduce surgical site infection after major gynecologic cancer surgery. Obstet Gynecol. 2016;127:1135-1144.
- Kalogera E, Nitschmann CC, Dowdy SC, et al. A prospective algorithm to reduce anastomotic leaks after rectosigmoid resection for gynecologic malignancies. Gynecol Oncol. 2017;144:343-347.
- Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study LAP2. J Clin Oncol. 2009;27:5331-5336.
- Fader AN, Weise RM, Sinno AK, et al. Utilization of minimally invasive surgery in endometrial cancer care: a quality and cost disparity. Obstet Gynecol. 2016;127:91-100.
Our specialty is focusing now more intently on perioperative optimization, commonly referred to as enhanced recovery after surgery (ERAS), a concept championed first and most visibly by colorectal surgeons in the 1990s.1 Both academic and nonacademic practices are challenging long-held beliefs about perioperative management.
The 3 tenets of ERAS
In multiple surgical specialties, proper implementation of 3 tenets—early feeding, perioperative euvolemia, and multimodal pain control—reduces the length of hospital stay, improves patient satisfaction, reduces complications, lowers health care costs, and most importantly hastens patient recovery.
1 Early feeding
Just as athletes hydrate and carbohydrate load prior to a competition, patients benefit if fluids and calories are not withheld in anticipation of a physiologically stressful surgical procedure. Similarly, modest benefit is associated with carbohydrate loading as a liquid supplement 2 hours before surgery.2 The American Society of Anesthesiologists guidelines state that while solid foods should not be consumed after midnight before surgery, clear liquids safely may be withheld for only 2 hours prior to anesthesia induction, and systematic reviews have failed to show harm.3,4 All patients, including those undergoing colonic resections, are allowed to eat a general diet as tolerated the evening before surgery, supplemented with caloric-dense nutritional supplements.
2 Multimodal pain control
Postsurgical pain is a top patient concern. Pain control is critical for rapid recovery; it helps avoid upregulation of the sympathetic axis and permits ambulation and resumption of normal activities. Although opioids relieve pain, they should not be considered a primary pain control approach.
Responding to the opioid epidemic, in 2015 the Centers for Disease Control and Prevention identified opioid overdose prevention as one of the top 5 public health challenges; notably, approximately 6% of patients will experience new, persistent opioid use following surgery.5 Optimal pain management therefore should provide effective pain relief while minimizing opioid use.
Preemptive oral acetaminophen, gabapentin, and celecoxib should be used routinely prior to incision; nonsteroidal anti-inflammatory drugs should be scheduled postoperatively. Even after a complex cytoreductive laparotomy, pain may be controlled with oral rather than intravenous (IV) medications in most patients, with opioid requirements averaging just 2 to 4 tablets of oxycodone in the first 48 hours after surgery, in our experience. The most critical need for pain medications occurs in the first 48 hours after surgery, which highlights the importance of local or regional analgesia. In one investigation, implementation of multimodal pain management that included incisional injection of liposomal bupivacaine reduced patient-controlled analgesia use to less than 5% after laparotomy.6 The need for opioids more than a week postoperatively is uncommon even after a laparotomy.
3 Perioperative euvolemia
Maintaining euvolemia is a central and underrecognized tenet of enhanced recovery pathways, and it facilitates the other 2 tenets of early feeding and optimal pain control. Overhydrated patients have more pain and prolonged recovery of bowel function. Unfortunately, euvolemia is the most difficult ERAS component to implement, requiring seamless communication between all members of the surgical team.
Continue to: Fluid therapy...
Fluid therapy should be respected as a pharmacologic agent with both benefits and risks. Recognizing that a single liter of lactated Ringer’s solution contains the sodium load of more than 30 bags of potato chips (and normal saline contains far more), one can imagine the impact of 10 L of solution on peripheral and bowel edema and on overall recovery. Importantly, euvolemia must be initiated during surgery. A meta-analysis of nearly 1,000 randomly assigned patients showed that benefits were limited when euvolemia was initiated in the postoperative period.7
When it comes to maintaining euvolemia, particular care must be taken to avoid erring toward hyperadherence. No difference in hospital length of stay, complications, or ileus was observed when patients were randomly assigned to goal-directed fluid therapy or standard practice.8 However, differences in the volume of fluid administered were relatively small, and while there was evidence of underhydration (likely responsible for acute kidney injury), there was no evidence of overhydration. For example, 4 L of fluid is likely superior to 15 L, but it may not be clinically different from 4.5 L. A threshold of fluid restriction is likely to be reached; that is, additional benefit is not achieved and, instead, detrimental effects may occur.
Rather than a specific directive, a more clinically relevant goal may be to replace insensible fluid losses and to maintain perfusion and blood pressure with the lowest volume possible. Note that estimation of fluid requirements is vastly simplified by omitting mechanical bowel preparation. Postoperatively, permissive oliguria (20 mL/h) is allowed since reduced urine output is a normal response to surgery (as a result of inappropriate secretion of antidiuretic hormone) and does not necessitate administration of a fluid bolus. Above all, anesthesiologists should acknowledge that fluid administration’s effects on a patient extend past the postanesthesia care unit, and the entire surgical team should be invested in the patient’s long-term recovery.
Our experience with ERAS
In 2011, Mayo Clinic was the first institution to implement enhanced recovery on a large scale in gynecologic surgery. We have subsequently made multiple pathway modifications in the spirit of continuous improvement (FIGURE 1).

For patients with ovarian cancer requiring extended procedures for cytoreduction via laparotomy (such as colon resection, splenectomy, diaphragm resection), enhanced recovery reduced the median hospital stay by 3 days, patient-controlled IV analgesia use by 88%, and postoperative opioid requirements by 90%.9,10
At 48 hours after surgery, 40% of our patients require no opioids or tramadol, and epidurals are not utilized because of their effects on ambulation and the potential for hypotension. These reductions were met with stable to improved pain scores, a 60% decrease in nausea, and a 50% reduction in adynamic ileus.9,10
Our initial efforts reduced 30-day costs of care by more than $850,000 in just 6 months, with savings of more than $7,600 for each patient undergoing a complex cytoreduction. Furthermore, these improvements allowed consolidation of our inpatient unit with those of other surgical specialties, serving higher volumes of patients within a smaller inpatient footprint. This contraction of inpatient services has accounted for an additional $1.1 million in savings every year since implementation (FIGURE 2).9,10
Our group is not alone in realizing these benefits, and interest has intensified as demonstrated by the fact that the ERAS Society guidelines are among the all-time most downloaded articles in Gynecologic Oncology.11,12 Although our research to demonstrate safety has focused on women undergoing complex oncologic operations, ERAS nevertheless hastens recovery, improves patient satisfaction, and adds value for all patients undergoing gynecologic surgery.

Continue to: Collateral improvements to practice...
Collateral improvements to practice
Clinical optimization using evidence-based practices such as enhanced recovery pathways can result in immediate patient benefit. Affecting such profound clinical improvements is energizing and creates a unique opportunity to transform the culture of the entire health care team. Irrespective of our provider roles (surgeon, anesthesiologist, nurse) or areas of interest (practice, research, education, leadership), we are united by a common purpose: to improve the human condition.13 Reaffirming this common purpose, through the collective effort involved in establishing a standardized enhanced recovery pathway, has allowed our practice and those of others to move beyond enhanced recovery and improve other areas of practice.
Other positive effects. The long-term collateral impact of this culture change at our institution is arguably more important than enhanced recovery itself. Examples of downstream impact include14,15:
- 80% reduction in surgical site infection
- 50% reduction in anastomotic leaks
- 60% reduction in blood utilization for patients undergoing surgery for ovarian cancer.
Team-based pragmatic strategies. Additionally, our willingness to make decisions as a division rather than as individuals facilitated universal implementation of sentinel lymph node biopsy for patients with endometrial cancer and standardized imaging, testing, and surgical decision making for patients with ovarian and endometrial cancer.
The interventions associated with these improvements were not tested in a randomized fashion; however, rather than await perfect data, we made informed decisions based on imperfect data together with a commitment to continuous data review. We find this to be an effective strategy if our goal is to ensure that tomorrow’s outcomes will be better than yesterday’s. In this way, pragmatic trials can be extremely effective in rural settings and tertiary centers.
