User login
Ibrutinib associated with decreased circulating malignant cells and restored T-cell function in CLL patients
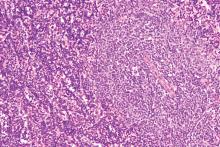
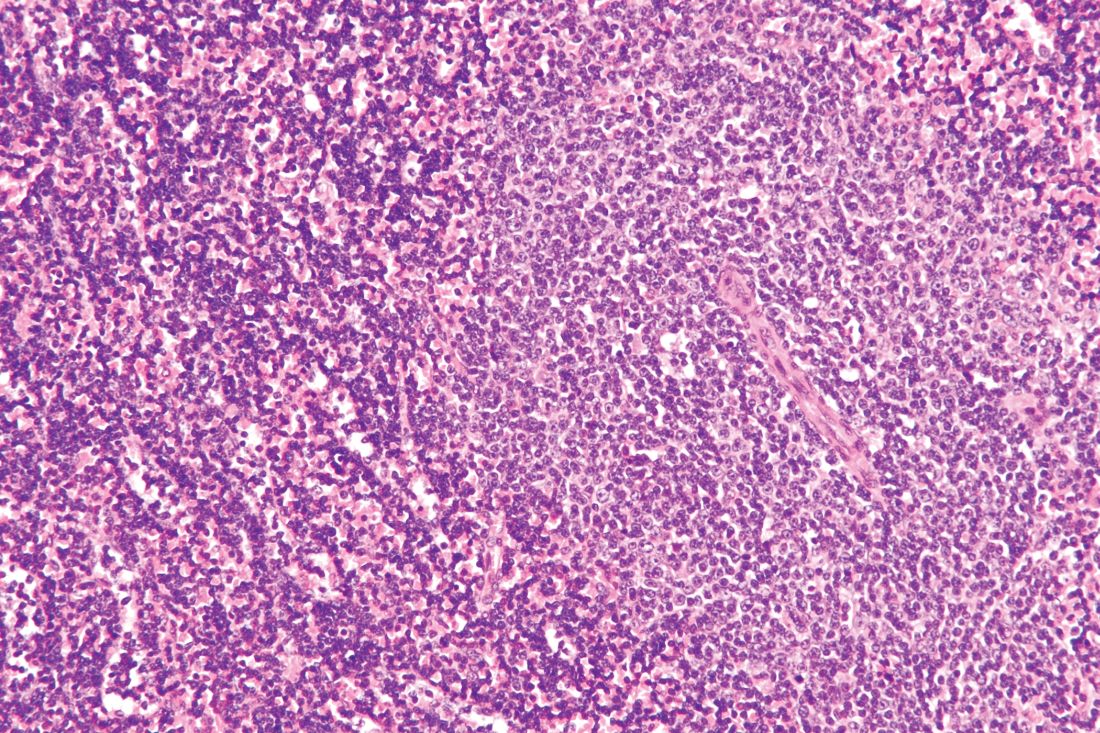
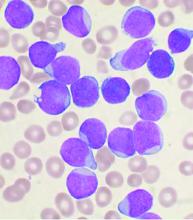
Ibrutinib showed significant impact on circulating malignant and nonmalignant immune cells and was found to restore healthy T-cell function in patients with chronic lymphocytic leukemia (CLL), according to the results of a comparative study of CLL patients and healthy controls.
Researchers compared circulating counts of 21 immune blood cell subsets throughout the first year of treatment in 55 patients with relapsed/refractory (R/R) CLL from the RESONATE trial and 50 previously untreated CLL patients from the RESONATE-2 trial with 20 untreated age-matched healthy donors, according to a report published online in Leukemia Research.
In addition, T-cell function was assessed in response to T-cell–receptor stimulation in 21 patients with R/R CLL, compared with 18 age-matched healthy donors, according to Isabelle G. Solman, MS, an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. and colleagues.
Positive indicators
Ibrutinib significantly decreased pathologically high circulating B cells, regulatory T cells, effector/memory CD4+ and CD8+ T cells (including exhausted and chronically activated T cells), natural killer (NK) T cells, and myeloid-derived suppressor cells; preserved naive T cells and NK cells; and increased circulating classical monocytes, according to the researchers.
Ibrutinib also significantly restored T-cell proliferative ability, degranulation, and cytokine secretion. Over the same period, ofatumumab or chlorambucil did not confer the same spectrum of normalization as ibrutinib in multiple immune subsets that were examined, they added.
“These results establish that ibrutinib has a significant and likely positive impact on circulating malignant and nonmalignant immune cells and restores healthy T-cell function,” the researchers indicated.
“Ibrutinib has a significant, progressively positive impact on both malignant and nonmalignant immune cells in CLL. These positive effects on circulating nonmalignant immune cells may contribute to long-term CLL disease control, overall health status, and decreased susceptibility to infection,” they concluded.
The study was funded by Pharmacyclics, an AbbVie Company. Ms. Solman is an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. as were several other authors.
SOURCE: Solman IG et al. Leuk Res. 2020;97. doi: 10.1016/j.leukres.2020.106432.
Ibrutinib showed significant impact on circulating malignant and nonmalignant immune cells and was found to restore healthy T-cell function in patients with chronic lymphocytic leukemia (CLL), according to the results of a comparative study of CLL patients and healthy controls.
Researchers compared circulating counts of 21 immune blood cell subsets throughout the first year of treatment in 55 patients with relapsed/refractory (R/R) CLL from the RESONATE trial and 50 previously untreated CLL patients from the RESONATE-2 trial with 20 untreated age-matched healthy donors, according to a report published online in Leukemia Research.
In addition, T-cell function was assessed in response to T-cell–receptor stimulation in 21 patients with R/R CLL, compared with 18 age-matched healthy donors, according to Isabelle G. Solman, MS, an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. and colleagues.
Positive indicators
Ibrutinib significantly decreased pathologically high circulating B cells, regulatory T cells, effector/memory CD4+ and CD8+ T cells (including exhausted and chronically activated T cells), natural killer (NK) T cells, and myeloid-derived suppressor cells; preserved naive T cells and NK cells; and increased circulating classical monocytes, according to the researchers.
Ibrutinib also significantly restored T-cell proliferative ability, degranulation, and cytokine secretion. Over the same period, ofatumumab or chlorambucil did not confer the same spectrum of normalization as ibrutinib in multiple immune subsets that were examined, they added.
“These results establish that ibrutinib has a significant and likely positive impact on circulating malignant and nonmalignant immune cells and restores healthy T-cell function,” the researchers indicated.
“Ibrutinib has a significant, progressively positive impact on both malignant and nonmalignant immune cells in CLL. These positive effects on circulating nonmalignant immune cells may contribute to long-term CLL disease control, overall health status, and decreased susceptibility to infection,” they concluded.
The study was funded by Pharmacyclics, an AbbVie Company. Ms. Solman is an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. as were several other authors.
SOURCE: Solman IG et al. Leuk Res. 2020;97. doi: 10.1016/j.leukres.2020.106432.
Ibrutinib showed significant impact on circulating malignant and nonmalignant immune cells and was found to restore healthy T-cell function in patients with chronic lymphocytic leukemia (CLL), according to the results of a comparative study of CLL patients and healthy controls.
Researchers compared circulating counts of 21 immune blood cell subsets throughout the first year of treatment in 55 patients with relapsed/refractory (R/R) CLL from the RESONATE trial and 50 previously untreated CLL patients from the RESONATE-2 trial with 20 untreated age-matched healthy donors, according to a report published online in Leukemia Research.
In addition, T-cell function was assessed in response to T-cell–receptor stimulation in 21 patients with R/R CLL, compared with 18 age-matched healthy donors, according to Isabelle G. Solman, MS, an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. and colleagues.
Positive indicators
Ibrutinib significantly decreased pathologically high circulating B cells, regulatory T cells, effector/memory CD4+ and CD8+ T cells (including exhausted and chronically activated T cells), natural killer (NK) T cells, and myeloid-derived suppressor cells; preserved naive T cells and NK cells; and increased circulating classical monocytes, according to the researchers.
Ibrutinib also significantly restored T-cell proliferative ability, degranulation, and cytokine secretion. Over the same period, ofatumumab or chlorambucil did not confer the same spectrum of normalization as ibrutinib in multiple immune subsets that were examined, they added.
“These results establish that ibrutinib has a significant and likely positive impact on circulating malignant and nonmalignant immune cells and restores healthy T-cell function,” the researchers indicated.
“Ibrutinib has a significant, progressively positive impact on both malignant and nonmalignant immune cells in CLL. These positive effects on circulating nonmalignant immune cells may contribute to long-term CLL disease control, overall health status, and decreased susceptibility to infection,” they concluded.
The study was funded by Pharmacyclics, an AbbVie Company. Ms. Solman is an employee of Translational Medicine, Pharmacyclics, Sunnyvale, Calif. as were several other authors.
SOURCE: Solman IG et al. Leuk Res. 2020;97. doi: 10.1016/j.leukres.2020.106432.
FROM LEUKEMIA RESEARCH
Beat AML: Precision medicine strategy feasible, superior to SOC for AML
The 30-day mortality rates were 3.7% versus 20.4% in 224 patients who enrolled in the Beat AML trial precision medicine substudies within 7 days of prospective genomic profiling and 103 who elected SOC chemotherapy, respectively, Amy Burd, PhD, vice president of research strategy for the Leukemia & Lymphoma Society, Rye Brook, N.Y. and her colleagues reported online in Nature Medicine.
