User login
In Case You Missed It: COVID
States ready plans to get Pfizer COVID vaccine to younger teens
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
after the Food and Drug Administration authorized its use in this age group May 10.
Some states hope to start the vaccinations as early as May 13, officials said at an Association of State and Territorial Health Officials news conference.
There are, however, two more steps before shots can reach younger arms. On May 12, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is expected to recommend use of the vaccine in this age group. Then CDC Director Rochelle Walensky, MD, must make a final decision to begin vaccinating 12- to 15-year-olds.
Some hoping to start this week
Both the CDC panel and Dr. Walensky are expected to sign off on the vaccine’s use. States have been making plans on how to tailor the vaccination message not just to the patient this time, but to parents and guardians as well, some of whom are hesitant to consent.
Some schools, assuming approval May 12, are ready to start vaccinating in cafeterias and gyms.
Anne Zink, MD, president-elect of the Association of State and Territorial Health Officials and Alaska chief medical officer, told reporters that many of her state’s boroughs and districts have booked in-person vaccines for their schools May 12 as the state has dismissal for summer as early as this week.
Maine is readying four types of distribution sites for the vaccines: primary care offices, Walgreen’s and CVS pharmacies, mass vaccination sites, and schools, said Nirav Shah, MD, current ASTHO president and director of the Maine Center for Disease Control and Prevention.
Starting later this week, he said, the state hopes to host large vaccination clinics for people age 12 and over.
Eliminating barriers
States are working to break down barriers through education and improving access.
In Alaska, many of the drive-through evening vaccination sites are being changed to Pfizer sites so parents just getting off work can take their kids.
It’s also important to get young people to speak to their peers about the importance of vaccines, she said. Some teen groups in Alaska are hosting Zoom calls where they share with children and families why they chose to get vaccinated.
In Maine, Dr. Shah said, “the notion of informed consent applies with equal force to adults as it does with adolescents.” But at least in Maine, it is not required that a parent be on site and present during the vaccination itself.
A parent could sign a form allowing the child to be vaccinated in a school-based clinic. Maine also allows verbal consent so a parent can give consent over the phone, Dr. Shah said.
Dividing vaccine trays
Vaccines going to pediatrician and family medicine offices presents a challenge in that smaller numbers of doses are needed for those venues than at large vaccination sites that get trays of 1,170 Pfizer doses each.
Dr. Shah says states have been talking with federal authorities on the need for smaller packaging.
“Breaking the trays up into smaller lot sizes takes a fair amount of effort,” Dr. Shah said. “We understand that later this month the lot size will be going down to 450.”
But even that will be too much for small offices, he said.
Similarly, an effort is being made in Maine to make sure doctors’ offices are not limited by their refrigeration capabilities. The Pfizer vaccine must be kept at ultra-cold temperatures that many primary care doctors’ offices may not have.
“If they need a cool cube with dry ice, we can furnish that to them,” Dr. Shah said.
Should they be mandated?
Dr. Zink said Alaska generally has high acceptance for recommendations around COVID-19 and has no plans to mandate the COVID-19 vaccines for children.
Umair A. Shah, MD, secretary of health at the Washington State Department of Health, said, “Our number one ability to get people vaccinated is for them to be encouraged to do so, to be incentivized to do so, to do everything we can to make the vaccine choice the easy choice,” including eliminating language, cultural and access barriers.
However, he said, “in higher education, University of Washington and Washington State University have indicated they are going to require COVID vaccines for kids to come back to school. I do think that is something that is increasingly being looked at.”
Though the messages will be tailored differently across the states the bottom line will be the same, Dr. Shah said: The vaccines work and they are safe.
But most critically, “Vaccines are our pathway to moving forward and once and for all ending this pandemic,” he said.
A version of this article first appeared on Medscape.com.
Adulterants in street drugs could increase susceptibility to COVID
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
The composition of street drugs like heroin and cocaine are changing. According to a new analysis, almost all contain at least one toxic adulterant, and many contain a plethora. Most adulterants have pharmacologic activities and toxicities. Their presence has added impact in the context of the COVID-19 pandemic, since some may cause a drastic drop in white blood cells that could leave drug users more vulnerable to infection.
“It’s remarkable that we just forgot to notice, in the horrendous transition from prescription opioid epidemic to the illicit opioid and psychostimulant epidemics, that we would have to pay special attention to what the medications are in the drugs that the person was exposed to – and for how long,” said Mark S. Gold, MD, a coauthor of the review.
The analysis showed that adulterants include new psychoactive substances, industrial compounds, fungicides, veterinary medications, and various impurities. In addition, other various medications are being found in street drugs, such as antipsychotics, antidepressants, anxiolytics, antihistamines, anthelmintics, anesthetics, anti-inflammatory agents, antipyretics, analgesics, antispasmodics, antiarrhythmics, antimalarials, bronchodilators, decongestants, expectorants, muscle relaxers, natural/synthetic hallucinogens, and sedatives.
Illicit drugs are by nature manufactured without Food and Drug Administration oversight, and it is becoming increasingly common that substances like leftover medicines and other active drugs are added to illicit drug batches to add weight, said Dr. Gold, a professor at Washington University,St. Louis. The study appeared in Current Psychopharmacology.
Effects of adulterants ‘terrifying’
The findings of adulterants and their consequences are concerning, according to Jean Lud Cadet, MD, who was asked to comment on the findings. “The blood dysplasia, the pulmonary problems that some of those adulterants can cause – it’s actually terrifying, to put it bluntly,” said Dr. Cadet, who is a senior investigator and chief of the Molecular Neuropsychiatry Research Branch at the National Institute on Drug Abuse.
Before 2000, street drugs were generally diluted with comparatively benign substances such as caffeine, sugars, or lidocaine. Drugs like phenacetin, levamisole, acetaminophen, and diltiazem began to appear in heroin and cocaine in the late 1990s, and by 2010, more powerful adulterants like fentanyl, ketamine, and quetiapine became common.
In 2015, the U.S. Department of State partnered with the Colombo Plan, an international organization based in Sri Lanka, to use field spectroscopy to detect toxins directly in cocaine and heroin samples found in Argentina, Brazil, Ecuador, Peru, Sri Lanka, Thailand, Honduras, Guatemala, Mexico, Colombia, and South Africa. They found a range of adulterants such as aminopyrine, diltiazem, metamizole, levamisole, and phenacetin.
A similar project with 431 heroin and cocaine samples from Vermont and Kentucky found that 69% of samples had five or more controlled drugs, toxic adulterants, or impurities. About 15% had nine or more, and 95% of samples had at least one toxic adulterant.
In the midst of the COVID-19 pandemic, these adulterants take on even greater significance. Individuals with substance use disorders often have other health conditions that can make them more vulnerable to viral infections, and this could be exacerbated by the effects of adulterants on white blood cells or other systems. The pandemic has also had an indirect effect by causing a shortage of street drugs. During production shortages, traffickers might boost potency by adding more cutting agents and adulterants. As a result, COVID-19 and opioid addiction tend to reinforce each other.
“The clinical message would be that our [substance use] patients will contract infectious disease and need to be prioritized for [COVID-19] vaccination,” said Dr. Gold.
The findings came as a surprise to Dr. Cadet, and that illustrates a need to publicize the presence of adulterants in street drugs.
“If I wasn’t aware of many of these, then the general public is also not going to be aware of them,” Dr. Cadet said. “Scientists, including myself, and government agencies need to do a better job [of communicating this issue].”
The study references individuals with substance use disorder, but Dr. Cadet cautioned that anyone who uses street drugs, even once or twice, could be a victim of adulterants. “You don’t need to have met criteria for diagnosis in order to suffer the consequences.”
The study had no funding. Dr. Gold and Dr. Cadet have no relevant financial disclosures.
FROM CURRENT PSYCHOPHARMACOLOGY
Support group for Asian Americans uses theater to cope with COVID
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
An online, culturally based peer support group that uses theater and other creative outlets is helping Asian Americans cope with the COVID-19 pandemic, new research shows.
The findings of the qualitative study suggest that the program could be a model to support the mental health of other minority community groups during the COVID pandemic and beyond, say investigators from the Yale University Child Study Center, New Haven, Conn.
The Yale Compassionate Home, Action Together (CHATogether) group was created to promote emotional wellness among Asian American youth, young adults, and their families.
Early in the pandemic, it expanded its purpose to serve as a COVID-19 support group. Through social media outreach, CHATogether encourages members to cope with COVID-19 by using productive and creative outlets.
“We are a community education program serving Asian American families,” said Eunice Yuen, MD, PhD, the program’s founder and director, who is with the Yale University Child Study Center.
“ such as family conflict and xenophobic attacks,” said Dr. Yuen.
She discussed the program at the annual meeting of the American Psychiatric Association, which was held as a virtual live event.
Skits, role playing
CHATogether groups consist of people with similar experiences and challenges who support each other through weekly online group meetings, she explained.
Group members work together to create family conflict scenarios and role-play dialogues on topics amplified during the COVID-19 pandemic, such as cross-cultural challenges among Asian Americans, academic expectations in home schooling, and Black Lives Matter and LGBTQ conflicts within Asian families.
Group members create skits that are based on their personal experiences and that allow them to work through their own internal conflicts and gain a sense of agency, said Dr. Yuen.
“CHATogether is really the interface of mental health, art, and theater, and we’re trying to create a vehicle that can be a lighthearted way for people to talk about mental health, especially for Asian American families,” said Dr. Yuen.
Preliminary results from a focus group with 10 CHATogether members who joined the program since the pandemic started identified four major ways in which the program has had a positive impact on the mental health and well-being of participants:
- It provides a safe and supportive environment, strengthens bonds between members, and increases the sense of belonging, thus encouraging engagement.
- It provides structural consistency/stability through regular meetings and consistent group functions. Weekly meetings provide a sense of control and hope in the midst of uncertainty during periods of sheltering in place.
- Through adapting the group to virtual platforms, group members experience the inherent strengths of a growth mindset and cognitive flexibility when facing challenges.
- It supports healthy coping skills through sublimation and altruism.
Looking ahead, Dr. Yuen said, the team plans to investigate the validity and effectiveness of this model and to expand the group to include other minorities, school educators, and medical education for trainees and medical students.