Barriers to innovation
The widely reported benefits of enhanced recovery beg the question, Why has enhanced recovery not been adopted universally as standard of care? The answer is multifaceted and highlights long-standing shortcomings in our health care system.
Most importantly, our health care system lacks a robust interface to link discovery of new techniques, treatments, and workflows to clinical practice. Perhaps the best example of this is the adoption of minimally invasive surgery (MIS) for endometrial cancer. Ten years have passed since randomized trials showed MIS has equivalent oncologic outcomes and superior recovery compared to laparotomy, yet in the United States less than 50% of women with endometrial cancer benefit.16,17
However, even surgeons who are knowledgeable about recent innovations and genuinely wish to promote improvements may face near-insurmountable skepticism. Blind faith in our abilities and outcomes, overprotection of autonomy, close-mindedness, and satisfaction with the status quo are common responses to innovation and are the enemies of change. Resistance often comes from good intentions, but our desire to avoid complications may result in actions that could just as accurately be labeled superstitious as conservative. These observations suggest that developing methods to incorporate evidence-based practice into routine clinical use is the rate-limiting step in improving surgical quality.
The national Improving Surgical Care and Recovery program is available to specifically aid with ERAS implementation. A collaboration between the Agency for Healthcare Research and Quality (AHRQ) and the American College of Surgeons, the program aims to diffuse enhanced recovery to 750 service lines in 4 surgical subspecialties, including gynecologic surgery, over the next 5 years. (Note: The author is the content expert for the gynecology portion of this program.) The program’s larger aim is to measurably improve patient outcomes, reduce health care utilization, and improve patient experience through the use of an adaptation to AHRQ’s Comprehensive Unit-based Safety Program (CUSP). The backbone for this program is the recent systematic review to establish best practices for gynecologic surgery.1 Free to all participants, the program includes resources such as webinars and coaching calls to assist with the inevitable barriers to ERAS implementation. For more information and to enroll, visit https://www.ahrq.gov/professionals/quality-patient-safety/hais/tools/enhanced-recovery /index.html. An important aspect of the program is a registry for tracking outcomes and identifying areas for improvement. For members who currently participate in the National Surgical Quality Improvement Program, clinical data are automatically uploaded into the database. Programs such as Improving Surgical Care and Recovery may be the most reliable way to facilitate diffusion of best practices and take collective responsibility for not only “my outcomes” but also for “our outcomes” as a national community of gynecologic surgeons.
Reference
1. Kalogera E, Nelson G, Liu J, et al. Surgical technical evidence review for gynecologic surgery conducted for the Agency for Healthcare Research and Quality Safety Program for Improving Surgical Care and Recovery. Am J Obstet Gynecol. 2018;219:563.e1-563.e19.
Principles essential to change
Various methodologies have been described to manage change and facilitate implementation of new workflows and practices. Irrespective of the method used, including the more formal discipline of implementation science, at least 4 principles must be followed:
1. Teamwork. Mutual trust, mutual respect, and a sense of common purpose are minimum requirements for any successful initiative. Standardization is difficult or impossible without these elements. Thus, establishing a healthy team is the first step in implementing change.
2. Stakeholder analysis. Feedback from surgeons, nurses, residents, fellows, anesthesiologists, pharmacists, nurse anesthetists, and administrators is necessary to obtain diverse perspectives, facilitate engagement, and promote collaborative management. Negativity and resistance are common reactions to change, and it is particularly important to include those who are most skeptical in the stakeholder analysis to mitigate sabotage.
3. Concrete metrics. Success is possible only if defined a priori by specific and achievable goals. Counterbalances also are important to ensure that interventions do not have unintended consequences. Once a goal is met (for example, reduced hospital length of stay or costs), relevant metrics should be monitored after project completion for a minimum of 3 years to avoid regression to the pre-project state.
4. Leadership. The project champion responsible for the initiative must objectively facilitate all of the above and ensure excellent communication between stakeholders to nurture long-term engagement. Despite best efforts, if a minority of the group rejects compromise, this creates an opportunity to compare outcomes between those who do and do not accept the proposed change. Progress realized by early adopters may convince resistors to conform at a later time. Alternatively, the project champion also must have the insight to recognize when a proposed change is impossible at that point in time with that particular group. For example, our own initial attempts to implement enhanced recovery stalled in 2008, but they were successful 3 years later in a different environment.
Continue to: Although a discussion of leadership styles...
Although a discussion of leadership styles is beyond the scope of this article, in our experience, the most successful model is one of servant leadership that is team oriented rather than star dominated. Rather than being led by a single surgeon, each of the 4 quality improvement projects reviewed above (ERAS, and reductions in anastomotic leak, surgical site infection, and blood transfusion) that grew from enhanced recovery included trainees and was led by a different champion, encouraging teamwork and promoting career development. Such a model also supports the Accreditation Council for Graduate Medical Education’s emphasis on quality improvement education.
Our specialty is focusing now more intently on perioperative optimization, commonly referred to as enhanced recovery after surgery (ERAS), a concept championed first and most visibly by colorectal surgeons in the 1990s.1 Both academic and nonacademic practices are challenging long-held beliefs about perioperative management.
The 3 tenets of ERAS
In multiple surgical specialties, proper implementation of 3 tenets—early feeding, perioperative euvolemia, and multimodal pain control—reduces the length of hospital stay, improves patient satisfaction, reduces complications, lowers health care costs, and most importantly hastens patient recovery.
1 Early feeding
Just as athletes hydrate and carbohydrate load prior to a competition, patients benefit if fluids and calories are not withheld in anticipation of a physiologically stressful surgical procedure. Similarly, modest benefit is associated with carbohydrate loading as a liquid supplement 2 hours before surgery.2 The American Society of Anesthesiologists guidelines state that while solid foods should not be consumed after midnight before surgery, clear liquids safely may be withheld for only 2 hours prior to anesthesia induction, and systematic reviews have failed to show harm.3,4 All patients, including those undergoing colonic resections, are allowed to eat a general diet as tolerated the evening before surgery, supplemented with caloric-dense nutritional supplements.
2 Multimodal pain control
Postsurgical pain is a top patient concern. Pain control is critical for rapid recovery; it helps avoid upregulation of the sympathetic axis and permits ambulation and resumption of normal activities. Although opioids relieve pain, they should not be considered a primary pain control approach.
Responding to the opioid epidemic, in 2015 the Centers for Disease Control and Prevention identified opioid overdose prevention as one of the top 5 public health challenges; notably, approximately 6% of patients will experience new, persistent opioid use following surgery.5 Optimal pain management therefore should provide effective pain relief while minimizing opioid use.
Preemptive oral acetaminophen, gabapentin, and celecoxib should be used routinely prior to incision; nonsteroidal anti-inflammatory drugs should be scheduled postoperatively. Even after a complex cytoreductive laparotomy, pain may be controlled with oral rather than intravenous (IV) medications in most patients, with opioid requirements averaging just 2 to 4 tablets of oxycodone in the first 48 hours after surgery, in our experience. The most critical need for pain medications occurs in the first 48 hours after surgery, which highlights the importance of local or regional analgesia. In one investigation, implementation of multimodal pain management that included incisional injection of liposomal bupivacaine reduced patient-controlled analgesia use to less than 5% after laparotomy.6 The need for opioids more than a week postoperatively is uncommon even after a laparotomy.
3 Perioperative euvolemia
Maintaining euvolemia is a central and underrecognized tenet of enhanced recovery pathways, and it facilitates the other 2 tenets of early feeding and optimal pain control. Overhydrated patients have more pain and prolonged recovery of bowel function. Unfortunately, euvolemia is the most difficult ERAS component to implement, requiring seamless communication between all members of the surgical team.
Continue to: Fluid therapy...
Fluid therapy should be respected as a pharmacologic agent with both benefits and risks. Recognizing that a single liter of lactated Ringer’s solution contains the sodium load of more than 30 bags of potato chips (and normal saline contains far more), one can imagine the impact of 10 L of solution on peripheral and bowel edema and on overall recovery. Importantly, euvolemia must be initiated during surgery. A meta-analysis of nearly 1,000 randomly assigned patients showed that benefits were limited when euvolemia was initiated in the postoperative period.7
When it comes to maintaining euvolemia, particular care must be taken to avoid erring toward hyperadherence. No difference in hospital length of stay, complications, or ileus was observed when patients were randomly assigned to goal-directed fluid therapy or standard practice.8 However, differences in the volume of fluid administered were relatively small, and while there was evidence of underhydration (likely responsible for acute kidney injury), there was no evidence of overhydration. For example, 4 L of fluid is likely superior to 15 L, but it may not be clinically different from 4.5 L. A threshold of fluid restriction is likely to be reached; that is, additional benefit is not achieved and, instead, detrimental effects may occur.
