User login
For MD-IQ use only
Psychogenic Purpura
To the Editor:
A 14-year-old Black adolescent girl presented with episodic, painful, edematous plaques that occurred symmetrically on the arms and legs of 5 years’ duration. The plaques evolved into hyperpigmented patches within 24 to 48 hours before eventually resolving. Fatigue, headache, arthralgias of the arms and legs, chest pain, abdominal pain, nausea, and vomiting variably accompanied these episodes.
Prior to visiting our clinic, the patient had been seen by numerous specialists. A review of her medical records revealed an initial diagnosis of Henoch-Schönlein purpura (HSP), then urticarial vasculitis. She had been treated with antihistamines, topical and systemic steroids, hydroxychloroquine, mycophenolate mofetil, dapsone, azathioprine, and gabapentin. All treatments were ineffectual. She underwent extensive diagnostic testing and imaging, which were normal or noncontributory, including type I allergy testing; multiple exhaustive batteries of hematologic testing; and computed tomography/magnetic resonance imaging/magnetic resonance angiography of the brain, chest, abdomen, and pelvic region. Biopsies from symptomatic segments of the gastrointestinal tract were normal.
Chronic treatment with systemic steroids over 9 months resulted in gastritis and an episode of hematemesis requiring emergent hospitalization. A lengthy multidisciplinary evaluation was conducted at the patient’s local community hospital; the team concluded that she had an urticarial-type rash with accompanying symptoms that did not have an autoimmune, rheumatologic, or inflammatory basis.
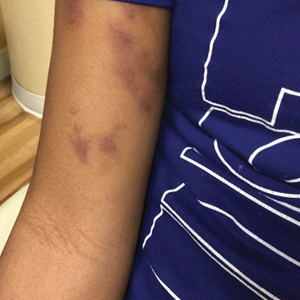
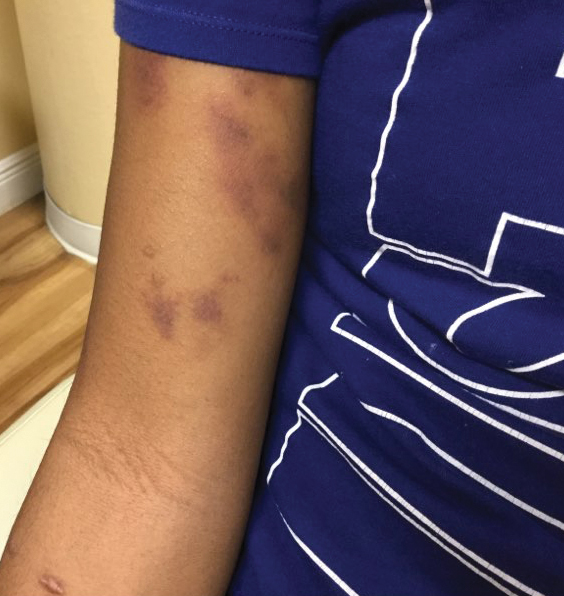
The patient’s medical history was remarkable for recent-onset panic attacks. Her family medical history was noncontributory. Physical examination revealed multiple violaceous hyperpigmented patches diffusely located on the proximal upper arms (Figure 1). There were no additional findings on physical examination.
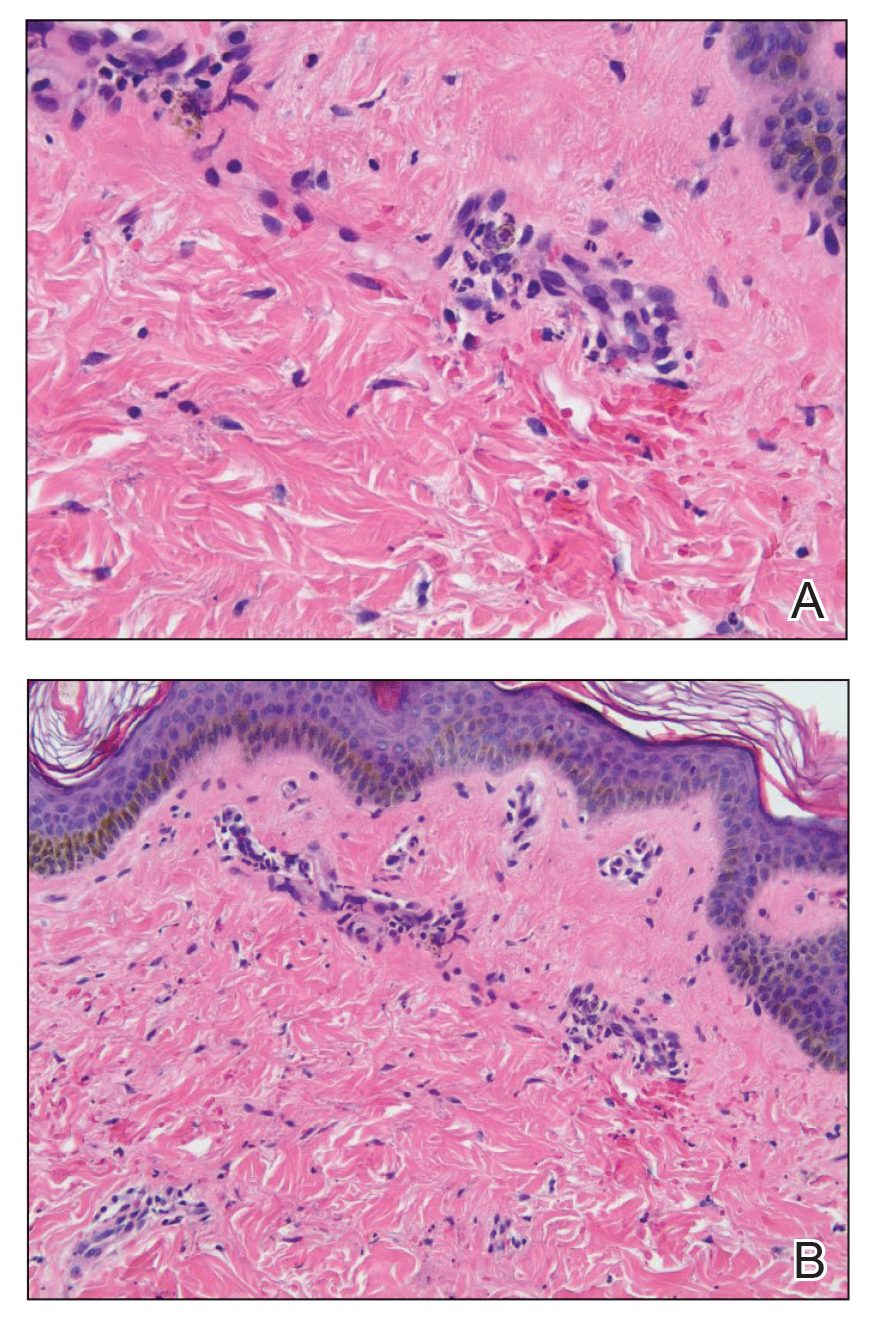
Punch biopsies were performed on lesional areas of the arm. Histopathology indicated a mild superficial perivascular dermal mixed infiltrate and extravasated erythrocytes (Figure 2). Direct immunofluorescence (DIF) testing was negative for vasculitis. Immunohistochemical stains for CD117 and tryptase demonstrated a slight increase in the number of dermal mast cells; however, the increase was not sufficient to diagnose cutaneous mastocytosis, which was in the differential. We proposed a diagnosis of psychogenic purpura (PP)(also known as Gardner-Diamond syndrome). She was treated with gabapentin, a selective serotonin reuptake inhibitor, and cognitive therapy. Unfortunately, after starting therapy the patient was lost to follow-up.
Psychogenic purpura is a rare vasculopathy of unknown etiology that may be a special form of factitious disorder.1,2 In one study, PP occurred predominantly in females aged 15 to 66 years, with a median onset age of 33 years.3 A prodrome of localized itching, burning, and/or pain precedes the development of edematous plaques. The plaques evolve into painful ecchymoses within 1 to 2 days and resolve in 10 days or fewer without treatment. Lesions most commonly occur on the extremities but may occur anywhere on the body. The most common associated finding is an underlying depressive disorder. Episodes may be accompanied by headache, dizziness, fatigue, fever, arthralgia, nausea, vomiting, abdominal pain, menstrual irregularities, myalgia, and urologic conditions.
In 1955, Gardner and Diamond4 described the first cases of PP in 4 female patients at Peter Bent Brigham Hospital in Boston, Massachusetts. The investigators were able to replicate the painful ecchymoses with intradermal injection of the patient’s own erythrocytes into the skin. They proposed that the underlying pathogenesis involved autosensitization to erythrocyte stroma.4 Since then, others have suggested that the pathogenesis may include autosensitization to erythrocyte phosphatidylserine, tonus dysregulation of venous capillaries, abnormal endothelial fibrin synthesis, and capillary wall instability.5-7
Histopathology typically reveals superficial and deep perivascular inflammation with extravasated erythrocytes. Direct immunofluorescence is negative for vasculitis.8 Diagnostics and laboratory findings for underlying systemic illness are negative or noncontributory. Cutaneous injection of 1 mL of the patient’s own washed erythrocytes may result in the formation of the characteristic painful plaques within 24 hours; however, this test is limited by lack of standardization and low sensitivity.3
Psychogenic purpura may share clinical features with cutaneous small vessel vasculitis, such as HSP or urticarial vasculitis. Some of the findings that our patient was experiencing, including purpura, arthralgia, and abdominal pain, are associated with HSP. However, HSP typically is self-limiting and classically features palpable purpura distributed across the lower extremities and buttocks. Histopathology demonstrates the classic findings of leukocytoclastic vasculitis; DIF typically is positive for perivascular IgA and C3 deposition. Increased serum IgA may be present.9 Urticarial vasculitis appears as erythematous indurated wheals that favor a proximal extremity and truncal distribution. They characteristically last longer than 24 hours, are frequently associated with nonprodromal pain or burning, and resolve with hyperpigmentation. Arthralgia and gastrointestinal, renal, pulmonary, cardiac, and neurologic symptoms may be present, especially in patients with low complement levels.10 Skin biopsy demonstrates leukocytoclasia that must be accompanied by vessel wall necrosis. Fibrinoid deposition, erythrocyte extravasation, or perivascular inflammation may be present. In 70% of cases revealing perivascular immunoglobulin, C3, and fibrinogen deposition, DIF is positive. Serum C1q autoantibody may be associated with the hypocomplementemic form.10
The classic histopathologic findings in leukocytoclastic vasculitis include transmural neutrophilic infiltration of the walls of small vessels, fibrinoid necrosis of vessel walls, leukocytoclasia, extravasated erythrocytes, and signs of endothelial cell damage.9 A prior punch biopsy in this patient demonstrated rare neutrophilic nuclear debris within the vessel walls without fibrin deposition. Although the presence of nuclear debris and extravasated erythrocytes could be compatible with a manifestation of urticarial vasculitis, the lack of direct evidence of vessel wall necrosis combined with subsequent biopsies unequivocally ruled out cutaneous small vessel vasculitis in our patient.
Psychogenic purpura has been reported to occur frequently in the background of psycho-emotional distress. In 1989, Ratnoff11 noted that many of the patients he was treating at the University Hospitals of Cleveland, Ohio, had a depressive syndrome. A review of patients treated at the Mayo Clinic in Rochester, Minnesota, illustrated concomitant psychiatric illnesses in 41 of 76 (54%) patients treated for PP, most commonly depressive, personality, and anxiety disorders.3
There is no consensus on therapy for PP. Treatment is based on providing symptomatic relief and relieving underlying psychiatric distress. Block et al12 found the use of selective serotonin reuptake inhibitors, tricyclic antidepressants, and psychotherapy to be successful in improving symptoms and reducing lesions at follow-up visits.
- Piette WW. Purpura: mechanisms and differential diagnosis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:376-389.
- Harth W, Taube KM, Gieler U. Factitious disorders in dermatology. J Dtsch Dermatol Ges. 2010;8:361-372.
- Sridharan M, Ali U, Hook CC, et al. The Mayo Clinic experience with psychogenic purpura (Gardner-Diamond syndrome). Am J Med Sci. 2019;357:411‐420.
- Gardner FH, Diamond LK. Autoerythrocyte sensitization; a form of purpura producing painful bruising following autosensitization to red blood cells in certain women. Blood. 1955;10:675-690.
- Groch GS, Finch SC, Rogoway W, et al. Studies in the pathogenesis of autoerythrocyte sensitization syndrome. Blood. 1966;28:19-33.
- Strunecká A, Krpejsová L, Palecek J, et al. Transbilayer redistribution of phosphatidylserine in erythrocytes of a patient with autoerythrocyte sensitization syndrome (psychogenic purpura). Folia Haematol Int Mag Klin Morphol Blutforsch. 1990;117:829-841.
- Merlen JF. Ecchymotic patches of the fingers and Gardner-Diamond vascular purpura. Phlebologie. 1987;40:473-487.
- Ivanov OL, Lvov AN, Michenko AV, et al. Autoerythrocyte sensitization syndrome (Gardner-Diamond syndrome): review of the literature. J Eur Acad Dermatol Venereol. 2009;23:499-504.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Hamad A, Jithpratuck W, Krishnaswamy G. Urticarial vasculitis and associated disorders. Ann Allergy Asthma Immunol. 2017;118:394-398.
- Ratnoff OD. Psychogenic purpura (autoerythrocyte sensitization): an unsolved dilemma. Am J Med. 1989;87:16N-21N.
- Block ME, Sitenga JL, Lehrer M, et al. Gardner‐Diamond syndrome: a systematic review of treatment options for a rare psychodermatological disorder. Int J Dermatol. 2019;58:782-787.
To the Editor:
A 14-year-old Black adolescent girl presented with episodic, painful, edematous plaques that occurred symmetrically on the arms and legs of 5 years’ duration. The plaques evolved into hyperpigmented patches within 24 to 48 hours before eventually resolving. Fatigue, headache, arthralgias of the arms and legs, chest pain, abdominal pain, nausea, and vomiting variably accompanied these episodes.
Prior to visiting our clinic, the patient had been seen by numerous specialists. A review of her medical records revealed an initial diagnosis of Henoch-Schönlein purpura (HSP), then urticarial vasculitis. She had been treated with antihistamines, topical and systemic steroids, hydroxychloroquine, mycophenolate mofetil, dapsone, azathioprine, and gabapentin. All treatments were ineffectual. She underwent extensive diagnostic testing and imaging, which were normal or noncontributory, including type I allergy testing; multiple exhaustive batteries of hematologic testing; and computed tomography/magnetic resonance imaging/magnetic resonance angiography of the brain, chest, abdomen, and pelvic region. Biopsies from symptomatic segments of the gastrointestinal tract were normal.
Chronic treatment with systemic steroids over 9 months resulted in gastritis and an episode of hematemesis requiring emergent hospitalization. A lengthy multidisciplinary evaluation was conducted at the patient’s local community hospital; the team concluded that she had an urticarial-type rash with accompanying symptoms that did not have an autoimmune, rheumatologic, or inflammatory basis.
The patient’s medical history was remarkable for recent-onset panic attacks. Her family medical history was noncontributory. Physical examination revealed multiple violaceous hyperpigmented patches diffusely located on the proximal upper arms (Figure 1). There were no additional findings on physical examination.

Punch biopsies were performed on lesional areas of the arm. Histopathology indicated a mild superficial perivascular dermal mixed infiltrate and extravasated erythrocytes (Figure 2). Direct immunofluorescence (DIF) testing was negative for vasculitis. Immunohistochemical stains for CD117 and tryptase demonstrated a slight increase in the number of dermal mast cells; however, the increase was not sufficient to diagnose cutaneous mastocytosis, which was in the differential. We proposed a diagnosis of psychogenic purpura (PP)(also known as Gardner-Diamond syndrome). She was treated with gabapentin, a selective serotonin reuptake inhibitor, and cognitive therapy. Unfortunately, after starting therapy the patient was lost to follow-up.

Psychogenic purpura is a rare vasculopathy of unknown etiology that may be a special form of factitious disorder.1,2 In one study, PP occurred predominantly in females aged 15 to 66 years, with a median onset age of 33 years.3 A prodrome of localized itching, burning, and/or pain precedes the development of edematous plaques. The plaques evolve into painful ecchymoses within 1 to 2 days and resolve in 10 days or fewer without treatment. Lesions most commonly occur on the extremities but may occur anywhere on the body. The most common associated finding is an underlying depressive disorder. Episodes may be accompanied by headache, dizziness, fatigue, fever, arthralgia, nausea, vomiting, abdominal pain, menstrual irregularities, myalgia, and urologic conditions.
