User login
Cancer patients report delays in treatment because of COVID-19
As the COVID-19 pandemic continues, many cancer patients are finding it increasingly difficult to receive the care they need and are facing financial challenges.
Half of the cancer patients and survivors who responded to a recent survey reported changes, delays, or disruptions to the care they were receiving. The survey, with 1,219 respondents, was conducted by the American Cancer Society Cancer Action Network (ACS CAN).
“The circumstances of this virus – from the fact cancer patients are at higher risk of severe complications should they be diagnosed with COVID-19, to the fact many patients are facing serious financial strain caused by the virus’ economic effect – make getting care especially difficult,” Keysha Brooks-Coley, vice president of federal advocacy for ACS CAN, told Medscape Medical News.
Nearly a quarter (24%) of survey respondents reported a delay in care or treatment. The proportion was slightly more (27%) among those currently receiving active treatment.
In addition, 12% (13% in active treatment) stated that not only was their care delayed but that they also have not been told when services would be rescheduled.
As previously reported by Medscape Medical News, many oncology groups have issued new guidelines for cancer care in reaction to the current crisis. These include recommendations to delay cancer treatment in order to avoid exposing cancer patients to the virus.
Half of those in active treatment report disruptions
The survey was initiated by ACS CAN on March 25 and was distributed over a 2-week period. The goal was to gain a better understanding of how COVID-19 was affecting cancer patients and survivors in the United States. Of the 1,219 respondents, half (51%) were cancer patients currently undergoing active treatment.
Among the patients and survivors who were currently in active treatment, 55% reported that there have been changes, delays, or disruptions in their care. The services most frequently affected included in-person provider visits (50%), supportive services (20%), and imaging procedures to monitor tumor growth (20%).
In addition, 8% reported that their treatment, including chemotherapy and immunotherapy, had been affected by the COVID-19 pandemic.
Financial concerns
Almost all of the survey respondents were covered by some type of insurance; 49% had coverage through an employer, 32% were covered by Medicare, 7% had privately purchased insurance, and 4% were covered through Medicaid.
Many cancer patients had already been having difficulty paying for their care, but for a substantial proportion of survey respondents, the COVID-19 pandemic has exacerbated the problem. More than one-third (38%) stated that COVID-19 “has had a notable impact on their financial situation that affects their ability to pay for health care.”
The most common financial problems that were related to access to care include reduced work hours (14%), reduced investment values (11%), having difficulty affording food and supplies because of staying at home to avoid contracting the virus (9%), and becoming unemployed (8%).
A reduction in work hours and job loss were of particular concern to respondents because of the possible effects these would have on their health insurance coverage. Of those who reported that they or a family member living with them had lost a job, 43% had employer-sponsored health insurance. Additionally, 58% of patients or a family member whose working hours had been reduced also had health insurance through their employer
Among the entire cohort, 28% reported that they were worried that the financial impact of COVID-19 would make it difficult to pay for the health care they need as cancer survivors. This concern was highly correlated with income. Almost half (46%) of patients who earned $30,000 or less reported that they were worried, but even in household with incomes over $110,000 per year, 21% were also concerned about the financial impact.
“Now more than ever, patients need to be able to get, keep, and afford health coverage to treat their disease,” commented Brooks-Coley.
Taking action
“ACS CAN is working every day to make clear to Congress and the administration the real and immediate challenges cancer patients and survivors face during this pandemic,” said Brooks-Coley.
With nearly 50 other professional and advocacy groups, ACS CAN has sent letters to congressional leadership and the Secretary of the Department of Health & Human Services asking them to make policy changes that would help patients.
The proposed action points include having insurers allow patients to use providers who are out of network if necessary; waiving site-specific precertification and prior authorization for cancer treatment; utilizing shared decision making between patients and providers in deciding whether to use home infusion without pressure from the insurer; allowing patients to obtain 90-day supplies of medication; increasing funding for state Medicaid programs and assistance for those who have lost employee-sponsored coverage; and improving telehealth services.
“We urge Congress and the administration to keep the needs of cancer patients and survivors in mind as they continue to address the public health crisis,” she said.
This article first appeared on Medscape.com.
As the COVID-19 pandemic continues, many cancer patients are finding it increasingly difficult to receive the care they need and are facing financial challenges.
Half of the cancer patients and survivors who responded to a recent survey reported changes, delays, or disruptions to the care they were receiving. The survey, with 1,219 respondents, was conducted by the American Cancer Society Cancer Action Network (ACS CAN).
“The circumstances of this virus – from the fact cancer patients are at higher risk of severe complications should they be diagnosed with COVID-19, to the fact many patients are facing serious financial strain caused by the virus’ economic effect – make getting care especially difficult,” Keysha Brooks-Coley, vice president of federal advocacy for ACS CAN, told Medscape Medical News.
Nearly a quarter (24%) of survey respondents reported a delay in care or treatment. The proportion was slightly more (27%) among those currently receiving active treatment.
In addition, 12% (13% in active treatment) stated that not only was their care delayed but that they also have not been told when services would be rescheduled.
As previously reported by Medscape Medical News, many oncology groups have issued new guidelines for cancer care in reaction to the current crisis. These include recommendations to delay cancer treatment in order to avoid exposing cancer patients to the virus.
Half of those in active treatment report disruptions
The survey was initiated by ACS CAN on March 25 and was distributed over a 2-week period. The goal was to gain a better understanding of how COVID-19 was affecting cancer patients and survivors in the United States. Of the 1,219 respondents, half (51%) were cancer patients currently undergoing active treatment.
Among the patients and survivors who were currently in active treatment, 55% reported that there have been changes, delays, or disruptions in their care. The services most frequently affected included in-person provider visits (50%), supportive services (20%), and imaging procedures to monitor tumor growth (20%).
In addition, 8% reported that their treatment, including chemotherapy and immunotherapy, had been affected by the COVID-19 pandemic.
Financial concerns
Almost all of the survey respondents were covered by some type of insurance; 49% had coverage through an employer, 32% were covered by Medicare, 7% had privately purchased insurance, and 4% were covered through Medicaid.
Many cancer patients had already been having difficulty paying for their care, but for a substantial proportion of survey respondents, the COVID-19 pandemic has exacerbated the problem. More than one-third (38%) stated that COVID-19 “has had a notable impact on their financial situation that affects their ability to pay for health care.”
The most common financial problems that were related to access to care include reduced work hours (14%), reduced investment values (11%), having difficulty affording food and supplies because of staying at home to avoid contracting the virus (9%), and becoming unemployed (8%).
A reduction in work hours and job loss were of particular concern to respondents because of the possible effects these would have on their health insurance coverage. Of those who reported that they or a family member living with them had lost a job, 43% had employer-sponsored health insurance. Additionally, 58% of patients or a family member whose working hours had been reduced also had health insurance through their employer
Among the entire cohort, 28% reported that they were worried that the financial impact of COVID-19 would make it difficult to pay for the health care they need as cancer survivors. This concern was highly correlated with income. Almost half (46%) of patients who earned $30,000 or less reported that they were worried, but even in household with incomes over $110,000 per year, 21% were also concerned about the financial impact.
“Now more than ever, patients need to be able to get, keep, and afford health coverage to treat their disease,” commented Brooks-Coley.
Taking action
“ACS CAN is working every day to make clear to Congress and the administration the real and immediate challenges cancer patients and survivors face during this pandemic,” said Brooks-Coley.
With nearly 50 other professional and advocacy groups, ACS CAN has sent letters to congressional leadership and the Secretary of the Department of Health & Human Services asking them to make policy changes that would help patients.
The proposed action points include having insurers allow patients to use providers who are out of network if necessary; waiving site-specific precertification and prior authorization for cancer treatment; utilizing shared decision making between patients and providers in deciding whether to use home infusion without pressure from the insurer; allowing patients to obtain 90-day supplies of medication; increasing funding for state Medicaid programs and assistance for those who have lost employee-sponsored coverage; and improving telehealth services.
“We urge Congress and the administration to keep the needs of cancer patients and survivors in mind as they continue to address the public health crisis,” she said.
This article first appeared on Medscape.com.
As the COVID-19 pandemic continues, many cancer patients are finding it increasingly difficult to receive the care they need and are facing financial challenges.
Half of the cancer patients and survivors who responded to a recent survey reported changes, delays, or disruptions to the care they were receiving. The survey, with 1,219 respondents, was conducted by the American Cancer Society Cancer Action Network (ACS CAN).
“The circumstances of this virus – from the fact cancer patients are at higher risk of severe complications should they be diagnosed with COVID-19, to the fact many patients are facing serious financial strain caused by the virus’ economic effect – make getting care especially difficult,” Keysha Brooks-Coley, vice president of federal advocacy for ACS CAN, told Medscape Medical News.
Nearly a quarter (24%) of survey respondents reported a delay in care or treatment. The proportion was slightly more (27%) among those currently receiving active treatment.
In addition, 12% (13% in active treatment) stated that not only was their care delayed but that they also have not been told when services would be rescheduled.
As previously reported by Medscape Medical News, many oncology groups have issued new guidelines for cancer care in reaction to the current crisis. These include recommendations to delay cancer treatment in order to avoid exposing cancer patients to the virus.
Half of those in active treatment report disruptions
The survey was initiated by ACS CAN on March 25 and was distributed over a 2-week period. The goal was to gain a better understanding of how COVID-19 was affecting cancer patients and survivors in the United States. Of the 1,219 respondents, half (51%) were cancer patients currently undergoing active treatment.
Among the patients and survivors who were currently in active treatment, 55% reported that there have been changes, delays, or disruptions in their care. The services most frequently affected included in-person provider visits (50%), supportive services (20%), and imaging procedures to monitor tumor growth (20%).
In addition, 8% reported that their treatment, including chemotherapy and immunotherapy, had been affected by the COVID-19 pandemic.
Financial concerns
Almost all of the survey respondents were covered by some type of insurance; 49% had coverage through an employer, 32% were covered by Medicare, 7% had privately purchased insurance, and 4% were covered through Medicaid.
Many cancer patients had already been having difficulty paying for their care, but for a substantial proportion of survey respondents, the COVID-19 pandemic has exacerbated the problem. More than one-third (38%) stated that COVID-19 “has had a notable impact on their financial situation that affects their ability to pay for health care.”
The most common financial problems that were related to access to care include reduced work hours (14%), reduced investment values (11%), having difficulty affording food and supplies because of staying at home to avoid contracting the virus (9%), and becoming unemployed (8%).
A reduction in work hours and job loss were of particular concern to respondents because of the possible effects these would have on their health insurance coverage. Of those who reported that they or a family member living with them had lost a job, 43% had employer-sponsored health insurance. Additionally, 58% of patients or a family member whose working hours had been reduced also had health insurance through their employer
Among the entire cohort, 28% reported that they were worried that the financial impact of COVID-19 would make it difficult to pay for the health care they need as cancer survivors. This concern was highly correlated with income. Almost half (46%) of patients who earned $30,000 or less reported that they were worried, but even in household with incomes over $110,000 per year, 21% were also concerned about the financial impact.
“Now more than ever, patients need to be able to get, keep, and afford health coverage to treat their disease,” commented Brooks-Coley.
Taking action
“ACS CAN is working every day to make clear to Congress and the administration the real and immediate challenges cancer patients and survivors face during this pandemic,” said Brooks-Coley.
With nearly 50 other professional and advocacy groups, ACS CAN has sent letters to congressional leadership and the Secretary of the Department of Health & Human Services asking them to make policy changes that would help patients.
The proposed action points include having insurers allow patients to use providers who are out of network if necessary; waiving site-specific precertification and prior authorization for cancer treatment; utilizing shared decision making between patients and providers in deciding whether to use home infusion without pressure from the insurer; allowing patients to obtain 90-day supplies of medication; increasing funding for state Medicaid programs and assistance for those who have lost employee-sponsored coverage; and improving telehealth services.
“We urge Congress and the administration to keep the needs of cancer patients and survivors in mind as they continue to address the public health crisis,” she said.
This article first appeared on Medscape.com.
Cancer care ‘transformed in space of a month’ because of pandemic
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
, the most “revolutionary” being a deep dive into telehealth, predicts Deborah Schrag, MD, MPH, a medical oncologist specializing in gastrointestinal cancers at the Dana Farber Cancer Institute in Boston, Massachusetts.
“In the space of a month, approaches and accepted norms of cancer care delivery have been transformed of necessity,” Schrag and colleagues write in an article published in JAMA on April 13.
