User login
For MD-IQ use only
Magnetic System May Improve Kidney Stone Removal
Kidney stones afflict approximately one in nine individuals, causing intense pain and serious infections. With over 1.3 million emergency room visits and healthcare expenditures exceeding $5 billion annually in the United States, they pose a significant health burden. Small, hard-to-extract fragments are often left behind, risking natural elimination. While technologies like focused ultrasound, fragment adhesion with biopolymers, and negative pressure aspiration have been explored, they face limitations, especially with standard ureteroscope channel sizes.
Magnetizing Renal Calculus Fragments
A published study introduced the Magnetic System for Total Nephrolith Extraction, a system designed to enhance the efficiency of renal calculus fragment removal. In this system, the stones are coated with a magnetic hydrogel and retrieved using a magnetic guidewire compatible with standard ureteroscopes.
In vitro, laser-obtained renal calculus fragments were separated by size and coated with either ferumoxytol alone or combined with chitosan (Hydrogel CF). Treated fragments were then subjected to a magnetic wire for fragment removal assessment. Additional tests included scanning electron microscopy and cell culture with human urothelial cells to evaluate the cytotoxicity of the magnetic hydrogel components. The hydrogel and its components underwent safety and efficacy evaluations in in vitro studies, human tissue samples, and murine models to assess their impact on urothelium and antibacterial properties.
Safe Fragment Removal
The Hydrogel CF, composed of ferumoxytol and chitosan, demonstrated 100% effectiveness in eliminating all tested fragments, even those measuring up to 4 mm, across various stone compositions. Particle tracing simulations indicated that small-sized stones (1 and 3 mm) could be captured several millimeters away. Scanning electron microscopy confirmed the binding of ferumoxytol and Hydrogel CF to the surface of calcium oxalate stones.
The components of Hydrogel CF did not induce significant cytotoxicity on human urothelial cells, even after a 4-hour exposure. Moreover, live mouse studies showed that Hydrogel CF caused less bladder urothelium exfoliation compared with chitosan, and the urothelium returned to normal within 12 hours. In addition, these components exhibited antibacterial properties, inhibiting the growth of uropathogenic bacteria such as Escherichia coli and Proteus mirabilis, comparable to that of ciprofloxacin.
The ability to eliminate lithiasic fragments, the absence of significant urothelial toxicity, and antibacterial activity suggest that the use of magnetic hydrogel could be integrated into laser treatments for renal stones through ureteroscopy without immediate complications. The antibacterial properties could offer potential postoperative benefits while reducing procedural time. Further animal studies are underway to assess the safety of Hydrogel CF before proceeding to human clinical trials.
This article was translated from JIM, which is part of the Medscape Professional Network. A version of this article appeared on Medscape.com.
Kidney stones afflict approximately one in nine individuals, causing intense pain and serious infections. With over 1.3 million emergency room visits and healthcare expenditures exceeding $5 billion annually in the United States, they pose a significant health burden. Small, hard-to-extract fragments are often left behind, risking natural elimination. While technologies like focused ultrasound, fragment adhesion with biopolymers, and negative pressure aspiration have been explored, they face limitations, especially with standard ureteroscope channel sizes.
Magnetizing Renal Calculus Fragments
A published study introduced the Magnetic System for Total Nephrolith Extraction, a system designed to enhance the efficiency of renal calculus fragment removal. In this system, the stones are coated with a magnetic hydrogel and retrieved using a magnetic guidewire compatible with standard ureteroscopes.
In vitro, laser-obtained renal calculus fragments were separated by size and coated with either ferumoxytol alone or combined with chitosan (Hydrogel CF). Treated fragments were then subjected to a magnetic wire for fragment removal assessment. Additional tests included scanning electron microscopy and cell culture with human urothelial cells to evaluate the cytotoxicity of the magnetic hydrogel components. The hydrogel and its components underwent safety and efficacy evaluations in in vitro studies, human tissue samples, and murine models to assess their impact on urothelium and antibacterial properties.
Safe Fragment Removal
The Hydrogel CF, composed of ferumoxytol and chitosan, demonstrated 100% effectiveness in eliminating all tested fragments, even those measuring up to 4 mm, across various stone compositions. Particle tracing simulations indicated that small-sized stones (1 and 3 mm) could be captured several millimeters away. Scanning electron microscopy confirmed the binding of ferumoxytol and Hydrogel CF to the surface of calcium oxalate stones.
The components of Hydrogel CF did not induce significant cytotoxicity on human urothelial cells, even after a 4-hour exposure. Moreover, live mouse studies showed that Hydrogel CF caused less bladder urothelium exfoliation compared with chitosan, and the urothelium returned to normal within 12 hours. In addition, these components exhibited antibacterial properties, inhibiting the growth of uropathogenic bacteria such as Escherichia coli and Proteus mirabilis, comparable to that of ciprofloxacin.
The ability to eliminate lithiasic fragments, the absence of significant urothelial toxicity, and antibacterial activity suggest that the use of magnetic hydrogel could be integrated into laser treatments for renal stones through ureteroscopy without immediate complications. The antibacterial properties could offer potential postoperative benefits while reducing procedural time. Further animal studies are underway to assess the safety of Hydrogel CF before proceeding to human clinical trials.
This article was translated from JIM, which is part of the Medscape Professional Network. A version of this article appeared on Medscape.com.
Kidney stones afflict approximately one in nine individuals, causing intense pain and serious infections. With over 1.3 million emergency room visits and healthcare expenditures exceeding $5 billion annually in the United States, they pose a significant health burden. Small, hard-to-extract fragments are often left behind, risking natural elimination. While technologies like focused ultrasound, fragment adhesion with biopolymers, and negative pressure aspiration have been explored, they face limitations, especially with standard ureteroscope channel sizes.
Magnetizing Renal Calculus Fragments
A published study introduced the Magnetic System for Total Nephrolith Extraction, a system designed to enhance the efficiency of renal calculus fragment removal. In this system, the stones are coated with a magnetic hydrogel and retrieved using a magnetic guidewire compatible with standard ureteroscopes.
In vitro, laser-obtained renal calculus fragments were separated by size and coated with either ferumoxytol alone or combined with chitosan (Hydrogel CF). Treated fragments were then subjected to a magnetic wire for fragment removal assessment. Additional tests included scanning electron microscopy and cell culture with human urothelial cells to evaluate the cytotoxicity of the magnetic hydrogel components. The hydrogel and its components underwent safety and efficacy evaluations in in vitro studies, human tissue samples, and murine models to assess their impact on urothelium and antibacterial properties.
Safe Fragment Removal
The Hydrogel CF, composed of ferumoxytol and chitosan, demonstrated 100% effectiveness in eliminating all tested fragments, even those measuring up to 4 mm, across various stone compositions. Particle tracing simulations indicated that small-sized stones (1 and 3 mm) could be captured several millimeters away. Scanning electron microscopy confirmed the binding of ferumoxytol and Hydrogel CF to the surface of calcium oxalate stones.
The components of Hydrogel CF did not induce significant cytotoxicity on human urothelial cells, even after a 4-hour exposure. Moreover, live mouse studies showed that Hydrogel CF caused less bladder urothelium exfoliation compared with chitosan, and the urothelium returned to normal within 12 hours. In addition, these components exhibited antibacterial properties, inhibiting the growth of uropathogenic bacteria such as Escherichia coli and Proteus mirabilis, comparable to that of ciprofloxacin.
The ability to eliminate lithiasic fragments, the absence of significant urothelial toxicity, and antibacterial activity suggest that the use of magnetic hydrogel could be integrated into laser treatments for renal stones through ureteroscopy without immediate complications. The antibacterial properties could offer potential postoperative benefits while reducing procedural time. Further animal studies are underway to assess the safety of Hydrogel CF before proceeding to human clinical trials.
This article was translated from JIM, which is part of the Medscape Professional Network. A version of this article appeared on Medscape.com.
Deaths Linked to Substance Use, CVD on the Rise
TOPLINE:
, with the most pronounced rise among women, American Indians, younger people, rural residents, and users of cannabis and psychostimulants, results of new research suggest.
METHODOLOGY:
- From the Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research (CDC WONDER) database and using International Classification of Diseases (ICD) codes, researchers collected data on deaths within the United States where both SU and CVD (SU+CVD) were a contributing or an underlying cause and gathered information on location of death (medical facility, home, hospice, nursing home/long-term care facility), demographics (sex, race/ethnicity, age), and region (urban-rural, state).
- Researchers determined crude and age-adjusted mortality rates (AAMRs) per 100,000 population, identified trends in AAMR using annual percent change (APC) and calculated the weighted average of APCs (AAPCs).
- Between 1999 and 2019, there were 636,572 deaths related to CVD+SU, 75.6% of which were among men and 70.6% among non-Hispanic White individuals, with 65% related to alcohol, and where location of death was available, 47.7% occurred in medical facilities.
TAKEAWAY:
- The overall SU+CVD-related AAMR from 1999 to 2019 was 14.3 (95% CI, 14.3-14.3) per 100,000 individuals, with the rate being higher in men (22.5) than in women (6.8) and highest in American Indians or Alaska Natives (37.7) compared with other races/ethnicities.
- Rural areas had higher SU+CVD-related AAMR (15.2; 95% CI, 15.1-15.3) than urban areas, with the District of Columbia having the highest AAMR geographically (25.4), individuals aged 55-69 years having the highest rate agewise (25.1), and alcohol accounting for the highest rate (9.09) among substance types.
- Temporal trends show that the overall SU+CVD-related AAMR increased from 9.9 in 1999 to 21.4 in 2019, a rate that started accelerating in 2012, with an AAPC of 4.0% (95% CI, 3.7-4.3); increases were across all ethnicities and age groups and were particularly pronounced among women (4.8%; 95% CI, 4.5-5.1).
- Cannabis had the highest AAPC of all substances (12.7%), but stimulants had an APC of 21.4 (95% CI, 20.0-22.8) from 2009 to 2019, a period during which stimulants were the fastest-growing substance abuse category.
IN PRACTICE:
These new results identify high-risk groups, which “is crucial for prioritizing preventive measures aiming to reduce substance use and cardiovascular disease-related mortality in these populations,” the researchers wrote.
SOURCE:
Abdul Mannan Khan Minhas, MD, Department of Medicine, University of Mississippi Medical Center, Jackson, Mississippi, and Jakrin Kewcharoen, MD, Division of Cardiology, Loma Linda University Medical Center, Loma Linda, California, were co-first authors of the study, which was published online in the Journal of the American Heart Association.
A version of this article first appeared on Medscape.com.
TOPLINE:
, with the most pronounced rise among women, American Indians, younger people, rural residents, and users of cannabis and psychostimulants, results of new research suggest.
METHODOLOGY:
- From the Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research (CDC WONDER) database and using International Classification of Diseases (ICD) codes, researchers collected data on deaths within the United States where both SU and CVD (SU+CVD) were a contributing or an underlying cause and gathered information on location of death (medical facility, home, hospice, nursing home/long-term care facility), demographics (sex, race/ethnicity, age), and region (urban-rural, state).
- Researchers determined crude and age-adjusted mortality rates (AAMRs) per 100,000 population, identified trends in AAMR using annual percent change (APC) and calculated the weighted average of APCs (AAPCs).
- Between 1999 and 2019, there were 636,572 deaths related to CVD+SU, 75.6% of which were among men and 70.6% among non-Hispanic White individuals, with 65% related to alcohol, and where location of death was available, 47.7% occurred in medical facilities.
TAKEAWAY:
- The overall SU+CVD-related AAMR from 1999 to 2019 was 14.3 (95% CI, 14.3-14.3) per 100,000 individuals, with the rate being higher in men (22.5) than in women (6.8) and highest in American Indians or Alaska Natives (37.7) compared with other races/ethnicities.
- Rural areas had higher SU+CVD-related AAMR (15.2; 95% CI, 15.1-15.3) than urban areas, with the District of Columbia having the highest AAMR geographically (25.4), individuals aged 55-69 years having the highest rate agewise (25.1), and alcohol accounting for the highest rate (9.09) among substance types.
- Temporal trends show that the overall SU+CVD-related AAMR increased from 9.9 in 1999 to 21.4 in 2019, a rate that started accelerating in 2012, with an AAPC of 4.0% (95% CI, 3.7-4.3); increases were across all ethnicities and age groups and were particularly pronounced among women (4.8%; 95% CI, 4.5-5.1).
- Cannabis had the highest AAPC of all substances (12.7%), but stimulants had an APC of 21.4 (95% CI, 20.0-22.8) from 2009 to 2019, a period during which stimulants were the fastest-growing substance abuse category.
IN PRACTICE:
These new results identify high-risk groups, which “is crucial for prioritizing preventive measures aiming to reduce substance use and cardiovascular disease-related mortality in these populations,” the researchers wrote.
SOURCE:
Abdul Mannan Khan Minhas, MD, Department of Medicine, University of Mississippi Medical Center, Jackson, Mississippi, and Jakrin Kewcharoen, MD, Division of Cardiology, Loma Linda University Medical Center, Loma Linda, California, were co-first authors of the study, which was published online in the Journal of the American Heart Association.
