User login
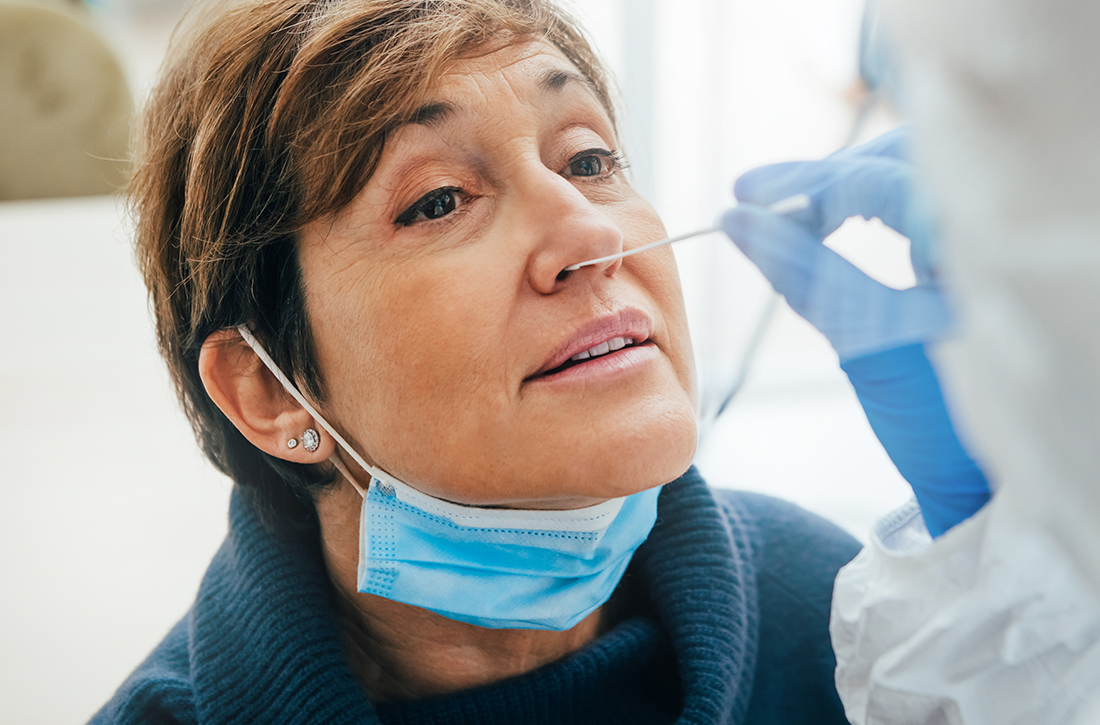
55-year-old woman • unilateral nasal drainage • salty taste • nasal redness • recent COVID-19 nasal swabs • Dx?
THE CASE
A 55-year-old woman was evaluated in a family medicine clinic for clear, right-side nasal drainage. She stated that the drainage began 5 months earlier after 2 hospitalizations for severe anxiety leading to emesis and hypokalemia. She reported 3 different COVID-19 nasal swab tests performed on the right nare. Chart review showed 2 negative COVID-19 tests, 6 days apart. Since the hospitalizations, the patient had been given antihistamines for rhinorrhea at an urgent care visit. Despite this treatment, the patient reported a constant drip from the right nare with a salty taste. She also reported experiencing occasional headaches but denied nausea/vomiting.
The patient’s history included uncontrolled hypertension, treatment-resistant anxiety and depression, obstructive sleep apnea, chronic sinus disease (observed on computed tomography [CT] scans), and type 2 diabetes. She was on amlodipine 10 mg/d for hypertension and was not taking any medication for diabetes.
On examination, the patient’s vital signs were within normal limits except for an elevated blood pressure of 158/88 mm Hg. The patient had persistent clear rhinorrhea fluid draining from the right nostril that was exacerbated when she looked down. Right nasal erythema was present.
THE DIAGNOSIS
The patient’s negative COVID-19 tests, lack of improvement on antihistamines, and description of the nasal fluid as salty tasting prompted us to suspect a cerebrospinal fluid (CSF) leak. The clinical work-up included a halo (“double-ring”) sign test, a β-2 transferrin test, and a sinus x-ray.
The halo sign test was negative for CSF fluid. Sinus/skull x-ray did not show a cribriform or other fracture. However, a sample of the nasal fluid collected in a sterile container was positive for β-2 transferrin, the gold-standard laboratory test to confirm a CSF leak.
The patient was sent for a maxillofacial CT scan without contrast. Results showed a 3-mm defect over the right ethmoid roof associated with a 10 × 16–mm low-attenuation structure in the right ethmoid labyrinth, suspicious for encephalocele. This defect, in the setting of the patient’s history of chronic sinus disease, furthered our suspicion of a CSF leak secondary to COVID-19 testing. Radiology confirmed the diagnosis.
DISCUSSION
CSF rhinorrhea is CSF leakage through the nasal cavity due to abnormal communication between the arachnoid membrane and nasal mucosa.1 The most commonly reported risk factors for this include female sex, middle age (fourth to fifth decade), obesity (body mass index > 40), intracranial hypertension, and obstructive sleep apnea.1,2
Continue to: Clear, unilateral rhinorrhea...
Clear, unilateral rhinorrhea drainage that increases at times of relatively increased intracranial pressure and has a metallic or salty taste is suspicious for CSF rhinorrhea.3 It can occur following skull‐base trauma (eg, cribriform plate, temporal bone), endoscopic sinus surgery, or neurosurgical procedures, or have a spontaneous etiology.3,4
Modalities to confirm CSF rhinorrhea include radionuclide cisternography and testing of fluid for the halo sign, glucose, and the CSF-specific proteins β‐2 transferrin and β-trace protein.3,4 High‐resolution CT is the imaging method most commonly used for localizing a CSF leak.4
Treatment is provided in the hospital
Patients with CSF rhinorrhea typically require inpatient management with bed rest, head-of-bed elevation, and frequent neurologic evaluation, as persistent CSF rhinorrhea increases the risk for meningitis, thus necessitating surgical intervention.3,5 Some cases resolve with bed rest alone. Endonasal endoscopic repair of CSF leaks has become the standard of care because of its high success rate and lower morbidity profile.4
The preferred treatment method for encephalocele is surgical removal after diagnosis is confirmed with CT or magnetic resonance imaging.6
Our patient underwent surgery to remove the encephalocele. The surgeons reported no evidence of fracture.
The final cause of her CSF leak is still uncertain. The surgeons felt confident it was due to ethmoidal encephalocele, a form of neural tube defect in which brain tissue herniates through structural weaknesses of the skull.6-8 While more common in infants, encephalocele can manifest in adulthood due to traumatic or iatrogenic causes.7,8
There is a previous report of encephalocele with CSF leak after COVID-19 testing.9 This case report suggests the possibility of a nasal swab causing trauma to a patient’s pre‐existing encephalocele—a probability in our patient’s case. It is unlikely, however, that the nasal swab itself violated the bony skull base.
THE TAKEAWAY
This case exemplifies how unexplained local symptoms, a high index of suspicion, and adequate work-up can lead to a rare diagnosis. Diagnostic strategies employed for cases of CSF rhinorrhea vary widely due to limited evidence-based guidance.4 Unilateral rhinorrhea with clear fluid that increases at times of increased intracranial pressure, such as bending over, should prompt suspicion for CSF rhinorrhea. With millions of people getting nasal swabs daily during the COVID-19 pandemic, it is even more important to keep CSF leak in our differential diagnosis.
CORRESPONDENCE
Eliana Lizeth Garcia, MD, BS, BA, University of New Mexico Health Sciences Center, 1209 University Boulevard NE, Albuquerque, NM 87131-5001; [email protected]
1. Keshri A, Jain R, Manogaran RS, et al. Management of spontaneous CSF rhinorrhea: an institutional experience. J Neurol Surg B Skull Base. 2019;80:493-499. doi: 10.1055/s-0038-1676334
2. Lobo BC, Baumanis MM, Nelson RF. Surgical repair of spontaneous cerebrospinal fluid (CSF) leaks: a systematic review. Laryngoscope Investig Otolaryngol. 2017;2:215-224. doi: 10.1002/lio2.75
3. Van Zele T, Dewaele F. Traumatic CSF leaks of the anterior skull base. B-ENT. 2016;suppl 26:19-27.
4. Oakley GM, Alt JA, Schlosser RJ, et al. Diagnosis of cerebrospinal fluid rhinorrhea: an evidence-based review with recommendations. Int Forum Allergy Rhinol. 2016;6:8-16. doi: 10.1002/alr.21637
5. Friedman JA, Ebersold MJ, Quast LM. Post-traumatic cerebrospinal fluid leakage. World J Surg. 2001;25:1062-1066. doi: 10.1007/s00268-001-0059-7
6. Tirumandas M, Sharma A, Gbenimacho I, et al. Nasal encephaloceles: a review of etiology, pathophysiology, clinical presentations, diagnosis, treatment, and complications. Childs Nerv Syst. 2013;29:739-744. doi: 10.1007/s00381-012-1998-z
7. Junaid M, Sobani ZU, Shamim AA, et al. Nasal encephaloceles presenting at later ages: experience of otorhinolaryngology department at a tertiary care center in Karachi, Pakistan. J Pak Med Assoc. 2012;62:74-76.
8. Dhirawani RB, Gupta R, Pathak S, et al. Frontoethmoidal encephalocele: case report and review on management. Ann Maxillofac Surg. 2014;4:195-197. doi: 10.4103/2231-0746.147140
9. Paquin R, Ryan L, Vale FL, et al. CSF leak after COVID-19 nasopharyngeal swab: a case report. Laryngoscope. 2021;131:1927-1929. doi: 10.1002/lary.29462
THE CASE
A 55-year-old woman was evaluated in a family medicine clinic for clear, right-side nasal drainage. She stated that the drainage began 5 months earlier after 2 hospitalizations for severe anxiety leading to emesis and hypokalemia. She reported 3 different COVID-19 nasal swab tests performed on the right nare. Chart review showed 2 negative COVID-19 tests, 6 days apart. Since the hospitalizations, the patient had been given antihistamines for rhinorrhea at an urgent care visit. Despite this treatment, the patient reported a constant drip from the right nare with a salty taste. She also reported experiencing occasional headaches but denied nausea/vomiting.
The patient’s history included uncontrolled hypertension, treatment-resistant anxiety and depression, obstructive sleep apnea, chronic sinus disease (observed on computed tomography [CT] scans), and type 2 diabetes. She was on amlodipine 10 mg/d for hypertension and was not taking any medication for diabetes.
On examination, the patient’s vital signs were within normal limits except for an elevated blood pressure of 158/88 mm Hg. The patient had persistent clear rhinorrhea fluid draining from the right nostril that was exacerbated when she looked down. Right nasal erythema was present.
THE DIAGNOSIS
The patient’s negative COVID-19 tests, lack of improvement on antihistamines, and description of the nasal fluid as salty tasting prompted us to suspect a cerebrospinal fluid (CSF) leak. The clinical work-up included a halo (“double-ring”) sign test, a β-2 transferrin test, and a sinus x-ray.
The halo sign test was negative for CSF fluid. Sinus/skull x-ray did not show a cribriform or other fracture. However, a sample of the nasal fluid collected in a sterile container was positive for β-2 transferrin, the gold-standard laboratory test to confirm a CSF leak.
The patient was sent for a maxillofacial CT scan without contrast. Results showed a 3-mm defect over the right ethmoid roof associated with a 10 × 16–mm low-attenuation structure in the right ethmoid labyrinth, suspicious for encephalocele. This defect, in the setting of the patient’s history of chronic sinus disease, furthered our suspicion of a CSF leak secondary to COVID-19 testing. Radiology confirmed the diagnosis.
DISCUSSION
CSF rhinorrhea is CSF leakage through the nasal cavity due to abnormal communication between the arachnoid membrane and nasal mucosa.1 The most commonly reported risk factors for this include female sex, middle age (fourth to fifth decade), obesity (body mass index > 40), intracranial hypertension, and obstructive sleep apnea.1,2
Continue to: Clear, unilateral rhinorrhea...
Clear, unilateral rhinorrhea drainage that increases at times of relatively increased intracranial pressure and has a metallic or salty taste is suspicious for CSF rhinorrhea.3 It can occur following skull‐base trauma (eg, cribriform plate, temporal bone), endoscopic sinus surgery, or neurosurgical procedures, or have a spontaneous etiology.3,4
Modalities to confirm CSF rhinorrhea include radionuclide cisternography and testing of fluid for the halo sign, glucose, and the CSF-specific proteins β‐2 transferrin and β-trace protein.3,4 High‐resolution CT is the imaging method most commonly used for localizing a CSF leak.4
Treatment is provided in the hospital
Patients with CSF rhinorrhea typically require inpatient management with bed rest, head-of-bed elevation, and frequent neurologic evaluation, as persistent CSF rhinorrhea increases the risk for meningitis, thus necessitating surgical intervention.3,5 Some cases resolve with bed rest alone. Endonasal endoscopic repair of CSF leaks has become the standard of care because of its high success rate and lower morbidity profile.4
The preferred treatment method for encephalocele is surgical removal after diagnosis is confirmed with CT or magnetic resonance imaging.6
Our patient underwent surgery to remove the encephalocele. The surgeons reported no evidence of fracture.
The final cause of her CSF leak is still uncertain. The surgeons felt confident it was due to ethmoidal encephalocele, a form of neural tube defect in which brain tissue herniates through structural weaknesses of the skull.6-8 While more common in infants, encephalocele can manifest in adulthood due to traumatic or iatrogenic causes.7,8
There is a previous report of encephalocele with CSF leak after COVID-19 testing.9 This case report suggests the possibility of a nasal swab causing trauma to a patient’s pre‐existing encephalocele—a probability in our patient’s case. It is unlikely, however, that the nasal swab itself violated the bony skull base.
THE TAKEAWAY
This case exemplifies how unexplained local symptoms, a high index of suspicion, and adequate work-up can lead to a rare diagnosis. Diagnostic strategies employed for cases of CSF rhinorrhea vary widely due to limited evidence-based guidance.4 Unilateral rhinorrhea with clear fluid that increases at times of increased intracranial pressure, such as bending over, should prompt suspicion for CSF rhinorrhea. With millions of people getting nasal swabs daily during the COVID-19 pandemic, it is even more important to keep CSF leak in our differential diagnosis.
CORRESPONDENCE
Eliana Lizeth Garcia, MD, BS, BA, University of New Mexico Health Sciences Center, 1209 University Boulevard NE, Albuquerque, NM 87131-5001; [email protected]
THE CASE
A 55-year-old woman was evaluated in a family medicine clinic for clear, right-side nasal drainage. She stated that the drainage began 5 months earlier after 2 hospitalizations for severe anxiety leading to emesis and hypokalemia. She reported 3 different COVID-19 nasal swab tests performed on the right nare. Chart review showed 2 negative COVID-19 tests, 6 days apart. Since the hospitalizations, the patient had been given antihistamines for rhinorrhea at an urgent care visit. Despite this treatment, the patient reported a constant drip from the right nare with a salty taste. She also reported experiencing occasional headaches but denied nausea/vomiting.
The patient’s history included uncontrolled hypertension, treatment-resistant anxiety and depression, obstructive sleep apnea, chronic sinus disease (observed on computed tomography [CT] scans), and type 2 diabetes. She was on amlodipine 10 mg/d for hypertension and was not taking any medication for diabetes.
On examination, the patient’s vital signs were within normal limits except for an elevated blood pressure of 158/88 mm Hg. The patient had persistent clear rhinorrhea fluid draining from the right nostril that was exacerbated when she looked down. Right nasal erythema was present.
THE DIAGNOSIS
The patient’s negative COVID-19 tests, lack of improvement on antihistamines, and description of the nasal fluid as salty tasting prompted us to suspect a cerebrospinal fluid (CSF) leak. The clinical work-up included a halo (“double-ring”) sign test, a β-2 transferrin test, and a sinus x-ray.
The halo sign test was negative for CSF fluid. Sinus/skull x-ray did not show a cribriform or other fracture. However, a sample of the nasal fluid collected in a sterile container was positive for β-2 transferrin, the gold-standard laboratory test to confirm a CSF leak.
The patient was sent for a maxillofacial CT scan without contrast. Results showed a 3-mm defect over the right ethmoid roof associated with a 10 × 16–mm low-attenuation structure in the right ethmoid labyrinth, suspicious for encephalocele. This defect, in the setting of the patient’s history of chronic sinus disease, furthered our suspicion of a CSF leak secondary to COVID-19 testing. Radiology confirmed the diagnosis.
DISCUSSION
CSF rhinorrhea is CSF leakage through the nasal cavity due to abnormal communication between the arachnoid membrane and nasal mucosa.1 The most commonly reported risk factors for this include female sex, middle age (fourth to fifth decade), obesity (body mass index > 40), intracranial hypertension, and obstructive sleep apnea.1,2
Continue to: Clear, unilateral rhinorrhea...
Clear, unilateral rhinorrhea drainage that increases at times of relatively increased intracranial pressure and has a metallic or salty taste is suspicious for CSF rhinorrhea.3 It can occur following skull‐base trauma (eg, cribriform plate, temporal bone), endoscopic sinus surgery, or neurosurgical procedures, or have a spontaneous etiology.3,4
Modalities to confirm CSF rhinorrhea include radionuclide cisternography and testing of fluid for the halo sign, glucose, and the CSF-specific proteins β‐2 transferrin and β-trace protein.3,4 High‐resolution CT is the imaging method most commonly used for localizing a CSF leak.4
Treatment is provided in the hospital
Patients with CSF rhinorrhea typically require inpatient management with bed rest, head-of-bed elevation, and frequent neurologic evaluation, as persistent CSF rhinorrhea increases the risk for meningitis, thus necessitating surgical intervention.3,5 Some cases resolve with bed rest alone. Endonasal endoscopic repair of CSF leaks has become the standard of care because of its high success rate and lower morbidity profile.4
The preferred treatment method for encephalocele is surgical removal after diagnosis is confirmed with CT or magnetic resonance imaging.6
Our patient underwent surgery to remove the encephalocele. The surgeons reported no evidence of fracture.
The final cause of her CSF leak is still uncertain. The surgeons felt confident it was due to ethmoidal encephalocele, a form of neural tube defect in which brain tissue herniates through structural weaknesses of the skull.6-8 While more common in infants, encephalocele can manifest in adulthood due to traumatic or iatrogenic causes.7,8
There is a previous report of encephalocele with CSF leak after COVID-19 testing.9 This case report suggests the possibility of a nasal swab causing trauma to a patient’s pre‐existing encephalocele—a probability in our patient’s case. It is unlikely, however, that the nasal swab itself violated the bony skull base.
THE TAKEAWAY
This case exemplifies how unexplained local symptoms, a high index of suspicion, and adequate work-up can lead to a rare diagnosis. Diagnostic strategies employed for cases of CSF rhinorrhea vary widely due to limited evidence-based guidance.4 Unilateral rhinorrhea with clear fluid that increases at times of increased intracranial pressure, such as bending over, should prompt suspicion for CSF rhinorrhea. With millions of people getting nasal swabs daily during the COVID-19 pandemic, it is even more important to keep CSF leak in our differential diagnosis.
