User login
Melanoma screening study stokes overdiagnosis debate
new research shows.
Without a corresponding decrease in melanoma mortality, an increase in the detection of those thin melanomas “raises the concern that early detection efforts, such as visual skin screening, may result in overdiagnosis,” the study authors wrote. “The value of a cancer screening program should most rigorously be measured not by the number of new, early cancers detected, but by its impact on the development of late-stage disease and its associated morbidity, cost, and mortality.”
The research, published in JAMA Dermatology, has reignited the controversy over the benefits and harms of primary care skin cancer screening, garnering two editorials that reflect different sides of the debate.
In one, Robert A. Swerlick, MD, pointed out that, “despite public messaging to the contrary, to my knowledge there is no evidence that routine skin examinations have any effect on melanoma mortality.
“The stage shift to smaller tumors should not be viewed as success and is very strong evidence of overdiagnosis,” wrote Dr. Swerlick, of the department of dermatology, Emory University, Atlanta.
The other editorial, however, argued that routine screening saves lives. “Most melanoma deaths are because of stage I disease, with an estimated 3%-15% of thin melanomas (≤ 1 mm) being lethal,” wrote a trio of editorialists from Oregon Health & Science University, Portland.
When considering the high mutation rate associated with melanoma and the current limits of treatment options, early diagnosis becomes “particularly important and counterbalances the risk of overdiagnosis,” the editorialists asserted.
Primary care screening study
The new findings come from an observational study of a quality improvement initiative conducted at the University of Pittsburgh Medical Center system between 2014 and 2018, in which primary care clinicians were offered training in melanoma identification through skin examination and were encouraged to offer annual skin cancer screening to patients aged 35 years and older.
Of 595,799 eligible patients, 144,851 (24.3%) were screened at least once during the study period. Those who received screening were more likely than unscreened patients to be older (median age, 59 vs. 55 years), women, and non-Hispanic White persons.
During a follow-up of 5 years, the researchers found that patients who received screening were significantly more likely than unscreened patients to be diagnosed with in situ melanoma (incidence, 30.4 vs. 14.4; hazard ratio, 2.6; P < .001) or thin invasive melanoma (incidence, 24.5 vs. 16.1; HR, 1.8; P < .001), after adjusting for factors that included age, sex, and race.
The screened patients were also more likely than unscreened patients to be diagnosed with in situ interval melanomas, defined as melanomas occurring at least 60 days after initial screening (incidence, 26.7 vs. 12.9; HR, 2.1; P < .001), as well as thin invasive interval melanomas (incidence, 18.5 vs. 14.4; HR, 1.3; P = .03).
The 60-day interval was included to account for the possible time to referral to a specialist for definitive diagnosis, the authors explained.
The incidence of the detection of melanomas thicker than 4 mm was lower in screened versus unscreened patients, but the difference was not statistically significant for all melanomas (2.7 vs. 3.3; HR, 0.8; P = .38) or interval melanomas (1.5 vs. 2.7; HR, 0.6; P = .15).
Experts weigh in
Although the follow-up period was of 5 years, not all patients were followed that long after undergoing screening. For instance, for some patients, follow-up occurred only 1 year after they had been screened.
The study’s senior author, Laura K. Ferris, MD, PhD, of the department of dermatology, University of Pittsburgh, noted that a longer follow-up could shift the results.
“When you look at the curves in our figures, you do start to see them separate more and more over time for the thicker melanomas,” Dr. Ferris said in an interview. “I do suspect that, if we followed patients longer, we might start to see a more significant difference.”
The findings nevertheless add to evidence that although routine screening substantially increases the detection of melanomas overall, these melanomas are often not the ones doctors are most worried about or that increase a person’s risk of mortality, Dr. Ferris noted.
When it comes to melanoma screening, balancing the risks and benefits is key. One major downside, Dr. Ferris said, is in regard to the burden such screening could place on the health care system, with potentially unproductive screenings causing delays in care for patients with more urgent needs.
“We are undersupplied in the dermatology workforce, and there is often a long wait to see dermatologists, so we really want to make sure, as trained professionals, that patients have access to us,” she said. “If we’re doing something that doesn’t have proven benefit and is increasing the wait time, that will come at the expense of other patients’ access.”
Costs involved in skin biopsies and excisions of borderline lesions as well as the potential to increase patients’ anxiety represent other important considerations, Dr. Ferris noted.
However, Sancy A. Leachman, MD, PhD, a coauthor of the editorial in favor of screening, said in an interview that “at the individual level, there are an almost infinite number of individual circumstances that could lead a person to decide that the potential benefits outweigh the harms.”
According to Dr. Leachman, who is chair of the department of dermatology, Oregon Health & Science University, these individual priorities may not align with those of the various decision-makers or with guidelines, such as those from the U.S. Preventive Services Task Force, which gives visual skin cancer screening of asymptomatic patients an “I” rating, indicating “insufficient evidence.”
“Many federal agencies and payer groups focus on minimizing costs and optimizing outcomes,” Dr. Leachman and coauthors wrote. As the only professional advocates for individual patients, physicians “have a responsibility to assure that the best interests of patients are served.”
The study was funded by the University of Pittsburgh Melanoma and Skin Cancer Program. Dr. Ferris and Dr. Swerlick disclosed no relevant financial relationships. Dr. Leachman is the principal investigator for War on Melanoma, an early-detection program in Oregon.
A version of this article first appeared on Medscape.com.
new research shows.
Without a corresponding decrease in melanoma mortality, an increase in the detection of those thin melanomas “raises the concern that early detection efforts, such as visual skin screening, may result in overdiagnosis,” the study authors wrote. “The value of a cancer screening program should most rigorously be measured not by the number of new, early cancers detected, but by its impact on the development of late-stage disease and its associated morbidity, cost, and mortality.”
The research, published in JAMA Dermatology, has reignited the controversy over the benefits and harms of primary care skin cancer screening, garnering two editorials that reflect different sides of the debate.
In one, Robert A. Swerlick, MD, pointed out that, “despite public messaging to the contrary, to my knowledge there is no evidence that routine skin examinations have any effect on melanoma mortality.
“The stage shift to smaller tumors should not be viewed as success and is very strong evidence of overdiagnosis,” wrote Dr. Swerlick, of the department of dermatology, Emory University, Atlanta.
The other editorial, however, argued that routine screening saves lives. “Most melanoma deaths are because of stage I disease, with an estimated 3%-15% of thin melanomas (≤ 1 mm) being lethal,” wrote a trio of editorialists from Oregon Health & Science University, Portland.
When considering the high mutation rate associated with melanoma and the current limits of treatment options, early diagnosis becomes “particularly important and counterbalances the risk of overdiagnosis,” the editorialists asserted.
Primary care screening study
The new findings come from an observational study of a quality improvement initiative conducted at the University of Pittsburgh Medical Center system between 2014 and 2018, in which primary care clinicians were offered training in melanoma identification through skin examination and were encouraged to offer annual skin cancer screening to patients aged 35 years and older.
Of 595,799 eligible patients, 144,851 (24.3%) were screened at least once during the study period. Those who received screening were more likely than unscreened patients to be older (median age, 59 vs. 55 years), women, and non-Hispanic White persons.
During a follow-up of 5 years, the researchers found that patients who received screening were significantly more likely than unscreened patients to be diagnosed with in situ melanoma (incidence, 30.4 vs. 14.4; hazard ratio, 2.6; P < .001) or thin invasive melanoma (incidence, 24.5 vs. 16.1; HR, 1.8; P < .001), after adjusting for factors that included age, sex, and race.
The screened patients were also more likely than unscreened patients to be diagnosed with in situ interval melanomas, defined as melanomas occurring at least 60 days after initial screening (incidence, 26.7 vs. 12.9; HR, 2.1; P < .001), as well as thin invasive interval melanomas (incidence, 18.5 vs. 14.4; HR, 1.3; P = .03).
The 60-day interval was included to account for the possible time to referral to a specialist for definitive diagnosis, the authors explained.
The incidence of the detection of melanomas thicker than 4 mm was lower in screened versus unscreened patients, but the difference was not statistically significant for all melanomas (2.7 vs. 3.3; HR, 0.8; P = .38) or interval melanomas (1.5 vs. 2.7; HR, 0.6; P = .15).
Experts weigh in
Although the follow-up period was of 5 years, not all patients were followed that long after undergoing screening. For instance, for some patients, follow-up occurred only 1 year after they had been screened.
The study’s senior author, Laura K. Ferris, MD, PhD, of the department of dermatology, University of Pittsburgh, noted that a longer follow-up could shift the results.
“When you look at the curves in our figures, you do start to see them separate more and more over time for the thicker melanomas,” Dr. Ferris said in an interview. “I do suspect that, if we followed patients longer, we might start to see a more significant difference.”
The findings nevertheless add to evidence that although routine screening substantially increases the detection of melanomas overall, these melanomas are often not the ones doctors are most worried about or that increase a person’s risk of mortality, Dr. Ferris noted.
When it comes to melanoma screening, balancing the risks and benefits is key. One major downside, Dr. Ferris said, is in regard to the burden such screening could place on the health care system, with potentially unproductive screenings causing delays in care for patients with more urgent needs.
“We are undersupplied in the dermatology workforce, and there is often a long wait to see dermatologists, so we really want to make sure, as trained professionals, that patients have access to us,” she said. “If we’re doing something that doesn’t have proven benefit and is increasing the wait time, that will come at the expense of other patients’ access.”
Costs involved in skin biopsies and excisions of borderline lesions as well as the potential to increase patients’ anxiety represent other important considerations, Dr. Ferris noted.
However, Sancy A. Leachman, MD, PhD, a coauthor of the editorial in favor of screening, said in an interview that “at the individual level, there are an almost infinite number of individual circumstances that could lead a person to decide that the potential benefits outweigh the harms.”
According to Dr. Leachman, who is chair of the department of dermatology, Oregon Health & Science University, these individual priorities may not align with those of the various decision-makers or with guidelines, such as those from the U.S. Preventive Services Task Force, which gives visual skin cancer screening of asymptomatic patients an “I” rating, indicating “insufficient evidence.”
“Many federal agencies and payer groups focus on minimizing costs and optimizing outcomes,” Dr. Leachman and coauthors wrote. As the only professional advocates for individual patients, physicians “have a responsibility to assure that the best interests of patients are served.”
The study was funded by the University of Pittsburgh Melanoma and Skin Cancer Program. Dr. Ferris and Dr. Swerlick disclosed no relevant financial relationships. Dr. Leachman is the principal investigator for War on Melanoma, an early-detection program in Oregon.
A version of this article first appeared on Medscape.com.
new research shows.
Without a corresponding decrease in melanoma mortality, an increase in the detection of those thin melanomas “raises the concern that early detection efforts, such as visual skin screening, may result in overdiagnosis,” the study authors wrote. “The value of a cancer screening program should most rigorously be measured not by the number of new, early cancers detected, but by its impact on the development of late-stage disease and its associated morbidity, cost, and mortality.”
The research, published in JAMA Dermatology, has reignited the controversy over the benefits and harms of primary care skin cancer screening, garnering two editorials that reflect different sides of the debate.
In one, Robert A. Swerlick, MD, pointed out that, “despite public messaging to the contrary, to my knowledge there is no evidence that routine skin examinations have any effect on melanoma mortality.
“The stage shift to smaller tumors should not be viewed as success and is very strong evidence of overdiagnosis,” wrote Dr. Swerlick, of the department of dermatology, Emory University, Atlanta.
