User login
Migraine: Helping patients through a difficult journey
Most clinicians who treat migraine know the statistics associated with this debilitating condition and can recite them almost verbatim. Likewise, we all know the long journey a patient experiencing headache can take before finding relief.
The question is, what can we do about it? It can sometimes feel easy to look at the statistics, accept them with little objection, and move on, but we must do the best we can for our patients in the face of the migraine challenge.
It’s a journey that I welcome you to take with me as we evaluate important migraine trends, treatments, and controversies, and figure out how to leverage these developments to improve outcomes and help our patients improve and feel and function better.
There are upwards of 39 million migraine sufferers in the US who are on a journey of their own that is often perplexing, frustrating, and can feel fruitless. According to the Migraine Research Foundation, most migraine sufferers do not seek medical treatment. More than half of patients who experience migraine are never diagnosed. Moreover, only 4% receive care from headache and pain specialists. Perhaps most disheartening, an estimated 5 million migraine sufferers, who we believe can benefit from preventive treatment, are not receiving it.
The patient journey can be extremely discouraging and often maddening. Results from the American Migraine Study show that nearly four in every 10 migraine patients suffer 3 years or more before being diagnosed. A cross-sectional study published in 2019 analyzed treatments, procedures, and follow-up approaches experienced by 456 migraine sufferers until their initial consult with a headache specialist. Patients reported an average headache frequency of approximately 16 days per month. More than half were found to have chronic migraine, and 3 in every 10 had migraine with aura. Despite these characteristics—which were apparently hiding in plain sight—it took patients in this study an average of about 17 years from pain onset to make the journey to an appointment with a headache specialist. That is hard to believe, let alone to understand. Along the way, many migraineurs—particularly those with chronic migraine—were subjected to unnecessary exams and treatments.
Results like these do not come cheaply for families, society, and the health care system. Migraine is estimated to cost more than $20 million per year in direct medical expenses and lost productivity in the US, according to the American Migraine Foundation. Others have estimated double that number. Sufferers, meanwhile, face the prospect of significant pain, stigma and ongoing disability. More than 8 in every 10participants in the American Migraine Study had at least some headache-related disability. More than half say their pain caused severe impairment, even requiring bed rest.
I believe we can help our migraine patients along this journey—and we can make it less arduous for them. We have education, tools, and treatments to help them. Learn how by joining me here each month. We will address the practical relevance of topics such as acute and preventive care (including the new gepants and CGRP-targeted treatments), new and more effective treatments for medication overuse headache, new treatment devices, behavioral approaches to migraine, and our role in headache advocacy, including stigma avoidance. We will not shy away from controversial topics such as the changing definition of chronic migraine, monoclonal antibody safety , high and low cerebrospinal fluid pressure syndromes, and more. See you next month.
Most clinicians who treat migraine know the statistics associated with this debilitating condition and can recite them almost verbatim. Likewise, we all know the long journey a patient experiencing headache can take before finding relief.
The question is, what can we do about it? It can sometimes feel easy to look at the statistics, accept them with little objection, and move on, but we must do the best we can for our patients in the face of the migraine challenge.
It’s a journey that I welcome you to take with me as we evaluate important migraine trends, treatments, and controversies, and figure out how to leverage these developments to improve outcomes and help our patients improve and feel and function better.
There are upwards of 39 million migraine sufferers in the US who are on a journey of their own that is often perplexing, frustrating, and can feel fruitless. According to the Migraine Research Foundation, most migraine sufferers do not seek medical treatment. More than half of patients who experience migraine are never diagnosed. Moreover, only 4% receive care from headache and pain specialists. Perhaps most disheartening, an estimated 5 million migraine sufferers, who we believe can benefit from preventive treatment, are not receiving it.
The patient journey can be extremely discouraging and often maddening. Results from the American Migraine Study show that nearly four in every 10 migraine patients suffer 3 years or more before being diagnosed. A cross-sectional study published in 2019 analyzed treatments, procedures, and follow-up approaches experienced by 456 migraine sufferers until their initial consult with a headache specialist. Patients reported an average headache frequency of approximately 16 days per month. More than half were found to have chronic migraine, and 3 in every 10 had migraine with aura. Despite these characteristics—which were apparently hiding in plain sight—it took patients in this study an average of about 17 years from pain onset to make the journey to an appointment with a headache specialist. That is hard to believe, let alone to understand. Along the way, many migraineurs—particularly those with chronic migraine—were subjected to unnecessary exams and treatments.
Results like these do not come cheaply for families, society, and the health care system. Migraine is estimated to cost more than $20 million per year in direct medical expenses and lost productivity in the US, according to the American Migraine Foundation. Others have estimated double that number. Sufferers, meanwhile, face the prospect of significant pain, stigma and ongoing disability. More than 8 in every 10participants in the American Migraine Study had at least some headache-related disability. More than half say their pain caused severe impairment, even requiring bed rest.
I believe we can help our migraine patients along this journey—and we can make it less arduous for them. We have education, tools, and treatments to help them. Learn how by joining me here each month. We will address the practical relevance of topics such as acute and preventive care (including the new gepants and CGRP-targeted treatments), new and more effective treatments for medication overuse headache, new treatment devices, behavioral approaches to migraine, and our role in headache advocacy, including stigma avoidance. We will not shy away from controversial topics such as the changing definition of chronic migraine, monoclonal antibody safety , high and low cerebrospinal fluid pressure syndromes, and more. See you next month.
Most clinicians who treat migraine know the statistics associated with this debilitating condition and can recite them almost verbatim. Likewise, we all know the long journey a patient experiencing headache can take before finding relief.
The question is, what can we do about it? It can sometimes feel easy to look at the statistics, accept them with little objection, and move on, but we must do the best we can for our patients in the face of the migraine challenge.
It’s a journey that I welcome you to take with me as we evaluate important migraine trends, treatments, and controversies, and figure out how to leverage these developments to improve outcomes and help our patients improve and feel and function better.
There are upwards of 39 million migraine sufferers in the US who are on a journey of their own that is often perplexing, frustrating, and can feel fruitless. According to the Migraine Research Foundation, most migraine sufferers do not seek medical treatment. More than half of patients who experience migraine are never diagnosed. Moreover, only 4% receive care from headache and pain specialists. Perhaps most disheartening, an estimated 5 million migraine sufferers, who we believe can benefit from preventive treatment, are not receiving it.
The patient journey can be extremely discouraging and often maddening. Results from the American Migraine Study show that nearly four in every 10 migraine patients suffer 3 years or more before being diagnosed. A cross-sectional study published in 2019 analyzed treatments, procedures, and follow-up approaches experienced by 456 migraine sufferers until their initial consult with a headache specialist. Patients reported an average headache frequency of approximately 16 days per month. More than half were found to have chronic migraine, and 3 in every 10 had migraine with aura. Despite these characteristics—which were apparently hiding in plain sight—it took patients in this study an average of about 17 years from pain onset to make the journey to an appointment with a headache specialist. That is hard to believe, let alone to understand. Along the way, many migraineurs—particularly those with chronic migraine—were subjected to unnecessary exams and treatments.
Results like these do not come cheaply for families, society, and the health care system. Migraine is estimated to cost more than $20 million per year in direct medical expenses and lost productivity in the US, according to the American Migraine Foundation. Others have estimated double that number. Sufferers, meanwhile, face the prospect of significant pain, stigma and ongoing disability. More than 8 in every 10participants in the American Migraine Study had at least some headache-related disability. More than half say their pain caused severe impairment, even requiring bed rest.
I believe we can help our migraine patients along this journey—and we can make it less arduous for them. We have education, tools, and treatments to help them. Learn how by joining me here each month. We will address the practical relevance of topics such as acute and preventive care (including the new gepants and CGRP-targeted treatments), new and more effective treatments for medication overuse headache, new treatment devices, behavioral approaches to migraine, and our role in headache advocacy, including stigma avoidance. We will not shy away from controversial topics such as the changing definition of chronic migraine, monoclonal antibody safety , high and low cerebrospinal fluid pressure syndromes, and more. See you next month.
Physicians react: Doctors worry about patients reading their clinical notes
Patients will soon be able to read the notes that physicians make during an episode of care, as well as information about diagnostic testing and imaging results, tests for STDs, fetal ultrasounds, and cancer biopsies. This open access is raising concerns among physicians.
As part of the 21st Century Cures Act, patients have the right to see their medical notes. Known as Open Notes, the policy will go into effect on April 5, 2021. The Department of Health & Human Services recently changed the original start date, which was to be Nov. 2, 2020.
The mandate has some physicians worrying about potential legal risks and possible violation of doctor-patient confidentiality. When asked to share their views on the new Open Notes mandate, many physicians expressed their concerns but also cited some of the positive effects that could come from this.
Potentially more legal woes for physicians?
A key concern raised by one physician commenter is that patients could misunderstand legitimate medical terminology or even put a physician in legal crosshairs. For example, a medical term such as “spontaneous abortion” could be misconstrued by patients. A physician might write notes with the idea that a patient is reading them and thus might alter those notes in a way that creates legal trouble.
“This layers another level of censorship and legal liability onto physicians, who in attempting to be [politically correct], may omit critical information or have to use euphemisms in order to avoid conflict,” one physician said.
She also questioned whether notes might now have to be run through legal counsel before being posted to avoid potential liability.
Another doctor questioned how physicians would be able to document patients suspected of faking injuries for pain medication, for example. Could such documentation lead to lawsuits for the doctor?
As one physician noted, some patients “are drug seekers. Some refuse to aid in their own care. Some are malingerers. Not documenting that is bad medicine.”
The possibility of violating doctor-patient confidentiality laws, particularly for teenagers, could be another negative effect of Open Notes, said one physician.
“Won’t this violate the statutes that teenagers have the right to confidential evaluations?” the commenter mused. “If charts are to be immediately available, then STDs and pregnancies they weren’t ready to talk about will now be suddenly known by their parents.”
One doctor has already faced this issue. “I already ran into this problem once,” he noted. “Now I warn those on their parents’ insurance before I start the visit. I have literally had a patient state, ‘well then we are done,’ and leave without being seen due to it.”
Another physician questioned the possibility of having to write notes differently than they do now, especially if the patients have lower reading comprehension abilities.
One physician who uses Open Notes said he receives patient requests for changes that have little to do with the actual diagnosis and relate to ancillary issues. He highlighted patients who “don’t want psych diagnosis in their chart or are concerned a diagnosis will raise their insurance premium, so they ask me to delete it.”
Will Open Notes erode patient communication?
One physician questioned whether it would lead to patients being less open and forthcoming about their medical concerns with doctors.
“The main problem I see is the patient not telling me the whole story, or worse, telling me the story, and then asking me not to document it (as many have done in the past) because they don’t want their spouse, family, etc. to read the notes and they have already given their permission for them to do so, for a variety of reasons,” he commented. “This includes topics of STDs, infidelity, depression, suicidal thoughts, and other symptoms the patient doesn’t want their family to read about.”
Some physicians envision positive developments
Many physicians are unconcerned by the new mandate. “I see some potential good in this, such as improving doctor-patient communication and more scrupulous charting,” one physician said.
A doctor working in the U.S. federal health care system noted that open access has been a part of that system for decades.
“Since health care providers work in this unveiled setting for their entire career, they usually know how to write appropriate clinical notes and what information needs to be included in them,” he wrote. “Now it’s time for the rest of the medical community to catch up to a reality that we have worked within for decades now.
“The world did not end, malpractice complaints did not increase, and physician/patient relationships were not damaged. Living in the information age, archaic practices like private notes were surely going to end at some point.”
One doctor who has been using Open Notes has had experiences in which the patient noted an error in the medical chart that needed correcting. “I have had one patient correct me on a timeline in the HPI which was helpful and I made the requested correction in that instance,” he said.
