User login
For MD-IQ only
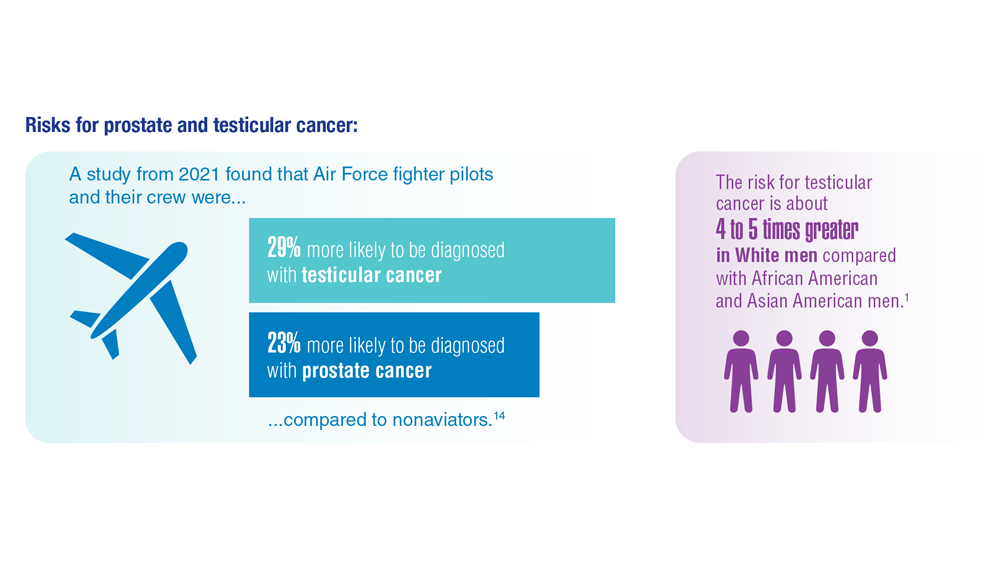
Risky drinking common in cancer survivors
An analysis of more than 15,000 adults with a cancer diagnosis revealed that nearly 80% were current drinkers. Among current drinkers, 13% consumed a moderate amount of alcohol in a typical day, while close to 40% engaged in hazardous drinking.
The numbers are “staggering,” Yin Cao, ScD, MPH, of Washington University in St. Louis, said in an interview. “Most concerning is that those on cancer treatment are engaged in a similar level of risky drinking.”
The study was published online in JAMA Network Open.
Drinking alcohol can increase a person’s risk for a variety of cancers, including oral and pharyngeal cancer as well as esophageal, colorectal, liver, and female breast cancers.
Consuming alcohol is also associated with numerous risks among people diagnosed with cancer. In the short term, alcohol consumption can worsen postsurgical outcomes as well as impair cognition and amplify cardiotoxicity in patients undergoing chemotherapy. In the long term, drinking alcohol can elevate a person’s risk of recurrence, secondary tumors, and mortality.
The American Society of Clinical Oncology recently issued a statement reinforcing the need to prioritize alcohol consumption as a key modifiable behavioral factor in the cancer control research agenda.
The current American Cancer Society guidelines indicate that it’s best to avoid or, at least, minimize alcohol consumption. Men should limit their intake to no more than two drinks per day and women should have no more than one drink per day.
Despite this data and guidelines, alcohol drinking patterns among cancer survivors in the United States remain poorly understood.
To explore further, the researchers identified 15,199 adult cancer survivors enrolled in the National Institutes of Health’s All of Us Research Program.
Overall, 78% of the cohort – more than 11,800 individuals – were current drinkers. In a typical day, 24% engaged in binge drinking – consuming six or more drinks on a single occasion – and 38% engaged in hazardous drinking. Using the Alcohol Use Disorders Identification Test–Consumption, the researchers classified hazardous drinking as scores of 4 or higher in men and 3 or higher in women.
Drinking patterns looked similar in the subset of 1,839 patients undergoing cancer treatment. In this group, 76% were current drinkers. Among current drinkers, 12% exceeded moderate drinking levels, 23% reported binge drinking, and 38% engaged in hazardous drinking. In this group, men, Hispanics, people diagnosed with cancer before age 18, and smokers were more likely to engage in risky drinking behaviors.
“We know that many people who are diagnosed with cancer continue to drink alcohol, but this study provides much more detailed information about that,” said Farhad Islami, MD, PhD, senior scientific director for cancer disparity research at the American Cancer Society, Atlanta, who was not involved in the study.
Given the degree of drinking identified in this population, Dr. Cao highlighted the importance of talking to patients about alcohol.
“Our findings highlight an opportunity for enhanced support and intervention concerning risky drinking behaviors” in oncology, Dr. Cao said. “Given the societal norms surrounding alcohol and the general lack of awareness of alcohol’s short- and long-term impact on cancer outcomes, gently educating patients/survivors about potential risks while understanding the cultural and societal contexts of drinking can make a difference.”
Dr. Islami agreed that oncologists should talk to their patients about alcohol, “especially those going through active treatment because alcohol may affect the treatment or may be associated with more complications of the treatment.”
“Many people now know that smoking causes cancer, but unfortunately, many people do not know about the association of alcohol with cancer,” he said.
Outside of an awareness gap, there are numerous risk factors for substance abuse among cancer survivors, Marleen Meyers, MD, director of the cancer survivorship program at NYU Langone Perlmutter Cancer Center, New York, explained.
Alcohol can help some cancer survivors dull feelings of isolation, fear, stress, and poor pain management that may accompany their diagnosis and treatment, said Dr. Meyers, who was not involved in the research. That is why “it is important for patients to be honest with their providers and for providers to ask about substance use in a nonjudgmental way.”
In these conversations, oncologists should educate patients about the safety risks associated with alcohol intake during or after treatment and that there is no established “safe” amount of alcohol. Incorporating a mental health screening and questions about a family history of substance abuse can also help identify patients “most at risk so providers can be proactive,” she said.
The study was supported by a grant from the NIH. Dr. Cao, Dr. Islami, and Dr. Meyers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An analysis of more than 15,000 adults with a cancer diagnosis revealed that nearly 80% were current drinkers. Among current drinkers, 13% consumed a moderate amount of alcohol in a typical day, while close to 40% engaged in hazardous drinking.
The numbers are “staggering,” Yin Cao, ScD, MPH, of Washington University in St. Louis, said in an interview. “Most concerning is that those on cancer treatment are engaged in a similar level of risky drinking.”
The study was published online in JAMA Network Open.
Drinking alcohol can increase a person’s risk for a variety of cancers, including oral and pharyngeal cancer as well as esophageal, colorectal, liver, and female breast cancers.
Consuming alcohol is also associated with numerous risks among people diagnosed with cancer. In the short term, alcohol consumption can worsen postsurgical outcomes as well as impair cognition and amplify cardiotoxicity in patients undergoing chemotherapy. In the long term, drinking alcohol can elevate a person’s risk of recurrence, secondary tumors, and mortality.
The American Society of Clinical Oncology recently issued a statement reinforcing the need to prioritize alcohol consumption as a key modifiable behavioral factor in the cancer control research agenda.
The current American Cancer Society guidelines indicate that it’s best to avoid or, at least, minimize alcohol consumption. Men should limit their intake to no more than two drinks per day and women should have no more than one drink per day.
Despite this data and guidelines, alcohol drinking patterns among cancer survivors in the United States remain poorly understood.
To explore further, the researchers identified 15,199 adult cancer survivors enrolled in the National Institutes of Health’s All of Us Research Program.
Overall, 78% of the cohort – more than 11,800 individuals – were current drinkers. In a typical day, 24% engaged in binge drinking – consuming six or more drinks on a single occasion – and 38% engaged in hazardous drinking. Using the Alcohol Use Disorders Identification Test–Consumption, the researchers classified hazardous drinking as scores of 4 or higher in men and 3 or higher in women.
Drinking patterns looked similar in the subset of 1,839 patients undergoing cancer treatment. In this group, 76% were current drinkers. Among current drinkers, 12% exceeded moderate drinking levels, 23% reported binge drinking, and 38% engaged in hazardous drinking. In this group, men, Hispanics, people diagnosed with cancer before age 18, and smokers were more likely to engage in risky drinking behaviors.
“We know that many people who are diagnosed with cancer continue to drink alcohol, but this study provides much more detailed information about that,” said Farhad Islami, MD, PhD, senior scientific director for cancer disparity research at the American Cancer Society, Atlanta, who was not involved in the study.
Given the degree of drinking identified in this population, Dr. Cao highlighted the importance of talking to patients about alcohol.
“Our findings highlight an opportunity for enhanced support and intervention concerning risky drinking behaviors” in oncology, Dr. Cao said. “Given the societal norms surrounding alcohol and the general lack of awareness of alcohol’s short- and long-term impact on cancer outcomes, gently educating patients/survivors about potential risks while understanding the cultural and societal contexts of drinking can make a difference.”
Dr. Islami agreed that oncologists should talk to their patients about alcohol, “especially those going through active treatment because alcohol may affect the treatment or may be associated with more complications of the treatment.”
“Many people now know that smoking causes cancer, but unfortunately, many people do not know about the association of alcohol with cancer,” he said.
Outside of an awareness gap, there are numerous risk factors for substance abuse among cancer survivors, Marleen Meyers, MD, director of the cancer survivorship program at NYU Langone Perlmutter Cancer Center, New York, explained.
Alcohol can help some cancer survivors dull feelings of isolation, fear, stress, and poor pain management that may accompany their diagnosis and treatment, said Dr. Meyers, who was not involved in the research. That is why “it is important for patients to be honest with their providers and for providers to ask about substance use in a nonjudgmental way.”
In these conversations, oncologists should educate patients about the safety risks associated with alcohol intake during or after treatment and that there is no established “safe” amount of alcohol. Incorporating a mental health screening and questions about a family history of substance abuse can also help identify patients “most at risk so providers can be proactive,” she said.
The study was supported by a grant from the NIH. Dr. Cao, Dr. Islami, and Dr. Meyers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An analysis of more than 15,000 adults with a cancer diagnosis revealed that nearly 80% were current drinkers. Among current drinkers, 13% consumed a moderate amount of alcohol in a typical day, while close to 40% engaged in hazardous drinking.
The numbers are “staggering,” Yin Cao, ScD, MPH, of Washington University in St. Louis, said in an interview. “Most concerning is that those on cancer treatment are engaged in a similar level of risky drinking.”
The study was published online in JAMA Network Open.
Drinking alcohol can increase a person’s risk for a variety of cancers, including oral and pharyngeal cancer as well as esophageal, colorectal, liver, and female breast cancers.
Consuming alcohol is also associated with numerous risks among people diagnosed with cancer. In the short term, alcohol consumption can worsen postsurgical outcomes as well as impair cognition and amplify cardiotoxicity in patients undergoing chemotherapy. In the long term, drinking alcohol can elevate a person’s risk of recurrence, secondary tumors, and mortality.
The American Society of Clinical Oncology recently issued a statement reinforcing the need to prioritize alcohol consumption as a key modifiable behavioral factor in the cancer control research agenda.
The current American Cancer Society guidelines indicate that it’s best to avoid or, at least, minimize alcohol consumption. Men should limit their intake to no more than two drinks per day and women should have no more than one drink per day.
Despite this data and guidelines, alcohol drinking patterns among cancer survivors in the United States remain poorly understood.
To explore further, the researchers identified 15,199 adult cancer survivors enrolled in the National Institutes of Health’s All of Us Research Program.
Overall, 78% of the cohort – more than 11,800 individuals – were current drinkers. In a typical day, 24% engaged in binge drinking – consuming six or more drinks on a single occasion – and 38% engaged in hazardous drinking. Using the Alcohol Use Disorders Identification Test–Consumption, the researchers classified hazardous drinking as scores of 4 or higher in men and 3 or higher in women.
Drinking patterns looked similar in the subset of 1,839 patients undergoing cancer treatment. In this group, 76% were current drinkers. Among current drinkers, 12% exceeded moderate drinking levels, 23% reported binge drinking, and 38% engaged in hazardous drinking. In this group, men, Hispanics, people diagnosed with cancer before age 18, and smokers were more likely to engage in risky drinking behaviors.
“We know that many people who are diagnosed with cancer continue to drink alcohol, but this study provides much more detailed information about that,” said Farhad Islami, MD, PhD, senior scientific director for cancer disparity research at the American Cancer Society, Atlanta, who was not involved in the study.
Given the degree of drinking identified in this population, Dr. Cao highlighted the importance of talking to patients about alcohol.
“Our findings highlight an opportunity for enhanced support and intervention concerning risky drinking behaviors” in oncology, Dr. Cao said. “Given the societal norms surrounding alcohol and the general lack of awareness of alcohol’s short- and long-term impact on cancer outcomes, gently educating patients/survivors about potential risks while understanding the cultural and societal contexts of drinking can make a difference.”
Dr. Islami agreed that oncologists should talk to their patients about alcohol, “especially those going through active treatment because alcohol may affect the treatment or may be associated with more complications of the treatment.”
“Many people now know that smoking causes cancer, but unfortunately, many people do not know about the association of alcohol with cancer,” he said.
Outside of an awareness gap, there are numerous risk factors for substance abuse among cancer survivors, Marleen Meyers, MD, director of the cancer survivorship program at NYU Langone Perlmutter Cancer Center, New York, explained.
Alcohol can help some cancer survivors dull feelings of isolation, fear, stress, and poor pain management that may accompany their diagnosis and treatment, said Dr. Meyers, who was not involved in the research. That is why “it is important for patients to be honest with their providers and for providers to ask about substance use in a nonjudgmental way.”
In these conversations, oncologists should educate patients about the safety risks associated with alcohol intake during or after treatment and that there is no established “safe” amount of alcohol. Incorporating a mental health screening and questions about a family history of substance abuse can also help identify patients “most at risk so providers can be proactive,” she said.
The study was supported by a grant from the NIH. Dr. Cao, Dr. Islami, and Dr. Meyers report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Race and Age-Related PSA Testing Disparities in Spinal Cord Injured Men: Analysis of National Veterans Health Administration Data
Prostate cancer will be diagnosed in 12.5% of men during their lifetime. It is the most commonly diagnosed solid organ cancer in men.1 However, prostate cancer screening for prostate-specific antigen (PSA) remains controversial due to concerns about overdiagnosis, as the overall risk of dying of prostate cancer is only 2.4%.1
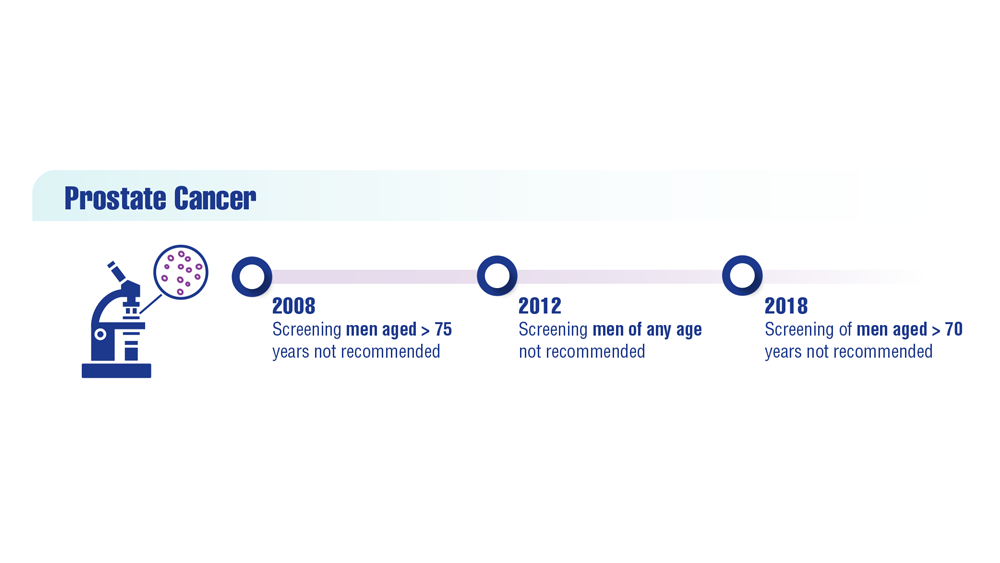
To address the risk and benefits of PSA testing, in 2012 the US Preventive Services Task Force (USPSTF) recommended against routine PSA testing.2 Updated 2018 recommendations continued this recommendation in men aged > 70 years but acknowledged a small potential benefit in men aged 55 to 69 years and suggested individualized shared decision making between patient and clinician.3 In addition, American Urological Association (AUA) guidelines for the early detection of prostate cancer recommend against PSA screening in men aged < 40 years or those aged > 70 years, shared decision making for individuals aged 55 to 70 years or in high-risk men aged 40 to 55 years (ie, family history of prostate cancer or African American race).4 PSA screening is not recommended for men with a life expectancy shorter than 10 to 15 years aged > 70 years.4
The Veterans Health Administration (VHA) is the largest integrated health care system in the US.5 In addition, the US Department of Veterans Affairs (VA) Spinal Cord Injury and Disorders System of Care operates 25 centers throughout the US.6 Life expectancy following spinal cord injury (SCI) increased significantly through the 1980s but has since plateaued, with life expectancy being impacted by age at injury, completeness of injury, and neurologic level.7,8 As part of a program of uniform care, all persons with SCI followed at the Spinal Cord Injury and Disorders System of Care centers are offered comprehensive annual evaluations, including screening laboratory tests, such as PSA level.9
Patients with SCI present a unique challenge when interpreting PSA levels, given potentially confounding factors, including neurogenic bladder management, high rates of bacteriuria, urinary tract infections (UTIs), testosterone deficiency, and pelvic innervation that differs from the noninjured population.10,11 Unfortunately, the literature on prostate cancer prevalence and average PSA levels in patients with SCI is limited by the small scope of studies and inconsistent data.10-16 Therefore, the purpose of the current investigation was to quantify and analyze the rates of annual PSA testing for all men with SCI in the VHA.