The other editorial, however, argued that routine screening saves lives. “Most melanoma deaths are because of stage I disease, with an estimated 3%-15% of thin melanomas (≤ 1 mm) being lethal,” wrote a trio of editorialists from Oregon Health & Science University, Portland.
When considering the high mutation rate associated with melanoma and the current limits of treatment options, early diagnosis becomes “particularly important and counterbalances the risk of overdiagnosis,” the editorialists asserted.
Primary care screening study
The new findings come from an observational study of a quality improvement initiative conducted at the University of Pittsburgh Medical Center system between 2014 and 2018, in which primary care clinicians were offered training in melanoma identification through skin examination and were encouraged to offer annual skin cancer screening to patients aged 35 years and older.
Of 595,799 eligible patients, 144,851 (24.3%) were screened at least once during the study period. Those who received screening were more likely than unscreened patients to be older (median age, 59 vs. 55 years), women, and non-Hispanic White persons.
During a follow-up of 5 years, the researchers found that patients who received screening were significantly more likely than unscreened patients to be diagnosed with in situ melanoma (incidence, 30.4 vs. 14.4; hazard ratio, 2.6; P < .001) or thin invasive melanoma (incidence, 24.5 vs. 16.1; HR, 1.8; P < .001), after adjusting for factors that included age, sex, and race.
The screened patients were also more likely than unscreened patients to be diagnosed with in situ interval melanomas, defined as melanomas occurring at least 60 days after initial screening (incidence, 26.7 vs. 12.9; HR, 2.1; P < .001), as well as thin invasive interval melanomas (incidence, 18.5 vs. 14.4; HR, 1.3; P = .03).
The 60-day interval was included to account for the possible time to referral to a specialist for definitive diagnosis, the authors explained.
The incidence of the detection of melanomas thicker than 4 mm was lower in screened versus unscreened patients, but the difference was not statistically significant for all melanomas (2.7 vs. 3.3; HR, 0.8; P = .38) or interval melanomas (1.5 vs. 2.7; HR, 0.6; P = .15).
Experts weigh in
Although the follow-up period was of 5 years, not all patients were followed that long after undergoing screening. For instance, for some patients, follow-up occurred only 1 year after they had been screened.
The study’s senior author, Laura K. Ferris, MD, PhD, of the department of dermatology, University of Pittsburgh, noted that a longer follow-up could shift the results.
“When you look at the curves in our figures, you do start to see them separate more and more over time for the thicker melanomas,” Dr. Ferris said in an interview. “I do suspect that, if we followed patients longer, we might start to see a more significant difference.”
The findings nevertheless add to evidence that although routine screening substantially increases the detection of melanomas overall, these melanomas are often not the ones doctors are most worried about or that increase a person’s risk of mortality, Dr. Ferris noted.
When it comes to melanoma screening, balancing the risks and benefits is key. One major downside, Dr. Ferris said, is in regard to the burden such screening could place on the health care system, with potentially unproductive screenings causing delays in care for patients with more urgent needs.
“We are undersupplied in the dermatology workforce, and there is often a long wait to see dermatologists, so we really want to make sure, as trained professionals, that patients have access to us,” she said. “If we’re doing something that doesn’t have proven benefit and is increasing the wait time, that will come at the expense of other patients’ access.”
Costs involved in skin biopsies and excisions of borderline lesions as well as the potential to increase patients’ anxiety represent other important considerations, Dr. Ferris noted.
However, Sancy A. Leachman, MD, PhD, a coauthor of the editorial in favor of screening, said in an interview that “at the individual level, there are an almost infinite number of individual circumstances that could lead a person to decide that the potential benefits outweigh the harms.”
According to Dr. Leachman, who is chair of the department of dermatology, Oregon Health & Science University, these individual priorities may not align with those of the various decision-makers or with guidelines, such as those from the U.S. Preventive Services Task Force, which gives visual skin cancer screening of asymptomatic patients an “I” rating, indicating “insufficient evidence.”
“Many federal agencies and payer groups focus on minimizing costs and optimizing outcomes,” Dr. Leachman and coauthors wrote. As the only professional advocates for individual patients, physicians “have a responsibility to assure that the best interests of patients are served.”
The study was funded by the University of Pittsburgh Melanoma and Skin Cancer Program. Dr. Ferris and Dr. Swerlick disclosed no relevant financial relationships. Dr. Leachman is the principal investigator for War on Melanoma, an early-detection program in Oregon.
A version of this article first appeared on Medscape.com.
FROM JAMA DERMATOLOGY
Hypocaloric diet controls joint activity in psoriatic arthritis – regardless of weight loss
, Brazilian researchers found.
Earlier research has reported that weight loss improves the symptoms of PsA.
Improvement in the Brazilian DIETA study was linked to both better eating patterns and better quality of diet, and while omega-3 supplementation caused relevant body composition changes, it did not improve disease activity, according to Beatriz F. Leite of the division of rheumatology at the Federal University of São Paulo and colleagues.
“The DIETA trial, a nonpharmacologic approach, is an inexpensive, suitable, and efficient approach that could be combined with standardized drug therapy,” the investigators wrote online in Advances in Rheumatology.
Dietary counseling aimed at losing or controlling weight could therefore be part of the global protocol for PsA patients, the researchers added. They conceded, however, that nonpharmacologic interventions traditionally have a low rate of adherence.
This recommendation aligns with a systematic review by the National Psoriasis Foundation, which found evidence of benefit with dietary weight reduction via a hypocaloric diet in overweight and obese patients with psoriasis and/or PsA.
The DIETA trial
The 12-week randomized, double-blind, placebo-controlled study, conducted at three hospitals in São Paulo from September 2012 to May 2014, assessed whether dietary changes, antioxidant supplementation, or weight loss of 5%-10% could improve skin and joint activity in 97 enrolled PsA patients.
Participants were randomized into the following supervised dietary groups:
- Diet-placebo (hypocaloric diet plus placebo supplementation).
- Diet-fish (hypocaloric diet plus 3 g/day of omega-3 supplementation).
- Placebo (with habitual diet).
Diets were carefully tailored to each individual patient. The regimen for overweight and obese patients included a 500-kcal restriction, while for eutrophic patients, diets were calculated to maintain weight with no caloric restriction.
In the 91 patients evaluable by multiple measures at 12 weeks, Ms. Leite and colleagues observed the following:
- The Disease Activity Score 28 (DAS28) for Rheumatoid Arthritis with C-Reactive Protein and the Bath Ankylosing Spondylitis Disease Activity Index improved, especially in the diet-placebo group (−0.6 ± 0.9, P = .004 and −1.39 ± 1.97, P = .001, respectively).
- Minimal disease activity improved in all groups.
- The diet-fish group showed significant weight loss (−1.79 ± 2.4 kg, P = .004), as well as reductions in waist circumference (−3.28 ± 3.5 cm, P < .001) and body fat (−1.2 ± 2.2 kg, P = .006).
Other findings from this study showed the following:
- No significant correlation was seen between weight loss and disease activity improvement.
- Each 1-unit increase in the Healthy Eating Index value reduced the likelihood of achieving remission by 4%.
- Each 100-calorie increase per day caused a 3.4-fold impairment on the DAS28-Erythrocyte Sedimentation Rate score.
The fact that no changes in PsA, medications, or physical activity were made during the study period reinforces the role of diet in the context of immunometabolism, the authors said. Supervised exercise, however, could contribute to weight loss, lean muscle mass, and better disease activity control.
The authors stressed that the data suggest “increased energy intake and worse diet quality may negatively affect joint activity and reduce the likelihood of achieving disease remission, regardless of weight loss or body composition changes.”
“There are other studies that have looked at the effect of weight loss from a very low-calorie diet, and they’ve suggested that PsA symptoms can improve, said rheumatologist Eric. M. Ruderman, MD, a professor of medicine at Northwestern Medicine in Chicago, in an interview. “The unique piece here is that they found that the improvement was really independent of weight loss.”
Dr. Ruderman, who was not involved in DIETA, cautioned, however, that the study is small and saw improvement in the placebo group as well, which could suggest that some of the improvement was related to the extra attention and regular communication with the nutritionist that came with participation in the study.
“Also, the absolute improvement was small, and the dietary restriction was pretty aggressive, so I’m not sure how generalizable this really is. While there are lots of benefits to maintaining a healthy diet and exercising, I don’t think that the results of this small study would justify taking an aggressive [dietary] approach as part of the clinical playbook for all PsA patients.”
This study was supported by the São Paulo Research Foundation and the Coordination for Improvement in Higher Education Foundation of the Ministry of Education, Brazil.
The authors had no competing interests to declare.
Dr. Ruderman disclosed no relevant competing interests.
, Brazilian researchers found.
Earlier research has reported that weight loss improves the symptoms of PsA.
Improvement in the Brazilian DIETA study was linked to both better eating patterns and better quality of diet, and while omega-3 supplementation caused relevant body composition changes, it did not improve disease activity, according to Beatriz F. Leite of the division of rheumatology at the Federal University of São Paulo and colleagues.
“The DIETA trial, a nonpharmacologic approach, is an inexpensive, suitable, and efficient approach that could be combined with standardized drug therapy,” the investigators wrote online in Advances in Rheumatology.
Dietary counseling aimed at losing or controlling weight could therefore be part of the global protocol for PsA patients, the researchers added. They conceded, however, that nonpharmacologic interventions traditionally have a low rate of adherence.
This recommendation aligns with a systematic review by the National Psoriasis Foundation, which found evidence of benefit with dietary weight reduction via a hypocaloric diet in overweight and obese patients with psoriasis and/or PsA.
The DIETA trial
The 12-week randomized, double-blind, placebo-controlled study, conducted at three hospitals in São Paulo from September 2012 to May 2014, assessed whether dietary changes, antioxidant supplementation, or weight loss of 5%-10% could improve skin and joint activity in 97 enrolled PsA patients.
Participants were randomized into the following supervised dietary groups:
- Diet-placebo (hypocaloric diet plus placebo supplementation).
- Diet-fish (hypocaloric diet plus 3 g/day of omega-3 supplementation).
- Placebo (with habitual diet).
Diets were carefully tailored to each individual patient. The regimen for overweight and obese patients included a 500-kcal restriction, while for eutrophic patients, diets were calculated to maintain weight with no caloric restriction.
In the 91 patients evaluable by multiple measures at 12 weeks, Ms. Leite and colleagues observed the following:
- The Disease Activity Score 28 (DAS28) for Rheumatoid Arthritis with C-Reactive Protein and the Bath Ankylosing Spondylitis Disease Activity Index improved, especially in the diet-placebo group (−0.6 ± 0.9, P = .004 and −1.39 ± 1.97, P = .001, respectively).
- Minimal disease activity improved in all groups.
- The diet-fish group showed significant weight loss (−1.79 ± 2.4 kg, P = .004), as well as reductions in waist circumference (−3.28 ± 3.5 cm, P < .001) and body fat (−1.2 ± 2.2 kg, P = .006).
Other findings from this study showed the following:
- No significant correlation was seen between weight loss and disease activity improvement.
- Each 1-unit increase in the Healthy Eating Index value reduced the likelihood of achieving remission by 4%.
- Each 100-calorie increase per day caused a 3.4-fold impairment on the DAS28-Erythrocyte Sedimentation Rate score.
The fact that no changes in PsA, medications, or physical activity were made during the study period reinforces the role of diet in the context of immunometabolism, the authors said. Supervised exercise, however, could contribute to weight loss, lean muscle mass, and better disease activity control.
