User login
Sexual activity alters the microbiome, with potential psychiatric implications
Evidence is strong that sexual partners transmit microbiota (bacteria, viruses, fungi, protozoa, and archaea) to each other. While microbial flora are abundant in the gastrointestinal tract, they are also present in the vagina, penis, urethra, mouth, and skin.1 For better or worse, sexual contact of all types means that participants will acquire each other’s microbiota.
The 39 trillion microbiota in the body (which exceed the 30 trillion cells in the body) are commensal and influence both the larger brain in the skull and the smaller enteric brain in the gut. The microbiota and their microbiome genes (1,000 times larger than the human genome) have been linked to depression, anxiety, psychosis, and autism.2-4 They produce 90% of the body’s serotonin, as well as catecholamines (norepinephrine, epinephrine, dopamine), make hormones (eg, cortisol), and modulate the immune system. Microbiota have several important functions, including food digestion, synthesis of vitamins, autoimmunity, hypothalamic-pituitary-adrenal axis regulation, and CNS modulation.
Consequences of dysbiosis
Everyone should be concerned about maintaining a healthy diversity of microbiota in their body, with a predominance of beneficial bacteria such as Lactobacillus and Bacteroides, and avoiding acquiring pathogenic bacteria such as Gardnerella, Prevotella, and Atopobium. Sexual activity involving a partner with unhealthy microbiota may increase the risk of dysbiosis, defined as a reduction in microbiota diversity, including a loss of beneficial bacteria and a rise in harmful bacteria.
Dysbiosis is associated with multiple symptoms, including5:
- brain “fog,” irritability, mood changes, and anxiety
- bloating, loss of intestinal permeability, and insufficient reclamation of nutrients
- congestion of certain organs, such as the liver, gallbladder, and pancreas
- production of antigen-antibody complexes in response to chemicals in partially digested food
- aggravation of inflammatory disorders such as migraine, arthritis, and autoimmune disorders.
Apart from intimate sexual contact, simply sharing a household with someone leads to sharing of gut microflora. Persons who live together, whether genetically related or not, have similar microbiota. Compared with people living in separate households, cohabiting human pairs, dog pairs, and human-dog pairs share most of their microbiota (especially in the skin).
A consequence of acquiring pathogenic microbiota in the vagina is bacterial vaginosis (BV), which is not an infection but an ecologic imbalance in the composition of the vaginal microbiota. BV is caused by a significant decline in the beneficial vaginal Lactobacillus and a marked increase in the non-Lactobacillus taxa (especially Gardnerella and Atopobium).6 It can last for a least 1 week after sexual intercourse. BV is rare or absent among virgins. For a male partner, penile microbiota changes significantly after unprotected sex.6
Pathogenic bacteria can be cultivated from the glans, the coronal sulcus, and the prepuce, as well as from the penile skin, semen, urethra, and urine.6 Diverse bacteria exist in human semen, regardless if the male is fertile or infertile.7Anaerococcus is a biomarker for low sperm quality. Many of the semen bacteria are also found in the vagina of women with BV.7 Semen is a medium for the transmission of bacteria and viruses between men and women, and can contribute to sexually transmitted diseases.8
There are approximately 21 million cases of BV in the United States each year, and BV can also increase the risk of HIV and poor obstetric outcomes.9 The microbiota in the penile skin and urethra in males who have monogamous relationships with females are very similar to the vaginal microbiota of their female partner.
Consequences of BV include:
- decrease in hydrogen peroxide–producing bacilli
- prevalence of anaerobic bacteria (Prevotella, Gardnerella, and Atopobium)
- alkalinization, fishy odor, and gray-white vaginal discharge
- increase in the rate of pelvic inflammatory disease, ectopic pregnancy, endometriosis, preterm birth, and tubal factor infertility.9
Circumcision decreases the risk of BV. There is an increased rate of BV bacterial taxa in men with extramarital affairs and in women with multiple partners. Both oral and vaginal sex increase the abundance of Lactobacillus in the male oral and penile microbiota. Gingivitis has also been reported after oral sex.10
A link to psychiatric disorders
Given that all forms of sexual contact (vaginal, oral, anal, or skin) can transmit microbiota bidirectionally between partners, it is vital to practice safe sex and consider a monogamous relationship rather than indiscriminate promiscuity. Unfortunately, certain psychiatric disorders, such as bipolar disorder, are associated with hypersexuality and multiple partners, which may disrupt the microbiota. This can further disrupt the diversity of an individual’s microbiome and may put them at risk for mood, anxiety, and other psychiatric disorders. Another problem is sexually transmitted infections such as gonorrhea or syphilis require antibiotic therapy. It is well established that antibiotics kill both the bad pathogenic and the good nonpathogenic microbiota, further exacerbating dysbiosis and leading to disruptions in the microbiota-gut-brain (MGB) axis, which then results in psychiatric disorders.
The MGB axis modulates neurological processes via the vagus nerve, the major “highway” connecting the gut and brain for bidirectional traffic. The MGB axis produces microbial metabolites and immune factors that can lead to changes in brain neurotransmitters as well as neuroinflammation and psychiatric symptoms such as depression and anxiety.5
Many researchers are focusing on how to exploit the microbiome to develop novel therapeutic strategies, and encouraging advances are emerging.5 But the exact mechanisms by which the gut microbiome can impact mental health is still a work in progress. It is highly likely that dysbiosis is associated with mood and anxiety symptoms.
The bottom line: Sexual activity—whether it is heavy kissing, vaginal intercourse, oral sex, anal sex, or extensive skin contact—can lead to the exchange of microbiota. If an individual has dysbiosis, that could impact the mental health of their sexual partner(s). This raises the question of whether counseling patients about avoiding indiscriminate sex and practicing safe sex is as important for mental health as diet and exercise counseling is for physical health.
1. Reid G, Younes JA, Van der Mei HC, et al. Microbiota restoration: natural and supplemented recovery of human microbial communities. Nat Rev Microbiol. 2011;9(1):27-38.
2. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13(10):701-712.
3. Peirce JM, Alviña K. The role of inflammation and the gut microbiome in depression and anxiety. J Neurosci Res. 2019;97(10):1223-1241.
4. Yolken R, Prandovszky E, Severance EG, et al. The oropharyngeal microbiome is altered in individuals with schizophrenia and mania. Schizophr Res. 2021;234:51-57.
5. Capuco A, Urits I, Hasoon J, et al. Current perspectives on gut microbiome dysbiosis and depression. Adv Ther. 2020;37(4):1328-1346.
6. Zozaya M, Ferris MJ, Siren JD, et al. Bacterial communities in penile skin, male urethra, and vagina of heterosexual couples with and without bacterial vaginosis. Microbiome. 2016;4:16. doi:10.1186/s40168-016-0161-6
7. Hou D, Zhou X, Zhong X, et al. Microbiota of the seminal fluid from healthy and infertile men. Fertil Steril. 2013;100(5):1261-1269.
8. Gallo MF, Warner L, King CC, et al. Association between semen exposure and incident bacterial vaginosis. Infect Dis Obstet Gynecol. 2011;2011:842652.
9. Liu CM, Hungate BA, Tobian AA, et al. Penile microbiota and female partner bacterial vaginosis in Rakai, Uganda. mBio. 2015;6(3):e00589. doi:10.1128/mBio.00589-15
10. Carda-Diéguez M, Cárdenas N, Aparicio M, et al. Variations in vaginal, penile, and oral microbiota after sexual intercourse: a case report. Front Med. 2019;6:178. doi:10.3389/fmed.2019.00178
Evidence is strong that sexual partners transmit microbiota (bacteria, viruses, fungi, protozoa, and archaea) to each other. While microbial flora are abundant in the gastrointestinal tract, they are also present in the vagina, penis, urethra, mouth, and skin.1 For better or worse, sexual contact of all types means that participants will acquire each other’s microbiota.
The 39 trillion microbiota in the body (which exceed the 30 trillion cells in the body) are commensal and influence both the larger brain in the skull and the smaller enteric brain in the gut. The microbiota and their microbiome genes (1,000 times larger than the human genome) have been linked to depression, anxiety, psychosis, and autism.2-4 They produce 90% of the body’s serotonin, as well as catecholamines (norepinephrine, epinephrine, dopamine), make hormones (eg, cortisol), and modulate the immune system. Microbiota have several important functions, including food digestion, synthesis of vitamins, autoimmunity, hypothalamic-pituitary-adrenal axis regulation, and CNS modulation.
Consequences of dysbiosis
Everyone should be concerned about maintaining a healthy diversity of microbiota in their body, with a predominance of beneficial bacteria such as Lactobacillus and Bacteroides, and avoiding acquiring pathogenic bacteria such as Gardnerella, Prevotella, and Atopobium. Sexual activity involving a partner with unhealthy microbiota may increase the risk of dysbiosis, defined as a reduction in microbiota diversity, including a loss of beneficial bacteria and a rise in harmful bacteria.
Dysbiosis is associated with multiple symptoms, including5:
- brain “fog,” irritability, mood changes, and anxiety
- bloating, loss of intestinal permeability, and insufficient reclamation of nutrients
- congestion of certain organs, such as the liver, gallbladder, and pancreas
- production of antigen-antibody complexes in response to chemicals in partially digested food
- aggravation of inflammatory disorders such as migraine, arthritis, and autoimmune disorders.
Apart from intimate sexual contact, simply sharing a household with someone leads to sharing of gut microflora. Persons who live together, whether genetically related or not, have similar microbiota. Compared with people living in separate households, cohabiting human pairs, dog pairs, and human-dog pairs share most of their microbiota (especially in the skin).
A consequence of acquiring pathogenic microbiota in the vagina is bacterial vaginosis (BV), which is not an infection but an ecologic imbalance in the composition of the vaginal microbiota. BV is caused by a significant decline in the beneficial vaginal Lactobacillus and a marked increase in the non-Lactobacillus taxa (especially Gardnerella and Atopobium).6 It can last for a least 1 week after sexual intercourse. BV is rare or absent among virgins. For a male partner, penile microbiota changes significantly after unprotected sex.6
Pathogenic bacteria can be cultivated from the glans, the coronal sulcus, and the prepuce, as well as from the penile skin, semen, urethra, and urine.6 Diverse bacteria exist in human semen, regardless if the male is fertile or infertile.7Anaerococcus is a biomarker for low sperm quality. Many of the semen bacteria are also found in the vagina of women with BV.7 Semen is a medium for the transmission of bacteria and viruses between men and women, and can contribute to sexually transmitted diseases.8
There are approximately 21 million cases of BV in the United States each year, and BV can also increase the risk of HIV and poor obstetric outcomes.9 The microbiota in the penile skin and urethra in males who have monogamous relationships with females are very similar to the vaginal microbiota of their female partner.
Consequences of BV include:
- decrease in hydrogen peroxide–producing bacilli
- prevalence of anaerobic bacteria (Prevotella, Gardnerella, and Atopobium)
- alkalinization, fishy odor, and gray-white vaginal discharge
- increase in the rate of pelvic inflammatory disease, ectopic pregnancy, endometriosis, preterm birth, and tubal factor infertility.9
Circumcision decreases the risk of BV. There is an increased rate of BV bacterial taxa in men with extramarital affairs and in women with multiple partners. Both oral and vaginal sex increase the abundance of Lactobacillus in the male oral and penile microbiota. Gingivitis has also been reported after oral sex.10
A link to psychiatric disorders
Given that all forms of sexual contact (vaginal, oral, anal, or skin) can transmit microbiota bidirectionally between partners, it is vital to practice safe sex and consider a monogamous relationship rather than indiscriminate promiscuity. Unfortunately, certain psychiatric disorders, such as bipolar disorder, are associated with hypersexuality and multiple partners, which may disrupt the microbiota. This can further disrupt the diversity of an individual’s microbiome and may put them at risk for mood, anxiety, and other psychiatric disorders. Another problem is sexually transmitted infections such as gonorrhea or syphilis require antibiotic therapy. It is well established that antibiotics kill both the bad pathogenic and the good nonpathogenic microbiota, further exacerbating dysbiosis and leading to disruptions in the microbiota-gut-brain (MGB) axis, which then results in psychiatric disorders.
The MGB axis modulates neurological processes via the vagus nerve, the major “highway” connecting the gut and brain for bidirectional traffic. The MGB axis produces microbial metabolites and immune factors that can lead to changes in brain neurotransmitters as well as neuroinflammation and psychiatric symptoms such as depression and anxiety.5
Many researchers are focusing on how to exploit the microbiome to develop novel therapeutic strategies, and encouraging advances are emerging.5 But the exact mechanisms by which the gut microbiome can impact mental health is still a work in progress. It is highly likely that dysbiosis is associated with mood and anxiety symptoms.
The bottom line: Sexual activity—whether it is heavy kissing, vaginal intercourse, oral sex, anal sex, or extensive skin contact—can lead to the exchange of microbiota. If an individual has dysbiosis, that could impact the mental health of their sexual partner(s). This raises the question of whether counseling patients about avoiding indiscriminate sex and practicing safe sex is as important for mental health as diet and exercise counseling is for physical health.
Evidence is strong that sexual partners transmit microbiota (bacteria, viruses, fungi, protozoa, and archaea) to each other. While microbial flora are abundant in the gastrointestinal tract, they are also present in the vagina, penis, urethra, mouth, and skin.1 For better or worse, sexual contact of all types means that participants will acquire each other’s microbiota.
The 39 trillion microbiota in the body (which exceed the 30 trillion cells in the body) are commensal and influence both the larger brain in the skull and the smaller enteric brain in the gut. The microbiota and their microbiome genes (1,000 times larger than the human genome) have been linked to depression, anxiety, psychosis, and autism.2-4 They produce 90% of the body’s serotonin, as well as catecholamines (norepinephrine, epinephrine, dopamine), make hormones (eg, cortisol), and modulate the immune system. Microbiota have several important functions, including food digestion, synthesis of vitamins, autoimmunity, hypothalamic-pituitary-adrenal axis regulation, and CNS modulation.
Consequences of dysbiosis
Everyone should be concerned about maintaining a healthy diversity of microbiota in their body, with a predominance of beneficial bacteria such as Lactobacillus and Bacteroides, and avoiding acquiring pathogenic bacteria such as Gardnerella, Prevotella, and Atopobium. Sexual activity involving a partner with unhealthy microbiota may increase the risk of dysbiosis, defined as a reduction in microbiota diversity, including a loss of beneficial bacteria and a rise in harmful bacteria.
Dysbiosis is associated with multiple symptoms, including5:
- brain “fog,” irritability, mood changes, and anxiety
- bloating, loss of intestinal permeability, and insufficient reclamation of nutrients
- congestion of certain organs, such as the liver, gallbladder, and pancreas
- production of antigen-antibody complexes in response to chemicals in partially digested food
- aggravation of inflammatory disorders such as migraine, arthritis, and autoimmune disorders.
Apart from intimate sexual contact, simply sharing a household with someone leads to sharing of gut microflora. Persons who live together, whether genetically related or not, have similar microbiota. Compared with people living in separate households, cohabiting human pairs, dog pairs, and human-dog pairs share most of their microbiota (especially in the skin).
A consequence of acquiring pathogenic microbiota in the vagina is bacterial vaginosis (BV), which is not an infection but an ecologic imbalance in the composition of the vaginal microbiota. BV is caused by a significant decline in the beneficial vaginal Lactobacillus and a marked increase in the non-Lactobacillus taxa (especially Gardnerella and Atopobium).6 It can last for a least 1 week after sexual intercourse. BV is rare or absent among virgins. For a male partner, penile microbiota changes significantly after unprotected sex.6
Pathogenic bacteria can be cultivated from the glans, the coronal sulcus, and the prepuce, as well as from the penile skin, semen, urethra, and urine.6 Diverse bacteria exist in human semen, regardless if the male is fertile or infertile.7Anaerococcus is a biomarker for low sperm quality. Many of the semen bacteria are also found in the vagina of women with BV.7 Semen is a medium for the transmission of bacteria and viruses between men and women, and can contribute to sexually transmitted diseases.8
There are approximately 21 million cases of BV in the United States each year, and BV can also increase the risk of HIV and poor obstetric outcomes.9 The microbiota in the penile skin and urethra in males who have monogamous relationships with females are very similar to the vaginal microbiota of their female partner.
Consequences of BV include:
- decrease in hydrogen peroxide–producing bacilli
- prevalence of anaerobic bacteria (Prevotella, Gardnerella, and Atopobium)
- alkalinization, fishy odor, and gray-white vaginal discharge
- increase in the rate of pelvic inflammatory disease, ectopic pregnancy, endometriosis, preterm birth, and tubal factor infertility.9
Circumcision decreases the risk of BV. There is an increased rate of BV bacterial taxa in men with extramarital affairs and in women with multiple partners. Both oral and vaginal sex increase the abundance of Lactobacillus in the male oral and penile microbiota. Gingivitis has also been reported after oral sex.10
A link to psychiatric disorders
Given that all forms of sexual contact (vaginal, oral, anal, or skin) can transmit microbiota bidirectionally between partners, it is vital to practice safe sex and consider a monogamous relationship rather than indiscriminate promiscuity. Unfortunately, certain psychiatric disorders, such as bipolar disorder, are associated with hypersexuality and multiple partners, which may disrupt the microbiota. This can further disrupt the diversity of an individual’s microbiome and may put them at risk for mood, anxiety, and other psychiatric disorders. Another problem is sexually transmitted infections such as gonorrhea or syphilis require antibiotic therapy. It is well established that antibiotics kill both the bad pathogenic and the good nonpathogenic microbiota, further exacerbating dysbiosis and leading to disruptions in the microbiota-gut-brain (MGB) axis, which then results in psychiatric disorders.
The MGB axis modulates neurological processes via the vagus nerve, the major “highway” connecting the gut and brain for bidirectional traffic. The MGB axis produces microbial metabolites and immune factors that can lead to changes in brain neurotransmitters as well as neuroinflammation and psychiatric symptoms such as depression and anxiety.5
Many researchers are focusing on how to exploit the microbiome to develop novel therapeutic strategies, and encouraging advances are emerging.5 But the exact mechanisms by which the gut microbiome can impact mental health is still a work in progress. It is highly likely that dysbiosis is associated with mood and anxiety symptoms.
The bottom line: Sexual activity—whether it is heavy kissing, vaginal intercourse, oral sex, anal sex, or extensive skin contact—can lead to the exchange of microbiota. If an individual has dysbiosis, that could impact the mental health of their sexual partner(s). This raises the question of whether counseling patients about avoiding indiscriminate sex and practicing safe sex is as important for mental health as diet and exercise counseling is for physical health.
1. Reid G, Younes JA, Van der Mei HC, et al. Microbiota restoration: natural and supplemented recovery of human microbial communities. Nat Rev Microbiol. 2011;9(1):27-38.
2. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13(10):701-712.
3. Peirce JM, Alviña K. The role of inflammation and the gut microbiome in depression and anxiety. J Neurosci Res. 2019;97(10):1223-1241.
4. Yolken R, Prandovszky E, Severance EG, et al. The oropharyngeal microbiome is altered in individuals with schizophrenia and mania. Schizophr Res. 2021;234:51-57.
5. Capuco A, Urits I, Hasoon J, et al. Current perspectives on gut microbiome dysbiosis and depression. Adv Ther. 2020;37(4):1328-1346.
6. Zozaya M, Ferris MJ, Siren JD, et al. Bacterial communities in penile skin, male urethra, and vagina of heterosexual couples with and without bacterial vaginosis. Microbiome. 2016;4:16. doi:10.1186/s40168-016-0161-6
7. Hou D, Zhou X, Zhong X, et al. Microbiota of the seminal fluid from healthy and infertile men. Fertil Steril. 2013;100(5):1261-1269.
8. Gallo MF, Warner L, King CC, et al. Association between semen exposure and incident bacterial vaginosis. Infect Dis Obstet Gynecol. 2011;2011:842652.
9. Liu CM, Hungate BA, Tobian AA, et al. Penile microbiota and female partner bacterial vaginosis in Rakai, Uganda. mBio. 2015;6(3):e00589. doi:10.1128/mBio.00589-15
10. Carda-Diéguez M, Cárdenas N, Aparicio M, et al. Variations in vaginal, penile, and oral microbiota after sexual intercourse: a case report. Front Med. 2019;6:178. doi:10.3389/fmed.2019.00178
1. Reid G, Younes JA, Van der Mei HC, et al. Microbiota restoration: natural and supplemented recovery of human microbial communities. Nat Rev Microbiol. 2011;9(1):27-38.
2. Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13(10):701-712.
3. Peirce JM, Alviña K. The role of inflammation and the gut microbiome in depression and anxiety. J Neurosci Res. 2019;97(10):1223-1241.
4. Yolken R, Prandovszky E, Severance EG, et al. The oropharyngeal microbiome is altered in individuals with schizophrenia and mania. Schizophr Res. 2021;234:51-57.
5. Capuco A, Urits I, Hasoon J, et al. Current perspectives on gut microbiome dysbiosis and depression. Adv Ther. 2020;37(4):1328-1346.
6. Zozaya M, Ferris MJ, Siren JD, et al. Bacterial communities in penile skin, male urethra, and vagina of heterosexual couples with and without bacterial vaginosis. Microbiome. 2016;4:16. doi:10.1186/s40168-016-0161-6
7. Hou D, Zhou X, Zhong X, et al. Microbiota of the seminal fluid from healthy and infertile men. Fertil Steril. 2013;100(5):1261-1269.
8. Gallo MF, Warner L, King CC, et al. Association between semen exposure and incident bacterial vaginosis. Infect Dis Obstet Gynecol. 2011;2011:842652.
9. Liu CM, Hungate BA, Tobian AA, et al. Penile microbiota and female partner bacterial vaginosis in Rakai, Uganda. mBio. 2015;6(3):e00589. doi:10.1128/mBio.00589-15
10. Carda-Diéguez M, Cárdenas N, Aparicio M, et al. Variations in vaginal, penile, and oral microbiota after sexual intercourse: a case report. Front Med. 2019;6:178. doi:10.3389/fmed.2019.00178
Treatment augmentation strategies for OCD: A review of 8 studies
Obsessive-compulsive disorder (OCD) is a chronic, debilitating neuropsychiatric disorder that affects 1% to 3% of the population worldwide.1,2 Together, serotonin reuptake inhibitors (SRIs) and cognitive-behavior therapy (CBT) are considered the first-line treatment for OCD.3 In children and adults, CBT is considered at least as effective as pharmacotherapy.4 Despite being an effective treatment, CBT continues to have barriers to its widespread use, including limited availability of trained CBT therapists, delayed clinical response, and high costs.5
Only approximately one-half of patients with OCD respond to SRI therapy, and a considerable percentage (30% to 40%) show significant residual symptoms even after multiple trials of SRIs.6-8 In addition, SRIs may have adverse effects (eg, sexual dysfunction, gastrointestinal symptoms) that impair patient adherence to these medications.9 Therefore, finding better treatment options is important for managing patients with OCD.