Rather than a specific directive, a more clinically relevant goal may be to replace insensible fluid losses and to maintain perfusion and blood pressure with the lowest volume possible. Note that estimation of fluid requirements is vastly simplified by omitting mechanical bowel preparation. Postoperatively, permissive oliguria (20 mL/h) is allowed since reduced urine output is a normal response to surgery (as a result of inappropriate secretion of antidiuretic hormone) and does not necessitate administration of a fluid bolus. Above all, anesthesiologists should acknowledge that fluid administration’s effects on a patient extend past the postanesthesia care unit, and the entire surgical team should be invested in the patient’s long-term recovery.
Our experience with ERAS
In 2011, Mayo Clinic was the first institution to implement enhanced recovery on a large scale in gynecologic surgery. We have subsequently made multiple pathway modifications in the spirit of continuous improvement (FIGURE 1).

For patients with ovarian cancer requiring extended procedures for cytoreduction via laparotomy (such as colon resection, splenectomy, diaphragm resection), enhanced recovery reduced the median hospital stay by 3 days, patient-controlled IV analgesia use by 88%, and postoperative opioid requirements by 90%.9,10
At 48 hours after surgery, 40% of our patients require no opioids or tramadol, and epidurals are not utilized because of their effects on ambulation and the potential for hypotension. These reductions were met with stable to improved pain scores, a 60% decrease in nausea, and a 50% reduction in adynamic ileus.9,10
Our initial efforts reduced 30-day costs of care by more than $850,000 in just 6 months, with savings of more than $7,600 for each patient undergoing a complex cytoreduction. Furthermore, these improvements allowed consolidation of our inpatient unit with those of other surgical specialties, serving higher volumes of patients within a smaller inpatient footprint. This contraction of inpatient services has accounted for an additional $1.1 million in savings every year since implementation (FIGURE 2).9,10
Our group is not alone in realizing these benefits, and interest has intensified as demonstrated by the fact that the ERAS Society guidelines are among the all-time most downloaded articles in Gynecologic Oncology.11,12 Although our research to demonstrate safety has focused on women undergoing complex oncologic operations, ERAS nevertheless hastens recovery, improves patient satisfaction, and adds value for all patients undergoing gynecologic surgery.

Continue to: Collateral improvements to practice...
Collateral improvements to practice
Clinical optimization using evidence-based practices such as enhanced recovery pathways can result in immediate patient benefit. Affecting such profound clinical improvements is energizing and creates a unique opportunity to transform the culture of the entire health care team. Irrespective of our provider roles (surgeon, anesthesiologist, nurse) or areas of interest (practice, research, education, leadership), we are united by a common purpose: to improve the human condition.13 Reaffirming this common purpose, through the collective effort involved in establishing a standardized enhanced recovery pathway, has allowed our practice and those of others to move beyond enhanced recovery and improve other areas of practice.
Other positive effects. The long-term collateral impact of this culture change at our institution is arguably more important than enhanced recovery itself. Examples of downstream impact include14,15:
- 80% reduction in surgical site infection
- 50% reduction in anastomotic leaks
- 60% reduction in blood utilization for patients undergoing surgery for ovarian cancer.
Team-based pragmatic strategies. Additionally, our willingness to make decisions as a division rather than as individuals facilitated universal implementation of sentinel lymph node biopsy for patients with endometrial cancer and standardized imaging, testing, and surgical decision making for patients with ovarian and endometrial cancer.
The interventions associated with these improvements were not tested in a randomized fashion; however, rather than await perfect data, we made informed decisions based on imperfect data together with a commitment to continuous data review. We find this to be an effective strategy if our goal is to ensure that tomorrow’s outcomes will be better than yesterday’s. In this way, pragmatic trials can be extremely effective in rural settings and tertiary centers.
Barriers to innovation
The widely reported benefits of enhanced recovery beg the question, Why has enhanced recovery not been adopted universally as standard of care? The answer is multifaceted and highlights long-standing shortcomings in our health care system.
Most importantly, our health care system lacks a robust interface to link discovery of new techniques, treatments, and workflows to clinical practice. Perhaps the best example of this is the adoption of minimally invasive surgery (MIS) for endometrial cancer. Ten years have passed since randomized trials showed MIS has equivalent oncologic outcomes and superior recovery compared to laparotomy, yet in the United States less than 50% of women with endometrial cancer benefit.16,17
However, even surgeons who are knowledgeable about recent innovations and genuinely wish to promote improvements may face near-insurmountable skepticism. Blind faith in our abilities and outcomes, overprotection of autonomy, close-mindedness, and satisfaction with the status quo are common responses to innovation and are the enemies of change. Resistance often comes from good intentions, but our desire to avoid complications may result in actions that could just as accurately be labeled superstitious as conservative. These observations suggest that developing methods to incorporate evidence-based practice into routine clinical use is the rate-limiting step in improving surgical quality.
The national Improving Surgical Care and Recovery program is available to specifically aid with ERAS implementation. A collaboration between the Agency for Healthcare Research and Quality (AHRQ) and the American College of Surgeons, the program aims to diffuse enhanced recovery to 750 service lines in 4 surgical subspecialties, including gynecologic surgery, over the next 5 years. (Note: The author is the content expert for the gynecology portion of this program.) The program’s larger aim is to measurably improve patient outcomes, reduce health care utilization, and improve patient experience through the use of an adaptation to AHRQ’s Comprehensive Unit-based Safety Program (CUSP). The backbone for this program is the recent systematic review to establish best practices for gynecologic surgery.1 Free to all participants, the program includes resources such as webinars and coaching calls to assist with the inevitable barriers to ERAS implementation. For more information and to enroll, visit https://www.ahrq.gov/professionals/quality-patient-safety/hais/tools/enhanced-recovery /index.html. An important aspect of the program is a registry for tracking outcomes and identifying areas for improvement. For members who currently participate in the National Surgical Quality Improvement Program, clinical data are automatically uploaded into the database. Programs such as Improving Surgical Care and Recovery may be the most reliable way to facilitate diffusion of best practices and take collective responsibility for not only “my outcomes” but also for “our outcomes” as a national community of gynecologic surgeons.
Reference
1. Kalogera E, Nelson G, Liu J, et al. Surgical technical evidence review for gynecologic surgery conducted for the Agency for Healthcare Research and Quality Safety Program for Improving Surgical Care and Recovery. Am J Obstet Gynecol. 2018;219:563.e1-563.e19.
Principles essential to change
Various methodologies have been described to manage change and facilitate implementation of new workflows and practices. Irrespective of the method used, including the more formal discipline of implementation science, at least 4 principles must be followed:
1. Teamwork. Mutual trust, mutual respect, and a sense of common purpose are minimum requirements for any successful initiative. Standardization is difficult or impossible without these elements. Thus, establishing a healthy team is the first step in implementing change.
2. Stakeholder analysis. Feedback from surgeons, nurses, residents, fellows, anesthesiologists, pharmacists, nurse anesthetists, and administrators is necessary to obtain diverse perspectives, facilitate engagement, and promote collaborative management. Negativity and resistance are common reactions to change, and it is particularly important to include those who are most skeptical in the stakeholder analysis to mitigate sabotage.
3. Concrete metrics. Success is possible only if defined a priori by specific and achievable goals. Counterbalances also are important to ensure that interventions do not have unintended consequences. Once a goal is met (for example, reduced hospital length of stay or costs), relevant metrics should be monitored after project completion for a minimum of 3 years to avoid regression to the pre-project state.
4. Leadership. The project champion responsible for the initiative must objectively facilitate all of the above and ensure excellent communication between stakeholders to nurture long-term engagement. Despite best efforts, if a minority of the group rejects compromise, this creates an opportunity to compare outcomes between those who do and do not accept the proposed change. Progress realized by early adopters may convince resistors to conform at a later time. Alternatively, the project champion also must have the insight to recognize when a proposed change is impossible at that point in time with that particular group. For example, our own initial attempts to implement enhanced recovery stalled in 2008, but they were successful 3 years later in a different environment.
