User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
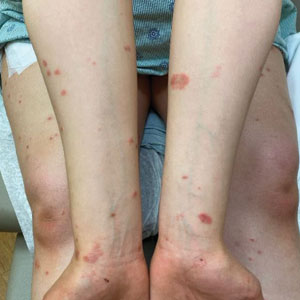
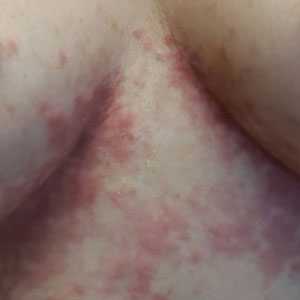
Can this tool forecast peanut allergies?
Pediatricians may have a new aid to better predict peanut allergies among infants with atopic dermatitis.
Their study of the implementation of the scorecard was presented at the Pediatric Academic Societies annual meeting.
Infants with atopic dermatitis or eczema are six times more likely to have an egg allergy and eleven times more likely to have a peanut allergy at age 12 months than are infants without atopic dermatitis.
The scorecard reflects recent directives from the National Institute of Allergy and Infectious Diseases to help combat the public health problem.
“When the NIAID prevention of peanut allergy guidelines first came out, it asked pediatricians to serve as frontline practitioners in implementing them by identifying children at risk for peanut allergy and guiding families on what to do next,” said Waheeda Samady, MD, professor of pediatrics at Northwestern University, Chicago. “The impetus for the study was to further support pediatricians in this role.”
Although pediatricians are trained to identify and even treat mild to moderate cases of atopic dermatitis, little emphasis has gone to categorizing the condition on the basis of severity and to correlating peanut allergy risk.
The predictive scorecard captures 14 images from one infant of mixed race, two White infants, two Black infants, and two Hispanic infants.
To create the card, two in-house pediatric dermatologists assessed 58 images from 13 children and categorized images from 0 (no signs of atopic dermatitis) to 4 (severe signs of atopic dermatitis). After a first pass on categorization, the doctors agreed on 84% of images.
Of 189 pediatricians who used the card, fewer than half reported that they “sometimes,” “very often,” or “always” used the scorecard for atopic dermatitis evaluation. A little fewer than three-quarters reported that their ability to diagnose and categorize atopic dermatitis improved.
“Severity staging of atopic dermatitis is not something that the general pediatrician necessarily performs on a day-to-day basis,” said Kawaljit Brar, MD, professor of pediatrics in the division of allergy and immunology at Hassenfeld Children’s Hospital in New York.
Dr. Brar explained that children who are identified as being at high risk are often referred to specialists such as her, who then perform allergy screenings and can determine whether introduction of food at home is safe or whether office feedings supervised by an allergist are necessary. Researchers have found that early introduction to peanuts for children with moderate to severe atopic dermatitis could prevent peanut allergy.
“This represents a wonderful initiative to educate pediatricians so that they understand which patients require screening for peanut allergy and which patients don’t and can just get introduced to peanuts at home,” Dr. Brar said.
The atopic dermatitis scorecard reflects a growing recognition that varying skin tones show levels of severity incongruously.
“Many of us in clinical practice have recognized that our education has not always been inclusive of patients with varying skin tones,” Dr. Samady said. “When we looked for photos of patients with different skin tones, we simply could not find any that we thought were appropriate. So we decided to take some ourselves, and we’re currently continuing to take photos in order to improve the scorecard we currently have.”
The study was funded by the National Institute of Health and Food Allergy Research and Education. Dr. Samady and Dr. Brar reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pediatricians may have a new aid to better predict peanut allergies among infants with atopic dermatitis.
Their study of the implementation of the scorecard was presented at the Pediatric Academic Societies annual meeting.
Infants with atopic dermatitis or eczema are six times more likely to have an egg allergy and eleven times more likely to have a peanut allergy at age 12 months than are infants without atopic dermatitis.
The scorecard reflects recent directives from the National Institute of Allergy and Infectious Diseases to help combat the public health problem.
“When the NIAID prevention of peanut allergy guidelines first came out, it asked pediatricians to serve as frontline practitioners in implementing them by identifying children at risk for peanut allergy and guiding families on what to do next,” said Waheeda Samady, MD, professor of pediatrics at Northwestern University, Chicago. “The impetus for the study was to further support pediatricians in this role.”
Although pediatricians are trained to identify and even treat mild to moderate cases of atopic dermatitis, little emphasis has gone to categorizing the condition on the basis of severity and to correlating peanut allergy risk.
The predictive scorecard captures 14 images from one infant of mixed race, two White infants, two Black infants, and two Hispanic infants.
To create the card, two in-house pediatric dermatologists assessed 58 images from 13 children and categorized images from 0 (no signs of atopic dermatitis) to 4 (severe signs of atopic dermatitis). After a first pass on categorization, the doctors agreed on 84% of images.
Of 189 pediatricians who used the card, fewer than half reported that they “sometimes,” “very often,” or “always” used the scorecard for atopic dermatitis evaluation. A little fewer than three-quarters reported that their ability to diagnose and categorize atopic dermatitis improved.
“Severity staging of atopic dermatitis is not something that the general pediatrician necessarily performs on a day-to-day basis,” said Kawaljit Brar, MD, professor of pediatrics in the division of allergy and immunology at Hassenfeld Children’s Hospital in New York.
Dr. Brar explained that children who are identified as being at high risk are often referred to specialists such as her, who then perform allergy screenings and can determine whether introduction of food at home is safe or whether office feedings supervised by an allergist are necessary. Researchers have found that early introduction to peanuts for children with moderate to severe atopic dermatitis could prevent peanut allergy.
“This represents a wonderful initiative to educate pediatricians so that they understand which patients require screening for peanut allergy and which patients don’t and can just get introduced to peanuts at home,” Dr. Brar said.
The atopic dermatitis scorecard reflects a growing recognition that varying skin tones show levels of severity incongruously.
“Many of us in clinical practice have recognized that our education has not always been inclusive of patients with varying skin tones,” Dr. Samady said. “When we looked for photos of patients with different skin tones, we simply could not find any that we thought were appropriate. So we decided to take some ourselves, and we’re currently continuing to take photos in order to improve the scorecard we currently have.”
The study was funded by the National Institute of Health and Food Allergy Research and Education. Dr. Samady and Dr. Brar reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pediatricians may have a new aid to better predict peanut allergies among infants with atopic dermatitis.
Their study of the implementation of the scorecard was presented at the Pediatric Academic Societies annual meeting.
Infants with atopic dermatitis or eczema are six times more likely to have an egg allergy and eleven times more likely to have a peanut allergy at age 12 months than are infants without atopic dermatitis.
The scorecard reflects recent directives from the National Institute of Allergy and Infectious Diseases to help combat the public health problem.
“When the NIAID prevention of peanut allergy guidelines first came out, it asked pediatricians to serve as frontline practitioners in implementing them by identifying children at risk for peanut allergy and guiding families on what to do next,” said Waheeda Samady, MD, professor of pediatrics at Northwestern University, Chicago. “The impetus for the study was to further support pediatricians in this role.”
Although pediatricians are trained to identify and even treat mild to moderate cases of atopic dermatitis, little emphasis has gone to categorizing the condition on the basis of severity and to correlating peanut allergy risk.
The predictive scorecard captures 14 images from one infant of mixed race, two White infants, two Black infants, and two Hispanic infants.
To create the card, two in-house pediatric dermatologists assessed 58 images from 13 children and categorized images from 0 (no signs of atopic dermatitis) to 4 (severe signs of atopic dermatitis). After a first pass on categorization, the doctors agreed on 84% of images.
Of 189 pediatricians who used the card, fewer than half reported that they “sometimes,” “very often,” or “always” used the scorecard for atopic dermatitis evaluation. A little fewer than three-quarters reported that their ability to diagnose and categorize atopic dermatitis improved.
“Severity staging of atopic dermatitis is not something that the general pediatrician necessarily performs on a day-to-day basis,” said Kawaljit Brar, MD, professor of pediatrics in the division of allergy and immunology at Hassenfeld Children’s Hospital in New York.
Dr. Brar explained that children who are identified as being at high risk are often referred to specialists such as her, who then perform allergy screenings and can determine whether introduction of food at home is safe or whether office feedings supervised by an allergist are necessary. Researchers have found that early introduction to peanuts for children with moderate to severe atopic dermatitis could prevent peanut allergy.
“This represents a wonderful initiative to educate pediatricians so that they understand which patients require screening for peanut allergy and which patients don’t and can just get introduced to peanuts at home,” Dr. Brar said.
The atopic dermatitis scorecard reflects a growing recognition that varying skin tones show levels of severity incongruously.
“Many of us in clinical practice have recognized that our education has not always been inclusive of patients with varying skin tones,” Dr. Samady said. “When we looked for photos of patients with different skin tones, we simply could not find any that we thought were appropriate. So we decided to take some ourselves, and we’re currently continuing to take photos in order to improve the scorecard we currently have.”
The study was funded by the National Institute of Health and Food Allergy Research and Education. Dr. Samady and Dr. Brar reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PAS 2023
Medical students gain momentum in effort to ban legacy admissions
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
, which they say offer preferential treatment to applicants based on their association with donors or alumni.
While an estimated 25% of public colleges and universities still use legacy admissions, a growing list of top medical schools have moved away from the practice over the last decade, including Johns Hopkins University, Baltimore, and Tufts University, Medford, Mass.
Legacy admissions contradict schools’ more inclusive policies, Senila Yasmin, MPH, a second-year medical student at Tufts University, said in an interview. While Tufts maintains legacy admissions for its undergraduate applicants, the medical school stopped the practice in 2021, said Ms. Yasmin, a member of a student group that lobbied against the school’s legacy preferences.
Describing herself as a low-income, first-generation Muslim-Pakistani American, Ms. Yasmin wants to use her experience at Tufts to improve accessibility for students like herself.
As a member of the American Medical Association (AMA) Medical Student Section, she coauthored a resolution stating that legacy admissions go against the AMA’s strategic plan to advance racial justice and health equity. The Student Section passed the resolution in November, and in June, the AMA House of Delegates will vote on whether to adopt the policy.
Along with a Supreme Court decision that could strike down race-conscious college admissions, an AMA policy could convince medical schools to rethink legacy admissions and how to maintain diverse student bodies. In June, the court is expected to issue a decision in the Students for Fair Admissions lawsuit against Harvard University, Cambridge, Mass., and the University of North Carolina, Chapel Hill, which alleges that considering race in holistic admissions constitutes racial discrimination and violates the Equal Protection Clause.
Opponents of legacy admissions, like Ms. Yasmin, say it penalizes students from racial minorities and lower socioeconomic backgrounds, hampering a fair and equitable admissions process that attracts diverse medical school admissions.
Diversity of medical applicants
Diversity in medical schools continued to increase last year with more Black, Hispanic, and female students applying and enrolling, according to a recent report by the Association of American Medical Colleges (AAMC). However, universities often include nonacademic criteria in their admission assessments to improve educational access for underrepresented minorities.
Medical schools carefully consider each applicant’s background “to yield a diverse class of students,” Geoffrey Young, PhD, AAMC’s senior director of transforming the health care workforce, told this news organization.
Some schools, such as Morehouse School of Medicine, Atlanta, the University of Virginia School of Medicine, Charlottesville, and the University of Arizona College of Medicine, Tucson, perform a thorough review of candidates while offering admissions practices designed specifically for legacy applicants. The schools assert that legacy designation doesn’t factor into the student’s likelihood of acceptance.
The arrangement may show that schools want to commit to equity and fairness but have trouble moving away from entrenched traditions, two professors from Penn State College of Medicine, Hershey, Pa., who sit on separate medical admissions subcommittees, wrote last year in Bioethics Today.
Legislation may hasten legacies’ end
In December, Ms. Yasmin and a group of Massachusetts Medical Society student-members presented another resolution to the state medical society, which adopted it.
The society’s new policy opposes the use of legacy status in medical school admissions and supports mechanisms to eliminate its inclusion from the application process, Theodore Calianos II, MD, FACS, president of the Massachusetts Medical Society, said in an interview.
“Legacy preferences limit racial and socioeconomic diversity on campuses, so we asked, ‘What can we do so that everyone has equal access to medical education?’ It is exciting to see the students and young physicians – the future of medicine – become involved in policymaking.”
Proposed laws may also hasten the end of legacy admissions. Last year, the U.S. Senate began considering a bill prohibiting colleges receiving federal financial aid from giving preferential treatment to students based on their relations to donors or alumni. However, the bill allows the Department of Education to make exceptions for institutions serving historically underrepresented groups.
The New York State Senate and the New York State Assembly also are reviewing bills that ban legacy and early admissions policies at public and private universities. Connecticut announced similar legislation last year. Massachusetts legislators are considering two bills: one that would ban the practice at the state’s public universities and another that would require all schools using legacy status to pay a “public service fee” equal to a percentage of its endowment. Colleges with endowment assets exceeding $2 billion must pay at least $2 million, according to the bill’s text.
At schools like Harvard, whose endowment surpasses $50 billion, the option to pay the penalty will make the law moot, Michael Walls, DO, MPH, president of the American Medical Student Association (AMSA), said in an interview. “Smaller schools wouldn’t be able to afford the fine and are less likely to be doing [legacy admissions] anyway,” he said. “The schools that want to continue doing it could just pay the fine.”
Dr. Walls said AMSA supports race-conscious admissions processes and anything that increases fairness for medical school applicants. “Whatever [fair] means is up for interpretation, but it would be great to eliminate legacy admissions,” he said.
A version of this article originally appeared on Medscape.com.
Picosecond laser applications continue to expand
PHOENIX – Ever since PicoSure became the first picosecond laser cleared by the Food and Drug Administration for the treatment of unwanted tattoos and pigmented lesions in 2012, new uses for this technology continue to expand.
Now, These include PicoWay, PicoSure, Enlighten, PicoPlus, PiQo4, and Quanta Pico, among others.
“PicoWay technology has integrated nicely into my practice in Houston, the most ethnically diverse city in the country, with its ability to safely treat a number of various benign, congenital, and acquired epidermal and dermal pigmented lesions with ultrashort pulse duration and low thermal impact, which greatly reduces the risk of postinflammatory hyperpigmentation even in darker skin types,” Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center, Houston, said at the annual conference of the American Society for Laser Medicine and Surgery.
He emphasized the importance of therapeutic clinical endpoints, noting that with q-switched lasers, “you’re looking for immediate whitening, whereas with picosecond lasers, your endpoint is slight whitening or slight darkening depending on wavelength, indication, and skin type. The ability to fractionate picosecond pulses has also allowed us to utilize this technology for photoaging as well as acne scarring.”
The PicoWay system includes a 730-nm picosecond titanium sapphire handpiece, which is FDA cleared for treatment of benign pigmented lesions and blue and green tattoo removal. Dr. Friedman said that he has seen good clinical results using the handpiece for café-au-lait macules, particularly in skin of color.
In an abstract presented at the ASLMS meeting, he and his colleagues presented a retrospective review of 12 patients with café-au-lait macules with Fitzpatrick skin types III-VI who were treated with the PicoWay 730 nm handpiece between April 2021 and January 2023. Patients received a mean of 3.1 treatments at intervals that ranged from 5 to 40 weeks. Clinical photographs were graded by three board-certified dermatologists using a 5-point visual analogue scale.