Commenting on the program, briefing moderator Jeffrey Borenstein, MD, president and CEO of the Brain and Behavior Research Foundation and editor-in-chief of Psychiatric News, described the initiative as a “great project that serves as a model that can be used not only for Asian Americans but for other groups.
“I think the key to it is that cultural sensitivity that we need to really take into account and cultural differences among people in order to best engage them and help support them. I think this program does that beautifully,” said Dr. Borenstein.
The work was supported by the APA’s Substance Abuse and Mental Health Services Administration Minority Fellowship, which provides a 1-year fellowship to psychiatry residents committed to addressing minority psychiatric mental health issues. Dr. Yuen and Dr. Borenstein disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Palliative care in the pandemic: How one hospital met the challenge
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
Clarissa Johnston, MD, said during a virtual presentation at the annual meeting of the Society of General Internal Medicine.
Dr. Johnston, of the University of Texas at Austin, and colleagues experienced an extreme COVID-19 surge when they reopened after initial closure in the first weeks of the pandemic.
“Our hospital and clinics are the health care safety net in Austin, and we serve a predominantly uninsured and Hispanic population that experienced a greater burden of COVID-19 than other populations in our area,” she said in the presentation.
The rapid onset and spread of COVID-19 locally required physicians and staff to innovate quickly, and “we developed and implemented collaborative and novel partnerships between generalists and palliative care specialists to help ensure that our core humanizing values were not lost in the pandemic,” Dr. Johnston emphasized.
Collaboration between internal medicine and palliative care involved developing relationship-centered communication for families and health care workers, as well as engaging medical students in a Transitions of Care elective, Dr. Johnston said.
The early weeks of the pandemic impacted families with the no visitor policy and the loss of death rituals, she said. Health care providers suffered, too, as nurses experienced an overload of work, fears for their own health and safety, and feelings of disconnect from their patients. Physicians dealt with the challenges of a unique illness, and their own fears and uncertainty, Dr. Johnston said.
Meeting communication challenges
One of the strategies used to bridge the communication gap caused by the lack of visitors and family contact was the adoption of the Meet My Loved One program, adapted from a similar program at the University of Alabama, said Dr. Johnston. Meet My Loved One was a collaborative effort focused on ICU patients, Dr. Johnston said. Members of the primary care team, including medical students in the Transitions of Care elective, called family members of ICU patients to collect personal details and humanizing information about the patient, such as preferred name, favorite foods, favorite activities, and some personal history (i.e. played basketball when he was young), and this information was collated, summarized, and posted on the door of the patient’s room.
Advanced care conversations
Advanced care planning (ACP) benefits include not only the promotion of patient-centered care, but also decreases in ICU admissions, length of stay, and cost. Dr. Johnston and colleagues developed a multipronged curriculum that trainees could use to have ACP conversations with clinic patients who would be considered high risk if they developed COVID-19 infections, Dr. Johnston explained. As part of the elective, medical students were trained to have ACP conversations with patients via telehealth; students practiced role-playing conversations with each other via Zoom and met virtually as a group to review the conversations, she said.
Maintaining Humanity
“COVID-19 has changed the way we interact with patients and families,” Dr. Johnston said in an interview. The inability to rely on face-to-face discussions means that “we really need to think carefully how we maintain humanity and the human touch,” she said.
Challenges in providing palliative care during the pandemic include “maintaining humanity, remembering that there is a person behind the prone, paralyzed patient, with family members who love them, and are desperate to be with them but unable,” Dr. Johnston said.
“The Meet My Loved One program helped, as well as multidisciplinary rounds, chaplain services, and frequent check ins with the bedside nurses,” she said.
“I tried hard to call families every day to start to build that trust and rapport that was lost by all the distancing and lack of visits. I didn’t realize how much the day in and day out care of ICU patients is witnessed by families when they are in the room,” she noted. “During COVID-19, it was so much harder to build trust, especially when you add in the inequities and structural racism problems in our health care system,” she said.
“Why would a family member believe and trust some random doctor calling them on the phone? Were we really trying our hardest? Families didn’t have a way to assess that, at least not like they do when they are at bedside and see how hard everyone works,” Dr. Johnston said. “Video visits helped but were not the same.”
Some key lessons about palliative care Dr. Johnson said she learned from the pandemic were how important it is to remember the patient and family, “how we need to work to build trust,” and that clinicians should be mindful that video visits don’t work for everyone, and to “ask, ask, ask about what you don’t know, including death rituals.”
Additional research needs in palliative care in the wake of COVID-19 include more information on what works and what doesn’t work, from the patient and family perspective, said Dr. Johnston. Communication strategies are important, and “we need to address how we can better communicate around serious illness and end-of-life issues with Black and Brown communities,” she said.
Challenges of COVID care
One of the main challenges to providing palliative care in the early days of the pandemic was navigating the constantly evolving science of COVID-19, Aziz Ansari, DO, of Loyola University Chicago, Maywood, Ill., said in an interview.
“It was, and remains, very hard to prognosticate on how a patient will do having respiratory failure with COVID,” said Dr. Ansari, who was the leader of the Palliative Care interest group at the SGIM meeting.
“So, the challenge was how to have a conversation on goals, values, and preferences when we really did not know the disease entity,” Dr. Ansari noted.
“We were surprised many times [when patients with COVID-19] recovered though it took a long time, so we could not really say that in the acute phase of COVID, it was a terminal illness,” he noted.
“Regardless, it still behooves us to have conversations with our patients and families about what are they willing to go through, and how they define a quality of life,” he said.
Strategies such as those used at the University of Texas show the importance of primary care palliative skill development, said Dr. Ansari. “Every physician should have the skill set of having conversations with patients and families on goals, values, and preferences even in unknown situations,” he said. That lifelong skill set development begins in medical school, he added.
Dr. Johnston and Dr. Ansari had no financial conflicts to disclose.
FROM SGIM 2021
Impact of the COVID-19 Pandemic on Multiple Sclerosis Care for Veterans
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
The following is a lightly edited transcript of a teleconference recorded in February 2021.
How has COVID impacted Veterans with multiple sclerosis?
Mitchell Wallin, MD, MPH: There has been a lot of concern in the multiple sclerosis (MS) patient community about getting infected with COVID-19 and what to do about it. Now that there are vaccines, the concern is whether and how to take a vaccine. At least here, in the Washington DC/Baltimore area where I practice, we have seen many veterans being hospitalized with COVID-19, some with multiple sclerosis (MS), and some who have died of COVID-19. So, there has been a lot of fear, especially in veterans that are older with comorbid diseases.
Rebecca Spain, MD, MSPH: There also has been an impact on our ability to provide care to our veterans with MS. There are challenges having them come into the office or providing virtual care. There are additional challenges and concerns this year about making changes in MS medications because we can’t see patients in person to or understand their needs or current status of their MS. So, providing care has been a challenge this year as well.
There has also been an impact on our day to day lives, like there has been for all of us, from the lockdown particularly not being able to exercise and socialize as much. There have been physical and social and emotional tolls that this disease has taken on veterans with MS.
Jodie Haselkorn, MD, MPH: The survivors of COVID-19, that are transferred to an inpatient multidisciplinary rehabilitation program unit to address impairments related to the cardiopulmonary, immobility, psychological impacts and other medical complications are highly motivated to work with the team to achieve a safe discharge. The US Department of Veterans Affairs (VA) Rehabilitation Services has much to offer them.
Heidi Maloni, PhD, NP: Veterans with MS are not at greater risk because they are diagnosed with MS. But, their comorbidities such as hypertension, obesity, or factors such as older age and increased disability can increase the risk of COVID-19 infection and poorer outcomes if infected. might place them at greater risk.
Veterans have asked “Am I at greater risk? Do I need to do something more to protect myself?” I have had innumerable veterans call and ask whether I can write them letters for their employer to ensure that they work at home longer rather than go into the workplace because they’re very nervous and don’t feel confident that masking and distancing is really going to be protective.
Mitchell Wallin: We are analyzing some of our data in the VA health care system related to COVID-19 infections in the MS population. We can’t say for sure what are numbers are, but our rates of infection and hospitalization are higher than the general population and we will soon have a report. We have a majority male population, which is different from the general MS population, which is predominantly female. The proportion of minority patients in VA mirrors those of the US population. These demographic factors along with a high level of comorbid disease put veterans at high risk for acquiring COVID-19. So, in some ways it’s hard to compare when you look at reports from other countries or the US National MS-COVID-19 Registry, which captures a population that is predominantly female. In the VA, our age range spans from the 20s to almost 100 years. We must understand our population to prevent COVID-19 and better care for the most vulnerable.
Rebecca Spain: Heidi, my understanding, although the numbers are small, that for the most part, Veterans with MS who are older are at higher risk of complications and death, which is also true of the general population. But that there is an additional risk for people with MS who have higher disability levels. My understanding from reading the literature, was that people with MS needing or requiring a cane to walk or greater assistance for mobility were at a higher risk for COVID-19 complications, including mortality. I have been particularly encouraged that in many places this special population of people with MS are getting vaccinated sooner.
Heidi Maloni: I completely agree, you said it very clearly, Becca. Their disability level puts them at risk
Rebecca Spain: Disability is a comorbidity.
Heidi Maloni: Yes. Just sitting in a wheelchair and not being able to get a full breath or having problems with respiratory effort really does put you at risk for doing well if you were to have COVID-19.
Are there other ancillary impacts from COVID-19 for patients with MS?
Jodie Haselkorn: Individuals who are hospitalized with COVID-19 miss social touch and social support from family and friends. They miss familiar conversations, a hug and having someone hold their hand. The acute phase of the infection limits professional face-to-face interaction with patients due to time and protective garments. There are reports of negative consequences with isolation and social reintegration of the COVID-19 survivors is necessary and a necessary part of rehabilitation.
Mitchell Wallin: For certain procedures (eg, magnetic resonance imaging [MRI]) or consultations, we need to bring people into the medical center. Many clinical encounters, however, can be done through telemedicine and both the VA and the US Department of Defense systems were set up to execute this type of visit. We had been doing telemedicine for a long time before the pandemic and we were in a better position than a lot of other health systems to shift to a virtual format with COVID-19. We had to ramp up a little bit and get our tools working a little more effectively for all clinics, but I think we were prepared to broadly execute telemedicine clinics for the pandemic.
