User login
Children With Chronic Skin Disorders Face Substantial Stigma
TOPLINE:
METHODOLOGY:
- Stigmatization has been addressed for several chronic medical conditions, such as HIV/AIDS, obesity, and mental illness; however, it has received limited attention in children living with chronic skin disorders.
- This cross-sectional, single-visit study examined the prevalence of stigma, its dependence on disease visibility and severity, and its association with mental health and QoL in children with chronic skin disorders.
- A total of 1671 children aged 8-17 years (57.9% girls; mean age, 13.7 years) were recruited from 32 pediatric dermatology centers in the United States and Canada from November 2018 to November 2021. The most common conditions were acne, atopic dermatitis/eczematous disorders, alopecia, and psoriasis, but rare genetic disorders were also represented.
- The primary outcome was the extent of stigmatization in relation to disease visibility, assessed using the Patient-Reported Outcomes Measurement Instrumentation System Pediatric Stigma-Skin.
- Secondary outcomes were the extent of stigmatization in relation to disease severity, along with QoL, depression, anxiety, and poor peer relationships.
TAKEAWAY:
- Approximately half (56.4%) of the children self-reported their skin condition as highly visible; 50.5% reported their disease severity as moderate, while 21.3% reported it as severe.
- Stigma was experienced by 73% of children and adolescents with chronic skin disease, with 43.8% reporting moderate stigma.
- Stigma scores correlated strongly with impaired QOL (Spearman’s rank correlation coefficient = 0.73) and child-reported scores for depression (Spearman’s rank correlation coefficient = 0.61) and moderately with anxiety (Spearman’s rank correlation coefficient = 0.54) and peer relationships (Spearman’s rank correlation coefficient = −0.49; all P < .001).
- Although stigma is increased for children with higher disease visibility and severity, the relatively weak correlation between child-assessed disease visibility and stigma (Spearman’s rank correlation coefficient = 0.22) showed that stigma is common in children even when diseases are not highly visible.
IN PRACTICE:
“Better treatment approaches for chronic skin diseases in children remain an unmet need. Increased awareness and instituting medical and psychological interventions to identify and reduce stigma and disease severity are important directions for improving QOL,” the authors concluded.
SOURCE:
Amy S. Paller, MD, professor of pediatrics and dermatology, Northwestern University, Chicago, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Stigmatization needs to be assessed in children from low- and middle-income countries. Investigators enrolled children who had physician-assessed moderate to severe disease severity and/or at least some visibility of skin disease while wearing clothing, which resulted in exclusion of children with mild chronic disease, and the pandemic limited enrollment.
DISCLOSURES:
This study was funded through a grant from the Pediatric Dermatology Research Alliance (PeDRA). The authors declared receiving grants, personal fees, and honorarium and having other ties with various sources.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Stigmatization has been addressed for several chronic medical conditions, such as HIV/AIDS, obesity, and mental illness; however, it has received limited attention in children living with chronic skin disorders.
- This cross-sectional, single-visit study examined the prevalence of stigma, its dependence on disease visibility and severity, and its association with mental health and QoL in children with chronic skin disorders.
- A total of 1671 children aged 8-17 years (57.9% girls; mean age, 13.7 years) were recruited from 32 pediatric dermatology centers in the United States and Canada from November 2018 to November 2021. The most common conditions were acne, atopic dermatitis/eczematous disorders, alopecia, and psoriasis, but rare genetic disorders were also represented.
- The primary outcome was the extent of stigmatization in relation to disease visibility, assessed using the Patient-Reported Outcomes Measurement Instrumentation System Pediatric Stigma-Skin.
- Secondary outcomes were the extent of stigmatization in relation to disease severity, along with QoL, depression, anxiety, and poor peer relationships.
TAKEAWAY:
- Approximately half (56.4%) of the children self-reported their skin condition as highly visible; 50.5% reported their disease severity as moderate, while 21.3% reported it as severe.
- Stigma was experienced by 73% of children and adolescents with chronic skin disease, with 43.8% reporting moderate stigma.
- Stigma scores correlated strongly with impaired QOL (Spearman’s rank correlation coefficient = 0.73) and child-reported scores for depression (Spearman’s rank correlation coefficient = 0.61) and moderately with anxiety (Spearman’s rank correlation coefficient = 0.54) and peer relationships (Spearman’s rank correlation coefficient = −0.49; all P < .001).
- Although stigma is increased for children with higher disease visibility and severity, the relatively weak correlation between child-assessed disease visibility and stigma (Spearman’s rank correlation coefficient = 0.22) showed that stigma is common in children even when diseases are not highly visible.
IN PRACTICE:
“Better treatment approaches for chronic skin diseases in children remain an unmet need. Increased awareness and instituting medical and psychological interventions to identify and reduce stigma and disease severity are important directions for improving QOL,” the authors concluded.
SOURCE:
Amy S. Paller, MD, professor of pediatrics and dermatology, Northwestern University, Chicago, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Stigmatization needs to be assessed in children from low- and middle-income countries. Investigators enrolled children who had physician-assessed moderate to severe disease severity and/or at least some visibility of skin disease while wearing clothing, which resulted in exclusion of children with mild chronic disease, and the pandemic limited enrollment.
DISCLOSURES:
This study was funded through a grant from the Pediatric Dermatology Research Alliance (PeDRA). The authors declared receiving grants, personal fees, and honorarium and having other ties with various sources.
A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- Stigmatization has been addressed for several chronic medical conditions, such as HIV/AIDS, obesity, and mental illness; however, it has received limited attention in children living with chronic skin disorders.
- This cross-sectional, single-visit study examined the prevalence of stigma, its dependence on disease visibility and severity, and its association with mental health and QoL in children with chronic skin disorders.
- A total of 1671 children aged 8-17 years (57.9% girls; mean age, 13.7 years) were recruited from 32 pediatric dermatology centers in the United States and Canada from November 2018 to November 2021. The most common conditions were acne, atopic dermatitis/eczematous disorders, alopecia, and psoriasis, but rare genetic disorders were also represented.
- The primary outcome was the extent of stigmatization in relation to disease visibility, assessed using the Patient-Reported Outcomes Measurement Instrumentation System Pediatric Stigma-Skin.
- Secondary outcomes were the extent of stigmatization in relation to disease severity, along with QoL, depression, anxiety, and poor peer relationships.
TAKEAWAY:
- Approximately half (56.4%) of the children self-reported their skin condition as highly visible; 50.5% reported their disease severity as moderate, while 21.3% reported it as severe.
- Stigma was experienced by 73% of children and adolescents with chronic skin disease, with 43.8% reporting moderate stigma.
- Stigma scores correlated strongly with impaired QOL (Spearman’s rank correlation coefficient = 0.73) and child-reported scores for depression (Spearman’s rank correlation coefficient = 0.61) and moderately with anxiety (Spearman’s rank correlation coefficient = 0.54) and peer relationships (Spearman’s rank correlation coefficient = −0.49; all P < .001).
- Although stigma is increased for children with higher disease visibility and severity, the relatively weak correlation between child-assessed disease visibility and stigma (Spearman’s rank correlation coefficient = 0.22) showed that stigma is common in children even when diseases are not highly visible.
IN PRACTICE:
“Better treatment approaches for chronic skin diseases in children remain an unmet need. Increased awareness and instituting medical and psychological interventions to identify and reduce stigma and disease severity are important directions for improving QOL,” the authors concluded.
SOURCE:
Amy S. Paller, MD, professor of pediatrics and dermatology, Northwestern University, Chicago, led the study, which was published online in JAMA Dermatology.
LIMITATIONS:
Stigmatization needs to be assessed in children from low- and middle-income countries. Investigators enrolled children who had physician-assessed moderate to severe disease severity and/or at least some visibility of skin disease while wearing clothing, which resulted in exclusion of children with mild chronic disease, and the pandemic limited enrollment.
DISCLOSURES:
This study was funded through a grant from the Pediatric Dermatology Research Alliance (PeDRA). The authors declared receiving grants, personal fees, and honorarium and having other ties with various sources.
A version of this article appeared on Medscape.com.
Microbiome Alterations Linked to Growth Hormone Deficiency
, said Chinese researchers.
The research, published recently in Pediatric Research, involved more than 80 children and showed that those with GHD had alterations in microbial populations that have been linked to longevity, as well as a microbial and metabolite signature that allowed accurate discrimination from ISS.
“These findings provide novel insights into potential early diagnosis and innovative treatment alternatives, such as fecal microbiota transplantation, for short stature with varying growth hormone levels,” the authors wrote.
Andrew Dauber, MD, MMSc, chief of endocrinology, Children’s National Hospital, Washington, who was not involved in the study, said that while this is “a really interesting area of research,” he expressed “hesitancy about getting too excited about this data yet.”
“One of the problems is how you define growth hormone deficiency,” as it is “not a black and white diagnosis,” and the etiology and child’s growth trajectory also need to be considered, Dr. Dauber told said.
He explained: “The problem is that, when you rely on the growth hormone stimulation test alone, there’s so many false positives and so much overlap between patients with true growth hormone deficiency and those without. And I think that this article fell prey to that.”
He added: “It would be really, really interesting and helpful to have a microbiome signature that allows you to distinguish between true growth hormone deficiency and patients with idiopathic short stature.”
“But you have to make sure that your groups are very well defined for this study to be really valid. And that’s one of my concerns here.”
Dr. Dauber continued: “Now, that being said, they did find some associations that correlated with growth hormone peak levels,” some which replicate previous findings, “so I do think that there are kernels of important findings here.”
‘Tease Out Influences’ to Isolate the Interaction
He pointed out that there are “many factors that influence the microbiome,” such as the use of antibiotics, diet, age, and geographic location. Therefore, a study that could truly tease out all these influences and isolate the interaction with growth hormone levels would need to be “very thoughtfully designed.”
A number of factors contribute to short stature, lead author Lan Li, MD, Department of Radiology, The Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University, Wenzhou, China, and colleagues.
These include genetic factors, environmental factors, and conditions such as being small for gestational age at birth, familial short stature, and chronic systemic diseases, as well as GHD and ISS.
Recent animal studies have suggested that there may be a bidirectional relationship between the gut microbiota and the growth hormone/insulin-like growth factor 1 axis, and it has been shown that individuals with GHD have significant alterations in their gut microbiota compared with healthy controls.
To investigate, they studied 36 children diagnosed with GHD, 32 with ISS, and 16 age- and sex-matched healthy controls, all of whom were recruited between February 2019 and June 2021 from the Pediatric Endocrinology Department of The Second Affiliated Hospital of Wenzhou Medical University.
Fecal samples obtained from the children underwent microbiome analysis using 16S ribosomal RNA gene sequencing, alongside nuclear MRI analysis of the metabolome, or the entire complement of small molecules in the samples.
Patients with GHD had a significantly higher body mass index than those with ISS (P < .05), and their peak growth hormone level was significantly lower (P < .001). Patients with GHD also had significantly higher total cholesterol and low-density lipoprotein cholesterol levels than patients with ISS (P < .05).
The team reports that the alpha diversity of the fecal microbiome, which measures the microbial diversity within a fecal sample, was similar between the three groups.
However, there was significant variation between the groups in the beta diversity, which quantifies the similarity or dissimilarity between two samples, and allows the overall taxonomic or functional diversity pattern to be linked to environmental features.
Compared with the healthy control group, the abundance of Pelomonas, Rodentibacter, and Rothia was significantly decreased in GHD and patients with ISS, while the abundance of Prevotellaceae_NK3B31_group was increased in the two patient groups, particularly in those with GHD.
In addition, the researchers found a decreased Firmicutes/Bacteroidota (F/B) ratio in participants with short stature, particularly in the GHD group. They noted that “emerging evidence suggests the F/B ratio may play a role in longevity.”
Nocardioides was substantially more common in the ISS group vs both patients with GHD and healthy controls, while Fusobacterium mortiferum was characteristic of GHD. The team suggests this “may serve as a critical intestinal factor contributing to the short stature observed in GHD.”
The metabolome analysis revealed that glucose, pyruvate, and pyrimidine metabolism may also play a significant role in distinguishing between patients with GHD and ISS and healthy control groups.
Finally, the team demonstrated that a panel combining 13 microbiome and metabolome markers was able to discriminate between GHD and ISS at an area under the receiver operating characteristic curve of 0.945, with a sensitivity of 87% and a specificity of 91%.
The study was supported by grants from the National Natural Science Foundation of China and Wenzhou Science and Technology Bureau in China. No relevant financial relationships were declared.
A version of this article appeared on Medscape.com.
, said Chinese researchers.
The research, published recently in Pediatric Research, involved more than 80 children and showed that those with GHD had alterations in microbial populations that have been linked to longevity, as well as a microbial and metabolite signature that allowed accurate discrimination from ISS.
“These findings provide novel insights into potential early diagnosis and innovative treatment alternatives, such as fecal microbiota transplantation, for short stature with varying growth hormone levels,” the authors wrote.
Andrew Dauber, MD, MMSc, chief of endocrinology, Children’s National Hospital, Washington, who was not involved in the study, said that while this is “a really interesting area of research,” he expressed “hesitancy about getting too excited about this data yet.”
“One of the problems is how you define growth hormone deficiency,” as it is “not a black and white diagnosis,” and the etiology and child’s growth trajectory also need to be considered, Dr. Dauber told said.
He explained: “The problem is that, when you rely on the growth hormone stimulation test alone, there’s so many false positives and so much overlap between patients with true growth hormone deficiency and those without. And I think that this article fell prey to that.”
He added: “It would be really, really interesting and helpful to have a microbiome signature that allows you to distinguish between true growth hormone deficiency and patients with idiopathic short stature.”
“But you have to make sure that your groups are very well defined for this study to be really valid. And that’s one of my concerns here.”
Dr. Dauber continued: “Now, that being said, they did find some associations that correlated with growth hormone peak levels,” some which replicate previous findings, “so I do think that there are kernels of important findings here.”
‘Tease Out Influences’ to Isolate the Interaction
He pointed out that there are “many factors that influence the microbiome,” such as the use of antibiotics, diet, age, and geographic location. Therefore, a study that could truly tease out all these influences and isolate the interaction with growth hormone levels would need to be “very thoughtfully designed.”
A number of factors contribute to short stature, lead author Lan Li, MD, Department of Radiology, The Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University, Wenzhou, China, and colleagues.
These include genetic factors, environmental factors, and conditions such as being small for gestational age at birth, familial short stature, and chronic systemic diseases, as well as GHD and ISS.
Recent animal studies have suggested that there may be a bidirectional relationship between the gut microbiota and the growth hormone/insulin-like growth factor 1 axis, and it has been shown that individuals with GHD have significant alterations in their gut microbiota compared with healthy controls.
To investigate, they studied 36 children diagnosed with GHD, 32 with ISS, and 16 age- and sex-matched healthy controls, all of whom were recruited between February 2019 and June 2021 from the Pediatric Endocrinology Department of The Second Affiliated Hospital of Wenzhou Medical University.
Fecal samples obtained from the children underwent microbiome analysis using 16S ribosomal RNA gene sequencing, alongside nuclear MRI analysis of the metabolome, or the entire complement of small molecules in the samples.
Patients with GHD had a significantly higher body mass index than those with ISS (P < .05), and their peak growth hormone level was significantly lower (P < .001). Patients with GHD also had significantly higher total cholesterol and low-density lipoprotein cholesterol levels than patients with ISS (P < .05).
The team reports that the alpha diversity of the fecal microbiome, which measures the microbial diversity within a fecal sample, was similar between the three groups.
