User login
Immediate or delayed pushing in the second stage of labor?
ILLUSTRATIVE CASE
A 27-year-old G1P000 at term with an uncomplicated pregnancy has been laboring for 6 hours with an epidural in place and a reassuring fetal heart tracing. She is at –2 station with complete cervical dilation and effacement. Should she push now or delay pushing to allow for more descent?
More than 10,000 women give birth each day in the United States, yet few of our approaches to labor management are evidence based.2 For example, there are no clear guidelines on whether immediate pushing or delayed pushing (waiting 1-2 hours) in the second stage of labor (the time from complete cervical dilation to delivery of the fetus) leads to better outcomes.
A recent Cochrane review, which included very low- to moderate-quality trials of nulliparous and multiparous women using epidural analgesia showed that delayed pushing resulted in more vaginal deliveries, longer duration of second stage of labor, and shorter duration of pushing.3 But many of the trials included in this Cochrane review were noted to have study design limitations and significant heterogeneity.
A recent retrospective study found that delayed pushing resulted in longer duration of pushing and increased risks for cesarean section, operative vaginal delivery, and postpartum hemorrhage in nulliparous patients with and without epidurals.4 The World Health Organization recommends delayed pushing in women with epidural analgesia if time and fetal monitoring resources are available.5
STUDY SUMMARY
Does the timing of second stage pushing efforts affect outcomes?
This multicenter randomized controlled trial (RCT) evaluated the effect on spontaneous vaginal delivery of delayed pushing vs immediate pushing in 2404 term nulliparous women using epidural analgesia.1 Patients were ≥ 37 weeks’ gestation. Once patients achieved 10 cm of cervical dilation, they were randomized in a 1:1 ratio to either immediate pushing or to delayed (for 60 minutes) pushing (unless there was an irresistible urge to push or they were otherwise instructed by their provider).
Outcome and results. The primary outcome was spontaneous vaginal delivery without the use of any operative support. The mean time to pushing after complete cervical dilation was 19 minutes in the immediate pushing group and 60 minutes in the delayed group. There was no difference in the rate of spontaneous vaginal delivery between the immediate and delayed pushing groups (86% vs 87%, respectively; P = .67). The immediate pushing group had a shorter duration of second stage of labor (102 minutes vs 134 minutes; mean difference [MD] = –32 minutes; 95% confidence interval [CI], –37 to –27; P < .001) and a slightly longer duration of active pushing (84 minutes vs 75 minutes; MD = 9.2 minutes; 95% CI, 6-13; P < .001).
There was no significant difference in operative vaginal or cesarean deliveries. Postpartum hemorrhage was lower in the immediate pushing group (2.3% vs 4%; risk ratio [RR] = 0.6; 95% CI, 0.3-0.9; P = .03; number needed to treat [NNT] = 58), as was chorioamnionitis (6.7% vs 9.1%; RR = 0.7; 95% CI, 0.66-0.90; P = .005; NNT = 40). There was no significant difference in neonatal morbidity between groups. And in subgroup analysis, there was no significant difference in rates of vaginal delivery based on fetal position (occiput anterior, posterior, or transverse) or station (defined as high [< 2 cm] or low [≥ 2 cm]) between groups. Recruitment was stopped early at 75% because there was no difference in the primary outcome and there was concern regarding an increased risk of hemorrhage in the delayed pushing group.
Continue to: WHAT'S NEW
WHAT’S NEW
There’s no good reason to delay pushing
Delaying pushing once the cervix is completely dilated is not indicated, even for nulliparous women receiving epidural analgesia, as it does not decrease the rate of spontaneous vaginal delivery. It does, however, increase the length of second stage labor and the risk of postpartum hemorrhage and chorioamnionitis.
CAVEATS
Study was stopped early, and groups were unblinded
This study was stopped early, so it is not known if it was underpowered for some of the secondary outcomes. Also, it was not possible to blind the groups, so it is not clear if any bias in patient management or diagnosis resulted.
CHALLENGES TO IMPLEMENTATION
Will current practice and culture pose obstacles?
Although the overt challenges to enacting a policy of immediate pushing are minimal, the inertia of current practice and culture could affect the implementation of this strategy.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Cahill AG, Srinivas SK, Tita ATN, et al. Effect of immediate vs delayed pushing on rates of spontaneous vaginal delivery among nulliparous women receiving neuraxial analgesia: a randomized clinical trial. JAMA. 2018;320:1444-1454.
2. Hamilton BE, Martin JA, Osterman MJK, et al. Births: provisional data for 2018. Vital Statistics Rapid Release. May 2019; Report No. 007. www.cdc.gov/nchs/data/vsrr/vsrr-007-508.pdf. Accessed April 22, 2020.
3. Lemos A, Amorim MM, Domales de Andrade A, et al. Pushing/bearing down methods for the second stage of labour. Cochrane Database Syst Rev. 2017;3:CD009124.
4. Yee LM, Sandoval G, Bailit J, et al. Maternal and neonatal outcomes with early compared with delayed pushing among nulliparous women. Obstet Gynecol. 2016;128:1039-1047.
5. WHO recommendations: intrapartum care for a positive childbirth experience. Geneva: World Health Organization; 2018. www.who.int/reproductivehealth/publications/intrapartum-care-guidelines/en/. Accessed April 22, 2020.
ILLUSTRATIVE CASE
A 27-year-old G1P000 at term with an uncomplicated pregnancy has been laboring for 6 hours with an epidural in place and a reassuring fetal heart tracing. She is at –2 station with complete cervical dilation and effacement. Should she push now or delay pushing to allow for more descent?
More than 10,000 women give birth each day in the United States, yet few of our approaches to labor management are evidence based.2 For example, there are no clear guidelines on whether immediate pushing or delayed pushing (waiting 1-2 hours) in the second stage of labor (the time from complete cervical dilation to delivery of the fetus) leads to better outcomes.
A recent Cochrane review, which included very low- to moderate-quality trials of nulliparous and multiparous women using epidural analgesia showed that delayed pushing resulted in more vaginal deliveries, longer duration of second stage of labor, and shorter duration of pushing.3 But many of the trials included in this Cochrane review were noted to have study design limitations and significant heterogeneity.
A recent retrospective study found that delayed pushing resulted in longer duration of pushing and increased risks for cesarean section, operative vaginal delivery, and postpartum hemorrhage in nulliparous patients with and without epidurals.4 The World Health Organization recommends delayed pushing in women with epidural analgesia if time and fetal monitoring resources are available.5
STUDY SUMMARY
Does the timing of second stage pushing efforts affect outcomes?
This multicenter randomized controlled trial (RCT) evaluated the effect on spontaneous vaginal delivery of delayed pushing vs immediate pushing in 2404 term nulliparous women using epidural analgesia.1 Patients were ≥ 37 weeks’ gestation. Once patients achieved 10 cm of cervical dilation, they were randomized in a 1:1 ratio to either immediate pushing or to delayed (for 60 minutes) pushing (unless there was an irresistible urge to push or they were otherwise instructed by their provider).
Outcome and results. The primary outcome was spontaneous vaginal delivery without the use of any operative support. The mean time to pushing after complete cervical dilation was 19 minutes in the immediate pushing group and 60 minutes in the delayed group. There was no difference in the rate of spontaneous vaginal delivery between the immediate and delayed pushing groups (86% vs 87%, respectively; P = .67). The immediate pushing group had a shorter duration of second stage of labor (102 minutes vs 134 minutes; mean difference [MD] = –32 minutes; 95% confidence interval [CI], –37 to –27; P < .001) and a slightly longer duration of active pushing (84 minutes vs 75 minutes; MD = 9.2 minutes; 95% CI, 6-13; P < .001).
There was no significant difference in operative vaginal or cesarean deliveries. Postpartum hemorrhage was lower in the immediate pushing group (2.3% vs 4%; risk ratio [RR] = 0.6; 95% CI, 0.3-0.9; P = .03; number needed to treat [NNT] = 58), as was chorioamnionitis (6.7% vs 9.1%; RR = 0.7; 95% CI, 0.66-0.90; P = .005; NNT = 40). There was no significant difference in neonatal morbidity between groups. And in subgroup analysis, there was no significant difference in rates of vaginal delivery based on fetal position (occiput anterior, posterior, or transverse) or station (defined as high [< 2 cm] or low [≥ 2 cm]) between groups. Recruitment was stopped early at 75% because there was no difference in the primary outcome and there was concern regarding an increased risk of hemorrhage in the delayed pushing group.
Continue to: WHAT'S NEW
WHAT’S NEW
There’s no good reason to delay pushing
Delaying pushing once the cervix is completely dilated is not indicated, even for nulliparous women receiving epidural analgesia, as it does not decrease the rate of spontaneous vaginal delivery. It does, however, increase the length of second stage labor and the risk of postpartum hemorrhage and chorioamnionitis.
CAVEATS
Study was stopped early, and groups were unblinded
This study was stopped early, so it is not known if it was underpowered for some of the secondary outcomes. Also, it was not possible to blind the groups, so it is not clear if any bias in patient management or diagnosis resulted.
CHALLENGES TO IMPLEMENTATION
Will current practice and culture pose obstacles?
Although the overt challenges to enacting a policy of immediate pushing are minimal, the inertia of current practice and culture could affect the implementation of this strategy.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 27-year-old G1P000 at term with an uncomplicated pregnancy has been laboring for 6 hours with an epidural in place and a reassuring fetal heart tracing. She is at –2 station with complete cervical dilation and effacement. Should she push now or delay pushing to allow for more descent?
More than 10,000 women give birth each day in the United States, yet few of our approaches to labor management are evidence based.2 For example, there are no clear guidelines on whether immediate pushing or delayed pushing (waiting 1-2 hours) in the second stage of labor (the time from complete cervical dilation to delivery of the fetus) leads to better outcomes.
A recent Cochrane review, which included very low- to moderate-quality trials of nulliparous and multiparous women using epidural analgesia showed that delayed pushing resulted in more vaginal deliveries, longer duration of second stage of labor, and shorter duration of pushing.3 But many of the trials included in this Cochrane review were noted to have study design limitations and significant heterogeneity.
A recent retrospective study found that delayed pushing resulted in longer duration of pushing and increased risks for cesarean section, operative vaginal delivery, and postpartum hemorrhage in nulliparous patients with and without epidurals.4 The World Health Organization recommends delayed pushing in women with epidural analgesia if time and fetal monitoring resources are available.5
STUDY SUMMARY
Does the timing of second stage pushing efforts affect outcomes?
This multicenter randomized controlled trial (RCT) evaluated the effect on spontaneous vaginal delivery of delayed pushing vs immediate pushing in 2404 term nulliparous women using epidural analgesia.1 Patients were ≥ 37 weeks’ gestation. Once patients achieved 10 cm of cervical dilation, they were randomized in a 1:1 ratio to either immediate pushing or to delayed (for 60 minutes) pushing (unless there was an irresistible urge to push or they were otherwise instructed by their provider).
Outcome and results. The primary outcome was spontaneous vaginal delivery without the use of any operative support. The mean time to pushing after complete cervical dilation was 19 minutes in the immediate pushing group and 60 minutes in the delayed group. There was no difference in the rate of spontaneous vaginal delivery between the immediate and delayed pushing groups (86% vs 87%, respectively; P = .67). The immediate pushing group had a shorter duration of second stage of labor (102 minutes vs 134 minutes; mean difference [MD] = –32 minutes; 95% confidence interval [CI], –37 to –27; P < .001) and a slightly longer duration of active pushing (84 minutes vs 75 minutes; MD = 9.2 minutes; 95% CI, 6-13; P < .001).
There was no significant difference in operative vaginal or cesarean deliveries. Postpartum hemorrhage was lower in the immediate pushing group (2.3% vs 4%; risk ratio [RR] = 0.6; 95% CI, 0.3-0.9; P = .03; number needed to treat [NNT] = 58), as was chorioamnionitis (6.7% vs 9.1%; RR = 0.7; 95% CI, 0.66-0.90; P = .005; NNT = 40). There was no significant difference in neonatal morbidity between groups. And in subgroup analysis, there was no significant difference in rates of vaginal delivery based on fetal position (occiput anterior, posterior, or transverse) or station (defined as high [< 2 cm] or low [≥ 2 cm]) between groups. Recruitment was stopped early at 75% because there was no difference in the primary outcome and there was concern regarding an increased risk of hemorrhage in the delayed pushing group.
Continue to: WHAT'S NEW
WHAT’S NEW
There’s no good reason to delay pushing
Delaying pushing once the cervix is completely dilated is not indicated, even for nulliparous women receiving epidural analgesia, as it does not decrease the rate of spontaneous vaginal delivery. It does, however, increase the length of second stage labor and the risk of postpartum hemorrhage and chorioamnionitis.
CAVEATS
Study was stopped early, and groups were unblinded
This study was stopped early, so it is not known if it was underpowered for some of the secondary outcomes. Also, it was not possible to blind the groups, so it is not clear if any bias in patient management or diagnosis resulted.
CHALLENGES TO IMPLEMENTATION
Will current practice and culture pose obstacles?
Although the overt challenges to enacting a policy of immediate pushing are minimal, the inertia of current practice and culture could affect the implementation of this strategy.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Cahill AG, Srinivas SK, Tita ATN, et al. Effect of immediate vs delayed pushing on rates of spontaneous vaginal delivery among nulliparous women receiving neuraxial analgesia: a randomized clinical trial. JAMA. 2018;320:1444-1454.
2. Hamilton BE, Martin JA, Osterman MJK, et al. Births: provisional data for 2018. Vital Statistics Rapid Release. May 2019; Report No. 007. www.cdc.gov/nchs/data/vsrr/vsrr-007-508.pdf. Accessed April 22, 2020.
3. Lemos A, Amorim MM, Domales de Andrade A, et al. Pushing/bearing down methods for the second stage of labour. Cochrane Database Syst Rev. 2017;3:CD009124.
4. Yee LM, Sandoval G, Bailit J, et al. Maternal and neonatal outcomes with early compared with delayed pushing among nulliparous women. Obstet Gynecol. 2016;128:1039-1047.
5. WHO recommendations: intrapartum care for a positive childbirth experience. Geneva: World Health Organization; 2018. www.who.int/reproductivehealth/publications/intrapartum-care-guidelines/en/. Accessed April 22, 2020.
1. Cahill AG, Srinivas SK, Tita ATN, et al. Effect of immediate vs delayed pushing on rates of spontaneous vaginal delivery among nulliparous women receiving neuraxial analgesia: a randomized clinical trial. JAMA. 2018;320:1444-1454.
2. Hamilton BE, Martin JA, Osterman MJK, et al. Births: provisional data for 2018. Vital Statistics Rapid Release. May 2019; Report No. 007. www.cdc.gov/nchs/data/vsrr/vsrr-007-508.pdf. Accessed April 22, 2020.
3. Lemos A, Amorim MM, Domales de Andrade A, et al. Pushing/bearing down methods for the second stage of labour. Cochrane Database Syst Rev. 2017;3:CD009124.
4. Yee LM, Sandoval G, Bailit J, et al. Maternal and neonatal outcomes with early compared with delayed pushing among nulliparous women. Obstet Gynecol. 2016;128:1039-1047.
5. WHO recommendations: intrapartum care for a positive childbirth experience. Geneva: World Health Organization; 2018. www.who.int/reproductivehealth/publications/intrapartum-care-guidelines/en/. Accessed April 22, 2020.
PRACTICE CHANGER
Recommend immediate, rather than delayed, pushing in the second stage of labor for nulliparous women receiving epidural analgesia. The rate of spontaneous vaginal delivery is the same, and there is a lower risk of postpartum hemorrhage and chorioamnionitis.
STRENGTH OF RECOMMENDATION
B: Based on an individual randomized controlled trial. 1
Cahill AG, Srinivas SK, Tita ATN, et al. Effect of immediate vs delayed pushing on rates of spontaneous vaginal delivery among nulliparous women receiving neuraxial analgesia: a randomized clinical trial. JAMA. 2018;320:1444-1454.
Justices appear split over birth control mandate case
U.S. Supreme Court justices appear divided over whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraception mandate.
During oral arguments on May 6, the court expressed differing perspectives about the administration’s authority to allow for more exemptions under the health law’s birth control mandate and whether the expansions were reasonable. Justices heard the consolidated cases – Little Sisters of the Poor v. Pennsylvania and Trump v. Pennsylvania – by teleconference because of the COVID-19 pandemic. They are expected to make a decision by the summer.
Associate justice Ruth Bader Ginsburg, who participated in the telephone conference call from a hospital where she was recovering from a gallbladder condition, said the exemptions ignored the intent of Congress to provide women with comprehensive coverage through the ACA.
“The glaring feature of what the government has done in expanding this exemption is to toss to the winds entirely Congress’s instruction that women need and shall have seamless, no-cost, comprehensive coverage,” she said during oral arguments. “This leaves the women to hunt for other government programs that might cover them, and for those who are not covered by Medicaid or one of the other government programs, they can get contraceptive coverage only from paying out of their own pocket, which is exactly what Congress didn’t want to happen.”
Associate Justice Samuel Alito Jr., meanwhile, indicated that a lower court opinion that had blocked the exemptions from going forward conflicts with the Supreme Court’s ruling in a related case, Burwell v. Hobby Lobby.
“Explain to me why the Third Circuit’s analysis of the question of substantial burden is not squarely inconsistent with our reasoning in Hobby Lobby,” Associate Justice Alito said during oral arguments. “Hobby Lobby held that, if a person sincerely believes that it is immoral to perform an act that has the effect of enabling another person to commit an immoral act, a federal court does not have the right to say that this person is wrong on the question of moral complicity. That’s precisely the situation here. Reading the Third Circuit’s discussion of the substantial burden question, I wondered whether they had read that part of the Hobby Lobby decision.”
The dispute surrounding the ACA’s birth control mandate and the extent of exemptions afforded has gone on for a decade and has led to numerous legal challenges. The ACA initially required all employers to cover birth control for employees with no copayments, but exempted group health plans of religious employers. Those religious employers were primarily churches and other houses of worship. After a number of complaints and lawsuits, the Obama administration created a workaround for nonprofit religious employers not included in that exemption to opt out of the mandate. However, critics argued the process itself was a violation of their religious freedom.
The issue led to the case of Zubik v. Burwell, a legal challenge over the mandate exemption that went before the U.S. Supreme Court in March 2016. The issue was never resolved however, and in May 2016, the Supreme Court vacated the lower court rulings related to Zubik v. Burwell and remanded the case back to the four appeals courts that had originally ruled on the issue.
In 2018, the Trump administration announced new rules aimed at broadening exemptions to the ACA’s contraceptive mandate to entities that object to services covered by the mandate on the basis of “sincerely held religious beliefs.” A second rule allowed nonprofit organizations and small businesses that had nonreligious moral convictions against the mandate to opt out.
Thirteen states and the District of Columbia then sued the Trump administration over the rules, as well as Pennsylvania and New Jersey in a separate case. Little Sisters of the Poor, a religious nonprofit operating a home in Pittsburgh, intervened in the case as an aggrieved party. An appeal court temporarily barred the regulations from moving forward.
During oral arguments, Solicitor General for the Department of Justice Noel J. Francisco said the exemptions are lawful because they are authorized under a provision of the ACA as well as the Religious Freedom Restoration Act (RFRA).
“RFRA at the very least authorizes the religious exemption,” Mr. Francisco said during oral arguments.
Chief Deputy Attorney General for Pennsylvania Michael J. Fischer argued that the Trump administration’s moral and religious exemption rules rest on overly broad assertions of agency authority.
“First, the agencies twist a narrow delegation that allows the Health Resources and Services Administration to decide which preventive services insurers must cover under the Women’s Health Amendment into a grant of authority so broad it allows them to permit virtually any employer or college to opt out of providing contraceptive coverage entirely, including for reasons as amorphous as vaguely defined moral beliefs,” he said during oral arguments. “Second, the agencies claim that RFRA, a statute that limits government action, affirmatively authorizes them to permit employers to deny women their rights to contraceptive coverage even in the absence of a RFRA violation in the first place.”
U.S. Supreme Court justices appear divided over whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraception mandate.
During oral arguments on May 6, the court expressed differing perspectives about the administration’s authority to allow for more exemptions under the health law’s birth control mandate and whether the expansions were reasonable. Justices heard the consolidated cases – Little Sisters of the Poor v. Pennsylvania and Trump v. Pennsylvania – by teleconference because of the COVID-19 pandemic. They are expected to make a decision by the summer.
Associate justice Ruth Bader Ginsburg, who participated in the telephone conference call from a hospital where she was recovering from a gallbladder condition, said the exemptions ignored the intent of Congress to provide women with comprehensive coverage through the ACA.
“The glaring feature of what the government has done in expanding this exemption is to toss to the winds entirely Congress’s instruction that women need and shall have seamless, no-cost, comprehensive coverage,” she said during oral arguments. “This leaves the women to hunt for other government programs that might cover them, and for those who are not covered by Medicaid or one of the other government programs, they can get contraceptive coverage only from paying out of their own pocket, which is exactly what Congress didn’t want to happen.”
Associate Justice Samuel Alito Jr., meanwhile, indicated that a lower court opinion that had blocked the exemptions from going forward conflicts with the Supreme Court’s ruling in a related case, Burwell v. Hobby Lobby.
“Explain to me why the Third Circuit’s analysis of the question of substantial burden is not squarely inconsistent with our reasoning in Hobby Lobby,” Associate Justice Alito said during oral arguments. “Hobby Lobby held that, if a person sincerely believes that it is immoral to perform an act that has the effect of enabling another person to commit an immoral act, a federal court does not have the right to say that this person is wrong on the question of moral complicity. That’s precisely the situation here. Reading the Third Circuit’s discussion of the substantial burden question, I wondered whether they had read that part of the Hobby Lobby decision.”
The dispute surrounding the ACA’s birth control mandate and the extent of exemptions afforded has gone on for a decade and has led to numerous legal challenges. The ACA initially required all employers to cover birth control for employees with no copayments, but exempted group health plans of religious employers. Those religious employers were primarily churches and other houses of worship. After a number of complaints and lawsuits, the Obama administration created a workaround for nonprofit religious employers not included in that exemption to opt out of the mandate. However, critics argued the process itself was a violation of their religious freedom.
The issue led to the case of Zubik v. Burwell, a legal challenge over the mandate exemption that went before the U.S. Supreme Court in March 2016. The issue was never resolved however, and in May 2016, the Supreme Court vacated the lower court rulings related to Zubik v. Burwell and remanded the case back to the four appeals courts that had originally ruled on the issue.
In 2018, the Trump administration announced new rules aimed at broadening exemptions to the ACA’s contraceptive mandate to entities that object to services covered by the mandate on the basis of “sincerely held religious beliefs.” A second rule allowed nonprofit organizations and small businesses that had nonreligious moral convictions against the mandate to opt out.
Thirteen states and the District of Columbia then sued the Trump administration over the rules, as well as Pennsylvania and New Jersey in a separate case. Little Sisters of the Poor, a religious nonprofit operating a home in Pittsburgh, intervened in the case as an aggrieved party. An appeal court temporarily barred the regulations from moving forward.
During oral arguments, Solicitor General for the Department of Justice Noel J. Francisco said the exemptions are lawful because they are authorized under a provision of the ACA as well as the Religious Freedom Restoration Act (RFRA).
“RFRA at the very least authorizes the religious exemption,” Mr. Francisco said during oral arguments.
Chief Deputy Attorney General for Pennsylvania Michael J. Fischer argued that the Trump administration’s moral and religious exemption rules rest on overly broad assertions of agency authority.
“First, the agencies twist a narrow delegation that allows the Health Resources and Services Administration to decide which preventive services insurers must cover under the Women’s Health Amendment into a grant of authority so broad it allows them to permit virtually any employer or college to opt out of providing contraceptive coverage entirely, including for reasons as amorphous as vaguely defined moral beliefs,” he said during oral arguments. “Second, the agencies claim that RFRA, a statute that limits government action, affirmatively authorizes them to permit employers to deny women their rights to contraceptive coverage even in the absence of a RFRA violation in the first place.”
U.S. Supreme Court justices appear divided over whether the Trump administration acted properly when it expanded exemptions under the Affordable Care Act’s contraception mandate.
During oral arguments on May 6, the court expressed differing perspectives about the administration’s authority to allow for more exemptions under the health law’s birth control mandate and whether the expansions were reasonable. Justices heard the consolidated cases – Little Sisters of the Poor v. Pennsylvania and Trump v. Pennsylvania – by teleconference because of the COVID-19 pandemic. They are expected to make a decision by the summer.
Associate justice Ruth Bader Ginsburg, who participated in the telephone conference call from a hospital where she was recovering from a gallbladder condition, said the exemptions ignored the intent of Congress to provide women with comprehensive coverage through the ACA.
“The glaring feature of what the government has done in expanding this exemption is to toss to the winds entirely Congress’s instruction that women need and shall have seamless, no-cost, comprehensive coverage,” she said during oral arguments. “This leaves the women to hunt for other government programs that might cover them, and for those who are not covered by Medicaid or one of the other government programs, they can get contraceptive coverage only from paying out of their own pocket, which is exactly what Congress didn’t want to happen.”
Associate Justice Samuel Alito Jr., meanwhile, indicated that a lower court opinion that had blocked the exemptions from going forward conflicts with the Supreme Court’s ruling in a related case, Burwell v. Hobby Lobby.
“Explain to me why the Third Circuit’s analysis of the question of substantial burden is not squarely inconsistent with our reasoning in Hobby Lobby,” Associate Justice Alito said during oral arguments. “Hobby Lobby held that, if a person sincerely believes that it is immoral to perform an act that has the effect of enabling another person to commit an immoral act, a federal court does not have the right to say that this person is wrong on the question of moral complicity. That’s precisely the situation here. Reading the Third Circuit’s discussion of the substantial burden question, I wondered whether they had read that part of the Hobby Lobby decision.”
The dispute surrounding the ACA’s birth control mandate and the extent of exemptions afforded has gone on for a decade and has led to numerous legal challenges. The ACA initially required all employers to cover birth control for employees with no copayments, but exempted group health plans of religious employers. Those religious employers were primarily churches and other houses of worship. After a number of complaints and lawsuits, the Obama administration created a workaround for nonprofit religious employers not included in that exemption to opt out of the mandate. However, critics argued the process itself was a violation of their religious freedom.
The issue led to the case of Zubik v. Burwell, a legal challenge over the mandate exemption that went before the U.S. Supreme Court in March 2016. The issue was never resolved however, and in May 2016, the Supreme Court vacated the lower court rulings related to Zubik v. Burwell and remanded the case back to the four appeals courts that had originally ruled on the issue.
In 2018, the Trump administration announced new rules aimed at broadening exemptions to the ACA’s contraceptive mandate to entities that object to services covered by the mandate on the basis of “sincerely held religious beliefs.” A second rule allowed nonprofit organizations and small businesses that had nonreligious moral convictions against the mandate to opt out.
Thirteen states and the District of Columbia then sued the Trump administration over the rules, as well as Pennsylvania and New Jersey in a separate case. Little Sisters of the Poor, a religious nonprofit operating a home in Pittsburgh, intervened in the case as an aggrieved party. An appeal court temporarily barred the regulations from moving forward.
During oral arguments, Solicitor General for the Department of Justice Noel J. Francisco said the exemptions are lawful because they are authorized under a provision of the ACA as well as the Religious Freedom Restoration Act (RFRA).
“RFRA at the very least authorizes the religious exemption,” Mr. Francisco said during oral arguments.
Chief Deputy Attorney General for Pennsylvania Michael J. Fischer argued that the Trump administration’s moral and religious exemption rules rest on overly broad assertions of agency authority.
“First, the agencies twist a narrow delegation that allows the Health Resources and Services Administration to decide which preventive services insurers must cover under the Women’s Health Amendment into a grant of authority so broad it allows them to permit virtually any employer or college to opt out of providing contraceptive coverage entirely, including for reasons as amorphous as vaguely defined moral beliefs,” he said during oral arguments. “Second, the agencies claim that RFRA, a statute that limits government action, affirmatively authorizes them to permit employers to deny women their rights to contraceptive coverage even in the absence of a RFRA violation in the first place.”
Postpartum IUD placement • breastfeeding • difficulty maintaining milk supply • Dx?
THE CASE
A 28-year-old G1P1 initially presented to the family medicine clinic 4 weeks postpartum to discuss possibilities for contraception. She had received her prenatal care through a midwife and had had a successful home delivery. She was exclusively breastfeeding her infant daughter but wanted to ensure adequate spacing between her pregnancies.
During the discussion of possible options, the patient revealed that she had previously had an intrauterine device (IUD) placed and expressed interest in using this method again. A levonorgestrel-releasing IUD (Mirena) was placed at 6 weeks postpartum, after a negative pregnancy test was obtained.
The patient returned to the clinic about 6 months later with complaints of increased difficulty maintaining her milk supply.
THE DIAGNOSIS
The patient had taken a home pregnancy test, which was positive—a finding confirmed in clinic via a urine pregnancy test.
Gestational age. Since the patient had an IUD in place and had been exclusively breastfeeding, gestational age was difficult to determine. A quantitative human chorionic gonadotropin (hCG) test showed an hCG level of 12,469 U/L, consistent with a 4-to-8-week pregnancy. An ultrasound performed the next day showed a single intrauterine pregnancy at 21 weeks.
IUD location. There was also the question of the location of the IUD and whether it would interfere with the patient’s ability to maintain the pregnancy. On ultrasound, the IUD was noted within the cervix and myometrium. After discussion of the risks, the patient chose to leave it in place.
DISCUSSION
IUDs are among the most effective forms of contraception; levonorgestrel-releasing IUDs are more effective than copper IUDs.1 The rates of failure in the first year of use are 0.8% and 0.2% for copper and levonorgestrel-releasing IUDs, respectively.1
Continue to: The Lactational Amenorrhea Method
The Lactational Amenorrhea Method (LAM), which is defined as providing infant nutrition exclusively through breastmilk during the first 6 months postpartum, also provides protection against pregnancy. LAM has a failure rate of 0% to 1.5%.2
It is not surprising that this patient thought she was adequately protected against pregnancy. That said, no contraceptive method is foolproof (as this case demonstrates).
Risks to the pregnancy. When pregnancy does occur with an IUD in place, the patient should be informed of the possible risks to the pregnancy. These include complications such as spontaneous abortion, chorioamnionitis, and preterm delivery.3 Risk is further increased if the IUD is malpositioned (as this one was), meaning that any part of the IUD is located in the lower uterine segment, myometrium, or endocervical canal.4,5
Removal of the IUD is generally recommended if the device and its strings can be located, although removal does not completely mitigate risk. In a study done in Egypt, 46 of 52 IUDs were removed successfully, with 2 spontaneous abortions as a result.6 Of note, the IUDs extracted in this study were Lippes loop and copper models, not levonorgestrel-releasing IUDs such as our patient had. There is a single case report7 of a patient who had a Mirena inserted very early in a pregnancy; the IUD had to be left in place due to the risk for miscarriage, but she was able to carry the infant to term and did not experience any adverse effects.