A version of this article first appeared on Medscape.com.
TOPLINE:
, with the most pronounced rise among women, American Indians, younger people, rural residents, and users of cannabis and psychostimulants, results of new research suggest.
METHODOLOGY:
- From the Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research (CDC WONDER) database and using International Classification of Diseases (ICD) codes, researchers collected data on deaths within the United States where both SU and CVD (SU+CVD) were a contributing or an underlying cause and gathered information on location of death (medical facility, home, hospice, nursing home/long-term care facility), demographics (sex, race/ethnicity, age), and region (urban-rural, state).
- Researchers determined crude and age-adjusted mortality rates (AAMRs) per 100,000 population, identified trends in AAMR using annual percent change (APC) and calculated the weighted average of APCs (AAPCs).
- Between 1999 and 2019, there were 636,572 deaths related to CVD+SU, 75.6% of which were among men and 70.6% among non-Hispanic White individuals, with 65% related to alcohol, and where location of death was available, 47.7% occurred in medical facilities.
TAKEAWAY:
- The overall SU+CVD-related AAMR from 1999 to 2019 was 14.3 (95% CI, 14.3-14.3) per 100,000 individuals, with the rate being higher in men (22.5) than in women (6.8) and highest in American Indians or Alaska Natives (37.7) compared with other races/ethnicities.
- Rural areas had higher SU+CVD-related AAMR (15.2; 95% CI, 15.1-15.3) than urban areas, with the District of Columbia having the highest AAMR geographically (25.4), individuals aged 55-69 years having the highest rate agewise (25.1), and alcohol accounting for the highest rate (9.09) among substance types.
- Temporal trends show that the overall SU+CVD-related AAMR increased from 9.9 in 1999 to 21.4 in 2019, a rate that started accelerating in 2012, with an AAPC of 4.0% (95% CI, 3.7-4.3); increases were across all ethnicities and age groups and were particularly pronounced among women (4.8%; 95% CI, 4.5-5.1).
- Cannabis had the highest AAPC of all substances (12.7%), but stimulants had an APC of 21.4 (95% CI, 20.0-22.8) from 2009 to 2019, a period during which stimulants were the fastest-growing substance abuse category.
IN PRACTICE:
These new results identify high-risk groups, which “is crucial for prioritizing preventive measures aiming to reduce substance use and cardiovascular disease-related mortality in these populations,” the researchers wrote.
SOURCE:
Abdul Mannan Khan Minhas, MD, Department of Medicine, University of Mississippi Medical Center, Jackson, Mississippi, and Jakrin Kewcharoen, MD, Division of Cardiology, Loma Linda University Medical Center, Loma Linda, California, were co-first authors of the study, which was published online in the Journal of the American Heart Association.
A version of this article first appeared on Medscape.com.
Researchers Uncover Nanoplastics in Water Bottles
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
FROM THE PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Testosterone Replacement May Cause ... Fracture?
This transcript has been edited for clarity.
I am showing you a graph without any labels.
What could this line represent? The stock price of some company that made a big splash but failed to live up to expectations? An outbreak curve charting the introduction of a new infectious agent to a population? The performance of a viral tweet?
I’ll tell you what it is in a moment, but I wanted you to recognize that there is something inherently wistful in this shape, something that speaks of past glory and inevitable declines. It’s a graph that induces a feeling of resistance — no, do not go gently into that good night.
The graph actually represents (roughly) the normal level of serum testosterone in otherwise-healthy men as they age.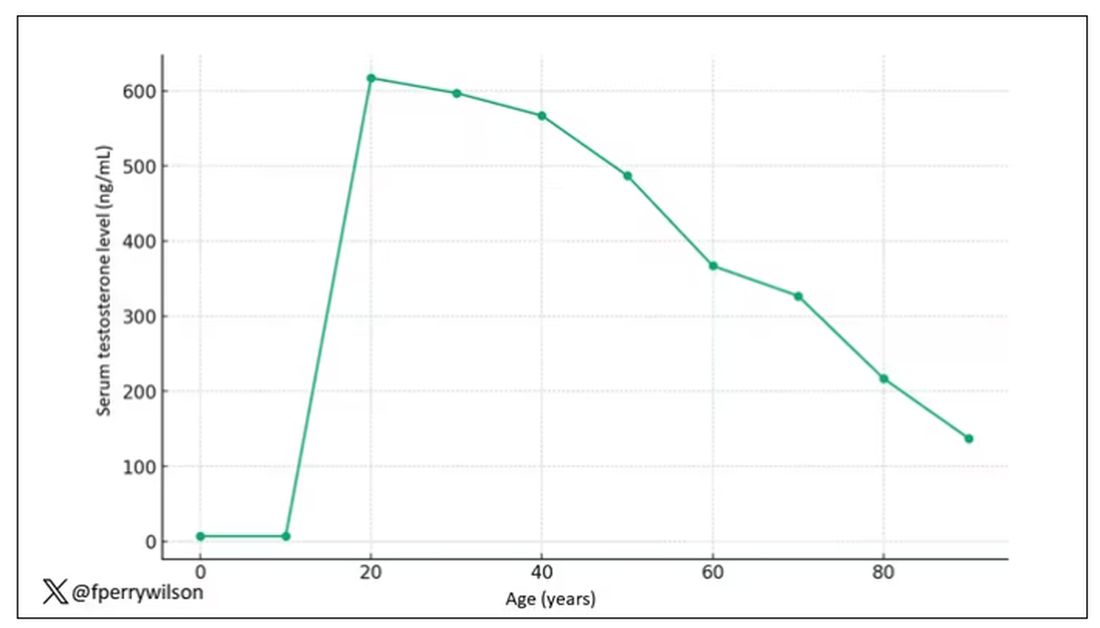
A caveat here: These numbers are not as well defined as I made them seem on this graph, particularly for those older than 65 years. But it is clear that testosterone levels decline with time, and the idea to supplement testosterone is hardly new. Like all treatments, testosterone supplementation has risks and benefits. Some risks are predictable, like exacerbating the symptoms of benign prostatic hyperplasia. Some risks seem to come completely out of left field. That’s what we have today, in a study suggesting that testosterone supplementation increases the risk for bone fractures.
Let me set the stage here by saying that nearly all prior research into the effects of testosterone supplementation has suggested that it is pretty good for bone health. It increases bone mineral density, bone strength, and improves bone architecture.
So if you were to do a randomized trial of testosterone supplementation and look at fracture risk in the testosterone group compared with the placebo group, you would expect the fracture risk would be much lower in those getting supplemented. Of course, this is why we actually do studies instead of assuming we know the answer already — because in this case, you’d be wrong.
I’m talking about this study, appearing in The New England Journal of Medicine.
It’s a prespecified secondary analysis of a randomized trial known as the TRAVERSE trial, which randomly assigned 5246 men with low testosterone levels to transdermal testosterone gel vs placebo. The primary goal of that trial was to assess the cardiovascular risk associated with testosterone supplementation, and the major take-home was that there was no difference in cardiovascular event rates between the testosterone and placebo groups.
This secondary analysis looked at fracture incidence. Researchers contacted participants multiple times in the first year of the study and yearly thereafter. Each time, they asked whether the participant had sustained a fracture. If they answered in the affirmative, a request for medical records was made and the researchers, still blinded to randomization status, adjudicated whether there was indeed a fracture or not, along with some details as to location, situation, and so on.
This was a big study, though, and that translates to just a 3.5% fracture rate in testosterone vs 2.5% in control, but the difference was statistically significant.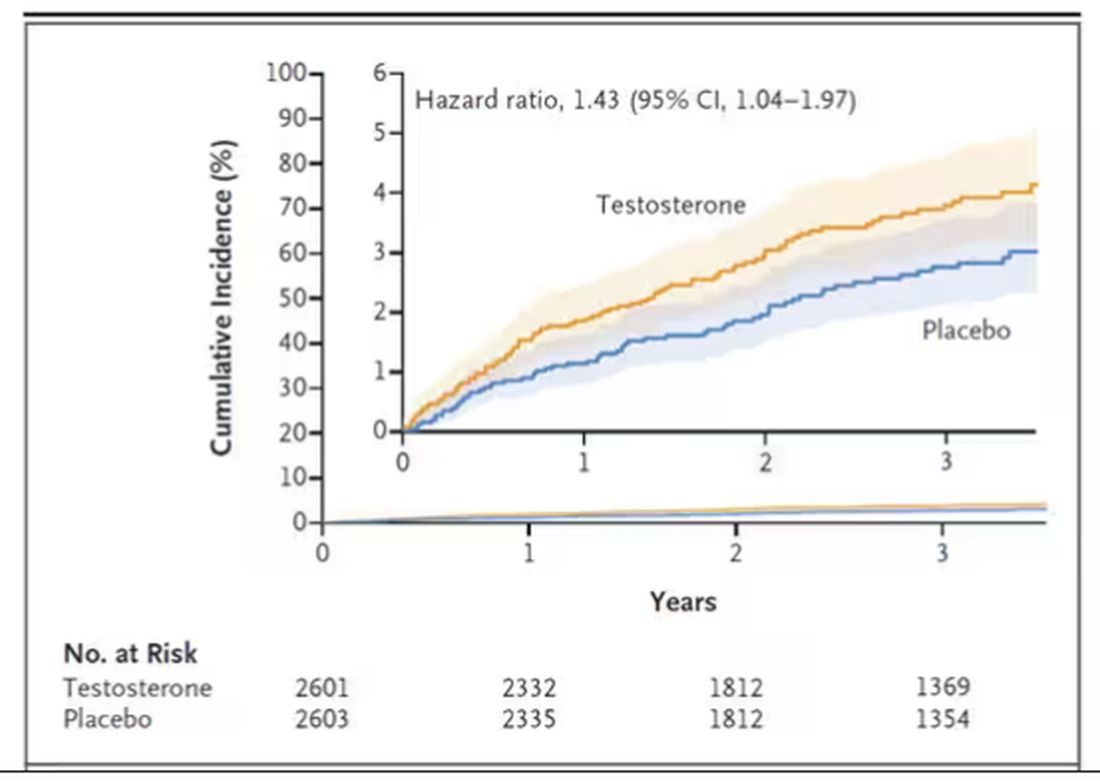
This difference persisted across various fracture types (non–high-impact fractures, for example) after excluding the small percentage of men taking osteoporosis medication.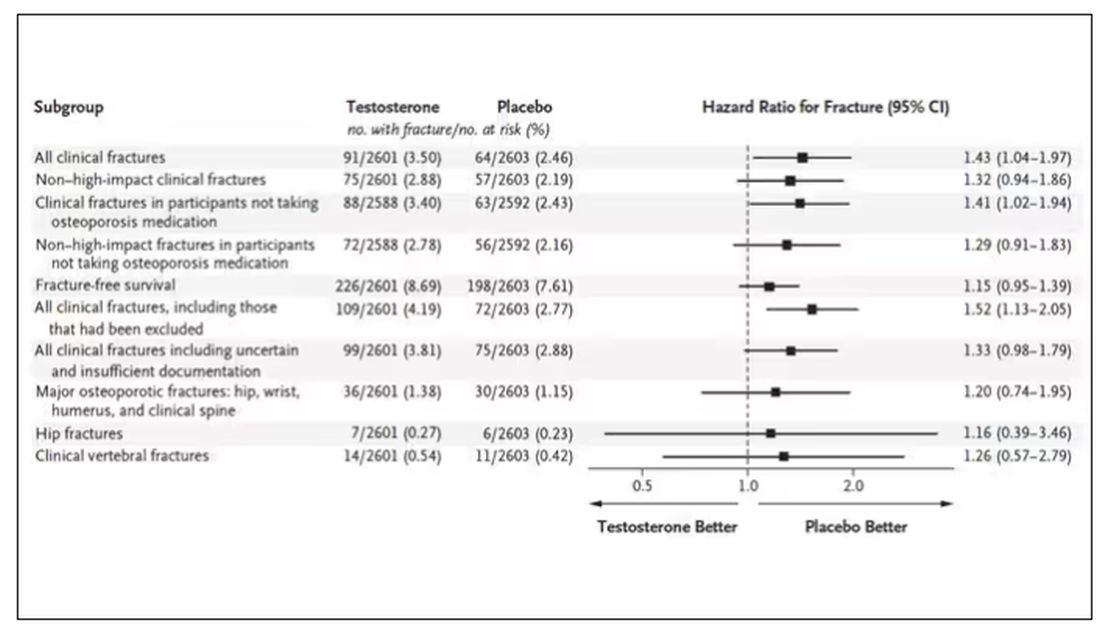
How does a drug that increases bone mineral density and bone strength increase the risk for fracture?
Well, one clue — and this was pointed out in a nice editorial by Matthis Grossman and Bradley Anawalt — is that the increased risk for fracture occurs quite soon after starting treatment, which is not consistent with direct bone effects. Rather, this might represent behavioral differences. Testosterone supplementation seems to increase energy levels; might it lead men to engage in activities that put them at higher risk for fracture?
