User login
Delaying second dose of SARS-CoV-2 vaccine not advisable in IBD patients treated with infliximab
Key clinical point: Patients with inflammatory bowel disease (IBD) treated with infliximab vs. vedolizumab showed attenuated immunogenicity to a single dose of BNT162b2 and adenovirus-ChAdOx1 nCoV-19 SARS-CoV-2 vaccines. Reassuringly, second dose of the vaccine led to seroconversion in most patients.
Major finding: Anti-SARS-CoV-2 antibody concentrations were lower in patients treated with infliximab vs. vedolizumab following vaccination with a single dose of either BNT162b2 (fold change [FC], 0.29; P less than .0001) or ChAdOx1 nCoV-19 (FC, 0.39; P less than .0001). After second vaccine dose, 85% of infliximab- and 86% of vedolizumab-treated patients seroconverted (P = .68).
Study details: Findings are from the CLARITY IBD study that included patients with IBD without evidence of prior SARS-CoV-2 infection who were treated with infliximab (n=865) or vedolizumab (n=428).
Disclosures: The study was funded by F. Hoffmann-La Roche AG (Switzerland), Biogen GmbH (Switzerland), Celltrion Healthcare (South Korea), and others. Some of the authors reported receiving grants, personal fees, and/or financial or nonfinancial support from multiple sources.
Source: Kennedy NA et al. Gut. 2021 Apr 26. doi: 10.1136/gutjnl-2021-324789.
Key clinical point: Patients with inflammatory bowel disease (IBD) treated with infliximab vs. vedolizumab showed attenuated immunogenicity to a single dose of BNT162b2 and adenovirus-ChAdOx1 nCoV-19 SARS-CoV-2 vaccines. Reassuringly, second dose of the vaccine led to seroconversion in most patients.
Major finding: Anti-SARS-CoV-2 antibody concentrations were lower in patients treated with infliximab vs. vedolizumab following vaccination with a single dose of either BNT162b2 (fold change [FC], 0.29; P less than .0001) or ChAdOx1 nCoV-19 (FC, 0.39; P less than .0001). After second vaccine dose, 85% of infliximab- and 86% of vedolizumab-treated patients seroconverted (P = .68).
Study details: Findings are from the CLARITY IBD study that included patients with IBD without evidence of prior SARS-CoV-2 infection who were treated with infliximab (n=865) or vedolizumab (n=428).
Disclosures: The study was funded by F. Hoffmann-La Roche AG (Switzerland), Biogen GmbH (Switzerland), Celltrion Healthcare (South Korea), and others. Some of the authors reported receiving grants, personal fees, and/or financial or nonfinancial support from multiple sources.
Source: Kennedy NA et al. Gut. 2021 Apr 26. doi: 10.1136/gutjnl-2021-324789.
Key clinical point: Patients with inflammatory bowel disease (IBD) treated with infliximab vs. vedolizumab showed attenuated immunogenicity to a single dose of BNT162b2 and adenovirus-ChAdOx1 nCoV-19 SARS-CoV-2 vaccines. Reassuringly, second dose of the vaccine led to seroconversion in most patients.
Major finding: Anti-SARS-CoV-2 antibody concentrations were lower in patients treated with infliximab vs. vedolizumab following vaccination with a single dose of either BNT162b2 (fold change [FC], 0.29; P less than .0001) or ChAdOx1 nCoV-19 (FC, 0.39; P less than .0001). After second vaccine dose, 85% of infliximab- and 86% of vedolizumab-treated patients seroconverted (P = .68).
Study details: Findings are from the CLARITY IBD study that included patients with IBD without evidence of prior SARS-CoV-2 infection who were treated with infliximab (n=865) or vedolizumab (n=428).
Disclosures: The study was funded by F. Hoffmann-La Roche AG (Switzerland), Biogen GmbH (Switzerland), Celltrion Healthcare (South Korea), and others. Some of the authors reported receiving grants, personal fees, and/or financial or nonfinancial support from multiple sources.
Source: Kennedy NA et al. Gut. 2021 Apr 26. doi: 10.1136/gutjnl-2021-324789.
Minnesota named best place to practice in 2021
For physicians who are just starting out or thinking about moving, the “Land of 10,000 Lakes” could be the land of opportunity, according to a recent Medscape analysis.
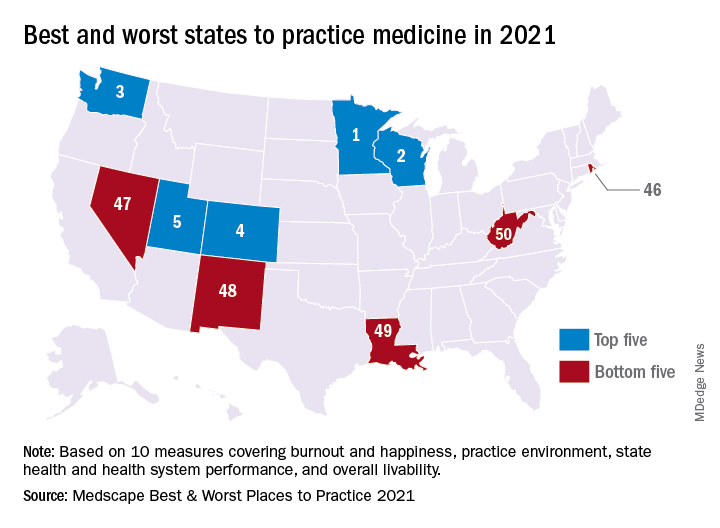
In a ranking of the 50 states, Minnesota “claimed top marks for livability, low incidence of adverse actions against doctors, and the performance of its health system,” Shelly Reese wrote in Medscape’s “Best & Worst Places to Practice 2021.”
Minnesota is below average where it’s good to be below average – share of physicians reporting burnout and/or depression – but above average in the share of physicians who say they’re “very happy” outside of work, Medscape said in the annual report.
and adverse actions and a high level of livability. Third place went to Washington (called the most livable state in the country by U.S. News and World Report), fourth to Colorado (physicians happy at and outside of work, high retention rate for residents), and fifth to Utah (low crime rate, high quality of life), Medscape said.
At the bottom of the list for 2021 is West Virginia, where physicians “may confront a bevy of challenges” in the form of low livability, a high rate of adverse actions, and relatively high malpractice payouts, Ms. Reese noted in the report.
State number 49 is Louisiana, where livability is low, malpractice payouts are high, and more than half of physicians say that they’re burned out and/or depressed. New Mexico is 48th (very high rate of adverse actions, poor resident retention), Nevada is 47th (low marks for avoidable hospital use and disparity in care), and Rhode Island is 46th (high malpractice payouts, low physician compensation), Medscape said.
Continuing with the group-of-five theme, America’s three most populous states finished in the top half of the ranking – California 16th, Texas 11th, and Florida 21st – but New York and Pennsylvania, numbers four and five by population size, did not.
The rankings are based on states’ performance in 10 different measures, three of which were sourced from Medscape surveys – happiness at work, happiness outside of work, and burnout/depression – and seven from other organizations: adverse actions against physicians, malpractice payouts, compensation (adjusted for cost of living), overall health, health system performance, overall livability, resident retention.
For physicians who are just starting out or thinking about moving, the “Land of 10,000 Lakes” could be the land of opportunity, according to a recent Medscape analysis.

In a ranking of the 50 states, Minnesota “claimed top marks for livability, low incidence of adverse actions against doctors, and the performance of its health system,” Shelly Reese wrote in Medscape’s “Best & Worst Places to Practice 2021.”
Minnesota is below average where it’s good to be below average – share of physicians reporting burnout and/or depression – but above average in the share of physicians who say they’re “very happy” outside of work, Medscape said in the annual report.
and adverse actions and a high level of livability. Third place went to Washington (called the most livable state in the country by U.S. News and World Report), fourth to Colorado (physicians happy at and outside of work, high retention rate for residents), and fifth to Utah (low crime rate, high quality of life), Medscape said.
At the bottom of the list for 2021 is West Virginia, where physicians “may confront a bevy of challenges” in the form of low livability, a high rate of adverse actions, and relatively high malpractice payouts, Ms. Reese noted in the report.
State number 49 is Louisiana, where livability is low, malpractice payouts are high, and more than half of physicians say that they’re burned out and/or depressed. New Mexico is 48th (very high rate of adverse actions, poor resident retention), Nevada is 47th (low marks for avoidable hospital use and disparity in care), and Rhode Island is 46th (high malpractice payouts, low physician compensation), Medscape said.
Continuing with the group-of-five theme, America’s three most populous states finished in the top half of the ranking – California 16th, Texas 11th, and Florida 21st – but New York and Pennsylvania, numbers four and five by population size, did not.
The rankings are based on states’ performance in 10 different measures, three of which were sourced from Medscape surveys – happiness at work, happiness outside of work, and burnout/depression – and seven from other organizations: adverse actions against physicians, malpractice payouts, compensation (adjusted for cost of living), overall health, health system performance, overall livability, resident retention.
For physicians who are just starting out or thinking about moving, the “Land of 10,000 Lakes” could be the land of opportunity, according to a recent Medscape analysis.

In a ranking of the 50 states, Minnesota “claimed top marks for livability, low incidence of adverse actions against doctors, and the performance of its health system,” Shelly Reese wrote in Medscape’s “Best & Worst Places to Practice 2021.”
Minnesota is below average where it’s good to be below average – share of physicians reporting burnout and/or depression – but above average in the share of physicians who say they’re “very happy” outside of work, Medscape said in the annual report.
and adverse actions and a high level of livability. Third place went to Washington (called the most livable state in the country by U.S. News and World Report), fourth to Colorado (physicians happy at and outside of work, high retention rate for residents), and fifth to Utah (low crime rate, high quality of life), Medscape said.
At the bottom of the list for 2021 is West Virginia, where physicians “may confront a bevy of challenges” in the form of low livability, a high rate of adverse actions, and relatively high malpractice payouts, Ms. Reese noted in the report.
State number 49 is Louisiana, where livability is low, malpractice payouts are high, and more than half of physicians say that they’re burned out and/or depressed. New Mexico is 48th (very high rate of adverse actions, poor resident retention), Nevada is 47th (low marks for avoidable hospital use and disparity in care), and Rhode Island is 46th (high malpractice payouts, low physician compensation), Medscape said.
Continuing with the group-of-five theme, America’s three most populous states finished in the top half of the ranking – California 16th, Texas 11th, and Florida 21st – but New York and Pennsylvania, numbers four and five by population size, did not.
The rankings are based on states’ performance in 10 different measures, three of which were sourced from Medscape surveys – happiness at work, happiness outside of work, and burnout/depression – and seven from other organizations: adverse actions against physicians, malpractice payouts, compensation (adjusted for cost of living), overall health, health system performance, overall livability, resident retention.
Clinical Edge Journal Scan Commentary: IBD June 2021
U.S. News releases Best Children’s Hospitals list, with changes
Released June 15, the 2021-2022 rankings, which acknowledge 50 U.S. centers for delivering exceptional care in several specialties, also give the Massachusetts hospital the top spot in 4 of 10 pediatric specialties assessed: nephrology, neurology and neurosurgery, pulmonology and lung surgery, and urology.
Children’s Hospital of Philadelphia retains second spot in the annually updated list, and Texas Children’s Hospital, in Houston, moves up a rung to third place, bumping Cincinnati Children’s Hospital Medical Center from third to fourth place. Children’s Hospital Los Angeles comes in at no. 5.
The remaining top 10 placements, in descending order, are as follows:
Children’s Hospital Colorado in Aurora; Children’s National Hospital in Washington; Nationwide Children’s Hospital in Columbus, Ohio; UPMS Children’s Hospital of Pittsburgh; and Lucile Packard Children’s Hospital Stanford (Calif.).
New regional rankings
This year’s edition offers something new, adding rankings within states and multiple-state rankings within seven regions to facilitate choice. “The Best Children’s Hospitals rankings have always highlighted hospitals that excel in specialized care,” said Ben Harder, chief of health analysis and managing editor at U.S. News, in a press release. “Now, this year’s new state and regional rankings can help families identify conveniently located hospitals capable of meeting their child’s needs. As the pandemic continues to affect travel, finding high-quality care close to home has never been more important.”
Across the seven regions, the top-ranked institutions are as follows:
- Mid-Atlantic – Children’s Hospital of Philadelphia.
- Midwest – Cincinnati Children’s Hospital Medical Center.
- New England – Boston Children’s Hospital.
- Pacific – Children’s Hospital Los Angeles.
- Rocky Mountains – Children’s Hospital Colorado.