In 1955, Gardner and Diamond4 described the first cases of PP in 4 female patients at Peter Bent Brigham Hospital in Boston, Massachusetts. The investigators were able to replicate the painful ecchymoses with intradermal injection of the patient’s own erythrocytes into the skin. They proposed that the underlying pathogenesis involved autosensitization to erythrocyte stroma.4 Since then, others have suggested that the pathogenesis may include autosensitization to erythrocyte phosphatidylserine, tonus dysregulation of venous capillaries, abnormal endothelial fibrin synthesis, and capillary wall instability.5-7
Histopathology typically reveals superficial and deep perivascular inflammation with extravasated erythrocytes. Direct immunofluorescence is negative for vasculitis.8 Diagnostics and laboratory findings for underlying systemic illness are negative or noncontributory. Cutaneous injection of 1 mL of the patient’s own washed erythrocytes may result in the formation of the characteristic painful plaques within 24 hours; however, this test is limited by lack of standardization and low sensitivity.3
Psychogenic purpura may share clinical features with cutaneous small vessel vasculitis, such as HSP or urticarial vasculitis. Some of the findings that our patient was experiencing, including purpura, arthralgia, and abdominal pain, are associated with HSP. However, HSP typically is self-limiting and classically features palpable purpura distributed across the lower extremities and buttocks. Histopathology demonstrates the classic findings of leukocytoclastic vasculitis; DIF typically is positive for perivascular IgA and C3 deposition. Increased serum IgA may be present.9 Urticarial vasculitis appears as erythematous indurated wheals that favor a proximal extremity and truncal distribution. They characteristically last longer than 24 hours, are frequently associated with nonprodromal pain or burning, and resolve with hyperpigmentation. Arthralgia and gastrointestinal, renal, pulmonary, cardiac, and neurologic symptoms may be present, especially in patients with low complement levels.10 Skin biopsy demonstrates leukocytoclasia that must be accompanied by vessel wall necrosis. Fibrinoid deposition, erythrocyte extravasation, or perivascular inflammation may be present. In 70% of cases revealing perivascular immunoglobulin, C3, and fibrinogen deposition, DIF is positive. Serum C1q autoantibody may be associated with the hypocomplementemic form.10
The classic histopathologic findings in leukocytoclastic vasculitis include transmural neutrophilic infiltration of the walls of small vessels, fibrinoid necrosis of vessel walls, leukocytoclasia, extravasated erythrocytes, and signs of endothelial cell damage.9 A prior punch biopsy in this patient demonstrated rare neutrophilic nuclear debris within the vessel walls without fibrin deposition. Although the presence of nuclear debris and extravasated erythrocytes could be compatible with a manifestation of urticarial vasculitis, the lack of direct evidence of vessel wall necrosis combined with subsequent biopsies unequivocally ruled out cutaneous small vessel vasculitis in our patient.
Psychogenic purpura has been reported to occur frequently in the background of psycho-emotional distress. In 1989, Ratnoff11 noted that many of the patients he was treating at the University Hospitals of Cleveland, Ohio, had a depressive syndrome. A review of patients treated at the Mayo Clinic in Rochester, Minnesota, illustrated concomitant psychiatric illnesses in 41 of 76 (54%) patients treated for PP, most commonly depressive, personality, and anxiety disorders.3
There is no consensus on therapy for PP. Treatment is based on providing symptomatic relief and relieving underlying psychiatric distress. Block et al12 found the use of selective serotonin reuptake inhibitors, tricyclic antidepressants, and psychotherapy to be successful in improving symptoms and reducing lesions at follow-up visits.
To the Editor:
A 14-year-old Black adolescent girl presented with episodic, painful, edematous plaques that occurred symmetrically on the arms and legs of 5 years’ duration. The plaques evolved into hyperpigmented patches within 24 to 48 hours before eventually resolving. Fatigue, headache, arthralgias of the arms and legs, chest pain, abdominal pain, nausea, and vomiting variably accompanied these episodes.
Prior to visiting our clinic, the patient had been seen by numerous specialists. A review of her medical records revealed an initial diagnosis of Henoch-Schönlein purpura (HSP), then urticarial vasculitis. She had been treated with antihistamines, topical and systemic steroids, hydroxychloroquine, mycophenolate mofetil, dapsone, azathioprine, and gabapentin. All treatments were ineffectual. She underwent extensive diagnostic testing and imaging, which were normal or noncontributory, including type I allergy testing; multiple exhaustive batteries of hematologic testing; and computed tomography/magnetic resonance imaging/magnetic resonance angiography of the brain, chest, abdomen, and pelvic region. Biopsies from symptomatic segments of the gastrointestinal tract were normal.
Chronic treatment with systemic steroids over 9 months resulted in gastritis and an episode of hematemesis requiring emergent hospitalization. A lengthy multidisciplinary evaluation was conducted at the patient’s local community hospital; the team concluded that she had an urticarial-type rash with accompanying symptoms that did not have an autoimmune, rheumatologic, or inflammatory basis.
The patient’s medical history was remarkable for recent-onset panic attacks. Her family medical history was noncontributory. Physical examination revealed multiple violaceous hyperpigmented patches diffusely located on the proximal upper arms (Figure 1). There were no additional findings on physical examination.

Punch biopsies were performed on lesional areas of the arm. Histopathology indicated a mild superficial perivascular dermal mixed infiltrate and extravasated erythrocytes (Figure 2). Direct immunofluorescence (DIF) testing was negative for vasculitis. Immunohistochemical stains for CD117 and tryptase demonstrated a slight increase in the number of dermal mast cells; however, the increase was not sufficient to diagnose cutaneous mastocytosis, which was in the differential. We proposed a diagnosis of psychogenic purpura (PP)(also known as Gardner-Diamond syndrome). She was treated with gabapentin, a selective serotonin reuptake inhibitor, and cognitive therapy. Unfortunately, after starting therapy the patient was lost to follow-up.

Psychogenic purpura is a rare vasculopathy of unknown etiology that may be a special form of factitious disorder.1,2 In one study, PP occurred predominantly in females aged 15 to 66 years, with a median onset age of 33 years.3 A prodrome of localized itching, burning, and/or pain precedes the development of edematous plaques. The plaques evolve into painful ecchymoses within 1 to 2 days and resolve in 10 days or fewer without treatment. Lesions most commonly occur on the extremities but may occur anywhere on the body. The most common associated finding is an underlying depressive disorder. Episodes may be accompanied by headache, dizziness, fatigue, fever, arthralgia, nausea, vomiting, abdominal pain, menstrual irregularities, myalgia, and urologic conditions.
In 1955, Gardner and Diamond4 described the first cases of PP in 4 female patients at Peter Bent Brigham Hospital in Boston, Massachusetts. The investigators were able to replicate the painful ecchymoses with intradermal injection of the patient’s own erythrocytes into the skin. They proposed that the underlying pathogenesis involved autosensitization to erythrocyte stroma.4 Since then, others have suggested that the pathogenesis may include autosensitization to erythrocyte phosphatidylserine, tonus dysregulation of venous capillaries, abnormal endothelial fibrin synthesis, and capillary wall instability.5-7
Histopathology typically reveals superficial and deep perivascular inflammation with extravasated erythrocytes. Direct immunofluorescence is negative for vasculitis.8 Diagnostics and laboratory findings for underlying systemic illness are negative or noncontributory. Cutaneous injection of 1 mL of the patient’s own washed erythrocytes may result in the formation of the characteristic painful plaques within 24 hours; however, this test is limited by lack of standardization and low sensitivity.3
Psychogenic purpura may share clinical features with cutaneous small vessel vasculitis, such as HSP or urticarial vasculitis. Some of the findings that our patient was experiencing, including purpura, arthralgia, and abdominal pain, are associated with HSP. However, HSP typically is self-limiting and classically features palpable purpura distributed across the lower extremities and buttocks. Histopathology demonstrates the classic findings of leukocytoclastic vasculitis; DIF typically is positive for perivascular IgA and C3 deposition. Increased serum IgA may be present.9 Urticarial vasculitis appears as erythematous indurated wheals that favor a proximal extremity and truncal distribution. They characteristically last longer than 24 hours, are frequently associated with nonprodromal pain or burning, and resolve with hyperpigmentation. Arthralgia and gastrointestinal, renal, pulmonary, cardiac, and neurologic symptoms may be present, especially in patients with low complement levels.10 Skin biopsy demonstrates leukocytoclasia that must be accompanied by vessel wall necrosis. Fibrinoid deposition, erythrocyte extravasation, or perivascular inflammation may be present. In 70% of cases revealing perivascular immunoglobulin, C3, and fibrinogen deposition, DIF is positive. Serum C1q autoantibody may be associated with the hypocomplementemic form.10
The classic histopathologic findings in leukocytoclastic vasculitis include transmural neutrophilic infiltration of the walls of small vessels, fibrinoid necrosis of vessel walls, leukocytoclasia, extravasated erythrocytes, and signs of endothelial cell damage.9 A prior punch biopsy in this patient demonstrated rare neutrophilic nuclear debris within the vessel walls without fibrin deposition. Although the presence of nuclear debris and extravasated erythrocytes could be compatible with a manifestation of urticarial vasculitis, the lack of direct evidence of vessel wall necrosis combined with subsequent biopsies unequivocally ruled out cutaneous small vessel vasculitis in our patient.
Psychogenic purpura has been reported to occur frequently in the background of psycho-emotional distress. In 1989, Ratnoff11 noted that many of the patients he was treating at the University Hospitals of Cleveland, Ohio, had a depressive syndrome. A review of patients treated at the Mayo Clinic in Rochester, Minnesota, illustrated concomitant psychiatric illnesses in 41 of 76 (54%) patients treated for PP, most commonly depressive, personality, and anxiety disorders.3
There is no consensus on therapy for PP. Treatment is based on providing symptomatic relief and relieving underlying psychiatric distress. Block et al12 found the use of selective serotonin reuptake inhibitors, tricyclic antidepressants, and psychotherapy to be successful in improving symptoms and reducing lesions at follow-up visits.
- Piette WW. Purpura: mechanisms and differential diagnosis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:376-389.
- Harth W, Taube KM, Gieler U. Factitious disorders in dermatology. J Dtsch Dermatol Ges. 2010;8:361-372.
- Sridharan M, Ali U, Hook CC, et al. The Mayo Clinic experience with psychogenic purpura (Gardner-Diamond syndrome). Am J Med Sci. 2019;357:411‐420.
- Gardner FH, Diamond LK. Autoerythrocyte sensitization; a form of purpura producing painful bruising following autosensitization to red blood cells in certain women. Blood. 1955;10:675-690.
- Groch GS, Finch SC, Rogoway W, et al. Studies in the pathogenesis of autoerythrocyte sensitization syndrome. Blood. 1966;28:19-33.
- Strunecká A, Krpejsová L, Palecek J, et al. Transbilayer redistribution of phosphatidylserine in erythrocytes of a patient with autoerythrocyte sensitization syndrome (psychogenic purpura). Folia Haematol Int Mag Klin Morphol Blutforsch. 1990;117:829-841.
- Merlen JF. Ecchymotic patches of the fingers and Gardner-Diamond vascular purpura. Phlebologie. 1987;40:473-487.
- Ivanov OL, Lvov AN, Michenko AV, et al. Autoerythrocyte sensitization syndrome (Gardner-Diamond syndrome): review of the literature. J Eur Acad Dermatol Venereol. 2009;23:499-504.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Hamad A, Jithpratuck W, Krishnaswamy G. Urticarial vasculitis and associated disorders. Ann Allergy Asthma Immunol. 2017;118:394-398.
- Ratnoff OD. Psychogenic purpura (autoerythrocyte sensitization): an unsolved dilemma. Am J Med. 1989;87:16N-21N.
- Block ME, Sitenga JL, Lehrer M, et al. Gardner‐Diamond syndrome: a systematic review of treatment options for a rare psychodermatological disorder. Int J Dermatol. 2019;58:782-787.
- Piette WW. Purpura: mechanisms and differential diagnosis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:376-389.
- Harth W, Taube KM, Gieler U. Factitious disorders in dermatology. J Dtsch Dermatol Ges. 2010;8:361-372.
- Sridharan M, Ali U, Hook CC, et al. The Mayo Clinic experience with psychogenic purpura (Gardner-Diamond syndrome). Am J Med Sci. 2019;357:411‐420.
- Gardner FH, Diamond LK. Autoerythrocyte sensitization; a form of purpura producing painful bruising following autosensitization to red blood cells in certain women. Blood. 1955;10:675-690.
- Groch GS, Finch SC, Rogoway W, et al. Studies in the pathogenesis of autoerythrocyte sensitization syndrome. Blood. 1966;28:19-33.
- Strunecká A, Krpejsová L, Palecek J, et al. Transbilayer redistribution of phosphatidylserine in erythrocytes of a patient with autoerythrocyte sensitization syndrome (psychogenic purpura). Folia Haematol Int Mag Klin Morphol Blutforsch. 1990;117:829-841.
- Merlen JF. Ecchymotic patches of the fingers and Gardner-Diamond vascular purpura. Phlebologie. 1987;40:473-487.
- Ivanov OL, Lvov AN, Michenko AV, et al. Autoerythrocyte sensitization syndrome (Gardner-Diamond syndrome): review of the literature. J Eur Acad Dermatol Venereol. 2009;23:499-504.
- Wetter DA, Dutz JP, Shinkai K, et al. Cutaneous vasculitis. In: Bolognia JL, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018:409-439.
- Hamad A, Jithpratuck W, Krishnaswamy G. Urticarial vasculitis and associated disorders. Ann Allergy Asthma Immunol. 2017;118:394-398.
- Ratnoff OD. Psychogenic purpura (autoerythrocyte sensitization): an unsolved dilemma. Am J Med. 1989;87:16N-21N.
- Block ME, Sitenga JL, Lehrer M, et al. Gardner‐Diamond syndrome: a systematic review of treatment options for a rare psychodermatological disorder. Int J Dermatol. 2019;58:782-787.
PRACTICE POINTS
- Psychogenic purpura is a rare vasculopathy characterized by painful recurrent episodes of purpura. It is a diagnosis of exclusion that may manifest with signs similar to cutaneous small vessel vasculitis.
- Awareness of this condition could help prevent unnecessary diagnostics, medications, and adverse events.
Surveillance for 21 Possible Effects of Endocrine Disruptors
Santé Publique France (SPF), the French national public health agency, has released the findings of the PEPS’PE study, which was launched in 2021. The study aims to prioritize, following extensive consultation, the health effects to be monitored for their potential link to endocrine disruptors (EDs). Out of 59 health effects suspected to be associated with exposure to EDs, 21 have been considered a priority for surveillance. Based on these results and others, SPF will expand the scope of the Agency’s surveillance by incorporating new pathologies.
As part of its environmental health program and the National Strategy on EDs, To incorporate new scientific knowledge, the PEPS’PE project aims to prioritize health effects related to EDs and identify health events to integrate into the agency’s current surveillance. The 59 health effects suspected to be associated with exposure to EDs were to be evaluated based on two criteria: The weight of evidence and the epidemiological and societal impact of the health effect. A diverse panel of international experts and French stakeholders in the field of EDs classified 21 health effects as a priority for surveillance.
Among these effects, six reproductive health effects are already monitored in the surveillance program: Cryptorchidism, hypospadias, early puberty, testicular cancer, alteration of sperm quality, and endometriosis. In addition, infertility and decreased fertility (which are not currently monitored for their link to EDs) have been included.
Metabolic effects (including overweight and obesity, cardiovascular diseases, type 2 diabetes, and metabolic syndrome), child neurodevelopmental disorders (including behavioral disorders, intellectual deficits, and attention-deficit disorders), cancers (including breast cancer, prostate cancer, lymphomas, and leukemias in children), and asthma have also been highlighted.
Furthermore, 22 effects were considered low priorities or deemed nonpriorities when, for example, they presented weak or moderate evidence with varying levels of interest in implementing surveillance. Finally, 16 health effects could not be prioritized because of a lack of scientific experts on these topics and a failure to achieve consensus (eg, bone disorders, adrenal disorders, and skin and eye disorders). Consensus was sought during this consultation using a Delphi method.
“These results indicate the need to expand the scope of the Agency’s surveillance beyond reproductive health, incorporating new pathologies when surveillance data are available,” SPF declared in a press release.
“With the initial decision elements obtained through this study, Santé Publique France will analyze the feasibility of implementing surveillance for effects classified as priorities.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Santé Publique France (SPF), the French national public health agency, has released the findings of the PEPS’PE study, which was launched in 2021. The study aims to prioritize, following extensive consultation, the health effects to be monitored for their potential link to endocrine disruptors (EDs). Out of 59 health effects suspected to be associated with exposure to EDs, 21 have been considered a priority for surveillance. Based on these results and others, SPF will expand the scope of the Agency’s surveillance by incorporating new pathologies.
As part of its environmental health program and the National Strategy on EDs, To incorporate new scientific knowledge, the PEPS’PE project aims to prioritize health effects related to EDs and identify health events to integrate into the agency’s current surveillance. The 59 health effects suspected to be associated with exposure to EDs were to be evaluated based on two criteria: The weight of evidence and the epidemiological and societal impact of the health effect. A diverse panel of international experts and French stakeholders in the field of EDs classified 21 health effects as a priority for surveillance.
Among these effects, six reproductive health effects are already monitored in the surveillance program: Cryptorchidism, hypospadias, early puberty, testicular cancer, alteration of sperm quality, and endometriosis. In addition, infertility and decreased fertility (which are not currently monitored for their link to EDs) have been included.
Metabolic effects (including overweight and obesity, cardiovascular diseases, type 2 diabetes, and metabolic syndrome), child neurodevelopmental disorders (including behavioral disorders, intellectual deficits, and attention-deficit disorders), cancers (including breast cancer, prostate cancer, lymphomas, and leukemias in children), and asthma have also been highlighted.
Furthermore, 22 effects were considered low priorities or deemed nonpriorities when, for example, they presented weak or moderate evidence with varying levels of interest in implementing surveillance. Finally, 16 health effects could not be prioritized because of a lack of scientific experts on these topics and a failure to achieve consensus (eg, bone disorders, adrenal disorders, and skin and eye disorders). Consensus was sought during this consultation using a Delphi method.
“These results indicate the need to expand the scope of the Agency’s surveillance beyond reproductive health, incorporating new pathologies when surveillance data are available,” SPF declared in a press release.
“With the initial decision elements obtained through this study, Santé Publique France will analyze the feasibility of implementing surveillance for effects classified as priorities.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Santé Publique France (SPF), the French national public health agency, has released the findings of the PEPS’PE study, which was launched in 2021. The study aims to prioritize, following extensive consultation, the health effects to be monitored for their potential link to endocrine disruptors (EDs). Out of 59 health effects suspected to be associated with exposure to EDs, 21 have been considered a priority for surveillance. Based on these results and others, SPF will expand the scope of the Agency’s surveillance by incorporating new pathologies.
