User login
COVID-19 in children, pregnant women: What do we know?
A novel coronavirus, the causative agent of the current pandemic of viral respiratory illness and pneumonia, was first identified in Wuhan, Hubei, China. The disease has been given the name, coronavirus disease 2019 (COVID-19). The virus at last report has spread to more than 100 countries. Much of what we suspect about this virus comes from work on other severe coronavirus respiratory disease outbreaks – Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). MERS-CoV was a viral respiratory disease, first reported in Saudi Arabia, that was identified in more than 27 additional countries. The disease was characterized by severe acute respiratory illness, including fever, cough, and shortness of breath. Among 2,499 cases, only two patients tested positive for MERS-CoV in the United States. SARS-CoV also caused a severe viral respiratory illness. SARS was first recognized in Asia in 2003 and was subsequently reported in approximately 25 countries. The last case reported was in 2004.
As of March 13, there are 137,066 cases worldwide of COVID-19 and 1,701 in the United States, according to the John Hopkins University Coronavirus COVID-19 resource center.
What about children?
The remarkable observation is how few seriously ill children have been identified in the face of global spread. Unlike the H1N1 influenza epidemic of 2009, where older adults were relatively spared and children were a major target population, COVID-19 appears to be relatively infrequent in children or too mild to come to diagnosis, to date. Specifically, among China’s first approximately 44,000 cases, less than 2% were identified in children less than 20 years of age, and severe disease was uncommon with no deaths in children less than 10 years of age reported. One child, 13 months of age, with acute respiratory distress syndrome and septic shock was reported in China. According to the Centers for Disease Control and Prevention webcast , children present with fever in about 50% of cases, cough, fatigue, and subsequently some (3%-30%) progress to shortness of breath. Some children and adults have presented with gastrointestinal disease initially. Viral RNA has been detected in respiratory secretions, blood, and stool of affected children; however, the samples were not cultured for virus so whether stool is a potential source for transmission is unclear. In adults, the disease appears to be most severe – with development of pneumonia – in the second week of illness. In both children and adults, the chest x-ray findings are an interstitial pneumonitis, ground glass appearance, and/or patchy infiltrates.

Are some children at greater risk? Are children the source of community transmission? Will children become a greater part of the disease pattern as further cases are identified and further testing is available? We cannot answer many of these questions about COVID-19 in children as yet, but as you are aware, data are accumulating daily, and the Centers for Disease Control and Prevention and the National Institutes of Health are providing regular updates.
A report from China gave us some idea about community transmission and infection risk for children. The Shenzhen CDC identified 391 COVID-19 cases and 1,286 close contacts. Household contacts and those persons traveling with a case of the virus were at highest risk of acquisition. The secondary attack rates within households was 15%; children were as likely to become infected as adults (medRxiv preprint. 2020. doi: 10.1101/2020.03.03.20028423).
What about pregnant women?
The data on pregnant women are even more limited. The concern about COVID-19 during pregnancy comes from our knowledge of adverse outcomes from other respiratory viral infections. For example, respiratory viral infections such as influenza have been associated with increased maternal risk of severe disease, and adverse neonatal outcomes, including low birth weight and preterm birth. The experience with SARS also is concerning for excess adverse maternal and neonatal complications such as spontaneous miscarriage, preterm delivery, intrauterine growth restriction, admission to the ICU, renal failure, and disseminated intravascular coagulopathy all were reported as complications of SARS infection during pregnancy.
Two studies on COVID-19 in pregnancy have been reported to date. In nine pregnant women reported by Chen et al., COVID-19 pneumonia was identified in the third trimester. The women presented with fever, cough, myalgia, sore throat, and/or malaise. Fetal distress was reported in two; all nine infants were born alive. Apgar scores were 8-10 at 1 minute. Five were found to have lymphopenia; three had increases in hepatic enzymes. None of the infants developed severe COVID-19 pneumonia. Amniotic fluid, cord blood, neonatal throat swab, and breast milk samples from six of the nine patients were tested for the novel coronavirus 2019, and all results were negative (Lancet. 2020 Feb 12. doi: 10.1016/S0140-6736[20]30360-3)https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30360-3/fulltext.
In a study by Zhu et al., nine pregnant women with confirmed COVID-19 infection were identified during Jan. 20-Feb. 5, 2020. The onset of clinical symptoms in these women occurred before delivery in four cases, on the day of delivery in two cases, and after delivery in three cases. Of the 10 neonates (one set of twins) many had clinical symptoms, but none were proven to be COVID-19 positive in their pharyngeal swabs. Shortness of breath was observed in six, fever in two, tachycardia in one. GI symptoms such as feeding intolerance, bloating, GI bleed, and vomiting also were observed. Chest radiography showed abnormalities in seven neonates at admission. Thrombocytopenia and/or disseminated intravascular coagulopathy also was reported. Five neonates recovered and were discharged, one died, and four neonates remained in hospital in a stable condition. It is unclear if the illness in these infants was related to COVID-19 (Transl Pediatrics. 2020 Feb. doi: 10.21037/tp.2020.02.06)http://tp.amegroups.com/article/view/35919/28274.
In the limited experience to date, no evidence of virus has been found in the breast milk of women with COVID-19, which is consistent with the SARS experience. Current recommendations are to separate the infant from known COVID-19 infected mothers either in a different room or in the mother’s room using a six foot rule, a barrier curtain of some type, and mask and hand washing prior to any contact between mother and infant. If the mother desires to breastfeed her child, the same precautions – mask and hand washing – should be in place.
What about treatment?
There are no proven effective therapies and supportive care has been the mainstay to date. Clinical trials of remdesivir have been initiated both by Gilead (compassionate use, open label) and by the National Institutes of Health (randomized remdesivirhttps://www.drugs.com/history/remdesivir.html vs. placebo) in adults based on in vitro data suggesting activity again COVID-19. Lopinavir/ritonavir (combination protease inhibitors) also have been administered off label, but no results are available as yet.
Keeping up
I suggest several valuable resources to keep yourself abreast of the rapidly changing COVID-19 story. First the CDC website or your local Department of Health. These are being updated frequently and include advisories on personal protective equipment, clusters of cases in your local community, and current recommendations for mitigation of the epidemic. I have listened to Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, and Robert R. Redfield, MD, the director of the CDC almost daily. I trust their viewpoints and transparency about what is and what is not known, as well as the why and wherefore of their guidance, remembering that each day brings new information and new guidance.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. He has no relevant financial disclosures. Email him at [email protected].
A novel coronavirus, the causative agent of the current pandemic of viral respiratory illness and pneumonia, was first identified in Wuhan, Hubei, China. The disease has been given the name, coronavirus disease 2019 (COVID-19). The virus at last report has spread to more than 100 countries. Much of what we suspect about this virus comes from work on other severe coronavirus respiratory disease outbreaks – Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). MERS-CoV was a viral respiratory disease, first reported in Saudi Arabia, that was identified in more than 27 additional countries. The disease was characterized by severe acute respiratory illness, including fever, cough, and shortness of breath. Among 2,499 cases, only two patients tested positive for MERS-CoV in the United States. SARS-CoV also caused a severe viral respiratory illness. SARS was first recognized in Asia in 2003 and was subsequently reported in approximately 25 countries. The last case reported was in 2004.
As of March 13, there are 137,066 cases worldwide of COVID-19 and 1,701 in the United States, according to the John Hopkins University Coronavirus COVID-19 resource center.
What about children?
The remarkable observation is how few seriously ill children have been identified in the face of global spread. Unlike the H1N1 influenza epidemic of 2009, where older adults were relatively spared and children were a major target population, COVID-19 appears to be relatively infrequent in children or too mild to come to diagnosis, to date. Specifically, among China’s first approximately 44,000 cases, less than 2% were identified in children less than 20 years of age, and severe disease was uncommon with no deaths in children less than 10 years of age reported. One child, 13 months of age, with acute respiratory distress syndrome and septic shock was reported in China. According to the Centers for Disease Control and Prevention webcast , children present with fever in about 50% of cases, cough, fatigue, and subsequently some (3%-30%) progress to shortness of breath. Some children and adults have presented with gastrointestinal disease initially. Viral RNA has been detected in respiratory secretions, blood, and stool of affected children; however, the samples were not cultured for virus so whether stool is a potential source for transmission is unclear. In adults, the disease appears to be most severe – with development of pneumonia – in the second week of illness. In both children and adults, the chest x-ray findings are an interstitial pneumonitis, ground glass appearance, and/or patchy infiltrates.

Are some children at greater risk? Are children the source of community transmission? Will children become a greater part of the disease pattern as further cases are identified and further testing is available? We cannot answer many of these questions about COVID-19 in children as yet, but as you are aware, data are accumulating daily, and the Centers for Disease Control and Prevention and the National Institutes of Health are providing regular updates.
A report from China gave us some idea about community transmission and infection risk for children. The Shenzhen CDC identified 391 COVID-19 cases and 1,286 close contacts. Household contacts and those persons traveling with a case of the virus were at highest risk of acquisition. The secondary attack rates within households was 15%; children were as likely to become infected as adults (medRxiv preprint. 2020. doi: 10.1101/2020.03.03.20028423).
What about pregnant women?
The data on pregnant women are even more limited. The concern about COVID-19 during pregnancy comes from our knowledge of adverse outcomes from other respiratory viral infections. For example, respiratory viral infections such as influenza have been associated with increased maternal risk of severe disease, and adverse neonatal outcomes, including low birth weight and preterm birth. The experience with SARS also is concerning for excess adverse maternal and neonatal complications such as spontaneous miscarriage, preterm delivery, intrauterine growth restriction, admission to the ICU, renal failure, and disseminated intravascular coagulopathy all were reported as complications of SARS infection during pregnancy.
Two studies on COVID-19 in pregnancy have been reported to date. In nine pregnant women reported by Chen et al., COVID-19 pneumonia was identified in the third trimester. The women presented with fever, cough, myalgia, sore throat, and/or malaise. Fetal distress was reported in two; all nine infants were born alive. Apgar scores were 8-10 at 1 minute. Five were found to have lymphopenia; three had increases in hepatic enzymes. None of the infants developed severe COVID-19 pneumonia. Amniotic fluid, cord blood, neonatal throat swab, and breast milk samples from six of the nine patients were tested for the novel coronavirus 2019, and all results were negative (Lancet. 2020 Feb 12. doi: 10.1016/S0140-6736[20]30360-3)https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30360-3/fulltext.
In a study by Zhu et al., nine pregnant women with confirmed COVID-19 infection were identified during Jan. 20-Feb. 5, 2020. The onset of clinical symptoms in these women occurred before delivery in four cases, on the day of delivery in two cases, and after delivery in three cases. Of the 10 neonates (one set of twins) many had clinical symptoms, but none were proven to be COVID-19 positive in their pharyngeal swabs. Shortness of breath was observed in six, fever in two, tachycardia in one. GI symptoms such as feeding intolerance, bloating, GI bleed, and vomiting also were observed. Chest radiography showed abnormalities in seven neonates at admission. Thrombocytopenia and/or disseminated intravascular coagulopathy also was reported. Five neonates recovered and were discharged, one died, and four neonates remained in hospital in a stable condition. It is unclear if the illness in these infants was related to COVID-19 (Transl Pediatrics. 2020 Feb. doi: 10.21037/tp.2020.02.06)http://tp.amegroups.com/article/view/35919/28274.
In the limited experience to date, no evidence of virus has been found in the breast milk of women with COVID-19, which is consistent with the SARS experience. Current recommendations are to separate the infant from known COVID-19 infected mothers either in a different room or in the mother’s room using a six foot rule, a barrier curtain of some type, and mask and hand washing prior to any contact between mother and infant. If the mother desires to breastfeed her child, the same precautions – mask and hand washing – should be in place.
What about treatment?
There are no proven effective therapies and supportive care has been the mainstay to date. Clinical trials of remdesivir have been initiated both by Gilead (compassionate use, open label) and by the National Institutes of Health (randomized remdesivirhttps://www.drugs.com/history/remdesivir.html vs. placebo) in adults based on in vitro data suggesting activity again COVID-19. Lopinavir/ritonavir (combination protease inhibitors) also have been administered off label, but no results are available as yet.
Keeping up
I suggest several valuable resources to keep yourself abreast of the rapidly changing COVID-19 story. First the CDC website or your local Department of Health. These are being updated frequently and include advisories on personal protective equipment, clusters of cases in your local community, and current recommendations for mitigation of the epidemic. I have listened to Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, and Robert R. Redfield, MD, the director of the CDC almost daily. I trust their viewpoints and transparency about what is and what is not known, as well as the why and wherefore of their guidance, remembering that each day brings new information and new guidance.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. He has no relevant financial disclosures. Email him at [email protected].
A novel coronavirus, the causative agent of the current pandemic of viral respiratory illness and pneumonia, was first identified in Wuhan, Hubei, China. The disease has been given the name, coronavirus disease 2019 (COVID-19). The virus at last report has spread to more than 100 countries. Much of what we suspect about this virus comes from work on other severe coronavirus respiratory disease outbreaks – Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). MERS-CoV was a viral respiratory disease, first reported in Saudi Arabia, that was identified in more than 27 additional countries. The disease was characterized by severe acute respiratory illness, including fever, cough, and shortness of breath. Among 2,499 cases, only two patients tested positive for MERS-CoV in the United States. SARS-CoV also caused a severe viral respiratory illness. SARS was first recognized in Asia in 2003 and was subsequently reported in approximately 25 countries. The last case reported was in 2004.
As of March 13, there are 137,066 cases worldwide of COVID-19 and 1,701 in the United States, according to the John Hopkins University Coronavirus COVID-19 resource center.
What about children?
The remarkable observation is how few seriously ill children have been identified in the face of global spread. Unlike the H1N1 influenza epidemic of 2009, where older adults were relatively spared and children were a major target population, COVID-19 appears to be relatively infrequent in children or too mild to come to diagnosis, to date. Specifically, among China’s first approximately 44,000 cases, less than 2% were identified in children less than 20 years of age, and severe disease was uncommon with no deaths in children less than 10 years of age reported. One child, 13 months of age, with acute respiratory distress syndrome and septic shock was reported in China. According to the Centers for Disease Control and Prevention webcast , children present with fever in about 50% of cases, cough, fatigue, and subsequently some (3%-30%) progress to shortness of breath. Some children and adults have presented with gastrointestinal disease initially. Viral RNA has been detected in respiratory secretions, blood, and stool of affected children; however, the samples were not cultured for virus so whether stool is a potential source for transmission is unclear. In adults, the disease appears to be most severe – with development of pneumonia – in the second week of illness. In both children and adults, the chest x-ray findings are an interstitial pneumonitis, ground glass appearance, and/or patchy infiltrates.