The authors stressed that the data suggest “increased energy intake and worse diet quality may negatively affect joint activity and reduce the likelihood of achieving disease remission, regardless of weight loss or body composition changes.”
“There are other studies that have looked at the effect of weight loss from a very low-calorie diet, and they’ve suggested that PsA symptoms can improve, said rheumatologist Eric. M. Ruderman, MD, a professor of medicine at Northwestern Medicine in Chicago, in an interview. “The unique piece here is that they found that the improvement was really independent of weight loss.”
Dr. Ruderman, who was not involved in DIETA, cautioned, however, that the study is small and saw improvement in the placebo group as well, which could suggest that some of the improvement was related to the extra attention and regular communication with the nutritionist that came with participation in the study.
“Also, the absolute improvement was small, and the dietary restriction was pretty aggressive, so I’m not sure how generalizable this really is. While there are lots of benefits to maintaining a healthy diet and exercising, I don’t think that the results of this small study would justify taking an aggressive [dietary] approach as part of the clinical playbook for all PsA patients.”
This study was supported by the São Paulo Research Foundation and the Coordination for Improvement in Higher Education Foundation of the Ministry of Education, Brazil.
The authors had no competing interests to declare.
Dr. Ruderman disclosed no relevant competing interests.
, Brazilian researchers found.
Earlier research has reported that weight loss improves the symptoms of PsA.
Improvement in the Brazilian DIETA study was linked to both better eating patterns and better quality of diet, and while omega-3 supplementation caused relevant body composition changes, it did not improve disease activity, according to Beatriz F. Leite of the division of rheumatology at the Federal University of São Paulo and colleagues.
“The DIETA trial, a nonpharmacologic approach, is an inexpensive, suitable, and efficient approach that could be combined with standardized drug therapy,” the investigators wrote online in Advances in Rheumatology.
Dietary counseling aimed at losing or controlling weight could therefore be part of the global protocol for PsA patients, the researchers added. They conceded, however, that nonpharmacologic interventions traditionally have a low rate of adherence.
This recommendation aligns with a systematic review by the National Psoriasis Foundation, which found evidence of benefit with dietary weight reduction via a hypocaloric diet in overweight and obese patients with psoriasis and/or PsA.
The DIETA trial
The 12-week randomized, double-blind, placebo-controlled study, conducted at three hospitals in São Paulo from September 2012 to May 2014, assessed whether dietary changes, antioxidant supplementation, or weight loss of 5%-10% could improve skin and joint activity in 97 enrolled PsA patients.
Participants were randomized into the following supervised dietary groups:
- Diet-placebo (hypocaloric diet plus placebo supplementation).
- Diet-fish (hypocaloric diet plus 3 g/day of omega-3 supplementation).
- Placebo (with habitual diet).
Diets were carefully tailored to each individual patient. The regimen for overweight and obese patients included a 500-kcal restriction, while for eutrophic patients, diets were calculated to maintain weight with no caloric restriction.
In the 91 patients evaluable by multiple measures at 12 weeks, Ms. Leite and colleagues observed the following:
- The Disease Activity Score 28 (DAS28) for Rheumatoid Arthritis with C-Reactive Protein and the Bath Ankylosing Spondylitis Disease Activity Index improved, especially in the diet-placebo group (−0.6 ± 0.9, P = .004 and −1.39 ± 1.97, P = .001, respectively).
- Minimal disease activity improved in all groups.
- The diet-fish group showed significant weight loss (−1.79 ± 2.4 kg, P = .004), as well as reductions in waist circumference (−3.28 ± 3.5 cm, P < .001) and body fat (−1.2 ± 2.2 kg, P = .006).
Other findings from this study showed the following:
- No significant correlation was seen between weight loss and disease activity improvement.
- Each 1-unit increase in the Healthy Eating Index value reduced the likelihood of achieving remission by 4%.
- Each 100-calorie increase per day caused a 3.4-fold impairment on the DAS28-Erythrocyte Sedimentation Rate score.
The fact that no changes in PsA, medications, or physical activity were made during the study period reinforces the role of diet in the context of immunometabolism, the authors said. Supervised exercise, however, could contribute to weight loss, lean muscle mass, and better disease activity control.
The authors stressed that the data suggest “increased energy intake and worse diet quality may negatively affect joint activity and reduce the likelihood of achieving disease remission, regardless of weight loss or body composition changes.”
“There are other studies that have looked at the effect of weight loss from a very low-calorie diet, and they’ve suggested that PsA symptoms can improve, said rheumatologist Eric. M. Ruderman, MD, a professor of medicine at Northwestern Medicine in Chicago, in an interview. “The unique piece here is that they found that the improvement was really independent of weight loss.”
Dr. Ruderman, who was not involved in DIETA, cautioned, however, that the study is small and saw improvement in the placebo group as well, which could suggest that some of the improvement was related to the extra attention and regular communication with the nutritionist that came with participation in the study.
“Also, the absolute improvement was small, and the dietary restriction was pretty aggressive, so I’m not sure how generalizable this really is. While there are lots of benefits to maintaining a healthy diet and exercising, I don’t think that the results of this small study would justify taking an aggressive [dietary] approach as part of the clinical playbook for all PsA patients.”
This study was supported by the São Paulo Research Foundation and the Coordination for Improvement in Higher Education Foundation of the Ministry of Education, Brazil.
The authors had no competing interests to declare.
Dr. Ruderman disclosed no relevant competing interests.
FROM ADVANCES IN RHEUMATOLOGY
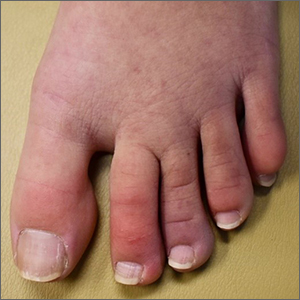
A case of cold, purple toes
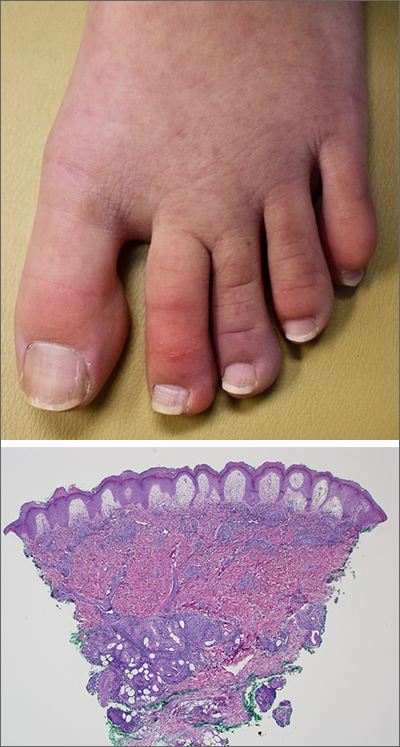
A punch-biopsy was performed on the left second toe where the erythema was the most intense. It demonstrated classic findings for pernio: superficial and deep perivascular lymphocytic inflammation and papillary dermal edema on the acral surface.
Pernio, alternatively known as chilblains, is characterized by erythema, violaceous changes, and swelling at acral sites (especially the toes or fingers). There can also be blistering, pain/tenderness, and itch. Pernio results in an abnormal localized inflammatory response to nonfreezing cold and is more common in damp climates. Pernio may also occur in occupational settings where patients handle frozen food. When a patient presents with the classic findings and consistent history, biopsy is not strictly necessary, but can aid in a definitive diagnosis.
The pathogenesis of pernio is not clearly understood. Inflammation secondary to vasospasm and type I interferon immune response to repeated or chronic cold exposure likely play a significant role. Symptoms can arise within 24 hours of exposure and resolve just as quickly. However, persistent and repeated exposure can also trigger ongoing symptoms that last for weeks.
As with most autoinflammatory conditions, pernio has a proclivity to affect younger women. It also affects children and the elderly. Because it is an inflammatory response to nonfreezing cold temperatures, the disease tends to occur during autumn in patients who live in homes without central heating.
A diagnosis of idiopathic pernio necessitates excluding several other similar, cold-induced entities. These include acrocyanosis (due to erythromelalgia, anorexia, medications), Raynaud phenomenon, cryoglobulinemia, cold urticaria, and chilblain lupus (among others). Pernio tends to lack other clinical findings such as true retiform purpura.
Of note, during the COVID-19 pandemic, physicians identified a spike in the incidence of pernio-like acral eruptions. This phenomenon has been coined “COVID toes.” While the direct temporal and causal relationships between COVID-19 and the observed eruption has not been clearly established, any patient who presents with a new onset pernio-like eruption should receive a COVID-19 test to ensure proper precautions are followed.1
In our patient, the work-up did not show any evidence of other underlying conditions. As her symptoms were minimal, we provided reassurance and counseling on preventive measures such as keeping her hands and feet warm and dry. In cases where treatment is needed, high-potency topical corticosteroids can be utilized judiciously during flares to decrease local inflammation. (There is minimal concern for adverse effects due to the thicker skin on acral surfaces.) Another treatment option is oral nifedipine (20-60 mg/d). One double-blinded trial showed it can improve symptoms in up to 70% of patients.2
Clinical image courtesy of Jiasen Wang, MD; microscopy image courtesy of Shelly Stepenaskie, MD. Text courtesy of Jiasen Wang, MD, Aimee Smidt, MD, Shelly Stepenaskie, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Cappel MA, Cappel JA, Wetter DA. Pernio (Chilblains), SARS-CoV-2, and covid toes unified through cutaneous and systemic mechanisms. Mayo Clin Proc. 2021;96:989-1005. doi: 10.1016/j.mayocp.2021.01.009
2. Simon TD, Soep JB, Hollister JR. Pernio in pediatrics. Pediatrics. 2005;116:e472-e475. doi: 10.1542/peds.2004-2681

A punch-biopsy was performed on the left second toe where the erythema was the most intense. It demonstrated classic findings for pernio: superficial and deep perivascular lymphocytic inflammation and papillary dermal edema on the acral surface.
Pernio, alternatively known as chilblains, is characterized by erythema, violaceous changes, and swelling at acral sites (especially the toes or fingers). There can also be blistering, pain/tenderness, and itch. Pernio results in an abnormal localized inflammatory response to nonfreezing cold and is more common in damp climates. Pernio may also occur in occupational settings where patients handle frozen food. When a patient presents with the classic findings and consistent history, biopsy is not strictly necessary, but can aid in a definitive diagnosis.
The pathogenesis of pernio is not clearly understood. Inflammation secondary to vasospasm and type I interferon immune response to repeated or chronic cold exposure likely play a significant role. Symptoms can arise within 24 hours of exposure and resolve just as quickly. However, persistent and repeated exposure can also trigger ongoing symptoms that last for weeks.
As with most autoinflammatory conditions, pernio has a proclivity to affect younger women. It also affects children and the elderly. Because it is an inflammatory response to nonfreezing cold temperatures, the disease tends to occur during autumn in patients who live in homes without central heating.
A diagnosis of idiopathic pernio necessitates excluding several other similar, cold-induced entities. These include acrocyanosis (due to erythromelalgia, anorexia, medications), Raynaud phenomenon, cryoglobulinemia, cold urticaria, and chilblain lupus (among others). Pernio tends to lack other clinical findings such as true retiform purpura.
