User login
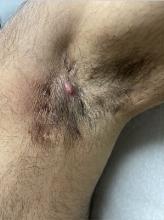
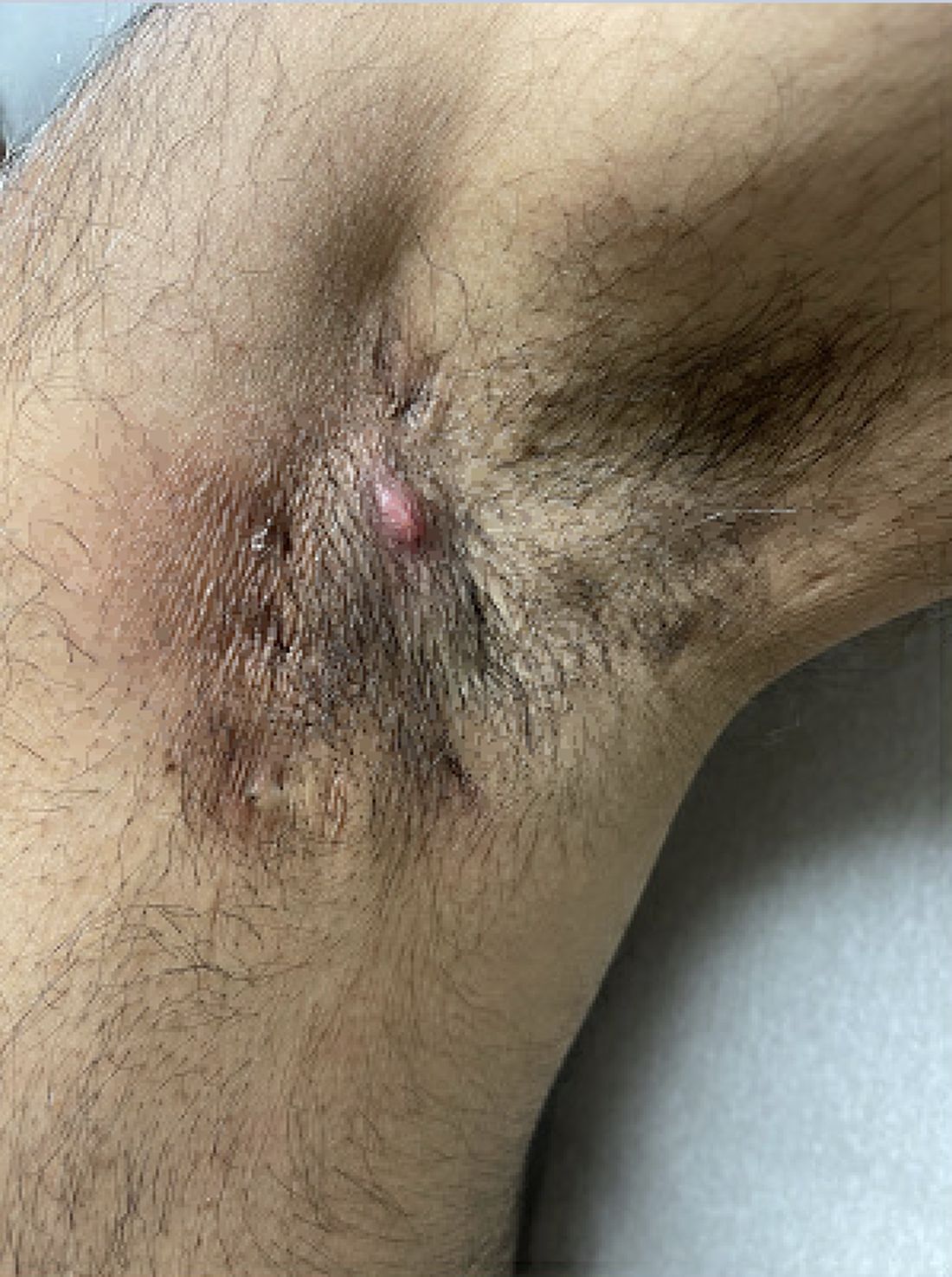
Treatments for Early HS Range From Topical Therapies to Laser Hair Removal
This can be challenging because to date, no Food and Drug Administration–approved treatments exist for early-stage HS and only two biologics exist for moderate to severe disease.
“For someone with occasional nodules and abscesses, we often use antibiotics and topical antiseptics,” Christopher Sayed, MD, a dermatologist at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill, told this news organization. “We may use these daily for weeks or months or just provide them to use for 1-2 weeks at a time for intermittent flares if a patient doesn’t want to take a pill every day,” he said. “For women, hormonal options like oral contraceptive pills and spironolactone can be a great option” if they don’t mind taking a daily pill.
Topical options that Jennifer L. Hsiao, MD, reaches for in her role as director of the HS clinic at the University of Southern California, Los Angeles, include chlorhexidine wash, topical clindamycin, and topical resorcinol. Systemic medications include oral antibiotics such as doxycycline or clindamycin, while hormonal options include oral contraceptives and/or spironolactone for women and finasteride for men.
Laser hair removal for both men and women can also help treat lesions and abscesses in the groin and axillae, since reducing hair follicles tends to result in fewer follicles that become inflamed and form nodules and abscesses over time, “but it requires multiple visits and not all patients have access to it,” Dr. Sayed said. “Once patients start to develop tunnels or scars or fail to respond to some of these other treatments, I am quick to open the conversation on biologics to help avoid progression and long-term need for surgery.”
Metformin Among Options to Consider
According to Dr. Hsiao, other treatment options to consider trying in patients with mild HS include metformin, “especially in patients who also have prediabetes, PCOS, or obesity;” isotretinoin if the patient has concomitant severe acne; botulinum toxin injections; apremilast or topical roflumilast, and antihyperhidrosis medications such as prescription aluminum chloride topicals, glycopyrronium wipes, and glycopyrrolate.
Recommending lifestyle modifications such as smoking cessation and weight loss for patients diagnosed with early-stage HS is “challenging,” Dr. Sayed said, “because the evidence on different triggers and lifestyle modifications isn’t very strong. There can also be a lot of stigmas around weight and smoking in HS, and it can alienate patients to go straight to these topics in the first visit.”
Many patients also ask what dietary changes they can make to improve their HS. “The most common things patients tend to bring up are dairy avoidance and reducing carbohydrates,” he said. “Supplements like zinc and turmeric are also frequently brought up by patients and some find them helpful. Once rapport is built, I may discuss smoking cessation as potentially helping prevent as much activity over time or weight loss as possibly helping improve response to treatments, but I don’t promise that these things always help since modifying them doesn’t always lead to improvement.”
Dr. Hsiao noted that existing research suggests that following a Mediterranean diet may benefit HS symptoms.
Early Data on Ruxolitinib Cream Promising
At the 2024 annual meeting of the American Academy of Dermatology, researchers reported on the results of a phase 2 study, which found that topical 1.5% ruxolitinib, a Janus kinase (JAK) inhibitor (currently FDA-approved for atopic dermatitis) was effective in reducing abscess and inflammatory nodule count in patients with mild HS. “There is a major need for this kind of option, and the early results are promising,” said Dr. Sayed, who was not involved with the study. “It’s very difficult to get this covered for patients currently since it is off label for HS. We’ve gotten it for a few patients, and one has really liked it, but it’s unclear how consistent the others were with their use, and their level of improvement was not clear to me.”
For mild HS, he added, “the most important area in which we’ve seen growing evidence is around hair removal lasers such as Nd:YAG and alexandrite lasers. Improving access for patients is a major priority in the coming years.”
According to Dr. Hsiao, other approaches being studied for treating mild HS include a topical aryl hydrocarbon receptor agonist known as AT193, and oral medications, such as phosphodiesterase-4 inhibitors. Laser therapies are also being studied, “such as fractional ablative CO2 laser therapy combined with topical triamcinolone,” she said. “However, the majority of ongoing HS trials are for moderate to severe disease, so there is certainly a need for more investigation into mild HS treatment approaches.”
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
This can be challenging because to date, no Food and Drug Administration–approved treatments exist for early-stage HS and only two biologics exist for moderate to severe disease.
“For someone with occasional nodules and abscesses, we often use antibiotics and topical antiseptics,” Christopher Sayed, MD, a dermatologist at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill, told this news organization. “We may use these daily for weeks or months or just provide them to use for 1-2 weeks at a time for intermittent flares if a patient doesn’t want to take a pill every day,” he said. “For women, hormonal options like oral contraceptive pills and spironolactone can be a great option” if they don’t mind taking a daily pill.
Topical options that Jennifer L. Hsiao, MD, reaches for in her role as director of the HS clinic at the University of Southern California, Los Angeles, include chlorhexidine wash, topical clindamycin, and topical resorcinol. Systemic medications include oral antibiotics such as doxycycline or clindamycin, while hormonal options include oral contraceptives and/or spironolactone for women and finasteride for men.
Laser hair removal for both men and women can also help treat lesions and abscesses in the groin and axillae, since reducing hair follicles tends to result in fewer follicles that become inflamed and form nodules and abscesses over time, “but it requires multiple visits and not all patients have access to it,” Dr. Sayed said. “Once patients start to develop tunnels or scars or fail to respond to some of these other treatments, I am quick to open the conversation on biologics to help avoid progression and long-term need for surgery.”
Metformin Among Options to Consider
According to Dr. Hsiao, other treatment options to consider trying in patients with mild HS include metformin, “especially in patients who also have prediabetes, PCOS, or obesity;” isotretinoin if the patient has concomitant severe acne; botulinum toxin injections; apremilast or topical roflumilast, and antihyperhidrosis medications such as prescription aluminum chloride topicals, glycopyrronium wipes, and glycopyrrolate.
Recommending lifestyle modifications such as smoking cessation and weight loss for patients diagnosed with early-stage HS is “challenging,” Dr. Sayed said, “because the evidence on different triggers and lifestyle modifications isn’t very strong. There can also be a lot of stigmas around weight and smoking in HS, and it can alienate patients to go straight to these topics in the first visit.”