Overall survival (OS) at a median of 7.1 months was also significantly longer with precision medicine than with SOC chemotherapy (median, 12.8 vs. 3.9 months), the investigators found.
In an additional 28 patients who selected an investigational therapy rather than a precision medicine strategy or SOC chemotherapy, median OS was not reached, and in 38 who chose palliative care, median OS was 0.6 months, they noted. Care type was unknown in two patients.
The results were similar after controlling for demographic, clinical, and molecular variables and did not change when patients with adverse events of special interest were excluded from the analysis or when only those with survival greater than 2 weeks were included in the analysis.
AML confers an adverse outcome in older adults and therefore is typically treated rapidly after diagnosis. This has precluded consideration of patients’ mutational profile for treatment decisions.
Beat AML, however, sought to prospectively assess the feasibility of quickly ascertaining cytogenetic and mutational data for the purpose of improving outcomes through targeted treatment.
“The study shows that delaying treatment up to 7 days is feasible and safe, and that patients who opted for the precision medicine approach experienced a lower early death rate and superior overall survival, compared with patients who opted for standard of care,” lead study author John C. Byrd, MD, the D. Warren Brown Chair of Leukemia Research of the Ohio State University, Columbus, noted in a press statement from the Leukemia & Lymphoma Society, which conducted the trial. “This patient-centric study shows that we can move away from chemotherapy treatment for patients who won’t respond or can’t withstand the harsh effects of the same chemotherapies we’ve been using for 40 years and match them with a treatment better suited for their individual cases.”
The ongoing Beat AML trial was launched by LLS in 2016 to assess various novel targeted therapies in newly diagnosed AML patients aged 60 years and older. Participants underwent next-generation genomic sequencing, were matched to the appropriate targeted therapy, and were given the option of enrolling on the relevant substudy or selecting an alternate treatment strategy. There are currently 11 substudies assessing novel therapies that have emerged in the wake of “significant progress in understanding the molecular pathogenesis of AML.”
The current findings represent outcomes in patients enrolled between Nov. 2016 and Jan. 2018. The patients had a mean age of 72 years, and those selecting precision medicine vs. SOC had similar demographic and genetic features, the authors noted.
LLS president and chief executive officer Louis J. DeGennaro, PhD, said the findings are practice changing and provide a template for studying precision medicine in other cancers.
“The study is changing significantly the way we look at treating patients with AML, showing that precision medicine ... can improve short- and long-term outcomes for patients with this deadly blood cancer,” he said in the LLS statement. “Further, BEAT AML has proven to be a viable model for other cancer clinical trials to emulate.”
In fact, the model has been applied to the recently launched Beat COVID trial, which looks at acalabrutinib in patients with hematologic cancers and COVID-19 infection, and other trials, including the LLS PedAL global precision medicine trial for children with relapsed acute leukemia, are planned.
“This study sets the path to establish the safety of precision medicine in AML and sets the stage to extend this same approach to younger patients with this disease and other cancers that are urgently treated as a single disease despite recognition of multiple subtypes, the authors concluded.
Dr. Burd is an employee of LLS, which received funding from AbbVie, Agios Pharmaceuticals, Alexion Pharmaceuticals, and a variety of other pharmaceutical and biotechnology companies. Dr. Byrd has received research support from Acerta Pharma, Genentech, Janssen Pharmaceutica, and Pharmacyclics and has served on the advisory board of Syndax Pharmaceuticals.
SOURCE: Burd A et al. Nature Medicine 2020 Oct 26. doi: 10.1038/s41591-020-1089-8.
The 30-day mortality rates were 3.7% versus 20.4% in 224 patients who enrolled in the Beat AML trial precision medicine substudies within 7 days of prospective genomic profiling and 103 who elected SOC chemotherapy, respectively, Amy Burd, PhD, vice president of research strategy for the Leukemia & Lymphoma Society, Rye Brook, N.Y. and her colleagues reported online in Nature Medicine.
Overall survival (OS) at a median of 7.1 months was also significantly longer with precision medicine than with SOC chemotherapy (median, 12.8 vs. 3.9 months), the investigators found.
In an additional 28 patients who selected an investigational therapy rather than a precision medicine strategy or SOC chemotherapy, median OS was not reached, and in 38 who chose palliative care, median OS was 0.6 months, they noted. Care type was unknown in two patients.
The results were similar after controlling for demographic, clinical, and molecular variables and did not change when patients with adverse events of special interest were excluded from the analysis or when only those with survival greater than 2 weeks were included in the analysis.
AML confers an adverse outcome in older adults and therefore is typically treated rapidly after diagnosis. This has precluded consideration of patients’ mutational profile for treatment decisions.
Beat AML, however, sought to prospectively assess the feasibility of quickly ascertaining cytogenetic and mutational data for the purpose of improving outcomes through targeted treatment.
“The study shows that delaying treatment up to 7 days is feasible and safe, and that patients who opted for the precision medicine approach experienced a lower early death rate and superior overall survival, compared with patients who opted for standard of care,” lead study author John C. Byrd, MD, the D. Warren Brown Chair of Leukemia Research of the Ohio State University, Columbus, noted in a press statement from the Leukemia & Lymphoma Society, which conducted the trial. “This patient-centric study shows that we can move away from chemotherapy treatment for patients who won’t respond or can’t withstand the harsh effects of the same chemotherapies we’ve been using for 40 years and match them with a treatment better suited for their individual cases.”
The ongoing Beat AML trial was launched by LLS in 2016 to assess various novel targeted therapies in newly diagnosed AML patients aged 60 years and older. Participants underwent next-generation genomic sequencing, were matched to the appropriate targeted therapy, and were given the option of enrolling on the relevant substudy or selecting an alternate treatment strategy. There are currently 11 substudies assessing novel therapies that have emerged in the wake of “significant progress in understanding the molecular pathogenesis of AML.”
The current findings represent outcomes in patients enrolled between Nov. 2016 and Jan. 2018. The patients had a mean age of 72 years, and those selecting precision medicine vs. SOC had similar demographic and genetic features, the authors noted.
LLS president and chief executive officer Louis J. DeGennaro, PhD, said the findings are practice changing and provide a template for studying precision medicine in other cancers.
“The study is changing significantly the way we look at treating patients with AML, showing that precision medicine ... can improve short- and long-term outcomes for patients with this deadly blood cancer,” he said in the LLS statement. “Further, BEAT AML has proven to be a viable model for other cancer clinical trials to emulate.”
In fact, the model has been applied to the recently launched Beat COVID trial, which looks at acalabrutinib in patients with hematologic cancers and COVID-19 infection, and other trials, including the LLS PedAL global precision medicine trial for children with relapsed acute leukemia, are planned.
“This study sets the path to establish the safety of precision medicine in AML and sets the stage to extend this same approach to younger patients with this disease and other cancers that are urgently treated as a single disease despite recognition of multiple subtypes, the authors concluded.
Dr. Burd is an employee of LLS, which received funding from AbbVie, Agios Pharmaceuticals, Alexion Pharmaceuticals, and a variety of other pharmaceutical and biotechnology companies. Dr. Byrd has received research support from Acerta Pharma, Genentech, Janssen Pharmaceutica, and Pharmacyclics and has served on the advisory board of Syndax Pharmaceuticals.
SOURCE: Burd A et al. Nature Medicine 2020 Oct 26. doi: 10.1038/s41591-020-1089-8.
The 30-day mortality rates were 3.7% versus 20.4% in 224 patients who enrolled in the Beat AML trial precision medicine substudies within 7 days of prospective genomic profiling and 103 who elected SOC chemotherapy, respectively, Amy Burd, PhD, vice president of research strategy for the Leukemia & Lymphoma Society, Rye Brook, N.Y. and her colleagues reported online in Nature Medicine.
Overall survival (OS) at a median of 7.1 months was also significantly longer with precision medicine than with SOC chemotherapy (median, 12.8 vs. 3.9 months), the investigators found.
In an additional 28 patients who selected an investigational therapy rather than a precision medicine strategy or SOC chemotherapy, median OS was not reached, and in 38 who chose palliative care, median OS was 0.6 months, they noted. Care type was unknown in two patients.
The results were similar after controlling for demographic, clinical, and molecular variables and did not change when patients with adverse events of special interest were excluded from the analysis or when only those with survival greater than 2 weeks were included in the analysis.
AML confers an adverse outcome in older adults and therefore is typically treated rapidly after diagnosis. This has precluded consideration of patients’ mutational profile for treatment decisions.
Beat AML, however, sought to prospectively assess the feasibility of quickly ascertaining cytogenetic and mutational data for the purpose of improving outcomes through targeted treatment.
“The study shows that delaying treatment up to 7 days is feasible and safe, and that patients who opted for the precision medicine approach experienced a lower early death rate and superior overall survival, compared with patients who opted for standard of care,” lead study author John C. Byrd, MD, the D. Warren Brown Chair of Leukemia Research of the Ohio State University, Columbus, noted in a press statement from the Leukemia & Lymphoma Society, which conducted the trial. “This patient-centric study shows that we can move away from chemotherapy treatment for patients who won’t respond or can’t withstand the harsh effects of the same chemotherapies we’ve been using for 40 years and match them with a treatment better suited for their individual cases.”
