User login
Ticagrelor/aspirin combo: Fewer repeat strokes and deaths, but more bleeds
, new data show. However, severe bleeding was more common in the ticagrelor/aspirin group than in the aspirin-only group.
“We found that ticagrelor plus aspirin reduced the risk of stroke or death, compared to aspirin alone in patients presenting acutely with stroke or TIA,” reported lead author S. Claiborne Johnston, MD, PhD, dean and vice president for medical affairs, Dell Medical School, the University of Texas, Austin.
Although the combination also increased the risk for major hemorrhage, that increase was small and would not overwhelm the benefit, he said.
The study was published online July 16 in The New England Journal of Medicine.
Attractive properties
“Lots of patients have stroke in the days to weeks after first presenting with a stroke or TIA,” said Dr. Johnston, who is also the Frank and Charmaine Denius Distinguished Dean’s Chair at Dell Medical School. “Aspirin has been the standard of care but is only partially effective. Clopidogrel plus aspirin is another option that has recently been proven, [but] ticagrelor has attractive properties as an antiplatelet agent and works synergistically with aspirin,” he added.
Ticagrelor is a direct-acting antiplatelet agent that does not depend on metabolic activation and that “reversibly binds” and inhibits the P2Y12 receptor on platelets. Previous research has evaluated clopidogrel and aspirin for the secondary prevention of ischemic stroke or TIA. In an earlier trial, ticagrelor was no better than aspirin in preventing these subsequent events. However, the investigators noted that the combination of the two drugs has not been well studied.
The randomized, placebo-controlled, double-blind trial involved 11,016 patients at 414 sites in 28 countries. Patients who had experienced mild to moderate acute noncardioembolic ischemic stroke (mean age, 65 years; 39% women; roughly 54% White) were randomly assigned to receive either ticagrelor plus aspirin (n = 5,523) or aspirin alone (n = 5,493) for 30 days. Of these patients, 91% had sustained a stroke, and 9% had sustained a TIA.
Thirty days was chosen as the treatment period because the risk for subsequent stroke tends to occur mainly in the first month after an acute ischemic stroke or TIA. The primary outcome was “a composite of stroke or death in a time-to-first-event analysis from randomization to 30 days of follow-up.” For the study, “stroke” encompassed ischemic, hemorrhagic, or stroke of undetermined type, and “death” included deaths of all causes. Secondary outcomes included first subsequent ischemic stroke and disability (defined as a score of >1 on the Rankin Scale).
Almost all patients (99.5%) were taking aspirin during the treatment period, and most were also taking an antihypertensive and a statin (74% and 83%, respectively).
Patients in the ticagrelor/aspirin group had fewer primary-outcome events in comparison with those in the aspirin-only group (303 patients [5.5%] vs. 362 patients [6.6%]; hazard ratio, 0.83; 95% confidence interval, 0.71-0.96; P = 0.02). Incidence of subsequent ischemic stroke were similarly lower in the ticagrelor/aspirin group in comparison with the aspirin-only group (276 patients [5.0%] vs. 345 patients [6.3%]; HR, 0.79; 95% CI, 0.68-0.93; P = .004).
On the other hand, there was no significant difference between the groups in the incidence of overall disability (23.8% of the patients in the ticagrelor/aspirin group and in 24.1% of the patients in the aspirin group; odds ratio, 0.98; 95% CI, 0.89-1.07; P = .61).
There were differences between the groups in severe bleeding, which occurred in 28 patients (0.5%) in the ticagrelor/aspirin group and in seven patients (0.15) in the ticagrelor group (HR, 3.99; 95% CI, 1.74-9.14; P = .001). Moreover, more patients in the ticagrelor/aspirin group experienced a composite of intracranial hemorrhage or fatal bleeding compared with the aspirin-only group (0.4% vs 0.1%). Fatal bleeding occurred in 0.2% of patients in the ticagrelor/aspirin group versus 0.1% of patients in the aspirin group. More patients in the ticagrelor-aspirin group permanently discontinued the treatment because of bleeding than in the aspirin-only group (2.8% vs. 0.6%).
“The benefit from treatment with ticagrelor/aspirin, as compared with aspirin alone, would be expected to result in a number needed to treat of 92 to prevent one primary outcome event, and a number needed to harm of 263 for severe bleeding,” the authors noted.
Risks versus benefits
Commenting on the study, Konark Malhotra, MD, a vascular neurologist at Allegheny Health Network, Pittsburgh, noted that ticagrelor is an antiplatelet medication “that adds to the armamentarium of stroke neurologists for the treatment of mild acute ischemic or high-risk TIA patients.” Dr. Malhotra, who was not involved with the study, added that the “combined use of ticagrelor and aspirin is effective in the reduction of ischemic events, however, at the expense of increased risk of bleeding events.”
In an accompanying editorial, Peter Rothwell, MD, PhD, of the Wolfson Center for Prevention of Stroke and Dementia, Nuffield Department of Clinical Neurosciences at the University of Oxford (England) who was not involved with the study, suggested that the “bleeding risk associated with ticagrelor and aspirin might exceed the benefit among lower-risk patients who make up the majority in practice, and so the results should not be overgeneralized.” Moreover, “regardless of which combination of antiplatelet therapy is favored for the high-risk minority, all patients should receive aspirin immediately after TIA, unless aspirin is contraindicated.”
He noted that “too many patients are sent home from emergency departments without this simple treatment that substantially reduces the risk and severity of early recurrent stroke.”
The study was supported by AstraZeneca. Dr. Johnston has received a grant from AstraZeneca and nonfinancial support from SANOFI. Dr. Rothwell has received personal fees from Bayer and BMS. Dr. Malhotra has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
, new data show. However, severe bleeding was more common in the ticagrelor/aspirin group than in the aspirin-only group.
“We found that ticagrelor plus aspirin reduced the risk of stroke or death, compared to aspirin alone in patients presenting acutely with stroke or TIA,” reported lead author S. Claiborne Johnston, MD, PhD, dean and vice president for medical affairs, Dell Medical School, the University of Texas, Austin.
Although the combination also increased the risk for major hemorrhage, that increase was small and would not overwhelm the benefit, he said.
The study was published online July 16 in The New England Journal of Medicine.
Attractive properties
“Lots of patients have stroke in the days to weeks after first presenting with a stroke or TIA,” said Dr. Johnston, who is also the Frank and Charmaine Denius Distinguished Dean’s Chair at Dell Medical School. “Aspirin has been the standard of care but is only partially effective. Clopidogrel plus aspirin is another option that has recently been proven, [but] ticagrelor has attractive properties as an antiplatelet agent and works synergistically with aspirin,” he added.
Ticagrelor is a direct-acting antiplatelet agent that does not depend on metabolic activation and that “reversibly binds” and inhibits the P2Y12 receptor on platelets. Previous research has evaluated clopidogrel and aspirin for the secondary prevention of ischemic stroke or TIA. In an earlier trial, ticagrelor was no better than aspirin in preventing these subsequent events. However, the investigators noted that the combination of the two drugs has not been well studied.
The randomized, placebo-controlled, double-blind trial involved 11,016 patients at 414 sites in 28 countries. Patients who had experienced mild to moderate acute noncardioembolic ischemic stroke (mean age, 65 years; 39% women; roughly 54% White) were randomly assigned to receive either ticagrelor plus aspirin (n = 5,523) or aspirin alone (n = 5,493) for 30 days. Of these patients, 91% had sustained a stroke, and 9% had sustained a TIA.
Thirty days was chosen as the treatment period because the risk for subsequent stroke tends to occur mainly in the first month after an acute ischemic stroke or TIA. The primary outcome was “a composite of stroke or death in a time-to-first-event analysis from randomization to 30 days of follow-up.” For the study, “stroke” encompassed ischemic, hemorrhagic, or stroke of undetermined type, and “death” included deaths of all causes. Secondary outcomes included first subsequent ischemic stroke and disability (defined as a score of >1 on the Rankin Scale).
Almost all patients (99.5%) were taking aspirin during the treatment period, and most were also taking an antihypertensive and a statin (74% and 83%, respectively).
Patients in the ticagrelor/aspirin group had fewer primary-outcome events in comparison with those in the aspirin-only group (303 patients [5.5%] vs. 362 patients [6.6%]; hazard ratio, 0.83; 95% confidence interval, 0.71-0.96; P = 0.02). Incidence of subsequent ischemic stroke were similarly lower in the ticagrelor/aspirin group in comparison with the aspirin-only group (276 patients [5.0%] vs. 345 patients [6.3%]; HR, 0.79; 95% CI, 0.68-0.93; P = .004).
On the other hand, there was no significant difference between the groups in the incidence of overall disability (23.8% of the patients in the ticagrelor/aspirin group and in 24.1% of the patients in the aspirin group; odds ratio, 0.98; 95% CI, 0.89-1.07; P = .61).
There were differences between the groups in severe bleeding, which occurred in 28 patients (0.5%) in the ticagrelor/aspirin group and in seven patients (0.15) in the ticagrelor group (HR, 3.99; 95% CI, 1.74-9.14; P = .001). Moreover, more patients in the ticagrelor/aspirin group experienced a composite of intracranial hemorrhage or fatal bleeding compared with the aspirin-only group (0.4% vs 0.1%). Fatal bleeding occurred in 0.2% of patients in the ticagrelor/aspirin group versus 0.1% of patients in the aspirin group. More patients in the ticagrelor-aspirin group permanently discontinued the treatment because of bleeding than in the aspirin-only group (2.8% vs. 0.6%).
“The benefit from treatment with ticagrelor/aspirin, as compared with aspirin alone, would be expected to result in a number needed to treat of 92 to prevent one primary outcome event, and a number needed to harm of 263 for severe bleeding,” the authors noted.
Risks versus benefits
Commenting on the study, Konark Malhotra, MD, a vascular neurologist at Allegheny Health Network, Pittsburgh, noted that ticagrelor is an antiplatelet medication “that adds to the armamentarium of stroke neurologists for the treatment of mild acute ischemic or high-risk TIA patients.” Dr. Malhotra, who was not involved with the study, added that the “combined use of ticagrelor and aspirin is effective in the reduction of ischemic events, however, at the expense of increased risk of bleeding events.”
In an accompanying editorial, Peter Rothwell, MD, PhD, of the Wolfson Center for Prevention of Stroke and Dementia, Nuffield Department of Clinical Neurosciences at the University of Oxford (England) who was not involved with the study, suggested that the “bleeding risk associated with ticagrelor and aspirin might exceed the benefit among lower-risk patients who make up the majority in practice, and so the results should not be overgeneralized.” Moreover, “regardless of which combination of antiplatelet therapy is favored for the high-risk minority, all patients should receive aspirin immediately after TIA, unless aspirin is contraindicated.”
He noted that “too many patients are sent home from emergency departments without this simple treatment that substantially reduces the risk and severity of early recurrent stroke.”
The study was supported by AstraZeneca. Dr. Johnston has received a grant from AstraZeneca and nonfinancial support from SANOFI. Dr. Rothwell has received personal fees from Bayer and BMS. Dr. Malhotra has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
, new data show. However, severe bleeding was more common in the ticagrelor/aspirin group than in the aspirin-only group.
“We found that ticagrelor plus aspirin reduced the risk of stroke or death, compared to aspirin alone in patients presenting acutely with stroke or TIA,” reported lead author S. Claiborne Johnston, MD, PhD, dean and vice president for medical affairs, Dell Medical School, the University of Texas, Austin.
Although the combination also increased the risk for major hemorrhage, that increase was small and would not overwhelm the benefit, he said.
The study was published online July 16 in The New England Journal of Medicine.
Attractive properties
“Lots of patients have stroke in the days to weeks after first presenting with a stroke or TIA,” said Dr. Johnston, who is also the Frank and Charmaine Denius Distinguished Dean’s Chair at Dell Medical School. “Aspirin has been the standard of care but is only partially effective. Clopidogrel plus aspirin is another option that has recently been proven, [but] ticagrelor has attractive properties as an antiplatelet agent and works synergistically with aspirin,” he added.
Ticagrelor is a direct-acting antiplatelet agent that does not depend on metabolic activation and that “reversibly binds” and inhibits the P2Y12 receptor on platelets. Previous research has evaluated clopidogrel and aspirin for the secondary prevention of ischemic stroke or TIA. In an earlier trial, ticagrelor was no better than aspirin in preventing these subsequent events. However, the investigators noted that the combination of the two drugs has not been well studied.
The randomized, placebo-controlled, double-blind trial involved 11,016 patients at 414 sites in 28 countries. Patients who had experienced mild to moderate acute noncardioembolic ischemic stroke (mean age, 65 years; 39% women; roughly 54% White) were randomly assigned to receive either ticagrelor plus aspirin (n = 5,523) or aspirin alone (n = 5,493) for 30 days. Of these patients, 91% had sustained a stroke, and 9% had sustained a TIA.
Thirty days was chosen as the treatment period because the risk for subsequent stroke tends to occur mainly in the first month after an acute ischemic stroke or TIA. The primary outcome was “a composite of stroke or death in a time-to-first-event analysis from randomization to 30 days of follow-up.” For the study, “stroke” encompassed ischemic, hemorrhagic, or stroke of undetermined type, and “death” included deaths of all causes. Secondary outcomes included first subsequent ischemic stroke and disability (defined as a score of >1 on the Rankin Scale).
Almost all patients (99.5%) were taking aspirin during the treatment period, and most were also taking an antihypertensive and a statin (74% and 83%, respectively).
Patients in the ticagrelor/aspirin group had fewer primary-outcome events in comparison with those in the aspirin-only group (303 patients [5.5%] vs. 362 patients [6.6%]; hazard ratio, 0.83; 95% confidence interval, 0.71-0.96; P = 0.02). Incidence of subsequent ischemic stroke were similarly lower in the ticagrelor/aspirin group in comparison with the aspirin-only group (276 patients [5.0%] vs. 345 patients [6.3%]; HR, 0.79; 95% CI, 0.68-0.93; P = .004).
On the other hand, there was no significant difference between the groups in the incidence of overall disability (23.8% of the patients in the ticagrelor/aspirin group and in 24.1% of the patients in the aspirin group; odds ratio, 0.98; 95% CI, 0.89-1.07; P = .61).
There were differences between the groups in severe bleeding, which occurred in 28 patients (0.5%) in the ticagrelor/aspirin group and in seven patients (0.15) in the ticagrelor group (HR, 3.99; 95% CI, 1.74-9.14; P = .001). Moreover, more patients in the ticagrelor/aspirin group experienced a composite of intracranial hemorrhage or fatal bleeding compared with the aspirin-only group (0.4% vs 0.1%). Fatal bleeding occurred in 0.2% of patients in the ticagrelor/aspirin group versus 0.1% of patients in the aspirin group. More patients in the ticagrelor-aspirin group permanently discontinued the treatment because of bleeding than in the aspirin-only group (2.8% vs. 0.6%).
“The benefit from treatment with ticagrelor/aspirin, as compared with aspirin alone, would be expected to result in a number needed to treat of 92 to prevent one primary outcome event, and a number needed to harm of 263 for severe bleeding,” the authors noted.
Risks versus benefits
Commenting on the study, Konark Malhotra, MD, a vascular neurologist at Allegheny Health Network, Pittsburgh, noted that ticagrelor is an antiplatelet medication “that adds to the armamentarium of stroke neurologists for the treatment of mild acute ischemic or high-risk TIA patients.” Dr. Malhotra, who was not involved with the study, added that the “combined use of ticagrelor and aspirin is effective in the reduction of ischemic events, however, at the expense of increased risk of bleeding events.”
In an accompanying editorial, Peter Rothwell, MD, PhD, of the Wolfson Center for Prevention of Stroke and Dementia, Nuffield Department of Clinical Neurosciences at the University of Oxford (England) who was not involved with the study, suggested that the “bleeding risk associated with ticagrelor and aspirin might exceed the benefit among lower-risk patients who make up the majority in practice, and so the results should not be overgeneralized.” Moreover, “regardless of which combination of antiplatelet therapy is favored for the high-risk minority, all patients should receive aspirin immediately after TIA, unless aspirin is contraindicated.”
He noted that “too many patients are sent home from emergency departments without this simple treatment that substantially reduces the risk and severity of early recurrent stroke.”
The study was supported by AstraZeneca. Dr. Johnston has received a grant from AstraZeneca and nonfinancial support from SANOFI. Dr. Rothwell has received personal fees from Bayer and BMS. Dr. Malhotra has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
From New England Journal of Medicine
50-year-old man • foot pain • “purple” toe • history of smoking • Dx?
