User login
Oral Biologics: The New Wave for Treating Psoriasis
Oral Biologics: The New Wave for Treating Psoriasis
Biologic therapies have transformed the treatment of psoriasis. Current biologics approved for psoriasis include monoclonal antibodies targeting various pathways: tumor necrosis factor α (TNF-α) inhibitors (infliximab, adalimumab, certolizumab, etanercept), the p40 subunit common to IL-12 and IL-23 (ustekinumab), the p19 subunit of IL-23 (guselkumab, tildrakizumab, risankizumab), IL-17A (secukinumab, ixekizumab), IL-17 receptor A (brodalumab), and dual IL-17A/IL-17F inhibition (bimekizumab). Recent research showed that risankizumab achieved the highest Psoriasis Area and Severity Index (PASI) 90 scores in short- and long-term treatment periods (4 and 16 weeks, respectively) compared to other biologics, and IL-23 inhibitors demonstrated the lowest short- and long-term adverse event rates and the most favorable long-term risk-benefit profile compared to IL-17, IL-12/23, and TNF-α inhibitors.1
Although these monoclonal antibodies have revolutionized psoriasis treatment, they are large proteins that must be administered subcutaneously or via intravenous injection. Emerging biologics are smaller proteins administered orally via a tablet or pill. In clinical trials, oral biologics have demonstrated efficacy (eTable), suggesting that oral biologics may be the future for psoriasis treatment, as this noninvasive delivery method may help improve patient compliance with treatment.
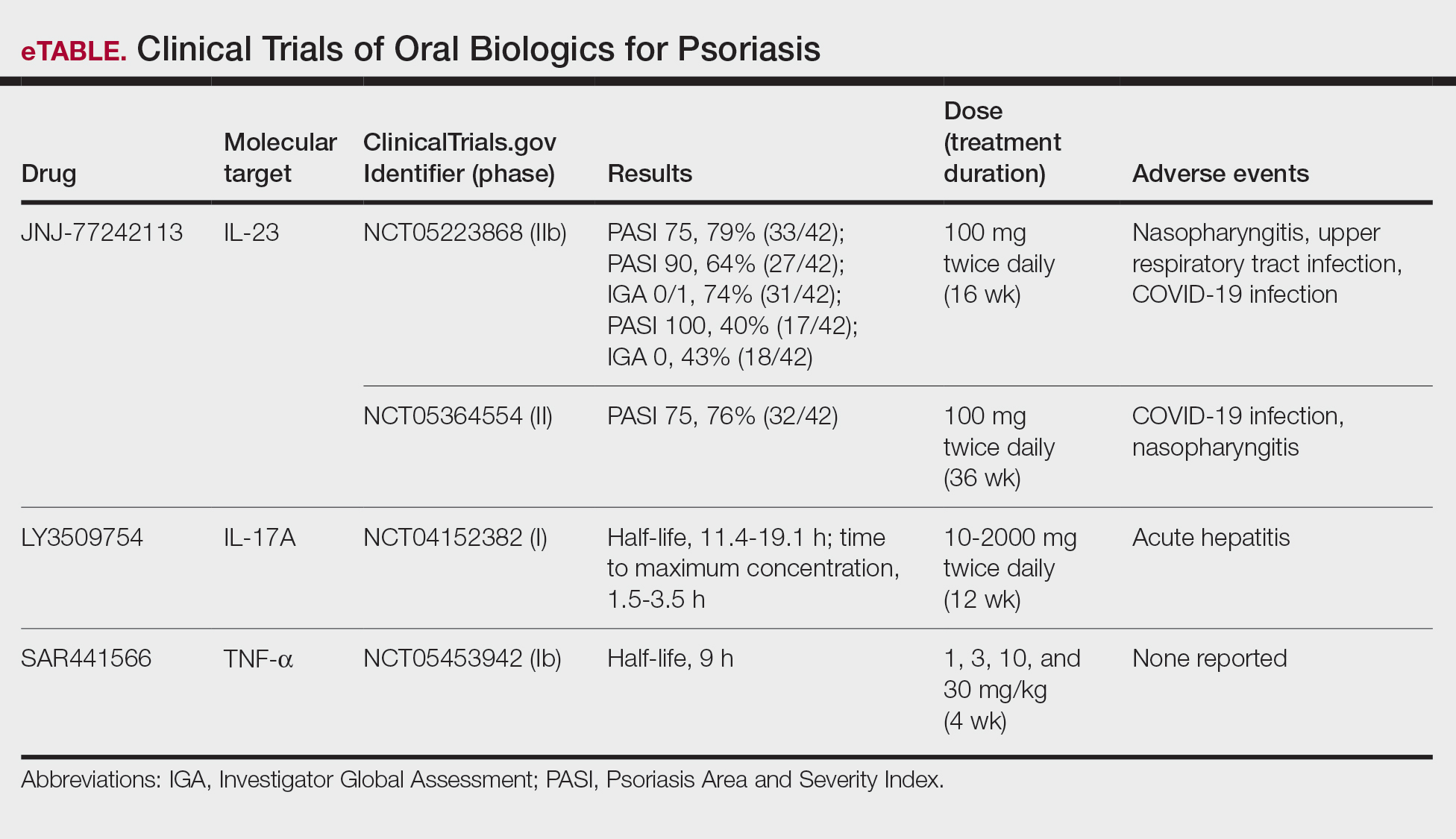
A major inflammatory pathway in psoriasis, IL-23 has been an effective and safe drug target. The novel oral IL-23 inhibitor, JNJ-2113, was discovered in 2017 and currently is being compared to deucravacitinib in the phase III ICONIC-LEAD trial (ClinicalTrials. gov Identifier NCT06095115) in patients with moderate to severe plaque psoriasis.2,3 In the phase IIb FRONTIER 1 trial, treatment with either 3 once-daily (25 mg, 50 mg, 100 mg) and 2 twice-daily (25 mg, 100 mg) doses of JNJ-2113 led to significant improvements in PASI 75 response at 16 weeks compared to placebo (P<.001).4 In the phase IIb long-term extension FRONTIER 2 trial, JNJ-2113 maintained high rates of skin clearance through 52 weeks in adults with moderate to severe plaque psoriasis, with the highest PASI 75 response observed in the 100-mg twice-daily group (32/42 [76.2%]).5 Responses were maintained through week 52 for all JNJ-2113 treatment groups for PASI 90 and PASI 100 endpoints. In addition to ICONIC-LEAD, JNJ-2113 is being evaluated in the phase III multicenter, randomized, double-blind, placebo-controlled trial ICONIC-TOTAL (NCT06095102) in patients with special area psoriasis and ANTHEM-UC (NCT06049017) in patients with ulcerative colitis to evaluate its efficacy and safety. The most common adverse events associated with JNJ-77242113 were mild to moderate and included COVID-19 infection and nasopharyngitis.6 Higher rates of COVID-19 infection likely were due to immune compromise in the setting of the recent pandemic. Similar percentages of at least 1 adverse event were found in JNJ-77242113 and placebo groups (52%-58.6% and 51%-65.7%, respectively).4,5,7
An orally administered small-molecule inhibitor of IL-17A, LY3509754, may represent a convenient alternative to IL-17A–
The small potent molecule SAR441566 inhibits TNF-α by stabilizing an asymmetrical form of the soluble TNF trimer. As the asymmetrical trimer is the biologically active form of TNF-α, stabilization of the trimer compromises downstream signaling and inhibits the functions of TNF-α in vitro and in vivo. Recently, SAR441566 was found to be safe and well tolerated in healthy participants, showing efficacy in mild to moderate psoriasis in a phase Ib trial.9 A phase II trial of SAR441566 (NCT06073119) is being developed to create a more convenient orally bioavailable treatment option for patients with psoriasis compared to established biologic drugs targeting TNF-α.10
Few trials have focused on investigating the antipsoriatic effects of orally administered small molecules. Some of these small molecules can enter cells and inhibit the activation of T lymphocytes, leukocyte trafficking, leukotriene activity/production and angiogenesis, and promote apoptosis. Oral administration of small molecules is the future of effective and affordable psoriasis treatment, but safety and efficacy must first be assessed in clinical trials. JNJ-77242113 has shown a more promising safety profile, has recently undergone phase III trials, and may represent the newest wave for psoriasis treatment. While LY3509754 had a strong pharmacokinetics profile, it was poorly tolerated, and study participants' laboratory results suggested the drug to be hepatotoxic.8 SAR441566 has been shown to be safe and well tolerated in treating psoriasis, and phase II readouts are expected later in 2025. We can expect a new wave of psoriasis treatments with emerging oral therapies.
- Wride AM, Chen GF, Spaulding SL, et al. Biologics for psoriasis. Dermatol Clin. 2024;42:339-355. doi:10.1016/j.det.2024.02.001
- New data shows JNJ-2113, the first and only investigational targeted oral peptide, maintained skin clearance in moderate-to-severe plaque psoriasis through one year. Johnson & Johnson website. March 9, 2024. Accessed August 29, 2024. https://www.jnj.com/media-center/press-releases/new-data-shows-jnj-2113-the-first-and-only-investigational-targeted-oral-peptide-maintained-skin-clearance-in-moderate-to-severe-plaque-psoriasis-through-one-year
- Drakos A, Torres T, Vender R. Emerging oral therapies for the treatment of psoriasis: a review of pipeline agents. Pharmaceutics. 2024;16:111. doi:10.3390/pharmaceutics16010111
- Bissonnette R. A phase 2, randomized, placebo-controlled, dose -ranging study of oral JNJ-77242113 for the treatment of moderate -to-severe plaque psoriasis: FRONTIER 1. Presented at: 25th World Congress of Dermatology; July 3, 2023; Suntec City, Singapore.
- Ferris L. S026. A phase 2b, long-term extension, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severeplaque psoriasis: FRONTIER 2. Presented at: Annual Meeting of the American Academy of Dermatology; San Diego, California; March 8-12, 2024.
- Inc PT. Protagonist announces two new phase 3 ICONIC studies in psoriasis evaluating JNJ-2113 in head-to-head comparisons with deucravacitinib. ACCESSWIRE website. November 27, 2023. Accessed August 29, 2024. https://www.accesswire.com/810075/protagonist-announces-two-new-phase-3-iconic-studies-in-psoriasis-evaluating-jnj-2113-in-head-to-head-comparisons-with-deucravacitinib
- Bissonnette R, Pinter A, Ferris LK, et al. An oral interleukin-23-receptor antagonist peptide for plaque psoriasis. N Engl J Med. 2024;390:510-521. doi:10.1056/NEJMoa2308713
- Datta-Mannan A, Regev A, Coutant DE, et al. Safety, tolerability, and pharmacokinetics of an oral small molecule inhibitor of IL-17A (LY3509754): a phase I randomized placebo-controlled study. Clin Pharmacol Ther. 2024;115:1152-1161. doi:10.1002/cpt.3185
- Vugler A, O’Connell J, Nguyen MA, et al. An orally available small molecule that targets soluble TNF to deliver anti-TNF biologic-like efficacy in rheumatoid arthritis. Front Pharmacol. 2022;13:1037983. doi:10.3389/fphar.2022.1037983
- Sanofi pipeline transformation to accelerate growth driven by record number of potential blockbuster launches, paving the way to industry leadership in immunology. News release. Sanofi; New York: Sanofi; Dec 7, 2023. https://www.sanofi.com/en/media-room/press-releases/2023/2023-12-07-02-30-00-2792186
Biologic therapies have transformed the treatment of psoriasis. Current biologics approved for psoriasis include monoclonal antibodies targeting various pathways: tumor necrosis factor α (TNF-α) inhibitors (infliximab, adalimumab, certolizumab, etanercept), the p40 subunit common to IL-12 and IL-23 (ustekinumab), the p19 subunit of IL-23 (guselkumab, tildrakizumab, risankizumab), IL-17A (secukinumab, ixekizumab), IL-17 receptor A (brodalumab), and dual IL-17A/IL-17F inhibition (bimekizumab). Recent research showed that risankizumab achieved the highest Psoriasis Area and Severity Index (PASI) 90 scores in short- and long-term treatment periods (4 and 16 weeks, respectively) compared to other biologics, and IL-23 inhibitors demonstrated the lowest short- and long-term adverse event rates and the most favorable long-term risk-benefit profile compared to IL-17, IL-12/23, and TNF-α inhibitors.1
Although these monoclonal antibodies have revolutionized psoriasis treatment, they are large proteins that must be administered subcutaneously or via intravenous injection. Emerging biologics are smaller proteins administered orally via a tablet or pill. In clinical trials, oral biologics have demonstrated efficacy (eTable), suggesting that oral biologics may be the future for psoriasis treatment, as this noninvasive delivery method may help improve patient compliance with treatment.

A major inflammatory pathway in psoriasis, IL-23 has been an effective and safe drug target. The novel oral IL-23 inhibitor, JNJ-2113, was discovered in 2017 and currently is being compared to deucravacitinib in the phase III ICONIC-LEAD trial (ClinicalTrials. gov Identifier NCT06095115) in patients with moderate to severe plaque psoriasis.2,3 In the phase IIb FRONTIER 1 trial, treatment with either 3 once-daily (25 mg, 50 mg, 100 mg) and 2 twice-daily (25 mg, 100 mg) doses of JNJ-2113 led to significant improvements in PASI 75 response at 16 weeks compared to placebo (P<.001).4 In the phase IIb long-term extension FRONTIER 2 trial, JNJ-2113 maintained high rates of skin clearance through 52 weeks in adults with moderate to severe plaque psoriasis, with the highest PASI 75 response observed in the 100-mg twice-daily group (32/42 [76.2%]).5 Responses were maintained through week 52 for all JNJ-2113 treatment groups for PASI 90 and PASI 100 endpoints. In addition to ICONIC-LEAD, JNJ-2113 is being evaluated in the phase III multicenter, randomized, double-blind, placebo-controlled trial ICONIC-TOTAL (NCT06095102) in patients with special area psoriasis and ANTHEM-UC (NCT06049017) in patients with ulcerative colitis to evaluate its efficacy and safety. The most common adverse events associated with JNJ-77242113 were mild to moderate and included COVID-19 infection and nasopharyngitis.6 Higher rates of COVID-19 infection likely were due to immune compromise in the setting of the recent pandemic. Similar percentages of at least 1 adverse event were found in JNJ-77242113 and placebo groups (52%-58.6% and 51%-65.7%, respectively).4,5,7
An orally administered small-molecule inhibitor of IL-17A, LY3509754, may represent a convenient alternative to IL-17A–
The small potent molecule SAR441566 inhibits TNF-α by stabilizing an asymmetrical form of the soluble TNF trimer. As the asymmetrical trimer is the biologically active form of TNF-α, stabilization of the trimer compromises downstream signaling and inhibits the functions of TNF-α in vitro and in vivo. Recently, SAR441566 was found to be safe and well tolerated in healthy participants, showing efficacy in mild to moderate psoriasis in a phase Ib trial.9 A phase II trial of SAR441566 (NCT06073119) is being developed to create a more convenient orally bioavailable treatment option for patients with psoriasis compared to established biologic drugs targeting TNF-α.10
Few trials have focused on investigating the antipsoriatic effects of orally administered small molecules. Some of these small molecules can enter cells and inhibit the activation of T lymphocytes, leukocyte trafficking, leukotriene activity/production and angiogenesis, and promote apoptosis. Oral administration of small molecules is the future of effective and affordable psoriasis treatment, but safety and efficacy must first be assessed in clinical trials. JNJ-77242113 has shown a more promising safety profile, has recently undergone phase III trials, and may represent the newest wave for psoriasis treatment. While LY3509754 had a strong pharmacokinetics profile, it was poorly tolerated, and study participants' laboratory results suggested the drug to be hepatotoxic.8 SAR441566 has been shown to be safe and well tolerated in treating psoriasis, and phase II readouts are expected later in 2025. We can expect a new wave of psoriasis treatments with emerging oral therapies.
Biologic therapies have transformed the treatment of psoriasis. Current biologics approved for psoriasis include monoclonal antibodies targeting various pathways: tumor necrosis factor α (TNF-α) inhibitors (infliximab, adalimumab, certolizumab, etanercept), the p40 subunit common to IL-12 and IL-23 (ustekinumab), the p19 subunit of IL-23 (guselkumab, tildrakizumab, risankizumab), IL-17A (secukinumab, ixekizumab), IL-17 receptor A (brodalumab), and dual IL-17A/IL-17F inhibition (bimekizumab). Recent research showed that risankizumab achieved the highest Psoriasis Area and Severity Index (PASI) 90 scores in short- and long-term treatment periods (4 and 16 weeks, respectively) compared to other biologics, and IL-23 inhibitors demonstrated the lowest short- and long-term adverse event rates and the most favorable long-term risk-benefit profile compared to IL-17, IL-12/23, and TNF-α inhibitors.1
Although these monoclonal antibodies have revolutionized psoriasis treatment, they are large proteins that must be administered subcutaneously or via intravenous injection. Emerging biologics are smaller proteins administered orally via a tablet or pill. In clinical trials, oral biologics have demonstrated efficacy (eTable), suggesting that oral biologics may be the future for psoriasis treatment, as this noninvasive delivery method may help improve patient compliance with treatment.

A major inflammatory pathway in psoriasis, IL-23 has been an effective and safe drug target. The novel oral IL-23 inhibitor, JNJ-2113, was discovered in 2017 and currently is being compared to deucravacitinib in the phase III ICONIC-LEAD trial (ClinicalTrials. gov Identifier NCT06095115) in patients with moderate to severe plaque psoriasis.2,3 In the phase IIb FRONTIER 1 trial, treatment with either 3 once-daily (25 mg, 50 mg, 100 mg) and 2 twice-daily (25 mg, 100 mg) doses of JNJ-2113 led to significant improvements in PASI 75 response at 16 weeks compared to placebo (P<.001).4 In the phase IIb long-term extension FRONTIER 2 trial, JNJ-2113 maintained high rates of skin clearance through 52 weeks in adults with moderate to severe plaque psoriasis, with the highest PASI 75 response observed in the 100-mg twice-daily group (32/42 [76.2%]).5 Responses were maintained through week 52 for all JNJ-2113 treatment groups for PASI 90 and PASI 100 endpoints. In addition to ICONIC-LEAD, JNJ-2113 is being evaluated in the phase III multicenter, randomized, double-blind, placebo-controlled trial ICONIC-TOTAL (NCT06095102) in patients with special area psoriasis and ANTHEM-UC (NCT06049017) in patients with ulcerative colitis to evaluate its efficacy and safety. The most common adverse events associated with JNJ-77242113 were mild to moderate and included COVID-19 infection and nasopharyngitis.6 Higher rates of COVID-19 infection likely were due to immune compromise in the setting of the recent pandemic. Similar percentages of at least 1 adverse event were found in JNJ-77242113 and placebo groups (52%-58.6% and 51%-65.7%, respectively).4,5,7
An orally administered small-molecule inhibitor of IL-17A, LY3509754, may represent a convenient alternative to IL-17A–
The small potent molecule SAR441566 inhibits TNF-α by stabilizing an asymmetrical form of the soluble TNF trimer. As the asymmetrical trimer is the biologically active form of TNF-α, stabilization of the trimer compromises downstream signaling and inhibits the functions of TNF-α in vitro and in vivo. Recently, SAR441566 was found to be safe and well tolerated in healthy participants, showing efficacy in mild to moderate psoriasis in a phase Ib trial.9 A phase II trial of SAR441566 (NCT06073119) is being developed to create a more convenient orally bioavailable treatment option for patients with psoriasis compared to established biologic drugs targeting TNF-α.10
Few trials have focused on investigating the antipsoriatic effects of orally administered small molecules. Some of these small molecules can enter cells and inhibit the activation of T lymphocytes, leukocyte trafficking, leukotriene activity/production and angiogenesis, and promote apoptosis. Oral administration of small molecules is the future of effective and affordable psoriasis treatment, but safety and efficacy must first be assessed in clinical trials. JNJ-77242113 has shown a more promising safety profile, has recently undergone phase III trials, and may represent the newest wave for psoriasis treatment. While LY3509754 had a strong pharmacokinetics profile, it was poorly tolerated, and study participants' laboratory results suggested the drug to be hepatotoxic.8 SAR441566 has been shown to be safe and well tolerated in treating psoriasis, and phase II readouts are expected later in 2025. We can expect a new wave of psoriasis treatments with emerging oral therapies.
- Wride AM, Chen GF, Spaulding SL, et al. Biologics for psoriasis. Dermatol Clin. 2024;42:339-355. doi:10.1016/j.det.2024.02.001
- New data shows JNJ-2113, the first and only investigational targeted oral peptide, maintained skin clearance in moderate-to-severe plaque psoriasis through one year. Johnson & Johnson website. March 9, 2024. Accessed August 29, 2024. https://www.jnj.com/media-center/press-releases/new-data-shows-jnj-2113-the-first-and-only-investigational-targeted-oral-peptide-maintained-skin-clearance-in-moderate-to-severe-plaque-psoriasis-through-one-year
- Drakos A, Torres T, Vender R. Emerging oral therapies for the treatment of psoriasis: a review of pipeline agents. Pharmaceutics. 2024;16:111. doi:10.3390/pharmaceutics16010111
- Bissonnette R. A phase 2, randomized, placebo-controlled, dose -ranging study of oral JNJ-77242113 for the treatment of moderate -to-severe plaque psoriasis: FRONTIER 1. Presented at: 25th World Congress of Dermatology; July 3, 2023; Suntec City, Singapore.
- Ferris L. S026. A phase 2b, long-term extension, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severeplaque psoriasis: FRONTIER 2. Presented at: Annual Meeting of the American Academy of Dermatology; San Diego, California; March 8-12, 2024.
- Inc PT. Protagonist announces two new phase 3 ICONIC studies in psoriasis evaluating JNJ-2113 in head-to-head comparisons with deucravacitinib. ACCESSWIRE website. November 27, 2023. Accessed August 29, 2024. https://www.accesswire.com/810075/protagonist-announces-two-new-phase-3-iconic-studies-in-psoriasis-evaluating-jnj-2113-in-head-to-head-comparisons-with-deucravacitinib
- Bissonnette R, Pinter A, Ferris LK, et al. An oral interleukin-23-receptor antagonist peptide for plaque psoriasis. N Engl J Med. 2024;390:510-521. doi:10.1056/NEJMoa2308713
- Datta-Mannan A, Regev A, Coutant DE, et al. Safety, tolerability, and pharmacokinetics of an oral small molecule inhibitor of IL-17A (LY3509754): a phase I randomized placebo-controlled study. Clin Pharmacol Ther. 2024;115:1152-1161. doi:10.1002/cpt.3185
- Vugler A, O’Connell J, Nguyen MA, et al. An orally available small molecule that targets soluble TNF to deliver anti-TNF biologic-like efficacy in rheumatoid arthritis. Front Pharmacol. 2022;13:1037983. doi:10.3389/fphar.2022.1037983
- Sanofi pipeline transformation to accelerate growth driven by record number of potential blockbuster launches, paving the way to industry leadership in immunology. News release. Sanofi; New York: Sanofi; Dec 7, 2023. https://www.sanofi.com/en/media-room/press-releases/2023/2023-12-07-02-30-00-2792186
- Wride AM, Chen GF, Spaulding SL, et al. Biologics for psoriasis. Dermatol Clin. 2024;42:339-355. doi:10.1016/j.det.2024.02.001
- New data shows JNJ-2113, the first and only investigational targeted oral peptide, maintained skin clearance in moderate-to-severe plaque psoriasis through one year. Johnson & Johnson website. March 9, 2024. Accessed August 29, 2024. https://www.jnj.com/media-center/press-releases/new-data-shows-jnj-2113-the-first-and-only-investigational-targeted-oral-peptide-maintained-skin-clearance-in-moderate-to-severe-plaque-psoriasis-through-one-year
- Drakos A, Torres T, Vender R. Emerging oral therapies for the treatment of psoriasis: a review of pipeline agents. Pharmaceutics. 2024;16:111. doi:10.3390/pharmaceutics16010111
- Bissonnette R. A phase 2, randomized, placebo-controlled, dose -ranging study of oral JNJ-77242113 for the treatment of moderate -to-severe plaque psoriasis: FRONTIER 1. Presented at: 25th World Congress of Dermatology; July 3, 2023; Suntec City, Singapore.
- Ferris L. S026. A phase 2b, long-term extension, dose-ranging study of oral JNJ-77242113 for the treatment of moderate-to-severeplaque psoriasis: FRONTIER 2. Presented at: Annual Meeting of the American Academy of Dermatology; San Diego, California; March 8-12, 2024.
- Inc PT. Protagonist announces two new phase 3 ICONIC studies in psoriasis evaluating JNJ-2113 in head-to-head comparisons with deucravacitinib. ACCESSWIRE website. November 27, 2023. Accessed August 29, 2024. https://www.accesswire.com/810075/protagonist-announces-two-new-phase-3-iconic-studies-in-psoriasis-evaluating-jnj-2113-in-head-to-head-comparisons-with-deucravacitinib
- Bissonnette R, Pinter A, Ferris LK, et al. An oral interleukin-23-receptor antagonist peptide for plaque psoriasis. N Engl J Med. 2024;390:510-521. doi:10.1056/NEJMoa2308713
- Datta-Mannan A, Regev A, Coutant DE, et al. Safety, tolerability, and pharmacokinetics of an oral small molecule inhibitor of IL-17A (LY3509754): a phase I randomized placebo-controlled study. Clin Pharmacol Ther. 2024;115:1152-1161. doi:10.1002/cpt.3185
- Vugler A, O’Connell J, Nguyen MA, et al. An orally available small molecule that targets soluble TNF to deliver anti-TNF biologic-like efficacy in rheumatoid arthritis. Front Pharmacol. 2022;13:1037983. doi:10.3389/fphar.2022.1037983
- Sanofi pipeline transformation to accelerate growth driven by record number of potential blockbuster launches, paving the way to industry leadership in immunology. News release. Sanofi; New York: Sanofi; Dec 7, 2023. https://www.sanofi.com/en/media-room/press-releases/2023/2023-12-07-02-30-00-2792186
Oral Biologics: The New Wave for Treating Psoriasis
Oral Biologics: The New Wave for Treating Psoriasis
PRACTICE POINTS
- The biologics that currently are approved for psoriasis are expensive and must be administered via injection due to their large molecule size.
- Emerging small-molecule oral therapies for psoriasis are effective and affordable and may represent the future for psoriasis patients.
Association Between Psoriasis and Sunburn Prevalence in US Adults
Association Between Psoriasis and Sunburn Prevalence in US Adults
To the Editor:
UV light plays an essential role in various environmental and biological processes.1 Excessive exposure to UV radiation can lead to sunburn, which is marked by skin erythema and pain.2 A study of more than 31,000 individuals found that 34.2% of adults aged 18 years and older reported at least 1 sunburn during the survey year.3 A lack of research regarding the incidence of sunburns in patients with psoriasis is particularly important considering the heightened incidence of skin cancer observed in this population.4 Thus, the aim of our study was to analyze the prevalence of sunburns among US adults with psoriasis utilizing data from the National Health and Nutrition Examination Survey (NHANES) database.5
Our analysis initially included 11,842 participants ranging in age from 20 to 59 years; 35 did not respond to questions assessing psoriasis and sunburn prevalence and thus were excluded. Multivariable logistic regression analyses were performed using Stata/SE 18 (StataCorp LLC) to assess the relationship between psoriasis and sunburns. Our models controlled for patient age, sex, income, race, education, diabetes status, tobacco use, and body mass index. A P value <.05 was considered statistically significant. The study period from January 2009 to December 2014 was chosen based on the availability of the most recent and comprehensive psoriasis data within the NHANES database.
In the NHANES data we evaluated, psoriasis status was assessed by asking, “Have you ever been told by a doctor or other health professional that you had psoriasis?” History of sunburns in the survey year was assessed by the question, “How many times in the past year have you had sunburn?” Patients who reported 1 or more sunburns were included in the sunburn cohort, while those who did not report a sunburn were included in the no sunburn cohort.
In our analysis, the prevalence of at least 1 sunburn in the survey year in patients with psoriasis was 55.4% (weighted), compared to 45.6% (weighted) among those without psoriasis (eTable 1). Although there was no statistically significant relationship between psoriasis and history of sunburn in patients aged 20 to 59 years, a subgroup analysis revealed a significant association between psoriasis and sunburn in adults aged 20 to 39 years after adjusting for potential confounding variables (adjusted OR, 1.57 [95% CI, 1.00-2.45]; P=.049)(eTable 2). Further analysis of subgroups showed no statistically significant results with adjustment of the logistic regression model. Characterizing response rates is important for assessing the validity of survey studies. The NHANES response rate from 2009 to 2014 was 72.9%, enhancing the reliability of our findings.
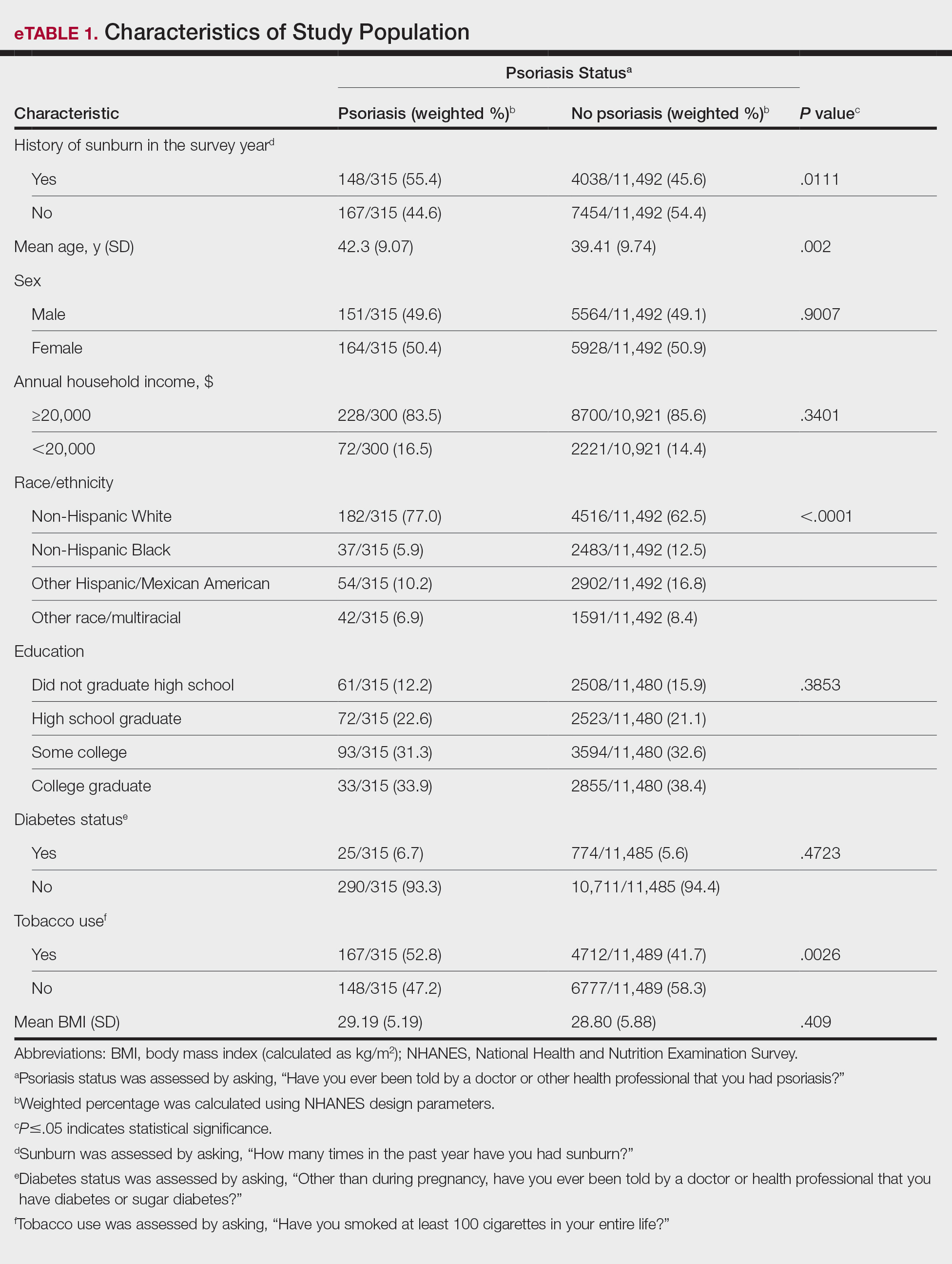
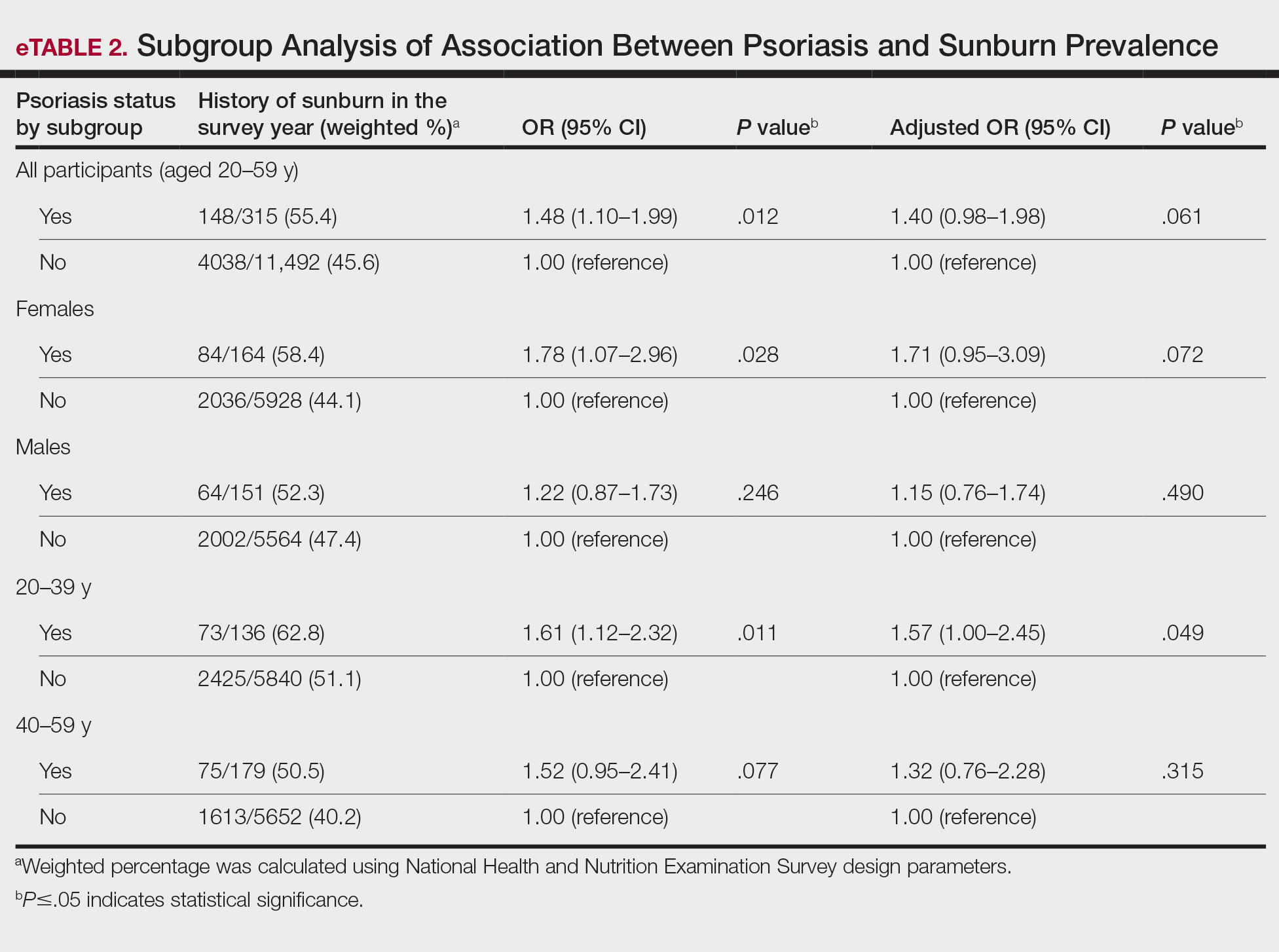
Our study revealed an increased prevalence of sunburn in US adults with psoriasis. A trend of increased sunburn prevalence among younger adults regardless of psoriasis status is corroborated by the literature. Surveys conducted in the United States in 2005, 2010, and 2015 showed that 43% to 50% of adults aged 18 to 39 years and 28% to 42% of those aged 40 to 59 years reported experiencing at least 1 sunburn within the respective survey year.6 Furthermore, in our study, patients with psoriasis reported higher rates of sunburn than their counterparts without psoriasis, both in those aged 20 to 39 years (psoriasis, 62.8% [73/136]; no psoriasis, 51.1% [2425/5840]) and those aged 40 to 59 years (psoriasis, 50.5% [n=75/179]; no psoriasis, 40.2% [1613/5652]), though it was only statistically significant in the 20-to-39 age group. This discrepancy may be attributed to differences in sun-protective behaviors in younger vs older adults. A study from the NHANES database found that, among individuals aged 20 to 39 years, 75.9% [4225/5493] reported staying in the shade, 50.0% [2346/5493] reported using sunscreen, and 31.2% [1874/5493] reported wearing sun-protective clothing.7 Interestingly, the likelihood of engaging in all 3 behaviors was 28% lower in the 20-to-39 age group vs the 40-to-59 age group (adjusted OR, 0.72; 95% CI, 0.62-0.83).7
While our analysis adjusted for age, race/ethnicity, and tobacco use to mitigate potential confounding, we acknowledge the statistically significant differences observed in these variables between study groups as presented in eTable 2. These differences may reflect inherent disparities in the study population. We employed multivariable regression analysis to control for these covariates in our primary analyses. Of note, there was a statistically significant difference associated with race/ethnicity when comparing non-Hispanic White individuals with psoriasis (77.0% [n=182/315]) and those without psoriasis (62.5% [n=4516/11,492])(P<.0001)(eTable 1). The higher proportion of non-Hispanic White patients in the psoriasis group may reflect an increased susceptibility to sunburn given their typically lighter skin pigmentation; however, our analysis controlled for race/ethnicity (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of racial/ethnic differences. There also were statistically significant differences in tobacco use (P=.0026) and age (P=.002) in our unadjusted findings (eTable 1). Again, our analysis controlled for these factors (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of tobacco use and age differences. This approach enhanced the reliability of our findings.
The association between psoriasis and skin cancer has previously been evaluated using the NHANES database—one study found that patients with psoriasis had a significantly higher prevalence of nonmelanoma skin cancer compared with those without psoriasis (3.0% vs 1.3%; relative risk, 2.29; P<.001).8 This difference remained significant after adjusting for confounding variables, as it was found that psoriasis was independently associated with a 1.5-fold increased risk for nonmelanoma skin cancer (adjusted relative risk, 2.06; P=.004).8
The relationship between psoriasis and sunburn may be due to behavioral choices, such as the use of phototherapy for managing psoriasis due to its recognized advantages.9 Patients may seek out both artificial and natural light sources more frequently, potentially increasing the risk for sunburn.10 Psoriasis-related sunburn susceptibility may stem from biological factors, including vitamin D insufficiency, as vitamin D is crucial for keratinocyte differentiation, immune function, and UV protection and repair.11 One study examined the effects of high-dose vitamin D3 on sunburn-induced inflammation.12 Patients who received high-dose vitamin D3 exhibited reduced skin inflammation, enhanced skin barrier repair, and increased anti-inflammatory response compared with those who did not receive the supplement. This improvement was associated with upregulation of arginase 1, an anti-inflammatory enzyme, leading to decreased levels of pro-inflammatory mediators such as tumor necrosis factor α and inducible nitric oxide synthase, thereby promoting tissue repair and reducing prolonged inflammation.12 These findings suggest that vitamin D insufficiency coupled with dysregulated immune responses may contribute to the heightened susceptibility of individuals with psoriasis to sunburn.