The ongoing Beat AML trial was launched by LLS in 2016 to assess various novel targeted therapies in newly diagnosed AML patients aged 60 years and older. Participants underwent next-generation genomic sequencing, were matched to the appropriate targeted therapy, and were given the option of enrolling on the relevant substudy or selecting an alternate treatment strategy. There are currently 11 substudies assessing novel therapies that have emerged in the wake of “significant progress in understanding the molecular pathogenesis of AML.”
The current findings represent outcomes in patients enrolled between Nov. 2016 and Jan. 2018. The patients had a mean age of 72 years, and those selecting precision medicine vs. SOC had similar demographic and genetic features, the authors noted.
LLS president and chief executive officer Louis J. DeGennaro, PhD, said the findings are practice changing and provide a template for studying precision medicine in other cancers.
“The study is changing significantly the way we look at treating patients with AML, showing that precision medicine ... can improve short- and long-term outcomes for patients with this deadly blood cancer,” he said in the LLS statement. “Further, BEAT AML has proven to be a viable model for other cancer clinical trials to emulate.”
In fact, the model has been applied to the recently launched Beat COVID trial, which looks at acalabrutinib in patients with hematologic cancers and COVID-19 infection, and other trials, including the LLS PedAL global precision medicine trial for children with relapsed acute leukemia, are planned.
“This study sets the path to establish the safety of precision medicine in AML and sets the stage to extend this same approach to younger patients with this disease and other cancers that are urgently treated as a single disease despite recognition of multiple subtypes, the authors concluded.
Dr. Burd is an employee of LLS, which received funding from AbbVie, Agios Pharmaceuticals, Alexion Pharmaceuticals, and a variety of other pharmaceutical and biotechnology companies. Dr. Byrd has received research support from Acerta Pharma, Genentech, Janssen Pharmaceutica, and Pharmacyclics and has served on the advisory board of Syndax Pharmaceuticals.
SOURCE: Burd A et al. Nature Medicine 2020 Oct 26. doi: 10.1038/s41591-020-1089-8.
FROM NATURE MEDICINE
Are HMAS appropriate for posttransplant maintenance in acute leukemias?
Hematopoietic stem cell transplantation (HCT) is one of the most important treatment options for acute leukemias. However, posttransplant cancer recurrence remains a continuing issue. And while there are reasons to think that hypomethylating agents (HMAS) could be helpful as maintenance tools to prevent cancer recurrence after HCT in leukemia, a hematologist/oncologist told colleagues that the treatment isn’t yet ready for prime time.
“I don’t think you can prefer hypomethylating agents over anything right now. Unfortunately, there’s no data that we can hang our hat on that says they are of benefit in the posttransplant setting,” said Frederick Appelbaum, MD, executive vice president and deputy director of the Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
However, there’s still plenty of room for improvement for patients following HCT, he said, pointing to the findings of a 2020 study. The report, which he cowrote, found that 200-day mortality after HCT fell by a third from 2003-2007 to 2013-20017, but also noted that “relapse of cancer remains the largest obstacle to better survival outcomes.”
Dr. Appelbaum described the findings this way: “Without a doubt, the major limitation to transplants for hematologic malignancies today is disease recurrence,” he said. “In fact, if you look at patients after day 100, over 60% of the reason for failure is tumor regrowth. Thus, people are very anxious to look at any method that we can to prevent posttransplant relapse, including the use of hypomethylating agents.”
In regard to strategy, “we don’t have to get rid of every last leukemic cell. Just delaying recurrence might be enough,” he said. “If you can keep the patient from relapsing for the first 3 months, and then take the brakes off the immune suppression and allow immunity to regrow, that may be enough to allow increased numbers of patients to be cured of their disease.”
A potential role
Why might HMAS be a possible option after transplant? They do appear to play a role after chemotherapy, he said, pointing to four 2019 studies: One that examined decitabine and three that examined azacytidine: Here, here, and here.
“These four studies provide convincing evidence that hypomethylating-agent therapy after conventional chemotherapy may either prevent or delay relapse when given as maintenance,” Dr. Appelbaum said.
If HMAS work after standard chemotherapy, why might they fail to work after transplantation? “For one, by the time the disease has been able to go through chemotherapy and transplant, you’re left with highly resistant cells,” he said. “Therefore, hypomethylating agents may not be enough to get rid of the disease. Secondly, any of you who have tried to give a maintenance therapy after transplantation know how difficult it can be with CMV [cytomegalovirus] reactivation, count suppression with ganciclovir, graft-versus-host disease [GVHD] causing nausea and vomiting, diarrhea and renal dysfunction caused by calcineurin inhibitors. These are daily events during the first 3 months after transplantation, making drug administration difficult.”
In addition, he said, “even if you can give the drug, the clinical and disease variability may make it very difficult to detect an effect.”
In another study, researchers “did make a valiant attempt to study azacitidine in the posttransplant setting by randomizing 181 patients to either azacitidine or observation,” Dr. Appelbaum said. “Unfortunately, as they reported in 2018, they could not detect a difference in either disease-free or overall survival.”
The researchers reported that nearly 75% of patients in the azacitidine arm failed to complete the planned 12 cycles of treatment, he said. “The reasons for stopping the drug were pretty profound. Half of the patients stopped because they relapsed. Others had stopped because of grades three or four toxicity, death, or severe GVHD or significant infections. It is very difficult to give the drug.”
In the future, “if we truly want to optimize the benefit of using hypomethylating agents after transplantation, it’s going to be very important for us to understand how they work,” he said. “Understanding that would then help us to select which drug we should use, what the dosing and schedule might be, and also to select patients that might benefit from it. Unfortunately, right now, it’s pretty much of a black box. We don’t really understand the effects of hypomethylating agents in the posttransplant period.”
Still, he added, “without question, the results that we have seen with the use of hypomethylating agents after conventional chemotherapy – prolonging disease-free and, probably, overall survival – are going to provide a very, very strong stimulus to study hypomethylating agents after transplantation as well.”
Dr. Appelbaum reports no disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Hematopoietic stem cell transplantation (HCT) is one of the most important treatment options for acute leukemias. However, posttransplant cancer recurrence remains a continuing issue. And while there are reasons to think that hypomethylating agents (HMAS) could be helpful as maintenance tools to prevent cancer recurrence after HCT in leukemia, a hematologist/oncologist told colleagues that the treatment isn’t yet ready for prime time.
“I don’t think you can prefer hypomethylating agents over anything right now. Unfortunately, there’s no data that we can hang our hat on that says they are of benefit in the posttransplant setting,” said Frederick Appelbaum, MD, executive vice president and deputy director of the Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
However, there’s still plenty of room for improvement for patients following HCT, he said, pointing to the findings of a 2020 study. The report, which he cowrote, found that 200-day mortality after HCT fell by a third from 2003-2007 to 2013-20017, but also noted that “relapse of cancer remains the largest obstacle to better survival outcomes.”
Dr. Appelbaum described the findings this way: “Without a doubt, the major limitation to transplants for hematologic malignancies today is disease recurrence,” he said. “In fact, if you look at patients after day 100, over 60% of the reason for failure is tumor regrowth. Thus, people are very anxious to look at any method that we can to prevent posttransplant relapse, including the use of hypomethylating agents.”
In regard to strategy, “we don’t have to get rid of every last leukemic cell. Just delaying recurrence might be enough,” he said. “If you can keep the patient from relapsing for the first 3 months, and then take the brakes off the immune suppression and allow immunity to regrow, that may be enough to allow increased numbers of patients to be cured of their disease.”
A potential role
Why might HMAS be a possible option after transplant? They do appear to play a role after chemotherapy, he said, pointing to four 2019 studies: One that examined decitabine and three that examined azacytidine: Here, here, and here.
“These four studies provide convincing evidence that hypomethylating-agent therapy after conventional chemotherapy may either prevent or delay relapse when given as maintenance,” Dr. Appelbaum said.
If HMAS work after standard chemotherapy, why might they fail to work after transplantation? “For one, by the time the disease has been able to go through chemotherapy and transplant, you’re left with highly resistant cells,” he said. “Therefore, hypomethylating agents may not be enough to get rid of the disease. Secondly, any of you who have tried to give a maintenance therapy after transplantation know how difficult it can be with CMV [cytomegalovirus] reactivation, count suppression with ganciclovir, graft-versus-host disease [GVHD] causing nausea and vomiting, diarrhea and renal dysfunction caused by calcineurin inhibitors. These are daily events during the first 3 months after transplantation, making drug administration difficult.”
In addition, he said, “even if you can give the drug, the clinical and disease variability may make it very difficult to detect an effect.”
In another study, researchers “did make a valiant attempt to study azacitidine in the posttransplant setting by randomizing 181 patients to either azacitidine or observation,” Dr. Appelbaum said. “Unfortunately, as they reported in 2018, they could not detect a difference in either disease-free or overall survival.”
The researchers reported that nearly 75% of patients in the azacitidine arm failed to complete the planned 12 cycles of treatment, he said. “The reasons for stopping the drug were pretty profound. Half of the patients stopped because they relapsed. Others had stopped because of grades three or four toxicity, death, or severe GVHD or significant infections. It is very difficult to give the drug.”
In the future, “if we truly want to optimize the benefit of using hypomethylating agents after transplantation, it’s going to be very important for us to understand how they work,” he said. “Understanding that would then help us to select which drug we should use, what the dosing and schedule might be, and also to select patients that might benefit from it. Unfortunately, right now, it’s pretty much of a black box. We don’t really understand the effects of hypomethylating agents in the posttransplant period.”
