User login
Is pediatric subspecialty training financially worth it?
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pursuing fellowship training is often financially costly in terms of lifetime earnings, compared with starting a career as a general pediatrician immediately after residency, a report suggests.
Researchers found that most pediatric subspecialists – including those practicing neurology, pulmonology, and adolescent medicine – do not see a financial return from additional training because of the delays in receiving increased compensation and the repayment of educational debt.
“Most pediatric subspecialists don’t experience a relative increase in compensation after training compared to a general pediatrician, so there isn’t a financial benefit to additional training,” lead author Eva Catenaccio, MD, from the division of pediatric neurology, department of neurology, Johns Hopkins University, Baltimore, told this news organization.
The findings, published online March 8 in Pediatrics, contribute to the ongoing debate about the length of pediatric fellowship training programs. The data also provide evidence for the potential effect of a pediatric subspecialty loan repayment program.
Pediatric subspecialty training rarely pays off
However, not all practitioners in pediatric subspecialties would find themselves in the red relative to their generalist peers. Three subspecialties had a positive financial return: cardiology, critical care, and neonatology. Dr. Catenaccio explained that this may be because these subspecialties tend to be “inpatient procedure oriented, which are often more [lucrative] than outpatient cognitive–oriented subspecialties, such as pediatric infectious diseases, endocrinology, or adolescent medicine.”
Enrolling in a pediatric fellowship program resulted in lifetime financial returns that ranged from an increase of $852,129 for cardiology, relative to general pediatrics, to a loss of $1,594,366 for adolescent medicine, researchers found.
For the study, researchers calculated the financial returns of 15 pediatric subspecialties – emergency medicine, neurology, cardiology, critical care, neonatology, hematology and oncology, pulmonology, hospitalist medicine, allergy and immunology, gastroenterology, rheumatology, nephrology, adolescent medicine, infectious diseases, and endocrinology – in comparison with returns of private practice general pediatrics on the basis of 2018-2019 data on fellowship stipends, compensation, and educational debt.
They obtained most of the data from the Association of American Medical Colleges Survey of Resident/Fellow Stipends and Benefits, AAMC’s annual Medical School Faculty Salary Report, and the AAMC Medical School Graduation Questionnaire.
Richard Mink, MD, department of pediatrics, Harbor-UCLA Medical Center, Torrance, Calif., noted that it would have been helpful to have also compared the lifetime earnings of practitioners in pediatric subspecialties to academic general pediatricians and not just those in private practice.
The financial gap has worsened
To better understand which aspects of fellowship training have the greatest effect on lifetime compensation, Dr. Catenaccio and colleagues evaluated the potential effects of shortening fellowship length, eliminating school debt, and implementing a federal loan repayment plan. These changes enhanced the returns of cardiology, critical care, and neonatology – subspecialties that had already seen financial returns before these changes – and resulted in a positive financial return for emergency medicine.
The changes also narrowed the financial gap between subspecialties and general pediatrics. However, the remaining subspecialties still earned less than private practice pediatrics.
The new study is an update to a 2011 report, which reflected 2007-2008 data for 11 subspecialties. This time around, the researchers included the subspecialty of hospitalist medicine, which was approved as a board-certified subspecialty by the American Board of Pediatrics in 2014, as well as neurology, allergy and immunology, and adolescent medicine.
“I was most surprised that the additional pediatric subspecialties we included since the 2011 report followed the same general trend, with pediatric subspecialty training having a lower lifetime earning potential than general pediatrics,” Dr. Catenaccio said.
Comparing results from the two study periods showed that the financial gap between general pediatrics and subspecialty pediatrics worsened over time. For example, the financial return for pediatric endocrinology decreased an additional $500,000 between 2007 and 2018.
The researchers believe a combination of increased educational debt burden, slow growth in compensation, and changing interest rates over time have caused the financial differences between general pediatrics and subspecialty pediatrics to become more pronounced.
‘Pediatric subspecialty training is worth it!’
Despite the financial gaps, Dr. Catenaccio and colleagues say pediatric subspecialty training is still worthwhile but that policymakers should address these financial differences to help guide workforce distribution in a way that meets the needs of patients.
“I think pediatric subspecialty training is worth it,” said Dr. Catenaccio, who’s pursuing pediatric subspecialty training. “There are so many factors that go into choosing a specialty or subspecialty in medicine, including the desire to care for a particular patient population, interest in certain diseases or organ systems, lifestyle considerations, and research opportunities.”
But it’s also important for trainees to be aware of economic considerations in their decision-making.
Dr. Mink, who wrote an accompanying commentary, agrees that young clinicians should not make career decisions on the basis of metrics such as lifetime earning measures.
“I think people who go into pediatrics have decided that money is not the driving force,” said Dr. Mink. He noted that pediatricians are usually not paid well, compared with other specialists. “To me the important thing is you have to like what you’re doing.”
A 2020 study found that trainees who chose a career in pediatric pulmonology, a subspecialty, said that financial considerations were not the driving factor in their decision-making. Nevertheless, Dr. Mink also believes young clinicians should take into account their educational debt.
The further widening of the financial gap between general pediatrics and pediatric subspecialties could lead to shortages in the pediatric subspecialty workforce.
The authors and Dr. Mink have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Smartphone ‘addiction’ tied to poor sleep in young adults
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Smartphone “addiction” may explain poor sleep quality in a significant proportion of young adults, new research suggests.
Investigators found that almost 40% of adults aged 18-30 years who self-reported excessive smartphone use also reported poor sleep.
“Our study provides further support to the growing body of evidence that smartphone addiction has a deleterious impact on sleep,” wrote the researchers.
The study was published online March 2 in Frontiers of Psychiatry.
Not a clinical diagnosis
Smartphone addiction is not formally recognized as a clinical diagnosis, but it’s an “active” area of research, lead investigator Ben Carter, PhD, King’s College London, noted in the report.
In a cross-sectional survey, 1,043 college students (aged 18-30 years, 73% women) completed the 10-question validated Smartphone Addiction Scale Short Version (SAS-SV) and the adapted Pittsburgh Sleep Quality Score Index.
On the SAS-SV, 406 students (38.9%) reported “addiction” to their smartphones. This estimated prevalence is consistent with other reported studies in young adult populations globally, which is in the range of 30%-45%, the researchers noted.
Overall, 61.6% of participants surveyed reported poor sleep; among those who reported smartphone addiction, 68.7% had poor sleep quality, vs. 57.1% of those who did not report smartphone addiction.
In multivariable analysis that adjusted for a variety of relevant factors, among those for whom there was evidence of smartphone addiction, the odds of poor sleep were increased by 41% (adjusted odds ratio [aOR] = 1.41; 95% confidence interval, 1.06-1.87, P = .018).
The findings also suggest that a greater amount of time spent using the phone and greater use late at night can raise the risk for smartphone addiction.
“Should smartphone addiction become firmly established as a focus of clinical concern, those using their phones after midnight or using their phones for four or more hours per day are likely to be at high risk, and should guide administration of the SAS-SV,” the researchers wrote.
Caveats, cautions, and concerns
Reached for comment, Paul Weigle, MD, psychiatrist with Hartford HealthCare and Hartford (Conn.) Hospital, and member of the American Academy of Child and Adolescent Psychiatry, said the finding of a relationship between addictive smartphone usage and poor sleep quality is not surprising.
“Great increases in adolescent screen media habits in recent decades have seen a concurrent increase in rates of insomnia among this population,” he said in an interview.
Dr. Weigle also noted that young people who use the phone excessively often do so in bed, “which decreases sleep onset by disrupting conditioning (the tendency for our bodies to relate bed with sleep) and by increasing physiological arousal, which makes it more difficult to fall asleep. The blue light from smartphones used at night disrupts our body’s natural circadian rhythms, confusing our brains regarding whether it is night or day, and further worsens sleep.”
Dr. Weigle said in an interview that some of his patients come to him seeking sleep medications, although the best treatment is to perform a “smartphone-ectomy” every evening.
Teenage patients will “beg, borrow, or steal” to be allowed to keep their phones by the bed with the promise not to use them overnight. Three-quarters of the time, when the parents are able to charge the phone in another room, “the sleep problem resolves,” Dr. Weigle said.
One caveat, he said, is that it’s “somewhat unclear whether this is best classified as an addiction or simply a seriously problematic habit. Either way, this type of habit causes a great deal of distress and dysfunction in the lives of those it affects, so it is important to understand,” he said.
In a statement, Bob Patton, PhD, lecturer in clinical psychology, University of Surrey, Guildford, England, noted that this is a cross-sectional study “and as such cannot lead to any firm conclusions about phone usage as the cause of reduced sleep quality.
“It does, however, provide some compelling evidence,” Dr. Patton said, “that the nature of smartphone usage and its related consequences are important considerations in addressing the emerging phenomenon of ‘smartphone addiction.’ ”
Also weighing in, Andrew Przybylski, PhD, director of research, Oxford (England) Internet Institute, University of Oxford, said by any global health body and is not a psychiatric disorder.
“The study is a correlational analysis of a sample of participants recruited on university campuses and therefore only reflects the experiences of those who had the purpose of the study explained to them. It can say nothing about behaviors in the general population,” Dr. Przybylski said in a statement.
“Readers should be cautious of making any firm conclusions about the impact of smartphone use in the general population, or the idea that they’re addictive in any objective sense, on the basis of this work,” he added. The study had no specific funding. Dr. Carter, Dr. Weigle, Dr. Patton, and Dr. Przybylski have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Is it possible to classify dermatologists and internists into different patterns of prescribing behavior?
An exploratory analysis recently published in the Journal of the American Academy of Dermatology examines whether it is possible to classify dermatologists and internists into different patterns of prescribing behavior for patients with acne.
“Prior research has highlighted that prescribing for acne may not be aligned with guideline recommendations, including the overuse of oral antibiotics and lack of use of concomitant topical medications such as topical retinoids,” the study’s corresponding author, John S. Barbieri, MD, MBA, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview.
“In addition, there is substantial variation in prescribing practices among clinicians. . By identifying such groups, it would facilitate future qualitative interviews to understand factors that might contribute to clinicians having certain prescribing patterns, which could help guide implementation science work to better align practices with evidence and guidelines.”
For the study, which appeared online on March 1, Dr. Barbieri and colleague David J. Margolis, MD, PhD, professor of dermatology and epidemiology at the University of Pennsylvania, evaluated all clinical encounters associated with an ICD-9 or ICD-10 code for acne that occurred in the university’s departments of dermatology and internal medicine between Jan. 1, 2011, and Dec. 31, 2019. They used a machine-learning method known as k-means clustering to cluster clinicians based on their relative use of acne medications, as well as the ratio of spironolactone versus tetracycline use among female patients and stratified their analyses by specialty.
Of the 116 dermatologists included in the analysis, the researchers identified three clusters. The first cluster included 17 dermatologists (14.7%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and low use of spironolactone, compared with oral antibiotics, among women with acne. Physicians in this cluster were more likely to be male and to have more years in practice.
The second cluster included 46 dermatologists (39.6%) and was marked by high use of spironolactone and low use of isotretinoin. The third cluster included 53 dermatologists (45.7%) and was characterized by high use of topical retinoids and frequent use of systemic medications.
Of the 86 internists included in the study, the researchers identified three clusters. The first cluster included 39 internists (45.4%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and limited use of spironolactone. The second cluster included 34 internists (39.5%) and was marked by low use of topical retinoids and systemic medications. The third cluster included 13 clinicians (15.1%), most of whom were nurse practitioners, physician assistants, and other advanced practice providers. This cluster was characterized by high use of topical retinoids and relatively high use of spironolactone.
“There are likely opportunities to improve the use of topical retinoids by internists caring for patients with acne, since these are a first-line treatment option that may be underutilized by internists,” Dr. Barbieri said in the interview. “Future work is needed to identify underlying factors associated with different prescribing phenotypes among both dermatologists and internists. By understanding these factors, we can develop implementation science efforts to align prescribing behavior with best practices based on the guidelines and available evidence.”
He acknowledged certain limitations of the analysis, including its single-center design and the lack of data on patient characteristics. “Future studies are needed to examine whether our results generalize to other settings,” he said.
Dr. Barbieri disclosed that he receives partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. The authors had no other disclosures.
An exploratory analysis recently published in the Journal of the American Academy of Dermatology examines whether it is possible to classify dermatologists and internists into different patterns of prescribing behavior for patients with acne.
“Prior research has highlighted that prescribing for acne may not be aligned with guideline recommendations, including the overuse of oral antibiotics and lack of use of concomitant topical medications such as topical retinoids,” the study’s corresponding author, John S. Barbieri, MD, MBA, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview.
“In addition, there is substantial variation in prescribing practices among clinicians. . By identifying such groups, it would facilitate future qualitative interviews to understand factors that might contribute to clinicians having certain prescribing patterns, which could help guide implementation science work to better align practices with evidence and guidelines.”
For the study, which appeared online on March 1, Dr. Barbieri and colleague David J. Margolis, MD, PhD, professor of dermatology and epidemiology at the University of Pennsylvania, evaluated all clinical encounters associated with an ICD-9 or ICD-10 code for acne that occurred in the university’s departments of dermatology and internal medicine between Jan. 1, 2011, and Dec. 31, 2019. They used a machine-learning method known as k-means clustering to cluster clinicians based on their relative use of acne medications, as well as the ratio of spironolactone versus tetracycline use among female patients and stratified their analyses by specialty.
Of the 116 dermatologists included in the analysis, the researchers identified three clusters. The first cluster included 17 dermatologists (14.7%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and low use of spironolactone, compared with oral antibiotics, among women with acne. Physicians in this cluster were more likely to be male and to have more years in practice.
The second cluster included 46 dermatologists (39.6%) and was marked by high use of spironolactone and low use of isotretinoin. The third cluster included 53 dermatologists (45.7%) and was characterized by high use of topical retinoids and frequent use of systemic medications.
Of the 86 internists included in the study, the researchers identified three clusters. The first cluster included 39 internists (45.4%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and limited use of spironolactone. The second cluster included 34 internists (39.5%) and was marked by low use of topical retinoids and systemic medications. The third cluster included 13 clinicians (15.1%), most of whom were nurse practitioners, physician assistants, and other advanced practice providers. This cluster was characterized by high use of topical retinoids and relatively high use of spironolactone.
“There are likely opportunities to improve the use of topical retinoids by internists caring for patients with acne, since these are a first-line treatment option that may be underutilized by internists,” Dr. Barbieri said in the interview. “Future work is needed to identify underlying factors associated with different prescribing phenotypes among both dermatologists and internists. By understanding these factors, we can develop implementation science efforts to align prescribing behavior with best practices based on the guidelines and available evidence.”
He acknowledged certain limitations of the analysis, including its single-center design and the lack of data on patient characteristics. “Future studies are needed to examine whether our results generalize to other settings,” he said.