As part of its environmental health program and the National Strategy on EDs, To incorporate new scientific knowledge, the PEPS’PE project aims to prioritize health effects related to EDs and identify health events to integrate into the agency’s current surveillance. The 59 health effects suspected to be associated with exposure to EDs were to be evaluated based on two criteria: The weight of evidence and the epidemiological and societal impact of the health effect. A diverse panel of international experts and French stakeholders in the field of EDs classified 21 health effects as a priority for surveillance.
Among these effects, six reproductive health effects are already monitored in the surveillance program: Cryptorchidism, hypospadias, early puberty, testicular cancer, alteration of sperm quality, and endometriosis. In addition, infertility and decreased fertility (which are not currently monitored for their link to EDs) have been included.
Metabolic effects (including overweight and obesity, cardiovascular diseases, type 2 diabetes, and metabolic syndrome), child neurodevelopmental disorders (including behavioral disorders, intellectual deficits, and attention-deficit disorders), cancers (including breast cancer, prostate cancer, lymphomas, and leukemias in children), and asthma have also been highlighted.
Furthermore, 22 effects were considered low priorities or deemed nonpriorities when, for example, they presented weak or moderate evidence with varying levels of interest in implementing surveillance. Finally, 16 health effects could not be prioritized because of a lack of scientific experts on these topics and a failure to achieve consensus (eg, bone disorders, adrenal disorders, and skin and eye disorders). Consensus was sought during this consultation using a Delphi method.
“These results indicate the need to expand the scope of the Agency’s surveillance beyond reproductive health, incorporating new pathologies when surveillance data are available,” SPF declared in a press release.
“With the initial decision elements obtained through this study, Santé Publique France will analyze the feasibility of implementing surveillance for effects classified as priorities.”
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
The Potential for Artificial Intelligence Tools in Residency Recruitment
According to Electronic Residency Application Service (ERAS) statistics, there were more than 1400 dermatology applicants in 2022, with an average of almost 560 applications received per program.1,2 With the goal to expand the diversity of board-certified dermatologists, there is increasing emphasis on the holistic review of applications, forgoing filtering by discrete metrics such as AOA (American Osteopathic Association) membership and US Medical Licensing Examination (USMLE) scores.3 According to the Association of American Medical Colleges, holistic review focuses on an individual applicant’s experience and unique attributes in addition to their academic achievements.4 Recent strategies to enhance the residency recruitment process have included the introduction of standardized letters of recommendation, preference signaling, and supplemental applications.5,6
Because it has become increasingly important to include applicant factors and achievements that extend beyond academics, the number of data points that are required for holistic review has expanded. If each application required 20 minutes to review, this would result in 166 total hours for complete holistic review of 500 applications. Tools that can facilitate holistic review of candidates and select applicants whose interests and career goals align with individual residency programs have the potential to optimize review. Artificial intelligence (AI) may aid in this process. This column highlights some of the published research on novel AI strategies that have the potential to impact dermatology residency recruitment.
Machine Learning to Screen Applicants
Artificial intelligence involves a machine-based system that can make decisions, predictions, and recommendations when provided a given set of human-defined objectives.7 Autonomous systems, machine learning (ML), and generative AI are examples of AI models.8 Machine learning has been explored to shorten and streamline the application review process and decrease bias. Because ML is a model in which the computer learns patterns based on large amounts of input data,9 it is possible that models could be developed and used in future cycles. Some studies found that applicants were discovered who traditionally would not have made it to the next stage of consideration based primarily on academic metrics.10,11 Burk-Rafel et al10 developed and validated an ML-based decision support tool for residency program directors to use for interview invitation decisions. The tool utilized 61 variables from ERAS data from more than 8000 applications in 3 prior application cycles at a single internal medicine residency program. An interview invitation was designated as the target outcome. Ultimately, the model would output a probability score for an interview invitation. The authors were able to tune the model to a 91% sensitivity and 85% specificity; for a pool of 2000 applicants and an invite rate of 15%, 1475 applicants would be screened out with a negative predictive value of 98% with maintenance of performance, even with removal of USMLE Step 1 examination scores. Their ML model was prospectively validated during an ongoing resident selection cycle, and when compared with human review, the AI model found an additional 20 applicants to invite for interviews. They concluded that this tool could potentially augment the human review process and reveal applicants who may have otherwise been overlooked.10
Rees and Ryder11 utilized another ML screening approach with the target outcome of ranked and matriculated compared with ranked applicants based on ERAS data using 72 unique variables for more than 5000 applicants. Their model was able to identify ranked candidates from the overall applicant pool with high accuracy; identification of ranked applicants that matriculated at the program was more modest but better than random probability.11Both the Burk-Rafel et al10 and Rees and Ryder11 models excluded some unstructured data components of the residency application, such as personal statements, medical student performance evaluation letters, transcripts, and letters of reference, that some may consider strongly in the holistic review process. Drum et al12 explored the value of extraction of this type of data. They created a program to extract “snippets” of text that pertained to values of successful residents for their internal medicine–pediatrics residency program that they previously validated via a modified Delphi method, which then were annotated by expert reviewers. Natural language processing was used to train an ML algorithm (MLA) to classify snippets into 11 value categories. Four values had more than 66% agreement with human annotation: academic strength; leadership; communication; and justice, equity, diversity, and inclusion. Although this MLA has not reached high enough levels of agreement for all the predetermined success values, the authors hope to generate a model that could produce a quantitative score to use as an initial screening tool to select applicants for interview.12 This type of analysis also could be incorporated into other MLAs for further refinement of the mentoring and application process.
Knapke et al13 evaluated the use of a natural language modeling platform to look for semantic patterns in medical school applications that could predict which students would be more likely to pursue family medicine residency, thus beginning the recruitment process even before residency application. This strategy could be particularly valuable for specialties for which there may be greater need in the workforce.
AI for Administrative Purposes
Artificial intelligence also has been used for nonapplication aspects of the residency recruitment process, such as interview scheduling. In the absence of coordinated interview release dates (as was implemented in dermatology starting in the 2020-2021 application cycle), a deluge of responses to schedule an interview comes flooding in as soon as invitations for interviewees are sent out, which can produce anxiety both for applicants and residency program staff as the schedule is sorted out and can create delays at both ends. Stephens et al14 utilized a computerized scheduling program for pediatric surgery fellowship applicants. It was used in 2016 to schedule 26 interviews, and it was found to reduce the average time to schedule an interview from 14.4 hours to 1.7 hours. It also reduced the number of email exchanges needed to finalize scheduling.14
Another aspect of residency recruitment that is amenable to AI is information gathering. Many would-be applicants turn to the internet and social media to learn about residency programs—their unique qualities, assets, and potential alignment of career goals.15 This exchange often is unidirectional, as the applicant clicks through the website searching for information. Yi et al16 explored the use of a chatbot, which mimics human conversation and exchange, on their institution’s pain fellowship website. Fellowship applicants could create specific prompts, such as “Show me faculty that trained at <applicant’s home program>,” and the chatbot would reply with the answer. The researchers sent a survey to all 258 applicants to the pain fellowship program that was completed by 48 applicants. Of these respondents, more than 70% (35/48) utilized the chatbot, and 84% (40/48) stated that they had found the information that was requested. The respondents overall found the chatbot to be a useful and positive experience.16
Specific Tools to Consider
There are some tools that are publicly available for programs and applicants to use that rely on AI.
In collaboration with ERAS and the Association of American Medical Colleges, Cortex powered by Thalamus (SJ MedConnect Inc)(https://thalamusgme.com/cortex-application-screening/) offers technology-assisted holistic review of residency and fellowship applications by utilizing natural language processing and optical character recognition to aggregate data from ERAS.
Tools also are being leveraged by applicants to help them find residency programs that fit their criteria, prepare for interviews, and complete portions of the application. Match A Resident (https://www.matcharesident.com/) is a resource for the international medical graduate community. As part of the service, the “Learn More with MARai” feature uses AI to generate information on residency programs to increase applicants’ confidence going into the interview process.17 Big Interview Medical (https://www.biginterviewmedical.com/ai-feedback), a paid interview preparation system developed by interview experts, utilizes AI to provide feedback to residents practicing for the interview process by measuring the amount of natural eye contact, language used, and pace of speech. A “Power Word” score is provided that incorporates aspects such as using filler words (“umm,” “uhh”). A Pace of Speech Tool provides rate of speaking feedback presuming that there is an ideal pace to decrease the impression that the applicant is nervous. Johnstone et al18 used ChatGPT (https://chat.openai.com/auth/login) to generate 2 personal statements for anesthesia residency applicants. Based on survey responses from 31 program directors, 22 rated the statements as good or excellent.18
Ethnical Concerns and Limitations of AI
The potential use of AI tools by residency applicants inevitably brings forth consideration of biases, ethics, and current limitations. These tools are highly dependent on the quality and quantity of data used for training and validation. Information considered valuable in the holistic review of applications includes unstructured data such as personal statements and letters of recommendation, and incorporating this information can be challenging in ML models, in contrast to discrete structured data such as grades, test scores, and awards. In addition, MLAs depend on large quantities of data to optimize performance.19 Depending on the size of the applicant pool and the amount of data available, this can present a limitation for smaller programs in developing ML tools for residency recruitment. Studies evaluating the use of AI in the residency application process often are from single institutions, and therefore generalizability is uncertain. The risk for latent bias—whereby a historical or pre-existing stereotype gets perpetuated through the system—must be considered, with the development of tools to detect and address this if found. Choosing which data to use to train the model can be tricky as well as choosing the outcome of interest. For these interventions to become more resilient, programs need to self-examine what defines their criteria for a successful match to their program to incorporate this data into their ML studies. The previously described models in this overview focused on outcomes such as whether an applicant was invited to interview, whether the applicant was ranked, and whether the applicant matriculated to their program.10,11 For supervised ML models that rely on outcomes to develop a prediction, continued research as to what outcomes represent resident success (eg, passing board certification examinations, correlation with clinical performance) would be important. There also is the possibility of applicants restructuring their applications to align with goals of an AI-assisted search and using AI to generate part or all of their application. The use of ChatGPT and other AI tools in the preparation of personal statements and curriculum vitae may provide benefits such as improved efficiency and grammar support.20 However, as use becomes more widespread, there is the potential increased similarity of personal statements and likely varied opinions on the use of such tools as writing aids.21,22 Continued efforts to develop guidance on generative AI use cases is ongoing; an example is the launch of VALID AI (https://validai.health/), a collaboration among health systems, health plans, and AI research organizations and nonprofits.23
Final Thoughts
Artificial intelligence tools may be a promising resource for residency and fellowship programs seeking to find meaningful ways to select applicants who are good matches for their training environment. Prioritizing the holistic review of applications has been promoted as a method to evaluate the applicant beyond their test scores and grades. The use of MLAs may streamline this review process, aid in scheduling interviews, and help discover trends in successful matriculants.
- Association of American Medical Colleges. ERAS® Statistics. Accessed January 16, 2024. https://www.aamc.org/data-reports/data/eras-statistics-data
- National Resident Matching Program, Data Release and ResearchCommittee: Results of the 2022 NRMP Program Director Survey. Accessed January 18, 2024. https://www.nrmp.org/wp-content/uploads/2022/09/PD-Survey-Report-2022_FINALrev.pdf
- Isaq NA, Bowers S, Chen ST. Taking a “step” toward diversity in dermatology: de-emphasizing USMLE Step 1 scores in residency applications. Int J Womens Dermatol. 2020;6:209-210. doi:10.1016/j.ijwd.2020.02.008
- Association of American Medical Colleges. Holistic review in medical school admissions. Accessed January 16, 2024. https://students-residents.aamc.org/choosing-medical-career/holistic-review-medical-school-admissions
- Association of American Medical Colleges. The MyERAS® application and program signaling for 2023-24. Accessed January 16, 2024. https://students-residents.aamc.org/applying-residencies-eras/myeras-application-and-program-signaling-2023-24
- Tavarez MM, Baghdassarian A, Bailey J, et al. A call to action for standardizing letters of recommendation. J Grad Med Educ. 2022;14:642-646. doi:10.4300/JGME-D-22-00131.1
- US Department of State. Artificial intelligence (AI). Accessed January 16, 2024. https://www.state.gov/artificial-intelligence/
- Stanford University Human-Centered Artificial Intelligence. Artificial intelligence definitions. Accessed January 16, 2024.https://hai.stanford.edu/sites/default/files/2023-03/AI-Key-Terms-Glossary-Definition.pdf
- Rajkomar A, Dean J, Kohane I. Machine learning in medicine. N Engl J Med. 2019;380:1347-1358. doi:10.1056/NEJMra1814259
- Burk-Rafel J, Reinstein I, Feng J, et al. Development and validation of a machine learning-based decision support tool for residency applicant screening and review. Acad Med. 2021;96(11S):S54-S61. doi:10.1097/ACM.0000000000004317
- Rees CA, Ryder HF. Machine learning for the prediction of ranked applicants and matriculants to an internal medicine residency program. Teach Learn Med. 2023;35:277-286. doi:10.1080/10401334.2022.2059664
- Drum B, Shi J, Peterson B, et al. Using natural language processing and machine learning to identify internal medicine-pediatrics residency values in applications. Acad Med. 2023;98:1278-1282. doi:10.1097/ACM.0000000000005352
- Knapke JM, Mount HR, McCabe E, et al. Early identification of family physicians using qualitative admissions data. Fam Med. 2023;55:245-252. doi:10.22454/FamMed.2023.596964
- Stephens CQ, Hamilton NA, Thompson AE, et al. Use of computerized interview scheduling program for pediatric surgery match applicants. J Pediatr Surg. 2017;52:1056-1059. doi:10.1016/j.jpedsurg.2017.03.033
- Nickles MA, Kulkarni V, Varghese JA, et al. Dermatology residency programs’ websites in the virtual era: a cross-sectional analysis. J Am Acad Dermatol. 2022;86:447-448. doi:10.1016/j.jaad.2021.09.064
- Yi PK, Ray ND, Segall N. A novel use of an artificially intelligent Chatbot and a live, synchronous virtual question-and answer session for fellowship recruitment. BMC Med Educ. 2023;23:152. doi:10.1186/s12909-022-03872-z
- Introducing “Learn More with MARai”—the key to understanding your target residency programs. Match A Resident website. Published September 23, 2023. Accessed January 16, 2024. https://blog.matcharesident.com/ai-powered-residency-insights/
- Johnstone RE, Neely G, Sizemore DC. Artificial intelligence softwarecan generate residency application personal statements that program directors find acceptable and difficult to distinguish from applicant compositions. J Clin Anesth. 2023;89:111185. doi:10.1016/j.jclinane.2023.111185
- Khalid N, Qayyum A, Bilal M, et al. Privacy-preserving artificial intelligence in healthcare: techniques and applications. Comput Biol Med. 2023;158:106848. doi:10.1016/j.compbiomed.2023.106848
- Birt J. How to optimize your resume for AI scanners (with tips). Indeed website. Updated December 30, 2022. Accessed January 16, 2024. https://www.indeed.com/career-advice/resumes-cover-letters/resume-ai
- Patel V, Deleonibus A, Wells MW, et al. Distinguishing authentic voices in the age of ChatGPT: comparing AI-generated and applicant-written personal statements for plastic surgery residency application. Ann Plast Surg. 2023;91:324-325. doi:10.1097/SAP.0000000000003653
- Woodfin MW. The personal statement in the age of artificial intelligence. Acad Med. 2023;98:869. doi:10.1097/ACM.0000000000005266
- Diaz N. UC Davis Health to lead new gen AI collaborative. Beckers Healthcare website. Published October 10, 2023. AccessedJanuary 16, 2024. https://www.beckershospitalreview.com/digital-health/uc-davis-health-to-lead-new-gen-ai-collaborative.html
According to Electronic Residency Application Service (ERAS) statistics, there were more than 1400 dermatology applicants in 2022, with an average of almost 560 applications received per program.1,2 With the goal to expand the diversity of board-certified dermatologists, there is increasing emphasis on the holistic review of applications, forgoing filtering by discrete metrics such as AOA (American Osteopathic Association) membership and US Medical Licensing Examination (USMLE) scores.3 According to the Association of American Medical Colleges, holistic review focuses on an individual applicant’s experience and unique attributes in addition to their academic achievements.4 Recent strategies to enhance the residency recruitment process have included the introduction of standardized letters of recommendation, preference signaling, and supplemental applications.5,6
Because it has become increasingly important to include applicant factors and achievements that extend beyond academics, the number of data points that are required for holistic review has expanded. If each application required 20 minutes to review, this would result in 166 total hours for complete holistic review of 500 applications. Tools that can facilitate holistic review of candidates and select applicants whose interests and career goals align with individual residency programs have the potential to optimize review. Artificial intelligence (AI) may aid in this process. This column highlights some of the published research on novel AI strategies that have the potential to impact dermatology residency recruitment.