Are some children at greater risk? Are children the source of community transmission? Will children become a greater part of the disease pattern as further cases are identified and further testing is available? We cannot answer many of these questions about COVID-19 in children as yet, but as you are aware, data are accumulating daily, and the Centers for Disease Control and Prevention and the National Institutes of Health are providing regular updates.
A report from China gave us some idea about community transmission and infection risk for children. The Shenzhen CDC identified 391 COVID-19 cases and 1,286 close contacts. Household contacts and those persons traveling with a case of the virus were at highest risk of acquisition. The secondary attack rates within households was 15%; children were as likely to become infected as adults (medRxiv preprint. 2020. doi: 10.1101/2020.03.03.20028423).
What about pregnant women?
The data on pregnant women are even more limited. The concern about COVID-19 during pregnancy comes from our knowledge of adverse outcomes from other respiratory viral infections. For example, respiratory viral infections such as influenza have been associated with increased maternal risk of severe disease, and adverse neonatal outcomes, including low birth weight and preterm birth. The experience with SARS also is concerning for excess adverse maternal and neonatal complications such as spontaneous miscarriage, preterm delivery, intrauterine growth restriction, admission to the ICU, renal failure, and disseminated intravascular coagulopathy all were reported as complications of SARS infection during pregnancy.
Two studies on COVID-19 in pregnancy have been reported to date. In nine pregnant women reported by Chen et al., COVID-19 pneumonia was identified in the third trimester. The women presented with fever, cough, myalgia, sore throat, and/or malaise. Fetal distress was reported in two; all nine infants were born alive. Apgar scores were 8-10 at 1 minute. Five were found to have lymphopenia; three had increases in hepatic enzymes. None of the infants developed severe COVID-19 pneumonia. Amniotic fluid, cord blood, neonatal throat swab, and breast milk samples from six of the nine patients were tested for the novel coronavirus 2019, and all results were negative (Lancet. 2020 Feb 12. doi: 10.1016/S0140-6736[20]30360-3)https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)30360-3/fulltext.
In a study by Zhu et al., nine pregnant women with confirmed COVID-19 infection were identified during Jan. 20-Feb. 5, 2020. The onset of clinical symptoms in these women occurred before delivery in four cases, on the day of delivery in two cases, and after delivery in three cases. Of the 10 neonates (one set of twins) many had clinical symptoms, but none were proven to be COVID-19 positive in their pharyngeal swabs. Shortness of breath was observed in six, fever in two, tachycardia in one. GI symptoms such as feeding intolerance, bloating, GI bleed, and vomiting also were observed. Chest radiography showed abnormalities in seven neonates at admission. Thrombocytopenia and/or disseminated intravascular coagulopathy also was reported. Five neonates recovered and were discharged, one died, and four neonates remained in hospital in a stable condition. It is unclear if the illness in these infants was related to COVID-19 (Transl Pediatrics. 2020 Feb. doi: 10.21037/tp.2020.02.06)http://tp.amegroups.com/article/view/35919/28274.
In the limited experience to date, no evidence of virus has been found in the breast milk of women with COVID-19, which is consistent with the SARS experience. Current recommendations are to separate the infant from known COVID-19 infected mothers either in a different room or in the mother’s room using a six foot rule, a barrier curtain of some type, and mask and hand washing prior to any contact between mother and infant. If the mother desires to breastfeed her child, the same precautions – mask and hand washing – should be in place.
What about treatment?
There are no proven effective therapies and supportive care has been the mainstay to date. Clinical trials of remdesivir have been initiated both by Gilead (compassionate use, open label) and by the National Institutes of Health (randomized remdesivirhttps://www.drugs.com/history/remdesivir.html vs. placebo) in adults based on in vitro data suggesting activity again COVID-19. Lopinavir/ritonavir (combination protease inhibitors) also have been administered off label, but no results are available as yet.
Keeping up
I suggest several valuable resources to keep yourself abreast of the rapidly changing COVID-19 story. First the CDC website or your local Department of Health. These are being updated frequently and include advisories on personal protective equipment, clusters of cases in your local community, and current recommendations for mitigation of the epidemic. I have listened to Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, and Robert R. Redfield, MD, the director of the CDC almost daily. I trust their viewpoints and transparency about what is and what is not known, as well as the why and wherefore of their guidance, remembering that each day brings new information and new guidance.
Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. He has no relevant financial disclosures. Email him at [email protected].
Detection of COVID-19 in children in early January 2020 in Wuhan, China
Clinical question: What were the clinical characteristics of children in Wuhan, China hospitalized with SARS-CoV-2?
Background: The coronavirus disease 2019 (COVID-19) was recently described by researchers in Wuhan, China.1 However, there has been limited discussion on how the disease has affected children. Based on the Chinese Center for Disease Control and Prevention report, Wu et al. found that 1% of the affected population was less than 10 years, and another 1% of the affected population was 10-19 years.2 However, little information regarding hospitalizations of children with viral infections was previously reported.
Study design: A retrospective analysis of hospitalized children.
Setting: Three sites of a multisite urban teaching hospital in central Wuhan, China.
Synopsis: Over an 8-day period, hospitalized pediatric patients were retrospectively enrolled into this study. The authors defined pediatric patients as those aged 16 years or younger. The patients had one throat swab specimen collected on admission. Throat swab specimens were tested for viral etiologies. In response to the COVID-19 outbreak, the throat samples were retrospectively tested for SARS-CoV-2. If two independent experiments and a clinically verified diagnostic test confirmed the SARS-CoV-2, the cases were confirmed as COVID-19 cases. During the 8-day period, 366 hospitalized pediatric patients were included in the study. Of the 366 patients, 6 tested positive for SARS-CoV-2, while 23 tested positive for influenza A and 20 tested positive for influenza B. The median age of the six patients was 3 years (range, 1-7 years), and all were previously healthy. All six pediatric patients with COVID-19 had high fevers (greater than 39°C), cough, and lymphopenia. Four of the six affected patients had vomiting and leukopenia, while three of the six patients had neutropenia. Four of the six affected patients had pneumonia, as diagnosed on CT scans. Of the six patients, one patient was admitted to the ICU and received intravenous immunoglobulin. The patient admitted to ICU underwent a CT scan which showed “patchy ground-glass opacities in both lungs,” while three of the five children requiring non-ICU hospitalization had chest radiographs showing “patchy shadows in both lungs.” The median length of stay in the hospital was 7.5 days (range, 5-13 days).
Bottom line: COVID-19 causes moderate to severe respiratory illness in pediatric patients with SARS-CoV-2, possibly leading to critical illness. During this time period of the Wuhan COVID-19 outbreak, pediatric patients were more likely to be hospitalized with influenza A or B, than they were with SARS-CoV-2.
Citation: Liu W et al. Detection of Covid-19 in Children in Early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
Dr. Kumar is clinical assistant professor of pediatrics at Case Western Reserve University, Cleveland, and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of the Hospitalist.
References
1. Zhu N et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727-33.
2. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24 (Epub ahead of print).
From the Hospitalist editors: The pediatrics “In the Literature” series generally focuses on original articles. However, given the urgency to learn more about SARS-CoV-2/COVID-19 pandemic and the limited literature about hospitalized pediatric patients with the disease, the editors of the Hospitalist thought it was appropriate to share an article reviewing this letter that was recently published in the New England Journal of Medicine.
Clinical question: What were the clinical characteristics of children in Wuhan, China hospitalized with SARS-CoV-2?
Background: The coronavirus disease 2019 (COVID-19) was recently described by researchers in Wuhan, China.1 However, there has been limited discussion on how the disease has affected children. Based on the Chinese Center for Disease Control and Prevention report, Wu et al. found that 1% of the affected population was less than 10 years, and another 1% of the affected population was 10-19 years.2 However, little information regarding hospitalizations of children with viral infections was previously reported.
Study design: A retrospective analysis of hospitalized children.
Setting: Three sites of a multisite urban teaching hospital in central Wuhan, China.
Synopsis: Over an 8-day period, hospitalized pediatric patients were retrospectively enrolled into this study. The authors defined pediatric patients as those aged 16 years or younger. The patients had one throat swab specimen collected on admission. Throat swab specimens were tested for viral etiologies. In response to the COVID-19 outbreak, the throat samples were retrospectively tested for SARS-CoV-2. If two independent experiments and a clinically verified diagnostic test confirmed the SARS-CoV-2, the cases were confirmed as COVID-19 cases. During the 8-day period, 366 hospitalized pediatric patients were included in the study. Of the 366 patients, 6 tested positive for SARS-CoV-2, while 23 tested positive for influenza A and 20 tested positive for influenza B. The median age of the six patients was 3 years (range, 1-7 years), and all were previously healthy. All six pediatric patients with COVID-19 had high fevers (greater than 39°C), cough, and lymphopenia. Four of the six affected patients had vomiting and leukopenia, while three of the six patients had neutropenia. Four of the six affected patients had pneumonia, as diagnosed on CT scans. Of the six patients, one patient was admitted to the ICU and received intravenous immunoglobulin. The patient admitted to ICU underwent a CT scan which showed “patchy ground-glass opacities in both lungs,” while three of the five children requiring non-ICU hospitalization had chest radiographs showing “patchy shadows in both lungs.” The median length of stay in the hospital was 7.5 days (range, 5-13 days).
Bottom line: COVID-19 causes moderate to severe respiratory illness in pediatric patients with SARS-CoV-2, possibly leading to critical illness. During this time period of the Wuhan COVID-19 outbreak, pediatric patients were more likely to be hospitalized with influenza A or B, than they were with SARS-CoV-2.
Citation: Liu W et al. Detection of Covid-19 in Children in Early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
Dr. Kumar is clinical assistant professor of pediatrics at Case Western Reserve University, Cleveland, and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of the Hospitalist.
References
1. Zhu N et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727-33.
2. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24 (Epub ahead of print).
From the Hospitalist editors: The pediatrics “In the Literature” series generally focuses on original articles. However, given the urgency to learn more about SARS-CoV-2/COVID-19 pandemic and the limited literature about hospitalized pediatric patients with the disease, the editors of the Hospitalist thought it was appropriate to share an article reviewing this letter that was recently published in the New England Journal of Medicine.
Clinical question: What were the clinical characteristics of children in Wuhan, China hospitalized with SARS-CoV-2?
Background: The coronavirus disease 2019 (COVID-19) was recently described by researchers in Wuhan, China.1 However, there has been limited discussion on how the disease has affected children. Based on the Chinese Center for Disease Control and Prevention report, Wu et al. found that 1% of the affected population was less than 10 years, and another 1% of the affected population was 10-19 years.2 However, little information regarding hospitalizations of children with viral infections was previously reported.
Study design: A retrospective analysis of hospitalized children.
Setting: Three sites of a multisite urban teaching hospital in central Wuhan, China.
Synopsis: Over an 8-day period, hospitalized pediatric patients were retrospectively enrolled into this study. The authors defined pediatric patients as those aged 16 years or younger. The patients had one throat swab specimen collected on admission. Throat swab specimens were tested for viral etiologies. In response to the COVID-19 outbreak, the throat samples were retrospectively tested for SARS-CoV-2. If two independent experiments and a clinically verified diagnostic test confirmed the SARS-CoV-2, the cases were confirmed as COVID-19 cases. During the 8-day period, 366 hospitalized pediatric patients were included in the study. Of the 366 patients, 6 tested positive for SARS-CoV-2, while 23 tested positive for influenza A and 20 tested positive for influenza B. The median age of the six patients was 3 years (range, 1-7 years), and all were previously healthy. All six pediatric patients with COVID-19 had high fevers (greater than 39°C), cough, and lymphopenia. Four of the six affected patients had vomiting and leukopenia, while three of the six patients had neutropenia. Four of the six affected patients had pneumonia, as diagnosed on CT scans. Of the six patients, one patient was admitted to the ICU and received intravenous immunoglobulin. The patient admitted to ICU underwent a CT scan which showed “patchy ground-glass opacities in both lungs,” while three of the five children requiring non-ICU hospitalization had chest radiographs showing “patchy shadows in both lungs.” The median length of stay in the hospital was 7.5 days (range, 5-13 days).
Bottom line: COVID-19 causes moderate to severe respiratory illness in pediatric patients with SARS-CoV-2, possibly leading to critical illness. During this time period of the Wuhan COVID-19 outbreak, pediatric patients were more likely to be hospitalized with influenza A or B, than they were with SARS-CoV-2.
Citation: Liu W et al. Detection of Covid-19 in Children in Early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
Dr. Kumar is clinical assistant professor of pediatrics at Case Western Reserve University, Cleveland, and a pediatric hospitalist at Cleveland Clinic Children’s. She is the pediatric editor of the Hospitalist.
References
1. Zhu N et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727-33.
2. Wu Z, McGoogan JM. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: Summary of a report of 72,314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 Feb 24 (Epub ahead of print).
From the Hospitalist editors: The pediatrics “In the Literature” series generally focuses on original articles. However, given the urgency to learn more about SARS-CoV-2/COVID-19 pandemic and the limited literature about hospitalized pediatric patients with the disease, the editors of the Hospitalist thought it was appropriate to share an article reviewing this letter that was recently published in the New England Journal of Medicine.
Flattening the curve: Viral graphic shows COVID-19 containment needs
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.
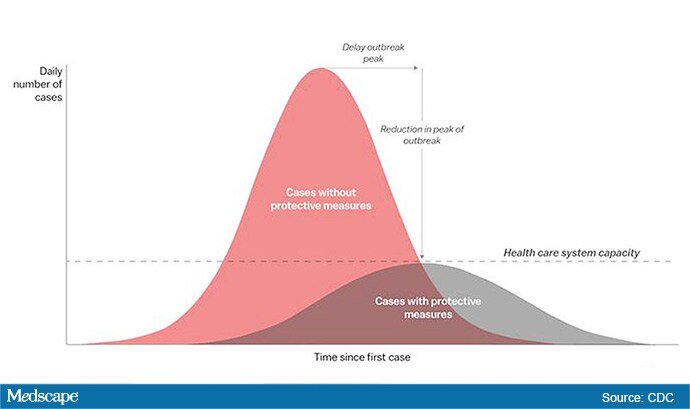
Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.

Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
Editor’s note: Find the latest COVID-19 news and guidance in Medscape’s Coronavirus Resource Center.
The “Flattening the Curve” graphic, which has, to not use the term lightly, gone viral on social media, visually explains the best currently available strategy to stop the COVID-19 spread, experts told Medscape Medical News.
The height of the curve is the number of potential cases in the United States; along the horizontal X axis, or the breadth, is the amount of time. The line across the middle represents the point at which too many cases in too short a time overwhelm the healthcare system.

Jeanne Marrazzo, MD, MPH, director of the Division of Infectious Diseases at the University of Alabama at Birmingham’s School of Medicine explained.