Many patients also ask what dietary changes they can make to improve their HS. “The most common things patients tend to bring up are dairy avoidance and reducing carbohydrates,” he said. “Supplements like zinc and turmeric are also frequently brought up by patients and some find them helpful. Once rapport is built, I may discuss smoking cessation as potentially helping prevent as much activity over time or weight loss as possibly helping improve response to treatments, but I don’t promise that these things always help since modifying them doesn’t always lead to improvement.”
Dr. Hsiao noted that existing research suggests that following a Mediterranean diet may benefit HS symptoms.
Early Data on Ruxolitinib Cream Promising
At the 2024 annual meeting of the American Academy of Dermatology, researchers reported on the results of a phase 2 study, which found that topical 1.5% ruxolitinib, a Janus kinase (JAK) inhibitor (currently FDA-approved for atopic dermatitis) was effective in reducing abscess and inflammatory nodule count in patients with mild HS. “There is a major need for this kind of option, and the early results are promising,” said Dr. Sayed, who was not involved with the study. “It’s very difficult to get this covered for patients currently since it is off label for HS. We’ve gotten it for a few patients, and one has really liked it, but it’s unclear how consistent the others were with their use, and their level of improvement was not clear to me.”
For mild HS, he added, “the most important area in which we’ve seen growing evidence is around hair removal lasers such as Nd:YAG and alexandrite lasers. Improving access for patients is a major priority in the coming years.”
According to Dr. Hsiao, other approaches being studied for treating mild HS include a topical aryl hydrocarbon receptor agonist known as AT193, and oral medications, such as phosphodiesterase-4 inhibitors. Laser therapies are also being studied, “such as fractional ablative CO2 laser therapy combined with topical triamcinolone,” she said. “However, the majority of ongoing HS trials are for moderate to severe disease, so there is certainly a need for more investigation into mild HS treatment approaches.”
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
This can be challenging because to date, no Food and Drug Administration–approved treatments exist for early-stage HS and only two biologics exist for moderate to severe disease.
“For someone with occasional nodules and abscesses, we often use antibiotics and topical antiseptics,” Christopher Sayed, MD, a dermatologist at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill, told this news organization. “We may use these daily for weeks or months or just provide them to use for 1-2 weeks at a time for intermittent flares if a patient doesn’t want to take a pill every day,” he said. “For women, hormonal options like oral contraceptive pills and spironolactone can be a great option” if they don’t mind taking a daily pill.
Topical options that Jennifer L. Hsiao, MD, reaches for in her role as director of the HS clinic at the University of Southern California, Los Angeles, include chlorhexidine wash, topical clindamycin, and topical resorcinol. Systemic medications include oral antibiotics such as doxycycline or clindamycin, while hormonal options include oral contraceptives and/or spironolactone for women and finasteride for men.
Laser hair removal for both men and women can also help treat lesions and abscesses in the groin and axillae, since reducing hair follicles tends to result in fewer follicles that become inflamed and form nodules and abscesses over time, “but it requires multiple visits and not all patients have access to it,” Dr. Sayed said. “Once patients start to develop tunnels or scars or fail to respond to some of these other treatments, I am quick to open the conversation on biologics to help avoid progression and long-term need for surgery.”
Metformin Among Options to Consider
According to Dr. Hsiao, other treatment options to consider trying in patients with mild HS include metformin, “especially in patients who also have prediabetes, PCOS, or obesity;” isotretinoin if the patient has concomitant severe acne; botulinum toxin injections; apremilast or topical roflumilast, and antihyperhidrosis medications such as prescription aluminum chloride topicals, glycopyrronium wipes, and glycopyrrolate.
Recommending lifestyle modifications such as smoking cessation and weight loss for patients diagnosed with early-stage HS is “challenging,” Dr. Sayed said, “because the evidence on different triggers and lifestyle modifications isn’t very strong. There can also be a lot of stigmas around weight and smoking in HS, and it can alienate patients to go straight to these topics in the first visit.”
Many patients also ask what dietary changes they can make to improve their HS. “The most common things patients tend to bring up are dairy avoidance and reducing carbohydrates,” he said. “Supplements like zinc and turmeric are also frequently brought up by patients and some find them helpful. Once rapport is built, I may discuss smoking cessation as potentially helping prevent as much activity over time or weight loss as possibly helping improve response to treatments, but I don’t promise that these things always help since modifying them doesn’t always lead to improvement.”
Dr. Hsiao noted that existing research suggests that following a Mediterranean diet may benefit HS symptoms.
Early Data on Ruxolitinib Cream Promising
At the 2024 annual meeting of the American Academy of Dermatology, researchers reported on the results of a phase 2 study, which found that topical 1.5% ruxolitinib, a Janus kinase (JAK) inhibitor (currently FDA-approved for atopic dermatitis) was effective in reducing abscess and inflammatory nodule count in patients with mild HS. “There is a major need for this kind of option, and the early results are promising,” said Dr. Sayed, who was not involved with the study. “It’s very difficult to get this covered for patients currently since it is off label for HS. We’ve gotten it for a few patients, and one has really liked it, but it’s unclear how consistent the others were with their use, and their level of improvement was not clear to me.”
For mild HS, he added, “the most important area in which we’ve seen growing evidence is around hair removal lasers such as Nd:YAG and alexandrite lasers. Improving access for patients is a major priority in the coming years.”
According to Dr. Hsiao, other approaches being studied for treating mild HS include a topical aryl hydrocarbon receptor agonist known as AT193, and oral medications, such as phosphodiesterase-4 inhibitors. Laser therapies are also being studied, “such as fractional ablative CO2 laser therapy combined with topical triamcinolone,” she said. “However, the majority of ongoing HS trials are for moderate to severe disease, so there is certainly a need for more investigation into mild HS treatment approaches.”
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
Diagnosing Mild Hidradenitis Suppurativa: Early Stage Can Mimic Other Diseases
, such as an infection, folliculitis, and acne.
According to 2019 guidelines from the United States and Canadian hidradenitis suppurativa foundations, the diagnostic criteria for HS in general are the presence of typical lesions such as abscesses, nodules, and tunnels in classic locations such as underarms, groins, and buttocks that recur over the course of at least 6 months. “There is no need for additional testing or imaging to make the diagnosis,” said Dr. Sayed, co-chair of the 2019 guidelines work group, who sees patients at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill. “In many ways, the diagnosis should be very simple since the presentation is classic in most cases, though it can be confusing in the first 6 months or so.”
Persistence, Recurrence Major Clues
Prior to being diagnosed with Hurley stage I HS — characterized by recurrent nodules and abscesses with minimal scars, according to the guidelines — most people figure they’ve been getting recurrent Staphylococcus aureus infections or are having trouble with ingrown hairs from shaving, he continued. They may also say they get “boils” without an understanding of what has been causing them.
“Early HS can mimic an intense folliculitis or furuncles that can sometimes be caused by Staphylococcus infections, but the history of persistence or recurrence for months, despite treatment that should cover something like a Staph infection is a major clue,” Dr. Sayed said. “Thanks to improved resources on the internet, more patients, compared to several years ago, come in asking about HS after they’ve done their own research. As public awareness improves, hopefully this trend will grow, and patients will be diagnosed and treated earlier.” Family history is also a strong predictor of HS, since about half of patients have first-degree relatives who have a history of HS, he noted.
Clinicians can use the Hurley staging system to characterize the extent of disease and the Dermatology Life Quality Index to measure the impact of HS on quality of life. “We perform these assessments in our specialty clinic at each visit, but they are not necessary for diagnosis,” Dr. Sayed told this news organization.
The ‘2-2-6 Rule’
When she sees a patient who might have HS, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, follows the “2-2-6 rule,” which involves asking patients if they have had 2 episodes of 2 or more abscesses in 6 months. “If the patient answers yes, there’s a high likelihood that person has HS,” she said.
Hurley stage I HS is defined as nodules and abscesses without sinus tracts (tunnels) or scarring. But in Dr. Hsiao’s opinion, the Hurley staging system “is not the best way to characterize disease activity” because some patients meet criteria for Hurley stage I disease, meaning they do not have any scars or sinus tracts/tunnels, “but they have high disease activity with several inflammatory nodules and large painful abscesses that are limiting their quality of life and ability to function.”
Most cases of early-stage HS can be diagnosed in a single clinic visit, but some patients may present with a limited history of disease. For example, they may report having only had one episode of an axillary abscess or one episode of a few folliculitis-like papules in the groin. “In the absence of other physical exam findings suggestive of HS, such as open or double-headed comedones in flexural regions, I tell the patient that it is too early to call their condition HS, and I recommend that if they have another episode to call the office for an appointment for evaluation,” Dr. Hsiao said in an interview.
“What sets HS apart from an isolated incidence of a Staphylococcus aureus furuncle is the history of recurrence,” she added. To better characterize HS disease severity, she uses the six-point HS Physician Global Assessment score, a scale from 0 to 5, which classifies a patient as having moderate HS if they have five or more inflammatory nodules, or one abscess and one or more inflammatory nodule(s), without the requirement of demonstrating a scar or tunnel on a physical exam.
To help guide management decisions, Dr. Hsiao also considers asking patients with early-stage HS the following questions:
- Do you have a primary care provider (PCP)? PCPs are important care partners for patients with HS doctor to help screen for the comorbidities associated with the condition.
- What seems to make your HS worse? This can help identify potential triggers to avoid.
- What other medical conditions do you have?
- How would you describe the impact HS has on your quality of life?
- For women: Does your HS get worse around your period? “This can help to identify a potential hormonal trigger,” she said. “If the patient answers ‘yes,’ I would strongly consider a combined oral contraceptive pill and/or spironolactone as part of the patient’s treatment regimen.”
‘Window of Opportunity’ to Intervene
According to Dr. Hsiao, there has been a paradigm shift in the approach to HS management that emphasizes a “window of opportunity,” where earlier initiation of appropriate long-term immunomodulator therapy is recommended to try to mitigate disease progression. The development of tunnels and scars is a telltale sign that permanent tissue destruction is occurring, and the patient’s HS is no longer mild.
