User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
Scrubs ad that insulted women and DOs pulled after outcry
A video that advertised scrubs but denigrated women and DOs has been removed from the company’s website after fierce backlash.
On Tuesday Kevin Klauer, DO, EJD, directed this tweet to the medical uniform company Figs: “@wearfigs REMOVE YOUR DO offensive web ad immediately or the @AOAforDOs will proceed promptly with a defamation lawsuit on behalf of our members and profession.”
Also on Tuesday, the American Association of Colleges of Osteopathic Medicine demanded a public apology.
The video ad featured a woman carrying a “Medical Terminology for Dummies” book upside down while modeling the pink scrubs from all angles and dancing. At one point in the ad, the camera zooms in on the badge clipped to her waistband that read “DO.”
Agnieszka Solberg, MD, a vascular and interventional radiologist and assistant clinical professor at the University of North Dakota in Grand Forks, was among those voicing pointed criticism on social media.
“This was another hit for our DO colleagues,” she said in an interview, emphasizing that MDs and DOs provide the same level of care.
AACOM tweeted: “We are outraged women physicians & doctors of osteopathic medicine are still attacked in ignorant marketing campaigns. A company like @wearfigs should be ashamed for promoting these stereotypes. We demand the respect we’ve earned AND a public apology.”
Dr. Solberg says this is not the first offense by the company. She said she had stopped buying the company’s scrubs a year ago because the ads “have been portraying female providers as dumb and silly. This was the final straw.”
She said the timing of the ad is suspect as DOs had been swept into a storm of negativity earlier this month, as Medscape Medical News reported, when some questioned the qualifications of President Donald Trump’s physician, Sean Conley, who is a DO.
The scrubs ad ignited criticism across specialties, provider levels, and genders.
Jessica K. Willett, MD, tweeted: “As women physicians in 2020, we still struggle to be taken seriously compared to our male counterparts, as we battle stereotypes like THIS EXACT ONE. We expect the brands we support to reflect the badasses we are.”
The company responded to her tweet: “Thank you so much for the feedback! Totally not our intent – we’re taking down both the men’s and women’s versions of this ASAP! I really appreciate you taking the time to share this.”
The company did not respond to a request for comment but issued an apology on social media: “A lot of you guys have pointed out an insensitive video we had on our site – we are incredibly sorry for any hurt this has caused you, especially our female DOs (who are amazing!) FIGS is a female founded company whose only mission is to make you guys feel awesome.”
The Los Angeles–based company, which Forbes estimated will make $250 million in sales this year, was founded by co-CEOs Heather Hasson and Trina Spear.
A med student wrote on Twitter: “As a female and a DO student, how would I ever “feel awesome” about myself knowing that this is how you view me??? And how you want others to view me??? Women and DO’s have fought stereotypes way too long for you to go ahead and put this out there. Do better.”
Even the company’s apology was tinged with disrespect, some noted, with the use of “you guys” and for what it didn’t include.
As Liesl Young, MD, tweeted: “We are not “guys”, we are women. MD = DO. We stand together.”
Dr. Solberg said the apology came across as an apology that feelings were hurt. It should have detailed the changes the company would make to prevent another incident and address the processes that led to the video.
Dr. Solberg said she is seeing something positive come from the whole incident in that, “women are taking up the torch of feminism in such a volatile and divisive time.”
Dr. Solberg reported no relevant financial relationships.
This article first appeared on Medscape.com.
A video that advertised scrubs but denigrated women and DOs has been removed from the company’s website after fierce backlash.
On Tuesday Kevin Klauer, DO, EJD, directed this tweet to the medical uniform company Figs: “@wearfigs REMOVE YOUR DO offensive web ad immediately or the @AOAforDOs will proceed promptly with a defamation lawsuit on behalf of our members and profession.”
Also on Tuesday, the American Association of Colleges of Osteopathic Medicine demanded a public apology.
The video ad featured a woman carrying a “Medical Terminology for Dummies” book upside down while modeling the pink scrubs from all angles and dancing. At one point in the ad, the camera zooms in on the badge clipped to her waistband that read “DO.”
Agnieszka Solberg, MD, a vascular and interventional radiologist and assistant clinical professor at the University of North Dakota in Grand Forks, was among those voicing pointed criticism on social media.
“This was another hit for our DO colleagues,” she said in an interview, emphasizing that MDs and DOs provide the same level of care.
AACOM tweeted: “We are outraged women physicians & doctors of osteopathic medicine are still attacked in ignorant marketing campaigns. A company like @wearfigs should be ashamed for promoting these stereotypes. We demand the respect we’ve earned AND a public apology.”
Dr. Solberg says this is not the first offense by the company. She said she had stopped buying the company’s scrubs a year ago because the ads “have been portraying female providers as dumb and silly. This was the final straw.”
She said the timing of the ad is suspect as DOs had been swept into a storm of negativity earlier this month, as Medscape Medical News reported, when some questioned the qualifications of President Donald Trump’s physician, Sean Conley, who is a DO.
The scrubs ad ignited criticism across specialties, provider levels, and genders.
Jessica K. Willett, MD, tweeted: “As women physicians in 2020, we still struggle to be taken seriously compared to our male counterparts, as we battle stereotypes like THIS EXACT ONE. We expect the brands we support to reflect the badasses we are.”
The company responded to her tweet: “Thank you so much for the feedback! Totally not our intent – we’re taking down both the men’s and women’s versions of this ASAP! I really appreciate you taking the time to share this.”
The company did not respond to a request for comment but issued an apology on social media: “A lot of you guys have pointed out an insensitive video we had on our site – we are incredibly sorry for any hurt this has caused you, especially our female DOs (who are amazing!) FIGS is a female founded company whose only mission is to make you guys feel awesome.”
The Los Angeles–based company, which Forbes estimated will make $250 million in sales this year, was founded by co-CEOs Heather Hasson and Trina Spear.
A med student wrote on Twitter: “As a female and a DO student, how would I ever “feel awesome” about myself knowing that this is how you view me??? And how you want others to view me??? Women and DO’s have fought stereotypes way too long for you to go ahead and put this out there. Do better.”
Even the company’s apology was tinged with disrespect, some noted, with the use of “you guys” and for what it didn’t include.
As Liesl Young, MD, tweeted: “We are not “guys”, we are women. MD = DO. We stand together.”
Dr. Solberg said the apology came across as an apology that feelings were hurt. It should have detailed the changes the company would make to prevent another incident and address the processes that led to the video.
Dr. Solberg said she is seeing something positive come from the whole incident in that, “women are taking up the torch of feminism in such a volatile and divisive time.”
Dr. Solberg reported no relevant financial relationships.
This article first appeared on Medscape.com.
A video that advertised scrubs but denigrated women and DOs has been removed from the company’s website after fierce backlash.
On Tuesday Kevin Klauer, DO, EJD, directed this tweet to the medical uniform company Figs: “@wearfigs REMOVE YOUR DO offensive web ad immediately or the @AOAforDOs will proceed promptly with a defamation lawsuit on behalf of our members and profession.”
Also on Tuesday, the American Association of Colleges of Osteopathic Medicine demanded a public apology.
The video ad featured a woman carrying a “Medical Terminology for Dummies” book upside down while modeling the pink scrubs from all angles and dancing. At one point in the ad, the camera zooms in on the badge clipped to her waistband that read “DO.”
Agnieszka Solberg, MD, a vascular and interventional radiologist and assistant clinical professor at the University of North Dakota in Grand Forks, was among those voicing pointed criticism on social media.
“This was another hit for our DO colleagues,” she said in an interview, emphasizing that MDs and DOs provide the same level of care.
AACOM tweeted: “We are outraged women physicians & doctors of osteopathic medicine are still attacked in ignorant marketing campaigns. A company like @wearfigs should be ashamed for promoting these stereotypes. We demand the respect we’ve earned AND a public apology.”
Dr. Solberg says this is not the first offense by the company. She said she had stopped buying the company’s scrubs a year ago because the ads “have been portraying female providers as dumb and silly. This was the final straw.”
She said the timing of the ad is suspect as DOs had been swept into a storm of negativity earlier this month, as Medscape Medical News reported, when some questioned the qualifications of President Donald Trump’s physician, Sean Conley, who is a DO.
The scrubs ad ignited criticism across specialties, provider levels, and genders.
Jessica K. Willett, MD, tweeted: “As women physicians in 2020, we still struggle to be taken seriously compared to our male counterparts, as we battle stereotypes like THIS EXACT ONE. We expect the brands we support to reflect the badasses we are.”
The company responded to her tweet: “Thank you so much for the feedback! Totally not our intent – we’re taking down both the men’s and women’s versions of this ASAP! I really appreciate you taking the time to share this.”
The company did not respond to a request for comment but issued an apology on social media: “A lot of you guys have pointed out an insensitive video we had on our site – we are incredibly sorry for any hurt this has caused you, especially our female DOs (who are amazing!) FIGS is a female founded company whose only mission is to make you guys feel awesome.”
The Los Angeles–based company, which Forbes estimated will make $250 million in sales this year, was founded by co-CEOs Heather Hasson and Trina Spear.
A med student wrote on Twitter: “As a female and a DO student, how would I ever “feel awesome” about myself knowing that this is how you view me??? And how you want others to view me??? Women and DO’s have fought stereotypes way too long for you to go ahead and put this out there. Do better.”
Even the company’s apology was tinged with disrespect, some noted, with the use of “you guys” and for what it didn’t include.
As Liesl Young, MD, tweeted: “We are not “guys”, we are women. MD = DO. We stand together.”
Dr. Solberg said the apology came across as an apology that feelings were hurt. It should have detailed the changes the company would make to prevent another incident and address the processes that led to the video.
Dr. Solberg said she is seeing something positive come from the whole incident in that, “women are taking up the torch of feminism in such a volatile and divisive time.”
Dr. Solberg reported no relevant financial relationships.
This article first appeared on Medscape.com.
Systemic sclerosis patients share their perspectives and needs in treatment trials
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
Patients with systemic sclerosis have variable disease progression but often experience debilitating fatigue, pain, and digestive issues – and they’re extremely concerned about progressive organ damage, according to those who spoke at and provided input at a public meeting on patient-focused drug development for the disease.
The virtual meeting was part of the Food and Drug Administration’s Patient-Focused Drug Development (PFDD) initiative, which began in 2012 and aims to provide a systematic way for patients’ experiences, needs, and priorities to be “captured and meaningfully incorporated” into drug development and evaluation.
Patients rate their most impactful symptoms
Dinesh Khanna, MBBS, MSc, a rheumatologist who directs a scleroderma research program at the University of Michigan, Ann Arbor, attended the meeting after giving an opening presentation on the disease to FDA officials, patients, and other participants. In a later interview, he said that patients’ ratings of their most impactful symptoms was especially striking.
Raynaud’s phenomenon, digestive symptoms, and fatigue were the top three answers to a poll question that asked patients what symptom had the most significant impact on daily life, he noted, “and none of these are being [strongly] addressed right now [in clinical trials] apart from Raynaud’s phenomenon, for which there are some trials ongoing.”
He and other researchers are “struggling with what outcomes measures to use [in their studies],” said Dr. Khanna, the Frederick G.L. Huetwell Professor of Rheumatology at the University. “My takeaway from the meeting as a clinical trialist is that we should be paying close attention to the symptoms that patients tell us are the most important. We should be including these in our trial designs as secondary endpoints, if not primary endpoints. We have not done that [thus far], really.”
Approximately 200,000 patients in the United States have scleroderma, and approximately 75,000-80,000 of these patients have systemic scleroderma, or systemic sclerosis, Dr. Khanna said in his opening presentation. Each year, he estimates, about 6,000 new diagnoses of systemic sclerosis are made.
More than 200 people – patients, FDA officials, and others – participated in the PFDD meeting. Patients participated in one of two panels – one focused on health effects and daily impacts, and the other on treatments – or submitted input electronically. All were invited to answer poll questions.
Raj Nair, MD, one of eight FDA leaders attending the meeting, noted in closing remarks that the pain experienced by patients with systemic sclerosis includes severe pain from Raynaud’s phenomenon and pain caused by digital ulcers and by calcinosis. “We heard about how paralyzing the pain from calcinosis is, and that there are very few options for alleviating this pain,” said Dr. Nair, of the division of rheumatology and transplant medicine.
Another takeaway, he said, is that the “fatigue can be severe and debilitating, leading to days where it is impossible to get out of bed,” and that digestive symptoms can also be severe. “Reflux,” he noted, “requires significant medical intervention.”
Patients describe their experiences
Rosemary Lyons, diagnosed with scleroderma 35 years ago, explained that while her skin is no longer hardened, she is overly sensitive to fabrics and skin care products and has difficulty with sleeping and eating. She moved away from family in the Northeast to live in the South where the climate is warmer, but even on a 90-degree night she needs a blanket and two comforters to curb the cold and attempt to sleep.
Impaired gastrointestinal motility has made food her “biggest problem” for the past 10 years, and because of GI symptoms, she can eat only one meal a day. She also experiences fainting, brain fog, and severe fatigue. On a good day, Ms. Lyons noted, she sometimes opts to do some house chores “knowing that I’ll have 1-3 days of recovery.”
Another patient, Amy Harding, said that 22 years after her scleroderma diagnosis, “the calcinosis I get in my fingers, elbows, toes, and ears tops all the prior symptoms.” The skin tightening and digital ulcers that she experienced in the first 10 years have tapered off, and while Raynaud’s symptoms and heartburn have worsened, they are at least partly manageable with medications, unlike the pain from calcinosis.
Treating symptoms vs. disease may be key in risk-benefit analysis
In questions after patient presentations, FDA officials probed for more perspective on issues such as how fatigue should be assessed, the differences between fatigue and brain fog, the impact of calcinosis on functioning, and how much risk patients would be willing to assume from treatments that have side effects and that may or may not modulate the disease and slow disease progression.
Most patients said in response to an FDA poll question that they definitely (almost 40%) or possibly (almost 50%) would be willing to try a hypothetical new self-injectable medication if it were shown to reduce their most impactful symptoms but had side effects.
“I think what [we’ve been hearing] today is that whether we’re working on the symptoms or the disease itself is [the key]” to patients’ risk-benefit analysis, said meeting moderator Capt. Robyn Bent, RN, MS, of the U.S. Public Health Service, and director of the PFDD.