CORRESPONDENCE
Eliana Lizeth Garcia, MD, BS, BA, University of New Mexico Health Sciences Center, 1209 University Boulevard NE, Albuquerque, NM 87131-5001; [email protected]
1. Keshri A, Jain R, Manogaran RS, et al. Management of spontaneous CSF rhinorrhea: an institutional experience. J Neurol Surg B Skull Base. 2019;80:493-499. doi: 10.1055/s-0038-1676334
2. Lobo BC, Baumanis MM, Nelson RF. Surgical repair of spontaneous cerebrospinal fluid (CSF) leaks: a systematic review. Laryngoscope Investig Otolaryngol. 2017;2:215-224. doi: 10.1002/lio2.75
3. Van Zele T, Dewaele F. Traumatic CSF leaks of the anterior skull base. B-ENT. 2016;suppl 26:19-27.
4. Oakley GM, Alt JA, Schlosser RJ, et al. Diagnosis of cerebrospinal fluid rhinorrhea: an evidence-based review with recommendations. Int Forum Allergy Rhinol. 2016;6:8-16. doi: 10.1002/alr.21637
5. Friedman JA, Ebersold MJ, Quast LM. Post-traumatic cerebrospinal fluid leakage. World J Surg. 2001;25:1062-1066. doi: 10.1007/s00268-001-0059-7
6. Tirumandas M, Sharma A, Gbenimacho I, et al. Nasal encephaloceles: a review of etiology, pathophysiology, clinical presentations, diagnosis, treatment, and complications. Childs Nerv Syst. 2013;29:739-744. doi: 10.1007/s00381-012-1998-z
7. Junaid M, Sobani ZU, Shamim AA, et al. Nasal encephaloceles presenting at later ages: experience of otorhinolaryngology department at a tertiary care center in Karachi, Pakistan. J Pak Med Assoc. 2012;62:74-76.
8. Dhirawani RB, Gupta R, Pathak S, et al. Frontoethmoidal encephalocele: case report and review on management. Ann Maxillofac Surg. 2014;4:195-197. doi: 10.4103/2231-0746.147140
9. Paquin R, Ryan L, Vale FL, et al. CSF leak after COVID-19 nasopharyngeal swab: a case report. Laryngoscope. 2021;131:1927-1929. doi: 10.1002/lary.29462
1. Keshri A, Jain R, Manogaran RS, et al. Management of spontaneous CSF rhinorrhea: an institutional experience. J Neurol Surg B Skull Base. 2019;80:493-499. doi: 10.1055/s-0038-1676334
2. Lobo BC, Baumanis MM, Nelson RF. Surgical repair of spontaneous cerebrospinal fluid (CSF) leaks: a systematic review. Laryngoscope Investig Otolaryngol. 2017;2:215-224. doi: 10.1002/lio2.75
3. Van Zele T, Dewaele F. Traumatic CSF leaks of the anterior skull base. B-ENT. 2016;suppl 26:19-27.
4. Oakley GM, Alt JA, Schlosser RJ, et al. Diagnosis of cerebrospinal fluid rhinorrhea: an evidence-based review with recommendations. Int Forum Allergy Rhinol. 2016;6:8-16. doi: 10.1002/alr.21637
5. Friedman JA, Ebersold MJ, Quast LM. Post-traumatic cerebrospinal fluid leakage. World J Surg. 2001;25:1062-1066. doi: 10.1007/s00268-001-0059-7
6. Tirumandas M, Sharma A, Gbenimacho I, et al. Nasal encephaloceles: a review of etiology, pathophysiology, clinical presentations, diagnosis, treatment, and complications. Childs Nerv Syst. 2013;29:739-744. doi: 10.1007/s00381-012-1998-z
7. Junaid M, Sobani ZU, Shamim AA, et al. Nasal encephaloceles presenting at later ages: experience of otorhinolaryngology department at a tertiary care center in Karachi, Pakistan. J Pak Med Assoc. 2012;62:74-76.
8. Dhirawani RB, Gupta R, Pathak S, et al. Frontoethmoidal encephalocele: case report and review on management. Ann Maxillofac Surg. 2014;4:195-197. doi: 10.4103/2231-0746.147140
9. Paquin R, Ryan L, Vale FL, et al. CSF leak after COVID-19 nasopharyngeal swab: a case report. Laryngoscope. 2021;131:1927-1929. doi: 10.1002/lary.29462
► Unilateral nasal drainage
► Salty taste
► Nasal redness
► Recent COVID-19 nasal swabs
Caring for the caregiver in dementia
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-
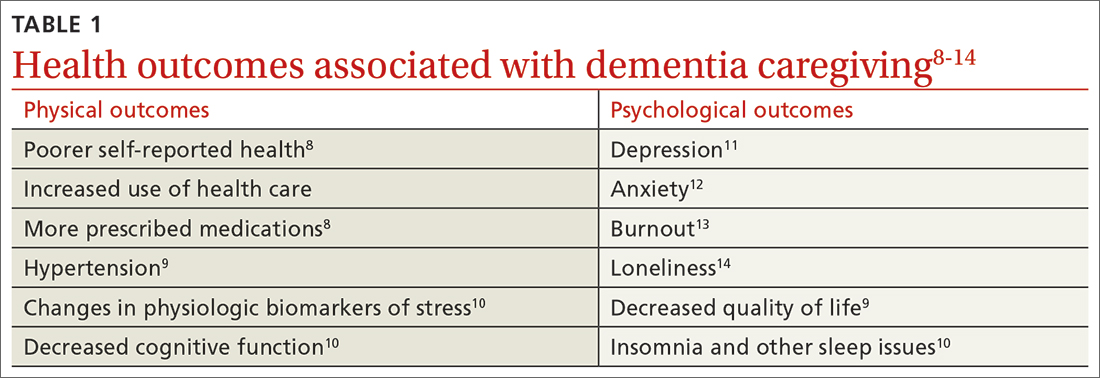
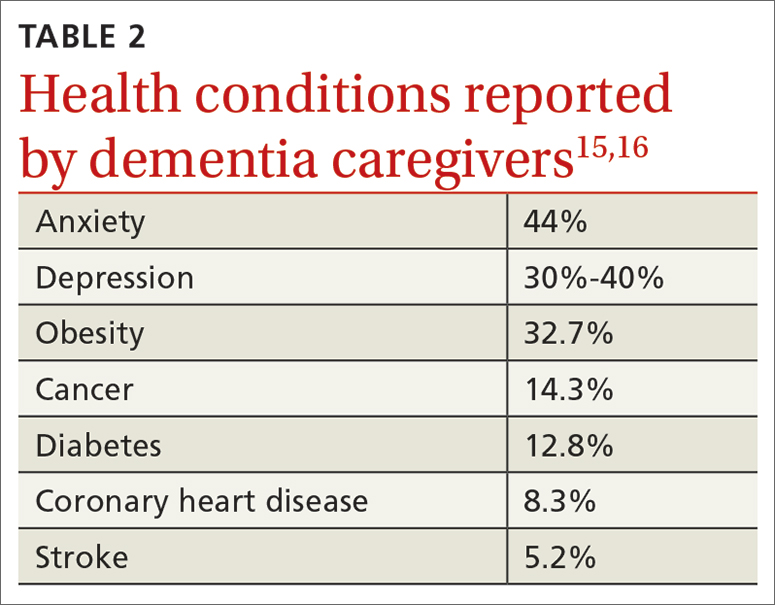
Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17
Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-

Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17

Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
THE CASE
Sam C* is a 68-year-old man who presented to his family physician in a rural health clinic due to concerns about weight loss. Since his visit 8 months prior, Mr. C unintentionally had lost 20 pounds. Upon questioning, Mr. C also reported feeling irritable and having difficulty with sleep and concentration.
A review of systems did not indicate the presence of infection or other medical conditions. In the 6 years since becoming a patient to the practice, he had reported no chronic health concerns, was taking no medications, and had only been to the clinic for his annual check-up appointments. He completed a Patient Health Questionnaire (PHQ-9) and scored 18, indicating moderately severe depression.
Mr. C had established care with his physician when he moved to the area from out of state so that he could be closer to his parents, who were in their mid-80s at the time. Mr. C’s physician also had been the family physician for his parents for the previous 20 years. Three years prior to Mr. C’s presentation for weight loss, his mother had received a diagnosis of acute leukemia; she died a year later.
Over the past year, Mr. C had needed to take a more active role in the care of his father, who was now in his early 90s. Mr. C’s father, who was previously in excellent health, had begun to develop significant health problems, including degenerative arthritis and progressive vascular dementia. He also had ataxia, leading to poor mobility, and a neurogenic bladder requiring self-catheterization, which required Mr. C’s assistance. Mr. C lived next door to his father and provided frequent assistance with activities of daily living. However, his father, who always had been the dominant figure in the family, was determined to maintain his independence and not relinquish control to others.
The strain of caregiving activities, along with managing his father’s inflexibility, was causing increasing distress for Mr. C. As he told his family physician, “I just don’t know what to do.”
●
* The patient’s name has been changed to protect his identity.
It is estimated that more than 11 million Americans provided more than 18 billion hours in unpaid support for individuals with dementia in 2022, averaging 30 hours of care per caregiver per week.1 As individuals with dementia progressively decline, they require increased assistance with activities of daily living (ADLs, such as bathing and dressing) and instrumental activities of daily living (IADLs, such as paying bills and using transportation). Most of this assistance comes from informal caregiving provided by family members and friends.
Caregiver burden can be defined as “the strain or load borne by a person who cares for a chronically ill, disabled, or elderly family member.”2 Caregiver stress has been found to be higher for dementia caregiving than other types of caregiving.3 In particular, caring for someone with greater behavioral and psychological symptoms of dementia (BPSDs) has been associated with higher caregiver burden.4-

Beyond the subjective burden of caregiving, there are other potential negative consequences for dementia caregivers (see TABLE 18-14 and TABLE 215,16). In addition, caregiver distress is related to a number of care recipient outcomes, including earlier institutionalization, more hospitalizations, more BPSDs, poorer quality of life, and greater likelihood of experiencing elder abuse.17

Assessment, reassessment are key to meeting needs
Numerous factors can foster caregiver well-being, including feelings of accomplishment and contribution, a strengthening of the relationship with the care recipient, and feeling supported by friends, family, and formal care systems.18,19 Family physicians can play an important role by assessing and supporting patients with dementia and their caregivers. Ideally, the individual with dementia and the caregiver will be assessed both together and separately.
A thorough assessment includes gathering information about the context and quality of the caregiving relationship; caregiver perception of the care recipient’s health and functional status; caregiver values and preferences; caregiver well-being (including mental health assessment); caregiver skills, abilities, and knowledge about caregiving; and positive and negative consequences of caregiving.20 Caregiver needs—including informational, care support, emotional, physical, and social needs—also should be assessed.
Continue to: Tools are available...
Tools are available to facilitate caregiver assessment. For example, the Zarit Burden Interview is a 22-item self-report measure that can be given to the caregiver21; shorter versions (4 and 12 items) are also available.22 Another resource available for caregiver assessment guidance is a toolkit developed by the Family Caregiver Alliance.20
Continually assess for changing needs
As the condition of the individual with dementia progresses, it will be important to reassess the caregiver, as stressors and needs will change over the course of the caregiving relationship. Support should be adapted accordingly.
In the early stage of dementia, caregivers may need information on disease progression and dementia care planning, ways to navigate the health care system, financial planning, and useful resources. Caregivers also may need emotional support to help them adapt to the role of caregiver, deal with denial, and manage their stress.23,24
With dementia progression, caregivers may need support related to increased decision-making responsibility, managing challenging behaviors, assisting with ADLs and IADLs, and identifying opportunities to meet personal social and well-being needs. They also may need support to accept the changes they are seeing in the individual with dementia and the shifts they are experiencing in their relationship with him or her.23,25
In late-stage dementia, caregiver needs tend to shift to determining the need for long-term care placement vs staying at home, end-of-life planning, loneliness, and anticipatory grief.23,26 Support with managing changing and accumulating stress typically remains a primary need throughout the progression of dementia.27
Continue to: Specific populations have distinct needs
Specific populations have distinct needs. Some caregivers, including members of the LGBTQ+ community and different racial and ethnic groups, as well as caregivers of people with younger-onset dementia, may have additional support needs.28
For example, African American and Latino caregivers tend to have caregiving relationships of longer duration, requiring more time-intensive care, but use fewer formal support services than White caregivers.29 Caregivers from non-White racial and ethnic groups also are more likely to experience discrimination when interacting with health care services on behalf of care recipients.30
Having an awareness of potential specialized needs may help to prevent or address potential care disparities, and cultural humility may help to improve caregiver experiences with primary care physicians.
Resources to support caregivers
Family physicians are well situated to provide informational and emotional support for both patients with dementia and their informal care providers.31 Given the variability of caregiver concerns, multicomponent interventions addressing informational, self-care, social support, and financial needs often are needed.31 Supportive counseling and psychoeducation can help dementia caregivers with stress management, self-care, coping, and skills training—supporting the development of self-efficacy.32,33
Outside resources. Although significant caregiver support can be provided directly by the physician, caregivers should be connected with outside resources, including support groups, counselors, psychotherapists, financial and legal support, and formal care services
Continue to: Psychosocial and complementary interventions
Psychosocial and complementary interventions. Various psychosocial interventions (eg, psychoeducation, cognitive behavioral therapy, support groups) have been found to be beneficial in alleviating caregiver symptoms of depression, anxiety, and stress and improving well-being, perceived burden, and quality of life. However, systematic reviews have found variability in the degree of helpfulness of these interventions.35,36
Some caregivers and care recipients may benefit from complementary and integrative medicine referrals. Mind–body therapies such as mindfulness, yoga, and Tai Chi have shown some beneficial effects.37
Online resources. Caregivers also can be directed to online resources from organizations such as the Alzheimer’s Association (www.alz.org), the National Institutes of Health (www.alzheimers.gov), and the Family Caregiver Alliance (www.caregiver.org).
In rural settings, such as the one in which this case took place, online resources may decrease some barriers to supporting caregivers.38 Internet-based interventions also have been found to have some benefit for dementia caregivers.31,39
However, some rural locations continue to have limited reliable Internet services.40 In affected areas, a strong relationship with a primary care physician may be even more important to the well-being of caregivers, since other support services may be less accessible.41
Continue to: Impacts of the pandemic
Impacts of the pandemic. Although our case took place prior to the COVID-19 pandemic, it is important to acknowledge ways the pandemic has impacted informal dementia caregiving.
Caregiver stress, depression, and anxiety increased during the pandemic, and the need for greater home confinement and social distancing amplified the negative impact of social isolation, including loneliness, on caregivers.42,43 Caregivers often needed to increase their caregiving responsibilities and had more difficulty with care coordination due to limited access to in-person resources.43 The pandemic led to increased reliance on technology and telehealth in the support of dementia caregivers.43
THE CASE
The physician prescribed mirtazapine for Mr. C, titrating the dose as needed to address depressive symptoms and promote weight gain. The physician connected Mr. C’s father with home health services, including physical therapy for fall risk reduction. Mr. C also hired part-time support to provide additional assistance with ADLs and IADLs, allowing Mr. C to have time to attend to his own needs. Though provided with information about a local caregiver support group, Mr. C chose not to attend. The physician also assisted the family with advanced directives.
A particular challenge that occurred during care for the family was addressing Mr. C’s father’s driving capacity, considering his strong need for independence. To address this concern, a family meeting was held with Mr. C, his father, and his siblings from out of town. Although Mr. C’s father was not willing to relinquish his driver’s license during that meeting, he agreed to complete a functional driving assessment.
The physician continued to meet with Mr. C and his father together, as well as with Mr. C individually, to provide supportive counseling as needed. As the father’s dementia progressed and it became more difficult to complete office appointments, the physician transitioned to home visits to provide care until the father’s death.
After the death of Mr. C’s father, the physician continued to serve as Mr. C’s primary care provider.
Keeping the “family”in family medicine
Through longitudinal assessment, needs identification, and provision of relevant information, emotional support, and resources, family physicians can provide care that can improve the quality of life and well-being and help alleviate burden experienced by dementia caregivers. Family physicians also are positioned to provide treatments that can address the negative physical and psychological health outcomes associated with informal dementia caregiving. By building relationships with multiple family members across generations, family physicians can understand the context of caregiving dynamics and work together with individuals with dementia and their caregivers throughout disease progression, providing consistent support to the family unit.
CORRESPONDENCE
Kathleen M. Young, PhD, MPH, Novant Health Family Medicine Wilmington, 2523 Delaney Avenue, Wilmington, NC 28403; [email protected]
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
1. Alzheimer’s Association. 2023 Alzheimer’s Disease Facts and Figures. Alzheimers Dement. 202319:1598-1695. doi: 10.1002/alz.13016
2. Liu Z, Heffernan C, Tan J. Caregiver burden: a concept analysis. Int J of Nurs Sci. 2020;7:448-435. doi: 10.1016/j.ijnss.2020.07.012
3. Ory MG, Hoffman RR III, Yee JL, et al. Prevalence and impacts of caregiving: a detailed comparison between dementia and nondementia caregivers. Gerontologist. 1999;39:177-185. doi: 10.1093/geront/39.2.177
4. Baharudin AD, Din NC, Subramaniam P, et al. The associations between behavioral-psychological symptoms of dementia (BPSD) and coping strategy, burden of care and personality style among low-income caregivers of patients with dementia. BMC Public Health. 2019;19(suppl 4):447. doi: 10.1186/s12889-019-6868-0
5. Cheng S-T. Dementia caregiver burden: a research update and critical analysis. Curr Psychiatry Rep. 2017;19:64. doi: 10.1007/s11920-017-0818-2
6. Reed C, Belger M, Andrews JS, et al. Factors associated with long-term impact on informal caregivers during Alzheimer’s disease dementia progression: 36-month results from GERAS. Int Psychogeriatr. 2020;32:267-277. doi: 10.1017/S1041610219000425
7. Gilhooly KJ, Gilhooly MLM, Sullivan MP, et al. A meta-review of stress, coping and interventions in dementia and dementia caregiving. BMC Geriatr. 2016;16:106. doi: 10.1186/s12877-016-0280-8
8. Haley WE, Levine EG, Brown SL, et al. Psychological, social, and health consequences of caring for a relative with senile dementia. J Am Geriatr Soc. 1987;35:405-411.
9. Bom J, Bakx P, Schut F, et al. The impact of informal caregiving for older adults on the health of various types of caregivers: a systematic review. The Gerontologist. 2019;59:e629-e642. doi: 10.1093/geront/gny137
10. Fonareva I, Oken BS. Physiological and functional consequences of caregiving for relatives with dementia. Int Psychogeriatr. 2014;26:725-747. doi: 10.1017/S1041610214000039
11. Del-Pino-Casado R, Rodriguez Cardosa M, Lopez-Martinez C, et al. The association between subjective caregiver burden and depressive symptoms in carers of older relatives: a systematic review and meta-analysis. PLoS One. 2019;14:e0217648. doi: 10.1371/journal.pone.0217648
12. Del-Pino-Casado R, Priego-Cubero E, Lopez-Martinez C, et al. Subjective caregiver burden and anxiety in informal caregivers: a systematic review and meta-analysis. PLoS One. 2020;16:e0247143. doi: 10.1371/journal.pone.0247143
13. De Souza Alves LC, Quirino Montiero D, Ricarte Bento S, et al. Burnout syndrome in informal caregivers of older adults with dementia: a systematic review. Dement Neuropsychol. 2019;13:415-421. doi: 10.1590/1980-57642018dn13-040008
14. Victor CR, Rippon I, Quinn C, et al. The prevalence and predictors of loneliness in caregivers of people with dementia: findings from the IDEAL programme. Aging Ment Health. 2021;25:1232-1238. doi: 10.1080/13607863.2020.1753014
15. Sallim AB, Sayampanathan AA, Cuttilan A, et al. Prevalence of mental health disorders among caregivers of patients with Alzheimer disease. J Am Med Dir Assoc. 2015;16:1034-1041. doi: 10.1016/j.jamda.2015.09.007
16. Unpublished data from the 2015, 2016 2017, 2020, and 2021 Behavioral Risk Factor Surveillance System survey, analyzed by and provided to the Alzheimer’s Association by the Alzheimer’s Disease and Healthy Aging Program (AD+HP), Centers for Disease Control and Prevention (CDC).