Our patient
The patient delivered a male infant vaginally at term without issue. However, the IUD was not expelled during this process. Ultrasound showed that it was embedded in the posterior myometrium with a hypoechoic tract. The patient was referred to Gynecology, and the IUD was successfully removed.
Continue to: THE TAKEAWAY
THE TAKEAWAY
Even the most reliable method of contraception can fail—so pregnancy should always be in the differential diagnosis for a sexually active woman. Location of IUD placement is important; it must be in the right place to be effective. The tenets of LAM must be followed precisely in order for breastfeeding to provide protection against pregnancy. Patients can successfully carry a pregnancy to term with an IUD, as this patient did, but it places them at higher risk for ectopic pregnancy, premature rupture of membranes, and infection.
The author thanks Jenny Walters, lactation consultant, for her assistance in the preparation of the manuscript.
CORRESPONDENCE
Hannah Maxfield, MD, 3901 Rainbow Boulevard, MS 4010, Kansas City, KS 66160; [email protected]
1. Heinemann K, Reed S, Moehner S, et al. Comparative contraceptive effectiveness of levonorgestrel-releasing and copper intrauterine devices: the European Active Surveillance Study for Intrauterine Devices. Contraception. 2015;91:280-283.
2. Labbok MH. Postpartum sexuality and the Lactational Amenorrhea Method for contraception. Clin Obstet Gynecol. 2015;58:915-927.
3. Ganer H, Levy A, Ohel I, et al. Pregnancy outcome in women with an intrauterine contraceptive device. Am J Obstet Gynecol. 2009;201:381.e1-e5.
4. Moschos E, Twickler D. Intrauterine devices in early pregnancy: findings on ultrasound and clinical outcomes. Am J Obstet Gynecol. 2011;204:427.e1-e6.
5. Ozgu-Erdinc AS, Tasdemir UG, Uygur D, et al. Outcome of intrauterine pregnancies with intrauterine device in place and effects of device location on prognosis. Contraception. 2014;89:426-430.
6. Assaf A, Gohar M, Saad S, et al. Removal of intrauterine devices with missing tails during early pregnancy. Contraception. 1992;45:541-546.
7. Gardyszewska A, Czajkowski K. Application of levonorgestrel-releasing intrauterine system in early pregnancy: a case report [article in Polish]. Ginekol Pol. 2012;83:950-952.
THE CASE
A 28-year-old G1P1 initially presented to the family medicine clinic 4 weeks postpartum to discuss possibilities for contraception. She had received her prenatal care through a midwife and had had a successful home delivery. She was exclusively breastfeeding her infant daughter but wanted to ensure adequate spacing between her pregnancies.
During the discussion of possible options, the patient revealed that she had previously had an intrauterine device (IUD) placed and expressed interest in using this method again. A levonorgestrel-releasing IUD (Mirena) was placed at 6 weeks postpartum, after a negative pregnancy test was obtained.
The patient returned to the clinic about 6 months later with complaints of increased difficulty maintaining her milk supply.
THE DIAGNOSIS
The patient had taken a home pregnancy test, which was positive—a finding confirmed in clinic via a urine pregnancy test.
Gestational age. Since the patient had an IUD in place and had been exclusively breastfeeding, gestational age was difficult to determine. A quantitative human chorionic gonadotropin (hCG) test showed an hCG level of 12,469 U/L, consistent with a 4-to-8-week pregnancy. An ultrasound performed the next day showed a single intrauterine pregnancy at 21 weeks.
IUD location. There was also the question of the location of the IUD and whether it would interfere with the patient’s ability to maintain the pregnancy. On ultrasound, the IUD was noted within the cervix and myometrium. After discussion of the risks, the patient chose to leave it in place.
DISCUSSION
IUDs are among the most effective forms of contraception; levonorgestrel-releasing IUDs are more effective than copper IUDs.1 The rates of failure in the first year of use are 0.8% and 0.2% for copper and levonorgestrel-releasing IUDs, respectively.1
Continue to: The Lactational Amenorrhea Method
The Lactational Amenorrhea Method (LAM), which is defined as providing infant nutrition exclusively through breastmilk during the first 6 months postpartum, also provides protection against pregnancy. LAM has a failure rate of 0% to 1.5%.2
It is not surprising that this patient thought she was adequately protected against pregnancy. That said, no contraceptive method is foolproof (as this case demonstrates).
Risks to the pregnancy. When pregnancy does occur with an IUD in place, the patient should be informed of the possible risks to the pregnancy. These include complications such as spontaneous abortion, chorioamnionitis, and preterm delivery.3 Risk is further increased if the IUD is malpositioned (as this one was), meaning that any part of the IUD is located in the lower uterine segment, myometrium, or endocervical canal.4,5
Removal of the IUD is generally recommended if the device and its strings can be located, although removal does not completely mitigate risk. In a study done in Egypt, 46 of 52 IUDs were removed successfully, with 2 spontaneous abortions as a result.6 Of note, the IUDs extracted in this study were Lippes loop and copper models, not levonorgestrel-releasing IUDs such as our patient had. There is a single case report7 of a patient who had a Mirena inserted very early in a pregnancy; the IUD had to be left in place due to the risk for miscarriage, but she was able to carry the infant to term and did not experience any adverse effects.
Our patient
The patient delivered a male infant vaginally at term without issue. However, the IUD was not expelled during this process. Ultrasound showed that it was embedded in the posterior myometrium with a hypoechoic tract. The patient was referred to Gynecology, and the IUD was successfully removed.
Continue to: THE TAKEAWAY
THE TAKEAWAY
Even the most reliable method of contraception can fail—so pregnancy should always be in the differential diagnosis for a sexually active woman. Location of IUD placement is important; it must be in the right place to be effective. The tenets of LAM must be followed precisely in order for breastfeeding to provide protection against pregnancy. Patients can successfully carry a pregnancy to term with an IUD, as this patient did, but it places them at higher risk for ectopic pregnancy, premature rupture of membranes, and infection.
The author thanks Jenny Walters, lactation consultant, for her assistance in the preparation of the manuscript.
CORRESPONDENCE
Hannah Maxfield, MD, 3901 Rainbow Boulevard, MS 4010, Kansas City, KS 66160; [email protected]
THE CASE
A 28-year-old G1P1 initially presented to the family medicine clinic 4 weeks postpartum to discuss possibilities for contraception. She had received her prenatal care through a midwife and had had a successful home delivery. She was exclusively breastfeeding her infant daughter but wanted to ensure adequate spacing between her pregnancies.
During the discussion of possible options, the patient revealed that she had previously had an intrauterine device (IUD) placed and expressed interest in using this method again. A levonorgestrel-releasing IUD (Mirena) was placed at 6 weeks postpartum, after a negative pregnancy test was obtained.
The patient returned to the clinic about 6 months later with complaints of increased difficulty maintaining her milk supply.
THE DIAGNOSIS
The patient had taken a home pregnancy test, which was positive—a finding confirmed in clinic via a urine pregnancy test.
Gestational age. Since the patient had an IUD in place and had been exclusively breastfeeding, gestational age was difficult to determine. A quantitative human chorionic gonadotropin (hCG) test showed an hCG level of 12,469 U/L, consistent with a 4-to-8-week pregnancy. An ultrasound performed the next day showed a single intrauterine pregnancy at 21 weeks.
IUD location. There was also the question of the location of the IUD and whether it would interfere with the patient’s ability to maintain the pregnancy. On ultrasound, the IUD was noted within the cervix and myometrium. After discussion of the risks, the patient chose to leave it in place.
DISCUSSION
IUDs are among the most effective forms of contraception; levonorgestrel-releasing IUDs are more effective than copper IUDs.1 The rates of failure in the first year of use are 0.8% and 0.2% for copper and levonorgestrel-releasing IUDs, respectively.1
Continue to: The Lactational Amenorrhea Method
The Lactational Amenorrhea Method (LAM), which is defined as providing infant nutrition exclusively through breastmilk during the first 6 months postpartum, also provides protection against pregnancy. LAM has a failure rate of 0% to 1.5%.2
It is not surprising that this patient thought she was adequately protected against pregnancy. That said, no contraceptive method is foolproof (as this case demonstrates).
Risks to the pregnancy. When pregnancy does occur with an IUD in place, the patient should be informed of the possible risks to the pregnancy. These include complications such as spontaneous abortion, chorioamnionitis, and preterm delivery.3 Risk is further increased if the IUD is malpositioned (as this one was), meaning that any part of the IUD is located in the lower uterine segment, myometrium, or endocervical canal.4,5
Removal of the IUD is generally recommended if the device and its strings can be located, although removal does not completely mitigate risk. In a study done in Egypt, 46 of 52 IUDs were removed successfully, with 2 spontaneous abortions as a result.6 Of note, the IUDs extracted in this study were Lippes loop and copper models, not levonorgestrel-releasing IUDs such as our patient had. There is a single case report7 of a patient who had a Mirena inserted very early in a pregnancy; the IUD had to be left in place due to the risk for miscarriage, but she was able to carry the infant to term and did not experience any adverse effects.
Our patient
The patient delivered a male infant vaginally at term without issue. However, the IUD was not expelled during this process. Ultrasound showed that it was embedded in the posterior myometrium with a hypoechoic tract. The patient was referred to Gynecology, and the IUD was successfully removed.
Continue to: THE TAKEAWAY
THE TAKEAWAY
Even the most reliable method of contraception can fail—so pregnancy should always be in the differential diagnosis for a sexually active woman. Location of IUD placement is important; it must be in the right place to be effective. The tenets of LAM must be followed precisely in order for breastfeeding to provide protection against pregnancy. Patients can successfully carry a pregnancy to term with an IUD, as this patient did, but it places them at higher risk for ectopic pregnancy, premature rupture of membranes, and infection.
The author thanks Jenny Walters, lactation consultant, for her assistance in the preparation of the manuscript.
CORRESPONDENCE
Hannah Maxfield, MD, 3901 Rainbow Boulevard, MS 4010, Kansas City, KS 66160; [email protected]
1. Heinemann K, Reed S, Moehner S, et al. Comparative contraceptive effectiveness of levonorgestrel-releasing and copper intrauterine devices: the European Active Surveillance Study for Intrauterine Devices. Contraception. 2015;91:280-283.
2. Labbok MH. Postpartum sexuality and the Lactational Amenorrhea Method for contraception. Clin Obstet Gynecol. 2015;58:915-927.
3. Ganer H, Levy A, Ohel I, et al. Pregnancy outcome in women with an intrauterine contraceptive device. Am J Obstet Gynecol. 2009;201:381.e1-e5.
4. Moschos E, Twickler D. Intrauterine devices in early pregnancy: findings on ultrasound and clinical outcomes. Am J Obstet Gynecol. 2011;204:427.e1-e6.
5. Ozgu-Erdinc AS, Tasdemir UG, Uygur D, et al. Outcome of intrauterine pregnancies with intrauterine device in place and effects of device location on prognosis. Contraception. 2014;89:426-430.
6. Assaf A, Gohar M, Saad S, et al. Removal of intrauterine devices with missing tails during early pregnancy. Contraception. 1992;45:541-546.
7. Gardyszewska A, Czajkowski K. Application of levonorgestrel-releasing intrauterine system in early pregnancy: a case report [article in Polish]. Ginekol Pol. 2012;83:950-952.
1. Heinemann K, Reed S, Moehner S, et al. Comparative contraceptive effectiveness of levonorgestrel-releasing and copper intrauterine devices: the European Active Surveillance Study for Intrauterine Devices. Contraception. 2015;91:280-283.
2. Labbok MH. Postpartum sexuality and the Lactational Amenorrhea Method for contraception. Clin Obstet Gynecol. 2015;58:915-927.
3. Ganer H, Levy A, Ohel I, et al. Pregnancy outcome in women with an intrauterine contraceptive device. Am J Obstet Gynecol. 2009;201:381.e1-e5.
4. Moschos E, Twickler D. Intrauterine devices in early pregnancy: findings on ultrasound and clinical outcomes. Am J Obstet Gynecol. 2011;204:427.e1-e6.
5. Ozgu-Erdinc AS, Tasdemir UG, Uygur D, et al. Outcome of intrauterine pregnancies with intrauterine device in place and effects of device location on prognosis. Contraception. 2014;89:426-430.
6. Assaf A, Gohar M, Saad S, et al. Removal of intrauterine devices with missing tails during early pregnancy. Contraception. 1992;45:541-546.
7. Gardyszewska A, Czajkowski K. Application of levonorgestrel-releasing intrauterine system in early pregnancy: a case report [article in Polish]. Ginekol Pol. 2012;83:950-952.
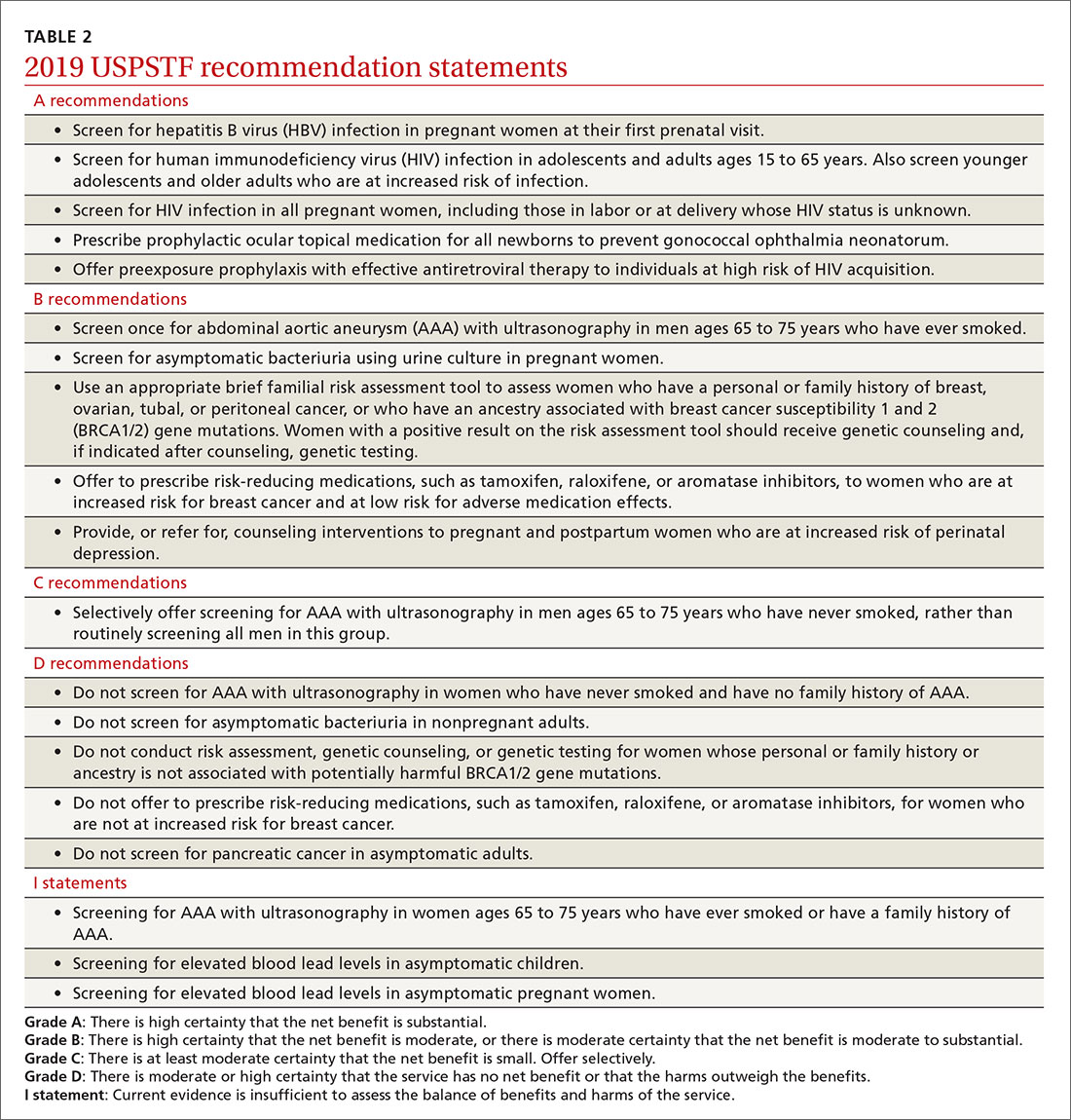
USPSTF round-up
In 2019, the US Preventive Services Task Force published 19 recommendation statements on 11 topics. Two of the topics are new; 9 are topics the Task Force had previously reviewed and has updated (TABLE 1). Three of these topics have been covered in Practice Alert podcasts (mdedge.com/familymedicine) and will not be discussed here: risk assessment, genetic counseling, and genetic testing for breast cancer susceptibility gene mutations (October 2019); medications to reduce the risk of breast cancer (December 2019); and preexposure prophylaxis to prevent HIV infections (January 2020).

Of the 19 recommendation statements made in 2019 (TABLE 2), 5 were rated “A” and 5 were “B,” meaning the evidence shows that benefits outweigh harms and these interventions should be offered in primary care practice. There were 5 “D” recommendations for interventions that should not be offered because they are either ineffective or harms exceed benefits. There were 3 “I” statements on interventions having insufficient evidence on benefits or harms to warrant a recommendation. Only 1 recommendation was rated “C” (selectively offer based on individual factors); this assessment is the hardest one to interpret and implement. Keep in mind that all “A” and “B” recommendations must be covered by commercial health plans with no out-of-pocket cost to the patient (ie, no co-pay or deductible).
New recommendation on preventing perinatal depression
One of 2 new topics reviewed in 2019 was the prevention of perinatal depression. (As noted, the other on preexposure prophylaxis to prevent HIV infection has already been covered in a Practice Alert podcast.) The Task Force found that the prevalence of depression is estimated at 8.9% among pregnant women and 37% at any point in the first year postpartum.1
Depression during pregnancy and the postpartum period is associated with adverse effects on the mother and infant, including higher rates of suicide and suicidal ideation and thoughts of harming the infant.1 Women with perinatal depression are also more likely to exhibit significantly lower levels of positive maternal behaviors, such as praising and playing with their child,2 and higher rates of negative maternal behaviors.2 Perinatal depression is also associated with increased rates of preterm birth and low birth weight.3
Mothers with postpartum depression have higher rates of early termination of breast feeding and lower adherence for recommended child preventive services including vaccination.1 Children of mothers with perinatal depression develop more behavior problems, have lower cognitive functioning, and have an increased risk of psychiatric disorders than do children of mothers without this condition.4,5
A number of risk factors are associated with perinatal depression, but no screening tool was found to have enough predictive value to be recommended. In deciding who should receive an offer or referral for counseling, the Task Force recommends as a practical approach providing “counseling interventions to women with 1 or more of the following: a history of depression, current depressive symptoms (that do not reach a diagnostic threshold), certain socioeconomic risk factors such as low income or adolescent or single parenthood, recent intimate partner violence, or mental health-related factors such as elevated anxiety symptoms or a history of significant negative life events.”1
There is no conclusive evidence to guide timing of counseling interventions, but most studies reviewed started them in the second trimester. These studies included cognitive behavioral therapy and interpersonal therapy and involved counseling sessions that ranged from 4 to 20 sessions and lasted for 4 to 70 weeks. They involved group and individual sessions, mostly in-person visits, and were provided by a variety of health professionals.6
Continue to: The studies reviewed showed...
The studies reviewed showed that counseling interventions reduced the likelihood of developing depression symptoms by 39%, with a number needed to treat of 13.5.6 Studies that looked at pregnancy and maternal and infant clinical outcomes were mixed but usually found little to no difference with counseling.6 Even so, the Task Force felt that a reduction in depression itself was enough to warrant a “B” recommendation.
Screening for abdominal aortic aneurisms
Ultrasound is underused in screening for abdominal aortic aneurisms (AAA) and preventing death from their rupture. (See “Whom should you screen for abdominal aortic aneurysm?”) The prevalence of AAA is the United States is unknown; in other western countries it varies from 1.2% to 3.3% in men and is declining due to decreased rates of smoking, the primary risk factor.
The risk of AAA rupture is related to the size of the aneurism, and surgical repair (either endovascular or open repair) is usually reserved for lesions > 5.5 cm in diameter or for smaller ones that are rapidly increasing in size. The standard of care for most aneurysms < 5.5 cm is to periodically monitor growth using ultrasound.
The 2019 recommendations on AAA screening are essentially the same as those made in 2004; evaluation of new evidence supported the previous recommendations. The Task Force recommends one-time screening for men ages 65 to 75 years who have ever smoked (B recommendation). Selective screening is recommended for men in this age group who have never smoked, based mainly on personal factors such as a family history of AAA, the presence of other arterial aneurisms, and the number of risk factors for cardiovascular disease (C recommendation).
The Task Force recommends against screening women ages 65 to 75 years with no history of smoking or family history of AAA, while the evidence was felt to be insufficient to make a recommendation for women in this age range who have either risk factor. This is problematic for family physicians since women with these risk factors are at increased risk of AAA compared with women without risk factors.8 And aneurisms in women appear to rupture more frequently at smaller sizes, although at a later age than in men.8 Operative mortality is also higher in women than in men8 and there is no direct evidence that screening improves outcomes for women.
Continue to: Screening for asymptomatic bacteriuria
Screening for asymptomatic bacteriuria
The Task Force re-examined and reconfirmed its previous recommendations on screening for asymptomatic bacteriuria in adults. It recommends in favor of it for pregnant women, using a urine culture to screen, and against it for all other adults. There is good evidence that treating screen-detected asymptomatic bacteriuria in pregnant women reduces the incidence of pyelonephritis in pregnancy.
The Task Force made this a “B” recommendation based on a lower prevalence of pyelonephritis found in more recent studies, making the overall magnitude of benefits moderate. There is also good evidence that treating asymptomatic bacteriuria in nonpregnant adults offers no benefits.9 The Task Force has re-examined this topic 5 times since 1996 with essentially the same results.
Screening for elevated lead levels in children and pregnant women
In 2019 the Task Force changed its 2006 recommendation on screening for elevated lead levels. The earlier recommendation advised against screening both children ages 1 to 5 years and pregnant women at average risk for elevated blood lead levels. In 2006 the Task Force also felt that evidence was insufficient to make a recommendation regarding children ages 1 to 5 years at elevated risk.
The Task Force now believes the evidence is insufficient to make a recommendation for all children ages 1 to 5 years and for pregnant women, thus moving from a “D” to an “I” recommendation for children and pregnant women with average risk. Even though there is little evidence to support screening for elevated lead levels in children ages 1 to 5 years and in pregnant women, the Task Force apparently did not feel comfortable recommending against testing, given that the cutoff for elevated blood lead levels has been lowered from 10 to 5 mcg/dL and that other sources of lead may now be more prevalent than in 2006.10
Remember that the Medicaid Early and Periodic Screening, Diagnostic, and Treatment program requires that all children receive a blood lead test twice, at ages 12 and 24 months, and that previously unscreened children ages 36 to 72 months must be tested once.
Continue to: Additional updates with no recommendation changes
Additional updates with no recommendation changes
Four other topics were re-examined by the Task Force in 2019, resulting in no significant changes to recommendations (TABLE 2):
- Screen for hepatitis B infection in pregnant women at the first prenatal visit (A recommendation; updated from 2009).
- Screen for HIV infection in adolescents and adults ages 15 to 65 years, and in those younger and older who are at high risk, and during pregnancy (A recommendation; updated from 2013).
- Provide topical medication for all newborns to prevent gonococcal ophthalmia neonatorum (A recommendation; first recommendation in 1996, updated in 2005 and 2011).
- Avoid screening for pancreatic cancer in asymptomatic adults (D recommendation; updated from 2004).
Affirmation of USPSTF’s value
In only 1 out of 9 reassessments of past topics did the Task Force modify its previous recommendations in any significant way. This demonstrates that recommendations will usually stand the test of time if they are made using robust, evidence-based methods (that consider both benefits and harms) and they are not made when evidence is insufficient. That only 2 new topics could be addressed in 2019 may reflect a need for more resources for the Task Force.
1. USPSTF. Interventions to prevent perinatal depression: US Preventive Services Task Force recommendation statement. 2019;321:580-587.
2. Lovejoy MC, Graczyk PA, O’Hare E, et al. Maternal depression and parenting behavior: a meta-analytic review. Clin Psychol Rev. 2000;20:561-592.
3. Szegda K, Markenson G, Bertone-Johnson ER, et al. Depression during pregnancy: a risk factor for adverse neonatal outcomes? A critical review of the literature. J Matern Fetal Neonatal Med. 2014;27:960-967.
4. Beck CT. The effects of postpartum depression on child development: a meta-analysis. Arch Psychiatr Nurs. 1998;12:12-20.
5. Santos IS, Matijasevich A, Barros AJ, et al. Antenatal and postnatal maternal mood symptoms and psychiatric disorders in pre-school children from the 2004 Pelotas Birth Cohort. J Affect Disord. 2014;164:112-117.
6. O’Connor E, Senger CA, Henniger ML, et al. Interventions to prevent perinatal depression. Evidence report and systematic review for the US preventive services task force. JAMA. 2019;321:588-601.
7. USPSTF. Screening for abdominal aortic aneurysm: US Preventive Services Task Force recommendation statement. 2019;322:2211-2218.
8. Guirguis-Blake JM, Beil TL, Senger CA, et al. Primary care screening for abdominal aortic aneurysm: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2019;322:2219-2238.
9. USPSTF. Owens DK, Davidson KW, Krist AH, et al. Screening for asymptomatic bacteriuria in adults: US Preventive Services Task Force recommendation statement. 2019;322:1188-1194.
10. USPSTF. Screening for elevated blood lead levels in children and pregnant women: US Preventive Services Task Force recommendation statement. 2019;321:1502-1509.
In 2019, the US Preventive Services Task Force published 19 recommendation statements on 11 topics. Two of the topics are new; 9 are topics the Task Force had previously reviewed and has updated (TABLE 1). Three of these topics have been covered in Practice Alert podcasts (mdedge.com/familymedicine) and will not be discussed here: risk assessment, genetic counseling, and genetic testing for breast cancer susceptibility gene mutations (October 2019); medications to reduce the risk of breast cancer (December 2019); and preexposure prophylaxis to prevent HIV infections (January 2020).

Of the 19 recommendation statements made in 2019 (TABLE 2), 5 were rated “A” and 5 were “B,” meaning the evidence shows that benefits outweigh harms and these interventions should be offered in primary care practice. There were 5 “D” recommendations for interventions that should not be offered because they are either ineffective or harms exceed benefits. There were 3 “I” statements on interventions having insufficient evidence on benefits or harms to warrant a recommendation. Only 1 recommendation was rated “C” (selectively offer based on individual factors); this assessment is the hardest one to interpret and implement. Keep in mind that all “A” and “B” recommendations must be covered by commercial health plans with no out-of-pocket cost to the patient (ie, no co-pay or deductible).

New recommendation on preventing perinatal depression
One of 2 new topics reviewed in 2019 was the prevention of perinatal depression. (As noted, the other on preexposure prophylaxis to prevent HIV infection has already been covered in a Practice Alert podcast.) The Task Force found that the prevalence of depression is estimated at 8.9% among pregnant women and 37% at any point in the first year postpartum.1
Depression during pregnancy and the postpartum period is associated with adverse effects on the mother and infant, including higher rates of suicide and suicidal ideation and thoughts of harming the infant.1 Women with perinatal depression are also more likely to exhibit significantly lower levels of positive maternal behaviors, such as praising and playing with their child,2 and higher rates of negative maternal behaviors.2 Perinatal depression is also associated with increased rates of preterm birth and low birth weight.3
Mothers with postpartum depression have higher rates of early termination of breast feeding and lower adherence for recommended child preventive services including vaccination.1 Children of mothers with perinatal depression develop more behavior problems, have lower cognitive functioning, and have an increased risk of psychiatric disorders than do children of mothers without this condition.4,5
A number of risk factors are associated with perinatal depression, but no screening tool was found to have enough predictive value to be recommended. In deciding who should receive an offer or referral for counseling, the Task Force recommends as a practical approach providing “counseling interventions to women with 1 or more of the following: a history of depression, current depressive symptoms (that do not reach a diagnostic threshold), certain socioeconomic risk factors such as low income or adolescent or single parenthood, recent intimate partner violence, or mental health-related factors such as elevated anxiety symptoms or a history of significant negative life events.”1
There is no conclusive evidence to guide timing of counseling interventions, but most studies reviewed started them in the second trimester. These studies included cognitive behavioral therapy and interpersonal therapy and involved counseling sessions that ranged from 4 to 20 sessions and lasted for 4 to 70 weeks. They involved group and individual sessions, mostly in-person visits, and were provided by a variety of health professionals.6
Continue to: The studies reviewed showed...
The studies reviewed showed that counseling interventions reduced the likelihood of developing depression symptoms by 39%, with a number needed to treat of 13.5.6 Studies that looked at pregnancy and maternal and infant clinical outcomes were mixed but usually found little to no difference with counseling.6 Even so, the Task Force felt that a reduction in depression itself was enough to warrant a “B” recommendation.
Screening for abdominal aortic aneurisms
Ultrasound is underused in screening for abdominal aortic aneurisms (AAA) and preventing death from their rupture. (See “Whom should you screen for abdominal aortic aneurysm?”) The prevalence of AAA is the United States is unknown; in other western countries it varies from 1.2% to 3.3% in men and is declining due to decreased rates of smoking, the primary risk factor.
The risk of AAA rupture is related to the size of the aneurism, and surgical repair (either endovascular or open repair) is usually reserved for lesions > 5.5 cm in diameter or for smaller ones that are rapidly increasing in size. The standard of care for most aneurysms < 5.5 cm is to periodically monitor growth using ultrasound.
The 2019 recommendations on AAA screening are essentially the same as those made in 2004; evaluation of new evidence supported the previous recommendations. The Task Force recommends one-time screening for men ages 65 to 75 years who have ever smoked (B recommendation). Selective screening is recommended for men in this age group who have never smoked, based mainly on personal factors such as a family history of AAA, the presence of other arterial aneurisms, and the number of risk factors for cardiovascular disease (C recommendation).
The Task Force recommends against screening women ages 65 to 75 years with no history of smoking or family history of AAA, while the evidence was felt to be insufficient to make a recommendation for women in this age range who have either risk factor. This is problematic for family physicians since women with these risk factors are at increased risk of AAA compared with women without risk factors.8 And aneurisms in women appear to rupture more frequently at smaller sizes, although at a later age than in men.8 Operative mortality is also higher in women than in men8 and there is no direct evidence that screening improves outcomes for women.
Continue to: Screening for asymptomatic bacteriuria
Screening for asymptomatic bacteriuria
The Task Force re-examined and reconfirmed its previous recommendations on screening for asymptomatic bacteriuria in adults. It recommends in favor of it for pregnant women, using a urine culture to screen, and against it for all other adults. There is good evidence that treating screen-detected asymptomatic bacteriuria in pregnant women reduces the incidence of pyelonephritis in pregnancy.
