User login
Formerly Skin & Allergy News
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')]
The leading independent newspaper covering dermatology news and commentary.
Phone outreach intervention feasible to reduce SLE readmissions
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
A nurse-led intervention aimed at reducing hospital readmission rates for systemic lupus erythematosus (SLE) is feasible but the jury is out as to whether it can achieve its primary goal, a study has found.
A paper published in Arthritis Care & Research presents the outcomes of a retrospective study using electronic health records that looked at the effect of a quality improvement initiative at the University of Colorado Hospital on readmission rates in two cohorts of 48 and 56 individuals with SLE.
Emily Bowers, MD, of the department of rheumatology at the University of Colorado at Denver, Aurora, and coauthors wrote that hospital readmission rates for SLE are as high as 36% for 30-day readmission. They are significantly higher than for other common chronic diseases such as heart failure, COPD, and diabetes. Readmission for SLE is associated with young age, ethnic or racial diversity, public health insurance, multiorgan involvement, and other comorbidities.
The intervention involved first alerting clinic nurses via the patient’s electronic medical record when the patient was discharged from hospital. The nurses would then call the patient within 48 hours to answer any questions and review their discharge information, and then consult with a rheumatologist on on-call if needed. This call was documented in the patient’s medical record.
In the preintervention cohort, there were 59 hospitalizations among 48 patients, 29% of which were followed by readmission within 30 days; 53% of these readmissions were lupus related. In the cohort that followed introduction of the intervention, there were 73 hospitalizations among 56 individuals, and 19% were followed by readmission within 30 days, 29% of which were lupus related.
After accounting for gender, age, race, and insurance type, the researchers calculated that there was an 89% higher odds of readmission in the nonintervention group than in the intervention group, but the difference was not statistically significant.
The authors noted that although the results were not statistically significant, the low cost of the intervention – requiring around 30 minutes of nursing time – meant even small reductions in the number of emergency department or hospital admissions would make it a cost-effective approach.
“Telephone outreach is an excellent method of providing additional support to patients, assessing clinical needs, reinforcing education about SLE, medications, and common complications such as drug side effects and infections, and allows for patients to ask pertinent questions to RN providers with expertise in the management of lupus,” the authors wrote.
The nurses also recorded qualitative information about the calls, which picked up some patient issues that could be addressed. For example, a patient was discharged with the wrong amount of prednisone, which the nurse was able to fix by adjusting the order and sending it to the pharmacy. Two other patients were confused by their medication instructions and were taking the medication incorrectly; the nurse arranged for the patients to come in for educational session. In another case, the nurse was able to arrange an infusion for the patient, and for one patient with concerns about infection, the nurse was able to advise that person on symptoms and how to seek care.
“To increase implementation of the intervention, we have discussed creating a discharge order set, which would include an automatic EMR message to the nurses,” the authors wrote. “Future studies should explore alternative ways of communicating with our patients after discharge, such as the use of text messaging, messaging through the patient portal in the EMR, or telehealth.”
The authors had no financial disclosures, and there was no outside financial support for the study.
SOURCE: Bowers E et al. Arthritis Care Res. 2020 Aug 29. doi: 10.1002/acr.24435.
FROM ARTHRITIS CARE & RESEARCH
Asymptomatic children may transmit COVID-19 in communities
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
About 22% of children with COVID-19 infections were asymptomatic, and 66% of the symptomatic children had unrecognized symptoms at the time of diagnosis, based on data from a case series of 91 confirmed cases.
Although recent reports suggest that COVID-19 infections in children are generally mild, data on the full spectrum of illness and duration of viral RNA in children are limited, wrote Mi Seon Han, MD, PhD, of Seoul (South Korea) Metropolitan Government–Seoul National University Boramae Medical Center, and colleagues.
To examine the full clinical course and duration of COVID-19 RNA detectability in children with confirmed infections, the researchers reviewed data from 91 individuals with confirmed infections. The children ranged in age from 27 days to 18 years, and 58% were male. The children were monitored at 20 hospitals and 2 isolation facilities for a mean 21.9 days. The findings were published in JAMA Pediatrics.
Overall, COVID-19 viral RNA was present in the study population for a mean 17.6 days, with testing done at a median interval of 3 days. A total of 20 children (22%) were asymptomatic throughout the study period. In these children, viral RNA was detected for a mean 14 days.
“The major hurdle implicated in this study in diagnosing and treating children with COVID-19 is that the researchers noted.
Of the 71 symptomatic children, 47 (66%) had unrecognized symptoms prior to diagnosis, 18 (25%) developed symptoms after diagnosis, and 6 (9%) were diagnosed at the time of symptom onset. The symptomatic children were symptomatic for a median of 11 days; 43 (61%) remained symptomatic at 7 days’ follow-up after the study period, 27 (38%) were symptomatic at 14 days, and 7 (10%) were symptomatic at 21 days.
A total of 41 children had upper respiratory infections (58%) and 22 children (24%) had lower respiratory tract infections. No difference in the duration of virus RNA was detected between children with upper respiratory tract infections and lower respiratory tract infections (average, 18.7 days vs. 19.9 days).
Among the symptomatic children, 46 (65%) had mild cases and 20 (28%) had moderate cases.
For treatment, 14 children (15%) received lopinavir-ritonavir and/or hydroxychloroquine. Two patients had severe illness and received oxygen via nasal prong, without the need for mechanical ventilation. All the children in the case series recovered from their infections with no fatalities.
The study’s main limitation was the inability to analyze the transmission potential of the children because of the quarantine and isolation policies in Korea, the researchers noted. In addition, the researchers did not perform follow-up testing at consistent intervals, so the duration of COVID-19 RNA detection may be inexact.
However, the results suggest “that suspecting and diagnosing COVID-19 in children based on their symptoms without epidemiologic information and virus testing is very challenging,” the researchers emphasized.
“Most of the children with COVID-19 have silent disease, but SARS-CoV-2 RNA can still be detected in the respiratory tract for a prolonged period,” they wrote. More research is needed to explore the potential for disease transmission by children in the community, and increased surveillance with laboratory screening can help identify children with unrecognized infections.
The study is the first known to focus on the frequency of asymptomatic infection in children and the duration of symptoms in both asymptomatic and symptomatic children, Roberta L. DeBiasi, MD, and Meghan Delaney, DO, both affiliated with Children’s National Hospital and Research Institute, Washington, and George Washington University, Washington, wrote in an accompanying editorial. The structure of the Korean public health system “allowed for the sequential observation, testing (median testing interval of every 3 days), and comparison of 91 asymptomatic, presymptomatic, and symptomatic children with mild to moderate upper and lower respiratory tract infection, identified primarily by contact tracing from laboratory-proven cases.”
Two take-home points from the study are that not all infected children are symptomatic, and the duration of symptoms in those who are varies widely, they noted. “Interestingly, this study aligns with adult data in which up to 40% of adults may remain asymptomatic in the face of infection.”
However, “The third and most important take-home point from this study relates to the duration of viral shedding in infected pediatric patients,” Dr. DeBiasi and Dr. Delaney said (JAMA Pediatr. 2020 Aug 28. doi: 10.1001/jamapediatrics.2020.3996).
“Fully half of symptomatic children with both upper and lower tract disease were still shedding virus at 21 days. These are striking data, particularly since 86 of 88 diagnosed children (98%) either had no symptoms or mild or moderate disease,” they explained. The results highlight the need for improvements in qualitative molecular testing and formal studies to identify differences in results from different testing scenarios, such as hospital entry, preprocedure screening, and symptomatic testing. In addition, “these findings are highly relevant to the development of public health strategies to mitigate and contain spread within communities, particularly as affected communities begin their recovery phases.”
The study is important because “schools are opening, and we don’t know what is going to happen,” Michael E. Pichichero, MD, of Rochester General Hospital, N.Y., said in an interview.
“Clinicians, parents, students, school administrators and politicians are worried,” he said. “This study adds to others recently published, bringing into focus the challenges to several suppositions that existed when the COVID-19 pandemic began and over the summer.”
“This study of 91 Korean children tells us that taking a child’s temperature as a screening tool to decide if they may enter school will not be a highly successful strategy,” he said. “Many children are without fever and asymptomatic when infected and contagious. The notion that children shed less virus or shed it for shorter lengths of time we keep learning from this type of research is not true. In another recent study the authors found that children shed as much of the SARS-CoV-2 virus as an adult in the ICU on a ventilator.”
Dr. Pichichero said he was not surprised by the study findings. “A similar paper was published last week in the Journal of Pediatrics from Massachusetts General Hospital, so the findings in the JAMA paper are similar to what has been reported in the United States.”
“Availability of testing will continue to be a challenge in some communities,” said Dr. Pichichero. “Here in the Rochester, New York, area we will use a screening questionnaire based on the CDC [Centers for Disease Control and Prevention] symptom criteria of SARS-CoV-2 infections to decide whom to test.”
As for additional research, “We have so much more to learn about SARS-CoV-2 in children,” he emphasized. “The focus has been on adults because the morbidity and mortality has been greatest in adults, especially the elderly and those with compromised health.”
“The National Institutes of Health has issued a call for more research in children to characterize the spectrum of SARS-CoV-2 illness, including the multisystem inflammatory syndrome in children [MIS-C] and try to identify biomarkers and/or biosignatures for a prognostic algorithm to predict the longitudinal risk of disease severity after a child is exposed to and may be infected with SARS-CoV-2,” said Dr. Pichichero. “NIH has asked researchers to answer the following questions.”
- Why do children have milder illness?
- Are there differences in childhood biology (e.g., gender, puberty, etc.) that contribute to illness severity?
- Are there genetic host differences associated with different disease severity phenotypes, including MIS-C?
- Are there innate mucosal, humoral, cellular and other adaptive immune profiles that are associated with reduced or increased risk of progressive disease, including previous coronavirus infections?
- Will SARS-CoV-2 reinfection cause worse disease as seen with antibody-dependent enhancement (ADE) in other viral infections (e.g., dengue)? Will future vaccines carry a risk of the ADE phenomenon?
- Does substance use (e.g., nicotine, marijuana) exacerbate or trigger MIS-C through immune activation?
“We have no knowledge yet about SARS-CoV-2 vaccination of children, especially young children,” Dr. Pichichero emphasized. “There are different types of vaccines – messenger RNA, adenovirus vector and purified spike proteins of the virus – among others, but questions remain: Will the vaccines work in children? What about side effects? Will the antibodies and cellular immunity protect partially or completely?”
The researchers and editorialists had no financial conflicts to disclose. Dr. Pichichero had no financial conflicts to disclose.
SOURCE: Han MS et al. JAMA Pediatr. 2020 Aug 28. doi:10.1001/jamapediatrics.2020.3988.
FROM JAMA PEDIATRICS
Durable response 5 years after adjuvant combo in melanoma
Adjuvant therapy for patients with high-risk resected melanomas is now a standard of care, but the durability of the benefit gained from this treatment is still unclear.
New data show that the benefit is maintained over the longer term.
At 5 years, just over half of patients (52%) with advanced melanoma who had received a year of adjuvant therapy with two targeted agents were still alive and remained relapse free, compared with 36% of patients who received placebo.
The combination of the investigators concluded.
These data come from the COMBI-AD phase 3 trial and were published online in the New England Journal of Medicine.
“The treatment duration of this adjuvant therapy was 12 months; however, we do not know whether this is the optimal treatment duration,” said lead author Reinhard Dummer, MD, vice chairman, department of dermatology, University of Zürich Hospital. “Early biomarker results suggest that, in a subgroup, longer treatment durations might be necessary. In other patients, a shorter treatment could be sufficient.”
Richard Carvajal, MD, director of the Melanoma Service at New York–Presbyterian Hospital and Columbia University Medical Center, also in New York, said the new data “address prior concerns that any benefit achieved with targeted therapy in the adjuvant setting may be limited in duration.”
“Indeed, with active therapy, over 50% of patients are alive without relapse and 65% of patients are alive without the development of distant metastasis,” he said. “Although overall survival data remain immature, numerical improvement in survival is also reported.”
In an interview, Dr. Carvajal said that the plateaus observed with relapse and distant metastasis-free survival suggest that true disease cures are being achieved with treatment. “Based upon these results, the discussion of adjuvant therapeutic options should include a 12-month course of adjuvant dabrafenib and trametinib, as well as the option of adjuvant anti-PD-1 [programmed death–1] therapy.”
As for how the MEK-BRAF inhibitor combination compares with immunotherapy in this setting, he pointed out that, since there has been no head-to-head comparison of adjuvant targeted therapy and adjuvant nivolumab (Opdivo) or pembrolizumab (Keytruda), it is not possible to conclusively state that one regimen is more effective than another.
“For patients with resected BRAF-mutant melanoma at high risk of disease recurrence, we now have data demonstrating the clinical benefit for a course of adjuvant dabrafenib and trametinib, adjuvant nivolumab and adjuvant pembrolizumab,” said Dr. Carvajal.
“Although the efficacy of adjuvant ipilimumab [Yervoy] as well as adjuvant interferon have also been previously demonstrated, these agents are now appropriate for consideration in extremely rare clinical circumstances given the clinical efficacy and improved toxicity profile of single agent anti-PD-1 therapy.”
“The selection of the most appropriate adjuvant therapy should take into account the preferences of individual patients in terms of toxicity profile and drug administration considerations,” he added.
Study details
The COMBI-AD was a randomized, double-blind, placebo-controlled, phase 3 study conducted in 870 patients with high-risk, stage III, BRAF-V600E/K–mutant melanoma who were treatment naive. Participating patients had undergone surgical resection and had been disease free for ≤12 weeks.
Interim results from this study, reported in 2017, showed 1 year of oral adjuvant therapy with dabrafenib and trametinib provided a 53% lower risk for 3-year recurrence, compared with placebo.
Now, the investigators reported on the 5-year results for relapse-free survival and survival without distant metastasis. They noted that they were unable to analyze overall survival since the required number of events had not been reached.
Patients had been randomly assigned to receive 12 months of oral dabrafenib (at a dose of 150 mg twice daily) plus trametinib (2 mg once daily) or two matched placebos. Patients were followed for 60 months (5 years) for dabrafenib plus trametinib and 58 months for placebo.
At 5 years, the median relapse-free survival was not reached for patients who received the combination therapy group versus 16.6 months in the placebo group (hazard ratio for relapse or death, 0.51).
The percentage of patients who were alive without distant metastasis at 5 years was 65% in the dabrafenib plus trametinib group and 54% in the placebo arm (HR for distant metastasis or death, 0.55).
The hazard ratio for relapse-free survival favored dabrafenib plus trametinib across all patient subgroups that were evaluated in the study, and survival without distant metastasis showed a similar benefit for the combination regardless of disease stage.
Subsequent therapy was needed in 40% of patients who received dabrafenib plus trametinib and by 54% of those in the placebo group, with the most common treatments being immunotherapy in the combination-therapy group [26%] and small molecule–targeted therapy in the placebo group (35%).