17. Stall NM, Kim SJ, Hardacre KA, et al. Association of informal caregiver distress with health outcomes of community-dwelling dementia care recipients: a systematic review. J Am Geriatr Soc. 2018;00:1-9. doi: 10.1111/jgs.15690
18. Lindeza P, Rodrigues M, Costa J, et al. Impact of dementia on informal care: a systematic review of family caregivers’ perceptions. BMJ Support Palliat Care. 2020;bmjspcare-2020-002242. doi: 10.1136/bmjspcare-2020-002242
19. Lethin C, Guiteras AR, Zwakhalen S, et al. Psychological well-being over time among informal caregivers caring for persons with dementia living at home. Aging and Ment Health. 2017; 21:1138-1146. doi: 10.1080/13607863.2016.1211621
20. Family Caregiver Alliance. Caregivers Count Too! A Toolkit to Help Practitioners Assess the Needs of Family Caregivers. Family Caregiver Alliance; 2006. Accessed May 16, 2023. www.caregiver.org/uploads/legacy/pdfs/Assessment_Toolkit_20060802.pdf
21. Zarit SH, Zarit JM. Instructions for the Burden Interview. Pennsylvania State University; 1987.
22. University of Wisconsin. Zarit Burden Interview: assessing caregiver burden. Accessed May 19, 2023. https://wai.wisc.edu/wp-content/uploads/sites/1129/2021/11/Zarit-Caregiver-Burden-Assessment-Instruments.pdf
23. Gallagher-Thompson D, Bilbrey AC, Apesoa-Varano EC, et al. Conceptual framework to guide intervention research across the trajectory of dementia caregiving. Gerontologist. 2020;60:S29-S40. doi: 10.1093/geront/gnz157
24. Queluz FNFR, Kervin E, Wozney L, et al. Understanding the needs of caregivers of persons with dementia: a scoping review. Int Psychogeriatr. 2020;32:35-52. doi: 10.1017/S1041610219000243
25. McCabe M, You E, Tatangelo G. Hearing their voice: a systematic review of dementia family caregivers’ needs. Gerontologist. 2016;56:e70-e88. doi: 10.1093/geront/gnw07
26. Zwaanswijk M, Peeters JM, van Beek AP, et al. Informal caregivers of people with dementia: problems, needs and support in the initial stage and in subsequent stages of dementia: a questionnaire survey. Open Nurs J. 2013;7:6-13. doi: 10.2174/1874434601307010006
27. Jennings LA, Palimaru A, Corona MG, et al. Patient and caregiver goals for dementia care. Qual Life Res. 2017;26:685-693. doi: 10.1007/s11136-016-1471-7
28. Brodaty H, Donkin M. Family caregivers of people with dementia. Dialogues Clin Neurosci. 2009;11:217-228. doi: 10.31887/DCNS.2009.11.2/hbrodaty
29. Rote SM, Angel JL, Moon H, et al. Caregiving across diverse populations: new evidence from the national study of caregiving and Hispanic EPESE. Innovation in Aging. 2019;3:1-11. doi: 10.1093/geroni/igz033
30. Alzheimer’s Association. 2021 Alzheimer’s Disease facts and figures. Special report—race, ethnicity, and Alzheimer’s in America. Alzheimers Dement. 2021;17:70-104. doi: 10.1002/alz.12328
31. Swartz K, Collins LG. Caregiver care. Am Fam Physician. 2019;99:699-706.
32. Cheng ST, Au A, Losada A, et al. Psychological interventions for dementia caregivers: what we have achieved, what we have learned. Curr Psychiatry Rep. 2019;21:59. doi: 10.1007/s11920-019-1045-9
33. Jennings LA, Reuben DB, Everston LC, et al. Unmet needs of caregivers of patients referred to a dementia care program. J Am Geriatr Soc. 2015;63:282-289. doi: 10.1111/jgs.13251
34. Soong A, Au ST, Kyaw BM, et al. Information needs and information seeking behaviour of people with dementia and their non-professional caregivers: a scoping review. BMC Geriatrics. 2020;20:61. doi: 10.1186/s12877-020-1454-y
35. Cheng S-T, Zhang F. A comprehensive meta-review of systematic reviews and meta-analyses on nonpharmacological interventions for informal dementia caregivers. BMC Geriatrics. 2020;20:137. doi: 10.1186/s12877-020-01547-2
36. Wiegelmann H, Speller S, Verhaert LM, et al. Psychosocial interventions to support the mental health of informal caregivers of persons living with dementia—a systematic literature review. BMC Geriatrics. 2021;21:94. doi: 10.1186/s12877-021-02020-4
37. Nguyen SA, Oughli HA, Lavretsky H. Complementary and integrative medicine for neurocognitive disorders and caregiver health. Current Psychiatry Reports. 2022;24:469-480. doi: 10.1007/s11920-022-01355-y
38. Gibson A, Holmes SD, Fields NL, et al. Providing care for persons with dementia in rural communities: informal caregivers’ perceptions of supports and services. J Gerontol Soc Work. 2019;62:630-648. doi: 10.1080/01634372.2019.1636332
39. Leng M, Zhao Y, Xiau H, et al. Internet-based supportive interventions for family caregivers of people with dementia: systematic review and meta-analysis. J Med Internet Res. 2020;22:e19468. doi: 10.2196/19468
40. Ruggiano N, Brown EL, Li J, et al. Rural dementia caregivers and technology. What is the evidence? Res Gerontol Nurs. 2018;11:216-224. doi: 10.3928/19404921-20180628-04
41. Shuffler J, Lee K, Fields, et al. Challenges experienced by rural informal caregivers of older adults in the United States: a scoping review. J Evid Based Soc Work. Published online 24 February 24, 2023. doi:10.1080/26408066.2023.2183102
42. Hughes MC, Liu Y, Baumbach A. Impact of COVID-19 on the health and well-being of informal caregivers of people with dementia: a rapid systematic review. Gerontol Geriatric Med. 2021;7:1-8. doi: 10.1177/2333721421102164
43. Paplickar A, Rajagopalan J, Alladi S. Care for dementia patients and caregivers amid COVID-19 pandemic. Cereb Circ Cogn Behav. 2022;3:100040. doi: 10.1016/j.cccb.2022.100040
Immediate statin after acute stroke reduces disability
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
MUNICH, GERMANY – without compromising safety, results of the INSPIRES trial show.
The research, presented at the annual European Stroke Organisation Conference, also showed that intensive antiplatelet therapy reduced the risk for recurrent stroke albeit at an increased in bleeding risk versus standard treatment.
The study involved more than 6,000 patients with acute mild ischemic stroke or TIA and intracranial or extracranial atherosclerosis (ICAS/ECAS), who were randomly assigned in a 2 x 2 factorial design to compare intensive versus standard antiplatelet therapy and intensive statin therapy within 24 hours versus waiting up to 72 hours after onset.
Intensive antiplatelet therapy with clopidogrel plus aspirin reduced the risk for recurrent stroke within 90 days by 21% versus standard single-agent therapy, although it also doubled the risk for moderate to severe bleeding.
Starting intensive statin therapy with atorvastatin within 24 hours of onset had no impact on recurrent stroke risk but did reduce the risk for a poor functional outcome versus waiting up to 72 hours by 16%.
Moreover, it was “safe, with no increased risk of bleeding, hepatotoxicity, or muscle toxicity,” said study presenter Yilong Wang, MD, department of neurology, Beijing Tiantan Hospital, National Clinical Research Center.
There was, however, a suggestion of an interaction between intensive antiplatelet therapy and immediate intensive statin therapy, he noted, with a trend toward increased bleeding vs delaying the start of statin therapy.
Approached for comment, session cochair Carlos Molina, MD, director of the stroke unit and brain hemodynamics in Hospital Universitari Vall d’Hebron, Barcelona, said that the study is “important because when we look at studies of minor stroke and TIA, they are just focused on long-term outcomes in terms of recurrent stroke.”
He said in an interview that “putting statins in the equation and looking at their impact on long-term outcomes, the study demonstrates that statins are associated ... in particular with reductions in disabling stoke, and that’s good.”
Recurrence and progression
Dr. Wang began by highlighting that acute mild stroke and high-risk TIA are common and underestimated, with a relatively high risk for recurrence and progression, often caused by ICAS/ECAS.
Numerous guidelines recommend intensive antiplatelet therapy in the first 24 hours after the event, but Wang pointed out that there is little evidence to support this, and a meta-analysis suggested the window for effective treatment may be up to 72 hours.
In addition, intense statin therapy appears to be beneficial for the secondary prevention of atherosclerotic stroke in the nonacute phase, although there is no evidence for any neuroprotective effects in the acute phase nor for the optimal timing of starting the drugs.
Dr. Wang also noted that there is the potential for an interaction between intensive antiplatelet and statin therapy that could increase the risk for bleeding.
To investigate further, the researchers conducted a multicenter study involving patients aged 35-80 years with acute ischemic stroke or TIA.
The former was defined as an acute single infarction with 50% or greater stenosis of a major intracranial or extracranial artery that “probably account for the infarction and symptoms,” or multiple infarctions of large artery origin, including nonstenotic vulnerable plaques.
Patients were required to have a National Institutes of Health Stroke Scale score of 4-5 24 hours or less from acute stoke onset or 0-5 between 24 and 72 hours of onset.
TIA was defined as 50% or more stenosis of major intracranial or extracranial arteries that probably account for the symptoms, and an ABCD2 score for stroke risk of 4 or more within 24-72 hours of onset.
Patients were excluded if they had received dual antiplatelet therapy with aspirin and clopidogrel or high-intensity statin therapy within 14 days of random assignment or had intravenous thrombolysis or endovascular therapy after acute stroke or TIA onset.
Those included in the trial were randomly assigned in a 2 x 2 factorial design to receive:
- Intensive or dual antiplatelet therapy with clopidogrel and aspirin plus immediate high-intensity statin therapy with atorvastatin
- Intensive antiplatelet therapy plus delayed high-intensity statin therapy
- Standard antiplatelet therapy with aspirin alone plus immediate high-intensity statin therapy
- Standard antiplatelet therapy plus delayed high-intensity statin therapy
In all, 6,100 patients were enrolled from 222 hospitals in 99 cities across 25 provinces in China. The mean age was 65 years, and 34.6%-37.0% were women. TIA was recorded in 12.2%-14.1% of patients; 19.5%-19.7% had a single acute infarction, and 66.4%-68.1% had acute multiple infarctions.
The time to randomization was 24 hours or less after event onset in 12.5%-13.2% of cases versus 24-48 hours in 41.2%-42.5% and 48 hours or more in 44.9%-45.7% of patients.
The primary efficacy outcome, defined as stroke at 90 days, was significantly less common with intensive versus standard antiplatelet therapy, at a cumulative probability of 9.2% versus 7.3% (hazard ratio, 0.79; 95% confidence interval, 0.66-0.94; P = .007).
Clopidogrel plus aspirin was also associated with a significant reduction in a composite vascular event of stroke, myocardial infarction, or vascular death versus aspirin alone, at 7.5% versus 9.3% (HR, 0.80; 95% CI, 0.67-0.95, P = .01), as well as a reduction in rates of ischemic stroke (P = .002), and TIA (P = .02).
The primary safety outcome, defined as moderate to severe bleeding on the GUSTO criteria, was increased with intensive antiplatelet therapy, at 0.9% versus 0.4% for aspirin alone (HR, 2.08; 95% CI, 1.07-4.03; P = .02).
Turning to statin use, Dr. Wang showed that there was no significant difference in rates of stroke at 90 days between delayed and immediate intensive therapy, at a cumulative probability of 8.4% versus 8.1% (HR, 0.95; P = .58).
There was also no difference in rates of moderate to severe bleeding, at 0.8% with immediate versus 0.6% for delayed intensive statin therapy (HR, 1.36; 95% CI, 0.73-2.54; P = .34).
Dr. Wang reported that there were no significant differences in key secondary efficacy and safety outcomes.
Analysis of the distribution of modified Rankin Scale scores at 90 days, however, indicated that there was a significant reduction in the risk for poor functional outcome, defined as a score of 2-6, with immediate versus delayed statin therapy (odds ratio, 0.84; 95% CI, 0.72-0.99; P = .04).
Finally, it was found that combining dual antiplatelet therapy with immediate intensive statin therapy was associated with an increase in moderate to severe bleeding versus delayed statin therapy, affecting 1.1% versus 0.7% of patients. The association nonetheless did not reach statistical significance (HR, 1.70; 95% CI, 0.78-3.71; P = .18).
The study was funded by the National Natural Science Foundation of China, the National Key R&D Program of China, the Beijing Outstanding Young Scientist Program, the Beijing Youth Scholar Program, and the Beijing Talent Project. The drug was provided by Sanofi and Jialin Pharmaceutical. No relevant financial relationships were declared.
A version of this article originally appeared on Medscape.com.
AT ESOC 2023
FDA panel unanimously endorses lecanemab for Alzheimer’s
“Overall, the study demonstrated clearly that this is an effective treatment,” said acting chair Robert C. Alexander, MD, chief scientific officer, Alzheimer’s Prevention Initiative, Banner Alzheimer’s Institute, and research professor, department of psychiatry, University of Arizona, Phoenix, during the meeting.
An intravenous infusion targeting amyloid-beta, lecanemab received accelerated FDA approved earlier in 2023 for the treatment of early Alzheimer’s disease (AD). The company was required to complete a confirmatory study to verify and describe the product’s clinical benefit.
The Peripheral and Central Nervous System Drugs Advisory Committee met to discuss this phase 3 study (CLARITY-AD). The multicenter, double-blind study included 1,795 patients (mean age, 71 years) who had mild cognitive impairment caused by AD or mild AD dementia.
Delayed progression
Study participants had a broad range of comorbidities, and many were concomitantly receiving other medications. Black people were underrepresented in the study at just 3% of the total cohort.
Patients were randomly assigned to receive placebo or lecanemab 10 mg/kg biweekly. In addition to a placebo-controlled period and safety follow-up, the study has an ongoing extension phase of up to 4 years.
The study met its primary endpoint, showing a highly statistically significant 27% less decline on the Clinical Dementia Rating-Sum of Boxes at 18 months (difference in adjusted mean, –0.45; 95% CI, –0.67 to –0.23; P = .00005).
This was supported by a significant 26% difference on the AD Assessment Scale–Cognitive Subscale with 14 tasks (ADAS-Cog 14).
The drug also affected function, with a 37% decrease, compared with placebo, on the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment.
Committee members heard that the results signal delays in disease progression by about 5 months, giving patients more time to live independently and participate in hobbies and interests.
Patients who received the active drug also experienced quality of life benefits. Compared with patients who received placebo, those who took lecanemab had 49% less decline as measured with the European Quality of Life–5 Dimensions scale and 56% less decline as measured by the Quality of Life in AD scale, and caregivers reported less burden.
Lecanemab also affected biomarkers of amyloid, tau, and neurodegeneration, providing a biological basis for the treatment effects consistent with slowing of disease progression.
Unanimous support
All six committee members agreed by vote that the study provides evidence of clinical benefit. They variously descried the study and results as “robust,” “compelling,” “well conducted,” “clear and consistent,” and “clinically meaningful.”
In the active treatment group, there was a higher incidence of amyloid-related imaging abnormalities (ARIAs), which can be serious and life-threatening but are usually asymptomatic. In this study, most ARIAs had resolved by 3 months.
Deaths occurred in 0.8% of the placebo and 0.7% of the treatment group. Dean Follmann, PhD, assistant director for biostatistics, National Institute of Allergy and Infectious Diseases, Bethesda, Md., noted that the numbers of deaths and serious adverse events were “quite similar” in the two groups.
“And for serious ARIA, there was an imbalance favoring placebo, but overall, these were pretty rare,” he said.
Subgroup concerns
Committee members discussed the risk/benefit profile for three subgroups of patients – those with apolipoprotein E4 (apo E4) allele, patients taking an anticoagulant, and those with cerebral amyloid angiopathy (CAA).
In the apo E4 group, the study’s primary endpoint did not favor the drug, but secondary endpoints did.
“I think the general feeling [for apo E4 status] is that the risk/benefit still remains favorable, especially when looking across multiple endpoints,” said Dr. Alexander.
However, some members supported recommending genetic testing before initiating the drug.
The views were more diverse for the use of lecanemab in the presence of an anticoagulant, which may increase the risk for cerebral hemorrhage. Some committee members strongly recommended that these patients not receive lecanemab, while others highlighted the need for more information, owing to uncertainties about the risks.
With respect to CAA, most members supported the idea of considering use of the drug in the presence of this condition, but only after discussing the risks with patients and their families and in the presence of a robust reporting system.
An Alzheimer’s Association representative was in attendance during the public hearing portion of the meeting to express support for traditional approval of lecanemab for people with early AD.
The association strongly favors full Medicare coverage for FDA-approved AD treatments. The Centers for Medicare & Medicaid Services has determined that AD treatments receiving traditional FDA approval will be covered if clinicians register and enter data in a registry.
“While this is an important signal that CMS wants to improve access to FDA-approved treatments, registry as a condition of coverage is an unnecessary and potentially harmful barrier,” said the Alzheimer’s Association in a press release following the meeting.
A version of this article first appeared on Medscape.com.
“Overall, the study demonstrated clearly that this is an effective treatment,” said acting chair Robert C. Alexander, MD, chief scientific officer, Alzheimer’s Prevention Initiative, Banner Alzheimer’s Institute, and research professor, department of psychiatry, University of Arizona, Phoenix, during the meeting.
An intravenous infusion targeting amyloid-beta, lecanemab received accelerated FDA approved earlier in 2023 for the treatment of early Alzheimer’s disease (AD). The company was required to complete a confirmatory study to verify and describe the product’s clinical benefit.
The Peripheral and Central Nervous System Drugs Advisory Committee met to discuss this phase 3 study (CLARITY-AD). The multicenter, double-blind study included 1,795 patients (mean age, 71 years) who had mild cognitive impairment caused by AD or mild AD dementia.
Delayed progression
Study participants had a broad range of comorbidities, and many were concomitantly receiving other medications. Black people were underrepresented in the study at just 3% of the total cohort.
Patients were randomly assigned to receive placebo or lecanemab 10 mg/kg biweekly. In addition to a placebo-controlled period and safety follow-up, the study has an ongoing extension phase of up to 4 years.
The study met its primary endpoint, showing a highly statistically significant 27% less decline on the Clinical Dementia Rating-Sum of Boxes at 18 months (difference in adjusted mean, –0.45; 95% CI, –0.67 to –0.23; P = .00005).
This was supported by a significant 26% difference on the AD Assessment Scale–Cognitive Subscale with 14 tasks (ADAS-Cog 14).
The drug also affected function, with a 37% decrease, compared with placebo, on the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment.
Committee members heard that the results signal delays in disease progression by about 5 months, giving patients more time to live independently and participate in hobbies and interests.
Patients who received the active drug also experienced quality of life benefits. Compared with patients who received placebo, those who took lecanemab had 49% less decline as measured with the European Quality of Life–5 Dimensions scale and 56% less decline as measured by the Quality of Life in AD scale, and caregivers reported less burden.
Lecanemab also affected biomarkers of amyloid, tau, and neurodegeneration, providing a biological basis for the treatment effects consistent with slowing of disease progression.
Unanimous support
All six committee members agreed by vote that the study provides evidence of clinical benefit. They variously descried the study and results as “robust,” “compelling,” “well conducted,” “clear and consistent,” and “clinically meaningful.”
In the active treatment group, there was a higher incidence of amyloid-related imaging abnormalities (ARIAs), which can be serious and life-threatening but are usually asymptomatic. In this study, most ARIAs had resolved by 3 months.
