User login
Combination treatments a must for the war on warts
When counseling patients with warts, Adam Friedman, MD, admits that he feels like a character from “Game of Thrones” since many treatment options are “medieval and painful,” from duct tape occlusion to the stings of liquid nitrogen and salicylic acid.
“We can combine destructive, immunologic, and cytotoxic approaches,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic & Surgical Conference. “It’s not one or the other, we want to be aggressive.
At the initial clinic visit, he advises asking patients how long the warts have been present, because sometimes they will go away within a year or two without treatment. “If someone says, ‘I’ve had these for years,’ you know you’re in for the long haul and you have to be aggressive with their therapy,” Dr. Friedman said. “Sometimes you’ll pick up plantar warts on a full-body skin exam and the patient may say, ‘I really don’t care. Please don’t touch them,’ so it’s important to understand how they are impacting quality of life.”
Patients should also be asked what treatments they have used previously, and it is important to set some realistic expectations and dispel some myths, Dr. Friedman said. “One of the most important things is that you must get these patients back. This is not often a one and done approach; you need to keep hitting them [with therapy], because if you let one infected keratinocyte survive, it’s going to come back and it’s still going to be contagious – more likely for that patient than for anyone else.”
The application of liquid nitrogen is a popular, inexpensive destructive treatment option, with spray canisters that cost about $600. “You have to consider the temperature of the liquid nitrogen spray because melanocytes die at negative 5 degrees Celsius, so you have to be mindful in patients with darker skin tones that you may leave with permanent dyschromia, meaning hypopigmentation or depigmentation when you do this,” he said. Because it is painful, “we’re limited when it comes to treating children with warts who are younger than 9 or 10. I don’t think the Q-tip method or dipping a hemostat in cryogen and touching the tip really works. You’ve got to create a nice ice ball that thaws and kills the infected keratinocytes.”
Dr. Friedman favors a 10-second freeze of the wart, usually for two to three cycles depending on its anatomic location, and he may give patients imiquimod or 5-FU to use at home for 5 nights of the week. A recently published study found that the use of ultrasound gel increases the efficacy of cryotherapy in the treatment of warts.
Another destructive treatment approach is cantharidin 0.7% applied topically in the office. It is believed to activate neutral serine proteases that cause degeneration of the desmosomal plaque, leading to detachment of tonofilaments from desmosomes. Repeat in-office applications within 14-21 days may be necessary for this treatment, which is not approved by the Food and Drug Administration. “It is painless on application unless there’s a break in the skin,” Dr. Friedman said.
For warts on thicker areas such as palms and soles, he often employs combination therapy with cantharidin 1%, salicylic acid 30%, and podophyllotoxin 5%. “This can hurt a little bit, but some patients require only one treatment for cure,” he said. “Efficacy depends on the size of the wart.”
VP-102, a proprietary, drug-device combination product containing cantharidin, 0.7% “is coming down the pike,” Dr. Friedman said. “From the data we have, it seems that pairing with a curette or a #15 blade first gets better penetration, which makes sense. Patients come back every 3-4 weeks for treatment. It is a big investment, but it is worth it. I tell patients it’s not worth starting if you’re not going to see it through. I tell them, ‘we’re going to see a lot of each other until this is clear.’ ”
As for immunomodulatory approaches, imiquimod 5% cream is approved for treating genital and perianal warts. In Dr. Friedman’s clinical experience, it has limited efficacy on keratinized skin unless the surface has been disrupted, “so don’t even waste your time unless you are using some approach to enhance skin penetration,” he advised. “Insurance coverage can be a challenge,” he added.
He recommends application with salicylic acid alternating with imiquimod 5% cream every night at bedtime – under occlusion for thicker skinned areas.
For patients who favor use of natural products, off-label ingenol mebutate is an option. A case series of its use in 17 patients with anogenital warts found that 16 experienced clearance of all warts treated with either 0.05% or 0.015% ingenol mebutate gel. Local irritation occurred within 24-48 hours and lasted 2-5 days.
A natural alternative treatment is Candida albicans skin test antigen (Candin), especially for cases of multiple lesions on the hands and feet, because a field effect can be achieved, Dr. Friedman said. “The idea here is simple. At most, you’re talking about injecting a sentinel wart with 0.3 mL Candin 2-10 times every 3 weeks. The wart may be in a field of warts. That will induce an immune reaction that brings in the cavalry. I find that it works very well but it is painful, so when you’re injecting the feet, get the foot positioned well, because that patient may inadvertently kick you in the face [upon injection].”
Authors of a recent systematic review and meta-analysis highlighted the efficacy for systemic retinoids in the treatment of warts, particularly recalcitrant or recurrent types (Dermatol Ther 2021 34[2]:e14793). “Tazarotene is going to be your best bet if you can get it,” Dr. Friedman said. “If you have to go lower like OTC adapalene or tretinoin, be my guest, but tazarotene works best by slowing down that rapid turnover that the virus is imparting on the basal keratinocyte layer. It can enhance penetration of drug but also thin the warts out.”
Dr. Friedman characterized human papilloma virus (HPV) vaccines, such as Gardasil 9, as “one of the greatest innovations” in the treatment of warts. While indicated as a preventive strategy, “it also works as treatment. I’ve had patients with recalcitrant genital warts who will clear after taking the vaccine. It is something to think about as an adjuvant to everything we do, because it can function as a treatment.”
Another immunologic treatment option is the oral H2-receptor antagonist cimetidine taken 30 mg/kg per day for 3-5 months. “There is mixed evidence of efficacy with this,” Dr. Friedman said. “I tend to use it in cases of innumerable flat warts.”
As for cytotoxic options for treating warts, bleomycin works at 250-1,000 U/mL injected per lesion, with lidocaine. “This is painful to patients both on application and post treatment,” he said. “But it works really well when used properly.”
In one study of 46 patients who received intralesional bleomycin, 74% patients had complete resolution of all warts with an average of 1.7 treatments. About 70% of patients experienced pain that lasted less than 2 days after treatment. In a separate study of patients treated with bleomycin for warts, researchers in India diluted bleomycin with lidocaine to help mitigate some of that pain.
An additional cytotoxic option, 5-FU in formulations of 5% cream/solution or 1% cream, can effectively treat warts. Dr. Friedman typically suggests application to the affected area twice daily for 3-5 weeks. “The cost can be high especially for off-label use,” he said. He noted that Skin Medicinals makes a compounded wart solution composed of 5% 5-FU and salicylic acid 30% solution. A 50 mL container sells for about $50.
Dr. Friedman had no relevant disclosures related to his presentation.
When counseling patients with warts, Adam Friedman, MD, admits that he feels like a character from “Game of Thrones” since many treatment options are “medieval and painful,” from duct tape occlusion to the stings of liquid nitrogen and salicylic acid.
“We can combine destructive, immunologic, and cytotoxic approaches,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic & Surgical Conference. “It’s not one or the other, we want to be aggressive.
At the initial clinic visit, he advises asking patients how long the warts have been present, because sometimes they will go away within a year or two without treatment. “If someone says, ‘I’ve had these for years,’ you know you’re in for the long haul and you have to be aggressive with their therapy,” Dr. Friedman said. “Sometimes you’ll pick up plantar warts on a full-body skin exam and the patient may say, ‘I really don’t care. Please don’t touch them,’ so it’s important to understand how they are impacting quality of life.”
Patients should also be asked what treatments they have used previously, and it is important to set some realistic expectations and dispel some myths, Dr. Friedman said. “One of the most important things is that you must get these patients back. This is not often a one and done approach; you need to keep hitting them [with therapy], because if you let one infected keratinocyte survive, it’s going to come back and it’s still going to be contagious – more likely for that patient than for anyone else.”
The application of liquid nitrogen is a popular, inexpensive destructive treatment option, with spray canisters that cost about $600. “You have to consider the temperature of the liquid nitrogen spray because melanocytes die at negative 5 degrees Celsius, so you have to be mindful in patients with darker skin tones that you may leave with permanent dyschromia, meaning hypopigmentation or depigmentation when you do this,” he said. Because it is painful, “we’re limited when it comes to treating children with warts who are younger than 9 or 10. I don’t think the Q-tip method or dipping a hemostat in cryogen and touching the tip really works. You’ve got to create a nice ice ball that thaws and kills the infected keratinocytes.”
Dr. Friedman favors a 10-second freeze of the wart, usually for two to three cycles depending on its anatomic location, and he may give patients imiquimod or 5-FU to use at home for 5 nights of the week. A recently published study found that the use of ultrasound gel increases the efficacy of cryotherapy in the treatment of warts.
Another destructive treatment approach is cantharidin 0.7% applied topically in the office. It is believed to activate neutral serine proteases that cause degeneration of the desmosomal plaque, leading to detachment of tonofilaments from desmosomes. Repeat in-office applications within 14-21 days may be necessary for this treatment, which is not approved by the Food and Drug Administration. “It is painless on application unless there’s a break in the skin,” Dr. Friedman said.
For warts on thicker areas such as palms and soles, he often employs combination therapy with cantharidin 1%, salicylic acid 30%, and podophyllotoxin 5%. “This can hurt a little bit, but some patients require only one treatment for cure,” he said. “Efficacy depends on the size of the wart.”
VP-102, a proprietary, drug-device combination product containing cantharidin, 0.7% “is coming down the pike,” Dr. Friedman said. “From the data we have, it seems that pairing with a curette or a #15 blade first gets better penetration, which makes sense. Patients come back every 3-4 weeks for treatment. It is a big investment, but it is worth it. I tell patients it’s not worth starting if you’re not going to see it through. I tell them, ‘we’re going to see a lot of each other until this is clear.’ ”
As for immunomodulatory approaches, imiquimod 5% cream is approved for treating genital and perianal warts. In Dr. Friedman’s clinical experience, it has limited efficacy on keratinized skin unless the surface has been disrupted, “so don’t even waste your time unless you are using some approach to enhance skin penetration,” he advised. “Insurance coverage can be a challenge,” he added.
He recommends application with salicylic acid alternating with imiquimod 5% cream every night at bedtime – under occlusion for thicker skinned areas.
For patients who favor use of natural products, off-label ingenol mebutate is an option. A case series of its use in 17 patients with anogenital warts found that 16 experienced clearance of all warts treated with either 0.05% or 0.015% ingenol mebutate gel. Local irritation occurred within 24-48 hours and lasted 2-5 days.
A natural alternative treatment is Candida albicans skin test antigen (Candin), especially for cases of multiple lesions on the hands and feet, because a field effect can be achieved, Dr. Friedman said. “The idea here is simple. At most, you’re talking about injecting a sentinel wart with 0.3 mL Candin 2-10 times every 3 weeks. The wart may be in a field of warts. That will induce an immune reaction that brings in the cavalry. I find that it works very well but it is painful, so when you’re injecting the feet, get the foot positioned well, because that patient may inadvertently kick you in the face [upon injection].”
Authors of a recent systematic review and meta-analysis highlighted the efficacy for systemic retinoids in the treatment of warts, particularly recalcitrant or recurrent types (Dermatol Ther 2021 34[2]:e14793). “Tazarotene is going to be your best bet if you can get it,” Dr. Friedman said. “If you have to go lower like OTC adapalene or tretinoin, be my guest, but tazarotene works best by slowing down that rapid turnover that the virus is imparting on the basal keratinocyte layer. It can enhance penetration of drug but also thin the warts out.”
Dr. Friedman characterized human papilloma virus (HPV) vaccines, such as Gardasil 9, as “one of the greatest innovations” in the treatment of warts. While indicated as a preventive strategy, “it also works as treatment. I’ve had patients with recalcitrant genital warts who will clear after taking the vaccine. It is something to think about as an adjuvant to everything we do, because it can function as a treatment.”
Another immunologic treatment option is the oral H2-receptor antagonist cimetidine taken 30 mg/kg per day for 3-5 months. “There is mixed evidence of efficacy with this,” Dr. Friedman said. “I tend to use it in cases of innumerable flat warts.”
As for cytotoxic options for treating warts, bleomycin works at 250-1,000 U/mL injected per lesion, with lidocaine. “This is painful to patients both on application and post treatment,” he said. “But it works really well when used properly.”
In one study of 46 patients who received intralesional bleomycin, 74% patients had complete resolution of all warts with an average of 1.7 treatments. About 70% of patients experienced pain that lasted less than 2 days after treatment. In a separate study of patients treated with bleomycin for warts, researchers in India diluted bleomycin with lidocaine to help mitigate some of that pain.
An additional cytotoxic option, 5-FU in formulations of 5% cream/solution or 1% cream, can effectively treat warts. Dr. Friedman typically suggests application to the affected area twice daily for 3-5 weeks. “The cost can be high especially for off-label use,” he said. He noted that Skin Medicinals makes a compounded wart solution composed of 5% 5-FU and salicylic acid 30% solution. A 50 mL container sells for about $50.
Dr. Friedman had no relevant disclosures related to his presentation.
When counseling patients with warts, Adam Friedman, MD, admits that he feels like a character from “Game of Thrones” since many treatment options are “medieval and painful,” from duct tape occlusion to the stings of liquid nitrogen and salicylic acid.
“We can combine destructive, immunologic, and cytotoxic approaches,” Dr. Friedman, professor and chair of dermatology at George Washington University, Washington, said at the ODAC Dermatology, Aesthetic & Surgical Conference. “It’s not one or the other, we want to be aggressive.