Jodie Haselkorn: I agree that the he VA infrastructure was ahead of most other health system in terms of readiness for telehealth and maintaining access to care. Not all health care providers (HCPs) were using it, but the system was there, and included a telehealth coordinator in all of the facilities who could gear health care professionals up quickly. Additionally, a system was in place to provide veterans and caregivers with telehealth home equipment and provide training. Another thing that really helped was the MISSION Act. Veterans who have difficulty travelling for an appointment may have the ability to seek care outside of the VA within their own community. They may be able to go into a local facility to get laboratory or radiologic studies done or continue rehabilitation closer to home.
VA MS Registry Data
Rebecca Spain: Mitch, there are many interesting things we can learn about the interplay between COVID-19 and MS using registries such as how it affects people based on rural vs metropolitan living, whether people are living in single family homes or not as a proxy marker for social support, and so on.
Mitchell Wallin: We have both an MS registry to track and follow patients through our clinical network and a specific COVID-19 registry as well in VA. We have identified the MS cases infected with CoVID-19 and are putting them together.
Jodie Haselkorn: There are a number of efforts in mental health that are moving forward to examine depression and in anxiety during COVID-19. Individuals with MS have increased rates of depression and anxiety above that of the general population during usual times. The literature reports an increase in anxiety and depression in general population associated with the pandemic and veterans with MS seem to be reporting these symptoms more frequently as well. We will be able to track use the registry to assess the impacts of COVID-19 on depression and anxiety in Veterans with MS.
Providing MS Care During COVID-19
Jodie Haselkorn: The transition to telehealth in COVID-19 has been surprisingly seamless with some additional training for veterans and HCPs. I initially experienced an inefficiency in my clinic visit productivity. It took me longer to see a veteran because I wasn’t doing telehealth in our clinic with support staff and residents, my examination had to change, my documentation template needed to be restructured, and the coding was different. Sometimes I saw a veteran in clinic the and my next appointment required me to move back to my office in another building for a telehealth appointment. Teaching virtual trainees who also participated in the clinic encounters had its own challenges and rewards. My ‘motor routine’ was disrupted.
Rebecca Spain: There’s a real learning curve for telehealth in terms of how comfortable you feel with the data you get by telephone or video and how reliable that is. There are issues based on technology factors—like the patient’s bandwidth—because determining how smooth their motions are is challenging if you have a jerky, intermittent signal. I learned quickly to always do the physical examination first because I might lose video connection partway through and have to switch to a phone visit!
It’s still an open question, how much are we missing by using a video and not in-person visits. And what are the long-term health outcomes and implications of that? That is something that needs to be studied in neurology where we pride ourselves on the physical examination. When move to a virtual physical examination, is there cost? There are incredible gains using telehealth in terms of convenience and access to care, which may outweigh some of the drawbacks in particular cases.
There are also pandemic challenge in terms of clinic workflow. At VA Portland Health Care System in Oregon, I have 3 clinics for Friday morning: telephone, virtual, and face-to-face clinics. It’s a real struggle for the schedulers. And because of that transition to new system workflows to accommodate this, some patient visits have been dropped, lost, or scheduled incorrectly.
Heidi Maloni: As the nurse in this group, I agree with everything that Becca and Jodie have said about telehealth. But, I have found some benefits, and one of them is a greater intimacy with my patients. What do I mean by that? For instance, if a patient has taken me to their kitchen and opened their cupboard to show me the breakfast cereal, I’m also observing that there’s nothing else in that cupboard other than cereal. I’m also putting some things together about health and wellness. Or, for the first time, I might meet their significant other who can’t come to clinic because they’re working, but they are at home with the patient. And then having that 3-way conversation with the patient and the significant other, that’s kind of opened up my sense of who that person is.
You are right about the neurological examination. It’s challenging to make exacting assessments. When gathering household objects, ice bags and pronged forks to assess sensation, you remember that this exam is subjective and there is meaning in this remote evaluation. But all in all, I have been blessed with telehealth. Patients don’t mind it at all. They’re completely open to the idea. They like the telehealth for the contact they are able to have with their HCP.
Jodie Haselkorn: As you were saying that, Heidi, I thought, I’ve been inside my veterans’ bathrooms virtually and have seen all of their equipment that they have at home. In a face-to-face clinic visit, you don’t have an opportunity to see all their canes and walkers, braces, and other assistive technology. Some of it’s stashed in a closet, some of it under the bed. In a virtual visit, I get to understand why some is not used, what veterans prefer, and see their own innovations for mobility and self-care.
Mitchell Wallin: There’s a typical ritual that patients talk about when they go to a clinic. They check in, sit down, and wait for the nurse to give them their vital signs and set them up in the room. And then they meet with their HCP, and finally they complete the tasks on the checklist. And part of that may mean scheduling an MRI or going to the lab. But some of these handoffs don’t happen as well on telehealth. Maybe we haven’t integrated these segments of a clinical visit into telehealth platforms. But it could be developed, and there could be new neurologic tools to improve the interview and physical examination. Twenty years ago, you couldn’t deposit a check on your phone; but now you can do everything on your phone you could do in a physical bank. With some creativity, we can improve parts of the neurological exam that are currently difficult to assess remotely.
Jodie Haselkorn: I have not used peripherals in video telehealth to home and I would need to become accustomed to their use with current technology and train patients and caregivers. I would like telehealth peripherals such as a stethoscope to listen to the abdomen of a veteran with neurogenic bowel or a user-friendly ultrasound probe to measure postvoid residual urine in an individual with symptoms of neurogenic bladder, in addition to devices that measure walking speed and pulmonary function. I look forward to the development, use, and the incorporation peripherals that will enable a more extensive virtual exam within the home.
What are the MS Centers of Excellence working on now?
Jodie Haselkorn: We are working to understand the healthcare needs of veterans with MS by evaluating not only care for MS within the VA, but also the types and quantity of MS specialty care VA that is being received in the community during the pandemic. Dr. Wallin is also using the registry to lead a telehealth study to capture the variety of different codes that VA health professionals in MS have used to document workload by telehealth, and face-to-face, and telephone encounters.
Rebecca Spain: The MS Center of Excellence (MSCoE) is coming out with note templates to be available for HCPs, which we can refine as we get experience. This is s one way we can promote high standards in MS care by making these ancillary tools more productive.
Jodie Haselkorn: We are looking at different ways to achieve a high-quality virtual examination using standardized examination strategies and patient and caregiver information to prepare for a specialty MS visit.
Rebecca Spain: I would like to, in more of a research setting, study health outcomes using telehealth vs in person and start tracking that long term.
Mitchell Wallin: We can probably do more in terms of standardization, such as the routine patient reported surveys and implementing the new Consortium of Multiple Sclerosis Centers’ International MRI criteria. The COVID pandemic has affected everything in medical care. But we want to have a regular standardized outcome to assess, and if we can start to do some of the standard data collection through telemedicine, it becomes part of our regular clinic data.
Heidi Maloni: We need better technology. You can do electrocardiograms on your watch. Could we do Dinamaps? Could we figure out strength? That’s a wish list.
Jodie Haselkorn: Since the MSCoE is a national program, we were set up to do what we needed to do for education. We were able to continue on with all of our HCP webinars, including the series with the National MS Society (NMSS). We also have a Specialty Care Access Network-Extension for Community Healthcare Outcomes (SCAN-ECHO) series with the Northwest ECHO VA program and collaborated with the Can Do MS program on patient education as well. We’ve sent out 2 printed newsletters for veterans. The training of HCPs for the future has continued as well. All of our postdoctoral fellows who have finished their programs on time and moved on to either clinical practice or received career development grants to continue their VA careers, a new fellow has joined, and our other fellows are continuing as planned.
The loss that we sustained was in-person meetings. We held MSCoE Regional Program meetings in the East and West that combined education and administrative goals. Both of these were well attended and successful. There was a lot of virtual education available from multiple sources. It was challenging this year was to anticipate what education programming people wanted from MSCoE. Interestingly, a lot of our regional HCPs did not want much more COVID-19 education. They wanted other education and we were able to meet those needs.
Did the pandemic impact the VA MS registry?
Mitchell Wallin: Like any electronic product, the VA MS Surveillance Registry must be maintained, and we have tried to encourage people to use it. Our biggest concern was to identify cases of MS that got infected with COVID-19 and to put those people into the registry. In some cases, Veterans with MS were in locations without a MS clinic. So, we’ve spent a lot more time identifying those cases and adjudicating them to make sure their infection and MS were documented correctly.
During the COVID-19 pandemic, the VA healthcare system has been taxed like others and so HCPs have been a lot busier than normal, forcing new workflows. It has been a hard year that way because a lot of health care providers have been doing many other jobs to help maintain patient care during the COVID-19 pandemic.
Heidi Maloni: The impact of COVID-19 has been positive for the registry because we’ve had more opportunities to populate it.
Jodie Haselkorn: Dr. Wallin and the COVID-19 Registry group began building the combined registry at the onset of the pandemic. We have developed the capacity to identify COVID-19 infections in veterans who have MS and receive care in the VA. We entered these cases in the MS Surveillance Registry and have developed a linkage with the COVID-19 national VA registry. We are in the middle of the grunt work part case entry, but it is a rich resource.
How has the pandemic impacted MS research?
Rebecca Spain: COVID-19 has put a big damper on clinical research progress, including some of our MSCoE studies. It has been difficult to have subjects come in for clinical visits. It’s been difficult to get approval for new studies. It’s shifted timelines dramatically, and then that always increases budgets in a time when there’s not a lot of extra money. So, for clinical research, it’s been a real struggle and a strain and an ever-moving target. For laboratory research most, if not all, centers that have laboratory research at some point were closed and have only slowly reopened. Some still haven’t reopened to any kind of research or laboratory. So, it’s been tough, I think, on research in general.
Heidi Maloni: I would say the word is devastating. The pandemic essentially put a stop to in-person research studies. Our hospital was in research phase I, meaning human subjects can only participate in a research study if they are an inpatient or outpatient with an established clinic visit (clinics open to 25% occupancy) or involved in a study requiring safety monitoring, This plan limits risk of COVID-19 exposure.
Rebecca Spain: There is risk for a higher dropout rate of subjects from studies meaning there’s less chance of success for finding answers if enough people don’t stay in. At a certain point, you have to say, “Is this going to be a successful study?”