“Not only are you spreading out the new cases but the rate at which people recover,” she told Medscape Medical News. “You have time to get people out of the hospital so you can get new people in and clear out those beds.”
The strategy, with its own Twitter hashtag, #Flattenthecurve, “is about all we have,” without a vaccine, Marrazzo said.
Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said avoiding spikes in cases could mean fewer deaths.
“If you look at the curves of outbreaks, you know, they go big peaks, and then they come down. What we need to do is flatten that down,” Fauci said March 10 in a White House briefing. “You do that by trying to interfere with the natural flow of the outbreak.”
Wuhan, China, at the epicenter of the pandemic, “had an explosive curve” and quickly got overwhelmed without early containment measures, Marrazzo noted. “If you look at Italy right now, it’s clearly in the same situation.”
The Race Is On to Interrupt the Spread
The race is on in the US to interrupt the transmission of the virus and slow the spread, meaning containment measures have increasingly higher and wider stakes.
Closing down Broadway shows and some theme parks and massive sporting events; the escalating numbers of people working from home; and businesses cutting hours or closing all demonstrate the level of US confidence that “social distancing” will work, Marrazzo said.
“We’re clearly ready to disrupt the economy and social infrastructure,” she said.
That appears to have made a difference in Wuhan, Marrazzo said, as the new infections are coming down.
The question, she said, is “we’re not China – so are Americans really going to take to this? Americans greatly value their liberty and there’s some skepticism about public health and its directives. People have never seen a pandemic like this before.”
Dena Grayson, MD, PhD, a Florida-based expert in Ebola and other pandemic threats, told Medscape Medical News that EvergreenHealth in Kirkland, Washington, is a good example of what it means when a virus overwhelms healthcare operations.
The New York Times reported that supplies were so strained at the facility that staff were using sanitary napkins to pad protective helmets.
As of March 11, 65 people who had come into the hospital have tested positive for the virus, and 15 of them had died.
Grayson points out that the COVID-19 cases come on top of a severe flu season and the usual cases hospitals see, so the bar on the graphic is even lower than it usually would be.
“We have a relatively limited capacity with ICU beds to begin with,” she said.
So far, closures, postponements, and cancellations are woefully inadequate, Grayson said.
“We can’t stop this virus. We can hope to contain it and slow down the rate of infection,” she said.
“We need to right now shut down all the schools, preschools, and universities,” Grayson said. “We need to look at shutting down public transportation. We need people to stay home – and not for a day but for a couple of weeks.”
The graphic was developed by visual-data journalist Rosamund Pearce, based on a graphic that had appeared in a Centers for Disease Control and Prevention (CDC) article titled “Community Mitigation Guidelines to Prevent Pandemic Influenza,” the Times reports.
Marrazzo and Grayson have disclosed no relevant financial relationships.
This story first appeared on Medscape.com .
President declares national emergency for COVID-19, ramps up testing capability
President Donald Trump has declared a national emergency to allow for additional resources to combat the COVID-19 pandemic and announced increased testing capacity in partnership with private industry.
During a March 13 press conference, the president said the declaration would “open up access to up to $50 billion” for states and territories in combating the spread of the disease.
He also called on all states to “set up emergency operation centers, effective immediately” and for every hospital “to activate its emergency preparedness plan so that they can meet the needs of Americans everywhere.”
Additionally, he said the declaration will confer broad new authority on the Department of Health & Human Services Secretary Alex Azar that will allow him to “immediately waive provisions of applicable laws and regulations to give doctors, all hospitals, and health care providers maximum flexibility to respond to the virus and care for patients.”
Some of the powers he highlighted included the ability to waive laws to enable telehealth; to waive certain federal license requirements to allow doctors licensed in one state to offer services in other states; the ability to waive limits on beds in critical access hospitals; and to waive rules that hinder hospitals from hiring additional physicians.
The president also announced that more testing capacity will be made available within the next week, in partnership with private industry.
“We want to make sure that those who need a test can get a test very safely, quickly, and conveniently, but we don’t want people to take a test if we feel that they shouldn’t be doing it,” he said.
To help make that determination, a website, developed with Google, is expected to be launched the weekend of March 13 to will allow individuals to input their symptoms and risk factors to help determine if they should be tested. If certain criteria are met, the website will provide locations for drive-through testing facilities. Individuals will be tested using a nasal swab and will receive results within 24-36 hours.
The testing is being done in partnership with retailers, including Target and Walmart (who are providing parking lot space for the pop-up testing facilities) and testing companies LabCorp and Quest Diagnostics.
The new test was developed by Roche and just received emergency use authorization from the Food and Drug Administration.
“We therefore expect up to a half-million additional tests will be available early next week,” President Trump said, adding that testing locations will “probably” be announced on Sunday, March 15.
A second application for a new test, submitted by Thermo Fisher, is currently under review at the FDA and is expected to be approved within the next 24 hours, he said. This would add an additional 1.4 million tests in the next week and 5 million within a month, according to the president.
President Donald Trump has declared a national emergency to allow for additional resources to combat the COVID-19 pandemic and announced increased testing capacity in partnership with private industry.
During a March 13 press conference, the president said the declaration would “open up access to up to $50 billion” for states and territories in combating the spread of the disease.
He also called on all states to “set up emergency operation centers, effective immediately” and for every hospital “to activate its emergency preparedness plan so that they can meet the needs of Americans everywhere.”
Additionally, he said the declaration will confer broad new authority on the Department of Health & Human Services Secretary Alex Azar that will allow him to “immediately waive provisions of applicable laws and regulations to give doctors, all hospitals, and health care providers maximum flexibility to respond to the virus and care for patients.”
Some of the powers he highlighted included the ability to waive laws to enable telehealth; to waive certain federal license requirements to allow doctors licensed in one state to offer services in other states; the ability to waive limits on beds in critical access hospitals; and to waive rules that hinder hospitals from hiring additional physicians.
The president also announced that more testing capacity will be made available within the next week, in partnership with private industry.
“We want to make sure that those who need a test can get a test very safely, quickly, and conveniently, but we don’t want people to take a test if we feel that they shouldn’t be doing it,” he said.
To help make that determination, a website, developed with Google, is expected to be launched the weekend of March 13 to will allow individuals to input their symptoms and risk factors to help determine if they should be tested. If certain criteria are met, the website will provide locations for drive-through testing facilities. Individuals will be tested using a nasal swab and will receive results within 24-36 hours.
The testing is being done in partnership with retailers, including Target and Walmart (who are providing parking lot space for the pop-up testing facilities) and testing companies LabCorp and Quest Diagnostics.
The new test was developed by Roche and just received emergency use authorization from the Food and Drug Administration.
“We therefore expect up to a half-million additional tests will be available early next week,” President Trump said, adding that testing locations will “probably” be announced on Sunday, March 15.
A second application for a new test, submitted by Thermo Fisher, is currently under review at the FDA and is expected to be approved within the next 24 hours, he said. This would add an additional 1.4 million tests in the next week and 5 million within a month, according to the president.
President Donald Trump has declared a national emergency to allow for additional resources to combat the COVID-19 pandemic and announced increased testing capacity in partnership with private industry.
During a March 13 press conference, the president said the declaration would “open up access to up to $50 billion” for states and territories in combating the spread of the disease.
He also called on all states to “set up emergency operation centers, effective immediately” and for every hospital “to activate its emergency preparedness plan so that they can meet the needs of Americans everywhere.”
Additionally, he said the declaration will confer broad new authority on the Department of Health & Human Services Secretary Alex Azar that will allow him to “immediately waive provisions of applicable laws and regulations to give doctors, all hospitals, and health care providers maximum flexibility to respond to the virus and care for patients.”
Some of the powers he highlighted included the ability to waive laws to enable telehealth; to waive certain federal license requirements to allow doctors licensed in one state to offer services in other states; the ability to waive limits on beds in critical access hospitals; and to waive rules that hinder hospitals from hiring additional physicians.
The president also announced that more testing capacity will be made available within the next week, in partnership with private industry.
“We want to make sure that those who need a test can get a test very safely, quickly, and conveniently, but we don’t want people to take a test if we feel that they shouldn’t be doing it,” he said.
To help make that determination, a website, developed with Google, is expected to be launched the weekend of March 13 to will allow individuals to input their symptoms and risk factors to help determine if they should be tested. If certain criteria are met, the website will provide locations for drive-through testing facilities. Individuals will be tested using a nasal swab and will receive results within 24-36 hours.
The testing is being done in partnership with retailers, including Target and Walmart (who are providing parking lot space for the pop-up testing facilities) and testing companies LabCorp and Quest Diagnostics.
The new test was developed by Roche and just received emergency use authorization from the Food and Drug Administration.
“We therefore expect up to a half-million additional tests will be available early next week,” President Trump said, adding that testing locations will “probably” be announced on Sunday, March 15.
A second application for a new test, submitted by Thermo Fisher, is currently under review at the FDA and is expected to be approved within the next 24 hours, he said. This would add an additional 1.4 million tests in the next week and 5 million within a month, according to the president.
After weeks of decline, influenza activity increases slightly
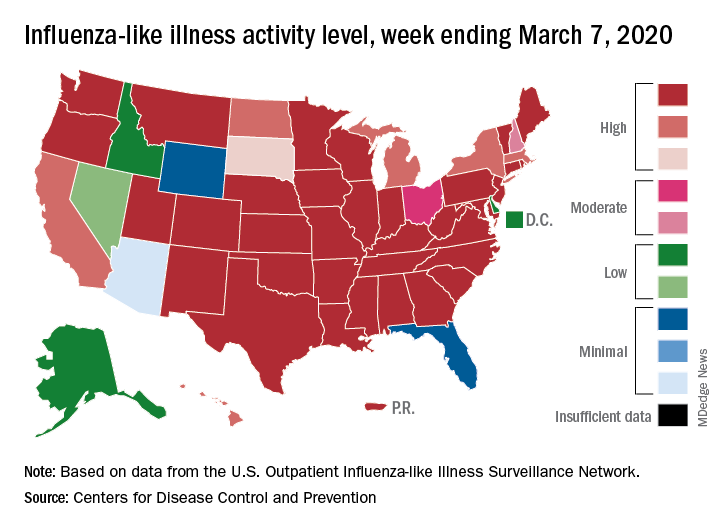
The two leading measures of influenza activity – the percentage of respiratory specimens testing positive for influenza and the proportion of visits to health care providers for influenza-like illness (ILI) – had been following a similar downward path since mid-February. But during the week ending March 7, their paths diverged, according to the Centers for Disease Control and Prevention.
The percentage of respiratory specimens testing positive for influenza dropped for the fourth consecutive week, falling from 26.1% to 21.5%, while the proportion of visits to health care providers for ILI increased from 5.1% to 5.2%, the CDC’s influenza division reported.
One possible explanation for that rise: “The largest increases in ILI activity occurred in areas of the country where COVID-19 is most prevalent. More people may be seeking care for respiratory illness than usual at this time,” the influenza division said March 13 in its weekly Fluview report.
This week’s map puts 34 states and Puerto Rico at level 10 on the CDC’s 1-10 scale of ILI activity, one more state than the week before, and 43 jurisdictions in the “high” range of 8-10, compared with 42 the previous week, the CDC said.
Rates of hospitalizations associated with influenza “remain moderate compared to recent seasons, but rates for children 0-4 years and adults 18-49 years are now the highest CDC has on record for these age groups, surpassing rates reported during the 2009 H1N1 pandemic,” the Fluview report said. Rates for children aged 5-17 years “are higher than any recent regular season but remain lower than rates experienced by this age group during the pandemic.”
The number of pediatric deaths this season is now up to 144, equaling the total for all of the 2018-2019 season. This year’s count led the CDC to invoke 2009 again, since it “is higher for the same time period than in every season since reporting began in 2004-2005, except for the 2009 pandemic.”
For the 2019-2020 season so far there have been 36 million flu illnesses, 370,000 hospitalizations, and 22,000 deaths from flu and pneumonia, the CDC estimated.

The two leading measures of influenza activity – the percentage of respiratory specimens testing positive for influenza and the proportion of visits to health care providers for influenza-like illness (ILI) – had been following a similar downward path since mid-February. But during the week ending March 7, their paths diverged, according to the Centers for Disease Control and Prevention.
The percentage of respiratory specimens testing positive for influenza dropped for the fourth consecutive week, falling from 26.1% to 21.5%, while the proportion of visits to health care providers for ILI increased from 5.1% to 5.2%, the CDC’s influenza division reported.
One possible explanation for that rise: “The largest increases in ILI activity occurred in areas of the country where COVID-19 is most prevalent. More people may be seeking care for respiratory illness than usual at this time,” the influenza division said March 13 in its weekly Fluview report.
This week’s map puts 34 states and Puerto Rico at level 10 on the CDC’s 1-10 scale of ILI activity, one more state than the week before, and 43 jurisdictions in the “high” range of 8-10, compared with 42 the previous week, the CDC said.
Rates of hospitalizations associated with influenza “remain moderate compared to recent seasons, but rates for children 0-4 years and adults 18-49 years are now the highest CDC has on record for these age groups, surpassing rates reported during the 2009 H1N1 pandemic,” the Fluview report said. Rates for children aged 5-17 years “are higher than any recent regular season but remain lower than rates experienced by this age group during the pandemic.”
The number of pediatric deaths this season is now up to 144, equaling the total for all of the 2018-2019 season. This year’s count led the CDC to invoke 2009 again, since it “is higher for the same time period than in every season since reporting began in 2004-2005, except for the 2009 pandemic.”
For the 2019-2020 season so far there have been 36 million flu illnesses, 370,000 hospitalizations, and 22,000 deaths from flu and pneumonia, the CDC estimated.

The two leading measures of influenza activity – the percentage of respiratory specimens testing positive for influenza and the proportion of visits to health care providers for influenza-like illness (ILI) – had been following a similar downward path since mid-February. But during the week ending March 7, their paths diverged, according to the Centers for Disease Control and Prevention.
The percentage of respiratory specimens testing positive for influenza dropped for the fourth consecutive week, falling from 26.1% to 21.5%, while the proportion of visits to health care providers for ILI increased from 5.1% to 5.2%, the CDC’s influenza division reported.
One possible explanation for that rise: “The largest increases in ILI activity occurred in areas of the country where COVID-19 is most prevalent. More people may be seeking care for respiratory illness than usual at this time,” the influenza division said March 13 in its weekly Fluview report.
This week’s map puts 34 states and Puerto Rico at level 10 on the CDC’s 1-10 scale of ILI activity, one more state than the week before, and 43 jurisdictions in the “high” range of 8-10, compared with 42 the previous week, the CDC said.
Rates of hospitalizations associated with influenza “remain moderate compared to recent seasons, but rates for children 0-4 years and adults 18-49 years are now the highest CDC has on record for these age groups, surpassing rates reported during the 2009 H1N1 pandemic,” the Fluview report said. Rates for children aged 5-17 years “are higher than any recent regular season but remain lower than rates experienced by this age group during the pandemic.”
