User login
Transplantation palliative care: The time is ripe
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
Over 10 years ago, a challenge was made in a surgical publication for increased collaboration between the fields of transplantation and palliative care.1
Since that time not much progress has been made bringing these fields together in a consistent way that would mutually benefit patients and the specialties. However, other progress has been made, particularly in the field of palliative care, which could brighten the prospects and broaden the opportunities to accomplish collaboration between palliative care and transplantation.
Growth of palliative services
During the past decade there has been a robust proliferation of hospital-based palliative care programs in the United States. In all, 67% of U.S. hospitals with 50 or more beds report palliative care teams, up from 63% in 2011 and 53% in 2008.
Only a decade ago, critical care and palliative care were generally considered mutually exclusive. Evidence is trickling in to suggest that this is no longer the case. Although palliative care was not an integral part of critical care at that time, patients, families, and even practitioners began to demand these services. Cook and Rocker have eloquently advocated the rightful place of palliative care in the ICU.2
Studies in recent years have shown that the integration of palliative care into critical care decreases in length of ICU and hospital stay, decreases costs, enhances patient/family satisfaction, and promotes a more rapid consensus about goals of care, without increasing mortality. The ICU experience to date could be considered a reassuring precedent for transplantation palliative care.
Integration of palliative care with transplantation
Early palliative care intervention has been shown to improve symptom burden and depression scores in end-stage liver disease patients awaiting transplant. In addition, early palliative care consultation in conjunction with cancer treatment has been associated with increased survival in non–small-cell lung cancer patients. It has been demonstrated that early integration of palliative care in the surgical ICU alongside disease-directed curative care can be accomplished without change in mortality, while improving end-of-life practice in liver transplant patients.3
What palliative care can do for transplant patients
What does palliative care mean for the person (and family) awaiting transplantation? For the cirrhotic patient with cachexia, ascites, and encephalopathy, it means access to the services of a team trained in the management of these symptoms. Palliative care teams can also provide psychosocial and spiritual support for patients and families who are intimidated by the complex navigation of the health care system and the existential threat that end-stage organ failure presents to them. Skilled palliative care and services can be the difference between failing and extended life with a higher quality of life for these very sick patients
Resuscitation of a patient, whether through restoration of organ function or interdicting the progression of disease, begins with resuscitation of hope. Nothing achieves this more quickly than amelioration of burdensome symptoms for the patient and family.
The barriers for transplant surgeons and teams referring and incorporating palliative care services in their practices are multiple and profound. The unique dilemma facing the transplant team is to balance the treatment of the failing organ, the treatment of the patient (and family and friends), and the best use of the graft, a precious gift of society.
Palliative surgery has been defined as any invasive procedure in which the main intention is to mitigate physical symptoms in patients with noncurable disease without causing premature death. The very success of transplantation over the past 3 decades has obscured our memory of transplantation as a type of palliative surgery. It is a well-known axiom of reconstructive surgery that the reconstructed site should be compared to what was there, not to “normal.” Even in the current era of improved immunosuppression and posttransplant support services, one could hardly describe even a successful transplant patient’s experience as “normal.” These patients’ lives may be extended and/or enhanced but they need palliative care before, during, and after transplantation. The growing availability of trained palliative care clinicians and teams, the increased familiarity of palliative and end-of-life care to surgical residents and fellows, and quality metrics measuring palliative care outcomes will provide reassurance and guidance to address reservations about the convergence of the two seemingly opposite realities.
A modest proposal
We propose that palliative care be presented to the entire spectrum of transplantation care: on the ward, in the ICU, and after transplantation. More specific “triggers” for palliative care for referral of transplant patients should be identified. Wentlandt et al.4 have described a promising model for an ambulatory clinic, which provides early, integrated palliative care to patients awaiting and receiving organ transplantation. In addition, we propose an application for grant funding for a conference and eventual formation of a work group of transplant surgeons and team members, palliative care clinicians, and patient/families who have experienced one of the aspects of the transplant spectrum. We await the subspecialty certification in hospice and palliative medicine of a transplant surgeon. Outside of transplantation, every other surgical specialty in the United States has diplomates certified in hospice and palliative medicine. We await the benefits that will accrue from research about the merging of these fields.
1. Molmenti EP, Dunn GP: Transplantation and palliative care: The convergence of two seemingly opposite realities. Surg Clin North Am. 2005;85:373-82.