A viable option
Dr. Dummer noted that, when this clinical trial was designed, all patients had to undergo aggressive surgery that involved lymph node dissection. “Nowadays, based on the lack of improvement on progression-free survival and overall survival, the surgical procedures are less aggressive and today we do not recommend aggressive lymph node dissection in patients that qualify for adjuvant therapy. In patients that do not have the BRAF mutation, there is the possibility of giving immunotherapy.”
He added that there is an urgent need for biomarkers that can identify early progression during adjuvant therapy. “Potentially, these patients would profit from immunotherapy alone or from combination using targeted therapy and immunotherapy,” Dr. Dummer said.
The study was funded by GlaxoSmithKline and Novartis. Dr. Dummer has declared multiple relationships with industry.
A version of this article originally appeared on Medscape.com.
Adjuvant therapy for patients with high-risk resected melanomas is now a standard of care, but the durability of the benefit gained from this treatment is still unclear.
New data show that the benefit is maintained over the longer term.
At 5 years, just over half of patients (52%) with advanced melanoma who had received a year of adjuvant therapy with two targeted agents were still alive and remained relapse free, compared with 36% of patients who received placebo.
The combination of the investigators concluded.
These data come from the COMBI-AD phase 3 trial and were published online in the New England Journal of Medicine.
“The treatment duration of this adjuvant therapy was 12 months; however, we do not know whether this is the optimal treatment duration,” said lead author Reinhard Dummer, MD, vice chairman, department of dermatology, University of Zürich Hospital. “Early biomarker results suggest that, in a subgroup, longer treatment durations might be necessary. In other patients, a shorter treatment could be sufficient.”
Richard Carvajal, MD, director of the Melanoma Service at New York–Presbyterian Hospital and Columbia University Medical Center, also in New York, said the new data “address prior concerns that any benefit achieved with targeted therapy in the adjuvant setting may be limited in duration.”
“Indeed, with active therapy, over 50% of patients are alive without relapse and 65% of patients are alive without the development of distant metastasis,” he said. “Although overall survival data remain immature, numerical improvement in survival is also reported.”
In an interview, Dr. Carvajal said that the plateaus observed with relapse and distant metastasis-free survival suggest that true disease cures are being achieved with treatment. “Based upon these results, the discussion of adjuvant therapeutic options should include a 12-month course of adjuvant dabrafenib and trametinib, as well as the option of adjuvant anti-PD-1 [programmed death–1] therapy.”
As for how the MEK-BRAF inhibitor combination compares with immunotherapy in this setting, he pointed out that, since there has been no head-to-head comparison of adjuvant targeted therapy and adjuvant nivolumab (Opdivo) or pembrolizumab (Keytruda), it is not possible to conclusively state that one regimen is more effective than another.
“For patients with resected BRAF-mutant melanoma at high risk of disease recurrence, we now have data demonstrating the clinical benefit for a course of adjuvant dabrafenib and trametinib, adjuvant nivolumab and adjuvant pembrolizumab,” said Dr. Carvajal.
“Although the efficacy of adjuvant ipilimumab [Yervoy] as well as adjuvant interferon have also been previously demonstrated, these agents are now appropriate for consideration in extremely rare clinical circumstances given the clinical efficacy and improved toxicity profile of single agent anti-PD-1 therapy.”
“The selection of the most appropriate adjuvant therapy should take into account the preferences of individual patients in terms of toxicity profile and drug administration considerations,” he added.
Study details
The COMBI-AD was a randomized, double-blind, placebo-controlled, phase 3 study conducted in 870 patients with high-risk, stage III, BRAF-V600E/K–mutant melanoma who were treatment naive. Participating patients had undergone surgical resection and had been disease free for ≤12 weeks.
Interim results from this study, reported in 2017, showed 1 year of oral adjuvant therapy with dabrafenib and trametinib provided a 53% lower risk for 3-year recurrence, compared with placebo.
Now, the investigators reported on the 5-year results for relapse-free survival and survival without distant metastasis. They noted that they were unable to analyze overall survival since the required number of events had not been reached.
Patients had been randomly assigned to receive 12 months of oral dabrafenib (at a dose of 150 mg twice daily) plus trametinib (2 mg once daily) or two matched placebos. Patients were followed for 60 months (5 years) for dabrafenib plus trametinib and 58 months for placebo.
At 5 years, the median relapse-free survival was not reached for patients who received the combination therapy group versus 16.6 months in the placebo group (hazard ratio for relapse or death, 0.51).
The percentage of patients who were alive without distant metastasis at 5 years was 65% in the dabrafenib plus trametinib group and 54% in the placebo arm (HR for distant metastasis or death, 0.55).
The hazard ratio for relapse-free survival favored dabrafenib plus trametinib across all patient subgroups that were evaluated in the study, and survival without distant metastasis showed a similar benefit for the combination regardless of disease stage.
Subsequent therapy was needed in 40% of patients who received dabrafenib plus trametinib and by 54% of those in the placebo group, with the most common treatments being immunotherapy in the combination-therapy group [26%] and small molecule–targeted therapy in the placebo group (35%).
A viable option
Dr. Dummer noted that, when this clinical trial was designed, all patients had to undergo aggressive surgery that involved lymph node dissection. “Nowadays, based on the lack of improvement on progression-free survival and overall survival, the surgical procedures are less aggressive and today we do not recommend aggressive lymph node dissection in patients that qualify for adjuvant therapy. In patients that do not have the BRAF mutation, there is the possibility of giving immunotherapy.”
He added that there is an urgent need for biomarkers that can identify early progression during adjuvant therapy. “Potentially, these patients would profit from immunotherapy alone or from combination using targeted therapy and immunotherapy,” Dr. Dummer said.
The study was funded by GlaxoSmithKline and Novartis. Dr. Dummer has declared multiple relationships with industry.
A version of this article originally appeared on Medscape.com.
Adjuvant therapy for patients with high-risk resected melanomas is now a standard of care, but the durability of the benefit gained from this treatment is still unclear.
New data show that the benefit is maintained over the longer term.
At 5 years, just over half of patients (52%) with advanced melanoma who had received a year of adjuvant therapy with two targeted agents were still alive and remained relapse free, compared with 36% of patients who received placebo.
The combination of the investigators concluded.
These data come from the COMBI-AD phase 3 trial and were published online in the New England Journal of Medicine.
“The treatment duration of this adjuvant therapy was 12 months; however, we do not know whether this is the optimal treatment duration,” said lead author Reinhard Dummer, MD, vice chairman, department of dermatology, University of Zürich Hospital. “Early biomarker results suggest that, in a subgroup, longer treatment durations might be necessary. In other patients, a shorter treatment could be sufficient.”
Richard Carvajal, MD, director of the Melanoma Service at New York–Presbyterian Hospital and Columbia University Medical Center, also in New York, said the new data “address prior concerns that any benefit achieved with targeted therapy in the adjuvant setting may be limited in duration.”
“Indeed, with active therapy, over 50% of patients are alive without relapse and 65% of patients are alive without the development of distant metastasis,” he said. “Although overall survival data remain immature, numerical improvement in survival is also reported.”
In an interview, Dr. Carvajal said that the plateaus observed with relapse and distant metastasis-free survival suggest that true disease cures are being achieved with treatment. “Based upon these results, the discussion of adjuvant therapeutic options should include a 12-month course of adjuvant dabrafenib and trametinib, as well as the option of adjuvant anti-PD-1 [programmed death–1] therapy.”
As for how the MEK-BRAF inhibitor combination compares with immunotherapy in this setting, he pointed out that, since there has been no head-to-head comparison of adjuvant targeted therapy and adjuvant nivolumab (Opdivo) or pembrolizumab (Keytruda), it is not possible to conclusively state that one regimen is more effective than another.
“For patients with resected BRAF-mutant melanoma at high risk of disease recurrence, we now have data demonstrating the clinical benefit for a course of adjuvant dabrafenib and trametinib, adjuvant nivolumab and adjuvant pembrolizumab,” said Dr. Carvajal.
“Although the efficacy of adjuvant ipilimumab [Yervoy] as well as adjuvant interferon have also been previously demonstrated, these agents are now appropriate for consideration in extremely rare clinical circumstances given the clinical efficacy and improved toxicity profile of single agent anti-PD-1 therapy.”
“The selection of the most appropriate adjuvant therapy should take into account the preferences of individual patients in terms of toxicity profile and drug administration considerations,” he added.
Study details
The COMBI-AD was a randomized, double-blind, placebo-controlled, phase 3 study conducted in 870 patients with high-risk, stage III, BRAF-V600E/K–mutant melanoma who were treatment naive. Participating patients had undergone surgical resection and had been disease free for ≤12 weeks.
Interim results from this study, reported in 2017, showed 1 year of oral adjuvant therapy with dabrafenib and trametinib provided a 53% lower risk for 3-year recurrence, compared with placebo.
Now, the investigators reported on the 5-year results for relapse-free survival and survival without distant metastasis. They noted that they were unable to analyze overall survival since the required number of events had not been reached.
Patients had been randomly assigned to receive 12 months of oral dabrafenib (at a dose of 150 mg twice daily) plus trametinib (2 mg once daily) or two matched placebos. Patients were followed for 60 months (5 years) for dabrafenib plus trametinib and 58 months for placebo.
At 5 years, the median relapse-free survival was not reached for patients who received the combination therapy group versus 16.6 months in the placebo group (hazard ratio for relapse or death, 0.51).
The percentage of patients who were alive without distant metastasis at 5 years was 65% in the dabrafenib plus trametinib group and 54% in the placebo arm (HR for distant metastasis or death, 0.55).
The hazard ratio for relapse-free survival favored dabrafenib plus trametinib across all patient subgroups that were evaluated in the study, and survival without distant metastasis showed a similar benefit for the combination regardless of disease stage.
Subsequent therapy was needed in 40% of patients who received dabrafenib plus trametinib and by 54% of those in the placebo group, with the most common treatments being immunotherapy in the combination-therapy group [26%] and small molecule–targeted therapy in the placebo group (35%).
A viable option
Dr. Dummer noted that, when this clinical trial was designed, all patients had to undergo aggressive surgery that involved lymph node dissection. “Nowadays, based on the lack of improvement on progression-free survival and overall survival, the surgical procedures are less aggressive and today we do not recommend aggressive lymph node dissection in patients that qualify for adjuvant therapy. In patients that do not have the BRAF mutation, there is the possibility of giving immunotherapy.”
He added that there is an urgent need for biomarkers that can identify early progression during adjuvant therapy. “Potentially, these patients would profit from immunotherapy alone or from combination using targeted therapy and immunotherapy,” Dr. Dummer said.
The study was funded by GlaxoSmithKline and Novartis. Dr. Dummer has declared multiple relationships with industry.
A version of this article originally appeared on Medscape.com.
Statins linked to reduced mortality in COVID-19
Treatment with statins was associated with a reduced risk of a severe or fatal course of COVID-19 by 30%, a meta-analysis of four published studies has shown.
In the analysis that included almost 9,000 COVID-19 patients, there was a significantly reduced risk for fatal or severe COVID-19 among patients who were users of statins, compared with nonusers (pooled hazard ratio, 0.70; 95% confidence interval, 0.53-0.94).
Based on the findings, “it may be time we shift our focus to statins as the potential therapeutic options in COVID-19 patients,” authors Syed Shahzad Hasan, PhD, University of Huddersfield (England), and Chia Siang Kow, MPharm, International Medical University, Kuala Lumpur, Malaysia, said in an interview.
The study was published online August 11 in The American Journal of Cardiology.
Moderate- to good-quality data
The analysis included four studies published up to July 27 of this year. Eligible studies included those with a cohort or case-control designs, enrolled patients with confirmed COVID-19, and had data available allowing comparison of the risk of severe illness and/or mortality among statin users versus nonusers in adjusted analyses, the authors noted.
The four studies – one of “moderate” quality and three of “good” quality – included a total of 8,990 COVID-19 patients.
In the pooled analysis, there was a significantly reduced risk for fatal or severe COVID-19 with use of statins, compared with non-use of statins (pooled HR, 0.70; 95% CI, 0.53-0.94).
Their findings also “discredited the suggestion of harms with the use of statins in COVID-19 patients,” the authors concluded.
“Since our meta-analysis included a fairly large total number of COVID-19 patients from four studies in which three are large-scale studies that adjusted extensively for multiple potential confounding factors, the findings can be considered reliable,” Dr. Hasan and Mr. Kow wrote in their article.
Based on the results, “moderate- to high-intensity statin therapy is likely to be beneficial” in patients with COVID-19, they said.
However, they cautioned that more data from prospective studies are needed to substantiate the findings and to determine the appropriate regimen for a statin in COVID-19 patients.
Yibin Wang, PhD, of the University of California, Los Angeles, said that “this is a very simple meta-analysis from four published studies which consistently reported a protective or neutral effect of statin usage on mortality or severe complications in COVID-19 patients.”
Although the scope of this meta-analysis was “quite limited, the conclusion was not unexpected, as most of the clinical analysis so far reported supports the benefits or safety of statin usage in COVID-19 patients,” Dr. Wang said in an interview.
Nonetheless, questions remain
While there is “almost no dispute” about the safety of continuing statin therapy in COVID-19 patients, it remains to be determined if statin therapy can be implemented as an adjuvant or independent therapy and a part of the standard care for COVID-19 patients regardless of their hyperlipidemia status, said Dr. Wang, who was not associated with Dr. Hasan’s and Mr. Kow’s research.
“While statin usage is associated with several beneficial effects such as anti-inflammation and cytoprotection, these effects are usually observed from long-term usage rather than short-term/acute administration. Therefore, prospective studies and randomized trials should be conducted to test the efficacy of stain usage for COVID-19 patients with mild to severe symptoms,” he noted.
“Considering the excellent record of statins as a safe and cheap drug, it is certainly a worthwhile effort to consider its broad-based usage for COVID-19 in order to lower the overall death and severe complications,” Dr. Wang concluded.
Guillermo Rodriguez-Nava, MD, department of internal medicine, AMITA Health Saint Francis Hospital, Evanston, Ill., is first author on one of the studies included in this meta-analysis.
The retrospective, single-center study found slower progression to death associated with atorvastatin in older patients with COVID-19 admitted to the ICU.
“Currently, there are hundreds of clinical trials evaluating a wide variety of pharmacological therapies for COVID-19. Unfortunately, these trials take time, and we are getting results in dribs and drabs,” Dr. Rodriguez-Nava said in an interview.
“In the meantime, the best available evidence is observational, and COVID-19 treatment regiments will continue to evolve. Whether atorvastatin is effective against COVID-19 is still under investigation. Nevertheless, clinicians should consider at least continuing them in patients with COVID-19,” he advised.
The study had no specific funding. Dr. Hasan, Mr. Kow, Dr. Wang, and Dr. Rodriguez-Nava disclosed no relationships relevant to this research.
A version of this article originally appeared on Medscape.com.