At the initial clinic visit, he advises asking patients how long the warts have been present, because sometimes they will go away within a year or two without treatment. “If someone says, ‘I’ve had these for years,’ you know you’re in for the long haul and you have to be aggressive with their therapy,” Dr. Friedman said. “Sometimes you’ll pick up plantar warts on a full-body skin exam and the patient may say, ‘I really don’t care. Please don’t touch them,’ so it’s important to understand how they are impacting quality of life.”
Patients should also be asked what treatments they have used previously, and it is important to set some realistic expectations and dispel some myths, Dr. Friedman said. “One of the most important things is that you must get these patients back. This is not often a one and done approach; you need to keep hitting them [with therapy], because if you let one infected keratinocyte survive, it’s going to come back and it’s still going to be contagious – more likely for that patient than for anyone else.”
The application of liquid nitrogen is a popular, inexpensive destructive treatment option, with spray canisters that cost about $600. “You have to consider the temperature of the liquid nitrogen spray because melanocytes die at negative 5 degrees Celsius, so you have to be mindful in patients with darker skin tones that you may leave with permanent dyschromia, meaning hypopigmentation or depigmentation when you do this,” he said. Because it is painful, “we’re limited when it comes to treating children with warts who are younger than 9 or 10. I don’t think the Q-tip method or dipping a hemostat in cryogen and touching the tip really works. You’ve got to create a nice ice ball that thaws and kills the infected keratinocytes.”
Dr. Friedman favors a 10-second freeze of the wart, usually for two to three cycles depending on its anatomic location, and he may give patients imiquimod or 5-FU to use at home for 5 nights of the week. A recently published study found that the use of ultrasound gel increases the efficacy of cryotherapy in the treatment of warts.
Another destructive treatment approach is cantharidin 0.7% applied topically in the office. It is believed to activate neutral serine proteases that cause degeneration of the desmosomal plaque, leading to detachment of tonofilaments from desmosomes. Repeat in-office applications within 14-21 days may be necessary for this treatment, which is not approved by the Food and Drug Administration. “It is painless on application unless there’s a break in the skin,” Dr. Friedman said.
For warts on thicker areas such as palms and soles, he often employs combination therapy with cantharidin 1%, salicylic acid 30%, and podophyllotoxin 5%. “This can hurt a little bit, but some patients require only one treatment for cure,” he said. “Efficacy depends on the size of the wart.”
VP-102, a proprietary, drug-device combination product containing cantharidin, 0.7% “is coming down the pike,” Dr. Friedman said. “From the data we have, it seems that pairing with a curette or a #15 blade first gets better penetration, which makes sense. Patients come back every 3-4 weeks for treatment. It is a big investment, but it is worth it. I tell patients it’s not worth starting if you’re not going to see it through. I tell them, ‘we’re going to see a lot of each other until this is clear.’ ”
As for immunomodulatory approaches, imiquimod 5% cream is approved for treating genital and perianal warts. In Dr. Friedman’s clinical experience, it has limited efficacy on keratinized skin unless the surface has been disrupted, “so don’t even waste your time unless you are using some approach to enhance skin penetration,” he advised. “Insurance coverage can be a challenge,” he added.
He recommends application with salicylic acid alternating with imiquimod 5% cream every night at bedtime – under occlusion for thicker skinned areas.
For patients who favor use of natural products, off-label ingenol mebutate is an option. A case series of its use in 17 patients with anogenital warts found that 16 experienced clearance of all warts treated with either 0.05% or 0.015% ingenol mebutate gel. Local irritation occurred within 24-48 hours and lasted 2-5 days.
A natural alternative treatment is Candida albicans skin test antigen (Candin), especially for cases of multiple lesions on the hands and feet, because a field effect can be achieved, Dr. Friedman said. “The idea here is simple. At most, you’re talking about injecting a sentinel wart with 0.3 mL Candin 2-10 times every 3 weeks. The wart may be in a field of warts. That will induce an immune reaction that brings in the cavalry. I find that it works very well but it is painful, so when you’re injecting the feet, get the foot positioned well, because that patient may inadvertently kick you in the face [upon injection].”
Authors of a recent systematic review and meta-analysis highlighted the efficacy for systemic retinoids in the treatment of warts, particularly recalcitrant or recurrent types (Dermatol Ther 2021 34[2]:e14793). “Tazarotene is going to be your best bet if you can get it,” Dr. Friedman said. “If you have to go lower like OTC adapalene or tretinoin, be my guest, but tazarotene works best by slowing down that rapid turnover that the virus is imparting on the basal keratinocyte layer. It can enhance penetration of drug but also thin the warts out.”
Dr. Friedman characterized human papilloma virus (HPV) vaccines, such as Gardasil 9, as “one of the greatest innovations” in the treatment of warts. While indicated as a preventive strategy, “it also works as treatment. I’ve had patients with recalcitrant genital warts who will clear after taking the vaccine. It is something to think about as an adjuvant to everything we do, because it can function as a treatment.”
Another immunologic treatment option is the oral H2-receptor antagonist cimetidine taken 30 mg/kg per day for 3-5 months. “There is mixed evidence of efficacy with this,” Dr. Friedman said. “I tend to use it in cases of innumerable flat warts.”
As for cytotoxic options for treating warts, bleomycin works at 250-1,000 U/mL injected per lesion, with lidocaine. “This is painful to patients both on application and post treatment,” he said. “But it works really well when used properly.”
In one study of 46 patients who received intralesional bleomycin, 74% patients had complete resolution of all warts with an average of 1.7 treatments. About 70% of patients experienced pain that lasted less than 2 days after treatment. In a separate study of patients treated with bleomycin for warts, researchers in India diluted bleomycin with lidocaine to help mitigate some of that pain.
An additional cytotoxic option, 5-FU in formulations of 5% cream/solution or 1% cream, can effectively treat warts. Dr. Friedman typically suggests application to the affected area twice daily for 3-5 weeks. “The cost can be high especially for off-label use,” he said. He noted that Skin Medicinals makes a compounded wart solution composed of 5% 5-FU and salicylic acid 30% solution. A 50 mL container sells for about $50.
Dr. Friedman had no relevant disclosures related to his presentation.
FROM ODAC 2022
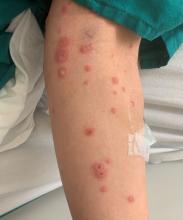
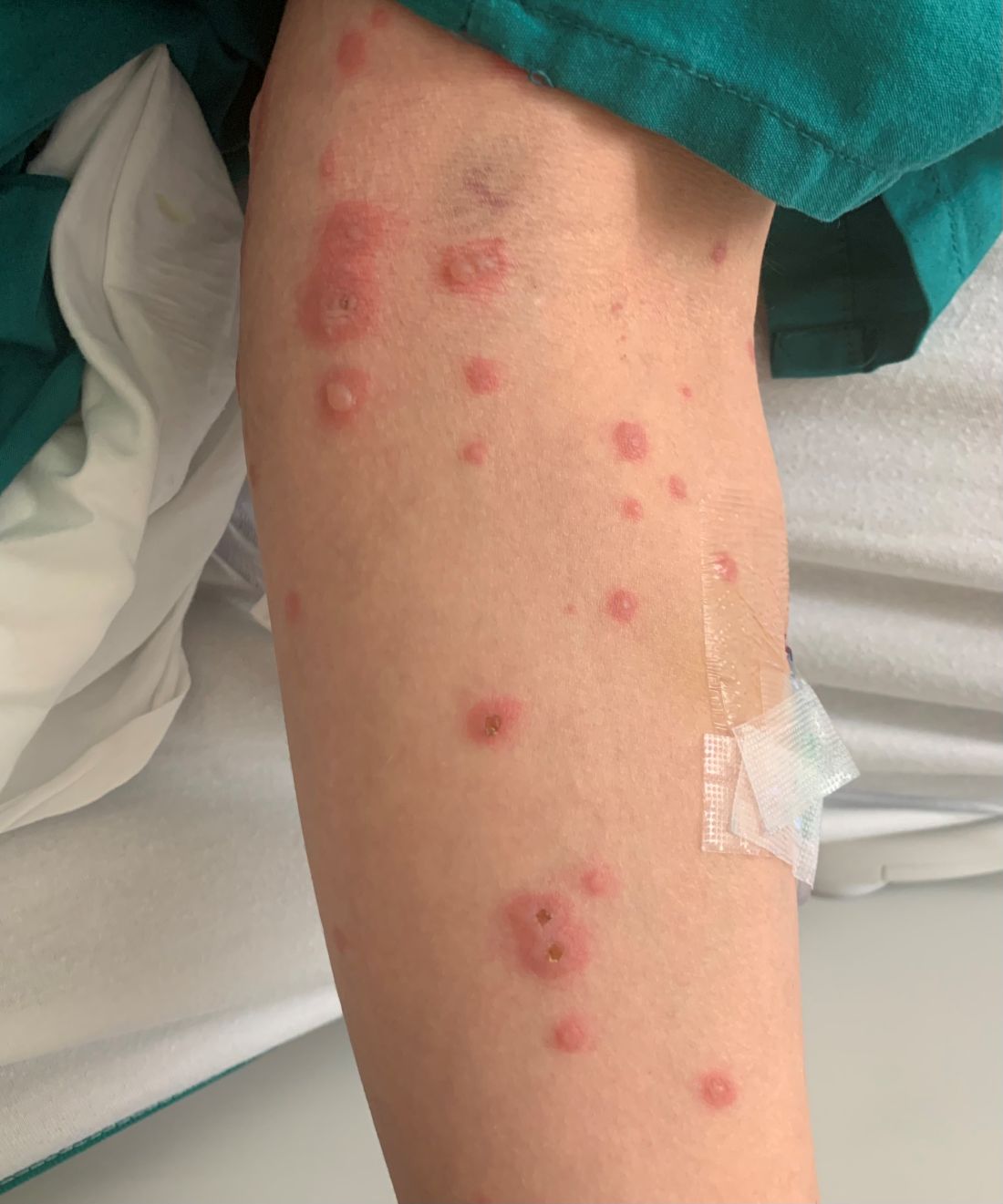
A 34-year-old male presented with 10 days of a pruritic rash
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Less frequently observable infectious agents associated with EM are Mycoplasma pneumoniae, Histoplasma capsulatum, and parapoxvirus (orf). Rarely, EM is triggered by drug eruption or systemic disease. Individuals of all age groups and races can be affected by EM. However, it is predominantly observed in young adult patients (20-40 years of age), and there is a male predominance.
Patients typically present with the abrupt onset of symmetrical red papules that evolve into typical and atypical targetoid lesions. Lesions evolve in 48-72 hours, favoring acrofacial sites that then spread down towards the trunk. Systemic symptoms such as fever and arthralgia may accompany the skin lesions.1-3
Erythema multiforme is recognized in two forms: EM minor and EM major. Both forms share the same characteristic of target lesions. However, the presence of mucosal involvement distinguishes the two. Mucosal involvement is absent or mild in EM minor, while mucosal involvement in EM major is often severe.2,3 Painful bullous lesions are commonly present in the mouth, genital, and ocular mucous membranes. Severe symptoms can often result in difficulty eating and drinking.
Diagnosis is largely clinical. Further histologic study may accompany diagnoses to exclude differential diagnosis. In EM, direct immunofluorescence (DIF) is negative. Histopathology reveals apoptosis of individual keratinocytes.1,2
Therapeutic treatment for painful bullous lesions in the mouth involve antiseptic rinses and anesthetic solutions. Preventive treatment for patients with HSV-associated EM recurrence includes oral acyclovir or valacyclovir.2
In this patient, a punch biopsy was performed, confirming the diagnosis. A DIF was negative, and a chest x-ray was negative. Treatment was initiated with oral acyclovir, doxycycline, and a prednisone taper. In addition, topical clobetasol propionate and magic mouthwash (Maalox/lidocaine/nystatin) was prescribed. The patient was placed on daily suppressive valacyclovir to prevent frequent recurrence of EM.
This case and photo were submitted by Ms. Pham, the University of California, Los Angeles, and Dr. Sateesh, San Diego Family Dermatology. Dr. Bilu-Martin edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Hafsi W and Badri T. Erythema Multiforme, in “StatPearls [Internet].” Treasure Island, Fla.: StatPearls Publishing, 2022 Jan.
2. Bolognia J et al. Dermatology. St. Louis: Mosby/Elsevier, 2008.
3. Oakley A. Erythema Multiforme. DermNet NZ. 2015 Oct.
Lipedema: A potentially devastating, often unrecognized disease
” according to C. William Hanke, MD, MPH.
“This disease is well known in Europe, especially in the Netherlands, Germany, and Austria, but in this country, I believe most dermatologists have never heard of it,” Dr. Hanke said at the ODAC Dermatology, Aesthetic & Surgical Conference.
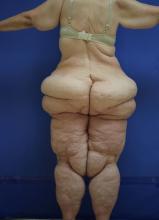
Clinically, patients with lipedema – also known as “two-body syndrome” – present with a symmetric, bilateral increase in subcutaneous fat, with “cuffs of fat” around the ankles. It usually affects the legs and thighs; the hands and feet are not affected.
“From the waist on up, the body looks like one person, and from the waist on down, it looks like an entirely different person,” said Dr. Hanke, a dermatologist who is program director for the micrographic surgery and dermatologic oncology fellowship training program at Ascension St. Vincent Hospital in Indianapolis. “Just think of the difficulty that the person has with their life in terms of buying clothes or social interactions. This is a devastating problem.”