“Most of these changes would not have occurred without the pandemic,” they add. They predict that some changes will last after the crisis is over.
“None of us want to be thrown in the deep end.... On the other hand, sometimes it works,” Schrag told Medscape Medical News.
“The in-person visit between patient and physician has been upended,” she said.
“I don’t think there’s any going back to the way it was before because cancer patients won’t stand for it,” she said. “They’re not going to drive in to get the results of a blood test.
“I think that on balance, of course, there are situations where you need eye-to-eye contact. No one wants to have an initial oncology meeting by telehealth – doctors or patients – that’s ridiculous,” she said. “But for follow-up visits, patients are now going to be more demanding, and doctors will be more willing.”
The “essential empathy” of oncologists can still “transcend the new physical barriers presented by masks and telehealth,” Schrag and colleagues comment.
“Doctors are figuring out how to deliver empathy by Zoom,” she told Medscape Medical News. “It’s not the same, but we all convey empathy to our elderly relatives over the phone.”
Pandemic impact on oncology
While the crisis has affected all of medicine – dismantling how care is delivered and forcing clinicians to make difficult decisions regarding triage – the fact that some cancers present an immediate threat to survival means that oncology “provides a lens into the major shifts currently underway in clinical care,” Schrag and colleagues write.
They illustrate the point by highlighting systemic chemotherapy, which is provided to a large proportion of patients with advanced cancer. The pandemic has tipped the risk-benefit ratio away from treatments that have a marginal effect on quality or quantity of life, they note. It has forced an “elimination of low-value treatments that were identified by the Choosing Wisely campaign,” the authors write. Up to now, the uptake of recommendations to eliminate these treatments has been slow.
“For example, for most metastatic solid tumors, chemotherapy beyond the third regimen does not improve survival for more than a few weeks; therefore, oncologists are advising supportive care instead. For patients receiving adjuvant therapy for curable cancers, delaying initiation or abbreviating the number of cycles is appropriate. Oncologists are postponing initiation of adjuvant chemotherapy for some estrogen receptor–negative stage II breast cancers by 8 weeks and administering 6 rather than 12 cycles of adjuvant chemotherapy for stage III colorectal cancers,” Schrag and colleagues write.
On the other hand, even in the epicenters of the pandemic, thus far, oncologists are still delivering cancer treatments that have the potential to cure and cannot safely be delayed, they point out. “This includes most patients with new diagnoses of acute leukemia, high-grade lymphoma, and those with chemotherapy-responsive tumors such as testicular, ovarian, and small cell lung cancer. Despite the risks, oncologists are not modifying such treatments because these cancers are likely more lethal than COVID-19.”
It’s the cancer patients who fall in between these two extremes who pose the biggest treatment challenge during this crisis – the patients for whom a delay would have “moderate clinically important adverse influence on quality of life or survival.” In these cases, oncologists are “prescribing marginally less effective regimens that have lower risk of precipitating hospitalization,” the authors note.
These treatments include the use of “white cell growth factor, more stringent neutrophil counts for proceeding with a next cycle of therapy, and omitting use of steroids to manage nausea.” In addition, where possible, oncologists are substituting oral agents for intravenous agents and “myriad other modifications to minimize visits and hospitalizations.”
Most hospitals and outpatient infusion centers now prohibit visitors from accompanying patients, and oncologists are prioritizing conversations with patients about advance directives, healthcare proxies, and end-of-life care preferences. Yet, even here, telehealth offers a new, enhanced layer to those conversations by enabling families to gather with their loved one and the doctor, she said.
This article first appeared on Medscape.com.
Breast-conserving surgery deemed okay in high-risk hereditary breast cancers
It’s okay to consider breast-conserving therapy in breast cancer patients with high-risk hereditary genetic mutations, according to guidelines published in the Journal of Clinical Oncology.
The presence of a germline BRCA1 or BRCA2 mutation shouldn’t preclude breast-conserving therapy as long as the patient is otherwise eligible for the procedure, according to the guidelines, which were developed by an expert panel convened by the American Society of Clinical Oncology, American Society for Radiation Oncology, and Society for Surgical Oncology.
Nadine M. Tung, MD, of Beth Israel Deaconess Medical Center in Boston and the rest of the expert panel reviewed evidence from 58 published articles to create the guidelines.
In addition to supporting use of breast-conserving therapy, the guidelines suggest that radiation shouldn’t be withheld because of mutation status, except in patients with TP53 mutations. Furthermore, BRCA1/2 mutation carriers with metastatic HER2-negative disease can receive the poly (ADP-ribose) polymerase (PARP) inhibitors olaparib and talazoparib as an “alternative to chemotherapy” for first-, second-, or third-line therapy.
However, it’s the “license to consider breast-conserving therapy” for high-risk individuals that is one of the most noteworthy points in the guidelines, and the one that may surprise some readers, according to William J. Gradishar, MD, of Northwestern University in Chicago, who was not involved in developing the guidelines.
“We don’t have to be as dogmatic with these patients with respect to local therapies as we were in the past,” Dr. Gradishar said in an interview. “That’s a good thing for patients, but you also have to understand the nuances that go into recommending [breast-conserving surgery] to a patient. Other variables, like the age at which the patient develops breast cancer, family history, etc., all go into it.”
Weighing options for surgery
The guidelines emphasize that, for patients with germline BRCA1/2 mutations, health care providers need to discuss treatment options for the breast cancer at hand. However, patients should also be made aware of their increased risk of contralateral and new ipsilateral breast cancer as compared with noncarriers.
When weighing breast-conserving therapy versus mastectomy in light of contralateral breast cancer risk, the guidelines recommend considering not only age at diagnosis – the strongest predictor of a later contralateral breast cancer – but also family history, comorbidities, life expectancy, ability to undergo MRI, and prognosis from breast or other cancers, such as ovarian cancer.
If a bilateral mastectomy isn’t performed in a BRCA1/2 mutation carrier, an annual mammogram and MRI are warranted thereafter for screening of the remaining breast tissue, according to the guidelines.
The guidelines say breast-conserving therapy should be offered to patients with mutations in moderate-penetrance genes, including PALB2, CHEK2, and ATM. However, there’s not much data regarding the risk of ipsilateral breast cancer after breast-conserving therapy in these patients.
Likewise, there’s limited evidence on contralateral breast cancer risk for patients with mutations in moderate-penetrance genes aside from CHEK2. The guidelines say the risk should be discussed with patients “in the context of shared decision making.”
Nipple-sparing mastectomy is “reasonable” to consider in certain newly diagnosed patients with BRCA1/2 mutations, as well as in newly diagnosed patients with moderate-risk mutations, the guidelines state.
Women with breast cancer and a deleterious BRCA1/2 mutation who are undergoing unilateral mastectomy should be offered contralateral risk-reducing mastectomy. Likewise, women with moderate-risk mutations should be offered contralateral risk-reducing mastectomy, but not solely based on mutation status, according to the guidelines. Data are limited on contralateral breast cancer risk related to those mutations.
Considerations for radiation
Radiation therapy in the context of breast-conserving therapy or mastectomy should not be withheld because of hereditary mutations, except in the case of TP53 mutations, according to the guidelines.
There’s no evidence that radiotherapy increases toxicity or contralateral breast cancer risk for most BRCA1/2 or moderate-penetrance gene mutations. However, the intact breast shouldn’t be irradiated in germline TP53 mutation carriers, the guidelines say, because of the important role that TP53 plays in the ability to repair DNA damage after cellular stress.
“Carriers of a TP53 mutation would be expected to be unable to repair tissue damage from DNA damaging radiotherapy and be at risk for significant [radiotherapy]-associated sequelae,” the guidelines state.
Chemotherapy and PARP inhibitors
For women with metastatic breast cancer harboring germline BRCA1/2 mutations, the guidelines say platinum chemotherapy should be preferred over taxanes for platinum-naive patients.
Provided the breast cancer is HER2 negative, the PARP inhibitors olaparib or talazoparib “should be offered as an alternative to chemotherapy in the first- to third-line settings,” the guidelines state.
The guidelines confirm that PARP inhibitors are a “valid starting point” for treatment of BCRA1/2–associated metastatic breast cancer, Dr. Gradishar said.
“When a patient progresses on a PARP inhibitor, assuming they’re not going on some other investigational drug or clinical trial, they’re going to get chemotherapy,” he said. “So the argument is that, if you have something that’s at least as good or maybe a little bit better and has fewer side effects, why not start with that and then move on to other things?”
By contrast, there’s not enough evidence to recommend PARP inhibitors for germline BRCA mutation carriers with nonmetastatic breast cancers, according to the guidelines, and there’s “no robust data” for using PARP inhibitors in patients with breast cancers with mutations in moderate-penetrance genes.
The guideline authors disclosed relationships with AstraZeneca, Myriad Genetics, Pfizer, Lilly, and other companies. Dr. Gradishar has relationships with AstraZeneca, Celltrion, Genentech, MacroGenics, Merck, Pfizer, and Seattle Genetics.
SOURCE: Tung NM et al. J Clin Oncol. 2020 Apr 3;JCO2000299. doi: 10.1200/JCO.20.00299.
It’s okay to consider breast-conserving therapy in breast cancer patients with high-risk hereditary genetic mutations, according to guidelines published in the Journal of Clinical Oncology.
The presence of a germline BRCA1 or BRCA2 mutation shouldn’t preclude breast-conserving therapy as long as the patient is otherwise eligible for the procedure, according to the guidelines, which were developed by an expert panel convened by the American Society of Clinical Oncology, American Society for Radiation Oncology, and Society for Surgical Oncology.
Nadine M. Tung, MD, of Beth Israel Deaconess Medical Center in Boston and the rest of the expert panel reviewed evidence from 58 published articles to create the guidelines.
In addition to supporting use of breast-conserving therapy, the guidelines suggest that radiation shouldn’t be withheld because of mutation status, except in patients with TP53 mutations. Furthermore, BRCA1/2 mutation carriers with metastatic HER2-negative disease can receive the poly (ADP-ribose) polymerase (PARP) inhibitors olaparib and talazoparib as an “alternative to chemotherapy” for first-, second-, or third-line therapy.
However, it’s the “license to consider breast-conserving therapy” for high-risk individuals that is one of the most noteworthy points in the guidelines, and the one that may surprise some readers, according to William J. Gradishar, MD, of Northwestern University in Chicago, who was not involved in developing the guidelines.
“We don’t have to be as dogmatic with these patients with respect to local therapies as we were in the past,” Dr. Gradishar said in an interview. “That’s a good thing for patients, but you also have to understand the nuances that go into recommending [breast-conserving surgery] to a patient. Other variables, like the age at which the patient develops breast cancer, family history, etc., all go into it.”
Weighing options for surgery
The guidelines emphasize that, for patients with germline BRCA1/2 mutations, health care providers need to discuss treatment options for the breast cancer at hand. However, patients should also be made aware of their increased risk of contralateral and new ipsilateral breast cancer as compared with noncarriers.
When weighing breast-conserving therapy versus mastectomy in light of contralateral breast cancer risk, the guidelines recommend considering not only age at diagnosis – the strongest predictor of a later contralateral breast cancer – but also family history, comorbidities, life expectancy, ability to undergo MRI, and prognosis from breast or other cancers, such as ovarian cancer.
If a bilateral mastectomy isn’t performed in a BRCA1/2 mutation carrier, an annual mammogram and MRI are warranted thereafter for screening of the remaining breast tissue, according to the guidelines.
The guidelines say breast-conserving therapy should be offered to patients with mutations in moderate-penetrance genes, including PALB2, CHEK2, and ATM. However, there’s not much data regarding the risk of ipsilateral breast cancer after breast-conserving therapy in these patients.
Likewise, there’s limited evidence on contralateral breast cancer risk for patients with mutations in moderate-penetrance genes aside from CHEK2. The guidelines say the risk should be discussed with patients “in the context of shared decision making.”
Nipple-sparing mastectomy is “reasonable” to consider in certain newly diagnosed patients with BRCA1/2 mutations, as well as in newly diagnosed patients with moderate-risk mutations, the guidelines state.
Women with breast cancer and a deleterious BRCA1/2 mutation who are undergoing unilateral mastectomy should be offered contralateral risk-reducing mastectomy. Likewise, women with moderate-risk mutations should be offered contralateral risk-reducing mastectomy, but not solely based on mutation status, according to the guidelines. Data are limited on contralateral breast cancer risk related to those mutations.