- Southeast – Children’s Healthcare of Atlanta and Monroe Carell Jr. Children’s Hospital of Vanderbilt, in Nashville, Tenn.
- Southwest – Texas Children’s Hospital.
Specialties
Boston Children’s not only topped the overall list but also led in four specialties. For the other six specialties that were ranked, the top hospitals on the honor roll are as follows:
- Cancer – Children’s Hospital of Philadelphia.
- Cardiology and heart surgery – Texas Children’s Hospital.
- Diabetes and endocrinology – Children’s Hospital of Philadelphia.
- Gastroenterology and gastrointestinal surgery – Children’s Hospital Colorado.
- Neonatology – Children’s National Hospital.
- Orthopedics – Children’s Hospital of Philadelphia.
For the past 15 years, the objective of the rankings has been to offer a starting point for parents in making decisions about the best place to take very sick children for high-quality care. The editors of the rankings acknowledge that considerations of travel costs and insurance coverage are other factors to consider.
Helpful for families
The rankings are helpful for families, according to Joe W. St. Geme, III, MD, Children’s Hospital of Philadelphia’s physician-in-chief and chair of its department of pediatrics. “Some parents, especially those coming from outside an area, find them useful when deciding on care away from home,” he told this news organization. “Most types of pediatric care are available in the community, but sometimes a child has an unusual disease or complex disease for which local care is not available.”
Dr. St. Geme said the new regional rankings may be useful in helping parents decide where to bring a child for care that is closer to where they live.
A top ranking from U.S. News is just one indication of a hospital›s overall performance, according to Angela Lorts, MD, MBA, director of the Ventricular Assist Device Program, at Cincinnati Children’s Hospital Medical Center.
“Parents seeking care for their child should use the data to ask questions and understand the limitations,” she told this news organization. “Rankings are only based on a small subset of the children we care for. Many of the metrics may not pertain to their child and may not reflect the care they will receive.”
In her view, ranking will not give parents all the information they need about medical care and outcomes for specific conditions.
Hospital reaction
Hospitals can use the rankings to target improvements, says Dr. St. Geme. “These rankings can provide an opportunity for some benchmarking, to see what other institutions are doing and how they’re able to deliver care. They can serve as a source of ideas and can influence planning,” he said.
He cautioned that the data are not as complete as they could be. “A number of services are not included, and we try to keep that in mind,” he said.
Rankings may also affect recruitment, Dr. St. Geme added, because higher-ranked institutions may find it easier to attract sought-after clinicians and investigators in needed areas.
Another sphere of influence is philanthropy and fund raising. “People are much more likely to consider making both small and large donations to a high-ranked institution,” said J. Howard Smart, MD, chair of pediatrics at Sharp Rees-Stealy Medical Group and chair-elect of the physician leadership council at Sharp Mary Birch Hospital for Women and Newborns in San Diego.
Dr. St. Geme agrees. “Philanthropists are interested in making investments where they feel they’re a sure bet, and rankings may indicate a sure bet. But their impact on government funding and grant support is probably less.”
Ultimately, however, some families may not have lot of choice in where to go when their children are sick, Dr. Smart said. “And people probably don’t choose a location to live in based on nearby children’s hospitals the way they do for schools,” he said.
What about hospitals that continue to rank much lower on the 50-institution list – excellent though they must be to make it onto the honor roll. “To be on the list but not to have risen in rank in recent years might be a disappointment,” said Dr. St. Geme. “But it might also motivate a hospital to think about making internal investments in order to strengthen a particular service. And it may motivate nonranked hospitals to improve care in order to break into the list.”
Dr. Lorts points out that the annual survey process requires hospitals to track the clinical outcomes of a subset of patients, which may lead to improvement in these areas. It also requires data collection on structure and process, which drives needs assessments of select hospital areas. “But ideally, all hospitals would be tracking important outcomes, benchmarking to peer hospitals, and improving where needed without the U.S. News incentive,” she said.
This year’s data, compiled by research and consulting firm RTI International, derive from feedback on more than 1,200 questions provided by 118 responding institutions. Details on each hospital on the list and the methodology used in the analysis are available on U.S. News & World Report’s website.
A version of this article first appeared on Medscape.com.
Released June 15, the 2021-2022 rankings, which acknowledge 50 U.S. centers for delivering exceptional care in several specialties, also give the Massachusetts hospital the top spot in 4 of 10 pediatric specialties assessed: nephrology, neurology and neurosurgery, pulmonology and lung surgery, and urology.
Children’s Hospital of Philadelphia retains second spot in the annually updated list, and Texas Children’s Hospital, in Houston, moves up a rung to third place, bumping Cincinnati Children’s Hospital Medical Center from third to fourth place. Children’s Hospital Los Angeles comes in at no. 5.
The remaining top 10 placements, in descending order, are as follows:
Children’s Hospital Colorado in Aurora; Children’s National Hospital in Washington; Nationwide Children’s Hospital in Columbus, Ohio; UPMS Children’s Hospital of Pittsburgh; and Lucile Packard Children’s Hospital Stanford (Calif.).
New regional rankings
This year’s edition offers something new, adding rankings within states and multiple-state rankings within seven regions to facilitate choice. “The Best Children’s Hospitals rankings have always highlighted hospitals that excel in specialized care,” said Ben Harder, chief of health analysis and managing editor at U.S. News, in a press release. “Now, this year’s new state and regional rankings can help families identify conveniently located hospitals capable of meeting their child’s needs. As the pandemic continues to affect travel, finding high-quality care close to home has never been more important.”
Across the seven regions, the top-ranked institutions are as follows:
- Mid-Atlantic – Children’s Hospital of Philadelphia.
- Midwest – Cincinnati Children’s Hospital Medical Center.
- New England – Boston Children’s Hospital.
- Pacific – Children’s Hospital Los Angeles.
- Rocky Mountains – Children’s Hospital Colorado.
- Southeast – Children’s Healthcare of Atlanta and Monroe Carell Jr. Children’s Hospital of Vanderbilt, in Nashville, Tenn.
- Southwest – Texas Children’s Hospital.
Specialties
Boston Children’s not only topped the overall list but also led in four specialties. For the other six specialties that were ranked, the top hospitals on the honor roll are as follows:
- Cancer – Children’s Hospital of Philadelphia.
- Cardiology and heart surgery – Texas Children’s Hospital.
- Diabetes and endocrinology – Children’s Hospital of Philadelphia.
- Gastroenterology and gastrointestinal surgery – Children’s Hospital Colorado.
- Neonatology – Children’s National Hospital.
- Orthopedics – Children’s Hospital of Philadelphia.
For the past 15 years, the objective of the rankings has been to offer a starting point for parents in making decisions about the best place to take very sick children for high-quality care. The editors of the rankings acknowledge that considerations of travel costs and insurance coverage are other factors to consider.
Helpful for families
The rankings are helpful for families, according to Joe W. St. Geme, III, MD, Children’s Hospital of Philadelphia’s physician-in-chief and chair of its department of pediatrics. “Some parents, especially those coming from outside an area, find them useful when deciding on care away from home,” he told this news organization. “Most types of pediatric care are available in the community, but sometimes a child has an unusual disease or complex disease for which local care is not available.”
Dr. St. Geme said the new regional rankings may be useful in helping parents decide where to bring a child for care that is closer to where they live.
A top ranking from U.S. News is just one indication of a hospital›s overall performance, according to Angela Lorts, MD, MBA, director of the Ventricular Assist Device Program, at Cincinnati Children’s Hospital Medical Center.
“Parents seeking care for their child should use the data to ask questions and understand the limitations,” she told this news organization. “Rankings are only based on a small subset of the children we care for. Many of the metrics may not pertain to their child and may not reflect the care they will receive.”
In her view, ranking will not give parents all the information they need about medical care and outcomes for specific conditions.
Hospital reaction
Hospitals can use the rankings to target improvements, says Dr. St. Geme. “These rankings can provide an opportunity for some benchmarking, to see what other institutions are doing and how they’re able to deliver care. They can serve as a source of ideas and can influence planning,” he said.
He cautioned that the data are not as complete as they could be. “A number of services are not included, and we try to keep that in mind,” he said.
Rankings may also affect recruitment, Dr. St. Geme added, because higher-ranked institutions may find it easier to attract sought-after clinicians and investigators in needed areas.
Another sphere of influence is philanthropy and fund raising. “People are much more likely to consider making both small and large donations to a high-ranked institution,” said J. Howard Smart, MD, chair of pediatrics at Sharp Rees-Stealy Medical Group and chair-elect of the physician leadership council at Sharp Mary Birch Hospital for Women and Newborns in San Diego.
Dr. St. Geme agrees. “Philanthropists are interested in making investments where they feel they’re a sure bet, and rankings may indicate a sure bet. But their impact on government funding and grant support is probably less.”
Ultimately, however, some families may not have lot of choice in where to go when their children are sick, Dr. Smart said. “And people probably don’t choose a location to live in based on nearby children’s hospitals the way they do for schools,” he said.
What about hospitals that continue to rank much lower on the 50-institution list – excellent though they must be to make it onto the honor roll. “To be on the list but not to have risen in rank in recent years might be a disappointment,” said Dr. St. Geme. “But it might also motivate a hospital to think about making internal investments in order to strengthen a particular service. And it may motivate nonranked hospitals to improve care in order to break into the list.”
Dr. Lorts points out that the annual survey process requires hospitals to track the clinical outcomes of a subset of patients, which may lead to improvement in these areas. It also requires data collection on structure and process, which drives needs assessments of select hospital areas. “But ideally, all hospitals would be tracking important outcomes, benchmarking to peer hospitals, and improving where needed without the U.S. News incentive,” she said.
This year’s data, compiled by research and consulting firm RTI International, derive from feedback on more than 1,200 questions provided by 118 responding institutions. Details on each hospital on the list and the methodology used in the analysis are available on U.S. News & World Report’s website.
A version of this article first appeared on Medscape.com.
Released June 15, the 2021-2022 rankings, which acknowledge 50 U.S. centers for delivering exceptional care in several specialties, also give the Massachusetts hospital the top spot in 4 of 10 pediatric specialties assessed: nephrology, neurology and neurosurgery, pulmonology and lung surgery, and urology.
Children’s Hospital of Philadelphia retains second spot in the annually updated list, and Texas Children’s Hospital, in Houston, moves up a rung to third place, bumping Cincinnati Children’s Hospital Medical Center from third to fourth place. Children’s Hospital Los Angeles comes in at no. 5.
The remaining top 10 placements, in descending order, are as follows:
Children’s Hospital Colorado in Aurora; Children’s National Hospital in Washington; Nationwide Children’s Hospital in Columbus, Ohio; UPMS Children’s Hospital of Pittsburgh; and Lucile Packard Children’s Hospital Stanford (Calif.).
New regional rankings
This year’s edition offers something new, adding rankings within states and multiple-state rankings within seven regions to facilitate choice. “The Best Children’s Hospitals rankings have always highlighted hospitals that excel in specialized care,” said Ben Harder, chief of health analysis and managing editor at U.S. News, in a press release. “Now, this year’s new state and regional rankings can help families identify conveniently located hospitals capable of meeting their child’s needs. As the pandemic continues to affect travel, finding high-quality care close to home has never been more important.”
Across the seven regions, the top-ranked institutions are as follows:
- Mid-Atlantic – Children’s Hospital of Philadelphia.
- Midwest – Cincinnati Children’s Hospital Medical Center.
- New England – Boston Children’s Hospital.
- Pacific – Children’s Hospital Los Angeles.
- Rocky Mountains – Children’s Hospital Colorado.
- Southeast – Children’s Healthcare of Atlanta and Monroe Carell Jr. Children’s Hospital of Vanderbilt, in Nashville, Tenn.
- Southwest – Texas Children’s Hospital.
Specialties
Boston Children’s not only topped the overall list but also led in four specialties. For the other six specialties that were ranked, the top hospitals on the honor roll are as follows:
- Cancer – Children’s Hospital of Philadelphia.
- Cardiology and heart surgery – Texas Children’s Hospital.
- Diabetes and endocrinology – Children’s Hospital of Philadelphia.
- Gastroenterology and gastrointestinal surgery – Children’s Hospital Colorado.
- Neonatology – Children’s National Hospital.
- Orthopedics – Children’s Hospital of Philadelphia.
For the past 15 years, the objective of the rankings has been to offer a starting point for parents in making decisions about the best place to take very sick children for high-quality care. The editors of the rankings acknowledge that considerations of travel costs and insurance coverage are other factors to consider.