Lipedema almost always affects women and is progressive from puberty. “Characteristically, patients have pain and bruise easily in the areas of lipedema,” said Dr. Hanke, who has served as president of the American Academy of Dermatology, the American Society for Dermatologic Surgery, the American College of Mohs Surgery, and the International Society for Dermatologic Surgery. The affected areas are painful to touch, making exercise uncomfortable for patients, he said.
Lipedema can be masked by obesity, “so, if you superimpose generalized obesity on lipedema, you have an even more difficult problem,” he added. “A physician who doesn’t understand the disease may perform standard nontumescent liposuction under general anesthesia, with cannulas, which traumatize lipedematous fat. Thereby, a patient with lipedema can then be inadvertently transformed into a patient with lympholipedema. Then you’ve got even an even worse problem.”
One might think that the rate of diabetes would be high among lipedema patients, “but diabetes is essentially nonexistent in this group,” he continued. However, patients with lipedema “may develop hypothyroidism, venous disease, joint pain, and fibrosis in the fat as the disease progresses.”
Lipedema stages, treatment
Lipedema is defined by three clinical stages: Stage one is characterized by an enlarged subcutaneous fat department, but the skin surface is smooth. In stage 2, the skin surface becomes wavy with irregularities and dents, and in stage 3, patients develop large deforming nodules and hanging flaps.
“If we can diagnose lipedema in the early stages and perform tumescent liposuction using tumescent local anesthesia, we can prevent the progression of the disease,” Dr. Hanke said. For patients who meet criteria for tumescent liposuction, three to six treatments may be required for stage 3 disease. “Tumescent local anesthesia should be used, because liposuction using tumescent local anesthesia is atraumatic to fat,” he said. “Usually, the most painful areas are treated first.”
In a single-center study from Germany that followed 85 patients who underwent tumescent liposuction for lipedema, researchers found that improvements in pain, bruising, and mobility were sustained at 4 and 8 years following the procedure. Patient quality of life and cosmetic appearance were also sustained.
In terms of liposuction’s cosmetic effects, “the goal of liposuction in lipedema patients is different,” Dr. Hanke said. “The goal is to get these people moving again, stabilize their weight, and minimize progression of the disease. Cosmetic improvement is secondary.”
A more recent follow-up study of 60 patients from the same single-center German study showed that the positive effects of liposuction lasted 12 years postoperatively without relevant progression of disease.
Following the first International Consensus Conference on Lipedema in Vienna in 2017, Dr. Hanke and colleagues published guidelines on preventing progression of lipedema with liposuction using tumescent local anesthesia.
“If patients with lipedema gain weight, the problem becomes even worse,” he said. “A sensible diet and nontraumatic exercise like water aerobics is ideal. If patients pursue yo-yo dieting, more and more fat stays in the legs after each cycle. Sometimes I’ll refer overweight patients with lipedema for a bariatric surgery consult.”
Dr. Hanke noted that Karen Herbst, MD, PhD, an endocrinologist at the University of Arizona, Tucson, who is widely considered an expert on the medical management of lipedema, has a website on lipedema care.
Dr. Hanke reported having no financial conflicts related to his presentation.
” according to C. William Hanke, MD, MPH.
“This disease is well known in Europe, especially in the Netherlands, Germany, and Austria, but in this country, I believe most dermatologists have never heard of it,” Dr. Hanke said at the ODAC Dermatology, Aesthetic & Surgical Conference.
Clinically, patients with lipedema – also known as “two-body syndrome” – present with a symmetric, bilateral increase in subcutaneous fat, with “cuffs of fat” around the ankles. It usually affects the legs and thighs; the hands and feet are not affected.
“From the waist on up, the body looks like one person, and from the waist on down, it looks like an entirely different person,” said Dr. Hanke, a dermatologist who is program director for the micrographic surgery and dermatologic oncology fellowship training program at Ascension St. Vincent Hospital in Indianapolis. “Just think of the difficulty that the person has with their life in terms of buying clothes or social interactions. This is a devastating problem.”
Lipedema almost always affects women and is progressive from puberty. “Characteristically, patients have pain and bruise easily in the areas of lipedema,” said Dr. Hanke, who has served as president of the American Academy of Dermatology, the American Society for Dermatologic Surgery, the American College of Mohs Surgery, and the International Society for Dermatologic Surgery. The affected areas are painful to touch, making exercise uncomfortable for patients, he said.
Lipedema can be masked by obesity, “so, if you superimpose generalized obesity on lipedema, you have an even more difficult problem,” he added. “A physician who doesn’t understand the disease may perform standard nontumescent liposuction under general anesthesia, with cannulas, which traumatize lipedematous fat. Thereby, a patient with lipedema can then be inadvertently transformed into a patient with lympholipedema. Then you’ve got even an even worse problem.”
One might think that the rate of diabetes would be high among lipedema patients, “but diabetes is essentially nonexistent in this group,” he continued. However, patients with lipedema “may develop hypothyroidism, venous disease, joint pain, and fibrosis in the fat as the disease progresses.”
Lipedema stages, treatment
Lipedema is defined by three clinical stages: Stage one is characterized by an enlarged subcutaneous fat department, but the skin surface is smooth. In stage 2, the skin surface becomes wavy with irregularities and dents, and in stage 3, patients develop large deforming nodules and hanging flaps.
“If we can diagnose lipedema in the early stages and perform tumescent liposuction using tumescent local anesthesia, we can prevent the progression of the disease,” Dr. Hanke said. For patients who meet criteria for tumescent liposuction, three to six treatments may be required for stage 3 disease. “Tumescent local anesthesia should be used, because liposuction using tumescent local anesthesia is atraumatic to fat,” he said. “Usually, the most painful areas are treated first.”
In a single-center study from Germany that followed 85 patients who underwent tumescent liposuction for lipedema, researchers found that improvements in pain, bruising, and mobility were sustained at 4 and 8 years following the procedure. Patient quality of life and cosmetic appearance were also sustained.
In terms of liposuction’s cosmetic effects, “the goal of liposuction in lipedema patients is different,” Dr. Hanke said. “The goal is to get these people moving again, stabilize their weight, and minimize progression of the disease. Cosmetic improvement is secondary.”
A more recent follow-up study of 60 patients from the same single-center German study showed that the positive effects of liposuction lasted 12 years postoperatively without relevant progression of disease.
Following the first International Consensus Conference on Lipedema in Vienna in 2017, Dr. Hanke and colleagues published guidelines on preventing progression of lipedema with liposuction using tumescent local anesthesia.
“If patients with lipedema gain weight, the problem becomes even worse,” he said. “A sensible diet and nontraumatic exercise like water aerobics is ideal. If patients pursue yo-yo dieting, more and more fat stays in the legs after each cycle. Sometimes I’ll refer overweight patients with lipedema for a bariatric surgery consult.”
Dr. Hanke noted that Karen Herbst, MD, PhD, an endocrinologist at the University of Arizona, Tucson, who is widely considered an expert on the medical management of lipedema, has a website on lipedema care.
Dr. Hanke reported having no financial conflicts related to his presentation.
” according to C. William Hanke, MD, MPH.
“This disease is well known in Europe, especially in the Netherlands, Germany, and Austria, but in this country, I believe most dermatologists have never heard of it,” Dr. Hanke said at the ODAC Dermatology, Aesthetic & Surgical Conference.
Clinically, patients with lipedema – also known as “two-body syndrome” – present with a symmetric, bilateral increase in subcutaneous fat, with “cuffs of fat” around the ankles. It usually affects the legs and thighs; the hands and feet are not affected.
“From the waist on up, the body looks like one person, and from the waist on down, it looks like an entirely different person,” said Dr. Hanke, a dermatologist who is program director for the micrographic surgery and dermatologic oncology fellowship training program at Ascension St. Vincent Hospital in Indianapolis. “Just think of the difficulty that the person has with their life in terms of buying clothes or social interactions. This is a devastating problem.”
Lipedema almost always affects women and is progressive from puberty. “Characteristically, patients have pain and bruise easily in the areas of lipedema,” said Dr. Hanke, who has served as president of the American Academy of Dermatology, the American Society for Dermatologic Surgery, the American College of Mohs Surgery, and the International Society for Dermatologic Surgery. The affected areas are painful to touch, making exercise uncomfortable for patients, he said.
Lipedema can be masked by obesity, “so, if you superimpose generalized obesity on lipedema, you have an even more difficult problem,” he added. “A physician who doesn’t understand the disease may perform standard nontumescent liposuction under general anesthesia, with cannulas, which traumatize lipedematous fat. Thereby, a patient with lipedema can then be inadvertently transformed into a patient with lympholipedema. Then you’ve got even an even worse problem.”
One might think that the rate of diabetes would be high among lipedema patients, “but diabetes is essentially nonexistent in this group,” he continued. However, patients with lipedema “may develop hypothyroidism, venous disease, joint pain, and fibrosis in the fat as the disease progresses.”
Lipedema stages, treatment
Lipedema is defined by three clinical stages: Stage one is characterized by an enlarged subcutaneous fat department, but the skin surface is smooth. In stage 2, the skin surface becomes wavy with irregularities and dents, and in stage 3, patients develop large deforming nodules and hanging flaps.
“If we can diagnose lipedema in the early stages and perform tumescent liposuction using tumescent local anesthesia, we can prevent the progression of the disease,” Dr. Hanke said. For patients who meet criteria for tumescent liposuction, three to six treatments may be required for stage 3 disease. “Tumescent local anesthesia should be used, because liposuction using tumescent local anesthesia is atraumatic to fat,” he said. “Usually, the most painful areas are treated first.”
In a single-center study from Germany that followed 85 patients who underwent tumescent liposuction for lipedema, researchers found that improvements in pain, bruising, and mobility were sustained at 4 and 8 years following the procedure. Patient quality of life and cosmetic appearance were also sustained.
In terms of liposuction’s cosmetic effects, “the goal of liposuction in lipedema patients is different,” Dr. Hanke said. “The goal is to get these people moving again, stabilize their weight, and minimize progression of the disease. Cosmetic improvement is secondary.”
A more recent follow-up study of 60 patients from the same single-center German study showed that the positive effects of liposuction lasted 12 years postoperatively without relevant progression of disease.
Following the first International Consensus Conference on Lipedema in Vienna in 2017, Dr. Hanke and colleagues published guidelines on preventing progression of lipedema with liposuction using tumescent local anesthesia.
“If patients with lipedema gain weight, the problem becomes even worse,” he said. “A sensible diet and nontraumatic exercise like water aerobics is ideal. If patients pursue yo-yo dieting, more and more fat stays in the legs after each cycle. Sometimes I’ll refer overweight patients with lipedema for a bariatric surgery consult.”
Dr. Hanke noted that Karen Herbst, MD, PhD, an endocrinologist at the University of Arizona, Tucson, who is widely considered an expert on the medical management of lipedema, has a website on lipedema care.
Dr. Hanke reported having no financial conflicts related to his presentation.
FROM ODAC 2022
Phase 2 studies of novel JAK1 inhibitor for HS show promise
results from two small phase 2 studies showed.
“INCB054707 is an oral, small-molecule JAK1 inhibitor with approximately 52-fold greater selectivity for JAK1 versus JAK2,” researchers led by Afsaneh Alavi, MD, of the Mayo Clinic in Rochester, Minn., wrote in an article published recently in the British Journal of Dermatology. “Specifically targeting JAK1, a critical regulator of proinflammatory cytokine signaling implicated in several immune-related diseases, may reduce cytokine signaling involved in HS pathogenesis while limiting JAK2-mediated cytopenias.”
For the first study, 10 patients received 15 mg INCB054707 once daily for 8 weeks (NCT03569371). For the second study, 35 patients were randomized to 30, 60, or 90 mg INCB054707 once daily or placebo (3:1 within each cohort) for 8 weeks (NCT03607487). Eligibility criteria for both studies included patients with Hurley stage II/III HS who aged 18-75 years with lesions present in two or more anatomic locations, and a total abscess and inflammatory nodule count of three or more. The primary endpoint for both studies was safety and tolerability. Secondary endpoints included HS Clinical Response (HiSCR) and other efficacy measures.
The researchers reported that 30% of patients in study 1 and 42.3% of patients who received INCB054707 in study 2 experienced one or more treatment-emergent adverse event, most commonly upper respiratory tract infection. Among evaluable patients, 3 patients (42.9%) in study 1 and 17 patients in study 2 (65.4%) achieved HiSCR at week 8, compared with 57.1% of those in the placebo group. By dosing, 55.6% in the 30-mg group achieved HiSCR at week 8, compared with 55.6% in the 60-mg group and 87.5% in the 90-mg group.
“In conclusion, safety and efficacy findings from these two phase 2 studies establish proof of concept for the JAK1 inhibitor INCB054707 in the treatment of moderate to severe HS,” the authors wrote. “A phase 2, dose-ranging, placebo-controlled study exploring three dose levels and including approximately 200 patients is ongoing (NCT04476043) and expected to provide additional evidence of the safety and efficacy profile of INCB054707 in patients with HS.”
INCB054707 is being developed by Incyte Corporation. Dr. Alavi disclosed that she has received honoraria as a consultant or advisory board participant from AbbVie, Janssen, Novartis, Boehringer Ingelheim, InflaRX, and UCB, and received honoraria as an investigator for Boehringer Ingelheim and Processa.
results from two small phase 2 studies showed.
“INCB054707 is an oral, small-molecule JAK1 inhibitor with approximately 52-fold greater selectivity for JAK1 versus JAK2,” researchers led by Afsaneh Alavi, MD, of the Mayo Clinic in Rochester, Minn., wrote in an article published recently in the British Journal of Dermatology. “Specifically targeting JAK1, a critical regulator of proinflammatory cytokine signaling implicated in several immune-related diseases, may reduce cytokine signaling involved in HS pathogenesis while limiting JAK2-mediated cytopenias.”