Jodie Haselkorn: Dr. Spain has done an amazing job leading a multisite, international clinical trial funded by the VA and the NMSS and kept it afloat, despite challenges. The pandemic has had impacts, but the study continues to move towards completion. I’ve appreciated the efforts of the Research Service at VA Puget Sound to ensure that we could safely obtain many of the 12-month outcomes for all the participants enrolled in that study.
Mitchell Wallin: The funding for some of our nonprofit partners, including the Paralyzed Veterans Association (PVA) and the NMSS, has suffered as well and so a lot of their funding programs have closed or been cut back during the pandemic. Despite that, we still have been able to use televideo technology for our clinical and educational programs with our network.
Jodie Haselkorn: MSCoE also does health services and epidemiological studies in addition to clinical trials and that work has continued. Quite a few of the studies that had human subjects in them were completed in terms of data collection, and so those are being analyzed. There will be a drop in funded studies, publications and posters as the pandemic continues and for a recovery period. We have a robust baseline for research productivity and a talented team. We’ll be able to track drop off and recovery over time.
Rebecca Spain: There’s going to be long-term consequences that we don’t see right now, especially for young researchers who have missed getting pilot data which would have led to additional small grants and then later large grants. There’s going to be an education gap that’s going on with all of the kids who are not able to go to school properly. It’s part of that whole swath of lost time and lost opportunity that we will have to deal with.
However, there are going to be some positive changes. We’re now busy designing clinical trials that can be done virtually to minimize any contact with the health facility, and then looking at things like shifting to research ideas that are more focused around health services.
Jodie Haselkorn: Given the current impacts of the pandemic on delivery of health care there is a strong interest in looking at how we can deliver health care in ways that accommodates the consumers and the providers perspectives. In the future we see marked impacts in our abilities to deliver care to Veterans with MS.
As a final thought, I wanted to put in a plug for this talented team. One of our pandemic resolutions was to innovatively find new possibilities and avoid negative focus on small changes. We are fortunate that all our staff have remained healthy and been supportive and compassionate with each other throughout this period. We have met our goals and are still moving forward.
MSCoE has benefited from the supportive leadership of Sharyl Martini, MD, PhD, and Glenn Graham, MD, PhD, in VA Specialty Care Neurology and leadership and space from VA Puget Sound, VA Portland Health Care System, the Washington DC VA Medical Center and VA Maryland Health Care System in Baltimore.
We also have a national advisory system that is actively involved, sets high standards and performs a rigorous annual review. We have rich inputs from the VA National Regional Programs and Veterans. Additionally, we have had the leadership and opportunities to collaborate with outside organizations including, the Consortium of MS Centers, the NMSS, and the PVA. We have been fortunate.
Small increase seen in new COVID-19 cases among children
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
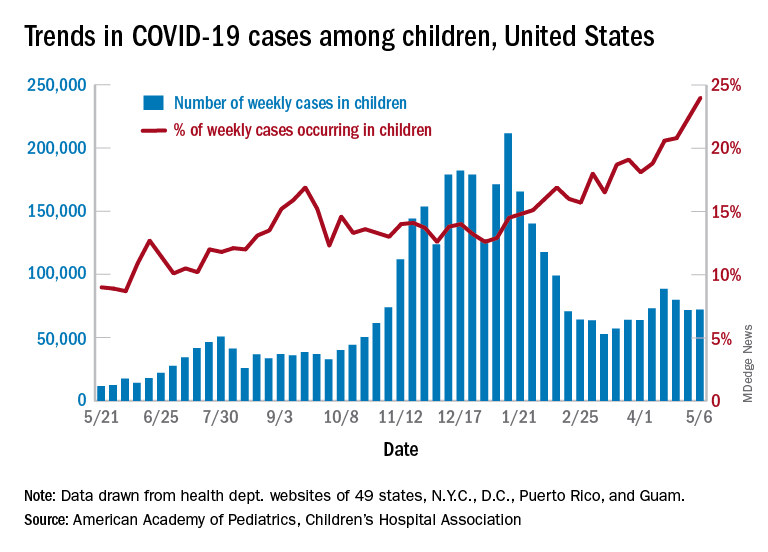
higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
After 2 consecutive weeks of declines, the number of new COVID-19 cases in children rose slightly, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

higher than at any other time during the pandemic, the AAP and CHA data show.
It is worth noting, however, that Rhode Island experienced a 30% increase in the last week, adding about 4,900 cases because of data revision and a lag in reporting, the AAP and CHA said in their weekly COVID-19 report.
All the new cases bring the total national count to just over 3.54 million in children, which represents 14.0% of all cases in 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam. The cumulative case rate as of May 6 was 5,122 per 100,000 children, the two organizations said.
All the new cases that were added to Rhode Island’s total give it the highest cumulative rate in the country: 9,614 cases per 100,000 children. North Dakota is right behind with 9,526 per 100,000, followed by Tennessee (8,898), Connecticut (8,281), and South Carolina (8,274). Vermont has the highest proportion of cases in children at 22.4%, with Alaska next at 20.3% and South Carolina third at 18.7%, according to the AAP and CHA.
Hawaii just reported its first COVID-19–related death in a child, which drops the number of states with zero deaths in children from 10 to 9. Two other new deaths in children from April 30 to May 6 bring the total number to 306 in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting the age distribution of deaths.
In a separate statement, AAP president Lee Savio Beers acknowledged the Food and Drug Administration’s authorization of the Pfizer-BioNTech vaccine for children aged 12-15 years as “a critically important step in bringing lifesaving vaccines to children and adolescents. ... We look forward to the discussion by the Advisory Committee on Immunization Practices of the CDC, which will make recommendations about the use of this vaccine in adolescents.”
Dr. Fauci: Feds may ease indoor mask mandates soon
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.
Federal guidance on indoor mask use may change soon, Anthony S. Fauci, MD, director of the National Institute of Allergy and Infectious Diseases, said on May 9.
He was asked whether it’s time to start relaxing indoor mask requirements.
“I think so, and I think you’re going to probably be seeing that as we go along and as more people get vaccinated,” Dr. Fauci said on ABC News’s This Week.Nearly 150 million adults in the United States – or about 58% of the adult population – have received at least one COVID-19 vaccine dose, according to the latest CDC tally. About 113 million adults, or 44%, are considered fully vaccinated.
“The CDC will be, you know, almost in real time … updating their recommendations and their guidelines,” Dr. Fauci said.
In April, the CDC relaxed its guidance for those who have been vaccinated against COVID-19. Those who have gotten a shot don’t need to wear a mask outdoors or in small indoor gatherings with other vaccinated people, but both vaccinated and unvaccinated people are still advised to wear masks in indoor public spaces.
“We do need to start being more liberal as we get more people vaccinated,” Dr. Fauci said. “As you get more people vaccinated, the number of cases per day will absolutely go down.”
The United States is averaging about 43,000 cases per day, he said, adding that the cases need to be “much, much lower.” When the case numbers drop and vaccination numbers increase, the risk of infection will fall dramatically indoors and outdoors, he said.
Even after the pandemic, though, wearing masks could become a seasonal habit, Dr. Fauci said May 9 on NBC News’s Meet the Press.“I think people have gotten used to the fact that wearing masks, clearly if you look at the data, it diminishes respiratory diseases. We’ve had practically a nonexistent flu season this year,” he said.
“So it is conceivable that as we go on, a year or 2 or more from now, that during certain seasonal periods when you have respiratory-borne viruses like the flu, people might actually elect to wear masks to diminish the likelihood that you’ll spread these respiratory-borne diseases,” he said.
Dr. Fauci was asked about indoor mask guidelines on May 9 after former FDA Commissioner Scott Gottlieb, MD, said face mask requirements should be relaxed.
“Certainly outdoors, we shouldn’t be putting limits on gatherings anymore,” Dr. Gottlieb said on CBS News’s Face the Nation.“The states where prevalence is low, vaccination rates are high, we have good testing in place, and we’re identifying infections, I think we could start lifting these restrictions indoors as well, on a broad basis,” he said.
Lifting pandemic-related restrictions in areas where they’re no longer necessary could also encourage people to implement them again if cases increase during future surges, such as this fall or winter, Dr. Gottlieb said.
At the same time, Americans should continue to follow CDC guidance and wait for new guidelines before changing their indoor mask use, Jeffrey Zients, the White House COVID-19 response coordinator, said on CNN’s State of the Union on May 9.
“We all want to get back to a normal lifestyle,” he said. “I think we’re on the path to do that, but stay disciplined, and let’s take advantage of the new privilege of being vaccinated and not wearing masks outdoors, for example, unless you’re in a crowded place.”
Mr. Zients pointed to President Joe Biden’s goal for 70% of adults to receive at least one vaccine dose by July 4.
“As we all move toward that 70% goal, there will be more and more advantages to being vaccinated,” he said. “And if you’re not vaccinated, you’re not protected.”
A version of this article first appeared on WebMD.com.
FDA authorizes Pfizer COVID vaccine for teens 12-15
The Food and Drug Administration on May 10 granted emergency use authorization (EUA) for the Pfizer coronavirus vaccine to be given to children 12-15 years old.
The much-expected decision increases the likelihood that schools in the United States will fully reopen in the fall – a goal of both the Biden and Trump administrations.
Acting FDA Commissioner Janet Woodcock, MD, called the decision “a significant step” in “returning to a sense of normalcy.”
“Today’s action allows for a younger population to be protected from COVID-19, bringing us closer to returning to a sense of normalcy and to ending the pandemic,” she said in a statement. “Parents and guardians can rest assured that the agency undertook a rigorous and thorough review of all available data, as we have with all of our COVID-19 vaccine emergency use authorizations.”
The Pfizer adolescent vaccine is not yet a done deal, though.
Next, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will decide on May 12 whether to recommend use of the vaccine in this age group. After that, CDC Director Rochelle Walensky, MD, will decide whether to give the green light for the vaccine to be administered to that age group.
The FDA action on May 10 amends the Dec. 11, 2020, emergency use authorization that allowed the Pfizer vaccine to be given to people 16 and older. Pfizer was the first company to receive an EUA for its adult vaccine and is the first to receive authorization for its adolescent vaccine. Pfizer is conducting clinical trials on much younger children, too.