Considerations for radiation
Radiation therapy in the context of breast-conserving therapy or mastectomy should not be withheld because of hereditary mutations, except in the case of TP53 mutations, according to the guidelines.
There’s no evidence that radiotherapy increases toxicity or contralateral breast cancer risk for most BRCA1/2 or moderate-penetrance gene mutations. However, the intact breast shouldn’t be irradiated in germline TP53 mutation carriers, the guidelines say, because of the important role that TP53 plays in the ability to repair DNA damage after cellular stress.
“Carriers of a TP53 mutation would be expected to be unable to repair tissue damage from DNA damaging radiotherapy and be at risk for significant [radiotherapy]-associated sequelae,” the guidelines state.
Chemotherapy and PARP inhibitors
For women with metastatic breast cancer harboring germline BRCA1/2 mutations, the guidelines say platinum chemotherapy should be preferred over taxanes for platinum-naive patients.
Provided the breast cancer is HER2 negative, the PARP inhibitors olaparib or talazoparib “should be offered as an alternative to chemotherapy in the first- to third-line settings,” the guidelines state.
The guidelines confirm that PARP inhibitors are a “valid starting point” for treatment of BCRA1/2–associated metastatic breast cancer, Dr. Gradishar said.
“When a patient progresses on a PARP inhibitor, assuming they’re not going on some other investigational drug or clinical trial, they’re going to get chemotherapy,” he said. “So the argument is that, if you have something that’s at least as good or maybe a little bit better and has fewer side effects, why not start with that and then move on to other things?”
By contrast, there’s not enough evidence to recommend PARP inhibitors for germline BRCA mutation carriers with nonmetastatic breast cancers, according to the guidelines, and there’s “no robust data” for using PARP inhibitors in patients with breast cancers with mutations in moderate-penetrance genes.
The guideline authors disclosed relationships with AstraZeneca, Myriad Genetics, Pfizer, Lilly, and other companies. Dr. Gradishar has relationships with AstraZeneca, Celltrion, Genentech, MacroGenics, Merck, Pfizer, and Seattle Genetics.
SOURCE: Tung NM et al. J Clin Oncol. 2020 Apr 3;JCO2000299. doi: 10.1200/JCO.20.00299.
It’s okay to consider breast-conserving therapy in breast cancer patients with high-risk hereditary genetic mutations, according to guidelines published in the Journal of Clinical Oncology.
The presence of a germline BRCA1 or BRCA2 mutation shouldn’t preclude breast-conserving therapy as long as the patient is otherwise eligible for the procedure, according to the guidelines, which were developed by an expert panel convened by the American Society of Clinical Oncology, American Society for Radiation Oncology, and Society for Surgical Oncology.
Nadine M. Tung, MD, of Beth Israel Deaconess Medical Center in Boston and the rest of the expert panel reviewed evidence from 58 published articles to create the guidelines.
In addition to supporting use of breast-conserving therapy, the guidelines suggest that radiation shouldn’t be withheld because of mutation status, except in patients with TP53 mutations. Furthermore, BRCA1/2 mutation carriers with metastatic HER2-negative disease can receive the poly (ADP-ribose) polymerase (PARP) inhibitors olaparib and talazoparib as an “alternative to chemotherapy” for first-, second-, or third-line therapy.
However, it’s the “license to consider breast-conserving therapy” for high-risk individuals that is one of the most noteworthy points in the guidelines, and the one that may surprise some readers, according to William J. Gradishar, MD, of Northwestern University in Chicago, who was not involved in developing the guidelines.
“We don’t have to be as dogmatic with these patients with respect to local therapies as we were in the past,” Dr. Gradishar said in an interview. “That’s a good thing for patients, but you also have to understand the nuances that go into recommending [breast-conserving surgery] to a patient. Other variables, like the age at which the patient develops breast cancer, family history, etc., all go into it.”
Weighing options for surgery
The guidelines emphasize that, for patients with germline BRCA1/2 mutations, health care providers need to discuss treatment options for the breast cancer at hand. However, patients should also be made aware of their increased risk of contralateral and new ipsilateral breast cancer as compared with noncarriers.
When weighing breast-conserving therapy versus mastectomy in light of contralateral breast cancer risk, the guidelines recommend considering not only age at diagnosis – the strongest predictor of a later contralateral breast cancer – but also family history, comorbidities, life expectancy, ability to undergo MRI, and prognosis from breast or other cancers, such as ovarian cancer.
If a bilateral mastectomy isn’t performed in a BRCA1/2 mutation carrier, an annual mammogram and MRI are warranted thereafter for screening of the remaining breast tissue, according to the guidelines.
The guidelines say breast-conserving therapy should be offered to patients with mutations in moderate-penetrance genes, including PALB2, CHEK2, and ATM. However, there’s not much data regarding the risk of ipsilateral breast cancer after breast-conserving therapy in these patients.
Likewise, there’s limited evidence on contralateral breast cancer risk for patients with mutations in moderate-penetrance genes aside from CHEK2. The guidelines say the risk should be discussed with patients “in the context of shared decision making.”
Nipple-sparing mastectomy is “reasonable” to consider in certain newly diagnosed patients with BRCA1/2 mutations, as well as in newly diagnosed patients with moderate-risk mutations, the guidelines state.
Women with breast cancer and a deleterious BRCA1/2 mutation who are undergoing unilateral mastectomy should be offered contralateral risk-reducing mastectomy. Likewise, women with moderate-risk mutations should be offered contralateral risk-reducing mastectomy, but not solely based on mutation status, according to the guidelines. Data are limited on contralateral breast cancer risk related to those mutations.
Considerations for radiation
Radiation therapy in the context of breast-conserving therapy or mastectomy should not be withheld because of hereditary mutations, except in the case of TP53 mutations, according to the guidelines.
There’s no evidence that radiotherapy increases toxicity or contralateral breast cancer risk for most BRCA1/2 or moderate-penetrance gene mutations. However, the intact breast shouldn’t be irradiated in germline TP53 mutation carriers, the guidelines say, because of the important role that TP53 plays in the ability to repair DNA damage after cellular stress.
“Carriers of a TP53 mutation would be expected to be unable to repair tissue damage from DNA damaging radiotherapy and be at risk for significant [radiotherapy]-associated sequelae,” the guidelines state.
Chemotherapy and PARP inhibitors
For women with metastatic breast cancer harboring germline BRCA1/2 mutations, the guidelines say platinum chemotherapy should be preferred over taxanes for platinum-naive patients.
Provided the breast cancer is HER2 negative, the PARP inhibitors olaparib or talazoparib “should be offered as an alternative to chemotherapy in the first- to third-line settings,” the guidelines state.
The guidelines confirm that PARP inhibitors are a “valid starting point” for treatment of BCRA1/2–associated metastatic breast cancer, Dr. Gradishar said.
“When a patient progresses on a PARP inhibitor, assuming they’re not going on some other investigational drug or clinical trial, they’re going to get chemotherapy,” he said. “So the argument is that, if you have something that’s at least as good or maybe a little bit better and has fewer side effects, why not start with that and then move on to other things?”
By contrast, there’s not enough evidence to recommend PARP inhibitors for germline BRCA mutation carriers with nonmetastatic breast cancers, according to the guidelines, and there’s “no robust data” for using PARP inhibitors in patients with breast cancers with mutations in moderate-penetrance genes.
The guideline authors disclosed relationships with AstraZeneca, Myriad Genetics, Pfizer, Lilly, and other companies. Dr. Gradishar has relationships with AstraZeneca, Celltrion, Genentech, MacroGenics, Merck, Pfizer, and Seattle Genetics.
SOURCE: Tung NM et al. J Clin Oncol. 2020 Apr 3;JCO2000299. doi: 10.1200/JCO.20.00299.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Oncologists need to advocate for scarce COVID-19 resources: ASCO
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
both at the institutional and regional level, according to new recommendations from the American Society for Clinical Oncology (ASCO).
“There was a lot of concern from the oncology community that if a patient had cancer, they would be arbitrarily excluded from consideration for critical care resources,” said Jonathan M. Marron, MD, chair-elect of ASCO’s Ethics Committee and lead author of the recommendations.
“The hope is that we’ll never have to make any of these decisions ... but the primary reason for putting together these recommendations was that if such decisions have to be made, we hope to inform them,” he told Medscape Medical News.
Marron, who is a pediatric hematologist at Boston Children’s Hospital, says ASCO’s main recommendation is that decisions about the allocation of resources must be separated from bedside clinical care, meaning that clinicians who are caring for individual patients should not also be the ones making the allocation decisions.
“Those dueling responsibilities are a conflict of interest and make that physician unable to make an unbiased decision,” he said.
“It’s also just an unbearable burden to try and do those two things simultaneously,” he added. “It’s an incredible burden to do them individually, but it’s multifold worse to try to do them both simultaneously.”
He said the vital role of oncologists who provide treatment is to offer the kind of personalized information that triage committees need in order to make appropriate decisions.
“They should be asked – maybe even must be asked – to provide the most high-quality evidence-based data about their patients’ diagnosis and prognosis,” Marron commented. “Because oncology is evolving so rapidly, and cancer is so many different diseases, it’s impossible for someone making these decisions to know everything they would need to know about why this patient is likely to survive their cancer and this patient is not.”
He says that during the COVID-19 pandemic, concerns regarding public health transcend the well-being of individual patients and that consideration must be given to providing the maximum benefit to the greatest number of people.
“That makes perfect sense and is the appropriate and laudable goal during a public health emergency like this ... but one of the challenges is that there is this belief that a diagnosis of cancer is uniformly fatal,” Marron said.
“It’s certainly conceivable that it would be a better use of resources to give the last ventilator to a young, otherwise healthy patient rather than a patient with multiply recurrent progressive metastatic cancer,” he continued. “However, we want to ensure that there is at least a discussion where that information is made available, rather than just saying, ‘She’s got cancer. She’s a lost cause.’ ”
Cancer patients are doing very well
Concerns about cancer misconceptions have been circulating in the oncology community since the start of the pandemic. “It’s really important that people understand that cancer patients are doing very well nowadays, and even with a diagnosis of cancer, they can potentially live for many years,” Anne Chiang, MD, PhD, from the Smilow Cancer Network, New Haven, Connecticut, told Medscape Medical News in a recent interview.
Thus far, even in hard-hit New York City, fears that cancer patients may not be receiving appropriate care have not materialized, according to Mark Robson, MD, a medical oncologist at Memorial Sloan Kettering Cancer Center (MSKCC). “I would emphasize that cancer patients are ABSOLUTELY getting the care they need, including patients with metastatic disease,” he recently tweeted. “NOONE at @sloan_kettering (or anywhere else) is being ‘triaged’ because of advanced cancer. Period.”
Robson told Medscape Medical News that although MSKCC continues to provide oncology care to patients with cancer, “we are [also] treating them if they develop COVID. ... I am trying to help pivot the institution towards care in this setting.”
He said he agrees with Craig Spencer, MD, MPH, director of global health in emergency medicine at the New York–Presbyterian/Columbia University Medical Center, who recently tweeted, “If you need a ventilator, you get a ventilator. Let’s be clear – this isn’t being ‘rationed.’ ”
Marron emphasized that an important safeguard against uninformed decision making is appropriate planning. For hospitalized patients, this means oncologists who provide treatment should offer information even before it is requested. But he said the “duty to plan” begins long before that.
“Clinicians haven’t always been great at talking about death and long-term outcomes with their patients, but this really cranks up the importance of having those conversations, and having them early, even though it’s incredibly hard. If someone has expressed that they would never want to be put on a ventilator, it’s important now even more so that is made clear,” he said.
He said early responses to the ASCO statement suggest that it has calmed some concerns in the oncology community, “but it still remains to be seen whether individual institutions will take this to heart, because this unto itself cannot enforce anything – it is up to individual institutions. I am hopeful this will get to the people it needs to get to.”
This article first appeared on Medscape.com.
ASCO announces its own COVID-19 and cancer registry
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
Data will not be commercialized, unlike CancerLinQ
Data will not be commercialized, unlike CancerLinQ
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
The American Society of Clinical Oncology (ASCO) has launched a registry to collect data on cancer patients with COVID-19 and is asking oncology practices across the United States to share information about their patients with the infection for educational purposes.
The new registry joins at least two other cancer and COVID-19 patient registries already underway in the U.S.
In a statement, ASCO President Howard “Skip” Burris III, MD said there is a need to know “how the virus is impacting our patients, their cancer treatment, and outcomes to inform current cancer care” and future care.