Methods
Approval was granted by the Richmond VA Medical Center (VAMC) Institutional Review Board in Virginia, and by the VA Informatics and Computing Infrastructure (VINCI) data access request tracker system for extraction of data from the VA Corporate Data Warehouse. Microsoft Structured Query Language was used for data programming and query design. Statistical analysis was conducted using Stata version 15.1 with assistance from professional biostatisticians.
Only male veterans with a nervous system disorder affecting the spinal cord or with myelopathy were included, based on International Classification of Diseases (ICD) version 9 and 10 codes, corresponding to traumatic and nontraumatic myelopathy. Veterans diagnosed with myelopathy based on ICD codes corresponding to progressive or degenerative myelopathies, such as multiple sclerosis or amyotrophic lateral sclerosis, were excluded.
For each veteran, extracted data included the unique identification number, date of birth, ICD code, date ICD code first appeared, race, gender, death status (yes/no), date of death (when applicable), date of each PSA test, PSA test values, and the VAMC where each test was performed. Only tests for total PSA were included. The date that the ICD code first appeared served as an approximation for the date of SCI. The time frame for the study included all PSA tests in the VINCI database for 2000 through 2017. However, only post-SCI PSA tests were included in the analysis. Duplicate tests (same date/time) were eliminated.
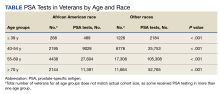
Race is considered a risk factor for prostate cancer only for African American patients, likely due to racial health disparities.17 Given this, we chose to categorize race as either African American or other, with a third category for missing/inconsistent reporting. Age at time of the PSA test was categorized into 4 groups (≤ 39, 40-54, 55-69, and ≥ 70 years) based on AUA guidelines.4 The annual PSA testing rate was calculated for each veteran with SCI as the number of PSA tests per year. A mean annual PSA test rate was then calculated as the weighted (by exposure time) mean value for all annual PSA testing rates from 2000 through 2017 for each age group and race. Annual exposure was calculated for each veteran and defined as the number of days a veteran was eligible to have a PSA test. This started with the date of SCI diagnosis and ended with either the date of death or the date of last PSA. If a veteran moved from one age group to another in 1 year, the first part of this year’s exposure was included in the calculation of the annual PSA testing rate for the younger group and the second part was included for the calculation of the older group. For deceased veterans, the death date was excluded from the exposure period, and their exposure period ended on the day before death.
Statistical Analysis
To compare PSA testing rates between African American race and other races, Poisson regression was used with exposure treated as an offset (exposures were summed across years for each veteran). An indicator (dummy) variable for African American race vs other races was coded, and statistical significance was set at P < .05. To check sensitivity for the Poisson assumption that the mean was equal to the variance, negative binomial regression was used. To assess for geographic PSA testing rate variability, the data were further analyzed based on the locations where PSA tests were performed. This subanalysis was limited to veterans who had all PSA tests in a single station. For each station, the average PSA testing rate was calculated for each veteran, and the mean for all annual PSA testing rates was used to determine station-specific PSA testing rates.
Results
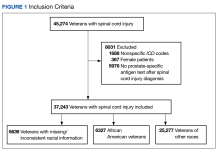
A total of 45,274 veterans were initially identified of which 367 females were excluded (Figure 1).
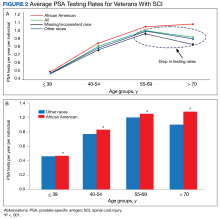
The PSA testing rate rose for veterans in the age groups ≤ 39, 40 to 54, and 55 to 69 years (Figure 2A).
Of the cohort of 37,243 veterans, 28,396 (76.2%) had their post-SCI tests done at a single facility, 6770 (18.1%) at 2 locations, and 2077 (5.5%) at > 2 locations. Single-station group data were included in a subanalysis to determine the mean (SD) PSA testing rates, which for the 123 locations was 0.98 (0.36) tests per veteran per year (range, 0.2-3.0 tests per veteran per year).
To assess the impact of the 2012 USPSTF recommendations on PSA testing rates in veterans with SCI, mean PSA testing rates were calculated for 5 years before the recommendations (2007-2011) and compared with the average PSA testing rate for 5 years following the updated recommendations (2013-2017). The USPSTF updated its recommendation again in 2018 and acknowledged the potential benefit for PSA screening in certain patient populations.2,3 Surprisingly, and despite recommendations, the results show a significant increase in PSA testing rates in all age groups for all races (P < .001) (Figure 4).
Discussion
The goal of this study was to establish testing rates and analyze PSA testing trends across races and age groups in veterans with SCI. This is the largest cohort of patients with SCI analyzed in the literature. The key findings of this study were that despite clear AUA guidelines recommending against PSA testing in patients aged ≤ 39 years and ≥ 70 years, there are high rates of testing in veterans with SCI in these age groups (0.46 tests per year in those aged ≤ 39 years and 0.91 tests per year in those aged ≥ 70 years). In terms of race, as expected based on increased risk,
Prostate Cancer Incidence
Although the exact mechanism behind alterations in prostate function in the SCI population have yet to be fully elucidated, research suggests that the prostate behaves differently after SCI. Animal models of prostate gland denervation show decreased prostate volume and suggest that SCI may lead to a reduction in prostatic secretory function associated with autonomic dysfunction. Shim and colleagues hypothesized that impaired autonomic prostate innervation alters the prostatic volume and PSA in patients with SCI.10
Additional studies looking at actual PSA levels in men with SCI reveal conflicting data.10-15,20 Toricelli and colleagues retrospectively studied 140 men with SCI, of whom 34 had PSA levels available and found that mean PSA was not significantly different for patients with SCI compared with controls, but patients using clean intermittent catheterization had 2-fold higher PSA levels.21 In contrast, Konety and colleagues found that mean PSA was not significantly different from uninjured controls in their cohort of 79 patients with SCI, though they did find a correlation between indwelling catheter use and a higher PSA.22
Studies have shown an overall decreased risk of prostate cancer in patients with SCI, though the mechanism remains unclear. A large cohort study from Taiwan showed a lower risk of prostate cancer for 54,401 patients with SCI with an adjusted hazard ratio of 0.73.23 Patel and colleagues found the overall rate of prostate cancer in the population of veterans with SCI was lower than the general uninjured VA population, though this study was limited by scope with only 350 patients with SCI.24 A more recent systematic review and meta-analysis of 9 studies evaluating the prevalence of prostate cancer in men with SCI found a reduction of up to 65% in the risk of prostate cancer in men with SCI, and PSA was found to be a poor screening tool for prostate cancer due to large study heterogeneity.16
PSA Screening
This study identified widespread overscreening using the PSA test in veterans with SCI, which is likely attributable to many factors. Per VHA Directive 1176, all eligible veterans are offered yearly interdisciplinary comprehensive evaluations, including laboratory testing, and as such veterans with SCI have high rates of annual visit attendance due to the complexity of their care.9 PSA testing is included in the standard battery of laboratory tests ordered for all patients with SCI during their annual examinations. Additionally, many SCI specialists use the PSA level in patients with SCI for identifying cystitis or prostatitis in patients with colonization who may not experience typical symptoms. Everaert and colleagues demonstrated the clinical utility for localizing UTIs to the upper or lower tract, with elevated PSA indicating prostatitis. They found that serum PSA has a sensitivity of 68% and a specificity of 100% in the differential diagnosis of prostatitis and pyelonephritis.25 As such, the high PSA screening rates may be reflective of diagnostic use for infection rather than for cancer screening.
Likely as a response to the USPSTF recommendations, there has been a national slow decline in overall PSA screening rates since 2012.26-28 A study from Vetterlein and colleagues examining changes in the PSA screening trends related to USPSTF recommendations found an 8.5% decline in overall PSA screening from 2012 to 2014.29 However, the increase in PSA testing across all ages and races in the VA population with SCI over the same period is not entirely understood and suggests the need for further research and education in this area.
Limitations
This study is limited by the use of data identified by ICD codes rather than by review of individual health records. This required the use of decision algorithms for data points, such as the date of SCI. In addition, analysis was not able to capture shared decision making that may have contributed to PSA screening outside the recommended age ranges based on additional risk factors, such as family history of lethal malignancy. Furthermore, a detailed attempt to define specific age-adjusted PSA levels was beyond the scope of this study but will be addressed in later publications. In addition, we did not exclude individuals with a diagnosis of prostate adenocarcinoma, prostatitis, or recurrent UTIs because the onset, duration, and severity of disease could not be definitively ascertained. Finally, veterans with SCI are unique and may not be reflective of individuals with SCI who do not receive care within the VA. However, despite these limitations, this is, to our knowledge, the largest and most comprehensive study evaluating PSA testing rates in individuals with SCI.
Conclusions
Currently, PSA screening is recommended following shared decision making for patients at average risk aged 55 to 70 years. Patients with SCI experience many conditions that may affect PSA values, but data regarding normal PSA ranges and rates of prostate cancer in this population remain sparse. The study demonstrated high rates of overtesting in veterans with SCI, higher than expected testing rates in African American veterans, a paradoxical increase in PSA testing rates after the 2012 publication of the USPSTF PSA guidelines, and wide variability in testing rates depending on VA location.
African American men were tested at higher rates across all age groups, including in patients aged > 70 years. To balance the benefits of detecting clinically significant prostate cancer vs the risks of invasive testing in high-risk populations with SCI, more work is needed to determine the clinical impact of screening practices. Future work is currently ongoing to define age-based PSA values in patients with SCI.
Acknowledgments
This research was supported in part through funding from the Center for Rehabilitation Science and Engineering, Virginia Commonwealth University Health System.
1. American Cancer Society. Key statistics for prostate cancer. Updated January 12, 2023. Accessed June 2, 2023. https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html
2. Moyer VA; U.S. Preventive Services Task Force. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;157(2):120-134. doi:10.7326/0003-4819-157-2-201207170-00459
3. US Preventive Services Task Force, Grossman DC, Curry SJ, et al. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(18):1901-1913. doi:10.1001/jama.2018.3710
4. Carter HB, Albertsen PC, Barry MJ, et al. Early detection of prostate cancer: AUA Guideline. J Urol. 2013;190(2):419-426. doi:10.1016/j.juro.2013.04.119
5. US Department of Veterans Affairs, Veterans Health Administration. Updated August 15, 2022. Accessed June 2, 2023. https://www.va.gov/health/aboutVHA.asp
6. US Department of Veterans Affairs. Spinal cord injuries and disorders system of care. Updated January 31, 2022. Accessed June 2, 2023. https://www.sci.va.gov/VAs_SCID_System_of_Care.asp
7. DeVivo MJ, Chen Y, Wen H. Cause of death trends among persons with spinal cord injury in the United States: 1960-2017. Arch Phys Med Rehabil. 2022;103(4):634-641. doi:10.1016/j.apmr.2021.09.019
8. Cao Y, DiPiro N, Krause JS. Health factors and spinal cord injury: a prospective study of risk of cause-specific mortality. Spinal Cord. 2019;57(7):594-602. doi:10.1038/s41393-019-0264-6
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1176(2): Spinal Cord Injuries and Disorders System of Care. Published September 30, 2019. Accessed June 2, 2023. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=8523
10. Shim HB, Jung TY, Lee JK, Ku JH. Prostate activity and prostate cancer in spinal cord injury. Prostate Cancer Prostatic Dis. 2006;9(2):115-120. doi:10.1038/sj.pcan.4500865
11. Lynne CM, Aballa TC, Wang TJ, Rittenhouse HG, Ferrell SM, Brackett NL. Serum and semen prostate specific antigen concentrations are different in young spinal cord injured men compared to normal controls. J Urol. 1999;162(1):89-91. doi:10.1097/00005392-199907000-00022
12. Bartoletti R, Gavazzi A, Cai T, et al. Prostate growth and prevalence of prostate diseases in early onset spinal cord injuries. Eur Urol. 2009;56(1):142-148. doi:10.1016/j.eururo.2008.01.088
13. Pannek J, Berges RR, Cubick G, Meindl R, Senge T. Prostate size and PSA serum levels in male patients with spinal cord injury. Urology. 2003;62(5):845-848. doi:10.1016/s0090-4295(03)00654-x
14. Pramudji CK, Mutchnik SE, DeConcini D, Boone TB. Prostate cancer screening with prostate specific antigen in spinal cord injured men. J Urol. 2002;167(3):1303-1305.