The number of pediatric deaths this season is now up to 144, equaling the total for all of the 2018-2019 season. This year’s count led the CDC to invoke 2009 again, since it “is higher for the same time period than in every season since reporting began in 2004-2005, except for the 2009 pandemic.”
For the 2019-2020 season so far there have been 36 million flu illnesses, 370,000 hospitalizations, and 22,000 deaths from flu and pneumonia, the CDC estimated.
FDA warns of serious infection risk after FMT
The Food and Drug Administration has issued a Safety Alert warning of the potential risk of serious, life-threatening infection in patients who receive fecal microbiota transplant for Clostridioides difficile infection.
The FDA has received six reports of infection associated with fecal microbiota transplant from a stool bank company based in the United States: Two patients had enteropathogenic Escherichia coli (EPEC) infection, and four had shiga toxin–producing E. coli (STEC). The two EPEC infections came from two separate donors, but the four STEC infections came from a single donor, according to the FDA.
In addition, two patients died after receiving fecal microbiota transplant from the donor associated with the STEC infections. These patients died before any of the STEC infections were reported to the FDA; as their stool was not tested for STEC, it is unclear whether it contributed to their deaths.
The use of fecal microbiota transplant is still investigational, and as such, patients should be made aware by health care providers of the risks, which include the potential for transmission of pathogenic bacteria and the resultant adverse events, the FDA said in the press release.
The Food and Drug Administration has issued a Safety Alert warning of the potential risk of serious, life-threatening infection in patients who receive fecal microbiota transplant for Clostridioides difficile infection.
The FDA has received six reports of infection associated with fecal microbiota transplant from a stool bank company based in the United States: Two patients had enteropathogenic Escherichia coli (EPEC) infection, and four had shiga toxin–producing E. coli (STEC). The two EPEC infections came from two separate donors, but the four STEC infections came from a single donor, according to the FDA.
In addition, two patients died after receiving fecal microbiota transplant from the donor associated with the STEC infections. These patients died before any of the STEC infections were reported to the FDA; as their stool was not tested for STEC, it is unclear whether it contributed to their deaths.
The use of fecal microbiota transplant is still investigational, and as such, patients should be made aware by health care providers of the risks, which include the potential for transmission of pathogenic bacteria and the resultant adverse events, the FDA said in the press release.
The Food and Drug Administration has issued a Safety Alert warning of the potential risk of serious, life-threatening infection in patients who receive fecal microbiota transplant for Clostridioides difficile infection.
The FDA has received six reports of infection associated with fecal microbiota transplant from a stool bank company based in the United States: Two patients had enteropathogenic Escherichia coli (EPEC) infection, and four had shiga toxin–producing E. coli (STEC). The two EPEC infections came from two separate donors, but the four STEC infections came from a single donor, according to the FDA.
In addition, two patients died after receiving fecal microbiota transplant from the donor associated with the STEC infections. These patients died before any of the STEC infections were reported to the FDA; as their stool was not tested for STEC, it is unclear whether it contributed to their deaths.
The use of fecal microbiota transplant is still investigational, and as such, patients should be made aware by health care providers of the risks, which include the potential for transmission of pathogenic bacteria and the resultant adverse events, the FDA said in the press release.
A point-of-care urine test is on the way for PrEP adherence
A simple, quick point-of-care urine test for tenofovir adherence, similar to an OTC-pregnancy test, has an accuracy of 99.6% versus laboratory testing, according to a report at the Conference on Retroviruses & Opportunistic Infections.
A few drops of urine yield results in 5 minutes, and tell if patients have been taking tenofovir, a key component of HIV preexposure prophylaxis (PrEP) medications, within the previous 4-7 days. Abbott Rapid Diagnostics is gearing up to market the test widely in the United States, and it won’t be very expensive, according to lead investigator Matthew Spinelli, MD, a clinical fellow and HIV/AIDS researcher at the University of California, San Francisco.
It’s an alternative to the usual approach, measuring tenofovir levels in hair, blood, or urine by liquid chromatography and mass spectrometry. That approach is expensive and requires trained personnel, and the results can take a while. Dr. Spinelli and colleagues saw the need for a quicker, easier way for use in the clinic, since real-time adherence results are most likely to make a difference, he said at the meeting, which was scheduled to be in Boston, but was held online instead this year because of concerns about spreading the COVID-19 virus.
Self-report, meanwhile, is notoriously unreliable. Over 90% of people in two previous PrEP studies said they were taking their medications, but only about a quarter had tenofovir in their plasma.
The investigators identified a tenofovir antibody in urine that could be read by enzyme-linked immunosorbent assay (ELISA), and validated it for adherence accuracy against liquid chromatography and mass spectrometry; they then put the antibody on a test strip to create a lateral flow immunoassay.
After hearing the presentation, moderator Susan Buchbinder, MD, director of HIV prevention research at the San Francisco Department of Public Health, called the work “important” and said it “really has the possibility of opening up a lot of new kinds of studies and new kinds of intervention for both prevention and treatment.”
Dr. Spinelli and colleagues pitted the test strip against their laboratory-based ELISA test using 684 stored urine samples from 324 men and women in disoproxil fumarate/emtricitabine (Truvada) PrEP projects in Africa and the United States.
Overall, the 505 samples that were positive for tenofovir in the lab test were also positive on the urine strip, yielding 100% sensitivity. Of the 179 negative samples on the lab test, 176 were also negative with the strip, yielding a specificity of 98.3%. The results calculated into nearly perfect accuracy.
“We believe that” the urine test strip “is ready for field testing,” and that “point-of-care adherence testing” will be a boon to both PrEP and HIV treatment. A negative test, for instance, would signal the need for immediate counseling, and the patient would still be in the office to hear it. For HIV, high adherence but also high viral load would signal the need for resistance testing, Dr. Spinelli said.
A white-coat effect is possible; people might take their medication when they know they have an upcoming doctor’s appointment. “We will need to evaluate for [that] with additional studies” comparing point-of-care testing with longer-term metrics, such as drug levels in hair, he said.
The study was published to coincide with Dr. Spinelli’s report (J Acquir Immune Defic Syndr. 2020 Mar 10. doi: 10.1097/QAI.0000000000002322).
The funding source wasn’t reported. Dr. Spinelli had no disclosures. Two investigators were Abbott employees.
SOURCE: Spinelli MA et al. 2020 CROI abstract 91.
A simple, quick point-of-care urine test for tenofovir adherence, similar to an OTC-pregnancy test, has an accuracy of 99.6% versus laboratory testing, according to a report at the Conference on Retroviruses & Opportunistic Infections.
A few drops of urine yield results in 5 minutes, and tell if patients have been taking tenofovir, a key component of HIV preexposure prophylaxis (PrEP) medications, within the previous 4-7 days. Abbott Rapid Diagnostics is gearing up to market the test widely in the United States, and it won’t be very expensive, according to lead investigator Matthew Spinelli, MD, a clinical fellow and HIV/AIDS researcher at the University of California, San Francisco.
It’s an alternative to the usual approach, measuring tenofovir levels in hair, blood, or urine by liquid chromatography and mass spectrometry. That approach is expensive and requires trained personnel, and the results can take a while. Dr. Spinelli and colleagues saw the need for a quicker, easier way for use in the clinic, since real-time adherence results are most likely to make a difference, he said at the meeting, which was scheduled to be in Boston, but was held online instead this year because of concerns about spreading the COVID-19 virus.
Self-report, meanwhile, is notoriously unreliable. Over 90% of people in two previous PrEP studies said they were taking their medications, but only about a quarter had tenofovir in their plasma.
The investigators identified a tenofovir antibody in urine that could be read by enzyme-linked immunosorbent assay (ELISA), and validated it for adherence accuracy against liquid chromatography and mass spectrometry; they then put the antibody on a test strip to create a lateral flow immunoassay.
After hearing the presentation, moderator Susan Buchbinder, MD, director of HIV prevention research at the San Francisco Department of Public Health, called the work “important” and said it “really has the possibility of opening up a lot of new kinds of studies and new kinds of intervention for both prevention and treatment.”
Dr. Spinelli and colleagues pitted the test strip against their laboratory-based ELISA test using 684 stored urine samples from 324 men and women in disoproxil fumarate/emtricitabine (Truvada) PrEP projects in Africa and the United States.
Overall, the 505 samples that were positive for tenofovir in the lab test were also positive on the urine strip, yielding 100% sensitivity. Of the 179 negative samples on the lab test, 176 were also negative with the strip, yielding a specificity of 98.3%. The results calculated into nearly perfect accuracy.
“We believe that” the urine test strip “is ready for field testing,” and that “point-of-care adherence testing” will be a boon to both PrEP and HIV treatment. A negative test, for instance, would signal the need for immediate counseling, and the patient would still be in the office to hear it. For HIV, high adherence but also high viral load would signal the need for resistance testing, Dr. Spinelli said.
A white-coat effect is possible; people might take their medication when they know they have an upcoming doctor’s appointment. “We will need to evaluate for [that] with additional studies” comparing point-of-care testing with longer-term metrics, such as drug levels in hair, he said.
The study was published to coincide with Dr. Spinelli’s report (J Acquir Immune Defic Syndr. 2020 Mar 10. doi: 10.1097/QAI.0000000000002322).
The funding source wasn’t reported. Dr. Spinelli had no disclosures. Two investigators were Abbott employees.
SOURCE: Spinelli MA et al. 2020 CROI abstract 91.
A simple, quick point-of-care urine test for tenofovir adherence, similar to an OTC-pregnancy test, has an accuracy of 99.6% versus laboratory testing, according to a report at the Conference on Retroviruses & Opportunistic Infections.
A few drops of urine yield results in 5 minutes, and tell if patients have been taking tenofovir, a key component of HIV preexposure prophylaxis (PrEP) medications, within the previous 4-7 days. Abbott Rapid Diagnostics is gearing up to market the test widely in the United States, and it won’t be very expensive, according to lead investigator Matthew Spinelli, MD, a clinical fellow and HIV/AIDS researcher at the University of California, San Francisco.
It’s an alternative to the usual approach, measuring tenofovir levels in hair, blood, or urine by liquid chromatography and mass spectrometry. That approach is expensive and requires trained personnel, and the results can take a while. Dr. Spinelli and colleagues saw the need for a quicker, easier way for use in the clinic, since real-time adherence results are most likely to make a difference, he said at the meeting, which was scheduled to be in Boston, but was held online instead this year because of concerns about spreading the COVID-19 virus.
Self-report, meanwhile, is notoriously unreliable. Over 90% of people in two previous PrEP studies said they were taking their medications, but only about a quarter had tenofovir in their plasma.
The investigators identified a tenofovir antibody in urine that could be read by enzyme-linked immunosorbent assay (ELISA), and validated it for adherence accuracy against liquid chromatography and mass spectrometry; they then put the antibody on a test strip to create a lateral flow immunoassay.
After hearing the presentation, moderator Susan Buchbinder, MD, director of HIV prevention research at the San Francisco Department of Public Health, called the work “important” and said it “really has the possibility of opening up a lot of new kinds of studies and new kinds of intervention for both prevention and treatment.”
Dr. Spinelli and colleagues pitted the test strip against their laboratory-based ELISA test using 684 stored urine samples from 324 men and women in disoproxil fumarate/emtricitabine (Truvada) PrEP projects in Africa and the United States.
Overall, the 505 samples that were positive for tenofovir in the lab test were also positive on the urine strip, yielding 100% sensitivity. Of the 179 negative samples on the lab test, 176 were also negative with the strip, yielding a specificity of 98.3%. The results calculated into nearly perfect accuracy.
“We believe that” the urine test strip “is ready for field testing,” and that “point-of-care adherence testing” will be a boon to both PrEP and HIV treatment. A negative test, for instance, would signal the need for immediate counseling, and the patient would still be in the office to hear it. For HIV, high adherence but also high viral load would signal the need for resistance testing, Dr. Spinelli said.
A white-coat effect is possible; people might take their medication when they know they have an upcoming doctor’s appointment. “We will need to evaluate for [that] with additional studies” comparing point-of-care testing with longer-term metrics, such as drug levels in hair, he said.
The study was published to coincide with Dr. Spinelli’s report (J Acquir Immune Defic Syndr. 2020 Mar 10. doi: 10.1097/QAI.0000000000002322).
The funding source wasn’t reported. Dr. Spinelli had no disclosures. Two investigators were Abbott employees.
SOURCE: Spinelli MA et al. 2020 CROI abstract 91.
FROM CROI 2020
Wuhan case review: COVID-19 characteristics differ in children vs. adults
Pediatric cases of COVID-19 infection are typically mild, but underlying coinfection may be more common in children than in adults, according to an analysis of clinical, laboratory, and chest CT features of pediatric inpatients in Wuhan, China.

The findings point toward a need for early chest CT with corresponding pathogen detection in children with suspected COVID-19 infection, Wei Xia, MD, of Huazhong University of Science and Technology, Wuhan, China, and colleagues reported in Pediatric Pulmonology.
The most common symptoms in 20 pediatric patients hospitalized between Jan. 23 and Feb. 8, 2020, with COVID-19 infection confirmed by the pharyngeal swab COVID-19 nucleic acid test were fever and cough, which occurred in 60% and 65% of patients, respectively. Coinfection was detected in eight patients (40%), they noted.
Clinical manifestations were similar to those seen in adults, but overall symptoms were relatively mild and overall prognosis was good. Of particular note, 7 of the 20 (35%) patients had a previously diagnosed congenital or acquired diseases, suggesting that children with underlying conditions may be more susceptible, Dr. Xia and colleagues wrote.
Laboratory findings also were notable in that 80% of the children had procalcitonin (PCT) elevations not typically seen in adults with COVID-19. PCT is a marker for bacterial infection and “[this finding] may suggest that routine antibacterial treatment should be considered in pediatric patients,” the investigators wrote.
As for imaging results, chest CT findings in children were similar to those in adults.“The typical manifestations were unilateral or bilateral subpleural ground-glass opacities, and consolidations with surrounding halo signs,” Dr. Xia and associates wrote, adding that consolidations with surrounding halo sign accounted for about half the pediatric cases and should be considered as “typical signs in pediatric patients.”
Pediatric cases were “rather rare” in the early days of the COVID-19 outbreak in Wuhan, where the first cases of infection were reported.
“As a pediatric group is usually susceptible to upper respiratory tract infection, because of their developing immune system, the delayed presence of pediatric patients is confusing,” the investigators wrote, noting that a low detection rate of pharyngeal swab COVID-19 nucleic acid test, distinguishing the virus from other common respiratory tract infectious pathogens in pediatric patients, “is still a problem.”