Ideally, a conversation about adalimumab, a tumor necrosis factor inhibitor, and secukinumab, an interleukin-17A antagonist (the two currently Food and Drug Administration–approved medications for HS, for moderate to severe disease/Hurley stage II/III) will have already been started with patients prior to development of a high tunnel or scar burden, signs of later-stage disease.
“Medications like this have the potential to slow and prevent that progression and reduce the surgical burden patients face over time, which is a major priority,” Dr. Sayed said. He noted that while comfort level with managing HS can vary among clinicians, “I’d encourage dermatologists to stay engaged with these patients because our training in the medical and surgical management of complex diseases like this is unmatched among other specialties,” he said. “Education of colleagues in other specialties should also be a big priority, especially for those in urgent care, emergency medicine, surgery, and ob.gyn. who often encounter these patients and may be less familiar” with HS.
Besides the North American clinical management guidelines for HS, which are expected to be updated in the next 18-24 months, as well as comorbidity screening recommendations for HS published in 2022, another resource Dr. Sayed and Dr. Hsiao recommend is the HS Foundation website, which features a link to Continuing Medical Education video lectures. The foundation also hosts an annual Symposium on HS Advances. This year’s event is scheduled in November in Austin, Texas.
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
, such as an infection, folliculitis, and acne.
According to 2019 guidelines from the United States and Canadian hidradenitis suppurativa foundations, the diagnostic criteria for HS in general are the presence of typical lesions such as abscesses, nodules, and tunnels in classic locations such as underarms, groins, and buttocks that recur over the course of at least 6 months. “There is no need for additional testing or imaging to make the diagnosis,” said Dr. Sayed, co-chair of the 2019 guidelines work group, who sees patients at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill. “In many ways, the diagnosis should be very simple since the presentation is classic in most cases, though it can be confusing in the first 6 months or so.”
Persistence, Recurrence Major Clues
Prior to being diagnosed with Hurley stage I HS — characterized by recurrent nodules and abscesses with minimal scars, according to the guidelines — most people figure they’ve been getting recurrent Staphylococcus aureus infections or are having trouble with ingrown hairs from shaving, he continued. They may also say they get “boils” without an understanding of what has been causing them.
“Early HS can mimic an intense folliculitis or furuncles that can sometimes be caused by Staphylococcus infections, but the history of persistence or recurrence for months, despite treatment that should cover something like a Staph infection is a major clue,” Dr. Sayed said. “Thanks to improved resources on the internet, more patients, compared to several years ago, come in asking about HS after they’ve done their own research. As public awareness improves, hopefully this trend will grow, and patients will be diagnosed and treated earlier.” Family history is also a strong predictor of HS, since about half of patients have first-degree relatives who have a history of HS, he noted.
Clinicians can use the Hurley staging system to characterize the extent of disease and the Dermatology Life Quality Index to measure the impact of HS on quality of life. “We perform these assessments in our specialty clinic at each visit, but they are not necessary for diagnosis,” Dr. Sayed told this news organization.
The ‘2-2-6 Rule’
When she sees a patient who might have HS, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, follows the “2-2-6 rule,” which involves asking patients if they have had 2 episodes of 2 or more abscesses in 6 months. “If the patient answers yes, there’s a high likelihood that person has HS,” she said.
Hurley stage I HS is defined as nodules and abscesses without sinus tracts (tunnels) or scarring. But in Dr. Hsiao’s opinion, the Hurley staging system “is not the best way to characterize disease activity” because some patients meet criteria for Hurley stage I disease, meaning they do not have any scars or sinus tracts/tunnels, “but they have high disease activity with several inflammatory nodules and large painful abscesses that are limiting their quality of life and ability to function.”
Most cases of early-stage HS can be diagnosed in a single clinic visit, but some patients may present with a limited history of disease. For example, they may report having only had one episode of an axillary abscess or one episode of a few folliculitis-like papules in the groin. “In the absence of other physical exam findings suggestive of HS, such as open or double-headed comedones in flexural regions, I tell the patient that it is too early to call their condition HS, and I recommend that if they have another episode to call the office for an appointment for evaluation,” Dr. Hsiao said in an interview.
“What sets HS apart from an isolated incidence of a Staphylococcus aureus furuncle is the history of recurrence,” she added. To better characterize HS disease severity, she uses the six-point HS Physician Global Assessment score, a scale from 0 to 5, which classifies a patient as having moderate HS if they have five or more inflammatory nodules, or one abscess and one or more inflammatory nodule(s), without the requirement of demonstrating a scar or tunnel on a physical exam.
To help guide management decisions, Dr. Hsiao also considers asking patients with early-stage HS the following questions:
- Do you have a primary care provider (PCP)? PCPs are important care partners for patients with HS doctor to help screen for the comorbidities associated with the condition.
- What seems to make your HS worse? This can help identify potential triggers to avoid.
- What other medical conditions do you have?
- How would you describe the impact HS has on your quality of life?
- For women: Does your HS get worse around your period? “This can help to identify a potential hormonal trigger,” she said. “If the patient answers ‘yes,’ I would strongly consider a combined oral contraceptive pill and/or spironolactone as part of the patient’s treatment regimen.”
‘Window of Opportunity’ to Intervene
According to Dr. Hsiao, there has been a paradigm shift in the approach to HS management that emphasizes a “window of opportunity,” where earlier initiation of appropriate long-term immunomodulator therapy is recommended to try to mitigate disease progression. The development of tunnels and scars is a telltale sign that permanent tissue destruction is occurring, and the patient’s HS is no longer mild.
Ideally, a conversation about adalimumab, a tumor necrosis factor inhibitor, and secukinumab, an interleukin-17A antagonist (the two currently Food and Drug Administration–approved medications for HS, for moderate to severe disease/Hurley stage II/III) will have already been started with patients prior to development of a high tunnel or scar burden, signs of later-stage disease.
“Medications like this have the potential to slow and prevent that progression and reduce the surgical burden patients face over time, which is a major priority,” Dr. Sayed said. He noted that while comfort level with managing HS can vary among clinicians, “I’d encourage dermatologists to stay engaged with these patients because our training in the medical and surgical management of complex diseases like this is unmatched among other specialties,” he said. “Education of colleagues in other specialties should also be a big priority, especially for those in urgent care, emergency medicine, surgery, and ob.gyn. who often encounter these patients and may be less familiar” with HS.
Besides the North American clinical management guidelines for HS, which are expected to be updated in the next 18-24 months, as well as comorbidity screening recommendations for HS published in 2022, another resource Dr. Sayed and Dr. Hsiao recommend is the HS Foundation website, which features a link to Continuing Medical Education video lectures. The foundation also hosts an annual Symposium on HS Advances. This year’s event is scheduled in November in Austin, Texas.
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
, such as an infection, folliculitis, and acne.
According to 2019 guidelines from the United States and Canadian hidradenitis suppurativa foundations, the diagnostic criteria for HS in general are the presence of typical lesions such as abscesses, nodules, and tunnels in classic locations such as underarms, groins, and buttocks that recur over the course of at least 6 months. “There is no need for additional testing or imaging to make the diagnosis,” said Dr. Sayed, co-chair of the 2019 guidelines work group, who sees patients at the HS and Follicular Disorders Clinic at the University of North Carolina, Chapel Hill. “In many ways, the diagnosis should be very simple since the presentation is classic in most cases, though it can be confusing in the first 6 months or so.”
Persistence, Recurrence Major Clues
Prior to being diagnosed with Hurley stage I HS — characterized by recurrent nodules and abscesses with minimal scars, according to the guidelines — most people figure they’ve been getting recurrent Staphylococcus aureus infections or are having trouble with ingrown hairs from shaving, he continued. They may also say they get “boils” without an understanding of what has been causing them.
“Early HS can mimic an intense folliculitis or furuncles that can sometimes be caused by Staphylococcus infections, but the history of persistence or recurrence for months, despite treatment that should cover something like a Staph infection is a major clue,” Dr. Sayed said. “Thanks to improved resources on the internet, more patients, compared to several years ago, come in asking about HS after they’ve done their own research. As public awareness improves, hopefully this trend will grow, and patients will be diagnosed and treated earlier.” Family history is also a strong predictor of HS, since about half of patients have first-degree relatives who have a history of HS, he noted.
Clinicians can use the Hurley staging system to characterize the extent of disease and the Dermatology Life Quality Index to measure the impact of HS on quality of life. “We perform these assessments in our specialty clinic at each visit, but they are not necessary for diagnosis,” Dr. Sayed told this news organization.
The ‘2-2-6 Rule’
When she sees a patient who might have HS, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, follows the “2-2-6 rule,” which involves asking patients if they have had 2 episodes of 2 or more abscesses in 6 months. “If the patient answers yes, there’s a high likelihood that person has HS,” she said.
Hurley stage I HS is defined as nodules and abscesses without sinus tracts (tunnels) or scarring. But in Dr. Hsiao’s opinion, the Hurley staging system “is not the best way to characterize disease activity” because some patients meet criteria for Hurley stage I disease, meaning they do not have any scars or sinus tracts/tunnels, “but they have high disease activity with several inflammatory nodules and large painful abscesses that are limiting their quality of life and ability to function.”
Most cases of early-stage HS can be diagnosed in a single clinic visit, but some patients may present with a limited history of disease. For example, they may report having only had one episode of an axillary abscess or one episode of a few folliculitis-like papules in the groin. “In the absence of other physical exam findings suggestive of HS, such as open or double-headed comedones in flexural regions, I tell the patient that it is too early to call their condition HS, and I recommend that if they have another episode to call the office for an appointment for evaluation,” Dr. Hsiao said in an interview.
“What sets HS apart from an isolated incidence of a Staphylococcus aureus furuncle is the history of recurrence,” she added. To better characterize HS disease severity, she uses the six-point HS Physician Global Assessment score, a scale from 0 to 5, which classifies a patient as having moderate HS if they have five or more inflammatory nodules, or one abscess and one or more inflammatory nodule(s), without the requirement of demonstrating a scar or tunnel on a physical exam.
To help guide management decisions, Dr. Hsiao also considers asking patients with early-stage HS the following questions:
- Do you have a primary care provider (PCP)? PCPs are important care partners for patients with HS doctor to help screen for the comorbidities associated with the condition.