Another physician agreed. “I’ve had patients add or correct valuable information I’ve missed. Good probably outweighs the bad if we set limits on behaviors expressed by the personality disordered group. The majority of people don’t seem to care and still ask me ‘what would you do’ or ‘tell me what to do.’ It’s all about patient/physician trust.”
Another talked about how Open Notes should have little or no impact. “Here’s a novel concept – talking to our patients,” he commented. “There is nothing in every one of my chart notes that has not already been discussed with my patients and I dictate (speech to text) my findings and plan in front of them. So, if they are reviewing my office notes, it will only serve to reinforce what we have already discussed.”
“I don’t intend to change anything,” he added. “Chances are if they were to see a test result before I have a chance to discuss it with them, they will have already ‘Googled’ its meaning and we can have more meaningful interaction if they have a basic understanding of the test.”
“I understand that this is anxiety provoking, but in general I think it is appropriate for patients to have access to their notes,” said another physician. “If physicians write lousy notes that say they did things they didn’t do, that fail to actually state a diagnosis and a plan (and they often do), that is the doc’s problem, not the patient’s.”
A version of this article first appeared on Medscape.com.
Patients will soon be able to read the notes that physicians make during an episode of care, as well as information about diagnostic testing and imaging results, tests for STDs, fetal ultrasounds, and cancer biopsies. This open access is raising concerns among physicians.
As part of the 21st Century Cures Act, patients have the right to see their medical notes. Known as Open Notes, the policy will go into effect on April 5, 2021. The Department of Health & Human Services recently changed the original start date, which was to be Nov. 2, 2020.
The mandate has some physicians worrying about potential legal risks and possible violation of doctor-patient confidentiality. When asked to share their views on the new Open Notes mandate, many physicians expressed their concerns but also cited some of the positive effects that could come from this.
Potentially more legal woes for physicians?
A key concern raised by one physician commenter is that patients could misunderstand legitimate medical terminology or even put a physician in legal crosshairs. For example, a medical term such as “spontaneous abortion” could be misconstrued by patients. A physician might write notes with the idea that a patient is reading them and thus might alter those notes in a way that creates legal trouble.
“This layers another level of censorship and legal liability onto physicians, who in attempting to be [politically correct], may omit critical information or have to use euphemisms in order to avoid conflict,” one physician said.
She also questioned whether notes might now have to be run through legal counsel before being posted to avoid potential liability.
Another doctor questioned how physicians would be able to document patients suspected of faking injuries for pain medication, for example. Could such documentation lead to lawsuits for the doctor?
As one physician noted, some patients “are drug seekers. Some refuse to aid in their own care. Some are malingerers. Not documenting that is bad medicine.”
The possibility of violating doctor-patient confidentiality laws, particularly for teenagers, could be another negative effect of Open Notes, said one physician.
“Won’t this violate the statutes that teenagers have the right to confidential evaluations?” the commenter mused. “If charts are to be immediately available, then STDs and pregnancies they weren’t ready to talk about will now be suddenly known by their parents.”
One doctor has already faced this issue. “I already ran into this problem once,” he noted. “Now I warn those on their parents’ insurance before I start the visit. I have literally had a patient state, ‘well then we are done,’ and leave without being seen due to it.”
Another physician questioned the possibility of having to write notes differently than they do now, especially if the patients have lower reading comprehension abilities.
One physician who uses Open Notes said he receives patient requests for changes that have little to do with the actual diagnosis and relate to ancillary issues. He highlighted patients who “don’t want psych diagnosis in their chart or are concerned a diagnosis will raise their insurance premium, so they ask me to delete it.”
Will Open Notes erode patient communication?
One physician questioned whether it would lead to patients being less open and forthcoming about their medical concerns with doctors.
“The main problem I see is the patient not telling me the whole story, or worse, telling me the story, and then asking me not to document it (as many have done in the past) because they don’t want their spouse, family, etc. to read the notes and they have already given their permission for them to do so, for a variety of reasons,” he commented. “This includes topics of STDs, infidelity, depression, suicidal thoughts, and other symptoms the patient doesn’t want their family to read about.”
Some physicians envision positive developments
Many physicians are unconcerned by the new mandate. “I see some potential good in this, such as improving doctor-patient communication and more scrupulous charting,” one physician said.
A doctor working in the U.S. federal health care system noted that open access has been a part of that system for decades.
“Since health care providers work in this unveiled setting for their entire career, they usually know how to write appropriate clinical notes and what information needs to be included in them,” he wrote. “Now it’s time for the rest of the medical community to catch up to a reality that we have worked within for decades now.
“The world did not end, malpractice complaints did not increase, and physician/patient relationships were not damaged. Living in the information age, archaic practices like private notes were surely going to end at some point.”
One doctor who has been using Open Notes has had experiences in which the patient noted an error in the medical chart that needed correcting. “I have had one patient correct me on a timeline in the HPI which was helpful and I made the requested correction in that instance,” he said.
Another physician agreed. “I’ve had patients add or correct valuable information I’ve missed. Good probably outweighs the bad if we set limits on behaviors expressed by the personality disordered group. The majority of people don’t seem to care and still ask me ‘what would you do’ or ‘tell me what to do.’ It’s all about patient/physician trust.”
Another talked about how Open Notes should have little or no impact. “Here’s a novel concept – talking to our patients,” he commented. “There is nothing in every one of my chart notes that has not already been discussed with my patients and I dictate (speech to text) my findings and plan in front of them. So, if they are reviewing my office notes, it will only serve to reinforce what we have already discussed.”
“I don’t intend to change anything,” he added. “Chances are if they were to see a test result before I have a chance to discuss it with them, they will have already ‘Googled’ its meaning and we can have more meaningful interaction if they have a basic understanding of the test.”
“I understand that this is anxiety provoking, but in general I think it is appropriate for patients to have access to their notes,” said another physician. “If physicians write lousy notes that say they did things they didn’t do, that fail to actually state a diagnosis and a plan (and they often do), that is the doc’s problem, not the patient’s.”
A version of this article first appeared on Medscape.com.
Patients will soon be able to read the notes that physicians make during an episode of care, as well as information about diagnostic testing and imaging results, tests for STDs, fetal ultrasounds, and cancer biopsies. This open access is raising concerns among physicians.
As part of the 21st Century Cures Act, patients have the right to see their medical notes. Known as Open Notes, the policy will go into effect on April 5, 2021. The Department of Health & Human Services recently changed the original start date, which was to be Nov. 2, 2020.
The mandate has some physicians worrying about potential legal risks and possible violation of doctor-patient confidentiality. When asked to share their views on the new Open Notes mandate, many physicians expressed their concerns but also cited some of the positive effects that could come from this.
Potentially more legal woes for physicians?
A key concern raised by one physician commenter is that patients could misunderstand legitimate medical terminology or even put a physician in legal crosshairs. For example, a medical term such as “spontaneous abortion” could be misconstrued by patients. A physician might write notes with the idea that a patient is reading them and thus might alter those notes in a way that creates legal trouble.
“This layers another level of censorship and legal liability onto physicians, who in attempting to be [politically correct], may omit critical information or have to use euphemisms in order to avoid conflict,” one physician said.
She also questioned whether notes might now have to be run through legal counsel before being posted to avoid potential liability.
Another doctor questioned how physicians would be able to document patients suspected of faking injuries for pain medication, for example. Could such documentation lead to lawsuits for the doctor?
As one physician noted, some patients “are drug seekers. Some refuse to aid in their own care. Some are malingerers. Not documenting that is bad medicine.”
The possibility of violating doctor-patient confidentiality laws, particularly for teenagers, could be another negative effect of Open Notes, said one physician.
“Won’t this violate the statutes that teenagers have the right to confidential evaluations?” the commenter mused. “If charts are to be immediately available, then STDs and pregnancies they weren’t ready to talk about will now be suddenly known by their parents.”
One doctor has already faced this issue. “I already ran into this problem once,” he noted. “Now I warn those on their parents’ insurance before I start the visit. I have literally had a patient state, ‘well then we are done,’ and leave without being seen due to it.”
Another physician questioned the possibility of having to write notes differently than they do now, especially if the patients have lower reading comprehension abilities.
One physician who uses Open Notes said he receives patient requests for changes that have little to do with the actual diagnosis and relate to ancillary issues. He highlighted patients who “don’t want psych diagnosis in their chart or are concerned a diagnosis will raise their insurance premium, so they ask me to delete it.”
Will Open Notes erode patient communication?
One physician questioned whether it would lead to patients being less open and forthcoming about their medical concerns with doctors.
“The main problem I see is the patient not telling me the whole story, or worse, telling me the story, and then asking me not to document it (as many have done in the past) because they don’t want their spouse, family, etc. to read the notes and they have already given their permission for them to do so, for a variety of reasons,” he commented. “This includes topics of STDs, infidelity, depression, suicidal thoughts, and other symptoms the patient doesn’t want their family to read about.”
Some physicians envision positive developments
Many physicians are unconcerned by the new mandate. “I see some potential good in this, such as improving doctor-patient communication and more scrupulous charting,” one physician said.
A doctor working in the U.S. federal health care system noted that open access has been a part of that system for decades.
“Since health care providers work in this unveiled setting for their entire career, they usually know how to write appropriate clinical notes and what information needs to be included in them,” he wrote. “Now it’s time for the rest of the medical community to catch up to a reality that we have worked within for decades now.
“The world did not end, malpractice complaints did not increase, and physician/patient relationships were not damaged. Living in the information age, archaic practices like private notes were surely going to end at some point.”
One doctor who has been using Open Notes has had experiences in which the patient noted an error in the medical chart that needed correcting. “I have had one patient correct me on a timeline in the HPI which was helpful and I made the requested correction in that instance,” he said.
Another physician agreed. “I’ve had patients add or correct valuable information I’ve missed. Good probably outweighs the bad if we set limits on behaviors expressed by the personality disordered group. The majority of people don’t seem to care and still ask me ‘what would you do’ or ‘tell me what to do.’ It’s all about patient/physician trust.”
Another talked about how Open Notes should have little or no impact. “Here’s a novel concept – talking to our patients,” he commented. “There is nothing in every one of my chart notes that has not already been discussed with my patients and I dictate (speech to text) my findings and plan in front of them. So, if they are reviewing my office notes, it will only serve to reinforce what we have already discussed.”
“I don’t intend to change anything,” he added. “Chances are if they were to see a test result before I have a chance to discuss it with them, they will have already ‘Googled’ its meaning and we can have more meaningful interaction if they have a basic understanding of the test.”
“I understand that this is anxiety provoking, but in general I think it is appropriate for patients to have access to their notes,” said another physician. “If physicians write lousy notes that say they did things they didn’t do, that fail to actually state a diagnosis and a plan (and they often do), that is the doc’s problem, not the patient’s.”
A version of this article first appeared on Medscape.com.
Sequential Targeted Treatment for a Geriatric Patient with Acute Myeloid Leukemia with Concurrent FLT3-TKD and IDH1 Mutations
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
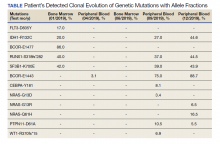
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
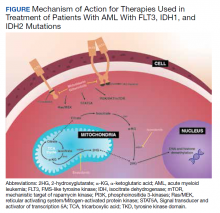
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
Which imaging criteria identify progressive forms of MS?
The role of imaging in diagnosing progressive multiple sclerosis (MS) and in assessing prognosis is the subject of a new review.
MRI is central in the diagnostic work-up of patients suspected of having MS, given its high sensitivity in detecting disease dissemination in space and over time and its notable ability to exclude mimics of MS, the authors noted. However, diagnosis of primary progressive MS remains challenging and is only possible retrospectively on the basis of clinical assessment.
they wrote.