The established correlation between sunburn and skin cancer4,8 coupled with our findings of increased prevalence of sunburn in individuals with psoriasis underscores the need for additional research to clarify the underlying biological and behavioral factors that may contribute to a higher prevalence of sunburn in these patients, along with the implications for skin cancer development. Limitations of our study included potential recall bias, as individuals self-reported their clinical conditions and the inability to incorporate psoriasis severity into our analysis, as this was not consistently captured in the NHANES questionnaire during the study period.
- Blaustein AR, Searle C. Ultraviolet radiation. In: Levin SA, ed. Encyclopedia of Biodiversity. 2nd ed. Academic Press; 2013:296-303.
- D’Orazio J, Jarrett S, Amaro-Ortiz A, et al. UV radiation and the skin. Int J Mol Sci. 2013;14:12222-12248
- Holman DM, Ding H, Guy GP Jr, et al. Prevalence of sun protection use and sunburn and association of demographic and behavioral characteristics with sunburn among US adults. JAMA Dermatol. 2018;154:561-568.
- Balda A, Wani I, Roohi TF, et al. Psoriasis and skin cancer—is there a link? Int Immunopharmacol. 2023;121:110464.
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed December 4, 2024. https://wwwn.cdc.gov/nchs/nhanes/Default.aspx
- Holman DM, Ding H, Berkowitz Z, et al. Sunburn prevalence among US adults, National Health Interview Survey 2005, 2010, and 2015. J Am Acad Dermatol. 2019;80:817-820.
- Challapalli SD, Shetty KR, Bui Q, et al. Sun protective behaviors among adolescents and young adults in the United States. J Natl Med Assoc. 2023;115:353-361.
- Herbosa CM, Hodges W, Mann C, et al. Risk of cancer in psoriasis: study of a nationally representative sample of the US population with comparison to a single]institution cohort. J Am Acad Dermatol Venereol. 2020;34:E529-E531.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Åkerla P, Pukkala E, Helminen M, et al. Skin cancer risk of narrow-band UV-B (TL-01) phototherapy: a multi-center registry study with 4,815 patients. Acta Derm Venereol. 2024;104:adv39927.
- Filoni A, Vestita M, Congedo M, et al. Association between psoriasis and vitamin D: duration of disease correlates with decreased vitamin D serum levels: an observational case-control study. Medicine (Baltimore). 2018;97:E11185.
- Scott JF, Das LM, Ahsanuddin S, et al. Oral vitamin D rapidly attenuates inflammation from sunburn: an interventional study. J Invest Dermatol. 2017;137:2078-2086.
To the Editor:
UV light plays an essential role in various environmental and biological processes.1 Excessive exposure to UV radiation can lead to sunburn, which is marked by skin erythema and pain.2 A study of more than 31,000 individuals found that 34.2% of adults aged 18 years and older reported at least 1 sunburn during the survey year.3 A lack of research regarding the incidence of sunburns in patients with psoriasis is particularly important considering the heightened incidence of skin cancer observed in this population.4 Thus, the aim of our study was to analyze the prevalence of sunburns among US adults with psoriasis utilizing data from the National Health and Nutrition Examination Survey (NHANES) database.5
Our analysis initially included 11,842 participants ranging in age from 20 to 59 years; 35 did not respond to questions assessing psoriasis and sunburn prevalence and thus were excluded. Multivariable logistic regression analyses were performed using Stata/SE 18 (StataCorp LLC) to assess the relationship between psoriasis and sunburns. Our models controlled for patient age, sex, income, race, education, diabetes status, tobacco use, and body mass index. A P value <.05 was considered statistically significant. The study period from January 2009 to December 2014 was chosen based on the availability of the most recent and comprehensive psoriasis data within the NHANES database.
In the NHANES data we evaluated, psoriasis status was assessed by asking, “Have you ever been told by a doctor or other health professional that you had psoriasis?” History of sunburns in the survey year was assessed by the question, “How many times in the past year have you had sunburn?” Patients who reported 1 or more sunburns were included in the sunburn cohort, while those who did not report a sunburn were included in the no sunburn cohort.
In our analysis, the prevalence of at least 1 sunburn in the survey year in patients with psoriasis was 55.4% (weighted), compared to 45.6% (weighted) among those without psoriasis (eTable 1). Although there was no statistically significant relationship between psoriasis and history of sunburn in patients aged 20 to 59 years, a subgroup analysis revealed a significant association between psoriasis and sunburn in adults aged 20 to 39 years after adjusting for potential confounding variables (adjusted OR, 1.57 [95% CI, 1.00-2.45]; P=.049)(eTable 2). Further analysis of subgroups showed no statistically significant results with adjustment of the logistic regression model. Characterizing response rates is important for assessing the validity of survey studies. The NHANES response rate from 2009 to 2014 was 72.9%, enhancing the reliability of our findings.


Our study revealed an increased prevalence of sunburn in US adults with psoriasis. A trend of increased sunburn prevalence among younger adults regardless of psoriasis status is corroborated by the literature. Surveys conducted in the United States in 2005, 2010, and 2015 showed that 43% to 50% of adults aged 18 to 39 years and 28% to 42% of those aged 40 to 59 years reported experiencing at least 1 sunburn within the respective survey year.6 Furthermore, in our study, patients with psoriasis reported higher rates of sunburn than their counterparts without psoriasis, both in those aged 20 to 39 years (psoriasis, 62.8% [73/136]; no psoriasis, 51.1% [2425/5840]) and those aged 40 to 59 years (psoriasis, 50.5% [n=75/179]; no psoriasis, 40.2% [1613/5652]), though it was only statistically significant in the 20-to-39 age group. This discrepancy may be attributed to differences in sun-protective behaviors in younger vs older adults. A study from the NHANES database found that, among individuals aged 20 to 39 years, 75.9% [4225/5493] reported staying in the shade, 50.0% [2346/5493] reported using sunscreen, and 31.2% [1874/5493] reported wearing sun-protective clothing.7 Interestingly, the likelihood of engaging in all 3 behaviors was 28% lower in the 20-to-39 age group vs the 40-to-59 age group (adjusted OR, 0.72; 95% CI, 0.62-0.83).7
While our analysis adjusted for age, race/ethnicity, and tobacco use to mitigate potential confounding, we acknowledge the statistically significant differences observed in these variables between study groups as presented in eTable 2. These differences may reflect inherent disparities in the study population. We employed multivariable regression analysis to control for these covariates in our primary analyses. Of note, there was a statistically significant difference associated with race/ethnicity when comparing non-Hispanic White individuals with psoriasis (77.0% [n=182/315]) and those without psoriasis (62.5% [n=4516/11,492])(P<.0001)(eTable 1). The higher proportion of non-Hispanic White patients in the psoriasis group may reflect an increased susceptibility to sunburn given their typically lighter skin pigmentation; however, our analysis controlled for race/ethnicity (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of racial/ethnic differences. There also were statistically significant differences in tobacco use (P=.0026) and age (P=.002) in our unadjusted findings (eTable 1). Again, our analysis controlled for these factors (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of tobacco use and age differences. This approach enhanced the reliability of our findings.
The association between psoriasis and skin cancer has previously been evaluated using the NHANES database—one study found that patients with psoriasis had a significantly higher prevalence of nonmelanoma skin cancer compared with those without psoriasis (3.0% vs 1.3%; relative risk, 2.29; P<.001).8 This difference remained significant after adjusting for confounding variables, as it was found that psoriasis was independently associated with a 1.5-fold increased risk for nonmelanoma skin cancer (adjusted relative risk, 2.06; P=.004).8
The relationship between psoriasis and sunburn may be due to behavioral choices, such as the use of phototherapy for managing psoriasis due to its recognized advantages.9 Patients may seek out both artificial and natural light sources more frequently, potentially increasing the risk for sunburn.10 Psoriasis-related sunburn susceptibility may stem from biological factors, including vitamin D insufficiency, as vitamin D is crucial for keratinocyte differentiation, immune function, and UV protection and repair.11 One study examined the effects of high-dose vitamin D3 on sunburn-induced inflammation.12 Patients who received high-dose vitamin D3 exhibited reduced skin inflammation, enhanced skin barrier repair, and increased anti-inflammatory response compared with those who did not receive the supplement. This improvement was associated with upregulation of arginase 1, an anti-inflammatory enzyme, leading to decreased levels of pro-inflammatory mediators such as tumor necrosis factor α and inducible nitric oxide synthase, thereby promoting tissue repair and reducing prolonged inflammation.12 These findings suggest that vitamin D insufficiency coupled with dysregulated immune responses may contribute to the heightened susceptibility of individuals with psoriasis to sunburn.
The established correlation between sunburn and skin cancer4,8 coupled with our findings of increased prevalence of sunburn in individuals with psoriasis underscores the need for additional research to clarify the underlying biological and behavioral factors that may contribute to a higher prevalence of sunburn in these patients, along with the implications for skin cancer development. Limitations of our study included potential recall bias, as individuals self-reported their clinical conditions and the inability to incorporate psoriasis severity into our analysis, as this was not consistently captured in the NHANES questionnaire during the study period.
To the Editor:
UV light plays an essential role in various environmental and biological processes.1 Excessive exposure to UV radiation can lead to sunburn, which is marked by skin erythema and pain.2 A study of more than 31,000 individuals found that 34.2% of adults aged 18 years and older reported at least 1 sunburn during the survey year.3 A lack of research regarding the incidence of sunburns in patients with psoriasis is particularly important considering the heightened incidence of skin cancer observed in this population.4 Thus, the aim of our study was to analyze the prevalence of sunburns among US adults with psoriasis utilizing data from the National Health and Nutrition Examination Survey (NHANES) database.5
Our analysis initially included 11,842 participants ranging in age from 20 to 59 years; 35 did not respond to questions assessing psoriasis and sunburn prevalence and thus were excluded. Multivariable logistic regression analyses were performed using Stata/SE 18 (StataCorp LLC) to assess the relationship between psoriasis and sunburns. Our models controlled for patient age, sex, income, race, education, diabetes status, tobacco use, and body mass index. A P value <.05 was considered statistically significant. The study period from January 2009 to December 2014 was chosen based on the availability of the most recent and comprehensive psoriasis data within the NHANES database.
In the NHANES data we evaluated, psoriasis status was assessed by asking, “Have you ever been told by a doctor or other health professional that you had psoriasis?” History of sunburns in the survey year was assessed by the question, “How many times in the past year have you had sunburn?” Patients who reported 1 or more sunburns were included in the sunburn cohort, while those who did not report a sunburn were included in the no sunburn cohort.
In our analysis, the prevalence of at least 1 sunburn in the survey year in patients with psoriasis was 55.4% (weighted), compared to 45.6% (weighted) among those without psoriasis (eTable 1). Although there was no statistically significant relationship between psoriasis and history of sunburn in patients aged 20 to 59 years, a subgroup analysis revealed a significant association between psoriasis and sunburn in adults aged 20 to 39 years after adjusting for potential confounding variables (adjusted OR, 1.57 [95% CI, 1.00-2.45]; P=.049)(eTable 2). Further analysis of subgroups showed no statistically significant results with adjustment of the logistic regression model. Characterizing response rates is important for assessing the validity of survey studies. The NHANES response rate from 2009 to 2014 was 72.9%, enhancing the reliability of our findings.


Our study revealed an increased prevalence of sunburn in US adults with psoriasis. A trend of increased sunburn prevalence among younger adults regardless of psoriasis status is corroborated by the literature. Surveys conducted in the United States in 2005, 2010, and 2015 showed that 43% to 50% of adults aged 18 to 39 years and 28% to 42% of those aged 40 to 59 years reported experiencing at least 1 sunburn within the respective survey year.6 Furthermore, in our study, patients with psoriasis reported higher rates of sunburn than their counterparts without psoriasis, both in those aged 20 to 39 years (psoriasis, 62.8% [73/136]; no psoriasis, 51.1% [2425/5840]) and those aged 40 to 59 years (psoriasis, 50.5% [n=75/179]; no psoriasis, 40.2% [1613/5652]), though it was only statistically significant in the 20-to-39 age group. This discrepancy may be attributed to differences in sun-protective behaviors in younger vs older adults. A study from the NHANES database found that, among individuals aged 20 to 39 years, 75.9% [4225/5493] reported staying in the shade, 50.0% [2346/5493] reported using sunscreen, and 31.2% [1874/5493] reported wearing sun-protective clothing.7 Interestingly, the likelihood of engaging in all 3 behaviors was 28% lower in the 20-to-39 age group vs the 40-to-59 age group (adjusted OR, 0.72; 95% CI, 0.62-0.83).7
While our analysis adjusted for age, race/ethnicity, and tobacco use to mitigate potential confounding, we acknowledge the statistically significant differences observed in these variables between study groups as presented in eTable 2. These differences may reflect inherent disparities in the study population. We employed multivariable regression analysis to control for these covariates in our primary analyses. Of note, there was a statistically significant difference associated with race/ethnicity when comparing non-Hispanic White individuals with psoriasis (77.0% [n=182/315]) and those without psoriasis (62.5% [n=4516/11,492])(P<.0001)(eTable 1). The higher proportion of non-Hispanic White patients in the psoriasis group may reflect an increased susceptibility to sunburn given their typically lighter skin pigmentation; however, our analysis controlled for race/ethnicity (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of racial/ethnic differences. There also were statistically significant differences in tobacco use (P=.0026) and age (P=.002) in our unadjusted findings (eTable 1). Again, our analysis controlled for these factors (eTable 2), thereby allowing us to isolate the effect of psoriasis on sunburn prevalence independent of tobacco use and age differences. This approach enhanced the reliability of our findings.
The association between psoriasis and skin cancer has previously been evaluated using the NHANES database—one study found that patients with psoriasis had a significantly higher prevalence of nonmelanoma skin cancer compared with those without psoriasis (3.0% vs 1.3%; relative risk, 2.29; P<.001).8 This difference remained significant after adjusting for confounding variables, as it was found that psoriasis was independently associated with a 1.5-fold increased risk for nonmelanoma skin cancer (adjusted relative risk, 2.06; P=.004).8
The relationship between psoriasis and sunburn may be due to behavioral choices, such as the use of phototherapy for managing psoriasis due to its recognized advantages.9 Patients may seek out both artificial and natural light sources more frequently, potentially increasing the risk for sunburn.10 Psoriasis-related sunburn susceptibility may stem from biological factors, including vitamin D insufficiency, as vitamin D is crucial for keratinocyte differentiation, immune function, and UV protection and repair.11 One study examined the effects of high-dose vitamin D3 on sunburn-induced inflammation.12 Patients who received high-dose vitamin D3 exhibited reduced skin inflammation, enhanced skin barrier repair, and increased anti-inflammatory response compared with those who did not receive the supplement. This improvement was associated with upregulation of arginase 1, an anti-inflammatory enzyme, leading to decreased levels of pro-inflammatory mediators such as tumor necrosis factor α and inducible nitric oxide synthase, thereby promoting tissue repair and reducing prolonged inflammation.12 These findings suggest that vitamin D insufficiency coupled with dysregulated immune responses may contribute to the heightened susceptibility of individuals with psoriasis to sunburn.
The established correlation between sunburn and skin cancer4,8 coupled with our findings of increased prevalence of sunburn in individuals with psoriasis underscores the need for additional research to clarify the underlying biological and behavioral factors that may contribute to a higher prevalence of sunburn in these patients, along with the implications for skin cancer development. Limitations of our study included potential recall bias, as individuals self-reported their clinical conditions and the inability to incorporate psoriasis severity into our analysis, as this was not consistently captured in the NHANES questionnaire during the study period.
- Blaustein AR, Searle C. Ultraviolet radiation. In: Levin SA, ed. Encyclopedia of Biodiversity. 2nd ed. Academic Press; 2013:296-303.
- D’Orazio J, Jarrett S, Amaro-Ortiz A, et al. UV radiation and the skin. Int J Mol Sci. 2013;14:12222-12248
- Holman DM, Ding H, Guy GP Jr, et al. Prevalence of sun protection use and sunburn and association of demographic and behavioral characteristics with sunburn among US adults. JAMA Dermatol. 2018;154:561-568.
- Balda A, Wani I, Roohi TF, et al. Psoriasis and skin cancer—is there a link? Int Immunopharmacol. 2023;121:110464.
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed December 4, 2024. https://wwwn.cdc.gov/nchs/nhanes/Default.aspx
- Holman DM, Ding H, Berkowitz Z, et al. Sunburn prevalence among US adults, National Health Interview Survey 2005, 2010, and 2015. J Am Acad Dermatol. 2019;80:817-820.
- Challapalli SD, Shetty KR, Bui Q, et al. Sun protective behaviors among adolescents and young adults in the United States. J Natl Med Assoc. 2023;115:353-361.
- Herbosa CM, Hodges W, Mann C, et al. Risk of cancer in psoriasis: study of a nationally representative sample of the US population with comparison to a single]institution cohort. J Am Acad Dermatol Venereol. 2020;34:E529-E531.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Åkerla P, Pukkala E, Helminen M, et al. Skin cancer risk of narrow-band UV-B (TL-01) phototherapy: a multi-center registry study with 4,815 patients. Acta Derm Venereol. 2024;104:adv39927.
- Filoni A, Vestita M, Congedo M, et al. Association between psoriasis and vitamin D: duration of disease correlates with decreased vitamin D serum levels: an observational case-control study. Medicine (Baltimore). 2018;97:E11185.
- Scott JF, Das LM, Ahsanuddin S, et al. Oral vitamin D rapidly attenuates inflammation from sunburn: an interventional study. J Invest Dermatol. 2017;137:2078-2086.
- Blaustein AR, Searle C. Ultraviolet radiation. In: Levin SA, ed. Encyclopedia of Biodiversity. 2nd ed. Academic Press; 2013:296-303.
- D’Orazio J, Jarrett S, Amaro-Ortiz A, et al. UV radiation and the skin. Int J Mol Sci. 2013;14:12222-12248
- Holman DM, Ding H, Guy GP Jr, et al. Prevalence of sun protection use and sunburn and association of demographic and behavioral characteristics with sunburn among US adults. JAMA Dermatol. 2018;154:561-568.
- Balda A, Wani I, Roohi TF, et al. Psoriasis and skin cancer—is there a link? Int Immunopharmacol. 2023;121:110464.
- Centers for Disease Control and Prevention. National Health and Nutrition Examination Survey. NHANES questionnaires, datasets, and related documentation. Accessed December 4, 2024. https://wwwn.cdc.gov/nchs/nhanes/Default.aspx
- Holman DM, Ding H, Berkowitz Z, et al. Sunburn prevalence among US adults, National Health Interview Survey 2005, 2010, and 2015. J Am Acad Dermatol. 2019;80:817-820.
- Challapalli SD, Shetty KR, Bui Q, et al. Sun protective behaviors among adolescents and young adults in the United States. J Natl Med Assoc. 2023;115:353-361.
- Herbosa CM, Hodges W, Mann C, et al. Risk of cancer in psoriasis: study of a nationally representative sample of the US population with comparison to a single]institution cohort. J Am Acad Dermatol Venereol. 2020;34:E529-E531.
- Elmets CA, Lim HW, Stoff B, et al. Joint American Academy of Dermatology-National Psoriasis Foundation guidelines of care for the management and treatment of psoriasis with phototherapy. J Am Acad Dermatol. 2019;81:775-804.
- Åkerla P, Pukkala E, Helminen M, et al. Skin cancer risk of narrow-band UV-B (TL-01) phototherapy: a multi-center registry study with 4,815 patients. Acta Derm Venereol. 2024;104:adv39927.
- Filoni A, Vestita M, Congedo M, et al. Association between psoriasis and vitamin D: duration of disease correlates with decreased vitamin D serum levels: an observational case-control study. Medicine (Baltimore). 2018;97:E11185.
- Scott JF, Das LM, Ahsanuddin S, et al. Oral vitamin D rapidly attenuates inflammation from sunburn: an interventional study. J Invest Dermatol. 2017;137:2078-2086.
Association Between Psoriasis and Sunburn Prevalence in US Adults
Association Between Psoriasis and Sunburn Prevalence in US Adults
PRACTICE POINTS
- It is important for dermatologists to encourage rigorous sun-safety practices in patients with psoriasis, particularly those aged 20 to 59 years.
- A thorough sunburn history should be taken for skin cancer risk assessment in patients with psoriasis.
The Post-PASI Era: Considering Comorbidities to Select Appropriate Systemic Psoriasis Treatments
The Post-PASI Era: Considering Comorbidities to Select Appropriate Systemic Psoriasis Treatments
Psoriasis treatments have come a long way in the past 20 years. We now have more than a dozen systemic targeted treatments for psoriatic disease, with more on the way; however, with each successive class of medications introduced, the gap has narrowed in terms of increasing efficacy. In an era of medications reporting complete clearance rates in the 70% range, the average improvement in Psoriasis Area and Severity Index (PASI) for most biologics has remained at 90% to 95% in the past half-decade. While this is a far cry from the mean PASI improvements of 70% seen with the first biologics,1 it is becoming more challenging to base our treatment decisions solely on PASI outcome measures.
How, then, do we approach rational selection of a systemic psoriasis treatment? We could try to delineate based on mechanism of action, but it may be disingenuous to dissect minor differences in pathways (eg, IL-17 vs IL-23) that are fundamentally related and on the same continuum in psoriasis pathophysiology. Therefore, the most meaningful way to select an appropriate therapeutic may be to adopt a patient-centered approach that accounts for both individual preferences and specific medical needs by evaluating for other comorbidities2 to exclude or select certain medicines or types of treatments. We have long known to avoid tumor necrosis factor (TNF) α inhibitors in patients with congestive heart failure or a history of demyelinating disorders while regularly considering the presence of psoriatic arthritis and family planning when making treatment decisions. Now, we can be more nuanced in our approaches to psoriasis biologics. Specifically, the most important comorbidities to consider broadly encompass cardiometabolic disorders, gastrointestinal conditions, and psychiatric conditions.
Cardiometabolic Disorders
Possibly the hottest topic in psoriasis for some years now, the relationship between cardiometabolic disorders and psoriasis is of great interest to clinicians, scientists, and patients alike. There is a clear link between development of atherosclerosis and Th17-related immune mechanisms that also are implicated in the pathogenesis of psoriasis.3 Furthermore, the incidence of cardiovascular disease is markedly increased in patients with psoriasis, which is an independent risk factor for myocardial infarction, particularly among younger patients.4,5 Although several retrospective studies6-8 have shown that TNF-α inhibitors are associated with a reduction in cardiovascular outcomes, it is yet to be seen whether biologic treatment actually has a direct impact on cardiovascular outcomes, multiple studies investigating the effect of biologics on arterial inflammation markers notwithstanding.9
There are some direct factors to keep in mind when considering cardiometabolic comorbidities in patients with psoriasis. Obesity is common in the psoriasis population and can have a direct negative effect on cardiovascular health.10 However, the data on obesity and psoriasis are somewhat mixed with regard to treatment outcomes. In general, with increased volumes of distribution for biologics in patients with obesity, it has been shown that treatment success is more difficult to achieve in those with a body mass index greater than 30.11 Rather surprisingly, a separate nationwide study in South Korea found that patients on biologics for psoriasis were more likely to experience weight gain, even after controlling for factors such as exercise, smoking, and drinking,12 but it is unclear whether this is driven mostly by a known connection between weight gain and TNF-α inhibitors.13 These contrasting results point to the need for further studies in this area, as our intuitive approach would involve promoting weight loss while starting on a systemic treatment for psoriasis—but perhaps it is important not to assume that one will come with the other in tow, reinforcing the need to discuss a healthy diet with our patients with psoriasis regardless of treatment decisions.
The data that we have do not directly answer the big questions about biologic treatment and cardiovascular health, but we are starting to see interesting signals. For example, in a report of tildrakizumab treatment in patients with and without metabolic syndrome, the rates of major adverse cardiovascular events as well as cardiac disorders were essentially the same in both groups after receiving treatment for up to 244 weeks.14 This is interesting, more because of the lack of an increase in cardiovascular adverse events in the metabolic syndrome group, who entered the trial on average 25 kg to 30 kg heavier than those without metabolic syndrome. There is an increased risk for adverse cardiovascular events among patients with metabolic syndrome, a roughly 2-fold relative risk in as few as 5 to 6 years of follow-up.15 While the cohorts in the tildrakizumab study14 were too small to draw firm conclusions, the data are interesting and a step in the right direction; we need much larger data sets for analysis. Among other agents, similar efficacy and safety have been reported for guselkumab in a long-term psoriasis study; as a class, IL-23 inhibitors also tend to perform well from an efficacy standpoint in patients with obesity.16
Overall, when assessing the evidence for cardiometabolic disorders, it is reasonable to consider starting a biologic from the IL-17 or IL-23 inhibitor classes— thus avoiding both the potential downside of weight gain and contraindication in patients with congestive heart failure associated with TNF-α inhibitors. It is important to counsel patients about weight loss in conjunction with these treatments, both to improve efficacy and reduce cardiovascular risk factors. There may be a preference for IL-23 inhibitors in patients with obesity, as this class of medications maintains efficacy particularly well in these patients. Patients with psoriasis should be counseled to follow up with a primary care physician given their higher risk for metabolic syndrome and adverse cardiovascular outcomes.
Gastrointestinal Conditions
Psoriasis and inflammatory bowel disease (IBD) have a bidirectional association, and patients with psoriasis are about 1.7 times more likely to have either Crohn disease or ulcerative colitis.17,18 This association may be related to a shared pathogenesis with regard to immune dysregulation and overactivated inflammatory pathways, but there are some important differences to consider from a therapeutic standpoint. Given the increased expression of IL-17 in patients with IBD,19 a phase II trial of secukinumab yielded surprising results—not only was secukinumab ineffective in treating Crohn disease, but there also were higher rates of adverse events20 (as noted on the product label for all IL-17 inhibitors). We have come to understand that there are regulatory subsets of IL-17 cells that are important in mucosal homeostasis and also regulate IL-10, which generally is considered an anti-inflammatory cytokine.21 Thus, while IL-17 inhibition can reduce some component of inflammatory signaling, it also can increase inflammatory signaling through indirect pathways while increasing intestinal permeability to microbes. Importantly, this process seems to occur via IL-23–independent pathways; as such, while direct inhibition of IL-17 can be deleterious, IL-23 inhibitors have become important therapeutics for IBD.22
IL-17 family, IL-17A clearly is the culprit for worsening colitis as evidenced by both human and animal models. On the contrary, IL-17F blockade has been shown to ameliorate colitis in a murine model, whereas IL-17A inhibition worsens it.23 Furthermore, dual blockade of IL-17A and IL-17F has a protective effect against colitis, suggesting that the IL-17F inhibition is dominant. This interesting finding has some mechanistic backing, since blockade of IL-17F induces Treg cells that serve to maintain gut epithelium homeostasis and integrity.24
Overall, IL-17A inhibitors should be avoided in patients with a history of IBD—namely, secukinumab and ixekizumab. While there may be theoretical reasons that brodalumab or bimekizumab may confer a somewhat different risk for IBD exacerbation, there may be better choices that would be expected to effectively treat both the psoriasis and IBD manifestations. Given the US Food and Drug Administration approval of IL-23 inhibitors for IBD and their high levels of efficacy in treating psoriasis, these likely will be the best choice for most patients. Another mainstay of IBD treatment is TNF-α inhibitors, but they come with other risks such as increased immunosuppression and increased risk for nonmelanoma skin cancer.
An important question remains: What about patients who do not have known IBD? Do we proactively change our treatment choice due to fear of IBD development given the higher incidence of both Crohn disease and ulcerative colitis in patients with psoriasis? What about patients with a family history of IBD? First-degree relatives of patients with Crohn disease and ulcerative colitis have an 8- and 4-fold higher risk for those same conditions, respectively.25 Postmarketing surveillance and database findings of low rates of IBD development with IL-17 inhibitors gives only modest reassurance, as dermatologists generally know to avoid these medications for patients with even questionable IBD symptoms. It is important to emphasize to our patients that in no case do we believe that a psoriasis medication actually will cause IBD—rather, someone with subclinical IBD could experience a flare and a first manifestation of colitis. The drug is not the culprit in inducing IBD but rather may serve to unmask existing disease.
One study suggested that for patients who move on to the IL-17 inhibitor secukinumab after being treated with TNF-α inhibitors for psoriasis, the rates of IBD development are higher (4.8%) than in those who start IL-17A inhibition without prior treatment (1%)(OR, 8.38; P=.018).26 This begs the question of whether subclinical IBD in many patients with psoriasis who are treated with TNF-α inhibitors can be unmasked later when they are transitioned to a treatment that either does not treat the IBD or could worsen it. There may be a mechanistic drive behind this sequencing of treatments that predisposes patients to colitis, which would suggest selecting an IL-23 inhibitor after failing/trying a TNF-α inhibitor. However, the data are very preliminary, and in real practice, other concerns such as severe psoriatic arthritis may outweigh these considerations, as the IL-17 inhibitor class still is considered to be more effective than IL-23 inhibition at treating psoriatic arthritis overall. For most patients with no personal history of IBD and no strong family history of IBD (ie, first-degree relatives), the choice of biologic should not be affected by concern over gastrointestinal issues.
Psychiatric Conditions
It has been well established that psoriasis is linked to higher rates of depression, anxiety, and suicidality.27 How do we take this into account when treating patients with psoriasis, especially when we have biologics with a warning label for suicidality and a Risk Evaluation and Mitigation Strategies program (brodalumab) and language around suicidal ideation in the label (bimekizumab)? While it is challenging to discuss mental health, it is not a conversation that we as dermatologists should shy away from. Appropriate treatment of psoriasis is an important tool to get our patients on the path to better mental health. A recent database study of more than 4000 patients showed that patients with psoriasis treated with biologics had a 17% lower risk for depression than those treated with conventional disease-modifying drugs such as methotrexate.28 The comparator of the conventional disease-modifying drug class is important as it serves as a control for disease severity. Too often, a higher rate of depression, anxiety, or suicidality can be attributed to a medication when we in fact may just be capturing the background of higher incidence of all 3 in patients with severe psoriasis.
Indeed, even with the medication that many worry about on this front (brodalumab), multiple studies have confirmed that the effect on mental health generally is a positive one, with decreases in depressive symptoms.29 In a cohort switched from TNF-α inhibitors to brodalumab, symptoms of depression actually improved,30 so attributing a direct treatment effect to negative mental health outcomes does not seem to be justified, especially in light of the low number of suicide events in global postmarketing surveillance for brodalumab, comparable to or lower than other biologics for psoriasis.31 Similarly, bimekizumab has language in the label about discussing suicidality with patients, although the rates of suicidal ideation and behavior are no different from other biologics and rates of depression improved with its use.32
Heightened awareness of our patients’ mental health is something that we as providers should embrace, even when it seems that we do not have much time to see each patient. The priority when a patient comes in with mental health symptoms should be to treat what is within our scope (ie, psoriasis) as quickly and effectively as possible— with a newer-generation biologic such as an IL-17 or IL-23 inhibitor—while encouraging the patient to seek care from a mental health professional. In these cases, one might even argue that the rapidity of action of IL-17 inhibitors may be of additional benefit.
Final Thoughts
We as dermatologists generally are tasked with seeing high volumes of patients, and an initial psoriasis consultation can be a lengthy visit; however, it is rewarding to establish this relationship with patients and a reminder of why we practice medicine to begin with. Psoriasis can be satisfying to treat, and we have so many highly effective medicines that can completely transform our patients’ lives. Applying an understanding of the interplay between psoriasis, its related comorbidities, and treatment choices can be a fulfilling exercise that captures the essence of shared decision-making, which can lead to better outcomes and satisfaction for both providers and patients.
- Leonardi CL, Powers JL, Matheson RT, et al. Etanercept as monotherapy in patients with psoriasis. N Engl J Med. 2003;349:2014-2022. doi:10.1056/NEJMoa030409
- Thatiparthi A, Martin A, Liu J, et al. Biologic treatment algorithms for moderate-to-severe psoriasis with comorbid conditions and special populations: a review. Am J Clin Dermatol. 2021;22:425-442. doi:10.1007/s40257-021-00603-w
- Packard RR, Lichtman AH, Libby P. Innate and adaptive immunity in atherosclerosis. Semin Immunopathol. 2009;31:5-22. doi:10.1007 /s00281-009-0153-8
- Gelfand JM, Neimann AL, Shin DB, et al. Risk of myocardial infarction in patients with psoriasis. JAMA. 2006;296:1735-1741. doi:10.1001/jama.296.14.1735
- Miller IM, Ellervik C, Yazdanyar S, et al. Meta-analysis of psoriasis, cardiovascular disease, and associated risk factors. J Am Acad Dermatol. 2013;69:1014-1024. doi:10.1016/j.jaad.2013.06.053
- Wu JJ, Guerin A, Sundaram M, et al. Cardiovascular event risk assessment in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus methotrexate. J Am Acad Dermatol. 2017;76:81-90. doi:10.1016/j.jaad.2016.07.042
- Wu JJ, Poon KY, Channual JC, et al. Association between tumor necrosis factor inhibitor therapy and myocardial infarction risk in patients with psoriasis. Arch Dermatol. 2012;148:1244-1250. doi:10.1001 /archdermatol.2012.2502
- Wu JJ, Sundaram M, Cloutier M, et al. The risk of cardiovascular events in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus phototherapy: an observational cohort study. J Am Acad Dermatol. 2018;79:60-68. doi:10.1016/j.jaad.2018.02.050
- Cai J, Cui L, Wang Y, et al. Cardiometabolic comorbidities in patients with psoriasis: focusing on risk, biological therapy, and pathogenesis. Front Pharmacol. 2021;12:774808. doi:10.3389/fphar.2021.774808
- Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:E984-E1010. doi:10.1161/CIR.0000000000000973
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925. doi:10.1007 /s40261-021-01080-z
- Kim H, Hong JY, Cheong S, et al. Impact of biologic agents on body weight and obesity-related disorders in patients with psoriasis: a nationwide population-based cohort study. Obes Res Clin Pract. 2023;17:210-217. doi:10.1016/j.orcp.2023.05.004
- Saraceno R, Schipani C, Mazzotta A, et al. Effect of anti-tumor necrosis factor-alpha therapies on body mass index in patients with psoriasis. Pharmacol Res. 2008;57:290-295. doi:10.1016/j.phrs.2008.02.006
- Fernandez AP, Dauden E, Gerdes S, et al. Tildrakizumab efficacy and safety in patients with psoriasis and concomitant metabolic syndrome: post hoc analysis of 5-year data from reSURFACE 1 and reSURFACE 2. J Eur Acad Dermatol Venereol. 2022;36:1774-1783. doi:10.1111/jdv.18167
- Mottillo S, Filion KB, Genest J, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113-1132. doi:10.1016/j.jacc.2010.05.034
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti-IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Alinaghi F, Tekin HG, Burisch J, et al. Global prevalence and bidirectional association between psoriasis and inflammatory bowel disease— a systematic review and meta-analysis. J Crohns Colitis. 2020;14:351-360. doi:10.1093/ecco-jcc/jjz152
- Fu Y, Lee CH, Chi CC. Association of psoriasis with inflammatory bowel disease: a systematic review and meta-analysis. JAMA Dermatol. 2018;154:1417-1423. doi:10.1001/jamadermatol.2018.3631
- Fujino S, Andoh A, Bamba S, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut. 2003;52:65-70. doi:10.1136/gut.52.1.65
- Hueber W, Sands BE, Lewitzky S, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: unexpected results of a randomised, double-blind placebocontrolled trial. Gut. 2012;61:1693-1700. doi:10.1136 /gutjnl-2011-301668
- Brockmann L, Tran A, Huang Y, et al. Intestinal microbiotaspecific Th17 cells possess regulatory properties and suppress effector T cells via c-MAF and IL-10. Immunity. 2023;56:2719-2735 e7. doi:10.1016/j.immuni.2023.11.003
- Lee JS, Tato CM, Joyce-Shaikh B, et al. Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity. 2015;43:727-738. doi:10.1016/j.immuni.2015.09.003
- Wedebye Schmidt EG, Larsen HL, Kristensen NN, et al. TH17 cell induction and effects of IL-17A and IL-17F blockade in experimental colitis. Inflamm Bowel Dis. 2013;19:1567-1576. doi:10.1097 /MIB.0b013e318286fa1c
- Tang C, Kakuta S, Shimizu K, et al. Suppression of IL-17F, but not of IL-17A, provides protection against colitis by inducing T(reg) cells through modification of the intestinal microbiota. Nat Immunol. 2018;19:755-765. doi:10.1038/s41590-018-0134-y
- El Hadad J, Schreiner P, Vavricka SR, Greuter T. The genetics of inflammatory bowel disease. Mol Diagn Ther. 2024;28:27-35. doi:10.1007 /s40291-023-00678-7
- Albayrak F, Gür M, Karatas¸ A, et al. Is the use of secukinumab after anti-TNF therapy greater than expected for the risk of developing inflammatory bowel disease? Reumatol Clin (Engl Ed). 2024;20:123-127. doi:10.1016/j.reumae.2023.11.002
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a populationbased cohort study. Arch Dermatol. 2010;146:891-895. doi:10.1001 /archdermatol.2010.186
- Strober B, Soliman AM, Truong B, et al. Association between biologic exposure and the risk of depression in patients with psoriasis: a retrospective analysis of large US administrative claims data. Am J Clin Dermatol. 2024;25:853-856. doi:10.1007/s40257 -024-00877-w
- Koo J, Ho RS, Thibodeaux Q. Depression and suicidality in psoriasis and clinical studies of brodalumab: a narrative review. Cutis. 2019;104:361-365.
- Andersch-Bjorkman Y, Micu E, Seifert O, et al. Effects of brodalumab on psoriasis and depressive symptoms in patients with insufficient response to TNF-alpha inhibitors. J Dermatol. 2023;50:1401-1414. doi:10.1111/1346-8138.16917
- Yeroushalmi S, Chung M, Bartholomew E, et al. Examining worldwide postmarketing suicides from biologics used for psoriasis with a focus on brodalumab: a cross-sectional analysis using the Food and Drug Administration Adverse Event Reporting System (FAERS). JAAD Int. 2022;9:119-121. doi:10.1016/j.jdin.2022.08.010
- Blauvelt A, Armstrong A, Merola JF, et al. Mental health outcomes in patients with moderate to severe psoriasis treated with bimekizumab: analysis of phase 2/3 randomized trials. J Am Acad Dermatol. 2024;91:72-81. doi:10.1016/j.jaad.2024.02.039
Psoriasis treatments have come a long way in the past 20 years. We now have more than a dozen systemic targeted treatments for psoriatic disease, with more on the way; however, with each successive class of medications introduced, the gap has narrowed in terms of increasing efficacy. In an era of medications reporting complete clearance rates in the 70% range, the average improvement in Psoriasis Area and Severity Index (PASI) for most biologics has remained at 90% to 95% in the past half-decade. While this is a far cry from the mean PASI improvements of 70% seen with the first biologics,1 it is becoming more challenging to base our treatment decisions solely on PASI outcome measures.