The web-based registry, known as the American Society of Clinical Oncology (ASCO) Survey on COVID-19 in Oncology Registry, is open to all U.S. oncology practices. Participating practices will receive an unspecified “nominal” payment for their data entry efforts.
The registry patient information will be stored on ASCO’s “Big Data” platform, known as CancerLinQ, but is being held apart from that pool of data. The registry information will not be available for commercial purposes, ASCO spokesperson Rachel Martin recently told Medscape Medical News.
Separately, CancerLinQ, which is a wholly owned subsidiary of ASCO, will continue to collect data from its participant oncology practices (as usual), including COVID-19 information.
CancerLinQ has been criticized by ethicists for allowing partner companies to sell access to its data (after stripping off patient identifiers), but without asking for patients’ permission, as reported last year by Medscape Medical News.
Eleven practices, including academic enterprises, have so far expressed interested in participating in the ASCO COVID-19 Registry.
Participating practices are requested to send in details about cancer patients with a confirmed COVID-19 diagnosis. As well as a baseline data capture form, they will need to provide details of subsequent status, treatment, and outcomes. Some patient-identifying data, including zip code, date of birth, gender, race, ethnicity, type of cancer, and comorbidities, will be collected for the purposes of analysis.
ASCO hopes to learn about characteristics of patients with cancer most impacted by COVID-19; estimates of disease severity; treatment modifications or delays; implementation of telemedicine in the cancer treatment setting; and clinical outcomes related to both COVID-19 and cancer.
ASCO says it will deliver periodic reports to the cancer community and the broader public on these and other “key learnings.” It also says that the registry is designed to capture point-in-time data as well as longitudinal data on how the virus will impact care and outcomes into 2021.
ASCO is not alone in its data collection efforts.
The COVID-19 and Cancer Consortium is already collecting information from more than 50 cancer centers and organizations on COVID-19 in patients with cancer. The American Society of Hematology (ASH) Research Collaborative COVID-19 Registry for Hematologic Malignancy is doing the same but with a focus on hematologic malignancies.
This article first appeared on Medscape.com.
Cancer prevalence among COVID-19 patients may be higher than previously reported
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
An early report pegged the prevalence of cancer among COVID-19 patients at 1%, but authors of a recent meta-analysis found an overall prevalence of 2% and up to 3% depending on the subset of data they reviewed.
However, those findings are limited by the retrospective nature of the studies published to date, according to the authors of the meta-analysis, led by Aakash Desai, MBBS, of the University of Connecticut, Farmington.
Nevertheless, the results do confirm that cancer patients and survivors are an important at-risk population for COVID-19, according to Dr. Desai and colleagues.
“We hope that additional data from China and Italy will provide information on the characteristics of patients with cancer at risk, types of cancer that confer higher risk, and systemic regimens that may increase COVID-19 infection complications,” the authors wrote in JCO Global Oncology.
More than 15 million individuals with cancer and many more cancer survivors are at increased risk of COVID-19 because of compromised immune systems, according to the authors.
Exactly how many individuals with cancer are among the COVID-19 cases remains unclear, though a previous report suggested the prevalence of cancer was 1% (95% confidence interval, 0.61%-1.65%) among COVID-19 patients in China (Lancet Oncol. 2020 Mar;21[3]:335-7). This “seems to be higher” than the 0.29% prevalence of cancer in the overall Chinese population, the investigators noted at the time.
That study revealed 18 cancer patients among 1,590 COVID-19 cases, though it was “hypothesis generating,” according to Dr. Desai and colleagues, who rolled that data into their meta-analysis of 11 reports including 3,661 COVID-19 cases.
Overall, Dr. Desai and colleagues found the pooled prevalence of cancer was 2.0% (95% CI, 2.0%-3.0%) in that population. In a subgroup analysis of five studies with sample sizes of less than 100 COVID-19 patients, the researchers found a “slightly higher” prevalence of 3.0% (95% CI, 1.0%-6.0%).
However, even that data wasn’t robust enough for Dr. Desai and colleagues to make any pronouncements on cancer prevalence. “Overall, current evidence on the association between cancer and COVID-19 remains inconclusive,” they wrote.
Though inconclusive, the findings raise questions about whether treatments or interventions might need to be postponed in certain patients, whether cancer patients and survivors need stronger personal protection, and how to deal with potential delays in cancer clinical trials, according to Dr. Desai and colleagues.
“As the evidence continues to rise, we must strive to answer the unanswered clinical questions,” the authors wrote.
Dr. Desai and colleagues reported no potential conflicts of interest related to the study.
SOURCE: Desai A et al. JCO Glob Oncol. 2020 Apr 6. doi: 10.1200/GO.20.00097.
FROM JCO GLOBAL ONCOLOGY
When to treat, delay, or omit breast cancer therapy in the face of COVID-19
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Nothing is business as usual during the COVID-19 pandemic, and that includes breast cancer therapy. That’s why two groups have released guidance documents on treating breast cancer patients during the pandemic.
A guidance on surgery, drug therapy, and radiotherapy was created by the COVID-19 Pandemic Breast Cancer Consortium. This guidance is set to be published in Breast Cancer Research and Treatment and can be downloaded from the American College of Surgeons website.
A group from Memorial Sloan Kettering Cancer Center (MSKCC) created a guidance document on radiotherapy for breast cancer patients, and that guidance was recently published in Advances in Radiation Oncology.
Prioritizing certain patients and treatments
As hospital beds and clinics fill with coronavirus-infected patients, oncologists must balance the need for timely therapy for their patients with the imperative to protect vulnerable, immunosuppressed patients from exposure and keep clinical resources as free as possible.
“As we’re taking care of breast cancer patients during this unprecedented pandemic, what we’re all trying to do is balance the most effective treatments for our patients against the risk of additional exposures, either from other patients [or] from being outside, and considerations about the safety of our staff,” said Steven Isakoff, MD, PhD, of Massachusetts General Hospital Cancer Center in Boston, who is an author of the COVID-19 Pandemic Breast Cancer Consortium guidance.
The consortium’s guidance recommends prioritizing treatment according to patient needs and the disease type and stage. The three basic categories for considering when to treat are:
- Priority A: Patients who have immediately life-threatening conditions, are clinically unstable, or would experience a significant change in prognosis with even a short delay in treatment.
- Priority B: Deferring treatment for a short time (6-12 weeks) would not impact overall outcomes in these patients.
- Priority C: These patients are stable enough that treatment can be delayed for the duration of the COVID-19 pandemic.
“The consortium highly recommends multidisciplinary discussion regarding priority for elective surgery and adjuvant treatments for your breast cancer patients,” the guidance authors wrote. “The COVID-19 pandemic may vary in severity over time, and these recommendations are subject to change with changing COVID-19 pandemic severity.”
For example, depending on local circumstances, the guidance recommends limiting immediate outpatient visits to patients with potentially unstable conditions such as infection or hematoma. Established patients with new problems or patients with a new diagnosis of noninvasive cancer might be managed with telemedicine visits, and patients who are on follow-up with no new issues or who have benign lesions might have their visits safely postponed.
Surgery and drug recommendations
High-priority surgical procedures include operative drainage of a breast abscess in a septic patient and evacuation of expanding hematoma in a hemodynamically unstable patient, according to the consortium guidance.
Other surgical situations are more nuanced. For example, for patients with triple-negative breast cancer (TNBC) or HER2-positive disease, the guidance recommends neoadjuvant chemotherapy or HER2-targeted chemotherapy in some cases. In other cases, institutions may proceed with surgery before chemotherapy, but “these decisions will depend on institutional resources and patient factors,” according to the authors.
The guidance states that chemotherapy and other drug treatments should not be delayed in patients with oncologic emergencies, such as febrile neutropenia, hypercalcemia, intolerable pain, symptomatic pleural effusions, or brain metastases.
In addition, patients with inflammatory breast cancer, TNBC, or HER2-positive breast cancer should receive neoadjuvant/adjuvant chemotherapy. Patients with metastatic disease that is likely to benefit from therapy should start chemotherapy, endocrine therapy, or targeted therapy. And patients who have already started neoadjuvant/adjuvant chemotherapy or oral adjuvant endocrine therapy should continue on these treatments.
Radiation therapy recommendations
The consortium guidance recommends administering radiation to patients with bleeding or painful inoperable locoregional disease, those with symptomatic metastatic disease, and patients who progress on neoadjuvant chemotherapy.
In contrast, older patients (aged 65-70 years) with lower-risk, stage I, hormone receptor–positive, HER2-negative cancers who are on adjuvant endocrine therapy can safely defer or omit radiation without affecting their overall survival, according to the guidance. Patients with ductal carcinoma in situ, especially those with estrogen receptor–positive disease on endocrine therapy, can safely omit radiation.
“There are clearly conditions where radiation might reduce the risk of recurrence but not improve overall survival, where a delay in treatment really will have minimal or no impact,” Dr. Isakoff said.
The MSKCC guidance recommends omitting radiation for some patients with favorable-risk disease and truncating or accelerating regimens using hypofractionation for others who require whole-breast radiation or post-mastectomy treatment.
The MSKCC guidance also contains recommendations for prioritization of patients according to disease state and the urgency of care. It divides cases into high, intermediate, and low priority for breast radiotherapy, as follows:
- Tier 1 (high priority): Patients with inflammatory breast cancer, residual node-positive disease after neoadjuvant chemotherapy, four or more positive nodes (N2), recurrent disease, node-positive TNBC, or extensive lymphovascular invasion.
- Tier 2 (intermediate priority): Patients with estrogen receptor–positive disease with one to three positive nodes (N1a), pathologic stage N0 after neoadjuvant chemotherapy, lymphovascular invasion not otherwise specified, or node-negative TNBC.
- Tier 3 (low priority): Patients with early-stage estrogen receptor-positive breast cancer (especially patients of advanced age), patients with ductal carcinoma in situ, or those who otherwise do not meet the criteria for tiers 1 or 2.
The MSKCC guidance also contains recommended hypofractionated or accelerated radiotherapy regimens for partial and whole-breast irradiation, post-mastectomy treatment, and breast and regional node irradiation, including recommended techniques (for example, 3-D conformal or intensity modulated approaches).
The authors of the MSKCC guidance disclosed relationships with eContour, Volastra Therapeutics, Sanofi, the Prostate Cancer Foundation, and Cancer Research UK. The authors of the COVID-19 Pandemic Breast Cancer Consortium guidance did not disclose any conflicts and said there was no funding source for the guidance.
SOURCES: Braunstein LZ et al. Adv Radiat Oncol. 2020 Apr 1. doi:10.1016/j.adro.2020.03.013; Dietz JR et al. 2020 Apr. Recommendations for prioritization, treatment and triage of breast cancer patients during the COVID-19 pandemic. Accepted for publication in Breast Cancer Research and Treatment.
Home-based chemo skyrockets at one U.S. center
Major organization opposes concept
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
Major organization opposes concept
Major organization opposes concept
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
In the fall of 2019, the University of Pennsylvania in Philadelphia started a pilot program of home-based chemotherapy for two treatment regimens (one via infusion and one via injection). Six months later, the Cancer Care at Home program had treated 40 patients.
The uptake within the university’s large regional health system was acceptable but not rapid, admitted Amy Laughlin, MD, a hematology-oncology fellow involved with the program.
Then COVID-19 arrived, along with related travel restrictions.
Suddenly, in a 5-week period (March to April 7), 175 patients had been treated – a 300% increase from the first half year. Program staff jumped from 12 to 80 employees. The list of chemotherapies delivered went from two to seven, with more coming.
“We’re not the pilot anymore – we’re the standard of care,” Laughlin told Medscape Medical News.
“The impact [on patients] is amazing,” she said. “As long as you are selecting the right patients and right therapy, it is feasible and even preferable for a lot of patients.”
For example, patients with hormone-positive breast cancer who receive leuprolide (to shut down the ovaries and suppress estrogen production) ordinarily would have to visit a Penn facility for an injection every month, potentially for years. Now, a nurse can meet patients at home (or before the COVID-19 pandemic, even at their place of work) and administer the injection, saving the patient travel time and associated costs.
This home-based chemotherapy service does not appear to be offered elsewhere in the United States, and a major oncology organization – the Community Oncology Alliance – is opposed to the practice because of patient safety concerns.