2. Cook D, Rocker G. Dying with dignity in the intensive care unit. N Engl J Med. 2014;370:2506-14.
3. Lamba S, Murphy P, McVicker S, Smith JH, and Mosenthal AC. Changing end-of-life care practice for liver transplant patients: structured palliative care intervention in the surgical intensive care unit. J Pain Symptom Manage. 2012; 44(4):508-19.
4. Wentlandt, K., Dall’Osto, A., Freeman, N., Le, L. W., Kaya, E., Ross, H., Singer, L. G., Abbey, S., Clarke, H. and Zimmermann, C. (2016), The Transplant Palliative Care Clinic: An early palliative care model for patients in a transplant program. Clin Transplant. 2016 Nov 4; doi: 10.1111/ctr.12838.
Dr. Azoulay is a transplantation specialist of Assistance Publique – Hôpitaux de Paris, and the University of Paris. Dr. Dunn is medical director of the Palliative Care Consultation Service at the University of Pittsburgh Medical Center Hamot, and vice-chair of the ACS Committee on Surgical Palliative Care.
SVS Now Accepting Abstracts for VAM 2017
Abstracts for the 2017 Vascular Annual Meeting are now being accepted. The submission site opened Monday, Nov. 14 for the meeting, to be held May 31 to June 3, 2017, in San Diego. Plenary sessions and exhibits will be June 1 to 3.
Participants may submit abstracts into any of 14 categories and a number of presentation types, including videos. In 2016, organizers selected approximately two-thirds of the submitted abstracts, and this year the VAM Program Committee is seeking additional venues for people to present their work in, including more sessions and other presentation formats.
Click here for abstract guidelines and more information. Abstracts themselves may be submitted here.
Abstracts for the 2017 Vascular Annual Meeting are now being accepted. The submission site opened Monday, Nov. 14 for the meeting, to be held May 31 to June 3, 2017, in San Diego. Plenary sessions and exhibits will be June 1 to 3.
Participants may submit abstracts into any of 14 categories and a number of presentation types, including videos. In 2016, organizers selected approximately two-thirds of the submitted abstracts, and this year the VAM Program Committee is seeking additional venues for people to present their work in, including more sessions and other presentation formats.
Click here for abstract guidelines and more information. Abstracts themselves may be submitted here.
Abstracts for the 2017 Vascular Annual Meeting are now being accepted. The submission site opened Monday, Nov. 14 for the meeting, to be held May 31 to June 3, 2017, in San Diego. Plenary sessions and exhibits will be June 1 to 3.
Participants may submit abstracts into any of 14 categories and a number of presentation types, including videos. In 2016, organizers selected approximately two-thirds of the submitted abstracts, and this year the VAM Program Committee is seeking additional venues for people to present their work in, including more sessions and other presentation formats.
Click here for abstract guidelines and more information. Abstracts themselves may be submitted here.
Best Practices: Protecting Dry Vulnerable Skin with CeraVe® Healing Ointment
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A supplement to Dermatology News. This advertising supplement is sponsored by Valeant Pharmaceuticals.
- Reinforcing the Skin Barrier
- NEA Seal of Acceptance
- A Preventative Approach to Dry, Cracked Skin
- CeraVe Ointment in the Clinical Setting
Faculty/Faculty Disclosure
Sheila Fallon Friedlander, MD
Professor of Clinical Dermatology & Pediatrics
Director, Pediatric Dermatology Fellowship Training Program
University of California at San Diego School of Medicine
Rady Children’s Hospital,
San Diego, California
Dr. Friedlander was compensated for her participation in the development of this article.
CeraVe is a registered trademark of Valeant Pharmaceuticals International, Inc. or its affiliates.
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.
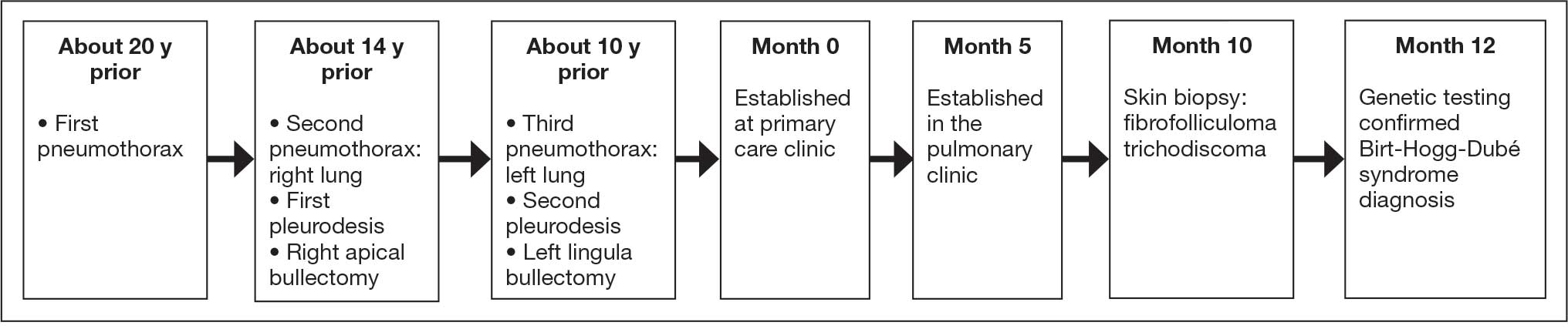
On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).