For the first study, 10 patients received 15 mg INCB054707 once daily for 8 weeks (NCT03569371). For the second study, 35 patients were randomized to 30, 60, or 90 mg INCB054707 once daily or placebo (3:1 within each cohort) for 8 weeks (NCT03607487). Eligibility criteria for both studies included patients with Hurley stage II/III HS who aged 18-75 years with lesions present in two or more anatomic locations, and a total abscess and inflammatory nodule count of three or more. The primary endpoint for both studies was safety and tolerability. Secondary endpoints included HS Clinical Response (HiSCR) and other efficacy measures.
The researchers reported that 30% of patients in study 1 and 42.3% of patients who received INCB054707 in study 2 experienced one or more treatment-emergent adverse event, most commonly upper respiratory tract infection. Among evaluable patients, 3 patients (42.9%) in study 1 and 17 patients in study 2 (65.4%) achieved HiSCR at week 8, compared with 57.1% of those in the placebo group. By dosing, 55.6% in the 30-mg group achieved HiSCR at week 8, compared with 55.6% in the 60-mg group and 87.5% in the 90-mg group.
“In conclusion, safety and efficacy findings from these two phase 2 studies establish proof of concept for the JAK1 inhibitor INCB054707 in the treatment of moderate to severe HS,” the authors wrote. “A phase 2, dose-ranging, placebo-controlled study exploring three dose levels and including approximately 200 patients is ongoing (NCT04476043) and expected to provide additional evidence of the safety and efficacy profile of INCB054707 in patients with HS.”
INCB054707 is being developed by Incyte Corporation. Dr. Alavi disclosed that she has received honoraria as a consultant or advisory board participant from AbbVie, Janssen, Novartis, Boehringer Ingelheim, InflaRX, and UCB, and received honoraria as an investigator for Boehringer Ingelheim and Processa.
results from two small phase 2 studies showed.
“INCB054707 is an oral, small-molecule JAK1 inhibitor with approximately 52-fold greater selectivity for JAK1 versus JAK2,” researchers led by Afsaneh Alavi, MD, of the Mayo Clinic in Rochester, Minn., wrote in an article published recently in the British Journal of Dermatology. “Specifically targeting JAK1, a critical regulator of proinflammatory cytokine signaling implicated in several immune-related diseases, may reduce cytokine signaling involved in HS pathogenesis while limiting JAK2-mediated cytopenias.”
For the first study, 10 patients received 15 mg INCB054707 once daily for 8 weeks (NCT03569371). For the second study, 35 patients were randomized to 30, 60, or 90 mg INCB054707 once daily or placebo (3:1 within each cohort) for 8 weeks (NCT03607487). Eligibility criteria for both studies included patients with Hurley stage II/III HS who aged 18-75 years with lesions present in two or more anatomic locations, and a total abscess and inflammatory nodule count of three or more. The primary endpoint for both studies was safety and tolerability. Secondary endpoints included HS Clinical Response (HiSCR) and other efficacy measures.
The researchers reported that 30% of patients in study 1 and 42.3% of patients who received INCB054707 in study 2 experienced one or more treatment-emergent adverse event, most commonly upper respiratory tract infection. Among evaluable patients, 3 patients (42.9%) in study 1 and 17 patients in study 2 (65.4%) achieved HiSCR at week 8, compared with 57.1% of those in the placebo group. By dosing, 55.6% in the 30-mg group achieved HiSCR at week 8, compared with 55.6% in the 60-mg group and 87.5% in the 90-mg group.
“In conclusion, safety and efficacy findings from these two phase 2 studies establish proof of concept for the JAK1 inhibitor INCB054707 in the treatment of moderate to severe HS,” the authors wrote. “A phase 2, dose-ranging, placebo-controlled study exploring three dose levels and including approximately 200 patients is ongoing (NCT04476043) and expected to provide additional evidence of the safety and efficacy profile of INCB054707 in patients with HS.”
INCB054707 is being developed by Incyte Corporation. Dr. Alavi disclosed that she has received honoraria as a consultant or advisory board participant from AbbVie, Janssen, Novartis, Boehringer Ingelheim, InflaRX, and UCB, and received honoraria as an investigator for Boehringer Ingelheim and Processa.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Survey: Medical cannabis use for skin conditions lags behind interest, acceptance
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
A , according to the results of a recent survey.
Almost 89% of respondents were in favor of medical cannabis use for dermatologic diseases, and 73% said that they would be comfortable seeing a dermatologist who recommended such products to them, Samuel Yeroushalmi, a 4th-year medical student at George Washington University, Washington, and associates reported.
“Consumers and patients are already using MCPs [medical cannabis products] to treat inflammatory skin conditions, such as acne, rosacea, atopic dermatitis, and psoriasis, even without guidance from a dermatologist. While acceptance was high, there were clear barriers reported limiting use and uptake, such as patient skepticism and a lack of understanding,” Adam Friedman, MD, senior author and chair of the department of dermatology at the university, said in a separate statement.
Dermatologic use of OTC cannabis products without the recommendation of a dermatologist was reported by 18% of the 504 of 700 adults who responded in the SurveyMonkey online panel. Of the two-thirds who had seen a dermatologist, 20% received a recommendation for an OTC product and 11% were recommended a product that required a department of health medical card, the investigators said.
Uptake among the patients who did receive a recommendation, however, was high: 76% for OTC products and 72% for those that required a medical card. Among those who had received an OTC recommendation, 32% used the cannabis product for psoriasis and 30% each for acne and rosacea, Mr. Yeroushalmi and his coauthors said.
The most common indication among the respondents with dermatologist recommendations for products requiring a medical card was for acne (68%), followed by psoriasis and rosacea (28% each). Cost was the main deterrent (60%) for those who declined to use the recommended cannabis product, with skepticism, limited understanding, and product illegality in their state each at 50%, the researchers said.
“Though cost and legality concerns are nonmodifiable barriers, dermatologists have an opportunity to educate those who know little in the way of medical cannabis or are skeptic[s],” they wrote. The survey results show that many patients are interested, and “the future should be bright for MCPs; we just need to show and disseminate the science,” Dr. Friedman commented in the statement.
One of the authors was from the University of Maryland, College Park. The authors had no disclosures to report.
FROM JOURNAL OF DRUGS IN DERMATOLOGY
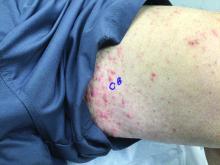
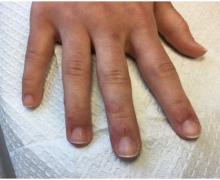
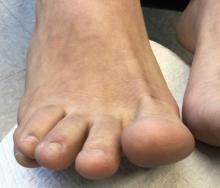
75-year-old White male presenting with progressive pruritus and a worsening rash
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
, although it can also be contracted through contaminated bedding and clothing. It can affect all races and ages.
Patients typically present with extremely pruritic, symmetric papules and excoriations. In nodular scabies, nodules and large papules are seen on exam. Thin lines in the skin called burrows may be present, especially in the webs between fingers. Female mites create burrows as they tunnel through the epidermis and lay eggs. The wrists, areola, waistline, and groin may all be involved, creating an imaginary circle between the areas described as the “circle of Hebra.” Penile and scrotal lesions are common in men.
Patients usually experience worse pruritus at night, which disturbs sleep. Crusted scabies is a severe form of scabies more often seen in those with immunocompromised immune systems. Clinically, thick crusted and scaly patches are present that are teeming with mites.
Diagnosis can be confirmed by performing a scabies prep, during which a burrow is scraped with a surgical blade. A drop of mineral oil is placed on the skin cells. The mite, ova, and feces can be visualized under the microscope. Wrists and hands usually have the highest yield for finding the parasites.
Topical treatments include permethrin 5% cream, lindane, benzyl benzoate, and crotamiton, and should be applied as two treatments a week apart. In the United States, permethrin is most commonly used. Ivermectin pills are used off label and are very effective and may be repeated for 1-2 weeks. All household contacts should be treated. Patients may still have pruritus for 2-4 weeks following treatment.
In this patient, a scabies prep was performed prior to performing repeat skin biopsies. Microscopic examination revealed ova, one mite, and feces. Treatment was initiated with ivermectin and permethrin.
Photos and case were submitted by Susannah Berke, MD, and Damon McClain, MD, Three Rivers Dermatology, Coraopolis, Pa.; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
Ophthalmologist who developed medical botox dies at 89
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
his family confirmed to National Public Radio.
Four decades ago, Alan Brown Scott, MD, a native of Berkeley, Calif., turned the drug, once a deadly poison, into a revolutionary treatment for obscure eye diseases. It later became a well-known blockbuster treatment for reducing the appearance of wrinkles and treating hyperhidrosis (excessive sweating). Other approved medical uses include treatment of overactive bladder and urinary incontinence.
According to the American Society of Plastic Surgeons, its popularity for cosmetic use was boosted further during the pandemic and it was the No. 1 minimally invasive cosmetic procedure performed in 2020. Among the 13.3 million procedures, 4.4 million involved Botox.
According to Bloomberg Businessweek, Ed Schantz, who was working in the military’s biological weapons program, was the one to first send the toxin to Dr. Scott, who wanted to explore its properties for medical use.
The same Bloomberg article also noted that the original botulinum toxin itself “is so powerful that a tiny amount can suffocate a person by paralyzing the muscles used for breathing.”
Dr. Scott was looking for a way to help his patients avoid extensive surgeries.
“Specifically, he was aiming to treat people with strabismus, or cross-eyes, and blepharospasm, which is an uncontrollable closure of eyes. Today, it’s also used as a treatment to help with migraines, hair loss, and drooling,” NPR reported.
The New York Times once described Botox as “medicine’s answer to duct tape.”
Dr. Scott was the executive director of the Smith-Kettlewell Eye Research Institute in San Francisco when he did his pioneering research with botulinum toxin in the 1970s and 1980s, according to a 2002 article in SFGate.
In 1991, Dr. Scott sold the drug to Allergan, when it was called Oculinum. The next year, the name was officially changed to Botox.
In 2002, Dr. Scott told SFGate, when asked about the more popular use for the drug, “I think that’s a charming, slightly frivolous use,” adding, “but it’s not along the lines of what I was into, applications for serious disorders.”
According to Scientific American in 2016, Dr. Scott, then age 83, kept working on the noncosmetic benefits of botulism-toxin injections for eye-related disorders at the Strabismus Research Foundation,
He told Scientific American he was proud that his efforts “are directly helpful to people.”
“There are interesting and difficult problems still to be solved, and I’m a practicing physician and I see them every day,” he said.
Dr. Scott’s daughter, Ann Scott, told NPR: “He definitely loved his work and he was also a really great father.” She said her dad involved his children in his research and work.
She added, “He was a really calm, more of a quiet reserved person,” and said he was committed to teaching his students, many of them international students.
“That was what he really loved,” she said.
Dr. Scott, who died Dec. 16, was in intensive care for the last 10 days from an unspecified illness, his daughter told NPR.
A version of this article first appeared on Medscape.com.
Prurigo nodularis has two disease endotypes, a cluster analysis shows
A cluster analysis of circulating plasma biomarkers in prurigo nodularis (PN) has identified two disease endotypes with inflammatory and noninflammatory biomarker profiles.
said senior author Shawn G. Kwatra, MD, of the department of dermatology at Johns Hopkins University, Baltimore. “This is the beginning of personalized medicine in prurigo nodularis.”
He and others have long observed significant clinical heterogeneity both in the presentation of PN – with the nodules in African American patients, for instance, appearing larger, thicker, and more fibrotic – and in patients’ response to immunomodulating and neuromodulating therapies.
To avoid the introduction of bias, the researchers used an unsupervised machine-learning approach to analyze the levels of 12 inflammatory biomarkers in 20 patients with PN and in matched healthy controls. The biomarkers were chosen based on their demonstrated dysregulation in PN and other inflammatory dermatoses.
The researchers then conducted a population-level analysis using multicenter electronic medical record data to explore inflammatory markers and verify findings from the cluster analysis. The study was published online Oct. 27, 2021, in the Journal of Investigative Dermatology.
One cluster of the 20 patients had higher levels of nine inflammatory biomarkers representing multiple immune axes: Higher interleukin-1 alpha, IL-4, IL-5, IL-6, IL-10, IL-17A, IL-22, and IL-25. This cluster had a higher percentage of Black patients, a higher severity of itch, and lower quality of life scores, the authors report in the preprint.
The other cluster – without such an inflammatory profile – had fewer Black patients and a higher percentage of patients with myelopathy (e.g. spinal stenosis, spinal trauma, degenerative disc disease). The rates of inflammatory comorbidities and of immune- and neuromodulating treatments at the time of blood draw were relatively equivalent between the two clusters.
In the subsequent population-level analysis, using data from a global health research network of EMRs from almost 50 health care organizations, Black patients with PN were found to have higher erythrocyte sedimentation rate, C-reactive protein, ferritin, and eosinophils, and lower transferrin, than White patients with PN. (The analysis included only Black and White patients.)
There are no Food and Drug Administration–approved therapies for PN, and “clinicians need to be really creative in managing these patients,” Dr. Kwatra said.
“There may be suggestions at the bedside that patients have more immune dysregulation, or maybe I’ll see increased circulating blood eosinophils,” he said. “And there are those who don’t seem to have any immune dysregulation and have more features of neurosensitization ... who may have a history of neck pain or back injury.”