Regardless of the cause, this adds to our knowledge about the rather complex mix of risks and benefits of testosterone supplementation and probably puts a bit more weight on the risks side. The truth is that testosterone levels do decline with age, as do many things, and it may not be appropriate to try to fight against that in all people. It’s worth noting that all of these studies use low levels of total serum testosterone as an entry criterion. But total testosterone is not what your body “sees.” It sees free testosterone, the portion not bound to sex hormone–binding globulin. And that binding protein is affected by lots of stuff — diabetes and obesity lower it, for example — making total testosterone levels seem low when free testosterone might be just fine.
In other words, testosterone supplementation is probably not terrible, but it is definitely not the cure for aging. In situations like this, we need better data to guide exactly who will benefit from the therapy and who will only be exposed to the risks.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I am showing you a graph without any labels.
What could this line represent? The stock price of some company that made a big splash but failed to live up to expectations? An outbreak curve charting the introduction of a new infectious agent to a population? The performance of a viral tweet?
I’ll tell you what it is in a moment, but I wanted you to recognize that there is something inherently wistful in this shape, something that speaks of past glory and inevitable declines. It’s a graph that induces a feeling of resistance — no, do not go gently into that good night.
The graph actually represents (roughly) the normal level of serum testosterone in otherwise-healthy men as they age.
A caveat here: These numbers are not as well defined as I made them seem on this graph, particularly for those older than 65 years. But it is clear that testosterone levels decline with time, and the idea to supplement testosterone is hardly new. Like all treatments, testosterone supplementation has risks and benefits. Some risks are predictable, like exacerbating the symptoms of benign prostatic hyperplasia. Some risks seem to come completely out of left field. That’s what we have today, in a study suggesting that testosterone supplementation increases the risk for bone fractures.
Let me set the stage here by saying that nearly all prior research into the effects of testosterone supplementation has suggested that it is pretty good for bone health. It increases bone mineral density, bone strength, and improves bone architecture.
So if you were to do a randomized trial of testosterone supplementation and look at fracture risk in the testosterone group compared with the placebo group, you would expect the fracture risk would be much lower in those getting supplemented. Of course, this is why we actually do studies instead of assuming we know the answer already — because in this case, you’d be wrong.
I’m talking about this study, appearing in The New England Journal of Medicine.
It’s a prespecified secondary analysis of a randomized trial known as the TRAVERSE trial, which randomly assigned 5246 men with low testosterone levels to transdermal testosterone gel vs placebo. The primary goal of that trial was to assess the cardiovascular risk associated with testosterone supplementation, and the major take-home was that there was no difference in cardiovascular event rates between the testosterone and placebo groups.
This secondary analysis looked at fracture incidence. Researchers contacted participants multiple times in the first year of the study and yearly thereafter. Each time, they asked whether the participant had sustained a fracture. If they answered in the affirmative, a request for medical records was made and the researchers, still blinded to randomization status, adjudicated whether there was indeed a fracture or not, along with some details as to location, situation, and so on.
This was a big study, though, and that translates to just a 3.5% fracture rate in testosterone vs 2.5% in control, but the difference was statistically significant.
This difference persisted across various fracture types (non–high-impact fractures, for example) after excluding the small percentage of men taking osteoporosis medication.
How does a drug that increases bone mineral density and bone strength increase the risk for fracture?
Well, one clue — and this was pointed out in a nice editorial by Matthis Grossman and Bradley Anawalt — is that the increased risk for fracture occurs quite soon after starting treatment, which is not consistent with direct bone effects. Rather, this might represent behavioral differences. Testosterone supplementation seems to increase energy levels; might it lead men to engage in activities that put them at higher risk for fracture?
Regardless of the cause, this adds to our knowledge about the rather complex mix of risks and benefits of testosterone supplementation and probably puts a bit more weight on the risks side. The truth is that testosterone levels do decline with age, as do many things, and it may not be appropriate to try to fight against that in all people. It’s worth noting that all of these studies use low levels of total serum testosterone as an entry criterion. But total testosterone is not what your body “sees.” It sees free testosterone, the portion not bound to sex hormone–binding globulin. And that binding protein is affected by lots of stuff — diabetes and obesity lower it, for example — making total testosterone levels seem low when free testosterone might be just fine.
In other words, testosterone supplementation is probably not terrible, but it is definitely not the cure for aging. In situations like this, we need better data to guide exactly who will benefit from the therapy and who will only be exposed to the risks.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I am showing you a graph without any labels.
What could this line represent? The stock price of some company that made a big splash but failed to live up to expectations? An outbreak curve charting the introduction of a new infectious agent to a population? The performance of a viral tweet?
I’ll tell you what it is in a moment, but I wanted you to recognize that there is something inherently wistful in this shape, something that speaks of past glory and inevitable declines. It’s a graph that induces a feeling of resistance — no, do not go gently into that good night.
The graph actually represents (roughly) the normal level of serum testosterone in otherwise-healthy men as they age.
A caveat here: These numbers are not as well defined as I made them seem on this graph, particularly for those older than 65 years. But it is clear that testosterone levels decline with time, and the idea to supplement testosterone is hardly new. Like all treatments, testosterone supplementation has risks and benefits. Some risks are predictable, like exacerbating the symptoms of benign prostatic hyperplasia. Some risks seem to come completely out of left field. That’s what we have today, in a study suggesting that testosterone supplementation increases the risk for bone fractures.
Let me set the stage here by saying that nearly all prior research into the effects of testosterone supplementation has suggested that it is pretty good for bone health. It increases bone mineral density, bone strength, and improves bone architecture.
So if you were to do a randomized trial of testosterone supplementation and look at fracture risk in the testosterone group compared with the placebo group, you would expect the fracture risk would be much lower in those getting supplemented. Of course, this is why we actually do studies instead of assuming we know the answer already — because in this case, you’d be wrong.
I’m talking about this study, appearing in The New England Journal of Medicine.
It’s a prespecified secondary analysis of a randomized trial known as the TRAVERSE trial, which randomly assigned 5246 men with low testosterone levels to transdermal testosterone gel vs placebo. The primary goal of that trial was to assess the cardiovascular risk associated with testosterone supplementation, and the major take-home was that there was no difference in cardiovascular event rates between the testosterone and placebo groups.
This secondary analysis looked at fracture incidence. Researchers contacted participants multiple times in the first year of the study and yearly thereafter. Each time, they asked whether the participant had sustained a fracture. If they answered in the affirmative, a request for medical records was made and the researchers, still blinded to randomization status, adjudicated whether there was indeed a fracture or not, along with some details as to location, situation, and so on.
This was a big study, though, and that translates to just a 3.5% fracture rate in testosterone vs 2.5% in control, but the difference was statistically significant.
This difference persisted across various fracture types (non–high-impact fractures, for example) after excluding the small percentage of men taking osteoporosis medication.
How does a drug that increases bone mineral density and bone strength increase the risk for fracture?
Well, one clue — and this was pointed out in a nice editorial by Matthis Grossman and Bradley Anawalt — is that the increased risk for fracture occurs quite soon after starting treatment, which is not consistent with direct bone effects. Rather, this might represent behavioral differences. Testosterone supplementation seems to increase energy levels; might it lead men to engage in activities that put them at higher risk for fracture?
Regardless of the cause, this adds to our knowledge about the rather complex mix of risks and benefits of testosterone supplementation and probably puts a bit more weight on the risks side. The truth is that testosterone levels do decline with age, as do many things, and it may not be appropriate to try to fight against that in all people. It’s worth noting that all of these studies use low levels of total serum testosterone as an entry criterion. But total testosterone is not what your body “sees.” It sees free testosterone, the portion not bound to sex hormone–binding globulin. And that binding protein is affected by lots of stuff — diabetes and obesity lower it, for example — making total testosterone levels seem low when free testosterone might be just fine.
In other words, testosterone supplementation is probably not terrible, but it is definitely not the cure for aging. In situations like this, we need better data to guide exactly who will benefit from the therapy and who will only be exposed to the risks.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Smoking Associated With Increased Risk for Hair Loss Among Men
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
Prostate Risks Similar for Testosterone Therapy and Placebo
TOPLINE:
including cancer.
METHODOLOGY:
- Uncertainty and concern exist about a link between prostate cancer risk and testosterone levels. Most professional society guidelines recommend against TRT in men with a history of or an increased risk for prostate cancer.
- The Testosterone Replacement Therapy for Assessment of Long-Term Vascular Events and Efficacy Response in Hypogonadal Men included 5204 men (ages 45-80, 17% Black, 80% White), randomly assigned to receive testosterone gel or placebo.
- Men with a history of cardiovascular disease or increased cardiovascular risk were evaluated to exclude those at increased prostate cancer risk (fasting testosterone < 300 ng/dL, ≥ 1 hypogonadal symptoms).
- The primary prostate safety endpoint was high-grade prostate cancer incidence (Gleason score, ≥ 4 + 3).
- Secondary endpoints were incidences of any prostate cancer, acute urinary retention, invasive procedure for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms.
TAKEAWAY:
- During 14,304 person-years of follow-up, high-grade prostate cancer incidence did not differ significantly between the TRT and placebo (0.19% vs 0.12%; P = .51) groups.
- The incidences of prostate cancer, acute urinary retention, invasive procedures for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms were also similar between the groups.
- TRT did not lead to an increase in lower urinary tract symptoms.
- The increase in prostate-specific antigen (PSA) levels was higher in the TRT group than in the placebo group (P < .001). However, the between-group difference did not widen after 12 months.
IN PRACTICE:
For “clinicians and patients who are considering testosterone replacement therapy for hypogonadism,” wrote the authors, “the study’s findings will facilitate a more informed appraisal of the potential prostate risks of testosterone replacement therapy.”
SOURCE:
Shalender Bhasin, MB, BS, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts, led the study. It was published online in JAMA Network Open.
LIMITATIONS:
- The study findings do not apply to men with known prostate cancer or higher PSA values or those without confirmed hypogonadism.
- Although the TRAVERSE study was longer than many contemporary trials, carcinogens may require many years to induce malignant neoplasms.
- The trial’s structured evaluation of men after PSA testing did not include prostate imaging or other biomarker tests, which could affect the decision to perform a biopsy.
DISCLOSURES:
This study was funded by a consortium of testosterone manufacturers led by AbbVie Inc with additional financial support from Endo Pharmaceuticals, Acerus Pharmaceuticals Corp, and Upsher-Smith Laboratories. Mr. Bhasin and two coauthors declared receiving grants, consulting and personal fees, and other ties with pharmaceutical and device companies and other sources.
A version of this article appeared on Medscape.com.
TOPLINE:
including cancer.
METHODOLOGY:
- Uncertainty and concern exist about a link between prostate cancer risk and testosterone levels. Most professional society guidelines recommend against TRT in men with a history of or an increased risk for prostate cancer.
- The Testosterone Replacement Therapy for Assessment of Long-Term Vascular Events and Efficacy Response in Hypogonadal Men included 5204 men (ages 45-80, 17% Black, 80% White), randomly assigned to receive testosterone gel or placebo.
- Men with a history of cardiovascular disease or increased cardiovascular risk were evaluated to exclude those at increased prostate cancer risk (fasting testosterone < 300 ng/dL, ≥ 1 hypogonadal symptoms).
- The primary prostate safety endpoint was high-grade prostate cancer incidence (Gleason score, ≥ 4 + 3).
- Secondary endpoints were incidences of any prostate cancer, acute urinary retention, invasive procedure for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms.
TAKEAWAY:
- During 14,304 person-years of follow-up, high-grade prostate cancer incidence did not differ significantly between the TRT and placebo (0.19% vs 0.12%; P = .51) groups.
- The incidences of prostate cancer, acute urinary retention, invasive procedures for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms were also similar between the groups.
- TRT did not lead to an increase in lower urinary tract symptoms.
- The increase in prostate-specific antigen (PSA) levels was higher in the TRT group than in the placebo group (P < .001). However, the between-group difference did not widen after 12 months.
IN PRACTICE:
For “clinicians and patients who are considering testosterone replacement therapy for hypogonadism,” wrote the authors, “the study’s findings will facilitate a more informed appraisal of the potential prostate risks of testosterone replacement therapy.”
SOURCE:
Shalender Bhasin, MB, BS, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts, led the study. It was published online in JAMA Network Open.
LIMITATIONS:
- The study findings do not apply to men with known prostate cancer or higher PSA values or those without confirmed hypogonadism.
- Although the TRAVERSE study was longer than many contemporary trials, carcinogens may require many years to induce malignant neoplasms.
- The trial’s structured evaluation of men after PSA testing did not include prostate imaging or other biomarker tests, which could affect the decision to perform a biopsy.
DISCLOSURES:
This study was funded by a consortium of testosterone manufacturers led by AbbVie Inc with additional financial support from Endo Pharmaceuticals, Acerus Pharmaceuticals Corp, and Upsher-Smith Laboratories. Mr. Bhasin and two coauthors declared receiving grants, consulting and personal fees, and other ties with pharmaceutical and device companies and other sources.