The Moderna and Johnson & Johnson vaccines are authorized for people 18 and up. Moderna also has launched clinical trials in children.
Most health experts have said the United States needs to vaccinate children before the COVID-19 pandemic can truly be brought under control. The 12- to 15-year-old group represents 17 million people, about 5% of the population. Thus far, 58% of U.S. adults have had at least one dose of a vaccine and 34.8% of all Americans are fully vaccinated.
American Academy of Pediatrics President Lee Savio Beers, MD, praised the agency’s decision, calling it a “critically important step in bringing life-saving vaccines to children and adolescents. Our youngest generations have shouldered heavy burdens over the past year, and the vaccine is a hopeful sign that they will be able to begin to experience all the activities that are so important for their health and development.”
President Joe Biden recently announced a new strategy for expanding vaccinations in which vaccinating 12- to 15-year-olds was a key component. He said the administration was ready to ship the adolescent vaccine directly to pharmacies and pediatricians to speed up the vaccination rate.
In March, Anthony S. Fauci, MD, told a Senate committee, “We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape. … We ultimately would like to get and have to get children into that mix.”
Pfizer submitted data to the FDA in late March showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12-15 in clinical trials.
Though most children have milder symptoms when infected with the coronavirus, about 1.5 million cases in children aged 11-17 were reported to the CDC between March 1, 2020, and April 30 of this year, the FDA news release said.
Albert Bourla, CEO of Pfizer, tweeted that “today brings very encouraging news for families and adolescents across the United States.
“While this is a meaningful step forward, we are still in a critical period of combating #COVID19 around the world. In the coming weeks, we hope to continue to receive authorizations from global regulators to support worldwide vaccination efforts,” he said.
“It’s essential for children to be vaccinated against COVID-19. According to data compiled by the AAP and Children’s Hospital Association, more than 3.8 million children have tested positive for COVID-19 in the United States since the start of the pandemic,” said Dr. Savio Beers. “While fewer children than adults have suffered the most severe disease, this is not a benign disease in children. Thousands of children have been hospitalized, and hundreds have died. We will soon have a very safe, highly effective vaccine that can prevent so much suffering. I encourage parents to talk with their pediatricians about how to get the vaccine for their adolescents as soon as they are eligible.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration on May 10 granted emergency use authorization (EUA) for the Pfizer coronavirus vaccine to be given to children 12-15 years old.
The much-expected decision increases the likelihood that schools in the United States will fully reopen in the fall – a goal of both the Biden and Trump administrations.
Acting FDA Commissioner Janet Woodcock, MD, called the decision “a significant step” in “returning to a sense of normalcy.”
“Today’s action allows for a younger population to be protected from COVID-19, bringing us closer to returning to a sense of normalcy and to ending the pandemic,” she said in a statement. “Parents and guardians can rest assured that the agency undertook a rigorous and thorough review of all available data, as we have with all of our COVID-19 vaccine emergency use authorizations.”
The Pfizer adolescent vaccine is not yet a done deal, though.
Next, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will decide on May 12 whether to recommend use of the vaccine in this age group. After that, CDC Director Rochelle Walensky, MD, will decide whether to give the green light for the vaccine to be administered to that age group.
The FDA action on May 10 amends the Dec. 11, 2020, emergency use authorization that allowed the Pfizer vaccine to be given to people 16 and older. Pfizer was the first company to receive an EUA for its adult vaccine and is the first to receive authorization for its adolescent vaccine. Pfizer is conducting clinical trials on much younger children, too.
The Moderna and Johnson & Johnson vaccines are authorized for people 18 and up. Moderna also has launched clinical trials in children.
Most health experts have said the United States needs to vaccinate children before the COVID-19 pandemic can truly be brought under control. The 12- to 15-year-old group represents 17 million people, about 5% of the population. Thus far, 58% of U.S. adults have had at least one dose of a vaccine and 34.8% of all Americans are fully vaccinated.
American Academy of Pediatrics President Lee Savio Beers, MD, praised the agency’s decision, calling it a “critically important step in bringing life-saving vaccines to children and adolescents. Our youngest generations have shouldered heavy burdens over the past year, and the vaccine is a hopeful sign that they will be able to begin to experience all the activities that are so important for their health and development.”
President Joe Biden recently announced a new strategy for expanding vaccinations in which vaccinating 12- to 15-year-olds was a key component. He said the administration was ready to ship the adolescent vaccine directly to pharmacies and pediatricians to speed up the vaccination rate.
In March, Anthony S. Fauci, MD, told a Senate committee, “We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape. … We ultimately would like to get and have to get children into that mix.”
Pfizer submitted data to the FDA in late March showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12-15 in clinical trials.
Though most children have milder symptoms when infected with the coronavirus, about 1.5 million cases in children aged 11-17 were reported to the CDC between March 1, 2020, and April 30 of this year, the FDA news release said.
Albert Bourla, CEO of Pfizer, tweeted that “today brings very encouraging news for families and adolescents across the United States.
“While this is a meaningful step forward, we are still in a critical period of combating #COVID19 around the world. In the coming weeks, we hope to continue to receive authorizations from global regulators to support worldwide vaccination efforts,” he said.
“It’s essential for children to be vaccinated against COVID-19. According to data compiled by the AAP and Children’s Hospital Association, more than 3.8 million children have tested positive for COVID-19 in the United States since the start of the pandemic,” said Dr. Savio Beers. “While fewer children than adults have suffered the most severe disease, this is not a benign disease in children. Thousands of children have been hospitalized, and hundreds have died. We will soon have a very safe, highly effective vaccine that can prevent so much suffering. I encourage parents to talk with their pediatricians about how to get the vaccine for their adolescents as soon as they are eligible.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration on May 10 granted emergency use authorization (EUA) for the Pfizer coronavirus vaccine to be given to children 12-15 years old.
The much-expected decision increases the likelihood that schools in the United States will fully reopen in the fall – a goal of both the Biden and Trump administrations.
Acting FDA Commissioner Janet Woodcock, MD, called the decision “a significant step” in “returning to a sense of normalcy.”
“Today’s action allows for a younger population to be protected from COVID-19, bringing us closer to returning to a sense of normalcy and to ending the pandemic,” she said in a statement. “Parents and guardians can rest assured that the agency undertook a rigorous and thorough review of all available data, as we have with all of our COVID-19 vaccine emergency use authorizations.”
The Pfizer adolescent vaccine is not yet a done deal, though.
Next, the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices will decide on May 12 whether to recommend use of the vaccine in this age group. After that, CDC Director Rochelle Walensky, MD, will decide whether to give the green light for the vaccine to be administered to that age group.
The FDA action on May 10 amends the Dec. 11, 2020, emergency use authorization that allowed the Pfizer vaccine to be given to people 16 and older. Pfizer was the first company to receive an EUA for its adult vaccine and is the first to receive authorization for its adolescent vaccine. Pfizer is conducting clinical trials on much younger children, too.
The Moderna and Johnson & Johnson vaccines are authorized for people 18 and up. Moderna also has launched clinical trials in children.
Most health experts have said the United States needs to vaccinate children before the COVID-19 pandemic can truly be brought under control. The 12- to 15-year-old group represents 17 million people, about 5% of the population. Thus far, 58% of U.S. adults have had at least one dose of a vaccine and 34.8% of all Americans are fully vaccinated.
American Academy of Pediatrics President Lee Savio Beers, MD, praised the agency’s decision, calling it a “critically important step in bringing life-saving vaccines to children and adolescents. Our youngest generations have shouldered heavy burdens over the past year, and the vaccine is a hopeful sign that they will be able to begin to experience all the activities that are so important for their health and development.”
President Joe Biden recently announced a new strategy for expanding vaccinations in which vaccinating 12- to 15-year-olds was a key component. He said the administration was ready to ship the adolescent vaccine directly to pharmacies and pediatricians to speed up the vaccination rate.
In March, Anthony S. Fauci, MD, told a Senate committee, “We don’t really know what that magical point of herd immunity is, but we do know that if we get the overwhelming population vaccinated, we’re going to be in good shape. … We ultimately would like to get and have to get children into that mix.”
Pfizer submitted data to the FDA in late March showing its mRNA vaccine was 100% effective at preventing COVID-19 infection in children ages 12-15 in clinical trials.
Though most children have milder symptoms when infected with the coronavirus, about 1.5 million cases in children aged 11-17 were reported to the CDC between March 1, 2020, and April 30 of this year, the FDA news release said.
Albert Bourla, CEO of Pfizer, tweeted that “today brings very encouraging news for families and adolescents across the United States.
“While this is a meaningful step forward, we are still in a critical period of combating #COVID19 around the world. In the coming weeks, we hope to continue to receive authorizations from global regulators to support worldwide vaccination efforts,” he said.
“It’s essential for children to be vaccinated against COVID-19. According to data compiled by the AAP and Children’s Hospital Association, more than 3.8 million children have tested positive for COVID-19 in the United States since the start of the pandemic,” said Dr. Savio Beers. “While fewer children than adults have suffered the most severe disease, this is not a benign disease in children. Thousands of children have been hospitalized, and hundreds have died. We will soon have a very safe, highly effective vaccine that can prevent so much suffering. I encourage parents to talk with their pediatricians about how to get the vaccine for their adolescents as soon as they are eligible.”
A version of this article first appeared on Medscape.com.
NSAIDs don’t make COVID-19 worse in hospitalized patients
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
NSAIDs don’t boost the risk of more severe disease or death in hospitalized patients with COVID-19, a new study finds.
“To our knowledge, our prospective study includes the largest number of patients admitted to hospital with COVID-19 to date, and adds to the literature on the safety of NSAIDs and in-hospital outcomes. NSAIDs do not appear to increase the risk of worse in-hospital outcomes ...” the study authors wrote. “NSAIDs are an important analgesic modality and have a vital opioid-sparing role in pain management. Patients and clinicians should be reassured by these findings that NSAIDs are safe in the context of the pandemic.”
The report was published online May 7 in The Lancet Rheumatology and led by clinical research fellow Thomas M. Drake, MBChB, of the University of Edinburgh’s Usher Institute.