Deaths occurred in 0.8% of the placebo and 0.7% of the treatment group. Dean Follmann, PhD, assistant director for biostatistics, National Institute of Allergy and Infectious Diseases, Bethesda, Md., noted that the numbers of deaths and serious adverse events were “quite similar” in the two groups.
“And for serious ARIA, there was an imbalance favoring placebo, but overall, these were pretty rare,” he said.
Subgroup concerns
Committee members discussed the risk/benefit profile for three subgroups of patients – those with apolipoprotein E4 (apo E4) allele, patients taking an anticoagulant, and those with cerebral amyloid angiopathy (CAA).
In the apo E4 group, the study’s primary endpoint did not favor the drug, but secondary endpoints did.
“I think the general feeling [for apo E4 status] is that the risk/benefit still remains favorable, especially when looking across multiple endpoints,” said Dr. Alexander.
However, some members supported recommending genetic testing before initiating the drug.
The views were more diverse for the use of lecanemab in the presence of an anticoagulant, which may increase the risk for cerebral hemorrhage. Some committee members strongly recommended that these patients not receive lecanemab, while others highlighted the need for more information, owing to uncertainties about the risks.
With respect to CAA, most members supported the idea of considering use of the drug in the presence of this condition, but only after discussing the risks with patients and their families and in the presence of a robust reporting system.
An Alzheimer’s Association representative was in attendance during the public hearing portion of the meeting to express support for traditional approval of lecanemab for people with early AD.
The association strongly favors full Medicare coverage for FDA-approved AD treatments. The Centers for Medicare & Medicaid Services has determined that AD treatments receiving traditional FDA approval will be covered if clinicians register and enter data in a registry.
“While this is an important signal that CMS wants to improve access to FDA-approved treatments, registry as a condition of coverage is an unnecessary and potentially harmful barrier,” said the Alzheimer’s Association in a press release following the meeting.
A version of this article first appeared on Medscape.com.
“Overall, the study demonstrated clearly that this is an effective treatment,” said acting chair Robert C. Alexander, MD, chief scientific officer, Alzheimer’s Prevention Initiative, Banner Alzheimer’s Institute, and research professor, department of psychiatry, University of Arizona, Phoenix, during the meeting.
An intravenous infusion targeting amyloid-beta, lecanemab received accelerated FDA approved earlier in 2023 for the treatment of early Alzheimer’s disease (AD). The company was required to complete a confirmatory study to verify and describe the product’s clinical benefit.
The Peripheral and Central Nervous System Drugs Advisory Committee met to discuss this phase 3 study (CLARITY-AD). The multicenter, double-blind study included 1,795 patients (mean age, 71 years) who had mild cognitive impairment caused by AD or mild AD dementia.
Delayed progression
Study participants had a broad range of comorbidities, and many were concomitantly receiving other medications. Black people were underrepresented in the study at just 3% of the total cohort.
Patients were randomly assigned to receive placebo or lecanemab 10 mg/kg biweekly. In addition to a placebo-controlled period and safety follow-up, the study has an ongoing extension phase of up to 4 years.
The study met its primary endpoint, showing a highly statistically significant 27% less decline on the Clinical Dementia Rating-Sum of Boxes at 18 months (difference in adjusted mean, –0.45; 95% CI, –0.67 to –0.23; P = .00005).
This was supported by a significant 26% difference on the AD Assessment Scale–Cognitive Subscale with 14 tasks (ADAS-Cog 14).
The drug also affected function, with a 37% decrease, compared with placebo, on the AD Cooperative Study–Activities of Daily Living Scale for Mild Cognitive Impairment.
Committee members heard that the results signal delays in disease progression by about 5 months, giving patients more time to live independently and participate in hobbies and interests.
Patients who received the active drug also experienced quality of life benefits. Compared with patients who received placebo, those who took lecanemab had 49% less decline as measured with the European Quality of Life–5 Dimensions scale and 56% less decline as measured by the Quality of Life in AD scale, and caregivers reported less burden.
Lecanemab also affected biomarkers of amyloid, tau, and neurodegeneration, providing a biological basis for the treatment effects consistent with slowing of disease progression.
Unanimous support
All six committee members agreed by vote that the study provides evidence of clinical benefit. They variously descried the study and results as “robust,” “compelling,” “well conducted,” “clear and consistent,” and “clinically meaningful.”
In the active treatment group, there was a higher incidence of amyloid-related imaging abnormalities (ARIAs), which can be serious and life-threatening but are usually asymptomatic. In this study, most ARIAs had resolved by 3 months.
Deaths occurred in 0.8% of the placebo and 0.7% of the treatment group. Dean Follmann, PhD, assistant director for biostatistics, National Institute of Allergy and Infectious Diseases, Bethesda, Md., noted that the numbers of deaths and serious adverse events were “quite similar” in the two groups.
“And for serious ARIA, there was an imbalance favoring placebo, but overall, these were pretty rare,” he said.
Subgroup concerns
Committee members discussed the risk/benefit profile for three subgroups of patients – those with apolipoprotein E4 (apo E4) allele, patients taking an anticoagulant, and those with cerebral amyloid angiopathy (CAA).
In the apo E4 group, the study’s primary endpoint did not favor the drug, but secondary endpoints did.
“I think the general feeling [for apo E4 status] is that the risk/benefit still remains favorable, especially when looking across multiple endpoints,” said Dr. Alexander.
However, some members supported recommending genetic testing before initiating the drug.
The views were more diverse for the use of lecanemab in the presence of an anticoagulant, which may increase the risk for cerebral hemorrhage. Some committee members strongly recommended that these patients not receive lecanemab, while others highlighted the need for more information, owing to uncertainties about the risks.
With respect to CAA, most members supported the idea of considering use of the drug in the presence of this condition, but only after discussing the risks with patients and their families and in the presence of a robust reporting system.
An Alzheimer’s Association representative was in attendance during the public hearing portion of the meeting to express support for traditional approval of lecanemab for people with early AD.
The association strongly favors full Medicare coverage for FDA-approved AD treatments. The Centers for Medicare & Medicaid Services has determined that AD treatments receiving traditional FDA approval will be covered if clinicians register and enter data in a registry.
“While this is an important signal that CMS wants to improve access to FDA-approved treatments, registry as a condition of coverage is an unnecessary and potentially harmful barrier,” said the Alzheimer’s Association in a press release following the meeting.
A version of this article first appeared on Medscape.com.
Muscle fat: A new risk factor for cognitive decline?
Investigators assessed muscle fat in more than 1,600 adults in their 70s and evaluated their cognitive function over a 10-year period. They found that increases in muscle adiposity from year 1 to year 6 were associated with greater cognitive decline over time, independent of total weight, other fat deposits, muscle characteristics, and traditional dementia risk factors.
The findings were similar between Black and White people and between men and women.
“Increasing adiposity – or fat deposition – in skeletal muscles predicted faster cognitive decline, irrespective of demographics or other disease, and this effect was distinct from that of other types of fat or other muscle characteristics, such as strength or mass,” study investigator Caterina Rosano MD, MPH, professor of epidemiology at the University of Pittsburgh, said in an interview.
The study was published in the Journal of the American Geriatrics Society.
Biologically plausible
“There has been a growing recognition that overall adiposity and muscle measures, such as strength and mass, are individual indicators of future dementia risk and both strengthen the algorithms to predict cognitive decline,” said Dr. Rosano, associate director for clinical translation at the University of Pittsburgh’s Aging Institute. “However, adiposity in the muscle has not been examined.”
Some evidence supports a “biologically plausible link” between muscle adiposity and dementia risk. For example, muscle adiposity increases the risk for type 2 diabetes and hypertension, both of which are dementia risk factors.
Skeletal muscle adiposity increases with older age, even in older adults who lose weight, and is “highly prevalent” among older adults of African ancestry.
The researchers examined a large, biracial sample of older adults participating in the Health, Aging and Body Composition study, which enrolled men and women aged between 70 and 79 years. Participants were followed for an average of 9.0 ± 1.8 years.
During years 1 and 6, participants’ body composition was analyzed, including intermuscular adipose tissue (IMAT), visceral and subcutaneous adiposity, total fat mass, and muscle area.
In years 1, 3, 5, 8, and 10, participants’ cognition was measured using the modified Mini-Mental State (3MS) exam.
The main independent variable was 5-year change in thigh IMAT (year 6 minus year 1), and the main dependent variable was 3MS decline (from year 5 to year 10).
The researchers adjusted all the models for traditional dementia risk factors at baseline including 3MS, education, apo E4 allele, diabetes, hypertension, and physical activity and also calculated interactions between IMAT change by race or sex.
These models also accounted for change in muscle strength, muscle area, body weight, abdominal subcutaneous and visceral adiposity, and total body fat mass as well as cytokines related to adiposity.
‘Rich and engaging crosstalk’
The final sample included 1634 participants (mean age, 73.38 years at baseline; 48% female; 35% Black; mean baseline 3MS score, 91.6).
Thigh IMAT increased by 39.0% in all participants from year 1 to year 6, which corresponded to an increase of 4.85 cm2 or 0.97 cm2/year. During the same time period, muscle strength decreased by 14.0% (P < .05), although thigh muscle area remained stable, decreasing less than 0.5%.
There were decreases in both abdominal subcutaneous and visceral adiposity of 3.92% and 6.43%, respectively (P < .05). There was a decrease of 3.3% in 3MS from year 5 to year 10.
Several variables were associated with 3MS decline, independent of any change in thigh IMAT: older age, less education, and having at least one copy of the APOe4 allele. These variables were included in the model of IMAT change predicting 3MS change.
A statistically significant association of IMAT increase with 3MS decline was found. The IMAT increase of 4.85 cm2 corresponded to a 3MS decline of an additional 3.6 points (P < .0001) from year 5 to year 10, “indicating a clinically important change.”
The association between increasing thigh IMAT with declining 3MS “remained statistically significant” after adjusting for race, age, education, and apo E4 (P < .0001) and was independent of changes in thigh muscle area, muscle strength, and other adiposity measures.
In participants with increased IMAT in years 1-6, the mean 3MS score fell to approximately 87 points at year 10, compared with those without increased IMAT, with a 3MS score that dropped to approximately 89 points.
Interactions by race and sex were not statistically significant (P > .08).
“Our results suggest that adiposity in muscles can predict cognitive decline, in addition to (not instead of) other traditional dementia risk factors,” said Dr. Rosano.
There is “a rich and engaging crosstalk between muscle, adipose tissue, and the brain all throughout our lives, happening through factors released in the bloodstream that can reach the brain, however, the specific identity of the factors responsible for the crosstalk of muscle adiposity and brain in older adults has not yet been discovered,” she noted.
Although muscle adiposity is “not yet routinely measured in clinical settings, it is being measured opportunistically on clinical CT scans obtained as part of routine patient care,” she added. “These CT measurements have already been validated in many studies of older adults; thus, clinicians could have access to this novel information without additional cost, time, or radiation exposure.”
Causality not proven
In a comment, Bruce Albala, PhD, professor, department of environmental and occupational health, University of California, Irvine, noted that the 3MS assessment is scored on a 100-point scale, with a score less than 78 “generally regarded as indicating cognitive impairment or approaching a dementia condition.” In the current study, the mean 3MS score of participants with increased IMAT was still “well above the dementia cut-off.”
Moreover, “even if there is a relationship or correlation between IMAT and cognition, this does not prove or even suggest causality, especially from a biological mechanistic approach,” said Dr. Albaba, an adjunct professor of neurology, who was not involved in the study. “Clearly, more research is needed even to understand the relationship between these two factors.”
The study was supported by the National Institute on Aging. Dr. Rosano and coauthors and Dr. Albala declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators assessed muscle fat in more than 1,600 adults in their 70s and evaluated their cognitive function over a 10-year period. They found that increases in muscle adiposity from year 1 to year 6 were associated with greater cognitive decline over time, independent of total weight, other fat deposits, muscle characteristics, and traditional dementia risk factors.
The findings were similar between Black and White people and between men and women.
“Increasing adiposity – or fat deposition – in skeletal muscles predicted faster cognitive decline, irrespective of demographics or other disease, and this effect was distinct from that of other types of fat or other muscle characteristics, such as strength or mass,” study investigator Caterina Rosano MD, MPH, professor of epidemiology at the University of Pittsburgh, said in an interview.
The study was published in the Journal of the American Geriatrics Society.
Biologically plausible
“There has been a growing recognition that overall adiposity and muscle measures, such as strength and mass, are individual indicators of future dementia risk and both strengthen the algorithms to predict cognitive decline,” said Dr. Rosano, associate director for clinical translation at the University of Pittsburgh’s Aging Institute. “However, adiposity in the muscle has not been examined.”
Some evidence supports a “biologically plausible link” between muscle adiposity and dementia risk. For example, muscle adiposity increases the risk for type 2 diabetes and hypertension, both of which are dementia risk factors.
Skeletal muscle adiposity increases with older age, even in older adults who lose weight, and is “highly prevalent” among older adults of African ancestry.
The researchers examined a large, biracial sample of older adults participating in the Health, Aging and Body Composition study, which enrolled men and women aged between 70 and 79 years. Participants were followed for an average of 9.0 ± 1.8 years.
During years 1 and 6, participants’ body composition was analyzed, including intermuscular adipose tissue (IMAT), visceral and subcutaneous adiposity, total fat mass, and muscle area.
In years 1, 3, 5, 8, and 10, participants’ cognition was measured using the modified Mini-Mental State (3MS) exam.
The main independent variable was 5-year change in thigh IMAT (year 6 minus year 1), and the main dependent variable was 3MS decline (from year 5 to year 10).
The researchers adjusted all the models for traditional dementia risk factors at baseline including 3MS, education, apo E4 allele, diabetes, hypertension, and physical activity and also calculated interactions between IMAT change by race or sex.
These models also accounted for change in muscle strength, muscle area, body weight, abdominal subcutaneous and visceral adiposity, and total body fat mass as well as cytokines related to adiposity.
‘Rich and engaging crosstalk’
The final sample included 1634 participants (mean age, 73.38 years at baseline; 48% female; 35% Black; mean baseline 3MS score, 91.6).
Thigh IMAT increased by 39.0% in all participants from year 1 to year 6, which corresponded to an increase of 4.85 cm2 or 0.97 cm2/year. During the same time period, muscle strength decreased by 14.0% (P < .05), although thigh muscle area remained stable, decreasing less than 0.5%.
There were decreases in both abdominal subcutaneous and visceral adiposity of 3.92% and 6.43%, respectively (P < .05). There was a decrease of 3.3% in 3MS from year 5 to year 10.
Several variables were associated with 3MS decline, independent of any change in thigh IMAT: older age, less education, and having at least one copy of the APOe4 allele. These variables were included in the model of IMAT change predicting 3MS change.
A statistically significant association of IMAT increase with 3MS decline was found. The IMAT increase of 4.85 cm2 corresponded to a 3MS decline of an additional 3.6 points (P < .0001) from year 5 to year 10, “indicating a clinically important change.”
The association between increasing thigh IMAT with declining 3MS “remained statistically significant” after adjusting for race, age, education, and apo E4 (P < .0001) and was independent of changes in thigh muscle area, muscle strength, and other adiposity measures.
In participants with increased IMAT in years 1-6, the mean 3MS score fell to approximately 87 points at year 10, compared with those without increased IMAT, with a 3MS score that dropped to approximately 89 points.
Interactions by race and sex were not statistically significant (P > .08).
“Our results suggest that adiposity in muscles can predict cognitive decline, in addition to (not instead of) other traditional dementia risk factors,” said Dr. Rosano.
There is “a rich and engaging crosstalk between muscle, adipose tissue, and the brain all throughout our lives, happening through factors released in the bloodstream that can reach the brain, however, the specific identity of the factors responsible for the crosstalk of muscle adiposity and brain in older adults has not yet been discovered,” she noted.
Although muscle adiposity is “not yet routinely measured in clinical settings, it is being measured opportunistically on clinical CT scans obtained as part of routine patient care,” she added. “These CT measurements have already been validated in many studies of older adults; thus, clinicians could have access to this novel information without additional cost, time, or radiation exposure.”
Causality not proven
In a comment, Bruce Albala, PhD, professor, department of environmental and occupational health, University of California, Irvine, noted that the 3MS assessment is scored on a 100-point scale, with a score less than 78 “generally regarded as indicating cognitive impairment or approaching a dementia condition.” In the current study, the mean 3MS score of participants with increased IMAT was still “well above the dementia cut-off.”
Moreover, “even if there is a relationship or correlation between IMAT and cognition, this does not prove or even suggest causality, especially from a biological mechanistic approach,” said Dr. Albaba, an adjunct professor of neurology, who was not involved in the study. “Clearly, more research is needed even to understand the relationship between these two factors.”
The study was supported by the National Institute on Aging. Dr. Rosano and coauthors and Dr. Albala declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Investigators assessed muscle fat in more than 1,600 adults in their 70s and evaluated their cognitive function over a 10-year period. They found that increases in muscle adiposity from year 1 to year 6 were associated with greater cognitive decline over time, independent of total weight, other fat deposits, muscle characteristics, and traditional dementia risk factors.
The findings were similar between Black and White people and between men and women.
“Increasing adiposity – or fat deposition – in skeletal muscles predicted faster cognitive decline, irrespective of demographics or other disease, and this effect was distinct from that of other types of fat or other muscle characteristics, such as strength or mass,” study investigator Caterina Rosano MD, MPH, professor of epidemiology at the University of Pittsburgh, said in an interview.
The study was published in the Journal of the American Geriatrics Society.
Biologically plausible
“There has been a growing recognition that overall adiposity and muscle measures, such as strength and mass, are individual indicators of future dementia risk and both strengthen the algorithms to predict cognitive decline,” said Dr. Rosano, associate director for clinical translation at the University of Pittsburgh’s Aging Institute. “However, adiposity in the muscle has not been examined.”
Some evidence supports a “biologically plausible link” between muscle adiposity and dementia risk. For example, muscle adiposity increases the risk for type 2 diabetes and hypertension, both of which are dementia risk factors.
Skeletal muscle adiposity increases with older age, even in older adults who lose weight, and is “highly prevalent” among older adults of African ancestry.
The researchers examined a large, biracial sample of older adults participating in the Health, Aging and Body Composition study, which enrolled men and women aged between 70 and 79 years. Participants were followed for an average of 9.0 ± 1.8 years.
During years 1 and 6, participants’ body composition was analyzed, including intermuscular adipose tissue (IMAT), visceral and subcutaneous adiposity, total fat mass, and muscle area.
In years 1, 3, 5, 8, and 10, participants’ cognition was measured using the modified Mini-Mental State (3MS) exam.
The main independent variable was 5-year change in thigh IMAT (year 6 minus year 1), and the main dependent variable was 3MS decline (from year 5 to year 10).
The researchers adjusted all the models for traditional dementia risk factors at baseline including 3MS, education, apo E4 allele, diabetes, hypertension, and physical activity and also calculated interactions between IMAT change by race or sex.
These models also accounted for change in muscle strength, muscle area, body weight, abdominal subcutaneous and visceral adiposity, and total body fat mass as well as cytokines related to adiposity.
‘Rich and engaging crosstalk’
The final sample included 1634 participants (mean age, 73.38 years at baseline; 48% female; 35% Black; mean baseline 3MS score, 91.6).
Thigh IMAT increased by 39.0% in all participants from year 1 to year 6, which corresponded to an increase of 4.85 cm2 or 0.97 cm2/year. During the same time period, muscle strength decreased by 14.0% (P < .05), although thigh muscle area remained stable, decreasing less than 0.5%.
There were decreases in both abdominal subcutaneous and visceral adiposity of 3.92% and 6.43%, respectively (P < .05). There was a decrease of 3.3% in 3MS from year 5 to year 10.