Diagnosis of progressive MS is limited by difficulties in distinguishing accumulating disability caused by inflammatory disease activity from that attributable to degenerative processes associated with secondary progressive MS. Moreover, there are no accepted clinical criteria for diagnosing secondary progressive MS, the authors explained.
This need has promoted extensive research in the field of imaging, facilitated by definition of novel MRI sequences, to identify imaging features reflecting pathophysiological mechanisms relevant to the pathobiology of progressive MS, the authors said.
The current review reports the conclusions of a workshop held in Milan in November 2019, at which an expert panel of neurologists and neuroradiologists addressed the role of MRI in progressive MS.
Massimo Filippi, MD, IRCCS San Raffaele Scientific Institute, Milan, was the lead author of the review, which was published online Dec. 14, 2020, in JAMA Neurology.
The authors concluded that no definitive, qualitative clinical, immunologic, histopathologic, or neuroimaging features differentiate primary progressive and secondary progressive forms of MS; both are characterized by neurodegenerative phenomena and a gradual and irreversible accumulation of clinical disability, which is also affected by aging and comorbidities.
A definitive diagnosis of primary progressive MS is more difficult than a diagnosis of relapsing remitting MS; in part, primary progressive MS is a diagnosis of exclusion because it can be mimicked by other conditions clinically and radiologically, the authors noted.
The writers did report that, although nonspecific, some spinal cord imaging features are typical of primary progressive MS. These include diffuse abnormalities and lesions involving gray matter and two or more white-matter columns, but confirmation of this is required.
In patients with primary progressive MS and those with relapse-onset MS, MRI features at disease onset predict long-term disability and a progressive disease course. These features include lesions in critical central nervous system regions (i.e., spinal cord, infratentorial regions, and gray matter) and high inflammatory activity in the first years after disease onset. These measures are evaluable in clinical practice, the authors said.
In patients with established MS, gray-matter involvement and neurodegeneration are associated with accelerated clinical worsening; however, detection validation and standardization need to be implemented at the individual patient level, they commented.
Novel candidate imaging biomarkers, such as subpial demyelination, and the presence of slowly expanding lesions or paramagnetic rim lesions may identify progressive MS but should be further investigated, they added.
Discovery of MRI markers capable of detecting evolution from relapsing-remitting to secondary progressive MS remains an unmet need that will probably require multiparametric MRI studies, because it is unlikely that a single MRI method will be able to allow clinicians to optimally distinguish among these stages, the authors said.
The contribution of these promising MRI measures combined with other biomarkers, such as quantification of serum neurofilament light chain levels or optical coherence tomography assessment, should be explored to improve the identification of patients with progressive MS, they concluded.
‘A comprehensive review’
In a comment, Jeffrey A. Cohen, MD, director of the Cleveland Clinic’s Mellen Center for MS Treatment and Research, said the article is a comprehensive review of the pathologic mechanisms that underlie progression in MS and the proxy measures of those processes (brain and spinal cord MRI, PET, optical coherence tomography, and biomarkers).
“The paper reports there is no qualitative difference between relapsing remitting and progressive MS; rather, the difference is quantitative,” Dr. Cohen noted. “In other words, the processes that underlie progression are present from the earliest stages of MS, becoming more prominent over time.”
The apparent transition to progressive MS, he added, “rather than representing a ‘transition,’ instead results from the accumulation of pathology over time, a shift from focal lesions to diffuse inflammation and damage, and unmasking of the damage due to decreased resiliency due to aging and failure of compensatory mechanisms (neuroplasticity and remyelination).”
Also commenting, Edward Fox, MD, director, MS Clinic of Central Texas and clinical associate professor, University of Texas, Austin, explained that loss of tissue is the main driver of progressive MS.
“We all look at imaging to confirm that the progressive symptoms expressed by the patient are related to demyelinating disease,” he said. “When I see MRI of the spinal cord showing multifocal lesions, especially if localized atrophy is seen in a region of the cord, I expect to hear a history of progressive deficits in gait and other signs of disability.”
Dr. Fox noted that, on MRI of the brain, gray matter atrophy both cortically and in the deep gray structures usually manifests as cognitive slowing and poorer performance in work and social situations.
“We hope that other biomarkers, such as neurofilament light chain, will add to this body of knowledge and give us a better grasp of the definition of neurodegeneration to confirm the clinical and radiographic findings,” he added.
Dr. Filippi has received compensation for consulting services and/or speaking activities from Bayer, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi, Genzyme, Takeda, and Teva Pharmaceutical Industries; and research support from ARiSLA, Biogen Idec, Fondazione Italiana Sclerosi Multipla, Italian Ministry of Health, Merck Serono, Novartis, Roche, and Teva.
A version of this article first appeared on Medscape.com.
The role of imaging in diagnosing progressive multiple sclerosis (MS) and in assessing prognosis is the subject of a new review.
MRI is central in the diagnostic work-up of patients suspected of having MS, given its high sensitivity in detecting disease dissemination in space and over time and its notable ability to exclude mimics of MS, the authors noted. However, diagnosis of primary progressive MS remains challenging and is only possible retrospectively on the basis of clinical assessment.
they wrote.
Diagnosis of progressive MS is limited by difficulties in distinguishing accumulating disability caused by inflammatory disease activity from that attributable to degenerative processes associated with secondary progressive MS. Moreover, there are no accepted clinical criteria for diagnosing secondary progressive MS, the authors explained.
This need has promoted extensive research in the field of imaging, facilitated by definition of novel MRI sequences, to identify imaging features reflecting pathophysiological mechanisms relevant to the pathobiology of progressive MS, the authors said.
The current review reports the conclusions of a workshop held in Milan in November 2019, at which an expert panel of neurologists and neuroradiologists addressed the role of MRI in progressive MS.
Massimo Filippi, MD, IRCCS San Raffaele Scientific Institute, Milan, was the lead author of the review, which was published online Dec. 14, 2020, in JAMA Neurology.
The authors concluded that no definitive, qualitative clinical, immunologic, histopathologic, or neuroimaging features differentiate primary progressive and secondary progressive forms of MS; both are characterized by neurodegenerative phenomena and a gradual and irreversible accumulation of clinical disability, which is also affected by aging and comorbidities.
A definitive diagnosis of primary progressive MS is more difficult than a diagnosis of relapsing remitting MS; in part, primary progressive MS is a diagnosis of exclusion because it can be mimicked by other conditions clinically and radiologically, the authors noted.
The writers did report that, although nonspecific, some spinal cord imaging features are typical of primary progressive MS. These include diffuse abnormalities and lesions involving gray matter and two or more white-matter columns, but confirmation of this is required.
In patients with primary progressive MS and those with relapse-onset MS, MRI features at disease onset predict long-term disability and a progressive disease course. These features include lesions in critical central nervous system regions (i.e., spinal cord, infratentorial regions, and gray matter) and high inflammatory activity in the first years after disease onset. These measures are evaluable in clinical practice, the authors said.
In patients with established MS, gray-matter involvement and neurodegeneration are associated with accelerated clinical worsening; however, detection validation and standardization need to be implemented at the individual patient level, they commented.
Novel candidate imaging biomarkers, such as subpial demyelination, and the presence of slowly expanding lesions or paramagnetic rim lesions may identify progressive MS but should be further investigated, they added.
Discovery of MRI markers capable of detecting evolution from relapsing-remitting to secondary progressive MS remains an unmet need that will probably require multiparametric MRI studies, because it is unlikely that a single MRI method will be able to allow clinicians to optimally distinguish among these stages, the authors said.
The contribution of these promising MRI measures combined with other biomarkers, such as quantification of serum neurofilament light chain levels or optical coherence tomography assessment, should be explored to improve the identification of patients with progressive MS, they concluded.
‘A comprehensive review’
In a comment, Jeffrey A. Cohen, MD, director of the Cleveland Clinic’s Mellen Center for MS Treatment and Research, said the article is a comprehensive review of the pathologic mechanisms that underlie progression in MS and the proxy measures of those processes (brain and spinal cord MRI, PET, optical coherence tomography, and biomarkers).
“The paper reports there is no qualitative difference between relapsing remitting and progressive MS; rather, the difference is quantitative,” Dr. Cohen noted. “In other words, the processes that underlie progression are present from the earliest stages of MS, becoming more prominent over time.”
The apparent transition to progressive MS, he added, “rather than representing a ‘transition,’ instead results from the accumulation of pathology over time, a shift from focal lesions to diffuse inflammation and damage, and unmasking of the damage due to decreased resiliency due to aging and failure of compensatory mechanisms (neuroplasticity and remyelination).”
Also commenting, Edward Fox, MD, director, MS Clinic of Central Texas and clinical associate professor, University of Texas, Austin, explained that loss of tissue is the main driver of progressive MS.
“We all look at imaging to confirm that the progressive symptoms expressed by the patient are related to demyelinating disease,” he said. “When I see MRI of the spinal cord showing multifocal lesions, especially if localized atrophy is seen in a region of the cord, I expect to hear a history of progressive deficits in gait and other signs of disability.”
Dr. Fox noted that, on MRI of the brain, gray matter atrophy both cortically and in the deep gray structures usually manifests as cognitive slowing and poorer performance in work and social situations.
“We hope that other biomarkers, such as neurofilament light chain, will add to this body of knowledge and give us a better grasp of the definition of neurodegeneration to confirm the clinical and radiographic findings,” he added.
Dr. Filippi has received compensation for consulting services and/or speaking activities from Bayer, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi, Genzyme, Takeda, and Teva Pharmaceutical Industries; and research support from ARiSLA, Biogen Idec, Fondazione Italiana Sclerosi Multipla, Italian Ministry of Health, Merck Serono, Novartis, Roche, and Teva.
A version of this article first appeared on Medscape.com.
The role of imaging in diagnosing progressive multiple sclerosis (MS) and in assessing prognosis is the subject of a new review.
MRI is central in the diagnostic work-up of patients suspected of having MS, given its high sensitivity in detecting disease dissemination in space and over time and its notable ability to exclude mimics of MS, the authors noted. However, diagnosis of primary progressive MS remains challenging and is only possible retrospectively on the basis of clinical assessment.
they wrote.
Diagnosis of progressive MS is limited by difficulties in distinguishing accumulating disability caused by inflammatory disease activity from that attributable to degenerative processes associated with secondary progressive MS. Moreover, there are no accepted clinical criteria for diagnosing secondary progressive MS, the authors explained.
This need has promoted extensive research in the field of imaging, facilitated by definition of novel MRI sequences, to identify imaging features reflecting pathophysiological mechanisms relevant to the pathobiology of progressive MS, the authors said.
The current review reports the conclusions of a workshop held in Milan in November 2019, at which an expert panel of neurologists and neuroradiologists addressed the role of MRI in progressive MS.
Massimo Filippi, MD, IRCCS San Raffaele Scientific Institute, Milan, was the lead author of the review, which was published online Dec. 14, 2020, in JAMA Neurology.
The authors concluded that no definitive, qualitative clinical, immunologic, histopathologic, or neuroimaging features differentiate primary progressive and secondary progressive forms of MS; both are characterized by neurodegenerative phenomena and a gradual and irreversible accumulation of clinical disability, which is also affected by aging and comorbidities.
A definitive diagnosis of primary progressive MS is more difficult than a diagnosis of relapsing remitting MS; in part, primary progressive MS is a diagnosis of exclusion because it can be mimicked by other conditions clinically and radiologically, the authors noted.
The writers did report that, although nonspecific, some spinal cord imaging features are typical of primary progressive MS. These include diffuse abnormalities and lesions involving gray matter and two or more white-matter columns, but confirmation of this is required.
In patients with primary progressive MS and those with relapse-onset MS, MRI features at disease onset predict long-term disability and a progressive disease course. These features include lesions in critical central nervous system regions (i.e., spinal cord, infratentorial regions, and gray matter) and high inflammatory activity in the first years after disease onset. These measures are evaluable in clinical practice, the authors said.
