User login
Do Health-Related Social Needs Raise Mortality Risk in Cancer Survivors?
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
Little is known about the specific association between health-related social needs (HRSNs) and mortality risk even though HRSNs, defined as challenges in affording food, housing, and other necessities of daily living, are potential challenges for cancer survivors, wrote Zhiyuan Zheng, PhD, of the American Cancer Society, Atlanta, and colleagues.
A 2020 study by Dr. Zheng and colleagues published in the Journal of the National Comprehensive Cancer Network (NCCN) showed that food insecurity and financial worries had a negative impact on cancer survivorship. In the new study, published in Cancer, the researchers identified cancer survivors using the 2013-2018 National Health Interview Survey (NHIS) and the NHIS Mortality File through December 31, 2019. The researchers examined mortality using the data from the Centers for Disease Control and Prevention’s National Death Index (NDI) through December 31, 2019, which links to the National Health Interview Survey Data used in the study.
Individuals’ HRSNs were categorized into three groups: severe, moderate, and minor/none. HRSNs included food insecurity and nonmedical financial concerns, such as housing costs (rent, mortgage). Medical financial hardship included material, psychological, and behavioral domains and was divided into three groups: 2-3 domains, 1 domain, or 0 domains.
What Are the Potential Financial Implications of this Research?
The high costs of cancer care often cause medical financial hardships for cancer survivors, and expenses also may cause psychological distress and nonmedical financial hardship as survivors try to make ends meet while facing medical bills, wrote Dr. Zheng and colleagues.
Policy makers are increasingly interested in adding HRSNs to insurance coverage; recent guidance from the Centers for Medicare & Medicaid Services (CMS) allows individual states to apply to provide nutrition and housing supports through state Medicaid programs, according to authors of a 2023 article published in JAMA Health Forum.
The new study adds to the understanding of how HRSNs impact people with cancer by examining the association with mortality risk, Yelak Biru, MSc, president and chief executive officer of the International Myeloma Foundation, said in an interview.
“This is a key area of study for addressing the disparities in treatments and outcomes that result in inequities,” said Mr. Biru, a patient advocate and multiple myeloma survivor who was not involved in the study.
What Does the New Study Show?
The new study characterized HRSNs in 5,855 adult cancer survivors aged 18-64 years and 5,918 aged 65-79 years. In the 18- to 64-year-old group, 25.5% reported moderate levels of HRSNs, and 18.3% reported severe HRSNs. In patients aged 65-79 years, 15.6% and 6.6% reported moderate HRSNs and severe HRSNs, respectively.
Severe HRSN was significantly associated with higher mortality risk in an adjusted analysis in patients aged 18-64 years (hazard ratio 2.00, P < .001).
Among adults aged 65-79 years, severe HRSN was not associated with higher mortality risk; however, in this older age group, those with 2-3 domains of medical financial hardship had significantly increased mortality risk compared with adults aged 65-79 years with zero domains of medical financial hardship (HR 1.58, P = .007).
Although the findings that HRSNs were associated with increased mortality risk, especially in the younger group, were not surprising, they serve as a call to action to address how HRSNs are contributing to cancer mortality, Mr. Biru said in an interview. “HRSNs, like food or housing insecurity, can lead to patients being unable to undergo the best treatment approach for their cancer,” he said.
What Are the Limitations and Research Gaps?
The study findings were limited by several factors including the use of self-reports to measure medical financial hardship, food insecurity, and nonmedical financial concerns in the NHIS, the researchers wrote in their discussion. More research with longer follow-up time beyond 1-5 years is needed, wrote Dr. Zheng and colleagues.
Studies also are needed to illustrate how patient navigation can help prevent patients from falling through the cracks with regard to social needs and financial hardships, Mr. Biru told this news organization.
Other areas for research include how addressing social needs affects health outcomes and whether programs designed to address social needs are effective, he said.
“Finally, qualitative research is needed to capture the lived experiences of cancer survivors facing these challenges. This knowledge can inform the development of more patient-centered interventions and policies that effectively address the social determinants of health and improve overall outcomes for all cancer survivors,” Mr. Biru said.
What Is the Takeaway Message for Clinicians?
HRSNs and financial hardship are significantly associated with increased risk of mortality in adult cancer survivors, Dr. Zheng and colleagues concluded. Looking ahead, comprehensive assessment of HRSNs and financial hardship may help clinicians connect patients with relevant services to mitigate the social and financial impacts of cancer, they wrote.
“The takeaway message for oncologists in practice is that addressing [HRSNs] and financial hardship is crucial for providing comprehensive and equitable cancer care,” Mr. Biru said during his interview.
“The impact of social determinants of health on cancer outcomes cannot be ignored, and oncologists play a vital role in identifying and addressing these needs,” he said. Sensitive, discussion-based screenings are needed to identify core needs such as food and transportation, but clinicians also can consider broader social factors and work with a team to connect patients to appropriate resources, he added.
“By recognizing the importance of HRSN screening and taking proactive steps to address these needs, oncologists can contribute to improving health outcomes, reducing healthcare disparities, and providing more equitable cancer care for their patients,” he said.
What Other Guidance Is Available?
“High-quality cancer care requires treating the whole person; measuring and addressing anything in their life that could result in poorer health outcomes is a key component of comprehensive care,” Mr. Biru emphasized.
In September 2023, the National Comprehensive Cancer Network (NCCN) convened a working group cochaired by Mr. Biru that developed recommendations for how oncology practices should routinely measure HRSNs (NCCN.org/social-needs).
“The working group proposed that every cancer patient be assessed for food, transportation access, and financial and housing security at least once a year, and be reassessed at every care transition point as well,” Mr. Biru said. Such screenings should include follow-up to connect patients with services to address any HRSNs they are experiencing, he added.
Lead author Dr. Zheng is employed by the American Cancer Society, which as a nonprofit receives funds from the public through fundraising and contributions, as well as some support from corporations and industry to support its mission programs and services. Mr. Biru had no financial conflicts to disclose.
FROM CANCER
Terminal Cancer: What Matters to Patients and Caregivers
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
New research found that patients and caregivers both tend to prioritize symptom control over life extension but often preferring a balance. Patients and caregivers, however, are less aligned on decisions about cost containment, with patients more likely to prioritize cost containment.
“Our research has revealed that patients and caregivers generally share similar end-of-life goals,” with a “notable exception” when it comes to costs, first author Semra Ozdemir, PhD, with the Lien Centre for Palliative Care, Duke-NUS Medical School, Singapore, told this news organization.
However, when patients and caregivers have a better understanding of the patient’s prognosis, both may be more inclined to avoid costly life-extending treatments and prioritize symptom management.
In other words, the survey suggests that “knowing the prognosis helps patients and their families set realistic expectations for care and adequately prepare for end-of-life decisions,” said Dr. Ozdemir.
This study was published online in JAMA Network Open.
Patients with advanced cancer often face difficult decisions: Do they opt for treatments that may — or may not — extend life or do they focus more on symptom control?
Family caregivers, who also play an important role in this decision-making process, may have different care goals. Some research suggests that caregivers tend to prioritize treatments that could extend life, whereas patients prioritize symptom management, but it’s less clear how these priorities may change over time and how patients and caregivers may influence each other.
In the current study, the researchers examined goals of care among patients with stage IV solid tumors and caregivers during the last 2 years of life, focusing on life extension vs symptom management and cost containment, as well as how these goals changed over time.
The survey included 210 patient-caregiver pairs, recruited from outpatient clinics at two major cancer centers in Singapore. Patients had a mean age of 63 years, and about half were men. The caregivers had a mean age of 49 years, and almost two third (63%) were women.
Overall, 34% patients and 29% caregivers prioritized symptom management over life extension, whereas 24% patients and 19% caregivers prioritized life extension. Most patients and caregivers preferred balancing the two, with 34%-47% patients and 37%-69% caregivers supporting this approach.
When balancing cost and treatment decisions, however, patients were more likely to prioritize containing costs — 28% vs 17% for caregivers — over extending life — 26% of patients vs 35% of caregivers.
Cost containment tended to be more of a priority for older patients, those with a higher symptom burden, and those with less family caregiver support. For caregivers, cost containment was more of a priority for those who reported that caregiving had a big impact on their finances, those with worse self-esteem related to their caregiving abilities, as well as those caring for older patients.
To better align cost containment priorities between patients and caregivers, it’s essential for families to engage in open and thorough discussions about the allocation of resources, Dr. Ozdemir said.
Although “patients, families, and physicians often avoid discussions about prognosis,” such conversations are essential for setting realistic expectations for care and adequately preparing for end-of-life decisions, Dr. Ozdemir told this news organization.
“These conversations should aim to balance competing interests and create care plans that are mutually acceptable to both patients and caregivers,” she said, adding that “this approach will help in minimizing any potential conflicts and ensure that both parties feel respected and understood in their decision-making process.”
Managing Unrealistic Expectations
As patients approached the end of life, neither patients nor caregivers shifted their priorities from life extension to symptom management.
This finding raises concerns because it suggests that many patients hold unrealistic expectations regarding their care and “underscores the need for continuous dialogue and reassessment of care goals throughout the progression of illness,” Dr. Ozdemir said.
“This stability in preferences over time suggests that initial care decisions are deeply ingrained or that there may be a lack of ongoing communication about evolving care needs and possibilities as conditions change,” Ozdemir said.
Yet, it can be hard to define what unrealistic expectations mean, said Olivia Seecof, MD, who wasn’t involved in the study.
“I think people are hopeful that a devastating diagnosis won’t lead to the end of their life and that there will be a treatment or something that will change [their prognosis], and they’ll get better,” said Dr. Seecof, palliative care expert with the Supportive Oncology Program at NYU Langone Health’s Perlmutter Cancer Center in New York City.
Giving patients and caregivers a realistic understanding of the prognosis is important, but “there’s more to it than just telling the patient their diagnosis,” she said.
“We have to plan for end of life, what it can look like,” said Dr. Seecof, adding that “often we don’t do a very good job of talking about that early on in an illness course.”
Overall, though, Dr. Seecof stressed that no two patients or situations are the same, and it’s important to understand what’s important in each scenario. End-of-life care requires “an individual approach because every patient is different, even if they have the same diagnosis as someone else,” she said.
This work was supported by funding from the Singapore Millennium Foundation and the Lien Centre for Palliative Care. Dr. Ozdemir and Dr. Seecof had no relevant disclosures.
A version of this article appeared on Medscape.com.
Global Quest to Cut CAR T Costs
In the United States, a stand-alone device could greatly reduce the expense of producing modified immune cells. In India, researchers hope homegrown technology is the key to getting costs under control. In Latin America, a partnership between the Brazilian government and a US nonprofit may be just the ticket.
At stake is expanded access to CAR T-cell therapy, a form of immunotherapy that in just the past few years has revolutionized the care of hematologic cancers.
“Among patients with lymphoma, leukemia, and myeloma, anywhere between 30% to 50% reach long-term remission after one CAR T-cell infusion,” Mayo Clinic–Rochester hematologist/oncologist Saad J. Kenderian, MB, ChB, said in an interview. “It’s such an important therapy.”
However, only a small percentage of eligible patients in the United States — perhaps 20% or fewer — are receiving the treatment, he added.
A 2024 report suggested that many patients in the United States who may benefit aren’t being treated because of a range of possible reasons, including high prices, manufacturing logistics, and far distance from the limited number of institutions offering the therapy.
“Taken together, the real-world cost of CAR T-cell therapy can range from $700,000 to $1 million, which may make the treatment unaffordable to those patients without robust financial and/or social support,” the report authors noted.
Outside Western countries, access to the therapy is even more limited, because of its exorbitant price. The 2024 report noted that “there is a wide use of CAR T-cell therapy in Europe and China, but access is limited in developing countries in Southeast Asia, Africa, and Latin America.”
Harnessing the Power of T-Cells
Several types of CAR T-cell therapy have been approved by the US Food and Drug Administration (FDA) for patients with relapsed/refractory blood cancers such as follicular lymphoma, large B-cell lymphoma, multiple myeloma, and B-cell precursor acute lymphoblastic leukemia. A 2023 review analyzed clinical trials and reported that complete response rates were 40%-54% in aggressive B-cell lymphoma, 67% in mantle cell lymphoma, and 69%-74% in indolent B-cell lymphoma.
Pediatric hematologist/oncologist Kirsten Williams, MD, who specializes in pediatric blood and marrow transplant and cellular therapy at the Aflac Cancer and Blood Disorders Center of Children’s Healthcare of Atlanta, described CAR T-cell therapy as “a very unique form of immunotherapy” that harnesses the power of the immune system’s T-cells.
These cells are effective tumor killers, but they typically aren’t assigned to control cancer, she said in an interview. “We have very few of them, and most of our T cells are focused on killing various viruses,” she said. The therapy “allows us to take the T cell that would have killed the flu or mono and instead target leukemia, B-cell leukemia, or lymphoma.”
As she explained, “T cells are collected by a machine that reserves white blood cells and gives back the rest of the blood to the patient. We insert a gene into the T cells that encodes for a B-cell receptor. This receptor acts as a GPS signal, pulling T cells to the cancer so that they can kill it.”
In addition, “with this genetic change, we also add some things that allow the T cell to be stronger, to have a higher signal to kill the cancer cell once it locks on.”
The therapy is unique for each patient, Dr. Williams said. “We have collected and modified your specific T cells, and they can now only be infused into you. If we try to give your product to someone else, those cells would either cause harm by attacking the patient or would be immediately killed by that patient’s own immune system. This is very different than all the other kinds of therapies. When you take other medicines, it doesn’t matter who receives that pill.”
Treatment: Individual, Complex, and Costly
Why is CAR T-cell therapy so expensive? While only a single treatment is needed, the T cells have to go through an “individualized, bespoke manufacturing” process that’s “highly technical,” pediatric oncologist Stephan A. Grupp, MD, PhD, section chief of the Cellular Therapy and Transplant Section at Children’s Hospital of Philadelphia, said in an interview. As he explained, the cells for a single patient have to go through the same testing as with a drug that might be given to 1,000 people.
“The first thing we need to do is collect the cells from a patient,” said Dr. Williams. “For adults, that process involves putting in two big IVs — one in each arm — and then pulling the blood through a machine. This typically involves an 8-hour collection in the hospital and very highly specialized people to oversee the collection process.”
Secondly, at some institutions, “the cells get sent to a company where they undergo the process where the gene is inserted,” she said. “This process needs to be done in a very sterile environment so there’s no infections, and it needs to have a lot of oversight.”
Finally, “after the cells are generated, they are typically frozen and shipped back to the site where the patient is at the hospital,” she said. “Then we give chemotherapy to the patient, which prepares the patient’s blood system. It removes some of the T-cells that are there, allowing for the T cells that we’re about to infuse to quickly be activated, find the cancer, and kill it.”
Side effects can boost costs even more. “Unfortunately, some significant toxicities can occur after we infuse these cells,” Dr. Williams noted. “Patients can have trouble breathing and sometimes need ventilatory support. They can have trouble maintaining their blood pressure and become swollen as fluid seeps into tissues. Or they can have high fevers and organ dysfunction. Many of those patients go to the intensive care unit, which is obviously expensive as well.”
Taking Gene Therapy In-House
As Dr. Williams explained, one way to reduce costs is to “perform the genetic manipulation and expansion of the cells outside of a company.” Several academic institutions in the United States are embracing this approach, including Children’s Hospital of Philadelphia, which is experimenting with an automated device developed by the German company Miltenyi Biotec and known as the CliniMACS Prodigy machine.
“The current manufacturing process is very manual and requires a lot of interaction with the product and highly trained personnel,” Dr. Grupp said. “If you have an automated device, you have those cells in the device over the 7 to 12 days that you actually need to grow the cells. There’s much less interaction, so you need fewer trained personnel.”