Several variables were associated with 3MS decline, independent of any change in thigh IMAT: older age, less education, and having at least one copy of the APOe4 allele. These variables were included in the model of IMAT change predicting 3MS change.
A statistically significant association of IMAT increase with 3MS decline was found. The IMAT increase of 4.85 cm2 corresponded to a 3MS decline of an additional 3.6 points (P < .0001) from year 5 to year 10, “indicating a clinically important change.”
The association between increasing thigh IMAT with declining 3MS “remained statistically significant” after adjusting for race, age, education, and apo E4 (P < .0001) and was independent of changes in thigh muscle area, muscle strength, and other adiposity measures.
In participants with increased IMAT in years 1-6, the mean 3MS score fell to approximately 87 points at year 10, compared with those without increased IMAT, with a 3MS score that dropped to approximately 89 points.
Interactions by race and sex were not statistically significant (P > .08).
“Our results suggest that adiposity in muscles can predict cognitive decline, in addition to (not instead of) other traditional dementia risk factors,” said Dr. Rosano.
There is “a rich and engaging crosstalk between muscle, adipose tissue, and the brain all throughout our lives, happening through factors released in the bloodstream that can reach the brain, however, the specific identity of the factors responsible for the crosstalk of muscle adiposity and brain in older adults has not yet been discovered,” she noted.
Although muscle adiposity is “not yet routinely measured in clinical settings, it is being measured opportunistically on clinical CT scans obtained as part of routine patient care,” she added. “These CT measurements have already been validated in many studies of older adults; thus, clinicians could have access to this novel information without additional cost, time, or radiation exposure.”
Causality not proven
In a comment, Bruce Albala, PhD, professor, department of environmental and occupational health, University of California, Irvine, noted that the 3MS assessment is scored on a 100-point scale, with a score less than 78 “generally regarded as indicating cognitive impairment or approaching a dementia condition.” In the current study, the mean 3MS score of participants with increased IMAT was still “well above the dementia cut-off.”
Moreover, “even if there is a relationship or correlation between IMAT and cognition, this does not prove or even suggest causality, especially from a biological mechanistic approach,” said Dr. Albaba, an adjunct professor of neurology, who was not involved in the study. “Clearly, more research is needed even to understand the relationship between these two factors.”
The study was supported by the National Institute on Aging. Dr. Rosano and coauthors and Dr. Albala declared no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN GERIATRICS SOCIETY
Blood biomarker may help predict who will develop Alzheimer’s
A blood biomarker that measures astrocyte reactivity may help determine who, among cognitively unimpaired older adults with amyloid-beta, will go on to develop Alzheimer’s disease (AD), new research suggests.
Investigators tested the blood of 1,000 cognitively healthy individuals with and without amyloid-beta pathology and found that only those with a combination of amyloid-beta burden and abnormal astrocyte activation subsequently progressed to AD.
“Our study argues that testing for the presence of brain amyloid along with blood biomarkers of astrocyte reactivity is the optimal screening to identify patients who are most at risk for progressing to Alzheimer’s disease,” senior investigator Tharick A. Pascoal, MD, PhD, associate professor of psychiatry and neurology, University of Pittsburgh, said in a release.
At this point, the biomarker is a research tool, but its application in clinical practice “is not very far away,” Dr. Pascoal told this news organization.
The study was published online in Nature Medicine.
Multicenter study
In AD, accumulation of amyloid-beta in the brain precedes tau pathology, but not everyone with amyloid-beta develops tau, and, consequently, clinical symptoms. Approximately 30% of older adults have brain amyloid but many never progress to AD, said Dr. Pascoal.
This suggests other biological processes may trigger the deleterious effects of amyloid-beta in the early stages of AD.
Finding predictive markers of early amyloid-beta–related tau pathology would help identify cognitively normal individuals who are more likely to develop AD.
Post-mortem studies show astrocyte reactivity – changes in glial cells in the brain and spinal cord because of an insult in the brain – is an early AD abnormality. Other research suggests a close link between amyloid-beta, astrocyte reactivity, and tau.
In addition, evidence suggests plasma measures of glial fibrillary acidic protein (GFAP) could be a strong proxy of astrocyte reactivity in the brain. Dr. Pascoal explained that when astrocytes are changed or become bigger, more GFAP is released.
The study included 1,016 cognitively normal individuals from three centers; some had amyloid pathology, some did not. Participants’ mean age was 69.6 years, and all were deemed negative or positive for astrocyte reactivity based on plasma GFAP levels.
Results showed amyloid-beta is associated with increased plasma phosphorylated tau only in individuals positive for astrocyte reactivity. In addition, analyses using PET scans showed an AD-like pattern of tau tangle accumulation as a function of amyloid-beta exclusively in those same individuals.
Early upstream event
The findings suggest abnormalities in astrocyte reactivity is an early upstream event that likely occurs prior to tau pathology, which is closely related to the development of neurodegeneration and cognitive decline.
It’s likely many types of insults or processes can lead to astrocyte reactivity, possibly including COVID, but more research in this area is needed, said Dr. Pascoal.
“Our study only looked at the consequence of having both amyloid and astrocyte reactivity; it did not elucidate what is causing either of them,” he said.
Although “we were able to have very good results” in the current study, additional studies are needed to better establish the cut-off for GFAP levels that signal progression, said Dr. Pascoal.
The effect of astrocyte reactivity on the association between amyloid-beta and tau phosphorylation was greater in men than women. Dr. Pascoal noted anti-amyloid therapies, which might be modifying the amyloid-beta-astrocyte-tau pathway, tend to have a much larger effect in men than women.
Further studies that measure amyloid-beta, tau, and GFAP biomarkers at multiple timepoints, and with long follow-up, are needed, the investigators note.
The results may have implications for clinical trials, which have increasingly focused on individuals in the earliest preclinical phases of AD. Future studies should include cognitively normal patients who are positive for both amyloid pathology and astrocyte reactivity but have no overt p-tau abnormality, said Dr. Pascoal.
This may provide a time window for interventions very early in the disease process in those at increased risk for AD-related progression.
The study did not determine whether participants with both amyloid and astrocyte reactivity will inevitably develop AD, and to do so would require a longer follow up. “Our outcome was correlation to tau in the brain, which is something we know will lead to AD.”
Although the cohort represents significant socioeconomic diversity, a main limitation of the study was that subjects were mainly White, which limits the generalizability of the findings to a more diverse population.
The study received support from the National Institute of Aging; National Heart Lung and Blood Institute; Alzheimer’s Association; Fonds de Recherche du Québec-Santé; Canadian Consortium of Neurodegeneration in Aging; Weston Brain Institute; Colin Adair Charitable Foundation; Swedish Research Council; Wallenberg Scholar; BrightFocus Foundation; Swedish Alzheimer Foundation; Swedish Brain Foundation; Agneta Prytz-Folkes & Gösta Folkes Foundation; European Union; Swedish State Support for Clinical Research; Alzheimer Drug Discovery Foundation; Bluefield Project, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden; the UK Dementia Research Institute at UCL; National Academy of Neuropsychology; Fundação de Amparo a pesquisa do Rio Grande do Sul; Instituto Serrapilheira; and Hjärnfonden.
Dr. Pascoal reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A blood biomarker that measures astrocyte reactivity may help determine who, among cognitively unimpaired older adults with amyloid-beta, will go on to develop Alzheimer’s disease (AD), new research suggests.
Investigators tested the blood of 1,000 cognitively healthy individuals with and without amyloid-beta pathology and found that only those with a combination of amyloid-beta burden and abnormal astrocyte activation subsequently progressed to AD.
“Our study argues that testing for the presence of brain amyloid along with blood biomarkers of astrocyte reactivity is the optimal screening to identify patients who are most at risk for progressing to Alzheimer’s disease,” senior investigator Tharick A. Pascoal, MD, PhD, associate professor of psychiatry and neurology, University of Pittsburgh, said in a release.
At this point, the biomarker is a research tool, but its application in clinical practice “is not very far away,” Dr. Pascoal told this news organization.
The study was published online in Nature Medicine.
Multicenter study
In AD, accumulation of amyloid-beta in the brain precedes tau pathology, but not everyone with amyloid-beta develops tau, and, consequently, clinical symptoms. Approximately 30% of older adults have brain amyloid but many never progress to AD, said Dr. Pascoal.
This suggests other biological processes may trigger the deleterious effects of amyloid-beta in the early stages of AD.
Finding predictive markers of early amyloid-beta–related tau pathology would help identify cognitively normal individuals who are more likely to develop AD.
Post-mortem studies show astrocyte reactivity – changes in glial cells in the brain and spinal cord because of an insult in the brain – is an early AD abnormality. Other research suggests a close link between amyloid-beta, astrocyte reactivity, and tau.
In addition, evidence suggests plasma measures of glial fibrillary acidic protein (GFAP) could be a strong proxy of astrocyte reactivity in the brain. Dr. Pascoal explained that when astrocytes are changed or become bigger, more GFAP is released.
The study included 1,016 cognitively normal individuals from three centers; some had amyloid pathology, some did not. Participants’ mean age was 69.6 years, and all were deemed negative or positive for astrocyte reactivity based on plasma GFAP levels.
Results showed amyloid-beta is associated with increased plasma phosphorylated tau only in individuals positive for astrocyte reactivity. In addition, analyses using PET scans showed an AD-like pattern of tau tangle accumulation as a function of amyloid-beta exclusively in those same individuals.
Early upstream event
The findings suggest abnormalities in astrocyte reactivity is an early upstream event that likely occurs prior to tau pathology, which is closely related to the development of neurodegeneration and cognitive decline.
It’s likely many types of insults or processes can lead to astrocyte reactivity, possibly including COVID, but more research in this area is needed, said Dr. Pascoal.
“Our study only looked at the consequence of having both amyloid and astrocyte reactivity; it did not elucidate what is causing either of them,” he said.
Although “we were able to have very good results” in the current study, additional studies are needed to better establish the cut-off for GFAP levels that signal progression, said Dr. Pascoal.
The effect of astrocyte reactivity on the association between amyloid-beta and tau phosphorylation was greater in men than women. Dr. Pascoal noted anti-amyloid therapies, which might be modifying the amyloid-beta-astrocyte-tau pathway, tend to have a much larger effect in men than women.
Further studies that measure amyloid-beta, tau, and GFAP biomarkers at multiple timepoints, and with long follow-up, are needed, the investigators note.
The results may have implications for clinical trials, which have increasingly focused on individuals in the earliest preclinical phases of AD. Future studies should include cognitively normal patients who are positive for both amyloid pathology and astrocyte reactivity but have no overt p-tau abnormality, said Dr. Pascoal.
This may provide a time window for interventions very early in the disease process in those at increased risk for AD-related progression.
The study did not determine whether participants with both amyloid and astrocyte reactivity will inevitably develop AD, and to do so would require a longer follow up. “Our outcome was correlation to tau in the brain, which is something we know will lead to AD.”
Although the cohort represents significant socioeconomic diversity, a main limitation of the study was that subjects were mainly White, which limits the generalizability of the findings to a more diverse population.
The study received support from the National Institute of Aging; National Heart Lung and Blood Institute; Alzheimer’s Association; Fonds de Recherche du Québec-Santé; Canadian Consortium of Neurodegeneration in Aging; Weston Brain Institute; Colin Adair Charitable Foundation; Swedish Research Council; Wallenberg Scholar; BrightFocus Foundation; Swedish Alzheimer Foundation; Swedish Brain Foundation; Agneta Prytz-Folkes & Gösta Folkes Foundation; European Union; Swedish State Support for Clinical Research; Alzheimer Drug Discovery Foundation; Bluefield Project, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden; the UK Dementia Research Institute at UCL; National Academy of Neuropsychology; Fundação de Amparo a pesquisa do Rio Grande do Sul; Instituto Serrapilheira; and Hjärnfonden.
Dr. Pascoal reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A blood biomarker that measures astrocyte reactivity may help determine who, among cognitively unimpaired older adults with amyloid-beta, will go on to develop Alzheimer’s disease (AD), new research suggests.
Investigators tested the blood of 1,000 cognitively healthy individuals with and without amyloid-beta pathology and found that only those with a combination of amyloid-beta burden and abnormal astrocyte activation subsequently progressed to AD.
“Our study argues that testing for the presence of brain amyloid along with blood biomarkers of astrocyte reactivity is the optimal screening to identify patients who are most at risk for progressing to Alzheimer’s disease,” senior investigator Tharick A. Pascoal, MD, PhD, associate professor of psychiatry and neurology, University of Pittsburgh, said in a release.
At this point, the biomarker is a research tool, but its application in clinical practice “is not very far away,” Dr. Pascoal told this news organization.
The study was published online in Nature Medicine.
Multicenter study
In AD, accumulation of amyloid-beta in the brain precedes tau pathology, but not everyone with amyloid-beta develops tau, and, consequently, clinical symptoms. Approximately 30% of older adults have brain amyloid but many never progress to AD, said Dr. Pascoal.
This suggests other biological processes may trigger the deleterious effects of amyloid-beta in the early stages of AD.
Finding predictive markers of early amyloid-beta–related tau pathology would help identify cognitively normal individuals who are more likely to develop AD.
Post-mortem studies show astrocyte reactivity – changes in glial cells in the brain and spinal cord because of an insult in the brain – is an early AD abnormality. Other research suggests a close link between amyloid-beta, astrocyte reactivity, and tau.
In addition, evidence suggests plasma measures of glial fibrillary acidic protein (GFAP) could be a strong proxy of astrocyte reactivity in the brain. Dr. Pascoal explained that when astrocytes are changed or become bigger, more GFAP is released.
The study included 1,016 cognitively normal individuals from three centers; some had amyloid pathology, some did not. Participants’ mean age was 69.6 years, and all were deemed negative or positive for astrocyte reactivity based on plasma GFAP levels.
Results showed amyloid-beta is associated with increased plasma phosphorylated tau only in individuals positive for astrocyte reactivity. In addition, analyses using PET scans showed an AD-like pattern of tau tangle accumulation as a function of amyloid-beta exclusively in those same individuals.
Early upstream event
The findings suggest abnormalities in astrocyte reactivity is an early upstream event that likely occurs prior to tau pathology, which is closely related to the development of neurodegeneration and cognitive decline.
It’s likely many types of insults or processes can lead to astrocyte reactivity, possibly including COVID, but more research in this area is needed, said Dr. Pascoal.
“Our study only looked at the consequence of having both amyloid and astrocyte reactivity; it did not elucidate what is causing either of them,” he said.
Although “we were able to have very good results” in the current study, additional studies are needed to better establish the cut-off for GFAP levels that signal progression, said Dr. Pascoal.
The effect of astrocyte reactivity on the association between amyloid-beta and tau phosphorylation was greater in men than women. Dr. Pascoal noted anti-amyloid therapies, which might be modifying the amyloid-beta-astrocyte-tau pathway, tend to have a much larger effect in men than women.
Further studies that measure amyloid-beta, tau, and GFAP biomarkers at multiple timepoints, and with long follow-up, are needed, the investigators note.
The results may have implications for clinical trials, which have increasingly focused on individuals in the earliest preclinical phases of AD. Future studies should include cognitively normal patients who are positive for both amyloid pathology and astrocyte reactivity but have no overt p-tau abnormality, said Dr. Pascoal.
This may provide a time window for interventions very early in the disease process in those at increased risk for AD-related progression.
The study did not determine whether participants with both amyloid and astrocyte reactivity will inevitably develop AD, and to do so would require a longer follow up. “Our outcome was correlation to tau in the brain, which is something we know will lead to AD.”
Although the cohort represents significant socioeconomic diversity, a main limitation of the study was that subjects were mainly White, which limits the generalizability of the findings to a more diverse population.
The study received support from the National Institute of Aging; National Heart Lung and Blood Institute; Alzheimer’s Association; Fonds de Recherche du Québec-Santé; Canadian Consortium of Neurodegeneration in Aging; Weston Brain Institute; Colin Adair Charitable Foundation; Swedish Research Council; Wallenberg Scholar; BrightFocus Foundation; Swedish Alzheimer Foundation; Swedish Brain Foundation; Agneta Prytz-Folkes & Gösta Folkes Foundation; European Union; Swedish State Support for Clinical Research; Alzheimer Drug Discovery Foundation; Bluefield Project, the Olav Thon Foundation, the Erling-Persson Family Foundation, Stiftelsen för Gamla Tjänarinnor, Hjärnfonden, Sweden; the UK Dementia Research Institute at UCL; National Academy of Neuropsychology; Fundação de Amparo a pesquisa do Rio Grande do Sul; Instituto Serrapilheira; and Hjärnfonden.
Dr. Pascoal reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Gout linked to smaller brain volume, higher likelihood of neurodegenerative diseases
Patients with gout may have smaller brain volumes and higher brain iron markers than people without gout, and also be more likely to develop Parkinson’s disease, probable essential tremor, and dementia, researchers in the United Kingdom report.
“We were surprised about the regions of the brain affected by gout, several of which are important for motor function. The other intriguing finding was that the risk of dementia amongst gout patients was strongly time-dependent: highest in the first 3 years after their gout diagnosis,” lead study author Anya Topiwala, BMBCh, DPhil, said in an interview.
“Our combination of traditional and genetic approaches increases the confidence that gout is causing the brain findings,” said Dr. Topiwala, a clinical research fellow and consultant psychiatrist in the Nuffield Department of Population Health at the University of Oxford, England.
“We suggest that clinicians be vigilant for cognitive and motor problems after gout diagnosis, particularly in the early stages,” she added.
Links between gout and neurodegenerative diseases debated in earlier studies
Gout, the most common inflammatory arthritis, affects around 1%-4% of people, the authors wrote, with monosodium urate crystal deposits causing acute flares of pain and swelling in joints and periarticular tissues.
Whether and how gout may affect the brain has been debated in the literature. Gout and hyperuricemia have been linked with elevated stroke risk; and although observational studies have linked hyperuricemia with lower dementia risk, especially Alzheimer’s disease, Mendelian randomization studies have had conflicting results in Alzheimer’s disease.
A novel approach that analyzes brain structure and genetics
In a study published in Nature Communications, Dr. Topiwala and her colleagues combined observational and Mendelian randomization techniques to explore relationships between gout and neurodegenerative diseases. They analyzed data from over 303,000 volunteer participants between 40 and 69 years of age recruited between 2006 and 2010 to contribute their detailed genetic and health information to the U.K. Biobank, a large-scale biomedical database and research resource.
Patients with gout tended to be older and male. At baseline, all participants’ serum urate levels were measured, and 30.8% of patients with gout reported that they currently used urate-lowering therapy.
MRI shows brain changes in patients with gout
In what the authors said is the first investigation of neuroimaging markers in patients with gout, they compared differences in gray matter volumes found in the 1,165 participants with gout and the 32,202 controls without gout who had MRI data.
They found no marked sex differences in associations. Urate was inversely linked with global brain volume and with gray and white matter volumes, and gout appeared to age global gray matter by 2 years.
Patients with gout and higher urate showed significant differences in regional gray matter volumes, especially in the cerebellum, pons, and midbrain, as well as subcortical differences in the nucleus accumbens, putamen, and caudate. They also showed significant differences in white matter tract microstructure in the fornix.
Patients with gout were more likely to develop dementia (average hazard ratio [HR] over study = 1.60), especially in the first 3 years after gout diagnosis (HR = 7.40). They were also at higher risk for vascular dementia (average HR = 2.41), compared with all-cause dementia, but not for Alzheimer’s disease (average HR = 1.62).
In asymptomatic participants though, urate and dementia were inversely linked (HR = 0.85), with no time dependence.
Gout was linked with higher incidence of Parkinson’s disease (HR = 1.43) and probable essential tremor (HR = 6.75). In asymptomatic participants, urate and Parkinson’s disease (HR = 0.89), but not probable essential tremor, were inversely linked.