In patients with established MS, gray-matter involvement and neurodegeneration are associated with accelerated clinical worsening; however, detection validation and standardization need to be implemented at the individual patient level, they commented.
Novel candidate imaging biomarkers, such as subpial demyelination, and the presence of slowly expanding lesions or paramagnetic rim lesions may identify progressive MS but should be further investigated, they added.
Discovery of MRI markers capable of detecting evolution from relapsing-remitting to secondary progressive MS remains an unmet need that will probably require multiparametric MRI studies, because it is unlikely that a single MRI method will be able to allow clinicians to optimally distinguish among these stages, the authors said.
The contribution of these promising MRI measures combined with other biomarkers, such as quantification of serum neurofilament light chain levels or optical coherence tomography assessment, should be explored to improve the identification of patients with progressive MS, they concluded.
‘A comprehensive review’
In a comment, Jeffrey A. Cohen, MD, director of the Cleveland Clinic’s Mellen Center for MS Treatment and Research, said the article is a comprehensive review of the pathologic mechanisms that underlie progression in MS and the proxy measures of those processes (brain and spinal cord MRI, PET, optical coherence tomography, and biomarkers).
“The paper reports there is no qualitative difference between relapsing remitting and progressive MS; rather, the difference is quantitative,” Dr. Cohen noted. “In other words, the processes that underlie progression are present from the earliest stages of MS, becoming more prominent over time.”
The apparent transition to progressive MS, he added, “rather than representing a ‘transition,’ instead results from the accumulation of pathology over time, a shift from focal lesions to diffuse inflammation and damage, and unmasking of the damage due to decreased resiliency due to aging and failure of compensatory mechanisms (neuroplasticity and remyelination).”
Also commenting, Edward Fox, MD, director, MS Clinic of Central Texas and clinical associate professor, University of Texas, Austin, explained that loss of tissue is the main driver of progressive MS.
“We all look at imaging to confirm that the progressive symptoms expressed by the patient are related to demyelinating disease,” he said. “When I see MRI of the spinal cord showing multifocal lesions, especially if localized atrophy is seen in a region of the cord, I expect to hear a history of progressive deficits in gait and other signs of disability.”
Dr. Fox noted that, on MRI of the brain, gray matter atrophy both cortically and in the deep gray structures usually manifests as cognitive slowing and poorer performance in work and social situations.
“We hope that other biomarkers, such as neurofilament light chain, will add to this body of knowledge and give us a better grasp of the definition of neurodegeneration to confirm the clinical and radiographic findings,” he added.
Dr. Filippi has received compensation for consulting services and/or speaking activities from Bayer, Biogen Idec, Merck Serono, Novartis, Roche, Sanofi, Genzyme, Takeda, and Teva Pharmaceutical Industries; and research support from ARiSLA, Biogen Idec, Fondazione Italiana Sclerosi Multipla, Italian Ministry of Health, Merck Serono, Novartis, Roche, and Teva.
A version of this article first appeared on Medscape.com.
Genomic analysis reveals insights into pathogenesis of neuroblastoma
Insights into the genetic drivers of the disease were identified based on data from whole-genome, whole-exome, and/or transcriptome sequencing of tumor samples.
“The comprehensive genome-wide analysis performed here allowed us to discover age-associated alterations in MYCN, TERT, PTPRD, and Ras pathway alterations, which, together with ATRX, represent the majority of common driver gene alterations in neuroblastoma,” wrote study author Samuel W. Brady, PhD, of St. Jude Children’s Research Hospital in Memphis, Tenn., and colleagues.
The group’s findings were published in Nature Communications.
The researchers integrated and analyzed data from 702 neuroblastomas encompassing all age and risk categories, with the goal of identifying rare driver events and age-related molecular aberrations. Among the samples, 23 were from patients who had relapsed.
The researchers found that 40% of samples had somatic alterations in known driver genes, with the most common alterations being MYCN (19%; primarily amplification), TERT (17%; structural variations [SVs]), SHANK2 (13%; SVs), PTPRD (11%; SVs and focal deletions), ALK (10%; single nucleotide variants [SNVs] and SVs), and ATRX (8%; multiple mutation types).
MYCN and TERT alterations were more common in younger children (median age of 2.3 years and 3.8 years, respectively), while ATRX alterations were more frequently seen in older patients (median age of 5.6 years).
“These findings suggest that the sympathetic nervous system, the tissue from which neuroblastoma arises, is susceptible to different oncogenic insults at different times during development, which could be explored in future investigations using animal models,” the researchers wrote.
Furthermore, they found evidence to suggest the COSMIC mutational signature 18 is the most common cause of driver SNVs in neuroblastoma, including most Ras-activating and ALK variants.
Signature 18 was enriched in neuroblastomas with increased expression of mitochondrial ribosome and electron transport–associated genes, 17q gain, and MYCN amplification.
“[T]his mutagenic process, which is caused by ROS [reactive oxygen species] in other settings (though not proven in neuroblastoma), may promote evolution and heterogeneity, as many driver SNVs, such as ALK mutations, are later events in neuroblastoma,” the researchers explained.
Based on these findings, the authors concluded that neuroblastomas with 17q gain may be amenable to precision medicines, possibly through targeting altered mitochondrial function.
“[Our] findings will identify patients who might be eligible for targeted therapy and those that may be at higher risk based on a combination of genetic alterations detected by these genome-wide sequencing methods,” commented study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
The study was supported by grants from the National Cancer Institute and by the American Lebanese Syrian Associated Charities of St. Jude Children’s Research Hospital. One author disclosed financial affiliations with Y-mabs Therapeutics, Abpro-Labs, Eureka Therapeutics, and Biotec Pharmacon.
SOURCE: Brady SW et al. Nat Commun. 2020 Oct 14. doi: 10.1038/s41467-020-18987-4.
Insights into the genetic drivers of the disease were identified based on data from whole-genome, whole-exome, and/or transcriptome sequencing of tumor samples.
“The comprehensive genome-wide analysis performed here allowed us to discover age-associated alterations in MYCN, TERT, PTPRD, and Ras pathway alterations, which, together with ATRX, represent the majority of common driver gene alterations in neuroblastoma,” wrote study author Samuel W. Brady, PhD, of St. Jude Children’s Research Hospital in Memphis, Tenn., and colleagues.
The group’s findings were published in Nature Communications.
The researchers integrated and analyzed data from 702 neuroblastomas encompassing all age and risk categories, with the goal of identifying rare driver events and age-related molecular aberrations. Among the samples, 23 were from patients who had relapsed.
The researchers found that 40% of samples had somatic alterations in known driver genes, with the most common alterations being MYCN (19%; primarily amplification), TERT (17%; structural variations [SVs]), SHANK2 (13%; SVs), PTPRD (11%; SVs and focal deletions), ALK (10%; single nucleotide variants [SNVs] and SVs), and ATRX (8%; multiple mutation types).
MYCN and TERT alterations were more common in younger children (median age of 2.3 years and 3.8 years, respectively), while ATRX alterations were more frequently seen in older patients (median age of 5.6 years).
“These findings suggest that the sympathetic nervous system, the tissue from which neuroblastoma arises, is susceptible to different oncogenic insults at different times during development, which could be explored in future investigations using animal models,” the researchers wrote.
Furthermore, they found evidence to suggest the COSMIC mutational signature 18 is the most common cause of driver SNVs in neuroblastoma, including most Ras-activating and ALK variants.
Signature 18 was enriched in neuroblastomas with increased expression of mitochondrial ribosome and electron transport–associated genes, 17q gain, and MYCN amplification.
“[T]his mutagenic process, which is caused by ROS [reactive oxygen species] in other settings (though not proven in neuroblastoma), may promote evolution and heterogeneity, as many driver SNVs, such as ALK mutations, are later events in neuroblastoma,” the researchers explained.
Based on these findings, the authors concluded that neuroblastomas with 17q gain may be amenable to precision medicines, possibly through targeting altered mitochondrial function.
“[Our] findings will identify patients who might be eligible for targeted therapy and those that may be at higher risk based on a combination of genetic alterations detected by these genome-wide sequencing methods,” commented study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
The study was supported by grants from the National Cancer Institute and by the American Lebanese Syrian Associated Charities of St. Jude Children’s Research Hospital. One author disclosed financial affiliations with Y-mabs Therapeutics, Abpro-Labs, Eureka Therapeutics, and Biotec Pharmacon.
SOURCE: Brady SW et al. Nat Commun. 2020 Oct 14. doi: 10.1038/s41467-020-18987-4.
Insights into the genetic drivers of the disease were identified based on data from whole-genome, whole-exome, and/or transcriptome sequencing of tumor samples.
“The comprehensive genome-wide analysis performed here allowed us to discover age-associated alterations in MYCN, TERT, PTPRD, and Ras pathway alterations, which, together with ATRX, represent the majority of common driver gene alterations in neuroblastoma,” wrote study author Samuel W. Brady, PhD, of St. Jude Children’s Research Hospital in Memphis, Tenn., and colleagues.
The group’s findings were published in Nature Communications.
The researchers integrated and analyzed data from 702 neuroblastomas encompassing all age and risk categories, with the goal of identifying rare driver events and age-related molecular aberrations. Among the samples, 23 were from patients who had relapsed.
The researchers found that 40% of samples had somatic alterations in known driver genes, with the most common alterations being MYCN (19%; primarily amplification), TERT (17%; structural variations [SVs]), SHANK2 (13%; SVs), PTPRD (11%; SVs and focal deletions), ALK (10%; single nucleotide variants [SNVs] and SVs), and ATRX (8%; multiple mutation types).
MYCN and TERT alterations were more common in younger children (median age of 2.3 years and 3.8 years, respectively), while ATRX alterations were more frequently seen in older patients (median age of 5.6 years).
“These findings suggest that the sympathetic nervous system, the tissue from which neuroblastoma arises, is susceptible to different oncogenic insults at different times during development, which could be explored in future investigations using animal models,” the researchers wrote.
Furthermore, they found evidence to suggest the COSMIC mutational signature 18 is the most common cause of driver SNVs in neuroblastoma, including most Ras-activating and ALK variants.
Signature 18 was enriched in neuroblastomas with increased expression of mitochondrial ribosome and electron transport–associated genes, 17q gain, and MYCN amplification.
“[T]his mutagenic process, which is caused by ROS [reactive oxygen species] in other settings (though not proven in neuroblastoma), may promote evolution and heterogeneity, as many driver SNVs, such as ALK mutations, are later events in neuroblastoma,” the researchers explained.
Based on these findings, the authors concluded that neuroblastomas with 17q gain may be amenable to precision medicines, possibly through targeting altered mitochondrial function.
“[Our] findings will identify patients who might be eligible for targeted therapy and those that may be at higher risk based on a combination of genetic alterations detected by these genome-wide sequencing methods,” commented study author Jinghui Zhang, PhD, of St. Jude Children’s Research Hospital.
The study was supported by grants from the National Cancer Institute and by the American Lebanese Syrian Associated Charities of St. Jude Children’s Research Hospital. One author disclosed financial affiliations with Y-mabs Therapeutics, Abpro-Labs, Eureka Therapeutics, and Biotec Pharmacon.
SOURCE: Brady SW et al. Nat Commun. 2020 Oct 14. doi: 10.1038/s41467-020-18987-4.