The Task Force made this a “B” recommendation based on a lower prevalence of pyelonephritis found in more recent studies, making the overall magnitude of benefits moderate. There is also good evidence that treating asymptomatic bacteriuria in nonpregnant adults offers no benefits.9 The Task Force has re-examined this topic 5 times since 1996 with essentially the same results.
Screening for elevated lead levels in children and pregnant women
In 2019 the Task Force changed its 2006 recommendation on screening for elevated lead levels. The earlier recommendation advised against screening both children ages 1 to 5 years and pregnant women at average risk for elevated blood lead levels. In 2006 the Task Force also felt that evidence was insufficient to make a recommendation regarding children ages 1 to 5 years at elevated risk.
The Task Force now believes the evidence is insufficient to make a recommendation for all children ages 1 to 5 years and for pregnant women, thus moving from a “D” to an “I” recommendation for children and pregnant women with average risk. Even though there is little evidence to support screening for elevated lead levels in children ages 1 to 5 years and in pregnant women, the Task Force apparently did not feel comfortable recommending against testing, given that the cutoff for elevated blood lead levels has been lowered from 10 to 5 mcg/dL and that other sources of lead may now be more prevalent than in 2006.10
Remember that the Medicaid Early and Periodic Screening, Diagnostic, and Treatment program requires that all children receive a blood lead test twice, at ages 12 and 24 months, and that previously unscreened children ages 36 to 72 months must be tested once.
Continue to: Additional updates with no recommendation changes
Additional updates with no recommendation changes
Four other topics were re-examined by the Task Force in 2019, resulting in no significant changes to recommendations (TABLE 2):
- Screen for hepatitis B infection in pregnant women at the first prenatal visit (A recommendation; updated from 2009).
- Screen for HIV infection in adolescents and adults ages 15 to 65 years, and in those younger and older who are at high risk, and during pregnancy (A recommendation; updated from 2013).
- Provide topical medication for all newborns to prevent gonococcal ophthalmia neonatorum (A recommendation; first recommendation in 1996, updated in 2005 and 2011).
- Avoid screening for pancreatic cancer in asymptomatic adults (D recommendation; updated from 2004).
Affirmation of USPSTF’s value
In only 1 out of 9 reassessments of past topics did the Task Force modify its previous recommendations in any significant way. This demonstrates that recommendations will usually stand the test of time if they are made using robust, evidence-based methods (that consider both benefits and harms) and they are not made when evidence is insufficient. That only 2 new topics could be addressed in 2019 may reflect a need for more resources for the Task Force.
In 2019, the US Preventive Services Task Force published 19 recommendation statements on 11 topics. Two of the topics are new; 9 are topics the Task Force had previously reviewed and has updated (TABLE 1). Three of these topics have been covered in Practice Alert podcasts (mdedge.com/familymedicine) and will not be discussed here: risk assessment, genetic counseling, and genetic testing for breast cancer susceptibility gene mutations (October 2019); medications to reduce the risk of breast cancer (December 2019); and preexposure prophylaxis to prevent HIV infections (January 2020).

Of the 19 recommendation statements made in 2019 (TABLE 2), 5 were rated “A” and 5 were “B,” meaning the evidence shows that benefits outweigh harms and these interventions should be offered in primary care practice. There were 5 “D” recommendations for interventions that should not be offered because they are either ineffective or harms exceed benefits. There were 3 “I” statements on interventions having insufficient evidence on benefits or harms to warrant a recommendation. Only 1 recommendation was rated “C” (selectively offer based on individual factors); this assessment is the hardest one to interpret and implement. Keep in mind that all “A” and “B” recommendations must be covered by commercial health plans with no out-of-pocket cost to the patient (ie, no co-pay or deductible).

New recommendation on preventing perinatal depression
One of 2 new topics reviewed in 2019 was the prevention of perinatal depression. (As noted, the other on preexposure prophylaxis to prevent HIV infection has already been covered in a Practice Alert podcast.) The Task Force found that the prevalence of depression is estimated at 8.9% among pregnant women and 37% at any point in the first year postpartum.1
Depression during pregnancy and the postpartum period is associated with adverse effects on the mother and infant, including higher rates of suicide and suicidal ideation and thoughts of harming the infant.1 Women with perinatal depression are also more likely to exhibit significantly lower levels of positive maternal behaviors, such as praising and playing with their child,2 and higher rates of negative maternal behaviors.2 Perinatal depression is also associated with increased rates of preterm birth and low birth weight.3
Mothers with postpartum depression have higher rates of early termination of breast feeding and lower adherence for recommended child preventive services including vaccination.1 Children of mothers with perinatal depression develop more behavior problems, have lower cognitive functioning, and have an increased risk of psychiatric disorders than do children of mothers without this condition.4,5
A number of risk factors are associated with perinatal depression, but no screening tool was found to have enough predictive value to be recommended. In deciding who should receive an offer or referral for counseling, the Task Force recommends as a practical approach providing “counseling interventions to women with 1 or more of the following: a history of depression, current depressive symptoms (that do not reach a diagnostic threshold), certain socioeconomic risk factors such as low income or adolescent or single parenthood, recent intimate partner violence, or mental health-related factors such as elevated anxiety symptoms or a history of significant negative life events.”1
There is no conclusive evidence to guide timing of counseling interventions, but most studies reviewed started them in the second trimester. These studies included cognitive behavioral therapy and interpersonal therapy and involved counseling sessions that ranged from 4 to 20 sessions and lasted for 4 to 70 weeks. They involved group and individual sessions, mostly in-person visits, and were provided by a variety of health professionals.6
Continue to: The studies reviewed showed...
The studies reviewed showed that counseling interventions reduced the likelihood of developing depression symptoms by 39%, with a number needed to treat of 13.5.6 Studies that looked at pregnancy and maternal and infant clinical outcomes were mixed but usually found little to no difference with counseling.6 Even so, the Task Force felt that a reduction in depression itself was enough to warrant a “B” recommendation.
Screening for abdominal aortic aneurisms
Ultrasound is underused in screening for abdominal aortic aneurisms (AAA) and preventing death from their rupture. (See “Whom should you screen for abdominal aortic aneurysm?”) The prevalence of AAA is the United States is unknown; in other western countries it varies from 1.2% to 3.3% in men and is declining due to decreased rates of smoking, the primary risk factor.
The risk of AAA rupture is related to the size of the aneurism, and surgical repair (either endovascular or open repair) is usually reserved for lesions > 5.5 cm in diameter or for smaller ones that are rapidly increasing in size. The standard of care for most aneurysms < 5.5 cm is to periodically monitor growth using ultrasound.
The 2019 recommendations on AAA screening are essentially the same as those made in 2004; evaluation of new evidence supported the previous recommendations. The Task Force recommends one-time screening for men ages 65 to 75 years who have ever smoked (B recommendation). Selective screening is recommended for men in this age group who have never smoked, based mainly on personal factors such as a family history of AAA, the presence of other arterial aneurisms, and the number of risk factors for cardiovascular disease (C recommendation).
The Task Force recommends against screening women ages 65 to 75 years with no history of smoking or family history of AAA, while the evidence was felt to be insufficient to make a recommendation for women in this age range who have either risk factor. This is problematic for family physicians since women with these risk factors are at increased risk of AAA compared with women without risk factors.8 And aneurisms in women appear to rupture more frequently at smaller sizes, although at a later age than in men.8 Operative mortality is also higher in women than in men8 and there is no direct evidence that screening improves outcomes for women.
Continue to: Screening for asymptomatic bacteriuria
Screening for asymptomatic bacteriuria
The Task Force re-examined and reconfirmed its previous recommendations on screening for asymptomatic bacteriuria in adults. It recommends in favor of it for pregnant women, using a urine culture to screen, and against it for all other adults. There is good evidence that treating screen-detected asymptomatic bacteriuria in pregnant women reduces the incidence of pyelonephritis in pregnancy.
The Task Force made this a “B” recommendation based on a lower prevalence of pyelonephritis found in more recent studies, making the overall magnitude of benefits moderate. There is also good evidence that treating asymptomatic bacteriuria in nonpregnant adults offers no benefits.9 The Task Force has re-examined this topic 5 times since 1996 with essentially the same results.
Screening for elevated lead levels in children and pregnant women
In 2019 the Task Force changed its 2006 recommendation on screening for elevated lead levels. The earlier recommendation advised against screening both children ages 1 to 5 years and pregnant women at average risk for elevated blood lead levels. In 2006 the Task Force also felt that evidence was insufficient to make a recommendation regarding children ages 1 to 5 years at elevated risk.
The Task Force now believes the evidence is insufficient to make a recommendation for all children ages 1 to 5 years and for pregnant women, thus moving from a “D” to an “I” recommendation for children and pregnant women with average risk. Even though there is little evidence to support screening for elevated lead levels in children ages 1 to 5 years and in pregnant women, the Task Force apparently did not feel comfortable recommending against testing, given that the cutoff for elevated blood lead levels has been lowered from 10 to 5 mcg/dL and that other sources of lead may now be more prevalent than in 2006.10
Remember that the Medicaid Early and Periodic Screening, Diagnostic, and Treatment program requires that all children receive a blood lead test twice, at ages 12 and 24 months, and that previously unscreened children ages 36 to 72 months must be tested once.
Continue to: Additional updates with no recommendation changes
Additional updates with no recommendation changes
Four other topics were re-examined by the Task Force in 2019, resulting in no significant changes to recommendations (TABLE 2):
- Screen for hepatitis B infection in pregnant women at the first prenatal visit (A recommendation; updated from 2009).
- Screen for HIV infection in adolescents and adults ages 15 to 65 years, and in those younger and older who are at high risk, and during pregnancy (A recommendation; updated from 2013).
- Provide topical medication for all newborns to prevent gonococcal ophthalmia neonatorum (A recommendation; first recommendation in 1996, updated in 2005 and 2011).
- Avoid screening for pancreatic cancer in asymptomatic adults (D recommendation; updated from 2004).
Affirmation of USPSTF’s value
In only 1 out of 9 reassessments of past topics did the Task Force modify its previous recommendations in any significant way. This demonstrates that recommendations will usually stand the test of time if they are made using robust, evidence-based methods (that consider both benefits and harms) and they are not made when evidence is insufficient. That only 2 new topics could be addressed in 2019 may reflect a need for more resources for the Task Force.
1. USPSTF. Interventions to prevent perinatal depression: US Preventive Services Task Force recommendation statement. 2019;321:580-587.
2. Lovejoy MC, Graczyk PA, O’Hare E, et al. Maternal depression and parenting behavior: a meta-analytic review. Clin Psychol Rev. 2000;20:561-592.
3. Szegda K, Markenson G, Bertone-Johnson ER, et al. Depression during pregnancy: a risk factor for adverse neonatal outcomes? A critical review of the literature. J Matern Fetal Neonatal Med. 2014;27:960-967.
4. Beck CT. The effects of postpartum depression on child development: a meta-analysis. Arch Psychiatr Nurs. 1998;12:12-20.
5. Santos IS, Matijasevich A, Barros AJ, et al. Antenatal and postnatal maternal mood symptoms and psychiatric disorders in pre-school children from the 2004 Pelotas Birth Cohort. J Affect Disord. 2014;164:112-117.
6. O’Connor E, Senger CA, Henniger ML, et al. Interventions to prevent perinatal depression. Evidence report and systematic review for the US preventive services task force. JAMA. 2019;321:588-601.
7. USPSTF. Screening for abdominal aortic aneurysm: US Preventive Services Task Force recommendation statement. 2019;322:2211-2218.
8. Guirguis-Blake JM, Beil TL, Senger CA, et al. Primary care screening for abdominal aortic aneurysm: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2019;322:2219-2238.
9. USPSTF. Owens DK, Davidson KW, Krist AH, et al. Screening for asymptomatic bacteriuria in adults: US Preventive Services Task Force recommendation statement. 2019;322:1188-1194.
10. USPSTF. Screening for elevated blood lead levels in children and pregnant women: US Preventive Services Task Force recommendation statement. 2019;321:1502-1509.
1. USPSTF. Interventions to prevent perinatal depression: US Preventive Services Task Force recommendation statement. 2019;321:580-587.
2. Lovejoy MC, Graczyk PA, O’Hare E, et al. Maternal depression and parenting behavior: a meta-analytic review. Clin Psychol Rev. 2000;20:561-592.
3. Szegda K, Markenson G, Bertone-Johnson ER, et al. Depression during pregnancy: a risk factor for adverse neonatal outcomes? A critical review of the literature. J Matern Fetal Neonatal Med. 2014;27:960-967.
4. Beck CT. The effects of postpartum depression on child development: a meta-analysis. Arch Psychiatr Nurs. 1998;12:12-20.
5. Santos IS, Matijasevich A, Barros AJ, et al. Antenatal and postnatal maternal mood symptoms and psychiatric disorders in pre-school children from the 2004 Pelotas Birth Cohort. J Affect Disord. 2014;164:112-117.
6. O’Connor E, Senger CA, Henniger ML, et al. Interventions to prevent perinatal depression. Evidence report and systematic review for the US preventive services task force. JAMA. 2019;321:588-601.
7. USPSTF. Screening for abdominal aortic aneurysm: US Preventive Services Task Force recommendation statement. 2019;322:2211-2218.
8. Guirguis-Blake JM, Beil TL, Senger CA, et al. Primary care screening for abdominal aortic aneurysm: evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2019;322:2219-2238.
9. USPSTF. Owens DK, Davidson KW, Krist AH, et al. Screening for asymptomatic bacteriuria in adults: US Preventive Services Task Force recommendation statement. 2019;322:1188-1194.
10. USPSTF. Screening for elevated blood lead levels in children and pregnant women: US Preventive Services Task Force recommendation statement. 2019;321:1502-1509.
AHA emphasizes the need for cardio-obstetrics teams
according to a statement from the American Heart Association.
Cardiovascular disease (CVD) remains the leading cause of pregnancy-related mortality in the United States, and accounted for approximately 17 deaths per 100,000 live births in 2015, wrote Laxmi S. Mehta, MD, of The Ohio State University, Columbus, and colleagues.
Ideally, a woman with CVD at the time of pregnancy should be managed by a multidisciplinary cardio-obstetrics team that can assess cardiovascular risk, obstetric risk, and fetal risk throughout pregnancy, delivery, and up to a year post partum. The team should develop a shared strategy to promote best outcomes, according to the statement. The cardio-obstetrics team may include obstetricians, cardiologists, anesthesiologists, maternal-fetal medicine specialists, geneticists, neurologists, nurses, and pharmacists, according to the statement.
Women with preexisting CVD should receive counseling about maternal and fetal risks before conception, if possible, to involve the women in shared decision-making and to develop strategies for each stage of pregnancy and delivery, Dr. Mehta and associates said. Such counseling should include a review of all medications and assessment of risk factors.
However, some women present already in the early stages of pregnancy even with severe conditions such as pulmonary arterial hypertension, severe ventricular dysfunction, severe left-sided heart obstruction, and significant aortic dilatation with underlying connective tissue disease. Women with these conditions often are counseled to avoid pregnancy, but if they already are pregnant, a high-risk cardio-obstetrics team will need to work together to discover the best strategies going forward to mitigate risk, Dr. Mehta and associates said.
Common CVD conditions that affect pregnancy include hypertensive disorders, notably preeclampsia, defined as systolic blood pressure greater than 140 mm Hg or diastolic blood pressure greater than 90 mm Hg in women after 20 weeks of gestation whose blood pressure was normal prior to pregnancy. A management strategy to reduce the risk of pregnancy-related complications from hypertension includes healthy lifestyle behaviors such as exercise, nutrition, and smoking cessation, according to the statement. However, patients with severe hypertension may require intravenous labetalol or hydralazine. The statement gives more information about handling preeclampsia with pulmonary edema, and prevention of eclampsia and treatment of seizures.
It is important to recognize that severe hypertension or superimposed preeclampsia may occur for the first time post partum. Early ambulatory visits in the first 1-2 weeks are sensible. Medications may be needed to keep a systolic blood pressure not higher than 150 mm Hg and a diastolic blood pressure not higher than 100 mm Hg, Dr. Mehta and associates said.
According to the statement, severe hypertriglyceridemia and familial hypercholesterolemia are the two most common conditions in which lipids should be addressed during pregnancy, with consideration of the fetal risks associated with certain medications.
“Statins are contraindicated during pregnancy, and all women who are on any lipid-lowering agents should review with their physician the safety of treatment during pregnancy and whether to discontinue treatment before pregnancy,” according to the statement. A heart-healthy lifestyle can help improve lipid profiles in all pregnant patients, Dr. Mehta and associates said. Patients with extremely high triglycerides above 500 mg/dL are at risk of pancreatitis and “may benefit from pharmacological agents (omega-3 fatty acids with or without fenofibrate or gemfibrozil) during the second trimester,” they noted. Pregnant women with familial hypercholesterolemia might take bile acid sequestrants, or as a last resort, low-density lipoprotein apheresis.
Other conditions calling for a multidisciplinary cardio-obstetric approach include preexisting coronary artery disease, cardiomyopathies, arrhythmias, valvular heart disease, cerebrovascular disease, and deep venous thrombosis, according to the statement, which provides information about the risks, diagnosis, and management.
When it is time for delivery, spontaneous labor and vaginal birth are preferable for most women with heart disease, as cesarean delivery is associated with increased risk of infection, thrombotic complications, and blood loss, according to the statement.
Women with CVD and associated complications will require “specialized long-term cardiovascular follow-up,” Dr. Mehta and associates said. “In women with a high-risk pregnancy, a cardio-obstetrics team is essential to prevent maternal morbidity and mortality during the length of the pregnancy and post partum.”
“The release of this document demonstrates the AHA’s recognition of the importance of CVD in pregnancy-related death and their commitment to education and ensuring best practices in this field,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and chief medical officer at Texas Children’s Health Plan, Bellaire.
“I think one of the most important outcomes from the release of this scientific statement from AHA will be increased implementation of cardio-obstetrics teams,” she said in an interview.
“In the United States, cardiovascular disease and cardiomyopathy together are now the leading cause of death in pregnancy and the postpartum period, and constitute 26.5% of pregnancy-related deaths, with higher rates of mortality among women of color and women with lower incomes,” she said. “The rising trend in cardiovascular-related maternal deaths appears to be due to acquired, not congenital, heart disease.”
During her tenure as president of ACOG, Dr. Hollier convened a task force on cardiovascular disease in pregnancy that developed guidance that outlines screening, diagnosis, and management of CVD for women from prepregnancy through post partum.
Dr. Hollier noted that COVID-19 emphasizes racial disparities for maternal mortality.
“Pregnant patients with comorbidities, like heart conditions, may be at increased risk for severe illness from COVID-19 – consistent with the general population with similar comorbidities,” she said. “And as we know, black women’s risk of dying from CVD-related pregnancy complications is 3.4 times higher than that of white women. During the COVID-19 pandemic, we are seeing these racial health disparities exacerbated.”
However, any pregnant patients should not hesitate to communicate with their health care providers despite the pandemic situation, Dr. Hollier emphasized. “Communication between a patient and her ob.gyn., cardiologist, or other clinician is even more critical now during the COVID-19 pandemic. We’re hearing reports that patients who are experiencing symptoms or those with known cardiac conditions are avoiding the hospital and delaying or not seeking necessary treatment. This has the very real possibility of worsening the devastating maternal mortality crisis that we’re already experiencing in this country.”
To help overcome barriers to treatment, “collaboration between ob.gyns. and cardiologists, such as the cardio-obstetrics team or pregnancy heart team, is critical,” said Dr. Hollier. “These collaborative teams with a multidisciplinary approach can prospectively reduce the communication gaps across specialties when patients are seen separately. They can also improve the communication during care transitions such as between outpatient and inpatient care.
“In reviews of maternal deaths, we have found that there are often delays in diagnosis of heart conditions during and after pregnancy,” Dr. Hollier added. “Most maternal deaths from CVD are due to either undiagnosed cardiovascular disease or new-onset cardiomyopathy. ACOG recommends that all women be assessed for cardiovascular disease in the antepartum and postpartum periods using a recently developed algorithm,” she said. “Women who have known CVD and women who have concerning symptoms should have a consultation with this team. With increased awareness and screening, women can receive the additional care that they need.
“Because management of cardiac conditions in pregnancy is so complex, it is important to ensure that women receive care with teams and in facilities that have appropriate resources,” explained Dr. Hollier. “Women with known heart disease should see a cardiologist prior to pregnancy and receive prepregnancy counseling,” as noted in the AHA statement. “Patients determined to have moderate and high-risk CVD should be managed during pregnancy, delivery, and post partum in a medical center that is able to provide a higher level of care, including a cardio-obstetrics team.”
Early recognition of cardiovascular conditions is essential to help manage care and reduce risks to mother and baby, said Dr. Hollier. “Identification before a woman becomes pregnant means the patient’s care can be properly managed throughout the pregnancy and a detailed delivery plan can be developed through shared decision making between the patient and provider. We must think of heart disease as a possibility in every pregnant or postpartum patient we see to detect and treat at-risk mothers,” she said.
Additional research should focus on identifying risk factors prior to pregnancy, said Dr. Hollier. “There are often delays in recognizing symptoms during pregnancy and post partum, particularly for black women. We need data to understand which protocols are best to identify heart disease,”
Dr. Hollier had no financial conflicts to disclose. The authors of the AHA statement had no financial conflicts to disclose. The scientific statement was produced on behalf of the American Heart Association Council on Clinical Cardiology; Council on Atherosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular and Stroke Nursing; and the Stroke Council.
SOURCE: Mehta LS et al. Circulation. 2020 May 4. doi: 10.1161/CIR.0000000000000772.
according to a statement from the American Heart Association.
Cardiovascular disease (CVD) remains the leading cause of pregnancy-related mortality in the United States, and accounted for approximately 17 deaths per 100,000 live births in 2015, wrote Laxmi S. Mehta, MD, of The Ohio State University, Columbus, and colleagues.
Ideally, a woman with CVD at the time of pregnancy should be managed by a multidisciplinary cardio-obstetrics team that can assess cardiovascular risk, obstetric risk, and fetal risk throughout pregnancy, delivery, and up to a year post partum. The team should develop a shared strategy to promote best outcomes, according to the statement. The cardio-obstetrics team may include obstetricians, cardiologists, anesthesiologists, maternal-fetal medicine specialists, geneticists, neurologists, nurses, and pharmacists, according to the statement.
Women with preexisting CVD should receive counseling about maternal and fetal risks before conception, if possible, to involve the women in shared decision-making and to develop strategies for each stage of pregnancy and delivery, Dr. Mehta and associates said. Such counseling should include a review of all medications and assessment of risk factors.
However, some women present already in the early stages of pregnancy even with severe conditions such as pulmonary arterial hypertension, severe ventricular dysfunction, severe left-sided heart obstruction, and significant aortic dilatation with underlying connective tissue disease. Women with these conditions often are counseled to avoid pregnancy, but if they already are pregnant, a high-risk cardio-obstetrics team will need to work together to discover the best strategies going forward to mitigate risk, Dr. Mehta and associates said.
Common CVD conditions that affect pregnancy include hypertensive disorders, notably preeclampsia, defined as systolic blood pressure greater than 140 mm Hg or diastolic blood pressure greater than 90 mm Hg in women after 20 weeks of gestation whose blood pressure was normal prior to pregnancy. A management strategy to reduce the risk of pregnancy-related complications from hypertension includes healthy lifestyle behaviors such as exercise, nutrition, and smoking cessation, according to the statement. However, patients with severe hypertension may require intravenous labetalol or hydralazine. The statement gives more information about handling preeclampsia with pulmonary edema, and prevention of eclampsia and treatment of seizures.
It is important to recognize that severe hypertension or superimposed preeclampsia may occur for the first time post partum. Early ambulatory visits in the first 1-2 weeks are sensible. Medications may be needed to keep a systolic blood pressure not higher than 150 mm Hg and a diastolic blood pressure not higher than 100 mm Hg, Dr. Mehta and associates said.
According to the statement, severe hypertriglyceridemia and familial hypercholesterolemia are the two most common conditions in which lipids should be addressed during pregnancy, with consideration of the fetal risks associated with certain medications.
“Statins are contraindicated during pregnancy, and all women who are on any lipid-lowering agents should review with their physician the safety of treatment during pregnancy and whether to discontinue treatment before pregnancy,” according to the statement. A heart-healthy lifestyle can help improve lipid profiles in all pregnant patients, Dr. Mehta and associates said. Patients with extremely high triglycerides above 500 mg/dL are at risk of pancreatitis and “may benefit from pharmacological agents (omega-3 fatty acids with or without fenofibrate or gemfibrozil) during the second trimester,” they noted. Pregnant women with familial hypercholesterolemia might take bile acid sequestrants, or as a last resort, low-density lipoprotein apheresis.
Other conditions calling for a multidisciplinary cardio-obstetric approach include preexisting coronary artery disease, cardiomyopathies, arrhythmias, valvular heart disease, cerebrovascular disease, and deep venous thrombosis, according to the statement, which provides information about the risks, diagnosis, and management.
When it is time for delivery, spontaneous labor and vaginal birth are preferable for most women with heart disease, as cesarean delivery is associated with increased risk of infection, thrombotic complications, and blood loss, according to the statement.
Women with CVD and associated complications will require “specialized long-term cardiovascular follow-up,” Dr. Mehta and associates said. “In women with a high-risk pregnancy, a cardio-obstetrics team is essential to prevent maternal morbidity and mortality during the length of the pregnancy and post partum.”
“The release of this document demonstrates the AHA’s recognition of the importance of CVD in pregnancy-related death and their commitment to education and ensuring best practices in this field,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and chief medical officer at Texas Children’s Health Plan, Bellaire.
“I think one of the most important outcomes from the release of this scientific statement from AHA will be increased implementation of cardio-obstetrics teams,” she said in an interview.
“In the United States, cardiovascular disease and cardiomyopathy together are now the leading cause of death in pregnancy and the postpartum period, and constitute 26.5% of pregnancy-related deaths, with higher rates of mortality among women of color and women with lower incomes,” she said. “The rising trend in cardiovascular-related maternal deaths appears to be due to acquired, not congenital, heart disease.”
During her tenure as president of ACOG, Dr. Hollier convened a task force on cardiovascular disease in pregnancy that developed guidance that outlines screening, diagnosis, and management of CVD for women from prepregnancy through post partum.
Dr. Hollier noted that COVID-19 emphasizes racial disparities for maternal mortality.
“Pregnant patients with comorbidities, like heart conditions, may be at increased risk for severe illness from COVID-19 – consistent with the general population with similar comorbidities,” she said. “And as we know, black women’s risk of dying from CVD-related pregnancy complications is 3.4 times higher than that of white women. During the COVID-19 pandemic, we are seeing these racial health disparities exacerbated.”
However, any pregnant patients should not hesitate to communicate with their health care providers despite the pandemic situation, Dr. Hollier emphasized. “Communication between a patient and her ob.gyn., cardiologist, or other clinician is even more critical now during the COVID-19 pandemic. We’re hearing reports that patients who are experiencing symptoms or those with known cardiac conditions are avoiding the hospital and delaying or not seeking necessary treatment. This has the very real possibility of worsening the devastating maternal mortality crisis that we’re already experiencing in this country.”
To help overcome barriers to treatment, “collaboration between ob.gyns. and cardiologists, such as the cardio-obstetrics team or pregnancy heart team, is critical,” said Dr. Hollier. “These collaborative teams with a multidisciplinary approach can prospectively reduce the communication gaps across specialties when patients are seen separately. They can also improve the communication during care transitions such as between outpatient and inpatient care.
“In reviews of maternal deaths, we have found that there are often delays in diagnosis of heart conditions during and after pregnancy,” Dr. Hollier added. “Most maternal deaths from CVD are due to either undiagnosed cardiovascular disease or new-onset cardiomyopathy. ACOG recommends that all women be assessed for cardiovascular disease in the antepartum and postpartum periods using a recently developed algorithm,” she said. “Women who have known CVD and women who have concerning symptoms should have a consultation with this team. With increased awareness and screening, women can receive the additional care that they need.
“Because management of cardiac conditions in pregnancy is so complex, it is important to ensure that women receive care with teams and in facilities that have appropriate resources,” explained Dr. Hollier. “Women with known heart disease should see a cardiologist prior to pregnancy and receive prepregnancy counseling,” as noted in the AHA statement. “Patients determined to have moderate and high-risk CVD should be managed during pregnancy, delivery, and post partum in a medical center that is able to provide a higher level of care, including a cardio-obstetrics team.”
Early recognition of cardiovascular conditions is essential to help manage care and reduce risks to mother and baby, said Dr. Hollier. “Identification before a woman becomes pregnant means the patient’s care can be properly managed throughout the pregnancy and a detailed delivery plan can be developed through shared decision making between the patient and provider. We must think of heart disease as a possibility in every pregnant or postpartum patient we see to detect and treat at-risk mothers,” she said.
Additional research should focus on identifying risk factors prior to pregnancy, said Dr. Hollier. “There are often delays in recognizing symptoms during pregnancy and post partum, particularly for black women. We need data to understand which protocols are best to identify heart disease,”
Dr. Hollier had no financial conflicts to disclose. The authors of the AHA statement had no financial conflicts to disclose. The scientific statement was produced on behalf of the American Heart Association Council on Clinical Cardiology; Council on Atherosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular and Stroke Nursing; and the Stroke Council.
SOURCE: Mehta LS et al. Circulation. 2020 May 4. doi: 10.1161/CIR.0000000000000772.
according to a statement from the American Heart Association.
Cardiovascular disease (CVD) remains the leading cause of pregnancy-related mortality in the United States, and accounted for approximately 17 deaths per 100,000 live births in 2015, wrote Laxmi S. Mehta, MD, of The Ohio State University, Columbus, and colleagues.
Ideally, a woman with CVD at the time of pregnancy should be managed by a multidisciplinary cardio-obstetrics team that can assess cardiovascular risk, obstetric risk, and fetal risk throughout pregnancy, delivery, and up to a year post partum. The team should develop a shared strategy to promote best outcomes, according to the statement. The cardio-obstetrics team may include obstetricians, cardiologists, anesthesiologists, maternal-fetal medicine specialists, geneticists, neurologists, nurses, and pharmacists, according to the statement.
Women with preexisting CVD should receive counseling about maternal and fetal risks before conception, if possible, to involve the women in shared decision-making and to develop strategies for each stage of pregnancy and delivery, Dr. Mehta and associates said. Such counseling should include a review of all medications and assessment of risk factors.
However, some women present already in the early stages of pregnancy even with severe conditions such as pulmonary arterial hypertension, severe ventricular dysfunction, severe left-sided heart obstruction, and significant aortic dilatation with underlying connective tissue disease. Women with these conditions often are counseled to avoid pregnancy, but if they already are pregnant, a high-risk cardio-obstetrics team will need to work together to discover the best strategies going forward to mitigate risk, Dr. Mehta and associates said.