Of note, during the COVID-19 pandemic, physicians identified a spike in the incidence of pernio-like acral eruptions. This phenomenon has been coined “COVID toes.” While the direct temporal and causal relationships between COVID-19 and the observed eruption has not been clearly established, any patient who presents with a new onset pernio-like eruption should receive a COVID-19 test to ensure proper precautions are followed.1
In our patient, the work-up did not show any evidence of other underlying conditions. As her symptoms were minimal, we provided reassurance and counseling on preventive measures such as keeping her hands and feet warm and dry. In cases where treatment is needed, high-potency topical corticosteroids can be utilized judiciously during flares to decrease local inflammation. (There is minimal concern for adverse effects due to the thicker skin on acral surfaces.) Another treatment option is oral nifedipine (20-60 mg/d). One double-blinded trial showed it can improve symptoms in up to 70% of patients.2
Clinical image courtesy of Jiasen Wang, MD; microscopy image courtesy of Shelly Stepenaskie, MD. Text courtesy of Jiasen Wang, MD, Aimee Smidt, MD, Shelly Stepenaskie, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A punch-biopsy was performed on the left second toe where the erythema was the most intense. It demonstrated classic findings for pernio: superficial and deep perivascular lymphocytic inflammation and papillary dermal edema on the acral surface.
Pernio, alternatively known as chilblains, is characterized by erythema, violaceous changes, and swelling at acral sites (especially the toes or fingers). There can also be blistering, pain/tenderness, and itch. Pernio results in an abnormal localized inflammatory response to nonfreezing cold and is more common in damp climates. Pernio may also occur in occupational settings where patients handle frozen food. When a patient presents with the classic findings and consistent history, biopsy is not strictly necessary, but can aid in a definitive diagnosis.
The pathogenesis of pernio is not clearly understood. Inflammation secondary to vasospasm and type I interferon immune response to repeated or chronic cold exposure likely play a significant role. Symptoms can arise within 24 hours of exposure and resolve just as quickly. However, persistent and repeated exposure can also trigger ongoing symptoms that last for weeks.
As with most autoinflammatory conditions, pernio has a proclivity to affect younger women. It also affects children and the elderly. Because it is an inflammatory response to nonfreezing cold temperatures, the disease tends to occur during autumn in patients who live in homes without central heating.
A diagnosis of idiopathic pernio necessitates excluding several other similar, cold-induced entities. These include acrocyanosis (due to erythromelalgia, anorexia, medications), Raynaud phenomenon, cryoglobulinemia, cold urticaria, and chilblain lupus (among others). Pernio tends to lack other clinical findings such as true retiform purpura.
Of note, during the COVID-19 pandemic, physicians identified a spike in the incidence of pernio-like acral eruptions. This phenomenon has been coined “COVID toes.” While the direct temporal and causal relationships between COVID-19 and the observed eruption has not been clearly established, any patient who presents with a new onset pernio-like eruption should receive a COVID-19 test to ensure proper precautions are followed.1
In our patient, the work-up did not show any evidence of other underlying conditions. As her symptoms were minimal, we provided reassurance and counseling on preventive measures such as keeping her hands and feet warm and dry. In cases where treatment is needed, high-potency topical corticosteroids can be utilized judiciously during flares to decrease local inflammation. (There is minimal concern for adverse effects due to the thicker skin on acral surfaces.) Another treatment option is oral nifedipine (20-60 mg/d). One double-blinded trial showed it can improve symptoms in up to 70% of patients.2
Clinical image courtesy of Jiasen Wang, MD; microscopy image courtesy of Shelly Stepenaskie, MD. Text courtesy of Jiasen Wang, MD, Aimee Smidt, MD, Shelly Stepenaskie, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Cappel MA, Cappel JA, Wetter DA. Pernio (Chilblains), SARS-CoV-2, and covid toes unified through cutaneous and systemic mechanisms. Mayo Clin Proc. 2021;96:989-1005. doi: 10.1016/j.mayocp.2021.01.009
2. Simon TD, Soep JB, Hollister JR. Pernio in pediatrics. Pediatrics. 2005;116:e472-e475. doi: 10.1542/peds.2004-2681
1. Cappel MA, Cappel JA, Wetter DA. Pernio (Chilblains), SARS-CoV-2, and covid toes unified through cutaneous and systemic mechanisms. Mayo Clin Proc. 2021;96:989-1005. doi: 10.1016/j.mayocp.2021.01.009
2. Simon TD, Soep JB, Hollister JR. Pernio in pediatrics. Pediatrics. 2005;116:e472-e475. doi: 10.1542/peds.2004-2681
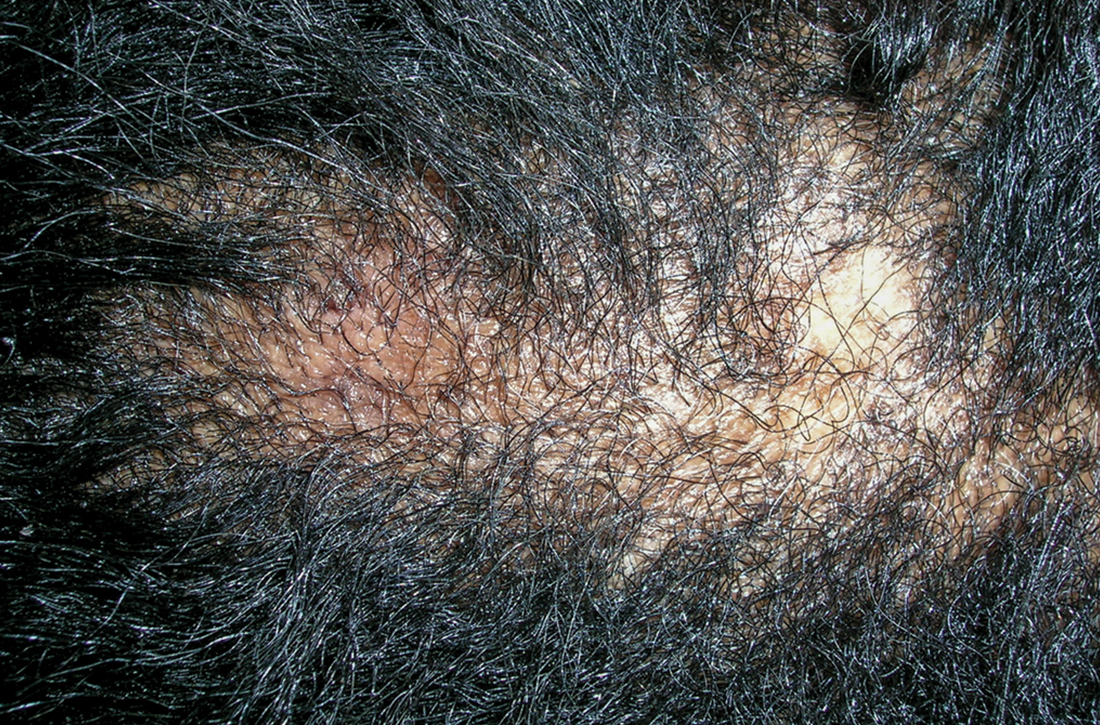
Central centrifugal cicatricial alopecia
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.
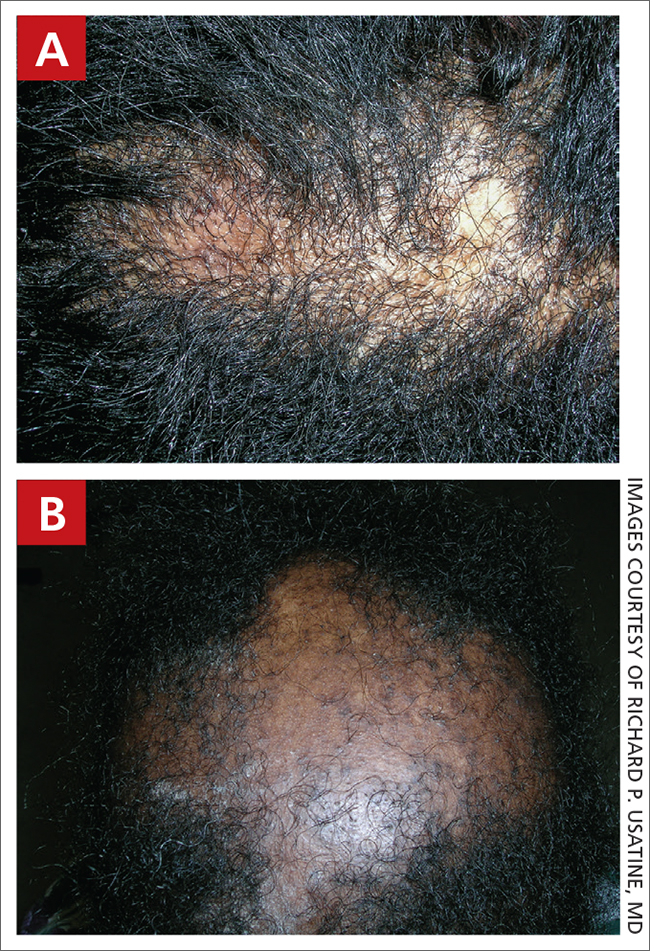
Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia.1 CCCA (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
CCCA predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. CCCA has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7 PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
- CCCA begins in the central scalp (crown area, vertex) and spreads centrifugally.
- Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
- Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
- Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
- CCCA can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Continue to: Due to the inflammatory...
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral antiinflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
1. Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/ j.1600-0560.2001.280701.x
2. Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/ archderm.147.12.1453
3. Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111/ 1346-8138.14217
4. Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
5. Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
6. Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
7. Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056/ NEJMe1900042
8. Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
9. Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
10. Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/ j.jdcr.2019.12.008.
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.

Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia.1 CCCA (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
CCCA predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. CCCA has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7 PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
- CCCA begins in the central scalp (crown area, vertex) and spreads centrifugally.
- Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
- Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
- Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
- CCCA can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Continue to: Due to the inflammatory...
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral antiinflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.

Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia.1 CCCA (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
CCCA predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. CCCA has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7 PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
- CCCA begins in the central scalp (crown area, vertex) and spreads centrifugally.
- Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
- Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
- Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
- CCCA can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Continue to: Due to the inflammatory...
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral antiinflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
1. Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/ j.1600-0560.2001.280701.x
2. Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/ archderm.147.12.1453
3. Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111/ 1346-8138.14217
4. Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
5. Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
6. Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
7. Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056/ NEJMe1900042
8. Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
9. Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
10. Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/ j.jdcr.2019.12.008.
1. Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/ j.1600-0560.2001.280701.x
2. Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/ archderm.147.12.1453
3. Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111/ 1346-8138.14217
4. Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
5. Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
6. Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
7. Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056/ NEJMe1900042
8. Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
9. Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
10. Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/ j.jdcr.2019.12.008.