Augmentation strategies are recommended for patients who show partial response to SRI treatment or poor response to multiple SRIs. Augmentation typically includes incorporating additional medications with the primary drug with the goal of boosting the therapeutic efficacy of the primary drug. Typically, these additional medications have different mechanisms of action. However, there are no large-scale randomized controlled trials (RCTs) to inform treatment augmentation after first-line treatments for OCD produce suboptimal outcomes. The available evidence is predominantly based on small-scale RCTs, open-label trials, and case series.
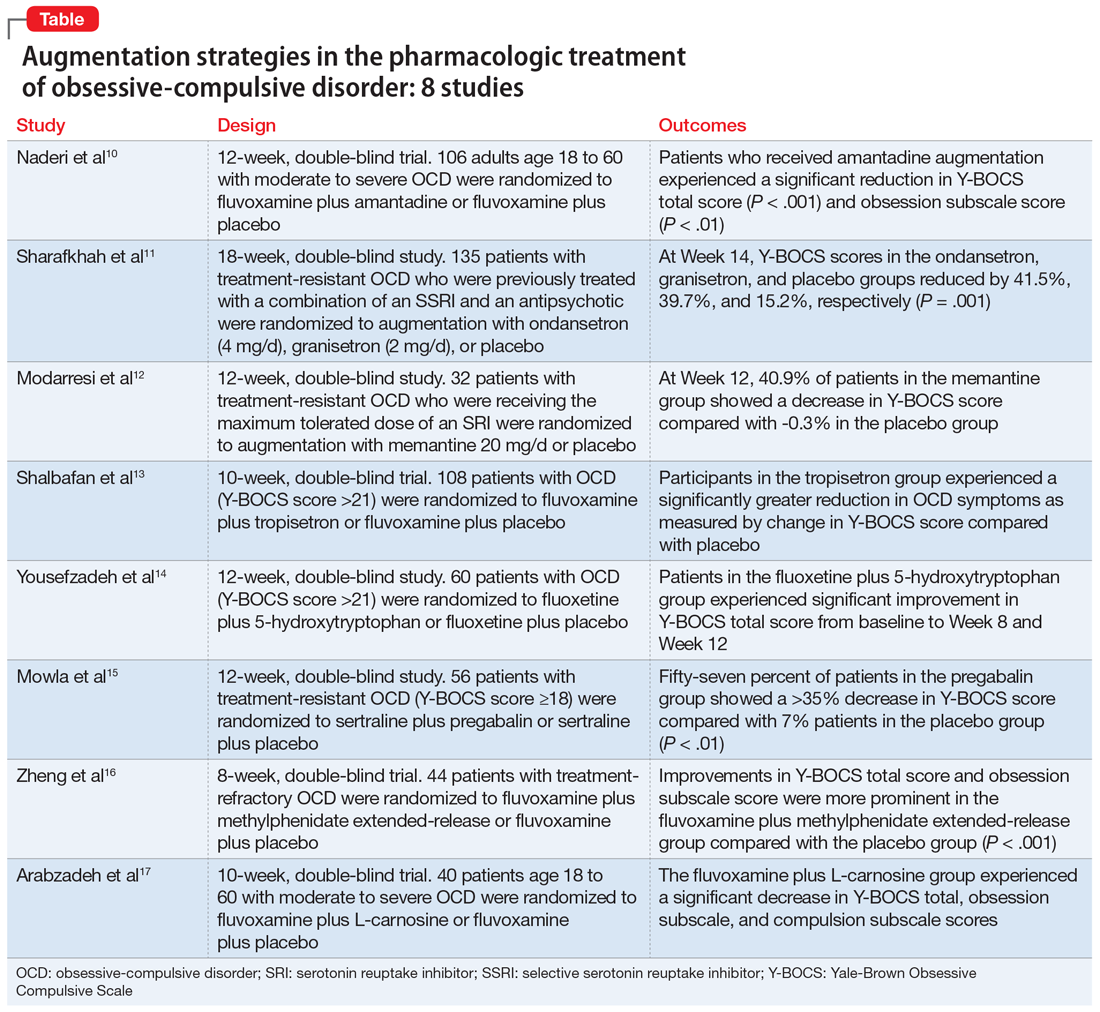
In this article, we review the evidence for treatment augmentation strategies for OCD and summarize 8 studies that show promising results (Table10-17). We focus only on pharmacologic agents and do not include other biological interventions, such as repetitive transcranial magnetic stimulation over supplementary motor area, ablative neurosurgery, or deep brain stimulation.
Continue to: Reference 1...
1. Naderi S, Faghih H , Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessivecompulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/ pcn.12803
Numerous studies support the role of glutamate dysregulation in the pathophysiology of OCD. Cortico-striato-thalamo-cortical (CSTC) abnormalities play a major role in the pathophysiology of OCD as suggested by neuroimaging research studies that indicate glutamate is the fundamental neurotransmitter of the CSTC circuit. Dysregulation of glutamatergic signaling within this circuit has been linked to OCD. Patients with OCD have been found to have an increase of glutamate in the CSF. As a result, medications that affect glutamate levels can be used to treat patients with OCD who do not respond to first-line agents. In patients already taking SRIs, augmentation of glutamate-modulating medications can reduce OCD symptoms. As an uncompetitive antagonist of the N-methyl-
Naderi et al10 evaluated amantadine as augmentative therapy to fluvoxamine for treating patients with moderate to severe OCD.
Study design
- This 12-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy and safety of amantadine as an augmentative agent to fluvoxamine in 106 patients age 18 to 60 with moderate to severe OCD.
- Participants met DSM-5 criteria for OCD and had a Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score >21. Participants were excluded if they had any substance dependence; an IQ <70; any other Axis I mental disorder; any serious cardiac, renal, or hepatic disease; had received psychotropic medications during the last 6 weeks, were pregnant or breastfeeding, or had rising liver transaminases to 3 times the upper limit of normal or higher.
- Participants received fluvoxamine 100 mg twice daily plus amantadine 100 mg/d, or fluvoxamine 100 mg twice daily plus placebo. All patients received fluvoxamine 100 mg/d for 28 days followed by 200 mg/d for the remainder of the trial.
- The primary outcome measure was difference in Y-BOCS total scores between the amantadine and placebo groups. The secondary outcome was the difference in Y-BOCS obsession and compulsion subscale scores.
Outcomes
- Patients who received amantadine augmentation experienced a significant reduction in Y-BOCS total score (P < .001) and obsession subscale score (P < .01).
- The amantadine group showed good tolerability and safety. There were no clinically significant adverse effects.
- Amantadine is an effective adjuvant to fluvoxamine for reducing OCD symptoms.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
2. Sharafkhah M, Aghakarim Alamdar M, Massoudifar A, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222- 233. doi:10.1097/YIC.0000000000000267
Although selective serotonin reuptake inhibitors (SSRIs) are considered a first-line treatment when teamed with CBT and antipsychotic augmentation, symptom resolution is not always achieved, and treatment resistance is a common problem. Sharafkhah et al11 compared the efficacy of ondansetron and granisetron augmentation specifically for patients with treatment-resistant OCD.
Study Design
- In this 18-week, randomized, double-blind, placebo-controlled study, 135 patients with treatment-resistant OCD who were previously treated with a combination of an SSRI and an antipsychotic received augmentation with ondansetron (n = 45, 4 mg/d), granisetron (n = 45, 2 mg/d), or placebo.
- Patients were rated using Y-BOCS every 2 weeks during phase I (intervention period), which lasted 14 weeks. After completing the intervention, patients were followed for 4 more weeks during phase II (discontinuation period).
- The aim of this study was to determine the safety, efficacy, and tolerability of ondansetron vs granisetron as augmentation for patients with treatment-resistant OCD. A secondary aim was to determine the rate of relapse of OCD symptoms after discontinuing ondansetron as compared with granisetron at 4 weeks after intervention.
Outcomes
- At Week 14, the reductions in Y-BOCS scores in the ondansetron, granisetron, and placebo groups were 41.5%, 39.7%, and 15.2%, respectively (P = .001). The reduction in Y-BOCS score in the ondansetron and granisetron groups was significantly greater than placebo at all phase I visits.
- Complete response was higher in the ondansetron group compared with the granisetron group (P = .041).
- Y-BOCS scores increased in both the ondansetron and granisetron groups during the discontinuation phase, but OCD symptoms were not significantly exacerbated.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
3. Modarresi A, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitorrefractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-120268
Increased glutamate levels in CSF, glutamatergic overactivity, and polymorphisms of genes coding the NMDA receptor have been shown to contribute to the occurrence of OCD. Memantine is a noncompetitive antagonist of the NMDA receptor. Various control trials have shown augmentation with memantine 5 mg/d to 20 mg/d significantly reduced symptom severity in patients with moderate to severe OCD. Modarresi et al12 evaluated memantine as a treatment option for patients with severe OCD who did not respond to SRI monotherapy.
Study design
- This 12-week, double-blind, randomized, placebo-controlled trial evaluated the efficacy of memantine augmentation in 32 patients age 18 to 40 who met DSM-5 criteria for OCD, had a Y-BOCS score ≥24, and no psychiatric comorbidity. Participants had not responded to ≥3 adequate trials (minimum 3 months) of SRI therapy, 1 of which was clomipramine.
- Individuals were excluded if they were undergoing CBT; had an additional anxiety disorder, mood disorder, or current drug or alcohol use disorder, or any systemic disorder; had a history of seizures; were pregnant or breastfeeding; or had a history of memantine use.
- Participants already receiving the maximum tolerated dose of an SRI received augmentation with memantine 20 mg/d or placebo.
- The primary outcome measure was change in Y-BOCS score from baseline. The secondary outcome was the number of individuals who achieved treatment response (defined as ≥35% reduction in Y-BOCS score).
Continue to: Outcomes...
Outcomes
- There was a statistically significant difference in Y-BOCS score in patients treated with memantine at Week 8 and Week 12 vs those who received placebo. By Week 8, 17.2% of patients in the memantine group showed a decrease in Y-BOCS score, compared with -0.8% patients in the placebo group. The difference became more significant by Week 12, with 40.9% in the memantine group showing a decrease in Y-BOCS score vs -0.3% in the placebo group. This resulted in 73.3% of patients achieving treatment response.
- Eight weeks of memantine augmentation was necessary to observe a significant improvement in OCD symptoms, and 12 weeks was needed for treatment response.
- The mean Y-BOCS total score decreased significantly in the memantine group from Week 4 to Week 8 (16.8%) and again from Week 8 to Week 12 (28.5%).
- The memantine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- Memantine augmentation in patients with severe OCD who do not respond to an SRI is effective and well-tolerated.
4. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407- 1414. doi:10.1177/0269881119878177
Studies have demonstrated the involvement of the amygdala, medial and lateral orbitofrontal cortex, and dorsal anterior cingulate cortex in OCD. Additionally, studies have also investigated the role of serotonin, dopamine, and glutamate system dysregulation in the pathology of OCD.
The 5-HT3 receptors are ligand-gated ion channels found in the prefrontal cortex, amygdala, and hippocampus. Studies of 5-HT3 receptor antagonists such as ondansetron and granisetron have shown beneficial results in augmentation with SSRIs for patients with OCD.11 Tropisetron, a 5-HT3 receptor antagonist, is highly lipophilic and able to cross the blood brain barrier. It also has dopamine-inhibiting properties that could have benefits in OCD management. Shalbafan et al13 evaluated the efficacy of tropisetron augmentation to fluvoxamine for patients with OCD.
Study design
- In a 10-week, randomized, double-blind, placebo-controlled, parallel-group trial, 108 individuals age 18 to 60 who met DSM-5 criteria for OCD and had a Y-BOCS score >21 received fluvoxamine plus tropisetron or fluvoxamine plus placebo. A total of 48 (44.4%) participants in each group completed the trial. Participants were evaluated using the Y-BOCS scale at baseline and at Week 4 and Week 10.
- The primary outcome was decrease in total Y-BOCS score from baseline to Week 10. The secondary outcome was the difference in change in Y-BOCS obsession and compulsion subscale scores between the groups.
Outcomes
- The Y-BOCS total score was not significantly different between the 2 groups (P = .975). Repeated measures analysis of variance determined a significant effect for time in both tropisetron and placebo groups (Greenhouse-Geisser F [2.72–2303.84] = 152.25, P < .001; and Greenhouse-Geisser F [1.37–1736.81] = 75.57, P < .001, respectively). At Week 10, 35 participants in the tropisetron group and 19 participants in the placebo group were complete responders.
- The baseline Y-BOCS obsession and compulsion subscales did not significantly differ between treatment groups.
Conclusion
- Compared with participants in the placebo group, those in the tropisetron group experienced a significantly greater reduction in OCD symptoms as measured by Y-BOCS score. More participants in the tropisetron group experienced complete response and remission.
- This study demonstrated that compared with placebo, when administered as augmentation with fluvoxamine, tropisetron can have beneficial effects for patients with OCD.
Continue to: Reference 5...
5. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a doubleblind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254- 262. doi:10.1097/YIC.0000000000000321
Nutraceuticals such as glycine, milk thistle, myoinositol, and serotonin (5-hydroxytryptophan) have been proposed as augmentation options for OCD. Yousefzadeh et al14 investigated the effectiveness of using 5-hydroxytryptophan in treating OCD.
Study design
- In a 12-week, randomized, double-blind study, 60 patients who met DSM-5 criteria for moderate to severe OCD (Y-BOCS score >21) were randomly assigned to receive fluoxetine plus 5-hydroxytryptophan 100 mg twice daily or fluoxetine plus placebo.
- All patients were administered fluoxetine 20 mg/d for the first 4 weeks of the study followed by fluoxetine 60 mg/d for the remainder of the trial.
- Symptoms were assessed using the Y-BOCS at baseline, Week 4, Week 8, and Week 12.
- The primary outcome measure was the difference between the 2 groups in change in Y-BOCS total score from baseline to the end of the trial. Secondary outcome measures were the differences in the Y-BOCS obsession and compulsion subscale scores from baseline to Week 12.
Outcomes
- Compared to the placebo group, the 5-hydroxytryptophan group experienced a statistically significant greater improvement in Y-BOCS total score from baseline to Week 8 (P = .002) and Week 12 (P < .001).
- General linear model repeated measure showed significant effects for time × treatment interaction on Y-BOCS total (F = 12.07, df = 2.29, P < .001), obsession subscale (F = 8.25, df = 1.91, P = .001), and compulsion subscale scores (F = 6.64, df = 2.01, P = .002).
- The 5-hydroxytryptophan group demonstrated higher partial and complete treatment response rates (P = .032 and P = .001, respectively) as determined by change in Y-BOCS total score.
- The 5-hydroxytryptophan group showed a significant improvement from baseline to Week 12 in Y-BOCS obsession subscale score (5.23 ± 2.33 vs 3.53 ± 2.13, P = .009).
- There was a significant change from baseline to the end of the trial in the Y-BOCS compulsion subscale score (3.88 ± 2.04 vs 2.30 ± 1.37, P = .002).
Conclusion
- This trial demonstrated the potential benefits of 5-hydroxytryptophan in combination with fluoxetine for patients with OCD.
6. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
Glutamatergic dysfunction has been identified as a potential cause of OCD. Studies have found elevated levels of glutamatergic transmission in the cortical-striatal-thalamic circuit of the brain and elevated glutamate concentration in the CSF in patients with OCD. Pregabalin has multiple mechanisms of action that inhibit the release of glutamate. Mowla et al15 evaluated pregabalin as an augmentation treatment for resistant OCD.
Study design
- This 12-week, double-blind, placebo-controlled clinical trial evaluated the efficacy of adjunctive pregabalin in 56 patients who met DSM-5 criteria for OCD and had not responded to ≥12 weeks of treatment with an adequate and stable dose of sertraline (baseline Y-BOCS score ≥18).
- Individuals who had other major psychiatric disorders, major medical problems, were pregnant, or had past substance or alcohol abuse were excluded.
- Participants were randomly assigned to receive sertraline plus pregabalin (n = 28) or sertraline plus placebo (n = 28). Mean sertraline dosage was 256.5 mg/d; range was 100 mg/d to 300 mg/d. Pregabalin was started at 75 mg/d and increased by 75 mg increments weekly. The mean dosage was 185.9 mg/d; range was 75 mg/d to 225 mg/d.
- The primary outcome measure was change in Y-BOCS score. A decrease >35% in Y-BOCS score was considered a significant response rate.
Outcomes
- There was a statistically significant decrease in Y-BOCS score in patients who received pregabalin. In the pregabalin group, 57.14% of patients (n = 16) showed a >35% decrease in Y-BOCS score compared with 7.14% of patients (n = 2) in the placebo group (P < .01).
- The pregabalin group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- In patients with treatment-resistant OCD who did not respond to sertraline monotherapy, augmentation with pregabalin significantly decreases Y-BOCS scores compared with placebo.
Continue to: Reference 7...
7. Zheng H, Jia F, Han H, et al. Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebocontrolled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro. 2018.12.010
Recent evidence suggests dysregulation of serotonin and dopamine in patients with OCD. Methylphenidate is a dopamine and norepinephrine inhibitor and releaser. A limited number of studies have suggested stimulants might be useful for OCD patients. Zheng et al16 conducted a pilot trial to determine whether methylphenidate augmentation may be of benefit in the management of outpatients with OCD.
Study design
- In an 8-week, double-blind, randomized, placebo-controlled trial, 44 patients (29 [66%] men, with a mean [SD] age of 24.7 [6]) with treatment-refractory OCD were randomized to receive fluvoxamine 250 mg/d plus methylphenidate extended-release (MPH-ER) 36 mg/d or fluvoxamine 250 mg/d plus placebo. The MPH-ER dose was 18 mg/d for the first 4 weeks and 36 mg/d for the rest of the trial.
- Biweekly assessments consisted of scores on the Y-BOCS, Hamilton Depression Rating Scale (HDRS), and Hamilton Anxiety Rating Scale (HAM-A).
- The primary outcomes were improvement in Y-BOCS score and the clinical response rate. Secondary outcomes included a change in score on the Y-BOCS subscales, HARS, and HAM-A. Data were analyzed with the intention-to-treat sample.
Outcomes
- Forty-one patients finished the trial. The baseline Y-BOCS total scores and subscale scores did not differ significantly between the 2 groups.
- Improvements in Y-BOCS total score and obsession subscale score were more prominent in the fluvoxamine plus MPH-ER group compared with the placebo group (P < .001).
- HDRS score decreased in both the placebo and MPH-ER groups. HAM-A scores decreased significantly in the MPH-ER plus fluvoxamine group compared with the placebo group.
Conclusion
- This study demonstrated that the combination of fluvoxamine and MPH-ER produces a higher and faster response rate than fluvoxamine plus placebo in patients with OCD.
8. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
Glutamate dysregulation is implicated in the pathogenesis of OCD. Glutamate-modulating agents have been used to treat OCD. Studies have shown L-carnosine has a neuroprotective role via its modulatory effect on glutamate. Arabzadeh et al17 evaluated the efficacy of L-carnosine as an adjuvant to fluvoxamine for treating OCD.
Study design
- This 10-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy of adjunctive L-carnosine in 40 patients age 18 to 60 who met DSM-5 criteria for OCD and had moderate to severe OCD (Y-BOCS score ≥21).
- Individuals with any other DSM-5 major psychiatric disorders, serious medical or neurologic illness, substance dependence (other than caffeine or nicotine), mental retardation (based on clinical judgment), were pregnant or breastfeeding, had any contraindication for the use of L‐carnosine or fluvoxamine, or received any psychotropic drugs in the previous 6 weeks were excluded.
- Participants received fluvoxamine 100 mg/d for the first 4 weeks and 200 mg/d for the next 6 weeks plus either L-carnosine 500 mg twice daily or placebo. This dosage of L-carnosine was chosen because previously it had been tolerated and effective.
- The primary outcome measure was difference in Y-BOCS total scores. Secondary outcomes were differences in Y-BOCS obsession and compulsion subscale scores and differences in change in score on Y-BOCS total and subscale scores from baseline.
Outcomes
- The L-carnosine group experienced a significant decrease in Y-BOCS total score (P < .001), obsession subscale score (P < .01), and compulsion subscale score (P < .01).
- The group that received fluvoxamine plus L-carnosine also experienced a more complete response (P = .03).
- The L-carnosine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- L-carnosine significantly reduces OCD symptoms when used as an adjuvant to fluvoxamine.
1. Kessler RC, Berglund P, Demler O, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
3. Eddy KT, Dutra L, Bradley, R, et al. A multidimensional meta-analysis of psychotherapy and pharmacotherapy for obsessive-compulsive disorder. Clin Psychol Rev. 2004;24(8):1011-1030.
4. Franklin ME, Foa EB. Treatment of obsessive compulsive disorder. Annu Rev Clin Psychol. 2011;7:229-243.
5. Koran LM, Hanna GL, Hollander E, et al. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Am J Psychiatry. 2007;164(7 Suppl):5-53.
6. Pittenger C, Bloch MH. Pharmacological treatment of obsessive-compulsive disorder. Psychiatr Clin North Am. 2014;37(3):375-391.
7. Pallanti S, Hollander E, Bienstock C, et al. Treatment non-response in OCD: methodological issues and operational definitions. Int J Neuropsychopharmacol. 2002;5(2):181-191.
8. Atmaca M. Treatment-refractory obsessive compulsive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:127-133.
9. Barth M, Kriston L, Klostermann S, et al. Efficacy of selective serotonin reuptake inhibitors and adverse events: meta-regression and mediation analysis of placebo-controlled trials. Br J Psychiatry. 2016;208(2):114-119.
10. NaderiS, Faghih H, Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/pcn.12803
11. SharafkhahM, Aghakarim Alamdar M, MassoudifarA, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222-233. doi:10.1097/YIC.0000000000000267
12. ModarresiA, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitor-refractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-12026
13. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407-1414. doi:10.1177/0269881119878177
14. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254-262. doi:10.1097/YIC.0000000000000321
15. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
16. Zheng H, Jia F, Han H, et al.Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebo-controlled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro.2018.12.010
17. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
Obsessive-compulsive disorder (OCD) is a chronic, debilitating neuropsychiatric disorder that affects 1% to 3% of the population worldwide.1,2 Together, serotonin reuptake inhibitors (SRIs) and cognitive-behavior therapy (CBT) are considered the first-line treatment for OCD.3 In children and adults, CBT is considered at least as effective as pharmacotherapy.4 Despite being an effective treatment, CBT continues to have barriers to its widespread use, including limited availability of trained CBT therapists, delayed clinical response, and high costs.5
Only approximately one-half of patients with OCD respond to SRI therapy, and a considerable percentage (30% to 40%) show significant residual symptoms even after multiple trials of SRIs.6-8 In addition, SRIs may have adverse effects (eg, sexual dysfunction, gastrointestinal symptoms) that impair patient adherence to these medications.9 Therefore, finding better treatment options is important for managing patients with OCD.
Augmentation strategies are recommended for patients who show partial response to SRI treatment or poor response to multiple SRIs. Augmentation typically includes incorporating additional medications with the primary drug with the goal of boosting the therapeutic efficacy of the primary drug. Typically, these additional medications have different mechanisms of action. However, there are no large-scale randomized controlled trials (RCTs) to inform treatment augmentation after first-line treatments for OCD produce suboptimal outcomes. The available evidence is predominantly based on small-scale RCTs, open-label trials, and case series.