Genetic analyses reinforce MRI results
Using Mendelian randomization estimates, the authors found that genetic links generally reflected their observational findings. Both genetically predicted gout and serum urate were significantly linked with regional gray matter volumes, including cerebellar, midbrain, pons, and brainstem.
They also found significant links with higher magnetic susceptibility in the putamen and caudate, markers of higher iron. But while genetically predicted gout was significantly linked with global gray matter volume, urate was not.
In males, but not in females, urate was positively linked with alcohol intake and lower socioeconomic status.
Dr. Topiwala acknowledged several limitations to the study, writing that “the results from the volunteer participants may not apply to other populations; the cross-sectional serum urate measurements may not reflect chronic exposure; and Parkinson’s disease and essential tremor may have been diagnostically confounded.”
A novel approach that suggests further related research
Asked to comment on the study, Puja Khanna, MD, MPH, a rheumatologist and clinical associate professor of medicine at the University of Michigan, Ann Arbor, called its novel use of neuroimaging interesting.
Dr. Khanna, who was not involved in the study, said she would like to know more about the role that horizontal pleiotropy – one genetic variant having independent effects on multiple traits – plays in this disease process, and about the impact of the antioxidative properties of urate in maintaining neuroprotection.
“[The] U.K. Biobank is an excellent database to look at questions of association,” John D. FitzGerald, MD, PhD, MPH, MBA, professor and clinical chief of rheumatology at the University of California, Los Angeles, said in an interview.
“This is a fairly rigorous study,” added Dr. FitzGerald, also not involved in the study. “While it has lots of strengths,” including its large sample size and Mendelian randomization, it also has “abundant weaknesses,” he added. “It is largely cross-sectional, with single urate measurement and single brain MRI.”
“Causation is the big question,” Dr. FitzGerald noted. “Does treating gout (or urate) help prevent dementia or neurodegenerative decline?”
Early diagnosis benefits patients
Dr. Khanna and Dr. FitzGerald joined the authors in advising doctors to monitor their gout patients for cognitive and motor symptoms of neurodegenerative disease.
“It is clearly important to pay close attention to the neurologic exam and history in gout, especially because it is a disease of the aging population,” Dr. Khanna advised. “Addressing dementia when gout is diagnosed can lead to prompt mitigation strategies that can hugely impact patients.”
Dr. Topiwala and her colleagues would like to investigate why the dementia risk was time-dependent. “Is this because of the acute inflammatory response in gout, or could it just be that patients with gout visit their doctors more frequently, so any cognitive problems are picked up sooner?” she asked.
The authors, and Dr. Khanna and Dr. FitzGerald, report no relevant financial relationships. The Wellcome Trust; the U.K. Medical Research Council; the European Commission Horizon 2020 research and innovation program; the British Heart Foundation; the U.S. National Institutes of Health; the Engineering and Physical Sciences Research Council; and the National Institute for Health and Care Research funded the study.
Patients with gout may have smaller brain volumes and higher brain iron markers than people without gout, and also be more likely to develop Parkinson’s disease, probable essential tremor, and dementia, researchers in the United Kingdom report.
“We were surprised about the regions of the brain affected by gout, several of which are important for motor function. The other intriguing finding was that the risk of dementia amongst gout patients was strongly time-dependent: highest in the first 3 years after their gout diagnosis,” lead study author Anya Topiwala, BMBCh, DPhil, said in an interview.
“Our combination of traditional and genetic approaches increases the confidence that gout is causing the brain findings,” said Dr. Topiwala, a clinical research fellow and consultant psychiatrist in the Nuffield Department of Population Health at the University of Oxford, England.
“We suggest that clinicians be vigilant for cognitive and motor problems after gout diagnosis, particularly in the early stages,” she added.
Links between gout and neurodegenerative diseases debated in earlier studies
Gout, the most common inflammatory arthritis, affects around 1%-4% of people, the authors wrote, with monosodium urate crystal deposits causing acute flares of pain and swelling in joints and periarticular tissues.
Whether and how gout may affect the brain has been debated in the literature. Gout and hyperuricemia have been linked with elevated stroke risk; and although observational studies have linked hyperuricemia with lower dementia risk, especially Alzheimer’s disease, Mendelian randomization studies have had conflicting results in Alzheimer’s disease.
A novel approach that analyzes brain structure and genetics
In a study published in Nature Communications, Dr. Topiwala and her colleagues combined observational and Mendelian randomization techniques to explore relationships between gout and neurodegenerative diseases. They analyzed data from over 303,000 volunteer participants between 40 and 69 years of age recruited between 2006 and 2010 to contribute their detailed genetic and health information to the U.K. Biobank, a large-scale biomedical database and research resource.
Patients with gout tended to be older and male. At baseline, all participants’ serum urate levels were measured, and 30.8% of patients with gout reported that they currently used urate-lowering therapy.
MRI shows brain changes in patients with gout
In what the authors said is the first investigation of neuroimaging markers in patients with gout, they compared differences in gray matter volumes found in the 1,165 participants with gout and the 32,202 controls without gout who had MRI data.
They found no marked sex differences in associations. Urate was inversely linked with global brain volume and with gray and white matter volumes, and gout appeared to age global gray matter by 2 years.
Patients with gout and higher urate showed significant differences in regional gray matter volumes, especially in the cerebellum, pons, and midbrain, as well as subcortical differences in the nucleus accumbens, putamen, and caudate. They also showed significant differences in white matter tract microstructure in the fornix.
Patients with gout were more likely to develop dementia (average hazard ratio [HR] over study = 1.60), especially in the first 3 years after gout diagnosis (HR = 7.40). They were also at higher risk for vascular dementia (average HR = 2.41), compared with all-cause dementia, but not for Alzheimer’s disease (average HR = 1.62).
In asymptomatic participants though, urate and dementia were inversely linked (HR = 0.85), with no time dependence.
Gout was linked with higher incidence of Parkinson’s disease (HR = 1.43) and probable essential tremor (HR = 6.75). In asymptomatic participants, urate and Parkinson’s disease (HR = 0.89), but not probable essential tremor, were inversely linked.
Genetic analyses reinforce MRI results
Using Mendelian randomization estimates, the authors found that genetic links generally reflected their observational findings. Both genetically predicted gout and serum urate were significantly linked with regional gray matter volumes, including cerebellar, midbrain, pons, and brainstem.
They also found significant links with higher magnetic susceptibility in the putamen and caudate, markers of higher iron. But while genetically predicted gout was significantly linked with global gray matter volume, urate was not.
In males, but not in females, urate was positively linked with alcohol intake and lower socioeconomic status.
Dr. Topiwala acknowledged several limitations to the study, writing that “the results from the volunteer participants may not apply to other populations; the cross-sectional serum urate measurements may not reflect chronic exposure; and Parkinson’s disease and essential tremor may have been diagnostically confounded.”
A novel approach that suggests further related research
Asked to comment on the study, Puja Khanna, MD, MPH, a rheumatologist and clinical associate professor of medicine at the University of Michigan, Ann Arbor, called its novel use of neuroimaging interesting.
Dr. Khanna, who was not involved in the study, said she would like to know more about the role that horizontal pleiotropy – one genetic variant having independent effects on multiple traits – plays in this disease process, and about the impact of the antioxidative properties of urate in maintaining neuroprotection.
“[The] U.K. Biobank is an excellent database to look at questions of association,” John D. FitzGerald, MD, PhD, MPH, MBA, professor and clinical chief of rheumatology at the University of California, Los Angeles, said in an interview.
“This is a fairly rigorous study,” added Dr. FitzGerald, also not involved in the study. “While it has lots of strengths,” including its large sample size and Mendelian randomization, it also has “abundant weaknesses,” he added. “It is largely cross-sectional, with single urate measurement and single brain MRI.”
“Causation is the big question,” Dr. FitzGerald noted. “Does treating gout (or urate) help prevent dementia or neurodegenerative decline?”
Early diagnosis benefits patients
Dr. Khanna and Dr. FitzGerald joined the authors in advising doctors to monitor their gout patients for cognitive and motor symptoms of neurodegenerative disease.
“It is clearly important to pay close attention to the neurologic exam and history in gout, especially because it is a disease of the aging population,” Dr. Khanna advised. “Addressing dementia when gout is diagnosed can lead to prompt mitigation strategies that can hugely impact patients.”
Dr. Topiwala and her colleagues would like to investigate why the dementia risk was time-dependent. “Is this because of the acute inflammatory response in gout, or could it just be that patients with gout visit their doctors more frequently, so any cognitive problems are picked up sooner?” she asked.
The authors, and Dr. Khanna and Dr. FitzGerald, report no relevant financial relationships. The Wellcome Trust; the U.K. Medical Research Council; the European Commission Horizon 2020 research and innovation program; the British Heart Foundation; the U.S. National Institutes of Health; the Engineering and Physical Sciences Research Council; and the National Institute for Health and Care Research funded the study.
Patients with gout may have smaller brain volumes and higher brain iron markers than people without gout, and also be more likely to develop Parkinson’s disease, probable essential tremor, and dementia, researchers in the United Kingdom report.
“We were surprised about the regions of the brain affected by gout, several of which are important for motor function. The other intriguing finding was that the risk of dementia amongst gout patients was strongly time-dependent: highest in the first 3 years after their gout diagnosis,” lead study author Anya Topiwala, BMBCh, DPhil, said in an interview.
“Our combination of traditional and genetic approaches increases the confidence that gout is causing the brain findings,” said Dr. Topiwala, a clinical research fellow and consultant psychiatrist in the Nuffield Department of Population Health at the University of Oxford, England.
“We suggest that clinicians be vigilant for cognitive and motor problems after gout diagnosis, particularly in the early stages,” she added.
Links between gout and neurodegenerative diseases debated in earlier studies
Gout, the most common inflammatory arthritis, affects around 1%-4% of people, the authors wrote, with monosodium urate crystal deposits causing acute flares of pain and swelling in joints and periarticular tissues.
Whether and how gout may affect the brain has been debated in the literature. Gout and hyperuricemia have been linked with elevated stroke risk; and although observational studies have linked hyperuricemia with lower dementia risk, especially Alzheimer’s disease, Mendelian randomization studies have had conflicting results in Alzheimer’s disease.
A novel approach that analyzes brain structure and genetics
In a study published in Nature Communications, Dr. Topiwala and her colleagues combined observational and Mendelian randomization techniques to explore relationships between gout and neurodegenerative diseases. They analyzed data from over 303,000 volunteer participants between 40 and 69 years of age recruited between 2006 and 2010 to contribute their detailed genetic and health information to the U.K. Biobank, a large-scale biomedical database and research resource.
Patients with gout tended to be older and male. At baseline, all participants’ serum urate levels were measured, and 30.8% of patients with gout reported that they currently used urate-lowering therapy.
MRI shows brain changes in patients with gout
In what the authors said is the first investigation of neuroimaging markers in patients with gout, they compared differences in gray matter volumes found in the 1,165 participants with gout and the 32,202 controls without gout who had MRI data.
They found no marked sex differences in associations. Urate was inversely linked with global brain volume and with gray and white matter volumes, and gout appeared to age global gray matter by 2 years.
Patients with gout and higher urate showed significant differences in regional gray matter volumes, especially in the cerebellum, pons, and midbrain, as well as subcortical differences in the nucleus accumbens, putamen, and caudate. They also showed significant differences in white matter tract microstructure in the fornix.
Patients with gout were more likely to develop dementia (average hazard ratio [HR] over study = 1.60), especially in the first 3 years after gout diagnosis (HR = 7.40). They were also at higher risk for vascular dementia (average HR = 2.41), compared with all-cause dementia, but not for Alzheimer’s disease (average HR = 1.62).
In asymptomatic participants though, urate and dementia were inversely linked (HR = 0.85), with no time dependence.
Gout was linked with higher incidence of Parkinson’s disease (HR = 1.43) and probable essential tremor (HR = 6.75). In asymptomatic participants, urate and Parkinson’s disease (HR = 0.89), but not probable essential tremor, were inversely linked.
Genetic analyses reinforce MRI results
Using Mendelian randomization estimates, the authors found that genetic links generally reflected their observational findings. Both genetically predicted gout and serum urate were significantly linked with regional gray matter volumes, including cerebellar, midbrain, pons, and brainstem.
They also found significant links with higher magnetic susceptibility in the putamen and caudate, markers of higher iron. But while genetically predicted gout was significantly linked with global gray matter volume, urate was not.
In males, but not in females, urate was positively linked with alcohol intake and lower socioeconomic status.
Dr. Topiwala acknowledged several limitations to the study, writing that “the results from the volunteer participants may not apply to other populations; the cross-sectional serum urate measurements may not reflect chronic exposure; and Parkinson’s disease and essential tremor may have been diagnostically confounded.”
A novel approach that suggests further related research
Asked to comment on the study, Puja Khanna, MD, MPH, a rheumatologist and clinical associate professor of medicine at the University of Michigan, Ann Arbor, called its novel use of neuroimaging interesting.
Dr. Khanna, who was not involved in the study, said she would like to know more about the role that horizontal pleiotropy – one genetic variant having independent effects on multiple traits – plays in this disease process, and about the impact of the antioxidative properties of urate in maintaining neuroprotection.
“[The] U.K. Biobank is an excellent database to look at questions of association,” John D. FitzGerald, MD, PhD, MPH, MBA, professor and clinical chief of rheumatology at the University of California, Los Angeles, said in an interview.
“This is a fairly rigorous study,” added Dr. FitzGerald, also not involved in the study. “While it has lots of strengths,” including its large sample size and Mendelian randomization, it also has “abundant weaknesses,” he added. “It is largely cross-sectional, with single urate measurement and single brain MRI.”
“Causation is the big question,” Dr. FitzGerald noted. “Does treating gout (or urate) help prevent dementia or neurodegenerative decline?”
Early diagnosis benefits patients
Dr. Khanna and Dr. FitzGerald joined the authors in advising doctors to monitor their gout patients for cognitive and motor symptoms of neurodegenerative disease.
“It is clearly important to pay close attention to the neurologic exam and history in gout, especially because it is a disease of the aging population,” Dr. Khanna advised. “Addressing dementia when gout is diagnosed can lead to prompt mitigation strategies that can hugely impact patients.”
Dr. Topiwala and her colleagues would like to investigate why the dementia risk was time-dependent. “Is this because of the acute inflammatory response in gout, or could it just be that patients with gout visit their doctors more frequently, so any cognitive problems are picked up sooner?” she asked.
The authors, and Dr. Khanna and Dr. FitzGerald, report no relevant financial relationships. The Wellcome Trust; the U.K. Medical Research Council; the European Commission Horizon 2020 research and innovation program; the British Heart Foundation; the U.S. National Institutes of Health; the Engineering and Physical Sciences Research Council; and the National Institute for Health and Care Research funded the study.
FROM NATURE COMMUNICATIONS
Game-changing Alzheimer’s research: The latest on biomarkers
The field of neurodegenerative dementias, particularly Alzheimer’s disease (AD), has been revolutionized by the development of imaging and cerebrospinal fluid biomarkers and is on the brink of a new development: emerging plasma biomarkers. Research now recognizes the relationship between the cognitive-behavioral syndromic diagnosis (that is, the illness) and the etiologic diagnosis (the disease) – and the need to consider each separately when developing a diagnostic formulation. The National Institute on Aging and Alzheimer’s Association Research Framework uses the amyloid, tau, and neurodegeneration system to define AD biologically in living patients. Here is an overview of the framework, which requires biomarker evidence of amyloid plaques (amyloid positivity) and neurofibrillary tangles (tau positivity), with evidence of neurodegeneration (neurodegeneration positivity) to support the diagnosis.
The diagnostic approach for symptomatic patients
The differential diagnosis in symptomatic patients with mild cognitive impairment (MCI), mild behavioral impairment, or dementia is broad and includes multiple neurodegenerative diseases (for example, AD, frontotemporal lobar degeneration, dementia with Lewy bodies, argyrophilic grain disease, hippocampal sclerosis); vascular ischemic brain injury (for example, stroke); tumors; infectious, inflammatory, paraneoplastic, or demyelinating diseases; trauma; hydrocephalus; toxic/metabolic insults; and other rare diseases. The patient’s clinical syndrome narrows the differential diagnosis.
Once the clinician has a prioritized differential diagnosis of the brain disease or condition that is probably causing or contributing to the patient’s signs and symptoms, they can then select appropriate assessments and tests, typically starting with a laboratory panel and brain MRI. Strong evidence backed by practice recommendations also supports the use of fluorodeoxyglucose PET as a marker of functional brain abnormalities associated with dementia. Although molecular biomarkers are typically considered at the later stage of the clinical workup, the anticipated future availability of plasma biomarkers will probably change the timing of molecular biomarker assessment in patients with suspected cognitive impairment owing to AD.
Molecular PET biomarkers
Three PET tracers approved by the U.S. Food and Drug Administration for the detection of cerebral amyloid plaques have high sensitivity (89%-98%) and specificity (88%-100%), compared with autopsy, the gold standard diagnostic tool. However, these scans are costly and are not reimbursed by Medicare and Medicaid. Because all amyloid PET scans are covered by the Veterans Administration, this test is more readily accessible for patients receiving VA benefits.
The appropriate-use criteria developed by the Amyloid Imaging Task Force recommends amyloid PET for patients with persistent or progressive MCI or dementia. In such patients, a negative amyloid PET scan would strongly weigh against AD, supporting a differential diagnosis of other etiologies. Although a positive amyloid PET scan in patients with MCI or dementia indicates the presence of amyloid plaques, it does not necessarily confirm AD as the cause. Cerebral amyloid plaques may coexist with other pathologies and increase with age, even in cognitively normal individuals.
The IDEAS study looked at the clinical utility of amyloid PET in a real-world dementia specialist setting. In the study, dementia subspecialists documented their presumed etiologic diagnosis (and level of confidence) before and after amyloid PET. Of the 11,409 patients who completed the study, the etiologic diagnosis changed from AD to non-AD in just over 25% of cases and from non-AD to AD in 10.5%. Clinical management changed in about 60% of patients with MCI and 63.5% of patients with dementia.
In May 2020, the FDA approved flortaucipir F-18, the first diagnostic tau radiotracer for use with PET to estimate the density and distribution of aggregated tau neurofibrillary tangles in adults with cognitive impairment undergoing evaluation for AD. Regulatory approval of flortaucipir F-18 was based on findings from two clinical trials of terminally ill patients who were followed to autopsy. The studies included patients with a spectrum of clinically diagnosed dementias and those with normal cognition. The primary outcome of the studies was accurate visual interpretation of the images in detecting advanced AD tau neurofibrillary tangle pathology (Braak stage V or VI tau pathology). Sensitivity of five trained readers ranged from 68% to 86%, and specificity ranged from 63% to 100%; interrater agreement was 0.87. Tau PET is not yet reimbursed and is therefore not yet readily available in the clinical setting. Moreover, appropriate use criteria have not yet been published.
Molecular fluid biomarkers
Cerebrospinal fluid (CSF) analysis is currently the most readily available and reimbursed test to aid in diagnosing AD, with appropriate-use criteria for patients with suspected AD. CSF biomarkers for AD are useful in cognitively impaired patients when the etiologic diagnosis is equivocal, there is only an intermediate level of diagnostic confidence, or there is very high confidence in the etiologic diagnosis. Testing for CSF biomarkers is also recommended for patients at very early clinical stages (for example, early MCI) or with atypical clinical presentations.