Treatment with statins was associated with a reduced risk of a severe or fatal course of COVID-19 by 30%, a meta-analysis of four published studies has shown.
In the analysis that included almost 9,000 COVID-19 patients, there was a significantly reduced risk for fatal or severe COVID-19 among patients who were users of statins, compared with nonusers (pooled hazard ratio, 0.70; 95% confidence interval, 0.53-0.94).
Based on the findings, “it may be time we shift our focus to statins as the potential therapeutic options in COVID-19 patients,” authors Syed Shahzad Hasan, PhD, University of Huddersfield (England), and Chia Siang Kow, MPharm, International Medical University, Kuala Lumpur, Malaysia, said in an interview.
The study was published online August 11 in The American Journal of Cardiology.
Moderate- to good-quality data
The analysis included four studies published up to July 27 of this year. Eligible studies included those with a cohort or case-control designs, enrolled patients with confirmed COVID-19, and had data available allowing comparison of the risk of severe illness and/or mortality among statin users versus nonusers in adjusted analyses, the authors noted.
The four studies – one of “moderate” quality and three of “good” quality – included a total of 8,990 COVID-19 patients.
In the pooled analysis, there was a significantly reduced risk for fatal or severe COVID-19 with use of statins, compared with non-use of statins (pooled HR, 0.70; 95% CI, 0.53-0.94).
Their findings also “discredited the suggestion of harms with the use of statins in COVID-19 patients,” the authors concluded.
“Since our meta-analysis included a fairly large total number of COVID-19 patients from four studies in which three are large-scale studies that adjusted extensively for multiple potential confounding factors, the findings can be considered reliable,” Dr. Hasan and Mr. Kow wrote in their article.
Based on the results, “moderate- to high-intensity statin therapy is likely to be beneficial” in patients with COVID-19, they said.
However, they cautioned that more data from prospective studies are needed to substantiate the findings and to determine the appropriate regimen for a statin in COVID-19 patients.
Yibin Wang, PhD, of the University of California, Los Angeles, said that “this is a very simple meta-analysis from four published studies which consistently reported a protective or neutral effect of statin usage on mortality or severe complications in COVID-19 patients.”
Although the scope of this meta-analysis was “quite limited, the conclusion was not unexpected, as most of the clinical analysis so far reported supports the benefits or safety of statin usage in COVID-19 patients,” Dr. Wang said in an interview.
Nonetheless, questions remain
While there is “almost no dispute” about the safety of continuing statin therapy in COVID-19 patients, it remains to be determined if statin therapy can be implemented as an adjuvant or independent therapy and a part of the standard care for COVID-19 patients regardless of their hyperlipidemia status, said Dr. Wang, who was not associated with Dr. Hasan’s and Mr. Kow’s research.
“While statin usage is associated with several beneficial effects such as anti-inflammation and cytoprotection, these effects are usually observed from long-term usage rather than short-term/acute administration. Therefore, prospective studies and randomized trials should be conducted to test the efficacy of stain usage for COVID-19 patients with mild to severe symptoms,” he noted.
“Considering the excellent record of statins as a safe and cheap drug, it is certainly a worthwhile effort to consider its broad-based usage for COVID-19 in order to lower the overall death and severe complications,” Dr. Wang concluded.
Guillermo Rodriguez-Nava, MD, department of internal medicine, AMITA Health Saint Francis Hospital, Evanston, Ill., is first author on one of the studies included in this meta-analysis.
The retrospective, single-center study found slower progression to death associated with atorvastatin in older patients with COVID-19 admitted to the ICU.
“Currently, there are hundreds of clinical trials evaluating a wide variety of pharmacological therapies for COVID-19. Unfortunately, these trials take time, and we are getting results in dribs and drabs,” Dr. Rodriguez-Nava said in an interview.
“In the meantime, the best available evidence is observational, and COVID-19 treatment regiments will continue to evolve. Whether atorvastatin is effective against COVID-19 is still under investigation. Nevertheless, clinicians should consider at least continuing them in patients with COVID-19,” he advised.
The study had no specific funding. Dr. Hasan, Mr. Kow, Dr. Wang, and Dr. Rodriguez-Nava disclosed no relationships relevant to this research.
A version of this article originally appeared on Medscape.com.
Treatment with statins was associated with a reduced risk of a severe or fatal course of COVID-19 by 30%, a meta-analysis of four published studies has shown.
In the analysis that included almost 9,000 COVID-19 patients, there was a significantly reduced risk for fatal or severe COVID-19 among patients who were users of statins, compared with nonusers (pooled hazard ratio, 0.70; 95% confidence interval, 0.53-0.94).
Based on the findings, “it may be time we shift our focus to statins as the potential therapeutic options in COVID-19 patients,” authors Syed Shahzad Hasan, PhD, University of Huddersfield (England), and Chia Siang Kow, MPharm, International Medical University, Kuala Lumpur, Malaysia, said in an interview.
The study was published online August 11 in The American Journal of Cardiology.
Moderate- to good-quality data
The analysis included four studies published up to July 27 of this year. Eligible studies included those with a cohort or case-control designs, enrolled patients with confirmed COVID-19, and had data available allowing comparison of the risk of severe illness and/or mortality among statin users versus nonusers in adjusted analyses, the authors noted.
The four studies – one of “moderate” quality and three of “good” quality – included a total of 8,990 COVID-19 patients.
In the pooled analysis, there was a significantly reduced risk for fatal or severe COVID-19 with use of statins, compared with non-use of statins (pooled HR, 0.70; 95% CI, 0.53-0.94).
Their findings also “discredited the suggestion of harms with the use of statins in COVID-19 patients,” the authors concluded.
“Since our meta-analysis included a fairly large total number of COVID-19 patients from four studies in which three are large-scale studies that adjusted extensively for multiple potential confounding factors, the findings can be considered reliable,” Dr. Hasan and Mr. Kow wrote in their article.
Based on the results, “moderate- to high-intensity statin therapy is likely to be beneficial” in patients with COVID-19, they said.
However, they cautioned that more data from prospective studies are needed to substantiate the findings and to determine the appropriate regimen for a statin in COVID-19 patients.
Yibin Wang, PhD, of the University of California, Los Angeles, said that “this is a very simple meta-analysis from four published studies which consistently reported a protective or neutral effect of statin usage on mortality or severe complications in COVID-19 patients.”
Although the scope of this meta-analysis was “quite limited, the conclusion was not unexpected, as most of the clinical analysis so far reported supports the benefits or safety of statin usage in COVID-19 patients,” Dr. Wang said in an interview.
Nonetheless, questions remain
While there is “almost no dispute” about the safety of continuing statin therapy in COVID-19 patients, it remains to be determined if statin therapy can be implemented as an adjuvant or independent therapy and a part of the standard care for COVID-19 patients regardless of their hyperlipidemia status, said Dr. Wang, who was not associated with Dr. Hasan’s and Mr. Kow’s research.
“While statin usage is associated with several beneficial effects such as anti-inflammation and cytoprotection, these effects are usually observed from long-term usage rather than short-term/acute administration. Therefore, prospective studies and randomized trials should be conducted to test the efficacy of stain usage for COVID-19 patients with mild to severe symptoms,” he noted.
“Considering the excellent record of statins as a safe and cheap drug, it is certainly a worthwhile effort to consider its broad-based usage for COVID-19 in order to lower the overall death and severe complications,” Dr. Wang concluded.
Guillermo Rodriguez-Nava, MD, department of internal medicine, AMITA Health Saint Francis Hospital, Evanston, Ill., is first author on one of the studies included in this meta-analysis.
The retrospective, single-center study found slower progression to death associated with atorvastatin in older patients with COVID-19 admitted to the ICU.
“Currently, there are hundreds of clinical trials evaluating a wide variety of pharmacological therapies for COVID-19. Unfortunately, these trials take time, and we are getting results in dribs and drabs,” Dr. Rodriguez-Nava said in an interview.
“In the meantime, the best available evidence is observational, and COVID-19 treatment regiments will continue to evolve. Whether atorvastatin is effective against COVID-19 is still under investigation. Nevertheless, clinicians should consider at least continuing them in patients with COVID-19,” he advised.
The study had no specific funding. Dr. Hasan, Mr. Kow, Dr. Wang, and Dr. Rodriguez-Nava disclosed no relationships relevant to this research.
A version of this article originally appeared on Medscape.com.
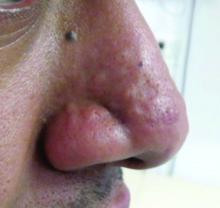
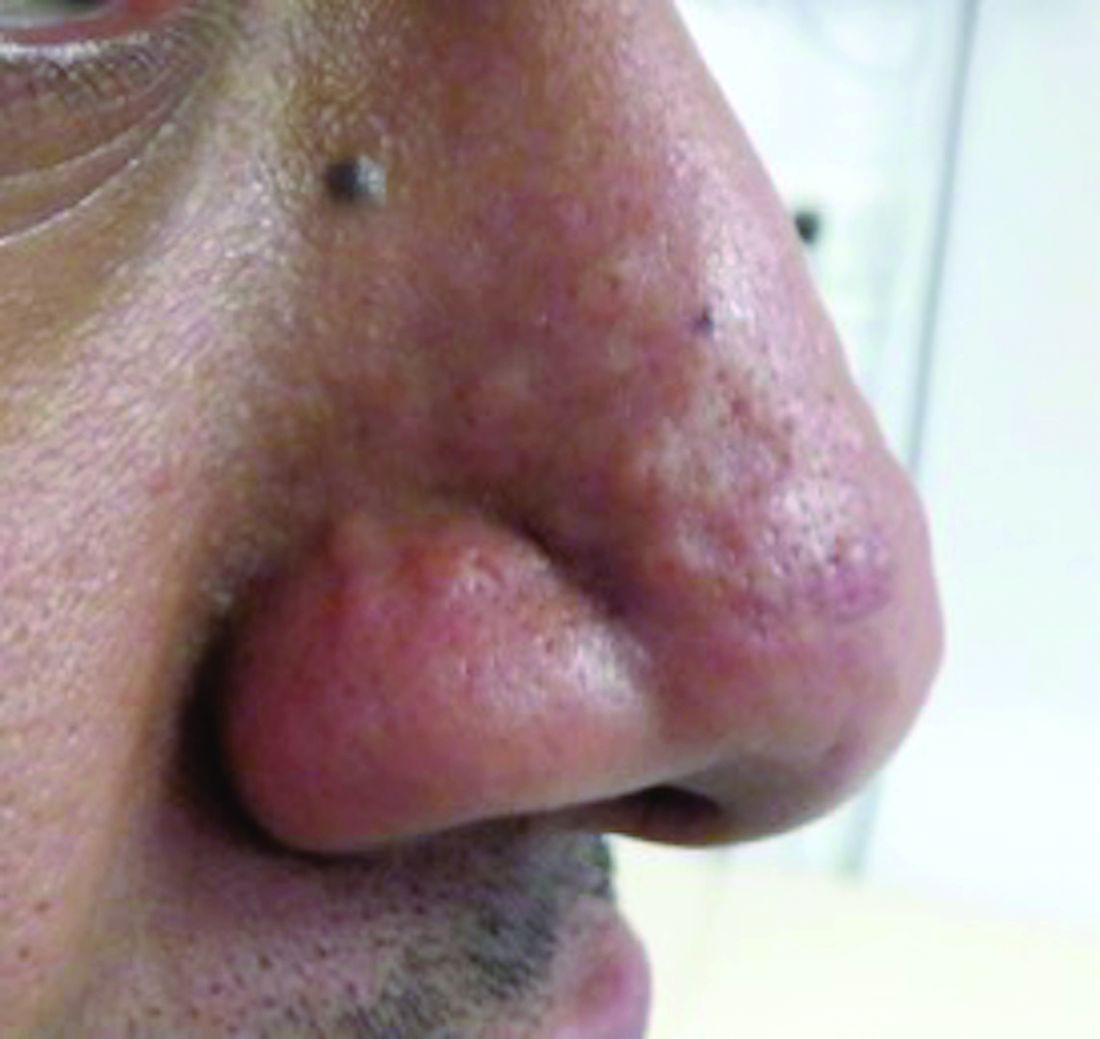
Post-acne nasal papules described in a series of patients
, but researchers believe the condition could be an underrecognized problem, affecting patients with skin of color in particular, according to the authors of a case series published in Pediatric Dermatology.
Jorge Roman, MD, and coauthors in the department of dermatology at New York (N.Y.) University identified 20 patients with a history of acne who had nasal papules, in a retrospective review of electronic medical records at NYU over 1 year (April 2018 to April 2019). The presentation ranged from “a few, small skin-colored papules to large, dome-shaped papulonodules, to more extensive rhinophymatous-like” changes with some patients having papular lesions on the chin in addition to the nose, they wrote in the report.
These papules greatly resembled angiofibromas, but appear to be a sequela of acne, according to the authors. In five patients who had biopsies, the results showed “a dome-shaped proliferation of spindle and stellate-shaped cells with thickened collagen bundles and dilated thin-walled blood vessels,” the authors wrote. “The histopathological findings of these nasal papules were indistinguishable from those of a conventional angiofibroma.”
In addition, the patients did not have evidence of underlying genetic conditions that could explain the angiofibroma-like lesions. “Although acne has not previously been implicated in the development of angiofibromas, based on the data available for our patients, it seems extremely unlikely that the lesions would be related to anything else,” Dr. Roman, a dermatology resident at New York University, said in an interview.
He said he first recognized the nasal papules in clinic as a first-year resident, but was surprised to find a lack of information on the condition. “Dermatology has a name for just about every skin disease imaginable, so I found it very odd when I couldn’t find much describing this condition,” he said. “There was a large disparity between what we were seeing in clinic and what was reported in the literature.”
Nearly all the patients were Hispanic (17 of 20) and adolescent males (17 patients), with a median age of 16 years at the time of presentation. There were two Black patients and one Asian patient. Race and ethnicity were not mentioned in two previous reports describing papular acne scarring, but Dr. Roman and colleagues noted that in their clinic, the condition appeared to affect adolescent patients with skin of color predominantly.
Reasons why nasal papules may be underreported are unclear, Dr. Roman noted. One possible explanation is lower use of dermatologic care among patients with skin of color. “Interestingly, previous research has shown that racial minorities are lower utilizers of dermatologic care. It is possible that the patient demographic most afflicted by this condition face significant barriers when seeking care,” he said.
Due to a low level of awareness of acne-related nasal papules, “clinicians may not recognize it as an acne-related scarring process. This is significant, as early recognition and treatment can prevent the development or progression of these potentially disfiguring sequelae,” Dr. Roman said.
Although the results are from a small case series at a single center, Dr. Roman said this condition may be more prevalent than realized. “Having been raised in a predominately Latino community in Texas, I can easily recall seeing people with these papules growing up. I don’t think it would be surprising for dermatologists reading our paper to say, ‘I’ve seen this in clinic before,’ ” he said.