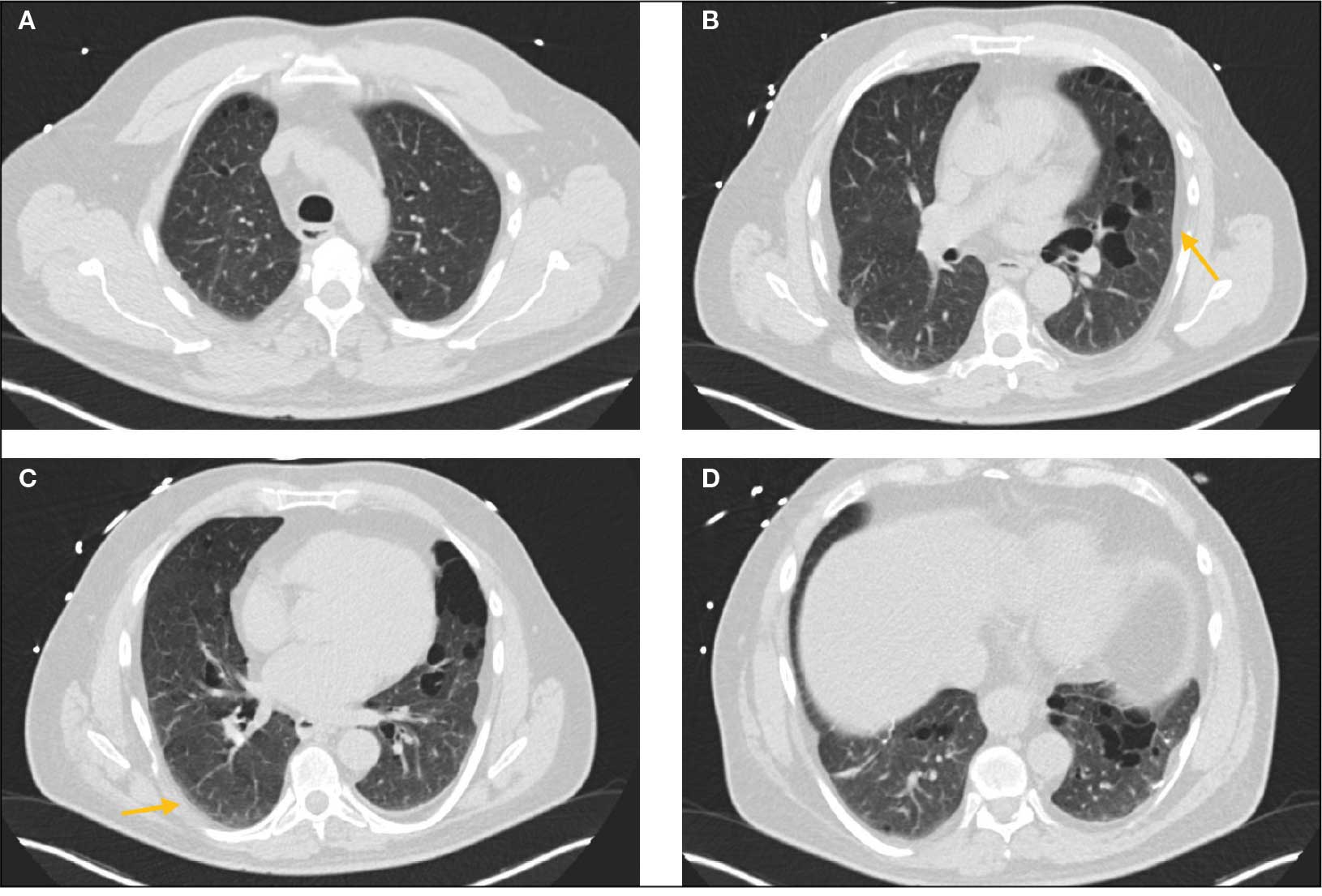
Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).