Dr. Barbieri disclosed that he receives partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. The authors had no other disclosures.
An exploratory analysis recently published in the Journal of the American Academy of Dermatology examines whether it is possible to classify dermatologists and internists into different patterns of prescribing behavior for patients with acne.
“Prior research has highlighted that prescribing for acne may not be aligned with guideline recommendations, including the overuse of oral antibiotics and lack of use of concomitant topical medications such as topical retinoids,” the study’s corresponding author, John S. Barbieri, MD, MBA, of the department of dermatology at the University of Pennsylvania, Philadelphia, said in an interview.
“In addition, there is substantial variation in prescribing practices among clinicians. . By identifying such groups, it would facilitate future qualitative interviews to understand factors that might contribute to clinicians having certain prescribing patterns, which could help guide implementation science work to better align practices with evidence and guidelines.”
For the study, which appeared online on March 1, Dr. Barbieri and colleague David J. Margolis, MD, PhD, professor of dermatology and epidemiology at the University of Pennsylvania, evaluated all clinical encounters associated with an ICD-9 or ICD-10 code for acne that occurred in the university’s departments of dermatology and internal medicine between Jan. 1, 2011, and Dec. 31, 2019. They used a machine-learning method known as k-means clustering to cluster clinicians based on their relative use of acne medications, as well as the ratio of spironolactone versus tetracycline use among female patients and stratified their analyses by specialty.
Of the 116 dermatologists included in the analysis, the researchers identified three clusters. The first cluster included 17 dermatologists (14.7%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and low use of spironolactone, compared with oral antibiotics, among women with acne. Physicians in this cluster were more likely to be male and to have more years in practice.
The second cluster included 46 dermatologists (39.6%) and was marked by high use of spironolactone and low use of isotretinoin. The third cluster included 53 dermatologists (45.7%) and was characterized by high use of topical retinoids and frequent use of systemic medications.
Of the 86 internists included in the study, the researchers identified three clusters. The first cluster included 39 internists (45.4%) and was characterized by low use of topical retinoids, high use of oral tetracycline, and limited use of spironolactone. The second cluster included 34 internists (39.5%) and was marked by low use of topical retinoids and systemic medications. The third cluster included 13 clinicians (15.1%), most of whom were nurse practitioners, physician assistants, and other advanced practice providers. This cluster was characterized by high use of topical retinoids and relatively high use of spironolactone.
“There are likely opportunities to improve the use of topical retinoids by internists caring for patients with acne, since these are a first-line treatment option that may be underutilized by internists,” Dr. Barbieri said in the interview. “Future work is needed to identify underlying factors associated with different prescribing phenotypes among both dermatologists and internists. By understanding these factors, we can develop implementation science efforts to align prescribing behavior with best practices based on the guidelines and available evidence.”
He acknowledged certain limitations of the analysis, including its single-center design and the lack of data on patient characteristics. “Future studies are needed to examine whether our results generalize to other settings,” he said.
Dr. Barbieri disclosed that he receives partial salary support through a Pfizer Fellowship in Dermatology Patient Oriented Research grant to the Trustees of the University of Pennsylvania. The authors had no other disclosures.
Vagisil offered teens a vaginal ‘glow up.’ Docs cry foul
Late one night in early February, Jen Gunter, MD, was scrolling online when she discovered a new “feminine hygiene” product being marketed for teen girls. The new vanilla clementine scented wipes and cleansers with confetti-colored packaging and a cute name (OMV!) irked Dr. Gunter because they are designed for girls to use to “freshen” their vaginal area.
Dr. Gunter, a San Francisco-based gynecologist and author of “The Vagina Bible,” has built a reputation as a fierce advocate for women’s health and debunker of pseudoscience. She has called out jade eggs and “detox pearls” and various other items that promise to improve the vagina but that she and other doctors warn could actually be harmful. And, in her view, this product is no different.
She fired off a tweet that became the first volley in a vociferous social media countercampaign: “Hey @vagisil going to call you out here for this predatory line of products aimed at teen girls. Why do you think teen vulvas need special cleaning? To be prepped for men? Because they are dirty. Anxiously awaiting your answer as are all my followers.”
Vagisil responded on Instagram that “we want to clarify any confusion or the underlying belief that OMV! was developed because there is something wrong with teens or that vulvas/vaginas are inherently dirty. That is not the case. All-Day Fresh Wash is an all-over body wash, that is safe, gentle, and pH-balanced for sensitive vulvar area skin.”
Dr. Gunter’s Feb. 4 tweet attracted more than 8,300 likes, 1,300 retweets and hundreds of comments, but that was just the beginning. Dr. Gunter has continued to tweet about the OMV! product line – and has inspired dozens of other gynecologists to join in.
‘Your vagina is fine’
Dr. Gunter and other gynecologists have long delivered the message that water alone is sufficient to cleanse the vulvar area and that the vagina itself is self-cleaning. Research into the vaginal microbiome reveals the role of lactobacilli in preventing urogenital diseases. “Disturbances in your vagina microbiome are hard to undo,” says Jocelyn Fitzgerald, MD, a urogynecologist and pelvic reconstructive surgeon at Magee-Womens Hospital at the University of Pittsburgh Medical Center.
To underscore that message, Dr. Fitzgerald recently tweeted in support of Dr. Gunter’s Twitter thread: “Honestly, the @vagisil marketing campaign is a brilliant one because using their products while your vagina is perfectly fine will destroy your microbiome, give you real Bacterial Vaginosis, and prompt you to buy more Vagisil. DON’T FALL FOR IT GIRLS YOUR VAGINA IS FINE.”
In an emailed response to this news organization, a Vagisil spokesperson said, “We follow industry best practices for testing and OMV! products are rigorously assessed for safety and quality. In addition, we work with respected, independent clinical labs that follow strict testing protocols, using board-certified gynecologists and dermatologists to test our products before launch.”
However, beyond the potential for irritation or misuse, the gynecologists zeroed in on the underlying message that girls would feel more confident if they used the wipes and cleanser. For example, the company suggested that teens could use the wipes to get rid of “period funk.”
“There is no such thing as period funk!” gynecologist Danielle Jones, MD, exclaimed in a video on YouTube, where she has a channel called Mama Doctor Jones – with 700,000 subscribers. “All you need is ordinary hygiene. Period funk is not a thing! And if you feel like something is going on because there’s an odor that is abnormal, you need to talk to your doctor.”
Adult women often use wipes and special cleansers in the vaginal area. An online survey of 1,435 Canadian women, published in BMC Women’s Health in 2018, found 42% had used vaginal wipes, 12% had used vaginal washes or cleansers – and 4% had used them internally.
When it launched OMV! in July, Vagisil said it had engaged 2,500 teens and their mothers in creating the product, which it said was “designed to meet the cleansing and care needs of a new generation of young women.”
That extension of a product most commonly used by adult women to teenagers – who often feel self-conscious about their bodies – is exactly what bothers Dr. Gunter. “BTW I am sorry I am subjecting you all to my @vagisil outrage, but preying on teens and amplifying patriarchal shame of normal bodily functions to sell an irritating product is not acceptable. I’m not stopping until they take that OMV! product line down everywhere,” she said in a Feb. 8 tweet that attracted more than 7,900 likes.
No ‘glow up’ needed
Dr. Gunter’s tweets tapped into collective anger over the shaming of women’s bodies. The OMV! marketing suggested that teens could get a “glow up” with the products.
“Your vulva doesn’t need a ‘glow up.’ It’s fine like it is. And if it’s not, talk to your doctor,” Dr. Jones said in her Feb. 8 video, which has had almost 350,000 views, with 28,000 likes and only 149 dislikes.
“They’re very clearly pathologizing normal physiology,” Dr. Jones says. “They’re creating language that makes people feel as though their normal bodily functions have to be somehow fixed or changed.”
Dr. Gunter says she specifically wanted to prevent Vagisil from leveraging social media to influence teen girls. With her stream of tweets and support from colleagues around the country, she has sparked a prolonged online conversation.
“I am encouraged by the strong response on social media from both other enraged ob.gyns. and health care professionals as well the response from a lot of women and men,” Dr. Gunter said in an interview. “We have effectively blocked [Vagisil] from using social media.”
In its response to this news organization, Vagisil noted, “We are a brand run by women with daughters of our own.” While defending the products, Vagisil acknowledged the criticisms: “We are always listening to our consumers and our expert partners so that we continuously evolve. We appreciate the perspective that our language choice surrounding periods may perpetuate an old idea and have already begun to make changes to address this.”
Dr. Gunter says she plans to stay on topic. “Given the number of people outraged, I suspect if they venture out on social media again the reaction will be swift,” she said. “Hopefully we have made OMV! toxic for influencers as well.”
In fact, she’s ready to take on “the entire predatory feminine hygiene market. I’m sick of their false claims about balancing pH and not-so-subtle suggestions that vaginas and vulvas and menstruation stink. These products cause psychological harm as well as physical harm from their irritants,” she said.
A version of this article first appeared on Medscape.com.
Late one night in early February, Jen Gunter, MD, was scrolling online when she discovered a new “feminine hygiene” product being marketed for teen girls. The new vanilla clementine scented wipes and cleansers with confetti-colored packaging and a cute name (OMV!) irked Dr. Gunter because they are designed for girls to use to “freshen” their vaginal area.
Dr. Gunter, a San Francisco-based gynecologist and author of “The Vagina Bible,” has built a reputation as a fierce advocate for women’s health and debunker of pseudoscience. She has called out jade eggs and “detox pearls” and various other items that promise to improve the vagina but that she and other doctors warn could actually be harmful. And, in her view, this product is no different.
She fired off a tweet that became the first volley in a vociferous social media countercampaign: “Hey @vagisil going to call you out here for this predatory line of products aimed at teen girls. Why do you think teen vulvas need special cleaning? To be prepped for men? Because they are dirty. Anxiously awaiting your answer as are all my followers.”
Vagisil responded on Instagram that “we want to clarify any confusion or the underlying belief that OMV! was developed because there is something wrong with teens or that vulvas/vaginas are inherently dirty. That is not the case. All-Day Fresh Wash is an all-over body wash, that is safe, gentle, and pH-balanced for sensitive vulvar area skin.”
Dr. Gunter’s Feb. 4 tweet attracted more than 8,300 likes, 1,300 retweets and hundreds of comments, but that was just the beginning. Dr. Gunter has continued to tweet about the OMV! product line – and has inspired dozens of other gynecologists to join in.
‘Your vagina is fine’
Dr. Gunter and other gynecologists have long delivered the message that water alone is sufficient to cleanse the vulvar area and that the vagina itself is self-cleaning. Research into the vaginal microbiome reveals the role of lactobacilli in preventing urogenital diseases. “Disturbances in your vagina microbiome are hard to undo,” says Jocelyn Fitzgerald, MD, a urogynecologist and pelvic reconstructive surgeon at Magee-Womens Hospital at the University of Pittsburgh Medical Center.
To underscore that message, Dr. Fitzgerald recently tweeted in support of Dr. Gunter’s Twitter thread: “Honestly, the @vagisil marketing campaign is a brilliant one because using their products while your vagina is perfectly fine will destroy your microbiome, give you real Bacterial Vaginosis, and prompt you to buy more Vagisil. DON’T FALL FOR IT GIRLS YOUR VAGINA IS FINE.”
In an emailed response to this news organization, a Vagisil spokesperson said, “We follow industry best practices for testing and OMV! products are rigorously assessed for safety and quality. In addition, we work with respected, independent clinical labs that follow strict testing protocols, using board-certified gynecologists and dermatologists to test our products before launch.”
However, beyond the potential for irritation or misuse, the gynecologists zeroed in on the underlying message that girls would feel more confident if they used the wipes and cleanser. For example, the company suggested that teens could use the wipes to get rid of “period funk.”
“There is no such thing as period funk!” gynecologist Danielle Jones, MD, exclaimed in a video on YouTube, where she has a channel called Mama Doctor Jones – with 700,000 subscribers. “All you need is ordinary hygiene. Period funk is not a thing! And if you feel like something is going on because there’s an odor that is abnormal, you need to talk to your doctor.”
Adult women often use wipes and special cleansers in the vaginal area. An online survey of 1,435 Canadian women, published in BMC Women’s Health in 2018, found 42% had used vaginal wipes, 12% had used vaginal washes or cleansers – and 4% had used them internally.
When it launched OMV! in July, Vagisil said it had engaged 2,500 teens and their mothers in creating the product, which it said was “designed to meet the cleansing and care needs of a new generation of young women.”
That extension of a product most commonly used by adult women to teenagers – who often feel self-conscious about their bodies – is exactly what bothers Dr. Gunter. “BTW I am sorry I am subjecting you all to my @vagisil outrage, but preying on teens and amplifying patriarchal shame of normal bodily functions to sell an irritating product is not acceptable. I’m not stopping until they take that OMV! product line down everywhere,” she said in a Feb. 8 tweet that attracted more than 7,900 likes.
No ‘glow up’ needed
Dr. Gunter’s tweets tapped into collective anger over the shaming of women’s bodies. The OMV! marketing suggested that teens could get a “glow up” with the products.
“Your vulva doesn’t need a ‘glow up.’ It’s fine like it is. And if it’s not, talk to your doctor,” Dr. Jones said in her Feb. 8 video, which has had almost 350,000 views, with 28,000 likes and only 149 dislikes.
“They’re very clearly pathologizing normal physiology,” Dr. Jones says. “They’re creating language that makes people feel as though their normal bodily functions have to be somehow fixed or changed.”
Dr. Gunter says she specifically wanted to prevent Vagisil from leveraging social media to influence teen girls. With her stream of tweets and support from colleagues around the country, she has sparked a prolonged online conversation.
“I am encouraged by the strong response on social media from both other enraged ob.gyns. and health care professionals as well the response from a lot of women and men,” Dr. Gunter said in an interview. “We have effectively blocked [Vagisil] from using social media.”
In its response to this news organization, Vagisil noted, “We are a brand run by women with daughters of our own.” While defending the products, Vagisil acknowledged the criticisms: “We are always listening to our consumers and our expert partners so that we continuously evolve. We appreciate the perspective that our language choice surrounding periods may perpetuate an old idea and have already begun to make changes to address this.”
Dr. Gunter says she plans to stay on topic. “Given the number of people outraged, I suspect if they venture out on social media again the reaction will be swift,” she said. “Hopefully we have made OMV! toxic for influencers as well.”
In fact, she’s ready to take on “the entire predatory feminine hygiene market. I’m sick of their false claims about balancing pH and not-so-subtle suggestions that vaginas and vulvas and menstruation stink. These products cause psychological harm as well as physical harm from their irritants,” she said.
A version of this article first appeared on Medscape.com.