How, then, do we approach rational selection of a systemic psoriasis treatment? We could try to delineate based on mechanism of action, but it may be disingenuous to dissect minor differences in pathways (eg, IL-17 vs IL-23) that are fundamentally related and on the same continuum in psoriasis pathophysiology. Therefore, the most meaningful way to select an appropriate therapeutic may be to adopt a patient-centered approach that accounts for both individual preferences and specific medical needs by evaluating for other comorbidities2 to exclude or select certain medicines or types of treatments. We have long known to avoid tumor necrosis factor (TNF) α inhibitors in patients with congestive heart failure or a history of demyelinating disorders while regularly considering the presence of psoriatic arthritis and family planning when making treatment decisions. Now, we can be more nuanced in our approaches to psoriasis biologics. Specifically, the most important comorbidities to consider broadly encompass cardiometabolic disorders, gastrointestinal conditions, and psychiatric conditions.
Cardiometabolic Disorders
Possibly the hottest topic in psoriasis for some years now, the relationship between cardiometabolic disorders and psoriasis is of great interest to clinicians, scientists, and patients alike. There is a clear link between development of atherosclerosis and Th17-related immune mechanisms that also are implicated in the pathogenesis of psoriasis.3 Furthermore, the incidence of cardiovascular disease is markedly increased in patients with psoriasis, which is an independent risk factor for myocardial infarction, particularly among younger patients.4,5 Although several retrospective studies6-8 have shown that TNF-α inhibitors are associated with a reduction in cardiovascular outcomes, it is yet to be seen whether biologic treatment actually has a direct impact on cardiovascular outcomes, multiple studies investigating the effect of biologics on arterial inflammation markers notwithstanding.9
There are some direct factors to keep in mind when considering cardiometabolic comorbidities in patients with psoriasis. Obesity is common in the psoriasis population and can have a direct negative effect on cardiovascular health.10 However, the data on obesity and psoriasis are somewhat mixed with regard to treatment outcomes. In general, with increased volumes of distribution for biologics in patients with obesity, it has been shown that treatment success is more difficult to achieve in those with a body mass index greater than 30.11 Rather surprisingly, a separate nationwide study in South Korea found that patients on biologics for psoriasis were more likely to experience weight gain, even after controlling for factors such as exercise, smoking, and drinking,12 but it is unclear whether this is driven mostly by a known connection between weight gain and TNF-α inhibitors.13 These contrasting results point to the need for further studies in this area, as our intuitive approach would involve promoting weight loss while starting on a systemic treatment for psoriasis—but perhaps it is important not to assume that one will come with the other in tow, reinforcing the need to discuss a healthy diet with our patients with psoriasis regardless of treatment decisions.
The data that we have do not directly answer the big questions about biologic treatment and cardiovascular health, but we are starting to see interesting signals. For example, in a report of tildrakizumab treatment in patients with and without metabolic syndrome, the rates of major adverse cardiovascular events as well as cardiac disorders were essentially the same in both groups after receiving treatment for up to 244 weeks.14 This is interesting, more because of the lack of an increase in cardiovascular adverse events in the metabolic syndrome group, who entered the trial on average 25 kg to 30 kg heavier than those without metabolic syndrome. There is an increased risk for adverse cardiovascular events among patients with metabolic syndrome, a roughly 2-fold relative risk in as few as 5 to 6 years of follow-up.15 While the cohorts in the tildrakizumab study14 were too small to draw firm conclusions, the data are interesting and a step in the right direction; we need much larger data sets for analysis. Among other agents, similar efficacy and safety have been reported for guselkumab in a long-term psoriasis study; as a class, IL-23 inhibitors also tend to perform well from an efficacy standpoint in patients with obesity.16
Overall, when assessing the evidence for cardiometabolic disorders, it is reasonable to consider starting a biologic from the IL-17 or IL-23 inhibitor classes— thus avoiding both the potential downside of weight gain and contraindication in patients with congestive heart failure associated with TNF-α inhibitors. It is important to counsel patients about weight loss in conjunction with these treatments, both to improve efficacy and reduce cardiovascular risk factors. There may be a preference for IL-23 inhibitors in patients with obesity, as this class of medications maintains efficacy particularly well in these patients. Patients with psoriasis should be counseled to follow up with a primary care physician given their higher risk for metabolic syndrome and adverse cardiovascular outcomes.
Gastrointestinal Conditions
Psoriasis and inflammatory bowel disease (IBD) have a bidirectional association, and patients with psoriasis are about 1.7 times more likely to have either Crohn disease or ulcerative colitis.17,18 This association may be related to a shared pathogenesis with regard to immune dysregulation and overactivated inflammatory pathways, but there are some important differences to consider from a therapeutic standpoint. Given the increased expression of IL-17 in patients with IBD,19 a phase II trial of secukinumab yielded surprising results—not only was secukinumab ineffective in treating Crohn disease, but there also were higher rates of adverse events20 (as noted on the product label for all IL-17 inhibitors). We have come to understand that there are regulatory subsets of IL-17 cells that are important in mucosal homeostasis and also regulate IL-10, which generally is considered an anti-inflammatory cytokine.21 Thus, while IL-17 inhibition can reduce some component of inflammatory signaling, it also can increase inflammatory signaling through indirect pathways while increasing intestinal permeability to microbes. Importantly, this process seems to occur via IL-23–independent pathways; as such, while direct inhibition of IL-17 can be deleterious, IL-23 inhibitors have become important therapeutics for IBD.22
IL-17 family, IL-17A clearly is the culprit for worsening colitis as evidenced by both human and animal models. On the contrary, IL-17F blockade has been shown to ameliorate colitis in a murine model, whereas IL-17A inhibition worsens it.23 Furthermore, dual blockade of IL-17A and IL-17F has a protective effect against colitis, suggesting that the IL-17F inhibition is dominant. This interesting finding has some mechanistic backing, since blockade of IL-17F induces Treg cells that serve to maintain gut epithelium homeostasis and integrity.24
Overall, IL-17A inhibitors should be avoided in patients with a history of IBD—namely, secukinumab and ixekizumab. While there may be theoretical reasons that brodalumab or bimekizumab may confer a somewhat different risk for IBD exacerbation, there may be better choices that would be expected to effectively treat both the psoriasis and IBD manifestations. Given the US Food and Drug Administration approval of IL-23 inhibitors for IBD and their high levels of efficacy in treating psoriasis, these likely will be the best choice for most patients. Another mainstay of IBD treatment is TNF-α inhibitors, but they come with other risks such as increased immunosuppression and increased risk for nonmelanoma skin cancer.
An important question remains: What about patients who do not have known IBD? Do we proactively change our treatment choice due to fear of IBD development given the higher incidence of both Crohn disease and ulcerative colitis in patients with psoriasis? What about patients with a family history of IBD? First-degree relatives of patients with Crohn disease and ulcerative colitis have an 8- and 4-fold higher risk for those same conditions, respectively.25 Postmarketing surveillance and database findings of low rates of IBD development with IL-17 inhibitors gives only modest reassurance, as dermatologists generally know to avoid these medications for patients with even questionable IBD symptoms. It is important to emphasize to our patients that in no case do we believe that a psoriasis medication actually will cause IBD—rather, someone with subclinical IBD could experience a flare and a first manifestation of colitis. The drug is not the culprit in inducing IBD but rather may serve to unmask existing disease.
One study suggested that for patients who move on to the IL-17 inhibitor secukinumab after being treated with TNF-α inhibitors for psoriasis, the rates of IBD development are higher (4.8%) than in those who start IL-17A inhibition without prior treatment (1%)(OR, 8.38; P=.018).26 This begs the question of whether subclinical IBD in many patients with psoriasis who are treated with TNF-α inhibitors can be unmasked later when they are transitioned to a treatment that either does not treat the IBD or could worsen it. There may be a mechanistic drive behind this sequencing of treatments that predisposes patients to colitis, which would suggest selecting an IL-23 inhibitor after failing/trying a TNF-α inhibitor. However, the data are very preliminary, and in real practice, other concerns such as severe psoriatic arthritis may outweigh these considerations, as the IL-17 inhibitor class still is considered to be more effective than IL-23 inhibition at treating psoriatic arthritis overall. For most patients with no personal history of IBD and no strong family history of IBD (ie, first-degree relatives), the choice of biologic should not be affected by concern over gastrointestinal issues.
Psychiatric Conditions
It has been well established that psoriasis is linked to higher rates of depression, anxiety, and suicidality.27 How do we take this into account when treating patients with psoriasis, especially when we have biologics with a warning label for suicidality and a Risk Evaluation and Mitigation Strategies program (brodalumab) and language around suicidal ideation in the label (bimekizumab)? While it is challenging to discuss mental health, it is not a conversation that we as dermatologists should shy away from. Appropriate treatment of psoriasis is an important tool to get our patients on the path to better mental health. A recent database study of more than 4000 patients showed that patients with psoriasis treated with biologics had a 17% lower risk for depression than those treated with conventional disease-modifying drugs such as methotrexate.28 The comparator of the conventional disease-modifying drug class is important as it serves as a control for disease severity. Too often, a higher rate of depression, anxiety, or suicidality can be attributed to a medication when we in fact may just be capturing the background of higher incidence of all 3 in patients with severe psoriasis.
Indeed, even with the medication that many worry about on this front (brodalumab), multiple studies have confirmed that the effect on mental health generally is a positive one, with decreases in depressive symptoms.29 In a cohort switched from TNF-α inhibitors to brodalumab, symptoms of depression actually improved,30 so attributing a direct treatment effect to negative mental health outcomes does not seem to be justified, especially in light of the low number of suicide events in global postmarketing surveillance for brodalumab, comparable to or lower than other biologics for psoriasis.31 Similarly, bimekizumab has language in the label about discussing suicidality with patients, although the rates of suicidal ideation and behavior are no different from other biologics and rates of depression improved with its use.32
Heightened awareness of our patients’ mental health is something that we as providers should embrace, even when it seems that we do not have much time to see each patient. The priority when a patient comes in with mental health symptoms should be to treat what is within our scope (ie, psoriasis) as quickly and effectively as possible— with a newer-generation biologic such as an IL-17 or IL-23 inhibitor—while encouraging the patient to seek care from a mental health professional. In these cases, one might even argue that the rapidity of action of IL-17 inhibitors may be of additional benefit.
Final Thoughts
We as dermatologists generally are tasked with seeing high volumes of patients, and an initial psoriasis consultation can be a lengthy visit; however, it is rewarding to establish this relationship with patients and a reminder of why we practice medicine to begin with. Psoriasis can be satisfying to treat, and we have so many highly effective medicines that can completely transform our patients’ lives. Applying an understanding of the interplay between psoriasis, its related comorbidities, and treatment choices can be a fulfilling exercise that captures the essence of shared decision-making, which can lead to better outcomes and satisfaction for both providers and patients.
Psoriasis treatments have come a long way in the past 20 years. We now have more than a dozen systemic targeted treatments for psoriatic disease, with more on the way; however, with each successive class of medications introduced, the gap has narrowed in terms of increasing efficacy. In an era of medications reporting complete clearance rates in the 70% range, the average improvement in Psoriasis Area and Severity Index (PASI) for most biologics has remained at 90% to 95% in the past half-decade. While this is a far cry from the mean PASI improvements of 70% seen with the first biologics,1 it is becoming more challenging to base our treatment decisions solely on PASI outcome measures.
How, then, do we approach rational selection of a systemic psoriasis treatment? We could try to delineate based on mechanism of action, but it may be disingenuous to dissect minor differences in pathways (eg, IL-17 vs IL-23) that are fundamentally related and on the same continuum in psoriasis pathophysiology. Therefore, the most meaningful way to select an appropriate therapeutic may be to adopt a patient-centered approach that accounts for both individual preferences and specific medical needs by evaluating for other comorbidities2 to exclude or select certain medicines or types of treatments. We have long known to avoid tumor necrosis factor (TNF) α inhibitors in patients with congestive heart failure or a history of demyelinating disorders while regularly considering the presence of psoriatic arthritis and family planning when making treatment decisions. Now, we can be more nuanced in our approaches to psoriasis biologics. Specifically, the most important comorbidities to consider broadly encompass cardiometabolic disorders, gastrointestinal conditions, and psychiatric conditions.
Cardiometabolic Disorders
Possibly the hottest topic in psoriasis for some years now, the relationship between cardiometabolic disorders and psoriasis is of great interest to clinicians, scientists, and patients alike. There is a clear link between development of atherosclerosis and Th17-related immune mechanisms that also are implicated in the pathogenesis of psoriasis.3 Furthermore, the incidence of cardiovascular disease is markedly increased in patients with psoriasis, which is an independent risk factor for myocardial infarction, particularly among younger patients.4,5 Although several retrospective studies6-8 have shown that TNF-α inhibitors are associated with a reduction in cardiovascular outcomes, it is yet to be seen whether biologic treatment actually has a direct impact on cardiovascular outcomes, multiple studies investigating the effect of biologics on arterial inflammation markers notwithstanding.9
There are some direct factors to keep in mind when considering cardiometabolic comorbidities in patients with psoriasis. Obesity is common in the psoriasis population and can have a direct negative effect on cardiovascular health.10 However, the data on obesity and psoriasis are somewhat mixed with regard to treatment outcomes. In general, with increased volumes of distribution for biologics in patients with obesity, it has been shown that treatment success is more difficult to achieve in those with a body mass index greater than 30.11 Rather surprisingly, a separate nationwide study in South Korea found that patients on biologics for psoriasis were more likely to experience weight gain, even after controlling for factors such as exercise, smoking, and drinking,12 but it is unclear whether this is driven mostly by a known connection between weight gain and TNF-α inhibitors.13 These contrasting results point to the need for further studies in this area, as our intuitive approach would involve promoting weight loss while starting on a systemic treatment for psoriasis—but perhaps it is important not to assume that one will come with the other in tow, reinforcing the need to discuss a healthy diet with our patients with psoriasis regardless of treatment decisions.
The data that we have do not directly answer the big questions about biologic treatment and cardiovascular health, but we are starting to see interesting signals. For example, in a report of tildrakizumab treatment in patients with and without metabolic syndrome, the rates of major adverse cardiovascular events as well as cardiac disorders were essentially the same in both groups after receiving treatment for up to 244 weeks.14 This is interesting, more because of the lack of an increase in cardiovascular adverse events in the metabolic syndrome group, who entered the trial on average 25 kg to 30 kg heavier than those without metabolic syndrome. There is an increased risk for adverse cardiovascular events among patients with metabolic syndrome, a roughly 2-fold relative risk in as few as 5 to 6 years of follow-up.15 While the cohorts in the tildrakizumab study14 were too small to draw firm conclusions, the data are interesting and a step in the right direction; we need much larger data sets for analysis. Among other agents, similar efficacy and safety have been reported for guselkumab in a long-term psoriasis study; as a class, IL-23 inhibitors also tend to perform well from an efficacy standpoint in patients with obesity.16
Overall, when assessing the evidence for cardiometabolic disorders, it is reasonable to consider starting a biologic from the IL-17 or IL-23 inhibitor classes— thus avoiding both the potential downside of weight gain and contraindication in patients with congestive heart failure associated with TNF-α inhibitors. It is important to counsel patients about weight loss in conjunction with these treatments, both to improve efficacy and reduce cardiovascular risk factors. There may be a preference for IL-23 inhibitors in patients with obesity, as this class of medications maintains efficacy particularly well in these patients. Patients with psoriasis should be counseled to follow up with a primary care physician given their higher risk for metabolic syndrome and adverse cardiovascular outcomes.
Gastrointestinal Conditions
Psoriasis and inflammatory bowel disease (IBD) have a bidirectional association, and patients with psoriasis are about 1.7 times more likely to have either Crohn disease or ulcerative colitis.17,18 This association may be related to a shared pathogenesis with regard to immune dysregulation and overactivated inflammatory pathways, but there are some important differences to consider from a therapeutic standpoint. Given the increased expression of IL-17 in patients with IBD,19 a phase II trial of secukinumab yielded surprising results—not only was secukinumab ineffective in treating Crohn disease, but there also were higher rates of adverse events20 (as noted on the product label for all IL-17 inhibitors). We have come to understand that there are regulatory subsets of IL-17 cells that are important in mucosal homeostasis and also regulate IL-10, which generally is considered an anti-inflammatory cytokine.21 Thus, while IL-17 inhibition can reduce some component of inflammatory signaling, it also can increase inflammatory signaling through indirect pathways while increasing intestinal permeability to microbes. Importantly, this process seems to occur via IL-23–independent pathways; as such, while direct inhibition of IL-17 can be deleterious, IL-23 inhibitors have become important therapeutics for IBD.22
IL-17 family, IL-17A clearly is the culprit for worsening colitis as evidenced by both human and animal models. On the contrary, IL-17F blockade has been shown to ameliorate colitis in a murine model, whereas IL-17A inhibition worsens it.23 Furthermore, dual blockade of IL-17A and IL-17F has a protective effect against colitis, suggesting that the IL-17F inhibition is dominant. This interesting finding has some mechanistic backing, since blockade of IL-17F induces Treg cells that serve to maintain gut epithelium homeostasis and integrity.24
Overall, IL-17A inhibitors should be avoided in patients with a history of IBD—namely, secukinumab and ixekizumab. While there may be theoretical reasons that brodalumab or bimekizumab may confer a somewhat different risk for IBD exacerbation, there may be better choices that would be expected to effectively treat both the psoriasis and IBD manifestations. Given the US Food and Drug Administration approval of IL-23 inhibitors for IBD and their high levels of efficacy in treating psoriasis, these likely will be the best choice for most patients. Another mainstay of IBD treatment is TNF-α inhibitors, but they come with other risks such as increased immunosuppression and increased risk for nonmelanoma skin cancer.
An important question remains: What about patients who do not have known IBD? Do we proactively change our treatment choice due to fear of IBD development given the higher incidence of both Crohn disease and ulcerative colitis in patients with psoriasis? What about patients with a family history of IBD? First-degree relatives of patients with Crohn disease and ulcerative colitis have an 8- and 4-fold higher risk for those same conditions, respectively.25 Postmarketing surveillance and database findings of low rates of IBD development with IL-17 inhibitors gives only modest reassurance, as dermatologists generally know to avoid these medications for patients with even questionable IBD symptoms. It is important to emphasize to our patients that in no case do we believe that a psoriasis medication actually will cause IBD—rather, someone with subclinical IBD could experience a flare and a first manifestation of colitis. The drug is not the culprit in inducing IBD but rather may serve to unmask existing disease.
One study suggested that for patients who move on to the IL-17 inhibitor secukinumab after being treated with TNF-α inhibitors for psoriasis, the rates of IBD development are higher (4.8%) than in those who start IL-17A inhibition without prior treatment (1%)(OR, 8.38; P=.018).26 This begs the question of whether subclinical IBD in many patients with psoriasis who are treated with TNF-α inhibitors can be unmasked later when they are transitioned to a treatment that either does not treat the IBD or could worsen it. There may be a mechanistic drive behind this sequencing of treatments that predisposes patients to colitis, which would suggest selecting an IL-23 inhibitor after failing/trying a TNF-α inhibitor. However, the data are very preliminary, and in real practice, other concerns such as severe psoriatic arthritis may outweigh these considerations, as the IL-17 inhibitor class still is considered to be more effective than IL-23 inhibition at treating psoriatic arthritis overall. For most patients with no personal history of IBD and no strong family history of IBD (ie, first-degree relatives), the choice of biologic should not be affected by concern over gastrointestinal issues.
Psychiatric Conditions
It has been well established that psoriasis is linked to higher rates of depression, anxiety, and suicidality.27 How do we take this into account when treating patients with psoriasis, especially when we have biologics with a warning label for suicidality and a Risk Evaluation and Mitigation Strategies program (brodalumab) and language around suicidal ideation in the label (bimekizumab)? While it is challenging to discuss mental health, it is not a conversation that we as dermatologists should shy away from. Appropriate treatment of psoriasis is an important tool to get our patients on the path to better mental health. A recent database study of more than 4000 patients showed that patients with psoriasis treated with biologics had a 17% lower risk for depression than those treated with conventional disease-modifying drugs such as methotrexate.28 The comparator of the conventional disease-modifying drug class is important as it serves as a control for disease severity. Too often, a higher rate of depression, anxiety, or suicidality can be attributed to a medication when we in fact may just be capturing the background of higher incidence of all 3 in patients with severe psoriasis.
Indeed, even with the medication that many worry about on this front (brodalumab), multiple studies have confirmed that the effect on mental health generally is a positive one, with decreases in depressive symptoms.29 In a cohort switched from TNF-α inhibitors to brodalumab, symptoms of depression actually improved,30 so attributing a direct treatment effect to negative mental health outcomes does not seem to be justified, especially in light of the low number of suicide events in global postmarketing surveillance for brodalumab, comparable to or lower than other biologics for psoriasis.31 Similarly, bimekizumab has language in the label about discussing suicidality with patients, although the rates of suicidal ideation and behavior are no different from other biologics and rates of depression improved with its use.32
Heightened awareness of our patients’ mental health is something that we as providers should embrace, even when it seems that we do not have much time to see each patient. The priority when a patient comes in with mental health symptoms should be to treat what is within our scope (ie, psoriasis) as quickly and effectively as possible— with a newer-generation biologic such as an IL-17 or IL-23 inhibitor—while encouraging the patient to seek care from a mental health professional. In these cases, one might even argue that the rapidity of action of IL-17 inhibitors may be of additional benefit.
Final Thoughts
We as dermatologists generally are tasked with seeing high volumes of patients, and an initial psoriasis consultation can be a lengthy visit; however, it is rewarding to establish this relationship with patients and a reminder of why we practice medicine to begin with. Psoriasis can be satisfying to treat, and we have so many highly effective medicines that can completely transform our patients’ lives. Applying an understanding of the interplay between psoriasis, its related comorbidities, and treatment choices can be a fulfilling exercise that captures the essence of shared decision-making, which can lead to better outcomes and satisfaction for both providers and patients.
- Leonardi CL, Powers JL, Matheson RT, et al. Etanercept as monotherapy in patients with psoriasis. N Engl J Med. 2003;349:2014-2022. doi:10.1056/NEJMoa030409
- Thatiparthi A, Martin A, Liu J, et al. Biologic treatment algorithms for moderate-to-severe psoriasis with comorbid conditions and special populations: a review. Am J Clin Dermatol. 2021;22:425-442. doi:10.1007/s40257-021-00603-w
- Packard RR, Lichtman AH, Libby P. Innate and adaptive immunity in atherosclerosis. Semin Immunopathol. 2009;31:5-22. doi:10.1007 /s00281-009-0153-8
- Gelfand JM, Neimann AL, Shin DB, et al. Risk of myocardial infarction in patients with psoriasis. JAMA. 2006;296:1735-1741. doi:10.1001/jama.296.14.1735
- Miller IM, Ellervik C, Yazdanyar S, et al. Meta-analysis of psoriasis, cardiovascular disease, and associated risk factors. J Am Acad Dermatol. 2013;69:1014-1024. doi:10.1016/j.jaad.2013.06.053
- Wu JJ, Guerin A, Sundaram M, et al. Cardiovascular event risk assessment in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus methotrexate. J Am Acad Dermatol. 2017;76:81-90. doi:10.1016/j.jaad.2016.07.042
- Wu JJ, Poon KY, Channual JC, et al. Association between tumor necrosis factor inhibitor therapy and myocardial infarction risk in patients with psoriasis. Arch Dermatol. 2012;148:1244-1250. doi:10.1001 /archdermatol.2012.2502
- Wu JJ, Sundaram M, Cloutier M, et al. The risk of cardiovascular events in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus phototherapy: an observational cohort study. J Am Acad Dermatol. 2018;79:60-68. doi:10.1016/j.jaad.2018.02.050
- Cai J, Cui L, Wang Y, et al. Cardiometabolic comorbidities in patients with psoriasis: focusing on risk, biological therapy, and pathogenesis. Front Pharmacol. 2021;12:774808. doi:10.3389/fphar.2021.774808
- Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:E984-E1010. doi:10.1161/CIR.0000000000000973
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925. doi:10.1007 /s40261-021-01080-z
- Kim H, Hong JY, Cheong S, et al. Impact of biologic agents on body weight and obesity-related disorders in patients with psoriasis: a nationwide population-based cohort study. Obes Res Clin Pract. 2023;17:210-217. doi:10.1016/j.orcp.2023.05.004
- Saraceno R, Schipani C, Mazzotta A, et al. Effect of anti-tumor necrosis factor-alpha therapies on body mass index in patients with psoriasis. Pharmacol Res. 2008;57:290-295. doi:10.1016/j.phrs.2008.02.006
- Fernandez AP, Dauden E, Gerdes S, et al. Tildrakizumab efficacy and safety in patients with psoriasis and concomitant metabolic syndrome: post hoc analysis of 5-year data from reSURFACE 1 and reSURFACE 2. J Eur Acad Dermatol Venereol. 2022;36:1774-1783. doi:10.1111/jdv.18167
- Mottillo S, Filion KB, Genest J, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113-1132. doi:10.1016/j.jacc.2010.05.034
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti-IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Alinaghi F, Tekin HG, Burisch J, et al. Global prevalence and bidirectional association between psoriasis and inflammatory bowel disease— a systematic review and meta-analysis. J Crohns Colitis. 2020;14:351-360. doi:10.1093/ecco-jcc/jjz152
- Fu Y, Lee CH, Chi CC. Association of psoriasis with inflammatory bowel disease: a systematic review and meta-analysis. JAMA Dermatol. 2018;154:1417-1423. doi:10.1001/jamadermatol.2018.3631
- Fujino S, Andoh A, Bamba S, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut. 2003;52:65-70. doi:10.1136/gut.52.1.65
- Hueber W, Sands BE, Lewitzky S, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: unexpected results of a randomised, double-blind placebocontrolled trial. Gut. 2012;61:1693-1700. doi:10.1136 /gutjnl-2011-301668
- Brockmann L, Tran A, Huang Y, et al. Intestinal microbiotaspecific Th17 cells possess regulatory properties and suppress effector T cells via c-MAF and IL-10. Immunity. 2023;56:2719-2735 e7. doi:10.1016/j.immuni.2023.11.003
- Lee JS, Tato CM, Joyce-Shaikh B, et al. Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity. 2015;43:727-738. doi:10.1016/j.immuni.2015.09.003
- Wedebye Schmidt EG, Larsen HL, Kristensen NN, et al. TH17 cell induction and effects of IL-17A and IL-17F blockade in experimental colitis. Inflamm Bowel Dis. 2013;19:1567-1576. doi:10.1097 /MIB.0b013e318286fa1c
- Tang C, Kakuta S, Shimizu K, et al. Suppression of IL-17F, but not of IL-17A, provides protection against colitis by inducing T(reg) cells through modification of the intestinal microbiota. Nat Immunol. 2018;19:755-765. doi:10.1038/s41590-018-0134-y
- El Hadad J, Schreiner P, Vavricka SR, Greuter T. The genetics of inflammatory bowel disease. Mol Diagn Ther. 2024;28:27-35. doi:10.1007 /s40291-023-00678-7
- Albayrak F, Gür M, Karatas¸ A, et al. Is the use of secukinumab after anti-TNF therapy greater than expected for the risk of developing inflammatory bowel disease? Reumatol Clin (Engl Ed). 2024;20:123-127. doi:10.1016/j.reumae.2023.11.002
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a populationbased cohort study. Arch Dermatol. 2010;146:891-895. doi:10.1001 /archdermatol.2010.186
- Strober B, Soliman AM, Truong B, et al. Association between biologic exposure and the risk of depression in patients with psoriasis: a retrospective analysis of large US administrative claims data. Am J Clin Dermatol. 2024;25:853-856. doi:10.1007/s40257 -024-00877-w
- Koo J, Ho RS, Thibodeaux Q. Depression and suicidality in psoriasis and clinical studies of brodalumab: a narrative review. Cutis. 2019;104:361-365.
- Andersch-Bjorkman Y, Micu E, Seifert O, et al. Effects of brodalumab on psoriasis and depressive symptoms in patients with insufficient response to TNF-alpha inhibitors. J Dermatol. 2023;50:1401-1414. doi:10.1111/1346-8138.16917
- Yeroushalmi S, Chung M, Bartholomew E, et al. Examining worldwide postmarketing suicides from biologics used for psoriasis with a focus on brodalumab: a cross-sectional analysis using the Food and Drug Administration Adverse Event Reporting System (FAERS). JAAD Int. 2022;9:119-121. doi:10.1016/j.jdin.2022.08.010
- Blauvelt A, Armstrong A, Merola JF, et al. Mental health outcomes in patients with moderate to severe psoriasis treated with bimekizumab: analysis of phase 2/3 randomized trials. J Am Acad Dermatol. 2024;91:72-81. doi:10.1016/j.jaad.2024.02.039
- Leonardi CL, Powers JL, Matheson RT, et al. Etanercept as monotherapy in patients with psoriasis. N Engl J Med. 2003;349:2014-2022. doi:10.1056/NEJMoa030409
- Thatiparthi A, Martin A, Liu J, et al. Biologic treatment algorithms for moderate-to-severe psoriasis with comorbid conditions and special populations: a review. Am J Clin Dermatol. 2021;22:425-442. doi:10.1007/s40257-021-00603-w
- Packard RR, Lichtman AH, Libby P. Innate and adaptive immunity in atherosclerosis. Semin Immunopathol. 2009;31:5-22. doi:10.1007 /s00281-009-0153-8
- Gelfand JM, Neimann AL, Shin DB, et al. Risk of myocardial infarction in patients with psoriasis. JAMA. 2006;296:1735-1741. doi:10.1001/jama.296.14.1735
- Miller IM, Ellervik C, Yazdanyar S, et al. Meta-analysis of psoriasis, cardiovascular disease, and associated risk factors. J Am Acad Dermatol. 2013;69:1014-1024. doi:10.1016/j.jaad.2013.06.053
- Wu JJ, Guerin A, Sundaram M, et al. Cardiovascular event risk assessment in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus methotrexate. J Am Acad Dermatol. 2017;76:81-90. doi:10.1016/j.jaad.2016.07.042
- Wu JJ, Poon KY, Channual JC, et al. Association between tumor necrosis factor inhibitor therapy and myocardial infarction risk in patients with psoriasis. Arch Dermatol. 2012;148:1244-1250. doi:10.1001 /archdermatol.2012.2502
- Wu JJ, Sundaram M, Cloutier M, et al. The risk of cardiovascular events in psoriasis patients treated with tumor necrosis factor-alpha inhibitors versus phototherapy: an observational cohort study. J Am Acad Dermatol. 2018;79:60-68. doi:10.1016/j.jaad.2018.02.050
- Cai J, Cui L, Wang Y, et al. Cardiometabolic comorbidities in patients with psoriasis: focusing on risk, biological therapy, and pathogenesis. Front Pharmacol. 2021;12:774808. doi:10.3389/fphar.2021.774808
- Powell-Wiley TM, Poirier P, Burke LE, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:E984-E1010. doi:10.1161/CIR.0000000000000973
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925. doi:10.1007 /s40261-021-01080-z
- Kim H, Hong JY, Cheong S, et al. Impact of biologic agents on body weight and obesity-related disorders in patients with psoriasis: a nationwide population-based cohort study. Obes Res Clin Pract. 2023;17:210-217. doi:10.1016/j.orcp.2023.05.004
- Saraceno R, Schipani C, Mazzotta A, et al. Effect of anti-tumor necrosis factor-alpha therapies on body mass index in patients with psoriasis. Pharmacol Res. 2008;57:290-295. doi:10.1016/j.phrs.2008.02.006
- Fernandez AP, Dauden E, Gerdes S, et al. Tildrakizumab efficacy and safety in patients with psoriasis and concomitant metabolic syndrome: post hoc analysis of 5-year data from reSURFACE 1 and reSURFACE 2. J Eur Acad Dermatol Venereol. 2022;36:1774-1783. doi:10.1111/jdv.18167
- Mottillo S, Filion KB, Genest J, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56:1113-1132. doi:10.1016/j.jacc.2010.05.034
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti-IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Alinaghi F, Tekin HG, Burisch J, et al. Global prevalence and bidirectional association between psoriasis and inflammatory bowel disease— a systematic review and meta-analysis. J Crohns Colitis. 2020;14:351-360. doi:10.1093/ecco-jcc/jjz152
- Fu Y, Lee CH, Chi CC. Association of psoriasis with inflammatory bowel disease: a systematic review and meta-analysis. JAMA Dermatol. 2018;154:1417-1423. doi:10.1001/jamadermatol.2018.3631
- Fujino S, Andoh A, Bamba S, et al. Increased expression of interleukin 17 in inflammatory bowel disease. Gut. 2003;52:65-70. doi:10.1136/gut.52.1.65
- Hueber W, Sands BE, Lewitzky S, et al. Secukinumab, a human anti-IL-17A monoclonal antibody, for moderate to severe Crohn’s disease: unexpected results of a randomised, double-blind placebocontrolled trial. Gut. 2012;61:1693-1700. doi:10.1136 /gutjnl-2011-301668
- Brockmann L, Tran A, Huang Y, et al. Intestinal microbiotaspecific Th17 cells possess regulatory properties and suppress effector T cells via c-MAF and IL-10. Immunity. 2023;56:2719-2735 e7. doi:10.1016/j.immuni.2023.11.003
- Lee JS, Tato CM, Joyce-Shaikh B, et al. Interleukin-23-independent IL-17 production regulates intestinal epithelial permeability. Immunity. 2015;43:727-738. doi:10.1016/j.immuni.2015.09.003
- Wedebye Schmidt EG, Larsen HL, Kristensen NN, et al. TH17 cell induction and effects of IL-17A and IL-17F blockade in experimental colitis. Inflamm Bowel Dis. 2013;19:1567-1576. doi:10.1097 /MIB.0b013e318286fa1c
- Tang C, Kakuta S, Shimizu K, et al. Suppression of IL-17F, but not of IL-17A, provides protection against colitis by inducing T(reg) cells through modification of the intestinal microbiota. Nat Immunol. 2018;19:755-765. doi:10.1038/s41590-018-0134-y
- El Hadad J, Schreiner P, Vavricka SR, Greuter T. The genetics of inflammatory bowel disease. Mol Diagn Ther. 2024;28:27-35. doi:10.1007 /s40291-023-00678-7
- Albayrak F, Gür M, Karatas¸ A, et al. Is the use of secukinumab after anti-TNF therapy greater than expected for the risk of developing inflammatory bowel disease? Reumatol Clin (Engl Ed). 2024;20:123-127. doi:10.1016/j.reumae.2023.11.002
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a populationbased cohort study. Arch Dermatol. 2010;146:891-895. doi:10.1001 /archdermatol.2010.186
- Strober B, Soliman AM, Truong B, et al. Association between biologic exposure and the risk of depression in patients with psoriasis: a retrospective analysis of large US administrative claims data. Am J Clin Dermatol. 2024;25:853-856. doi:10.1007/s40257 -024-00877-w
- Koo J, Ho RS, Thibodeaux Q. Depression and suicidality in psoriasis and clinical studies of brodalumab: a narrative review. Cutis. 2019;104:361-365.
- Andersch-Bjorkman Y, Micu E, Seifert O, et al. Effects of brodalumab on psoriasis and depressive symptoms in patients with insufficient response to TNF-alpha inhibitors. J Dermatol. 2023;50:1401-1414. doi:10.1111/1346-8138.16917
- Yeroushalmi S, Chung M, Bartholomew E, et al. Examining worldwide postmarketing suicides from biologics used for psoriasis with a focus on brodalumab: a cross-sectional analysis using the Food and Drug Administration Adverse Event Reporting System (FAERS). JAAD Int. 2022;9:119-121. doi:10.1016/j.jdin.2022.08.010
- Blauvelt A, Armstrong A, Merola JF, et al. Mental health outcomes in patients with moderate to severe psoriasis treated with bimekizumab: analysis of phase 2/3 randomized trials. J Am Acad Dermatol. 2024;91:72-81. doi:10.1016/j.jaad.2024.02.039
The Post-PASI Era: Considering Comorbidities to Select Appropriate Systemic Psoriasis Treatments
The Post-PASI Era: Considering Comorbidities to Select Appropriate Systemic Psoriasis Treatments
Treatment with Tildrakizumab Effective for Scalp Psoriasis in Phase 3b Study
TOPLINE:
METHODOLOGY:
- A 72-week, multicenter, randomized, double-blind, placebo-controlled phase 3b trial enrolled 231 patients with moderate to severe plaque psoriasis of the scalp.
- Patients were randomly assigned to receive placebo (n = 114) or tildrakizumab (n = 117) until week 16, when patients in the placebo group switched to receive tildrakizumab.
- The primary endpoint, Investigator Global Assessment modified 2011 (IGA) scalp response, was defined as a score of 0 (clear) or 1 (almost clear) or an improvement of at least two points at week 16.
- The treatment was stopped at week 52, and participants were observed for another 20 weeks for safety and tolerability.
TAKEAWAY:
- At week 16, the response rate was higher in the tildrakizumab group than in the placebo group (49.4% vs 7.3%; P < .00001), and it increased to 62.9% and 56.1% (after crossover), respectively, at week 52.
- Psoriasis Scalp Severity Index 90 (PSSI 90) response rates were 60.7% and 4.9% at week 16 in the tildrakizumab and placebo groups, rising to 65.2% and 57.3%, respectively, at week 52.
- More than 80% of the week 16 responders maintained IGA and PSSI 90 responses at week 52.
- More than 50% of patients in both groups experienced adverse events, with no treatment-related serious toxicity.
IN PRACTICE:
“Tildrakizumab maintains improvements in scalp psoriasis for up to 52 weeks,” the authors wrote.
SOURCE:
Howard L. Sofen, MD, University of California, Los Angeles, led the study, which was published online on December 22, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
This study excluded patients with predominantly scalp involvement and minimal whole body psoriasis, who might respond differently to the treatment. Results were obtained under controlled clinical conditions and may not be generalizable to clinical practice.
DISCLOSURES:
This study and analyses were funded by Sun Pharma. Sofen reported serving as a clinical investigator for various pharmaceutical companies, including Sun Pharma. Five authors were current or former employees of Sun Pharma and associated companies. Others also disclosed financial ties outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A 72-week, multicenter, randomized, double-blind, placebo-controlled phase 3b trial enrolled 231 patients with moderate to severe plaque psoriasis of the scalp.
- Patients were randomly assigned to receive placebo (n = 114) or tildrakizumab (n = 117) until week 16, when patients in the placebo group switched to receive tildrakizumab.
- The primary endpoint, Investigator Global Assessment modified 2011 (IGA) scalp response, was defined as a score of 0 (clear) or 1 (almost clear) or an improvement of at least two points at week 16.
- The treatment was stopped at week 52, and participants were observed for another 20 weeks for safety and tolerability.
TAKEAWAY:
- At week 16, the response rate was higher in the tildrakizumab group than in the placebo group (49.4% vs 7.3%; P < .00001), and it increased to 62.9% and 56.1% (after crossover), respectively, at week 52.
- Psoriasis Scalp Severity Index 90 (PSSI 90) response rates were 60.7% and 4.9% at week 16 in the tildrakizumab and placebo groups, rising to 65.2% and 57.3%, respectively, at week 52.
- More than 80% of the week 16 responders maintained IGA and PSSI 90 responses at week 52.
- More than 50% of patients in both groups experienced adverse events, with no treatment-related serious toxicity.
IN PRACTICE:
“Tildrakizumab maintains improvements in scalp psoriasis for up to 52 weeks,” the authors wrote.