For more than a year, researchers worldwide have debated about whether NSAIDs spell trouble for people at risk of COVID-19. In March 2020, French health officials announced that use of the painkillers such as NSAIDs may increase the severity of the disease, and they recommended that patients take acetaminophen instead. The National Health Service in the United Kingdom made a similar recommendation. But other agencies didn’t believe there was enough evidence to support ditching NSAIDs, and recent research studies published in Annals of the Rheumatic Diseases and PLoS Medicine suggested they may be right.
For the new study, researchers identified 72,179 patients who were treated for COVID-19 in British hospitals during January-August 2020. About 56% were men, 74% were White, and 6% took NSAIDs on a regular basis before they entered the hospital. The average age was 70.
The researchers examined whether the patients in either group were more or less likely to die in the hospital, be admitted into a critical care unit, need oxygen treatment, need a ventilator, or suffer kidney injury.
In terms of outcomes, there weren’t any major gaps between the groups overall. The differences in most comparisons were statistically insignificant. For example, 31% of those who didn’t take NSAIDs died vs. 30% of those who did (P = .227). In both groups, 14% required critical care admission (P = .476).
The researchers then focused on two matched groups of 4,205 patients: One group used NSAIDs regularly, and the other group didn’t. The difference in risk of death in those who took NSAIDs vs. those who didn’t was statistically insignificant (odds ratio, 0.95; 95% confidence interval, 0.84-1.07; P = .35). Other comparisons were also statistically insignificant.
The findings offer insight into whether the use of NSAIDs might actually be helpful for patients who develop COVID-19. Scientists believe that COVID-19 is linked to inflammation in the body, and NSAIDs, of course, reduce inflammation. But the researchers didn’t turn up any sign of a benefit.
The new study has some weaknesses: It doesn’t say anything about whether NSAIDs have an impact on whether people get COVID-19 in the first place. Researchers don’t know if high use of NSAIDs may affect the severity of the disease. And it doesn’t examine the potential effect of acetaminophen, although other research suggests the drug also may not cause harm in patients with COVID-19.
Still, the researchers say the study is the largest of its kind to look at the use of NSAIDs by patients who are admitted to the hospital with COVID-19. “Considering all the evidence, if there was an extreme effect of NSAIDs on COVID-19 outcomes or severity, this would have been observed in one or more of the studies that have been done, including the present study,” they wrote.
In a commentary that accompanied the study, three physicians from hospitals in Denmark, led by Kristian Kragholm, MD, of Aalborg University Hospital, praised the research and wrote that it adds to “a growing body of evidence” that NSAIDs don’t make things worse for patients with COVID-19.
The study was funded by the U.K. National Institute for Health Research and the U.K. Medical Research Council. The study and commentary authors reported no relevant disclosures.
FROM THE LANCET RHEUMATOLOGY
Recommendations for Pregnant Members of Dermatology Health Care Teams During the COVID-19 Pandemic
Information is scarce regarding the impact of COVID-19 on pregnant women and newborns; health care workers (HCWs), particularly pregnant women,1 who are caring for patients during the pandemic might experience concern and uncertainty. The American College of Obstetricians and Gynecologists (ACOG) released recommendations, based on expert consensus, regarding pregnant HCWs on December 14, 2020.2 We propose an appropriation of the ACOG recommendations for dermatologists and their teams caring for patients during the COVID-19 pandemic.
Risks to Pregnant HCWs
Worldwide, viral pneumonia is a leading cause of death during pregnancy,3 with higher mortality documented among pregnant patients during the 1918 influenza pandemic and the 2003 severe acute respiratory syndrome–associated coronavirus pandemic,3 and an increased rate of hospital admission documented among these patients compared to the general population during the 2009 H1N1 influenza pandemic.4
Data from the Centers for Disease Control and Prevention (CDC) suggest that pregnant women with symptomatic COVID-19 (n=30,415) are at increased risk for the following (compared to nonpregnant women with symptomatic COVID-19 [n=431,410])5:
• Admission to the intensive care unit (10.5 of every 1000 cases vs 3.9 of every 1000 cases; adjusted risk ratio [aRR]=3.0; 95% CI, 2.6-3.4)
• Receipt of invasive ventilation (2.9 of every 1000 cases vs 1.1 of every 1000 cases; aRR=2.9; 95% CI, 2.2-3.8)
• Receipt of extracorporeal membrane oxygenation (0.7 of every 1000 cases vs 0.3 of every 1000 cases; aRR=2.4; 95% CI, 1.5-4.0)
• Death (1.5 of every 1000 cases vs 1.2 of every 1000 cases; aRR=1.7; 95% CI, 1.2-2.4).
Although the absolute risk of severe COVID-19–related outcomes is low, the CDC includes pregnant women in its increased risk category for COVID-19. Furthermore, in a systematic review of 61 studies comprising 790 COVID-19–positive pregnant women and 548 newborns, the rates of cesarean delivery, premature birth, low birth weight, and adverse pregnancy events (the latter comprising preterm birth, death or stillbirth, and early termination of pregnancy) were estimated to be 72%, 23%, 7%, and 27%, respectively.6 In a systematic review of 39 studies (case series and cohort studies), comprising 936 SARS-CoV-2–tested newborns of mothers with COVID-19, mother-to-fetus transmission of SARS-CoV-2 occurred during the third trimester in approximately 3.2% of infected mothers.7
In pregnant women with COVID-19 who develop cytokine storm syndrome, a fetal inflammatory response syndrome can ensue, which has been shown to cause ventricular expansion and bleeding in animal models.8 In addition, underlying conditions, such as cardiovascular disease, diabetes mellitus, pre-existing lung disease, and obesity, which are well-established risks factors for severe COVID-19 in nonpregnant patients, can increase the severity of COVID-19 in pregnant women.5,9-11
Recommendations From ACOG for Pregnant HCWs
The American College of Obstetricians and Gynecologists recommends that health care facilities consider limiting the exposure of pregnant HCWs to patients with confirmed or suspected COVID-19. They also recommend that pregnant women continue to work in patient-facing roles if they want to, if recommended personal protective equipment (PPE) is available for them to wear.2 The US Food and Drug Administration issued an Emergency Use Authorization for 2 messenger RNA COVID-19 vaccines. Although these vaccines have not been tested in pregnant women, ACOG recommends that COVID-19 vaccines not be withheld from pregnant women who fulfill the criteria for vaccination; pregnant women who decline vaccination should be supported in their decision.12 In dermatology, telemedicine is an effective alternative to face-to-face visits, reducing the risk of transmitting SARS-CoV-2 to physicians and patients.
Ideally, pregnant dermatology attending physicians and residents can continue to provide care through teledermatology. They also can continue to provide in-person care, if they choose to; however, higher-risk procedures should be avoided.12 In dermatology, that might include ablative laser procedures to the face, prolonged surgery, such as hair transplantation, and intraoral or intranasal procedures. Alternatively, pregnant dermatology residents can be allocated to clinical rotations in which face-to-face contact with patients is not required such as dermatopathology and a research rotation. Likewise, telework options can be encouraged for other pregnant members of dermatology teams, including front-desk staff, nurses, medical assistants, and remaining ancillary staff.
Guidance on Face Masks for Pregnant HCWs
Universal masking of HCWs has been shown to reduce the rate of health care–related acquisition of SARS-CoV-2.13 However, extended use or reuse of N95 respirators might contribute to SARS-CoV-2 transmission.14 The American College of Obstetricians and Gynecologists recommends that all HCWs wear a face mask at all times while working in a health care facility, even if patients are wearing a face covering or face mask.2 Based on CDC guidelines,15 HCWs in regions where community transmission is moderate or substantial should wear eye protection in addition to a face mask, and they should wear an N95, N95-equivalent, or higher-level respirator instead of a face mask when performing aerosol-generating procedures and surgical procedures. If working in a patient-facing role caring for patients with suspected or confirmed COVID-19, HCWs should wear an N95, N95-equivalent, or higher-level respirator; gown; gloves; and eye protection (goggles or a disposable face shield).15
Final Thoughts
COVID-19 has brought about acute and likely permanent changes to the US health care system. Dermatologists are integral members of that system and are essential to the treatment of patients with skin, hair, and nail disorders. Pregnant dermatologists and residents should refrain from patient-facing roles when feasible; however, when all recommended PPE are available, they may continue to work in patient-facing roles until they give birth if they desire to do so. Alternatively, teledermatology and non–face-to-face rotations should be encouraged. Higher-risk and aerosol-generating procedures are of particular concern regarding the risk for transmitting SARS-CoV-2 and should be avoided. Correct and universal use of PPE is paramount; when all recommended PPE is not available, pregnant HCWs should avoid exposure to patients with suspected or confirmed COVID-19. These recommendations will help safeguard pregnant members of dermatology teams during the COVID-19 pandemic while maximizing patient care.
- Rashidi Fakari F, Simbar M. Coronavirus pandemic and worries during pregnancy; a letter to editor. Arch Acad Emerg Med. 2020;8:E21.
- The American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, obstetrics. 2020. Accessed April 21, 2021. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics
- Schwartz DA, Graham AL. Potential maternal and infant outcomes from (Wuhan) coronavirus 2019-nCoV infecting pregnant women: lessons from SARS, MERS, and other human coronavirus infections. Viruses. 2020;12:194. doi:10.3390/v12020194
- Yan J, Guo J, Fan C, et al. Coronavirus disease 2019 in pregnant women: a report based on 116 cases. Am J Obstet Gynecol. 2020;223:111.e1-111.e14. doi:10.1016/j.ajog.2020.04.014
- Zambrano LD, Ellington S, Strid P, et al; . Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3
- Dubey P, Reddy SY, Manuel S, et al. Maternal and neonatal characteristics and outcomes among COVID-19 infected women: an updated systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;252:490-501. doi:10.1016/j.ejogrb.2020.07.034
- Kotlyar AM, Grechukhina O, Chen A, et al. Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis. Am J Obstet Gynecol. 2020;224:35-53.e3. doi:10.1016/j.ajog.2020.07.049
- Mitchell T, MacDonald JW, Srinouanpranchanh S, et al. Evidence of cardiac involvement in the fetal inflammatory response syndrome: disruption of gene networks programming cardiac development in nonhuman primates. Am J Obstet Gynecol. 2018;218:438.e1-438.e16. doi:10.1016/j.ajog.2018.01.009
- Ellington S, Strid P, Tong VT, et al. Characteristics of women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–June 7, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:769-775. doi:10.15585/mmwr.mm6925a1
- Panagiotakopoulos L, Myers TR, Gee J, et al. SARS-CoV-2 infection among hospitalized pregnant women: reasons for admission and pregnancy characteristics—eight U.S. health care centers, March 1–May 30, 2020. 2020. doi:10.15585/mmwr.mm6938e2
- Knight M, Bunch K, Vousden N, et al; . Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107. doi:10.1136/bmj.m2107
- The American College of Obstetricians and Gynecologists. Vaccinating pregnant and lactating patients against COVID-19. December 2020. Updated March 24, 2021. Accessed April 28, 2021. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/vaccinating-pregnant-and-lactating-patients-against-covid-19
- Seidelman JL, Lewis SS, Advani SD, et al. Universal masking is an effective strategy to flatten the severe acute respiratory coronavirus virus 2 (SARS-CoV-2) healthcare worker epidemiologic curve. Infect Control Hosp Epidemiol. 2020;41:1466-1467. doi:10.1017/ice.2020.31314.