Helpful for families
The rankings are helpful for families, according to Joe W. St. Geme, III, MD, Children’s Hospital of Philadelphia’s physician-in-chief and chair of its department of pediatrics. “Some parents, especially those coming from outside an area, find them useful when deciding on care away from home,” he told this news organization. “Most types of pediatric care are available in the community, but sometimes a child has an unusual disease or complex disease for which local care is not available.”
Dr. St. Geme said the new regional rankings may be useful in helping parents decide where to bring a child for care that is closer to where they live.
A top ranking from U.S. News is just one indication of a hospital›s overall performance, according to Angela Lorts, MD, MBA, director of the Ventricular Assist Device Program, at Cincinnati Children’s Hospital Medical Center.
“Parents seeking care for their child should use the data to ask questions and understand the limitations,” she told this news organization. “Rankings are only based on a small subset of the children we care for. Many of the metrics may not pertain to their child and may not reflect the care they will receive.”
In her view, ranking will not give parents all the information they need about medical care and outcomes for specific conditions.
Hospital reaction
Hospitals can use the rankings to target improvements, says Dr. St. Geme. “These rankings can provide an opportunity for some benchmarking, to see what other institutions are doing and how they’re able to deliver care. They can serve as a source of ideas and can influence planning,” he said.
He cautioned that the data are not as complete as they could be. “A number of services are not included, and we try to keep that in mind,” he said.
Rankings may also affect recruitment, Dr. St. Geme added, because higher-ranked institutions may find it easier to attract sought-after clinicians and investigators in needed areas.
Another sphere of influence is philanthropy and fund raising. “People are much more likely to consider making both small and large donations to a high-ranked institution,” said J. Howard Smart, MD, chair of pediatrics at Sharp Rees-Stealy Medical Group and chair-elect of the physician leadership council at Sharp Mary Birch Hospital for Women and Newborns in San Diego.
Dr. St. Geme agrees. “Philanthropists are interested in making investments where they feel they’re a sure bet, and rankings may indicate a sure bet. But their impact on government funding and grant support is probably less.”
Ultimately, however, some families may not have lot of choice in where to go when their children are sick, Dr. Smart said. “And people probably don’t choose a location to live in based on nearby children’s hospitals the way they do for schools,” he said.
What about hospitals that continue to rank much lower on the 50-institution list – excellent though they must be to make it onto the honor roll. “To be on the list but not to have risen in rank in recent years might be a disappointment,” said Dr. St. Geme. “But it might also motivate a hospital to think about making internal investments in order to strengthen a particular service. And it may motivate nonranked hospitals to improve care in order to break into the list.”
Dr. Lorts points out that the annual survey process requires hospitals to track the clinical outcomes of a subset of patients, which may lead to improvement in these areas. It also requires data collection on structure and process, which drives needs assessments of select hospital areas. “But ideally, all hospitals would be tracking important outcomes, benchmarking to peer hospitals, and improving where needed without the U.S. News incentive,” she said.
This year’s data, compiled by research and consulting firm RTI International, derive from feedback on more than 1,200 questions provided by 118 responding institutions. Details on each hospital on the list and the methodology used in the analysis are available on U.S. News & World Report’s website.
A version of this article first appeared on Medscape.com.
Third COVID-19 vaccine dose helped some transplant recipients
All of those with low titers before the third dose had high titers after receiving the additional shot, but only about 33% of those with negative initial responses had detectable antibodies after the third dose, according to the paper, published in Annals of Internal Medicine.
Researchers at Johns Hopkins, Baltimore, who keep a COVID-19 vaccine registry, perform antibody tests on all registry subjects and inform them of their results. Registry participants were asked to inform the research team if they received a third dose, and, the research team tracked the immune responses of those who did.
The participants in this case series had low antibody levels and received a third dose of the vaccine on their own between March 20 and May 10 of 2021.
Third dose results
In this cases series – thought to be the first to look at third vaccine shots in this type of patient group – all six of those who had low antibody titers before the third dose had high-positive titers after the third dose.
Of the 24 individuals who had negative antibody titers before the third dose, just 6 had high titers after the third dose.
Two of the participants had low-positive titers, and 16 were negative.
“Several of those boosted very nicely into ranges seen, using these assays, in healthy persons,” said William Werbel, MD, a fellow in infectious disease at Johns Hopkins Medicine, Baltimore, who helped lead the study. Those with negative levels, even if they responded, tended to have lower titers, he said.
“The benefits at least from an antibody perspective were not the same for everybody and so this is obviously something that needs to be considered when thinking about selecting patients” for a COVID-19 prevention strategy, he said.
Reactions to the vaccine were low to moderate, such as some arm pain and fatigue.
“Showing that something is safe in that special, vulnerable population is important,” Dr. Werbel said. “We’re all wanting to make sure that we’re doing no harm.”
Dr. Werbel noted that there was no pattern in the small series based on the organ transplanted or in the vaccines used. As their third shot, 15 of the patients received the Johnson & Johnson vaccine; 9 received Moderna; and 6 received Pfizer-BioNTech.
Welcome news, but larger studies needed
“To think that a third dose could confer protection for a significant number of people is of course extremely welcome news,” said Christian Larsen, MD, DPhil, professor of surgery in the transplantation division at Emory University, Atlanta, who was not involved in the study. “It’s the easiest conceivable next intervention.”
He added, “We just want studies to confirm that – larger studies.”
Dr. Werbel stressed the importance of looking at third doses in these patients in a more controlled fashion in a randomized trial, to more carefully monitor safety and how patients fare when starting with one type of vaccine and switching to another, for example.
Richard Wender, MD, chair of family medicine and community health at the University of Pennsylvania, Philadelphia, said the findings are a reminder that there is still a lot that is unknown about COVID-19 and vaccination.
“We still don’t know who will or will not benefit from a third dose,” he said. “And our knowledge is evolving. For example, a recent study suggested that people with previous infection and who are vaccinated may have better and longer protection than people with vaccination alone. We’re still learning.”
He added that specialists, not primary care clinicians, should be relied upon to respond to this emerging vaccination data. Primary care doctors are very busy in other ways – such as in getting children caught up on vaccinations and helping adults return to managing their chronic diseases, Dr. Wender noted.
“Their focus needs to be on helping to overcome hesitancy, mistrust, lack of information, or antivaccination sentiment to help more people feel comfortable being vaccinated – this is a lot of work and needs constant focus. In short, primary care clinicians need to focus chiefly on the unvaccinated,” he said.
“Monitoring immunization recommendations for unique at-risk populations should be the chief responsibility of teams providing subspecialty care, [such as for] transplant patients, people with chronic kidney disease, cancer patients, and people with other chronic illnesses. This will allow primary care clinicians to tackle their many complex jobs.”
Possible solutions for those with low antibody responses
Dr. Larsen said that those with ongoing low antibody responses might still have other immune responses, such as a T-cell response. Such patients also could consider changing their vaccine type, he said.
“At the more significant intervention level, there may be circumstances where one could change the immunosuppressive drugs in a controlled way that might allow a better response,” suggested Dr. Larsen. “That’s obviously going to be something that requires a lot more thought and careful study.”
Dr. Werbel said that other options might need to be considered for those having no response following a third dose. One possibility is trying a vaccine with an adjuvant, such as the Novavax version, which might be more widely available soon.
“If you’re given a third dose of a very immunogenic vaccine – something that should work – and you just have no antibody development, it seems relatively unlikely that doing the same thing again is going to help you from that perspective, and for all we know might expose you to more risk,” Dr. Werbel noted.
Participant details
None of the 30 patients were thought to have ever had COVID-19. On average, patients had received their transplant 4.5 years before their original vaccination. In 25 patients, maintenance immunosuppression included tacrolimus or cyclosporine along with mycophenolate. Corticosteroids were also used for 24 patients, sirolimus was used for one patient, and belatacept was used for another patient.
Fifty-seven percent of patients had received the Pfizer/BioNTech vaccine originally, and 43% the Moderna vaccine. Most of the patients were kidney recipients, with two heart, three liver, one lung, one pancreas and one kidney-pancreas.
Dr. Werbel, Dr. Wender, and Dr. Larsen reported no relevant disclosures.
All of those with low titers before the third dose had high titers after receiving the additional shot, but only about 33% of those with negative initial responses had detectable antibodies after the third dose, according to the paper, published in Annals of Internal Medicine.
Researchers at Johns Hopkins, Baltimore, who keep a COVID-19 vaccine registry, perform antibody tests on all registry subjects and inform them of their results. Registry participants were asked to inform the research team if they received a third dose, and, the research team tracked the immune responses of those who did.
The participants in this case series had low antibody levels and received a third dose of the vaccine on their own between March 20 and May 10 of 2021.
Third dose results
In this cases series – thought to be the first to look at third vaccine shots in this type of patient group – all six of those who had low antibody titers before the third dose had high-positive titers after the third dose.
Of the 24 individuals who had negative antibody titers before the third dose, just 6 had high titers after the third dose.
Two of the participants had low-positive titers, and 16 were negative.
“Several of those boosted very nicely into ranges seen, using these assays, in healthy persons,” said William Werbel, MD, a fellow in infectious disease at Johns Hopkins Medicine, Baltimore, who helped lead the study. Those with negative levels, even if they responded, tended to have lower titers, he said.
“The benefits at least from an antibody perspective were not the same for everybody and so this is obviously something that needs to be considered when thinking about selecting patients” for a COVID-19 prevention strategy, he said.
Reactions to the vaccine were low to moderate, such as some arm pain and fatigue.
“Showing that something is safe in that special, vulnerable population is important,” Dr. Werbel said. “We’re all wanting to make sure that we’re doing no harm.”
Dr. Werbel noted that there was no pattern in the small series based on the organ transplanted or in the vaccines used. As their third shot, 15 of the patients received the Johnson & Johnson vaccine; 9 received Moderna; and 6 received Pfizer-BioNTech.
Welcome news, but larger studies needed
“To think that a third dose could confer protection for a significant number of people is of course extremely welcome news,” said Christian Larsen, MD, DPhil, professor of surgery in the transplantation division at Emory University, Atlanta, who was not involved in the study. “It’s the easiest conceivable next intervention.”
He added, “We just want studies to confirm that – larger studies.”
Dr. Werbel stressed the importance of looking at third doses in these patients in a more controlled fashion in a randomized trial, to more carefully monitor safety and how patients fare when starting with one type of vaccine and switching to another, for example.
Richard Wender, MD, chair of family medicine and community health at the University of Pennsylvania, Philadelphia, said the findings are a reminder that there is still a lot that is unknown about COVID-19 and vaccination.
“We still don’t know who will or will not benefit from a third dose,” he said. “And our knowledge is evolving. For example, a recent study suggested that people with previous infection and who are vaccinated may have better and longer protection than people with vaccination alone. We’re still learning.”
He added that specialists, not primary care clinicians, should be relied upon to respond to this emerging vaccination data. Primary care doctors are very busy in other ways – such as in getting children caught up on vaccinations and helping adults return to managing their chronic diseases, Dr. Wender noted.
“Their focus needs to be on helping to overcome hesitancy, mistrust, lack of information, or antivaccination sentiment to help more people feel comfortable being vaccinated – this is a lot of work and needs constant focus. In short, primary care clinicians need to focus chiefly on the unvaccinated,” he said.
“Monitoring immunization recommendations for unique at-risk populations should be the chief responsibility of teams providing subspecialty care, [such as for] transplant patients, people with chronic kidney disease, cancer patients, and people with other chronic illnesses. This will allow primary care clinicians to tackle their many complex jobs.”
Possible solutions for those with low antibody responses
Dr. Larsen said that those with ongoing low antibody responses might still have other immune responses, such as a T-cell response. Such patients also could consider changing their vaccine type, he said.
“At the more significant intervention level, there may be circumstances where one could change the immunosuppressive drugs in a controlled way that might allow a better response,” suggested Dr. Larsen. “That’s obviously going to be something that requires a lot more thought and careful study.”
Dr. Werbel said that other options might need to be considered for those having no response following a third dose. One possibility is trying a vaccine with an adjuvant, such as the Novavax version, which might be more widely available soon.
“If you’re given a third dose of a very immunogenic vaccine – something that should work – and you just have no antibody development, it seems relatively unlikely that doing the same thing again is going to help you from that perspective, and for all we know might expose you to more risk,” Dr. Werbel noted.
Participant details
None of the 30 patients were thought to have ever had COVID-19. On average, patients had received their transplant 4.5 years before their original vaccination. In 25 patients, maintenance immunosuppression included tacrolimus or cyclosporine along with mycophenolate. Corticosteroids were also used for 24 patients, sirolimus was used for one patient, and belatacept was used for another patient.