Common CVD conditions that affect pregnancy include hypertensive disorders, notably preeclampsia, defined as systolic blood pressure greater than 140 mm Hg or diastolic blood pressure greater than 90 mm Hg in women after 20 weeks of gestation whose blood pressure was normal prior to pregnancy. A management strategy to reduce the risk of pregnancy-related complications from hypertension includes healthy lifestyle behaviors such as exercise, nutrition, and smoking cessation, according to the statement. However, patients with severe hypertension may require intravenous labetalol or hydralazine. The statement gives more information about handling preeclampsia with pulmonary edema, and prevention of eclampsia and treatment of seizures.
It is important to recognize that severe hypertension or superimposed preeclampsia may occur for the first time post partum. Early ambulatory visits in the first 1-2 weeks are sensible. Medications may be needed to keep a systolic blood pressure not higher than 150 mm Hg and a diastolic blood pressure not higher than 100 mm Hg, Dr. Mehta and associates said.
According to the statement, severe hypertriglyceridemia and familial hypercholesterolemia are the two most common conditions in which lipids should be addressed during pregnancy, with consideration of the fetal risks associated with certain medications.
“Statins are contraindicated during pregnancy, and all women who are on any lipid-lowering agents should review with their physician the safety of treatment during pregnancy and whether to discontinue treatment before pregnancy,” according to the statement. A heart-healthy lifestyle can help improve lipid profiles in all pregnant patients, Dr. Mehta and associates said. Patients with extremely high triglycerides above 500 mg/dL are at risk of pancreatitis and “may benefit from pharmacological agents (omega-3 fatty acids with or without fenofibrate or gemfibrozil) during the second trimester,” they noted. Pregnant women with familial hypercholesterolemia might take bile acid sequestrants, or as a last resort, low-density lipoprotein apheresis.
Other conditions calling for a multidisciplinary cardio-obstetric approach include preexisting coronary artery disease, cardiomyopathies, arrhythmias, valvular heart disease, cerebrovascular disease, and deep venous thrombosis, according to the statement, which provides information about the risks, diagnosis, and management.
When it is time for delivery, spontaneous labor and vaginal birth are preferable for most women with heart disease, as cesarean delivery is associated with increased risk of infection, thrombotic complications, and blood loss, according to the statement.
Women with CVD and associated complications will require “specialized long-term cardiovascular follow-up,” Dr. Mehta and associates said. “In women with a high-risk pregnancy, a cardio-obstetrics team is essential to prevent maternal morbidity and mortality during the length of the pregnancy and post partum.”
“The release of this document demonstrates the AHA’s recognition of the importance of CVD in pregnancy-related death and their commitment to education and ensuring best practices in this field,” said Lisa M. Hollier, MD, past president of the American College of Obstetricians and Gynecologists and chief medical officer at Texas Children’s Health Plan, Bellaire.
“I think one of the most important outcomes from the release of this scientific statement from AHA will be increased implementation of cardio-obstetrics teams,” she said in an interview.
“In the United States, cardiovascular disease and cardiomyopathy together are now the leading cause of death in pregnancy and the postpartum period, and constitute 26.5% of pregnancy-related deaths, with higher rates of mortality among women of color and women with lower incomes,” she said. “The rising trend in cardiovascular-related maternal deaths appears to be due to acquired, not congenital, heart disease.”
During her tenure as president of ACOG, Dr. Hollier convened a task force on cardiovascular disease in pregnancy that developed guidance that outlines screening, diagnosis, and management of CVD for women from prepregnancy through post partum.
Dr. Hollier noted that COVID-19 emphasizes racial disparities for maternal mortality.
“Pregnant patients with comorbidities, like heart conditions, may be at increased risk for severe illness from COVID-19 – consistent with the general population with similar comorbidities,” she said. “And as we know, black women’s risk of dying from CVD-related pregnancy complications is 3.4 times higher than that of white women. During the COVID-19 pandemic, we are seeing these racial health disparities exacerbated.”
However, any pregnant patients should not hesitate to communicate with their health care providers despite the pandemic situation, Dr. Hollier emphasized. “Communication between a patient and her ob.gyn., cardiologist, or other clinician is even more critical now during the COVID-19 pandemic. We’re hearing reports that patients who are experiencing symptoms or those with known cardiac conditions are avoiding the hospital and delaying or not seeking necessary treatment. This has the very real possibility of worsening the devastating maternal mortality crisis that we’re already experiencing in this country.”
To help overcome barriers to treatment, “collaboration between ob.gyns. and cardiologists, such as the cardio-obstetrics team or pregnancy heart team, is critical,” said Dr. Hollier. “These collaborative teams with a multidisciplinary approach can prospectively reduce the communication gaps across specialties when patients are seen separately. They can also improve the communication during care transitions such as between outpatient and inpatient care.
“In reviews of maternal deaths, we have found that there are often delays in diagnosis of heart conditions during and after pregnancy,” Dr. Hollier added. “Most maternal deaths from CVD are due to either undiagnosed cardiovascular disease or new-onset cardiomyopathy. ACOG recommends that all women be assessed for cardiovascular disease in the antepartum and postpartum periods using a recently developed algorithm,” she said. “Women who have known CVD and women who have concerning symptoms should have a consultation with this team. With increased awareness and screening, women can receive the additional care that they need.
“Because management of cardiac conditions in pregnancy is so complex, it is important to ensure that women receive care with teams and in facilities that have appropriate resources,” explained Dr. Hollier. “Women with known heart disease should see a cardiologist prior to pregnancy and receive prepregnancy counseling,” as noted in the AHA statement. “Patients determined to have moderate and high-risk CVD should be managed during pregnancy, delivery, and post partum in a medical center that is able to provide a higher level of care, including a cardio-obstetrics team.”
Early recognition of cardiovascular conditions is essential to help manage care and reduce risks to mother and baby, said Dr. Hollier. “Identification before a woman becomes pregnant means the patient’s care can be properly managed throughout the pregnancy and a detailed delivery plan can be developed through shared decision making between the patient and provider. We must think of heart disease as a possibility in every pregnant or postpartum patient we see to detect and treat at-risk mothers,” she said.
Additional research should focus on identifying risk factors prior to pregnancy, said Dr. Hollier. “There are often delays in recognizing symptoms during pregnancy and post partum, particularly for black women. We need data to understand which protocols are best to identify heart disease,”
Dr. Hollier had no financial conflicts to disclose. The authors of the AHA statement had no financial conflicts to disclose. The scientific statement was produced on behalf of the American Heart Association Council on Clinical Cardiology; Council on Atherosclerosis, Thrombosis and Vascular Biology; Council on Cardiovascular and Stroke Nursing; and the Stroke Council.
SOURCE: Mehta LS et al. Circulation. 2020 May 4. doi: 10.1161/CIR.0000000000000772.
FROM CIRCULATION
Triage, L&D, postpartum care during the COVID-19 pandemic
The meteoric rise in the number of test-positive and clinical cases of COVID-19 because of infection with the SARS coronavirus (SARS-CoV-2) in states and cities across the United States has added urgency to the efforts to develop protocols for hospital triage, admission, labor and delivery management, and other aspects of obstetrical care.
Emerging data suggest that, while SARS-CoV-2 is less lethal overall than the severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) proved to be, it is significantly more contagious. Although a severe disease, the limited worldwide data so far available (as of early May) do not indicate that pregnant women are at greater risk of severe disease, compared with the general population. However, there remains a critical need for data on maternal and perinatal outcomes in women infected with SARS-CoV-2.
Multiple physiological changes in pregnancy, from reduced cell-based immune competence to changes in respiratory tract and pulmonary function – e.g., edema of the respiratory tract, increases in secretions and oxygen consumption, elevation of the diaphragm, and decrease in functional residual capacity – have historically contributed to worse obstetric outcomes in pregnant women who have had viral pneumonias. Furthermore, limited published experience with COVID-19 in China suggests worse perinatal outcomes in some affected pregnancies, including prematurity and perinatal death.
With evolution of the pandemic and accumulation of experience, it is expected that data-driven guidelines on assessment and management of infected pregnant women will contribute to improved maternal and perinatal outcomes. What is clear now, however, is that,
Here are my recommendations, based on a currently limited body of literature on COVID-19 and other communicable viral respiratory disorders, as well my experience in the greater Detroit area, a COVID-19 hot spot.
Preparing for hospital evaluation and admission
The obstetric triage or labor and delivery (L&D) unit should be notified prior to the arrival of a patient suspected of or known to be infected with the virus. This will minimize staff exposure and allow sufficient time to prepare appropriate accommodations, equipment, and supplies for the patient’s care. Hospital infection control should be promptly notified by L&D of the expected arrival of such a patient. Placement ideally should be in a negative-pressure room, which allows outside air to flow into the room but prevents contaminated air from escaping. In the absence of a negative-pressure room, an infection isolation area should be utilized.
The patient and one accompanying support individual should wear either medical-grade masks brought from home or supplied upon entry to the hospital or homemade masks or bandanas. This will reduce the risk of viral transmission to hospital workers and other individuals encountered in the hospital prior to arriving in L&D. An ideal setup is to have separate entry areas, access corridors, and elevators for patients known or suspected to have COVID-19 infection. The patient and visitor should be expeditiously escorted to the prepared area for evaluation. Patients who are not known or suspected to be infected ideally should be tested.
Screening of patients & support individuals
Proper screening of patients and support individuals is critical to protecting both patients and staff in the L&D unit. This should include an expanded questionnaire that asks about disturbances of smell and taste and GI symptoms like loss of appetite – not only the more commonly queried symptoms of fever, shortness of breath, coughing, and exposure to someone who may have been ill.
Recent studies regarding presenting symptoms cast significant doubt, in fact, on the validity of patients with “asymptomatic COVID-19.” Over 15% of patients with confirmed infection in one published case series had solely GI symptoms and almost all had some digestive symptoms, for example, and almost 90% in another study had absent or reduced sense of smell and/or taste.1,2 In fact, the use of the term “paucisymptomatic” rather than “asymptomatic” may be most appropriate.
Support individuals also should undergo temperature screening, ideally with laser noncontact thermometers on entry to the hospital or triage.
Visitor policy
The number of visitors/support individuals should be kept to a minimum to reduce transmission risk. The actual number will be determined by hospital or state policy, but up to one visitor in the labor room appears reasonable. Very strong individual justification should be required to exceed this threshold! The visitor should not only be screened for an expanded list of symptoms, but they also should be queried for underlying illnesses (e.g., diabetes, cardiovascular disease, significant lung disease, undergoing cancer therapy) as well as for age over 65 years, each of which increase the chances of severe COVID-19 disease should infection occur. The visitor should be informed of such risks and, especially when accompanying a patient with known or suspected COVID-19, provided the option of voluntarily revoking their visitor status. A visitor with known or suspected COVID-19 infection based on testing or screening should not be allowed into the L&D unit.
In addition, institutions may be considered to have obligations to the visitor/support person beyond screening. These include instructions in proper mask usage, hand washing, and limiting the touching of surfaces to lower infection risk.
“Visitor relays” where one visitor replaces another should be strongly discouraged. Visitors should similarly not be allowed to wander around the hospital (to use phones, for instance); transiting back and forth to obtain food and coffee should be kept to a strict minimum. For visitors accompanying COVID-19–-infected women, “visitor’s plates” provided by the hospital at reasonable cost is a much-preferred arrangement for obtaining meals during the course of the hospital stay. In addition, visitors should be sent out of the room during the performance of aerosolizing procedures.
Labor and delivery management
The successful management of patients with COVID-19 requires a rigorous infection control protocol informed by guidelines from national entities, such as the Centers for Disease Control and Prevention, the Society for Maternal-Fetal Medicine, and the American College of Obstetricians and Gynecologists, and by state health departments when available.
Strict limits on the number of obstetricians and other health care workers (HCWs) entering the patient’s room should be enforced and documented to minimize risk to the HCWs attending to patients who have a positive diagnosis or who are under investigation. Only in cases of demonstrable clinical benefit should repeat visits by the same or additional HCWs be permitted. Conventional and electronic tablets present an excellent opportunity for patient follow-up visits without room entry. In our institution, this has been successfully piloted in nonpregnant patients. Obstetricians and others caring for obstetrical patients – especially those who are infected or under investigation for infection – should always wear a properly fitted N95 mask.
Because patients with COVID-19 may have or go on to develop a constellation of organ abnormalities (e.g., cardiovascular, renal, pulmonary), it is vital that a standardized panel of baseline laboratory studies be developed for pregnant patients. This will minimize the need for repeated blood draws and other testing which may increase HCW exposure.
A negative screen based on nonreport of symptoms, lack of temperature elevation, and reported nonexposure to individuals with COVID-19 symptoms still has limitations in terms of disease detection. A recent report from a tertiary care hospital in New York City found that close to one-third of pregnant patients with confirmed COVID-19 admitted over a 2-week period had no viral symptoms or instructive history on initial admission.3 This is consistent with our clinical experience. Most importantly, therefore, routine quantitative reverse transcription polymerase chain reaction testing should be performed on all patients admitted to the L&D unit.
Given the reported variability in the accuracy of polymerase chain reaction testing induced by variable effectiveness of sampling techniques, stage of infection, and inherent test accuracy issues, symptomatic patients with a negative test should first obtain clearance from infectious disease specialists before isolation precautions are discontinued. Repeat testing in 24 hours, including testing of multiple sites, may subsequently yield a positive result in persistently symptomatic patients.
Intrapartum management
As much as possible, standard obstetric indications should guide the timing and route of delivery. In the case of a COVID-19–positive patient or a patient under investigation, nonobstetric factors may bear heavily on decision making, and management flexibility is of great value. For example, in cases of severe or critical disease status, evidence suggests that early delivery regardless of gestational age can improve maternal oxygenation; this supports the liberal use of C-sections in these circumstances. In addition, shortening labor length as well as duration of hospitalization may be expected to reduce the risk of transmission to HCWs, other staff, and other patients.
High rates of cesarean delivery unsurprisingly have been reported thus far: One review of 108 case reports and series of test-positive COVID-19 pregnancies found a 92% C-section rate, and another review and meta-analysis of studies of SARS, MERS, and COVID-19 during pregnancy similarly found that the majority of patients – 84% across all coronavirus infections and 91% in COVID-19 pregnancies – were delivered by C-section.4,5 Given these high rates of cesarean deliveries, the early placement of neuraxial anesthesia while the patient is stable appears to be prudent and obviates the need for intubation, the latter of which is associated with increased aerosol generation and increased virus transmission risk.
Strict protocols for the optimal protection of staff should be observed, including proper personal protective equipment (PPE) protection. Protocols have been detailed in various guidelines and publications; they include the wearing of shoe covers, gowns, N95 masks, goggles, face shields, and two layers of gloves.
For institutions that currently do not offer routine COVID-19 testing to pregnant patients – especially those in areas of outbreaks – N95 masks and eye protection should still be provided to all HCWs involved in the intrapartum management of untested asymptomatic patients, particularly those in the active phase of labor. This protection is justified given the limitations of symptom- and history-based screening and the not-uncommon experience of the patient with a negative screen who subsequently develops the clinical syndrome.
Obstetric management of labor requires close patient contact that potentially elevates the risk of contamination and infection. During the active stage of labor, patient shouting, rapid mouth breathing, and other behaviors inherent to labor all increase the risk of aerosolization of oronasal secretions. In addition, nasal-prong oxygen administration is believed to independently increase the risk of aerosolization of secretions. The casual practice of nasal oxygen application should thus be discontinued and, where felt to be absolutely necessary, a mask should be worn on top of the prongs.
Regarding operative delivery, each participating obstetric surgeon should observe guidelines and recommendations of governing national organizations and professional groups – including the American College of Surgeons – regarding the safe conduct of operations on patients with COVID-19. Written guidelines should be tailored as needed to the performance of C-sections and readily available in L&D. Drills and simulations are generally valuable, and expertise and support should always be available in the labor room to assist with donning and doffing of PPE.
Postpartum care
Expeditious separation of the COVID-19–positive mother from her infant is recommended, including avoidance of delayed cord clamping because of insufficient evidence of benefit to the infant. Insufficient evidence exists to support vertical transmission, but the possibility of maternal-infant transmission is clinically accepted based on small case reports of infection in a neonate at 30 hours of life and in infants of mothers with suspected or confirmed COVID-19.6,7 Accordingly, it is recommended that the benefit of early infant separation should be discussed with the mother. If approved, the infant should be kept in a separate isolation area and observed.
There is no evidence of breast milk transmission of the virus. For those electing to breastfeed, the patient should be provided with a breast pump to express and store the milk for subsequent bottle feeding. For mothers who elect to room in with the infant, a separation distance of 6 feet is recommended with an intervening barrier curtain. For COVID-19–positive mothers who elect breastfeeding, meticulous hand and face washing, continuous wearing of a mask, and cleansing of the breast prior to feeding needs to be maintained.
Restrictive visiting policies of no more than one visitor should be maintained. For severely or critically ill patients with COVID-19, it has been suggested that no visitors be allowed. As with other hospitalizations of COVID-19 patients, the HCW contact should be kept at a justifiable minimum to reduce the risk of transmission.
Protecting the obstetrician and other HCWs
Protecting the health of obstetricians and other HCWs is central to any successful strategy to fight the COVID-19 epidemic. For the individual obstetrician, careful attention to national and local hospital guidelines is required as these are rapidly evolving.
Physicians and their leadership must maintain an ongoing dialogue with hospital leadership to continually upgrade and optimize infection prevention and control measures, and to uphold best practices. The experience in Wuhan, China, illustrates the effectiveness of the proper use of PPE along with population control measures to reduce infections in HCWs. Prior to understanding the mechanism of virus transmission and using protective equipment, infection rates of 3%-29% were reported among HCWs. With the meticulous utilization of mitigation strategies and population control measures – including consistent use of PPE – the rate of infection of HCWs reportedly fell to zero.
In outpatient offices, all staff and HCWs should wear masks at all times and engage in social distancing and in frequent hand sanitization. Patients should be strongly encouraged to wear masks during office visits and on all other occasions when they will be in physical proximity to other individuals outside of the home.
Reports from epidemic areas describe transmission from household sources as a significant cause of HCW infection. The information emphasizes the need for ongoing vigilance and attention to sanitization measures even when at home with one’s family. An additional benefit is reduced risk of transmission from HCWs to family members.
Dr. Bahado-Singh is professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System.
References
1. Luo S et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.043.
2. Lechien JR et al. Eur Arch Otorhinolaryngol. 2020 Apr 6. doi: 10.1007/s00405-020-05965-1.
3. Breslin N et al. Am J Obstet Gynecol MFM. 2020 Apr 9. doi: 10.1016/j.ajogmf.2020.100118.
4. Zaigham M, Andersson O. Acta Obstet Gynecol Scand. 2020 Apr 7. doi: 10.1111/aogs.13867.
5. Di Mascio D et al. Am J Obstet Gynecol MFM. 2020 Mar 25. doi: 10.1016/j.ajogmf.2020.100107.
6. Ital J. Pediatr 2020;46(1) doi: 10.1186/s13052-020-0820-x.
7. Int J Gynaecol Obstet. 2020;149(2):130-6.
*This article was updated 5/6/2020.
The meteoric rise in the number of test-positive and clinical cases of COVID-19 because of infection with the SARS coronavirus (SARS-CoV-2) in states and cities across the United States has added urgency to the efforts to develop protocols for hospital triage, admission, labor and delivery management, and other aspects of obstetrical care.
Emerging data suggest that, while SARS-CoV-2 is less lethal overall than the severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) proved to be, it is significantly more contagious. Although a severe disease, the limited worldwide data so far available (as of early May) do not indicate that pregnant women are at greater risk of severe disease, compared with the general population. However, there remains a critical need for data on maternal and perinatal outcomes in women infected with SARS-CoV-2.
Multiple physiological changes in pregnancy, from reduced cell-based immune competence to changes in respiratory tract and pulmonary function – e.g., edema of the respiratory tract, increases in secretions and oxygen consumption, elevation of the diaphragm, and decrease in functional residual capacity – have historically contributed to worse obstetric outcomes in pregnant women who have had viral pneumonias. Furthermore, limited published experience with COVID-19 in China suggests worse perinatal outcomes in some affected pregnancies, including prematurity and perinatal death.
With evolution of the pandemic and accumulation of experience, it is expected that data-driven guidelines on assessment and management of infected pregnant women will contribute to improved maternal and perinatal outcomes. What is clear now, however, is that,
Here are my recommendations, based on a currently limited body of literature on COVID-19 and other communicable viral respiratory disorders, as well my experience in the greater Detroit area, a COVID-19 hot spot.
Preparing for hospital evaluation and admission
The obstetric triage or labor and delivery (L&D) unit should be notified prior to the arrival of a patient suspected of or known to be infected with the virus. This will minimize staff exposure and allow sufficient time to prepare appropriate accommodations, equipment, and supplies for the patient’s care. Hospital infection control should be promptly notified by L&D of the expected arrival of such a patient. Placement ideally should be in a negative-pressure room, which allows outside air to flow into the room but prevents contaminated air from escaping. In the absence of a negative-pressure room, an infection isolation area should be utilized.
The patient and one accompanying support individual should wear either medical-grade masks brought from home or supplied upon entry to the hospital or homemade masks or bandanas. This will reduce the risk of viral transmission to hospital workers and other individuals encountered in the hospital prior to arriving in L&D. An ideal setup is to have separate entry areas, access corridors, and elevators for patients known or suspected to have COVID-19 infection. The patient and visitor should be expeditiously escorted to the prepared area for evaluation. Patients who are not known or suspected to be infected ideally should be tested.
Screening of patients & support individuals
Proper screening of patients and support individuals is critical to protecting both patients and staff in the L&D unit. This should include an expanded questionnaire that asks about disturbances of smell and taste and GI symptoms like loss of appetite – not only the more commonly queried symptoms of fever, shortness of breath, coughing, and exposure to someone who may have been ill.
Recent studies regarding presenting symptoms cast significant doubt, in fact, on the validity of patients with “asymptomatic COVID-19.” Over 15% of patients with confirmed infection in one published case series had solely GI symptoms and almost all had some digestive symptoms, for example, and almost 90% in another study had absent or reduced sense of smell and/or taste.1,2 In fact, the use of the term “paucisymptomatic” rather than “asymptomatic” may be most appropriate.
Support individuals also should undergo temperature screening, ideally with laser noncontact thermometers on entry to the hospital or triage.
Visitor policy
The number of visitors/support individuals should be kept to a minimum to reduce transmission risk. The actual number will be determined by hospital or state policy, but up to one visitor in the labor room appears reasonable. Very strong individual justification should be required to exceed this threshold! The visitor should not only be screened for an expanded list of symptoms, but they also should be queried for underlying illnesses (e.g., diabetes, cardiovascular disease, significant lung disease, undergoing cancer therapy) as well as for age over 65 years, each of which increase the chances of severe COVID-19 disease should infection occur. The visitor should be informed of such risks and, especially when accompanying a patient with known or suspected COVID-19, provided the option of voluntarily revoking their visitor status. A visitor with known or suspected COVID-19 infection based on testing or screening should not be allowed into the L&D unit.
In addition, institutions may be considered to have obligations to the visitor/support person beyond screening. These include instructions in proper mask usage, hand washing, and limiting the touching of surfaces to lower infection risk.
“Visitor relays” where one visitor replaces another should be strongly discouraged. Visitors should similarly not be allowed to wander around the hospital (to use phones, for instance); transiting back and forth to obtain food and coffee should be kept to a strict minimum. For visitors accompanying COVID-19–-infected women, “visitor’s plates” provided by the hospital at reasonable cost is a much-preferred arrangement for obtaining meals during the course of the hospital stay. In addition, visitors should be sent out of the room during the performance of aerosolizing procedures.
Labor and delivery management
The successful management of patients with COVID-19 requires a rigorous infection control protocol informed by guidelines from national entities, such as the Centers for Disease Control and Prevention, the Society for Maternal-Fetal Medicine, and the American College of Obstetricians and Gynecologists, and by state health departments when available.
Strict limits on the number of obstetricians and other health care workers (HCWs) entering the patient’s room should be enforced and documented to minimize risk to the HCWs attending to patients who have a positive diagnosis or who are under investigation. Only in cases of demonstrable clinical benefit should repeat visits by the same or additional HCWs be permitted. Conventional and electronic tablets present an excellent opportunity for patient follow-up visits without room entry. In our institution, this has been successfully piloted in nonpregnant patients. Obstetricians and others caring for obstetrical patients – especially those who are infected or under investigation for infection – should always wear a properly fitted N95 mask.
Because patients with COVID-19 may have or go on to develop a constellation of organ abnormalities (e.g., cardiovascular, renal, pulmonary), it is vital that a standardized panel of baseline laboratory studies be developed for pregnant patients. This will minimize the need for repeated blood draws and other testing which may increase HCW exposure.
A negative screen based on nonreport of symptoms, lack of temperature elevation, and reported nonexposure to individuals with COVID-19 symptoms still has limitations in terms of disease detection. A recent report from a tertiary care hospital in New York City found that close to one-third of pregnant patients with confirmed COVID-19 admitted over a 2-week period had no viral symptoms or instructive history on initial admission.3 This is consistent with our clinical experience. Most importantly, therefore, routine quantitative reverse transcription polymerase chain reaction testing should be performed on all patients admitted to the L&D unit.
Given the reported variability in the accuracy of polymerase chain reaction testing induced by variable effectiveness of sampling techniques, stage of infection, and inherent test accuracy issues, symptomatic patients with a negative test should first obtain clearance from infectious disease specialists before isolation precautions are discontinued. Repeat testing in 24 hours, including testing of multiple sites, may subsequently yield a positive result in persistently symptomatic patients.
Intrapartum management
As much as possible, standard obstetric indications should guide the timing and route of delivery. In the case of a COVID-19–positive patient or a patient under investigation, nonobstetric factors may bear heavily on decision making, and management flexibility is of great value. For example, in cases of severe or critical disease status, evidence suggests that early delivery regardless of gestational age can improve maternal oxygenation; this supports the liberal use of C-sections in these circumstances. In addition, shortening labor length as well as duration of hospitalization may be expected to reduce the risk of transmission to HCWs, other staff, and other patients.
High rates of cesarean delivery unsurprisingly have been reported thus far: One review of 108 case reports and series of test-positive COVID-19 pregnancies found a 92% C-section rate, and another review and meta-analysis of studies of SARS, MERS, and COVID-19 during pregnancy similarly found that the majority of patients – 84% across all coronavirus infections and 91% in COVID-19 pregnancies – were delivered by C-section.4,5 Given these high rates of cesarean deliveries, the early placement of neuraxial anesthesia while the patient is stable appears to be prudent and obviates the need for intubation, the latter of which is associated with increased aerosol generation and increased virus transmission risk.
Strict protocols for the optimal protection of staff should be observed, including proper personal protective equipment (PPE) protection. Protocols have been detailed in various guidelines and publications; they include the wearing of shoe covers, gowns, N95 masks, goggles, face shields, and two layers of gloves.
For institutions that currently do not offer routine COVID-19 testing to pregnant patients – especially those in areas of outbreaks – N95 masks and eye protection should still be provided to all HCWs involved in the intrapartum management of untested asymptomatic patients, particularly those in the active phase of labor. This protection is justified given the limitations of symptom- and history-based screening and the not-uncommon experience of the patient with a negative screen who subsequently develops the clinical syndrome.
Obstetric management of labor requires close patient contact that potentially elevates the risk of contamination and infection. During the active stage of labor, patient shouting, rapid mouth breathing, and other behaviors inherent to labor all increase the risk of aerosolization of oronasal secretions. In addition, nasal-prong oxygen administration is believed to independently increase the risk of aerosolization of secretions. The casual practice of nasal oxygen application should thus be discontinued and, where felt to be absolutely necessary, a mask should be worn on top of the prongs.
Regarding operative delivery, each participating obstetric surgeon should observe guidelines and recommendations of governing national organizations and professional groups – including the American College of Surgeons – regarding the safe conduct of operations on patients with COVID-19. Written guidelines should be tailored as needed to the performance of C-sections and readily available in L&D. Drills and simulations are generally valuable, and expertise and support should always be available in the labor room to assist with donning and doffing of PPE.
Postpartum care
Expeditious separation of the COVID-19–positive mother from her infant is recommended, including avoidance of delayed cord clamping because of insufficient evidence of benefit to the infant. Insufficient evidence exists to support vertical transmission, but the possibility of maternal-infant transmission is clinically accepted based on small case reports of infection in a neonate at 30 hours of life and in infants of mothers with suspected or confirmed COVID-19.6,7 Accordingly, it is recommended that the benefit of early infant separation should be discussed with the mother. If approved, the infant should be kept in a separate isolation area and observed.
There is no evidence of breast milk transmission of the virus. For those electing to breastfeed, the patient should be provided with a breast pump to express and store the milk for subsequent bottle feeding. For mothers who elect to room in with the infant, a separation distance of 6 feet is recommended with an intervening barrier curtain. For COVID-19–positive mothers who elect breastfeeding, meticulous hand and face washing, continuous wearing of a mask, and cleansing of the breast prior to feeding needs to be maintained.
Restrictive visiting policies of no more than one visitor should be maintained. For severely or critically ill patients with COVID-19, it has been suggested that no visitors be allowed. As with other hospitalizations of COVID-19 patients, the HCW contact should be kept at a justifiable minimum to reduce the risk of transmission.
Protecting the obstetrician and other HCWs
Protecting the health of obstetricians and other HCWs is central to any successful strategy to fight the COVID-19 epidemic. For the individual obstetrician, careful attention to national and local hospital guidelines is required as these are rapidly evolving.
Physicians and their leadership must maintain an ongoing dialogue with hospital leadership to continually upgrade and optimize infection prevention and control measures, and to uphold best practices. The experience in Wuhan, China, illustrates the effectiveness of the proper use of PPE along with population control measures to reduce infections in HCWs. Prior to understanding the mechanism of virus transmission and using protective equipment, infection rates of 3%-29% were reported among HCWs. With the meticulous utilization of mitigation strategies and population control measures – including consistent use of PPE – the rate of infection of HCWs reportedly fell to zero.
In outpatient offices, all staff and HCWs should wear masks at all times and engage in social distancing and in frequent hand sanitization. Patients should be strongly encouraged to wear masks during office visits and on all other occasions when they will be in physical proximity to other individuals outside of the home.
Reports from epidemic areas describe transmission from household sources as a significant cause of HCW infection. The information emphasizes the need for ongoing vigilance and attention to sanitization measures even when at home with one’s family. An additional benefit is reduced risk of transmission from HCWs to family members.
Dr. Bahado-Singh is professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System.