FROM NATURE COMMUNICATIONS
Guideline Concordance with Durvalumab in Unresectable Stage III Non-Small Cell Lung Cancer: A Single Center Veterans Hospital Experience
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
The US Food and Drug Administration (FDA) approved the use of durvalumab for patients with unresectable stage III non-small cell lung cancer (NSCLC) whose disease has not progressed following concurrent platinum-based chemotherapy and radiation therapy (CRT).1 After 2 randomized phase 3 studies in 2017 and 2018 showed significant progression-free and overall survival respectively,2,3 durvalumab became a category 1 recommendation for the above indication per National Comprehensive Cancer Network (NCCN) guidelines.4 Adherence to guidelines have been shown to improve patient survival across several cancer types.5-7 However, guideline adherence rates have been variable across health institutions. Therefore, further study is warranted to evaluate nonadherent practices with the goal of improving the quality of cancer care delivery.8,9
Stage III NSCLC is associated with poor survival rates.10 Concurrent CRT remains the standard of care in patients with good performance status based on clinical trial populations.4 Lung cancer remains a disease of the elderly, with a median age at diagnosis of 70 years.11 Discrepancies in the treatment of lung cancer in older adults can vary widely due to a lack of evidence surrounding the treatment in those who have comorbidities and poor performance status, widening the gap between clinical trial and real-world populations.11
A recent review by Passaro and colleagues revealed that at least 11 pivotal randomized controlled trials have shown the activity of immune checkpoint inhibitors (ICI) in locally advanced and metastatic lung cancer. However, these studies have mostly excluded patients with a performance status of the Eastern Cooperative Oncology Group (ECOG) level ≥ 2.11
Durvalumab is one of many new therapies to enter clinical practice to demonstrate survival benefit, but its use among veterans with stage III NSCLC in adherence with National Comprehensive Cancer Network (NCCN) guidelines was not robust at the Birmingham Veterans Affairs Medical Center (VAMC) in Alabama. Therefore, we decided to study the level of adherence and to identify barriers to conformity to the category 1 NCCN recommendations.
Methods
The Birmingham VAMC Outpatient Oncology Clinic billing data identified all individuals diagnosed with lung cancer treated between October 2017 and August 2019. Patients who did not have NSCLC that was stage III and unresectable were excluded from our study. Patients who did not receive a majority of their treatment at US Department of Veterans Affairs (VA) facilities were excluded as well. Each patient’s demographic, functional level, and tumor characteristics during the treatment planning phase and follow-up visits were obtained. Two investigators who evaluated health care provider documentation using the VA Computerized Patient Record System (CPRS) conducted chart reviews.
The primary outcomes were the proportion of patients who received concurrent CRT and the proportion who received durvalumab consolidation. Our chart review also categorized reasons for nonreceipt of concurrent CRT and subsequent durvalumab. Documented reasons for guideline discordancy were generated empirically and broadly. We noted if documentation was unclear and included reasons for why a veteran was not a candidate for CRT, the presence of toxicities associated with CRT, and a patient’s refusal for therapy despite medical advice. Descriptive data were analyzed for all clinical or demographic characteristics and outcomes.
This was considered an internal quality improvement initiative. As such, Birmingham VAMC did not require institutional review board approval for the study. The facility is accredited by the American College of Surgeons Commission on Cancer.
Results
A total of 41 veterans with stage III NSCLC were identified to have established care in the Birmingham VAMC Oncology Clinic between October 2017 and August 2019. Of these, 7 received the majority of their treatment from community-based non-VA facilities and 14 were not candidates for CRT and were excluded from this study.
The mean (SD) age of study participants was 70.0 (8.4) years (range, 57 to 92 years). Most of the study veterans (33; 97.1%) were male and 20 (58.8%) were African American (Table). Eighteen (53%) of study participants had clinical stage IIIa NSCLC; 19 (56%) showed a squamous subtype of NSCLC. A majority (53%) of the veterans studied were evaluated to be functionally fit with an ECOG status of 0 to 1, although documentation of ECOG status was lacking in 5 (14.7%) patients in the initial treatment planning visit records. It was unclear if performance status had been reevaluated and changes noted over the course of concurrent CRT.
CRT Patients
The relative distribution of veterans who underwent CRT for stage III NSCLC plus the reasons they did not receive guideline-based treatment with durvalumab is shown in the Figure. Fourteen patients (41%) were inappropriate candidates for CRT; the most common reason for this was their poor performance status upon initial evaluation and 3 patients (8.8%) in the study had extensive disease or were upstaged upon follow-up clinic visit.
Twenty (59%) veterans in the study initiated CRT. However, only 16 (47.1%) completed CRT. Those who dropped out of CRT did so because of toxicities that included various cytopenia, gastrointestinal toxicities due to radiation and/or chemotherapy, or failure to thrive.
Durvalumab Treatment
After initiation of CRT, 9 (26.5%) patients did not go on to receive durvalumab. Three patients (8.8%) suffered toxicities during CRT. One study patient was found to have a severe respiratory infection requiring intensive care unit admission. Another study patient was found to have a new sternal lesion on follow-up positron emission tomography. One declined because of a history of severe antineutrophil cytoplasmic antibodies vasculitis, which made durvalumab use unsafe. Three patients (8.8%) declined treatment with CRT or durvalumab because of personal preference. Documentation was unclear as to why durvalumab was prescribed to one patient who had completed CRT.
Discussion
NCCN guidelines on the use of durvalumab in NSCLC are based on the phase 3 PACIFIC placebo-controlled randomized clinical trial. This trial, which included only patients with documented performance status of ECOG 0 or 1, reported that grade 3 or 4 events occurred in 30.5% of patients randomized to consolidative durvalumab. Treatment was discontinued in 15.4% of patients due to adverse events.3
Our study examined consolidation therapy with durvalumab in patients with unresectable stage III NSCLC with an ECOG performance status of 0 to 1 who had not progressed after 2 or more cycles of definitive concurrent CRT.4 Patients with previous exposure to immunotherapy, a history of immunodeficiency, active infection, unresolved toxicity from CRT, autoimmune disease, and patients who received sequential CRT were excluded.2 Surprisingly, the adherence rate to guidelines was close to 100% with appropriate documentation and justification of CRT initiation and durvalumab use. Five (14.7%) of veterans with unresectable stage III NSCLC did not have clear documentation of ECOG status on initial visit and only 1 veteran who completed CRT did not have clear documentation as to why durvalumab was not provided. Unfortunately, 23 (68.6%) veterans in the study were unable to receive durvalumab, a potentially disease-modifying drug; nearly one-third (10) of veterans were deemed poor candidates for concurrent CRT despite the fact that 52.9% (18) of veterans in the study had a documented ECOG of 0 or 1 on initial evaluation.
Clinical Trials vs Real World
The heterogeneity between anticipated study populations, those who were able to receive durvalumab in the PACIFIC trial, compared with our observed real-world veteran population, likely stems from the lack of information about how comorbidity and fitness can affect the choice of therapeutic intervention in patients with lung cancer.12 In addition, older adults who participated in randomized controlled trials (RCTs) are not representative of the average older adult who presents to medical oncology clinics, making the application of guideline concordant care difficult.13
Similar real-world observations parallel to our analyses have confirmed, complemented and/or refuted findings of RCTs, and have helped impact the treatment of multiple acute and chronic conditions including influenza, cardiovascular disease, and diabetes.14
A component of socioeconomic barriers and access to supportive care played roles in the decisions of certain patients who chose not to undergo concurrent CRT despite medical advice. These 2 obstacles also affected the decision making for some in the study when considering the use of durvalumab (administered by a 60-minute IV infusion every 2 weeks for 1 year) per recommended guidelines.1 These hurdles need further study in the context of their effect on quality of life and the difficulties generated by various social determinants of health.
Limitations
Study limitations included the biased and confounding factors previously described about retrospective and nonrandomized observational studies that are controlled for during RCTs.15 Electronic health record data may have been incorrectly collected resulting in missing or wrong data points that affect the validity of our conclusion. Recall bias with regard to documentation by health care providers describing reasons why CRT or durvalumab were not initiated or the patient’s ability to recall previous treatments and report ECOG status or toxicities also may have impacted our findings. Comorbidities and poor performance status, frequently occurring among veterans, negatively impact cancer treatment decisions and may result in a detection bias. For example, tobacco use, cardiovascular disease, including heart failure, and chronic obstructive pulmonary disease, are notoriously higher in the US veteran population when compared with civilian cohorts.16-18 Also, veterans with poorly controlled depression and posttraumatic stress disorder resulting in functional impairment are a factor.19 Steps were taken to address some of these biases by performing repeat checks of tabulated data and employing 2 independent reviewers to evaluate all relevant clinical documentation, compare results, and reach a consensus.
Conlcusions
This retrospective analysis of adherence to category 1 NCCN guidelines for durvalumab use among patients at the Birmingham VAMC Oncology Clinic reinforced our practice and identified minor deficiencies in documentation that would impact future clinical visits. More importantly, it depicted the massive disparity in treatment candidacy among Birmingham veterans compared with clinical trial populations. Efforts will be made to address factors impacting a veteran’s candidacy for CRT and explore other variables such as socioeconomic barriers to treatment. Multiple complementary tools to assess patients’ frailty, such as the Charlson Comorbidity Index (CCI), are now being used for a variety of disorders including cancers. More robust data and standardization are needed to validate the use of these assessments in predicting response to immune checkpoint inhibitors.
Immune checkpoint inhibitors are currently being evaluated in stage III NSCLC studies and may be implemented as routine practice in the future.12 It is important to distinguish fit from frail veterans with lung cancer for treatment selection. We would like to see the expansion of the eligibility criteria for clinical trials to include patients with a performance status of ECOG 2 in order for results to be truly generalizable to the real-world population. Our hope is that such work will improve not only the quality of lung cancer care, but also the quality of care across multiple tumor types.
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
1. US Food and Drug Administration. FDA approves durvalumab after chemoradiation for unresectable stage II. Published February 20, 2018. Accessed October 9, 2020. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-durvalumab-after-chemoradiation-unresectable-stage-iii-nsclc
2. Antonia SJ, Villegas A, Daniel D, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377(20):1919-1929. doi:10.1056/NEJMoa1709937
3. Antonia SJ, Villegas A, Daniel D, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379(24):2342-2350. doi:10.1056/NEJMoa1809697
4. Ettinger DS, Wood DE, Aisner DL et al. NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version8.2020. Updated September 15, 2020. Accessed October 9, 2020. https://www.nccn.org/professionals/physician_gls/pdf/nscl.pdf
5. Bristow RE, Chang J, Ziogas A, Campos B, Chavez LR, Anton-Culver H. Impact of National Cancer Institute Comprehensive Cancer Centers on ovarian cancer treatment and survival. J Am Coll Surg. 2015;220(5):940-950. doi:10.1016/j.jamcollsurg.2015.01.056
6. Boland GM, Chang GJ, Haynes AB, et al. Association between adherence to National Comprehensive Cancer Network treatment guidelines and improved survival in patients with colon cancer. Cancer. 2013;119(8):1593-1601. doi:10.1002/cncr.27935
7. Schwentner L, Wöckel A, König J, et al. Adherence to treatment guidelines and survival in triple-negative breast cancer: a retrospective multi-center cohort study with 9,156 patients. BMC Cancer. 2013;13:487. Published 2013 Oct 21. doi:10.1186/1471-2407-13-487
8. Jazieh A, Alkaiyat MO, Ali Y, Hashim MA, Abdelhafiz N, Al Olayan A. Improving adherence to lung cancer guidelines: a quality improvement project that uses chart review, audit and feedback approach. BMJ Open Qual. 2019;8(3):e000436. Published 2019 Aug 26. doi:10.1136/bmjoq-2018-000436
9. Shaverdian N, Offin MD, Rimner A, et al. Utilization and factors precluding the initiation of consolidative durvalumab in unresectable stage III non-small cell lung cancer. Radiother Oncol. 2020;144:101-104. doi:10.1016/j.radonc.2019.11.015
10. National Cancer Institute. SEER cancer statistics review, 1975-2015, Table 15.1 cancer of the lung and bronchus. Accessed October 19, 2020 https://seer.cancer.gov/archive/csr/1975_2015/results_merged/sect_15_lung_bronchus.pdf. Updated September 10, 2018
11. Passaro A, Spitaleri G, Gyawali B, de Marinis F. Immunotherapy in non-small-cell lung cancer patients with performance status 2: clinical decision making with scant evidence. J Clin Oncol. 2019;37(22):1863-1867. doi:10.1200/JCO.18.02118
12. Driessen EJM, Janssen-Heijnen MLG, Maas HA, Dingemans AC, van Loon JGM. Study protocol of the NVALT25-ELDAPT trial: selecting the optimal treatment for older patients with stage III non-small-cell lung cancer. Clin Lung Cancer. 2018;19(6):e849-e852. doi:10.1016/j.cllc.2018.07.003
13. Schulkes KJ, Nguyen C, van den Bos F, van Elden LJ, Hamaker ME. Selection of Patients in Ongoing Clinical Trials on Lung Cancer. Lung. 2016;194(6):967-974. doi:10.1007/s00408-016-9943-7
14. Blonde L, Khunti K, Harris SB, Meizinger C, Skolnik NS. Interpretation and impact of real-world clinical data for the practicing clinician. Adv Ther. 2018;35(11):1763-1774. doi:10.1007/s12325-018-0805-y
15. Garrison LP Jr, Neumann PJ, Erickson P, Marshall D, Mullins CD. Using real-world data for coverage and payment decisions: the ISPOR Real-World Data Task Force report. Value Health. 2007;10(5):326-335. doi:10.1111/j.1524-4733.2007.00186.x
16. Assari S. Veterans and risk of heart disease in the United States: a cohort with 20 years of follow up. Int J Prev Med. 2014;5(6):703-709.