The service is not offered at a sample of cancer centers queried by Medscape Medical News, including the Dana-Farber Cancer Institute in Boston, the Moffitt Cancer Center in Tampa, the Huntsman Cancer Institute in Salt Lake City, Utah, and Moores Cancer Center, the University of California, San Diego.
Opposition because of safety concerns
On April 9, the Community Oncology Alliance (COA) issued a statement saying it “fundamentally opposes home infusion of chemotherapy, cancer immunotherapy, and cancer treatment supportive drugs because of serious patient safety concerns.”
The COA warned that “many of the side effects caused by cancer treatment can have a rapid, unpredictable onset that places patients in incredible jeopardy and can even be life-threatening.”
In contrast, in a recent communication related to COVID-19, the National Comprehensive Cancer Network tacitly endorsed the concept, stating that a number of chemotherapies may potentially be administered at home, but it did not include guidelines for doing so.
The American Society of Clinical Oncology said that chemotherapy at home is “an issue [we] are monitoring closely,” according to a spokesperson.
What’s involved
Criteria for home-based chemotherapy at Penn include use of anticancer therapies that a patient has previously tolerated and low toxicity (that can be readily managed in the home setting). In addition, patients must be capable of following a med chart.
The chemotherapy is reconstituted at a Penn facility in a Philadelphia suburb. A courier then delivers the drug to the patient’s home, where it is administered by an oncology-trained nurse. Drugs must be stable for at least a few hours to qualify for the program.
The Penn program started with two regimens: EPOCH (etoposide, vincristine, doxorubicin, cyclophosphamide, and prednisone) for lymphoma, and leuprolide acetate injections for either breast or prostate cancer.
The two treatments are polar opposites in terms of complexity, common usage, and time required, which was intended, said Laughlin.
Time to deliver the chemo varies from a matter of minutes with leuprolide to more than 2 hours for rituximab, a lymphoma drug that may be added to EPOCH.
The current list of at-home chemo agents in the Penn program also includes bortezomib, lanreotide, zoledronic acid, and denosumab. Soon to come are rituximab and pembrolizumab for lung cancer and head and neck cancer.
Already practiced in some European countries
Home-based chemotherapy dates from at least the 1980s in the medical literature and is practiced in some European countries.
A 2018 randomized study of adjuvant treatment with capecitabine and oxaliplatin for stage II/III colon cancer in Denmark, where home-based care has been practiced for the past 2 years and is growing in use, concluded that “it might be a valuable alternative to treatment at an outpatient clinic.”
However, in the study, there was no difference in quality of life between the home and outpatient settings, which is somewhat surprising, inasmuch as a major appeal to receiving chemotherapy at home is that it is less disruptive compared to receiving it in a hospital or clinic, which requires travel.
Also, chemo at home “may be resource intensive” and have a “lower throughput of patients due to transportation time,” cautioned the Danish investigators, who were from Herlev and Gentofte Hospital.
A 2015 review called home chemo “a safe and patient‐centered alternative to hospital‐ and outpatient‐based service.” Jenna Evans, PhD, McMaster University, Toronto, Canada, and lead author of that review, says there are two major barriers to infusion chemotherapy in homes.
One is inadequate resources in the community, such as oncology-trained nurses to deliver treatment, and the other is perceptions of safety and quality, including among healthcare providers.
COVID-19 might prompt more chemo at home, said Evans, a health policy expert, in an email to Medscape Medical News. “It is not unusual for change of this type and scale to require a seismic event to become more mainstream,” she argued.
Reimbursement for home-based chemo is usually the same as for chemo in a free-standing infusion suite, says Cassandra Redmond, PharmD, MBA, director of pharmacy, Penn Home Infusion Therapy.
Private insurers and Medicare cover a subset of infused medications at home, but coverage is limited. “The opportunity now is to expand these initiatives ... to include other cancer therapies,” she said about coverage.
This article first appeared on Medscape.com.
Conducting cancer trials amid the COVID-19 pandemic
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
More than three-quarters of cancer clinical research programs have experienced operational changes during the COVID-19 pandemic, according to a survey conducted by the Association of Community Cancer Centers (ACCC) during a recent webinar.
The webinar included insights into how some cancer research programs have adapted to the pandemic, a review of guidance for conducting cancer trials during this time, and a discussion of how the cancer research landscape may be affected by COVID-19 going forward.
The webinar was led by Randall A. Oyer, MD, president of the ACCC and medical director of the oncology program at Penn Medicine Lancaster General Health in Pennsylvania.
The impact of COVID-19 on cancer research
Dr. Oyer observed that planning and implementation for COVID-19–related illness at U.S. health care institutions has had a predictable effect of limiting patient access and staff availability for nonessential services.
Coronavirus-related exposure and/or illness has relegated cancer research to a lower-level priority. As a result, ACCC institutions have made adjustments in their cancer research programs, including moving clinical research coordinators off-campus and deploying them in clinical areas.
New clinical trials have not been opened. In some cases, new accruals have been halted, particularly for registry, prevention, and symptom control trials.
Standards that have changed and those that have not
Guidance documents for conducting clinical trials during the pandemic have been developed by the Food and Drug Administration, the National Cancer Institute’s Cancer Therapy Evaluation Program and Central Institutional Review Board, and the National Institutes of Health’s Office of Extramural Research. Industry sponsors and parent institutions of research programs have also disseminated guidance.
Among other topics, guidance documents have addressed:
- How COVID-19-related protocol deviations will be judged at monitoring visits and audits
- Missed office visits and endpoint evaluations
- Providing investigational oral medications to patients via mail and potential issues of medication unavailability
- Processes for patients to have interim visits with providers at external institutions, including providers who may not be personally engaged in or credentialed for the research trial
- Potential delays in submitting protocol amendments for institutional review board (IRB) review
- Recommendations for patients confirmed or suspected of having a coronavirus infection.
Dr. Oyer emphasized that patient safety must remain the highest priority for patient management, on or off study. He advised continuing investigational therapy when potential benefit from treatment is anticipated and identifying alternative methods to face-to-face visits for monitoring and access to treatment.
Dr. Oyer urged programs to:
- Maintain good clinical practice standards
- Consult with sponsors and IRBs when questions arise but implement changes that affect patient safety prior to IRB review if necessary
- Document all deviations and COVID-19 related adaptations in a log or spreadsheet in anticipation of future questions from sponsors, monitors, and other entities.
New questions and considerations
In the short-term, Dr. Oyer predicts fewer available trials and a decreased rate of accrual to existing studies. This may result in delays in trial completion and the possibility of redesign for some trials.
He predicts the emergence of COVID-19-focused research questions, including those assessing the course of coronavirus infection in various malignant settings and the impact of cancer-directed treatments and supportive care interventions (e.g., treatment for graft-versus-host disease) on response to COVID-19.
To facilitate developing a clinically and research-relevant database, Dr. Oyer stressed the importance of documentation in the research record, reporting infections as serious adverse events. Documentation should specify whether the infection was confirmed or suspected coronavirus or related to another organism.
In general, when coronavirus infection is strongly suspected, Dr. Oyer said investigational treatments should be interrupted, but study-specific criteria will be forthcoming on that issue.
Looking to the future
For patients with advanced cancers, clinical trials provide an important option for hope and clinical benefit. Disrupting the conduct of clinical trials could endanger the lives of participants and delay the emergence of promising treatments and diagnostic tests.
When the coronavirus pandemic recedes, advancing knowledge and treatments for cancer will demand renewed commitment across the oncology care community.
Going forward, Dr. Oyer advised that clinical research staff protect their own health and the safety of trial participants. He encouraged programs to work with sponsors and IRBs to solve logistical problems and clarify individual issues.
He was optimistic that resumption of more normal conduct of studies will enable the successful completion of ongoing trials, enhanced by the creative solutions that were devised during the crisis and by additional prospective, clinically annotated, carefully recorded data from academic and community research sites.
Dr. Lyss was a community-based medical oncologist and clinical researcher for more than 35 years before his recent retirement. His clinical and research interests were focused on breast and lung cancers as well as expanding clinical trial access to medically underserved populations. He is based in St. Louis. He has no conflicts of interest.
Evaluating and managing the patient with nipple discharge
CASE Young woman with discharge from one nipple
A 26-year-old African American woman presents with a 10-month history of left nipple discharge. The patient describes the discharge as spontaneous, colored dark brown to yellow, and occurring from a single opening in the nipple. The discharge is associated with left breast pain and fullness, without a palpable lump. The patient has no family or personal history of breast cancer.
Nipple discharge is the third most common breast-related symptom (after palpable masses and breast pain), with an estimated prevalence of 5% to 8% among premenopausal women.1 While most causes of nipple discharge reflect benign issues, approximately 5% to 12% of breast cancers have nipple discharge as the only symptom.2 Not surprisingly, nipple discharge creates anxiety for both patients and clinicians.
In this article, we—a breast imaging radiologist, gynecologist, and breast surgeon—outline key steps for evaluating and managing patients with nipple discharge.
Two types of nipple discharge
Nipple discharge can be characterized as physiologic or pathologic. The distinction is based on the patient’s history in conjunction with the clinical breast exam.
Physiologic nipple discharge often is bilateral, nonspontaneous, and white, yellow, green, or brown (TABLE).3 It often is due to nipple stimulation, and the patient can elicit discharge by manually manipulating the breast. Usually, multiple ducts are involved. Galactorrhea refers specifically to milky discharge and occurs most commonly during pregnancy or lactation.2 Galactorrhea that is not associated with pregnancy or lactation often is related to elevated prolactin or thyroid-stimulating hormone levels or to medications. One study reported that no cancers were found when discharge was nonspontaneous and colored or milky.4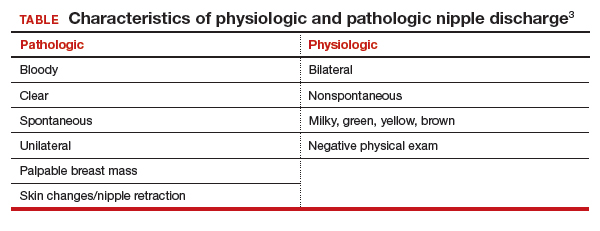
Pathologic nipple discharge is defined as a spontaneous, bloody, clear, or single-duct discharge. A palpable mass in the same breast automatically increases the suspicion of the discharge, regardless of its color or spontaneity.2 The most common cause of pathologic nipple discharge is an intraductal papilloma, a benign epithelial tumor, which accounts for approximately 57% of cases.5
Although the risk of malignancy is low for all patients with nipple discharge, increasing age is associated with increased risk of breast cancer. One study demonstrated that among women aged 40 to 60 years presenting with nipple discharge, the prevalence of invasive cancer is 10%, and the percentage jumps to 32% among women older than 60.6
Breast exam. For any patient with nonlactational nipple discharge, we recommend a thorough breast examination. Deep palpation of all quadrants of the symptomatic breast, especially near the nipple areolar complex, should elicit nipple discharge without any direct squeezing of the nipple. If the patient’s history and physical exam are consistent with physiologic discharge, no further workup is needed. Reassure the patient and recommend appropriate breast cancer screening. Encourage the patient to decrease stimulation or manual manipulation of the nipples if the discharge bothers her.
Continue to: CASE Continued: Workup...
CASE Continued: Workup
On physical exam, the patient’s breasts are noted to be cup size DDD and asymmetric, with the left breast larger than the right; there is no contour deformity. There is no skin or nipple retraction, skin rash, swelling, or nipple changes bilaterally. No dominant masses are appreciated bilaterally. Manual compression elicits no nipple discharge.
Although the discharge is nonbloody, its spontaneity, unilaterality, and single-duct/orifice origin suggest a pathologic cause. The patient is referred for breast imaging.
Imaging workup for pathologic discharge
The American College of Radiology (ACR) Appropriateness Criteria is a useful tool that provides an evidence-based, easy-to-use algorithm for breast imaging in the patient with pathologic nipple discharge (FIGURE 1).6 The algorithm is categorized by patient age, with diagnostic mammography recommended for women aged 30 and older.6 Diagnostic mammography is recommended if the patient has not had a mammogram study in the last 6 months.6 For patients with no prior mammograms, we recommend bilateral diagnostic mammography to compare symmetry of the breasts.