- What seems to make your HS worse? This can help identify potential triggers to avoid.
- What other medical conditions do you have?
- How would you describe the impact HS has on your quality of life?
- For women: Does your HS get worse around your period? “This can help to identify a potential hormonal trigger,” she said. “If the patient answers ‘yes,’ I would strongly consider a combined oral contraceptive pill and/or spironolactone as part of the patient’s treatment regimen.”
‘Window of Opportunity’ to Intervene
According to Dr. Hsiao, there has been a paradigm shift in the approach to HS management that emphasizes a “window of opportunity,” where earlier initiation of appropriate long-term immunomodulator therapy is recommended to try to mitigate disease progression. The development of tunnels and scars is a telltale sign that permanent tissue destruction is occurring, and the patient’s HS is no longer mild.
Ideally, a conversation about adalimumab, a tumor necrosis factor inhibitor, and secukinumab, an interleukin-17A antagonist (the two currently Food and Drug Administration–approved medications for HS, for moderate to severe disease/Hurley stage II/III) will have already been started with patients prior to development of a high tunnel or scar burden, signs of later-stage disease.
“Medications like this have the potential to slow and prevent that progression and reduce the surgical burden patients face over time, which is a major priority,” Dr. Sayed said. He noted that while comfort level with managing HS can vary among clinicians, “I’d encourage dermatologists to stay engaged with these patients because our training in the medical and surgical management of complex diseases like this is unmatched among other specialties,” he said. “Education of colleagues in other specialties should also be a big priority, especially for those in urgent care, emergency medicine, surgery, and ob.gyn. who often encounter these patients and may be less familiar” with HS.
Besides the North American clinical management guidelines for HS, which are expected to be updated in the next 18-24 months, as well as comorbidity screening recommendations for HS published in 2022, another resource Dr. Sayed and Dr. Hsiao recommend is the HS Foundation website, which features a link to Continuing Medical Education video lectures. The foundation also hosts an annual Symposium on HS Advances. This year’s event is scheduled in November in Austin, Texas.
Dr. Sayed disclosed that he is secretary of the HS Foundation and a member of the European HS Foundation. He has served as a consultant for AbbVie, Alumis, AstraZeneca, Incyte, InflaRx, Novartis, Sanofi, Sonoma Biotherapeutics, and UCB; as a speaker for AbbVie, Novartis, and UCB; and as an investigator for Chemocentryx, Incyte, InflaRx, Novartis, and UCB. Dr. Hsiao disclosed that she is a member of the board of directors for the HS Foundation and has served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie, Novartis, Sanofi Regeneron, and UCB; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
A version of this article first appeared on Medscape.com.
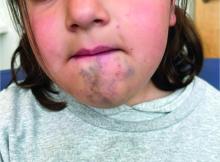
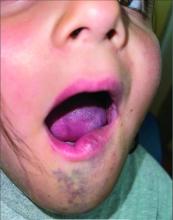
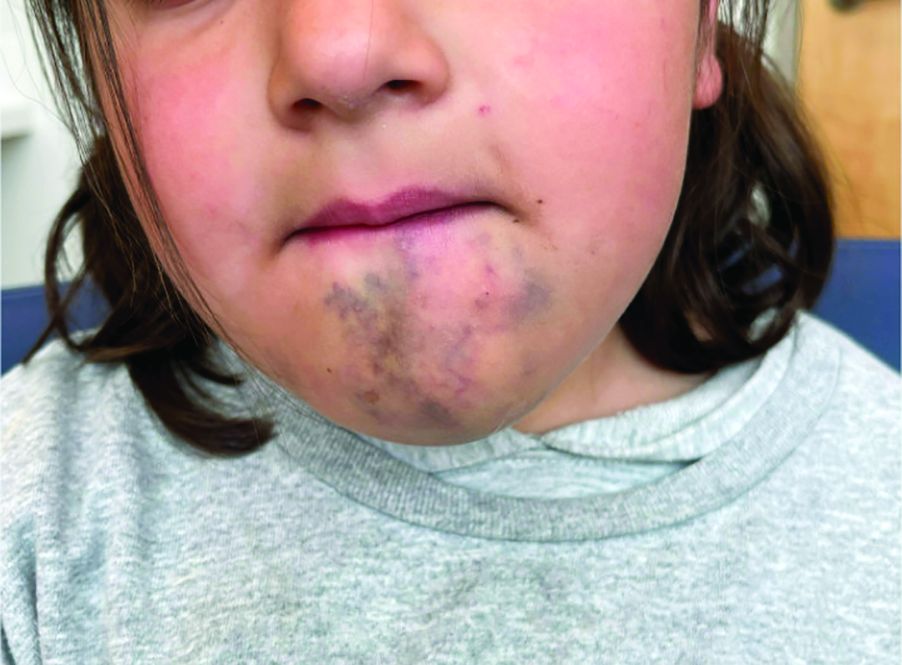
An 8-year-old girl presented with papules on her bilateral eyelid margins
, with an equal distribution across genders and ethnicities.1 It is caused by mutations in the ECM1 gene2 on chromosome 1q21. This leads to the abnormal deposition of hyaline material in various tissues across different organ systems, with the classic manifestations known as the “string of pearls” sign and a hoarse cry or voice.
The rarity of lipoid proteinosis often leads to challenges in diagnosis. Particularly when deviating from the common association with consanguinity, the potential for de novo mutations or a broader genetic variability in disease expression is highlighted. Our patient presents with symptoms that are pathognomonic to LP with moniliform blepharosis and hoarseness of the voice, in addition to scarring of the extremities.
Other common clinical manifestations in patients with LP include cobblestoning of the mucosa; hyperkeratosis of the elbows, knees, and hands; and calcification of the amygdala with neuroimaging.3
Genetic testing that identifies a loss-of-function mutation in ECM1 offers diagnostic confirmation. Patients often need multidisciplinary care involving dermatology; ear, nose, throat; neurology; and genetics. Treatment of LP is mostly symptomatic with unsatisfactory resolution of cutaneous changes, with retinoids such as acitretin used as the first-line option and surgery as a consideration for laryngeal hyaline deposits.2 Although LP can affect different organ systems, patients tend to have a normal lifespan.
LP is a rare disorder that dermatologists often learn about during textbook sessions or didactics in residency but do not see in practice for decades, or if ever. This case highlights the need to review the classic presentations of rare conditions.
This case and the photos were submitted by Ms. Chang, BS, Western University of Health Sciences, College of Osteopathic Medicine, Pomona, California; Dr. Connie Chang, Verdugo Dermatology, Glendale, California; and Dr. Yuchieh Kathryn Chang, MD Anderson Cancer Center, Houston, Texas. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Mcgrath JA. Handb Clin Neurol. 2015:132:317-22. doi: 10.1016/B978-0-444-62702-5.00023-8.
2. Hamada Tet al. Hum Mol Genet. 2002 Apr 1;11(7):833-40. doi: 10.1093/hmg/11.7.833.
3. Frenkel B et al. Clin Oral Investig. 2017 Sep;21(7):2245-51 doi: 10.1007/s00784-016-2017-7.
, with an equal distribution across genders and ethnicities.1 It is caused by mutations in the ECM1 gene2 on chromosome 1q21. This leads to the abnormal deposition of hyaline material in various tissues across different organ systems, with the classic manifestations known as the “string of pearls” sign and a hoarse cry or voice.
The rarity of lipoid proteinosis often leads to challenges in diagnosis. Particularly when deviating from the common association with consanguinity, the potential for de novo mutations or a broader genetic variability in disease expression is highlighted. Our patient presents with symptoms that are pathognomonic to LP with moniliform blepharosis and hoarseness of the voice, in addition to scarring of the extremities.
Other common clinical manifestations in patients with LP include cobblestoning of the mucosa; hyperkeratosis of the elbows, knees, and hands; and calcification of the amygdala with neuroimaging.3
Genetic testing that identifies a loss-of-function mutation in ECM1 offers diagnostic confirmation. Patients often need multidisciplinary care involving dermatology; ear, nose, throat; neurology; and genetics. Treatment of LP is mostly symptomatic with unsatisfactory resolution of cutaneous changes, with retinoids such as acitretin used as the first-line option and surgery as a consideration for laryngeal hyaline deposits.2 Although LP can affect different organ systems, patients tend to have a normal lifespan.
LP is a rare disorder that dermatologists often learn about during textbook sessions or didactics in residency but do not see in practice for decades, or if ever. This case highlights the need to review the classic presentations of rare conditions.
This case and the photos were submitted by Ms. Chang, BS, Western University of Health Sciences, College of Osteopathic Medicine, Pomona, California; Dr. Connie Chang, Verdugo Dermatology, Glendale, California; and Dr. Yuchieh Kathryn Chang, MD Anderson Cancer Center, Houston, Texas. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Mcgrath JA. Handb Clin Neurol. 2015:132:317-22. doi: 10.1016/B978-0-444-62702-5.00023-8.
2. Hamada Tet al. Hum Mol Genet. 2002 Apr 1;11(7):833-40. doi: 10.1093/hmg/11.7.833.
3. Frenkel B et al. Clin Oral Investig. 2017 Sep;21(7):2245-51 doi: 10.1007/s00784-016-2017-7.
, with an equal distribution across genders and ethnicities.1 It is caused by mutations in the ECM1 gene2 on chromosome 1q21. This leads to the abnormal deposition of hyaline material in various tissues across different organ systems, with the classic manifestations known as the “string of pearls” sign and a hoarse cry or voice.
The rarity of lipoid proteinosis often leads to challenges in diagnosis. Particularly when deviating from the common association with consanguinity, the potential for de novo mutations or a broader genetic variability in disease expression is highlighted. Our patient presents with symptoms that are pathognomonic to LP with moniliform blepharosis and hoarseness of the voice, in addition to scarring of the extremities.