THE CASE
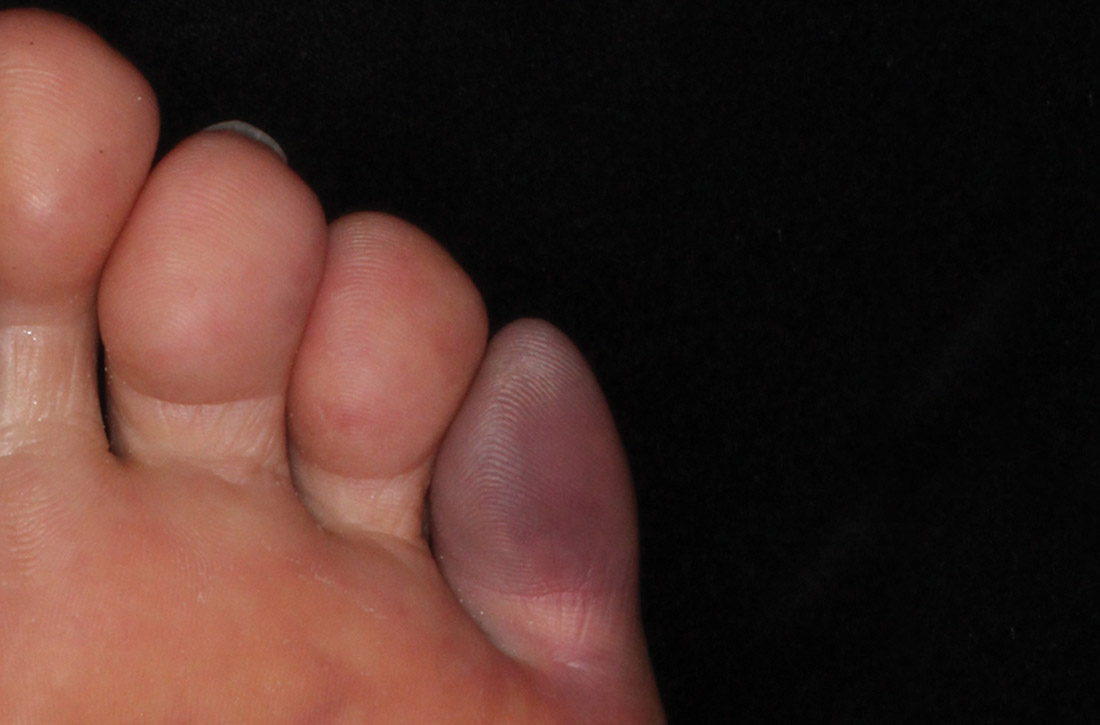
A 50-year-old man presented to the primary care office for evaluation of foot pain. The day before, his left fifth toe had become exquisitely tender. He distinctly remembered that when he awoke, there was no discoloration or pain, but the toe later became “purple.” He denied any trauma. His medical record was notable for an extensive smoking history and a family history of early cardiovascular disease.
The patient appeared well but in obvious distress, secondary to the pain. His vital signs were unremarkable. His head, neck, lung, and cardiac exams revealed no abnormalities. Physical examination revealed a left fifth toe that was dusky purple and warm to the touch. Pain disproportionate to examination was noted on the anterior aspect of the toe, with limited range of motion. The patient walked with a compensated gait. Pulses were palpable on the posterior tibial (PT) and dorsalis pedis (DP) regions.
DIAGNOSIS
Based on our exam findings, we suspected a vascular injury and recommended an emergency consult by Podiatry, for which he was scheduled the following morning. The podiatric evaluation confirmed concern for a vascular injury and prompted a request for an emergent evaluation by Vascular Surgery.
The patient was seen emergently on Day 4 for a vascular surgery evaluation. Examination at that time showed a nearly absent femoral pulse on the left side and diminished and monophasic DP and PT pulses. His left foot demonstrated nonblanchable purpura that was clinically consistent with cholesterol embolization syndrome (CES).
We calculated the patient’s ankle-brachial index, and computed tomography angiography (CTA) was performed. While results were pending, the patient was started on aspirin 81 mg, clopidogrel 75 mg, and atorvastatin 40 mg, for a suspected slowly progressing iliac artery stenosis with a resulting acute atheroembolic event.
The CTA report showed a high-grade stenosis at the bifurcation of the left iliac artery, extending into both external and internal arteries. Of note, mild atherosclerotic disease without significant occlusion and runoff to the foot was observed into the tibial arteries. The stenosis extended into the profonda femoris artery, as well.
DISCUSSION
Atherosclerotic plaques are commonly encountered in patients with atherosclerotic disease; however, there are 2 varieties of emboli that arise from these plaques and one is often overlooked.1-4 The more common of these variants, thromboemboli, originates from an atherosclerotic plaque and can become lodged in a medium or large vessel as a single embolus.
Continue to: By contrast...
By contrast, atheroemboli (commonly known as cholesterol emboli or cholesterol crystal embolization) originate from atherosclerotic plaques in the aorta or another large artery,5 which are prone to embolize if the underlying plaque experiences stress. As the plaque erodes, cholesterol crystals break off and embolize distally. These smaller crystals flood into the circulation, allowing a shower of emboli over time to occlude the arterioles. As occlusion spreads through the arterioles, multiple organ systems are affected. (It was previously thought that procedure-associated cases were common, but a literature review has not borne this out.5)
The shower of emboli often triggers a systemic inflammatory response, causing nondescript abnormalities of laboratory inflammatory markers.6,7 Interestingly, hypereosinophilia is noted in about 80% of patients with CES.8
No disease-specific testing. A confounding factor in validating the diagnosis of CES is the lack of disease-specific testing. However, CES should be considered in a patient with acute kidney injury and hypereosinophilia. Making the diagnosis requires a high degree of clinical suspicion. Any organ can be affected, although the brain, kidneys, gastrointestinal tract, skin, and skeletal muscles of the lower extremities are most frequently involved.9 If left undiagnosed, the results can be devastating: slow and chronic injury to a variety of organ systems over time, which may not be recognized as a harbinger of an insidious underlying process causing end-organ damage.
Technically, definitive diagnosis can be made by biopsy of an affected organ. However, biopsy’s utility is limited due to potential for sampling error, accessibility (as noted, the location of the involved organ[s] may make biopsy nearly impossible without additional surgical risk9), and risk of poor healing to the biopsy site.10
Treatment is two-fold: supportive care for the affected end organ and prevention of subsequent embolic events. The latter entails aggressive risk factor reduction strategies, such as smoking cessation, statin therapy, blood pressure control, and blood sugar control. Warfarin is not recommended for treatment of CES due to the risk of further plaque rupture, hemorrhage, acute and chronic renal failure, and cholesterol microembolization to other organs.11,12
Continue to: Our patient
Our patient. After testing confirmed the diagnosis, the patient underwent an angioplasty. A stent was placed in his left iliac artery. He was continued on antiplatelet and statin therapy and was again counseled regarding smoking cessation.
THE TAKEAWAY
When patients present with symptoms suggestive of a vascular origin, consider CES. Although it can affect a multitude of organs, acute kidney injury and hypereosinophilia are the most common signs. Immediate intervention is required to save the affected organ; strategizing to reduce the risk for further embolic events is also key.
Prompt recognition of vascular emergencies, including those that are harbingers of atherosclerotic disease, is essential. As clinicians, it is imperative that we use all resources to address significant population health burdens. If CES is more prevalent than commonly thought, consideration should be given to increasing education about early detection and treatment of this disorder, including the reinforcement of primary prevention and aggressive treatment of risk factors for atherosclerotic cardiovascular disease.
CORRESPONDENCE
Meagan Vermeulen, MD, FAAFP, Department of Family Medicine, Rowan University School of Osteopathic Medicine, 42 East Laurel Road, Suite 2100A, Stratford, NJ 08084; [email protected]
1. Tunick PA, Kronzon I. Atheromas of the thoracic aorta: clinical and therapeutic update. J Am Coll Cardiol. 2000;35:545-554.
2. Amarenco P, Duyckaerts C, Tzourio C, et al. The prevalence of ulcerated plaques in the aortic arch in patients with stroke. N Engl J Med. 1992;326:221-225.
3. Amarenco P, Cohen A, Tzourio C, et al. Atherosclerotic disease of the aortic arch and the risk of ischemic stroke. N Engl J Med. 1994;331:1474-1479.
4. Amarenco P, Cohen A, et al; French Study of Aortic Plaques in Stroke Group. Atherosclerotic disease of the aortic arch as a risk factor for recurrent ischemic stroke. N Engl J Med. 1996;334:1216-1221.
5. Ong HT, Elmsly WG, Friedlander DH. Cholesterol atheroembolism: an increasingly frequent complication of cardiac catheterisation. Med J Aust. 1991;154:412-414.
6. Kronzon I, Saric M. Cholesterol embolization syndrome. Circulation. 2010;122:631-641.
7. Saric M, Kronzon I. Cholesterol embolization syndrome. Curr Opin Cardiol. 2011;26:472-479.
8. Kasinath BS, Lewis EJ. Eosinophilia as a clue to the diagnosis of atheroembolic renal disease. Arch Intern Med. 1987;147:1384-1385.
9. Quinones A, Saric M. The cholesterol emboli syndrome in atherosclerosis. Curr Atheroscler Rep. 2013;15:315.
10. Jucgla A, Moreso F, Muniesa C, et al. Cholesterol embolism: still an unrecognized entity with a high mortality rate. J Am Acad Dermatol. 2006;55:786-793.
11. Kim H, Zhen DB, Lieske JC, et al. Treatment of cholesterol embolization syndrome in the setting of an acute indication for anticoagulation therapy. J Med Cases. 2014;5:376-379.
12. Igarashi Y, Akimoto T, Kobayashi T, et al. Performing anticoagulation: a puzzling case of cholesterol embolization syndrome. Clin Med Insights Case Rep. 2017;10:1179547616684649. doi:10.1177/1179547616684649.
THE CASE
A 50-year-old man presented to the primary care office for evaluation of foot pain. The day before, his left fifth toe had become exquisitely tender. He distinctly remembered that when he awoke, there was no discoloration or pain, but the toe later became “purple.” He denied any trauma. His medical record was notable for an extensive smoking history and a family history of early cardiovascular disease.
The patient appeared well but in obvious distress, secondary to the pain. His vital signs were unremarkable. His head, neck, lung, and cardiac exams revealed no abnormalities. Physical examination revealed a left fifth toe that was dusky purple and warm to the touch. Pain disproportionate to examination was noted on the anterior aspect of the toe, with limited range of motion. The patient walked with a compensated gait. Pulses were palpable on the posterior tibial (PT) and dorsalis pedis (DP) regions.
DIAGNOSIS
Based on our exam findings, we suspected a vascular injury and recommended an emergency consult by Podiatry, for which he was scheduled the following morning. The podiatric evaluation confirmed concern for a vascular injury and prompted a request for an emergent evaluation by Vascular Surgery.
The patient was seen emergently on Day 4 for a vascular surgery evaluation. Examination at that time showed a nearly absent femoral pulse on the left side and diminished and monophasic DP and PT pulses. His left foot demonstrated nonblanchable purpura that was clinically consistent with cholesterol embolization syndrome (CES).
We calculated the patient’s ankle-brachial index, and computed tomography angiography (CTA) was performed. While results were pending, the patient was started on aspirin 81 mg, clopidogrel 75 mg, and atorvastatin 40 mg, for a suspected slowly progressing iliac artery stenosis with a resulting acute atheroembolic event.
The CTA report showed a high-grade stenosis at the bifurcation of the left iliac artery, extending into both external and internal arteries. Of note, mild atherosclerotic disease without significant occlusion and runoff to the foot was observed into the tibial arteries. The stenosis extended into the profonda femoris artery, as well.
DISCUSSION
Atherosclerotic plaques are commonly encountered in patients with atherosclerotic disease; however, there are 2 varieties of emboli that arise from these plaques and one is often overlooked.1-4 The more common of these variants, thromboemboli, originates from an atherosclerotic plaque and can become lodged in a medium or large vessel as a single embolus.
Continue to: By contrast...
By contrast, atheroemboli (commonly known as cholesterol emboli or cholesterol crystal embolization) originate from atherosclerotic plaques in the aorta or another large artery,5 which are prone to embolize if the underlying plaque experiences stress. As the plaque erodes, cholesterol crystals break off and embolize distally. These smaller crystals flood into the circulation, allowing a shower of emboli over time to occlude the arterioles. As occlusion spreads through the arterioles, multiple organ systems are affected. (It was previously thought that procedure-associated cases were common, but a literature review has not borne this out.5)
The shower of emboli often triggers a systemic inflammatory response, causing nondescript abnormalities of laboratory inflammatory markers.6,7 Interestingly, hypereosinophilia is noted in about 80% of patients with CES.8
No disease-specific testing. A confounding factor in validating the diagnosis of CES is the lack of disease-specific testing. However, CES should be considered in a patient with acute kidney injury and hypereosinophilia. Making the diagnosis requires a high degree of clinical suspicion. Any organ can be affected, although the brain, kidneys, gastrointestinal tract, skin, and skeletal muscles of the lower extremities are most frequently involved.9 If left undiagnosed, the results can be devastating: slow and chronic injury to a variety of organ systems over time, which may not be recognized as a harbinger of an insidious underlying process causing end-organ damage.
Technically, definitive diagnosis can be made by biopsy of an affected organ. However, biopsy’s utility is limited due to potential for sampling error, accessibility (as noted, the location of the involved organ[s] may make biopsy nearly impossible without additional surgical risk9), and risk of poor healing to the biopsy site.10
Treatment is two-fold: supportive care for the affected end organ and prevention of subsequent embolic events. The latter entails aggressive risk factor reduction strategies, such as smoking cessation, statin therapy, blood pressure control, and blood sugar control. Warfarin is not recommended for treatment of CES due to the risk of further plaque rupture, hemorrhage, acute and chronic renal failure, and cholesterol microembolization to other organs.11,12
Continue to: Our patient
Our patient. After testing confirmed the diagnosis, the patient underwent an angioplasty. A stent was placed in his left iliac artery. He was continued on antiplatelet and statin therapy and was again counseled regarding smoking cessation.
THE TAKEAWAY
When patients present with symptoms suggestive of a vascular origin, consider CES. Although it can affect a multitude of organs, acute kidney injury and hypereosinophilia are the most common signs. Immediate intervention is required to save the affected organ; strategizing to reduce the risk for further embolic events is also key.
Prompt recognition of vascular emergencies, including those that are harbingers of atherosclerotic disease, is essential. As clinicians, it is imperative that we use all resources to address significant population health burdens. If CES is more prevalent than commonly thought, consideration should be given to increasing education about early detection and treatment of this disorder, including the reinforcement of primary prevention and aggressive treatment of risk factors for atherosclerotic cardiovascular disease.
CORRESPONDENCE
Meagan Vermeulen, MD, FAAFP, Department of Family Medicine, Rowan University School of Osteopathic Medicine, 42 East Laurel Road, Suite 2100A, Stratford, NJ 08084; [email protected]
THE CASE
A 50-year-old man presented to the primary care office for evaluation of foot pain. The day before, his left fifth toe had become exquisitely tender. He distinctly remembered that when he awoke, there was no discoloration or pain, but the toe later became “purple.” He denied any trauma. His medical record was notable for an extensive smoking history and a family history of early cardiovascular disease.
The patient appeared well but in obvious distress, secondary to the pain. His vital signs were unremarkable. His head, neck, lung, and cardiac exams revealed no abnormalities. Physical examination revealed a left fifth toe that was dusky purple and warm to the touch. Pain disproportionate to examination was noted on the anterior aspect of the toe, with limited range of motion. The patient walked with a compensated gait. Pulses were palpable on the posterior tibial (PT) and dorsalis pedis (DP) regions.
DIAGNOSIS
Based on our exam findings, we suspected a vascular injury and recommended an emergency consult by Podiatry, for which he was scheduled the following morning. The podiatric evaluation confirmed concern for a vascular injury and prompted a request for an emergent evaluation by Vascular Surgery.
The patient was seen emergently on Day 4 for a vascular surgery evaluation. Examination at that time showed a nearly absent femoral pulse on the left side and diminished and monophasic DP and PT pulses. His left foot demonstrated nonblanchable purpura that was clinically consistent with cholesterol embolization syndrome (CES).
We calculated the patient’s ankle-brachial index, and computed tomography angiography (CTA) was performed. While results were pending, the patient was started on aspirin 81 mg, clopidogrel 75 mg, and atorvastatin 40 mg, for a suspected slowly progressing iliac artery stenosis with a resulting acute atheroembolic event.
The CTA report showed a high-grade stenosis at the bifurcation of the left iliac artery, extending into both external and internal arteries. Of note, mild atherosclerotic disease without significant occlusion and runoff to the foot was observed into the tibial arteries. The stenosis extended into the profonda femoris artery, as well.
DISCUSSION
Atherosclerotic plaques are commonly encountered in patients with atherosclerotic disease; however, there are 2 varieties of emboli that arise from these plaques and one is often overlooked.1-4 The more common of these variants, thromboemboli, originates from an atherosclerotic plaque and can become lodged in a medium or large vessel as a single embolus.
Continue to: By contrast...
By contrast, atheroemboli (commonly known as cholesterol emboli or cholesterol crystal embolization) originate from atherosclerotic plaques in the aorta or another large artery,5 which are prone to embolize if the underlying plaque experiences stress. As the plaque erodes, cholesterol crystals break off and embolize distally. These smaller crystals flood into the circulation, allowing a shower of emboli over time to occlude the arterioles. As occlusion spreads through the arterioles, multiple organ systems are affected. (It was previously thought that procedure-associated cases were common, but a literature review has not borne this out.5)
The shower of emboli often triggers a systemic inflammatory response, causing nondescript abnormalities of laboratory inflammatory markers.6,7 Interestingly, hypereosinophilia is noted in about 80% of patients with CES.8
No disease-specific testing. A confounding factor in validating the diagnosis of CES is the lack of disease-specific testing. However, CES should be considered in a patient with acute kidney injury and hypereosinophilia. Making the diagnosis requires a high degree of clinical suspicion. Any organ can be affected, although the brain, kidneys, gastrointestinal tract, skin, and skeletal muscles of the lower extremities are most frequently involved.9 If left undiagnosed, the results can be devastating: slow and chronic injury to a variety of organ systems over time, which may not be recognized as a harbinger of an insidious underlying process causing end-organ damage.
Technically, definitive diagnosis can be made by biopsy of an affected organ. However, biopsy’s utility is limited due to potential for sampling error, accessibility (as noted, the location of the involved organ[s] may make biopsy nearly impossible without additional surgical risk9), and risk of poor healing to the biopsy site.10
Treatment is two-fold: supportive care for the affected end organ and prevention of subsequent embolic events. The latter entails aggressive risk factor reduction strategies, such as smoking cessation, statin therapy, blood pressure control, and blood sugar control. Warfarin is not recommended for treatment of CES due to the risk of further plaque rupture, hemorrhage, acute and chronic renal failure, and cholesterol microembolization to other organs.11,12
Continue to: Our patient
Our patient. After testing confirmed the diagnosis, the patient underwent an angioplasty. A stent was placed in his left iliac artery. He was continued on antiplatelet and statin therapy and was again counseled regarding smoking cessation.