Fifty-seven percent of patients had received the Pfizer/BioNTech vaccine originally, and 43% the Moderna vaccine. Most of the patients were kidney recipients, with two heart, three liver, one lung, one pancreas and one kidney-pancreas.
Dr. Werbel, Dr. Wender, and Dr. Larsen reported no relevant disclosures.
All of those with low titers before the third dose had high titers after receiving the additional shot, but only about 33% of those with negative initial responses had detectable antibodies after the third dose, according to the paper, published in Annals of Internal Medicine.
Researchers at Johns Hopkins, Baltimore, who keep a COVID-19 vaccine registry, perform antibody tests on all registry subjects and inform them of their results. Registry participants were asked to inform the research team if they received a third dose, and, the research team tracked the immune responses of those who did.
The participants in this case series had low antibody levels and received a third dose of the vaccine on their own between March 20 and May 10 of 2021.
Third dose results
In this cases series – thought to be the first to look at third vaccine shots in this type of patient group – all six of those who had low antibody titers before the third dose had high-positive titers after the third dose.
Of the 24 individuals who had negative antibody titers before the third dose, just 6 had high titers after the third dose.
Two of the participants had low-positive titers, and 16 were negative.
“Several of those boosted very nicely into ranges seen, using these assays, in healthy persons,” said William Werbel, MD, a fellow in infectious disease at Johns Hopkins Medicine, Baltimore, who helped lead the study. Those with negative levels, even if they responded, tended to have lower titers, he said.
“The benefits at least from an antibody perspective were not the same for everybody and so this is obviously something that needs to be considered when thinking about selecting patients” for a COVID-19 prevention strategy, he said.
Reactions to the vaccine were low to moderate, such as some arm pain and fatigue.
“Showing that something is safe in that special, vulnerable population is important,” Dr. Werbel said. “We’re all wanting to make sure that we’re doing no harm.”
Dr. Werbel noted that there was no pattern in the small series based on the organ transplanted or in the vaccines used. As their third shot, 15 of the patients received the Johnson & Johnson vaccine; 9 received Moderna; and 6 received Pfizer-BioNTech.
Welcome news, but larger studies needed
“To think that a third dose could confer protection for a significant number of people is of course extremely welcome news,” said Christian Larsen, MD, DPhil, professor of surgery in the transplantation division at Emory University, Atlanta, who was not involved in the study. “It’s the easiest conceivable next intervention.”
He added, “We just want studies to confirm that – larger studies.”
Dr. Werbel stressed the importance of looking at third doses in these patients in a more controlled fashion in a randomized trial, to more carefully monitor safety and how patients fare when starting with one type of vaccine and switching to another, for example.
Richard Wender, MD, chair of family medicine and community health at the University of Pennsylvania, Philadelphia, said the findings are a reminder that there is still a lot that is unknown about COVID-19 and vaccination.
“We still don’t know who will or will not benefit from a third dose,” he said. “And our knowledge is evolving. For example, a recent study suggested that people with previous infection and who are vaccinated may have better and longer protection than people with vaccination alone. We’re still learning.”
He added that specialists, not primary care clinicians, should be relied upon to respond to this emerging vaccination data. Primary care doctors are very busy in other ways – such as in getting children caught up on vaccinations and helping adults return to managing their chronic diseases, Dr. Wender noted.
“Their focus needs to be on helping to overcome hesitancy, mistrust, lack of information, or antivaccination sentiment to help more people feel comfortable being vaccinated – this is a lot of work and needs constant focus. In short, primary care clinicians need to focus chiefly on the unvaccinated,” he said.
“Monitoring immunization recommendations for unique at-risk populations should be the chief responsibility of teams providing subspecialty care, [such as for] transplant patients, people with chronic kidney disease, cancer patients, and people with other chronic illnesses. This will allow primary care clinicians to tackle their many complex jobs.”
Possible solutions for those with low antibody responses
Dr. Larsen said that those with ongoing low antibody responses might still have other immune responses, such as a T-cell response. Such patients also could consider changing their vaccine type, he said.
“At the more significant intervention level, there may be circumstances where one could change the immunosuppressive drugs in a controlled way that might allow a better response,” suggested Dr. Larsen. “That’s obviously going to be something that requires a lot more thought and careful study.”
Dr. Werbel said that other options might need to be considered for those having no response following a third dose. One possibility is trying a vaccine with an adjuvant, such as the Novavax version, which might be more widely available soon.
“If you’re given a third dose of a very immunogenic vaccine – something that should work – and you just have no antibody development, it seems relatively unlikely that doing the same thing again is going to help you from that perspective, and for all we know might expose you to more risk,” Dr. Werbel noted.
Participant details
None of the 30 patients were thought to have ever had COVID-19. On average, patients had received their transplant 4.5 years before their original vaccination. In 25 patients, maintenance immunosuppression included tacrolimus or cyclosporine along with mycophenolate. Corticosteroids were also used for 24 patients, sirolimus was used for one patient, and belatacept was used for another patient.
Fifty-seven percent of patients had received the Pfizer/BioNTech vaccine originally, and 43% the Moderna vaccine. Most of the patients were kidney recipients, with two heart, three liver, one lung, one pancreas and one kidney-pancreas.
Dr. Werbel, Dr. Wender, and Dr. Larsen reported no relevant disclosures.
Women in GI: Career-spanning strategies to overcome gender bias
The gender gap in gastroenterology persists – currently, women constitute 39% of fellows, but only 22% of senior AGA members and less than 18% of all practicing gastroenterologists – and it has gained even greater significance within the “current historical moment” of the COVID pandemic and growing cognizance of systemic sexism and racism, according to experts.
During the pandemic, women have been more likely to stay home to care for ill family members and children affected by school closures, which increases their already disproportionate share of unpaid work, wrote Jessica Bernica, MD, of Baylor College of Medicine in Houston with her associates in Techniques and Innovations in Gastrointestinal Endoscopy. They noted that, according to one study, this “holds true for female physicians, who despite their more privileged positions, also experience higher demands at home, impacting their ability to contribute to teaching, service, and research.”
At the same time, the pandemic has brought into focus which jobs are “truly essential” – and that they are “overwhelmingly [held] by women and people of color, who are often underpaid and undervalued,” the experts wrote. The growing focus on systemic racism has also increased awareness of the chronic gender discrimination faced by female minorities, as well as by women in general, they added. In the field of gastroenterology, inherent gender bias – both systemic and self-directed – can bar women from advancing beginning as early as medical school.
To help address these issues, the experts outlined key opportunities for change as women navigate professional “forks in the road” throughout their careers.
Throughout their careers
During medical school and residency, women can specifically request gastroenterology rotations (“ideally with both inpatient and outpatient exposure”), attend society conferences, participate in research themselves, and join a research track or serve as chief medical resident. When applying for gastroenterology fellowships, they can prioritize programs with female faculty, which were recently found to be more likely to hire female fellows.
During fellowship, women can avail themselves of female mentors, who can help them strategize about ways to address gender bias, connect with GI groups and societies, and learn endoscopy techniques, including “unique approaches ... [that] overcome the challenges of standard scope sizes and accessibility.” At the institutional level, opportunities to affect positive changes for women trainees include “formal education on the benefits of hands-on learning and encouraging explicit and open communication between parties regarding invitation to, comfort with, and type of physical contact prior to a case.”
After fellowship, early-career gastroenterologists should scrutinize contracts for details on pay and research support, and they should ideally join a practice that either already has many women physicians on staff, or that ensures salary transparency and has “parental leave policies that are compatible with [applicants’] personal and professional goals.” But the experts advocated caution about part-time positions, which may purport to offer more flexibility but turn into full-time work for part-time pay and can preclude participation in practice management.
The experts recommended midcareer female gastroenterologists call out their own achievements rather than waiting for recognition, “actively seek promotion and tenure,” negotiate their salaries (as men tend to do routinely), and think twice before accepting professional roles that are uncompensated or do not clearly promote career advancement.
Senior gastroenterologists have unique opportunities to spearhead changes in institutional policies and practices, according to the experts. Specific examples include “explicitly stating [in job listings] that salary is negotiable, creating transparent written compensation plans, and conducting audits of job offers” to help mitigate any inequities in pay or hiring practices. In addition, senior women gastroenterologists can mentor individual women in the field, implement formal trainings on implicit bias, ensure that their practice or department tracks the gender of gastroenterologists who join, leave, or are promoted.
The experts did not report receiving funding for the work. They reported having no conflicts of interest.
Gastroenterology is a male-dominated field; women represent only 18% of current practicing gastroenterologists. Fortunately more women are entering medicine, including our field of gastroenterology, with current statistics showing that 39% of fellows are women. There have been historical barriers to women’s entry into the gastroenterology field, but thanks to the efforts of great female leaders in gastroenterology and men who are allies of women in our field, we have seen some of these barriers start to weaken. However, there is much work yet to be done. In fact, many would argue our work is just beginning.
Hopefully we will all learn something from Bernica and colleagues’ important piece and continue to sponsor and encourage women to practice this great field so that someday our workforce will look more like the patients we are caring for.
Laura E. Raffals, MD, is with the department of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. She has no conflicts of interest.
Gastroenterology is a male-dominated field; women represent only 18% of current practicing gastroenterologists. Fortunately more women are entering medicine, including our field of gastroenterology, with current statistics showing that 39% of fellows are women. There have been historical barriers to women’s entry into the gastroenterology field, but thanks to the efforts of great female leaders in gastroenterology and men who are allies of women in our field, we have seen some of these barriers start to weaken. However, there is much work yet to be done. In fact, many would argue our work is just beginning.
Hopefully we will all learn something from Bernica and colleagues’ important piece and continue to sponsor and encourage women to practice this great field so that someday our workforce will look more like the patients we are caring for.
Laura E. Raffals, MD, is with the department of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. She has no conflicts of interest.
Gastroenterology is a male-dominated field; women represent only 18% of current practicing gastroenterologists. Fortunately more women are entering medicine, including our field of gastroenterology, with current statistics showing that 39% of fellows are women. There have been historical barriers to women’s entry into the gastroenterology field, but thanks to the efforts of great female leaders in gastroenterology and men who are allies of women in our field, we have seen some of these barriers start to weaken. However, there is much work yet to be done. In fact, many would argue our work is just beginning.
Hopefully we will all learn something from Bernica and colleagues’ important piece and continue to sponsor and encourage women to practice this great field so that someday our workforce will look more like the patients we are caring for.
Laura E. Raffals, MD, is with the department of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn. She has no conflicts of interest.
The gender gap in gastroenterology persists – currently, women constitute 39% of fellows, but only 22% of senior AGA members and less than 18% of all practicing gastroenterologists – and it has gained even greater significance within the “current historical moment” of the COVID pandemic and growing cognizance of systemic sexism and racism, according to experts.
During the pandemic, women have been more likely to stay home to care for ill family members and children affected by school closures, which increases their already disproportionate share of unpaid work, wrote Jessica Bernica, MD, of Baylor College of Medicine in Houston with her associates in Techniques and Innovations in Gastrointestinal Endoscopy. They noted that, according to one study, this “holds true for female physicians, who despite their more privileged positions, also experience higher demands at home, impacting their ability to contribute to teaching, service, and research.”
At the same time, the pandemic has brought into focus which jobs are “truly essential” – and that they are “overwhelmingly [held] by women and people of color, who are often underpaid and undervalued,” the experts wrote. The growing focus on systemic racism has also increased awareness of the chronic gender discrimination faced by female minorities, as well as by women in general, they added. In the field of gastroenterology, inherent gender bias – both systemic and self-directed – can bar women from advancing beginning as early as medical school.
To help address these issues, the experts outlined key opportunities for change as women navigate professional “forks in the road” throughout their careers.
Throughout their careers
During medical school and residency, women can specifically request gastroenterology rotations (“ideally with both inpatient and outpatient exposure”), attend society conferences, participate in research themselves, and join a research track or serve as chief medical resident. When applying for gastroenterology fellowships, they can prioritize programs with female faculty, which were recently found to be more likely to hire female fellows.
During fellowship, women can avail themselves of female mentors, who can help them strategize about ways to address gender bias, connect with GI groups and societies, and learn endoscopy techniques, including “unique approaches ... [that] overcome the challenges of standard scope sizes and accessibility.” At the institutional level, opportunities to affect positive changes for women trainees include “formal education on the benefits of hands-on learning and encouraging explicit and open communication between parties regarding invitation to, comfort with, and type of physical contact prior to a case.”