In this article, we review the evidence for treatment augmentation strategies for OCD and summarize 8 studies that show promising results (Table10-17). We focus only on pharmacologic agents and do not include other biological interventions, such as repetitive transcranial magnetic stimulation over supplementary motor area, ablative neurosurgery, or deep brain stimulation.

Continue to: Reference 1...
1. Naderi S, Faghih H , Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessivecompulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/ pcn.12803
Numerous studies support the role of glutamate dysregulation in the pathophysiology of OCD. Cortico-striato-thalamo-cortical (CSTC) abnormalities play a major role in the pathophysiology of OCD as suggested by neuroimaging research studies that indicate glutamate is the fundamental neurotransmitter of the CSTC circuit. Dysregulation of glutamatergic signaling within this circuit has been linked to OCD. Patients with OCD have been found to have an increase of glutamate in the CSF. As a result, medications that affect glutamate levels can be used to treat patients with OCD who do not respond to first-line agents. In patients already taking SRIs, augmentation of glutamate-modulating medications can reduce OCD symptoms. As an uncompetitive antagonist of the N-methyl-
Naderi et al10 evaluated amantadine as augmentative therapy to fluvoxamine for treating patients with moderate to severe OCD.
Study design
- This 12-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy and safety of amantadine as an augmentative agent to fluvoxamine in 106 patients age 18 to 60 with moderate to severe OCD.
- Participants met DSM-5 criteria for OCD and had a Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score >21. Participants were excluded if they had any substance dependence; an IQ <70; any other Axis I mental disorder; any serious cardiac, renal, or hepatic disease; had received psychotropic medications during the last 6 weeks, were pregnant or breastfeeding, or had rising liver transaminases to 3 times the upper limit of normal or higher.
- Participants received fluvoxamine 100 mg twice daily plus amantadine 100 mg/d, or fluvoxamine 100 mg twice daily plus placebo. All patients received fluvoxamine 100 mg/d for 28 days followed by 200 mg/d for the remainder of the trial.
- The primary outcome measure was difference in Y-BOCS total scores between the amantadine and placebo groups. The secondary outcome was the difference in Y-BOCS obsession and compulsion subscale scores.
Outcomes
- Patients who received amantadine augmentation experienced a significant reduction in Y-BOCS total score (P < .001) and obsession subscale score (P < .01).
- The amantadine group showed good tolerability and safety. There were no clinically significant adverse effects.
- Amantadine is an effective adjuvant to fluvoxamine for reducing OCD symptoms.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
2. Sharafkhah M, Aghakarim Alamdar M, Massoudifar A, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222- 233. doi:10.1097/YIC.0000000000000267
Although selective serotonin reuptake inhibitors (SSRIs) are considered a first-line treatment when teamed with CBT and antipsychotic augmentation, symptom resolution is not always achieved, and treatment resistance is a common problem. Sharafkhah et al11 compared the efficacy of ondansetron and granisetron augmentation specifically for patients with treatment-resistant OCD.
Study Design
- In this 18-week, randomized, double-blind, placebo-controlled study, 135 patients with treatment-resistant OCD who were previously treated with a combination of an SSRI and an antipsychotic received augmentation with ondansetron (n = 45, 4 mg/d), granisetron (n = 45, 2 mg/d), or placebo.
- Patients were rated using Y-BOCS every 2 weeks during phase I (intervention period), which lasted 14 weeks. After completing the intervention, patients were followed for 4 more weeks during phase II (discontinuation period).
- The aim of this study was to determine the safety, efficacy, and tolerability of ondansetron vs granisetron as augmentation for patients with treatment-resistant OCD. A secondary aim was to determine the rate of relapse of OCD symptoms after discontinuing ondansetron as compared with granisetron at 4 weeks after intervention.
Outcomes
- At Week 14, the reductions in Y-BOCS scores in the ondansetron, granisetron, and placebo groups were 41.5%, 39.7%, and 15.2%, respectively (P = .001). The reduction in Y-BOCS score in the ondansetron and granisetron groups was significantly greater than placebo at all phase I visits.
- Complete response was higher in the ondansetron group compared with the granisetron group (P = .041).
- Y-BOCS scores increased in both the ondansetron and granisetron groups during the discontinuation phase, but OCD symptoms were not significantly exacerbated.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
3. Modarresi A, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitorrefractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-120268
Increased glutamate levels in CSF, glutamatergic overactivity, and polymorphisms of genes coding the NMDA receptor have been shown to contribute to the occurrence of OCD. Memantine is a noncompetitive antagonist of the NMDA receptor. Various control trials have shown augmentation with memantine 5 mg/d to 20 mg/d significantly reduced symptom severity in patients with moderate to severe OCD. Modarresi et al12 evaluated memantine as a treatment option for patients with severe OCD who did not respond to SRI monotherapy.
Study design
- This 12-week, double-blind, randomized, placebo-controlled trial evaluated the efficacy of memantine augmentation in 32 patients age 18 to 40 who met DSM-5 criteria for OCD, had a Y-BOCS score ≥24, and no psychiatric comorbidity. Participants had not responded to ≥3 adequate trials (minimum 3 months) of SRI therapy, 1 of which was clomipramine.
- Individuals were excluded if they were undergoing CBT; had an additional anxiety disorder, mood disorder, or current drug or alcohol use disorder, or any systemic disorder; had a history of seizures; were pregnant or breastfeeding; or had a history of memantine use.
- Participants already receiving the maximum tolerated dose of an SRI received augmentation with memantine 20 mg/d or placebo.
- The primary outcome measure was change in Y-BOCS score from baseline. The secondary outcome was the number of individuals who achieved treatment response (defined as ≥35% reduction in Y-BOCS score).
Continue to: Outcomes...
Outcomes
- There was a statistically significant difference in Y-BOCS score in patients treated with memantine at Week 8 and Week 12 vs those who received placebo. By Week 8, 17.2% of patients in the memantine group showed a decrease in Y-BOCS score, compared with -0.8% patients in the placebo group. The difference became more significant by Week 12, with 40.9% in the memantine group showing a decrease in Y-BOCS score vs -0.3% in the placebo group. This resulted in 73.3% of patients achieving treatment response.
- Eight weeks of memantine augmentation was necessary to observe a significant improvement in OCD symptoms, and 12 weeks was needed for treatment response.
- The mean Y-BOCS total score decreased significantly in the memantine group from Week 4 to Week 8 (16.8%) and again from Week 8 to Week 12 (28.5%).
- The memantine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- Memantine augmentation in patients with severe OCD who do not respond to an SRI is effective and well-tolerated.
4. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407- 1414. doi:10.1177/0269881119878177
Studies have demonstrated the involvement of the amygdala, medial and lateral orbitofrontal cortex, and dorsal anterior cingulate cortex in OCD. Additionally, studies have also investigated the role of serotonin, dopamine, and glutamate system dysregulation in the pathology of OCD.
The 5-HT3 receptors are ligand-gated ion channels found in the prefrontal cortex, amygdala, and hippocampus. Studies of 5-HT3 receptor antagonists such as ondansetron and granisetron have shown beneficial results in augmentation with SSRIs for patients with OCD.11 Tropisetron, a 5-HT3 receptor antagonist, is highly lipophilic and able to cross the blood brain barrier. It also has dopamine-inhibiting properties that could have benefits in OCD management. Shalbafan et al13 evaluated the efficacy of tropisetron augmentation to fluvoxamine for patients with OCD.
Study design
- In a 10-week, randomized, double-blind, placebo-controlled, parallel-group trial, 108 individuals age 18 to 60 who met DSM-5 criteria for OCD and had a Y-BOCS score >21 received fluvoxamine plus tropisetron or fluvoxamine plus placebo. A total of 48 (44.4%) participants in each group completed the trial. Participants were evaluated using the Y-BOCS scale at baseline and at Week 4 and Week 10.
- The primary outcome was decrease in total Y-BOCS score from baseline to Week 10. The secondary outcome was the difference in change in Y-BOCS obsession and compulsion subscale scores between the groups.
Outcomes
- The Y-BOCS total score was not significantly different between the 2 groups (P = .975). Repeated measures analysis of variance determined a significant effect for time in both tropisetron and placebo groups (Greenhouse-Geisser F [2.72–2303.84] = 152.25, P < .001; and Greenhouse-Geisser F [1.37–1736.81] = 75.57, P < .001, respectively). At Week 10, 35 participants in the tropisetron group and 19 participants in the placebo group were complete responders.
- The baseline Y-BOCS obsession and compulsion subscales did not significantly differ between treatment groups.
Conclusion
- Compared with participants in the placebo group, those in the tropisetron group experienced a significantly greater reduction in OCD symptoms as measured by Y-BOCS score. More participants in the tropisetron group experienced complete response and remission.
- This study demonstrated that compared with placebo, when administered as augmentation with fluvoxamine, tropisetron can have beneficial effects for patients with OCD.
Continue to: Reference 5...
5. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a doubleblind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254- 262. doi:10.1097/YIC.0000000000000321
Nutraceuticals such as glycine, milk thistle, myoinositol, and serotonin (5-hydroxytryptophan) have been proposed as augmentation options for OCD. Yousefzadeh et al14 investigated the effectiveness of using 5-hydroxytryptophan in treating OCD.
Study design
- In a 12-week, randomized, double-blind study, 60 patients who met DSM-5 criteria for moderate to severe OCD (Y-BOCS score >21) were randomly assigned to receive fluoxetine plus 5-hydroxytryptophan 100 mg twice daily or fluoxetine plus placebo.
- All patients were administered fluoxetine 20 mg/d for the first 4 weeks of the study followed by fluoxetine 60 mg/d for the remainder of the trial.
- Symptoms were assessed using the Y-BOCS at baseline, Week 4, Week 8, and Week 12.
- The primary outcome measure was the difference between the 2 groups in change in Y-BOCS total score from baseline to the end of the trial. Secondary outcome measures were the differences in the Y-BOCS obsession and compulsion subscale scores from baseline to Week 12.
Outcomes
- Compared to the placebo group, the 5-hydroxytryptophan group experienced a statistically significant greater improvement in Y-BOCS total score from baseline to Week 8 (P = .002) and Week 12 (P < .001).
- General linear model repeated measure showed significant effects for time × treatment interaction on Y-BOCS total (F = 12.07, df = 2.29, P < .001), obsession subscale (F = 8.25, df = 1.91, P = .001), and compulsion subscale scores (F = 6.64, df = 2.01, P = .002).
- The 5-hydroxytryptophan group demonstrated higher partial and complete treatment response rates (P = .032 and P = .001, respectively) as determined by change in Y-BOCS total score.
- The 5-hydroxytryptophan group showed a significant improvement from baseline to Week 12 in Y-BOCS obsession subscale score (5.23 ± 2.33 vs 3.53 ± 2.13, P = .009).
- There was a significant change from baseline to the end of the trial in the Y-BOCS compulsion subscale score (3.88 ± 2.04 vs 2.30 ± 1.37, P = .002).
Conclusion
- This trial demonstrated the potential benefits of 5-hydroxytryptophan in combination with fluoxetine for patients with OCD.
6. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
Glutamatergic dysfunction has been identified as a potential cause of OCD. Studies have found elevated levels of glutamatergic transmission in the cortical-striatal-thalamic circuit of the brain and elevated glutamate concentration in the CSF in patients with OCD. Pregabalin has multiple mechanisms of action that inhibit the release of glutamate. Mowla et al15 evaluated pregabalin as an augmentation treatment for resistant OCD.
Study design
- This 12-week, double-blind, placebo-controlled clinical trial evaluated the efficacy of adjunctive pregabalin in 56 patients who met DSM-5 criteria for OCD and had not responded to ≥12 weeks of treatment with an adequate and stable dose of sertraline (baseline Y-BOCS score ≥18).
- Individuals who had other major psychiatric disorders, major medical problems, were pregnant, or had past substance or alcohol abuse were excluded.
- Participants were randomly assigned to receive sertraline plus pregabalin (n = 28) or sertraline plus placebo (n = 28). Mean sertraline dosage was 256.5 mg/d; range was 100 mg/d to 300 mg/d. Pregabalin was started at 75 mg/d and increased by 75 mg increments weekly. The mean dosage was 185.9 mg/d; range was 75 mg/d to 225 mg/d.
- The primary outcome measure was change in Y-BOCS score. A decrease >35% in Y-BOCS score was considered a significant response rate.
Outcomes
- There was a statistically significant decrease in Y-BOCS score in patients who received pregabalin. In the pregabalin group, 57.14% of patients (n = 16) showed a >35% decrease in Y-BOCS score compared with 7.14% of patients (n = 2) in the placebo group (P < .01).
- The pregabalin group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- In patients with treatment-resistant OCD who did not respond to sertraline monotherapy, augmentation with pregabalin significantly decreases Y-BOCS scores compared with placebo.
Continue to: Reference 7...
7. Zheng H, Jia F, Han H, et al. Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebocontrolled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro. 2018.12.010
Recent evidence suggests dysregulation of serotonin and dopamine in patients with OCD. Methylphenidate is a dopamine and norepinephrine inhibitor and releaser. A limited number of studies have suggested stimulants might be useful for OCD patients. Zheng et al16 conducted a pilot trial to determine whether methylphenidate augmentation may be of benefit in the management of outpatients with OCD.
Study design
- In an 8-week, double-blind, randomized, placebo-controlled trial, 44 patients (29 [66%] men, with a mean [SD] age of 24.7 [6]) with treatment-refractory OCD were randomized to receive fluvoxamine 250 mg/d plus methylphenidate extended-release (MPH-ER) 36 mg/d or fluvoxamine 250 mg/d plus placebo. The MPH-ER dose was 18 mg/d for the first 4 weeks and 36 mg/d for the rest of the trial.
- Biweekly assessments consisted of scores on the Y-BOCS, Hamilton Depression Rating Scale (HDRS), and Hamilton Anxiety Rating Scale (HAM-A).
- The primary outcomes were improvement in Y-BOCS score and the clinical response rate. Secondary outcomes included a change in score on the Y-BOCS subscales, HARS, and HAM-A. Data were analyzed with the intention-to-treat sample.
Outcomes
- Forty-one patients finished the trial. The baseline Y-BOCS total scores and subscale scores did not differ significantly between the 2 groups.
- Improvements in Y-BOCS total score and obsession subscale score were more prominent in the fluvoxamine plus MPH-ER group compared with the placebo group (P < .001).
- HDRS score decreased in both the placebo and MPH-ER groups. HAM-A scores decreased significantly in the MPH-ER plus fluvoxamine group compared with the placebo group.
Conclusion
- This study demonstrated that the combination of fluvoxamine and MPH-ER produces a higher and faster response rate than fluvoxamine plus placebo in patients with OCD.
8. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
Glutamate dysregulation is implicated in the pathogenesis of OCD. Glutamate-modulating agents have been used to treat OCD. Studies have shown L-carnosine has a neuroprotective role via its modulatory effect on glutamate. Arabzadeh et al17 evaluated the efficacy of L-carnosine as an adjuvant to fluvoxamine for treating OCD.
Study design
- This 10-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy of adjunctive L-carnosine in 40 patients age 18 to 60 who met DSM-5 criteria for OCD and had moderate to severe OCD (Y-BOCS score ≥21).
- Individuals with any other DSM-5 major psychiatric disorders, serious medical or neurologic illness, substance dependence (other than caffeine or nicotine), mental retardation (based on clinical judgment), were pregnant or breastfeeding, had any contraindication for the use of L‐carnosine or fluvoxamine, or received any psychotropic drugs in the previous 6 weeks were excluded.
- Participants received fluvoxamine 100 mg/d for the first 4 weeks and 200 mg/d for the next 6 weeks plus either L-carnosine 500 mg twice daily or placebo. This dosage of L-carnosine was chosen because previously it had been tolerated and effective.
- The primary outcome measure was difference in Y-BOCS total scores. Secondary outcomes were differences in Y-BOCS obsession and compulsion subscale scores and differences in change in score on Y-BOCS total and subscale scores from baseline.
Outcomes
- The L-carnosine group experienced a significant decrease in Y-BOCS total score (P < .001), obsession subscale score (P < .01), and compulsion subscale score (P < .01).
- The group that received fluvoxamine plus L-carnosine also experienced a more complete response (P = .03).
- The L-carnosine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- L-carnosine significantly reduces OCD symptoms when used as an adjuvant to fluvoxamine.
Obsessive-compulsive disorder (OCD) is a chronic, debilitating neuropsychiatric disorder that affects 1% to 3% of the population worldwide.1,2 Together, serotonin reuptake inhibitors (SRIs) and cognitive-behavior therapy (CBT) are considered the first-line treatment for OCD.3 In children and adults, CBT is considered at least as effective as pharmacotherapy.4 Despite being an effective treatment, CBT continues to have barriers to its widespread use, including limited availability of trained CBT therapists, delayed clinical response, and high costs.5
Only approximately one-half of patients with OCD respond to SRI therapy, and a considerable percentage (30% to 40%) show significant residual symptoms even after multiple trials of SRIs.6-8 In addition, SRIs may have adverse effects (eg, sexual dysfunction, gastrointestinal symptoms) that impair patient adherence to these medications.9 Therefore, finding better treatment options is important for managing patients with OCD.
Augmentation strategies are recommended for patients who show partial response to SRI treatment or poor response to multiple SRIs. Augmentation typically includes incorporating additional medications with the primary drug with the goal of boosting the therapeutic efficacy of the primary drug. Typically, these additional medications have different mechanisms of action. However, there are no large-scale randomized controlled trials (RCTs) to inform treatment augmentation after first-line treatments for OCD produce suboptimal outcomes. The available evidence is predominantly based on small-scale RCTs, open-label trials, and case series.
In this article, we review the evidence for treatment augmentation strategies for OCD and summarize 8 studies that show promising results (Table10-17). We focus only on pharmacologic agents and do not include other biological interventions, such as repetitive transcranial magnetic stimulation over supplementary motor area, ablative neurosurgery, or deep brain stimulation.

Continue to: Reference 1...
1. Naderi S, Faghih H , Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessivecompulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/ pcn.12803
Numerous studies support the role of glutamate dysregulation in the pathophysiology of OCD. Cortico-striato-thalamo-cortical (CSTC) abnormalities play a major role in the pathophysiology of OCD as suggested by neuroimaging research studies that indicate glutamate is the fundamental neurotransmitter of the CSTC circuit. Dysregulation of glutamatergic signaling within this circuit has been linked to OCD. Patients with OCD have been found to have an increase of glutamate in the CSF. As a result, medications that affect glutamate levels can be used to treat patients with OCD who do not respond to first-line agents. In patients already taking SRIs, augmentation of glutamate-modulating medications can reduce OCD symptoms. As an uncompetitive antagonist of the N-methyl-
Naderi et al10 evaluated amantadine as augmentative therapy to fluvoxamine for treating patients with moderate to severe OCD.
Study design
- This 12-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy and safety of amantadine as an augmentative agent to fluvoxamine in 106 patients age 18 to 60 with moderate to severe OCD.
- Participants met DSM-5 criteria for OCD and had a Yale-Brown Obsessive Compulsive Scale (Y-BOCS) score >21. Participants were excluded if they had any substance dependence; an IQ <70; any other Axis I mental disorder; any serious cardiac, renal, or hepatic disease; had received psychotropic medications during the last 6 weeks, were pregnant or breastfeeding, or had rising liver transaminases to 3 times the upper limit of normal or higher.
- Participants received fluvoxamine 100 mg twice daily plus amantadine 100 mg/d, or fluvoxamine 100 mg twice daily plus placebo. All patients received fluvoxamine 100 mg/d for 28 days followed by 200 mg/d for the remainder of the trial.
- The primary outcome measure was difference in Y-BOCS total scores between the amantadine and placebo groups. The secondary outcome was the difference in Y-BOCS obsession and compulsion subscale scores.
Outcomes
- Patients who received amantadine augmentation experienced a significant reduction in Y-BOCS total score (P < .001) and obsession subscale score (P < .01).
- The amantadine group showed good tolerability and safety. There were no clinically significant adverse effects.
- Amantadine is an effective adjuvant to fluvoxamine for reducing OCD symptoms.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
2. Sharafkhah M, Aghakarim Alamdar M, Massoudifar A, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222- 233. doi:10.1097/YIC.0000000000000267
Although selective serotonin reuptake inhibitors (SSRIs) are considered a first-line treatment when teamed with CBT and antipsychotic augmentation, symptom resolution is not always achieved, and treatment resistance is a common problem. Sharafkhah et al11 compared the efficacy of ondansetron and granisetron augmentation specifically for patients with treatment-resistant OCD.
Study Design
- In this 18-week, randomized, double-blind, placebo-controlled study, 135 patients with treatment-resistant OCD who were previously treated with a combination of an SSRI and an antipsychotic received augmentation with ondansetron (n = 45, 4 mg/d), granisetron (n = 45, 2 mg/d), or placebo.
- Patients were rated using Y-BOCS every 2 weeks during phase I (intervention period), which lasted 14 weeks. After completing the intervention, patients were followed for 4 more weeks during phase II (discontinuation period).
- The aim of this study was to determine the safety, efficacy, and tolerability of ondansetron vs granisetron as augmentation for patients with treatment-resistant OCD. A secondary aim was to determine the rate of relapse of OCD symptoms after discontinuing ondansetron as compared with granisetron at 4 weeks after intervention.
Outcomes
- At Week 14, the reductions in Y-BOCS scores in the ondansetron, granisetron, and placebo groups were 41.5%, 39.7%, and 15.2%, respectively (P = .001). The reduction in Y-BOCS score in the ondansetron and granisetron groups was significantly greater than placebo at all phase I visits.
- Complete response was higher in the ondansetron group compared with the granisetron group (P = .041).
- Y-BOCS scores increased in both the ondansetron and granisetron groups during the discontinuation phase, but OCD symptoms were not significantly exacerbated.
Conclusion
- Ondansetron and granisetron can be beneficial as an augmentation strategy for patients with treatment-resistant OCD.
3. Modarresi A, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitorrefractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-120268
Increased glutamate levels in CSF, glutamatergic overactivity, and polymorphisms of genes coding the NMDA receptor have been shown to contribute to the occurrence of OCD. Memantine is a noncompetitive antagonist of the NMDA receptor. Various control trials have shown augmentation with memantine 5 mg/d to 20 mg/d significantly reduced symptom severity in patients with moderate to severe OCD. Modarresi et al12 evaluated memantine as a treatment option for patients with severe OCD who did not respond to SRI monotherapy.
Study design
- This 12-week, double-blind, randomized, placebo-controlled trial evaluated the efficacy of memantine augmentation in 32 patients age 18 to 40 who met DSM-5 criteria for OCD, had a Y-BOCS score ≥24, and no psychiatric comorbidity. Participants had not responded to ≥3 adequate trials (minimum 3 months) of SRI therapy, 1 of which was clomipramine.
- Individuals were excluded if they were undergoing CBT; had an additional anxiety disorder, mood disorder, or current drug or alcohol use disorder, or any systemic disorder; had a history of seizures; were pregnant or breastfeeding; or had a history of memantine use.