THE TAKEAWAY
When patients present with symptoms suggestive of a vascular origin, consider CES. Although it can affect a multitude of organs, acute kidney injury and hypereosinophilia are the most common signs. Immediate intervention is required to save the affected organ; strategizing to reduce the risk for further embolic events is also key.
Prompt recognition of vascular emergencies, including those that are harbingers of atherosclerotic disease, is essential. As clinicians, it is imperative that we use all resources to address significant population health burdens. If CES is more prevalent than commonly thought, consideration should be given to increasing education about early detection and treatment of this disorder, including the reinforcement of primary prevention and aggressive treatment of risk factors for atherosclerotic cardiovascular disease.
CORRESPONDENCE
Meagan Vermeulen, MD, FAAFP, Department of Family Medicine, Rowan University School of Osteopathic Medicine, 42 East Laurel Road, Suite 2100A, Stratford, NJ 08084; [email protected]
1. Tunick PA, Kronzon I. Atheromas of the thoracic aorta: clinical and therapeutic update. J Am Coll Cardiol. 2000;35:545-554.
2. Amarenco P, Duyckaerts C, Tzourio C, et al. The prevalence of ulcerated plaques in the aortic arch in patients with stroke. N Engl J Med. 1992;326:221-225.
3. Amarenco P, Cohen A, Tzourio C, et al. Atherosclerotic disease of the aortic arch and the risk of ischemic stroke. N Engl J Med. 1994;331:1474-1479.
4. Amarenco P, Cohen A, et al; French Study of Aortic Plaques in Stroke Group. Atherosclerotic disease of the aortic arch as a risk factor for recurrent ischemic stroke. N Engl J Med. 1996;334:1216-1221.
5. Ong HT, Elmsly WG, Friedlander DH. Cholesterol atheroembolism: an increasingly frequent complication of cardiac catheterisation. Med J Aust. 1991;154:412-414.
6. Kronzon I, Saric M. Cholesterol embolization syndrome. Circulation. 2010;122:631-641.
7. Saric M, Kronzon I. Cholesterol embolization syndrome. Curr Opin Cardiol. 2011;26:472-479.
8. Kasinath BS, Lewis EJ. Eosinophilia as a clue to the diagnosis of atheroembolic renal disease. Arch Intern Med. 1987;147:1384-1385.
9. Quinones A, Saric M. The cholesterol emboli syndrome in atherosclerosis. Curr Atheroscler Rep. 2013;15:315.
10. Jucgla A, Moreso F, Muniesa C, et al. Cholesterol embolism: still an unrecognized entity with a high mortality rate. J Am Acad Dermatol. 2006;55:786-793.
11. Kim H, Zhen DB, Lieske JC, et al. Treatment of cholesterol embolization syndrome in the setting of an acute indication for anticoagulation therapy. J Med Cases. 2014;5:376-379.
12. Igarashi Y, Akimoto T, Kobayashi T, et al. Performing anticoagulation: a puzzling case of cholesterol embolization syndrome. Clin Med Insights Case Rep. 2017;10:1179547616684649. doi:10.1177/1179547616684649.
1. Tunick PA, Kronzon I. Atheromas of the thoracic aorta: clinical and therapeutic update. J Am Coll Cardiol. 2000;35:545-554.
2. Amarenco P, Duyckaerts C, Tzourio C, et al. The prevalence of ulcerated plaques in the aortic arch in patients with stroke. N Engl J Med. 1992;326:221-225.
3. Amarenco P, Cohen A, Tzourio C, et al. Atherosclerotic disease of the aortic arch and the risk of ischemic stroke. N Engl J Med. 1994;331:1474-1479.
4. Amarenco P, Cohen A, et al; French Study of Aortic Plaques in Stroke Group. Atherosclerotic disease of the aortic arch as a risk factor for recurrent ischemic stroke. N Engl J Med. 1996;334:1216-1221.
5. Ong HT, Elmsly WG, Friedlander DH. Cholesterol atheroembolism: an increasingly frequent complication of cardiac catheterisation. Med J Aust. 1991;154:412-414.
6. Kronzon I, Saric M. Cholesterol embolization syndrome. Circulation. 2010;122:631-641.
7. Saric M, Kronzon I. Cholesterol embolization syndrome. Curr Opin Cardiol. 2011;26:472-479.
8. Kasinath BS, Lewis EJ. Eosinophilia as a clue to the diagnosis of atheroembolic renal disease. Arch Intern Med. 1987;147:1384-1385.
9. Quinones A, Saric M. The cholesterol emboli syndrome in atherosclerosis. Curr Atheroscler Rep. 2013;15:315.
10. Jucgla A, Moreso F, Muniesa C, et al. Cholesterol embolism: still an unrecognized entity with a high mortality rate. J Am Acad Dermatol. 2006;55:786-793.
11. Kim H, Zhen DB, Lieske JC, et al. Treatment of cholesterol embolization syndrome in the setting of an acute indication for anticoagulation therapy. J Med Cases. 2014;5:376-379.
12. Igarashi Y, Akimoto T, Kobayashi T, et al. Performing anticoagulation: a puzzling case of cholesterol embolization syndrome. Clin Med Insights Case Rep. 2017;10:1179547616684649. doi:10.1177/1179547616684649.
Quitting smoking after MI has huge benefits in young adults
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Young adult smokers who stop smoking in the first year after an initial myocardial infarction are far less likely to die over the next 10 years than their peers who continue to smoke. Yet nearly two-thirds keep smoking after the event, according to new data from the Partners YOUNG-MI Registry.
“Smoking is one of the most common risk factors for developing an MI at a young age. ... This reinforces the need to have more young individuals avoid, or quit, the use of tobacco,” Ron Blankstein, MD, Brigham and Women’s Hospital and Harvard Medical School, Boston, said in an interview.
Yet, the finding that 62% of young adults continue to smoke 1 year after MI points to an “enormous need for better smoking cessation efforts following a heart attack,” he said.
“Powerful” message for clinicians
“This study joins an incredibly powerful body of evidence that says if you quit smoking, you’re going to live longer,” said Michael Fiore, MD, MPH, MBA, director of the University of Wisconsin Center for Tobacco Research and Intervention, Madison, who wasn’t involved in the study.
“As physicians, there is nothing we can do that will have a greater impact for our patients than quitting smoking. The study is a powerful call for clinicians to intervene with their patients that smoke – both if you have an MI or if you don’t,” Dr. Fiore told this news organization.
The study involved 2,072 individuals 50 years or younger (median age, 45 years; 81% male) who were hospitalized for an initial MI at two large academic medical centers in Boston. Of these, 33.9% were never-smokers, 13.6% were former smokers, and 52.5% were smokers at the time of their MI.
During a median follow-up of 10.2 years, those who quit smoking had a significantly lower rate of death from any cause (unadjusted hazard ratio, 0.35; 95% confidence interval, 0.19-0.63; P < .001) and a cardiovascular cause (HR, 0.29; 95% CI, 0.11-0.79; P = .02), relative to those who continued to smoke.
The results remained statistically significant in a propensity-matched analysis for both all-cause (HR, 0.30; 95% CI, 0.16-0.56; P < .001) and CV mortality (HR, 0.19; 95% CI, 0.06-0.56; P = .003).
“Although patients who quit smoking were similar to those who continued to smoke with respect to their baseline characteristics, smoking cessation was associated with an approximate 70%-80% reduction in all-cause and CV mortality,” the authors note in their article, published online July 8 in JAMA Network Open.
They say it’s also noteworthy that long-term death rates of never-smokers and former smokers who quit before the MI were nearly identical.
‘A failure of our health care system’
The bottom line, said Dr. Blankstein, is that it is “never too late to quit, and those who experience an MI should do so right away. Our health care system must help promote such efforts, as there is immense room for improvement.”
Dr. Fiore said: “When I see an article like this, it just reminds me that, if you’re really thinking about staying healthy, there is nothing better you can do to improve the quality and longevity of your life than quitting smoking.”
The observation that many patients continue to smoke after MI is a “failure of our health care system, and it’s an individual failure in that these individuals are not able to overcome their powerful nicotine dependence. It’s an unfortunate occurrence that’s resulting in unnecessary deaths,” said Dr. Fiore.
There is no “magic bullet” to overcome nicotine addiction, but there are approved treatments that can “substantially boost quit rates,” he noted.
The two most effective smoking-cessation treatments are varenicline (Chantix) and combination nicotine replacement therapy, a patch combined ideally with nicotine mini lozenges, particularly when combined with some brief counseling, said Fiore.
He encourages cardiologists to get their patients to commit to quitting and then link them to resources such as 1-800-QUIT-NOW or SmokeFree.gov.
Funding for the study was provided by grants from the National Heart, Lung, and Blood Institute. Dr. Blankstein reported receiving research support from Amgen and Astellas. Dr. Fiore had no relevant disclosures.
A version of this article originally appeared on Medscape.com.
CMS to broaden transcatheter mitral valve repair coverage, and change its name
The first order of business in the long-awaited, recently released Centers for Medicare & Medicaid Services (CMS) proposed national coverage decision (NCD) for transcatheter mitral valve repair (TMVR) was to get rid of its familiar moniker.
The document tosses the term TMVR in favor of transcatheter edge-to-edge repair (TEER) “to more precisely define the treatment addressed in this NCD” and differentiate it from other therapies that repair or replace the mitral valve.
(In an off-the-cuff Twitter poll launched right after the CMS document’s release, 80.3% of respondents answered that they “hate” the new acronym and the remainder said they “love” it; those two were the poll’s only choices.)
The NCD proposal goes on to say that CMS coverage of TEER would expand to include treatment of symptomatic moderate-to-severe or severe functional mitral regurgitation (MR) when used with maximally tolerated guideline-directed medical therapy.
The proposed NCD has been expected since March 2019 when the US Food and Drug Administration (FDA) approved the MitraClip (Abbott Vascular) for secondary functional MR. Medicare has covered MitraClip for primary degenerative MR since 2014.
Abbott announced in October 2019 that it would ramp up production of the MitraClip, which is currently the only FDA-approved TEER device.
Further specifications
Even as the new proposed NCD would add CMS coverage for functional MR, it would also decline a coverage statement for degenerative MR. Instead, it proposes to leave such coverage decisions to local Medical Administrative Contractors (MACs), given a relatively low incidence of clip intervention for degenerative MR. Less than 1% of the Medicare population undergo TEER of the mitral valve for that indication, the document says.
“The MACs are structured to be able to take into account local patient, physician, and institutional factors, which are especially important when overall prevalence is very low.”
The proposal also emphasizes that patients undergoing such covered TEER procedures be “under the care of a heart failure physician specialist experienced in the care and treatment of mitral valve disease,» with additional care provided by a heart team that includes a cardiac surgeon, interventional cardiologist, interventional echocardiographer.
The new document is generally consistent with a Consensus Statement from the American Association for Thoracic Surgery, the American College of Cardiology (ACC), the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons published in December 2019 and covered then by theheart.org / Medscape Cardiology.
In anticipation the CMS coverage proposal, the ACC earlier this year published a Focused Update of the 2017 Expert Consensus Decision Pathway on the Management of Mitral Regurgitation to reflect new evidence in the field, mainly the recent clinical trial data on functional MR from the MITRA-FR and COAPT trials.
“The proposed criteria are nicely guided by the multisociety consensus document, which sought to foster optimal patient outcomes while also maintaining access to TEER,” Sammy Elmariah, MD, MPH, from Massachusetts General Hospital in Boston, commented by email.
“These criteria, in conjunction with results of the COAPT trial, establish TEER as the standard of care for patients with symptomatic functional MR despite guideline-directed medical therapy who do not possess an alternative indication for cardiac surgery,” said Elmariah, a coauthor on both the Consensus Statement and the Focused Update.
The proposed NCD seems “reasonable,” cardiothoracic surgeon Michael J. Reardon, MD, Houston Methodist Hospital, said by email. But he thought there might be some objections to the requirement for TEER centers to have a surgery program with a minimum annual volume for mitral-valve surgeries.
The proposed NCD says a hospital must have “a surgical program that performs ≥25 total mitral valve surgical procedures for severe MR per year, of which at least 10 must be mitral valve repairs.”
“There is a very definite relationship between mitral valve surgery volume and surgical outcomes and between TEER volume and TEER outcomes, but no real relationship between mitral valve surgery volumes and TEER outcomes,” Reardon said. “A mitral valve surgery program is important, but how many cases do you need to be able to start and run a TEER program?”
Edwards Lifesciences is currently testing its own device for TEER: the PASCAL transcatheter mitral valve repair system. Early findings from the company’s ongoing CLASP IID trial, a head-to-head comparison of Pascal and MitraClip, are expected in December 2023.
CMS is seeking comments on the proposed national coverage determination, and will render a final decision within 60 days of the end of the 30-day public comment period.
Elmariah discloses receiving research grants from the American Heart Association, the National Institutes of Health, Edwards Lifesciences, Svelte Medical, and Medtronic, and consulting fees from AstraZeneca. Reardon recently reported no relevant conflicts of interest.
This article first appeared on Medscape.com.
The first order of business in the long-awaited, recently released Centers for Medicare & Medicaid Services (CMS) proposed national coverage decision (NCD) for transcatheter mitral valve repair (TMVR) was to get rid of its familiar moniker.
The document tosses the term TMVR in favor of transcatheter edge-to-edge repair (TEER) “to more precisely define the treatment addressed in this NCD” and differentiate it from other therapies that repair or replace the mitral valve.
(In an off-the-cuff Twitter poll launched right after the CMS document’s release, 80.3% of respondents answered that they “hate” the new acronym and the remainder said they “love” it; those two were the poll’s only choices.)
The NCD proposal goes on to say that CMS coverage of TEER would expand to include treatment of symptomatic moderate-to-severe or severe functional mitral regurgitation (MR) when used with maximally tolerated guideline-directed medical therapy.
The proposed NCD has been expected since March 2019 when the US Food and Drug Administration (FDA) approved the MitraClip (Abbott Vascular) for secondary functional MR. Medicare has covered MitraClip for primary degenerative MR since 2014.
Abbott announced in October 2019 that it would ramp up production of the MitraClip, which is currently the only FDA-approved TEER device.
Further specifications
Even as the new proposed NCD would add CMS coverage for functional MR, it would also decline a coverage statement for degenerative MR. Instead, it proposes to leave such coverage decisions to local Medical Administrative Contractors (MACs), given a relatively low incidence of clip intervention for degenerative MR. Less than 1% of the Medicare population undergo TEER of the mitral valve for that indication, the document says.
“The MACs are structured to be able to take into account local patient, physician, and institutional factors, which are especially important when overall prevalence is very low.”
The proposal also emphasizes that patients undergoing such covered TEER procedures be “under the care of a heart failure physician specialist experienced in the care and treatment of mitral valve disease,» with additional care provided by a heart team that includes a cardiac surgeon, interventional cardiologist, interventional echocardiographer.
The new document is generally consistent with a Consensus Statement from the American Association for Thoracic Surgery, the American College of Cardiology (ACC), the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons published in December 2019 and covered then by theheart.org / Medscape Cardiology.
In anticipation the CMS coverage proposal, the ACC earlier this year published a Focused Update of the 2017 Expert Consensus Decision Pathway on the Management of Mitral Regurgitation to reflect new evidence in the field, mainly the recent clinical trial data on functional MR from the MITRA-FR and COAPT trials.
“The proposed criteria are nicely guided by the multisociety consensus document, which sought to foster optimal patient outcomes while also maintaining access to TEER,” Sammy Elmariah, MD, MPH, from Massachusetts General Hospital in Boston, commented by email.
“These criteria, in conjunction with results of the COAPT trial, establish TEER as the standard of care for patients with symptomatic functional MR despite guideline-directed medical therapy who do not possess an alternative indication for cardiac surgery,” said Elmariah, a coauthor on both the Consensus Statement and the Focused Update.
The proposed NCD seems “reasonable,” cardiothoracic surgeon Michael J. Reardon, MD, Houston Methodist Hospital, said by email. But he thought there might be some objections to the requirement for TEER centers to have a surgery program with a minimum annual volume for mitral-valve surgeries.
The proposed NCD says a hospital must have “a surgical program that performs ≥25 total mitral valve surgical procedures for severe MR per year, of which at least 10 must be mitral valve repairs.”
“There is a very definite relationship between mitral valve surgery volume and surgical outcomes and between TEER volume and TEER outcomes, but no real relationship between mitral valve surgery volumes and TEER outcomes,” Reardon said. “A mitral valve surgery program is important, but how many cases do you need to be able to start and run a TEER program?”
Edwards Lifesciences is currently testing its own device for TEER: the PASCAL transcatheter mitral valve repair system. Early findings from the company’s ongoing CLASP IID trial, a head-to-head comparison of Pascal and MitraClip, are expected in December 2023.
CMS is seeking comments on the proposed national coverage determination, and will render a final decision within 60 days of the end of the 30-day public comment period.
Elmariah discloses receiving research grants from the American Heart Association, the National Institutes of Health, Edwards Lifesciences, Svelte Medical, and Medtronic, and consulting fees from AstraZeneca. Reardon recently reported no relevant conflicts of interest.
This article first appeared on Medscape.com.
The first order of business in the long-awaited, recently released Centers for Medicare & Medicaid Services (CMS) proposed national coverage decision (NCD) for transcatheter mitral valve repair (TMVR) was to get rid of its familiar moniker.
The document tosses the term TMVR in favor of transcatheter edge-to-edge repair (TEER) “to more precisely define the treatment addressed in this NCD” and differentiate it from other therapies that repair or replace the mitral valve.