Overall, patients were rated to have a mean improvement of 26%-50%. Two patients achieved 100% clearance after four to five treatment sessions. “Café-au-lait macules with smooth borders responded less well to laser treatment, confirming prior studies at our center,” he said. “We often educate parents that café-au-lait macules may recur over time, especially with repeated sun exposure.”
Treating melasma
Dr. Friedman’s go-to devices for melasma include the low-density, low-energy 1,927-nm fractional diode laser; the 1,064 nm picosecond Nd:YAG, the low-fluence 1,064 nm Q-switched Nd:YAG with a nanosecond pulse duration, and the 595-nm pulsed dye laser for lesions exhibiting underlying vascularity. He said that combining therapies that target pigment and vasculature may be ideal to prevent relapses. “Melasma is a multifactorial condition so by improving patient education and expectation alongside advances in laser treatment of melasma, we have ultimately improved our ability to treat this condition,” he said.
“We’re approaching it from all angles, with ultraviolet photography and spectrocolorimetry, behavioral modifications, topical skin-lightening agents, broad spectrum sunscreens with protection against visible light, and oral tranexamic acid in advanced cases. Then, we intervene with these energy-based modalities, and the bottom line is, less energy and density is more, with lengthened treatment intervals. In 2023, we’re better than we’ve ever been in terms of our ability to safely and effectively improve melasma.”
Novel lasers
Dr. Friedman also described the UltraClear, a novel ablative fractional 2,910-nm erbium-doped glass fiber laser that delivers a customized blend of ablation and coagulation based on the patient’s condition, skin type, and tolerability for down time. He provided an overview of the versatility of what he described as highly customizable technology for conditions such as photoaging and dyschromia in patients of various skin types, making it a very versatile platform in his practice.
The AVAVA MIRIA system is a “next generation” laser “where you’re able to use a focal point. Basically, you’re treating the skin from the inside out in a 3D manner and you’re able to focus intradermally up to 1 mm with high energy 1,064 nm or 1,550 nm,” he said. “It’s a unique conical geometry that spares the epidermis, combined with sapphire tip cooling and images the skin at the same time with the potential for personalized treatments of dyschromia and photoaging in all skin types. It’s truly remarkable where the technology is heading.”
Dr. Friedman disclosed that he has received consulting fees from Allergan, Galderma, Acclaro, Merz Aesthetics, Solta Medical, and Cytrellis. He has conducted contracted research for Sofwave and is a member of the speakers bureau for Solta Medical and Candela.
PHOENIX – Ever since PicoSure became the first picosecond laser cleared by the Food and Drug Administration for the treatment of unwanted tattoos and pigmented lesions in 2012, new uses for this technology continue to expand.
Now, These include PicoWay, PicoSure, Enlighten, PicoPlus, PiQo4, and Quanta Pico, among others.
“PicoWay technology has integrated nicely into my practice in Houston, the most ethnically diverse city in the country, with its ability to safely treat a number of various benign, congenital, and acquired epidermal and dermal pigmented lesions with ultrashort pulse duration and low thermal impact, which greatly reduces the risk of postinflammatory hyperpigmentation even in darker skin types,” Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center, Houston, said at the annual conference of the American Society for Laser Medicine and Surgery.
He emphasized the importance of therapeutic clinical endpoints, noting that with q-switched lasers, “you’re looking for immediate whitening, whereas with picosecond lasers, your endpoint is slight whitening or slight darkening depending on wavelength, indication, and skin type. The ability to fractionate picosecond pulses has also allowed us to utilize this technology for photoaging as well as acne scarring.”
The PicoWay system includes a 730-nm picosecond titanium sapphire handpiece, which is FDA cleared for treatment of benign pigmented lesions and blue and green tattoo removal. Dr. Friedman said that he has seen good clinical results using the handpiece for café-au-lait macules, particularly in skin of color.
In an abstract presented at the ASLMS meeting, he and his colleagues presented a retrospective review of 12 patients with café-au-lait macules with Fitzpatrick skin types III-VI who were treated with the PicoWay 730 nm handpiece between April 2021 and January 2023. Patients received a mean of 3.1 treatments at intervals that ranged from 5 to 40 weeks. Clinical photographs were graded by three board-certified dermatologists using a 5-point visual analogue scale.
Overall, patients were rated to have a mean improvement of 26%-50%. Two patients achieved 100% clearance after four to five treatment sessions. “Café-au-lait macules with smooth borders responded less well to laser treatment, confirming prior studies at our center,” he said. “We often educate parents that café-au-lait macules may recur over time, especially with repeated sun exposure.”
Treating melasma
Dr. Friedman’s go-to devices for melasma include the low-density, low-energy 1,927-nm fractional diode laser; the 1,064 nm picosecond Nd:YAG, the low-fluence 1,064 nm Q-switched Nd:YAG with a nanosecond pulse duration, and the 595-nm pulsed dye laser for lesions exhibiting underlying vascularity. He said that combining therapies that target pigment and vasculature may be ideal to prevent relapses. “Melasma is a multifactorial condition so by improving patient education and expectation alongside advances in laser treatment of melasma, we have ultimately improved our ability to treat this condition,” he said.
“We’re approaching it from all angles, with ultraviolet photography and spectrocolorimetry, behavioral modifications, topical skin-lightening agents, broad spectrum sunscreens with protection against visible light, and oral tranexamic acid in advanced cases. Then, we intervene with these energy-based modalities, and the bottom line is, less energy and density is more, with lengthened treatment intervals. In 2023, we’re better than we’ve ever been in terms of our ability to safely and effectively improve melasma.”
Novel lasers
Dr. Friedman also described the UltraClear, a novel ablative fractional 2,910-nm erbium-doped glass fiber laser that delivers a customized blend of ablation and coagulation based on the patient’s condition, skin type, and tolerability for down time. He provided an overview of the versatility of what he described as highly customizable technology for conditions such as photoaging and dyschromia in patients of various skin types, making it a very versatile platform in his practice.
The AVAVA MIRIA system is a “next generation” laser “where you’re able to use a focal point. Basically, you’re treating the skin from the inside out in a 3D manner and you’re able to focus intradermally up to 1 mm with high energy 1,064 nm or 1,550 nm,” he said. “It’s a unique conical geometry that spares the epidermis, combined with sapphire tip cooling and images the skin at the same time with the potential for personalized treatments of dyschromia and photoaging in all skin types. It’s truly remarkable where the technology is heading.”
Dr. Friedman disclosed that he has received consulting fees from Allergan, Galderma, Acclaro, Merz Aesthetics, Solta Medical, and Cytrellis. He has conducted contracted research for Sofwave and is a member of the speakers bureau for Solta Medical and Candela.
PHOENIX – Ever since PicoSure became the first picosecond laser cleared by the Food and Drug Administration for the treatment of unwanted tattoos and pigmented lesions in 2012, new uses for this technology continue to expand.
Now, These include PicoWay, PicoSure, Enlighten, PicoPlus, PiQo4, and Quanta Pico, among others.
“PicoWay technology has integrated nicely into my practice in Houston, the most ethnically diverse city in the country, with its ability to safely treat a number of various benign, congenital, and acquired epidermal and dermal pigmented lesions with ultrashort pulse duration and low thermal impact, which greatly reduces the risk of postinflammatory hyperpigmentation even in darker skin types,” Paul M. Friedman, MD, director of the Dermatology and Laser Surgery Center, Houston, said at the annual conference of the American Society for Laser Medicine and Surgery.
He emphasized the importance of therapeutic clinical endpoints, noting that with q-switched lasers, “you’re looking for immediate whitening, whereas with picosecond lasers, your endpoint is slight whitening or slight darkening depending on wavelength, indication, and skin type. The ability to fractionate picosecond pulses has also allowed us to utilize this technology for photoaging as well as acne scarring.”
The PicoWay system includes a 730-nm picosecond titanium sapphire handpiece, which is FDA cleared for treatment of benign pigmented lesions and blue and green tattoo removal. Dr. Friedman said that he has seen good clinical results using the handpiece for café-au-lait macules, particularly in skin of color.
In an abstract presented at the ASLMS meeting, he and his colleagues presented a retrospective review of 12 patients with café-au-lait macules with Fitzpatrick skin types III-VI who were treated with the PicoWay 730 nm handpiece between April 2021 and January 2023. Patients received a mean of 3.1 treatments at intervals that ranged from 5 to 40 weeks. Clinical photographs were graded by three board-certified dermatologists using a 5-point visual analogue scale.
Overall, patients were rated to have a mean improvement of 26%-50%. Two patients achieved 100% clearance after four to five treatment sessions. “Café-au-lait macules with smooth borders responded less well to laser treatment, confirming prior studies at our center,” he said. “We often educate parents that café-au-lait macules may recur over time, especially with repeated sun exposure.”
Treating melasma
Dr. Friedman’s go-to devices for melasma include the low-density, low-energy 1,927-nm fractional diode laser; the 1,064 nm picosecond Nd:YAG, the low-fluence 1,064 nm Q-switched Nd:YAG with a nanosecond pulse duration, and the 595-nm pulsed dye laser for lesions exhibiting underlying vascularity. He said that combining therapies that target pigment and vasculature may be ideal to prevent relapses. “Melasma is a multifactorial condition so by improving patient education and expectation alongside advances in laser treatment of melasma, we have ultimately improved our ability to treat this condition,” he said.
“We’re approaching it from all angles, with ultraviolet photography and spectrocolorimetry, behavioral modifications, topical skin-lightening agents, broad spectrum sunscreens with protection against visible light, and oral tranexamic acid in advanced cases. Then, we intervene with these energy-based modalities, and the bottom line is, less energy and density is more, with lengthened treatment intervals. In 2023, we’re better than we’ve ever been in terms of our ability to safely and effectively improve melasma.”
Novel lasers
Dr. Friedman also described the UltraClear, a novel ablative fractional 2,910-nm erbium-doped glass fiber laser that delivers a customized blend of ablation and coagulation based on the patient’s condition, skin type, and tolerability for down time. He provided an overview of the versatility of what he described as highly customizable technology for conditions such as photoaging and dyschromia in patients of various skin types, making it a very versatile platform in his practice.
The AVAVA MIRIA system is a “next generation” laser “where you’re able to use a focal point. Basically, you’re treating the skin from the inside out in a 3D manner and you’re able to focus intradermally up to 1 mm with high energy 1,064 nm or 1,550 nm,” he said. “It’s a unique conical geometry that spares the epidermis, combined with sapphire tip cooling and images the skin at the same time with the potential for personalized treatments of dyschromia and photoaging in all skin types. It’s truly remarkable where the technology is heading.”
Dr. Friedman disclosed that he has received consulting fees from Allergan, Galderma, Acclaro, Merz Aesthetics, Solta Medical, and Cytrellis. He has conducted contracted research for Sofwave and is a member of the speakers bureau for Solta Medical and Candela.
FROM ASLMS 2023
Five ways docs may qualify for discounts on medical malpractice premiums
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Getting a better deal might simply mean taking advantage of incentives and discounts your insurer may already offer. These include claims-free, new-to-practice, and working part-time discounts.
However, if you decide to shop around, keep in mind that discounts are just one factor that can affect your premium price – insurers look at your specialty, location, and claims history.
One of the most common ways physicians can earn discounts is by participating in risk management programs. With this type of program, physicians evaluate elements of their practice and documentation practices and identify areas that might leave them at risk for a lawsuit. While they save money, physician risk management programs also are designed to reduce malpractice claims, which ultimately minimizes the potential for bigger financial losses, insurance experts say.
“It’s a win-win situation when liability insurers and physicians work together to minimize risk, and it’s a win for patients,” said Gary Price, MD, president of The Physicians Foundation.
Doctors in private practice or employed by small hospitals that are not self-insured can qualify for these discounts, said David Zetter, president of Zetter HealthCare Management Consultants.
“I do a lot of work with medical malpractice companies trying to find clients policies. All the carriers are transparent about what physicians have to do to lower their premiums. Physicians can receive the discounts if they follow through and meet the insurer’s requirements,” said Mr. Zetter.
State insurance departments regulate medical malpractice insurance, including the premium credits insurers offer. Most states cap discounts at 25%, but some go as high as 70%, according to The Doctors Company, a national physician-owned medical malpractice insurer.
Insurers typically offer doctors several ways to earn discounts. The size of the discount also can depend on whether a doctor is new to a practice, remains claims free, or takes risk management courses.
In addition to the premium discount, some online risk management classes and webinars are eligible for CME credits.
“The credits can add up and they can be used for recertification or relicensure,” said Susan Boisvert, senior patient safety risk manager at The Doctors Company.
Here are five ways you may qualify for discounts with your insurer.
1. Make use of discounts available to new doctors
Doctors can earn hefty discounts on their premiums when they are no longer interns or residents and start practicing medicine. The Doctors Company usually gives a 50% discount on member premiums the first year they’re in practice and a 25% discount credit in their second year. The discounts end after that.
Other insurance carriers offer similar discounts to doctors starting to practice medicine. The deepest one is offered in the first year (at least 50%) and a smaller one (20%-25%) the second year, according to medical malpractice brokers.
“The new-to-practice discount is based solely on when the physician left their formal training to begin their practice for the first time; it is not based on claim-free history,” explained Mr. Zetter.
This is a very common discount used by different insurer carriers, said Dr. Price. “New physicians don’t have the same amount of risk of a lawsuit when they’re starting out. It’s unlikely they will have a claim and most liability actions have a 2-year time limit from the date of injury to be filed.”
2. Take advantage of being claims free
If you’ve been claims free for at least a few years, you may be eligible for a large discount.
“Doctors without claims are a better risk. Once a doctor has one claim, they’re likely to have a second, which the research shows,” said Mr. Zetter.
The most common credit The Doctors Company offers is 3 years of being claim free – this earns doctors up to 25%, he said. Mr. Zetter explained that the criteria and size of The Doctors Company credit may depend on the state where physicians practice.
“We allowed insurance carriers that we acquired to continue with their own claim-free discount program such as Florida’s First Professionals Insurance Company we acquired in 2011,” he said.
Doctors with other medical malpractice insurers may also be eligible for a credit up to 25%. In some instances, they may have to be claims free for 5 or 10 years, say insurance experts.
It pays to shop around before purchasing insurance.
3. If you work part time, make sure your premium reflects that
Physicians who see patients part time can receive up to a 75% discount on their medical liability insurance premiums.
The discounts are based on the hours the physician works per week. The fewer hours worked, the larger the discount. This type of discount does not vary by specialty.
According to The Doctors Company, working 10 hours or less per week may entitle doctors to a 75% discount; working 11-20 hours per week may entitle them to a 50% discount, and working 21-30 hours per week may entitle them to a 25% discount. If you are in this situation, it pays to ask your insurer if there is a discount available to you.
4. Look into your professional medical society insurance company
“I would look at your state medical association [or] state specialty society and talk to your colleagues to learn what premiums they’re paying and about any discounts they’re getting,” advised Mr. Zetter.
Some state medical societies have formed their own liability companies and offer lower premiums to their members because “they’re organized and managed by doctors, which makes their premiums more competitive,” Dr. Price said.
Other state medical societies endorse specific insurance carriers and offer their members a 5% discount for enrolling with them.
5. Enroll in a risk management program
Most insurers offer online educational activities designed to improve patient safety and reduce the risk of a lawsuit. Physicians may be eligible for both premium discounts and CME credits.