A version of this article appeared on Medscape.com.
TOPLINE:
including cancer.
METHODOLOGY:
- Uncertainty and concern exist about a link between prostate cancer risk and testosterone levels. Most professional society guidelines recommend against TRT in men with a history of or an increased risk for prostate cancer.
- The Testosterone Replacement Therapy for Assessment of Long-Term Vascular Events and Efficacy Response in Hypogonadal Men included 5204 men (ages 45-80, 17% Black, 80% White), randomly assigned to receive testosterone gel or placebo.
- Men with a history of cardiovascular disease or increased cardiovascular risk were evaluated to exclude those at increased prostate cancer risk (fasting testosterone < 300 ng/dL, ≥ 1 hypogonadal symptoms).
- The primary prostate safety endpoint was high-grade prostate cancer incidence (Gleason score, ≥ 4 + 3).
- Secondary endpoints were incidences of any prostate cancer, acute urinary retention, invasive procedure for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms.
TAKEAWAY:
- During 14,304 person-years of follow-up, high-grade prostate cancer incidence did not differ significantly between the TRT and placebo (0.19% vs 0.12%; P = .51) groups.
- The incidences of prostate cancer, acute urinary retention, invasive procedures for benign prostatic hyperplasia, prostate biopsy, and new pharmacologic treatment for lower urinary tract symptoms were also similar between the groups.
- TRT did not lead to an increase in lower urinary tract symptoms.
- The increase in prostate-specific antigen (PSA) levels was higher in the TRT group than in the placebo group (P < .001). However, the between-group difference did not widen after 12 months.
IN PRACTICE:
For “clinicians and patients who are considering testosterone replacement therapy for hypogonadism,” wrote the authors, “the study’s findings will facilitate a more informed appraisal of the potential prostate risks of testosterone replacement therapy.”
SOURCE:
Shalender Bhasin, MB, BS, Brigham and Women’s Hospital, Harvard Medical School, Boston, Massachusetts, led the study. It was published online in JAMA Network Open.
LIMITATIONS:
- The study findings do not apply to men with known prostate cancer or higher PSA values or those without confirmed hypogonadism.
- Although the TRAVERSE study was longer than many contemporary trials, carcinogens may require many years to induce malignant neoplasms.
- The trial’s structured evaluation of men after PSA testing did not include prostate imaging or other biomarker tests, which could affect the decision to perform a biopsy.
DISCLOSURES:
This study was funded by a consortium of testosterone manufacturers led by AbbVie Inc with additional financial support from Endo Pharmaceuticals, Acerus Pharmaceuticals Corp, and Upsher-Smith Laboratories. Mr. Bhasin and two coauthors declared receiving grants, consulting and personal fees, and other ties with pharmaceutical and device companies and other sources.
A version of this article appeared on Medscape.com.
Radiation Oncologists Fight for Payment Reform Amid Cuts
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
Oncologists Sound the Alarm About Rise of White Bagging
For years, oncologist John DiPersio, MD, PhD, had faced frustrating encounters with insurers that only cover medications through a process called white bagging.
Instead of the traditional buy-and-bill pathway where oncologists purchase specialty drugs, such as infusion medications, directly from the distributor or manufacturer, white bagging requires physicians to receive these drugs from a specialty pharmacy.
On its face, the differences may seem minor. However, as Dr. DiPersio knows well, the consequences for oncologists and patients are not.
That is why Dr. DiPersio’s cancer center does not allow white bagging.
And when insurers refuse to reconsider the white bagging policy, his cancer team is left with few options.
“Sometimes, we have to redirect patients to other places,” said Dr. DiPersio, a bone marrow transplant specialist at Siteman Cancer Center, Washington University, St. Louis.
In emergency instances where patients cannot wait, Dr. DiPersio’s team will administer their own stock of a drug. In such cases, “we accept the fact that by not allowing white bagging, there may be nonpayment. We take the hit as far as cost.”
Increasingly, white bagging mandates are becoming harder for practices to avoid.
In a 2021 survey, 87% of Association of Community Cancer Centers members said white bagging has become an insurer mandate for some of their patients.
A 2023 analysis from Adam J. Fein, PhD, of Drug Channels Institute, Philadelphia, found that white bagging accounted for 17% of infused oncology product sourcing from clinics and 38% from hospital outpatient departments, up from 15% to 28% in 2019. Another practice called brown bagging, where specialty pharmacies send drugs directly to patients, creates many of the same issues but is much less prevalent than white bagging.
This change reflects “the broader battle over oncology margins” and insurers’ “attempts to shift costs to providers, patients, and manufacturers,” Dr. Fein wrote in his 2023 report.
White Bagging: Who Benefits?
At its core, white bagging changes how drugs are covered and reimbursed. Under buy and bill, drugs fall under a patient’s medical benefit. Oncologists purchase drugs directly from the manufacturer or distributor and receive reimbursement from the insurance company for both the cost of the drug as well as for administering it to patients.
Under white bagging, drugs fall under a patient’s pharmacy benefit. In these instances, a specialty pharmacy prepares the infusion ahead of time and ships it directly to the physician’s office or clinic. Because oncologists do not purchase the drug directly, they cannot bill insurers for it; instead, the pharmacy receives reimbursement for the drug and the provider is reimbursed for administering it.
Insurance companies argue that white bagging reduces patients’ out-of-pocket costs “by preventing hospitals and physicians from charging exorbitant fees to buy and store specialty medicines themselves,” according to advocacy group America’s Health Insurance Plans (AHIP).
Data from AHIP suggested that hospitals mark up the price of cancer drugs considerably, charging about twice as much as a specialty pharmacy, and that physician’s offices also charge about 23% more. However, these figures highlight how much insurers are billed, not necessarily how much patients ultimately pay.
Other evidence shows that white bagging raises costs for patients while reducing reimbursement for oncologists and saving insurance companies money.
A recent analysis in JAMA Network Open, which looked at 50 cancer drugs associated with the highest total spending from the 2020 Medicare Part B, found that mean insurance payments to providers were more than $2000 lower for drugs distributed under bagging than traditional buy and bill: $7405 vs $9547 per patient per month. Investigators found the same pattern in median insurance payments: $5746 vs $6681. Patients also paid more out-of-pocket each month with bagging vs buy and bill: $315 vs $145.
For patients with private insurance, “out-of-pocket costs were higher under bagging practice than the traditional buy-and-bill practice,” said lead author Ya-Chen Tina Shih, PhD, a professor in the department of radiation oncology at UCLA Health, Los Angeles.
White bagging is entirely for the profit of health insurers, specialty pharmacies, and pharmacy benefit managers, the middlemen who negotiate drug prices on behalf of payers.
Many people may not realize the underlying money-making strategies behind white bagging, explained Ted Okon, executive director for Community Oncology Alliance, which opposes the practice. Often, an insurer, pharmacy benefit manager, and mail order pharmacy involved in the process are all affiliated with the same corporation. In such cases, an insurer has a financial motive to control the source of medications and steer business to its affiliated pharmacies, Mr. Okon said.
When a single corporation owns numerous parts of the drug supply chain, insurers end up having “sway over what drug to use and then how the patient is going to get it,” Mr. Okon said. If the specialty pharmacy is a 340B contract pharmacy, it likely also receives a sizable discount on the drug and can make more money through white bagging.
Dangerous to Patients?
On the safety front, proponents of white bagging say the process is safe and efficient.
Specialty pharmacies are used only for prescription drugs that can be safely delivered, said AHIP spokesman David Allen.
In addition to having the same supply chain safety requirements as any other dispensing pharmacy, “specialty pharmacies also must meet additional safety requirements for specialty drugs” to ensure “the safe storage, handling, and dispensing of the drugs,” Mr. Allen explained.
However, oncologists argue that white bagging can be dangerous.
With white bagging, specialty pharmacies send a specified dose to practices, which does not allow practices to source and mix the drug themselves or make essential last-minute dose-related changes — something that happens every day in the clinic, said Debra Patt, MD, PhD, MBA, executive vice president for policy and strategy for Texas Oncology, Dallas.
White bagging also increases the risk for drug contamination, results in drug waste if the medication can’t be used, and can create delays in care.
Essentially, white bagging takes control away from oncologists and makes patient care more unpredictable and complex, explained Dr. Patt, president of the Texas Society of Clinical Oncology, Rockville, Maryland.
Dr. Patt, who does not allow white bagging in her practice, recalled a recent patient with metastatic breast cancer who came to the clinic for trastuzumab deruxtecan. The patient had been experiencing acute abdominal pain. After an exam and CT, Dr. Patt found the breast cancer had grown and moved into the patient’s liver.
“I had to discontinue that plan and change to a different chemotherapy,” she said. “If we had white bagged, that would have been a waste of several thousand dollars. Also, the patient would have to wait for the new medication to be white bagged, a delay that would be at least a week and the patient would have to come back at another time.”
When asked about the safety concerns associated with white bagging, Lemrey “Al” Carter, MS, PharmD, RPh, executive director of the National Association of Boards of Pharmacy (NABP), said the NABP “acknowledges that all these issues exist.
“It is unfortunate if patient care or costs are negatively impacted,” Dr. Carter said, adding that “boards of pharmacy can investigate if they are made aware of safety concerns at the pharmacy level. If a violation of the pharmacy laws or rules is found, boards can take action.”
More Legislation to Prevent Bagging
As white bagging mandates from insurance companies ramp up, more practices and states are banning it.
In the Association of Community Cancer Centers’ 2021 survey, 59% of members said their cancer program or practice does not allow white bagging.
At least 15 states have introduced legislation that restricts and/or prohibits white and brown bagging practices, according to a 2023 report by the Institute for Clinical and Economic Review. Some of the proposed laws would restrict mandates by stipulating that physicians are reimbursed at the contracted amount for clinician-administered drugs, whether obtained from a pharmacy or the manufacturer.
Louisiana, Vermont, and Minnesota were the first to enact anti–white bagging laws. Louisiana’s law, for example, enacted in 2021, bans white bagging and requires insurers to reimburse providers for physician-administered drugs if obtained from out-of-network pharmacies.
When the legislation passed, white bagging was just starting to enter the healthcare market in Louisiana, and the state wanted to act proactively, said Kathy W. Oubre, MS, CEO of the Pontchartrain Cancer Center, Covington, Louisiana, and president of the Coalition of Hematology and Oncology Practices, Mountain View, California.
“We recognized the growing concern around it,” Ms. Oubre said. The state legislature at the time included physicians and pharmacists who “really understood from a practice and patient perspective, the harm that policy could do.”
Ms. Oubre would like to see more legislation in other states and believes Louisiana’s law is a good model.
At the federal level, the American Hospital Association and American Society of Health-System Pharmacists have also urged the US Food and Drug Administration to take appropriate enforcement action to protect patients from white bagging.
Legislation that bars white bagging mandates is the most reasonable way to support timely and appropriate access to cancer care, Dr. Patt said. In the absence of such legislation, she said oncologists can only opt out of insurance contracts that may require the practice.
“That is a difficult position to put oncologists in,” she said.
A version of this article appeared on Medscape.com.
For years, oncologist John DiPersio, MD, PhD, had faced frustrating encounters with insurers that only cover medications through a process called white bagging.
Instead of the traditional buy-and-bill pathway where oncologists purchase specialty drugs, such as infusion medications, directly from the distributor or manufacturer, white bagging requires physicians to receive these drugs from a specialty pharmacy.
On its face, the differences may seem minor. However, as Dr. DiPersio knows well, the consequences for oncologists and patients are not.
That is why Dr. DiPersio’s cancer center does not allow white bagging.
And when insurers refuse to reconsider the white bagging policy, his cancer team is left with few options.
“Sometimes, we have to redirect patients to other places,” said Dr. DiPersio, a bone marrow transplant specialist at Siteman Cancer Center, Washington University, St. Louis.
In emergency instances where patients cannot wait, Dr. DiPersio’s team will administer their own stock of a drug. In such cases, “we accept the fact that by not allowing white bagging, there may be nonpayment. We take the hit as far as cost.”
Increasingly, white bagging mandates are becoming harder for practices to avoid.
In a 2021 survey, 87% of Association of Community Cancer Centers members said white bagging has become an insurer mandate for some of their patients.
A 2023 analysis from Adam J. Fein, PhD, of Drug Channels Institute, Philadelphia, found that white bagging accounted for 17% of infused oncology product sourcing from clinics and 38% from hospital outpatient departments, up from 15% to 28% in 2019. Another practice called brown bagging, where specialty pharmacies send drugs directly to patients, creates many of the same issues but is much less prevalent than white bagging.
This change reflects “the broader battle over oncology margins” and insurers’ “attempts to shift costs to providers, patients, and manufacturers,” Dr. Fein wrote in his 2023 report.
White Bagging: Who Benefits?