- Participants already receiving the maximum tolerated dose of an SRI received augmentation with memantine 20 mg/d or placebo.
- The primary outcome measure was change in Y-BOCS score from baseline. The secondary outcome was the number of individuals who achieved treatment response (defined as ≥35% reduction in Y-BOCS score).
Continue to: Outcomes...
Outcomes
- There was a statistically significant difference in Y-BOCS score in patients treated with memantine at Week 8 and Week 12 vs those who received placebo. By Week 8, 17.2% of patients in the memantine group showed a decrease in Y-BOCS score, compared with -0.8% patients in the placebo group. The difference became more significant by Week 12, with 40.9% in the memantine group showing a decrease in Y-BOCS score vs -0.3% in the placebo group. This resulted in 73.3% of patients achieving treatment response.
- Eight weeks of memantine augmentation was necessary to observe a significant improvement in OCD symptoms, and 12 weeks was needed for treatment response.
- The mean Y-BOCS total score decreased significantly in the memantine group from Week 4 to Week 8 (16.8%) and again from Week 8 to Week 12 (28.5%).
- The memantine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- Memantine augmentation in patients with severe OCD who do not respond to an SRI is effective and well-tolerated.
4. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407- 1414. doi:10.1177/0269881119878177
Studies have demonstrated the involvement of the amygdala, medial and lateral orbitofrontal cortex, and dorsal anterior cingulate cortex in OCD. Additionally, studies have also investigated the role of serotonin, dopamine, and glutamate system dysregulation in the pathology of OCD.
The 5-HT3 receptors are ligand-gated ion channels found in the prefrontal cortex, amygdala, and hippocampus. Studies of 5-HT3 receptor antagonists such as ondansetron and granisetron have shown beneficial results in augmentation with SSRIs for patients with OCD.11 Tropisetron, a 5-HT3 receptor antagonist, is highly lipophilic and able to cross the blood brain barrier. It also has dopamine-inhibiting properties that could have benefits in OCD management. Shalbafan et al13 evaluated the efficacy of tropisetron augmentation to fluvoxamine for patients with OCD.
Study design
- In a 10-week, randomized, double-blind, placebo-controlled, parallel-group trial, 108 individuals age 18 to 60 who met DSM-5 criteria for OCD and had a Y-BOCS score >21 received fluvoxamine plus tropisetron or fluvoxamine plus placebo. A total of 48 (44.4%) participants in each group completed the trial. Participants were evaluated using the Y-BOCS scale at baseline and at Week 4 and Week 10.
- The primary outcome was decrease in total Y-BOCS score from baseline to Week 10. The secondary outcome was the difference in change in Y-BOCS obsession and compulsion subscale scores between the groups.
Outcomes
- The Y-BOCS total score was not significantly different between the 2 groups (P = .975). Repeated measures analysis of variance determined a significant effect for time in both tropisetron and placebo groups (Greenhouse-Geisser F [2.72–2303.84] = 152.25, P < .001; and Greenhouse-Geisser F [1.37–1736.81] = 75.57, P < .001, respectively). At Week 10, 35 participants in the tropisetron group and 19 participants in the placebo group were complete responders.
- The baseline Y-BOCS obsession and compulsion subscales did not significantly differ between treatment groups.
Conclusion
- Compared with participants in the placebo group, those in the tropisetron group experienced a significantly greater reduction in OCD symptoms as measured by Y-BOCS score. More participants in the tropisetron group experienced complete response and remission.
- This study demonstrated that compared with placebo, when administered as augmentation with fluvoxamine, tropisetron can have beneficial effects for patients with OCD.
Continue to: Reference 5...
5. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a doubleblind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254- 262. doi:10.1097/YIC.0000000000000321
Nutraceuticals such as glycine, milk thistle, myoinositol, and serotonin (5-hydroxytryptophan) have been proposed as augmentation options for OCD. Yousefzadeh et al14 investigated the effectiveness of using 5-hydroxytryptophan in treating OCD.
Study design
- In a 12-week, randomized, double-blind study, 60 patients who met DSM-5 criteria for moderate to severe OCD (Y-BOCS score >21) were randomly assigned to receive fluoxetine plus 5-hydroxytryptophan 100 mg twice daily or fluoxetine plus placebo.
- All patients were administered fluoxetine 20 mg/d for the first 4 weeks of the study followed by fluoxetine 60 mg/d for the remainder of the trial.
- Symptoms were assessed using the Y-BOCS at baseline, Week 4, Week 8, and Week 12.
- The primary outcome measure was the difference between the 2 groups in change in Y-BOCS total score from baseline to the end of the trial. Secondary outcome measures were the differences in the Y-BOCS obsession and compulsion subscale scores from baseline to Week 12.
Outcomes
- Compared to the placebo group, the 5-hydroxytryptophan group experienced a statistically significant greater improvement in Y-BOCS total score from baseline to Week 8 (P = .002) and Week 12 (P < .001).
- General linear model repeated measure showed significant effects for time × treatment interaction on Y-BOCS total (F = 12.07, df = 2.29, P < .001), obsession subscale (F = 8.25, df = 1.91, P = .001), and compulsion subscale scores (F = 6.64, df = 2.01, P = .002).
- The 5-hydroxytryptophan group demonstrated higher partial and complete treatment response rates (P = .032 and P = .001, respectively) as determined by change in Y-BOCS total score.
- The 5-hydroxytryptophan group showed a significant improvement from baseline to Week 12 in Y-BOCS obsession subscale score (5.23 ± 2.33 vs 3.53 ± 2.13, P = .009).
- There was a significant change from baseline to the end of the trial in the Y-BOCS compulsion subscale score (3.88 ± 2.04 vs 2.30 ± 1.37, P = .002).
Conclusion
- This trial demonstrated the potential benefits of 5-hydroxytryptophan in combination with fluoxetine for patients with OCD.
6. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
Glutamatergic dysfunction has been identified as a potential cause of OCD. Studies have found elevated levels of glutamatergic transmission in the cortical-striatal-thalamic circuit of the brain and elevated glutamate concentration in the CSF in patients with OCD. Pregabalin has multiple mechanisms of action that inhibit the release of glutamate. Mowla et al15 evaluated pregabalin as an augmentation treatment for resistant OCD.
Study design
- This 12-week, double-blind, placebo-controlled clinical trial evaluated the efficacy of adjunctive pregabalin in 56 patients who met DSM-5 criteria for OCD and had not responded to ≥12 weeks of treatment with an adequate and stable dose of sertraline (baseline Y-BOCS score ≥18).
- Individuals who had other major psychiatric disorders, major medical problems, were pregnant, or had past substance or alcohol abuse were excluded.
- Participants were randomly assigned to receive sertraline plus pregabalin (n = 28) or sertraline plus placebo (n = 28). Mean sertraline dosage was 256.5 mg/d; range was 100 mg/d to 300 mg/d. Pregabalin was started at 75 mg/d and increased by 75 mg increments weekly. The mean dosage was 185.9 mg/d; range was 75 mg/d to 225 mg/d.
- The primary outcome measure was change in Y-BOCS score. A decrease >35% in Y-BOCS score was considered a significant response rate.
Outcomes
- There was a statistically significant decrease in Y-BOCS score in patients who received pregabalin. In the pregabalin group, 57.14% of patients (n = 16) showed a >35% decrease in Y-BOCS score compared with 7.14% of patients (n = 2) in the placebo group (P < .01).
- The pregabalin group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- In patients with treatment-resistant OCD who did not respond to sertraline monotherapy, augmentation with pregabalin significantly decreases Y-BOCS scores compared with placebo.
Continue to: Reference 7...
7. Zheng H, Jia F, Han H, et al. Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebocontrolled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro. 2018.12.010
Recent evidence suggests dysregulation of serotonin and dopamine in patients with OCD. Methylphenidate is a dopamine and norepinephrine inhibitor and releaser. A limited number of studies have suggested stimulants might be useful for OCD patients. Zheng et al16 conducted a pilot trial to determine whether methylphenidate augmentation may be of benefit in the management of outpatients with OCD.
Study design
- In an 8-week, double-blind, randomized, placebo-controlled trial, 44 patients (29 [66%] men, with a mean [SD] age of 24.7 [6]) with treatment-refractory OCD were randomized to receive fluvoxamine 250 mg/d plus methylphenidate extended-release (MPH-ER) 36 mg/d or fluvoxamine 250 mg/d plus placebo. The MPH-ER dose was 18 mg/d for the first 4 weeks and 36 mg/d for the rest of the trial.
- Biweekly assessments consisted of scores on the Y-BOCS, Hamilton Depression Rating Scale (HDRS), and Hamilton Anxiety Rating Scale (HAM-A).
- The primary outcomes were improvement in Y-BOCS score and the clinical response rate. Secondary outcomes included a change in score on the Y-BOCS subscales, HARS, and HAM-A. Data were analyzed with the intention-to-treat sample.
Outcomes
- Forty-one patients finished the trial. The baseline Y-BOCS total scores and subscale scores did not differ significantly between the 2 groups.
- Improvements in Y-BOCS total score and obsession subscale score were more prominent in the fluvoxamine plus MPH-ER group compared with the placebo group (P < .001).
- HDRS score decreased in both the placebo and MPH-ER groups. HAM-A scores decreased significantly in the MPH-ER plus fluvoxamine group compared with the placebo group.
Conclusion
- This study demonstrated that the combination of fluvoxamine and MPH-ER produces a higher and faster response rate than fluvoxamine plus placebo in patients with OCD.
8. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
Glutamate dysregulation is implicated in the pathogenesis of OCD. Glutamate-modulating agents have been used to treat OCD. Studies have shown L-carnosine has a neuroprotective role via its modulatory effect on glutamate. Arabzadeh et al17 evaluated the efficacy of L-carnosine as an adjuvant to fluvoxamine for treating OCD.
Study design
- This 10-week, randomized, double-blind, placebo-controlled trial evaluated the efficacy of adjunctive L-carnosine in 40 patients age 18 to 60 who met DSM-5 criteria for OCD and had moderate to severe OCD (Y-BOCS score ≥21).
- Individuals with any other DSM-5 major psychiatric disorders, serious medical or neurologic illness, substance dependence (other than caffeine or nicotine), mental retardation (based on clinical judgment), were pregnant or breastfeeding, had any contraindication for the use of L‐carnosine or fluvoxamine, or received any psychotropic drugs in the previous 6 weeks were excluded.
- Participants received fluvoxamine 100 mg/d for the first 4 weeks and 200 mg/d for the next 6 weeks plus either L-carnosine 500 mg twice daily or placebo. This dosage of L-carnosine was chosen because previously it had been tolerated and effective.
- The primary outcome measure was difference in Y-BOCS total scores. Secondary outcomes were differences in Y-BOCS obsession and compulsion subscale scores and differences in change in score on Y-BOCS total and subscale scores from baseline.
Outcomes
- The L-carnosine group experienced a significant decrease in Y-BOCS total score (P < .001), obsession subscale score (P < .01), and compulsion subscale score (P < .01).
- The group that received fluvoxamine plus L-carnosine also experienced a more complete response (P = .03).
- The L-carnosine group showed good tolerability and safety. There were no clinically significant adverse effects.
Conclusion
- L-carnosine significantly reduces OCD symptoms when used as an adjuvant to fluvoxamine.
1. Kessler RC, Berglund P, Demler O, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
3. Eddy KT, Dutra L, Bradley, R, et al. A multidimensional meta-analysis of psychotherapy and pharmacotherapy for obsessive-compulsive disorder. Clin Psychol Rev. 2004;24(8):1011-1030.
4. Franklin ME, Foa EB. Treatment of obsessive compulsive disorder. Annu Rev Clin Psychol. 2011;7:229-243.
5. Koran LM, Hanna GL, Hollander E, et al. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Am J Psychiatry. 2007;164(7 Suppl):5-53.
6. Pittenger C, Bloch MH. Pharmacological treatment of obsessive-compulsive disorder. Psychiatr Clin North Am. 2014;37(3):375-391.
7. Pallanti S, Hollander E, Bienstock C, et al. Treatment non-response in OCD: methodological issues and operational definitions. Int J Neuropsychopharmacol. 2002;5(2):181-191.
8. Atmaca M. Treatment-refractory obsessive compulsive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:127-133.
9. Barth M, Kriston L, Klostermann S, et al. Efficacy of selective serotonin reuptake inhibitors and adverse events: meta-regression and mediation analysis of placebo-controlled trials. Br J Psychiatry. 2016;208(2):114-119.
10. NaderiS, Faghih H, Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/pcn.12803
11. SharafkhahM, Aghakarim Alamdar M, MassoudifarA, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222-233. doi:10.1097/YIC.0000000000000267
12. ModarresiA, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitor-refractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-12026
13. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407-1414. doi:10.1177/0269881119878177
14. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254-262. doi:10.1097/YIC.0000000000000321
15. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
16. Zheng H, Jia F, Han H, et al.Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebo-controlled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro.2018.12.010
17. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
1. Kessler RC, Berglund P, Demler O, et al. Lifetime prevalence and age-of-onset distributions of DSM-IV disorders in the National Comorbidity Survey Replication. Arch Gen Psychiatry. 2005;62(6):593-602.
2. Ruscio AM, Stein DJ, Chiu WT, et al. The epidemiology of obsessive-compulsive disorder in the National Comorbidity Survey Replication. Mol Psychiatry. 2010;15(1):53-63.
3. Eddy KT, Dutra L, Bradley, R, et al. A multidimensional meta-analysis of psychotherapy and pharmacotherapy for obsessive-compulsive disorder. Clin Psychol Rev. 2004;24(8):1011-1030.
4. Franklin ME, Foa EB. Treatment of obsessive compulsive disorder. Annu Rev Clin Psychol. 2011;7:229-243.
5. Koran LM, Hanna GL, Hollander E, et al. Practice guideline for the treatment of patients with obsessive-compulsive disorder. Am J Psychiatry. 2007;164(7 Suppl):5-53.
6. Pittenger C, Bloch MH. Pharmacological treatment of obsessive-compulsive disorder. Psychiatr Clin North Am. 2014;37(3):375-391.
7. Pallanti S, Hollander E, Bienstock C, et al. Treatment non-response in OCD: methodological issues and operational definitions. Int J Neuropsychopharmacol. 2002;5(2):181-191.
8. Atmaca M. Treatment-refractory obsessive compulsive disorder. Prog Neuropsychopharmacol Biol Psychiatry. 2016;70:127-133.
9. Barth M, Kriston L, Klostermann S, et al. Efficacy of selective serotonin reuptake inhibitors and adverse events: meta-regression and mediation analysis of placebo-controlled trials. Br J Psychiatry. 2016;208(2):114-119.
10. NaderiS, Faghih H, Aqamolaei A, et al. Amantadine as adjuvant therapy in the treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Psychiatry Clin Neurosci. 2019;73(4):169-174. doi:10.1111/pcn.12803
11. SharafkhahM, Aghakarim Alamdar M, MassoudifarA, et al. Comparing the efficacy of ondansetron and granisetron augmentation in treatment-resistant obsessive-compulsive disorder: a randomized double-blind placebo-controlled study. Int Clin Psychopharmacol. 2019;34(5):222-233. doi:10.1097/YIC.0000000000000267
12. ModarresiA, Sayyah M, Razooghi S, et al. Memantine augmentation improves symptoms in serotonin reuptake inhibitor-refractory obsessive-compulsive disorder: a randomized controlled trial. Pharmacopsychiatry. 2018;51(6):263-269. doi:10.1055/s-0043-12026
13. Shalbafan M, Malekpour F, Tadayon Najafabadi B, et al. Fluvoxamine combination therapy with tropisetron for obsessive-compulsive disorder patients: a placebo-controlled, randomized clinical trial. J Psychopharmacol. 2019;33(11):1407-1414. doi:10.1177/0269881119878177
14. Yousefzadeh F, Sahebolzamani E, Sadri A, et al. 5-Hydroxytryptophan as adjuvant therapy in treatment of moderate to severe obsessive-compulsive disorder: a double-blind randomized trial with placebo control. Int Clin Psychopharmacol. 2020;35(5):254-262. doi:10.1097/YIC.0000000000000321
15. Mowla A, Ghaedsharaf M. Pregabalin augmentation for resistant obsessive-compulsive disorder: a double-blind placebo-controlled clinical trial. CNS Spectr. 2020;25(4):552-556. doi:10.1017/S1092852919001500
16. Zheng H, Jia F, Han H, et al.Combined fluvoxamine and extended-release methylphenidate improved treatment response compared to fluvoxamine alone in patients with treatment-refractory obsessive-compulsive disorder: a randomized double-blind, placebo-controlled study. Eur Neuropsychopharmacol. 2019;29(3):397-404. doi:10.1016/j.euroneuro.2018.12.010
17. Arabzadeh S, Shahhossenie M, Mesgarpour B, et al. L-carnosine as an adjuvant to fluvoxamine in treatment of obsessive compulsive disorder: a randomized double-blind study. Hum Psychopharmacol. 2017;32(4). doi:10.1002/hup.2584
Managing a COVID-19–positive psychiatric patient on a medical unit
With the COVID-19 pandemic turning the world on its head, we have seen more first-episode psychotic breaks and quick deterioration in previously stable patients. Early in the pandemic, care was particularly complicated for psychiatric patients who had been infected with the virus. Many of these patients required immediate psychiatric hospitalization. At that time, many community hospital psychiatric inpatient units did not have the capacity, staffing, or infrastructure to safely admit such patients, so they needed to be managed on a medical unit. Here, I discuss the case of a COVID-19–positive woman with psychiatric illness who we managed while she was in quarantine on a medical unit.
Case report
Early in the COVID-19 pandemic, Ms. B, a 35-year-old teacher with a history of depression, was evaluated in the emergency department for bizarre behavior and paranoid delusions regarding her family. Initial laboratory and imaging testing was negative for any potential medical causes of her psychiatric symptoms. Psychiatric hospitalization was recommended, but before Ms. B could be transferred to the psychiatric unit, she tested positive for COVID-19. At that time, our community hospital did not have a designated wing on our psychiatric unit for patients infected with COVID-19. Thus, Ms. B was admitted to the medical floor, where she was quarantined in her room. She would need to remain asymptomatic and test negative for COVID-19 before she could be transferred to the psychiatric unit.
Upon arriving at the medical unit, Ms. B was hostile and uncooperative. She frequently attempted to leave her room and required restraints throughout the day. Our consultation-liaison (CL) team was consulted to assist in managing her. During the initial interview, we noticed that she had covered all 4 walls of her room with papers filled with handwritten notes. Ms. B had cut her gown to expose her stomach and legs. She had pressured speech, tangential thinking, and was religiously preoccupied. She denied any visual and auditory hallucinations, but her persecutory delusions involving her family persisted. We believed that her signs and symptoms were consistent with a manic episode from underlying, and likely undiagnosed, bipolar I disorder that was precipitated by her COVID-19 infection.
We first addressed Ms. B’s and the staff’s safety by transferring her to a larger room with a vestibule at the end of the hallway so she had more room to walk and minimal exposure to the stimuli of the medical unit. We initiated one-on-one observation to redirect her and prevent elopement. We incentivized her cooperation with staff by providing her with paper, pencils, reading material, and phone privileges. We started oral risperidone 2 mg twice daily and lorazepam 2 mg 3 times daily for short-term behavioral control and acute treatment of her symptoms, with the goal of deferring additional treatment decisions to the inpatient psychiatry team after she was transferred to the psychiatric unit. Ms. B’s agitation and impulsivity improved. She began participating with the medical team and was eventually transferred out of our medical unit to a psychiatric unit at a different facility.
COVID-19 and psychiatric illness: Clinical concerns
While infection from COVID-19 and widespread social distancing of the general population have been linked to depression and anxiety, manic and psychotic symptoms secondary to the COVID-19 pandemic have not been well described. The association between influenza infection and psychosis has been reported since the Spanish Flu pandemic,1 but there is limited data on the association between COVID-19 and psychosis. A review of 14 studies found that 0.9% to 4% of people exposed to a virus during an epidemic or pandemic develop psychosis or psychotic symptoms.1 Psychosis was associated with viral exposure, treatments used to manage the infection (steroid therapy), and psychosocial stress. This study also found that treatment with low doses of antipsychotic medication—notably aripiprazole—seemed to have been effective.1
Nonetheless, it is important to keep in mind a thorough differential diagnosis and rule out any potential organic etiologies in a COVID-19–positive patient who presents with psychiatric symptoms.2 For Ms. B, we began by ruling out drug-induced psychosis and electrolyte imbalance, and obtained brain imaging to rule out malignancy. We considered an interictal behavior syndrome of temporal lobe epilepsy, a neuropsychiatric disorder characterized by alterations in sexual behavior, religiosity, and extensive and compulsive writing and drawing.3 Neurology was consulted to evaluate the patient and possibly use EEG to detect interictal spikes, a tall task given the patient’s restlessness and paranoia. Ultimately, we determined the patient was most likely exhibiting symptoms of previously undetected bipolar disorder.
Managing patients with psychiatric illness on a medical floor during a pandemic such as COVID-19 requires the psychiatrist to truly serve as a consultant and liaison between the patient and the treatment team.4 Clinical management should address both infection control and psychiatric symptoms.5 We visited with Ms. B frequently, provided psychoeducation, engaged her in treatment, and updated her on the treatment plan.
As the medical world continues to adjust to treating patients during the pandemic, CL psychiatrists may be tasked with managing patients with acute psychiatric illness on the medical unit while they await transfer to a psychiatric unit. A creative, multifaceted, and team-based approach is key to ensure effective care and safety for all involved.
1. Brown E, Gray R, Lo Monaco S, et al. The potential impact of COVID-19 on psychosis: a rapid review of contemporary epidemic and pandemic research. Schizophr Res. 2020;222:79-87. doi:10.1016/j.schres.2020.05.005
2. Byrne P. Managing the acute psychotic episode. BMJ. 2007;334(7595):686-692. doi:10.1136/bmj.39148.668160.80
3. Waxman SG, Geschwind N. The interictal behavior syndrome of temporal lobe epilepsy. Arch Gen Psychiatry. 1975;32(12):1580-1586. doi:10.1001/archpsyc.1975.01760300118011
4. Stern TA, Freudenreich O, Smith FA, et al. Psychotic patients. In: Massachusetts General Hospital: Handbook of General Hospital Psychiatry. Mosby; 1997:109-121.
5. Deshpande S, Livingstone A. First-onset psychosis in older adults: social isolation influence during COVID pandemic—a UK case series. Progress in Neurology and Psychiatry. 2021;25(1):14-18. doi:10.1002/pnp.692
With the COVID-19 pandemic turning the world on its head, we have seen more first-episode psychotic breaks and quick deterioration in previously stable patients. Early in the pandemic, care was particularly complicated for psychiatric patients who had been infected with the virus. Many of these patients required immediate psychiatric hospitalization. At that time, many community hospital psychiatric inpatient units did not have the capacity, staffing, or infrastructure to safely admit such patients, so they needed to be managed on a medical unit. Here, I discuss the case of a COVID-19–positive woman with psychiatric illness who we managed while she was in quarantine on a medical unit.