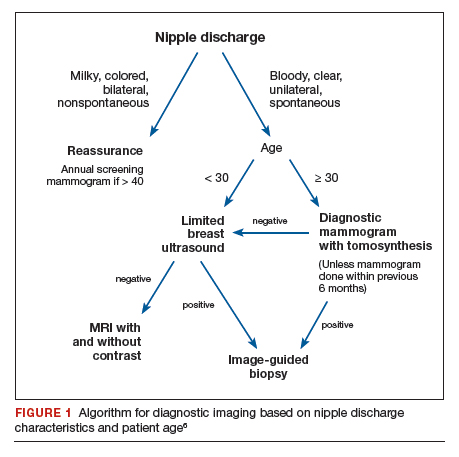
Currently, no studies show that digital breast tomosynthesis (3-D mammography) has a benefit compared with standard 2-D mammography in women with pathologic nipple discharge.6 Given the increased sensitivity of digital breast tomosynthesis for cancer detection, however, in our practice it is standard to use tomosynthesis in the diagnostic evaluation of most patients.
Mammography
On mammography, ductal carcinoma in situ (DCIS) usually presents as calcifications. Both the morphology and distribution of calcifications are used to characterize them as suspicious or, typically, benign. DCIS usually presents as fine pleomorphic or fine linear branching calcifications in a segmental or linear distribution. In patients with pathologic nipple discharge and no other symptoms, the radiologist must closely examine the retroareolar region of the breast to assess for faint calcifications. Magnification views also can be performed to better characterize calcifications.
The sensitivity of mammography for nipple discharge varies in the literature, ranging from approximately 15% to 68%, with a specificity range of 38% to 98%.6 This results in a relatively low positive predictive value but a high negative predictive value of 90%.7 Mammographic sensitivity largely is limited by increased breast density. As more data emerge on the utility of digital breast tomosynthesis in dense breasts, mammographic sensitivity for nipple discharge will likely increase.
Ultrasonography
As an adjunct to mammography, the ACR Appropriateness Criteria recommends targeted (or “limited”) ultrasonography of the retroareolar region of the affected breast for patients aged 30 and older. Ultrasonography is useful to assess for intraductal masses and architectural distortion, and it has higher sensitivity but lower specificity than mammography. The sensitivity of ultrasonography for detecting breast cancer in patients presenting with nipple discharge is reported to be 56% to 80%.6 Ultrasonography can identify lesions not visible mammographically in 63% to 69% of cases.8 Although DCIS usually presents as calcifications, it also can present as an intraductal mass on ultrasonography.
The ACR recommends targeted ultrasonography for patients with nipple discharge and a negative mammogram, or to evaluate a suspicious mammographic abnormality such as architectural distortion, focal asymmetry, or a mass.6 For patient comfort, ultrasonography is the preferred modality for image-guided biopsy.
For women younger than 30 years, targeted ultrasonography is the initial imaging study of choice, according to the ACR criteria.6 Women younger than 30 years with pathologic nipple discharge have a very low risk of breast cancer and tend to have higher breast density, making mammography less useful. Although the radiation dose from mammography is negligible given technological improvements and dose-reduction techniques, ultrasonography remains the preferred initial imaging modality in young women, not only for nipple discharge but also for palpable lumps and focal breast pain.
Mammography is used as an adjunct to ultrasonography in women younger than 30 years when a suspicious abnormality is detected on ultrasonography, such as an intraductal mass or architectural distortion. In these cases, mammography can be used to assess for extent of disease or to visualize suspicious calcifications not well seen on ultrasonography.
For practical purposes regarding which imaging study to order for a patient, it is most efficient to order both a diagnostic mammogram (with tomosynthesis, if possible) and a targeted ultrasound scan of the affected breast. Even if both orders are not needed, having them available increases efficiency for both the radiologist and the ordering physician.
Continue to: CASE Continued: Imaging findings...
CASE Continued: Imaging findings
Given her age, the patient initially undergoes targeted ultrasonography. The grayscale image (FIGURE 2) demonstrates multiple mildly dilated ducts (white arrows) with surrounding hyperechogenicity of the fat (red arrows), indicating soft tissue edema. No intraductal mass is imaged. Given that the ultrasonography findings are not completely negative and are equivocal for malignancy, bilateral diagnostic mammography (FIGURE 3, left breast only) is performed. Standard full-field craniocaudal (FIGURE 3A) and mediolateral oblique (FIGURE 3B) mammographic views demonstrate a heterogeneously dense breast with a few calcifications in the retroareolar left breast (red ovals). No associated mass or architectural distortions are noted. The mammographic and sonographic findings do not reveal a definitive biopsy target.

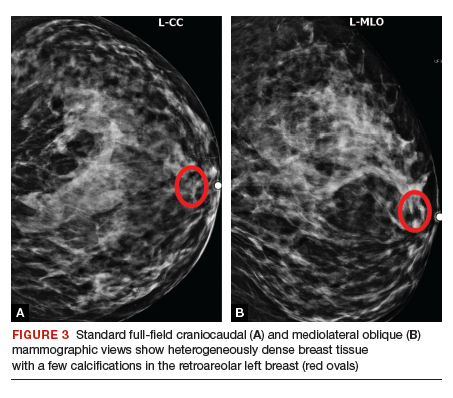
Ductography
When a suspicious abnormality is visualized on either mammography or ultrasonography, the standard of care is to perform an image-guided biopsy of the abnormality. When the standard workup is negative or equivocal, the standard of care historically was to perform ductography.
Ductography is an invasive procedure that involves cannulating the suspicious duct with a small catheter and injecting radiopaque dye into the duct under mammographic guidance. While the sensitivity of ductography is higher than that of both mammography and ultrasonography, its specificity is lower than that of either modality.
Most cases of pathologic discharge are spontaneous and are not reproducible on the day of the procedure. If the procedural radiologist cannot visualize the duct that is producing the discharge, the procedure cannot be performed. Although most patients tolerate the procedure well, ductography produces patient discomfort from cannulation of the duct and injection of contrast.
Magnetic resonance imaging
Dynamic contrast-enhanced magnetic resonance imaging (MRI) is the most sensitive imaging study for evaluating pathologic nipple discharge, and it has largely replaced ductography as an adjunct to mammography and ultrasonography. MRI’s sensitivity for detecting breast cancer ranges from 93% to 100%.6 In addition, MRI allows visualization of the entire breast and areas peripheral to the field of view of a standard ductogram or ultrasound scan.9
Clinicians commonly ask, “Why not skip the mammogram and ultrasound scan and go straight to MRI, since it is so much more sensitive?” Breast MRI has several limitations, including relatively low specificity, cost, use of intravenous contrast, and patient discomfort (that is, claustrophobia, prone positioning). MRI should be utilized for pathologic discharge only when the mammogram and/or targeted ultrasound scans are negative or equivocal.
CASE Continued: Additional imaging
A contrast-enhanced MRI of the breasts (FIGURE 4) demonstrates a large area of non-mass enhancement (red oval) in the left breast, which involves most of the upper breast extending from the nipple to the posterior breast tissue; it measures approximately 7.3 x 14 x 9.1 cm in transverse, anteroposterior, and craniocaudal dimensions, respectively. There is no evidence of left pectoralis muscle involvement. An MRI-directed second look left breast ultrasonography (FIGURE 5) is performed, revealing a small irregular mass in the left breast 1 o’clock position, 10 to 11 cm from the nipple (red arrow). This area had not been imaged in the prior ultrasound scan due to its posterior location far from the nipple. Ultrasound-guided core needle biopsy is performed; moderately differentiated invasive ductal carcinoma (IDC) with high-grade DCIS is found.

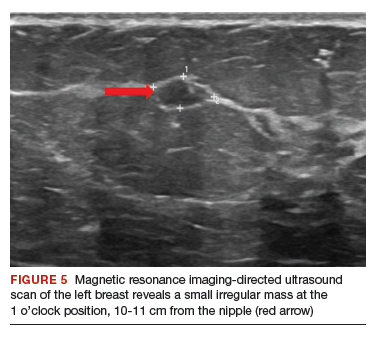
Continue to: When to refer for surgery...
When to refer for surgery
No surgical evaluation or intervention is needed for physiologic nipple discharge. As mentioned previously, reassure the patient and recommend appropriate breast cancer screening. In the setting of pathologic discharge, however, referral to a breast surgeon may be indicated after appropriate imaging workup has been done.
Since abnormal imaging almost always results in a recommendation for image-guided biopsy, typically the biopsy is performed prior to the surgical consultation. Once the pathology report from the biopsy is available, the radiologist makes a radiologic-pathologic concordance statement and recommends surgical consultation. This process allows the surgeon to have all the necessary information at the initial visit.
However, in the setting of pathologic nipple discharge with normal breast imaging, the surgeon and patient may opt for close observation or surgery for definitive diagnosis. Surgical options include single-duct excision when nipple discharge is localized to one duct or central duct excision when nipple discharge cannot be localized to one duct.
CASE Continued: Follow-up
The patient was referred to a breast surgeon. Given the extent of disease in the left breast, breast conservation was not possible. The patient underwent left breast simple mastectomy with sentinel lymph node biopsy and prophylactic right simple mastectomy. Final pathology results revealed stage IA IDC with DCIS. Sentinel lymph nodes were negative for malignancy. The patient underwent adjuvant left chest wall radiation, endocrine therapy with tamoxifen, and implant reconstruction. After 2 years of follow-up, she is disease free.
In summary
Nipple discharge can be classified as physiologic or pathologic. For pathologic discharge, a thorough physical examination should be performed with subsequent imaging evaluation. First-line tools, based on patient age, include diagnostic mammography and targeted ultrasonography. Contrast-enhanced MRI is then recommended for negative or equivocal cases. All patients with pathologic nipple discharge should be referred to a breast surgeon following appropriate imaging evaluation. ●
- Alcock C, Layer GT. Predicting occult malignancy in nipple discharge. ANZ J Surg. 2010;80:646-649.
- Patel BK, Falcon S, Drukteinis J. Management of nipple discharge and the associated imaging findings. Am J Med. 2015;128:353-360.
- Mazzarello S, Arnaout A. Five things to know about nipple discharge. CMAJ. 2015;187:599.
- Goksel HA, Yagmurdur MC, Demirhan B, et al. Management strategies for patients with nipple discharge. Langenbecks Arch Surg. 2005;390:52-58.
- Vargas HI, Vargas MP, Eldrageely K, et al. Outcomes of clinical and surgical assessment of women with pathological nipple discharge. Am Surg. 2006;72:124-128.
- Expert Panel on Breast Imaging; Lee S, Tikha S, Moy L, et al. American College of Radiology Appropriateness Criteria: Evaluation of nipple discharge. https://acsearch.acr.org /docs/3099312/Narrative/. Accessed February 2, 2020.
- Cabioglu N, Hunt KK, Singletary SE, et al. Surgical decision making and factors determining a diagnosis of breast carcinoma in women presenting with nipple discharge. J Am Coll Surg. 2003;196:354-364.
- Morrogh M, Park A, Elkin EB, et al. Lessons learned from 416 cases of nipple discharge of the breast. Am J Surg. 2010;200:73-80.
- Morrogh M, Morris EA, Liberman L, et al. The predictive value of ductography and magnetic resonance imaging in the management of nipple discharge. Ann Surg Oncol. 2007;14:3369-3377.
CASE Young woman with discharge from one nipple
A 26-year-old African American woman presents with a 10-month history of left nipple discharge. The patient describes the discharge as spontaneous, colored dark brown to yellow, and occurring from a single opening in the nipple. The discharge is associated with left breast pain and fullness, without a palpable lump. The patient has no family or personal history of breast cancer.
Nipple discharge is the third most common breast-related symptom (after palpable masses and breast pain), with an estimated prevalence of 5% to 8% among premenopausal women.1 While most causes of nipple discharge reflect benign issues, approximately 5% to 12% of breast cancers have nipple discharge as the only symptom.2 Not surprisingly, nipple discharge creates anxiety for both patients and clinicians.
In this article, we—a breast imaging radiologist, gynecologist, and breast surgeon—outline key steps for evaluating and managing patients with nipple discharge.
Two types of nipple discharge
Nipple discharge can be characterized as physiologic or pathologic. The distinction is based on the patient’s history in conjunction with the clinical breast exam.