Other common clinical manifestations in patients with LP include cobblestoning of the mucosa; hyperkeratosis of the elbows, knees, and hands; and calcification of the amygdala with neuroimaging.3
Genetic testing that identifies a loss-of-function mutation in ECM1 offers diagnostic confirmation. Patients often need multidisciplinary care involving dermatology; ear, nose, throat; neurology; and genetics. Treatment of LP is mostly symptomatic with unsatisfactory resolution of cutaneous changes, with retinoids such as acitretin used as the first-line option and surgery as a consideration for laryngeal hyaline deposits.2 Although LP can affect different organ systems, patients tend to have a normal lifespan.
LP is a rare disorder that dermatologists often learn about during textbook sessions or didactics in residency but do not see in practice for decades, or if ever. This case highlights the need to review the classic presentations of rare conditions.
This case and the photos were submitted by Ms. Chang, BS, Western University of Health Sciences, College of Osteopathic Medicine, Pomona, California; Dr. Connie Chang, Verdugo Dermatology, Glendale, California; and Dr. Yuchieh Kathryn Chang, MD Anderson Cancer Center, Houston, Texas. The column was edited by Donna Bilu Martin, MD.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Florida. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Mcgrath JA. Handb Clin Neurol. 2015:132:317-22. doi: 10.1016/B978-0-444-62702-5.00023-8.
2. Hamada Tet al. Hum Mol Genet. 2002 Apr 1;11(7):833-40. doi: 10.1093/hmg/11.7.833.
3. Frenkel B et al. Clin Oral Investig. 2017 Sep;21(7):2245-51 doi: 10.1007/s00784-016-2017-7.
Specialists Are ‘Underwater’ With Some Insurance-Preferred Biosimilars
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Editor’s note: This article is adapted from an explanatory statement that Dr. Feldman wrote for the Coalition of State Rheumatology Organizations (CSRO).
According to the Guinness Book of World records, the longest time someone has held their breath underwater voluntarily is 24 minutes and 37.36 seconds. While certainly an amazing feat, UnitedHealthcare, many of the Blues, and other national “payers” are expecting rheumatologists and other specialists to live “underwater” in order to take care of their patients. In other words, these insurance companies are mandating that specialists use certain provider-administered biosimilars whose acquisition cost is higher than what the insurance company is willing to reimburse them. Essentially, the insurance companies expect the rheumatologists to pay them to take care of their patients. Because of the substantial and destabilizing financial losses incurred, many practices and free-standing infusion centers have been forced to cease offering these biosimilars. Most rheumatologists will provide patients with appropriate alternatives when available and permitted by the insurer; otherwise, they must refer patients to hospital-based infusion centers. That results in delayed care and increased costs for patients and the system, because hospital-based infusion typically costs more than twice what office-based infusion costs.
Quantifying the Problem
To help quantify the magnitude of this issue, the Coalition of State Rheumatology Organizations (CSRO) recently conducted a survey of its membership. A shocking 97% of respondents reported that their practice had been affected by reimbursement rates for some biosimilars being lower than acquisition costs, with 91% of respondents stating that this issue is more pronounced for certain biosimilars than others. Across the board, respondents most frequently identified Inflectra (infliximab-dyyb) and Avsola (infliximab-axxq) as being especially affected: Over 88% and over 85% of respondents identified these two products, respectively, as being underwater. These results support the ongoing anecdotal reports CSRO continues to receive from rheumatology practices.
However, the survey results indicated that this issue is by no means confined to those two biosimilars. Truxima (rituximab-abbs) — a biosimilar for Rituxan — was frequently mentioned as well. Notably, respondents almost uniformly identified biosimilars in the infliximab and rituximab families, which illustrates that this issue is no longer confined to one or two early-to-market biosimilars but has almost become a hallmark of this particular biosimilars market. Remarkably, one respondent commented that the brand products are now cheaper to acquire than the biosimilars. Furthermore, the survey included respondents from across the country, indicating that this issue is not confined to a particular region.
How Did This Happen?
Biosimilars held promise for increasing availability and decreasing biologic costs for patients but, thus far, no patients have seen their cost go down. It appears that the only biosimilars that have made it to “preferred” status on the formulary are the ones that have made more money for the middlemen in the drug supply chain, particularly those that construct formularies. Now, we have provider-administered biosimilars whose acquisition cost exceeds the reimbursement for these drugs. This disparity was ultimately created by biosimilar manufacturers “over-rebating” their drugs to health insurance companies to gain “fail-first” status on the formulary.
For example, the manufacturer of Inflectra offered substantial rebates to health insurers for preferred formulary placement. These rebates are factored into the sales price of the medication, which then results in a rapidly declining average sales price (ASP) for the biosimilar. Unfortunately, the acquisition cost for the drug does not experience commensurate reductions, resulting in physicians being reimbursed far less for the drug than it costs to acquire. The financial losses for physicians put them underwater as a result of the acquisition costs for the preferred drugs far surpassing the reimbursement from the health insurance company that constructed the formulary.
While various factors affect ASPs and acquisition costs, this particular consequence of formulary placement based on price concessions is a major driver of the underwater situation in which physicians have found themselves with many biosimilars. Not only does that lead to a lower uptake of biosimilars, but it also results in patients being referred to the hospital outpatient infusion sites to receive this care, as freestanding infusion centers cannot treat these patients either. Hospitals incur higher costs because of facility fees and elevated rates, and this makes private rheumatology in-office infusion centers a much lower-cost option. Similarly, home infusion services, while convenient, are marginally more expensive than private practices and, in cases of biologic infusions, it is important to note that physicians’ offices have a greater safety profile than home infusion of biologics. The overall result of these “fail-first underwater drugs” is delayed and more costly care for the patient and the “system,” particularly self-insured employers.
What Is Being Done to Correct This?
Since ASPs are updated quarterly, it is possible that acquisition costs and reimbursements might stabilize over time, making the drugs affordable again to practices. However, that does not appear to be happening in the near future, so that possibility does not offer immediate relief to struggling practices. It doesn’t promise a favorable outlook for future biosimilar entries of provider-administered medications if formularies continue to prefer the highest-rebated medication.
This dynamic between ASP and acquisition cost does not happen on the pharmacy side because the price concessions on specific drug rebates and fees are proprietary. There appears to be no equivalent to a publicly known ASP on the pharmacy side, which has led to myriad pricing definitions and manipulation on the pharmacy benefit side of medications. In any event, the savings from rebates and other manufacturer price concessions on pharmacy drugs do not influence ASPs of medical benefit drugs.
The Inflation Reduction Act provided a temporary increase in the add-on payment for biosimilars from ASP+6% to ASP+8%, but as long as the biosimilar’s ASP is lower than the reference brand’s ASP, that temporary increase does not appear to make up for the large differential between ASP and acquisition cost. It should be noted that any federal attempt to artificially lower the ASP of a provider-administered drug without a pathway assuring that the acquisition cost for the provider is less than the reimbursement is going to result in loss of access for patients to those medications and/or higher hospital site of care costs.
A Few Partial Fixes, But Most Complaints Go Ignored
Considering the higher costs of hospital-based infusion, insurers should be motivated to keep patients within private practices. Perhaps through insurers’ recognition of that fact, some practices have successfully negotiated exceptions for specific patients by discussing this situation with insurers. From the feedback that CSRO has received from rheumatology practices, it appears that most insurers have been ignoring the complaints from physicians. The few who have responded have resulted in only partial fixes, with some of the biosimilars still left underwater.
Ultimate Solution?
This issue is a direct result of the “rebate game,” whereby price concessions from drug manufacturers drive formulary placement. For provider-administered medications, this results in an artificially lowered ASP, not as a consequence of free-market incentives that benefit the patient, but as a result of misaligned incentives created by Safe Harbor–protected “kickbacks,” distorting the free market and paradoxically reducing access to these medications, delaying care, and increasing prices for patients and the healthcare system.
While federal and state governments are not likely to address this particular situation in the biosimilars market, CSRO is highlighting this issue as a prime example of why the current formulary construction system urgently requires federal reform. At this time, the biosimilars most affected are Inflectra and Avsola, but if nothing changes, more and more biosimilars will fall victim to the short-sighted pricing strategy of aggressive rebating to gain formulary position, with physician purchasers and patients left to navigate the aftermath. The existing system, which necessitates drug companies purchasing formulary access from pharmacy benefit managers, has led to delayed and even denied patient access to certain provider-administered drugs. Moreover, it now appears to be hindering the adoption of biosimilars.
To address this, a multifaceted approach is required. It not only involves reevaluating the rebate system and its impact on formulary construction and ASP, but also ensuring that acquisition costs for providers are aligned with reimbursement rates. Insurers must recognize the economic and clinical value of maintaining infusions within private practices and immediately update their policies to ensure that physician in-office infusion is financially feasible for these “fail-first” biosimilars.
Ultimately, the goal should be to create a sustainable model that promotes the use of affordable biosimilars, enhances patient access to affordable care, and supports the financial viability of medical practices. Concerted efforts to reform the current formulary construction system are required to achieve a healthcare environment that is both cost effective and patient centric.
Dr. Feldman is a rheumatologist in private practice with The Rheumatology Group in New Orleans. She is the CSRO’s vice president of advocacy and government affairs and its immediate past president, as well as past chair of the Alliance for Safe Biologic Medicines and a past member of the American College of Rheumatology insurance subcommittee. You can reach her at [email protected].
Pediatric Dermatologists Beat ChatGPT on Board Questions
In an experiment that pitted the wits of results from a small single-center study showed.
“We were relieved to find that the pediatric dermatologists in our study performed better than ChatGPT on both multiple choice and case-based questions; however, the latest iteration of ChatGPT (4.0) was very close,” one of the study’s first authors Charles Huang, a fourth-year medical student at Thomas Jefferson University, Philadelphia, said in an interview. “Something else that was interesting in our data was that the pediatric dermatologists performed much better than ChatGPT on questions related to procedural dermatology/surgical techniques, perhaps indicating that knowledge/reasoning gained through practical experience isn’t easily replicated in AI tools such as ChatGPT.”