Medical Liability Mutual Insurance Company, owned by Berkshire Hathaway, operates in New York and offers physicians a premium discount of up to 5%, CME credit, and maintenance of certification credit for successfully completing its risk management program every other year.
ProAssurance members nationwide can earn 5% in premium discounts if they complete a 2-hour video series called “Back to Basics: Loss Prevention and Navigating Everyday Risks: Using Data to Drive Change.”
They can earn one credit for completing each webinar on topics such as “Medication Management: Minimizing Errors and Improving Safety” and “Opioid Prescribing: Keeping Patients Safe.”
MagMutual offers its insured physicians 1 CME credit for completing their specialty’s risk assessment and courses, which may be applied toward their premium discounts.
The Doctors Company offers its members a 5% premium discount if they complete 4 CME credits. One of its most popular courses is “How To Get Rid of a Difficult Patient.”
“Busy residents like the shorter case studies worth one-quarter credit that they can complete in 15 minutes,” said Ms. Boisvert.
“This is a good bargain from the physician’s standpoint and the fact that risk management education is offered online makes it a lot easier than going to a seminar in person,” said Dr. Price.
A version of this article first appeared on Medscape.com.
Two phase 3 trials show benefits of dupilumab for prurigo nodularis
The results, which were published online in Nature Medicine, were the basis for the FDA approval of dupilumab (Dupixent) for adults with PN in September 2022, the first treatment approved for treating PN in the United States.
“These positive studies support the involvement of type 2 cytokines in driving PN disease pathogenesis and the targeting of the [interleukin]-4/IL-13 axis as a novel therapeutic paradigm for patients with PN,” wrote the researchers, who were led by principal investigator Gil Yosipovitch, MD, professor of dermatology at the University of Miami, Fla. Dupilumab, an IL-4 receptor alpha antagonist, blocks the shared receptor component (IL-4R alpha) for IL-4 and IL-13.
For the two phase 3 trials, which were called LIBERTY-PN PRIME and PRIME2 and were sponsored by Sanofi and Regeneron Pharmaceuticals, researchers randomized adults with PN with 20 or more nodules and severe itch uncontrolled with topical therapies 1:1 to 300 mg dupilumab or placebo subcutaneously every 2 weeks for 24 weeks. The primary endpoint was pruritus improvement, which was measured by the proportion of patients with a 4-point or greater reduction in Worst Itch Numeric Rating Scale (WI-NRS) from baseline at week 24 (PRIME) or week 12 (PRIME2). Key secondary endpoints included a reduction in the number of nodules to 5 or fewer at week 24.
PRIME and PRIME2 enrolled 151 and 160 patients, respectively. In PRIME, 60% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 24, compared with 18.4% of patients in the placebo arm (P < .001). In PRIME2, 37.2% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 12, compared with 22% of patients in the placebo arm (P = .022).
The researchers also reported that, from an initial baseline of 20 to greater than 100 nodules, 32.0% of dupilumab-treated patients in PRIME and 25.6% in PRIME2 showed a reduction to 5 nodules or fewer, which corresponded to a response of “clear” or “almost clear” skin at week 12, compared with 11.8% and 12.2% of placebo-treated patients, respectively. This treatment effect on skin lesions continued to improve after week 12, with 48% of dupilumab-treated patients in PRIME and 44.9% in PRIME2 having five nodules or fewer at week 24, compared with 18.4% and 15.9% of placebo-treated patients, respectively. Safety was consistent with the known dupilumab safety profile.
“Validation is the first success of this paper,” said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study. “While both the safety and efficacy of dupilumab in these two phase 3 programs is the meat of the matter, nuanced highlights for me include the rigid nature of the exclusion criteria to ensure a study population that truly has PN as a stand-alone disease, rather than a secondary finding as we once believed to be the entire story. I think it’s important for us to recognize that it’s not one or the other, rather there is both ‘primary’ prurigo nodularis, and then there is secondary prurigo nodularis associated with something else [a wide range of underlying medical conditions], just like we divide primary and secondary hyperhidrosis.”
Dr. Yosipovitch reported having competing interests with several pharmaceutical companies, including Regeneron and Sanofi. Dr. Friedman disclosed that he is a consultant to and a speaker for Regeneron.
The results, which were published online in Nature Medicine, were the basis for the FDA approval of dupilumab (Dupixent) for adults with PN in September 2022, the first treatment approved for treating PN in the United States.
“These positive studies support the involvement of type 2 cytokines in driving PN disease pathogenesis and the targeting of the [interleukin]-4/IL-13 axis as a novel therapeutic paradigm for patients with PN,” wrote the researchers, who were led by principal investigator Gil Yosipovitch, MD, professor of dermatology at the University of Miami, Fla. Dupilumab, an IL-4 receptor alpha antagonist, blocks the shared receptor component (IL-4R alpha) for IL-4 and IL-13.
For the two phase 3 trials, which were called LIBERTY-PN PRIME and PRIME2 and were sponsored by Sanofi and Regeneron Pharmaceuticals, researchers randomized adults with PN with 20 or more nodules and severe itch uncontrolled with topical therapies 1:1 to 300 mg dupilumab or placebo subcutaneously every 2 weeks for 24 weeks. The primary endpoint was pruritus improvement, which was measured by the proportion of patients with a 4-point or greater reduction in Worst Itch Numeric Rating Scale (WI-NRS) from baseline at week 24 (PRIME) or week 12 (PRIME2). Key secondary endpoints included a reduction in the number of nodules to 5 or fewer at week 24.
PRIME and PRIME2 enrolled 151 and 160 patients, respectively. In PRIME, 60% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 24, compared with 18.4% of patients in the placebo arm (P < .001). In PRIME2, 37.2% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 12, compared with 22% of patients in the placebo arm (P = .022).
The researchers also reported that, from an initial baseline of 20 to greater than 100 nodules, 32.0% of dupilumab-treated patients in PRIME and 25.6% in PRIME2 showed a reduction to 5 nodules or fewer, which corresponded to a response of “clear” or “almost clear” skin at week 12, compared with 11.8% and 12.2% of placebo-treated patients, respectively. This treatment effect on skin lesions continued to improve after week 12, with 48% of dupilumab-treated patients in PRIME and 44.9% in PRIME2 having five nodules or fewer at week 24, compared with 18.4% and 15.9% of placebo-treated patients, respectively. Safety was consistent with the known dupilumab safety profile.
“Validation is the first success of this paper,” said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study. “While both the safety and efficacy of dupilumab in these two phase 3 programs is the meat of the matter, nuanced highlights for me include the rigid nature of the exclusion criteria to ensure a study population that truly has PN as a stand-alone disease, rather than a secondary finding as we once believed to be the entire story. I think it’s important for us to recognize that it’s not one or the other, rather there is both ‘primary’ prurigo nodularis, and then there is secondary prurigo nodularis associated with something else [a wide range of underlying medical conditions], just like we divide primary and secondary hyperhidrosis.”
Dr. Yosipovitch reported having competing interests with several pharmaceutical companies, including Regeneron and Sanofi. Dr. Friedman disclosed that he is a consultant to and a speaker for Regeneron.
The results, which were published online in Nature Medicine, were the basis for the FDA approval of dupilumab (Dupixent) for adults with PN in September 2022, the first treatment approved for treating PN in the United States.
“These positive studies support the involvement of type 2 cytokines in driving PN disease pathogenesis and the targeting of the [interleukin]-4/IL-13 axis as a novel therapeutic paradigm for patients with PN,” wrote the researchers, who were led by principal investigator Gil Yosipovitch, MD, professor of dermatology at the University of Miami, Fla. Dupilumab, an IL-4 receptor alpha antagonist, blocks the shared receptor component (IL-4R alpha) for IL-4 and IL-13.
For the two phase 3 trials, which were called LIBERTY-PN PRIME and PRIME2 and were sponsored by Sanofi and Regeneron Pharmaceuticals, researchers randomized adults with PN with 20 or more nodules and severe itch uncontrolled with topical therapies 1:1 to 300 mg dupilumab or placebo subcutaneously every 2 weeks for 24 weeks. The primary endpoint was pruritus improvement, which was measured by the proportion of patients with a 4-point or greater reduction in Worst Itch Numeric Rating Scale (WI-NRS) from baseline at week 24 (PRIME) or week 12 (PRIME2). Key secondary endpoints included a reduction in the number of nodules to 5 or fewer at week 24.
PRIME and PRIME2 enrolled 151 and 160 patients, respectively. In PRIME, 60% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 24, compared with 18.4% of patients in the placebo arm (P < .001). In PRIME2, 37.2% of patients in the dupilumab arm achieved a 4-point or greater reduction in the WI-NRS at week 12, compared with 22% of patients in the placebo arm (P = .022).
The researchers also reported that, from an initial baseline of 20 to greater than 100 nodules, 32.0% of dupilumab-treated patients in PRIME and 25.6% in PRIME2 showed a reduction to 5 nodules or fewer, which corresponded to a response of “clear” or “almost clear” skin at week 12, compared with 11.8% and 12.2% of placebo-treated patients, respectively. This treatment effect on skin lesions continued to improve after week 12, with 48% of dupilumab-treated patients in PRIME and 44.9% in PRIME2 having five nodules or fewer at week 24, compared with 18.4% and 15.9% of placebo-treated patients, respectively. Safety was consistent with the known dupilumab safety profile.
“Validation is the first success of this paper,” said Adam Friedman, MD, professor and chair of dermatology at George Washington University, Washington, who was asked to comment on the study. “While both the safety and efficacy of dupilumab in these two phase 3 programs is the meat of the matter, nuanced highlights for me include the rigid nature of the exclusion criteria to ensure a study population that truly has PN as a stand-alone disease, rather than a secondary finding as we once believed to be the entire story. I think it’s important for us to recognize that it’s not one or the other, rather there is both ‘primary’ prurigo nodularis, and then there is secondary prurigo nodularis associated with something else [a wide range of underlying medical conditions], just like we divide primary and secondary hyperhidrosis.”
Dr. Yosipovitch reported having competing interests with several pharmaceutical companies, including Regeneron and Sanofi. Dr. Friedman disclosed that he is a consultant to and a speaker for Regeneron.
FROM NATURE MEDICINE
Boys may carry the weight, or overweight, of adults’ infertility
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
Overweight boy, infertile man?
When it comes to causes of infertility, history and science have generally focused on women. A lot of the research overlooks men, but some previous studies have suggested that male infertility contributes to about half of the cases of couple infertility. The reason for much of that male infertility, however, has been a mystery. Until now.
A group of Italian investigators looked at the declining trend in sperm counts over the past 40 years and the increase of childhood obesity. Is there a correlation? The researchers think so. Childhood obesity can be linked to multiple causes, but the researchers zeroed in on the effect that obesity has on metabolic rates and, therefore, testicular growth.
Collecting data on testicular volume, body mass index (BMI), and insulin resistance from 268 boys aged 2-18 years, the researchers discovered that those with normal weight and normal insulin levels had testicular volumes 1.5 times higher than their overweight counterparts and 1.5-2 times higher than those with hyperinsulinemia, building a case for obesity being a factor for infertility later in life.
Since low testicular volume is associated with lower sperm count and production as an adult, putting two and two together makes a compelling argument for childhood obesity being a major male infertility culprit. It also creates even more urgency for the health care industry and community decision makers to focus on childhood obesity.
It sure would be nice to be able to take one of the many risk factors for future human survival off the table. Maybe by taking something, like cake, off the table.
Fecal transplantation moves to the kitchen
Fecal microbiota transplantation is an effective way to treat Clostridioides difficile infection, but, in the end, it’s still a transplantation procedure involving a nasogastric or colorectal tube or rather large oral capsules with a demanding (30-40 capsules over 2 days) dosage. Please, Science, tell us there’s a better way.
Science, in the form of investigators at the University of Geneva and Lausanne University Hospital in Switzerland, has spoken, and there may be a better way. Presenting fecal beads: All the bacterial goodness of donor stool without the tubal insertions or massive quantities of giant capsules.
We know you’re scoffing out there, but it’s true. All you need is a little alginate, which is a “biocompatible polysaccharide isolated from brown algae” of the Phaeophyceae family. The donor feces is microencapsulated by mixing it with the alginate, dropping that mixture into water containing calcium chloride, turning it into a gel, and then freeze-drying the gel into small (just 2 mm), solid beads.
Sounds plausible enough, but what do you do with them? “These brownish beads can be easily dispersed in a liquid or food that is pleasant to eat. They also have no taste,” senior author Eric Allémann, PhD, said in a statement released by the University of Geneva.
Pleasant to eat? No taste? So which is it? If you really want to know, watch fecal beads week on the new season of “The Great British Baking Show,” when Paul and Prue judge poop baked into crumpets, crepes, and crostatas. Yum.
We’re on the low-oxygen diet
Nine out of ten doctors agree: Oxygen is more important to your continued well-being than food. After all, a human can go weeks without food, but just minutes without oxygen. However, ten out of ten doctors agree that the United States has an obesity problem. They all also agree that previous research has shown soldiers who train at high altitudes lose more weight than those training at lower altitudes.
So, on the one hand, we have a country full of overweight people, and on the other, we have low oxygen levels causing weight loss. The solution, then, is obvious: Stop breathing.
More specifically (and somewhat less facetiously), researchers from Louisiana have launched the Low Oxygen and Weight Status trial and are currently recruiting individuals with BMIs of 30-40 to, uh, suffocate themselves. No, no, it’s okay, it’s just when they’re sleeping.
Fine, straight face. Participants in the LOWS trial will undergo an 8-week period when they will consume a controlled weight-loss diet and spend their nights in a hypoxic sealed tent, where they will sleep in an environment with an oxygen level equivalent to 8,500 feet above sea level (roughly equivalent to Aspen, Colo.). They will be compared with people on the same diet who sleep in a normal, sea-level oxygen environment.
The study’s goal is to determine whether or not spending time in a low-oxygen environment will suppress appetite, increase energy expenditure, and improve weight loss and insulin sensitivity. Excessive weight loss in high-altitude environments isn’t a good thing for soldiers – they kind of need their muscles and body weight to do the whole soldiering thing – but it could be great for people struggling to lose those last few pounds. And it also may prove LOTME’s previous thesis: Air is not good.
New-Onset Pemphigoid Gestationis Following COVID-19 Vaccination
To the Editor:
Pemphigoid gestationis (PG), or gestational pemphigoid, is a rare autoimmune bullous disease (AIBD) occurring in 1 in 50,000 pregnancies. It is characterized by abrupt development of intensely pruritic papules and urticarial plaques, followed by an eruption of blisters.1 We present a case of new-onset PG that erupted 10 days following SARs-CoV-2 messenger RNA (mRNA) vaccination with BNT162b2 (Pfizer-BioNTech).
A 36-year-old pregnant woman (gravida 1, para 0, aborta 0) at 37 weeks’ gestation presented to our AIBD clinic with a pruritic dermatitis of 6 weeks’ duration that developed 10 days after receiving the second dose of BNT162b2. Multiple intensely pruritic, red bumps presented first on the forearms and within days spread to the thighs, hands, and abdomen, followed by progression to the ankles, feet, and back 2 weeks later. An initial biopsy was consistent with subacute spongiotic dermatitis with rare eosinophils. She found minimal relief from diphenhydramine or topical steroids. She denied oral, nasal, ocular, or genital involvement or history of any other skin disease. The pregnancy had been otherwise uneventful.