Late one night in early February, Jen Gunter, MD, was scrolling online when she discovered a new “feminine hygiene” product being marketed for teen girls. The new vanilla clementine scented wipes and cleansers with confetti-colored packaging and a cute name (OMV!) irked Dr. Gunter because they are designed for girls to use to “freshen” their vaginal area.
Dr. Gunter, a San Francisco-based gynecologist and author of “The Vagina Bible,” has built a reputation as a fierce advocate for women’s health and debunker of pseudoscience. She has called out jade eggs and “detox pearls” and various other items that promise to improve the vagina but that she and other doctors warn could actually be harmful. And, in her view, this product is no different.
She fired off a tweet that became the first volley in a vociferous social media countercampaign: “Hey @vagisil going to call you out here for this predatory line of products aimed at teen girls. Why do you think teen vulvas need special cleaning? To be prepped for men? Because they are dirty. Anxiously awaiting your answer as are all my followers.”
Vagisil responded on Instagram that “we want to clarify any confusion or the underlying belief that OMV! was developed because there is something wrong with teens or that vulvas/vaginas are inherently dirty. That is not the case. All-Day Fresh Wash is an all-over body wash, that is safe, gentle, and pH-balanced for sensitive vulvar area skin.”
Dr. Gunter’s Feb. 4 tweet attracted more than 8,300 likes, 1,300 retweets and hundreds of comments, but that was just the beginning. Dr. Gunter has continued to tweet about the OMV! product line – and has inspired dozens of other gynecologists to join in.
‘Your vagina is fine’
Dr. Gunter and other gynecologists have long delivered the message that water alone is sufficient to cleanse the vulvar area and that the vagina itself is self-cleaning. Research into the vaginal microbiome reveals the role of lactobacilli in preventing urogenital diseases. “Disturbances in your vagina microbiome are hard to undo,” says Jocelyn Fitzgerald, MD, a urogynecologist and pelvic reconstructive surgeon at Magee-Womens Hospital at the University of Pittsburgh Medical Center.
To underscore that message, Dr. Fitzgerald recently tweeted in support of Dr. Gunter’s Twitter thread: “Honestly, the @vagisil marketing campaign is a brilliant one because using their products while your vagina is perfectly fine will destroy your microbiome, give you real Bacterial Vaginosis, and prompt you to buy more Vagisil. DON’T FALL FOR IT GIRLS YOUR VAGINA IS FINE.”
In an emailed response to this news organization, a Vagisil spokesperson said, “We follow industry best practices for testing and OMV! products are rigorously assessed for safety and quality. In addition, we work with respected, independent clinical labs that follow strict testing protocols, using board-certified gynecologists and dermatologists to test our products before launch.”
However, beyond the potential for irritation or misuse, the gynecologists zeroed in on the underlying message that girls would feel more confident if they used the wipes and cleanser. For example, the company suggested that teens could use the wipes to get rid of “period funk.”
“There is no such thing as period funk!” gynecologist Danielle Jones, MD, exclaimed in a video on YouTube, where she has a channel called Mama Doctor Jones – with 700,000 subscribers. “All you need is ordinary hygiene. Period funk is not a thing! And if you feel like something is going on because there’s an odor that is abnormal, you need to talk to your doctor.”
Adult women often use wipes and special cleansers in the vaginal area. An online survey of 1,435 Canadian women, published in BMC Women’s Health in 2018, found 42% had used vaginal wipes, 12% had used vaginal washes or cleansers – and 4% had used them internally.
When it launched OMV! in July, Vagisil said it had engaged 2,500 teens and their mothers in creating the product, which it said was “designed to meet the cleansing and care needs of a new generation of young women.”
That extension of a product most commonly used by adult women to teenagers – who often feel self-conscious about their bodies – is exactly what bothers Dr. Gunter. “BTW I am sorry I am subjecting you all to my @vagisil outrage, but preying on teens and amplifying patriarchal shame of normal bodily functions to sell an irritating product is not acceptable. I’m not stopping until they take that OMV! product line down everywhere,” she said in a Feb. 8 tweet that attracted more than 7,900 likes.
No ‘glow up’ needed
Dr. Gunter’s tweets tapped into collective anger over the shaming of women’s bodies. The OMV! marketing suggested that teens could get a “glow up” with the products.
“Your vulva doesn’t need a ‘glow up.’ It’s fine like it is. And if it’s not, talk to your doctor,” Dr. Jones said in her Feb. 8 video, which has had almost 350,000 views, with 28,000 likes and only 149 dislikes.
“They’re very clearly pathologizing normal physiology,” Dr. Jones says. “They’re creating language that makes people feel as though their normal bodily functions have to be somehow fixed or changed.”
Dr. Gunter says she specifically wanted to prevent Vagisil from leveraging social media to influence teen girls. With her stream of tweets and support from colleagues around the country, she has sparked a prolonged online conversation.
“I am encouraged by the strong response on social media from both other enraged ob.gyns. and health care professionals as well the response from a lot of women and men,” Dr. Gunter said in an interview. “We have effectively blocked [Vagisil] from using social media.”
In its response to this news organization, Vagisil noted, “We are a brand run by women with daughters of our own.” While defending the products, Vagisil acknowledged the criticisms: “We are always listening to our consumers and our expert partners so that we continuously evolve. We appreciate the perspective that our language choice surrounding periods may perpetuate an old idea and have already begun to make changes to address this.”
Dr. Gunter says she plans to stay on topic. “Given the number of people outraged, I suspect if they venture out on social media again the reaction will be swift,” she said. “Hopefully we have made OMV! toxic for influencers as well.”
In fact, she’s ready to take on “the entire predatory feminine hygiene market. I’m sick of their false claims about balancing pH and not-so-subtle suggestions that vaginas and vulvas and menstruation stink. These products cause psychological harm as well as physical harm from their irritants,” she said.
A version of this article first appeared on Medscape.com.
‘I think I’m transgender’: A clinician’s guide to next steps
A 16-year-old patient sits in front of you and says, “I think I’m transgender.”
What do you do?
Whether you are an endocrinologist, family physician, pediatrician, or emergency physician, it’s probably a situation for which medical school education did not sufficiently prepare you. What you know is that you want to do your best to guide your patient and offer every resource necessary for a healthy and happy life. The good news is that your patient trusted you enough to disclose this information to you.
Sadly, this isn’t always the case. About 23% of transgender adults responding to the 2015 U.S. Transgender Survey put off necessary health care because they fear being mistreated or disrespected. Nearly one-third (31%) of survey respondents reported that none of their health care providers knew they were transgender.
Now that your patient feels comfortable enough with you to share this information, you must make sure you do everything in your power to continue to earn your patient’s trust.
Language matters
First, make sure that you are respectful with your terminology. Ask the patient for their name, pronouns, and gender identity. For example: “My name is Dr. Pine, and my pronouns are she/her. What are your pronouns? How do you describe your gender identity?” Each person may have terminology that is specific to their own experience, so allowing people to use their own language is the most respectful method.
People may identify as male, female, transwoman, transman, gender fluid, nonbinary, agender, neutrois, pangender, two-spirit, or other options not listed here. Physicians can be supportive by ensuring that their paperwork or electronic medical systems are sensitive to the needs of the transgender community. Having an option for the patient’s chosen name is courteous to all patients, regardless of gender identity; not everyone uses their legal name in everyday conversation.
Paperwork and electronic medical systems should ask for gender identity and sex assigned at birth, allow write-in options for issues of gender and sexual orientation, and ask for an anatomical inventory or organ inventory so that cancer screening can be conducted for the appropriate body parts.
Questions to ask
Ask patients about their gender journey: How long have they felt this way? How did they come to understand themselves and their gender? When did they start to disclose their experience with others? With pediatric patients, I ask if they have discussed this with their parents/guardians, and if they would like to have that conversation together.
Ask how you can support the patient on their journey. Are they interested in therapy, puberty blockers, hormones, or surgery? People may seek therapy for help coping with internalized transphobia, family rejection, or stigma. They may also want information or support with accessing hormones or surgery. In addition to individual therapy, there are numerous support groups for children, individuals, parents/guardians, and partners, such as PFLAG.org and Genderspectrum.org.
If you are the right kind of doctor and ready to prescribe, you can begin counseling. If not, you should know how to find local resources. The World Professional Association for Transgender Health has a directory of providers, and there are other resources listed below.
What does gender transitioning entail?
There are many components to gender transition. Some transitions may consist primarily of a social transition, with people using a different name, pronouns, and external expression, such as hairstyle and clothing. For others, there may be a medical component.
Mental health care is also an important component of gender transition for children, adolescents, adults, and family members. Mental health concerns are significantly greater in transgender and gender-nonconforming people, with higher rates of depression, anxiety, suicidal ideation, self-harm, substance abuse, eating disorders, and neurodiversity on the autism spectrum. In a study of more than 6,400 transgender people in the United States, 41% reported attempting suicide – a rate 25 times higher than that of the general population. Numerous studies show that hormonal treatment decreases depression, suicidal ideation, and anxiety, and also improves quality of life.
One common misperception, especially when working with children, is that youth transition involves a “sex change” (an outdated term) or any type of surgery. In reality, the main intervention before puberty is psychological support and social transition. The use of a chosen name at school, home, work, and with friends was shown to be associated with lower depression, suicidal ideation, and suicidal behavior. Another study demonstrated that children supported in their identities have mental health similar to that of cisgender siblings and peers.
When puberty approaches, if there is distress around natal pubertal development, then gonadotropin-releasing hormone agonists or “puberty blockers” may be used to temporarily pause the pubertal process, but only after Tanner stage II-III is reached. These medications have been safely used for decades for patients with central precocious puberty. Access to puberty-blocking medication in adolescence (when desired) has been associated with lower rates of suicidal ideation in adulthood and can truly be a lifesaving intervention.
When teens are older, they may choose to take gender-affirming hormone therapy to go through the puberty that is concordant with their affirmed gender. Adults who decide to transition medically may decide to take hormone therapy and may have gender-confirmation surgery, if desired, to align the body with their gender identity and alleviate gender dysphoria. Overall, access to medical care and hormone therapy have been shown to decrease depression, anxiety, and suicidality, and improve quality of life.
Sexuality and fertility
It is important to understand that sexuality is separate from gender identity, and that attraction and sexual activity cannot be assumed. Take a sensitive romantic and sexual history from your patient to get the information necessary to counsel patients about safe relationships and sexual practices. It is important not to make assumptions; a patient who is asexual may still be having sex, and a transgender man may be having receptive vaginal intercourse and may need information about contraception and family planning.
Also, be careful about using clinical language. Some people may want to use anatomical terms, but others may be uncomfortable or triggered by them. For instance, a transgender man may use “chest” for breasts, or “genital canal,” and “junk,” “dick,” or “front hole” for the vagina. Ask patients what terms they prefer to use.
It is also important to consider the impact that medical and surgical interventions may have on fertility, especially when discussing the topic with children and adolescents who may not have spent much time thinking about family planning. Be careful not to make assumptions about plans for parenthood, and remember that there are many paths to becoming a parent.
What does the patient need right now?
When I was a fourth-year medical student on my outpatient child psychiatry rotation, a 5-year-old child assigned male at birth was guarded and frightened of me until their mother said: “It’s okay, Dr. Elyse likes girl things too,” at which point the child became animated and happy while chatting with me about Barbie dolls. My patient had already endured teasing about gender nonconformity, starting in kindergarten; it was unclear to my patient whether I would be a safe person or a bully.
The mother was kind and affirming, but she also wanted answers. Would her child grow up to be a gay man? Or a transgender woman? Would her child be able to live freely, or would they always be quiet around others, scared of what people might say? Would her child be safe?
We can’t predict the future, but as doctors, we want to use all of our knowledge and tools to help our patients live healthy lives. In this case, it meant helping the mother know how to support her child’s identity, how to advocate for a safe school and community, where to connect with other gender-creative children, and how to tolerate ambiguity and celebrate the child she has, not the child she expected.
We know that people with higher support and higher self-esteem can have greater resilience and greater success. This family may need medical resources for puberty blockers, hormone therapy, and even surgery someday, but reassurance is what was needed in the moment. When your patient comes out to you, they are trusting you. It is your obligation and privilege as a medical professional to help them begin a journey to an authentic life.
A version of this article first appeared on Medscape.com.
A 16-year-old patient sits in front of you and says, “I think I’m transgender.”
What do you do?
Whether you are an endocrinologist, family physician, pediatrician, or emergency physician, it’s probably a situation for which medical school education did not sufficiently prepare you. What you know is that you want to do your best to guide your patient and offer every resource necessary for a healthy and happy life. The good news is that your patient trusted you enough to disclose this information to you.
Sadly, this isn’t always the case. About 23% of transgender adults responding to the 2015 U.S. Transgender Survey put off necessary health care because they fear being mistreated or disrespected. Nearly one-third (31%) of survey respondents reported that none of their health care providers knew they were transgender.
Now that your patient feels comfortable enough with you to share this information, you must make sure you do everything in your power to continue to earn your patient’s trust.
Language matters
First, make sure that you are respectful with your terminology. Ask the patient for their name, pronouns, and gender identity. For example: “My name is Dr. Pine, and my pronouns are she/her. What are your pronouns? How do you describe your gender identity?” Each person may have terminology that is specific to their own experience, so allowing people to use their own language is the most respectful method.
People may identify as male, female, transwoman, transman, gender fluid, nonbinary, agender, neutrois, pangender, two-spirit, or other options not listed here. Physicians can be supportive by ensuring that their paperwork or electronic medical systems are sensitive to the needs of the transgender community. Having an option for the patient’s chosen name is courteous to all patients, regardless of gender identity; not everyone uses their legal name in everyday conversation.
Paperwork and electronic medical systems should ask for gender identity and sex assigned at birth, allow write-in options for issues of gender and sexual orientation, and ask for an anatomical inventory or organ inventory so that cancer screening can be conducted for the appropriate body parts.
Questions to ask
Ask patients about their gender journey: How long have they felt this way? How did they come to understand themselves and their gender? When did they start to disclose their experience with others? With pediatric patients, I ask if they have discussed this with their parents/guardians, and if they would like to have that conversation together.
Ask how you can support the patient on their journey. Are they interested in therapy, puberty blockers, hormones, or surgery? People may seek therapy for help coping with internalized transphobia, family rejection, or stigma. They may also want information or support with accessing hormones or surgery. In addition to individual therapy, there are numerous support groups for children, individuals, parents/guardians, and partners, such as PFLAG.org and Genderspectrum.org.
If you are the right kind of doctor and ready to prescribe, you can begin counseling. If not, you should know how to find local resources. The World Professional Association for Transgender Health has a directory of providers, and there are other resources listed below.
What does gender transitioning entail?
There are many components to gender transition. Some transitions may consist primarily of a social transition, with people using a different name, pronouns, and external expression, such as hairstyle and clothing. For others, there may be a medical component.