Continue to: Although a discussion of leadership styles...
Although a discussion of leadership styles is beyond the scope of this article, in our experience, the most successful model is one of servant leadership that is team oriented rather than star dominated. Rather than being led by a single surgeon, each of the 4 quality improvement projects reviewed above (ERAS, and reductions in anastomotic leak, surgical site infection, and blood transfusion) that grew from enhanced recovery included trainees and was led by a different champion, encouraging teamwork and promoting career development. Such a model also supports the Accreditation Council for Graduate Medical Education’s emphasis on quality improvement education.
- Nygren J, Thacker J, Carli F, et al; Enhanced Recovery After Surgery (ERAS) Society, for Perioperative Care; European Society for Clinical Nutrition and Metabolism (ESPEN); International Association for Surgical Metabolism and Nutrition (IASMEN). Guidelines for perioperative care in elective rectal/pelvic surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations. World J Surg. 2013;37:285-305.
- American Society of Anesthesiologists Committee. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: an updated report by the American Society of Anesthesiologists Committee on Standards and Practice Parameters. Anesthesiology. 2011;114: 495-511.
- Brady M, Kinn S, Stuart P. Preoperative fasting for adults to prevent perioperative complications. Cochrane Database Syst Rev. 2003;(4):CD004423.
- Nygren J, Soop M, Thorell A, et al. Preoperative oral carbohydrate administration reduces postoperative insulin resistance. Clin Nutr. 1998;17:65-71.
- Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152:e170504.
- Kalogera, E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Rahbari NN, Zimmermann JB, Schmidt T, et al. Meta-analysis of standard, restrictive and supplemental fluid administration in colorectal surgery. Br J Surg. 2009;96:331-341.
- Myles PS, Bellomo R, Corcoran T, et al; Australian and New Zealand College of Anaesthetists Clinical Trials Network and the Australian and New Zealand Intensive Care Society Clinical Trials Group. Restrictive versus liberal fluid therapy for major abdominal surgery. N Engl J Med. 2018;378:2263-2274.
- Kalogera E, Bakkum-Gamez JN, Jankowski CJ, et al. Enhanced recovery in gynecologic surgery. Obstet Gynecol. 2013;122(2 pt 1):319-328.
- Kalogera E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Nelson G, Altman AD, Nick A, et al. Guidelines for pre- and intra-operative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part I. Gynecol Oncol. 2016;140:313-322.
- Nelson G, Altman AD, Nick A, et al. Guidelines for postoperative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part II. Gynecol Oncol. 2016;140:323-332.
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481.
- Johnson MP, Kim SJ, Langstraat CL, et al. Using bundled interventions to reduce surgical site infection after major gynecologic cancer surgery. Obstet Gynecol. 2016;127:1135-1144.
- Kalogera E, Nitschmann CC, Dowdy SC, et al. A prospective algorithm to reduce anastomotic leaks after rectosigmoid resection for gynecologic malignancies. Gynecol Oncol. 2017;144:343-347.
- Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study LAP2. J Clin Oncol. 2009;27:5331-5336.
- Fader AN, Weise RM, Sinno AK, et al. Utilization of minimally invasive surgery in endometrial cancer care: a quality and cost disparity. Obstet Gynecol. 2016;127:91-100.
- Nygren J, Thacker J, Carli F, et al; Enhanced Recovery After Surgery (ERAS) Society, for Perioperative Care; European Society for Clinical Nutrition and Metabolism (ESPEN); International Association for Surgical Metabolism and Nutrition (IASMEN). Guidelines for perioperative care in elective rectal/pelvic surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations. World J Surg. 2013;37:285-305.
- American Society of Anesthesiologists Committee. Practice guidelines for preoperative fasting and the use of pharmacologic agents to reduce the risk of pulmonary aspiration: application to healthy patients undergoing elective procedures: an updated report by the American Society of Anesthesiologists Committee on Standards and Practice Parameters. Anesthesiology. 2011;114: 495-511.
- Brady M, Kinn S, Stuart P. Preoperative fasting for adults to prevent perioperative complications. Cochrane Database Syst Rev. 2003;(4):CD004423.
- Nygren J, Soop M, Thorell A, et al. Preoperative oral carbohydrate administration reduces postoperative insulin resistance. Clin Nutr. 1998;17:65-71.
- Brummett CM, Waljee JF, Goesling J, et al. New persistent opioid use after minor and major surgical procedures in US adults. JAMA Surg. 2017;152:e170504.
- Kalogera, E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Rahbari NN, Zimmermann JB, Schmidt T, et al. Meta-analysis of standard, restrictive and supplemental fluid administration in colorectal surgery. Br J Surg. 2009;96:331-341.
- Myles PS, Bellomo R, Corcoran T, et al; Australian and New Zealand College of Anaesthetists Clinical Trials Network and the Australian and New Zealand Intensive Care Society Clinical Trials Group. Restrictive versus liberal fluid therapy for major abdominal surgery. N Engl J Med. 2018;378:2263-2274.
- Kalogera E, Bakkum-Gamez JN, Jankowski CJ, et al. Enhanced recovery in gynecologic surgery. Obstet Gynecol. 2013;122(2 pt 1):319-328.
- Kalogera E, Bakkum-Gamez JN, Weaver AL, et al. Abdominal incision injection of liposomal bupivacaine and opioid use after laparotomy for gynecologic malignancies. Obstet Gynecol. 2016;128:1009-1017.
- Nelson G, Altman AD, Nick A, et al. Guidelines for pre- and intra-operative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part I. Gynecol Oncol. 2016;140:313-322.
- Nelson G, Altman AD, Nick A, et al. Guidelines for postoperative care in gynecologic/oncology surgery: Enhanced Recovery After Surgery (ERAS) Society recommendations--part II. Gynecol Oncol. 2016;140:323-332.
- Porter ME. What is value in health care? N Engl J Med. 2010;363:2477-2481.
- Johnson MP, Kim SJ, Langstraat CL, et al. Using bundled interventions to reduce surgical site infection after major gynecologic cancer surgery. Obstet Gynecol. 2016;127:1135-1144.
- Kalogera E, Nitschmann CC, Dowdy SC, et al. A prospective algorithm to reduce anastomotic leaks after rectosigmoid resection for gynecologic malignancies. Gynecol Oncol. 2017;144:343-347.
- Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: Gynecologic Oncology Group Study LAP2. J Clin Oncol. 2009;27:5331-5336.
- Fader AN, Weise RM, Sinno AK, et al. Utilization of minimally invasive surgery in endometrial cancer care: a quality and cost disparity. Obstet Gynecol. 2016;127:91-100.
2019 Update on prenatal exome sequencing
Prenatal diagnosis of genetic anomalies is important for diagnosing lethal genetic conditions before birth. It can provide information for parents regarding pregnancy options and allow for recurrence risk counseling and the potential use of preimplantation genetic testing in the next pregnancy. For decades, a karyotype was used to analyze amniocentesis and chorionic villus sampling specimens; in recent years, chromosomal microarray analysis provides more information about significant chromosomal abnormalities, including microdeletions and microduplications. However, microarrays also have limitations, as they do not identify base pair changes associated with single-gene disorders.
The advent of next-generation sequencing has substantially reduced the cost of DNA sequencing. Whole genome sequencing (WGS) can sequence the entire genome— both the coding (exonic) and noncoding (intronic) regions—while exome sequencing analyzes only the protein-coding exons, which make up 1% to 2% of the genome and about 85% of the protein-coding genes associated with known human disease. Exome sequencing increasingly is used in cases of suspected genetic disorders when other tests have been unrevealing.
In this Update, we review recent reports of prenatal exome sequencing, including studies exploring the yield in fetuses with structural anomalies; the importance of prenatal phenotyping; the perspectives of parents and health care professionals who were involved in prenatal exome sequencing studies; and a summary of a joint position statement from 3 societies regarding prenatal sequencing.
Prenatal whole exome sequencing has potential utility, with some limitations
Petrovski S, Aggarwal V, Giordano JL, et al. Whole-exome sequencing in the evaluation of fetal structural anomalies: a prospective cohort study. Lancet. 2019;393:758-767.
Lord J, McMullan DJ, Eberhardt RY, et al; for the Prenatal Assessment of Genomes and Exomes Consortium. Prenatal exome sequencing analysis in fetal structural anomalies detected by ultrasonography (PAGE): a cohort study. Lancet. 2019;393:747-757.