SOURCE:
Howard L. Sofen, MD, University of California, Los Angeles, led the study, which was published online on December 22, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
This study excluded patients with predominantly scalp involvement and minimal whole body psoriasis, who might respond differently to the treatment. Results were obtained under controlled clinical conditions and may not be generalizable to clinical practice.
DISCLOSURES:
This study and analyses were funded by Sun Pharma. Sofen reported serving as a clinical investigator for various pharmaceutical companies, including Sun Pharma. Five authors were current or former employees of Sun Pharma and associated companies. Others also disclosed financial ties outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
TOPLINE:
METHODOLOGY:
- A 72-week, multicenter, randomized, double-blind, placebo-controlled phase 3b trial enrolled 231 patients with moderate to severe plaque psoriasis of the scalp.
- Patients were randomly assigned to receive placebo (n = 114) or tildrakizumab (n = 117) until week 16, when patients in the placebo group switched to receive tildrakizumab.
- The primary endpoint, Investigator Global Assessment modified 2011 (IGA) scalp response, was defined as a score of 0 (clear) or 1 (almost clear) or an improvement of at least two points at week 16.
- The treatment was stopped at week 52, and participants were observed for another 20 weeks for safety and tolerability.
TAKEAWAY:
- At week 16, the response rate was higher in the tildrakizumab group than in the placebo group (49.4% vs 7.3%; P < .00001), and it increased to 62.9% and 56.1% (after crossover), respectively, at week 52.
- Psoriasis Scalp Severity Index 90 (PSSI 90) response rates were 60.7% and 4.9% at week 16 in the tildrakizumab and placebo groups, rising to 65.2% and 57.3%, respectively, at week 52.
- More than 80% of the week 16 responders maintained IGA and PSSI 90 responses at week 52.
- More than 50% of patients in both groups experienced adverse events, with no treatment-related serious toxicity.
IN PRACTICE:
“Tildrakizumab maintains improvements in scalp psoriasis for up to 52 weeks,” the authors wrote.
SOURCE:
Howard L. Sofen, MD, University of California, Los Angeles, led the study, which was published online on December 22, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
This study excluded patients with predominantly scalp involvement and minimal whole body psoriasis, who might respond differently to the treatment. Results were obtained under controlled clinical conditions and may not be generalizable to clinical practice.
DISCLOSURES:
This study and analyses were funded by Sun Pharma. Sofen reported serving as a clinical investigator for various pharmaceutical companies, including Sun Pharma. Five authors were current or former employees of Sun Pharma and associated companies. Others also disclosed financial ties outside this work.
This article was created using several editorial tools, including artificial intelligence, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Cardiac Risks of Newer Psoriasis Biologics vs. TNF Inhibitors Compared
TOPLINE:
The newer biologics — .
METHODOLOGY:
- In a retrospective cohort study, researchers conducted an emulated target trial analysis using data of 32,098 biologic-naive patients with psoriasis or PsA who were treated with one of the newer biologics (infliximab, adalimumab, etanercept, certolizumab pegol, secukinumab, ixekizumab, brodalumab, ustekinumab, risankizumab, guselkumab, and tildrakizumab) from the TriNetX Research Network between 2014 and 2022.
- Patients received TNF inhibitors (n = 20,314), IL-17 inhibitors (n = 5073), IL-12/23 inhibitors (n = 3573), or IL-23 inhibitors (n = 3138).
- A propensity-matched analysis compared each class of newer biologics with TNF inhibitors, adjusting for demographics, comorbidities, and medication use.
- The primary outcomes were major adverse cardiovascular events (MACE; myocardial infarction and stroke) or venous thromboembolic events (VTE).
TAKEAWAY:
- Compared with patients who received TNF inhibitors, the risk for MACE was not significantly different between patients who received IL-17 inhibitors (incidence rate ratio [IRR], 1.14; 95% CI, 0.86-1.52), IL-12/23 inhibitors (IRR, 1.24; 95% CI, 0.84-1.78), or IL-23 inhibitors (IRR, 0.93; 95% CI, 0.61-1.38)
- The VTE risk was also not significantly different between patients who received IL-17 inhibitors (IRR, 1.12; 95% CI, 0.63-2.08), IL-12/23 inhibitors (IRR, 1.51; 95% CI, 0.73-3.19), or IL-23 inhibitors (IRR, 1.42; 95% CI, 0.64-3.25) compared with those who received TNF inhibitors.
- Subgroup analyses for psoriasis or psoriatic arthritis alone confirmed consistent findings.
- Patients with preexisting hyperlipidemia and diabetes mellitus showed lower risks for MACE and VTE with newer biologics compared with TNF inhibitors.
IN PRACTICE:
“No significant MACE and VTE risk differences were detected in patients with psoriasis or PsA between those receiving IL-17, IL-12/23, and IL-23 inhibitors and those with TNF inhibitors,” the authors concluded. These findings, they added “can be considered by physicians and patients when making treatment decisions” and also provide “evidence for future pharmacovigilance studies.”
SOURCE:
The study was led by Tai-Li Chen, MD, of the Department of Dermatology, Taipei Veterans General Hospital in Taipei, Taiwan. It was published online on December 27, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Study limitations included potential residual confounding factors, lack of information on disease severity, and inclusion of predominantly White individuals.
DISCLOSURES:
The study received support from Taipei Veterans General Hospital and Ministry of Science and Technology, Taiwan. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The newer biologics — .
METHODOLOGY:
- In a retrospective cohort study, researchers conducted an emulated target trial analysis using data of 32,098 biologic-naive patients with psoriasis or PsA who were treated with one of the newer biologics (infliximab, adalimumab, etanercept, certolizumab pegol, secukinumab, ixekizumab, brodalumab, ustekinumab, risankizumab, guselkumab, and tildrakizumab) from the TriNetX Research Network between 2014 and 2022.
- Patients received TNF inhibitors (n = 20,314), IL-17 inhibitors (n = 5073), IL-12/23 inhibitors (n = 3573), or IL-23 inhibitors (n = 3138).
- A propensity-matched analysis compared each class of newer biologics with TNF inhibitors, adjusting for demographics, comorbidities, and medication use.
- The primary outcomes were major adverse cardiovascular events (MACE; myocardial infarction and stroke) or venous thromboembolic events (VTE).
TAKEAWAY:
- Compared with patients who received TNF inhibitors, the risk for MACE was not significantly different between patients who received IL-17 inhibitors (incidence rate ratio [IRR], 1.14; 95% CI, 0.86-1.52), IL-12/23 inhibitors (IRR, 1.24; 95% CI, 0.84-1.78), or IL-23 inhibitors (IRR, 0.93; 95% CI, 0.61-1.38)
- The VTE risk was also not significantly different between patients who received IL-17 inhibitors (IRR, 1.12; 95% CI, 0.63-2.08), IL-12/23 inhibitors (IRR, 1.51; 95% CI, 0.73-3.19), or IL-23 inhibitors (IRR, 1.42; 95% CI, 0.64-3.25) compared with those who received TNF inhibitors.
- Subgroup analyses for psoriasis or psoriatic arthritis alone confirmed consistent findings.
- Patients with preexisting hyperlipidemia and diabetes mellitus showed lower risks for MACE and VTE with newer biologics compared with TNF inhibitors.
IN PRACTICE:
“No significant MACE and VTE risk differences were detected in patients with psoriasis or PsA between those receiving IL-17, IL-12/23, and IL-23 inhibitors and those with TNF inhibitors,” the authors concluded. These findings, they added “can be considered by physicians and patients when making treatment decisions” and also provide “evidence for future pharmacovigilance studies.”
SOURCE:
The study was led by Tai-Li Chen, MD, of the Department of Dermatology, Taipei Veterans General Hospital in Taipei, Taiwan. It was published online on December 27, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Study limitations included potential residual confounding factors, lack of information on disease severity, and inclusion of predominantly White individuals.
DISCLOSURES:
The study received support from Taipei Veterans General Hospital and Ministry of Science and Technology, Taiwan. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
TOPLINE:
The newer biologics — .
METHODOLOGY:
- In a retrospective cohort study, researchers conducted an emulated target trial analysis using data of 32,098 biologic-naive patients with psoriasis or PsA who were treated with one of the newer biologics (infliximab, adalimumab, etanercept, certolizumab pegol, secukinumab, ixekizumab, brodalumab, ustekinumab, risankizumab, guselkumab, and tildrakizumab) from the TriNetX Research Network between 2014 and 2022.
- Patients received TNF inhibitors (n = 20,314), IL-17 inhibitors (n = 5073), IL-12/23 inhibitors (n = 3573), or IL-23 inhibitors (n = 3138).
- A propensity-matched analysis compared each class of newer biologics with TNF inhibitors, adjusting for demographics, comorbidities, and medication use.
- The primary outcomes were major adverse cardiovascular events (MACE; myocardial infarction and stroke) or venous thromboembolic events (VTE).
TAKEAWAY:
- Compared with patients who received TNF inhibitors, the risk for MACE was not significantly different between patients who received IL-17 inhibitors (incidence rate ratio [IRR], 1.14; 95% CI, 0.86-1.52), IL-12/23 inhibitors (IRR, 1.24; 95% CI, 0.84-1.78), or IL-23 inhibitors (IRR, 0.93; 95% CI, 0.61-1.38)
- The VTE risk was also not significantly different between patients who received IL-17 inhibitors (IRR, 1.12; 95% CI, 0.63-2.08), IL-12/23 inhibitors (IRR, 1.51; 95% CI, 0.73-3.19), or IL-23 inhibitors (IRR, 1.42; 95% CI, 0.64-3.25) compared with those who received TNF inhibitors.
- Subgroup analyses for psoriasis or psoriatic arthritis alone confirmed consistent findings.
- Patients with preexisting hyperlipidemia and diabetes mellitus showed lower risks for MACE and VTE with newer biologics compared with TNF inhibitors.
IN PRACTICE:
“No significant MACE and VTE risk differences were detected in patients with psoriasis or PsA between those receiving IL-17, IL-12/23, and IL-23 inhibitors and those with TNF inhibitors,” the authors concluded. These findings, they added “can be considered by physicians and patients when making treatment decisions” and also provide “evidence for future pharmacovigilance studies.”
SOURCE:
The study was led by Tai-Li Chen, MD, of the Department of Dermatology, Taipei Veterans General Hospital in Taipei, Taiwan. It was published online on December 27, 2024, in the Journal of the American Academy of Dermatology.
LIMITATIONS:
Study limitations included potential residual confounding factors, lack of information on disease severity, and inclusion of predominantly White individuals.
DISCLOSURES:
The study received support from Taipei Veterans General Hospital and Ministry of Science and Technology, Taiwan. The authors reported no conflicts of interest.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article first appeared on Medscape.com.
Dermatologic Implications of Glycemic Control Medications for Patients with Type 2 Diabetes Mellitus
Dermatologic Implications of Glycemic Control Medications for Patients with Type 2 Diabetes Mellitus
Type 2 diabetes mellitus (T2DM) is a chronic disease characterized by uncontrolled hyperglycemia. Over the past few decades, its prevalence has steadily increased, now affecting approximately 10% of adults worldwide and ranking among the top 10 leading causes of death globally.1 The pathophysiology of T2DM involves persistent hyperglycemia that drives insulin resistance and a progressive decline in insulin production from the pancreas.2 Medical management of this condition aims to reduce blood glucose levels or enhance insulin production and sensitivity. Aside from lifestyle modifications, metformin is considered the first-line treatment for glycemic control according to the 2023 American Association of Clinical Endocrinology’s T2DM management algorithm.3 These updated guidelines stratify adjunct treatments by individualized glycemic targets and patient needs. For patients who are overweight or obese, glucagonlike peptide 1 (GLP-1) and dual GLP-1/ gastric inhibitory polypeptide (GIP) agonists are the preferred adjunct or second-line treatments.3
In this review, we highlight the dermatologic adverse effects and potential therapeutic benefits of metformin as well as GLP-1 and GLP-1/GIP agonists.
METFORMIN
Metformin is a biguanide agent used as a first-line treatment for T2DM because of its ability to reduce hepatic glucose production and increase peripheral tissue glucose uptake.4 In addition to its effects on glucose, metformin has been shown to have anti-inflammatory properties via inhibition of the nuclear factor κB and mammalian target of rapamycin (mTOR) pathways, leading to decreased production of cytokines associated with T helper (Th) 1 and Th17 cell responses, such as IL-17, interferon gamma (IFN-γ), and tumor necrosis factor α (TNF-α).5-7 These findings have spurred interest among clinicians in the potential use of metformin for inflammatory conditions, including dermatologic diseases such as psoriasis and hidradenitis suppurativa (HS).8
Adverse Effects
Metformin is administered orally and generally is well tolerated. The most common adverse effects include gastrointestinal symptoms such as diarrhea, nausea, vomiting, and abdominal pain.9 While cutaneous adverse effects are rare, multiple dermatologic adverse reactions to metformin have been reported,10,11 including leukocytoclastic vasculitis,11-13 fixed drug eruptions,14-17 drug rash with eosinophilia and systemic symptoms (DRESS) syndrome,18 and photosensitivity reactions.19 Leukocytoclastic vasculitis and DRESS syndrome typically develop within the first month following metformin initiation, while fixed drug eruption and photosensitivity reactions have more variable timing, occurring weeks to years after treatment initiation.12-19
Dermatologic Implications
Acanthosis Nigricans—Acanthosis nigricans (AN) is characterized by hyperpigmentation and velvety skin thickening, typically in intertriginous areas such as the back of the neck, axillae, and groin.20 It commonly is associated with insulin resistance and obesity.21-23 Treatments for AN primarily center around insulin sensitivity and weight loss,24,25 with some benefit observed from the use of keratolytic agents.26,27 Metformin may have utility in treating AN through its effects on insulin sensitivity and glycemic control. Multiple case reports have noted marked improvements in AN in patients with and without obesity with the addition of metformin to their existing treatment regimens in doses ranging from 500 mg to 1700 mg daily.28-30 However, an unblinded randomized controlled trial (RCT) comparing the efficacy of metformin (500 mg 3 times daily) with rosiglitazone (4 mg/d), another T2DM medication, on AN neck lesions in patients who were overweight and obese found no significant effects in lesion severity and only modest improvements in skin texture in both groups at 12 weeks following treatment initiation.31 Another RCT comparing metformin (500 mg twice daily) with a twice-daily capsule containing α-lipoic acid, biotin, chromium polynicotinate, and zinc sulfate, showed significant (P<.001) improvements in AN neck lesions in both groups after 12 weeks.32 According to Sung et al,8 longer duration of therapy (>6 months), higher doses (1700–2000 mg), and lower baseline weight were associated with higher efficacy of metformin for treatment of AN. Overall, the use of metformin as an adjunct treatment for AN, particularly in patients with underlying hyperglycemia, is supported in the literature, but further studies are needed to clarify dosing, duration of therapy, and patient populations that will benefit most from adding metformin to their treatment regimens.
Hirsutism—Hirsutism, which is characterized by excessive hair growth in androgen-dependent areas, can be challenging to treat. Metformin has been shown to reduce circulating insulin, luteinizing hormone, androstenedione, and testosterone, thus improving underlying hyperandrogenism, particularly in patients with polycystic ovary syndrome (PCOS).33-35 Although single studies evaluating the efficacy of metformin for treatment of hirsutism in patients with PCOS have shown potential benefits,36-38 meta-analyses showed no significant effects of metformin compared to placebo or oral contraceptives and decreased benefits compared to spironolactone and flutamide.39 Given these findings showing that metformin was no more effective than placebo or other treatments, the current Endocrine Society guidelines recommend against the use of metformin for hirsutism.39,40 There may be a role for metformin as an adjuvant therapy in certain populations (eg, patients with comorbid T2DM), although further studies stratifying risk factors such as body mass index and age are needed.41
Hidradenitis Suppurativa—Hidradenitis suppurativa is a follicular occlusive disease characterized by recurrent inflamed nodules leading to chronic dermal abscesses, fibrosis, and sinus tract formation primarily in intertriginous areas such as the axillae and groin.42 Medical management depends on disease severity but usually involves antibiotic treatment with adjunct therapies such as oral contraceptives, antiandrogenic medications (eg, spironolactone), biologic medications, and metformin.42 Preclinical and clinical data suggest that metformin can impact HS through metabolic and immunomodulatory mechanisms.5,42 Like many chronic inflammatory disorders, HS is associated with metabolic syndrome.43,44 A study evaluating insulin secretion after oral glucose tolerance testing showed increased insulin levels in patients with HS compared to controls (P=.02), with 60% (6/10) of patients with HS meeting criteria for insulin resistance. In addition, serum insulin levels in insulin-resistant patients with HS correlated with increased lesional skin mTOR gene expression at 30 (r=.80) and 60 (r=1.00) minutes, and mTOR was found to be upregulated in lesional and extralesional skin in patients with HS compared to healthy controls (P<.01).45 Insulin activates mTOR signaling, which mediates cell growth and survival, among other processes.46 Thus, metformin’s ability to increase insulin sensitivity and inhibit mTOR signaling could be beneficial in the setting of HS. Additionally, insulin and insulinlike growth factor 1 (IGF-1) increase androgen signaling, a process that has been implicated in HS.47
Metformin also may impact HS through its effects on testosterone and other hormones.48 A study evaluating peripheral blood mononuclear cells in patients with HS showed reduced IL-17, IFN-γ, TNF-α, and IL-6 levels in patients who were taking metformin (dose not reported) for longer than 6 months compared to patients who were not on metformin. Further analysis of ex vivo HS lesions cultured with metformin showed decreased IL-17, IFN-γ, TNF-α, and IL-8 expression in tissue, suggesting an antiinflammatory role of metformin in HS.5
Although there are no known RCTs assessing the efficacy of metformin in HS, existing clinical data are supportive of the use of metformin for refractory HS.49 Following a case report describing a patient with T2DM and stable HS while on metformin,50 several cohort studies have assessed the efficacy of metformin for the treatment of HS. A prospective study evaluating the efficacy of metformin monotherapy (starting dose of 500 mg/d, titrated to 500 mg 3 times daily) in patients with and without T2DM with HS refractory to other therapies found clinical improvement in 72% (18/25) of patients using the Sartorius Hidradenitis Suppurativa Score, improving from a mean (SD) score of 34.40 (12.46) to 26.76 (11.22) at 12 weeks (P=.0055,) and 22.39 (11.30) at 24 weeks (P=.0001). Additionally, 64% (16/25) of patients showed improved quality of life as evaluated by the Dermatology Life Quality Index (DLQI), which decreased from a mean (SD) score of 15.00 (4.96) to 10.08 (5.96)(P=.0017) at 12 weeks and 7.65 (7.12)(P=.000009) at 24 weeks on treatment.48 In a retrospective study of 53 patients with HS taking metformin started at 500 mg daily and increased to 500 mg twice daily after 2 weeks (when tolerated), 68% (36/53) showed some clinical response, with 19% (7/36) of those patients having achieved complete response to metformin monotherapy (defined as no active HS).51 Similarly, a retrospective study of pediatric patients with HS evaluating metformin (doses ranging from 500-2000 mg daily) as an adjunct therapy described a subset of patients with decreased frequency of HS flares with metformin.52 These studies emphasize the safety profile of metformin and support its current use as an adjunctive therapy for HS.
Acne Vulgaris—Acne vulgaris (AV) is a chronic inflammatory disorder affecting the pilosebaceous follicles.11 Similar to HS, AV has metabolic and hormonal influences that can be targeted by metformin.53 In AV, androgens lead to increased sebum production by binding to androgen receptors on sebocytes, which in turn attracts Cutibacterium acnes and promotes hyperkeratinization, inducing inflammation.54 Thus, the antiandrogenic effects of metformin may be beneficial for treatment of AV. Additionally, sebocytes express receptors for insulin and IGF-1, which can increase the size and number of sebocytes, as well as promote lipogenesis and inflammatory response, influencing sebum production.54 Serum levels for IGF-1 have been observed to be increased in patients with AV55 and reduced by metformin.56 A recent meta-analysis assessing the efficacy of metformin on AV indicated that 87% (13/15) of studies noted disease improvement on metformin, with 47% (7/15) of studies showing statistically significant (P<0.05) decreases in acne severity.57 Although most studies showed improvement, 47% (7/15) did not find significant differences between metformin and other interventions, indicating the availability of comparable treatment options. Overall, there has been a positive association between metformin use and acne improvement.57 However, it is important to note that most studies have focused on females with PCOS,57 and the main benefits of metformin in acne might be seen when managing comorbid conditions, particularly those associated with metabolic dysregulation and insulin resistance. Further studies are needed to determine the generalizability of prior results.
Psoriasis—Psoriasis is a chronic autoinflammatory disease characterized by epidermal hyperplasia with multiple cutaneous manifestations and potential for multiorgan involvement. Comorbid conditions include psoriatic arthritis, metabolic syndrome, and cardiovascular disease.58 Current treatment options depend on several factors (eg, disease severity, location of cutaneous lesions, comorbidities) and include topical, systemic, and phototherapy options, many of which target the immune system.58,59 A meta-analysis of 3 RCTs showed that metformin (500 mg/d or 1000 mg/d) was associated with significantly improved Psoriasis Area and Severity Index (PASI) 75% reductions (odds ratio [OR], 22.02; 95% CI, 2.12-228.49; P=.01) and 75% reductions in erythema, scaling, and induration (OR, 9.12; 95% CI, 2.13-39.02; P=.003) compared to placebo.60 In addition, an RCT evaluating the efficacy of metformin (1000 mg/d) or pioglitazone (30 mg/d) for 12 weeks in patients with psoriasis with metabolic syndrome found significant improvements in PASI75 (P=.001) and erythema, scaling, and induration (P=.016) scores as well as in Physician Global Assessment scores (P=.012) compared to placebo and no differences compared to pioglitazone.61 While current psoriasis management guidelines do not include metformin, its use may be worth consideration as an adjunct therapy in patients with psoriasis and comorbidities such as T2DM and metabolic syndrome.59 Metformin’s potential benefits in psoriasis may lie outside its metabolic influences and occur secondary to its immunomodulatory effects, including targeting of the Th17 axis or cytokine-specific pathways such as TNF-α, which are known to be involved in psoriasis pathogenesis.58
Central Centrifugal Cicatricial Alopecia—Central centrifugal cicatricial alopecia (CCCA) is a form of scarring alopecia characterized by chronic inflammation leading to permanent loss of hair follicles on the crown of the scalp.62 Current treatments include topical and intralesional corticosteroids, as well as oral antibiotics. In addition, therapies including the antimalarial hydroxychloroquine and immunosuppressants mycophenolate and cyclosporine are used in refractory disease.63,64 A case report described 2 patients with hair regrowth after 4 and 6 months of treatment with topical metformin 10% compounded in a proprietary transdermal vehicle.65 The authors speculated that metformin’s effects on CCCA could be attributed to its known agonistic effects on the adenosine monophosphate-activated protein kinase (AMPK) pathway with subsequent reduction in inflammation-induced fibrosis.65,66 Microarray67 and proteomic68 analysis have shown that AMPK is known to be downregulated in CCCA , making it an interesting therapeutic target in this disease. A recent retrospective case series demonstrated that 67% (8/12) of patients with refractory CCCA had symptomatic improvement, and 50% (6/12) showed hair regrowth after 6 months of low-dose (500 mg/d) oral metformin treatment.62 In addition, metformin therapy showed antifibrotic and anti-inflammatory effects when comparing scalp biopsies before and after treatment. Results showed decreased expression of fibrosisrelated genes (matrix metalloproteinase 7, collagen type IV á 1 chain), and gene set variation analysis showing reduced Th17 (P=.04) and increased AMPK signaling (P=.02) gene set expression.62 These findings are consistent with previous studies describing the upregulation of AMPK66 and downregulation of Th176 following metformin treatment. The immunomodulatory effects of metformin could be attributed to AMPK-mediated mTOR and NF-κB downregulation,62 although more studies are needed to understand these mechanisms and further explore the use of metformin in CCCA.
Skin Cancer—Metformin also has been evaluated in the setting of skin malignancies, including melanoma, squamous cell carcinoma, and basal cell carcinoma. Preclinical data suggest that metformin decreases cell viability in tumors through interactions with pathways involved in proinflammatory and prosurvival mechanisms such as NF-κB and mTOR.69,70 Additionally, given metformin’s inhibitory effects on oxidative phosphorylation, it has been postulated that it could be used to overcome treatment resistance driven by metabolic reprogramming.71,72 Most studies related to metformin and skin malignancies are still in preclinical stages; however, a meta-analysis of RCTs and cohort studies did not find significant associations between metformin use and skin cancer risk, although data trended toward a modest reduction in skin cancer among metformin users.73 A retrospective cohort study of melanoma in patients with T2DM taking metformin (250-2000 mg/d) found that the 5-year incidence of recurrence was lower in the metformin cohort compared to nonusers (43.8% vs 58.2%, respectively)(P=.002), and overall survival rates trended upward in the higher body mass index (>30) and melanoma stages 1 and 2 groups but did not reach statistical significance.74 In addition, a whole population casecontrol study in Iceland reported that metformin use at least 2 years before first-time basal cell carcinoma diagnosis was associated with a lower risk for disease (adjusted OR, 0.71; 95% CI, 0.61-0.83) with no significant dose-dependent differences; there were no notable effects on squamous cell carcinoma risk.75 Further preclinical and clinical data are needed to elucidate metformin’s effects on skin malignancies.
GLP-1 AND DUAL GLP-1/GIP AGONISTS
Glucagonlike peptide 1 and dual GLP-1/GIP agonists are emerging classes of medications currently approved as adjunct and second-line therapies for T2DM, particularly in patients who are overweight or obese as well as in those who are at risk for hypoglycemia.3 Currently approved GLP-1 agonists for T2DM include semaglutide, dulaglutide, exenatide, liraglutide, and lixisenatide, while tirzepatide is the only approved dual GLP-1/GIP agonist. Activating GLP-1 and GIP receptors stimulates insulin secretion and decreases glucagon production by the pancreas, thereby reducing blood glucose levels. Additionally, some of these medications are approved for obesity given their effects in delayed gastric emptying and increased satiety, among other factors.
Over the past few years, multiple case reports have described the associations between GLP-1 agonist use and improvement of dermatologic conditions, particularly those associated with T2DM and obesity, including HS and psoriasis.76,77 The mechanisms through which this occurs are not fully elucidated, although basic science and clinical studies have shown that GLP-1 agonists have immunomodulatory effects by reducing proinflammatory cytokines and altering immune cell populations.77-80 The numerous ongoing clinical trials and research studies will help further elucidate their benefits in other disease settings.81
Adverse Reactions
Most GLP-1 and GLP-1/GIP agonists are administered subcutaneously, and the most commonly reported cutaneous adverse effects are injection site reactions.82 Anaphylactic reactions to these medications also have been reported, although it is unclear if these were specific to the active ingredients or to injection excipients.83,84 A review of 33 cases of cutaneous reactions to GLP-1 agonists reported 11 (33%) dermal hypersensitivity reactions occurring as early as 4 weeks and as late as 3 years after treatment initiation. It also described 10 (30%) cases of eosinophilic panniculitis that developed within 3 weeks to 5 months of GLP-1 treatment, 3 (9%) cases of bullous pemphigoid that occurred within the first 2 months, 2 (6%) morbilliform drug eruptions that occurred within 5 weeks, 2 (6%) cases of angioedema that occurred 15 minutes to 2 weeks after treatment initiation, and 7 (21%) other isolated cutaneous reactions. Extended-release exenatide had the most reported reactions followed by liraglutide and subcutaneous semaglutide.85
In a different study, semaglutide use was most commonly associated with injection site reactions followed by alopecia, especially with oral administration. Unique cases of angioedema (2 days after injection), cutaneous hypersensitivity (within 10 months on treatment), bullous pemphigoid (within 2 months on treatment), eosinophilic fasciitis (within 2 weeks on treatment), and leukocytoclastic vasculitis (unclear timing), most of which resolved after discontinuation, also were reported.86 A recent case report linked semaglutide (0.5 mg/wk) to a case of drug-induced systemic lupus erythematosus that developed within 3 months of treatment initiation and described systemic lupus erythematosus–like symptoms in a subset of patients using this medication, namely females older than 60 years, within the first month of treatment.87 Hyperhidrosis was listed as a common adverse event in exenatide clinical trials, and various cases of panniculitis with exenatide use have been reported.82,88 Alopecia, mainly attributed to accelerated telogen effluvium secondary to rapid weight loss, also has been reported, although hair loss is not officially listed as an adverse effect of GLP-1 agonists, and reports are highly variable.89 Also secondary to weight loss, facial changes including sunken eyes, development of wrinkles, sagging jowls around the neck and jaw, and a hollowed appearance, among others, are recognized as undesirable adverse effects.90 Mansour et al90 described the potential challenges and considerations to these rising concerns associated with GLP1-agonist use.
Dermatologic Implications
Hidradenitis Suppurativa—Weight loss commonly is recommended as a lifestyle modification in the management of HS. Multiple reports have described clinical improvement of HS following weight loss with other medical interventions, such as dietary measures and bariatric surgery.91-94 Thus, it has been postulated that medically supported weight loss with GLP-1 agonists can help improve HS95; however, the data on the effectiveness of GLP-1 agonists on HS are still scarce and mostly have been reported in individual patients. One case report described a patient with improvements in their recalcitrant HS and DLQI score following weight loss on liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d).76 In addition, a recent case report described improvements in HS and DLQI score following concomitant tirzepatide (initial dose of 2.5 mg/0.5 mL weekly, titrated to 7.5 mg/0.5 mL weekly) and infliximab treatment.96 The off-label use of these medications for HS is debated, and further studies regarding the benefits of GLP-1 agonists on HS still are needed.
Psoriasis—Similarly, several case reports have commented on the effects of GLP-1 agonists on psoriasis.97,98 An early study found GLP-1 receptors were expressed in psoriasis plaques but not in healthy skin and discussed that this could be due to immune infiltration in the plaques, providing a potential rationale for using anti-inflammatory GLP-1 agonists for psoriasis.99 Two prospective cohort studies observed improvements in PASI and DLQI scores in patients with psoriasis and T2DM after liraglutide treatment and noted important changes in immune cell populations.80,100 A recent RCT also found improvements in DLQI and PASI scores (P<.05) in patients with T2DM following liraglutide (1.8 mg/d) treatment, along with overall decreases in inflammatory cytokines, such as IL-23, IL-17, and TNF-α.77 However, another RCT in patients with obesity did not observe significant improvements in PASI and DLQI scores compared to placebo after 8 weeks of liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d) treatment. 99 Although these results could have been influenced by the short length of treatment compared to other studies, which observed participants for more than 10 weeks, they highlight the need for tailored studies considering the different comorbidities to identify patients who could benefit the most from these therapies.
Alopecia—Although some studies have reported increased rates of alopecia following GLP-1 agonist treatment, others have speculated about the potential role of these medications in treating hair loss through improved insulin sensitivity and scalp blood flow.86,89 For example, a case report described a patient with improvement in androgenetic alopecia within 6 months of tirzepatide monotherapy at 2.5 mg weekly for the first 3 months followed by an increased dose of 5 mg weekly.101 The authors described the role of insulin in increasing dihydrotestosterone levels, which leads to miniaturization of the dermal papilla of hair follicles and argued that improvement of insulin resistance could benefit hair loss. Further studies can help elucidate the role of these medications on alopecia.
FINAL THOUGHTS
Standard T2DM treatments including metformin and GLP-1 and GLP-1/GIP agonists exhibit metabolic, immunologic, and hormonal effects that should be explored in other disease contexts. We reviewed the current data on T2DM medications in dermatologic conditions to highlight the need for additional studies to better understand the role that these medications play across diverse patient populations. Type 2 diabetes mellitus is a common comorbidity in dermatology patients, and understanding the multifactorial effects of these medications can help optimize treatment strategies, especially in patients with coexisting dermatologic and metabolic diseases.
- Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14:88-98. doi:10.1038/nrendo.2017.151
- Ahmad E, Lim S, Lamptey R, et al. Type 2 diabetes. Lancet. 2022;400: 1803-1820. doi:10.1016/s0140-6736(22)01655-5
- Samson SL, Vellanki P, Blonde L, et al. American Association of Clinical Endocrinology Consensus Statement: comprehensive type 2 diabetes management algorithm—2023 update. Endocr Pract. 2023;29:305-340. doi:10.1016/j.eprac.2023.02.001
- LaMoia TE, Shulman GI. Cellular and molecular mechanisms of metformin action. Endocr Rev. 2021;42:77-96. doi:10.1210/endrev/bnaa023
- Petrasca A, Hambly R, Kearney N, et al. Metformin has antiinflammatory effects and induces immunometabolic reprogramming via multiple mechanisms in hidradenitis suppurativa. Br J Dermatol. 2023;189:730-740. doi:10.1093/bjd/ljad305
- Duan W, Ding Y, Yu X, et al. Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production. Am J Transl Res. 2019;11:2393-2402.
- Bharath LP, Nikolajczyk BS. The intersection of metformin and inflammation. Am J Physiol Cell Physiol. 2021;320:C873-C879. doi:10.1152 /ajpcell.00604.2020
- Sung CT, Chao T, Lee A, et al. Oral metformin for treating dermatological diseases: a systematic review. J Drugs Dermatol. 2020;19:713-720. doi:10.36849/jdd.2020.4874
- Feng J, Wang X, Ye X, et al. Mitochondria as an important target of metformin: the mechanism of action, toxic and side effects, and new therapeutic applications. Pharmacol Res. 2022;177:106114. doi:10.1016/j.phrs.2022.106114
- Klapholz L, Leitersdorf E, Weinrauch L. Leucocytoclastic vasculitis and pneumonitis induced by metformin. Br Med J (Clin Res Ed). 1986;293:483. doi:10.1136/bmj.293.6545.483
- Badr D, Kurban M, Abbas O. Metformin in dermatology: an overview. J Eur Acad Dermatol Venereol. 2013;27:1329-1335. doi:10.1111/jdv.12116
- Czarnowicki T, Ramot Y, Ingber A, et al. Metformin-induced leukocytoclastic vasculitis: a case report. Am J Clin Dermatol. 2012;13:61-63. doi:10.2165/11593230-000000000-00000
- Ben Salem C, Hmouda H, Slim R, et al. Rare case of metformininduced leukocytoclastic vasculitis. Ann Pharmacother. 2006;40:1685-1687. doi:10.1345/aph.1H155
- Abtahi-Naeini B, Momen T, Amiri R, et al. Metformin-induced generalized bullous fixed-drug eruption with a positive dechallengerechallenge test: a case report and literature review. Case Rep Dermatol Med. 2023;2023:6353919. doi:10.1155/2023/6353919
- Al Masri D, Fleifel M, Hirbli K. Fixed drug eruption secondary to four anti-diabetic medications: an unusual case of polysensitivity. Cureus. 2021;13:E18599. doi:10.7759/cureus.18599
- Ramírez-Bellver JL, Lopez J, Macias E, et al. Metformin-induced generalized fixed drug eruption with cutaneous hemophagocytosis. Am J Dermatopathol. 2017;39:471-475. doi:10.1097/dad.0000000000000800
- Steber CJ, Perkins SL, Harris KB. Metformin-induced fixed-drug eruption confirmed by multiple exposures. Am J Case Rep. 2016;17:231-234. doi:10.12659/ajcr.896424
- Voore P, Odigwe C, Mirrakhimov AE, et al. DRESS syndrome following metformin administration: a case report and review of the literature. Am J Ther. 2016;23:E1970-E1973. doi:10.1097/mjt.0000000000000292
- Kastalli S, El Aïdli S, Chaabane A, et al. Photosensitivity induced by metformin: a report of 3 cases. Article in French. Tunis Med. 2009;87:703-705.
- Karadağ AS, You Y, Danarti R, et al. Acanthosis nigricans and the metabolic syndrome. Clin Dermatol. 2018;36:48-53. doi:10.1016/j.clindermatol.2017.09.008
- Kong AS, Williams RL, Smith M, et al. Acanthosis nigricans and diabetes risk factors: prevalence in young persons seen in southwestern US primary care practices. Ann Fam Med. 2007;5:202-208. doi:10.1370/afm.678
- Stuart CA, Gilkison CR, Smith MM, et al. Acanthosis nigricans as a risk factor for non-insulin dependent diabetes mellitus. Clin Pediatr (Phila). 1998;37:73-79. doi:10.1177/000992289803700203
- Hud JA Jr, Cohen JB, Wagner JM, et al. Prevalence and significance of acanthosis nigricans in an adult obese population. Arch Dermatol. 1992;128:941-944.
- Novotny R, Davis J, Butel J, et al. Effect of the Children’s Healthy Living Program on young child overweight, obesity, and acanthosis nigricans in the US-affiliated Pacific region: a randomized clinical trial. JAMA Netw Open. 2018;1:E183896. doi:10.1001/jamanetworkopen.2018.3896
- Romo A, Benavides S. Treatment options in insulin resistance obesityrelated acanthosis nigricans. Ann Pharmacother. 2008;42:1090-1094. doi:10.1345/aph.1K446
- Treesirichod A, Chaithirayanon S, Chaikul T, et al. The randomized trials of 10% urea cream and 0.025% tretinoin cream in the treatment of acanthosis nigricans. J Dermatolog Treat. 2021;32:837-842. doi:10.108 0/09546634.2019.1708855
- Treesirichod A, Chaithirayanon S, Wongjitrat N. Comparison of the efficacy and safety of 0.1% adapalene gel and 0.025% tretinoin cream in the treatment of childhood acanthosis nigricans. Pediatr Dermatol. 2019;36:330-334. doi:10.1111/pde.13799
- Hermanns-Lê T, Hermanns JF, Piérard GE. Juvenile acanthosis nigricans and insulin resistance. Pediatr Dermatol. 2002;19:12-14. doi:10.1046 /j.1525-1470.2002.00013.x
- Walling HW, Messingham M, Myers LM, et al. Improvement of acanthosis nigricans on isotretinoin and metformin. J Drugs Dermatol. 2003;2:677-681.
- Giri D, Alsaffar H, Ramakrishnan R. Acanthosis nigricans and its response to metformin. Pediatr Dermatol. 2017;34:e281-e282. doi:10.1111/pde.13206
- Bellot-Rojas P, Posadas-Sanchez R, Caracas-Portilla N, et al. Comparison of metformin versus rosiglitazone in patients with acanthosis nigricans: a pilot study. J Drugs Dermatol. 2006;5:884-889.
- Sett A, Pradhan S, Sancheti K, et al. Effectiveness and safety of metformin versus Canthex™ in patients with acanthosis nigricans: a randomized, double-blind controlled trial. Indian J Dermatol. 2019;64:115-121. doi:10.4103/ijd.IJD_417_17
- Genazzani AD, Battaglia C, Malavasi B, et al. Metformin administration modulates and restores luteinizing hormone spontaneous episodic secretion and ovarian function in nonobese patients with polycystic ovary syndrome. Fertil Steril. 2004;81:114-119. doi:10.1016 /j.fertnstert.2003.05.020
- Kazerooni T, Dehghan-Kooshkghazi M. Effects of metformin therapy on hyperandrogenism in women with polycystic ovarian syndrome. Gynecol Endocrinol. 2003;17:51-56.