Regarding treatment, there is an ongoing investigation into what treatments are effective for the acne-related nasal papules. “Physical treatment modalities such as ablative laser or surgical removal seem to be the most efficacious,” Dr. Roman said. “In the future, a prospective clinical study will help to better define the prevalence and risk factors for the condition,” he said.
He and coauthors reported no conflicts of interest. No funding source was listed.
SOURCE: Roman J et al. Pediatr Dermatol. 2020 Aug 7. doi: 10.1111/pde.14319.
, but researchers believe the condition could be an underrecognized problem, affecting patients with skin of color in particular, according to the authors of a case series published in Pediatric Dermatology.
Jorge Roman, MD, and coauthors in the department of dermatology at New York (N.Y.) University identified 20 patients with a history of acne who had nasal papules, in a retrospective review of electronic medical records at NYU over 1 year (April 2018 to April 2019). The presentation ranged from “a few, small skin-colored papules to large, dome-shaped papulonodules, to more extensive rhinophymatous-like” changes with some patients having papular lesions on the chin in addition to the nose, they wrote in the report.
These papules greatly resembled angiofibromas, but appear to be a sequela of acne, according to the authors. In five patients who had biopsies, the results showed “a dome-shaped proliferation of spindle and stellate-shaped cells with thickened collagen bundles and dilated thin-walled blood vessels,” the authors wrote. “The histopathological findings of these nasal papules were indistinguishable from those of a conventional angiofibroma.”
In addition, the patients did not have evidence of underlying genetic conditions that could explain the angiofibroma-like lesions. “Although acne has not previously been implicated in the development of angiofibromas, based on the data available for our patients, it seems extremely unlikely that the lesions would be related to anything else,” Dr. Roman, a dermatology resident at New York University, said in an interview.
He said he first recognized the nasal papules in clinic as a first-year resident, but was surprised to find a lack of information on the condition. “Dermatology has a name for just about every skin disease imaginable, so I found it very odd when I couldn’t find much describing this condition,” he said. “There was a large disparity between what we were seeing in clinic and what was reported in the literature.”
Nearly all the patients were Hispanic (17 of 20) and adolescent males (17 patients), with a median age of 16 years at the time of presentation. There were two Black patients and one Asian patient. Race and ethnicity were not mentioned in two previous reports describing papular acne scarring, but Dr. Roman and colleagues noted that in their clinic, the condition appeared to affect adolescent patients with skin of color predominantly.
Reasons why nasal papules may be underreported are unclear, Dr. Roman noted. One possible explanation is lower use of dermatologic care among patients with skin of color. “Interestingly, previous research has shown that racial minorities are lower utilizers of dermatologic care. It is possible that the patient demographic most afflicted by this condition face significant barriers when seeking care,” he said.
Due to a low level of awareness of acne-related nasal papules, “clinicians may not recognize it as an acne-related scarring process. This is significant, as early recognition and treatment can prevent the development or progression of these potentially disfiguring sequelae,” Dr. Roman said.
Although the results are from a small case series at a single center, Dr. Roman said this condition may be more prevalent than realized. “Having been raised in a predominately Latino community in Texas, I can easily recall seeing people with these papules growing up. I don’t think it would be surprising for dermatologists reading our paper to say, ‘I’ve seen this in clinic before,’ ” he said.
Regarding treatment, there is an ongoing investigation into what treatments are effective for the acne-related nasal papules. “Physical treatment modalities such as ablative laser or surgical removal seem to be the most efficacious,” Dr. Roman said. “In the future, a prospective clinical study will help to better define the prevalence and risk factors for the condition,” he said.
He and coauthors reported no conflicts of interest. No funding source was listed.
SOURCE: Roman J et al. Pediatr Dermatol. 2020 Aug 7. doi: 10.1111/pde.14319.
, but researchers believe the condition could be an underrecognized problem, affecting patients with skin of color in particular, according to the authors of a case series published in Pediatric Dermatology.
Jorge Roman, MD, and coauthors in the department of dermatology at New York (N.Y.) University identified 20 patients with a history of acne who had nasal papules, in a retrospective review of electronic medical records at NYU over 1 year (April 2018 to April 2019). The presentation ranged from “a few, small skin-colored papules to large, dome-shaped papulonodules, to more extensive rhinophymatous-like” changes with some patients having papular lesions on the chin in addition to the nose, they wrote in the report.
These papules greatly resembled angiofibromas, but appear to be a sequela of acne, according to the authors. In five patients who had biopsies, the results showed “a dome-shaped proliferation of spindle and stellate-shaped cells with thickened collagen bundles and dilated thin-walled blood vessels,” the authors wrote. “The histopathological findings of these nasal papules were indistinguishable from those of a conventional angiofibroma.”
In addition, the patients did not have evidence of underlying genetic conditions that could explain the angiofibroma-like lesions. “Although acne has not previously been implicated in the development of angiofibromas, based on the data available for our patients, it seems extremely unlikely that the lesions would be related to anything else,” Dr. Roman, a dermatology resident at New York University, said in an interview.
He said he first recognized the nasal papules in clinic as a first-year resident, but was surprised to find a lack of information on the condition. “Dermatology has a name for just about every skin disease imaginable, so I found it very odd when I couldn’t find much describing this condition,” he said. “There was a large disparity between what we were seeing in clinic and what was reported in the literature.”
Nearly all the patients were Hispanic (17 of 20) and adolescent males (17 patients), with a median age of 16 years at the time of presentation. There were two Black patients and one Asian patient. Race and ethnicity were not mentioned in two previous reports describing papular acne scarring, but Dr. Roman and colleagues noted that in their clinic, the condition appeared to affect adolescent patients with skin of color predominantly.
Reasons why nasal papules may be underreported are unclear, Dr. Roman noted. One possible explanation is lower use of dermatologic care among patients with skin of color. “Interestingly, previous research has shown that racial minorities are lower utilizers of dermatologic care. It is possible that the patient demographic most afflicted by this condition face significant barriers when seeking care,” he said.
Due to a low level of awareness of acne-related nasal papules, “clinicians may not recognize it as an acne-related scarring process. This is significant, as early recognition and treatment can prevent the development or progression of these potentially disfiguring sequelae,” Dr. Roman said.
Although the results are from a small case series at a single center, Dr. Roman said this condition may be more prevalent than realized. “Having been raised in a predominately Latino community in Texas, I can easily recall seeing people with these papules growing up. I don’t think it would be surprising for dermatologists reading our paper to say, ‘I’ve seen this in clinic before,’ ” he said.
Regarding treatment, there is an ongoing investigation into what treatments are effective for the acne-related nasal papules. “Physical treatment modalities such as ablative laser or surgical removal seem to be the most efficacious,” Dr. Roman said. “In the future, a prospective clinical study will help to better define the prevalence and risk factors for the condition,” he said.
He and coauthors reported no conflicts of interest. No funding source was listed.
SOURCE: Roman J et al. Pediatr Dermatol. 2020 Aug 7. doi: 10.1111/pde.14319.
FROM PEDIATRIC DERMATOLOGY
Who’s better off: Employed or self-employed physicians?
Self-employed physicians have the highest salaries, largest homes, and greatest wealth – yet they feel the least fairly compensated, according to an analysis of data from over 17,000 physicians.
A new examination of survey responses from the Medscape Physician Compensation Report 2020, which included information about income, job satisfaction, and more, compared responses from self-employed physicians, independent contractors, and employed physicians.
Income and wealth, benefits, and job satisfaction were compared. From the results of the questionnaire, self-employed physicians stand out among their peers across all categories: They enjoy greater income, wealth, and benefits and appear to be more satisfied by their choice of practice.
“The survey confirms that self-employed is the most satisfying, although the trend in health care is to take employed positions,” said Robert Scroggins, JD, CPA, certified health care business consultant with ScrogginsGreer, Cincinnati. “Doctors who become employees primarily do that to escape the management responsibilities for the practice. It seems to be more a decision to get away from something than to go toward something.”
The financial and work picture for self-employed physicians
Self-employed physicians reported the largest salaries for 2019 (average, $360,752), followed by independent contractors ($336,005). Employees reported the lowest average salary ($297,332).
The largest percentage of self-employed physicians (46%) work in an office-based group practice, followed by those in office-based solo practices (30%). Almost two-thirds of self-employed respondents are owners and 37% are partners.
Self-employed physicians are more likely to be older than 45 years; 79% fall into that age bracket, compared with 57% of employees and 70% of independent contractors.
Self-employed physicians reported the highest levels of wealth among their peers. About 44% of self-employed respondents declared a net wealth of over $2 million, compared with 25% of employees. Only 6% of contractors and employed physicians reported a net wealth of over $5 million, compared with 13% of self-employed physicians.
Self-employed physicians also managed their personal expenses slightly differently. They were more likely to pool their income with their spouse in a common account used for bills and expenses, regardless of how much they each earned (63% of self-employed respondents, compared with 58% of employees and 50% of independent contractors).
Perhaps unsurprisingly, self-employed physicians also reported having the largest homes, with an average square footage of 3,629 square feet, compared with 3,023 square feet for employees and 2,984 square feet for independent contractors. Self-employed physicians’ mortgages (average, $240,389) were similar to those of employed physicians’ mortgages but were higher than those of independent contractors’ mortgages (average, $213,740).
Self-employed physicians were also most likely to highly appraise their own performance: Half of all self-employed respondents felt “very satisfied” with their job performance, compared with 40% of employees and 44% of independent contractors.
When asked what they consider to be the most rewarding aspect of their job, self-employed physicians were more likely to choose gratitude and patient relationships than their peers (32%, compared with 26% of employees and 19% of independent contractors).
Despite their higher net wealth and larger salaries, self-employed physicians were least likely to feel fairly compensated; 49% of self-employed physicians said they did not feel fairly compensated for their work, compared with 40% of employees and 40% of independent contractors.
“Self-employed physicians may be better compensated than others of the same specialty who are employees, so some of that may be perception,” said Mr. Scroggins. “Or they feel they should be compensated to a far greater degree than those who are employed.”
Self-employed physicians were also more likely to respond that they would choose the same practice setting again, though across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
The financial and work picture for employed physicians
About a third (32%) of employed physician respondents work in hospitals; 28% work in private practices.
Employed physicians were most likely to report a salary increase from 2018 to 2019: 74%, compared with 45% of self-employed and 52% of independent contractors.
As for declines in income, self-employed physicians and independent contractors suffered a comparable loss, with 13% and 12% of them, respectively, reporting salary cuts greater than 10%. Decreases of up to 10% were felt mostly by the self-employed, with 17% experiencing such cuts, compared with 7% of employees and 10% of independent contractors.
In contrast, employees were the least likely of the three categories to have incurred large financial losses over the past year: 77% of employed respondents indicated that they had not experienced any significant financial losses in the past year, compared with 63% of self-employed physicians and 63% of independent contractors. They were also least likely to have made any investments at all over the past year – 21% of employees reported having made none at all in 2019, compared to 11% of self-employed physicians and 16% of independent contractors.
The financial and work picture for independent contractors
Just over half (52%) of all independent contractors who responded to our questionnaire work in hospitals, 15% work in group practices, 9% work in outpatient clinics, and just 2% work in solo practices.
Independent contractors were less likely than their peers to have received employment benefits such as health insurance, malpractice coverage, and paid time off. They were also less likely to be saving for retirement. Almost half (45%) of independent contractors said they received no employment benefits at all, compared to 20% of self-employed physicians and just 8% of employees.
What’s more, 27% of independent contractors do not currently put money into a 401(k) retirement account or tax-deferred college savings account on a regular basis, compared with 16% of self-employed physicians and 8% of employees. Similarly, they were less likely to put money into a taxable savings account (39% responded that they do not, compared with 32% of self-employed physicians and 27% of employees).
“Net worth and retirement funding findings do line up with what I’ve observed,” said Mr. Scroggins. “Those who have independent practices as opposed to working for a hospital do tend to more heavily fund retirement plan accounts, which is typically the biggest driver of building net worth.”
Despite the lack of retirement planning, independent contractors were more likely than their peers to derive satisfaction from making money at a job they like (18%, compared with 12% of employees and 11% of self-employed physicians). They’re also far more likely to be in emergency medicine (22% of independent contractors, compared with 3% of self-employed and 5% of employees) or psychiatry (11% of independent contractors, compared with 5% of self-employed and 6% of employees).
Among the three categories of physicians, independent contractors were least likely to say that they would choose the same practice setting again. Across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
Physicians who are considering leaving their own practice for a hospital setting should do so with caution and fully understand what they are getting into, said Mr. Scroggins. “If they’re just looking at compensation, they also should be looking very carefully at retirement plan benefits. If that’s their main method of saving and building net worth, then that’s a dramatic difference.”
And of course, there’s always the intangible value of feeling connected to a practice and its patients: “Physicians got into this line of work to treat patients and help people become healthier, and in hospitals they end up being more disconnected from their patients,” Mr. Scroggins said. “That’s a big factor as well.”
Editor’s note: Only differences that are statistically significant at a 95% confidence level between categories of employment have been included. Of the 13,893 responses included in this analysis, 3,860 physicians identified as self-employed, 9,262 as employees, and 772 as independent contractors.
A version of this article originally appeared on Medscape.com.
Self-employed physicians have the highest salaries, largest homes, and greatest wealth – yet they feel the least fairly compensated, according to an analysis of data from over 17,000 physicians.
A new examination of survey responses from the Medscape Physician Compensation Report 2020, which included information about income, job satisfaction, and more, compared responses from self-employed physicians, independent contractors, and employed physicians.
Income and wealth, benefits, and job satisfaction were compared. From the results of the questionnaire, self-employed physicians stand out among their peers across all categories: They enjoy greater income, wealth, and benefits and appear to be more satisfied by their choice of practice.
“The survey confirms that self-employed is the most satisfying, although the trend in health care is to take employed positions,” said Robert Scroggins, JD, CPA, certified health care business consultant with ScrogginsGreer, Cincinnati. “Doctors who become employees primarily do that to escape the management responsibilities for the practice. It seems to be more a decision to get away from something than to go toward something.”
The financial and work picture for self-employed physicians
Self-employed physicians reported the largest salaries for 2019 (average, $360,752), followed by independent contractors ($336,005). Employees reported the lowest average salary ($297,332).
The largest percentage of self-employed physicians (46%) work in an office-based group practice, followed by those in office-based solo practices (30%). Almost two-thirds of self-employed respondents are owners and 37% are partners.
Self-employed physicians are more likely to be older than 45 years; 79% fall into that age bracket, compared with 57% of employees and 70% of independent contractors.
Self-employed physicians reported the highest levels of wealth among their peers. About 44% of self-employed respondents declared a net wealth of over $2 million, compared with 25% of employees. Only 6% of contractors and employed physicians reported a net wealth of over $5 million, compared with 13% of self-employed physicians.
Self-employed physicians also managed their personal expenses slightly differently. They were more likely to pool their income with their spouse in a common account used for bills and expenses, regardless of how much they each earned (63% of self-employed respondents, compared with 58% of employees and 50% of independent contractors).