Physical examination revealed annular edematous plaques on the trunk and buttocks; excoriated and erythematous papules on the neck, trunk, arms, and legs; and scattered vesicles along the fingers, arms, hands, abdomen, back, legs, and feet (Figure 1). The Bullous Pemphigoid Disease Area Index (BPDAI) total skin activity score was 25.3, corresponding to moderate disease activity (validated at 20–56).2 The BPDAI total pruritus component score was 20. A repeat biopsy for direct immunofluorescence showed faint linear deposits of IgG and bright linear deposits of C3 along the basement membrane zone. Indirect immunofluorescence showed linear deposits of IgG localized to the blister roof of salt-split skin at a dilution of 1:40. An enzyme-linked immunosorbent assay for anti-BP180 was 62 U/mL (negative, <9 U/mL; positive, ≥9 U/mL), and anti-BP230 autoantibodies were less than 9 U/mL (negative <9 U/mL; positive, ≥9 U/mL). Given these clinical and histopathologic findings, PG was diagnosed.
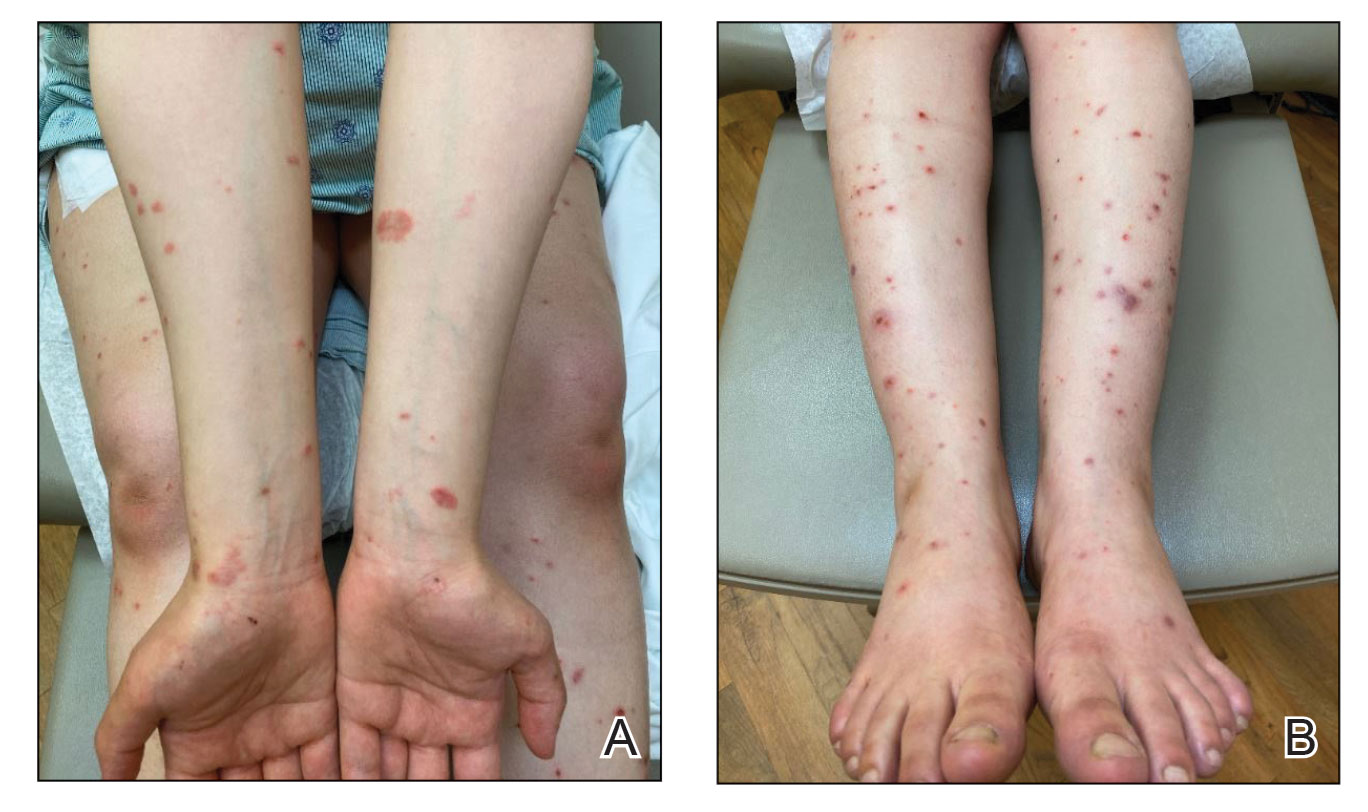
The patient was started on prednisone 20 mg and antihistamines while continuing topical steroids. Pruritus and blistering improved close to delivery. Fetal monitoring with regular biophysical profiles remained normal. The patient delivered a healthy neonate without skin lesions at 40 weeks’ gestation. The disease flared 2 days after delivery, and prednisone was increased to 40 mg and slowly tapered. Two months after delivery, the patient remained on prednisone 10 mg daily with ongoing but reduced blistering and pruritus (Figure 2). The BPDAI total skin activity and pruritus component scores remained elevated at 20.3 and 14, respectively, and anti-BP180 was 44 U/mL. After a discussion with the patient on safe systemic therapy while breastfeeding, intravenous immunoglobulin (IVIG) was initiated. The patient received 3 monthly infusions at 2 g/kg and was able to taper the prednisone to 5 mg every other day without new lesions. Four months after completion of IVIG therapy, she achieved complete remission off all therapy.
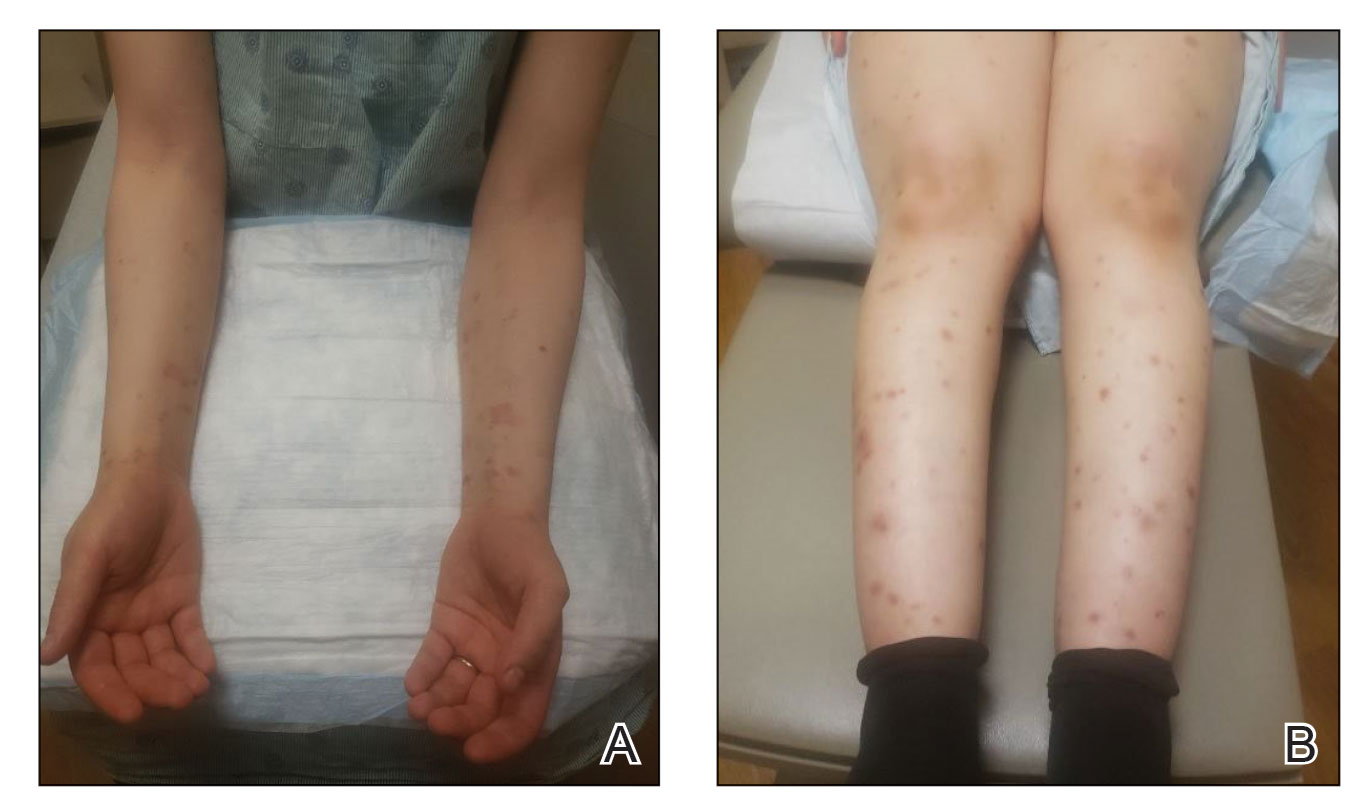
Management of PG begins with topical corticosteroids, but most patients require systemic steroid therapy.1 Remission commonly occurs close to delivery, and 75% of patients flare post partum, though the disease typically resolves 6 months following delivery.1,3,4 For persistent intrapartum cases requiring more than prednisone 20 mg daily, therapy can include dapsone, IVIG, azathioprine, rituximab, or plasmapheresis.4,5 Dapsone and IVIG are compatible with breastfeeding postpartum, but if dapsone is selected, the infant must be monitored for hemolytic anemia.5 Pemphigoid gestationis increases the risk for a premature or small-for-gestational-age neonate, necessitating regular fetal monitoring until delivery.1 Cutaneous lesions may affect the newborn, though this occurrence is rare and self-limiting.6 Pemphigoid gestationis may recur in subsequent pregnancies at a rate of 33% to 55%, with earlier and more severe presentations.4
Clinically and histologically, PG closely resembles bullous pemphigoid (BP), but the exact pathogenesis is not fully understood. Recently, another case of what was termed pseudo-PG has been described 3 days following administration of the second dose of the Pfizer-BioNTech COVID-19 vaccine.7 Since the introduction of COVID-19 mRNA vaccines, cases of postvaccination BP, BP-like eruptions, and pemphigus vulgaris have been described.8-11 Tomayko et al10 reported 12 cases of subepidermal eruptions, including BP, in which 7 patients developed blisters after the second dose of either the Pfizer-BioNTech or Moderna mRNA vaccine. Three patients who developed BP after the first dose of the vaccine and chose to receive the second dose tolerated it well, with a mild flare observed in 1 patient.10 Similarly, subsequent vaccine doses in reports of vaccine-associated AIBD resulted in increased disease activity in 21% of cases.12 COVID-19 vaccine–associated BP, similar to drug-induced BP, seemingly displays a milder course of disease compared to the classic form of BP.10,13 More follow-up is needed to better understand these reactions and inform appropriate discussions on the administration of booster doses. Currently, completion of the vaccination series against COVID-19 is advisable given the paucity of reports of postvaccination AIBD and the risk for COVID-19 infection, but careful discussions on a case-by-case basis are warranted related to the risk for disease exacerbation following subsequent vaccinations.
The clinical presentation and diagnostic evaluation of our patient’s rash were consistent with PG. The temporal relationship between vaccine administration and PG lesion onset suggests the mRNA vaccine triggered AIBD in our patient. Interestingly, AIBD associated with COVID-19 is not unique to only the vaccines and has been observed following infection with the virus itself.14 The high rate of vaccination against COVID-19 in contrast with the low number of reported cases of AIBD after vaccination supports the overall safety of COVID-19 vaccines but identifies a need for further understanding of the processes that lead to the development of autoimmune conditions in at-risk populations.
- Wiznia LE, Pomeranz MK. Skin changes and diseases in pregnancy. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Masmoudi W, Vaillant M, Vassileva S, et al. International validation of the Bullous Pemphigoid Disease Area Index severity score and calculation of cut-off values for defining mild, moderate and severe types of bullous pemphigoid. Br J Dermatol. 2021;184:1106-1112. doi:10.1111/bjd.19611
- Semkova K, Black M. Pemphigoid gestationis: current insights into pathogenesis and treatment. Eur J Obstet Gynecol Reprod Biol. 2009;145:138-144.
- Savervall C, Sand FL, Thomsen SF. Pemphigoid gestationis: current perspectives. Clin Cosmet Investig Dermatol. 2017;10:441-449.
- Braunstein I, Werth V. Treatment of dermatologic connective tissue disease and autoimmune blistering disorders in pregnancy. Dermatol Ther. 2013;26:354-363.
- Lipozencic J, Ljubojevic S, Bukvic-Mokos Z. Pemphigoid gestationis. Clin Dermatol. 2012;30:51-55.
- de Lorenzi C, Kaya G, Toutous Trellu L. Pseudo-pemphigoid gestationis eruption following SARS-CoV-2 vaccination with mRNA vaccine. Dermatopathology (Basel). 2022;9:203-206. doi:10.3390/dermatopathology9030025
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Solimani F, Mansour Y, Didona D, et al. Development of severe pemphigus vulgaris following SARS-CoV-2 vaccination with BNT162b2. J Eur Acad Dermatol Venereol. 2021;35:E649-E651.
- Tomayko MM, Damsky W, Fathy R, et al. Subepidermal blistering eruptions, including bullous pemphigoid, following COVID-19 vaccination. J Allergy Clin Immunol. 2021;148:750-751.
- Coto-Segura P, Fernandez-Prada M, Mir-Bonafe M, et al. Vesiculobullous skin reactions induced by COVID-19 mRNA vaccine: report of four cases and review of the literature. Clin Exp Dermatol. 2022;47:141-143.
- Kasperkiewicz M, Woodley DT. Association between vaccination and autoimmune bullous diseases: a systematic review. J Am Acad Dermatol. 2022;86:1160-1164.
- Stavropoulos PG, Soura E, Antoniou C. Drug-induced pemphigoid: a review of the literature. J Eur Acad Dermatol Venereol. 2014;28:1133-1140.
- Olson N, Eckhardt D, Delano A. New-onset bullous pemphigoid in a COVID-19 patient. Case Rep Dermatol Med. 2021;2021:5575111.
To the Editor:
Pemphigoid gestationis (PG), or gestational pemphigoid, is a rare autoimmune bullous disease (AIBD) occurring in 1 in 50,000 pregnancies. It is characterized by abrupt development of intensely pruritic papules and urticarial plaques, followed by an eruption of blisters.1 We present a case of new-onset PG that erupted 10 days following SARs-CoV-2 messenger RNA (mRNA) vaccination with BNT162b2 (Pfizer-BioNTech).
A 36-year-old pregnant woman (gravida 1, para 0, aborta 0) at 37 weeks’ gestation presented to our AIBD clinic with a pruritic dermatitis of 6 weeks’ duration that developed 10 days after receiving the second dose of BNT162b2. Multiple intensely pruritic, red bumps presented first on the forearms and within days spread to the thighs, hands, and abdomen, followed by progression to the ankles, feet, and back 2 weeks later. An initial biopsy was consistent with subacute spongiotic dermatitis with rare eosinophils. She found minimal relief from diphenhydramine or topical steroids. She denied oral, nasal, ocular, or genital involvement or history of any other skin disease. The pregnancy had been otherwise uneventful.