- Kolodziejczyk B, Duleba AJ, Spaczynski RZ, et al. Metformin therapy decreases hyperandrogenism and hyperinsulinemia in women with polycystic ovary syndrome. Fertil Steril. 2000;73:1149-1154. doi:10.1016 /s0015-0282(00)00501-x
- Kelly CJ, Gordon D. The effect of metformin on hirsutism in polycystic ovary syndrome. Eur J Endocrinol. 2002;147:217-221. doi:10.1530/eje.0.1470217
- Harborne L, Fleming R, Lyall H, et al. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88:4116-4123. doi:10.1210/jc.2003-030424
- Rezvanian H, Adibi N, Siavash M, et al. Increased insulin sensitivity by metformin enhances intense-pulsed-light-assisted hair removal in patients with polycystic ovary syndrome. Dermatology. 2009;218: 231-236. doi:10.1159/000187718
- Cosma M, Swiglo BA, Flynn DN, et al. Clinical review: insulin sensitizers for the treatment of hirsutism: a systematic review and metaanalyses of randomized controlled trials. J Clin Endocrinol Metab. 2008;93:1135-1142. doi:10.1210/jc.2007-2429
- Martin KA, Anderson RR, Chang RJ, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1233-1257.
- Fraison E, Kostova E, Moran LJ, et al. Metformin versus the combined oral contraceptive pill for hirsutism, acne, and menstrual pattern in polycystic ovary syndrome. Cochrane Database Syst Rev. 2020;8:CD005552. doi:10.1002/14651858.CD005552.pub3
- Hambly R, Kearney N, Hughes R, et al. Metformin treatment of hidradenitis suppurativa: effect on metabolic parameters, inflammation, cardiovascular risk biomarkers, and immune mediators. Int J Mol Sci. 2023;24:6969. doi:10.3390/ijms24086969
- Gold DA, Reeder VJ, Mahan MG, et al. The prevalence of metabolic syndrome in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2014;70:699-703. doi:10.1016/j.jaad.2013.11.014
- Miller IM, Ellervik C, Vinding GR, et al. Association of metabolic syndrome and hidradenitis suppurativa. JAMA Dermatol. 2014;150: 1273-1280. doi:10.1001/jamadermatol.2014.1165
- Monfrecola G, Balato A, Caiazzo G, et al. Mammalian target of rapamycin, insulin resistance and hidradenitis suppurativa: a possible metabolic loop. J Eur Acad Dermatol Venereol. 2016;30:1631-1633. doi:10.1111/jdv.13233
- Yoon MS. The role of mammalian target of rapamycin (mTOR) in insulin signaling. Nutrients. 2017;9:1176. doi:10.3390/nu9111176
- Abu Rached N, Gambichler T, Dietrich JW, et al. The role of hormones in hidradenitis suppurativa: a systematic review. Int J Mol Sci. 2022;23:15250. doi:10.3390/ijms232315250
- Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi:10.1111/j.1468-3083.2012.04668.x
- Tsentemeidou A, Vakirlis E, Papadimitriou I, et al. Metformin in hidradenitis suppurativa: is it worth pursuing further? Skin Appendage Disord. 2023;9:187-190. doi:10.1159/000529359
- Arun B, Loffeld A. Long-standing hidradenitis suppurativa treated effectively with metformin. Clin Exp Dermatol. 2009;34:920-921. doi:10.1111/j.1365-2230.2008.03121.x
- Jennings L, Hambly R, Hughes R, et al. Metformin use in hidradenitis suppurativa. J Dermatolog Treat. 2020;31:261-263. doi:10.1080/09546634 .2019.1592100
- Moussa C, Wadowski L, Price H, et al. Metformin as adjunctive therapy for pediatric patients with hidradenitis suppurativa. J Drugs Dermatol. 2020;19:1231-1234. doi:10.36849/jdd.2020.5447
- Cho M, Woo YR, Cho SH, et al. Metformin: a potential treatment for acne, hidradenitis suppurativa and rosacea. Acta Derm Venereol. 2023;103:adv18392. doi:10.2340/actadv.v103.18392
- Del Rosso JQ, Kircik L. The cutaneous effects of androgens and androgen-mediated sebum production and their pathophysiologic and therapeutic importance in acne vulgaris. J Dermatolog Treat. 2024;35:2298878. doi:10.1080/09546634.2023.2298878
- El-Tahlawi S, Ezzat Mohammad N, Mohamed El-Amir A, et al. Survivin and insulin-like growth factor-I: potential role in the pathogenesis of acne and post-acne scar. Scars Burn Heal. 2019;5:2059513118818031. doi:10.1177/2059513118818031
- Albalat W, Darwish H, Abd-Elaal WH, et al. The potential role of insulin-like growth factor 1 in acne vulgaris and its correlation with the clinical response before and after treatment with metformin. J Cosmet Dermatol. 2022;21:6209-6214. doi:10.1111/jocd.15210
- Nguyen S, Nguyen ML, Roberts WS, et al. The efficacy of metformin as a therapeutic agent in the treatment of acne vulgaris: a systematic review. Cureus. 2024;16:E56246. doi:10.7759/cureus.56246
- Boehncke WH, Schön MP. Psoriasis. Lancet. 2015;386:983-994. doi:10.1016 /s0140-6736(14)61909-7
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi:10.1016/j.jaad.2018.11.058
- Huang Z, Li J, Chen H, et al. The efficacy of metformin for the treatment of psoriasis: a meta-analysis study. Postepy Dermatol Alergol. 2023;40:606-610. doi:10.5114/ada.2023.130524
- Singh S, Bhansali A. Randomized placebo control study of insulin sensitizers (metformin and pioglitazone) in psoriasis patients with metabolic syndrome (topical treatment cohort). BMC Dermatol. 2016;16:12. doi:10.1186 /s12895-016-0049-y
- Bao A, Qadri A, Gadre A, et al. Low-dose metformin and profibrotic signature in central centrifugal cicatricial alopecia. JAMA Dermatol. 2024;E243062. doi:10.1001/jamadermatol.2024.3062
- Lawson CN, Bakayoko A, Callender VD. Central centrifugal cicatricial alopecia: challenges and treatments. Dermatol Clin. 2021;39:389-405. doi:10.1016/j.det.2021.03.004
- Gathers RC, Lim HW. Central centrifugal cicatricial alopecia: past, present, and future. J Am Acad Dermatol. 2009;60:660-668. doi:10.1016/j .jaad.2008.09.066
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j.jdcr.2019.12.008
- Foretz M, Guigas B, Bertrand L, et al. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20:953-966. doi:10.1016 /j.cmet.2014.09.018
- Aguh C, Dina Y, Talbot CC Jr, et al. Fibroproliferative genes are preferentially expressed in central centrifugal cicatricial alopecia. J Am Acad Dermatol. 2018;79:904-912.e1. doi:10.1016/j.jaad.2018.05.1257
- Gadre A, Dyson T, Jedrych J, et al. Proteomic profiling of central centrifugal cicatricial alopecia reveals role of humoral immune response pathway and metabolic dysregulation. JID Innov. 2024;4:100263. doi:10.1016/j.xjidi.2024.100263
- Chaudhary SC, Kurundkar D, Elmets CA, et al. Metformin, an antidiabetic agent reduces growth of cutaneous squamous cell carcinoma by targeting mTOR signaling pathway. Photochem Photobiol. 2012;88:1149-1156. doi:10.1111/j.1751-1097.2012.01165.x
- Tomic T, Botton T, Cerezo M, et al. Metformin inhibits melanoma development through autophagy and apoptosis mechanisms. Cell Death Dis. 2011;2:e199. doi:10.1038/cddis.2011.86
- Mascaraque-Checa M, Gallego-Rentero M, Nicolás-Morala J, et al. Metformin overcomes metabolic reprogramming-induced resistance of skin squamous cell carcinoma to photodynamic therapy. Mol Metab. 2022;60:101496. doi:10.1016/j.molmet.2022.101496
- Mascaraque M, Delgado-Wicke P, Nuevo-Tapioles C, et al. Metformin as an adjuvant to photodynamic therapy in resistant basal cell carcinoma cells. Cancers (Basel). 2020;12:668. doi:10.3390/cancers12030668
- Chang MS, Hartman RI, Xue J, et al. Risk of skin cancer associated with metformin use: a meta-analysis of randomized controlled trials and observational studies. Cancer Prev Res (Phila). 2021;14:77-84. doi:10.1158/1940-6207.Capr-20-0376
- Augustin RC, Huang Z, Ding F, et al. Metformin is associated with improved clinical outcomes in patients with melanoma: a retrospective, multi-institutional study. Front Oncol. 2023;13:1075823. doi:10.3389 /fonc.2023.1075823
- Adalsteinsson JA, Muzumdar S, Waldman R, et al. Metformin is associated with decreased risk of basal cell carcinoma: a whole-population casecontrol study from Iceland. J Am Acad Dermatol. 2021;85:56-61. doi:10.1016/j.jaad.2021.02.042
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Lin L, Xu X, Yu Y, et al. Glucagon-like peptide-1 receptor agonist liraglutide therapy for psoriasis patients with type 2 diabetes: a randomized-controlled trial. J Dermatolog Treat. 2022;33: 1428-1434. doi:10.1080/09546634.2020.1826392
- Karacabeyli D, Lacaille D. Glucagon-like peptide 1 receptor agonists in patients with inflammatory arthritis or psoriasis: a scoping review. J Clin Rheumatol. 2024;30:26-31. doi:10.1097/rhu.0000000000001949
- Yang J, Wang Z, Zhang X. GLP-1 receptor agonist impairs keratinocytes inflammatory signals by activating AMPK. Exp Mol Pathol. 2019;107: 124-128. doi:10.1016/j.yexmp.2019.01.014
- Buysschaert M, Baeck M, Preumont V, et al. Improvement of psoriasis during glucagon-like peptide-1 analogue therapy in type 2 diabetes is associated with decreasing dermal Υϛ T-cell number: a prospective case-series study. Br J Dermatol. 2014;171:155-161. doi:10.1111/bjd.12886
- Wilbon SS, Kolonin MG. GLP1 receptor agonists-effects beyond obesity and diabetes. Cells. 2023;13:65. doi:10.3390/cells13010065
- Filippatos TD, Panagiotopoulou TV, Elisaf MS. Adverse effects of GLP-1 receptor agonists. Rev Diabet Stud. 2014;11:202-230. doi:10.1900 /rds.2014.11.202
- He Z, Tabe AN, Rana S, et al. Tirzepatide-induced biphasic anaphylactic reaction: a case report. Cureus. 2023;15:e50112. doi:10.7759/cureus.50112
- Anthony MS, Aroda VR, Parlett LE, et al. Risk of anaphylaxis among new users of glp-1 receptor agonists: a cohort study. Diabetes Care. 2024;47:712-719. doi:10.2337/dc23-1911
- Salazar CE, Patil MK, Aihie O, et al. Rare cutaneous adverse reactions associated with GLP-1 agonists: a review of the published literature. Arch Dermatol Res. 2024;316:248. doi:10.1007/s00403-024-02969-3
- Tran MM, Mirza FN, Lee AC, et al. Dermatologic findings associated with semaglutide use: a scoping review. J Am Acad Dermatol. 2024;91:166-168. doi:10.1016/j.jaad.2024.03.021
- Castellanos V, Workneh H, Malik A, et al. Semaglutide-induced lupus erythematosus with multiorgan involvement. Cureus. 2024;16:E55324. doi:10.7759/cureus.55324
- Boccardi A, Shubrook JH. Cutaneous reactions to antidiabetic agents: a narrative review. Diabetology. 2022;3:97-107.
- Desai DD, Sikora M, Nohria A, et al. GLP-1 agonists and hair loss: a call for further investigation. Int J Dermatol. 2024;63:1128-1130. doi:10.1111 /ijd.17246
- Mansour MR, Hannawa OM, Yaldo MM, et al. The rise of “Ozempic face”: analyzing trends and treatment challenges associated with rapid facial weight loss induced by GLP-1 agonists. J Plast Reconstr Aesthet Surg. 2024;96:225-227. doi:10.1016/j.bjps.2024.07.051
- Sivanand A, Gulliver WP, Josan CK, et al. Weight loss and dietary interventions for hidradenitis suppurativa: a systematic review. J Cutan Med Surg. 2020;24:64-72. doi:10.1177/1203475419874412
- Boer J. Resolution of hidradenitis suppurativa after weight loss by dietary measures, especially on frictional locations. J Eur Acad Dermatol Venereol. 2016;30:895-896. doi:10.1111/jdv.13059
- Thomas CL, Gordon KD, Mortimer PS. Rapid resolution of hidradenitis suppurativa after bariatric surgical intervention. Clin Exp Dermatol. 2014;39:315-7; quiz 317-8. doi:10.1111/ced.12269
- Mandour MO, Al-Musawi S, Idowu E, et al. Metabolic endoscopy and a simplified low-carbohydrate-high-dietary fiber template as novel treatments for hidradenitis suppurativa—a case series. JAAD Case Rep. 2023;34:23-26. doi:10.1016/j.jdcr.2023.01.035
- Henry T, Cahn B, Haber R, et al. Therapeutic potential of GLP-1 agonists for hidradenitis suppurativa. Int J Dermatol. 2023;62:1543-1544. doi:10.1111/ijd.16892
- Chan LJ, Kaur M, Kaffenberger BH. A case of recalcitrant hidradenitis suppurativa concomitantly treated with tirzepatide. JAAD Case Rep. 2024;52:101-102. doi:10.1016/j.jdcr.2024.02.023
- Costanzo G, Curatolo S, Busà B, et al. Two birds one stone: semaglutide is highly effective against severe psoriasis in a type 2 diabetic patient. Endocrinol Diabetes Metab Case Rep. 2021;2021:21-00007. doi:10.1530 /edm-21-0007
- Buysschaert M, Tennstedt D, Preumont V. Improvement of psoriasis during exenatide treatment in a patient with diabetes. Diabetes Metab. 2012;38:86-88. doi:10.1016/j.diabet.2011.11.004
- Faurschou A, Gyldenløve M, Rohde U, et al. Lack of effect of the glucagonlike peptide-1 receptor agonist liraglutide on psoriasis in glucose-tolerant patients--a randomized placebo-controlled trial. J Eur Acad Dermatol Venereol. 2015;29:555-559. doi:10.1111/jdv.12629
- Ahern T, Tobin AM, Corrigan M, et al. Glucagon-like peptide-1 analogue therapy for psoriasis patients with obesity and type 2 diabetes: a prospective cohort study. J Eur Acad Dermatol Venereol. 2013;27:1440-1443. doi:10.1111/j.1468-3083.2012.04609.x
- Gordon ER, Musleh S, Bordone LA. Treatment of insulin resistance with tirzepatide leading to improvement of hair loss. JAAD Case Rep. 2024;50:123-125. doi:10.1016/j.jdcr.2024.06.001
Type 2 diabetes mellitus (T2DM) is a chronic disease characterized by uncontrolled hyperglycemia. Over the past few decades, its prevalence has steadily increased, now affecting approximately 10% of adults worldwide and ranking among the top 10 leading causes of death globally.1 The pathophysiology of T2DM involves persistent hyperglycemia that drives insulin resistance and a progressive decline in insulin production from the pancreas.2 Medical management of this condition aims to reduce blood glucose levels or enhance insulin production and sensitivity. Aside from lifestyle modifications, metformin is considered the first-line treatment for glycemic control according to the 2023 American Association of Clinical Endocrinology’s T2DM management algorithm.3 These updated guidelines stratify adjunct treatments by individualized glycemic targets and patient needs. For patients who are overweight or obese, glucagonlike peptide 1 (GLP-1) and dual GLP-1/ gastric inhibitory polypeptide (GIP) agonists are the preferred adjunct or second-line treatments.3
In this review, we highlight the dermatologic adverse effects and potential therapeutic benefits of metformin as well as GLP-1 and GLP-1/GIP agonists.
METFORMIN
Metformin is a biguanide agent used as a first-line treatment for T2DM because of its ability to reduce hepatic glucose production and increase peripheral tissue glucose uptake.4 In addition to its effects on glucose, metformin has been shown to have anti-inflammatory properties via inhibition of the nuclear factor κB and mammalian target of rapamycin (mTOR) pathways, leading to decreased production of cytokines associated with T helper (Th) 1 and Th17 cell responses, such as IL-17, interferon gamma (IFN-γ), and tumor necrosis factor α (TNF-α).5-7 These findings have spurred interest among clinicians in the potential use of metformin for inflammatory conditions, including dermatologic diseases such as psoriasis and hidradenitis suppurativa (HS).8
Adverse Effects
Metformin is administered orally and generally is well tolerated. The most common adverse effects include gastrointestinal symptoms such as diarrhea, nausea, vomiting, and abdominal pain.9 While cutaneous adverse effects are rare, multiple dermatologic adverse reactions to metformin have been reported,10,11 including leukocytoclastic vasculitis,11-13 fixed drug eruptions,14-17 drug rash with eosinophilia and systemic symptoms (DRESS) syndrome,18 and photosensitivity reactions.19 Leukocytoclastic vasculitis and DRESS syndrome typically develop within the first month following metformin initiation, while fixed drug eruption and photosensitivity reactions have more variable timing, occurring weeks to years after treatment initiation.12-19
Dermatologic Implications
Acanthosis Nigricans—Acanthosis nigricans (AN) is characterized by hyperpigmentation and velvety skin thickening, typically in intertriginous areas such as the back of the neck, axillae, and groin.20 It commonly is associated with insulin resistance and obesity.21-23 Treatments for AN primarily center around insulin sensitivity and weight loss,24,25 with some benefit observed from the use of keratolytic agents.26,27 Metformin may have utility in treating AN through its effects on insulin sensitivity and glycemic control. Multiple case reports have noted marked improvements in AN in patients with and without obesity with the addition of metformin to their existing treatment regimens in doses ranging from 500 mg to 1700 mg daily.28-30 However, an unblinded randomized controlled trial (RCT) comparing the efficacy of metformin (500 mg 3 times daily) with rosiglitazone (4 mg/d), another T2DM medication, on AN neck lesions in patients who were overweight and obese found no significant effects in lesion severity and only modest improvements in skin texture in both groups at 12 weeks following treatment initiation.31 Another RCT comparing metformin (500 mg twice daily) with a twice-daily capsule containing α-lipoic acid, biotin, chromium polynicotinate, and zinc sulfate, showed significant (P<.001) improvements in AN neck lesions in both groups after 12 weeks.32 According to Sung et al,8 longer duration of therapy (>6 months), higher doses (1700–2000 mg), and lower baseline weight were associated with higher efficacy of metformin for treatment of AN. Overall, the use of metformin as an adjunct treatment for AN, particularly in patients with underlying hyperglycemia, is supported in the literature, but further studies are needed to clarify dosing, duration of therapy, and patient populations that will benefit most from adding metformin to their treatment regimens.
Hirsutism—Hirsutism, which is characterized by excessive hair growth in androgen-dependent areas, can be challenging to treat. Metformin has been shown to reduce circulating insulin, luteinizing hormone, androstenedione, and testosterone, thus improving underlying hyperandrogenism, particularly in patients with polycystic ovary syndrome (PCOS).33-35 Although single studies evaluating the efficacy of metformin for treatment of hirsutism in patients with PCOS have shown potential benefits,36-38 meta-analyses showed no significant effects of metformin compared to placebo or oral contraceptives and decreased benefits compared to spironolactone and flutamide.39 Given these findings showing that metformin was no more effective than placebo or other treatments, the current Endocrine Society guidelines recommend against the use of metformin for hirsutism.39,40 There may be a role for metformin as an adjuvant therapy in certain populations (eg, patients with comorbid T2DM), although further studies stratifying risk factors such as body mass index and age are needed.41
Hidradenitis Suppurativa—Hidradenitis suppurativa is a follicular occlusive disease characterized by recurrent inflamed nodules leading to chronic dermal abscesses, fibrosis, and sinus tract formation primarily in intertriginous areas such as the axillae and groin.42 Medical management depends on disease severity but usually involves antibiotic treatment with adjunct therapies such as oral contraceptives, antiandrogenic medications (eg, spironolactone), biologic medications, and metformin.42 Preclinical and clinical data suggest that metformin can impact HS through metabolic and immunomodulatory mechanisms.5,42 Like many chronic inflammatory disorders, HS is associated with metabolic syndrome.43,44 A study evaluating insulin secretion after oral glucose tolerance testing showed increased insulin levels in patients with HS compared to controls (P=.02), with 60% (6/10) of patients with HS meeting criteria for insulin resistance. In addition, serum insulin levels in insulin-resistant patients with HS correlated with increased lesional skin mTOR gene expression at 30 (r=.80) and 60 (r=1.00) minutes, and mTOR was found to be upregulated in lesional and extralesional skin in patients with HS compared to healthy controls (P<.01).45 Insulin activates mTOR signaling, which mediates cell growth and survival, among other processes.46 Thus, metformin’s ability to increase insulin sensitivity and inhibit mTOR signaling could be beneficial in the setting of HS. Additionally, insulin and insulinlike growth factor 1 (IGF-1) increase androgen signaling, a process that has been implicated in HS.47
Metformin also may impact HS through its effects on testosterone and other hormones.48 A study evaluating peripheral blood mononuclear cells in patients with HS showed reduced IL-17, IFN-γ, TNF-α, and IL-6 levels in patients who were taking metformin (dose not reported) for longer than 6 months compared to patients who were not on metformin. Further analysis of ex vivo HS lesions cultured with metformin showed decreased IL-17, IFN-γ, TNF-α, and IL-8 expression in tissue, suggesting an antiinflammatory role of metformin in HS.5
Although there are no known RCTs assessing the efficacy of metformin in HS, existing clinical data are supportive of the use of metformin for refractory HS.49 Following a case report describing a patient with T2DM and stable HS while on metformin,50 several cohort studies have assessed the efficacy of metformin for the treatment of HS. A prospective study evaluating the efficacy of metformin monotherapy (starting dose of 500 mg/d, titrated to 500 mg 3 times daily) in patients with and without T2DM with HS refractory to other therapies found clinical improvement in 72% (18/25) of patients using the Sartorius Hidradenitis Suppurativa Score, improving from a mean (SD) score of 34.40 (12.46) to 26.76 (11.22) at 12 weeks (P=.0055,) and 22.39 (11.30) at 24 weeks (P=.0001). Additionally, 64% (16/25) of patients showed improved quality of life as evaluated by the Dermatology Life Quality Index (DLQI), which decreased from a mean (SD) score of 15.00 (4.96) to 10.08 (5.96)(P=.0017) at 12 weeks and 7.65 (7.12)(P=.000009) at 24 weeks on treatment.48 In a retrospective study of 53 patients with HS taking metformin started at 500 mg daily and increased to 500 mg twice daily after 2 weeks (when tolerated), 68% (36/53) showed some clinical response, with 19% (7/36) of those patients having achieved complete response to metformin monotherapy (defined as no active HS).51 Similarly, a retrospective study of pediatric patients with HS evaluating metformin (doses ranging from 500-2000 mg daily) as an adjunct therapy described a subset of patients with decreased frequency of HS flares with metformin.52 These studies emphasize the safety profile of metformin and support its current use as an adjunctive therapy for HS.
Acne Vulgaris—Acne vulgaris (AV) is a chronic inflammatory disorder affecting the pilosebaceous follicles.11 Similar to HS, AV has metabolic and hormonal influences that can be targeted by metformin.53 In AV, androgens lead to increased sebum production by binding to androgen receptors on sebocytes, which in turn attracts Cutibacterium acnes and promotes hyperkeratinization, inducing inflammation.54 Thus, the antiandrogenic effects of metformin may be beneficial for treatment of AV. Additionally, sebocytes express receptors for insulin and IGF-1, which can increase the size and number of sebocytes, as well as promote lipogenesis and inflammatory response, influencing sebum production.54 Serum levels for IGF-1 have been observed to be increased in patients with AV55 and reduced by metformin.56 A recent meta-analysis assessing the efficacy of metformin on AV indicated that 87% (13/15) of studies noted disease improvement on metformin, with 47% (7/15) of studies showing statistically significant (P<0.05) decreases in acne severity.57 Although most studies showed improvement, 47% (7/15) did not find significant differences between metformin and other interventions, indicating the availability of comparable treatment options. Overall, there has been a positive association between metformin use and acne improvement.57 However, it is important to note that most studies have focused on females with PCOS,57 and the main benefits of metformin in acne might be seen when managing comorbid conditions, particularly those associated with metabolic dysregulation and insulin resistance. Further studies are needed to determine the generalizability of prior results.
Psoriasis—Psoriasis is a chronic autoinflammatory disease characterized by epidermal hyperplasia with multiple cutaneous manifestations and potential for multiorgan involvement. Comorbid conditions include psoriatic arthritis, metabolic syndrome, and cardiovascular disease.58 Current treatment options depend on several factors (eg, disease severity, location of cutaneous lesions, comorbidities) and include topical, systemic, and phototherapy options, many of which target the immune system.58,59 A meta-analysis of 3 RCTs showed that metformin (500 mg/d or 1000 mg/d) was associated with significantly improved Psoriasis Area and Severity Index (PASI) 75% reductions (odds ratio [OR], 22.02; 95% CI, 2.12-228.49; P=.01) and 75% reductions in erythema, scaling, and induration (OR, 9.12; 95% CI, 2.13-39.02; P=.003) compared to placebo.60 In addition, an RCT evaluating the efficacy of metformin (1000 mg/d) or pioglitazone (30 mg/d) for 12 weeks in patients with psoriasis with metabolic syndrome found significant improvements in PASI75 (P=.001) and erythema, scaling, and induration (P=.016) scores as well as in Physician Global Assessment scores (P=.012) compared to placebo and no differences compared to pioglitazone.61 While current psoriasis management guidelines do not include metformin, its use may be worth consideration as an adjunct therapy in patients with psoriasis and comorbidities such as T2DM and metabolic syndrome.59 Metformin’s potential benefits in psoriasis may lie outside its metabolic influences and occur secondary to its immunomodulatory effects, including targeting of the Th17 axis or cytokine-specific pathways such as TNF-α, which are known to be involved in psoriasis pathogenesis.58
Central Centrifugal Cicatricial Alopecia—Central centrifugal cicatricial alopecia (CCCA) is a form of scarring alopecia characterized by chronic inflammation leading to permanent loss of hair follicles on the crown of the scalp.62 Current treatments include topical and intralesional corticosteroids, as well as oral antibiotics. In addition, therapies including the antimalarial hydroxychloroquine and immunosuppressants mycophenolate and cyclosporine are used in refractory disease.63,64 A case report described 2 patients with hair regrowth after 4 and 6 months of treatment with topical metformin 10% compounded in a proprietary transdermal vehicle.65 The authors speculated that metformin’s effects on CCCA could be attributed to its known agonistic effects on the adenosine monophosphate-activated protein kinase (AMPK) pathway with subsequent reduction in inflammation-induced fibrosis.65,66 Microarray67 and proteomic68 analysis have shown that AMPK is known to be downregulated in CCCA , making it an interesting therapeutic target in this disease. A recent retrospective case series demonstrated that 67% (8/12) of patients with refractory CCCA had symptomatic improvement, and 50% (6/12) showed hair regrowth after 6 months of low-dose (500 mg/d) oral metformin treatment.62 In addition, metformin therapy showed antifibrotic and anti-inflammatory effects when comparing scalp biopsies before and after treatment. Results showed decreased expression of fibrosisrelated genes (matrix metalloproteinase 7, collagen type IV á 1 chain), and gene set variation analysis showing reduced Th17 (P=.04) and increased AMPK signaling (P=.02) gene set expression.62 These findings are consistent with previous studies describing the upregulation of AMPK66 and downregulation of Th176 following metformin treatment. The immunomodulatory effects of metformin could be attributed to AMPK-mediated mTOR and NF-κB downregulation,62 although more studies are needed to understand these mechanisms and further explore the use of metformin in CCCA.
Skin Cancer—Metformin also has been evaluated in the setting of skin malignancies, including melanoma, squamous cell carcinoma, and basal cell carcinoma. Preclinical data suggest that metformin decreases cell viability in tumors through interactions with pathways involved in proinflammatory and prosurvival mechanisms such as NF-κB and mTOR.69,70 Additionally, given metformin’s inhibitory effects on oxidative phosphorylation, it has been postulated that it could be used to overcome treatment resistance driven by metabolic reprogramming.71,72 Most studies related to metformin and skin malignancies are still in preclinical stages; however, a meta-analysis of RCTs and cohort studies did not find significant associations between metformin use and skin cancer risk, although data trended toward a modest reduction in skin cancer among metformin users.73 A retrospective cohort study of melanoma in patients with T2DM taking metformin (250-2000 mg/d) found that the 5-year incidence of recurrence was lower in the metformin cohort compared to nonusers (43.8% vs 58.2%, respectively)(P=.002), and overall survival rates trended upward in the higher body mass index (>30) and melanoma stages 1 and 2 groups but did not reach statistical significance.74 In addition, a whole population casecontrol study in Iceland reported that metformin use at least 2 years before first-time basal cell carcinoma diagnosis was associated with a lower risk for disease (adjusted OR, 0.71; 95% CI, 0.61-0.83) with no significant dose-dependent differences; there were no notable effects on squamous cell carcinoma risk.75 Further preclinical and clinical data are needed to elucidate metformin’s effects on skin malignancies.
GLP-1 AND DUAL GLP-1/GIP AGONISTS
Glucagonlike peptide 1 and dual GLP-1/GIP agonists are emerging classes of medications currently approved as adjunct and second-line therapies for T2DM, particularly in patients who are overweight or obese as well as in those who are at risk for hypoglycemia.3 Currently approved GLP-1 agonists for T2DM include semaglutide, dulaglutide, exenatide, liraglutide, and lixisenatide, while tirzepatide is the only approved dual GLP-1/GIP agonist. Activating GLP-1 and GIP receptors stimulates insulin secretion and decreases glucagon production by the pancreas, thereby reducing blood glucose levels. Additionally, some of these medications are approved for obesity given their effects in delayed gastric emptying and increased satiety, among other factors.
Over the past few years, multiple case reports have described the associations between GLP-1 agonist use and improvement of dermatologic conditions, particularly those associated with T2DM and obesity, including HS and psoriasis.76,77 The mechanisms through which this occurs are not fully elucidated, although basic science and clinical studies have shown that GLP-1 agonists have immunomodulatory effects by reducing proinflammatory cytokines and altering immune cell populations.77-80 The numerous ongoing clinical trials and research studies will help further elucidate their benefits in other disease settings.81
Adverse Reactions
Most GLP-1 and GLP-1/GIP agonists are administered subcutaneously, and the most commonly reported cutaneous adverse effects are injection site reactions.82 Anaphylactic reactions to these medications also have been reported, although it is unclear if these were specific to the active ingredients or to injection excipients.83,84 A review of 33 cases of cutaneous reactions to GLP-1 agonists reported 11 (33%) dermal hypersensitivity reactions occurring as early as 4 weeks and as late as 3 years after treatment initiation. It also described 10 (30%) cases of eosinophilic panniculitis that developed within 3 weeks to 5 months of GLP-1 treatment, 3 (9%) cases of bullous pemphigoid that occurred within the first 2 months, 2 (6%) morbilliform drug eruptions that occurred within 5 weeks, 2 (6%) cases of angioedema that occurred 15 minutes to 2 weeks after treatment initiation, and 7 (21%) other isolated cutaneous reactions. Extended-release exenatide had the most reported reactions followed by liraglutide and subcutaneous semaglutide.85
In a different study, semaglutide use was most commonly associated with injection site reactions followed by alopecia, especially with oral administration. Unique cases of angioedema (2 days after injection), cutaneous hypersensitivity (within 10 months on treatment), bullous pemphigoid (within 2 months on treatment), eosinophilic fasciitis (within 2 weeks on treatment), and leukocytoclastic vasculitis (unclear timing), most of which resolved after discontinuation, also were reported.86 A recent case report linked semaglutide (0.5 mg/wk) to a case of drug-induced systemic lupus erythematosus that developed within 3 months of treatment initiation and described systemic lupus erythematosus–like symptoms in a subset of patients using this medication, namely females older than 60 years, within the first month of treatment.87 Hyperhidrosis was listed as a common adverse event in exenatide clinical trials, and various cases of panniculitis with exenatide use have been reported.82,88 Alopecia, mainly attributed to accelerated telogen effluvium secondary to rapid weight loss, also has been reported, although hair loss is not officially listed as an adverse effect of GLP-1 agonists, and reports are highly variable.89 Also secondary to weight loss, facial changes including sunken eyes, development of wrinkles, sagging jowls around the neck and jaw, and a hollowed appearance, among others, are recognized as undesirable adverse effects.90 Mansour et al90 described the potential challenges and considerations to these rising concerns associated with GLP1-agonist use.
Dermatologic Implications
Hidradenitis Suppurativa—Weight loss commonly is recommended as a lifestyle modification in the management of HS. Multiple reports have described clinical improvement of HS following weight loss with other medical interventions, such as dietary measures and bariatric surgery.91-94 Thus, it has been postulated that medically supported weight loss with GLP-1 agonists can help improve HS95; however, the data on the effectiveness of GLP-1 agonists on HS are still scarce and mostly have been reported in individual patients. One case report described a patient with improvements in their recalcitrant HS and DLQI score following weight loss on liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d).76 In addition, a recent case report described improvements in HS and DLQI score following concomitant tirzepatide (initial dose of 2.5 mg/0.5 mL weekly, titrated to 7.5 mg/0.5 mL weekly) and infliximab treatment.96 The off-label use of these medications for HS is debated, and further studies regarding the benefits of GLP-1 agonists on HS still are needed.
Psoriasis—Similarly, several case reports have commented on the effects of GLP-1 agonists on psoriasis.97,98 An early study found GLP-1 receptors were expressed in psoriasis plaques but not in healthy skin and discussed that this could be due to immune infiltration in the plaques, providing a potential rationale for using anti-inflammatory GLP-1 agonists for psoriasis.99 Two prospective cohort studies observed improvements in PASI and DLQI scores in patients with psoriasis and T2DM after liraglutide treatment and noted important changes in immune cell populations.80,100 A recent RCT also found improvements in DLQI and PASI scores (P<.05) in patients with T2DM following liraglutide (1.8 mg/d) treatment, along with overall decreases in inflammatory cytokines, such as IL-23, IL-17, and TNF-α.77 However, another RCT in patients with obesity did not observe significant improvements in PASI and DLQI scores compared to placebo after 8 weeks of liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d) treatment. 99 Although these results could have been influenced by the short length of treatment compared to other studies, which observed participants for more than 10 weeks, they highlight the need for tailored studies considering the different comorbidities to identify patients who could benefit the most from these therapies.
Alopecia—Although some studies have reported increased rates of alopecia following GLP-1 agonist treatment, others have speculated about the potential role of these medications in treating hair loss through improved insulin sensitivity and scalp blood flow.86,89 For example, a case report described a patient with improvement in androgenetic alopecia within 6 months of tirzepatide monotherapy at 2.5 mg weekly for the first 3 months followed by an increased dose of 5 mg weekly.101 The authors described the role of insulin in increasing dihydrotestosterone levels, which leads to miniaturization of the dermal papilla of hair follicles and argued that improvement of insulin resistance could benefit hair loss. Further studies can help elucidate the role of these medications on alopecia.
FINAL THOUGHTS
Standard T2DM treatments including metformin and GLP-1 and GLP-1/GIP agonists exhibit metabolic, immunologic, and hormonal effects that should be explored in other disease contexts. We reviewed the current data on T2DM medications in dermatologic conditions to highlight the need for additional studies to better understand the role that these medications play across diverse patient populations. Type 2 diabetes mellitus is a common comorbidity in dermatology patients, and understanding the multifactorial effects of these medications can help optimize treatment strategies, especially in patients with coexisting dermatologic and metabolic diseases.
Type 2 diabetes mellitus (T2DM) is a chronic disease characterized by uncontrolled hyperglycemia. Over the past few decades, its prevalence has steadily increased, now affecting approximately 10% of adults worldwide and ranking among the top 10 leading causes of death globally.1 The pathophysiology of T2DM involves persistent hyperglycemia that drives insulin resistance and a progressive decline in insulin production from the pancreas.2 Medical management of this condition aims to reduce blood glucose levels or enhance insulin production and sensitivity. Aside from lifestyle modifications, metformin is considered the first-line treatment for glycemic control according to the 2023 American Association of Clinical Endocrinology’s T2DM management algorithm.3 These updated guidelines stratify adjunct treatments by individualized glycemic targets and patient needs. For patients who are overweight or obese, glucagonlike peptide 1 (GLP-1) and dual GLP-1/ gastric inhibitory polypeptide (GIP) agonists are the preferred adjunct or second-line treatments.3
In this review, we highlight the dermatologic adverse effects and potential therapeutic benefits of metformin as well as GLP-1 and GLP-1/GIP agonists.
METFORMIN
Metformin is a biguanide agent used as a first-line treatment for T2DM because of its ability to reduce hepatic glucose production and increase peripheral tissue glucose uptake.4 In addition to its effects on glucose, metformin has been shown to have anti-inflammatory properties via inhibition of the nuclear factor κB and mammalian target of rapamycin (mTOR) pathways, leading to decreased production of cytokines associated with T helper (Th) 1 and Th17 cell responses, such as IL-17, interferon gamma (IFN-γ), and tumor necrosis factor α (TNF-α).5-7 These findings have spurred interest among clinicians in the potential use of metformin for inflammatory conditions, including dermatologic diseases such as psoriasis and hidradenitis suppurativa (HS).8
Adverse Effects
Metformin is administered orally and generally is well tolerated. The most common adverse effects include gastrointestinal symptoms such as diarrhea, nausea, vomiting, and abdominal pain.9 While cutaneous adverse effects are rare, multiple dermatologic adverse reactions to metformin have been reported,10,11 including leukocytoclastic vasculitis,11-13 fixed drug eruptions,14-17 drug rash with eosinophilia and systemic symptoms (DRESS) syndrome,18 and photosensitivity reactions.19 Leukocytoclastic vasculitis and DRESS syndrome typically develop within the first month following metformin initiation, while fixed drug eruption and photosensitivity reactions have more variable timing, occurring weeks to years after treatment initiation.12-19
Dermatologic Implications
Acanthosis Nigricans—Acanthosis nigricans (AN) is characterized by hyperpigmentation and velvety skin thickening, typically in intertriginous areas such as the back of the neck, axillae, and groin.20 It commonly is associated with insulin resistance and obesity.21-23 Treatments for AN primarily center around insulin sensitivity and weight loss,24,25 with some benefit observed from the use of keratolytic agents.26,27 Metformin may have utility in treating AN through its effects on insulin sensitivity and glycemic control. Multiple case reports have noted marked improvements in AN in patients with and without obesity with the addition of metformin to their existing treatment regimens in doses ranging from 500 mg to 1700 mg daily.28-30 However, an unblinded randomized controlled trial (RCT) comparing the efficacy of metformin (500 mg 3 times daily) with rosiglitazone (4 mg/d), another T2DM medication, on AN neck lesions in patients who were overweight and obese found no significant effects in lesion severity and only modest improvements in skin texture in both groups at 12 weeks following treatment initiation.31 Another RCT comparing metformin (500 mg twice daily) with a twice-daily capsule containing α-lipoic acid, biotin, chromium polynicotinate, and zinc sulfate, showed significant (P<.001) improvements in AN neck lesions in both groups after 12 weeks.32 According to Sung et al,8 longer duration of therapy (>6 months), higher doses (1700–2000 mg), and lower baseline weight were associated with higher efficacy of metformin for treatment of AN. Overall, the use of metformin as an adjunct treatment for AN, particularly in patients with underlying hyperglycemia, is supported in the literature, but further studies are needed to clarify dosing, duration of therapy, and patient populations that will benefit most from adding metformin to their treatment regimens.