At its core, white bagging changes how drugs are covered and reimbursed. Under buy and bill, drugs fall under a patient’s medical benefit. Oncologists purchase drugs directly from the manufacturer or distributor and receive reimbursement from the insurance company for both the cost of the drug as well as for administering it to patients.
Under white bagging, drugs fall under a patient’s pharmacy benefit. In these instances, a specialty pharmacy prepares the infusion ahead of time and ships it directly to the physician’s office or clinic. Because oncologists do not purchase the drug directly, they cannot bill insurers for it; instead, the pharmacy receives reimbursement for the drug and the provider is reimbursed for administering it.
Insurance companies argue that white bagging reduces patients’ out-of-pocket costs “by preventing hospitals and physicians from charging exorbitant fees to buy and store specialty medicines themselves,” according to advocacy group America’s Health Insurance Plans (AHIP).
Data from AHIP suggested that hospitals mark up the price of cancer drugs considerably, charging about twice as much as a specialty pharmacy, and that physician’s offices also charge about 23% more. However, these figures highlight how much insurers are billed, not necessarily how much patients ultimately pay.
Other evidence shows that white bagging raises costs for patients while reducing reimbursement for oncologists and saving insurance companies money.
A recent analysis in JAMA Network Open, which looked at 50 cancer drugs associated with the highest total spending from the 2020 Medicare Part B, found that mean insurance payments to providers were more than $2000 lower for drugs distributed under bagging than traditional buy and bill: $7405 vs $9547 per patient per month. Investigators found the same pattern in median insurance payments: $5746 vs $6681. Patients also paid more out-of-pocket each month with bagging vs buy and bill: $315 vs $145.
For patients with private insurance, “out-of-pocket costs were higher under bagging practice than the traditional buy-and-bill practice,” said lead author Ya-Chen Tina Shih, PhD, a professor in the department of radiation oncology at UCLA Health, Los Angeles.
White bagging is entirely for the profit of health insurers, specialty pharmacies, and pharmacy benefit managers, the middlemen who negotiate drug prices on behalf of payers.
Many people may not realize the underlying money-making strategies behind white bagging, explained Ted Okon, executive director for Community Oncology Alliance, which opposes the practice. Often, an insurer, pharmacy benefit manager, and mail order pharmacy involved in the process are all affiliated with the same corporation. In such cases, an insurer has a financial motive to control the source of medications and steer business to its affiliated pharmacies, Mr. Okon said.
When a single corporation owns numerous parts of the drug supply chain, insurers end up having “sway over what drug to use and then how the patient is going to get it,” Mr. Okon said. If the specialty pharmacy is a 340B contract pharmacy, it likely also receives a sizable discount on the drug and can make more money through white bagging.
Dangerous to Patients?
On the safety front, proponents of white bagging say the process is safe and efficient.
Specialty pharmacies are used only for prescription drugs that can be safely delivered, said AHIP spokesman David Allen.
In addition to having the same supply chain safety requirements as any other dispensing pharmacy, “specialty pharmacies also must meet additional safety requirements for specialty drugs” to ensure “the safe storage, handling, and dispensing of the drugs,” Mr. Allen explained.
However, oncologists argue that white bagging can be dangerous.
With white bagging, specialty pharmacies send a specified dose to practices, which does not allow practices to source and mix the drug themselves or make essential last-minute dose-related changes — something that happens every day in the clinic, said Debra Patt, MD, PhD, MBA, executive vice president for policy and strategy for Texas Oncology, Dallas.
White bagging also increases the risk for drug contamination, results in drug waste if the medication can’t be used, and can create delays in care.
Essentially, white bagging takes control away from oncologists and makes patient care more unpredictable and complex, explained Dr. Patt, president of the Texas Society of Clinical Oncology, Rockville, Maryland.
Dr. Patt, who does not allow white bagging in her practice, recalled a recent patient with metastatic breast cancer who came to the clinic for trastuzumab deruxtecan. The patient had been experiencing acute abdominal pain. After an exam and CT, Dr. Patt found the breast cancer had grown and moved into the patient’s liver.
“I had to discontinue that plan and change to a different chemotherapy,” she said. “If we had white bagged, that would have been a waste of several thousand dollars. Also, the patient would have to wait for the new medication to be white bagged, a delay that would be at least a week and the patient would have to come back at another time.”
When asked about the safety concerns associated with white bagging, Lemrey “Al” Carter, MS, PharmD, RPh, executive director of the National Association of Boards of Pharmacy (NABP), said the NABP “acknowledges that all these issues exist.
“It is unfortunate if patient care or costs are negatively impacted,” Dr. Carter said, adding that “boards of pharmacy can investigate if they are made aware of safety concerns at the pharmacy level. If a violation of the pharmacy laws or rules is found, boards can take action.”
More Legislation to Prevent Bagging
As white bagging mandates from insurance companies ramp up, more practices and states are banning it.
In the Association of Community Cancer Centers’ 2021 survey, 59% of members said their cancer program or practice does not allow white bagging.
At least 15 states have introduced legislation that restricts and/or prohibits white and brown bagging practices, according to a 2023 report by the Institute for Clinical and Economic Review. Some of the proposed laws would restrict mandates by stipulating that physicians are reimbursed at the contracted amount for clinician-administered drugs, whether obtained from a pharmacy or the manufacturer.
Louisiana, Vermont, and Minnesota were the first to enact anti–white bagging laws. Louisiana’s law, for example, enacted in 2021, bans white bagging and requires insurers to reimburse providers for physician-administered drugs if obtained from out-of-network pharmacies.
When the legislation passed, white bagging was just starting to enter the healthcare market in Louisiana, and the state wanted to act proactively, said Kathy W. Oubre, MS, CEO of the Pontchartrain Cancer Center, Covington, Louisiana, and president of the Coalition of Hematology and Oncology Practices, Mountain View, California.
“We recognized the growing concern around it,” Ms. Oubre said. The state legislature at the time included physicians and pharmacists who “really understood from a practice and patient perspective, the harm that policy could do.”
Ms. Oubre would like to see more legislation in other states and believes Louisiana’s law is a good model.
At the federal level, the American Hospital Association and American Society of Health-System Pharmacists have also urged the US Food and Drug Administration to take appropriate enforcement action to protect patients from white bagging.
Legislation that bars white bagging mandates is the most reasonable way to support timely and appropriate access to cancer care, Dr. Patt said. In the absence of such legislation, she said oncologists can only opt out of insurance contracts that may require the practice.
“That is a difficult position to put oncologists in,” she said.
A version of this article appeared on Medscape.com.
For years, oncologist John DiPersio, MD, PhD, had faced frustrating encounters with insurers that only cover medications through a process called white bagging.
Instead of the traditional buy-and-bill pathway where oncologists purchase specialty drugs, such as infusion medications, directly from the distributor or manufacturer, white bagging requires physicians to receive these drugs from a specialty pharmacy.
On its face, the differences may seem minor. However, as Dr. DiPersio knows well, the consequences for oncologists and patients are not.
That is why Dr. DiPersio’s cancer center does not allow white bagging.
And when insurers refuse to reconsider the white bagging policy, his cancer team is left with few options.
“Sometimes, we have to redirect patients to other places,” said Dr. DiPersio, a bone marrow transplant specialist at Siteman Cancer Center, Washington University, St. Louis.
In emergency instances where patients cannot wait, Dr. DiPersio’s team will administer their own stock of a drug. In such cases, “we accept the fact that by not allowing white bagging, there may be nonpayment. We take the hit as far as cost.”
Increasingly, white bagging mandates are becoming harder for practices to avoid.
In a 2021 survey, 87% of Association of Community Cancer Centers members said white bagging has become an insurer mandate for some of their patients.
A 2023 analysis from Adam J. Fein, PhD, of Drug Channels Institute, Philadelphia, found that white bagging accounted for 17% of infused oncology product sourcing from clinics and 38% from hospital outpatient departments, up from 15% to 28% in 2019. Another practice called brown bagging, where specialty pharmacies send drugs directly to patients, creates many of the same issues but is much less prevalent than white bagging.
This change reflects “the broader battle over oncology margins” and insurers’ “attempts to shift costs to providers, patients, and manufacturers,” Dr. Fein wrote in his 2023 report.
White Bagging: Who Benefits?
At its core, white bagging changes how drugs are covered and reimbursed. Under buy and bill, drugs fall under a patient’s medical benefit. Oncologists purchase drugs directly from the manufacturer or distributor and receive reimbursement from the insurance company for both the cost of the drug as well as for administering it to patients.
Under white bagging, drugs fall under a patient’s pharmacy benefit. In these instances, a specialty pharmacy prepares the infusion ahead of time and ships it directly to the physician’s office or clinic. Because oncologists do not purchase the drug directly, they cannot bill insurers for it; instead, the pharmacy receives reimbursement for the drug and the provider is reimbursed for administering it.
Insurance companies argue that white bagging reduces patients’ out-of-pocket costs “by preventing hospitals and physicians from charging exorbitant fees to buy and store specialty medicines themselves,” according to advocacy group America’s Health Insurance Plans (AHIP).
Data from AHIP suggested that hospitals mark up the price of cancer drugs considerably, charging about twice as much as a specialty pharmacy, and that physician’s offices also charge about 23% more. However, these figures highlight how much insurers are billed, not necessarily how much patients ultimately pay.
Other evidence shows that white bagging raises costs for patients while reducing reimbursement for oncologists and saving insurance companies money.
A recent analysis in JAMA Network Open, which looked at 50 cancer drugs associated with the highest total spending from the 2020 Medicare Part B, found that mean insurance payments to providers were more than $2000 lower for drugs distributed under bagging than traditional buy and bill: $7405 vs $9547 per patient per month. Investigators found the same pattern in median insurance payments: $5746 vs $6681. Patients also paid more out-of-pocket each month with bagging vs buy and bill: $315 vs $145.
For patients with private insurance, “out-of-pocket costs were higher under bagging practice than the traditional buy-and-bill practice,” said lead author Ya-Chen Tina Shih, PhD, a professor in the department of radiation oncology at UCLA Health, Los Angeles.
White bagging is entirely for the profit of health insurers, specialty pharmacies, and pharmacy benefit managers, the middlemen who negotiate drug prices on behalf of payers.
Many people may not realize the underlying money-making strategies behind white bagging, explained Ted Okon, executive director for Community Oncology Alliance, which opposes the practice. Often, an insurer, pharmacy benefit manager, and mail order pharmacy involved in the process are all affiliated with the same corporation. In such cases, an insurer has a financial motive to control the source of medications and steer business to its affiliated pharmacies, Mr. Okon said.
When a single corporation owns numerous parts of the drug supply chain, insurers end up having “sway over what drug to use and then how the patient is going to get it,” Mr. Okon said. If the specialty pharmacy is a 340B contract pharmacy, it likely also receives a sizable discount on the drug and can make more money through white bagging.
Dangerous to Patients?
On the safety front, proponents of white bagging say the process is safe and efficient.
Specialty pharmacies are used only for prescription drugs that can be safely delivered, said AHIP spokesman David Allen.
In addition to having the same supply chain safety requirements as any other dispensing pharmacy, “specialty pharmacies also must meet additional safety requirements for specialty drugs” to ensure “the safe storage, handling, and dispensing of the drugs,” Mr. Allen explained.
However, oncologists argue that white bagging can be dangerous.
With white bagging, specialty pharmacies send a specified dose to practices, which does not allow practices to source and mix the drug themselves or make essential last-minute dose-related changes — something that happens every day in the clinic, said Debra Patt, MD, PhD, MBA, executive vice president for policy and strategy for Texas Oncology, Dallas.
White bagging also increases the risk for drug contamination, results in drug waste if the medication can’t be used, and can create delays in care.
Essentially, white bagging takes control away from oncologists and makes patient care more unpredictable and complex, explained Dr. Patt, president of the Texas Society of Clinical Oncology, Rockville, Maryland.
Dr. Patt, who does not allow white bagging in her practice, recalled a recent patient with metastatic breast cancer who came to the clinic for trastuzumab deruxtecan. The patient had been experiencing acute abdominal pain. After an exam and CT, Dr. Patt found the breast cancer had grown and moved into the patient’s liver.
“I had to discontinue that plan and change to a different chemotherapy,” she said. “If we had white bagged, that would have been a waste of several thousand dollars. Also, the patient would have to wait for the new medication to be white bagged, a delay that would be at least a week and the patient would have to come back at another time.”
When asked about the safety concerns associated with white bagging, Lemrey “Al” Carter, MS, PharmD, RPh, executive director of the National Association of Boards of Pharmacy (NABP), said the NABP “acknowledges that all these issues exist.
“It is unfortunate if patient care or costs are negatively impacted,” Dr. Carter said, adding that “boards of pharmacy can investigate if they are made aware of safety concerns at the pharmacy level. If a violation of the pharmacy laws or rules is found, boards can take action.”
More Legislation to Prevent Bagging
As white bagging mandates from insurance companies ramp up, more practices and states are banning it.
In the Association of Community Cancer Centers’ 2021 survey, 59% of members said their cancer program or practice does not allow white bagging.