Physiologic nipple discharge often is bilateral, nonspontaneous, and white, yellow, green, or brown (TABLE).3 It often is due to nipple stimulation, and the patient can elicit discharge by manually manipulating the breast. Usually, multiple ducts are involved. Galactorrhea refers specifically to milky discharge and occurs most commonly during pregnancy or lactation.2 Galactorrhea that is not associated with pregnancy or lactation often is related to elevated prolactin or thyroid-stimulating hormone levels or to medications. One study reported that no cancers were found when discharge was nonspontaneous and colored or milky.4
Pathologic nipple discharge is defined as a spontaneous, bloody, clear, or single-duct discharge. A palpable mass in the same breast automatically increases the suspicion of the discharge, regardless of its color or spontaneity.2 The most common cause of pathologic nipple discharge is an intraductal papilloma, a benign epithelial tumor, which accounts for approximately 57% of cases.5
Although the risk of malignancy is low for all patients with nipple discharge, increasing age is associated with increased risk of breast cancer. One study demonstrated that among women aged 40 to 60 years presenting with nipple discharge, the prevalence of invasive cancer is 10%, and the percentage jumps to 32% among women older than 60.6
Breast exam. For any patient with nonlactational nipple discharge, we recommend a thorough breast examination. Deep palpation of all quadrants of the symptomatic breast, especially near the nipple areolar complex, should elicit nipple discharge without any direct squeezing of the nipple. If the patient’s history and physical exam are consistent with physiologic discharge, no further workup is needed. Reassure the patient and recommend appropriate breast cancer screening. Encourage the patient to decrease stimulation or manual manipulation of the nipples if the discharge bothers her.
Continue to: CASE Continued: Workup...
CASE Continued: Workup
On physical exam, the patient’s breasts are noted to be cup size DDD and asymmetric, with the left breast larger than the right; there is no contour deformity. There is no skin or nipple retraction, skin rash, swelling, or nipple changes bilaterally. No dominant masses are appreciated bilaterally. Manual compression elicits no nipple discharge.
Although the discharge is nonbloody, its spontaneity, unilaterality, and single-duct/orifice origin suggest a pathologic cause. The patient is referred for breast imaging.
Imaging workup for pathologic discharge
The American College of Radiology (ACR) Appropriateness Criteria is a useful tool that provides an evidence-based, easy-to-use algorithm for breast imaging in the patient with pathologic nipple discharge (FIGURE 1).6 The algorithm is categorized by patient age, with diagnostic mammography recommended for women aged 30 and older.6 Diagnostic mammography is recommended if the patient has not had a mammogram study in the last 6 months.6 For patients with no prior mammograms, we recommend bilateral diagnostic mammography to compare symmetry of the breasts.

Currently, no studies show that digital breast tomosynthesis (3-D mammography) has a benefit compared with standard 2-D mammography in women with pathologic nipple discharge.6 Given the increased sensitivity of digital breast tomosynthesis for cancer detection, however, in our practice it is standard to use tomosynthesis in the diagnostic evaluation of most patients.
Mammography
On mammography, ductal carcinoma in situ (DCIS) usually presents as calcifications. Both the morphology and distribution of calcifications are used to characterize them as suspicious or, typically, benign. DCIS usually presents as fine pleomorphic or fine linear branching calcifications in a segmental or linear distribution. In patients with pathologic nipple discharge and no other symptoms, the radiologist must closely examine the retroareolar region of the breast to assess for faint calcifications. Magnification views also can be performed to better characterize calcifications.
The sensitivity of mammography for nipple discharge varies in the literature, ranging from approximately 15% to 68%, with a specificity range of 38% to 98%.6 This results in a relatively low positive predictive value but a high negative predictive value of 90%.7 Mammographic sensitivity largely is limited by increased breast density. As more data emerge on the utility of digital breast tomosynthesis in dense breasts, mammographic sensitivity for nipple discharge will likely increase.
Ultrasonography
As an adjunct to mammography, the ACR Appropriateness Criteria recommends targeted (or “limited”) ultrasonography of the retroareolar region of the affected breast for patients aged 30 and older. Ultrasonography is useful to assess for intraductal masses and architectural distortion, and it has higher sensitivity but lower specificity than mammography. The sensitivity of ultrasonography for detecting breast cancer in patients presenting with nipple discharge is reported to be 56% to 80%.6 Ultrasonography can identify lesions not visible mammographically in 63% to 69% of cases.8 Although DCIS usually presents as calcifications, it also can present as an intraductal mass on ultrasonography.
The ACR recommends targeted ultrasonography for patients with nipple discharge and a negative mammogram, or to evaluate a suspicious mammographic abnormality such as architectural distortion, focal asymmetry, or a mass.6 For patient comfort, ultrasonography is the preferred modality for image-guided biopsy.
For women younger than 30 years, targeted ultrasonography is the initial imaging study of choice, according to the ACR criteria.6 Women younger than 30 years with pathologic nipple discharge have a very low risk of breast cancer and tend to have higher breast density, making mammography less useful. Although the radiation dose from mammography is negligible given technological improvements and dose-reduction techniques, ultrasonography remains the preferred initial imaging modality in young women, not only for nipple discharge but also for palpable lumps and focal breast pain.
Mammography is used as an adjunct to ultrasonography in women younger than 30 years when a suspicious abnormality is detected on ultrasonography, such as an intraductal mass or architectural distortion. In these cases, mammography can be used to assess for extent of disease or to visualize suspicious calcifications not well seen on ultrasonography.
For practical purposes regarding which imaging study to order for a patient, it is most efficient to order both a diagnostic mammogram (with tomosynthesis, if possible) and a targeted ultrasound scan of the affected breast. Even if both orders are not needed, having them available increases efficiency for both the radiologist and the ordering physician.
Continue to: CASE Continued: Imaging findings...
CASE Continued: Imaging findings
Given her age, the patient initially undergoes targeted ultrasonography. The grayscale image (FIGURE 2) demonstrates multiple mildly dilated ducts (white arrows) with surrounding hyperechogenicity of the fat (red arrows), indicating soft tissue edema. No intraductal mass is imaged. Given that the ultrasonography findings are not completely negative and are equivocal for malignancy, bilateral diagnostic mammography (FIGURE 3, left breast only) is performed. Standard full-field craniocaudal (FIGURE 3A) and mediolateral oblique (FIGURE 3B) mammographic views demonstrate a heterogeneously dense breast with a few calcifications in the retroareolar left breast (red ovals). No associated mass or architectural distortions are noted. The mammographic and sonographic findings do not reveal a definitive biopsy target.


Ductography
When a suspicious abnormality is visualized on either mammography or ultrasonography, the standard of care is to perform an image-guided biopsy of the abnormality. When the standard workup is negative or equivocal, the standard of care historically was to perform ductography.
Ductography is an invasive procedure that involves cannulating the suspicious duct with a small catheter and injecting radiopaque dye into the duct under mammographic guidance. While the sensitivity of ductography is higher than that of both mammography and ultrasonography, its specificity is lower than that of either modality.
Most cases of pathologic discharge are spontaneous and are not reproducible on the day of the procedure. If the procedural radiologist cannot visualize the duct that is producing the discharge, the procedure cannot be performed. Although most patients tolerate the procedure well, ductography produces patient discomfort from cannulation of the duct and injection of contrast.
Magnetic resonance imaging
Dynamic contrast-enhanced magnetic resonance imaging (MRI) is the most sensitive imaging study for evaluating pathologic nipple discharge, and it has largely replaced ductography as an adjunct to mammography and ultrasonography. MRI’s sensitivity for detecting breast cancer ranges from 93% to 100%.6 In addition, MRI allows visualization of the entire breast and areas peripheral to the field of view of a standard ductogram or ultrasound scan.9
Clinicians commonly ask, “Why not skip the mammogram and ultrasound scan and go straight to MRI, since it is so much more sensitive?” Breast MRI has several limitations, including relatively low specificity, cost, use of intravenous contrast, and patient discomfort (that is, claustrophobia, prone positioning). MRI should be utilized for pathologic discharge only when the mammogram and/or targeted ultrasound scans are negative or equivocal.
CASE Continued: Additional imaging
A contrast-enhanced MRI of the breasts (FIGURE 4) demonstrates a large area of non-mass enhancement (red oval) in the left breast, which involves most of the upper breast extending from the nipple to the posterior breast tissue; it measures approximately 7.3 x 14 x 9.1 cm in transverse, anteroposterior, and craniocaudal dimensions, respectively. There is no evidence of left pectoralis muscle involvement. An MRI-directed second look left breast ultrasonography (FIGURE 5) is performed, revealing a small irregular mass in the left breast 1 o’clock position, 10 to 11 cm from the nipple (red arrow). This area had not been imaged in the prior ultrasound scan due to its posterior location far from the nipple. Ultrasound-guided core needle biopsy is performed; moderately differentiated invasive ductal carcinoma (IDC) with high-grade DCIS is found.


Continue to: When to refer for surgery...
When to refer for surgery
No surgical evaluation or intervention is needed for physiologic nipple discharge. As mentioned previously, reassure the patient and recommend appropriate breast cancer screening. In the setting of pathologic discharge, however, referral to a breast surgeon may be indicated after appropriate imaging workup has been done.
Since abnormal imaging almost always results in a recommendation for image-guided biopsy, typically the biopsy is performed prior to the surgical consultation. Once the pathology report from the biopsy is available, the radiologist makes a radiologic-pathologic concordance statement and recommends surgical consultation. This process allows the surgeon to have all the necessary information at the initial visit.
However, in the setting of pathologic nipple discharge with normal breast imaging, the surgeon and patient may opt for close observation or surgery for definitive diagnosis. Surgical options include single-duct excision when nipple discharge is localized to one duct or central duct excision when nipple discharge cannot be localized to one duct.
CASE Continued: Follow-up
The patient was referred to a breast surgeon. Given the extent of disease in the left breast, breast conservation was not possible. The patient underwent left breast simple mastectomy with sentinel lymph node biopsy and prophylactic right simple mastectomy. Final pathology results revealed stage IA IDC with DCIS. Sentinel lymph nodes were negative for malignancy. The patient underwent adjuvant left chest wall radiation, endocrine therapy with tamoxifen, and implant reconstruction. After 2 years of follow-up, she is disease free.
In summary
Nipple discharge can be classified as physiologic or pathologic. For pathologic discharge, a thorough physical examination should be performed with subsequent imaging evaluation. First-line tools, based on patient age, include diagnostic mammography and targeted ultrasonography. Contrast-enhanced MRI is then recommended for negative or equivocal cases. All patients with pathologic nipple discharge should be referred to a breast surgeon following appropriate imaging evaluation. ●
CASE Young woman with discharge from one nipple
A 26-year-old African American woman presents with a 10-month history of left nipple discharge. The patient describes the discharge as spontaneous, colored dark brown to yellow, and occurring from a single opening in the nipple. The discharge is associated with left breast pain and fullness, without a palpable lump. The patient has no family or personal history of breast cancer.
Nipple discharge is the third most common breast-related symptom (after palpable masses and breast pain), with an estimated prevalence of 5% to 8% among premenopausal women.1 While most causes of nipple discharge reflect benign issues, approximately 5% to 12% of breast cancers have nipple discharge as the only symptom.2 Not surprisingly, nipple discharge creates anxiety for both patients and clinicians.
In this article, we—a breast imaging radiologist, gynecologist, and breast surgeon—outline key steps for evaluating and managing patients with nipple discharge.
Two types of nipple discharge
Nipple discharge can be characterized as physiologic or pathologic. The distinction is based on the patient’s history in conjunction with the clinical breast exam.
Physiologic nipple discharge often is bilateral, nonspontaneous, and white, yellow, green, or brown (TABLE).3 It often is due to nipple stimulation, and the patient can elicit discharge by manually manipulating the breast. Usually, multiple ducts are involved. Galactorrhea refers specifically to milky discharge and occurs most commonly during pregnancy or lactation.2 Galactorrhea that is not associated with pregnancy or lactation often is related to elevated prolactin or thyroid-stimulating hormone levels or to medications. One study reported that no cancers were found when discharge was nonspontaneous and colored or milky.4
Pathologic nipple discharge is defined as a spontaneous, bloody, clear, or single-duct discharge. A palpable mass in the same breast automatically increases the suspicion of the discharge, regardless of its color or spontaneity.2 The most common cause of pathologic nipple discharge is an intraductal papilloma, a benign epithelial tumor, which accounts for approximately 57% of cases.5
Although the risk of malignancy is low for all patients with nipple discharge, increasing age is associated with increased risk of breast cancer. One study demonstrated that among women aged 40 to 60 years presenting with nipple discharge, the prevalence of invasive cancer is 10%, and the percentage jumps to 32% among women older than 60.6
Breast exam. For any patient with nonlactational nipple discharge, we recommend a thorough breast examination. Deep palpation of all quadrants of the symptomatic breast, especially near the nipple areolar complex, should elicit nipple discharge without any direct squeezing of the nipple. If the patient’s history and physical exam are consistent with physiologic discharge, no further workup is needed. Reassure the patient and recommend appropriate breast cancer screening. Encourage the patient to decrease stimulation or manual manipulation of the nipples if the discharge bothers her.