References
1. Luo S et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.043.
2. Lechien JR et al. Eur Arch Otorhinolaryngol. 2020 Apr 6. doi: 10.1007/s00405-020-05965-1.
3. Breslin N et al. Am J Obstet Gynecol MFM. 2020 Apr 9. doi: 10.1016/j.ajogmf.2020.100118.
4. Zaigham M, Andersson O. Acta Obstet Gynecol Scand. 2020 Apr 7. doi: 10.1111/aogs.13867.
5. Di Mascio D et al. Am J Obstet Gynecol MFM. 2020 Mar 25. doi: 10.1016/j.ajogmf.2020.100107.
6. Ital J. Pediatr 2020;46(1) doi: 10.1186/s13052-020-0820-x.
7. Int J Gynaecol Obstet. 2020;149(2):130-6.
*This article was updated 5/6/2020.
The meteoric rise in the number of test-positive and clinical cases of COVID-19 because of infection with the SARS coronavirus (SARS-CoV-2) in states and cities across the United States has added urgency to the efforts to develop protocols for hospital triage, admission, labor and delivery management, and other aspects of obstetrical care.
Emerging data suggest that, while SARS-CoV-2 is less lethal overall than the severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) proved to be, it is significantly more contagious. Although a severe disease, the limited worldwide data so far available (as of early May) do not indicate that pregnant women are at greater risk of severe disease, compared with the general population. However, there remains a critical need for data on maternal and perinatal outcomes in women infected with SARS-CoV-2.
Multiple physiological changes in pregnancy, from reduced cell-based immune competence to changes in respiratory tract and pulmonary function – e.g., edema of the respiratory tract, increases in secretions and oxygen consumption, elevation of the diaphragm, and decrease in functional residual capacity – have historically contributed to worse obstetric outcomes in pregnant women who have had viral pneumonias. Furthermore, limited published experience with COVID-19 in China suggests worse perinatal outcomes in some affected pregnancies, including prematurity and perinatal death.
With evolution of the pandemic and accumulation of experience, it is expected that data-driven guidelines on assessment and management of infected pregnant women will contribute to improved maternal and perinatal outcomes. What is clear now, however, is that,
Here are my recommendations, based on a currently limited body of literature on COVID-19 and other communicable viral respiratory disorders, as well my experience in the greater Detroit area, a COVID-19 hot spot.
Preparing for hospital evaluation and admission
The obstetric triage or labor and delivery (L&D) unit should be notified prior to the arrival of a patient suspected of or known to be infected with the virus. This will minimize staff exposure and allow sufficient time to prepare appropriate accommodations, equipment, and supplies for the patient’s care. Hospital infection control should be promptly notified by L&D of the expected arrival of such a patient. Placement ideally should be in a negative-pressure room, which allows outside air to flow into the room but prevents contaminated air from escaping. In the absence of a negative-pressure room, an infection isolation area should be utilized.
The patient and one accompanying support individual should wear either medical-grade masks brought from home or supplied upon entry to the hospital or homemade masks or bandanas. This will reduce the risk of viral transmission to hospital workers and other individuals encountered in the hospital prior to arriving in L&D. An ideal setup is to have separate entry areas, access corridors, and elevators for patients known or suspected to have COVID-19 infection. The patient and visitor should be expeditiously escorted to the prepared area for evaluation. Patients who are not known or suspected to be infected ideally should be tested.
Screening of patients & support individuals
Proper screening of patients and support individuals is critical to protecting both patients and staff in the L&D unit. This should include an expanded questionnaire that asks about disturbances of smell and taste and GI symptoms like loss of appetite – not only the more commonly queried symptoms of fever, shortness of breath, coughing, and exposure to someone who may have been ill.
Recent studies regarding presenting symptoms cast significant doubt, in fact, on the validity of patients with “asymptomatic COVID-19.” Over 15% of patients with confirmed infection in one published case series had solely GI symptoms and almost all had some digestive symptoms, for example, and almost 90% in another study had absent or reduced sense of smell and/or taste.1,2 In fact, the use of the term “paucisymptomatic” rather than “asymptomatic” may be most appropriate.
Support individuals also should undergo temperature screening, ideally with laser noncontact thermometers on entry to the hospital or triage.
Visitor policy
The number of visitors/support individuals should be kept to a minimum to reduce transmission risk. The actual number will be determined by hospital or state policy, but up to one visitor in the labor room appears reasonable. Very strong individual justification should be required to exceed this threshold! The visitor should not only be screened for an expanded list of symptoms, but they also should be queried for underlying illnesses (e.g., diabetes, cardiovascular disease, significant lung disease, undergoing cancer therapy) as well as for age over 65 years, each of which increase the chances of severe COVID-19 disease should infection occur. The visitor should be informed of such risks and, especially when accompanying a patient with known or suspected COVID-19, provided the option of voluntarily revoking their visitor status. A visitor with known or suspected COVID-19 infection based on testing or screening should not be allowed into the L&D unit.
In addition, institutions may be considered to have obligations to the visitor/support person beyond screening. These include instructions in proper mask usage, hand washing, and limiting the touching of surfaces to lower infection risk.
“Visitor relays” where one visitor replaces another should be strongly discouraged. Visitors should similarly not be allowed to wander around the hospital (to use phones, for instance); transiting back and forth to obtain food and coffee should be kept to a strict minimum. For visitors accompanying COVID-19–-infected women, “visitor’s plates” provided by the hospital at reasonable cost is a much-preferred arrangement for obtaining meals during the course of the hospital stay. In addition, visitors should be sent out of the room during the performance of aerosolizing procedures.
Labor and delivery management
The successful management of patients with COVID-19 requires a rigorous infection control protocol informed by guidelines from national entities, such as the Centers for Disease Control and Prevention, the Society for Maternal-Fetal Medicine, and the American College of Obstetricians and Gynecologists, and by state health departments when available.
Strict limits on the number of obstetricians and other health care workers (HCWs) entering the patient’s room should be enforced and documented to minimize risk to the HCWs attending to patients who have a positive diagnosis or who are under investigation. Only in cases of demonstrable clinical benefit should repeat visits by the same or additional HCWs be permitted. Conventional and electronic tablets present an excellent opportunity for patient follow-up visits without room entry. In our institution, this has been successfully piloted in nonpregnant patients. Obstetricians and others caring for obstetrical patients – especially those who are infected or under investigation for infection – should always wear a properly fitted N95 mask.
Because patients with COVID-19 may have or go on to develop a constellation of organ abnormalities (e.g., cardiovascular, renal, pulmonary), it is vital that a standardized panel of baseline laboratory studies be developed for pregnant patients. This will minimize the need for repeated blood draws and other testing which may increase HCW exposure.
A negative screen based on nonreport of symptoms, lack of temperature elevation, and reported nonexposure to individuals with COVID-19 symptoms still has limitations in terms of disease detection. A recent report from a tertiary care hospital in New York City found that close to one-third of pregnant patients with confirmed COVID-19 admitted over a 2-week period had no viral symptoms or instructive history on initial admission.3 This is consistent with our clinical experience. Most importantly, therefore, routine quantitative reverse transcription polymerase chain reaction testing should be performed on all patients admitted to the L&D unit.
Given the reported variability in the accuracy of polymerase chain reaction testing induced by variable effectiveness of sampling techniques, stage of infection, and inherent test accuracy issues, symptomatic patients with a negative test should first obtain clearance from infectious disease specialists before isolation precautions are discontinued. Repeat testing in 24 hours, including testing of multiple sites, may subsequently yield a positive result in persistently symptomatic patients.
Intrapartum management
As much as possible, standard obstetric indications should guide the timing and route of delivery. In the case of a COVID-19–positive patient or a patient under investigation, nonobstetric factors may bear heavily on decision making, and management flexibility is of great value. For example, in cases of severe or critical disease status, evidence suggests that early delivery regardless of gestational age can improve maternal oxygenation; this supports the liberal use of C-sections in these circumstances. In addition, shortening labor length as well as duration of hospitalization may be expected to reduce the risk of transmission to HCWs, other staff, and other patients.
High rates of cesarean delivery unsurprisingly have been reported thus far: One review of 108 case reports and series of test-positive COVID-19 pregnancies found a 92% C-section rate, and another review and meta-analysis of studies of SARS, MERS, and COVID-19 during pregnancy similarly found that the majority of patients – 84% across all coronavirus infections and 91% in COVID-19 pregnancies – were delivered by C-section.4,5 Given these high rates of cesarean deliveries, the early placement of neuraxial anesthesia while the patient is stable appears to be prudent and obviates the need for intubation, the latter of which is associated with increased aerosol generation and increased virus transmission risk.
Strict protocols for the optimal protection of staff should be observed, including proper personal protective equipment (PPE) protection. Protocols have been detailed in various guidelines and publications; they include the wearing of shoe covers, gowns, N95 masks, goggles, face shields, and two layers of gloves.
For institutions that currently do not offer routine COVID-19 testing to pregnant patients – especially those in areas of outbreaks – N95 masks and eye protection should still be provided to all HCWs involved in the intrapartum management of untested asymptomatic patients, particularly those in the active phase of labor. This protection is justified given the limitations of symptom- and history-based screening and the not-uncommon experience of the patient with a negative screen who subsequently develops the clinical syndrome.
Obstetric management of labor requires close patient contact that potentially elevates the risk of contamination and infection. During the active stage of labor, patient shouting, rapid mouth breathing, and other behaviors inherent to labor all increase the risk of aerosolization of oronasal secretions. In addition, nasal-prong oxygen administration is believed to independently increase the risk of aerosolization of secretions. The casual practice of nasal oxygen application should thus be discontinued and, where felt to be absolutely necessary, a mask should be worn on top of the prongs.
Regarding operative delivery, each participating obstetric surgeon should observe guidelines and recommendations of governing national organizations and professional groups – including the American College of Surgeons – regarding the safe conduct of operations on patients with COVID-19. Written guidelines should be tailored as needed to the performance of C-sections and readily available in L&D. Drills and simulations are generally valuable, and expertise and support should always be available in the labor room to assist with donning and doffing of PPE.
Postpartum care
Expeditious separation of the COVID-19–positive mother from her infant is recommended, including avoidance of delayed cord clamping because of insufficient evidence of benefit to the infant. Insufficient evidence exists to support vertical transmission, but the possibility of maternal-infant transmission is clinically accepted based on small case reports of infection in a neonate at 30 hours of life and in infants of mothers with suspected or confirmed COVID-19.6,7 Accordingly, it is recommended that the benefit of early infant separation should be discussed with the mother. If approved, the infant should be kept in a separate isolation area and observed.
There is no evidence of breast milk transmission of the virus. For those electing to breastfeed, the patient should be provided with a breast pump to express and store the milk for subsequent bottle feeding. For mothers who elect to room in with the infant, a separation distance of 6 feet is recommended with an intervening barrier curtain. For COVID-19–positive mothers who elect breastfeeding, meticulous hand and face washing, continuous wearing of a mask, and cleansing of the breast prior to feeding needs to be maintained.
Restrictive visiting policies of no more than one visitor should be maintained. For severely or critically ill patients with COVID-19, it has been suggested that no visitors be allowed. As with other hospitalizations of COVID-19 patients, the HCW contact should be kept at a justifiable minimum to reduce the risk of transmission.
Protecting the obstetrician and other HCWs
Protecting the health of obstetricians and other HCWs is central to any successful strategy to fight the COVID-19 epidemic. For the individual obstetrician, careful attention to national and local hospital guidelines is required as these are rapidly evolving.
Physicians and their leadership must maintain an ongoing dialogue with hospital leadership to continually upgrade and optimize infection prevention and control measures, and to uphold best practices. The experience in Wuhan, China, illustrates the effectiveness of the proper use of PPE along with population control measures to reduce infections in HCWs. Prior to understanding the mechanism of virus transmission and using protective equipment, infection rates of 3%-29% were reported among HCWs. With the meticulous utilization of mitigation strategies and population control measures – including consistent use of PPE – the rate of infection of HCWs reportedly fell to zero.
In outpatient offices, all staff and HCWs should wear masks at all times and engage in social distancing and in frequent hand sanitization. Patients should be strongly encouraged to wear masks during office visits and on all other occasions when they will be in physical proximity to other individuals outside of the home.
Reports from epidemic areas describe transmission from household sources as a significant cause of HCW infection. The information emphasizes the need for ongoing vigilance and attention to sanitization measures even when at home with one’s family. An additional benefit is reduced risk of transmission from HCWs to family members.
Dr. Bahado-Singh is professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System.
References
1. Luo S et al. Clin Gastroenterol Hepatol. 2020 Mar 20. doi: 10.1016/j.cgh.2020.03.043.
2. Lechien JR et al. Eur Arch Otorhinolaryngol. 2020 Apr 6. doi: 10.1007/s00405-020-05965-1.
3. Breslin N et al. Am J Obstet Gynecol MFM. 2020 Apr 9. doi: 10.1016/j.ajogmf.2020.100118.
4. Zaigham M, Andersson O. Acta Obstet Gynecol Scand. 2020 Apr 7. doi: 10.1111/aogs.13867.
5. Di Mascio D et al. Am J Obstet Gynecol MFM. 2020 Mar 25. doi: 10.1016/j.ajogmf.2020.100107.
6. Ital J. Pediatr 2020;46(1) doi: 10.1186/s13052-020-0820-x.
7. Int J Gynaecol Obstet. 2020;149(2):130-6.
*This article was updated 5/6/2020.
Obstetrics during the COVID-19 pandemic
The identification of the SARS coronavirus (SARS-CoV-2) and emergence of the associated infectious respiratory disease, COVID-19, in late 2019 catapulted the citizens of the world, especially those in the health care professions, into an era of considerable uncertainty. At this moment in human history, calm reassurance – founded in fact and evidence – seems its greatest need. Much of the focus within the biomedical community has been on containment, prevention, and treatment of this highly contagious and, for some, extremely virulent disease.
However, for ob.gyns on the front lines of the COVID-19 fight, there is the additional challenge of caring for at least two patients simultaneously: the mother and her unborn baby. Studies in mother-baby dyads, while being published at an incredible pace, are still quite scarce. In addition, published reports are limited by the small sample size of the patient population (many are single-case reports), lack of uniformity in the timing and types of clinical samples collected, testing delays, and varying isolation protocols in cases where the mother has confirmed SARS-CoV-2.
Five months into a pandemic that has swept the world, we still know very little about COVID-19 infection in the general population, let alone the obstetric one. We do not know if having and resolving COVID-19 infection provides any long-term protection against future disease. We do not know if vertical transmission of SARS-CoV-2 occurs. We do not know if maternal infection confers any immunologic benefit to the neonate. The list goes on.
What we do know is that taking extra precautions works. Use of personal protective equipment saves health care practitioner and patient lives. Prohibiting or restricting visitors to only one person in hospitals reduces risk of transmission to vulnerable patients.
Additionally, we know that leading with compassion is vital to easing patient – and practitioner – anxiety and stress. Most importantly, we know that people are extraordinarily resilient, especially when it comes to safeguarding the health of their families.
To address some of the major concerns that many ob.gyns. have regarding their risk of coronavirus exposure when caring for patients, we have invited Ray Bahado-Singh, MD, professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System, who works in a suburb of Detroit, one of our nation’s COVID-19 hot spots.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland School of Medicine as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
The identification of the SARS coronavirus (SARS-CoV-2) and emergence of the associated infectious respiratory disease, COVID-19, in late 2019 catapulted the citizens of the world, especially those in the health care professions, into an era of considerable uncertainty. At this moment in human history, calm reassurance – founded in fact and evidence – seems its greatest need. Much of the focus within the biomedical community has been on containment, prevention, and treatment of this highly contagious and, for some, extremely virulent disease.
However, for ob.gyns on the front lines of the COVID-19 fight, there is the additional challenge of caring for at least two patients simultaneously: the mother and her unborn baby. Studies in mother-baby dyads, while being published at an incredible pace, are still quite scarce. In addition, published reports are limited by the small sample size of the patient population (many are single-case reports), lack of uniformity in the timing and types of clinical samples collected, testing delays, and varying isolation protocols in cases where the mother has confirmed SARS-CoV-2.
Five months into a pandemic that has swept the world, we still know very little about COVID-19 infection in the general population, let alone the obstetric one. We do not know if having and resolving COVID-19 infection provides any long-term protection against future disease. We do not know if vertical transmission of SARS-CoV-2 occurs. We do not know if maternal infection confers any immunologic benefit to the neonate. The list goes on.
What we do know is that taking extra precautions works. Use of personal protective equipment saves health care practitioner and patient lives. Prohibiting or restricting visitors to only one person in hospitals reduces risk of transmission to vulnerable patients.
Additionally, we know that leading with compassion is vital to easing patient – and practitioner – anxiety and stress. Most importantly, we know that people are extraordinarily resilient, especially when it comes to safeguarding the health of their families.
To address some of the major concerns that many ob.gyns. have regarding their risk of coronavirus exposure when caring for patients, we have invited Ray Bahado-Singh, MD, professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System, who works in a suburb of Detroit, one of our nation’s COVID-19 hot spots.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland School of Medicine as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
The identification of the SARS coronavirus (SARS-CoV-2) and emergence of the associated infectious respiratory disease, COVID-19, in late 2019 catapulted the citizens of the world, especially those in the health care professions, into an era of considerable uncertainty. At this moment in human history, calm reassurance – founded in fact and evidence – seems its greatest need. Much of the focus within the biomedical community has been on containment, prevention, and treatment of this highly contagious and, for some, extremely virulent disease.
However, for ob.gyns on the front lines of the COVID-19 fight, there is the additional challenge of caring for at least two patients simultaneously: the mother and her unborn baby. Studies in mother-baby dyads, while being published at an incredible pace, are still quite scarce. In addition, published reports are limited by the small sample size of the patient population (many are single-case reports), lack of uniformity in the timing and types of clinical samples collected, testing delays, and varying isolation protocols in cases where the mother has confirmed SARS-CoV-2.
Five months into a pandemic that has swept the world, we still know very little about COVID-19 infection in the general population, let alone the obstetric one. We do not know if having and resolving COVID-19 infection provides any long-term protection against future disease. We do not know if vertical transmission of SARS-CoV-2 occurs. We do not know if maternal infection confers any immunologic benefit to the neonate. The list goes on.
What we do know is that taking extra precautions works. Use of personal protective equipment saves health care practitioner and patient lives. Prohibiting or restricting visitors to only one person in hospitals reduces risk of transmission to vulnerable patients.
Additionally, we know that leading with compassion is vital to easing patient – and practitioner – anxiety and stress. Most importantly, we know that people are extraordinarily resilient, especially when it comes to safeguarding the health of their families.
To address some of the major concerns that many ob.gyns. have regarding their risk of coronavirus exposure when caring for patients, we have invited Ray Bahado-Singh, MD, professor and chair of obstetrics and gynecology at Oakland University, Rochester, Mich., and health system chair for obstetrics and gynecology at Beaumont Health System, who works in a suburb of Detroit, one of our nation’s COVID-19 hot spots.
Dr. Reece, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland School of Medicine as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the school of medicine. He is the medical editor of this column. He said he had no relevant financial disclosures. Contact him at [email protected].
NSDUH data might underestimate substance use by pregnant women
New study suggests rate of alcohol use might be almost 19%
The use of alcohol, tobacco products, and drugs by pregnant women is a substantial problem that may be more prevalent than previously thought, according to researcher Kimberly Yonkers, MD.
Higher levels of substance use during pregnancy means more negative impacts on maternal and fetal, neonatal, and child health. However, one bit of good news is that pregnant women still are less likely than nonpregnant women to engage in such behavior, said Dr. Yonkers, director of psychological medicine and the Center for Wellbeing of Women and Mothers at Yale University, New Haven, Conn.
Dr. Yonkers said in a featured presentation at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Tobacco predominates among substances of concern used by pregnant women, with 11.6% reporting past-month use in 2018, according to the National Survey on Drug Use and Health, Dr. Yonkers said. Alcohol was next, with 9.9% of pregnant women reporting use in the past month, followed by drugs at 5.4%, of which marijuana was the most common.
Those numbers may jump much higher when focusing on substance use that’s biologically verified, she added, referring to a recent three-center cross-sectional study she and her colleagues published in Addiction (2019 Jun 19. doi: 10.1111/add.14651). In that study, alcohol use was as high as 18.9% among pregnant women who either had positive urine or self-reported use. Similarly, rates of nicotine or nicotine byproduct detected were 27% at one center in the study, and tetrahydrocannabinol reached 29.4% at that same center.
“These numbers are impressive,” Dr. Yonkers told attendees at the meeting. “So what we may be seeing in terms of the National Survey on Drug Use and Health, as valuable as it is, is in all likelihood it underestimates the use of substances in pregnancy.”
Substance use goes down in pregnancy as some women become more mindful of perinatal health, though unfortunately, that abstinence is offset by a dramatic rise in substance use in the 6-12 months’ post partum, research suggests.
Interestingly, big differences are found in both abstinence and relapse rates, with some data sets showing that, while alcohol is stopped fairly early, cigarettes are stopped much later, if at all.
On the postpartum side of the equation, relapse rates look similar for cigarettes, alcohol, and marijuana, but for some reason, cocaine relapse rates are much lower “That’s kind of nice, and we’d like to be able to understand what it is about this whole process that enabled that relative period of wellness,” Dr. Yonkers said.
Opioid use disorder is rising among pregnant women, just like it is in the general population, and 50% – or possibly even as high as 80% – of babies born to these women will experience neonatal opioid withdrawal, Dr. Yonkers said.
Maternal mortality in the United States increased by 34% from 2008 to 2016; while that’s a sobering statistic, Dr. Yonkers said, opioid-related maternal mortality doubled over that same time period.
“We really have to be mindful that we’re not just talking about taking care of kids and offspring, but we have to take care of moms – it’s really critical,” she said.
With the increasing legalization of cannabis, it’s expected that a lot more cannabis-exposed pregnancies will be seen in clinical practice, and some studies are starting to show an increase in prevalence in the preconception, prenatal, and postpartum period.
While some people feel that cannabis is benign, more data are needed, according to Dr. Yonkers, who said that cannabis and its metabolites cross the blood/placenta and blood/milk barriers, and that cannabinoid receptors are “very important” to fertility, implantation, and fetal development. One study recently published linked cannabis use in pregnancy to significant increases in preterm birth rates (JAMA. 2019 Jun 18;322[2]:145-52).
“We don’t really have a context for this, so we don’t really know what’s going to have an impact, and what’s not,” Dr. Yonkers said.
While both standard and novel treatments could help in the quest to achieve and maintain well-being in this unique patient population, Dr. Yonkers said that health equity and universal approaches to care might be needed to more comprehensively address the problem, which means taking a close look at how much money women have, the resources available to them, and where they live.
In many communities, eliminating inequalities in care will be critical to successfully addressing substance use issues in pregnant women, agreed Maureen Sayres Van Niel, MD, a specialist in women’s psychiatry in Boston and president of the Women’s Caucus of the APA.
“What we see is such a disparity in the delivery of care to women who are poor and living in communities where the socioeconomic and financial problems are very severe,” Dr. Van Niel said in an interview. “Unless we address these disparities, women will not be getting the kind of health care that they really need to have in the perinatal period.”
Dr. Yonkers reported a disclosure related to UpToDate.
New study suggests rate of alcohol use might be almost 19%
New study suggests rate of alcohol use might be almost 19%
The use of alcohol, tobacco products, and drugs by pregnant women is a substantial problem that may be more prevalent than previously thought, according to researcher Kimberly Yonkers, MD.
Higher levels of substance use during pregnancy means more negative impacts on maternal and fetal, neonatal, and child health. However, one bit of good news is that pregnant women still are less likely than nonpregnant women to engage in such behavior, said Dr. Yonkers, director of psychological medicine and the Center for Wellbeing of Women and Mothers at Yale University, New Haven, Conn.
Dr. Yonkers said in a featured presentation at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Tobacco predominates among substances of concern used by pregnant women, with 11.6% reporting past-month use in 2018, according to the National Survey on Drug Use and Health, Dr. Yonkers said. Alcohol was next, with 9.9% of pregnant women reporting use in the past month, followed by drugs at 5.4%, of which marijuana was the most common.
Those numbers may jump much higher when focusing on substance use that’s biologically verified, she added, referring to a recent three-center cross-sectional study she and her colleagues published in Addiction (2019 Jun 19. doi: 10.1111/add.14651). In that study, alcohol use was as high as 18.9% among pregnant women who either had positive urine or self-reported use. Similarly, rates of nicotine or nicotine byproduct detected were 27% at one center in the study, and tetrahydrocannabinol reached 29.4% at that same center.
“These numbers are impressive,” Dr. Yonkers told attendees at the meeting. “So what we may be seeing in terms of the National Survey on Drug Use and Health, as valuable as it is, is in all likelihood it underestimates the use of substances in pregnancy.”
Substance use goes down in pregnancy as some women become more mindful of perinatal health, though unfortunately, that abstinence is offset by a dramatic rise in substance use in the 6-12 months’ post partum, research suggests.
Interestingly, big differences are found in both abstinence and relapse rates, with some data sets showing that, while alcohol is stopped fairly early, cigarettes are stopped much later, if at all.
On the postpartum side of the equation, relapse rates look similar for cigarettes, alcohol, and marijuana, but for some reason, cocaine relapse rates are much lower “That’s kind of nice, and we’d like to be able to understand what it is about this whole process that enabled that relative period of wellness,” Dr. Yonkers said.
Opioid use disorder is rising among pregnant women, just like it is in the general population, and 50% – or possibly even as high as 80% – of babies born to these women will experience neonatal opioid withdrawal, Dr. Yonkers said.
Maternal mortality in the United States increased by 34% from 2008 to 2016; while that’s a sobering statistic, Dr. Yonkers said, opioid-related maternal mortality doubled over that same time period.
“We really have to be mindful that we’re not just talking about taking care of kids and offspring, but we have to take care of moms – it’s really critical,” she said.
With the increasing legalization of cannabis, it’s expected that a lot more cannabis-exposed pregnancies will be seen in clinical practice, and some studies are starting to show an increase in prevalence in the preconception, prenatal, and postpartum period.
While some people feel that cannabis is benign, more data are needed, according to Dr. Yonkers, who said that cannabis and its metabolites cross the blood/placenta and blood/milk barriers, and that cannabinoid receptors are “very important” to fertility, implantation, and fetal development. One study recently published linked cannabis use in pregnancy to significant increases in preterm birth rates (JAMA. 2019 Jun 18;322[2]:145-52).
“We don’t really have a context for this, so we don’t really know what’s going to have an impact, and what’s not,” Dr. Yonkers said.
While both standard and novel treatments could help in the quest to achieve and maintain well-being in this unique patient population, Dr. Yonkers said that health equity and universal approaches to care might be needed to more comprehensively address the problem, which means taking a close look at how much money women have, the resources available to them, and where they live.
In many communities, eliminating inequalities in care will be critical to successfully addressing substance use issues in pregnant women, agreed Maureen Sayres Van Niel, MD, a specialist in women’s psychiatry in Boston and president of the Women’s Caucus of the APA.
“What we see is such a disparity in the delivery of care to women who are poor and living in communities where the socioeconomic and financial problems are very severe,” Dr. Van Niel said in an interview. “Unless we address these disparities, women will not be getting the kind of health care that they really need to have in the perinatal period.”
Dr. Yonkers reported a disclosure related to UpToDate.
The use of alcohol, tobacco products, and drugs by pregnant women is a substantial problem that may be more prevalent than previously thought, according to researcher Kimberly Yonkers, MD.
Higher levels of substance use during pregnancy means more negative impacts on maternal and fetal, neonatal, and child health. However, one bit of good news is that pregnant women still are less likely than nonpregnant women to engage in such behavior, said Dr. Yonkers, director of psychological medicine and the Center for Wellbeing of Women and Mothers at Yale University, New Haven, Conn.
Dr. Yonkers said in a featured presentation at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Tobacco predominates among substances of concern used by pregnant women, with 11.6% reporting past-month use in 2018, according to the National Survey on Drug Use and Health, Dr. Yonkers said. Alcohol was next, with 9.9% of pregnant women reporting use in the past month, followed by drugs at 5.4%, of which marijuana was the most common.
Those numbers may jump much higher when focusing on substance use that’s biologically verified, she added, referring to a recent three-center cross-sectional study she and her colleagues published in Addiction (2019 Jun 19. doi: 10.1111/add.14651). In that study, alcohol use was as high as 18.9% among pregnant women who either had positive urine or self-reported use. Similarly, rates of nicotine or nicotine byproduct detected were 27% at one center in the study, and tetrahydrocannabinol reached 29.4% at that same center.
“These numbers are impressive,” Dr. Yonkers told attendees at the meeting. “So what we may be seeing in terms of the National Survey on Drug Use and Health, as valuable as it is, is in all likelihood it underestimates the use of substances in pregnancy.”
Substance use goes down in pregnancy as some women become more mindful of perinatal health, though unfortunately, that abstinence is offset by a dramatic rise in substance use in the 6-12 months’ post partum, research suggests.
Interestingly, big differences are found in both abstinence and relapse rates, with some data sets showing that, while alcohol is stopped fairly early, cigarettes are stopped much later, if at all.
On the postpartum side of the equation, relapse rates look similar for cigarettes, alcohol, and marijuana, but for some reason, cocaine relapse rates are much lower “That’s kind of nice, and we’d like to be able to understand what it is about this whole process that enabled that relative period of wellness,” Dr. Yonkers said.
Opioid use disorder is rising among pregnant women, just like it is in the general population, and 50% – or possibly even as high as 80% – of babies born to these women will experience neonatal opioid withdrawal, Dr. Yonkers said.
Maternal mortality in the United States increased by 34% from 2008 to 2016; while that’s a sobering statistic, Dr. Yonkers said, opioid-related maternal mortality doubled over that same time period.
“We really have to be mindful that we’re not just talking about taking care of kids and offspring, but we have to take care of moms – it’s really critical,” she said.
With the increasing legalization of cannabis, it’s expected that a lot more cannabis-exposed pregnancies will be seen in clinical practice, and some studies are starting to show an increase in prevalence in the preconception, prenatal, and postpartum period.
While some people feel that cannabis is benign, more data are needed, according to Dr. Yonkers, who said that cannabis and its metabolites cross the blood/placenta and blood/milk barriers, and that cannabinoid receptors are “very important” to fertility, implantation, and fetal development. One study recently published linked cannabis use in pregnancy to significant increases in preterm birth rates (JAMA. 2019 Jun 18;322[2]:145-52).
“We don’t really have a context for this, so we don’t really know what’s going to have an impact, and what’s not,” Dr. Yonkers said.
While both standard and novel treatments could help in the quest to achieve and maintain well-being in this unique patient population, Dr. Yonkers said that health equity and universal approaches to care might be needed to more comprehensively address the problem, which means taking a close look at how much money women have, the resources available to them, and where they live.
In many communities, eliminating inequalities in care will be critical to successfully addressing substance use issues in pregnant women, agreed Maureen Sayres Van Niel, MD, a specialist in women’s psychiatry in Boston and president of the Women’s Caucus of the APA.
“What we see is such a disparity in the delivery of care to women who are poor and living in communities where the socioeconomic and financial problems are very severe,” Dr. Van Niel said in an interview. “Unless we address these disparities, women will not be getting the kind of health care that they really need to have in the perinatal period.”