At least 15 states have introduced legislation that restricts and/or prohibits white and brown bagging practices, according to a 2023 report by the Institute for Clinical and Economic Review. Some of the proposed laws would restrict mandates by stipulating that physicians are reimbursed at the contracted amount for clinician-administered drugs, whether obtained from a pharmacy or the manufacturer.
Louisiana, Vermont, and Minnesota were the first to enact anti–white bagging laws. Louisiana’s law, for example, enacted in 2021, bans white bagging and requires insurers to reimburse providers for physician-administered drugs if obtained from out-of-network pharmacies.
When the legislation passed, white bagging was just starting to enter the healthcare market in Louisiana, and the state wanted to act proactively, said Kathy W. Oubre, MS, CEO of the Pontchartrain Cancer Center, Covington, Louisiana, and president of the Coalition of Hematology and Oncology Practices, Mountain View, California.
“We recognized the growing concern around it,” Ms. Oubre said. The state legislature at the time included physicians and pharmacists who “really understood from a practice and patient perspective, the harm that policy could do.”
Ms. Oubre would like to see more legislation in other states and believes Louisiana’s law is a good model.
At the federal level, the American Hospital Association and American Society of Health-System Pharmacists have also urged the US Food and Drug Administration to take appropriate enforcement action to protect patients from white bagging.
Legislation that bars white bagging mandates is the most reasonable way to support timely and appropriate access to cancer care, Dr. Patt said. In the absence of such legislation, she said oncologists can only opt out of insurance contracts that may require the practice.
“That is a difficult position to put oncologists in,” she said.
A version of this article appeared on Medscape.com.
Tips and Techniques to Boost Colonoscopy Quality
This transcript has been edited for clarity.
When it comes to the use of colonoscopy to reduce the risk for cancer, quality is key.
There are a number of performance improvements we can make in our practices so that we can do better. This is evident in several recently published studies and a recent review article on the topic, which I’d like to profile for you; many of these key quality indicators you can implement now.
Even though it may take more time before they’re supported in the guidelines, you’ll see that the evidence behind these is extraordinarily strong.
Increasing the Adenoma Detection Rate
Certainly, we all do what we can to increase the adenoma detection rate (ADR).
However, at the moment, the nationally recommended benchmark is to achieve an ADR of 25%, which is inordinately low. The ADR rate reported in the GIQuIC registry data is closer to 39%, and in high-level detectors, it’s actually in the greater-than-50% range.
There’s no question that we can do more, and there are a number of ways to do that.
First, This may actually decrease your withdrawal time because you don’t spend so much time trying to face these folds.
In considering tools to aid ADR, don’t forget electronic chromoendoscopy (eg, narrow-band imaging).
There are a number of new artificial intelligence options out there as well, which have been reported to increase the ADR by approximately 10%. Of importance, this improvement even occurs among expert endoscopists.
There’s also important emerging data about ADR in fecal immunochemical test (FIT)–positive patients. FIT-positive status increases the ADR threshold by 15%-20%. This places you in an ADR range of approximately 50%, which is really the norm when screening patients that present for colonoscopy because of FIT positivity.
Adenoma Per Colonoscopy: A Possible ADR Substitute
Growing evidence supports the use of adenoma per colonoscopy (APC) as a substitute to ADR. This would allow you to record every adenoma and attribute it to that index colonoscopy.
A high-quality paper showed that the APC value should be around 0.6 to achieve the current ADR minimum threshold of 25%. Having the APC < 0.6 seems to be associated with an increased risk for residual polyp. Sessile serrated lesions also increased the hazard ratio for interval colorectal cancer. This was evaluated recently with data from the New Hampshire Colonoscopy Registry, which Dr Joseph Anderson has led for so long. They showed that 21% of endoscopists had an ADR of 25% or greater but still had APCs < 0.6.
Therefore, when it comes to remedial corrective work, doctors need to be reevaluated, retrained, and educated in the ways that they can incorporate this. The APC in high-level detectors is > 1.0.
APC may be something you want to consider using internally. It does require that you place each polyp into an individual jar, which can increase incremental cost. Nonetheless, there is clear evidence that APC positively changes outcomes.
Including Sessile Serrated Lesions in ADR Detectors
Unfortunately, some of the high-level ADR detectors aren’t so “high level” when it comes to detecting sessile serrated lesions. It’s not quite as concordant as we had previously thought.
Nonetheless, there are many adjunctive things you can do with sessile serrated lesions, including narrow-band imaging and chromoendoscopy.
When it comes to establishing a discriminant, the numbers should be 5%-6% if we’re going to set a quality ratio and an index. However, this is somewhat dependent on your pathologist because they have to read these correctly. Lesions that are ≥ 6 mm above the sigmoid colon and anything in the right colon should be evaluated really closely as a sessile serrated lesion.
I’ve had indications where the pathologist says the lesion is hyperplastic, to which I say, “I’m going to follow as a sessile serrated lesion.” This is because it’s apparent to me in the endoscopic appearance and the narrow-band imaging appearance that it was characteristic of sessile serrated lesions.
Best Practices in Bowel Preparations
The US Multi-Society Task Force recommends that adequate bowel preparation should occur in 85% or more of outpatients, and for the European Society for Gastrointestinal Endoscopy, it’s 90% or more.
I’ll pass along a tip I use in my patients undergoing bowel preparation: I make them aware that during this process, they want to see a clear, yellow, urine-like color to their stool. Otherwise, many patients will think if they’ve had some diarrhea, they don’t need to finish prep. Setting that expectation for them upfront is really important.
The nurses also should be aware of this because if there’s a murky brown effluent upon presentation for the colonoscopy, there’s a greater than 50% chance that they’re going to have had an inadequate preparation. In such cases, you would want to preempt the colonoscopy and perhaps send them out for a re-prep that day or bring them back for a rescheduled appointment.
Resection Considerations
There is substantial variation when it comes to lesion resection, which makes it an important quality indicator on which to focus: High-level detectors aren’t always high-level resectors.
There are two validated instruments that you can use to gauge the adequacy of resection. Those aren’t really ready for prime time in every practice, though they may be seen in fellowship programs.
The idea here is that you want to get a ≥ 2 mm margin for cold snare polypectomy in lesions 1-10 mm in size. This has been a challenge, as findings indicate we don’t do this that well.
Joseph Anderson and colleagues recently published a study using a 2-mm resection margin. They reported that this was only possible in approximately 28% of polyps. For a 1-mm margin, the rate was 84%.
We simply need to set clearer margins when setting our snare. Make sure you’re close enough to the polyp, push down on the snare, and get a good margin of tissue.
When the sample contracts are placed into the formalin, it’s not quite so simple to define the margin at the time of the surgical resection. This often requires an audit evaluation by the pathologist.
There are two other considerations regarding resection.
The first is about the referral for surgery. Referral should not occur for any benign lesions ascribed by your endoscopic advanced imaging techniques and classifications that are not thought to have intramucosal carcinoma. These should be referred to an expert endoscopic evaluation. If you can’t do it, then somebody else should. And you shouldn’t attempt it unless you can get it totally because resection of partially resected lesions is much more complicated. The European Society of Gastrointestinal Endoscopy says this applies to any benign lesion of any size, which I think really is the emerging standard of care. You should consider and offer that to the patient. It may require a referral for outside of your institution.
The second additional consideration is around the minimization of cold forceps for removal of polyps. The US Multi-Society Task Force says cold forceps shouldn’t be used for any lesions > 2 mm, whereas for the European Society of Gastrointestinal Endoscopy, it is > 3 mm. However, it’s still done very commonly in clinical practice. Nibbling the polyp is not an option. Cold snare is actually quicker, more effective, has better outcomes, and is something that you can bill for when you look at the coding.
In summary, there are a lot of things that we can do now to improve colonoscopy. Quality indicators continue to emerge with a compelling, excellent evidence base that strongly supports their use. Given that, I think most of these are actionable now, and it’s not necessary to wait for the guidelines to begin using them. I’d therefore challenge all of us to incorporate them in our continual efforts to do better.
Dr. Johnson is professor of medicine and chief of gastroenterology at Eastern Virginia Medical School in Norfolk, and a past president of the American College of Gastroenterology. His primary focus is the clinical practice of gastroenterology. He has published extensively in the internal medicine/gastroenterology literature, with principal research interests in esophageal and colon disease, and more recently in sleep and microbiome effects on gastrointestinal health and disease. He has disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
When it comes to the use of colonoscopy to reduce the risk for cancer, quality is key.
There are a number of performance improvements we can make in our practices so that we can do better. This is evident in several recently published studies and a recent review article on the topic, which I’d like to profile for you; many of these key quality indicators you can implement now.
Even though it may take more time before they’re supported in the guidelines, you’ll see that the evidence behind these is extraordinarily strong.
Increasing the Adenoma Detection Rate
Certainly, we all do what we can to increase the adenoma detection rate (ADR).
However, at the moment, the nationally recommended benchmark is to achieve an ADR of 25%, which is inordinately low. The ADR rate reported in the GIQuIC registry data is closer to 39%, and in high-level detectors, it’s actually in the greater-than-50% range.
There’s no question that we can do more, and there are a number of ways to do that.
First, This may actually decrease your withdrawal time because you don’t spend so much time trying to face these folds.
In considering tools to aid ADR, don’t forget electronic chromoendoscopy (eg, narrow-band imaging).
There are a number of new artificial intelligence options out there as well, which have been reported to increase the ADR by approximately 10%. Of importance, this improvement even occurs among expert endoscopists.
There’s also important emerging data about ADR in fecal immunochemical test (FIT)–positive patients. FIT-positive status increases the ADR threshold by 15%-20%. This places you in an ADR range of approximately 50%, which is really the norm when screening patients that present for colonoscopy because of FIT positivity.
Adenoma Per Colonoscopy: A Possible ADR Substitute
Growing evidence supports the use of adenoma per colonoscopy (APC) as a substitute to ADR. This would allow you to record every adenoma and attribute it to that index colonoscopy.
A high-quality paper showed that the APC value should be around 0.6 to achieve the current ADR minimum threshold of 25%. Having the APC < 0.6 seems to be associated with an increased risk for residual polyp. Sessile serrated lesions also increased the hazard ratio for interval colorectal cancer. This was evaluated recently with data from the New Hampshire Colonoscopy Registry, which Dr Joseph Anderson has led for so long. They showed that 21% of endoscopists had an ADR of 25% or greater but still had APCs < 0.6.
Therefore, when it comes to remedial corrective work, doctors need to be reevaluated, retrained, and educated in the ways that they can incorporate this. The APC in high-level detectors is > 1.0.
APC may be something you want to consider using internally. It does require that you place each polyp into an individual jar, which can increase incremental cost. Nonetheless, there is clear evidence that APC positively changes outcomes.
Including Sessile Serrated Lesions in ADR Detectors
Unfortunately, some of the high-level ADR detectors aren’t so “high level” when it comes to detecting sessile serrated lesions. It’s not quite as concordant as we had previously thought.
Nonetheless, there are many adjunctive things you can do with sessile serrated lesions, including narrow-band imaging and chromoendoscopy.
When it comes to establishing a discriminant, the numbers should be 5%-6% if we’re going to set a quality ratio and an index. However, this is somewhat dependent on your pathologist because they have to read these correctly. Lesions that are ≥ 6 mm above the sigmoid colon and anything in the right colon should be evaluated really closely as a sessile serrated lesion.
I’ve had indications where the pathologist says the lesion is hyperplastic, to which I say, “I’m going to follow as a sessile serrated lesion.” This is because it’s apparent to me in the endoscopic appearance and the narrow-band imaging appearance that it was characteristic of sessile serrated lesions.
Best Practices in Bowel Preparations
The US Multi-Society Task Force recommends that adequate bowel preparation should occur in 85% or more of outpatients, and for the European Society for Gastrointestinal Endoscopy, it’s 90% or more.
I’ll pass along a tip I use in my patients undergoing bowel preparation: I make them aware that during this process, they want to see a clear, yellow, urine-like color to their stool. Otherwise, many patients will think if they’ve had some diarrhea, they don’t need to finish prep. Setting that expectation for them upfront is really important.
The nurses also should be aware of this because if there’s a murky brown effluent upon presentation for the colonoscopy, there’s a greater than 50% chance that they’re going to have had an inadequate preparation. In such cases, you would want to preempt the colonoscopy and perhaps send them out for a re-prep that day or bring them back for a rescheduled appointment.
Resection Considerations
There is substantial variation when it comes to lesion resection, which makes it an important quality indicator on which to focus: High-level detectors aren’t always high-level resectors.
There are two validated instruments that you can use to gauge the adequacy of resection. Those aren’t really ready for prime time in every practice, though they may be seen in fellowship programs.
The idea here is that you want to get a ≥ 2 mm margin for cold snare polypectomy in lesions 1-10 mm in size. This has been a challenge, as findings indicate we don’t do this that well.
Joseph Anderson and colleagues recently published a study using a 2-mm resection margin. They reported that this was only possible in approximately 28% of polyps. For a 1-mm margin, the rate was 84%.