Still, he added, “without question, the results that we have seen with the use of hypomethylating agents after conventional chemotherapy – prolonging disease-free and, probably, overall survival – are going to provide a very, very strong stimulus to study hypomethylating agents after transplantation as well.”
Dr. Appelbaum reports no disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Hematopoietic stem cell transplantation (HCT) is one of the most important treatment options for acute leukemias. However, posttransplant cancer recurrence remains a continuing issue. And while there are reasons to think that hypomethylating agents (HMAS) could be helpful as maintenance tools to prevent cancer recurrence after HCT in leukemia, a hematologist/oncologist told colleagues that the treatment isn’t yet ready for prime time.
“I don’t think you can prefer hypomethylating agents over anything right now. Unfortunately, there’s no data that we can hang our hat on that says they are of benefit in the posttransplant setting,” said Frederick Appelbaum, MD, executive vice president and deputy director of the Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
However, there’s still plenty of room for improvement for patients following HCT, he said, pointing to the findings of a 2020 study. The report, which he cowrote, found that 200-day mortality after HCT fell by a third from 2003-2007 to 2013-20017, but also noted that “relapse of cancer remains the largest obstacle to better survival outcomes.”
Dr. Appelbaum described the findings this way: “Without a doubt, the major limitation to transplants for hematologic malignancies today is disease recurrence,” he said. “In fact, if you look at patients after day 100, over 60% of the reason for failure is tumor regrowth. Thus, people are very anxious to look at any method that we can to prevent posttransplant relapse, including the use of hypomethylating agents.”
In regard to strategy, “we don’t have to get rid of every last leukemic cell. Just delaying recurrence might be enough,” he said. “If you can keep the patient from relapsing for the first 3 months, and then take the brakes off the immune suppression and allow immunity to regrow, that may be enough to allow increased numbers of patients to be cured of their disease.”
A potential role
Why might HMAS be a possible option after transplant? They do appear to play a role after chemotherapy, he said, pointing to four 2019 studies: One that examined decitabine and three that examined azacytidine: Here, here, and here.
“These four studies provide convincing evidence that hypomethylating-agent therapy after conventional chemotherapy may either prevent or delay relapse when given as maintenance,” Dr. Appelbaum said.
If HMAS work after standard chemotherapy, why might they fail to work after transplantation? “For one, by the time the disease has been able to go through chemotherapy and transplant, you’re left with highly resistant cells,” he said. “Therefore, hypomethylating agents may not be enough to get rid of the disease. Secondly, any of you who have tried to give a maintenance therapy after transplantation know how difficult it can be with CMV [cytomegalovirus] reactivation, count suppression with ganciclovir, graft-versus-host disease [GVHD] causing nausea and vomiting, diarrhea and renal dysfunction caused by calcineurin inhibitors. These are daily events during the first 3 months after transplantation, making drug administration difficult.”
In addition, he said, “even if you can give the drug, the clinical and disease variability may make it very difficult to detect an effect.”
In another study, researchers “did make a valiant attempt to study azacitidine in the posttransplant setting by randomizing 181 patients to either azacitidine or observation,” Dr. Appelbaum said. “Unfortunately, as they reported in 2018, they could not detect a difference in either disease-free or overall survival.”
The researchers reported that nearly 75% of patients in the azacitidine arm failed to complete the planned 12 cycles of treatment, he said. “The reasons for stopping the drug were pretty profound. Half of the patients stopped because they relapsed. Others had stopped because of grades three or four toxicity, death, or severe GVHD or significant infections. It is very difficult to give the drug.”
In the future, “if we truly want to optimize the benefit of using hypomethylating agents after transplantation, it’s going to be very important for us to understand how they work,” he said. “Understanding that would then help us to select which drug we should use, what the dosing and schedule might be, and also to select patients that might benefit from it. Unfortunately, right now, it’s pretty much of a black box. We don’t really understand the effects of hypomethylating agents in the posttransplant period.”
Still, he added, “without question, the results that we have seen with the use of hypomethylating agents after conventional chemotherapy – prolonging disease-free and, probably, overall survival – are going to provide a very, very strong stimulus to study hypomethylating agents after transplantation as well.”
Dr. Appelbaum reports no disclosures.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
FROM ALF 2020
Novel agents hold promise for frontline AML treatment
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
Novel therapies are poised to dramatically change frontline therapy for acute myeloid leukemia (AML), and they have the potential to replace chemotherapy, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus.
But more work needs to be done, noted Alexander Perl, MD, MS, associate professor at the University of Pennsylvania, Philadelphia. While advances have transformed AML treatment in the relapsed/refractory setting, “we’re just not seeing that substantive improvement” for newly diagnosed patients, he said. “We need to find the disease-modifying drugs that work in the relapsed/refractory setting and move those frontline. That’s where we’re going to see the transformations.”
Research suggests that low-intensity therapy holds tremendous promise, he said, “with the idea that we could make therapy much more tolerable for the vast majority of patients affected by AML, who, as we know, are older patients.”
Dr. Perl highlighted the 2020 VIALE-A study – venetoclax/azacitidine versus azacitidine/placebo – which reported that “in previously untreated patients who were ineligible for intensive chemotherapy, overall survival was longer and the incidence of remission was higher among patients who received azacitidine plus venetoclax than among those who received azacitidine alone.”
Venetoclax promotes apoptosis in leukemia cells, Dr. Perl said. “To a certain extent, you can think of it as putting the rubber to the road in terms of what actually chemotherapy is designed to do, which is to make leukemic blasts apoptose. It does so without DNA damage and with much less toxicity to the patient. Therefore it can be added to any number of regimens – granted, with mild suppression, but with relatively little extramedullary toxicity.”
Dr. Perl noted that the venetoclax arm “showed a higher response rate than azacitidine in pretty much every subgroup that was looked at, whether patients had de novo leukemia, secondary leukemia, multiple mutational complements, various different karyotypes. The response rates on this study are as high as what we often will see with intensive chemotherapy.” He added that “the winning arm on this trial seems to hold up against any low-intensity therapy, and I would argue against many high-intensity therapies in older patients.”
As for other targeted agents, isocitrate dehydrogenase (IDH) inhibitors “are very promising drugs in the relapsed/refractory setting, which is primarily where these drugs are given. In regard to frontline treatment, “data are coming from a very small study, but they’re very encouraging. It’s hard to entirely say that we’re ready to change practice based on this. But it’s very encouraging – the idea that earlier use of a drug-targeting IDH mutation might lead to substantially better outcomes.”
Moving forward, he said, “we could put all of our eggs in one basket and use many active drugs [at] front line. Or we can perhaps be smart about sequencing these drugs one after another, or using more intensive approaches followed by maintenance approaches followed by more intensive approaches.”
This approach is similar to strategies in myeloma patients “who less and less are relying on an autologous transplant for durable control of their disease, and more and more are using low-intensity biologically targeted drugs,” he said.
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Perl reported numerous disclosures, including relationships with Daiichi Sankyo, Abbvie, and Astellas.
FROM ALF 2020
Novel treatments under study for chronic graft-versus-host disease in allo-HCT
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Physicians are gaining a greater understanding of the pathophysiology of chronic graft-versus-host disease (cGVHD) in allo-hematopoietic cell transplantation (allo-HCT), a hematologist/oncologist told colleagues, and novel treatments are being tested.
However, options remain limited. There’s only one Food and Drug Administration–approved therapy for cGVHD that’s failed one or more treatments, and clinical trials remain a crucial option in some cases, said Mary E.D. Flowers, MD, professor of medicine at the University of Washington’s Fred Hutchinson Cancer Research Center, Seattle, in a presentation at the virtual Acute Leukemia Forum of Hemedicus.
According to Dr. Flowers, cGVHD – a product of a graft’s “immunological assault” against the person receiving a transplant – occurs in 40% of patients within a year after allo-HCT. The disorder “is associated with a poor quality of life, disability, and increased mortality after allo-transplantation,” she said. “It’s a syndrome that can be inflammatory and fibrotic. It involves several organs – the skin, the mouth, the eyes, the lungs, the GI tract.”
The median length of treatment after peripheral blood stem cell transplant is 3.5 years, Dr. Flowers said. Seven years after treatment, 10% of those who are alive – and have avoided relapse – will still need treatment. “Corticosteroids remain the first-line [treatment], at 0.5-1.0 [mg/kg], but they do not control at least 40% of the patients with cGVHD.”
In regard to pathophysiology, she highlighted a 2017 report that presented findings about the pathophysiology of cGVHD. The findings, the report authors wrote, “have yielded a raft of potential new therapeutics, centered on naive T-cell depletion, interleukin-17/21 inhibition, kinase inhibition, regulatory T-cell restoration, and CSF-1 inhibition.”
For now, no agents other than corticosteroids have shown benefit in cGVHD as initial therapy, Dr. Flowers said. In fact, several trials closed early from lack of benefit. But trials continue, she said: Results are pending for a completed phase 3 trial of ibrutinib, a Bruton tyrosine kinase inhibitor, plus steroids for initial treatment of cGVHD. Nearly 500 patients were enrolled, she said. And there’s an ongoing phase 2/3 trial of itacitinib, a Janus kinase 1 inhibitor plus steroids as initial treatment.