Exome sequencing has been shown to identify an underlying genetic cause in 25% to 30% of children with an undiagnosed suspected genetic disorder. Two studies recently published in the Lancet sought to determine the incremental diagnostic yield of prenatal whole exome sequencing (WES) in the setting of fetal structural anomalies when karyotype and microarray results were normal.
Continue to: Details of the studies...
Details of the studies
In a prospective cohort study by Petrovski and colleagues, DNA samples from 234 fetuses with a structural anomaly (identified on ultrasonography) and both parents (parent-fetus "trios") were used for analysis. WES identified diagnostic genetic variants in 24 trios (10%). An additional 46 (20%) had variants that indicated pathogenicity but without sufficient evidence to be considered diagnostic.
The anomalies with the highest frequency of a genetic diagnosis were lymphatic, 24%; skeletal, 24%; central nervous system, 22%; and renal, 16%; while cardiac anomalies had the lowest yield at 5%.
In another prospective cohort study, known as the Prenatal Assessment of Genomes and Exomes (PAGE), Lord and colleagues sequenced DNA samples from 610 parent-fetus trios, but they restricted sequencing to a predefined list of 1,628 genes. Diagnostic genetic variants were identified in 52 fetuses (8.5%), while 24 (3.9%) had a variant of uncertain significance that was thought to be of potential clinical usefulness.
Fetuses with multiple anomalies had the highest genetic yield (15.4%), followed by skeletal (15.4%) and cardiac anomalies (11.1%), with the lowest yield in fetuses with isolated increased nuchal translucency (3.2%).
Diagnostic yield is high, but prenatal utility is limited
Both studies showed a clinically significant diagnostic yield of 8% to 10% for prenatal exome sequencing in cases of fetal structural anomalies with normal karyotype and microarray testing. While this yield demonstrates the utility of prenatal exome sequencing, it is significantly lower than what has been reported in postnatal studies. One of the reasons for this is the inherent limitation of prenatal phenotyping (discussed below).
The cohort studies by both Petrovski and Lord and their colleagues show the feasibility and potential diagnostic utility of exome sequencing in cases of fetal structural anomalies where karyotype and microarray are not diagnostic. However, the lower yield found in these studies compared with those in postnatal studies highlights in part the limitations of prenatal phenotyping.
The importance of prenatal phenotyping
Aarabi M, Sniezek O, Jiang H, et al. Importance of complete phenotyping in prenatal whole exome sequencing. Hum Genet. 2018;137:175-181.
In postnatal exome sequencing, the physical exam, imaging findings, and laboratory results are components of the phenotype that are used to interpret the sequencing data. Prenatal phenotyping, however, is limited to the use of fetal ultrasonography and, occasionally, the addition of magnetic resonance imaging. Prenatal phenotyping is without the benefit of an exam to detect more subtle anomalies or functional status, such as developmental delay, seizures, or failure to thrive.
When a structural anomaly is identified on prenatal ultrasonography, it is especially important that detailed imaging be undertaken to detect other anomalies, including more subtle facial features and dysmorphology.
Value of reanalyzing exome sequencing data
Aarabi and colleagues conducted a retrospective study of 20 fetuses with structural anomalies and normal karyotype and microarray. They performed trio exome sequencing first using information available only prenatally and then conducted a reanalysis using information available after delivery.
With prenatal phenotyping only, the investigators identified no pathogenic, or likely pathogenic, variants. On reanalysis of combined prenatal and postnatal findings, however, they identified pathogenic variants in 20% of cases.
Significance of the findings
This study highlights both the importance of a careful, detailed fetal ultrasonography study and the possible additional benefit of a postnatal examination (such as an autopsy) in order to yield improved results. In addition, the authors noted that the development of a prenatal phenotype-genotype database would significantly help exome sequencing interpretation in the prenatal setting.
Careful prenatal ultrasonography is crucial to help in the interpretation of prenatal exome sequencing. Patients who have undergone prenatal clinical exome sequencing may benefit from reanalysis of the genetic data based on detailed postnatal findings.
Social impact of WES: Parent and provider perspectives
Wou K, Weitz T, McCormack C, et al. Parental perceptions of prenatal whole exome sequencing (PPPWES) study. Prenat Diagn. 2018;38:801-811.
Horn R, Parker M. Health professionals' and researchers' perspectives on prenatal whole genome and exome sequencing: 'We can't shut the door now, the genie's out, we need to refine it.' PLoS One. 2018;13:e0204158.
As health care providers enter a new era of prenatal genetic testing with exome sequencing, it is crucial to the path forward that we obtain perspectives from the parents and providers who participated in these studies. Notably, in both of the previously discussed Lancet reports, the authors interviewed the participants to discuss the challenges involved and identify strategies for improving future testing.
Continue to: What parents want...
What parents want
To ascertain the perceptions of couples who underwent prenatal WES, Wou and colleagues conducted semi-structured interviews with participants from the Fetal Sequencing Study regarding their experience. They interviewed 29 parents from 17 pregnancies, including a mix of those who had pathogenic prenatal results, terminated prior to receiving the results, and had normal results.
Expressed feelings and desires. Parents recalled feelings of anxiety and stress around the time of diagnosis and the need for help with coping while awaiting results. The majority of parents reported that they would like to be told about uncertain results, but that desire decreased as the certainty of results decreased.
Parents were overall satisfied with the prenatal genetic testing experience, but they added that they would have liked to receive written materials beforehand and a written report of the test results (including negative cases). They also would like to have connected with other families with similar experiences, to have received results sooner, and to have an in-person meeting after telephone disclosure of the results.
Health professionals articulate complexity of prenatal genomics
In a qualitative interview study to explore critical issues involved in the clinical practice use of prenatal genomics, Horn and Parker conducted interviews with 20 health care professionals who were involved in the previously described PAGE trial. Patient recruiters, midwives, genetic counselors, research assistants, and laboratory staff were included.
Interviewees cited numerous challenges involved in their day-to-day work with prenatal whole genome and exome sequencing, including:
- the complexity of achieving valid parental consent at a time of vulnerability
- management of parent expectations
- transmitting and comprehending complex information
- the usefulness of information
- the difficulty of a long turnaround time for study results.
All the interviewees agreed that prenatal exome sequencing studies contribute to knowledge generation and the advancement of technology.
The authors concluded that an appropriate next step would be the development of appropriate guidelines for good ethical practice that address the concerns encountered in genomics clinical practice.
The prenatal experience can be overwhelming for parents. Pretest and posttest counseling on genetic testing and results are of the utmost importance, as is finding ways to help support parents through this anxious time.
Societies offer guidance on using genome and exome sequencing
International Society for Prenatal Diagnosis, Society for Maternal and Fetal Medicine, Perinatal Quality Foundation. Joint Position Statement from the International Society for Prenatal Diagnosis (ISPD), the Society for Maternal Fetal Medicine (SMFM), and the Perinatal Quality Foundation (PQF) on the use of genome-wide sequencing for fetal diagnosis. Prenat Diagn. 2018;38:6-9.
In response to the rapid integration of exome sequencing for genetic diagnosis, several professional societies—the International Society for Prenatal Diagnosis, Society for Maternal Fetal Medicine, and Perinatal Quality Foundation—issued a joint statement addressing the clinical use of prenatal diagnostic genome wide sequencing, including exome sequencing.
Continue to: Guidance at a glance...
Guidance at a glance
The societies' recommendations are summarized as follows:
- Exome sequencing is best done as a trio analysis, with fetal and both parental samples sequenced and analyzed together.
- Extensive pretest education, counseling, and informed consent, as well as posttest counseling, are essential. This should include:
—the types of results to be conveyed (variants that are pathogenic, likely pathogenic, of uncertain significance, likely benign, and benign)
—the possibility that results will not be obtained or may not be available before the birth of the fetus
—realistic expectations regarding the likelihood that a significant result will be obtained
—the timeframe to results
—the option to include or exclude in the results incidental or secondary findings (such as an unexpected childhood disorder, cancer susceptibility genes, adult-onset disorders)
—the possibility of uncovering nonpaternity or consanguinity
—the potential reanalysis of results over time
—how data are stored, who has access, and for what purpose.