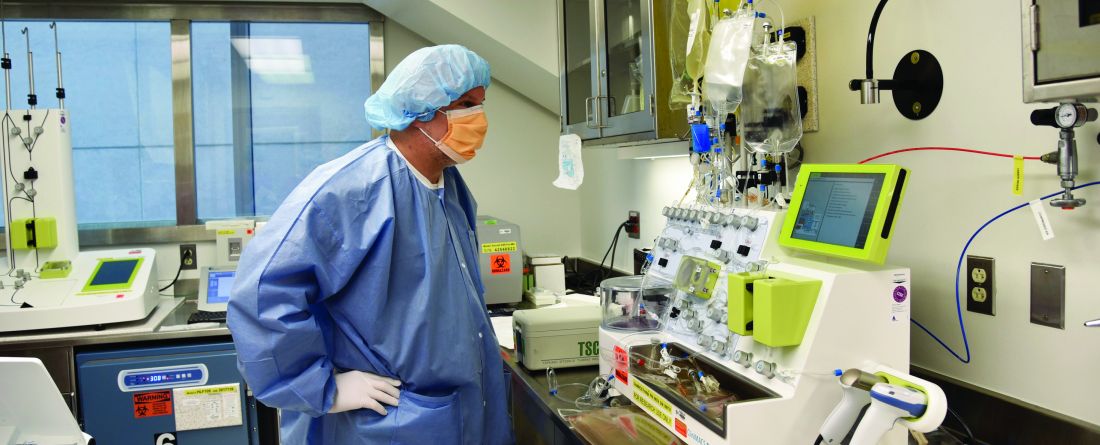
The device is experimental and not yet FDA approved, Dr. Grupp noted, so that patients are all in clinical trials. Children’s Hospital of Philadelphia has treated more than a dozen patients with the device, he said.
Another member of Children’s Hospital of Philadelphia’s CAR T-cell team told WHYY-FM that a single patient’s treatment would run about $30,000 for labor and testing, but not other expenses such as facility costs.
Dr. Grupp estimated that about half a dozen of these devices are in use in the United States, and many more worldwide. “They’re all just like we are — at the absolute beginning. We’ve only been doing this for about a year.”
In the big picture, Dr. Grupp said, “this is where cell therapy is going. Whether it’s point of care or not, automated cell manufacturing is the obvious next step.”
India: Big Hopes for Homegrown Technology
In India, researchers are hoping that their homegrown approach to CAR T-cell therapy will expand access by greatly lowering treatment prices.
Last fall, India’s equivalent of the FDA-granted approval for actalycabtagene autoleucel (NexCAR19), which was developed by Indian scientists who worked closely with the US National Institutes of Health (NIH). The therapy’s developer is a company called ImmunoACT.
In an interview, ImmunoACT founder Rahul Purwar, PhD, MSc, associate professor at Indian Institute of Technology Bombay, said the treatment costs about $40,000. The price is much lower than in the United States because staffing, facility construction, and maintenance are less expensive in India, he said.
Results of small early clinical trials have been promising, with complete responses in 68% of 38 lymphoma patients and 72% of 15 leukemia patients. Updated data will be presented at the annual American Society of Hematology meeting in December 2024, Dr. Purwar said.
According to the NIH, at first ImmunoACT hopes to treat about 1,200 patients a year. The immediate goal is to “focus and stabilize our operation in India,” Dr. Purwar said. “Then, if opportunities come, we will try to bring CAR T to all who might benefit from these technologies. A majority of countries don’t have access to these technologies.”
A US-Brazil Partnership Holds Promise
Meanwhile, a US nonprofit known as Caring Cross announced this year that it has partnered with Fundação Oswaldo Cruz (Fiocruz), a Brazilian government foundation, to manufacture CAR T cells at point-of-care in South America.
“Our model is different than traditional biotech/pharma,” Boro Dropulic, PhD, MBA, cofounder and executive director of Caring Cross, said in an interview. “Our goal is to develop technologies and transfer them to organizations like Fiocruz to enable them to manufacture these transformative therapies for patients in their regions. We believe this model is an important solution for therapies that are priced so high that they are not accessible to many patients that need them, particularly underserved populations and those in low- and middle-income countries.”
According to Dr. Dropulic: “We have developed a production process where the material cost is about $20,000 per dose.” When labor and infrastructure costs are added, the total expense won’t be more than $37,000-$47,500, he said.
The research process for the CAR T-cell technology is at an earlier stage than in India. Scientists plan to start clinical trials of the technology in the United States by the end of 2024 and then begin them in Brazil in 2025, after safety and efficacy have been demonstrated. The first trial, a phase I/II study, will enroll about 20 patients, Dr. Dropulic said.
Dr. Kenderian reported ties with Novartis, Capstan Bio, Kite/Gilead, Juno/BMS, Humanigen, Tolero, Leah Labs, Lentigen, Luminary, Sunesis/Viracta, Morphosys, Troque, Carisma, Sendero, and LifEngine. Dr. Williams disclosed grants from National Institutes of Health and philanthropic organizations. Dr. Grupp reported relationships with Novartis, Kite, Vertex and Servier, Roche, GSK, Humanigen, CBMG, Eureka, Janssen/JNJ, Jazz, Adaptimmune, TCR2, Cellectis, Juno, Allogene, and Cabaletta. Dr. Purwar is the founder of ImmunoACT. Dr. Dropulic serves as executive director of Caring Cross and CEO of Vector BioMed, which provides vectors for gene therapy.
In the United States, a stand-alone device could greatly reduce the expense of producing modified immune cells. In India, researchers hope homegrown technology is the key to getting costs under control. In Latin America, a partnership between the Brazilian government and a US nonprofit may be just the ticket.
At stake is expanded access to CAR T-cell therapy, a form of immunotherapy that in just the past few years has revolutionized the care of hematologic cancers.
“Among patients with lymphoma, leukemia, and myeloma, anywhere between 30% to 50% reach long-term remission after one CAR T-cell infusion,” Mayo Clinic–Rochester hematologist/oncologist Saad J. Kenderian, MB, ChB, said in an interview. “It’s such an important therapy.”
However, only a small percentage of eligible patients in the United States — perhaps 20% or fewer — are receiving the treatment, he added.
A 2024 report suggested that many patients in the United States who may benefit aren’t being treated because of a range of possible reasons, including high prices, manufacturing logistics, and far distance from the limited number of institutions offering the therapy.
“Taken together, the real-world cost of CAR T-cell therapy can range from $700,000 to $1 million, which may make the treatment unaffordable to those patients without robust financial and/or social support,” the report authors noted.
Outside Western countries, access to the therapy is even more limited, because of its exorbitant price. The 2024 report noted that “there is a wide use of CAR T-cell therapy in Europe and China, but access is limited in developing countries in Southeast Asia, Africa, and Latin America.”
Harnessing the Power of T-Cells
Several types of CAR T-cell therapy have been approved by the US Food and Drug Administration (FDA) for patients with relapsed/refractory blood cancers such as follicular lymphoma, large B-cell lymphoma, multiple myeloma, and B-cell precursor acute lymphoblastic leukemia. A 2023 review analyzed clinical trials and reported that complete response rates were 40%-54% in aggressive B-cell lymphoma, 67% in mantle cell lymphoma, and 69%-74% in indolent B-cell lymphoma.
Pediatric hematologist/oncologist Kirsten Williams, MD, who specializes in pediatric blood and marrow transplant and cellular therapy at the Aflac Cancer and Blood Disorders Center of Children’s Healthcare of Atlanta, described CAR T-cell therapy as “a very unique form of immunotherapy” that harnesses the power of the immune system’s T-cells.
These cells are effective tumor killers, but they typically aren’t assigned to control cancer, she said in an interview. “We have very few of them, and most of our T cells are focused on killing various viruses,” she said. The therapy “allows us to take the T cell that would have killed the flu or mono and instead target leukemia, B-cell leukemia, or lymphoma.”
As she explained, “T cells are collected by a machine that reserves white blood cells and gives back the rest of the blood to the patient. We insert a gene into the T cells that encodes for a B-cell receptor. This receptor acts as a GPS signal, pulling T cells to the cancer so that they can kill it.”
In addition, “with this genetic change, we also add some things that allow the T cell to be stronger, to have a higher signal to kill the cancer cell once it locks on.”
The therapy is unique for each patient, Dr. Williams said. “We have collected and modified your specific T cells, and they can now only be infused into you. If we try to give your product to someone else, those cells would either cause harm by attacking the patient or would be immediately killed by that patient’s own immune system. This is very different than all the other kinds of therapies. When you take other medicines, it doesn’t matter who receives that pill.”
Treatment: Individual, Complex, and Costly
Why is CAR T-cell therapy so expensive? While only a single treatment is needed, the T cells have to go through an “individualized, bespoke manufacturing” process that’s “highly technical,” pediatric oncologist Stephan A. Grupp, MD, PhD, section chief of the Cellular Therapy and Transplant Section at Children’s Hospital of Philadelphia, said in an interview. As he explained, the cells for a single patient have to go through the same testing as with a drug that might be given to 1,000 people.
“The first thing we need to do is collect the cells from a patient,” said Dr. Williams. “For adults, that process involves putting in two big IVs — one in each arm — and then pulling the blood through a machine. This typically involves an 8-hour collection in the hospital and very highly specialized people to oversee the collection process.”
Secondly, at some institutions, “the cells get sent to a company where they undergo the process where the gene is inserted,” she said. “This process needs to be done in a very sterile environment so there’s no infections, and it needs to have a lot of oversight.”
Finally, “after the cells are generated, they are typically frozen and shipped back to the site where the patient is at the hospital,” she said. “Then we give chemotherapy to the patient, which prepares the patient’s blood system. It removes some of the T-cells that are there, allowing for the T cells that we’re about to infuse to quickly be activated, find the cancer, and kill it.”
Side effects can boost costs even more. “Unfortunately, some significant toxicities can occur after we infuse these cells,” Dr. Williams noted. “Patients can have trouble breathing and sometimes need ventilatory support. They can have trouble maintaining their blood pressure and become swollen as fluid seeps into tissues. Or they can have high fevers and organ dysfunction. Many of those patients go to the intensive care unit, which is obviously expensive as well.”
Taking Gene Therapy In-House
As Dr. Williams explained, one way to reduce costs is to “perform the genetic manipulation and expansion of the cells outside of a company.” Several academic institutions in the United States are embracing this approach, including Children’s Hospital of Philadelphia, which is experimenting with an automated device developed by the German company Miltenyi Biotec and known as the CliniMACS Prodigy machine.
“The current manufacturing process is very manual and requires a lot of interaction with the product and highly trained personnel,” Dr. Grupp said. “If you have an automated device, you have those cells in the device over the 7 to 12 days that you actually need to grow the cells. There’s much less interaction, so you need fewer trained personnel.”

The device is experimental and not yet FDA approved, Dr. Grupp noted, so that patients are all in clinical trials. Children’s Hospital of Philadelphia has treated more than a dozen patients with the device, he said.
Another member of Children’s Hospital of Philadelphia’s CAR T-cell team told WHYY-FM that a single patient’s treatment would run about $30,000 for labor and testing, but not other expenses such as facility costs.
Dr. Grupp estimated that about half a dozen of these devices are in use in the United States, and many more worldwide. “They’re all just like we are — at the absolute beginning. We’ve only been doing this for about a year.”
In the big picture, Dr. Grupp said, “this is where cell therapy is going. Whether it’s point of care or not, automated cell manufacturing is the obvious next step.”
India: Big Hopes for Homegrown Technology
In India, researchers are hoping that their homegrown approach to CAR T-cell therapy will expand access by greatly lowering treatment prices.
Last fall, India’s equivalent of the FDA-granted approval for actalycabtagene autoleucel (NexCAR19), which was developed by Indian scientists who worked closely with the US National Institutes of Health (NIH). The therapy’s developer is a company called ImmunoACT.
In an interview, ImmunoACT founder Rahul Purwar, PhD, MSc, associate professor at Indian Institute of Technology Bombay, said the treatment costs about $40,000. The price is much lower than in the United States because staffing, facility construction, and maintenance are less expensive in India, he said.
Results of small early clinical trials have been promising, with complete responses in 68% of 38 lymphoma patients and 72% of 15 leukemia patients. Updated data will be presented at the annual American Society of Hematology meeting in December 2024, Dr. Purwar said.
According to the NIH, at first ImmunoACT hopes to treat about 1,200 patients a year. The immediate goal is to “focus and stabilize our operation in India,” Dr. Purwar said. “Then, if opportunities come, we will try to bring CAR T to all who might benefit from these technologies. A majority of countries don’t have access to these technologies.”
A US-Brazil Partnership Holds Promise
Meanwhile, a US nonprofit known as Caring Cross announced this year that it has partnered with Fundação Oswaldo Cruz (Fiocruz), a Brazilian government foundation, to manufacture CAR T cells at point-of-care in South America.
“Our model is different than traditional biotech/pharma,” Boro Dropulic, PhD, MBA, cofounder and executive director of Caring Cross, said in an interview. “Our goal is to develop technologies and transfer them to organizations like Fiocruz to enable them to manufacture these transformative therapies for patients in their regions. We believe this model is an important solution for therapies that are priced so high that they are not accessible to many patients that need them, particularly underserved populations and those in low- and middle-income countries.”
According to Dr. Dropulic: “We have developed a production process where the material cost is about $20,000 per dose.” When labor and infrastructure costs are added, the total expense won’t be more than $37,000-$47,500, he said.
The research process for the CAR T-cell technology is at an earlier stage than in India. Scientists plan to start clinical trials of the technology in the United States by the end of 2024 and then begin them in Brazil in 2025, after safety and efficacy have been demonstrated. The first trial, a phase I/II study, will enroll about 20 patients, Dr. Dropulic said.
Dr. Kenderian reported ties with Novartis, Capstan Bio, Kite/Gilead, Juno/BMS, Humanigen, Tolero, Leah Labs, Lentigen, Luminary, Sunesis/Viracta, Morphosys, Troque, Carisma, Sendero, and LifEngine. Dr. Williams disclosed grants from National Institutes of Health and philanthropic organizations. Dr. Grupp reported relationships with Novartis, Kite, Vertex and Servier, Roche, GSK, Humanigen, CBMG, Eureka, Janssen/JNJ, Jazz, Adaptimmune, TCR2, Cellectis, Juno, Allogene, and Cabaletta. Dr. Purwar is the founder of ImmunoACT. Dr. Dropulic serves as executive director of Caring Cross and CEO of Vector BioMed, which provides vectors for gene therapy.
In the United States, a stand-alone device could greatly reduce the expense of producing modified immune cells. In India, researchers hope homegrown technology is the key to getting costs under control. In Latin America, a partnership between the Brazilian government and a US nonprofit may be just the ticket.
At stake is expanded access to CAR T-cell therapy, a form of immunotherapy that in just the past few years has revolutionized the care of hematologic cancers.
“Among patients with lymphoma, leukemia, and myeloma, anywhere between 30% to 50% reach long-term remission after one CAR T-cell infusion,” Mayo Clinic–Rochester hematologist/oncologist Saad J. Kenderian, MB, ChB, said in an interview. “It’s such an important therapy.”
However, only a small percentage of eligible patients in the United States — perhaps 20% or fewer — are receiving the treatment, he added.
A 2024 report suggested that many patients in the United States who may benefit aren’t being treated because of a range of possible reasons, including high prices, manufacturing logistics, and far distance from the limited number of institutions offering the therapy.
“Taken together, the real-world cost of CAR T-cell therapy can range from $700,000 to $1 million, which may make the treatment unaffordable to those patients without robust financial and/or social support,” the report authors noted.
Outside Western countries, access to the therapy is even more limited, because of its exorbitant price. The 2024 report noted that “there is a wide use of CAR T-cell therapy in Europe and China, but access is limited in developing countries in Southeast Asia, Africa, and Latin America.”
Harnessing the Power of T-Cells
Several types of CAR T-cell therapy have been approved by the US Food and Drug Administration (FDA) for patients with relapsed/refractory blood cancers such as follicular lymphoma, large B-cell lymphoma, multiple myeloma, and B-cell precursor acute lymphoblastic leukemia. A 2023 review analyzed clinical trials and reported that complete response rates were 40%-54% in aggressive B-cell lymphoma, 67% in mantle cell lymphoma, and 69%-74% in indolent B-cell lymphoma.
Pediatric hematologist/oncologist Kirsten Williams, MD, who specializes in pediatric blood and marrow transplant and cellular therapy at the Aflac Cancer and Blood Disorders Center of Children’s Healthcare of Atlanta, described CAR T-cell therapy as “a very unique form of immunotherapy” that harnesses the power of the immune system’s T-cells.
These cells are effective tumor killers, but they typically aren’t assigned to control cancer, she said in an interview. “We have very few of them, and most of our T cells are focused on killing various viruses,” she said. The therapy “allows us to take the T cell that would have killed the flu or mono and instead target leukemia, B-cell leukemia, or lymphoma.”
As she explained, “T cells are collected by a machine that reserves white blood cells and gives back the rest of the blood to the patient. We insert a gene into the T cells that encodes for a B-cell receptor. This receptor acts as a GPS signal, pulling T cells to the cancer so that they can kill it.”