15. Alexandrino AP, Rodrigues MA, Matsuo T. Evaluation of serum and seminal levels of prostate specific antigen in men with spinal cord injury. J Urol. 2004;171(6 Pt 1):2230-2232. doi:10.1097/01.ju.0000125241.77517.10
16. Barbonetti A, D’Andrea S, Martorella A, Felzani G, Francavilla S, Francavilla F. Risk of prostate cancer in men with spinal cord injury: a systematic review and meta-analysis. Asian J Androl. 2018;20(6):555-560. doi:10.4103/aja.aja_31_18
17. Vince RA Jr, Jiang R, Bank M, et al. Evaluation of social determinants of health and prostate cancer outcomes among black and white patients: a systematic review and meta-analysis. JAMA Netw Open. 2023;6(1):e2250416. Published 2023 Jan 3. doi:10.1001/jamanetworkopen.2022.50416
18. Smith ZL, Eggener SE, Murphy AB. African-American prostate cancer disparities. Curr Urol Rep. 2017;18(10):81. Published 2017 Aug 14. doi:10.1007/s11934-017-0724-5
19. Jeong SH, Werneburg GT, Abouassaly R, Wood H. Acquired and congenital spinal cord injury is associated with lower likelihood of prostate specific antigen screening. Urology. 2022;164:178-183. doi:10.1016/j.urology.2022.01.044
20. Benaim EA, Montoya JD, Saboorian MH, Litwiller S, Roehrborn CG. Characterization of prostate size, PSA and endocrine profiles in patients with spinal cord injuries. Prostate Cancer Prostatic Dis. 1998;1(5):250-255. doi:10.1038/sj.pcan.4500246
21. Torricelli FC, Lucon M, Vicentini F, Gomes CM, Srougi M, Bruschini H. PSA levels in men with spinal cord injury and under intermittent catheterization. Neurourol Urodyn. 2011;30(8):1522-1524. doi:10.1002/nau.21119
22. Konety BR, Nguyen TT, Brenes G, et al. Evaluation of the effect of spinal cord injury on serum PSA levels. Urology. 2000;56(1):82-86. doi:10.1016/s0090-4295(00)00548-3
23. Lee WY, Sun LM, Lin CL, et al. Risk of prostate and bladder cancers in patients with spinal cord injury: a population-based cohort study. Urol Oncol. 2014;32(1):51.e1-51.e517. doi:10.1016/j.urolonc.2013.07.019
24. Patel N, Ngo K, Hastings J, Ketchum N, Sepahpanah F. Prevalence of prostate cancer in patients with chronic spinal cord injury. PM R. 2011;3(7):633-636. doi:10.1016/j.pmrj.2011.04.024
25. Everaert K, Oostra C, Delanghe J, Vande Walle J, Van Laere M, Oosterlinck W. Diagnosis and localization of a complicated urinary tract infection in neurogenic bladder disease by tubular proteinuria and serum prostate specific antigen. Spinal Cord. 1998;36(1):33-38. doi:10.1038/sj.sc.3100520
26. Drazer MW, Huo D, Eggener SE. National prostate cancer screening rates after the 2012 US Preventive Services Task Force recommendation discouraging prostate-specific antigen-based screening. J Clin Oncol. 2015;33(22):2416-2423. doi:10.1200/JCO.2015.61.6532
27. Sammon JD, Abdollah F, Choueiri TK, et al. Prostate-specific antigen screening after 2012 US Preventive Services Task Force recommendations. JAMA. 2015;314(19):2077-2079. doi:10.1001/jama.2015.7273
28. Jemal A, Fedewa SA, Ma J, et al. Prostate cancer incidence and PSA testing patterns in relation to USPSTF screening recommendations. JAMA. 2015;314(19):2054-2061. doi:10.1001/jama.2015.14905
29. Vetterlein MW, Dalela D, Sammon JD, et al. State-by-state variation in prostate-specific antigen screening trends following the 2011 United States Preventive Services Task Force panel update. Urology. 2018;112:56-65. doi:10.1016/j.urology.2017.08.055
Prostate cancer will be diagnosed in 12.5% of men during their lifetime. It is the most commonly diagnosed solid organ cancer in men.1 However, prostate cancer screening for prostate-specific antigen (PSA) remains controversial due to concerns about overdiagnosis, as the overall risk of dying of prostate cancer is only 2.4%.1
To address the risk and benefits of PSA testing, in 2012 the US Preventive Services Task Force (USPSTF) recommended against routine PSA testing.2 Updated 2018 recommendations continued this recommendation in men aged > 70 years but acknowledged a small potential benefit in men aged 55 to 69 years and suggested individualized shared decision making between patient and clinician.3 In addition, American Urological Association (AUA) guidelines for the early detection of prostate cancer recommend against PSA screening in men aged < 40 years or those aged > 70 years, shared decision making for individuals aged 55 to 70 years or in high-risk men aged 40 to 55 years (ie, family history of prostate cancer or African American race).4 PSA screening is not recommended for men with a life expectancy shorter than 10 to 15 years aged > 70 years.4
The Veterans Health Administration (VHA) is the largest integrated health care system in the US.5 In addition, the US Department of Veterans Affairs (VA) Spinal Cord Injury and Disorders System of Care operates 25 centers throughout the US.6 Life expectancy following spinal cord injury (SCI) increased significantly through the 1980s but has since plateaued, with life expectancy being impacted by age at injury, completeness of injury, and neurologic level.7,8 As part of a program of uniform care, all persons with SCI followed at the Spinal Cord Injury and Disorders System of Care centers are offered comprehensive annual evaluations, including screening laboratory tests, such as PSA level.9
Patients with SCI present a unique challenge when interpreting PSA levels, given potentially confounding factors, including neurogenic bladder management, high rates of bacteriuria, urinary tract infections (UTIs), testosterone deficiency, and pelvic innervation that differs from the noninjured population.10,11 Unfortunately, the literature on prostate cancer prevalence and average PSA levels in patients with SCI is limited by the small scope of studies and inconsistent data.10-16 Therefore, the purpose of the current investigation was to quantify and analyze the rates of annual PSA testing for all men with SCI in the VHA.
Methods
Approval was granted by the Richmond VA Medical Center (VAMC) Institutional Review Board in Virginia, and by the VA Informatics and Computing Infrastructure (VINCI) data access request tracker system for extraction of data from the VA Corporate Data Warehouse. Microsoft Structured Query Language was used for data programming and query design. Statistical analysis was conducted using Stata version 15.1 with assistance from professional biostatisticians.
Only male veterans with a nervous system disorder affecting the spinal cord or with myelopathy were included, based on International Classification of Diseases (ICD) version 9 and 10 codes, corresponding to traumatic and nontraumatic myelopathy. Veterans diagnosed with myelopathy based on ICD codes corresponding to progressive or degenerative myelopathies, such as multiple sclerosis or amyotrophic lateral sclerosis, were excluded.
For each veteran, extracted data included the unique identification number, date of birth, ICD code, date ICD code first appeared, race, gender, death status (yes/no), date of death (when applicable), date of each PSA test, PSA test values, and the VAMC where each test was performed. Only tests for total PSA were included. The date that the ICD code first appeared served as an approximation for the date of SCI. The time frame for the study included all PSA tests in the VINCI database for 2000 through 2017. However, only post-SCI PSA tests were included in the analysis. Duplicate tests (same date/time) were eliminated.
Race is considered a risk factor for prostate cancer only for African American patients, likely due to racial health disparities.17 Given this, we chose to categorize race as either African American or other, with a third category for missing/inconsistent reporting. Age at time of the PSA test was categorized into 4 groups (≤ 39, 40-54, 55-69, and ≥ 70 years) based on AUA guidelines.4 The annual PSA testing rate was calculated for each veteran with SCI as the number of PSA tests per year. A mean annual PSA test rate was then calculated as the weighted (by exposure time) mean value for all annual PSA testing rates from 2000 through 2017 for each age group and race. Annual exposure was calculated for each veteran and defined as the number of days a veteran was eligible to have a PSA test. This started with the date of SCI diagnosis and ended with either the date of death or the date of last PSA. If a veteran moved from one age group to another in 1 year, the first part of this year’s exposure was included in the calculation of the annual PSA testing rate for the younger group and the second part was included for the calculation of the older group. For deceased veterans, the death date was excluded from the exposure period, and their exposure period ended on the day before death.
Statistical Analysis
To compare PSA testing rates between African American race and other races, Poisson regression was used with exposure treated as an offset (exposures were summed across years for each veteran). An indicator (dummy) variable for African American race vs other races was coded, and statistical significance was set at P < .05. To check sensitivity for the Poisson assumption that the mean was equal to the variance, negative binomial regression was used. To assess for geographic PSA testing rate variability, the data were further analyzed based on the locations where PSA tests were performed. This subanalysis was limited to veterans who had all PSA tests in a single station. For each station, the average PSA testing rate was calculated for each veteran, and the mean for all annual PSA testing rates was used to determine station-specific PSA testing rates.
Results
A total of 45,274 veterans were initially identified of which 367 females were excluded (Figure 1).
The PSA testing rate rose for veterans in the age groups ≤ 39, 40 to 54, and 55 to 69 years (Figure 2A).
Of the cohort of 37,243 veterans, 28,396 (76.2%) had their post-SCI tests done at a single facility, 6770 (18.1%) at 2 locations, and 2077 (5.5%) at > 2 locations. Single-station group data were included in a subanalysis to determine the mean (SD) PSA testing rates, which for the 123 locations was 0.98 (0.36) tests per veteran per year (range, 0.2-3.0 tests per veteran per year).
To assess the impact of the 2012 USPSTF recommendations on PSA testing rates in veterans with SCI, mean PSA testing rates were calculated for 5 years before the recommendations (2007-2011) and compared with the average PSA testing rate for 5 years following the updated recommendations (2013-2017). The USPSTF updated its recommendation again in 2018 and acknowledged the potential benefit for PSA screening in certain patient populations.2,3 Surprisingly, and despite recommendations, the results show a significant increase in PSA testing rates in all age groups for all races (P < .001) (Figure 4).
Discussion
The goal of this study was to establish testing rates and analyze PSA testing trends across races and age groups in veterans with SCI. This is the largest cohort of patients with SCI analyzed in the literature. The key findings of this study were that despite clear AUA guidelines recommending against PSA testing in patients aged ≤ 39 years and ≥ 70 years, there are high rates of testing in veterans with SCI in these age groups (0.46 tests per year in those aged ≤ 39 years and 0.91 tests per year in those aged ≥ 70 years). In terms of race, as expected based on increased risk,
Prostate Cancer Incidence
Although the exact mechanism behind alterations in prostate function in the SCI population have yet to be fully elucidated, research suggests that the prostate behaves differently after SCI. Animal models of prostate gland denervation show decreased prostate volume and suggest that SCI may lead to a reduction in prostatic secretory function associated with autonomic dysfunction. Shim and colleagues hypothesized that impaired autonomic prostate innervation alters the prostatic volume and PSA in patients with SCI.10
Additional studies looking at actual PSA levels in men with SCI reveal conflicting data.10-15,20 Toricelli and colleagues retrospectively studied 140 men with SCI, of whom 34 had PSA levels available and found that mean PSA was not significantly different for patients with SCI compared with controls, but patients using clean intermittent catheterization had 2-fold higher PSA levels.21 In contrast, Konety and colleagues found that mean PSA was not significantly different from uninjured controls in their cohort of 79 patients with SCI, though they did find a correlation between indwelling catheter use and a higher PSA.22
Studies have shown an overall decreased risk of prostate cancer in patients with SCI, though the mechanism remains unclear. A large cohort study from Taiwan showed a lower risk of prostate cancer for 54,401 patients with SCI with an adjusted hazard ratio of 0.73.23 Patel and colleagues found the overall rate of prostate cancer in the population of veterans with SCI was lower than the general uninjured VA population, though this study was limited by scope with only 350 patients with SCI.24 A more recent systematic review and meta-analysis of 9 studies evaluating the prevalence of prostate cancer in men with SCI found a reduction of up to 65% in the risk of prostate cancer in men with SCI, and PSA was found to be a poor screening tool for prostate cancer due to large study heterogeneity.16
PSA Screening
This study identified widespread overscreening using the PSA test in veterans with SCI, which is likely attributable to many factors. Per VHA Directive 1176, all eligible veterans are offered yearly interdisciplinary comprehensive evaluations, including laboratory testing, and as such veterans with SCI have high rates of annual visit attendance due to the complexity of their care.9 PSA testing is included in the standard battery of laboratory tests ordered for all patients with SCI during their annual examinations. Additionally, many SCI specialists use the PSA level in patients with SCI for identifying cystitis or prostatitis in patients with colonization who may not experience typical symptoms. Everaert and colleagues demonstrated the clinical utility for localizing UTIs to the upper or lower tract, with elevated PSA indicating prostatitis. They found that serum PSA has a sensitivity of 68% and a specificity of 100% in the differential diagnosis of prostatitis and pyelonephritis.25 As such, the high PSA screening rates may be reflective of diagnostic use for infection rather than for cancer screening.
Likely as a response to the USPSTF recommendations, there has been a national slow decline in overall PSA screening rates since 2012.26-28 A study from Vetterlein and colleagues examining changes in the PSA screening trends related to USPSTF recommendations found an 8.5% decline in overall PSA screening from 2012 to 2014.29 However, the increase in PSA testing across all ages and races in the VA population with SCI over the same period is not entirely understood and suggests the need for further research and education in this area.
Limitations
This study is limited by the use of data identified by ICD codes rather than by review of individual health records. This required the use of decision algorithms for data points, such as the date of SCI. In addition, analysis was not able to capture shared decision making that may have contributed to PSA screening outside the recommended age ranges based on additional risk factors, such as family history of lethal malignancy. Furthermore, a detailed attempt to define specific age-adjusted PSA levels was beyond the scope of this study but will be addressed in later publications. In addition, we did not exclude individuals with a diagnosis of prostate adenocarcinoma, prostatitis, or recurrent UTIs because the onset, duration, and severity of disease could not be definitively ascertained. Finally, veterans with SCI are unique and may not be reflective of individuals with SCI who do not receive care within the VA. However, despite these limitations, this is, to our knowledge, the largest and most comprehensive study evaluating PSA testing rates in individuals with SCI.
Conclusions
Currently, PSA screening is recommended following shared decision making for patients at average risk aged 55 to 70 years. Patients with SCI experience many conditions that may affect PSA values, but data regarding normal PSA ranges and rates of prostate cancer in this population remain sparse. The study demonstrated high rates of overtesting in veterans with SCI, higher than expected testing rates in African American veterans, a paradoxical increase in PSA testing rates after the 2012 publication of the USPSTF PSA guidelines, and wide variability in testing rates depending on VA location.
African American men were tested at higher rates across all age groups, including in patients aged > 70 years. To balance the benefits of detecting clinically significant prostate cancer vs the risks of invasive testing in high-risk populations with SCI, more work is needed to determine the clinical impact of screening practices. Future work is currently ongoing to define age-based PSA values in patients with SCI.
Acknowledgments
This research was supported in part through funding from the Center for Rehabilitation Science and Engineering, Virginia Commonwealth University Health System.
Prostate cancer will be diagnosed in 12.5% of men during their lifetime. It is the most commonly diagnosed solid organ cancer in men.1 However, prostate cancer screening for prostate-specific antigen (PSA) remains controversial due to concerns about overdiagnosis, as the overall risk of dying of prostate cancer is only 2.4%.1
To address the risk and benefits of PSA testing, in 2012 the US Preventive Services Task Force (USPSTF) recommended against routine PSA testing.2 Updated 2018 recommendations continued this recommendation in men aged > 70 years but acknowledged a small potential benefit in men aged 55 to 69 years and suggested individualized shared decision making between patient and clinician.3 In addition, American Urological Association (AUA) guidelines for the early detection of prostate cancer recommend against PSA screening in men aged < 40 years or those aged > 70 years, shared decision making for individuals aged 55 to 70 years or in high-risk men aged 40 to 55 years (ie, family history of prostate cancer or African American race).4 PSA screening is not recommended for men with a life expectancy shorter than 10 to 15 years aged > 70 years.4
The Veterans Health Administration (VHA) is the largest integrated health care system in the US.5 In addition, the US Department of Veterans Affairs (VA) Spinal Cord Injury and Disorders System of Care operates 25 centers throughout the US.6 Life expectancy following spinal cord injury (SCI) increased significantly through the 1980s but has since plateaued, with life expectancy being impacted by age at injury, completeness of injury, and neurologic level.7,8 As part of a program of uniform care, all persons with SCI followed at the Spinal Cord Injury and Disorders System of Care centers are offered comprehensive annual evaluations, including screening laboratory tests, such as PSA level.9
Patients with SCI present a unique challenge when interpreting PSA levels, given potentially confounding factors, including neurogenic bladder management, high rates of bacteriuria, urinary tract infections (UTIs), testosterone deficiency, and pelvic innervation that differs from the noninjured population.10,11 Unfortunately, the literature on prostate cancer prevalence and average PSA levels in patients with SCI is limited by the small scope of studies and inconsistent data.10-16 Therefore, the purpose of the current investigation was to quantify and analyze the rates of annual PSA testing for all men with SCI in the VHA.
Methods
Approval was granted by the Richmond VA Medical Center (VAMC) Institutional Review Board in Virginia, and by the VA Informatics and Computing Infrastructure (VINCI) data access request tracker system for extraction of data from the VA Corporate Data Warehouse. Microsoft Structured Query Language was used for data programming and query design. Statistical analysis was conducted using Stata version 15.1 with assistance from professional biostatisticians.
Only male veterans with a nervous system disorder affecting the spinal cord or with myelopathy were included, based on International Classification of Diseases (ICD) version 9 and 10 codes, corresponding to traumatic and nontraumatic myelopathy. Veterans diagnosed with myelopathy based on ICD codes corresponding to progressive or degenerative myelopathies, such as multiple sclerosis or amyotrophic lateral sclerosis, were excluded.
For each veteran, extracted data included the unique identification number, date of birth, ICD code, date ICD code first appeared, race, gender, death status (yes/no), date of death (when applicable), date of each PSA test, PSA test values, and the VAMC where each test was performed. Only tests for total PSA were included. The date that the ICD code first appeared served as an approximation for the date of SCI. The time frame for the study included all PSA tests in the VINCI database for 2000 through 2017. However, only post-SCI PSA tests were included in the analysis. Duplicate tests (same date/time) were eliminated.
Race is considered a risk factor for prostate cancer only for African American patients, likely due to racial health disparities.17 Given this, we chose to categorize race as either African American or other, with a third category for missing/inconsistent reporting. Age at time of the PSA test was categorized into 4 groups (≤ 39, 40-54, 55-69, and ≥ 70 years) based on AUA guidelines.4 The annual PSA testing rate was calculated for each veteran with SCI as the number of PSA tests per year. A mean annual PSA test rate was then calculated as the weighted (by exposure time) mean value for all annual PSA testing rates from 2000 through 2017 for each age group and race. Annual exposure was calculated for each veteran and defined as the number of days a veteran was eligible to have a PSA test. This started with the date of SCI diagnosis and ended with either the date of death or the date of last PSA. If a veteran moved from one age group to another in 1 year, the first part of this year’s exposure was included in the calculation of the annual PSA testing rate for the younger group and the second part was included for the calculation of the older group. For deceased veterans, the death date was excluded from the exposure period, and their exposure period ended on the day before death.