Machine Learning to Screen Applicants
Artificial intelligence involves a machine-based system that can make decisions, predictions, and recommendations when provided a given set of human-defined objectives.7 Autonomous systems, machine learning (ML), and generative AI are examples of AI models.8 Machine learning has been explored to shorten and streamline the application review process and decrease bias. Because ML is a model in which the computer learns patterns based on large amounts of input data,9 it is possible that models could be developed and used in future cycles. Some studies found that applicants were discovered who traditionally would not have made it to the next stage of consideration based primarily on academic metrics.10,11 Burk-Rafel et al10 developed and validated an ML-based decision support tool for residency program directors to use for interview invitation decisions. The tool utilized 61 variables from ERAS data from more than 8000 applications in 3 prior application cycles at a single internal medicine residency program. An interview invitation was designated as the target outcome. Ultimately, the model would output a probability score for an interview invitation. The authors were able to tune the model to a 91% sensitivity and 85% specificity; for a pool of 2000 applicants and an invite rate of 15%, 1475 applicants would be screened out with a negative predictive value of 98% with maintenance of performance, even with removal of USMLE Step 1 examination scores. Their ML model was prospectively validated during an ongoing resident selection cycle, and when compared with human review, the AI model found an additional 20 applicants to invite for interviews. They concluded that this tool could potentially augment the human review process and reveal applicants who may have otherwise been overlooked.10
Rees and Ryder11 utilized another ML screening approach with the target outcome of ranked and matriculated compared with ranked applicants based on ERAS data using 72 unique variables for more than 5000 applicants. Their model was able to identify ranked candidates from the overall applicant pool with high accuracy; identification of ranked applicants that matriculated at the program was more modest but better than random probability.11Both the Burk-Rafel et al10 and Rees and Ryder11 models excluded some unstructured data components of the residency application, such as personal statements, medical student performance evaluation letters, transcripts, and letters of reference, that some may consider strongly in the holistic review process. Drum et al12 explored the value of extraction of this type of data. They created a program to extract “snippets” of text that pertained to values of successful residents for their internal medicine–pediatrics residency program that they previously validated via a modified Delphi method, which then were annotated by expert reviewers. Natural language processing was used to train an ML algorithm (MLA) to classify snippets into 11 value categories. Four values had more than 66% agreement with human annotation: academic strength; leadership; communication; and justice, equity, diversity, and inclusion. Although this MLA has not reached high enough levels of agreement for all the predetermined success values, the authors hope to generate a model that could produce a quantitative score to use as an initial screening tool to select applicants for interview.12 This type of analysis also could be incorporated into other MLAs for further refinement of the mentoring and application process.
Knapke et al13 evaluated the use of a natural language modeling platform to look for semantic patterns in medical school applications that could predict which students would be more likely to pursue family medicine residency, thus beginning the recruitment process even before residency application. This strategy could be particularly valuable for specialties for which there may be greater need in the workforce.
AI for Administrative Purposes
Artificial intelligence also has been used for nonapplication aspects of the residency recruitment process, such as interview scheduling. In the absence of coordinated interview release dates (as was implemented in dermatology starting in the 2020-2021 application cycle), a deluge of responses to schedule an interview comes flooding in as soon as invitations for interviewees are sent out, which can produce anxiety both for applicants and residency program staff as the schedule is sorted out and can create delays at both ends. Stephens et al14 utilized a computerized scheduling program for pediatric surgery fellowship applicants. It was used in 2016 to schedule 26 interviews, and it was found to reduce the average time to schedule an interview from 14.4 hours to 1.7 hours. It also reduced the number of email exchanges needed to finalize scheduling.14
Another aspect of residency recruitment that is amenable to AI is information gathering. Many would-be applicants turn to the internet and social media to learn about residency programs—their unique qualities, assets, and potential alignment of career goals.15 This exchange often is unidirectional, as the applicant clicks through the website searching for information. Yi et al16 explored the use of a chatbot, which mimics human conversation and exchange, on their institution’s pain fellowship website. Fellowship applicants could create specific prompts, such as “Show me faculty that trained at <applicant’s home program>,” and the chatbot would reply with the answer. The researchers sent a survey to all 258 applicants to the pain fellowship program that was completed by 48 applicants. Of these respondents, more than 70% (35/48) utilized the chatbot, and 84% (40/48) stated that they had found the information that was requested. The respondents overall found the chatbot to be a useful and positive experience.16
Specific Tools to Consider
There are some tools that are publicly available for programs and applicants to use that rely on AI.
In collaboration with ERAS and the Association of American Medical Colleges, Cortex powered by Thalamus (SJ MedConnect Inc)(https://thalamusgme.com/cortex-application-screening/) offers technology-assisted holistic review of residency and fellowship applications by utilizing natural language processing and optical character recognition to aggregate data from ERAS.
Tools also are being leveraged by applicants to help them find residency programs that fit their criteria, prepare for interviews, and complete portions of the application. Match A Resident (https://www.matcharesident.com/) is a resource for the international medical graduate community. As part of the service, the “Learn More with MARai” feature uses AI to generate information on residency programs to increase applicants’ confidence going into the interview process.17 Big Interview Medical (https://www.biginterviewmedical.com/ai-feedback), a paid interview preparation system developed by interview experts, utilizes AI to provide feedback to residents practicing for the interview process by measuring the amount of natural eye contact, language used, and pace of speech. A “Power Word” score is provided that incorporates aspects such as using filler words (“umm,” “uhh”). A Pace of Speech Tool provides rate of speaking feedback presuming that there is an ideal pace to decrease the impression that the applicant is nervous. Johnstone et al18 used ChatGPT (https://chat.openai.com/auth/login) to generate 2 personal statements for anesthesia residency applicants. Based on survey responses from 31 program directors, 22 rated the statements as good or excellent.18
Ethnical Concerns and Limitations of AI
The potential use of AI tools by residency applicants inevitably brings forth consideration of biases, ethics, and current limitations. These tools are highly dependent on the quality and quantity of data used for training and validation. Information considered valuable in the holistic review of applications includes unstructured data such as personal statements and letters of recommendation, and incorporating this information can be challenging in ML models, in contrast to discrete structured data such as grades, test scores, and awards. In addition, MLAs depend on large quantities of data to optimize performance.19 Depending on the size of the applicant pool and the amount of data available, this can present a limitation for smaller programs in developing ML tools for residency recruitment. Studies evaluating the use of AI in the residency application process often are from single institutions, and therefore generalizability is uncertain. The risk for latent bias—whereby a historical or pre-existing stereotype gets perpetuated through the system—must be considered, with the development of tools to detect and address this if found. Choosing which data to use to train the model can be tricky as well as choosing the outcome of interest. For these interventions to become more resilient, programs need to self-examine what defines their criteria for a successful match to their program to incorporate this data into their ML studies. The previously described models in this overview focused on outcomes such as whether an applicant was invited to interview, whether the applicant was ranked, and whether the applicant matriculated to their program.10,11 For supervised ML models that rely on outcomes to develop a prediction, continued research as to what outcomes represent resident success (eg, passing board certification examinations, correlation with clinical performance) would be important. There also is the possibility of applicants restructuring their applications to align with goals of an AI-assisted search and using AI to generate part or all of their application. The use of ChatGPT and other AI tools in the preparation of personal statements and curriculum vitae may provide benefits such as improved efficiency and grammar support.20 However, as use becomes more widespread, there is the potential increased similarity of personal statements and likely varied opinions on the use of such tools as writing aids.21,22 Continued efforts to develop guidance on generative AI use cases is ongoing; an example is the launch of VALID AI (https://validai.health/), a collaboration among health systems, health plans, and AI research organizations and nonprofits.23
Final Thoughts
Artificial intelligence tools may be a promising resource for residency and fellowship programs seeking to find meaningful ways to select applicants who are good matches for their training environment. Prioritizing the holistic review of applications has been promoted as a method to evaluate the applicant beyond their test scores and grades. The use of MLAs may streamline this review process, aid in scheduling interviews, and help discover trends in successful matriculants.
According to Electronic Residency Application Service (ERAS) statistics, there were more than 1400 dermatology applicants in 2022, with an average of almost 560 applications received per program.1,2 With the goal to expand the diversity of board-certified dermatologists, there is increasing emphasis on the holistic review of applications, forgoing filtering by discrete metrics such as AOA (American Osteopathic Association) membership and US Medical Licensing Examination (USMLE) scores.3 According to the Association of American Medical Colleges, holistic review focuses on an individual applicant’s experience and unique attributes in addition to their academic achievements.4 Recent strategies to enhance the residency recruitment process have included the introduction of standardized letters of recommendation, preference signaling, and supplemental applications.5,6
Because it has become increasingly important to include applicant factors and achievements that extend beyond academics, the number of data points that are required for holistic review has expanded. If each application required 20 minutes to review, this would result in 166 total hours for complete holistic review of 500 applications. Tools that can facilitate holistic review of candidates and select applicants whose interests and career goals align with individual residency programs have the potential to optimize review. Artificial intelligence (AI) may aid in this process. This column highlights some of the published research on novel AI strategies that have the potential to impact dermatology residency recruitment.
Machine Learning to Screen Applicants
Artificial intelligence involves a machine-based system that can make decisions, predictions, and recommendations when provided a given set of human-defined objectives.7 Autonomous systems, machine learning (ML), and generative AI are examples of AI models.8 Machine learning has been explored to shorten and streamline the application review process and decrease bias. Because ML is a model in which the computer learns patterns based on large amounts of input data,9 it is possible that models could be developed and used in future cycles. Some studies found that applicants were discovered who traditionally would not have made it to the next stage of consideration based primarily on academic metrics.10,11 Burk-Rafel et al10 developed and validated an ML-based decision support tool for residency program directors to use for interview invitation decisions. The tool utilized 61 variables from ERAS data from more than 8000 applications in 3 prior application cycles at a single internal medicine residency program. An interview invitation was designated as the target outcome. Ultimately, the model would output a probability score for an interview invitation. The authors were able to tune the model to a 91% sensitivity and 85% specificity; for a pool of 2000 applicants and an invite rate of 15%, 1475 applicants would be screened out with a negative predictive value of 98% with maintenance of performance, even with removal of USMLE Step 1 examination scores. Their ML model was prospectively validated during an ongoing resident selection cycle, and when compared with human review, the AI model found an additional 20 applicants to invite for interviews. They concluded that this tool could potentially augment the human review process and reveal applicants who may have otherwise been overlooked.10
Rees and Ryder11 utilized another ML screening approach with the target outcome of ranked and matriculated compared with ranked applicants based on ERAS data using 72 unique variables for more than 5000 applicants. Their model was able to identify ranked candidates from the overall applicant pool with high accuracy; identification of ranked applicants that matriculated at the program was more modest but better than random probability.11Both the Burk-Rafel et al10 and Rees and Ryder11 models excluded some unstructured data components of the residency application, such as personal statements, medical student performance evaluation letters, transcripts, and letters of reference, that some may consider strongly in the holistic review process. Drum et al12 explored the value of extraction of this type of data. They created a program to extract “snippets” of text that pertained to values of successful residents for their internal medicine–pediatrics residency program that they previously validated via a modified Delphi method, which then were annotated by expert reviewers. Natural language processing was used to train an ML algorithm (MLA) to classify snippets into 11 value categories. Four values had more than 66% agreement with human annotation: academic strength; leadership; communication; and justice, equity, diversity, and inclusion. Although this MLA has not reached high enough levels of agreement for all the predetermined success values, the authors hope to generate a model that could produce a quantitative score to use as an initial screening tool to select applicants for interview.12 This type of analysis also could be incorporated into other MLAs for further refinement of the mentoring and application process.
Knapke et al13 evaluated the use of a natural language modeling platform to look for semantic patterns in medical school applications that could predict which students would be more likely to pursue family medicine residency, thus beginning the recruitment process even before residency application. This strategy could be particularly valuable for specialties for which there may be greater need in the workforce.
AI for Administrative Purposes
Artificial intelligence also has been used for nonapplication aspects of the residency recruitment process, such as interview scheduling. In the absence of coordinated interview release dates (as was implemented in dermatology starting in the 2020-2021 application cycle), a deluge of responses to schedule an interview comes flooding in as soon as invitations for interviewees are sent out, which can produce anxiety both for applicants and residency program staff as the schedule is sorted out and can create delays at both ends. Stephens et al14 utilized a computerized scheduling program for pediatric surgery fellowship applicants. It was used in 2016 to schedule 26 interviews, and it was found to reduce the average time to schedule an interview from 14.4 hours to 1.7 hours. It also reduced the number of email exchanges needed to finalize scheduling.14
Another aspect of residency recruitment that is amenable to AI is information gathering. Many would-be applicants turn to the internet and social media to learn about residency programs—their unique qualities, assets, and potential alignment of career goals.15 This exchange often is unidirectional, as the applicant clicks through the website searching for information. Yi et al16 explored the use of a chatbot, which mimics human conversation and exchange, on their institution’s pain fellowship website. Fellowship applicants could create specific prompts, such as “Show me faculty that trained at <applicant’s home program>,” and the chatbot would reply with the answer. The researchers sent a survey to all 258 applicants to the pain fellowship program that was completed by 48 applicants. Of these respondents, more than 70% (35/48) utilized the chatbot, and 84% (40/48) stated that they had found the information that was requested. The respondents overall found the chatbot to be a useful and positive experience.16
Specific Tools to Consider
There are some tools that are publicly available for programs and applicants to use that rely on AI.
In collaboration with ERAS and the Association of American Medical Colleges, Cortex powered by Thalamus (SJ MedConnect Inc)(https://thalamusgme.com/cortex-application-screening/) offers technology-assisted holistic review of residency and fellowship applications by utilizing natural language processing and optical character recognition to aggregate data from ERAS.
Tools also are being leveraged by applicants to help them find residency programs that fit their criteria, prepare for interviews, and complete portions of the application. Match A Resident (https://www.matcharesident.com/) is a resource for the international medical graduate community. As part of the service, the “Learn More with MARai” feature uses AI to generate information on residency programs to increase applicants’ confidence going into the interview process.17 Big Interview Medical (https://www.biginterviewmedical.com/ai-feedback), a paid interview preparation system developed by interview experts, utilizes AI to provide feedback to residents practicing for the interview process by measuring the amount of natural eye contact, language used, and pace of speech. A “Power Word” score is provided that incorporates aspects such as using filler words (“umm,” “uhh”). A Pace of Speech Tool provides rate of speaking feedback presuming that there is an ideal pace to decrease the impression that the applicant is nervous. Johnstone et al18 used ChatGPT (https://chat.openai.com/auth/login) to generate 2 personal statements for anesthesia residency applicants. Based on survey responses from 31 program directors, 22 rated the statements as good or excellent.18
Ethnical Concerns and Limitations of AI
The potential use of AI tools by residency applicants inevitably brings forth consideration of biases, ethics, and current limitations. These tools are highly dependent on the quality and quantity of data used for training and validation. Information considered valuable in the holistic review of applications includes unstructured data such as personal statements and letters of recommendation, and incorporating this information can be challenging in ML models, in contrast to discrete structured data such as grades, test scores, and awards. In addition, MLAs depend on large quantities of data to optimize performance.19 Depending on the size of the applicant pool and the amount of data available, this can present a limitation for smaller programs in developing ML tools for residency recruitment. Studies evaluating the use of AI in the residency application process often are from single institutions, and therefore generalizability is uncertain. The risk for latent bias—whereby a historical or pre-existing stereotype gets perpetuated through the system—must be considered, with the development of tools to detect and address this if found. Choosing which data to use to train the model can be tricky as well as choosing the outcome of interest. For these interventions to become more resilient, programs need to self-examine what defines their criteria for a successful match to their program to incorporate this data into their ML studies. The previously described models in this overview focused on outcomes such as whether an applicant was invited to interview, whether the applicant was ranked, and whether the applicant matriculated to their program.10,11 For supervised ML models that rely on outcomes to develop a prediction, continued research as to what outcomes represent resident success (eg, passing board certification examinations, correlation with clinical performance) would be important. There also is the possibility of applicants restructuring their applications to align with goals of an AI-assisted search and using AI to generate part or all of their application. The use of ChatGPT and other AI tools in the preparation of personal statements and curriculum vitae may provide benefits such as improved efficiency and grammar support.20 However, as use becomes more widespread, there is the potential increased similarity of personal statements and likely varied opinions on the use of such tools as writing aids.21,22 Continued efforts to develop guidance on generative AI use cases is ongoing; an example is the launch of VALID AI (https://validai.health/), a collaboration among health systems, health plans, and AI research organizations and nonprofits.23
Final Thoughts
Artificial intelligence tools may be a promising resource for residency and fellowship programs seeking to find meaningful ways to select applicants who are good matches for their training environment. Prioritizing the holistic review of applications has been promoted as a method to evaluate the applicant beyond their test scores and grades. The use of MLAs may streamline this review process, aid in scheduling interviews, and help discover trends in successful matriculants.