In addition, “with this genetic change, we also add some things that allow the T cell to be stronger, to have a higher signal to kill the cancer cell once it locks on.”
The therapy is unique for each patient, Dr. Williams said. “We have collected and modified your specific T cells, and they can now only be infused into you. If we try to give your product to someone else, those cells would either cause harm by attacking the patient or would be immediately killed by that patient’s own immune system. This is very different than all the other kinds of therapies. When you take other medicines, it doesn’t matter who receives that pill.”
Treatment: Individual, Complex, and Costly
Why is CAR T-cell therapy so expensive? While only a single treatment is needed, the T cells have to go through an “individualized, bespoke manufacturing” process that’s “highly technical,” pediatric oncologist Stephan A. Grupp, MD, PhD, section chief of the Cellular Therapy and Transplant Section at Children’s Hospital of Philadelphia, said in an interview. As he explained, the cells for a single patient have to go through the same testing as with a drug that might be given to 1,000 people.
“The first thing we need to do is collect the cells from a patient,” said Dr. Williams. “For adults, that process involves putting in two big IVs — one in each arm — and then pulling the blood through a machine. This typically involves an 8-hour collection in the hospital and very highly specialized people to oversee the collection process.”
Secondly, at some institutions, “the cells get sent to a company where they undergo the process where the gene is inserted,” she said. “This process needs to be done in a very sterile environment so there’s no infections, and it needs to have a lot of oversight.”
Finally, “after the cells are generated, they are typically frozen and shipped back to the site where the patient is at the hospital,” she said. “Then we give chemotherapy to the patient, which prepares the patient’s blood system. It removes some of the T-cells that are there, allowing for the T cells that we’re about to infuse to quickly be activated, find the cancer, and kill it.”
Side effects can boost costs even more. “Unfortunately, some significant toxicities can occur after we infuse these cells,” Dr. Williams noted. “Patients can have trouble breathing and sometimes need ventilatory support. They can have trouble maintaining their blood pressure and become swollen as fluid seeps into tissues. Or they can have high fevers and organ dysfunction. Many of those patients go to the intensive care unit, which is obviously expensive as well.”
Taking Gene Therapy In-House
As Dr. Williams explained, one way to reduce costs is to “perform the genetic manipulation and expansion of the cells outside of a company.” Several academic institutions in the United States are embracing this approach, including Children’s Hospital of Philadelphia, which is experimenting with an automated device developed by the German company Miltenyi Biotec and known as the CliniMACS Prodigy machine.
“The current manufacturing process is very manual and requires a lot of interaction with the product and highly trained personnel,” Dr. Grupp said. “If you have an automated device, you have those cells in the device over the 7 to 12 days that you actually need to grow the cells. There’s much less interaction, so you need fewer trained personnel.”

The device is experimental and not yet FDA approved, Dr. Grupp noted, so that patients are all in clinical trials. Children’s Hospital of Philadelphia has treated more than a dozen patients with the device, he said.
Another member of Children’s Hospital of Philadelphia’s CAR T-cell team told WHYY-FM that a single patient’s treatment would run about $30,000 for labor and testing, but not other expenses such as facility costs.
Dr. Grupp estimated that about half a dozen of these devices are in use in the United States, and many more worldwide. “They’re all just like we are — at the absolute beginning. We’ve only been doing this for about a year.”
In the big picture, Dr. Grupp said, “this is where cell therapy is going. Whether it’s point of care or not, automated cell manufacturing is the obvious next step.”
India: Big Hopes for Homegrown Technology
In India, researchers are hoping that their homegrown approach to CAR T-cell therapy will expand access by greatly lowering treatment prices.
Last fall, India’s equivalent of the FDA-granted approval for actalycabtagene autoleucel (NexCAR19), which was developed by Indian scientists who worked closely with the US National Institutes of Health (NIH). The therapy’s developer is a company called ImmunoACT.
In an interview, ImmunoACT founder Rahul Purwar, PhD, MSc, associate professor at Indian Institute of Technology Bombay, said the treatment costs about $40,000. The price is much lower than in the United States because staffing, facility construction, and maintenance are less expensive in India, he said.
Results of small early clinical trials have been promising, with complete responses in 68% of 38 lymphoma patients and 72% of 15 leukemia patients. Updated data will be presented at the annual American Society of Hematology meeting in December 2024, Dr. Purwar said.
According to the NIH, at first ImmunoACT hopes to treat about 1,200 patients a year. The immediate goal is to “focus and stabilize our operation in India,” Dr. Purwar said. “Then, if opportunities come, we will try to bring CAR T to all who might benefit from these technologies. A majority of countries don’t have access to these technologies.”
A US-Brazil Partnership Holds Promise
Meanwhile, a US nonprofit known as Caring Cross announced this year that it has partnered with Fundação Oswaldo Cruz (Fiocruz), a Brazilian government foundation, to manufacture CAR T cells at point-of-care in South America.
“Our model is different than traditional biotech/pharma,” Boro Dropulic, PhD, MBA, cofounder and executive director of Caring Cross, said in an interview. “Our goal is to develop technologies and transfer them to organizations like Fiocruz to enable them to manufacture these transformative therapies for patients in their regions. We believe this model is an important solution for therapies that are priced so high that they are not accessible to many patients that need them, particularly underserved populations and those in low- and middle-income countries.”
According to Dr. Dropulic: “We have developed a production process where the material cost is about $20,000 per dose.” When labor and infrastructure costs are added, the total expense won’t be more than $37,000-$47,500, he said.
The research process for the CAR T-cell technology is at an earlier stage than in India. Scientists plan to start clinical trials of the technology in the United States by the end of 2024 and then begin them in Brazil in 2025, after safety and efficacy have been demonstrated. The first trial, a phase I/II study, will enroll about 20 patients, Dr. Dropulic said.
Dr. Kenderian reported ties with Novartis, Capstan Bio, Kite/Gilead, Juno/BMS, Humanigen, Tolero, Leah Labs, Lentigen, Luminary, Sunesis/Viracta, Morphosys, Troque, Carisma, Sendero, and LifEngine. Dr. Williams disclosed grants from National Institutes of Health and philanthropic organizations. Dr. Grupp reported relationships with Novartis, Kite, Vertex and Servier, Roche, GSK, Humanigen, CBMG, Eureka, Janssen/JNJ, Jazz, Adaptimmune, TCR2, Cellectis, Juno, Allogene, and Cabaletta. Dr. Purwar is the founder of ImmunoACT. Dr. Dropulic serves as executive director of Caring Cross and CEO of Vector BioMed, which provides vectors for gene therapy.
The Long, Controversial Search for a ‘Cancer Microbiome’
Last year, the controversy heightened when experts questioned a high-profile study — a 2020 analysis claiming that the tumors of 33 different cancers had their own unique microbiomes — on whether the “signature” of these bacterial compositions could help diagnose cancer.
The incident renewed the spotlight on “tumor microbiomes” because of the bold claims of the original paper and the strongly worded refutations of those claims. The broader field has focused primarily on ways the body’s microbiome interacts with cancers and cancer treatment.
This controversy has highlighted the challenges of making headway in a field where researchers may not even have the tools yet to puzzle-out the wide-ranging implications the microbiome holds for cancer diagnosis and treatment.
But it is also part of a provocative question within that larger field: whether tumors in the body, far from the natural microbiome in the gut, have their own thriving communities of bacteria, viruses, and fungi. And, if they do, how do those tumor microbiomes affect the development and progression of the cancer and the effectiveness of cancer therapies?
Cancer Controversy
The evidence is undeniable that some microbes can directly cause certain cancers and that the human gut microbiome can influence the effectiveness of certain therapies. Beyond that established science, however, the research has raised as many questions as answers about what we do and don’t know about microbiota and cancer.
The only confirmed microbiomes are on the skin and in the gut, mouth, and vagina, which are all areas with an easy direct route for bacteria to enter and grow in or on the body. A series of papers in recent years have suggested that other internal organs, and tumors within them, may have their own microbiomes.
“Whether microbes exist in tumors of internal organs beyond body surfaces exposed to the environment is a different matter,” said Ivan Vujkovic-Cvijin, PhD, an assistant professor of biomedical sciences and gastroenterology at Cedars-Sinai Medical Center in Los Angeles, whose lab studies how human gut microbes affect inflammatory diseases. “We’ve only recently had the tools to study that question on a molecular level, and the reported results have been conflicting.”
For example, research allegedly identified microbiota in the human placenta nearly one decade ago. But subsequent research contradicted those claims and showed that the source of the “placental microbiome” was actually contamination. Subsequent similar studies for other parts of the body faced the same scrutiny and, often, eventual debunking.
“Most likely, our immune system has undergone selective pressure to eliminate everything that crosses the gut barrier because there’s not much benefit to the body to have bacteria run amok in our internal organs,” Dr. Vujkovic-Cvijin said. “That can only disrupt the functioning of our tissues, to have an external organism living inside them.”
The controversy that erupted last summer, surrounding research from the lab of Rob Knight, PhD, at the University of California, San Diego, centered on a slightly different but related question: Could tumors harbor their own microbiomes?
This news organization spoke with two of the authors who published a paper contesting Dr. Knight’s findings: Steven Salzberg, PhD, a professor of biomedical engineering at John Hopkins Medicine, Baltimore, Maryland, and Abraham Gihawi, PhD, a research fellow at Norwich Medical School at the University of East Anglia in the United Kingdom.
Dr. Salzberg described two major problems with Dr. Knight’s study.
“What they found were false positives because of contamination in the database and flaws in their methods,” Dr. Salzberg said. “I can’t prove there’s no cancer microbiome, but I can say the cancer microbiomes that they reported don’t exist because the species they were finding aren’t there.”
Dr. Knight disagrees with Dr. Salzberg’s findings, noting that Dr. Salzberg and his co-authors did not examine the publicly available databases used in his study. In a written response, he said that his team’s examination of the database revealed that less than 1% of the microbial genomes overlapped with human ones and that removing them did not change their findings.
Dr. Knight also noted that his team could still “distinguish cancer types by their microbiome” even after running their analysis without the technique that Dr. Salzberg found fault with.
Dr. Salzberg said that the database linked above is not the one Dr. Knight’s study used, however. “The primary database in their study was never made public (it’s too large, they said), and it has/had about 69,000 genomes,” Dr. Salzberg said by email. “But even if we did, this is irrelevant. He’s trying to distract from the primary errors in their study,” which Dr. Salzberg said Dr. Knight’s team has not addressed.
The critiques Dr. Salzberg raised have been leveled at other studies investigating microbiomes specifically within tumors and independent of the body’s microbiome.
For example, a 2019 study in Nature described a fungal microbiome in pancreatic cancer that a Nature paper 4 years later directly contradicted, citing flaws that invalidated the original findings. A different 2019 study in Cell examined pancreatic tumor microbiota and patient outcomes, but it’s unclear whether the microorganisms moved from the gut to the pancreas or “constitute a durably colonized community that lives inside the tumor,” which remains a matter of debate, Dr. Vujkovic-Cvijin said.
A 2020 study in Science suggested diverse microbial communities in seven tumor types, but those findings were similarly called into question. That study stated that “bacteria were first detected in human tumors more than 100 years ago” and that “bacteria are well-known residents in human tumors,” but Dr. Salzberg considers those statements misleading.
It’s true that bacteria and viruses have been detected in tumors because “there’s very good evidence that an acute infection caused by a very small number of viruses and bacteria can cause a tumor,” Dr. Salzberg said. Human papillomavirus, for example, can cause six different types of cancer. Inflammation and ulcers caused by Helicobacter pylori may progress to stomach cancer, and Fusobacterium nucleatum and Enterococcus faecalis have been shown to contribute to colorectal cancer. Those examples differ from a microbiome; this “a community of bacteria and possibly other microscopic bugs, like fungi, that are happily living in the tumor” the same way microbes reside in our guts, he said.
Dr. Knight said that many bacteria his team identified “have been confirmed independently in subsequent work.” He acknowledged, however, that more research is needed.
Several of the contested studies above were among a lengthy list that Dr. Knight provided, noting that most of the disagreements “have two sides to them, and critiques from one particular group does not immediately invalidate a reported finding.”
Yet, many of the papers Dr. Knight listed are precisely the types that skeptics like Dr. Salzberg believe are too flawed to draw reliable conclusions.
“I think many agree that microbes may exist within tumors that are exposed to the environment, like tumors of the skin, gut, and mouth,” Dr. Vujkovic-Cvijin said. It’s less clear, however, whether tumors further from the body’s microbiome harbor any microbes or where they came from if they do. Microbial signals in organs elsewhere in the body become faint quickly, he said.
Underdeveloped Technology
Though Dr. Salzberg said that the concept of a tumor microbiome is “implausible” because there’s no easy route for bacteria to reach internal organs, it’s unclear whether scientists have the technology yet to adequately answer this question.
For one thing, samples in these types of studies are typically “ultra-low biomass samples, where the signal — the amount of microbes in the sample — is so low that it’s comparable to how much would be expected to be found in reagents and environmental contamination through processing,” Dr. Vujkovic-Cvijin explained. Many polymerases used to amplify a DNA signal, for example, are made in bacteria and may retain trace amounts identified in these studies.
Dr. Knight agreed that low biomass is a challenge in this field but is not an unsurmountable one.
Another challenge is that study samples, as with Dr. Knight’s work, were collected during routine surgeries without the intent to find a microbial signal. Simply using a scalpel to cut through the skin means cutting through a layer of bacteria, and surgery rooms are not designed to eliminate all bacteria. Some work has even shown there is a “hospital microbiome,” so “you can easily have that creep into your signal and mistake it for tumor-resident bacteria,” Dr. Vujkovic-Cvijin said.
Dr. Knight asserted that the samples are taken under sterile conditions, but other researchers do not think the level of sterility necessary for completely clean samples is possible.
“Just because it’s in your sample doesn’t mean it was in your tumor,” Dr. Gihawi said.
Even if scientists can retrieve a reliable sample without contamination, analyzing it requires comparing the genetic material to existing databases of microbial genomes. Yet, contamination and misclassification of genetic sequences can be problems in those reference genomes too, Dr. Gihawi explained.
Machine learning algorithms have a role in interpreting data, but “we need to be careful of what we use them for,” he added.
“These techniques are in their infancy, and we’re starting to chase them down, which is why we need to move microbiome research in a way that can be used clinically,” Dr. Gihawi said.
Influence on Cancer Treatment Outcomes
Again, however, the question of whether microbiomes exist within tumors is only one slice of the much larger field looking at microbiomes and cancer, including its influence on cancer treatment outcomes. Although much remains to be learned, less controversy exists over the thousands of studies in the past two decades that have gradually revealed how the body’s microbiome can affect both the course of a cancer and the effectiveness of different treatments.
The growing research showing the importance of the gut microbiome in cancer treatments is not surprising given its role in immunity more broadly. Because the human immune system must recognize and defend against microbes, the microbiome helps train it, Dr. Vujkovic-Cvijin said.
Some bacteria can escape the gut — a phenomenon called bacterial translocation — and may aid in fighting tumors. To grow large enough to be seen on imaging, tumors need to evolve several abilities, such as growing enough vascularization to receive blood flow and shutting down local immune responses.
“Any added boost, like immunotherapy, has a chance of breaking through that immune forcefield and killing the tumor cells,” Dr. Vujkovic-Cvijin said. Escaped gut bacteria may provide that boost.
“There’s a lot of evidence that depletion of the gut microbiome impairs immunotherapy and chemotherapy. The thinking behind some of those studies is that gut microbes can cross the gut barrier and when they do, they activate the immune system,” he said.
In mice engineered to have sterile guts, for example, the lack of bacteria results in less effective immune systems, Dr. Vujkovic-Cvijin pointed out. A host of research has shown that antibiotic exposure during and even 6 months before immunotherapy dramatically reduces survival rates. “That’s pretty convincing to me that gut microbes are important,” he said.
Dr. Vujkovic-Cvijin cautioned that there continues to be controversy on understanding which bacteria are important for response to immunotherapy. “The field is still in its infancy in terms of understanding which bacteria are most important for these effects,” he said.
Dr. Knight suggested that escaped bacteria may be the genesis of the ones that he and other researchers believe exist in tumors. “Because tumor microbes must come from somewhere, it is to be expected that some of those microbes will be co-opted from body-site specific commensals.”
It’s also possible that metabolites released from gut bacteria escape the gut and could theoretically affect distant tumor growth, Dr. Gihawi said. The most promising avenue of research in this area is metabolites being used as biomarkers, added Dr. Gihawi, whose lab published research on a link between bacteria detected in men’s urine and a more aggressive subset of prostate cancers. But that research is not far enough along to develop lab tests for clinical use, he noted.