Perhaps unsurprisingly, self-employed physicians also reported having the largest homes, with an average square footage of 3,629 square feet, compared with 3,023 square feet for employees and 2,984 square feet for independent contractors. Self-employed physicians’ mortgages (average, $240,389) were similar to those of employed physicians’ mortgages but were higher than those of independent contractors’ mortgages (average, $213,740).
Self-employed physicians were also most likely to highly appraise their own performance: Half of all self-employed respondents felt “very satisfied” with their job performance, compared with 40% of employees and 44% of independent contractors.
When asked what they consider to be the most rewarding aspect of their job, self-employed physicians were more likely to choose gratitude and patient relationships than their peers (32%, compared with 26% of employees and 19% of independent contractors).
Despite their higher net wealth and larger salaries, self-employed physicians were least likely to feel fairly compensated; 49% of self-employed physicians said they did not feel fairly compensated for their work, compared with 40% of employees and 40% of independent contractors.
“Self-employed physicians may be better compensated than others of the same specialty who are employees, so some of that may be perception,” said Mr. Scroggins. “Or they feel they should be compensated to a far greater degree than those who are employed.”
Self-employed physicians were also more likely to respond that they would choose the same practice setting again, though across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
The financial and work picture for employed physicians
About a third (32%) of employed physician respondents work in hospitals; 28% work in private practices.
Employed physicians were most likely to report a salary increase from 2018 to 2019: 74%, compared with 45% of self-employed and 52% of independent contractors.
As for declines in income, self-employed physicians and independent contractors suffered a comparable loss, with 13% and 12% of them, respectively, reporting salary cuts greater than 10%. Decreases of up to 10% were felt mostly by the self-employed, with 17% experiencing such cuts, compared with 7% of employees and 10% of independent contractors.
In contrast, employees were the least likely of the three categories to have incurred large financial losses over the past year: 77% of employed respondents indicated that they had not experienced any significant financial losses in the past year, compared with 63% of self-employed physicians and 63% of independent contractors. They were also least likely to have made any investments at all over the past year – 21% of employees reported having made none at all in 2019, compared to 11% of self-employed physicians and 16% of independent contractors.
The financial and work picture for independent contractors
Just over half (52%) of all independent contractors who responded to our questionnaire work in hospitals, 15% work in group practices, 9% work in outpatient clinics, and just 2% work in solo practices.
Independent contractors were less likely than their peers to have received employment benefits such as health insurance, malpractice coverage, and paid time off. They were also less likely to be saving for retirement. Almost half (45%) of independent contractors said they received no employment benefits at all, compared to 20% of self-employed physicians and just 8% of employees.
What’s more, 27% of independent contractors do not currently put money into a 401(k) retirement account or tax-deferred college savings account on a regular basis, compared with 16% of self-employed physicians and 8% of employees. Similarly, they were less likely to put money into a taxable savings account (39% responded that they do not, compared with 32% of self-employed physicians and 27% of employees).
“Net worth and retirement funding findings do line up with what I’ve observed,” said Mr. Scroggins. “Those who have independent practices as opposed to working for a hospital do tend to more heavily fund retirement plan accounts, which is typically the biggest driver of building net worth.”
Despite the lack of retirement planning, independent contractors were more likely than their peers to derive satisfaction from making money at a job they like (18%, compared with 12% of employees and 11% of self-employed physicians). They’re also far more likely to be in emergency medicine (22% of independent contractors, compared with 3% of self-employed and 5% of employees) or psychiatry (11% of independent contractors, compared with 5% of self-employed and 6% of employees).
Among the three categories of physicians, independent contractors were least likely to say that they would choose the same practice setting again. Across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
Physicians who are considering leaving their own practice for a hospital setting should do so with caution and fully understand what they are getting into, said Mr. Scroggins. “If they’re just looking at compensation, they also should be looking very carefully at retirement plan benefits. If that’s their main method of saving and building net worth, then that’s a dramatic difference.”
And of course, there’s always the intangible value of feeling connected to a practice and its patients: “Physicians got into this line of work to treat patients and help people become healthier, and in hospitals they end up being more disconnected from their patients,” Mr. Scroggins said. “That’s a big factor as well.”
Editor’s note: Only differences that are statistically significant at a 95% confidence level between categories of employment have been included. Of the 13,893 responses included in this analysis, 3,860 physicians identified as self-employed, 9,262 as employees, and 772 as independent contractors.
A version of this article originally appeared on Medscape.com.
Self-employed physicians have the highest salaries, largest homes, and greatest wealth – yet they feel the least fairly compensated, according to an analysis of data from over 17,000 physicians.
A new examination of survey responses from the Medscape Physician Compensation Report 2020, which included information about income, job satisfaction, and more, compared responses from self-employed physicians, independent contractors, and employed physicians.
Income and wealth, benefits, and job satisfaction were compared. From the results of the questionnaire, self-employed physicians stand out among their peers across all categories: They enjoy greater income, wealth, and benefits and appear to be more satisfied by their choice of practice.
“The survey confirms that self-employed is the most satisfying, although the trend in health care is to take employed positions,” said Robert Scroggins, JD, CPA, certified health care business consultant with ScrogginsGreer, Cincinnati. “Doctors who become employees primarily do that to escape the management responsibilities for the practice. It seems to be more a decision to get away from something than to go toward something.”
The financial and work picture for self-employed physicians
Self-employed physicians reported the largest salaries for 2019 (average, $360,752), followed by independent contractors ($336,005). Employees reported the lowest average salary ($297,332).
The largest percentage of self-employed physicians (46%) work in an office-based group practice, followed by those in office-based solo practices (30%). Almost two-thirds of self-employed respondents are owners and 37% are partners.
Self-employed physicians are more likely to be older than 45 years; 79% fall into that age bracket, compared with 57% of employees and 70% of independent contractors.
Self-employed physicians reported the highest levels of wealth among their peers. About 44% of self-employed respondents declared a net wealth of over $2 million, compared with 25% of employees. Only 6% of contractors and employed physicians reported a net wealth of over $5 million, compared with 13% of self-employed physicians.
Self-employed physicians also managed their personal expenses slightly differently. They were more likely to pool their income with their spouse in a common account used for bills and expenses, regardless of how much they each earned (63% of self-employed respondents, compared with 58% of employees and 50% of independent contractors).
Perhaps unsurprisingly, self-employed physicians also reported having the largest homes, with an average square footage of 3,629 square feet, compared with 3,023 square feet for employees and 2,984 square feet for independent contractors. Self-employed physicians’ mortgages (average, $240,389) were similar to those of employed physicians’ mortgages but were higher than those of independent contractors’ mortgages (average, $213,740).
Self-employed physicians were also most likely to highly appraise their own performance: Half of all self-employed respondents felt “very satisfied” with their job performance, compared with 40% of employees and 44% of independent contractors.
When asked what they consider to be the most rewarding aspect of their job, self-employed physicians were more likely to choose gratitude and patient relationships than their peers (32%, compared with 26% of employees and 19% of independent contractors).
Despite their higher net wealth and larger salaries, self-employed physicians were least likely to feel fairly compensated; 49% of self-employed physicians said they did not feel fairly compensated for their work, compared with 40% of employees and 40% of independent contractors.
“Self-employed physicians may be better compensated than others of the same specialty who are employees, so some of that may be perception,” said Mr. Scroggins. “Or they feel they should be compensated to a far greater degree than those who are employed.”
Self-employed physicians were also more likely to respond that they would choose the same practice setting again, though across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
The financial and work picture for employed physicians
About a third (32%) of employed physician respondents work in hospitals; 28% work in private practices.
Employed physicians were most likely to report a salary increase from 2018 to 2019: 74%, compared with 45% of self-employed and 52% of independent contractors.
As for declines in income, self-employed physicians and independent contractors suffered a comparable loss, with 13% and 12% of them, respectively, reporting salary cuts greater than 10%. Decreases of up to 10% were felt mostly by the self-employed, with 17% experiencing such cuts, compared with 7% of employees and 10% of independent contractors.
In contrast, employees were the least likely of the three categories to have incurred large financial losses over the past year: 77% of employed respondents indicated that they had not experienced any significant financial losses in the past year, compared with 63% of self-employed physicians and 63% of independent contractors. They were also least likely to have made any investments at all over the past year – 21% of employees reported having made none at all in 2019, compared to 11% of self-employed physicians and 16% of independent contractors.
The financial and work picture for independent contractors
Just over half (52%) of all independent contractors who responded to our questionnaire work in hospitals, 15% work in group practices, 9% work in outpatient clinics, and just 2% work in solo practices.
Independent contractors were less likely than their peers to have received employment benefits such as health insurance, malpractice coverage, and paid time off. They were also less likely to be saving for retirement. Almost half (45%) of independent contractors said they received no employment benefits at all, compared to 20% of self-employed physicians and just 8% of employees.
What’s more, 27% of independent contractors do not currently put money into a 401(k) retirement account or tax-deferred college savings account on a regular basis, compared with 16% of self-employed physicians and 8% of employees. Similarly, they were less likely to put money into a taxable savings account (39% responded that they do not, compared with 32% of self-employed physicians and 27% of employees).
“Net worth and retirement funding findings do line up with what I’ve observed,” said Mr. Scroggins. “Those who have independent practices as opposed to working for a hospital do tend to more heavily fund retirement plan accounts, which is typically the biggest driver of building net worth.”
Despite the lack of retirement planning, independent contractors were more likely than their peers to derive satisfaction from making money at a job they like (18%, compared with 12% of employees and 11% of self-employed physicians). They’re also far more likely to be in emergency medicine (22% of independent contractors, compared with 3% of self-employed and 5% of employees) or psychiatry (11% of independent contractors, compared with 5% of self-employed and 6% of employees).
Among the three categories of physicians, independent contractors were least likely to say that they would choose the same practice setting again. Across all three categories, fewer than 50% of respondents would do so: 34% of self-employed physicians, compared with 29% of employees and 28% of independent contractors.
Physicians who are considering leaving their own practice for a hospital setting should do so with caution and fully understand what they are getting into, said Mr. Scroggins. “If they’re just looking at compensation, they also should be looking very carefully at retirement plan benefits. If that’s their main method of saving and building net worth, then that’s a dramatic difference.”
And of course, there’s always the intangible value of feeling connected to a practice and its patients: “Physicians got into this line of work to treat patients and help people become healthier, and in hospitals they end up being more disconnected from their patients,” Mr. Scroggins said. “That’s a big factor as well.”
Editor’s note: Only differences that are statistically significant at a 95% confidence level between categories of employment have been included. Of the 13,893 responses included in this analysis, 3,860 physicians identified as self-employed, 9,262 as employees, and 772 as independent contractors.
A version of this article originally appeared on Medscape.com.
High mortality rates reported in large COVID-19 study
Factors including older age and certain comorbidities have been linked to more serious COVID-19 outcomes in previous research, and now a large dataset collected from hundreds of hospitals nationwide provides more detailed data regarding risk for mechanical ventilation and death.
History of pulmonary disease or smoking, interestingly, were not.
One expert urges caution when interpreting the results, however. Although the study found a number of risk factors for ventilation and mortality, she says the dataset lacks information on race and disease severity, and the sample may not be nationally representative.
The investigators hope their level of granularity will further assist researchers searching for effective treatments and clinicians seeking to triage patients during the COVID-19 pandemic.
The study was published online August 28 in Clinical Infectious Diseases.
COVID-19 and comorbidities
“What I found most illuminating was this whole concept of comorbid conditions. This provides suggestive data about who we need to worry about most and who we may need to worry about less,” study author Robert S. Brown Jr, MD, MPH, told Medscape Medical News.
Comorbid conditions included hypertension in 47% of patients, diabetes in 28%, and cardiovascular disease in 19%. Another 16% were obese and 12% had chronic kidney disease. People with comorbid obesity, chronic kidney disease, and cardiovascular disease were more likely to receive mechanical ventilation compared to those without a history of these conditions in an adjusted, multivariable logistic analysis.
With the exception of obesity, the same factors were associated with risk for death during hospitalization.
In contrast, hypertension, history of smoking, and history of pulmonary disease were associated with a lower risk of needing mechanical ventilation and/or lower risk for mortality.
Furthermore, people with liver disease, gastrointestinal diseases, and even autoimmune diseases – which are likely associated with immunosuppression – “are not at that much of an increased risk that we noticed it in our data,” Brown said.
“As I tell many of my patients who have mild liver disease, for example, I would rather have mild liver disease and be on immunosuppressant therapy than be an older, obese male,” he added.
Assessing data for people in 38 U.S. states, and not limiting outcomes to patients in a particular COVID-19 hot spot, was a unique aspect of the research, said Brown, clinical chief of the Division of Gastroenterology and Hepatology at Weill Cornell Medicine in New York City.
Brown, lead author Michael W. Fried, MD, from TARGET PharmaSolutions in Durham, North Carolina, and colleagues studied adults from a commercially available Target Real-World Evidence (RWE) dataset of nearly 70,000 patients. They examined hospital chargemaster data and ICD-10 codes for COVID-19 inpatients between February 15 and April 20.
This population tended to be older, with 60% older than 60 years. A little more than half of participants, 53%, were men.
Key findings
A total of 21% of patients died after a median hospital length of stay of 8 days.
Older patients were significantly more likely to die, particularly those older than 60 years (P < .0001).
“This confirms some of the things we know about age and its impact on outcome,” Brown said.
The risk for mortality among patients older than 60 years was 7.2 times that of patients between 18 and 40 years in an adjusted multivariate analysis. The risk for death for those between 41 and 60 years of age was lower (odds ratio [OR], 2.6), compared with the youngest cohort.
Men were more likely to die than women (OR, 1.5).
When asked if he was surprised by the high mortality rates, Brown said, “Having worked here in New York? No, I was not.”
Mechanical ventilation and mortality
Male sex, age older than 40 years, obesity, and presence of cardiovascular or chronic kidney disease were risk factors for mechanical ventilation.
Among the nearly 2,000 hospitalized adults requiring mechanical ventilation in the current report, only 27% were discharged alive. “The outcomes of people who are mechanically ventilated are really quite sobering,” Brown said.
People who ever required mechanical ventilation were 32 times more likely to die compared with others whose highest level of oxygenation was low-flow, high-flow, or no-oxygen therapy in an analysis that controlled for demographics and comorbidities.
Furthermore, patients placed on mechanical ventilation earlier – within 24 hours of admission – tended to experience better outcomes.
COVID-19 therapies?
Brown and colleagues also evaluated outcomes in patients who were taking either remdesivir or hydroxychloroquine. A total of 48 people were treated with remdesivir.
The four individuals receiving remdesivir who died were among 11 who were taking remdesivir and also on mechanical ventilation.
“The data for remdesivir is very encouraging,” Brown said.
Many more participants were treated with hydroxychloroquine, more than 4,200 or 36% of the total study population.