(In an off-the-cuff Twitter poll launched right after the CMS document’s release, 80.3% of respondents answered that they “hate” the new acronym and the remainder said they “love” it; those two were the poll’s only choices.)
The NCD proposal goes on to say that CMS coverage of TEER would expand to include treatment of symptomatic moderate-to-severe or severe functional mitral regurgitation (MR) when used with maximally tolerated guideline-directed medical therapy.
The proposed NCD has been expected since March 2019 when the US Food and Drug Administration (FDA) approved the MitraClip (Abbott Vascular) for secondary functional MR. Medicare has covered MitraClip for primary degenerative MR since 2014.
Abbott announced in October 2019 that it would ramp up production of the MitraClip, which is currently the only FDA-approved TEER device.
Further specifications
Even as the new proposed NCD would add CMS coverage for functional MR, it would also decline a coverage statement for degenerative MR. Instead, it proposes to leave such coverage decisions to local Medical Administrative Contractors (MACs), given a relatively low incidence of clip intervention for degenerative MR. Less than 1% of the Medicare population undergo TEER of the mitral valve for that indication, the document says.
“The MACs are structured to be able to take into account local patient, physician, and institutional factors, which are especially important when overall prevalence is very low.”
The proposal also emphasizes that patients undergoing such covered TEER procedures be “under the care of a heart failure physician specialist experienced in the care and treatment of mitral valve disease,» with additional care provided by a heart team that includes a cardiac surgeon, interventional cardiologist, interventional echocardiographer.
The new document is generally consistent with a Consensus Statement from the American Association for Thoracic Surgery, the American College of Cardiology (ACC), the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons published in December 2019 and covered then by theheart.org / Medscape Cardiology.
In anticipation the CMS coverage proposal, the ACC earlier this year published a Focused Update of the 2017 Expert Consensus Decision Pathway on the Management of Mitral Regurgitation to reflect new evidence in the field, mainly the recent clinical trial data on functional MR from the MITRA-FR and COAPT trials.
“The proposed criteria are nicely guided by the multisociety consensus document, which sought to foster optimal patient outcomes while also maintaining access to TEER,” Sammy Elmariah, MD, MPH, from Massachusetts General Hospital in Boston, commented by email.
“These criteria, in conjunction with results of the COAPT trial, establish TEER as the standard of care for patients with symptomatic functional MR despite guideline-directed medical therapy who do not possess an alternative indication for cardiac surgery,” said Elmariah, a coauthor on both the Consensus Statement and the Focused Update.
The proposed NCD seems “reasonable,” cardiothoracic surgeon Michael J. Reardon, MD, Houston Methodist Hospital, said by email. But he thought there might be some objections to the requirement for TEER centers to have a surgery program with a minimum annual volume for mitral-valve surgeries.
The proposed NCD says a hospital must have “a surgical program that performs ≥25 total mitral valve surgical procedures for severe MR per year, of which at least 10 must be mitral valve repairs.”
“There is a very definite relationship between mitral valve surgery volume and surgical outcomes and between TEER volume and TEER outcomes, but no real relationship between mitral valve surgery volumes and TEER outcomes,” Reardon said. “A mitral valve surgery program is important, but how many cases do you need to be able to start and run a TEER program?”
Edwards Lifesciences is currently testing its own device for TEER: the PASCAL transcatheter mitral valve repair system. Early findings from the company’s ongoing CLASP IID trial, a head-to-head comparison of Pascal and MitraClip, are expected in December 2023.
CMS is seeking comments on the proposed national coverage determination, and will render a final decision within 60 days of the end of the 30-day public comment period.
Elmariah discloses receiving research grants from the American Heart Association, the National Institutes of Health, Edwards Lifesciences, Svelte Medical, and Medtronic, and consulting fees from AstraZeneca. Reardon recently reported no relevant conflicts of interest.
This article first appeared on Medscape.com.
Used together, troponin and coronary calcium improve CV risk assessment
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
If either high sensitivity cardiac troponin (hs-cTnT) or coronary artery calcium (CAC) are elevated, the 10-year risk of atherosclerotic cardiovascular disease (ASCVD) climbs substantially, which suggests these biomarkers yield more prognostic information when they are used together, according to a cohort study with a median 15 years of follow-up.
Among those with a double negative result, meaning hs-cTnT was less than the limit of detection (<3 ng/L) and the CAC score was zero, only 2.8% developed ASCVD within 10 years, but the rates climbed to 4.6% if hs-cTnT was detectable and to 9.8% if the CAC score exceeded zero even when the other biomarker was negative.
“The increased risk for ASCVD among those with discordant results indicate that their prognostic information is complementary, favoring their conjoined use for risk prediction,” reported a multicenter team of investigators led by Allan S. Jaffe, MD, professor of laboratory medicine and pathology, Mayo Clinic, Rochester, Minn.
The study was performed with data from 6,749 participants in the Multi-Ethnic Study of Atherosclerosis (MESA), which is a longitudinal, community-based study funded by the National Heart, Lung, and Blood Institute. Over the course of long-term follow-up in a patient population that was about half female, 39% non-Hispanic white, 28% Black, 22% Hispanic American, and 12% Asian, ASCVD events were evaluated in relation to both biomarkers measured at baseline.
At baseline, both biomarkers were negative in 22%, both positive in 40%, and discordant in 38%.
After a median follow-up of 15 years, when 1,002 ASCVD events had occurred, the crude rate of ASCVD was 2.8 per 1,000 person-years in the double-negative group. When compared with this, the adjusted hazard ratio for ASCVD among those with double positive biomarkers was 3.5 (P < .00001). Increased risk was also highly significant if just hs-cTnT was positive (HR, 1.59; P = .003) or if just CAC was positive (HR, 2.74; P < .00001).
The added value of using both biomarkers to identify individuals at very low risk of ASCVD makes sense, according to the authors of an accompanying editorial. Written by a team led by John W. McEvoy, MB, BCh, National University of Ireland, Galway, the editorial explained why the information is complementary.
“CAC indicates subclinical atherosclerosis, whereas hs-cTnT indicates myocardial ischemia or damage, not just from coronary stenosis but also due to other conditions like hypertensive heart and left ventricular hypertrophy,” the authors stated.
Although they maintained that adding N-terminal pro-brain natriuretic peptide, which could be drawn from the same blood sample as hs-cTnT, might prove to be an even better but still simple strategy to identify low-risk patients, they praised the concept of combining biomarkers.
“If one’s wish is to identify truly low-risk individuals, then it appears that it takes two negative ASCVD biomarkers to make that wish come true,” the authors of the editorial concluded.
Relative to alternative methods of ASCVD risk assessment, measurement of these biomarkers might be useful for sparing patients from interventions, such as lipid lowering with statin therapy, being considered on the basis of conventional risk factors alone.
Dr. Jaffe said in an interview that he considers the two-biomarker assessment to be a useful tool in the low-risk population that he studied, but he does not consider this strategy as a substitute for other methods, such as those outline in the 2019 ACC/AHA guidelines that address the entire spectrum of risk, although work is planned to see if this approach can be extended to this broader group.*
“The data we have presented now is a good start and suggests that these two objective measures can identify those who are at very low risk and avoid adding individuals who may not be at as low risk if only one of the two tests is used,” Dr. Jaffe explained.
“Given there are now techniques to measure coronary calcium from any chest CT study, and that high sensitivity cardiac troponin is a relatively inexpensive test, putting them together should really help risk stratify patients,” he added.
When asked whether this approach will eventually replace conventional methods of ASCVD risk assessment, such as those proposed in the 2019 American College of Cardiology/American Heart Association guidelines for the primary prevention of cardiovascular disease (Circulation. 2019;140:e596-e646), he said maybe.
“The answer is that we will probe that question in our ongoing studies using continuous data in an attempt to evaluate how to use this approach to risk stratify larger numbers of individuals,” Dr. Jaffe replied.
The senior investigator, Dr. Jaffe, has consulting relationships with many pharmaceutical companies. The editorial authors had no relevant disclosures.
SOURCE: Sandoval Y et al. J Am Coll Cardiol. 2020;76:357-370.
*Correction, 7/27/20: An earlier version of this article mischaracterized Dr. Jaffe's statement.
FROM JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Hot-off-the-press insights on heart failure
Hospitalists frequently encounter patients with heart failure – a complex, clinical syndrome, which has high prevalence, mortality, hospitalization rates, and health care costs.
The HM20 Virtual session “Updates in Heart Failure” will provide literature updates for all types of heart failure patient scenarios – patients with acute and chronic heart failure, those who are hospitalized with heart failure, and patients with heart failure with reduced ejection fraction (HFrEF) and heart failure with preserved ejection fraction (HFpEF). The popular session with questions and answers will be held on Aug. 25.
Presenter Dustin Smith, MD, SFHM, associate professor of medicine in the department of medicine at Emory University, Atlanta, and section chief for education in medical specialty at the Atlanta Veterans Affairs Medical Center, will discuss recent trends, diagnostics, therapeutics, and prognostics for heart failure. He’ll also provide a summary of recent changes to clinical practice guidelines.
“The significance of staying knowledgeable and updated regarding this common admission diagnosis cannot be overstated,” Dr. Smith said. Attendees of this clinical update should learn important practices from new evidence in literature, including an unearthed risk grade predictor of acute heart failure mortality, a diagnostic tool for HFpEF in euvolemic patients with unexplained dyspnea, an examination of the potassium “repletion reflex” in patients hospitalized with heart failure, dietary patterns associated with incident heart failure, and therapies efficacious for HFrEF and/or HFpEF.
“The goal of this session is for attendees to incorporate this new information into their clinical practice so they can optimally manage patients with heart failure,” Dr. Smith said.
The session is specifically curated to impact the clinical practice of hospitalists who provide care for patients with heart failure in the acute care setting and beyond. Key impact areas of clinical practice that will be tackled include:
- Augmenting one’s clinical acumen to diagnose HFpEF.
- Calculating mortality risk for patients with acute heart failure.
- Recognizing other predictors of risk for patients hospitalized with heart failure.
- Recommending dietary, medication, and interventional therapies to prevent future heart failure morbidity and mortality.
Dr. Smith will conclude each literature review with a summary of take-home learning points carefully selected to either change, modify, or confirm the current practice and teaching for providers who care for heart failure patients.
Although Dr. Smith has presented the “Updates in Heart Failure” session in various educational arenas in the past, this is a new update. He has gained vast experience and expertise in this area from conducting extensive and in-depth literature reviews on managing heart failure while preparing for presentations on this topic.
In addition, Dr. Smith has contributed to original research manuscripts, book chapters, and board review–style exam questions in cardiology – including heart failure – and evidence-based medicine topics as an author and editor. He has also sought out additional training and completed faculty development programs targeted at improving his knowledge and skill set to teach evidence-based clinical practice.
Dr. Smith had no relevant financial conflicts to disclose.
Updates in Heart Failure
Live Q&A – Tuesday, Aug. 25 1:00 p.m. to 2:00 p.m.
Hospitalists frequently encounter patients with heart failure – a complex, clinical syndrome, which has high prevalence, mortality, hospitalization rates, and health care costs.
The HM20 Virtual session “Updates in Heart Failure” will provide literature updates for all types of heart failure patient scenarios – patients with acute and chronic heart failure, those who are hospitalized with heart failure, and patients with heart failure with reduced ejection fraction (HFrEF) and heart failure with preserved ejection fraction (HFpEF). The popular session with questions and answers will be held on Aug. 25.
Presenter Dustin Smith, MD, SFHM, associate professor of medicine in the department of medicine at Emory University, Atlanta, and section chief for education in medical specialty at the Atlanta Veterans Affairs Medical Center, will discuss recent trends, diagnostics, therapeutics, and prognostics for heart failure. He’ll also provide a summary of recent changes to clinical practice guidelines.
“The significance of staying knowledgeable and updated regarding this common admission diagnosis cannot be overstated,” Dr. Smith said. Attendees of this clinical update should learn important practices from new evidence in literature, including an unearthed risk grade predictor of acute heart failure mortality, a diagnostic tool for HFpEF in euvolemic patients with unexplained dyspnea, an examination of the potassium “repletion reflex” in patients hospitalized with heart failure, dietary patterns associated with incident heart failure, and therapies efficacious for HFrEF and/or HFpEF.
“The goal of this session is for attendees to incorporate this new information into their clinical practice so they can optimally manage patients with heart failure,” Dr. Smith said.
The session is specifically curated to impact the clinical practice of hospitalists who provide care for patients with heart failure in the acute care setting and beyond. Key impact areas of clinical practice that will be tackled include:
- Augmenting one’s clinical acumen to diagnose HFpEF.
- Calculating mortality risk for patients with acute heart failure.
- Recognizing other predictors of risk for patients hospitalized with heart failure.
- Recommending dietary, medication, and interventional therapies to prevent future heart failure morbidity and mortality.
Dr. Smith will conclude each literature review with a summary of take-home learning points carefully selected to either change, modify, or confirm the current practice and teaching for providers who care for heart failure patients.
Although Dr. Smith has presented the “Updates in Heart Failure” session in various educational arenas in the past, this is a new update. He has gained vast experience and expertise in this area from conducting extensive and in-depth literature reviews on managing heart failure while preparing for presentations on this topic.
In addition, Dr. Smith has contributed to original research manuscripts, book chapters, and board review–style exam questions in cardiology – including heart failure – and evidence-based medicine topics as an author and editor. He has also sought out additional training and completed faculty development programs targeted at improving his knowledge and skill set to teach evidence-based clinical practice.
Dr. Smith had no relevant financial conflicts to disclose.
Updates in Heart Failure
Live Q&A – Tuesday, Aug. 25 1:00 p.m. to 2:00 p.m.
Hospitalists frequently encounter patients with heart failure – a complex, clinical syndrome, which has high prevalence, mortality, hospitalization rates, and health care costs.
The HM20 Virtual session “Updates in Heart Failure” will provide literature updates for all types of heart failure patient scenarios – patients with acute and chronic heart failure, those who are hospitalized with heart failure, and patients with heart failure with reduced ejection fraction (HFrEF) and heart failure with preserved ejection fraction (HFpEF). The popular session with questions and answers will be held on Aug. 25.
Presenter Dustin Smith, MD, SFHM, associate professor of medicine in the department of medicine at Emory University, Atlanta, and section chief for education in medical specialty at the Atlanta Veterans Affairs Medical Center, will discuss recent trends, diagnostics, therapeutics, and prognostics for heart failure. He’ll also provide a summary of recent changes to clinical practice guidelines.
“The significance of staying knowledgeable and updated regarding this common admission diagnosis cannot be overstated,” Dr. Smith said. Attendees of this clinical update should learn important practices from new evidence in literature, including an unearthed risk grade predictor of acute heart failure mortality, a diagnostic tool for HFpEF in euvolemic patients with unexplained dyspnea, an examination of the potassium “repletion reflex” in patients hospitalized with heart failure, dietary patterns associated with incident heart failure, and therapies efficacious for HFrEF and/or HFpEF.
“The goal of this session is for attendees to incorporate this new information into their clinical practice so they can optimally manage patients with heart failure,” Dr. Smith said.
The session is specifically curated to impact the clinical practice of hospitalists who provide care for patients with heart failure in the acute care setting and beyond. Key impact areas of clinical practice that will be tackled include:
- Augmenting one’s clinical acumen to diagnose HFpEF.
- Calculating mortality risk for patients with acute heart failure.
- Recognizing other predictors of risk for patients hospitalized with heart failure.
- Recommending dietary, medication, and interventional therapies to prevent future heart failure morbidity and mortality.
Dr. Smith will conclude each literature review with a summary of take-home learning points carefully selected to either change, modify, or confirm the current practice and teaching for providers who care for heart failure patients.
Although Dr. Smith has presented the “Updates in Heart Failure” session in various educational arenas in the past, this is a new update. He has gained vast experience and expertise in this area from conducting extensive and in-depth literature reviews on managing heart failure while preparing for presentations on this topic.
In addition, Dr. Smith has contributed to original research manuscripts, book chapters, and board review–style exam questions in cardiology – including heart failure – and evidence-based medicine topics as an author and editor. He has also sought out additional training and completed faculty development programs targeted at improving his knowledge and skill set to teach evidence-based clinical practice.
Dr. Smith had no relevant financial conflicts to disclose.
Updates in Heart Failure
Live Q&A – Tuesday, Aug. 25 1:00 p.m. to 2:00 p.m.
Does concurrent use of clopidogrel and PPIs increase CV risk in patients with ACS?
EVIDENCE SUMMARY
A double-blind, double-dummy, placebo-controlled RCT comparing a combination of clopidogrel, aspirin, and omeprazole with clopidogrel, aspirin, and placebo found no increase in composite CV outcomes with the PPI (TABLE).1 Using a PPI did, however, significantly reduce gastrointestinal (GI) bleeding (hazard ratio [HR] = 0.13; 95% confidence interval [CI], 0.03-0.56).Although several meta-analyses have been conducted, they all rely on this single RCT that directly addresses the question, plus post-hoc analyses of other RCTs.