The existence of endotypes in PN suggests that patients may benefit from personalized therapies with either immunomodulating or neuromodulating treatments, he and his colleagues wrote. “Further neuroimmune phenotyping studies of PN may pave the way for a future precision medicine management approach.”
Studies of PN conducted in Europe have been almost exclusively in White patients, Dr. Kwatra noted, even though PN has been shown to disproportionately affect Black and other racial/ethnic-minority patients.
Black patients with PN were found to have the highest all-cause mortality over 20 years post diagnosis in a separate analysis of over 22,000 patients with PN. Using data from the same health research network, Dr. Kwatra and coinvestigators stratified patients by race/ethnicity and compared each subgroup with a corresponding subgroup of similar race/ethnicity to control for inherent differences in mortality.
Overall, patients with PN had higher all-cause mortality than controls (hazard ratio, 1.70), likely because of a high comorbidity burden, they wrote in their research letter. Black patients with PN had the highest mortality (HR, 2.07), followed by White (HR, 1.74) and Hispanic (HR, 1.62) patients.
PN may exacerbate existing racial disparities in the social determinants of health, and Black patients may suffer from greater systemic inflammation, Dr. Kwatra and coauthors wrote. Certainly, he said, these findings, as well as the finding of a distinct inflammatory signature in Black patients with PN, support “that the disease burden is much higher” in these patients.
Dr Kwatra disclosed that he is an advisory board member/consultant for Celldex Therapeutics, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals and has received grant funding from several companies. His research is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Grants from the Dermatology Foundation and the Skin of Color Society also helped fund the cluster analysis.
A cluster analysis of circulating plasma biomarkers in prurigo nodularis (PN) has identified two disease endotypes with inflammatory and noninflammatory biomarker profiles.
said senior author Shawn G. Kwatra, MD, of the department of dermatology at Johns Hopkins University, Baltimore. “This is the beginning of personalized medicine in prurigo nodularis.”
He and others have long observed significant clinical heterogeneity both in the presentation of PN – with the nodules in African American patients, for instance, appearing larger, thicker, and more fibrotic – and in patients’ response to immunomodulating and neuromodulating therapies.
To avoid the introduction of bias, the researchers used an unsupervised machine-learning approach to analyze the levels of 12 inflammatory biomarkers in 20 patients with PN and in matched healthy controls. The biomarkers were chosen based on their demonstrated dysregulation in PN and other inflammatory dermatoses.
The researchers then conducted a population-level analysis using multicenter electronic medical record data to explore inflammatory markers and verify findings from the cluster analysis. The study was published online Oct. 27, 2021, in the Journal of Investigative Dermatology.
One cluster of the 20 patients had higher levels of nine inflammatory biomarkers representing multiple immune axes: Higher interleukin-1 alpha, IL-4, IL-5, IL-6, IL-10, IL-17A, IL-22, and IL-25. This cluster had a higher percentage of Black patients, a higher severity of itch, and lower quality of life scores, the authors report in the preprint.
The other cluster – without such an inflammatory profile – had fewer Black patients and a higher percentage of patients with myelopathy (e.g. spinal stenosis, spinal trauma, degenerative disc disease). The rates of inflammatory comorbidities and of immune- and neuromodulating treatments at the time of blood draw were relatively equivalent between the two clusters.
In the subsequent population-level analysis, using data from a global health research network of EMRs from almost 50 health care organizations, Black patients with PN were found to have higher erythrocyte sedimentation rate, C-reactive protein, ferritin, and eosinophils, and lower transferrin, than White patients with PN. (The analysis included only Black and White patients.)
There are no Food and Drug Administration–approved therapies for PN, and “clinicians need to be really creative in managing these patients,” Dr. Kwatra said.
“There may be suggestions at the bedside that patients have more immune dysregulation, or maybe I’ll see increased circulating blood eosinophils,” he said. “And there are those who don’t seem to have any immune dysregulation and have more features of neurosensitization ... who may have a history of neck pain or back injury.”
The existence of endotypes in PN suggests that patients may benefit from personalized therapies with either immunomodulating or neuromodulating treatments, he and his colleagues wrote. “Further neuroimmune phenotyping studies of PN may pave the way for a future precision medicine management approach.”
Studies of PN conducted in Europe have been almost exclusively in White patients, Dr. Kwatra noted, even though PN has been shown to disproportionately affect Black and other racial/ethnic-minority patients.
Black patients with PN were found to have the highest all-cause mortality over 20 years post diagnosis in a separate analysis of over 22,000 patients with PN. Using data from the same health research network, Dr. Kwatra and coinvestigators stratified patients by race/ethnicity and compared each subgroup with a corresponding subgroup of similar race/ethnicity to control for inherent differences in mortality.
Overall, patients with PN had higher all-cause mortality than controls (hazard ratio, 1.70), likely because of a high comorbidity burden, they wrote in their research letter. Black patients with PN had the highest mortality (HR, 2.07), followed by White (HR, 1.74) and Hispanic (HR, 1.62) patients.
PN may exacerbate existing racial disparities in the social determinants of health, and Black patients may suffer from greater systemic inflammation, Dr. Kwatra and coauthors wrote. Certainly, he said, these findings, as well as the finding of a distinct inflammatory signature in Black patients with PN, support “that the disease burden is much higher” in these patients.
Dr Kwatra disclosed that he is an advisory board member/consultant for Celldex Therapeutics, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals and has received grant funding from several companies. His research is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Grants from the Dermatology Foundation and the Skin of Color Society also helped fund the cluster analysis.
A cluster analysis of circulating plasma biomarkers in prurigo nodularis (PN) has identified two disease endotypes with inflammatory and noninflammatory biomarker profiles.
said senior author Shawn G. Kwatra, MD, of the department of dermatology at Johns Hopkins University, Baltimore. “This is the beginning of personalized medicine in prurigo nodularis.”
He and others have long observed significant clinical heterogeneity both in the presentation of PN – with the nodules in African American patients, for instance, appearing larger, thicker, and more fibrotic – and in patients’ response to immunomodulating and neuromodulating therapies.
To avoid the introduction of bias, the researchers used an unsupervised machine-learning approach to analyze the levels of 12 inflammatory biomarkers in 20 patients with PN and in matched healthy controls. The biomarkers were chosen based on their demonstrated dysregulation in PN and other inflammatory dermatoses.
The researchers then conducted a population-level analysis using multicenter electronic medical record data to explore inflammatory markers and verify findings from the cluster analysis. The study was published online Oct. 27, 2021, in the Journal of Investigative Dermatology.
One cluster of the 20 patients had higher levels of nine inflammatory biomarkers representing multiple immune axes: Higher interleukin-1 alpha, IL-4, IL-5, IL-6, IL-10, IL-17A, IL-22, and IL-25. This cluster had a higher percentage of Black patients, a higher severity of itch, and lower quality of life scores, the authors report in the preprint.
The other cluster – without such an inflammatory profile – had fewer Black patients and a higher percentage of patients with myelopathy (e.g. spinal stenosis, spinal trauma, degenerative disc disease). The rates of inflammatory comorbidities and of immune- and neuromodulating treatments at the time of blood draw were relatively equivalent between the two clusters.
In the subsequent population-level analysis, using data from a global health research network of EMRs from almost 50 health care organizations, Black patients with PN were found to have higher erythrocyte sedimentation rate, C-reactive protein, ferritin, and eosinophils, and lower transferrin, than White patients with PN. (The analysis included only Black and White patients.)
There are no Food and Drug Administration–approved therapies for PN, and “clinicians need to be really creative in managing these patients,” Dr. Kwatra said.
“There may be suggestions at the bedside that patients have more immune dysregulation, or maybe I’ll see increased circulating blood eosinophils,” he said. “And there are those who don’t seem to have any immune dysregulation and have more features of neurosensitization ... who may have a history of neck pain or back injury.”
The existence of endotypes in PN suggests that patients may benefit from personalized therapies with either immunomodulating or neuromodulating treatments, he and his colleagues wrote. “Further neuroimmune phenotyping studies of PN may pave the way for a future precision medicine management approach.”
Studies of PN conducted in Europe have been almost exclusively in White patients, Dr. Kwatra noted, even though PN has been shown to disproportionately affect Black and other racial/ethnic-minority patients.
Black patients with PN were found to have the highest all-cause mortality over 20 years post diagnosis in a separate analysis of over 22,000 patients with PN. Using data from the same health research network, Dr. Kwatra and coinvestigators stratified patients by race/ethnicity and compared each subgroup with a corresponding subgroup of similar race/ethnicity to control for inherent differences in mortality.
Overall, patients with PN had higher all-cause mortality than controls (hazard ratio, 1.70), likely because of a high comorbidity burden, they wrote in their research letter. Black patients with PN had the highest mortality (HR, 2.07), followed by White (HR, 1.74) and Hispanic (HR, 1.62) patients.
PN may exacerbate existing racial disparities in the social determinants of health, and Black patients may suffer from greater systemic inflammation, Dr. Kwatra and coauthors wrote. Certainly, he said, these findings, as well as the finding of a distinct inflammatory signature in Black patients with PN, support “that the disease burden is much higher” in these patients.
Dr Kwatra disclosed that he is an advisory board member/consultant for Celldex Therapeutics, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals and has received grant funding from several companies. His research is supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases. Grants from the Dermatology Foundation and the Skin of Color Society also helped fund the cluster analysis.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
Dermatologists take to TikTok to share their own ‘hacks’
A young woman is having her lip swabbed with an unknown substance, smiling, on the TikTok video. Seconds later, another young woman, wearing gloves, pushes a hyaluron pen against the first woman’s lips, who, in the next cut, is smiling, happy. “My first syringe down and already 1,000x more confident,” the caption reads.
That video is one of thousands showing hyaluron pen use on TikTok. The pens are sold online and are unapproved – which led to a Food and Drug Administration warning in October 2021 that use could cause bleeding, infection, blood vessel occlusion that could result in blindness or stroke, allergic reactions, and other injuries.
The warning has not stopped many TikTokkers, who also use the medium to promote all sorts of skin and aesthetic products and procedures, a large number unproven, unapproved, or ill advised. which, more often than not, comes from “skinfluencers,” aestheticians, and other laypeople, not board-certified dermatologists.
The suggested “hacks” can be harmless or ineffective, but they also can be misleading, fraudulent, or even dangerous.
Skinfluencers take the lead
TikTok has a reported 1 billion monthly users. Two-thirds are aged 10-29 years, according to data reported in February 2021 in the Journal of the American Academy of Dermatology by David X. Zheng, BA, and colleagues at Case Western Reserve University, Cleveland, and the department of dermatology, Johns Hopkins University, Baltimore.
Visitors consume information in video bits that run from 15 seconds to up to 3 minutes and can follow their favorite TikTokkers, browse for people or hashtags with a search function, or click on content recommended by the platform, which uses algorithms based on the user’s viewing habits to determine what might be of interest.
Some of the biggest “skinfluencers” have millions of followers: Hyram Yarbro, (@skincarebyhyram) for instance, has 6.6 million followers and his own line of skin care products at Sephora. Mr. Yarbro is seen as a no-nonsense debunker of skin care myths, as is British influencer James Welsh, who has 124,000 followers.
“The reason why people trust your average influencer person who’s not a doctor is because they’re relatable,” said Muneeb Shah, MD, a dermatology resident at Atlantic Dermatology in Wilmington, N.C. – known to his 11.4 million TikTok followers as @dermdoctor.
To Sandra Lee, MD, the popularity of nonprofessionals is easy to explain. “You have to think about the fact that a lot of people can’t see dermatologists – they don’t have the money, they don’t have the time to travel there, they don’t have health insurance, or they’re scared of doctors, so they’re willing to try to find an answer, and one of the easiest ways, one of the more entertaining ways to get information, is on social media.”
Dr. Lee is in private practice in Upland, Calif., but is better known as “Dr. Pimple Popper,” through her television show of the same name and her social media accounts, including on TikTok, where she has 14.4 million followers after having started in 2020.
“We’re all looking for that no-down-time, no-expense, no-lines, no-wrinkles, stay-young-forever magic bullet,” said Dr. Lee.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, agreed that people are looking for a quick fix. They don’t want to wait 12 weeks for an acne medication or 16 weeks for a biologic to work. “They want something simple, easy, do-it-yourself,” and “natural,” he said.
Laypeople are still the dominant producers – and have the most views – of dermatology content.
Morgan Nguyen, BA, at Northwestern University, Chicago, and colleagues looked at hashtags for the top 10 dermatologic diagnoses and procedures and analyzed the content of the first 40 TikTok videos in each category. About half the videos were produced by an individual, and 39% by a health care provider, according to the study, published in the International Journal of Women’s Dermatology. About 40% of the videos were educational, focusing on skin care, procedures, and disease treatment.
Viewership was highest for videos by laypeople, followed by those produced by business or industry accounts. Those produced by health care providers received only 18% of the views.
The most popular videos were about dermatologic diagnoses, with 2.5 billion views, followed by dermatologic procedures, with 708 million views.
Ms. Nguyen noted in the study that the most liked and most viewed posts were related to #skincare but that board-certified dermatologists produced only 2.5% of the #skincare videos.
Dermatologists take to TikTok
Some dermatologists have started their own TikTok accounts, seeking both to counteract misinformation and provide education.
Dr. Shah has become one of the top influencers on the platform. In a year-end wrap, TikTok put Dr. Shah at No. 7 on its top creators list for 2021.