Anita Devine, diagnosed 13 years ago with systemic sclerosis, was one of several panel members who said she would accept more bothersome treatment side effects and risks “if the gain was control of disease progression and overall quality of life ... and organ preservation.” Ms. Devine, who has needed kidney dialysis and multiple hand surgeries, noted that she previously took anti-neoplastic and anti-inflammatory agents “to try to stem the course of my disease, but unfortunately the disease did not abate.”
Treatments for systemic sclerosis include vasodilators, immunosuppressive medications, antifibrotic therapies, and stem cell transplants, Dr. Khanna said in his opening remarks.
Trials of drugs for scleroderma have focused on early disease that may be amenable to treatment, with the exception of trials for pulmonary arterial hypertension, which affects some patients with systemic sclerosis. There are multiple FDA-approved drugs for pulmonary arterial hypertension and more trials are underway.
Outcomes such as pain and fatigue are included in many of the trials currently underway, but they tend to be lower-level secondary outcomes measures that cannot be incorporated into drug labeling or are more “exploratory in nature,” Dr. Khanna said in the interview.
Dr. Khanna disclosed that he is the chief medical officer (an equity position) for CiVi Biopharma/Eicos Sciences Inc., which is developing a drug for Raynaud’s, and serves as a consultant and grant recipient for numerous companies that make or are developing drugs for systemic sclerosis.
The FDA will accept patient comments until Dec. 15, 2020, at which time comments will be compiled into a summary report, Ms. Bent said.
FROM AN FDA PATIENT-FOCUSED DRUG DEVELOPMENT MEETING
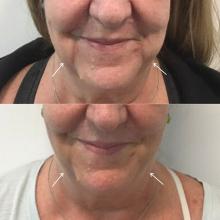
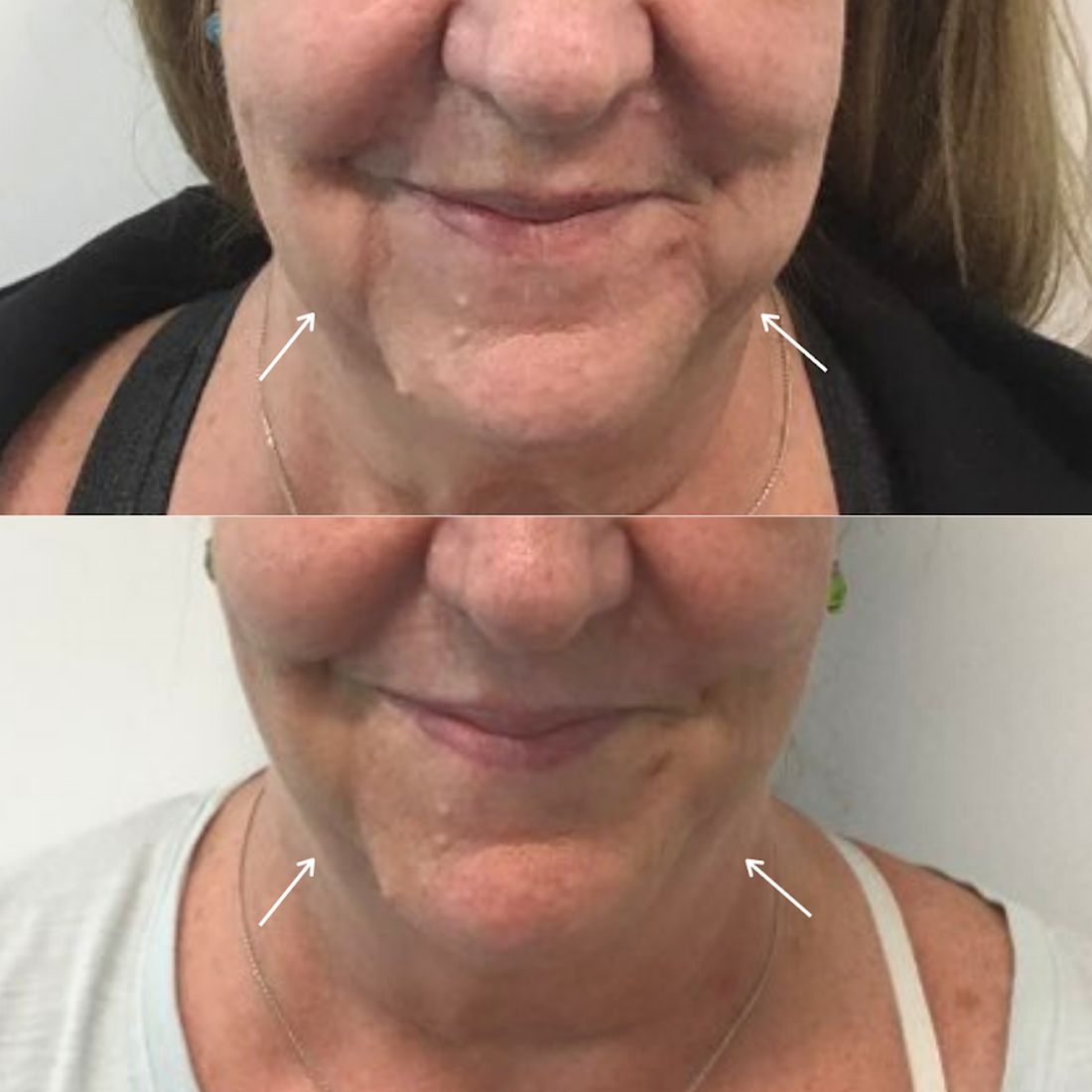
Treating the jowl fat overhang with deoxycholic acid
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
Rejuvenation of the lower face often involves treatment of the submentum and the jowls. Energy-based devices such as lasers, radiofrequency, radiofrequency microneedling, CoolSculpting, and ultrasound have been used in the tightening of the neck and jowls.
However, the only noninvasive injectable treatment approved for the reduction of submental fat is deoxycholic acid (Kybella). The mechanism of action of deoxycholic acid has been documented as adipocyte lysis, followed by a local tissue response with neutrophil infiltration, septal thickening, neocollagenesis, and neovascularization within the subcutaneous layer, with no adverse changes in the dermis or epidermis. This treatment, which has a dose-dependent response, is highly effective for submental fat reduction and jaw contouring.
In my practice, I have found that multiple consecutive treatments with deoxycholic acid (an off-label use) are effective in permanently reducing the jowl overhang with minimal adverse effects.
Jowl fat is a common cause of sagging of the jowls, and there are few alternatives to treatment with surgery or liposuction. Jowl overhang results from multiple factors related to aging, including skeletal resorption, subcutaneous atrophy, superior and inferior fat pad compartment displacement, or mandibular septum dehiscence, which allows for the accumulation of fat pockets to migrate into the neck.
A prospective study published earlier this year describes results in 66 adults with excess jowl fat, who were treated with 2 mg/cm2 of deoxycholic acid. Injections were done in patients with “pinchable fat on the jawline” and “relatively” minimal skin laxity of 0.2 mL spaced approximately 1 cm apart or 0.1 mL spaced 0.5 cm-0.75 cm apart; the mean injection volume was 0.8 mL. After 6 months, 98% of the patients experienced improvement with a mean of 1.8 treatments. Common injection site adverse events included edema, numbness, tenderness, and bruising.
In my experience, injection volumes from 1.0 mL to 1.5 mL of deoxycholic acid can be used in each jowl with minimal adverse events if proper landmarks are followed. It is crucial that the correct patient is selected (one with minimal skin laxity), and that during injection, the fat and skin are pinched away from the underlying musculature and neurovascular structures to avoid injection near the marginal mandibular nerve. Volumes less than 1.0 mL have minimal visible improvements and will require more than 3-4 treatment sessions for optimal results.
. I often see a marked improvement in patients who present prominent marionette lines who have been unhappy with fillers in the lower face. Often, the marionette lines are a result of significant overhang from jowl fat and hyaluronic acid fillers are a temporary and often unsatisfactory treatment option. The use of deoxycholic acid in the treatment of the jowl fat is a highly effective option to minimize the appearance of marionette lines caused by displaced fat pockets in the aging lower face.
Dr. Talakoub and Dr. Wesley are cocontributors to this column. Dr. Talakoub is in private practice in McLean, Va. Dr. Wesley practices dermatology in Beverly Hills, Calif. This month’s column is by Dr. Talakoub. Write to them at [email protected] . They had no relevant disclosures.
Baricitinib reduces adult atopic dermatitis severity in phase 3 study
in the phase 3, double-blind, placebo-controlled, BREEZE-AD7 study.
The study enrolled patients with inadequate responses to topical corticosteroids, according to Kristian Reich, MD, University Medical Center Hamburg-Eppendorf, Hamburg, Germany, and his coauthors.
First test of baricitinib plus topical steroids
Baricitinib, an oral selective Janus kinase (JAK)1/JAK2 inhibitor, inhibits several cytokines in AD pathogenesis, and in two monotherapy studies (BREEZE-AD1 and BREEZE-AD2), it was superior to placebo for reducing several AD clinical signs and symptoms. The current BREEZE-AD7 study is the first to test baricitinib plus background topical corticosteroid therapy, more closely mirroring clinical practice, the authors noted.
BREEZE-AD7 was conducted at 68 centers in 10 countries in Asia, Australia, Europe, and South America. It included 329 adults with moderate to severe AD (mean age around 34 years, and around 34% were female) with inadequate responses to topical corticosteroids documented within the last 6 months. They were randomized 1:1:1 to daily baricitinib 4 mg, daily baricitinib 2 mg, or placebo for 16 weeks. All patients received moderate- and/or low-potency topical corticosteroids (such as 0.1%triamcinolone cream and 2.5% hydrocortisone ointment, respectively) for active lesions.
Significant benefit at 4 mg
At week 16, 31% of AD patients receiving baricitinib 4 mg achieved Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores of 0 (clear) or 1 (almost clear) versus 15% in the placebo group (odds ratio, 2.8; 95% confidence interval, 1.4-5.6; P = .004). Among patients receiving baricitinib 2 mg, 24% achieved vI-GA-AD scores of 0 or 1 (OR, 1.9; 95% CI, 0.9-3.9; P = .08).
The same pattern of improving scores from placebo to baricitinib 2 mg to baricitinib 4 mg persisted, as reflected with secondary endpoints at week 16. Among patients receiving baricitinib 4 mg, 48% achieved Eczema Area Severity Index (EASI) 75 responses, versus 43% and 23% in 2 mg and placebo groups, respectively. Percent changes from baseline in total EASI score were –67%, –58%, –45% for baricitinib 4 mg, baricitinib 2 mg, and placebo, respectively; the proportion of patients achieving 4-point or greater improvements in Itch Numeric Rating Scale (NRS) was 44%, 38%, and 20% for baricitinib 4 mg, baricitinib 2 mg and placebo, respectively.
Similarly, mean change from baseline on the Skin Pain numeric rating scale was –3.7, –3.2, and –2.1 for baricitinib 4 mg, baricitinib 2 mg and placebo. Nighttime itch awakenings were also reduced in a similar progression from placebo to the higher baricitinib dose.
Adverse events dose related
Treatment-related adverse events were reported more frequently in the baricitinib groups (58% baricitinib 4 mg, 56% baricitinib 2 mg) versus placebo 38%. Nasopharyngitis was most common, followed by oral herpes, upper respiratory tract infection, acne, diarrhea, and back pain. Serious adverse event rates were similar across treatment groups. Permanent discontinuation rates were low at 5% for baricitinib 4 mg, 0% for baricitinib 2 mg, and 1% for placebo. The side-effect profile for baricitinib was consistent with prior studies, Dr. Reich and his coauthors reported.
The authors noted further, “data in this study suggest that patients with AD treated with baricitinib may be able to reduce the frequency and total quantity of concomitant TCSs [topical corticosteroids] used, thus mitigating concerns associated with continual or sustained application of topical treatments.”
“Overall, this study provides further evidence to support the efficacy and safety profile of baricitinib for the treatment of moderate-severe AD,” commented one of the authors, Jonathan I. Silverberg, MD, PhD, MPH, of the department of dermatology at George Washington University in Washington.
“In particular, this study shows that adding topical corticosteroids to baricitinib increases the rate of treatment success compared with the efficacy seen in baricitinib monotherapy studies. These data will be important to guide the use of baricitinib with topical corticosteroids in clinical practice. I think these data are also important because they show that baricitinib 4 mg may be more effective than 2 mg in some patients,” he said in an interview.
In late September, the European Medicines Agency’s Committee for Medicinal Products for Human Use recommended approval of oral baricitinib for adults with moderate to severe AD who are candidates for systemic therapy. Baricitinib is approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis. If approved in Europe, it will be the first JAK inhibitor and first oral medication indicated to treat patients with AD.
The study was funded by Eli Lilly and Company under license from Incyte Corporation. Dr. Reich reported receiving fees to the institution for participation in clinical trials from Eli Lilly and Company during the conduct of the study and personal fees for lectures. Dr. Silverberg reported receiving fees from Eli Lilly and Company during the conduct of the study, and fees from companies outside of this work. Other authors also reported disclosures related to Eli Lilly and other pharmaceutical companies, and several authors were Eli Lilly employees.
SOURCE: Reich K et al. JAMA Dermatol. 2020 Sep 30. doi: 10.1001/jamadermatol.2020.3260.
in the phase 3, double-blind, placebo-controlled, BREEZE-AD7 study.
The study enrolled patients with inadequate responses to topical corticosteroids, according to Kristian Reich, MD, University Medical Center Hamburg-Eppendorf, Hamburg, Germany, and his coauthors.
First test of baricitinib plus topical steroids
Baricitinib, an oral selective Janus kinase (JAK)1/JAK2 inhibitor, inhibits several cytokines in AD pathogenesis, and in two monotherapy studies (BREEZE-AD1 and BREEZE-AD2), it was superior to placebo for reducing several AD clinical signs and symptoms. The current BREEZE-AD7 study is the first to test baricitinib plus background topical corticosteroid therapy, more closely mirroring clinical practice, the authors noted.
BREEZE-AD7 was conducted at 68 centers in 10 countries in Asia, Australia, Europe, and South America. It included 329 adults with moderate to severe AD (mean age around 34 years, and around 34% were female) with inadequate responses to topical corticosteroids documented within the last 6 months. They were randomized 1:1:1 to daily baricitinib 4 mg, daily baricitinib 2 mg, or placebo for 16 weeks. All patients received moderate- and/or low-potency topical corticosteroids (such as 0.1%triamcinolone cream and 2.5% hydrocortisone ointment, respectively) for active lesions.