No Consensus Yet
Even before the controversy erupted around Dr. Knight’s research, he co-founded the company Micronoma to develop cancer tests based on his microbe findings. The company has raised $17.5 million from private investors as of August 2023 and received the US Food and Drug Administration’s Breakthrough Device designation, allowing the firm to fast-track clinical trials testing the technology. The recent critiques have not changed the company’s plans.
It’s safe to say that scientists will continue to research and debate the possibility of tumor microbiomes until a consensus emerges.
“The field is evolving and studies testing the reproducibility of tumor-resident microbial signals are essential for developing our understanding in this area,” Dr. Vujkovic-Cvijin said.
Even if that path ultimately leads nowhere, as Dr. Salzberg expects, research into microbiomes and cancer has plenty of other directions to go.
“I’m actually quite an optimist,” Dr. Gihawi said. “I think there’s a lot of scope for some really good research here, especially in the sites where we know there is a strong microbiome, such as the gastrointestinal tract.”
A version of this article appeared on Medscape.com.
Last year, the controversy heightened when experts questioned a high-profile study — a 2020 analysis claiming that the tumors of 33 different cancers had their own unique microbiomes — on whether the “signature” of these bacterial compositions could help diagnose cancer.
The incident renewed the spotlight on “tumor microbiomes” because of the bold claims of the original paper and the strongly worded refutations of those claims. The broader field has focused primarily on ways the body’s microbiome interacts with cancers and cancer treatment.
This controversy has highlighted the challenges of making headway in a field where researchers may not even have the tools yet to puzzle-out the wide-ranging implications the microbiome holds for cancer diagnosis and treatment.
But it is also part of a provocative question within that larger field: whether tumors in the body, far from the natural microbiome in the gut, have their own thriving communities of bacteria, viruses, and fungi. And, if they do, how do those tumor microbiomes affect the development and progression of the cancer and the effectiveness of cancer therapies?
Cancer Controversy
The evidence is undeniable that some microbes can directly cause certain cancers and that the human gut microbiome can influence the effectiveness of certain therapies. Beyond that established science, however, the research has raised as many questions as answers about what we do and don’t know about microbiota and cancer.
The only confirmed microbiomes are on the skin and in the gut, mouth, and vagina, which are all areas with an easy direct route for bacteria to enter and grow in or on the body. A series of papers in recent years have suggested that other internal organs, and tumors within them, may have their own microbiomes.
“Whether microbes exist in tumors of internal organs beyond body surfaces exposed to the environment is a different matter,” said Ivan Vujkovic-Cvijin, PhD, an assistant professor of biomedical sciences and gastroenterology at Cedars-Sinai Medical Center in Los Angeles, whose lab studies how human gut microbes affect inflammatory diseases. “We’ve only recently had the tools to study that question on a molecular level, and the reported results have been conflicting.”
For example, research allegedly identified microbiota in the human placenta nearly one decade ago. But subsequent research contradicted those claims and showed that the source of the “placental microbiome” was actually contamination. Subsequent similar studies for other parts of the body faced the same scrutiny and, often, eventual debunking.
“Most likely, our immune system has undergone selective pressure to eliminate everything that crosses the gut barrier because there’s not much benefit to the body to have bacteria run amok in our internal organs,” Dr. Vujkovic-Cvijin said. “That can only disrupt the functioning of our tissues, to have an external organism living inside them.”
The controversy that erupted last summer, surrounding research from the lab of Rob Knight, PhD, at the University of California, San Diego, centered on a slightly different but related question: Could tumors harbor their own microbiomes?
This news organization spoke with two of the authors who published a paper contesting Dr. Knight’s findings: Steven Salzberg, PhD, a professor of biomedical engineering at John Hopkins Medicine, Baltimore, Maryland, and Abraham Gihawi, PhD, a research fellow at Norwich Medical School at the University of East Anglia in the United Kingdom.
Dr. Salzberg described two major problems with Dr. Knight’s study.
“What they found were false positives because of contamination in the database and flaws in their methods,” Dr. Salzberg said. “I can’t prove there’s no cancer microbiome, but I can say the cancer microbiomes that they reported don’t exist because the species they were finding aren’t there.”
Dr. Knight disagrees with Dr. Salzberg’s findings, noting that Dr. Salzberg and his co-authors did not examine the publicly available databases used in his study. In a written response, he said that his team’s examination of the database revealed that less than 1% of the microbial genomes overlapped with human ones and that removing them did not change their findings.
Dr. Knight also noted that his team could still “distinguish cancer types by their microbiome” even after running their analysis without the technique that Dr. Salzberg found fault with.
Dr. Salzberg said that the database linked above is not the one Dr. Knight’s study used, however. “The primary database in their study was never made public (it’s too large, they said), and it has/had about 69,000 genomes,” Dr. Salzberg said by email. “But even if we did, this is irrelevant. He’s trying to distract from the primary errors in their study,” which Dr. Salzberg said Dr. Knight’s team has not addressed.
The critiques Dr. Salzberg raised have been leveled at other studies investigating microbiomes specifically within tumors and independent of the body’s microbiome.
For example, a 2019 study in Nature described a fungal microbiome in pancreatic cancer that a Nature paper 4 years later directly contradicted, citing flaws that invalidated the original findings. A different 2019 study in Cell examined pancreatic tumor microbiota and patient outcomes, but it’s unclear whether the microorganisms moved from the gut to the pancreas or “constitute a durably colonized community that lives inside the tumor,” which remains a matter of debate, Dr. Vujkovic-Cvijin said.
A 2020 study in Science suggested diverse microbial communities in seven tumor types, but those findings were similarly called into question. That study stated that “bacteria were first detected in human tumors more than 100 years ago” and that “bacteria are well-known residents in human tumors,” but Dr. Salzberg considers those statements misleading.
It’s true that bacteria and viruses have been detected in tumors because “there’s very good evidence that an acute infection caused by a very small number of viruses and bacteria can cause a tumor,” Dr. Salzberg said. Human papillomavirus, for example, can cause six different types of cancer. Inflammation and ulcers caused by Helicobacter pylori may progress to stomach cancer, and Fusobacterium nucleatum and Enterococcus faecalis have been shown to contribute to colorectal cancer. Those examples differ from a microbiome; this “a community of bacteria and possibly other microscopic bugs, like fungi, that are happily living in the tumor” the same way microbes reside in our guts, he said.
Dr. Knight said that many bacteria his team identified “have been confirmed independently in subsequent work.” He acknowledged, however, that more research is needed.
Several of the contested studies above were among a lengthy list that Dr. Knight provided, noting that most of the disagreements “have two sides to them, and critiques from one particular group does not immediately invalidate a reported finding.”
Yet, many of the papers Dr. Knight listed are precisely the types that skeptics like Dr. Salzberg believe are too flawed to draw reliable conclusions.
“I think many agree that microbes may exist within tumors that are exposed to the environment, like tumors of the skin, gut, and mouth,” Dr. Vujkovic-Cvijin said. It’s less clear, however, whether tumors further from the body’s microbiome harbor any microbes or where they came from if they do. Microbial signals in organs elsewhere in the body become faint quickly, he said.
Underdeveloped Technology
Though Dr. Salzberg said that the concept of a tumor microbiome is “implausible” because there’s no easy route for bacteria to reach internal organs, it’s unclear whether scientists have the technology yet to adequately answer this question.
For one thing, samples in these types of studies are typically “ultra-low biomass samples, where the signal — the amount of microbes in the sample — is so low that it’s comparable to how much would be expected to be found in reagents and environmental contamination through processing,” Dr. Vujkovic-Cvijin explained. Many polymerases used to amplify a DNA signal, for example, are made in bacteria and may retain trace amounts identified in these studies.
Dr. Knight agreed that low biomass is a challenge in this field but is not an unsurmountable one.
Another challenge is that study samples, as with Dr. Knight’s work, were collected during routine surgeries without the intent to find a microbial signal. Simply using a scalpel to cut through the skin means cutting through a layer of bacteria, and surgery rooms are not designed to eliminate all bacteria. Some work has even shown there is a “hospital microbiome,” so “you can easily have that creep into your signal and mistake it for tumor-resident bacteria,” Dr. Vujkovic-Cvijin said.
Dr. Knight asserted that the samples are taken under sterile conditions, but other researchers do not think the level of sterility necessary for completely clean samples is possible.
“Just because it’s in your sample doesn’t mean it was in your tumor,” Dr. Gihawi said.
Even if scientists can retrieve a reliable sample without contamination, analyzing it requires comparing the genetic material to existing databases of microbial genomes. Yet, contamination and misclassification of genetic sequences can be problems in those reference genomes too, Dr. Gihawi explained.
Machine learning algorithms have a role in interpreting data, but “we need to be careful of what we use them for,” he added.
“These techniques are in their infancy, and we’re starting to chase them down, which is why we need to move microbiome research in a way that can be used clinically,” Dr. Gihawi said.
Influence on Cancer Treatment Outcomes
Again, however, the question of whether microbiomes exist within tumors is only one slice of the much larger field looking at microbiomes and cancer, including its influence on cancer treatment outcomes. Although much remains to be learned, less controversy exists over the thousands of studies in the past two decades that have gradually revealed how the body’s microbiome can affect both the course of a cancer and the effectiveness of different treatments.
The growing research showing the importance of the gut microbiome in cancer treatments is not surprising given its role in immunity more broadly. Because the human immune system must recognize and defend against microbes, the microbiome helps train it, Dr. Vujkovic-Cvijin said.
Some bacteria can escape the gut — a phenomenon called bacterial translocation — and may aid in fighting tumors. To grow large enough to be seen on imaging, tumors need to evolve several abilities, such as growing enough vascularization to receive blood flow and shutting down local immune responses.
“Any added boost, like immunotherapy, has a chance of breaking through that immune forcefield and killing the tumor cells,” Dr. Vujkovic-Cvijin said. Escaped gut bacteria may provide that boost.
“There’s a lot of evidence that depletion of the gut microbiome impairs immunotherapy and chemotherapy. The thinking behind some of those studies is that gut microbes can cross the gut barrier and when they do, they activate the immune system,” he said.
In mice engineered to have sterile guts, for example, the lack of bacteria results in less effective immune systems, Dr. Vujkovic-Cvijin pointed out. A host of research has shown that antibiotic exposure during and even 6 months before immunotherapy dramatically reduces survival rates. “That’s pretty convincing to me that gut microbes are important,” he said.
Dr. Vujkovic-Cvijin cautioned that there continues to be controversy on understanding which bacteria are important for response to immunotherapy. “The field is still in its infancy in terms of understanding which bacteria are most important for these effects,” he said.
Dr. Knight suggested that escaped bacteria may be the genesis of the ones that he and other researchers believe exist in tumors. “Because tumor microbes must come from somewhere, it is to be expected that some of those microbes will be co-opted from body-site specific commensals.”
It’s also possible that metabolites released from gut bacteria escape the gut and could theoretically affect distant tumor growth, Dr. Gihawi said. The most promising avenue of research in this area is metabolites being used as biomarkers, added Dr. Gihawi, whose lab published research on a link between bacteria detected in men’s urine and a more aggressive subset of prostate cancers. But that research is not far enough along to develop lab tests for clinical use, he noted.
No Consensus Yet
Even before the controversy erupted around Dr. Knight’s research, he co-founded the company Micronoma to develop cancer tests based on his microbe findings. The company has raised $17.5 million from private investors as of August 2023 and received the US Food and Drug Administration’s Breakthrough Device designation, allowing the firm to fast-track clinical trials testing the technology. The recent critiques have not changed the company’s plans.
It’s safe to say that scientists will continue to research and debate the possibility of tumor microbiomes until a consensus emerges.
“The field is evolving and studies testing the reproducibility of tumor-resident microbial signals are essential for developing our understanding in this area,” Dr. Vujkovic-Cvijin said.
Even if that path ultimately leads nowhere, as Dr. Salzberg expects, research into microbiomes and cancer has plenty of other directions to go.
“I’m actually quite an optimist,” Dr. Gihawi said. “I think there’s a lot of scope for some really good research here, especially in the sites where we know there is a strong microbiome, such as the gastrointestinal tract.”
A version of this article appeared on Medscape.com.
Last year, the controversy heightened when experts questioned a high-profile study — a 2020 analysis claiming that the tumors of 33 different cancers had their own unique microbiomes — on whether the “signature” of these bacterial compositions could help diagnose cancer.
The incident renewed the spotlight on “tumor microbiomes” because of the bold claims of the original paper and the strongly worded refutations of those claims. The broader field has focused primarily on ways the body’s microbiome interacts with cancers and cancer treatment.
This controversy has highlighted the challenges of making headway in a field where researchers may not even have the tools yet to puzzle-out the wide-ranging implications the microbiome holds for cancer diagnosis and treatment.
But it is also part of a provocative question within that larger field: whether tumors in the body, far from the natural microbiome in the gut, have their own thriving communities of bacteria, viruses, and fungi. And, if they do, how do those tumor microbiomes affect the development and progression of the cancer and the effectiveness of cancer therapies?
Cancer Controversy
The evidence is undeniable that some microbes can directly cause certain cancers and that the human gut microbiome can influence the effectiveness of certain therapies. Beyond that established science, however, the research has raised as many questions as answers about what we do and don’t know about microbiota and cancer.
The only confirmed microbiomes are on the skin and in the gut, mouth, and vagina, which are all areas with an easy direct route for bacteria to enter and grow in or on the body. A series of papers in recent years have suggested that other internal organs, and tumors within them, may have their own microbiomes.
“Whether microbes exist in tumors of internal organs beyond body surfaces exposed to the environment is a different matter,” said Ivan Vujkovic-Cvijin, PhD, an assistant professor of biomedical sciences and gastroenterology at Cedars-Sinai Medical Center in Los Angeles, whose lab studies how human gut microbes affect inflammatory diseases. “We’ve only recently had the tools to study that question on a molecular level, and the reported results have been conflicting.”
For example, research allegedly identified microbiota in the human placenta nearly one decade ago. But subsequent research contradicted those claims and showed that the source of the “placental microbiome” was actually contamination. Subsequent similar studies for other parts of the body faced the same scrutiny and, often, eventual debunking.
“Most likely, our immune system has undergone selective pressure to eliminate everything that crosses the gut barrier because there’s not much benefit to the body to have bacteria run amok in our internal organs,” Dr. Vujkovic-Cvijin said. “That can only disrupt the functioning of our tissues, to have an external organism living inside them.”
The controversy that erupted last summer, surrounding research from the lab of Rob Knight, PhD, at the University of California, San Diego, centered on a slightly different but related question: Could tumors harbor their own microbiomes?
This news organization spoke with two of the authors who published a paper contesting Dr. Knight’s findings: Steven Salzberg, PhD, a professor of biomedical engineering at John Hopkins Medicine, Baltimore, Maryland, and Abraham Gihawi, PhD, a research fellow at Norwich Medical School at the University of East Anglia in the United Kingdom.
Dr. Salzberg described two major problems with Dr. Knight’s study.
“What they found were false positives because of contamination in the database and flaws in their methods,” Dr. Salzberg said. “I can’t prove there’s no cancer microbiome, but I can say the cancer microbiomes that they reported don’t exist because the species they were finding aren’t there.”
Dr. Knight disagrees with Dr. Salzberg’s findings, noting that Dr. Salzberg and his co-authors did not examine the publicly available databases used in his study. In a written response, he said that his team’s examination of the database revealed that less than 1% of the microbial genomes overlapped with human ones and that removing them did not change their findings.
Dr. Knight also noted that his team could still “distinguish cancer types by their microbiome” even after running their analysis without the technique that Dr. Salzberg found fault with.
Dr. Salzberg said that the database linked above is not the one Dr. Knight’s study used, however. “The primary database in their study was never made public (it’s too large, they said), and it has/had about 69,000 genomes,” Dr. Salzberg said by email. “But even if we did, this is irrelevant. He’s trying to distract from the primary errors in their study,” which Dr. Salzberg said Dr. Knight’s team has not addressed.
The critiques Dr. Salzberg raised have been leveled at other studies investigating microbiomes specifically within tumors and independent of the body’s microbiome.