- Association of American Medical Colleges. ERAS® Statistics. Accessed January 16, 2024. https://www.aamc.org/data-reports/data/eras-statistics-data
- National Resident Matching Program, Data Release and ResearchCommittee: Results of the 2022 NRMP Program Director Survey. Accessed January 18, 2024. https://www.nrmp.org/wp-content/uploads/2022/09/PD-Survey-Report-2022_FINALrev.pdf
- Isaq NA, Bowers S, Chen ST. Taking a “step” toward diversity in dermatology: de-emphasizing USMLE Step 1 scores in residency applications. Int J Womens Dermatol. 2020;6:209-210. doi:10.1016/j.ijwd.2020.02.008
- Association of American Medical Colleges. Holistic review in medical school admissions. Accessed January 16, 2024. https://students-residents.aamc.org/choosing-medical-career/holistic-review-medical-school-admissions
- Association of American Medical Colleges. The MyERAS® application and program signaling for 2023-24. Accessed January 16, 2024. https://students-residents.aamc.org/applying-residencies-eras/myeras-application-and-program-signaling-2023-24
- Tavarez MM, Baghdassarian A, Bailey J, et al. A call to action for standardizing letters of recommendation. J Grad Med Educ. 2022;14:642-646. doi:10.4300/JGME-D-22-00131.1
- US Department of State. Artificial intelligence (AI). Accessed January 16, 2024. https://www.state.gov/artificial-intelligence/
- Stanford University Human-Centered Artificial Intelligence. Artificial intelligence definitions. Accessed January 16, 2024.https://hai.stanford.edu/sites/default/files/2023-03/AI-Key-Terms-Glossary-Definition.pdf
- Rajkomar A, Dean J, Kohane I. Machine learning in medicine. N Engl J Med. 2019;380:1347-1358. doi:10.1056/NEJMra1814259
- Burk-Rafel J, Reinstein I, Feng J, et al. Development and validation of a machine learning-based decision support tool for residency applicant screening and review. Acad Med. 2021;96(11S):S54-S61. doi:10.1097/ACM.0000000000004317
- Rees CA, Ryder HF. Machine learning for the prediction of ranked applicants and matriculants to an internal medicine residency program. Teach Learn Med. 2023;35:277-286. doi:10.1080/10401334.2022.2059664
- Drum B, Shi J, Peterson B, et al. Using natural language processing and machine learning to identify internal medicine-pediatrics residency values in applications. Acad Med. 2023;98:1278-1282. doi:10.1097/ACM.0000000000005352
- Knapke JM, Mount HR, McCabe E, et al. Early identification of family physicians using qualitative admissions data. Fam Med. 2023;55:245-252. doi:10.22454/FamMed.2023.596964
- Stephens CQ, Hamilton NA, Thompson AE, et al. Use of computerized interview scheduling program for pediatric surgery match applicants. J Pediatr Surg. 2017;52:1056-1059. doi:10.1016/j.jpedsurg.2017.03.033
- Nickles MA, Kulkarni V, Varghese JA, et al. Dermatology residency programs’ websites in the virtual era: a cross-sectional analysis. J Am Acad Dermatol. 2022;86:447-448. doi:10.1016/j.jaad.2021.09.064
- Yi PK, Ray ND, Segall N. A novel use of an artificially intelligent Chatbot and a live, synchronous virtual question-and answer session for fellowship recruitment. BMC Med Educ. 2023;23:152. doi:10.1186/s12909-022-03872-z
- Introducing “Learn More with MARai”—the key to understanding your target residency programs. Match A Resident website. Published September 23, 2023. Accessed January 16, 2024. https://blog.matcharesident.com/ai-powered-residency-insights/
- Johnstone RE, Neely G, Sizemore DC. Artificial intelligence softwarecan generate residency application personal statements that program directors find acceptable and difficult to distinguish from applicant compositions. J Clin Anesth. 2023;89:111185. doi:10.1016/j.jclinane.2023.111185
- Khalid N, Qayyum A, Bilal M, et al. Privacy-preserving artificial intelligence in healthcare: techniques and applications. Comput Biol Med. 2023;158:106848. doi:10.1016/j.compbiomed.2023.106848
- Birt J. How to optimize your resume for AI scanners (with tips). Indeed website. Updated December 30, 2022. Accessed January 16, 2024. https://www.indeed.com/career-advice/resumes-cover-letters/resume-ai
- Patel V, Deleonibus A, Wells MW, et al. Distinguishing authentic voices in the age of ChatGPT: comparing AI-generated and applicant-written personal statements for plastic surgery residency application. Ann Plast Surg. 2023;91:324-325. doi:10.1097/SAP.0000000000003653
- Woodfin MW. The personal statement in the age of artificial intelligence. Acad Med. 2023;98:869. doi:10.1097/ACM.0000000000005266
- Diaz N. UC Davis Health to lead new gen AI collaborative. Beckers Healthcare website. Published October 10, 2023. AccessedJanuary 16, 2024. https://www.beckershospitalreview.com/digital-health/uc-davis-health-to-lead-new-gen-ai-collaborative.html
- Association of American Medical Colleges. ERAS® Statistics. Accessed January 16, 2024. https://www.aamc.org/data-reports/data/eras-statistics-data
- National Resident Matching Program, Data Release and ResearchCommittee: Results of the 2022 NRMP Program Director Survey. Accessed January 18, 2024. https://www.nrmp.org/wp-content/uploads/2022/09/PD-Survey-Report-2022_FINALrev.pdf
- Isaq NA, Bowers S, Chen ST. Taking a “step” toward diversity in dermatology: de-emphasizing USMLE Step 1 scores in residency applications. Int J Womens Dermatol. 2020;6:209-210. doi:10.1016/j.ijwd.2020.02.008
- Association of American Medical Colleges. Holistic review in medical school admissions. Accessed January 16, 2024. https://students-residents.aamc.org/choosing-medical-career/holistic-review-medical-school-admissions
- Association of American Medical Colleges. The MyERAS® application and program signaling for 2023-24. Accessed January 16, 2024. https://students-residents.aamc.org/applying-residencies-eras/myeras-application-and-program-signaling-2023-24
- Tavarez MM, Baghdassarian A, Bailey J, et al. A call to action for standardizing letters of recommendation. J Grad Med Educ. 2022;14:642-646. doi:10.4300/JGME-D-22-00131.1
- US Department of State. Artificial intelligence (AI). Accessed January 16, 2024. https://www.state.gov/artificial-intelligence/
- Stanford University Human-Centered Artificial Intelligence. Artificial intelligence definitions. Accessed January 16, 2024.https://hai.stanford.edu/sites/default/files/2023-03/AI-Key-Terms-Glossary-Definition.pdf
- Rajkomar A, Dean J, Kohane I. Machine learning in medicine. N Engl J Med. 2019;380:1347-1358. doi:10.1056/NEJMra1814259
- Burk-Rafel J, Reinstein I, Feng J, et al. Development and validation of a machine learning-based decision support tool for residency applicant screening and review. Acad Med. 2021;96(11S):S54-S61. doi:10.1097/ACM.0000000000004317
- Rees CA, Ryder HF. Machine learning for the prediction of ranked applicants and matriculants to an internal medicine residency program. Teach Learn Med. 2023;35:277-286. doi:10.1080/10401334.2022.2059664
- Drum B, Shi J, Peterson B, et al. Using natural language processing and machine learning to identify internal medicine-pediatrics residency values in applications. Acad Med. 2023;98:1278-1282. doi:10.1097/ACM.0000000000005352
- Knapke JM, Mount HR, McCabe E, et al. Early identification of family physicians using qualitative admissions data. Fam Med. 2023;55:245-252. doi:10.22454/FamMed.2023.596964
- Stephens CQ, Hamilton NA, Thompson AE, et al. Use of computerized interview scheduling program for pediatric surgery match applicants. J Pediatr Surg. 2017;52:1056-1059. doi:10.1016/j.jpedsurg.2017.03.033
- Nickles MA, Kulkarni V, Varghese JA, et al. Dermatology residency programs’ websites in the virtual era: a cross-sectional analysis. J Am Acad Dermatol. 2022;86:447-448. doi:10.1016/j.jaad.2021.09.064
- Yi PK, Ray ND, Segall N. A novel use of an artificially intelligent Chatbot and a live, synchronous virtual question-and answer session for fellowship recruitment. BMC Med Educ. 2023;23:152. doi:10.1186/s12909-022-03872-z
- Introducing “Learn More with MARai”—the key to understanding your target residency programs. Match A Resident website. Published September 23, 2023. Accessed January 16, 2024. https://blog.matcharesident.com/ai-powered-residency-insights/
- Johnstone RE, Neely G, Sizemore DC. Artificial intelligence softwarecan generate residency application personal statements that program directors find acceptable and difficult to distinguish from applicant compositions. J Clin Anesth. 2023;89:111185. doi:10.1016/j.jclinane.2023.111185
- Khalid N, Qayyum A, Bilal M, et al. Privacy-preserving artificial intelligence in healthcare: techniques and applications. Comput Biol Med. 2023;158:106848. doi:10.1016/j.compbiomed.2023.106848
- Birt J. How to optimize your resume for AI scanners (with tips). Indeed website. Updated December 30, 2022. Accessed January 16, 2024. https://www.indeed.com/career-advice/resumes-cover-letters/resume-ai
- Patel V, Deleonibus A, Wells MW, et al. Distinguishing authentic voices in the age of ChatGPT: comparing AI-generated and applicant-written personal statements for plastic surgery residency application. Ann Plast Surg. 2023;91:324-325. doi:10.1097/SAP.0000000000003653
- Woodfin MW. The personal statement in the age of artificial intelligence. Acad Med. 2023;98:869. doi:10.1097/ACM.0000000000005266
- Diaz N. UC Davis Health to lead new gen AI collaborative. Beckers Healthcare website. Published October 10, 2023. AccessedJanuary 16, 2024. https://www.beckershospitalreview.com/digital-health/uc-davis-health-to-lead-new-gen-ai-collaborative.html
Practice Points
- Artificial intelligence solutions may increase the efficiency of the holistic review process and enhance the opportunity to find applicants who may have been overlooked by a traditional review process.
- Artificial intelligence support also may be utilized by applicants to aid in discovering training programs that fit their interests, practice interview strategies, and refine their written application.
Ob.Gyns. Face ‘Occupational Crisis’ Navigating Abortion Ban
A 14-year-old girl arrived at a South Carolina clinic just one day after the state’s anti-abortion law would have allowed her to terminate a pregnancy in instances of rape or incest.
Angela Dempsey-Fanning, MD, MPH, an ob.gyn. in Charleston, had to inform the teenager, a victim of incest, that she could not legally provide abortion care, so the girl and her mother decided to seek treatment in a different state.
“I couldn’t shake the sense that so many principles of medical ethics were being violated in denying care to her,” said Dr. Dempsey-Fanning, president of the Society of Family Planning, a nonprofit that advocates for abortion access. “When I interact with patients in these situations ... I carry the emotional and mental burden for weeks.”
South Carolina is one of 16 states to put in place severe abortion restrictions in the wake of the US Supreme Court ruling in June 2022 on the Dobbs v. Jackson Women’s Health Organization case that overturned Roe v. Wade.
according to a study recently published in the JAMA Network Open.
Public discourse on the Dobbs v. Jackson ruling has mostly centered on the impact to patients, according to Mara Buchbinder, PhD, professor and vice chair in the Department of Social Medicine at University of North Carolina Chapel Hill School of Medicine, and a coauthor of the study.
“We were interested in what the impacts would be for the obstetric workforce as well,” she said.
In 2022 and 2023, Dr. Buchbinder and her colleagues interviewed 54 ob.gyns. practicing in 13 states where abortion had become illegal with limited exceptions, including Texas, West Virginia, and South Dakota.
Clinicians who participated in the study described instances in which the state restrictions on abortion forced them to delay what they deemed to be medically necessary care until a patient faced severe complications or even death. More than 90% reported moral distress concerning situations where legal constraints prevented them or their colleagues from following clinical standards.
“You have somebody hemorrhaging with an intrauterine pregnancy with a heartbeat ... I [didn’t yet] have legal coverage for that, but there’s only so many times you can transfuse somebody and they’re begging for their life before you say, ‘This is unconscionable,’ ” one clinician reported to researchers.
Another clinician said, “Is a 5% risk of death enough? Does it take 20%? Does it take 50%? What is enough legally?”
The US Department of Health and Human Services has announced a new team to ensure hospitals in all states comply with the Emergency Medical Treatment and Labor Act, which, according to the Biden administration, includes emergency abortions. Still, some hospitals may not have clear policies that define pregnancy-related emergencies, making it challenging for clinicians to feel protected in clinically complex situations.
The study also highlighted aiding and abetting clauses, which prevent ob.gyns. from providing referrals for abortions or discussing the option with patients. Participants described the limitations as undermining their medical expertise.
“Some of the harm that is done to these ob.gyns. is not only from the laws themselves, but from their own institutions,” Dr. Buchbinder said. “Hospitals have to decide, ‘what does this law mean and how are we going to put it to practice here?’ ”
Angela Hawkins, MD, a hospitalist practicing in Oklahoma, encountered a patient who was experiencing an obvious miscarriage. But because the situation could not yet be established as life-threatening, Dr. Hawkins felt that she could not intervene.
“There are things I know are straightforward and I would’ve handled them completely differently in the past,” Dr. Hawkins said, adding that she needed to seek reassurance from her hospital employer that she would not face legal ramifications if she provided care.
“It’s frustrating to know that this is medicine and I can’t practice it without calling legal and ethics in the middle of the night,” said Dr. Hawkins, chair of the Oklahoma section of the American College of Obstetrics and Gynecology.
Still, more than half of Oklahoma’s 77 counties are considered maternity care deserts, meaning they have little to no obstetric services available for pregnant patients. Dr. Hawkins recently completed her own survey of practicing ob.gyns. in the state. In soon-to-be published research, almost 60% of the 63 respondents reported thinking about leaving or were planning to leave the state to practice in areas that are less restrictive.
“That’s very concerning to the ob.gyns. that are left,” she said. “I feel like, if everyone leaves, who is left to take care of the patients?”
The study in JAMA Network Open also highlighted that 11% of participants had moved their practices to less restrictive states with stronger abortion protections.
In addition to losing existing clinicians, the laws have made it difficult for medical centers to recruit new ones, according to Kavita Shah Arora, MD, MBE, MS, director for Division of General Obstetrics, Gynecology, and Midwifery at the University of North Carolina at Chapel Hill, and a coauthor of the study. North Carolina enacted a new law in July 2023 that reduced the time allowed for an abortion from 20 weeks to 12 weeks under most circumstances.
“Our department faces new challenges in recruitment and retention being in a restrictive state that we haven’t had to deal with before,” Dr. Arora said. “It’s impacting how medical students choose which residency programs to apply to.”
Ob.gyns. may not be the only clinicians who feel the effect of laws restricting abortion, according to Deborah Nucatola, MD, chief medical officer of Planned Parenthood Great Northwest, Hawai’i, Alaska, Indiana, Kentucky.
Patients who live in areas with limited access to obstetrics services often present to urgent care facilities or emergency departments for medical care that are staffed with family, internal medicine, and emergency physicians, Dr. Nucatola said.
“I don’t want anyone by any means to think this is isolated to one specialty,” said Dr. Nucatola, who was not involved with the study. “It’s going to affect everyone who cares for these patients; you lose the ability to use your medical knowledge and then have to navigate this legal restriction that doesn’t correlate with anything that happens in medicine.”
Dr. Dempsey-Fanning’s 14-year-old patient did eventually receive abortion care outside of South Carolina. Dr. Dempsey-Fanning said that she and her colleagues have spent hours coordinating for patients to receive care in a different state. Then, patients and their families must come up with the money for travel and any missed work to get to another clinician working where abortion is legal.
Despite this, she said, “You are left still feeling as though you abandoned this patient in many practical ways.
“I know I weigh the decision about my future practice almost daily, wondering how long I can stay and keep fighting for patients in an environment ripe with fear, worry, and an overriding sense of injustice,” said Dr. Dempsey-Fanning.
The study authors and experts quoted in the story report no relevant disclosures.
A version of this article appeared on Medscape.com.
A 14-year-old girl arrived at a South Carolina clinic just one day after the state’s anti-abortion law would have allowed her to terminate a pregnancy in instances of rape or incest.
Angela Dempsey-Fanning, MD, MPH, an ob.gyn. in Charleston, had to inform the teenager, a victim of incest, that she could not legally provide abortion care, so the girl and her mother decided to seek treatment in a different state.
“I couldn’t shake the sense that so many principles of medical ethics were being violated in denying care to her,” said Dr. Dempsey-Fanning, president of the Society of Family Planning, a nonprofit that advocates for abortion access. “When I interact with patients in these situations ... I carry the emotional and mental burden for weeks.”
South Carolina is one of 16 states to put in place severe abortion restrictions in the wake of the US Supreme Court ruling in June 2022 on the Dobbs v. Jackson Women’s Health Organization case that overturned Roe v. Wade.
according to a study recently published in the JAMA Network Open.
Public discourse on the Dobbs v. Jackson ruling has mostly centered on the impact to patients, according to Mara Buchbinder, PhD, professor and vice chair in the Department of Social Medicine at University of North Carolina Chapel Hill School of Medicine, and a coauthor of the study.
“We were interested in what the impacts would be for the obstetric workforce as well,” she said.
In 2022 and 2023, Dr. Buchbinder and her colleagues interviewed 54 ob.gyns. practicing in 13 states where abortion had become illegal with limited exceptions, including Texas, West Virginia, and South Dakota.
Clinicians who participated in the study described instances in which the state restrictions on abortion forced them to delay what they deemed to be medically necessary care until a patient faced severe complications or even death. More than 90% reported moral distress concerning situations where legal constraints prevented them or their colleagues from following clinical standards.
“You have somebody hemorrhaging with an intrauterine pregnancy with a heartbeat ... I [didn’t yet] have legal coverage for that, but there’s only so many times you can transfuse somebody and they’re begging for their life before you say, ‘This is unconscionable,’ ” one clinician reported to researchers.
Another clinician said, “Is a 5% risk of death enough? Does it take 20%? Does it take 50%? What is enough legally?”
The US Department of Health and Human Services has announced a new team to ensure hospitals in all states comply with the Emergency Medical Treatment and Labor Act, which, according to the Biden administration, includes emergency abortions. Still, some hospitals may not have clear policies that define pregnancy-related emergencies, making it challenging for clinicians to feel protected in clinically complex situations.
The study also highlighted aiding and abetting clauses, which prevent ob.gyns. from providing referrals for abortions or discussing the option with patients. Participants described the limitations as undermining their medical expertise.
“Some of the harm that is done to these ob.gyns. is not only from the laws themselves, but from their own institutions,” Dr. Buchbinder said. “Hospitals have to decide, ‘what does this law mean and how are we going to put it to practice here?’ ”
Angela Hawkins, MD, a hospitalist practicing in Oklahoma, encountered a patient who was experiencing an obvious miscarriage. But because the situation could not yet be established as life-threatening, Dr. Hawkins felt that she could not intervene.
“There are things I know are straightforward and I would’ve handled them completely differently in the past,” Dr. Hawkins said, adding that she needed to seek reassurance from her hospital employer that she would not face legal ramifications if she provided care.