Significant benefit at 4 mg
At week 16, 31% of AD patients receiving baricitinib 4 mg achieved Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores of 0 (clear) or 1 (almost clear) versus 15% in the placebo group (odds ratio, 2.8; 95% confidence interval, 1.4-5.6; P = .004). Among patients receiving baricitinib 2 mg, 24% achieved vI-GA-AD scores of 0 or 1 (OR, 1.9; 95% CI, 0.9-3.9; P = .08).
The same pattern of improving scores from placebo to baricitinib 2 mg to baricitinib 4 mg persisted, as reflected with secondary endpoints at week 16. Among patients receiving baricitinib 4 mg, 48% achieved Eczema Area Severity Index (EASI) 75 responses, versus 43% and 23% in 2 mg and placebo groups, respectively. Percent changes from baseline in total EASI score were –67%, –58%, –45% for baricitinib 4 mg, baricitinib 2 mg, and placebo, respectively; the proportion of patients achieving 4-point or greater improvements in Itch Numeric Rating Scale (NRS) was 44%, 38%, and 20% for baricitinib 4 mg, baricitinib 2 mg and placebo, respectively.
Similarly, mean change from baseline on the Skin Pain numeric rating scale was –3.7, –3.2, and –2.1 for baricitinib 4 mg, baricitinib 2 mg and placebo. Nighttime itch awakenings were also reduced in a similar progression from placebo to the higher baricitinib dose.
Adverse events dose related
Treatment-related adverse events were reported more frequently in the baricitinib groups (58% baricitinib 4 mg, 56% baricitinib 2 mg) versus placebo 38%. Nasopharyngitis was most common, followed by oral herpes, upper respiratory tract infection, acne, diarrhea, and back pain. Serious adverse event rates were similar across treatment groups. Permanent discontinuation rates were low at 5% for baricitinib 4 mg, 0% for baricitinib 2 mg, and 1% for placebo. The side-effect profile for baricitinib was consistent with prior studies, Dr. Reich and his coauthors reported.
The authors noted further, “data in this study suggest that patients with AD treated with baricitinib may be able to reduce the frequency and total quantity of concomitant TCSs [topical corticosteroids] used, thus mitigating concerns associated with continual or sustained application of topical treatments.”
“Overall, this study provides further evidence to support the efficacy and safety profile of baricitinib for the treatment of moderate-severe AD,” commented one of the authors, Jonathan I. Silverberg, MD, PhD, MPH, of the department of dermatology at George Washington University in Washington.
“In particular, this study shows that adding topical corticosteroids to baricitinib increases the rate of treatment success compared with the efficacy seen in baricitinib monotherapy studies. These data will be important to guide the use of baricitinib with topical corticosteroids in clinical practice. I think these data are also important because they show that baricitinib 4 mg may be more effective than 2 mg in some patients,” he said in an interview.
In late September, the European Medicines Agency’s Committee for Medicinal Products for Human Use recommended approval of oral baricitinib for adults with moderate to severe AD who are candidates for systemic therapy. Baricitinib is approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis. If approved in Europe, it will be the first JAK inhibitor and first oral medication indicated to treat patients with AD.
The study was funded by Eli Lilly and Company under license from Incyte Corporation. Dr. Reich reported receiving fees to the institution for participation in clinical trials from Eli Lilly and Company during the conduct of the study and personal fees for lectures. Dr. Silverberg reported receiving fees from Eli Lilly and Company during the conduct of the study, and fees from companies outside of this work. Other authors also reported disclosures related to Eli Lilly and other pharmaceutical companies, and several authors were Eli Lilly employees.
SOURCE: Reich K et al. JAMA Dermatol. 2020 Sep 30. doi: 10.1001/jamadermatol.2020.3260.
in the phase 3, double-blind, placebo-controlled, BREEZE-AD7 study.
The study enrolled patients with inadequate responses to topical corticosteroids, according to Kristian Reich, MD, University Medical Center Hamburg-Eppendorf, Hamburg, Germany, and his coauthors.
First test of baricitinib plus topical steroids
Baricitinib, an oral selective Janus kinase (JAK)1/JAK2 inhibitor, inhibits several cytokines in AD pathogenesis, and in two monotherapy studies (BREEZE-AD1 and BREEZE-AD2), it was superior to placebo for reducing several AD clinical signs and symptoms. The current BREEZE-AD7 study is the first to test baricitinib plus background topical corticosteroid therapy, more closely mirroring clinical practice, the authors noted.
BREEZE-AD7 was conducted at 68 centers in 10 countries in Asia, Australia, Europe, and South America. It included 329 adults with moderate to severe AD (mean age around 34 years, and around 34% were female) with inadequate responses to topical corticosteroids documented within the last 6 months. They were randomized 1:1:1 to daily baricitinib 4 mg, daily baricitinib 2 mg, or placebo for 16 weeks. All patients received moderate- and/or low-potency topical corticosteroids (such as 0.1%triamcinolone cream and 2.5% hydrocortisone ointment, respectively) for active lesions.
Significant benefit at 4 mg
At week 16, 31% of AD patients receiving baricitinib 4 mg achieved Validated Investigator Global Assessment for Atopic Dermatitis (vIGA-AD) scores of 0 (clear) or 1 (almost clear) versus 15% in the placebo group (odds ratio, 2.8; 95% confidence interval, 1.4-5.6; P = .004). Among patients receiving baricitinib 2 mg, 24% achieved vI-GA-AD scores of 0 or 1 (OR, 1.9; 95% CI, 0.9-3.9; P = .08).
The same pattern of improving scores from placebo to baricitinib 2 mg to baricitinib 4 mg persisted, as reflected with secondary endpoints at week 16. Among patients receiving baricitinib 4 mg, 48% achieved Eczema Area Severity Index (EASI) 75 responses, versus 43% and 23% in 2 mg and placebo groups, respectively. Percent changes from baseline in total EASI score were –67%, –58%, –45% for baricitinib 4 mg, baricitinib 2 mg, and placebo, respectively; the proportion of patients achieving 4-point or greater improvements in Itch Numeric Rating Scale (NRS) was 44%, 38%, and 20% for baricitinib 4 mg, baricitinib 2 mg and placebo, respectively.
Similarly, mean change from baseline on the Skin Pain numeric rating scale was –3.7, –3.2, and –2.1 for baricitinib 4 mg, baricitinib 2 mg and placebo. Nighttime itch awakenings were also reduced in a similar progression from placebo to the higher baricitinib dose.
Adverse events dose related
Treatment-related adverse events were reported more frequently in the baricitinib groups (58% baricitinib 4 mg, 56% baricitinib 2 mg) versus placebo 38%. Nasopharyngitis was most common, followed by oral herpes, upper respiratory tract infection, acne, diarrhea, and back pain. Serious adverse event rates were similar across treatment groups. Permanent discontinuation rates were low at 5% for baricitinib 4 mg, 0% for baricitinib 2 mg, and 1% for placebo. The side-effect profile for baricitinib was consistent with prior studies, Dr. Reich and his coauthors reported.
The authors noted further, “data in this study suggest that patients with AD treated with baricitinib may be able to reduce the frequency and total quantity of concomitant TCSs [topical corticosteroids] used, thus mitigating concerns associated with continual or sustained application of topical treatments.”
“Overall, this study provides further evidence to support the efficacy and safety profile of baricitinib for the treatment of moderate-severe AD,” commented one of the authors, Jonathan I. Silverberg, MD, PhD, MPH, of the department of dermatology at George Washington University in Washington.
“In particular, this study shows that adding topical corticosteroids to baricitinib increases the rate of treatment success compared with the efficacy seen in baricitinib monotherapy studies. These data will be important to guide the use of baricitinib with topical corticosteroids in clinical practice. I think these data are also important because they show that baricitinib 4 mg may be more effective than 2 mg in some patients,” he said in an interview.
In late September, the European Medicines Agency’s Committee for Medicinal Products for Human Use recommended approval of oral baricitinib for adults with moderate to severe AD who are candidates for systemic therapy. Baricitinib is approved in the European Union and the United States to treat moderate to severe active rheumatoid arthritis. If approved in Europe, it will be the first JAK inhibitor and first oral medication indicated to treat patients with AD.
The study was funded by Eli Lilly and Company under license from Incyte Corporation. Dr. Reich reported receiving fees to the institution for participation in clinical trials from Eli Lilly and Company during the conduct of the study and personal fees for lectures. Dr. Silverberg reported receiving fees from Eli Lilly and Company during the conduct of the study, and fees from companies outside of this work. Other authors also reported disclosures related to Eli Lilly and other pharmaceutical companies, and several authors were Eli Lilly employees.
SOURCE: Reich K et al. JAMA Dermatol. 2020 Sep 30. doi: 10.1001/jamadermatol.2020.3260.
FROM JAMA DERMATOLOGY
Women make progress in pediatric dermatology leadership
Women account for approximately 78% of the pediatric dermatology workforce, and continue to gain influence through increased numbers of leadership positions and published research, based on data from a review of professional society leaders, grant recipients, and annual meeting presenters from 2010 to 2019.
“Despite extensive research on gender equality in general dermatology, studies have yet to explore the evolving representation of women as leaders and researchers in pediatric dermatology, a field where the majority of board-certified physicians are women,” wrote Catherine Baker, MD, and colleagues. Dr. Baker was a medical student at Geisel School of Medicine at Dartmouth, Hanover, N.H., at the time of the study and is now a resident physician at Brigham and Women’s Hospital, Boston.
In a study published in Pediatric Dermatology, the researchers reviewed data on society leadership, research grants, and annual meeting speakers in order to evaluate the impact of women in pediatric dermatology.
Overall, the Society for Pediatric Dermatology has had 20 women presidents since its founding in 1975 (45%), and 7 of the last 10 since 2011 have been women (70%). The Pediatric Dermatology Research Alliance, founded in 2013, has two cochairs each year, and 75% have been women.
The percentage of women as lead authors of published research in pediatric dermatology increased significantly from 1983 to 2019; 71% of first authors and 65% of senior authors of papers in the journal Pediatric Dermatology in 2019 were women.
In addition, 26 of the 31 physicians (84%) who received SPD/PeDRA pilot project awards between 2008 and 2018 were women, as were 88% of SPD/PeDRA team/collaborative grant winners from 2016 to 2018.
However, named lectures at annual meetings remain an area in which women are underrepresented, the researchers wrote. Although women have been well represented at PeDRA meetings, accounting for 65% of plenary speakers, but they accounted for less than half (44%) of Hurwitz and Founders’ lectures at SPD annual meetings from 2010 to 2019.
The study findings were limited by a lack of data on nonbinary genders and the possibility of error in assessing gender based on name and online profiles, the researchers noted. However, the results suggest that women have increased their influence in pediatric dermatology through leadership and research, although a gender gap persists in roles as senior authors and named lecturers at meetings, they wrote.
Overall, “we expect increasing gender equity in these positions as women continue to play important roles as leaders and researchers in pediatric dermatology,” the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Baker C et al. Pediatr Dermatol. 2020 Jul 9. doi: 10.1111/pde.14266.
Women account for approximately 78% of the pediatric dermatology workforce, and continue to gain influence through increased numbers of leadership positions and published research, based on data from a review of professional society leaders, grant recipients, and annual meeting presenters from 2010 to 2019.
“Despite extensive research on gender equality in general dermatology, studies have yet to explore the evolving representation of women as leaders and researchers in pediatric dermatology, a field where the majority of board-certified physicians are women,” wrote Catherine Baker, MD, and colleagues. Dr. Baker was a medical student at Geisel School of Medicine at Dartmouth, Hanover, N.H., at the time of the study and is now a resident physician at Brigham and Women’s Hospital, Boston.
In a study published in Pediatric Dermatology, the researchers reviewed data on society leadership, research grants, and annual meeting speakers in order to evaluate the impact of women in pediatric dermatology.
Overall, the Society for Pediatric Dermatology has had 20 women presidents since its founding in 1975 (45%), and 7 of the last 10 since 2011 have been women (70%). The Pediatric Dermatology Research Alliance, founded in 2013, has two cochairs each year, and 75% have been women.
The percentage of women as lead authors of published research in pediatric dermatology increased significantly from 1983 to 2019; 71% of first authors and 65% of senior authors of papers in the journal Pediatric Dermatology in 2019 were women.
In addition, 26 of the 31 physicians (84%) who received SPD/PeDRA pilot project awards between 2008 and 2018 were women, as were 88% of SPD/PeDRA team/collaborative grant winners from 2016 to 2018.
However, named lectures at annual meetings remain an area in which women are underrepresented, the researchers wrote. Although women have been well represented at PeDRA meetings, accounting for 65% of plenary speakers, but they accounted for less than half (44%) of Hurwitz and Founders’ lectures at SPD annual meetings from 2010 to 2019.
The study findings were limited by a lack of data on nonbinary genders and the possibility of error in assessing gender based on name and online profiles, the researchers noted. However, the results suggest that women have increased their influence in pediatric dermatology through leadership and research, although a gender gap persists in roles as senior authors and named lecturers at meetings, they wrote.
Overall, “we expect increasing gender equity in these positions as women continue to play important roles as leaders and researchers in pediatric dermatology,” the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Baker C et al. Pediatr Dermatol. 2020 Jul 9. doi: 10.1111/pde.14266.
Women account for approximately 78% of the pediatric dermatology workforce, and continue to gain influence through increased numbers of leadership positions and published research, based on data from a review of professional society leaders, grant recipients, and annual meeting presenters from 2010 to 2019.
“Despite extensive research on gender equality in general dermatology, studies have yet to explore the evolving representation of women as leaders and researchers in pediatric dermatology, a field where the majority of board-certified physicians are women,” wrote Catherine Baker, MD, and colleagues. Dr. Baker was a medical student at Geisel School of Medicine at Dartmouth, Hanover, N.H., at the time of the study and is now a resident physician at Brigham and Women’s Hospital, Boston.
In a study published in Pediatric Dermatology, the researchers reviewed data on society leadership, research grants, and annual meeting speakers in order to evaluate the impact of women in pediatric dermatology.
Overall, the Society for Pediatric Dermatology has had 20 women presidents since its founding in 1975 (45%), and 7 of the last 10 since 2011 have been women (70%). The Pediatric Dermatology Research Alliance, founded in 2013, has two cochairs each year, and 75% have been women.
The percentage of women as lead authors of published research in pediatric dermatology increased significantly from 1983 to 2019; 71% of first authors and 65% of senior authors of papers in the journal Pediatric Dermatology in 2019 were women.