However, there was significant variation between the groups in the beta diversity, which quantifies the similarity or dissimilarity between two samples, and allows the overall taxonomic or functional diversity pattern to be linked to environmental features.
Compared with the healthy control group, the abundance of Pelomonas, Rodentibacter, and Rothia was significantly decreased in GHD and patients with ISS, while the abundance of Prevotellaceae_NK3B31_group was increased in the two patient groups, particularly in those with GHD.
In addition, the researchers found a decreased Firmicutes/Bacteroidota (F/B) ratio in participants with short stature, particularly in the GHD group. They noted that “emerging evidence suggests the F/B ratio may play a role in longevity.”
Nocardioides was substantially more common in the ISS group vs both patients with GHD and healthy controls, while Fusobacterium mortiferum was characteristic of GHD. The team suggests this “may serve as a critical intestinal factor contributing to the short stature observed in GHD.”
The metabolome analysis revealed that glucose, pyruvate, and pyrimidine metabolism may also play a significant role in distinguishing between patients with GHD and ISS and healthy control groups.
Finally, the team demonstrated that a panel combining 13 microbiome and metabolome markers was able to discriminate between GHD and ISS at an area under the receiver operating characteristic curve of 0.945, with a sensitivity of 87% and a specificity of 91%.
The study was supported by grants from the National Natural Science Foundation of China and Wenzhou Science and Technology Bureau in China. No relevant financial relationships were declared.
A version of this article appeared on Medscape.com.
, said Chinese researchers.
The research, published recently in Pediatric Research, involved more than 80 children and showed that those with GHD had alterations in microbial populations that have been linked to longevity, as well as a microbial and metabolite signature that allowed accurate discrimination from ISS.
“These findings provide novel insights into potential early diagnosis and innovative treatment alternatives, such as fecal microbiota transplantation, for short stature with varying growth hormone levels,” the authors wrote.
Andrew Dauber, MD, MMSc, chief of endocrinology, Children’s National Hospital, Washington, who was not involved in the study, said that while this is “a really interesting area of research,” he expressed “hesitancy about getting too excited about this data yet.”
“One of the problems is how you define growth hormone deficiency,” as it is “not a black and white diagnosis,” and the etiology and child’s growth trajectory also need to be considered, Dr. Dauber told said.
He explained: “The problem is that, when you rely on the growth hormone stimulation test alone, there’s so many false positives and so much overlap between patients with true growth hormone deficiency and those without. And I think that this article fell prey to that.”
He added: “It would be really, really interesting and helpful to have a microbiome signature that allows you to distinguish between true growth hormone deficiency and patients with idiopathic short stature.”
“But you have to make sure that your groups are very well defined for this study to be really valid. And that’s one of my concerns here.”
Dr. Dauber continued: “Now, that being said, they did find some associations that correlated with growth hormone peak levels,” some which replicate previous findings, “so I do think that there are kernels of important findings here.”
‘Tease Out Influences’ to Isolate the Interaction
He pointed out that there are “many factors that influence the microbiome,” such as the use of antibiotics, diet, age, and geographic location. Therefore, a study that could truly tease out all these influences and isolate the interaction with growth hormone levels would need to be “very thoughtfully designed.”
A number of factors contribute to short stature, lead author Lan Li, MD, Department of Radiology, The Second Affiliated Hospital and Yuying Children’s Hospital of Wenzhou Medical University, Wenzhou, China, and colleagues.
These include genetic factors, environmental factors, and conditions such as being small for gestational age at birth, familial short stature, and chronic systemic diseases, as well as GHD and ISS.
Recent animal studies have suggested that there may be a bidirectional relationship between the gut microbiota and the growth hormone/insulin-like growth factor 1 axis, and it has been shown that individuals with GHD have significant alterations in their gut microbiota compared with healthy controls.
To investigate, they studied 36 children diagnosed with GHD, 32 with ISS, and 16 age- and sex-matched healthy controls, all of whom were recruited between February 2019 and June 2021 from the Pediatric Endocrinology Department of The Second Affiliated Hospital of Wenzhou Medical University.
Fecal samples obtained from the children underwent microbiome analysis using 16S ribosomal RNA gene sequencing, alongside nuclear MRI analysis of the metabolome, or the entire complement of small molecules in the samples.
Patients with GHD had a significantly higher body mass index than those with ISS (P < .05), and their peak growth hormone level was significantly lower (P < .001). Patients with GHD also had significantly higher total cholesterol and low-density lipoprotein cholesterol levels than patients with ISS (P < .05).
The team reports that the alpha diversity of the fecal microbiome, which measures the microbial diversity within a fecal sample, was similar between the three groups.
However, there was significant variation between the groups in the beta diversity, which quantifies the similarity or dissimilarity between two samples, and allows the overall taxonomic or functional diversity pattern to be linked to environmental features.
Compared with the healthy control group, the abundance of Pelomonas, Rodentibacter, and Rothia was significantly decreased in GHD and patients with ISS, while the abundance of Prevotellaceae_NK3B31_group was increased in the two patient groups, particularly in those with GHD.
In addition, the researchers found a decreased Firmicutes/Bacteroidota (F/B) ratio in participants with short stature, particularly in the GHD group. They noted that “emerging evidence suggests the F/B ratio may play a role in longevity.”
Nocardioides was substantially more common in the ISS group vs both patients with GHD and healthy controls, while Fusobacterium mortiferum was characteristic of GHD. The team suggests this “may serve as a critical intestinal factor contributing to the short stature observed in GHD.”
The metabolome analysis revealed that glucose, pyruvate, and pyrimidine metabolism may also play a significant role in distinguishing between patients with GHD and ISS and healthy control groups.
Finally, the team demonstrated that a panel combining 13 microbiome and metabolome markers was able to discriminate between GHD and ISS at an area under the receiver operating characteristic curve of 0.945, with a sensitivity of 87% and a specificity of 91%.
The study was supported by grants from the National Natural Science Foundation of China and Wenzhou Science and Technology Bureau in China. No relevant financial relationships were declared.
A version of this article appeared on Medscape.com.
FROM PEDIATRIC RESEARCH
Syphilis Treatment Falls Short for Pregnant Patients
Approximately one third of pregnant individuals with syphilis were inadequately treated or not treated for syphilis despite receiving timely prenatal care, based on data from nearly 1500 patients.
Although congenital syphilis is preventable with treatment before or early in pregnancy, data from the Centers for Disease Control and Prevention (CDC) show a doubling of syphilis rates in the United States between 2018 and 2021 wrote Ayzsa Tannis, MPH, of the Centers for Disease Control and Prevention, Atlanta, and colleagues.
To better understand factors contributing to inadequate syphilis treatment during pregnancy, the researchers examined data from 1476 individuals with syphilis during pregnancy. The study population came from six jurisdictions that participated in the Surveillance for Emerging Threats to Pregnant People and Infants Network, and sources included case investigations, medical records, and links between laboratory data and vital records.
The researchers characterized the status of syphilis during pregnancy as adequate, inadequate, or not treated based on the CDC’s Sexually Transmitted Infections Treatment Guidelines, 2021. Prenatal care was defined as timely (at least 30 days prior to pregnancy outcome), nontimely (less than 30 days before pregnancy outcome), and no prenatal care. The findings were published in Obstetrics & Gynecology.
Of the 1476 individuals studied, 855 (57.9%) were adequately treated for syphilis and 621 (42.1%) were inadequately or not treated.
Overall, 82% of the study population received timely prenatal care. However, 32.1% of those who received timely prenatal care were inadequately treated, including 14.8% who received no syphilis treatment. Individuals with nontimely or no prenatal care were significantly more likely to receive inadequate or no treatment for syphilis than those who received timely care (risk ratio, 2.50 and 2.73, respectively).
The findings were consistent with previous studies of missed opportunities for prevention and treatment, the researchers noted. Factors behind nontimely treatment (less than 30 days before pregnancy outcome) may include intermittent shortages of benzathine penicillin G, the standard treatment for syphilis, as well as the lack of time and administrative support for clinicians to communicate with patients and health departments, and to expedite treatment, the researchers wrote.
The results were limited by several factors including the use of data from six US jurisdictions that may not generalize to other areas, the variations in reporting years for the different jurisdictions, and variation in mandates for syphilis screening during pregnancy, the researchers noted.
More research is needed to improve syphilis testing itself, and to develop more treatment options, the researchers concluded. Partnerships among public health, patient advocacy groups, prenatal care clinicians, and other clinicians outside the prenatal care setting also are needed for effective intervention in pregnant individuals with syphilis, they said.
The study was carried out as part of the regular work of the CDC, supported by the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases Cooperative Agreement and through contractual mechanisms including the Local Health Department Initiative to Chickasaw Health Consulting. The researchers had no financial conflicts to disclose.
Approximately one third of pregnant individuals with syphilis were inadequately treated or not treated for syphilis despite receiving timely prenatal care, based on data from nearly 1500 patients.
Although congenital syphilis is preventable with treatment before or early in pregnancy, data from the Centers for Disease Control and Prevention (CDC) show a doubling of syphilis rates in the United States between 2018 and 2021 wrote Ayzsa Tannis, MPH, of the Centers for Disease Control and Prevention, Atlanta, and colleagues.
To better understand factors contributing to inadequate syphilis treatment during pregnancy, the researchers examined data from 1476 individuals with syphilis during pregnancy. The study population came from six jurisdictions that participated in the Surveillance for Emerging Threats to Pregnant People and Infants Network, and sources included case investigations, medical records, and links between laboratory data and vital records.
The researchers characterized the status of syphilis during pregnancy as adequate, inadequate, or not treated based on the CDC’s Sexually Transmitted Infections Treatment Guidelines, 2021. Prenatal care was defined as timely (at least 30 days prior to pregnancy outcome), nontimely (less than 30 days before pregnancy outcome), and no prenatal care. The findings were published in Obstetrics & Gynecology.
Of the 1476 individuals studied, 855 (57.9%) were adequately treated for syphilis and 621 (42.1%) were inadequately or not treated.
Overall, 82% of the study population received timely prenatal care. However, 32.1% of those who received timely prenatal care were inadequately treated, including 14.8% who received no syphilis treatment. Individuals with nontimely or no prenatal care were significantly more likely to receive inadequate or no treatment for syphilis than those who received timely care (risk ratio, 2.50 and 2.73, respectively).
The findings were consistent with previous studies of missed opportunities for prevention and treatment, the researchers noted. Factors behind nontimely treatment (less than 30 days before pregnancy outcome) may include intermittent shortages of benzathine penicillin G, the standard treatment for syphilis, as well as the lack of time and administrative support for clinicians to communicate with patients and health departments, and to expedite treatment, the researchers wrote.
The results were limited by several factors including the use of data from six US jurisdictions that may not generalize to other areas, the variations in reporting years for the different jurisdictions, and variation in mandates for syphilis screening during pregnancy, the researchers noted.
More research is needed to improve syphilis testing itself, and to develop more treatment options, the researchers concluded. Partnerships among public health, patient advocacy groups, prenatal care clinicians, and other clinicians outside the prenatal care setting also are needed for effective intervention in pregnant individuals with syphilis, they said.
The study was carried out as part of the regular work of the CDC, supported by the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases Cooperative Agreement and through contractual mechanisms including the Local Health Department Initiative to Chickasaw Health Consulting. The researchers had no financial conflicts to disclose.
Approximately one third of pregnant individuals with syphilis were inadequately treated or not treated for syphilis despite receiving timely prenatal care, based on data from nearly 1500 patients.
Although congenital syphilis is preventable with treatment before or early in pregnancy, data from the Centers for Disease Control and Prevention (CDC) show a doubling of syphilis rates in the United States between 2018 and 2021 wrote Ayzsa Tannis, MPH, of the Centers for Disease Control and Prevention, Atlanta, and colleagues.
To better understand factors contributing to inadequate syphilis treatment during pregnancy, the researchers examined data from 1476 individuals with syphilis during pregnancy. The study population came from six jurisdictions that participated in the Surveillance for Emerging Threats to Pregnant People and Infants Network, and sources included case investigations, medical records, and links between laboratory data and vital records.
The researchers characterized the status of syphilis during pregnancy as adequate, inadequate, or not treated based on the CDC’s Sexually Transmitted Infections Treatment Guidelines, 2021. Prenatal care was defined as timely (at least 30 days prior to pregnancy outcome), nontimely (less than 30 days before pregnancy outcome), and no prenatal care. The findings were published in Obstetrics & Gynecology.
Of the 1476 individuals studied, 855 (57.9%) were adequately treated for syphilis and 621 (42.1%) were inadequately or not treated.
Overall, 82% of the study population received timely prenatal care. However, 32.1% of those who received timely prenatal care were inadequately treated, including 14.8% who received no syphilis treatment. Individuals with nontimely or no prenatal care were significantly more likely to receive inadequate or no treatment for syphilis than those who received timely care (risk ratio, 2.50 and 2.73, respectively).
The findings were consistent with previous studies of missed opportunities for prevention and treatment, the researchers noted. Factors behind nontimely treatment (less than 30 days before pregnancy outcome) may include intermittent shortages of benzathine penicillin G, the standard treatment for syphilis, as well as the lack of time and administrative support for clinicians to communicate with patients and health departments, and to expedite treatment, the researchers wrote.
The results were limited by several factors including the use of data from six US jurisdictions that may not generalize to other areas, the variations in reporting years for the different jurisdictions, and variation in mandates for syphilis screening during pregnancy, the researchers noted.
More research is needed to improve syphilis testing itself, and to develop more treatment options, the researchers concluded. Partnerships among public health, patient advocacy groups, prenatal care clinicians, and other clinicians outside the prenatal care setting also are needed for effective intervention in pregnant individuals with syphilis, they said.
The study was carried out as part of the regular work of the CDC, supported by the Epidemiology and Laboratory Capacity for Prevention and Control of Emerging Infectious Diseases Cooperative Agreement and through contractual mechanisms including the Local Health Department Initiative to Chickasaw Health Consulting. The researchers had no financial conflicts to disclose.
FROM OBSTETRICS & GYNECOLOGY
Occipital Scalp Nodule in a Newborn
The Diagnosis: Subcutaneous Fat Necrosis
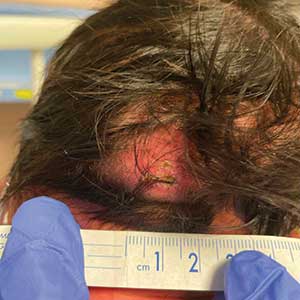
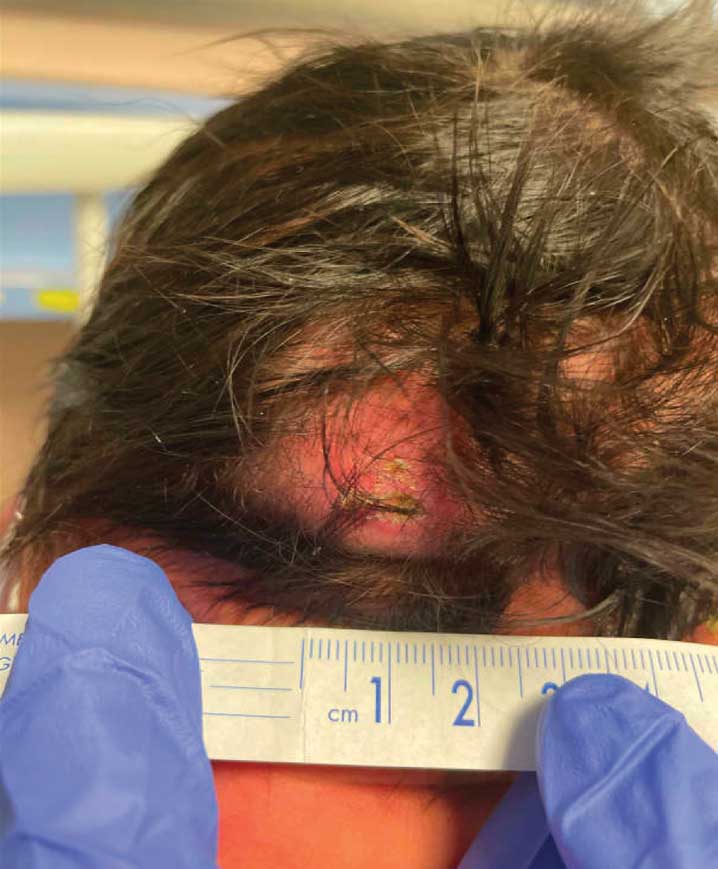
Histopathology revealed lobular panniculitis with lymphohistiocytic inflammation, lipid crystals, and calcifications in our patient (Figure). Subcutaneous fat necrosis (SCFN) was diagnosed based on these characteristic histopathologic findings. No further treatment was pursued.