Dr. Yonkers reported a disclosure related to UpToDate.
EXPERT ANALYSIS FROM APA 2020
The American maternal mortality crisis: The role of racism and bias
April 11-17 marked the third annual national Black Maternal Health Week, an event launched in 2017 by the Atlanta-based Black Mamas Matter Alliance (BMMA), in part to “deepen the national conversation about black maternal health.”
Around the same time, emerging data showing higher mortality rates among black patients versus patients of other races with COVID-19 opened similar dialogue fraught with questions about what might explain the disturbing health disparities.
“It’s kind of surprising to me that people are shocked by these [COVID-19] disparities,” Rebekah Gee, MD, an ob.gyn. who is director of the Louisiana State University Health System in New Orleans and a driving force behind initiatives addressing racial disparities in maternal health, said in an interview. If this is it, great – and certainly every moment is a moment for learning – but these COVID-19 disparities should not be surprising to people who have been looking at data.”
Veronica Gillispie, MD, an ob.gyn. and medical director of the Louisiana Perinatal Quality Collaborative and Pregnancy-Associated Mortality Review, was similarly baffled that the news was treated as a revelation.
That news includes outcomes data from New York showing that in March there were 92.3 and 74.3 deaths per 100,000 black and Hispanic COVID-19 patients, respectively, compared with 45.2 per 100,000 white patients.
“Now there’s a task force and all these initiatives to look at why this is happening, and I think those of us who work in maternal mortality are all saying, ‘We know why it’s happening,’ ” she said. “It’s the same thing we’ve been telling people why it’s been happening in maternal mortality.
“It’s implicit bias and structural racism.”
Facing hard numbers, harder conversations
The U.S. maternal mortality rate in 2018 was 17 per 100,000 live births – the highest of any similarly wealthy industrialized nation, the Centers for Disease Control and Prevention’s National Center for Health Statistics (NCHS) reported in January. That’s a striking statistic in its own right. Perhaps more striking is the breakdown by race.
Hispanic women had the lowest maternal mortality rate at 12 per 100,000 live births, followed by non-Hispanic white women at 15.
The rate for non-Hispanic black women was 37 per 100,000 live births.
Numerous factors contribute to these disparities. Among those listed by the American College of Obstetricians and Gynecologists’ chief executive officer Maureen G. Phipps, MD, in a press statement on the NCHS data, are care access issues, lack of standardization of care, bias, and racism. All of these must be addressed if the disparities in maternal and other areas of care are to be eliminated, according to Dr. Phipps.
“The NCHS data confirmed what we have known from other data sources: The rate of maternal deaths for non-Hispanic black women is substantially higher than the rates for non-Hispanic white women,” she wrote. “Continued efforts to improve the standardization of data and review processes related to U.S. maternal mortality are a necessary step to achieving the goal of eliminating disparities and preventable maternal mortality.”
However, such efforts frequently encounter roadblocks constructed by the reluctance among “many academics, policy makers, scientists, elected officials, journalists, and others responsible for defining and responding to the public discourse” to identify racism as a root cause of health disparities, according to Zinzi D. Bailey, ScD, former director of research and evaluation for the New York City Department of Health and Mental Hygiene, and colleagues.
In the third of a three-part conceptual report in The Lancet, entitled America: Equity and Equality in Health, Dr. Bailey and colleagues argued that advancing health equity requires a focus on structural racism – which they defined as “the totality of ways in which societies foster racial discrimination via mutually reinforcing inequitable systems (e.g., in housing, education, employment, earning, benefits, credit, media, healthcare, and criminal justice, etc.) that in turn reinforce discriminatory beliefs, values, and distribution of resources.”
In their series, the authors peeled back layer upon layer of sociological and political contributors to structural racism throughout history, revealing how each laid a foundation for health inequity over time. They particularly home in on health care quality and access.
“Interpersonal racism, bias, and discrimination in healthcare settings can directly affect health through poor health care,” they wrote, noting that “almost 15 years ago, the Institute of Medicine report entitled Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, documented systematic and pervasive bias in the treatment of people of color resulting in substandard care.”
That report concluded that “bias, stereotyping, prejudice, and clinical uncertainty on the part of healthcare providers” likely play a role in the continuation of health disparities. More recent data – including the NCHS maternal mortality data – show an ongoing crisis.
A study of 210 experienced primary care providers and 190 community members in the Denver area, for example, found substantial evidence of implicit bias against both Latino and African American patients. The authors defined implicit bias as “unintentional and even unconscious” negative evaluation of one group and its members relative to another that is expressed implicitly, such as through negative nonverbal behavior.
“Activated by situational cues (e.g., a person’s skin color), implicit bias can quickly and unknowingly exert its influence on perception, memory, and behavior,” they wrote.
In their study, Implicit Association Test and self-report measures of bias showed similar rates of implicit bias among the providers and community members, with only a slight weakening of ethnic/racial bias among providers after adjustment for background characteristics, which suggests “a wider societal problem,” they said.
A specific example of how implicit bias can manifest was described in a 2016 report addressing the well-documented under-treatment of pain among black versus white patients. Kelly M. Hoffman, PhD, and colleagues demonstrated that a substantial number of individuals with at least some medical training endorse false beliefs regarding biological differences between black and white patients. For example, 25% of 28 white residents surveyed agreed black individuals have thicker skin, and 4% believed black individuals have faster blood coagulation and less sensitivity in their nerve endings.
Those who more strongly endorsed such erroneous beliefs were more likely to underestimate and undertreat pain among black patients, the authors found.
Another study, which underscored the insidiousness of structural racism, was reported in Science. The authors identified significant racial bias in an algorithm widely used by health systems, insurers, and practitioners to allocate health care resources for patients with complex health needs. The algorithm, which affects millions of patients, uses predictions of future health care costs rather than future illness to determine who should receive extra medical care.
The problem is that unequal care access for black patients skews lower the foundational cost data used for making those predictions. Correcting the algorithm would increase the percentage of black patients receiving additional medical help from 17.7% to 46.5%, the authors concluded.
This evidence of persistent racism and bias in medicine, however, doesn’t mean progress is lacking.
ACOG has partnered with numerous other organizations to promote awareness and change, including through legislation. A recent win was the enactment of the Preventing Maternal Deaths Act of 2018, a bipartisan bill designed to promote and support maternal mortality review committees in every state. A major focus of BMMA’s Black Maternal Health Week was the Black Maternal Health Momnibus Act of 2020, a nine-bill package introduced in March to comprehensively address the crisis.
But efforts like these, whether they aim to elucidate the contributors to health disparities or to directly target structural and overt racism and root out implicit bias in medical care, are nothing new. As Dr. Bailey and colleagues noted, a challenge is getting the message across because efforts to avoid tough conversations around these topics are nothing new, either.
Dr. Gee attested to that during a maternal mortality panel discussion at the 2019 ACOG meeting where she spoke about the resistance she encountered in 2016 when she was appointed secretary of the Louisiana Health Department and worked to make racism and bias a foundational part of the discussion on improving maternal and fetal outcomes.
She established the first Office of Health Equity in the state – and the first in the nation to not only require measurement outcomes by race but also explicitly address racial bias at the outset. The goal wasn’t just to talk about it but to “plan for addressing equity in every single aspect of what we do with ... our case equity action teams.”
“At our first maternal mortality quality meetings we insisted on focusing on equity at the very outset, and we had people that left when we started talking about racism,” she said, noting that others said it was “too political” to discuss during an election year or that equity was something to address later.
“We said no. We insisted on it, and I think that was very important because all ships don’t rise with the tide, not with health disparities,” she said, recounting an earlier experience when she led the Louisiana Birth Outcomes Initiative: “I asked to have a brown-bag focused on racism at the department so we could talk about the impact of implicit bias on decision making, and I was told that I was a Yankee who didn’t understand the South and that racism didn’t really exist here, and what did I know about it – and I couldn’t have the brown-bag.”
That was in 2011.
Fast-forward to April 24, 2020. As Dr. Gee shared her perspective on addressing racism and bias in medicine, she was preparing for a call regarding the racial disparities in COVID-19 outcomes – the first health equity action sanctioned by Louisiana Governor John Bel Edwards (D).
“I think we really set the stage for these discussions,” she said.
Addressing equity to enact change
The efforts in Louisiana also set the stage for better maternal outcomes. At the 2019 ACOG meeting where she spoke as part of the President’s Panel, Dr. Gee said Louisiana had the highest maternal mortality rates in the nation. The NCHC data released in January, however, suggest that may no longer be the case.
Inconsistencies in how the latest and prior data were reported, including in how maternal mortality was defined, make direct comparison impossible. But in the latest report, Louisiana ranked seventh among states with available data.
“Ninety percent of the deliveries in the state happen at hospitals that we worked with,” Dr. Gee said, highlighting the reach of the efforts to improve outcomes there.
She also described a recent case involving an anemic patient whose bleeding risk was identified early thanks to the programs put in place. That enabled early preparation in the event of complications.
The patient experienced a massive hemorrhage, but the preparation, including having units of blood on hand in case of such an emergency, saved her life.
“So we clearly have not just data, but individual stories of people whose lives have been saved by this work,” she said.
More tangible data on maternal morbidity further show that the efforts in the state are making a difference, Dr. Gillispie said, citing preliminary outcomes data from the Pregnancy-Associated Mortality Review launched in 2018.
“We started with an initial goal of reducing severe maternal morbidity related to hypertension and hemorrhage by 20%, as well as reducing the black/white disparity gap by Mother’s Day 2020,” she said.
Final analyses have been delayed because of COVID-19, but early assessments showed a reduction in the disparity gap, she said, again highlighting the importance of focusing on equity.
“Definitely from the standpoint of the Quality Collaborative side ... we’ve been working with our facilities to make them aware of what implicit bias is, helping them to also do the Harvard Implicit Bias Test so they can figure out what their own biases are, start working to acknowledge them and address them, and start working to fight against letting that bias change how they treat individuals,” Dr. Gillispie said.
The work started through these initiatives will continue because there is much left to be done, she said.
Indeed, the surprised reactions in recent weeks to the reports of disparities in COVID-19 outcomes further underscore that reality, and the maternal mortality statistics – with use of the voices of those directly affected by structural and overt racism and bias in maternal care as a megaphone – speak for themselves.
Hearing implicit bias from patients’ perspective
Just ask Timoria McQueen Saba, a black woman who nearly died from a postpartum hemorrhage in 2010. At ACOG 2019, she spoke about how she had to switch ob.gyns. three times during her first pregnancy because she felt she had not received quality care – one doctor neglected to tell her she had placenta previa. She also experienced excessive wait times at prenatal appointments and had been on the receiving end of microaggressions and degrading questions such as “Are you still married?” and “Is your husband your baby’s father?” – and these are all things her white friends who recommended those physicians never experienced, she said.
“The health care system has just sometimes beaten people down so much, just like the world has – people of color, especially – to where you’re dismissed, your concerns are invalidated,” she said. “Some doctors don’t even think black people feel pain [or that] our pain is less.”
Mrs. Saba also spoke about how her health care “improved a billion percent” when her white husband accompanied her to appointments.
Just ask Charles S. Johnson IV, whose wife Kira Dixon Johnson died in 2016 during surgery for postpartum bleeding complications – after he and other family members spent 10 hours pleading for help for her.
Speaking at the ACOG panel discussion with Mrs. Saba, Mr. Johnson described “a clear disconnect” between the medical staff at the hospital and the way they viewed and valued Kira. He shared his frustration in wanting to advocate for his wife, but knowing that, as an African American male, he risked being seen as a threat and removed from the hospital if he didn’t stay calm, if he “tapped into those natural instincts as a man and a husband who wants to just protect his family.”
He fought back emotions, struggling to get the words out, saying that’s what haunts him and keeps him up at night – wondering if he should have “fought harder, grabbed the doctor by the collar, raised his voice, slammed on the counter.
“Maybe they would have done something,” he said.
Such experiences cross all socioeconomic boundaries. Ask U.S. Track and Field Olympic gold medalist Allyson Felix, who testified at a U.S. House Ways and Means Committee hearing on May 16, 2019 after developing severe preeclampsia that threatened her life and that of her baby. Ask tennis champion Serena Williams, who demanded assessment for pulmonary embolism following the birth of her child; she knew the signs, but her health care providers initially dismissed her concerns.
Their experiences aren’t just anecdotal. Data consistently show how racism and bias affect patient treatment and outcomes. Dr. Gee, for example, shared findings from a retrospective assessment of 47 confirmed pregnancy-related deaths in Louisiana between 2011 and 2016 that looked specifically at whether the deaths could potentially have been prevented if blood was given sooner, cardiomyopathy was recognized sooner, hypertension was treated on time, or other changes were made to care.
The answer was “Yes” in 9% of cases involving white patients – and in 59% of cases involving black patients (odds ratio, 14.6).
The study, reported in February in Obstetrics & Gynecology, showed that 27 of the deaths (58%) occurred at level III or IV birth facilities and that those deaths were not less likely than those at level I or II facilities to be categorized as preventable (OR, 2.0).
Findings from the Giving Voice to Mothers study, published in Reproductive Health in 2019, showed how mistreatment during childbirth might contribute to such outcomes.
In an online cross-sectional survey of more than 2,100 U.S. women, one in six reported at least one type of mistreatment, such as loss of autonomy, being yelled at or threatened, being ignored or having requests for help ignored, Saraswathi Vedam, SciD, of the Birth Place Lab at the University of British Columbia, Vancouver, and colleagues reported.
Race was among the factors associated with likelihood of mistreatment, and the rates of mistreatment for women of color were consistently higher – even when looking at interactions between race and other characteristics, such as socioeconomic status (SES). For example, 27.2% of women of color with low SES, compared with 18.7% of white women with low SES, reported mistreatment. Having a partner who was black, regardless of maternal race, was also associated with an increased rate of mistreatment, the authors found.
“I often get the question, ‘Do you think Kira would be alive if she was white,’ ” Mr. Johnson said. “The first way I respond to that question is [by saying that] the simple fact that you have to ask me is a problem.
“When this first happened, I was in so much pain that I couldn’t process the fact that something so egregious and outrageous happened to my wife because of the color of her skin, but as I began to process and really think about it and unpack this scenario, I have to be really frank ... do I think that she would have sat there for 10 hours while we begged and pleaded? Absolutely not.”
He stressed that his words aren’t “an indictment of the profession.”
“This is not an indictment saying that all people are racist or prejudiced,” he said. “But here’s the reality: If you are in this profession, if you are responsible for the well-being of patients and their families, and you are not able to see them in the same way that you see your mother, your wife, your sister, you have two options – you need to find something else to do or you need to take steps to get better.”
Fixing systems, finding solutions
Dr. Gee acknowledged the work that physicians need to do to help improve outcomes.
“The average time we give a patient to talk is 11 seconds before we interrupt them,” she said, as one example. “We have to recognize that.”
But efforts to improve outcomes shouldn’t just focus on changing physician behavior, she said.
“We really need to focus, as has the U.K. – very effectively – on using midwives, doulas, other health care professionals as complements to physicians to make sure that we have women-centered birth experiences.
“So, instead of just blaming the doctors, I think we need to change the system,” Dr. Gee emphasized.
The disruptions in health systems caused by COVID-19 present a unique opportunity to do that, she said. There is now an opportunity to build them back.
“We have a chance to build the systems back, and when we do so, we ought to build them back correcting for implicit bias and some of the systemic issues that lead to poor outcomes for people of color in our country,” she said.
Solutions proposed by Dr. Gee and others include more diversity in the workforce, more inclusion of patient advocates in maternal care, development of culturally appropriate literacy and numeracy communications, measurement by race (and action on the outcomes), standardization of care, and development of new ways to improve care access.
We will focus more specifically on these solutions in Part 2 of this article in our maternal mortality series. Previous articles in the series are available at mdedge.com/obgyn/maternal-mortality.
April 11-17 marked the third annual national Black Maternal Health Week, an event launched in 2017 by the Atlanta-based Black Mamas Matter Alliance (BMMA), in part to “deepen the national conversation about black maternal health.”
Around the same time, emerging data showing higher mortality rates among black patients versus patients of other races with COVID-19 opened similar dialogue fraught with questions about what might explain the disturbing health disparities.
“It’s kind of surprising to me that people are shocked by these [COVID-19] disparities,” Rebekah Gee, MD, an ob.gyn. who is director of the Louisiana State University Health System in New Orleans and a driving force behind initiatives addressing racial disparities in maternal health, said in an interview. If this is it, great – and certainly every moment is a moment for learning – but these COVID-19 disparities should not be surprising to people who have been looking at data.”
Veronica Gillispie, MD, an ob.gyn. and medical director of the Louisiana Perinatal Quality Collaborative and Pregnancy-Associated Mortality Review, was similarly baffled that the news was treated as a revelation.
That news includes outcomes data from New York showing that in March there were 92.3 and 74.3 deaths per 100,000 black and Hispanic COVID-19 patients, respectively, compared with 45.2 per 100,000 white patients.
“Now there’s a task force and all these initiatives to look at why this is happening, and I think those of us who work in maternal mortality are all saying, ‘We know why it’s happening,’ ” she said. “It’s the same thing we’ve been telling people why it’s been happening in maternal mortality.
“It’s implicit bias and structural racism.”
Facing hard numbers, harder conversations
The U.S. maternal mortality rate in 2018 was 17 per 100,000 live births – the highest of any similarly wealthy industrialized nation, the Centers for Disease Control and Prevention’s National Center for Health Statistics (NCHS) reported in January. That’s a striking statistic in its own right. Perhaps more striking is the breakdown by race.
Hispanic women had the lowest maternal mortality rate at 12 per 100,000 live births, followed by non-Hispanic white women at 15.
The rate for non-Hispanic black women was 37 per 100,000 live births.
Numerous factors contribute to these disparities. Among those listed by the American College of Obstetricians and Gynecologists’ chief executive officer Maureen G. Phipps, MD, in a press statement on the NCHS data, are care access issues, lack of standardization of care, bias, and racism. All of these must be addressed if the disparities in maternal and other areas of care are to be eliminated, according to Dr. Phipps.
“The NCHS data confirmed what we have known from other data sources: The rate of maternal deaths for non-Hispanic black women is substantially higher than the rates for non-Hispanic white women,” she wrote. “Continued efforts to improve the standardization of data and review processes related to U.S. maternal mortality are a necessary step to achieving the goal of eliminating disparities and preventable maternal mortality.”
However, such efforts frequently encounter roadblocks constructed by the reluctance among “many academics, policy makers, scientists, elected officials, journalists, and others responsible for defining and responding to the public discourse” to identify racism as a root cause of health disparities, according to Zinzi D. Bailey, ScD, former director of research and evaluation for the New York City Department of Health and Mental Hygiene, and colleagues.
In the third of a three-part conceptual report in The Lancet, entitled America: Equity and Equality in Health, Dr. Bailey and colleagues argued that advancing health equity requires a focus on structural racism – which they defined as “the totality of ways in which societies foster racial discrimination via mutually reinforcing inequitable systems (e.g., in housing, education, employment, earning, benefits, credit, media, healthcare, and criminal justice, etc.) that in turn reinforce discriminatory beliefs, values, and distribution of resources.”
In their series, the authors peeled back layer upon layer of sociological and political contributors to structural racism throughout history, revealing how each laid a foundation for health inequity over time. They particularly home in on health care quality and access.
“Interpersonal racism, bias, and discrimination in healthcare settings can directly affect health through poor health care,” they wrote, noting that “almost 15 years ago, the Institute of Medicine report entitled Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, documented systematic and pervasive bias in the treatment of people of color resulting in substandard care.”
That report concluded that “bias, stereotyping, prejudice, and clinical uncertainty on the part of healthcare providers” likely play a role in the continuation of health disparities. More recent data – including the NCHS maternal mortality data – show an ongoing crisis.
A study of 210 experienced primary care providers and 190 community members in the Denver area, for example, found substantial evidence of implicit bias against both Latino and African American patients. The authors defined implicit bias as “unintentional and even unconscious” negative evaluation of one group and its members relative to another that is expressed implicitly, such as through negative nonverbal behavior.
“Activated by situational cues (e.g., a person’s skin color), implicit bias can quickly and unknowingly exert its influence on perception, memory, and behavior,” they wrote.
In their study, Implicit Association Test and self-report measures of bias showed similar rates of implicit bias among the providers and community members, with only a slight weakening of ethnic/racial bias among providers after adjustment for background characteristics, which suggests “a wider societal problem,” they said.
A specific example of how implicit bias can manifest was described in a 2016 report addressing the well-documented under-treatment of pain among black versus white patients. Kelly M. Hoffman, PhD, and colleagues demonstrated that a substantial number of individuals with at least some medical training endorse false beliefs regarding biological differences between black and white patients. For example, 25% of 28 white residents surveyed agreed black individuals have thicker skin, and 4% believed black individuals have faster blood coagulation and less sensitivity in their nerve endings.
Those who more strongly endorsed such erroneous beliefs were more likely to underestimate and undertreat pain among black patients, the authors found.
Another study, which underscored the insidiousness of structural racism, was reported in Science. The authors identified significant racial bias in an algorithm widely used by health systems, insurers, and practitioners to allocate health care resources for patients with complex health needs. The algorithm, which affects millions of patients, uses predictions of future health care costs rather than future illness to determine who should receive extra medical care.
The problem is that unequal care access for black patients skews lower the foundational cost data used for making those predictions. Correcting the algorithm would increase the percentage of black patients receiving additional medical help from 17.7% to 46.5%, the authors concluded.
This evidence of persistent racism and bias in medicine, however, doesn’t mean progress is lacking.
ACOG has partnered with numerous other organizations to promote awareness and change, including through legislation. A recent win was the enactment of the Preventing Maternal Deaths Act of 2018, a bipartisan bill designed to promote and support maternal mortality review committees in every state. A major focus of BMMA’s Black Maternal Health Week was the Black Maternal Health Momnibus Act of 2020, a nine-bill package introduced in March to comprehensively address the crisis.
But efforts like these, whether they aim to elucidate the contributors to health disparities or to directly target structural and overt racism and root out implicit bias in medical care, are nothing new. As Dr. Bailey and colleagues noted, a challenge is getting the message across because efforts to avoid tough conversations around these topics are nothing new, either.
Dr. Gee attested to that during a maternal mortality panel discussion at the 2019 ACOG meeting where she spoke about the resistance she encountered in 2016 when she was appointed secretary of the Louisiana Health Department and worked to make racism and bias a foundational part of the discussion on improving maternal and fetal outcomes.
She established the first Office of Health Equity in the state – and the first in the nation to not only require measurement outcomes by race but also explicitly address racial bias at the outset. The goal wasn’t just to talk about it but to “plan for addressing equity in every single aspect of what we do with ... our case equity action teams.”
“At our first maternal mortality quality meetings we insisted on focusing on equity at the very outset, and we had people that left when we started talking about racism,” she said, noting that others said it was “too political” to discuss during an election year or that equity was something to address later.
“We said no. We insisted on it, and I think that was very important because all ships don’t rise with the tide, not with health disparities,” she said, recounting an earlier experience when she led the Louisiana Birth Outcomes Initiative: “I asked to have a brown-bag focused on racism at the department so we could talk about the impact of implicit bias on decision making, and I was told that I was a Yankee who didn’t understand the South and that racism didn’t really exist here, and what did I know about it – and I couldn’t have the brown-bag.”
That was in 2011.
Fast-forward to April 24, 2020. As Dr. Gee shared her perspective on addressing racism and bias in medicine, she was preparing for a call regarding the racial disparities in COVID-19 outcomes – the first health equity action sanctioned by Louisiana Governor John Bel Edwards (D).
“I think we really set the stage for these discussions,” she said.
Addressing equity to enact change
The efforts in Louisiana also set the stage for better maternal outcomes. At the 2019 ACOG meeting where she spoke as part of the President’s Panel, Dr. Gee said Louisiana had the highest maternal mortality rates in the nation. The NCHC data released in January, however, suggest that may no longer be the case.
Inconsistencies in how the latest and prior data were reported, including in how maternal mortality was defined, make direct comparison impossible. But in the latest report, Louisiana ranked seventh among states with available data.
“Ninety percent of the deliveries in the state happen at hospitals that we worked with,” Dr. Gee said, highlighting the reach of the efforts to improve outcomes there.
She also described a recent case involving an anemic patient whose bleeding risk was identified early thanks to the programs put in place. That enabled early preparation in the event of complications.
The patient experienced a massive hemorrhage, but the preparation, including having units of blood on hand in case of such an emergency, saved her life.
“So we clearly have not just data, but individual stories of people whose lives have been saved by this work,” she said.
More tangible data on maternal morbidity further show that the efforts in the state are making a difference, Dr. Gillispie said, citing preliminary outcomes data from the Pregnancy-Associated Mortality Review launched in 2018.
“We started with an initial goal of reducing severe maternal morbidity related to hypertension and hemorrhage by 20%, as well as reducing the black/white disparity gap by Mother’s Day 2020,” she said.
Final analyses have been delayed because of COVID-19, but early assessments showed a reduction in the disparity gap, she said, again highlighting the importance of focusing on equity.
“Definitely from the standpoint of the Quality Collaborative side ... we’ve been working with our facilities to make them aware of what implicit bias is, helping them to also do the Harvard Implicit Bias Test so they can figure out what their own biases are, start working to acknowledge them and address them, and start working to fight against letting that bias change how they treat individuals,” Dr. Gillispie said.
The work started through these initiatives will continue because there is much left to be done, she said.
Indeed, the surprised reactions in recent weeks to the reports of disparities in COVID-19 outcomes further underscore that reality, and the maternal mortality statistics – with use of the voices of those directly affected by structural and overt racism and bias in maternal care as a megaphone – speak for themselves.
Hearing implicit bias from patients’ perspective
Just ask Timoria McQueen Saba, a black woman who nearly died from a postpartum hemorrhage in 2010. At ACOG 2019, she spoke about how she had to switch ob.gyns. three times during her first pregnancy because she felt she had not received quality care – one doctor neglected to tell her she had placenta previa. She also experienced excessive wait times at prenatal appointments and had been on the receiving end of microaggressions and degrading questions such as “Are you still married?” and “Is your husband your baby’s father?” – and these are all things her white friends who recommended those physicians never experienced, she said.
“The health care system has just sometimes beaten people down so much, just like the world has – people of color, especially – to where you’re dismissed, your concerns are invalidated,” she said. “Some doctors don’t even think black people feel pain [or that] our pain is less.”
Mrs. Saba also spoke about how her health care “improved a billion percent” when her white husband accompanied her to appointments.
Just ask Charles S. Johnson IV, whose wife Kira Dixon Johnson died in 2016 during surgery for postpartum bleeding complications – after he and other family members spent 10 hours pleading for help for her.
Speaking at the ACOG panel discussion with Mrs. Saba, Mr. Johnson described “a clear disconnect” between the medical staff at the hospital and the way they viewed and valued Kira. He shared his frustration in wanting to advocate for his wife, but knowing that, as an African American male, he risked being seen as a threat and removed from the hospital if he didn’t stay calm, if he “tapped into those natural instincts as a man and a husband who wants to just protect his family.”
He fought back emotions, struggling to get the words out, saying that’s what haunts him and keeps him up at night – wondering if he should have “fought harder, grabbed the doctor by the collar, raised his voice, slammed on the counter.
“Maybe they would have done something,” he said.
Such experiences cross all socioeconomic boundaries. Ask U.S. Track and Field Olympic gold medalist Allyson Felix, who testified at a U.S. House Ways and Means Committee hearing on May 16, 2019 after developing severe preeclampsia that threatened her life and that of her baby. Ask tennis champion Serena Williams, who demanded assessment for pulmonary embolism following the birth of her child; she knew the signs, but her health care providers initially dismissed her concerns.
Their experiences aren’t just anecdotal. Data consistently show how racism and bias affect patient treatment and outcomes. Dr. Gee, for example, shared findings from a retrospective assessment of 47 confirmed pregnancy-related deaths in Louisiana between 2011 and 2016 that looked specifically at whether the deaths could potentially have been prevented if blood was given sooner, cardiomyopathy was recognized sooner, hypertension was treated on time, or other changes were made to care.
The answer was “Yes” in 9% of cases involving white patients – and in 59% of cases involving black patients (odds ratio, 14.6).
The study, reported in February in Obstetrics & Gynecology, showed that 27 of the deaths (58%) occurred at level III or IV birth facilities and that those deaths were not less likely than those at level I or II facilities to be categorized as preventable (OR, 2.0).
Findings from the Giving Voice to Mothers study, published in Reproductive Health in 2019, showed how mistreatment during childbirth might contribute to such outcomes.
In an online cross-sectional survey of more than 2,100 U.S. women, one in six reported at least one type of mistreatment, such as loss of autonomy, being yelled at or threatened, being ignored or having requests for help ignored, Saraswathi Vedam, SciD, of the Birth Place Lab at the University of British Columbia, Vancouver, and colleagues reported.
Race was among the factors associated with likelihood of mistreatment, and the rates of mistreatment for women of color were consistently higher – even when looking at interactions between race and other characteristics, such as socioeconomic status (SES). For example, 27.2% of women of color with low SES, compared with 18.7% of white women with low SES, reported mistreatment. Having a partner who was black, regardless of maternal race, was also associated with an increased rate of mistreatment, the authors found.
“I often get the question, ‘Do you think Kira would be alive if she was white,’ ” Mr. Johnson said. “The first way I respond to that question is [by saying that] the simple fact that you have to ask me is a problem.
“When this first happened, I was in so much pain that I couldn’t process the fact that something so egregious and outrageous happened to my wife because of the color of her skin, but as I began to process and really think about it and unpack this scenario, I have to be really frank ... do I think that she would have sat there for 10 hours while we begged and pleaded? Absolutely not.”
He stressed that his words aren’t “an indictment of the profession.”
“This is not an indictment saying that all people are racist or prejudiced,” he said. “But here’s the reality: If you are in this profession, if you are responsible for the well-being of patients and their families, and you are not able to see them in the same way that you see your mother, your wife, your sister, you have two options – you need to find something else to do or you need to take steps to get better.”
Fixing systems, finding solutions
Dr. Gee acknowledged the work that physicians need to do to help improve outcomes.
“The average time we give a patient to talk is 11 seconds before we interrupt them,” she said, as one example. “We have to recognize that.”
But efforts to improve outcomes shouldn’t just focus on changing physician behavior, she said.
“We really need to focus, as has the U.K. – very effectively – on using midwives, doulas, other health care professionals as complements to physicians to make sure that we have women-centered birth experiences.
“So, instead of just blaming the doctors, I think we need to change the system,” Dr. Gee emphasized.
The disruptions in health systems caused by COVID-19 present a unique opportunity to do that, she said. There is now an opportunity to build them back.
“We have a chance to build the systems back, and when we do so, we ought to build them back correcting for implicit bias and some of the systemic issues that lead to poor outcomes for people of color in our country,” she said.