In addition, 26 of the 31 physicians (84%) who received SPD/PeDRA pilot project awards between 2008 and 2018 were women, as were 88% of SPD/PeDRA team/collaborative grant winners from 2016 to 2018.
However, named lectures at annual meetings remain an area in which women are underrepresented, the researchers wrote. Although women have been well represented at PeDRA meetings, accounting for 65% of plenary speakers, but they accounted for less than half (44%) of Hurwitz and Founders’ lectures at SPD annual meetings from 2010 to 2019.
The study findings were limited by a lack of data on nonbinary genders and the possibility of error in assessing gender based on name and online profiles, the researchers noted. However, the results suggest that women have increased their influence in pediatric dermatology through leadership and research, although a gender gap persists in roles as senior authors and named lecturers at meetings, they wrote.
Overall, “we expect increasing gender equity in these positions as women continue to play important roles as leaders and researchers in pediatric dermatology,” the researchers concluded.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Baker C et al. Pediatr Dermatol. 2020 Jul 9. doi: 10.1111/pde.14266.
FROM PEDIATRIC DERMATOLOGY
COVID-19 antibody response not reduced with diabetes
Neither diabetes per se nor hyperglycemia appear to impair the antibody response to SARS-CoV-2, suggesting that a COVID-19 vaccine would be just as effective in people with diabetes as in those without, new research finds.
Results from a study involving 480 patients with confirmed COVID-19 seen at an Italian hospital between February 25 and April 19 were published online October 8 in Diabetologia by Vito Lampasona, MD, and colleagues.
Antibody responses against multiple SARS-CoV-2 antigens among the 27% of patients with COVID-19 and diabetes (preexisting and newly diagnosed) were similar with regard to timing, titers, and classes to those of patients with COVID-19 and without diabetes, and the results did not differ by glucose levels.
Moreover, positivity for immunoglobulin G (IgG) against the SARS-CoV-2 spike receptor-binding domain (RBD) was associated with improved survival regardless of diabetes status.
And as previously shown, high blood glucose levels were strongly associated with greater COVID-19 mortality even in those without diabetes.
This is the first study of the immunologic humoral response against SARS-CoV-2 in patients with hyperglycemia, the authors say.
“The immunological response to a future SARS-CoV-2 vaccine will be assessed when the vaccine becomes available. However, our data allow a cautious optimism regarding effective immunization in individuals with diabetes, as well as in the general population,” wrote Dr. Lampasona of San Raffaele Diabetes Research Institute, IRCCS Ospedale San Raffaele in Milan, and colleagues.
Diabetes and hyperglycemia worsen COVID-19 outcomes
The investigators analyzed the presence of three types of antibody to multiple SARS-CoV-2 antigens in 509 participants: IgG, which is evidence of past infection; IgM, which indicates more recent or current infection; and IgA, which is involved in the mucosal immune response, for example, in the nose where the virus enters the body.
Overall, 452 (88.8%) patients were hospitalized, 79 (15.5%) patients were admitted to intensive care, and 93 (18.3%) patients died during follow-up.
Of the 139 patients with diabetes, 90 (17.7% of the study cohort) already had a diagnosis of diabetes, and 49 (9.6%) were newly diagnosed.
Those with diabetes were older, had a higher body mass index (BMI), and were more likely to have cardiovascular comorbidities, hypertension, and chronic kidney disease. As has been previously reported for diabetes and COVID-19, diabetes was also associated with increased levels of inflammatory biomarkers, hypercoagulopathy, leukocytosis, and neutrophilia.
In multivariate analysis, diabetes status (hazard ratio, 2.32; P = .001), mean fasting plasma glucose (P < .001), and glucose variability (P = .002) were all independently associated with increased mortality and ICU admission. And fasting plasma glucose was associated with increased mortality risk even among those without diabetes (P < .001).
Antibody response similar in patients with and without diabetes
The humoral response against SARS-CoV-2 in patients with diabetes was present and superimposable in terms of timing and antibody titers to that of patients without diabetes, with marginal differences, and was not influenced by glucose levels.
After adjustment for sex, age, and diabetes status and stratification by symptom duration at time of sampling, the development of SARS-CoV-2 RBD IgG antibodies was associated with improved survival, with an HR for time to death of 0.4 (P = .002).
“Of the measured antibody responses, positivity for IgG against the SARS-CoV-2 spike RBD was predictive of survival rate, both in the presence or absence of diabetes,” the authors stressed, with similar HRs for those with diabetes (0.37; P = .013) and without diabetes (0.43; P = .038).
These data confirm “the relevance for patient survival rate of the specific antigen response against spike RBD even in the presence of diabetes, and it underlines how the mechanism explaining the worse clinical outcome in patients with diabetes is unrelated to the antibody response,” they explain.
They added, “This, together with evidence that increased blood glucose levels do predict a poor prognosis even in nondiabetic individuals and the association with increased levels of inflammatory biomarkers and hypercoagulopathy, as well as leukocytosis and neutrophilia, support the speculation that glucose per se could be an independent biological negative factor, acting as a direct regulator of innate immunity.”
“The observed increased severity and mortality risk of COVID-19 pneumonia in patients with hyperglycemia was not the result of an impaired humoral response against SARS-CoV-2.”
“RBD IgG positivity was associated with a remarkable protective effect, allowing for a cautious optimism about the efficacy of future vaccines against SARS-COV-2 in people with diabetes,” they reiterated.
The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Neither diabetes per se nor hyperglycemia appear to impair the antibody response to SARS-CoV-2, suggesting that a COVID-19 vaccine would be just as effective in people with diabetes as in those without, new research finds.
Results from a study involving 480 patients with confirmed COVID-19 seen at an Italian hospital between February 25 and April 19 were published online October 8 in Diabetologia by Vito Lampasona, MD, and colleagues.
Antibody responses against multiple SARS-CoV-2 antigens among the 27% of patients with COVID-19 and diabetes (preexisting and newly diagnosed) were similar with regard to timing, titers, and classes to those of patients with COVID-19 and without diabetes, and the results did not differ by glucose levels.
Moreover, positivity for immunoglobulin G (IgG) against the SARS-CoV-2 spike receptor-binding domain (RBD) was associated with improved survival regardless of diabetes status.
And as previously shown, high blood glucose levels were strongly associated with greater COVID-19 mortality even in those without diabetes.
This is the first study of the immunologic humoral response against SARS-CoV-2 in patients with hyperglycemia, the authors say.
“The immunological response to a future SARS-CoV-2 vaccine will be assessed when the vaccine becomes available. However, our data allow a cautious optimism regarding effective immunization in individuals with diabetes, as well as in the general population,” wrote Dr. Lampasona of San Raffaele Diabetes Research Institute, IRCCS Ospedale San Raffaele in Milan, and colleagues.
Diabetes and hyperglycemia worsen COVID-19 outcomes
The investigators analyzed the presence of three types of antibody to multiple SARS-CoV-2 antigens in 509 participants: IgG, which is evidence of past infection; IgM, which indicates more recent or current infection; and IgA, which is involved in the mucosal immune response, for example, in the nose where the virus enters the body.
Overall, 452 (88.8%) patients were hospitalized, 79 (15.5%) patients were admitted to intensive care, and 93 (18.3%) patients died during follow-up.
Of the 139 patients with diabetes, 90 (17.7% of the study cohort) already had a diagnosis of diabetes, and 49 (9.6%) were newly diagnosed.
Those with diabetes were older, had a higher body mass index (BMI), and were more likely to have cardiovascular comorbidities, hypertension, and chronic kidney disease. As has been previously reported for diabetes and COVID-19, diabetes was also associated with increased levels of inflammatory biomarkers, hypercoagulopathy, leukocytosis, and neutrophilia.
In multivariate analysis, diabetes status (hazard ratio, 2.32; P = .001), mean fasting plasma glucose (P < .001), and glucose variability (P = .002) were all independently associated with increased mortality and ICU admission. And fasting plasma glucose was associated with increased mortality risk even among those without diabetes (P < .001).
Antibody response similar in patients with and without diabetes
The humoral response against SARS-CoV-2 in patients with diabetes was present and superimposable in terms of timing and antibody titers to that of patients without diabetes, with marginal differences, and was not influenced by glucose levels.
After adjustment for sex, age, and diabetes status and stratification by symptom duration at time of sampling, the development of SARS-CoV-2 RBD IgG antibodies was associated with improved survival, with an HR for time to death of 0.4 (P = .002).
“Of the measured antibody responses, positivity for IgG against the SARS-CoV-2 spike RBD was predictive of survival rate, both in the presence or absence of diabetes,” the authors stressed, with similar HRs for those with diabetes (0.37; P = .013) and without diabetes (0.43; P = .038).
These data confirm “the relevance for patient survival rate of the specific antigen response against spike RBD even in the presence of diabetes, and it underlines how the mechanism explaining the worse clinical outcome in patients with diabetes is unrelated to the antibody response,” they explain.
They added, “This, together with evidence that increased blood glucose levels do predict a poor prognosis even in nondiabetic individuals and the association with increased levels of inflammatory biomarkers and hypercoagulopathy, as well as leukocytosis and neutrophilia, support the speculation that glucose per se could be an independent biological negative factor, acting as a direct regulator of innate immunity.”
“The observed increased severity and mortality risk of COVID-19 pneumonia in patients with hyperglycemia was not the result of an impaired humoral response against SARS-CoV-2.”
“RBD IgG positivity was associated with a remarkable protective effect, allowing for a cautious optimism about the efficacy of future vaccines against SARS-COV-2 in people with diabetes,” they reiterated.
The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Neither diabetes per se nor hyperglycemia appear to impair the antibody response to SARS-CoV-2, suggesting that a COVID-19 vaccine would be just as effective in people with diabetes as in those without, new research finds.
Results from a study involving 480 patients with confirmed COVID-19 seen at an Italian hospital between February 25 and April 19 were published online October 8 in Diabetologia by Vito Lampasona, MD, and colleagues.
Antibody responses against multiple SARS-CoV-2 antigens among the 27% of patients with COVID-19 and diabetes (preexisting and newly diagnosed) were similar with regard to timing, titers, and classes to those of patients with COVID-19 and without diabetes, and the results did not differ by glucose levels.
Moreover, positivity for immunoglobulin G (IgG) against the SARS-CoV-2 spike receptor-binding domain (RBD) was associated with improved survival regardless of diabetes status.
And as previously shown, high blood glucose levels were strongly associated with greater COVID-19 mortality even in those without diabetes.
This is the first study of the immunologic humoral response against SARS-CoV-2 in patients with hyperglycemia, the authors say.
“The immunological response to a future SARS-CoV-2 vaccine will be assessed when the vaccine becomes available. However, our data allow a cautious optimism regarding effective immunization in individuals with diabetes, as well as in the general population,” wrote Dr. Lampasona of San Raffaele Diabetes Research Institute, IRCCS Ospedale San Raffaele in Milan, and colleagues.
Diabetes and hyperglycemia worsen COVID-19 outcomes
The investigators analyzed the presence of three types of antibody to multiple SARS-CoV-2 antigens in 509 participants: IgG, which is evidence of past infection; IgM, which indicates more recent or current infection; and IgA, which is involved in the mucosal immune response, for example, in the nose where the virus enters the body.
Overall, 452 (88.8%) patients were hospitalized, 79 (15.5%) patients were admitted to intensive care, and 93 (18.3%) patients died during follow-up.
Of the 139 patients with diabetes, 90 (17.7% of the study cohort) already had a diagnosis of diabetes, and 49 (9.6%) were newly diagnosed.
Those with diabetes were older, had a higher body mass index (BMI), and were more likely to have cardiovascular comorbidities, hypertension, and chronic kidney disease. As has been previously reported for diabetes and COVID-19, diabetes was also associated with increased levels of inflammatory biomarkers, hypercoagulopathy, leukocytosis, and neutrophilia.
In multivariate analysis, diabetes status (hazard ratio, 2.32; P = .001), mean fasting plasma glucose (P < .001), and glucose variability (P = .002) were all independently associated with increased mortality and ICU admission. And fasting plasma glucose was associated with increased mortality risk even among those without diabetes (P < .001).
Antibody response similar in patients with and without diabetes
The humoral response against SARS-CoV-2 in patients with diabetes was present and superimposable in terms of timing and antibody titers to that of patients without diabetes, with marginal differences, and was not influenced by glucose levels.
After adjustment for sex, age, and diabetes status and stratification by symptom duration at time of sampling, the development of SARS-CoV-2 RBD IgG antibodies was associated with improved survival, with an HR for time to death of 0.4 (P = .002).
“Of the measured antibody responses, positivity for IgG against the SARS-CoV-2 spike RBD was predictive of survival rate, both in the presence or absence of diabetes,” the authors stressed, with similar HRs for those with diabetes (0.37; P = .013) and without diabetes (0.43; P = .038).
These data confirm “the relevance for patient survival rate of the specific antigen response against spike RBD even in the presence of diabetes, and it underlines how the mechanism explaining the worse clinical outcome in patients with diabetes is unrelated to the antibody response,” they explain.
They added, “This, together with evidence that increased blood glucose levels do predict a poor prognosis even in nondiabetic individuals and the association with increased levels of inflammatory biomarkers and hypercoagulopathy, as well as leukocytosis and neutrophilia, support the speculation that glucose per se could be an independent biological negative factor, acting as a direct regulator of innate immunity.”
“The observed increased severity and mortality risk of COVID-19 pneumonia in patients with hyperglycemia was not the result of an impaired humoral response against SARS-CoV-2.”
“RBD IgG positivity was associated with a remarkable protective effect, allowing for a cautious optimism about the efficacy of future vaccines against SARS-COV-2 in people with diabetes,” they reiterated.
The authors have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Older age, r/r disease in lymphoma patients tied to increased COVID-19 death rate
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
Patients with B-cell lymphoma are immunocompromised because of the disease and its treatments. This presents the question of their outcomes upon infection with SARS-CoV-2. Researchers assessed the characteristics of patients with lymphoma hospitalized for COVID-19 and analyzed determinants of mortality in a retrospective database study. The investigators looked at data from adult patients with lymphoma who were hospitalized for COVID-19 in March and April 2020 in three French regions.
Older age and relapsed/refractory (r/r) disease in B-cell lymphoma patients were both found to be independent risk factors of increased death rate from COVID-19, according to the online report in EClinicalMedicine, published by The Lancet.