For example, a 2019 study in Nature described a fungal microbiome in pancreatic cancer that a Nature paper 4 years later directly contradicted, citing flaws that invalidated the original findings. A different 2019 study in Cell examined pancreatic tumor microbiota and patient outcomes, but it’s unclear whether the microorganisms moved from the gut to the pancreas or “constitute a durably colonized community that lives inside the tumor,” which remains a matter of debate, Dr. Vujkovic-Cvijin said.
A 2020 study in Science suggested diverse microbial communities in seven tumor types, but those findings were similarly called into question. That study stated that “bacteria were first detected in human tumors more than 100 years ago” and that “bacteria are well-known residents in human tumors,” but Dr. Salzberg considers those statements misleading.
It’s true that bacteria and viruses have been detected in tumors because “there’s very good evidence that an acute infection caused by a very small number of viruses and bacteria can cause a tumor,” Dr. Salzberg said. Human papillomavirus, for example, can cause six different types of cancer. Inflammation and ulcers caused by Helicobacter pylori may progress to stomach cancer, and Fusobacterium nucleatum and Enterococcus faecalis have been shown to contribute to colorectal cancer. Those examples differ from a microbiome; this “a community of bacteria and possibly other microscopic bugs, like fungi, that are happily living in the tumor” the same way microbes reside in our guts, he said.
Dr. Knight said that many bacteria his team identified “have been confirmed independently in subsequent work.” He acknowledged, however, that more research is needed.
Several of the contested studies above were among a lengthy list that Dr. Knight provided, noting that most of the disagreements “have two sides to them, and critiques from one particular group does not immediately invalidate a reported finding.”
Yet, many of the papers Dr. Knight listed are precisely the types that skeptics like Dr. Salzberg believe are too flawed to draw reliable conclusions.
“I think many agree that microbes may exist within tumors that are exposed to the environment, like tumors of the skin, gut, and mouth,” Dr. Vujkovic-Cvijin said. It’s less clear, however, whether tumors further from the body’s microbiome harbor any microbes or where they came from if they do. Microbial signals in organs elsewhere in the body become faint quickly, he said.
Underdeveloped Technology
Though Dr. Salzberg said that the concept of a tumor microbiome is “implausible” because there’s no easy route for bacteria to reach internal organs, it’s unclear whether scientists have the technology yet to adequately answer this question.
For one thing, samples in these types of studies are typically “ultra-low biomass samples, where the signal — the amount of microbes in the sample — is so low that it’s comparable to how much would be expected to be found in reagents and environmental contamination through processing,” Dr. Vujkovic-Cvijin explained. Many polymerases used to amplify a DNA signal, for example, are made in bacteria and may retain trace amounts identified in these studies.
Dr. Knight agreed that low biomass is a challenge in this field but is not an unsurmountable one.
Another challenge is that study samples, as with Dr. Knight’s work, were collected during routine surgeries without the intent to find a microbial signal. Simply using a scalpel to cut through the skin means cutting through a layer of bacteria, and surgery rooms are not designed to eliminate all bacteria. Some work has even shown there is a “hospital microbiome,” so “you can easily have that creep into your signal and mistake it for tumor-resident bacteria,” Dr. Vujkovic-Cvijin said.
Dr. Knight asserted that the samples are taken under sterile conditions, but other researchers do not think the level of sterility necessary for completely clean samples is possible.
“Just because it’s in your sample doesn’t mean it was in your tumor,” Dr. Gihawi said.
Even if scientists can retrieve a reliable sample without contamination, analyzing it requires comparing the genetic material to existing databases of microbial genomes. Yet, contamination and misclassification of genetic sequences can be problems in those reference genomes too, Dr. Gihawi explained.
Machine learning algorithms have a role in interpreting data, but “we need to be careful of what we use them for,” he added.
“These techniques are in their infancy, and we’re starting to chase them down, which is why we need to move microbiome research in a way that can be used clinically,” Dr. Gihawi said.
Influence on Cancer Treatment Outcomes
Again, however, the question of whether microbiomes exist within tumors is only one slice of the much larger field looking at microbiomes and cancer, including its influence on cancer treatment outcomes. Although much remains to be learned, less controversy exists over the thousands of studies in the past two decades that have gradually revealed how the body’s microbiome can affect both the course of a cancer and the effectiveness of different treatments.
The growing research showing the importance of the gut microbiome in cancer treatments is not surprising given its role in immunity more broadly. Because the human immune system must recognize and defend against microbes, the microbiome helps train it, Dr. Vujkovic-Cvijin said.
Some bacteria can escape the gut — a phenomenon called bacterial translocation — and may aid in fighting tumors. To grow large enough to be seen on imaging, tumors need to evolve several abilities, such as growing enough vascularization to receive blood flow and shutting down local immune responses.
“Any added boost, like immunotherapy, has a chance of breaking through that immune forcefield and killing the tumor cells,” Dr. Vujkovic-Cvijin said. Escaped gut bacteria may provide that boost.
“There’s a lot of evidence that depletion of the gut microbiome impairs immunotherapy and chemotherapy. The thinking behind some of those studies is that gut microbes can cross the gut barrier and when they do, they activate the immune system,” he said.
In mice engineered to have sterile guts, for example, the lack of bacteria results in less effective immune systems, Dr. Vujkovic-Cvijin pointed out. A host of research has shown that antibiotic exposure during and even 6 months before immunotherapy dramatically reduces survival rates. “That’s pretty convincing to me that gut microbes are important,” he said.
Dr. Vujkovic-Cvijin cautioned that there continues to be controversy on understanding which bacteria are important for response to immunotherapy. “The field is still in its infancy in terms of understanding which bacteria are most important for these effects,” he said.
Dr. Knight suggested that escaped bacteria may be the genesis of the ones that he and other researchers believe exist in tumors. “Because tumor microbes must come from somewhere, it is to be expected that some of those microbes will be co-opted from body-site specific commensals.”
It’s also possible that metabolites released from gut bacteria escape the gut and could theoretically affect distant tumor growth, Dr. Gihawi said. The most promising avenue of research in this area is metabolites being used as biomarkers, added Dr. Gihawi, whose lab published research on a link between bacteria detected in men’s urine and a more aggressive subset of prostate cancers. But that research is not far enough along to develop lab tests for clinical use, he noted.
No Consensus Yet
Even before the controversy erupted around Dr. Knight’s research, he co-founded the company Micronoma to develop cancer tests based on his microbe findings. The company has raised $17.5 million from private investors as of August 2023 and received the US Food and Drug Administration’s Breakthrough Device designation, allowing the firm to fast-track clinical trials testing the technology. The recent critiques have not changed the company’s plans.
It’s safe to say that scientists will continue to research and debate the possibility of tumor microbiomes until a consensus emerges.
“The field is evolving and studies testing the reproducibility of tumor-resident microbial signals are essential for developing our understanding in this area,” Dr. Vujkovic-Cvijin said.
Even if that path ultimately leads nowhere, as Dr. Salzberg expects, research into microbiomes and cancer has plenty of other directions to go.
“I’m actually quite an optimist,” Dr. Gihawi said. “I think there’s a lot of scope for some really good research here, especially in the sites where we know there is a strong microbiome, such as the gastrointestinal tract.”
A version of this article appeared on Medscape.com.
Do No Harm: What Smoldering Myeloma Teaches Us
My approach to treating SMM takes into account what its history can teach us about 1) how advancements in imaging and diagnostic reclassifications can revise the entire natural history of a disease, and 2) how evidence generated by even the best of studies may have an expiration date.
Much of what we know about SMM today dates to a pivotal study by Robert A. Kyle, MD, and colleagues, published in 2007. That inspirational team of investigators followed people diagnosed with SMM from 1970 to 1995 and established the first natural history of the condition. Their monumental effort and the data and conclusions it generated (eg,10% risk annually of SMM becoming MM for the first 5 years) are still cited today in references, papers, and slide sets.
Despite the seminal importance of this work, from today’s perspective the 2007 study might just as well have been describing a different disease. Back then people were diagnosed with SMM if their blood work detected a monoclonal protein and a follow-up bone marrow biopsy found at least 10% plasma cells (or a monoclonal protein exceeding 3g/dL). If there were no signs of end-organ damage (ie, no anemia or kidney problems) and an x-ray showed no fractures or lesions in the bones, the diagnosis was determined to be SMM.
What’s different in 2024? First and foremost: advanced, highly sensitive imaging techniques. MRIs can pick up small lytic lesions (and even the precursor to lytic lesions) that would not appear on an x-ray. In fact, relying solely on x-rays risks missing half of the lytic lesions.
Therefore, using the same criteria, many people who in the past were diagnosed with SMM would today be diagnosed with MM. Furthermore, in 2014 a diagnostic change reclassified people’s diagnosis from the highest risk category of SMM to the category of active MM.
Due to these scientific advances and classification changes, I believe that the natural history of SMM is unknown. Risk stratification models for SMM derived from data sets of people who had not undergone rigorous advanced imaging likely are skewed by data from people who had MM. In addition, current risk stratification models have very poor concordance with each other. I routinely see people whose 2-year risk according to different models varies by more than 30%-40%.
All this information tells us that SMM today is more indolent than the SMM of the past. Paradoxically, however, our therapies keep getting more and more aggressive, exposing this vulnerable group of people to intense treatment regimens that they may not require. Therapies tested on people diagnosed with SMM include an aggressive three-drug regimen, autologous stem cell transplant, and 2 years of additional therapy, as well as, more recently CAR T-cell therapy which so far has at least a 4%-5% treatment-related mortality risk in people with myeloma and a strong signal for secondary cancer risk. Other trials are testing bispecific therapies such as talquetamab, a drug which in my experience causes horrendous skin toxicity, profound weight loss, and one’s nails to fall off.
Doctors routinely keep showing slides from Kyle’s pivotal work to describe the natural history of SMM and to justify the need for treatment, and trials continue to use outdated progression prediction models. In my opinion, as people with MM keep living longer and treatments for MM keep getting better, the threshold for intervening with asymptomatic, healthy people with SMM should be getting higher, not lower.
I strongly believe that the current landscape of SMM treatment exemplifies good intentions leading to bad outcomes. A routine blood test in a completely healthy person that finds elevated total protein in the blood could culminate in well-intentioned but aggressive therapies that can lead to many serious side effects. (I repeat: Secondary cancers and deaths from infections have all occurred in SMM trials.)
With no control arm, we simply don’t know how well these people might have fared without any therapy. For all we know, treatment may have shortened their lives due to complications up to and including death — all because of a blood test often conducted for reasons that have no evidentiary basis.
For example, plasma cell diseases are not linked to low bone density or auto-immune diseases, yet these labs are sent routinely as part of a workup for those conditions, leading to increasing anxiety and costs.
So, what is my approach? When treating people with SMM, I hold nuanced discussions of this data to help prioritize and reach informed decisions. After our honest conversation about the limitations of SMM models, older data, and the limitations of prospective data studying pharmacological treatment, almost no one signs up for treatment.
I want these people to stay safe, and I’m proud to be a part of a trial (SPOTLIGHT, NCT06212323) that aims to show prospectively that these people can be watched off treatment with monitoring via advanced imaging modalities.
In conclusion: SMM teaches us how, even in the absence of pharmacological interventions, the natural history of a disease can change over time, simply via better imaging techniques and changes in diagnostic classifications. Unfortunately, SMM also illustrates how good intentions can lead to harm.
Dr. Mohyuddin is assistant professor in the multiple myeloma program at the Huntsman Cancer Institute at the University of Utah in Salt Lake City.
My approach to treating SMM takes into account what its history can teach us about 1) how advancements in imaging and diagnostic reclassifications can revise the entire natural history of a disease, and 2) how evidence generated by even the best of studies may have an expiration date.
Much of what we know about SMM today dates to a pivotal study by Robert A. Kyle, MD, and colleagues, published in 2007. That inspirational team of investigators followed people diagnosed with SMM from 1970 to 1995 and established the first natural history of the condition. Their monumental effort and the data and conclusions it generated (eg,10% risk annually of SMM becoming MM for the first 5 years) are still cited today in references, papers, and slide sets.
Despite the seminal importance of this work, from today’s perspective the 2007 study might just as well have been describing a different disease. Back then people were diagnosed with SMM if their blood work detected a monoclonal protein and a follow-up bone marrow biopsy found at least 10% plasma cells (or a monoclonal protein exceeding 3g/dL). If there were no signs of end-organ damage (ie, no anemia or kidney problems) and an x-ray showed no fractures or lesions in the bones, the diagnosis was determined to be SMM.
What’s different in 2024? First and foremost: advanced, highly sensitive imaging techniques. MRIs can pick up small lytic lesions (and even the precursor to lytic lesions) that would not appear on an x-ray. In fact, relying solely on x-rays risks missing half of the lytic lesions.
Therefore, using the same criteria, many people who in the past were diagnosed with SMM would today be diagnosed with MM. Furthermore, in 2014 a diagnostic change reclassified people’s diagnosis from the highest risk category of SMM to the category of active MM.
Due to these scientific advances and classification changes, I believe that the natural history of SMM is unknown. Risk stratification models for SMM derived from data sets of people who had not undergone rigorous advanced imaging likely are skewed by data from people who had MM. In addition, current risk stratification models have very poor concordance with each other. I routinely see people whose 2-year risk according to different models varies by more than 30%-40%.
All this information tells us that SMM today is more indolent than the SMM of the past. Paradoxically, however, our therapies keep getting more and more aggressive, exposing this vulnerable group of people to intense treatment regimens that they may not require. Therapies tested on people diagnosed with SMM include an aggressive three-drug regimen, autologous stem cell transplant, and 2 years of additional therapy, as well as, more recently CAR T-cell therapy which so far has at least a 4%-5% treatment-related mortality risk in people with myeloma and a strong signal for secondary cancer risk. Other trials are testing bispecific therapies such as talquetamab, a drug which in my experience causes horrendous skin toxicity, profound weight loss, and one’s nails to fall off.
Doctors routinely keep showing slides from Kyle’s pivotal work to describe the natural history of SMM and to justify the need for treatment, and trials continue to use outdated progression prediction models. In my opinion, as people with MM keep living longer and treatments for MM keep getting better, the threshold for intervening with asymptomatic, healthy people with SMM should be getting higher, not lower.
I strongly believe that the current landscape of SMM treatment exemplifies good intentions leading to bad outcomes. A routine blood test in a completely healthy person that finds elevated total protein in the blood could culminate in well-intentioned but aggressive therapies that can lead to many serious side effects. (I repeat: Secondary cancers and deaths from infections have all occurred in SMM trials.)
With no control arm, we simply don’t know how well these people might have fared without any therapy. For all we know, treatment may have shortened their lives due to complications up to and including death — all because of a blood test often conducted for reasons that have no evidentiary basis.
For example, plasma cell diseases are not linked to low bone density or auto-immune diseases, yet these labs are sent routinely as part of a workup for those conditions, leading to increasing anxiety and costs.
So, what is my approach? When treating people with SMM, I hold nuanced discussions of this data to help prioritize and reach informed decisions. After our honest conversation about the limitations of SMM models, older data, and the limitations of prospective data studying pharmacological treatment, almost no one signs up for treatment.
I want these people to stay safe, and I’m proud to be a part of a trial (SPOTLIGHT, NCT06212323) that aims to show prospectively that these people can be watched off treatment with monitoring via advanced imaging modalities.
In conclusion: SMM teaches us how, even in the absence of pharmacological interventions, the natural history of a disease can change over time, simply via better imaging techniques and changes in diagnostic classifications. Unfortunately, SMM also illustrates how good intentions can lead to harm.
Dr. Mohyuddin is assistant professor in the multiple myeloma program at the Huntsman Cancer Institute at the University of Utah in Salt Lake City.
My approach to treating SMM takes into account what its history can teach us about 1) how advancements in imaging and diagnostic reclassifications can revise the entire natural history of a disease, and 2) how evidence generated by even the best of studies may have an expiration date.
Much of what we know about SMM today dates to a pivotal study by Robert A. Kyle, MD, and colleagues, published in 2007. That inspirational team of investigators followed people diagnosed with SMM from 1970 to 1995 and established the first natural history of the condition. Their monumental effort and the data and conclusions it generated (eg,10% risk annually of SMM becoming MM for the first 5 years) are still cited today in references, papers, and slide sets.
Despite the seminal importance of this work, from today’s perspective the 2007 study might just as well have been describing a different disease. Back then people were diagnosed with SMM if their blood work detected a monoclonal protein and a follow-up bone marrow biopsy found at least 10% plasma cells (or a monoclonal protein exceeding 3g/dL). If there were no signs of end-organ damage (ie, no anemia or kidney problems) and an x-ray showed no fractures or lesions in the bones, the diagnosis was determined to be SMM.