To better characterize the clinical and imaging features in children versus adults with COVID-19, Dr. Xia and associates reviewed these 20 pediatric cases, including 13 boys and 7 girls with ages ranging from less than 1 month to 14 years, 7 months (median 2 years, 1.5 months). Thirteen had an identified close contact with a COVID-19–diagnosed family member, and all were treated in an isolation ward. A total of 18 children were cured and discharged after an average stay of 13 days, and 2 neonates remained under observation because of positive swab results with negative CT findings. The investigators speculated that the different findings in neonates were perhaps caused by the influence of delivery on sampling or the specific CT manifestations for neonates, adding that more samples are needed for further clarification.
Based on these findings, “the CT imaging of COVID-19 infection should be differentiated with other virus pneumonias such as influenza virus, parainfluenza virus, respiratory syncytial virus, and adenovirus,” they concluded. It also should “be differentiated from bacterial pneumonia, mycoplasma pneumonia, and chlamydia pneumonia ... the density of pneumonia lesions caused by the latter pathogens is relatively higher.”
However, Dr. Xia and colleagues noted that chest CT manifestations of pneumonia caused by different pathogens overlap, and COVID-19 pneumonia “can be superimposed with serious and complex imaging manifestations, so epidemiological and etiological examinations should be combined.”
The investigators concluded that COVID-19 virus pneumonia in children is generally mild, and that the characteristic changes of subpleural ground-glass opacities and consolidations with surrounding halo on chest CT provide an “effective means for follow-up and evaluating the changes of lung lesions.”
“In the case that the positive rate of COVID-19 nucleic acid test from pharyngeal swab samples is not high, the early detection of lesions by CT is conducive to reasonable management and early treatment for pediatric patients. However, the diagnosis of COVID-19 pneumonia by CT imaging alone is not sufficient enough, especially in the case of coinfection with other pathogens,” Dr. Xia and associates wrote. “Therefore, early chest CT screening and timely follow-up, combined with corresponding pathogen detection, is a feasible clinical protocol in children.”
An early study
In a separate retrospective analysis described in a letter to the editor of the New England Journal of Medicine, Weiyong Liu, PhD, of Tongji Hospital of Huazhong University of Science and Technology and colleagues found that the most frequently detected pathogens in 366 children under the age of 16 years hospitalized with respiratory infections in Wuhan during Jan. 7-15, 2020, were influenza A virus (6.3% of cases) and influenza B virus (5.5% of cases), whereas COVID-19 was detected in 1.6% of cases.
The median age of the COVID-19 patients in that series was 3 years (range 1-7 years), and in contrast to the findings of Xia et al., all previously had been “completely healthy.” Common characteristics were high fever and cough in all six patients, and vomiting in four patients. Five had pneumonia as assessed by X-ray, and CTs showed typical viral pneumonia patterns.
One patient was admitted to a pediatric ICU. All patients received antiviral agents, antibiotic agents, and supportive therapies; all recovered after a median hospital stay of 7.5 days (median range, 5-13 days).
In contrast with the findings of Xia et al., the findings of Liu et al. showed COVID-19 caused moderate to severe respiratory illness in children, and that infections in children were occurring early in the epidemic.
Some perspective
In an interview regarding the findings by Xia et al., Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University, and director of pediatric infectious diseases at Boston Medical Center, noted the absence of fever in 40% of cases.
“This is important, as the criteria for testing by public health departments has been high fever, cough, and shortness of breath,” he said. “The absence of fever is not inconsistent with COVID-19 disease.”
Another important point regarding the findings by Xia et al. is that the highest attack rates appear to be in children under 1 year of age, he said, further noting that the finding of concurrent influenza A, influenza B, or respiratory syncytial virus underscores that “concurrent infection can occur, and the presence of another virus in diagnostic tests does not mean that COVID-19 is not causal.”
As for whether the finding of elevated procalcitonin levels in 80% of cases reflects COVID-19 disease or coinfection with bacteria, the answer is unclear. But none of the children in the study were proven to have bacterial disease, he said, adding that “this marker will need to be interpreted with caution in the setting of COVID-19 disease.”
Dr. Xia and colleagues reported having no disclosures. Dr. Liu and associates also reported having no disclosures. The study by Liu et al. was supported by the Ministry of Science and Technology of China, the National Mega Project on Major Infectious Disease Prevention, and the National Key Research and Development Program of China.
SOURCES: Xia W et al. Ped Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718; Liu W et al. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
Pediatric cases of COVID-19 infection are typically mild, but underlying coinfection may be more common in children than in adults, according to an analysis of clinical, laboratory, and chest CT features of pediatric inpatients in Wuhan, China.

The findings point toward a need for early chest CT with corresponding pathogen detection in children with suspected COVID-19 infection, Wei Xia, MD, of Huazhong University of Science and Technology, Wuhan, China, and colleagues reported in Pediatric Pulmonology.
The most common symptoms in 20 pediatric patients hospitalized between Jan. 23 and Feb. 8, 2020, with COVID-19 infection confirmed by the pharyngeal swab COVID-19 nucleic acid test were fever and cough, which occurred in 60% and 65% of patients, respectively. Coinfection was detected in eight patients (40%), they noted.
Clinical manifestations were similar to those seen in adults, but overall symptoms were relatively mild and overall prognosis was good. Of particular note, 7 of the 20 (35%) patients had a previously diagnosed congenital or acquired diseases, suggesting that children with underlying conditions may be more susceptible, Dr. Xia and colleagues wrote.
Laboratory findings also were notable in that 80% of the children had procalcitonin (PCT) elevations not typically seen in adults with COVID-19. PCT is a marker for bacterial infection and “[this finding] may suggest that routine antibacterial treatment should be considered in pediatric patients,” the investigators wrote.
As for imaging results, chest CT findings in children were similar to those in adults.“The typical manifestations were unilateral or bilateral subpleural ground-glass opacities, and consolidations with surrounding halo signs,” Dr. Xia and associates wrote, adding that consolidations with surrounding halo sign accounted for about half the pediatric cases and should be considered as “typical signs in pediatric patients.”
Pediatric cases were “rather rare” in the early days of the COVID-19 outbreak in Wuhan, where the first cases of infection were reported.
“As a pediatric group is usually susceptible to upper respiratory tract infection, because of their developing immune system, the delayed presence of pediatric patients is confusing,” the investigators wrote, noting that a low detection rate of pharyngeal swab COVID-19 nucleic acid test, distinguishing the virus from other common respiratory tract infectious pathogens in pediatric patients, “is still a problem.”
To better characterize the clinical and imaging features in children versus adults with COVID-19, Dr. Xia and associates reviewed these 20 pediatric cases, including 13 boys and 7 girls with ages ranging from less than 1 month to 14 years, 7 months (median 2 years, 1.5 months). Thirteen had an identified close contact with a COVID-19–diagnosed family member, and all were treated in an isolation ward. A total of 18 children were cured and discharged after an average stay of 13 days, and 2 neonates remained under observation because of positive swab results with negative CT findings. The investigators speculated that the different findings in neonates were perhaps caused by the influence of delivery on sampling or the specific CT manifestations for neonates, adding that more samples are needed for further clarification.
Based on these findings, “the CT imaging of COVID-19 infection should be differentiated with other virus pneumonias such as influenza virus, parainfluenza virus, respiratory syncytial virus, and adenovirus,” they concluded. It also should “be differentiated from bacterial pneumonia, mycoplasma pneumonia, and chlamydia pneumonia ... the density of pneumonia lesions caused by the latter pathogens is relatively higher.”
However, Dr. Xia and colleagues noted that chest CT manifestations of pneumonia caused by different pathogens overlap, and COVID-19 pneumonia “can be superimposed with serious and complex imaging manifestations, so epidemiological and etiological examinations should be combined.”
The investigators concluded that COVID-19 virus pneumonia in children is generally mild, and that the characteristic changes of subpleural ground-glass opacities and consolidations with surrounding halo on chest CT provide an “effective means for follow-up and evaluating the changes of lung lesions.”
“In the case that the positive rate of COVID-19 nucleic acid test from pharyngeal swab samples is not high, the early detection of lesions by CT is conducive to reasonable management and early treatment for pediatric patients. However, the diagnosis of COVID-19 pneumonia by CT imaging alone is not sufficient enough, especially in the case of coinfection with other pathogens,” Dr. Xia and associates wrote. “Therefore, early chest CT screening and timely follow-up, combined with corresponding pathogen detection, is a feasible clinical protocol in children.”
An early study
In a separate retrospective analysis described in a letter to the editor of the New England Journal of Medicine, Weiyong Liu, PhD, of Tongji Hospital of Huazhong University of Science and Technology and colleagues found that the most frequently detected pathogens in 366 children under the age of 16 years hospitalized with respiratory infections in Wuhan during Jan. 7-15, 2020, were influenza A virus (6.3% of cases) and influenza B virus (5.5% of cases), whereas COVID-19 was detected in 1.6% of cases.
The median age of the COVID-19 patients in that series was 3 years (range 1-7 years), and in contrast to the findings of Xia et al., all previously had been “completely healthy.” Common characteristics were high fever and cough in all six patients, and vomiting in four patients. Five had pneumonia as assessed by X-ray, and CTs showed typical viral pneumonia patterns.
One patient was admitted to a pediatric ICU. All patients received antiviral agents, antibiotic agents, and supportive therapies; all recovered after a median hospital stay of 7.5 days (median range, 5-13 days).
In contrast with the findings of Xia et al., the findings of Liu et al. showed COVID-19 caused moderate to severe respiratory illness in children, and that infections in children were occurring early in the epidemic.
Some perspective
In an interview regarding the findings by Xia et al., Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University, and director of pediatric infectious diseases at Boston Medical Center, noted the absence of fever in 40% of cases.
“This is important, as the criteria for testing by public health departments has been high fever, cough, and shortness of breath,” he said. “The absence of fever is not inconsistent with COVID-19 disease.”
Another important point regarding the findings by Xia et al. is that the highest attack rates appear to be in children under 1 year of age, he said, further noting that the finding of concurrent influenza A, influenza B, or respiratory syncytial virus underscores that “concurrent infection can occur, and the presence of another virus in diagnostic tests does not mean that COVID-19 is not causal.”
As for whether the finding of elevated procalcitonin levels in 80% of cases reflects COVID-19 disease or coinfection with bacteria, the answer is unclear. But none of the children in the study were proven to have bacterial disease, he said, adding that “this marker will need to be interpreted with caution in the setting of COVID-19 disease.”
Dr. Xia and colleagues reported having no disclosures. Dr. Liu and associates also reported having no disclosures. The study by Liu et al. was supported by the Ministry of Science and Technology of China, the National Mega Project on Major Infectious Disease Prevention, and the National Key Research and Development Program of China.
SOURCES: Xia W et al. Ped Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718; Liu W et al. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
Pediatric cases of COVID-19 infection are typically mild, but underlying coinfection may be more common in children than in adults, according to an analysis of clinical, laboratory, and chest CT features of pediatric inpatients in Wuhan, China.

The findings point toward a need for early chest CT with corresponding pathogen detection in children with suspected COVID-19 infection, Wei Xia, MD, of Huazhong University of Science and Technology, Wuhan, China, and colleagues reported in Pediatric Pulmonology.
The most common symptoms in 20 pediatric patients hospitalized between Jan. 23 and Feb. 8, 2020, with COVID-19 infection confirmed by the pharyngeal swab COVID-19 nucleic acid test were fever and cough, which occurred in 60% and 65% of patients, respectively. Coinfection was detected in eight patients (40%), they noted.
Clinical manifestations were similar to those seen in adults, but overall symptoms were relatively mild and overall prognosis was good. Of particular note, 7 of the 20 (35%) patients had a previously diagnosed congenital or acquired diseases, suggesting that children with underlying conditions may be more susceptible, Dr. Xia and colleagues wrote.
Laboratory findings also were notable in that 80% of the children had procalcitonin (PCT) elevations not typically seen in adults with COVID-19. PCT is a marker for bacterial infection and “[this finding] may suggest that routine antibacterial treatment should be considered in pediatric patients,” the investigators wrote.
As for imaging results, chest CT findings in children were similar to those in adults.“The typical manifestations were unilateral or bilateral subpleural ground-glass opacities, and consolidations with surrounding halo signs,” Dr. Xia and associates wrote, adding that consolidations with surrounding halo sign accounted for about half the pediatric cases and should be considered as “typical signs in pediatric patients.”
Pediatric cases were “rather rare” in the early days of the COVID-19 outbreak in Wuhan, where the first cases of infection were reported.
“As a pediatric group is usually susceptible to upper respiratory tract infection, because of their developing immune system, the delayed presence of pediatric patients is confusing,” the investigators wrote, noting that a low detection rate of pharyngeal swab COVID-19 nucleic acid test, distinguishing the virus from other common respiratory tract infectious pathogens in pediatric patients, “is still a problem.”
To better characterize the clinical and imaging features in children versus adults with COVID-19, Dr. Xia and associates reviewed these 20 pediatric cases, including 13 boys and 7 girls with ages ranging from less than 1 month to 14 years, 7 months (median 2 years, 1.5 months). Thirteen had an identified close contact with a COVID-19–diagnosed family member, and all were treated in an isolation ward. A total of 18 children were cured and discharged after an average stay of 13 days, and 2 neonates remained under observation because of positive swab results with negative CT findings. The investigators speculated that the different findings in neonates were perhaps caused by the influence of delivery on sampling or the specific CT manifestations for neonates, adding that more samples are needed for further clarification.
Based on these findings, “the CT imaging of COVID-19 infection should be differentiated with other virus pneumonias such as influenza virus, parainfluenza virus, respiratory syncytial virus, and adenovirus,” they concluded. It also should “be differentiated from bacterial pneumonia, mycoplasma pneumonia, and chlamydia pneumonia ... the density of pneumonia lesions caused by the latter pathogens is relatively higher.”
However, Dr. Xia and colleagues noted that chest CT manifestations of pneumonia caused by different pathogens overlap, and COVID-19 pneumonia “can be superimposed with serious and complex imaging manifestations, so epidemiological and etiological examinations should be combined.”
The investigators concluded that COVID-19 virus pneumonia in children is generally mild, and that the characteristic changes of subpleural ground-glass opacities and consolidations with surrounding halo on chest CT provide an “effective means for follow-up and evaluating the changes of lung lesions.”
“In the case that the positive rate of COVID-19 nucleic acid test from pharyngeal swab samples is not high, the early detection of lesions by CT is conducive to reasonable management and early treatment for pediatric patients. However, the diagnosis of COVID-19 pneumonia by CT imaging alone is not sufficient enough, especially in the case of coinfection with other pathogens,” Dr. Xia and associates wrote. “Therefore, early chest CT screening and timely follow-up, combined with corresponding pathogen detection, is a feasible clinical protocol in children.”