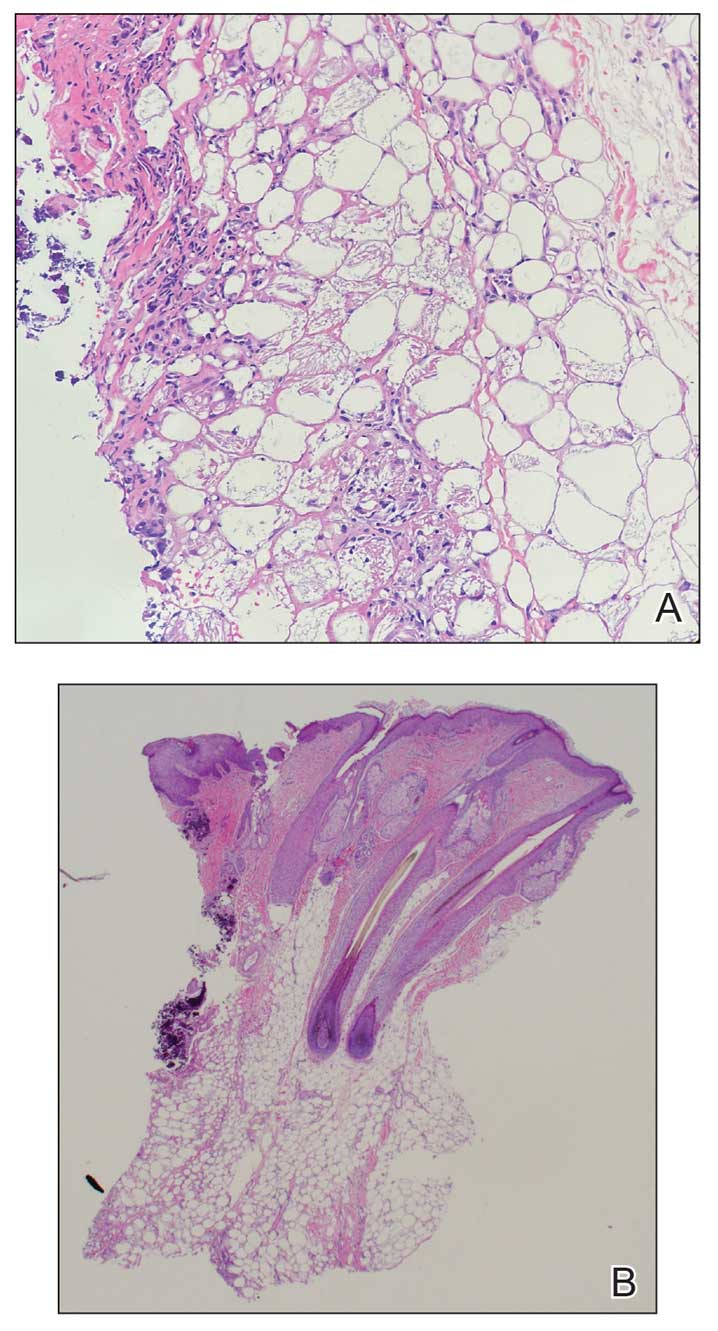
Subcutaneous fat necrosis is a rare, self-limiting panniculitis that typically resolves within several weeks to months without scarring. It manifests as red or violaceous subcutaneous nodules or plaques most commonly on the buttocks, trunk, proximal arms and legs, and cheeks.1 Histopathology reveals lobular panniculitis with dense granulomatous infiltrates of histiocytes, eosinophils, and multinucleated giant cells with needle-shaped crystals. Focal areas of fat necrosis with calcification also can be seen.2
The epidemiology of SCFN is unknown. Most cases occur in healthy full-term to postterm neonates who experience hypoxia, other prenatal stressors, or therapeutic hypothermia for the treatment of hypoxic-ischemic encephalopathy.3 Although the etiology is unclear, certain inciting factors such as local tissue hypoxia, cold exposure, meconium aspiration, maternal diabetes, preeclampsia, and mechanical pressure have been proposed. Our patient underwent hypothermic cooling protocol, and it has been suggested that the increased saturated to unsaturated fat concentration in the skin of newborns increases the melting point, thus predisposing them to fat crystalization.4 Cases of SCFN involving the scalp are rare; therefore, any newborns receiving hypothermic therapy for hypoxic-ischemic encephalopathy should have a thorough skin examination with possible biopsy of lesions that are characteristic of SCFN, such as red or violaceous subcutaneous nodules or plaques, for specific disease identification.
The main complication of SCFN is hypercalcemia, which occurs in approximately 50% of cases. Other serum abnormalities include hyperglycemia, hypertriglyceridemia, and thrombocytopenia, though these findings are not as well associated.4 Patients with associated hypercalcemia may be asymptomatic, as in our patient, but other presentations include irritability, weakness, anorexia, vomiting, renal failure, failure to thrive, and encephalopathy. Nephrocalcinosis is a common complication of severe hypercalcemia; however, there is little evidence of associated major renal dysfunction.5 The exact mechanism of hypercalcemia is poorly understood. A widely accepted theory postulates that a granulomatous inflammatory infiltrate upregulates 1-α-hydroxylase activity, which enzymatically converts 25-hydroxyvitamin D to its active form, 1,25-dihydroxycholecalciferol, which increases bone resorption and calcium absorption through the gastrointestinal tract and renal systems. Treatments for hypercalcemia include hyperhydration, calcium-wasting diuretics, and low calcium intake.6 Furthermore, calcium levels should be obtained at the time of diagnosis and 30, 45, and 60 days after the lesions resolve.4
Subcutaneous fat necrosis needs to be differentiated from the more severe panniculitis, sclerema neonatorum (SN), which typically affects critically ill, preterm, and small-for-gestational-age newborns. It is associated with a high mortality rate and is characterized by skin and subadjacent tissue structures. The process typically begins in the thighs, buttocks, or trunk and spreads diffusely, sparing the fat-free palms, soles, and genitalia.7 Although our patient was born preterm, the physical characteristics of the nodule and the lack of severe illness placed SN lower on our differential. Histopathologic differences between SCFN and SN involve the extent of tissue fibrosis and presence of inflammatory cells. Sclerema neonatorum typically manifests with thickened connective tissue with a sparse inflammatory infiltrate, including lymphocytes, histiocytes, and multinucleated giant cells.7 Conversely, SCFN manifests with fat necrosis with an extensive inflammatory infiltrate. It is important to be able to distinguish between these 2 conditions, as both have vastly different prognoses.
Cold panniculitis, sometimes called “popsicle panniculitis,” is a phenomenon in which cold contact with the skin causes eruption of firm, erythematous, indurated plaques at the site of exposure. This self-limiting condition typically appears hours to days after cold exposure and spontaneously resolves in a few weeks.8 Therapeutic hypothermic protocol treatment involves using cooling devices to lower the body temperature for a short duration. The temperature typically is lowered to approximately 32 °C to 36 °C. These temperatures are not low enough to induce cold panniculitis, which is more commonly seen in facial ice applications when managing supraventricular tachycardia in neonates.
Cephalohematoma is a birthing injury that causes blood accumulation within the subperiosteal space. During parturition, the compressive and sheering forces on the calvarium rupture the vessels passing through the periosteum, causing blood to pool slowly into the subperiostium; thus, a cephalohematoma usually manifests later at 1 to 3 days of life as localized head swelling.9 The bleeding typically does not cross suture lines and is primarily found in the occipital or parietal regions. The incidence has been reported to be 0.4% to 2.5% of all live births.10 Although the location of the nodule in our patient was in the occipital region, imaging and biopsy results did not show hemorrhagic findings consistent with cephalohematoma. Management of cephalohematoma mainly is observational, as the mass slowly regresses and the accumulated blood gradually is reabsorbed.
Fungal scalp infections (tinea capitis) are common in the pediatric population. The peak incidence of this infection has been reported in children aged 3 to 7 years, with Trichophyton tonsurans and Microsporum canis as the usual causative organisms.11 Clinical features of tinea capitis include scaly patches with hair loss, hair loss with black pigmented dots at the follicular openings, diffuse scalp scaling with subtle hair loss, and cervical lymphadenopathy.12 Although less common, tinea capitis can progress to a more severe form known as a kerion, which is characterized by a tender plaque with pustules and crusting. A kerion can result in permanent scarring and alopecia if left untreated.12 In our patient, a nodule with scaling and faint erythema was observed, but no black pigmented dots at the follicular orifices were present. Therefore, a potassium hydroxide wet mount preparation used to diagnose tinea capitis was unnecessary. Systemic oral antifungal therapy such as fluconazole or terbinafine is the standard treatment for tinea capitis.
- Coondoo A, Lahiry R, Choudhury A, et al. Tender skin nodules in a newborn. Indian J Dermatol. 2013;58:328. doi:10.4103/0019-5154.113983
- Mitra S, Dove J, Somisetty SK. Subcutaneous fat necrosis in newbornan unusual case and review of literature. Eur J Pediatr. 2011;170:1107- 1110. doi:10.1007/s00431-011-1405-x
- Velasquez JH, Mendez MD. Newborn subcutaneous fat necrosis. In: StatPearls. StatPearls Publishing; 2022.
- Stefanko NS, Drolet BA. Subcutaneous fat necrosis of the newborn and associated hypercalcemia: a systematic review of the literature. Pediatr Dermatol. 2019;36:24-30. doi:10.1111/pde.13640
- Shumer DE, Thaker V, Taylor GA, et al. Severe hypercalcaemia due to subcutaneous fat necrosis: presentation, management and complications. Arch Dis Child Fetal Neonatal Ed. 2014;99:F419-F421. doi:10.1136/ archdischild-2014-306069
- Farooque A, Moss C, Zehnder D, et al. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in subcutaneous fat necrosis. Br J Dermatol. 2009;160:423-425. doi:10.1111/j.1365-2133.2008.08844.x
- Zeb A, Darmstadt GL. Sclerema neonatorum: a review of nomenclature, clinical presentation, histological features, differential diagnoses and management. J Perinatol. 2008;28:453-460. doi:10.1038/jp.2008.33
- Quesada-Cortés A, Campos-Muñoz L, Díaz-Díaz RM, et al. Cold panniculitis. Dermatol Clin. 2008;26:485-489, vii. doi:10.1016 /j.det.2008.05.015
- Raines DA, Krawiec C, Jain S. Cephalohematoma. In: StatPearls. StatPearls Publishing; 2023.
- Chung HY, Chung JY, Lee DG, et al. Surgical treatment of ossified cephalhematoma. J Craniofac Surg. 2004;15:774-779. doi:10.1097/00001665- 200409000-00015
- Leung AKC, Hon KL, Leong KF, et al. Tinea capitis: an updated review. Recent Pat Inflamm Allergy Drug Discov. 2020;14:58-68. doi:10.2174/1872 213x14666200106145624
- Kovitwanichkanont T, Chong A. Superficial fungal infections. Aust J Gen Pract. 2019;48:706-711. doi:10.31128/ajgp-05-19-4930
The Diagnosis: Subcutaneous Fat Necrosis
Histopathology revealed lobular panniculitis with lymphohistiocytic inflammation, lipid crystals, and calcifications in our patient (Figure). Subcutaneous fat necrosis (SCFN) was diagnosed based on these characteristic histopathologic findings. No further treatment was pursued.

Subcutaneous fat necrosis is a rare, self-limiting panniculitis that typically resolves within several weeks to months without scarring. It manifests as red or violaceous subcutaneous nodules or plaques most commonly on the buttocks, trunk, proximal arms and legs, and cheeks.1 Histopathology reveals lobular panniculitis with dense granulomatous infiltrates of histiocytes, eosinophils, and multinucleated giant cells with needle-shaped crystals. Focal areas of fat necrosis with calcification also can be seen.2
The epidemiology of SCFN is unknown. Most cases occur in healthy full-term to postterm neonates who experience hypoxia, other prenatal stressors, or therapeutic hypothermia for the treatment of hypoxic-ischemic encephalopathy.3 Although the etiology is unclear, certain inciting factors such as local tissue hypoxia, cold exposure, meconium aspiration, maternal diabetes, preeclampsia, and mechanical pressure have been proposed. Our patient underwent hypothermic cooling protocol, and it has been suggested that the increased saturated to unsaturated fat concentration in the skin of newborns increases the melting point, thus predisposing them to fat crystalization.4 Cases of SCFN involving the scalp are rare; therefore, any newborns receiving hypothermic therapy for hypoxic-ischemic encephalopathy should have a thorough skin examination with possible biopsy of lesions that are characteristic of SCFN, such as red or violaceous subcutaneous nodules or plaques, for specific disease identification.
The main complication of SCFN is hypercalcemia, which occurs in approximately 50% of cases. Other serum abnormalities include hyperglycemia, hypertriglyceridemia, and thrombocytopenia, though these findings are not as well associated.4 Patients with associated hypercalcemia may be asymptomatic, as in our patient, but other presentations include irritability, weakness, anorexia, vomiting, renal failure, failure to thrive, and encephalopathy. Nephrocalcinosis is a common complication of severe hypercalcemia; however, there is little evidence of associated major renal dysfunction.5 The exact mechanism of hypercalcemia is poorly understood. A widely accepted theory postulates that a granulomatous inflammatory infiltrate upregulates 1-α-hydroxylase activity, which enzymatically converts 25-hydroxyvitamin D to its active form, 1,25-dihydroxycholecalciferol, which increases bone resorption and calcium absorption through the gastrointestinal tract and renal systems. Treatments for hypercalcemia include hyperhydration, calcium-wasting diuretics, and low calcium intake.6 Furthermore, calcium levels should be obtained at the time of diagnosis and 30, 45, and 60 days after the lesions resolve.4
Subcutaneous fat necrosis needs to be differentiated from the more severe panniculitis, sclerema neonatorum (SN), which typically affects critically ill, preterm, and small-for-gestational-age newborns. It is associated with a high mortality rate and is characterized by skin and subadjacent tissue structures. The process typically begins in the thighs, buttocks, or trunk and spreads diffusely, sparing the fat-free palms, soles, and genitalia.7 Although our patient was born preterm, the physical characteristics of the nodule and the lack of severe illness placed SN lower on our differential. Histopathologic differences between SCFN and SN involve the extent of tissue fibrosis and presence of inflammatory cells. Sclerema neonatorum typically manifests with thickened connective tissue with a sparse inflammatory infiltrate, including lymphocytes, histiocytes, and multinucleated giant cells.7 Conversely, SCFN manifests with fat necrosis with an extensive inflammatory infiltrate. It is important to be able to distinguish between these 2 conditions, as both have vastly different prognoses.