Mental health care is also an important component of gender transition for children, adolescents, adults, and family members. Mental health concerns are significantly greater in transgender and gender-nonconforming people, with higher rates of depression, anxiety, suicidal ideation, self-harm, substance abuse, eating disorders, and neurodiversity on the autism spectrum. In a study of more than 6,400 transgender people in the United States, 41% reported attempting suicide – a rate 25 times higher than that of the general population. Numerous studies show that hormonal treatment decreases depression, suicidal ideation, and anxiety, and also improves quality of life.
One common misperception, especially when working with children, is that youth transition involves a “sex change” (an outdated term) or any type of surgery. In reality, the main intervention before puberty is psychological support and social transition. The use of a chosen name at school, home, work, and with friends was shown to be associated with lower depression, suicidal ideation, and suicidal behavior. Another study demonstrated that children supported in their identities have mental health similar to that of cisgender siblings and peers.
When puberty approaches, if there is distress around natal pubertal development, then gonadotropin-releasing hormone agonists or “puberty blockers” may be used to temporarily pause the pubertal process, but only after Tanner stage II-III is reached. These medications have been safely used for decades for patients with central precocious puberty. Access to puberty-blocking medication in adolescence (when desired) has been associated with lower rates of suicidal ideation in adulthood and can truly be a lifesaving intervention.
When teens are older, they may choose to take gender-affirming hormone therapy to go through the puberty that is concordant with their affirmed gender. Adults who decide to transition medically may decide to take hormone therapy and may have gender-confirmation surgery, if desired, to align the body with their gender identity and alleviate gender dysphoria. Overall, access to medical care and hormone therapy have been shown to decrease depression, anxiety, and suicidality, and improve quality of life.
Sexuality and fertility
It is important to understand that sexuality is separate from gender identity, and that attraction and sexual activity cannot be assumed. Take a sensitive romantic and sexual history from your patient to get the information necessary to counsel patients about safe relationships and sexual practices. It is important not to make assumptions; a patient who is asexual may still be having sex, and a transgender man may be having receptive vaginal intercourse and may need information about contraception and family planning.
Also, be careful about using clinical language. Some people may want to use anatomical terms, but others may be uncomfortable or triggered by them. For instance, a transgender man may use “chest” for breasts, or “genital canal,” and “junk,” “dick,” or “front hole” for the vagina. Ask patients what terms they prefer to use.
It is also important to consider the impact that medical and surgical interventions may have on fertility, especially when discussing the topic with children and adolescents who may not have spent much time thinking about family planning. Be careful not to make assumptions about plans for parenthood, and remember that there are many paths to becoming a parent.
What does the patient need right now?
When I was a fourth-year medical student on my outpatient child psychiatry rotation, a 5-year-old child assigned male at birth was guarded and frightened of me until their mother said: “It’s okay, Dr. Elyse likes girl things too,” at which point the child became animated and happy while chatting with me about Barbie dolls. My patient had already endured teasing about gender nonconformity, starting in kindergarten; it was unclear to my patient whether I would be a safe person or a bully.
The mother was kind and affirming, but she also wanted answers. Would her child grow up to be a gay man? Or a transgender woman? Would her child be able to live freely, or would they always be quiet around others, scared of what people might say? Would her child be safe?
We can’t predict the future, but as doctors, we want to use all of our knowledge and tools to help our patients live healthy lives. In this case, it meant helping the mother know how to support her child’s identity, how to advocate for a safe school and community, where to connect with other gender-creative children, and how to tolerate ambiguity and celebrate the child she has, not the child she expected.
We know that people with higher support and higher self-esteem can have greater resilience and greater success. This family may need medical resources for puberty blockers, hormone therapy, and even surgery someday, but reassurance is what was needed in the moment. When your patient comes out to you, they are trusting you. It is your obligation and privilege as a medical professional to help them begin a journey to an authentic life.
A version of this article first appeared on Medscape.com.
A 16-year-old patient sits in front of you and says, “I think I’m transgender.”
What do you do?
Whether you are an endocrinologist, family physician, pediatrician, or emergency physician, it’s probably a situation for which medical school education did not sufficiently prepare you. What you know is that you want to do your best to guide your patient and offer every resource necessary for a healthy and happy life. The good news is that your patient trusted you enough to disclose this information to you.
Sadly, this isn’t always the case. About 23% of transgender adults responding to the 2015 U.S. Transgender Survey put off necessary health care because they fear being mistreated or disrespected. Nearly one-third (31%) of survey respondents reported that none of their health care providers knew they were transgender.
Now that your patient feels comfortable enough with you to share this information, you must make sure you do everything in your power to continue to earn your patient’s trust.
Language matters
First, make sure that you are respectful with your terminology. Ask the patient for their name, pronouns, and gender identity. For example: “My name is Dr. Pine, and my pronouns are she/her. What are your pronouns? How do you describe your gender identity?” Each person may have terminology that is specific to their own experience, so allowing people to use their own language is the most respectful method.
People may identify as male, female, transwoman, transman, gender fluid, nonbinary, agender, neutrois, pangender, two-spirit, or other options not listed here. Physicians can be supportive by ensuring that their paperwork or electronic medical systems are sensitive to the needs of the transgender community. Having an option for the patient’s chosen name is courteous to all patients, regardless of gender identity; not everyone uses their legal name in everyday conversation.
Paperwork and electronic medical systems should ask for gender identity and sex assigned at birth, allow write-in options for issues of gender and sexual orientation, and ask for an anatomical inventory or organ inventory so that cancer screening can be conducted for the appropriate body parts.
Questions to ask
Ask patients about their gender journey: How long have they felt this way? How did they come to understand themselves and their gender? When did they start to disclose their experience with others? With pediatric patients, I ask if they have discussed this with their parents/guardians, and if they would like to have that conversation together.
Ask how you can support the patient on their journey. Are they interested in therapy, puberty blockers, hormones, or surgery? People may seek therapy for help coping with internalized transphobia, family rejection, or stigma. They may also want information or support with accessing hormones or surgery. In addition to individual therapy, there are numerous support groups for children, individuals, parents/guardians, and partners, such as PFLAG.org and Genderspectrum.org.
If you are the right kind of doctor and ready to prescribe, you can begin counseling. If not, you should know how to find local resources. The World Professional Association for Transgender Health has a directory of providers, and there are other resources listed below.
What does gender transitioning entail?
There are many components to gender transition. Some transitions may consist primarily of a social transition, with people using a different name, pronouns, and external expression, such as hairstyle and clothing. For others, there may be a medical component.
Mental health care is also an important component of gender transition for children, adolescents, adults, and family members. Mental health concerns are significantly greater in transgender and gender-nonconforming people, with higher rates of depression, anxiety, suicidal ideation, self-harm, substance abuse, eating disorders, and neurodiversity on the autism spectrum. In a study of more than 6,400 transgender people in the United States, 41% reported attempting suicide – a rate 25 times higher than that of the general population. Numerous studies show that hormonal treatment decreases depression, suicidal ideation, and anxiety, and also improves quality of life.
One common misperception, especially when working with children, is that youth transition involves a “sex change” (an outdated term) or any type of surgery. In reality, the main intervention before puberty is psychological support and social transition. The use of a chosen name at school, home, work, and with friends was shown to be associated with lower depression, suicidal ideation, and suicidal behavior. Another study demonstrated that children supported in their identities have mental health similar to that of cisgender siblings and peers.
When puberty approaches, if there is distress around natal pubertal development, then gonadotropin-releasing hormone agonists or “puberty blockers” may be used to temporarily pause the pubertal process, but only after Tanner stage II-III is reached. These medications have been safely used for decades for patients with central precocious puberty. Access to puberty-blocking medication in adolescence (when desired) has been associated with lower rates of suicidal ideation in adulthood and can truly be a lifesaving intervention.
When teens are older, they may choose to take gender-affirming hormone therapy to go through the puberty that is concordant with their affirmed gender. Adults who decide to transition medically may decide to take hormone therapy and may have gender-confirmation surgery, if desired, to align the body with their gender identity and alleviate gender dysphoria. Overall, access to medical care and hormone therapy have been shown to decrease depression, anxiety, and suicidality, and improve quality of life.
Sexuality and fertility
It is important to understand that sexuality is separate from gender identity, and that attraction and sexual activity cannot be assumed. Take a sensitive romantic and sexual history from your patient to get the information necessary to counsel patients about safe relationships and sexual practices. It is important not to make assumptions; a patient who is asexual may still be having sex, and a transgender man may be having receptive vaginal intercourse and may need information about contraception and family planning.
Also, be careful about using clinical language. Some people may want to use anatomical terms, but others may be uncomfortable or triggered by them. For instance, a transgender man may use “chest” for breasts, or “genital canal,” and “junk,” “dick,” or “front hole” for the vagina. Ask patients what terms they prefer to use.
It is also important to consider the impact that medical and surgical interventions may have on fertility, especially when discussing the topic with children and adolescents who may not have spent much time thinking about family planning. Be careful not to make assumptions about plans for parenthood, and remember that there are many paths to becoming a parent.
What does the patient need right now?
When I was a fourth-year medical student on my outpatient child psychiatry rotation, a 5-year-old child assigned male at birth was guarded and frightened of me until their mother said: “It’s okay, Dr. Elyse likes girl things too,” at which point the child became animated and happy while chatting with me about Barbie dolls. My patient had already endured teasing about gender nonconformity, starting in kindergarten; it was unclear to my patient whether I would be a safe person or a bully.
The mother was kind and affirming, but she also wanted answers. Would her child grow up to be a gay man? Or a transgender woman? Would her child be able to live freely, or would they always be quiet around others, scared of what people might say? Would her child be safe?
We can’t predict the future, but as doctors, we want to use all of our knowledge and tools to help our patients live healthy lives. In this case, it meant helping the mother know how to support her child’s identity, how to advocate for a safe school and community, where to connect with other gender-creative children, and how to tolerate ambiguity and celebrate the child she has, not the child she expected.
We know that people with higher support and higher self-esteem can have greater resilience and greater success. This family may need medical resources for puberty blockers, hormone therapy, and even surgery someday, but reassurance is what was needed in the moment. When your patient comes out to you, they are trusting you. It is your obligation and privilege as a medical professional to help them begin a journey to an authentic life.
A version of this article first appeared on Medscape.com.
Heavier girls hit hormonal puberty earlier, but develop breasts later
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
Girls with more body fat experienced earlier menarche and hormone changes, but later full breast development, compared with those with normal weight, according to longitudinal data from 90 girls aged 8-15 years.
A link between obesity and early puberty has been observed among U.S. girls for decades, but more recent studies suggest that “girls with greater childhood adiposity have earlier thelarche and progress through puberty at a faster rate than normal weight girls,” wrote Madison T. Ortega, MD, of the National Institute of Environmental Health Sciences, Durham, N.C., and colleagues. However, studies involving hormone levels have yielded mixed results, they said.
In a study published in the Journal of Clinical Endocrinology & Metabolism , the researchers followed 36 girls with overweight or obesity and 54 girls with normal weight for 4 years; normal weight was defined as body mass index in the 5th to 85th percentile, overweight was defined as BMI in the 85th to 95th percentile, and obese was defined as greater than 95th percentile. Overweight and obese were combined into one category for comparison with normal weight girls.
Participants had an average of 2.8 study visits during this period and provided additional information by phone and online. Visits included measurement of total body fat using dual-energy x-ray absorptiometry (DXA), Tanner staging, breast ultrasound for morphological staging (BMORPH; A-E), pelvic ultrasound, hormone tests, and menarchal status assessment.
Overall, girls with overweight/obesity (OW/OB) had significantly more advanced breast development at baseline than did those with normal weight (NW), but these girls progressed through BMORPH stage D later than did NW girls. Early-stage breast development was not affected by total body fat. However, “an increase of 5 percentage points in mean total body fat, for example, was associated with a 26% decrease in the transition rate out of stage D,” the researchers noted.
Hormone levels were similar at baseline for follicle-stimulating hormone, inhibin B, estrone (E1), total and free testosterone, and androstenedione. However, these levels increased more quickly after 1 year for girls with OW/OB, while they plateaued in girls with NW and dropped among girls with lower total body fat. Total body fat had no apparent effect on other reproductive hormones including luteinizing hormone, modified vaginal maturation index, and estradiol 2.
The average age of menarche was 12.4 years across all participants, but girls with higher total body fat at baseline were more likely to reach menarche at a younger age. “For every 1-unit increase in visit one total body fat, the chance of achieving menarche at any given time point was 3% higher,” the researchers said. No interaction appeared between race and total body fat with regard to menarche.
Several surprising findings
The study is important because “there have been no longitudinal studies in U.S. girls to examine how total body fat affects serum reproductive hormones or the development of the breast and ovaries using ultrasound imaging,” corresponding author Natalie Shaw, MD, of the National Institute of Environmental Health Sciences, said in an interview.
Dr. Shaw said she was surprised by several of the study findings. “Others have reported increased male-like hormones (androgens) in overweight/obese girls in cross-sectional studies; however, we were surprised to find that FSH and inhibin B were also elevated in girls with excess body fat,” she said. “We also found, unexpectedly, that even though the breast bud appears earlier in overweight/obese girls (thelarche), which signals the onset of puberty, the breast matured more slowly during the course of puberty in overweight/obese girls compared with normal weight girls,” she noted.
“The main take-home message is that puberty looks different in girls with excess body fat; they develop breast tissue earlier, yet take longer to achieve a fully mature breast, and they undergo menarche earlier,” Dr. Shaw said. Clinicians should be aware of the hormonal differences based on body fat, Dr. Shaw emphasized. “Girls with greater body fat had higher levels of FSH (a pituitary hormone), inhibin B (an ovarian hormone), and male-like reproductive hormones (e.g., testosterone) that are made by the adrenal glands and the ovaries in the late stages of puberty,” she said.
Potential implications for adulthood
“The findings in this study contribute to better understanding how total body fat impacts hormonal findings of puberty,” M. Susan Jay, MD, of the Medical College of Wisconsin and Children’s Hospital of Wisconsin, Milwaukee, said in an interview. “Prior studies have linked weight gain as a factor that contributes to pubertal development, but this study is attempting to longitudinally investigate how body weight may affect clinical and biochemical pubertal markers in girls,” she noted.
“The take-home message is that this study and other earlier studies have illustrated that puberty is not a fixed pattern in all individual girls,” Dr. Jay emphasized. “Rather, there are environmental factors which can impact pubertal course,” she said. “In effect, there are pathways through puberty in individual adolescents that require greater ongoing studies to further identify the arc of puberty and the impact of how the length in various stages may affect exposure to estrogen and other neurohormonal factors,” she explained. These factors impact not only adolescence but also future health in adulthood, she said.