Solutions proposed by Dr. Gee and others include more diversity in the workforce, more inclusion of patient advocates in maternal care, development of culturally appropriate literacy and numeracy communications, measurement by race (and action on the outcomes), standardization of care, and development of new ways to improve care access.
We will focus more specifically on these solutions in Part 2 of this article in our maternal mortality series. Previous articles in the series are available at mdedge.com/obgyn/maternal-mortality.
April 11-17 marked the third annual national Black Maternal Health Week, an event launched in 2017 by the Atlanta-based Black Mamas Matter Alliance (BMMA), in part to “deepen the national conversation about black maternal health.”
Around the same time, emerging data showing higher mortality rates among black patients versus patients of other races with COVID-19 opened similar dialogue fraught with questions about what might explain the disturbing health disparities.
“It’s kind of surprising to me that people are shocked by these [COVID-19] disparities,” Rebekah Gee, MD, an ob.gyn. who is director of the Louisiana State University Health System in New Orleans and a driving force behind initiatives addressing racial disparities in maternal health, said in an interview. If this is it, great – and certainly every moment is a moment for learning – but these COVID-19 disparities should not be surprising to people who have been looking at data.”
Veronica Gillispie, MD, an ob.gyn. and medical director of the Louisiana Perinatal Quality Collaborative and Pregnancy-Associated Mortality Review, was similarly baffled that the news was treated as a revelation.
That news includes outcomes data from New York showing that in March there were 92.3 and 74.3 deaths per 100,000 black and Hispanic COVID-19 patients, respectively, compared with 45.2 per 100,000 white patients.
“Now there’s a task force and all these initiatives to look at why this is happening, and I think those of us who work in maternal mortality are all saying, ‘We know why it’s happening,’ ” she said. “It’s the same thing we’ve been telling people why it’s been happening in maternal mortality.
“It’s implicit bias and structural racism.”
Facing hard numbers, harder conversations
The U.S. maternal mortality rate in 2018 was 17 per 100,000 live births – the highest of any similarly wealthy industrialized nation, the Centers for Disease Control and Prevention’s National Center for Health Statistics (NCHS) reported in January. That’s a striking statistic in its own right. Perhaps more striking is the breakdown by race.
Hispanic women had the lowest maternal mortality rate at 12 per 100,000 live births, followed by non-Hispanic white women at 15.
The rate for non-Hispanic black women was 37 per 100,000 live births.
Numerous factors contribute to these disparities. Among those listed by the American College of Obstetricians and Gynecologists’ chief executive officer Maureen G. Phipps, MD, in a press statement on the NCHS data, are care access issues, lack of standardization of care, bias, and racism. All of these must be addressed if the disparities in maternal and other areas of care are to be eliminated, according to Dr. Phipps.
“The NCHS data confirmed what we have known from other data sources: The rate of maternal deaths for non-Hispanic black women is substantially higher than the rates for non-Hispanic white women,” she wrote. “Continued efforts to improve the standardization of data and review processes related to U.S. maternal mortality are a necessary step to achieving the goal of eliminating disparities and preventable maternal mortality.”
However, such efforts frequently encounter roadblocks constructed by the reluctance among “many academics, policy makers, scientists, elected officials, journalists, and others responsible for defining and responding to the public discourse” to identify racism as a root cause of health disparities, according to Zinzi D. Bailey, ScD, former director of research and evaluation for the New York City Department of Health and Mental Hygiene, and colleagues.
In the third of a three-part conceptual report in The Lancet, entitled America: Equity and Equality in Health, Dr. Bailey and colleagues argued that advancing health equity requires a focus on structural racism – which they defined as “the totality of ways in which societies foster racial discrimination via mutually reinforcing inequitable systems (e.g., in housing, education, employment, earning, benefits, credit, media, healthcare, and criminal justice, etc.) that in turn reinforce discriminatory beliefs, values, and distribution of resources.”
In their series, the authors peeled back layer upon layer of sociological and political contributors to structural racism throughout history, revealing how each laid a foundation for health inequity over time. They particularly home in on health care quality and access.
“Interpersonal racism, bias, and discrimination in healthcare settings can directly affect health through poor health care,” they wrote, noting that “almost 15 years ago, the Institute of Medicine report entitled Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care, documented systematic and pervasive bias in the treatment of people of color resulting in substandard care.”
That report concluded that “bias, stereotyping, prejudice, and clinical uncertainty on the part of healthcare providers” likely play a role in the continuation of health disparities. More recent data – including the NCHS maternal mortality data – show an ongoing crisis.
A study of 210 experienced primary care providers and 190 community members in the Denver area, for example, found substantial evidence of implicit bias against both Latino and African American patients. The authors defined implicit bias as “unintentional and even unconscious” negative evaluation of one group and its members relative to another that is expressed implicitly, such as through negative nonverbal behavior.
“Activated by situational cues (e.g., a person’s skin color), implicit bias can quickly and unknowingly exert its influence on perception, memory, and behavior,” they wrote.
In their study, Implicit Association Test and self-report measures of bias showed similar rates of implicit bias among the providers and community members, with only a slight weakening of ethnic/racial bias among providers after adjustment for background characteristics, which suggests “a wider societal problem,” they said.
A specific example of how implicit bias can manifest was described in a 2016 report addressing the well-documented under-treatment of pain among black versus white patients. Kelly M. Hoffman, PhD, and colleagues demonstrated that a substantial number of individuals with at least some medical training endorse false beliefs regarding biological differences between black and white patients. For example, 25% of 28 white residents surveyed agreed black individuals have thicker skin, and 4% believed black individuals have faster blood coagulation and less sensitivity in their nerve endings.
Those who more strongly endorsed such erroneous beliefs were more likely to underestimate and undertreat pain among black patients, the authors found.
Another study, which underscored the insidiousness of structural racism, was reported in Science. The authors identified significant racial bias in an algorithm widely used by health systems, insurers, and practitioners to allocate health care resources for patients with complex health needs. The algorithm, which affects millions of patients, uses predictions of future health care costs rather than future illness to determine who should receive extra medical care.
The problem is that unequal care access for black patients skews lower the foundational cost data used for making those predictions. Correcting the algorithm would increase the percentage of black patients receiving additional medical help from 17.7% to 46.5%, the authors concluded.
This evidence of persistent racism and bias in medicine, however, doesn’t mean progress is lacking.
ACOG has partnered with numerous other organizations to promote awareness and change, including through legislation. A recent win was the enactment of the Preventing Maternal Deaths Act of 2018, a bipartisan bill designed to promote and support maternal mortality review committees in every state. A major focus of BMMA’s Black Maternal Health Week was the Black Maternal Health Momnibus Act of 2020, a nine-bill package introduced in March to comprehensively address the crisis.
But efforts like these, whether they aim to elucidate the contributors to health disparities or to directly target structural and overt racism and root out implicit bias in medical care, are nothing new. As Dr. Bailey and colleagues noted, a challenge is getting the message across because efforts to avoid tough conversations around these topics are nothing new, either.
Dr. Gee attested to that during a maternal mortality panel discussion at the 2019 ACOG meeting where she spoke about the resistance she encountered in 2016 when she was appointed secretary of the Louisiana Health Department and worked to make racism and bias a foundational part of the discussion on improving maternal and fetal outcomes.
She established the first Office of Health Equity in the state – and the first in the nation to not only require measurement outcomes by race but also explicitly address racial bias at the outset. The goal wasn’t just to talk about it but to “plan for addressing equity in every single aspect of what we do with ... our case equity action teams.”
“At our first maternal mortality quality meetings we insisted on focusing on equity at the very outset, and we had people that left when we started talking about racism,” she said, noting that others said it was “too political” to discuss during an election year or that equity was something to address later.
“We said no. We insisted on it, and I think that was very important because all ships don’t rise with the tide, not with health disparities,” she said, recounting an earlier experience when she led the Louisiana Birth Outcomes Initiative: “I asked to have a brown-bag focused on racism at the department so we could talk about the impact of implicit bias on decision making, and I was told that I was a Yankee who didn’t understand the South and that racism didn’t really exist here, and what did I know about it – and I couldn’t have the brown-bag.”
That was in 2011.
Fast-forward to April 24, 2020. As Dr. Gee shared her perspective on addressing racism and bias in medicine, she was preparing for a call regarding the racial disparities in COVID-19 outcomes – the first health equity action sanctioned by Louisiana Governor John Bel Edwards (D).
“I think we really set the stage for these discussions,” she said.
Addressing equity to enact change
The efforts in Louisiana also set the stage for better maternal outcomes. At the 2019 ACOG meeting where she spoke as part of the President’s Panel, Dr. Gee said Louisiana had the highest maternal mortality rates in the nation. The NCHC data released in January, however, suggest that may no longer be the case.
Inconsistencies in how the latest and prior data were reported, including in how maternal mortality was defined, make direct comparison impossible. But in the latest report, Louisiana ranked seventh among states with available data.
“Ninety percent of the deliveries in the state happen at hospitals that we worked with,” Dr. Gee said, highlighting the reach of the efforts to improve outcomes there.
She also described a recent case involving an anemic patient whose bleeding risk was identified early thanks to the programs put in place. That enabled early preparation in the event of complications.
The patient experienced a massive hemorrhage, but the preparation, including having units of blood on hand in case of such an emergency, saved her life.
“So we clearly have not just data, but individual stories of people whose lives have been saved by this work,” she said.
More tangible data on maternal morbidity further show that the efforts in the state are making a difference, Dr. Gillispie said, citing preliminary outcomes data from the Pregnancy-Associated Mortality Review launched in 2018.
“We started with an initial goal of reducing severe maternal morbidity related to hypertension and hemorrhage by 20%, as well as reducing the black/white disparity gap by Mother’s Day 2020,” she said.
Final analyses have been delayed because of COVID-19, but early assessments showed a reduction in the disparity gap, she said, again highlighting the importance of focusing on equity.
“Definitely from the standpoint of the Quality Collaborative side ... we’ve been working with our facilities to make them aware of what implicit bias is, helping them to also do the Harvard Implicit Bias Test so they can figure out what their own biases are, start working to acknowledge them and address them, and start working to fight against letting that bias change how they treat individuals,” Dr. Gillispie said.
The work started through these initiatives will continue because there is much left to be done, she said.
Indeed, the surprised reactions in recent weeks to the reports of disparities in COVID-19 outcomes further underscore that reality, and the maternal mortality statistics – with use of the voices of those directly affected by structural and overt racism and bias in maternal care as a megaphone – speak for themselves.
Hearing implicit bias from patients’ perspective
Just ask Timoria McQueen Saba, a black woman who nearly died from a postpartum hemorrhage in 2010. At ACOG 2019, she spoke about how she had to switch ob.gyns. three times during her first pregnancy because she felt she had not received quality care – one doctor neglected to tell her she had placenta previa. She also experienced excessive wait times at prenatal appointments and had been on the receiving end of microaggressions and degrading questions such as “Are you still married?” and “Is your husband your baby’s father?” – and these are all things her white friends who recommended those physicians never experienced, she said.
“The health care system has just sometimes beaten people down so much, just like the world has – people of color, especially – to where you’re dismissed, your concerns are invalidated,” she said. “Some doctors don’t even think black people feel pain [or that] our pain is less.”
Mrs. Saba also spoke about how her health care “improved a billion percent” when her white husband accompanied her to appointments.
Just ask Charles S. Johnson IV, whose wife Kira Dixon Johnson died in 2016 during surgery for postpartum bleeding complications – after he and other family members spent 10 hours pleading for help for her.
Speaking at the ACOG panel discussion with Mrs. Saba, Mr. Johnson described “a clear disconnect” between the medical staff at the hospital and the way they viewed and valued Kira. He shared his frustration in wanting to advocate for his wife, but knowing that, as an African American male, he risked being seen as a threat and removed from the hospital if he didn’t stay calm, if he “tapped into those natural instincts as a man and a husband who wants to just protect his family.”
He fought back emotions, struggling to get the words out, saying that’s what haunts him and keeps him up at night – wondering if he should have “fought harder, grabbed the doctor by the collar, raised his voice, slammed on the counter.
“Maybe they would have done something,” he said.
Such experiences cross all socioeconomic boundaries. Ask U.S. Track and Field Olympic gold medalist Allyson Felix, who testified at a U.S. House Ways and Means Committee hearing on May 16, 2019 after developing severe preeclampsia that threatened her life and that of her baby. Ask tennis champion Serena Williams, who demanded assessment for pulmonary embolism following the birth of her child; she knew the signs, but her health care providers initially dismissed her concerns.
Their experiences aren’t just anecdotal. Data consistently show how racism and bias affect patient treatment and outcomes. Dr. Gee, for example, shared findings from a retrospective assessment of 47 confirmed pregnancy-related deaths in Louisiana between 2011 and 2016 that looked specifically at whether the deaths could potentially have been prevented if blood was given sooner, cardiomyopathy was recognized sooner, hypertension was treated on time, or other changes were made to care.
The answer was “Yes” in 9% of cases involving white patients – and in 59% of cases involving black patients (odds ratio, 14.6).
The study, reported in February in Obstetrics & Gynecology, showed that 27 of the deaths (58%) occurred at level III or IV birth facilities and that those deaths were not less likely than those at level I or II facilities to be categorized as preventable (OR, 2.0).
Findings from the Giving Voice to Mothers study, published in Reproductive Health in 2019, showed how mistreatment during childbirth might contribute to such outcomes.
In an online cross-sectional survey of more than 2,100 U.S. women, one in six reported at least one type of mistreatment, such as loss of autonomy, being yelled at or threatened, being ignored or having requests for help ignored, Saraswathi Vedam, SciD, of the Birth Place Lab at the University of British Columbia, Vancouver, and colleagues reported.
Race was among the factors associated with likelihood of mistreatment, and the rates of mistreatment for women of color were consistently higher – even when looking at interactions between race and other characteristics, such as socioeconomic status (SES). For example, 27.2% of women of color with low SES, compared with 18.7% of white women with low SES, reported mistreatment. Having a partner who was black, regardless of maternal race, was also associated with an increased rate of mistreatment, the authors found.
“I often get the question, ‘Do you think Kira would be alive if she was white,’ ” Mr. Johnson said. “The first way I respond to that question is [by saying that] the simple fact that you have to ask me is a problem.
“When this first happened, I was in so much pain that I couldn’t process the fact that something so egregious and outrageous happened to my wife because of the color of her skin, but as I began to process and really think about it and unpack this scenario, I have to be really frank ... do I think that she would have sat there for 10 hours while we begged and pleaded? Absolutely not.”
He stressed that his words aren’t “an indictment of the profession.”
“This is not an indictment saying that all people are racist or prejudiced,” he said. “But here’s the reality: If you are in this profession, if you are responsible for the well-being of patients and their families, and you are not able to see them in the same way that you see your mother, your wife, your sister, you have two options – you need to find something else to do or you need to take steps to get better.”
Fixing systems, finding solutions
Dr. Gee acknowledged the work that physicians need to do to help improve outcomes.
“The average time we give a patient to talk is 11 seconds before we interrupt them,” she said, as one example. “We have to recognize that.”
But efforts to improve outcomes shouldn’t just focus on changing physician behavior, she said.
“We really need to focus, as has the U.K. – very effectively – on using midwives, doulas, other health care professionals as complements to physicians to make sure that we have women-centered birth experiences.
“So, instead of just blaming the doctors, I think we need to change the system,” Dr. Gee emphasized.
The disruptions in health systems caused by COVID-19 present a unique opportunity to do that, she said. There is now an opportunity to build them back.
“We have a chance to build the systems back, and when we do so, we ought to build them back correcting for implicit bias and some of the systemic issues that lead to poor outcomes for people of color in our country,” she said.
Solutions proposed by Dr. Gee and others include more diversity in the workforce, more inclusion of patient advocates in maternal care, development of culturally appropriate literacy and numeracy communications, measurement by race (and action on the outcomes), standardization of care, and development of new ways to improve care access.
We will focus more specifically on these solutions in Part 2 of this article in our maternal mortality series. Previous articles in the series are available at mdedge.com/obgyn/maternal-mortality.
Primary care physicians reshuffle their work, lives in a pandemic
During his shift at a COVID-19 drive-through triage screening area set up outside the University of Arkansas for Medical Sciences in Little Rock, Robert Hopkins Jr., MD, noticed a woman bowled over in the front seat of her car.
A nurse practitioner had just informed her that she had met the criteria for undergoing testing for the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
“She was very upset and was crying nearly inconsolably,” said Dr. Hopkins, who directs the division of general internal medicine at the University of Arkansas Medical Sciences College of Medicine. “I went over and visited with her for a few minutes. She was scared to death that we [had] told her she was going to die. In her mind, if she had COVID-19 that meant a death sentence, and if we were testing her that meant she was likely to not survive.”
Dr. Hopkins tried his best to put testing in perspective for the woman. “At least she came to a level of comfort and realized that we were doing this for her, that this was not a death sentence, that this was not her fault,” he said. “She was worried about infecting her kids and her grandkids and ending up in the hospital and being a burden. Being able to spend that few minutes with her and help to bring down her level of anxiety – I think that’s where we need to put our efforts as physicians right now, helping people understand, ‘Yes, this is serious. Yes, we need to continue to social distance. Yes, we need to be cautious. But, we will get through this if we all work together to do so.’ ”
Prior to the COVID-19 pandemic, Dr. Hopkins spent part of his time seeing patients in the university’s main hospital, but most of it in an outpatient clinic where he and about 20 other primary care physicians care for patients and precept medical residents. Now, medical residents have been deployed to other services, primarily in the hospital, and he and his physician colleagues are conducting 80%-90% of patient visits by video conferencing or by telephone. It’s a whole new world.
“We’ve gone from a relatively traditional inpatient/outpatient practice where we’re seeing patients face to face to doing some face-to-face visits, but an awful lot of what we do now is in the technology domain,” said Dr. Hopkins, who also assisted with health care relief efforts during hurricanes Rita and Katrina.
“A group of six of us has been redeployed to assist with the surge unit for the inpatient facility, so our outpatient duties are being taken on by some of our partners.”
He also pitches in at the drive-through COVID-19 screening clinic, which was set up on March 27 and operates between 8 a.m. and 8 p.m., 7 days a week. “We’re able to measure people’s temperature, take a quick screening history, decide whether their risk is such that we need to do a COVID-19 PCR [polymerase chain reaction] test,” he said. “Then we make a determination of whether they need to go home on quarantine awaiting those results, or if they don’t have anything that needs to be evaluated, or whether they need to be triaged to an urgent care setting or to the emergency department.”
To minimize his risk of acquiring COVID-19, he follows personal hygiene practices recommended by the Centers for Disease Control and Prevention. He also places his work shoes in a shoebox, which he keeps in his car. “I put them on when I get to the parking deck at work, do my work, and then I put them in the shoebox, slip on another pair of shoes and drive home so I’m not tracking in things I potentially had on me,” said Dr. Hopkins, who is married and a father to two college-aged sons and a daughter in fourth grade. “When I get home I immediately shower, and then I exercise or have dinner with my family.”
Despite the longer-than-usual work hours and upheaval to the traditional medical practice model brought on by the pandemic, Dr. Hopkins, a self-described “glass half full person,” said that he does his best to keep watch over his patients and colleagues. “I’m trying to keep an eye out on my team members – physicians, nurses, medical assistants, and folks at the front desk – trying to make sure that people are getting rest, trying to make sure that people are not overcommitting,” he said. “Because if we’re not all working together and working for the long term, we’re going to be in trouble. This is not going to be a sprint; this is going to be a marathon for us to get through.”
To keep mentally centered, he engages in at least 40 minutes of exercise each day on his bicycle or on the elliptical machine at home. Dr. Hopkins hopes that the current efforts to redeploy resources, expand clinician skill sets, and forge relationships with colleagues in other disciplines will carry over into the delivery of health care when COVID-19 is a distant memory. “I hope that some of those relationships are going to continue and result in better care for all of our patients,” he said.
"We are in dire need of hugs"
Typically, Dr. Dakkak, a family physician at Boston University, practices a mix of clinic-based family medicine and obstetrics, and works in inpatient medicine 6 weeks a year. Currently, she is leading a COVID-19 team full time at Boston Medical Center, a 300-bed safety-net hospital located on the campus of Boston University Medical Center.
COVID-19 has also shaken up her life at home.
When Dr. Dakkak volunteered to take on her new role, the first thing that came to her mind was how making the switch would affect the well-being of her 8-year-old son and 10-year-old daughter.
“I thought, ‘How do I get my children somewhere where I don’t have to worry about them?’ ” Dr. Dakkak said.
She floated the idea with her husband of flying their children out to stay with her recently retired parents, who live outside of Sacramento, Calif., until the pandemic eases up. “I was thinking to myself, ‘Am I overreacting? Is the pandemic not going to be that bad?’ because the rest of the country seemed to be in some amount of denial,” she said. “So, I called my dad, who’s a retired pediatric anesthesiologist. He’s from Egypt so he’s done crisis medicine in his time. He encouraged me to send the kids.”
On the same day that Dr. Dakkak began her first 12-hour COVID-19 shift at the hospital, her husband and children boarded a plane to California, where the kids remain in the care of her parents. Her husband returned after staying there for 2 weeks. “Every day when I’m working, I validate my decision,” she said. “When I first started, I worked 5 nights in a row, had 2 days off, and then worked 6 nights in a row. I was busy so I didn’t think about [being away from my kids], but at the same time I was grateful that I didn’t have to come home and worry about homeschooling the kids or infecting them.”
She checks in with them as she can via cell phone or FaceTime. “My son has been very honest,” Dr. Dakkak said. “He says, ‘FaceTime makes me miss you more, and I don’t like it,’ which I understand. I’ll call my mom, and if they want to talk to me, they’ll talk to me. If they don’t want to talk to me, I’m okay. This is about them being healthy and safe. I sent them a care package a few days ago with cards and some workbooks. I’m optimistic that in June I can at least see them if not bring them home.”
Dr. Dakkak describes leading a COVID-19 team as a grueling experience that challenges her medical know-how nearly every day, with seemingly ever-changing algorithms. “Our knowledge of this disease is five steps behind, and changing at lightning speed,” said Dr. Dakkak, who completed a fellowship in surgical and high-risk obstetrics. “It’s hard to balance continuing to teach evidence-based medicine for everything else in medicine [with continuing] to practice minimal and ever-changing evidence-based COVID medicine. We just don’t know enough [about the virus] yet. This is nothing like we were taught in medical school. Everyone has elevated d-dimers with COVID-19, and we don’t get CT pulmonary angiograms [CTPAs] on all of them; we wouldn’t physically be able to. Some patients have d-dimers in the thousands, and only some are stable to get CTPAs. We are also finding pulmonary embolisms. Now we’re basing our algorithm on anticoagulation due to d-dimers because sometimes you can’t always do a CTPA even if you want to. On the other hand, we have people who are coming into the hospital too late. We’ve had a few who have come in after having days of stroke symptoms. I worry about our patients at home who hesitate to come in when they really should.”
Sometimes she feels sad for the medical residents on her team because their instinct is to go in and check on each patient, “but I don’t want them to get exposed,” she said. “So, we check in by phone, or if they need a physical check-in, we minimize the check-ins; only one of us goes in. I’m more willing to put myself in the room than to put them in the room. I also feel for them because they came into medicine for the humanity of medicine – not the charting or the ordering of medicine. I also worry about the acuity and sadness they’re seeing. This is a rough introduction to medicine for them.”
When interviewed for this story in late April, Dr. Dakkak had kept track of her intubated COVID-19 patients. “Most of my patients get to go home without having been intubated, but those aren’t the ones I worry about,” she said. “I have two patients I have been watching. One of them has just been extubated and I’m still worried about him, but I’m hoping he’s going to be fine. The other one is the first pregnant woman we intubated. She is now extubated, doing really well, and went home. Her fetus is doing well, never had any issues while she was intubated. Those cases make me happy. They were both under the age of 35. It is nice to follow those intubations and find that the majority are doing okay.”
The first patient she had cared for who died was a young man “who was always in good spirits,” she recalled. “We called his brother right before intubating him. After intubation, his oxygen saturation didn’t jump up, which made me worry a bit.” About a week later, the young man died. “I kept thinking, ‘We intubated him when he was still comfortable talking. Should I have put it off and had him call more people to say goodbye? Should I have known that he wasn’t going to wake up?’ ” said Dr. Dakkak, who is also women’s health director at Manet Community Health Centers. “A lot of us have worked on our end-of-life discussions in the past month, just being able to tell somebody, ‘This might be your last time to call family. Call family and talk to whoever you want.’ Guilt isn’t the right word, but it’s unsettling if I’m the last person a patient talks to. I feel that, if that’s the case, then I didn’t do a good enough job trying to get them to their family or friends. If I am worried about a patient’s clinical status declining, I tell families now, when I call them, ‘I hope I’m wrong; I hope they don’t need to be intubated, but I think this is the time to talk.’ ”
To keep herself grounded during off hours, Dr. Dakkak spends time resting, checking in with her family, journaling “to get a lot of feelings out,” gardening, hiking, and joining Zoom chats with friends. Once recentered, she draws from a sense of obligation to others as she prepares for her next shift caring for COVID-19 patients.
“I have a lot of love for the world that I get to expend by doing this hard work,” she said. “I love humanity and I love humanity in times of crisis. The interactions I have with patients and their families are still central to why I do this work. I love my medical teams, and I would never want to let them down. It is nice to feel the sense of teamwork across the hospital. The nurses that I sit with and experience this with are amazing. I keep saying that the only thing I want to do when this pandemic is over is hug everyone. I think we are in dire need of hugs.”
Finding light in the darkness
Internist Katie Jobbins, DO, also has worked in a professional role that was created because of COVID-19.
Shortly before Dr. Jobbins was deployed to Baystate Medical Center in Springfield, Ma., for 2 weeks in April of 2020 to help clinicians with an anticipated surge of COVID-19 cases, she encountered a patient who walked into Baystate’s High Street Health Center.
“I think I have COVID-19,” the patient proclaimed to her, at the outpatient clinic that serves mostly inner-city, Medicaid patients.
Prior to becoming an ambulatory internist, Dr. Jobbins was a surgical resident. “So I went into that mode of ‘I need to do this, this, and this,’ ” she said. “I went through a checklist in my head to make sure I was prepared to take care of the patient.”
She applied that same systems approach during her redeployment assignment in the tertiary care hospital, which typically involved 10-hour shifts overseeing internal medicine residents in a medical telemetry unit. “We would take care of people under investigation for COVID-19, but we were not assigned to the actual COVID unit,” said Dr. Jobbins, who is also associate program director for the internal medicine residency program at the University of Massachusetts Medical School–Baystate Springfield. “They tried to redeploy other people to those units who had special training, and we were trying to back fill into where those people that got moved to the COVID units or the ICU units were actually working. We were taking more of the medical side of the floors.”
Even so, one patient on the unit was suspected of having COVID-19, so Dr. Jobbins suited up with personal protective equipment and conducted a thorough exam with residents waiting outside the patient’s room, a safe distance away. “I explained everything I found on the exam to the residents, trying to give them some educational benefit, even though they couldn’t physically examine the patient because we’re trying to protect them since they’re in training,” she said. “It was anxiety provoking, on some level, knowing that there’s a potential risk of exposure [to the virus], but knowing that Baystate Health has gone to extraordinary measures to make sure we have the correct PPE and support us is reassuring. I knew I had the right equipment and the right tools to take care of the patient, which calmed my nerves and made me feel like I could do the job. That’s the most important thing as a physician during this time: knowing that you have people supporting you who have your back at all times.”
Like Dr. Dakkak, Dr. Jobbins had to make some adjustments to her interaction with her family.
Before she began the deployment, Dr. Jobbins engaged in a frank discussion with her husband and her two young boys about the risks she faced working in a hospital caring for patients with COVID-19. “My husband and I made sure our wills were up to date, and we talked about what we would do if either of us got the virus,” she said. To minimize the potential risk of transmitting the virus to her loved ones during the two-week deployment, she considered living away from her family in a nearby home owned by her father, but decided against that and to “take it day by day.” Following her hospital shifts, Dr. Jobbins changed into a fresh set of clothes before leaving the hospital. Once she arrived home, she showered to reduce the risk of possibly becoming a vector to her family.
She had to tell her kids: “You can’t kiss me right now.”
“As much as it’s hard for them to understand, we had a conversation [in which I explained] ‘This is a virus. It will go away eventually, but it’s a virus we’re fighting.’ It’s interesting to watch a 3-year-old try to process that and take his play samurai sword or Marvel toys and decide he’s going to run around the neighborhood and try to kill the virus.”
At the High Street Health Center, Dr. Jobbins and her colleagues have transitioned to conducting most patient encounters via telephone or video appointments. “We have tried to maintain as much continuity for our patients to address their chronic medical needs through these visits, such as hypertension management and diabetes care,” she said. “We have begun a rigorous screening process to triage and treat patients suspicious for COVID-19 through telehealth in hopes of keeping them safe and in their own homes. We also continue to see patients for nonrespiratory urgent care needs in person once they have screened negative for COVID-19.”
“In terms of the inpatient setting, we’ve noticed that a lot of people are choosing not to go to the hospital now, unless they’re extremely ill,” Dr. Jobbins noted. “We’re going to need to find a balance with when do people truly need to go to the hospital and when do they not? What can we manage as an outpatient versus having someone go to the emergency department? That’s really the role of the primary care physician. We need to help people understand, ‘You don’t need to go to the ED for everything, but here are the things you really need to go for.’ ”
“It will be interesting to see what health care looks like in 6 months or a year. I’m excited to see where we land,” Dr. Jobbins added.
Hopes for the Future of Telemedicine
When the practice of medicine enters a post–COVID-19 era, Dr. Jobbins hopes that telemedicine will be incorporated more into the delivery of patient care. “I’ve found that many of my patients who often are no-shows to the inpatient version of their visits have had a higher success rate of follow-through when we do the telephone visits,” she said. “It’s been very successful. I hope that the insurance companies and [Centers for Medicare & Medicaid] will continue to reimburse this as they see this is a benefit to our patients.
Dr. Hopkins is also hopeful that physicians will be able to successfully see patients via telemedicine in the postpandemic world.
“For the ups and downs we’ve had with telemedicine, I’d love for us to be able to enhance the positives and incorporate that into our practice going forward. If we can reach our patients and help treat them where they are, rather than them having to come to us, that may be a plus,” he said.
In the meantime, Dr. Jobbins presses on as the curve of COVID-19 cases flattens in Western Massachusetts and remains grateful that she chose to practice medicine.
“The commitment I have to being an educator in addition to being a physician is part of why I keep doing this,” Dr. Jobbins said. “I find this to be one of the most fulfilling jobs and careers you could ever have: being there for people when they need you the most. That’s really what a physician’s job is: being there for people when a family member has passed away or when they just need to talk because they’re having anxiety. At the end of the day, if we can impart that to those we work with and bring in a positive attitude, it’s infectious and it makes people see this is a reason we keep doing what we’re doing.”