Cold panniculitis, sometimes called “popsicle panniculitis,” is a phenomenon in which cold contact with the skin causes eruption of firm, erythematous, indurated plaques at the site of exposure. This self-limiting condition typically appears hours to days after cold exposure and spontaneously resolves in a few weeks.8 Therapeutic hypothermic protocol treatment involves using cooling devices to lower the body temperature for a short duration. The temperature typically is lowered to approximately 32 °C to 36 °C. These temperatures are not low enough to induce cold panniculitis, which is more commonly seen in facial ice applications when managing supraventricular tachycardia in neonates.
Cephalohematoma is a birthing injury that causes blood accumulation within the subperiosteal space. During parturition, the compressive and sheering forces on the calvarium rupture the vessels passing through the periosteum, causing blood to pool slowly into the subperiostium; thus, a cephalohematoma usually manifests later at 1 to 3 days of life as localized head swelling.9 The bleeding typically does not cross suture lines and is primarily found in the occipital or parietal regions. The incidence has been reported to be 0.4% to 2.5% of all live births.10 Although the location of the nodule in our patient was in the occipital region, imaging and biopsy results did not show hemorrhagic findings consistent with cephalohematoma. Management of cephalohematoma mainly is observational, as the mass slowly regresses and the accumulated blood gradually is reabsorbed.
Fungal scalp infections (tinea capitis) are common in the pediatric population. The peak incidence of this infection has been reported in children aged 3 to 7 years, with Trichophyton tonsurans and Microsporum canis as the usual causative organisms.11 Clinical features of tinea capitis include scaly patches with hair loss, hair loss with black pigmented dots at the follicular openings, diffuse scalp scaling with subtle hair loss, and cervical lymphadenopathy.12 Although less common, tinea capitis can progress to a more severe form known as a kerion, which is characterized by a tender plaque with pustules and crusting. A kerion can result in permanent scarring and alopecia if left untreated.12 In our patient, a nodule with scaling and faint erythema was observed, but no black pigmented dots at the follicular orifices were present. Therefore, a potassium hydroxide wet mount preparation used to diagnose tinea capitis was unnecessary. Systemic oral antifungal therapy such as fluconazole or terbinafine is the standard treatment for tinea capitis.
The Diagnosis: Subcutaneous Fat Necrosis
Histopathology revealed lobular panniculitis with lymphohistiocytic inflammation, lipid crystals, and calcifications in our patient (Figure). Subcutaneous fat necrosis (SCFN) was diagnosed based on these characteristic histopathologic findings. No further treatment was pursued.

Subcutaneous fat necrosis is a rare, self-limiting panniculitis that typically resolves within several weeks to months without scarring. It manifests as red or violaceous subcutaneous nodules or plaques most commonly on the buttocks, trunk, proximal arms and legs, and cheeks.1 Histopathology reveals lobular panniculitis with dense granulomatous infiltrates of histiocytes, eosinophils, and multinucleated giant cells with needle-shaped crystals. Focal areas of fat necrosis with calcification also can be seen.2
The epidemiology of SCFN is unknown. Most cases occur in healthy full-term to postterm neonates who experience hypoxia, other prenatal stressors, or therapeutic hypothermia for the treatment of hypoxic-ischemic encephalopathy.3 Although the etiology is unclear, certain inciting factors such as local tissue hypoxia, cold exposure, meconium aspiration, maternal diabetes, preeclampsia, and mechanical pressure have been proposed. Our patient underwent hypothermic cooling protocol, and it has been suggested that the increased saturated to unsaturated fat concentration in the skin of newborns increases the melting point, thus predisposing them to fat crystalization.4 Cases of SCFN involving the scalp are rare; therefore, any newborns receiving hypothermic therapy for hypoxic-ischemic encephalopathy should have a thorough skin examination with possible biopsy of lesions that are characteristic of SCFN, such as red or violaceous subcutaneous nodules or plaques, for specific disease identification.
The main complication of SCFN is hypercalcemia, which occurs in approximately 50% of cases. Other serum abnormalities include hyperglycemia, hypertriglyceridemia, and thrombocytopenia, though these findings are not as well associated.4 Patients with associated hypercalcemia may be asymptomatic, as in our patient, but other presentations include irritability, weakness, anorexia, vomiting, renal failure, failure to thrive, and encephalopathy. Nephrocalcinosis is a common complication of severe hypercalcemia; however, there is little evidence of associated major renal dysfunction.5 The exact mechanism of hypercalcemia is poorly understood. A widely accepted theory postulates that a granulomatous inflammatory infiltrate upregulates 1-α-hydroxylase activity, which enzymatically converts 25-hydroxyvitamin D to its active form, 1,25-dihydroxycholecalciferol, which increases bone resorption and calcium absorption through the gastrointestinal tract and renal systems. Treatments for hypercalcemia include hyperhydration, calcium-wasting diuretics, and low calcium intake.6 Furthermore, calcium levels should be obtained at the time of diagnosis and 30, 45, and 60 days after the lesions resolve.4
Subcutaneous fat necrosis needs to be differentiated from the more severe panniculitis, sclerema neonatorum (SN), which typically affects critically ill, preterm, and small-for-gestational-age newborns. It is associated with a high mortality rate and is characterized by skin and subadjacent tissue structures. The process typically begins in the thighs, buttocks, or trunk and spreads diffusely, sparing the fat-free palms, soles, and genitalia.7 Although our patient was born preterm, the physical characteristics of the nodule and the lack of severe illness placed SN lower on our differential. Histopathologic differences between SCFN and SN involve the extent of tissue fibrosis and presence of inflammatory cells. Sclerema neonatorum typically manifests with thickened connective tissue with a sparse inflammatory infiltrate, including lymphocytes, histiocytes, and multinucleated giant cells.7 Conversely, SCFN manifests with fat necrosis with an extensive inflammatory infiltrate. It is important to be able to distinguish between these 2 conditions, as both have vastly different prognoses.
Cold panniculitis, sometimes called “popsicle panniculitis,” is a phenomenon in which cold contact with the skin causes eruption of firm, erythematous, indurated plaques at the site of exposure. This self-limiting condition typically appears hours to days after cold exposure and spontaneously resolves in a few weeks.8 Therapeutic hypothermic protocol treatment involves using cooling devices to lower the body temperature for a short duration. The temperature typically is lowered to approximately 32 °C to 36 °C. These temperatures are not low enough to induce cold panniculitis, which is more commonly seen in facial ice applications when managing supraventricular tachycardia in neonates.
Cephalohematoma is a birthing injury that causes blood accumulation within the subperiosteal space. During parturition, the compressive and sheering forces on the calvarium rupture the vessels passing through the periosteum, causing blood to pool slowly into the subperiostium; thus, a cephalohematoma usually manifests later at 1 to 3 days of life as localized head swelling.9 The bleeding typically does not cross suture lines and is primarily found in the occipital or parietal regions. The incidence has been reported to be 0.4% to 2.5% of all live births.10 Although the location of the nodule in our patient was in the occipital region, imaging and biopsy results did not show hemorrhagic findings consistent with cephalohematoma. Management of cephalohematoma mainly is observational, as the mass slowly regresses and the accumulated blood gradually is reabsorbed.
Fungal scalp infections (tinea capitis) are common in the pediatric population. The peak incidence of this infection has been reported in children aged 3 to 7 years, with Trichophyton tonsurans and Microsporum canis as the usual causative organisms.11 Clinical features of tinea capitis include scaly patches with hair loss, hair loss with black pigmented dots at the follicular openings, diffuse scalp scaling with subtle hair loss, and cervical lymphadenopathy.12 Although less common, tinea capitis can progress to a more severe form known as a kerion, which is characterized by a tender plaque with pustules and crusting. A kerion can result in permanent scarring and alopecia if left untreated.12 In our patient, a nodule with scaling and faint erythema was observed, but no black pigmented dots at the follicular orifices were present. Therefore, a potassium hydroxide wet mount preparation used to diagnose tinea capitis was unnecessary. Systemic oral antifungal therapy such as fluconazole or terbinafine is the standard treatment for tinea capitis.
- Coondoo A, Lahiry R, Choudhury A, et al. Tender skin nodules in a newborn. Indian J Dermatol. 2013;58:328. doi:10.4103/0019-5154.113983
- Mitra S, Dove J, Somisetty SK. Subcutaneous fat necrosis in newbornan unusual case and review of literature. Eur J Pediatr. 2011;170:1107- 1110. doi:10.1007/s00431-011-1405-x
- Velasquez JH, Mendez MD. Newborn subcutaneous fat necrosis. In: StatPearls. StatPearls Publishing; 2022.
- Stefanko NS, Drolet BA. Subcutaneous fat necrosis of the newborn and associated hypercalcemia: a systematic review of the literature. Pediatr Dermatol. 2019;36:24-30. doi:10.1111/pde.13640
- Shumer DE, Thaker V, Taylor GA, et al. Severe hypercalcaemia due to subcutaneous fat necrosis: presentation, management and complications. Arch Dis Child Fetal Neonatal Ed. 2014;99:F419-F421. doi:10.1136/ archdischild-2014-306069
- Farooque A, Moss C, Zehnder D, et al. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in subcutaneous fat necrosis. Br J Dermatol. 2009;160:423-425. doi:10.1111/j.1365-2133.2008.08844.x
- Zeb A, Darmstadt GL. Sclerema neonatorum: a review of nomenclature, clinical presentation, histological features, differential diagnoses and management. J Perinatol. 2008;28:453-460. doi:10.1038/jp.2008.33
- Quesada-Cortés A, Campos-Muñoz L, Díaz-Díaz RM, et al. Cold panniculitis. Dermatol Clin. 2008;26:485-489, vii. doi:10.1016 /j.det.2008.05.015
- Raines DA, Krawiec C, Jain S. Cephalohematoma. In: StatPearls. StatPearls Publishing; 2023.
- Chung HY, Chung JY, Lee DG, et al. Surgical treatment of ossified cephalhematoma. J Craniofac Surg. 2004;15:774-779. doi:10.1097/00001665- 200409000-00015
- Leung AKC, Hon KL, Leong KF, et al. Tinea capitis: an updated review. Recent Pat Inflamm Allergy Drug Discov. 2020;14:58-68. doi:10.2174/1872 213x14666200106145624
- Kovitwanichkanont T, Chong A. Superficial fungal infections. Aust J Gen Pract. 2019;48:706-711. doi:10.31128/ajgp-05-19-4930
- Coondoo A, Lahiry R, Choudhury A, et al. Tender skin nodules in a newborn. Indian J Dermatol. 2013;58:328. doi:10.4103/0019-5154.113983
- Mitra S, Dove J, Somisetty SK. Subcutaneous fat necrosis in newbornan unusual case and review of literature. Eur J Pediatr. 2011;170:1107- 1110. doi:10.1007/s00431-011-1405-x
- Velasquez JH, Mendez MD. Newborn subcutaneous fat necrosis. In: StatPearls. StatPearls Publishing; 2022.
- Stefanko NS, Drolet BA. Subcutaneous fat necrosis of the newborn and associated hypercalcemia: a systematic review of the literature. Pediatr Dermatol. 2019;36:24-30. doi:10.1111/pde.13640
- Shumer DE, Thaker V, Taylor GA, et al. Severe hypercalcaemia due to subcutaneous fat necrosis: presentation, management and complications. Arch Dis Child Fetal Neonatal Ed. 2014;99:F419-F421. doi:10.1136/ archdischild-2014-306069
- Farooque A, Moss C, Zehnder D, et al. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in subcutaneous fat necrosis. Br J Dermatol. 2009;160:423-425. doi:10.1111/j.1365-2133.2008.08844.x
- Zeb A, Darmstadt GL. Sclerema neonatorum: a review of nomenclature, clinical presentation, histological features, differential diagnoses and management. J Perinatol. 2008;28:453-460. doi:10.1038/jp.2008.33
- Quesada-Cortés A, Campos-Muñoz L, Díaz-Díaz RM, et al. Cold panniculitis. Dermatol Clin. 2008;26:485-489, vii. doi:10.1016 /j.det.2008.05.015
- Raines DA, Krawiec C, Jain S. Cephalohematoma. In: StatPearls. StatPearls Publishing; 2023.
- Chung HY, Chung JY, Lee DG, et al. Surgical treatment of ossified cephalhematoma. J Craniofac Surg. 2004;15:774-779. doi:10.1097/00001665- 200409000-00015
- Leung AKC, Hon KL, Leong KF, et al. Tinea capitis: an updated review. Recent Pat Inflamm Allergy Drug Discov. 2020;14:58-68. doi:10.2174/1872 213x14666200106145624
- Kovitwanichkanont T, Chong A. Superficial fungal infections. Aust J Gen Pract. 2019;48:706-711. doi:10.31128/ajgp-05-19-4930
A 4-week-old male infant was referred to dermatology for evaluation of a nodule on the occipital protuberance of 2 weeks’ duration. The patient was born at 36 weeks and 6 days’ gestation via an emergency cesarean delivery due to fetal distress. He later was found to have hypoxic-ischemic encephalopathy, pulmonary hypertension, and hypertrophic cardiomyopathy. He underwent therapeutic hypothermia protocol treatment starting at less than 6 hours after birth. At the current presentation, physical examination showed a 2.5-cm, erythematous, firm, mobile nodule on the occipital scalp with some overlying crusting and minimal surrounding erythema. No other cutaneous features or lesions were present. Initial laboratory findings were remarkable for hypercalcemia at 11 mg/dL (reference range, 8.5-10.5 mg/dL). Magnetic resonance imaging showed a faint abnormality in the subcutaneous tissue in this region without a noted connection to the underlying brain/meningeal matter. A punch biopsy was performed.
The Obesogenic Environment of Preschool and Day Care
Thirty years ago I had an experience in the office that influenced my approach to obesity for the rest of my career. The patient was a 4-year-old whom I had been seeing since her birth. At her annual well-child visit her weight had jumped up significantly from the previous year’s visit. She appeared well, but the change in her growth trajectory prompted a bit more in-depth history taking.
It turned out that finances had forced the family to employ one of the child’s grandmothers as the day care provider. Unfortunately, this grandmother’s passion was cooking and she was particularly adept at baking. She had no other hobbies and a sore hip limited her mobility, so she seldom went outside. When I eventually met her she was a cheerful, overweight, and delightful woman.
Deconstructing this obesogenic environment without disrupting this otherwise healthy family was an exercise that required tact, patience, and creativity. Fortunately, the young girl’s mother had already harbored some concerns about her child’s weight and was more than willing to participate in this environmental re-engineering project. It’s a long story, but she and I achieved our goals and the child eventually coasted back toward her previous growth curve.
I have always suspected that this scenario is being replayed hundreds of thousands of time across this country. But, sadly most don’t share this one’s happy ending. Parents don’t alway perceive the seriousness of the problem. The economic hurdles are often too steep to overcome, even when the most creative minds are involved.
How prevalent are obesogenic day care environments? We certainly know childhood obesity is a problem and the statistics in the preschool age group are particularly concerning. More than 14 million children are in non-parental early care and education programs; these environments would seem to be a logical place to target our prevention strategies. Understandably, there seems to be a hesitancy to point fingers, but how many day care providers are similar to the well-intentioned grandmother in the scenario I described? We must at least suspect that the example set by the adults in the preschool and day care environment might be having some influence on the children under their care.