These results encourage “the application of standard Covid-19 treatment, including intubation, for lymphoma patients with Covid-19 lymphoma diagnosis, under first- or second-line chemotherapy, or in remission,” according to Sylvain Lamure, MD, of Montellier (France) University, and colleagues.
The study examined a series of 89 consecutive patients from three French regions who had lymphoma and were hospitalized for COVID-19 in March and April 2020. The population was homogeneous; most patients were diagnosed with B-cell non-Hodgkin lymphoma (NHL) and had been treated for their lymphoma within 1 year.
Promising results for many
There were a significant associations between 30-day mortality and increasing age (over age 70 years) and r/r lymphoma. However, in the absence of those factors, mortality of the lymphoma patients with COVID-19 was comparable with that of the reference French COVID-19 population. In addition, there was no significant impact of active lymphoma treatment that had been given within 1 year, except for those patients who received bendamustine, which was associated with greater mortality, according to the researchers.
With a median follow-up of 33 days from admission, the Kaplan-Meier estimate of 30-day overall survival was 71% (95% confidence interval, 62%-81%). According to histological type of the lymphoma, 30-day overall survival rates were 80% (95% CI, 45%-100%) for Hodgkin lymphoma, 71% (95% CI, 61%-82%) for B-cell non-Hodgkin Lymphoma, and 71% (95% CI, 38%-100%) for T-cell non-Hodgkin Lymphoma.
The main factors associated with mortality were age 70 years and older (hazard ratio, 3.78; 95% CI, 1.73-8.25; P = .0009), hypertension (HR, 2.20; 95% CI, 1.06-4.59; P = .03), previous cancer (HR, 2.11; 95% CI, 0.90-4.92; P = .08), use of bendamustine within 12 months before admission to hospital (HR, 3.05; 95% CI, 1.31-7.11; P = .01), and r/r lymphoma (HR, 2.62; 95% CI, 1.20-5.72; P = .02).
Overall, the Kaplan-Meier estimates of 30-day overall survival were 61% for patients with r/r lymphoma, 52% in patients age 70 years with non–r/r lymphoma, and 88% for patients younger than 70 years with non–r/r, which was comparable with general population survival data among French populations, according to the researchers.
“Longer term clinical follow-up and biological monitoring of immune responses is warranted to explore the impact of lymphoma and its treatment on the immunity and prolonged outcome of Covid-19 patients,” they concluded.
The study was unsponsored. Several of the authors reported financial relationships with a number of biotechnology and pharmaceutical companies.
SOURCE: Lamure S et al. EClinicalMedicine. 2020 Oct 12. doi: 10.1016/j.eclinm.2020.100549.
FROM ECLINICALMEDICINE
Non-Whites remain sorely underrepresented in phase 3 psoriasis trials
Non-White patient participation in phase 3 therapeutic trials for plaque psoriasis is less than 15%, according to a recently published analysis of data from the ClinicalTrials.gov database.
The exact figure drawn from the survey of 82 trials was 14.2%, but 20 (24%) of the trials did not include ethnoracial data at all, and only 65% of those with data had complete data, according to a report in the British Journal of Dermatology by a team of investigators from the department of dermatology at the University of California, San Francisco.
“The remaining studies reported the percentage of white participants only or white participants and one additional ethnoracial group,” reported the investigators, led by Vidhatha D. Reddy, a medical student at UCSF.
The investigators broke down participation by race in all phase 3 plaque psoriasis trials that enrolled adults and had posted results by May 2020. Data from trials of medications yet to be approved were excluded.
Most trials were multinational. The medications evaluated included 11 biologics, 10 topicals, 2 oral systemic agents, and a phosphodiesterase type-4 inhibitor. The 82 trials included in this analysis enrolled 48,846 collectively.
From trials that identified race, 85.8% of 39,161 participants were White, 3.09% of 25,565 patients were Black, 19.55% of 11,364 patients were Hispanic or Latino, and 9.21% of 30,009 patients were Asian. Of trials that included Native Americans or Pacific Islanders, fewer than 2% of participants represented this category.
Non-White patients remain underrepresented even when recognizing differences in the prevalence of psoriasis. For example, one recent survey found the U.S, prevalence of psoriasis to be about half as great in Blacks as it is in Whites (1.9% vs. 3.9%), but the representation of Blacks in the phase 3 trials evaluated by Mr. Reddy and colleagues was more than 20 times lower.
There are many reasons to suspect that lack of diversification in psoriasis trials is impeding optimal care in those underrepresented. Of several examples offered by the authors, one involved differential responses to adalimumab among patients with hidradenitis suppurativa with genetic variants in the BCL2 gene, but the authors reported racially associated genetic differences are not uncommon.
“Estimates have shown that approximately one-fifth of newly developed medications demonstrate interracial/ethnic variability in regard to various factors, such as pharmacokinetics, safety and efficacy profiles, dosing, and pharmacogenetics,” Mr. Reddy and his coinvestigators stated.
Although racial diversity in the design and recruitment for clinical trials has not been a priority in trials involving psoriasis, other skin diseases, or most diseases in general, the authors cited some evidence that this is changing.
“Since 2017, research funded by the National Institutes of Health has been required to report race and ethnicity of participants following an amendment to the Health Revitalization Act,” according to the authors, who suggested that other such initiatives are needed. They advocated “explicit goals to increase recruitment of people of color” as a standard step in clinical trial conduct.
Hypertension trials were cited as an example in which diversity has made a difference.
“Although Black patients are at an elevated risk of developing hypertension, it was not until the enrollment of a substantial proportion of black participants in ALLHAT (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) that enough data on Black patients were available to make specific treatment recommendations in this population,” they noted.
Impossible to know treatment benefits without ethnoracial data
Another investigator who has considered this issue, Junko Takeshita, MD, PhD, an assistant professor of dermatology at the University of Pennsylvania, Philadelphia, agreed.
“Lack of diversity among participants in phase 3 clinical trials for psoriasis is a problem,” said Dr. Takeshita, who led a study of racial differences in perceptions of psoriasis therapies that was published last year.
In that study, “my research group not only found differences in perceptions about biologics between Black and White patients with psoriasis, but we have also shown that Black patients with psoriasis are less likely to receive biologic treatment,” she reported. There are many explanations. For example, she found in another study that Black patients are underrepresented in direct-to-consumer advertisements for biologics.
This problem is not unique to psoriasis. Underrepresentation of Blacks and other ethnoracial groups is true of other skin diseases and many diseases in general, according to Dr. Takeshita. However, she cautioned that the 3% figure for Black participation in psoriasis trials reported by Mr. Reddy and colleagues is not necessarily reflective of trials in the United States.
“This study included international study sites that are recruiting patients from populations with different demographics than the U.S.,” she noted. By including sites with only Asian patients or countries with few Blacks in the population, it dilutes Black representation. She would expect the exact proportion of Black participants to be somewhat higher even if they are “still likely to be underrepresented” if the analysis has been limited to U.S. data.
The research had no funding source. Three of the nine authors reported financial relationships with pharmaceutical companies.
SOURCE: Reddy VD et al. Br J Dermatol. 2020 Sep 17. doi: 10.1111/bjd.19468.
Non-White patient participation in phase 3 therapeutic trials for plaque psoriasis is less than 15%, according to a recently published analysis of data from the ClinicalTrials.gov database.
The exact figure drawn from the survey of 82 trials was 14.2%, but 20 (24%) of the trials did not include ethnoracial data at all, and only 65% of those with data had complete data, according to a report in the British Journal of Dermatology by a team of investigators from the department of dermatology at the University of California, San Francisco.
“The remaining studies reported the percentage of white participants only or white participants and one additional ethnoracial group,” reported the investigators, led by Vidhatha D. Reddy, a medical student at UCSF.
The investigators broke down participation by race in all phase 3 plaque psoriasis trials that enrolled adults and had posted results by May 2020. Data from trials of medications yet to be approved were excluded.
Most trials were multinational. The medications evaluated included 11 biologics, 10 topicals, 2 oral systemic agents, and a phosphodiesterase type-4 inhibitor. The 82 trials included in this analysis enrolled 48,846 collectively.
From trials that identified race, 85.8% of 39,161 participants were White, 3.09% of 25,565 patients were Black, 19.55% of 11,364 patients were Hispanic or Latino, and 9.21% of 30,009 patients were Asian. Of trials that included Native Americans or Pacific Islanders, fewer than 2% of participants represented this category.
Non-White patients remain underrepresented even when recognizing differences in the prevalence of psoriasis. For example, one recent survey found the U.S, prevalence of psoriasis to be about half as great in Blacks as it is in Whites (1.9% vs. 3.9%), but the representation of Blacks in the phase 3 trials evaluated by Mr. Reddy and colleagues was more than 20 times lower.
There are many reasons to suspect that lack of diversification in psoriasis trials is impeding optimal care in those underrepresented. Of several examples offered by the authors, one involved differential responses to adalimumab among patients with hidradenitis suppurativa with genetic variants in the BCL2 gene, but the authors reported racially associated genetic differences are not uncommon.
“Estimates have shown that approximately one-fifth of newly developed medications demonstrate interracial/ethnic variability in regard to various factors, such as pharmacokinetics, safety and efficacy profiles, dosing, and pharmacogenetics,” Mr. Reddy and his coinvestigators stated.
Although racial diversity in the design and recruitment for clinical trials has not been a priority in trials involving psoriasis, other skin diseases, or most diseases in general, the authors cited some evidence that this is changing.
“Since 2017, research funded by the National Institutes of Health has been required to report race and ethnicity of participants following an amendment to the Health Revitalization Act,” according to the authors, who suggested that other such initiatives are needed. They advocated “explicit goals to increase recruitment of people of color” as a standard step in clinical trial conduct.
Hypertension trials were cited as an example in which diversity has made a difference.
“Although Black patients are at an elevated risk of developing hypertension, it was not until the enrollment of a substantial proportion of black participants in ALLHAT (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) that enough data on Black patients were available to make specific treatment recommendations in this population,” they noted.
Impossible to know treatment benefits without ethnoracial data
Another investigator who has considered this issue, Junko Takeshita, MD, PhD, an assistant professor of dermatology at the University of Pennsylvania, Philadelphia, agreed.
“Lack of diversity among participants in phase 3 clinical trials for psoriasis is a problem,” said Dr. Takeshita, who led a study of racial differences in perceptions of psoriasis therapies that was published last year.
In that study, “my research group not only found differences in perceptions about biologics between Black and White patients with psoriasis, but we have also shown that Black patients with psoriasis are less likely to receive biologic treatment,” she reported. There are many explanations. For example, she found in another study that Black patients are underrepresented in direct-to-consumer advertisements for biologics.
This problem is not unique to psoriasis. Underrepresentation of Blacks and other ethnoracial groups is true of other skin diseases and many diseases in general, according to Dr. Takeshita. However, she cautioned that the 3% figure for Black participation in psoriasis trials reported by Mr. Reddy and colleagues is not necessarily reflective of trials in the United States.
“This study included international study sites that are recruiting patients from populations with different demographics than the U.S.,” she noted. By including sites with only Asian patients or countries with few Blacks in the population, it dilutes Black representation. She would expect the exact proportion of Black participants to be somewhat higher even if they are “still likely to be underrepresented” if the analysis has been limited to U.S. data.
The research had no funding source. Three of the nine authors reported financial relationships with pharmaceutical companies.
SOURCE: Reddy VD et al. Br J Dermatol. 2020 Sep 17. doi: 10.1111/bjd.19468.
Non-White patient participation in phase 3 therapeutic trials for plaque psoriasis is less than 15%, according to a recently published analysis of data from the ClinicalTrials.gov database.
The exact figure drawn from the survey of 82 trials was 14.2%, but 20 (24%) of the trials did not include ethnoracial data at all, and only 65% of those with data had complete data, according to a report in the British Journal of Dermatology by a team of investigators from the department of dermatology at the University of California, San Francisco.
“The remaining studies reported the percentage of white participants only or white participants and one additional ethnoracial group,” reported the investigators, led by Vidhatha D. Reddy, a medical student at UCSF.
The investigators broke down participation by race in all phase 3 plaque psoriasis trials that enrolled adults and had posted results by May 2020. Data from trials of medications yet to be approved were excluded.
Most trials were multinational. The medications evaluated included 11 biologics, 10 topicals, 2 oral systemic agents, and a phosphodiesterase type-4 inhibitor. The 82 trials included in this analysis enrolled 48,846 collectively.
From trials that identified race, 85.8% of 39,161 participants were White, 3.09% of 25,565 patients were Black, 19.55% of 11,364 patients were Hispanic or Latino, and 9.21% of 30,009 patients were Asian. Of trials that included Native Americans or Pacific Islanders, fewer than 2% of participants represented this category.
Non-White patients remain underrepresented even when recognizing differences in the prevalence of psoriasis. For example, one recent survey found the U.S, prevalence of psoriasis to be about half as great in Blacks as it is in Whites (1.9% vs. 3.9%), but the representation of Blacks in the phase 3 trials evaluated by Mr. Reddy and colleagues was more than 20 times lower.
There are many reasons to suspect that lack of diversification in psoriasis trials is impeding optimal care in those underrepresented. Of several examples offered by the authors, one involved differential responses to adalimumab among patients with hidradenitis suppurativa with genetic variants in the BCL2 gene, but the authors reported racially associated genetic differences are not uncommon.
“Estimates have shown that approximately one-fifth of newly developed medications demonstrate interracial/ethnic variability in regard to various factors, such as pharmacokinetics, safety and efficacy profiles, dosing, and pharmacogenetics,” Mr. Reddy and his coinvestigators stated.
Although racial diversity in the design and recruitment for clinical trials has not been a priority in trials involving psoriasis, other skin diseases, or most diseases in general, the authors cited some evidence that this is changing.
“Since 2017, research funded by the National Institutes of Health has been required to report race and ethnicity of participants following an amendment to the Health Revitalization Act,” according to the authors, who suggested that other such initiatives are needed. They advocated “explicit goals to increase recruitment of people of color” as a standard step in clinical trial conduct.
Hypertension trials were cited as an example in which diversity has made a difference.
“Although Black patients are at an elevated risk of developing hypertension, it was not until the enrollment of a substantial proportion of black participants in ALLHAT (Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial) that enough data on Black patients were available to make specific treatment recommendations in this population,” they noted.