A higher proportion of people treated with hydroxychloroquine received mechanical ventilation, at 25%, versus 12% not treated with hydroxychloroquine.
The unadjusted mortality rate was also higher among those treated with the agent, at 25%, compared to 20% not receiving hydroxychloroquine.
The data with hydroxychloroquine can lead to two conclusions, Brown said: “One, it doesn’t work. Or two, it doesn’t work in the way that we use it.”
The researchers cautioned that their hydroxychloroquine findings must be interpreted carefully because those treated with the agent were also more likely to have comorbidities and greater COVID-19 disease severity.
“This study greatly contributes to understanding the natural course of COVID-19 infection by describing characteristics and outcomes of patients with COVID-19 hospitalized throughout the US,” the investigators note. “It identified categories of patients at greatest risk for poor outcomes, which should be used to prioritize prevention and treatment strategies in the future.”
Some limitations
“The findings that patients with hypertension and who were smokers had lower ventilation rates, and patients with hypertension, pulmonary disease, who were smokers had lower mortality risks was very surprising,” Ninez A. Ponce, PhD, MPP, told Medscape Medical News when asked to comment on the study.
Although the study identified multiple risk factors for ventilation and mortality, “unfortunately the dataset did not have race available or disease severity,” said Ponce, director of the UCLA Center for Health Policy Research and professor in the Department of Health Policy and Management at the UCLA Fielding School of Public Health.
“These omitted variables could have a considerable effect on the significance, magnitude, and direction of point estimates provided, so I would be cautious in interpreting the results as a picture of a nationally representative sample,” she said.
On a positive note, the study and dataset could illuminate the utility of medications used to treat COVID-19, Ponce said. In addition, as the authors note, “the data will expand over time.”
Brown has reported receiving grants and consulting for Gilead. Ponce has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Factors including older age and certain comorbidities have been linked to more serious COVID-19 outcomes in previous research, and now a large dataset collected from hundreds of hospitals nationwide provides more detailed data regarding risk for mechanical ventilation and death.
History of pulmonary disease or smoking, interestingly, were not.
One expert urges caution when interpreting the results, however. Although the study found a number of risk factors for ventilation and mortality, she says the dataset lacks information on race and disease severity, and the sample may not be nationally representative.
The investigators hope their level of granularity will further assist researchers searching for effective treatments and clinicians seeking to triage patients during the COVID-19 pandemic.
The study was published online August 28 in Clinical Infectious Diseases.
COVID-19 and comorbidities
“What I found most illuminating was this whole concept of comorbid conditions. This provides suggestive data about who we need to worry about most and who we may need to worry about less,” study author Robert S. Brown Jr, MD, MPH, told Medscape Medical News.
Comorbid conditions included hypertension in 47% of patients, diabetes in 28%, and cardiovascular disease in 19%. Another 16% were obese and 12% had chronic kidney disease. People with comorbid obesity, chronic kidney disease, and cardiovascular disease were more likely to receive mechanical ventilation compared to those without a history of these conditions in an adjusted, multivariable logistic analysis.
With the exception of obesity, the same factors were associated with risk for death during hospitalization.
In contrast, hypertension, history of smoking, and history of pulmonary disease were associated with a lower risk of needing mechanical ventilation and/or lower risk for mortality.
Furthermore, people with liver disease, gastrointestinal diseases, and even autoimmune diseases – which are likely associated with immunosuppression – “are not at that much of an increased risk that we noticed it in our data,” Brown said.
“As I tell many of my patients who have mild liver disease, for example, I would rather have mild liver disease and be on immunosuppressant therapy than be an older, obese male,” he added.
Assessing data for people in 38 U.S. states, and not limiting outcomes to patients in a particular COVID-19 hot spot, was a unique aspect of the research, said Brown, clinical chief of the Division of Gastroenterology and Hepatology at Weill Cornell Medicine in New York City.
Brown, lead author Michael W. Fried, MD, from TARGET PharmaSolutions in Durham, North Carolina, and colleagues studied adults from a commercially available Target Real-World Evidence (RWE) dataset of nearly 70,000 patients. They examined hospital chargemaster data and ICD-10 codes for COVID-19 inpatients between February 15 and April 20.
This population tended to be older, with 60% older than 60 years. A little more than half of participants, 53%, were men.
Key findings
A total of 21% of patients died after a median hospital length of stay of 8 days.
Older patients were significantly more likely to die, particularly those older than 60 years (P < .0001).
“This confirms some of the things we know about age and its impact on outcome,” Brown said.
The risk for mortality among patients older than 60 years was 7.2 times that of patients between 18 and 40 years in an adjusted multivariate analysis. The risk for death for those between 41 and 60 years of age was lower (odds ratio [OR], 2.6), compared with the youngest cohort.
Men were more likely to die than women (OR, 1.5).
When asked if he was surprised by the high mortality rates, Brown said, “Having worked here in New York? No, I was not.”
Mechanical ventilation and mortality
Male sex, age older than 40 years, obesity, and presence of cardiovascular or chronic kidney disease were risk factors for mechanical ventilation.
Among the nearly 2,000 hospitalized adults requiring mechanical ventilation in the current report, only 27% were discharged alive. “The outcomes of people who are mechanically ventilated are really quite sobering,” Brown said.
People who ever required mechanical ventilation were 32 times more likely to die compared with others whose highest level of oxygenation was low-flow, high-flow, or no-oxygen therapy in an analysis that controlled for demographics and comorbidities.
Furthermore, patients placed on mechanical ventilation earlier – within 24 hours of admission – tended to experience better outcomes.
COVID-19 therapies?
Brown and colleagues also evaluated outcomes in patients who were taking either remdesivir or hydroxychloroquine. A total of 48 people were treated with remdesivir.
The four individuals receiving remdesivir who died were among 11 who were taking remdesivir and also on mechanical ventilation.
“The data for remdesivir is very encouraging,” Brown said.
Many more participants were treated with hydroxychloroquine, more than 4,200 or 36% of the total study population.
A higher proportion of people treated with hydroxychloroquine received mechanical ventilation, at 25%, versus 12% not treated with hydroxychloroquine.
The unadjusted mortality rate was also higher among those treated with the agent, at 25%, compared to 20% not receiving hydroxychloroquine.
The data with hydroxychloroquine can lead to two conclusions, Brown said: “One, it doesn’t work. Or two, it doesn’t work in the way that we use it.”
The researchers cautioned that their hydroxychloroquine findings must be interpreted carefully because those treated with the agent were also more likely to have comorbidities and greater COVID-19 disease severity.
“This study greatly contributes to understanding the natural course of COVID-19 infection by describing characteristics and outcomes of patients with COVID-19 hospitalized throughout the US,” the investigators note. “It identified categories of patients at greatest risk for poor outcomes, which should be used to prioritize prevention and treatment strategies in the future.”
Some limitations
“The findings that patients with hypertension and who were smokers had lower ventilation rates, and patients with hypertension, pulmonary disease, who were smokers had lower mortality risks was very surprising,” Ninez A. Ponce, PhD, MPP, told Medscape Medical News when asked to comment on the study.
Although the study identified multiple risk factors for ventilation and mortality, “unfortunately the dataset did not have race available or disease severity,” said Ponce, director of the UCLA Center for Health Policy Research and professor in the Department of Health Policy and Management at the UCLA Fielding School of Public Health.
“These omitted variables could have a considerable effect on the significance, magnitude, and direction of point estimates provided, so I would be cautious in interpreting the results as a picture of a nationally representative sample,” she said.
On a positive note, the study and dataset could illuminate the utility of medications used to treat COVID-19, Ponce said. In addition, as the authors note, “the data will expand over time.”
Brown has reported receiving grants and consulting for Gilead. Ponce has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Factors including older age and certain comorbidities have been linked to more serious COVID-19 outcomes in previous research, and now a large dataset collected from hundreds of hospitals nationwide provides more detailed data regarding risk for mechanical ventilation and death.
History of pulmonary disease or smoking, interestingly, were not.
One expert urges caution when interpreting the results, however. Although the study found a number of risk factors for ventilation and mortality, she says the dataset lacks information on race and disease severity, and the sample may not be nationally representative.
The investigators hope their level of granularity will further assist researchers searching for effective treatments and clinicians seeking to triage patients during the COVID-19 pandemic.
The study was published online August 28 in Clinical Infectious Diseases.
COVID-19 and comorbidities
“What I found most illuminating was this whole concept of comorbid conditions. This provides suggestive data about who we need to worry about most and who we may need to worry about less,” study author Robert S. Brown Jr, MD, MPH, told Medscape Medical News.
Comorbid conditions included hypertension in 47% of patients, diabetes in 28%, and cardiovascular disease in 19%. Another 16% were obese and 12% had chronic kidney disease. People with comorbid obesity, chronic kidney disease, and cardiovascular disease were more likely to receive mechanical ventilation compared to those without a history of these conditions in an adjusted, multivariable logistic analysis.
With the exception of obesity, the same factors were associated with risk for death during hospitalization.
In contrast, hypertension, history of smoking, and history of pulmonary disease were associated with a lower risk of needing mechanical ventilation and/or lower risk for mortality.
Furthermore, people with liver disease, gastrointestinal diseases, and even autoimmune diseases – which are likely associated with immunosuppression – “are not at that much of an increased risk that we noticed it in our data,” Brown said.
“As I tell many of my patients who have mild liver disease, for example, I would rather have mild liver disease and be on immunosuppressant therapy than be an older, obese male,” he added.
Assessing data for people in 38 U.S. states, and not limiting outcomes to patients in a particular COVID-19 hot spot, was a unique aspect of the research, said Brown, clinical chief of the Division of Gastroenterology and Hepatology at Weill Cornell Medicine in New York City.
Brown, lead author Michael W. Fried, MD, from TARGET PharmaSolutions in Durham, North Carolina, and colleagues studied adults from a commercially available Target Real-World Evidence (RWE) dataset of nearly 70,000 patients. They examined hospital chargemaster data and ICD-10 codes for COVID-19 inpatients between February 15 and April 20.
This population tended to be older, with 60% older than 60 years. A little more than half of participants, 53%, were men.
Key findings
A total of 21% of patients died after a median hospital length of stay of 8 days.
Older patients were significantly more likely to die, particularly those older than 60 years (P < .0001).
“This confirms some of the things we know about age and its impact on outcome,” Brown said.
The risk for mortality among patients older than 60 years was 7.2 times that of patients between 18 and 40 years in an adjusted multivariate analysis. The risk for death for those between 41 and 60 years of age was lower (odds ratio [OR], 2.6), compared with the youngest cohort.
Men were more likely to die than women (OR, 1.5).
When asked if he was surprised by the high mortality rates, Brown said, “Having worked here in New York? No, I was not.”
Mechanical ventilation and mortality
Male sex, age older than 40 years, obesity, and presence of cardiovascular or chronic kidney disease were risk factors for mechanical ventilation.
Among the nearly 2,000 hospitalized adults requiring mechanical ventilation in the current report, only 27% were discharged alive. “The outcomes of people who are mechanically ventilated are really quite sobering,” Brown said.
People who ever required mechanical ventilation were 32 times more likely to die compared with others whose highest level of oxygenation was low-flow, high-flow, or no-oxygen therapy in an analysis that controlled for demographics and comorbidities.
Furthermore, patients placed on mechanical ventilation earlier – within 24 hours of admission – tended to experience better outcomes.
COVID-19 therapies?
Brown and colleagues also evaluated outcomes in patients who were taking either remdesivir or hydroxychloroquine. A total of 48 people were treated with remdesivir.
The four individuals receiving remdesivir who died were among 11 who were taking remdesivir and also on mechanical ventilation.
“The data for remdesivir is very encouraging,” Brown said.
Many more participants were treated with hydroxychloroquine, more than 4,200 or 36% of the total study population.
A higher proportion of people treated with hydroxychloroquine received mechanical ventilation, at 25%, versus 12% not treated with hydroxychloroquine.
The unadjusted mortality rate was also higher among those treated with the agent, at 25%, compared to 20% not receiving hydroxychloroquine.
The data with hydroxychloroquine can lead to two conclusions, Brown said: “One, it doesn’t work. Or two, it doesn’t work in the way that we use it.”
The researchers cautioned that their hydroxychloroquine findings must be interpreted carefully because those treated with the agent were also more likely to have comorbidities and greater COVID-19 disease severity.
“This study greatly contributes to understanding the natural course of COVID-19 infection by describing characteristics and outcomes of patients with COVID-19 hospitalized throughout the US,” the investigators note. “It identified categories of patients at greatest risk for poor outcomes, which should be used to prioritize prevention and treatment strategies in the future.”
Some limitations
“The findings that patients with hypertension and who were smokers had lower ventilation rates, and patients with hypertension, pulmonary disease, who were smokers had lower mortality risks was very surprising,” Ninez A. Ponce, PhD, MPP, told Medscape Medical News when asked to comment on the study.
Although the study identified multiple risk factors for ventilation and mortality, “unfortunately the dataset did not have race available or disease severity,” said Ponce, director of the UCLA Center for Health Policy Research and professor in the Department of Health Policy and Management at the UCLA Fielding School of Public Health.
“These omitted variables could have a considerable effect on the significance, magnitude, and direction of point estimates provided, so I would be cautious in interpreting the results as a picture of a nationally representative sample,” she said.
On a positive note, the study and dataset could illuminate the utility of medications used to treat COVID-19, Ponce said. In addition, as the authors note, “the data will expand over time.”
Brown has reported receiving grants and consulting for Gilead. Ponce has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Fatal pediatric melanomas diverse in presentation
results of a retrospective multicenter study showed.
“The most striking thing that we learned from this study is that pediatric melanoma can present in so many different ways, and it’s distinct from the adult population in that we see more presentations associated with congenital nevi, or spitz melanoma, which is a special class of pigmented lesions that looks a little different under the microscope,” Elena B. Hawryluk, MD, PhD, of the department of dermatology at Massachusetts General Hospital (MGH) and Harvard University, Boston, said in an interview. Dr. Hawryluk is lead author of the study, which was published online ahead of print in the Journal of the American Academy of Dermatology.
Dr. Hawryluk and colleagues at MGH and 11 other centers conducted a retrospective review of all cases of fatal pediatric melanoma among patients younger than 20 years diagnosed from late 1994 through early 2017.
They identified a total of 38 fatal cases over more than 2 decades. The cases were distinguished primarily by their heterogeneous clinical presentation and by the diversity of the patients, their precursor lesions, and the tumor histopathology, she said in an interview.
“We were surprised to find that patients with each of these presentations could end up with a fatal course, it wasn’t just all the adolescents, or all the patients with giant congenital nevi; it really presented quite diversely.”
Rare malignancy
Melanoma is far less common in the pediatric population than in adults, with an annual incidence of 18 per 1 million among adolescents aged 15-18 years, and 1 per 1 million in children under 10 years, the authors noted.