There has been some research that sheds some light on this question. A paper from the University of Oklahoma has looked at the predictors of overweight and obesity in early care and education (ECE) teachers in hopes of “finding modifiable opportunities to enhance the health of this critical workforce.” In their paper, the investigators refer to other research that has found the prevalence of overweight and obesity among ECE teachers is higher than our national average and their waist circumference is significantly greater than the standard recommendation for women.
A study from Norway has looked at the association between preschool staff’s activity level and that of the children under their care using accelerometers. This particular investigation couldn’t determine whether it was the staff’s activity level that influenced the children or vice versa because it wasn’t an observational study. Common sense would lead one to believe it was the staff’s relative inactivity that was being reflected in the children’s.
It is interesting that in this Norwegian study when the teachers were asked about their attitudes toward activity and their self-perception of their own activity, there was no relationship between the staff’s and the children’s level of activity. In other words, the educators and caregivers bought into the importance of activity but had difficulty translating this philosophy into own behavior.
So where does this leave us? It turns out my experience decades ago was not a one-off event, but instead represents the tip of very large iceberg. Should we immediately create a system of day care provider boot camps? Let’s remember that each educator and caregiver is one of us. They may be slight outliers but not a group of individuals deserving of forced marches and half-rations to get them in shape.
ECEs have listened to the same message we have all heard about diet and activity and their importance for a child’s health. It’s for their own health and that of their charges. This could be as simple as providing accelerometers or step-counting smartwatches. Or, by having physical educators perform on-site audits that could then be used to create site-specific plans for increasing both teacher and student activity.
Modifying the educators’ diet is a more complex procedure and can quickly become entangled in the socio-economic background of each individual teacher. A healthy diet is not always equally available to everyone. The solution may involve providing the teachers with food to be eaten at work and to be prepared at home. But, creative answers can be found if we look for them.
Before we get too far down the obesity-is-a-disease pathway, we must take a closer look at the role the early care and early school milieu are playing in the obesity problem. A little common sense behavior modification when children are in the controlled environment of school/day care may allow us to be less reliant on the those new wonder drugs in the long run.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Thirty years ago I had an experience in the office that influenced my approach to obesity for the rest of my career. The patient was a 4-year-old whom I had been seeing since her birth. At her annual well-child visit her weight had jumped up significantly from the previous year’s visit. She appeared well, but the change in her growth trajectory prompted a bit more in-depth history taking.
It turned out that finances had forced the family to employ one of the child’s grandmothers as the day care provider. Unfortunately, this grandmother’s passion was cooking and she was particularly adept at baking. She had no other hobbies and a sore hip limited her mobility, so she seldom went outside. When I eventually met her she was a cheerful, overweight, and delightful woman.
Deconstructing this obesogenic environment without disrupting this otherwise healthy family was an exercise that required tact, patience, and creativity. Fortunately, the young girl’s mother had already harbored some concerns about her child’s weight and was more than willing to participate in this environmental re-engineering project. It’s a long story, but she and I achieved our goals and the child eventually coasted back toward her previous growth curve.
I have always suspected that this scenario is being replayed hundreds of thousands of time across this country. But, sadly most don’t share this one’s happy ending. Parents don’t alway perceive the seriousness of the problem. The economic hurdles are often too steep to overcome, even when the most creative minds are involved.
How prevalent are obesogenic day care environments? We certainly know childhood obesity is a problem and the statistics in the preschool age group are particularly concerning. More than 14 million children are in non-parental early care and education programs; these environments would seem to be a logical place to target our prevention strategies. Understandably, there seems to be a hesitancy to point fingers, but how many day care providers are similar to the well-intentioned grandmother in the scenario I described? We must at least suspect that the example set by the adults in the preschool and day care environment might be having some influence on the children under their care.
There has been some research that sheds some light on this question. A paper from the University of Oklahoma has looked at the predictors of overweight and obesity in early care and education (ECE) teachers in hopes of “finding modifiable opportunities to enhance the health of this critical workforce.” In their paper, the investigators refer to other research that has found the prevalence of overweight and obesity among ECE teachers is higher than our national average and their waist circumference is significantly greater than the standard recommendation for women.
A study from Norway has looked at the association between preschool staff’s activity level and that of the children under their care using accelerometers. This particular investigation couldn’t determine whether it was the staff’s activity level that influenced the children or vice versa because it wasn’t an observational study. Common sense would lead one to believe it was the staff’s relative inactivity that was being reflected in the children’s.
It is interesting that in this Norwegian study when the teachers were asked about their attitudes toward activity and their self-perception of their own activity, there was no relationship between the staff’s and the children’s level of activity. In other words, the educators and caregivers bought into the importance of activity but had difficulty translating this philosophy into own behavior.
So where does this leave us? It turns out my experience decades ago was not a one-off event, but instead represents the tip of very large iceberg. Should we immediately create a system of day care provider boot camps? Let’s remember that each educator and caregiver is one of us. They may be slight outliers but not a group of individuals deserving of forced marches and half-rations to get them in shape.
ECEs have listened to the same message we have all heard about diet and activity and their importance for a child’s health. It’s for their own health and that of their charges. This could be as simple as providing accelerometers or step-counting smartwatches. Or, by having physical educators perform on-site audits that could then be used to create site-specific plans for increasing both teacher and student activity.
Modifying the educators’ diet is a more complex procedure and can quickly become entangled in the socio-economic background of each individual teacher. A healthy diet is not always equally available to everyone. The solution may involve providing the teachers with food to be eaten at work and to be prepared at home. But, creative answers can be found if we look for them.
Before we get too far down the obesity-is-a-disease pathway, we must take a closer look at the role the early care and early school milieu are playing in the obesity problem. A little common sense behavior modification when children are in the controlled environment of school/day care may allow us to be less reliant on the those new wonder drugs in the long run.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Thirty years ago I had an experience in the office that influenced my approach to obesity for the rest of my career. The patient was a 4-year-old whom I had been seeing since her birth. At her annual well-child visit her weight had jumped up significantly from the previous year’s visit. She appeared well, but the change in her growth trajectory prompted a bit more in-depth history taking.
It turned out that finances had forced the family to employ one of the child’s grandmothers as the day care provider. Unfortunately, this grandmother’s passion was cooking and she was particularly adept at baking. She had no other hobbies and a sore hip limited her mobility, so she seldom went outside. When I eventually met her she was a cheerful, overweight, and delightful woman.
Deconstructing this obesogenic environment without disrupting this otherwise healthy family was an exercise that required tact, patience, and creativity. Fortunately, the young girl’s mother had already harbored some concerns about her child’s weight and was more than willing to participate in this environmental re-engineering project. It’s a long story, but she and I achieved our goals and the child eventually coasted back toward her previous growth curve.
I have always suspected that this scenario is being replayed hundreds of thousands of time across this country. But, sadly most don’t share this one’s happy ending. Parents don’t alway perceive the seriousness of the problem. The economic hurdles are often too steep to overcome, even when the most creative minds are involved.
How prevalent are obesogenic day care environments? We certainly know childhood obesity is a problem and the statistics in the preschool age group are particularly concerning. More than 14 million children are in non-parental early care and education programs; these environments would seem to be a logical place to target our prevention strategies. Understandably, there seems to be a hesitancy to point fingers, but how many day care providers are similar to the well-intentioned grandmother in the scenario I described? We must at least suspect that the example set by the adults in the preschool and day care environment might be having some influence on the children under their care.
There has been some research that sheds some light on this question. A paper from the University of Oklahoma has looked at the predictors of overweight and obesity in early care and education (ECE) teachers in hopes of “finding modifiable opportunities to enhance the health of this critical workforce.” In their paper, the investigators refer to other research that has found the prevalence of overweight and obesity among ECE teachers is higher than our national average and their waist circumference is significantly greater than the standard recommendation for women.
A study from Norway has looked at the association between preschool staff’s activity level and that of the children under their care using accelerometers. This particular investigation couldn’t determine whether it was the staff’s activity level that influenced the children or vice versa because it wasn’t an observational study. Common sense would lead one to believe it was the staff’s relative inactivity that was being reflected in the children’s.
It is interesting that in this Norwegian study when the teachers were asked about their attitudes toward activity and their self-perception of their own activity, there was no relationship between the staff’s and the children’s level of activity. In other words, the educators and caregivers bought into the importance of activity but had difficulty translating this philosophy into own behavior.
So where does this leave us? It turns out my experience decades ago was not a one-off event, but instead represents the tip of very large iceberg. Should we immediately create a system of day care provider boot camps? Let’s remember that each educator and caregiver is one of us. They may be slight outliers but not a group of individuals deserving of forced marches and half-rations to get them in shape.
ECEs have listened to the same message we have all heard about diet and activity and their importance for a child’s health. It’s for their own health and that of their charges. This could be as simple as providing accelerometers or step-counting smartwatches. Or, by having physical educators perform on-site audits that could then be used to create site-specific plans for increasing both teacher and student activity.
Modifying the educators’ diet is a more complex procedure and can quickly become entangled in the socio-economic background of each individual teacher. A healthy diet is not always equally available to everyone. The solution may involve providing the teachers with food to be eaten at work and to be prepared at home. But, creative answers can be found if we look for them.
Before we get too far down the obesity-is-a-disease pathway, we must take a closer look at the role the early care and early school milieu are playing in the obesity problem. A little common sense behavior modification when children are in the controlled environment of school/day care may allow us to be less reliant on the those new wonder drugs in the long run.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Pediatric Patients With MS May Do Best on High-Efficacy DMTs
DENVER — Patients with pediatric-onset multiple sclerosis (POMS) are often prescribed low-efficacy disease-modifying therapies (DMTs), but a new retrospective analysis suggests that, like adults, this patient population may benefit from early treatment with high-efficacy DMTs.
“I think it’s very important to highlight that we are seeing that traditionally, kids are just started on lower-efficacy treatments and they keep relapsing. If we can show that when they get transitioned to high-efficacy treatments, the relapses are lessening, I’m hoping that can then push for better clinical trials with pediatric patients included,” said Frederick Bassal, DO, who presented the study during a poster session at the 2024 annual meeting of the American Academy of Neurology. He is a pediatric neurologist at University of California, Davis.
The first line for POMS is generally low-efficacy DMTs like interferon-beta and glatiramer acetate, but these medications may not effectively control disease progression, according to the study authors, and this could lead to pediatric patients being changed to more potent therapies. That can include moderate-efficacy drugs like S1P inhibitors and fumarates, or high-efficacy DMTS such as B cell depletors and alpha 4 integrin receptor antibodies.
Treatment Strategies
“Right now what we’re seeing is the conservative approach — starting low and working up with the younger and adolescent patients. I’m speculating, and I want to look more into it. Is [it maybe] because of insurance approval?” said study coauthor Amara Miller, a medical student at the University of Arizona College of Medicine in Phoenix.
The findings aren’t surprising, according to Barbara Giesser, MD, who was asked to comment on the study. “It is in line with what we think we know about people with adult MS — that if you start early on with a more effective therapy, you tend to have better outcomes,” said Dr. Giesser, director of the MS program at the Pacific Neuroscience Institute.
Another reason to consider higher-efficacy DMTs is that children with MS can have cognitive problems and delays. “There’s a suggestion that if you treat with highly-effective DMT that you might be able to abrogate some of that,” said Dr. Giesser.
Among the approximately two dozen FDA-approved disease-modifying therapies for MS, only fingolimod (Gilenya, Novartis) is approved for children and adolescents. “All of the others are used off label, but I think perhaps, if you have more studies that [show] that children do better if you treat with more effective therapies early on, perhaps we might see more on-label indications for use in a pediatric population,” said Dr. Giesser.
The finding that obesity was associated with a higher likelihood of having multiple therapies is also interesting, she said. “We’re beginning to see that obesity in adults as well seems to portend less favorable neurologic outcomes.”
Study Methodology
The researchers analyzed data from 135 POMS patients between 2012 and 2023.
The mean age of participants was 15 years, 60% were female, and 120 of 135 were White, while 76 were of Hispanic ethnicity. Overweight and obesity were common, with 36 and 43 participants in each category. The initial therapy was a low-efficacy DMT in 23.0% of participants, moderate-efficacy in 37.0%, and high-efficacy in 24.4%, while 1.5% received some other kind of medication and 14.1% received no medication. The annualized relapse rate was 0.932, and the mean EDSS score was 0.88.
Patients who underwent three or medication changes had lower EDSS scores than those who underwent zero to 2 (P = .00607).
Over the course of the study, the percentage of patients who received high-efficacy DMTs rose from 25.9% to 48.9%, largely due to changes in medication. Of those initially prescribed low-efficacy DMTs, 77.4% were eventually switched to high-efficacy DMTs.
Every patient who underwent three or more medication changes was initially prescribed a low-efficacy DMT.
Patients started on low-efficacy drugs had a mean of 1.42 medication changes, compared with 0.94 in the moderate-efficacy group and 0.51 in the high-efficacy group. The reasons for changing from the first medication included relapse (36), side effects (11), patient choice or compliance (8), and pregnancy (2).
Patients 10 years or younger were more likely to be initially prescribed a low-efficacy therapy (odds ratio [OR], 5.72; P = .0366), while older patients were more likely to be prescribed moderate- or high-efficacy therapies (OR, 14.44; P = .0012).
There were more total medication changes in the low-efficacy group than the high initial DMT group (P = .000305).
Asked what advice they would give to physicians treating POMS patients, Ms. Miller suggested a top-down approach. “We want to look at if maybe we can start with higher efficacy DMT’s and maybe titering it down. That may be an option,” said Ms. Miller.
Dr. Bassal highlighted the importance of shared decision-making. “We want to go over the options, that we recommend higher-efficacy [DMTs] for these reasons. But every individual is different. And there may be fears that are very reasonable that families have. I think in those cases, it’s also reasonable to make a shared decision. And if that means going with something like an oral, moderate- to lower-efficacy [therapy], that’s okay, because compliance is key, and if you start something where the family is afraid of side effects, or there are side effects, then you kind of lost that opportunity,” he said.
Dr. Bassal, Dr. Giesser, and Ms. Miller have no relevant financial disclosures.
DENVER — Patients with pediatric-onset multiple sclerosis (POMS) are often prescribed low-efficacy disease-modifying therapies (DMTs), but a new retrospective analysis suggests that, like adults, this patient population may benefit from early treatment with high-efficacy DMTs.
“I think it’s very important to highlight that we are seeing that traditionally, kids are just started on lower-efficacy treatments and they keep relapsing. If we can show that when they get transitioned to high-efficacy treatments, the relapses are lessening, I’m hoping that can then push for better clinical trials with pediatric patients included,” said Frederick Bassal, DO, who presented the study during a poster session at the 2024 annual meeting of the American Academy of Neurology. He is a pediatric neurologist at University of California, Davis.
The first line for POMS is generally low-efficacy DMTs like interferon-beta and glatiramer acetate, but these medications may not effectively control disease progression, according to the study authors, and this could lead to pediatric patients being changed to more potent therapies. That can include moderate-efficacy drugs like S1P inhibitors and fumarates, or high-efficacy DMTS such as B cell depletors and alpha 4 integrin receptor antibodies.
Treatment Strategies
“Right now what we’re seeing is the conservative approach — starting low and working up with the younger and adolescent patients. I’m speculating, and I want to look more into it. Is [it maybe] because of insurance approval?” said study coauthor Amara Miller, a medical student at the University of Arizona College of Medicine in Phoenix.
The findings aren’t surprising, according to Barbara Giesser, MD, who was asked to comment on the study. “It is in line with what we think we know about people with adult MS — that if you start early on with a more effective therapy, you tend to have better outcomes,” said Dr. Giesser, director of the MS program at the Pacific Neuroscience Institute.