An early study
In a separate retrospective analysis described in a letter to the editor of the New England Journal of Medicine, Weiyong Liu, PhD, of Tongji Hospital of Huazhong University of Science and Technology and colleagues found that the most frequently detected pathogens in 366 children under the age of 16 years hospitalized with respiratory infections in Wuhan during Jan. 7-15, 2020, were influenza A virus (6.3% of cases) and influenza B virus (5.5% of cases), whereas COVID-19 was detected in 1.6% of cases.
The median age of the COVID-19 patients in that series was 3 years (range 1-7 years), and in contrast to the findings of Xia et al., all previously had been “completely healthy.” Common characteristics were high fever and cough in all six patients, and vomiting in four patients. Five had pneumonia as assessed by X-ray, and CTs showed typical viral pneumonia patterns.
One patient was admitted to a pediatric ICU. All patients received antiviral agents, antibiotic agents, and supportive therapies; all recovered after a median hospital stay of 7.5 days (median range, 5-13 days).
In contrast with the findings of Xia et al., the findings of Liu et al. showed COVID-19 caused moderate to severe respiratory illness in children, and that infections in children were occurring early in the epidemic.
Some perspective
In an interview regarding the findings by Xia et al., Stephen I. Pelton, MD, professor of pediatrics and epidemiology at Boston University, and director of pediatric infectious diseases at Boston Medical Center, noted the absence of fever in 40% of cases.
“This is important, as the criteria for testing by public health departments has been high fever, cough, and shortness of breath,” he said. “The absence of fever is not inconsistent with COVID-19 disease.”
Another important point regarding the findings by Xia et al. is that the highest attack rates appear to be in children under 1 year of age, he said, further noting that the finding of concurrent influenza A, influenza B, or respiratory syncytial virus underscores that “concurrent infection can occur, and the presence of another virus in diagnostic tests does not mean that COVID-19 is not causal.”
As for whether the finding of elevated procalcitonin levels in 80% of cases reflects COVID-19 disease or coinfection with bacteria, the answer is unclear. But none of the children in the study were proven to have bacterial disease, he said, adding that “this marker will need to be interpreted with caution in the setting of COVID-19 disease.”
Dr. Xia and colleagues reported having no disclosures. Dr. Liu and associates also reported having no disclosures. The study by Liu et al. was supported by the Ministry of Science and Technology of China, the National Mega Project on Major Infectious Disease Prevention, and the National Key Research and Development Program of China.
SOURCES: Xia W et al. Ped Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718; Liu W et al. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
FROM PEDIATRIC PULMONOLOGY
COVID-19: Older patients with cancer especially vulnerable
For oncologists and other clinicians caring for patients with cancer, the COVID-19 pandemic represents a dynamic clinical challenge that is changing daily and that can feel overwhelming at times, say experts.
“Oncology clinicians are well versed in caring for immunosuppressed patients with cancer, of all ages,” Merry-Jennifer Markham, MD, interim chief of the Division of Hematology and Oncology at the University of Florida Health, Gainesville, told Medscape Medical News.
However, she emphasized that, during this COVID-19 outbreak, “we must be especially diligent about screening for symptoms and exposure, and we must recognize that our older patients with cancer may be especially vulnerable.”
Patients with cancer who are in active treatment are immunosuppressed and are more susceptible to infection and to complications from infection, Markham pointed out. “While we don’t yet have much data on how COVID-19 impacts patients with cancer, I have to suspect that patients undergoing active cancer treatment may be especially vulnerable to the more severe illness associated with COVID-19,” she said.
Indeed, a recent report from China that was published in the Lancet Oncology supports this. The authors suggest that patients with cancer are at higher risk for COVID-19 and have a worse prognosis if they become infected than do those without cancer.
Commonsense rules
Commonsense rules apply for all patients with cancer, regardless of age, said Markham. Measures include thorough handwashing, staying home when sick, and avoiding sick contacts.
Markham, who acts as an expert spokesperson for the American Society of Clinical Oncology, provides information on what patients with cancer need to know about COVID-19 at Cancer.net, the society’s website for patients with cancer.
“Unfortunately, this outbreak of COVID-19 is happening rapidly and in real time,” Markham noted. “The entire medical community is learning as we go, rather than having the luxury of years of evidence-based literature to guide us.”
Another expert agrees. “Unfortunately, there are not a lot of data on how COVID-19 affects cancer patients,” Cardinale Smith, MD, PhD, director of Quality for Cancer Services in the Mount Sinai Health System, New York City, said in an interview.
“We need to minimize the risk for patients and minimize our own exposure by treating this situation like we would a really bad flu season,” Smith told Medscape Medical News. “Some patients have had a bad outcome, but the vast majority do not. The best we can do is stay calm and focused.”
At Mount Sinai, for patients with cancer, routine, nonurgent appointments are being rescheduled for May, Smith said. Those in active treatment are screened by telephone 24 to 48 hours before arrival, after which they undergo a full risk assessment in an isolation room. Those with a respiratory infection are given a mask.
“Patients are very anxious and worried about COVID-19,” said Smith, who has young children and an elderly parent at home. “We don’t have all the answers, and this can heighten anxiety.”
To help allay fears, social workers are asking patients with cancer who express anxiety to discuss their concerns and provide information. A one-page handout on both flu and COVID-10 is available in the waiting room.
The Web portal MyChart gives patients access to updated information on COVID-19 precautions and provides links to the hospital website and to the US Centers for Disease Control and Prevention. Patients who are not feeling well can speak to someone or get answers if they have additional questions.
When counseling patients, Smith advises them to use “an abundance of caution” and to be creative in efforts to minimize risk. “My suggestion is to use FaceTime and Skype to connect and communicate with your community,” she said.
Some churches are conducting services via teleconferencing to minimize risk, and seniors’ centers that offer yoga and other classes are also beginning to provide services virtually, she pointed out.
Data from China
A report published February 14 in the Lancet Oncology appears to be the first analysis in the literature to focus on COVID-19 in patients with cancer.
“Patients with cancer are more susceptible to infection than individuals without cancer because of their systemic immunosuppressive state caused by the malignancy and anticancer treatments, such as chemotherapy or surgery,” write the authors, led by Wenhua Liang, MD, of Guangzhou Medical University. However, in correspondence published in the Lancet Oncology, other experts in China question some of Liang’s and colleagues’ findings.
The report by Liang and colleagues concerns a prospective cohort of 1590 patients with COVID-19.
There were 2007 laboratory-confirmed cases of COVID-19 among patients admitted to 575 hospitals throughout China as of January 31. Of those cases, 417 were excluded from the analysis because of insufficient information regarding disease history.
The team reports that of 18 patients with cancer and COVID-19, 39% were at significantly higher risk for “severe events.” By comparison, of 1572 patients with COVID-19 who did not have cancer, 8% were at significantly higher risk (P = .0003). These events included rapid clinical deterioration that required admission to intensive care; invasive ventilation; or death.
Patients with cancer experienced a much more rapid deterioration in clinical status than did those without cancer. The median time to severe events was 13 days, vs 43 days (hazard ratio [HR] adjusted for age, 3.56; P < .0001).
The analysis also shows that patients who underwent chemotherapy or surgery in the past month had a 75% risk of experiencing clinically severe events, compared with a 43% risk for those who had not received recent treatment.
After adjusting for other risk factors, including age and smoking history, older age was the only risk factor for severe events (odds ratio [OR], 1.43; 95% confidence interval [CI], 0.97 – 2.12; P = .072), the study authors say.
Patients with lung cancer did not have a higher probability of severe events compared with patients with other cancer types (20% vs 62%, respectively; P = .294).
Liang and colleagues conclude that these findings provide “a timely reminder to physicians that more intensive attention should be paid to patients with cancer, in case of rapid deterioration.”
The team also proposes three strategies for managing patients with cancer who are at risk for COVID-19 or any other severe infectious disease. They recommend that intentional postponement of adjuvant chemotherapy or elective surgery be considered for patients with stable cancer who live in areas where disease is endemic. Stronger “personal protection provisions” could also be made for patients with cancer or for cancer survivors. Lastly, for patients with cancer who have COVID-19, especially those who are older or who have comorbidities, more intensive surveillance or treatment should be considered.
However, in comments in the Lancet Oncology, other authors in China say these findings should be interpreted with caution.
One group suggests that the increased susceptibility to COVID-19 in patients with cancer could be the result of higher rates of smoking compared with patients who did not have cancer. “Overall, current evidence remains insufficient to explain a conclusive association between cancer and COVID-19,” say Huahao Shen, PhD, of Zhejiang University School of Medicine, Hangzhou, Zhejiang, and colleagues.
Another group suggests that the significantly higher median age of patients with cancer compared with noncancer patients (63 years vs 49 years) may have contributed to poor prognosis.
These authors, led by Li Zhang, MD, PhD, and Hanping Wang, MD, of Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, emphasize that patients with cancer need online medical counseling and that critical cases need to be identified and treated.
“In endemic areas outside Wuhan, decisions on whether or not to postpone cancer treatment need to made on a patient-by-patient basis and according to the risk to the patient and the prevailing situation because delays could lead to tumor progression and ultimately poorer outcomes,” they write.
The study was funded by the China National Science Foundation and the Key Project of Guangzhou Scientific Research Project. Liang and coauthors, Shen and coauthors, Zhang, Wang, and Smith have disclosed no relevant financial relationships. Markham has relationships with Aduro Biotech, Lilly, Tesaro, Novartis, and VBL Therapeutics.
This article first appeared on Medscape.com.
For oncologists and other clinicians caring for patients with cancer, the COVID-19 pandemic represents a dynamic clinical challenge that is changing daily and that can feel overwhelming at times, say experts.
“Oncology clinicians are well versed in caring for immunosuppressed patients with cancer, of all ages,” Merry-Jennifer Markham, MD, interim chief of the Division of Hematology and Oncology at the University of Florida Health, Gainesville, told Medscape Medical News.
However, she emphasized that, during this COVID-19 outbreak, “we must be especially diligent about screening for symptoms and exposure, and we must recognize that our older patients with cancer may be especially vulnerable.”
Patients with cancer who are in active treatment are immunosuppressed and are more susceptible to infection and to complications from infection, Markham pointed out. “While we don’t yet have much data on how COVID-19 impacts patients with cancer, I have to suspect that patients undergoing active cancer treatment may be especially vulnerable to the more severe illness associated with COVID-19,” she said.
Indeed, a recent report from China that was published in the Lancet Oncology supports this. The authors suggest that patients with cancer are at higher risk for COVID-19 and have a worse prognosis if they become infected than do those without cancer.
Commonsense rules
Commonsense rules apply for all patients with cancer, regardless of age, said Markham. Measures include thorough handwashing, staying home when sick, and avoiding sick contacts.
Markham, who acts as an expert spokesperson for the American Society of Clinical Oncology, provides information on what patients with cancer need to know about COVID-19 at Cancer.net, the society’s website for patients with cancer.
“Unfortunately, this outbreak of COVID-19 is happening rapidly and in real time,” Markham noted. “The entire medical community is learning as we go, rather than having the luxury of years of evidence-based literature to guide us.”
Another expert agrees. “Unfortunately, there are not a lot of data on how COVID-19 affects cancer patients,” Cardinale Smith, MD, PhD, director of Quality for Cancer Services in the Mount Sinai Health System, New York City, said in an interview.
“We need to minimize the risk for patients and minimize our own exposure by treating this situation like we would a really bad flu season,” Smith told Medscape Medical News. “Some patients have had a bad outcome, but the vast majority do not. The best we can do is stay calm and focused.”
At Mount Sinai, for patients with cancer, routine, nonurgent appointments are being rescheduled for May, Smith said. Those in active treatment are screened by telephone 24 to 48 hours before arrival, after which they undergo a full risk assessment in an isolation room. Those with a respiratory infection are given a mask.
“Patients are very anxious and worried about COVID-19,” said Smith, who has young children and an elderly parent at home. “We don’t have all the answers, and this can heighten anxiety.”
To help allay fears, social workers are asking patients with cancer who express anxiety to discuss their concerns and provide information. A one-page handout on both flu and COVID-10 is available in the waiting room.
The Web portal MyChart gives patients access to updated information on COVID-19 precautions and provides links to the hospital website and to the US Centers for Disease Control and Prevention. Patients who are not feeling well can speak to someone or get answers if they have additional questions.
When counseling patients, Smith advises them to use “an abundance of caution” and to be creative in efforts to minimize risk. “My suggestion is to use FaceTime and Skype to connect and communicate with your community,” she said.
Some churches are conducting services via teleconferencing to minimize risk, and seniors’ centers that offer yoga and other classes are also beginning to provide services virtually, she pointed out.
Data from China
A report published February 14 in the Lancet Oncology appears to be the first analysis in the literature to focus on COVID-19 in patients with cancer.
“Patients with cancer are more susceptible to infection than individuals without cancer because of their systemic immunosuppressive state caused by the malignancy and anticancer treatments, such as chemotherapy or surgery,” write the authors, led by Wenhua Liang, MD, of Guangzhou Medical University. However, in correspondence published in the Lancet Oncology, other experts in China question some of Liang’s and colleagues’ findings.
The report by Liang and colleagues concerns a prospective cohort of 1590 patients with COVID-19.
There were 2007 laboratory-confirmed cases of COVID-19 among patients admitted to 575 hospitals throughout China as of January 31. Of those cases, 417 were excluded from the analysis because of insufficient information regarding disease history.
The team reports that of 18 patients with cancer and COVID-19, 39% were at significantly higher risk for “severe events.” By comparison, of 1572 patients with COVID-19 who did not have cancer, 8% were at significantly higher risk (P = .0003). These events included rapid clinical deterioration that required admission to intensive care; invasive ventilation; or death.
Patients with cancer experienced a much more rapid deterioration in clinical status than did those without cancer. The median time to severe events was 13 days, vs 43 days (hazard ratio [HR] adjusted for age, 3.56; P < .0001).
The analysis also shows that patients who underwent chemotherapy or surgery in the past month had a 75% risk of experiencing clinically severe events, compared with a 43% risk for those who had not received recent treatment.
After adjusting for other risk factors, including age and smoking history, older age was the only risk factor for severe events (odds ratio [OR], 1.43; 95% confidence interval [CI], 0.97 – 2.12; P = .072), the study authors say.
Patients with lung cancer did not have a higher probability of severe events compared with patients with other cancer types (20% vs 62%, respectively; P = .294).
Liang and colleagues conclude that these findings provide “a timely reminder to physicians that more intensive attention should be paid to patients with cancer, in case of rapid deterioration.”
The team also proposes three strategies for managing patients with cancer who are at risk for COVID-19 or any other severe infectious disease. They recommend that intentional postponement of adjuvant chemotherapy or elective surgery be considered for patients with stable cancer who live in areas where disease is endemic. Stronger “personal protection provisions” could also be made for patients with cancer or for cancer survivors. Lastly, for patients with cancer who have COVID-19, especially those who are older or who have comorbidities, more intensive surveillance or treatment should be considered.