Dr. Flowers highlighted the case of a patient with moderate cGVHD. The patient was treated with infection prophylaxis, supportive care for oral and eyes manifestations, and prednisone 0.5 mg/kg (at a lower dose because of diabetes) plus a substitution of tacrolimus with sirolimus, a calcineurin inhibitor.
Why sirolimus? At this early point in progression, she said, the patient didn’t necessarily need systemwide chemo-suppression, and calcineurin inhibitors can be “quite effective” in management of inflammation in the liver. “It would be a completely different story once the patient develops severe cGVHR.” In that case, she said, calcineurin inhibitors wouldn’t be appropriate.
The patient’s status deteriorated to severe cGVHD, and sirolimus was replaced with ibrutinib. Other drugs were added to prevent infection and treat bronchiolitis obliterans syndrome.
In general, “the goal of the treatment is get adequate control of clinical manifestations and prevent more severe disease from developing,” Dr. Flowers said.
In response to a question about polypharmacy in patient with advanced disease – “we tend not to peel those drugs off” – Dr. Flowers said she does see new patients who appear to be taking too many medications. “They are on five drugs, and I say, ‘What are we doing?’ ”
Quite often, Dr. Flowers said, she doesn’t add therapies to existing ones but instead looks for substitutes. “A clinical lesson that I feel that I learned over time is: Ask your questions first. What would you like to see in 3 months? Or 6 months? Before you just add another therapy, do you really know what the trajectory of a disease might be?”
Dr. Flowers discloses research support (Pharmacyclics, Incyte), speaker honorarium (Janssen, Johnson & Johnson, Astellas, Mallinckrodt), and consulting relationships (Pharmacyclics, CSL Behring, Fresenius Kabi).
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
FROM ALF 2020
Final ASCEND study data: Acalabrutinib beat standard of care for r/r CLL
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
FROM SOHO 2020
Are oncologists ready to confront a second wave of COVID-19?
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
Canceled appointments, postponed surgeries, and delayed cancer diagnoses – all are a recipe for exhaustion for oncologists around the world, struggling to reach and treat their patients during the pandemic. Physicians and their teams felt the pain as COVID-19 took its initial march around the globe.
“We saw the distress of people with cancer who could no longer get to anyone on the phone. Their medical visit was usually canceled. Their radiotherapy session was postponed or modified, and chemotherapy postponed,” says Axel Kahn, MD, chairman of the board of directors of La Ligue Nationale Contre le Cancer (National League Against Cancer). “In the vast majority of cases, cancer treatment can be postponed or readjusted, without affecting the patient’s chances of survival, but there has been a lot of anxiety because the patients do not know that.”
The stay-at-home factor was one that played out across many months during the first wave.
“I believe that the ‘stay-home’ message that we transmitted was rigorously followed by patients who should have come to the emergency room much earlier and who, therefore, were admitted with a much more deteriorated general condition than in non-COVID-19 times,” says Benjamín Domingo Arrué, MD, from the department of medical oncology at Hospital Universitari i Politècnic La Fe in Valencia, Spain.
And in Brazil, some of the impact from the initial hit of COVID-19 on oncology is only now being felt, according to Laura Testa, MD, head of breast medical oncology, Instituto do Câncer do Estado de São Paulo.
“We are starting to see a lot of cancer cases that didn’t show up at the beginning of the pandemic, but now they are arriving to us already in advanced stages,” she said. “These patients need hospital care. If the situation worsens and goes back to what we saw at the peak of the curve, I fear the public system won’t be able to treat properly the oncology patients that need hospital care and the patients with cancer who also have COVID-19.”
But even as health care worker fatigue and concerns linger, oncologists say that what they have learned in the last 6 months has helped them prepare as COVID-19 cases increase and a second global wave kicks up.
Lessons from the first wave
In the United States, COVID-19 hit different regions at different times and to different degrees. One of the areas hit first was Seattle.
“We jumped on top of this, we were evidence based, we put things in place very, very quickly,” said Julie Gralow, MD, professor at the University of Washington and the Fred Hutchinson Cancer Research Center, both in Seattle.
“We did a really good job keeping COVID out of our cancer centers,” Dr. Gralow said. “We learned how to be super safe, and to keep symptomatic people out of the building, and to limit the extra people they could bring with them. It’s all about the number of contacts you have.”
The story was different, though, for oncologists in several other countries, and sometimes it varied immensely within each nation.
“We treated fewer patients with cancer during the first wave,” says Dirk Arnold, MD, medical director of the Asklepios Tumor Center Hamburg (Germany), in an interview. “In part, this was because staff were quarantined and because we had a completely different infrastructure in all of the hospitals. But also fewer patients with cancer came to the clinic at all. A lot of resources were directed toward COVID-19.”
In Spain, telemedicine helped keep up with visits, but other areas felt the effect of COVID-19 patient loads.
“At least in the oncology department of our center, we have practically maintained 100% of visits, mostly by telephone,” says Dr. Arrué, “but the reality is that our country has not yet been prepared for telemedicine.”
Laura Mezquita, MD, of the department of medical oncology at Hospital Clinic de Barcelona, describes a more dramatic situation: “We have seen how some of our patients, especially with metastatic disease, have been dismissed for intensive care and life-support treatments, as well as specific treatments against COVID-19 (tocilizumab, remdesivir, etc.) due to the general health collapse of the former wave,” she said. She adds that specific oncologic populations, such as those with thoracic tumors, have been more affected.
Distress among oncologists
Many oncologists are still feeling stressed and fatigued after the first wave, just as a second string of outbreaks is on its way.
A survey presented at last month’s ESMO 2020 Congress found that, in July-August, moral distress was reported by one-third of the oncologists who responded, and more than half reported a feeling of exhaustion.
“The tiredness and team exhaustion is noticeable,” said Dr. Arnold. “We recently had a task force discussion about what will happen when we have a second wave and how the department and our services will adapt. It was clear that those who were at the very front in the first wave had only a limited desire to do that again in the second wave.”
Another concern: COVID-19’s effect on staffing levels.
“We have a population of young caregivers who are affected by the COVID-19 disease with an absenteeism rate that is quite unprecedented,” said Sophie Beaupère, general delegate of Unicancer since January.
She said that, in general, the absenteeism rate in the cancer centers averages 5%-6%, depending on the year. But that rate is now skyrocketing.
Stop-start cycle for surgery
As caregivers quarantined around the world, more than 10% of patients with cancer had treatment canceled or delayed during the first wave of the pandemic, according to another survey from ESMO, involving 109 oncologists from 18 countries.
Difficulties were reported for surgeries by 34% of the centers, but also difficulties with delivering chemotherapy (22% of centers), radiotherapy (13.7%), and therapy with checkpoint inhibitors (9.1%), monoclonal antibodies (9%), and oral targeted therapy (3.7%).
Stopping surgery is a real concern in France, noted Dr. Kahn, the National League Against Cancer chair. He says that in regions that were badly hit by COVID-19, “it was not possible to have access to the operating room for people who absolutely needed surgery; for example, patients with lung cancer that was still operable. Most of the recovery rooms were mobilized for resuscitation.”
There may be some solutions, suggested Thierry Breton, director general of the National Institute of Cancer in France. “We are getting prepared, with the health ministry, for a possible increase in hospital tension, which would lead to a situation where we would have to reschedule operations. Nationally, regionally, and locally, we are seeing how we can resume and prioritize surgeries that have not been done.”
Delays in cancer diagnosis
While COVID-19 affected treatment, many oncologists say the major impact of the first wave was a delay in diagnosing cancer. Some of this was a result of the suspension of cancer screening programs, but there was also fear among the general public about visiting clinics and hospitals during a pandemic.
“We didn’t do so well with cancer during the first wave here in the U.K.,” said Karol Sikora, PhD, MBBChir, professor of cancer medicine and founding dean at the University of Buckingham Medical School, London. “Cancer diagnostic pathways virtually stalled partly because patients didn’t seek help, but getting scans and biopsies was also very difficult. Even patients referred urgently under the ‘2-weeks-wait’ rule were turned down.”
In France, “the delay in diagnosis is indisputable,” said Dr. Kahn. “About 50% of the cancer diagnoses one would expect during this period were missed.”
“I am worried that there remains a major traffic jam that has not been caught up with, and, in the meantime, the health crisis is worsening,” he added.
In Seattle, Dr. Gralow said the first COVID-19 wave had little impact on treatment for breast cancer, but it was in screening for breast cancer “where things really got messed up.”
“Even though we’ve been fully ramped up again,” she said, concerns remain. To ensure that screening mammography is maintained, “we have spaced out the visits to keep our waiting rooms less populated, with a longer time between using the machine so we can clean it. To do this, we have extended operating hours and are now opening on Saturday.
“So we’re actually at 100% of our capacity, but I’m really nervous, though, that a lot of people put off their screening mammogram and aren’t going to come in and get it.
“Not only did people get the message to stay home and not do nonessential things, but I think a lot of people lost their health insurance when they lost their jobs,” she said, and without health insurance, they are not covered for cancer screening.
Looking ahead, with a plan
Many oncologists agree that access to care can and must be improved – and there were some positive moves.
“Some regimens changed during the first months of the pandemic, and I don’t see them going back to the way they were anytime soon,” said Dr. Testa. “The changes/adaptations that were made to minimize the chance of SARS-CoV-2 infection are still in place and will go on for a while. In this context, telemedicine helped a lot. The pandemic forced the stakeholders to step up and put it in place in March. And now it’s here to stay.”