Another reason to consider higher-efficacy DMTs is that children with MS can have cognitive problems and delays. “There’s a suggestion that if you treat with highly-effective DMT that you might be able to abrogate some of that,” said Dr. Giesser.
Among the approximately two dozen FDA-approved disease-modifying therapies for MS, only fingolimod (Gilenya, Novartis) is approved for children and adolescents. “All of the others are used off label, but I think perhaps, if you have more studies that [show] that children do better if you treat with more effective therapies early on, perhaps we might see more on-label indications for use in a pediatric population,” said Dr. Giesser.
The finding that obesity was associated with a higher likelihood of having multiple therapies is also interesting, she said. “We’re beginning to see that obesity in adults as well seems to portend less favorable neurologic outcomes.”
Study Methodology
The researchers analyzed data from 135 POMS patients between 2012 and 2023.
The mean age of participants was 15 years, 60% were female, and 120 of 135 were White, while 76 were of Hispanic ethnicity. Overweight and obesity were common, with 36 and 43 participants in each category. The initial therapy was a low-efficacy DMT in 23.0% of participants, moderate-efficacy in 37.0%, and high-efficacy in 24.4%, while 1.5% received some other kind of medication and 14.1% received no medication. The annualized relapse rate was 0.932, and the mean EDSS score was 0.88.
Patients who underwent three or medication changes had lower EDSS scores than those who underwent zero to 2 (P = .00607).
Over the course of the study, the percentage of patients who received high-efficacy DMTs rose from 25.9% to 48.9%, largely due to changes in medication. Of those initially prescribed low-efficacy DMTs, 77.4% were eventually switched to high-efficacy DMTs.
Every patient who underwent three or more medication changes was initially prescribed a low-efficacy DMT.
Patients started on low-efficacy drugs had a mean of 1.42 medication changes, compared with 0.94 in the moderate-efficacy group and 0.51 in the high-efficacy group. The reasons for changing from the first medication included relapse (36), side effects (11), patient choice or compliance (8), and pregnancy (2).
Patients 10 years or younger were more likely to be initially prescribed a low-efficacy therapy (odds ratio [OR], 5.72; P = .0366), while older patients were more likely to be prescribed moderate- or high-efficacy therapies (OR, 14.44; P = .0012).
There were more total medication changes in the low-efficacy group than the high initial DMT group (P = .000305).
Asked what advice they would give to physicians treating POMS patients, Ms. Miller suggested a top-down approach. “We want to look at if maybe we can start with higher efficacy DMT’s and maybe titering it down. That may be an option,” said Ms. Miller.
Dr. Bassal highlighted the importance of shared decision-making. “We want to go over the options, that we recommend higher-efficacy [DMTs] for these reasons. But every individual is different. And there may be fears that are very reasonable that families have. I think in those cases, it’s also reasonable to make a shared decision. And if that means going with something like an oral, moderate- to lower-efficacy [therapy], that’s okay, because compliance is key, and if you start something where the family is afraid of side effects, or there are side effects, then you kind of lost that opportunity,” he said.
Dr. Bassal, Dr. Giesser, and Ms. Miller have no relevant financial disclosures.
DENVER — Patients with pediatric-onset multiple sclerosis (POMS) are often prescribed low-efficacy disease-modifying therapies (DMTs), but a new retrospective analysis suggests that, like adults, this patient population may benefit from early treatment with high-efficacy DMTs.
“I think it’s very important to highlight that we are seeing that traditionally, kids are just started on lower-efficacy treatments and they keep relapsing. If we can show that when they get transitioned to high-efficacy treatments, the relapses are lessening, I’m hoping that can then push for better clinical trials with pediatric patients included,” said Frederick Bassal, DO, who presented the study during a poster session at the 2024 annual meeting of the American Academy of Neurology. He is a pediatric neurologist at University of California, Davis.
The first line for POMS is generally low-efficacy DMTs like interferon-beta and glatiramer acetate, but these medications may not effectively control disease progression, according to the study authors, and this could lead to pediatric patients being changed to more potent therapies. That can include moderate-efficacy drugs like S1P inhibitors and fumarates, or high-efficacy DMTS such as B cell depletors and alpha 4 integrin receptor antibodies.
Treatment Strategies
“Right now what we’re seeing is the conservative approach — starting low and working up with the younger and adolescent patients. I’m speculating, and I want to look more into it. Is [it maybe] because of insurance approval?” said study coauthor Amara Miller, a medical student at the University of Arizona College of Medicine in Phoenix.
The findings aren’t surprising, according to Barbara Giesser, MD, who was asked to comment on the study. “It is in line with what we think we know about people with adult MS — that if you start early on with a more effective therapy, you tend to have better outcomes,” said Dr. Giesser, director of the MS program at the Pacific Neuroscience Institute.
Another reason to consider higher-efficacy DMTs is that children with MS can have cognitive problems and delays. “There’s a suggestion that if you treat with highly-effective DMT that you might be able to abrogate some of that,” said Dr. Giesser.
Among the approximately two dozen FDA-approved disease-modifying therapies for MS, only fingolimod (Gilenya, Novartis) is approved for children and adolescents. “All of the others are used off label, but I think perhaps, if you have more studies that [show] that children do better if you treat with more effective therapies early on, perhaps we might see more on-label indications for use in a pediatric population,” said Dr. Giesser.
The finding that obesity was associated with a higher likelihood of having multiple therapies is also interesting, she said. “We’re beginning to see that obesity in adults as well seems to portend less favorable neurologic outcomes.”
Study Methodology
The researchers analyzed data from 135 POMS patients between 2012 and 2023.
The mean age of participants was 15 years, 60% were female, and 120 of 135 were White, while 76 were of Hispanic ethnicity. Overweight and obesity were common, with 36 and 43 participants in each category. The initial therapy was a low-efficacy DMT in 23.0% of participants, moderate-efficacy in 37.0%, and high-efficacy in 24.4%, while 1.5% received some other kind of medication and 14.1% received no medication. The annualized relapse rate was 0.932, and the mean EDSS score was 0.88.
Patients who underwent three or medication changes had lower EDSS scores than those who underwent zero to 2 (P = .00607).
Over the course of the study, the percentage of patients who received high-efficacy DMTs rose from 25.9% to 48.9%, largely due to changes in medication. Of those initially prescribed low-efficacy DMTs, 77.4% were eventually switched to high-efficacy DMTs.
Every patient who underwent three or more medication changes was initially prescribed a low-efficacy DMT.
Patients started on low-efficacy drugs had a mean of 1.42 medication changes, compared with 0.94 in the moderate-efficacy group and 0.51 in the high-efficacy group. The reasons for changing from the first medication included relapse (36), side effects (11), patient choice or compliance (8), and pregnancy (2).
Patients 10 years or younger were more likely to be initially prescribed a low-efficacy therapy (odds ratio [OR], 5.72; P = .0366), while older patients were more likely to be prescribed moderate- or high-efficacy therapies (OR, 14.44; P = .0012).
There were more total medication changes in the low-efficacy group than the high initial DMT group (P = .000305).
Asked what advice they would give to physicians treating POMS patients, Ms. Miller suggested a top-down approach. “We want to look at if maybe we can start with higher efficacy DMT’s and maybe titering it down. That may be an option,” said Ms. Miller.
Dr. Bassal highlighted the importance of shared decision-making. “We want to go over the options, that we recommend higher-efficacy [DMTs] for these reasons. But every individual is different. And there may be fears that are very reasonable that families have. I think in those cases, it’s also reasonable to make a shared decision. And if that means going with something like an oral, moderate- to lower-efficacy [therapy], that’s okay, because compliance is key, and if you start something where the family is afraid of side effects, or there are side effects, then you kind of lost that opportunity,” he said.
Dr. Bassal, Dr. Giesser, and Ms. Miller have no relevant financial disclosures.
FROM AAN 2024
How Does Moderate to Severe Eczema Affect Growth in Children?
FROM AAD 2024
SAN DIEGO — , results from an ongoing 10-year observational study showed.
“We need to sort out whether this is reversed by newer treatments, especially in the 6- to 11-year-olds, as well as the factors that underlie it in atopic dermatitis,” said the study’s first author Amy S. Paller, MD, chair of dermatology, Northwestern University, Chicago, Illinois, following the annual meeting of the American Academy of Dermatology, where the study was presented during a poster session.
Atopic Dermatitis Impacts Growth
In the ongoing international study called PEDISTAD, researchers enrolled 1326 children younger than 12 years with moderate to severe atopic dermatitis inadequately controlled by topical therapies who were candidates to receive systemic medications. They assessed the percentage of patients above the 50th percentile and the mean percentiles for height, weight, and body mass index (BMI) at baseline against the Centers for Disease Control and Prevention’s (CDC’s) Learning Management System reference healthy population, by age in months, and compared results to the CDC’s standardized growth curves for healthy children aged 0-12 years.
The investigators found that at baseline, compared with the age-specific population norms, 50% of men and 51% of women in the PEDISTAD study were above the 50th percentile for weight, but only 38% and 52%, respectively, were above the 50th percentile for height. Among patients aged 5-12 years, only 28% of men and 47% of women were above the 50th percentile for height, while 69% of men and 71% of women were above the 50th percentile for BMI.
Dr. Paller said that she was “not really surprised by the reduction in linear growth, since there are so many factors that may contribute,” including chronic inflammation, poor sleep, and the use of topical and systemic steroids. “But [it’s] good to have this data as an opportunity to see if our improved therapies can reverse this.”
She said that she was “a bit surprised by the increase in weight and body mass index, but this could reflect less physical activity/sports [participation and] deserves more investigation,” and added that the findings “mesh nicely with new attention on bone growth with good control of atopic dermatitis in this age group.”
Dr. Paller acknowledged certain limitations of the study, including the fact that those enrolled are a heterogeneous cohort with variable treatment regimens.
Some Answers, More Questions
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, who was asked to comment on the findings, said that atopic dermatitis “should be considered the cutaneous manifestations of a systemic inflammatory disease, though even if it were not, the impact on daily and nightly activities [such as sleep] could indirectly have systemic medical consequences.”
The data presented “highlights that children with moderate to severe disease have higher BMIs and shorter height than matched counterparts, likely owing to the treasure trove of direct and indirect consequences of uncontrolled type 2 inflammation,” he said. “What I would like to know, and as the authors astutely noted, could treatment, and especially early intervention, prevent or even alter this impact?”
Dr. Paller disclosed that she is a consultant for several pharmaceutical companies, including Sanofi and Regeneron, the study sponsor. She is also an investigator for AbbVie, Dermavant, Eli Lilly, Incyte, Janssen, Krystal, LEO Pharma, and UCB and is a member of the data monitoring safety board for AbbVie, Abeona, Catawba, Galderma, and InMed. Dr. Friedman, who was not involved with the study, had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM AAD 2024
SAN DIEGO — , results from an ongoing 10-year observational study showed.
“We need to sort out whether this is reversed by newer treatments, especially in the 6- to 11-year-olds, as well as the factors that underlie it in atopic dermatitis,” said the study’s first author Amy S. Paller, MD, chair of dermatology, Northwestern University, Chicago, Illinois, following the annual meeting of the American Academy of Dermatology, where the study was presented during a poster session.
Atopic Dermatitis Impacts Growth
In the ongoing international study called PEDISTAD, researchers enrolled 1326 children younger than 12 years with moderate to severe atopic dermatitis inadequately controlled by topical therapies who were candidates to receive systemic medications. They assessed the percentage of patients above the 50th percentile and the mean percentiles for height, weight, and body mass index (BMI) at baseline against the Centers for Disease Control and Prevention’s (CDC’s) Learning Management System reference healthy population, by age in months, and compared results to the CDC’s standardized growth curves for healthy children aged 0-12 years.
The investigators found that at baseline, compared with the age-specific population norms, 50% of men and 51% of women in the PEDISTAD study were above the 50th percentile for weight, but only 38% and 52%, respectively, were above the 50th percentile for height. Among patients aged 5-12 years, only 28% of men and 47% of women were above the 50th percentile for height, while 69% of men and 71% of women were above the 50th percentile for BMI.
Dr. Paller said that she was “not really surprised by the reduction in linear growth, since there are so many factors that may contribute,” including chronic inflammation, poor sleep, and the use of topical and systemic steroids. “But [it’s] good to have this data as an opportunity to see if our improved therapies can reverse this.”
She said that she was “a bit surprised by the increase in weight and body mass index, but this could reflect less physical activity/sports [participation and] deserves more investigation,” and added that the findings “mesh nicely with new attention on bone growth with good control of atopic dermatitis in this age group.”
Dr. Paller acknowledged certain limitations of the study, including the fact that those enrolled are a heterogeneous cohort with variable treatment regimens.
Some Answers, More Questions
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, who was asked to comment on the findings, said that atopic dermatitis “should be considered the cutaneous manifestations of a systemic inflammatory disease, though even if it were not, the impact on daily and nightly activities [such as sleep] could indirectly have systemic medical consequences.”
The data presented “highlights that children with moderate to severe disease have higher BMIs and shorter height than matched counterparts, likely owing to the treasure trove of direct and indirect consequences of uncontrolled type 2 inflammation,” he said. “What I would like to know, and as the authors astutely noted, could treatment, and especially early intervention, prevent or even alter this impact?”
Dr. Paller disclosed that she is a consultant for several pharmaceutical companies, including Sanofi and Regeneron, the study sponsor. She is also an investigator for AbbVie, Dermavant, Eli Lilly, Incyte, Janssen, Krystal, LEO Pharma, and UCB and is a member of the data monitoring safety board for AbbVie, Abeona, Catawba, Galderma, and InMed. Dr. Friedman, who was not involved with the study, had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM AAD 2024
SAN DIEGO — , results from an ongoing 10-year observational study showed.
“We need to sort out whether this is reversed by newer treatments, especially in the 6- to 11-year-olds, as well as the factors that underlie it in atopic dermatitis,” said the study’s first author Amy S. Paller, MD, chair of dermatology, Northwestern University, Chicago, Illinois, following the annual meeting of the American Academy of Dermatology, where the study was presented during a poster session.
Atopic Dermatitis Impacts Growth
In the ongoing international study called PEDISTAD, researchers enrolled 1326 children younger than 12 years with moderate to severe atopic dermatitis inadequately controlled by topical therapies who were candidates to receive systemic medications. They assessed the percentage of patients above the 50th percentile and the mean percentiles for height, weight, and body mass index (BMI) at baseline against the Centers for Disease Control and Prevention’s (CDC’s) Learning Management System reference healthy population, by age in months, and compared results to the CDC’s standardized growth curves for healthy children aged 0-12 years.
The investigators found that at baseline, compared with the age-specific population norms, 50% of men and 51% of women in the PEDISTAD study were above the 50th percentile for weight, but only 38% and 52%, respectively, were above the 50th percentile for height. Among patients aged 5-12 years, only 28% of men and 47% of women were above the 50th percentile for height, while 69% of men and 71% of women were above the 50th percentile for BMI.
Dr. Paller said that she was “not really surprised by the reduction in linear growth, since there are so many factors that may contribute,” including chronic inflammation, poor sleep, and the use of topical and systemic steroids. “But [it’s] good to have this data as an opportunity to see if our improved therapies can reverse this.”
She said that she was “a bit surprised by the increase in weight and body mass index, but this could reflect less physical activity/sports [participation and] deserves more investigation,” and added that the findings “mesh nicely with new attention on bone growth with good control of atopic dermatitis in this age group.”