Impossible to know treatment benefits without ethnoracial data
Another investigator who has considered this issue, Junko Takeshita, MD, PhD, an assistant professor of dermatology at the University of Pennsylvania, Philadelphia, agreed.
“Lack of diversity among participants in phase 3 clinical trials for psoriasis is a problem,” said Dr. Takeshita, who led a study of racial differences in perceptions of psoriasis therapies that was published last year.
In that study, “my research group not only found differences in perceptions about biologics between Black and White patients with psoriasis, but we have also shown that Black patients with psoriasis are less likely to receive biologic treatment,” she reported. There are many explanations. For example, she found in another study that Black patients are underrepresented in direct-to-consumer advertisements for biologics.
This problem is not unique to psoriasis. Underrepresentation of Blacks and other ethnoracial groups is true of other skin diseases and many diseases in general, according to Dr. Takeshita. However, she cautioned that the 3% figure for Black participation in psoriasis trials reported by Mr. Reddy and colleagues is not necessarily reflective of trials in the United States.
“This study included international study sites that are recruiting patients from populations with different demographics than the U.S.,” she noted. By including sites with only Asian patients or countries with few Blacks in the population, it dilutes Black representation. She would expect the exact proportion of Black participants to be somewhat higher even if they are “still likely to be underrepresented” if the analysis has been limited to U.S. data.
The research had no funding source. Three of the nine authors reported financial relationships with pharmaceutical companies.
SOURCE: Reddy VD et al. Br J Dermatol. 2020 Sep 17. doi: 10.1111/bjd.19468.
FROM THE BRITISH JOURNAL OF DERMATOLOGY
Link between vitamin D and ICU outcomes unclear
We can “stop putting money on vitamin D” to help patients who require critical care, said Todd Rice, MD, FCCP.
“Results from vitamin D trials have not been uniformly one way, but they have been pretty uniformly disappointing,” Dr. Rice, from Vanderbilt University Medical Center, Nashville, Tenn., reported at the annual meeting of the American College of Chest Physicians.
Low levels of vitamin D in critically ill COVID-19 patients have been reported in numerous recent studies, and researchers are looking for ways to boost those levels and improve outcomes.
We are seeing “the exact same story” in the critically ill COVID-19 population as we see in the general ICU population, said Dr. Rice. “The whole scenario is repeating itself. I’m pessimistic.”
Still, vitamin D levels can be elevated so, in theory, “the concept makes sense,” he said. There is evidence that, “when given enterally, the levels rise nicely” and vitamin D is absorbed reasonably well.” But is that enough?
When patients are admitted to the ICU, some biomarkers in the body are too high and others are too low. Vitamin D is often too low. So far, though, “supplementing vitamin D in the ICU has not significantly improved outcomes,” said Dr. Rice.
In the Vitamin D to Improve Outcomes by Leveraging Early Treatment (VIOLET) trial, Dr. Rice and colleagues found no statistical benefit when a 540,000 IU boost of vitamin D was administered to 2,624 critically ill patients, as reported by Medscape Medical News.
“Early administration of high-dose enteral vitamin D3 did not provide an advantage over placebo with respect to 90-day mortality or other nonfatal outcomes among critically ill, vitamin D–deficient patients,” the researchers write in their recent report.
In fact, VIOLET ended before enrollment had reached the planned 3,000-patient cohort because the statistical analysis clearly did not show benefit. Those enrolled were in the ICU because of, among other things, pneumonia, sepsis, the need for mechanical ventilation or vasopressors, and risk for acute respiratory distress syndrome.
“It doesn’t look like vitamin D is going to be the answer to our critical care problems,” Dr. Rice said in an interview.
Maintenance dose needed?
One theory suggests that VIOLET might have failed because a maintenance dose is needed after the initial boost of vitamin D.
In the ongoing VITDALIZE trial, critically ill patients with severe vitamin D deficiency (12 ng/mL or less at admission) receive an initial 540,000-IU dose followed by 4,000 IU per day.
The highly anticipated VITDALIZE results are expected in the middle of next year, Dr. Rice reported, so “let’s wait to see.”
“Vitamin D may not have an acute effect,” he theorized. “We can raise your levels, but that doesn’t give you all the benefits of having a sufficient level for a long period of time.”
Another theory suggests that a low level of vitamin D is simply a signal of the severity of disease, not a direct influence on disease pathology.
Some observational data have shown an association between low levels of vitamin D and outcomes in COVID-19 patients (Nutrients. 2020 May 9;12[5]:1359; medRxiv 2020 Apr 24. doi: 10.1101/2020.04.24.20075838; JAMA Netw Open. 2020;3[9]:e2019722; FEBS J. 2020 Jul 23;10.1111/febs.15495; Clin Endocrinol [Oxf]. 2020 Jul 3;10.1111/cen.14276), but some have shown no association (medRxiv. 2020 Jun 26. doi: 10.1101/2020.06.26.20140921; J Public Health [Oxf]. 2020 Aug 18;42[3]:451-60).
Dr. Rice conducted a search of Clinicaltrials.gov immediately before his presentation on Sunday, and found 41 ongoing interventional studies – “not observational studies” – looking at COVID-19 and vitamin D.
“They’re recruiting, they’re enrolling; hopefully we’ll have data soon,” he said.
Researchers have checked a lot of boxes with a resounding yes on the vitamin D question, so there’s reason to think an association does exist for ICU patients, whether or not they have COVID-19.
“Is there a theoretical benefit of vitamin D in the ICU?” Dr. Rice asked. “Yes. Is vitamin D deficient in patients in the ICU? Yes. Is that deficiency associated with poor outcomes? Yes. Can it be replaced safely? Yes.”
However, “we’re not really sure that it improves outcomes,” he said.
A chronic issue?
“Do you think it’s really an issue of the patients being critically ill with vitamin D,” or is it “a chronic issue of having low vitamin D?” asked session moderator Antine Stenbit, MD, PhD, from the University of California, San Diego.
“We don’t know for sure,” Dr. Rice said. Vitamin D might not have a lot of acute effects; it might have effects that are chronic, that work with levels over a period of time, he explained.
“It’s not clear we can correct that with a single dose or with a few days of giving a level that is adequate,” he acknowledged.
Dr. Rice is an investigator in the PETAL network. Dr. Stenbit disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
We can “stop putting money on vitamin D” to help patients who require critical care, said Todd Rice, MD, FCCP.
“Results from vitamin D trials have not been uniformly one way, but they have been pretty uniformly disappointing,” Dr. Rice, from Vanderbilt University Medical Center, Nashville, Tenn., reported at the annual meeting of the American College of Chest Physicians.
Low levels of vitamin D in critically ill COVID-19 patients have been reported in numerous recent studies, and researchers are looking for ways to boost those levels and improve outcomes.
We are seeing “the exact same story” in the critically ill COVID-19 population as we see in the general ICU population, said Dr. Rice. “The whole scenario is repeating itself. I’m pessimistic.”
Still, vitamin D levels can be elevated so, in theory, “the concept makes sense,” he said. There is evidence that, “when given enterally, the levels rise nicely” and vitamin D is absorbed reasonably well.” But is that enough?
When patients are admitted to the ICU, some biomarkers in the body are too high and others are too low. Vitamin D is often too low. So far, though, “supplementing vitamin D in the ICU has not significantly improved outcomes,” said Dr. Rice.
In the Vitamin D to Improve Outcomes by Leveraging Early Treatment (VIOLET) trial, Dr. Rice and colleagues found no statistical benefit when a 540,000 IU boost of vitamin D was administered to 2,624 critically ill patients, as reported by Medscape Medical News.
“Early administration of high-dose enteral vitamin D3 did not provide an advantage over placebo with respect to 90-day mortality or other nonfatal outcomes among critically ill, vitamin D–deficient patients,” the researchers write in their recent report.
In fact, VIOLET ended before enrollment had reached the planned 3,000-patient cohort because the statistical analysis clearly did not show benefit. Those enrolled were in the ICU because of, among other things, pneumonia, sepsis, the need for mechanical ventilation or vasopressors, and risk for acute respiratory distress syndrome.
“It doesn’t look like vitamin D is going to be the answer to our critical care problems,” Dr. Rice said in an interview.
Maintenance dose needed?
One theory suggests that VIOLET might have failed because a maintenance dose is needed after the initial boost of vitamin D.
In the ongoing VITDALIZE trial, critically ill patients with severe vitamin D deficiency (12 ng/mL or less at admission) receive an initial 540,000-IU dose followed by 4,000 IU per day.
The highly anticipated VITDALIZE results are expected in the middle of next year, Dr. Rice reported, so “let’s wait to see.”
“Vitamin D may not have an acute effect,” he theorized. “We can raise your levels, but that doesn’t give you all the benefits of having a sufficient level for a long period of time.”
Another theory suggests that a low level of vitamin D is simply a signal of the severity of disease, not a direct influence on disease pathology.
Some observational data have shown an association between low levels of vitamin D and outcomes in COVID-19 patients (Nutrients. 2020 May 9;12[5]:1359; medRxiv 2020 Apr 24. doi: 10.1101/2020.04.24.20075838; JAMA Netw Open. 2020;3[9]:e2019722; FEBS J. 2020 Jul 23;10.1111/febs.15495; Clin Endocrinol [Oxf]. 2020 Jul 3;10.1111/cen.14276), but some have shown no association (medRxiv. 2020 Jun 26. doi: 10.1101/2020.06.26.20140921; J Public Health [Oxf]. 2020 Aug 18;42[3]:451-60).
Dr. Rice conducted a search of Clinicaltrials.gov immediately before his presentation on Sunday, and found 41 ongoing interventional studies – “not observational studies” – looking at COVID-19 and vitamin D.
“They’re recruiting, they’re enrolling; hopefully we’ll have data soon,” he said.
Researchers have checked a lot of boxes with a resounding yes on the vitamin D question, so there’s reason to think an association does exist for ICU patients, whether or not they have COVID-19.
“Is there a theoretical benefit of vitamin D in the ICU?” Dr. Rice asked. “Yes. Is vitamin D deficient in patients in the ICU? Yes. Is that deficiency associated with poor outcomes? Yes. Can it be replaced safely? Yes.”
However, “we’re not really sure that it improves outcomes,” he said.
A chronic issue?
“Do you think it’s really an issue of the patients being critically ill with vitamin D,” or is it “a chronic issue of having low vitamin D?” asked session moderator Antine Stenbit, MD, PhD, from the University of California, San Diego.
“We don’t know for sure,” Dr. Rice said. Vitamin D might not have a lot of acute effects; it might have effects that are chronic, that work with levels over a period of time, he explained.
“It’s not clear we can correct that with a single dose or with a few days of giving a level that is adequate,” he acknowledged.
Dr. Rice is an investigator in the PETAL network. Dr. Stenbit disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
We can “stop putting money on vitamin D” to help patients who require critical care, said Todd Rice, MD, FCCP.
“Results from vitamin D trials have not been uniformly one way, but they have been pretty uniformly disappointing,” Dr. Rice, from Vanderbilt University Medical Center, Nashville, Tenn., reported at the annual meeting of the American College of Chest Physicians.
Low levels of vitamin D in critically ill COVID-19 patients have been reported in numerous recent studies, and researchers are looking for ways to boost those levels and improve outcomes.
We are seeing “the exact same story” in the critically ill COVID-19 population as we see in the general ICU population, said Dr. Rice. “The whole scenario is repeating itself. I’m pessimistic.”
Still, vitamin D levels can be elevated so, in theory, “the concept makes sense,” he said. There is evidence that, “when given enterally, the levels rise nicely” and vitamin D is absorbed reasonably well.” But is that enough?
When patients are admitted to the ICU, some biomarkers in the body are too high and others are too low. Vitamin D is often too low. So far, though, “supplementing vitamin D in the ICU has not significantly improved outcomes,” said Dr. Rice.
In the Vitamin D to Improve Outcomes by Leveraging Early Treatment (VIOLET) trial, Dr. Rice and colleagues found no statistical benefit when a 540,000 IU boost of vitamin D was administered to 2,624 critically ill patients, as reported by Medscape Medical News.
“Early administration of high-dose enteral vitamin D3 did not provide an advantage over placebo with respect to 90-day mortality or other nonfatal outcomes among critically ill, vitamin D–deficient patients,” the researchers write in their recent report.
In fact, VIOLET ended before enrollment had reached the planned 3,000-patient cohort because the statistical analysis clearly did not show benefit. Those enrolled were in the ICU because of, among other things, pneumonia, sepsis, the need for mechanical ventilation or vasopressors, and risk for acute respiratory distress syndrome.
“It doesn’t look like vitamin D is going to be the answer to our critical care problems,” Dr. Rice said in an interview.
Maintenance dose needed?
One theory suggests that VIOLET might have failed because a maintenance dose is needed after the initial boost of vitamin D.
In the ongoing VITDALIZE trial, critically ill patients with severe vitamin D deficiency (12 ng/mL or less at admission) receive an initial 540,000-IU dose followed by 4,000 IU per day.
The highly anticipated VITDALIZE results are expected in the middle of next year, Dr. Rice reported, so “let’s wait to see.”
“Vitamin D may not have an acute effect,” he theorized. “We can raise your levels, but that doesn’t give you all the benefits of having a sufficient level for a long period of time.”
Another theory suggests that a low level of vitamin D is simply a signal of the severity of disease, not a direct influence on disease pathology.
Some observational data have shown an association between low levels of vitamin D and outcomes in COVID-19 patients (Nutrients. 2020 May 9;12[5]:1359; medRxiv 2020 Apr 24. doi: 10.1101/2020.04.24.20075838; JAMA Netw Open. 2020;3[9]:e2019722; FEBS J. 2020 Jul 23;10.1111/febs.15495; Clin Endocrinol [Oxf]. 2020 Jul 3;10.1111/cen.14276), but some have shown no association (medRxiv. 2020 Jun 26. doi: 10.1101/2020.06.26.20140921; J Public Health [Oxf]. 2020 Aug 18;42[3]:451-60).
Dr. Rice conducted a search of Clinicaltrials.gov immediately before his presentation on Sunday, and found 41 ongoing interventional studies – “not observational studies” – looking at COVID-19 and vitamin D.
“They’re recruiting, they’re enrolling; hopefully we’ll have data soon,” he said.
Researchers have checked a lot of boxes with a resounding yes on the vitamin D question, so there’s reason to think an association does exist for ICU patients, whether or not they have COVID-19.