“Ongoing prospective studies are needed to identify how factors such as body weight can affect adolescent pubertal development and the possible impact long after adolescence for health issues such as breast cancer,” Dr. Jay added.
The study findings were limited by several factors including the available data from only two completed study visits for most participants, as well as the racial differences among body weight groups and lack of standardized timing for blood draws, the researchers noted.
The study was supported in part by the National Institute of Environmental Health Sciences, and corresponding author Dr. Shaw disclosed support as a Lasker Clinical Research Scholar. The other researchers, as well as Dr. Jay, had no disclosures.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM
Tips offered for treating co-occurring ADHD and SUDs
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
When Frances R. Levin, MD, began her clinical psychiatry career in the mid-1990s, she spent a lot of time educating colleagues about the validity of an ADHD diagnosis in adults.
“That’s no longer an issue,” Dr. Levin, the Kennedy-Leavy Professor of Psychiatry at Columbia University, New York, said during an annual psychopharmacology update held by the Nevada Psychiatric Association. “But at the time, we often thought, ‘ADHD is something that’s specific to people who are stimulant users.’ In fact, what we found over the years was that these rates are elevated in a range of substance use populations.”
According to National Comorbidity Survey, a nontreatment sample of more than 3,000 adults, individuals who have SUD have two to three times the risk of having ADHD, while individuals who have ADHD have about three times the rate of having an SUD, compared with those who don’t (Am J Psychiatry. 2006;163[4]:716-23). “When you move to treatment samples, the rates also remain quite high,” said Dr. Levin, who is also chief of the division of substance use disorders at the medical center.
“In the general population, the rates of ADHD are 2%-4%. When we look at people who are coming in specifically for treatment of their SUD, the rates are substantially higher, ranging from 10% to 24%.”
According to a 2014 review of medical literature, potential reasons for the association between ADHD and SUD vary and include underlying biologic deficits, such as parental SUDs and genetics; conduct disorder symptoms, such as defiance, rule breaking, and delinquency; poor performance in school, such as low grades, grade retention, or drop-out; and social difficulties, such as rejection from conventional groups or few quality friendships (Annu Rev Clin Psychol. 2014;10:607-39). Other potential pathways include neurocognitive deficits, stress-negative affect models, impulsive anger, and other underlying traits.
One key reason to treat ADHD in patients with SUDs is that they tend to develop the SUD earlier when the ADHD is present, Dr. Levin said. They’re also less likely to be retained in treatment and have a reduced likelihood of going into remission if dependence develops. “Even when they do achieve remission, it seems to take longer for people to reach remission,” she said. “They have more treatment exposure yet do less well in treatment. This can make it more challenging to treat this population.”
One common assumption from clinicians regarding patients with ADHD and a concomitant SUD is that standard treatments for ADHD do not work in active substance users. Another is that, even if treatments work for ADHD, they do not affect the substance use disorder. “Understandably, there is also concern that active substance abusers will misuse and divert their medications,” she said. “Finally, there are often additional psychiatric comorbidities that may make it harder to effectively treat individuals with ADHD and SUD.”
Since 2002, 15 double-blind outpatient studies using stimulants/atomoxetine to treat substance abusers with ADHD have appeared in the medical literature, Dr. Levin said. Only three have included adolescents. “That’s surprising, because up to 40% of kids who come in for treatment, often for cannabis use disorder, will have ADHD, yet there is very little guidance from empirical studies as to how to best treat them,” she said. “There have been several studies looking at atomoxetine to treat substance abusers with ADHD, but results have been mixed. In the cannabis use populations, atomoxetine has not been shown to be effective in treating the substance use disorder, and results are mixed regarding superiority in reducing ADHD symptoms. There is one study showing that ADHD is more likely to be improved in adults with alcohol use disorders with mixed results regarding the alcohol use.”
Overall, most of the outpatient and inpatient studies conducted in this population have demonstrated some signal in terms of reducing ADHD, she said, while a minority of the outpatient studies suggest some benefit in terms of substance use. “What’s interesting is that when you see a response in terms of the ADHD, you often see an improvement in the substance use as well,” Dr. Levin said. “This potentially suggests that patients may be self-medicating their ADHD symptoms or that if the ADHD responds to treatment, then the patient may benefit from the psychosocial interventions that targets the SUD.”
A separate meta-analysis involving more than 1,000 patients found mixed results from pharmacologic interventions and concluded that, while they modestly improved ADHD symptoms, no beneficial effect was seen on drug abstinence or on treatment discontinuation (J Psychopharmacol. 2015 Jan;29[1]:15-23). “I would argue that you don’t need to be as nihilistic about this as the meta-analysis might suggest, because the devil’s in the details,” said Dr. Levin, whose own research was included in the work.
“First of all, many of the studies had high drop-out rates. The outcome measures were variable, and some of the studies used formulations with poor bioavailability. Also, trials that evaluated atomoxetine or stimulants were combined, which may be problematic given the different mechanisms of action. Further, the meta-analysis did not include two recent placebo-controlled trials in adults with stimulant-use disorders that both found that higher dosing of a long-acting stimulant resulted in greater improvements in ADHD symptoms and stimulant use” (Addict. 2014;109[3]:440-9 and JAMA Psychiatry. 2015;72[6]:593-602).
Dr. Levin went on to note that there are few empirical data to guide treatment for those who have multiple psychiatric disorders, let alone treatment for ADHD and SUDs without additional psychiatric disorders. The challenge is what to treat first and/or how to treat the concomitant conditions safely.
“Generally, if possible, treat what is most clinically impairing first,” she said. “Overall, both stimulants and atomoxetine may work for ADHD even in the presence of additional depression, anxiety disorders, and substance use disorders.”
She cautioned against treating a patient with ADHD medication if there is a preexisting psychosis or bipolar illness. “If you start a stimulant or atomoxetine and psychosis or mania occurs, you clearly want to stop the medication and reassess,” she said. Researchers found that the risk of precipitating mania with a stimulant is uncommon if you alleviate symptoms first with a mood stabilizer. “This is a situation where you probably want to treat the bipolar illness first, but it does not preclude the treatment of ADHD once the mood stabilization has occurred,” she said.
In patients with ADHD and anxiety, she often treats the ADHD first, “because oftentimes the anxiety is driven by the procrastination and the inability to get things done,” she explained. “It’s important to determine whether the anxiety is an independent disorder rather than symptoms of ADHD. Inner restlessness can be described as anxiety.”
When there are concerns that preclude the use of a controlled medication, there are medications, in addition to atomoxetine, that might be considered. While bupropion is not Food and Drug Administration approved for ADHD, it might be useful in comorbid mood disorders for nicotine dependence. Other off-label medications that may help include guanfacine, modafinil, and tricyclic antidepressants.
“To date, robust dosing of long-acting amphetamine or methylphenidate formulations have been shown to be effective for patients with stimulant-use disorder, but as mentioned earlier, the data only come from two studies,” she said.
In order to determine whether stimulant treatment is yielding a benefit in a patient with co-occurring ADHD and SUD, she recommends carrying out a structured assessment of ADHD symptoms. Monitoring for functional improvement is also key.
“If there is no improvement in social, occupational, or academic settings and the patient is still actively using drugs, then there is no reason to keep prescribing,” she said. Close monitoring for cardiovascular or other psychiatric symptoms are key as well. Further, for those individuals with both ADHD and a substance-use disorder, it is critical that both are targeted for treatment.
Dr. Levin reported that she has received research, training, or salary support from the National Institute on Drug Abuse, New York state, and the Substance Abuse and Mental Health Services Administration. She has also received or currently receives industry support from Indivior and U.S. World Meds and for medication and from Major League Baseball. In addition, Dr. Levin has been an unpaid scientific advisory board member for Alkermes, Indivior, and Novartis.
FROM NPA 2021
Emerging research shows link between suicidality, ‘high-potency’ cannabis products
Number of suicides positive for marijuana on rise soared among Colorado youth
In the days since recreational sales of marijuana became legal in Colorado in January 2014, concerning trends have emerged among the state’s young cannabis users.
According to a report from the Rocky Mountain High Intensity Drug Trafficking Area, between 2014 and 2017, the number of suicides positive for marijuana increased 250% among those aged 10-19 years (from 4 to 14) and 22% among those aged 20 and older (from 118 to 144). “Other states are seeing something similar, and there is an emerging research showing a relationship between suicidality and the use of marijuana, especially high-potency products that are available in legalized markets,” Paula D. Riggs, MD, reported during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During that same 3-year time span, the proportion of Colorado youth aged 12 years and older who used marijuana in the past month jumped by 45%, which is more than 85% above the national average. “Similarly, among college-age students, we’ve seen an 18% increase in past-month marijuana use, which is 60% above the national average,” said Dr. Riggs, professor and vice chair of psychiatry at the University of Colorado at Denver, Aurora.
Among adolescents, state health officials have observed a 5% increase in the proportion of those who used marijuana in the past month, which is more than 54% above the national average. “But a concerning trend is that we’re seeing an increase in the use of concentrates such as dabs and waxes,” she said. “That’s worrisome in terms of exposure to high-potency products.”
In other findings, 48% of young marijuana users reported going to work high (40% at least once per week), and there has been a 170% increase in youth ED urgent care visits for marijuana-related illnesses such as cannabinoid hyperemesis syndrome or first-episode psychosis. State health officials have also observed a 148% increase in marijuana-related hospitalizations.
According to Dr. Riggs, who also directs the University of Colorado’s division of addiction science, prevention, and treatment, the average marijuana joint in the 1960s contained about 3% tetrahydrocannabinol (THC), a level that crept up to the 4%-6% range in 2002. In today’s postlegalization era, the average joint now contains 13%-23% THC. “What’s concerning is that the concentrates – the dabs, waxes, shatter, and butane hash oils – contain upward of 70%-95% THC,” Dr. Riggs said. “Those are highly potent products that represent about 25% of the market share now. That’s a very big concern because the higher the potency the cannabis product used, the greater the abuse liability and addictive potential.”
The use of high-potency products also doubles the risk of developing generalized anxiety disorder, triples the risk of tobacco dependence, doubles the risk of other illicit substance disorders, and it at least quadruples the risk of developing first-episode psychosis in young people. “So, when you’re taking a cannabis use history, it’s important to ask patients about the potency of the products being used,” she said.
In the 2019 Monitoring the Future survey, 12% of U.S. 8th graders self-reported marijuana use in the past year and 7% in the past month, compared with 29% and 18% of 10th graders, respectively. Self-reported use by 12th graders was even more elevated (36% in the past year and 29% in the past month). “The concern is, this survey doesn’t really capture what’s happening with marijuana concentrates,” Dr. Riggs said.
A survey of Colorado youth conducted by the state’s Department of Public Health and Environment found that the percentage of students who reported using concentrated forms of marijuana has risen steadily in recent years and now stands at roughly 34%. “The use of edibles has also crept up,” said Dr. Riggs, who noted that marijuana dispensaries in Colorado outnumber Starbucks locations and McDonald’s restaurants. “You might not think that’s particularly concerning, except that the use of edibles is even more associated with onset of psychosis than other forms. This is probably because when you eat a marijuana product, you can’t control the exposure or the dose that you’re ingesting. We need to be concerned about these trends.”
European studies report that 30%-50% of new cases of first-onset psychosis are attributed to high-potency cannabis. “There is a dose-response relationship between cannabis and psychosis,” Dr. Riggs said. “That is, the frequency and duration of cannabis use, or the use of high-potency products, and the age of onset, are strongly associated with the risk of first-episode psychosis.
Researchers have known for some time that alterations in the endocannabinoid system are associated with psychosis independent of cannabis exposure. “Dysregulation of that endocannabinoid system occurs in patients at all stages of the psychosis continuum,” she continued. “It also means that the endocannabinoid system is a potential therapeutic target for psychosis.”
According to Dr. Riggs, THC exposure acutely increases dopamine in the ventral striatum and it can produce transient psychotomimetic effects in clinical and nonclinical populations. Genetic differences in the dopaminergic system can also interact with cannabis use to increase the risk of psychosis.
“For example, the COMT (catechol-O-methyltransferase) breaks down catecholamines such as dopamine in the prefrontal cortex,” she explained. “If you have a COMT gene polymorphism, that increases your risk of developing psychosis due to increased levels of dopamine signaling.”
She emphasized the importance of clinicians to understand that the age of cannabis use onset, the duration, frequency, and THC potency is related to the psychosis risk and worse prognosis. The earlier the initiation of marijuana use, the greater potential for first-episode psychosis. “Those who continue using cannabis after a first-episode psychosis have greater severity of psychotic illness and more treatment resistance, and they’re less likely to engage or be compliant with treatment recommendations,” Dr. Riggs said. “So, Because if they resume cannabis use, this can turn into a more chronic psychotic disorder.”
She added that, while insufficient evidence exists to determine whether cannabis plays a causal role in the development of schizophrenia or not, mounting evidence suggests that cannabis use may precipitate earlier onset of schizophrenia in those with other risk factors for the disorder. “There is considerable evidence that cannabis use increases the risk of psychosis in a dose-related manner, especially with an onset before age 16,” Dr. Riggs said. “However, this does not mean that cannabis is safe for young adults. Cannabis-induced psychotic symptoms often develop during young adulthood and may become chronic.”
Dr. Riggs disclosed that she had received grant funding from the National Institute on Drug Abuse. She is also executive director for Encompass, which provides integrated treatment for adolescents and young adults.
Number of suicides positive for marijuana on rise soared among Colorado youth
Number of suicides positive for marijuana on rise soared among Colorado youth
In the days since recreational sales of marijuana became legal in Colorado in January 2014, concerning trends have emerged among the state’s young cannabis users.
According to a report from the Rocky Mountain High Intensity Drug Trafficking Area, between 2014 and 2017, the number of suicides positive for marijuana increased 250% among those aged 10-19 years (from 4 to 14) and 22% among those aged 20 and older (from 118 to 144). “Other states are seeing something similar, and there is an emerging research showing a relationship between suicidality and the use of marijuana, especially high-potency products that are available in legalized markets,” Paula D. Riggs, MD, reported during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During that same 3-year time span, the proportion of Colorado youth aged 12 years and older who used marijuana in the past month jumped by 45%, which is more than 85% above the national average. “Similarly, among college-age students, we’ve seen an 18% increase in past-month marijuana use, which is 60% above the national average,” said Dr. Riggs, professor and vice chair of psychiatry at the University of Colorado at Denver, Aurora.
Among adolescents, state health officials have observed a 5% increase in the proportion of those who used marijuana in the past month, which is more than 54% above the national average. “But a concerning trend is that we’re seeing an increase in the use of concentrates such as dabs and waxes,” she said. “That’s worrisome in terms of exposure to high-potency products.”