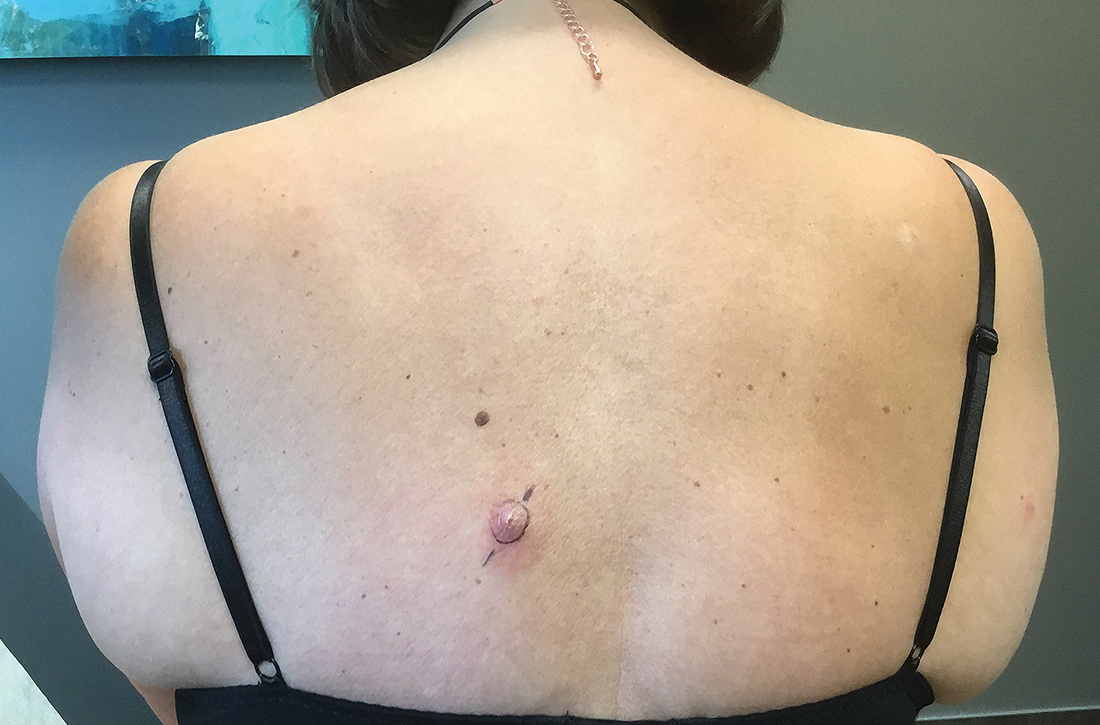
Nonhealing boils
A healthy woman in her 60s presented to the clinic with a 1-month history of red, itchy, and slightly painful nodules on the scalp and back. The patient had travelled to Belize for a vacation in the weeks prior to the onset of the lesions. She was initially given a course of cephalexin for presumed furunculosis at another clinic, without improvement.
Examination revealed inflamed nodules with a central open pore on the left upper back (FIGURE 1) and the occipital scalp. Notably, when the lesions were observed with a dermatoscope, intermittent air bubbles were seen through the skin opening.
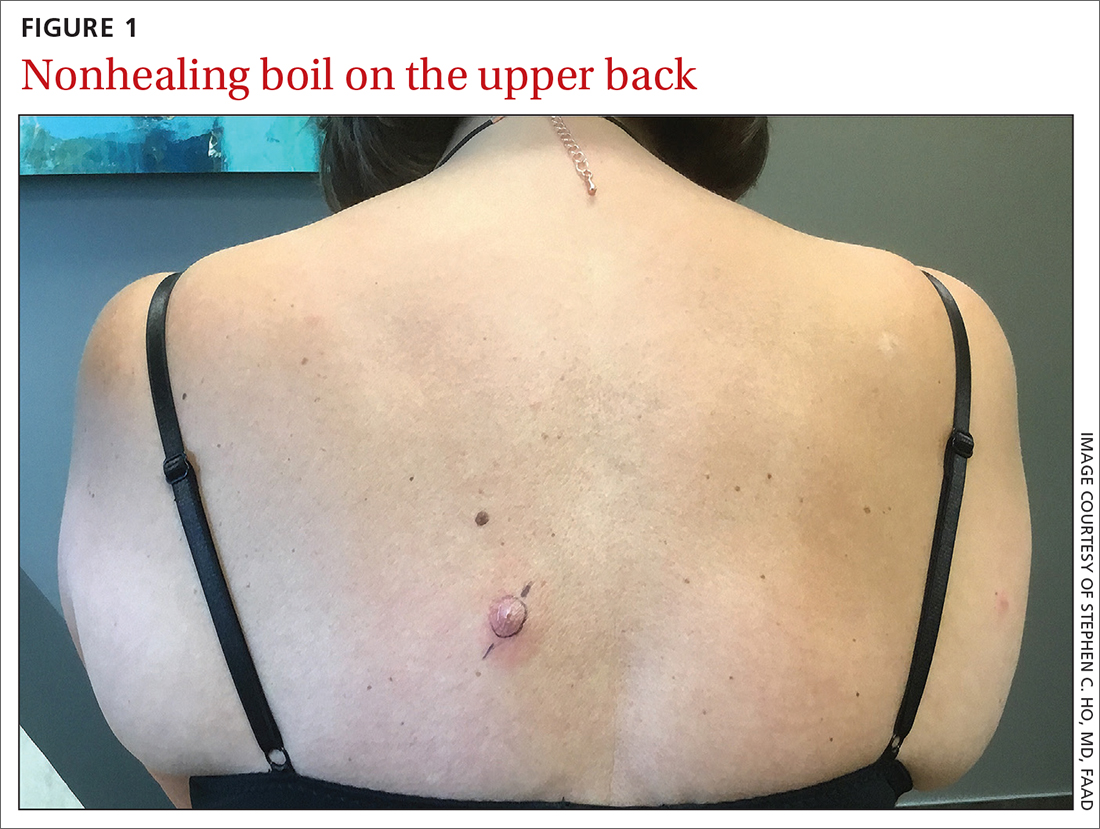
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Furuncular myiasis
Given the patient’s clinical presentation and travel history, furuncular myiasis infestation was suspected and confirmed by punch biopsy. Pathologic exam revealed botfly larvae in both wounds, consistent with the human botfly, Dermatobia hominis (FIGURE 2). Myiasis is not common in the United States but should be suspected in patients who have recently traveled to tropical or subtropical areas. Furuncular myiasis describes the condition in which fly larvae penetrate healthy skin in a localized fashion, leading to the development of a furuncle-like nodule with 1 or more larvae within it.

Dermatobia hominis is the most common causative organism for furuncular myiasis in the regions of the Americas. Patients typically present with 1 lesion on an exposed part of the body (eg, scalp, face, extremities). The lesions typically contain a central pore with purulent or serosanguinous exudate.1 Upon dermatoscopic inspection, one can often see the posterior part of the larva or the respiratory spiracles, which look like tiny black dots on the surface of the wound. The organism may also be indirectly viewed through respiratory bubble formation within the exudate.1,2 Diagnosis is confirmed by extracting the larvae from the wound and having the species identified by an experienced pathologist or parasitologist.
Mode of transmission
Phoresis is the name of the process by which Dermatobia hominis invades the skin.3 The female fly lays her eggs onto captured mosquitos using a quick-drying adhesive. The eggs are then transferred to the host by a mosquito bite. The host’s body heat induces egg hatching, and the larvae burrow into follicular openings or skin perforations. This leads to the development of a small erythematous papule, which can further lead to a furuncle-like nodule with a central pore that allows the organism to respirate. When ready to pupate, the larvae work their way to the skin surface and drop to the soil, where they can further develop. After pupation, the fly hatches and develops into an adult, and the cycle repeats.
Common infectious and inflammatory lesions are in the differential Dx
The differential diagnosis includes bacterial abscess, exaggerated arthropod reaction, and ruptured epidermal cyst. With our patient, the travel history and unique exam findings led to the suspicion of myiasis.
Bacterial abscess is likely to have more notable purulence with response to appropriate oral antibiotics and no bubbling phenomenon on exam. It is also unlikely to be present for 1 month without progressive worsening.
Continue to: Exaggerated arthropod reaction
Exaggerated arthropod reaction usually manifests as a smooth, red, papular lesion without bubble formation from a central pore.
Ruptured epidermal cyst would be considered if there was a known preexisting cyst that recently changed. No bubble formation would be observed from the central pore.
Extraction is the treatment of choice
Treatment of furuncular myiasis involves removing the larvae or forcing them out of the lesion. Wounds can be covered with a substance, such as petrolatum, nail polish, beeswax, paraffin, or mineral oil, to block respiration.3 Occlusion may be needed for 24 hours to create adequate localized hypoxia to force the larvae to migrate from the wound and allow for easier manual extraction. Surgical removal of the larvae is also effective. A cruciate incision can be made adjacent to the central pore to avoid damaging the organisms.3 A topical, broad-based antiparasitic, such as a 10% ivermectin solution, has also been successfully used to treat furuncular myiasis. This approach works by either inducing larval migration outward or simply killing the larvae.3
Our patient recovered well after we performed a punch biopsy to make a larger wound opening and remove the intact larvae.
ACKNOWLEDGMENT
We thank Richard Pollack, PhD, at IdentifyUS, LLC, for providing the botfly larvae photo.
1. Francesconi F, Lupi O. Myiasis. Clin Microbiol Rev. 2012;25:79-105. doi: 10.1128/cmr.00010-11
2. Diaz JH. The epidemiology, diagnosis, management, and prevention of ectoparasitic diseases in travelers. J Travel Med. 2006;13:100-111. doi: 10.1111/j.1708-8305.2006.00021.x
3. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926. doi: 10.1016/j.jaad.2008.03.014
A healthy woman in her 60s presented to the clinic with a 1-month history of red, itchy, and slightly painful nodules on the scalp and back. The patient had travelled to Belize for a vacation in the weeks prior to the onset of the lesions. She was initially given a course of cephalexin for presumed furunculosis at another clinic, without improvement.
Examination revealed inflamed nodules with a central open pore on the left upper back (FIGURE 1) and the occipital scalp. Notably, when the lesions were observed with a dermatoscope, intermittent air bubbles were seen through the skin opening.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Furuncular myiasis
Given the patient’s clinical presentation and travel history, furuncular myiasis infestation was suspected and confirmed by punch biopsy. Pathologic exam revealed botfly larvae in both wounds, consistent with the human botfly, Dermatobia hominis (FIGURE 2). Myiasis is not common in the United States but should be suspected in patients who have recently traveled to tropical or subtropical areas. Furuncular myiasis describes the condition in which fly larvae penetrate healthy skin in a localized fashion, leading to the development of a furuncle-like nodule with 1 or more larvae within it.

Dermatobia hominis is the most common causative organism for furuncular myiasis in the regions of the Americas. Patients typically present with 1 lesion on an exposed part of the body (eg, scalp, face, extremities). The lesions typically contain a central pore with purulent or serosanguinous exudate.1 Upon dermatoscopic inspection, one can often see the posterior part of the larva or the respiratory spiracles, which look like tiny black dots on the surface of the wound. The organism may also be indirectly viewed through respiratory bubble formation within the exudate.1,2 Diagnosis is confirmed by extracting the larvae from the wound and having the species identified by an experienced pathologist or parasitologist.
Mode of transmission
Phoresis is the name of the process by which Dermatobia hominis invades the skin.3 The female fly lays her eggs onto captured mosquitos using a quick-drying adhesive. The eggs are then transferred to the host by a mosquito bite. The host’s body heat induces egg hatching, and the larvae burrow into follicular openings or skin perforations. This leads to the development of a small erythematous papule, which can further lead to a furuncle-like nodule with a central pore that allows the organism to respirate. When ready to pupate, the larvae work their way to the skin surface and drop to the soil, where they can further develop. After pupation, the fly hatches and develops into an adult, and the cycle repeats.
Common infectious and inflammatory lesions are in the differential Dx
The differential diagnosis includes bacterial abscess, exaggerated arthropod reaction, and ruptured epidermal cyst. With our patient, the travel history and unique exam findings led to the suspicion of myiasis.