Physical examination revealed annular edematous plaques on the trunk and buttocks; excoriated and erythematous papules on the neck, trunk, arms, and legs; and scattered vesicles along the fingers, arms, hands, abdomen, back, legs, and feet (Figure 1). The Bullous Pemphigoid Disease Area Index (BPDAI) total skin activity score was 25.3, corresponding to moderate disease activity (validated at 20–56).2 The BPDAI total pruritus component score was 20. A repeat biopsy for direct immunofluorescence showed faint linear deposits of IgG and bright linear deposits of C3 along the basement membrane zone. Indirect immunofluorescence showed linear deposits of IgG localized to the blister roof of salt-split skin at a dilution of 1:40. An enzyme-linked immunosorbent assay for anti-BP180 was 62 U/mL (negative, <9 U/mL; positive, ≥9 U/mL), and anti-BP230 autoantibodies were less than 9 U/mL (negative <9 U/mL; positive, ≥9 U/mL). Given these clinical and histopathologic findings, PG was diagnosed.

The patient was started on prednisone 20 mg and antihistamines while continuing topical steroids. Pruritus and blistering improved close to delivery. Fetal monitoring with regular biophysical profiles remained normal. The patient delivered a healthy neonate without skin lesions at 40 weeks’ gestation. The disease flared 2 days after delivery, and prednisone was increased to 40 mg and slowly tapered. Two months after delivery, the patient remained on prednisone 10 mg daily with ongoing but reduced blistering and pruritus (Figure 2). The BPDAI total skin activity and pruritus component scores remained elevated at 20.3 and 14, respectively, and anti-BP180 was 44 U/mL. After a discussion with the patient on safe systemic therapy while breastfeeding, intravenous immunoglobulin (IVIG) was initiated. The patient received 3 monthly infusions at 2 g/kg and was able to taper the prednisone to 5 mg every other day without new lesions. Four months after completion of IVIG therapy, she achieved complete remission off all therapy.

Management of PG begins with topical corticosteroids, but most patients require systemic steroid therapy.1 Remission commonly occurs close to delivery, and 75% of patients flare post partum, though the disease typically resolves 6 months following delivery.1,3,4 For persistent intrapartum cases requiring more than prednisone 20 mg daily, therapy can include dapsone, IVIG, azathioprine, rituximab, or plasmapheresis.4,5 Dapsone and IVIG are compatible with breastfeeding postpartum, but if dapsone is selected, the infant must be monitored for hemolytic anemia.5 Pemphigoid gestationis increases the risk for a premature or small-for-gestational-age neonate, necessitating regular fetal monitoring until delivery.1 Cutaneous lesions may affect the newborn, though this occurrence is rare and self-limiting.6 Pemphigoid gestationis may recur in subsequent pregnancies at a rate of 33% to 55%, with earlier and more severe presentations.4
Clinically and histologically, PG closely resembles bullous pemphigoid (BP), but the exact pathogenesis is not fully understood. Recently, another case of what was termed pseudo-PG has been described 3 days following administration of the second dose of the Pfizer-BioNTech COVID-19 vaccine.7 Since the introduction of COVID-19 mRNA vaccines, cases of postvaccination BP, BP-like eruptions, and pemphigus vulgaris have been described.8-11 Tomayko et al10 reported 12 cases of subepidermal eruptions, including BP, in which 7 patients developed blisters after the second dose of either the Pfizer-BioNTech or Moderna mRNA vaccine. Three patients who developed BP after the first dose of the vaccine and chose to receive the second dose tolerated it well, with a mild flare observed in 1 patient.10 Similarly, subsequent vaccine doses in reports of vaccine-associated AIBD resulted in increased disease activity in 21% of cases.12 COVID-19 vaccine–associated BP, similar to drug-induced BP, seemingly displays a milder course of disease compared to the classic form of BP.10,13 More follow-up is needed to better understand these reactions and inform appropriate discussions on the administration of booster doses. Currently, completion of the vaccination series against COVID-19 is advisable given the paucity of reports of postvaccination AIBD and the risk for COVID-19 infection, but careful discussions on a case-by-case basis are warranted related to the risk for disease exacerbation following subsequent vaccinations.
The clinical presentation and diagnostic evaluation of our patient’s rash were consistent with PG. The temporal relationship between vaccine administration and PG lesion onset suggests the mRNA vaccine triggered AIBD in our patient. Interestingly, AIBD associated with COVID-19 is not unique to only the vaccines and has been observed following infection with the virus itself.14 The high rate of vaccination against COVID-19 in contrast with the low number of reported cases of AIBD after vaccination supports the overall safety of COVID-19 vaccines but identifies a need for further understanding of the processes that lead to the development of autoimmune conditions in at-risk populations.
To the Editor:
Pemphigoid gestationis (PG), or gestational pemphigoid, is a rare autoimmune bullous disease (AIBD) occurring in 1 in 50,000 pregnancies. It is characterized by abrupt development of intensely pruritic papules and urticarial plaques, followed by an eruption of blisters.1 We present a case of new-onset PG that erupted 10 days following SARs-CoV-2 messenger RNA (mRNA) vaccination with BNT162b2 (Pfizer-BioNTech).
A 36-year-old pregnant woman (gravida 1, para 0, aborta 0) at 37 weeks’ gestation presented to our AIBD clinic with a pruritic dermatitis of 6 weeks’ duration that developed 10 days after receiving the second dose of BNT162b2. Multiple intensely pruritic, red bumps presented first on the forearms and within days spread to the thighs, hands, and abdomen, followed by progression to the ankles, feet, and back 2 weeks later. An initial biopsy was consistent with subacute spongiotic dermatitis with rare eosinophils. She found minimal relief from diphenhydramine or topical steroids. She denied oral, nasal, ocular, or genital involvement or history of any other skin disease. The pregnancy had been otherwise uneventful.
Physical examination revealed annular edematous plaques on the trunk and buttocks; excoriated and erythematous papules on the neck, trunk, arms, and legs; and scattered vesicles along the fingers, arms, hands, abdomen, back, legs, and feet (Figure 1). The Bullous Pemphigoid Disease Area Index (BPDAI) total skin activity score was 25.3, corresponding to moderate disease activity (validated at 20–56).2 The BPDAI total pruritus component score was 20. A repeat biopsy for direct immunofluorescence showed faint linear deposits of IgG and bright linear deposits of C3 along the basement membrane zone. Indirect immunofluorescence showed linear deposits of IgG localized to the blister roof of salt-split skin at a dilution of 1:40. An enzyme-linked immunosorbent assay for anti-BP180 was 62 U/mL (negative, <9 U/mL; positive, ≥9 U/mL), and anti-BP230 autoantibodies were less than 9 U/mL (negative <9 U/mL; positive, ≥9 U/mL). Given these clinical and histopathologic findings, PG was diagnosed.

The patient was started on prednisone 20 mg and antihistamines while continuing topical steroids. Pruritus and blistering improved close to delivery. Fetal monitoring with regular biophysical profiles remained normal. The patient delivered a healthy neonate without skin lesions at 40 weeks’ gestation. The disease flared 2 days after delivery, and prednisone was increased to 40 mg and slowly tapered. Two months after delivery, the patient remained on prednisone 10 mg daily with ongoing but reduced blistering and pruritus (Figure 2). The BPDAI total skin activity and pruritus component scores remained elevated at 20.3 and 14, respectively, and anti-BP180 was 44 U/mL. After a discussion with the patient on safe systemic therapy while breastfeeding, intravenous immunoglobulin (IVIG) was initiated. The patient received 3 monthly infusions at 2 g/kg and was able to taper the prednisone to 5 mg every other day without new lesions. Four months after completion of IVIG therapy, she achieved complete remission off all therapy.

Management of PG begins with topical corticosteroids, but most patients require systemic steroid therapy.1 Remission commonly occurs close to delivery, and 75% of patients flare post partum, though the disease typically resolves 6 months following delivery.1,3,4 For persistent intrapartum cases requiring more than prednisone 20 mg daily, therapy can include dapsone, IVIG, azathioprine, rituximab, or plasmapheresis.4,5 Dapsone and IVIG are compatible with breastfeeding postpartum, but if dapsone is selected, the infant must be monitored for hemolytic anemia.5 Pemphigoid gestationis increases the risk for a premature or small-for-gestational-age neonate, necessitating regular fetal monitoring until delivery.1 Cutaneous lesions may affect the newborn, though this occurrence is rare and self-limiting.6 Pemphigoid gestationis may recur in subsequent pregnancies at a rate of 33% to 55%, with earlier and more severe presentations.4
Clinically and histologically, PG closely resembles bullous pemphigoid (BP), but the exact pathogenesis is not fully understood. Recently, another case of what was termed pseudo-PG has been described 3 days following administration of the second dose of the Pfizer-BioNTech COVID-19 vaccine.7 Since the introduction of COVID-19 mRNA vaccines, cases of postvaccination BP, BP-like eruptions, and pemphigus vulgaris have been described.8-11 Tomayko et al10 reported 12 cases of subepidermal eruptions, including BP, in which 7 patients developed blisters after the second dose of either the Pfizer-BioNTech or Moderna mRNA vaccine. Three patients who developed BP after the first dose of the vaccine and chose to receive the second dose tolerated it well, with a mild flare observed in 1 patient.10 Similarly, subsequent vaccine doses in reports of vaccine-associated AIBD resulted in increased disease activity in 21% of cases.12 COVID-19 vaccine–associated BP, similar to drug-induced BP, seemingly displays a milder course of disease compared to the classic form of BP.10,13 More follow-up is needed to better understand these reactions and inform appropriate discussions on the administration of booster doses. Currently, completion of the vaccination series against COVID-19 is advisable given the paucity of reports of postvaccination AIBD and the risk for COVID-19 infection, but careful discussions on a case-by-case basis are warranted related to the risk for disease exacerbation following subsequent vaccinations.
The clinical presentation and diagnostic evaluation of our patient’s rash were consistent with PG. The temporal relationship between vaccine administration and PG lesion onset suggests the mRNA vaccine triggered AIBD in our patient. Interestingly, AIBD associated with COVID-19 is not unique to only the vaccines and has been observed following infection with the virus itself.14 The high rate of vaccination against COVID-19 in contrast with the low number of reported cases of AIBD after vaccination supports the overall safety of COVID-19 vaccines but identifies a need for further understanding of the processes that lead to the development of autoimmune conditions in at-risk populations.
- Wiznia LE, Pomeranz MK. Skin changes and diseases in pregnancy. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Masmoudi W, Vaillant M, Vassileva S, et al. International validation of the Bullous Pemphigoid Disease Area Index severity score and calculation of cut-off values for defining mild, moderate and severe types of bullous pemphigoid. Br J Dermatol. 2021;184:1106-1112. doi:10.1111/bjd.19611
- Semkova K, Black M. Pemphigoid gestationis: current insights into pathogenesis and treatment. Eur J Obstet Gynecol Reprod Biol. 2009;145:138-144.
- Savervall C, Sand FL, Thomsen SF. Pemphigoid gestationis: current perspectives. Clin Cosmet Investig Dermatol. 2017;10:441-449.
- Braunstein I, Werth V. Treatment of dermatologic connective tissue disease and autoimmune blistering disorders in pregnancy. Dermatol Ther. 2013;26:354-363.
- Lipozencic J, Ljubojevic S, Bukvic-Mokos Z. Pemphigoid gestationis. Clin Dermatol. 2012;30:51-55.
- de Lorenzi C, Kaya G, Toutous Trellu L. Pseudo-pemphigoid gestationis eruption following SARS-CoV-2 vaccination with mRNA vaccine. Dermatopathology (Basel). 2022;9:203-206. doi:10.3390/dermatopathology9030025
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Solimani F, Mansour Y, Didona D, et al. Development of severe pemphigus vulgaris following SARS-CoV-2 vaccination with BNT162b2. J Eur Acad Dermatol Venereol. 2021;35:E649-E651.
- Tomayko MM, Damsky W, Fathy R, et al. Subepidermal blistering eruptions, including bullous pemphigoid, following COVID-19 vaccination. J Allergy Clin Immunol. 2021;148:750-751.
- Coto-Segura P, Fernandez-Prada M, Mir-Bonafe M, et al. Vesiculobullous skin reactions induced by COVID-19 mRNA vaccine: report of four cases and review of the literature. Clin Exp Dermatol. 2022;47:141-143.
- Kasperkiewicz M, Woodley DT. Association between vaccination and autoimmune bullous diseases: a systematic review. J Am Acad Dermatol. 2022;86:1160-1164.
- Stavropoulos PG, Soura E, Antoniou C. Drug-induced pemphigoid: a review of the literature. J Eur Acad Dermatol Venereol. 2014;28:1133-1140.
- Olson N, Eckhardt D, Delano A. New-onset bullous pemphigoid in a COVID-19 patient. Case Rep Dermatol Med. 2021;2021:5575111.
- Wiznia LE, Pomeranz MK. Skin changes and diseases in pregnancy. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick’s Dermatology. 9th ed. McGraw-Hill Education; 2019.
- Masmoudi W, Vaillant M, Vassileva S, et al. International validation of the Bullous Pemphigoid Disease Area Index severity score and calculation of cut-off values for defining mild, moderate and severe types of bullous pemphigoid. Br J Dermatol. 2021;184:1106-1112. doi:10.1111/bjd.19611
- Semkova K, Black M. Pemphigoid gestationis: current insights into pathogenesis and treatment. Eur J Obstet Gynecol Reprod Biol. 2009;145:138-144.
- Savervall C, Sand FL, Thomsen SF. Pemphigoid gestationis: current perspectives. Clin Cosmet Investig Dermatol. 2017;10:441-449.
- Braunstein I, Werth V. Treatment of dermatologic connective tissue disease and autoimmune blistering disorders in pregnancy. Dermatol Ther. 2013;26:354-363.
- Lipozencic J, Ljubojevic S, Bukvic-Mokos Z. Pemphigoid gestationis. Clin Dermatol. 2012;30:51-55.
- de Lorenzi C, Kaya G, Toutous Trellu L. Pseudo-pemphigoid gestationis eruption following SARS-CoV-2 vaccination with mRNA vaccine. Dermatopathology (Basel). 2022;9:203-206. doi:10.3390/dermatopathology9030025
- McMahon DE, Kovarik CL, Damsky W, et al. Clinical and pathologic correlation of cutaneous COVID-19 vaccine reactions including V-REPP: a registry-based study. J Am Acad Dermatol. 2022;86:113-121.
- Solimani F, Mansour Y, Didona D, et al. Development of severe pemphigus vulgaris following SARS-CoV-2 vaccination with BNT162b2. J Eur Acad Dermatol Venereol. 2021;35:E649-E651.
- Tomayko MM, Damsky W, Fathy R, et al. Subepidermal blistering eruptions, including bullous pemphigoid, following COVID-19 vaccination. J Allergy Clin Immunol. 2021;148:750-751.
- Coto-Segura P, Fernandez-Prada M, Mir-Bonafe M, et al. Vesiculobullous skin reactions induced by COVID-19 mRNA vaccine: report of four cases and review of the literature. Clin Exp Dermatol. 2022;47:141-143.
- Kasperkiewicz M, Woodley DT. Association between vaccination and autoimmune bullous diseases: a systematic review. J Am Acad Dermatol. 2022;86:1160-1164.