“Is there a theoretical benefit of vitamin D in the ICU?” Dr. Rice asked. “Yes. Is vitamin D deficient in patients in the ICU? Yes. Is that deficiency associated with poor outcomes? Yes. Can it be replaced safely? Yes.”
However, “we’re not really sure that it improves outcomes,” he said.
A chronic issue?
“Do you think it’s really an issue of the patients being critically ill with vitamin D,” or is it “a chronic issue of having low vitamin D?” asked session moderator Antine Stenbit, MD, PhD, from the University of California, San Diego.
“We don’t know for sure,” Dr. Rice said. Vitamin D might not have a lot of acute effects; it might have effects that are chronic, that work with levels over a period of time, he explained.
“It’s not clear we can correct that with a single dose or with a few days of giving a level that is adequate,” he acknowledged.
Dr. Rice is an investigator in the PETAL network. Dr. Stenbit disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM CHEST 2020
COVID-19 transforms medical education: No ‘back to normal’
The COVID-19 pandemic has thrown a monkey wrench into the medical education landscape across the entire health care spectrum, disrupting the plans of medical students, residents, fellows, and program directors.
As cases of COVID-19 spread across the United States in early 2020, it became clear to training program directors that immediate action was required to meet the needs of medical learners. The challenges were unlike those surrounding the Ebola virus in 2014, “where we could more easily prevent students and trainees from exposure due to the fact that there were simply not significant numbers of cases in the United States,” Tiffany Murano, MD, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. Dr. Murano is professor of emergency medicine at Rutgers New Jersey Medical School, Newark, and president-elect of the Council of Residency Directors in Emergency Medicine. “COVID was a completely different scenario. We quickly realized that not only was personal protective equipment in short supply, but we also lacked the testing and tracking capabilities for potential exposures. Medical students and other supportive workers who were considered nonessential were removed from the clinical setting. This was after a trial of limiting who the students saw, essentially dampening the risk of exposure. But this proved to be flawed as COVID patients presented with symptoms that were unexpected.”
To complicate matters, she continued, many medical clinics either shut down, had limited access, or converted to telemedicine. Elective surgeries were canceled. This led to an overall pause in clinical medical student rotations and no direct patient care activities. As social distancing mandates were instituted, licensing examination testing centers were closed, and exams and on-campus activities were postponed.
Limiting trainee exposure
On the graduate medical education front, some training programs attempted to limit exposure of their trainees to persons under investigation for COVID-19. “As the number of COVID cases grew and encompassed most of what we were seeing in the hospital, it was obvious that residents had to play a vital part in the care of these patients,” said Dr. Murano, who is also a member of the American Council of Graduate Medical Education’s emergency review and recognition committee. “However, there was a consensus among all of the specialties that the procedures that posed the highest risk of exposure would be limited to the most senior or experienced trainees or professionals, and closely supervised by the faculty.”
ACGME activities such as accreditation site visits, clinical environment learning reviews, self-study, and resident and faculty surveys were suspended, postponed, or modified in some way, she said. The ACGME created stages of COVID status to guide sponsoring institutions to suspend learning curricula in order for patients to be cared for. Stage 1 was business as usual, “so there was no significant impact on patient care,” Dr. Murano said. “Stage 2 was increased but manageable clinical demand, while stage 3 was pandemic emergency status, where there were extraordinary circumstances where the clinical demand was so high and strenuous that the routine patient care and education really needed to be reconfigured in order to care for the patients.”
New requirements to manage training
The ACGME also implemented four requirements to manage training that were consistent among institutions, regardless of their COVID stage status. These included making sure that trainees continued to be held to work-hour limit requirements, ensuring adequate resources for training, ensuring that all residents had the appropriate level of supervision at all times, and allowing fellows to function in the core specialty in which they completed their residency training. “This was only possible if the fellows were ABMS [American Board of Medical Specialties] or AOA [American Osteopathic Association] board-eligible, or certified in their core specialty,” Dr. Murano said. “The fellows had to be appointed to the medical staff at the sponsoring institution, and their time spent on the core specialty service would be limited to 20% of their annual education time in any academic year.”
Mindful that there may have been trainees who required a 2-week quarantine period following exposure or potential exposure to COVID-19, some specialty boards showed leniency in residency time required to sit for the written exam. “Testing centers were being forced to close to observe social distancing requirements and heed sanitation recommendations, so exams were either canceled or postponed,” Dr. Murano said. “This posed a special concern for the board certification process, and those specialties with oral examinations had to make a heavy decision regarding whether or not they would allow these exams to take place. Naturally, travel among institutions was suspended or limited, or had quarantine requirements upon returning home from endemic areas. Conferences were either being canceled or converted to virtual formats.”
Subani Chandra, MD, FCCP, of the division of pulmonary, allergy, and critical care medicine at Columbia University, New York, is the internal medicine residency program director and the associate vice-chair of education for the department of medicine, and she recognized the problem created for medical trainees by the changes necessitated by the pandemic.
“The variability in caseloads and clinical exposure has given thrust to the move toward competency-based assessments rather than number- or time-based criteria for determining proficiency and graduation,” she wrote in an email interview. In addition, she noted the impact on medical meetings and the need to adapt. “Early on, before large regional and national conferences adapted to a virtual format, many were canceled altogether. Students, residents, and fellows expecting to have the opportunity to present their scholarly work were suddenly no longer able to do so. Understanding the importance of scholarly interaction, the virtual format of CHEST 2020 is designed with opportunities to present, interact with experts in the field, ask questions, network, and meet mentors.”
No return to ‘normal’
By April 2020, cases in the northeast continued to rise, particularly in the New York, New Jersey, and Connecticut region. “These states were essentially shut down in order to contain spread of the virus,” she said. “This was a real turning point because we realized that things were not going to return to ‘normal’ in the foreseeable future.” With the clinical experience essentially halted for medical students during this time, some medical schools allowed their senior students who met requirements to graduate early. “There were a lot of mixed feelings about this, recognizing that PPE [personal protective equipment] was still in short supply in many areas,” Dr. Murano said. “So, institutions took on these early graduates into roles in which they were not learners in particular, but rather medical workers. They were helping with informatics and technology, telehealth, virtual or telephone call follow-ups, and other tasks like this. There was a movement to virtual learning for the preclinical undergraduate learners, so classes were now online, recorded, or livestreamed.”
Early graduation, matching, and residencies
On April 3, the ACGME released a statement regarding graduating students early and appointing them early to the clinical learning environment. “They pointed out that institutions that were in emergency pandemic status lacked the ability to offer the comprehensive orientation and training in PPE and direct supervision required for new residents at the start of their residency,” Dr. Murano said. “Their opinion maintained that graduating medical students matriculate in their previously matched program, the National Resident Match Program start date, or other date that would be nationally determined to be the beginning of the 2020-2021 academic year.”
As May 2020 rolled around, the overriding feeling was uncertainty regarding when, if, and how medical schools were going to open in the early summer and fall. “There was also uncertainty about how graduating medical students were going to function in their new role as residents,” she said. “Same for the graduating residents. There were some who had signed contracts for jobs months before, and had them rescinded, and physicians were being furloughed due to financial hardships that institutions faced. There was also postponement of board certification exams, so people were uncertain about when they would become board certified.”
July 2020 ushered in what Dr. Murano characterized as “a whole new level of stress.” For medical students in particular, “we were entering the application season for residency positions,” she said. “Due to travel restrictions placed by various states and institutions, away rotations were limited or nonexistent. Application release dates through the Electronic Residency Application Service were moved to later in the year. The United States Medical Licensing Examination clinical skills exam was suspended, and there were modifications made for Education Commission for Foreign Medical Graduates requirements. Letters of recommendation were also going to be limited, so there had to be some degree of leniency within specialties to take a more holistic approach to review of applications for residencies.”
On the graduate medical education front, the ACGME sunsetted the initial stages and created two categories: nonemergency, which was formerly stages 1 and 2, and emergency, which was formerly stage 3. “All emergency stages are applied for and granted at 1-month intervals,” Dr. Murano said. Board certification exams were modified to accommodate either later exams or online formats, and specialties with oral examinations faced the task of potentially creating virtual oral exams.
Despite the challenges, Dr. Chandra has seen medical training programs respond with new ideas. “The flexibility and agile adaptability of the entire educational enterprise has been remarkable. The inherent uncertainty in a very dynamic and changing learning environment can be challenging. Recognizing this, many programs are creating additional ways to support the mental, emotional, physical, and financial health of students, residents, and fellows and all health care workers. The importance of this innovative response cannot be overstated.”
New learning formats
The pandemic forced Dr. Murano and other medical educators to consider unorthodox learning formats, and virtual learning took center stage. “Residency programs had shared national livestream conferences and grand rounds, and there were virtual curricula made for medical students as well as virtual simulation,” she said. “Telemedicine and telehealth really became important parts of education as well, as this may have been the only face-to-face contact that students and residents had with patients who had non–COVID-related complaints.”
To level the playing field for medical residents during this unprecedented time, a work group of the Coalition for Physician Accountability developed a set of recommendations that include limiting the number of letters of recommendation accepted, limiting the number of away rotations, and allowing alternative or less conventional letters of recommendation. “Keeping an open mind and taking a more holistic approach to applicants has really been needed during this time,” Dr. Murano said. “Virtual interview days have been agreed upon for all specialties. They’re safer, and they allow for students to virtually meet faculty and residents from distant programs that in the past would have been a deterrent due to distance and travel costs. This is not without its own downside, as it’s difficult to determine how well a student will fit into a program without [him or her] actually visiting the institution.”
Dr. Chandra agreed that virtual interviews are necessary but have inherent limitations. However, “we will all learn a lot, and very likely the future process will blend the benefits of both virtual and in-person interviews.”
‘We need to keep moving forward’
Dr. Murano concluded her presentation by noting that the COVID-19 pandemic has created opportunities for growth and innovation in medical education, “so we need to keep moving forward. I’ve heard many say that they can’t wait for things to go back to normal. But I think it’s important to go ahead to new and better ways of learning. We’re now thinking outside of the typical education model and are embracing technology and alternative means of education. We don’t know yet if this education is better, worse, or equivalent to traditional methods, but that will be determined and studied in months and years to come, so we’re certainly looking to the future.”
Dr. Murano and Dr. Chandra reported having no financial disclosures.
The COVID-19 pandemic has thrown a monkey wrench into the medical education landscape across the entire health care spectrum, disrupting the plans of medical students, residents, fellows, and program directors.
As cases of COVID-19 spread across the United States in early 2020, it became clear to training program directors that immediate action was required to meet the needs of medical learners. The challenges were unlike those surrounding the Ebola virus in 2014, “where we could more easily prevent students and trainees from exposure due to the fact that there were simply not significant numbers of cases in the United States,” Tiffany Murano, MD, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. Dr. Murano is professor of emergency medicine at Rutgers New Jersey Medical School, Newark, and president-elect of the Council of Residency Directors in Emergency Medicine. “COVID was a completely different scenario. We quickly realized that not only was personal protective equipment in short supply, but we also lacked the testing and tracking capabilities for potential exposures. Medical students and other supportive workers who were considered nonessential were removed from the clinical setting. This was after a trial of limiting who the students saw, essentially dampening the risk of exposure. But this proved to be flawed as COVID patients presented with symptoms that were unexpected.”
To complicate matters, she continued, many medical clinics either shut down, had limited access, or converted to telemedicine. Elective surgeries were canceled. This led to an overall pause in clinical medical student rotations and no direct patient care activities. As social distancing mandates were instituted, licensing examination testing centers were closed, and exams and on-campus activities were postponed.
Limiting trainee exposure
On the graduate medical education front, some training programs attempted to limit exposure of their trainees to persons under investigation for COVID-19. “As the number of COVID cases grew and encompassed most of what we were seeing in the hospital, it was obvious that residents had to play a vital part in the care of these patients,” said Dr. Murano, who is also a member of the American Council of Graduate Medical Education’s emergency review and recognition committee. “However, there was a consensus among all of the specialties that the procedures that posed the highest risk of exposure would be limited to the most senior or experienced trainees or professionals, and closely supervised by the faculty.”
ACGME activities such as accreditation site visits, clinical environment learning reviews, self-study, and resident and faculty surveys were suspended, postponed, or modified in some way, she said. The ACGME created stages of COVID status to guide sponsoring institutions to suspend learning curricula in order for patients to be cared for. Stage 1 was business as usual, “so there was no significant impact on patient care,” Dr. Murano said. “Stage 2 was increased but manageable clinical demand, while stage 3 was pandemic emergency status, where there were extraordinary circumstances where the clinical demand was so high and strenuous that the routine patient care and education really needed to be reconfigured in order to care for the patients.”
New requirements to manage training
The ACGME also implemented four requirements to manage training that were consistent among institutions, regardless of their COVID stage status. These included making sure that trainees continued to be held to work-hour limit requirements, ensuring adequate resources for training, ensuring that all residents had the appropriate level of supervision at all times, and allowing fellows to function in the core specialty in which they completed their residency training. “This was only possible if the fellows were ABMS [American Board of Medical Specialties] or AOA [American Osteopathic Association] board-eligible, or certified in their core specialty,” Dr. Murano said. “The fellows had to be appointed to the medical staff at the sponsoring institution, and their time spent on the core specialty service would be limited to 20% of their annual education time in any academic year.”
Mindful that there may have been trainees who required a 2-week quarantine period following exposure or potential exposure to COVID-19, some specialty boards showed leniency in residency time required to sit for the written exam. “Testing centers were being forced to close to observe social distancing requirements and heed sanitation recommendations, so exams were either canceled or postponed,” Dr. Murano said. “This posed a special concern for the board certification process, and those specialties with oral examinations had to make a heavy decision regarding whether or not they would allow these exams to take place. Naturally, travel among institutions was suspended or limited, or had quarantine requirements upon returning home from endemic areas. Conferences were either being canceled or converted to virtual formats.”
Subani Chandra, MD, FCCP, of the division of pulmonary, allergy, and critical care medicine at Columbia University, New York, is the internal medicine residency program director and the associate vice-chair of education for the department of medicine, and she recognized the problem created for medical trainees by the changes necessitated by the pandemic.
“The variability in caseloads and clinical exposure has given thrust to the move toward competency-based assessments rather than number- or time-based criteria for determining proficiency and graduation,” she wrote in an email interview. In addition, she noted the impact on medical meetings and the need to adapt. “Early on, before large regional and national conferences adapted to a virtual format, many were canceled altogether. Students, residents, and fellows expecting to have the opportunity to present their scholarly work were suddenly no longer able to do so. Understanding the importance of scholarly interaction, the virtual format of CHEST 2020 is designed with opportunities to present, interact with experts in the field, ask questions, network, and meet mentors.”