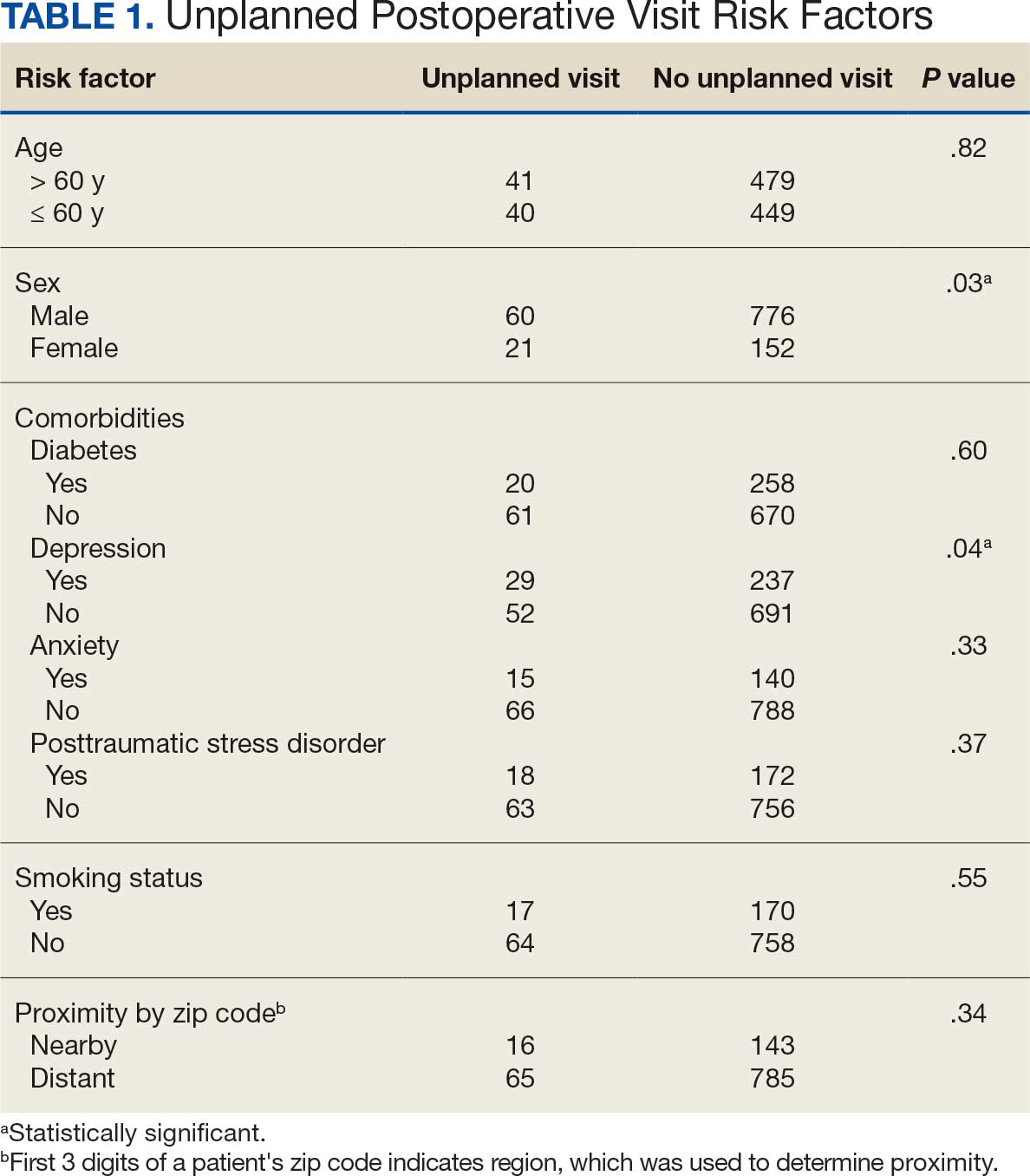

Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
Patients make unplanned appointments after elective soft tissue hand surgery for real or perceived complications when they experience pain, anxiety, or fear. Unplanned appointments can create travel and financial burdens for patients and families. These appointments take time away from scheduled appointments and can contribute to late arrivals and delays in other clinics. Unscheduled appointments contribute to poor access when staff are diverted from scheduled appointments. If predictive factors can be identified, unplanned appointments may either be ameliorated or avoided with better perioperative risk management or education.