“Melanoma in children and adolescents often has distinct clinical presentations such as association with a congenital melanocytic nevus (CMN), spitzoid melanoma, or amelanotic melanoma, which are more rarely observed in adult melanoma patients. Unique pediatric-specific clinical detection criteria have been proposed to highlight these differences, such as a tendency to present amelanotically,” they wrote.
Factors associated with worse prognosis, such as higher Breslow thickness and mitotic index, are more frequently present at the time of diagnosis in children compared with adults, particularly those diagnosed before age 11 years.
“It is unclear if this difference is secondary to diagnostic delays due to low clinical suspicion, atypical clinical presentations, or more rapid tumor growth rate, as many childhood melanomas are of nodular or spitzoid subtypes,” Dr. Hawryluk and her coauthors wrote.
Study details
The investigators sought to characterize the clinical and histopathologic features of fatal pediatric melanomas.
They found that 21 of the 38 patients (57%) were of White heritage, 7 (19%) were of Hispanic or Latino background, 1 (3%) was of Asian lineage, and 1 each were of Black African American or Black Hispanic background. The remaining children were classified as “other” or did not have their ethnic backgrounds recorded.
The “striking prevalence” of Hispanic patients observed in the study is consistent with surveillance reports of an increasing incidence of melanoma among children of Hispanic background, they noted.
The mean age at diagnosis was 12.7 years, and the mean age at death was 15.6 years.
Of the 16 cases with known identifiable disease subtypes, 8 (50%) were nodular, 5 (31%) were superficial spreading, and 3 (19%) were spitzoid melanomas. Of the 38 fatal melanomas, 10 were thought to have originated from congenital melanocytic nevi.
Outlook improving
Recent therapeutic breakthroughs such as targeted agents and immunotherapy with checkpoint inhibitors augur well for children diagnosed with melanoma, Dr. Hawryluk said.
“Fortunately, it’s not superaggressive in children at high frequency, so we generally use adult algorithms to inform treatment decisions,” she said. “It’s just important to note that melanomas that arise in congenital nevi tend to have different driver mutations than those that arise in older patients who may have lots of sun exposure.”
“Nowadays, we’re lucky to have a lot of extra tests and workups so that, if a patient does have metastatic or advance disease, they can have a better genetic profile that would guide our choice of medications,” she added.
The study was supported by a Pediatric Dermatology Research Alliance Study Support grant and Society for Pediatric Dermatology, Pediatric Dermatology Research Alliance Pilot award. Dr. Hawryluk is supported by the Dermatology Foundation and the Harvard Medical School Eleanor and Miles Shore Fellowship award. The authors reported no conflicts of interest.
SOURCE: Hawryluk EB et al. J Am Acad Dermatol. 2020 Jul 1. doi: 10.1016/j.jaad.2020.06.1010.
results of a retrospective multicenter study showed.
“The most striking thing that we learned from this study is that pediatric melanoma can present in so many different ways, and it’s distinct from the adult population in that we see more presentations associated with congenital nevi, or spitz melanoma, which is a special class of pigmented lesions that looks a little different under the microscope,” Elena B. Hawryluk, MD, PhD, of the department of dermatology at Massachusetts General Hospital (MGH) and Harvard University, Boston, said in an interview. Dr. Hawryluk is lead author of the study, which was published online ahead of print in the Journal of the American Academy of Dermatology.
Dr. Hawryluk and colleagues at MGH and 11 other centers conducted a retrospective review of all cases of fatal pediatric melanoma among patients younger than 20 years diagnosed from late 1994 through early 2017.
They identified a total of 38 fatal cases over more than 2 decades. The cases were distinguished primarily by their heterogeneous clinical presentation and by the diversity of the patients, their precursor lesions, and the tumor histopathology, she said in an interview.
“We were surprised to find that patients with each of these presentations could end up with a fatal course, it wasn’t just all the adolescents, or all the patients with giant congenital nevi; it really presented quite diversely.”
Rare malignancy
Melanoma is far less common in the pediatric population than in adults, with an annual incidence of 18 per 1 million among adolescents aged 15-18 years, and 1 per 1 million in children under 10 years, the authors noted.
“Melanoma in children and adolescents often has distinct clinical presentations such as association with a congenital melanocytic nevus (CMN), spitzoid melanoma, or amelanotic melanoma, which are more rarely observed in adult melanoma patients. Unique pediatric-specific clinical detection criteria have been proposed to highlight these differences, such as a tendency to present amelanotically,” they wrote.
Factors associated with worse prognosis, such as higher Breslow thickness and mitotic index, are more frequently present at the time of diagnosis in children compared with adults, particularly those diagnosed before age 11 years.
“It is unclear if this difference is secondary to diagnostic delays due to low clinical suspicion, atypical clinical presentations, or more rapid tumor growth rate, as many childhood melanomas are of nodular or spitzoid subtypes,” Dr. Hawryluk and her coauthors wrote.
Study details
The investigators sought to characterize the clinical and histopathologic features of fatal pediatric melanomas.
They found that 21 of the 38 patients (57%) were of White heritage, 7 (19%) were of Hispanic or Latino background, 1 (3%) was of Asian lineage, and 1 each were of Black African American or Black Hispanic background. The remaining children were classified as “other” or did not have their ethnic backgrounds recorded.
The “striking prevalence” of Hispanic patients observed in the study is consistent with surveillance reports of an increasing incidence of melanoma among children of Hispanic background, they noted.
The mean age at diagnosis was 12.7 years, and the mean age at death was 15.6 years.
Of the 16 cases with known identifiable disease subtypes, 8 (50%) were nodular, 5 (31%) were superficial spreading, and 3 (19%) were spitzoid melanomas. Of the 38 fatal melanomas, 10 were thought to have originated from congenital melanocytic nevi.
Outlook improving
Recent therapeutic breakthroughs such as targeted agents and immunotherapy with checkpoint inhibitors augur well for children diagnosed with melanoma, Dr. Hawryluk said.
“Fortunately, it’s not superaggressive in children at high frequency, so we generally use adult algorithms to inform treatment decisions,” she said. “It’s just important to note that melanomas that arise in congenital nevi tend to have different driver mutations than those that arise in older patients who may have lots of sun exposure.”
“Nowadays, we’re lucky to have a lot of extra tests and workups so that, if a patient does have metastatic or advance disease, they can have a better genetic profile that would guide our choice of medications,” she added.
The study was supported by a Pediatric Dermatology Research Alliance Study Support grant and Society for Pediatric Dermatology, Pediatric Dermatology Research Alliance Pilot award. Dr. Hawryluk is supported by the Dermatology Foundation and the Harvard Medical School Eleanor and Miles Shore Fellowship award. The authors reported no conflicts of interest.
SOURCE: Hawryluk EB et al. J Am Acad Dermatol. 2020 Jul 1. doi: 10.1016/j.jaad.2020.06.1010.
results of a retrospective multicenter study showed.
“The most striking thing that we learned from this study is that pediatric melanoma can present in so many different ways, and it’s distinct from the adult population in that we see more presentations associated with congenital nevi, or spitz melanoma, which is a special class of pigmented lesions that looks a little different under the microscope,” Elena B. Hawryluk, MD, PhD, of the department of dermatology at Massachusetts General Hospital (MGH) and Harvard University, Boston, said in an interview. Dr. Hawryluk is lead author of the study, which was published online ahead of print in the Journal of the American Academy of Dermatology.
Dr. Hawryluk and colleagues at MGH and 11 other centers conducted a retrospective review of all cases of fatal pediatric melanoma among patients younger than 20 years diagnosed from late 1994 through early 2017.
They identified a total of 38 fatal cases over more than 2 decades. The cases were distinguished primarily by their heterogeneous clinical presentation and by the diversity of the patients, their precursor lesions, and the tumor histopathology, she said in an interview.
“We were surprised to find that patients with each of these presentations could end up with a fatal course, it wasn’t just all the adolescents, or all the patients with giant congenital nevi; it really presented quite diversely.”
Rare malignancy
Melanoma is far less common in the pediatric population than in adults, with an annual incidence of 18 per 1 million among adolescents aged 15-18 years, and 1 per 1 million in children under 10 years, the authors noted.
“Melanoma in children and adolescents often has distinct clinical presentations such as association with a congenital melanocytic nevus (CMN), spitzoid melanoma, or amelanotic melanoma, which are more rarely observed in adult melanoma patients. Unique pediatric-specific clinical detection criteria have been proposed to highlight these differences, such as a tendency to present amelanotically,” they wrote.
Factors associated with worse prognosis, such as higher Breslow thickness and mitotic index, are more frequently present at the time of diagnosis in children compared with adults, particularly those diagnosed before age 11 years.
“It is unclear if this difference is secondary to diagnostic delays due to low clinical suspicion, atypical clinical presentations, or more rapid tumor growth rate, as many childhood melanomas are of nodular or spitzoid subtypes,” Dr. Hawryluk and her coauthors wrote.
Study details
The investigators sought to characterize the clinical and histopathologic features of fatal pediatric melanomas.
They found that 21 of the 38 patients (57%) were of White heritage, 7 (19%) were of Hispanic or Latino background, 1 (3%) was of Asian lineage, and 1 each were of Black African American or Black Hispanic background. The remaining children were classified as “other” or did not have their ethnic backgrounds recorded.
The “striking prevalence” of Hispanic patients observed in the study is consistent with surveillance reports of an increasing incidence of melanoma among children of Hispanic background, they noted.
The mean age at diagnosis was 12.7 years, and the mean age at death was 15.6 years.
Of the 16 cases with known identifiable disease subtypes, 8 (50%) were nodular, 5 (31%) were superficial spreading, and 3 (19%) were spitzoid melanomas. Of the 38 fatal melanomas, 10 were thought to have originated from congenital melanocytic nevi.
Outlook improving
Recent therapeutic breakthroughs such as targeted agents and immunotherapy with checkpoint inhibitors augur well for children diagnosed with melanoma, Dr. Hawryluk said.
“Fortunately, it’s not superaggressive in children at high frequency, so we generally use adult algorithms to inform treatment decisions,” she said. “It’s just important to note that melanomas that arise in congenital nevi tend to have different driver mutations than those that arise in older patients who may have lots of sun exposure.”
“Nowadays, we’re lucky to have a lot of extra tests and workups so that, if a patient does have metastatic or advance disease, they can have a better genetic profile that would guide our choice of medications,” she added.
The study was supported by a Pediatric Dermatology Research Alliance Study Support grant and Society for Pediatric Dermatology, Pediatric Dermatology Research Alliance Pilot award. Dr. Hawryluk is supported by the Dermatology Foundation and the Harvard Medical School Eleanor and Miles Shore Fellowship award. The authors reported no conflicts of interest.
SOURCE: Hawryluk EB et al. J Am Acad Dermatol. 2020 Jul 1. doi: 10.1016/j.jaad.2020.06.1010.
FROM JAAD
Psoriasis, PsA, and pregnancy: Tailoring treatment with increasing data
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
With an average age of diagnosis of 28 years, and one of two incidence peaks occurring at 15-30 years, psoriasis affects many women in the midst of their reproductive years. The prospect of pregnancy – or the reality of a surprise pregnancy – drives questions about heritability of the disease in offspring, the impact of the disease on pregnancy outcomes and breastfeeding, and how to best balance risks of treatments with risks of uncontrolled psoriasis and/or psoriatic arthritis (PsA).
While answers to these questions are not always clear, discussions about pregnancy and psoriasis management “shouldn’t be scary,” said Jenny E. Murase, MD, a dermatologist who speaks and writes widely about her research and experience with psoriasis and pregnancy. “We have access to information and data and educational resources to [work with] and reassure our patients – we just need to use it. Right now, there’s unnecessary suffering [with some patients unnecessarily stopping all treatment].”
Much has been learned in the past 2 decades about the course of psoriasis in pregnancy, and pregnancy outcomes data on the safety of biologics during pregnancy are increasingly emerging – particularly for tumor necrosis factor (TNF)–alpha inhibitors.
Ideally, since half of all pregnancies are unplanned, the implications of therapeutic options should be discussed with all women with psoriasis who are of reproductive age, whether they are sexually active or not. “The onus is on us to make sure that we’re considering the possibility [that our patient] could become pregnant without consulting us first,” said Dr. Murase, associate professor of dermatology at the University of California, San Francisco, and director of medical consultative dermatology for the Palo Alto Foundation Medical Group in Mountain View, Calif.
Lisa R. Sammaritano, MD, associate professor of clinical medicine at Weill Cornell Medicine and a rheumatologist at the Hospital for Special Surgery, both in New York, urges similar attention for PsA. “Pregnancy is best planned while patients have quiescent disease on pregnancy-compatible medications,” she said. “We encourage [more] rheumatologists to be actively involved in pregnancy planning [in order] to guide therapy.”
The impact of estrogen
Dr. Murase was inspired to study psoriasis and pregnancy in part by a patient she met as a medical student. “She had severe psoriasis covering her body, and she said that the only times her psoriasis cleared was during her three pregnancies,” Dr. Murase recalled. “I wondered: What about the pregnancies resulted in such a substantial reduction of her psoriasis?”
She subsequently led a study, published in 2005, of 47 pregnant and 27 nonpregnant patients with psoriasis. More than half of the patients – 55% – reported improvements in their psoriasis during pregnancy, 21% reported no change, and 23% reported worsening. Among the 16 patients who had 10% or greater psoriatic body surface area (BSA) involvement and reported improvements, lesions decreased by 84%.
In the postpartum period, only 9% reported improvement, 26% reported no change, and 65% reported worsening. The increased BSA values observed 6 weeks postpartum did not exceed those of the first trimester, suggesting a return to the patients’ baseline status.
Earlier and smaller retrospective studies had also shown that approximately half of patients improve during pregnancy, and it was believed that progesterone was most likely responsible for this improvement. Dr. Murase’s study moved the needle in that it examined BSA in pregnancy and the postpartum period. It also turned the spotlight on estrogen: Patients who had higher levels of improvement also had higher levels of estradiol, estrone, and the ratio of estrogen to progesterone. However, there was no correlation between psoriatic change and levels of progesterone.
To promote fetal survival, pregnancy triggers a shift from Th1 cell–mediated immunity – and Th17 immunity – to Th2 immunity. While there’s no proof of a causative effect, increased estrogen appears to play a role in this shift and in the reduced production of Th1 and Th17 cytokines. Psoriasis is believed to be primarily a Th17-mediated disease, with some Th1 involvement, so this down-regulation can result in improved disease status, Dr. Murase said. (A host of other autoimmune diseases categorized as Th1 mediated similarly tend to improve during pregnancy, she added.)
Information on the effect of pregnancy on PsA is “conflicting,” Dr. Sammaritano said. “Some [of a limited number of studies] suggest a beneficial effect as is generally seen for rheumatoid arthritis. Others, however, have found an increased risk of disease activity during pregnancy ... It may be that psoriatic arthritis can be quite variable from patient to patient in its clinical presentation.”