The dermatology resident said that TikTok is a good tool for reaching patients who might not otherwise interact with dermatologists. He recounted the story of an individual who came into his office with the idea that they had hidradenitis suppurativa.
The person had self-diagnosed after seeing one of Dr. Shah’s TikTok videos on the condition. It was a pleasant surprise, said Dr. Shah. People with hidradenitis suppurativa often avoid treatment, and it’s underdiagnosed and improperly treated, despite an American Academy of Dermatology awareness campaign.
“Dermatologists on social media are almost like the communications department for dermatology,” Dr. Shah commented.
A key to making TikTok work to advance dermatologists’ goals is knowing what makes it unique.
Dr. Lee said she prefers it to Instagram, because TikTok’s algorithms and its younger-skewing audience help her reach a more specific audience.
The algorithm “creates a positive feedback loop in which popular content creators or viral trends are prioritized on the users’ homepages, in turn providing the creators of these videos with an even larger audience,” Mr. Zheng, of University Hospitals Cleveland Medical Center, and coauthors noted in their letter in the Journal of the American Academy of Dermatology.
TikTok also celebrates the everyday – someone doesn’t have to be a celebrity to make something go viral, said Dr. Lee. She believes that TikTok users are more accepting of average people with real problems – which helps when someone is TikTokking about a skin condition.
Doris Day, MD, who goes by @drdorisday on TikTok, agreed with Dr. Lee. “There are so many creative ways you can convey information with it that’s different than what you have on Instagram,” said Dr. Day, who is in private practice in New York. And, she added, “it does really lend itself to getting points out super-fast.”
Dermatologists on TikTok also said they like the “duets” and the “stitch” features, which allow users to add on to an existing video, essentially chiming in or responding to what might have already been posted, in a side-by-side format.
Dr. Shah said he often duets videos that have questionable content. “It allows me to directly respond to people. A lot of times, if something is going really viral and it’s not accurate, you’ll have a response from me or one of the other doctors” within hours or days.
Dr. Shah’s duets are labeled with “DermDoctor Reacts” or “DermDoctor Explains.” In one duet, with more than 2.8 million views, the upper half of the video is someone squeezing a blackhead, while Dr. Shah, in the bottom half, in green scrubs, opines over some hip-hop music: “This is just a blackhead. But once it gets to this point, they do need to be extracted because topical treatments won’t help.”
Dr. Lee – whose TikTok and other accounts capitalize on teens’ obsession with popping pimples – has a duet in which she advised that although popping will leave scars, there are more ideal times to pop, if they must. The duet has at least 21 million views.
Sometimes a TikTok video effectively takes on a trend without being a duet. Nurse practitioner Uy Dam (@uy.np) has a video that demonstrates the dangers of hyaluron pens. He uses both a pen and a needle to inject fluid into a block of jello. The pen delivers a scattershot load of differing depths, while the needle is exact. It’s visual and easy to understand and has at least 1.3 million views.
Still, TikTok, like other forms of social media, is full of misinformation and false accounts, including people who claim to be doctors. “It’s hard for the regular person, myself included, sometimes to be able to root through that and find out whether something is real or not,” said Dr. Lee.
Dr. Friedman said he’s concerned about the lack of accountability. A doctor could lose his or her license for promoting unproven cures, especially if they are harmful. But for influencers, “there’s no accountability for posting information that can actually hurt people.”
TikTok trends gone bad
And some people are being hurt by emulating what they see on TikTok.
Dr. Friedman had a patient with extreme irritant contact dermatitis, “almost like chemical burns to her underarms,” he said. He determined that she saw a video “hack” that recommended using baking soda to stop hyperhidrosis. The patient used so much that it burned her skin.
In 2020, do-it-yourself freckles – with henna or sewing needles impregnated with ink – went viral. Tilly Whitfeld, a 21-year-old reality TV star on Australia’s Big Brother show, told the New York Times that she tried it at home after seeing a TikTok video. She ordered brown tattoo ink online and later found out that it was contaminated with lead, according to the Times. Ms. Whitfeld developed an infection and temporary vision loss and has permanent scarring.
She has since put out a cautionary TikTok video that’s been viewed some 300,000 times.
TikTokkers have also flocked to the idea of using sunscreen to “contour” the face. Selected areas are left without sunscreen to burn or tan. In a duet, a plastic surgeon shakes his head as a young woman explains that “it works.”
Scalp-popping – in which the hair is yanked so hard that it pulls the galea off the skull – has been mostly shut down by TikTok. A search of “scalp popping” brings up the message: “Learn how to recognize harmful challenges and hoaxes.” At-home mole and skin tag removal, pimple-popping, and supposed acne cures such as drinking chlorophyll are all avidly documented and shared on TikTok.
Dr. Shah had a back-and-forth video dialog with someone who had stubbed a toe and then drilled a hole into the nail to drain the hematoma. In a reaction video, Dr. Shah said it was likely to turn into an infection. When it did, the man revealed the infection in a video where he tagged Dr. Shah and later posted a video at the podiatrist’s office having his nail removed, again tagging Dr. Shah.
“I think that pretty much no procedure for skin is good to do at home,” said Dr. Shah, who repeatedly admonishes against mole removal by a nonphysician. He tells followers that “it’s extremely dangerous – not only is it going to cause scarring, but you are potentially discarding a cancerous lesion.”
Unfortunately, most will not follow the advice, said Dr. Shah. That’s especially true of pimple-popping. Aiming for the least harm, he suggests in some TikTok videos that poppers keep the area clean, wear gloves, and consult a physician to get an antibiotic prescription. “You might as well at least guide them in the right direction,” he added.
Dr. Lee believes that lack of access to physicians, insurance, or money may play into how TikTok trends evolve. “Probably those people who injected their lips with this air gun thing, maybe they didn’t have the money necessarily to get filler,” she said.
Also, she noted, while TikTok may try to police its content, creators are incentivized to be outrageous. “The more inflammatory your post is, the more engagement you get.”
Dr. Shah thinks TikTok is self-correcting. “If you’re not being ethical or contradicting yourself, putting out information that’s not accurate, people are going to catch on very quickly,” he said. “The only value, the only currency you have on social media is the trust that you build with people that follow you.”
What it takes to be a TikTokker
For dermatologists, conveying their credentials and experience is one way to build that currency. Dr. Lee advised fellow doctors on TikTok to “showcase your training and how many years it took to become a dermatologist.”
Plunging into TikTok is not for everyone, though. It’s time consuming, said Dr. Lee, who now devotes most of her nonclinical time to TikTok. She creates her own content, leaving others to manage her Instagram account.
Many of those in the medical field who have dived into TikTok are residents, like Dr. Shah. “They are attuned to it and understand it more,” said Dr. Lee. “It’s harder for a lot of us who are older, who really weren’t involved that much in social media at all. It’s very hard to jump in.” There’s a learning curve, and it takes hours to create a single video. “You have to enjoy it and it has to be a part of your life,” she said.
Dr. Shah started experimenting with TikTok at the beginning of the pandemic in 2020 and has never turned back. Fast-talking, curious, and with an infectious sense of fun, he shares tidbits about his personal life – putting his wife in some of his videos – and always seems upbeat.
He said that, as his following grew, users began to see him as an authority figure and started “tagging” him more often, seeking his opinion on other videos. Although still a resident, he believes he has specialized knowledge to share. “Even if you’re not the world’s leading expert in a particular topic, you’re still adding value for the person who doesn’t know much.”
Dr. Shah also occasionally does promotional TikToks, identified as sponsored content. He said he only works with companies that he believes have legitimate products. “You do have to monetize at some point,” he said, noting that many dermatologists, himself included, are trading clinic time for TikTok. “There’s no universe where they can do this for free.”
Product endorsements are likely more rewarding for influencers and other users like Dr. Shah than the remuneration from TikTok, the company. The platform pays user accounts $20 per 1 million views, Dr. Shah said. “Financially, it’s not a big winner for a practicing dermatologist, but the educational outreach is worthwhile.”
To be successful also means understanding what drives viewership.
Using “trending” sounds has “been shown to increase the likelihood of a video amassing millions of views” and may increase engagement with dermatologists’ TikTok videos, wrote Bina Kassamali, BA, and colleagues at the Brigham and Women’s Hospital in Boston and the Ponce Health Science University School of Medicine in Ponce, Puerto Rico, in a letter published in the Journal of the American Academy of Dermatology in July 2021.
Certain content is more likely to engage viewers. In their analysis of top trending dermatologic hashtags, acne-related content was viewed 6.7 billion times, followed by alopecia, with 1.1 billion views. Psoriasis content had 84 million views, putting it eighth on the list of topics.
Dermatologists are still cracking TikTok. They are accumulating more followers on TikTok than on Instagram but have greater engagement on Instagram reels, wrote Mindy D. Szeto, MS, and colleagues at the University of Colorado at Denver, Aurora, and Rocky Vista University in Parker, Colo., in the Journal of the American Academy of Dermatology in April 2021.
Dr. Lee and Dr. Shah had the highest engagement rate on TikTok, according to Ms. Szeto. The engagement rate is calculated as (likes + comments per post)/(total followers) x 100.
“TikTok may currently be the leading avenue for audience education by dermatologist influencers,” they wrote, urging dermatologists to use the platform to answer the call as more of the public “continues to turn to social media for medical advice.”
Dr. Day said she will keep trying to build her TikTok audience. She has just 239 followers, compared with her 44,500 on Instagram. “The more I do TikTok, the more I do any of these mediums, the better I get at it,” she said. “We just have to put a little time and effort into it and try to get more followers and just keep sharing the information.”
Dr. Friedman sees it as a positive that some dermatologists have taken to TikTok to dispel myths and put “good information out there in small bites.” But to be more effective, they need more followers.
“The truth is that 14-year-old is probably going to listen more to a Hyram than a dermatologist,” he said. “Maybe we need to work with these other individuals who know how to take these messages and convert them to a language that can be digested by a 14-year-old, by a 12-year-old, by a 23-year-old. We need to come to the table together and not fight.”
A version of this article first appeared on Medscape.com.
A young woman is having her lip swabbed with an unknown substance, smiling, on the TikTok video. Seconds later, another young woman, wearing gloves, pushes a hyaluron pen against the first woman’s lips, who, in the next cut, is smiling, happy. “My first syringe down and already 1,000x more confident,” the caption reads.
That video is one of thousands showing hyaluron pen use on TikTok. The pens are sold online and are unapproved – which led to a Food and Drug Administration warning in October 2021 that use could cause bleeding, infection, blood vessel occlusion that could result in blindness or stroke, allergic reactions, and other injuries.
The warning has not stopped many TikTokkers, who also use the medium to promote all sorts of skin and aesthetic products and procedures, a large number unproven, unapproved, or ill advised. which, more often than not, comes from “skinfluencers,” aestheticians, and other laypeople, not board-certified dermatologists.
The suggested “hacks” can be harmless or ineffective, but they also can be misleading, fraudulent, or even dangerous.
Skinfluencers take the lead
TikTok has a reported 1 billion monthly users. Two-thirds are aged 10-29 years, according to data reported in February 2021 in the Journal of the American Academy of Dermatology by David X. Zheng, BA, and colleagues at Case Western Reserve University, Cleveland, and the department of dermatology, Johns Hopkins University, Baltimore.
Visitors consume information in video bits that run from 15 seconds to up to 3 minutes and can follow their favorite TikTokkers, browse for people or hashtags with a search function, or click on content recommended by the platform, which uses algorithms based on the user’s viewing habits to determine what might be of interest.
Some of the biggest “skinfluencers” have millions of followers: Hyram Yarbro, (@skincarebyhyram) for instance, has 6.6 million followers and his own line of skin care products at Sephora. Mr. Yarbro is seen as a no-nonsense debunker of skin care myths, as is British influencer James Welsh, who has 124,000 followers.
“The reason why people trust your average influencer person who’s not a doctor is because they’re relatable,” said Muneeb Shah, MD, a dermatology resident at Atlantic Dermatology in Wilmington, N.C. – known to his 11.4 million TikTok followers as @dermdoctor.
To Sandra Lee, MD, the popularity of nonprofessionals is easy to explain. “You have to think about the fact that a lot of people can’t see dermatologists – they don’t have the money, they don’t have the time to travel there, they don’t have health insurance, or they’re scared of doctors, so they’re willing to try to find an answer, and one of the easiest ways, one of the more entertaining ways to get information, is on social media.”
Dr. Lee is in private practice in Upland, Calif., but is better known as “Dr. Pimple Popper,” through her television show of the same name and her social media accounts, including on TikTok, where she has 14.4 million followers after having started in 2020.
“We’re all looking for that no-down-time, no-expense, no-lines, no-wrinkles, stay-young-forever magic bullet,” said Dr. Lee.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, agreed that people are looking for a quick fix. They don’t want to wait 12 weeks for an acne medication or 16 weeks for a biologic to work. “They want something simple, easy, do-it-yourself,” and “natural,” he said.
Laypeople are still the dominant producers – and have the most views – of dermatology content.
Morgan Nguyen, BA, at Northwestern University, Chicago, and colleagues looked at hashtags for the top 10 dermatologic diagnoses and procedures and analyzed the content of the first 40 TikTok videos in each category. About half the videos were produced by an individual, and 39% by a health care provider, according to the study, published in the International Journal of Women’s Dermatology. About 40% of the videos were educational, focusing on skin care, procedures, and disease treatment.
Viewership was highest for videos by laypeople, followed by those produced by business or industry accounts. Those produced by health care providers received only 18% of the views.