Methods
The US Department of Veterans Affairs (VA) North Florida/South Georgia Veterans Health System (NFSGVAHS) and University of Florida Institutional Review Board approved a retrospective chart review of all plastic surgery cases performed at the Malcom Randall VA Medical Center (MRVAMC) and Lake City VAMC operating rooms from July 1, 2018, through December 31, 2019, and January 1, 2021, through June 30, 2022 (nonurgent surgeries were discouraged during the COVID-19 pandemic). Elective soft tissue hand surgery cases were identified based on the operative description found in the Surgical Service Surgeon Staffing Report reviewed monthly by the Service Chief. Potential indicators of unplanned visits were recorded, including age; sex; diagnosis of diabetes, depression, anxiety, or posttraumatic stress disorder (PTSD); current smoking status; and residential zip code. We used the first 3 digits of the patients’ zip codes, which indicate region, as an estimate of proximity to the MRVAMC, which has a 50-county catchment area across North Florida and South Georgia. Diagnoses were found on the “problem list” from the electronic health record documented in the history and physical examinations before surgery. Clinic notes were examined for 3 months postsurgery to identify unplanned postoperative visits and the reason for the appointment. A χ2 analysis was conducted using Excel Version 2402. P < .05 was used to determine whether age (> 60 years), sex, proximity to MRVAMC, diabetes, smoking, depression, anxiety, or PTSD were statistically significant independent risk factors for these appointments.
Results
A total of 1009 elective soft tissue hand surgeries at MRVAMC were reviewed. The patients median age was 61 years. Patients included 173 women (17.1%) and 836 men (82.9%). Eighty-one patients (8.0%) returned for unplanned visits. Age (P = .82); proximity to MRVAMC (P = .34); and diabetes (P = .60), smoking (P = .55), anxiety (P = .33), or PTSD (P = .37) were not statistically significant predictors of unplanned appointments. Depression diagnosis (P = .04) and female sex (P = .03) were found to be independent risk factors for an unplanned appointment (Table 1). The most common indication for the requested appointment was pain-related, followed closely by noninfectious wound concerns and persistent symptoms (Table 2).


Discussion
Improved access, quality, and efficiency for patients are goals for the VA.1-3 The MRVAMC Plastic and Hand Surgery service provides care for the NFSGVAHS and receives an average of 15 to 20 consultation requests daily. The Veterans Health Administration is frequently challenged by staff shortages, and surgical services struggle to respond to consultation requests and treat patients within reasonable time frames.4,5
The objective of this study was to identify risk factors for unplanned postoperative appointments following elective hand surgery. Unplanned appointments prevent scheduled patients from being seen on time and contribute to backlogs and delays. When patients schedule multiple appointments on the same day, delays in the first clinic’s scheduled appointments create delays for the second and third clinics. Hand surgery clinics can provide a better experience for patients and staff by identifying and mitigating factors prompting unplanned visits.
We anticipated that wound complications would prompt unscheduled visits. Diabetes is a known risk factor for wound healing complications after plastic and hand surgery.6,7 A hemoglobin A1c (HbA1c) screening protocol used by the NFSGVAHS plastic surgery service since 2015 to identify poorly controlled patients with diabetes before surgery may partially explain this finding.8 We did not find a statistically significant difference between patients with diabetes and patients without diabetes for scheduling unplanned appointments. The plastic surgery service does not perform elective hand surgery unless the patient’s HbA1c level is < 9%, or violate the flexor sheath unless HbA1c level is < 8%. However, Zhuang et al found an increase in soft tissue infections after hand surgery with HbA1c levels ≥ 7%.9
Smoking is a potential factor in postoperative hand surgery complications.10,11 Lans et al found an increased incidence of 30-day emergency room visits in current and former smokers after outpatient upper extremity fracture surgery.12 The MRVAMC Plastic Surgery Service counsels patients about the risk of skin necrosis and delayed wound healing, but does not cancel cases or obtain laboratory values to verify abstinence in patients undergoing hand surgery. The VA has multiple resources available for patients interested in smoking cessation through mental health services.13
MRVAMC patients have been known to resist returning for scheduled appointments due to the costs or availability of transportation. We suspected that patients who lived further from MRVAMC would be less likely to return for unscheduled visits. We used the first 3 digits of the patients’ mailing zip code to estimate residential proximity to MRVAMC. An acknowledged limitation to this approach is that some veterans have primary addresses in other regions but still spend significant time in the MRVAMC catchment area and use the facility for their health care during the winter months. These “snowbirds” might reside near the facility despite having official addresses that are more distant. Additionally, there was no increased risk of unplanned visits after hand surgery in patients aged > 61 years (the median age of study participants) (P = .82). Dependence on a third party for transportation in older veterans could impact this finding.