Bacterial abscess is likely to have more notable purulence with response to appropriate oral antibiotics and no bubbling phenomenon on exam. It is also unlikely to be present for 1 month without progressive worsening.
Continue to: Exaggerated arthropod reaction
Exaggerated arthropod reaction usually manifests as a smooth, red, papular lesion without bubble formation from a central pore.
Ruptured epidermal cyst would be considered if there was a known preexisting cyst that recently changed. No bubble formation would be observed from the central pore.
Extraction is the treatment of choice
Treatment of furuncular myiasis involves removing the larvae or forcing them out of the lesion. Wounds can be covered with a substance, such as petrolatum, nail polish, beeswax, paraffin, or mineral oil, to block respiration.3 Occlusion may be needed for 24 hours to create adequate localized hypoxia to force the larvae to migrate from the wound and allow for easier manual extraction. Surgical removal of the larvae is also effective. A cruciate incision can be made adjacent to the central pore to avoid damaging the organisms.3 A topical, broad-based antiparasitic, such as a 10% ivermectin solution, has also been successfully used to treat furuncular myiasis. This approach works by either inducing larval migration outward or simply killing the larvae.3
Our patient recovered well after we performed a punch biopsy to make a larger wound opening and remove the intact larvae.
ACKNOWLEDGMENT
We thank Richard Pollack, PhD, at IdentifyUS, LLC, for providing the botfly larvae photo.
A healthy woman in her 60s presented to the clinic with a 1-month history of red, itchy, and slightly painful nodules on the scalp and back. The patient had travelled to Belize for a vacation in the weeks prior to the onset of the lesions. She was initially given a course of cephalexin for presumed furunculosis at another clinic, without improvement.
Examination revealed inflamed nodules with a central open pore on the left upper back (FIGURE 1) and the occipital scalp. Notably, when the lesions were observed with a dermatoscope, intermittent air bubbles were seen through the skin opening.

WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Furuncular myiasis
Given the patient’s clinical presentation and travel history, furuncular myiasis infestation was suspected and confirmed by punch biopsy. Pathologic exam revealed botfly larvae in both wounds, consistent with the human botfly, Dermatobia hominis (FIGURE 2). Myiasis is not common in the United States but should be suspected in patients who have recently traveled to tropical or subtropical areas. Furuncular myiasis describes the condition in which fly larvae penetrate healthy skin in a localized fashion, leading to the development of a furuncle-like nodule with 1 or more larvae within it.

Dermatobia hominis is the most common causative organism for furuncular myiasis in the regions of the Americas. Patients typically present with 1 lesion on an exposed part of the body (eg, scalp, face, extremities). The lesions typically contain a central pore with purulent or serosanguinous exudate.1 Upon dermatoscopic inspection, one can often see the posterior part of the larva or the respiratory spiracles, which look like tiny black dots on the surface of the wound. The organism may also be indirectly viewed through respiratory bubble formation within the exudate.1,2 Diagnosis is confirmed by extracting the larvae from the wound and having the species identified by an experienced pathologist or parasitologist.
Mode of transmission
Phoresis is the name of the process by which Dermatobia hominis invades the skin.3 The female fly lays her eggs onto captured mosquitos using a quick-drying adhesive. The eggs are then transferred to the host by a mosquito bite. The host’s body heat induces egg hatching, and the larvae burrow into follicular openings or skin perforations. This leads to the development of a small erythematous papule, which can further lead to a furuncle-like nodule with a central pore that allows the organism to respirate. When ready to pupate, the larvae work their way to the skin surface and drop to the soil, where they can further develop. After pupation, the fly hatches and develops into an adult, and the cycle repeats.
Common infectious and inflammatory lesions are in the differential Dx
The differential diagnosis includes bacterial abscess, exaggerated arthropod reaction, and ruptured epidermal cyst. With our patient, the travel history and unique exam findings led to the suspicion of myiasis.
Bacterial abscess is likely to have more notable purulence with response to appropriate oral antibiotics and no bubbling phenomenon on exam. It is also unlikely to be present for 1 month without progressive worsening.
Continue to: Exaggerated arthropod reaction
Exaggerated arthropod reaction usually manifests as a smooth, red, papular lesion without bubble formation from a central pore.
Ruptured epidermal cyst would be considered if there was a known preexisting cyst that recently changed. No bubble formation would be observed from the central pore.
Extraction is the treatment of choice
Treatment of furuncular myiasis involves removing the larvae or forcing them out of the lesion. Wounds can be covered with a substance, such as petrolatum, nail polish, beeswax, paraffin, or mineral oil, to block respiration.3 Occlusion may be needed for 24 hours to create adequate localized hypoxia to force the larvae to migrate from the wound and allow for easier manual extraction. Surgical removal of the larvae is also effective. A cruciate incision can be made adjacent to the central pore to avoid damaging the organisms.3 A topical, broad-based antiparasitic, such as a 10% ivermectin solution, has also been successfully used to treat furuncular myiasis. This approach works by either inducing larval migration outward or simply killing the larvae.3
Our patient recovered well after we performed a punch biopsy to make a larger wound opening and remove the intact larvae.
ACKNOWLEDGMENT
We thank Richard Pollack, PhD, at IdentifyUS, LLC, for providing the botfly larvae photo.
1. Francesconi F, Lupi O. Myiasis. Clin Microbiol Rev. 2012;25:79-105. doi: 10.1128/cmr.00010-11
2. Diaz JH. The epidemiology, diagnosis, management, and prevention of ectoparasitic diseases in travelers. J Travel Med. 2006;13:100-111. doi: 10.1111/j.1708-8305.2006.00021.x
3. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926. doi: 10.1016/j.jaad.2008.03.014
1. Francesconi F, Lupi O. Myiasis. Clin Microbiol Rev. 2012;25:79-105. doi: 10.1128/cmr.00010-11
2. Diaz JH. The epidemiology, diagnosis, management, and prevention of ectoparasitic diseases in travelers. J Travel Med. 2006;13:100-111. doi: 10.1111/j.1708-8305.2006.00021.x
3. McGraw TA, Turiansky GW. Cutaneous myiasis. J Am Acad Dermatol. 2008;58:907-926. doi: 10.1016/j.jaad.2008.03.014
PLA testing brings nuance to the diagnosis of early-stage melanoma
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
AT AAD 22
Study finds discrepancies in biopsy decisions, diagnoses based on skin type
BOSTON – compared with White patients, new research shows.
“Our findings suggest diagnostic biases based on skin color exist in dermatology practice,” lead author Loren Krueger, MD, assistant professor in the department of dermatology, Emory University School of Medicine, Atlanta, said at the Annual Skin of Color Society Scientific Symposium. “A lower likelihood of biopsy of malignancy in darker skin types could contribute to disparities in cutaneous malignancies,” she added.
Disparities in dermatologic care among Black patients, compared with White patients, have been well documented. Recent evidence includes a 2020 study that showed significant shortcomings among medical students in correctly diagnosing squamous cell carcinoma, urticaria, and atopic dermatitis for patients with skin of color.
“It’s no secret that our images do not accurately or in the right quantity include skin of color,” Dr. Krueger said. “Yet few papers talk about how these biases actually impact our care. Importantly, this study demonstrates that diagnostic bias develops as early as the medical student level.”
To further investigate the role of skin color in the assessment of neoplastic and inflammatory skin conditions and decisions to perform biopsy, Dr. Krueger and her colleagues surveyed 144 dermatology residents and attending dermatologists to evaluate their clinical decisionmaking skills in assessing skin conditions for patients with lighter skin and those with darker skin. Almost 80% (113) provided complete responses and were included in the study.
For the survey, participants were shown photos of 10 neoplastic and 10 inflammatory skin conditions. Each image was matched in lighter (skin types I-II) and darker (skin types IV-VI) skinned patients in random order. Participants were asked to identify the suspected underlying etiology (neoplastic–benign, neoplastic–malignant, papulosquamous, lichenoid, infectious, bullous, or no suspected etiology) and whether they would choose to perform biopsy for the pictured condition.
Overall, their responses showed a slightly higher probability of recommending a biopsy for patients with skin types IV-V (odds ratio, 1.18; P = .054).
However, respondents were more than twice as likely to recommend a biopsy for benign neoplasms for patients with skin of color, compared with those with lighter skin types (OR, 2.57; P < .0001). They were significantly less likely to recommend a biopsy for a malignant neoplasm for patients with skin of color (OR, 0.42; P < .0001).
In addition, the correct etiology was much more commonly missed in diagnosing patients with skin of color, even after adjusting for years in dermatology practice (OR, 0.569; P < .0001).
Conversely, respondents were significantly less likely to recommend a biopsy for benign neoplasms and were more likely to recommend a biopsy for malignant neoplasms among White patients. Etiology was more commonly correct.
The findings underscore that “for skin of color patients, you’re more likely to have a benign neoplasm biopsied, you’re less likely to have a malignant neoplasm biopsied, and more often, your etiology may be missed,” Dr. Krueger said at the meeting.
Of note, while 45% of respondents were dermatology residents or fellows, 20.4% had 1-5 years of experience, and about 28% had 10 to more than 25 years of experience.
And while 75% of the dermatology residents, fellows, and attendings were White, there was no difference in the probability of correctly identifying the underlying etiology in dark or light skin types based on the provider’s self-identified race.
Importantly, the patterns in the study of diagnostic discrepancies are reflected in broader dermatologic outcomes. The 5-year melanoma survival rate is 74.1% among Black patients and 92.9% among White patients. Dr. Krueger referred to data showing that only 52.6% of Black patients have stage I melanoma at diagnosis, whereas among White patients, the rate is much higher, at 75.9%.
“We know skin malignancy can be more aggressive and late-stage in skin of color populations, leading to increased morbidity and later stage at initial diagnosis,” Dr. Krueger told this news organization. “We routinely attribute this to limited access to care and lack of awareness on skin malignancy. However, we have no evidence on how we, as dermatologists, may be playing a role.”
Furthermore, the decision to perform biopsy or not can affect the size and stage at diagnosis of a cutaneous malignancy, she noted.
Key changes needed to prevent the disparities – and their implications – should start at the training level, she emphasized. “I would love to see increased photo representation in training materials – this is a great place to start,” Dr. Krueger said.
In addition, “encouraging medical students, residents, and dermatologists to learn from skin of color experts is vital,” she said. “We should also provide hands-on experience and training with diverse patient populations.”
The first step to addressing biases “is to acknowledge they exist,” Dr. Krueger added. “I am hopeful this inspires others to continue to investigate these biases, as well as how we can eliminate them.”
The study was funded by the Rudin Resident Research Award. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – compared with White patients, new research shows.
“Our findings suggest diagnostic biases based on skin color exist in dermatology practice,” lead author Loren Krueger, MD, assistant professor in the department of dermatology, Emory University School of Medicine, Atlanta, said at the Annual Skin of Color Society Scientific Symposium. “A lower likelihood of biopsy of malignancy in darker skin types could contribute to disparities in cutaneous malignancies,” she added.
Disparities in dermatologic care among Black patients, compared with White patients, have been well documented. Recent evidence includes a 2020 study that showed significant shortcomings among medical students in correctly diagnosing squamous cell carcinoma, urticaria, and atopic dermatitis for patients with skin of color.
“It’s no secret that our images do not accurately or in the right quantity include skin of color,” Dr. Krueger said. “Yet few papers talk about how these biases actually impact our care. Importantly, this study demonstrates that diagnostic bias develops as early as the medical student level.”