Dr. Paller acknowledged certain limitations of the study, including the fact that those enrolled are a heterogeneous cohort with variable treatment regimens.
Some Answers, More Questions
Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, DC, who was asked to comment on the findings, said that atopic dermatitis “should be considered the cutaneous manifestations of a systemic inflammatory disease, though even if it were not, the impact on daily and nightly activities [such as sleep] could indirectly have systemic medical consequences.”
The data presented “highlights that children with moderate to severe disease have higher BMIs and shorter height than matched counterparts, likely owing to the treasure trove of direct and indirect consequences of uncontrolled type 2 inflammation,” he said. “What I would like to know, and as the authors astutely noted, could treatment, and especially early intervention, prevent or even alter this impact?”
Dr. Paller disclosed that she is a consultant for several pharmaceutical companies, including Sanofi and Regeneron, the study sponsor. She is also an investigator for AbbVie, Dermavant, Eli Lilly, Incyte, Janssen, Krystal, LEO Pharma, and UCB and is a member of the data monitoring safety board for AbbVie, Abeona, Catawba, Galderma, and InMed. Dr. Friedman, who was not involved with the study, had no relevant disclosures.
A version of this article appeared on Medscape.com.
Childhood Loneliness Predictive of Subsequent Psychosis?
BUDAPEST, HUNGARY — Self-perceived loneliness during childhood is linked to a more than twofold increased risk for subsequent first-episode psychosis (FEP) — new findings that may point to a novel marker for the disorder.
The association between loneliness and FEP “appears to extend beyond the effects of objective social isolation,” said study presenter Covadonga M. Díaz-Caneja, MD, PhD, Institute of Psychiatry and Mental Health, Hospital General Universitario Gregorio Marañón, Madrid, and “is particularly pronounced in females.”
“These findings suggest the potential of childhood loneliness as an early risk marker for psychosis that could help guide targeted interventions,” she added.
The results were presented at the European Psychiatric Association 2024 Congress.
Isolation a Major Risk Factor
There are two components to isolation, both of which are “major risk factors” for morbidity, mortality, and the onset of mental disorders, said Dr. Díaz-Caneja.
The first is “objective social isolation,” which consists of a demonstrable lack of social connections, including social interactions, contacts, and relationships, while the other is a perceived sense of isolation, or “loneliness,” defined as a “subjective feeling of distress associated with a lack of meaningful relationships,” regardless of the amount of actual social contact an individual experiences.
Childhood loneliness occurs before age 12 and is becoming increasingly prevalent, said Dr. Díaz-Caneja. A recent survey shows that approximately one third of children report they often feel lonely.
Genetic and observational research has shown there is a bidirectional relationship between loneliness and psychosis and that patients with schizophrenia are more likely to report loneliness than is the general population.
Dr. Díaz-Caneja noted that there is no previous research that has assessed the potential association between childhood loneliness and subsequent psychosis.
To investigate, the researchers conducted an observational, case-control study in seven university hospitals in Madrid. It included individuals aged 7-40 years, including FEP patients with a psychosis duration of less than 2 years, and healthy controls from the same geographic areas.
They assessed childhood objective social isolation using the Premorbid Adjustment Scale and examined childhood loneliness with the single item: “Have you ever felt lonely for more than 6 months before the age of 12?”
A range of measures and questionnaires were also administered to assess participants’ symptom scores, alongside the Global Assessment of Functioning (GAF).
Alone vs Lonely
Two hundred eighty-five patients with FEP participated in the study. They had a mean age of 24.5 years, and 32.6% were female. The study also included 261 healthy controls (average age, 25.9 years; 48.7% female).
After the researchers adjusted for age, gender, ethnicity, and socioeconomic status, loneliness during childhood was associated with a significantly increased risk for FEP (odds ratio [OR], 2.17; 95% CI, 1.40-3.51), which increased (OR, 2.70; 95% CI, 1.58-4.62) after further adjustment for objective social isolation.
Further analysis revealed that in those who did not have objective social isolation in childhood, loneliness was associated with a significantly increased risk for FEP (OR, 2.68; 95% CI, 1.56-4.60).
However, the relationship between loneliness and FEP was not significant in participants who were objectively socially isolated during childhood (OR, 0.33; 95% CI, 0.08-1.45).
Compared with males, females reporting loneliness had a markedly increased risk for FEP (OR, 4.74; 95% CI, 2.23-10.05 vs OR, 1.17; 95% CI, 0.63-2.19).
However, females had a reduced risk of receiving a diagnosis of schizophrenia spectrum disorder (OR, 0.155; 95% CI, 0.048-0.506), indicating that loneliness influenced the type of diagnosis, she noted.
There was a significant positive relationship between loneliness in childhood and symptom scores in men, and a negative association with GAF scores in men.
Dr. Díaz-Caneja noted that the study is preliminary and a “work in progress.” The investigators plan to increase the sample size and will conduct more complex analyses, she said.
“We also of course have to bear in mind that it is a cross-sectional study and that there may be some kind of recall biases [because] we are asking patients now about what happened in the past.”
She noted that it’s unclear whether the results can be extrapolated to individuals who are currently experiencing loneliness because “the determinants of loneliness 10 years ago or 15 years ago may be different.”
How, When to Intervene
Session chair Judit Lazáry, MD, PhD, Department of Clinical and Theoretical Mental Health, Kútvölgyi Clinical Center, Semmelweis University, Budapest, Hungary, told this news organization that the association between loneliness and FEP was “not surprising.”
She explained there are a lot of data indicating that premorbid symptoms in childhood are “predictive signs for the later onset of psychosis,” and loneliness may be “a part of that.”
Individuals experiencing loneliness are more anxious and have difficulties in cultivating and maintaining relationships. In addition, they tend to socially isolate, she said.
The key question, said Dr. Lazáry, is: “How can we intervene to prevent the onset of psychosis? What is the point at which we can support the young person?”
This is challenging, she added, because while “you can detect that a kid is always alone, you cannot detect the feeling of loneliness,” and children can’t always easily express themselves.
Another potential confounder is that in adults with current psychosis, the self-perception that they were lonely during childhood may be a consequence of the disorder.
In addition, she said, individuals with psychosis often experience cognitive impairment, which could affect memory reliability.
Nevertheless, said Dr. Lazáry, the study’s findings suggest that a young person reporting loneliness in childhood may be “another symptom that we have to investigate.”
No funding was declared.
Dr. Díaz-Caneja declared a relationship with Angelini, Janssen, and Viatris and grant support from Instituto de Salud Carlos III, the Spanish Ministry of Science and Innovation, and the European Commission.
A version of this article appeared on Medscape.com.
BUDAPEST, HUNGARY — Self-perceived loneliness during childhood is linked to a more than twofold increased risk for subsequent first-episode psychosis (FEP) — new findings that may point to a novel marker for the disorder.
The association between loneliness and FEP “appears to extend beyond the effects of objective social isolation,” said study presenter Covadonga M. Díaz-Caneja, MD, PhD, Institute of Psychiatry and Mental Health, Hospital General Universitario Gregorio Marañón, Madrid, and “is particularly pronounced in females.”
“These findings suggest the potential of childhood loneliness as an early risk marker for psychosis that could help guide targeted interventions,” she added.
The results were presented at the European Psychiatric Association 2024 Congress.
Isolation a Major Risk Factor
There are two components to isolation, both of which are “major risk factors” for morbidity, mortality, and the onset of mental disorders, said Dr. Díaz-Caneja.
The first is “objective social isolation,” which consists of a demonstrable lack of social connections, including social interactions, contacts, and relationships, while the other is a perceived sense of isolation, or “loneliness,” defined as a “subjective feeling of distress associated with a lack of meaningful relationships,” regardless of the amount of actual social contact an individual experiences.
Childhood loneliness occurs before age 12 and is becoming increasingly prevalent, said Dr. Díaz-Caneja. A recent survey shows that approximately one third of children report they often feel lonely.
Genetic and observational research has shown there is a bidirectional relationship between loneliness and psychosis and that patients with schizophrenia are more likely to report loneliness than is the general population.
Dr. Díaz-Caneja noted that there is no previous research that has assessed the potential association between childhood loneliness and subsequent psychosis.
To investigate, the researchers conducted an observational, case-control study in seven university hospitals in Madrid. It included individuals aged 7-40 years, including FEP patients with a psychosis duration of less than 2 years, and healthy controls from the same geographic areas.
They assessed childhood objective social isolation using the Premorbid Adjustment Scale and examined childhood loneliness with the single item: “Have you ever felt lonely for more than 6 months before the age of 12?”
A range of measures and questionnaires were also administered to assess participants’ symptom scores, alongside the Global Assessment of Functioning (GAF).
Alone vs Lonely
Two hundred eighty-five patients with FEP participated in the study. They had a mean age of 24.5 years, and 32.6% were female. The study also included 261 healthy controls (average age, 25.9 years; 48.7% female).
After the researchers adjusted for age, gender, ethnicity, and socioeconomic status, loneliness during childhood was associated with a significantly increased risk for FEP (odds ratio [OR], 2.17; 95% CI, 1.40-3.51), which increased (OR, 2.70; 95% CI, 1.58-4.62) after further adjustment for objective social isolation.
Further analysis revealed that in those who did not have objective social isolation in childhood, loneliness was associated with a significantly increased risk for FEP (OR, 2.68; 95% CI, 1.56-4.60).
However, the relationship between loneliness and FEP was not significant in participants who were objectively socially isolated during childhood (OR, 0.33; 95% CI, 0.08-1.45).
Compared with males, females reporting loneliness had a markedly increased risk for FEP (OR, 4.74; 95% CI, 2.23-10.05 vs OR, 1.17; 95% CI, 0.63-2.19).
However, females had a reduced risk of receiving a diagnosis of schizophrenia spectrum disorder (OR, 0.155; 95% CI, 0.048-0.506), indicating that loneliness influenced the type of diagnosis, she noted.
There was a significant positive relationship between loneliness in childhood and symptom scores in men, and a negative association with GAF scores in men.
Dr. Díaz-Caneja noted that the study is preliminary and a “work in progress.” The investigators plan to increase the sample size and will conduct more complex analyses, she said.
“We also of course have to bear in mind that it is a cross-sectional study and that there may be some kind of recall biases [because] we are asking patients now about what happened in the past.”
She noted that it’s unclear whether the results can be extrapolated to individuals who are currently experiencing loneliness because “the determinants of loneliness 10 years ago or 15 years ago may be different.”
How, When to Intervene
Session chair Judit Lazáry, MD, PhD, Department of Clinical and Theoretical Mental Health, Kútvölgyi Clinical Center, Semmelweis University, Budapest, Hungary, told this news organization that the association between loneliness and FEP was “not surprising.”
She explained there are a lot of data indicating that premorbid symptoms in childhood are “predictive signs for the later onset of psychosis,” and loneliness may be “a part of that.”
Individuals experiencing loneliness are more anxious and have difficulties in cultivating and maintaining relationships. In addition, they tend to socially isolate, she said.
The key question, said Dr. Lazáry, is: “How can we intervene to prevent the onset of psychosis? What is the point at which we can support the young person?”
This is challenging, she added, because while “you can detect that a kid is always alone, you cannot detect the feeling of loneliness,” and children can’t always easily express themselves.
Another potential confounder is that in adults with current psychosis, the self-perception that they were lonely during childhood may be a consequence of the disorder.
In addition, she said, individuals with psychosis often experience cognitive impairment, which could affect memory reliability.
Nevertheless, said Dr. Lazáry, the study’s findings suggest that a young person reporting loneliness in childhood may be “another symptom that we have to investigate.”
No funding was declared.
Dr. Díaz-Caneja declared a relationship with Angelini, Janssen, and Viatris and grant support from Instituto de Salud Carlos III, the Spanish Ministry of Science and Innovation, and the European Commission.
A version of this article appeared on Medscape.com.
BUDAPEST, HUNGARY — Self-perceived loneliness during childhood is linked to a more than twofold increased risk for subsequent first-episode psychosis (FEP) — new findings that may point to a novel marker for the disorder.
The association between loneliness and FEP “appears to extend beyond the effects of objective social isolation,” said study presenter Covadonga M. Díaz-Caneja, MD, PhD, Institute of Psychiatry and Mental Health, Hospital General Universitario Gregorio Marañón, Madrid, and “is particularly pronounced in females.”
“These findings suggest the potential of childhood loneliness as an early risk marker for psychosis that could help guide targeted interventions,” she added.
The results were presented at the European Psychiatric Association 2024 Congress.
Isolation a Major Risk Factor
There are two components to isolation, both of which are “major risk factors” for morbidity, mortality, and the onset of mental disorders, said Dr. Díaz-Caneja.
The first is “objective social isolation,” which consists of a demonstrable lack of social connections, including social interactions, contacts, and relationships, while the other is a perceived sense of isolation, or “loneliness,” defined as a “subjective feeling of distress associated with a lack of meaningful relationships,” regardless of the amount of actual social contact an individual experiences.
Childhood loneliness occurs before age 12 and is becoming increasingly prevalent, said Dr. Díaz-Caneja. A recent survey shows that approximately one third of children report they often feel lonely.
Genetic and observational research has shown there is a bidirectional relationship between loneliness and psychosis and that patients with schizophrenia are more likely to report loneliness than is the general population.
Dr. Díaz-Caneja noted that there is no previous research that has assessed the potential association between childhood loneliness and subsequent psychosis.
To investigate, the researchers conducted an observational, case-control study in seven university hospitals in Madrid. It included individuals aged 7-40 years, including FEP patients with a psychosis duration of less than 2 years, and healthy controls from the same geographic areas.
They assessed childhood objective social isolation using the Premorbid Adjustment Scale and examined childhood loneliness with the single item: “Have you ever felt lonely for more than 6 months before the age of 12?”
A range of measures and questionnaires were also administered to assess participants’ symptom scores, alongside the Global Assessment of Functioning (GAF).
Alone vs Lonely
Two hundred eighty-five patients with FEP participated in the study. They had a mean age of 24.5 years, and 32.6% were female. The study also included 261 healthy controls (average age, 25.9 years; 48.7% female).
After the researchers adjusted for age, gender, ethnicity, and socioeconomic status, loneliness during childhood was associated with a significantly increased risk for FEP (odds ratio [OR], 2.17; 95% CI, 1.40-3.51), which increased (OR, 2.70; 95% CI, 1.58-4.62) after further adjustment for objective social isolation.
Further analysis revealed that in those who did not have objective social isolation in childhood, loneliness was associated with a significantly increased risk for FEP (OR, 2.68; 95% CI, 1.56-4.60).
However, the relationship between loneliness and FEP was not significant in participants who were objectively socially isolated during childhood (OR, 0.33; 95% CI, 0.08-1.45).
Compared with males, females reporting loneliness had a markedly increased risk for FEP (OR, 4.74; 95% CI, 2.23-10.05 vs OR, 1.17; 95% CI, 0.63-2.19).
However, females had a reduced risk of receiving a diagnosis of schizophrenia spectrum disorder (OR, 0.155; 95% CI, 0.048-0.506), indicating that loneliness influenced the type of diagnosis, she noted.
There was a significant positive relationship between loneliness in childhood and symptom scores in men, and a negative association with GAF scores in men.
Dr. Díaz-Caneja noted that the study is preliminary and a “work in progress.” The investigators plan to increase the sample size and will conduct more complex analyses, she said.
“We also of course have to bear in mind that it is a cross-sectional study and that there may be some kind of recall biases [because] we are asking patients now about what happened in the past.”