For the study, which was published on May 9 in Pediatric Dermatology, Mr. Huang, and co-first author Esther Zhang, BS, a medical student at the University of Pennsylvania, Philadelphia, and coauthors from the Department of Dermatology, Children’s Hospital of Philadelphia, asked five pediatric dermatologists to answer 24 text-based questions including 16 single-answer, multiple-choice questions and two multiple answer questions drawn from the American Board of Dermatology 2021 Certification Sample Test and six free-response case-based questions drawn from the “Photoquiz” section of Pediatric Dermatology between July 2022 and July 2023. The researchers then processed the same set of questions through ChatGPT versions 3.5 and 4.0 and used statistical analysis to compare responses between the pediatric dermatologists and ChatGPT. A 5-point scale adapted from current AI tools was used to score replies to case-based questions.
On average, study participants had 5.6 years of clinical experience. Pediatric dermatologists performed significantly better than ChatGPT version 3.5 on multiple-choice and multiple answer questions (91.4% vs 76.2%, respectively; P = .021) but not significantly better than ChatGPT version 4.0 (90.5%; P = .44). As for replies to case-based questions, the average performance based on the 5-point scale was 3.81 for pediatric dermatologists and 3.53 for ChatGPT overall. The mean scores were significantly greater for pediatric dermatologists than for ChatGPT version 3.5 (P = .039) but not ChatGPT version 4.0 (P = .43).
The researchers acknowledged certain limitations of the analysis, including the evolving nature of AI tools, which may affect the reproducibility of results with subsequent model updates. And, while participating pediatric dermatologists said they were unfamiliar with the questions and cases used in the study, “there is potential for prior exposure through other dermatology board examination review processes,” they wrote.
“AI tools such as ChatGPT and similar large language models can be a valuable tool in your clinical practice, but be aware of potential pitfalls such as patient privacy, medical inaccuracies, [and] intrinsic biases in the tools,” Mr. Huang told this news organization. “As these technologies continue to advance, it is essential for all of us as medical clinicians to gain familiarity and stay abreast of new developments, just as we adapted to electronic health records and the use of the Internet.”
Maria Buethe, MD, PhD, a pediatric dermatology fellow at Rady Children’s Hospital–San Diego, who was asked to comment on the study, said she found it “interesting” that ChatGPT’s version 4.0 started to produce comparable results to clinician responses in some of the tested scenarios.
“The authors propose a set of best practices for pediatric dermatology clinicians using ChatGPT and other AI tools,” said Dr. Buethe, who was senior author of a recent literature review on AI and its application to pediatric dermatology. It was published in SKIN The Journal of Cutaneous Medicine. “One interesting recommended use for AI tools is to utilize it to generate differential diagnosis, which can broaden the list of pathologies previously considered.”
Asked to comment on the study, Erum Ilyas, MD, who practices dermatology in King of Prussia, Pennsylvania, and is a member of the Society for Pediatric Dermatology, said she was not surprised that ChatGPT “can perform fairly well on multiple-choice questions as we find available in testing circumstances,” as presented in the study. “Just as board questions only support testing a base of medical knowledge and facts for clinicians to master, they do not necessarily provide real-life circumstances that apply to caring for patients, which is inherently nuanced.”
In addition, the study “highlights that ChatGPT can be an aid to support thinking through differentials based on data entered by a clinician who understands how to phrase queries, especially if provided with enough data while respecting patient privacy, in the context of fact checking responses,” Dr. Ilyas said. “This underscores the fact that AI tools can be helpful to clinicians in assimilating various data points entered. However, ultimately, the tool is only able to support an output based on the information it has access to.” She added, “ChatGPT cannot be relied on to provide a single diagnosis with the clinician still responsible for making a final diagnosis. The tool is not definitive and cannot assimilate data that is not entered correctly.”
The study was not funded, and the study authors reported having no disclosures. Dr. Buethe and Dr. Ilyas, who were not involved with the study, had no disclosures.
A version of this article appeared on Medscape.com .
In an experiment that pitted the wits of results from a small single-center study showed.
“We were relieved to find that the pediatric dermatologists in our study performed better than ChatGPT on both multiple choice and case-based questions; however, the latest iteration of ChatGPT (4.0) was very close,” one of the study’s first authors Charles Huang, a fourth-year medical student at Thomas Jefferson University, Philadelphia, said in an interview. “Something else that was interesting in our data was that the pediatric dermatologists performed much better than ChatGPT on questions related to procedural dermatology/surgical techniques, perhaps indicating that knowledge/reasoning gained through practical experience isn’t easily replicated in AI tools such as ChatGPT.”
For the study, which was published on May 9 in Pediatric Dermatology, Mr. Huang, and co-first author Esther Zhang, BS, a medical student at the University of Pennsylvania, Philadelphia, and coauthors from the Department of Dermatology, Children’s Hospital of Philadelphia, asked five pediatric dermatologists to answer 24 text-based questions including 16 single-answer, multiple-choice questions and two multiple answer questions drawn from the American Board of Dermatology 2021 Certification Sample Test and six free-response case-based questions drawn from the “Photoquiz” section of Pediatric Dermatology between July 2022 and July 2023. The researchers then processed the same set of questions through ChatGPT versions 3.5 and 4.0 and used statistical analysis to compare responses between the pediatric dermatologists and ChatGPT. A 5-point scale adapted from current AI tools was used to score replies to case-based questions.
On average, study participants had 5.6 years of clinical experience. Pediatric dermatologists performed significantly better than ChatGPT version 3.5 on multiple-choice and multiple answer questions (91.4% vs 76.2%, respectively; P = .021) but not significantly better than ChatGPT version 4.0 (90.5%; P = .44). As for replies to case-based questions, the average performance based on the 5-point scale was 3.81 for pediatric dermatologists and 3.53 for ChatGPT overall. The mean scores were significantly greater for pediatric dermatologists than for ChatGPT version 3.5 (P = .039) but not ChatGPT version 4.0 (P = .43).
The researchers acknowledged certain limitations of the analysis, including the evolving nature of AI tools, which may affect the reproducibility of results with subsequent model updates. And, while participating pediatric dermatologists said they were unfamiliar with the questions and cases used in the study, “there is potential for prior exposure through other dermatology board examination review processes,” they wrote.
“AI tools such as ChatGPT and similar large language models can be a valuable tool in your clinical practice, but be aware of potential pitfalls such as patient privacy, medical inaccuracies, [and] intrinsic biases in the tools,” Mr. Huang told this news organization. “As these technologies continue to advance, it is essential for all of us as medical clinicians to gain familiarity and stay abreast of new developments, just as we adapted to electronic health records and the use of the Internet.”
Maria Buethe, MD, PhD, a pediatric dermatology fellow at Rady Children’s Hospital–San Diego, who was asked to comment on the study, said she found it “interesting” that ChatGPT’s version 4.0 started to produce comparable results to clinician responses in some of the tested scenarios.
“The authors propose a set of best practices for pediatric dermatology clinicians using ChatGPT and other AI tools,” said Dr. Buethe, who was senior author of a recent literature review on AI and its application to pediatric dermatology. It was published in SKIN The Journal of Cutaneous Medicine. “One interesting recommended use for AI tools is to utilize it to generate differential diagnosis, which can broaden the list of pathologies previously considered.”
Asked to comment on the study, Erum Ilyas, MD, who practices dermatology in King of Prussia, Pennsylvania, and is a member of the Society for Pediatric Dermatology, said she was not surprised that ChatGPT “can perform fairly well on multiple-choice questions as we find available in testing circumstances,” as presented in the study. “Just as board questions only support testing a base of medical knowledge and facts for clinicians to master, they do not necessarily provide real-life circumstances that apply to caring for patients, which is inherently nuanced.”
In addition, the study “highlights that ChatGPT can be an aid to support thinking through differentials based on data entered by a clinician who understands how to phrase queries, especially if provided with enough data while respecting patient privacy, in the context of fact checking responses,” Dr. Ilyas said. “This underscores the fact that AI tools can be helpful to clinicians in assimilating various data points entered. However, ultimately, the tool is only able to support an output based on the information it has access to.” She added, “ChatGPT cannot be relied on to provide a single diagnosis with the clinician still responsible for making a final diagnosis. The tool is not definitive and cannot assimilate data that is not entered correctly.”
The study was not funded, and the study authors reported having no disclosures. Dr. Buethe and Dr. Ilyas, who were not involved with the study, had no disclosures.
A version of this article appeared on Medscape.com .
In an experiment that pitted the wits of results from a small single-center study showed.
“We were relieved to find that the pediatric dermatologists in our study performed better than ChatGPT on both multiple choice and case-based questions; however, the latest iteration of ChatGPT (4.0) was very close,” one of the study’s first authors Charles Huang, a fourth-year medical student at Thomas Jefferson University, Philadelphia, said in an interview. “Something else that was interesting in our data was that the pediatric dermatologists performed much better than ChatGPT on questions related to procedural dermatology/surgical techniques, perhaps indicating that knowledge/reasoning gained through practical experience isn’t easily replicated in AI tools such as ChatGPT.”
For the study, which was published on May 9 in Pediatric Dermatology, Mr. Huang, and co-first author Esther Zhang, BS, a medical student at the University of Pennsylvania, Philadelphia, and coauthors from the Department of Dermatology, Children’s Hospital of Philadelphia, asked five pediatric dermatologists to answer 24 text-based questions including 16 single-answer, multiple-choice questions and two multiple answer questions drawn from the American Board of Dermatology 2021 Certification Sample Test and six free-response case-based questions drawn from the “Photoquiz” section of Pediatric Dermatology between July 2022 and July 2023. The researchers then processed the same set of questions through ChatGPT versions 3.5 and 4.0 and used statistical analysis to compare responses between the pediatric dermatologists and ChatGPT. A 5-point scale adapted from current AI tools was used to score replies to case-based questions.
On average, study participants had 5.6 years of clinical experience. Pediatric dermatologists performed significantly better than ChatGPT version 3.5 on multiple-choice and multiple answer questions (91.4% vs 76.2%, respectively; P = .021) but not significantly better than ChatGPT version 4.0 (90.5%; P = .44). As for replies to case-based questions, the average performance based on the 5-point scale was 3.81 for pediatric dermatologists and 3.53 for ChatGPT overall. The mean scores were significantly greater for pediatric dermatologists than for ChatGPT version 3.5 (P = .039) but not ChatGPT version 4.0 (P = .43).