Based on the observation that most patients needed reassurance rather than an intervention when they returned for unscheduled appointments, diagnoses of depression, anxiety, and PTSD were evaluated as separate predictive factors. In previous research, anxiety was found to be a risk factor for problematic recovery following carpal tunnel surgery.14 In the current study, depression was found to be a statistically significant predictor of unscheduled postoperative appointments (P = .04), while anxiety (P = .33) and PTSD (P = .37) were not statistically significant predictors. This is consistent with other studies that have found preexisting depression can predict complications after hand surgery.15,16 Vranceanu et al found that depression predicted pain intensity and disability after elective hand surgery.16 Similarly, Oflazoglu et al found a 12% incidence of depression based on the Patient Health Questionnaire-9 in new and returning hand patients who presented to an academic practice.17 They suggest patients should be assessed at all levels of care and that those with poor responses to surgical or nonsurgical management should be evaluated for depression. MRVAMC has a large mental health service consisting of psychiatrists, psychologists, addiction specialists, social workers, and homeless outreach, and patients tend to already have a diagnosis and mental health practitioner when they present to the clinic.
Recent studies found that wound problems, pain, and stiffness were the most common reasons for return visits.18,19 Shetty et al identified younger age, worse preoperative pain scores, and poor access to transportation as predictors of preventable emergency room visits, which generate higher health care expenditures than an office visit.19 Our study’s top reasons for appointments (pain, wound/scar concerns, persistent symptoms) can be addressed with additional presurgery patient and family education. Additionally, clinicians encourage nonnarcotic pain management strategies including anti-inflammatories, acetaminophen, elevation, splinting, and hand therapy, and the hospital employs experienced, fellowship-trained anesthesia block faculty who help limit perioperative narcotic use. Patients are advised that pain can be used to guide them through the postoperative recovery by preventing overuse and alerting them to a problem that would be masked with narcotics, and long-standing problems such as chronic nerve compressions may continue to cause pain after surgery.
Patients and families can be given consistent and repetitive verbal and written information, instructions, and expectations at the initial consultation, preoperative appointment, and on the day of surgery. Postoperatively, outside their scheduled appointments, patients are encouraged to use the My HealtheVet secure messaging system or call the clinic to access an experienced registered nurse before making a long drive. Access to virtual or phone visits can reduce emergent in-person visits in a VA population.20
Ozdag et al found that 42% of patients who had elective carpal tunnel surgery made unplanned electronic messages or phone contact within 2 weeks postsurgery. The authors point out the uncompensated administrative burden on the staff answering these messages and suggest pre-empting the contacts with more up-front education regarding postoperative pain expectations and management strategies.21
Fisher et al found that attending hand therapy reduced the number of emergency department visits in postoperative infection cases.22 At MRVAMC, a postoperative emergency department visit for a patient prompts an urgent unplanned appointment to the plastic surgery clinic, often on the same day. The MRVAMC occupational therapy clinic employed 3 on-site certified hand therapists during the study period. Because all hand surgery patients at the clinic receive hand therapy on the same day as their first postoperative appointment, attendance at hand therapy was not evaluated as a predictor of unplanned visits. Scheduled hand therapy is another point of contact where the clinic can provide reassurance and patient education.
While females made up 17.1% of the patients in this study, they constituted 12.5% of all veterans in Florida in fiscal year 2023.23 This study found that women were more likely to present for unplanned postoperative appointments (P = .03). This is consistent with existing literature which has found that women are higher users of health care and office-based appointments.24,25 This finding suggests the need for further study into whether our methods of communicating instructions to female patients undergoing plastic surgery may not be optimal.
Strengths and Limitations
As a retrospective review, the authors used information documented by multiple different health care practitioners, including trainees. The electronic medical record problem lists and templates provide consistency of information; however, less seasoned clinicians may interpret what they see and hear differently from more experienced clinicians in the postoperative setting. This study occurred in one part of the country with demographics that may not mirror other VA systems or the general population. The authors hope this study can be a starting point for other health care facilities to investigate ways to minimize the burden of unscheduled appointments. A strength of the study is that it was conducted within a closed system, as patients tend to stay within the VA system and documentation and communication among clinicians, even outside the immediate facility, are easily accessed through the electronic health record.