The experience gained in the last several months has driven preparation for the next wave.
“We are not going to see the disorganization that we saw during the first wave,” said Florence Joly, MD, PhD, head of medical oncology at the Centre François Baclesse in Caen, France. “The difference between now and earlier this year is that COVID diagnostic tests are available. That was one of the problems in the first wave. We had no way to diagnose.”
On the East Coast of the United States, medical oncologist Charu Aggarwal, MD, MPH, is also optimistic: “I think we’re at a place where we can manage.”
“I believe if there was going to be a new wave of COVID-19 cases we would be: better psychologically prepared and better organized,” said Dr. Aggarwal, assistant professor of medicine in the hematology-oncology division at the University of Pennsylvania, Philadelphia. “We already have experience with all of the tools, we have telemedicine available, we have screening protocols available, we have testing, we are already universally masking, everyone’s hand-washing, so I do think that means we would be okay.”
Dr. Arnold agreed that “we are much better prepared than for the first wave, but … we have immense tasks in the area of patient management, the digitization of patient care, the clear allocation of resources when there is a second or third wave. In many areas of preparation, I believe, unfortunately, we are not as well positioned as we had actually hoped.”
The first wave of COVID hit cancer services in the United Kingdom particularly hard: One modeling study suggested that delays in cancer referrals will lead to thousands of additional deaths and tens of thousands of life-years lost.
“Cancer services are working at near normal levels now, but they are still fragile and could be severely compromised again if the NHS [National Health Service] gets flooded by COVID patients,” said Dr. Sikora.
The second wave may be different. “Although the number of infections has increased, the hospitalizations have only risen a little. Let’s see what happens,” he said in an interview. Since then, however, infections have continued to rise, and there has been an increase in hospitalizations. New social distancing measures in the United Kingdom were put into place on Oct. 12, with the aim of protecting the NHS from overload.
Dr. Arrué describes it this way: “The reality is that the ‘second wave’ has left behind the initial grief and shock that both patients and health professionals experienced when faced with something that, until now, we had only seen in the movies.” The second wave has led to new restrictions – including a partial lockdown since the beginning of October.
Dr. Aggarwal says her department recently had a conference with Anthony Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, about the impact of COVID-19 on oncology.
“I asked him what advice he’d give oncologists, and he said to go back to as much screening as you were doing previously as quickly as possible. That’s what must be relayed to our oncologists in the community – and also to primary care physicians – because they are often the ones who are ordering and championing the screening efforts.”
This article was originated by Aude Lecrubier, Medscape French edition, and developed by Zosia Chustecka, Medscape Oncology. With additional reporting by Kate Johnson, freelance medical journalist, Claudia Gottschling for Medscape Germany, Leoleli Schwartz for Medscape em português, Tim Locke for Medscape United Kingdom, and Carla Nieto Martínez, freelance medical journalist for Medscape Spanish edition.
This article first appeared on Medscape.com.
ESMO offers new clinical practice guideline for CLL
An updated European Society for Medical Oncology (ESMO) clinical practice guidelines were released to provide key recommendations on the management of chronic lymphocytic leukemia (CLL).
The guidelines were developed by a multidisciplinary group of experts from different institutions and countries in Europe and provide levels of evidence and grades of recommendation where applicable for issues regarding prognosis and treatment decisions in CLL. Such decisions depend on genetic and clinical factors such as age, stage, and comorbidities. The guidelines also focus on new therapies targeting B-cell-receptor pathways or defect mechanism of apoptosis, which have been found to induce long-lasting remissions. The guidelines were endorsed by the European Hematology Association (EHA) through the Scientific Working Group on CLL/European Research Initiative on CLL (ERIC), according to the report published online the Annals of Oncology.
These clinical practice guidelines were developed in accordance with the ESMO standard operating procedures for clinical practice guidelines development with use of relevant literature selected by the expert authors. Statements without grading were considered justified as standard clinical practice by the experts and the ESMO faculty.
Below are some highlights of the guidelines, which cover a wide array of topics regarding the diagnosis, staging, treatment, and progression of CLL disease.
Diagnosis
The guidelines indicate that CLL diagnosis is usually possible by immunophenotyping of peripheral blood only and that lymph node (LN) biopsy and/or bone marrow biopsy may be helpful if immunophenotyping is not conclusive for the diagnosis of CLL, according to Barbara Eichhorst, MD, of the University of Cologne (Germany) and colleagues on behalf of the ESMO guidelines committee.
Staging and risk assessment
Early asymptomatic-stage disease does not need further risk assessment, but after the first year, when all patients should be seen at 3-monthly intervals, patients can be followed every 3-12 months. The interval would depend on burden and dynamics of the disease obtained by the using history and physical examinations, including a careful palpation of all LN areas, spleen, and liver, as well as assessing complete blood cell count and differential count, according to the report.
Advanced- and symptomatic-stage disease requires a broader examination including imaging, history and status of relevant infections, and fluorescent in situ hybridization (FISH) assays for the detection of deletion of the chromosome 17 (del[17p]) affecting the tumor protein p53 expression and, in the absence of del(17p), TP53 sequencing for detection of TP53 gene mutation, according to the authors.
Prognostication
Two clinical staging systems are typically used in CLL. Both Binet and Rai staging systems separate three groups of patients with different prognosis, although “as a consequence of more effective therapy, the overall survival (OS) of patients with advanced stage has improved and the relevance of the staging systems for prognostication has decreased,” according to the report.
The recent addition of genetic markers has also proved highly relevant to identifying patients with different prognoses and to guide treatment.
Therapy
Although CLL is an incurable disease, choice and application of treatment are strongly tied to the length of survival, according to the authors. The guidelines recommend Binet and Rai staging with clinical symptoms as relevant for treatment indication. In addition, the identification of del(17p), TP53 mutations, and IGHV status are relevant for choice of therapy and should be assessed prior to treatment.
The guidelines discuss specific treatment modalities for various stages of the disease, from early stages to relapse.
For frontline therapy, different treatment strategies are available including continuous treatment with Bruton tyrosine kinase (BTK)–inhibitors, such as ibrutinib, until progression or time-limited therapy with ChT backbone and CD20 antibodies. In addition, the Food and Drug Administration and European Medicines Agency have recently approved the combination of venetoclax plus obinutuzumab for first-line therapy of CLL.
Treatment decisions should include an assessment of IGHV and TP53 status, as well as patient-related factors such as comedication, comorbidities, preferences, drug availability, and potential of treatment adherence, according to the guidelines.
In case of symptomatic relapse within 3 years after fixed-duration therapy or nonresponse to therapy, the guidelines recommend that the therapeutic regimen should be changed, regardless of the type of first-line either to venetoclax plus rituximab for 24 months or to ibrutinib, acalabrutinib, or other BTK inhibitors (if available) as continuous therapy.
The guidelines also discuss the possible roles for hematopoietic stem cell transplantation and cellular therapies, as well as the treatment of the various complications that can arise in patients with CLL, and dealing with various aspects of disease progression.
No external funds were provided for the production of the guidelines. The authors of the report and members of the ESMO Guidelines Committee reported numerous disclosures regarding pharmaceutical and biotechnology companies.
SOURCE: Eichhorst B et al. Ann Oncol. 2020 Oct 19. doi: 10.1016/j.annonc.2020.09.019.
An updated European Society for Medical Oncology (ESMO) clinical practice guidelines were released to provide key recommendations on the management of chronic lymphocytic leukemia (CLL).
The guidelines were developed by a multidisciplinary group of experts from different institutions and countries in Europe and provide levels of evidence and grades of recommendation where applicable for issues regarding prognosis and treatment decisions in CLL. Such decisions depend on genetic and clinical factors such as age, stage, and comorbidities. The guidelines also focus on new therapies targeting B-cell-receptor pathways or defect mechanism of apoptosis, which have been found to induce long-lasting remissions. The guidelines were endorsed by the European Hematology Association (EHA) through the Scientific Working Group on CLL/European Research Initiative on CLL (ERIC), according to the report published online the Annals of Oncology.
These clinical practice guidelines were developed in accordance with the ESMO standard operating procedures for clinical practice guidelines development with use of relevant literature selected by the expert authors. Statements without grading were considered justified as standard clinical practice by the experts and the ESMO faculty.
Below are some highlights of the guidelines, which cover a wide array of topics regarding the diagnosis, staging, treatment, and progression of CLL disease.
Diagnosis
The guidelines indicate that CLL diagnosis is usually possible by immunophenotyping of peripheral blood only and that lymph node (LN) biopsy and/or bone marrow biopsy may be helpful if immunophenotyping is not conclusive for the diagnosis of CLL, according to Barbara Eichhorst, MD, of the University of Cologne (Germany) and colleagues on behalf of the ESMO guidelines committee.
Staging and risk assessment
Early asymptomatic-stage disease does not need further risk assessment, but after the first year, when all patients should be seen at 3-monthly intervals, patients can be followed every 3-12 months. The interval would depend on burden and dynamics of the disease obtained by the using history and physical examinations, including a careful palpation of all LN areas, spleen, and liver, as well as assessing complete blood cell count and differential count, according to the report.
Advanced- and symptomatic-stage disease requires a broader examination including imaging, history and status of relevant infections, and fluorescent in situ hybridization (FISH) assays for the detection of deletion of the chromosome 17 (del[17p]) affecting the tumor protein p53 expression and, in the absence of del(17p), TP53 sequencing for detection of TP53 gene mutation, according to the authors.