Case report
Early in the COVID-19 pandemic, Ms. B, a 35-year-old teacher with a history of depression, was evaluated in the emergency department for bizarre behavior and paranoid delusions regarding her family. Initial laboratory and imaging testing was negative for any potential medical causes of her psychiatric symptoms. Psychiatric hospitalization was recommended, but before Ms. B could be transferred to the psychiatric unit, she tested positive for COVID-19. At that time, our community hospital did not have a designated wing on our psychiatric unit for patients infected with COVID-19. Thus, Ms. B was admitted to the medical floor, where she was quarantined in her room. She would need to remain asymptomatic and test negative for COVID-19 before she could be transferred to the psychiatric unit.
Upon arriving at the medical unit, Ms. B was hostile and uncooperative. She frequently attempted to leave her room and required restraints throughout the day. Our consultation-liaison (CL) team was consulted to assist in managing her. During the initial interview, we noticed that she had covered all 4 walls of her room with papers filled with handwritten notes. Ms. B had cut her gown to expose her stomach and legs. She had pressured speech, tangential thinking, and was religiously preoccupied. She denied any visual and auditory hallucinations, but her persecutory delusions involving her family persisted. We believed that her signs and symptoms were consistent with a manic episode from underlying, and likely undiagnosed, bipolar I disorder that was precipitated by her COVID-19 infection.
We first addressed Ms. B’s and the staff’s safety by transferring her to a larger room with a vestibule at the end of the hallway so she had more room to walk and minimal exposure to the stimuli of the medical unit. We initiated one-on-one observation to redirect her and prevent elopement. We incentivized her cooperation with staff by providing her with paper, pencils, reading material, and phone privileges. We started oral risperidone 2 mg twice daily and lorazepam 2 mg 3 times daily for short-term behavioral control and acute treatment of her symptoms, with the goal of deferring additional treatment decisions to the inpatient psychiatry team after she was transferred to the psychiatric unit. Ms. B’s agitation and impulsivity improved. She began participating with the medical team and was eventually transferred out of our medical unit to a psychiatric unit at a different facility.
COVID-19 and psychiatric illness: Clinical concerns
While infection from COVID-19 and widespread social distancing of the general population have been linked to depression and anxiety, manic and psychotic symptoms secondary to the COVID-19 pandemic have not been well described. The association between influenza infection and psychosis has been reported since the Spanish Flu pandemic,1 but there is limited data on the association between COVID-19 and psychosis. A review of 14 studies found that 0.9% to 4% of people exposed to a virus during an epidemic or pandemic develop psychosis or psychotic symptoms.1 Psychosis was associated with viral exposure, treatments used to manage the infection (steroid therapy), and psychosocial stress. This study also found that treatment with low doses of antipsychotic medication—notably aripiprazole—seemed to have been effective.1
Nonetheless, it is important to keep in mind a thorough differential diagnosis and rule out any potential organic etiologies in a COVID-19–positive patient who presents with psychiatric symptoms.2 For Ms. B, we began by ruling out drug-induced psychosis and electrolyte imbalance, and obtained brain imaging to rule out malignancy. We considered an interictal behavior syndrome of temporal lobe epilepsy, a neuropsychiatric disorder characterized by alterations in sexual behavior, religiosity, and extensive and compulsive writing and drawing.3 Neurology was consulted to evaluate the patient and possibly use EEG to detect interictal spikes, a tall task given the patient’s restlessness and paranoia. Ultimately, we determined the patient was most likely exhibiting symptoms of previously undetected bipolar disorder.
Managing patients with psychiatric illness on a medical floor during a pandemic such as COVID-19 requires the psychiatrist to truly serve as a consultant and liaison between the patient and the treatment team.4 Clinical management should address both infection control and psychiatric symptoms.5 We visited with Ms. B frequently, provided psychoeducation, engaged her in treatment, and updated her on the treatment plan.
As the medical world continues to adjust to treating patients during the pandemic, CL psychiatrists may be tasked with managing patients with acute psychiatric illness on the medical unit while they await transfer to a psychiatric unit. A creative, multifaceted, and team-based approach is key to ensure effective care and safety for all involved.
With the COVID-19 pandemic turning the world on its head, we have seen more first-episode psychotic breaks and quick deterioration in previously stable patients. Early in the pandemic, care was particularly complicated for psychiatric patients who had been infected with the virus. Many of these patients required immediate psychiatric hospitalization. At that time, many community hospital psychiatric inpatient units did not have the capacity, staffing, or infrastructure to safely admit such patients, so they needed to be managed on a medical unit. Here, I discuss the case of a COVID-19–positive woman with psychiatric illness who we managed while she was in quarantine on a medical unit.
Case report
Early in the COVID-19 pandemic, Ms. B, a 35-year-old teacher with a history of depression, was evaluated in the emergency department for bizarre behavior and paranoid delusions regarding her family. Initial laboratory and imaging testing was negative for any potential medical causes of her psychiatric symptoms. Psychiatric hospitalization was recommended, but before Ms. B could be transferred to the psychiatric unit, she tested positive for COVID-19. At that time, our community hospital did not have a designated wing on our psychiatric unit for patients infected with COVID-19. Thus, Ms. B was admitted to the medical floor, where she was quarantined in her room. She would need to remain asymptomatic and test negative for COVID-19 before she could be transferred to the psychiatric unit.
Upon arriving at the medical unit, Ms. B was hostile and uncooperative. She frequently attempted to leave her room and required restraints throughout the day. Our consultation-liaison (CL) team was consulted to assist in managing her. During the initial interview, we noticed that she had covered all 4 walls of her room with papers filled with handwritten notes. Ms. B had cut her gown to expose her stomach and legs. She had pressured speech, tangential thinking, and was religiously preoccupied. She denied any visual and auditory hallucinations, but her persecutory delusions involving her family persisted. We believed that her signs and symptoms were consistent with a manic episode from underlying, and likely undiagnosed, bipolar I disorder that was precipitated by her COVID-19 infection.
We first addressed Ms. B’s and the staff’s safety by transferring her to a larger room with a vestibule at the end of the hallway so she had more room to walk and minimal exposure to the stimuli of the medical unit. We initiated one-on-one observation to redirect her and prevent elopement. We incentivized her cooperation with staff by providing her with paper, pencils, reading material, and phone privileges. We started oral risperidone 2 mg twice daily and lorazepam 2 mg 3 times daily for short-term behavioral control and acute treatment of her symptoms, with the goal of deferring additional treatment decisions to the inpatient psychiatry team after she was transferred to the psychiatric unit. Ms. B’s agitation and impulsivity improved. She began participating with the medical team and was eventually transferred out of our medical unit to a psychiatric unit at a different facility.
COVID-19 and psychiatric illness: Clinical concerns
While infection from COVID-19 and widespread social distancing of the general population have been linked to depression and anxiety, manic and psychotic symptoms secondary to the COVID-19 pandemic have not been well described. The association between influenza infection and psychosis has been reported since the Spanish Flu pandemic,1 but there is limited data on the association between COVID-19 and psychosis. A review of 14 studies found that 0.9% to 4% of people exposed to a virus during an epidemic or pandemic develop psychosis or psychotic symptoms.1 Psychosis was associated with viral exposure, treatments used to manage the infection (steroid therapy), and psychosocial stress. This study also found that treatment with low doses of antipsychotic medication—notably aripiprazole—seemed to have been effective.1
Nonetheless, it is important to keep in mind a thorough differential diagnosis and rule out any potential organic etiologies in a COVID-19–positive patient who presents with psychiatric symptoms.2 For Ms. B, we began by ruling out drug-induced psychosis and electrolyte imbalance, and obtained brain imaging to rule out malignancy. We considered an interictal behavior syndrome of temporal lobe epilepsy, a neuropsychiatric disorder characterized by alterations in sexual behavior, religiosity, and extensive and compulsive writing and drawing.3 Neurology was consulted to evaluate the patient and possibly use EEG to detect interictal spikes, a tall task given the patient’s restlessness and paranoia. Ultimately, we determined the patient was most likely exhibiting symptoms of previously undetected bipolar disorder.
Managing patients with psychiatric illness on a medical floor during a pandemic such as COVID-19 requires the psychiatrist to truly serve as a consultant and liaison between the patient and the treatment team.4 Clinical management should address both infection control and psychiatric symptoms.5 We visited with Ms. B frequently, provided psychoeducation, engaged her in treatment, and updated her on the treatment plan.
As the medical world continues to adjust to treating patients during the pandemic, CL psychiatrists may be tasked with managing patients with acute psychiatric illness on the medical unit while they await transfer to a psychiatric unit. A creative, multifaceted, and team-based approach is key to ensure effective care and safety for all involved.
1. Brown E, Gray R, Lo Monaco S, et al. The potential impact of COVID-19 on psychosis: a rapid review of contemporary epidemic and pandemic research. Schizophr Res. 2020;222:79-87. doi:10.1016/j.schres.2020.05.005
2. Byrne P. Managing the acute psychotic episode. BMJ. 2007;334(7595):686-692. doi:10.1136/bmj.39148.668160.80
3. Waxman SG, Geschwind N. The interictal behavior syndrome of temporal lobe epilepsy. Arch Gen Psychiatry. 1975;32(12):1580-1586. doi:10.1001/archpsyc.1975.01760300118011
4. Stern TA, Freudenreich O, Smith FA, et al. Psychotic patients. In: Massachusetts General Hospital: Handbook of General Hospital Psychiatry. Mosby; 1997:109-121.
5. Deshpande S, Livingstone A. First-onset psychosis in older adults: social isolation influence during COVID pandemic—a UK case series. Progress in Neurology and Psychiatry. 2021;25(1):14-18. doi:10.1002/pnp.692
1. Brown E, Gray R, Lo Monaco S, et al. The potential impact of COVID-19 on psychosis: a rapid review of contemporary epidemic and pandemic research. Schizophr Res. 2020;222:79-87. doi:10.1016/j.schres.2020.05.005
2. Byrne P. Managing the acute psychotic episode. BMJ. 2007;334(7595):686-692. doi:10.1136/bmj.39148.668160.80
3. Waxman SG, Geschwind N. The interictal behavior syndrome of temporal lobe epilepsy. Arch Gen Psychiatry. 1975;32(12):1580-1586. doi:10.1001/archpsyc.1975.01760300118011
4. Stern TA, Freudenreich O, Smith FA, et al. Psychotic patients. In: Massachusetts General Hospital: Handbook of General Hospital Psychiatry. Mosby; 1997:109-121.
5. Deshpande S, Livingstone A. First-onset psychosis in older adults: social isolation influence during COVID pandemic—a UK case series. Progress in Neurology and Psychiatry. 2021;25(1):14-18. doi:10.1002/pnp.692
Celebrating our colleagues
several of whom I am privileged to work with on a daily basis. We also welcome the newest members of AGA’s Governing Board, Maria T. Abreu, MD, AGAF, who is an outstanding leader and representative of a much larger group of volunteer members who work tirelessly to advance AGA’s initiatives to enhance the clinical practice of gastroenterology and improve patient outcomes. The nominating committee also appointed the following slate of councilors, which is subject to membership vote: Kim Barrett, PhD, AGAF; Lawrence Kosinski, MD, MBA, AGAF; and Sheryl Pfeil, MD, AGAF.
This month’s issue also highlights two newly-developed clinical risk-prediction tools – one designed to assist clinicians in predicting alcoholic hepatitis mortality, and another designed to identify inflammatory bowel disease (IBD) patients at high-risk of developing venous thromboembolism (VTE) post-hospitalization. While no prediction model is perfect, these tools can positively impact clinical decision-making and contribute to improved patient outcomes. We also include recommendations on managing IBD in older patients, and report on a study suggesting an increase in late-stage cancer diagnoses in the wake of the COVID-19 pandemic. AGA’s new clinical guideline on systemic therapy for hepatocellular carcinoma and Clinical Practice Update on non-invasive colorectal cancer screening also are featured. Finally, in this month’s Practice Management Toolbox, Dr. Feuerstein, Dr. Sofia, Dr. Guha, and Dr. Streett offer timely recommendations regarding how to overcome existing barriers to achieve high-value IBD care.
Megan A. Adams, MD, JD, MSc
Editor in Chief
several of whom I am privileged to work with on a daily basis. We also welcome the newest members of AGA’s Governing Board, Maria T. Abreu, MD, AGAF, who is an outstanding leader and representative of a much larger group of volunteer members who work tirelessly to advance AGA’s initiatives to enhance the clinical practice of gastroenterology and improve patient outcomes. The nominating committee also appointed the following slate of councilors, which is subject to membership vote: Kim Barrett, PhD, AGAF; Lawrence Kosinski, MD, MBA, AGAF; and Sheryl Pfeil, MD, AGAF.
This month’s issue also highlights two newly-developed clinical risk-prediction tools – one designed to assist clinicians in predicting alcoholic hepatitis mortality, and another designed to identify inflammatory bowel disease (IBD) patients at high-risk of developing venous thromboembolism (VTE) post-hospitalization. While no prediction model is perfect, these tools can positively impact clinical decision-making and contribute to improved patient outcomes. We also include recommendations on managing IBD in older patients, and report on a study suggesting an increase in late-stage cancer diagnoses in the wake of the COVID-19 pandemic. AGA’s new clinical guideline on systemic therapy for hepatocellular carcinoma and Clinical Practice Update on non-invasive colorectal cancer screening also are featured. Finally, in this month’s Practice Management Toolbox, Dr. Feuerstein, Dr. Sofia, Dr. Guha, and Dr. Streett offer timely recommendations regarding how to overcome existing barriers to achieve high-value IBD care.
Megan A. Adams, MD, JD, MSc
Editor in Chief
several of whom I am privileged to work with on a daily basis. We also welcome the newest members of AGA’s Governing Board, Maria T. Abreu, MD, AGAF, who is an outstanding leader and representative of a much larger group of volunteer members who work tirelessly to advance AGA’s initiatives to enhance the clinical practice of gastroenterology and improve patient outcomes. The nominating committee also appointed the following slate of councilors, which is subject to membership vote: Kim Barrett, PhD, AGAF; Lawrence Kosinski, MD, MBA, AGAF; and Sheryl Pfeil, MD, AGAF.
This month’s issue also highlights two newly-developed clinical risk-prediction tools – one designed to assist clinicians in predicting alcoholic hepatitis mortality, and another designed to identify inflammatory bowel disease (IBD) patients at high-risk of developing venous thromboembolism (VTE) post-hospitalization. While no prediction model is perfect, these tools can positively impact clinical decision-making and contribute to improved patient outcomes. We also include recommendations on managing IBD in older patients, and report on a study suggesting an increase in late-stage cancer diagnoses in the wake of the COVID-19 pandemic. AGA’s new clinical guideline on systemic therapy for hepatocellular carcinoma and Clinical Practice Update on non-invasive colorectal cancer screening also are featured. Finally, in this month’s Practice Management Toolbox, Dr. Feuerstein, Dr. Sofia, Dr. Guha, and Dr. Streett offer timely recommendations regarding how to overcome existing barriers to achieve high-value IBD care.
Megan A. Adams, MD, JD, MSc
Editor in Chief
How and why the language of medicine must change
The United States has never achieved a single high standard of medical care equity for all of its people, and the trend line does not appear favorable. The closest we have reached is basic Medicare (Parts A and B), military medicine, the Veterans Health Administration, and large nonprofit groups like Kaiser Permanente. It seems that the nature of we individualistic Americans is to always try to seek an advantage.
But even achieving equity in medical care would not ensure equity in health. The social determinants of health (income level, education, politics, government, geography, neighborhood, country of origin, language spoken, literacy, gender, and yes – race and ethnicity) have far more influence on health equity than does medical care.
Narratives can both reflect and influence culture. Considering the harmful effects of the current political divisiveness in the United States, the timing is ideal for our three leading medical and health education organizations – the American Medical Association, the Association of American Medical Colleges (AAMC), and the Centers for Disease Control and Prevention – to publish a definitive position paper called “Advancing Health Equity: A Guide to Language, Narrative and Concepts.”
What’s in a word?
According to William Shakespeare, “A rose by any other name would smell as sweet” (Romeo and Juliet). Maybe. But if the word used were “thorn” or “thistle,” it just would not be the same.
Words comprise language and wield enormous power with human beings. Wars are fought over geographic boundaries often defined by the language spoken by the people: think 2022, Russian-speaking Ukrainians. Think Winston Churchill’s massive 1,500-page “A History of the English-Speaking Peoples.” Think about the political power of French in Quebec, Canada.
Thus, it should be no surprise that words, acronyms, and abbreviations become rallying cries for political activists of all stripes: PC, January 6, Woke, 1619, BLM, Critical Race Theory, 1776, Remember Pearl Harbor, Remember the Alamo, the Civil War or the War Between the States, the War for Southern Independence, the War of Northern Aggression, the War of the Rebellion, or simply “The Lost Cause.” How about Realpolitik?
Is “medical language” the language of the people or of the profession? Physicians must understand each other, and physicians also must communicate clearly with patients using words that convey neutral meanings and don’t interfere with objective understanding. Medical editors prefer the brevity of one or a few words to clearly convey meaning.
I consider this document from the AMA and AAMC to be both profound and profoundly important for the healing professions. The contributors frequently use words like “humility” as they describe their efforts and products, knowing full well that they (and their organizations) stand to be figuratively torn limb from limb by a host of critics – or worse, ignored and marginalized.
Part 1 of the Health Equity Guide is titled “Language for promoting health equity.”(the reader is referred to the Health Equity Guide for the reasoning and explanations for all).
Part 2 of the Health Equity Guide is called “Why narratives matter.” It includes features of dominant narratives; a substantial section on the narrative of race and the narrative of individualism; the purpose of a health equity–based narrative; how to change the narrative; and how to see and think critically through dialogue.
Part 3 of the Health Equity Guide is a glossary of 138 key terms such as “class,” “discrimination,” “gender dysphoria,” “non-White,” “racial capitalism,” and “structural competency.”
The CDC also has a toolkit for inclusive communication, the “Health Equity Guiding Principles for Inclusive Communication.”
The substantive message of the Health Equity Guide could affect what you say, write, and do (even how you think) every day as well as how those with whom you interact view you. It can affect the entire communication milieu in which you live, whether or not you like it. Read it seriously, as though your professional life depended on it. It may.
Dr. Lundberg is consulting professor of health research policy and pathology at Stanford (Calif.) University. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The United States has never achieved a single high standard of medical care equity for all of its people, and the trend line does not appear favorable. The closest we have reached is basic Medicare (Parts A and B), military medicine, the Veterans Health Administration, and large nonprofit groups like Kaiser Permanente. It seems that the nature of we individualistic Americans is to always try to seek an advantage.
But even achieving equity in medical care would not ensure equity in health. The social determinants of health (income level, education, politics, government, geography, neighborhood, country of origin, language spoken, literacy, gender, and yes – race and ethnicity) have far more influence on health equity than does medical care.
Narratives can both reflect and influence culture. Considering the harmful effects of the current political divisiveness in the United States, the timing is ideal for our three leading medical and health education organizations – the American Medical Association, the Association of American Medical Colleges (AAMC), and the Centers for Disease Control and Prevention – to publish a definitive position paper called “Advancing Health Equity: A Guide to Language, Narrative and Concepts.”
What’s in a word?
According to William Shakespeare, “A rose by any other name would smell as sweet” (Romeo and Juliet). Maybe. But if the word used were “thorn” or “thistle,” it just would not be the same.
Words comprise language and wield enormous power with human beings. Wars are fought over geographic boundaries often defined by the language spoken by the people: think 2022, Russian-speaking Ukrainians. Think Winston Churchill’s massive 1,500-page “A History of the English-Speaking Peoples.” Think about the political power of French in Quebec, Canada.
Thus, it should be no surprise that words, acronyms, and abbreviations become rallying cries for political activists of all stripes: PC, January 6, Woke, 1619, BLM, Critical Race Theory, 1776, Remember Pearl Harbor, Remember the Alamo, the Civil War or the War Between the States, the War for Southern Independence, the War of Northern Aggression, the War of the Rebellion, or simply “The Lost Cause.” How about Realpolitik?
Is “medical language” the language of the people or of the profession? Physicians must understand each other, and physicians also must communicate clearly with patients using words that convey neutral meanings and don’t interfere with objective understanding. Medical editors prefer the brevity of one or a few words to clearly convey meaning.
I consider this document from the AMA and AAMC to be both profound and profoundly important for the healing professions. The contributors frequently use words like “humility” as they describe their efforts and products, knowing full well that they (and their organizations) stand to be figuratively torn limb from limb by a host of critics – or worse, ignored and marginalized.
Part 1 of the Health Equity Guide is titled “Language for promoting health equity.”(the reader is referred to the Health Equity Guide for the reasoning and explanations for all).

Part 2 of the Health Equity Guide is called “Why narratives matter.” It includes features of dominant narratives; a substantial section on the narrative of race and the narrative of individualism; the purpose of a health equity–based narrative; how to change the narrative; and how to see and think critically through dialogue.
Part 3 of the Health Equity Guide is a glossary of 138 key terms such as “class,” “discrimination,” “gender dysphoria,” “non-White,” “racial capitalism,” and “structural competency.”
The CDC also has a toolkit for inclusive communication, the “Health Equity Guiding Principles for Inclusive Communication.”
The substantive message of the Health Equity Guide could affect what you say, write, and do (even how you think) every day as well as how those with whom you interact view you. It can affect the entire communication milieu in which you live, whether or not you like it. Read it seriously, as though your professional life depended on it. It may.
Dr. Lundberg is consulting professor of health research policy and pathology at Stanford (Calif.) University. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
The United States has never achieved a single high standard of medical care equity for all of its people, and the trend line does not appear favorable. The closest we have reached is basic Medicare (Parts A and B), military medicine, the Veterans Health Administration, and large nonprofit groups like Kaiser Permanente. It seems that the nature of we individualistic Americans is to always try to seek an advantage.
But even achieving equity in medical care would not ensure equity in health. The social determinants of health (income level, education, politics, government, geography, neighborhood, country of origin, language spoken, literacy, gender, and yes – race and ethnicity) have far more influence on health equity than does medical care.
Narratives can both reflect and influence culture. Considering the harmful effects of the current political divisiveness in the United States, the timing is ideal for our three leading medical and health education organizations – the American Medical Association, the Association of American Medical Colleges (AAMC), and the Centers for Disease Control and Prevention – to publish a definitive position paper called “Advancing Health Equity: A Guide to Language, Narrative and Concepts.”
What’s in a word?
According to William Shakespeare, “A rose by any other name would smell as sweet” (Romeo and Juliet). Maybe. But if the word used were “thorn” or “thistle,” it just would not be the same.
Words comprise language and wield enormous power with human beings. Wars are fought over geographic boundaries often defined by the language spoken by the people: think 2022, Russian-speaking Ukrainians. Think Winston Churchill’s massive 1,500-page “A History of the English-Speaking Peoples.” Think about the political power of French in Quebec, Canada.