The most popular videos were about dermatologic diagnoses, with 2.5 billion views, followed by dermatologic procedures, with 708 million views.
Ms. Nguyen noted in the study that the most liked and most viewed posts were related to #skincare but that board-certified dermatologists produced only 2.5% of the #skincare videos.
Dermatologists take to TikTok
Some dermatologists have started their own TikTok accounts, seeking both to counteract misinformation and provide education.
Dr. Shah has become one of the top influencers on the platform. In a year-end wrap, TikTok put Dr. Shah at No. 7 on its top creators list for 2021.

The dermatology resident said that TikTok is a good tool for reaching patients who might not otherwise interact with dermatologists. He recounted the story of an individual who came into his office with the idea that they had hidradenitis suppurativa.
The person had self-diagnosed after seeing one of Dr. Shah’s TikTok videos on the condition. It was a pleasant surprise, said Dr. Shah. People with hidradenitis suppurativa often avoid treatment, and it’s underdiagnosed and improperly treated, despite an American Academy of Dermatology awareness campaign.
“Dermatologists on social media are almost like the communications department for dermatology,” Dr. Shah commented.
A key to making TikTok work to advance dermatologists’ goals is knowing what makes it unique.
Dr. Lee said she prefers it to Instagram, because TikTok’s algorithms and its younger-skewing audience help her reach a more specific audience.
The algorithm “creates a positive feedback loop in which popular content creators or viral trends are prioritized on the users’ homepages, in turn providing the creators of these videos with an even larger audience,” Mr. Zheng, of University Hospitals Cleveland Medical Center, and coauthors noted in their letter in the Journal of the American Academy of Dermatology.
TikTok also celebrates the everyday – someone doesn’t have to be a celebrity to make something go viral, said Dr. Lee. She believes that TikTok users are more accepting of average people with real problems – which helps when someone is TikTokking about a skin condition.
Doris Day, MD, who goes by @drdorisday on TikTok, agreed with Dr. Lee. “There are so many creative ways you can convey information with it that’s different than what you have on Instagram,” said Dr. Day, who is in private practice in New York. And, she added, “it does really lend itself to getting points out super-fast.”
Dermatologists on TikTok also said they like the “duets” and the “stitch” features, which allow users to add on to an existing video, essentially chiming in or responding to what might have already been posted, in a side-by-side format.
Dr. Shah said he often duets videos that have questionable content. “It allows me to directly respond to people. A lot of times, if something is going really viral and it’s not accurate, you’ll have a response from me or one of the other doctors” within hours or days.
Dr. Shah’s duets are labeled with “DermDoctor Reacts” or “DermDoctor Explains.” In one duet, with more than 2.8 million views, the upper half of the video is someone squeezing a blackhead, while Dr. Shah, in the bottom half, in green scrubs, opines over some hip-hop music: “This is just a blackhead. But once it gets to this point, they do need to be extracted because topical treatments won’t help.”
Dr. Lee – whose TikTok and other accounts capitalize on teens’ obsession with popping pimples – has a duet in which she advised that although popping will leave scars, there are more ideal times to pop, if they must. The duet has at least 21 million views.
Sometimes a TikTok video effectively takes on a trend without being a duet. Nurse practitioner Uy Dam (@uy.np) has a video that demonstrates the dangers of hyaluron pens. He uses both a pen and a needle to inject fluid into a block of jello. The pen delivers a scattershot load of differing depths, while the needle is exact. It’s visual and easy to understand and has at least 1.3 million views.
Still, TikTok, like other forms of social media, is full of misinformation and false accounts, including people who claim to be doctors. “It’s hard for the regular person, myself included, sometimes to be able to root through that and find out whether something is real or not,” said Dr. Lee.
Dr. Friedman said he’s concerned about the lack of accountability. A doctor could lose his or her license for promoting unproven cures, especially if they are harmful. But for influencers, “there’s no accountability for posting information that can actually hurt people.”
TikTok trends gone bad
And some people are being hurt by emulating what they see on TikTok.
Dr. Friedman had a patient with extreme irritant contact dermatitis, “almost like chemical burns to her underarms,” he said. He determined that she saw a video “hack” that recommended using baking soda to stop hyperhidrosis. The patient used so much that it burned her skin.
In 2020, do-it-yourself freckles – with henna or sewing needles impregnated with ink – went viral. Tilly Whitfeld, a 21-year-old reality TV star on Australia’s Big Brother show, told the New York Times that she tried it at home after seeing a TikTok video. She ordered brown tattoo ink online and later found out that it was contaminated with lead, according to the Times. Ms. Whitfeld developed an infection and temporary vision loss and has permanent scarring.
She has since put out a cautionary TikTok video that’s been viewed some 300,000 times.
TikTokkers have also flocked to the idea of using sunscreen to “contour” the face. Selected areas are left without sunscreen to burn or tan. In a duet, a plastic surgeon shakes his head as a young woman explains that “it works.”
Scalp-popping – in which the hair is yanked so hard that it pulls the galea off the skull – has been mostly shut down by TikTok. A search of “scalp popping” brings up the message: “Learn how to recognize harmful challenges and hoaxes.” At-home mole and skin tag removal, pimple-popping, and supposed acne cures such as drinking chlorophyll are all avidly documented and shared on TikTok.
Dr. Shah had a back-and-forth video dialog with someone who had stubbed a toe and then drilled a hole into the nail to drain the hematoma. In a reaction video, Dr. Shah said it was likely to turn into an infection. When it did, the man revealed the infection in a video where he tagged Dr. Shah and later posted a video at the podiatrist’s office having his nail removed, again tagging Dr. Shah.
“I think that pretty much no procedure for skin is good to do at home,” said Dr. Shah, who repeatedly admonishes against mole removal by a nonphysician. He tells followers that “it’s extremely dangerous – not only is it going to cause scarring, but you are potentially discarding a cancerous lesion.”
Unfortunately, most will not follow the advice, said Dr. Shah. That’s especially true of pimple-popping. Aiming for the least harm, he suggests in some TikTok videos that poppers keep the area clean, wear gloves, and consult a physician to get an antibiotic prescription. “You might as well at least guide them in the right direction,” he added.
Dr. Lee believes that lack of access to physicians, insurance, or money may play into how TikTok trends evolve. “Probably those people who injected their lips with this air gun thing, maybe they didn’t have the money necessarily to get filler,” she said.
Also, she noted, while TikTok may try to police its content, creators are incentivized to be outrageous. “The more inflammatory your post is, the more engagement you get.”
Dr. Shah thinks TikTok is self-correcting. “If you’re not being ethical or contradicting yourself, putting out information that’s not accurate, people are going to catch on very quickly,” he said. “The only value, the only currency you have on social media is the trust that you build with people that follow you.”
What it takes to be a TikTokker
For dermatologists, conveying their credentials and experience is one way to build that currency. Dr. Lee advised fellow doctors on TikTok to “showcase your training and how many years it took to become a dermatologist.”
Plunging into TikTok is not for everyone, though. It’s time consuming, said Dr. Lee, who now devotes most of her nonclinical time to TikTok. She creates her own content, leaving others to manage her Instagram account.
Many of those in the medical field who have dived into TikTok are residents, like Dr. Shah. “They are attuned to it and understand it more,” said Dr. Lee. “It’s harder for a lot of us who are older, who really weren’t involved that much in social media at all. It’s very hard to jump in.” There’s a learning curve, and it takes hours to create a single video. “You have to enjoy it and it has to be a part of your life,” she said.
Dr. Shah started experimenting with TikTok at the beginning of the pandemic in 2020 and has never turned back. Fast-talking, curious, and with an infectious sense of fun, he shares tidbits about his personal life – putting his wife in some of his videos – and always seems upbeat.
He said that, as his following grew, users began to see him as an authority figure and started “tagging” him more often, seeking his opinion on other videos. Although still a resident, he believes he has specialized knowledge to share. “Even if you’re not the world’s leading expert in a particular topic, you’re still adding value for the person who doesn’t know much.”
Dr. Shah also occasionally does promotional TikToks, identified as sponsored content. He said he only works with companies that he believes have legitimate products. “You do have to monetize at some point,” he said, noting that many dermatologists, himself included, are trading clinic time for TikTok. “There’s no universe where they can do this for free.”
Product endorsements are likely more rewarding for influencers and other users like Dr. Shah than the remuneration from TikTok, the company. The platform pays user accounts $20 per 1 million views, Dr. Shah said. “Financially, it’s not a big winner for a practicing dermatologist, but the educational outreach is worthwhile.”
To be successful also means understanding what drives viewership.
Using “trending” sounds has “been shown to increase the likelihood of a video amassing millions of views” and may increase engagement with dermatologists’ TikTok videos, wrote Bina Kassamali, BA, and colleagues at the Brigham and Women’s Hospital in Boston and the Ponce Health Science University School of Medicine in Ponce, Puerto Rico, in a letter published in the Journal of the American Academy of Dermatology in July 2021.
Certain content is more likely to engage viewers. In their analysis of top trending dermatologic hashtags, acne-related content was viewed 6.7 billion times, followed by alopecia, with 1.1 billion views. Psoriasis content had 84 million views, putting it eighth on the list of topics.
Dermatologists are still cracking TikTok. They are accumulating more followers on TikTok than on Instagram but have greater engagement on Instagram reels, wrote Mindy D. Szeto, MS, and colleagues at the University of Colorado at Denver, Aurora, and Rocky Vista University in Parker, Colo., in the Journal of the American Academy of Dermatology in April 2021.
Dr. Lee and Dr. Shah had the highest engagement rate on TikTok, according to Ms. Szeto. The engagement rate is calculated as (likes + comments per post)/(total followers) x 100.
“TikTok may currently be the leading avenue for audience education by dermatologist influencers,” they wrote, urging dermatologists to use the platform to answer the call as more of the public “continues to turn to social media for medical advice.”
Dr. Day said she will keep trying to build her TikTok audience. She has just 239 followers, compared with her 44,500 on Instagram. “The more I do TikTok, the more I do any of these mediums, the better I get at it,” she said. “We just have to put a little time and effort into it and try to get more followers and just keep sharing the information.”
Dr. Friedman sees it as a positive that some dermatologists have taken to TikTok to dispel myths and put “good information out there in small bites.” But to be more effective, they need more followers.
“The truth is that 14-year-old is probably going to listen more to a Hyram than a dermatologist,” he said. “Maybe we need to work with these other individuals who know how to take these messages and convert them to a language that can be digested by a 14-year-old, by a 12-year-old, by a 23-year-old. We need to come to the table together and not fight.”
A version of this article first appeared on Medscape.com.
A young woman is having her lip swabbed with an unknown substance, smiling, on the TikTok video. Seconds later, another young woman, wearing gloves, pushes a hyaluron pen against the first woman’s lips, who, in the next cut, is smiling, happy. “My first syringe down and already 1,000x more confident,” the caption reads.
That video is one of thousands showing hyaluron pen use on TikTok. The pens are sold online and are unapproved – which led to a Food and Drug Administration warning in October 2021 that use could cause bleeding, infection, blood vessel occlusion that could result in blindness or stroke, allergic reactions, and other injuries.
The warning has not stopped many TikTokkers, who also use the medium to promote all sorts of skin and aesthetic products and procedures, a large number unproven, unapproved, or ill advised. which, more often than not, comes from “skinfluencers,” aestheticians, and other laypeople, not board-certified dermatologists.
The suggested “hacks” can be harmless or ineffective, but they also can be misleading, fraudulent, or even dangerous.
Skinfluencers take the lead
TikTok has a reported 1 billion monthly users. Two-thirds are aged 10-29 years, according to data reported in February 2021 in the Journal of the American Academy of Dermatology by David X. Zheng, BA, and colleagues at Case Western Reserve University, Cleveland, and the department of dermatology, Johns Hopkins University, Baltimore.
Visitors consume information in video bits that run from 15 seconds to up to 3 minutes and can follow their favorite TikTokkers, browse for people or hashtags with a search function, or click on content recommended by the platform, which uses algorithms based on the user’s viewing habits to determine what might be of interest.
Some of the biggest “skinfluencers” have millions of followers: Hyram Yarbro, (@skincarebyhyram) for instance, has 6.6 million followers and his own line of skin care products at Sephora. Mr. Yarbro is seen as a no-nonsense debunker of skin care myths, as is British influencer James Welsh, who has 124,000 followers.
“The reason why people trust your average influencer person who’s not a doctor is because they’re relatable,” said Muneeb Shah, MD, a dermatology resident at Atlantic Dermatology in Wilmington, N.C. – known to his 11.4 million TikTok followers as @dermdoctor.
To Sandra Lee, MD, the popularity of nonprofessionals is easy to explain. “You have to think about the fact that a lot of people can’t see dermatologists – they don’t have the money, they don’t have the time to travel there, they don’t have health insurance, or they’re scared of doctors, so they’re willing to try to find an answer, and one of the easiest ways, one of the more entertaining ways to get information, is on social media.”
Dr. Lee is in private practice in Upland, Calif., but is better known as “Dr. Pimple Popper,” through her television show of the same name and her social media accounts, including on TikTok, where she has 14.4 million followers after having started in 2020.
“We’re all looking for that no-down-time, no-expense, no-lines, no-wrinkles, stay-young-forever magic bullet,” said Dr. Lee.
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, agreed that people are looking for a quick fix. They don’t want to wait 12 weeks for an acne medication or 16 weeks for a biologic to work. “They want something simple, easy, do-it-yourself,” and “natural,” he said.
Laypeople are still the dominant producers – and have the most views – of dermatology content.