In other findings, 48% of young marijuana users reported going to work high (40% at least once per week), and there has been a 170% increase in youth ED urgent care visits for marijuana-related illnesses such as cannabinoid hyperemesis syndrome or first-episode psychosis. State health officials have also observed a 148% increase in marijuana-related hospitalizations.
According to Dr. Riggs, who also directs the University of Colorado’s division of addiction science, prevention, and treatment, the average marijuana joint in the 1960s contained about 3% tetrahydrocannabinol (THC), a level that crept up to the 4%-6% range in 2002. In today’s postlegalization era, the average joint now contains 13%-23% THC. “What’s concerning is that the concentrates – the dabs, waxes, shatter, and butane hash oils – contain upward of 70%-95% THC,” Dr. Riggs said. “Those are highly potent products that represent about 25% of the market share now. That’s a very big concern because the higher the potency the cannabis product used, the greater the abuse liability and addictive potential.”
The use of high-potency products also doubles the risk of developing generalized anxiety disorder, triples the risk of tobacco dependence, doubles the risk of other illicit substance disorders, and it at least quadruples the risk of developing first-episode psychosis in young people. “So, when you’re taking a cannabis use history, it’s important to ask patients about the potency of the products being used,” she said.
In the 2019 Monitoring the Future survey, 12% of U.S. 8th graders self-reported marijuana use in the past year and 7% in the past month, compared with 29% and 18% of 10th graders, respectively. Self-reported use by 12th graders was even more elevated (36% in the past year and 29% in the past month). “The concern is, this survey doesn’t really capture what’s happening with marijuana concentrates,” Dr. Riggs said.
A survey of Colorado youth conducted by the state’s Department of Public Health and Environment found that the percentage of students who reported using concentrated forms of marijuana has risen steadily in recent years and now stands at roughly 34%. “The use of edibles has also crept up,” said Dr. Riggs, who noted that marijuana dispensaries in Colorado outnumber Starbucks locations and McDonald’s restaurants. “You might not think that’s particularly concerning, except that the use of edibles is even more associated with onset of psychosis than other forms. This is probably because when you eat a marijuana product, you can’t control the exposure or the dose that you’re ingesting. We need to be concerned about these trends.”
European studies report that 30%-50% of new cases of first-onset psychosis are attributed to high-potency cannabis. “There is a dose-response relationship between cannabis and psychosis,” Dr. Riggs said. “That is, the frequency and duration of cannabis use, or the use of high-potency products, and the age of onset, are strongly associated with the risk of first-episode psychosis.
Researchers have known for some time that alterations in the endocannabinoid system are associated with psychosis independent of cannabis exposure. “Dysregulation of that endocannabinoid system occurs in patients at all stages of the psychosis continuum,” she continued. “It also means that the endocannabinoid system is a potential therapeutic target for psychosis.”
According to Dr. Riggs, THC exposure acutely increases dopamine in the ventral striatum and it can produce transient psychotomimetic effects in clinical and nonclinical populations. Genetic differences in the dopaminergic system can also interact with cannabis use to increase the risk of psychosis.
“For example, the COMT (catechol-O-methyltransferase) breaks down catecholamines such as dopamine in the prefrontal cortex,” she explained. “If you have a COMT gene polymorphism, that increases your risk of developing psychosis due to increased levels of dopamine signaling.”
She emphasized the importance of clinicians to understand that the age of cannabis use onset, the duration, frequency, and THC potency is related to the psychosis risk and worse prognosis. The earlier the initiation of marijuana use, the greater potential for first-episode psychosis. “Those who continue using cannabis after a first-episode psychosis have greater severity of psychotic illness and more treatment resistance, and they’re less likely to engage or be compliant with treatment recommendations,” Dr. Riggs said. “So, Because if they resume cannabis use, this can turn into a more chronic psychotic disorder.”
She added that, while insufficient evidence exists to determine whether cannabis plays a causal role in the development of schizophrenia or not, mounting evidence suggests that cannabis use may precipitate earlier onset of schizophrenia in those with other risk factors for the disorder. “There is considerable evidence that cannabis use increases the risk of psychosis in a dose-related manner, especially with an onset before age 16,” Dr. Riggs said. “However, this does not mean that cannabis is safe for young adults. Cannabis-induced psychotic symptoms often develop during young adulthood and may become chronic.”
Dr. Riggs disclosed that she had received grant funding from the National Institute on Drug Abuse. She is also executive director for Encompass, which provides integrated treatment for adolescents and young adults.
In the days since recreational sales of marijuana became legal in Colorado in January 2014, concerning trends have emerged among the state’s young cannabis users.
According to a report from the Rocky Mountain High Intensity Drug Trafficking Area, between 2014 and 2017, the number of suicides positive for marijuana increased 250% among those aged 10-19 years (from 4 to 14) and 22% among those aged 20 and older (from 118 to 144). “Other states are seeing something similar, and there is an emerging research showing a relationship between suicidality and the use of marijuana, especially high-potency products that are available in legalized markets,” Paula D. Riggs, MD, reported during an annual psychopharmacology update held by the Nevada Psychiatric Association.
During that same 3-year time span, the proportion of Colorado youth aged 12 years and older who used marijuana in the past month jumped by 45%, which is more than 85% above the national average. “Similarly, among college-age students, we’ve seen an 18% increase in past-month marijuana use, which is 60% above the national average,” said Dr. Riggs, professor and vice chair of psychiatry at the University of Colorado at Denver, Aurora.
Among adolescents, state health officials have observed a 5% increase in the proportion of those who used marijuana in the past month, which is more than 54% above the national average. “But a concerning trend is that we’re seeing an increase in the use of concentrates such as dabs and waxes,” she said. “That’s worrisome in terms of exposure to high-potency products.”
In other findings, 48% of young marijuana users reported going to work high (40% at least once per week), and there has been a 170% increase in youth ED urgent care visits for marijuana-related illnesses such as cannabinoid hyperemesis syndrome or first-episode psychosis. State health officials have also observed a 148% increase in marijuana-related hospitalizations.
According to Dr. Riggs, who also directs the University of Colorado’s division of addiction science, prevention, and treatment, the average marijuana joint in the 1960s contained about 3% tetrahydrocannabinol (THC), a level that crept up to the 4%-6% range in 2002. In today’s postlegalization era, the average joint now contains 13%-23% THC. “What’s concerning is that the concentrates – the dabs, waxes, shatter, and butane hash oils – contain upward of 70%-95% THC,” Dr. Riggs said. “Those are highly potent products that represent about 25% of the market share now. That’s a very big concern because the higher the potency the cannabis product used, the greater the abuse liability and addictive potential.”
The use of high-potency products also doubles the risk of developing generalized anxiety disorder, triples the risk of tobacco dependence, doubles the risk of other illicit substance disorders, and it at least quadruples the risk of developing first-episode psychosis in young people. “So, when you’re taking a cannabis use history, it’s important to ask patients about the potency of the products being used,” she said.
In the 2019 Monitoring the Future survey, 12% of U.S. 8th graders self-reported marijuana use in the past year and 7% in the past month, compared with 29% and 18% of 10th graders, respectively. Self-reported use by 12th graders was even more elevated (36% in the past year and 29% in the past month). “The concern is, this survey doesn’t really capture what’s happening with marijuana concentrates,” Dr. Riggs said.
A survey of Colorado youth conducted by the state’s Department of Public Health and Environment found that the percentage of students who reported using concentrated forms of marijuana has risen steadily in recent years and now stands at roughly 34%. “The use of edibles has also crept up,” said Dr. Riggs, who noted that marijuana dispensaries in Colorado outnumber Starbucks locations and McDonald’s restaurants. “You might not think that’s particularly concerning, except that the use of edibles is even more associated with onset of psychosis than other forms. This is probably because when you eat a marijuana product, you can’t control the exposure or the dose that you’re ingesting. We need to be concerned about these trends.”
European studies report that 30%-50% of new cases of first-onset psychosis are attributed to high-potency cannabis. “There is a dose-response relationship between cannabis and psychosis,” Dr. Riggs said. “That is, the frequency and duration of cannabis use, or the use of high-potency products, and the age of onset, are strongly associated with the risk of first-episode psychosis.
Researchers have known for some time that alterations in the endocannabinoid system are associated with psychosis independent of cannabis exposure. “Dysregulation of that endocannabinoid system occurs in patients at all stages of the psychosis continuum,” she continued. “It also means that the endocannabinoid system is a potential therapeutic target for psychosis.”
According to Dr. Riggs, THC exposure acutely increases dopamine in the ventral striatum and it can produce transient psychotomimetic effects in clinical and nonclinical populations. Genetic differences in the dopaminergic system can also interact with cannabis use to increase the risk of psychosis.
“For example, the COMT (catechol-O-methyltransferase) breaks down catecholamines such as dopamine in the prefrontal cortex,” she explained. “If you have a COMT gene polymorphism, that increases your risk of developing psychosis due to increased levels of dopamine signaling.”
She emphasized the importance of clinicians to understand that the age of cannabis use onset, the duration, frequency, and THC potency is related to the psychosis risk and worse prognosis. The earlier the initiation of marijuana use, the greater potential for first-episode psychosis. “Those who continue using cannabis after a first-episode psychosis have greater severity of psychotic illness and more treatment resistance, and they’re less likely to engage or be compliant with treatment recommendations,” Dr. Riggs said. “So, Because if they resume cannabis use, this can turn into a more chronic psychotic disorder.”
She added that, while insufficient evidence exists to determine whether cannabis plays a causal role in the development of schizophrenia or not, mounting evidence suggests that cannabis use may precipitate earlier onset of schizophrenia in those with other risk factors for the disorder. “There is considerable evidence that cannabis use increases the risk of psychosis in a dose-related manner, especially with an onset before age 16,” Dr. Riggs said. “However, this does not mean that cannabis is safe for young adults. Cannabis-induced psychotic symptoms often develop during young adulthood and may become chronic.”
Dr. Riggs disclosed that she had received grant funding from the National Institute on Drug Abuse. She is also executive director for Encompass, which provides integrated treatment for adolescents and young adults.
FROM NPA 2021
Family medicine: Who cares for the children?
according to new research.
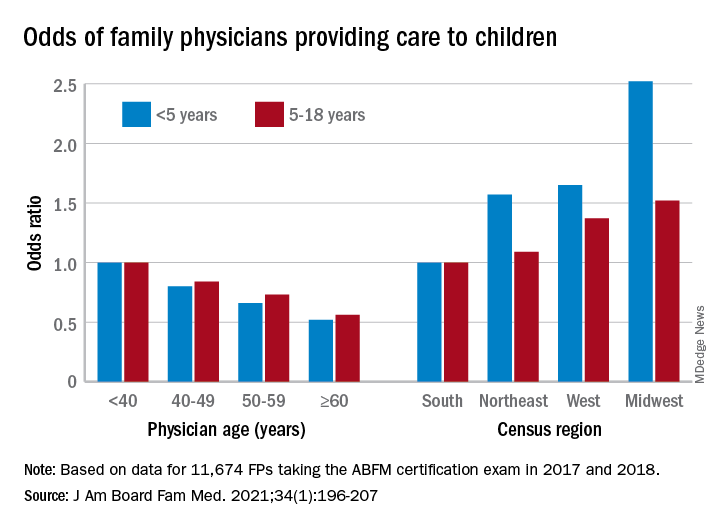
This the latest sign of a long-term decline, and it “poses a broader concern for a specialty that defines itself by its comprehensive scope of practice,” said the study investigators of the Robert Graham Center in Washington, D.C., in a written statement. “This is consistent with previous Robert Graham Center research that reported a similar steady decline from 1992 to 2002.”
Self-reported data from family physicians indicate that 84.3% cared for children aged 18 years and under in 2017, compared with 83.0% in 2018, based on a cross-sectional analysis of data gathered from 11,674 family physicians who completed the practice demographic questionnaire attached to the American Board of Family Medicine’s certification exam in 2017 and 2018.
“This current trend is unsettling, because family physicians provide the majority of pediatric care in rural and pediatrically underserved areas of the United States,” study author Anuradha Jetty, MPH, and coauthors said in the statement.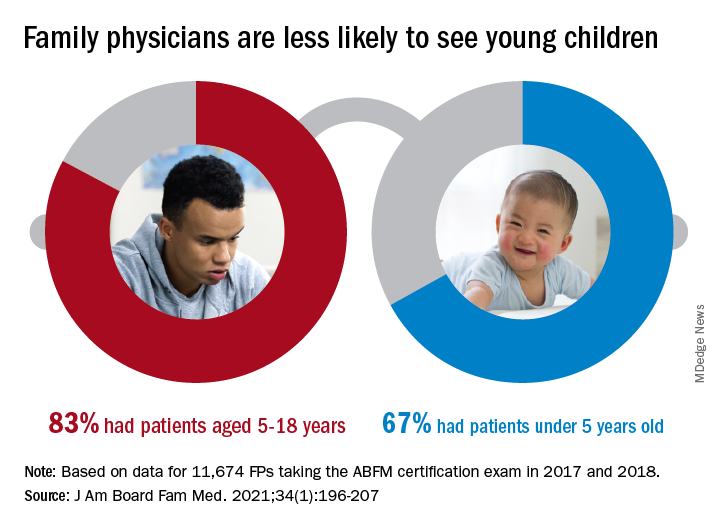
The analysis also offers a snapshot of the current state of pediatric care offered by family physicians. In 2017 and 2018, FPs were more likely to see patients aged 5-18 years than those under age 5 (83.0% vs. 67.0%), with variation by age, location, and race/ethnicity, said Ms. Jetty and colleagues, in their new paper.
FPs aged 60 years and older were much less likely to see pediatric patients, compared with those under age 40: odds ratios were 0.52 for children under 5 and 0.56 for children 5-18. Regional variation was even more pronounced: Compared with their colleagues in the Southern states, Midwestern FPs were 1.52 times as likely to treat children aged 5-18 and 2.52 times as likely to treat children under age 5, the investigators reported.
Non-Hispanic Asian and Hispanic family physicians had significantly lower odds of seeing pediatric patients, relative to non-Hispanic White family physicians, as did FPs who were international medical graduates (OR, 0.74), compared with those who trained in the United States, they said.
“Female gender was associated with seeing pediatric patients in a prior study using 2006-2009 [American Board of Family Medicine] data; however, we found no such association in 2017-2018,” Ms. Jetty and associates noted.
“Many diverse drivers likely influence the findings we observed, including organizational, personal, social, and economic factors,” they wrote, suggesting that the policies of some HMOs “may limit scope of practice for employed physicians,” while those who practice in areas of low pediatrician density might “capitalize on a market opportunity ... more than physicians in pediatrician-saturated areas with greater competition for young patients.”
The overall shortage of primary pediatric care may be a matter of debate, the investigators said, but “there is undoubtedly significant variability in the regional supply of pediatric primary care physicians and thus areas where family physicians are needed to meet current pediatric workforce demand.”