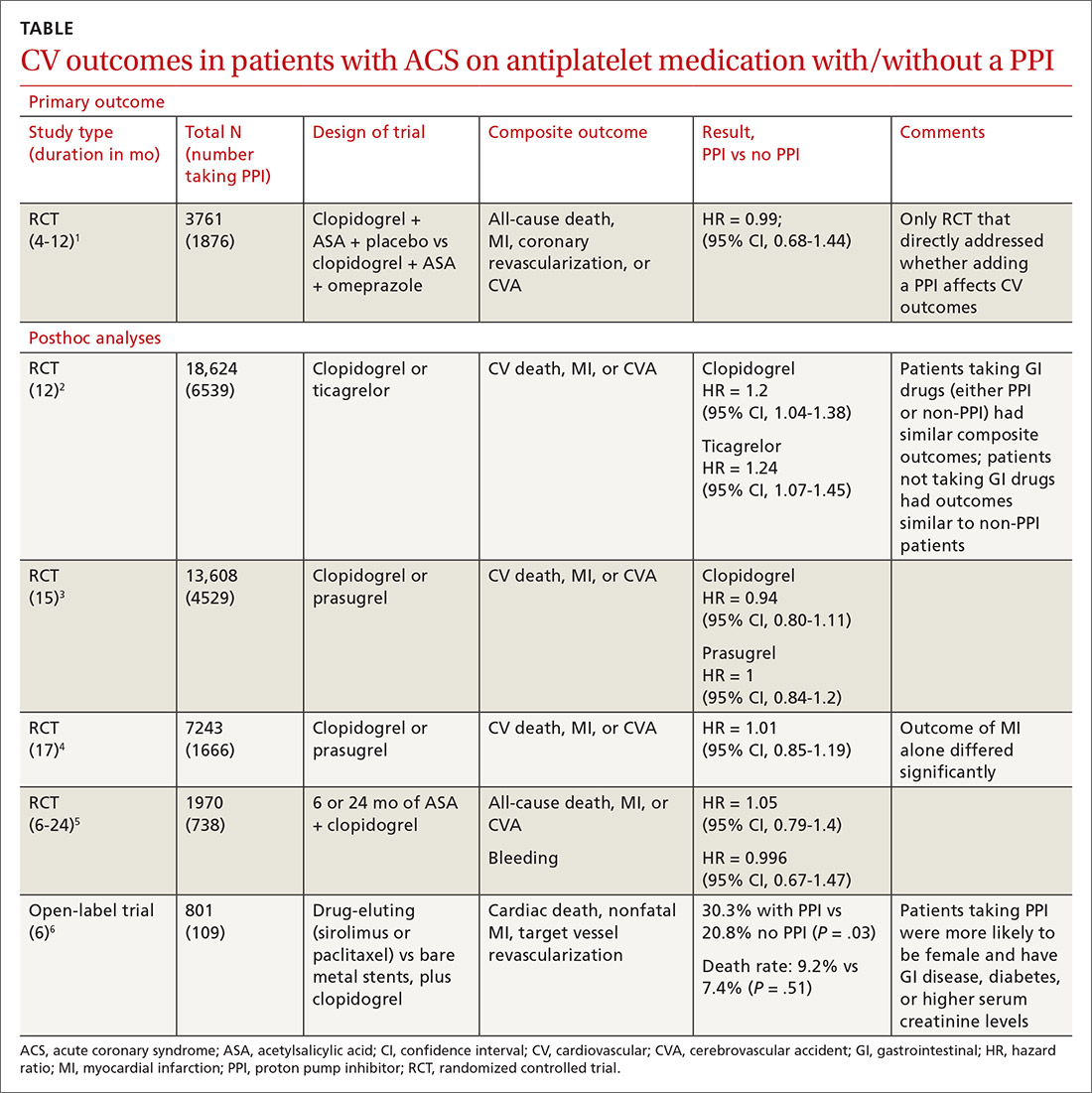
Four of 5 analyses find little or no difference in CV outcomes with a PPI
Four of 5 posthoc analyses (which weren’t themselves randomized) of RCTs found unclear or no differences in composite CV outcomes with concurrent use of a PPI and antiplatelet therapy, after multivariate adjustment for differences in populations taking or not taking a PPI.
Posthoc analysis of the largest study found worse CV outcomes for both clopidogrel and ticagrelor with concomitant PPI use.2 However, patients on any GI drugs (PPI or non-PPI) had composite outcomes similar to patients on a PPI (PPI vs non-PPI GI treatment: HR = 0.98; 95% CI, 0.79-1.23), and patients not taking GI drugs had fewer composite outcomes compared with patients on a PPI (clopidogrel vs no GI therapy: HR = 1.29; 95% CI, 1.12-1.49; ticagrelor vs no GI therapy: HR = 1.30; 95% CI, 1.14-1.49). Researchers postulated that because the rate of composite outcomes increased equally for patients on any GI drug, the higher rate of CV adverse events with a PPI might have been related to GI disease rather than PPI use.
A similar posthoc analysis found no differences with or without PPI use among patients with ACS undergoing planned percutaneous coronary intervention (PCI) and assigned to clopidogrel or prasugrel.3 Researchers performed multivariate adjustment for differences in age, gender, ethnicity, and initial presence of unstable angina/non-ST-elevation MI.
A smaller study also found no significant differences in composite CV outcomes in patients using PPIs.4 Patients did have higher rates of MI (HR = 0.62; 95% CI, 0.42-0.91), but they were more likely to be older and have a previous diagnosis of non-ST-elevation MI, higher incidence of previous coronary artery bypass graft surgery, and history of peptic ulcer disease.
The fourth posthoc analysis of an RCT found that concomitant PPI use (91% of patients on lansoprazole) didn’t alter outcomes among patients undergoing PCI and receiving dual antiplatelet therapy with clopidogrel and aspirin.5 Researchers used a multivariate adjustment for differences in age, gender, and renal function and found no difference in outcomes during the 6-month or 24-month period. PPI prescription was at physician discretion. Researchers didn’t assess for dose-dependent effects of PPI.
A fifth, flawed study finds more adverse events with PPIs
A posthoc analysis of a smaller, open-label trial found increased major adverse cardiac events with PPI use among patients taking clopidogrel after PCI.6 Researchers didn’t adjust for differences in populations at baseline, however, and patients taking PPIs were more likely to be female or older and have diabetes, GI disease, or higher serum creatinine levels.
Continue to: Editor's takeaway
Editor’s takeaway
The best evidence (a large RCT) found that adding a PPI to antiplatelet therapy didn’t alter CV outcomes in patients with ACS, but it did reduce GI bleeds. Hopefully this will give providers the confidence to use PPIs, if clinically indicated, in patients taking antiplatelet therapy with clopidogrel or prasugrel.
1. Bhatt DL, Cryer BL, Contant CF, et al. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med. 2010;363:1909-1917.
2. Goodman SG, Clare R, Pieper KS, et al. Association of proton pump inhibitor use on cardiovascular outcomes with clopidogrel and ticagrelor: insights from the platelet inhibition and patient outcomes trial. Circulation. 2012;125:978-986.
3. O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet. 2009;374:989-997.
4. Nicolau JC, Bhatt DL, Roe MT, et al. Concomitant proton-pump inhibitor use, platelet activity, and clinical outcomes in patients with acute coronary syndromes treated with prasugrel versus clopidogrel and managed without revascularization: insights from the Targeted Platelet Inhibition to Clarify the Optimal Strategy to Medically Manage Acute Coronary Syndromes trial. Am Heart J. 2015;170:683-694.e3.
5. Gargiulo G, Costa F, Ariotti S, et al. Impact of proton pump inhibitors on clinical outcomes in patients treated with a 6- or 24-month dual-antiplatelet therapy duration: insights from the PROlonging Dual-antiplatelet treatment after Grading stent-induced Intimal hyperplasia studY trial. Am Heart J. 2016;174:95-102.
6. Burkard T, Kaiser CA, Brunner-La Rocca H, et al. Combined clopidogrel and proton pump inhibitor therapy is associated with higher cardiovascular event rates after percutaneous coronary intervention: a report from the BASKET trial. J Intern Med. 2012;271:257-263.
EVIDENCE SUMMARY
A double-blind, double-dummy, placebo-controlled RCT comparing a combination of clopidogrel, aspirin, and omeprazole with clopidogrel, aspirin, and placebo found no increase in composite CV outcomes with the PPI (TABLE).1 Using a PPI did, however, significantly reduce gastrointestinal (GI) bleeding (hazard ratio [HR] = 0.13; 95% confidence interval [CI], 0.03-0.56).Although several meta-analyses have been conducted, they all rely on this single RCT that directly addresses the question, plus post-hoc analyses of other RCTs.

Four of 5 analyses find little or no difference in CV outcomes with a PPI
Four of 5 posthoc analyses (which weren’t themselves randomized) of RCTs found unclear or no differences in composite CV outcomes with concurrent use of a PPI and antiplatelet therapy, after multivariate adjustment for differences in populations taking or not taking a PPI.
Posthoc analysis of the largest study found worse CV outcomes for both clopidogrel and ticagrelor with concomitant PPI use.2 However, patients on any GI drugs (PPI or non-PPI) had composite outcomes similar to patients on a PPI (PPI vs non-PPI GI treatment: HR = 0.98; 95% CI, 0.79-1.23), and patients not taking GI drugs had fewer composite outcomes compared with patients on a PPI (clopidogrel vs no GI therapy: HR = 1.29; 95% CI, 1.12-1.49; ticagrelor vs no GI therapy: HR = 1.30; 95% CI, 1.14-1.49). Researchers postulated that because the rate of composite outcomes increased equally for patients on any GI drug, the higher rate of CV adverse events with a PPI might have been related to GI disease rather than PPI use.
A similar posthoc analysis found no differences with or without PPI use among patients with ACS undergoing planned percutaneous coronary intervention (PCI) and assigned to clopidogrel or prasugrel.3 Researchers performed multivariate adjustment for differences in age, gender, ethnicity, and initial presence of unstable angina/non-ST-elevation MI.
A smaller study also found no significant differences in composite CV outcomes in patients using PPIs.4 Patients did have higher rates of MI (HR = 0.62; 95% CI, 0.42-0.91), but they were more likely to be older and have a previous diagnosis of non-ST-elevation MI, higher incidence of previous coronary artery bypass graft surgery, and history of peptic ulcer disease.
The fourth posthoc analysis of an RCT found that concomitant PPI use (91% of patients on lansoprazole) didn’t alter outcomes among patients undergoing PCI and receiving dual antiplatelet therapy with clopidogrel and aspirin.5 Researchers used a multivariate adjustment for differences in age, gender, and renal function and found no difference in outcomes during the 6-month or 24-month period. PPI prescription was at physician discretion. Researchers didn’t assess for dose-dependent effects of PPI.
A fifth, flawed study finds more adverse events with PPIs
A posthoc analysis of a smaller, open-label trial found increased major adverse cardiac events with PPI use among patients taking clopidogrel after PCI.6 Researchers didn’t adjust for differences in populations at baseline, however, and patients taking PPIs were more likely to be female or older and have diabetes, GI disease, or higher serum creatinine levels.
Continue to: Editor's takeaway
Editor’s takeaway
The best evidence (a large RCT) found that adding a PPI to antiplatelet therapy didn’t alter CV outcomes in patients with ACS, but it did reduce GI bleeds. Hopefully this will give providers the confidence to use PPIs, if clinically indicated, in patients taking antiplatelet therapy with clopidogrel or prasugrel.
EVIDENCE SUMMARY
A double-blind, double-dummy, placebo-controlled RCT comparing a combination of clopidogrel, aspirin, and omeprazole with clopidogrel, aspirin, and placebo found no increase in composite CV outcomes with the PPI (TABLE).1 Using a PPI did, however, significantly reduce gastrointestinal (GI) bleeding (hazard ratio [HR] = 0.13; 95% confidence interval [CI], 0.03-0.56).Although several meta-analyses have been conducted, they all rely on this single RCT that directly addresses the question, plus post-hoc analyses of other RCTs.

Four of 5 analyses find little or no difference in CV outcomes with a PPI
Four of 5 posthoc analyses (which weren’t themselves randomized) of RCTs found unclear or no differences in composite CV outcomes with concurrent use of a PPI and antiplatelet therapy, after multivariate adjustment for differences in populations taking or not taking a PPI.
Posthoc analysis of the largest study found worse CV outcomes for both clopidogrel and ticagrelor with concomitant PPI use.2 However, patients on any GI drugs (PPI or non-PPI) had composite outcomes similar to patients on a PPI (PPI vs non-PPI GI treatment: HR = 0.98; 95% CI, 0.79-1.23), and patients not taking GI drugs had fewer composite outcomes compared with patients on a PPI (clopidogrel vs no GI therapy: HR = 1.29; 95% CI, 1.12-1.49; ticagrelor vs no GI therapy: HR = 1.30; 95% CI, 1.14-1.49). Researchers postulated that because the rate of composite outcomes increased equally for patients on any GI drug, the higher rate of CV adverse events with a PPI might have been related to GI disease rather than PPI use.
A similar posthoc analysis found no differences with or without PPI use among patients with ACS undergoing planned percutaneous coronary intervention (PCI) and assigned to clopidogrel or prasugrel.3 Researchers performed multivariate adjustment for differences in age, gender, ethnicity, and initial presence of unstable angina/non-ST-elevation MI.
A smaller study also found no significant differences in composite CV outcomes in patients using PPIs.4 Patients did have higher rates of MI (HR = 0.62; 95% CI, 0.42-0.91), but they were more likely to be older and have a previous diagnosis of non-ST-elevation MI, higher incidence of previous coronary artery bypass graft surgery, and history of peptic ulcer disease.
The fourth posthoc analysis of an RCT found that concomitant PPI use (91% of patients on lansoprazole) didn’t alter outcomes among patients undergoing PCI and receiving dual antiplatelet therapy with clopidogrel and aspirin.5 Researchers used a multivariate adjustment for differences in age, gender, and renal function and found no difference in outcomes during the 6-month or 24-month period. PPI prescription was at physician discretion. Researchers didn’t assess for dose-dependent effects of PPI.
A fifth, flawed study finds more adverse events with PPIs
A posthoc analysis of a smaller, open-label trial found increased major adverse cardiac events with PPI use among patients taking clopidogrel after PCI.6 Researchers didn’t adjust for differences in populations at baseline, however, and patients taking PPIs were more likely to be female or older and have diabetes, GI disease, or higher serum creatinine levels.
Continue to: Editor's takeaway
Editor’s takeaway
The best evidence (a large RCT) found that adding a PPI to antiplatelet therapy didn’t alter CV outcomes in patients with ACS, but it did reduce GI bleeds. Hopefully this will give providers the confidence to use PPIs, if clinically indicated, in patients taking antiplatelet therapy with clopidogrel or prasugrel.
1. Bhatt DL, Cryer BL, Contant CF, et al. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med. 2010;363:1909-1917.
2. Goodman SG, Clare R, Pieper KS, et al. Association of proton pump inhibitor use on cardiovascular outcomes with clopidogrel and ticagrelor: insights from the platelet inhibition and patient outcomes trial. Circulation. 2012;125:978-986.
3. O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet. 2009;374:989-997.
4. Nicolau JC, Bhatt DL, Roe MT, et al. Concomitant proton-pump inhibitor use, platelet activity, and clinical outcomes in patients with acute coronary syndromes treated with prasugrel versus clopidogrel and managed without revascularization: insights from the Targeted Platelet Inhibition to Clarify the Optimal Strategy to Medically Manage Acute Coronary Syndromes trial. Am Heart J. 2015;170:683-694.e3.
5. Gargiulo G, Costa F, Ariotti S, et al. Impact of proton pump inhibitors on clinical outcomes in patients treated with a 6- or 24-month dual-antiplatelet therapy duration: insights from the PROlonging Dual-antiplatelet treatment after Grading stent-induced Intimal hyperplasia studY trial. Am Heart J. 2016;174:95-102.
6. Burkard T, Kaiser CA, Brunner-La Rocca H, et al. Combined clopidogrel and proton pump inhibitor therapy is associated with higher cardiovascular event rates after percutaneous coronary intervention: a report from the BASKET trial. J Intern Med. 2012;271:257-263.
1. Bhatt DL, Cryer BL, Contant CF, et al. Clopidogrel with or without omeprazole in coronary artery disease. N Engl J Med. 2010;363:1909-1917.
2. Goodman SG, Clare R, Pieper KS, et al. Association of proton pump inhibitor use on cardiovascular outcomes with clopidogrel and ticagrelor: insights from the platelet inhibition and patient outcomes trial. Circulation. 2012;125:978-986.
3. O’Donoghue ML, Braunwald E, Antman EM, et al. Pharmacodynamic effect and clinical efficacy of clopidogrel and prasugrel with or without a proton-pump inhibitor: an analysis of two randomised trials. Lancet. 2009;374:989-997.
4. Nicolau JC, Bhatt DL, Roe MT, et al. Concomitant proton-pump inhibitor use, platelet activity, and clinical outcomes in patients with acute coronary syndromes treated with prasugrel versus clopidogrel and managed without revascularization: insights from the Targeted Platelet Inhibition to Clarify the Optimal Strategy to Medically Manage Acute Coronary Syndromes trial. Am Heart J. 2015;170:683-694.e3.
5. Gargiulo G, Costa F, Ariotti S, et al. Impact of proton pump inhibitors on clinical outcomes in patients treated with a 6- or 24-month dual-antiplatelet therapy duration: insights from the PROlonging Dual-antiplatelet treatment after Grading stent-induced Intimal hyperplasia studY trial. Am Heart J. 2016;174:95-102.
6. Burkard T, Kaiser CA, Brunner-La Rocca H, et al. Combined clopidogrel and proton pump inhibitor therapy is associated with higher cardiovascular event rates after percutaneous coronary intervention: a report from the BASKET trial. J Intern Med. 2012;271:257-263.
EVIDENCE-BASED ANSWER:
No. Adding a proton pump inhibitor (PPI) in patients taking antiplatelet medications such as clopidogrel for acute coronary syndrome (ACS) doesn’t increase the composite risk of cardiovascular (CV) events: CV death, myocardial infarction (MI), and cerebrovascular accident (CVA) (strength of recommendation: B, randomized, controlled trial [RCT] and prepon-derance of posthoc analyses of large RCTs).
Psoriatic disease inflammation linked to heart failure
Patients with psoriatic disease are known to be at increased risk of heart failure. A new cohort study suggests that part of the risk may be attributable to the disease itself, not just traditional cardiovascular risk factors like obesity and metabolic abnormalities that are common comorbidities in psoriatic disease. There may also be differences in the risk profiles of patients with ischemic and nonischemic heart failure.