17. Shahoumian TA, Phillips BR, Backus LI. Cigarette smoking, reduction and quit attempts: prevalence among veterans with coronary heart disease. Prev Chronic Dis. 2016;13:E41. Published 2016 Mar 24. doi:10.5888/pcd13.150282
18. Murphy DE, Chaudhry Z, Almoosa KF, Panos RJ. High prevalence of chronic obstructive pulmonary disease among veterans in the urban midwest. Mil Med. 2011;176(5):552-560. doi:10.7205/milmed-d-10-00377
19. Kozel FA, Didehbani N, DeLaRosa B, et al. Factors impacting functional status in veterans of recent conflicts with PTSD. J Neuropsychiatry Clin Neurosci. 2016;28(2):112-117. doi:10.1176/appi.neuropsych.15070183
Migraine nerve stimulation device now available over the counter
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
The Food and Drug Administration has cleared Cefaly Dual (Cefaly Technology) which was previously available only by prescription.
Most migraines involve the trigeminal nerve, which can be accessed through the skin on the forehead. Cefaly Dual stimulates the trigeminal nerve using a reusable self-adhesive electrode placed on the forehead.
The device has two settings, ACUTE and PREVENT. In the ACUTE setting, the individual wears the device for 60 minutes at headache onset or during a migraine attack. In the PREVENT setting, the individual wears the device for 20 minutes daily to help prevent future episodes.
At the start of a session, the wearer may feel a slight tingling sensation, which gradually increases and spreads throughout the forehead and the front part of the head. After about 14 minutes, the intensity stabilizes and remains constant until the treatment session is over, according to the company. The device automatically shuts off at the end of each session. It can be used as a stand-alone option or with existing treatment, the company noted.
“For millions of people across the U.S., living with migraine pain and coping with debilitating symptoms are daily realities. It is our mission to provide consumers with increased access to an effective and safe dual modality migraine treatment that is scientifically proven to reduce the number of monthly migraine days by almost half,” Jennifer Trainor McDermott, CEO of Cefaly Technology, said in a news release.
The FDA’s over-the-counter clearance of Cefaly Dual was based on several randomized, controlled clinical trials supporting the efficacy and safety of the device, the company said.
An earlier version of the Cefaly device was approved in the United States in March 2014 to help prevent migraine headache in adults aged 18 or older. The next-generation Cefaly Dual device is “small and sleek in comparison to its older model, which uses bands along the sides to create room for batteries. The newest device is palm-sized, more portable, and uses a battery that is rechargeable via USB,” the company said.
Last spring, the company announced a buyback program where customers in the United States may return their original device and receive a discount of the purchase of the Cefaly Dual device.
A version of this article originally appeared on Medscape.com.
In MS, serious adverse effects are more common in rituximab versus ocrelizumab
, a new postmarketing analysis finds, and AE-related deaths were not unusual. Serious AEs, and those linked to death, were more common in the rituximab group, although the reported infection rate was higher in the ocrelizumab group.
The analysis, published Aug. 21 in the Multiple Sclerosis Journal, highlights the importance of monitoring patients for infections and encouraging them to do the same, the authors said.
“This report points out the impact of treatments in terms of unrecognized or underappreciated complications,” said Mark Gudesblatt, MD, medical director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Patchogue, N.Y., who reviewed the study findings. “These medications have a significant downside.”
Lead author Natalia Gonzalez Caldito, MD, of the University of Texas Southwestern Medical Center, Dallas, and colleagues analyzed AEs for the drugs in the Food and Drug Administration’s Adverse Event Reporting System. They only included cases in which the drugs were solely used to treat MS and were indicated as the cause of the AEs.
Rituximab (Rituxan) and ocrelizumab (Ocrevus) are both monoclonal antibodies. Rituximab is not FDA approved for MS but is used off label; ocrelizumab is approved for the relapsing forms of MS and primary progressive MS.
The researchers found 623 AE reports and 1,466 total AEs for rituximab and 7,948 and 23,613, respectively, for ocrelizumab. The average ages for the groups were 48.76 versus 43.89, respectively, (P < .001), and 71% in each group were women.
Among total AEs, serious AEs were more common in the rituximab group versus the ocrelizumab group (64.8% vs. 56.3%, respectively, P < .001). Adverse events that caused death were also more common in the rituximab group versus the ocrelizumab group (5.75% vs. 2.11%, P < .001).
Infections and infestations were more common in the ocrelizumab group than the rituximab group (21.93% vs. 11.05%, respectively, P < .001). However, certain AEs were more common in the rituximab group than the ocrelizumab group: Those in the blood and lymphatic system category (2.86% vs. 0.91%, respectively, P < .001), and those in the neoplasms category (4.02% vs. 1.28%, P < .001, respectively).
Researchers found a highly strong association between rituximab and a rare side effects – ear pruritus (itching, 0.8%). They also identified signals for infusion-related reaction (4.82%), throat irritation (4.01%) and throat tightness (1.44%), malignant melanoma (0.8%), breast cancer (1.77%) and neutropenia (2.57%).
Among the ocrelizumab AEs, researchers found the strongest association with oral herpes (2.21%), and they found other signals for herpes zoster (2.89%), urinary tract infection (10.52%), nasopharyngitis (9.79%), infusion-related reaction (4.76%), throat irritation (3.08%), and notably MS relapses (4.1%).
“Additional pharmacovigilance studies are needed to explore and further characterize these findings,” the researchers wrote. “Furthermore, these observations suggest that the AE profile of other second-generation anti-CD20 [monoclonal antibodies] may also differ from those of rituximab and ocrelizumab.”
Dr. Gudesblatt praised the analysis and said the findings make sense. “Use of B-cell–depleting agents lead to accumulative immune deficiency in routine care, which leads to higher rates of infection,” he said. He added that, “in the clinical trials for ocrelizumab, patients with IgG and IgM deficiency were excluded, but there is no advisement to exclude such patients in real care. The rates of infection in those patients with MS who have preexisting immune deficiencies and who are treated with these agents are unknown.”
The prospect of AEs is especially worrisome, he said, since “this information is only short term. Who knows what effect the prolonged use of unopposed B-cell depletion will have on infections in the long run?”
Neurologist Mitchell Wallin, MD, MPH, of George Washington University, Washington, and the University of Maryland, Baltimore County, said in an interview that the analysis is rigorous and especially useful because it includes a wider array of subjects – including those who are older and sicker – than took part in earlier clinical trials. “It’s really important to look at this real-world evidence,” he said, “and basically put this in the back of your head when you follow up with your patients.”
No study funding was reported. The corresponding author reported various disclosures. Dr. Gudesblatt and Dr. Wallin reported no disclosures.
SOURCE: Gonzalez Caldito N et al. Mult Scler J. 2020 Aug 21. doi: 10.1177/1352458520949986.
, a new postmarketing analysis finds, and AE-related deaths were not unusual. Serious AEs, and those linked to death, were more common in the rituximab group, although the reported infection rate was higher in the ocrelizumab group.
The analysis, published Aug. 21 in the Multiple Sclerosis Journal, highlights the importance of monitoring patients for infections and encouraging them to do the same, the authors said.
“This report points out the impact of treatments in terms of unrecognized or underappreciated complications,” said Mark Gudesblatt, MD, medical director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Patchogue, N.Y., who reviewed the study findings. “These medications have a significant downside.”
Lead author Natalia Gonzalez Caldito, MD, of the University of Texas Southwestern Medical Center, Dallas, and colleagues analyzed AEs for the drugs in the Food and Drug Administration’s Adverse Event Reporting System. They only included cases in which the drugs were solely used to treat MS and were indicated as the cause of the AEs.
Rituximab (Rituxan) and ocrelizumab (Ocrevus) are both monoclonal antibodies. Rituximab is not FDA approved for MS but is used off label; ocrelizumab is approved for the relapsing forms of MS and primary progressive MS.
The researchers found 623 AE reports and 1,466 total AEs for rituximab and 7,948 and 23,613, respectively, for ocrelizumab. The average ages for the groups were 48.76 versus 43.89, respectively, (P < .001), and 71% in each group were women.
Among total AEs, serious AEs were more common in the rituximab group versus the ocrelizumab group (64.8% vs. 56.3%, respectively, P < .001). Adverse events that caused death were also more common in the rituximab group versus the ocrelizumab group (5.75% vs. 2.11%, P < .001).
Infections and infestations were more common in the ocrelizumab group than the rituximab group (21.93% vs. 11.05%, respectively, P < .001). However, certain AEs were more common in the rituximab group than the ocrelizumab group: Those in the blood and lymphatic system category (2.86% vs. 0.91%, respectively, P < .001), and those in the neoplasms category (4.02% vs. 1.28%, P < .001, respectively).
Researchers found a highly strong association between rituximab and a rare side effects – ear pruritus (itching, 0.8%). They also identified signals for infusion-related reaction (4.82%), throat irritation (4.01%) and throat tightness (1.44%), malignant melanoma (0.8%), breast cancer (1.77%) and neutropenia (2.57%).
Among the ocrelizumab AEs, researchers found the strongest association with oral herpes (2.21%), and they found other signals for herpes zoster (2.89%), urinary tract infection (10.52%), nasopharyngitis (9.79%), infusion-related reaction (4.76%), throat irritation (3.08%), and notably MS relapses (4.1%).
“Additional pharmacovigilance studies are needed to explore and further characterize these findings,” the researchers wrote. “Furthermore, these observations suggest that the AE profile of other second-generation anti-CD20 [monoclonal antibodies] may also differ from those of rituximab and ocrelizumab.”
Dr. Gudesblatt praised the analysis and said the findings make sense. “Use of B-cell–depleting agents lead to accumulative immune deficiency in routine care, which leads to higher rates of infection,” he said. He added that, “in the clinical trials for ocrelizumab, patients with IgG and IgM deficiency were excluded, but there is no advisement to exclude such patients in real care. The rates of infection in those patients with MS who have preexisting immune deficiencies and who are treated with these agents are unknown.”
The prospect of AEs is especially worrisome, he said, since “this information is only short term. Who knows what effect the prolonged use of unopposed B-cell depletion will have on infections in the long run?”