Morgan Nguyen, BA, at Northwestern University, Chicago, and colleagues looked at hashtags for the top 10 dermatologic diagnoses and procedures and analyzed the content of the first 40 TikTok videos in each category. About half the videos were produced by an individual, and 39% by a health care provider, according to the study, published in the International Journal of Women’s Dermatology. About 40% of the videos were educational, focusing on skin care, procedures, and disease treatment.
Viewership was highest for videos by laypeople, followed by those produced by business or industry accounts. Those produced by health care providers received only 18% of the views.
The most popular videos were about dermatologic diagnoses, with 2.5 billion views, followed by dermatologic procedures, with 708 million views.
Ms. Nguyen noted in the study that the most liked and most viewed posts were related to #skincare but that board-certified dermatologists produced only 2.5% of the #skincare videos.
Dermatologists take to TikTok
Some dermatologists have started their own TikTok accounts, seeking both to counteract misinformation and provide education.
Dr. Shah has become one of the top influencers on the platform. In a year-end wrap, TikTok put Dr. Shah at No. 7 on its top creators list for 2021.

The dermatology resident said that TikTok is a good tool for reaching patients who might not otherwise interact with dermatologists. He recounted the story of an individual who came into his office with the idea that they had hidradenitis suppurativa.
The person had self-diagnosed after seeing one of Dr. Shah’s TikTok videos on the condition. It was a pleasant surprise, said Dr. Shah. People with hidradenitis suppurativa often avoid treatment, and it’s underdiagnosed and improperly treated, despite an American Academy of Dermatology awareness campaign.
“Dermatologists on social media are almost like the communications department for dermatology,” Dr. Shah commented.
A key to making TikTok work to advance dermatologists’ goals is knowing what makes it unique.
Dr. Lee said she prefers it to Instagram, because TikTok’s algorithms and its younger-skewing audience help her reach a more specific audience.
The algorithm “creates a positive feedback loop in which popular content creators or viral trends are prioritized on the users’ homepages, in turn providing the creators of these videos with an even larger audience,” Mr. Zheng, of University Hospitals Cleveland Medical Center, and coauthors noted in their letter in the Journal of the American Academy of Dermatology.
TikTok also celebrates the everyday – someone doesn’t have to be a celebrity to make something go viral, said Dr. Lee. She believes that TikTok users are more accepting of average people with real problems – which helps when someone is TikTokking about a skin condition.
Doris Day, MD, who goes by @drdorisday on TikTok, agreed with Dr. Lee. “There are so many creative ways you can convey information with it that’s different than what you have on Instagram,” said Dr. Day, who is in private practice in New York. And, she added, “it does really lend itself to getting points out super-fast.”
Dermatologists on TikTok also said they like the “duets” and the “stitch” features, which allow users to add on to an existing video, essentially chiming in or responding to what might have already been posted, in a side-by-side format.
Dr. Shah said he often duets videos that have questionable content. “It allows me to directly respond to people. A lot of times, if something is going really viral and it’s not accurate, you’ll have a response from me or one of the other doctors” within hours or days.
Dr. Shah’s duets are labeled with “DermDoctor Reacts” or “DermDoctor Explains.” In one duet, with more than 2.8 million views, the upper half of the video is someone squeezing a blackhead, while Dr. Shah, in the bottom half, in green scrubs, opines over some hip-hop music: “This is just a blackhead. But once it gets to this point, they do need to be extracted because topical treatments won’t help.”
Dr. Lee – whose TikTok and other accounts capitalize on teens’ obsession with popping pimples – has a duet in which she advised that although popping will leave scars, there are more ideal times to pop, if they must. The duet has at least 21 million views.
Sometimes a TikTok video effectively takes on a trend without being a duet. Nurse practitioner Uy Dam (@uy.np) has a video that demonstrates the dangers of hyaluron pens. He uses both a pen and a needle to inject fluid into a block of jello. The pen delivers a scattershot load of differing depths, while the needle is exact. It’s visual and easy to understand and has at least 1.3 million views.
Still, TikTok, like other forms of social media, is full of misinformation and false accounts, including people who claim to be doctors. “It’s hard for the regular person, myself included, sometimes to be able to root through that and find out whether something is real or not,” said Dr. Lee.
Dr. Friedman said he’s concerned about the lack of accountability. A doctor could lose his or her license for promoting unproven cures, especially if they are harmful. But for influencers, “there’s no accountability for posting information that can actually hurt people.”
TikTok trends gone bad
And some people are being hurt by emulating what they see on TikTok.
Dr. Friedman had a patient with extreme irritant contact dermatitis, “almost like chemical burns to her underarms,” he said. He determined that she saw a video “hack” that recommended using baking soda to stop hyperhidrosis. The patient used so much that it burned her skin.
In 2020, do-it-yourself freckles – with henna or sewing needles impregnated with ink – went viral. Tilly Whitfeld, a 21-year-old reality TV star on Australia’s Big Brother show, told the New York Times that she tried it at home after seeing a TikTok video. She ordered brown tattoo ink online and later found out that it was contaminated with lead, according to the Times. Ms. Whitfeld developed an infection and temporary vision loss and has permanent scarring.
She has since put out a cautionary TikTok video that’s been viewed some 300,000 times.
TikTokkers have also flocked to the idea of using sunscreen to “contour” the face. Selected areas are left without sunscreen to burn or tan. In a duet, a plastic surgeon shakes his head as a young woman explains that “it works.”
Scalp-popping – in which the hair is yanked so hard that it pulls the galea off the skull – has been mostly shut down by TikTok. A search of “scalp popping” brings up the message: “Learn how to recognize harmful challenges and hoaxes.” At-home mole and skin tag removal, pimple-popping, and supposed acne cures such as drinking chlorophyll are all avidly documented and shared on TikTok.
Dr. Shah had a back-and-forth video dialog with someone who had stubbed a toe and then drilled a hole into the nail to drain the hematoma. In a reaction video, Dr. Shah said it was likely to turn into an infection. When it did, the man revealed the infection in a video where he tagged Dr. Shah and later posted a video at the podiatrist’s office having his nail removed, again tagging Dr. Shah.
“I think that pretty much no procedure for skin is good to do at home,” said Dr. Shah, who repeatedly admonishes against mole removal by a nonphysician. He tells followers that “it’s extremely dangerous – not only is it going to cause scarring, but you are potentially discarding a cancerous lesion.”
Unfortunately, most will not follow the advice, said Dr. Shah. That’s especially true of pimple-popping. Aiming for the least harm, he suggests in some TikTok videos that poppers keep the area clean, wear gloves, and consult a physician to get an antibiotic prescription. “You might as well at least guide them in the right direction,” he added.
Dr. Lee believes that lack of access to physicians, insurance, or money may play into how TikTok trends evolve. “Probably those people who injected their lips with this air gun thing, maybe they didn’t have the money necessarily to get filler,” she said.
Also, she noted, while TikTok may try to police its content, creators are incentivized to be outrageous. “The more inflammatory your post is, the more engagement you get.”
Dr. Shah thinks TikTok is self-correcting. “If you’re not being ethical or contradicting yourself, putting out information that’s not accurate, people are going to catch on very quickly,” he said. “The only value, the only currency you have on social media is the trust that you build with people that follow you.”
What it takes to be a TikTokker
For dermatologists, conveying their credentials and experience is one way to build that currency. Dr. Lee advised fellow doctors on TikTok to “showcase your training and how many years it took to become a dermatologist.”
Plunging into TikTok is not for everyone, though. It’s time consuming, said Dr. Lee, who now devotes most of her nonclinical time to TikTok. She creates her own content, leaving others to manage her Instagram account.
Many of those in the medical field who have dived into TikTok are residents, like Dr. Shah. “They are attuned to it and understand it more,” said Dr. Lee. “It’s harder for a lot of us who are older, who really weren’t involved that much in social media at all. It’s very hard to jump in.” There’s a learning curve, and it takes hours to create a single video. “You have to enjoy it and it has to be a part of your life,” she said.
Dr. Shah started experimenting with TikTok at the beginning of the pandemic in 2020 and has never turned back. Fast-talking, curious, and with an infectious sense of fun, he shares tidbits about his personal life – putting his wife in some of his videos – and always seems upbeat.
He said that, as his following grew, users began to see him as an authority figure and started “tagging” him more often, seeking his opinion on other videos. Although still a resident, he believes he has specialized knowledge to share. “Even if you’re not the world’s leading expert in a particular topic, you’re still adding value for the person who doesn’t know much.”
Dr. Shah also occasionally does promotional TikToks, identified as sponsored content. He said he only works with companies that he believes have legitimate products. “You do have to monetize at some point,” he said, noting that many dermatologists, himself included, are trading clinic time for TikTok. “There’s no universe where they can do this for free.”
Product endorsements are likely more rewarding for influencers and other users like Dr. Shah than the remuneration from TikTok, the company. The platform pays user accounts $20 per 1 million views, Dr. Shah said. “Financially, it’s not a big winner for a practicing dermatologist, but the educational outreach is worthwhile.”
To be successful also means understanding what drives viewership.
Using “trending” sounds has “been shown to increase the likelihood of a video amassing millions of views” and may increase engagement with dermatologists’ TikTok videos, wrote Bina Kassamali, BA, and colleagues at the Brigham and Women’s Hospital in Boston and the Ponce Health Science University School of Medicine in Ponce, Puerto Rico, in a letter published in the Journal of the American Academy of Dermatology in July 2021.
Certain content is more likely to engage viewers. In their analysis of top trending dermatologic hashtags, acne-related content was viewed 6.7 billion times, followed by alopecia, with 1.1 billion views. Psoriasis content had 84 million views, putting it eighth on the list of topics.
Dermatologists are still cracking TikTok. They are accumulating more followers on TikTok than on Instagram but have greater engagement on Instagram reels, wrote Mindy D. Szeto, MS, and colleagues at the University of Colorado at Denver, Aurora, and Rocky Vista University in Parker, Colo., in the Journal of the American Academy of Dermatology in April 2021.
Dr. Lee and Dr. Shah had the highest engagement rate on TikTok, according to Ms. Szeto. The engagement rate is calculated as (likes + comments per post)/(total followers) x 100.
“TikTok may currently be the leading avenue for audience education by dermatologist influencers,” they wrote, urging dermatologists to use the platform to answer the call as more of the public “continues to turn to social media for medical advice.”
Dr. Day said she will keep trying to build her TikTok audience. She has just 239 followers, compared with her 44,500 on Instagram. “The more I do TikTok, the more I do any of these mediums, the better I get at it,” she said. “We just have to put a little time and effort into it and try to get more followers and just keep sharing the information.”
Dr. Friedman sees it as a positive that some dermatologists have taken to TikTok to dispel myths and put “good information out there in small bites.” But to be more effective, they need more followers.
“The truth is that 14-year-old is probably going to listen more to a Hyram than a dermatologist,” he said. “Maybe we need to work with these other individuals who know how to take these messages and convert them to a language that can be digested by a 14-year-old, by a 12-year-old, by a 23-year-old. We need to come to the table together and not fight.”
A version of this article first appeared on Medscape.com.
A 22-year-old presented with erythematous papules on her fingers and toes
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.
than men. Clinically, distal extremities such as toes, fingertips and heels, as well as the rims of the ears or nose develop erythematous to purple plaques. Lesions may be painful or pruritic. Over time, lesions may develop atrophy and resemble those of discoid lupus. While the pathogenesis is unknown, exposure to cold or wet environments can precipitate lesions.
Histopathology reveals a deep and superficial lymphocytic infiltrate with perieccrine involvement and fibrin deposition in vessels. Dermal edema is often present. Direct immunofluorescence shows an interface dermatitis positive for IgM, IgA, and C3.
The Mayo Clinic developed diagnostic criteria for diagnosing chilblains lupus. Two major criteria are acral skin lesions induced by cold exposure and evidence of lupus erythematosus in skin lesions (histopathologically or by direct immunofluorescence). Three minor criteria are the coexistence of systemic lupus erythematosus or discoid lupus erythematosus, response to antilupus treatment, and negative cryoglobulin and cold agglutinin studies.
Chilblains, or perniosis, has a similar clinical presentation to chilblain lupus erythematosus. However, serologic evidence of lupus, such as a positive antinuclear antibody (ANA), will be absent. Lupus pernio (Besnier-Tenneson syndrome) is a form of sarcoidosis that tends to favor the nose. These lesions are not precipitated by cold. It can be differentiated on histology. “COVID toes” is an entity described during the coronavirus pandemic, during which dermatologists noted pernio-like lesions in patients testing positive for coronavirus.
The patient’s labs revealed a positive ANA at 1:320 in a nucleolar speckled pattern, elevated double-stranded DNA, low C3 and C4 levels, elevated cardiolipin IgM Ab, and elevated sedimentation rate. COVID-19 antigen testing and COVID-19 antibodies were negative. A serum protein electrophoresis was negative. Cryoglobulins were negative.
Treatment includes protection from cold. Smoking cessation should be discussed. Topical steroids and topical calcineurin inhibitors are first-line treatments for mild disease. Antimalarials, such as hydroxychloroquine can be helpful. Systemic calcium channel blockers, systemic steroids, mycophenolate mofetil, and tacrolimus have all been reported as treatments. This patient responded well to hydroxychloroquine and topical steroids with full resolution of lesions.
This case was submitted by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Su WP et al. Cutis. 1994 Dec;54(6):395-9.
Werth V and Newman S. Chilblain lupus (SLE pernio). Dermatology Advisor. 2017.