What’s different in 2024? First and foremost: advanced, highly sensitive imaging techniques. MRIs can pick up small lytic lesions (and even the precursor to lytic lesions) that would not appear on an x-ray. In fact, relying solely on x-rays risks missing half of the lytic lesions.
Therefore, using the same criteria, many people who in the past were diagnosed with SMM would today be diagnosed with MM. Furthermore, in 2014 a diagnostic change reclassified people’s diagnosis from the highest risk category of SMM to the category of active MM.
Due to these scientific advances and classification changes, I believe that the natural history of SMM is unknown. Risk stratification models for SMM derived from data sets of people who had not undergone rigorous advanced imaging likely are skewed by data from people who had MM. In addition, current risk stratification models have very poor concordance with each other. I routinely see people whose 2-year risk according to different models varies by more than 30%-40%.
All this information tells us that SMM today is more indolent than the SMM of the past. Paradoxically, however, our therapies keep getting more and more aggressive, exposing this vulnerable group of people to intense treatment regimens that they may not require. Therapies tested on people diagnosed with SMM include an aggressive three-drug regimen, autologous stem cell transplant, and 2 years of additional therapy, as well as, more recently CAR T-cell therapy which so far has at least a 4%-5% treatment-related mortality risk in people with myeloma and a strong signal for secondary cancer risk. Other trials are testing bispecific therapies such as talquetamab, a drug which in my experience causes horrendous skin toxicity, profound weight loss, and one’s nails to fall off.
Doctors routinely keep showing slides from Kyle’s pivotal work to describe the natural history of SMM and to justify the need for treatment, and trials continue to use outdated progression prediction models. In my opinion, as people with MM keep living longer and treatments for MM keep getting better, the threshold for intervening with asymptomatic, healthy people with SMM should be getting higher, not lower.
I strongly believe that the current landscape of SMM treatment exemplifies good intentions leading to bad outcomes. A routine blood test in a completely healthy person that finds elevated total protein in the blood could culminate in well-intentioned but aggressive therapies that can lead to many serious side effects. (I repeat: Secondary cancers and deaths from infections have all occurred in SMM trials.)
With no control arm, we simply don’t know how well these people might have fared without any therapy. For all we know, treatment may have shortened their lives due to complications up to and including death — all because of a blood test often conducted for reasons that have no evidentiary basis.
For example, plasma cell diseases are not linked to low bone density or auto-immune diseases, yet these labs are sent routinely as part of a workup for those conditions, leading to increasing anxiety and costs.
So, what is my approach? When treating people with SMM, I hold nuanced discussions of this data to help prioritize and reach informed decisions. After our honest conversation about the limitations of SMM models, older data, and the limitations of prospective data studying pharmacological treatment, almost no one signs up for treatment.
I want these people to stay safe, and I’m proud to be a part of a trial (SPOTLIGHT, NCT06212323) that aims to show prospectively that these people can be watched off treatment with monitoring via advanced imaging modalities.
In conclusion: SMM teaches us how, even in the absence of pharmacological interventions, the natural history of a disease can change over time, simply via better imaging techniques and changes in diagnostic classifications. Unfortunately, SMM also illustrates how good intentions can lead to harm.
Dr. Mohyuddin is assistant professor in the multiple myeloma program at the Huntsman Cancer Institute at the University of Utah in Salt Lake City.
Few Cancer Survivors Meet ACS Nutrition, Exercise Guidelines
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The ACS has published nutrition and exercise guidelines for cancer survivors, which include recommendations to maintain a healthy weight and diet, cut out alcohol, and participate in regular physical activities. Engaging in these behaviors is associated with longer survival among cancer survivors, but whether survivors follow these nutrition and activity recommendations has not been systematically tracked.
- Researchers evaluated data on 10,020 individuals (mean age, 64.2 years) who had completed cancer treatment. Data came from the Behavioral Risk Factor Surveillance System telephone-based survey administered in 2017, 2019, and 2021, which represents 2.7 million cancer survivors.
- The researchers estimated survivors’ adherence to guidelines across four domains: Weight, physical activity, fruit and vegetable consumption, and alcohol intake. Factors associated with adherence were also evaluated.
- Overall, 9,121 survivors (91%) completed questionnaires for all four domains.
TAKEAWAY:
Only 4% of patients (365 of 9121) followed ACS guidelines in all four categories.
When assessing adherence to each category, the researchers found that 72% of cancer survivors reported engaging in recommended levels of physical activity, 68% maintained a nonobese weight, 50% said they did not consume alcohol, and 12% said they consumed recommended quantities of fruits and vegetables.
Compared with people in the general population, cancer survivors generally engaged in fewer healthy behaviors than those who had never been diagnosed with cancer.
The authors identified certain factors associated with greater guideline adherence, including female sex, older age, Black (vs White) race, and higher education level (college graduate).
IN PRACTICE:
This study highlights a potential “gap between published guidelines regarding behavioral modifications for cancer survivors and uptake of these behaviors,” the authors wrote, adding that “it is essential for oncologists and general internists to improve widespread and systematic counseling on these guidelines to improve uptake of healthy behaviors in this vulnerable patient population.”
SOURCE:
This work, led by Carter Baughman, MD, from the Division of Internal Medicine at Beth Israel Deaconess Medical Center, Boston, Massachusetts, was published online in JAMA Oncology.
LIMITATIONS:
The authors reported several study limitations, most notably that self-reported data may introduce biases.
DISCLOSURES:
The study funding source was not reported. One author received grants from the US Highbush Blueberry Council outside the submitted work. No other disclosures were reported.
A version of this article appeared on Medscape.com.
Most Targeted Cancer Drugs Lack Substantial Clinical Benefit
TOPLINE:
METHODOLOGY:
- The strength and quality of evidence supporting genome-targeted cancer drug approvals vary. A big reason is the growing number of cancer drug approvals based on surrogate endpoints, such as disease-free and progression-free survival, instead of clinical endpoints, such as overall survival or quality of life. The US Food and Drug Administration (FDA) has also approved genome-targeted cancer drugs based on phase 1 or single-arm trials.
- Given these less rigorous considerations for approval, “the validity and value of the targets and surrogate measures underlying FDA genome-targeted cancer drug approvals are uncertain,” the researchers explained.
- In the current analysis, researchers assessed the validity of the molecular targets as well as the clinical benefits of genome-targeted cancer drugs approved in the United States from 2015 to 2022 based on results from pivotal trials.
- The researchers evaluated the strength of evidence supporting molecular targetability using the European Society for Medical Oncology (ESMO) Scale for Clinical Actionability of Molecular Targets (ESCAT) and the clinical benefit using the ESMO–Magnitude of Clinical Benefit Scale (ESMO-MCBS).
- The authors defined a substantial clinical benefit as an A or B grade for curative intent and a 4 or 5 for noncurative intent. High-benefit genomic-based cancer treatments were defined as those associated with a substantial clinical benefit (ESMO-MCBS) and that qualified as ESCAT category level I-A (a clinical benefit based on prospective randomized data) or I-B (prospective nonrandomized data).
TAKEAWAY:
- The analyses focused on 50 molecular-targeted cancer drugs covering 84 indications. Of which, 45 indications (54%) were approved based on phase 1 or 2 pivotal trials, 45 (54%) were supported by single-arm pivotal trials and the remaining 39 (46%) by randomized trial, and 48 (57%) were approved based on subgroup analyses.
- Among the 84 indications, more than half (55%) of the pivotal trials supporting approval used overall response rate as a primary endpoint, 31% used progression-free survival, and 6% used disease-free survival. Only seven indications (8%) were supported by pivotal trials demonstrating an improvement in overall survival.
- Among the 84 trials, 24 (29%) met the ESMO-MCBS threshold for substantial clinical benefit.
- Overall, when combining all ratings, only 24 of the 84 indications (29%) were considered high-benefit genomic-based cancer treatments.
IN PRACTICE:
“We applied the ESMO-MCBS and ESCAT value frameworks to identify therapies and molecular targets providing high clinical value that should be widely available to patients” and “found that drug indications supported by these characteristics represent a minority of cancer drug approvals in recent years,” the authors said. Using these value frameworks could help payers, governments, and individual patients “prioritize the availability of high-value molecular-targeted therapies.”
SOURCE:
The study, with first author Ariadna Tibau, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, was published online in JAMA Oncology.
LIMITATIONS:
The study evaluated only trials that supported regulatory approval and did not include outcomes of postapproval clinical studies, which could lead to changes in ESMO-MCBS grades and ESCAT levels of evidence over time.
DISCLOSURES:
The study was funded by the Kaiser Permanente Institute for Health Policy, Arnold Ventures, and the Commonwealth Fund. The authors had no relevant disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The strength and quality of evidence supporting genome-targeted cancer drug approvals vary. A big reason is the growing number of cancer drug approvals based on surrogate endpoints, such as disease-free and progression-free survival, instead of clinical endpoints, such as overall survival or quality of life. The US Food and Drug Administration (FDA) has also approved genome-targeted cancer drugs based on phase 1 or single-arm trials.
- Given these less rigorous considerations for approval, “the validity and value of the targets and surrogate measures underlying FDA genome-targeted cancer drug approvals are uncertain,” the researchers explained.
- In the current analysis, researchers assessed the validity of the molecular targets as well as the clinical benefits of genome-targeted cancer drugs approved in the United States from 2015 to 2022 based on results from pivotal trials.
- The researchers evaluated the strength of evidence supporting molecular targetability using the European Society for Medical Oncology (ESMO) Scale for Clinical Actionability of Molecular Targets (ESCAT) and the clinical benefit using the ESMO–Magnitude of Clinical Benefit Scale (ESMO-MCBS).
- The authors defined a substantial clinical benefit as an A or B grade for curative intent and a 4 or 5 for noncurative intent. High-benefit genomic-based cancer treatments were defined as those associated with a substantial clinical benefit (ESMO-MCBS) and that qualified as ESCAT category level I-A (a clinical benefit based on prospective randomized data) or I-B (prospective nonrandomized data).
TAKEAWAY:
- The analyses focused on 50 molecular-targeted cancer drugs covering 84 indications. Of which, 45 indications (54%) were approved based on phase 1 or 2 pivotal trials, 45 (54%) were supported by single-arm pivotal trials and the remaining 39 (46%) by randomized trial, and 48 (57%) were approved based on subgroup analyses.
- Among the 84 indications, more than half (55%) of the pivotal trials supporting approval used overall response rate as a primary endpoint, 31% used progression-free survival, and 6% used disease-free survival. Only seven indications (8%) were supported by pivotal trials demonstrating an improvement in overall survival.
- Among the 84 trials, 24 (29%) met the ESMO-MCBS threshold for substantial clinical benefit.
- Overall, when combining all ratings, only 24 of the 84 indications (29%) were considered high-benefit genomic-based cancer treatments.
IN PRACTICE:
“We applied the ESMO-MCBS and ESCAT value frameworks to identify therapies and molecular targets providing high clinical value that should be widely available to patients” and “found that drug indications supported by these characteristics represent a minority of cancer drug approvals in recent years,” the authors said. Using these value frameworks could help payers, governments, and individual patients “prioritize the availability of high-value molecular-targeted therapies.”
SOURCE:
The study, with first author Ariadna Tibau, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, was published online in JAMA Oncology.
LIMITATIONS:
The study evaluated only trials that supported regulatory approval and did not include outcomes of postapproval clinical studies, which could lead to changes in ESMO-MCBS grades and ESCAT levels of evidence over time.
DISCLOSURES:
The study was funded by the Kaiser Permanente Institute for Health Policy, Arnold Ventures, and the Commonwealth Fund. The authors had no relevant disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- The strength and quality of evidence supporting genome-targeted cancer drug approvals vary. A big reason is the growing number of cancer drug approvals based on surrogate endpoints, such as disease-free and progression-free survival, instead of clinical endpoints, such as overall survival or quality of life. The US Food and Drug Administration (FDA) has also approved genome-targeted cancer drugs based on phase 1 or single-arm trials.
- Given these less rigorous considerations for approval, “the validity and value of the targets and surrogate measures underlying FDA genome-targeted cancer drug approvals are uncertain,” the researchers explained.
- In the current analysis, researchers assessed the validity of the molecular targets as well as the clinical benefits of genome-targeted cancer drugs approved in the United States from 2015 to 2022 based on results from pivotal trials.
- The researchers evaluated the strength of evidence supporting molecular targetability using the European Society for Medical Oncology (ESMO) Scale for Clinical Actionability of Molecular Targets (ESCAT) and the clinical benefit using the ESMO–Magnitude of Clinical Benefit Scale (ESMO-MCBS).
- The authors defined a substantial clinical benefit as an A or B grade for curative intent and a 4 or 5 for noncurative intent. High-benefit genomic-based cancer treatments were defined as those associated with a substantial clinical benefit (ESMO-MCBS) and that qualified as ESCAT category level I-A (a clinical benefit based on prospective randomized data) or I-B (prospective nonrandomized data).
TAKEAWAY:
- The analyses focused on 50 molecular-targeted cancer drugs covering 84 indications. Of which, 45 indications (54%) were approved based on phase 1 or 2 pivotal trials, 45 (54%) were supported by single-arm pivotal trials and the remaining 39 (46%) by randomized trial, and 48 (57%) were approved based on subgroup analyses.
- Among the 84 indications, more than half (55%) of the pivotal trials supporting approval used overall response rate as a primary endpoint, 31% used progression-free survival, and 6% used disease-free survival. Only seven indications (8%) were supported by pivotal trials demonstrating an improvement in overall survival.
- Among the 84 trials, 24 (29%) met the ESMO-MCBS threshold for substantial clinical benefit.
- Overall, when combining all ratings, only 24 of the 84 indications (29%) were considered high-benefit genomic-based cancer treatments.
IN PRACTICE:
“We applied the ESMO-MCBS and ESCAT value frameworks to identify therapies and molecular targets providing high clinical value that should be widely available to patients” and “found that drug indications supported by these characteristics represent a minority of cancer drug approvals in recent years,” the authors said. Using these value frameworks could help payers, governments, and individual patients “prioritize the availability of high-value molecular-targeted therapies.”
SOURCE:
The study, with first author Ariadna Tibau, MD, PhD, Brigham and Women’s Hospital and Harvard Medical School, Boston, was published online in JAMA Oncology.
LIMITATIONS:
The study evaluated only trials that supported regulatory approval and did not include outcomes of postapproval clinical studies, which could lead to changes in ESMO-MCBS grades and ESCAT levels of evidence over time.
DISCLOSURES:
The study was funded by the Kaiser Permanente Institute for Health Policy, Arnold Ventures, and the Commonwealth Fund. The authors had no relevant disclosures.
A version of this article appeared on Medscape.com.
Panel: MRD Tests May Speed Myeloma Tx Approvals
The Oncologic Drugs Advisory Committee (ODAC) of the US Food and Drug Administration (FDA) voted 12-0 on April 12 on the following question: Does the evidence support the use of MRD as an accelerated approval endpoint in multiple myeloma clinical trials?
The FDA is not bound to accept the recommendations of its panels, but often does so.
ODAC panelists said they felt comfortable in this recommendation because they expected the FDA to mandate confirmatory studies of any drugs to be given accelerated approval based on MRD data.
There’s a risk that MRD results might mislead regulators into clearing a drug later found to lack benefit, said Christopher Hourigan, DM, DPhil, an ODAC panelist and a physician-scientist at Virginia Tech, Blacksburg, Virginia, who treats people with blood cancer. Further tests would ultimately show if drugs cleared based on MRD data actually delivered benefits such as extending progression-free survival (PFS).
“That’s why we’re talking about accelerated approval,” Dr. Hourigan said. “There is harm to inaction. We’re not currently curing people in multiple myeloma. I’m not willing to make patients wait on principle for a theoretical perfect that may never come.”
“Our responsibility is to accept the world as messy and be agile enough to adapt and iterate that the evidence develops rather than create barriers to the work of discovering effective new therapies for these patients,” he added.
Advances in testing now allow for detection of the presence of malignant cells at orders of magnitude below previous assessments. MRD assays used in tracking what’s happening with myeloma generally have a sensitivity level of 10-5, or a detection capacity of one cell of 100,000, said Ola Landgren, MD, PhD, of the University of Miami, Miami, Florida, during a presentation at the meeting.
The April 12 meeting was somewhat unusual for ODAC.
Instead of reviewing the benefits and risks of a specific drug, the panel reviewed results from two separate major research efforts done to see how MRD could be used in development of drugs.