- Stavropoulos PG, Soura E, Antoniou C. Drug-induced pemphigoid: a review of the literature. J Eur Acad Dermatol Venereol. 2014;28:1133-1140.
- Olson N, Eckhardt D, Delano A. New-onset bullous pemphigoid in a COVID-19 patient. Case Rep Dermatol Med. 2021;2021:5575111.
Practice Points
- Dermatologists should be aware that COVID-19 messenger RNA vaccinations may present with various cutaneous complications.
- Pemphigoid gestationis should be considered in a pregnant or postpartum woman with an unexplained eruption of persistent, pruritic, urticarial lesions and blisters occurring postvaccination. Treatments include high-potency topical steroids and frequently systemic corticosteroids, along with steroid-sparing agents in severe cases.
Papular Reticulated Rash
The Diagnosis: Prurigo Pigmentosa
Histopathology of the punch biopsy revealed subcorneal collections of neutrophils flanked by a spongiotic epidermis with neutrophil and eosinophil exocytosis. Rare dyskeratotic keratinocytes were identified at the dermoepidermal junction, and grampositive bacterial organisms were seen in a follicular infundibulum with purulent inflammation. The dermis demonstrated a mildly dense superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, scattered neutrophils, and eosinophils (Figure).
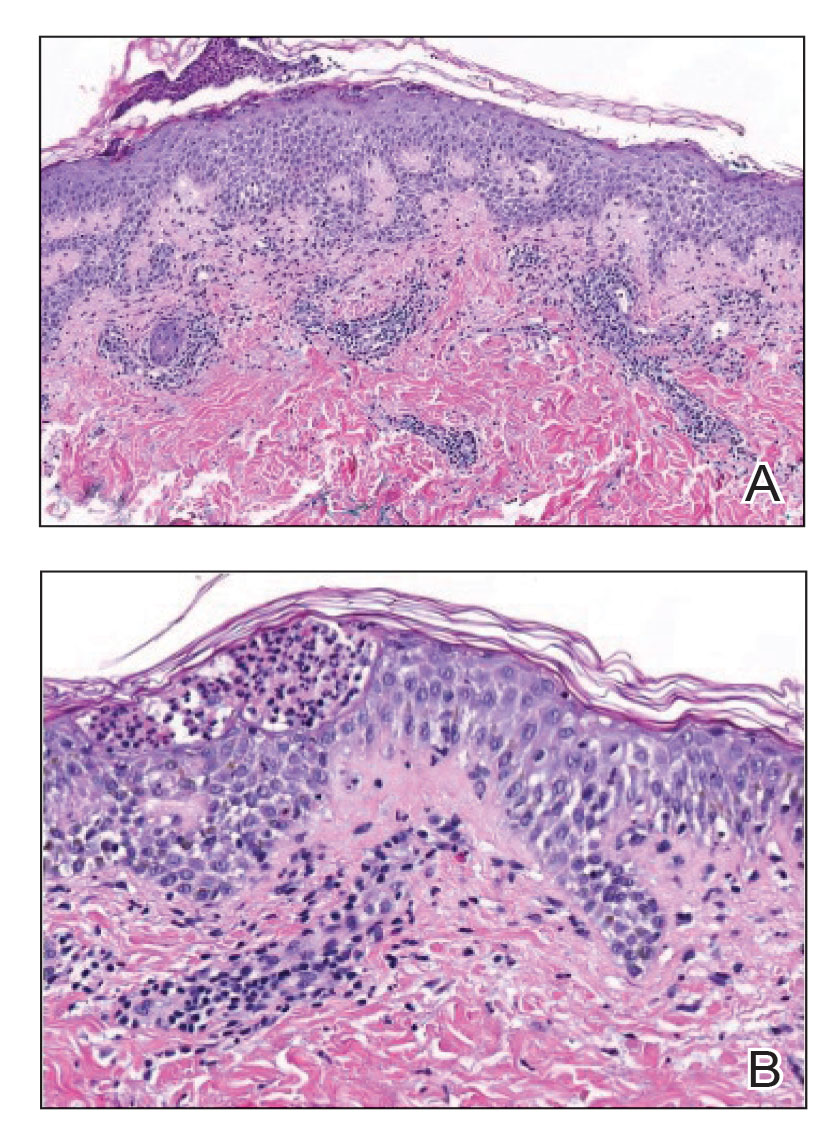
Given the combination of clinical and histologic findings, a diagnosis of prurigo pigmentosa (PP) was rendered and a urinalysis was ordered, which confirmed ketonuria. The patient was started on minocycline 100 mg twice daily and was advised to reintroduce carbohydrates into her diet. Resolution of the inflammatory component of the rash was achieved at 3-week follow-up, with residual reticulated postinflammatory hyperpigmentation.
Prurigo pigmentosa is a rare, albeit globally underrecognized, inflammatory dermatosis characterized by pruritic, symmetric, erythematous papules and plaques on the chest, back, neck, and rarely the arms and forehead that subsequently involute, leaving reticular postinflammatory hyperpigmentation.1 Prurigo pigmentosa is predominant in females (2.6:1 ratio). The mean age at presentation is 24.4 years, and it most commonly has been documented among populations in Asian countries, though it is unclear if a genetic predilection exists, as reports of PP are increasing globally with improved clinical awareness.1,2
The etiology of PP remains unknown; however, associations are well documented between PP and a ketogenic state secondary to uncontrolled diabetes, a low-carbohydrate diet, anorexia nervosa, or bariatric surgery.3 It is theorized that high serum ketones lead to perivascular ketone deposition, which induces neutrophil migration and chemotaxis,4 as substantiated by evidence of rash resolution with correction of the ketogenic state and improvement after administration of tetracyclines, a drug class known for neutrophil chemotaxis inhibition.5 Improvement of PP via these treatment mechanisms suggests that ketone bodies may play a role in the pathogenesis of PP.
Interestingly, Kafle et al6 reported that patients with PP commonly have bacterial colonies and associated inflammatory sequelae at the level of the hair follicles, which suggests that follicular involvement plays a role in the pathogenesis of PP. These findings are consistent with our patient’s histopathology consisting of gram-positive organisms and purulent inflammation at the infundibulum. The histopathologic features of PP are stage specific.1 Early stages are characterized by a superficial perivascular infiltrate of neutrophils that then spread to dermal papillae. Neutrophils then quickly sweep through the epidermis, causing spongiosis, ballooning, necrotic keratocytes, and consequent surface epithelium abscess formation. Over time, the dermal infiltrate assumes a lichenoid pattern as eosinophils and lymphocytes invade and predominate over neutrophils. Eventually, melanophages appear in the dermis as the epidermis undergoes hyperplasia, parakeratosis, and hyperpigmentation.1 The histologic differential diagnosis for PP is broad and varies based on the stage-specific progression of clinical and histopathologic findings.
Similar to PP, subacute cutaneous lupus erythematosus has a female predominance and resolves with subsequent dyspigmentation; however, it initially is characterized by annular plaques with central clearing or papulosquamous lesions restricted to sun-exposed skin. Photosensitivity is a prominent feature, and roughly 50% of patients meet diagnostic criteria for systemic lupus erythematosus.7 Histopathology shows interface changes with increased dermal mucin and a perivascular lymphoplasmacytic inflammatory infiltrate.
Papular pityriasis rosea can present as a pruritic papular rash on the back and chest; however, it most commonly is associated with a herald patch and typically follows a flulike prodrome.8 Biopsy reveals mounds of parakeratosis with mild spongiosis, perivascular inflammation, and extravasated erythrocytes.
Galli-Galli disease can present as a pruritic rash with follicular papules under the breasts and other flexural areas but histopathologically shows elongated rete ridges with dermal melanosis and acantholysis.9
Hailey-Hailey disease commonly presents in the third decade of life and can manifest as painful, pruritic, vesicular lesions on erythematous skin distributed on the back, neck, and inframammary region, as seen in our case; however, it is histopathologically associated with widespread epidermal acantholysis unlike the findings seen in our patient.10
First-line treatment of PP includes antibiotics such as minocycline, doxycycline, and dapsone due to their anti-inflammatory properties and ability to inhibit neutrophil chemotaxis. In patients with nutritional deficiencies or ketosis, reintroduction of carbohydrates alone has been effective.5,11
Prurigo pigmentosa is an underrecognized inflammatory dermatosis with a complex stage-dependent clinicopathologic presentation. Clinicians should be aware of the etiologic and histopathologic patterns of this unique dermatosis. Rash presentation in the context of a low-carbohydrate diet should prompt biopsy as well as treatment with antibiotics and dietary reintroduction of carbohydrates.
- Böer A, Misago N, Wolter M, et al. Prurigo pigmentosa: a distinctive inflammatory disease of the skin. Am J Dermatopathol. 2003;25:117-129. doi:10.1097/00000372-200304000-00005
- de Sousa Vargas TJ, Abreu Raposo CM, Lima RB, et al. Prurigo pigmentosa: report of 3 cases from Brazil and literature review. Am J Dermatopathol. 2017;39:267-274. doi:10.1097/DAD.0000000000000643
- Mufti A, Mirali S, Abduelmula A, et al. Clinical manifestations and treatment outcomes in prurigo pigmentosa (Nagashima disease): a systematic review of the literature. JAAD Int. 2021;3:79. doi:10.1016/J .JDIN.2021.03.003
- Beutler BD, Cohen PR, Lee RA. Prurigo pigmentosa: literature review. Am J Clin Dermatol. 2015;16:533-543. doi:10.1007/S40257-015-0154-4
- Chiam LYT, Goh BK, Lim KS, et al. Prurigo pigmentosa: a report of two cases that responded to minocycline. Clin Exp Dermatol. 2009;34. doi:10.1111/J.1365-2230.2009.03253.X
- Kafle SU, Swe SM, Hsiao PF, et al. Folliculitis in prurigo pigmentosa: a proposed pathogenesis based on clinical and pathological observation. J Cutan Pathol. 2017;44:20-27. doi:10.1111/CUP.12829
- Sontheimer RD. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263. doi:10.1016/J .AUTREV.2004.10.00
- Leung AKC, Lam JM, Leong KF, et al. Pityriasis rosea: an updated review. Curr Pediatr Rev. 2021;17:201-211. doi:10.2174/15733963166662 00923161330
- Sprecher E, Indelman M, Khamaysi Z, et al. Galli-Galli disease is an acantholytic variant of Dowling-Degos disease. Br J Dermatol. 2007;156:572-574. doi:10.1111/J.1365-2133.2006.07703.X
- Burge SM. Hailey-Hailey disease: the clinical features, response to treatment and prognosis. Br J Dermatol. 1992;126:275-282. doi:10.1111/J.1365-2133.1992.TB00658
- Lu L-Y, Chen C-B. Keto rash: ketoacidosis-induced prurigo pigmentosa. Mayo Clin Proc. 2022;97:20-21. doi:10.1016/j.mayocp.2021.11.019
The Diagnosis: Prurigo Pigmentosa
Histopathology of the punch biopsy revealed subcorneal collections of neutrophils flanked by a spongiotic epidermis with neutrophil and eosinophil exocytosis. Rare dyskeratotic keratinocytes were identified at the dermoepidermal junction, and grampositive bacterial organisms were seen in a follicular infundibulum with purulent inflammation. The dermis demonstrated a mildly dense superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, scattered neutrophils, and eosinophils (Figure).

Given the combination of clinical and histologic findings, a diagnosis of prurigo pigmentosa (PP) was rendered and a urinalysis was ordered, which confirmed ketonuria. The patient was started on minocycline 100 mg twice daily and was advised to reintroduce carbohydrates into her diet. Resolution of the inflammatory component of the rash was achieved at 3-week follow-up, with residual reticulated postinflammatory hyperpigmentation.
Prurigo pigmentosa is a rare, albeit globally underrecognized, inflammatory dermatosis characterized by pruritic, symmetric, erythematous papules and plaques on the chest, back, neck, and rarely the arms and forehead that subsequently involute, leaving reticular postinflammatory hyperpigmentation.1 Prurigo pigmentosa is predominant in females (2.6:1 ratio). The mean age at presentation is 24.4 years, and it most commonly has been documented among populations in Asian countries, though it is unclear if a genetic predilection exists, as reports of PP are increasing globally with improved clinical awareness.1,2
The etiology of PP remains unknown; however, associations are well documented between PP and a ketogenic state secondary to uncontrolled diabetes, a low-carbohydrate diet, anorexia nervosa, or bariatric surgery.3 It is theorized that high serum ketones lead to perivascular ketone deposition, which induces neutrophil migration and chemotaxis,4 as substantiated by evidence of rash resolution with correction of the ketogenic state and improvement after administration of tetracyclines, a drug class known for neutrophil chemotaxis inhibition.5 Improvement of PP via these treatment mechanisms suggests that ketone bodies may play a role in the pathogenesis of PP.
Interestingly, Kafle et al6 reported that patients with PP commonly have bacterial colonies and associated inflammatory sequelae at the level of the hair follicles, which suggests that follicular involvement plays a role in the pathogenesis of PP. These findings are consistent with our patient’s histopathology consisting of gram-positive organisms and purulent inflammation at the infundibulum. The histopathologic features of PP are stage specific.1 Early stages are characterized by a superficial perivascular infiltrate of neutrophils that then spread to dermal papillae. Neutrophils then quickly sweep through the epidermis, causing spongiosis, ballooning, necrotic keratocytes, and consequent surface epithelium abscess formation. Over time, the dermal infiltrate assumes a lichenoid pattern as eosinophils and lymphocytes invade and predominate over neutrophils. Eventually, melanophages appear in the dermis as the epidermis undergoes hyperplasia, parakeratosis, and hyperpigmentation.1 The histologic differential diagnosis for PP is broad and varies based on the stage-specific progression of clinical and histopathologic findings.
Similar to PP, subacute cutaneous lupus erythematosus has a female predominance and resolves with subsequent dyspigmentation; however, it initially is characterized by annular plaques with central clearing or papulosquamous lesions restricted to sun-exposed skin. Photosensitivity is a prominent feature, and roughly 50% of patients meet diagnostic criteria for systemic lupus erythematosus.7 Histopathology shows interface changes with increased dermal mucin and a perivascular lymphoplasmacytic inflammatory infiltrate.
Papular pityriasis rosea can present as a pruritic papular rash on the back and chest; however, it most commonly is associated with a herald patch and typically follows a flulike prodrome.8 Biopsy reveals mounds of parakeratosis with mild spongiosis, perivascular inflammation, and extravasated erythrocytes.
Galli-Galli disease can present as a pruritic rash with follicular papules under the breasts and other flexural areas but histopathologically shows elongated rete ridges with dermal melanosis and acantholysis.9
Hailey-Hailey disease commonly presents in the third decade of life and can manifest as painful, pruritic, vesicular lesions on erythematous skin distributed on the back, neck, and inframammary region, as seen in our case; however, it is histopathologically associated with widespread epidermal acantholysis unlike the findings seen in our patient.10
First-line treatment of PP includes antibiotics such as minocycline, doxycycline, and dapsone due to their anti-inflammatory properties and ability to inhibit neutrophil chemotaxis. In patients with nutritional deficiencies or ketosis, reintroduction of carbohydrates alone has been effective.5,11
Prurigo pigmentosa is an underrecognized inflammatory dermatosis with a complex stage-dependent clinicopathologic presentation. Clinicians should be aware of the etiologic and histopathologic patterns of this unique dermatosis. Rash presentation in the context of a low-carbohydrate diet should prompt biopsy as well as treatment with antibiotics and dietary reintroduction of carbohydrates.