Conclusions
This study found that depression diagnosis and female sex are statistically significant predictors of unplanned postoperative visits after elective soft tissue hand surgery. More effective patient education during the preoperative period, particularly in patients with depression, may be warranted.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
- Apaydin EA, Paige NM, Begashaw MM, et al. Veterans Health Administration (VA) vs. non-VA healthcare quality: a systematic review. J Gen Intern Med. 2023;38:2179-2188. doi:10.1007/s11606-023-08207-2
- Blegen M, Ko J, Salzman G, et al. Comparing quality of surgical care between the US Department of Veterans Affairs and non-Veterans Affairs settings: a systematic review. J Am Coll Surg. 2023;237:352-361. doi:10.1097/XCS.0000000000000720
- Valsangkar NP, Eppstein AC, Lawson RA, et al. Effect of lean processes on surgical wait times and efficiency in a tertiary care veterans affairs medical center. JAMA Surg. 2017;152:42-47. doi:10.1001/jamasurg.2016.2808
- National Association of Veterans Affairs Physicians and Dentists. Physicians remain at top of staffing shortage in VA. NAVAPD. December 20, 2023. Accessed March 16, 2026. https://www.navapd.org/news/physicians-remain-at-top-of-staffing-shortage-in-va
- OIG Determination of Veterans Health Administration’s severe occupational staffing shortages fiscal year 2024. Veterans Affairs Office of Inspector General. August 7, 2024. Accessed February 4, 2026. https://www.vaoig.gov/reports/national-healthcare-review/oig-determination-veterans-health-administrations-severe-0
- Goltsman D, Morrison KA, Ascherman JA. Defining the association between diabetes and plastic surgery outcomes: an analysis of nearly 40,000 patients. Plast Reconstr Surg Glob Open. 2017;5:e1461. doi:10.1097/GOX.0000000000001461 7.
- Cox CT, Sierra S, Egan A, et al. Elevated hemoglobin A1c and the risk of postoperative complications in elective hand and upper extremity surgery. Cureus. 2023;15:e48373. doi:10.7759/cureus.48373
- Coady-Fariborzian L, Anstead C. HbA1c and infection in diabetic elective hand surgery: a Veterans Affair Medical Center experience 2012-2018. Hand (NY). 2023;18:994-998. doi:10.1177/1558944720937363<
- Zhuang T, Shapiro LM, Fogel N, et al. Perioperative laboratory markers as risk factors for surgical site infection after elective hand surgery. J Hand Surg Am. 2021;46:675-684. doi:10.1016/j.jhsa.2021.04.001
- Cho BH, Aziz KT, Giladi AM. The impact of smoking on early postoperative complications in hand surgery. J Hand Surg Am. 2021;46:336.e1-336.e11. doi:10.1016/j.jhsa.2020.07.01411.
- Del Core MA, Ahn J, Golden AS, et al. Effect of smoking on short-term postoperative complications after elective upper extremity surgery. Hand (N Y). 2022;17:231-238. doi:10.1177/1558944720926638
- Lans J, Beagles CB, Watkins IT, et al. Unplanned postoperative emergency department visits after upper extremity fracture surgery. J Orthop Trauma. 2025;39:22-27. doi:10.1097/BOT.0000000000002925
- Tobacco and health - how to quit. US Dept of Veterans Affairs. Updated October 29, 2025. Accessed February 4, 2026. https://www.mentalhealth.va.gov/quit-tobacco/how-to-quit.asp
- Ryan C, Miner H, Ramachandran S, et al. General anxiety is associated with problematic initial recovery after carpal tunnel release. Clin Orthop Relat Res. 2022;480:1576-1581. doi:10.1097/CORR.0000000000002115
- Crijns TJ, Bernstein DN, Ring D, et al. Depression and pain interference correlate with physical function in patients recovering from hand surgery. Hand (N Y). 2019;14:830-835. doi:10.1177/1558944718777814
- Vranceanu AM, Jupiter JB, Mudgal CS, et al. Predictors of pain intensity and disability after minor hand surgery. J Hand Surg Am. 2010;35:956-960. doi:10.1016/j.jhsa.2010.02.00117.