To further investigate the role of skin color in the assessment of neoplastic and inflammatory skin conditions and decisions to perform biopsy, Dr. Krueger and her colleagues surveyed 144 dermatology residents and attending dermatologists to evaluate their clinical decisionmaking skills in assessing skin conditions for patients with lighter skin and those with darker skin. Almost 80% (113) provided complete responses and were included in the study.
For the survey, participants were shown photos of 10 neoplastic and 10 inflammatory skin conditions. Each image was matched in lighter (skin types I-II) and darker (skin types IV-VI) skinned patients in random order. Participants were asked to identify the suspected underlying etiology (neoplastic–benign, neoplastic–malignant, papulosquamous, lichenoid, infectious, bullous, or no suspected etiology) and whether they would choose to perform biopsy for the pictured condition.
Overall, their responses showed a slightly higher probability of recommending a biopsy for patients with skin types IV-V (odds ratio, 1.18; P = .054).
However, respondents were more than twice as likely to recommend a biopsy for benign neoplasms for patients with skin of color, compared with those with lighter skin types (OR, 2.57; P < .0001). They were significantly less likely to recommend a biopsy for a malignant neoplasm for patients with skin of color (OR, 0.42; P < .0001).
In addition, the correct etiology was much more commonly missed in diagnosing patients with skin of color, even after adjusting for years in dermatology practice (OR, 0.569; P < .0001).
Conversely, respondents were significantly less likely to recommend a biopsy for benign neoplasms and were more likely to recommend a biopsy for malignant neoplasms among White patients. Etiology was more commonly correct.
The findings underscore that “for skin of color patients, you’re more likely to have a benign neoplasm biopsied, you’re less likely to have a malignant neoplasm biopsied, and more often, your etiology may be missed,” Dr. Krueger said at the meeting.
Of note, while 45% of respondents were dermatology residents or fellows, 20.4% had 1-5 years of experience, and about 28% had 10 to more than 25 years of experience.
And while 75% of the dermatology residents, fellows, and attendings were White, there was no difference in the probability of correctly identifying the underlying etiology in dark or light skin types based on the provider’s self-identified race.
Importantly, the patterns in the study of diagnostic discrepancies are reflected in broader dermatologic outcomes. The 5-year melanoma survival rate is 74.1% among Black patients and 92.9% among White patients. Dr. Krueger referred to data showing that only 52.6% of Black patients have stage I melanoma at diagnosis, whereas among White patients, the rate is much higher, at 75.9%.
“We know skin malignancy can be more aggressive and late-stage in skin of color populations, leading to increased morbidity and later stage at initial diagnosis,” Dr. Krueger told this news organization. “We routinely attribute this to limited access to care and lack of awareness on skin malignancy. However, we have no evidence on how we, as dermatologists, may be playing a role.”
Furthermore, the decision to perform biopsy or not can affect the size and stage at diagnosis of a cutaneous malignancy, she noted.
Key changes needed to prevent the disparities – and their implications – should start at the training level, she emphasized. “I would love to see increased photo representation in training materials – this is a great place to start,” Dr. Krueger said.
In addition, “encouraging medical students, residents, and dermatologists to learn from skin of color experts is vital,” she said. “We should also provide hands-on experience and training with diverse patient populations.”
The first step to addressing biases “is to acknowledge they exist,” Dr. Krueger added. “I am hopeful this inspires others to continue to investigate these biases, as well as how we can eliminate them.”
The study was funded by the Rudin Resident Research Award. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
BOSTON – compared with White patients, new research shows.
“Our findings suggest diagnostic biases based on skin color exist in dermatology practice,” lead author Loren Krueger, MD, assistant professor in the department of dermatology, Emory University School of Medicine, Atlanta, said at the Annual Skin of Color Society Scientific Symposium. “A lower likelihood of biopsy of malignancy in darker skin types could contribute to disparities in cutaneous malignancies,” she added.
Disparities in dermatologic care among Black patients, compared with White patients, have been well documented. Recent evidence includes a 2020 study that showed significant shortcomings among medical students in correctly diagnosing squamous cell carcinoma, urticaria, and atopic dermatitis for patients with skin of color.
“It’s no secret that our images do not accurately or in the right quantity include skin of color,” Dr. Krueger said. “Yet few papers talk about how these biases actually impact our care. Importantly, this study demonstrates that diagnostic bias develops as early as the medical student level.”
To further investigate the role of skin color in the assessment of neoplastic and inflammatory skin conditions and decisions to perform biopsy, Dr. Krueger and her colleagues surveyed 144 dermatology residents and attending dermatologists to evaluate their clinical decisionmaking skills in assessing skin conditions for patients with lighter skin and those with darker skin. Almost 80% (113) provided complete responses and were included in the study.
For the survey, participants were shown photos of 10 neoplastic and 10 inflammatory skin conditions. Each image was matched in lighter (skin types I-II) and darker (skin types IV-VI) skinned patients in random order. Participants were asked to identify the suspected underlying etiology (neoplastic–benign, neoplastic–malignant, papulosquamous, lichenoid, infectious, bullous, or no suspected etiology) and whether they would choose to perform biopsy for the pictured condition.
Overall, their responses showed a slightly higher probability of recommending a biopsy for patients with skin types IV-V (odds ratio, 1.18; P = .054).
However, respondents were more than twice as likely to recommend a biopsy for benign neoplasms for patients with skin of color, compared with those with lighter skin types (OR, 2.57; P < .0001). They were significantly less likely to recommend a biopsy for a malignant neoplasm for patients with skin of color (OR, 0.42; P < .0001).
In addition, the correct etiology was much more commonly missed in diagnosing patients with skin of color, even after adjusting for years in dermatology practice (OR, 0.569; P < .0001).
Conversely, respondents were significantly less likely to recommend a biopsy for benign neoplasms and were more likely to recommend a biopsy for malignant neoplasms among White patients. Etiology was more commonly correct.
The findings underscore that “for skin of color patients, you’re more likely to have a benign neoplasm biopsied, you’re less likely to have a malignant neoplasm biopsied, and more often, your etiology may be missed,” Dr. Krueger said at the meeting.
Of note, while 45% of respondents were dermatology residents or fellows, 20.4% had 1-5 years of experience, and about 28% had 10 to more than 25 years of experience.
And while 75% of the dermatology residents, fellows, and attendings were White, there was no difference in the probability of correctly identifying the underlying etiology in dark or light skin types based on the provider’s self-identified race.
Importantly, the patterns in the study of diagnostic discrepancies are reflected in broader dermatologic outcomes. The 5-year melanoma survival rate is 74.1% among Black patients and 92.9% among White patients. Dr. Krueger referred to data showing that only 52.6% of Black patients have stage I melanoma at diagnosis, whereas among White patients, the rate is much higher, at 75.9%.
“We know skin malignancy can be more aggressive and late-stage in skin of color populations, leading to increased morbidity and later stage at initial diagnosis,” Dr. Krueger told this news organization. “We routinely attribute this to limited access to care and lack of awareness on skin malignancy. However, we have no evidence on how we, as dermatologists, may be playing a role.”
Furthermore, the decision to perform biopsy or not can affect the size and stage at diagnosis of a cutaneous malignancy, she noted.
Key changes needed to prevent the disparities – and their implications – should start at the training level, she emphasized. “I would love to see increased photo representation in training materials – this is a great place to start,” Dr. Krueger said.
In addition, “encouraging medical students, residents, and dermatologists to learn from skin of color experts is vital,” she said. “We should also provide hands-on experience and training with diverse patient populations.”
The first step to addressing biases “is to acknowledge they exist,” Dr. Krueger added. “I am hopeful this inspires others to continue to investigate these biases, as well as how we can eliminate them.”
The study was funded by the Rudin Resident Research Award. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Apremilast has neutral effect on vascular inflammation in psoriasis study
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
BOSTON – Treatment with , and glucose metabolism, in a study presented at the 2022 American Academy of Dermatology annual meeting.
In the phase 4, open-label, single arm trial, participants also lost subcutaneous and visceral fat after 16 weeks on the oral medication, a phosphodiesterase 4 (PDE4) inhibitor, and maintained that loss at 52 weeks.
People with psoriasis have an increased risk of obesity, type 2 diabetes, and cardiovascular events. Patients with more significant psoriasis “tend to die about 5 years younger than they should, based on their risk factors for mortality,” Joel Gelfand, MD, MSCE, professor of dermatology and epidemiology and vice chair of clinical research in dermatology at the University of Pennsylvania Perelman School of Medicine, Philadelphia, told this news organization.
He led the research and presented the findings at the AAD meeting March 26. “As a result, there has been a keen interest in understanding how psoriasis therapies impact cardiovascular risk, the idea being that by controlling inflammation, you may lower the risk of these patients developing cardiovascular disease over time,” he said.
Previous trials looking at the effect of psoriasis therapies on vascular inflammation “have been, for the most part, inconclusive,” Michael Garshick, MD, a cardiologist at NYU Langone Health, told this news organization. Dr. Garshick was not involved with the research. A 2021 systematic review of psoriasis clinicals trials reported that the tumor necrosis factor (TNF) blocker adalimumab (Humira) and phototherapy had the greatest effect on cardiometabolic markers, while ustekinumab (Stelara), an interleukin (IL)-12 and IL-23 antagonist, was the only treatment that improved vascular inflammation. These variable findings make this area “ripe for study,” noted Dr. Garshick.
To observe how apremilast, which is approved by the FDA for treating psoriasis and psoriatic arthritis, affected vascular inflammation, adiposity, and blood-based cardiometabolic markers, Dr. Gelfand organized an open-label study in adults with moderate-to-severe psoriasis. All participants were 18 years or older, had psoriasis for at least 6 months, and were candidates for systemic therapy. All patients underwent FDG PET/CT scans to assess aortic vascular inflammation and had blood work at baseline. Of the 70 patients originally enrolled in the study, 60 remained in the study at week 16, including 57 who underwent imaging for the second time. Thirty-nine participants remained in the study until week 52, and all except one had another scan.
The average age of participants was 47 years, and their mean BMI was 30. More than 80% of participants were White (83%) and 77% were male. The study population had lived with psoriasis for an average of 16 years and 8 patients also had psoriatic arthritis. At baseline, on average, participants had a Psoriasis Area and Severity Index (PASI) score of 18.62, a dermatology life quality index (DLQI) score of 11.60, and 22% of participants’ BSA (body surface area) were affected. The mean TBRmax, the marker for vascular inflammation, was 1.61.
Treatment responses were as expected for apremilast, with 35% of patients achieving PASI 75 and 65% of participants reporting DLQI scores of 5 or less by 16 weeks. At 52 weeks, 31% of the cohort had achieved PASI 75, and 67% reported DLQI score of 5 or higher. All psoriasis endpoints had improved since baseline (P = .001).
Throughout the study period, there was no significant change in TBRmax. However, in a sensitivity analysis, the 16 patients with a baseline TBRmax of 1.6 or higher had an absolute reduction of 0.21 in TBR by week 52. “That suggests that maybe a subset of people who have higher levels of aortic inflammation at baseline may experience some reduction that portend, potentially, some health benefits over time,” Dr. Gelfand said. “Ultimately, I wouldn’t hang my hat on the finding,” he said, noting that additional research comparing the treatment to placebo is necessary.