We simply need to set clearer margins when setting our snare. Make sure you’re close enough to the polyp, push down on the snare, and get a good margin of tissue.
When the sample contracts are placed into the formalin, it’s not quite so simple to define the margin at the time of the surgical resection. This often requires an audit evaluation by the pathologist.
There are two other considerations regarding resection.
The first is about the referral for surgery. Referral should not occur for any benign lesions ascribed by your endoscopic advanced imaging techniques and classifications that are not thought to have intramucosal carcinoma. These should be referred to an expert endoscopic evaluation. If you can’t do it, then somebody else should. And you shouldn’t attempt it unless you can get it totally because resection of partially resected lesions is much more complicated. The European Society of Gastrointestinal Endoscopy says this applies to any benign lesion of any size, which I think really is the emerging standard of care. You should consider and offer that to the patient. It may require a referral for outside of your institution.
The second additional consideration is around the minimization of cold forceps for removal of polyps. The US Multi-Society Task Force says cold forceps shouldn’t be used for any lesions > 2 mm, whereas for the European Society of Gastrointestinal Endoscopy, it is > 3 mm. However, it’s still done very commonly in clinical practice. Nibbling the polyp is not an option. Cold snare is actually quicker, more effective, has better outcomes, and is something that you can bill for when you look at the coding.
In summary, there are a lot of things that we can do now to improve colonoscopy. Quality indicators continue to emerge with a compelling, excellent evidence base that strongly supports their use. Given that, I think most of these are actionable now, and it’s not necessary to wait for the guidelines to begin using them. I’d therefore challenge all of us to incorporate them in our continual efforts to do better.
Dr. Johnson is professor of medicine and chief of gastroenterology at Eastern Virginia Medical School in Norfolk, and a past president of the American College of Gastroenterology. His primary focus is the clinical practice of gastroenterology. He has published extensively in the internal medicine/gastroenterology literature, with principal research interests in esophageal and colon disease, and more recently in sleep and microbiome effects on gastrointestinal health and disease. He has disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
When it comes to the use of colonoscopy to reduce the risk for cancer, quality is key.
There are a number of performance improvements we can make in our practices so that we can do better. This is evident in several recently published studies and a recent review article on the topic, which I’d like to profile for you; many of these key quality indicators you can implement now.
Even though it may take more time before they’re supported in the guidelines, you’ll see that the evidence behind these is extraordinarily strong.
Increasing the Adenoma Detection Rate
Certainly, we all do what we can to increase the adenoma detection rate (ADR).
However, at the moment, the nationally recommended benchmark is to achieve an ADR of 25%, which is inordinately low. The ADR rate reported in the GIQuIC registry data is closer to 39%, and in high-level detectors, it’s actually in the greater-than-50% range.
There’s no question that we can do more, and there are a number of ways to do that.
First, This may actually decrease your withdrawal time because you don’t spend so much time trying to face these folds.
In considering tools to aid ADR, don’t forget electronic chromoendoscopy (eg, narrow-band imaging).
There are a number of new artificial intelligence options out there as well, which have been reported to increase the ADR by approximately 10%. Of importance, this improvement even occurs among expert endoscopists.
There’s also important emerging data about ADR in fecal immunochemical test (FIT)–positive patients. FIT-positive status increases the ADR threshold by 15%-20%. This places you in an ADR range of approximately 50%, which is really the norm when screening patients that present for colonoscopy because of FIT positivity.
Adenoma Per Colonoscopy: A Possible ADR Substitute
Growing evidence supports the use of adenoma per colonoscopy (APC) as a substitute to ADR. This would allow you to record every adenoma and attribute it to that index colonoscopy.
A high-quality paper showed that the APC value should be around 0.6 to achieve the current ADR minimum threshold of 25%. Having the APC < 0.6 seems to be associated with an increased risk for residual polyp. Sessile serrated lesions also increased the hazard ratio for interval colorectal cancer. This was evaluated recently with data from the New Hampshire Colonoscopy Registry, which Dr Joseph Anderson has led for so long. They showed that 21% of endoscopists had an ADR of 25% or greater but still had APCs < 0.6.
Therefore, when it comes to remedial corrective work, doctors need to be reevaluated, retrained, and educated in the ways that they can incorporate this. The APC in high-level detectors is > 1.0.
APC may be something you want to consider using internally. It does require that you place each polyp into an individual jar, which can increase incremental cost. Nonetheless, there is clear evidence that APC positively changes outcomes.
Including Sessile Serrated Lesions in ADR Detectors
Unfortunately, some of the high-level ADR detectors aren’t so “high level” when it comes to detecting sessile serrated lesions. It’s not quite as concordant as we had previously thought.
Nonetheless, there are many adjunctive things you can do with sessile serrated lesions, including narrow-band imaging and chromoendoscopy.
When it comes to establishing a discriminant, the numbers should be 5%-6% if we’re going to set a quality ratio and an index. However, this is somewhat dependent on your pathologist because they have to read these correctly. Lesions that are ≥ 6 mm above the sigmoid colon and anything in the right colon should be evaluated really closely as a sessile serrated lesion.
I’ve had indications where the pathologist says the lesion is hyperplastic, to which I say, “I’m going to follow as a sessile serrated lesion.” This is because it’s apparent to me in the endoscopic appearance and the narrow-band imaging appearance that it was characteristic of sessile serrated lesions.
Best Practices in Bowel Preparations
The US Multi-Society Task Force recommends that adequate bowel preparation should occur in 85% or more of outpatients, and for the European Society for Gastrointestinal Endoscopy, it’s 90% or more.
I’ll pass along a tip I use in my patients undergoing bowel preparation: I make them aware that during this process, they want to see a clear, yellow, urine-like color to their stool. Otherwise, many patients will think if they’ve had some diarrhea, they don’t need to finish prep. Setting that expectation for them upfront is really important.
The nurses also should be aware of this because if there’s a murky brown effluent upon presentation for the colonoscopy, there’s a greater than 50% chance that they’re going to have had an inadequate preparation. In such cases, you would want to preempt the colonoscopy and perhaps send them out for a re-prep that day or bring them back for a rescheduled appointment.
Resection Considerations
There is substantial variation when it comes to lesion resection, which makes it an important quality indicator on which to focus: High-level detectors aren’t always high-level resectors.
There are two validated instruments that you can use to gauge the adequacy of resection. Those aren’t really ready for prime time in every practice, though they may be seen in fellowship programs.
The idea here is that you want to get a ≥ 2 mm margin for cold snare polypectomy in lesions 1-10 mm in size. This has been a challenge, as findings indicate we don’t do this that well.
Joseph Anderson and colleagues recently published a study using a 2-mm resection margin. They reported that this was only possible in approximately 28% of polyps. For a 1-mm margin, the rate was 84%.
We simply need to set clearer margins when setting our snare. Make sure you’re close enough to the polyp, push down on the snare, and get a good margin of tissue.
When the sample contracts are placed into the formalin, it’s not quite so simple to define the margin at the time of the surgical resection. This often requires an audit evaluation by the pathologist.
There are two other considerations regarding resection.
The first is about the referral for surgery. Referral should not occur for any benign lesions ascribed by your endoscopic advanced imaging techniques and classifications that are not thought to have intramucosal carcinoma. These should be referred to an expert endoscopic evaluation. If you can’t do it, then somebody else should. And you shouldn’t attempt it unless you can get it totally because resection of partially resected lesions is much more complicated. The European Society of Gastrointestinal Endoscopy says this applies to any benign lesion of any size, which I think really is the emerging standard of care. You should consider and offer that to the patient. It may require a referral for outside of your institution.
The second additional consideration is around the minimization of cold forceps for removal of polyps. The US Multi-Society Task Force says cold forceps shouldn’t be used for any lesions > 2 mm, whereas for the European Society of Gastrointestinal Endoscopy, it is > 3 mm. However, it’s still done very commonly in clinical practice. Nibbling the polyp is not an option. Cold snare is actually quicker, more effective, has better outcomes, and is something that you can bill for when you look at the coding.
In summary, there are a lot of things that we can do now to improve colonoscopy. Quality indicators continue to emerge with a compelling, excellent evidence base that strongly supports their use. Given that, I think most of these are actionable now, and it’s not necessary to wait for the guidelines to begin using them. I’d therefore challenge all of us to incorporate them in our continual efforts to do better.
Dr. Johnson is professor of medicine and chief of gastroenterology at Eastern Virginia Medical School in Norfolk, and a past president of the American College of Gastroenterology. His primary focus is the clinical practice of gastroenterology. He has published extensively in the internal medicine/gastroenterology literature, with principal research interests in esophageal and colon disease, and more recently in sleep and microbiome effects on gastrointestinal health and disease. He has disclosed ties with ISOTHRIVE and Johnson & Johnson.
A version of this article appeared on Medscape.com.
Superficial Vascular Anomaly of the Glabella Mimicking a Cutaneous Cyst
To the Editor:
Cutaneous cysts commonly are treated by dermatologists and typically are diagnosed clinically, followed by intraoperative or histologic confirmation; however, cyst mimickers can be misdiagnosed due to similar appearance and limited diagnostic guidelines.1 Vascular anomalies (VAs) of the face such as a facial aneurysm are rare.2 Preoperative assessment of findings suggestive of vascular etiology vs other common cutaneous tumors such as an epidermal inclusion cyst (EIC) and lipoma can help guide dermatologic management. We present a case of a VA of the glabella manifesting as a flesh-colored nodule that clinically mimicked a cyst and discuss the subsequent surgical management.
A 61-year-old man with a history of benign prostatic hyperplasia was evaluated at our dermatology clinic for an enlarging forehead mass of 1 year’s duration. Physical examination yielded a soft, flesh-colored, 2.5-cm nodule located superficially in the midline glabellar region without pulsation or palpable thrill. The differential diagnosis at the time included lipoma or EIC.
Excision of the lesion was performed utilizing superficial incisions with a descending depth of 1-mm increments to safely reach the target, identify the type of tumor, and prevent rupture of the suspected EIC. After the third incision to the level of the dermis, nonpulsatile bleeding was more than expected for a cyst. Digital pressure was applied, and the area was explored with blunt dissection to identify the source of bleeding. A fusiform, thin-walled aneurysm was identified in the dermal plane with additional tributaries coursing deep into the subcutaneous plane. The visualized tributaries were ligated with 3-0 polyglactin, figure-of-eight sutures resulting in hemostasis. The wound was closed with 5-0 nylon simple interrupted sutures. The patient was closely followed postoperatively for 1 week (Figure) and was referred for head imaging to evaluate for a possible associated intracranial aneurysm. Based on the thin vessel wall and continuous nonpulsatile hemorrhage, this VA was most consistent with venous aneurysm.
A VA can be encountered unexpectedly during dermatologic surgery. An aneurysm is a type of VA and is defined as an abnormal dilatation of a blood vessel that can be arterial, venous, or an arteriovenous malformation. Most reported aneurysms of the head and neck are cirsoid aneurysms or involve the superficial temporal artery.2,3 Reports of superficial venous aneurysms are rare.4 Preoperatively, cutaneous nodules can be evaluated for findings suggestive of a VA in the dermatologist’s office through physical examination. Arterial aneurysms may reveal palpable pulsation and audible bruit, while a venous aneurysm may exhibit a blue color, a size reduction with compression, and variable size with Valsalva maneuver.
The gold standard diagnostic tool for most dermatologic conditions is histopathology; however, dermatologic ultrasonography can provide noninvasive, real-time, important diagnostic characteristics of cutaneous pathologies as well as VA.5-7 Crisan et al6 outlined specific sonographic findings of lipomas, EICs, trichilemmal cysts, and other dermatologic conditions as well as the associated surgical pertinence. Ultrasonography of a venous aneurysm may show a heterogeneous, contiguous, echoic lesion with an adjacent superficial vein, which may be easily compressed by the probe.8 Advanced imaging such as computed tomography with contrast or magnetic resonance imaging may be performed, but these are more costly than ultrasonography. Additionally, point-of-care ultrasonography is becoming more popular and accessible for physicians to carry at bedside with portable tablet options available. Dermatologists may want to consider incorporating it into the outpatient setting to improve procedural planning.9
In conclusion, VAs should be included in the differential diagnosis of soft cutaneous nodules, as management differs from a cyst or lipoma. Dermatologists should use their clinical judgment preoperatively—including a comprehensive history, physical examination, and consideration of color Doppler ultrasonography to assess for findings of VA. We do not recommend intentional surgical exploration of cutaneous aneurysms in the ambulatory setting due to risk for hemorrhage. Furthermore, when clinical suspicion of EIC or lipoma is high, it still is preferable to descend the incision slowly at 1 to 2 mm per cut until the tumor is visualized.
- Ring CM, Kornreich DA, Lee JB. Clinical simulators of cysts. J Am Acad Dermatol. 2016;75:1255-1257.
- Evans CC, Larson MJ, Eichhorn PJ, et al. Traumatic pseudoaneurysm of the superficial temporal artery: two cases and review of the literature. J Am Acad Dermatol. 2003;49(5 suppl):S286-S288.