After fellowship, early-career gastroenterologists should scrutinize contracts for details on pay and research support, and they should ideally join a practice that either already has many women physicians on staff, or that ensures salary transparency and has “parental leave policies that are compatible with [applicants’] personal and professional goals.” But the experts advocated caution about part-time positions, which may purport to offer more flexibility but turn into full-time work for part-time pay and can preclude participation in practice management.
The experts recommended midcareer female gastroenterologists call out their own achievements rather than waiting for recognition, “actively seek promotion and tenure,” negotiate their salaries (as men tend to do routinely), and think twice before accepting professional roles that are uncompensated or do not clearly promote career advancement.
Senior gastroenterologists have unique opportunities to spearhead changes in institutional policies and practices, according to the experts. Specific examples include “explicitly stating [in job listings] that salary is negotiable, creating transparent written compensation plans, and conducting audits of job offers” to help mitigate any inequities in pay or hiring practices. In addition, senior women gastroenterologists can mentor individual women in the field, implement formal trainings on implicit bias, ensure that their practice or department tracks the gender of gastroenterologists who join, leave, or are promoted.
The experts did not report receiving funding for the work. They reported having no conflicts of interest.
The gender gap in gastroenterology persists – currently, women constitute 39% of fellows, but only 22% of senior AGA members and less than 18% of all practicing gastroenterologists – and it has gained even greater significance within the “current historical moment” of the COVID pandemic and growing cognizance of systemic sexism and racism, according to experts.
During the pandemic, women have been more likely to stay home to care for ill family members and children affected by school closures, which increases their already disproportionate share of unpaid work, wrote Jessica Bernica, MD, of Baylor College of Medicine in Houston with her associates in Techniques and Innovations in Gastrointestinal Endoscopy. They noted that, according to one study, this “holds true for female physicians, who despite their more privileged positions, also experience higher demands at home, impacting their ability to contribute to teaching, service, and research.”
At the same time, the pandemic has brought into focus which jobs are “truly essential” – and that they are “overwhelmingly [held] by women and people of color, who are often underpaid and undervalued,” the experts wrote. The growing focus on systemic racism has also increased awareness of the chronic gender discrimination faced by female minorities, as well as by women in general, they added. In the field of gastroenterology, inherent gender bias – both systemic and self-directed – can bar women from advancing beginning as early as medical school.
To help address these issues, the experts outlined key opportunities for change as women navigate professional “forks in the road” throughout their careers.
Throughout their careers
During medical school and residency, women can specifically request gastroenterology rotations (“ideally with both inpatient and outpatient exposure”), attend society conferences, participate in research themselves, and join a research track or serve as chief medical resident. When applying for gastroenterology fellowships, they can prioritize programs with female faculty, which were recently found to be more likely to hire female fellows.
During fellowship, women can avail themselves of female mentors, who can help them strategize about ways to address gender bias, connect with GI groups and societies, and learn endoscopy techniques, including “unique approaches ... [that] overcome the challenges of standard scope sizes and accessibility.” At the institutional level, opportunities to affect positive changes for women trainees include “formal education on the benefits of hands-on learning and encouraging explicit and open communication between parties regarding invitation to, comfort with, and type of physical contact prior to a case.”
After fellowship, early-career gastroenterologists should scrutinize contracts for details on pay and research support, and they should ideally join a practice that either already has many women physicians on staff, or that ensures salary transparency and has “parental leave policies that are compatible with [applicants’] personal and professional goals.” But the experts advocated caution about part-time positions, which may purport to offer more flexibility but turn into full-time work for part-time pay and can preclude participation in practice management.
The experts recommended midcareer female gastroenterologists call out their own achievements rather than waiting for recognition, “actively seek promotion and tenure,” negotiate their salaries (as men tend to do routinely), and think twice before accepting professional roles that are uncompensated or do not clearly promote career advancement.
Senior gastroenterologists have unique opportunities to spearhead changes in institutional policies and practices, according to the experts. Specific examples include “explicitly stating [in job listings] that salary is negotiable, creating transparent written compensation plans, and conducting audits of job offers” to help mitigate any inequities in pay or hiring practices. In addition, senior women gastroenterologists can mentor individual women in the field, implement formal trainings on implicit bias, ensure that their practice or department tracks the gender of gastroenterologists who join, leave, or are promoted.
The experts did not report receiving funding for the work. They reported having no conflicts of interest.
FROM TECHNIQUES AND INNOVATIONS IN GASTROINTESTINAL ENDOSCOPY
Bariatric surgery tied to fewer HFpEF hospitalizations
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
Patients who underwent metabolic and bariatric surgery had fewer than half the number of hospitalizations for both acute and chronic episodes of heart failure with preserved ejection fraction (HFpEF) in a retrospective analysis of more than 2 million Americans collected in a national database.
In a multivariate analysis that adjusted for several variables patients without a history of bariatric surgery had three- to fivefold more hospitalizations for acute events involving HFpEF, and more than double the rate of hospitalizations for chronic HFpEF events, David R. Funes, MD, said at the annual meeting of the American Society for Metabolic and Bariatric Surgery.
While this analysis has the limitations of being retrospective, observational, and entirely reliant on procedure codes to define medical histories and outcomes, it had the advantage of using a large database designed to represent the U.S. adult population, said Dr. Funes, a bariatric surgeon at the Cleveland Clinic in Weston, Fla.
HFpEF effects could ‘extend’ surgery’s use
The report “adds an important article to the literature where there is a true void in trying to discern the effect of bariatric surgery on HFpEF,” commented Tammy L. Kindel, MD, PhD, director of the bariatric surgery program at the Medical College of Wisconsin, Milwaukee, and designated discussant for the report. “Minimal studies [up to now] demonstrate that weight loss in any form can modify diastolic dysfunction in patients with HFpEF. Studies that investigate the impact of bariatric surgery on clinical outcomes in patients with HFpEF are probably the most important for extending use of metabolic surgery,” Dr. Kindel said.
She added that “one of the most difficult parts of studying HFpEF” is making a firm diagnosis that often involves excluding other potential causes. She also questioned Dr. Funes about his confidence that his analysis correctly identified patients only with HFpEF. Dr. Funes replied that the diagnostic codes his team used allowed for a clear distinction between patients identified with HFpEF and those with heart failure with reduced ejection fraction, but he also admitted that his study’s complete reliance on these codes introduced a limitation to the analysis.
Including patients with diastolic dysfunction as well as HFpEF
The study used data collected during 2010-2015 by the National Inpatient Sample, run by the U.S. Department of Health & Human Services in a case-control analysis that included 296,041 patients who had undergone some form of bariatric surgery and 2,004,804 people with no history of bariatric surgery selected as controls on the basis of their obesity.
The absolute numbers showed that, during the observation period, the incidence of acute HFpEF hospitalizations was 0.19% among those with prior bariatric surgery and 0.86% among those with no surgery, and the incidence of chronic heart failure hospitalizations was 0.01% among people with prior bariatric surgery and 0.05% among those without prior surgery. Dr. Funes said. He noted that, during the period studied patients, with HFpEF were usually identified as having diastolic heart failure, an older name for the same disease.
In multivariate analyses that adjusted for age, sex, race, hypertension, diabetes, smoking, and coronary artery disease, people without prior bariatric surgery and with hypertension had a 2.8-fold increased rate of acute hospitalizations for HFpEF, while those without hypertension or prior bariatric surgery had a 5.2-fold increased rate. In addition, control patients, regardless of hypertension status, had a 2.9-fold increased rate of hospitalizations for chronic HFpEF events. All these differences were statistically significant.
Dr. Funes also reported results from additional analyses that focused on a roughly 68,000-patient subgroup of those included in the study who had a history of coronary artery disease, including about 62,000 with no prior bariatric surgery and nearly 6,000 people with prior bariatric surgery. In a multivariate analysis of this subgroup, people without prior bariatric surgery had a 2.65-fold increased rate of hospitalization for a HFpEF event (either acute or chronic), compared with those who had undergone bariatric surgery.
Dr. Funes and associates and Dr. Kindel had no relevant disclosures.
FROM ASMBS 2021
Risankizumab shows efficacy, tolerability in patients with PsA
Risankizumab (Skyrizi) was effective for treating psoriatic arthritis (PsA) in patients who did not respond to or who could not tolerate other biologics or standard disease-modifying antirheumatic drugs (DMARDs), according to a study presented at the annual European Congress of Rheumatology. It was also well tolerated.
“Treatment with risankizumab resulted in significantly greater improvements in signs and symptoms of psoriatic arthritis, including assessments of disease activity in joints and skin and patient-reported outcomes, compared with placebo, in patients who did not respond to or were intolerant to biologics or DMARDs,” reported Andrew Ostor, MD, of Monash University and Cabrini Hospital, both in Melbourne,. The safety profile was “consistent with that established for risankizumab in the treatment moderate to severe psoriasis,” he told attendees.
Risankizumab is approved in the United States for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. It is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits cytokine interleukin-23 by binding to its p19 subunit. IL-23 has been implicated in the development of PsA.
This was a phase 3 trial with “promising results in line with the ACR 20 response [at least 20% improvement in American College of Rheumatology response criteria] of other biologics in psoriatic arthritis,” according to Gaëlle Varkas, MD, PhD, of the Ghent (the Netherlands) University VIB Center for Inflammation Research and the department of rheumatology, Ghent University Hospital. “Especially in patients with severe and/or refractory skin disease or inadequate response at the level of the joint to other DMARDs or biologics, risankizumab is filling a void,” Dr. Varkas, who was not involved in the research, said in an interview.
There were no major safety problems, although long-term data, especially in regard to cancer and cardiovascular effects, “are always of interest, as they can be missed in randomized, controlled trials,” she said. In addition, “efficacy in concomitant axial disease, uveitis, and inflammatory bowel disease might favor one treatment over the other.” Another clinically significant takeaway was risankizumab’s “better effect on skin psoriasis while maintaining the effect on joint manifestations.”
Details of 24-week trial results
The phase 3, randomized, placebo-controlled, double-blind KEEPSAKE 2 trial involved 444 patients who had active PsA, defined as at least five swollen joints and at least five tender joints. All the patients either had an inadequate response to or were intolerant of one or two biologics or at least one conventional synthetic DMARD.
A total of 224 patients were randomly assigned to receive 150 mg of subcutaneous risankizumab at baseline and at 4 and 16 weeks after baseline; 220 participants received placebo injections. The primary endpoint was the proportion of patients who had at least 20% improvement in American College of Rheumatology response criteria at week 24.
Demographic and clinical characteristics were similar in both groups at baseline. Among the participants, the total mean number of swollen joints was 13.3, and the total mean number of tender joints was 22.6. The participants had PsA for an average of 8.2 years. The proportions of patients previously treated with biologics and DMARDs were similar in both groups, as were the proportions of patients currently taking glucocorticoids, NSAIDs, or methotrexate or another DMARD. At week 24, there remained 199 patients in the placebo group and 215 in the risankizumab group.
Just over half (51.3%) of patients who took risankizumab achieved at least 20% improvement in their ACR 20 score, compared with just over a quarter (26.5%) of those who received placebo (P < .001). All secondary endpoints also showed statistically significant improvements (P < .001 for all except P < .009 for the Fatigue Functional Assessment of Chronic Illness Therapy–Fatigue [FACIT-Fatigue] secondary endpoint).
Scores on the Health Assessment Questionnaire–Disability Index were –0.22 in the risankizumab group and –0.05 in the placebo group (P < .001). In the risankizumab group, 55% of patients achieved at least a 90% reduction in scores on the Psoriasis Area Severity Index, compared with 10.2% of patients who received placebo. Similarly, 25.6% of patients who took risankizumab and 11.4% of patients who received placebo had minimal disease activity 24 weeks after baseline.
In the 36-item Short Form Health Survey Physical Component Summary, the score change among risankizumab patients was 5.9, compared with 2 among the patients who received placebo. The change in FACIT-Fatigue score was 4.9 for patients who took risankizumab and 2.6 for patients who received placebo.
The researchers also assessed how many patients achieved higher levels of response to treatment. At least a 50% improvement in ACR response criteria occurred among 26.3% of patients taking risankizumab and 9.3% of patients taking placebo (P < .001). ACR 70 responses were seen in 12% of patients receiving risankizumab, compared with 5.9% of patients receiving placebo (P < .02). In the risankizumab group, 72.5% of patients had resolution of dactylitis and 42.9% had resolution of enthesitis, compared with 42.1% and 30.4%, respectively, in the placebo group.