At least one study, Dr. Sammaritano added, “has shown that the arthritis in pregnancy patients with PsA did not improve, compared to control nonpregnant patients, while the psoriasis rash did improve.”
The mixed findings don’t surprise Dr. Murase. “It harder to quantify joint disease in general,” she said. “And during pregnancy, physiologic changes relating to the pregnancy itself can cause discomfort – your joints ache. The numbers [of improved] cases aren’t as high with PsA, but it’s a more complex question.”
In the postpartum period, however, research findings “all suggest an increased risk of flare” of PsA, Dr. Sammaritano said, just as with psoriasis.
Assessing risk of treatment
Understanding the immunologic effects of pregnancy on psoriasis and PsA – and appreciating the concept of a hormonal component – is an important part of treatment decision making. So is understanding pregnancy outcomes data.
Researchers have looked at a host of pregnancy outcomes – including congenital malformations, preterm birth, spontaneous abortion, low birth weight, macrosomia, and gestational diabetes and hypertension – in women with psoriasis or psoriasis/PsA, compared with control groups. Some studies have suggested a link between disease activity and pregnancy complications or adverse pregnancy outcomes, “just as a result of having moderate to severe disease,” while others have found no evidence of increased risk, Dr. Murase said.
“It’s a bit unclear and a difficult question to answer; it depends on what study you look at and what data you believe. It would be nice to have some clarity, but basically the jury is still out,” said Dr. Murase, who, with coauthors Alice B. Gottlieb, MD, PhD, of the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, and Caitriona Ryan, MD, of the Blackrock Clinic and Charles Institute of Dermatology, University College Dublin, discussed the pregnancy outcomes data in a recently published review of psoriasis in women.
“In my opinion, because we have therapies that are so low risk and well tolerated, it’s better to make sure that the inflammatory cascade and inflammation created by psoriasis is under control,” she said. “So whether or not the pregnancy itself causes the patient to go into remission, or whether you have to use therapy to help the patient stay in remission, it’s important to control the inflammation.”
Contraindicated in pregnancy are oral psoralen, methotrexate, and acitretin, the latter of which should be avoided for several years before pregnancy and “therefore shouldn’t be used in a woman of childbearing age,” said Dr. Murase. Methotrexate, said Dr. Sammaritano, should generally be stopped 1-3 months prior to conception.
For psoriasis, the therapy that’s “classically considered the safest in pregnancy is UVB light therapy, specifically the 300-nm wavelength of light, which works really well as an anti-inflammatory,” Dr. Murase said. Because of the potential for maternal folate degradation with phototherapy and the long-known association of folate deficiency with neural tube defects, women of childbearing age who are receiving light therapy should take daily folic acid supplementation. (She prescribes a daily prenatal vitamin containing at least 1 mg of folic acid for women who are utilizing light therapy.)
Many topical agents can be used during pregnancy, Dr. Murase said. Topical corticosteroids, she noted, have the most safety-affirming data of any topical medication.
Regarding oral therapies, Dr. Murase recommends against the use of apremilast (Otezla) for her patients. “It’s not contraindicated, but the animal studies don’t look promising, so I don’t use that one in women of childbearing age just in case. There’s just very little data to support the safety of this medication [in pregnancy].”
There are no therapeutic guidelines in the United States for guiding the management of psoriasis in women who are considering pregnancy. In 2012, the medical board of the National Psoriasis Foundation published a review of treatment options for psoriasis in pregnant or lactating women, the “closest thing to guidelines that we’ve had,” said Dr. Murase. (Now almost a decade old, the review addresses TNF inhibitors but does not cover the anti-interleukin agents more recently approved for moderate to severe psoriasis and PsA.)
For treating PsA, rheumatologists now have the American College of Rheumatology’s first guideline for the management of reproductive health in rheumatic and musculoskeletal diseases to reference. The 2020 guideline does not address PsA specifically, but its section on pregnancy and lactation includes recommendations on biologic and other therapies used to treat the disease.
Guidelines aside, physician-patient discussions over drug safety have the potential to be much more meaningful now that drug labels offer clinical summaries, data, and risk summaries regarding potential use in pregnancy. The labels have “more of a narrative, which is a more useful way to counsel patients and make risk-benefit decisions” than the former system of five-letter categories, said Dr. Murase. (The changes were made per the Pregnancy and Lactation Labeling Rule of 2015.)
MothertoBaby, a service of the nonprofit Organization of Teratology Information Specialists, also provides good evidence-based information to physicians and mothers, Dr. Sammaritano noted.
The use of biologic therapies
In a 2017 review of biologic safety for patients with psoriasis during pregnancy, Alexa B. Kimball, MD, MPH, professor of dermatology at Harvard Medical School, Boston; Martina L. Porter, MD, currently with the department of dermatology at Beth Israel Deaconess Medical Center, Boston; and Stephen J. Lockwood, MD, MPH, of the department of dermatology at Harvard Medical School, concluded that an increasing body of literature suggests that biologic agents can be used during pregnancy and breastfeeding. Anti-TNF agents “should be considered over IL-12/23 and IL-17 inhibitors due to the increased availability of long-term data,” they wrote.
“In general,” said Dr. Murase, “there’s more and more data coming out from gastroenterology and rheumatology to reassure patients and prescribing physicians that the TNF-blocker class is likely safe to use in pregnancy,” particularly during the first trimester and early second trimester, when the transport of maternal antibodies across the placenta is “essentially nonexistent.” In the third trimester, the active transport of IgG antibodies increases rapidly.
If possible, said Dr. Sammaritano, who served as lead author of the ACR’s reproductive health guideline, TNF inhibitors “will be stopped prior to the third trimester to avoid [the possibility of] high drug levels in the infant at birth, which raises concern for immunosuppression in the newborn. If disease is very active, however, they can be continued throughout the pregnancy.”
The TNF inhibitor certolizumab pegol (Cimzia) has the advantage of being transported only minimally across the placenta, if at all, she and Dr. Murase both explained. “To be actively carried across, antibodies need what’s called an Fc region for the placenta to grab onto,” Dr. Murase said. Certolizumab – a pegylated anti–binding fragment antibody – lacks this Fc region.
Two recent studies – CRIB and a UCB Pharma safety database analysis – showed “essentially no medication crossing – there were barely detectable levels,” Dr. Murase said. Certolizumab’s label contains this information and other clinical trial data as well as findings from safety database analyses/surveillance registries.
“Before we had much data for the biologics, I’d advise transitioning patients to light therapy from their biologics and a lot of times their psoriasis would improve, but it was more of a dance,” she said. “Now we tend to look at [certolizumab] when they’re of childbearing age and keep them on the treatment. I know that the baby is not being immunosuppressed.”
Consideration of the use of certolizumab when treatment with biologic agents is required throughout the pregnancy is a recommendation included in Dr. Kimball’s 2017 review.
As newer anti-interleukin agents – the IL-12/23 and IL-17 inhibitors – play a growing role in the treatment of psoriasis and PsA, questions loom about their safety profile. Dr. Murase and Dr. Sammaritano are waiting for more data. “In general,” Dr. Sammaritano said, “we recommend stopping them at the time pregnancy is detected, based on a lack of data at this time.”
Small-molecule drugs are also less well studied, she noted. “Because of their low molecular weight, we anticipate they will easily cross the placenta, so we recommend avoiding use during pregnancy until more information is available.”
Postpartum care
The good news, both experts say, is that the vast majority of medications, including biologics, are safe to use during breastfeeding. Methotrexate should be avoided, Dr. Sammaritano pointed out, and the impact of novel small-molecule therapies on breast milk has not been studied.
In her 2019 review of psoriasis in women, Dr. Murase and coauthors wrote that too many dermatologists believe that breastfeeding women should either not be on biologics or are uncertain about biologic use during breastfeeding. However, “biologics are considered compatible for use while breastfeeding due to their large molecular size and the proteolytic environment in the neonatal gastrointestinal tract,” they added.
Counseling and support for breastfeeding is especially important for women with psoriasis, Dr. Murase emphasized. “Breastfeeding is very traumatizing to the skin, and psoriasis can form in skin that’s injured. I have my patients set up an office visit very soon after the pregnancy to make sure they’re doing alright with their breastfeeding and that they’re coating their nipple area with some type of moisturizer and keeping the health of their nipples in good shape.”
Timely reviews of therapy and adjustments are also a priority, she said. “We need to prepare for 6 weeks post partum” when psoriasis will often flare without treatment.
Dr. Murase disclosed that she is a consultant for Dermira, UCB Pharma, Sanofi, Ferndale, and Regeneron. She is also coeditor in chief of the International Journal of Women’s Dermatology. Dr. Sammaritano reported that she has no disclosures relating to the treatment of PsA.
First randomized trial reassures on ACEIs, ARBs in COVID-19
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The first randomized study to compare continuing versus stopping ACE inhibitors or angiotensin receptor blockers (ARBs) for patients with COVID-19 has shown no difference in key outcomes between the two approaches.
The BRACE CORONA trial – conducted in patients had been taking an ACE inhibitor or an ARB on a long-term basis and who were subsequently hospitalized with COVID-19 – showed no difference in the primary endpoint of number of days alive and out of hospital among those whose medication was suspended for 30 days and those who continued undergoing treatment with these agents.
“Because these data indicate that there is no clinical benefit from routinely interrupting these medications in hospitalized patients with mild to moderate COVID-19, they should generally be continued for those with an indication,” principal investigator Renato Lopes, MD, of Duke Clinical Research Institute, Durham, N.C., concluded.
The BRACE CORONA trial was presented at the European Society of Cardiology Congress 2020 on Sept. 1.
Dr. Lopes explained that there are two conflicting hypotheses about the role of ACE inhibitors and ARBs in COVID-19.
One hypothesis suggests that use of these drugs could be harmful by increasing the expression of ACE2 receptors (which the SARS-CoV-2 virus uses to gain entry into cells), thus potentially enhancing viral binding and viral entry. The other suggests that ACE inhibitors and ARBs could be protective by reducing production of angiotensin II and enhancing the generation of angiotensin 1-7, which attenuates inflammation and fibrosis and therefore could attenuate lung injury.
The BRACE CORONA trial was an academic-led randomized study that tested two strategies: temporarily stopping the ACE inhibitor/ARB for 30 days or continuing these drugs for patients who had been taking these medications on a long-term basis and were hospitalized with a confirmed diagnosis of COVID-19.
The primary outcome was the number of days alive and out of hospital at 30 days. Patients who were using more than three antihypertensive drugs or sacubitril/valsartan or who were hemodynamically unstable at presentation were excluded from the study.
The trial enrolled 659 patients from 29 sites in Brazil. The mean age of patients was 56 years, 40% were women, and 52% were obese. ACE inhibitors were being taken by 15% of the trial participants; ARBs were being taken by 85%. The median duration of ACE inhibitor/ARB treatment was 5 years.
Patients were a median of 6 days from COVID-19 symptom onset. For 30% of the patients, oxygen saturation was below 94% at entry. In terms of COVID-19 symptoms, 57% were classified as mild, and 43% as moderate.
Those with severe COVID-19 symptoms who needed intubation or vasoactive drugs were excluded. Antihypertensive therapy would generally be discontinued in these patients anyway, Dr. Lopes said.
Results showed that the average number of days alive and out of hospital was 21.9 days for patients who stopped taking ACE inhibitors/ARBs and 22.9 days for patients who continued taking these medications. The average difference between groups was –1.1 days.
The average ratio of days alive and out of hospital between the suspending and continuing groups was 0.95 (95% CI, 0.90-1.01; P = .09).
The proportion of patients alive and out of hospital by the end of 30 days in the suspending ACE inhibitor/ARB group was 91.8% versus 95% in the continuing group.
A similar 30-day mortality rate was seen for patients who continued and those who suspended ACE inhibitor/ARB therapy, at 2.8% and 2.7%, respectively (hazard ratio, 0.97). The median number of days that patients were alive and out of hospital was 25 in both groups.
Dr. Lopes said that there was no difference between the two groups with regard to many other secondary outcomes. These included COVID-19 disease progression (need for intubation, ventilation, need for vasoactive drugs, or imaging results) and cardiovascular endpoints (MI, stroke, thromboembolic events, worsening heart failure, myocarditis, or hypertensive crisis).
“Our results endorse with reliable and more definitive data what most medical and cardiovascular societies are recommending – that patients do not stop ACE inhibitor or ARB medication. This has been based on observational data so far, but BRACE CORONA now provides randomized data to support this recommendation,” Dr. Lopes concluded.
Dr. Lopes noted that several subgroups had been prespecified for analysis. Factors included age, obesity, difference between ACE inhibitors/ARBs, difference in oxygen saturation at presentation, time since COVID-19 symptom onset, degree of lung involvement on CT, and symptom severity on presentation.
“We saw very consistent effects of our main findings across all these subgroups, and we plan to report more details of these in the near future,” he said.
Protective for older patients?
The discussant of the study at the ESC Hotline session, Gianfranco Parati, MD, University of Milan-Bicocca and San Luca Hospital, Milan, congratulated Lopes and his team for conducting this important trial at such a difficult time.
He pointed out that patients in the BRACE CORONA trial were quite young (average age, 56 years) and that observational data so far suggest that ACE inhibitors and ARBs have a stronger protective effect in older COVID-19 patients.
He also noted that the percentage of patients alive and out of hospital at 30 days was higher for the patients who continued on treatment in this study (95% vs. 91.8%), which suggested an advantage in maintaining the medication.
Dr. Lopes replied that one-quarter of the population in the BRACE CORONA trial was older than 65 years, which he said was a “reasonable number.”
“Subgroup analysis by age did not show a significant interaction, but the effect of continuing treatment does seem to be more favorable in older patients and also in those who were sicker and had more comorbidities,” he added.
Dr. Parati also suggested that it would have been difficult to discern differences between ACE inhibitors and ARBs in the BRACE CORONA trial, because so few patents were taking ACE inhibitors; the follow-up period of 30 days was relatively short, inasmuch as these drugs may have long-term effects; and it would have been difficult to show differences in the main outcomes used in the study – mortality and time out of hospital – in these patients with mild to moderate disease.
Franz H. Messerli, MD, and Christoph Gräni, MD, University of Bern (Switzerland), said in a joint statement: “The BRACE CORONA trial provides answers to what we know from retrospective studies: if you have already COVID, don’t stop renin-angiotensin system blocker medication.”
But they added that the study does not answer the question about the risk/benefit of ACE inhibitors or ARBs with regard to possible enhanced viral entry through the ACE2 receptor. “What about all those on these drugs who are not infected with COVID? Do they need to stop them? We simply don’t know yet,” they said.
Dr. Messerli and Dr. Gräni added that they would like to see a study that compared patients before SARS-CoV-2 infection who were without hypertension, patients with hypertension who were taking ACE inhibitors or ARBs, and patients with hypertension taking other antihypertensive drugs.
The BRACE CORONA trial was sponsored by D’Or Institute for Research and Education and the Brazilian Clinical Research Institute. Dr. Lopes has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.