The researchers acknowledged certain limitations of the analysis, including the evolving nature of AI tools, which may affect the reproducibility of results with subsequent model updates. And, while participating pediatric dermatologists said they were unfamiliar with the questions and cases used in the study, “there is potential for prior exposure through other dermatology board examination review processes,” they wrote.
“AI tools such as ChatGPT and similar large language models can be a valuable tool in your clinical practice, but be aware of potential pitfalls such as patient privacy, medical inaccuracies, [and] intrinsic biases in the tools,” Mr. Huang told this news organization. “As these technologies continue to advance, it is essential for all of us as medical clinicians to gain familiarity and stay abreast of new developments, just as we adapted to electronic health records and the use of the Internet.”
Maria Buethe, MD, PhD, a pediatric dermatology fellow at Rady Children’s Hospital–San Diego, who was asked to comment on the study, said she found it “interesting” that ChatGPT’s version 4.0 started to produce comparable results to clinician responses in some of the tested scenarios.
“The authors propose a set of best practices for pediatric dermatology clinicians using ChatGPT and other AI tools,” said Dr. Buethe, who was senior author of a recent literature review on AI and its application to pediatric dermatology. It was published in SKIN The Journal of Cutaneous Medicine. “One interesting recommended use for AI tools is to utilize it to generate differential diagnosis, which can broaden the list of pathologies previously considered.”
Asked to comment on the study, Erum Ilyas, MD, who practices dermatology in King of Prussia, Pennsylvania, and is a member of the Society for Pediatric Dermatology, said she was not surprised that ChatGPT “can perform fairly well on multiple-choice questions as we find available in testing circumstances,” as presented in the study. “Just as board questions only support testing a base of medical knowledge and facts for clinicians to master, they do not necessarily provide real-life circumstances that apply to caring for patients, which is inherently nuanced.”
In addition, the study “highlights that ChatGPT can be an aid to support thinking through differentials based on data entered by a clinician who understands how to phrase queries, especially if provided with enough data while respecting patient privacy, in the context of fact checking responses,” Dr. Ilyas said. “This underscores the fact that AI tools can be helpful to clinicians in assimilating various data points entered. However, ultimately, the tool is only able to support an output based on the information it has access to.” She added, “ChatGPT cannot be relied on to provide a single diagnosis with the clinician still responsible for making a final diagnosis. The tool is not definitive and cannot assimilate data that is not entered correctly.”
The study was not funded, and the study authors reported having no disclosures. Dr. Buethe and Dr. Ilyas, who were not involved with the study, had no disclosures.
A version of this article appeared on Medscape.com .
Global Analysis Identifies Drugs Associated With SJS-TEN in Children
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- SJS and TEN are rare, life-threatening mucocutaneous reactions mainly associated with medications, but large pharmacovigilance studies of drugs associated with SJS-TEN in the pediatric population are still lacking.
- Using the WHO’s pharmacovigilance database (VigiBase) containing individual case safety reports from January 1967 to July 2022, researchers identified 7342 adverse drug reaction reports of SJS-TEN in children (younger than 18 years; median age, 9 years) in all six continents. Median onset was 5 days, and 3.2% were fatal.
- They analyzed drugs reported as suspected treatments, and for each molecule, they performed a case–non-case study to assess a potential pharmacovigilance signal by computing the information component (IC).
- A positive IC value suggested more frequent reporting of a specific drug-adverse reaction pair. A positive IC025, a traditional threshold for statistical signal detection, is suggestive of a potential pharmacovigilance signal.
TAKEAWAY:
- Overall, 165 drugs were associated with a diagnosis of SJS-TEN; antiepileptic and anti-infectious drugs were the most common drug classes represented.
- The five most frequently reported drugs were carbamazepine (11.7%), lamotrigine (10.6%), sulfamethoxazole-trimethoprim (9%), acetaminophen (8.4%), and phenytoin (6.6%). The five drugs with the highest IC025 were lamotrigine, carbamazepine, phenobarbital, phenytoin, and nimesulide.
- All antiepileptics, many antibiotic families, dapsone, antiretroviral drugs, some antifungal drugs, and nonsteroidal anti-inflammatory drugs were identified in reports, with penicillins the most frequently reported antibiotic family and sulfonamides having the strongest pharmacovigilance signal.
- Vaccines were not associated with significant signals.
IN PRACTICE:
The study provides an update on “the spectrum of drugs potentially associated with SJS-TEN in the pediatric population,” the authors concluded, and “underlines the importance of reporting to pharmacovigilance the suspicion of this severe side effect of drugs with the most precise and detailed clinical description possible.”
SOURCE:
The study, led by Pauline Bataille, MD, of the Department of Pediatric Dermatology, Hôpital Necker-Enfants Malades, Paris City University, France, was published online in the Journal of the European Academy of Dermatology and Venereology.
LIMITATIONS:
Limitations include the possibility that some cases could have had an infectious or idiopathic cause not related to a drug and the lack of detailed clinical data in the database.
DISCLOSURES:
This study did not receive any funding. The authors declared no conflict of interest.
A version of this article first appeared on Medscape.com.
Aquagenic Wrinkling Among Skin-Related Signs of Cystic Fibrosis
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Patients with CF, caused by a mutation in the CF Transmembrane Conductance Regulator (CFTR) gene, can develop diverse dermatologic manifestations.
- Researchers reviewed the literature and provided their own clinical experience regarding dermatologic manifestations of CF.
- They also reviewed the cutaneous side effects of CFTR modulators and antibiotics used to treat CF.
TAKEAWAY:
- Aquagenic wrinkling of the palm is common in individuals with CF, affecting up to 80% of patients (and 25% of CF gene carriers), and can be an early manifestation of CF. Treatments include topical medications (such as aluminum chloride, corticosteroids, and salicylic acid), botulinum toxin injections, and recently, CFTR-modulating treatments.
- CF nutrient deficiency dermatitis, often in a diaper distribution, usually appears in infancy and, before newborn screening was available, was sometimes the first sign of CF in some cases. It usually resolves with an adequate diet, pancreatic enzymes, and/or nutritional supplements. Zinc and essential fatty acid deficiencies can lead to acrodermatitis enteropathica–like symptoms and psoriasiform rashes, respectively.
- CF is also associated with vascular disorders, including cutaneous and, rarely, systemic vasculitis. Treatment includes topical and oral steroids and immune-modulating therapies.
- CFTR modulators, now the most common and highly effective treatment for CF, are associated with several skin reactions, which can be managed with treatments that include topical steroids and oral antihistamines. Frequent antibiotic treatment can also trigger skin reactions.
IN PRACTICE:
“Recognition and familiarity with dermatologic clinical manifestations of CF are important for multidisciplinary care” for patients with CF, the authors wrote, adding that “dermatology providers may play a significant role in the diagnosis and management of CF cutaneous comorbidities.”
SOURCE:
Aaron D. Smith, BS, from the University of Virginia (UVA) School of Medicine, Charlottesville, and coauthors were from the departments of dermatology and pulmonology/critical care medicine at UVA. The study was published online in the Journal of the American Academy of Dermatology.
LIMITATIONS:
The authors did not make a comment about the limitations of their review.
DISCLOSURES:
No funding was received for the review. The authors had no disclosures.
A version of this article first appeared on Medscape.com.
Survey Spotlights Identification of Dermatologic Adverse Events From Cancer Therapies
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
“New cancer therapies have brought a diversity of treatment-related dermatologic adverse events (dAEs) beyond those experienced with conventional chemotherapy, which has demanded an evolving assessment of toxicities,” researchers led by Nicole R. LeBoeuf, MD, MPH, of the Department of Dermatology at Brigham and Women’s Hospital and the Center for Cutaneous Oncology at the Dana-Farber Brigham Cancer Center, Boston, wrote in a poster presented at the American Academy of Dermatology annual meeting.
The authors noted that “Version 5.0 of the Common Terminology Criteria for Adverse Events (CTCAE v5.0)” serves as the current, broadly accepted criteria for classification and grading during routine medical care and clinical trials. But despite extensive utilization of CTCAE, there is little data regarding its application.”
To evaluate how CTCAE is being used in clinical practice, they sent a four-case survey of dAEs to 81 dermatologists and 182 medical oncologists at six US-based academic institutions. For three of the cases, respondents were asked to classify and grade morbilliform, psoriasiform, and papulopustular rashes based on a review of photographs and text descriptions. For the fourth case, respondents were asked to grade a dAE using only a clinic note text description. The researchers used chi-square tests in R software to compare survey responses.
Compared with medical oncologists, dermatologists were significantly more likely to provide correct responses in characterizing morbilliform and psoriasiform eruptions. “As low as 12%” of medical oncologists were correct, and “as low as 87%” of dermatologists were correct (P < .001). Similarly, dermatologists were significantly more likely to grade the psoriasiform, papulopustular, and written cases correctly compared with medical oncologists (P < .001 for all associations).
“These cases demonstrated poor concordance of classification and grading between specialties and across medical oncology,” the authors concluded in their poster, noting that 87% of medical oncologists were interested in additional educational tools on dAEs. “With correct classification as low as 12%, medical oncologists may have more difficulty delivering appropriate, toxicity-specific therapy and may consider banal eruptions dangerous.”
Poor concordance of grading among the two groups of clinicians “raises the question of whether CTCAE v5.0 is an appropriate determinant for patient continuation on therapy or in trials,” they added. “As anticancer therapy becomes more complex — with new toxicities from novel agents and combinations — we must ensure we have a grading system that is valid across investigators and does not harm patients by instituting unnecessary treatment stops.”
Future studies, they said, “can explore what interventions beyond involvement of dermatologists improve classification and grading in practice.”
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study, noted that with the continued expansion and introduction of new targeted and immunotherapies in the oncology space, “you can be sure we will continue to appreciate the importance and value of the field of supportive oncodermatology, as hair, skin, and nails are almost guaranteed collateral damage in this story.