Serious adverse events occurred in 4% of patients who received risankizumab and 5.5% of patients who received placebo. Serious infections occurred in 0.9% of those receiving risankizumab and 2.3% of those receiving placebo. Rates of treatment-emergent adverse events were also similar in the risankizumab (55.4%) and placebo (54.8%) groups.
In response to a question about whether it was possible to identify patients who might respond better to IL-23 inhibitors, compared with IL-17 inhibitors, Dr. Ostor acknowledged that rheumatologic practice is not yet proficient at using biomarkers to direct therapy, so the benefit from these drugs lay elsewhere.
“What I think is great is the luxury of choice these days,” Dr. Ostor told attendees. “We have these agents now, including risankizumab, that do work very effectively across the spectrum of the clinical features. It’s just lovely to have these agents available that can truly make a difference to the clinical picture of the individual.”
The trial was sponsored by AbbVie. Dr. Ostor has received research grants or speaking or consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Janssen, Lilly, Merck, Novartis, Pfizer, Roche, Sanofi, and UCB. Dr. Varkas has received research grants or speaker fees from AbbVie and Pfizer.
A version of this article first appeared on Medscape.com.
Risankizumab (Skyrizi) was effective for treating psoriatic arthritis (PsA) in patients who did not respond to or who could not tolerate other biologics or standard disease-modifying antirheumatic drugs (DMARDs), according to a study presented at the annual European Congress of Rheumatology. It was also well tolerated.
“Treatment with risankizumab resulted in significantly greater improvements in signs and symptoms of psoriatic arthritis, including assessments of disease activity in joints and skin and patient-reported outcomes, compared with placebo, in patients who did not respond to or were intolerant to biologics or DMARDs,” reported Andrew Ostor, MD, of Monash University and Cabrini Hospital, both in Melbourne,. The safety profile was “consistent with that established for risankizumab in the treatment moderate to severe psoriasis,” he told attendees.
Risankizumab is approved in the United States for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. It is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits cytokine interleukin-23 by binding to its p19 subunit. IL-23 has been implicated in the development of PsA.
This was a phase 3 trial with “promising results in line with the ACR 20 response [at least 20% improvement in American College of Rheumatology response criteria] of other biologics in psoriatic arthritis,” according to Gaëlle Varkas, MD, PhD, of the Ghent (the Netherlands) University VIB Center for Inflammation Research and the department of rheumatology, Ghent University Hospital. “Especially in patients with severe and/or refractory skin disease or inadequate response at the level of the joint to other DMARDs or biologics, risankizumab is filling a void,” Dr. Varkas, who was not involved in the research, said in an interview.
There were no major safety problems, although long-term data, especially in regard to cancer and cardiovascular effects, “are always of interest, as they can be missed in randomized, controlled trials,” she said. In addition, “efficacy in concomitant axial disease, uveitis, and inflammatory bowel disease might favor one treatment over the other.” Another clinically significant takeaway was risankizumab’s “better effect on skin psoriasis while maintaining the effect on joint manifestations.”
Details of 24-week trial results
The phase 3, randomized, placebo-controlled, double-blind KEEPSAKE 2 trial involved 444 patients who had active PsA, defined as at least five swollen joints and at least five tender joints. All the patients either had an inadequate response to or were intolerant of one or two biologics or at least one conventional synthetic DMARD.
A total of 224 patients were randomly assigned to receive 150 mg of subcutaneous risankizumab at baseline and at 4 and 16 weeks after baseline; 220 participants received placebo injections. The primary endpoint was the proportion of patients who had at least 20% improvement in American College of Rheumatology response criteria at week 24.
Demographic and clinical characteristics were similar in both groups at baseline. Among the participants, the total mean number of swollen joints was 13.3, and the total mean number of tender joints was 22.6. The participants had PsA for an average of 8.2 years. The proportions of patients previously treated with biologics and DMARDs were similar in both groups, as were the proportions of patients currently taking glucocorticoids, NSAIDs, or methotrexate or another DMARD. At week 24, there remained 199 patients in the placebo group and 215 in the risankizumab group.
Just over half (51.3%) of patients who took risankizumab achieved at least 20% improvement in their ACR 20 score, compared with just over a quarter (26.5%) of those who received placebo (P < .001). All secondary endpoints also showed statistically significant improvements (P < .001 for all except P < .009 for the Fatigue Functional Assessment of Chronic Illness Therapy–Fatigue [FACIT-Fatigue] secondary endpoint).
Scores on the Health Assessment Questionnaire–Disability Index were –0.22 in the risankizumab group and –0.05 in the placebo group (P < .001). In the risankizumab group, 55% of patients achieved at least a 90% reduction in scores on the Psoriasis Area Severity Index, compared with 10.2% of patients who received placebo. Similarly, 25.6% of patients who took risankizumab and 11.4% of patients who received placebo had minimal disease activity 24 weeks after baseline.
In the 36-item Short Form Health Survey Physical Component Summary, the score change among risankizumab patients was 5.9, compared with 2 among the patients who received placebo. The change in FACIT-Fatigue score was 4.9 for patients who took risankizumab and 2.6 for patients who received placebo.
The researchers also assessed how many patients achieved higher levels of response to treatment. At least a 50% improvement in ACR response criteria occurred among 26.3% of patients taking risankizumab and 9.3% of patients taking placebo (P < .001). ACR 70 responses were seen in 12% of patients receiving risankizumab, compared with 5.9% of patients receiving placebo (P < .02). In the risankizumab group, 72.5% of patients had resolution of dactylitis and 42.9% had resolution of enthesitis, compared with 42.1% and 30.4%, respectively, in the placebo group.
Serious adverse events occurred in 4% of patients who received risankizumab and 5.5% of patients who received placebo. Serious infections occurred in 0.9% of those receiving risankizumab and 2.3% of those receiving placebo. Rates of treatment-emergent adverse events were also similar in the risankizumab (55.4%) and placebo (54.8%) groups.
In response to a question about whether it was possible to identify patients who might respond better to IL-23 inhibitors, compared with IL-17 inhibitors, Dr. Ostor acknowledged that rheumatologic practice is not yet proficient at using biomarkers to direct therapy, so the benefit from these drugs lay elsewhere.
“What I think is great is the luxury of choice these days,” Dr. Ostor told attendees. “We have these agents now, including risankizumab, that do work very effectively across the spectrum of the clinical features. It’s just lovely to have these agents available that can truly make a difference to the clinical picture of the individual.”
The trial was sponsored by AbbVie. Dr. Ostor has received research grants or speaking or consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Janssen, Lilly, Merck, Novartis, Pfizer, Roche, Sanofi, and UCB. Dr. Varkas has received research grants or speaker fees from AbbVie and Pfizer.
A version of this article first appeared on Medscape.com.
Risankizumab (Skyrizi) was effective for treating psoriatic arthritis (PsA) in patients who did not respond to or who could not tolerate other biologics or standard disease-modifying antirheumatic drugs (DMARDs), according to a study presented at the annual European Congress of Rheumatology. It was also well tolerated.
“Treatment with risankizumab resulted in significantly greater improvements in signs and symptoms of psoriatic arthritis, including assessments of disease activity in joints and skin and patient-reported outcomes, compared with placebo, in patients who did not respond to or were intolerant to biologics or DMARDs,” reported Andrew Ostor, MD, of Monash University and Cabrini Hospital, both in Melbourne,. The safety profile was “consistent with that established for risankizumab in the treatment moderate to severe psoriasis,” he told attendees.
Risankizumab is approved in the United States for the treatment of moderate to severe plaque psoriasis in adults who are candidates for systemic therapy or phototherapy. It is a humanized immunoglobulin G1 monoclonal antibody that selectively inhibits cytokine interleukin-23 by binding to its p19 subunit. IL-23 has been implicated in the development of PsA.
This was a phase 3 trial with “promising results in line with the ACR 20 response [at least 20% improvement in American College of Rheumatology response criteria] of other biologics in psoriatic arthritis,” according to Gaëlle Varkas, MD, PhD, of the Ghent (the Netherlands) University VIB Center for Inflammation Research and the department of rheumatology, Ghent University Hospital. “Especially in patients with severe and/or refractory skin disease or inadequate response at the level of the joint to other DMARDs or biologics, risankizumab is filling a void,” Dr. Varkas, who was not involved in the research, said in an interview.
There were no major safety problems, although long-term data, especially in regard to cancer and cardiovascular effects, “are always of interest, as they can be missed in randomized, controlled trials,” she said. In addition, “efficacy in concomitant axial disease, uveitis, and inflammatory bowel disease might favor one treatment over the other.” Another clinically significant takeaway was risankizumab’s “better effect on skin psoriasis while maintaining the effect on joint manifestations.”
Details of 24-week trial results
The phase 3, randomized, placebo-controlled, double-blind KEEPSAKE 2 trial involved 444 patients who had active PsA, defined as at least five swollen joints and at least five tender joints. All the patients either had an inadequate response to or were intolerant of one or two biologics or at least one conventional synthetic DMARD.
A total of 224 patients were randomly assigned to receive 150 mg of subcutaneous risankizumab at baseline and at 4 and 16 weeks after baseline; 220 participants received placebo injections. The primary endpoint was the proportion of patients who had at least 20% improvement in American College of Rheumatology response criteria at week 24.
Demographic and clinical characteristics were similar in both groups at baseline. Among the participants, the total mean number of swollen joints was 13.3, and the total mean number of tender joints was 22.6. The participants had PsA for an average of 8.2 years. The proportions of patients previously treated with biologics and DMARDs were similar in both groups, as were the proportions of patients currently taking glucocorticoids, NSAIDs, or methotrexate or another DMARD. At week 24, there remained 199 patients in the placebo group and 215 in the risankizumab group.
Just over half (51.3%) of patients who took risankizumab achieved at least 20% improvement in their ACR 20 score, compared with just over a quarter (26.5%) of those who received placebo (P < .001). All secondary endpoints also showed statistically significant improvements (P < .001 for all except P < .009 for the Fatigue Functional Assessment of Chronic Illness Therapy–Fatigue [FACIT-Fatigue] secondary endpoint).
Scores on the Health Assessment Questionnaire–Disability Index were –0.22 in the risankizumab group and –0.05 in the placebo group (P < .001). In the risankizumab group, 55% of patients achieved at least a 90% reduction in scores on the Psoriasis Area Severity Index, compared with 10.2% of patients who received placebo. Similarly, 25.6% of patients who took risankizumab and 11.4% of patients who received placebo had minimal disease activity 24 weeks after baseline.
In the 36-item Short Form Health Survey Physical Component Summary, the score change among risankizumab patients was 5.9, compared with 2 among the patients who received placebo. The change in FACIT-Fatigue score was 4.9 for patients who took risankizumab and 2.6 for patients who received placebo.
The researchers also assessed how many patients achieved higher levels of response to treatment. At least a 50% improvement in ACR response criteria occurred among 26.3% of patients taking risankizumab and 9.3% of patients taking placebo (P < .001). ACR 70 responses were seen in 12% of patients receiving risankizumab, compared with 5.9% of patients receiving placebo (P < .02). In the risankizumab group, 72.5% of patients had resolution of dactylitis and 42.9% had resolution of enthesitis, compared with 42.1% and 30.4%, respectively, in the placebo group.
Serious adverse events occurred in 4% of patients who received risankizumab and 5.5% of patients who received placebo. Serious infections occurred in 0.9% of those receiving risankizumab and 2.3% of those receiving placebo. Rates of treatment-emergent adverse events were also similar in the risankizumab (55.4%) and placebo (54.8%) groups.
In response to a question about whether it was possible to identify patients who might respond better to IL-23 inhibitors, compared with IL-17 inhibitors, Dr. Ostor acknowledged that rheumatologic practice is not yet proficient at using biomarkers to direct therapy, so the benefit from these drugs lay elsewhere.
“What I think is great is the luxury of choice these days,” Dr. Ostor told attendees. “We have these agents now, including risankizumab, that do work very effectively across the spectrum of the clinical features. It’s just lovely to have these agents available that can truly make a difference to the clinical picture of the individual.”
The trial was sponsored by AbbVie. Dr. Ostor has received research grants or speaking or consulting fees from AbbVie, Bristol-Myers Squibb, Celgene, Janssen, Lilly, Merck, Novartis, Pfizer, Roche, Sanofi, and UCB. Dr. Varkas has received research grants or speaker fees from AbbVie and Pfizer.
A version of this article first appeared on Medscape.com.
Simple risk assessment predicts post-PCI ischemic events
A patient’s risk for ischemic events, but not bleeding, after percutaneous coronary intervention (PCI) can be predicted simply based on whether they have one or more guideline-based standardized risk criteria, a large-scale real-world analysis suggests.