Prognostication
Two clinical staging systems are typically used in CLL. Both Binet and Rai staging systems separate three groups of patients with different prognosis, although “as a consequence of more effective therapy, the overall survival (OS) of patients with advanced stage has improved and the relevance of the staging systems for prognostication has decreased,” according to the report.
The recent addition of genetic markers has also proved highly relevant to identifying patients with different prognoses and to guide treatment.
Therapy
Although CLL is an incurable disease, choice and application of treatment are strongly tied to the length of survival, according to the authors. The guidelines recommend Binet and Rai staging with clinical symptoms as relevant for treatment indication. In addition, the identification of del(17p), TP53 mutations, and IGHV status are relevant for choice of therapy and should be assessed prior to treatment.
The guidelines discuss specific treatment modalities for various stages of the disease, from early stages to relapse.
For frontline therapy, different treatment strategies are available including continuous treatment with Bruton tyrosine kinase (BTK)–inhibitors, such as ibrutinib, until progression or time-limited therapy with ChT backbone and CD20 antibodies. In addition, the Food and Drug Administration and European Medicines Agency have recently approved the combination of venetoclax plus obinutuzumab for first-line therapy of CLL.
Treatment decisions should include an assessment of IGHV and TP53 status, as well as patient-related factors such as comedication, comorbidities, preferences, drug availability, and potential of treatment adherence, according to the guidelines.
In case of symptomatic relapse within 3 years after fixed-duration therapy or nonresponse to therapy, the guidelines recommend that the therapeutic regimen should be changed, regardless of the type of first-line either to venetoclax plus rituximab for 24 months or to ibrutinib, acalabrutinib, or other BTK inhibitors (if available) as continuous therapy.
The guidelines also discuss the possible roles for hematopoietic stem cell transplantation and cellular therapies, as well as the treatment of the various complications that can arise in patients with CLL, and dealing with various aspects of disease progression.
No external funds were provided for the production of the guidelines. The authors of the report and members of the ESMO Guidelines Committee reported numerous disclosures regarding pharmaceutical and biotechnology companies.
SOURCE: Eichhorst B et al. Ann Oncol. 2020 Oct 19. doi: 10.1016/j.annonc.2020.09.019.
An updated European Society for Medical Oncology (ESMO) clinical practice guidelines were released to provide key recommendations on the management of chronic lymphocytic leukemia (CLL).
The guidelines were developed by a multidisciplinary group of experts from different institutions and countries in Europe and provide levels of evidence and grades of recommendation where applicable for issues regarding prognosis and treatment decisions in CLL. Such decisions depend on genetic and clinical factors such as age, stage, and comorbidities. The guidelines also focus on new therapies targeting B-cell-receptor pathways or defect mechanism of apoptosis, which have been found to induce long-lasting remissions. The guidelines were endorsed by the European Hematology Association (EHA) through the Scientific Working Group on CLL/European Research Initiative on CLL (ERIC), according to the report published online the Annals of Oncology.
These clinical practice guidelines were developed in accordance with the ESMO standard operating procedures for clinical practice guidelines development with use of relevant literature selected by the expert authors. Statements without grading were considered justified as standard clinical practice by the experts and the ESMO faculty.
Below are some highlights of the guidelines, which cover a wide array of topics regarding the diagnosis, staging, treatment, and progression of CLL disease.
Diagnosis
The guidelines indicate that CLL diagnosis is usually possible by immunophenotyping of peripheral blood only and that lymph node (LN) biopsy and/or bone marrow biopsy may be helpful if immunophenotyping is not conclusive for the diagnosis of CLL, according to Barbara Eichhorst, MD, of the University of Cologne (Germany) and colleagues on behalf of the ESMO guidelines committee.
Staging and risk assessment
Early asymptomatic-stage disease does not need further risk assessment, but after the first year, when all patients should be seen at 3-monthly intervals, patients can be followed every 3-12 months. The interval would depend on burden and dynamics of the disease obtained by the using history and physical examinations, including a careful palpation of all LN areas, spleen, and liver, as well as assessing complete blood cell count and differential count, according to the report.
Advanced- and symptomatic-stage disease requires a broader examination including imaging, history and status of relevant infections, and fluorescent in situ hybridization (FISH) assays for the detection of deletion of the chromosome 17 (del[17p]) affecting the tumor protein p53 expression and, in the absence of del(17p), TP53 sequencing for detection of TP53 gene mutation, according to the authors.
Prognostication
Two clinical staging systems are typically used in CLL. Both Binet and Rai staging systems separate three groups of patients with different prognosis, although “as a consequence of more effective therapy, the overall survival (OS) of patients with advanced stage has improved and the relevance of the staging systems for prognostication has decreased,” according to the report.
The recent addition of genetic markers has also proved highly relevant to identifying patients with different prognoses and to guide treatment.
Therapy
Although CLL is an incurable disease, choice and application of treatment are strongly tied to the length of survival, according to the authors. The guidelines recommend Binet and Rai staging with clinical symptoms as relevant for treatment indication. In addition, the identification of del(17p), TP53 mutations, and IGHV status are relevant for choice of therapy and should be assessed prior to treatment.
The guidelines discuss specific treatment modalities for various stages of the disease, from early stages to relapse.
For frontline therapy, different treatment strategies are available including continuous treatment with Bruton tyrosine kinase (BTK)–inhibitors, such as ibrutinib, until progression or time-limited therapy with ChT backbone and CD20 antibodies. In addition, the Food and Drug Administration and European Medicines Agency have recently approved the combination of venetoclax plus obinutuzumab for first-line therapy of CLL.
Treatment decisions should include an assessment of IGHV and TP53 status, as well as patient-related factors such as comedication, comorbidities, preferences, drug availability, and potential of treatment adherence, according to the guidelines.
In case of symptomatic relapse within 3 years after fixed-duration therapy or nonresponse to therapy, the guidelines recommend that the therapeutic regimen should be changed, regardless of the type of first-line either to venetoclax plus rituximab for 24 months or to ibrutinib, acalabrutinib, or other BTK inhibitors (if available) as continuous therapy.
The guidelines also discuss the possible roles for hematopoietic stem cell transplantation and cellular therapies, as well as the treatment of the various complications that can arise in patients with CLL, and dealing with various aspects of disease progression.
No external funds were provided for the production of the guidelines. The authors of the report and members of the ESMO Guidelines Committee reported numerous disclosures regarding pharmaceutical and biotechnology companies.
SOURCE: Eichhorst B et al. Ann Oncol. 2020 Oct 19. doi: 10.1016/j.annonc.2020.09.019.
FROM ANNALS OF ONCOLOGY
Can AML patients be too old for cell transplantation?
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
How old is too old for a patient to undergo hematopoietic cell transplantation (HCT)? That’s the wrong question to ask, a hematologist/oncologist told colleagues at the virtual Acute Leukemia Forum of Hemedicus. Instead, he said, look at other factors such as disease status and genetics.
“Transplantation for older patients, even beyond the age of 70, is acceptable, as long as it’s done with caution, care, and wisdom. So we’re all not too old for transplantation, at least not today,” said Daniel Weisdorf, MD, professor of medicine and deputy director of the University of Minnesota Clinical and Translational Science Institute.
As he noted, acute myeloid leukemia (AML) is often fatal. Among the general population, “the expected survival life expectancy at age 75 is 98% at 1 year, and most people living at 75 go on to live more than 10 years,” he said. “But if you have AML, at age 75, you have 20% survival at 1 year, 4% at 3 years. And since the median age of AML diagnosis is 68, and 75% of patients are diagnosed beyond the age of 55, this becomes relevant.”
Risk factors that affect survival after transplantation “certainly include age, but that interacts directly with the comorbidities people accumulate with age, their assessments of frailty, and their Karnofsky performance status, as well as the disease phenotype and molecular genetic markers,” Dr. Weisdorf said. “Perhaps most importantly, though not addressed very much, is patients’ willingness to undertake intensive therapy and their life outlook related to patient-reported outcomes when they get older.”
Despite the lack of indications that higher age by itself is an influential factor in survival after transplant, “we are generally reluctant to push the age of eligibility,” Dr. Weisdorf said. He noted that recently published American Society of Hematology guidelines for treatment of AML over the age of 55 “don’t discuss anything about transplantation fitness because they didn’t want to tackle that.”
Overall survival (OS) at 1 year after allogenic transplants only dipped slightly from ages 51-60 to 71 and above, according to Dr. Weisdorf’s analysis of U.S. data collected by the Center for International Blood and Marrow Transplant Research for the time period 2005-2019.
OS was 67.6% (66.8%-68.3%) for the 41-50 age group (n = 9,287) and 57.9% (56.1%-59.8%) for the 71 and older group, Dr. Weisdorf found. Overall, OS dropped by about 4 percentage points per decade of age, he said, revealing a “modest influence” of advancing years.
His analysis of autologous transplant data from the same source, also for 2005-2019, revealed “essentially no age influence.” OS was 90.8% (90.3%-91.2%) for the 41-50 age group (n = 15,075) and 86.6% (85.9%-87.3%) for the 71 and older group (n = 7,247).