Neurologist Mitchell Wallin, MD, MPH, of George Washington University, Washington, and the University of Maryland, Baltimore County, said in an interview that the analysis is rigorous and especially useful because it includes a wider array of subjects – including those who are older and sicker – than took part in earlier clinical trials. “It’s really important to look at this real-world evidence,” he said, “and basically put this in the back of your head when you follow up with your patients.”
No study funding was reported. The corresponding author reported various disclosures. Dr. Gudesblatt and Dr. Wallin reported no disclosures.
SOURCE: Gonzalez Caldito N et al. Mult Scler J. 2020 Aug 21. doi: 10.1177/1352458520949986.
, a new postmarketing analysis finds, and AE-related deaths were not unusual. Serious AEs, and those linked to death, were more common in the rituximab group, although the reported infection rate was higher in the ocrelizumab group.
The analysis, published Aug. 21 in the Multiple Sclerosis Journal, highlights the importance of monitoring patients for infections and encouraging them to do the same, the authors said.
“This report points out the impact of treatments in terms of unrecognized or underappreciated complications,” said Mark Gudesblatt, MD, medical director of the Comprehensive MS Care Center at South Shore Neurologic Associates in Patchogue, N.Y., who reviewed the study findings. “These medications have a significant downside.”
Lead author Natalia Gonzalez Caldito, MD, of the University of Texas Southwestern Medical Center, Dallas, and colleagues analyzed AEs for the drugs in the Food and Drug Administration’s Adverse Event Reporting System. They only included cases in which the drugs were solely used to treat MS and were indicated as the cause of the AEs.
Rituximab (Rituxan) and ocrelizumab (Ocrevus) are both monoclonal antibodies. Rituximab is not FDA approved for MS but is used off label; ocrelizumab is approved for the relapsing forms of MS and primary progressive MS.
The researchers found 623 AE reports and 1,466 total AEs for rituximab and 7,948 and 23,613, respectively, for ocrelizumab. The average ages for the groups were 48.76 versus 43.89, respectively, (P < .001), and 71% in each group were women.
Among total AEs, serious AEs were more common in the rituximab group versus the ocrelizumab group (64.8% vs. 56.3%, respectively, P < .001). Adverse events that caused death were also more common in the rituximab group versus the ocrelizumab group (5.75% vs. 2.11%, P < .001).
Infections and infestations were more common in the ocrelizumab group than the rituximab group (21.93% vs. 11.05%, respectively, P < .001). However, certain AEs were more common in the rituximab group than the ocrelizumab group: Those in the blood and lymphatic system category (2.86% vs. 0.91%, respectively, P < .001), and those in the neoplasms category (4.02% vs. 1.28%, P < .001, respectively).
Researchers found a highly strong association between rituximab and a rare side effects – ear pruritus (itching, 0.8%). They also identified signals for infusion-related reaction (4.82%), throat irritation (4.01%) and throat tightness (1.44%), malignant melanoma (0.8%), breast cancer (1.77%) and neutropenia (2.57%).
Among the ocrelizumab AEs, researchers found the strongest association with oral herpes (2.21%), and they found other signals for herpes zoster (2.89%), urinary tract infection (10.52%), nasopharyngitis (9.79%), infusion-related reaction (4.76%), throat irritation (3.08%), and notably MS relapses (4.1%).
“Additional pharmacovigilance studies are needed to explore and further characterize these findings,” the researchers wrote. “Furthermore, these observations suggest that the AE profile of other second-generation anti-CD20 [monoclonal antibodies] may also differ from those of rituximab and ocrelizumab.”
Dr. Gudesblatt praised the analysis and said the findings make sense. “Use of B-cell–depleting agents lead to accumulative immune deficiency in routine care, which leads to higher rates of infection,” he said. He added that, “in the clinical trials for ocrelizumab, patients with IgG and IgM deficiency were excluded, but there is no advisement to exclude such patients in real care. The rates of infection in those patients with MS who have preexisting immune deficiencies and who are treated with these agents are unknown.”
The prospect of AEs is especially worrisome, he said, since “this information is only short term. Who knows what effect the prolonged use of unopposed B-cell depletion will have on infections in the long run?”
Neurologist Mitchell Wallin, MD, MPH, of George Washington University, Washington, and the University of Maryland, Baltimore County, said in an interview that the analysis is rigorous and especially useful because it includes a wider array of subjects – including those who are older and sicker – than took part in earlier clinical trials. “It’s really important to look at this real-world evidence,” he said, “and basically put this in the back of your head when you follow up with your patients.”
No study funding was reported. The corresponding author reported various disclosures. Dr. Gudesblatt and Dr. Wallin reported no disclosures.
SOURCE: Gonzalez Caldito N et al. Mult Scler J. 2020 Aug 21. doi: 10.1177/1352458520949986.
FROM MULTIPLE SCLEROSIS JOURNAL
Lowering rituximab dose in patients with MS proves safe and effective
“Given its favorable cost-effectiveness profile, [rituximab] remains a valuable treatment option in the current landscape of MS treatments, even at the reduced dose,” wrote Giulio Disanto, MD, PhD, of the Neurocenter of Southern Switzerland in Lugano, and coauthors. The study was published in Multiple Sclerosis Journal.
To determine the clinical and radiologic effectiveness of deescalating rituximab dosage – along with assessing any adverse outcomes – this observational, single-center study examined 59 patients with MS who had been treated with rituximab at 1,000 mg for at least 1 year before the study began. Roughly 63% (n = 37) of the patients had relapsing remitting MS (RRMS), while the rest (n = 22) had secondary progressive disease (SPD). Their median age was 51, and nearly 75% were women.
All patients underwent neurologic examinations at baseline and then every 3 months for 1 year, with new symptoms, infections, or adverse events being assessed via the Expanded Disability Status Scale (EDSS). They also underwent brain and spinal MRI at baseline and at 12 months while blood samples were taken at baseline and then every 3 months for 1 year, with previous data for both collected when available.
Study results
All 59 patients completed 12-month follow-up, and no relapses occurred in the year after lowering rituximab dosage to 500 mg. No significant differences were observed when comparing EDSS scores at the start of the 1,000-mg dose with the start of the 500-mg dose (Wilcoxon P = .131) as well as from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .284). Analyzing RRMS and SPD patients separately also led to no differences in EDSS scores from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .531; Wilcoxon P = .408).
During the 1,000-mg treatment period the number of patients who developed at least one new T2 lesion on their brain or spine was 9 and 4, respectively. During the 500-mg period, just one patient developed a new T2 brain lesion and two patients developed new T2 spine lesions. IgG and IgM levels did not change from the start of 500-mg treatment, although total dose of rituximab was inversely associated with IgG concentrations when previous treatment with 1,000 mg was factored in (coefficient, −0.439; P = 0.041).
A total of 33 patients reported at least one adverse event during the 500-mg treatment period, with only three events being classified as serious: one pancreatitis, one coronary stenting, and one neutropenia.
Validating clinical experience
“This randomized trial is an important step,” said Timothy Vollmer, MD, of the Rocky Mountain MS Center in Westminster, Colo., in an interview. “It clearly supports that you can lessen the dose, which will allow us to use this revolutionary drug for a longer period of time in patients.”
Dr. Vollmer noted that, at his center, they have been using 500 mg of rituximab over a 6-month period since 2010 without a formal clinical trial and with no notable difference in adverse outcomes on MRIs or disability scales. “This validates what we’ve been doing, which we appreciate,” he said.
“The next thing you have to do is determine whether you really have to give it every 6 months,” he added, “because the treatment effect in most patients will last, in terms of B-cell depletion, about a year or more. What we should be testing next is giving the 500 mg and waiting until patients begin to recover B cells before we give them the next cycle, to see if that helps decrease the major side effect, which is a drop in IgG levels.”
The authors acknowledged their study’s limitations, including a moderate sample size, a short follow-up period after 500-mg dosage, and an inability to confirm consistency among 1,000-mg dose administration among all patients, which “may well influence efficacy and safety measures.”
The study was supported by the Neurocenter of Southern Switzerland. One author declared numerous potential conflicts of interest, including receiving speaker fees, research fees, and travel support, and serving on advisory boards for various foundations, universities, and pharmaceutical companies.
SOURCE: Disanto G et al. Mult Scler J. 2020 Aug 25. doi: 10.1177/1352458520952036.
“Given its favorable cost-effectiveness profile, [rituximab] remains a valuable treatment option in the current landscape of MS treatments, even at the reduced dose,” wrote Giulio Disanto, MD, PhD, of the Neurocenter of Southern Switzerland in Lugano, and coauthors. The study was published in Multiple Sclerosis Journal.
To determine the clinical and radiologic effectiveness of deescalating rituximab dosage – along with assessing any adverse outcomes – this observational, single-center study examined 59 patients with MS who had been treated with rituximab at 1,000 mg for at least 1 year before the study began. Roughly 63% (n = 37) of the patients had relapsing remitting MS (RRMS), while the rest (n = 22) had secondary progressive disease (SPD). Their median age was 51, and nearly 75% were women.
All patients underwent neurologic examinations at baseline and then every 3 months for 1 year, with new symptoms, infections, or adverse events being assessed via the Expanded Disability Status Scale (EDSS). They also underwent brain and spinal MRI at baseline and at 12 months while blood samples were taken at baseline and then every 3 months for 1 year, with previous data for both collected when available.
Study results
All 59 patients completed 12-month follow-up, and no relapses occurred in the year after lowering rituximab dosage to 500 mg. No significant differences were observed when comparing EDSS scores at the start of the 1,000-mg dose with the start of the 500-mg dose (Wilcoxon P = .131) as well as from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .284). Analyzing RRMS and SPD patients separately also led to no differences in EDSS scores from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .531; Wilcoxon P = .408).
During the 1,000-mg treatment period the number of patients who developed at least one new T2 lesion on their brain or spine was 9 and 4, respectively. During the 500-mg period, just one patient developed a new T2 brain lesion and two patients developed new T2 spine lesions. IgG and IgM levels did not change from the start of 500-mg treatment, although total dose of rituximab was inversely associated with IgG concentrations when previous treatment with 1,000 mg was factored in (coefficient, −0.439; P = 0.041).
A total of 33 patients reported at least one adverse event during the 500-mg treatment period, with only three events being classified as serious: one pancreatitis, one coronary stenting, and one neutropenia.
Validating clinical experience
“This randomized trial is an important step,” said Timothy Vollmer, MD, of the Rocky Mountain MS Center in Westminster, Colo., in an interview. “It clearly supports that you can lessen the dose, which will allow us to use this revolutionary drug for a longer period of time in patients.”
Dr. Vollmer noted that, at his center, they have been using 500 mg of rituximab over a 6-month period since 2010 without a formal clinical trial and with no notable difference in adverse outcomes on MRIs or disability scales. “This validates what we’ve been doing, which we appreciate,” he said.
“The next thing you have to do is determine whether you really have to give it every 6 months,” he added, “because the treatment effect in most patients will last, in terms of B-cell depletion, about a year or more. What we should be testing next is giving the 500 mg and waiting until patients begin to recover B cells before we give them the next cycle, to see if that helps decrease the major side effect, which is a drop in IgG levels.”
The authors acknowledged their study’s limitations, including a moderate sample size, a short follow-up period after 500-mg dosage, and an inability to confirm consistency among 1,000-mg dose administration among all patients, which “may well influence efficacy and safety measures.”
The study was supported by the Neurocenter of Southern Switzerland. One author declared numerous potential conflicts of interest, including receiving speaker fees, research fees, and travel support, and serving on advisory boards for various foundations, universities, and pharmaceutical companies.
SOURCE: Disanto G et al. Mult Scler J. 2020 Aug 25. doi: 10.1177/1352458520952036.