Both visceral and subcutaneous adipose tissue (VAT and SAT) decreased by week 16, and this reduction was maintained through week 52. In the first 16 weeks of the study, VAT decreased by 5.32% (P = .0009), and SAT decreased by 5.53% (P = .0005). From baseline to 52 weeks, VAT decreased by 5.52% (P = .0148), and SAT decreased by 5.50% (P = .0096). There were no significant differences between week 16 and week 52 in VAT or SAT.
Of the 68 blood biomarkers analyzed, there were significant decreases in the inflammatory markers ferritin (P = .015) and IL-beta (P = .006), the lipid metabolism biomarker HDL-cholesterol efflux (P = .008), and ketone bodies (P = .006). There were also increases in the inflammatory marker IL-8 (P = .003), the lipid metabolism marker ApoA (P = .05), and insulin (P = .05). Ferritin was the only biomarker that was reduced on both week 16 and week 52.
“If you want to be a purist, this was a negative trial,” said Dr. Garshick, because apremilast was not found to decrease vascular inflammation; however, he noted that the biomarker changes “were hopeful secondary endpoints.” It could be, he said, that another outcome measure may be better able to show changes in vascular inflammation compared with FDG. “It’s always hard to figure out what a good surrogate endpoint is in cardiovascular trials,” he noted, “so it may be that FDG/PET is too noisy or not reliable enough to see the outcome that we want to see.”
Dr. Gelfand reports consulting fees/grants from Amgen, AbbVie, BMS, Boehringer Ingelheim, Janssen Biologics, Novartis Corp, Pfizer, and UCB (DSMB). He serves as the Deputy Editor for the Journal of Investigative Dermatology and the Chief Medical Editor at Healio Psoriatic Disease and receives honoraria for both roles. Dr. Garshick has received consulting fees from AbbVie.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Trichotillomania: What you should know about this common hair-pulling disorder
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
Trichotillomania is a chronic psychiatric disorder that causes people to repeatedly pull out their own hair. Not only does it result in alopecia with no other underlying causes but it can have significant psychosocial ramifications and rare, but serious, complications. Though the reported prevalence rates are up to approximately 2%, it’s probable that you’ll come upon a patient suffering with this disorder at your practice, if you haven’t already.
To find out more about the best methods for diagnosing and treating this disorder, we spoke with Jon E. Grant, JD, MD, MPH, a leading trichotillomania researcher and part of the department of psychiatry and behavioral neuroscience at the University of Chicago.
Defining trichotillomania
What were the earliest descriptions of trichotillomania in medical literature?
The first real discussion of it probably goes back to Hippocrates, but from a modern medical perspective, discussion began in the 19th century with reports from the French dermatologist François Hallopeau.
They didn’t really call them disorders then – it was long before the Diagnostic and Statistical Manual of Mental Disorders (DSM-5) – but they described this in young men who kept pulling their hair for unclear reasons. These early case reports don’t provide a lot of psychological perspective, but they seem consistent with what we see now.
What are the diagnostic criteria for trichotillomania?
The current DSM-5 criteria are recurrent pulling out of hair, an inability to stop it, the pulling resulting in some noticeable thinning or hair loss, and that it causes some level of distress or some type of impairment in functioning.
At what age do most people experience an onset of symptoms?
Generally speaking, it’s in early adolescence, post puberty, around 12-15 years of age. Having said that, we do see children as young as 1-2 years who are pulling their hair, and we occasionally see somebody far older who is doing it for the first time, a sort of geriatric onset.
Overlap and differences with other disorders
You’ve written that although trichotillomania is grouped with obsessive-compulsive disorder (OCD) in the DSM-5, the thinking around that has recently shifted. Why is that?
At first, it was noticed that many of these people pulled their hair repetitively in an almost ritualized manner, perhaps every night before bed. That looked like a compulsion of OCD.
When DSM-5 came out in 2013, they grouped it with OCD. Yet people shifted to thinking that it’s kind of a cousin of OCD because it has this compulsive quality but doesn’t really have obsessive thinking that drives it. Many people just pull their hair. They’re not even always aware of it: sometimes yes, sometimes no.
We know that it has some links to OCD. You’ll see more OCD in folks with trichotillomania, but it clearly is not just the same as OCD. One of the biggest pieces of evidence for that is that our first-line treatment for OCD – a selective serotonin reuptake inhibitor antidepressant – does not really help hair pulling.
Having said that, if people are looking for help with trichotillomania, they often are best served by therapists and doctors who have a familiarity with OCD and have kept it on their radar over the past couple of decades.
How does trichotillomania overlap with skin picking disorder, which is another condition that you’ve closely researched?
It does have some overlap with skin picking in the sense that it often seems familial. For example, the mother may pull her hair and child picks their skin.
It also has a fair amount of comorbidity with skin picking. Many people who pull will pick a little bit or did at some point. Many people who pick pulled their hair at some point. It seems closely related to nail biting as well.
Studies have also shown that one of the things that runs in the histories of most families of people with trichotillomania might be substance abuse – alcohol or drug addiction.
All of this has led people to believe that there might be subtypes of trichotillomania: one that’s more like an OCD and one that’s more like an addiction. That’s similar to the debate with other mental health conditions, that there are probably multiple types of depression, multiple types of schizophrenia.
Is there a component of this that could be defined as self-harm?
That’s been its own debate. It doesn’t seem to have the same developmental trajectory that we see with self-harm, or even some of the personality features.
However, there may be a small segment of folks with trichotillomania that might more appropriately fit that category. For example, those with family histories of trauma, higher rates of posttraumatic stress disorder, or borderline personality. But it wouldn’t be the majority.
The problem is, if you look at some of the pediatrician data, they often group picking, pulling, and cutting. I think that’s far too all-inclusive.
A gap in clinician education
Are adolescent patients likely to self-report this behavior, or is it something that physicians need to suss out for themselves?
Clearly, if child psychologists, psychiatrists, or pediatricians see young people with patches of alopecia – eyebrows or eyelashes missing, head hair with spots – in addition to a dermatologic assessment, they should simply ask, “Do you pull your hair?”
But it’s interesting that with the internet, young people are much more likely to disclose and actually come forward and tell their parents that they think they have trichotillomania.
I also hear from a lot of the adolescents that they have to educate their doctors about trichotillomania because so often physicians don’t know much about it and will assume that it’s self-injury or just a symptom of anxiety. It’s a little bit of a flip from what we might have seen 20 years ago.
I’ve seen several patients who’ve said, basically, “I’m tired of no professionals seeming to know about this. I shouldn’t have to be educating my doctors about this.” I tell them that I completely agree. It’s a shame because if a doctor doesn’t know about it, then how can they get the appropriate care?
What are the complications that accompany trichotillomania?
A small percentage, maybe about 10%, will ingest their hair, much like people who bite and swallow their fingernails. The concern there is that because hair is nondigestible, it could create an intestinal plug that could rupture and be potentially life-threatening. That makes it all the more important to ask those who pull their hair what they do with the hair once they pull it.
However, with most people, the real problem is with self-esteem. Young people may not want to socialize, go on dates, or do other things they would normally do because of it. In adults, you may find that they’re far more educated than their job allows but don’t want to go to an interview because they don’t want to have somebody sit there and look at them and notice that perhaps they don’t have any eyebrows, or that they’re wearing a wig. Those psychosocial implications are huge for so many people.
Treatment options
In a 2021 study, you showed that nearly one-quarter of people with trichotillomania do naturally recover from it. What characteristics do they seem to have?
It’s interesting because we see natural recovery across many mental health problems: alcohol addition, gambling, OCD. The question then becomes why is that some people can seemingly just stop doing a behavior? Can we learn from those people?
We did see that those who naturally recovered were less likely to have some other mental health comorbidities. It seems like when you have other things such as skin picking or OCD plus trichotillomania, that it probably speaks to something that perhaps synergistically is keeping it going. But this is just a first study; learning how to harness and understand it is the next step.
What’s the goal of treating trichotillomania?
The desired goal is zero pulling. The realistic goal is more likely significantly reduced pulling that then leads to greater function in life, greater quality-of-life.
One doesn’t have to go from 100 to 0 in order to do that. I always tell people that maybe every now and then, every few months, when something is going on in life, you might find yourself pulling a hair or two. That’s okay. If you’re not pulling every day and it’s significantly reduced, we’ll call that a success. I think that setting reasonable goals at this point is really important.
And what would the treatment pathway look like for most patients?
The standard approach is probably some type of habit-reversal therapy, of which there have been many variants over the years. It involves doing something different with your hand, identifying the triggers that may set you off, and then doing something in response to those triggers that is not pulling and might neutralize whatever that anxious or stressed feeling is. That could be different with each person.
At this point, there is no drug approved by the U.S. Food and Drug Administration for trichotillomania. Our best approaches have included N-acetylcysteine, a glutamate modulator, which we’ve done research in.
That’s kind of a go-to option for people because its side-effect profile is generally innocuous. The data show that it could be beneficial in many people with very few, if any, side effects. That would be one “medication,” although it’s actually an over-the-counter vitamin. But we’re constantly looking for better and better treatments.
Do you have any final advice for clinicians or researchers?
Given how common it is, I don’t think clinicians should just see it as an innocuous little habit that people should be able to stop on their own. Clinicians should educate themselves about trichotillomania and know where the person should get the appropriate care.
From the research perspective, given the fact that we see this in animals of multiple species – that they overgroom – this seems to be deeply ingrained in us as animals. So when it comes to the underlying neuroscience, people should pay more attention because it probably has a lot to do with our understanding of habit and compulsive behaviors. It arguably can cut across a lot of different behaviors.
A version of this article first appeared on Medscape.com.
Bilateral palmar rash
A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855

A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A biopsy was performed and the pathology report showed ectatic, thin-walled vessels consistent with telangiectasias. There were no other inflammatory, infectious, or malignant changes.
Telangiectasias are caused by permanent dilatation of subpapillary plexus end vessels. Unlike petechiae and angiomata, telangiectasias blanch with pressure. They usually manifest as small, bright red, nonpulsatile vascular lesions with a fine, netlike pattern on the surface of the skin. Telangiectasis can affect many organs (eg, intestines, bladder, brain, eyes) and may occur in patients with certain genetic disorders and environmental exposures (eg, radiation).1
Palmar telangiectasias are specifically associated with hereditary hemorrhagic telangiectasia, dermatomyositis, Grave disease, CREST syndrome, systemic lupus erythematosus, and smoking.2 Sun exposure and smoking are the main risk factors for the development of telangiectasias.1
This patient had no history of autoimmune disease or hyperthyroidism, and no one in her family had telangiectasis. Thus, the likely cause of her lesions was smoking. While the pathophysiology is not fully understood, it is likely related to the vasoconstrictive quality of nicotine, causing ischemia in the dermis. This chronic, low-grade ischemia may trigger the compensatory development of telangiectasias.2
This patient was informed that her telangiectasias were most likely caused by her smoking and that the lesions themselves did not require treatment. She was encouraged to continue her smoking cessation efforts with her primary care provider.
Photos courtesy of Daniel Stulberg, MD. Text courtesy of Mia MJ Coleman, BA, BS, University of New Mexico School of Medicine, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855
1. Schieving JH, Shoenaker MHD, Weemaes CM, et al. Telangiectasias: Small lesions referring to serious disorders. Eur J Paediatr Neurol. 2017;21:807-815. doi: 10.1016/j.ejpn.2017.07.016
2. Levi A, Shechter R, Lapidoth M, et al. Palmar telangiectasias: a cutaneous sign for smoking. Dermatology. 2017;233:390-395. doi: 10.1159/000481855