These were Dr. Landgren’s EVIDENCE (Evaluating minimal residual disease as an intermediate clinical endpoint for multiple myeloma) meta-analysis, and the similar work of the i2TEAMM group, affiliated with the International Myeloma Foundation.
In its review, the FDA staff noted differences in the approaches of the two groups. In its analysis, the i2TEAMM removed information about patients with missing MRD data, while the University of Miami team retained information about these kinds of patients in the analyses and assigned their status to be MRD positive.
The FDA staff also noted in their review and presentations weaknesses in the case for MRD. For example, the FDA staff noted that the treatment effect on MRD negativity was not statistically significant in 4 of the 8 treatment comparisons in the work from Dr. Landgren and colleagues.
The FDA staff looked at what these analyses suggested at both an individual level and trial level. The data from these two research projects taken as a whole showed “strong individual-level” associations between negative MRD findings and later positive outcomes for patients, although trial-level associations were “weak to moderate” in some cases, the staff wrote.
The FDA staff concluded that the research appeared to support arguments in favor of the “prognostic value,” even with outstanding questions about how best to use this test.
In the briefing document for the meeting, the FDA also emphasized the need for new treatments.
Multiple myeloma remains an incurable disease with a 5-year relative survival rate of 59.8%, even after significant recent progress in treatment, the agency said. In the past decade, the FDA has approved 15 new drugs and greater than 20 new indications have been approved for the treatment of patients with multiple myeloma.
The FDA has been working with drugmakers and academic researchers for several years to address the potential of MRD in development of blood cancers. The agency in 2020 issued a guidance document on this issue.
Several ODAC members praised the i2TEAMM and Dr. Landgren’s EVIDENCE teams for their work, which took place across several nations and extended over many years.
“This was a herculean effort. It really changes the playbook for how we think about biomarkers across all cancer types,” said ODAC panelist Neil Vasan, MD, PhD, of Columbia University, New York, NY. “To me, the important word was reasonable. Is this a reasonable surrogate endpoint? Is this a reasonable intermediate endpoint? I think it is more than reasonable.”
Still, ODAC panelist Jorge Nieva, MD, raised a point of concern about how use of MRD as an endpoint could change the design of studies. He urged caution among researchers about potential ramping up of collection of MRD tests in search of more robust data, which could lead to more testing for patients.
“I have this tremendous fear that this is going to mean every myeloma protocol has a marrow biopsy every six weeks on the patients forever,” said Dr. Nieva of the Keck School of Medicine, University of Southern California, Los Angeles. “I just don’t want to see that happen. So I think we need to balance these two things.”
The Oncologic Drugs Advisory Committee (ODAC) of the US Food and Drug Administration (FDA) voted 12-0 on April 12 on the following question: Does the evidence support the use of MRD as an accelerated approval endpoint in multiple myeloma clinical trials?
The FDA is not bound to accept the recommendations of its panels, but often does so.
ODAC panelists said they felt comfortable in this recommendation because they expected the FDA to mandate confirmatory studies of any drugs to be given accelerated approval based on MRD data.
There’s a risk that MRD results might mislead regulators into clearing a drug later found to lack benefit, said Christopher Hourigan, DM, DPhil, an ODAC panelist and a physician-scientist at Virginia Tech, Blacksburg, Virginia, who treats people with blood cancer. Further tests would ultimately show if drugs cleared based on MRD data actually delivered benefits such as extending progression-free survival (PFS).
“That’s why we’re talking about accelerated approval,” Dr. Hourigan said. “There is harm to inaction. We’re not currently curing people in multiple myeloma. I’m not willing to make patients wait on principle for a theoretical perfect that may never come.”
“Our responsibility is to accept the world as messy and be agile enough to adapt and iterate that the evidence develops rather than create barriers to the work of discovering effective new therapies for these patients,” he added.
Advances in testing now allow for detection of the presence of malignant cells at orders of magnitude below previous assessments. MRD assays used in tracking what’s happening with myeloma generally have a sensitivity level of 10-5, or a detection capacity of one cell of 100,000, said Ola Landgren, MD, PhD, of the University of Miami, Miami, Florida, during a presentation at the meeting.
The April 12 meeting was somewhat unusual for ODAC.
Instead of reviewing the benefits and risks of a specific drug, the panel reviewed results from two separate major research efforts done to see how MRD could be used in development of drugs.
These were Dr. Landgren’s EVIDENCE (Evaluating minimal residual disease as an intermediate clinical endpoint for multiple myeloma) meta-analysis, and the similar work of the i2TEAMM group, affiliated with the International Myeloma Foundation.
In its review, the FDA staff noted differences in the approaches of the two groups. In its analysis, the i2TEAMM removed information about patients with missing MRD data, while the University of Miami team retained information about these kinds of patients in the analyses and assigned their status to be MRD positive.
The FDA staff also noted in their review and presentations weaknesses in the case for MRD. For example, the FDA staff noted that the treatment effect on MRD negativity was not statistically significant in 4 of the 8 treatment comparisons in the work from Dr. Landgren and colleagues.
The FDA staff looked at what these analyses suggested at both an individual level and trial level. The data from these two research projects taken as a whole showed “strong individual-level” associations between negative MRD findings and later positive outcomes for patients, although trial-level associations were “weak to moderate” in some cases, the staff wrote.
The FDA staff concluded that the research appeared to support arguments in favor of the “prognostic value,” even with outstanding questions about how best to use this test.
In the briefing document for the meeting, the FDA also emphasized the need for new treatments.
Multiple myeloma remains an incurable disease with a 5-year relative survival rate of 59.8%, even after significant recent progress in treatment, the agency said. In the past decade, the FDA has approved 15 new drugs and greater than 20 new indications have been approved for the treatment of patients with multiple myeloma.
The FDA has been working with drugmakers and academic researchers for several years to address the potential of MRD in development of blood cancers. The agency in 2020 issued a guidance document on this issue.
Several ODAC members praised the i2TEAMM and Dr. Landgren’s EVIDENCE teams for their work, which took place across several nations and extended over many years.
“This was a herculean effort. It really changes the playbook for how we think about biomarkers across all cancer types,” said ODAC panelist Neil Vasan, MD, PhD, of Columbia University, New York, NY. “To me, the important word was reasonable. Is this a reasonable surrogate endpoint? Is this a reasonable intermediate endpoint? I think it is more than reasonable.”
Still, ODAC panelist Jorge Nieva, MD, raised a point of concern about how use of MRD as an endpoint could change the design of studies. He urged caution among researchers about potential ramping up of collection of MRD tests in search of more robust data, which could lead to more testing for patients.
“I have this tremendous fear that this is going to mean every myeloma protocol has a marrow biopsy every six weeks on the patients forever,” said Dr. Nieva of the Keck School of Medicine, University of Southern California, Los Angeles. “I just don’t want to see that happen. So I think we need to balance these two things.”
The Oncologic Drugs Advisory Committee (ODAC) of the US Food and Drug Administration (FDA) voted 12-0 on April 12 on the following question: Does the evidence support the use of MRD as an accelerated approval endpoint in multiple myeloma clinical trials?
The FDA is not bound to accept the recommendations of its panels, but often does so.
ODAC panelists said they felt comfortable in this recommendation because they expected the FDA to mandate confirmatory studies of any drugs to be given accelerated approval based on MRD data.
There’s a risk that MRD results might mislead regulators into clearing a drug later found to lack benefit, said Christopher Hourigan, DM, DPhil, an ODAC panelist and a physician-scientist at Virginia Tech, Blacksburg, Virginia, who treats people with blood cancer. Further tests would ultimately show if drugs cleared based on MRD data actually delivered benefits such as extending progression-free survival (PFS).
“That’s why we’re talking about accelerated approval,” Dr. Hourigan said. “There is harm to inaction. We’re not currently curing people in multiple myeloma. I’m not willing to make patients wait on principle for a theoretical perfect that may never come.”
“Our responsibility is to accept the world as messy and be agile enough to adapt and iterate that the evidence develops rather than create barriers to the work of discovering effective new therapies for these patients,” he added.
Advances in testing now allow for detection of the presence of malignant cells at orders of magnitude below previous assessments. MRD assays used in tracking what’s happening with myeloma generally have a sensitivity level of 10-5, or a detection capacity of one cell of 100,000, said Ola Landgren, MD, PhD, of the University of Miami, Miami, Florida, during a presentation at the meeting.
The April 12 meeting was somewhat unusual for ODAC.
Instead of reviewing the benefits and risks of a specific drug, the panel reviewed results from two separate major research efforts done to see how MRD could be used in development of drugs.
These were Dr. Landgren’s EVIDENCE (Evaluating minimal residual disease as an intermediate clinical endpoint for multiple myeloma) meta-analysis, and the similar work of the i2TEAMM group, affiliated with the International Myeloma Foundation.
In its review, the FDA staff noted differences in the approaches of the two groups. In its analysis, the i2TEAMM removed information about patients with missing MRD data, while the University of Miami team retained information about these kinds of patients in the analyses and assigned their status to be MRD positive.
The FDA staff also noted in their review and presentations weaknesses in the case for MRD. For example, the FDA staff noted that the treatment effect on MRD negativity was not statistically significant in 4 of the 8 treatment comparisons in the work from Dr. Landgren and colleagues.
The FDA staff looked at what these analyses suggested at both an individual level and trial level. The data from these two research projects taken as a whole showed “strong individual-level” associations between negative MRD findings and later positive outcomes for patients, although trial-level associations were “weak to moderate” in some cases, the staff wrote.
The FDA staff concluded that the research appeared to support arguments in favor of the “prognostic value,” even with outstanding questions about how best to use this test.
In the briefing document for the meeting, the FDA also emphasized the need for new treatments.
Multiple myeloma remains an incurable disease with a 5-year relative survival rate of 59.8%, even after significant recent progress in treatment, the agency said. In the past decade, the FDA has approved 15 new drugs and greater than 20 new indications have been approved for the treatment of patients with multiple myeloma.
The FDA has been working with drugmakers and academic researchers for several years to address the potential of MRD in development of blood cancers. The agency in 2020 issued a guidance document on this issue.
Several ODAC members praised the i2TEAMM and Dr. Landgren’s EVIDENCE teams for their work, which took place across several nations and extended over many years.
“This was a herculean effort. It really changes the playbook for how we think about biomarkers across all cancer types,” said ODAC panelist Neil Vasan, MD, PhD, of Columbia University, New York, NY. “To me, the important word was reasonable. Is this a reasonable surrogate endpoint? Is this a reasonable intermediate endpoint? I think it is more than reasonable.”
Still, ODAC panelist Jorge Nieva, MD, raised a point of concern about how use of MRD as an endpoint could change the design of studies. He urged caution among researchers about potential ramping up of collection of MRD tests in search of more robust data, which could lead to more testing for patients.
“I have this tremendous fear that this is going to mean every myeloma protocol has a marrow biopsy every six weeks on the patients forever,” said Dr. Nieva of the Keck School of Medicine, University of Southern California, Los Angeles. “I just don’t want to see that happen. So I think we need to balance these two things.”
No Routine Cancer Screening Option? New MCED Tests May Help
Analyses presented during a session at the American Association for Cancer Research annual meeting, revealed that three new MCED tests — CanScan, MERCURY, and OncoSeek — could detect a range of cancers and recognize the tissue of origin with high accuracy. One — OncoSeek — could also provide an affordable cancer screening option for individuals living in lower-income countries.
The need for these noninvasive liquid biopsy tests that can accurately identify multiple cancer types with a single blood draw, especially cancers without routine screening strategies, is pressing. “We know that the current cancer standard of care screening will identify less than 50% of all cancers, while more than 50% of all cancer deaths occur in types of cancer with no recommended screening,” said co-moderator Marie E. Wood, MD, of the University of Colorado Anschutz Medical Campus, in Aurora, Colorado.
That being said, “the clinical utility of multicancer detection tests has not been established and we’re concerned about issues of overdiagnosis and overtreatment,” she noted.
The Early Data
One new MCED test called CanScan, developed by Geneseeq Technology, uses plasma cell-free DNA fragment patterns to detect cancer signals as well as identify the tissue of origin across 13 cancer types.
Overall, the CanScan test covers cancer types that contribute to two thirds of new cancer cases and 74% of morality globally, said presenter Shanshan Yang, of Geneseeq Research Institute, in Nanjing, China.
However, only five of these cancer types have screening recommendations issued by the US Preventive Services Task Force (USPSTF), Dr. Yang added.
The interim data comes from an ongoing large-scale prospective study evaluating the MCED test in a cohort of asymptomatic individuals between ages 45 and 75 years with an average risk for cancer and no cancer-related symptoms on enrollment.
Patients at baseline had their blood collected for the CanScan test and subsequently received annual routine physical exams once a year for 3 consecutive years, with an additional 2 years of follow-up.
The analysis included 3724 participants with analyzable samples at the data cutoff in September 2023. Among the 3724 participants, 29 had confirmed cancer diagnoses. Among these cases, 14 patients had their cancer confirmed through USPSTF recommended screening and 15 were detected through outside of standard USPSTF screening, such as a thyroid ultrasound, Dr. Yang explained.
Almost 90% of the cancers (26 of 29) were detected in the stage I or II, and eight (27.5%) were not one of the test’s 13 targeted cancer types.
The CanScan test had a sensitivity of 55.2%, identifying 16 of 29 of the patients with cancer, including 10 of 21 individuals with stage I (47.6%), and two of three with stage II (66.7%).
The test had a high specificity of 97.9%, meaning out of 100 people screened, only two had false negative findings.
Among the 15 patients who had their cancer detected outside of USPSTF screening recommendations, eight (53.3%) were found using a CanScan test, including patients with liver and endometrial cancers.
Compared with a positive predictive value of (PPV) of 1.6% with screening or physical exam methods alone, the CanScan test had a PPV of 17.4%, Dr. Yang reported.
“The MCED test holds significant potential for early cancer screening in asymptomatic populations,” Dr. Yang and colleagues concluded.
Another new MCED test called MERCURY, also developed by Geneseeq Technology and presented during the session, used a similar method to detect cancer signals and predict the tissue of origin across 13 cancer types.
The researchers initially validated the test using 3076 patients with cancer and 3477 healthy controls with a target specificity of 99%. In this group, researchers reported a sensitivity of 0.865 and a specificity of 0.989.
The team then performed an independent validation analysis with 1465 participants, 732 with cancer and 733 with no cancer, and confirmed a high sensitivity and specificity of 0.874 and 0.978, respectively. The sensitivity increased incrementally by cancer stage — 0.768 for stage I, 0.840 for stage II, 0.923 for stage III, and 0.971 for stage IV.
The test identified the tissue of origin with high accuracy, the researchers noted, but cautioned that the test needs “to be further validated in a prospective cohort study.”
MCED in Low-Income Settings
The session also featured findings on a new affordable MCED test called OncoSeek, which could provide greater access to cancer testing in low- and middle-income countries.
The OncoSeek algorithm identifies the presence of cancer using seven protein tumor markers alongside clinical information, such as gender and age. Like other tests, the test also predicts the possible tissue of origin.
The test can be run on clinical protein assay instruments that are already widely available, such as Roche cobas analyzer, Mao Mao, MD, PhD, the founder and CEO of SeekIn, of Shenzhen, China, told this news organization.
This “feature makes the test accessible worldwide, even in low- and middle-income countries,” he said. “These instruments are fully-automated and part of today’s clinical practice. Therefore, the test does not require additional infrastructure building and lab personal training.”
Another notable advantage: the OncoSeek test only costs about $20, compared with other MCED tests, which can cost anywhere from $200 to $1000.
To validate the technology in a large, diverse cohort, Dr. Mao and colleagues enrolled approximately 10,000 participants, including 2003 cancer cases and 7888 non-cancer cases.
Peripheral blood was collected from each participant and analyzed using a panel of the seven protein tumor markers — AFP, CA125, CA15-3, CA19-9, CA72-4, CEA, and CYFRA 21-1.
To reduce the risk for false positive findings, the team designed the OncoSeek algorithm to achieve a specificity of 93%. Dr. Mao and colleagues found a sensitivity of 51.7%, resulting in an overall accuracy of 84.6%.
The performance was consistent in additional validation cohorts in Brazil, China, and the United States, with sensitivities ranging from 39.0% to 77.6% for detecting nine common cancer types, including breast, colorectal, liver, lung, lymphoma, esophagus, ovary, pancreas, and stomach. The sensitivity for pancreatic cancer was at the high end of 77.6%.
The test could predict the tissue of origin in about two thirds of cases.