A decreased concentration of amyloid-beta 42 in CSF is a marker of amyloid neuritic plaques in the brain. An increased concentration of total tau in CSF reflects injury to neurons, and an increased concentration of specific isoforms of hyperphosphorylated tau reflects neurofibrillary tangles. Presently, the ratios of t-tau to amyloid-beta 42, amyloid-beta 42 to amyloid-beta 40, and phosphorylated-tau 181 to amyloid-beta 42 are the best-performing markers of AD neuropathologic changes and are more accurate than assessing individual biomarkers. These CSF biomarkers of AD have been validated against autopsy, and ratio values of CSF amyloid-beta 42 have been further validated against amyloid PET, with overall sensitivity and specificity of approximately 90% and 84%, respectively.
Some of the most exciting recent advances in AD center around the measurement of these proteins and others in plasma. Appropriate-use criteria for plasma biomarkers in the evaluation of patients with cognitive impairment were published in 2022. In addition to their use in clinical trials, these criteria cautiously recommend using these biomarkers in specialized memory clinics in the diagnostic workup of patients with cognitive symptoms, along with confirmatory CSF markers or PET. Additional data are needed before plasma biomarkers of AD are used as standalone diagnostic markers or considered in the primary care setting.
We have made remarkable progress toward more precise molecular diagnosis of brain diseases underlying cognitive impairment and dementia. Ongoing efforts to evaluate the utility of these measures in clinical practice include the need to increase diversity of patients and providers. Ultimately, the tremendous progress in molecular biomarkers for the diseases causing dementia will help the field work toward our common goal of early and accurate diagnosis, better management, and hope for people living with these diseases.
Bradford C. Dickerson, MD, MMSc, is a professor, department of neurology, Harvard Medical School, and director, Frontotemporal Disorders Unit, department of neurology, at Massachusetts General Hospital, both in Boston.
A version of this article first appeared on Medscape.com.
The field of neurodegenerative dementias, particularly Alzheimer’s disease (AD), has been revolutionized by the development of imaging and cerebrospinal fluid biomarkers and is on the brink of a new development: emerging plasma biomarkers. Research now recognizes the relationship between the cognitive-behavioral syndromic diagnosis (that is, the illness) and the etiologic diagnosis (the disease) – and the need to consider each separately when developing a diagnostic formulation. The National Institute on Aging and Alzheimer’s Association Research Framework uses the amyloid, tau, and neurodegeneration system to define AD biologically in living patients. Here is an overview of the framework, which requires biomarker evidence of amyloid plaques (amyloid positivity) and neurofibrillary tangles (tau positivity), with evidence of neurodegeneration (neurodegeneration positivity) to support the diagnosis.
The diagnostic approach for symptomatic patients
The differential diagnosis in symptomatic patients with mild cognitive impairment (MCI), mild behavioral impairment, or dementia is broad and includes multiple neurodegenerative diseases (for example, AD, frontotemporal lobar degeneration, dementia with Lewy bodies, argyrophilic grain disease, hippocampal sclerosis); vascular ischemic brain injury (for example, stroke); tumors; infectious, inflammatory, paraneoplastic, or demyelinating diseases; trauma; hydrocephalus; toxic/metabolic insults; and other rare diseases. The patient’s clinical syndrome narrows the differential diagnosis.
Once the clinician has a prioritized differential diagnosis of the brain disease or condition that is probably causing or contributing to the patient’s signs and symptoms, they can then select appropriate assessments and tests, typically starting with a laboratory panel and brain MRI. Strong evidence backed by practice recommendations also supports the use of fluorodeoxyglucose PET as a marker of functional brain abnormalities associated with dementia. Although molecular biomarkers are typically considered at the later stage of the clinical workup, the anticipated future availability of plasma biomarkers will probably change the timing of molecular biomarker assessment in patients with suspected cognitive impairment owing to AD.
Molecular PET biomarkers
Three PET tracers approved by the U.S. Food and Drug Administration for the detection of cerebral amyloid plaques have high sensitivity (89%-98%) and specificity (88%-100%), compared with autopsy, the gold standard diagnostic tool. However, these scans are costly and are not reimbursed by Medicare and Medicaid. Because all amyloid PET scans are covered by the Veterans Administration, this test is more readily accessible for patients receiving VA benefits.
The appropriate-use criteria developed by the Amyloid Imaging Task Force recommends amyloid PET for patients with persistent or progressive MCI or dementia. In such patients, a negative amyloid PET scan would strongly weigh against AD, supporting a differential diagnosis of other etiologies. Although a positive amyloid PET scan in patients with MCI or dementia indicates the presence of amyloid plaques, it does not necessarily confirm AD as the cause. Cerebral amyloid plaques may coexist with other pathologies and increase with age, even in cognitively normal individuals.
The IDEAS study looked at the clinical utility of amyloid PET in a real-world dementia specialist setting. In the study, dementia subspecialists documented their presumed etiologic diagnosis (and level of confidence) before and after amyloid PET. Of the 11,409 patients who completed the study, the etiologic diagnosis changed from AD to non-AD in just over 25% of cases and from non-AD to AD in 10.5%. Clinical management changed in about 60% of patients with MCI and 63.5% of patients with dementia.
In May 2020, the FDA approved flortaucipir F-18, the first diagnostic tau radiotracer for use with PET to estimate the density and distribution of aggregated tau neurofibrillary tangles in adults with cognitive impairment undergoing evaluation for AD. Regulatory approval of flortaucipir F-18 was based on findings from two clinical trials of terminally ill patients who were followed to autopsy. The studies included patients with a spectrum of clinically diagnosed dementias and those with normal cognition. The primary outcome of the studies was accurate visual interpretation of the images in detecting advanced AD tau neurofibrillary tangle pathology (Braak stage V or VI tau pathology). Sensitivity of five trained readers ranged from 68% to 86%, and specificity ranged from 63% to 100%; interrater agreement was 0.87. Tau PET is not yet reimbursed and is therefore not yet readily available in the clinical setting. Moreover, appropriate use criteria have not yet been published.
Molecular fluid biomarkers
Cerebrospinal fluid (CSF) analysis is currently the most readily available and reimbursed test to aid in diagnosing AD, with appropriate-use criteria for patients with suspected AD. CSF biomarkers for AD are useful in cognitively impaired patients when the etiologic diagnosis is equivocal, there is only an intermediate level of diagnostic confidence, or there is very high confidence in the etiologic diagnosis. Testing for CSF biomarkers is also recommended for patients at very early clinical stages (for example, early MCI) or with atypical clinical presentations.
A decreased concentration of amyloid-beta 42 in CSF is a marker of amyloid neuritic plaques in the brain. An increased concentration of total tau in CSF reflects injury to neurons, and an increased concentration of specific isoforms of hyperphosphorylated tau reflects neurofibrillary tangles. Presently, the ratios of t-tau to amyloid-beta 42, amyloid-beta 42 to amyloid-beta 40, and phosphorylated-tau 181 to amyloid-beta 42 are the best-performing markers of AD neuropathologic changes and are more accurate than assessing individual biomarkers. These CSF biomarkers of AD have been validated against autopsy, and ratio values of CSF amyloid-beta 42 have been further validated against amyloid PET, with overall sensitivity and specificity of approximately 90% and 84%, respectively.
Some of the most exciting recent advances in AD center around the measurement of these proteins and others in plasma. Appropriate-use criteria for plasma biomarkers in the evaluation of patients with cognitive impairment were published in 2022. In addition to their use in clinical trials, these criteria cautiously recommend using these biomarkers in specialized memory clinics in the diagnostic workup of patients with cognitive symptoms, along with confirmatory CSF markers or PET. Additional data are needed before plasma biomarkers of AD are used as standalone diagnostic markers or considered in the primary care setting.
We have made remarkable progress toward more precise molecular diagnosis of brain diseases underlying cognitive impairment and dementia. Ongoing efforts to evaluate the utility of these measures in clinical practice include the need to increase diversity of patients and providers. Ultimately, the tremendous progress in molecular biomarkers for the diseases causing dementia will help the field work toward our common goal of early and accurate diagnosis, better management, and hope for people living with these diseases.
Bradford C. Dickerson, MD, MMSc, is a professor, department of neurology, Harvard Medical School, and director, Frontotemporal Disorders Unit, department of neurology, at Massachusetts General Hospital, both in Boston.
A version of this article first appeared on Medscape.com.
The field of neurodegenerative dementias, particularly Alzheimer’s disease (AD), has been revolutionized by the development of imaging and cerebrospinal fluid biomarkers and is on the brink of a new development: emerging plasma biomarkers. Research now recognizes the relationship between the cognitive-behavioral syndromic diagnosis (that is, the illness) and the etiologic diagnosis (the disease) – and the need to consider each separately when developing a diagnostic formulation. The National Institute on Aging and Alzheimer’s Association Research Framework uses the amyloid, tau, and neurodegeneration system to define AD biologically in living patients. Here is an overview of the framework, which requires biomarker evidence of amyloid plaques (amyloid positivity) and neurofibrillary tangles (tau positivity), with evidence of neurodegeneration (neurodegeneration positivity) to support the diagnosis.
The diagnostic approach for symptomatic patients
The differential diagnosis in symptomatic patients with mild cognitive impairment (MCI), mild behavioral impairment, or dementia is broad and includes multiple neurodegenerative diseases (for example, AD, frontotemporal lobar degeneration, dementia with Lewy bodies, argyrophilic grain disease, hippocampal sclerosis); vascular ischemic brain injury (for example, stroke); tumors; infectious, inflammatory, paraneoplastic, or demyelinating diseases; trauma; hydrocephalus; toxic/metabolic insults; and other rare diseases. The patient’s clinical syndrome narrows the differential diagnosis.
Once the clinician has a prioritized differential diagnosis of the brain disease or condition that is probably causing or contributing to the patient’s signs and symptoms, they can then select appropriate assessments and tests, typically starting with a laboratory panel and brain MRI. Strong evidence backed by practice recommendations also supports the use of fluorodeoxyglucose PET as a marker of functional brain abnormalities associated with dementia. Although molecular biomarkers are typically considered at the later stage of the clinical workup, the anticipated future availability of plasma biomarkers will probably change the timing of molecular biomarker assessment in patients with suspected cognitive impairment owing to AD.
Molecular PET biomarkers
Three PET tracers approved by the U.S. Food and Drug Administration for the detection of cerebral amyloid plaques have high sensitivity (89%-98%) and specificity (88%-100%), compared with autopsy, the gold standard diagnostic tool. However, these scans are costly and are not reimbursed by Medicare and Medicaid. Because all amyloid PET scans are covered by the Veterans Administration, this test is more readily accessible for patients receiving VA benefits.
The appropriate-use criteria developed by the Amyloid Imaging Task Force recommends amyloid PET for patients with persistent or progressive MCI or dementia. In such patients, a negative amyloid PET scan would strongly weigh against AD, supporting a differential diagnosis of other etiologies. Although a positive amyloid PET scan in patients with MCI or dementia indicates the presence of amyloid plaques, it does not necessarily confirm AD as the cause. Cerebral amyloid plaques may coexist with other pathologies and increase with age, even in cognitively normal individuals.
The IDEAS study looked at the clinical utility of amyloid PET in a real-world dementia specialist setting. In the study, dementia subspecialists documented their presumed etiologic diagnosis (and level of confidence) before and after amyloid PET. Of the 11,409 patients who completed the study, the etiologic diagnosis changed from AD to non-AD in just over 25% of cases and from non-AD to AD in 10.5%. Clinical management changed in about 60% of patients with MCI and 63.5% of patients with dementia.
In May 2020, the FDA approved flortaucipir F-18, the first diagnostic tau radiotracer for use with PET to estimate the density and distribution of aggregated tau neurofibrillary tangles in adults with cognitive impairment undergoing evaluation for AD. Regulatory approval of flortaucipir F-18 was based on findings from two clinical trials of terminally ill patients who were followed to autopsy. The studies included patients with a spectrum of clinically diagnosed dementias and those with normal cognition. The primary outcome of the studies was accurate visual interpretation of the images in detecting advanced AD tau neurofibrillary tangle pathology (Braak stage V or VI tau pathology). Sensitivity of five trained readers ranged from 68% to 86%, and specificity ranged from 63% to 100%; interrater agreement was 0.87. Tau PET is not yet reimbursed and is therefore not yet readily available in the clinical setting. Moreover, appropriate use criteria have not yet been published.
Molecular fluid biomarkers
Cerebrospinal fluid (CSF) analysis is currently the most readily available and reimbursed test to aid in diagnosing AD, with appropriate-use criteria for patients with suspected AD. CSF biomarkers for AD are useful in cognitively impaired patients when the etiologic diagnosis is equivocal, there is only an intermediate level of diagnostic confidence, or there is very high confidence in the etiologic diagnosis. Testing for CSF biomarkers is also recommended for patients at very early clinical stages (for example, early MCI) or with atypical clinical presentations.
A decreased concentration of amyloid-beta 42 in CSF is a marker of amyloid neuritic plaques in the brain. An increased concentration of total tau in CSF reflects injury to neurons, and an increased concentration of specific isoforms of hyperphosphorylated tau reflects neurofibrillary tangles. Presently, the ratios of t-tau to amyloid-beta 42, amyloid-beta 42 to amyloid-beta 40, and phosphorylated-tau 181 to amyloid-beta 42 are the best-performing markers of AD neuropathologic changes and are more accurate than assessing individual biomarkers. These CSF biomarkers of AD have been validated against autopsy, and ratio values of CSF amyloid-beta 42 have been further validated against amyloid PET, with overall sensitivity and specificity of approximately 90% and 84%, respectively.
Some of the most exciting recent advances in AD center around the measurement of these proteins and others in plasma. Appropriate-use criteria for plasma biomarkers in the evaluation of patients with cognitive impairment were published in 2022. In addition to their use in clinical trials, these criteria cautiously recommend using these biomarkers in specialized memory clinics in the diagnostic workup of patients with cognitive symptoms, along with confirmatory CSF markers or PET. Additional data are needed before plasma biomarkers of AD are used as standalone diagnostic markers or considered in the primary care setting.
We have made remarkable progress toward more precise molecular diagnosis of brain diseases underlying cognitive impairment and dementia. Ongoing efforts to evaluate the utility of these measures in clinical practice include the need to increase diversity of patients and providers. Ultimately, the tremendous progress in molecular biomarkers for the diseases causing dementia will help the field work toward our common goal of early and accurate diagnosis, better management, and hope for people living with these diseases.
Bradford C. Dickerson, MD, MMSc, is a professor, department of neurology, Harvard Medical School, and director, Frontotemporal Disorders Unit, department of neurology, at Massachusetts General Hospital, both in Boston.
A version of this article first appeared on Medscape.com.
Flavanol supplement improves memory in adults with poor diets
Taking a daily flavanol supplement improves hippocampal-dependent memory in older adults who have a relatively poor diet, results of a large new study suggest.
There’s increasing evidence that certain nutrients are important for the aging body and brain, study investigator Scott Small, MD, the Boris and Rose Katz Professor of Neurology, Columbia University Vagelos College of Physicians and Surgeons, New York, told this news organization.
“With this new study, I think we can begin to say flavanols might be the first one that really is a nutrient for the aging brain.”
These findings, said Dr. Small, represent “the beginning of a new era” that will eventually lead to formal recommendations” related to ideal intake of flavanols to reduce cognitive aging.
The findings were published online in the Proceedings of the National Academy of Science.
Better cognitive aging
Cognitive aging refers to the decline in cognitive abilities that are not thought to be caused by neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. Cognitive aging targets two areas of the brain: the hippocampus, which is related to memory function, and the prefrontal cortex, which is related to attention and executive function.
Previous research has linked flavanols, which are found in foods like apples, pears, berries, and cocoa beans, to improved cognitive aging. The evidence shows that consuming these nutrients might be associated with the hippocampal-dependent memory component of cognitive aging.
The new study, known as COcoa Supplement and Multivitamin Outcomes Study-Web (COSMOS-Web), included 3,562 generally healthy men and women, mean age 71 years, who were mostly well-educated and non-Hispanic/non-Latinx White individuals.
Participants were randomly assigned to receive oral flavanol-containing cocoa extract (500 mg of cocoa flavanols, including 80 mg of epicatechin) or a placebo daily.
The primary endpoint was hippocampal-dependent memory at year 1 as assessed with the ModRey, a neuropsychological test designed to measure hippocampal function.
Results showed participants in both groups had a typical learning (practice) effect, with similar improvements (d = 0.025; P = .42).
Researchers used other tests to measure cognition: the Color/Directional Flanker Task, a measure of prefrontal cortex function, and the ModBent, a measure that’s sensitive to dentate gyrus function. The flavanol intervention did not affect ModBent results or performance on the Flanker test after 1 year.
However, it was a different story for those with a poor diet at baseline. Researchers stratified participants into tertiles on the basis of diet quality as measured by the Healthy Eating Index (HEI) scores. Those in the lowest tertile had poorer baseline hippocampal-dependent memory performance but not memory related to the prefrontal cortex.
The flavanol intervention improved performance on the ModRey test, compared with placebo in participants in the low HEI tertile (overall effect: d = 0.086; P = .011) but not among those with a medium or high HEI at baseline.
“We confirmed that the flavanol intervention only benefits people who are relatively deficient at baseline,” said Dr. Small.
The correlation with hippocampal-dependent memory was confirmed in a subset of 1,361 study participants who provided a urine sample. Researchers measured urinary 5-(3′,4′-dihydroxyphenyl)-gamma-valerolactone metabolite (gVLM) concentrations, a validated biomarker of flavanol consumption.
After stratifying these results into tertiles, researchers found performance on the ModRey was significantly improved with the dietary flavanol intervention (overall effect: d = 0.141; P = .006) in the lowest gVLM tertile.
Memory restored
When participants in the lowest tertile consumed the supplement, “their flavanol levels went back to normal, and when that happened, their memory was restored,” said Dr. Small.
It appears that there is a sort of ceiling effect to the flavanol benefits. “It seems what you need to do is normalize your flavanol levels; if you go above normal, there was no evidence that your memory keeps on getting better,” said Dr. Small.
The study included only older adults, so it’s unclear what the impact of flavanol supplementation is in younger adults. But cognitive aging “begins its slippery side” in the 40s, said Dr. Small. “If this is truly a nutrient that is taken to prevent that slide from happening, it might be beneficial to start in our 40s.”
He recognized that the effect size is not large but said this is “very dependent” on baseline factors and most study participants had a rather healthy diet. “None of our participants were really highly deficient” in flavanols, he said.
“To see a stronger effect size, we need to do another study where we recruit people who are very low, truly deficient, in flavanols, and then see what happens.”
Showing that flavanols are linked to the hippocampal and not to the prefrontal component of cognitive aging “speaks to the mechanism,” said Dr. Small.
Though the exact mechanism linking flavanols with enhanced memory isn’t clear, there are some clues; for example, research suggests cognitive aging affects the dentate gyrus, a subregion of the hippocampus.
The flavanol supplements were well tolerated. “I can say with close to certainty that this is very safe,” said Dr. Small, adding the flavanols have now been used in numerous studies.
The findings suggest flavanol consumption might be part of future dietary guidelines. “I suspect that once there is sufficient evidence, flavanols will be part of the dietary recommendations for healthy aging,” said Dr. Small.
A word of caution
Heather M. Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, said that though science suggests a balanced diet is good for overall brain health, no single food, beverage, ingredient, vitamin, or supplement has yet been proven to prevent dementia, treat or cure Alzheimer’s, or benefit cognitive function or brain health.
Experts agree the best source of vitamins and other nutrients is from whole foods as part of a balanced diet. “We recognize that, for a variety of reasons, this may not always be possible,” said Dr. Snyder.
However, she noted, dietary supplements are not subject to the same rigorous review and regulation process as medications.
“The Alzheimer’s Association strongly encourages individuals to have conversations with their physicians about all medications and dietary supplements they are currently taking or interested in starting.”