However, in comments in the Lancet Oncology, other authors in China say these findings should be interpreted with caution.
One group suggests that the increased susceptibility to COVID-19 in patients with cancer could be the result of higher rates of smoking compared with patients who did not have cancer. “Overall, current evidence remains insufficient to explain a conclusive association between cancer and COVID-19,” say Huahao Shen, PhD, of Zhejiang University School of Medicine, Hangzhou, Zhejiang, and colleagues.
Another group suggests that the significantly higher median age of patients with cancer compared with noncancer patients (63 years vs 49 years) may have contributed to poor prognosis.
These authors, led by Li Zhang, MD, PhD, and Hanping Wang, MD, of Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, emphasize that patients with cancer need online medical counseling and that critical cases need to be identified and treated.
“In endemic areas outside Wuhan, decisions on whether or not to postpone cancer treatment need to made on a patient-by-patient basis and according to the risk to the patient and the prevailing situation because delays could lead to tumor progression and ultimately poorer outcomes,” they write.
The study was funded by the China National Science Foundation and the Key Project of Guangzhou Scientific Research Project. Liang and coauthors, Shen and coauthors, Zhang, Wang, and Smith have disclosed no relevant financial relationships. Markham has relationships with Aduro Biotech, Lilly, Tesaro, Novartis, and VBL Therapeutics.
This article first appeared on Medscape.com.
For oncologists and other clinicians caring for patients with cancer, the COVID-19 pandemic represents a dynamic clinical challenge that is changing daily and that can feel overwhelming at times, say experts.
“Oncology clinicians are well versed in caring for immunosuppressed patients with cancer, of all ages,” Merry-Jennifer Markham, MD, interim chief of the Division of Hematology and Oncology at the University of Florida Health, Gainesville, told Medscape Medical News.
However, she emphasized that, during this COVID-19 outbreak, “we must be especially diligent about screening for symptoms and exposure, and we must recognize that our older patients with cancer may be especially vulnerable.”
Patients with cancer who are in active treatment are immunosuppressed and are more susceptible to infection and to complications from infection, Markham pointed out. “While we don’t yet have much data on how COVID-19 impacts patients with cancer, I have to suspect that patients undergoing active cancer treatment may be especially vulnerable to the more severe illness associated with COVID-19,” she said.
Indeed, a recent report from China that was published in the Lancet Oncology supports this. The authors suggest that patients with cancer are at higher risk for COVID-19 and have a worse prognosis if they become infected than do those without cancer.
Commonsense rules
Commonsense rules apply for all patients with cancer, regardless of age, said Markham. Measures include thorough handwashing, staying home when sick, and avoiding sick contacts.
Markham, who acts as an expert spokesperson for the American Society of Clinical Oncology, provides information on what patients with cancer need to know about COVID-19 at Cancer.net, the society’s website for patients with cancer.
“Unfortunately, this outbreak of COVID-19 is happening rapidly and in real time,” Markham noted. “The entire medical community is learning as we go, rather than having the luxury of years of evidence-based literature to guide us.”
Another expert agrees. “Unfortunately, there are not a lot of data on how COVID-19 affects cancer patients,” Cardinale Smith, MD, PhD, director of Quality for Cancer Services in the Mount Sinai Health System, New York City, said in an interview.
“We need to minimize the risk for patients and minimize our own exposure by treating this situation like we would a really bad flu season,” Smith told Medscape Medical News. “Some patients have had a bad outcome, but the vast majority do not. The best we can do is stay calm and focused.”
At Mount Sinai, for patients with cancer, routine, nonurgent appointments are being rescheduled for May, Smith said. Those in active treatment are screened by telephone 24 to 48 hours before arrival, after which they undergo a full risk assessment in an isolation room. Those with a respiratory infection are given a mask.
“Patients are very anxious and worried about COVID-19,” said Smith, who has young children and an elderly parent at home. “We don’t have all the answers, and this can heighten anxiety.”
To help allay fears, social workers are asking patients with cancer who express anxiety to discuss their concerns and provide information. A one-page handout on both flu and COVID-10 is available in the waiting room.
The Web portal MyChart gives patients access to updated information on COVID-19 precautions and provides links to the hospital website and to the US Centers for Disease Control and Prevention. Patients who are not feeling well can speak to someone or get answers if they have additional questions.
When counseling patients, Smith advises them to use “an abundance of caution” and to be creative in efforts to minimize risk. “My suggestion is to use FaceTime and Skype to connect and communicate with your community,” she said.
Some churches are conducting services via teleconferencing to minimize risk, and seniors’ centers that offer yoga and other classes are also beginning to provide services virtually, she pointed out.
Data from China
A report published February 14 in the Lancet Oncology appears to be the first analysis in the literature to focus on COVID-19 in patients with cancer.
“Patients with cancer are more susceptible to infection than individuals without cancer because of their systemic immunosuppressive state caused by the malignancy and anticancer treatments, such as chemotherapy or surgery,” write the authors, led by Wenhua Liang, MD, of Guangzhou Medical University. However, in correspondence published in the Lancet Oncology, other experts in China question some of Liang’s and colleagues’ findings.
The report by Liang and colleagues concerns a prospective cohort of 1590 patients with COVID-19.
There were 2007 laboratory-confirmed cases of COVID-19 among patients admitted to 575 hospitals throughout China as of January 31. Of those cases, 417 were excluded from the analysis because of insufficient information regarding disease history.
The team reports that of 18 patients with cancer and COVID-19, 39% were at significantly higher risk for “severe events.” By comparison, of 1572 patients with COVID-19 who did not have cancer, 8% were at significantly higher risk (P = .0003). These events included rapid clinical deterioration that required admission to intensive care; invasive ventilation; or death.
Patients with cancer experienced a much more rapid deterioration in clinical status than did those without cancer. The median time to severe events was 13 days, vs 43 days (hazard ratio [HR] adjusted for age, 3.56; P < .0001).
The analysis also shows that patients who underwent chemotherapy or surgery in the past month had a 75% risk of experiencing clinically severe events, compared with a 43% risk for those who had not received recent treatment.
After adjusting for other risk factors, including age and smoking history, older age was the only risk factor for severe events (odds ratio [OR], 1.43; 95% confidence interval [CI], 0.97 – 2.12; P = .072), the study authors say.
Patients with lung cancer did not have a higher probability of severe events compared with patients with other cancer types (20% vs 62%, respectively; P = .294).
Liang and colleagues conclude that these findings provide “a timely reminder to physicians that more intensive attention should be paid to patients with cancer, in case of rapid deterioration.”
The team also proposes three strategies for managing patients with cancer who are at risk for COVID-19 or any other severe infectious disease. They recommend that intentional postponement of adjuvant chemotherapy or elective surgery be considered for patients with stable cancer who live in areas where disease is endemic. Stronger “personal protection provisions” could also be made for patients with cancer or for cancer survivors. Lastly, for patients with cancer who have COVID-19, especially those who are older or who have comorbidities, more intensive surveillance or treatment should be considered.
However, in comments in the Lancet Oncology, other authors in China say these findings should be interpreted with caution.
One group suggests that the increased susceptibility to COVID-19 in patients with cancer could be the result of higher rates of smoking compared with patients who did not have cancer. “Overall, current evidence remains insufficient to explain a conclusive association between cancer and COVID-19,” say Huahao Shen, PhD, of Zhejiang University School of Medicine, Hangzhou, Zhejiang, and colleagues.
Another group suggests that the significantly higher median age of patients with cancer compared with noncancer patients (63 years vs 49 years) may have contributed to poor prognosis.
These authors, led by Li Zhang, MD, PhD, and Hanping Wang, MD, of Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, emphasize that patients with cancer need online medical counseling and that critical cases need to be identified and treated.
“In endemic areas outside Wuhan, decisions on whether or not to postpone cancer treatment need to made on a patient-by-patient basis and according to the risk to the patient and the prevailing situation because delays could lead to tumor progression and ultimately poorer outcomes,” they write.
The study was funded by the China National Science Foundation and the Key Project of Guangzhou Scientific Research Project. Liang and coauthors, Shen and coauthors, Zhang, Wang, and Smith have disclosed no relevant financial relationships. Markham has relationships with Aduro Biotech, Lilly, Tesaro, Novartis, and VBL Therapeutics.
This article first appeared on Medscape.com.
Health professionals fight against COVID-19 myths and misinformation
Misinformation about the COVID-19 travels faster than the virus and complicates the job of doctors who are treating those infected and responding to concerns of their other patients.
An array of myths springing up around this disease can be found on the Internet. The main themes appear to be false narratives about the origin of the virus, the size of the outbreak in the United States and in other countries, the availability of cures and treatments, and ways to prevent infection. Widespread misinformation hampers public health efforts to control the disease outbreak, confuses the public, and requires medical professionals to spend time refuting myths and re-educating patients.
A group of infectious disease experts became so alarmed by the misinformation trend they published a statement in The Lancet decrying the spread of false statements being circulated by some media outlets. “The rapid, open, and transparent sharing of data on this outbreak is now being threatened by rumours and misinformation ... Conspiracy theories do nothing but create fear, rumours, and prejudice that jeopardise our global collaboration in the fight against this virus,” wrote Charles H. Calisher, PhD, of Colorado State University, Fort Collins, and colleagues.
What can physicians do to counter misinformation?
Pulmonologist and critical care physician Cedric “Jamie” Rutland, MD, who practices in Riverside, Calif., sees misinformation about the novel coronavirus every day at home and on the job. His patients worry that everyone who gets infected will die or end up in the ICU. His neighbors ask him to pilfer surgical masks to protect them from the false notion that Chinese people in their community posed some kind of COVID-19 risk.
As he pondered how to counter myths with facts, Dr. Rutland turned to an unusual resource: His 7-year-old daughter Amelia. He explained to her how COVID-19 works and found that she could easily understand the basics. Now, Dr. Rutland draws upon the lessons from chats with his daughter as he explains COVID-19 to his patient audience on his YouTube channel “Medicine Deconstructed.” Simplicity, but not too much simplicity, is key, he said. Dr. Rutland uses a visual aid – a rough drawing of a virus – and shows how inflammation and antibodies enter the picture after infection. “I just teach them that if you’re a healthy person, this is how the body works, and this is what the immune system will do,” he said. “For the most part, you can calm people down when you make time for education.”
What are best practices? In a series of interviews, specialists emphasized the importance of fact-finding, wide-ranging communication, and – perhaps most difficult of all – humility.
Dr. Rutland emphasizes thoughtful communication based on facts and humility when communicating to patients about this potential health risk. “A lot of people finish medical school and think, ‘Everyone should trust me because I’m the pulmonologist or the GI doc.’ That’s not how it works. You still have to earn people’s trust,” he said.
Make sure all staff get reliable information
Hospitals are scrambling to keep staff safe with up-to-date directives and debunk false narratives about the virus. Keeping all hospital staff informed with verified and authoritative facts about the coronavirus is a key objective of the Massachusetts General Hospital’s Center for Disaster Medicine. The Center’s coronavirus educational materials are distributed to all staffers from physicians to janitors. “These provide information that they need to understand the risks and keep themselves safe,” said Eileen Searle, PhD, the Biothreats Clinical Operations program manager in the CDM.
According to Dr. Searle, the hospital keeps a continually updated COVID-19 Frequently Asked Questions document in its internal computer system. All employees can access it, she said, and it’s updated to include questions as they come up.
Even valets and front-desk volunteers are encouraged to read the FAQ, she said, since “they’re the first people that family and patients are interacting with.” The document “gives them reassurance about delivering messages,” she said.
Use patience with your patients
Dr. Rutland urges colleagues to take the time to listen to patients and educate them. “Reduce the gap between you and them,” said Dr. Rutland, who treats patients in Orange and Riverside counties. “Take off your white coat, sit down, and talk to the person about their concerns.”
Boston cardiologist Haider Warraich, MD, of Brigham and Women’s Hospital, Boston, said it’s important to “put medical information into a greater human context.” For example, he has told patients that he’s still taking his daughter to school despite COVID-19 risks. “I take the information I provide and apply it to my own life,” he said.
The Washington State Department of Health offers this advice to physicians to counter false information and stigma: “Stay updated and informed on COVID-19 to avoid miscommunication or inaccurate information. Talk openly about the harm of stigma. View people directly impacted by stigma as people first. Be conscious of your language. Acknowledge access and language barriers.”
Speak out on social media – but don’t fight
Should medical professionals speak out about COVID-19 misinformation via social media? It’s an individual decision, Dr. Warraich said, “but my sense is that it’s never been more important for physicians to be part of the fray and help quell the epidemic of misinformation that almost always follows any type of medial calamity.”
Dr. Rutland, vice president and founding member of the Association for Healthcare Social Media, cautioned that effective communication via social media requires care. Avoid confrontation, he advised. “Don’t call people stupid or say things like, ‘I went to medical school and I’m smarter than you.’ ”
Instead, he said, “it’s important to just state the facts: These are the people who are dying, these are the people who are getting infected.”
And, he added, remember to push the most important message of all: Wash your hands!
Public health organizations fight the ‘infodemic’
In a trend that hearkens back to the days of snake oil cures for all maladies, advertisements for fake treatments are popping up on the Internet and on other media.
Facebook and Amazon have acted to remove these ads but these messages continue to flood social media such as Twitter, WhatsApp, and other sites. Discussion groups on platforms such as Reddit continue to pump out misinformation about COVID-19. Conspiracy theories that link the virus to espionage and bioweapons are making the rounds on the Internet and talk radio. Wrong information about the effectiveness of non-N95 face masks to protect wearers against infection is widespread, leading to shortages for medical personnel and price gouging. Pernicious rumors about the effectiveness of substances such a vinegar, silver, garlic, lemon juice, and even vodka to disinfect hands and surfaces abound on the Internet. An especially dangerous stream of misinformation stigmatizes ethnic groups and individuals as sources of the infection.
The World Health Organization identified early in the COVID-19 outbreak the global wave of misinformation about the virus and dubbed the problem the “infodemic.” The WHO “Q & A” page on COVID-19 is updated frequently and addresses myths and rumors currently circulating.
According to the WHO website, the agency has reached out to social media players such as Facebook, Twitter, Instagram, LinkedIn, Pinterest, TikTok, and Weibo, the microblogging site in China. WHO has worked with these sites to curb the “infodemic” of misinformation and has used these sites for public education outreach on COVID-19. “Myth busting” infographics posted on a WHO web page are also reposted on major social media sites.