Statistical Analysis
To compare PSA testing rates between African American race and other races, Poisson regression was used with exposure treated as an offset (exposures were summed across years for each veteran). An indicator (dummy) variable for African American race vs other races was coded, and statistical significance was set at P < .05. To check sensitivity for the Poisson assumption that the mean was equal to the variance, negative binomial regression was used. To assess for geographic PSA testing rate variability, the data were further analyzed based on the locations where PSA tests were performed. This subanalysis was limited to veterans who had all PSA tests in a single station. For each station, the average PSA testing rate was calculated for each veteran, and the mean for all annual PSA testing rates was used to determine station-specific PSA testing rates.
Results
A total of 45,274 veterans were initially identified of which 367 females were excluded (Figure 1).
The PSA testing rate rose for veterans in the age groups ≤ 39, 40 to 54, and 55 to 69 years (Figure 2A).
Of the cohort of 37,243 veterans, 28,396 (76.2%) had their post-SCI tests done at a single facility, 6770 (18.1%) at 2 locations, and 2077 (5.5%) at > 2 locations. Single-station group data were included in a subanalysis to determine the mean (SD) PSA testing rates, which for the 123 locations was 0.98 (0.36) tests per veteran per year (range, 0.2-3.0 tests per veteran per year).
To assess the impact of the 2012 USPSTF recommendations on PSA testing rates in veterans with SCI, mean PSA testing rates were calculated for 5 years before the recommendations (2007-2011) and compared with the average PSA testing rate for 5 years following the updated recommendations (2013-2017). The USPSTF updated its recommendation again in 2018 and acknowledged the potential benefit for PSA screening in certain patient populations.2,3 Surprisingly, and despite recommendations, the results show a significant increase in PSA testing rates in all age groups for all races (P < .001) (Figure 4).
Discussion
The goal of this study was to establish testing rates and analyze PSA testing trends across races and age groups in veterans with SCI. This is the largest cohort of patients with SCI analyzed in the literature. The key findings of this study were that despite clear AUA guidelines recommending against PSA testing in patients aged ≤ 39 years and ≥ 70 years, there are high rates of testing in veterans with SCI in these age groups (0.46 tests per year in those aged ≤ 39 years and 0.91 tests per year in those aged ≥ 70 years). In terms of race, as expected based on increased risk,
Prostate Cancer Incidence
Although the exact mechanism behind alterations in prostate function in the SCI population have yet to be fully elucidated, research suggests that the prostate behaves differently after SCI. Animal models of prostate gland denervation show decreased prostate volume and suggest that SCI may lead to a reduction in prostatic secretory function associated with autonomic dysfunction. Shim and colleagues hypothesized that impaired autonomic prostate innervation alters the prostatic volume and PSA in patients with SCI.10
Additional studies looking at actual PSA levels in men with SCI reveal conflicting data.10-15,20 Toricelli and colleagues retrospectively studied 140 men with SCI, of whom 34 had PSA levels available and found that mean PSA was not significantly different for patients with SCI compared with controls, but patients using clean intermittent catheterization had 2-fold higher PSA levels.21 In contrast, Konety and colleagues found that mean PSA was not significantly different from uninjured controls in their cohort of 79 patients with SCI, though they did find a correlation between indwelling catheter use and a higher PSA.22
Studies have shown an overall decreased risk of prostate cancer in patients with SCI, though the mechanism remains unclear. A large cohort study from Taiwan showed a lower risk of prostate cancer for 54,401 patients with SCI with an adjusted hazard ratio of 0.73.23 Patel and colleagues found the overall rate of prostate cancer in the population of veterans with SCI was lower than the general uninjured VA population, though this study was limited by scope with only 350 patients with SCI.24 A more recent systematic review and meta-analysis of 9 studies evaluating the prevalence of prostate cancer in men with SCI found a reduction of up to 65% in the risk of prostate cancer in men with SCI, and PSA was found to be a poor screening tool for prostate cancer due to large study heterogeneity.16
PSA Screening
This study identified widespread overscreening using the PSA test in veterans with SCI, which is likely attributable to many factors. Per VHA Directive 1176, all eligible veterans are offered yearly interdisciplinary comprehensive evaluations, including laboratory testing, and as such veterans with SCI have high rates of annual visit attendance due to the complexity of their care.9 PSA testing is included in the standard battery of laboratory tests ordered for all patients with SCI during their annual examinations. Additionally, many SCI specialists use the PSA level in patients with SCI for identifying cystitis or prostatitis in patients with colonization who may not experience typical symptoms. Everaert and colleagues demonstrated the clinical utility for localizing UTIs to the upper or lower tract, with elevated PSA indicating prostatitis. They found that serum PSA has a sensitivity of 68% and a specificity of 100% in the differential diagnosis of prostatitis and pyelonephritis.25 As such, the high PSA screening rates may be reflective of diagnostic use for infection rather than for cancer screening.
Likely as a response to the USPSTF recommendations, there has been a national slow decline in overall PSA screening rates since 2012.26-28 A study from Vetterlein and colleagues examining changes in the PSA screening trends related to USPSTF recommendations found an 8.5% decline in overall PSA screening from 2012 to 2014.29 However, the increase in PSA testing across all ages and races in the VA population with SCI over the same period is not entirely understood and suggests the need for further research and education in this area.
Limitations
This study is limited by the use of data identified by ICD codes rather than by review of individual health records. This required the use of decision algorithms for data points, such as the date of SCI. In addition, analysis was not able to capture shared decision making that may have contributed to PSA screening outside the recommended age ranges based on additional risk factors, such as family history of lethal malignancy. Furthermore, a detailed attempt to define specific age-adjusted PSA levels was beyond the scope of this study but will be addressed in later publications. In addition, we did not exclude individuals with a diagnosis of prostate adenocarcinoma, prostatitis, or recurrent UTIs because the onset, duration, and severity of disease could not be definitively ascertained. Finally, veterans with SCI are unique and may not be reflective of individuals with SCI who do not receive care within the VA. However, despite these limitations, this is, to our knowledge, the largest and most comprehensive study evaluating PSA testing rates in individuals with SCI.
Conclusions
Currently, PSA screening is recommended following shared decision making for patients at average risk aged 55 to 70 years. Patients with SCI experience many conditions that may affect PSA values, but data regarding normal PSA ranges and rates of prostate cancer in this population remain sparse. The study demonstrated high rates of overtesting in veterans with SCI, higher than expected testing rates in African American veterans, a paradoxical increase in PSA testing rates after the 2012 publication of the USPSTF PSA guidelines, and wide variability in testing rates depending on VA location.
African American men were tested at higher rates across all age groups, including in patients aged > 70 years. To balance the benefits of detecting clinically significant prostate cancer vs the risks of invasive testing in high-risk populations with SCI, more work is needed to determine the clinical impact of screening practices. Future work is currently ongoing to define age-based PSA values in patients with SCI.
Acknowledgments
This research was supported in part through funding from the Center for Rehabilitation Science and Engineering, Virginia Commonwealth University Health System.
1. American Cancer Society. Key statistics for prostate cancer. Updated January 12, 2023. Accessed June 2, 2023. https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html
2. Moyer VA; U.S. Preventive Services Task Force. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;157(2):120-134. doi:10.7326/0003-4819-157-2-201207170-00459
3. US Preventive Services Task Force, Grossman DC, Curry SJ, et al. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(18):1901-1913. doi:10.1001/jama.2018.3710
4. Carter HB, Albertsen PC, Barry MJ, et al. Early detection of prostate cancer: AUA Guideline. J Urol. 2013;190(2):419-426. doi:10.1016/j.juro.2013.04.119
5. US Department of Veterans Affairs, Veterans Health Administration. Updated August 15, 2022. Accessed June 2, 2023. https://www.va.gov/health/aboutVHA.asp
6. US Department of Veterans Affairs. Spinal cord injuries and disorders system of care. Updated January 31, 2022. Accessed June 2, 2023. https://www.sci.va.gov/VAs_SCID_System_of_Care.asp
7. DeVivo MJ, Chen Y, Wen H. Cause of death trends among persons with spinal cord injury in the United States: 1960-2017. Arch Phys Med Rehabil. 2022;103(4):634-641. doi:10.1016/j.apmr.2021.09.019
8. Cao Y, DiPiro N, Krause JS. Health factors and spinal cord injury: a prospective study of risk of cause-specific mortality. Spinal Cord. 2019;57(7):594-602. doi:10.1038/s41393-019-0264-6
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1176(2): Spinal Cord Injuries and Disorders System of Care. Published September 30, 2019. Accessed June 2, 2023. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=8523
10. Shim HB, Jung TY, Lee JK, Ku JH. Prostate activity and prostate cancer in spinal cord injury. Prostate Cancer Prostatic Dis. 2006;9(2):115-120. doi:10.1038/sj.pcan.4500865
11. Lynne CM, Aballa TC, Wang TJ, Rittenhouse HG, Ferrell SM, Brackett NL. Serum and semen prostate specific antigen concentrations are different in young spinal cord injured men compared to normal controls. J Urol. 1999;162(1):89-91. doi:10.1097/00005392-199907000-00022
12. Bartoletti R, Gavazzi A, Cai T, et al. Prostate growth and prevalence of prostate diseases in early onset spinal cord injuries. Eur Urol. 2009;56(1):142-148. doi:10.1016/j.eururo.2008.01.088
13. Pannek J, Berges RR, Cubick G, Meindl R, Senge T. Prostate size and PSA serum levels in male patients with spinal cord injury. Urology. 2003;62(5):845-848. doi:10.1016/s0090-4295(03)00654-x
14. Pramudji CK, Mutchnik SE, DeConcini D, Boone TB. Prostate cancer screening with prostate specific antigen in spinal cord injured men. J Urol. 2002;167(3):1303-1305.
15. Alexandrino AP, Rodrigues MA, Matsuo T. Evaluation of serum and seminal levels of prostate specific antigen in men with spinal cord injury. J Urol. 2004;171(6 Pt 1):2230-2232. doi:10.1097/01.ju.0000125241.77517.10
16. Barbonetti A, D’Andrea S, Martorella A, Felzani G, Francavilla S, Francavilla F. Risk of prostate cancer in men with spinal cord injury: a systematic review and meta-analysis. Asian J Androl. 2018;20(6):555-560. doi:10.4103/aja.aja_31_18
17. Vince RA Jr, Jiang R, Bank M, et al. Evaluation of social determinants of health and prostate cancer outcomes among black and white patients: a systematic review and meta-analysis. JAMA Netw Open. 2023;6(1):e2250416. Published 2023 Jan 3. doi:10.1001/jamanetworkopen.2022.50416
18. Smith ZL, Eggener SE, Murphy AB. African-American prostate cancer disparities. Curr Urol Rep. 2017;18(10):81. Published 2017 Aug 14. doi:10.1007/s11934-017-0724-5
19. Jeong SH, Werneburg GT, Abouassaly R, Wood H. Acquired and congenital spinal cord injury is associated with lower likelihood of prostate specific antigen screening. Urology. 2022;164:178-183. doi:10.1016/j.urology.2022.01.044
20. Benaim EA, Montoya JD, Saboorian MH, Litwiller S, Roehrborn CG. Characterization of prostate size, PSA and endocrine profiles in patients with spinal cord injuries. Prostate Cancer Prostatic Dis. 1998;1(5):250-255. doi:10.1038/sj.pcan.4500246
21. Torricelli FC, Lucon M, Vicentini F, Gomes CM, Srougi M, Bruschini H. PSA levels in men with spinal cord injury and under intermittent catheterization. Neurourol Urodyn. 2011;30(8):1522-1524. doi:10.1002/nau.21119
22. Konety BR, Nguyen TT, Brenes G, et al. Evaluation of the effect of spinal cord injury on serum PSA levels. Urology. 2000;56(1):82-86. doi:10.1016/s0090-4295(00)00548-3
23. Lee WY, Sun LM, Lin CL, et al. Risk of prostate and bladder cancers in patients with spinal cord injury: a population-based cohort study. Urol Oncol. 2014;32(1):51.e1-51.e517. doi:10.1016/j.urolonc.2013.07.019
24. Patel N, Ngo K, Hastings J, Ketchum N, Sepahpanah F. Prevalence of prostate cancer in patients with chronic spinal cord injury. PM R. 2011;3(7):633-636. doi:10.1016/j.pmrj.2011.04.024
25. Everaert K, Oostra C, Delanghe J, Vande Walle J, Van Laere M, Oosterlinck W. Diagnosis and localization of a complicated urinary tract infection in neurogenic bladder disease by tubular proteinuria and serum prostate specific antigen. Spinal Cord. 1998;36(1):33-38. doi:10.1038/sj.sc.3100520
26. Drazer MW, Huo D, Eggener SE. National prostate cancer screening rates after the 2012 US Preventive Services Task Force recommendation discouraging prostate-specific antigen-based screening. J Clin Oncol. 2015;33(22):2416-2423. doi:10.1200/JCO.2015.61.6532
27. Sammon JD, Abdollah F, Choueiri TK, et al. Prostate-specific antigen screening after 2012 US Preventive Services Task Force recommendations. JAMA. 2015;314(19):2077-2079. doi:10.1001/jama.2015.7273
28. Jemal A, Fedewa SA, Ma J, et al. Prostate cancer incidence and PSA testing patterns in relation to USPSTF screening recommendations. JAMA. 2015;314(19):2054-2061. doi:10.1001/jama.2015.14905
29. Vetterlein MW, Dalela D, Sammon JD, et al. State-by-state variation in prostate-specific antigen screening trends following the 2011 United States Preventive Services Task Force panel update. Urology. 2018;112:56-65. doi:10.1016/j.urology.2017.08.055
1. American Cancer Society. Key statistics for prostate cancer. Updated January 12, 2023. Accessed June 2, 2023. https://www.cancer.org/cancer/prostate-cancer/about/key-statistics.html
2. Moyer VA; U.S. Preventive Services Task Force. Screening for prostate cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2012;157(2):120-134. doi:10.7326/0003-4819-157-2-201207170-00459
3. US Preventive Services Task Force, Grossman DC, Curry SJ, et al. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA. 2018;319(18):1901-1913. doi:10.1001/jama.2018.3710
4. Carter HB, Albertsen PC, Barry MJ, et al. Early detection of prostate cancer: AUA Guideline. J Urol. 2013;190(2):419-426. doi:10.1016/j.juro.2013.04.119
5. US Department of Veterans Affairs, Veterans Health Administration. Updated August 15, 2022. Accessed June 2, 2023. https://www.va.gov/health/aboutVHA.asp
6. US Department of Veterans Affairs. Spinal cord injuries and disorders system of care. Updated January 31, 2022. Accessed June 2, 2023. https://www.sci.va.gov/VAs_SCID_System_of_Care.asp
7. DeVivo MJ, Chen Y, Wen H. Cause of death trends among persons with spinal cord injury in the United States: 1960-2017. Arch Phys Med Rehabil. 2022;103(4):634-641. doi:10.1016/j.apmr.2021.09.019
8. Cao Y, DiPiro N, Krause JS. Health factors and spinal cord injury: a prospective study of risk of cause-specific mortality. Spinal Cord. 2019;57(7):594-602. doi:10.1038/s41393-019-0264-6
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Directive 1176(2): Spinal Cord Injuries and Disorders System of Care. Published September 30, 2019. Accessed June 2, 2023. https://www.va.gov/vhapublications/ViewPublication.asp?pub_ID=8523
10. Shim HB, Jung TY, Lee JK, Ku JH. Prostate activity and prostate cancer in spinal cord injury. Prostate Cancer Prostatic Dis. 2006;9(2):115-120. doi:10.1038/sj.pcan.4500865
11. Lynne CM, Aballa TC, Wang TJ, Rittenhouse HG, Ferrell SM, Brackett NL. Serum and semen prostate specific antigen concentrations are different in young spinal cord injured men compared to normal controls. J Urol. 1999;162(1):89-91. doi:10.1097/00005392-199907000-00022
12. Bartoletti R, Gavazzi A, Cai T, et al. Prostate growth and prevalence of prostate diseases in early onset spinal cord injuries. Eur Urol. 2009;56(1):142-148. doi:10.1016/j.eururo.2008.01.088
13. Pannek J, Berges RR, Cubick G, Meindl R, Senge T. Prostate size and PSA serum levels in male patients with spinal cord injury. Urology. 2003;62(5):845-848. doi:10.1016/s0090-4295(03)00654-x
14. Pramudji CK, Mutchnik SE, DeConcini D, Boone TB. Prostate cancer screening with prostate specific antigen in spinal cord injured men. J Urol. 2002;167(3):1303-1305.