She’s also been heartened by the kindness of strangers during this pandemic, from those who made and donated face shields when they were in short supply, to those who delivered food to the hospital as a gesture of thanks.
“I had a patient who made homemade masks and sent them to my office,” she said. “There’s obviously good and bad during this time, but I get hope from seeing all of the good things that are coming out of this, the whole idea of finding the light in the darkness.”
During his shift at a COVID-19 drive-through triage screening area set up outside the University of Arkansas for Medical Sciences in Little Rock, Robert Hopkins Jr., MD, noticed a woman bowled over in the front seat of her car.
A nurse practitioner had just informed her that she had met the criteria for undergoing testing for the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
“She was very upset and was crying nearly inconsolably,” said Dr. Hopkins, who directs the division of general internal medicine at the University of Arkansas Medical Sciences College of Medicine. “I went over and visited with her for a few minutes. She was scared to death that we [had] told her she was going to die. In her mind, if she had COVID-19 that meant a death sentence, and if we were testing her that meant she was likely to not survive.”
Dr. Hopkins tried his best to put testing in perspective for the woman. “At least she came to a level of comfort and realized that we were doing this for her, that this was not a death sentence, that this was not her fault,” he said. “She was worried about infecting her kids and her grandkids and ending up in the hospital and being a burden. Being able to spend that few minutes with her and help to bring down her level of anxiety – I think that’s where we need to put our efforts as physicians right now, helping people understand, ‘Yes, this is serious. Yes, we need to continue to social distance. Yes, we need to be cautious. But, we will get through this if we all work together to do so.’ ”
Prior to the COVID-19 pandemic, Dr. Hopkins spent part of his time seeing patients in the university’s main hospital, but most of it in an outpatient clinic where he and about 20 other primary care physicians care for patients and precept medical residents. Now, medical residents have been deployed to other services, primarily in the hospital, and he and his physician colleagues are conducting 80%-90% of patient visits by video conferencing or by telephone. It’s a whole new world.
“We’ve gone from a relatively traditional inpatient/outpatient practice where we’re seeing patients face to face to doing some face-to-face visits, but an awful lot of what we do now is in the technology domain,” said Dr. Hopkins, who also assisted with health care relief efforts during hurricanes Rita and Katrina.
“A group of six of us has been redeployed to assist with the surge unit for the inpatient facility, so our outpatient duties are being taken on by some of our partners.”
He also pitches in at the drive-through COVID-19 screening clinic, which was set up on March 27 and operates between 8 a.m. and 8 p.m., 7 days a week. “We’re able to measure people’s temperature, take a quick screening history, decide whether their risk is such that we need to do a COVID-19 PCR [polymerase chain reaction] test,” he said. “Then we make a determination of whether they need to go home on quarantine awaiting those results, or if they don’t have anything that needs to be evaluated, or whether they need to be triaged to an urgent care setting or to the emergency department.”
To minimize his risk of acquiring COVID-19, he follows personal hygiene practices recommended by the Centers for Disease Control and Prevention. He also places his work shoes in a shoebox, which he keeps in his car. “I put them on when I get to the parking deck at work, do my work, and then I put them in the shoebox, slip on another pair of shoes and drive home so I’m not tracking in things I potentially had on me,” said Dr. Hopkins, who is married and a father to two college-aged sons and a daughter in fourth grade. “When I get home I immediately shower, and then I exercise or have dinner with my family.”
Despite the longer-than-usual work hours and upheaval to the traditional medical practice model brought on by the pandemic, Dr. Hopkins, a self-described “glass half full person,” said that he does his best to keep watch over his patients and colleagues. “I’m trying to keep an eye out on my team members – physicians, nurses, medical assistants, and folks at the front desk – trying to make sure that people are getting rest, trying to make sure that people are not overcommitting,” he said. “Because if we’re not all working together and working for the long term, we’re going to be in trouble. This is not going to be a sprint; this is going to be a marathon for us to get through.”
To keep mentally centered, he engages in at least 40 minutes of exercise each day on his bicycle or on the elliptical machine at home. Dr. Hopkins hopes that the current efforts to redeploy resources, expand clinician skill sets, and forge relationships with colleagues in other disciplines will carry over into the delivery of health care when COVID-19 is a distant memory. “I hope that some of those relationships are going to continue and result in better care for all of our patients,” he said.
"We are in dire need of hugs"
Typically, Dr. Dakkak, a family physician at Boston University, practices a mix of clinic-based family medicine and obstetrics, and works in inpatient medicine 6 weeks a year. Currently, she is leading a COVID-19 team full time at Boston Medical Center, a 300-bed safety-net hospital located on the campus of Boston University Medical Center.
COVID-19 has also shaken up her life at home.
When Dr. Dakkak volunteered to take on her new role, the first thing that came to her mind was how making the switch would affect the well-being of her 8-year-old son and 10-year-old daughter.
“I thought, ‘How do I get my children somewhere where I don’t have to worry about them?’ ” Dr. Dakkak said.
She floated the idea with her husband of flying their children out to stay with her recently retired parents, who live outside of Sacramento, Calif., until the pandemic eases up. “I was thinking to myself, ‘Am I overreacting? Is the pandemic not going to be that bad?’ because the rest of the country seemed to be in some amount of denial,” she said. “So, I called my dad, who’s a retired pediatric anesthesiologist. He’s from Egypt so he’s done crisis medicine in his time. He encouraged me to send the kids.”
On the same day that Dr. Dakkak began her first 12-hour COVID-19 shift at the hospital, her husband and children boarded a plane to California, where the kids remain in the care of her parents. Her husband returned after staying there for 2 weeks. “Every day when I’m working, I validate my decision,” she said. “When I first started, I worked 5 nights in a row, had 2 days off, and then worked 6 nights in a row. I was busy so I didn’t think about [being away from my kids], but at the same time I was grateful that I didn’t have to come home and worry about homeschooling the kids or infecting them.”
She checks in with them as she can via cell phone or FaceTime. “My son has been very honest,” Dr. Dakkak said. “He says, ‘FaceTime makes me miss you more, and I don’t like it,’ which I understand. I’ll call my mom, and if they want to talk to me, they’ll talk to me. If they don’t want to talk to me, I’m okay. This is about them being healthy and safe. I sent them a care package a few days ago with cards and some workbooks. I’m optimistic that in June I can at least see them if not bring them home.”
Dr. Dakkak describes leading a COVID-19 team as a grueling experience that challenges her medical know-how nearly every day, with seemingly ever-changing algorithms. “Our knowledge of this disease is five steps behind, and changing at lightning speed,” said Dr. Dakkak, who completed a fellowship in surgical and high-risk obstetrics. “It’s hard to balance continuing to teach evidence-based medicine for everything else in medicine [with continuing] to practice minimal and ever-changing evidence-based COVID medicine. We just don’t know enough [about the virus] yet. This is nothing like we were taught in medical school. Everyone has elevated d-dimers with COVID-19, and we don’t get CT pulmonary angiograms [CTPAs] on all of them; we wouldn’t physically be able to. Some patients have d-dimers in the thousands, and only some are stable to get CTPAs. We are also finding pulmonary embolisms. Now we’re basing our algorithm on anticoagulation due to d-dimers because sometimes you can’t always do a CTPA even if you want to. On the other hand, we have people who are coming into the hospital too late. We’ve had a few who have come in after having days of stroke symptoms. I worry about our patients at home who hesitate to come in when they really should.”
Sometimes she feels sad for the medical residents on her team because their instinct is to go in and check on each patient, “but I don’t want them to get exposed,” she said. “So, we check in by phone, or if they need a physical check-in, we minimize the check-ins; only one of us goes in. I’m more willing to put myself in the room than to put them in the room. I also feel for them because they came into medicine for the humanity of medicine – not the charting or the ordering of medicine. I also worry about the acuity and sadness they’re seeing. This is a rough introduction to medicine for them.”
When interviewed for this story in late April, Dr. Dakkak had kept track of her intubated COVID-19 patients. “Most of my patients get to go home without having been intubated, but those aren’t the ones I worry about,” she said. “I have two patients I have been watching. One of them has just been extubated and I’m still worried about him, but I’m hoping he’s going to be fine. The other one is the first pregnant woman we intubated. She is now extubated, doing really well, and went home. Her fetus is doing well, never had any issues while she was intubated. Those cases make me happy. They were both under the age of 35. It is nice to follow those intubations and find that the majority are doing okay.”
The first patient she had cared for who died was a young man “who was always in good spirits,” she recalled. “We called his brother right before intubating him. After intubation, his oxygen saturation didn’t jump up, which made me worry a bit.” About a week later, the young man died. “I kept thinking, ‘We intubated him when he was still comfortable talking. Should I have put it off and had him call more people to say goodbye? Should I have known that he wasn’t going to wake up?’ ” said Dr. Dakkak, who is also women’s health director at Manet Community Health Centers. “A lot of us have worked on our end-of-life discussions in the past month, just being able to tell somebody, ‘This might be your last time to call family. Call family and talk to whoever you want.’ Guilt isn’t the right word, but it’s unsettling if I’m the last person a patient talks to. I feel that, if that’s the case, then I didn’t do a good enough job trying to get them to their family or friends. If I am worried about a patient’s clinical status declining, I tell families now, when I call them, ‘I hope I’m wrong; I hope they don’t need to be intubated, but I think this is the time to talk.’ ”
To keep herself grounded during off hours, Dr. Dakkak spends time resting, checking in with her family, journaling “to get a lot of feelings out,” gardening, hiking, and joining Zoom chats with friends. Once recentered, she draws from a sense of obligation to others as she prepares for her next shift caring for COVID-19 patients.
“I have a lot of love for the world that I get to expend by doing this hard work,” she said. “I love humanity and I love humanity in times of crisis. The interactions I have with patients and their families are still central to why I do this work. I love my medical teams, and I would never want to let them down. It is nice to feel the sense of teamwork across the hospital. The nurses that I sit with and experience this with are amazing. I keep saying that the only thing I want to do when this pandemic is over is hug everyone. I think we are in dire need of hugs.”
Finding light in the darkness
Internist Katie Jobbins, DO, also has worked in a professional role that was created because of COVID-19.
Shortly before Dr. Jobbins was deployed to Baystate Medical Center in Springfield, Ma., for 2 weeks in April of 2020 to help clinicians with an anticipated surge of COVID-19 cases, she encountered a patient who walked into Baystate’s High Street Health Center.
“I think I have COVID-19,” the patient proclaimed to her, at the outpatient clinic that serves mostly inner-city, Medicaid patients.
Prior to becoming an ambulatory internist, Dr. Jobbins was a surgical resident. “So I went into that mode of ‘I need to do this, this, and this,’ ” she said. “I went through a checklist in my head to make sure I was prepared to take care of the patient.”
She applied that same systems approach during her redeployment assignment in the tertiary care hospital, which typically involved 10-hour shifts overseeing internal medicine residents in a medical telemetry unit. “We would take care of people under investigation for COVID-19, but we were not assigned to the actual COVID unit,” said Dr. Jobbins, who is also associate program director for the internal medicine residency program at the University of Massachusetts Medical School–Baystate Springfield. “They tried to redeploy other people to those units who had special training, and we were trying to back fill into where those people that got moved to the COVID units or the ICU units were actually working. We were taking more of the medical side of the floors.”
Even so, one patient on the unit was suspected of having COVID-19, so Dr. Jobbins suited up with personal protective equipment and conducted a thorough exam with residents waiting outside the patient’s room, a safe distance away. “I explained everything I found on the exam to the residents, trying to give them some educational benefit, even though they couldn’t physically examine the patient because we’re trying to protect them since they’re in training,” she said. “It was anxiety provoking, on some level, knowing that there’s a potential risk of exposure [to the virus], but knowing that Baystate Health has gone to extraordinary measures to make sure we have the correct PPE and support us is reassuring. I knew I had the right equipment and the right tools to take care of the patient, which calmed my nerves and made me feel like I could do the job. That’s the most important thing as a physician during this time: knowing that you have people supporting you who have your back at all times.”
Like Dr. Dakkak, Dr. Jobbins had to make some adjustments to her interaction with her family.
Before she began the deployment, Dr. Jobbins engaged in a frank discussion with her husband and her two young boys about the risks she faced working in a hospital caring for patients with COVID-19. “My husband and I made sure our wills were up to date, and we talked about what we would do if either of us got the virus,” she said. To minimize the potential risk of transmitting the virus to her loved ones during the two-week deployment, she considered living away from her family in a nearby home owned by her father, but decided against that and to “take it day by day.” Following her hospital shifts, Dr. Jobbins changed into a fresh set of clothes before leaving the hospital. Once she arrived home, she showered to reduce the risk of possibly becoming a vector to her family.
She had to tell her kids: “You can’t kiss me right now.”
“As much as it’s hard for them to understand, we had a conversation [in which I explained] ‘This is a virus. It will go away eventually, but it’s a virus we’re fighting.’ It’s interesting to watch a 3-year-old try to process that and take his play samurai sword or Marvel toys and decide he’s going to run around the neighborhood and try to kill the virus.”
At the High Street Health Center, Dr. Jobbins and her colleagues have transitioned to conducting most patient encounters via telephone or video appointments. “We have tried to maintain as much continuity for our patients to address their chronic medical needs through these visits, such as hypertension management and diabetes care,” she said. “We have begun a rigorous screening process to triage and treat patients suspicious for COVID-19 through telehealth in hopes of keeping them safe and in their own homes. We also continue to see patients for nonrespiratory urgent care needs in person once they have screened negative for COVID-19.”
“In terms of the inpatient setting, we’ve noticed that a lot of people are choosing not to go to the hospital now, unless they’re extremely ill,” Dr. Jobbins noted. “We’re going to need to find a balance with when do people truly need to go to the hospital and when do they not? What can we manage as an outpatient versus having someone go to the emergency department? That’s really the role of the primary care physician. We need to help people understand, ‘You don’t need to go to the ED for everything, but here are the things you really need to go for.’ ”
“It will be interesting to see what health care looks like in 6 months or a year. I’m excited to see where we land,” Dr. Jobbins added.
Hopes for the Future of Telemedicine
When the practice of medicine enters a post–COVID-19 era, Dr. Jobbins hopes that telemedicine will be incorporated more into the delivery of patient care. “I’ve found that many of my patients who often are no-shows to the inpatient version of their visits have had a higher success rate of follow-through when we do the telephone visits,” she said. “It’s been very successful. I hope that the insurance companies and [Centers for Medicare & Medicaid] will continue to reimburse this as they see this is a benefit to our patients.
Dr. Hopkins is also hopeful that physicians will be able to successfully see patients via telemedicine in the postpandemic world.
“For the ups and downs we’ve had with telemedicine, I’d love for us to be able to enhance the positives and incorporate that into our practice going forward. If we can reach our patients and help treat them where they are, rather than them having to come to us, that may be a plus,” he said.
In the meantime, Dr. Jobbins presses on as the curve of COVID-19 cases flattens in Western Massachusetts and remains grateful that she chose to practice medicine.
“The commitment I have to being an educator in addition to being a physician is part of why I keep doing this,” Dr. Jobbins said. “I find this to be one of the most fulfilling jobs and careers you could ever have: being there for people when they need you the most. That’s really what a physician’s job is: being there for people when a family member has passed away or when they just need to talk because they’re having anxiety. At the end of the day, if we can impart that to those we work with and bring in a positive attitude, it’s infectious and it makes people see this is a reason we keep doing what we’re doing.”
She’s also been heartened by the kindness of strangers during this pandemic, from those who made and donated face shields when they were in short supply, to those who delivered food to the hospital as a gesture of thanks.
“I had a patient who made homemade masks and sent them to my office,” she said. “There’s obviously good and bad during this time, but I get hope from seeing all of the good things that are coming out of this, the whole idea of finding the light in the darkness.”
During his shift at a COVID-19 drive-through triage screening area set up outside the University of Arkansas for Medical Sciences in Little Rock, Robert Hopkins Jr., MD, noticed a woman bowled over in the front seat of her car.
A nurse practitioner had just informed her that she had met the criteria for undergoing testing for the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).
“She was very upset and was crying nearly inconsolably,” said Dr. Hopkins, who directs the division of general internal medicine at the University of Arkansas Medical Sciences College of Medicine. “I went over and visited with her for a few minutes. She was scared to death that we [had] told her she was going to die. In her mind, if she had COVID-19 that meant a death sentence, and if we were testing her that meant she was likely to not survive.”
Dr. Hopkins tried his best to put testing in perspective for the woman. “At least she came to a level of comfort and realized that we were doing this for her, that this was not a death sentence, that this was not her fault,” he said. “She was worried about infecting her kids and her grandkids and ending up in the hospital and being a burden. Being able to spend that few minutes with her and help to bring down her level of anxiety – I think that’s where we need to put our efforts as physicians right now, helping people understand, ‘Yes, this is serious. Yes, we need to continue to social distance. Yes, we need to be cautious. But, we will get through this if we all work together to do so.’ ”
Prior to the COVID-19 pandemic, Dr. Hopkins spent part of his time seeing patients in the university’s main hospital, but most of it in an outpatient clinic where he and about 20 other primary care physicians care for patients and precept medical residents. Now, medical residents have been deployed to other services, primarily in the hospital, and he and his physician colleagues are conducting 80%-90% of patient visits by video conferencing or by telephone. It’s a whole new world.
“We’ve gone from a relatively traditional inpatient/outpatient practice where we’re seeing patients face to face to doing some face-to-face visits, but an awful lot of what we do now is in the technology domain,” said Dr. Hopkins, who also assisted with health care relief efforts during hurricanes Rita and Katrina.
“A group of six of us has been redeployed to assist with the surge unit for the inpatient facility, so our outpatient duties are being taken on by some of our partners.”
He also pitches in at the drive-through COVID-19 screening clinic, which was set up on March 27 and operates between 8 a.m. and 8 p.m., 7 days a week. “We’re able to measure people’s temperature, take a quick screening history, decide whether their risk is such that we need to do a COVID-19 PCR [polymerase chain reaction] test,” he said. “Then we make a determination of whether they need to go home on quarantine awaiting those results, or if they don’t have anything that needs to be evaluated, or whether they need to be triaged to an urgent care setting or to the emergency department.”
To minimize his risk of acquiring COVID-19, he follows personal hygiene practices recommended by the Centers for Disease Control and Prevention. He also places his work shoes in a shoebox, which he keeps in his car. “I put them on when I get to the parking deck at work, do my work, and then I put them in the shoebox, slip on another pair of shoes and drive home so I’m not tracking in things I potentially had on me,” said Dr. Hopkins, who is married and a father to two college-aged sons and a daughter in fourth grade. “When I get home I immediately shower, and then I exercise or have dinner with my family.”
Despite the longer-than-usual work hours and upheaval to the traditional medical practice model brought on by the pandemic, Dr. Hopkins, a self-described “glass half full person,” said that he does his best to keep watch over his patients and colleagues. “I’m trying to keep an eye out on my team members – physicians, nurses, medical assistants, and folks at the front desk – trying to make sure that people are getting rest, trying to make sure that people are not overcommitting,” he said. “Because if we’re not all working together and working for the long term, we’re going to be in trouble. This is not going to be a sprint; this is going to be a marathon for us to get through.”
To keep mentally centered, he engages in at least 40 minutes of exercise each day on his bicycle or on the elliptical machine at home. Dr. Hopkins hopes that the current efforts to redeploy resources, expand clinician skill sets, and forge relationships with colleagues in other disciplines will carry over into the delivery of health care when COVID-19 is a distant memory. “I hope that some of those relationships are going to continue and result in better care for all of our patients,” he said.
"We are in dire need of hugs"
Typically, Dr. Dakkak, a family physician at Boston University, practices a mix of clinic-based family medicine and obstetrics, and works in inpatient medicine 6 weeks a year. Currently, she is leading a COVID-19 team full time at Boston Medical Center, a 300-bed safety-net hospital located on the campus of Boston University Medical Center.
COVID-19 has also shaken up her life at home.
When Dr. Dakkak volunteered to take on her new role, the first thing that came to her mind was how making the switch would affect the well-being of her 8-year-old son and 10-year-old daughter.
“I thought, ‘How do I get my children somewhere where I don’t have to worry about them?’ ” Dr. Dakkak said.
She floated the idea with her husband of flying their children out to stay with her recently retired parents, who live outside of Sacramento, Calif., until the pandemic eases up. “I was thinking to myself, ‘Am I overreacting? Is the pandemic not going to be that bad?’ because the rest of the country seemed to be in some amount of denial,” she said. “So, I called my dad, who’s a retired pediatric anesthesiologist. He’s from Egypt so he’s done crisis medicine in his time. He encouraged me to send the kids.”
On the same day that Dr. Dakkak began her first 12-hour COVID-19 shift at the hospital, her husband and children boarded a plane to California, where the kids remain in the care of her parents. Her husband returned after staying there for 2 weeks. “Every day when I’m working, I validate my decision,” she said. “When I first started, I worked 5 nights in a row, had 2 days off, and then worked 6 nights in a row. I was busy so I didn’t think about [being away from my kids], but at the same time I was grateful that I didn’t have to come home and worry about homeschooling the kids or infecting them.”
She checks in with them as she can via cell phone or FaceTime. “My son has been very honest,” Dr. Dakkak said. “He says, ‘FaceTime makes me miss you more, and I don’t like it,’ which I understand. I’ll call my mom, and if they want to talk to me, they’ll talk to me. If they don’t want to talk to me, I’m okay. This is about them being healthy and safe. I sent them a care package a few days ago with cards and some workbooks. I’m optimistic that in June I can at least see them if not bring them home.”
Dr. Dakkak describes leading a COVID-19 team as a grueling experience that challenges her medical know-how nearly every day, with seemingly ever-changing algorithms. “Our knowledge of this disease is five steps behind, and changing at lightning speed,” said Dr. Dakkak, who completed a fellowship in surgical and high-risk obstetrics. “It’s hard to balance continuing to teach evidence-based medicine for everything else in medicine [with continuing] to practice minimal and ever-changing evidence-based COVID medicine. We just don’t know enough [about the virus] yet. This is nothing like we were taught in medical school. Everyone has elevated d-dimers with COVID-19, and we don’t get CT pulmonary angiograms [CTPAs] on all of them; we wouldn’t physically be able to. Some patients have d-dimers in the thousands, and only some are stable to get CTPAs. We are also finding pulmonary embolisms. Now we’re basing our algorithm on anticoagulation due to d-dimers because sometimes you can’t always do a CTPA even if you want to. On the other hand, we have people who are coming into the hospital too late. We’ve had a few who have come in after having days of stroke symptoms. I worry about our patients at home who hesitate to come in when they really should.”
Sometimes she feels sad for the medical residents on her team because their instinct is to go in and check on each patient, “but I don’t want them to get exposed,” she said. “So, we check in by phone, or if they need a physical check-in, we minimize the check-ins; only one of us goes in. I’m more willing to put myself in the room than to put them in the room. I also feel for them because they came into medicine for the humanity of medicine – not the charting or the ordering of medicine. I also worry about the acuity and sadness they’re seeing. This is a rough introduction to medicine for them.”
When interviewed for this story in late April, Dr. Dakkak had kept track of her intubated COVID-19 patients. “Most of my patients get to go home without having been intubated, but those aren’t the ones I worry about,” she said. “I have two patients I have been watching. One of them has just been extubated and I’m still worried about him, but I’m hoping he’s going to be fine. The other one is the first pregnant woman we intubated. She is now extubated, doing really well, and went home. Her fetus is doing well, never had any issues while she was intubated. Those cases make me happy. They were both under the age of 35. It is nice to follow those intubations and find that the majority are doing okay.”
The first patient she had cared for who died was a young man “who was always in good spirits,” she recalled. “We called his brother right before intubating him. After intubation, his oxygen saturation didn’t jump up, which made me worry a bit.” About a week later, the young man died. “I kept thinking, ‘We intubated him when he was still comfortable talking. Should I have put it off and had him call more people to say goodbye? Should I have known that he wasn’t going to wake up?’ ” said Dr. Dakkak, who is also women’s health director at Manet Community Health Centers. “A lot of us have worked on our end-of-life discussions in the past month, just being able to tell somebody, ‘This might be your last time to call family. Call family and talk to whoever you want.’ Guilt isn’t the right word, but it’s unsettling if I’m the last person a patient talks to. I feel that, if that’s the case, then I didn’t do a good enough job trying to get them to their family or friends. If I am worried about a patient’s clinical status declining, I tell families now, when I call them, ‘I hope I’m wrong; I hope they don’t need to be intubated, but I think this is the time to talk.’ ”
To keep herself grounded during off hours, Dr. Dakkak spends time resting, checking in with her family, journaling “to get a lot of feelings out,” gardening, hiking, and joining Zoom chats with friends. Once recentered, she draws from a sense of obligation to others as she prepares for her next shift caring for COVID-19 patients.
“I have a lot of love for the world that I get to expend by doing this hard work,” she said. “I love humanity and I love humanity in times of crisis. The interactions I have with patients and their families are still central to why I do this work. I love my medical teams, and I would never want to let them down. It is nice to feel the sense of teamwork across the hospital. The nurses that I sit with and experience this with are amazing. I keep saying that the only thing I want to do when this pandemic is over is hug everyone. I think we are in dire need of hugs.”
Finding light in the darkness
Internist Katie Jobbins, DO, also has worked in a professional role that was created because of COVID-19.
Shortly before Dr. Jobbins was deployed to Baystate Medical Center in Springfield, Ma., for 2 weeks in April of 2020 to help clinicians with an anticipated surge of COVID-19 cases, she encountered a patient who walked into Baystate’s High Street Health Center.
“I think I have COVID-19,” the patient proclaimed to her, at the outpatient clinic that serves mostly inner-city, Medicaid patients.
Prior to becoming an ambulatory internist, Dr. Jobbins was a surgical resident. “So I went into that mode of ‘I need to do this, this, and this,’ ” she said. “I went through a checklist in my head to make sure I was prepared to take care of the patient.”
She applied that same systems approach during her redeployment assignment in the tertiary care hospital, which typically involved 10-hour shifts overseeing internal medicine residents in a medical telemetry unit. “We would take care of people under investigation for COVID-19, but we were not assigned to the actual COVID unit,” said Dr. Jobbins, who is also associate program director for the internal medicine residency program at the University of Massachusetts Medical School–Baystate Springfield. “They tried to redeploy other people to those units who had special training, and we were trying to back fill into where those people that got moved to the COVID units or the ICU units were actually working. We were taking more of the medical side of the floors.”
Even so, one patient on the unit was suspected of having COVID-19, so Dr. Jobbins suited up with personal protective equipment and conducted a thorough exam with residents waiting outside the patient’s room, a safe distance away. “I explained everything I found on the exam to the residents, trying to give them some educational benefit, even though they couldn’t physically examine the patient because we’re trying to protect them since they’re in training,” she said. “It was anxiety provoking, on some level, knowing that there’s a potential risk of exposure [to the virus], but knowing that Baystate Health has gone to extraordinary measures to make sure we have the correct PPE and support us is reassuring. I knew I had the right equipment and the right tools to take care of the patient, which calmed my nerves and made me feel like I could do the job. That’s the most important thing as a physician during this time: knowing that you have people supporting you who have your back at all times.”
Like Dr. Dakkak, Dr. Jobbins had to make some adjustments to her interaction with her family.
Before she began the deployment, Dr. Jobbins engaged in a frank discussion with her husband and her two young boys about the risks she faced working in a hospital caring for patients with COVID-19. “My husband and I made sure our wills were up to date, and we talked about what we would do if either of us got the virus,” she said. To minimize the potential risk of transmitting the virus to her loved ones during the two-week deployment, she considered living away from her family in a nearby home owned by her father, but decided against that and to “take it day by day.” Following her hospital shifts, Dr. Jobbins changed into a fresh set of clothes before leaving the hospital. Once she arrived home, she showered to reduce the risk of possibly becoming a vector to her family.
She had to tell her kids: “You can’t kiss me right now.”
“As much as it’s hard for them to understand, we had a conversation [in which I explained] ‘This is a virus. It will go away eventually, but it’s a virus we’re fighting.’ It’s interesting to watch a 3-year-old try to process that and take his play samurai sword or Marvel toys and decide he’s going to run around the neighborhood and try to kill the virus.”
At the High Street Health Center, Dr. Jobbins and her colleagues have transitioned to conducting most patient encounters via telephone or video appointments. “We have tried to maintain as much continuity for our patients to address their chronic medical needs through these visits, such as hypertension management and diabetes care,” she said. “We have begun a rigorous screening process to triage and treat patients suspicious for COVID-19 through telehealth in hopes of keeping them safe and in their own homes. We also continue to see patients for nonrespiratory urgent care needs in person once they have screened negative for COVID-19.”
“In terms of the inpatient setting, we’ve noticed that a lot of people are choosing not to go to the hospital now, unless they’re extremely ill,” Dr. Jobbins noted. “We’re going to need to find a balance with when do people truly need to go to the hospital and when do they not? What can we manage as an outpatient versus having someone go to the emergency department? That’s really the role of the primary care physician. We need to help people understand, ‘You don’t need to go to the ED for everything, but here are the things you really need to go for.’ ”
“It will be interesting to see what health care looks like in 6 months or a year. I’m excited to see where we land,” Dr. Jobbins added.
Hopes for the Future of Telemedicine
When the practice of medicine enters a post–COVID-19 era, Dr. Jobbins hopes that telemedicine will be incorporated more into the delivery of patient care. “I’ve found that many of my patients who often are no-shows to the inpatient version of their visits have had a higher success rate of follow-through when we do the telephone visits,” she said. “It’s been very successful. I hope that the insurance companies and [Centers for Medicare & Medicaid] will continue to reimburse this as they see this is a benefit to our patients.
Dr. Hopkins is also hopeful that physicians will be able to successfully see patients via telemedicine in the postpandemic world.
“For the ups and downs we’ve had with telemedicine, I’d love for us to be able to enhance the positives and incorporate that into our practice going forward. If we can reach our patients and help treat them where they are, rather than them having to come to us, that may be a plus,” he said.
In the meantime, Dr. Jobbins presses on as the curve of COVID-19 cases flattens in Western Massachusetts and remains grateful that she chose to practice medicine.
“The commitment I have to being an educator in addition to being a physician is part of why I keep doing this,” Dr. Jobbins said. “I find this to be one of the most fulfilling jobs and careers you could ever have: being there for people when they need you the most. That’s really what a physician’s job is: being there for people when a family member has passed away or when they just need to talk because they’re having anxiety. At the end of the day, if we can impart that to those we work with and bring in a positive attitude, it’s infectious and it makes people see this is a reason we keep doing what we’re doing.”
She’s also been heartened by the kindness of strangers during this pandemic, from those who made and donated face shields when they were in short supply, to those who delivered food to the hospital as a gesture of thanks.
“I had a patient who made homemade masks and sent them to my office,” she said. “There’s obviously good and bad during this time, but I get hope from seeing all of the good things that are coming out of this, the whole idea of finding the light in the darkness.”