Thus, it should be no surprise that words, acronyms, and abbreviations become rallying cries for political activists of all stripes: PC, January 6, Woke, 1619, BLM, Critical Race Theory, 1776, Remember Pearl Harbor, Remember the Alamo, the Civil War or the War Between the States, the War for Southern Independence, the War of Northern Aggression, the War of the Rebellion, or simply “The Lost Cause.” How about Realpolitik?
Is “medical language” the language of the people or of the profession? Physicians must understand each other, and physicians also must communicate clearly with patients using words that convey neutral meanings and don’t interfere with objective understanding. Medical editors prefer the brevity of one or a few words to clearly convey meaning.
I consider this document from the AMA and AAMC to be both profound and profoundly important for the healing professions. The contributors frequently use words like “humility” as they describe their efforts and products, knowing full well that they (and their organizations) stand to be figuratively torn limb from limb by a host of critics – or worse, ignored and marginalized.
Part 1 of the Health Equity Guide is titled “Language for promoting health equity.”(the reader is referred to the Health Equity Guide for the reasoning and explanations for all).

Part 2 of the Health Equity Guide is called “Why narratives matter.” It includes features of dominant narratives; a substantial section on the narrative of race and the narrative of individualism; the purpose of a health equity–based narrative; how to change the narrative; and how to see and think critically through dialogue.
Part 3 of the Health Equity Guide is a glossary of 138 key terms such as “class,” “discrimination,” “gender dysphoria,” “non-White,” “racial capitalism,” and “structural competency.”
The CDC also has a toolkit for inclusive communication, the “Health Equity Guiding Principles for Inclusive Communication.”
The substantive message of the Health Equity Guide could affect what you say, write, and do (even how you think) every day as well as how those with whom you interact view you. It can affect the entire communication milieu in which you live, whether or not you like it. Read it seriously, as though your professional life depended on it. It may.
Dr. Lundberg is consulting professor of health research policy and pathology at Stanford (Calif.) University. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Microdosing psychedelics: Untapped potential in psychiatry?
In her month-long memoir, A Really Good Day: How Microdosing Made a Mega Difference in My Mood, My Marriage, and My Life (Knopf, 2017), author Ayelet Waldman turns herself into a one-woman experiment.
Over a single month she takes one-tenth of a recreational dose of LSD every third day. She plots her emotions, her productivity, and her pain along the way. Ms. Waldman obtains the LSD in a single vial, enough for 10 doses, from a researcher, who is retiring. What she’s looking for, she tells the reader, is a really good day – something that has been elusive in her turbulent life.
Although psychedelics remain illegal for both recreational and therapeutic use, they are increasingly being studied at academic centers, and there is hope that they will offer something that our traditional medications might not. However, these are not “micro” doses, but full doses of psychedelic agents that induce clinically-monitored “trips” in order to treat conditions such as depression, anorexia nervosa, or for smoking cessation, to name just a few.
Yet
Because these drugs are illegal under most circumstances, many of the studies involve surveys of users in their natural environments who are already microdosing in an uncontrolled manner. In a 2019 study published in PLOS One, Vince Polito and Richard Stevenson, from Macquarie University, Sydney, gave daily surveys of psychological functioning to 98 microdosers over 6 weeks. Several participants were excluded for using doses that were too high or for concurrent use of other illicit substances.
Whereas the authors found that many people claimed to have positive experiences, there was an increase in neuroticism in some of the subjects. There was no control group and no uniformity to what the subject claimed to be ingesting with regard to dose, frequency, substance, or verification of the chemical content.
University of Chicago neuroscientist Harriet De Wit, PhD, leads one of the few laboratories that conducts controlled, double-blind studies looking at microdosing LSD.
“With microdosing there are expectations, and we don’t know if it’s the expectation or the agent that is making a difference,” she explained. And when asked who in her experience is experimenting with microdosing psychedelics, she expounded “Everybody under the sun!”
Dr. De Wit notes that people microdose to increase their creativity, productivity, focus, and energy, to heighten their spiritual awareness, improve empathy and social relational skills, and to improve their mood – all purported benefits of low-dose psychedelics.
Her group published a study in Addiction Biology, in which 39 subjects were administered low doses of LSD four times over 2 weeks. To address the issues of expectation, the subjects were not told they were participating in a study of hallucinogens specifically but were instead given a list of pharmaceuticals in different classes that they might be given. Microdoses of LSD did not improve either mood or performance, but they did appear to be safe, and they produced no adverse effects.
To date, studies on microdosing have looked at their effects on healthy populations, and the practice has been associated with “Silicon Valley techies” looking for performance enhancement. Ms. Waldman, however, is different.
She is open about her diagnosis of bipolar disorder, and her long history with therapy and medications. As she describes herself in the beginning of her book, she is emotionally uncomfortable, and both irritable and reactive to the point that her life is propelled by interpersonal chaos. In her uncontrolled ‘study,’ she is an N of 1, and she is pleased with the results. Microdosing, she believes, helped her become less irritable, more resilient, and in fact, have some very good days.
By the end of her memoir, she was looking for a way to continue microdosing but was unsure how to safely obtain more LSD and be certain of its purity. Her experience does raise the possibility that microdoses may have therapeutic benefits in people with certain psychiatric conditions, but this has yet to be studied.
J. Raymond DePaulo Jr., MD, is the chair of the National Network of Depression Centers and a distinguished service professor at Johns Hopkins Hospital, Baltimore. “Microdosing of psychedelics is very problematic for two equally serious reasons,” he cautioned. “There is no control over what it is that people are actually taking, it is completely unstudied scientifically, and there is no agreement on what a ‘micro’ dose is.”
He noted that one of his patients thought he was taking psilocybin. A chemical analysis was done that revealed the agent to contain a combination of THC, a stimulant, morphine, and fluoxetine. There wasn’t a trace of psilocybin. “Mislabeling is the rule, not the exception,” Dr. DePaulo has concluded.
He also believes the placebo effect has a powerful role with microdosing. “It’s not working because of what is in the pill, more likely it is working because of what is advertised to be in it.”
Ms. De Wit noted that when she started her study, she tried to find people who were elevated on measures of depression or anxiety, but she was not looking for a specific clinical population of patients with these clinical diagnoses. “We found a handful of people, and they improved, but so did those in the placebo group; they all got better.”
Psychedelic agents interact with antidepressants, so subjects in controlled studies need to go off their medications before enrolling – this is a limiting factor in studies of both macro- and microdosing. Ms. De Wit also notes that there are logistical and practical obstacles – it is difficult to get approval to use these agents, and the patients have to remain in the lab and be observed for several hours after they are administered, just as with standard doses.
As might be expected, data collection and anecdotal microdosing experiences are rampant on the internet. The social media forum Reddit alone boasts 192,000 members in its microdosing group, while Imperial College London invites microdosers to take part in surveys intended to add to the body of knowledge. But despite its popularity, there is little in the way of prospective, agent-verified, placebo-controlled research exploring whether or not microdosing is truly beneficial beyond just anecdotal evidence.
Perhaps microdosing is a fad, or perhaps it offers some benefits to some people. Given the current interest in the therapeutic uses of psychedelics, it would be useful to have controlled studies of lower doses that don’t carry the risk of “bad trips.”
Certainly, psychiatry could use more agents to address mental health issues, and society might benefit from the use of agents that are proven to be evidence-based options for improving creativity and productivity. Anything that has potential to reduce psychiatric suffering seems worthy of further study to delineate which populations could be helped or harmed.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.
A version of this article first appeared on Medscape.com.
In her month-long memoir, A Really Good Day: How Microdosing Made a Mega Difference in My Mood, My Marriage, and My Life (Knopf, 2017), author Ayelet Waldman turns herself into a one-woman experiment.
Over a single month she takes one-tenth of a recreational dose of LSD every third day. She plots her emotions, her productivity, and her pain along the way. Ms. Waldman obtains the LSD in a single vial, enough for 10 doses, from a researcher, who is retiring. What she’s looking for, she tells the reader, is a really good day – something that has been elusive in her turbulent life.
Although psychedelics remain illegal for both recreational and therapeutic use, they are increasingly being studied at academic centers, and there is hope that they will offer something that our traditional medications might not. However, these are not “micro” doses, but full doses of psychedelic agents that induce clinically-monitored “trips” in order to treat conditions such as depression, anorexia nervosa, or for smoking cessation, to name just a few.
Yet
Because these drugs are illegal under most circumstances, many of the studies involve surveys of users in their natural environments who are already microdosing in an uncontrolled manner. In a 2019 study published in PLOS One, Vince Polito and Richard Stevenson, from Macquarie University, Sydney, gave daily surveys of psychological functioning to 98 microdosers over 6 weeks. Several participants were excluded for using doses that were too high or for concurrent use of other illicit substances.
Whereas the authors found that many people claimed to have positive experiences, there was an increase in neuroticism in some of the subjects. There was no control group and no uniformity to what the subject claimed to be ingesting with regard to dose, frequency, substance, or verification of the chemical content.
University of Chicago neuroscientist Harriet De Wit, PhD, leads one of the few laboratories that conducts controlled, double-blind studies looking at microdosing LSD.
“With microdosing there are expectations, and we don’t know if it’s the expectation or the agent that is making a difference,” she explained. And when asked who in her experience is experimenting with microdosing psychedelics, she expounded “Everybody under the sun!”
Dr. De Wit notes that people microdose to increase their creativity, productivity, focus, and energy, to heighten their spiritual awareness, improve empathy and social relational skills, and to improve their mood – all purported benefits of low-dose psychedelics.
Her group published a study in Addiction Biology, in which 39 subjects were administered low doses of LSD four times over 2 weeks. To address the issues of expectation, the subjects were not told they were participating in a study of hallucinogens specifically but were instead given a list of pharmaceuticals in different classes that they might be given. Microdoses of LSD did not improve either mood or performance, but they did appear to be safe, and they produced no adverse effects.
To date, studies on microdosing have looked at their effects on healthy populations, and the practice has been associated with “Silicon Valley techies” looking for performance enhancement. Ms. Waldman, however, is different.
She is open about her diagnosis of bipolar disorder, and her long history with therapy and medications. As she describes herself in the beginning of her book, she is emotionally uncomfortable, and both irritable and reactive to the point that her life is propelled by interpersonal chaos. In her uncontrolled ‘study,’ she is an N of 1, and she is pleased with the results. Microdosing, she believes, helped her become less irritable, more resilient, and in fact, have some very good days.
By the end of her memoir, she was looking for a way to continue microdosing but was unsure how to safely obtain more LSD and be certain of its purity. Her experience does raise the possibility that microdoses may have therapeutic benefits in people with certain psychiatric conditions, but this has yet to be studied.
J. Raymond DePaulo Jr., MD, is the chair of the National Network of Depression Centers and a distinguished service professor at Johns Hopkins Hospital, Baltimore. “Microdosing of psychedelics is very problematic for two equally serious reasons,” he cautioned. “There is no control over what it is that people are actually taking, it is completely unstudied scientifically, and there is no agreement on what a ‘micro’ dose is.”
He noted that one of his patients thought he was taking psilocybin. A chemical analysis was done that revealed the agent to contain a combination of THC, a stimulant, morphine, and fluoxetine. There wasn’t a trace of psilocybin. “Mislabeling is the rule, not the exception,” Dr. DePaulo has concluded.
He also believes the placebo effect has a powerful role with microdosing. “It’s not working because of what is in the pill, more likely it is working because of what is advertised to be in it.”
Ms. De Wit noted that when she started her study, she tried to find people who were elevated on measures of depression or anxiety, but she was not looking for a specific clinical population of patients with these clinical diagnoses. “We found a handful of people, and they improved, but so did those in the placebo group; they all got better.”
Psychedelic agents interact with antidepressants, so subjects in controlled studies need to go off their medications before enrolling – this is a limiting factor in studies of both macro- and microdosing. Ms. De Wit also notes that there are logistical and practical obstacles – it is difficult to get approval to use these agents, and the patients have to remain in the lab and be observed for several hours after they are administered, just as with standard doses.
As might be expected, data collection and anecdotal microdosing experiences are rampant on the internet. The social media forum Reddit alone boasts 192,000 members in its microdosing group, while Imperial College London invites microdosers to take part in surveys intended to add to the body of knowledge. But despite its popularity, there is little in the way of prospective, agent-verified, placebo-controlled research exploring whether or not microdosing is truly beneficial beyond just anecdotal evidence.
Perhaps microdosing is a fad, or perhaps it offers some benefits to some people. Given the current interest in the therapeutic uses of psychedelics, it would be useful to have controlled studies of lower doses that don’t carry the risk of “bad trips.”
Certainly, psychiatry could use more agents to address mental health issues, and society might benefit from the use of agents that are proven to be evidence-based options for improving creativity and productivity. Anything that has potential to reduce psychiatric suffering seems worthy of further study to delineate which populations could be helped or harmed.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.
A version of this article first appeared on Medscape.com.
In her month-long memoir, A Really Good Day: How Microdosing Made a Mega Difference in My Mood, My Marriage, and My Life (Knopf, 2017), author Ayelet Waldman turns herself into a one-woman experiment.
Over a single month she takes one-tenth of a recreational dose of LSD every third day. She plots her emotions, her productivity, and her pain along the way. Ms. Waldman obtains the LSD in a single vial, enough for 10 doses, from a researcher, who is retiring. What she’s looking for, she tells the reader, is a really good day – something that has been elusive in her turbulent life.
Although psychedelics remain illegal for both recreational and therapeutic use, they are increasingly being studied at academic centers, and there is hope that they will offer something that our traditional medications might not. However, these are not “micro” doses, but full doses of psychedelic agents that induce clinically-monitored “trips” in order to treat conditions such as depression, anorexia nervosa, or for smoking cessation, to name just a few.
Yet
Because these drugs are illegal under most circumstances, many of the studies involve surveys of users in their natural environments who are already microdosing in an uncontrolled manner. In a 2019 study published in PLOS One, Vince Polito and Richard Stevenson, from Macquarie University, Sydney, gave daily surveys of psychological functioning to 98 microdosers over 6 weeks. Several participants were excluded for using doses that were too high or for concurrent use of other illicit substances.
Whereas the authors found that many people claimed to have positive experiences, there was an increase in neuroticism in some of the subjects. There was no control group and no uniformity to what the subject claimed to be ingesting with regard to dose, frequency, substance, or verification of the chemical content.
University of Chicago neuroscientist Harriet De Wit, PhD, leads one of the few laboratories that conducts controlled, double-blind studies looking at microdosing LSD.
“With microdosing there are expectations, and we don’t know if it’s the expectation or the agent that is making a difference,” she explained. And when asked who in her experience is experimenting with microdosing psychedelics, she expounded “Everybody under the sun!”
Dr. De Wit notes that people microdose to increase their creativity, productivity, focus, and energy, to heighten their spiritual awareness, improve empathy and social relational skills, and to improve their mood – all purported benefits of low-dose psychedelics.
Her group published a study in Addiction Biology, in which 39 subjects were administered low doses of LSD four times over 2 weeks. To address the issues of expectation, the subjects were not told they were participating in a study of hallucinogens specifically but were instead given a list of pharmaceuticals in different classes that they might be given. Microdoses of LSD did not improve either mood or performance, but they did appear to be safe, and they produced no adverse effects.
To date, studies on microdosing have looked at their effects on healthy populations, and the practice has been associated with “Silicon Valley techies” looking for performance enhancement. Ms. Waldman, however, is different.
She is open about her diagnosis of bipolar disorder, and her long history with therapy and medications. As she describes herself in the beginning of her book, she is emotionally uncomfortable, and both irritable and reactive to the point that her life is propelled by interpersonal chaos. In her uncontrolled ‘study,’ she is an N of 1, and she is pleased with the results. Microdosing, she believes, helped her become less irritable, more resilient, and in fact, have some very good days.
By the end of her memoir, she was looking for a way to continue microdosing but was unsure how to safely obtain more LSD and be certain of its purity. Her experience does raise the possibility that microdoses may have therapeutic benefits in people with certain psychiatric conditions, but this has yet to be studied.
J. Raymond DePaulo Jr., MD, is the chair of the National Network of Depression Centers and a distinguished service professor at Johns Hopkins Hospital, Baltimore. “Microdosing of psychedelics is very problematic for two equally serious reasons,” he cautioned. “There is no control over what it is that people are actually taking, it is completely unstudied scientifically, and there is no agreement on what a ‘micro’ dose is.”
He noted that one of his patients thought he was taking psilocybin. A chemical analysis was done that revealed the agent to contain a combination of THC, a stimulant, morphine, and fluoxetine. There wasn’t a trace of psilocybin. “Mislabeling is the rule, not the exception,” Dr. DePaulo has concluded.
He also believes the placebo effect has a powerful role with microdosing. “It’s not working because of what is in the pill, more likely it is working because of what is advertised to be in it.”
Ms. De Wit noted that when she started her study, she tried to find people who were elevated on measures of depression or anxiety, but she was not looking for a specific clinical population of patients with these clinical diagnoses. “We found a handful of people, and they improved, but so did those in the placebo group; they all got better.”
Psychedelic agents interact with antidepressants, so subjects in controlled studies need to go off their medications before enrolling – this is a limiting factor in studies of both macro- and microdosing. Ms. De Wit also notes that there are logistical and practical obstacles – it is difficult to get approval to use these agents, and the patients have to remain in the lab and be observed for several hours after they are administered, just as with standard doses.
As might be expected, data collection and anecdotal microdosing experiences are rampant on the internet. The social media forum Reddit alone boasts 192,000 members in its microdosing group, while Imperial College London invites microdosers to take part in surveys intended to add to the body of knowledge. But despite its popularity, there is little in the way of prospective, agent-verified, placebo-controlled research exploring whether or not microdosing is truly beneficial beyond just anecdotal evidence.
Perhaps microdosing is a fad, or perhaps it offers some benefits to some people. Given the current interest in the therapeutic uses of psychedelics, it would be useful to have controlled studies of lower doses that don’t carry the risk of “bad trips.”
Certainly, psychiatry could use more agents to address mental health issues, and society might benefit from the use of agents that are proven to be evidence-based options for improving creativity and productivity. Anything that has potential to reduce psychiatric suffering seems worthy of further study to delineate which populations could be helped or harmed.
Dr. Miller is a coauthor of “Committed: The Battle Over Involuntary Psychiatric Care” (Baltimore: Johns Hopkins University Press, 2016). She has a private practice and is assistant professor of psychiatry and behavioral sciences at Johns Hopkins in Baltimore. Dr. Miller has no conflicts of interest.
A version of this article first appeared on Medscape.com.
A little-known offshoot of hem/onc opens pathway for professional development
Only a small number of pediatric hematologist oncologists and even fewer of our adult counterparts feel comfortable evaluating and treating vascular anomalies.
While admittedly rare, these conditions are still common enough that clinicians in many disciplines encounter them. Hematologist/oncologists are most likely to see vascular malformations, which often present as mass lesions. Complications of these disorders occur across the hematology-oncology spectrum and include clots, pulmonary emboli, cancer predisposition, and an array of functional and psychosocial disorders.
Vascular anomalies are broadly categorized as vascular tumors or malformations. The tumors include hemangiomas, locally aggressive lesions, and true cancers. Malformations can be isolated disorders of one or more blood vessel types (veins, arteries, capillaries or lymphatics), or they can be one part of syndromic disorders. Lymphedema also falls under the heading of vascular anomalies. To make the terminology less confusing, in 2018 the International Society for the Study of Vascular Anomalies refined its classification scheme.
Vascular malformations are thought to be congenital. Although some are obvious at birth, others aren’t apparent until adulthood. In most cases, they grow with a child and may do so disproportionately at puberty and with pregnancies. The fact that vascular malformations persist into adulthood is one reason why their care should be integral to medical hematology-oncology.
Although the cause of a vascular malformation is not always known, a wide range of genetic mutations thought to be pathogenic have been reported. These mutations are usually somatic (only within the involved tissues, not in the blood or germ cells and therefore, not heritable) and tend to cluster in the VEGF-PIK3CA and RAS-MAP signaling pathways.
These genes and pathways will be familiar to any oncologist who cares for patients with solid tumors, notably breast cancer or melanoma. However, unlike the clonal expansion seen in cancers, most vascular malformations will express pathogenic mutations in less than 20% of vascular endothelium within a malformation.
Since 2008, medical management has been limited to sirolimus (rapamycin), a mammalian target of rapamycin inhibitor, which can be effective even when mTOR mutations aren’t apparent. In a seminal phase 2 trial of 57 patients with complex vascular anomalies who were aged 0-29 years, 47 patients had a partial response, 3 patients had stable disease, and 7 patients had progressive disease. None had complete responses. These data highlight the need for more effective treatments.
Recently, vascular anomalists have begun to repurpose drugs from adult oncology that specifically target pathogenic mutations. Some studies underway include Novartis’ international Alpelisib (Piqray) clinical trial for adults and children with PIK3CA-related overgrowth syndromes (NCT04589650) and Merck’s follow-up study of the AKT inhibitor miransertib for PROS and Proteus syndrome. Doses tend to be lower than those used to treat cancers. To date, these have been generally well-tolerated, with sometimes striking but preliminary evidence of efficacy.
During the past 2 years, symposia on vascular anomalies at the annual meeting of the American Society of Hematology have launched what we are hoping is just the start of a broader discussion. In 2020, Fran Blei, MD, chaired Vascular Anomalies 101: Case-Based Discussion on the Diagnosis, Treatment and Lifelong Care of These Patients, and in 2021, Adrienne Hammill, MD, PhD, and Dr. Raj Kasthuri, MBBS, MD, chaired a more specialized symposium: Hereditary Hemorrhagic Telangiectasia (HHT): A Practical Guide to Management.
Dr. Blatt is in the division of pediatric hematology oncology at the University of North Carolina at Chapel Hill. She disclosed no relevant financial relationships.
Only a small number of pediatric hematologist oncologists and even fewer of our adult counterparts feel comfortable evaluating and treating vascular anomalies.
While admittedly rare, these conditions are still common enough that clinicians in many disciplines encounter them. Hematologist/oncologists are most likely to see vascular malformations, which often present as mass lesions. Complications of these disorders occur across the hematology-oncology spectrum and include clots, pulmonary emboli, cancer predisposition, and an array of functional and psychosocial disorders.
Vascular anomalies are broadly categorized as vascular tumors or malformations. The tumors include hemangiomas, locally aggressive lesions, and true cancers. Malformations can be isolated disorders of one or more blood vessel types (veins, arteries, capillaries or lymphatics), or they can be one part of syndromic disorders. Lymphedema also falls under the heading of vascular anomalies. To make the terminology less confusing, in 2018 the International Society for the Study of Vascular Anomalies refined its classification scheme.
Vascular malformations are thought to be congenital. Although some are obvious at birth, others aren’t apparent until adulthood. In most cases, they grow with a child and may do so disproportionately at puberty and with pregnancies. The fact that vascular malformations persist into adulthood is one reason why their care should be integral to medical hematology-oncology.