“Ensuring early identification and consistent grading severity is not only important for the plethora of patients who are currently developing the litany of cutaneous adverse events but to evaluate potential mitigation strategies and even push along countermeasures down the FDA approval pathway,” Dr. Friedman said. In this study, the investigators demonstrated that work “is sorely needed, not just in dermatology but even more so for our colleagues across the aisle. A central tenet of supportive oncodermatology must also be education for all stakeholders, and the good news is our oncology partners will welcome it.”
Dr. LeBoeuf disclosed that she is a consultant to and has received honoraria from Bayer, Seattle Genetics, Sanofi, Silverback, Fortress Biotech, and Synox Therapeutics outside the submitted work. No other authors reported having financial disclosures. Dr. Friedman directs the supportive oncodermatology program at GW that received independent funding from La Roche-Posay.
A version of this article first appeared on Medscape.com.
FROM AAD 2024
A 6-Year-Old Female Presents With a Bruise-Like Lesion on the Lip, Tongue, and Chin Area Present Since Birth
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
Diagnosis: Venous Malformation
Although present at birth, they are not always clinically evident early in life. They also tend to grow with the child without spontaneous regression, causing potential cosmetic concerns or complications from impingement on surrounding tissue.
Venous malformations appear with a bluish color appearing beneath the skin and can vary significantly in size and severity. Venous malformations are compressible and characterized by low to stagnant blood flow, which can spontaneously thrombose. Clinically, this may cause pain, swelling, skin changes, tissue and limb overgrowth, or functional impairment depending on location and size.
Venous malformations result from disorganized angiogenesis secondary to sporadic mutations in somatic cells. The most common implicated gene is TEK, a receptor tyrosine kinase. PIK3CA has also been involved. Both genes are involved in the PI3K/AKT/mTOR pathway, which regulates cell growth, proliferation, and angiogenesis. In venous endothelial cells, abnormal angiogenesis and vessel maturation may lead to venous malformation formation. Dysplastic vessels frequently separate from normal veins but may be contiguous with the deep venous system.
Diagnosis involves clinical history and physical examination. Imaging with ultrasound and magnetic resonance imaging (MRI) may be utilized. While ultrasound may be preferred for superficial venous malformations, MRI or MRI with MR angiography (MRA) is the preferred method for venous malformation assessment. Genetic testing may be appropriate for complex malformations, as classification of lesions by underlying mutation may allow targeted therapy.
This patient’s past MRI and MRA findings were consistent with a venous malformation.
Treatment
Venous malformations rarely regress spontaneously. Treatment is required if venous malformations are symptomatic, which may include pain, swelling, deformity, thrombosis, or interference with daily activities of living. Treatment plans require consideration of patient goals of care. The main categories of therapy are embolization/sclerotherapy, surgical resection, and molecular targeted therapy.
Sclerotherapy is a well-tolerated and efficacious first-line therapy. It can be used as either nonsurgical curative therapy or preoperative adjunct therapy to minimize blood loss before surgical resection. While surgical resection may cause scarring, multimodal approaches with sclerotherapy or laser therapy can decrease complications. Molecular therapies aim to reduce vascular proliferation and symptoms. Referral to hematology/oncology for evaluation and consideration of chemotherapeutic agents may be required. Sirolimus has been shown in mice models to inhibit an endothelial cell tyrosine kinase receptor that plays a role in venous malformation growth. Multiple studies have proved its efficacy in managing complicated vascular anomalies, including venous malformations. Alpelisib is an inhibitor of PI3KCA, which is part of the pathway that contributes to venous malformation formation. Dactolisib, a dual inhibitor of the PI3KA and mTOR pathways, and rebastinib, a TEK inhibitor, are being investigated.
Differential Diagnoses
The differential diagnosis includes dermal melanocytosis, nevus of Ota, hemangioma of infancy, and ashy dermatosis. In addition, venous malformations can be part of more complex vascular malformations.
Dermal melanocytosis, also known as Mongolian spots, are blue-gray patches of discoloration on the skin that appear at birth or shortly after. They result from the arrest of dermal melanocytes in the dermis during fetal life and tissue modeling. They are commonly observed in those of Asian or African descent with darker skin types. Most often, they are located in the lumbar or sacral-gluteal region. Unlike venous malformations, they are benign and do not involve vascular abnormalities. They typically fade over time.
Nevus of Ota is a benign congenital condition that presents with blue-gray or brown patches of pigmentation on the skin around the eyes, cheeks, and forehead. They are dermal melanocytes with a speckled instead of uniform appearance. Nevus of Ota primarily affects individuals of Asian descent and typically presents in the trigeminal nerve distribution region. Treatment can be done to minimize deformity, generally with pigmented laser surgery.
Hemangiomas of infancy are common benign tumors of infancy caused by endothelial cell proliferation. They are characterized by rapid growth followed by spontaneous involution within the first year of life and for several years. Hemangiomas can be superficial, deep, or mixed with features of both superficial and deep. Superficial hemangiomas present as raised, lobulated, and bright red while deep hemangiomas present as a bluish-hued nodule, plaque, or tumor. They are diagnosed clinically but skin biopsies and imaging can confirm the suspected diagnosis. While hemangiomas may self-resolve, complicated hemangiomas can be treated with topical timolol, oral propranolol, topical and intralesional corticosteroids, pulsed-dye laser, and surgical resection.
Ashy dermatosis is a term for asymptomatic, gray-blue or ashy patches distributed symmetrically on the trunk, head, neck, and upper extremities. It primarily affects individuals with darker skin types (Fitzpatrick III-V), and is more common in patients with Hispanic, Asian, or African backgrounds. The direct cause of ashy dermatosis is unknown but it is thought to be linked to drug ingestion, genetics, infection, and immune-mediated mechanisms. The general treatment includes topical corticosteroids, clofazimine, topical calcineurin inhibitors, oral dapsone, phototherapy, topical retinoids, or isotretinoin to reduce inflammation and pigmentation.
Danny Lee and Samuel Le serve as research fellows and Jolina Bui as research associate in the Pediatric Dermatology Division of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. Dr. Eichenfield is Distinguished Professor of Dermatology and Pediatrics and Vice-Chair of the Department of Dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. The authors have no relevant financial disclosures.
Suggested Reading
Agarwal P, Patel BC. Nevus of Ota and Ito. [Updated 2023 Jul 10]. In: StatPearls [Internet]. StatPearls Publishing; 2024.
Dompmartin A et al. The VASCERN-VASCA Working Group Diagnostic and Management Pathways for Venous Malformations. J Vasc Anom (Phila). 2023 Mar 23;4(2):e064.
Dompmartin A et al. Venous malformation: Update on aetiopathogenesis, diagnosis and management. Phlebology. 2010 Oct;25(5):224-235.
Gupta D, Thappa DM. Mongolian spots. Indian J Dermatol Venereol Leprol. 2013 Jul-Aug;79(4):469-478.
Krowchuk DP et al. Clinical Practice Guideline for the Management of Infantile Hemangiomas. Pediatrics. 2019 Jan;143(1):e20183475.
Nguyen K, Khachemoune A. Ashy dermatosis: A review. Dermatol Online J. 2019 May 15;25(5):13030/qt44f462s8.
Patel ND, Chong AT et al. Venous Malformations. Semin Intervent Radiol. 2022 Dec 20;39(5):498-507.
A 6-year-old girl presents with a bruise-like lesion on the lip, tongue, and chin area present since birth. The family states that her tongue has been increasing in size and is painful. On physical exam, she presents with left lower mucosal lip fullness and an overlying violaceous hue extending into the oral mucosa and onto the left tongue. The left portion of the dorsal tongue displays an increased thickness and bluish discoloration and there is a pink, smooth papule on the left anterolateral tongue.
Study Evaluates CVD, Mortality Risks In Patients With Prurigo Nodularis
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.
TOPLINE:
, particularly among women and White patients.
METHODOLOGY:
- Studies have shown increased risks for cardiovascular diseases in patients with PN, but limited sample sizes have hindered further subgroup analysis. Given PN’s pronounced sex and ethnicity skew, it is important to examine underrepresented groups to accurately assess their cardiovascular risk.
- In this propensity-score matched analysis, researchers identified 64,801 patients (59.44% women) with PN using electronic health reports from the Global Collaborative Network of TriNetX and matched to individuals without PN.
- Researchers calculated risks for 15 cardiovascular endpoints and all-cause mortality within 10 years of diagnosis. Major adverse cardiovascular events (MACE) included acute cerebral and myocardial infarction (MI), heart failure, ventricular arrhythmia, and sudden cardiac death.
TAKEAWAY:
- Patients with PN showed a higher risk for death (hazard ratio [HR], 1.1243) and MACE (HR, 1.117) (P < .0001 for both).
- PN was also associated with a higher risk for heart failure (HR, 1.062), thrombotic venous disease (HR, 1.26), angina pectoris (HR, 1.096), and peripheral arterial diseases (HR, 1.082) (P < .0001 for all) and for acute MI (HR, 1.11; P = .0015) and valve disorders (HR, 1.08; P = .0018).
- White patients with PN had a significantly increased risk for MACE, death, heart failure, cardiac arrest, vascular diseases, and acute MI, but this was not observed in people of color.
- Women exhibited a higher risk for MACE, heart failure, peripheral artery disease, acute MI, conduction disease, and valve disorders, while men did not have an increased risk for major or acute cardiovascular events. Both men and women had a higher risk for death, chronic ischemic heart disease, and venous disease.
IN PRACTICE:
“Although no novel PN-specific treatment rationale can be derived from the presented data, the potential risk of subsequent cardiovascular disease should be considered in the care of patients with PN, which includes screening and optimal management of other additional cardiovascular risk factors,” the authors wrote.
LIMITATIONS:
Retrospective observational design introduced inherent biases. Misdiagnosis or false coding in electronic health records could affect the data accuracy and ethnicity-specific analyses.
SOURCE:
This work, led by Henning Olbrich, from the Department of Dermatology, University of Lübeck, Germany, was published online in eBioMedicine.
DISCLOSURES:
The study was supported by the University of Lübeck, the Deutsche Forschungsgemeinschaft, and the State of Schleswig-Holstein. One author declared financial ties outside this work, and one author is an employee of TriNetX.
A version of this article appeared on Medscape.com.