15. Alexandrino AP, Rodrigues MA, Matsuo T. Evaluation of serum and seminal levels of prostate specific antigen in men with spinal cord injury. J Urol. 2004;171(6 Pt 1):2230-2232. doi:10.1097/01.ju.0000125241.77517.10
16. Barbonetti A, D’Andrea S, Martorella A, Felzani G, Francavilla S, Francavilla F. Risk of prostate cancer in men with spinal cord injury: a systematic review and meta-analysis. Asian J Androl. 2018;20(6):555-560. doi:10.4103/aja.aja_31_18
17. Vince RA Jr, Jiang R, Bank M, et al. Evaluation of social determinants of health and prostate cancer outcomes among black and white patients: a systematic review and meta-analysis. JAMA Netw Open. 2023;6(1):e2250416. Published 2023 Jan 3. doi:10.1001/jamanetworkopen.2022.50416
18. Smith ZL, Eggener SE, Murphy AB. African-American prostate cancer disparities. Curr Urol Rep. 2017;18(10):81. Published 2017 Aug 14. doi:10.1007/s11934-017-0724-5
19. Jeong SH, Werneburg GT, Abouassaly R, Wood H. Acquired and congenital spinal cord injury is associated with lower likelihood of prostate specific antigen screening. Urology. 2022;164:178-183. doi:10.1016/j.urology.2022.01.044
20. Benaim EA, Montoya JD, Saboorian MH, Litwiller S, Roehrborn CG. Characterization of prostate size, PSA and endocrine profiles in patients with spinal cord injuries. Prostate Cancer Prostatic Dis. 1998;1(5):250-255. doi:10.1038/sj.pcan.4500246
21. Torricelli FC, Lucon M, Vicentini F, Gomes CM, Srougi M, Bruschini H. PSA levels in men with spinal cord injury and under intermittent catheterization. Neurourol Urodyn. 2011;30(8):1522-1524. doi:10.1002/nau.21119
22. Konety BR, Nguyen TT, Brenes G, et al. Evaluation of the effect of spinal cord injury on serum PSA levels. Urology. 2000;56(1):82-86. doi:10.1016/s0090-4295(00)00548-3
23. Lee WY, Sun LM, Lin CL, et al. Risk of prostate and bladder cancers in patients with spinal cord injury: a population-based cohort study. Urol Oncol. 2014;32(1):51.e1-51.e517. doi:10.1016/j.urolonc.2013.07.019
24. Patel N, Ngo K, Hastings J, Ketchum N, Sepahpanah F. Prevalence of prostate cancer in patients with chronic spinal cord injury. PM R. 2011;3(7):633-636. doi:10.1016/j.pmrj.2011.04.024
25. Everaert K, Oostra C, Delanghe J, Vande Walle J, Van Laere M, Oosterlinck W. Diagnosis and localization of a complicated urinary tract infection in neurogenic bladder disease by tubular proteinuria and serum prostate specific antigen. Spinal Cord. 1998;36(1):33-38. doi:10.1038/sj.sc.3100520
26. Drazer MW, Huo D, Eggener SE. National prostate cancer screening rates after the 2012 US Preventive Services Task Force recommendation discouraging prostate-specific antigen-based screening. J Clin Oncol. 2015;33(22):2416-2423. doi:10.1200/JCO.2015.61.6532
27. Sammon JD, Abdollah F, Choueiri TK, et al. Prostate-specific antigen screening after 2012 US Preventive Services Task Force recommendations. JAMA. 2015;314(19):2077-2079. doi:10.1001/jama.2015.7273
28. Jemal A, Fedewa SA, Ma J, et al. Prostate cancer incidence and PSA testing patterns in relation to USPSTF screening recommendations. JAMA. 2015;314(19):2054-2061. doi:10.1001/jama.2015.14905
29. Vetterlein MW, Dalela D, Sammon JD, et al. State-by-state variation in prostate-specific antigen screening trends following the 2011 United States Preventive Services Task Force panel update. Urology. 2018;112:56-65. doi:10.1016/j.urology.2017.08.055
FDA OKs combo therapy of niraparib, abiraterone acetate for prostate cancer
The Food and Drug Administration has approved niraparib and abiraterone acetate (Akeega, Janssen Pharmaceuticals) to treat BRCA-positive, metastatic castration-resistant prostate cancer in adult patients with deleterious or suspected deleterious disease, as determined by an FDA-approved test.
The FDA’s approval was based on findings from the phase 3 MAGNITUDE precision medicine study, a randomized, placebo-controlled trial with 423 patients, 225 (53%) of whom had BRCA gene mutations as determined using a tissue assay such as FoundationOne CDx.
Among the subgroup with a BRCA mutation, radiographic progression-free survival was a median of 16.6 months vs. 10.9 months (hazard ratio [HR], 0.53; 95% confidence interval [CI], 0.36-0.79; P = .0014). In this subgroup, an exploratory overall survival analysis demonstrated a median of 30.4 months vs. 28.6 months (HR, 0.79; 95% CI, 0.55-1.12), favoring the treatment arm.
Although the overall cohort (those with and without BRCA mutations) demonstrated a significant improvement in radiographic progression-free survival, the subgroup with non-BRCA homologous recombination repair mutations did not demonstrate a significant improvement in radiographic progression-free survival, which indicates that the benefit observed was “primarily attributed” to the results in the subgroup of patients with BRCA mutations, according to the FDA.
The safety profile of niraparib and abiraterone acetate plus prednisone was consistent with the known safety profile of each FDA-approved monotherapy. Serious adverse events occurred in 41% of patients in the treatment arm. These most often included musculoskeletal pain (44% vs. 42%), fatigue (43% vs. 30%), constipation (34% vs. 20%), hypertension (33% vs. 27%), and nausea (33% vs. 21%).
An adverse reaction led to permanent discontinuation of treatment in 15% of patients.
“As a physician, identifying patients with a worse prognosis is a priority, especially those whose cancers have a BRCA mutation,” principal investigator Kim Chi, MD, stated in the Janssen press release. “We prospectively designed the MAGNITUDE study to identify the subset of patients most likely to benefit from targeted treatment with AKEEGA and to help us understand how we can potentially achieve better health outcomes for patients.”
About 10%-15% of patients who develop metastatic castration-resistant prostate cancer have BRCA gene alterations, and those patients are more likely to have aggressive disease, poor outcomes, and shorter survival. Therefore, this new agent “brings an important treatment option to patients with prostate cancer as they consider their road ahead,” said Shelby Moneer, vice president of patient programs and education at ZERO Prostate Cancer.
The prescribing information lists the recommended dose at 200 mg niraparib and 1,000 mg abiraterone once daily in combination with 10 mg of prednisone daily until disease progression or unacceptable toxicity. Patients should also receive a gonadotropin-releasing hormone analog concurrently or should have had bilateral orchiectomy.
Health care professionals should report all serious adverse events suspected to be associated with the use of any medicine and device by using the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article appeared on Medscape.com.
The Food and Drug Administration has approved niraparib and abiraterone acetate (Akeega, Janssen Pharmaceuticals) to treat BRCA-positive, metastatic castration-resistant prostate cancer in adult patients with deleterious or suspected deleterious disease, as determined by an FDA-approved test.
The FDA’s approval was based on findings from the phase 3 MAGNITUDE precision medicine study, a randomized, placebo-controlled trial with 423 patients, 225 (53%) of whom had BRCA gene mutations as determined using a tissue assay such as FoundationOne CDx.
Among the subgroup with a BRCA mutation, radiographic progression-free survival was a median of 16.6 months vs. 10.9 months (hazard ratio [HR], 0.53; 95% confidence interval [CI], 0.36-0.79; P = .0014). In this subgroup, an exploratory overall survival analysis demonstrated a median of 30.4 months vs. 28.6 months (HR, 0.79; 95% CI, 0.55-1.12), favoring the treatment arm.
Although the overall cohort (those with and without BRCA mutations) demonstrated a significant improvement in radiographic progression-free survival, the subgroup with non-BRCA homologous recombination repair mutations did not demonstrate a significant improvement in radiographic progression-free survival, which indicates that the benefit observed was “primarily attributed” to the results in the subgroup of patients with BRCA mutations, according to the FDA.
The safety profile of niraparib and abiraterone acetate plus prednisone was consistent with the known safety profile of each FDA-approved monotherapy. Serious adverse events occurred in 41% of patients in the treatment arm. These most often included musculoskeletal pain (44% vs. 42%), fatigue (43% vs. 30%), constipation (34% vs. 20%), hypertension (33% vs. 27%), and nausea (33% vs. 21%).
An adverse reaction led to permanent discontinuation of treatment in 15% of patients.
“As a physician, identifying patients with a worse prognosis is a priority, especially those whose cancers have a BRCA mutation,” principal investigator Kim Chi, MD, stated in the Janssen press release. “We prospectively designed the MAGNITUDE study to identify the subset of patients most likely to benefit from targeted treatment with AKEEGA and to help us understand how we can potentially achieve better health outcomes for patients.”
About 10%-15% of patients who develop metastatic castration-resistant prostate cancer have BRCA gene alterations, and those patients are more likely to have aggressive disease, poor outcomes, and shorter survival. Therefore, this new agent “brings an important treatment option to patients with prostate cancer as they consider their road ahead,” said Shelby Moneer, vice president of patient programs and education at ZERO Prostate Cancer.
The prescribing information lists the recommended dose at 200 mg niraparib and 1,000 mg abiraterone once daily in combination with 10 mg of prednisone daily until disease progression or unacceptable toxicity. Patients should also receive a gonadotropin-releasing hormone analog concurrently or should have had bilateral orchiectomy.
Health care professionals should report all serious adverse events suspected to be associated with the use of any medicine and device by using the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article appeared on Medscape.com.
The Food and Drug Administration has approved niraparib and abiraterone acetate (Akeega, Janssen Pharmaceuticals) to treat BRCA-positive, metastatic castration-resistant prostate cancer in adult patients with deleterious or suspected deleterious disease, as determined by an FDA-approved test.
The FDA’s approval was based on findings from the phase 3 MAGNITUDE precision medicine study, a randomized, placebo-controlled trial with 423 patients, 225 (53%) of whom had BRCA gene mutations as determined using a tissue assay such as FoundationOne CDx.
Among the subgroup with a BRCA mutation, radiographic progression-free survival was a median of 16.6 months vs. 10.9 months (hazard ratio [HR], 0.53; 95% confidence interval [CI], 0.36-0.79; P = .0014). In this subgroup, an exploratory overall survival analysis demonstrated a median of 30.4 months vs. 28.6 months (HR, 0.79; 95% CI, 0.55-1.12), favoring the treatment arm.
Although the overall cohort (those with and without BRCA mutations) demonstrated a significant improvement in radiographic progression-free survival, the subgroup with non-BRCA homologous recombination repair mutations did not demonstrate a significant improvement in radiographic progression-free survival, which indicates that the benefit observed was “primarily attributed” to the results in the subgroup of patients with BRCA mutations, according to the FDA.
The safety profile of niraparib and abiraterone acetate plus prednisone was consistent with the known safety profile of each FDA-approved monotherapy. Serious adverse events occurred in 41% of patients in the treatment arm. These most often included musculoskeletal pain (44% vs. 42%), fatigue (43% vs. 30%), constipation (34% vs. 20%), hypertension (33% vs. 27%), and nausea (33% vs. 21%).
An adverse reaction led to permanent discontinuation of treatment in 15% of patients.
“As a physician, identifying patients with a worse prognosis is a priority, especially those whose cancers have a BRCA mutation,” principal investigator Kim Chi, MD, stated in the Janssen press release. “We prospectively designed the MAGNITUDE study to identify the subset of patients most likely to benefit from targeted treatment with AKEEGA and to help us understand how we can potentially achieve better health outcomes for patients.”
About 10%-15% of patients who develop metastatic castration-resistant prostate cancer have BRCA gene alterations, and those patients are more likely to have aggressive disease, poor outcomes, and shorter survival. Therefore, this new agent “brings an important treatment option to patients with prostate cancer as they consider their road ahead,” said Shelby Moneer, vice president of patient programs and education at ZERO Prostate Cancer.
The prescribing information lists the recommended dose at 200 mg niraparib and 1,000 mg abiraterone once daily in combination with 10 mg of prednisone daily until disease progression or unacceptable toxicity. Patients should also receive a gonadotropin-releasing hormone analog concurrently or should have had bilateral orchiectomy.
Health care professionals should report all serious adverse events suspected to be associated with the use of any medicine and device by using the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article appeared on Medscape.com.
MRI-guided SBRT cuts radiation toxicity in prostate cancer
TOPLINE
The use of magnetic resonance–guided daily adaptive stereotactic body radiotherapy for patients with prostate cancer reduces the risk of acute urinary side effects of grade 2 or higher by 44% and the risk of acute bowel side effects of grade 2 or higher by 60%, compared with standard CT-guided SBRT (CT‐SBRT).
METHODOLOGY
- With the use of magnetic resonance–guided daily adaptive SBRT, clinicians can customize radiation dosing to accommodate changes in prostate anatomy during treatment, which may also make SBRT safer and less toxic for patients.
- To determine whether this approach does reduce patient side effects, investigators ran a meta-analysis that included 29 studies with 2547 patients comparing the incidence of short-term, physician-assessed bowel and genitourinary side effects between the MRI-guided approach and standard CT-SBRT.
- The investigators reported no statistically significant differences in age, prescribed radiation doses, planning target volumes, or International Prostatism Symptom Scores between the two groups; the use of rectal spacers and the number of patients who received pelvic lymph node radiation were low in both.
- The average window for collecting acute toxicity data was 70 days in the MRI-guided investigations and 94 days in CT-SBRT investigations.
TAKEAWAY
- (odds ratio, 0.56; P = .04).
- The pooled estimate for grade 2 or higher gastrointestinal toxicity was 4% with the MRI approach versus 9% with CT-SBRT (OR, 0.40; P = .04).
- There were no differences in grade 3 or higher events, which were rare, between the groups.
- There was also no difference in toxicity among CT‐SBRT studies that used fiducial markers and those that did not.
IN PRACTICE
“These findings suggest that the technical advantages in precision of radiotherapy delivery afforded by [MRI-guided] SBRT translate to measurable clinical benefit,” the authors concluded. Potential reasons for the reduced risk of acute toxicity with the MRI-guided approach include “daily online adaptive planning, MRI‐based contouring that results in smaller treatment volumes, and MRI tracking, all of which may facilitate the precision and accuracy of treatment delivery.”
SOURCE
The study was led by Jonathan Leeman, MD, of the Dana-Farber Cancer Institute, Boston, and was published July 24 in Cancer.
LIMITATIONS
- The analysis did not account for differences in dosimetry, radiation planning, and toxicity management and assessment between the studies.
- Late toxicity and cancer control rates were not tracked and may have differed between the two approaches.
DISCLOSURES
- No external funding was reported.
- The investigators reported grants and consulting, personal, and other payments from Novartis, AstraZeneca, Janssen, and other companies.
A version of this article appeared on Medscape.com.
TOPLINE
The use of magnetic resonance–guided daily adaptive stereotactic body radiotherapy for patients with prostate cancer reduces the risk of acute urinary side effects of grade 2 or higher by 44% and the risk of acute bowel side effects of grade 2 or higher by 60%, compared with standard CT-guided SBRT (CT‐SBRT).
METHODOLOGY
- With the use of magnetic resonance–guided daily adaptive SBRT, clinicians can customize radiation dosing to accommodate changes in prostate anatomy during treatment, which may also make SBRT safer and less toxic for patients.
- To determine whether this approach does reduce patient side effects, investigators ran a meta-analysis that included 29 studies with 2547 patients comparing the incidence of short-term, physician-assessed bowel and genitourinary side effects between the MRI-guided approach and standard CT-SBRT.
- The investigators reported no statistically significant differences in age, prescribed radiation doses, planning target volumes, or International Prostatism Symptom Scores between the two groups; the use of rectal spacers and the number of patients who received pelvic lymph node radiation were low in both.
- The average window for collecting acute toxicity data was 70 days in the MRI-guided investigations and 94 days in CT-SBRT investigations.