The Diagnosis: Prurigo Pigmentosa
Histopathology of the punch biopsy revealed subcorneal collections of neutrophils flanked by a spongiotic epidermis with neutrophil and eosinophil exocytosis. Rare dyskeratotic keratinocytes were identified at the dermoepidermal junction, and grampositive bacterial organisms were seen in a follicular infundibulum with purulent inflammation. The dermis demonstrated a mildly dense superficial perivascular and interstitial infiltrate composed of lymphocytes, histiocytes, scattered neutrophils, and eosinophils (Figure).

Given the combination of clinical and histologic findings, a diagnosis of prurigo pigmentosa (PP) was rendered and a urinalysis was ordered, which confirmed ketonuria. The patient was started on minocycline 100 mg twice daily and was advised to reintroduce carbohydrates into her diet. Resolution of the inflammatory component of the rash was achieved at 3-week follow-up, with residual reticulated postinflammatory hyperpigmentation.
Prurigo pigmentosa is a rare, albeit globally underrecognized, inflammatory dermatosis characterized by pruritic, symmetric, erythematous papules and plaques on the chest, back, neck, and rarely the arms and forehead that subsequently involute, leaving reticular postinflammatory hyperpigmentation.1 Prurigo pigmentosa is predominant in females (2.6:1 ratio). The mean age at presentation is 24.4 years, and it most commonly has been documented among populations in Asian countries, though it is unclear if a genetic predilection exists, as reports of PP are increasing globally with improved clinical awareness.1,2
The etiology of PP remains unknown; however, associations are well documented between PP and a ketogenic state secondary to uncontrolled diabetes, a low-carbohydrate diet, anorexia nervosa, or bariatric surgery.3 It is theorized that high serum ketones lead to perivascular ketone deposition, which induces neutrophil migration and chemotaxis,4 as substantiated by evidence of rash resolution with correction of the ketogenic state and improvement after administration of tetracyclines, a drug class known for neutrophil chemotaxis inhibition.5 Improvement of PP via these treatment mechanisms suggests that ketone bodies may play a role in the pathogenesis of PP.
Interestingly, Kafle et al6 reported that patients with PP commonly have bacterial colonies and associated inflammatory sequelae at the level of the hair follicles, which suggests that follicular involvement plays a role in the pathogenesis of PP. These findings are consistent with our patient’s histopathology consisting of gram-positive organisms and purulent inflammation at the infundibulum. The histopathologic features of PP are stage specific.1 Early stages are characterized by a superficial perivascular infiltrate of neutrophils that then spread to dermal papillae. Neutrophils then quickly sweep through the epidermis, causing spongiosis, ballooning, necrotic keratocytes, and consequent surface epithelium abscess formation. Over time, the dermal infiltrate assumes a lichenoid pattern as eosinophils and lymphocytes invade and predominate over neutrophils. Eventually, melanophages appear in the dermis as the epidermis undergoes hyperplasia, parakeratosis, and hyperpigmentation.1 The histologic differential diagnosis for PP is broad and varies based on the stage-specific progression of clinical and histopathologic findings.
Similar to PP, subacute cutaneous lupus erythematosus has a female predominance and resolves with subsequent dyspigmentation; however, it initially is characterized by annular plaques with central clearing or papulosquamous lesions restricted to sun-exposed skin. Photosensitivity is a prominent feature, and roughly 50% of patients meet diagnostic criteria for systemic lupus erythematosus.7 Histopathology shows interface changes with increased dermal mucin and a perivascular lymphoplasmacytic inflammatory infiltrate.
Papular pityriasis rosea can present as a pruritic papular rash on the back and chest; however, it most commonly is associated with a herald patch and typically follows a flulike prodrome.8 Biopsy reveals mounds of parakeratosis with mild spongiosis, perivascular inflammation, and extravasated erythrocytes.
Galli-Galli disease can present as a pruritic rash with follicular papules under the breasts and other flexural areas but histopathologically shows elongated rete ridges with dermal melanosis and acantholysis.9
Hailey-Hailey disease commonly presents in the third decade of life and can manifest as painful, pruritic, vesicular lesions on erythematous skin distributed on the back, neck, and inframammary region, as seen in our case; however, it is histopathologically associated with widespread epidermal acantholysis unlike the findings seen in our patient.10
First-line treatment of PP includes antibiotics such as minocycline, doxycycline, and dapsone due to their anti-inflammatory properties and ability to inhibit neutrophil chemotaxis. In patients with nutritional deficiencies or ketosis, reintroduction of carbohydrates alone has been effective.5,11
Prurigo pigmentosa is an underrecognized inflammatory dermatosis with a complex stage-dependent clinicopathologic presentation. Clinicians should be aware of the etiologic and histopathologic patterns of this unique dermatosis. Rash presentation in the context of a low-carbohydrate diet should prompt biopsy as well as treatment with antibiotics and dietary reintroduction of carbohydrates.
- Böer A, Misago N, Wolter M, et al. Prurigo pigmentosa: a distinctive inflammatory disease of the skin. Am J Dermatopathol. 2003;25:117-129. doi:10.1097/00000372-200304000-00005
- de Sousa Vargas TJ, Abreu Raposo CM, Lima RB, et al. Prurigo pigmentosa: report of 3 cases from Brazil and literature review. Am J Dermatopathol. 2017;39:267-274. doi:10.1097/DAD.0000000000000643
- Mufti A, Mirali S, Abduelmula A, et al. Clinical manifestations and treatment outcomes in prurigo pigmentosa (Nagashima disease): a systematic review of the literature. JAAD Int. 2021;3:79. doi:10.1016/J .JDIN.2021.03.003
- Beutler BD, Cohen PR, Lee RA. Prurigo pigmentosa: literature review. Am J Clin Dermatol. 2015;16:533-543. doi:10.1007/S40257-015-0154-4
- Chiam LYT, Goh BK, Lim KS, et al. Prurigo pigmentosa: a report of two cases that responded to minocycline. Clin Exp Dermatol. 2009;34. doi:10.1111/J.1365-2230.2009.03253.X
- Kafle SU, Swe SM, Hsiao PF, et al. Folliculitis in prurigo pigmentosa: a proposed pathogenesis based on clinical and pathological observation. J Cutan Pathol. 2017;44:20-27. doi:10.1111/CUP.12829
- Sontheimer RD. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263. doi:10.1016/J .AUTREV.2004.10.00
- Leung AKC, Lam JM, Leong KF, et al. Pityriasis rosea: an updated review. Curr Pediatr Rev. 2021;17:201-211. doi:10.2174/15733963166662 00923161330
- Sprecher E, Indelman M, Khamaysi Z, et al. Galli-Galli disease is an acantholytic variant of Dowling-Degos disease. Br J Dermatol. 2007;156:572-574. doi:10.1111/J.1365-2133.2006.07703.X
- Burge SM. Hailey-Hailey disease: the clinical features, response to treatment and prognosis. Br J Dermatol. 1992;126:275-282. doi:10.1111/J.1365-2133.1992.TB00658
- Lu L-Y, Chen C-B. Keto rash: ketoacidosis-induced prurigo pigmentosa. Mayo Clin Proc. 2022;97:20-21. doi:10.1016/j.mayocp.2021.11.019
- Böer A, Misago N, Wolter M, et al. Prurigo pigmentosa: a distinctive inflammatory disease of the skin. Am J Dermatopathol. 2003;25:117-129. doi:10.1097/00000372-200304000-00005
- de Sousa Vargas TJ, Abreu Raposo CM, Lima RB, et al. Prurigo pigmentosa: report of 3 cases from Brazil and literature review. Am J Dermatopathol. 2017;39:267-274. doi:10.1097/DAD.0000000000000643
- Mufti A, Mirali S, Abduelmula A, et al. Clinical manifestations and treatment outcomes in prurigo pigmentosa (Nagashima disease): a systematic review of the literature. JAAD Int. 2021;3:79. doi:10.1016/J .JDIN.2021.03.003
- Beutler BD, Cohen PR, Lee RA. Prurigo pigmentosa: literature review. Am J Clin Dermatol. 2015;16:533-543. doi:10.1007/S40257-015-0154-4
- Chiam LYT, Goh BK, Lim KS, et al. Prurigo pigmentosa: a report of two cases that responded to minocycline. Clin Exp Dermatol. 2009;34. doi:10.1111/J.1365-2230.2009.03253.X
- Kafle SU, Swe SM, Hsiao PF, et al. Folliculitis in prurigo pigmentosa: a proposed pathogenesis based on clinical and pathological observation. J Cutan Pathol. 2017;44:20-27. doi:10.1111/CUP.12829
- Sontheimer RD. Subacute cutaneous lupus erythematosus: 25-year evolution of a prototypic subset (subphenotype) of lupus erythematosus defined by characteristic cutaneous, pathological, immunological, and genetic findings. Autoimmun Rev. 2005;4:253-263. doi:10.1016/J .AUTREV.2004.10.00
- Leung AKC, Lam JM, Leong KF, et al. Pityriasis rosea: an updated review. Curr Pediatr Rev. 2021;17:201-211. doi:10.2174/15733963166662 00923161330
- Sprecher E, Indelman M, Khamaysi Z, et al. Galli-Galli disease is an acantholytic variant of Dowling-Degos disease. Br J Dermatol. 2007;156:572-574. doi:10.1111/J.1365-2133.2006.07703.X
- Burge SM. Hailey-Hailey disease: the clinical features, response to treatment and prognosis. Br J Dermatol. 1992;126:275-282. doi:10.1111/J.1365-2133.1992.TB00658
- Lu L-Y, Chen C-B. Keto rash: ketoacidosis-induced prurigo pigmentosa. Mayo Clin Proc. 2022;97:20-21. doi:10.1016/j.mayocp.2021.11.019
An otherwise healthy 22-year-old woman presented with a painful eruption with burning and pruritus that had been slowly worsening as it spread over the last 4 weeks. The rash first appeared on the lower chest and inframammary folds (top) and spread to the upper chest, neck, back (bottom), arms, and lower face. Physical examination revealed multiple illdefined, erythematous papules, patches, and plaques on the chest, back, neck, and upper abdomen. Individual lesions coalesced into plaques that displayed a reticular configuration. There were no lesions in the axillae. The patient had been following a low-carbohydrate diet for 4 months. A punch biopsy was performed.
Mohs surgery improves survival in early-stage Merkel cell carcinoma
SEATTLE – The use of
Compared with conventional wide local excision, survival was significantly improved among patients treated with Mohs, and a subgroup analysis showed that the survival benefit remained for patients with risk factors.
“At 10 years, overall survival was about 21% higher for those treated with Mohs surgery versus those treated with conventional surgery,” said lead author Shayan Cheraghlou, MD, a dermatology resident at the New York University School of Medicine. “On multivariable analysis, which controlled for tumor and patient factors, Mohs was associated with an over 40% reduction in the hazard for death.”
The findings were presented at the annual meeting of the American College of Mohs Surgery.
MCC is a rare, aggressive, neuroendocrine cutaneous malignancy that carries a high mortality rate. The estimated 5-year survival for patients with localized disease is about 50%, Dr. Cheraghlou noted. “That extrapolates to about 55% for T1 tumors and down to about 30% for T4 tumors.”
Although it’s considered to be a rare cancer, the incidence of MCC has been rapidly rising, and in fact it doubled during the period from the 1990s to the 2010s.
Most commonly treated with wide local excision with or without adjuvant radiation therapy, Mohs as monotherapy may offer an alternative treatment option for patients with MCC. It is generally accepted that the optimal treatment for tumors without regional lymph node involvement is surgical, but the data regarding the optimal surgical approach are mixed. Current National Comprehensive Cancer Network guidelines state that either Mohs surgery or wide local excision can be used.
“However, these guidelines do not indicate a preference for one modality over the other,” said Dr. Cheraghlou, “and present them as interchangeable treatment options.”
A growing body of literature supports Mohs surgery for many types of rare tumors, including MCC. For example, as previously reported at the 2021 ACMS meeting, one study found that Mohs surgery compared favorably with the standard treatment approach when it came to recurrence rates for patients with MCC. The 5-year disease-specific survival rate was 91.2% for patients with stage I disease and 68.6% for patients with stage IIa. These rates were comparable with rates for historical control patients who were treated with wide local excision, with or without radiation (81%-87% for stage I disease, and 63%-67% for stage II).
Study details
In the current study, Dr. Cheraghlou and colleagues sought to evaluate the association of the type of surgical approach with patient survival after excision of early-stage MCC. They conducted a retrospective cohort study using the National Cancer Database to identify cases of MCC with T1/T2 tumors. A total of 2,313 patients who were diagnosed from 2004 to 2018 with pathologically confirmed negative lymph node involvement and who were treated with Mohs surgery or wide lesion excision were included in the analysis.
“About 90% were T1 tumors, about 40% were located on the head and neck, and the vast majority – about 60% – were treated with wide local excision,” he explained. “Only about 5% received Mohs surgery for treatment of the primary tumor.”
But when the researchers assessed survival outcomes, they found that treatment with Mohs surgery was associated with significantly improved overall survival.
The unadjusted 3-, 5-, and 10-year survival rates for patients treated with Mohs was 87.4% (SE: 3.4%), 84.5% (SE: 3.9%), and 81.8% (SE: 4.6%), respectively, while for wide lesion excision, the rates were 86.1% (SE: 0.9%), 76.9% (SE: 1.2%), and 60.9% (SE: 2.0%), respectively.
For patients who underwent treatment with narrow margin excision, survival rates were similar as for those treated with wide lesion excision, with 3-, 5-, and 10-year survival rates of 84.8% (SE: 1.4%), 78.3% (SE: 1.7%), and 60.8% (SE: 3.6%), respectively.
On multivariable survival analysis, Mohs surgery was associated with significantly improved survival, compared with wide lesion excision (hazard ratio, 0.594; P = .038). This was also true after multivariable analysis for patients who had one or more NCCN risk factors, for whom improved survival was also seen with Mohs (HR, 0.530; P = .026).
The results did not differ after a sensitivity analysis that included T3 and T4 tumors.
Given that the use of Mohs was so infrequent, compared with standard surgery, the researchers investigated the factors that were associated with the use of Mohs. High-volume MCC centers were significantly more likely to utilize Mohs than wide lesion excision (odds ratio, 1.993; P < .001), compared with other facilities.
“This study has important implications going forward,” Dr. Cheraghlou concluded. “We think it’s important how few patients were treated with Mohs for Merkel cell, and it was slightly more likely to happen in a high-volume center.”
The reasoning for that may be that high-volume centers are more likely to have a surgeon trained to perform Mohs surgery for MCC. “Or perhaps they are more attuned to the benefits of this procedure,” he said. “We can’t tell that from our data, but its notable that it’s such a small proportion of patients – especially when we consider that it is associated with improved survival for the patients who receive it.”
He added that efforts to increase the utilization of Mohs may yield improved local control and overall survival for these patients. “And perhaps with more data, future versions of guidelines may indicate a preference for Mohs over conventional incisions.”