Hirsutism—Hirsutism, which is characterized by excessive hair growth in androgen-dependent areas, can be challenging to treat. Metformin has been shown to reduce circulating insulin, luteinizing hormone, androstenedione, and testosterone, thus improving underlying hyperandrogenism, particularly in patients with polycystic ovary syndrome (PCOS).33-35 Although single studies evaluating the efficacy of metformin for treatment of hirsutism in patients with PCOS have shown potential benefits,36-38 meta-analyses showed no significant effects of metformin compared to placebo or oral contraceptives and decreased benefits compared to spironolactone and flutamide.39 Given these findings showing that metformin was no more effective than placebo or other treatments, the current Endocrine Society guidelines recommend against the use of metformin for hirsutism.39,40 There may be a role for metformin as an adjuvant therapy in certain populations (eg, patients with comorbid T2DM), although further studies stratifying risk factors such as body mass index and age are needed.41
Hidradenitis Suppurativa—Hidradenitis suppurativa is a follicular occlusive disease characterized by recurrent inflamed nodules leading to chronic dermal abscesses, fibrosis, and sinus tract formation primarily in intertriginous areas such as the axillae and groin.42 Medical management depends on disease severity but usually involves antibiotic treatment with adjunct therapies such as oral contraceptives, antiandrogenic medications (eg, spironolactone), biologic medications, and metformin.42 Preclinical and clinical data suggest that metformin can impact HS through metabolic and immunomodulatory mechanisms.5,42 Like many chronic inflammatory disorders, HS is associated with metabolic syndrome.43,44 A study evaluating insulin secretion after oral glucose tolerance testing showed increased insulin levels in patients with HS compared to controls (P=.02), with 60% (6/10) of patients with HS meeting criteria for insulin resistance. In addition, serum insulin levels in insulin-resistant patients with HS correlated with increased lesional skin mTOR gene expression at 30 (r=.80) and 60 (r=1.00) minutes, and mTOR was found to be upregulated in lesional and extralesional skin in patients with HS compared to healthy controls (P<.01).45 Insulin activates mTOR signaling, which mediates cell growth and survival, among other processes.46 Thus, metformin’s ability to increase insulin sensitivity and inhibit mTOR signaling could be beneficial in the setting of HS. Additionally, insulin and insulinlike growth factor 1 (IGF-1) increase androgen signaling, a process that has been implicated in HS.47
Metformin also may impact HS through its effects on testosterone and other hormones.48 A study evaluating peripheral blood mononuclear cells in patients with HS showed reduced IL-17, IFN-γ, TNF-α, and IL-6 levels in patients who were taking metformin (dose not reported) for longer than 6 months compared to patients who were not on metformin. Further analysis of ex vivo HS lesions cultured with metformin showed decreased IL-17, IFN-γ, TNF-α, and IL-8 expression in tissue, suggesting an antiinflammatory role of metformin in HS.5
Although there are no known RCTs assessing the efficacy of metformin in HS, existing clinical data are supportive of the use of metformin for refractory HS.49 Following a case report describing a patient with T2DM and stable HS while on metformin,50 several cohort studies have assessed the efficacy of metformin for the treatment of HS. A prospective study evaluating the efficacy of metformin monotherapy (starting dose of 500 mg/d, titrated to 500 mg 3 times daily) in patients with and without T2DM with HS refractory to other therapies found clinical improvement in 72% (18/25) of patients using the Sartorius Hidradenitis Suppurativa Score, improving from a mean (SD) score of 34.40 (12.46) to 26.76 (11.22) at 12 weeks (P=.0055,) and 22.39 (11.30) at 24 weeks (P=.0001). Additionally, 64% (16/25) of patients showed improved quality of life as evaluated by the Dermatology Life Quality Index (DLQI), which decreased from a mean (SD) score of 15.00 (4.96) to 10.08 (5.96)(P=.0017) at 12 weeks and 7.65 (7.12)(P=.000009) at 24 weeks on treatment.48 In a retrospective study of 53 patients with HS taking metformin started at 500 mg daily and increased to 500 mg twice daily after 2 weeks (when tolerated), 68% (36/53) showed some clinical response, with 19% (7/36) of those patients having achieved complete response to metformin monotherapy (defined as no active HS).51 Similarly, a retrospective study of pediatric patients with HS evaluating metformin (doses ranging from 500-2000 mg daily) as an adjunct therapy described a subset of patients with decreased frequency of HS flares with metformin.52 These studies emphasize the safety profile of metformin and support its current use as an adjunctive therapy for HS.
Acne Vulgaris—Acne vulgaris (AV) is a chronic inflammatory disorder affecting the pilosebaceous follicles.11 Similar to HS, AV has metabolic and hormonal influences that can be targeted by metformin.53 In AV, androgens lead to increased sebum production by binding to androgen receptors on sebocytes, which in turn attracts Cutibacterium acnes and promotes hyperkeratinization, inducing inflammation.54 Thus, the antiandrogenic effects of metformin may be beneficial for treatment of AV. Additionally, sebocytes express receptors for insulin and IGF-1, which can increase the size and number of sebocytes, as well as promote lipogenesis and inflammatory response, influencing sebum production.54 Serum levels for IGF-1 have been observed to be increased in patients with AV55 and reduced by metformin.56 A recent meta-analysis assessing the efficacy of metformin on AV indicated that 87% (13/15) of studies noted disease improvement on metformin, with 47% (7/15) of studies showing statistically significant (P<0.05) decreases in acne severity.57 Although most studies showed improvement, 47% (7/15) did not find significant differences between metformin and other interventions, indicating the availability of comparable treatment options. Overall, there has been a positive association between metformin use and acne improvement.57 However, it is important to note that most studies have focused on females with PCOS,57 and the main benefits of metformin in acne might be seen when managing comorbid conditions, particularly those associated with metabolic dysregulation and insulin resistance. Further studies are needed to determine the generalizability of prior results.
Psoriasis—Psoriasis is a chronic autoinflammatory disease characterized by epidermal hyperplasia with multiple cutaneous manifestations and potential for multiorgan involvement. Comorbid conditions include psoriatic arthritis, metabolic syndrome, and cardiovascular disease.58 Current treatment options depend on several factors (eg, disease severity, location of cutaneous lesions, comorbidities) and include topical, systemic, and phototherapy options, many of which target the immune system.58,59 A meta-analysis of 3 RCTs showed that metformin (500 mg/d or 1000 mg/d) was associated with significantly improved Psoriasis Area and Severity Index (PASI) 75% reductions (odds ratio [OR], 22.02; 95% CI, 2.12-228.49; P=.01) and 75% reductions in erythema, scaling, and induration (OR, 9.12; 95% CI, 2.13-39.02; P=.003) compared to placebo.60 In addition, an RCT evaluating the efficacy of metformin (1000 mg/d) or pioglitazone (30 mg/d) for 12 weeks in patients with psoriasis with metabolic syndrome found significant improvements in PASI75 (P=.001) and erythema, scaling, and induration (P=.016) scores as well as in Physician Global Assessment scores (P=.012) compared to placebo and no differences compared to pioglitazone.61 While current psoriasis management guidelines do not include metformin, its use may be worth consideration as an adjunct therapy in patients with psoriasis and comorbidities such as T2DM and metabolic syndrome.59 Metformin’s potential benefits in psoriasis may lie outside its metabolic influences and occur secondary to its immunomodulatory effects, including targeting of the Th17 axis or cytokine-specific pathways such as TNF-α, which are known to be involved in psoriasis pathogenesis.58
Central Centrifugal Cicatricial Alopecia—Central centrifugal cicatricial alopecia (CCCA) is a form of scarring alopecia characterized by chronic inflammation leading to permanent loss of hair follicles on the crown of the scalp.62 Current treatments include topical and intralesional corticosteroids, as well as oral antibiotics. In addition, therapies including the antimalarial hydroxychloroquine and immunosuppressants mycophenolate and cyclosporine are used in refractory disease.63,64 A case report described 2 patients with hair regrowth after 4 and 6 months of treatment with topical metformin 10% compounded in a proprietary transdermal vehicle.65 The authors speculated that metformin’s effects on CCCA could be attributed to its known agonistic effects on the adenosine monophosphate-activated protein kinase (AMPK) pathway with subsequent reduction in inflammation-induced fibrosis.65,66 Microarray67 and proteomic68 analysis have shown that AMPK is known to be downregulated in CCCA , making it an interesting therapeutic target in this disease. A recent retrospective case series demonstrated that 67% (8/12) of patients with refractory CCCA had symptomatic improvement, and 50% (6/12) showed hair regrowth after 6 months of low-dose (500 mg/d) oral metformin treatment.62 In addition, metformin therapy showed antifibrotic and anti-inflammatory effects when comparing scalp biopsies before and after treatment. Results showed decreased expression of fibrosisrelated genes (matrix metalloproteinase 7, collagen type IV á 1 chain), and gene set variation analysis showing reduced Th17 (P=.04) and increased AMPK signaling (P=.02) gene set expression.62 These findings are consistent with previous studies describing the upregulation of AMPK66 and downregulation of Th176 following metformin treatment. The immunomodulatory effects of metformin could be attributed to AMPK-mediated mTOR and NF-κB downregulation,62 although more studies are needed to understand these mechanisms and further explore the use of metformin in CCCA.
Skin Cancer—Metformin also has been evaluated in the setting of skin malignancies, including melanoma, squamous cell carcinoma, and basal cell carcinoma. Preclinical data suggest that metformin decreases cell viability in tumors through interactions with pathways involved in proinflammatory and prosurvival mechanisms such as NF-κB and mTOR.69,70 Additionally, given metformin’s inhibitory effects on oxidative phosphorylation, it has been postulated that it could be used to overcome treatment resistance driven by metabolic reprogramming.71,72 Most studies related to metformin and skin malignancies are still in preclinical stages; however, a meta-analysis of RCTs and cohort studies did not find significant associations between metformin use and skin cancer risk, although data trended toward a modest reduction in skin cancer among metformin users.73 A retrospective cohort study of melanoma in patients with T2DM taking metformin (250-2000 mg/d) found that the 5-year incidence of recurrence was lower in the metformin cohort compared to nonusers (43.8% vs 58.2%, respectively)(P=.002), and overall survival rates trended upward in the higher body mass index (>30) and melanoma stages 1 and 2 groups but did not reach statistical significance.74 In addition, a whole population casecontrol study in Iceland reported that metformin use at least 2 years before first-time basal cell carcinoma diagnosis was associated with a lower risk for disease (adjusted OR, 0.71; 95% CI, 0.61-0.83) with no significant dose-dependent differences; there were no notable effects on squamous cell carcinoma risk.75 Further preclinical and clinical data are needed to elucidate metformin’s effects on skin malignancies.
GLP-1 AND DUAL GLP-1/GIP AGONISTS
Glucagonlike peptide 1 and dual GLP-1/GIP agonists are emerging classes of medications currently approved as adjunct and second-line therapies for T2DM, particularly in patients who are overweight or obese as well as in those who are at risk for hypoglycemia.3 Currently approved GLP-1 agonists for T2DM include semaglutide, dulaglutide, exenatide, liraglutide, and lixisenatide, while tirzepatide is the only approved dual GLP-1/GIP agonist. Activating GLP-1 and GIP receptors stimulates insulin secretion and decreases glucagon production by the pancreas, thereby reducing blood glucose levels. Additionally, some of these medications are approved for obesity given their effects in delayed gastric emptying and increased satiety, among other factors.
Over the past few years, multiple case reports have described the associations between GLP-1 agonist use and improvement of dermatologic conditions, particularly those associated with T2DM and obesity, including HS and psoriasis.76,77 The mechanisms through which this occurs are not fully elucidated, although basic science and clinical studies have shown that GLP-1 agonists have immunomodulatory effects by reducing proinflammatory cytokines and altering immune cell populations.77-80 The numerous ongoing clinical trials and research studies will help further elucidate their benefits in other disease settings.81
Adverse Reactions
Most GLP-1 and GLP-1/GIP agonists are administered subcutaneously, and the most commonly reported cutaneous adverse effects are injection site reactions.82 Anaphylactic reactions to these medications also have been reported, although it is unclear if these were specific to the active ingredients or to injection excipients.83,84 A review of 33 cases of cutaneous reactions to GLP-1 agonists reported 11 (33%) dermal hypersensitivity reactions occurring as early as 4 weeks and as late as 3 years after treatment initiation. It also described 10 (30%) cases of eosinophilic panniculitis that developed within 3 weeks to 5 months of GLP-1 treatment, 3 (9%) cases of bullous pemphigoid that occurred within the first 2 months, 2 (6%) morbilliform drug eruptions that occurred within 5 weeks, 2 (6%) cases of angioedema that occurred 15 minutes to 2 weeks after treatment initiation, and 7 (21%) other isolated cutaneous reactions. Extended-release exenatide had the most reported reactions followed by liraglutide and subcutaneous semaglutide.85
In a different study, semaglutide use was most commonly associated with injection site reactions followed by alopecia, especially with oral administration. Unique cases of angioedema (2 days after injection), cutaneous hypersensitivity (within 10 months on treatment), bullous pemphigoid (within 2 months on treatment), eosinophilic fasciitis (within 2 weeks on treatment), and leukocytoclastic vasculitis (unclear timing), most of which resolved after discontinuation, also were reported.86 A recent case report linked semaglutide (0.5 mg/wk) to a case of drug-induced systemic lupus erythematosus that developed within 3 months of treatment initiation and described systemic lupus erythematosus–like symptoms in a subset of patients using this medication, namely females older than 60 years, within the first month of treatment.87 Hyperhidrosis was listed as a common adverse event in exenatide clinical trials, and various cases of panniculitis with exenatide use have been reported.82,88 Alopecia, mainly attributed to accelerated telogen effluvium secondary to rapid weight loss, also has been reported, although hair loss is not officially listed as an adverse effect of GLP-1 agonists, and reports are highly variable.89 Also secondary to weight loss, facial changes including sunken eyes, development of wrinkles, sagging jowls around the neck and jaw, and a hollowed appearance, among others, are recognized as undesirable adverse effects.90 Mansour et al90 described the potential challenges and considerations to these rising concerns associated with GLP1-agonist use.
Dermatologic Implications
Hidradenitis Suppurativa—Weight loss commonly is recommended as a lifestyle modification in the management of HS. Multiple reports have described clinical improvement of HS following weight loss with other medical interventions, such as dietary measures and bariatric surgery.91-94 Thus, it has been postulated that medically supported weight loss with GLP-1 agonists can help improve HS95; however, the data on the effectiveness of GLP-1 agonists on HS are still scarce and mostly have been reported in individual patients. One case report described a patient with improvements in their recalcitrant HS and DLQI score following weight loss on liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d).76 In addition, a recent case report described improvements in HS and DLQI score following concomitant tirzepatide (initial dose of 2.5 mg/0.5 mL weekly, titrated to 7.5 mg/0.5 mL weekly) and infliximab treatment.96 The off-label use of these medications for HS is debated, and further studies regarding the benefits of GLP-1 agonists on HS still are needed.
Psoriasis—Similarly, several case reports have commented on the effects of GLP-1 agonists on psoriasis.97,98 An early study found GLP-1 receptors were expressed in psoriasis plaques but not in healthy skin and discussed that this could be due to immune infiltration in the plaques, providing a potential rationale for using anti-inflammatory GLP-1 agonists for psoriasis.99 Two prospective cohort studies observed improvements in PASI and DLQI scores in patients with psoriasis and T2DM after liraglutide treatment and noted important changes in immune cell populations.80,100 A recent RCT also found improvements in DLQI and PASI scores (P<.05) in patients with T2DM following liraglutide (1.8 mg/d) treatment, along with overall decreases in inflammatory cytokines, such as IL-23, IL-17, and TNF-α.77 However, another RCT in patients with obesity did not observe significant improvements in PASI and DLQI scores compared to placebo after 8 weeks of liraglutide (initial dose of 0.6 mg/d, titrated to 1.8 mg/d) treatment. 99 Although these results could have been influenced by the short length of treatment compared to other studies, which observed participants for more than 10 weeks, they highlight the need for tailored studies considering the different comorbidities to identify patients who could benefit the most from these therapies.
Alopecia—Although some studies have reported increased rates of alopecia following GLP-1 agonist treatment, others have speculated about the potential role of these medications in treating hair loss through improved insulin sensitivity and scalp blood flow.86,89 For example, a case report described a patient with improvement in androgenetic alopecia within 6 months of tirzepatide monotherapy at 2.5 mg weekly for the first 3 months followed by an increased dose of 5 mg weekly.101 The authors described the role of insulin in increasing dihydrotestosterone levels, which leads to miniaturization of the dermal papilla of hair follicles and argued that improvement of insulin resistance could benefit hair loss. Further studies can help elucidate the role of these medications on alopecia.
FINAL THOUGHTS
Standard T2DM treatments including metformin and GLP-1 and GLP-1/GIP agonists exhibit metabolic, immunologic, and hormonal effects that should be explored in other disease contexts. We reviewed the current data on T2DM medications in dermatologic conditions to highlight the need for additional studies to better understand the role that these medications play across diverse patient populations. Type 2 diabetes mellitus is a common comorbidity in dermatology patients, and understanding the multifactorial effects of these medications can help optimize treatment strategies, especially in patients with coexisting dermatologic and metabolic diseases.
- Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14:88-98. doi:10.1038/nrendo.2017.151
- Ahmad E, Lim S, Lamptey R, et al. Type 2 diabetes. Lancet. 2022;400: 1803-1820. doi:10.1016/s0140-6736(22)01655-5
- Samson SL, Vellanki P, Blonde L, et al. American Association of Clinical Endocrinology Consensus Statement: comprehensive type 2 diabetes management algorithm—2023 update. Endocr Pract. 2023;29:305-340. doi:10.1016/j.eprac.2023.02.001
- LaMoia TE, Shulman GI. Cellular and molecular mechanisms of metformin action. Endocr Rev. 2021;42:77-96. doi:10.1210/endrev/bnaa023
- Petrasca A, Hambly R, Kearney N, et al. Metformin has antiinflammatory effects and induces immunometabolic reprogramming via multiple mechanisms in hidradenitis suppurativa. Br J Dermatol. 2023;189:730-740. doi:10.1093/bjd/ljad305
- Duan W, Ding Y, Yu X, et al. Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production. Am J Transl Res. 2019;11:2393-2402.
- Bharath LP, Nikolajczyk BS. The intersection of metformin and inflammation. Am J Physiol Cell Physiol. 2021;320:C873-C879. doi:10.1152 /ajpcell.00604.2020
- Sung CT, Chao T, Lee A, et al. Oral metformin for treating dermatological diseases: a systematic review. J Drugs Dermatol. 2020;19:713-720. doi:10.36849/jdd.2020.4874
- Feng J, Wang X, Ye X, et al. Mitochondria as an important target of metformin: the mechanism of action, toxic and side effects, and new therapeutic applications. Pharmacol Res. 2022;177:106114. doi:10.1016/j.phrs.2022.106114
- Klapholz L, Leitersdorf E, Weinrauch L. Leucocytoclastic vasculitis and pneumonitis induced by metformin. Br Med J (Clin Res Ed). 1986;293:483. doi:10.1136/bmj.293.6545.483
- Badr D, Kurban M, Abbas O. Metformin in dermatology: an overview. J Eur Acad Dermatol Venereol. 2013;27:1329-1335. doi:10.1111/jdv.12116
- Czarnowicki T, Ramot Y, Ingber A, et al. Metformin-induced leukocytoclastic vasculitis: a case report. Am J Clin Dermatol. 2012;13:61-63. doi:10.2165/11593230-000000000-00000
- Ben Salem C, Hmouda H, Slim R, et al. Rare case of metformininduced leukocytoclastic vasculitis. Ann Pharmacother. 2006;40:1685-1687. doi:10.1345/aph.1H155
- Abtahi-Naeini B, Momen T, Amiri R, et al. Metformin-induced generalized bullous fixed-drug eruption with a positive dechallengerechallenge test: a case report and literature review. Case Rep Dermatol Med. 2023;2023:6353919. doi:10.1155/2023/6353919
- Al Masri D, Fleifel M, Hirbli K. Fixed drug eruption secondary to four anti-diabetic medications: an unusual case of polysensitivity. Cureus. 2021;13:E18599. doi:10.7759/cureus.18599
- Ramírez-Bellver JL, Lopez J, Macias E, et al. Metformin-induced generalized fixed drug eruption with cutaneous hemophagocytosis. Am J Dermatopathol. 2017;39:471-475. doi:10.1097/dad.0000000000000800
- Steber CJ, Perkins SL, Harris KB. Metformin-induced fixed-drug eruption confirmed by multiple exposures. Am J Case Rep. 2016;17:231-234. doi:10.12659/ajcr.896424
- Voore P, Odigwe C, Mirrakhimov AE, et al. DRESS syndrome following metformin administration: a case report and review of the literature. Am J Ther. 2016;23:E1970-E1973. doi:10.1097/mjt.0000000000000292
- Kastalli S, El Aïdli S, Chaabane A, et al. Photosensitivity induced by metformin: a report of 3 cases. Article in French. Tunis Med. 2009;87:703-705.
- Karadağ AS, You Y, Danarti R, et al. Acanthosis nigricans and the metabolic syndrome. Clin Dermatol. 2018;36:48-53. doi:10.1016/j.clindermatol.2017.09.008
- Kong AS, Williams RL, Smith M, et al. Acanthosis nigricans and diabetes risk factors: prevalence in young persons seen in southwestern US primary care practices. Ann Fam Med. 2007;5:202-208. doi:10.1370/afm.678
- Stuart CA, Gilkison CR, Smith MM, et al. Acanthosis nigricans as a risk factor for non-insulin dependent diabetes mellitus. Clin Pediatr (Phila). 1998;37:73-79. doi:10.1177/000992289803700203
- Hud JA Jr, Cohen JB, Wagner JM, et al. Prevalence and significance of acanthosis nigricans in an adult obese population. Arch Dermatol. 1992;128:941-944.
- Novotny R, Davis J, Butel J, et al. Effect of the Children’s Healthy Living Program on young child overweight, obesity, and acanthosis nigricans in the US-affiliated Pacific region: a randomized clinical trial. JAMA Netw Open. 2018;1:E183896. doi:10.1001/jamanetworkopen.2018.3896
- Romo A, Benavides S. Treatment options in insulin resistance obesityrelated acanthosis nigricans. Ann Pharmacother. 2008;42:1090-1094. doi:10.1345/aph.1K446
- Treesirichod A, Chaithirayanon S, Chaikul T, et al. The randomized trials of 10% urea cream and 0.025% tretinoin cream in the treatment of acanthosis nigricans. J Dermatolog Treat. 2021;32:837-842. doi:10.108 0/09546634.2019.1708855
- Treesirichod A, Chaithirayanon S, Wongjitrat N. Comparison of the efficacy and safety of 0.1% adapalene gel and 0.025% tretinoin cream in the treatment of childhood acanthosis nigricans. Pediatr Dermatol. 2019;36:330-334. doi:10.1111/pde.13799
- Hermanns-Lê T, Hermanns JF, Piérard GE. Juvenile acanthosis nigricans and insulin resistance. Pediatr Dermatol. 2002;19:12-14. doi:10.1046 /j.1525-1470.2002.00013.x
- Walling HW, Messingham M, Myers LM, et al. Improvement of acanthosis nigricans on isotretinoin and metformin. J Drugs Dermatol. 2003;2:677-681.
- Giri D, Alsaffar H, Ramakrishnan R. Acanthosis nigricans and its response to metformin. Pediatr Dermatol. 2017;34:e281-e282. doi:10.1111/pde.13206
- Bellot-Rojas P, Posadas-Sanchez R, Caracas-Portilla N, et al. Comparison of metformin versus rosiglitazone in patients with acanthosis nigricans: a pilot study. J Drugs Dermatol. 2006;5:884-889.
- Sett A, Pradhan S, Sancheti K, et al. Effectiveness and safety of metformin versus Canthex™ in patients with acanthosis nigricans: a randomized, double-blind controlled trial. Indian J Dermatol. 2019;64:115-121. doi:10.4103/ijd.IJD_417_17
- Genazzani AD, Battaglia C, Malavasi B, et al. Metformin administration modulates and restores luteinizing hormone spontaneous episodic secretion and ovarian function in nonobese patients with polycystic ovary syndrome. Fertil Steril. 2004;81:114-119. doi:10.1016 /j.fertnstert.2003.05.020
- Kazerooni T, Dehghan-Kooshkghazi M. Effects of metformin therapy on hyperandrogenism in women with polycystic ovarian syndrome. Gynecol Endocrinol. 2003;17:51-56.
- Kolodziejczyk B, Duleba AJ, Spaczynski RZ, et al. Metformin therapy decreases hyperandrogenism and hyperinsulinemia in women with polycystic ovary syndrome. Fertil Steril. 2000;73:1149-1154. doi:10.1016 /s0015-0282(00)00501-x
- Kelly CJ, Gordon D. The effect of metformin on hirsutism in polycystic ovary syndrome. Eur J Endocrinol. 2002;147:217-221. doi:10.1530/eje.0.1470217
- Harborne L, Fleming R, Lyall H, et al. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88:4116-4123. doi:10.1210/jc.2003-030424
- Rezvanian H, Adibi N, Siavash M, et al. Increased insulin sensitivity by metformin enhances intense-pulsed-light-assisted hair removal in patients with polycystic ovary syndrome. Dermatology. 2009;218: 231-236. doi:10.1159/000187718
- Cosma M, Swiglo BA, Flynn DN, et al. Clinical review: insulin sensitizers for the treatment of hirsutism: a systematic review and metaanalyses of randomized controlled trials. J Clin Endocrinol Metab. 2008;93:1135-1142. doi:10.1210/jc.2007-2429
- Martin KA, Anderson RR, Chang RJ, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1233-1257.
- Fraison E, Kostova E, Moran LJ, et al. Metformin versus the combined oral contraceptive pill for hirsutism, acne, and menstrual pattern in polycystic ovary syndrome. Cochrane Database Syst Rev. 2020;8:CD005552. doi:10.1002/14651858.CD005552.pub3
- Hambly R, Kearney N, Hughes R, et al. Metformin treatment of hidradenitis suppurativa: effect on metabolic parameters, inflammation, cardiovascular risk biomarkers, and immune mediators. Int J Mol Sci. 2023;24:6969. doi:10.3390/ijms24086969
- Gold DA, Reeder VJ, Mahan MG, et al. The prevalence of metabolic syndrome in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2014;70:699-703. doi:10.1016/j.jaad.2013.11.014
- Miller IM, Ellervik C, Vinding GR, et al. Association of metabolic syndrome and hidradenitis suppurativa. JAMA Dermatol. 2014;150: 1273-1280. doi:10.1001/jamadermatol.2014.1165
- Monfrecola G, Balato A, Caiazzo G, et al. Mammalian target of rapamycin, insulin resistance and hidradenitis suppurativa: a possible metabolic loop. J Eur Acad Dermatol Venereol. 2016;30:1631-1633. doi:10.1111/jdv.13233
- Yoon MS. The role of mammalian target of rapamycin (mTOR) in insulin signaling. Nutrients. 2017;9:1176. doi:10.3390/nu9111176
- Abu Rached N, Gambichler T, Dietrich JW, et al. The role of hormones in hidradenitis suppurativa: a systematic review. Int J Mol Sci. 2022;23:15250. doi:10.3390/ijms232315250
- Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi:10.1111/j.1468-3083.2012.04668.x
- Tsentemeidou A, Vakirlis E, Papadimitriou I, et al. Metformin in hidradenitis suppurativa: is it worth pursuing further? Skin Appendage Disord. 2023;9:187-190. doi:10.1159/000529359
- Arun B, Loffeld A. Long-standing hidradenitis suppurativa treated effectively with metformin. Clin Exp Dermatol. 2009;34:920-921. doi:10.1111/j.1365-2230.2008.03121.x
- Jennings L, Hambly R, Hughes R, et al. Metformin use in hidradenitis suppurativa. J Dermatolog Treat. 2020;31:261-263. doi:10.1080/09546634 .2019.1592100
- Moussa C, Wadowski L, Price H, et al. Metformin as adjunctive therapy for pediatric patients with hidradenitis suppurativa. J Drugs Dermatol. 2020;19:1231-1234. doi:10.36849/jdd.2020.5447
- Cho M, Woo YR, Cho SH, et al. Metformin: a potential treatment for acne, hidradenitis suppurativa and rosacea. Acta Derm Venereol. 2023;103:adv18392. doi:10.2340/actadv.v103.18392
- Del Rosso JQ, Kircik L. The cutaneous effects of androgens and androgen-mediated sebum production and their pathophysiologic and therapeutic importance in acne vulgaris. J Dermatolog Treat. 2024;35:2298878. doi:10.1080/09546634.2023.2298878
- El-Tahlawi S, Ezzat Mohammad N, Mohamed El-Amir A, et al. Survivin and insulin-like growth factor-I: potential role in the pathogenesis of acne and post-acne scar. Scars Burn Heal. 2019;5:2059513118818031. doi:10.1177/2059513118818031
- Albalat W, Darwish H, Abd-Elaal WH, et al. The potential role of insulin-like growth factor 1 in acne vulgaris and its correlation with the clinical response before and after treatment with metformin. J Cosmet Dermatol. 2022;21:6209-6214. doi:10.1111/jocd.15210
- Nguyen S, Nguyen ML, Roberts WS, et al. The efficacy of metformin as a therapeutic agent in the treatment of acne vulgaris: a systematic review. Cureus. 2024;16:E56246. doi:10.7759/cureus.56246
- Boehncke WH, Schön MP. Psoriasis. Lancet. 2015;386:983-994. doi:10.1016 /s0140-6736(14)61909-7
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi:10.1016/j.jaad.2018.11.058
- Huang Z, Li J, Chen H, et al. The efficacy of metformin for the treatment of psoriasis: a meta-analysis study. Postepy Dermatol Alergol. 2023;40:606-610. doi:10.5114/ada.2023.130524
- Singh S, Bhansali A. Randomized placebo control study of insulin sensitizers (metformin and pioglitazone) in psoriasis patients with metabolic syndrome (topical treatment cohort). BMC Dermatol. 2016;16:12. doi:10.1186 /s12895-016-0049-y
- Bao A, Qadri A, Gadre A, et al. Low-dose metformin and profibrotic signature in central centrifugal cicatricial alopecia. JAMA Dermatol. 2024;E243062. doi:10.1001/jamadermatol.2024.3062
- Lawson CN, Bakayoko A, Callender VD. Central centrifugal cicatricial alopecia: challenges and treatments. Dermatol Clin. 2021;39:389-405. doi:10.1016/j.det.2021.03.004
- Gathers RC, Lim HW. Central centrifugal cicatricial alopecia: past, present, and future. J Am Acad Dermatol. 2009;60:660-668. doi:10.1016/j .jaad.2008.09.066
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j.jdcr.2019.12.008
- Foretz M, Guigas B, Bertrand L, et al. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20:953-966. doi:10.1016 /j.cmet.2014.09.018
- Aguh C, Dina Y, Talbot CC Jr, et al. Fibroproliferative genes are preferentially expressed in central centrifugal cicatricial alopecia. J Am Acad Dermatol. 2018;79:904-912.e1. doi:10.1016/j.jaad.2018.05.1257
- Gadre A, Dyson T, Jedrych J, et al. Proteomic profiling of central centrifugal cicatricial alopecia reveals role of humoral immune response pathway and metabolic dysregulation. JID Innov. 2024;4:100263. doi:10.1016/j.xjidi.2024.100263
- Chaudhary SC, Kurundkar D, Elmets CA, et al. Metformin, an antidiabetic agent reduces growth of cutaneous squamous cell carcinoma by targeting mTOR signaling pathway. Photochem Photobiol. 2012;88:1149-1156. doi:10.1111/j.1751-1097.2012.01165.x
- Tomic T, Botton T, Cerezo M, et al. Metformin inhibits melanoma development through autophagy and apoptosis mechanisms. Cell Death Dis. 2011;2:e199. doi:10.1038/cddis.2011.86
- Mascaraque-Checa M, Gallego-Rentero M, Nicolás-Morala J, et al. Metformin overcomes metabolic reprogramming-induced resistance of skin squamous cell carcinoma to photodynamic therapy. Mol Metab. 2022;60:101496. doi:10.1016/j.molmet.2022.101496
- Mascaraque M, Delgado-Wicke P, Nuevo-Tapioles C, et al. Metformin as an adjuvant to photodynamic therapy in resistant basal cell carcinoma cells. Cancers (Basel). 2020;12:668. doi:10.3390/cancers12030668
- Chang MS, Hartman RI, Xue J, et al. Risk of skin cancer associated with metformin use: a meta-analysis of randomized controlled trials and observational studies. Cancer Prev Res (Phila). 2021;14:77-84. doi:10.1158/1940-6207.Capr-20-0376
- Augustin RC, Huang Z, Ding F, et al. Metformin is associated with improved clinical outcomes in patients with melanoma: a retrospective, multi-institutional study. Front Oncol. 2023;13:1075823. doi:10.3389 /fonc.2023.1075823
- Adalsteinsson JA, Muzumdar S, Waldman R, et al. Metformin is associated with decreased risk of basal cell carcinoma: a whole-population casecontrol study from Iceland. J Am Acad Dermatol. 2021;85:56-61. doi:10.1016/j.jaad.2021.02.042
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Lin L, Xu X, Yu Y, et al. Glucagon-like peptide-1 receptor agonist liraglutide therapy for psoriasis patients with type 2 diabetes: a randomized-controlled trial. J Dermatolog Treat. 2022;33: 1428-1434. doi:10.1080/09546634.2020.1826392
- Karacabeyli D, Lacaille D. Glucagon-like peptide 1 receptor agonists in patients with inflammatory arthritis or psoriasis: a scoping review. J Clin Rheumatol. 2024;30:26-31. doi:10.1097/rhu.0000000000001949
- Yang J, Wang Z, Zhang X. GLP-1 receptor agonist impairs keratinocytes inflammatory signals by activating AMPK. Exp Mol Pathol. 2019;107: 124-128. doi:10.1016/j.yexmp.2019.01.014
- Buysschaert M, Baeck M, Preumont V, et al. Improvement of psoriasis during glucagon-like peptide-1 analogue therapy in type 2 diabetes is associated with decreasing dermal Υϛ T-cell number: a prospective case-series study. Br J Dermatol. 2014;171:155-161. doi:10.1111/bjd.12886
- Wilbon SS, Kolonin MG. GLP1 receptor agonists-effects beyond obesity and diabetes. Cells. 2023;13:65. doi:10.3390/cells13010065
- Filippatos TD, Panagiotopoulou TV, Elisaf MS. Adverse effects of GLP-1 receptor agonists. Rev Diabet Stud. 2014;11:202-230. doi:10.1900 /rds.2014.11.202
- He Z, Tabe AN, Rana S, et al. Tirzepatide-induced biphasic anaphylactic reaction: a case report. Cureus. 2023;15:e50112. doi:10.7759/cureus.50112
- Anthony MS, Aroda VR, Parlett LE, et al. Risk of anaphylaxis among new users of glp-1 receptor agonists: a cohort study. Diabetes Care. 2024;47:712-719. doi:10.2337/dc23-1911
- Salazar CE, Patil MK, Aihie O, et al. Rare cutaneous adverse reactions associated with GLP-1 agonists: a review of the published literature. Arch Dermatol Res. 2024;316:248. doi:10.1007/s00403-024-02969-3
- Tran MM, Mirza FN, Lee AC, et al. Dermatologic findings associated with semaglutide use: a scoping review. J Am Acad Dermatol. 2024;91:166-168. doi:10.1016/j.jaad.2024.03.021
- Castellanos V, Workneh H, Malik A, et al. Semaglutide-induced lupus erythematosus with multiorgan involvement. Cureus. 2024;16:E55324. doi:10.7759/cureus.55324
- Boccardi A, Shubrook JH. Cutaneous reactions to antidiabetic agents: a narrative review. Diabetology. 2022;3:97-107.
- Desai DD, Sikora M, Nohria A, et al. GLP-1 agonists and hair loss: a call for further investigation. Int J Dermatol. 2024;63:1128-1130. doi:10.1111 /ijd.17246
- Mansour MR, Hannawa OM, Yaldo MM, et al. The rise of “Ozempic face”: analyzing trends and treatment challenges associated with rapid facial weight loss induced by GLP-1 agonists. J Plast Reconstr Aesthet Surg. 2024;96:225-227. doi:10.1016/j.bjps.2024.07.051
- Sivanand A, Gulliver WP, Josan CK, et al. Weight loss and dietary interventions for hidradenitis suppurativa: a systematic review. J Cutan Med Surg. 2020;24:64-72. doi:10.1177/1203475419874412
- Boer J. Resolution of hidradenitis suppurativa after weight loss by dietary measures, especially on frictional locations. J Eur Acad Dermatol Venereol. 2016;30:895-896. doi:10.1111/jdv.13059
- Thomas CL, Gordon KD, Mortimer PS. Rapid resolution of hidradenitis suppurativa after bariatric surgical intervention. Clin Exp Dermatol. 2014;39:315-7; quiz 317-8. doi:10.1111/ced.12269
- Mandour MO, Al-Musawi S, Idowu E, et al. Metabolic endoscopy and a simplified low-carbohydrate-high-dietary fiber template as novel treatments for hidradenitis suppurativa—a case series. JAAD Case Rep. 2023;34:23-26. doi:10.1016/j.jdcr.2023.01.035
- Henry T, Cahn B, Haber R, et al. Therapeutic potential of GLP-1 agonists for hidradenitis suppurativa. Int J Dermatol. 2023;62:1543-1544. doi:10.1111/ijd.16892
- Chan LJ, Kaur M, Kaffenberger BH. A case of recalcitrant hidradenitis suppurativa concomitantly treated with tirzepatide. JAAD Case Rep. 2024;52:101-102. doi:10.1016/j.jdcr.2024.02.023
- Costanzo G, Curatolo S, Busà B, et al. Two birds one stone: semaglutide is highly effective against severe psoriasis in a type 2 diabetic patient. Endocrinol Diabetes Metab Case Rep. 2021;2021:21-00007. doi:10.1530 /edm-21-0007
- Buysschaert M, Tennstedt D, Preumont V. Improvement of psoriasis during exenatide treatment in a patient with diabetes. Diabetes Metab. 2012;38:86-88. doi:10.1016/j.diabet.2011.11.004
- Faurschou A, Gyldenløve M, Rohde U, et al. Lack of effect of the glucagonlike peptide-1 receptor agonist liraglutide on psoriasis in glucose-tolerant patients--a randomized placebo-controlled trial. J Eur Acad Dermatol Venereol. 2015;29:555-559. doi:10.1111/jdv.12629
- Ahern T, Tobin AM, Corrigan M, et al. Glucagon-like peptide-1 analogue therapy for psoriasis patients with obesity and type 2 diabetes: a prospective cohort study. J Eur Acad Dermatol Venereol. 2013;27:1440-1443. doi:10.1111/j.1468-3083.2012.04609.x
- Gordon ER, Musleh S, Bordone LA. Treatment of insulin resistance with tirzepatide leading to improvement of hair loss. JAAD Case Rep. 2024;50:123-125. doi:10.1016/j.jdcr.2024.06.001
- Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14:88-98. doi:10.1038/nrendo.2017.151
- Ahmad E, Lim S, Lamptey R, et al. Type 2 diabetes. Lancet. 2022;400: 1803-1820. doi:10.1016/s0140-6736(22)01655-5
- Samson SL, Vellanki P, Blonde L, et al. American Association of Clinical Endocrinology Consensus Statement: comprehensive type 2 diabetes management algorithm—2023 update. Endocr Pract. 2023;29:305-340. doi:10.1016/j.eprac.2023.02.001
- LaMoia TE, Shulman GI. Cellular and molecular mechanisms of metformin action. Endocr Rev. 2021;42:77-96. doi:10.1210/endrev/bnaa023
- Petrasca A, Hambly R, Kearney N, et al. Metformin has antiinflammatory effects and induces immunometabolic reprogramming via multiple mechanisms in hidradenitis suppurativa. Br J Dermatol. 2023;189:730-740. doi:10.1093/bjd/ljad305
- Duan W, Ding Y, Yu X, et al. Metformin mitigates autoimmune insulitis by inhibiting Th1 and Th17 responses while promoting Treg production. Am J Transl Res. 2019;11:2393-2402.