- Degesys NF, Wang RC, Kwan E, et al. Correlation between N95 extended use and reuse and fit failure in an emergency department. JAMA. 2020;324:94-96. doi:10.1001/jama.2020.9843
- Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic 2020. Updated February 23, 2021. Accessed April 21, 2021. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Finfection-control%2Fcontrol-recommendations.html
Information is scarce regarding the impact of COVID-19 on pregnant women and newborns; health care workers (HCWs), particularly pregnant women,1 who are caring for patients during the pandemic might experience concern and uncertainty. The American College of Obstetricians and Gynecologists (ACOG) released recommendations, based on expert consensus, regarding pregnant HCWs on December 14, 2020.2 We propose an appropriation of the ACOG recommendations for dermatologists and their teams caring for patients during the COVID-19 pandemic.
Risks to Pregnant HCWs
Worldwide, viral pneumonia is a leading cause of death during pregnancy,3 with higher mortality documented among pregnant patients during the 1918 influenza pandemic and the 2003 severe acute respiratory syndrome–associated coronavirus pandemic,3 and an increased rate of hospital admission documented among these patients compared to the general population during the 2009 H1N1 influenza pandemic.4
Data from the Centers for Disease Control and Prevention (CDC) suggest that pregnant women with symptomatic COVID-19 (n=30,415) are at increased risk for the following (compared to nonpregnant women with symptomatic COVID-19 [n=431,410])5:
• Admission to the intensive care unit (10.5 of every 1000 cases vs 3.9 of every 1000 cases; adjusted risk ratio [aRR]=3.0; 95% CI, 2.6-3.4)
• Receipt of invasive ventilation (2.9 of every 1000 cases vs 1.1 of every 1000 cases; aRR=2.9; 95% CI, 2.2-3.8)
• Receipt of extracorporeal membrane oxygenation (0.7 of every 1000 cases vs 0.3 of every 1000 cases; aRR=2.4; 95% CI, 1.5-4.0)
• Death (1.5 of every 1000 cases vs 1.2 of every 1000 cases; aRR=1.7; 95% CI, 1.2-2.4).
Although the absolute risk of severe COVID-19–related outcomes is low, the CDC includes pregnant women in its increased risk category for COVID-19. Furthermore, in a systematic review of 61 studies comprising 790 COVID-19–positive pregnant women and 548 newborns, the rates of cesarean delivery, premature birth, low birth weight, and adverse pregnancy events (the latter comprising preterm birth, death or stillbirth, and early termination of pregnancy) were estimated to be 72%, 23%, 7%, and 27%, respectively.6 In a systematic review of 39 studies (case series and cohort studies), comprising 936 SARS-CoV-2–tested newborns of mothers with COVID-19, mother-to-fetus transmission of SARS-CoV-2 occurred during the third trimester in approximately 3.2% of infected mothers.7
In pregnant women with COVID-19 who develop cytokine storm syndrome, a fetal inflammatory response syndrome can ensue, which has been shown to cause ventricular expansion and bleeding in animal models.8 In addition, underlying conditions, such as cardiovascular disease, diabetes mellitus, pre-existing lung disease, and obesity, which are well-established risks factors for severe COVID-19 in nonpregnant patients, can increase the severity of COVID-19 in pregnant women.5,9-11
Recommendations From ACOG for Pregnant HCWs
The American College of Obstetricians and Gynecologists recommends that health care facilities consider limiting the exposure of pregnant HCWs to patients with confirmed or suspected COVID-19. They also recommend that pregnant women continue to work in patient-facing roles if they want to, if recommended personal protective equipment (PPE) is available for them to wear.2 The US Food and Drug Administration issued an Emergency Use Authorization for 2 messenger RNA COVID-19 vaccines. Although these vaccines have not been tested in pregnant women, ACOG recommends that COVID-19 vaccines not be withheld from pregnant women who fulfill the criteria for vaccination; pregnant women who decline vaccination should be supported in their decision.12 In dermatology, telemedicine is an effective alternative to face-to-face visits, reducing the risk of transmitting SARS-CoV-2 to physicians and patients.
Ideally, pregnant dermatology attending physicians and residents can continue to provide care through teledermatology. They also can continue to provide in-person care, if they choose to; however, higher-risk procedures should be avoided.12 In dermatology, that might include ablative laser procedures to the face, prolonged surgery, such as hair transplantation, and intraoral or intranasal procedures. Alternatively, pregnant dermatology residents can be allocated to clinical rotations in which face-to-face contact with patients is not required such as dermatopathology and a research rotation. Likewise, telework options can be encouraged for other pregnant members of dermatology teams, including front-desk staff, nurses, medical assistants, and remaining ancillary staff.
Guidance on Face Masks for Pregnant HCWs
Universal masking of HCWs has been shown to reduce the rate of health care–related acquisition of SARS-CoV-2.13 However, extended use or reuse of N95 respirators might contribute to SARS-CoV-2 transmission.14 The American College of Obstetricians and Gynecologists recommends that all HCWs wear a face mask at all times while working in a health care facility, even if patients are wearing a face covering or face mask.2 Based on CDC guidelines,15 HCWs in regions where community transmission is moderate or substantial should wear eye protection in addition to a face mask, and they should wear an N95, N95-equivalent, or higher-level respirator instead of a face mask when performing aerosol-generating procedures and surgical procedures. If working in a patient-facing role caring for patients with suspected or confirmed COVID-19, HCWs should wear an N95, N95-equivalent, or higher-level respirator; gown; gloves; and eye protection (goggles or a disposable face shield).15
Final Thoughts
COVID-19 has brought about acute and likely permanent changes to the US health care system. Dermatologists are integral members of that system and are essential to the treatment of patients with skin, hair, and nail disorders. Pregnant dermatologists and residents should refrain from patient-facing roles when feasible; however, when all recommended PPE are available, they may continue to work in patient-facing roles until they give birth if they desire to do so. Alternatively, teledermatology and non–face-to-face rotations should be encouraged. Higher-risk and aerosol-generating procedures are of particular concern regarding the risk for transmitting SARS-CoV-2 and should be avoided. Correct and universal use of PPE is paramount; when all recommended PPE is not available, pregnant HCWs should avoid exposure to patients with suspected or confirmed COVID-19. These recommendations will help safeguard pregnant members of dermatology teams during the COVID-19 pandemic while maximizing patient care.
Information is scarce regarding the impact of COVID-19 on pregnant women and newborns; health care workers (HCWs), particularly pregnant women,1 who are caring for patients during the pandemic might experience concern and uncertainty. The American College of Obstetricians and Gynecologists (ACOG) released recommendations, based on expert consensus, regarding pregnant HCWs on December 14, 2020.2 We propose an appropriation of the ACOG recommendations for dermatologists and their teams caring for patients during the COVID-19 pandemic.
Risks to Pregnant HCWs
Worldwide, viral pneumonia is a leading cause of death during pregnancy,3 with higher mortality documented among pregnant patients during the 1918 influenza pandemic and the 2003 severe acute respiratory syndrome–associated coronavirus pandemic,3 and an increased rate of hospital admission documented among these patients compared to the general population during the 2009 H1N1 influenza pandemic.4
Data from the Centers for Disease Control and Prevention (CDC) suggest that pregnant women with symptomatic COVID-19 (n=30,415) are at increased risk for the following (compared to nonpregnant women with symptomatic COVID-19 [n=431,410])5:
• Admission to the intensive care unit (10.5 of every 1000 cases vs 3.9 of every 1000 cases; adjusted risk ratio [aRR]=3.0; 95% CI, 2.6-3.4)
• Receipt of invasive ventilation (2.9 of every 1000 cases vs 1.1 of every 1000 cases; aRR=2.9; 95% CI, 2.2-3.8)
• Receipt of extracorporeal membrane oxygenation (0.7 of every 1000 cases vs 0.3 of every 1000 cases; aRR=2.4; 95% CI, 1.5-4.0)
• Death (1.5 of every 1000 cases vs 1.2 of every 1000 cases; aRR=1.7; 95% CI, 1.2-2.4).
Although the absolute risk of severe COVID-19–related outcomes is low, the CDC includes pregnant women in its increased risk category for COVID-19. Furthermore, in a systematic review of 61 studies comprising 790 COVID-19–positive pregnant women and 548 newborns, the rates of cesarean delivery, premature birth, low birth weight, and adverse pregnancy events (the latter comprising preterm birth, death or stillbirth, and early termination of pregnancy) were estimated to be 72%, 23%, 7%, and 27%, respectively.6 In a systematic review of 39 studies (case series and cohort studies), comprising 936 SARS-CoV-2–tested newborns of mothers with COVID-19, mother-to-fetus transmission of SARS-CoV-2 occurred during the third trimester in approximately 3.2% of infected mothers.7
In pregnant women with COVID-19 who develop cytokine storm syndrome, a fetal inflammatory response syndrome can ensue, which has been shown to cause ventricular expansion and bleeding in animal models.8 In addition, underlying conditions, such as cardiovascular disease, diabetes mellitus, pre-existing lung disease, and obesity, which are well-established risks factors for severe COVID-19 in nonpregnant patients, can increase the severity of COVID-19 in pregnant women.5,9-11
Recommendations From ACOG for Pregnant HCWs
The American College of Obstetricians and Gynecologists recommends that health care facilities consider limiting the exposure of pregnant HCWs to patients with confirmed or suspected COVID-19. They also recommend that pregnant women continue to work in patient-facing roles if they want to, if recommended personal protective equipment (PPE) is available for them to wear.2 The US Food and Drug Administration issued an Emergency Use Authorization for 2 messenger RNA COVID-19 vaccines. Although these vaccines have not been tested in pregnant women, ACOG recommends that COVID-19 vaccines not be withheld from pregnant women who fulfill the criteria for vaccination; pregnant women who decline vaccination should be supported in their decision.12 In dermatology, telemedicine is an effective alternative to face-to-face visits, reducing the risk of transmitting SARS-CoV-2 to physicians and patients.