- Oflazoglu K, Mellema JJ, Menendez ME, et al. Prevalence of and factors associated with major depression in patients with upper extremity conditions. J Hand Surg Am. 2016;41:263-269. doi:10.1016/j.jhsa.2015.11.019
- Townsend CB, Henry TW, Lutsky KF, et al. Unplanned office visits following outpatient hand surgery. Hand (N Y). 2022;17:1264-1268. doi:10.1177/15589447211028932
- Shetty PN, Guarino GM, Zhang G, et al. Risk factors for preventable emergency department use after outpatient hand surgery. J Hand Surg Am. 2022;47:855-864. doi:10.1016/j.jhsa.2022.05.012
- Sommers-Olson B, Christianson J, Neumann T, et al. Reducing nonemergent visits to the emergency department in a Veterans Affairs multistate system. J Emerg Nurs. 2023;49:539-545. doi:10.1016/j.jen.2023.02.010
- Ozdag Y, Manzar S, El Koussaify J, et al. Unplanned postoperative phone calls and electronic messages for patients with and without opioid prescriptions after carpal tunnel release. J Hand Surg Glob Online. 2024;6:363-368. doi:10.1016/j.jhsg.2024.02.006
- Fisher AH, Gandhi J, Nelson Z, et al. Immediate interventions after surgery to reduce readmission for upper extremity infections. Ann Plast Surg. 2022;88:S163-S169. doi:10.1097/SAP.0000000000003141
- Florida Department of Veterans Affairs Fast Facts. Florida Department of Veterans Affairs. Accessed February 4, 2026. https://floridavets.org/our-veterans/profilefast-facts/
- Bertakis KD, Azari R, Helms LJ, et al. Gender differences in the utilization of health care services. J Fam Pract. 2000;49:147-152.
- Ashman JJ, Santo L, Okeyode T. Characteristics of office-based physician visits, 2018. NCHS Data Brief. 2021;408:1-8.
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Predictors of Unplanned Postoperative Visits in a Veterans Affairs Hand Surgery Practice
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
TOPLINE:
In a meta-analysis, patients with hidradenitis suppurativa (HS) faced a more than 80% higher risk for cancer overall than the general population, with particularly elevated risks for gastrointestinal, head and neck, hematologic, and respiratory system cancers.
METHODOLOGY:
- Researchers conducted a meta-analysis including 11 studies from PubMed, Embase, and Web of Science databases published between 2001 and 2024; these studies examined the risk for cancer in patients with HS compared with that in the general population.
- These studies included 624,721 patients diagnosed with HS (mean age, 33.6-43.8 years) and 393,691,636 control individuals from the general population.
- Researchers performed an inverse variance-weighted random-effects analysis to calculate pooled odds ratios (ORs) for cancer overall and specific cancer subtypes.
- Cancer types were categorized into 11 groups for subgroup analysis: bone and soft tissue cancers, breast cancer, central nervous system cancers, endocrine-related cancers, gastrointestinal cancers, head and neck cancers, hematologic cancers, respiratory system cancers, skin cancers, urogenital cancers, and unspecified cancers.
TAKEAWAY:
- Patients with HS demonstrated a significantly higher risk for cancer overall than control individuals (crude OR, 1.82; P = .018).
- Patients with HS showed an increased risk for gastrointestinal cancers (crude OR, 1.61; P = .0002), head and neck cancers (crude OR, 2.41; P = .00001), hematologic cancers (crude OR, 1.71; P = .00005), and respiratory system cancers (crude OR, 1.81; P = .04).
- Patients with HS demonstrated significantly elevated risks for both Hodgkin lymphoma (OR, 2.44; P = .0001) and non-Hodgkin lymphoma (OR, 1.15; P = .012).
- A non-significant increased risk for skin cancer was observed in patients with HS (crude OR, 1.48; P = .08). No increased risks for bone and soft tissue cancers, central nervous system cancers, breast cancer, or urogenital cancers were observed in patients with HS.
IN PRACTICE:
"HS was associated with an increased overall risk of cancer, including several specific subtypes, compared with controls," the authors wrote, suggesting that "studies are adjusting for confounders and assess long-term associations between HS and cancer risk are highly needed to investigate which factors contribute to this cancer risk."
SOURCE:
This study was led by Daniel Isufi, Department of Dermatology and Allergy, Copenhagen University Hospital-Herlev and Gentofte Hospital, University of Copenhagen, Copenhagen, Denmark. It was published online on March 11, 2026, in Dermatology and Therapy.
LIMITATIONS:
Limited data on cancer subtypes hindered meta-analyses of rare cancers, and the lack of reporting on anti‑inflammatory treatment and disease severity prevented subgroup analyses. Most studies originated from North America, introducing potential geographic bias. No study reported BMI, and ethnicity was poorly documented. Only few studies adjusted for key confounders (smoking, obesity, and alcohol intake), limiting the determination of whether the increased risk for cancer was due to HS itself or shared lifestyle and metabolic factors.
DISCLOSURES:
This study did not receive any funding or sponsorship. Two authors reported receiving research grant funding from the LEO Foundation and having other ties with various other sources.
This article was created using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication.
A version of this article first appeared on Medscape.com.
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types
Hidradenitis Suppurativa Associated With Elevated Risks for Multiple Cancer Types