- Bharath LP, Nikolajczyk BS. The intersection of metformin and inflammation. Am J Physiol Cell Physiol. 2021;320:C873-C879. doi:10.1152 /ajpcell.00604.2020
- Sung CT, Chao T, Lee A, et al. Oral metformin for treating dermatological diseases: a systematic review. J Drugs Dermatol. 2020;19:713-720. doi:10.36849/jdd.2020.4874
- Feng J, Wang X, Ye X, et al. Mitochondria as an important target of metformin: the mechanism of action, toxic and side effects, and new therapeutic applications. Pharmacol Res. 2022;177:106114. doi:10.1016/j.phrs.2022.106114
- Klapholz L, Leitersdorf E, Weinrauch L. Leucocytoclastic vasculitis and pneumonitis induced by metformin. Br Med J (Clin Res Ed). 1986;293:483. doi:10.1136/bmj.293.6545.483
- Badr D, Kurban M, Abbas O. Metformin in dermatology: an overview. J Eur Acad Dermatol Venereol. 2013;27:1329-1335. doi:10.1111/jdv.12116
- Czarnowicki T, Ramot Y, Ingber A, et al. Metformin-induced leukocytoclastic vasculitis: a case report. Am J Clin Dermatol. 2012;13:61-63. doi:10.2165/11593230-000000000-00000
- Ben Salem C, Hmouda H, Slim R, et al. Rare case of metformininduced leukocytoclastic vasculitis. Ann Pharmacother. 2006;40:1685-1687. doi:10.1345/aph.1H155
- Abtahi-Naeini B, Momen T, Amiri R, et al. Metformin-induced generalized bullous fixed-drug eruption with a positive dechallengerechallenge test: a case report and literature review. Case Rep Dermatol Med. 2023;2023:6353919. doi:10.1155/2023/6353919
- Al Masri D, Fleifel M, Hirbli K. Fixed drug eruption secondary to four anti-diabetic medications: an unusual case of polysensitivity. Cureus. 2021;13:E18599. doi:10.7759/cureus.18599
- Ramírez-Bellver JL, Lopez J, Macias E, et al. Metformin-induced generalized fixed drug eruption with cutaneous hemophagocytosis. Am J Dermatopathol. 2017;39:471-475. doi:10.1097/dad.0000000000000800
- Steber CJ, Perkins SL, Harris KB. Metformin-induced fixed-drug eruption confirmed by multiple exposures. Am J Case Rep. 2016;17:231-234. doi:10.12659/ajcr.896424
- Voore P, Odigwe C, Mirrakhimov AE, et al. DRESS syndrome following metformin administration: a case report and review of the literature. Am J Ther. 2016;23:E1970-E1973. doi:10.1097/mjt.0000000000000292
- Kastalli S, El Aïdli S, Chaabane A, et al. Photosensitivity induced by metformin: a report of 3 cases. Article in French. Tunis Med. 2009;87:703-705.
- Karadağ AS, You Y, Danarti R, et al. Acanthosis nigricans and the metabolic syndrome. Clin Dermatol. 2018;36:48-53. doi:10.1016/j.clindermatol.2017.09.008
- Kong AS, Williams RL, Smith M, et al. Acanthosis nigricans and diabetes risk factors: prevalence in young persons seen in southwestern US primary care practices. Ann Fam Med. 2007;5:202-208. doi:10.1370/afm.678
- Stuart CA, Gilkison CR, Smith MM, et al. Acanthosis nigricans as a risk factor for non-insulin dependent diabetes mellitus. Clin Pediatr (Phila). 1998;37:73-79. doi:10.1177/000992289803700203
- Hud JA Jr, Cohen JB, Wagner JM, et al. Prevalence and significance of acanthosis nigricans in an adult obese population. Arch Dermatol. 1992;128:941-944.
- Novotny R, Davis J, Butel J, et al. Effect of the Children’s Healthy Living Program on young child overweight, obesity, and acanthosis nigricans in the US-affiliated Pacific region: a randomized clinical trial. JAMA Netw Open. 2018;1:E183896. doi:10.1001/jamanetworkopen.2018.3896
- Romo A, Benavides S. Treatment options in insulin resistance obesityrelated acanthosis nigricans. Ann Pharmacother. 2008;42:1090-1094. doi:10.1345/aph.1K446
- Treesirichod A, Chaithirayanon S, Chaikul T, et al. The randomized trials of 10% urea cream and 0.025% tretinoin cream in the treatment of acanthosis nigricans. J Dermatolog Treat. 2021;32:837-842. doi:10.108 0/09546634.2019.1708855
- Treesirichod A, Chaithirayanon S, Wongjitrat N. Comparison of the efficacy and safety of 0.1% adapalene gel and 0.025% tretinoin cream in the treatment of childhood acanthosis nigricans. Pediatr Dermatol. 2019;36:330-334. doi:10.1111/pde.13799
- Hermanns-Lê T, Hermanns JF, Piérard GE. Juvenile acanthosis nigricans and insulin resistance. Pediatr Dermatol. 2002;19:12-14. doi:10.1046 /j.1525-1470.2002.00013.x
- Walling HW, Messingham M, Myers LM, et al. Improvement of acanthosis nigricans on isotretinoin and metformin. J Drugs Dermatol. 2003;2:677-681.
- Giri D, Alsaffar H, Ramakrishnan R. Acanthosis nigricans and its response to metformin. Pediatr Dermatol. 2017;34:e281-e282. doi:10.1111/pde.13206
- Bellot-Rojas P, Posadas-Sanchez R, Caracas-Portilla N, et al. Comparison of metformin versus rosiglitazone in patients with acanthosis nigricans: a pilot study. J Drugs Dermatol. 2006;5:884-889.
- Sett A, Pradhan S, Sancheti K, et al. Effectiveness and safety of metformin versus Canthex™ in patients with acanthosis nigricans: a randomized, double-blind controlled trial. Indian J Dermatol. 2019;64:115-121. doi:10.4103/ijd.IJD_417_17
- Genazzani AD, Battaglia C, Malavasi B, et al. Metformin administration modulates and restores luteinizing hormone spontaneous episodic secretion and ovarian function in nonobese patients with polycystic ovary syndrome. Fertil Steril. 2004;81:114-119. doi:10.1016 /j.fertnstert.2003.05.020
- Kazerooni T, Dehghan-Kooshkghazi M. Effects of metformin therapy on hyperandrogenism in women with polycystic ovarian syndrome. Gynecol Endocrinol. 2003;17:51-56.
- Kolodziejczyk B, Duleba AJ, Spaczynski RZ, et al. Metformin therapy decreases hyperandrogenism and hyperinsulinemia in women with polycystic ovary syndrome. Fertil Steril. 2000;73:1149-1154. doi:10.1016 /s0015-0282(00)00501-x
- Kelly CJ, Gordon D. The effect of metformin on hirsutism in polycystic ovary syndrome. Eur J Endocrinol. 2002;147:217-221. doi:10.1530/eje.0.1470217
- Harborne L, Fleming R, Lyall H, et al. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88:4116-4123. doi:10.1210/jc.2003-030424
- Rezvanian H, Adibi N, Siavash M, et al. Increased insulin sensitivity by metformin enhances intense-pulsed-light-assisted hair removal in patients with polycystic ovary syndrome. Dermatology. 2009;218: 231-236. doi:10.1159/000187718
- Cosma M, Swiglo BA, Flynn DN, et al. Clinical review: insulin sensitizers for the treatment of hirsutism: a systematic review and metaanalyses of randomized controlled trials. J Clin Endocrinol Metab. 2008;93:1135-1142. doi:10.1210/jc.2007-2429
- Martin KA, Anderson RR, Chang RJ, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103:1233-1257.
- Fraison E, Kostova E, Moran LJ, et al. Metformin versus the combined oral contraceptive pill for hirsutism, acne, and menstrual pattern in polycystic ovary syndrome. Cochrane Database Syst Rev. 2020;8:CD005552. doi:10.1002/14651858.CD005552.pub3
- Hambly R, Kearney N, Hughes R, et al. Metformin treatment of hidradenitis suppurativa: effect on metabolic parameters, inflammation, cardiovascular risk biomarkers, and immune mediators. Int J Mol Sci. 2023;24:6969. doi:10.3390/ijms24086969
- Gold DA, Reeder VJ, Mahan MG, et al. The prevalence of metabolic syndrome in patients with hidradenitis suppurativa. J Am Acad Dermatol. 2014;70:699-703. doi:10.1016/j.jaad.2013.11.014
- Miller IM, Ellervik C, Vinding GR, et al. Association of metabolic syndrome and hidradenitis suppurativa. JAMA Dermatol. 2014;150: 1273-1280. doi:10.1001/jamadermatol.2014.1165
- Monfrecola G, Balato A, Caiazzo G, et al. Mammalian target of rapamycin, insulin resistance and hidradenitis suppurativa: a possible metabolic loop. J Eur Acad Dermatol Venereol. 2016;30:1631-1633. doi:10.1111/jdv.13233
- Yoon MS. The role of mammalian target of rapamycin (mTOR) in insulin signaling. Nutrients. 2017;9:1176. doi:10.3390/nu9111176
- Abu Rached N, Gambichler T, Dietrich JW, et al. The role of hormones in hidradenitis suppurativa: a systematic review. Int J Mol Sci. 2022;23:15250. doi:10.3390/ijms232315250
- Verdolini R, Clayton N, Smith A, et al. Metformin for the treatment of hidradenitis suppurativa: a little help along the way. J Eur Acad Dermatol Venereol. 2013;27:1101-1108. doi:10.1111/j.1468-3083.2012.04668.x
- Tsentemeidou A, Vakirlis E, Papadimitriou I, et al. Metformin in hidradenitis suppurativa: is it worth pursuing further? Skin Appendage Disord. 2023;9:187-190. doi:10.1159/000529359
- Arun B, Loffeld A. Long-standing hidradenitis suppurativa treated effectively with metformin. Clin Exp Dermatol. 2009;34:920-921. doi:10.1111/j.1365-2230.2008.03121.x
- Jennings L, Hambly R, Hughes R, et al. Metformin use in hidradenitis suppurativa. J Dermatolog Treat. 2020;31:261-263. doi:10.1080/09546634 .2019.1592100
- Moussa C, Wadowski L, Price H, et al. Metformin as adjunctive therapy for pediatric patients with hidradenitis suppurativa. J Drugs Dermatol. 2020;19:1231-1234. doi:10.36849/jdd.2020.5447
- Cho M, Woo YR, Cho SH, et al. Metformin: a potential treatment for acne, hidradenitis suppurativa and rosacea. Acta Derm Venereol. 2023;103:adv18392. doi:10.2340/actadv.v103.18392
- Del Rosso JQ, Kircik L. The cutaneous effects of androgens and androgen-mediated sebum production and their pathophysiologic and therapeutic importance in acne vulgaris. J Dermatolog Treat. 2024;35:2298878. doi:10.1080/09546634.2023.2298878
- El-Tahlawi S, Ezzat Mohammad N, Mohamed El-Amir A, et al. Survivin and insulin-like growth factor-I: potential role in the pathogenesis of acne and post-acne scar. Scars Burn Heal. 2019;5:2059513118818031. doi:10.1177/2059513118818031
- Albalat W, Darwish H, Abd-Elaal WH, et al. The potential role of insulin-like growth factor 1 in acne vulgaris and its correlation with the clinical response before and after treatment with metformin. J Cosmet Dermatol. 2022;21:6209-6214. doi:10.1111/jocd.15210
- Nguyen S, Nguyen ML, Roberts WS, et al. The efficacy of metformin as a therapeutic agent in the treatment of acne vulgaris: a systematic review. Cureus. 2024;16:E56246. doi:10.7759/cureus.56246
- Boehncke WH, Schön MP. Psoriasis. Lancet. 2015;386:983-994. doi:10.1016 /s0140-6736(14)61909-7
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113. doi:10.1016/j.jaad.2018.11.058
- Huang Z, Li J, Chen H, et al. The efficacy of metformin for the treatment of psoriasis: a meta-analysis study. Postepy Dermatol Alergol. 2023;40:606-610. doi:10.5114/ada.2023.130524
- Singh S, Bhansali A. Randomized placebo control study of insulin sensitizers (metformin and pioglitazone) in psoriasis patients with metabolic syndrome (topical treatment cohort). BMC Dermatol. 2016;16:12. doi:10.1186 /s12895-016-0049-y
- Bao A, Qadri A, Gadre A, et al. Low-dose metformin and profibrotic signature in central centrifugal cicatricial alopecia. JAMA Dermatol. 2024;E243062. doi:10.1001/jamadermatol.2024.3062
- Lawson CN, Bakayoko A, Callender VD. Central centrifugal cicatricial alopecia: challenges and treatments. Dermatol Clin. 2021;39:389-405. doi:10.1016/j.det.2021.03.004
- Gathers RC, Lim HW. Central centrifugal cicatricial alopecia: past, present, and future. J Am Acad Dermatol. 2009;60:660-668. doi:10.1016/j .jaad.2008.09.066
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j.jdcr.2019.12.008
- Foretz M, Guigas B, Bertrand L, et al. Metformin: from mechanisms of action to therapies. Cell Metab. 2014;20:953-966. doi:10.1016 /j.cmet.2014.09.018
- Aguh C, Dina Y, Talbot CC Jr, et al. Fibroproliferative genes are preferentially expressed in central centrifugal cicatricial alopecia. J Am Acad Dermatol. 2018;79:904-912.e1. doi:10.1016/j.jaad.2018.05.1257
- Gadre A, Dyson T, Jedrych J, et al. Proteomic profiling of central centrifugal cicatricial alopecia reveals role of humoral immune response pathway and metabolic dysregulation. JID Innov. 2024;4:100263. doi:10.1016/j.xjidi.2024.100263
- Chaudhary SC, Kurundkar D, Elmets CA, et al. Metformin, an antidiabetic agent reduces growth of cutaneous squamous cell carcinoma by targeting mTOR signaling pathway. Photochem Photobiol. 2012;88:1149-1156. doi:10.1111/j.1751-1097.2012.01165.x
- Tomic T, Botton T, Cerezo M, et al. Metformin inhibits melanoma development through autophagy and apoptosis mechanisms. Cell Death Dis. 2011;2:e199. doi:10.1038/cddis.2011.86
- Mascaraque-Checa M, Gallego-Rentero M, Nicolás-Morala J, et al. Metformin overcomes metabolic reprogramming-induced resistance of skin squamous cell carcinoma to photodynamic therapy. Mol Metab. 2022;60:101496. doi:10.1016/j.molmet.2022.101496
- Mascaraque M, Delgado-Wicke P, Nuevo-Tapioles C, et al. Metformin as an adjuvant to photodynamic therapy in resistant basal cell carcinoma cells. Cancers (Basel). 2020;12:668. doi:10.3390/cancers12030668
- Chang MS, Hartman RI, Xue J, et al. Risk of skin cancer associated with metformin use: a meta-analysis of randomized controlled trials and observational studies. Cancer Prev Res (Phila). 2021;14:77-84. doi:10.1158/1940-6207.Capr-20-0376
- Augustin RC, Huang Z, Ding F, et al. Metformin is associated with improved clinical outcomes in patients with melanoma: a retrospective, multi-institutional study. Front Oncol. 2023;13:1075823. doi:10.3389 /fonc.2023.1075823
- Adalsteinsson JA, Muzumdar S, Waldman R, et al. Metformin is associated with decreased risk of basal cell carcinoma: a whole-population casecontrol study from Iceland. J Am Acad Dermatol. 2021;85:56-61. doi:10.1016/j.jaad.2021.02.042
- Jennings L, Nestor L, Molloy O, et al. The treatment of hidradenitis suppurativa with the glucagon-like peptide-1 agonist liraglutide. Br J Dermatol. 2017;177:858-859. doi:10.1111/bjd.15233
- Lin L, Xu X, Yu Y, et al. Glucagon-like peptide-1 receptor agonist liraglutide therapy for psoriasis patients with type 2 diabetes: a randomized-controlled trial. J Dermatolog Treat. 2022;33: 1428-1434. doi:10.1080/09546634.2020.1826392
- Karacabeyli D, Lacaille D. Glucagon-like peptide 1 receptor agonists in patients with inflammatory arthritis or psoriasis: a scoping review. J Clin Rheumatol. 2024;30:26-31. doi:10.1097/rhu.0000000000001949
- Yang J, Wang Z, Zhang X. GLP-1 receptor agonist impairs keratinocytes inflammatory signals by activating AMPK. Exp Mol Pathol. 2019;107: 124-128. doi:10.1016/j.yexmp.2019.01.014
- Buysschaert M, Baeck M, Preumont V, et al. Improvement of psoriasis during glucagon-like peptide-1 analogue therapy in type 2 diabetes is associated with decreasing dermal Υϛ T-cell number: a prospective case-series study. Br J Dermatol. 2014;171:155-161. doi:10.1111/bjd.12886
- Wilbon SS, Kolonin MG. GLP1 receptor agonists-effects beyond obesity and diabetes. Cells. 2023;13:65. doi:10.3390/cells13010065
- Filippatos TD, Panagiotopoulou TV, Elisaf MS. Adverse effects of GLP-1 receptor agonists. Rev Diabet Stud. 2014;11:202-230. doi:10.1900 /rds.2014.11.202
- He Z, Tabe AN, Rana S, et al. Tirzepatide-induced biphasic anaphylactic reaction: a case report. Cureus. 2023;15:e50112. doi:10.7759/cureus.50112
- Anthony MS, Aroda VR, Parlett LE, et al. Risk of anaphylaxis among new users of glp-1 receptor agonists: a cohort study. Diabetes Care. 2024;47:712-719. doi:10.2337/dc23-1911
- Salazar CE, Patil MK, Aihie O, et al. Rare cutaneous adverse reactions associated with GLP-1 agonists: a review of the published literature. Arch Dermatol Res. 2024;316:248. doi:10.1007/s00403-024-02969-3
- Tran MM, Mirza FN, Lee AC, et al. Dermatologic findings associated with semaglutide use: a scoping review. J Am Acad Dermatol. 2024;91:166-168. doi:10.1016/j.jaad.2024.03.021
- Castellanos V, Workneh H, Malik A, et al. Semaglutide-induced lupus erythematosus with multiorgan involvement. Cureus. 2024;16:E55324. doi:10.7759/cureus.55324
- Boccardi A, Shubrook JH. Cutaneous reactions to antidiabetic agents: a narrative review. Diabetology. 2022;3:97-107.
- Desai DD, Sikora M, Nohria A, et al. GLP-1 agonists and hair loss: a call for further investigation. Int J Dermatol. 2024;63:1128-1130. doi:10.1111 /ijd.17246
- Mansour MR, Hannawa OM, Yaldo MM, et al. The rise of “Ozempic face”: analyzing trends and treatment challenges associated with rapid facial weight loss induced by GLP-1 agonists. J Plast Reconstr Aesthet Surg. 2024;96:225-227. doi:10.1016/j.bjps.2024.07.051
- Sivanand A, Gulliver WP, Josan CK, et al. Weight loss and dietary interventions for hidradenitis suppurativa: a systematic review. J Cutan Med Surg. 2020;24:64-72. doi:10.1177/1203475419874412
- Boer J. Resolution of hidradenitis suppurativa after weight loss by dietary measures, especially on frictional locations. J Eur Acad Dermatol Venereol. 2016;30:895-896. doi:10.1111/jdv.13059
- Thomas CL, Gordon KD, Mortimer PS. Rapid resolution of hidradenitis suppurativa after bariatric surgical intervention. Clin Exp Dermatol. 2014;39:315-7; quiz 317-8. doi:10.1111/ced.12269
- Mandour MO, Al-Musawi S, Idowu E, et al. Metabolic endoscopy and a simplified low-carbohydrate-high-dietary fiber template as novel treatments for hidradenitis suppurativa—a case series. JAAD Case Rep. 2023;34:23-26. doi:10.1016/j.jdcr.2023.01.035
- Henry T, Cahn B, Haber R, et al. Therapeutic potential of GLP-1 agonists for hidradenitis suppurativa. Int J Dermatol. 2023;62:1543-1544. doi:10.1111/ijd.16892
- Chan LJ, Kaur M, Kaffenberger BH. A case of recalcitrant hidradenitis suppurativa concomitantly treated with tirzepatide. JAAD Case Rep. 2024;52:101-102. doi:10.1016/j.jdcr.2024.02.023
- Costanzo G, Curatolo S, Busà B, et al. Two birds one stone: semaglutide is highly effective against severe psoriasis in a type 2 diabetic patient. Endocrinol Diabetes Metab Case Rep. 2021;2021:21-00007. doi:10.1530 /edm-21-0007
- Buysschaert M, Tennstedt D, Preumont V. Improvement of psoriasis during exenatide treatment in a patient with diabetes. Diabetes Metab. 2012;38:86-88. doi:10.1016/j.diabet.2011.11.004
- Faurschou A, Gyldenløve M, Rohde U, et al. Lack of effect of the glucagonlike peptide-1 receptor agonist liraglutide on psoriasis in glucose-tolerant patients--a randomized placebo-controlled trial. J Eur Acad Dermatol Venereol. 2015;29:555-559. doi:10.1111/jdv.12629
- Ahern T, Tobin AM, Corrigan M, et al. Glucagon-like peptide-1 analogue therapy for psoriasis patients with obesity and type 2 diabetes: a prospective cohort study. J Eur Acad Dermatol Venereol. 2013;27:1440-1443. doi:10.1111/j.1468-3083.2012.04609.x
- Gordon ER, Musleh S, Bordone LA. Treatment of insulin resistance with tirzepatide leading to improvement of hair loss. JAAD Case Rep. 2024;50:123-125. doi:10.1016/j.jdcr.2024.06.001
Dermatologic Implications of Glycemic Control Medications for Patients with Type 2 Diabetes Mellitus
Dermatologic Implications of Glycemic Control Medications for Patients with Type 2 Diabetes Mellitus
PRACTICE POINTS
- Type 2 diabetes mellitus (T2DM) is highly prevalent in patients with various dermatologic conditions; therefore, it is important for dermatologists to understand the adverse effects of T2DM medications to optimize treatment strategies.
- In addition to glycemic control and management, the hormonal and immunologic effects of T2DM medications can be leveraged to treat dermatologic conditions, particularly those associated with metabolic dysregulation.
Combined Clinics, Personalized Medicine for Psoriatic Disease Face Barriers
NEW YORK CITY — The idea of having dermatologists and rheumatologists under one roof to see patients with psoriasis prone to psoriatic arthritis (PsA) — a concept known as combined clinics — has been around for more than a decade, and the idea of personalized medicine for these patients even longer than that, yet both approaches to care have encountered a host of obstacles, a longtime research rheumatologist said.
“It’s important that we work together, but there is a problem in terms of staffing — managing the meetings with patients together — and in the states in particular it’s a matter of who’s charging for what,” Dafna Gladman, MD, a rheumatologist at the University of Toronto, in Ontario, Canada, told attendees at the annual New York University (NYU) Langone Advanced Seminar in Psoriasis and Psoriatic Arthritis. Her institution has one of the 44 worldwide combined clinics registered in the Psoriasis & Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN), of which Gladman is an advisory board member.
Barriers to Combined Clinics
“Some of the barriers are physical in the sense that, for the dermatologists and rheumatologists to work at the same time, you need the right space, and in many places, you just don’t have the space to have the two specialists sitting at the same time,” Gladman told Medscape Medical News.
Some centers get around this by having the dermatology and rheumatology clinics next to or near each other. “So these two specialists are close enough to be able to go from room to room,” she added.
Another challenge facing combined clinics lies in the nature of how dermatologists and rheumatologists see patients. “The dermatologist sees patients a lot faster than the rheumatologist, so if the dermatologist and rheumatologist are sitting together, the dermatologist may not see as many patients as they would otherwise and therefore may not get reimbursed properly,” Gladman said.
To overcome these challenges, different models have emerged, Gladman said. If space allows, the ideal model is to have both specialties in one clinic, she said, while compensating for the different pace at which dermatologists and rheumatologists see patients.
The other model is to locate the two clinics close enough so that a person with suspected PsA can get to the rheumatology clinic soon after their dermatologic consult, or the rheumatologist can go to the dermatology clinic, Gladman said. Or the situation may be reversed when the rheumatologist needs a dermatology consult, she added.
When that’s not possible, a virtual visit may be the solution, Gladman said. She noted that PPACMAN offers ways to overcome the challenges of running a combined clinic.
Whatever combined clinic model a center chooses, clinicians must be mindful of preventing patients from falling through the cracks, Gladman said.
“When you treat patients separately, the patient sees the rheumatologist, and the rheumatologist wants to do one thing; then they go to the dermatologist and the dermatologist wants to do another thing, and the patient doesn’t do anything because they don’t know what to choose,” she said.
The combined clinic allows the patient to get the opinions of both specialists and avoid the uncertainty about the course of treatment, Gladman added.
Some combined clinics may also house other specialists, such as gastroenterologists, cardiologists, and nurse practitioners, noted Jose U. Scher, MD, director of the Arthritis Clinic and Psoriatic Arthritis Center at NYU Langone Health in New York City. Such centers are typically in academic centers “given challenges with space, scheduling, and reimbursement,” he told Medscape Medical News. NYU has a PPACMAN-registered combined clinic.
Regardless of how combined clinics are organized, Scher said, “We have found that the most important aspect of combined clinics is the open communication and integration of care between and amongst specialists and patients.”
The Potential of Personalized Medicine
“Personalized medicine is where we need to get to,” Gladman told seminar attendees. She said she had hoped it would be further along by now and be more integrated into the care of patients with psoriasis and PsA. “The idea is to identify psoriasis patients that are destined to develop psoriatic arthritis,” she said.
Besides that, identifying biomarkers is key to advancing personalized medicine for psoriasis, Gladman noted.
“In the skin, it’s easy; even the patient can assess their psoriasis,” she said. “But in the joints, it’s very difficult, so it would be nice to have some kind of biomarker, whether it’s the blood or an imaging modality. We want to identify the biomarkers for drug response or lack thereof so we know what drugs would be appropriate for the individual patient, and therefore, we can provide the right drug for the right person and fortunately at the right time.”
In explaining why personalized medicine isn’t further along in dermatology and rheumatology, Gladman told Medscape Medical News, “It’s a matter of finding the right things; we haven’t solved the mystery.” She cited a previous discussion at the seminar about the pathogenesis of PsA. “One person thinks it’s the bone marrow and another thinks it’s the T cells, so we haven’t quite put it all together to have a definitive answer.”
Personalized medicine in psoriasis and PsA is a “key unmet need,” Scher said. “Multiomics” — a biological analysis approach that uses multiple “omes,” such as the genome and microbiome — digital features, and wearables “can unlock novel diagnostic and therapeutic pathways that are desperately needed to enhance clinical response in PsA,” he said.
Also emerging are humanized animal models for laboratory research, which Scher called “potentially very useful tools to personalize approaches to PsA pathogenesis and treatment.”
Gladman disclosed financial relationships with AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Scher had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW YORK CITY — The idea of having dermatologists and rheumatologists under one roof to see patients with psoriasis prone to psoriatic arthritis (PsA) — a concept known as combined clinics — has been around for more than a decade, and the idea of personalized medicine for these patients even longer than that, yet both approaches to care have encountered a host of obstacles, a longtime research rheumatologist said.
“It’s important that we work together, but there is a problem in terms of staffing — managing the meetings with patients together — and in the states in particular it’s a matter of who’s charging for what,” Dafna Gladman, MD, a rheumatologist at the University of Toronto, in Ontario, Canada, told attendees at the annual New York University (NYU) Langone Advanced Seminar in Psoriasis and Psoriatic Arthritis. Her institution has one of the 44 worldwide combined clinics registered in the Psoriasis & Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN), of which Gladman is an advisory board member.
Barriers to Combined Clinics
“Some of the barriers are physical in the sense that, for the dermatologists and rheumatologists to work at the same time, you need the right space, and in many places, you just don’t have the space to have the two specialists sitting at the same time,” Gladman told Medscape Medical News.
Some centers get around this by having the dermatology and rheumatology clinics next to or near each other. “So these two specialists are close enough to be able to go from room to room,” she added.
Another challenge facing combined clinics lies in the nature of how dermatologists and rheumatologists see patients. “The dermatologist sees patients a lot faster than the rheumatologist, so if the dermatologist and rheumatologist are sitting together, the dermatologist may not see as many patients as they would otherwise and therefore may not get reimbursed properly,” Gladman said.
To overcome these challenges, different models have emerged, Gladman said. If space allows, the ideal model is to have both specialties in one clinic, she said, while compensating for the different pace at which dermatologists and rheumatologists see patients.
The other model is to locate the two clinics close enough so that a person with suspected PsA can get to the rheumatology clinic soon after their dermatologic consult, or the rheumatologist can go to the dermatology clinic, Gladman said. Or the situation may be reversed when the rheumatologist needs a dermatology consult, she added.
When that’s not possible, a virtual visit may be the solution, Gladman said. She noted that PPACMAN offers ways to overcome the challenges of running a combined clinic.
Whatever combined clinic model a center chooses, clinicians must be mindful of preventing patients from falling through the cracks, Gladman said.
“When you treat patients separately, the patient sees the rheumatologist, and the rheumatologist wants to do one thing; then they go to the dermatologist and the dermatologist wants to do another thing, and the patient doesn’t do anything because they don’t know what to choose,” she said.
The combined clinic allows the patient to get the opinions of both specialists and avoid the uncertainty about the course of treatment, Gladman added.
Some combined clinics may also house other specialists, such as gastroenterologists, cardiologists, and nurse practitioners, noted Jose U. Scher, MD, director of the Arthritis Clinic and Psoriatic Arthritis Center at NYU Langone Health in New York City. Such centers are typically in academic centers “given challenges with space, scheduling, and reimbursement,” he told Medscape Medical News. NYU has a PPACMAN-registered combined clinic.
Regardless of how combined clinics are organized, Scher said, “We have found that the most important aspect of combined clinics is the open communication and integration of care between and amongst specialists and patients.”
The Potential of Personalized Medicine
“Personalized medicine is where we need to get to,” Gladman told seminar attendees. She said she had hoped it would be further along by now and be more integrated into the care of patients with psoriasis and PsA. “The idea is to identify psoriasis patients that are destined to develop psoriatic arthritis,” she said.
Besides that, identifying biomarkers is key to advancing personalized medicine for psoriasis, Gladman noted.
“In the skin, it’s easy; even the patient can assess their psoriasis,” she said. “But in the joints, it’s very difficult, so it would be nice to have some kind of biomarker, whether it’s the blood or an imaging modality. We want to identify the biomarkers for drug response or lack thereof so we know what drugs would be appropriate for the individual patient, and therefore, we can provide the right drug for the right person and fortunately at the right time.”
In explaining why personalized medicine isn’t further along in dermatology and rheumatology, Gladman told Medscape Medical News, “It’s a matter of finding the right things; we haven’t solved the mystery.” She cited a previous discussion at the seminar about the pathogenesis of PsA. “One person thinks it’s the bone marrow and another thinks it’s the T cells, so we haven’t quite put it all together to have a definitive answer.”
Personalized medicine in psoriasis and PsA is a “key unmet need,” Scher said. “Multiomics” — a biological analysis approach that uses multiple “omes,” such as the genome and microbiome — digital features, and wearables “can unlock novel diagnostic and therapeutic pathways that are desperately needed to enhance clinical response in PsA,” he said.
Also emerging are humanized animal models for laboratory research, which Scher called “potentially very useful tools to personalize approaches to PsA pathogenesis and treatment.”
Gladman disclosed financial relationships with AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Scher had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
NEW YORK CITY — The idea of having dermatologists and rheumatologists under one roof to see patients with psoriasis prone to psoriatic arthritis (PsA) — a concept known as combined clinics — has been around for more than a decade, and the idea of personalized medicine for these patients even longer than that, yet both approaches to care have encountered a host of obstacles, a longtime research rheumatologist said.
“It’s important that we work together, but there is a problem in terms of staffing — managing the meetings with patients together — and in the states in particular it’s a matter of who’s charging for what,” Dafna Gladman, MD, a rheumatologist at the University of Toronto, in Ontario, Canada, told attendees at the annual New York University (NYU) Langone Advanced Seminar in Psoriasis and Psoriatic Arthritis. Her institution has one of the 44 worldwide combined clinics registered in the Psoriasis & Psoriatic Arthritis Clinics Multicenter Advancement Network (PPACMAN), of which Gladman is an advisory board member.
Barriers to Combined Clinics
“Some of the barriers are physical in the sense that, for the dermatologists and rheumatologists to work at the same time, you need the right space, and in many places, you just don’t have the space to have the two specialists sitting at the same time,” Gladman told Medscape Medical News.
Some centers get around this by having the dermatology and rheumatology clinics next to or near each other. “So these two specialists are close enough to be able to go from room to room,” she added.
Another challenge facing combined clinics lies in the nature of how dermatologists and rheumatologists see patients. “The dermatologist sees patients a lot faster than the rheumatologist, so if the dermatologist and rheumatologist are sitting together, the dermatologist may not see as many patients as they would otherwise and therefore may not get reimbursed properly,” Gladman said.
To overcome these challenges, different models have emerged, Gladman said. If space allows, the ideal model is to have both specialties in one clinic, she said, while compensating for the different pace at which dermatologists and rheumatologists see patients.
The other model is to locate the two clinics close enough so that a person with suspected PsA can get to the rheumatology clinic soon after their dermatologic consult, or the rheumatologist can go to the dermatology clinic, Gladman said. Or the situation may be reversed when the rheumatologist needs a dermatology consult, she added.
When that’s not possible, a virtual visit may be the solution, Gladman said. She noted that PPACMAN offers ways to overcome the challenges of running a combined clinic.
Whatever combined clinic model a center chooses, clinicians must be mindful of preventing patients from falling through the cracks, Gladman said.
“When you treat patients separately, the patient sees the rheumatologist, and the rheumatologist wants to do one thing; then they go to the dermatologist and the dermatologist wants to do another thing, and the patient doesn’t do anything because they don’t know what to choose,” she said.
The combined clinic allows the patient to get the opinions of both specialists and avoid the uncertainty about the course of treatment, Gladman added.
Some combined clinics may also house other specialists, such as gastroenterologists, cardiologists, and nurse practitioners, noted Jose U. Scher, MD, director of the Arthritis Clinic and Psoriatic Arthritis Center at NYU Langone Health in New York City. Such centers are typically in academic centers “given challenges with space, scheduling, and reimbursement,” he told Medscape Medical News. NYU has a PPACMAN-registered combined clinic.
Regardless of how combined clinics are organized, Scher said, “We have found that the most important aspect of combined clinics is the open communication and integration of care between and amongst specialists and patients.”
The Potential of Personalized Medicine
“Personalized medicine is where we need to get to,” Gladman told seminar attendees. She said she had hoped it would be further along by now and be more integrated into the care of patients with psoriasis and PsA. “The idea is to identify psoriasis patients that are destined to develop psoriatic arthritis,” she said.
Besides that, identifying biomarkers is key to advancing personalized medicine for psoriasis, Gladman noted.
“In the skin, it’s easy; even the patient can assess their psoriasis,” she said. “But in the joints, it’s very difficult, so it would be nice to have some kind of biomarker, whether it’s the blood or an imaging modality. We want to identify the biomarkers for drug response or lack thereof so we know what drugs would be appropriate for the individual patient, and therefore, we can provide the right drug for the right person and fortunately at the right time.”
In explaining why personalized medicine isn’t further along in dermatology and rheumatology, Gladman told Medscape Medical News, “It’s a matter of finding the right things; we haven’t solved the mystery.” She cited a previous discussion at the seminar about the pathogenesis of PsA. “One person thinks it’s the bone marrow and another thinks it’s the T cells, so we haven’t quite put it all together to have a definitive answer.”
Personalized medicine in psoriasis and PsA is a “key unmet need,” Scher said. “Multiomics” — a biological analysis approach that uses multiple “omes,” such as the genome and microbiome — digital features, and wearables “can unlock novel diagnostic and therapeutic pathways that are desperately needed to enhance clinical response in PsA,” he said.
Also emerging are humanized animal models for laboratory research, which Scher called “potentially very useful tools to personalize approaches to PsA pathogenesis and treatment.”
Gladman disclosed financial relationships with AbbVie, Amgen, Bristol Myers Squibb, Eli Lilly, Galapagos, Gilead, Janssen, Novartis, Pfizer, and UCB. Scher had no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA Approves Ustekinumab Biosimilar Steqeyma, the Seventh of Its Kind
The Food and Drug Administration (FDA) has approved ustekinumab-stba (Steqeyma) as a biosimilar to the interleukin-12 and -23 inhibitor ustekinumab (Stelara) for the treatment of adults with active Crohn’s disease or ulcerative colitis and for both children aged ≥ 6 years and adults with moderate to severe plaque psoriasis or active psoriatic arthritis.
This is the seventh ustekinumab biosimilar approved by the FDA. The biosimilar, developed by Celltrion, has a license entry date in February 2025 as part of the settlement and license agreement with the manufacturer of the reference biologic, Johnson & Johnson.
Ustekinumab-stba will be available in two formulations: A subcutaneous injection in two strengths — a 45 mg/0.5 mL or 90 mg/1 mL solution in a single-dose, prefilled syringe — and an intravenous infusion of a 130 mg/26 mL (5 mg/mL) solution in a single-dose vial.
“The approval of Steqeyma reflects Celltrion’s continued investment in providing treatment options to patients diagnosed with ulcerative colitis, Crohn’s disease, psoriasis, and psoriatic arthritis,” said Thomas Nusbickel, Chief Commercial Officer at Celltrion USA, Jersey City, New Jersey, in a press release.
The FDA has previously approved the company’s adalimumab biosimilar Yuflyma and its infliximab biosimilar Zymfentra.
The full prescribing information for ustekinumab-stba is available here.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved ustekinumab-stba (Steqeyma) as a biosimilar to the interleukin-12 and -23 inhibitor ustekinumab (Stelara) for the treatment of adults with active Crohn’s disease or ulcerative colitis and for both children aged ≥ 6 years and adults with moderate to severe plaque psoriasis or active psoriatic arthritis.
This is the seventh ustekinumab biosimilar approved by the FDA. The biosimilar, developed by Celltrion, has a license entry date in February 2025 as part of the settlement and license agreement with the manufacturer of the reference biologic, Johnson & Johnson.
Ustekinumab-stba will be available in two formulations: A subcutaneous injection in two strengths — a 45 mg/0.5 mL or 90 mg/1 mL solution in a single-dose, prefilled syringe — and an intravenous infusion of a 130 mg/26 mL (5 mg/mL) solution in a single-dose vial.
“The approval of Steqeyma reflects Celltrion’s continued investment in providing treatment options to patients diagnosed with ulcerative colitis, Crohn’s disease, psoriasis, and psoriatic arthritis,” said Thomas Nusbickel, Chief Commercial Officer at Celltrion USA, Jersey City, New Jersey, in a press release.
The FDA has previously approved the company’s adalimumab biosimilar Yuflyma and its infliximab biosimilar Zymfentra.