No return to ‘normal’
By April 2020, cases in the northeast continued to rise, particularly in the New York, New Jersey, and Connecticut region. “These states were essentially shut down in order to contain spread of the virus,” she said. “This was a real turning point because we realized that things were not going to return to ‘normal’ in the foreseeable future.” With the clinical experience essentially halted for medical students during this time, some medical schools allowed their senior students who met requirements to graduate early. “There were a lot of mixed feelings about this, recognizing that PPE [personal protective equipment] was still in short supply in many areas,” Dr. Murano said. “So, institutions took on these early graduates into roles in which they were not learners in particular, but rather medical workers. They were helping with informatics and technology, telehealth, virtual or telephone call follow-ups, and other tasks like this. There was a movement to virtual learning for the preclinical undergraduate learners, so classes were now online, recorded, or livestreamed.”
Early graduation, matching, and residencies
On April 3, the ACGME released a statement regarding graduating students early and appointing them early to the clinical learning environment. “They pointed out that institutions that were in emergency pandemic status lacked the ability to offer the comprehensive orientation and training in PPE and direct supervision required for new residents at the start of their residency,” Dr. Murano said. “Their opinion maintained that graduating medical students matriculate in their previously matched program, the National Resident Match Program start date, or other date that would be nationally determined to be the beginning of the 2020-2021 academic year.”
As May 2020 rolled around, the overriding feeling was uncertainty regarding when, if, and how medical schools were going to open in the early summer and fall. “There was also uncertainty about how graduating medical students were going to function in their new role as residents,” she said. “Same for the graduating residents. There were some who had signed contracts for jobs months before, and had them rescinded, and physicians were being furloughed due to financial hardships that institutions faced. There was also postponement of board certification exams, so people were uncertain about when they would become board certified.”
July 2020 ushered in what Dr. Murano characterized as “a whole new level of stress.” For medical students in particular, “we were entering the application season for residency positions,” she said. “Due to travel restrictions placed by various states and institutions, away rotations were limited or nonexistent. Application release dates through the Electronic Residency Application Service were moved to later in the year. The United States Medical Licensing Examination clinical skills exam was suspended, and there were modifications made for Education Commission for Foreign Medical Graduates requirements. Letters of recommendation were also going to be limited, so there had to be some degree of leniency within specialties to take a more holistic approach to review of applications for residencies.”
On the graduate medical education front, the ACGME sunsetted the initial stages and created two categories: nonemergency, which was formerly stages 1 and 2, and emergency, which was formerly stage 3. “All emergency stages are applied for and granted at 1-month intervals,” Dr. Murano said. Board certification exams were modified to accommodate either later exams or online formats, and specialties with oral examinations faced the task of potentially creating virtual oral exams.
Despite the challenges, Dr. Chandra has seen medical training programs respond with new ideas. “The flexibility and agile adaptability of the entire educational enterprise has been remarkable. The inherent uncertainty in a very dynamic and changing learning environment can be challenging. Recognizing this, many programs are creating additional ways to support the mental, emotional, physical, and financial health of students, residents, and fellows and all health care workers. The importance of this innovative response cannot be overstated.”
New learning formats
The pandemic forced Dr. Murano and other medical educators to consider unorthodox learning formats, and virtual learning took center stage. “Residency programs had shared national livestream conferences and grand rounds, and there were virtual curricula made for medical students as well as virtual simulation,” she said. “Telemedicine and telehealth really became important parts of education as well, as this may have been the only face-to-face contact that students and residents had with patients who had non–COVID-related complaints.”
To level the playing field for medical residents during this unprecedented time, a work group of the Coalition for Physician Accountability developed a set of recommendations that include limiting the number of letters of recommendation accepted, limiting the number of away rotations, and allowing alternative or less conventional letters of recommendation. “Keeping an open mind and taking a more holistic approach to applicants has really been needed during this time,” Dr. Murano said. “Virtual interview days have been agreed upon for all specialties. They’re safer, and they allow for students to virtually meet faculty and residents from distant programs that in the past would have been a deterrent due to distance and travel costs. This is not without its own downside, as it’s difficult to determine how well a student will fit into a program without [him or her] actually visiting the institution.”
Dr. Chandra agreed that virtual interviews are necessary but have inherent limitations. However, “we will all learn a lot, and very likely the future process will blend the benefits of both virtual and in-person interviews.”
‘We need to keep moving forward’
Dr. Murano concluded her presentation by noting that the COVID-19 pandemic has created opportunities for growth and innovation in medical education, “so we need to keep moving forward. I’ve heard many say that they can’t wait for things to go back to normal. But I think it’s important to go ahead to new and better ways of learning. We’re now thinking outside of the typical education model and are embracing technology and alternative means of education. We don’t know yet if this education is better, worse, or equivalent to traditional methods, but that will be determined and studied in months and years to come, so we’re certainly looking to the future.”
Dr. Murano and Dr. Chandra reported having no financial disclosures.
The COVID-19 pandemic has thrown a monkey wrench into the medical education landscape across the entire health care spectrum, disrupting the plans of medical students, residents, fellows, and program directors.
As cases of COVID-19 spread across the United States in early 2020, it became clear to training program directors that immediate action was required to meet the needs of medical learners. The challenges were unlike those surrounding the Ebola virus in 2014, “where we could more easily prevent students and trainees from exposure due to the fact that there were simply not significant numbers of cases in the United States,” Tiffany Murano, MD, said at a Society for Critical Care virtual meeting: COVID-19: What’s Next. Dr. Murano is professor of emergency medicine at Rutgers New Jersey Medical School, Newark, and president-elect of the Council of Residency Directors in Emergency Medicine. “COVID was a completely different scenario. We quickly realized that not only was personal protective equipment in short supply, but we also lacked the testing and tracking capabilities for potential exposures. Medical students and other supportive workers who were considered nonessential were removed from the clinical setting. This was after a trial of limiting who the students saw, essentially dampening the risk of exposure. But this proved to be flawed as COVID patients presented with symptoms that were unexpected.”
To complicate matters, she continued, many medical clinics either shut down, had limited access, or converted to telemedicine. Elective surgeries were canceled. This led to an overall pause in clinical medical student rotations and no direct patient care activities. As social distancing mandates were instituted, licensing examination testing centers were closed, and exams and on-campus activities were postponed.
Limiting trainee exposure
On the graduate medical education front, some training programs attempted to limit exposure of their trainees to persons under investigation for COVID-19. “As the number of COVID cases grew and encompassed most of what we were seeing in the hospital, it was obvious that residents had to play a vital part in the care of these patients,” said Dr. Murano, who is also a member of the American Council of Graduate Medical Education’s emergency review and recognition committee. “However, there was a consensus among all of the specialties that the procedures that posed the highest risk of exposure would be limited to the most senior or experienced trainees or professionals, and closely supervised by the faculty.”
ACGME activities such as accreditation site visits, clinical environment learning reviews, self-study, and resident and faculty surveys were suspended, postponed, or modified in some way, she said. The ACGME created stages of COVID status to guide sponsoring institutions to suspend learning curricula in order for patients to be cared for. Stage 1 was business as usual, “so there was no significant impact on patient care,” Dr. Murano said. “Stage 2 was increased but manageable clinical demand, while stage 3 was pandemic emergency status, where there were extraordinary circumstances where the clinical demand was so high and strenuous that the routine patient care and education really needed to be reconfigured in order to care for the patients.”
New requirements to manage training
The ACGME also implemented four requirements to manage training that were consistent among institutions, regardless of their COVID stage status. These included making sure that trainees continued to be held to work-hour limit requirements, ensuring adequate resources for training, ensuring that all residents had the appropriate level of supervision at all times, and allowing fellows to function in the core specialty in which they completed their residency training. “This was only possible if the fellows were ABMS [American Board of Medical Specialties] or AOA [American Osteopathic Association] board-eligible, or certified in their core specialty,” Dr. Murano said. “The fellows had to be appointed to the medical staff at the sponsoring institution, and their time spent on the core specialty service would be limited to 20% of their annual education time in any academic year.”
Mindful that there may have been trainees who required a 2-week quarantine period following exposure or potential exposure to COVID-19, some specialty boards showed leniency in residency time required to sit for the written exam. “Testing centers were being forced to close to observe social distancing requirements and heed sanitation recommendations, so exams were either canceled or postponed,” Dr. Murano said. “This posed a special concern for the board certification process, and those specialties with oral examinations had to make a heavy decision regarding whether or not they would allow these exams to take place. Naturally, travel among institutions was suspended or limited, or had quarantine requirements upon returning home from endemic areas. Conferences were either being canceled or converted to virtual formats.”
Subani Chandra, MD, FCCP, of the division of pulmonary, allergy, and critical care medicine at Columbia University, New York, is the internal medicine residency program director and the associate vice-chair of education for the department of medicine, and she recognized the problem created for medical trainees by the changes necessitated by the pandemic.
“The variability in caseloads and clinical exposure has given thrust to the move toward competency-based assessments rather than number- or time-based criteria for determining proficiency and graduation,” she wrote in an email interview. In addition, she noted the impact on medical meetings and the need to adapt. “Early on, before large regional and national conferences adapted to a virtual format, many were canceled altogether. Students, residents, and fellows expecting to have the opportunity to present their scholarly work were suddenly no longer able to do so. Understanding the importance of scholarly interaction, the virtual format of CHEST 2020 is designed with opportunities to present, interact with experts in the field, ask questions, network, and meet mentors.”
No return to ‘normal’
By April 2020, cases in the northeast continued to rise, particularly in the New York, New Jersey, and Connecticut region. “These states were essentially shut down in order to contain spread of the virus,” she said. “This was a real turning point because we realized that things were not going to return to ‘normal’ in the foreseeable future.” With the clinical experience essentially halted for medical students during this time, some medical schools allowed their senior students who met requirements to graduate early. “There were a lot of mixed feelings about this, recognizing that PPE [personal protective equipment] was still in short supply in many areas,” Dr. Murano said. “So, institutions took on these early graduates into roles in which they were not learners in particular, but rather medical workers. They were helping with informatics and technology, telehealth, virtual or telephone call follow-ups, and other tasks like this. There was a movement to virtual learning for the preclinical undergraduate learners, so classes were now online, recorded, or livestreamed.”
Early graduation, matching, and residencies
On April 3, the ACGME released a statement regarding graduating students early and appointing them early to the clinical learning environment. “They pointed out that institutions that were in emergency pandemic status lacked the ability to offer the comprehensive orientation and training in PPE and direct supervision required for new residents at the start of their residency,” Dr. Murano said. “Their opinion maintained that graduating medical students matriculate in their previously matched program, the National Resident Match Program start date, or other date that would be nationally determined to be the beginning of the 2020-2021 academic year.”
As May 2020 rolled around, the overriding feeling was uncertainty regarding when, if, and how medical schools were going to open in the early summer and fall. “There was also uncertainty about how graduating medical students were going to function in their new role as residents,” she said. “Same for the graduating residents. There were some who had signed contracts for jobs months before, and had them rescinded, and physicians were being furloughed due to financial hardships that institutions faced. There was also postponement of board certification exams, so people were uncertain about when they would become board certified.”
July 2020 ushered in what Dr. Murano characterized as “a whole new level of stress.” For medical students in particular, “we were entering the application season for residency positions,” she said. “Due to travel restrictions placed by various states and institutions, away rotations were limited or nonexistent. Application release dates through the Electronic Residency Application Service were moved to later in the year. The United States Medical Licensing Examination clinical skills exam was suspended, and there were modifications made for Education Commission for Foreign Medical Graduates requirements. Letters of recommendation were also going to be limited, so there had to be some degree of leniency within specialties to take a more holistic approach to review of applications for residencies.”
On the graduate medical education front, the ACGME sunsetted the initial stages and created two categories: nonemergency, which was formerly stages 1 and 2, and emergency, which was formerly stage 3. “All emergency stages are applied for and granted at 1-month intervals,” Dr. Murano said. Board certification exams were modified to accommodate either later exams or online formats, and specialties with oral examinations faced the task of potentially creating virtual oral exams.
Despite the challenges, Dr. Chandra has seen medical training programs respond with new ideas. “The flexibility and agile adaptability of the entire educational enterprise has been remarkable. The inherent uncertainty in a very dynamic and changing learning environment can be challenging. Recognizing this, many programs are creating additional ways to support the mental, emotional, physical, and financial health of students, residents, and fellows and all health care workers. The importance of this innovative response cannot be overstated.”
New learning formats
The pandemic forced Dr. Murano and other medical educators to consider unorthodox learning formats, and virtual learning took center stage. “Residency programs had shared national livestream conferences and grand rounds, and there were virtual curricula made for medical students as well as virtual simulation,” she said. “Telemedicine and telehealth really became important parts of education as well, as this may have been the only face-to-face contact that students and residents had with patients who had non–COVID-related complaints.”
To level the playing field for medical residents during this unprecedented time, a work group of the Coalition for Physician Accountability developed a set of recommendations that include limiting the number of letters of recommendation accepted, limiting the number of away rotations, and allowing alternative or less conventional letters of recommendation. “Keeping an open mind and taking a more holistic approach to applicants has really been needed during this time,” Dr. Murano said. “Virtual interview days have been agreed upon for all specialties. They’re safer, and they allow for students to virtually meet faculty and residents from distant programs that in the past would have been a deterrent due to distance and travel costs. This is not without its own downside, as it’s difficult to determine how well a student will fit into a program without [him or her] actually visiting the institution.”
Dr. Chandra agreed that virtual interviews are necessary but have inherent limitations. However, “we will all learn a lot, and very likely the future process will blend the benefits of both virtual and in-person interviews.”
‘We need to keep moving forward’
Dr. Murano concluded her presentation by noting that the COVID-19 pandemic has created opportunities for growth and innovation in medical education, “so we need to keep moving forward. I’ve heard many say that they can’t wait for things to go back to normal. But I think it’s important to go ahead to new and better ways of learning. We’re now thinking outside of the typical education model and are embracing technology and alternative means of education. We don’t know yet if this education is better, worse, or equivalent to traditional methods, but that will be determined and studied in months and years to come, so we’re certainly looking to the future.”
Dr. Murano and Dr. Chandra reported having no financial disclosures.
FROM AN SCCM VIRTUAL MEETING