- Fetal sequencing may be beneficial in the following scenarios:
—multiple fetal anomalies or a single major anomaly suggestive of a genetic disorder, when the microarray is negative
—no microarray result is available, but the fetus exhibits a pattern of anomalies strongly suggestive of a single-gene disorder
—a prior undiagnosed fetus (or child) with anomalies suggestive of a genetic etiology, and with similar anomalies in the current pregnancy, with normal karyotype or microarray. Providers also can consider sequencing samples from both parents prior to preimplantation genetic testing to check for shared carrier status for autosomal recessive mutations, although obtaining exome sequencing from the prior affected fetus (or child) is ideal.
—history of recurrent stillbirths of unknown etiology, with a recurrent pattern of anomalies in the current pregnancy, with normal karyotype or microarray.
- Interpretation of results should be done using a multidisciplinary team-based approach, including clinical scientists, geneticists, genetic counselors, and experts in prenatal diagnosis.
- Where possible and after informed consent, reanalysis of results should be undertaken if a future pregnancy is planned or ongoing, and a significant amount of time has elapsed since the time the result was last reported.
- Parents should be given a written report of test results.
Three professional societies have convened to issue consensus opinion that includes current indications for prenatal exome sequencing and important factors to include in the consent process. We follow these guidelines in our own practice.
Summary
Exome sequencing is increasingly becoming mainstream in postnatal genetic testing, and it is emerging as the newest diagnostic frontier in prenatal genetic testing. However, there are limitations to prenatal exome sequencing, including issues with consent at a vulnerable time for parents, limited information available regarding the phenotype, and results that may not be available before the birth of a fetus. Providers should be familiar with the indications for testing, the possible results, the limitations of prenatal phenotyping, and the implications for future pregnancies.
Prenatal diagnosis of genetic anomalies is important for diagnosing lethal genetic conditions before birth. It can provide information for parents regarding pregnancy options and allow for recurrence risk counseling and the potential use of preimplantation genetic testing in the next pregnancy. For decades, a karyotype was used to analyze amniocentesis and chorionic villus sampling specimens; in recent years, chromosomal microarray analysis provides more information about significant chromosomal abnormalities, including microdeletions and microduplications. However, microarrays also have limitations, as they do not identify base pair changes associated with single-gene disorders.
The advent of next-generation sequencing has substantially reduced the cost of DNA sequencing. Whole genome sequencing (WGS) can sequence the entire genome— both the coding (exonic) and noncoding (intronic) regions—while exome sequencing analyzes only the protein-coding exons, which make up 1% to 2% of the genome and about 85% of the protein-coding genes associated with known human disease. Exome sequencing increasingly is used in cases of suspected genetic disorders when other tests have been unrevealing.
In this Update, we review recent reports of prenatal exome sequencing, including studies exploring the yield in fetuses with structural anomalies; the importance of prenatal phenotyping; the perspectives of parents and health care professionals who were involved in prenatal exome sequencing studies; and a summary of a joint position statement from 3 societies regarding prenatal sequencing.
Prenatal whole exome sequencing has potential utility, with some limitations
Petrovski S, Aggarwal V, Giordano JL, et al. Whole-exome sequencing in the evaluation of fetal structural anomalies: a prospective cohort study. Lancet. 2019;393:758-767.
Lord J, McMullan DJ, Eberhardt RY, et al; for the Prenatal Assessment of Genomes and Exomes Consortium. Prenatal exome sequencing analysis in fetal structural anomalies detected by ultrasonography (PAGE): a cohort study. Lancet. 2019;393:747-757.
Exome sequencing has been shown to identify an underlying genetic cause in 25% to 30% of children with an undiagnosed suspected genetic disorder. Two studies recently published in the Lancet sought to determine the incremental diagnostic yield of prenatal whole exome sequencing (WES) in the setting of fetal structural anomalies when karyotype and microarray results were normal.
Continue to: Details of the studies...
Details of the studies
In a prospective cohort study by Petrovski and colleagues, DNA samples from 234 fetuses with a structural anomaly (identified on ultrasonography) and both parents (parent-fetus "trios") were used for analysis. WES identified diagnostic genetic variants in 24 trios (10%). An additional 46 (20%) had variants that indicated pathogenicity but without sufficient evidence to be considered diagnostic.
The anomalies with the highest frequency of a genetic diagnosis were lymphatic, 24%; skeletal, 24%; central nervous system, 22%; and renal, 16%; while cardiac anomalies had the lowest yield at 5%.
In another prospective cohort study, known as the Prenatal Assessment of Genomes and Exomes (PAGE), Lord and colleagues sequenced DNA samples from 610 parent-fetus trios, but they restricted sequencing to a predefined list of 1,628 genes. Diagnostic genetic variants were identified in 52 fetuses (8.5%), while 24 (3.9%) had a variant of uncertain significance that was thought to be of potential clinical usefulness.
Fetuses with multiple anomalies had the highest genetic yield (15.4%), followed by skeletal (15.4%) and cardiac anomalies (11.1%), with the lowest yield in fetuses with isolated increased nuchal translucency (3.2%).
Diagnostic yield is high, but prenatal utility is limited
Both studies showed a clinically significant diagnostic yield of 8% to 10% for prenatal exome sequencing in cases of fetal structural anomalies with normal karyotype and microarray testing. While this yield demonstrates the utility of prenatal exome sequencing, it is significantly lower than what has been reported in postnatal studies. One of the reasons for this is the inherent limitation of prenatal phenotyping (discussed below).
The cohort studies by both Petrovski and Lord and their colleagues show the feasibility and potential diagnostic utility of exome sequencing in cases of fetal structural anomalies where karyotype and microarray are not diagnostic. However, the lower yield found in these studies compared with those in postnatal studies highlights in part the limitations of prenatal phenotyping.
The importance of prenatal phenotyping
Aarabi M, Sniezek O, Jiang H, et al. Importance of complete phenotyping in prenatal whole exome sequencing. Hum Genet. 2018;137:175-181.
In postnatal exome sequencing, the physical exam, imaging findings, and laboratory results are components of the phenotype that are used to interpret the sequencing data. Prenatal phenotyping, however, is limited to the use of fetal ultrasonography and, occasionally, the addition of magnetic resonance imaging. Prenatal phenotyping is without the benefit of an exam to detect more subtle anomalies or functional status, such as developmental delay, seizures, or failure to thrive.
When a structural anomaly is identified on prenatal ultrasonography, it is especially important that detailed imaging be undertaken to detect other anomalies, including more subtle facial features and dysmorphology.
Value of reanalyzing exome sequencing data
Aarabi and colleagues conducted a retrospective study of 20 fetuses with structural anomalies and normal karyotype and microarray. They performed trio exome sequencing first using information available only prenatally and then conducted a reanalysis using information available after delivery.
With prenatal phenotyping only, the investigators identified no pathogenic, or likely pathogenic, variants. On reanalysis of combined prenatal and postnatal findings, however, they identified pathogenic variants in 20% of cases.
Significance of the findings
This study highlights both the importance of a careful, detailed fetal ultrasonography study and the possible additional benefit of a postnatal examination (such as an autopsy) in order to yield improved results. In addition, the authors noted that the development of a prenatal phenotype-genotype database would significantly help exome sequencing interpretation in the prenatal setting.
Careful prenatal ultrasonography is crucial to help in the interpretation of prenatal exome sequencing. Patients who have undergone prenatal clinical exome sequencing may benefit from reanalysis of the genetic data based on detailed postnatal findings.
Social impact of WES: Parent and provider perspectives
Wou K, Weitz T, McCormack C, et al. Parental perceptions of prenatal whole exome sequencing (PPPWES) study. Prenat Diagn. 2018;38:801-811.
Horn R, Parker M. Health professionals' and researchers' perspectives on prenatal whole genome and exome sequencing: 'We can't shut the door now, the genie's out, we need to refine it.' PLoS One. 2018;13:e0204158.
As health care providers enter a new era of prenatal genetic testing with exome sequencing, it is crucial to the path forward that we obtain perspectives from the parents and providers who participated in these studies. Notably, in both of the previously discussed Lancet reports, the authors interviewed the participants to discuss the challenges involved and identify strategies for improving future testing.
Continue to: What parents want...