The authors reported no conflicts.
according to new research.

This the latest sign of a long-term decline, and it “poses a broader concern for a specialty that defines itself by its comprehensive scope of practice,” said the study investigators of the Robert Graham Center in Washington, D.C., in a written statement. “This is consistent with previous Robert Graham Center research that reported a similar steady decline from 1992 to 2002.”
Self-reported data from family physicians indicate that 84.3% cared for children aged 18 years and under in 2017, compared with 83.0% in 2018, based on a cross-sectional analysis of data gathered from 11,674 family physicians who completed the practice demographic questionnaire attached to the American Board of Family Medicine’s certification exam in 2017 and 2018.
“This current trend is unsettling, because family physicians provide the majority of pediatric care in rural and pediatrically underserved areas of the United States,” study author Anuradha Jetty, MPH, and coauthors said in the statement.
The analysis also offers a snapshot of the current state of pediatric care offered by family physicians. In 2017 and 2018, FPs were more likely to see patients aged 5-18 years than those under age 5 (83.0% vs. 67.0%), with variation by age, location, and race/ethnicity, said Ms. Jetty and colleagues, in their new paper.
FPs aged 60 years and older were much less likely to see pediatric patients, compared with those under age 40: odds ratios were 0.52 for children under 5 and 0.56 for children 5-18. Regional variation was even more pronounced: Compared with their colleagues in the Southern states, Midwestern FPs were 1.52 times as likely to treat children aged 5-18 and 2.52 times as likely to treat children under age 5, the investigators reported.
Non-Hispanic Asian and Hispanic family physicians had significantly lower odds of seeing pediatric patients, relative to non-Hispanic White family physicians, as did FPs who were international medical graduates (OR, 0.74), compared with those who trained in the United States, they said.
“Female gender was associated with seeing pediatric patients in a prior study using 2006-2009 [American Board of Family Medicine] data; however, we found no such association in 2017-2018,” Ms. Jetty and associates noted.
“Many diverse drivers likely influence the findings we observed, including organizational, personal, social, and economic factors,” they wrote, suggesting that the policies of some HMOs “may limit scope of practice for employed physicians,” while those who practice in areas of low pediatrician density might “capitalize on a market opportunity ... more than physicians in pediatrician-saturated areas with greater competition for young patients.”
The overall shortage of primary pediatric care may be a matter of debate, the investigators said, but “there is undoubtedly significant variability in the regional supply of pediatric primary care physicians and thus areas where family physicians are needed to meet current pediatric workforce demand.”
The authors reported no conflicts.
according to new research.

This the latest sign of a long-term decline, and it “poses a broader concern for a specialty that defines itself by its comprehensive scope of practice,” said the study investigators of the Robert Graham Center in Washington, D.C., in a written statement. “This is consistent with previous Robert Graham Center research that reported a similar steady decline from 1992 to 2002.”
Self-reported data from family physicians indicate that 84.3% cared for children aged 18 years and under in 2017, compared with 83.0% in 2018, based on a cross-sectional analysis of data gathered from 11,674 family physicians who completed the practice demographic questionnaire attached to the American Board of Family Medicine’s certification exam in 2017 and 2018.
“This current trend is unsettling, because family physicians provide the majority of pediatric care in rural and pediatrically underserved areas of the United States,” study author Anuradha Jetty, MPH, and coauthors said in the statement.
The analysis also offers a snapshot of the current state of pediatric care offered by family physicians. In 2017 and 2018, FPs were more likely to see patients aged 5-18 years than those under age 5 (83.0% vs. 67.0%), with variation by age, location, and race/ethnicity, said Ms. Jetty and colleagues, in their new paper.
FPs aged 60 years and older were much less likely to see pediatric patients, compared with those under age 40: odds ratios were 0.52 for children under 5 and 0.56 for children 5-18. Regional variation was even more pronounced: Compared with their colleagues in the Southern states, Midwestern FPs were 1.52 times as likely to treat children aged 5-18 and 2.52 times as likely to treat children under age 5, the investigators reported.
Non-Hispanic Asian and Hispanic family physicians had significantly lower odds of seeing pediatric patients, relative to non-Hispanic White family physicians, as did FPs who were international medical graduates (OR, 0.74), compared with those who trained in the United States, they said.
“Female gender was associated with seeing pediatric patients in a prior study using 2006-2009 [American Board of Family Medicine] data; however, we found no such association in 2017-2018,” Ms. Jetty and associates noted.
“Many diverse drivers likely influence the findings we observed, including organizational, personal, social, and economic factors,” they wrote, suggesting that the policies of some HMOs “may limit scope of practice for employed physicians,” while those who practice in areas of low pediatrician density might “capitalize on a market opportunity ... more than physicians in pediatrician-saturated areas with greater competition for young patients.”
The overall shortage of primary pediatric care may be a matter of debate, the investigators said, but “there is undoubtedly significant variability in the regional supply of pediatric primary care physicians and thus areas where family physicians are needed to meet current pediatric workforce demand.”
The authors reported no conflicts.
FROM THE JOURNAL OF THE AMERICAN BOARD OF FAMILY MEDICINE
The importance of family acceptance for LGBTQ youth
It is well established that LGBTQ individuals experience more health disparities compared with their cisgender, heterosexual counterparts. In general, LGBTQ adolescents and young adults have higher levels of depression, suicide attempts, and substance use than those of their heterosexual peers. However, a key protective factor is family acceptance and support. By encouraging families to modify and change behaviors that are experienced by their LGBTQ children as rejecting and to engage in supportive and affirming behaviors, providers can help families to decrease risk and promote healthy outcomes for LGBTQ youth and young adults. 
We all know that a supportive family can make a difference for any child, but this is especially true for LGBTQ youth and is critical during a pandemic when young people are confined with families and separated from peers and supportive adults outside the home. Several research studies show that family support can improve outcomes related to suicide, depression, homelessness, drug use, and HIV in LGBTQ young people. Family acceptance improves health outcomes, while rejection undermines family relationships and worsens both health and other serious outcomes such as homelessness and placement in custodial care. Pediatricians can help their patients by educating parents and caregivers with LGBTQ children about the critical role of family support – both those who see themselves as accepting and those who believe that being gay or transgender is wrong and are struggling with parenting a child who identifies as LGBTQ or who is gender diverse.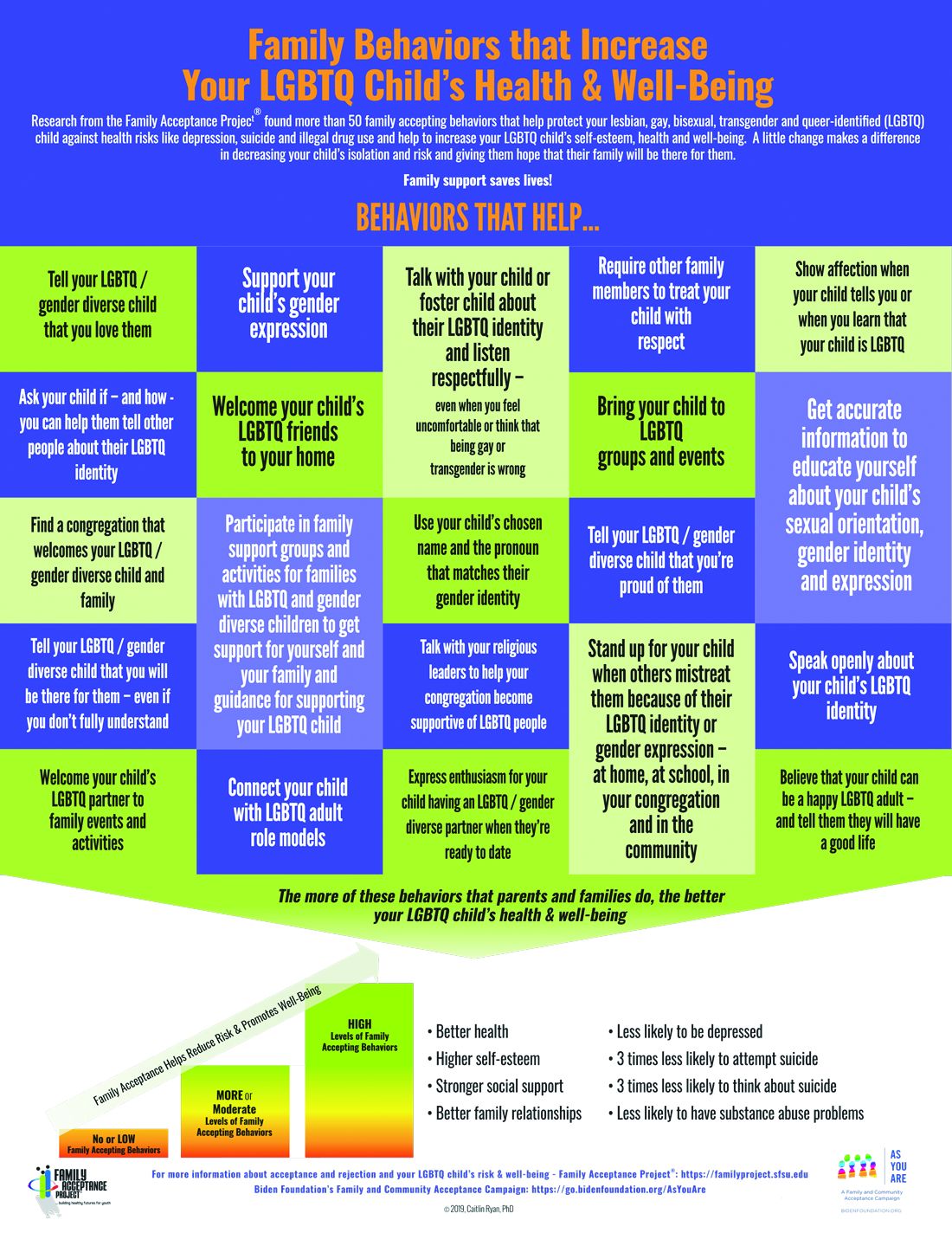
The Family Acceptance Project (FAP) at San Francisco State University conducted the first research on LGBTQ youth and families, developed the first evidence-informed family support model, and has published a range of studies and evidence-based resources that demonstrate the harm caused by family rejection, validate the importance of family acceptance, and provide guidance to increase family support. FAP’s research found that parents and caregivers that engage in rejecting behaviors are typically motivated by care and concern and by trying to protect their children from harm. They believe such behaviors will help their LGBTQ children fit in, have a good life, meet cultural and religious expectations, and be respected by others.1 FAP’s research identified and measured more than 50 rejecting behaviors that parents and caregivers use to respond to their LGBTQ children. Some of these commonly expressed rejecting behaviors include ridiculing and making disparaging comments about their child and other LGBTQ people; excluding them from family activities; blaming their child when others mistreat them because they are LGBTQ; blocking access to LGBTQ resources including friends, support groups, and activities; and trying to change their child’s sexual orientation and gender identity.2 LGBTQ youth experience these and other such behaviors as hurtful, harmful, and traumatic and may feel that they need to hide or repress their identity which can affect their self-esteem, increase isolation, depression, and risky behaviors.3 Providers working with families of LGBTQ youth should focus on shared goals, such as reducing risk and having a happy, healthy child. Most parents love their children and fear for their well-being. However, many are uninformed about their child’s gender identity and sexual orientation and don’t know how to nurture and support them.
In FAP’s initial study, LGB young people who reported higher levels of family rejection had substantially higher rates of attempted suicide, depression, illegal drug use, and unprotected sex.4 These rates were even more significant among Latino gay and bisexual men.4 Those who are rejected by family are less likely to want to have a family or to be parents themselves5 and have lower educational and income levels.6
To reduce risk, pediatricians should ask LGBTQ patients about family rejecting behaviors and help parents and caregivers to identify and understand the effect of such behaviors to reduce health risks and conflict that can lead to running away, expulsion, and removal from the home. Even decreasing rejecting behaviors to moderate levels can significantly improve negative outcomes.5
Caitlin Ryan, PhD, and her team also identified and measured more than 50 family accepting behaviors that help protect against risk and promote well-being. They found that young adults who experience high levels of family acceptance during adolescence report significantly higher levels of self-esteem, social support, and general health with much lower levels of depression, suicidality, and substance abuse.7 Family accepting and supportive behaviors include talking with the child about their LGBTQ identity; advocating for their LGBTQ child when others mistreat them; requiring other family members to treat their LGBTQ child with respect; and supporting their child’s gender identity.5 FAP has developed an evidence-informed family support model and multilingual educational resources for families, providers, youth and religious leaders to decrease rejection and increase family support. These are available in print copies and for download at familyproject.sfsu.edu.
In addition, Dr. Ryan and colleagues1,4,8 recommend the following guidance for providers:
- Ask LGBTQ adolescents about family reactions to their sexual orientation, gender identity, and expression, and refer to LGBTQ community support programs and for supportive counseling, as needed.
- Identify LGBTQ community support programs and online resources to educate parents about how to help their children. Parents need culturally relevant peer support to help decrease rejection and increase family support.
- Advise parents that negative reactions to their adolescent’s LGBTQ identity may negatively impact their child’s health and mental health while supportive and affirming reactions promote well-being.
- Advise parents and caregivers to modify and change family rejecting behaviors that increase their child’s risk for suicide, depression, substance abuse ,and risky sexual behaviors.
- Expand anticipatory guidance to include information on the need for support and the link between family rejection and negative health problems.
- Provide guidance on sexual orientation and gender identity as part of normative child development during well-baby and early childhood care.
- Use FAP’s multilingual family education booklets and Healthy Futures poster series in family and patient education and provide these materials in clinical and community settings. FAP’s Healthy Futures posters include a poster guidance, a version on family acceptance, a version on family rejection and a family acceptance version for conservative families and settings. They are available in camera-ready art in four sizes in English and Spanish and are forthcoming in five Asian languages: familyproject.sfsu.edu/poster.
Dr. Lawlis is assistant professor of pediatrics at the University of Oklahoma Health Sciences Center, Oklahoma City, and an adolescent medicine specialist at OU Children’s. She has no relevant financial disclosures.
Resources
• Family Acceptance Project – consultation and training; evidence-based educational materials for families, providers, religious leaders and youth.
• PFLAG – peer support for parents and friends with LGBTQ children in all states and several other countries.
References
1. Ryan C. Generating a revolution in prevention, wellness & care for LGBT children & youth. Temple Political & Civil Rights Law Review. 2014;23(2):331-44.
2. Ryan C. Healthy Futures Poster Series – Family Accepting & Rejecting Behaviors That Impact LGBTQ Children’s Health & Well-Being. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2019.
3. Ryan C. Family Acceptance Project: Culturally grounded framework for supporting LGBTQ children and youth. J Am Acad Child Adolesc Psychiatr. 2019;58(10):S58-9.