COSMOS is supported by an investigator-initiated grant from Mars Edge, a segment of Mars, company engaged in flavanol research and flavanol-related commercial activities, which included infrastructure support and the donation of study pills and packaging. Small reports receiving an unrestricted research grant from Mars.
A version of this article first appeared on Medscape.com.
Taking a daily flavanol supplement improves hippocampal-dependent memory in older adults who have a relatively poor diet, results of a large new study suggest.
There’s increasing evidence that certain nutrients are important for the aging body and brain, study investigator Scott Small, MD, the Boris and Rose Katz Professor of Neurology, Columbia University Vagelos College of Physicians and Surgeons, New York, told this news organization.
“With this new study, I think we can begin to say flavanols might be the first one that really is a nutrient for the aging brain.”
These findings, said Dr. Small, represent “the beginning of a new era” that will eventually lead to formal recommendations” related to ideal intake of flavanols to reduce cognitive aging.
The findings were published online in the Proceedings of the National Academy of Science.
Better cognitive aging
Cognitive aging refers to the decline in cognitive abilities that are not thought to be caused by neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. Cognitive aging targets two areas of the brain: the hippocampus, which is related to memory function, and the prefrontal cortex, which is related to attention and executive function.
Previous research has linked flavanols, which are found in foods like apples, pears, berries, and cocoa beans, to improved cognitive aging. The evidence shows that consuming these nutrients might be associated with the hippocampal-dependent memory component of cognitive aging.
The new study, known as COcoa Supplement and Multivitamin Outcomes Study-Web (COSMOS-Web), included 3,562 generally healthy men and women, mean age 71 years, who were mostly well-educated and non-Hispanic/non-Latinx White individuals.
Participants were randomly assigned to receive oral flavanol-containing cocoa extract (500 mg of cocoa flavanols, including 80 mg of epicatechin) or a placebo daily.
The primary endpoint was hippocampal-dependent memory at year 1 as assessed with the ModRey, a neuropsychological test designed to measure hippocampal function.
Results showed participants in both groups had a typical learning (practice) effect, with similar improvements (d = 0.025; P = .42).
Researchers used other tests to measure cognition: the Color/Directional Flanker Task, a measure of prefrontal cortex function, and the ModBent, a measure that’s sensitive to dentate gyrus function. The flavanol intervention did not affect ModBent results or performance on the Flanker test after 1 year.
However, it was a different story for those with a poor diet at baseline. Researchers stratified participants into tertiles on the basis of diet quality as measured by the Healthy Eating Index (HEI) scores. Those in the lowest tertile had poorer baseline hippocampal-dependent memory performance but not memory related to the prefrontal cortex.
The flavanol intervention improved performance on the ModRey test, compared with placebo in participants in the low HEI tertile (overall effect: d = 0.086; P = .011) but not among those with a medium or high HEI at baseline.
“We confirmed that the flavanol intervention only benefits people who are relatively deficient at baseline,” said Dr. Small.
The correlation with hippocampal-dependent memory was confirmed in a subset of 1,361 study participants who provided a urine sample. Researchers measured urinary 5-(3′,4′-dihydroxyphenyl)-gamma-valerolactone metabolite (gVLM) concentrations, a validated biomarker of flavanol consumption.
After stratifying these results into tertiles, researchers found performance on the ModRey was significantly improved with the dietary flavanol intervention (overall effect: d = 0.141; P = .006) in the lowest gVLM tertile.
Memory restored
When participants in the lowest tertile consumed the supplement, “their flavanol levels went back to normal, and when that happened, their memory was restored,” said Dr. Small.
It appears that there is a sort of ceiling effect to the flavanol benefits. “It seems what you need to do is normalize your flavanol levels; if you go above normal, there was no evidence that your memory keeps on getting better,” said Dr. Small.
The study included only older adults, so it’s unclear what the impact of flavanol supplementation is in younger adults. But cognitive aging “begins its slippery side” in the 40s, said Dr. Small. “If this is truly a nutrient that is taken to prevent that slide from happening, it might be beneficial to start in our 40s.”
He recognized that the effect size is not large but said this is “very dependent” on baseline factors and most study participants had a rather healthy diet. “None of our participants were really highly deficient” in flavanols, he said.
“To see a stronger effect size, we need to do another study where we recruit people who are very low, truly deficient, in flavanols, and then see what happens.”
Showing that flavanols are linked to the hippocampal and not to the prefrontal component of cognitive aging “speaks to the mechanism,” said Dr. Small.
Though the exact mechanism linking flavanols with enhanced memory isn’t clear, there are some clues; for example, research suggests cognitive aging affects the dentate gyrus, a subregion of the hippocampus.
The flavanol supplements were well tolerated. “I can say with close to certainty that this is very safe,” said Dr. Small, adding the flavanols have now been used in numerous studies.
The findings suggest flavanol consumption might be part of future dietary guidelines. “I suspect that once there is sufficient evidence, flavanols will be part of the dietary recommendations for healthy aging,” said Dr. Small.
A word of caution
Heather M. Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, said that though science suggests a balanced diet is good for overall brain health, no single food, beverage, ingredient, vitamin, or supplement has yet been proven to prevent dementia, treat or cure Alzheimer’s, or benefit cognitive function or brain health.
Experts agree the best source of vitamins and other nutrients is from whole foods as part of a balanced diet. “We recognize that, for a variety of reasons, this may not always be possible,” said Dr. Snyder.
However, she noted, dietary supplements are not subject to the same rigorous review and regulation process as medications.
“The Alzheimer’s Association strongly encourages individuals to have conversations with their physicians about all medications and dietary supplements they are currently taking or interested in starting.”
COSMOS is supported by an investigator-initiated grant from Mars Edge, a segment of Mars, company engaged in flavanol research and flavanol-related commercial activities, which included infrastructure support and the donation of study pills and packaging. Small reports receiving an unrestricted research grant from Mars.
A version of this article first appeared on Medscape.com.
Taking a daily flavanol supplement improves hippocampal-dependent memory in older adults who have a relatively poor diet, results of a large new study suggest.
There’s increasing evidence that certain nutrients are important for the aging body and brain, study investigator Scott Small, MD, the Boris and Rose Katz Professor of Neurology, Columbia University Vagelos College of Physicians and Surgeons, New York, told this news organization.
“With this new study, I think we can begin to say flavanols might be the first one that really is a nutrient for the aging brain.”
These findings, said Dr. Small, represent “the beginning of a new era” that will eventually lead to formal recommendations” related to ideal intake of flavanols to reduce cognitive aging.
The findings were published online in the Proceedings of the National Academy of Science.
Better cognitive aging
Cognitive aging refers to the decline in cognitive abilities that are not thought to be caused by neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease. Cognitive aging targets two areas of the brain: the hippocampus, which is related to memory function, and the prefrontal cortex, which is related to attention and executive function.
Previous research has linked flavanols, which are found in foods like apples, pears, berries, and cocoa beans, to improved cognitive aging. The evidence shows that consuming these nutrients might be associated with the hippocampal-dependent memory component of cognitive aging.
The new study, known as COcoa Supplement and Multivitamin Outcomes Study-Web (COSMOS-Web), included 3,562 generally healthy men and women, mean age 71 years, who were mostly well-educated and non-Hispanic/non-Latinx White individuals.
Participants were randomly assigned to receive oral flavanol-containing cocoa extract (500 mg of cocoa flavanols, including 80 mg of epicatechin) or a placebo daily.
The primary endpoint was hippocampal-dependent memory at year 1 as assessed with the ModRey, a neuropsychological test designed to measure hippocampal function.
Results showed participants in both groups had a typical learning (practice) effect, with similar improvements (d = 0.025; P = .42).
Researchers used other tests to measure cognition: the Color/Directional Flanker Task, a measure of prefrontal cortex function, and the ModBent, a measure that’s sensitive to dentate gyrus function. The flavanol intervention did not affect ModBent results or performance on the Flanker test after 1 year.
However, it was a different story for those with a poor diet at baseline. Researchers stratified participants into tertiles on the basis of diet quality as measured by the Healthy Eating Index (HEI) scores. Those in the lowest tertile had poorer baseline hippocampal-dependent memory performance but not memory related to the prefrontal cortex.
The flavanol intervention improved performance on the ModRey test, compared with placebo in participants in the low HEI tertile (overall effect: d = 0.086; P = .011) but not among those with a medium or high HEI at baseline.
“We confirmed that the flavanol intervention only benefits people who are relatively deficient at baseline,” said Dr. Small.
The correlation with hippocampal-dependent memory was confirmed in a subset of 1,361 study participants who provided a urine sample. Researchers measured urinary 5-(3′,4′-dihydroxyphenyl)-gamma-valerolactone metabolite (gVLM) concentrations, a validated biomarker of flavanol consumption.
After stratifying these results into tertiles, researchers found performance on the ModRey was significantly improved with the dietary flavanol intervention (overall effect: d = 0.141; P = .006) in the lowest gVLM tertile.
Memory restored
When participants in the lowest tertile consumed the supplement, “their flavanol levels went back to normal, and when that happened, their memory was restored,” said Dr. Small.
It appears that there is a sort of ceiling effect to the flavanol benefits. “It seems what you need to do is normalize your flavanol levels; if you go above normal, there was no evidence that your memory keeps on getting better,” said Dr. Small.
The study included only older adults, so it’s unclear what the impact of flavanol supplementation is in younger adults. But cognitive aging “begins its slippery side” in the 40s, said Dr. Small. “If this is truly a nutrient that is taken to prevent that slide from happening, it might be beneficial to start in our 40s.”
He recognized that the effect size is not large but said this is “very dependent” on baseline factors and most study participants had a rather healthy diet. “None of our participants were really highly deficient” in flavanols, he said.
“To see a stronger effect size, we need to do another study where we recruit people who are very low, truly deficient, in flavanols, and then see what happens.”
Showing that flavanols are linked to the hippocampal and not to the prefrontal component of cognitive aging “speaks to the mechanism,” said Dr. Small.
Though the exact mechanism linking flavanols with enhanced memory isn’t clear, there are some clues; for example, research suggests cognitive aging affects the dentate gyrus, a subregion of the hippocampus.
The flavanol supplements were well tolerated. “I can say with close to certainty that this is very safe,” said Dr. Small, adding the flavanols have now been used in numerous studies.
The findings suggest flavanol consumption might be part of future dietary guidelines. “I suspect that once there is sufficient evidence, flavanols will be part of the dietary recommendations for healthy aging,” said Dr. Small.
A word of caution
Heather M. Snyder, PhD, vice president of medical and scientific relations, Alzheimer’s Association, said that though science suggests a balanced diet is good for overall brain health, no single food, beverage, ingredient, vitamin, or supplement has yet been proven to prevent dementia, treat or cure Alzheimer’s, or benefit cognitive function or brain health.
Experts agree the best source of vitamins and other nutrients is from whole foods as part of a balanced diet. “We recognize that, for a variety of reasons, this may not always be possible,” said Dr. Snyder.
However, she noted, dietary supplements are not subject to the same rigorous review and regulation process as medications.
“The Alzheimer’s Association strongly encourages individuals to have conversations with their physicians about all medications and dietary supplements they are currently taking or interested in starting.”
COSMOS is supported by an investigator-initiated grant from Mars Edge, a segment of Mars, company engaged in flavanol research and flavanol-related commercial activities, which included infrastructure support and the donation of study pills and packaging. Small reports receiving an unrestricted research grant from Mars.
A version of this article first appeared on Medscape.com.
Potential new treatment for REM sleep behavior disorder
Dual orexin receptor antagonists (DORAs), a class of drugs approved to treat insomnia, may also be effective for rapid eye movement sleep behavior disorder (RBD), a study suggests.
About 3 million people in the United States have RBD, which is often a precursor to Parkinson’s disease. People with the disorder act out their dreams by talking, flailing their arms and legs, punching, kicking, and exhibiting other behaviors while asleep.
Researchers used an animal model for the study, which they say is the first to identify a new form of treatment for RBD.
“REM behavior disorder is difficult to treat, and the treatments are mostly limited to clonazepam and melatonin,” which may have side effects, senior investigator Andrew Varga, MD, PhD, associate professor of pulmonary, critical care, and sleep medicine at the Icahn School of Medicine at Mount Sinai, New York, told this news organization. “We’re using something completely different, which raises the possibility this might be something useful for REM behavior disorders.”
The findings, with Mount Sinai assistant professor Korey Kam, PhD, as lead author, were published online in the Journal of Neuroscience.
A new model for RBD?
RBD can signal risk for synucleinopathies, a group of neurological conditions such as Parkinson’s disease that involve the formation of clumps of alpha-synuclein protein in the brain.
Prior research on RBD was done in synucleinopathy mouse models. For this study, however, researchers used a tauopathy mouse model to investigate how the abnormal accumulation of tau protein might affect RBD.
Researchers collected data on biophysical properties when the mice were awake and in REM and non-REM sleep. They examined length of sleep, transitions from waking to sleep, and how some factors are related to age.
Nearly a third of the older animals showed behaviors similar to REM sleep behavior disorder in humans, including chewing and limb extension.
But after researchers administered a DORA medication twice during a 24-hour period, they noted that the medication not only helped the animals fall asleep faster and for longer, it also reduced levels of dream enactment that are a hallmark of RBD.
The ‘bigger highlight’
Finding RBD behaviors in a tauopathy animal model was surprising, Dr. Varga said, because RBD has been previously linked to synucleinopathies. There was no known correlation between RBD and abnormal accumulation of tau.
Another unexpected finding was the detection of RBD in some of the younger animals, who had not yet shown evidence of tau accumulation.
“It appears to be a biomarker or a signature of something that’s going on that predicts the impending tauopathy at a time where there is very little, or no, tau pathology going on in the brain,” Dr. Varga said.
If RBD is an early predictor of future tau accumulation, the model could guide future prevention and treatment. However, the more important finding is the potential new treatment for the condition.
“The bigger highlight here is less about what’s causing the RBD [than about] what you can do to make it better,” he said.
The next step in the work is to study whether the effect of DORAs on RBD seen in this tauopathy mouse model is evidenced in other animals and whether it is effective in humans with RBD, Dr. Varga said.
The study was funded by the Alzheimer’s Association and Merck Investigator Studies Program. Dr. Kam, Dr. Varga, and coauthors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Dual orexin receptor antagonists (DORAs), a class of drugs approved to treat insomnia, may also be effective for rapid eye movement sleep behavior disorder (RBD), a study suggests.
About 3 million people in the United States have RBD, which is often a precursor to Parkinson’s disease. People with the disorder act out their dreams by talking, flailing their arms and legs, punching, kicking, and exhibiting other behaviors while asleep.
Researchers used an animal model for the study, which they say is the first to identify a new form of treatment for RBD.
“REM behavior disorder is difficult to treat, and the treatments are mostly limited to clonazepam and melatonin,” which may have side effects, senior investigator Andrew Varga, MD, PhD, associate professor of pulmonary, critical care, and sleep medicine at the Icahn School of Medicine at Mount Sinai, New York, told this news organization. “We’re using something completely different, which raises the possibility this might be something useful for REM behavior disorders.”
The findings, with Mount Sinai assistant professor Korey Kam, PhD, as lead author, were published online in the Journal of Neuroscience.
A new model for RBD?
RBD can signal risk for synucleinopathies, a group of neurological conditions such as Parkinson’s disease that involve the formation of clumps of alpha-synuclein protein in the brain.
Prior research on RBD was done in synucleinopathy mouse models. For this study, however, researchers used a tauopathy mouse model to investigate how the abnormal accumulation of tau protein might affect RBD.
Researchers collected data on biophysical properties when the mice were awake and in REM and non-REM sleep. They examined length of sleep, transitions from waking to sleep, and how some factors are related to age.
Nearly a third of the older animals showed behaviors similar to REM sleep behavior disorder in humans, including chewing and limb extension.
But after researchers administered a DORA medication twice during a 24-hour period, they noted that the medication not only helped the animals fall asleep faster and for longer, it also reduced levels of dream enactment that are a hallmark of RBD.
The ‘bigger highlight’
Finding RBD behaviors in a tauopathy animal model was surprising, Dr. Varga said, because RBD has been previously linked to synucleinopathies. There was no known correlation between RBD and abnormal accumulation of tau.
Another unexpected finding was the detection of RBD in some of the younger animals, who had not yet shown evidence of tau accumulation.
“It appears to be a biomarker or a signature of something that’s going on that predicts the impending tauopathy at a time where there is very little, or no, tau pathology going on in the brain,” Dr. Varga said.
If RBD is an early predictor of future tau accumulation, the model could guide future prevention and treatment. However, the more important finding is the potential new treatment for the condition.
“The bigger highlight here is less about what’s causing the RBD [than about] what you can do to make it better,” he said.
The next step in the work is to study whether the effect of DORAs on RBD seen in this tauopathy mouse model is evidenced in other animals and whether it is effective in humans with RBD, Dr. Varga said.
The study was funded by the Alzheimer’s Association and Merck Investigator Studies Program. Dr. Kam, Dr. Varga, and coauthors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Dual orexin receptor antagonists (DORAs), a class of drugs approved to treat insomnia, may also be effective for rapid eye movement sleep behavior disorder (RBD), a study suggests.
About 3 million people in the United States have RBD, which is often a precursor to Parkinson’s disease. People with the disorder act out their dreams by talking, flailing their arms and legs, punching, kicking, and exhibiting other behaviors while asleep.
Researchers used an animal model for the study, which they say is the first to identify a new form of treatment for RBD.
“REM behavior disorder is difficult to treat, and the treatments are mostly limited to clonazepam and melatonin,” which may have side effects, senior investigator Andrew Varga, MD, PhD, associate professor of pulmonary, critical care, and sleep medicine at the Icahn School of Medicine at Mount Sinai, New York, told this news organization. “We’re using something completely different, which raises the possibility this might be something useful for REM behavior disorders.”
The findings, with Mount Sinai assistant professor Korey Kam, PhD, as lead author, were published online in the Journal of Neuroscience.
A new model for RBD?
RBD can signal risk for synucleinopathies, a group of neurological conditions such as Parkinson’s disease that involve the formation of clumps of alpha-synuclein protein in the brain.
Prior research on RBD was done in synucleinopathy mouse models. For this study, however, researchers used a tauopathy mouse model to investigate how the abnormal accumulation of tau protein might affect RBD.
Researchers collected data on biophysical properties when the mice were awake and in REM and non-REM sleep. They examined length of sleep, transitions from waking to sleep, and how some factors are related to age.
Nearly a third of the older animals showed behaviors similar to REM sleep behavior disorder in humans, including chewing and limb extension.
But after researchers administered a DORA medication twice during a 24-hour period, they noted that the medication not only helped the animals fall asleep faster and for longer, it also reduced levels of dream enactment that are a hallmark of RBD.
The ‘bigger highlight’
Finding RBD behaviors in a tauopathy animal model was surprising, Dr. Varga said, because RBD has been previously linked to synucleinopathies. There was no known correlation between RBD and abnormal accumulation of tau.
Another unexpected finding was the detection of RBD in some of the younger animals, who had not yet shown evidence of tau accumulation.
“It appears to be a biomarker or a signature of something that’s going on that predicts the impending tauopathy at a time where there is very little, or no, tau pathology going on in the brain,” Dr. Varga said.
If RBD is an early predictor of future tau accumulation, the model could guide future prevention and treatment. However, the more important finding is the potential new treatment for the condition.
“The bigger highlight here is less about what’s causing the RBD [than about] what you can do to make it better,” he said.
The next step in the work is to study whether the effect of DORAs on RBD seen in this tauopathy mouse model is evidenced in other animals and whether it is effective in humans with RBD, Dr. Varga said.
The study was funded by the Alzheimer’s Association and Merck Investigator Studies Program. Dr. Kam, Dr. Varga, and coauthors report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF NEUROSCIENCE