Although the cause of a vascular malformation is not always known, a wide range of genetic mutations thought to be pathogenic have been reported. These mutations are usually somatic (only within the involved tissues, not in the blood or germ cells and therefore, not heritable) and tend to cluster in the VEGF-PIK3CA and RAS-MAP signaling pathways.
These genes and pathways will be familiar to any oncologist who cares for patients with solid tumors, notably breast cancer or melanoma. However, unlike the clonal expansion seen in cancers, most vascular malformations will express pathogenic mutations in less than 20% of vascular endothelium within a malformation.
Since 2008, medical management has been limited to sirolimus (rapamycin), a mammalian target of rapamycin inhibitor, which can be effective even when mTOR mutations aren’t apparent. In a seminal phase 2 trial of 57 patients with complex vascular anomalies who were aged 0-29 years, 47 patients had a partial response, 3 patients had stable disease, and 7 patients had progressive disease. None had complete responses. These data highlight the need for more effective treatments.
Recently, vascular anomalists have begun to repurpose drugs from adult oncology that specifically target pathogenic mutations. Some studies underway include Novartis’ international Alpelisib (Piqray) clinical trial for adults and children with PIK3CA-related overgrowth syndromes (NCT04589650) and Merck’s follow-up study of the AKT inhibitor miransertib for PROS and Proteus syndrome. Doses tend to be lower than those used to treat cancers. To date, these have been generally well-tolerated, with sometimes striking but preliminary evidence of efficacy.
During the past 2 years, symposia on vascular anomalies at the annual meeting of the American Society of Hematology have launched what we are hoping is just the start of a broader discussion. In 2020, Fran Blei, MD, chaired Vascular Anomalies 101: Case-Based Discussion on the Diagnosis, Treatment and Lifelong Care of These Patients, and in 2021, Adrienne Hammill, MD, PhD, and Dr. Raj Kasthuri, MBBS, MD, chaired a more specialized symposium: Hereditary Hemorrhagic Telangiectasia (HHT): A Practical Guide to Management.
Dr. Blatt is in the division of pediatric hematology oncology at the University of North Carolina at Chapel Hill. She disclosed no relevant financial relationships.
Only a small number of pediatric hematologist oncologists and even fewer of our adult counterparts feel comfortable evaluating and treating vascular anomalies.
While admittedly rare, these conditions are still common enough that clinicians in many disciplines encounter them. Hematologist/oncologists are most likely to see vascular malformations, which often present as mass lesions. Complications of these disorders occur across the hematology-oncology spectrum and include clots, pulmonary emboli, cancer predisposition, and an array of functional and psychosocial disorders.
Vascular anomalies are broadly categorized as vascular tumors or malformations. The tumors include hemangiomas, locally aggressive lesions, and true cancers. Malformations can be isolated disorders of one or more blood vessel types (veins, arteries, capillaries or lymphatics), or they can be one part of syndromic disorders. Lymphedema also falls under the heading of vascular anomalies. To make the terminology less confusing, in 2018 the International Society for the Study of Vascular Anomalies refined its classification scheme.
Vascular malformations are thought to be congenital. Although some are obvious at birth, others aren’t apparent until adulthood. In most cases, they grow with a child and may do so disproportionately at puberty and with pregnancies. The fact that vascular malformations persist into adulthood is one reason why their care should be integral to medical hematology-oncology.
Although the cause of a vascular malformation is not always known, a wide range of genetic mutations thought to be pathogenic have been reported. These mutations are usually somatic (only within the involved tissues, not in the blood or germ cells and therefore, not heritable) and tend to cluster in the VEGF-PIK3CA and RAS-MAP signaling pathways.
These genes and pathways will be familiar to any oncologist who cares for patients with solid tumors, notably breast cancer or melanoma. However, unlike the clonal expansion seen in cancers, most vascular malformations will express pathogenic mutations in less than 20% of vascular endothelium within a malformation.
Since 2008, medical management has been limited to sirolimus (rapamycin), a mammalian target of rapamycin inhibitor, which can be effective even when mTOR mutations aren’t apparent. In a seminal phase 2 trial of 57 patients with complex vascular anomalies who were aged 0-29 years, 47 patients had a partial response, 3 patients had stable disease, and 7 patients had progressive disease. None had complete responses. These data highlight the need for more effective treatments.
Recently, vascular anomalists have begun to repurpose drugs from adult oncology that specifically target pathogenic mutations. Some studies underway include Novartis’ international Alpelisib (Piqray) clinical trial for adults and children with PIK3CA-related overgrowth syndromes (NCT04589650) and Merck’s follow-up study of the AKT inhibitor miransertib for PROS and Proteus syndrome. Doses tend to be lower than those used to treat cancers. To date, these have been generally well-tolerated, with sometimes striking but preliminary evidence of efficacy.
During the past 2 years, symposia on vascular anomalies at the annual meeting of the American Society of Hematology have launched what we are hoping is just the start of a broader discussion. In 2020, Fran Blei, MD, chaired Vascular Anomalies 101: Case-Based Discussion on the Diagnosis, Treatment and Lifelong Care of These Patients, and in 2021, Adrienne Hammill, MD, PhD, and Dr. Raj Kasthuri, MBBS, MD, chaired a more specialized symposium: Hereditary Hemorrhagic Telangiectasia (HHT): A Practical Guide to Management.
Dr. Blatt is in the division of pediatric hematology oncology at the University of North Carolina at Chapel Hill. She disclosed no relevant financial relationships.
Late to the game: Parenting after 40
As they rolled me down the hallway to the OR, ceiling lights rhythmically passing above, I zoned out into a 1,000-mile stare. How did I get here? I started humming “Swing Low, Sweet Chariot,” praying for a miracle to happen. I thought back to my birth plan, meticulously crafted, a one-pager so that the no-nonsense labor and delivery nurses wouldn›t think me completely off my rocker. No C-section unless medically necessary. Those words laughed back at me – cackling, even. I’d planned out the whole birthing process and here we were, my team almost jogging me to the OR. I lay still, utterly gobsmacked and partially anesthetized.
If I squint my eyes and hallucinate just a bit, that is sort of what motherhood has been like.
It’s about knowing all the things that could go wrong and meeting the unplanned head-on. Motherhood has indeed been a whirlwind – so many physical, psychological, and emotional transformations. And to top it off, the added effort of giving birth in a pandemic. As an over-40 physician, you’d think I would have been better prepared.
I was, but in a sense, I was not. The knowledge, the wisdom, the experience of my medical training surrounded me, but even I panicked at times in the beginning: Am I feeding her correctly? Am I making enough food for her? Am I doing the best that I can for her? What more could I be doing for her?
Over time, I’ve learned to lighten up. Some. In those teachable moments with my daughter Gia, I’ve learned to not sugarcoat reality but encourage the hopeful. If Gia falls on the ground? “You’re okay, sweetie. Now get back up.” If Gia has a tantrum and starts hitting herself? “Honey, our hands are for hugs, not hurting ourselves. Let’s go play.” Eighty percent of motherhood right now is redirection and the other 20% is patience.
I remember this one time I was rushing out the door for work. After getting in the car with my keys, I realized I forgot my coffee back in the house. I left the car, went back in the house to grab the blessed joe, went back to the car, and couldn’t get in because it was locked. I panicked at that moment, went back inside the house, and found Gia playing with my extra key fob. My own daughter locked me out of my car. Of course, it wasn’t her fault. Deep breath and I offered her another kiss while simultaneously taking the key fob from her.
Before Gia could walk, she could climb the stairs in our home. Her father and I sometimes refer to her as “Lil Bamm-Bamm” because she is so strong. One day, Daddy was supposed to be watching her while Mommy was folding laundry upstairs. She was not allowed on the stairs, but what should I hear? Literally, the pitter-patter of little feet, running down the upstairs hallway. Her father had drifted off watching yet another episode of something Star Wars–related. My strong little girl made it up the stairs all by herself and Dad received a strong word. The Force was with me that day.
I would say that I feel like having a child ages you, but what does that really mean when you’re already old? I’ve become acutely aware of my lack of endurance, stamina, and bodily strength. My knees will creak when taking her upstairs to bed, an osseous dirge of a lullaby. Date nights become unintentionally less and less frequent. Friday night dress-up becomes Friday night dress-down. I’ve replaced stiletto heels with comfy sweats.
Once we put Gia down for the night, we are usually exhausted from the day, and the couch and TV are welcome respites. We exhale. As over-40 parents, we knew that having children late in life would bring its challenges. But I’d like to think that we are meeting them the best way that we can. Often I encourage my body to meet Gia at her eye level, see what she sees, play with her on her own terms, and match her energy. She absolutely loves it when I do this. I’m out of breath and my knees are sore by the end of our play session, but I wouldn’t have it any other way.
We are learning from each other. She has a bright and assertive personality, and I am protective of that innocence. Her innocence is without fear. I often wonder what she is thinking when I see her facial expressions. A side-eye, a fleeting giggle. Is she secretly contemplating the chronicity of the cosmos, or is it just gas? I look at her in stolen moments and still can’t believe that I grew a human inside me, and said human was extracted from me and is now walking around my house commanding her bidding. So surreal. The unromanticized, scientific ingredients that are at play from conception to delivery are nothing short of miraculous. And the miracles of parenting over 40 are present every day.
Dr. Tolliver is a family medicine physician at The Ohio State University Wexner Medical Center in Columbus. A version of this article first appeared on Medscape.com.
As they rolled me down the hallway to the OR, ceiling lights rhythmically passing above, I zoned out into a 1,000-mile stare. How did I get here? I started humming “Swing Low, Sweet Chariot,” praying for a miracle to happen. I thought back to my birth plan, meticulously crafted, a one-pager so that the no-nonsense labor and delivery nurses wouldn›t think me completely off my rocker. No C-section unless medically necessary. Those words laughed back at me – cackling, even. I’d planned out the whole birthing process and here we were, my team almost jogging me to the OR. I lay still, utterly gobsmacked and partially anesthetized.
If I squint my eyes and hallucinate just a bit, that is sort of what motherhood has been like.
It’s about knowing all the things that could go wrong and meeting the unplanned head-on. Motherhood has indeed been a whirlwind – so many physical, psychological, and emotional transformations. And to top it off, the added effort of giving birth in a pandemic. As an over-40 physician, you’d think I would have been better prepared.
I was, but in a sense, I was not. The knowledge, the wisdom, the experience of my medical training surrounded me, but even I panicked at times in the beginning: Am I feeding her correctly? Am I making enough food for her? Am I doing the best that I can for her? What more could I be doing for her?
Over time, I’ve learned to lighten up. Some. In those teachable moments with my daughter Gia, I’ve learned to not sugarcoat reality but encourage the hopeful. If Gia falls on the ground? “You’re okay, sweetie. Now get back up.” If Gia has a tantrum and starts hitting herself? “Honey, our hands are for hugs, not hurting ourselves. Let’s go play.” Eighty percent of motherhood right now is redirection and the other 20% is patience.
I remember this one time I was rushing out the door for work. After getting in the car with my keys, I realized I forgot my coffee back in the house. I left the car, went back in the house to grab the blessed joe, went back to the car, and couldn’t get in because it was locked. I panicked at that moment, went back inside the house, and found Gia playing with my extra key fob. My own daughter locked me out of my car. Of course, it wasn’t her fault. Deep breath and I offered her another kiss while simultaneously taking the key fob from her.
Before Gia could walk, she could climb the stairs in our home. Her father and I sometimes refer to her as “Lil Bamm-Bamm” because she is so strong. One day, Daddy was supposed to be watching her while Mommy was folding laundry upstairs. She was not allowed on the stairs, but what should I hear? Literally, the pitter-patter of little feet, running down the upstairs hallway. Her father had drifted off watching yet another episode of something Star Wars–related. My strong little girl made it up the stairs all by herself and Dad received a strong word. The Force was with me that day.
I would say that I feel like having a child ages you, but what does that really mean when you’re already old? I’ve become acutely aware of my lack of endurance, stamina, and bodily strength. My knees will creak when taking her upstairs to bed, an osseous dirge of a lullaby. Date nights become unintentionally less and less frequent. Friday night dress-up becomes Friday night dress-down. I’ve replaced stiletto heels with comfy sweats.
Once we put Gia down for the night, we are usually exhausted from the day, and the couch and TV are welcome respites. We exhale. As over-40 parents, we knew that having children late in life would bring its challenges. But I’d like to think that we are meeting them the best way that we can. Often I encourage my body to meet Gia at her eye level, see what she sees, play with her on her own terms, and match her energy. She absolutely loves it when I do this. I’m out of breath and my knees are sore by the end of our play session, but I wouldn’t have it any other way.
We are learning from each other. She has a bright and assertive personality, and I am protective of that innocence. Her innocence is without fear. I often wonder what she is thinking when I see her facial expressions. A side-eye, a fleeting giggle. Is she secretly contemplating the chronicity of the cosmos, or is it just gas? I look at her in stolen moments and still can’t believe that I grew a human inside me, and said human was extracted from me and is now walking around my house commanding her bidding. So surreal. The unromanticized, scientific ingredients that are at play from conception to delivery are nothing short of miraculous. And the miracles of parenting over 40 are present every day.
Dr. Tolliver is a family medicine physician at The Ohio State University Wexner Medical Center in Columbus. A version of this article first appeared on Medscape.com.
As they rolled me down the hallway to the OR, ceiling lights rhythmically passing above, I zoned out into a 1,000-mile stare. How did I get here? I started humming “Swing Low, Sweet Chariot,” praying for a miracle to happen. I thought back to my birth plan, meticulously crafted, a one-pager so that the no-nonsense labor and delivery nurses wouldn›t think me completely off my rocker. No C-section unless medically necessary. Those words laughed back at me – cackling, even. I’d planned out the whole birthing process and here we were, my team almost jogging me to the OR. I lay still, utterly gobsmacked and partially anesthetized.
If I squint my eyes and hallucinate just a bit, that is sort of what motherhood has been like.
It’s about knowing all the things that could go wrong and meeting the unplanned head-on. Motherhood has indeed been a whirlwind – so many physical, psychological, and emotional transformations. And to top it off, the added effort of giving birth in a pandemic. As an over-40 physician, you’d think I would have been better prepared.
I was, but in a sense, I was not. The knowledge, the wisdom, the experience of my medical training surrounded me, but even I panicked at times in the beginning: Am I feeding her correctly? Am I making enough food for her? Am I doing the best that I can for her? What more could I be doing for her?
Over time, I’ve learned to lighten up. Some. In those teachable moments with my daughter Gia, I’ve learned to not sugarcoat reality but encourage the hopeful. If Gia falls on the ground? “You’re okay, sweetie. Now get back up.” If Gia has a tantrum and starts hitting herself? “Honey, our hands are for hugs, not hurting ourselves. Let’s go play.” Eighty percent of motherhood right now is redirection and the other 20% is patience.
I remember this one time I was rushing out the door for work. After getting in the car with my keys, I realized I forgot my coffee back in the house. I left the car, went back in the house to grab the blessed joe, went back to the car, and couldn’t get in because it was locked. I panicked at that moment, went back inside the house, and found Gia playing with my extra key fob. My own daughter locked me out of my car. Of course, it wasn’t her fault. Deep breath and I offered her another kiss while simultaneously taking the key fob from her.
Before Gia could walk, she could climb the stairs in our home. Her father and I sometimes refer to her as “Lil Bamm-Bamm” because she is so strong. One day, Daddy was supposed to be watching her while Mommy was folding laundry upstairs. She was not allowed on the stairs, but what should I hear? Literally, the pitter-patter of little feet, running down the upstairs hallway. Her father had drifted off watching yet another episode of something Star Wars–related. My strong little girl made it up the stairs all by herself and Dad received a strong word. The Force was with me that day.
I would say that I feel like having a child ages you, but what does that really mean when you’re already old? I’ve become acutely aware of my lack of endurance, stamina, and bodily strength. My knees will creak when taking her upstairs to bed, an osseous dirge of a lullaby. Date nights become unintentionally less and less frequent. Friday night dress-up becomes Friday night dress-down. I’ve replaced stiletto heels with comfy sweats.
Once we put Gia down for the night, we are usually exhausted from the day, and the couch and TV are welcome respites. We exhale. As over-40 parents, we knew that having children late in life would bring its challenges. But I’d like to think that we are meeting them the best way that we can. Often I encourage my body to meet Gia at her eye level, see what she sees, play with her on her own terms, and match her energy. She absolutely loves it when I do this. I’m out of breath and my knees are sore by the end of our play session, but I wouldn’t have it any other way.
We are learning from each other. She has a bright and assertive personality, and I am protective of that innocence. Her innocence is without fear. I often wonder what she is thinking when I see her facial expressions. A side-eye, a fleeting giggle. Is she secretly contemplating the chronicity of the cosmos, or is it just gas? I look at her in stolen moments and still can’t believe that I grew a human inside me, and said human was extracted from me and is now walking around my house commanding her bidding. So surreal. The unromanticized, scientific ingredients that are at play from conception to delivery are nothing short of miraculous. And the miracles of parenting over 40 are present every day.
Dr. Tolliver is a family medicine physician at The Ohio State University Wexner Medical Center in Columbus. A version of this article first appeared on Medscape.com.
Will we ever outgrow the Goldwater rule?
Since it appeared in the first edition of the American Psychiatric Association’s Principles of Medical Ethics in 1973, the “Goldwater rule” – often referred to in terms of where in the APA’s guideline it can be found, Section 7.3 – has placed a stringent prohibition on psychiatrists offering professional opinions about public figures “unless he or she has conducted an examination and has been granted proper authorization for such a statement.”1
Some psychiatrists experienced the restrictive nature of Section 7.3 more acutely perhaps than ever during the Trump presidency. This spurred numerous articles criticizing the guideline as an outdated “gag rule”2 that harms the public image of psychiatry.3 Some psychiatrists violated the rule to warn the public of the dangers of a president with “incipient dementia”4 occupying the most powerful position on earth.
Following President Trump’s exit from the White House, the alarm bells surrounding his presidency have quieted. Criticisms of the Goldwater rule, on the other hand, have persisted. Many of these criticisms now call for the rule to be refined, allowing for psychiatrists to give their professional opinions about public figures, but with certain guidelines on how to do so.5 Few have yet to make a sober case for the outright abolition of Section 7.3.6
Self-regulating and internal policing are important factors in the continued independence of the medical profession, and we should continue to hold each other to high professional standards. That being said, do psychiatrists need training wheels to prevent us from devolving into unprofessional social commentators? Other medical specialties do not see the need to implement a rule preventing their colleagues from expressing expertise in fear of embarrassment. Do we not have faith in our ability to conduct ourselves professionally? Is the Goldwater rule an admission of a juvenile lack of self-control within our field?
Not only do other medical specialties not forcibly handhold their members in public settings, but other “providers” in the realm of mental health likewise do not implement such strict self-restraints. Psychiatry staying silent on the matter of public figures leaves a void filled by other, arguably less qualified, individuals. Subsequently, the public discord risks being flooded with pseudoscientific pontification and distorted views of psychiatric illness. The cycle of speculating on the mental fitness of the president has outlived President Trump, with concerns about Joe Biden’s incoherence and waning cognition.7 Therein is an important argument to be made for the public duty of psychiatrists, with their greater expertise and clinical acumen, to weigh in on matters of societal importance in an attempt to dispel dangerous misconceptions.
Practical limitations are often raised and serve as the cornerstone for the Goldwater rule. Specifically, the limitation being that a psychiatrist cannot provide a professional opinion about an individual without a proper in-person evaluation. The psychiatric interview could be considered the most in-depth and comprehensive evaluation in all of medicine. Even so, is a trained psychiatrist presented with grandiosity, flight-of-ideas, and pressured speech unable to comment on the possibility of mania without a lengthy and comprehensive evaluation? How much disorganization of behavior and dialogue does one need to observe to recognize psychosis? For the experienced psychiatrist, many of these behavioral hallmarks are akin to an ST elevation on an EKG representing a heart attack.
When considering less extreme examples of mental affliction, such as depression and anxiety, many signs – including demeanor, motor activity, manner of speaking, and other aspects of behavior – are apparent to the perceptive psychiatrist without needing an extensive interview that dives into the depths of a person’s social history and childhood. After all, our own criteria for depression and mania do not require the presence of social stressors or childhood trauma. Even personality disorders can be reasonably postulated when a person behaves in a particular fashion. The recognition of transitional objects, items used to provide psychological comfort, including the “teddy bear sign” are common and scientifically studied methods to recognize personality disorder.8
The necessity for an in-person evaluation has become less compelling over the years. In our modern age, important social moments are memorialized in countless videos that are arguably more relevant, more accurate, and less subjective than a psychiatric interview. Furthermore, forensic psychiatrists routinely comment on individuals they have not examined for a variety of reasons, from postmortem analysis to the refusal of the client to be interviewed. Moreover, and with significant contradiction, many leaders in the field of psychiatry view integrated care, the practice of psychiatrists advising primary care doctors, often without even seeing patients, to be the future of psychiatry.9
Some reading this may scoff at the above examples. Perhaps Section 7.3 speaks to an underlying insecurity in our field regarding our ability to accurately diagnose. That insecurity is not unfounded. In terms of the DSM-5, the bar for reliability has been lowered to a kappa of 0.2-0.4, from a previous standard of 0.6, in an attempt to avoid critiques of unreliability.10 Yet herein lies a powerful recognition of the necessity of the Goldwater rule. If psychiatrists cannot reliably agree on the presence of diagnoses in the controlled setting of scientific study, how can we expect to speak with coherence and consistency on highly mediatized and provoking topics?
The defense – that the difficulty psychiatrists have at providing an accurate diagnosis stems from the immense complexity of the system being evaluated, the human mind – is a valid one. Attempts to force such complex pathology, with all its many variables, into the check-box approach implemented in the DSM inevitably leads to problems with diagnostic reliability. Still, as psychiatrists we retain a level of expertise in assessing and treating complex disorders of the mind that no other field can claim.
The duty physicians have not only to work toward the health of their individual patients, but also to act in service of the public health and well-being of communities in which our patients live, is well established. How ethical is it then for psychiatry to absolve itself from duty when it comes to public figures at the center of shaping public opinion? There are numerous recent, high-profile instances where our expertise may have helped shine light in an otherwise murky public discussion filled with disinformation. The death of George Floyd and the year of turmoil that followed is a salient example. The conservatorship of Britney Spears and the resulting societal outcry is another. Even setting the matter of diagnosis aside, we can help illuminate the societal implications of conservatorship laws,11 in addition to providing input on how to safely and responsibly approach an individual who is in crisis, under the influence of multiple illicit substances, and possibly suffering from excited delirium.
Whether psychiatry has progressed enough as a medical specialty to trust ourselves with the option of providing professional opinions on public figures is an ongoing debate. The persistence of the Goldwater rule is a strong testament to the internal lack of confidence among psychiatrists regarding our ability to provide accurate diagnoses, act with integrity in the public space, and foster a positive public image. That lack of confidence may be well deserved. However, it is possible that our field will never go through the necessary pains of maturing as long as Section 7.3 remains in place.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Compton has no conflicts of interest. Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. He has no conflicts of interest.
References
1. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Section 7. American Psychiatric Association; 2013 edition.