4. Ryan C et al. Family rejection as a predictor of negative health outcomes in White and Latino lesbian, gay, and bisexual young adults. Pediatrics. 2009;123(1):346-52.
5. Ryan C. Supportive families, healthy children: Helping families with lesbian, gay, bisexual & transgender children. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2009.
6. Ryan C et al. Parent-initiated sexual orientation change efforts with LGBT adolescents: Implications for young adult mental health and adjustment. J Homosexuality. 2020;67(2):159-73.
7. Ryan C et al. Family acceptance in adolescence and the health of LGBT young adults. J Child Adolesc Psychiatr Nursing. 2010;23(4):205-13. 8. Substance Abuse and Mental Health Services Administration. A Practitioner’s Guide: Helping Families to Support Their LGBT Children. In: Administration SAaMhS, ed. Vol PEP14-LGBTKIDS. Rockville, MD: HHS Publication; 2014.
It is well established that LGBTQ individuals experience more health disparities compared with their cisgender, heterosexual counterparts. In general, LGBTQ adolescents and young adults have higher levels of depression, suicide attempts, and substance use than those of their heterosexual peers. However, a key protective factor is family acceptance and support. By encouraging families to modify and change behaviors that are experienced by their LGBTQ children as rejecting and to engage in supportive and affirming behaviors, providers can help families to decrease risk and promote healthy outcomes for LGBTQ youth and young adults. 
We all know that a supportive family can make a difference for any child, but this is especially true for LGBTQ youth and is critical during a pandemic when young people are confined with families and separated from peers and supportive adults outside the home. Several research studies show that family support can improve outcomes related to suicide, depression, homelessness, drug use, and HIV in LGBTQ young people. Family acceptance improves health outcomes, while rejection undermines family relationships and worsens both health and other serious outcomes such as homelessness and placement in custodial care. Pediatricians can help their patients by educating parents and caregivers with LGBTQ children about the critical role of family support – both those who see themselves as accepting and those who believe that being gay or transgender is wrong and are struggling with parenting a child who identifies as LGBTQ or who is gender diverse.
The Family Acceptance Project (FAP) at San Francisco State University conducted the first research on LGBTQ youth and families, developed the first evidence-informed family support model, and has published a range of studies and evidence-based resources that demonstrate the harm caused by family rejection, validate the importance of family acceptance, and provide guidance to increase family support. FAP’s research found that parents and caregivers that engage in rejecting behaviors are typically motivated by care and concern and by trying to protect their children from harm. They believe such behaviors will help their LGBTQ children fit in, have a good life, meet cultural and religious expectations, and be respected by others.1 FAP’s research identified and measured more than 50 rejecting behaviors that parents and caregivers use to respond to their LGBTQ children. Some of these commonly expressed rejecting behaviors include ridiculing and making disparaging comments about their child and other LGBTQ people; excluding them from family activities; blaming their child when others mistreat them because they are LGBTQ; blocking access to LGBTQ resources including friends, support groups, and activities; and trying to change their child’s sexual orientation and gender identity.2 LGBTQ youth experience these and other such behaviors as hurtful, harmful, and traumatic and may feel that they need to hide or repress their identity which can affect their self-esteem, increase isolation, depression, and risky behaviors.3 Providers working with families of LGBTQ youth should focus on shared goals, such as reducing risk and having a happy, healthy child. Most parents love their children and fear for their well-being. However, many are uninformed about their child’s gender identity and sexual orientation and don’t know how to nurture and support them.
In FAP’s initial study, LGB young people who reported higher levels of family rejection had substantially higher rates of attempted suicide, depression, illegal drug use, and unprotected sex.4 These rates were even more significant among Latino gay and bisexual men.4 Those who are rejected by family are less likely to want to have a family or to be parents themselves5 and have lower educational and income levels.6
To reduce risk, pediatricians should ask LGBTQ patients about family rejecting behaviors and help parents and caregivers to identify and understand the effect of such behaviors to reduce health risks and conflict that can lead to running away, expulsion, and removal from the home. Even decreasing rejecting behaviors to moderate levels can significantly improve negative outcomes.5
Caitlin Ryan, PhD, and her team also identified and measured more than 50 family accepting behaviors that help protect against risk and promote well-being. They found that young adults who experience high levels of family acceptance during adolescence report significantly higher levels of self-esteem, social support, and general health with much lower levels of depression, suicidality, and substance abuse.7 Family accepting and supportive behaviors include talking with the child about their LGBTQ identity; advocating for their LGBTQ child when others mistreat them; requiring other family members to treat their LGBTQ child with respect; and supporting their child’s gender identity.5 FAP has developed an evidence-informed family support model and multilingual educational resources for families, providers, youth and religious leaders to decrease rejection and increase family support. These are available in print copies and for download at familyproject.sfsu.edu.
In addition, Dr. Ryan and colleagues1,4,8 recommend the following guidance for providers:
- Ask LGBTQ adolescents about family reactions to their sexual orientation, gender identity, and expression, and refer to LGBTQ community support programs and for supportive counseling, as needed.
- Identify LGBTQ community support programs and online resources to educate parents about how to help their children. Parents need culturally relevant peer support to help decrease rejection and increase family support.
- Advise parents that negative reactions to their adolescent’s LGBTQ identity may negatively impact their child’s health and mental health while supportive and affirming reactions promote well-being.
- Advise parents and caregivers to modify and change family rejecting behaviors that increase their child’s risk for suicide, depression, substance abuse ,and risky sexual behaviors.
- Expand anticipatory guidance to include information on the need for support and the link between family rejection and negative health problems.
- Provide guidance on sexual orientation and gender identity as part of normative child development during well-baby and early childhood care.
- Use FAP’s multilingual family education booklets and Healthy Futures poster series in family and patient education and provide these materials in clinical and community settings. FAP’s Healthy Futures posters include a poster guidance, a version on family acceptance, a version on family rejection and a family acceptance version for conservative families and settings. They are available in camera-ready art in four sizes in English and Spanish and are forthcoming in five Asian languages: familyproject.sfsu.edu/poster.
Dr. Lawlis is assistant professor of pediatrics at the University of Oklahoma Health Sciences Center, Oklahoma City, and an adolescent medicine specialist at OU Children’s. She has no relevant financial disclosures.
Resources
• Family Acceptance Project – consultation and training; evidence-based educational materials for families, providers, religious leaders and youth.
• PFLAG – peer support for parents and friends with LGBTQ children in all states and several other countries.
References
1. Ryan C. Generating a revolution in prevention, wellness & care for LGBT children & youth. Temple Political & Civil Rights Law Review. 2014;23(2):331-44.
2. Ryan C. Healthy Futures Poster Series – Family Accepting & Rejecting Behaviors That Impact LGBTQ Children’s Health & Well-Being. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2019.
3. Ryan C. Family Acceptance Project: Culturally grounded framework for supporting LGBTQ children and youth. J Am Acad Child Adolesc Psychiatr. 2019;58(10):S58-9.
4. Ryan C et al. Family rejection as a predictor of negative health outcomes in White and Latino lesbian, gay, and bisexual young adults. Pediatrics. 2009;123(1):346-52.
5. Ryan C. Supportive families, healthy children: Helping families with lesbian, gay, bisexual & transgender children. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2009.
6. Ryan C et al. Parent-initiated sexual orientation change efforts with LGBT adolescents: Implications for young adult mental health and adjustment. J Homosexuality. 2020;67(2):159-73.
7. Ryan C et al. Family acceptance in adolescence and the health of LGBT young adults. J Child Adolesc Psychiatr Nursing. 2010;23(4):205-13. 8. Substance Abuse and Mental Health Services Administration. A Practitioner’s Guide: Helping Families to Support Their LGBT Children. In: Administration SAaMhS, ed. Vol PEP14-LGBTKIDS. Rockville, MD: HHS Publication; 2014.
It is well established that LGBTQ individuals experience more health disparities compared with their cisgender, heterosexual counterparts. In general, LGBTQ adolescents and young adults have higher levels of depression, suicide attempts, and substance use than those of their heterosexual peers. However, a key protective factor is family acceptance and support. By encouraging families to modify and change behaviors that are experienced by their LGBTQ children as rejecting and to engage in supportive and affirming behaviors, providers can help families to decrease risk and promote healthy outcomes for LGBTQ youth and young adults. 
We all know that a supportive family can make a difference for any child, but this is especially true for LGBTQ youth and is critical during a pandemic when young people are confined with families and separated from peers and supportive adults outside the home. Several research studies show that family support can improve outcomes related to suicide, depression, homelessness, drug use, and HIV in LGBTQ young people. Family acceptance improves health outcomes, while rejection undermines family relationships and worsens both health and other serious outcomes such as homelessness and placement in custodial care. Pediatricians can help their patients by educating parents and caregivers with LGBTQ children about the critical role of family support – both those who see themselves as accepting and those who believe that being gay or transgender is wrong and are struggling with parenting a child who identifies as LGBTQ or who is gender diverse.
The Family Acceptance Project (FAP) at San Francisco State University conducted the first research on LGBTQ youth and families, developed the first evidence-informed family support model, and has published a range of studies and evidence-based resources that demonstrate the harm caused by family rejection, validate the importance of family acceptance, and provide guidance to increase family support. FAP’s research found that parents and caregivers that engage in rejecting behaviors are typically motivated by care and concern and by trying to protect their children from harm. They believe such behaviors will help their LGBTQ children fit in, have a good life, meet cultural and religious expectations, and be respected by others.1 FAP’s research identified and measured more than 50 rejecting behaviors that parents and caregivers use to respond to their LGBTQ children. Some of these commonly expressed rejecting behaviors include ridiculing and making disparaging comments about their child and other LGBTQ people; excluding them from family activities; blaming their child when others mistreat them because they are LGBTQ; blocking access to LGBTQ resources including friends, support groups, and activities; and trying to change their child’s sexual orientation and gender identity.2 LGBTQ youth experience these and other such behaviors as hurtful, harmful, and traumatic and may feel that they need to hide or repress their identity which can affect their self-esteem, increase isolation, depression, and risky behaviors.3 Providers working with families of LGBTQ youth should focus on shared goals, such as reducing risk and having a happy, healthy child. Most parents love their children and fear for their well-being. However, many are uninformed about their child’s gender identity and sexual orientation and don’t know how to nurture and support them.
In FAP’s initial study, LGB young people who reported higher levels of family rejection had substantially higher rates of attempted suicide, depression, illegal drug use, and unprotected sex.4 These rates were even more significant among Latino gay and bisexual men.4 Those who are rejected by family are less likely to want to have a family or to be parents themselves5 and have lower educational and income levels.6
To reduce risk, pediatricians should ask LGBTQ patients about family rejecting behaviors and help parents and caregivers to identify and understand the effect of such behaviors to reduce health risks and conflict that can lead to running away, expulsion, and removal from the home. Even decreasing rejecting behaviors to moderate levels can significantly improve negative outcomes.5
Caitlin Ryan, PhD, and her team also identified and measured more than 50 family accepting behaviors that help protect against risk and promote well-being. They found that young adults who experience high levels of family acceptance during adolescence report significantly higher levels of self-esteem, social support, and general health with much lower levels of depression, suicidality, and substance abuse.7 Family accepting and supportive behaviors include talking with the child about their LGBTQ identity; advocating for their LGBTQ child when others mistreat them; requiring other family members to treat their LGBTQ child with respect; and supporting their child’s gender identity.5 FAP has developed an evidence-informed family support model and multilingual educational resources for families, providers, youth and religious leaders to decrease rejection and increase family support. These are available in print copies and for download at familyproject.sfsu.edu.
In addition, Dr. Ryan and colleagues1,4,8 recommend the following guidance for providers:
- Ask LGBTQ adolescents about family reactions to their sexual orientation, gender identity, and expression, and refer to LGBTQ community support programs and for supportive counseling, as needed.
- Identify LGBTQ community support programs and online resources to educate parents about how to help their children. Parents need culturally relevant peer support to help decrease rejection and increase family support.
- Advise parents that negative reactions to their adolescent’s LGBTQ identity may negatively impact their child’s health and mental health while supportive and affirming reactions promote well-being.
- Advise parents and caregivers to modify and change family rejecting behaviors that increase their child’s risk for suicide, depression, substance abuse ,and risky sexual behaviors.
- Expand anticipatory guidance to include information on the need for support and the link between family rejection and negative health problems.
- Provide guidance on sexual orientation and gender identity as part of normative child development during well-baby and early childhood care.
- Use FAP’s multilingual family education booklets and Healthy Futures poster series in family and patient education and provide these materials in clinical and community settings. FAP’s Healthy Futures posters include a poster guidance, a version on family acceptance, a version on family rejection and a family acceptance version for conservative families and settings. They are available in camera-ready art in four sizes in English and Spanish and are forthcoming in five Asian languages: familyproject.sfsu.edu/poster.
Dr. Lawlis is assistant professor of pediatrics at the University of Oklahoma Health Sciences Center, Oklahoma City, and an adolescent medicine specialist at OU Children’s. She has no relevant financial disclosures.
Resources
• Family Acceptance Project – consultation and training; evidence-based educational materials for families, providers, religious leaders and youth.
• PFLAG – peer support for parents and friends with LGBTQ children in all states and several other countries.
References
1. Ryan C. Generating a revolution in prevention, wellness & care for LGBT children & youth. Temple Political & Civil Rights Law Review. 2014;23(2):331-44.
2. Ryan C. Healthy Futures Poster Series – Family Accepting & Rejecting Behaviors That Impact LGBTQ Children’s Health & Well-Being. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2019.
3. Ryan C. Family Acceptance Project: Culturally grounded framework for supporting LGBTQ children and youth. J Am Acad Child Adolesc Psychiatr. 2019;58(10):S58-9.
4. Ryan C et al. Family rejection as a predictor of negative health outcomes in White and Latino lesbian, gay, and bisexual young adults. Pediatrics. 2009;123(1):346-52.
5. Ryan C. Supportive families, healthy children: Helping families with lesbian, gay, bisexual & transgender children. In: Family Acceptance Project Marian Wright Edelman Institute SFSU, ed. San Francisco, CA2009.
6. Ryan C et al. Parent-initiated sexual orientation change efforts with LGBT adolescents: Implications for young adult mental health and adjustment. J Homosexuality. 2020;67(2):159-73.
7. Ryan C et al. Family acceptance in adolescence and the health of LGBT young adults. J Child Adolesc Psychiatr Nursing. 2010;23(4):205-13. 8. Substance Abuse and Mental Health Services Administration. A Practitioner’s Guide: Helping Families to Support Their LGBT Children. In: Administration SAaMhS, ed. Vol PEP14-LGBTKIDS. Rockville, MD: HHS Publication; 2014.