Ideally, pregnant dermatology attending physicians and residents can continue to provide care through teledermatology. They also can continue to provide in-person care, if they choose to; however, higher-risk procedures should be avoided.12 In dermatology, that might include ablative laser procedures to the face, prolonged surgery, such as hair transplantation, and intraoral or intranasal procedures. Alternatively, pregnant dermatology residents can be allocated to clinical rotations in which face-to-face contact with patients is not required such as dermatopathology and a research rotation. Likewise, telework options can be encouraged for other pregnant members of dermatology teams, including front-desk staff, nurses, medical assistants, and remaining ancillary staff.
Guidance on Face Masks for Pregnant HCWs
Universal masking of HCWs has been shown to reduce the rate of health care–related acquisition of SARS-CoV-2.13 However, extended use or reuse of N95 respirators might contribute to SARS-CoV-2 transmission.14 The American College of Obstetricians and Gynecologists recommends that all HCWs wear a face mask at all times while working in a health care facility, even if patients are wearing a face covering or face mask.2 Based on CDC guidelines,15 HCWs in regions where community transmission is moderate or substantial should wear eye protection in addition to a face mask, and they should wear an N95, N95-equivalent, or higher-level respirator instead of a face mask when performing aerosol-generating procedures and surgical procedures. If working in a patient-facing role caring for patients with suspected or confirmed COVID-19, HCWs should wear an N95, N95-equivalent, or higher-level respirator; gown; gloves; and eye protection (goggles or a disposable face shield).15
Final Thoughts
COVID-19 has brought about acute and likely permanent changes to the US health care system. Dermatologists are integral members of that system and are essential to the treatment of patients with skin, hair, and nail disorders. Pregnant dermatologists and residents should refrain from patient-facing roles when feasible; however, when all recommended PPE are available, they may continue to work in patient-facing roles until they give birth if they desire to do so. Alternatively, teledermatology and non–face-to-face rotations should be encouraged. Higher-risk and aerosol-generating procedures are of particular concern regarding the risk for transmitting SARS-CoV-2 and should be avoided. Correct and universal use of PPE is paramount; when all recommended PPE is not available, pregnant HCWs should avoid exposure to patients with suspected or confirmed COVID-19. These recommendations will help safeguard pregnant members of dermatology teams during the COVID-19 pandemic while maximizing patient care.
- Rashidi Fakari F, Simbar M. Coronavirus pandemic and worries during pregnancy; a letter to editor. Arch Acad Emerg Med. 2020;8:E21.
- The American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, obstetrics. 2020. Accessed April 21, 2021. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics
- Schwartz DA, Graham AL. Potential maternal and infant outcomes from (Wuhan) coronavirus 2019-nCoV infecting pregnant women: lessons from SARS, MERS, and other human coronavirus infections. Viruses. 2020;12:194. doi:10.3390/v12020194
- Yan J, Guo J, Fan C, et al. Coronavirus disease 2019 in pregnant women: a report based on 116 cases. Am J Obstet Gynecol. 2020;223:111.e1-111.e14. doi:10.1016/j.ajog.2020.04.014
- Zambrano LD, Ellington S, Strid P, et al; . Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3
- Dubey P, Reddy SY, Manuel S, et al. Maternal and neonatal characteristics and outcomes among COVID-19 infected women: an updated systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;252:490-501. doi:10.1016/j.ejogrb.2020.07.034
- Kotlyar AM, Grechukhina O, Chen A, et al. Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis. Am J Obstet Gynecol. 2020;224:35-53.e3. doi:10.1016/j.ajog.2020.07.049
- Mitchell T, MacDonald JW, Srinouanpranchanh S, et al. Evidence of cardiac involvement in the fetal inflammatory response syndrome: disruption of gene networks programming cardiac development in nonhuman primates. Am J Obstet Gynecol. 2018;218:438.e1-438.e16. doi:10.1016/j.ajog.2018.01.009
- Ellington S, Strid P, Tong VT, et al. Characteristics of women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–June 7, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:769-775. doi:10.15585/mmwr.mm6925a1
- Panagiotakopoulos L, Myers TR, Gee J, et al. SARS-CoV-2 infection among hospitalized pregnant women: reasons for admission and pregnancy characteristics—eight U.S. health care centers, March 1–May 30, 2020. 2020. doi:10.15585/mmwr.mm6938e2
- Knight M, Bunch K, Vousden N, et al; . Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107. doi:10.1136/bmj.m2107
- The American College of Obstetricians and Gynecologists. Vaccinating pregnant and lactating patients against COVID-19. December 2020. Updated March 24, 2021. Accessed April 28, 2021. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/vaccinating-pregnant-and-lactating-patients-against-covid-19
- Seidelman JL, Lewis SS, Advani SD, et al. Universal masking is an effective strategy to flatten the severe acute respiratory coronavirus virus 2 (SARS-CoV-2) healthcare worker epidemiologic curve. Infect Control Hosp Epidemiol. 2020;41:1466-1467. doi:10.1017/ice.2020.31314.
- Degesys NF, Wang RC, Kwan E, et al. Correlation between N95 extended use and reuse and fit failure in an emergency department. JAMA. 2020;324:94-96. doi:10.1001/jama.2020.9843
- Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic 2020. Updated February 23, 2021. Accessed April 21, 2021. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Finfection-control%2Fcontrol-recommendations.html
- Rashidi Fakari F, Simbar M. Coronavirus pandemic and worries during pregnancy; a letter to editor. Arch Acad Emerg Med. 2020;8:E21.
- The American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, obstetrics. 2020. Accessed April 21, 2021. https://www.acog.org/clinical-information/physician-faqs/covid-19-faqs-for-ob-gyns-obstetrics
- Schwartz DA, Graham AL. Potential maternal and infant outcomes from (Wuhan) coronavirus 2019-nCoV infecting pregnant women: lessons from SARS, MERS, and other human coronavirus infections. Viruses. 2020;12:194. doi:10.3390/v12020194
- Yan J, Guo J, Fan C, et al. Coronavirus disease 2019 in pregnant women: a report based on 116 cases. Am J Obstet Gynecol. 2020;223:111.e1-111.e14. doi:10.1016/j.ajog.2020.04.014
- Zambrano LD, Ellington S, Strid P, et al; . Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–October 3, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:1641-1647. doi:10.15585/mmwr.mm6944e3
- Dubey P, Reddy SY, Manuel S, et al. Maternal and neonatal characteristics and outcomes among COVID-19 infected women: an updated systematic review and meta-analysis. Eur J Obstet Gynecol Reprod Biol. 2020;252:490-501. doi:10.1016/j.ejogrb.2020.07.034
- Kotlyar AM, Grechukhina O, Chen A, et al. Vertical transmission of coronavirus disease 2019: a systematic review and meta-analysis. Am J Obstet Gynecol. 2020;224:35-53.e3. doi:10.1016/j.ajog.2020.07.049
- Mitchell T, MacDonald JW, Srinouanpranchanh S, et al. Evidence of cardiac involvement in the fetal inflammatory response syndrome: disruption of gene networks programming cardiac development in nonhuman primates. Am J Obstet Gynecol. 2018;218:438.e1-438.e16. doi:10.1016/j.ajog.2018.01.009
- Ellington S, Strid P, Tong VT, et al. Characteristics of women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status—United States, January 22–June 7, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:769-775. doi:10.15585/mmwr.mm6925a1
- Panagiotakopoulos L, Myers TR, Gee J, et al. SARS-CoV-2 infection among hospitalized pregnant women: reasons for admission and pregnancy characteristics—eight U.S. health care centers, March 1–May 30, 2020. 2020. doi:10.15585/mmwr.mm6938e2
- Knight M, Bunch K, Vousden N, et al; . Characteristics and outcomes of pregnant women admitted to hospital with confirmed SARS-CoV-2 infection in UK: national population based cohort study. BMJ. 2020;369:m2107. doi:10.1136/bmj.m2107
- The American College of Obstetricians and Gynecologists. Vaccinating pregnant and lactating patients against COVID-19. December 2020. Updated March 24, 2021. Accessed April 28, 2021. https://www.acog.org/clinical/clinical-guidance/practice-advisory/articles/2020/12/vaccinating-pregnant-and-lactating-patients-against-covid-19
- Seidelman JL, Lewis SS, Advani SD, et al. Universal masking is an effective strategy to flatten the severe acute respiratory coronavirus virus 2 (SARS-CoV-2) healthcare worker epidemiologic curve. Infect Control Hosp Epidemiol. 2020;41:1466-1467. doi:10.1017/ice.2020.31314.
- Degesys NF, Wang RC, Kwan E, et al. Correlation between N95 extended use and reuse and fit failure in an emergency department. JAMA. 2020;324:94-96. doi:10.1001/jama.2020.9843
- Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for healthcare personnel during the coronavirus disease 2019 (COVID-19) pandemic 2020. Updated February 23, 2021. Accessed April 21, 2021. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fcoronavirus%2F2019-ncov%2Finfection-control%2Fcontrol-recommendations.html
Practice Points
- Pregnant women are at an increased risk for severe illness due to COVID-19 compared with nonpregnant women; therefore, it is important to protect pregnant health care workers who are caring for patients during the current pandemic.
- Although currently available COVID-19 vaccines have not been tested in pregnant women, they should not be withheld from pregnant individuals.
- Pregnant attending physicians and residents in dermatology can continue to provide care through telemedicine; if they choose to, and if all recommended personal protective equipment (PPE) are available, they can continue to provide in-person care.
- Correct and comprehensive use of PPE by pregnant health care workers is paramount to minimizing exposure to SARS-CoV-2.