Dr. Weisdorf also highlighted unpublished research that suggests that cord-blood transplant recipients older than 70 face a significantly higher risk of death than that of younger patients in the same category. Cord blood “may be option of last resort” because of a lack of other options, he explained. “And it may be part of the learning curve of cord blood transplantation, which grew a little bit in the early 2000s, and maybe past 2010, and then fell off as everybody got enamored with the haploidentical transplant option.”
How can physicians make decisions about transplants in older patients? “The transplant comorbidity index, the specific comorbidities themselves, performance score, and frailty are all measures of somebody’s fitness to be a good candidate for transplant, really at any age,” Dr. Weisdorf said. “But we also have to recognize that disease status, genetics, and the risk phenotype remain critical and should influence decision making.”
However, even as transplant survival improves overall, “very few people are incorporating any very specific biological markers” in decision-making, he said. “We’ve gotten to measures of frailty, but we haven’t gotten to any biologic measures of cytokines or other things that would predict poor chances for doing well. So I’m afraid we’re still standing at the foot of the bed saying: ‘You look okay.’ Or we’re measuring their comorbidity index. But it is disappointing that we’re using mostly very simple clinical measures to decide if somebody is sturdy enough to proceed, and we perhaps need something better. But I don’t have a great suggestion what it should be.”
The Acute Leukemia Forum is held by Hemedicus, which is owned by the same company as this news organization.
Dr. Weisdorf disclosed consulting fees from Fate Therapeutics and Incyte Corp.
SOURCE: “The Ever-Increasing Upper Age for Transplant: Is This Evidence-Based?” Acute Leukemia Forum of Hemedicus, Oct. 15, 2020.
FROM ALF 2020
Study: Complications from childhood ALL and its treatment are common, but can be managed
Despite survival after treatment of acute lymphoblastic leukemia (ALL), a high percentage of children suffered acute complications, even without relapse, according to a report published online in Clinical Lymphoma, Myeloma & Leukemia.
In a retrospective study of 110 children with acute lymphoblastic leukemia (ALL), Ayse Pınar Öztürk, MD, and colleagues at Istanbul University, Cerrahpasa Faculty of Medicine, evaluated the acute complications that occurred during the treatment of childhood ALL and documented their survival rates. The 110 patients, comprising 65 boys and 45 girls, were all treated with the Children’s Oncology Group protocol from 1999 to 2014.
The mean age at admission was 8.3 years and 97 patients (88.2%) were diagnosed with pre–B-cell ALL, 11 (10%) with T-cell ALL, 1 (0.9%) with mixed phenotype acute leukemia, and 1 (0.9%) with mature B-cell acute leukemia. A total of 36.3% were evaluated to be in the standard-risk group and the rest were in the high-risk group. Regular follow-up and evaluation for acute complications was available for 105 of the patients.
Survival and complications
Of the 110 patients, 98 were assessed in the survival analyses. The 5- and 10-year overall survival rates were both 85.9%, while the relapse-free survival rates at 1, 3, and 5 years were 97.9%, 91.3%, and 86.3%, respectively. These results are favorable and in line with good results reported in the literature, according to the researchers.
In terms of acute complications, infection was the most common (88.5%), followed by gastrointestinal (27.6%), neurologic (26.6%), metabolic/endocrine (15.2%), drug-related hypersensitivity (15.2%), avascular necrosis (12.3%), thrombotic (10.4%), severe psychiatric (1.9%), and various other complications (11.4%).
In the present study, 13 of the 98 patients (13.3%) died. All 13 patients had been in the high-risk group and 9 had had relapsed ALL. Of the 13 deaths, 8 (8.2%) had resulted from treatment resistance and toxicity and 5 (5.1%) from severe infection (sepsis).
During ALL treatment, various complications can occur related to the disease itself or the treatment, according to the authors. However, they added that in regularly and closely monitored patients, complications can be effectively prevented, treated, and eliminated by aggressive observation and prompt intervention.
“In our study, the short hospitalization period, prompt implementation of protocol updates, rapid analysis of laboratory tests, continuous supportive care, efficient education given to the parents of children, and consistently undertaking patient care and treatment management by the same expert team increased the success of the therapy and ensured low complication rates,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Öztürk AP et al. Clin Lymphoma Myeloma Leuk. 2020 Sep 17. doi: 10.1016/j.clml.2020.08.025.
Despite survival after treatment of acute lymphoblastic leukemia (ALL), a high percentage of children suffered acute complications, even without relapse, according to a report published online in Clinical Lymphoma, Myeloma & Leukemia.
In a retrospective study of 110 children with acute lymphoblastic leukemia (ALL), Ayse Pınar Öztürk, MD, and colleagues at Istanbul University, Cerrahpasa Faculty of Medicine, evaluated the acute complications that occurred during the treatment of childhood ALL and documented their survival rates. The 110 patients, comprising 65 boys and 45 girls, were all treated with the Children’s Oncology Group protocol from 1999 to 2014.
The mean age at admission was 8.3 years and 97 patients (88.2%) were diagnosed with pre–B-cell ALL, 11 (10%) with T-cell ALL, 1 (0.9%) with mixed phenotype acute leukemia, and 1 (0.9%) with mature B-cell acute leukemia. A total of 36.3% were evaluated to be in the standard-risk group and the rest were in the high-risk group. Regular follow-up and evaluation for acute complications was available for 105 of the patients.
Survival and complications
Of the 110 patients, 98 were assessed in the survival analyses. The 5- and 10-year overall survival rates were both 85.9%, while the relapse-free survival rates at 1, 3, and 5 years were 97.9%, 91.3%, and 86.3%, respectively. These results are favorable and in line with good results reported in the literature, according to the researchers.
In terms of acute complications, infection was the most common (88.5%), followed by gastrointestinal (27.6%), neurologic (26.6%), metabolic/endocrine (15.2%), drug-related hypersensitivity (15.2%), avascular necrosis (12.3%), thrombotic (10.4%), severe psychiatric (1.9%), and various other complications (11.4%).
In the present study, 13 of the 98 patients (13.3%) died. All 13 patients had been in the high-risk group and 9 had had relapsed ALL. Of the 13 deaths, 8 (8.2%) had resulted from treatment resistance and toxicity and 5 (5.1%) from severe infection (sepsis).
During ALL treatment, various complications can occur related to the disease itself or the treatment, according to the authors. However, they added that in regularly and closely monitored patients, complications can be effectively prevented, treated, and eliminated by aggressive observation and prompt intervention.
“In our study, the short hospitalization period, prompt implementation of protocol updates, rapid analysis of laboratory tests, continuous supportive care, efficient education given to the parents of children, and consistently undertaking patient care and treatment management by the same expert team increased the success of the therapy and ensured low complication rates,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Öztürk AP et al. Clin Lymphoma Myeloma Leuk. 2020 Sep 17. doi: 10.1016/j.clml.2020.08.025.
Despite survival after treatment of acute lymphoblastic leukemia (ALL), a high percentage of children suffered acute complications, even without relapse, according to a report published online in Clinical Lymphoma, Myeloma & Leukemia.
In a retrospective study of 110 children with acute lymphoblastic leukemia (ALL), Ayse Pınar Öztürk, MD, and colleagues at Istanbul University, Cerrahpasa Faculty of Medicine, evaluated the acute complications that occurred during the treatment of childhood ALL and documented their survival rates. The 110 patients, comprising 65 boys and 45 girls, were all treated with the Children’s Oncology Group protocol from 1999 to 2014.
The mean age at admission was 8.3 years and 97 patients (88.2%) were diagnosed with pre–B-cell ALL, 11 (10%) with T-cell ALL, 1 (0.9%) with mixed phenotype acute leukemia, and 1 (0.9%) with mature B-cell acute leukemia. A total of 36.3% were evaluated to be in the standard-risk group and the rest were in the high-risk group. Regular follow-up and evaluation for acute complications was available for 105 of the patients.
Survival and complications
Of the 110 patients, 98 were assessed in the survival analyses. The 5- and 10-year overall survival rates were both 85.9%, while the relapse-free survival rates at 1, 3, and 5 years were 97.9%, 91.3%, and 86.3%, respectively. These results are favorable and in line with good results reported in the literature, according to the researchers.
In terms of acute complications, infection was the most common (88.5%), followed by gastrointestinal (27.6%), neurologic (26.6%), metabolic/endocrine (15.2%), drug-related hypersensitivity (15.2%), avascular necrosis (12.3%), thrombotic (10.4%), severe psychiatric (1.9%), and various other complications (11.4%).
In the present study, 13 of the 98 patients (13.3%) died. All 13 patients had been in the high-risk group and 9 had had relapsed ALL. Of the 13 deaths, 8 (8.2%) had resulted from treatment resistance and toxicity and 5 (5.1%) from severe infection (sepsis).
During ALL treatment, various complications can occur related to the disease itself or the treatment, according to the authors. However, they added that in regularly and closely monitored patients, complications can be effectively prevented, treated, and eliminated by aggressive observation and prompt intervention.
“In our study, the short hospitalization period, prompt implementation of protocol updates, rapid analysis of laboratory tests, continuous supportive care, efficient education given to the parents of children, and consistently undertaking patient care and treatment management by the same expert team increased the success of the therapy and ensured low complication rates,” the researchers concluded.
The authors reported that they had no conflicts of interest.
SOURCE: Öztürk AP et al. Clin Lymphoma Myeloma Leuk. 2020 Sep 17. doi: 10.1016/j.clml.2020.08.025.
FROM Clinical Lymphoma, Myeloma & Leukemia