2. Glass LL. The Goldwater rule is broken. Here’s how to fix it. STAT News. 2018 June 18.
3. Plymyer D. The Goldwater rule paradox. 2020 Aug 7.
4. Lieberman JA. Trump’s brain and the 25th Amendment. Vice. 2017 Sep 8.
5. Blotcky AD et al. The Goldwater rule is fine, if refined. Here’s how to do it. Psychiatric Times. 2022 Jan 6;39(1).
6. Blotcky AD and Norrholm SD. After Trump, end the Goldwater rule once and for all. New York Daily News. 2020 Dec 22.
7. Stephens B. Biden should not run again – And he should say he won’t. New York Times. 2021 Dec 14.
8. Schmaling KB et al. The positive teddy bear sign: Transitional objects in the medical setting. J Nerv Ment Dis. 1994 Dec;182(12):725.
9. Badre N et al. Psychopharmacologic management in integrated care: Challenges for residency education. Acad Psychiatry. 2015; 39(4):466-9.
10. Kraemer HC et al. DSM-5: How reliable is reliable enough? Am J Psychiatry. 2012 Jan;169(1):13-5.
11. Badre N and Compton C. Britney Spears – Reflections on conservatorship. Clinical Psychiatry News. 2021 Nov 16.
Since it appeared in the first edition of the American Psychiatric Association’s Principles of Medical Ethics in 1973, the “Goldwater rule” – often referred to in terms of where in the APA’s guideline it can be found, Section 7.3 – has placed a stringent prohibition on psychiatrists offering professional opinions about public figures “unless he or she has conducted an examination and has been granted proper authorization for such a statement.”1
Some psychiatrists experienced the restrictive nature of Section 7.3 more acutely perhaps than ever during the Trump presidency. This spurred numerous articles criticizing the guideline as an outdated “gag rule”2 that harms the public image of psychiatry.3 Some psychiatrists violated the rule to warn the public of the dangers of a president with “incipient dementia”4 occupying the most powerful position on earth.
Following President Trump’s exit from the White House, the alarm bells surrounding his presidency have quieted. Criticisms of the Goldwater rule, on the other hand, have persisted. Many of these criticisms now call for the rule to be refined, allowing for psychiatrists to give their professional opinions about public figures, but with certain guidelines on how to do so.5 Few have yet to make a sober case for the outright abolition of Section 7.3.6
Self-regulating and internal policing are important factors in the continued independence of the medical profession, and we should continue to hold each other to high professional standards. That being said, do psychiatrists need training wheels to prevent us from devolving into unprofessional social commentators? Other medical specialties do not see the need to implement a rule preventing their colleagues from expressing expertise in fear of embarrassment. Do we not have faith in our ability to conduct ourselves professionally? Is the Goldwater rule an admission of a juvenile lack of self-control within our field?
Not only do other medical specialties not forcibly handhold their members in public settings, but other “providers” in the realm of mental health likewise do not implement such strict self-restraints. Psychiatry staying silent on the matter of public figures leaves a void filled by other, arguably less qualified, individuals. Subsequently, the public discord risks being flooded with pseudoscientific pontification and distorted views of psychiatric illness. The cycle of speculating on the mental fitness of the president has outlived President Trump, with concerns about Joe Biden’s incoherence and waning cognition.7 Therein is an important argument to be made for the public duty of psychiatrists, with their greater expertise and clinical acumen, to weigh in on matters of societal importance in an attempt to dispel dangerous misconceptions.
Practical limitations are often raised and serve as the cornerstone for the Goldwater rule. Specifically, the limitation being that a psychiatrist cannot provide a professional opinion about an individual without a proper in-person evaluation. The psychiatric interview could be considered the most in-depth and comprehensive evaluation in all of medicine. Even so, is a trained psychiatrist presented with grandiosity, flight-of-ideas, and pressured speech unable to comment on the possibility of mania without a lengthy and comprehensive evaluation? How much disorganization of behavior and dialogue does one need to observe to recognize psychosis? For the experienced psychiatrist, many of these behavioral hallmarks are akin to an ST elevation on an EKG representing a heart attack.
When considering less extreme examples of mental affliction, such as depression and anxiety, many signs – including demeanor, motor activity, manner of speaking, and other aspects of behavior – are apparent to the perceptive psychiatrist without needing an extensive interview that dives into the depths of a person’s social history and childhood. After all, our own criteria for depression and mania do not require the presence of social stressors or childhood trauma. Even personality disorders can be reasonably postulated when a person behaves in a particular fashion. The recognition of transitional objects, items used to provide psychological comfort, including the “teddy bear sign” are common and scientifically studied methods to recognize personality disorder.8
The necessity for an in-person evaluation has become less compelling over the years. In our modern age, important social moments are memorialized in countless videos that are arguably more relevant, more accurate, and less subjective than a psychiatric interview. Furthermore, forensic psychiatrists routinely comment on individuals they have not examined for a variety of reasons, from postmortem analysis to the refusal of the client to be interviewed. Moreover, and with significant contradiction, many leaders in the field of psychiatry view integrated care, the practice of psychiatrists advising primary care doctors, often without even seeing patients, to be the future of psychiatry.9
Some reading this may scoff at the above examples. Perhaps Section 7.3 speaks to an underlying insecurity in our field regarding our ability to accurately diagnose. That insecurity is not unfounded. In terms of the DSM-5, the bar for reliability has been lowered to a kappa of 0.2-0.4, from a previous standard of 0.6, in an attempt to avoid critiques of unreliability.10 Yet herein lies a powerful recognition of the necessity of the Goldwater rule. If psychiatrists cannot reliably agree on the presence of diagnoses in the controlled setting of scientific study, how can we expect to speak with coherence and consistency on highly mediatized and provoking topics?
The defense – that the difficulty psychiatrists have at providing an accurate diagnosis stems from the immense complexity of the system being evaluated, the human mind – is a valid one. Attempts to force such complex pathology, with all its many variables, into the check-box approach implemented in the DSM inevitably leads to problems with diagnostic reliability. Still, as psychiatrists we retain a level of expertise in assessing and treating complex disorders of the mind that no other field can claim.
The duty physicians have not only to work toward the health of their individual patients, but also to act in service of the public health and well-being of communities in which our patients live, is well established. How ethical is it then for psychiatry to absolve itself from duty when it comes to public figures at the center of shaping public opinion? There are numerous recent, high-profile instances where our expertise may have helped shine light in an otherwise murky public discussion filled with disinformation. The death of George Floyd and the year of turmoil that followed is a salient example. The conservatorship of Britney Spears and the resulting societal outcry is another. Even setting the matter of diagnosis aside, we can help illuminate the societal implications of conservatorship laws,11 in addition to providing input on how to safely and responsibly approach an individual who is in crisis, under the influence of multiple illicit substances, and possibly suffering from excited delirium.
Whether psychiatry has progressed enough as a medical specialty to trust ourselves with the option of providing professional opinions on public figures is an ongoing debate. The persistence of the Goldwater rule is a strong testament to the internal lack of confidence among psychiatrists regarding our ability to provide accurate diagnoses, act with integrity in the public space, and foster a positive public image. That lack of confidence may be well deserved. However, it is possible that our field will never go through the necessary pains of maturing as long as Section 7.3 remains in place.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Compton has no conflicts of interest. Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. He has no conflicts of interest.
References
1. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Section 7. American Psychiatric Association; 2013 edition.
2. Glass LL. The Goldwater rule is broken. Here’s how to fix it. STAT News. 2018 June 18.
3. Plymyer D. The Goldwater rule paradox. 2020 Aug 7.
4. Lieberman JA. Trump’s brain and the 25th Amendment. Vice. 2017 Sep 8.
5. Blotcky AD et al. The Goldwater rule is fine, if refined. Here’s how to do it. Psychiatric Times. 2022 Jan 6;39(1).
6. Blotcky AD and Norrholm SD. After Trump, end the Goldwater rule once and for all. New York Daily News. 2020 Dec 22.
7. Stephens B. Biden should not run again – And he should say he won’t. New York Times. 2021 Dec 14.
8. Schmaling KB et al. The positive teddy bear sign: Transitional objects in the medical setting. J Nerv Ment Dis. 1994 Dec;182(12):725.
9. Badre N et al. Psychopharmacologic management in integrated care: Challenges for residency education. Acad Psychiatry. 2015; 39(4):466-9.
10. Kraemer HC et al. DSM-5: How reliable is reliable enough? Am J Psychiatry. 2012 Jan;169(1):13-5.
11. Badre N and Compton C. Britney Spears – Reflections on conservatorship. Clinical Psychiatry News. 2021 Nov 16.
Since it appeared in the first edition of the American Psychiatric Association’s Principles of Medical Ethics in 1973, the “Goldwater rule” – often referred to in terms of where in the APA’s guideline it can be found, Section 7.3 – has placed a stringent prohibition on psychiatrists offering professional opinions about public figures “unless he or she has conducted an examination and has been granted proper authorization for such a statement.”1
Some psychiatrists experienced the restrictive nature of Section 7.3 more acutely perhaps than ever during the Trump presidency. This spurred numerous articles criticizing the guideline as an outdated “gag rule”2 that harms the public image of psychiatry.3 Some psychiatrists violated the rule to warn the public of the dangers of a president with “incipient dementia”4 occupying the most powerful position on earth.
Following President Trump’s exit from the White House, the alarm bells surrounding his presidency have quieted. Criticisms of the Goldwater rule, on the other hand, have persisted. Many of these criticisms now call for the rule to be refined, allowing for psychiatrists to give their professional opinions about public figures, but with certain guidelines on how to do so.5 Few have yet to make a sober case for the outright abolition of Section 7.3.6
Self-regulating and internal policing are important factors in the continued independence of the medical profession, and we should continue to hold each other to high professional standards. That being said, do psychiatrists need training wheels to prevent us from devolving into unprofessional social commentators? Other medical specialties do not see the need to implement a rule preventing their colleagues from expressing expertise in fear of embarrassment. Do we not have faith in our ability to conduct ourselves professionally? Is the Goldwater rule an admission of a juvenile lack of self-control within our field?
Not only do other medical specialties not forcibly handhold their members in public settings, but other “providers” in the realm of mental health likewise do not implement such strict self-restraints. Psychiatry staying silent on the matter of public figures leaves a void filled by other, arguably less qualified, individuals. Subsequently, the public discord risks being flooded with pseudoscientific pontification and distorted views of psychiatric illness. The cycle of speculating on the mental fitness of the president has outlived President Trump, with concerns about Joe Biden’s incoherence and waning cognition.7 Therein is an important argument to be made for the public duty of psychiatrists, with their greater expertise and clinical acumen, to weigh in on matters of societal importance in an attempt to dispel dangerous misconceptions.
Practical limitations are often raised and serve as the cornerstone for the Goldwater rule. Specifically, the limitation being that a psychiatrist cannot provide a professional opinion about an individual without a proper in-person evaluation. The psychiatric interview could be considered the most in-depth and comprehensive evaluation in all of medicine. Even so, is a trained psychiatrist presented with grandiosity, flight-of-ideas, and pressured speech unable to comment on the possibility of mania without a lengthy and comprehensive evaluation? How much disorganization of behavior and dialogue does one need to observe to recognize psychosis? For the experienced psychiatrist, many of these behavioral hallmarks are akin to an ST elevation on an EKG representing a heart attack.
When considering less extreme examples of mental affliction, such as depression and anxiety, many signs – including demeanor, motor activity, manner of speaking, and other aspects of behavior – are apparent to the perceptive psychiatrist without needing an extensive interview that dives into the depths of a person’s social history and childhood. After all, our own criteria for depression and mania do not require the presence of social stressors or childhood trauma. Even personality disorders can be reasonably postulated when a person behaves in a particular fashion. The recognition of transitional objects, items used to provide psychological comfort, including the “teddy bear sign” are common and scientifically studied methods to recognize personality disorder.8
The necessity for an in-person evaluation has become less compelling over the years. In our modern age, important social moments are memorialized in countless videos that are arguably more relevant, more accurate, and less subjective than a psychiatric interview. Furthermore, forensic psychiatrists routinely comment on individuals they have not examined for a variety of reasons, from postmortem analysis to the refusal of the client to be interviewed. Moreover, and with significant contradiction, many leaders in the field of psychiatry view integrated care, the practice of psychiatrists advising primary care doctors, often without even seeing patients, to be the future of psychiatry.9
Some reading this may scoff at the above examples. Perhaps Section 7.3 speaks to an underlying insecurity in our field regarding our ability to accurately diagnose. That insecurity is not unfounded. In terms of the DSM-5, the bar for reliability has been lowered to a kappa of 0.2-0.4, from a previous standard of 0.6, in an attempt to avoid critiques of unreliability.10 Yet herein lies a powerful recognition of the necessity of the Goldwater rule. If psychiatrists cannot reliably agree on the presence of diagnoses in the controlled setting of scientific study, how can we expect to speak with coherence and consistency on highly mediatized and provoking topics?
The defense – that the difficulty psychiatrists have at providing an accurate diagnosis stems from the immense complexity of the system being evaluated, the human mind – is a valid one. Attempts to force such complex pathology, with all its many variables, into the check-box approach implemented in the DSM inevitably leads to problems with diagnostic reliability. Still, as psychiatrists we retain a level of expertise in assessing and treating complex disorders of the mind that no other field can claim.
The duty physicians have not only to work toward the health of their individual patients, but also to act in service of the public health and well-being of communities in which our patients live, is well established. How ethical is it then for psychiatry to absolve itself from duty when it comes to public figures at the center of shaping public opinion? There are numerous recent, high-profile instances where our expertise may have helped shine light in an otherwise murky public discussion filled with disinformation. The death of George Floyd and the year of turmoil that followed is a salient example. The conservatorship of Britney Spears and the resulting societal outcry is another. Even setting the matter of diagnosis aside, we can help illuminate the societal implications of conservatorship laws,11 in addition to providing input on how to safely and responsibly approach an individual who is in crisis, under the influence of multiple illicit substances, and possibly suffering from excited delirium.
Whether psychiatry has progressed enough as a medical specialty to trust ourselves with the option of providing professional opinions on public figures is an ongoing debate. The persistence of the Goldwater rule is a strong testament to the internal lack of confidence among psychiatrists regarding our ability to provide accurate diagnoses, act with integrity in the public space, and foster a positive public image. That lack of confidence may be well deserved. However, it is possible that our field will never go through the necessary pains of maturing as long as Section 7.3 remains in place.
Dr. Compton is a psychiatry resident at University of California, San Diego. His background includes medical education, mental health advocacy, work with underserved populations, and brain cancer research. Dr. Compton has no conflicts of interest. Dr. Badre is a clinical and forensic psychiatrist in San Diego. He holds teaching positions at the University of California, San Diego, and the University of San Diego. He teaches medical education, psychopharmacology, ethics in psychiatry, and correctional care. Dr. Badre can be reached at his website, BadreMD.com. He has no conflicts of interest.
References
1. American Psychiatric Association. The principles of medical ethics with annotations especially applicable to psychiatry. Section 7. American Psychiatric Association; 2013 edition.
2. Glass LL. The Goldwater rule is broken. Here’s how to fix it. STAT News. 2018 June 18.
3. Plymyer D. The Goldwater rule paradox. 2020 Aug 7.
4. Lieberman JA. Trump’s brain and the 25th Amendment. Vice. 2017 Sep 8.
5. Blotcky AD et al. The Goldwater rule is fine, if refined. Here’s how to do it. Psychiatric Times. 2022 Jan 6;39(1).
6. Blotcky AD and Norrholm SD. After Trump, end the Goldwater rule once and for all. New York Daily News. 2020 Dec 22.
7. Stephens B. Biden should not run again – And he should say he won’t. New York Times. 2021 Dec 14.
8. Schmaling KB et al. The positive teddy bear sign: Transitional objects in the medical setting. J Nerv Ment Dis. 1994 Dec;182(12):725.
9. Badre N et al. Psychopharmacologic management in integrated care: Challenges for residency education. Acad Psychiatry. 2015; 39(4):466-9.
10. Kraemer HC et al. DSM-5: How reliable is reliable enough? Am J Psychiatry. 2012 Jan;169(1):13-5.
11. Badre N and Compton C. Britney Spears – Reflections on conservatorship. Clinical Psychiatry News. 2021 Nov 16.
Does hustling equate to success?
Thank Goodness it’s Monday? Sincerely yours, #hustle.
The COVID-19 pandemic has given us the opportunity to reevaluate what we believe is important and valuable in our life. For some, it’s the opportunity to perform meaningful work; for others, it’s increased financial compensation; and, for the remaining, it may be autonomy (e.g., control over their time). One example of where this mindset has manifested has been in the Great Resignation.
The Great Resignation refers to the significant increase in resignations that was recorded in April 2021. Resignation rates tend to be higher in fields with high turnover rates (e.g., health care, tech) as a result of increased demand and burnout. Although hustle culture has been an ongoing trend for the last few years, the pandemic has given somewhat of a reality check of the future.
Hustle culture refers to the embracing of work as a lifestyle such that it takes over other important aspects of your life – in other words, when work-life balance becomes work-work (im)balance. It has also been aptly referred to as burnout culture or grind culture. It’s a bit ironic or counterintuitive to think that stopping work means increased productivity – but it’s true.
During my undergraduate years, I was always hustling – there wasn’t a moment where I wasn’t studying, doing research, training for my sport, or thinking about how I could do better and be better. It was all about working 24/7 – an illusion to think I was being productive. Now don’t get me wrong, I think the time and effort I invested during those years paid off. However, it also resulted in a sense of dissatisfaction; that is, dissatisfaction that I didn’t explore other potential paths, that I didn’t have the courage to try new things and to be okay with making mistakes. I had extremely narrow tunnel vision because my one and only goal was to go to medical school.
However, after entering graduate school and actually taking the time to explore other options and career pathways in health, as well as realize that nontraditional pathways are becoming more and more conventional, there is a sense of relief that “failure” is not about changing paths or making mistakes.
The part of hustle culture that has me hung up is being able to take the time to reflect whether this is what you truly want.
The pandemic has shaped a lot of the way we think, what we value, and how we proceed forward. Who we are and what we value is a continuing and ever-growing process, and how we choose to live our lives will play a part.
I’m curious to hear from you, do you believe in #hustle? Are you part of the #grind culture? Or do you believe we can achieve success, greatness, and satisfaction without the hustle culture?
Ms. Lui is an MSc candidate at the University of Toronto, and is with the Mood Disorders Psychopharmacology Unit, Toronto Western Hospital. She has received income from Braxia Scientific. A version of this article first appeared on Medscape.com.
Thank Goodness it’s Monday? Sincerely yours, #hustle.
The COVID-19 pandemic has given us the opportunity to reevaluate what we believe is important and valuable in our life. For some, it’s the opportunity to perform meaningful work; for others, it’s increased financial compensation; and, for the remaining, it may be autonomy (e.g., control over their time). One example of where this mindset has manifested has been in the Great Resignation.
The Great Resignation refers to the significant increase in resignations that was recorded in April 2021. Resignation rates tend to be higher in fields with high turnover rates (e.g., health care, tech) as a result of increased demand and burnout. Although hustle culture has been an ongoing trend for the last few years, the pandemic has given somewhat of a reality check of the future.
Hustle culture refers to the embracing of work as a lifestyle such that it takes over other important aspects of your life – in other words, when work-life balance becomes work-work (im)balance. It has also been aptly referred to as burnout culture or grind culture. It’s a bit ironic or counterintuitive to think that stopping work means increased productivity – but it’s true.
During my undergraduate years, I was always hustling – there wasn’t a moment where I wasn’t studying, doing research, training for my sport, or thinking about how I could do better and be better. It was all about working 24/7 – an illusion to think I was being productive. Now don’t get me wrong, I think the time and effort I invested during those years paid off. However, it also resulted in a sense of dissatisfaction; that is, dissatisfaction that I didn’t explore other potential paths, that I didn’t have the courage to try new things and to be okay with making mistakes. I had extremely narrow tunnel vision because my one and only goal was to go to medical school.
However, after entering graduate school and actually taking the time to explore other options and career pathways in health, as well as realize that nontraditional pathways are becoming more and more conventional, there is a sense of relief that “failure” is not about changing paths or making mistakes.
The part of hustle culture that has me hung up is being able to take the time to reflect whether this is what you truly want.
The pandemic has shaped a lot of the way we think, what we value, and how we proceed forward. Who we are and what we value is a continuing and ever-growing process, and how we choose to live our lives will play a part.
I’m curious to hear from you, do you believe in #hustle? Are you part of the #grind culture? Or do you believe we can achieve success, greatness, and satisfaction without the hustle culture?
Ms. Lui is an MSc candidate at the University of Toronto, and is with the Mood Disorders Psychopharmacology Unit, Toronto Western Hospital. She has received income from Braxia Scientific. A version of this article first appeared on Medscape.com.
Thank Goodness it’s Monday? Sincerely yours, #hustle.
The COVID-19 pandemic has given us the opportunity to reevaluate what we believe is important and valuable in our life. For some, it’s the opportunity to perform meaningful work; for others, it’s increased financial compensation; and, for the remaining, it may be autonomy (e.g., control over their time). One example of where this mindset has manifested has been in the Great Resignation.
The Great Resignation refers to the significant increase in resignations that was recorded in April 2021. Resignation rates tend to be higher in fields with high turnover rates (e.g., health care, tech) as a result of increased demand and burnout. Although hustle culture has been an ongoing trend for the last few years, the pandemic has given somewhat of a reality check of the future.
Hustle culture refers to the embracing of work as a lifestyle such that it takes over other important aspects of your life – in other words, when work-life balance becomes work-work (im)balance. It has also been aptly referred to as burnout culture or grind culture. It’s a bit ironic or counterintuitive to think that stopping work means increased productivity – but it’s true.
During my undergraduate years, I was always hustling – there wasn’t a moment where I wasn’t studying, doing research, training for my sport, or thinking about how I could do better and be better. It was all about working 24/7 – an illusion to think I was being productive. Now don’t get me wrong, I think the time and effort I invested during those years paid off. However, it also resulted in a sense of dissatisfaction; that is, dissatisfaction that I didn’t explore other potential paths, that I didn’t have the courage to try new things and to be okay with making mistakes. I had extremely narrow tunnel vision because my one and only goal was to go to medical school.
However, after entering graduate school and actually taking the time to explore other options and career pathways in health, as well as realize that nontraditional pathways are becoming more and more conventional, there is a sense of relief that “failure” is not about changing paths or making mistakes.
The part of hustle culture that has me hung up is being able to take the time to reflect whether this is what you truly want.
The pandemic has shaped a lot of the way we think, what we value, and how we proceed forward. Who we are and what we value is a continuing and ever-growing process, and how we choose to live our lives will play a part.
I’m curious to hear from you, do you believe in #hustle? Are you part of the #grind culture? Or do you believe we can achieve success, greatness, and satisfaction without the hustle culture?
Ms. Lui is an MSc candidate at the University of Toronto, and is with the Mood Disorders Psychopharmacology Unit, Toronto Western Hospital. She has received income from Braxia Scientific. A version of this article first appeared on Medscape.com.