TAKEAWAY
- (odds ratio, 0.56; P = .04).
- The pooled estimate for grade 2 or higher gastrointestinal toxicity was 4% with the MRI approach versus 9% with CT-SBRT (OR, 0.40; P = .04).
- There were no differences in grade 3 or higher events, which were rare, between the groups.
- There was also no difference in toxicity among CT‐SBRT studies that used fiducial markers and those that did not.
IN PRACTICE
“These findings suggest that the technical advantages in precision of radiotherapy delivery afforded by [MRI-guided] SBRT translate to measurable clinical benefit,” the authors concluded. Potential reasons for the reduced risk of acute toxicity with the MRI-guided approach include “daily online adaptive planning, MRI‐based contouring that results in smaller treatment volumes, and MRI tracking, all of which may facilitate the precision and accuracy of treatment delivery.”
SOURCE
The study was led by Jonathan Leeman, MD, of the Dana-Farber Cancer Institute, Boston, and was published July 24 in Cancer.
LIMITATIONS
- The analysis did not account for differences in dosimetry, radiation planning, and toxicity management and assessment between the studies.
- Late toxicity and cancer control rates were not tracked and may have differed between the two approaches.
DISCLOSURES
- No external funding was reported.
- The investigators reported grants and consulting, personal, and other payments from Novartis, AstraZeneca, Janssen, and other companies.
A version of this article appeared on Medscape.com.
TOPLINE
The use of magnetic resonance–guided daily adaptive stereotactic body radiotherapy for patients with prostate cancer reduces the risk of acute urinary side effects of grade 2 or higher by 44% and the risk of acute bowel side effects of grade 2 or higher by 60%, compared with standard CT-guided SBRT (CT‐SBRT).
METHODOLOGY
- With the use of magnetic resonance–guided daily adaptive SBRT, clinicians can customize radiation dosing to accommodate changes in prostate anatomy during treatment, which may also make SBRT safer and less toxic for patients.
- To determine whether this approach does reduce patient side effects, investigators ran a meta-analysis that included 29 studies with 2547 patients comparing the incidence of short-term, physician-assessed bowel and genitourinary side effects between the MRI-guided approach and standard CT-SBRT.
- The investigators reported no statistically significant differences in age, prescribed radiation doses, planning target volumes, or International Prostatism Symptom Scores between the two groups; the use of rectal spacers and the number of patients who received pelvic lymph node radiation were low in both.
- The average window for collecting acute toxicity data was 70 days in the MRI-guided investigations and 94 days in CT-SBRT investigations.
TAKEAWAY
- (odds ratio, 0.56; P = .04).
- The pooled estimate for grade 2 or higher gastrointestinal toxicity was 4% with the MRI approach versus 9% with CT-SBRT (OR, 0.40; P = .04).
- There were no differences in grade 3 or higher events, which were rare, between the groups.
- There was also no difference in toxicity among CT‐SBRT studies that used fiducial markers and those that did not.
IN PRACTICE
“These findings suggest that the technical advantages in precision of radiotherapy delivery afforded by [MRI-guided] SBRT translate to measurable clinical benefit,” the authors concluded. Potential reasons for the reduced risk of acute toxicity with the MRI-guided approach include “daily online adaptive planning, MRI‐based contouring that results in smaller treatment volumes, and MRI tracking, all of which may facilitate the precision and accuracy of treatment delivery.”
SOURCE
The study was led by Jonathan Leeman, MD, of the Dana-Farber Cancer Institute, Boston, and was published July 24 in Cancer.
LIMITATIONS
- The analysis did not account for differences in dosimetry, radiation planning, and toxicity management and assessment between the studies.
- Late toxicity and cancer control rates were not tracked and may have differed between the two approaches.
DISCLOSURES
- No external funding was reported.
- The investigators reported grants and consulting, personal, and other payments from Novartis, AstraZeneca, Janssen, and other companies.
A version of this article appeared on Medscape.com.
FROM CANCER
‘Treatment holiday’ in prostate cancer with tailored dosing
and improve patient outcomes, new research suggests.
The findings indicate that implementing a personalized dosing strategy with the radioligand therapy “allowed for treatment holidays in excellent responders, continuous 6-weekly treatments in moderate responders, and [allowed us] to consider changing or adding treatment in limited responders,” said study author Andrew Nguyen, MBBS, FRACP, AANMS, senior staff specialist in the department of theranostics and nuclear medicine at St. Vincent’s Hospital in Sydney.
The research was presented at the annual meeting of the Society of Nuclear Medicine and Molecular Imaging.
Although clinical trials have demonstrated that 177Lu-PSMA is an effective treatment for metastatic castration-resistant prostate cancer, the question remains: Can patient outcomes be improved through the use of biomarkers and by escalating or deescalating treatment as appropriate? asked Dr. Nguyen, who presented the findings at the meeting.
Clinical trials use standardized dosing intervals. Adjusting treatment intervals through the use of early-biomarker responses could give some patients a break from treatment and improve overall survival outcomes, Dr. Nguyen explained. For example, the 2021 REALITY study showed that overall survival was significantly better for patients who received 177Lu-PSMA plus standard care, compared with patients who received standard care alone (median, 15.3 vs. 11.3 months), and that overall survival was better among patients with early prostate-specific antigen (PSA) responses.
In the current study, Dr. Nguyen and colleagues used composite early biomarkers of PSA, imaging with 177Lu-PSMA SPECT, and diagnostic CT to guide a personalized dosing interval strategy for patients with metastatic castration-resistant prostate cancer receiving 177Lu-PSMA. The team evaluated progression-free survival and overall survival among these patients to determine whether personalizing dosing on the basis of early biomarker levels was associated with survival outcomes.
The cohort included 125 men who received six weekly doses of 177Lu-PSMA and who underwent imaging with 177Lu-SPECT/CT after each dose. After the second dose, investigators used the composite of PSA and 177Lu SPECT/CT response to determine which patients had a partial response, which had stable disease, and which had progressive disease.
The men were divided into three groups on the basis of their level of response. Group 1, which included 35% of participants, achieved a significant reduction in PSA levels and a partial response on 177Lu-SPECT. These patients were advised to discontinue treatment until PSA levels increased. This treatment holiday lasted a median of about 6 months.
Group 2, which represented 34% of the cohort, had stable or reduced PSA levels as well as stable disease on SPECT imaging. For these patients, the treatment regimen continued.
Group 3 demonstrated rising PSA levels and progressive disease on SPECT imaging. These men were offered an alternative therapy.
Overall, median PSA progression-free survival was 12.1 months in group 1, 6.1 months in group 2, and 2.6 months in group 3. Median overall survival was also significantly better among patients who showed early responses to therapy: 19.2 months in group 1, 13.2 months in group 2, and 11. 2 months in group 3.
Dr. Nguyen noted several limitations to the findings, including the study’s retrospective nature and the fact that some patients in group 1 chose not to resume further treatment after their PSA levels rose.
“Personalizing dosing intervals using early-response biomarkers with 177Lu-PSMA has the potential to achieve similar overall treatment responses to that published for continuous dosing, while allowing treatment holidays in responders and early crossover to potentially more effective therapies in nonresponders,” the authors conclude.
Given the effectiveness of this strategy, Dr. Nguyen says his team “now routinely uses these composite biomarkers when treating clinical patients.”
A version of this article appeared on Medscape.com.
and improve patient outcomes, new research suggests.
The findings indicate that implementing a personalized dosing strategy with the radioligand therapy “allowed for treatment holidays in excellent responders, continuous 6-weekly treatments in moderate responders, and [allowed us] to consider changing or adding treatment in limited responders,” said study author Andrew Nguyen, MBBS, FRACP, AANMS, senior staff specialist in the department of theranostics and nuclear medicine at St. Vincent’s Hospital in Sydney.
The research was presented at the annual meeting of the Society of Nuclear Medicine and Molecular Imaging.
Although clinical trials have demonstrated that 177Lu-PSMA is an effective treatment for metastatic castration-resistant prostate cancer, the question remains: Can patient outcomes be improved through the use of biomarkers and by escalating or deescalating treatment as appropriate? asked Dr. Nguyen, who presented the findings at the meeting.
Clinical trials use standardized dosing intervals. Adjusting treatment intervals through the use of early-biomarker responses could give some patients a break from treatment and improve overall survival outcomes, Dr. Nguyen explained. For example, the 2021 REALITY study showed that overall survival was significantly better for patients who received 177Lu-PSMA plus standard care, compared with patients who received standard care alone (median, 15.3 vs. 11.3 months), and that overall survival was better among patients with early prostate-specific antigen (PSA) responses.
In the current study, Dr. Nguyen and colleagues used composite early biomarkers of PSA, imaging with 177Lu-PSMA SPECT, and diagnostic CT to guide a personalized dosing interval strategy for patients with metastatic castration-resistant prostate cancer receiving 177Lu-PSMA. The team evaluated progression-free survival and overall survival among these patients to determine whether personalizing dosing on the basis of early biomarker levels was associated with survival outcomes.
The cohort included 125 men who received six weekly doses of 177Lu-PSMA and who underwent imaging with 177Lu-SPECT/CT after each dose. After the second dose, investigators used the composite of PSA and 177Lu SPECT/CT response to determine which patients had a partial response, which had stable disease, and which had progressive disease.
The men were divided into three groups on the basis of their level of response. Group 1, which included 35% of participants, achieved a significant reduction in PSA levels and a partial response on 177Lu-SPECT. These patients were advised to discontinue treatment until PSA levels increased. This treatment holiday lasted a median of about 6 months.
Group 2, which represented 34% of the cohort, had stable or reduced PSA levels as well as stable disease on SPECT imaging. For these patients, the treatment regimen continued.
Group 3 demonstrated rising PSA levels and progressive disease on SPECT imaging. These men were offered an alternative therapy.
Overall, median PSA progression-free survival was 12.1 months in group 1, 6.1 months in group 2, and 2.6 months in group 3. Median overall survival was also significantly better among patients who showed early responses to therapy: 19.2 months in group 1, 13.2 months in group 2, and 11. 2 months in group 3.
Dr. Nguyen noted several limitations to the findings, including the study’s retrospective nature and the fact that some patients in group 1 chose not to resume further treatment after their PSA levels rose.
“Personalizing dosing intervals using early-response biomarkers with 177Lu-PSMA has the potential to achieve similar overall treatment responses to that published for continuous dosing, while allowing treatment holidays in responders and early crossover to potentially more effective therapies in nonresponders,” the authors conclude.
Given the effectiveness of this strategy, Dr. Nguyen says his team “now routinely uses these composite biomarkers when treating clinical patients.”
A version of this article appeared on Medscape.com.
and improve patient outcomes, new research suggests.
The findings indicate that implementing a personalized dosing strategy with the radioligand therapy “allowed for treatment holidays in excellent responders, continuous 6-weekly treatments in moderate responders, and [allowed us] to consider changing or adding treatment in limited responders,” said study author Andrew Nguyen, MBBS, FRACP, AANMS, senior staff specialist in the department of theranostics and nuclear medicine at St. Vincent’s Hospital in Sydney.
The research was presented at the annual meeting of the Society of Nuclear Medicine and Molecular Imaging.
Although clinical trials have demonstrated that 177Lu-PSMA is an effective treatment for metastatic castration-resistant prostate cancer, the question remains: Can patient outcomes be improved through the use of biomarkers and by escalating or deescalating treatment as appropriate? asked Dr. Nguyen, who presented the findings at the meeting.
Clinical trials use standardized dosing intervals. Adjusting treatment intervals through the use of early-biomarker responses could give some patients a break from treatment and improve overall survival outcomes, Dr. Nguyen explained. For example, the 2021 REALITY study showed that overall survival was significantly better for patients who received 177Lu-PSMA plus standard care, compared with patients who received standard care alone (median, 15.3 vs. 11.3 months), and that overall survival was better among patients with early prostate-specific antigen (PSA) responses.
In the current study, Dr. Nguyen and colleagues used composite early biomarkers of PSA, imaging with 177Lu-PSMA SPECT, and diagnostic CT to guide a personalized dosing interval strategy for patients with metastatic castration-resistant prostate cancer receiving 177Lu-PSMA. The team evaluated progression-free survival and overall survival among these patients to determine whether personalizing dosing on the basis of early biomarker levels was associated with survival outcomes.
The cohort included 125 men who received six weekly doses of 177Lu-PSMA and who underwent imaging with 177Lu-SPECT/CT after each dose. After the second dose, investigators used the composite of PSA and 177Lu SPECT/CT response to determine which patients had a partial response, which had stable disease, and which had progressive disease.
The men were divided into three groups on the basis of their level of response. Group 1, which included 35% of participants, achieved a significant reduction in PSA levels and a partial response on 177Lu-SPECT. These patients were advised to discontinue treatment until PSA levels increased. This treatment holiday lasted a median of about 6 months.
Group 2, which represented 34% of the cohort, had stable or reduced PSA levels as well as stable disease on SPECT imaging. For these patients, the treatment regimen continued.
Group 3 demonstrated rising PSA levels and progressive disease on SPECT imaging. These men were offered an alternative therapy.
Overall, median PSA progression-free survival was 12.1 months in group 1, 6.1 months in group 2, and 2.6 months in group 3. Median overall survival was also significantly better among patients who showed early responses to therapy: 19.2 months in group 1, 13.2 months in group 2, and 11. 2 months in group 3.
Dr. Nguyen noted several limitations to the findings, including the study’s retrospective nature and the fact that some patients in group 1 chose not to resume further treatment after their PSA levels rose.
“Personalizing dosing intervals using early-response biomarkers with 177Lu-PSMA has the potential to achieve similar overall treatment responses to that published for continuous dosing, while allowing treatment holidays in responders and early crossover to potentially more effective therapies in nonresponders,” the authors conclude.
Given the effectiveness of this strategy, Dr. Nguyen says his team “now routinely uses these composite biomarkers when treating clinical patients.”
A version of this article appeared on Medscape.com.
FROM SNMMI 2023
Cancer Data Trends 2023
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
Federal Practitioner and the Association of VA Hematology/Oncology (AVAHO) present the 2023 edition of Cancer Data Trends (click to view the digital edition). This special issue provides updates on some of the top cancers and related concerns affecting veterans through original infographics and visual storytelling.
In this issue:
- COVID-19 Outcomes in Veterans With Hematologic Cancers
- Promising New Approaches for Testicular and Prostate Cancer
- Screening Guideline Updates and New Treatments in Colon Cancer
- Exposure-Related Cancers: A Look at the PACT Act
- New Classifications and Emerging Treatments in Brain Cancer
- Gender Disparity in Breast Cancer Among US Veterans
- Lung Cancer Screening in Veterans
- Necessary Updates to Skin Cancer Risk Stratification
- Innovation in Cancer Treatment
Promising New Approaches for Testicular and Prostate Cancer
- Risk factors for testicular cancer. American Cancer Society. Updated May 17, 2018. Accessed December 15, 2022. https://www.cancer.org/cancer/testicular-cancer/causes-risks-prevention/risk-factors.html
- Chovanec M, Cheng L. BMJ. 2022;379:e070499. doi:10.1136/bmj-2022-070499
- Tavares NT et al. J Pathol. 2022. doi:10.1002/path.6037
- Bryant AK et al. JAMA Oncol. 2022;e224319. doi:10.1001/jamaoncol.2022.4319
- Kabasakal L et al. Nucl Med Commun. 2017;38(2):149-155. doi:10.1097/MNM.0000000000000617
- Sartor O et al; VISION Investigators. N Engl J Med. 2021;385(12):1091-1103. doi:10.1056/NEJMoa2107322
- Rowe SP et al. Annu Rev Med. 2019;70:461-477. doi:10.1146/annurev-med-062117-073027
- Pomykala KL et al. Eur Urol Oncol. 2022;S2588-9311(22)00177-8. doi:10.1016/j.euo.2022.10.007
- Keam SJ. Mol Diagn Ther. 2022;26(4):467-475. doi:10.1007/s40291-022-00594-2
- Lovejoy LA et al. Mil Med. 2022:usac297. doi:10.1093/milmed/usac297
- Smith ZL et al. Med Clin North Am. 2018;102(2):251-264. doi:10.1016/j.mcna.2017.10.003
- Hohnloser JH et al. Eur J Med Res.1996;1(11):509-514.