“It’s frustrating to know that this is medicine and I can’t practice it without calling legal and ethics in the middle of the night,” said Dr. Hawkins, chair of the Oklahoma section of the American College of Obstetrics and Gynecology.
Still, more than half of Oklahoma’s 77 counties are considered maternity care deserts, meaning they have little to no obstetric services available for pregnant patients. Dr. Hawkins recently completed her own survey of practicing ob.gyns. in the state. In soon-to-be published research, almost 60% of the 63 respondents reported thinking about leaving or were planning to leave the state to practice in areas that are less restrictive.
“That’s very concerning to the ob.gyns. that are left,” she said. “I feel like, if everyone leaves, who is left to take care of the patients?”
The study in JAMA Network Open also highlighted that 11% of participants had moved their practices to less restrictive states with stronger abortion protections.
In addition to losing existing clinicians, the laws have made it difficult for medical centers to recruit new ones, according to Kavita Shah Arora, MD, MBE, MS, director for Division of General Obstetrics, Gynecology, and Midwifery at the University of North Carolina at Chapel Hill, and a coauthor of the study. North Carolina enacted a new law in July 2023 that reduced the time allowed for an abortion from 20 weeks to 12 weeks under most circumstances.
“Our department faces new challenges in recruitment and retention being in a restrictive state that we haven’t had to deal with before,” Dr. Arora said. “It’s impacting how medical students choose which residency programs to apply to.”
Ob.gyns. may not be the only clinicians who feel the effect of laws restricting abortion, according to Deborah Nucatola, MD, chief medical officer of Planned Parenthood Great Northwest, Hawai’i, Alaska, Indiana, Kentucky.
Patients who live in areas with limited access to obstetrics services often present to urgent care facilities or emergency departments for medical care that are staffed with family, internal medicine, and emergency physicians, Dr. Nucatola said.
“I don’t want anyone by any means to think this is isolated to one specialty,” said Dr. Nucatola, who was not involved with the study. “It’s going to affect everyone who cares for these patients; you lose the ability to use your medical knowledge and then have to navigate this legal restriction that doesn’t correlate with anything that happens in medicine.”
Dr. Dempsey-Fanning’s 14-year-old patient did eventually receive abortion care outside of South Carolina. Dr. Dempsey-Fanning said that she and her colleagues have spent hours coordinating for patients to receive care in a different state. Then, patients and their families must come up with the money for travel and any missed work to get to another clinician working where abortion is legal.
Despite this, she said, “You are left still feeling as though you abandoned this patient in many practical ways.
“I know I weigh the decision about my future practice almost daily, wondering how long I can stay and keep fighting for patients in an environment ripe with fear, worry, and an overriding sense of injustice,” said Dr. Dempsey-Fanning.
The study authors and experts quoted in the story report no relevant disclosures.
A version of this article appeared on Medscape.com.
A 14-year-old girl arrived at a South Carolina clinic just one day after the state’s anti-abortion law would have allowed her to terminate a pregnancy in instances of rape or incest.
Angela Dempsey-Fanning, MD, MPH, an ob.gyn. in Charleston, had to inform the teenager, a victim of incest, that she could not legally provide abortion care, so the girl and her mother decided to seek treatment in a different state.
“I couldn’t shake the sense that so many principles of medical ethics were being violated in denying care to her,” said Dr. Dempsey-Fanning, president of the Society of Family Planning, a nonprofit that advocates for abortion access. “When I interact with patients in these situations ... I carry the emotional and mental burden for weeks.”
South Carolina is one of 16 states to put in place severe abortion restrictions in the wake of the US Supreme Court ruling in June 2022 on the Dobbs v. Jackson Women’s Health Organization case that overturned Roe v. Wade.
according to a study recently published in the JAMA Network Open.
Public discourse on the Dobbs v. Jackson ruling has mostly centered on the impact to patients, according to Mara Buchbinder, PhD, professor and vice chair in the Department of Social Medicine at University of North Carolina Chapel Hill School of Medicine, and a coauthor of the study.
“We were interested in what the impacts would be for the obstetric workforce as well,” she said.
In 2022 and 2023, Dr. Buchbinder and her colleagues interviewed 54 ob.gyns. practicing in 13 states where abortion had become illegal with limited exceptions, including Texas, West Virginia, and South Dakota.
Clinicians who participated in the study described instances in which the state restrictions on abortion forced them to delay what they deemed to be medically necessary care until a patient faced severe complications or even death. More than 90% reported moral distress concerning situations where legal constraints prevented them or their colleagues from following clinical standards.
“You have somebody hemorrhaging with an intrauterine pregnancy with a heartbeat ... I [didn’t yet] have legal coverage for that, but there’s only so many times you can transfuse somebody and they’re begging for their life before you say, ‘This is unconscionable,’ ” one clinician reported to researchers.
Another clinician said, “Is a 5% risk of death enough? Does it take 20%? Does it take 50%? What is enough legally?”
The US Department of Health and Human Services has announced a new team to ensure hospitals in all states comply with the Emergency Medical Treatment and Labor Act, which, according to the Biden administration, includes emergency abortions. Still, some hospitals may not have clear policies that define pregnancy-related emergencies, making it challenging for clinicians to feel protected in clinically complex situations.
The study also highlighted aiding and abetting clauses, which prevent ob.gyns. from providing referrals for abortions or discussing the option with patients. Participants described the limitations as undermining their medical expertise.
“Some of the harm that is done to these ob.gyns. is not only from the laws themselves, but from their own institutions,” Dr. Buchbinder said. “Hospitals have to decide, ‘what does this law mean and how are we going to put it to practice here?’ ”
Angela Hawkins, MD, a hospitalist practicing in Oklahoma, encountered a patient who was experiencing an obvious miscarriage. But because the situation could not yet be established as life-threatening, Dr. Hawkins felt that she could not intervene.
“There are things I know are straightforward and I would’ve handled them completely differently in the past,” Dr. Hawkins said, adding that she needed to seek reassurance from her hospital employer that she would not face legal ramifications if she provided care.
“It’s frustrating to know that this is medicine and I can’t practice it without calling legal and ethics in the middle of the night,” said Dr. Hawkins, chair of the Oklahoma section of the American College of Obstetrics and Gynecology.
Still, more than half of Oklahoma’s 77 counties are considered maternity care deserts, meaning they have little to no obstetric services available for pregnant patients. Dr. Hawkins recently completed her own survey of practicing ob.gyns. in the state. In soon-to-be published research, almost 60% of the 63 respondents reported thinking about leaving or were planning to leave the state to practice in areas that are less restrictive.
“That’s very concerning to the ob.gyns. that are left,” she said. “I feel like, if everyone leaves, who is left to take care of the patients?”
The study in JAMA Network Open also highlighted that 11% of participants had moved their practices to less restrictive states with stronger abortion protections.
In addition to losing existing clinicians, the laws have made it difficult for medical centers to recruit new ones, according to Kavita Shah Arora, MD, MBE, MS, director for Division of General Obstetrics, Gynecology, and Midwifery at the University of North Carolina at Chapel Hill, and a coauthor of the study. North Carolina enacted a new law in July 2023 that reduced the time allowed for an abortion from 20 weeks to 12 weeks under most circumstances.
“Our department faces new challenges in recruitment and retention being in a restrictive state that we haven’t had to deal with before,” Dr. Arora said. “It’s impacting how medical students choose which residency programs to apply to.”
Ob.gyns. may not be the only clinicians who feel the effect of laws restricting abortion, according to Deborah Nucatola, MD, chief medical officer of Planned Parenthood Great Northwest, Hawai’i, Alaska, Indiana, Kentucky.
Patients who live in areas with limited access to obstetrics services often present to urgent care facilities or emergency departments for medical care that are staffed with family, internal medicine, and emergency physicians, Dr. Nucatola said.
“I don’t want anyone by any means to think this is isolated to one specialty,” said Dr. Nucatola, who was not involved with the study. “It’s going to affect everyone who cares for these patients; you lose the ability to use your medical knowledge and then have to navigate this legal restriction that doesn’t correlate with anything that happens in medicine.”
Dr. Dempsey-Fanning’s 14-year-old patient did eventually receive abortion care outside of South Carolina. Dr. Dempsey-Fanning said that she and her colleagues have spent hours coordinating for patients to receive care in a different state. Then, patients and their families must come up with the money for travel and any missed work to get to another clinician working where abortion is legal.
Despite this, she said, “You are left still feeling as though you abandoned this patient in many practical ways.
“I know I weigh the decision about my future practice almost daily, wondering how long I can stay and keep fighting for patients in an environment ripe with fear, worry, and an overriding sense of injustice,” said Dr. Dempsey-Fanning.
The study authors and experts quoted in the story report no relevant disclosures.
A version of this article appeared on Medscape.com.
Medical Aid in Dying Should Be Legal, Says Ethicist
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
Hi. I’m Art Caplan. I’m at the Division of Medical Ethics at the NYU Grossman School of Medicine.
Right now, there are 10 states and the District of Columbia that have had some version of medical assistance in dying approved and on the books. That basically means that about 20% of Americans have access where they live to a physician who can prescribe a lethal dose of medication to them if they’re terminally ill and can ingest the medication themselves. That leaves many Americans not covered by this kind of access to this kind of service.
Many of you watching this may live in states where it is legal, like Oregon, Washington, New Jersey, Colorado, and Hawaii. I know many doctors say, “I’m not going to do that.” It’s not something that anyone is compelling a doctor to do. For some Americans, access is not just about where they live but whether there is a doctor willing to participate with them in bringing about their accelerated death, knowing that they’re inevitably going to die.
There’s not much we can do about that. It’s up to the conscience of each physician as to what they’re comfortable with. Certainly, there are other things that can be done to extend the possibility of having this available.
One thing that’s taking place is that, after lawsuits were filed, Vermont and Oregon have given up on their residency requirement, so you don’t have to be there 6 months or a year in order to use this opportunity. It’s legal now to move to the state or visit the state, and as soon as you get there, sign up for this kind of end-of-life intervention.
New Jersey is also being sued. I’ll predict that every state that has a residency requirement, when sued in court, is going to lose because we’ve long recognized the right of Americans to seek out healthcare in the United States, wherever they want to go.
If some states have made this a legitimate medical procedure, courts are going to say you can’t restrict it only to state residents. If someone wants to use a service, they’re entitled to show up from another state or another place and use it. I’m not sure about foreign nationals, but I’m very sure that Americans can go state to state in search of legitimate medical procedures.
The other bills that are out there, however, are basically saying they want to emulate Oregon, Washington, and the other states and say that the terminally ill, with severe restrictions, are going to be able to get this service without going anywhere.
The restrictions include a diagnosis of terminal illness and that you have to be deemed mentally competent. You can’t use this if you have Alzheimer’s or severe depression. You have to make a request twice with a week or two in between to make sure that your request is authentic. And obviously, everyone is on board to make sure that you’re not being coerced or pushed somehow into requesting a somewhat earlier death than you would have experienced without having the availability of the pills.
You also have to take the pills yourself or be able to pull a switch so that you could use a feeding tube–type administration. If you can’t do that, say due to ALS, you’re not eligible to use medical aid in dying. It’s a pretty restricted intervention.
Many people who get pills after going through these restrictions in the states that permit it don’t use it. As many as one third say they like having it there as a safety valve or a parachute, but once they know they could end their life sooner, then they’re going to stick it out.
Should states make this legal? New York, Massachusetts, Florida, and many other states have bills that are moving through. I’m going to say yes. We’ve had Oregon and Washington since the late 1990s with medical aid in dying on the books. There doesn’t seem to be any evidence of pushing people to use this, of bias against the disabled, or bigotry against particular ethnic or racial groups being used to encourage people to end their life sooner.
I think it is an option that Americans want. I think it’s an option that makes some sense. I’m well aware that we also have to make sure that people know about hospice. In some of these states, medical aid in dying is offered as a part of hospice — not all, but a few. Not everybody wants hospice once they realize that they’re dying and that it is coming relatively soon. They may want to leave with family present, with a ceremony, or with a quality of life that they desire.
Past experience says let’s continue to expand availability in each state. Let’s also realize that we have to keep the restrictions in place on how it’s used because they have protected us against abuse. Let’s understand that every doctor has an option to do this or not do this. It’s a matter of conscience and a matter of comfort.
I think legalization is the direction we’re going to be going in. Getting rid of the residency requirements that have been around, as I think courts are going to overturn them, also gives a push to the idea that once the service is in this many states, it’s something that should be available if there are doctors willing to do it.
I’m Art Caplan at the Division of Medical Ethics at NYU Grossman School of Medicine. New York, NY. Thank you for watching.
Arthur L. Caplan, PhD, has disclosed the following relevant financial relationships:
- Served as a director, officer, partner, employee, advisor, consultant, or trustee for: Johnson & Johnson’s Panel for Compassionate Drug Use (unpaid position)
- Serves as a contributing author and adviser for: Medscape
A version of this article appeared on Medscape.com.
The Emerging Physician-Scientist Crisis in America
Recent reporting has shown that That’s a problem, because physician-scientists are uniquely equipped to make scientific discoveries in the laboratory and translate them to the clinic. Indeed, many of the discoveries that have transformed medicine for the better were made by physician-scientists. For example, Jonas Salk developed the polio vaccine, Timothy Ley sequenced the first cancer genome, and Anthony Fauci coordinated public health responses to both the HIV/AIDS and COVID-19 pandemics. Indicative of their sheer impact, at least a third and as many as half of all Nobel Prizes and Lasker Awards in physiology/medicine have gone to physician-scientists.
So why is the supply of physician-scientists shrinking so precipitously at a time when medical discoveries are being made at a record-high rate? Immunotherapy and proton therapy are transforming cancer care; RNA technology led to COVID vaccines; CRISPR is facilitating gene editing and treatment of diseases like sickle cell anemia. Yet, as exciting as medical science has become, only 1.5% of American doctors work as physician-scientists, more than a threefold drop compared with 30 years ago when the figure was a more robust 4.7%. What’s going on?
Residency training programs at prestigious academic medical centers have standard infolded research years; for example, neurosurgery residents at academic medical centers will often get 2 years of protected research time. And the National Institutes of Health has training grants dedicated to physician-scientists, such as the K08 award program. Several foundations are also dedicated to supporting early-career physician-scientists. Yet, the number of physicians deciding to become physician-scientists remains low, and, more troubling, the attrition rate of those who do decide to go this route is quite high.
The underlying issue is multifold. First, funding rates from the federal government for grants have become competitive to the point of being unrealistic. For example, the current funding rate for the flagship R01 program from the National Cancer Institute is only 12%. Promotions are typically tied to these grant awards, which means physician-scientists who are unable to acquire substantial grant funding are unable to pay for their research or win promotion — and often exit the physician-scientist track altogether.
Compounding this issue is a lack of mentorship for early-career physician-scientists. With the rise of “careerism” in medicine, senior-level physician-scientists may have less incentive to mentor those who are earlier in their careers. Rather, there seems to be greater reward to “managing up” — that is, spending time to please hospital administrators and departmental leadership. Being involved in countless committees appears to carry more value in advancing an established investigator’s career than does mentorship.
Finally, physician-scientists typically earn less than their clinician colleagues, despite juggling both scientific and clinical responsibilities. While many are comfortable with this arrangement when embarking on this track, the disparity may become untenable after a while, especially as departmental leadership will often turn to physician-scientists to fill clinical coverage gaps when faculty leave the department, or as the medical center expands to satellite centers outside the primary hospital. Indeed, physician-scientists get pulled in several directions, which can lead to burnout and attrition, with many who are highly equipped for this track ultimately hanging up their cleats and seeking more clinical or private industry–oriented opportunities.
Every academic medical center operates differently. Some clearly have done a better job than others promoting and fostering physician-scientists. What we find in the centers that manage to retain physician-scientists is leadership plays a major role: If a medical center values the importance of physician-scientists, they will do things to foster the success of those people, such as assembling mentorship committees, establishing clear criteria for promotion and career advancement, protecting research time while maintaining some level of pay equity, advocating for team science approaches, and supporting investigators in cases of gaps in federal funding. Different countries also have different models for physician-scientist training, with Germany, for example, allowing medical residents to have 3 years of protected time to engage in research after their second year of residency.
The stakes here are high. If we can’t address the physician-scientist recruitment and retention crisis in America now, we risk falling behind other countries in our ability to innovate and deliver world-class care.
Dr Chaudhuri is a tenure-track physician-scientist at Washington University in St. Louis, a Paul and Daisy Soros Fellow, and a Public Voices Fellow of The OpEd Project.
Aadel Chaudhuri, MD, PhD, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Recent reporting has shown that That’s a problem, because physician-scientists are uniquely equipped to make scientific discoveries in the laboratory and translate them to the clinic. Indeed, many of the discoveries that have transformed medicine for the better were made by physician-scientists. For example, Jonas Salk developed the polio vaccine, Timothy Ley sequenced the first cancer genome, and Anthony Fauci coordinated public health responses to both the HIV/AIDS and COVID-19 pandemics. Indicative of their sheer impact, at least a third and as many as half of all Nobel Prizes and Lasker Awards in physiology/medicine have gone to physician-scientists.
So why is the supply of physician-scientists shrinking so precipitously at a time when medical discoveries are being made at a record-high rate? Immunotherapy and proton therapy are transforming cancer care; RNA technology led to COVID vaccines; CRISPR is facilitating gene editing and treatment of diseases like sickle cell anemia. Yet, as exciting as medical science has become, only 1.5% of American doctors work as physician-scientists, more than a threefold drop compared with 30 years ago when the figure was a more robust 4.7%. What’s going on?