The CDC has followed with its own “frequently asked questions” page to address questions and rumors. State health agencies have put up COVID-19 pages to address public concerns and offer advice on prevention. The Maryland Department of Health web page directly addresses dangerous misinformation: “Do not stigmatize people of any specific ethnicities or racial background. Viruses do not target people from specific populations, ethnicities or racial backgrounds. Stay informed and seek information from reliable, official sources. Be wary of myths, rumors and misinformation circulating online and elsewhere. Health information shared through social media is frequently inaccurate, unless coming from an official, reliable source such as the CDC, MDH or local health departments.”
The Washington State Department of Health has taken a more assertive stance on stigma. The COVID-19 web page recommends to the public: “Show compassion and support for individuals and communities more closely impacted. Avoid stigmatizing people who are in quarantine. They are making the right choice for their communities. Do not make assumptions about someone’s health status based on their ethnicity, race or national origin.”
Misinformation about the COVID-19 travels faster than the virus and complicates the job of doctors who are treating those infected and responding to concerns of their other patients.
An array of myths springing up around this disease can be found on the Internet. The main themes appear to be false narratives about the origin of the virus, the size of the outbreak in the United States and in other countries, the availability of cures and treatments, and ways to prevent infection. Widespread misinformation hampers public health efforts to control the disease outbreak, confuses the public, and requires medical professionals to spend time refuting myths and re-educating patients.
A group of infectious disease experts became so alarmed by the misinformation trend they published a statement in The Lancet decrying the spread of false statements being circulated by some media outlets. “The rapid, open, and transparent sharing of data on this outbreak is now being threatened by rumours and misinformation ... Conspiracy theories do nothing but create fear, rumours, and prejudice that jeopardise our global collaboration in the fight against this virus,” wrote Charles H. Calisher, PhD, of Colorado State University, Fort Collins, and colleagues.
What can physicians do to counter misinformation?
Pulmonologist and critical care physician Cedric “Jamie” Rutland, MD, who practices in Riverside, Calif., sees misinformation about the novel coronavirus every day at home and on the job. His patients worry that everyone who gets infected will die or end up in the ICU. His neighbors ask him to pilfer surgical masks to protect them from the false notion that Chinese people in their community posed some kind of COVID-19 risk.
As he pondered how to counter myths with facts, Dr. Rutland turned to an unusual resource: His 7-year-old daughter Amelia. He explained to her how COVID-19 works and found that she could easily understand the basics. Now, Dr. Rutland draws upon the lessons from chats with his daughter as he explains COVID-19 to his patient audience on his YouTube channel “Medicine Deconstructed.” Simplicity, but not too much simplicity, is key, he said. Dr. Rutland uses a visual aid – a rough drawing of a virus – and shows how inflammation and antibodies enter the picture after infection. “I just teach them that if you’re a healthy person, this is how the body works, and this is what the immune system will do,” he said. “For the most part, you can calm people down when you make time for education.”
What are best practices? In a series of interviews, specialists emphasized the importance of fact-finding, wide-ranging communication, and – perhaps most difficult of all – humility.
Dr. Rutland emphasizes thoughtful communication based on facts and humility when communicating to patients about this potential health risk. “A lot of people finish medical school and think, ‘Everyone should trust me because I’m the pulmonologist or the GI doc.’ That’s not how it works. You still have to earn people’s trust,” he said.
Make sure all staff get reliable information
Hospitals are scrambling to keep staff safe with up-to-date directives and debunk false narratives about the virus. Keeping all hospital staff informed with verified and authoritative facts about the coronavirus is a key objective of the Massachusetts General Hospital’s Center for Disaster Medicine. The Center’s coronavirus educational materials are distributed to all staffers from physicians to janitors. “These provide information that they need to understand the risks and keep themselves safe,” said Eileen Searle, PhD, the Biothreats Clinical Operations program manager in the CDM.
According to Dr. Searle, the hospital keeps a continually updated COVID-19 Frequently Asked Questions document in its internal computer system. All employees can access it, she said, and it’s updated to include questions as they come up.
Even valets and front-desk volunteers are encouraged to read the FAQ, she said, since “they’re the first people that family and patients are interacting with.” The document “gives them reassurance about delivering messages,” she said.
Use patience with your patients
Dr. Rutland urges colleagues to take the time to listen to patients and educate them. “Reduce the gap between you and them,” said Dr. Rutland, who treats patients in Orange and Riverside counties. “Take off your white coat, sit down, and talk to the person about their concerns.”
Boston cardiologist Haider Warraich, MD, of Brigham and Women’s Hospital, Boston, said it’s important to “put medical information into a greater human context.” For example, he has told patients that he’s still taking his daughter to school despite COVID-19 risks. “I take the information I provide and apply it to my own life,” he said.
The Washington State Department of Health offers this advice to physicians to counter false information and stigma: “Stay updated and informed on COVID-19 to avoid miscommunication or inaccurate information. Talk openly about the harm of stigma. View people directly impacted by stigma as people first. Be conscious of your language. Acknowledge access and language barriers.”
Speak out on social media – but don’t fight
Should medical professionals speak out about COVID-19 misinformation via social media? It’s an individual decision, Dr. Warraich said, “but my sense is that it’s never been more important for physicians to be part of the fray and help quell the epidemic of misinformation that almost always follows any type of medial calamity.”
Dr. Rutland, vice president and founding member of the Association for Healthcare Social Media, cautioned that effective communication via social media requires care. Avoid confrontation, he advised. “Don’t call people stupid or say things like, ‘I went to medical school and I’m smarter than you.’ ”
Instead, he said, “it’s important to just state the facts: These are the people who are dying, these are the people who are getting infected.”
And, he added, remember to push the most important message of all: Wash your hands!
Public health organizations fight the ‘infodemic’
In a trend that hearkens back to the days of snake oil cures for all maladies, advertisements for fake treatments are popping up on the Internet and on other media.
Facebook and Amazon have acted to remove these ads but these messages continue to flood social media such as Twitter, WhatsApp, and other sites. Discussion groups on platforms such as Reddit continue to pump out misinformation about COVID-19. Conspiracy theories that link the virus to espionage and bioweapons are making the rounds on the Internet and talk radio. Wrong information about the effectiveness of non-N95 face masks to protect wearers against infection is widespread, leading to shortages for medical personnel and price gouging. Pernicious rumors about the effectiveness of substances such a vinegar, silver, garlic, lemon juice, and even vodka to disinfect hands and surfaces abound on the Internet. An especially dangerous stream of misinformation stigmatizes ethnic groups and individuals as sources of the infection.
The World Health Organization identified early in the COVID-19 outbreak the global wave of misinformation about the virus and dubbed the problem the “infodemic.” The WHO “Q & A” page on COVID-19 is updated frequently and addresses myths and rumors currently circulating.
According to the WHO website, the agency has reached out to social media players such as Facebook, Twitter, Instagram, LinkedIn, Pinterest, TikTok, and Weibo, the microblogging site in China. WHO has worked with these sites to curb the “infodemic” of misinformation and has used these sites for public education outreach on COVID-19. “Myth busting” infographics posted on a WHO web page are also reposted on major social media sites.
The CDC has followed with its own “frequently asked questions” page to address questions and rumors. State health agencies have put up COVID-19 pages to address public concerns and offer advice on prevention. The Maryland Department of Health web page directly addresses dangerous misinformation: “Do not stigmatize people of any specific ethnicities or racial background. Viruses do not target people from specific populations, ethnicities or racial backgrounds. Stay informed and seek information from reliable, official sources. Be wary of myths, rumors and misinformation circulating online and elsewhere. Health information shared through social media is frequently inaccurate, unless coming from an official, reliable source such as the CDC, MDH or local health departments.”
The Washington State Department of Health has taken a more assertive stance on stigma. The COVID-19 web page recommends to the public: “Show compassion and support for individuals and communities more closely impacted. Avoid stigmatizing people who are in quarantine. They are making the right choice for their communities. Do not make assumptions about someone’s health status based on their ethnicity, race or national origin.”
Misinformation about the COVID-19 travels faster than the virus and complicates the job of doctors who are treating those infected and responding to concerns of their other patients.
An array of myths springing up around this disease can be found on the Internet. The main themes appear to be false narratives about the origin of the virus, the size of the outbreak in the United States and in other countries, the availability of cures and treatments, and ways to prevent infection. Widespread misinformation hampers public health efforts to control the disease outbreak, confuses the public, and requires medical professionals to spend time refuting myths and re-educating patients.
A group of infectious disease experts became so alarmed by the misinformation trend they published a statement in The Lancet decrying the spread of false statements being circulated by some media outlets. “The rapid, open, and transparent sharing of data on this outbreak is now being threatened by rumours and misinformation ... Conspiracy theories do nothing but create fear, rumours, and prejudice that jeopardise our global collaboration in the fight against this virus,” wrote Charles H. Calisher, PhD, of Colorado State University, Fort Collins, and colleagues.
What can physicians do to counter misinformation?
Pulmonologist and critical care physician Cedric “Jamie” Rutland, MD, who practices in Riverside, Calif., sees misinformation about the novel coronavirus every day at home and on the job. His patients worry that everyone who gets infected will die or end up in the ICU. His neighbors ask him to pilfer surgical masks to protect them from the false notion that Chinese people in their community posed some kind of COVID-19 risk.
As he pondered how to counter myths with facts, Dr. Rutland turned to an unusual resource: His 7-year-old daughter Amelia. He explained to her how COVID-19 works and found that she could easily understand the basics. Now, Dr. Rutland draws upon the lessons from chats with his daughter as he explains COVID-19 to his patient audience on his YouTube channel “Medicine Deconstructed.” Simplicity, but not too much simplicity, is key, he said. Dr. Rutland uses a visual aid – a rough drawing of a virus – and shows how inflammation and antibodies enter the picture after infection. “I just teach them that if you’re a healthy person, this is how the body works, and this is what the immune system will do,” he said. “For the most part, you can calm people down when you make time for education.”
What are best practices? In a series of interviews, specialists emphasized the importance of fact-finding, wide-ranging communication, and – perhaps most difficult of all – humility.
Dr. Rutland emphasizes thoughtful communication based on facts and humility when communicating to patients about this potential health risk. “A lot of people finish medical school and think, ‘Everyone should trust me because I’m the pulmonologist or the GI doc.’ That’s not how it works. You still have to earn people’s trust,” he said.
Make sure all staff get reliable information
Hospitals are scrambling to keep staff safe with up-to-date directives and debunk false narratives about the virus. Keeping all hospital staff informed with verified and authoritative facts about the coronavirus is a key objective of the Massachusetts General Hospital’s Center for Disaster Medicine. The Center’s coronavirus educational materials are distributed to all staffers from physicians to janitors. “These provide information that they need to understand the risks and keep themselves safe,” said Eileen Searle, PhD, the Biothreats Clinical Operations program manager in the CDM.
According to Dr. Searle, the hospital keeps a continually updated COVID-19 Frequently Asked Questions document in its internal computer system. All employees can access it, she said, and it’s updated to include questions as they come up.
Even valets and front-desk volunteers are encouraged to read the FAQ, she said, since “they’re the first people that family and patients are interacting with.” The document “gives them reassurance about delivering messages,” she said.
Use patience with your patients
Dr. Rutland urges colleagues to take the time to listen to patients and educate them. “Reduce the gap between you and them,” said Dr. Rutland, who treats patients in Orange and Riverside counties. “Take off your white coat, sit down, and talk to the person about their concerns.”
Boston cardiologist Haider Warraich, MD, of Brigham and Women’s Hospital, Boston, said it’s important to “put medical information into a greater human context.” For example, he has told patients that he’s still taking his daughter to school despite COVID-19 risks. “I take the information I provide and apply it to my own life,” he said.
The Washington State Department of Health offers this advice to physicians to counter false information and stigma: “Stay updated and informed on COVID-19 to avoid miscommunication or inaccurate information. Talk openly about the harm of stigma. View people directly impacted by stigma as people first. Be conscious of your language. Acknowledge access and language barriers.”
Speak out on social media – but don’t fight
Should medical professionals speak out about COVID-19 misinformation via social media? It’s an individual decision, Dr. Warraich said, “but my sense is that it’s never been more important for physicians to be part of the fray and help quell the epidemic of misinformation that almost always follows any type of medial calamity.”
Dr. Rutland, vice president and founding member of the Association for Healthcare Social Media, cautioned that effective communication via social media requires care. Avoid confrontation, he advised. “Don’t call people stupid or say things like, ‘I went to medical school and I’m smarter than you.’ ”
Instead, he said, “it’s important to just state the facts: These are the people who are dying, these are the people who are getting infected.”
And, he added, remember to push the most important message of all: Wash your hands!
Public health organizations fight the ‘infodemic’
In a trend that hearkens back to the days of snake oil cures for all maladies, advertisements for fake treatments are popping up on the Internet and on other media.
Facebook and Amazon have acted to remove these ads but these messages continue to flood social media such as Twitter, WhatsApp, and other sites. Discussion groups on platforms such as Reddit continue to pump out misinformation about COVID-19. Conspiracy theories that link the virus to espionage and bioweapons are making the rounds on the Internet and talk radio. Wrong information about the effectiveness of non-N95 face masks to protect wearers against infection is widespread, leading to shortages for medical personnel and price gouging. Pernicious rumors about the effectiveness of substances such a vinegar, silver, garlic, lemon juice, and even vodka to disinfect hands and surfaces abound on the Internet. An especially dangerous stream of misinformation stigmatizes ethnic groups and individuals as sources of the infection.
The World Health Organization identified early in the COVID-19 outbreak the global wave of misinformation about the virus and dubbed the problem the “infodemic.” The WHO “Q & A” page on COVID-19 is updated frequently and addresses myths and rumors currently circulating.
According to the WHO website, the agency has reached out to social media players such as Facebook, Twitter, Instagram, LinkedIn, Pinterest, TikTok, and Weibo, the microblogging site in China. WHO has worked with these sites to curb the “infodemic” of misinformation and has used these sites for public education outreach on COVID-19. “Myth busting” infographics posted on a WHO web page are also reposted on major social media sites.
The CDC has followed with its own “frequently asked questions” page to address questions and rumors. State health agencies have put up COVID-19 pages to address public concerns and offer advice on prevention. The Maryland Department of Health web page directly addresses dangerous misinformation: “Do not stigmatize people of any specific ethnicities or racial background. Viruses do not target people from specific populations, ethnicities or racial backgrounds. Stay informed and seek information from reliable, official sources. Be wary of myths, rumors and misinformation circulating online and elsewhere. Health information shared through social media is frequently inaccurate, unless coming from an official, reliable source such as the CDC, MDH or local health departments.”
The Washington State Department of Health has taken a more assertive stance on stigma. The COVID-19 web page recommends to the public: “Show compassion and support for individuals and communities more closely impacted. Avoid stigmatizing people who are in quarantine. They are making the right choice for their communities. Do not make assumptions about someone’s health status based on their ethnicity, race or national origin.”