- Johns Hopkins Medicine website. Testicular Cancer tumor Markers. Accessed December 2022. https://www.hopkinsmedicine.org/health/conditions-and-diseases/testicular-cancer/testicular-cancer-tumor-markers
- Webber BJ et al. J Occup Environ Med. 2022;64(1):71-78. doi:10.1097/JOM.0000000000002353
- Risk factors for testicular cancer. American Cancer Society. Updated May 17, 2018. Accessed December 15, 2022. https://www.cancer.org/cancer/testicular-cancer/causes-risks-prevention/risk-factors.html
- Chovanec M, Cheng L. BMJ. 2022;379:e070499. doi:10.1136/bmj-2022-070499
- Tavares NT et al. J Pathol. 2022. doi:10.1002/path.6037
- Bryant AK et al. JAMA Oncol. 2022;e224319. doi:10.1001/jamaoncol.2022.4319
- Kabasakal L et al. Nucl Med Commun. 2017;38(2):149-155. doi:10.1097/MNM.0000000000000617
- Sartor O et al; VISION Investigators. N Engl J Med. 2021;385(12):1091-1103. doi:10.1056/NEJMoa2107322
- Rowe SP et al. Annu Rev Med. 2019;70:461-477. doi:10.1146/annurev-med-062117-073027
- Pomykala KL et al. Eur Urol Oncol. 2022;S2588-9311(22)00177-8. doi:10.1016/j.euo.2022.10.007
- Keam SJ. Mol Diagn Ther. 2022;26(4):467-475. doi:10.1007/s40291-022-00594-2
- Lovejoy LA et al. Mil Med. 2022:usac297. doi:10.1093/milmed/usac297
- Smith ZL et al. Med Clin North Am. 2018;102(2):251-264. doi:10.1016/j.mcna.2017.10.003
- Hohnloser JH et al. Eur J Med Res.1996;1(11):509-514.
- Johns Hopkins Medicine website. Testicular Cancer tumor Markers. Accessed December 2022. https://www.hopkinsmedicine.org/health/conditions-and-diseases/testicular-cancer/testicular-cancer-tumor-markers
- Webber BJ et al. J Occup Environ Med. 2022;64(1):71-78. doi:10.1097/JOM.0000000000002353
- Risk factors for testicular cancer. American Cancer Society. Updated May 17, 2018. Accessed December 15, 2022. https://www.cancer.org/cancer/testicular-cancer/causes-risks-prevention/risk-factors.html
- Chovanec M, Cheng L. BMJ. 2022;379:e070499. doi:10.1136/bmj-2022-070499
- Tavares NT et al. J Pathol. 2022. doi:10.1002/path.6037
- Bryant AK et al. JAMA Oncol. 2022;e224319. doi:10.1001/jamaoncol.2022.4319
- Kabasakal L et al. Nucl Med Commun. 2017;38(2):149-155. doi:10.1097/MNM.0000000000000617
- Sartor O et al; VISION Investigators. N Engl J Med. 2021;385(12):1091-1103. doi:10.1056/NEJMoa2107322
- Rowe SP et al. Annu Rev Med. 2019;70:461-477. doi:10.1146/annurev-med-062117-073027
- Pomykala KL et al. Eur Urol Oncol. 2022;S2588-9311(22)00177-8. doi:10.1016/j.euo.2022.10.007
- Keam SJ. Mol Diagn Ther. 2022;26(4):467-475. doi:10.1007/s40291-022-00594-2
- Lovejoy LA et al. Mil Med. 2022:usac297. doi:10.1093/milmed/usac297
- Smith ZL et al. Med Clin North Am. 2018;102(2):251-264. doi:10.1016/j.mcna.2017.10.003
- Hohnloser JH et al. Eur J Med Res.1996;1(11):509-514.
- Johns Hopkins Medicine website. Testicular Cancer tumor Markers. Accessed December 2022. https://www.hopkinsmedicine.org/health/conditions-and-diseases/testicular-cancer/testicular-cancer-tumor-markers
- Webber BJ et al. J Occup Environ Med. 2022;64(1):71-78. doi:10.1097/JOM.0000000000002353
Risk of falls seen with newer antiandrogens for prostate cancer
Second-generation antiandrogens (AAs) – abiraterone, apalutamide, darolutamide, and enzalutamide – are a cornerstone of modern prostate cancer treatment, improving outcomes and survival.
However, they carry a significant caveat, according to a new meta-analysis of 12 clinical trials with over 13,000 patients.
the authors reported.
These findings carry “important public health indications” because use of second-generation AAs, currently first-line treatment for advanced and castration-resistant prostate cancer, is expanding with new indications, meaning that the pool of men at risk for such problems is large and growing, the team wrote.
The take-home message is that the findings give men – and the physicians who counsel them – a fuller idea of what to expect when considering using the agents, the researchers comment. This information is key at a time when so much of prostate cancer treatment involves carefully weighing the risks and benefits, they added.
The study was published in JAMA Oncology. It was conducted by a team of researchers from the University of Texas MD Anderson Cancer Center, Houston, and was led by Malgorzata Nowakowska, a medical student at Baylor College of Medicine, Houston.
Two prostate cancer specialists agreed and gave an example to bring the point home in an accompanying editorial.
The risk-benefit ratio of adding a second-generation AA to treatment may be different for a patient who wants to stay alert and sharp to keep a complex job “versus someone whose primary goal is to see their young children graduate high school,” Alexandra Sokolova, MD, of the Oregon Health and Science University, Portland, and Julie Graff, MD, of the VA Portland (Ore.) Health Care System, wrote in their editorial.
The study fills a “critical gap” when it comes to counseling men about the drugs and will help guide discussions, they said.
The investigators said their study also highlights the need for additional research to identify who is most at risk for the side effects and the best way to prevent and treat them. “Interventions currently under investigation include donepezil, methylphenidate, low-fat diet, acupuncture, martial arts, and high-intensity exercise, among many others,” Ms. Nowakowska and colleagues noted.
Study details
The 12 trials in the meta-analysis, which compared second-generation AAs with placebo, were conducted from 2008 to 2021. These trials were multinational investigations that included patients with metastatic disease as well as those with nonmetastatic disease. The median age across the studies ranged from 67 to 74 years, and trial follow-up ranged from 3.9 to 48 months.
The rates of adverse cognitive effects and attention disorders and disturbances ranged from 2% to 8% among patients who received second-generation AAs versus 2%-3% among those who received placebo, a more than doubling of the risk of cognitive toxic effects (P = .002).
Fatigue of any grade was reported in 5%-45% of participants taking second-generation AAs versus 2%-42% of patients taking placebos, which translates to a 34% higher risk (P < .001).
The use of AAs was associated with an 87% increase in the risk of falls in comparison with placebo, regardless of severity. For falls of grade 3 or higher that required hospitalization or invasive treatment, the increase in risk with second-generation AAs was 72% (P = .05).
The findings were consistent for cognitive toxicity and fatigue in studies that included traditional hormone therapy in both the treatment and control arms. Increased age was associated with a greater risk of fatigue.
Study limits include the fact that it was not known how long patients were taking the drugs before they encountered problems. In addition, the findings were not broken down with respect to medication, so it’s unknown whether such problems are worse with some second-generation AAs than with others.
The editorialists noted that real-world patients tend to be older and sicker than patients in trials, so the risk of falls, fatigue, and cognition problems might be higher among everyday patients.
The study was funded by the National Institutes of Health and others. The investigators disclosed no relevant financial relationships. Dr. Sokolova has received personal fees from Lantheus and travel grants from AstraZeneca. Dr. Graff has received nonfinancial support from Janssen, Pfizer/Astellas, and Sanofi.
A version of this article first appeared on Medscape.com.
Second-generation antiandrogens (AAs) – abiraterone, apalutamide, darolutamide, and enzalutamide – are a cornerstone of modern prostate cancer treatment, improving outcomes and survival.
However, they carry a significant caveat, according to a new meta-analysis of 12 clinical trials with over 13,000 patients.
the authors reported.
These findings carry “important public health indications” because use of second-generation AAs, currently first-line treatment for advanced and castration-resistant prostate cancer, is expanding with new indications, meaning that the pool of men at risk for such problems is large and growing, the team wrote.
The take-home message is that the findings give men – and the physicians who counsel them – a fuller idea of what to expect when considering using the agents, the researchers comment. This information is key at a time when so much of prostate cancer treatment involves carefully weighing the risks and benefits, they added.
The study was published in JAMA Oncology. It was conducted by a team of researchers from the University of Texas MD Anderson Cancer Center, Houston, and was led by Malgorzata Nowakowska, a medical student at Baylor College of Medicine, Houston.
Two prostate cancer specialists agreed and gave an example to bring the point home in an accompanying editorial.
The risk-benefit ratio of adding a second-generation AA to treatment may be different for a patient who wants to stay alert and sharp to keep a complex job “versus someone whose primary goal is to see their young children graduate high school,” Alexandra Sokolova, MD, of the Oregon Health and Science University, Portland, and Julie Graff, MD, of the VA Portland (Ore.) Health Care System, wrote in their editorial.
The study fills a “critical gap” when it comes to counseling men about the drugs and will help guide discussions, they said.
The investigators said their study also highlights the need for additional research to identify who is most at risk for the side effects and the best way to prevent and treat them. “Interventions currently under investigation include donepezil, methylphenidate, low-fat diet, acupuncture, martial arts, and high-intensity exercise, among many others,” Ms. Nowakowska and colleagues noted.
Study details
The 12 trials in the meta-analysis, which compared second-generation AAs with placebo, were conducted from 2008 to 2021. These trials were multinational investigations that included patients with metastatic disease as well as those with nonmetastatic disease. The median age across the studies ranged from 67 to 74 years, and trial follow-up ranged from 3.9 to 48 months.
The rates of adverse cognitive effects and attention disorders and disturbances ranged from 2% to 8% among patients who received second-generation AAs versus 2%-3% among those who received placebo, a more than doubling of the risk of cognitive toxic effects (P = .002).
Fatigue of any grade was reported in 5%-45% of participants taking second-generation AAs versus 2%-42% of patients taking placebos, which translates to a 34% higher risk (P < .001).
The use of AAs was associated with an 87% increase in the risk of falls in comparison with placebo, regardless of severity. For falls of grade 3 or higher that required hospitalization or invasive treatment, the increase in risk with second-generation AAs was 72% (P = .05).
The findings were consistent for cognitive toxicity and fatigue in studies that included traditional hormone therapy in both the treatment and control arms. Increased age was associated with a greater risk of fatigue.
Study limits include the fact that it was not known how long patients were taking the drugs before they encountered problems. In addition, the findings were not broken down with respect to medication, so it’s unknown whether such problems are worse with some second-generation AAs than with others.
The editorialists noted that real-world patients tend to be older and sicker than patients in trials, so the risk of falls, fatigue, and cognition problems might be higher among everyday patients.
The study was funded by the National Institutes of Health and others. The investigators disclosed no relevant financial relationships. Dr. Sokolova has received personal fees from Lantheus and travel grants from AstraZeneca. Dr. Graff has received nonfinancial support from Janssen, Pfizer/Astellas, and Sanofi.
A version of this article first appeared on Medscape.com.
Second-generation antiandrogens (AAs) – abiraterone, apalutamide, darolutamide, and enzalutamide – are a cornerstone of modern prostate cancer treatment, improving outcomes and survival.
However, they carry a significant caveat, according to a new meta-analysis of 12 clinical trials with over 13,000 patients.
the authors reported.
These findings carry “important public health indications” because use of second-generation AAs, currently first-line treatment for advanced and castration-resistant prostate cancer, is expanding with new indications, meaning that the pool of men at risk for such problems is large and growing, the team wrote.
The take-home message is that the findings give men – and the physicians who counsel them – a fuller idea of what to expect when considering using the agents, the researchers comment. This information is key at a time when so much of prostate cancer treatment involves carefully weighing the risks and benefits, they added.
The study was published in JAMA Oncology. It was conducted by a team of researchers from the University of Texas MD Anderson Cancer Center, Houston, and was led by Malgorzata Nowakowska, a medical student at Baylor College of Medicine, Houston.
Two prostate cancer specialists agreed and gave an example to bring the point home in an accompanying editorial.
The risk-benefit ratio of adding a second-generation AA to treatment may be different for a patient who wants to stay alert and sharp to keep a complex job “versus someone whose primary goal is to see their young children graduate high school,” Alexandra Sokolova, MD, of the Oregon Health and Science University, Portland, and Julie Graff, MD, of the VA Portland (Ore.) Health Care System, wrote in their editorial.
The study fills a “critical gap” when it comes to counseling men about the drugs and will help guide discussions, they said.
The investigators said their study also highlights the need for additional research to identify who is most at risk for the side effects and the best way to prevent and treat them. “Interventions currently under investigation include donepezil, methylphenidate, low-fat diet, acupuncture, martial arts, and high-intensity exercise, among many others,” Ms. Nowakowska and colleagues noted.
Study details
The 12 trials in the meta-analysis, which compared second-generation AAs with placebo, were conducted from 2008 to 2021. These trials were multinational investigations that included patients with metastatic disease as well as those with nonmetastatic disease. The median age across the studies ranged from 67 to 74 years, and trial follow-up ranged from 3.9 to 48 months.
The rates of adverse cognitive effects and attention disorders and disturbances ranged from 2% to 8% among patients who received second-generation AAs versus 2%-3% among those who received placebo, a more than doubling of the risk of cognitive toxic effects (P = .002).
Fatigue of any grade was reported in 5%-45% of participants taking second-generation AAs versus 2%-42% of patients taking placebos, which translates to a 34% higher risk (P < .001).
The use of AAs was associated with an 87% increase in the risk of falls in comparison with placebo, regardless of severity. For falls of grade 3 or higher that required hospitalization or invasive treatment, the increase in risk with second-generation AAs was 72% (P = .05).
The findings were consistent for cognitive toxicity and fatigue in studies that included traditional hormone therapy in both the treatment and control arms. Increased age was associated with a greater risk of fatigue.
Study limits include the fact that it was not known how long patients were taking the drugs before they encountered problems. In addition, the findings were not broken down with respect to medication, so it’s unknown whether such problems are worse with some second-generation AAs than with others.
The editorialists noted that real-world patients tend to be older and sicker than patients in trials, so the risk of falls, fatigue, and cognition problems might be higher among everyday patients.
The study was funded by the National Institutes of Health and others. The investigators disclosed no relevant financial relationships. Dr. Sokolova has received personal fees from Lantheus and travel grants from AstraZeneca. Dr. Graff has received nonfinancial support from Janssen, Pfizer/Astellas, and Sanofi.
A version of this article first appeared on Medscape.com.
FROM JAMA ONCOLOGY
COVID can mimic prostate cancer symptoms
This patient has a strong likelihood of aggressive prostate cancer, right? If that same patient also presents with severe, burning bone pain with no precipitating trauma to the area and rest and over-the-counter painkillers are not helping, you’d think, “check for metastases,” right?
That patient was me in late January 2023.
As a research scientist member of the American Urological Association, I knew enough to know I had to consult my urologist ASAP.
With the above symptoms, I’ll admit I was scared. Fortunately, if that’s the right word, I was no stranger to a rapid, dramatic spike in PSA. In 2021 I was temporarily living in a new city, and I wanted to form a relationship with a good local urologist. The urologist that I was referred to gave me a thorough consultation, including a vigorous digital rectal exam (DRE) and sent me across the street for a blood draw.
To my shock, my PSA had spiked over 2 points, to 9.9 from 7.8 a few months earlier. I freaked. Had my 3-cm tumor burst out into an aggressive cancer? Research on PubMed provided an array of studies showing what could cause PSA to suddenly rise, including a DRE performed 72 hours before the blood draw.1 A week later, my PSA was back down to its normal 7.6.
But in January 2023, I had none of those previously reported experiences that could suddenly trigger a spike in PSA, like a DRE or riding on a thin bicycle seat for a few hours before the lab visit.
The COVID effect
I went back to PubMed and found a new circumstance that could cause a surge in PSA: COVID-19. A recent study2 of 91 men with benign prostatic hypertrophy by researchers in Turkey found that PSA spiked from 0 to 5 points during the COVID infection period and up to 2 points higher 3 months after the infection had cleared. I had tested positive for COVID-19 in mid-December 2022, 4 weeks before my 9.9 PSA reading.
Using Google translate, I communicated with the team in Turkey and found out that the PSA spike can last up to 6 months.
That study helps explain why my PSA dropped over 1.5 points to 8.5 just 2 weeks after the 9.9 reading, with the expectation that it would return to its previous normal of 7.8 within 6 months of infection with SARS-CoV-2. To be safe, my urologist scheduled another PSA test in May, along with an updated multiparametric MRI, which may be followed by an in-bore MRI-guided biopsy of the 3-cm tumor if the mass has enlarged.