Previous studies have shown that heart failure risk in patients with psoriatic arthritis is 32% higher than in the general population, and with psoriasis, it is 22%-53% higher. However, those studies were based on administrative databases with no clinical information to back up the accuracy of diagnoses, Sahil Koppikar, MD, from the University of Toronto, said during a presentation of the research at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA).
The finding that psoriatic disease inflammation may be a direct risk factor for heart failure might be good news for patients. “By controlling inflammation, we may be able to reduce the risk of heart failure in these patients,” Dr. Koppikar said.
During a question and answer session, discussant Deepak Jadon, MBChB, PhD, director of the rheumatology research unit and lead for psoriatic arthritis at Addenbrooke’s Hospital, Cambridge (England), noted that patients with conditions like lupus and systemic sclerosis may undergo regular echocardiograms, chest CTs, or other surveillance, and asked if Dr. Koppikar could recommend a framework for similar surveillance in psoriatic arthritis.
“With the current data we have, I don’t know if we can make recommendations. What we learned from our study is that patients that have elevated inflammatory disease, with elevated inflammatory markers for a prolonged period of time, were at higher risk than [if they had elevated markers only] just before the event. So poorly controlled patients might be something you should be more aware of, and maybe get cardiology involved. But I don’t think it’s something we should be doing right now for all patients,” Dr. Koppikar said.
The researchers analyzed data from a psoriasis cohort at the University of Toronto that began in 2006. Every 6-12 months, they were assessed by a rheumatologist and underwent imaging assessment and laboratory tests. The primary outcome of the study was the first heart failure event, which the researchers identified by linking the cohort database with provincial hospitalization and mortality databases. They verified all events by examining medical records. They also assessed the association between heart failure and disease activity over time rather than just before the event.
The analysis included 1,994 patients. A total of 64 new heart failure events occurred during a mean follow-up of 11.3 years (2.85 per 1,000 person-years), including 38 ischemic and 26 nonischemic events. A multivariate analysis found that heart failure was associated with adjusted mean (AM) tender joint count (hazard ratio, 1.51; P = .02), AM swollen joint count (HR, 1.82; P = .04), AM erythrocyte sedimentation rate (HR, 1.26; P = .009), AM C-reactive protein (HR, 1.27; P = .001), Health Assessment Questionnaire (HR, 1.95; P = .001), and minimum disease activity state (HR, 0.40; P = .04). The multivariate analysis was adjusted for sex, hypertension, diabetes mellitus, body mass index, ischemic heart disease, lipids, and smoking status.
When the researchers separated the analysis into ischemic and nonischemic heart failure, some interesting associations popped out. Nonischemic heart failure was associated with AM tender joint count (HR, 1.83; P = .004), but ischemic heart failure was not. Other factors associated with nonischemic but not ischemic heart failure included AM swollen joint count (HR, 3.56; P = .0003), damaged joint count (HR, 1.29; P = .04), and pain score (HR, 1.22; P = .047). Minimum disease activity had the opposite result: It was associated with only ischemic heart failure (HR, 0.40; P = .04).
The study cohort more closely resembles a rheumatology cohort than a dermatology cohort, and it suggests that patients with psoriatic arthritis have different cardiovascular comorbidities than those with pure psoriasis, according to Diamant Thaçi, MD, PhD, professor and chairman of the department of dermatology at the University of Lübeck (Germany). “It shows how it important it is to look for comorbidity in the rheumatologic setting,” Dr. Thaçi said in an interview.
The study was supported by the Arthritis Society. Dr. Koppikar and Dr. Thaçi have no relevant financial disclosures.
SOURCE: Koppikar S et al. GRAPPA 2020 Virtual Annual Meeting.
Patients with psoriatic disease are known to be at increased risk of heart failure. A new cohort study suggests that part of the risk may be attributable to the disease itself, not just traditional cardiovascular risk factors like obesity and metabolic abnormalities that are common comorbidities in psoriatic disease. There may also be differences in the risk profiles of patients with ischemic and nonischemic heart failure.
Previous studies have shown that heart failure risk in patients with psoriatic arthritis is 32% higher than in the general population, and with psoriasis, it is 22%-53% higher. However, those studies were based on administrative databases with no clinical information to back up the accuracy of diagnoses, Sahil Koppikar, MD, from the University of Toronto, said during a presentation of the research at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA).
The finding that psoriatic disease inflammation may be a direct risk factor for heart failure might be good news for patients. “By controlling inflammation, we may be able to reduce the risk of heart failure in these patients,” Dr. Koppikar said.
During a question and answer session, discussant Deepak Jadon, MBChB, PhD, director of the rheumatology research unit and lead for psoriatic arthritis at Addenbrooke’s Hospital, Cambridge (England), noted that patients with conditions like lupus and systemic sclerosis may undergo regular echocardiograms, chest CTs, or other surveillance, and asked if Dr. Koppikar could recommend a framework for similar surveillance in psoriatic arthritis.
“With the current data we have, I don’t know if we can make recommendations. What we learned from our study is that patients that have elevated inflammatory disease, with elevated inflammatory markers for a prolonged period of time, were at higher risk than [if they had elevated markers only] just before the event. So poorly controlled patients might be something you should be more aware of, and maybe get cardiology involved. But I don’t think it’s something we should be doing right now for all patients,” Dr. Koppikar said.
The researchers analyzed data from a psoriasis cohort at the University of Toronto that began in 2006. Every 6-12 months, they were assessed by a rheumatologist and underwent imaging assessment and laboratory tests. The primary outcome of the study was the first heart failure event, which the researchers identified by linking the cohort database with provincial hospitalization and mortality databases. They verified all events by examining medical records. They also assessed the association between heart failure and disease activity over time rather than just before the event.
The analysis included 1,994 patients. A total of 64 new heart failure events occurred during a mean follow-up of 11.3 years (2.85 per 1,000 person-years), including 38 ischemic and 26 nonischemic events. A multivariate analysis found that heart failure was associated with adjusted mean (AM) tender joint count (hazard ratio, 1.51; P = .02), AM swollen joint count (HR, 1.82; P = .04), AM erythrocyte sedimentation rate (HR, 1.26; P = .009), AM C-reactive protein (HR, 1.27; P = .001), Health Assessment Questionnaire (HR, 1.95; P = .001), and minimum disease activity state (HR, 0.40; P = .04). The multivariate analysis was adjusted for sex, hypertension, diabetes mellitus, body mass index, ischemic heart disease, lipids, and smoking status.
When the researchers separated the analysis into ischemic and nonischemic heart failure, some interesting associations popped out. Nonischemic heart failure was associated with AM tender joint count (HR, 1.83; P = .004), but ischemic heart failure was not. Other factors associated with nonischemic but not ischemic heart failure included AM swollen joint count (HR, 3.56; P = .0003), damaged joint count (HR, 1.29; P = .04), and pain score (HR, 1.22; P = .047). Minimum disease activity had the opposite result: It was associated with only ischemic heart failure (HR, 0.40; P = .04).
The study cohort more closely resembles a rheumatology cohort than a dermatology cohort, and it suggests that patients with psoriatic arthritis have different cardiovascular comorbidities than those with pure psoriasis, according to Diamant Thaçi, MD, PhD, professor and chairman of the department of dermatology at the University of Lübeck (Germany). “It shows how it important it is to look for comorbidity in the rheumatologic setting,” Dr. Thaçi said in an interview.
The study was supported by the Arthritis Society. Dr. Koppikar and Dr. Thaçi have no relevant financial disclosures.
SOURCE: Koppikar S et al. GRAPPA 2020 Virtual Annual Meeting.
Patients with psoriatic disease are known to be at increased risk of heart failure. A new cohort study suggests that part of the risk may be attributable to the disease itself, not just traditional cardiovascular risk factors like obesity and metabolic abnormalities that are common comorbidities in psoriatic disease. There may also be differences in the risk profiles of patients with ischemic and nonischemic heart failure.
Previous studies have shown that heart failure risk in patients with psoriatic arthritis is 32% higher than in the general population, and with psoriasis, it is 22%-53% higher. However, those studies were based on administrative databases with no clinical information to back up the accuracy of diagnoses, Sahil Koppikar, MD, from the University of Toronto, said during a presentation of the research at the virtual annual meeting of the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA).
The finding that psoriatic disease inflammation may be a direct risk factor for heart failure might be good news for patients. “By controlling inflammation, we may be able to reduce the risk of heart failure in these patients,” Dr. Koppikar said.
During a question and answer session, discussant Deepak Jadon, MBChB, PhD, director of the rheumatology research unit and lead for psoriatic arthritis at Addenbrooke’s Hospital, Cambridge (England), noted that patients with conditions like lupus and systemic sclerosis may undergo regular echocardiograms, chest CTs, or other surveillance, and asked if Dr. Koppikar could recommend a framework for similar surveillance in psoriatic arthritis.
“With the current data we have, I don’t know if we can make recommendations. What we learned from our study is that patients that have elevated inflammatory disease, with elevated inflammatory markers for a prolonged period of time, were at higher risk than [if they had elevated markers only] just before the event. So poorly controlled patients might be something you should be more aware of, and maybe get cardiology involved. But I don’t think it’s something we should be doing right now for all patients,” Dr. Koppikar said.
The researchers analyzed data from a psoriasis cohort at the University of Toronto that began in 2006. Every 6-12 months, they were assessed by a rheumatologist and underwent imaging assessment and laboratory tests. The primary outcome of the study was the first heart failure event, which the researchers identified by linking the cohort database with provincial hospitalization and mortality databases. They verified all events by examining medical records. They also assessed the association between heart failure and disease activity over time rather than just before the event.
The analysis included 1,994 patients. A total of 64 new heart failure events occurred during a mean follow-up of 11.3 years (2.85 per 1,000 person-years), including 38 ischemic and 26 nonischemic events. A multivariate analysis found that heart failure was associated with adjusted mean (AM) tender joint count (hazard ratio, 1.51; P = .02), AM swollen joint count (HR, 1.82; P = .04), AM erythrocyte sedimentation rate (HR, 1.26; P = .009), AM C-reactive protein (HR, 1.27; P = .001), Health Assessment Questionnaire (HR, 1.95; P = .001), and minimum disease activity state (HR, 0.40; P = .04). The multivariate analysis was adjusted for sex, hypertension, diabetes mellitus, body mass index, ischemic heart disease, lipids, and smoking status.
When the researchers separated the analysis into ischemic and nonischemic heart failure, some interesting associations popped out. Nonischemic heart failure was associated with AM tender joint count (HR, 1.83; P = .004), but ischemic heart failure was not. Other factors associated with nonischemic but not ischemic heart failure included AM swollen joint count (HR, 3.56; P = .0003), damaged joint count (HR, 1.29; P = .04), and pain score (HR, 1.22; P = .047). Minimum disease activity had the opposite result: It was associated with only ischemic heart failure (HR, 0.40; P = .04).
The study cohort more closely resembles a rheumatology cohort than a dermatology cohort, and it suggests that patients with psoriatic arthritis have different cardiovascular comorbidities than those with pure psoriasis, according to Diamant Thaçi, MD, PhD, professor and chairman of the department of dermatology at the University of Lübeck (Germany). “It shows how it important it is to look for comorbidity in the rheumatologic setting,” Dr. Thaçi said in an interview.
The study was supported by the Arthritis Society. Dr. Koppikar and Dr. Thaçi have no relevant financial disclosures.
SOURCE: Koppikar S et al. GRAPPA 2020 Virtual Annual Meeting.
FROM GRAPPA 2020 VIRTUAL ANNUAL MEETING
Empagliflozin failed to improve exercise capacity in heart failure
Empagliflozin showed favorable effects on diuretic use and congestion symptoms in patients with heart failure with reduced ejection fraction (HFrEF), but the oral sodium glucose cotransporter 2 (SGLT2) inhibitor did not improve the primary endpoint of improved exercise capacity in the EMPERIAL-Reduced trial, investigators reported at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
In the matching EMPERIAL-Preserved trial, conducted in patients with heart failure with preserved ejection fraction (HFpEF), empagliflozin (Jardiance) produced modest improvements in diuretic use, as well as a reduction in unscheduled outpatient visits, compared with placebo-treated controls, although these trends failed to achieve statistical significance. And as in the EMPERIAL-Reduced trial, the SGLT2 inhibitor didn’t move the needle at all on the primary endpoint of improved exercise capacity as measured by 6-minute hall walk distance.
EMPERIAL-Reduced and -Preserved were identically designed, concurrent, phase 3, double-blind, 12-week randomized trials of empagliflozin versus placebo in 312 patients with HFrEF and 315 with HFpEF, defined in EMPERIAL-preserved as a left ventricular ejection fraction above 40%. The majority of participants had type 2 diabetes.
From a baseline median 6-minute walk distance of about 300 meters, the 6-minute walk distance at week 12 was actually 4.0 meters worse in the empagliflozin-treated HFrEF patients than it was in controls and a mere 4.0 meters better than with placebo in empagliflozin-treated patients with HFpEF, reported William T. Abraham, MD, professor of medicine, director of the division of cardiovascular medicine, and associate dean at Ohio State University, Columbus.
He indicated that the audience shouldn’t make too much of the failure to achieve the primary endpoint in the two trials in light of the studies’ major limitations: namely, their relatively small size for purposes of evaluating clinical outcomes and the relatively short 12-week duration.
“In many ways, I would say it’s remarkable that we can observe a positive signal, a favorable signal, in outcomes around congestion. In the case of HFrEF it’s statistically significant, and in HFpEF it’s a trend towards improvement. Of course, there are larger trials ongoing that may confirm these observations. Hopefully the EMPERIAL trials predict a good outcome for those ongoing trials,” Dr. Abraham said.
Piotr Ponikowski, MD, presented the study results for the secondary outcomes of congestion symptoms, diuretic use, and utilization of health care resources. In EMPERIAL-Reduced, intensification of diuretic therapy – often a prelude to acute decompensation and a trip to the hospital – occurred at a rate of 4.5% with empagliflozin and 16.1% with placebo, for a highly significant 73% relative risk reduction. Intensification of loop diuretics occurred in 2.6% of the empagliflozin group and 14.2% of controls, for a 82% risk reduction.
“That’s a pretty significant effect,” observed Dr. Ponikowski, professor of cardiology and head of the department of heart diseases at the Medical University of Wroclaw (Poland).
Moreover, a congestion symptoms score comprising a summary of orthopnea, jugular veinous distention, and edema improved by 47% after 12 weeks on empagliflozin, a statistically significant and clinically meaningful improvement that grew in magnitude over time and at 12 weeks was twice as large, compared with the reduction in placebo group, he added.
There was a trend for fewer unscheduled outpatient visits in the empagliflozin arm of EMPERIAL-Reduced with a rate of 10.4%, compared with 25.8% in controls; however, this 26% reduction in relative risk did not achieve statistical significance.
Intensification of loop diuretics occurred in 9% of EMPERIAL-Preserved participants on empagliflozin and 13.5% on placebo, but this 34% reduction in risk didn’t reach significance.
Adverse events in the EMPERIAL trials were similar across the active treatment and placebo arms. The benign safety profile was similar to what was seen in the earlier major clinical trials of empagliflozin for treatment of type 2 diabetes.
Session chair Stephane Heymans, MD, PhD, of the University of Maastricht (the Netherlands) noted that a substantial minority of patients in EMPERIAL-Reduced were on the combined neprilysin inhibitor sacubitril and the angiotensin receptor blocker valsartan (Entresto), whereas far fewer were in EMPERIAL-Preserved. He wondered if this greater use of background sacubitril/valsartan could explain empagliflozin’s greater efficacy in EMPERIAL-Reduced.
Highly unlikely, according to the investigators.
“It looks like, as is the case with most of our heart failure therapies, that we do see incremental value here. If you met the criteria for these trials, it appears you derived benefit from empagliflozin regardless of whether you were on an angiotensin receptor neprilysin inhibitor or not. I think that speaks to the incremental benefit of SGLT2 inhibitors on top of current guideline-directed medical therapy,” Dr. Abraham said.
Dr. Ponikowski observed that the same point was underscored in the DAPA-HF trial of the SGLT2 inhibitor dapagliflozin (Farxiga) in patients with heart failure (DAPA-HF: N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“You’ll see that the mortality and morbidity and quality-of-life benefit is in those treated with dapagliflozin with or without angiotensin receptor neprilysin inhibition; so, regardless of background therapy. And the effect is especially clear in patients on both therapies,” the cardiologist said.
The EMPERIAL trials were sponsored by Boehringer Ingelheim. Dr. Abraham and Dr. Ponikowksi reported receiving consultant fees from the company for serving on the trials’ executive committee.
Empagliflozin showed favorable effects on diuretic use and congestion symptoms in patients with heart failure with reduced ejection fraction (HFrEF), but the oral sodium glucose cotransporter 2 (SGLT2) inhibitor did not improve the primary endpoint of improved exercise capacity in the EMPERIAL-Reduced trial, investigators reported at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
In the matching EMPERIAL-Preserved trial, conducted in patients with heart failure with preserved ejection fraction (HFpEF), empagliflozin (Jardiance) produced modest improvements in diuretic use, as well as a reduction in unscheduled outpatient visits, compared with placebo-treated controls, although these trends failed to achieve statistical significance. And as in the EMPERIAL-Reduced trial, the SGLT2 inhibitor didn’t move the needle at all on the primary endpoint of improved exercise capacity as measured by 6-minute hall walk distance.
EMPERIAL-Reduced and -Preserved were identically designed, concurrent, phase 3, double-blind, 12-week randomized trials of empagliflozin versus placebo in 312 patients with HFrEF and 315 with HFpEF, defined in EMPERIAL-preserved as a left ventricular ejection fraction above 40%. The majority of participants had type 2 diabetes.