Haoyu Wang, MD, and colleagues showed that having at least one high-risk feature, as outlined in the 2018 European Society of Cardiology and European Association for Cardiothoracic Surgery (ESC/EACTS) Guidelines on Myocardial Revascularization, was associated with an increased risk for target vessel failure by 48% and for a patient-oriented composite outcome by 44%.
Moreover, they showed that implantation of at least three stents and the presence of diabetes and diffuse multivessel disease were the only high-risk features from the guidelines that were independent predictors of the two outcomes.
The study of more than 10,000 PCI patients also showed that determining whether patients were at high bleeding risk (HBR) did not modify their ischemic risk.
This, said Dr. Wang, from the National Center for Cardiovascular Diseases, Fuwai Hospital, Beijing, underscores the importance of applying the high ischemic risk (HIR) criteria from the ESC/EACTS guidelines when tailoring dual antiplatelet therapy (DAPT).
The research was presented at the European Atherosclerosis Society 2021 Virtual Congress on June 2, and published online in the Journal of Atherosclerosis and Thrombosis.
Dr. Wang told theheart.org | Medscape Cardiology that they conducted the study to determine which – HIR or HBR – is “most important to balance when treating patients undergoing PCI and then having dual antiplatelet therapy.”
The results showed that when patients have both a HIR and HBR, it is the ESC/EACTS guideline HIR criteria that have “a higher impact” than the bleeding risk, and that this can be “used to guide our choice of the duration of dual anti-platelet therapy.”
“Maybe we can extend, or use more potent, P2Y12 inhibitors” in those situations, he said.
S. Lale Tokgözoglu, MD, PhD, professor of cardiology, Hacettepe University, Ankara, Turkey, who was not involved in the study, said the HIR assessment “performed well,” adding that the HBR score might have been expected to attenuate its “prognostic advantage.”
She told this news organization that the results “are interesting since previous observations have suggested that Asian patients may be more prone to medication side effects and bleeding.”
These findings emphasize the importance of assessing HIR in daily PCI practice and confirm that it “performs well in different populations in real life,” added Dr. Tokgözoglu, a former president of the EAS.
The ESC/EACTS guidelines aimed to standardize the definition of HIR, Dr. Wang said during the presentation.
They set out 10 high-risk features for ischemic events for patients undergoing revascularization, which included patient medical history, comorbid conditions, and the characteristics of the PCI procedure.
Although the goals of the criteria are to inform decision-making and stimulate research, Dr. Wang said that their “prevalence and prognostic association with clinical outcomes are yet to be established in real-world PCI practice.”
Alongside, the Predicting Bleeding Complication in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score was developed to predict out-of-hospital bleeding in patients receiving DAPT after stent implantation.
Although a PRECISE-DAPT score of at least 25 constitutes a patient at high bleeding risk, Dr. Wang pointed out that such patients are typically also at risk for ischemic events after PCI, and it is “unclear” whether being at HBR modifies this risk.
To investigate further, they used the prospective, real-world Fuwai PCI registry to collate an all-comer patient population with unselected use of drug-eluting stents at the National Center for Cardiovascular Diseases at Fuwai Hospital.
They excluded individuals who were treated with balloon angioplasty alone, bioresorbable scaffolds, or bare metal stents, leaving a total population of 10,167 patients who were treated in 2013.
In that cohort, 5,149 patients (50.6%) met at least one risk criterion from the ESC/EACTS guidelines (HIR patients) and 5,018 (49.4%) met none of the risk criteria (non-HIR patients).
The most common criteria were implantation of at least three stents (23.5%); total stent length greater than 60 mm (20.2%); diffuse multivessel disease, especially in diabetic patients (18.5%); and a history of ST-segment elevation myocardial infarction (13.9%).
HIR patients were significantly older than non-HIR patients (average age, 58.86 vs. 57.77 years; P < .001), were more likely to have diabetes mellitus (42.6% vs. 16.9%; P < .001); and were more likely to have already had a myocardial infarction (32.2% vs. 5.2%; P < .001).
HIR patients also had higher average PRECISE-ADAPT scores than those without HIR (11.22 vs. 9.94; P < .001), and were conversely less likely to have the left anterior descending artery as the target vessel than non-HIR patients (86.0% vs. 94.6%; P < .001).
Cox regression analysis taking into account a range of patient and clinical factors revealed that HIR patients were significantly more likely than their non-HIR counterparts to experience target vessel failure (hazard ratio, 1.48; 95% confidence interval, 1.25-1.74; P < .001).
They were also significantly more likely to have a patient-oriented composite outcome, defined as all-cause death, any myocardial infarction, or any revascularization (HR, 1.44; 95% CI, 1.28-1.63; P < .001).
There was also a significantly higher risk for cardiac death in HIR than in non-HIR patients (HR, 1.95; 95% CI, 1.16-3.29; P = .012).
However, there was no significant association between HIR status and clinically relevant bleeding (HR, 0.84; 95% CI, 0.66-1.06; P = .143).
When the researchers looked at individual ischemic risk features, they found that, on fully adjusted analyses, only two were independent predictors of target vessel failure and the patient-oriented composite outcome.
Having at least three stents implanted was significantly associated with target vessel failure (HR, 1.36; 95% CI, 1.02-1.80; P = .038), and borderline significantly associated with the patient oriented composite outcome (HR, 1.23; 95% CI, 1.00-1.53; P = .056).
Diffuse multivessel disease, especially in diabetic patients, was significantly associated with both target vessel failure (HR, 1.24; 95% CI, 1.02-1.51; P = .035) and with the patient-oriented composite outcome (HR, 1.20; 95% CI, 1.04-1.39; P = .012).
Neither risk feature was significantly associated with clinically relevant bleeding, Dr. Wang noted.
Stratifying the patients by HBR status, the team found that rates of target vessel failure, the patient-oriented composite outcome, cardiac death, myocardial infarction, and definite/probable stent thrombosis were higher in patients with both HIR and HBR than those with neither HIR nor HBR (P < .001).
Further stratifying patients by PRECISE-ADAPT scores – 10 or less indicating very low risk, 11-17 indicating low risk, 18-24 indicating moderate risk, and at least 25 indicating high risk – showed that HIR features had a consistent effect on ischemic and bleeding outcomes, regardless of bleeding risk.
No funding declared. No relevant financial relationships declared.
A version of this article first appeared on Medscape.com.
A patient’s risk for ischemic events, but not bleeding, after percutaneous coronary intervention (PCI) can be predicted simply based on whether they have one or more guideline-based standardized risk criteria, a large-scale real-world analysis suggests.
Haoyu Wang, MD, and colleagues showed that having at least one high-risk feature, as outlined in the 2018 European Society of Cardiology and European Association for Cardiothoracic Surgery (ESC/EACTS) Guidelines on Myocardial Revascularization, was associated with an increased risk for target vessel failure by 48% and for a patient-oriented composite outcome by 44%.
Moreover, they showed that implantation of at least three stents and the presence of diabetes and diffuse multivessel disease were the only high-risk features from the guidelines that were independent predictors of the two outcomes.
The study of more than 10,000 PCI patients also showed that determining whether patients were at high bleeding risk (HBR) did not modify their ischemic risk.
This, said Dr. Wang, from the National Center for Cardiovascular Diseases, Fuwai Hospital, Beijing, underscores the importance of applying the high ischemic risk (HIR) criteria from the ESC/EACTS guidelines when tailoring dual antiplatelet therapy (DAPT).
The research was presented at the European Atherosclerosis Society 2021 Virtual Congress on June 2, and published online in the Journal of Atherosclerosis and Thrombosis.
Dr. Wang told theheart.org | Medscape Cardiology that they conducted the study to determine which – HIR or HBR – is “most important to balance when treating patients undergoing PCI and then having dual antiplatelet therapy.”
The results showed that when patients have both a HIR and HBR, it is the ESC/EACTS guideline HIR criteria that have “a higher impact” than the bleeding risk, and that this can be “used to guide our choice of the duration of dual anti-platelet therapy.”
“Maybe we can extend, or use more potent, P2Y12 inhibitors” in those situations, he said.
S. Lale Tokgözoglu, MD, PhD, professor of cardiology, Hacettepe University, Ankara, Turkey, who was not involved in the study, said the HIR assessment “performed well,” adding that the HBR score might have been expected to attenuate its “prognostic advantage.”
She told this news organization that the results “are interesting since previous observations have suggested that Asian patients may be more prone to medication side effects and bleeding.”
These findings emphasize the importance of assessing HIR in daily PCI practice and confirm that it “performs well in different populations in real life,” added Dr. Tokgözoglu, a former president of the EAS.
The ESC/EACTS guidelines aimed to standardize the definition of HIR, Dr. Wang said during the presentation.
They set out 10 high-risk features for ischemic events for patients undergoing revascularization, which included patient medical history, comorbid conditions, and the characteristics of the PCI procedure.
Although the goals of the criteria are to inform decision-making and stimulate research, Dr. Wang said that their “prevalence and prognostic association with clinical outcomes are yet to be established in real-world PCI practice.”
Alongside, the Predicting Bleeding Complication in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score was developed to predict out-of-hospital bleeding in patients receiving DAPT after stent implantation.
Although a PRECISE-DAPT score of at least 25 constitutes a patient at high bleeding risk, Dr. Wang pointed out that such patients are typically also at risk for ischemic events after PCI, and it is “unclear” whether being at HBR modifies this risk.
To investigate further, they used the prospective, real-world Fuwai PCI registry to collate an all-comer patient population with unselected use of drug-eluting stents at the National Center for Cardiovascular Diseases at Fuwai Hospital.
They excluded individuals who were treated with balloon angioplasty alone, bioresorbable scaffolds, or bare metal stents, leaving a total population of 10,167 patients who were treated in 2013.
In that cohort, 5,149 patients (50.6%) met at least one risk criterion from the ESC/EACTS guidelines (HIR patients) and 5,018 (49.4%) met none of the risk criteria (non-HIR patients).
The most common criteria were implantation of at least three stents (23.5%); total stent length greater than 60 mm (20.2%); diffuse multivessel disease, especially in diabetic patients (18.5%); and a history of ST-segment elevation myocardial infarction (13.9%).
HIR patients were significantly older than non-HIR patients (average age, 58.86 vs. 57.77 years; P < .001), were more likely to have diabetes mellitus (42.6% vs. 16.9%; P < .001); and were more likely to have already had a myocardial infarction (32.2% vs. 5.2%; P < .001).
HIR patients also had higher average PRECISE-ADAPT scores than those without HIR (11.22 vs. 9.94; P < .001), and were conversely less likely to have the left anterior descending artery as the target vessel than non-HIR patients (86.0% vs. 94.6%; P < .001).
Cox regression analysis taking into account a range of patient and clinical factors revealed that HIR patients were significantly more likely than their non-HIR counterparts to experience target vessel failure (hazard ratio, 1.48; 95% confidence interval, 1.25-1.74; P < .001).
They were also significantly more likely to have a patient-oriented composite outcome, defined as all-cause death, any myocardial infarction, or any revascularization (HR, 1.44; 95% CI, 1.28-1.63; P < .001).
There was also a significantly higher risk for cardiac death in HIR than in non-HIR patients (HR, 1.95; 95% CI, 1.16-3.29; P = .012).
However, there was no significant association between HIR status and clinically relevant bleeding (HR, 0.84; 95% CI, 0.66-1.06; P = .143).
When the researchers looked at individual ischemic risk features, they found that, on fully adjusted analyses, only two were independent predictors of target vessel failure and the patient-oriented composite outcome.
Having at least three stents implanted was significantly associated with target vessel failure (HR, 1.36; 95% CI, 1.02-1.80; P = .038), and borderline significantly associated with the patient oriented composite outcome (HR, 1.23; 95% CI, 1.00-1.53; P = .056).
Diffuse multivessel disease, especially in diabetic patients, was significantly associated with both target vessel failure (HR, 1.24; 95% CI, 1.02-1.51; P = .035) and with the patient-oriented composite outcome (HR, 1.20; 95% CI, 1.04-1.39; P = .012).
Neither risk feature was significantly associated with clinically relevant bleeding, Dr. Wang noted.
Stratifying the patients by HBR status, the team found that rates of target vessel failure, the patient-oriented composite outcome, cardiac death, myocardial infarction, and definite/probable stent thrombosis were higher in patients with both HIR and HBR than those with neither HIR nor HBR (P < .001).
Further stratifying patients by PRECISE-ADAPT scores – 10 or less indicating very low risk, 11-17 indicating low risk, 18-24 indicating moderate risk, and at least 25 indicating high risk – showed that HIR features had a consistent effect on ischemic and bleeding outcomes, regardless of bleeding risk.