What parents want
To ascertain the perceptions of couples who underwent prenatal WES, Wou and colleagues conducted semi-structured interviews with participants from the Fetal Sequencing Study regarding their experience. They interviewed 29 parents from 17 pregnancies, including a mix of those who had pathogenic prenatal results, terminated prior to receiving the results, and had normal results.
Expressed feelings and desires. Parents recalled feelings of anxiety and stress around the time of diagnosis and the need for help with coping while awaiting results. The majority of parents reported that they would like to be told about uncertain results, but that desire decreased as the certainty of results decreased.
Parents were overall satisfied with the prenatal genetic testing experience, but they added that they would have liked to receive written materials beforehand and a written report of the test results (including negative cases). They also would like to have connected with other families with similar experiences, to have received results sooner, and to have an in-person meeting after telephone disclosure of the results.
Health professionals articulate complexity of prenatal genomics
In a qualitative interview study to explore critical issues involved in the clinical practice use of prenatal genomics, Horn and Parker conducted interviews with 20 health care professionals who were involved in the previously described PAGE trial. Patient recruiters, midwives, genetic counselors, research assistants, and laboratory staff were included.
Interviewees cited numerous challenges involved in their day-to-day work with prenatal whole genome and exome sequencing, including:
- the complexity of achieving valid parental consent at a time of vulnerability
- management of parent expectations
- transmitting and comprehending complex information
- the usefulness of information
- the difficulty of a long turnaround time for study results.
All the interviewees agreed that prenatal exome sequencing studies contribute to knowledge generation and the advancement of technology.
The authors concluded that an appropriate next step would be the development of appropriate guidelines for good ethical practice that address the concerns encountered in genomics clinical practice.
The prenatal experience can be overwhelming for parents. Pretest and posttest counseling on genetic testing and results are of the utmost importance, as is finding ways to help support parents through this anxious time.
Societies offer guidance on using genome and exome sequencing
International Society for Prenatal Diagnosis, Society for Maternal and Fetal Medicine, Perinatal Quality Foundation. Joint Position Statement from the International Society for Prenatal Diagnosis (ISPD), the Society for Maternal Fetal Medicine (SMFM), and the Perinatal Quality Foundation (PQF) on the use of genome-wide sequencing for fetal diagnosis. Prenat Diagn. 2018;38:6-9.
In response to the rapid integration of exome sequencing for genetic diagnosis, several professional societies—the International Society for Prenatal Diagnosis, Society for Maternal Fetal Medicine, and Perinatal Quality Foundation—issued a joint statement addressing the clinical use of prenatal diagnostic genome wide sequencing, including exome sequencing.
Continue to: Guidance at a glance...
Guidance at a glance
The societies' recommendations are summarized as follows:
- Exome sequencing is best done as a trio analysis, with fetal and both parental samples sequenced and analyzed together.
- Extensive pretest education, counseling, and informed consent, as well as posttest counseling, are essential. This should include:
—the types of results to be conveyed (variants that are pathogenic, likely pathogenic, of uncertain significance, likely benign, and benign)
—the possibility that results will not be obtained or may not be available before the birth of the fetus
—realistic expectations regarding the likelihood that a significant result will be obtained
—the timeframe to results
—the option to include or exclude in the results incidental or secondary findings (such as an unexpected childhood disorder, cancer susceptibility genes, adult-onset disorders)
—the possibility of uncovering nonpaternity or consanguinity
—the potential reanalysis of results over time
—how data are stored, who has access, and for what purpose.
- Fetal sequencing may be beneficial in the following scenarios:
—multiple fetal anomalies or a single major anomaly suggestive of a genetic disorder, when the microarray is negative
—no microarray result is available, but the fetus exhibits a pattern of anomalies strongly suggestive of a single-gene disorder
—a prior undiagnosed fetus (or child) with anomalies suggestive of a genetic etiology, and with similar anomalies in the current pregnancy, with normal karyotype or microarray. Providers also can consider sequencing samples from both parents prior to preimplantation genetic testing to check for shared carrier status for autosomal recessive mutations, although obtaining exome sequencing from the prior affected fetus (or child) is ideal.
—history of recurrent stillbirths of unknown etiology, with a recurrent pattern of anomalies in the current pregnancy, with normal karyotype or microarray.
- Interpretation of results should be done using a multidisciplinary team-based approach, including clinical scientists, geneticists, genetic counselors, and experts in prenatal diagnosis.
- Where possible and after informed consent, reanalysis of results should be undertaken if a future pregnancy is planned or ongoing, and a significant amount of time has elapsed since the time the result was last reported.
- Parents should be given a written report of test results.
Three professional societies have convened to issue consensus opinion that includes current indications for prenatal exome sequencing and important factors to include in the consent process. We follow these guidelines in our own practice.
Summary
Exome sequencing is increasingly becoming mainstream in postnatal genetic testing, and it is emerging as the newest diagnostic frontier in prenatal genetic testing. However, there are limitations to prenatal exome sequencing, including issues with consent at a vulnerable time for parents, limited information available regarding the phenotype, and results that may not be available before the birth of a fetus. Providers should be familiar with the indications for testing, the possible results, the limitations of prenatal phenotyping, and the implications for future pregnancies.
Prenatal diagnosis of genetic anomalies is important for diagnosing lethal genetic conditions before birth. It can provide information for parents regarding pregnancy options and allow for recurrence risk counseling and the potential use of preimplantation genetic testing in the next pregnancy. For decades, a karyotype was used to analyze amniocentesis and chorionic villus sampling specimens; in recent years, chromosomal microarray analysis provides more information about significant chromosomal abnormalities, including microdeletions and microduplications. However, microarrays also have limitations, as they do not identify base pair changes associated with single-gene disorders.
The advent of next-generation sequencing has substantially reduced the cost of DNA sequencing. Whole genome sequencing (WGS) can sequence the entire genome— both the coding (exonic) and noncoding (intronic) regions—while exome sequencing analyzes only the protein-coding exons, which make up 1% to 2% of the genome and about 85% of the protein-coding genes associated with known human disease. Exome sequencing increasingly is used in cases of suspected genetic disorders when other tests have been unrevealing.
In this Update, we review recent reports of prenatal exome sequencing, including studies exploring the yield in fetuses with structural anomalies; the importance of prenatal phenotyping; the perspectives of parents and health care professionals who were involved in prenatal exome sequencing studies; and a summary of a joint position statement from 3 societies regarding prenatal sequencing.
Prenatal whole exome sequencing has potential utility, with some limitations
Petrovski S, Aggarwal V, Giordano JL, et al. Whole-exome sequencing in the evaluation of fetal structural anomalies: a prospective cohort study. Lancet. 2019;393:758-767.
Lord J, McMullan DJ, Eberhardt RY, et al; for the Prenatal Assessment of Genomes and Exomes Consortium. Prenatal exome sequencing analysis in fetal structural anomalies detected by ultrasonography (PAGE): a cohort study. Lancet. 2019;393:747-757.
Exome sequencing has been shown to identify an underlying genetic cause in 25% to 30% of children with an undiagnosed suspected genetic disorder. Two studies recently published in the Lancet sought to determine the incremental diagnostic yield of prenatal whole exome sequencing (WES) in the setting of fetal structural anomalies when karyotype and microarray results were normal.
Continue to: Details of the studies...
Details of the studies
In a prospective cohort study by Petrovski and colleagues, DNA samples from 234 fetuses with a structural anomaly (identified on ultrasonography) and both parents (parent-fetus "trios") were used for analysis. WES identified diagnostic genetic variants in 24 trios (10%). An additional 46 (20%) had variants that indicated pathogenicity but without sufficient evidence to be considered diagnostic.
The anomalies with the highest frequency of a genetic diagnosis were lymphatic, 24%; skeletal, 24%; central nervous system, 22%; and renal, 16%; while cardiac anomalies had the lowest yield at 5%.
In another prospective cohort study, known as the Prenatal Assessment of Genomes and Exomes (PAGE), Lord and colleagues sequenced DNA samples from 610 parent-fetus trios, but they restricted sequencing to a predefined list of 1,628 genes. Diagnostic genetic variants were identified in 52 fetuses (8.5%), while 24 (3.9%) had a variant of uncertain significance that was thought to be of potential clinical usefulness.
Fetuses with multiple anomalies had the highest genetic yield (15.4%), followed by skeletal (15.4%) and cardiac anomalies (11.1%), with the lowest yield in fetuses with isolated increased nuchal translucency (3.2%).