No changes to current practice
Asked to comment on the study, Anthony J. Olszanski, RPh, MD, associate professor, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that while the results are intriguing, they must be interpreted with caution.
“This study was retrospective in nature, and unrecognized biases can influence results,” he said. “Additionally, given the relative rarity of Merkel cell carcinoma, the sample size is expectantly small.”
But importantly, Dr. Olszanski emphasized, Mohs may more often have been recommended for patients with lesions that appear less aggressive. “Many patients undergoing wide lesion excision may have been referred by Mohs surgeons secondary to features or characteristics of lesions which were worrisome,” he explained. “The results of this study do not opine on why Mohs would impact overall survival over wide lesion excision, a point worthy of consideration. Presently, both modalities can be considered for patients with T1/T2 MCC. The results of this study should not change current practice and would lend themselves to a more robust study.”
No external funding of the study was reported. Dr. Cheraghlou has disclosed no relevant financial relationships. Dr. Olszanski has received financial support from Merck and BMS for participated on advisory boards.
A version of this article originally appeared on Medscape.com.
SEATTLE – The use of
Compared with conventional wide local excision, survival was significantly improved among patients treated with Mohs, and a subgroup analysis showed that the survival benefit remained for patients with risk factors.
“At 10 years, overall survival was about 21% higher for those treated with Mohs surgery versus those treated with conventional surgery,” said lead author Shayan Cheraghlou, MD, a dermatology resident at the New York University School of Medicine. “On multivariable analysis, which controlled for tumor and patient factors, Mohs was associated with an over 40% reduction in the hazard for death.”
The findings were presented at the annual meeting of the American College of Mohs Surgery.
MCC is a rare, aggressive, neuroendocrine cutaneous malignancy that carries a high mortality rate. The estimated 5-year survival for patients with localized disease is about 50%, Dr. Cheraghlou noted. “That extrapolates to about 55% for T1 tumors and down to about 30% for T4 tumors.”
Although it’s considered to be a rare cancer, the incidence of MCC has been rapidly rising, and in fact it doubled during the period from the 1990s to the 2010s.
Most commonly treated with wide local excision with or without adjuvant radiation therapy, Mohs as monotherapy may offer an alternative treatment option for patients with MCC. It is generally accepted that the optimal treatment for tumors without regional lymph node involvement is surgical, but the data regarding the optimal surgical approach are mixed. Current National Comprehensive Cancer Network guidelines state that either Mohs surgery or wide local excision can be used.
“However, these guidelines do not indicate a preference for one modality over the other,” said Dr. Cheraghlou, “and present them as interchangeable treatment options.”
A growing body of literature supports Mohs surgery for many types of rare tumors, including MCC. For example, as previously reported at the 2021 ACMS meeting, one study found that Mohs surgery compared favorably with the standard treatment approach when it came to recurrence rates for patients with MCC. The 5-year disease-specific survival rate was 91.2% for patients with stage I disease and 68.6% for patients with stage IIa. These rates were comparable with rates for historical control patients who were treated with wide local excision, with or without radiation (81%-87% for stage I disease, and 63%-67% for stage II).
Study details
In the current study, Dr. Cheraghlou and colleagues sought to evaluate the association of the type of surgical approach with patient survival after excision of early-stage MCC. They conducted a retrospective cohort study using the National Cancer Database to identify cases of MCC with T1/T2 tumors. A total of 2,313 patients who were diagnosed from 2004 to 2018 with pathologically confirmed negative lymph node involvement and who were treated with Mohs surgery or wide lesion excision were included in the analysis.
“About 90% were T1 tumors, about 40% were located on the head and neck, and the vast majority – about 60% – were treated with wide local excision,” he explained. “Only about 5% received Mohs surgery for treatment of the primary tumor.”
But when the researchers assessed survival outcomes, they found that treatment with Mohs surgery was associated with significantly improved overall survival.
The unadjusted 3-, 5-, and 10-year survival rates for patients treated with Mohs was 87.4% (SE: 3.4%), 84.5% (SE: 3.9%), and 81.8% (SE: 4.6%), respectively, while for wide lesion excision, the rates were 86.1% (SE: 0.9%), 76.9% (SE: 1.2%), and 60.9% (SE: 2.0%), respectively.
For patients who underwent treatment with narrow margin excision, survival rates were similar as for those treated with wide lesion excision, with 3-, 5-, and 10-year survival rates of 84.8% (SE: 1.4%), 78.3% (SE: 1.7%), and 60.8% (SE: 3.6%), respectively.
On multivariable survival analysis, Mohs surgery was associated with significantly improved survival, compared with wide lesion excision (hazard ratio, 0.594; P = .038). This was also true after multivariable analysis for patients who had one or more NCCN risk factors, for whom improved survival was also seen with Mohs (HR, 0.530; P = .026).
The results did not differ after a sensitivity analysis that included T3 and T4 tumors.
Given that the use of Mohs was so infrequent, compared with standard surgery, the researchers investigated the factors that were associated with the use of Mohs. High-volume MCC centers were significantly more likely to utilize Mohs than wide lesion excision (odds ratio, 1.993; P < .001), compared with other facilities.
“This study has important implications going forward,” Dr. Cheraghlou concluded. “We think it’s important how few patients were treated with Mohs for Merkel cell, and it was slightly more likely to happen in a high-volume center.”
The reasoning for that may be that high-volume centers are more likely to have a surgeon trained to perform Mohs surgery for MCC. “Or perhaps they are more attuned to the benefits of this procedure,” he said. “We can’t tell that from our data, but its notable that it’s such a small proportion of patients – especially when we consider that it is associated with improved survival for the patients who receive it.”
He added that efforts to increase the utilization of Mohs may yield improved local control and overall survival for these patients. “And perhaps with more data, future versions of guidelines may indicate a preference for Mohs over conventional incisions.”
No changes to current practice
Asked to comment on the study, Anthony J. Olszanski, RPh, MD, associate professor, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that while the results are intriguing, they must be interpreted with caution.
“This study was retrospective in nature, and unrecognized biases can influence results,” he said. “Additionally, given the relative rarity of Merkel cell carcinoma, the sample size is expectantly small.”
But importantly, Dr. Olszanski emphasized, Mohs may more often have been recommended for patients with lesions that appear less aggressive. “Many patients undergoing wide lesion excision may have been referred by Mohs surgeons secondary to features or characteristics of lesions which were worrisome,” he explained. “The results of this study do not opine on why Mohs would impact overall survival over wide lesion excision, a point worthy of consideration. Presently, both modalities can be considered for patients with T1/T2 MCC. The results of this study should not change current practice and would lend themselves to a more robust study.”
No external funding of the study was reported. Dr. Cheraghlou has disclosed no relevant financial relationships. Dr. Olszanski has received financial support from Merck and BMS for participated on advisory boards.
A version of this article originally appeared on Medscape.com.
SEATTLE – The use of
Compared with conventional wide local excision, survival was significantly improved among patients treated with Mohs, and a subgroup analysis showed that the survival benefit remained for patients with risk factors.
“At 10 years, overall survival was about 21% higher for those treated with Mohs surgery versus those treated with conventional surgery,” said lead author Shayan Cheraghlou, MD, a dermatology resident at the New York University School of Medicine. “On multivariable analysis, which controlled for tumor and patient factors, Mohs was associated with an over 40% reduction in the hazard for death.”
The findings were presented at the annual meeting of the American College of Mohs Surgery.
MCC is a rare, aggressive, neuroendocrine cutaneous malignancy that carries a high mortality rate. The estimated 5-year survival for patients with localized disease is about 50%, Dr. Cheraghlou noted. “That extrapolates to about 55% for T1 tumors and down to about 30% for T4 tumors.”
Although it’s considered to be a rare cancer, the incidence of MCC has been rapidly rising, and in fact it doubled during the period from the 1990s to the 2010s.
Most commonly treated with wide local excision with or without adjuvant radiation therapy, Mohs as monotherapy may offer an alternative treatment option for patients with MCC. It is generally accepted that the optimal treatment for tumors without regional lymph node involvement is surgical, but the data regarding the optimal surgical approach are mixed. Current National Comprehensive Cancer Network guidelines state that either Mohs surgery or wide local excision can be used.
“However, these guidelines do not indicate a preference for one modality over the other,” said Dr. Cheraghlou, “and present them as interchangeable treatment options.”
A growing body of literature supports Mohs surgery for many types of rare tumors, including MCC. For example, as previously reported at the 2021 ACMS meeting, one study found that Mohs surgery compared favorably with the standard treatment approach when it came to recurrence rates for patients with MCC. The 5-year disease-specific survival rate was 91.2% for patients with stage I disease and 68.6% for patients with stage IIa. These rates were comparable with rates for historical control patients who were treated with wide local excision, with or without radiation (81%-87% for stage I disease, and 63%-67% for stage II).
Study details
In the current study, Dr. Cheraghlou and colleagues sought to evaluate the association of the type of surgical approach with patient survival after excision of early-stage MCC. They conducted a retrospective cohort study using the National Cancer Database to identify cases of MCC with T1/T2 tumors. A total of 2,313 patients who were diagnosed from 2004 to 2018 with pathologically confirmed negative lymph node involvement and who were treated with Mohs surgery or wide lesion excision were included in the analysis.
“About 90% were T1 tumors, about 40% were located on the head and neck, and the vast majority – about 60% – were treated with wide local excision,” he explained. “Only about 5% received Mohs surgery for treatment of the primary tumor.”
But when the researchers assessed survival outcomes, they found that treatment with Mohs surgery was associated with significantly improved overall survival.
The unadjusted 3-, 5-, and 10-year survival rates for patients treated with Mohs was 87.4% (SE: 3.4%), 84.5% (SE: 3.9%), and 81.8% (SE: 4.6%), respectively, while for wide lesion excision, the rates were 86.1% (SE: 0.9%), 76.9% (SE: 1.2%), and 60.9% (SE: 2.0%), respectively.
For patients who underwent treatment with narrow margin excision, survival rates were similar as for those treated with wide lesion excision, with 3-, 5-, and 10-year survival rates of 84.8% (SE: 1.4%), 78.3% (SE: 1.7%), and 60.8% (SE: 3.6%), respectively.
On multivariable survival analysis, Mohs surgery was associated with significantly improved survival, compared with wide lesion excision (hazard ratio, 0.594; P = .038). This was also true after multivariable analysis for patients who had one or more NCCN risk factors, for whom improved survival was also seen with Mohs (HR, 0.530; P = .026).
The results did not differ after a sensitivity analysis that included T3 and T4 tumors.
Given that the use of Mohs was so infrequent, compared with standard surgery, the researchers investigated the factors that were associated with the use of Mohs. High-volume MCC centers were significantly more likely to utilize Mohs than wide lesion excision (odds ratio, 1.993; P < .001), compared with other facilities.
“This study has important implications going forward,” Dr. Cheraghlou concluded. “We think it’s important how few patients were treated with Mohs for Merkel cell, and it was slightly more likely to happen in a high-volume center.”
The reasoning for that may be that high-volume centers are more likely to have a surgeon trained to perform Mohs surgery for MCC. “Or perhaps they are more attuned to the benefits of this procedure,” he said. “We can’t tell that from our data, but its notable that it’s such a small proportion of patients – especially when we consider that it is associated with improved survival for the patients who receive it.”
He added that efforts to increase the utilization of Mohs may yield improved local control and overall survival for these patients. “And perhaps with more data, future versions of guidelines may indicate a preference for Mohs over conventional incisions.”
No changes to current practice
Asked to comment on the study, Anthony J. Olszanski, RPh, MD, associate professor, department of hematology/oncology, Fox Chase Cancer Center, Philadelphia, noted that while the results are intriguing, they must be interpreted with caution.
“This study was retrospective in nature, and unrecognized biases can influence results,” he said. “Additionally, given the relative rarity of Merkel cell carcinoma, the sample size is expectantly small.”
But importantly, Dr. Olszanski emphasized, Mohs may more often have been recommended for patients with lesions that appear less aggressive. “Many patients undergoing wide lesion excision may have been referred by Mohs surgeons secondary to features or characteristics of lesions which were worrisome,” he explained. “The results of this study do not opine on why Mohs would impact overall survival over wide lesion excision, a point worthy of consideration. Presently, both modalities can be considered for patients with T1/T2 MCC. The results of this study should not change current practice and would lend themselves to a more robust study.”
No external funding of the study was reported. Dr. Cheraghlou has disclosed no relevant financial relationships. Dr. Olszanski has received financial support from Merck and BMS for participated on advisory boards.
A version of this article originally appeared on Medscape.com.
AT ACMS 2023
Study shifts burden of IgG4-related disease to women
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
The incidence and prevalence of IgG4-related disease each rose considerably from 2015 to 2019 in the United States, and the risk of death in those with the immune-mediated condition is about 2.5 times higher than those who are not affected, based on an analysis of claims data from commercially insured adults.
The first population-based study of IgG4-RD incidence, prevalence, and mortality establishes “key benchmarks for informing the diagnosis and management of patients” with a condition “that causes fibrosing inflammatory lesions at nearly any anatomic site,” and wasn’t initially described until 2001, Zachary S. Wallace, MD, and associates said in Annals of the Rheumatic Diseases.
The increases in incidence and prevalence likely reflected increased disease awareness, they suggested. Overall U.S. incidence was 1.2 per 100,000 person-years for the 5-year period of 2015-2019, rising 86% from 0.78 per 100,000 person-years to 1.45 in 2018 before dropping to 1.39 in 2019. The change in prevalence was even greater, increasing 122% from 2.41 per 100,000 persons in 2015 to 5.34 per 100,000 in 2019, the investigators said.
Previous studies had indicated that the majority of patients with IgG4-RD were male, but the current study, using Optum’s Clinformatics Data Mart, which includes commercial health plan and Medicare Advantage members in all 50 states, showed that both incidence and prevalence (see graph) were higher among women, noted Dr. Wallace of Massachusetts General Hospital, Boston, and associates. They identified 524 patients (57.6% female) in the database who met the criteria for IgG4-RD from Jan. 1, 2010, to Dec. 31, 2019.
Incidence over the course of the study “was similar in patients identified as Asian or White but lower in those identified as Black or Hispanic,” they noted, adding that “the prevalence of IgG4-RD during this period reflected similar trends.” A jump in prevalence from 2018 to 2019, however, left White patients with a much higher rate (6.13 per 100,000 persons) than Asian patients (4.54 per 100,000), Black patients (3.42), and Hispanic patients (3.02).
For the mortality analysis, 516 patients with IgG4-RD were age-, sex-, and race-matched with 5,160 patients without IgG4-RD. Mortality was 3.42 and 1.46 per 100 person-years, respectively, over the 5.5 years of follow-up, so IgG4-RD was associated with a 2.5-fold higher risk of death. “The association of IgG4-RD with a higher risk of death was observed across the age spectrum and among both male and female patients,” the researchers said.
“Clinicians across specialties should be aware of IgG4-RD given the incidence, prevalence, and excess risk of death associated with this condition. ... Additional studies are urgently needed to define optimal management strategies to improve survival,” they wrote.
The study was supported by a grant to Massachusetts General Hospital from Sanofi, and Dr. Wallace received funding from the National Institutes of Health/National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Rheumatology Research Foundation. He has received research support and consulting fees from several companies, and four coinvestigators are employees of Sanofi.
FROM ANNALS OF THE RHEUMATIC DISEASES