Given its low cost, OncoSeek represents an affordable and accessible option for cancer screening, the authors concluded.
Overall, “I think MCEDs have the potential to enhance cancer screening,” Dr. Wood told this news organization.
Still, questions remain about the optimal use of these tests, such as whether they are best for average-risk or higher risk populations, and how to integrate them into standard screening, she said.
Dr. Wood also cautioned that the studies presented in the session represent early data, and it is likely that the numbers, such as sensitivity and specificity, will change with further prospective analyses.
And ultimately, these tests should complement, not replace, standard screening. “A negative testing should not be taken as a sign to avoid standard screening,” Dr. Wood said.
Dr. Yang is an employee of Geneseeq Technology, Inc., and Dr. Mao is an employee of SeekIn. Dr. Wood had no disclosures to report.
A version of this article appeared on Medscape.com.
Analyses presented during a session at the American Association for Cancer Research annual meeting, revealed that three new MCED tests — CanScan, MERCURY, and OncoSeek — could detect a range of cancers and recognize the tissue of origin with high accuracy. One — OncoSeek — could also provide an affordable cancer screening option for individuals living in lower-income countries.
The need for these noninvasive liquid biopsy tests that can accurately identify multiple cancer types with a single blood draw, especially cancers without routine screening strategies, is pressing. “We know that the current cancer standard of care screening will identify less than 50% of all cancers, while more than 50% of all cancer deaths occur in types of cancer with no recommended screening,” said co-moderator Marie E. Wood, MD, of the University of Colorado Anschutz Medical Campus, in Aurora, Colorado.
That being said, “the clinical utility of multicancer detection tests has not been established and we’re concerned about issues of overdiagnosis and overtreatment,” she noted.
The Early Data
One new MCED test called CanScan, developed by Geneseeq Technology, uses plasma cell-free DNA fragment patterns to detect cancer signals as well as identify the tissue of origin across 13 cancer types.
Overall, the CanScan test covers cancer types that contribute to two thirds of new cancer cases and 74% of morality globally, said presenter Shanshan Yang, of Geneseeq Research Institute, in Nanjing, China.
However, only five of these cancer types have screening recommendations issued by the US Preventive Services Task Force (USPSTF), Dr. Yang added.
The interim data comes from an ongoing large-scale prospective study evaluating the MCED test in a cohort of asymptomatic individuals between ages 45 and 75 years with an average risk for cancer and no cancer-related symptoms on enrollment.
Patients at baseline had their blood collected for the CanScan test and subsequently received annual routine physical exams once a year for 3 consecutive years, with an additional 2 years of follow-up.
The analysis included 3724 participants with analyzable samples at the data cutoff in September 2023. Among the 3724 participants, 29 had confirmed cancer diagnoses. Among these cases, 14 patients had their cancer confirmed through USPSTF recommended screening and 15 were detected through outside of standard USPSTF screening, such as a thyroid ultrasound, Dr. Yang explained.
Almost 90% of the cancers (26 of 29) were detected in the stage I or II, and eight (27.5%) were not one of the test’s 13 targeted cancer types.
The CanScan test had a sensitivity of 55.2%, identifying 16 of 29 of the patients with cancer, including 10 of 21 individuals with stage I (47.6%), and two of three with stage II (66.7%).
The test had a high specificity of 97.9%, meaning out of 100 people screened, only two had false negative findings.
Among the 15 patients who had their cancer detected outside of USPSTF screening recommendations, eight (53.3%) were found using a CanScan test, including patients with liver and endometrial cancers.
Compared with a positive predictive value of (PPV) of 1.6% with screening or physical exam methods alone, the CanScan test had a PPV of 17.4%, Dr. Yang reported.
“The MCED test holds significant potential for early cancer screening in asymptomatic populations,” Dr. Yang and colleagues concluded.
Another new MCED test called MERCURY, also developed by Geneseeq Technology and presented during the session, used a similar method to detect cancer signals and predict the tissue of origin across 13 cancer types.
The researchers initially validated the test using 3076 patients with cancer and 3477 healthy controls with a target specificity of 99%. In this group, researchers reported a sensitivity of 0.865 and a specificity of 0.989.
The team then performed an independent validation analysis with 1465 participants, 732 with cancer and 733 with no cancer, and confirmed a high sensitivity and specificity of 0.874 and 0.978, respectively. The sensitivity increased incrementally by cancer stage — 0.768 for stage I, 0.840 for stage II, 0.923 for stage III, and 0.971 for stage IV.
The test identified the tissue of origin with high accuracy, the researchers noted, but cautioned that the test needs “to be further validated in a prospective cohort study.”
MCED in Low-Income Settings
The session also featured findings on a new affordable MCED test called OncoSeek, which could provide greater access to cancer testing in low- and middle-income countries.
The OncoSeek algorithm identifies the presence of cancer using seven protein tumor markers alongside clinical information, such as gender and age. Like other tests, the test also predicts the possible tissue of origin.
The test can be run on clinical protein assay instruments that are already widely available, such as Roche cobas analyzer, Mao Mao, MD, PhD, the founder and CEO of SeekIn, of Shenzhen, China, told this news organization.
This “feature makes the test accessible worldwide, even in low- and middle-income countries,” he said. “These instruments are fully-automated and part of today’s clinical practice. Therefore, the test does not require additional infrastructure building and lab personal training.”
Another notable advantage: the OncoSeek test only costs about $20, compared with other MCED tests, which can cost anywhere from $200 to $1000.
To validate the technology in a large, diverse cohort, Dr. Mao and colleagues enrolled approximately 10,000 participants, including 2003 cancer cases and 7888 non-cancer cases.
Peripheral blood was collected from each participant and analyzed using a panel of the seven protein tumor markers — AFP, CA125, CA15-3, CA19-9, CA72-4, CEA, and CYFRA 21-1.
To reduce the risk for false positive findings, the team designed the OncoSeek algorithm to achieve a specificity of 93%. Dr. Mao and colleagues found a sensitivity of 51.7%, resulting in an overall accuracy of 84.6%.
The performance was consistent in additional validation cohorts in Brazil, China, and the United States, with sensitivities ranging from 39.0% to 77.6% for detecting nine common cancer types, including breast, colorectal, liver, lung, lymphoma, esophagus, ovary, pancreas, and stomach. The sensitivity for pancreatic cancer was at the high end of 77.6%.
The test could predict the tissue of origin in about two thirds of cases.
Given its low cost, OncoSeek represents an affordable and accessible option for cancer screening, the authors concluded.
Overall, “I think MCEDs have the potential to enhance cancer screening,” Dr. Wood told this news organization.
Still, questions remain about the optimal use of these tests, such as whether they are best for average-risk or higher risk populations, and how to integrate them into standard screening, she said.
Dr. Wood also cautioned that the studies presented in the session represent early data, and it is likely that the numbers, such as sensitivity and specificity, will change with further prospective analyses.
And ultimately, these tests should complement, not replace, standard screening. “A negative testing should not be taken as a sign to avoid standard screening,” Dr. Wood said.
Dr. Yang is an employee of Geneseeq Technology, Inc., and Dr. Mao is an employee of SeekIn. Dr. Wood had no disclosures to report.
A version of this article appeared on Medscape.com.
Analyses presented during a session at the American Association for Cancer Research annual meeting, revealed that three new MCED tests — CanScan, MERCURY, and OncoSeek — could detect a range of cancers and recognize the tissue of origin with high accuracy. One — OncoSeek — could also provide an affordable cancer screening option for individuals living in lower-income countries.
The need for these noninvasive liquid biopsy tests that can accurately identify multiple cancer types with a single blood draw, especially cancers without routine screening strategies, is pressing. “We know that the current cancer standard of care screening will identify less than 50% of all cancers, while more than 50% of all cancer deaths occur in types of cancer with no recommended screening,” said co-moderator Marie E. Wood, MD, of the University of Colorado Anschutz Medical Campus, in Aurora, Colorado.
That being said, “the clinical utility of multicancer detection tests has not been established and we’re concerned about issues of overdiagnosis and overtreatment,” she noted.
The Early Data
One new MCED test called CanScan, developed by Geneseeq Technology, uses plasma cell-free DNA fragment patterns to detect cancer signals as well as identify the tissue of origin across 13 cancer types.
Overall, the CanScan test covers cancer types that contribute to two thirds of new cancer cases and 74% of morality globally, said presenter Shanshan Yang, of Geneseeq Research Institute, in Nanjing, China.
However, only five of these cancer types have screening recommendations issued by the US Preventive Services Task Force (USPSTF), Dr. Yang added.
The interim data comes from an ongoing large-scale prospective study evaluating the MCED test in a cohort of asymptomatic individuals between ages 45 and 75 years with an average risk for cancer and no cancer-related symptoms on enrollment.
Patients at baseline had their blood collected for the CanScan test and subsequently received annual routine physical exams once a year for 3 consecutive years, with an additional 2 years of follow-up.
The analysis included 3724 participants with analyzable samples at the data cutoff in September 2023. Among the 3724 participants, 29 had confirmed cancer diagnoses. Among these cases, 14 patients had their cancer confirmed through USPSTF recommended screening and 15 were detected through outside of standard USPSTF screening, such as a thyroid ultrasound, Dr. Yang explained.
Almost 90% of the cancers (26 of 29) were detected in the stage I or II, and eight (27.5%) were not one of the test’s 13 targeted cancer types.
The CanScan test had a sensitivity of 55.2%, identifying 16 of 29 of the patients with cancer, including 10 of 21 individuals with stage I (47.6%), and two of three with stage II (66.7%).
The test had a high specificity of 97.9%, meaning out of 100 people screened, only two had false negative findings.
Among the 15 patients who had their cancer detected outside of USPSTF screening recommendations, eight (53.3%) were found using a CanScan test, including patients with liver and endometrial cancers.
Compared with a positive predictive value of (PPV) of 1.6% with screening or physical exam methods alone, the CanScan test had a PPV of 17.4%, Dr. Yang reported.
“The MCED test holds significant potential for early cancer screening in asymptomatic populations,” Dr. Yang and colleagues concluded.
Another new MCED test called MERCURY, also developed by Geneseeq Technology and presented during the session, used a similar method to detect cancer signals and predict the tissue of origin across 13 cancer types.
The researchers initially validated the test using 3076 patients with cancer and 3477 healthy controls with a target specificity of 99%. In this group, researchers reported a sensitivity of 0.865 and a specificity of 0.989.
The team then performed an independent validation analysis with 1465 participants, 732 with cancer and 733 with no cancer, and confirmed a high sensitivity and specificity of 0.874 and 0.978, respectively. The sensitivity increased incrementally by cancer stage — 0.768 for stage I, 0.840 for stage II, 0.923 for stage III, and 0.971 for stage IV.
The test identified the tissue of origin with high accuracy, the researchers noted, but cautioned that the test needs “to be further validated in a prospective cohort study.”
MCED in Low-Income Settings
The session also featured findings on a new affordable MCED test called OncoSeek, which could provide greater access to cancer testing in low- and middle-income countries.
The OncoSeek algorithm identifies the presence of cancer using seven protein tumor markers alongside clinical information, such as gender and age. Like other tests, the test also predicts the possible tissue of origin.
The test can be run on clinical protein assay instruments that are already widely available, such as Roche cobas analyzer, Mao Mao, MD, PhD, the founder and CEO of SeekIn, of Shenzhen, China, told this news organization.
This “feature makes the test accessible worldwide, even in low- and middle-income countries,” he said. “These instruments are fully-automated and part of today’s clinical practice. Therefore, the test does not require additional infrastructure building and lab personal training.”
Another notable advantage: the OncoSeek test only costs about $20, compared with other MCED tests, which can cost anywhere from $200 to $1000.
To validate the technology in a large, diverse cohort, Dr. Mao and colleagues enrolled approximately 10,000 participants, including 2003 cancer cases and 7888 non-cancer cases.
Peripheral blood was collected from each participant and analyzed using a panel of the seven protein tumor markers — AFP, CA125, CA15-3, CA19-9, CA72-4, CEA, and CYFRA 21-1.
To reduce the risk for false positive findings, the team designed the OncoSeek algorithm to achieve a specificity of 93%. Dr. Mao and colleagues found a sensitivity of 51.7%, resulting in an overall accuracy of 84.6%.
The performance was consistent in additional validation cohorts in Brazil, China, and the United States, with sensitivities ranging from 39.0% to 77.6% for detecting nine common cancer types, including breast, colorectal, liver, lung, lymphoma, esophagus, ovary, pancreas, and stomach. The sensitivity for pancreatic cancer was at the high end of 77.6%.
The test could predict the tissue of origin in about two thirds of cases.
Given its low cost, OncoSeek represents an affordable and accessible option for cancer screening, the authors concluded.
Overall, “I think MCEDs have the potential to enhance cancer screening,” Dr. Wood told this news organization.
Still, questions remain about the optimal use of these tests, such as whether they are best for average-risk or higher risk populations, and how to integrate them into standard screening, she said.
Dr. Wood also cautioned that the studies presented in the session represent early data, and it is likely that the numbers, such as sensitivity and specificity, will change with further prospective analyses.
And ultimately, these tests should complement, not replace, standard screening. “A negative testing should not be taken as a sign to avoid standard screening,” Dr. Wood said.
Dr. Yang is an employee of Geneseeq Technology, Inc., and Dr. Mao is an employee of SeekIn. Dr. Wood had no disclosures to report.
A version of this article appeared on Medscape.com.
Oncologists Voice Ethical Concerns Over AI in Cancer Care
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.
TOPLINE:
Most respondents, for instance, said patients should not be expected to understand how AI tools work, but many also felt patients could make treatment decisions based on AI-generated recommendations. Most oncologists also felt responsible for protecting patients from biased AI, but few were confident that they could do so.
METHODOLOGY:
- The US Food and Drug Administration (FDA) has for use in various medical specialties over the past few decades, and increasingly, AI tools are being integrated into cancer care.
- However, the uptake of these tools in oncology has raised ethical questions and concerns, including challenges with AI bias, error, or misuse, as well as issues explaining how an AI model reached a result.
- In the current study, researchers asked 204 oncologists from 37 states for their views on the ethical implications of using AI for cancer care.
- Among the survey respondents, 64% were men and 63% were non-Hispanic White; 29% were from academic practices, 47% had received some education on AI use in healthcare, and 45% were familiar with clinical decision models.
- The researchers assessed respondents’ answers to various questions, including whether to provide informed consent for AI use and how oncologists would approach a scenario where the AI model and the oncologist recommended a different treatment regimen.
TAKEAWAY:
- Overall, 81% of oncologists supported having patient consent to use an AI model during treatment decisions, and 85% felt that oncologists needed to be able to explain an AI-based clinical decision model to use it in the clinic; however, only 23% felt that patients also needed to be able to explain an AI model.
- When an AI decision model recommended a different treatment regimen than the treating oncologist, the most common response (36.8%) was to present both options to the patient and let the patient decide. Oncologists from academic settings were about 2.5 times more likely than those from other settings to let the patient decide. About 34% of respondents said they would present both options but recommend the oncologist’s regimen, whereas about 22% said they would present both but recommend the AI’s regimen. A small percentage would only present the oncologist’s regimen (5%) or the AI’s regimen (about 2.5%).
- About three of four respondents (76.5%) agreed that oncologists should protect patients from biased AI tools; however, only about one of four (27.9%) felt confident they could identify biased AI models.
- Most oncologists (91%) felt that AI developers were responsible for the medico-legal problems associated with AI use; less than half (47%) said oncologists or hospitals (43%) shared this responsibility.
IN PRACTICE:
“Together, these data characterize barriers that may impede the ethical adoption of AI into cancer care. The findings suggest that the implementation of AI in oncology must include rigorous assessments of its effect on care decisions, as well as decisional responsibility when problems related to AI use arise,” the authors concluded.
SOURCE:
The study, with first author Andrew Hantel, MD, from Dana-Farber Cancer Institute, Boston, was published last month in JAMA Network Open.
LIMITATIONS:
The study had a moderate sample size and response rate, although demographics of participating oncologists appear to be nationally representative. The cross-sectional study design limited the generalizability of the findings over time as AI is integrated into cancer care.
DISCLOSURES:
The study was funded by the National Cancer Institute, the Dana-Farber McGraw/Patterson Research Fund, and the Mark Foundation Emerging Leader Award. Dr. Hantel reported receiving personal fees from AbbVie, AstraZeneca, the American Journal of Managed Care, Genentech, and GSK.
A version of this article appeared on Medscape.com.