Diagnostic yield is high, but prenatal utility is limited
Both studies showed a clinically significant diagnostic yield of 8% to 10% for prenatal exome sequencing in cases of fetal structural anomalies with normal karyotype and microarray testing. While this yield demonstrates the utility of prenatal exome sequencing, it is significantly lower than what has been reported in postnatal studies. One of the reasons for this is the inherent limitation of prenatal phenotyping (discussed below).
The cohort studies by both Petrovski and Lord and their colleagues show the feasibility and potential diagnostic utility of exome sequencing in cases of fetal structural anomalies where karyotype and microarray are not diagnostic. However, the lower yield found in these studies compared with those in postnatal studies highlights in part the limitations of prenatal phenotyping.
The importance of prenatal phenotyping
Aarabi M, Sniezek O, Jiang H, et al. Importance of complete phenotyping in prenatal whole exome sequencing. Hum Genet. 2018;137:175-181.
In postnatal exome sequencing, the physical exam, imaging findings, and laboratory results are components of the phenotype that are used to interpret the sequencing data. Prenatal phenotyping, however, is limited to the use of fetal ultrasonography and, occasionally, the addition of magnetic resonance imaging. Prenatal phenotyping is without the benefit of an exam to detect more subtle anomalies or functional status, such as developmental delay, seizures, or failure to thrive.
When a structural anomaly is identified on prenatal ultrasonography, it is especially important that detailed imaging be undertaken to detect other anomalies, including more subtle facial features and dysmorphology.
Value of reanalyzing exome sequencing data
Aarabi and colleagues conducted a retrospective study of 20 fetuses with structural anomalies and normal karyotype and microarray. They performed trio exome sequencing first using information available only prenatally and then conducted a reanalysis using information available after delivery.
With prenatal phenotyping only, the investigators identified no pathogenic, or likely pathogenic, variants. On reanalysis of combined prenatal and postnatal findings, however, they identified pathogenic variants in 20% of cases.
Significance of the findings
This study highlights both the importance of a careful, detailed fetal ultrasonography study and the possible additional benefit of a postnatal examination (such as an autopsy) in order to yield improved results. In addition, the authors noted that the development of a prenatal phenotype-genotype database would significantly help exome sequencing interpretation in the prenatal setting.
Careful prenatal ultrasonography is crucial to help in the interpretation of prenatal exome sequencing. Patients who have undergone prenatal clinical exome sequencing may benefit from reanalysis of the genetic data based on detailed postnatal findings.
Social impact of WES: Parent and provider perspectives
Wou K, Weitz T, McCormack C, et al. Parental perceptions of prenatal whole exome sequencing (PPPWES) study. Prenat Diagn. 2018;38:801-811.
Horn R, Parker M. Health professionals' and researchers' perspectives on prenatal whole genome and exome sequencing: 'We can't shut the door now, the genie's out, we need to refine it.' PLoS One. 2018;13:e0204158.
As health care providers enter a new era of prenatal genetic testing with exome sequencing, it is crucial to the path forward that we obtain perspectives from the parents and providers who participated in these studies. Notably, in both of the previously discussed Lancet reports, the authors interviewed the participants to discuss the challenges involved and identify strategies for improving future testing.
Continue to: What parents want...
What parents want
To ascertain the perceptions of couples who underwent prenatal WES, Wou and colleagues conducted semi-structured interviews with participants from the Fetal Sequencing Study regarding their experience. They interviewed 29 parents from 17 pregnancies, including a mix of those who had pathogenic prenatal results, terminated prior to receiving the results, and had normal results.
Expressed feelings and desires. Parents recalled feelings of anxiety and stress around the time of diagnosis and the need for help with coping while awaiting results. The majority of parents reported that they would like to be told about uncertain results, but that desire decreased as the certainty of results decreased.
Parents were overall satisfied with the prenatal genetic testing experience, but they added that they would have liked to receive written materials beforehand and a written report of the test results (including negative cases). They also would like to have connected with other families with similar experiences, to have received results sooner, and to have an in-person meeting after telephone disclosure of the results.
Health professionals articulate complexity of prenatal genomics
In a qualitative interview study to explore critical issues involved in the clinical practice use of prenatal genomics, Horn and Parker conducted interviews with 20 health care professionals who were involved in the previously described PAGE trial. Patient recruiters, midwives, genetic counselors, research assistants, and laboratory staff were included.
Interviewees cited numerous challenges involved in their day-to-day work with prenatal whole genome and exome sequencing, including:
- the complexity of achieving valid parental consent at a time of vulnerability
- management of parent expectations
- transmitting and comprehending complex information
- the usefulness of information
- the difficulty of a long turnaround time for study results.
All the interviewees agreed that prenatal exome sequencing studies contribute to knowledge generation and the advancement of technology.
The authors concluded that an appropriate next step would be the development of appropriate guidelines for good ethical practice that address the concerns encountered in genomics clinical practice.
The prenatal experience can be overwhelming for parents. Pretest and posttest counseling on genetic testing and results are of the utmost importance, as is finding ways to help support parents through this anxious time.
Societies offer guidance on using genome and exome sequencing
International Society for Prenatal Diagnosis, Society for Maternal and Fetal Medicine, Perinatal Quality Foundation. Joint Position Statement from the International Society for Prenatal Diagnosis (ISPD), the Society for Maternal Fetal Medicine (SMFM), and the Perinatal Quality Foundation (PQF) on the use of genome-wide sequencing for fetal diagnosis. Prenat Diagn. 2018;38:6-9.
In response to the rapid integration of exome sequencing for genetic diagnosis, several professional societies—the International Society for Prenatal Diagnosis, Society for Maternal Fetal Medicine, and Perinatal Quality Foundation—issued a joint statement addressing the clinical use of prenatal diagnostic genome wide sequencing, including exome sequencing.
Continue to: Guidance at a glance...
Guidance at a glance
The societies' recommendations are summarized as follows:
- Exome sequencing is best done as a trio analysis, with fetal and both parental samples sequenced and analyzed together.
- Extensive pretest education, counseling, and informed consent, as well as posttest counseling, are essential. This should include:
—the types of results to be conveyed (variants that are pathogenic, likely pathogenic, of uncertain significance, likely benign, and benign)
—the possibility that results will not be obtained or may not be available before the birth of the fetus
—realistic expectations regarding the likelihood that a significant result will be obtained
—the timeframe to results
—the option to include or exclude in the results incidental or secondary findings (such as an unexpected childhood disorder, cancer susceptibility genes, adult-onset disorders)
—the possibility of uncovering nonpaternity or consanguinity
—the potential reanalysis of results over time
—how data are stored, who has access, and for what purpose.
- Fetal sequencing may be beneficial in the following scenarios:
—multiple fetal anomalies or a single major anomaly suggestive of a genetic disorder, when the microarray is negative
—no microarray result is available, but the fetus exhibits a pattern of anomalies strongly suggestive of a single-gene disorder
—a prior undiagnosed fetus (or child) with anomalies suggestive of a genetic etiology, and with similar anomalies in the current pregnancy, with normal karyotype or microarray. Providers also can consider sequencing samples from both parents prior to preimplantation genetic testing to check for shared carrier status for autosomal recessive mutations, although obtaining exome sequencing from the prior affected fetus (or child) is ideal.
—history of recurrent stillbirths of unknown etiology, with a recurrent pattern of anomalies in the current pregnancy, with normal karyotype or microarray.
- Interpretation of results should be done using a multidisciplinary team-based approach, including clinical scientists, geneticists, genetic counselors, and experts in prenatal diagnosis.
- Where possible and after informed consent, reanalysis of results should be undertaken if a future pregnancy is planned or ongoing, and a significant amount of time has elapsed since the time the result was last reported.
- Parents should be given a written report of test results.
Three professional societies have convened to issue consensus opinion that includes current indications for prenatal exome sequencing and important factors to include in the consent process. We follow these guidelines in our own practice.
Summary
Exome sequencing is increasingly becoming mainstream in postnatal genetic testing, and it is emerging as the newest diagnostic frontier in prenatal genetic testing. However, there are limitations to prenatal exome sequencing, including issues with consent at a vulnerable time for parents, limited information available regarding the phenotype, and results that may not be available before the birth of a fetus. Providers should be familiar with the indications for testing, the possible results, the limitations of prenatal phenotyping, and the implications for future pregnancies.