Residency training programs at prestigious academic medical centers have standard infolded research years; for example, neurosurgery residents at academic medical centers will often get 2 years of protected research time. And the National Institutes of Health has training grants dedicated to physician-scientists, such as the K08 award program. Several foundations are also dedicated to supporting early-career physician-scientists. Yet, the number of physicians deciding to become physician-scientists remains low, and, more troubling, the attrition rate of those who do decide to go this route is quite high.
The underlying issue is multifold. First, funding rates from the federal government for grants have become competitive to the point of being unrealistic. For example, the current funding rate for the flagship R01 program from the National Cancer Institute is only 12%. Promotions are typically tied to these grant awards, which means physician-scientists who are unable to acquire substantial grant funding are unable to pay for their research or win promotion — and often exit the physician-scientist track altogether.
Compounding this issue is a lack of mentorship for early-career physician-scientists. With the rise of “careerism” in medicine, senior-level physician-scientists may have less incentive to mentor those who are earlier in their careers. Rather, there seems to be greater reward to “managing up” — that is, spending time to please hospital administrators and departmental leadership. Being involved in countless committees appears to carry more value in advancing an established investigator’s career than does mentorship.
Finally, physician-scientists typically earn less than their clinician colleagues, despite juggling both scientific and clinical responsibilities. While many are comfortable with this arrangement when embarking on this track, the disparity may become untenable after a while, especially as departmental leadership will often turn to physician-scientists to fill clinical coverage gaps when faculty leave the department, or as the medical center expands to satellite centers outside the primary hospital. Indeed, physician-scientists get pulled in several directions, which can lead to burnout and attrition, with many who are highly equipped for this track ultimately hanging up their cleats and seeking more clinical or private industry–oriented opportunities.
Every academic medical center operates differently. Some clearly have done a better job than others promoting and fostering physician-scientists. What we find in the centers that manage to retain physician-scientists is leadership plays a major role: If a medical center values the importance of physician-scientists, they will do things to foster the success of those people, such as assembling mentorship committees, establishing clear criteria for promotion and career advancement, protecting research time while maintaining some level of pay equity, advocating for team science approaches, and supporting investigators in cases of gaps in federal funding. Different countries also have different models for physician-scientist training, with Germany, for example, allowing medical residents to have 3 years of protected time to engage in research after their second year of residency.
The stakes here are high. If we can’t address the physician-scientist recruitment and retention crisis in America now, we risk falling behind other countries in our ability to innovate and deliver world-class care.
Dr Chaudhuri is a tenure-track physician-scientist at Washington University in St. Louis, a Paul and Daisy Soros Fellow, and a Public Voices Fellow of The OpEd Project.
Aadel Chaudhuri, MD, PhD, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Recent reporting has shown that That’s a problem, because physician-scientists are uniquely equipped to make scientific discoveries in the laboratory and translate them to the clinic. Indeed, many of the discoveries that have transformed medicine for the better were made by physician-scientists. For example, Jonas Salk developed the polio vaccine, Timothy Ley sequenced the first cancer genome, and Anthony Fauci coordinated public health responses to both the HIV/AIDS and COVID-19 pandemics. Indicative of their sheer impact, at least a third and as many as half of all Nobel Prizes and Lasker Awards in physiology/medicine have gone to physician-scientists.
So why is the supply of physician-scientists shrinking so precipitously at a time when medical discoveries are being made at a record-high rate? Immunotherapy and proton therapy are transforming cancer care; RNA technology led to COVID vaccines; CRISPR is facilitating gene editing and treatment of diseases like sickle cell anemia. Yet, as exciting as medical science has become, only 1.5% of American doctors work as physician-scientists, more than a threefold drop compared with 30 years ago when the figure was a more robust 4.7%. What’s going on?
Residency training programs at prestigious academic medical centers have standard infolded research years; for example, neurosurgery residents at academic medical centers will often get 2 years of protected research time. And the National Institutes of Health has training grants dedicated to physician-scientists, such as the K08 award program. Several foundations are also dedicated to supporting early-career physician-scientists. Yet, the number of physicians deciding to become physician-scientists remains low, and, more troubling, the attrition rate of those who do decide to go this route is quite high.
The underlying issue is multifold. First, funding rates from the federal government for grants have become competitive to the point of being unrealistic. For example, the current funding rate for the flagship R01 program from the National Cancer Institute is only 12%. Promotions are typically tied to these grant awards, which means physician-scientists who are unable to acquire substantial grant funding are unable to pay for their research or win promotion — and often exit the physician-scientist track altogether.
Compounding this issue is a lack of mentorship for early-career physician-scientists. With the rise of “careerism” in medicine, senior-level physician-scientists may have less incentive to mentor those who are earlier in their careers. Rather, there seems to be greater reward to “managing up” — that is, spending time to please hospital administrators and departmental leadership. Being involved in countless committees appears to carry more value in advancing an established investigator’s career than does mentorship.
Finally, physician-scientists typically earn less than their clinician colleagues, despite juggling both scientific and clinical responsibilities. While many are comfortable with this arrangement when embarking on this track, the disparity may become untenable after a while, especially as departmental leadership will often turn to physician-scientists to fill clinical coverage gaps when faculty leave the department, or as the medical center expands to satellite centers outside the primary hospital. Indeed, physician-scientists get pulled in several directions, which can lead to burnout and attrition, with many who are highly equipped for this track ultimately hanging up their cleats and seeking more clinical or private industry–oriented opportunities.
Every academic medical center operates differently. Some clearly have done a better job than others promoting and fostering physician-scientists. What we find in the centers that manage to retain physician-scientists is leadership plays a major role: If a medical center values the importance of physician-scientists, they will do things to foster the success of those people, such as assembling mentorship committees, establishing clear criteria for promotion and career advancement, protecting research time while maintaining some level of pay equity, advocating for team science approaches, and supporting investigators in cases of gaps in federal funding. Different countries also have different models for physician-scientist training, with Germany, for example, allowing medical residents to have 3 years of protected time to engage in research after their second year of residency.
The stakes here are high. If we can’t address the physician-scientist recruitment and retention crisis in America now, we risk falling behind other countries in our ability to innovate and deliver world-class care.
Dr Chaudhuri is a tenure-track physician-scientist at Washington University in St. Louis, a Paul and Daisy Soros Fellow, and a Public Voices Fellow of The OpEd Project.
Aadel Chaudhuri, MD, PhD, has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Europe Is Facing a Pancreatic Cancer Emergency
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
“It’s a health emergency for society, with mortality rates at over 90%,” warned Professor Alfredo Carrato, MD, PhD, the chairperson of Pancreatic Cancer Europe.
There are many challenges with pancreatic cancer: Lack of awareness, difficult to diagnose, no screening programs for the general population, poor survival rate, and limited treatment options.
Life expectancy at the time of diagnosis is just 4.6 months. Only 3% of people diagnosed with pancreatic cancer will survive for 5 years.
A 2024 systematic analysis in The Lancet suggested that people living in Western Europe are more likely to develop pancreatic cancer than those living anywhere else in the world.
Dr. Carrato, emeritus professor of medical oncology at the University of Alcalá, Madrid, Spain, wasn’t surprised. He told this news organization: “I think the lifestyle in Europe plays a part. We have all of the risk factors in society like obesity, our sedentary behavior, too much red meat consumption, and excess alcohol intake.”
Other risk factors include smoking, diabetes, chronic pancreatitis, and a family history.
The incidence of pancreatic cancer increases with age, so the longer life expectancy in Western Europe could also contribute to the region’s high rates.
A Silent Killer
Pancreatic cancer is difficult to identify. It is often asymptomatic. Symptoms that do show themselves, like back pain, weight loss, and nausea, are nonspecific and make early diagnosis challenging.
Professor Mattias Löhr from the Karolinska Institutet, Stockholm, Sweden, told this news organization: “It’s a dismal disease. It’s not accessible for any easy screening or surveillance. Even early diagnosis is too late with pancreatic cancer.”
There have been few advancements in patient outcomes over the past few decades.
Only about 20% of patients are suitable candidates for surgery that could prolong their lives.
Also, cancer reoccurs in two thirds of surgical candidates, said Dr. Carrato, and oncologists don’t know how to identify them in advance. “I have patients who survive 3 months and others who survive 4 or 5 years, so there’s a need to identify these subtypes at a molecular level for personalized, clinical, and translational research and therapies.”
Dr. Löhr agreed: “All of the medical therapies are not really working well enough for pancreatic cancer in sharp contrast to other cancers.”
How Can Rates Be Reduced?
“Pancreatic Cancer Europe is working in every EU state to try to raise awareness,” said Dr. Carrato. “We should have primary prevention programs to modify lifestyle risks. We also need funds for translational and clinical research. Secondary prevention isn’t possible yet as we haven’t identified the higher-risk population who would be the target for screening.”
Screening programs are available for the 10% of people who have a family history of pancreatic cancer. But, for the vast majority, there are no tests or screenings that allow for earlier detection.
“We need blood or stool tests that have high specificity and sensitivity that are cost-effective,” said Dr. Carrato.
“It’s a type of cancer with a particular and very aggressive biology. There is a lack of pancreatic tumor tissue for research, as many patients are diagnosed by fine-needle aspiration cytology. It’s a challenge for researchers. We have no biomarkers available to direct our decisions; no precision oncology,” he added.
Still, there are some encouraging developments.
The European PANCAID project (pancreatic cancer initial detection via liquid biopsy) is trying to find biomarkers to screen at-risk groups for earlier diagnosis via a blood test.
Also, the European Union (EU)-funded PANCAIM project (pancreatic cancer artificial intelligence [AI] for genomics and personalized medicine) has developed an AI algorithm that detects small cancers in CT scans that even experienced radiologists might easily overlook.
The project’s head, Henkjan Huisman, is professor of medical imaging AI at Radboud University Medical Center, Nijmegen, the Netherlands. He told this news organization: “It’s an extremely important step, as 20% of people with pancreas cancer have the ability to undergo surgery, which means they might live substantially longer. We believe if the tumors are found earlier, thanks to the algorithm, they are smaller and more contained, and so substantially more than 20% of patients would be suitable for surgery, which would be a breakthrough.”
Dr. Löhr added that a messenger RNA vaccine is being developed in the United States to prevent pancreatic cancer from returning after surgery and is demonstrating encouraging results in its early trials.
The Road Toward Better Care
To improve cancer care in Europe, Dr. Carrato said: “Reference centers should be a requirement in health policy programs because the outcomes are much better than in centers which only perform fewer surgeries, and Pancreatic Cancer Europe is working with the EU in this direction.”
Finland is a country that appears to have succeeded in this regard. Its 2005 Health Care Act, for example, ensures that cancer patients are able to receive care in one of its five specialized hospitals.
More research funding is also needed. According to Pancreatic Cancer Europe, only 2% of EU funding on cancer is spent on pancreatic cancer.
The American Cancer Society’s Cancer Facts & Figures 2024 makes room for some optimism, with the 5-year survival rate in the United States jumping to 13% from 6% in the society’s 2014 report, as a result of earlier diagnoses and more personalized treatment. But, even with potentially longer survival rates, said Dr. Löhr, “we are still on the trajectory of pancreatic cancer being number two for cancer deaths by 2030.”
“We need more money on research, centralized surgery, and networking between European countries,” said Dr. Carrato. “This networking would need more money for prevention, better diagnosis, and treatment. The problem is pancreatic cancer incidence is increasing and mortality is also in parallel, and we are not making real progress in this scenario.”
A version of this article appeared on Medscape.com.
Think Outside the Traditional Toolbox to Treat Itch
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
ORLANDO — “Itch may not be as sexy as Mohs surgery or aesthetic procedures,” but treating it is important and meaningful to patients, particularly those who’ve found little relief previously, Shawn G. Kwatra, MD, said at the annual ODAC Dermatology, Aesthetic & Surgery Conference.
In doctors’ defense, it can be highly challenging to know which approach is optimal for each individual with pruritus, added Dr. Kwatra, associate professor of dermatology at Johns Hopkins University, Baltimore, Maryland.
Cooling agents, topical capsaicin, topical anesthetics like pramoxine 1%, various forms of lidocaine, strontium, opioid modulators like naltrexone, oral Janus kinase inhibitor (JAK) inhibitors, and medical marijuana are among some of the “outside the box” tools in Dr. Kwatra’s itch toolbox.
Often a Medical Puzzle
Frequently, patients come to the dermatologist complaining of itch, “but you don’t see much on their skin.” After a trial of antihistamines, and some topical steroids, the doctor might put up their hands and think: I tried, but I don’t know what else to do. “This actually happens a lot,” said Dr. Kwatra, who is also director of the Johns Hopkins Itch Center.
This means itch can frustrate providers as well. But for patients, the impact on their quality of life can be on the same level as recovering from a stroke or living with heart failure, Dr. Kwatra said. Finding relief for their itch is where “we can make a big difference for patients.”
Consider Cooling Agents
Many of these therapies are inexpensive and widely available. Cooling agents like menthol, camphor, or calamine can reduce activity of the transient receptor potential (TRP) channels in the skin associated with itch. This ion channel also senses temperature, pressure, and other sensations.
Another option is topical capsaicin, which works through the same ion channels. It binds to the TRPV1 receptors in sensory nerve fibers and causes desensitization. Initially, four to six applications a day are required to reduce itch. After that, patients can apply the medication less frequently. “You have to tell folks we know it’s going to work, but it’s going to burn a lot initially,” Dr. Kwatra said. “In real world practice, I’m not using it often.”
A 1.8% capsaicin patch, approved for treating postherpetic neuralgia, can be used to treat pruritus as well. “You put the patch on for one hour and you can have a true clinical response,” he noted.
Another option for itch relief, the topical anesthetic pramoxine 1%, “is probably underutilized for our patients,” Dr. Kwatra said. Pramoxine 1% works fast — as quickly as 2 minutes — and lasts up to 8 hours and is well-tolerated with low toxicity, he added. The agent is applied three to four times a day and relieves itch by reducing the transmembrane permeability of sodium ions on the skin. “This is something widely available and cheap.”
Lidocaine, another topical anesthetic, is available compounded, over the counter, and as a spray or patch. “I would be careful before you use high doses, like 10%” because of tolerability issues, Dr. Kwatra cautioned. He generally starts with lower concentrations.
Topical strontium is really interesting as a strategy, Dr. Kwatra said. Strontium is a soft, white metal that competes with calcium for receptor binding. There are over-the-counter formulations available as a scalp solution or lotion, which, he said, “are ways to go with more episodic itching.”
Topical oatmeal can also relieve itch in some patients. “There is actually some good scientific evidence for topical oatmeal preparations,” he said.
Steroid-Sparing Novel Topicals
Topical ruxolitinib (a JAK inhibitor approved for atopic dermatitis and vitiligo); topical roflumilast (a phosphodiesterase-4 inhibitor) and topical tapinarof (an aryl hydrocarbon receptor agonist), both approved for treating psoriasis; and the atopic dermatitis drug crisaborole fall into this category of topicals with potential for treating itch, he said, noting that use for treating itch is off label.
Off-label use of biologic agents are also possible treatment options for itch, dupilumab and tralokinumab, both US Food and Drug Administration (FDA)–approved for treating atopic dermatitis. Emerging agents that may prove useful for treating itch include lebrikizumab, nemolizumab, amlitelimab, and rocatinlimab, he said.
In terms of oral therapies, the FDA has approved two oral JAK inhibitors for atopic dermatitis, abrocitinib and upadacitinib, which could prove useful for itch as an off-label indication, according to Dr. Kwatra.
Naltrexone Off Label
An emerging therapeutic concept for treating itch is using an opioid antagonist like naltrexone. Morphine causes more itch, so the theory is a reversal agent might help reduce it. The challenge is that naltrexone only comes as a 50 mg tablet, “and I find the high dose makes people nauseous and vomit,” he added.
Don’t Forget Devices
He referred to a “great paper” that he said has been “totally overlooked,” published in 2001, which evaluated a device that stimulates C fibers in the skin to reduce itch. In the study, 19 patients used the device to treat local areas 20 minutes daily for 5 weeks. Punch biopsies of the affected areas were taken at baseline and after treatment. Mean itch ratings decreased from 78% to 42%, and the number of immunoreactive nerve fibers in the epidermis decreased by 40% at the end of treatment.
“Electrical neurostimulation is better for localized pruritus. There is limited case series evidence, but it’s something to think about,” Dr. Kwatra said.
He and his colleagues also have a case study in press that explored the use of injected botulinum toxin to relieve recalcitrant, chronic itch in a 65-year-old man “who failed everything.”
Dr. Kwatra is a consultant or advisory board member for AbbVie, Amgen, Arcutis Biotherapeutics, ASLAN Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Celldex Therapeutics, Galderma, Incyte Corporation, Johnson & Johnson, LEO Pharma, Novartis, Pfizer, Regeneron, and Sanofi.
A version of this article appeared on Medscape.com.
FROM ODAC 2024
Cancer Identified as a New Cardiovascular Risk Factor
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
A history of cancer is an independent predictor of major cardiovascular events in patients undergoing coronary angioplasty. Cancer should be considered a new cardiovascular risk factor in primary and secondary prevention, according to a study presented at the 2023 American Heart Association Congress in Philadelphia.
, for example, equating it to the situation of a patient with diabetes or chronic renal failure,” said lead author Renzo Melchiori, MD, a cardiologist at the University Hospital Austral in Pilar, Argentina.
The researchers also advocate for intensifying cardiovascular control measures in secondary prevention for these patients, reconsidering goals, and ensuring compliance with prescribed pharmacological regimens and healthy lifestyle habits.
“Previously, when a patient had oncological pathology, thinking about associated cardiovascular risk seemed somewhat superfluous. But today, oncological diseases are treated so effectively, increasing survival and life expectancy, that we begin to focus on what happens with the arteries of these patients after treatment,” said Dr. Melchiori.