She noted that it’s unclear whether the results can be extrapolated to individuals who are currently experiencing loneliness because “the determinants of loneliness 10 years ago or 15 years ago may be different.”
How, When to Intervene
Session chair Judit Lazáry, MD, PhD, Department of Clinical and Theoretical Mental Health, Kútvölgyi Clinical Center, Semmelweis University, Budapest, Hungary, told this news organization that the association between loneliness and FEP was “not surprising.”
She explained there are a lot of data indicating that premorbid symptoms in childhood are “predictive signs for the later onset of psychosis,” and loneliness may be “a part of that.”
Individuals experiencing loneliness are more anxious and have difficulties in cultivating and maintaining relationships. In addition, they tend to socially isolate, she said.
The key question, said Dr. Lazáry, is: “How can we intervene to prevent the onset of psychosis? What is the point at which we can support the young person?”
This is challenging, she added, because while “you can detect that a kid is always alone, you cannot detect the feeling of loneliness,” and children can’t always easily express themselves.
Another potential confounder is that in adults with current psychosis, the self-perception that they were lonely during childhood may be a consequence of the disorder.
In addition, she said, individuals with psychosis often experience cognitive impairment, which could affect memory reliability.
Nevertheless, said Dr. Lazáry, the study’s findings suggest that a young person reporting loneliness in childhood may be “another symptom that we have to investigate.”
No funding was declared.
Dr. Díaz-Caneja declared a relationship with Angelini, Janssen, and Viatris and grant support from Instituto de Salud Carlos III, the Spanish Ministry of Science and Innovation, and the European Commission.
A version of this article appeared on Medscape.com.
First Consensus Statement on Improving Healthcare for Children with Neurodevelopmental Disabilities
The statement was published in Pediatrics.
The disparities in healthcare culture, mindset, and practice often start in childhood for young people with conditions including autism spectrum disorder (ASD), intellectual disability, and attention-deficit/hyperactivity disorder (ADHD), wrote co–first authors Carol Weitzman, MD, co-director of the Autism Spectrum Center at Boston Children’s Hospital, Boston, Massachusetts, and Cy Nadler, PhD, section chief of Autism Psychology at Children’s Mercy in Kansas City, Missouri, and colleagues.
Without better access to safe and appropriate care, people with NDDs experience more seclusion, accidents, restraints, and injury in healthcare encounters, the researchers wrote.
‘Accessible, Humane, Effective Care’
“At the heart of this consensus statement is an affirmation that all people are entitled to healthcare that is accessible, humane, and effective,” they wrote.
The consensus statement was developed as part of the Supporting Access for Everyone (SAFE) Initiative, launched by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. The consensus panel comprised professionals, caregivers, and adults with NDDs. After a 2-day public forum, the consensus panel held a conference and developed a statement on SAFE care, an NDD Health Care Bill of Rights and Transition Considerations. They developed 10 statements across five domains: training; communication; access and planning; diversity, equity, inclusion, belonging, and anti-ableism; and policy and structural change.
Asking the Patient ‘What do You Need?’
One theme in the statement that may have the most impact is “the importance of asking the person in front of you what they need,” and building a care plan around that, said senior author Marilyn Augustyn, MD, Director of the Division of Developmental and Behavioral Pediatrics at Boston University Chobanian & Avedisian School of Medicine, Boston, Massachusetts. “The medical community hasn’t done that very well for individuals with neurodevelopmental disabilities.”
Dr. Weitzman added: “Traditionally in healthcare settings, we’ve asked people to check their disabilities at the door.” Many people with neurodevelopmental disabilities often have “invisible disabilities,” she said, explaining that patients may have accommodation needs that aren’t immediately obvious, but could improve their access to care, so asking them what they need is critical.
Examples of ‘Ableism’
The consensus statement also calls attention to structural “ableism” or policies or practices that favor able-bodied people over those with disabilities and details the need for more training and changed policies.
The paper gives some examples of ableism, such as inappropriately excluding people with NDDs from research; staff assuming nonspeaking patients have no capacity for communication; or lack of awareness of sensory needs before using cold stethoscopes or flashing direct light into eyes.
Dr. Weitzman says this work is just the beginning of a complex process. It is intended to be the driver for developing curriculum to train all clinicians and others working with patients about neurodevelopmental disabilities. The hope is it will lead to more research to formalize best practices and make policies mandatory rather than optional.
The urgency in highlighting these issues is partly related to the prevalence of children and adolescents with neurodevelopmental disabilities, which the paper states is approximately 1 in 6.
But there are personal reasons as well for the team who developed the statement.
“We just believe that it is just a human right,” Dr. Weitzman said. “Having a neurodevelopmental disability does not make you any less entitled to good care. “
Dr. Augustyn added, “The children I’ve had the honor of caring for for the last 30 years deserve all this care and more. I think it’s time.”
This work was supported by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. Dr. Weitzman is a past consultant for Helios/Meliora. The other authors report no relevant financial relationships.
The statement was published in Pediatrics.
The disparities in healthcare culture, mindset, and practice often start in childhood for young people with conditions including autism spectrum disorder (ASD), intellectual disability, and attention-deficit/hyperactivity disorder (ADHD), wrote co–first authors Carol Weitzman, MD, co-director of the Autism Spectrum Center at Boston Children’s Hospital, Boston, Massachusetts, and Cy Nadler, PhD, section chief of Autism Psychology at Children’s Mercy in Kansas City, Missouri, and colleagues.
Without better access to safe and appropriate care, people with NDDs experience more seclusion, accidents, restraints, and injury in healthcare encounters, the researchers wrote.
‘Accessible, Humane, Effective Care’
“At the heart of this consensus statement is an affirmation that all people are entitled to healthcare that is accessible, humane, and effective,” they wrote.
The consensus statement was developed as part of the Supporting Access for Everyone (SAFE) Initiative, launched by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. The consensus panel comprised professionals, caregivers, and adults with NDDs. After a 2-day public forum, the consensus panel held a conference and developed a statement on SAFE care, an NDD Health Care Bill of Rights and Transition Considerations. They developed 10 statements across five domains: training; communication; access and planning; diversity, equity, inclusion, belonging, and anti-ableism; and policy and structural change.
Asking the Patient ‘What do You Need?’
One theme in the statement that may have the most impact is “the importance of asking the person in front of you what they need,” and building a care plan around that, said senior author Marilyn Augustyn, MD, Director of the Division of Developmental and Behavioral Pediatrics at Boston University Chobanian & Avedisian School of Medicine, Boston, Massachusetts. “The medical community hasn’t done that very well for individuals with neurodevelopmental disabilities.”
Dr. Weitzman added: “Traditionally in healthcare settings, we’ve asked people to check their disabilities at the door.” Many people with neurodevelopmental disabilities often have “invisible disabilities,” she said, explaining that patients may have accommodation needs that aren’t immediately obvious, but could improve their access to care, so asking them what they need is critical.
Examples of ‘Ableism’
The consensus statement also calls attention to structural “ableism” or policies or practices that favor able-bodied people over those with disabilities and details the need for more training and changed policies.
The paper gives some examples of ableism, such as inappropriately excluding people with NDDs from research; staff assuming nonspeaking patients have no capacity for communication; or lack of awareness of sensory needs before using cold stethoscopes or flashing direct light into eyes.
Dr. Weitzman says this work is just the beginning of a complex process. It is intended to be the driver for developing curriculum to train all clinicians and others working with patients about neurodevelopmental disabilities. The hope is it will lead to more research to formalize best practices and make policies mandatory rather than optional.
The urgency in highlighting these issues is partly related to the prevalence of children and adolescents with neurodevelopmental disabilities, which the paper states is approximately 1 in 6.
But there are personal reasons as well for the team who developed the statement.
“We just believe that it is just a human right,” Dr. Weitzman said. “Having a neurodevelopmental disability does not make you any less entitled to good care. “
Dr. Augustyn added, “The children I’ve had the honor of caring for for the last 30 years deserve all this care and more. I think it’s time.”
This work was supported by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. Dr. Weitzman is a past consultant for Helios/Meliora. The other authors report no relevant financial relationships.
The statement was published in Pediatrics.
The disparities in healthcare culture, mindset, and practice often start in childhood for young people with conditions including autism spectrum disorder (ASD), intellectual disability, and attention-deficit/hyperactivity disorder (ADHD), wrote co–first authors Carol Weitzman, MD, co-director of the Autism Spectrum Center at Boston Children’s Hospital, Boston, Massachusetts, and Cy Nadler, PhD, section chief of Autism Psychology at Children’s Mercy in Kansas City, Missouri, and colleagues.
Without better access to safe and appropriate care, people with NDDs experience more seclusion, accidents, restraints, and injury in healthcare encounters, the researchers wrote.
‘Accessible, Humane, Effective Care’
“At the heart of this consensus statement is an affirmation that all people are entitled to healthcare that is accessible, humane, and effective,” they wrote.
The consensus statement was developed as part of the Supporting Access for Everyone (SAFE) Initiative, launched by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. The consensus panel comprised professionals, caregivers, and adults with NDDs. After a 2-day public forum, the consensus panel held a conference and developed a statement on SAFE care, an NDD Health Care Bill of Rights and Transition Considerations. They developed 10 statements across five domains: training; communication; access and planning; diversity, equity, inclusion, belonging, and anti-ableism; and policy and structural change.
Asking the Patient ‘What do You Need?’
One theme in the statement that may have the most impact is “the importance of asking the person in front of you what they need,” and building a care plan around that, said senior author Marilyn Augustyn, MD, Director of the Division of Developmental and Behavioral Pediatrics at Boston University Chobanian & Avedisian School of Medicine, Boston, Massachusetts. “The medical community hasn’t done that very well for individuals with neurodevelopmental disabilities.”
Dr. Weitzman added: “Traditionally in healthcare settings, we’ve asked people to check their disabilities at the door.” Many people with neurodevelopmental disabilities often have “invisible disabilities,” she said, explaining that patients may have accommodation needs that aren’t immediately obvious, but could improve their access to care, so asking them what they need is critical.
Examples of ‘Ableism’
The consensus statement also calls attention to structural “ableism” or policies or practices that favor able-bodied people over those with disabilities and details the need for more training and changed policies.
The paper gives some examples of ableism, such as inappropriately excluding people with NDDs from research; staff assuming nonspeaking patients have no capacity for communication; or lack of awareness of sensory needs before using cold stethoscopes or flashing direct light into eyes.
Dr. Weitzman says this work is just the beginning of a complex process. It is intended to be the driver for developing curriculum to train all clinicians and others working with patients about neurodevelopmental disabilities. The hope is it will lead to more research to formalize best practices and make policies mandatory rather than optional.
The urgency in highlighting these issues is partly related to the prevalence of children and adolescents with neurodevelopmental disabilities, which the paper states is approximately 1 in 6.
But there are personal reasons as well for the team who developed the statement.
“We just believe that it is just a human right,” Dr. Weitzman said. “Having a neurodevelopmental disability does not make you any less entitled to good care. “
Dr. Augustyn added, “The children I’ve had the honor of caring for for the last 30 years deserve all this care and more. I think it’s time.”
This work was supported by the Developmental Behavioral Pediatric Research Network and the Association of University Centers on Disability. Dr. Weitzman is a past consultant for Helios/Meliora. The other authors report no relevant financial relationships.
Combined Pediatric Derm-Rheum Clinics Supported by Survey Respondents
TOPLINE:
.
METHODOLOGY:
- Combined pediatric dermatology-rheumatology clinics can improve patient outcomes and experiences, particularly for pediatric autoimmune conditions presenting with both cutaneous and systemic manifestations.
- The researchers surveyed 208 pediatric dermatologists working in combined pediatric dermatology-rheumatology clinics.
- A total of 13 member responses were recorded from three countries: 10 from the United States, two from Mexico, and one from Canada.
TAKEAWAY:
- Perceived benefits of combined clinics were improved patient care through coordinated treatment decisions and timely communication between providers.
- Patient satisfaction was favorable, and patients and families endorsed the combined clinic approach.
- Barriers to clinic establishment included differences in the pace between dermatology and rheumatology clinic flow, the need to generate more relative value units, resistance from colleagues, and limited time.
- Areas that needed improvement included more time for patient visits, dedicated research assistants, new patient referrals, additional patient rooms, resources for research, and patient care infrastructure.
IN PRACTICE:
The insights from this survey “will hopefully inspire further development of these combined clinics,” the authors wrote.
SOURCE:
The investigation, led by Olga S. Cherepakhin, BS, University of Washington, Seattle, Washington, was published in Pediatric Dermatology.
LIMITATIONS:
Limitations included the subjective nature, lack of some information, selection bias, and small number of respondents, and the survey reflected the perspective of the pediatric dermatologists only.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported full-time employment at Janssen R&D, and the other authors had no disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Combined pediatric dermatology-rheumatology clinics can improve patient outcomes and experiences, particularly for pediatric autoimmune conditions presenting with both cutaneous and systemic manifestations.
- The researchers surveyed 208 pediatric dermatologists working in combined pediatric dermatology-rheumatology clinics.
- A total of 13 member responses were recorded from three countries: 10 from the United States, two from Mexico, and one from Canada.
TAKEAWAY:
- Perceived benefits of combined clinics were improved patient care through coordinated treatment decisions and timely communication between providers.
- Patient satisfaction was favorable, and patients and families endorsed the combined clinic approach.
- Barriers to clinic establishment included differences in the pace between dermatology and rheumatology clinic flow, the need to generate more relative value units, resistance from colleagues, and limited time.
- Areas that needed improvement included more time for patient visits, dedicated research assistants, new patient referrals, additional patient rooms, resources for research, and patient care infrastructure.
IN PRACTICE:
The insights from this survey “will hopefully inspire further development of these combined clinics,” the authors wrote.
SOURCE:
The investigation, led by Olga S. Cherepakhin, BS, University of Washington, Seattle, Washington, was published in Pediatric Dermatology.
LIMITATIONS:
Limitations included the subjective nature, lack of some information, selection bias, and small number of respondents, and the survey reflected the perspective of the pediatric dermatologists only.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported full-time employment at Janssen R&D, and the other authors had no disclosures.
A version of this article appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- Combined pediatric dermatology-rheumatology clinics can improve patient outcomes and experiences, particularly for pediatric autoimmune conditions presenting with both cutaneous and systemic manifestations.
- The researchers surveyed 208 pediatric dermatologists working in combined pediatric dermatology-rheumatology clinics.
- A total of 13 member responses were recorded from three countries: 10 from the United States, two from Mexico, and one from Canada.
TAKEAWAY:
- Perceived benefits of combined clinics were improved patient care through coordinated treatment decisions and timely communication between providers.
- Patient satisfaction was favorable, and patients and families endorsed the combined clinic approach.
- Barriers to clinic establishment included differences in the pace between dermatology and rheumatology clinic flow, the need to generate more relative value units, resistance from colleagues, and limited time.
- Areas that needed improvement included more time for patient visits, dedicated research assistants, new patient referrals, additional patient rooms, resources for research, and patient care infrastructure.
IN PRACTICE:
The insights from this survey “will hopefully inspire further development of these combined clinics,” the authors wrote.
SOURCE:
The investigation, led by Olga S. Cherepakhin, BS, University of Washington, Seattle, Washington, was published in Pediatric Dermatology.
LIMITATIONS:
Limitations included the subjective nature, lack of some information, selection bias, and small number of respondents, and the survey reflected the perspective of the pediatric dermatologists only.
DISCLOSURES:
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. One author reported full-time employment at Janssen R&D, and the other authors had no disclosures.
A version of this article appeared on Medscape.com.