From a baseline median 6-minute walk distance of about 300 meters, the 6-minute walk distance at week 12 was actually 4.0 meters worse in the empagliflozin-treated HFrEF patients than it was in controls and a mere 4.0 meters better than with placebo in empagliflozin-treated patients with HFpEF, reported William T. Abraham, MD, professor of medicine, director of the division of cardiovascular medicine, and associate dean at Ohio State University, Columbus.
He indicated that the audience shouldn’t make too much of the failure to achieve the primary endpoint in the two trials in light of the studies’ major limitations: namely, their relatively small size for purposes of evaluating clinical outcomes and the relatively short 12-week duration.
“In many ways, I would say it’s remarkable that we can observe a positive signal, a favorable signal, in outcomes around congestion. In the case of HFrEF it’s statistically significant, and in HFpEF it’s a trend towards improvement. Of course, there are larger trials ongoing that may confirm these observations. Hopefully the EMPERIAL trials predict a good outcome for those ongoing trials,” Dr. Abraham said.
Piotr Ponikowski, MD, presented the study results for the secondary outcomes of congestion symptoms, diuretic use, and utilization of health care resources. In EMPERIAL-Reduced, intensification of diuretic therapy – often a prelude to acute decompensation and a trip to the hospital – occurred at a rate of 4.5% with empagliflozin and 16.1% with placebo, for a highly significant 73% relative risk reduction. Intensification of loop diuretics occurred in 2.6% of the empagliflozin group and 14.2% of controls, for a 82% risk reduction.
“That’s a pretty significant effect,” observed Dr. Ponikowski, professor of cardiology and head of the department of heart diseases at the Medical University of Wroclaw (Poland).
Moreover, a congestion symptoms score comprising a summary of orthopnea, jugular veinous distention, and edema improved by 47% after 12 weeks on empagliflozin, a statistically significant and clinically meaningful improvement that grew in magnitude over time and at 12 weeks was twice as large, compared with the reduction in placebo group, he added.
There was a trend for fewer unscheduled outpatient visits in the empagliflozin arm of EMPERIAL-Reduced with a rate of 10.4%, compared with 25.8% in controls; however, this 26% reduction in relative risk did not achieve statistical significance.
Intensification of loop diuretics occurred in 9% of EMPERIAL-Preserved participants on empagliflozin and 13.5% on placebo, but this 34% reduction in risk didn’t reach significance.
Adverse events in the EMPERIAL trials were similar across the active treatment and placebo arms. The benign safety profile was similar to what was seen in the earlier major clinical trials of empagliflozin for treatment of type 2 diabetes.
Session chair Stephane Heymans, MD, PhD, of the University of Maastricht (the Netherlands) noted that a substantial minority of patients in EMPERIAL-Reduced were on the combined neprilysin inhibitor sacubitril and the angiotensin receptor blocker valsartan (Entresto), whereas far fewer were in EMPERIAL-Preserved. He wondered if this greater use of background sacubitril/valsartan could explain empagliflozin’s greater efficacy in EMPERIAL-Reduced.
Highly unlikely, according to the investigators.
“It looks like, as is the case with most of our heart failure therapies, that we do see incremental value here. If you met the criteria for these trials, it appears you derived benefit from empagliflozin regardless of whether you were on an angiotensin receptor neprilysin inhibitor or not. I think that speaks to the incremental benefit of SGLT2 inhibitors on top of current guideline-directed medical therapy,” Dr. Abraham said.
Dr. Ponikowski observed that the same point was underscored in the DAPA-HF trial of the SGLT2 inhibitor dapagliflozin (Farxiga) in patients with heart failure (DAPA-HF: N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“You’ll see that the mortality and morbidity and quality-of-life benefit is in those treated with dapagliflozin with or without angiotensin receptor neprilysin inhibition; so, regardless of background therapy. And the effect is especially clear in patients on both therapies,” the cardiologist said.
The EMPERIAL trials were sponsored by Boehringer Ingelheim. Dr. Abraham and Dr. Ponikowksi reported receiving consultant fees from the company for serving on the trials’ executive committee.
Empagliflozin showed favorable effects on diuretic use and congestion symptoms in patients with heart failure with reduced ejection fraction (HFrEF), but the oral sodium glucose cotransporter 2 (SGLT2) inhibitor did not improve the primary endpoint of improved exercise capacity in the EMPERIAL-Reduced trial, investigators reported at the European Society of Cardiology Heart Failure Discoveries virtual meeting.
In the matching EMPERIAL-Preserved trial, conducted in patients with heart failure with preserved ejection fraction (HFpEF), empagliflozin (Jardiance) produced modest improvements in diuretic use, as well as a reduction in unscheduled outpatient visits, compared with placebo-treated controls, although these trends failed to achieve statistical significance. And as in the EMPERIAL-Reduced trial, the SGLT2 inhibitor didn’t move the needle at all on the primary endpoint of improved exercise capacity as measured by 6-minute hall walk distance.
EMPERIAL-Reduced and -Preserved were identically designed, concurrent, phase 3, double-blind, 12-week randomized trials of empagliflozin versus placebo in 312 patients with HFrEF and 315 with HFpEF, defined in EMPERIAL-preserved as a left ventricular ejection fraction above 40%. The majority of participants had type 2 diabetes.
From a baseline median 6-minute walk distance of about 300 meters, the 6-minute walk distance at week 12 was actually 4.0 meters worse in the empagliflozin-treated HFrEF patients than it was in controls and a mere 4.0 meters better than with placebo in empagliflozin-treated patients with HFpEF, reported William T. Abraham, MD, professor of medicine, director of the division of cardiovascular medicine, and associate dean at Ohio State University, Columbus.
He indicated that the audience shouldn’t make too much of the failure to achieve the primary endpoint in the two trials in light of the studies’ major limitations: namely, their relatively small size for purposes of evaluating clinical outcomes and the relatively short 12-week duration.
“In many ways, I would say it’s remarkable that we can observe a positive signal, a favorable signal, in outcomes around congestion. In the case of HFrEF it’s statistically significant, and in HFpEF it’s a trend towards improvement. Of course, there are larger trials ongoing that may confirm these observations. Hopefully the EMPERIAL trials predict a good outcome for those ongoing trials,” Dr. Abraham said.
Piotr Ponikowski, MD, presented the study results for the secondary outcomes of congestion symptoms, diuretic use, and utilization of health care resources. In EMPERIAL-Reduced, intensification of diuretic therapy – often a prelude to acute decompensation and a trip to the hospital – occurred at a rate of 4.5% with empagliflozin and 16.1% with placebo, for a highly significant 73% relative risk reduction. Intensification of loop diuretics occurred in 2.6% of the empagliflozin group and 14.2% of controls, for a 82% risk reduction.
“That’s a pretty significant effect,” observed Dr. Ponikowski, professor of cardiology and head of the department of heart diseases at the Medical University of Wroclaw (Poland).
Moreover, a congestion symptoms score comprising a summary of orthopnea, jugular veinous distention, and edema improved by 47% after 12 weeks on empagliflozin, a statistically significant and clinically meaningful improvement that grew in magnitude over time and at 12 weeks was twice as large, compared with the reduction in placebo group, he added.
There was a trend for fewer unscheduled outpatient visits in the empagliflozin arm of EMPERIAL-Reduced with a rate of 10.4%, compared with 25.8% in controls; however, this 26% reduction in relative risk did not achieve statistical significance.
Intensification of loop diuretics occurred in 9% of EMPERIAL-Preserved participants on empagliflozin and 13.5% on placebo, but this 34% reduction in risk didn’t reach significance.
Adverse events in the EMPERIAL trials were similar across the active treatment and placebo arms. The benign safety profile was similar to what was seen in the earlier major clinical trials of empagliflozin for treatment of type 2 diabetes.
Session chair Stephane Heymans, MD, PhD, of the University of Maastricht (the Netherlands) noted that a substantial minority of patients in EMPERIAL-Reduced were on the combined neprilysin inhibitor sacubitril and the angiotensin receptor blocker valsartan (Entresto), whereas far fewer were in EMPERIAL-Preserved. He wondered if this greater use of background sacubitril/valsartan could explain empagliflozin’s greater efficacy in EMPERIAL-Reduced.
Highly unlikely, according to the investigators.
“It looks like, as is the case with most of our heart failure therapies, that we do see incremental value here. If you met the criteria for these trials, it appears you derived benefit from empagliflozin regardless of whether you were on an angiotensin receptor neprilysin inhibitor or not. I think that speaks to the incremental benefit of SGLT2 inhibitors on top of current guideline-directed medical therapy,” Dr. Abraham said.
Dr. Ponikowski observed that the same point was underscored in the DAPA-HF trial of the SGLT2 inhibitor dapagliflozin (Farxiga) in patients with heart failure (DAPA-HF: N Engl J Med. 2019 Nov 21;381[21]:1995-2008).
“You’ll see that the mortality and morbidity and quality-of-life benefit is in those treated with dapagliflozin with or without angiotensin receptor neprilysin inhibition; so, regardless of background therapy. And the effect is especially clear in patients on both therapies,” the cardiologist said.
The EMPERIAL trials were sponsored by Boehringer Ingelheim. Dr. Abraham and Dr. Ponikowksi reported receiving consultant fees from the company for serving on the trials’ executive committee.
FROM ESC HEART FAILURE 2020
Why doctors keep monitoring kids who recover from mysterious COVID-linked illness
He’s a 5-year-old boy and would much rather talk about cartoons or the ideas for inventions that constantly pop into his head.
“Hold your horses, I think I know what I’m gonna make,” he said, holding up a finger in the middle of a conversation. “I’m gonna make something that lights up and attaches to things with glue, so if you don’t have a flashlight, you can just use it!”
In New York, at least 237 kids, including Israel, appear to have Multisystem Inflammatory Syndrome in Children (MIS-C). And state officials continue to track the syndrome, but the Centers for Disease Control and Prevention did not respond to repeated requests for information on how many children nationwide have been diagnosed so far with MIS-C.
A study published June 29 in the New England Journal of Medicine reported on 186 patients in 26 states who had been diagnosed with MIS-C. A researcher writing in the same issue added reports from other countries, finding that about 1,000 children worldwide have been diagnosed with MIS-C.
Tracking the long-term health effects of MIS-C
Israel is friendly and energetic, but he’s also really good at sitting still. During a recent checkup at the Children’s Hospital at Montefiore, New York, he had no complaints about all the stickers and wires a health aide attached to him for an EKG. And when Marc Foca, MD, an infectious disease specialist, came by to listen to his heart and lungs, and prod his abdomen, Israel barely seemed to notice.
There were still some tests pending, but overall, Dr. Foca said, “Israel looks like a totally healthy 5-year-old.”
“Stay safe!” Israel called out, as Dr. Foca left. It’s his new sign-off, instead of goodbye. His mother, Janelle Moholland, explained Israel came up with it himself. And she’s also hoping that, after a harrowing couple of weeks in early May, Israel himself will “stay safe.”
That’s why they’ve been returning to Montefiore for the periodic checkups, even though Israel seems to have recovered fully from both COVID-19 and MIS-C.
MIS-C is relatively rare, and it apparently responds well to treatment, but it is new enough – and mysterious enough – that doctors here want to make sure the children who recover don’t experience any related health complications in the future.
“We’ve seen these kids get really sick, and get better and recover and go home, yet we don’t know what the long-term outcomes are,” said Nadine Choueiter, MD, a pediatric cardiologist at Montefiore. “So that’s why we will be seeing them.”
When Israel first got sick at the end of April, his illness didn’t exactly look like COVID-19. He had persistent high fevers, with his temperature reaching 104° F – but no problems breathing. He wasn’t eating. He was barely drinking. He wasn’t using the bathroom. He had abdominal pains. His eyes were red.
They went to the ED a couple of times and visited an urgent care center, but the doctors sent them home without testing him for the coronavirus. Ms. Moholland, 29, said she felt powerless.
“There was nothing I could do but make him comfortable,” she said. “I literally had to just trust in a higher power and just hope that He would come through for us. It taught me a lot about patience and faith.”
As Israel grew sicker, and they still had no answers, Ms. Moholland grew frustrated. “I wish his pediatrician and [the ED and urgent care staff] had done what they were supposed to do and given him a test” when Israel first got sick, Ms. Moholland said. “What harm would it have done? He suffered for about 10 or 11 days that could have been avoided.”
In a later interview, she talked with NPR about how COVID-19 has disproportionately affected the African American community because of a combination of underlying health conditions and lack of access to good health care. She said she felt she, too, had fallen victim to those disparities.
“It affects me, personally, because I am African American, but you just never know,” she said. “It’s hard. We’re living in uncertain times – very uncertain times.”
Finally, the Children’s Hospital at Montefiore admitted Israel – and the test she’d been trying to get for days confirmed he had the virus.
“I was literally in tears, like begging them not to discharge me because I knew he was not fine,” she recalled.
Israel was in shock, and by the time he got to the hospital, doctors were on the lookout for MIS-C, so they recognized his symptoms – which were distinct from most people with COVID-19.
Doctors gave Israel fluids and intravenous immunoglobulin, a substance obtained from donated human plasma, which is used to treat deficiencies in the immune system.
Immunoglobulin has been effective in children like Israel because MIS-C appears to be caused by an immune overreaction to the initial coronavirus infection, according to Dr. Choueiter.
“The immune system starts attacking the body itself, including the arteries of the heart,” she said.
In some MIS-C cases – though not Israel’s – the attack occurs in the coronary arteries, inflaming and dilating them. That also happens in a different syndrome affecting children, Kawasaki disease. About 5% of Kawasaki patients experience aneurysms – which can fatally rupture blood vessels – after the initial condition subsides.
Dr. Choueiter and colleagues want to make sure MIS-C patients don’t face similar risks. So far, they’re cautiously optimistic.
“We have not seen any new decrease in heart function or any new coronary artery dilations,” she said. “When we check their blood, their inflammatory markers are back to normal. For the parents, the child is back to baseline, and it’s as if this illness is a nightmare that’s long gone.”
For a Pennsylvania teen, the MIS-C diagnosis came much later
Not every child who develops MIS-C tests positive for the coronavirus, though many will test positive for antibodies to the coronavirus, indicating they had been infected previously. That was the case with Andrew Lis, a boy from Pennsylvania who was the first MIS-C patient seen at the Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del.
Andrew had been a healthy 14-year-old boy before he got sick. He and his twin brother love sports and video games. He said the first symptom was a bad headache. He developed a fever the next day, then constipation and intense stomach pain.
“It was terrible,” Andrew said. “It was unbearable. I couldn’t really move a lot.”
His mother, Ingrid Lis, said they were thinking appendicitis, not coronavirus, at first. In fact, she hesitated to take Andrew to the hospital, for fear of exposing him to the virus. But after Andrew stopped eating because of his headache and stomach discomfort, “I knew I couldn’t keep him home anymore,” Mrs. Lis said.
Andrew was admitted to the hospital April 12, but that was before reports of the mysterious syndrome had started trickling out of Europe.
Over about 5 days in the pediatric ICU, Andrew’s condition deteriorated rapidly, as doctors struggled to figure out what was wrong. Puzzled, they tried treatments for scarlet fever, strep throat, and toxic shock syndrome. Andrew’s body broke out in rashes, then his heart began failing and he was put on a ventilator. Andrew’s father, Ed Lis, said doctors told the family to brace for the worst: “We’ve got a healthy kid who a few days ago was just having these sort of strange symptoms. And now they’re telling us that we could lose him.”
Though Andrew’s symptoms were atypical for Kawasaki disease, doctors decided to give him the standard treatment for that condition – administering intravenous immunoglobulin, the same treatment Israel Shippy received.
“Within the 24 hours of the infusion, he was a different person,” Mrs. Lis said. Andrew was removed from the ventilator, and his appetite eventually returned. “That’s when we knew that we had turned that corner.”
It wasn’t until after Andrew’s discharge that his doctors learned about MIS-C from colleagues in Europe. They recommended the whole family be tested for antibodies to the coronavirus. Although Andrew tested positive, the rest of the family – both parents, Andrew’s twin brother and two older siblings – all tested negative. Andrew’s mother is still not sure how he was exposed since the family had been observing a strict lockdown since mid-March. Both she and her husband were working remotely from home, and she says they all wore masks and were conscientious about hand-washing when they ventured out for groceries. She thinks Andrew must have been exposed at least a month before his illness began.
And she’s puzzled why the rest of her close-knit family wasn’t infected as well. “We are a Latino family,” Mrs. Lis said. “We are very used to being together, clustering in the same room.” Even when Andrew was sick, she says, all six of them huddled in his bedroom to comfort him.
Meanwhile, Andrew has made a quick recovery. Not long after his discharge in April, he turned 15 and resumed an exercise routine involving running, push-ups, and sit-ups. A few weeks later, an ECG showed Andrew’s heart was “perfect,” Mr. Lis said. Still, doctors have asked Andrew to follow up with a cardiologist every 3 months.
An eye on the long-term effects
The medical team at Montefiore is tracking the 40 children they have already treated and discharged. With kids showing few symptoms in the immediate aftermath, Dr. Choueiter hopes the long-term trajectory after MIS-C will be similar to what happens after Kawasaki disease.
“Usually children who have had coronary artery dilations [from Kawasaki disease] that have resolved within the first 6 weeks of the illness do well long-term,” said Dr. Choueiter, who runs the Kawasaki disease program at Montefiore.
The Montefiore team is asking patients affected by MIS-C to return for a checkup 1 week after discharge, then after 1 month, 3 months, 6 months, and a year. They will be evaluated by pediatric cardiologists, hematologists, rheumatologists and infectious disease specialists.
Montefiore and other children’s hospitals around the country are sharing information. Dr. Choueiter wants to establish an even longer-term monitoring program for MIS-C, comparable with registries that exist for other diseases.
Ms. Moholland is glad the hospital is being vigilant.