“Given its favorable cost-effectiveness profile, [rituximab] remains a valuable treatment option in the current landscape of MS treatments, even at the reduced dose,” wrote Giulio Disanto, MD, PhD, of the Neurocenter of Southern Switzerland in Lugano, and coauthors. The study was published in Multiple Sclerosis Journal.
To determine the clinical and radiologic effectiveness of deescalating rituximab dosage – along with assessing any adverse outcomes – this observational, single-center study examined 59 patients with MS who had been treated with rituximab at 1,000 mg for at least 1 year before the study began. Roughly 63% (n = 37) of the patients had relapsing remitting MS (RRMS), while the rest (n = 22) had secondary progressive disease (SPD). Their median age was 51, and nearly 75% were women.
All patients underwent neurologic examinations at baseline and then every 3 months for 1 year, with new symptoms, infections, or adverse events being assessed via the Expanded Disability Status Scale (EDSS). They also underwent brain and spinal MRI at baseline and at 12 months while blood samples were taken at baseline and then every 3 months for 1 year, with previous data for both collected when available.
Study results
All 59 patients completed 12-month follow-up, and no relapses occurred in the year after lowering rituximab dosage to 500 mg. No significant differences were observed when comparing EDSS scores at the start of the 1,000-mg dose with the start of the 500-mg dose (Wilcoxon P = .131) as well as from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .284). Analyzing RRMS and SPD patients separately also led to no differences in EDSS scores from the start of the 500-mg dose to the end of follow-up (Wilcoxon P = .531; Wilcoxon P = .408).
During the 1,000-mg treatment period the number of patients who developed at least one new T2 lesion on their brain or spine was 9 and 4, respectively. During the 500-mg period, just one patient developed a new T2 brain lesion and two patients developed new T2 spine lesions. IgG and IgM levels did not change from the start of 500-mg treatment, although total dose of rituximab was inversely associated with IgG concentrations when previous treatment with 1,000 mg was factored in (coefficient, −0.439; P = 0.041).
A total of 33 patients reported at least one adverse event during the 500-mg treatment period, with only three events being classified as serious: one pancreatitis, one coronary stenting, and one neutropenia.
Validating clinical experience
“This randomized trial is an important step,” said Timothy Vollmer, MD, of the Rocky Mountain MS Center in Westminster, Colo., in an interview. “It clearly supports that you can lessen the dose, which will allow us to use this revolutionary drug for a longer period of time in patients.”
Dr. Vollmer noted that, at his center, they have been using 500 mg of rituximab over a 6-month period since 2010 without a formal clinical trial and with no notable difference in adverse outcomes on MRIs or disability scales. “This validates what we’ve been doing, which we appreciate,” he said.
“The next thing you have to do is determine whether you really have to give it every 6 months,” he added, “because the treatment effect in most patients will last, in terms of B-cell depletion, about a year or more. What we should be testing next is giving the 500 mg and waiting until patients begin to recover B cells before we give them the next cycle, to see if that helps decrease the major side effect, which is a drop in IgG levels.”
The authors acknowledged their study’s limitations, including a moderate sample size, a short follow-up period after 500-mg dosage, and an inability to confirm consistency among 1,000-mg dose administration among all patients, which “may well influence efficacy and safety measures.”
The study was supported by the Neurocenter of Southern Switzerland. One author declared numerous potential conflicts of interest, including receiving speaker fees, research fees, and travel support, and serving on advisory boards for various foundations, universities, and pharmaceutical companies.
SOURCE: Disanto G et al. Mult Scler J. 2020 Aug 25. doi: 10.1177/1352458520952036.
FROM MULTIPLE SCLEROSIS JOURNAL
FDA approves ofatumumab (Kesimpta) for relapsing forms of MS
including relapsing-remitting MS, active secondary progressive MS, and clinically isolated syndrome, Novartis announced in a press release. This is the first FDA approval of a self-administered, targeted B-cell therapy for these conditions, and is delivered via an autoinjector pen.
“This approval is wonderful news for patients with relapsing multiple sclerosis,” Stephen Hauser, MD, director of the Weill Institute for Neurosciences at the University of California, San Francisco, said in the press release. “Through its favorable safety profile and well-tolerated monthly injection regimen, patients can self-administer the treatment at home, avoiding visits to the infusion center,” he noted.
Dr. Hauser is cochair of the steering committee for the phase 3 ASCLEPIOS I and II studies that were part of the basis for the FDA’s approval.
Bruce Bebo, PhD, executive vice president of research at the National MS Society, said because response to disease-modifying treatments varies among individuals with MS, it’s important to have a range of treatment options available with differing mechanisms of action. “We are pleased to have an additional option approved for the treatment of relapsing forms of MS,” he said.
Twin studies
Formerly known as OMB157, ofatumumab is a precisely-dosed anti-CD20 monoclonal antibody administered subcutaneously via once-monthly injection. However, Novartis noted that initial doses are given at weeks 0, 1, and 2 – with the first injection occurring with a health care professional present.
The drug “is thought to work by binding to a distinct epitope on the CD20 molecule inducing potent B-cell lysis and depletion,” the manufacturer noted.
As previously reported, results for the ACLEPIOS I and II studies were presented at the 2019 Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), with additional results presented at the 2020 Virtual Annual Meeting of the Consortium of Multiple Sclerosis Centers. In addition, the findings were published in the New England Journal of Medicine.
The twin, identically designed phase 3 studies assessed the safety and efficacy of the drug at a monthly subcutaneous dose of 20 mg versus once daily teriflunomide 14-mg oral tablets. Together, the studies included 1,882 adult patients at more than 350 sites in 37 countries.
Results showed that the study drug reduced the annualized relapse rate (ARR) by 51% in the first study and by 59% in the second versus teriflunomide (P < .001 in both studies), meeting the primary endpoint. Both studies also showed significant reductions of gadolinium-enhancing (Gd+) T1 lesions (by 98% and 94%, respectively) and new or enlarging T2 lesions (by 82% and 85%).
The most commonly observed treatment-related adverse events for ofatumumab were upper respiratory tract infection, headache, and injection-related reactions.
Although the FDA first approved ofatumumab in 2009 for treating chronic lymphocytic leukemia (CLL), it was administered as a high-dose intravenous infusion by a healthcare provider. “This is a different dosing regimen and route of administration than was previously approved for the CLL indication,” the company noted.
The drug is expected to be available in the United States in September.
A version of this article originally appeared on Medscape.com.
including relapsing-remitting MS, active secondary progressive MS, and clinically isolated syndrome, Novartis announced in a press release. This is the first FDA approval of a self-administered, targeted B-cell therapy for these conditions, and is delivered via an autoinjector pen.
“This approval is wonderful news for patients with relapsing multiple sclerosis,” Stephen Hauser, MD, director of the Weill Institute for Neurosciences at the University of California, San Francisco, said in the press release. “Through its favorable safety profile and well-tolerated monthly injection regimen, patients can self-administer the treatment at home, avoiding visits to the infusion center,” he noted.
Dr. Hauser is cochair of the steering committee for the phase 3 ASCLEPIOS I and II studies that were part of the basis for the FDA’s approval.
Bruce Bebo, PhD, executive vice president of research at the National MS Society, said because response to disease-modifying treatments varies among individuals with MS, it’s important to have a range of treatment options available with differing mechanisms of action. “We are pleased to have an additional option approved for the treatment of relapsing forms of MS,” he said.
Twin studies
Formerly known as OMB157, ofatumumab is a precisely-dosed anti-CD20 monoclonal antibody administered subcutaneously via once-monthly injection. However, Novartis noted that initial doses are given at weeks 0, 1, and 2 – with the first injection occurring with a health care professional present.
The drug “is thought to work by binding to a distinct epitope on the CD20 molecule inducing potent B-cell lysis and depletion,” the manufacturer noted.
As previously reported, results for the ACLEPIOS I and II studies were presented at the 2019 Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), with additional results presented at the 2020 Virtual Annual Meeting of the Consortium of Multiple Sclerosis Centers. In addition, the findings were published in the New England Journal of Medicine.
The twin, identically designed phase 3 studies assessed the safety and efficacy of the drug at a monthly subcutaneous dose of 20 mg versus once daily teriflunomide 14-mg oral tablets. Together, the studies included 1,882 adult patients at more than 350 sites in 37 countries.
Results showed that the study drug reduced the annualized relapse rate (ARR) by 51% in the first study and by 59% in the second versus teriflunomide (P < .001 in both studies), meeting the primary endpoint. Both studies also showed significant reductions of gadolinium-enhancing (Gd+) T1 lesions (by 98% and 94%, respectively) and new or enlarging T2 lesions (by 82% and 85%).
The most commonly observed treatment-related adverse events for ofatumumab were upper respiratory tract infection, headache, and injection-related reactions.
Although the FDA first approved ofatumumab in 2009 for treating chronic lymphocytic leukemia (CLL), it was administered as a high-dose intravenous infusion by a healthcare provider. “This is a different dosing regimen and route of administration than was previously approved for the CLL indication,” the company noted.
The drug is expected to be available in the United States in September.
A version of this article originally appeared on Medscape.com.
including relapsing-remitting MS, active secondary progressive MS, and clinically isolated syndrome, Novartis announced in a press release. This is the first FDA approval of a self-administered, targeted B-cell therapy for these conditions, and is delivered via an autoinjector pen.
“This approval is wonderful news for patients with relapsing multiple sclerosis,” Stephen Hauser, MD, director of the Weill Institute for Neurosciences at the University of California, San Francisco, said in the press release. “Through its favorable safety profile and well-tolerated monthly injection regimen, patients can self-administer the treatment at home, avoiding visits to the infusion center,” he noted.
Dr. Hauser is cochair of the steering committee for the phase 3 ASCLEPIOS I and II studies that were part of the basis for the FDA’s approval.
Bruce Bebo, PhD, executive vice president of research at the National MS Society, said because response to disease-modifying treatments varies among individuals with MS, it’s important to have a range of treatment options available with differing mechanisms of action. “We are pleased to have an additional option approved for the treatment of relapsing forms of MS,” he said.
Twin studies
Formerly known as OMB157, ofatumumab is a precisely-dosed anti-CD20 monoclonal antibody administered subcutaneously via once-monthly injection. However, Novartis noted that initial doses are given at weeks 0, 1, and 2 – with the first injection occurring with a health care professional present.
The drug “is thought to work by binding to a distinct epitope on the CD20 molecule inducing potent B-cell lysis and depletion,” the manufacturer noted.
As previously reported, results for the ACLEPIOS I and II studies were presented at the 2019 Congress of the European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS), with additional results presented at the 2020 Virtual Annual Meeting of the Consortium of Multiple Sclerosis Centers. In addition, the findings were published in the New England Journal of Medicine.
The twin, identically designed phase 3 studies assessed the safety and efficacy of the drug at a monthly subcutaneous dose of 20 mg versus once daily teriflunomide 14-mg oral tablets. Together, the studies included 1,882 adult patients at more than 350 sites in 37 countries.
Results showed that the study drug reduced the annualized relapse rate (ARR) by 51% in the first study and by 59% in the second versus teriflunomide (P < .001 in both studies), meeting the primary endpoint. Both studies also showed significant reductions of gadolinium-enhancing (Gd+) T1 lesions (by 98% and 94%, respectively) and new or enlarging T2 lesions (by 82% and 85%).
The most commonly observed treatment-related adverse events for ofatumumab were upper respiratory tract infection, headache, and injection-related reactions.
Although the FDA first approved ofatumumab in 2009 for treating chronic lymphocytic leukemia (CLL), it was administered as a high-dose intravenous infusion by a healthcare provider. “This is a different dosing regimen and route of administration than was previously approved for the CLL indication,” the company noted.
The drug is expected to be available in the United States in September.
A version of this article originally appeared on Medscape.com.