- Sofela A, Osunronbi T, Hettige S. Scalp cirsoid aneurysms: case illustration and systematic review of literature. Neurosurgery. 2020;86:E98-E107.
- McKesey J, Cohen PR. Spontaneous venous aneurysm: report of a non-traumatic superficial venous aneurysm on the distal arm. Cureus. 2018;10:E2641.
- Wortsman X, Alfageme F, Roustan G, et al. Guidelines for performing dermatologic ultrasound examinations by the DERMUS Group. J Ultrasound Med. 2016;35:577-580.
- Crisan D, Wortsman X, Alfageme F, et al. Ultrasonography in dermatologic surgery: revealing the unseen for improved surgical planning [published online May 26, 2022]. J Dtsch Dermatol Ges. doi:10.1111/ddg.14781
- Corvino A, Catalano O, Corvino F, et al. Superficial temporal artery pseudoaneurysm: what is the role of ultrasound. J Ultrasound. 2016;19:197-201.
- Lee HY, Lee W, Cho YK, et al. Superficial venous aneurysm: reports of 3 cases and literature review. J Ultrasound Med. 2006;25:771-776.
- Hadian Y, Link D, Dahle SE, et al. Ultrasound as a diagnostic and interventional aid at point-of-care in dermatology clinic: a case report. J Dermatolog Treat. 2020;31:74-76.
To the Editor:
Cutaneous cysts commonly are treated by dermatologists and typically are diagnosed clinically, followed by intraoperative or histologic confirmation; however, cyst mimickers can be misdiagnosed due to similar appearance and limited diagnostic guidelines.1 Vascular anomalies (VAs) of the face such as a facial aneurysm are rare.2 Preoperative assessment of findings suggestive of vascular etiology vs other common cutaneous tumors such as an epidermal inclusion cyst (EIC) and lipoma can help guide dermatologic management. We present a case of a VA of the glabella manifesting as a flesh-colored nodule that clinically mimicked a cyst and discuss the subsequent surgical management.
A 61-year-old man with a history of benign prostatic hyperplasia was evaluated at our dermatology clinic for an enlarging forehead mass of 1 year’s duration. Physical examination yielded a soft, flesh-colored, 2.5-cm nodule located superficially in the midline glabellar region without pulsation or palpable thrill. The differential diagnosis at the time included lipoma or EIC.
Excision of the lesion was performed utilizing superficial incisions with a descending depth of 1-mm increments to safely reach the target, identify the type of tumor, and prevent rupture of the suspected EIC. After the third incision to the level of the dermis, nonpulsatile bleeding was more than expected for a cyst. Digital pressure was applied, and the area was explored with blunt dissection to identify the source of bleeding. A fusiform, thin-walled aneurysm was identified in the dermal plane with additional tributaries coursing deep into the subcutaneous plane. The visualized tributaries were ligated with 3-0 polyglactin, figure-of-eight sutures resulting in hemostasis. The wound was closed with 5-0 nylon simple interrupted sutures. The patient was closely followed postoperatively for 1 week (Figure) and was referred for head imaging to evaluate for a possible associated intracranial aneurysm. Based on the thin vessel wall and continuous nonpulsatile hemorrhage, this VA was most consistent with venous aneurysm.

A VA can be encountered unexpectedly during dermatologic surgery. An aneurysm is a type of VA and is defined as an abnormal dilatation of a blood vessel that can be arterial, venous, or an arteriovenous malformation. Most reported aneurysms of the head and neck are cirsoid aneurysms or involve the superficial temporal artery.2,3 Reports of superficial venous aneurysms are rare.4 Preoperatively, cutaneous nodules can be evaluated for findings suggestive of a VA in the dermatologist’s office through physical examination. Arterial aneurysms may reveal palpable pulsation and audible bruit, while a venous aneurysm may exhibit a blue color, a size reduction with compression, and variable size with Valsalva maneuver.
The gold standard diagnostic tool for most dermatologic conditions is histopathology; however, dermatologic ultrasonography can provide noninvasive, real-time, important diagnostic characteristics of cutaneous pathologies as well as VA.5-7 Crisan et al6 outlined specific sonographic findings of lipomas, EICs, trichilemmal cysts, and other dermatologic conditions as well as the associated surgical pertinence. Ultrasonography of a venous aneurysm may show a heterogeneous, contiguous, echoic lesion with an adjacent superficial vein, which may be easily compressed by the probe.8 Advanced imaging such as computed tomography with contrast or magnetic resonance imaging may be performed, but these are more costly than ultrasonography. Additionally, point-of-care ultrasonography is becoming more popular and accessible for physicians to carry at bedside with portable tablet options available. Dermatologists may want to consider incorporating it into the outpatient setting to improve procedural planning.9
In conclusion, VAs should be included in the differential diagnosis of soft cutaneous nodules, as management differs from a cyst or lipoma. Dermatologists should use their clinical judgment preoperatively—including a comprehensive history, physical examination, and consideration of color Doppler ultrasonography to assess for findings of VA. We do not recommend intentional surgical exploration of cutaneous aneurysms in the ambulatory setting due to risk for hemorrhage. Furthermore, when clinical suspicion of EIC or lipoma is high, it still is preferable to descend the incision slowly at 1 to 2 mm per cut until the tumor is visualized.
To the Editor:
Cutaneous cysts commonly are treated by dermatologists and typically are diagnosed clinically, followed by intraoperative or histologic confirmation; however, cyst mimickers can be misdiagnosed due to similar appearance and limited diagnostic guidelines.1 Vascular anomalies (VAs) of the face such as a facial aneurysm are rare.2 Preoperative assessment of findings suggestive of vascular etiology vs other common cutaneous tumors such as an epidermal inclusion cyst (EIC) and lipoma can help guide dermatologic management. We present a case of a VA of the glabella manifesting as a flesh-colored nodule that clinically mimicked a cyst and discuss the subsequent surgical management.
A 61-year-old man with a history of benign prostatic hyperplasia was evaluated at our dermatology clinic for an enlarging forehead mass of 1 year’s duration. Physical examination yielded a soft, flesh-colored, 2.5-cm nodule located superficially in the midline glabellar region without pulsation or palpable thrill. The differential diagnosis at the time included lipoma or EIC.
Excision of the lesion was performed utilizing superficial incisions with a descending depth of 1-mm increments to safely reach the target, identify the type of tumor, and prevent rupture of the suspected EIC. After the third incision to the level of the dermis, nonpulsatile bleeding was more than expected for a cyst. Digital pressure was applied, and the area was explored with blunt dissection to identify the source of bleeding. A fusiform, thin-walled aneurysm was identified in the dermal plane with additional tributaries coursing deep into the subcutaneous plane. The visualized tributaries were ligated with 3-0 polyglactin, figure-of-eight sutures resulting in hemostasis. The wound was closed with 5-0 nylon simple interrupted sutures. The patient was closely followed postoperatively for 1 week (Figure) and was referred for head imaging to evaluate for a possible associated intracranial aneurysm. Based on the thin vessel wall and continuous nonpulsatile hemorrhage, this VA was most consistent with venous aneurysm.

A VA can be encountered unexpectedly during dermatologic surgery. An aneurysm is a type of VA and is defined as an abnormal dilatation of a blood vessel that can be arterial, venous, or an arteriovenous malformation. Most reported aneurysms of the head and neck are cirsoid aneurysms or involve the superficial temporal artery.2,3 Reports of superficial venous aneurysms are rare.4 Preoperatively, cutaneous nodules can be evaluated for findings suggestive of a VA in the dermatologist’s office through physical examination. Arterial aneurysms may reveal palpable pulsation and audible bruit, while a venous aneurysm may exhibit a blue color, a size reduction with compression, and variable size with Valsalva maneuver.
The gold standard diagnostic tool for most dermatologic conditions is histopathology; however, dermatologic ultrasonography can provide noninvasive, real-time, important diagnostic characteristics of cutaneous pathologies as well as VA.5-7 Crisan et al6 outlined specific sonographic findings of lipomas, EICs, trichilemmal cysts, and other dermatologic conditions as well as the associated surgical pertinence. Ultrasonography of a venous aneurysm may show a heterogeneous, contiguous, echoic lesion with an adjacent superficial vein, which may be easily compressed by the probe.8 Advanced imaging such as computed tomography with contrast or magnetic resonance imaging may be performed, but these are more costly than ultrasonography. Additionally, point-of-care ultrasonography is becoming more popular and accessible for physicians to carry at bedside with portable tablet options available. Dermatologists may want to consider incorporating it into the outpatient setting to improve procedural planning.9
In conclusion, VAs should be included in the differential diagnosis of soft cutaneous nodules, as management differs from a cyst or lipoma. Dermatologists should use their clinical judgment preoperatively—including a comprehensive history, physical examination, and consideration of color Doppler ultrasonography to assess for findings of VA. We do not recommend intentional surgical exploration of cutaneous aneurysms in the ambulatory setting due to risk for hemorrhage. Furthermore, when clinical suspicion of EIC or lipoma is high, it still is preferable to descend the incision slowly at 1 to 2 mm per cut until the tumor is visualized.
- Ring CM, Kornreich DA, Lee JB. Clinical simulators of cysts. J Am Acad Dermatol. 2016;75:1255-1257.
- Evans CC, Larson MJ, Eichhorn PJ, et al. Traumatic pseudoaneurysm of the superficial temporal artery: two cases and review of the literature. J Am Acad Dermatol. 2003;49(5 suppl):S286-S288.
- Sofela A, Osunronbi T, Hettige S. Scalp cirsoid aneurysms: case illustration and systematic review of literature. Neurosurgery. 2020;86:E98-E107.
- McKesey J, Cohen PR. Spontaneous venous aneurysm: report of a non-traumatic superficial venous aneurysm on the distal arm. Cureus. 2018;10:E2641.
- Wortsman X, Alfageme F, Roustan G, et al. Guidelines for performing dermatologic ultrasound examinations by the DERMUS Group. J Ultrasound Med. 2016;35:577-580.
- Crisan D, Wortsman X, Alfageme F, et al. Ultrasonography in dermatologic surgery: revealing the unseen for improved surgical planning [published online May 26, 2022]. J Dtsch Dermatol Ges. doi:10.1111/ddg.14781
- Corvino A, Catalano O, Corvino F, et al. Superficial temporal artery pseudoaneurysm: what is the role of ultrasound. J Ultrasound. 2016;19:197-201.
- Lee HY, Lee W, Cho YK, et al. Superficial venous aneurysm: reports of 3 cases and literature review. J Ultrasound Med. 2006;25:771-776.
- Hadian Y, Link D, Dahle SE, et al. Ultrasound as a diagnostic and interventional aid at point-of-care in dermatology clinic: a case report. J Dermatolog Treat. 2020;31:74-76.
- Ring CM, Kornreich DA, Lee JB. Clinical simulators of cysts. J Am Acad Dermatol. 2016;75:1255-1257.
- Evans CC, Larson MJ, Eichhorn PJ, et al. Traumatic pseudoaneurysm of the superficial temporal artery: two cases and review of the literature. J Am Acad Dermatol. 2003;49(5 suppl):S286-S288.
- Sofela A, Osunronbi T, Hettige S. Scalp cirsoid aneurysms: case illustration and systematic review of literature. Neurosurgery. 2020;86:E98-E107.
- McKesey J, Cohen PR. Spontaneous venous aneurysm: report of a non-traumatic superficial venous aneurysm on the distal arm. Cureus. 2018;10:E2641.
- Wortsman X, Alfageme F, Roustan G, et al. Guidelines for performing dermatologic ultrasound examinations by the DERMUS Group. J Ultrasound Med. 2016;35:577-580.
- Crisan D, Wortsman X, Alfageme F, et al. Ultrasonography in dermatologic surgery: revealing the unseen for improved surgical planning [published online May 26, 2022]. J Dtsch Dermatol Ges. doi:10.1111/ddg.14781
- Corvino A, Catalano O, Corvino F, et al. Superficial temporal artery pseudoaneurysm: what is the role of ultrasound. J Ultrasound. 2016;19:197-201.
- Lee HY, Lee W, Cho YK, et al. Superficial venous aneurysm: reports of 3 cases and literature review. J Ultrasound Med. 2006;25:771-776.
- Hadian Y, Link D, Dahle SE, et al. Ultrasound as a diagnostic and interventional aid at point-of-care in dermatology clinic: a case report. J Dermatolog Treat. 2020;31:74-76.
Practice Points
- Vascular anomalies should be included in the differential diagnosis of soft cutaneous nodules, as management differs from cysts or lipomas.
- Preoperative evaluation for a cutaneous cyst excision on the head and neck should include ruling out findings of a vascular lesion through history, physical examination, and consideration of color Doppler ultrasonography in unclear cases.
- Surgical technique should involve sequential superficial incisions, descending at 1 to 2 mm per cut, until the suspected capsule is identified to minimize the risk for inadvertent injury to a cyst mimicker such as a vascular anomaly.