“The uncertainty of not knowing whether it could come back in his future is a little unsettling,” she said. “But I am hopeful.”
This story is part of a partnership that includes WNYC, NPR, and Kaiser Health News. A version of this article originally appeared on Kaiser Health News.
He’s a 5-year-old boy and would much rather talk about cartoons or the ideas for inventions that constantly pop into his head.
“Hold your horses, I think I know what I’m gonna make,” he said, holding up a finger in the middle of a conversation. “I’m gonna make something that lights up and attaches to things with glue, so if you don’t have a flashlight, you can just use it!”
In New York, at least 237 kids, including Israel, appear to have Multisystem Inflammatory Syndrome in Children (MIS-C). And state officials continue to track the syndrome, but the Centers for Disease Control and Prevention did not respond to repeated requests for information on how many children nationwide have been diagnosed so far with MIS-C.
A study published June 29 in the New England Journal of Medicine reported on 186 patients in 26 states who had been diagnosed with MIS-C. A researcher writing in the same issue added reports from other countries, finding that about 1,000 children worldwide have been diagnosed with MIS-C.
Tracking the long-term health effects of MIS-C
Israel is friendly and energetic, but he’s also really good at sitting still. During a recent checkup at the Children’s Hospital at Montefiore, New York, he had no complaints about all the stickers and wires a health aide attached to him for an EKG. And when Marc Foca, MD, an infectious disease specialist, came by to listen to his heart and lungs, and prod his abdomen, Israel barely seemed to notice.
There were still some tests pending, but overall, Dr. Foca said, “Israel looks like a totally healthy 5-year-old.”
“Stay safe!” Israel called out, as Dr. Foca left. It’s his new sign-off, instead of goodbye. His mother, Janelle Moholland, explained Israel came up with it himself. And she’s also hoping that, after a harrowing couple of weeks in early May, Israel himself will “stay safe.”
That’s why they’ve been returning to Montefiore for the periodic checkups, even though Israel seems to have recovered fully from both COVID-19 and MIS-C.
MIS-C is relatively rare, and it apparently responds well to treatment, but it is new enough – and mysterious enough – that doctors here want to make sure the children who recover don’t experience any related health complications in the future.
“We’ve seen these kids get really sick, and get better and recover and go home, yet we don’t know what the long-term outcomes are,” said Nadine Choueiter, MD, a pediatric cardiologist at Montefiore. “So that’s why we will be seeing them.”
When Israel first got sick at the end of April, his illness didn’t exactly look like COVID-19. He had persistent high fevers, with his temperature reaching 104° F – but no problems breathing. He wasn’t eating. He was barely drinking. He wasn’t using the bathroom. He had abdominal pains. His eyes were red.
They went to the ED a couple of times and visited an urgent care center, but the doctors sent them home without testing him for the coronavirus. Ms. Moholland, 29, said she felt powerless.
“There was nothing I could do but make him comfortable,” she said. “I literally had to just trust in a higher power and just hope that He would come through for us. It taught me a lot about patience and faith.”
As Israel grew sicker, and they still had no answers, Ms. Moholland grew frustrated. “I wish his pediatrician and [the ED and urgent care staff] had done what they were supposed to do and given him a test” when Israel first got sick, Ms. Moholland said. “What harm would it have done? He suffered for about 10 or 11 days that could have been avoided.”
In a later interview, she talked with NPR about how COVID-19 has disproportionately affected the African American community because of a combination of underlying health conditions and lack of access to good health care. She said she felt she, too, had fallen victim to those disparities.
“It affects me, personally, because I am African American, but you just never know,” she said. “It’s hard. We’re living in uncertain times – very uncertain times.”
Finally, the Children’s Hospital at Montefiore admitted Israel – and the test she’d been trying to get for days confirmed he had the virus.
“I was literally in tears, like begging them not to discharge me because I knew he was not fine,” she recalled.
Israel was in shock, and by the time he got to the hospital, doctors were on the lookout for MIS-C, so they recognized his symptoms – which were distinct from most people with COVID-19.
Doctors gave Israel fluids and intravenous immunoglobulin, a substance obtained from donated human plasma, which is used to treat deficiencies in the immune system.
Immunoglobulin has been effective in children like Israel because MIS-C appears to be caused by an immune overreaction to the initial coronavirus infection, according to Dr. Choueiter.
“The immune system starts attacking the body itself, including the arteries of the heart,” she said.
In some MIS-C cases – though not Israel’s – the attack occurs in the coronary arteries, inflaming and dilating them. That also happens in a different syndrome affecting children, Kawasaki disease. About 5% of Kawasaki patients experience aneurysms – which can fatally rupture blood vessels – after the initial condition subsides.
Dr. Choueiter and colleagues want to make sure MIS-C patients don’t face similar risks. So far, they’re cautiously optimistic.
“We have not seen any new decrease in heart function or any new coronary artery dilations,” she said. “When we check their blood, their inflammatory markers are back to normal. For the parents, the child is back to baseline, and it’s as if this illness is a nightmare that’s long gone.”
For a Pennsylvania teen, the MIS-C diagnosis came much later
Not every child who develops MIS-C tests positive for the coronavirus, though many will test positive for antibodies to the coronavirus, indicating they had been infected previously. That was the case with Andrew Lis, a boy from Pennsylvania who was the first MIS-C patient seen at the Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del.
Andrew had been a healthy 14-year-old boy before he got sick. He and his twin brother love sports and video games. He said the first symptom was a bad headache. He developed a fever the next day, then constipation and intense stomach pain.
“It was terrible,” Andrew said. “It was unbearable. I couldn’t really move a lot.”
His mother, Ingrid Lis, said they were thinking appendicitis, not coronavirus, at first. In fact, she hesitated to take Andrew to the hospital, for fear of exposing him to the virus. But after Andrew stopped eating because of his headache and stomach discomfort, “I knew I couldn’t keep him home anymore,” Mrs. Lis said.
Andrew was admitted to the hospital April 12, but that was before reports of the mysterious syndrome had started trickling out of Europe.
Over about 5 days in the pediatric ICU, Andrew’s condition deteriorated rapidly, as doctors struggled to figure out what was wrong. Puzzled, they tried treatments for scarlet fever, strep throat, and toxic shock syndrome. Andrew’s body broke out in rashes, then his heart began failing and he was put on a ventilator. Andrew’s father, Ed Lis, said doctors told the family to brace for the worst: “We’ve got a healthy kid who a few days ago was just having these sort of strange symptoms. And now they’re telling us that we could lose him.”
Though Andrew’s symptoms were atypical for Kawasaki disease, doctors decided to give him the standard treatment for that condition – administering intravenous immunoglobulin, the same treatment Israel Shippy received.
“Within the 24 hours of the infusion, he was a different person,” Mrs. Lis said. Andrew was removed from the ventilator, and his appetite eventually returned. “That’s when we knew that we had turned that corner.”
It wasn’t until after Andrew’s discharge that his doctors learned about MIS-C from colleagues in Europe. They recommended the whole family be tested for antibodies to the coronavirus. Although Andrew tested positive, the rest of the family – both parents, Andrew’s twin brother and two older siblings – all tested negative. Andrew’s mother is still not sure how he was exposed since the family had been observing a strict lockdown since mid-March. Both she and her husband were working remotely from home, and she says they all wore masks and were conscientious about hand-washing when they ventured out for groceries. She thinks Andrew must have been exposed at least a month before his illness began.
And she’s puzzled why the rest of her close-knit family wasn’t infected as well. “We are a Latino family,” Mrs. Lis said. “We are very used to being together, clustering in the same room.” Even when Andrew was sick, she says, all six of them huddled in his bedroom to comfort him.
Meanwhile, Andrew has made a quick recovery. Not long after his discharge in April, he turned 15 and resumed an exercise routine involving running, push-ups, and sit-ups. A few weeks later, an ECG showed Andrew’s heart was “perfect,” Mr. Lis said. Still, doctors have asked Andrew to follow up with a cardiologist every 3 months.
An eye on the long-term effects
The medical team at Montefiore is tracking the 40 children they have already treated and discharged. With kids showing few symptoms in the immediate aftermath, Dr. Choueiter hopes the long-term trajectory after MIS-C will be similar to what happens after Kawasaki disease.
“Usually children who have had coronary artery dilations [from Kawasaki disease] that have resolved within the first 6 weeks of the illness do well long-term,” said Dr. Choueiter, who runs the Kawasaki disease program at Montefiore.
The Montefiore team is asking patients affected by MIS-C to return for a checkup 1 week after discharge, then after 1 month, 3 months, 6 months, and a year. They will be evaluated by pediatric cardiologists, hematologists, rheumatologists and infectious disease specialists.
Montefiore and other children’s hospitals around the country are sharing information. Dr. Choueiter wants to establish an even longer-term monitoring program for MIS-C, comparable with registries that exist for other diseases.
Ms. Moholland is glad the hospital is being vigilant.
“The uncertainty of not knowing whether it could come back in his future is a little unsettling,” she said. “But I am hopeful.”
This story is part of a partnership that includes WNYC, NPR, and Kaiser Health News. A version of this article originally appeared on Kaiser Health News.
He’s a 5-year-old boy and would much rather talk about cartoons or the ideas for inventions that constantly pop into his head.
“Hold your horses, I think I know what I’m gonna make,” he said, holding up a finger in the middle of a conversation. “I’m gonna make something that lights up and attaches to things with glue, so if you don’t have a flashlight, you can just use it!”
In New York, at least 237 kids, including Israel, appear to have Multisystem Inflammatory Syndrome in Children (MIS-C). And state officials continue to track the syndrome, but the Centers for Disease Control and Prevention did not respond to repeated requests for information on how many children nationwide have been diagnosed so far with MIS-C.
A study published June 29 in the New England Journal of Medicine reported on 186 patients in 26 states who had been diagnosed with MIS-C. A researcher writing in the same issue added reports from other countries, finding that about 1,000 children worldwide have been diagnosed with MIS-C.
Tracking the long-term health effects of MIS-C
Israel is friendly and energetic, but he’s also really good at sitting still. During a recent checkup at the Children’s Hospital at Montefiore, New York, he had no complaints about all the stickers and wires a health aide attached to him for an EKG. And when Marc Foca, MD, an infectious disease specialist, came by to listen to his heart and lungs, and prod his abdomen, Israel barely seemed to notice.
There were still some tests pending, but overall, Dr. Foca said, “Israel looks like a totally healthy 5-year-old.”
“Stay safe!” Israel called out, as Dr. Foca left. It’s his new sign-off, instead of goodbye. His mother, Janelle Moholland, explained Israel came up with it himself. And she’s also hoping that, after a harrowing couple of weeks in early May, Israel himself will “stay safe.”
That’s why they’ve been returning to Montefiore for the periodic checkups, even though Israel seems to have recovered fully from both COVID-19 and MIS-C.
MIS-C is relatively rare, and it apparently responds well to treatment, but it is new enough – and mysterious enough – that doctors here want to make sure the children who recover don’t experience any related health complications in the future.
“We’ve seen these kids get really sick, and get better and recover and go home, yet we don’t know what the long-term outcomes are,” said Nadine Choueiter, MD, a pediatric cardiologist at Montefiore. “So that’s why we will be seeing them.”
When Israel first got sick at the end of April, his illness didn’t exactly look like COVID-19. He had persistent high fevers, with his temperature reaching 104° F – but no problems breathing. He wasn’t eating. He was barely drinking. He wasn’t using the bathroom. He had abdominal pains. His eyes were red.
They went to the ED a couple of times and visited an urgent care center, but the doctors sent them home without testing him for the coronavirus. Ms. Moholland, 29, said she felt powerless.
“There was nothing I could do but make him comfortable,” she said. “I literally had to just trust in a higher power and just hope that He would come through for us. It taught me a lot about patience and faith.”
As Israel grew sicker, and they still had no answers, Ms. Moholland grew frustrated. “I wish his pediatrician and [the ED and urgent care staff] had done what they were supposed to do and given him a test” when Israel first got sick, Ms. Moholland said. “What harm would it have done? He suffered for about 10 or 11 days that could have been avoided.”
In a later interview, she talked with NPR about how COVID-19 has disproportionately affected the African American community because of a combination of underlying health conditions and lack of access to good health care. She said she felt she, too, had fallen victim to those disparities.
“It affects me, personally, because I am African American, but you just never know,” she said. “It’s hard. We’re living in uncertain times – very uncertain times.”
Finally, the Children’s Hospital at Montefiore admitted Israel – and the test she’d been trying to get for days confirmed he had the virus.
“I was literally in tears, like begging them not to discharge me because I knew he was not fine,” she recalled.
Israel was in shock, and by the time he got to the hospital, doctors were on the lookout for MIS-C, so they recognized his symptoms – which were distinct from most people with COVID-19.
Doctors gave Israel fluids and intravenous immunoglobulin, a substance obtained from donated human plasma, which is used to treat deficiencies in the immune system.
Immunoglobulin has been effective in children like Israel because MIS-C appears to be caused by an immune overreaction to the initial coronavirus infection, according to Dr. Choueiter.
“The immune system starts attacking the body itself, including the arteries of the heart,” she said.
In some MIS-C cases – though not Israel’s – the attack occurs in the coronary arteries, inflaming and dilating them. That also happens in a different syndrome affecting children, Kawasaki disease. About 5% of Kawasaki patients experience aneurysms – which can fatally rupture blood vessels – after the initial condition subsides.
Dr. Choueiter and colleagues want to make sure MIS-C patients don’t face similar risks. So far, they’re cautiously optimistic.
“We have not seen any new decrease in heart function or any new coronary artery dilations,” she said. “When we check their blood, their inflammatory markers are back to normal. For the parents, the child is back to baseline, and it’s as if this illness is a nightmare that’s long gone.”
For a Pennsylvania teen, the MIS-C diagnosis came much later
Not every child who develops MIS-C tests positive for the coronavirus, though many will test positive for antibodies to the coronavirus, indicating they had been infected previously. That was the case with Andrew Lis, a boy from Pennsylvania who was the first MIS-C patient seen at the Nemours/Alfred I. duPont Hospital for Children in Wilmington, Del.
Andrew had been a healthy 14-year-old boy before he got sick. He and his twin brother love sports and video games. He said the first symptom was a bad headache. He developed a fever the next day, then constipation and intense stomach pain.
“It was terrible,” Andrew said. “It was unbearable. I couldn’t really move a lot.”
His mother, Ingrid Lis, said they were thinking appendicitis, not coronavirus, at first. In fact, she hesitated to take Andrew to the hospital, for fear of exposing him to the virus. But after Andrew stopped eating because of his headache and stomach discomfort, “I knew I couldn’t keep him home anymore,” Mrs. Lis said.
Andrew was admitted to the hospital April 12, but that was before reports of the mysterious syndrome had started trickling out of Europe.
Over about 5 days in the pediatric ICU, Andrew’s condition deteriorated rapidly, as doctors struggled to figure out what was wrong. Puzzled, they tried treatments for scarlet fever, strep throat, and toxic shock syndrome. Andrew’s body broke out in rashes, then his heart began failing and he was put on a ventilator. Andrew’s father, Ed Lis, said doctors told the family to brace for the worst: “We’ve got a healthy kid who a few days ago was just having these sort of strange symptoms. And now they’re telling us that we could lose him.”
Though Andrew’s symptoms were atypical for Kawasaki disease, doctors decided to give him the standard treatment for that condition – administering intravenous immunoglobulin, the same treatment Israel Shippy received.
“Within the 24 hours of the infusion, he was a different person,” Mrs. Lis said. Andrew was removed from the ventilator, and his appetite eventually returned. “That’s when we knew that we had turned that corner.”
It wasn’t until after Andrew’s discharge that his doctors learned about MIS-C from colleagues in Europe. They recommended the whole family be tested for antibodies to the coronavirus. Although Andrew tested positive, the rest of the family – both parents, Andrew’s twin brother and two older siblings – all tested negative. Andrew’s mother is still not sure how he was exposed since the family had been observing a strict lockdown since mid-March. Both she and her husband were working remotely from home, and she says they all wore masks and were conscientious about hand-washing when they ventured out for groceries. She thinks Andrew must have been exposed at least a month before his illness began.
And she’s puzzled why the rest of her close-knit family wasn’t infected as well. “We are a Latino family,” Mrs. Lis said. “We are very used to being together, clustering in the same room.” Even when Andrew was sick, she says, all six of them huddled in his bedroom to comfort him.
Meanwhile, Andrew has made a quick recovery. Not long after his discharge in April, he turned 15 and resumed an exercise routine involving running, push-ups, and sit-ups. A few weeks later, an ECG showed Andrew’s heart was “perfect,” Mr. Lis said. Still, doctors have asked Andrew to follow up with a cardiologist every 3 months.
An eye on the long-term effects
The medical team at Montefiore is tracking the 40 children they have already treated and discharged. With kids showing few symptoms in the immediate aftermath, Dr. Choueiter hopes the long-term trajectory after MIS-C will be similar to what happens after Kawasaki disease.
“Usually children who have had coronary artery dilations [from Kawasaki disease] that have resolved within the first 6 weeks of the illness do well long-term,” said Dr. Choueiter, who runs the Kawasaki disease program at Montefiore.
The Montefiore team is asking patients affected by MIS-C to return for a checkup 1 week after discharge, then after 1 month, 3 months, 6 months, and a year. They will be evaluated by pediatric cardiologists, hematologists, rheumatologists and infectious disease specialists.
Montefiore and other children’s hospitals around the country are sharing information. Dr. Choueiter wants to establish an even longer-term monitoring program for MIS-C, comparable with registries that exist for other diseases.
Ms. Moholland is glad the hospital is being vigilant.
“The uncertainty of not knowing whether it could come back in his future is a little unsettling,” she said. “But I am hopeful.”
This story is part of a partnership that includes WNYC, NPR, and Kaiser Health News. A version of this article originally appeared on Kaiser Health News.