The full prescribing information for ustekinumab-stba is available here.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration (FDA) has approved ustekinumab-stba (Steqeyma) as a biosimilar to the interleukin-12 and -23 inhibitor ustekinumab (Stelara) for the treatment of adults with active Crohn’s disease or ulcerative colitis and for both children aged ≥ 6 years and adults with moderate to severe plaque psoriasis or active psoriatic arthritis.
This is the seventh ustekinumab biosimilar approved by the FDA. The biosimilar, developed by Celltrion, has a license entry date in February 2025 as part of the settlement and license agreement with the manufacturer of the reference biologic, Johnson & Johnson.
Ustekinumab-stba will be available in two formulations: A subcutaneous injection in two strengths — a 45 mg/0.5 mL or 90 mg/1 mL solution in a single-dose, prefilled syringe — and an intravenous infusion of a 130 mg/26 mL (5 mg/mL) solution in a single-dose vial.
“The approval of Steqeyma reflects Celltrion’s continued investment in providing treatment options to patients diagnosed with ulcerative colitis, Crohn’s disease, psoriasis, and psoriatic arthritis,” said Thomas Nusbickel, Chief Commercial Officer at Celltrion USA, Jersey City, New Jersey, in a press release.
The FDA has previously approved the company’s adalimumab biosimilar Yuflyma and its infliximab biosimilar Zymfentra.
The full prescribing information for ustekinumab-stba is available here.
A version of this article first appeared on Medscape.com.
Optimal Exercise Levels for Dermatology Patients With Psoriasis
Optimal Exercise Levels for Dermatology Patients With Psoriasis
There is a direct link between psoriasis and metabolic conditions such as diabetes mellitus and obesity.1 Exercise of varied intensity in patients with chronic inflammatory and metabolic conditions can help improve quality of life and severity of disease; however, there has not been a clear consensus on the recommended duration and types of exercise that are most advantageous.1-5 We reviewed the literature to identify physical and mental health impacts of exercise on patients with psoriasis, and we present the recommended duration and types of exercise that are most impactful for these patients.
One indicator of the link between psoriasis and exercise is the level of peroxisome proliferator activated receptor gamma coactivator-1 α (PGC-1α) in muscle cells.2 This marker reduces inflammation. When levels are low in muscle cells, an induction occurs that leads to systemic or local inflammation; however, skeletal muscle PGC-1α levels increase following exercise, indicating reduced inflammation.2 The level of PGC-1α is measured through muscle biopsy and polymerase chain reaction.6 Another indicator of the correlation between exercise and inflammation is lipoprotein-associated phospholipase A2, which is produced by inflammatory cells and has a correlation with cardiovascular disease. Exercise reduces lipoprotein-associated phospholipase A2 levels, and a sedentary lifestyle correlates with increased levels of this marker.3 Lipoprotein-associated phospholipase A2 is measured through an enzyme-linked immunosorbent assay of the blood, with levels around 200 ng/mL considered high.7 Patients with psoriasis are 30% less likely to participate in physical activity compared to patients without psoriasis, which can be attributed to psychosocial impairment and other factors. Sedentary lifestyle is associated with new or worsening metabolic disease and prevalence of psoriatic lesions.1
A metabolic equivalent task score is a classification system that measures the rate of the body’s oxygen uptake for any given activity.4 A score of 20.9 or more metabolic equivalent task hours of vigorous exercise per week—equal to 105 minutes of running or 180 minutes of swimming or playing tennis—is linked with a 25% to 30% risk reduction of psoriasis in women.4 Therefore, we recommend 30 minutes of exercise 4 to 5 times per week for women. These periods of exercise should consist mainly of activities that will not cause psoriasis flares due to excessive sweating, skin trauma, or prolonged sun exposure.5 Walking, yoga, and bike riding all could be good exercise options for those with psoriasis. The National Psoriasis Foundation offers guidance on physical activity in patients with psoriasis or psoriatic arthritis.8 Psoriasis has apparent physical and psychosocial impacts on patients that can be prevented and improved through the exercise recommendations presented in this article. Dermatologists should use these recommendations to address psoriasis in their everyday practice.
- Do YK, Lakhani N, Malhotra R, et al. Association between psoriasis and leisure-time physical activity: findings from the National Health and Nutrition Examination Survey. J Dermatol. 2015;42:148-153. doi:10.1111/1346-8138.12721
- Handschin C, Spiegelman BM. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature. 2008;454: 463-469. doi:10.1038/nature07206
- Clark K, Sharp S, Womack CJ, et al. Increased sedentary time and decreased physical activity increases lipoprotein associated phospholipase A2 in obese individuals. Nutr Metab Cardiovasc Dis. 2022;32:1703-1710. doi:10.1016/j.numecd.2022.04.023
- Yeh C, Flatley E, Elkattawy O, et al. Exercise in dermatology: exercise’s influence on skin aging, skin cancer, psoriasis, venous ulcers, and androgenetic alopecia. J Am Acad Dermatol. 2022;87:183-184. doi:10.1016/j.jaad.2021.07.023
- Sheppard R, Gan WK, Onambele-Pearson GL, et al. Developing an aerobic exercise intervention for patients with psoriasis to support lifestyle behaviour change and improve health outcomes. Clin Exp Dermatol. 2023;48:5-11. doi:10.1093/ced/llac008
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904.023
- National Psoriasis Foundation. Active and mindful lifestyles. https://www.psoriasis.org/active-and-mindful-lifestyles/
There is a direct link between psoriasis and metabolic conditions such as diabetes mellitus and obesity.1 Exercise of varied intensity in patients with chronic inflammatory and metabolic conditions can help improve quality of life and severity of disease; however, there has not been a clear consensus on the recommended duration and types of exercise that are most advantageous.1-5 We reviewed the literature to identify physical and mental health impacts of exercise on patients with psoriasis, and we present the recommended duration and types of exercise that are most impactful for these patients.
One indicator of the link between psoriasis and exercise is the level of peroxisome proliferator activated receptor gamma coactivator-1 α (PGC-1α) in muscle cells.2 This marker reduces inflammation. When levels are low in muscle cells, an induction occurs that leads to systemic or local inflammation; however, skeletal muscle PGC-1α levels increase following exercise, indicating reduced inflammation.2 The level of PGC-1α is measured through muscle biopsy and polymerase chain reaction.6 Another indicator of the correlation between exercise and inflammation is lipoprotein-associated phospholipase A2, which is produced by inflammatory cells and has a correlation with cardiovascular disease. Exercise reduces lipoprotein-associated phospholipase A2 levels, and a sedentary lifestyle correlates with increased levels of this marker.3 Lipoprotein-associated phospholipase A2 is measured through an enzyme-linked immunosorbent assay of the blood, with levels around 200 ng/mL considered high.7 Patients with psoriasis are 30% less likely to participate in physical activity compared to patients without psoriasis, which can be attributed to psychosocial impairment and other factors. Sedentary lifestyle is associated with new or worsening metabolic disease and prevalence of psoriatic lesions.1
A metabolic equivalent task score is a classification system that measures the rate of the body’s oxygen uptake for any given activity.4 A score of 20.9 or more metabolic equivalent task hours of vigorous exercise per week—equal to 105 minutes of running or 180 minutes of swimming or playing tennis—is linked with a 25% to 30% risk reduction of psoriasis in women.4 Therefore, we recommend 30 minutes of exercise 4 to 5 times per week for women. These periods of exercise should consist mainly of activities that will not cause psoriasis flares due to excessive sweating, skin trauma, or prolonged sun exposure.5 Walking, yoga, and bike riding all could be good exercise options for those with psoriasis. The National Psoriasis Foundation offers guidance on physical activity in patients with psoriasis or psoriatic arthritis.8 Psoriasis has apparent physical and psychosocial impacts on patients that can be prevented and improved through the exercise recommendations presented in this article. Dermatologists should use these recommendations to address psoriasis in their everyday practice.
There is a direct link between psoriasis and metabolic conditions such as diabetes mellitus and obesity.1 Exercise of varied intensity in patients with chronic inflammatory and metabolic conditions can help improve quality of life and severity of disease; however, there has not been a clear consensus on the recommended duration and types of exercise that are most advantageous.1-5 We reviewed the literature to identify physical and mental health impacts of exercise on patients with psoriasis, and we present the recommended duration and types of exercise that are most impactful for these patients.
One indicator of the link between psoriasis and exercise is the level of peroxisome proliferator activated receptor gamma coactivator-1 α (PGC-1α) in muscle cells.2 This marker reduces inflammation. When levels are low in muscle cells, an induction occurs that leads to systemic or local inflammation; however, skeletal muscle PGC-1α levels increase following exercise, indicating reduced inflammation.2 The level of PGC-1α is measured through muscle biopsy and polymerase chain reaction.6 Another indicator of the correlation between exercise and inflammation is lipoprotein-associated phospholipase A2, which is produced by inflammatory cells and has a correlation with cardiovascular disease. Exercise reduces lipoprotein-associated phospholipase A2 levels, and a sedentary lifestyle correlates with increased levels of this marker.3 Lipoprotein-associated phospholipase A2 is measured through an enzyme-linked immunosorbent assay of the blood, with levels around 200 ng/mL considered high.7 Patients with psoriasis are 30% less likely to participate in physical activity compared to patients without psoriasis, which can be attributed to psychosocial impairment and other factors. Sedentary lifestyle is associated with new or worsening metabolic disease and prevalence of psoriatic lesions.1
A metabolic equivalent task score is a classification system that measures the rate of the body’s oxygen uptake for any given activity.4 A score of 20.9 or more metabolic equivalent task hours of vigorous exercise per week—equal to 105 minutes of running or 180 minutes of swimming or playing tennis—is linked with a 25% to 30% risk reduction of psoriasis in women.4 Therefore, we recommend 30 minutes of exercise 4 to 5 times per week for women. These periods of exercise should consist mainly of activities that will not cause psoriasis flares due to excessive sweating, skin trauma, or prolonged sun exposure.5 Walking, yoga, and bike riding all could be good exercise options for those with psoriasis. The National Psoriasis Foundation offers guidance on physical activity in patients with psoriasis or psoriatic arthritis.8 Psoriasis has apparent physical and psychosocial impacts on patients that can be prevented and improved through the exercise recommendations presented in this article. Dermatologists should use these recommendations to address psoriasis in their everyday practice.
- Do YK, Lakhani N, Malhotra R, et al. Association between psoriasis and leisure-time physical activity: findings from the National Health and Nutrition Examination Survey. J Dermatol. 2015;42:148-153. doi:10.1111/1346-8138.12721
- Handschin C, Spiegelman BM. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature. 2008;454: 463-469. doi:10.1038/nature07206
- Clark K, Sharp S, Womack CJ, et al. Increased sedentary time and decreased physical activity increases lipoprotein associated phospholipase A2 in obese individuals. Nutr Metab Cardiovasc Dis. 2022;32:1703-1710. doi:10.1016/j.numecd.2022.04.023
- Yeh C, Flatley E, Elkattawy O, et al. Exercise in dermatology: exercise’s influence on skin aging, skin cancer, psoriasis, venous ulcers, and androgenetic alopecia. J Am Acad Dermatol. 2022;87:183-184. doi:10.1016/j.jaad.2021.07.023
- Sheppard R, Gan WK, Onambele-Pearson GL, et al. Developing an aerobic exercise intervention for patients with psoriasis to support lifestyle behaviour change and improve health outcomes. Clin Exp Dermatol. 2023;48:5-11. doi:10.1093/ced/llac008
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904.023
- National Psoriasis Foundation. Active and mindful lifestyles. https://www.psoriasis.org/active-and-mindful-lifestyles/
- Do YK, Lakhani N, Malhotra R, et al. Association between psoriasis and leisure-time physical activity: findings from the National Health and Nutrition Examination Survey. J Dermatol. 2015;42:148-153. doi:10.1111/1346-8138.12721
- Handschin C, Spiegelman BM. The role of exercise and PGC1alpha in inflammation and chronic disease. Nature. 2008;454: 463-469. doi:10.1038/nature07206
- Clark K, Sharp S, Womack CJ, et al. Increased sedentary time and decreased physical activity increases lipoprotein associated phospholipase A2 in obese individuals. Nutr Metab Cardiovasc Dis. 2022;32:1703-1710. doi:10.1016/j.numecd.2022.04.023
- Yeh C, Flatley E, Elkattawy O, et al. Exercise in dermatology: exercise’s influence on skin aging, skin cancer, psoriasis, venous ulcers, and androgenetic alopecia. J Am Acad Dermatol. 2022;87:183-184. doi:10.1016/j.jaad.2021.07.023
- Sheppard R, Gan WK, Onambele-Pearson GL, et al. Developing an aerobic exercise intervention for patients with psoriasis to support lifestyle behaviour change and improve health outcomes. Clin Exp Dermatol. 2023;48:5-11. doi:10.1093/ced/llac008
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904
- Lin J, Wu H, Tarr P, et al. Transcriptional co-activator PGC-1a drives the formation of slow-twitch muscle fibres. Nature. 2002;418:797-801. doi:10.1038/nature00904.023
- National Psoriasis Foundation. Active and mindful lifestyles. https://www.psoriasis.org/active-and-mindful-lifestyles/
Optimal Exercise Levels for Dermatology Patients With Psoriasis
Optimal Exercise Levels for Dermatology Patients With Psoriasis
PRACTICE POINTS
- Patients with psoriasis should exercise for less time (~30 min) more frequently (4–5 times per week).
- Exercise that involves excessive sweating should be avoided; recommended types of exercise for patients with psoriasis include walking, yoga, and bike riding.
- Physicians should educate patients on the processes behind psoriasis and direct them to the National Psoriasis Foundation’s website when needed.
Geriatric Dermatology: Q&A With Daniel C. Butler, MD
Daniel C. Butler, MD, is associate professor of dermatology and director of the new Inflammatory and Aging Skin Research Program in the Division of Dermatology at the University of Arizona College of Medicine, Tucson, Arizona. Before returning to Arizona, where he had attended medical school, Butler practiced and was a researcher at the University of California, San Francisco, and its geriatric dermatology clinic. He is a co-founder and continues to co-lead the American Academy of Dermatology (AAD) Geriatric Dermatology Expert Resource Group (ERG).
Butler’s interest in geriatric dermatology is rooted in his experience growing up with four grandparents and witnessing their wisdom, relationships, moments with loved ones, and other unique and desirable parts of growing old. “When I looked later at how aging was perceived in dermatology, I found it was a lot about ‘antiaging,’” he told this news organization. “I thought there was a needed voice in dermatology for healthy aging, for all the desirable things that only growing old can provide, along with all the incredible ‘antiaging’ things we can do.”
In interviews, Butler spoke about research priorities in geriatric dermatology, how the “4M” model of geriatrics should be applied within dermatology, how dermatologists can best work with older complex patients, and more. The conversation was edited for clarity and length.
What is geriatric dermatology? It is described by the AAD’s Geriatric Dermatology ERG as “an emerging subspecialty.” Yet it’s also viewed more broadly. Please speak about its various identities and meanings and its importance for dermatology.
If you’re a Mohs surgeon, you’re seeing a strong majority of over 65 patients. And in various specialty clinics, such as inflammatory skin disease, geriatric dermatology pertains to you. In many ways, it can be viewed as a mindset.
From a framework standpoint, and as a field, geriatric dermatology is a basic science initiative, a clinical initiative, an educational initiative, and an advocacy initiative. The goal is to be able to influence, grow, and learn in each of these categories for our older patients. This is happening: Research in this field has progressed, and education has progressed, which has driven some progress in clinical care.
How has research progressed in the basic science of aging skin? What are key questions for dermatology?
There has been a lot of basic science research on aging skin and on how an aging immune system, for instance, is reflected in conditions such as bullous pemphigoid, atopic dermatitis (AD), and chronic itch. But aging involves more than immunosenescence. I think of aging skin as a three-headed monster that involves changes in the skin barrier and the microbiome as well. But is there a primary piece of aging in the skin? What comes first or influences the other? More research on these questions can potentially influence our treatments.
With respect to the immune system, what we’re finding in the skin is that age-related change is not a decline in the immune system per se, but rather aberrance in response. Parts of the system tend to become overactive, with a skew toward overexpression of type 2 inflammation. This can be problematic, driving conditions such as chronic itch.
With respect to the skin barrier, we lose essential fatty acids, and we lose a lot of our recovery ability and our ability to respond quickly to environmental stressors. But are barrier changes triggering the immune system? Or is it the other way around?
The microbiome, which is a big focus of research, involves similar chicken-and-egg discussions. Is it the microbiome that changes and alters the barrier, which then entices the immune system? Which one happens first? We have a lot to learn, and there’s probably not one answer for every patient.
Please speak about research more broadly. What questions and issues need to be answered and addressed to improve the dermatologic care of older adults?
In general, research in dermatology is very disease-specific and not particularly conducive to looking at the larger demographic populations. We have a huge opportunity, therefore, to break the mold and grow geriatric dermatology as an area of population-based research — so that geriatric dermatology research encompasses not only the melanoma researcher who’s trying to understand how aging influences the melanocytes but also the epidemiologic researcher looking at how our diagnoses and coding and prescription practices are different in the 65-plus age group.
Clinically speaking, researchers want to better understand how aging influences the clinical presentations of our diseases. And there’s research to be done on best practices. For example, what are the best practices for treating basal cell carcinomas in patients with mild cognitive impairment? How should we consider the use of topicals in a patient who has severe arthritis or who lives alone? And then how should we teach practical approaches to help providers meet people where they are?
Looking at it from a healthcare system standpoint, there are many care delivery and access issues — practical pieces — to research, and we’re getting a lot better with this. We’re also advocating not only for more inclusion of older adults in clinical trials of treatments but also for the use of evaluations and outcomes that are relevant and important for older adults.
One piece of good news is that we’re seeing safer treatment options with tremendous efficacy that target known pathways for diseases like AD and chronic itch that affect older adults. Again, now we must find ways to improve access to these novel, safe options.
Our research program at the University of Arizona College of Medicine, which we’re just getting off the ground, aims to be dual-sided, looking both at the basic science of aging skin and at access and care delivery issues, such as how to ensure that patients on Medicare have access to medications that are at least on par with others with private insurance.
What are the most common dermatologic problems experienced by older adults?
Based on my experience and on research that we expect to be published soon, it’s absolutely nonmelanoma skin cancers, precancers like actinic keratoses — and on the inflammatory disease side, itch, AD, and psoriasis. Of course, also common are the age-related changes to the skin that we put in the benign category, such as solar lentigines.
How does age influence dermatologic diseases from a pathophysiological and clinical standpoint?
Diseases overall are very similar and respond to the same treatments, but age in and of itself does influence little pieces. For example, there is more crossover in the presentation of psoriasis and AD in older adults, leading to delays in the diagnosis of psoriasis.
With AD, we’ve found that itch is the predominant symptom for older adults rather than the red rash. We see higher or more severe itch scores in older adults with AD with less visual changes on the skin than in younger cohorts. And rash occurs in different locations than in young patients. Older adults typically present with it on their chest, back, and across the trunk, rather than in folded areas. They’re also more likely to get it on their legs in a nummular pattern as opposed to the more traditional flexural area presentation.
What unique considerations need to be made in treating older adults? How should the 4M model of geriatrics be applied to dermatologic care?
Our care model pushes us to be very algorithmic, but at the end of the day, what’s really important are the 4Ms: Mobility, medication, mentation, and “what matters most.” As you’re having your shared decision-making conversations with your patients and their families, these should be your priorities.
A patient with physical limitations, for instance, may not be able to apply a topical cream twice a day all over the body. They may have comorbidities and treatments for these comorbidities that may conflict with medications you’re considering.
And then mentation is so important. For a long time, we used antihistamines for older adults, but this has been proven to be bad for their mentation and risky in other ways. We need to be sure we’re prioritizing their ability to be clear mentally when we’re prescribing medications and even when we’re considering surgical approaches. Do they show capacity for that procedure or treatment, and how will they respond to that treatment later on?
Using the 4M model to drive conversations is a way to get all of us to connect to the patient and learn about what’s most important for them. In many ways, geriatrics is about taking a step back from your specialist skills and thinking about how you would want a family member treated.
We want to avoid treating just the lesion or the pathologic diagnosis. We want to avoid the “conveyor belt” from a biopsy to Mohs. I have 95-year-olds who say, “Heck yeah, if Mohs is the best treatment, that’s what I want.” And I have 70-year-olds who say, “I think I’ll go with another option,” and that’s the right decision for them. It’s having the conversation that matters.
In practice, given time constraints and other confines, how can dermatologists best work with more complex older patients? What are your practical tips?
People talk about having 45-minute “golden year” conversations with their older patients, but it doesn’t have to be this way. In pursuing geriatric dermatology, I decided early on that I wanted to make sure it was practical, so I’ve focused on maximizing shorter visits and on embracing the concept that relationships can be developed over time. Each time we meet with someone, we’re building equity to have bigger conversations later on.
I can have a 15-minute conversation about whether my patient may want to have Mohs surgery, for instance, or escalate treatment to a systemic agent for their chronic inflammatory disease. If that time isn’t enough, I can encourage further thought about treatment options, acknowledge that decisions aren’t necessarily easy, and schedule a follow-up or offer to call the patient after clinic to continue the conversation.
Sometimes, when I’m at an impasse and my patient is unsure how to proceed, I’ll use clear metrics relevant to older adults — sleep, activity level, and caregiver burden — to help my patient. If someone is not sleeping because of their lesion — if they’re so itchy or their inflammatory disease is uncontrolled, for instance — I’ll point out that the side effects of not sleeping are worse than the medications or surgery we’d pursue. If someone removes themselves from an activity due to their skin condition, that’s a red flag. And if the caregiver in the room is overwhelmed or frustrated by having to put cream on twice a day, I’ll use this to advance treatment.
What resources are available for dermatologists interested in improving their geriatric dermatology skills or advancing the area?
For those interested in investigating these issues or improving their practices, the AAD’s Geriatric Dermatology ERG is always welcoming of new members. The ERG will have an all-inclusive meeting at the 2025 annual AAD meeting in March.
The AAD also has educational modules on geriatric dermatology that were recently published as an initiative of our ERG. More information is available on the website. Also valuable is the ElderDerm conference hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC; the second such conference takes place in May 2025.
Butler reported that he had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
Daniel C. Butler, MD, is associate professor of dermatology and director of the new Inflammatory and Aging Skin Research Program in the Division of Dermatology at the University of Arizona College of Medicine, Tucson, Arizona. Before returning to Arizona, where he had attended medical school, Butler practiced and was a researcher at the University of California, San Francisco, and its geriatric dermatology clinic. He is a co-founder and continues to co-lead the American Academy of Dermatology (AAD) Geriatric Dermatology Expert Resource Group (ERG).
Butler’s interest in geriatric dermatology is rooted in his experience growing up with four grandparents and witnessing their wisdom, relationships, moments with loved ones, and other unique and desirable parts of growing old. “When I looked later at how aging was perceived in dermatology, I found it was a lot about ‘antiaging,’” he told this news organization. “I thought there was a needed voice in dermatology for healthy aging, for all the desirable things that only growing old can provide, along with all the incredible ‘antiaging’ things we can do.”
In interviews, Butler spoke about research priorities in geriatric dermatology, how the “4M” model of geriatrics should be applied within dermatology, how dermatologists can best work with older complex patients, and more. The conversation was edited for clarity and length.
What is geriatric dermatology? It is described by the AAD’s Geriatric Dermatology ERG as “an emerging subspecialty.” Yet it’s also viewed more broadly. Please speak about its various identities and meanings and its importance for dermatology.
If you’re a Mohs surgeon, you’re seeing a strong majority of over 65 patients. And in various specialty clinics, such as inflammatory skin disease, geriatric dermatology pertains to you. In many ways, it can be viewed as a mindset.
From a framework standpoint, and as a field, geriatric dermatology is a basic science initiative, a clinical initiative, an educational initiative, and an advocacy initiative. The goal is to be able to influence, grow, and learn in each of these categories for our older patients. This is happening: Research in this field has progressed, and education has progressed, which has driven some progress in clinical care.
How has research progressed in the basic science of aging skin? What are key questions for dermatology?
There has been a lot of basic science research on aging skin and on how an aging immune system, for instance, is reflected in conditions such as bullous pemphigoid, atopic dermatitis (AD), and chronic itch. But aging involves more than immunosenescence. I think of aging skin as a three-headed monster that involves changes in the skin barrier and the microbiome as well. But is there a primary piece of aging in the skin? What comes first or influences the other? More research on these questions can potentially influence our treatments.
With respect to the immune system, what we’re finding in the skin is that age-related change is not a decline in the immune system per se, but rather aberrance in response. Parts of the system tend to become overactive, with a skew toward overexpression of type 2 inflammation. This can be problematic, driving conditions such as chronic itch.
With respect to the skin barrier, we lose essential fatty acids, and we lose a lot of our recovery ability and our ability to respond quickly to environmental stressors. But are barrier changes triggering the immune system? Or is it the other way around?
The microbiome, which is a big focus of research, involves similar chicken-and-egg discussions. Is it the microbiome that changes and alters the barrier, which then entices the immune system? Which one happens first? We have a lot to learn, and there’s probably not one answer for every patient.
Please speak about research more broadly. What questions and issues need to be answered and addressed to improve the dermatologic care of older adults?
In general, research in dermatology is very disease-specific and not particularly conducive to looking at the larger demographic populations. We have a huge opportunity, therefore, to break the mold and grow geriatric dermatology as an area of population-based research — so that geriatric dermatology research encompasses not only the melanoma researcher who’s trying to understand how aging influences the melanocytes but also the epidemiologic researcher looking at how our diagnoses and coding and prescription practices are different in the 65-plus age group.
Clinically speaking, researchers want to better understand how aging influences the clinical presentations of our diseases. And there’s research to be done on best practices. For example, what are the best practices for treating basal cell carcinomas in patients with mild cognitive impairment? How should we consider the use of topicals in a patient who has severe arthritis or who lives alone? And then how should we teach practical approaches to help providers meet people where they are?
Looking at it from a healthcare system standpoint, there are many care delivery and access issues — practical pieces — to research, and we’re getting a lot better with this. We’re also advocating not only for more inclusion of older adults in clinical trials of treatments but also for the use of evaluations and outcomes that are relevant and important for older adults.
One piece of good news is that we’re seeing safer treatment options with tremendous efficacy that target known pathways for diseases like AD and chronic itch that affect older adults. Again, now we must find ways to improve access to these novel, safe options.
Our research program at the University of Arizona College of Medicine, which we’re just getting off the ground, aims to be dual-sided, looking both at the basic science of aging skin and at access and care delivery issues, such as how to ensure that patients on Medicare have access to medications that are at least on par with others with private insurance.
What are the most common dermatologic problems experienced by older adults?
Based on my experience and on research that we expect to be published soon, it’s absolutely nonmelanoma skin cancers, precancers like actinic keratoses — and on the inflammatory disease side, itch, AD, and psoriasis. Of course, also common are the age-related changes to the skin that we put in the benign category, such as solar lentigines.
How does age influence dermatologic diseases from a pathophysiological and clinical standpoint?
Diseases overall are very similar and respond to the same treatments, but age in and of itself does influence little pieces. For example, there is more crossover in the presentation of psoriasis and AD in older adults, leading to delays in the diagnosis of psoriasis.
With AD, we’ve found that itch is the predominant symptom for older adults rather than the red rash. We see higher or more severe itch scores in older adults with AD with less visual changes on the skin than in younger cohorts. And rash occurs in different locations than in young patients. Older adults typically present with it on their chest, back, and across the trunk, rather than in folded areas. They’re also more likely to get it on their legs in a nummular pattern as opposed to the more traditional flexural area presentation.
What unique considerations need to be made in treating older adults? How should the 4M model of geriatrics be applied to dermatologic care?
Our care model pushes us to be very algorithmic, but at the end of the day, what’s really important are the 4Ms: Mobility, medication, mentation, and “what matters most.” As you’re having your shared decision-making conversations with your patients and their families, these should be your priorities.
A patient with physical limitations, for instance, may not be able to apply a topical cream twice a day all over the body. They may have comorbidities and treatments for these comorbidities that may conflict with medications you’re considering.
And then mentation is so important. For a long time, we used antihistamines for older adults, but this has been proven to be bad for their mentation and risky in other ways. We need to be sure we’re prioritizing their ability to be clear mentally when we’re prescribing medications and even when we’re considering surgical approaches. Do they show capacity for that procedure or treatment, and how will they respond to that treatment later on?
Using the 4M model to drive conversations is a way to get all of us to connect to the patient and learn about what’s most important for them. In many ways, geriatrics is about taking a step back from your specialist skills and thinking about how you would want a family member treated.
We want to avoid treating just the lesion or the pathologic diagnosis. We want to avoid the “conveyor belt” from a biopsy to Mohs. I have 95-year-olds who say, “Heck yeah, if Mohs is the best treatment, that’s what I want.” And I have 70-year-olds who say, “I think I’ll go with another option,” and that’s the right decision for them. It’s having the conversation that matters.
In practice, given time constraints and other confines, how can dermatologists best work with more complex older patients? What are your practical tips?
People talk about having 45-minute “golden year” conversations with their older patients, but it doesn’t have to be this way. In pursuing geriatric dermatology, I decided early on that I wanted to make sure it was practical, so I’ve focused on maximizing shorter visits and on embracing the concept that relationships can be developed over time. Each time we meet with someone, we’re building equity to have bigger conversations later on.
I can have a 15-minute conversation about whether my patient may want to have Mohs surgery, for instance, or escalate treatment to a systemic agent for their chronic inflammatory disease. If that time isn’t enough, I can encourage further thought about treatment options, acknowledge that decisions aren’t necessarily easy, and schedule a follow-up or offer to call the patient after clinic to continue the conversation.
Sometimes, when I’m at an impasse and my patient is unsure how to proceed, I’ll use clear metrics relevant to older adults — sleep, activity level, and caregiver burden — to help my patient. If someone is not sleeping because of their lesion — if they’re so itchy or their inflammatory disease is uncontrolled, for instance — I’ll point out that the side effects of not sleeping are worse than the medications or surgery we’d pursue. If someone removes themselves from an activity due to their skin condition, that’s a red flag. And if the caregiver in the room is overwhelmed or frustrated by having to put cream on twice a day, I’ll use this to advance treatment.
What resources are available for dermatologists interested in improving their geriatric dermatology skills or advancing the area?
For those interested in investigating these issues or improving their practices, the AAD’s Geriatric Dermatology ERG is always welcoming of new members. The ERG will have an all-inclusive meeting at the 2025 annual AAD meeting in March.
The AAD also has educational modules on geriatric dermatology that were recently published as an initiative of our ERG. More information is available on the website. Also valuable is the ElderDerm conference hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC; the second such conference takes place in May 2025.
Butler reported that he had no relevant financial disclosures.
A version of this article appeared on Medscape.com.
Daniel C. Butler, MD, is associate professor of dermatology and director of the new Inflammatory and Aging Skin Research Program in the Division of Dermatology at the University of Arizona College of Medicine, Tucson, Arizona. Before returning to Arizona, where he had attended medical school, Butler practiced and was a researcher at the University of California, San Francisco, and its geriatric dermatology clinic. He is a co-founder and continues to co-lead the American Academy of Dermatology (AAD) Geriatric Dermatology Expert Resource Group (ERG).
Butler’s interest in geriatric dermatology is rooted in his experience growing up with four grandparents and witnessing their wisdom, relationships, moments with loved ones, and other unique and desirable parts of growing old. “When I looked later at how aging was perceived in dermatology, I found it was a lot about ‘antiaging,’” he told this news organization. “I thought there was a needed voice in dermatology for healthy aging, for all the desirable things that only growing old can provide, along with all the incredible ‘antiaging’ things we can do.”
In interviews, Butler spoke about research priorities in geriatric dermatology, how the “4M” model of geriatrics should be applied within dermatology, how dermatologists can best work with older complex patients, and more. The conversation was edited for clarity and length.
What is geriatric dermatology? It is described by the AAD’s Geriatric Dermatology ERG as “an emerging subspecialty.” Yet it’s also viewed more broadly. Please speak about its various identities and meanings and its importance for dermatology.
If you’re a Mohs surgeon, you’re seeing a strong majority of over 65 patients. And in various specialty clinics, such as inflammatory skin disease, geriatric dermatology pertains to you. In many ways, it can be viewed as a mindset.
From a framework standpoint, and as a field, geriatric dermatology is a basic science initiative, a clinical initiative, an educational initiative, and an advocacy initiative. The goal is to be able to influence, grow, and learn in each of these categories for our older patients. This is happening: Research in this field has progressed, and education has progressed, which has driven some progress in clinical care.
How has research progressed in the basic science of aging skin? What are key questions for dermatology?
There has been a lot of basic science research on aging skin and on how an aging immune system, for instance, is reflected in conditions such as bullous pemphigoid, atopic dermatitis (AD), and chronic itch. But aging involves more than immunosenescence. I think of aging skin as a three-headed monster that involves changes in the skin barrier and the microbiome as well. But is there a primary piece of aging in the skin? What comes first or influences the other? More research on these questions can potentially influence our treatments.
With respect to the immune system, what we’re finding in the skin is that age-related change is not a decline in the immune system per se, but rather aberrance in response. Parts of the system tend to become overactive, with a skew toward overexpression of type 2 inflammation. This can be problematic, driving conditions such as chronic itch.
With respect to the skin barrier, we lose essential fatty acids, and we lose a lot of our recovery ability and our ability to respond quickly to environmental stressors. But are barrier changes triggering the immune system? Or is it the other way around?
The microbiome, which is a big focus of research, involves similar chicken-and-egg discussions. Is it the microbiome that changes and alters the barrier, which then entices the immune system? Which one happens first? We have a lot to learn, and there’s probably not one answer for every patient.
Please speak about research more broadly. What questions and issues need to be answered and addressed to improve the dermatologic care of older adults?
In general, research in dermatology is very disease-specific and not particularly conducive to looking at the larger demographic populations. We have a huge opportunity, therefore, to break the mold and grow geriatric dermatology as an area of population-based research — so that geriatric dermatology research encompasses not only the melanoma researcher who’s trying to understand how aging influences the melanocytes but also the epidemiologic researcher looking at how our diagnoses and coding and prescription practices are different in the 65-plus age group.
Clinically speaking, researchers want to better understand how aging influences the clinical presentations of our diseases. And there’s research to be done on best practices. For example, what are the best practices for treating basal cell carcinomas in patients with mild cognitive impairment? How should we consider the use of topicals in a patient who has severe arthritis or who lives alone? And then how should we teach practical approaches to help providers meet people where they are?
Looking at it from a healthcare system standpoint, there are many care delivery and access issues — practical pieces — to research, and we’re getting a lot better with this. We’re also advocating not only for more inclusion of older adults in clinical trials of treatments but also for the use of evaluations and outcomes that are relevant and important for older adults.
One piece of good news is that we’re seeing safer treatment options with tremendous efficacy that target known pathways for diseases like AD and chronic itch that affect older adults. Again, now we must find ways to improve access to these novel, safe options.
Our research program at the University of Arizona College of Medicine, which we’re just getting off the ground, aims to be dual-sided, looking both at the basic science of aging skin and at access and care delivery issues, such as how to ensure that patients on Medicare have access to medications that are at least on par with others with private insurance.
What are the most common dermatologic problems experienced by older adults?
Based on my experience and on research that we expect to be published soon, it’s absolutely nonmelanoma skin cancers, precancers like actinic keratoses — and on the inflammatory disease side, itch, AD, and psoriasis. Of course, also common are the age-related changes to the skin that we put in the benign category, such as solar lentigines.
How does age influence dermatologic diseases from a pathophysiological and clinical standpoint?
Diseases overall are very similar and respond to the same treatments, but age in and of itself does influence little pieces. For example, there is more crossover in the presentation of psoriasis and AD in older adults, leading to delays in the diagnosis of psoriasis.
With AD, we’ve found that itch is the predominant symptom for older adults rather than the red rash. We see higher or more severe itch scores in older adults with AD with less visual changes on the skin than in younger cohorts. And rash occurs in different locations than in young patients. Older adults typically present with it on their chest, back, and across the trunk, rather than in folded areas. They’re also more likely to get it on their legs in a nummular pattern as opposed to the more traditional flexural area presentation.
What unique considerations need to be made in treating older adults? How should the 4M model of geriatrics be applied to dermatologic care?
Our care model pushes us to be very algorithmic, but at the end of the day, what’s really important are the 4Ms: Mobility, medication, mentation, and “what matters most.” As you’re having your shared decision-making conversations with your patients and their families, these should be your priorities.
A patient with physical limitations, for instance, may not be able to apply a topical cream twice a day all over the body. They may have comorbidities and treatments for these comorbidities that may conflict with medications you’re considering.
And then mentation is so important. For a long time, we used antihistamines for older adults, but this has been proven to be bad for their mentation and risky in other ways. We need to be sure we’re prioritizing their ability to be clear mentally when we’re prescribing medications and even when we’re considering surgical approaches. Do they show capacity for that procedure or treatment, and how will they respond to that treatment later on?
Using the 4M model to drive conversations is a way to get all of us to connect to the patient and learn about what’s most important for them. In many ways, geriatrics is about taking a step back from your specialist skills and thinking about how you would want a family member treated.
We want to avoid treating just the lesion or the pathologic diagnosis. We want to avoid the “conveyor belt” from a biopsy to Mohs. I have 95-year-olds who say, “Heck yeah, if Mohs is the best treatment, that’s what I want.” And I have 70-year-olds who say, “I think I’ll go with another option,” and that’s the right decision for them. It’s having the conversation that matters.
In practice, given time constraints and other confines, how can dermatologists best work with more complex older patients? What are your practical tips?
People talk about having 45-minute “golden year” conversations with their older patients, but it doesn’t have to be this way. In pursuing geriatric dermatology, I decided early on that I wanted to make sure it was practical, so I’ve focused on maximizing shorter visits and on embracing the concept that relationships can be developed over time. Each time we meet with someone, we’re building equity to have bigger conversations later on.
I can have a 15-minute conversation about whether my patient may want to have Mohs surgery, for instance, or escalate treatment to a systemic agent for their chronic inflammatory disease. If that time isn’t enough, I can encourage further thought about treatment options, acknowledge that decisions aren’t necessarily easy, and schedule a follow-up or offer to call the patient after clinic to continue the conversation.
Sometimes, when I’m at an impasse and my patient is unsure how to proceed, I’ll use clear metrics relevant to older adults — sleep, activity level, and caregiver burden — to help my patient. If someone is not sleeping because of their lesion — if they’re so itchy or their inflammatory disease is uncontrolled, for instance — I’ll point out that the side effects of not sleeping are worse than the medications or surgery we’d pursue. If someone removes themselves from an activity due to their skin condition, that’s a red flag. And if the caregiver in the room is overwhelmed or frustrated by having to put cream on twice a day, I’ll use this to advance treatment.
What resources are available for dermatologists interested in improving their geriatric dermatology skills or advancing the area?
For those interested in investigating these issues or improving their practices, the AAD’s Geriatric Dermatology ERG is always welcoming of new members. The ERG will have an all-inclusive meeting at the 2025 annual AAD meeting in March.
The AAD also has educational modules on geriatric dermatology that were recently published as an initiative of our ERG. More information is available on the website. Also valuable is the ElderDerm conference hosted by the George Washington University School of Medicine and Health Sciences, Washington, DC; the second such conference takes place in May 2025.
Butler reported that he had no relevant financial disclosures.
A version of this article appeared on Medscape.com.