Continue to: CASE Continued: Workup...
CASE Continued: Workup
On physical exam, the patient’s breasts are noted to be cup size DDD and asymmetric, with the left breast larger than the right; there is no contour deformity. There is no skin or nipple retraction, skin rash, swelling, or nipple changes bilaterally. No dominant masses are appreciated bilaterally. Manual compression elicits no nipple discharge.
Although the discharge is nonbloody, its spontaneity, unilaterality, and single-duct/orifice origin suggest a pathologic cause. The patient is referred for breast imaging.
Imaging workup for pathologic discharge
The American College of Radiology (ACR) Appropriateness Criteria is a useful tool that provides an evidence-based, easy-to-use algorithm for breast imaging in the patient with pathologic nipple discharge (FIGURE 1).6 The algorithm is categorized by patient age, with diagnostic mammography recommended for women aged 30 and older.6 Diagnostic mammography is recommended if the patient has not had a mammogram study in the last 6 months.6 For patients with no prior mammograms, we recommend bilateral diagnostic mammography to compare symmetry of the breasts.

Currently, no studies show that digital breast tomosynthesis (3-D mammography) has a benefit compared with standard 2-D mammography in women with pathologic nipple discharge.6 Given the increased sensitivity of digital breast tomosynthesis for cancer detection, however, in our practice it is standard to use tomosynthesis in the diagnostic evaluation of most patients.
Mammography
On mammography, ductal carcinoma in situ (DCIS) usually presents as calcifications. Both the morphology and distribution of calcifications are used to characterize them as suspicious or, typically, benign. DCIS usually presents as fine pleomorphic or fine linear branching calcifications in a segmental or linear distribution. In patients with pathologic nipple discharge and no other symptoms, the radiologist must closely examine the retroareolar region of the breast to assess for faint calcifications. Magnification views also can be performed to better characterize calcifications.
The sensitivity of mammography for nipple discharge varies in the literature, ranging from approximately 15% to 68%, with a specificity range of 38% to 98%.6 This results in a relatively low positive predictive value but a high negative predictive value of 90%.7 Mammographic sensitivity largely is limited by increased breast density. As more data emerge on the utility of digital breast tomosynthesis in dense breasts, mammographic sensitivity for nipple discharge will likely increase.
Ultrasonography
As an adjunct to mammography, the ACR Appropriateness Criteria recommends targeted (or “limited”) ultrasonography of the retroareolar region of the affected breast for patients aged 30 and older. Ultrasonography is useful to assess for intraductal masses and architectural distortion, and it has higher sensitivity but lower specificity than mammography. The sensitivity of ultrasonography for detecting breast cancer in patients presenting with nipple discharge is reported to be 56% to 80%.6 Ultrasonography can identify lesions not visible mammographically in 63% to 69% of cases.8 Although DCIS usually presents as calcifications, it also can present as an intraductal mass on ultrasonography.
The ACR recommends targeted ultrasonography for patients with nipple discharge and a negative mammogram, or to evaluate a suspicious mammographic abnormality such as architectural distortion, focal asymmetry, or a mass.6 For patient comfort, ultrasonography is the preferred modality for image-guided biopsy.
For women younger than 30 years, targeted ultrasonography is the initial imaging study of choice, according to the ACR criteria.6 Women younger than 30 years with pathologic nipple discharge have a very low risk of breast cancer and tend to have higher breast density, making mammography less useful. Although the radiation dose from mammography is negligible given technological improvements and dose-reduction techniques, ultrasonography remains the preferred initial imaging modality in young women, not only for nipple discharge but also for palpable lumps and focal breast pain.
Mammography is used as an adjunct to ultrasonography in women younger than 30 years when a suspicious abnormality is detected on ultrasonography, such as an intraductal mass or architectural distortion. In these cases, mammography can be used to assess for extent of disease or to visualize suspicious calcifications not well seen on ultrasonography.
For practical purposes regarding which imaging study to order for a patient, it is most efficient to order both a diagnostic mammogram (with tomosynthesis, if possible) and a targeted ultrasound scan of the affected breast. Even if both orders are not needed, having them available increases efficiency for both the radiologist and the ordering physician.
Continue to: CASE Continued: Imaging findings...
CASE Continued: Imaging findings
Given her age, the patient initially undergoes targeted ultrasonography. The grayscale image (FIGURE 2) demonstrates multiple mildly dilated ducts (white arrows) with surrounding hyperechogenicity of the fat (red arrows), indicating soft tissue edema. No intraductal mass is imaged. Given that the ultrasonography findings are not completely negative and are equivocal for malignancy, bilateral diagnostic mammography (FIGURE 3, left breast only) is performed. Standard full-field craniocaudal (FIGURE 3A) and mediolateral oblique (FIGURE 3B) mammographic views demonstrate a heterogeneously dense breast with a few calcifications in the retroareolar left breast (red ovals). No associated mass or architectural distortions are noted. The mammographic and sonographic findings do not reveal a definitive biopsy target.


Ductography
When a suspicious abnormality is visualized on either mammography or ultrasonography, the standard of care is to perform an image-guided biopsy of the abnormality. When the standard workup is negative or equivocal, the standard of care historically was to perform ductography.
Ductography is an invasive procedure that involves cannulating the suspicious duct with a small catheter and injecting radiopaque dye into the duct under mammographic guidance. While the sensitivity of ductography is higher than that of both mammography and ultrasonography, its specificity is lower than that of either modality.
Most cases of pathologic discharge are spontaneous and are not reproducible on the day of the procedure. If the procedural radiologist cannot visualize the duct that is producing the discharge, the procedure cannot be performed. Although most patients tolerate the procedure well, ductography produces patient discomfort from cannulation of the duct and injection of contrast.
Magnetic resonance imaging
Dynamic contrast-enhanced magnetic resonance imaging (MRI) is the most sensitive imaging study for evaluating pathologic nipple discharge, and it has largely replaced ductography as an adjunct to mammography and ultrasonography. MRI’s sensitivity for detecting breast cancer ranges from 93% to 100%.6 In addition, MRI allows visualization of the entire breast and areas peripheral to the field of view of a standard ductogram or ultrasound scan.9
Clinicians commonly ask, “Why not skip the mammogram and ultrasound scan and go straight to MRI, since it is so much more sensitive?” Breast MRI has several limitations, including relatively low specificity, cost, use of intravenous contrast, and patient discomfort (that is, claustrophobia, prone positioning). MRI should be utilized for pathologic discharge only when the mammogram and/or targeted ultrasound scans are negative or equivocal.
CASE Continued: Additional imaging
A contrast-enhanced MRI of the breasts (FIGURE 4) demonstrates a large area of non-mass enhancement (red oval) in the left breast, which involves most of the upper breast extending from the nipple to the posterior breast tissue; it measures approximately 7.3 x 14 x 9.1 cm in transverse, anteroposterior, and craniocaudal dimensions, respectively. There is no evidence of left pectoralis muscle involvement. An MRI-directed second look left breast ultrasonography (FIGURE 5) is performed, revealing a small irregular mass in the left breast 1 o’clock position, 10 to 11 cm from the nipple (red arrow). This area had not been imaged in the prior ultrasound scan due to its posterior location far from the nipple. Ultrasound-guided core needle biopsy is performed; moderately differentiated invasive ductal carcinoma (IDC) with high-grade DCIS is found.


Continue to: When to refer for surgery...
When to refer for surgery
No surgical evaluation or intervention is needed for physiologic nipple discharge. As mentioned previously, reassure the patient and recommend appropriate breast cancer screening. In the setting of pathologic discharge, however, referral to a breast surgeon may be indicated after appropriate imaging workup has been done.
Since abnormal imaging almost always results in a recommendation for image-guided biopsy, typically the biopsy is performed prior to the surgical consultation. Once the pathology report from the biopsy is available, the radiologist makes a radiologic-pathologic concordance statement and recommends surgical consultation. This process allows the surgeon to have all the necessary information at the initial visit.
However, in the setting of pathologic nipple discharge with normal breast imaging, the surgeon and patient may opt for close observation or surgery for definitive diagnosis. Surgical options include single-duct excision when nipple discharge is localized to one duct or central duct excision when nipple discharge cannot be localized to one duct.
CASE Continued: Follow-up
The patient was referred to a breast surgeon. Given the extent of disease in the left breast, breast conservation was not possible. The patient underwent left breast simple mastectomy with sentinel lymph node biopsy and prophylactic right simple mastectomy. Final pathology results revealed stage IA IDC with DCIS. Sentinel lymph nodes were negative for malignancy. The patient underwent adjuvant left chest wall radiation, endocrine therapy with tamoxifen, and implant reconstruction. After 2 years of follow-up, she is disease free.
In summary
Nipple discharge can be classified as physiologic or pathologic. For pathologic discharge, a thorough physical examination should be performed with subsequent imaging evaluation. First-line tools, based on patient age, include diagnostic mammography and targeted ultrasonography. Contrast-enhanced MRI is then recommended for negative or equivocal cases. All patients with pathologic nipple discharge should be referred to a breast surgeon following appropriate imaging evaluation. ●
- Alcock C, Layer GT. Predicting occult malignancy in nipple discharge. ANZ J Surg. 2010;80:646-649.
- Patel BK, Falcon S, Drukteinis J. Management of nipple discharge and the associated imaging findings. Am J Med. 2015;128:353-360.
- Mazzarello S, Arnaout A. Five things to know about nipple discharge. CMAJ. 2015;187:599.
- Goksel HA, Yagmurdur MC, Demirhan B, et al. Management strategies for patients with nipple discharge. Langenbecks Arch Surg. 2005;390:52-58.
- Vargas HI, Vargas MP, Eldrageely K, et al. Outcomes of clinical and surgical assessment of women with pathological nipple discharge. Am Surg. 2006;72:124-128.
- Expert Panel on Breast Imaging; Lee S, Tikha S, Moy L, et al. American College of Radiology Appropriateness Criteria: Evaluation of nipple discharge. https://acsearch.acr.org /docs/3099312/Narrative/. Accessed February 2, 2020.
- Cabioglu N, Hunt KK, Singletary SE, et al. Surgical decision making and factors determining a diagnosis of breast carcinoma in women presenting with nipple discharge. J Am Coll Surg. 2003;196:354-364.
- Morrogh M, Park A, Elkin EB, et al. Lessons learned from 416 cases of nipple discharge of the breast. Am J Surg. 2010;200:73-80.
- Morrogh M, Morris EA, Liberman L, et al. The predictive value of ductography and magnetic resonance imaging in the management of nipple discharge. Ann Surg Oncol. 2007;14:3369-3377.
- Alcock C, Layer GT. Predicting occult malignancy in nipple discharge. ANZ J Surg. 2010;80:646-649.
- Patel BK, Falcon S, Drukteinis J. Management of nipple discharge and the associated imaging findings. Am J Med. 2015;128:353-360.
- Mazzarello S, Arnaout A. Five things to know about nipple discharge. CMAJ. 2015;187:599.
- Goksel HA, Yagmurdur MC, Demirhan B, et al. Management strategies for patients with nipple discharge. Langenbecks Arch Surg. 2005;390:52-58.
- Vargas HI, Vargas MP, Eldrageely K, et al. Outcomes of clinical and surgical assessment of women with pathological nipple discharge. Am Surg. 2006;72:124-128.
- Expert Panel on Breast Imaging; Lee S, Tikha S, Moy L, et al. American College of Radiology Appropriateness Criteria: Evaluation of nipple discharge. https://acsearch.acr.org /docs/3099312/Narrative/. Accessed February 2, 2020.
- Cabioglu N, Hunt KK, Singletary SE, et al. Surgical decision making and factors determining a diagnosis of breast carcinoma in women presenting with nipple discharge. J Am Coll Surg. 2003;196:354-364.
- Morrogh M, Park A, Elkin EB, et al. Lessons learned from 416 cases of nipple discharge of the breast. Am J Surg. 2010;200:73-80.
- Morrogh M, Morris EA, Liberman L, et al. The predictive value of ductography and magnetic resonance imaging in the management of nipple discharge. Ann Surg Oncol. 2007;14:3369-3377.