COVID-19 pain
What about my burning bone pain in my upper right humerus and right rotator cuff that was not precipitated by trauma or strain? A radiograph found no evidence of metastasis, thank goodness. And my research showed that several studies3 have found that COVID-19 can cause burning musculoskeletal pain, including enthesopathy, which is what I had per the radiology report. So my PSA spike and searing pain were likely consequences of the infection.
To avoid the risk for a gross misdiagnosis after a radical spike in PSA, the informed urologist should ask the patient if he has had COVID-19 in the previous 6 months. Overlooking that question could lead to the wrong diagnostic decisions about a rapid jump in PSA or unexplained bone pain.
References
1. Bossens MM et al. Eur J Cancer. 1995;31A:682-5.
2. Cinislioglu AE et al. Urology. 2022;159:16-21.
3. Ciaffi J et al. Joint Bone Spine. 2021;88:105158.
Dr. Keller is founder of the Keller Research Institute, Jacksonville, Fla. He reported serving as a research scientist for the American Urological Association, serving on the advisory board of Active Surveillance Patient’s International, and serving on the boards of numerous nonprofit organizations.
A version of this article first appeared on Medscape.com.
This patient has a strong likelihood of aggressive prostate cancer, right? If that same patient also presents with severe, burning bone pain with no precipitating trauma to the area and rest and over-the-counter painkillers are not helping, you’d think, “check for metastases,” right?
That patient was me in late January 2023.
As a research scientist member of the American Urological Association, I knew enough to know I had to consult my urologist ASAP.
With the above symptoms, I’ll admit I was scared. Fortunately, if that’s the right word, I was no stranger to a rapid, dramatic spike in PSA. In 2021 I was temporarily living in a new city, and I wanted to form a relationship with a good local urologist. The urologist that I was referred to gave me a thorough consultation, including a vigorous digital rectal exam (DRE) and sent me across the street for a blood draw.
To my shock, my PSA had spiked over 2 points, to 9.9 from 7.8 a few months earlier. I freaked. Had my 3-cm tumor burst out into an aggressive cancer? Research on PubMed provided an array of studies showing what could cause PSA to suddenly rise, including a DRE performed 72 hours before the blood draw.1 A week later, my PSA was back down to its normal 7.6.
But in January 2023, I had none of those previously reported experiences that could suddenly trigger a spike in PSA, like a DRE or riding on a thin bicycle seat for a few hours before the lab visit.
The COVID effect
I went back to PubMed and found a new circumstance that could cause a surge in PSA: COVID-19. A recent study2 of 91 men with benign prostatic hypertrophy by researchers in Turkey found that PSA spiked from 0 to 5 points during the COVID infection period and up to 2 points higher 3 months after the infection had cleared. I had tested positive for COVID-19 in mid-December 2022, 4 weeks before my 9.9 PSA reading.
Using Google translate, I communicated with the team in Turkey and found out that the PSA spike can last up to 6 months.
That study helps explain why my PSA dropped over 1.5 points to 8.5 just 2 weeks after the 9.9 reading, with the expectation that it would return to its previous normal of 7.8 within 6 months of infection with SARS-CoV-2. To be safe, my urologist scheduled another PSA test in May, along with an updated multiparametric MRI, which may be followed by an in-bore MRI-guided biopsy of the 3-cm tumor if the mass has enlarged.
COVID-19 pain
What about my burning bone pain in my upper right humerus and right rotator cuff that was not precipitated by trauma or strain? A radiograph found no evidence of metastasis, thank goodness. And my research showed that several studies3 have found that COVID-19 can cause burning musculoskeletal pain, including enthesopathy, which is what I had per the radiology report. So my PSA spike and searing pain were likely consequences of the infection.
To avoid the risk for a gross misdiagnosis after a radical spike in PSA, the informed urologist should ask the patient if he has had COVID-19 in the previous 6 months. Overlooking that question could lead to the wrong diagnostic decisions about a rapid jump in PSA or unexplained bone pain.
References
1. Bossens MM et al. Eur J Cancer. 1995;31A:682-5.
2. Cinislioglu AE et al. Urology. 2022;159:16-21.
3. Ciaffi J et al. Joint Bone Spine. 2021;88:105158.
Dr. Keller is founder of the Keller Research Institute, Jacksonville, Fla. He reported serving as a research scientist for the American Urological Association, serving on the advisory board of Active Surveillance Patient’s International, and serving on the boards of numerous nonprofit organizations.
A version of this article first appeared on Medscape.com.
This patient has a strong likelihood of aggressive prostate cancer, right? If that same patient also presents with severe, burning bone pain with no precipitating trauma to the area and rest and over-the-counter painkillers are not helping, you’d think, “check for metastases,” right?
That patient was me in late January 2023.
As a research scientist member of the American Urological Association, I knew enough to know I had to consult my urologist ASAP.
With the above symptoms, I’ll admit I was scared. Fortunately, if that’s the right word, I was no stranger to a rapid, dramatic spike in PSA. In 2021 I was temporarily living in a new city, and I wanted to form a relationship with a good local urologist. The urologist that I was referred to gave me a thorough consultation, including a vigorous digital rectal exam (DRE) and sent me across the street for a blood draw.
To my shock, my PSA had spiked over 2 points, to 9.9 from 7.8 a few months earlier. I freaked. Had my 3-cm tumor burst out into an aggressive cancer? Research on PubMed provided an array of studies showing what could cause PSA to suddenly rise, including a DRE performed 72 hours before the blood draw.1 A week later, my PSA was back down to its normal 7.6.
But in January 2023, I had none of those previously reported experiences that could suddenly trigger a spike in PSA, like a DRE or riding on a thin bicycle seat for a few hours before the lab visit.
The COVID effect
I went back to PubMed and found a new circumstance that could cause a surge in PSA: COVID-19. A recent study2 of 91 men with benign prostatic hypertrophy by researchers in Turkey found that PSA spiked from 0 to 5 points during the COVID infection period and up to 2 points higher 3 months after the infection had cleared. I had tested positive for COVID-19 in mid-December 2022, 4 weeks before my 9.9 PSA reading.
Using Google translate, I communicated with the team in Turkey and found out that the PSA spike can last up to 6 months.
That study helps explain why my PSA dropped over 1.5 points to 8.5 just 2 weeks after the 9.9 reading, with the expectation that it would return to its previous normal of 7.8 within 6 months of infection with SARS-CoV-2. To be safe, my urologist scheduled another PSA test in May, along with an updated multiparametric MRI, which may be followed by an in-bore MRI-guided biopsy of the 3-cm tumor if the mass has enlarged.
COVID-19 pain
What about my burning bone pain in my upper right humerus and right rotator cuff that was not precipitated by trauma or strain? A radiograph found no evidence of metastasis, thank goodness. And my research showed that several studies3 have found that COVID-19 can cause burning musculoskeletal pain, including enthesopathy, which is what I had per the radiology report. So my PSA spike and searing pain were likely consequences of the infection.
To avoid the risk for a gross misdiagnosis after a radical spike in PSA, the informed urologist should ask the patient if he has had COVID-19 in the previous 6 months. Overlooking that question could lead to the wrong diagnostic decisions about a rapid jump in PSA or unexplained bone pain.
References
1. Bossens MM et al. Eur J Cancer. 1995;31A:682-5.
2. Cinislioglu AE et al. Urology. 2022;159:16-21.
3. Ciaffi J et al. Joint Bone Spine. 2021;88:105158.
Dr. Keller is founder of the Keller Research Institute, Jacksonville, Fla. He reported serving as a research scientist for the American Urological Association, serving on the advisory board of Active Surveillance Patient’s International, and serving on the boards of numerous nonprofit organizations.
A version of this article first appeared on Medscape.com.
Reports of dysuria and nocturia
The history and findings in this case are suggestive of small cell carcinoma of the prostate (SCCP).
SCCP is a rare and aggressive cancer that comprises 1%–5% of all prostate cancers (if mixed cases with adenocarcinoma are included). Similar to small cell carcinoma of the lung or other small cell primaries, SCCP is characterized by a primary tumor of the prostate gland that expresses small cell morphology and high-grade features, including minimal cytoplasm, nuclear molding, fine chromatin pattern, extensive tumor necrosis and apoptosis, variable tumor giant cells, and a high mitotic rate. Patients often have disproportionally low PSA levels despite having large metastatic burden and visceral disease. Pathologic diagnosis is made on the basis of prostate biopsy using characteristics of small cell tumors and immunohistochemical staining for neuroendocrine markers, such as CD56, chromogranin A, synaptophysin, and neuron-specific enolase.
SCCP arises de novo in approximately 50% of cases; it also occurs in patients with previous or concomitant prostate adenocarcinoma. Patients are often symptomatic at diagnosis because of the extent of the tumor. The aggressive nature and high proliferation rate associated with SCCP result in an increased risk for lytic or blastic bone, visceral, and brain metastases. In addition, paraneoplastic syndromes (eg, the syndrome of inappropriate antidiuretic hormone secretion, Cushing syndrome, and hypercalcemia) frequently occur as a result of the release of peptides.
SCCP metastasizes early in its course and is associated with a poor prognosis. It has a median survival of < 1 year. Fluorodeoxyglucose PET-CT are useful for staging and monitoring treatment response; in addition, given the disease's predilection for brain metastases, MRI of the brain should be considered.
The optimal treatment for patients with metastatic SCCP has not yet been determined. Localized SCCP is treated aggressively, typically with a multimodality approach involving chemotherapy with concurrent or consolidative radiotherapy.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), platinum-based combination chemotherapy (cisplatin-etoposide, carboplatin-etoposide, docetaxel-carboplatin, cabazitaxel-carboplatin) is the first-line approach for patients with metastatic disease.
Physicians are also advised to consult the NCCN guidelines for small cell lung cancer because the behavior of SCCP is similar to that of small cell carcinoma of the lung. Immunotherapy with pembrolizumab may be used for platinum-resistant extrapulmonary small cell carcinoma. However, sipuleucel-T is not recommended for patients with SCCP.
Chad R. Tracy, MD, Professor; Director, Minimally Invasive Surgery, Department of Urology, University of Iowa Hospitals and Clinics, Iowa City, Iowa
Chad R. Tracy, MD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: CVICO Medical Solutions.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are suggestive of small cell carcinoma of the prostate (SCCP).
SCCP is a rare and aggressive cancer that comprises 1%–5% of all prostate cancers (if mixed cases with adenocarcinoma are included). Similar to small cell carcinoma of the lung or other small cell primaries, SCCP is characterized by a primary tumor of the prostate gland that expresses small cell morphology and high-grade features, including minimal cytoplasm, nuclear molding, fine chromatin pattern, extensive tumor necrosis and apoptosis, variable tumor giant cells, and a high mitotic rate. Patients often have disproportionally low PSA levels despite having large metastatic burden and visceral disease. Pathologic diagnosis is made on the basis of prostate biopsy using characteristics of small cell tumors and immunohistochemical staining for neuroendocrine markers, such as CD56, chromogranin A, synaptophysin, and neuron-specific enolase.
SCCP arises de novo in approximately 50% of cases; it also occurs in patients with previous or concomitant prostate adenocarcinoma. Patients are often symptomatic at diagnosis because of the extent of the tumor. The aggressive nature and high proliferation rate associated with SCCP result in an increased risk for lytic or blastic bone, visceral, and brain metastases. In addition, paraneoplastic syndromes (eg, the syndrome of inappropriate antidiuretic hormone secretion, Cushing syndrome, and hypercalcemia) frequently occur as a result of the release of peptides.
SCCP metastasizes early in its course and is associated with a poor prognosis. It has a median survival of < 1 year. Fluorodeoxyglucose PET-CT are useful for staging and monitoring treatment response; in addition, given the disease's predilection for brain metastases, MRI of the brain should be considered.
The optimal treatment for patients with metastatic SCCP has not yet been determined. Localized SCCP is treated aggressively, typically with a multimodality approach involving chemotherapy with concurrent or consolidative radiotherapy.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), platinum-based combination chemotherapy (cisplatin-etoposide, carboplatin-etoposide, docetaxel-carboplatin, cabazitaxel-carboplatin) is the first-line approach for patients with metastatic disease.
Physicians are also advised to consult the NCCN guidelines for small cell lung cancer because the behavior of SCCP is similar to that of small cell carcinoma of the lung. Immunotherapy with pembrolizumab may be used for platinum-resistant extrapulmonary small cell carcinoma. However, sipuleucel-T is not recommended for patients with SCCP.
Chad R. Tracy, MD, Professor; Director, Minimally Invasive Surgery, Department of Urology, University of Iowa Hospitals and Clinics, Iowa City, Iowa
Chad R. Tracy, MD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: CVICO Medical Solutions.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
The history and findings in this case are suggestive of small cell carcinoma of the prostate (SCCP).
SCCP is a rare and aggressive cancer that comprises 1%–5% of all prostate cancers (if mixed cases with adenocarcinoma are included). Similar to small cell carcinoma of the lung or other small cell primaries, SCCP is characterized by a primary tumor of the prostate gland that expresses small cell morphology and high-grade features, including minimal cytoplasm, nuclear molding, fine chromatin pattern, extensive tumor necrosis and apoptosis, variable tumor giant cells, and a high mitotic rate. Patients often have disproportionally low PSA levels despite having large metastatic burden and visceral disease. Pathologic diagnosis is made on the basis of prostate biopsy using characteristics of small cell tumors and immunohistochemical staining for neuroendocrine markers, such as CD56, chromogranin A, synaptophysin, and neuron-specific enolase.
SCCP arises de novo in approximately 50% of cases; it also occurs in patients with previous or concomitant prostate adenocarcinoma. Patients are often symptomatic at diagnosis because of the extent of the tumor. The aggressive nature and high proliferation rate associated with SCCP result in an increased risk for lytic or blastic bone, visceral, and brain metastases. In addition, paraneoplastic syndromes (eg, the syndrome of inappropriate antidiuretic hormone secretion, Cushing syndrome, and hypercalcemia) frequently occur as a result of the release of peptides.
SCCP metastasizes early in its course and is associated with a poor prognosis. It has a median survival of < 1 year. Fluorodeoxyglucose PET-CT are useful for staging and monitoring treatment response; in addition, given the disease's predilection for brain metastases, MRI of the brain should be considered.
The optimal treatment for patients with metastatic SCCP has not yet been determined. Localized SCCP is treated aggressively, typically with a multimodality approach involving chemotherapy with concurrent or consolidative radiotherapy.
According to 2023 guidelines from the National Comprehensive Cancer Network (NCCN), platinum-based combination chemotherapy (cisplatin-etoposide, carboplatin-etoposide, docetaxel-carboplatin, cabazitaxel-carboplatin) is the first-line approach for patients with metastatic disease.
Physicians are also advised to consult the NCCN guidelines for small cell lung cancer because the behavior of SCCP is similar to that of small cell carcinoma of the lung. Immunotherapy with pembrolizumab may be used for platinum-resistant extrapulmonary small cell carcinoma. However, sipuleucel-T is not recommended for patients with SCCP.
Chad R. Tracy, MD, Professor; Director, Minimally Invasive Surgery, Department of Urology, University of Iowa Hospitals and Clinics, Iowa City, Iowa
Chad R. Tracy, MD, has disclosed the following relevant financial relationships:
Serve(d) as a consultant for: CVICO Medical Solutions.
Image Quizzes are fictional or fictionalized clinical scenarios intended to provide evidence-based educational takeaways.
A 69-year-old nonsmoking African American man presents with reports of dysuria, nocturia, and unintentional weight loss. He reveals no other lower urinary tract symptoms, pelvic pain, night sweats, back pain, or excessive fatigue. Digital rectal exam reveals an enlarged prostate with a firm, irregular nodule at the right side of the gland. Laboratory tests reveal a prostate-specific antigen (PSA) level of 2.22 ng/mL; a comprehensive metabolic panel and CBC are within normal limits. The patient is 6 ft 1 in and weighs 187 lb.
A transrectal ultrasound-guided prostate biopsy is performed. Histologic examination reveals immunoreactivity for the neuroendocrine markers synaptophysin, chromogranin A, and expression of transcription factor 1. A proliferation of small cells (> 4 lymphocytes in diameter) is noted, with scant cytoplasm, poorly defined borders, finely granular salt-and-pepper chromatin, inconspicuous nucleoli, and a high mitotic count. Evidence of perineural invasion is noted.