Higher Incidence Density
The retrospective analysis included 937 patients of both sexes aged 18 years and older who underwent coronary angioplasty for acute coronary syndrome between 2008 and 2022 at a university hospital. Of these participants, 89 (9.5%) had a history of cancer, with a median time since oncologic diagnosis of around 2 years for solid and hematologic tumors. Most participants had treated and resolved cancer.
Over a median follow-up of 45 months (range, 14-72 months), the cumulative incidence rates of a major cardiovascular event (nonfatal stroke, nonfatal acute myocardial infarction, cardiovascular death, or new angioplasty) were 22.2% (155/698) and 28.4% (25/88) in the groups without and with a history of cancer, respectively. The incidence density was significantly higher in the group with an oncologic history than in the group without such a history: 0.78 events/100 patients/month vs 0.48 events/100 patients/month (P = .01).
Kaplan-Meier analysis showed a higher probability of a major cardiovascular event in the group of patients with cancer or a history of cancer (P = .0086). In multivariate Cox regression analysis, cancer history was an independent predictor of major cardiovascular events adjusted for other risk factors such as age, hypertension, diabetes, smoking, sedentary lifestyle, and family history (hazard ratio, 1.66; P = .025).
Dr. Melchiori clarified that the increased incidence of cardiovascular events in patients with cancer or a history of cancer cannot be attributed to differences in percutaneous intervention or the indication or compliance of post-treatment pharmacological therapy.
In addition, the specialist acknowledged that due to the sample size, discrimination by cancer type, disease stage, or therapeutic strategies couldn’t be performed. A subanalysis, which has not been presented, indicated that the effect could not be explained solely by the application of radiotherapy or chemotherapy in the 90 days before angioplasty — two factors that cause arterial inflammation.
Intensifying Prevention Measures
Two independent experts told this news organization that the new study is "interesting" and reinforces the close connection between oncologic and cardiovascular pathology.
Andrés Daniele, MD, cardiologist and president of the Argentine Cardio-Oncology Association, a local chapter of the International Cardio-Oncology Society, emphasized that the study “reiterates an observation seen in other works: A higher rate of atherosclerotic pathology and cardiovascular events in patients with a history of cancer. And that has a reason to be: Both pathologies present common risk factors, and on the other hand, there is greater endothelial dysfunction secondary to the inflammatory syndrome and oncologic therapies.”
“There needs to be a continuum in the intensification of measures in primary and secondary cardiovascular prevention in cancer survivors, whether in remission or with chronic disease. We need to be very aggressive in managing risk factors and insist that patients who have had a cardiovascular event enter cardiovascular rehabilitation therapies,” said Dr. Daniele, who also heads the Cardio-Oncology Department at the centenary Roffo Institute of Oncology at the University of Buenos Aires, Argentina.
The study provides a valuable contribution because “we need to understand the epidemiology and natural history of patients with cancer at risk of developing cardiovascular complications to implement personalized cardiovascular prevention strategies,” said Teresa López Fernández, MD, cardiologist, coordinator of the Cardio-Oncology Program at La Paz University Hospital in Madrid, member of the Cardio-Oncology Working Group of the Spanish Society of Cardiology, member of the board of the International Cardio-Oncology Society, and cochair of the first clinical practice guidelines in cardio-oncology of the European Society of Cardiology.
“We have to be aware that perhaps we should not guide ourselves in these patients with the usual risk stratification scores as cancer or cardiotoxic treatment are not included as variables. However, they require our attention and effort to improve their quality and quantity of life, avoiding potentially preventable cardiovascular events that could negatively impact the survival achieved thanks to advances in cancer treatments,” said Dr. López Fernández.
Dr. Melchiori and Dr. Daniele declared no relevant economic conflicts of interest. Dr. López Fernández reported relationships with Daiichi Sankyo, Almirall España, Janssen-Cilag, Bayer, Roche, Philips, and Incyte.
This article was translated from the Medscape Spanish edition. A version of this article appeared on Medscape.com.
Even Intentional Weight Loss Linked With Cancer
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.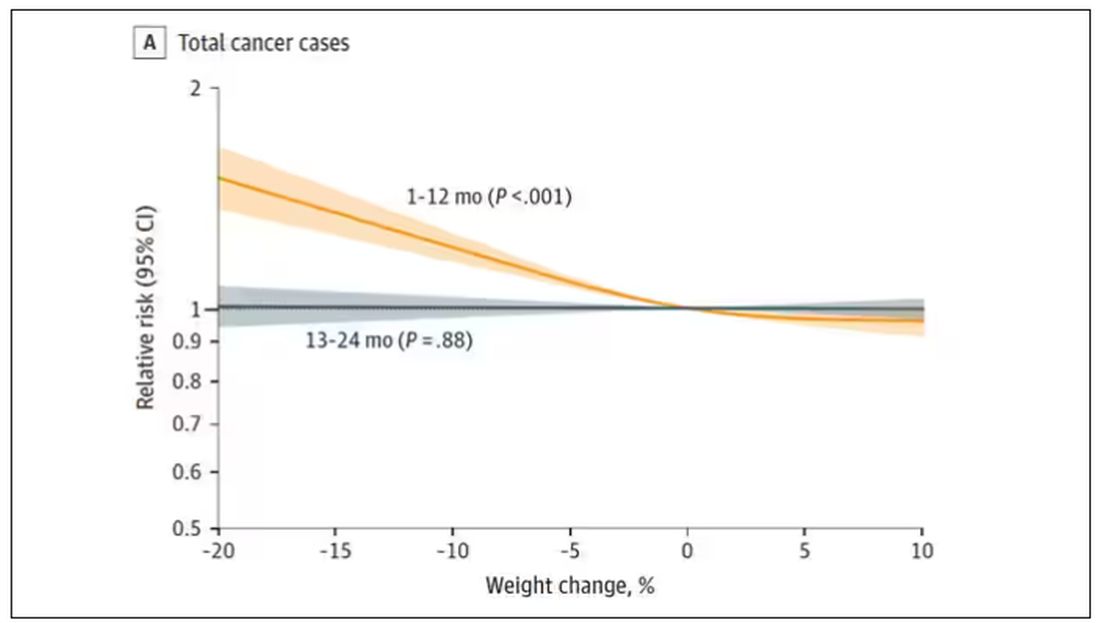
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.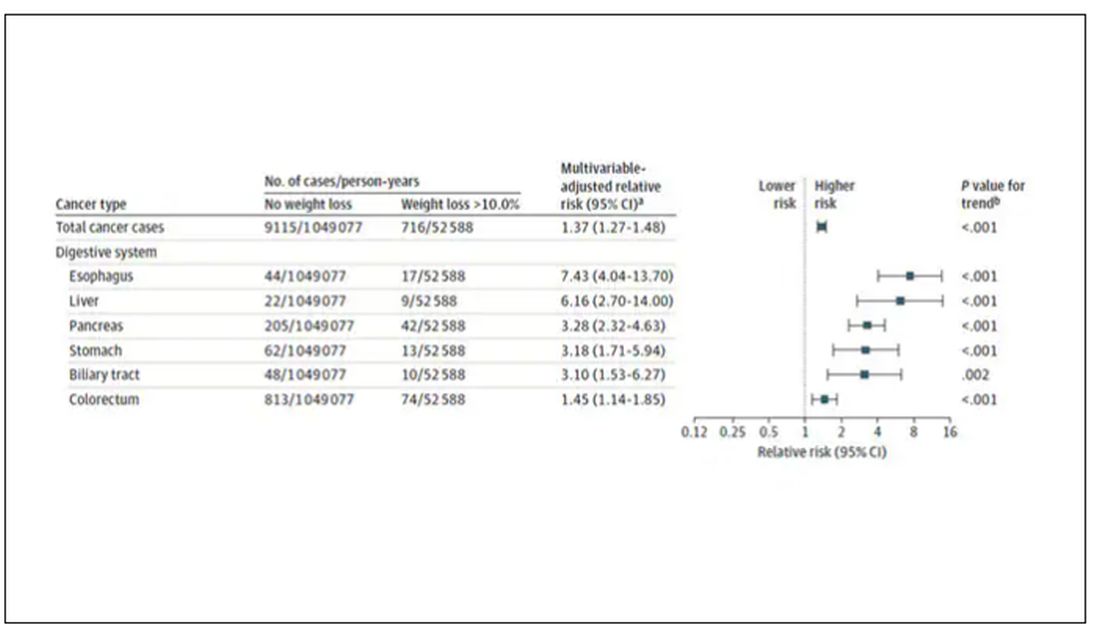
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.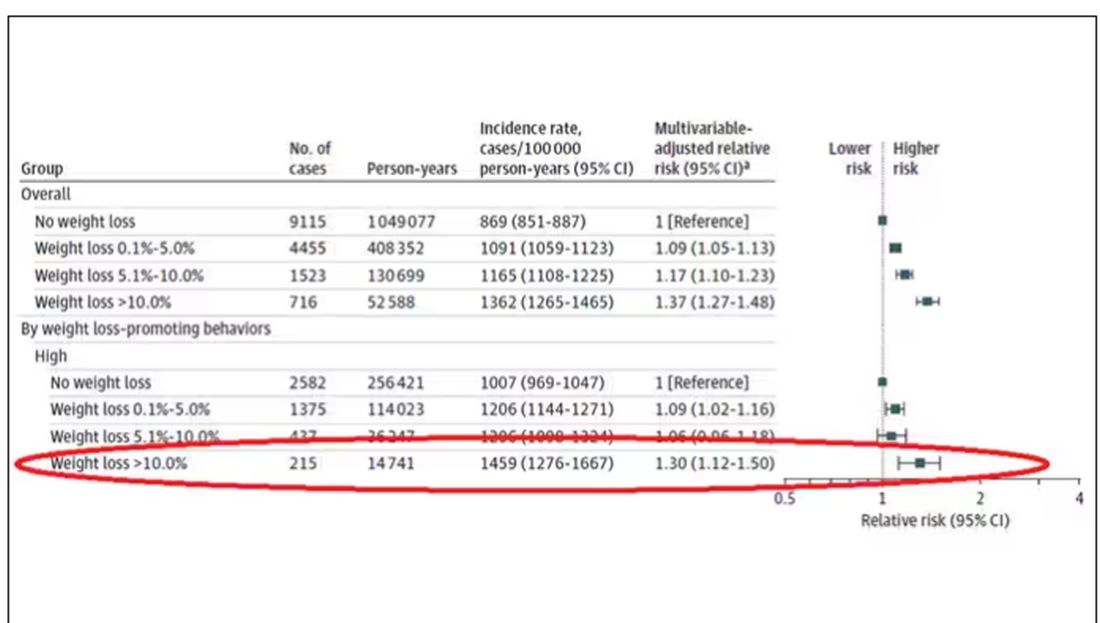
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.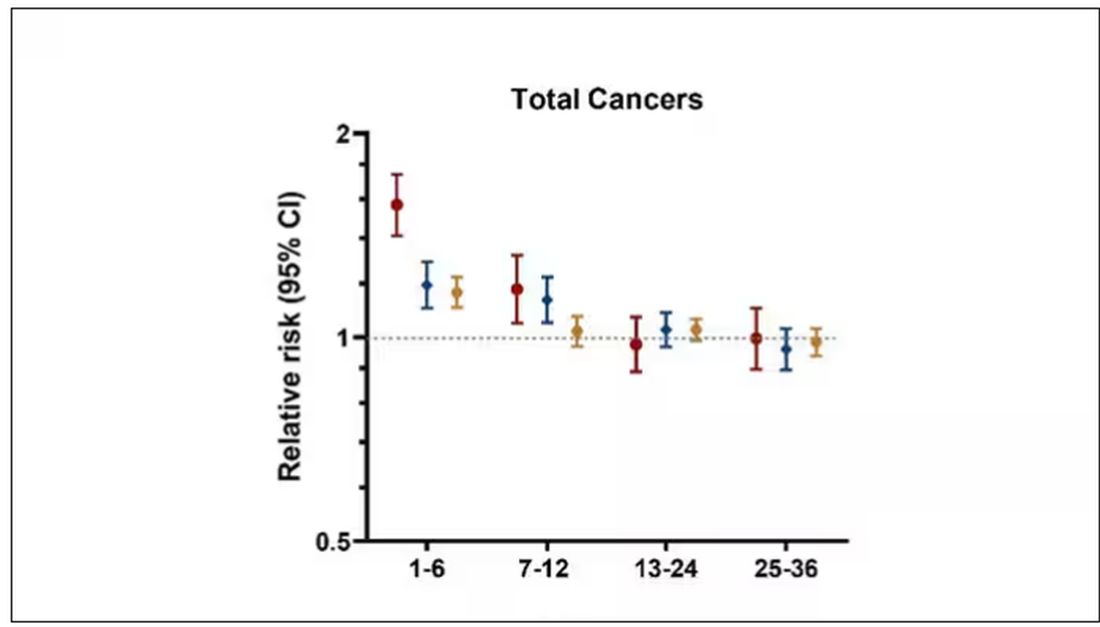
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
As anyone who has been through medical training will tell you, some little scenes just stick with you. I had been seeing a patient in our resident clinic in West Philly for a couple of years. She was in her mid-60s with diabetes and hypertension and a distant smoking history. She was overweight and had been trying to improve her diet and lose weight since I started seeing her. One day she came in and was delighted to report that she had finally started shedding some pounds — about 15 in the past 2 months.
I enthusiastically told my preceptor that my careful dietary counseling had finally done the job. She looked through the chart for a moment and asked, “Is she up to date on her cancer screening?” A workup revealed adenocarcinoma of the lung. The patient did well, actually, but the story stuck with me.
The textbooks call it “unintentional weight loss,” often in big, scary letters, and every doctor will go just a bit pale if a patient tells them that, despite efforts not to, they are losing weight. But true unintentional weight loss is not that common. After all, most of us are at least half-heartedly trying to lose weight all the time. Should doctors be worried when we are successful?
A new study suggests that perhaps they should. We’re talking about this study, appearing in JAMA, which combined participants from two long-running observational cohorts: 120,000 women from the Nurses’ Health Study, and 50,000 men from the Health Professionals Follow-Up Study. (These cohorts started in the 1970s and 1980s, so we’ll give them a pass on the gender-specific study designs.)
The rationale of enrolling healthcare providers in these cohort studies is that they would be reliable witnesses of their own health status. If a nurse or doctor says they have pancreatic cancer, it’s likely that they truly have pancreatic cancer. Detailed health surveys were distributed to the participants every other year, and the average follow-up was more than a decade.
Participants recorded their weight — as an aside, a nested study found that self-reported rate was extremely well correlated with professionally measured weight — and whether they had received a cancer diagnosis since the last survey.
This allowed researchers to look at the phenomenon described above. Would weight loss precede a new diagnosis of cancer? And, more interestingly, would intentional weight loss precede a new diagnosis of cancer.
I don’t think it will surprise you to hear that individuals in the highest category of weight loss, those who lost more than 10% of their body weight over a 2-year period, had a larger risk of being diagnosed with cancer in the next year. That’s the yellow line in this graph. In fact, they had about a 40% higher risk than those who did not lose weight.
Increased risk was found across multiple cancer types, though cancers of the gastrointestinal tract, not surprisingly, were most strongly associated with antecedent weight loss.
What about intentionality of weight loss? Unfortunately, the surveys did not ask participants whether they were trying to lose weight. Rather, the surveys asked about exercise and dietary habits. The researchers leveraged these responses to create three categories of participants: those who seemed to be trying to lose weight (defined as people who had increased their exercise and dietary quality); those who didn’t seem to be trying to lose weight (they changed neither exercise nor dietary behaviors); and a middle group, which changed one or the other of these behaviors but not both.
Let’s look at those who really seemed to be trying to lose weight. Over 2 years, they got more exercise and improved their diet.
If they succeeded in losing 10% or more of their body weight, they still had a higher risk for cancer than those who had not lost weight — about 30% higher, which is not that different from the 40% increased risk when you include those folks who weren’t changing their lifestyle.
This is why this study is important. The classic teaching is that unintentional weight loss is a bad thing and needs a workup. That’s fine. But we live in a world where perhaps the majority of people are, at any given time, trying to lose weight.
We need to be careful here. I am not by any means trying to say that people who have successfully lost weight have cancer. Both of the following statements can be true:
Significant weight loss, whether intentional or not, is associated with a higher risk for cancer.
Most people with significant weight loss will not have cancer.
Both of these can be true because cancer is, fortunately, rare. Of people who lose weight, the vast majority will lose weight because they are engaging in healthier behaviors. A small number may lose weight because something else is wrong. It’s just hard to tell the two apart.
Out of the nearly 200,000 people in this study, only around 16,000 developed cancer during follow-up. Again, although the chance of having cancer is slightly higher if someone has experienced weight loss, the chance is still very low.
We also need to avoid suggesting that weight loss causes cancer. Some people lose weight because of an existing, as of yet undiagnosed cancer and its metabolic effects. This is borne out if you look at the risk of being diagnosed with cancer as you move further away from the interval of weight loss.
The further you get from the year of that 10% weight loss, the less likely you are to be diagnosed with cancer. Most of these cancers are diagnosed within a year of losing weight. In other words, if you’re reading this and getting worried that you lost weight 10 years ago, you’re probably out of the woods. That was, most likely, just you getting healthier.
Last thing: We have methods for weight loss now that are way more effective than diet or exercise. I’m looking at you, Ozempic. But aside from the weight loss wonder drugs, we have surgery and other interventions. This study did not capture any of that data. Ozempic wasn’t even on the market during this study, so we can’t say anything about the relationship between weight loss and cancer among people using nonlifestyle mechanisms to lose weight.
It’s a complicated system. But the clinically actionable point here is to notice if patients have lost weight. If they’ve lost it without trying, further workup is reasonable. If they’ve lost it but were trying to lose it, tell them “good job.” And consider a workup anyway.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.