No funding declared. No relevant financial relationships declared.
A version of this article first appeared on Medscape.com.
A patient’s risk for ischemic events, but not bleeding, after percutaneous coronary intervention (PCI) can be predicted simply based on whether they have one or more guideline-based standardized risk criteria, a large-scale real-world analysis suggests.
Haoyu Wang, MD, and colleagues showed that having at least one high-risk feature, as outlined in the 2018 European Society of Cardiology and European Association for Cardiothoracic Surgery (ESC/EACTS) Guidelines on Myocardial Revascularization, was associated with an increased risk for target vessel failure by 48% and for a patient-oriented composite outcome by 44%.
Moreover, they showed that implantation of at least three stents and the presence of diabetes and diffuse multivessel disease were the only high-risk features from the guidelines that were independent predictors of the two outcomes.
The study of more than 10,000 PCI patients also showed that determining whether patients were at high bleeding risk (HBR) did not modify their ischemic risk.
This, said Dr. Wang, from the National Center for Cardiovascular Diseases, Fuwai Hospital, Beijing, underscores the importance of applying the high ischemic risk (HIR) criteria from the ESC/EACTS guidelines when tailoring dual antiplatelet therapy (DAPT).
The research was presented at the European Atherosclerosis Society 2021 Virtual Congress on June 2, and published online in the Journal of Atherosclerosis and Thrombosis.
Dr. Wang told theheart.org | Medscape Cardiology that they conducted the study to determine which – HIR or HBR – is “most important to balance when treating patients undergoing PCI and then having dual antiplatelet therapy.”
The results showed that when patients have both a HIR and HBR, it is the ESC/EACTS guideline HIR criteria that have “a higher impact” than the bleeding risk, and that this can be “used to guide our choice of the duration of dual anti-platelet therapy.”
“Maybe we can extend, or use more potent, P2Y12 inhibitors” in those situations, he said.
S. Lale Tokgözoglu, MD, PhD, professor of cardiology, Hacettepe University, Ankara, Turkey, who was not involved in the study, said the HIR assessment “performed well,” adding that the HBR score might have been expected to attenuate its “prognostic advantage.”
She told this news organization that the results “are interesting since previous observations have suggested that Asian patients may be more prone to medication side effects and bleeding.”
These findings emphasize the importance of assessing HIR in daily PCI practice and confirm that it “performs well in different populations in real life,” added Dr. Tokgözoglu, a former president of the EAS.
The ESC/EACTS guidelines aimed to standardize the definition of HIR, Dr. Wang said during the presentation.
They set out 10 high-risk features for ischemic events for patients undergoing revascularization, which included patient medical history, comorbid conditions, and the characteristics of the PCI procedure.
Although the goals of the criteria are to inform decision-making and stimulate research, Dr. Wang said that their “prevalence and prognostic association with clinical outcomes are yet to be established in real-world PCI practice.”
Alongside, the Predicting Bleeding Complication in Patients Undergoing Stent Implantation and Subsequent Dual Antiplatelet Therapy (PRECISE-DAPT) score was developed to predict out-of-hospital bleeding in patients receiving DAPT after stent implantation.
Although a PRECISE-DAPT score of at least 25 constitutes a patient at high bleeding risk, Dr. Wang pointed out that such patients are typically also at risk for ischemic events after PCI, and it is “unclear” whether being at HBR modifies this risk.
To investigate further, they used the prospective, real-world Fuwai PCI registry to collate an all-comer patient population with unselected use of drug-eluting stents at the National Center for Cardiovascular Diseases at Fuwai Hospital.
They excluded individuals who were treated with balloon angioplasty alone, bioresorbable scaffolds, or bare metal stents, leaving a total population of 10,167 patients who were treated in 2013.
In that cohort, 5,149 patients (50.6%) met at least one risk criterion from the ESC/EACTS guidelines (HIR patients) and 5,018 (49.4%) met none of the risk criteria (non-HIR patients).
The most common criteria were implantation of at least three stents (23.5%); total stent length greater than 60 mm (20.2%); diffuse multivessel disease, especially in diabetic patients (18.5%); and a history of ST-segment elevation myocardial infarction (13.9%).
HIR patients were significantly older than non-HIR patients (average age, 58.86 vs. 57.77 years; P < .001), were more likely to have diabetes mellitus (42.6% vs. 16.9%; P < .001); and were more likely to have already had a myocardial infarction (32.2% vs. 5.2%; P < .001).
HIR patients also had higher average PRECISE-ADAPT scores than those without HIR (11.22 vs. 9.94; P < .001), and were conversely less likely to have the left anterior descending artery as the target vessel than non-HIR patients (86.0% vs. 94.6%; P < .001).
Cox regression analysis taking into account a range of patient and clinical factors revealed that HIR patients were significantly more likely than their non-HIR counterparts to experience target vessel failure (hazard ratio, 1.48; 95% confidence interval, 1.25-1.74; P < .001).
They were also significantly more likely to have a patient-oriented composite outcome, defined as all-cause death, any myocardial infarction, or any revascularization (HR, 1.44; 95% CI, 1.28-1.63; P < .001).
There was also a significantly higher risk for cardiac death in HIR than in non-HIR patients (HR, 1.95; 95% CI, 1.16-3.29; P = .012).
However, there was no significant association between HIR status and clinically relevant bleeding (HR, 0.84; 95% CI, 0.66-1.06; P = .143).
When the researchers looked at individual ischemic risk features, they found that, on fully adjusted analyses, only two were independent predictors of target vessel failure and the patient-oriented composite outcome.
Having at least three stents implanted was significantly associated with target vessel failure (HR, 1.36; 95% CI, 1.02-1.80; P = .038), and borderline significantly associated with the patient oriented composite outcome (HR, 1.23; 95% CI, 1.00-1.53; P = .056).
Diffuse multivessel disease, especially in diabetic patients, was significantly associated with both target vessel failure (HR, 1.24; 95% CI, 1.02-1.51; P = .035) and with the patient-oriented composite outcome (HR, 1.20; 95% CI, 1.04-1.39; P = .012).
Neither risk feature was significantly associated with clinically relevant bleeding, Dr. Wang noted.
Stratifying the patients by HBR status, the team found that rates of target vessel failure, the patient-oriented composite outcome, cardiac death, myocardial infarction, and definite/probable stent thrombosis were higher in patients with both HIR and HBR than those with neither HIR nor HBR (P < .001).
Further stratifying patients by PRECISE-ADAPT scores – 10 or less indicating very low risk, 11-17 indicating low risk, 18-24 indicating moderate risk, and at least 25 indicating high risk – showed that HIR features had a consistent effect on ischemic and bleeding outcomes, regardless of bleeding risk.
No funding declared. No relevant financial relationships declared.
A version of this article first appeared on Medscape.com.
2021 Update on menopause
Among the studies we review in this Update are a follow-up of the US Women’s Health Initiative clinical trials and a large observational study from the United Kingdom, which exlore the impact of different hormone therapies (HTs) on breast cancer risk. We look at the interesting patterns found by authors of a study in Canada that analyzed predictors of unnecessary bilateral salpingo-oophorectomy. In addition, we review a study that investigates whether hormone therapy can be effective, alone or adjunctively, in peri- and postmenopausal women with depression. Finally, Dr. Chrisandra Shufelt and Dr. JoAnn Manson summarize highlights from the recent American Heart Association’s scientific statement on the menopause transition and increasing risk factors for cardiovascular disease, and how this period can be viewed as an opportunity to encourage healthy, cardiovascular risk–reducing behaviors.
Studies clarify menopausal HT’s impact on breast cancer risk
Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
For many menopausal women, the most worrisome concern related to the use of HT is that it might increase breast cancer risk. In the summer and fall of 2020, 2 important articles were published that addressed how the use of menopausal HT impacts the risk of breast cancer.
The Women’s Health Initiative (WHI) represents the largest and longest-term randomized trial assessing the health impacts of systemic HT. A 2013 WHI report found that with a median of 13 years’ cumulative follow-up, estrogen-only HT (ET) reduced the risk for breast cancer while estrogen-progestin therapy (EPT) increased the risk.1 In a July 2020 issue of JAMA, WHI investigators analyzed longer-term data (cumulative median follow-up >20 years), which allowed assessment of whether these trends (breast cancer incidence) persisted and if they led to changes in mortality from breast cancer.2
WHI data on breast cancer risk trends in ET vs EPT users
In the ET trial, in which Chlebowski and colleagues studied 10,739 women with prior hysterectomy, 238 versus 296 new cases of breast cancer were diagnosed in women in the ET versus placebo groups, respectively (annualized incidence, 0.30% [ET] vs 0.37% [placebo]; hazard ratio [HR], 0.78; P = .005). ET also was associated with significantly lower mortality from breast cancer: 30 versus 46 deaths (annualized mortality, 0.031% [ET] vs 0.046% [placebo]; HR, 0.60; P = 0.04).
In the EPT trial, which included 16,608 participants with an intact uterus, EPT compared with placebo was associated with significantly elevated risk for incident breast cancer: 584 versus 447 new cases, respectively (annualized incidence, 0.45% [EPT] vs 0.36% [placebo]; HR, 1.28; P<.001). However, mortality from breast cancer was similar in the EPT and placebo groups: 71 and 53 deaths (annualized mortality, 0.045% [EPT] and 0.035% [placebo]; HR, 1.35; P = .11).2
For women with previous hysterectomy who are considering initiating or continuing ET for treatment of bothersome menopausal symptoms, the breast cancer mortality benefit documented in this long-term WHI analysis could, as editorialists point out, “tip the scales” in favor of ET.3 Furthermore, the mortality benefit raises the possibility that ET could be evaluated as a risk-reduction strategy for selected high-risk menopausal women who have undergone hysterectomy. Although tamoxifen and aromatase inhibitors are approved for breast cancer chemoprophylaxis in high-risk menopausal women, these agents have not been found to lower breast cancer mortality.2
UK data analysis and risk for breast cancer in HT users
In an October 2020 issue of BMJ, Vinogradova and colleagues described their analysis of 2 primary care databases in the United Kingdom that in aggregate included roughly 99,000 women with breast cancer diagnosed between 1998 and 2018 (age range, 50–79; mean age at diagnosis, 63; >95% White); these were matched with more than 450,000 women without breast cancer (controls).4 Analyses were adjusted for smoking, body mass index (BMI), ethnicity, and mammography.
In this study, ever-use of EPT was associated with an adjusted odds ratio (OR) for breast cancer of 1.26 (95% confidence interval [CI], 1.24–1.29), while ET had an OR of 1.06 (95% CI, 1.03–1.10). In women aged 50 to 59 who used EPT for 5 years or more, 15 additional breast cancers were diagnosed per 10,000 woman-years; for ET users, the attributable risk was 3. Although risk rose with longer HT duration, this trend was less evident with ET than EPT.
In addition, the increased risk associated with ET use was less pronounced in women with a BMI greater than 30 kg/m2. Among EPT users, risks were similar with the progestins medroxyprogesterone acetate (MPA), norethindrone (NET), and levonorgestrel (LNG). Likewise, risks were similar regardless of estrogen dose and route of administration (that is, oral vs transdermal). Vaginal estrogen was not associated with a higher or lower risk for breast cancer. Among past users of ET or EPT (with MPA), no increased risk was noted 5 years or more after stopping HT. For users of EPT (with NET or LNG), risks diminished 5 years or more after stopping HT but remained modestly elevated compared with risk in never-users.4
In this large observational UK study, ET was associated with minimally elevated risk for breast cancer, while in the WHI study, ET reduced the risk for breast cancer. For EPT, the excess risk in both studies was identical. As the authors note, mean BMI in the UK study participants was slightly lower than that in the WHI participants, a distinction that might explain the differing findings with ET use.
In our practice, for women with an intact uterus who are considering the use of EPT for treatment of bothersome menopausal symptoms, we counsel that long-term use of HT slightly elevates the risk for breast cancer. By contrast, we advise posthysterectomy women with bothersome menopausal symptoms that ET does not appear to increase the risk for breast cancer.
Continue to: Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women...
Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women
Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2020;28:8-11. doi: 10.1097/GME.0000000000001652.
While prevention of ovarian cancer is an important benefit of bilateral salpingo-oophorectomy (BSO), performing a BSO at the time of hysterectomy in pre- or perimenopausal patients not only will induce surgical menopause but also is associated with significantly increased overall mortality and an increased risk of mortality due to cardiovascular disease in patients younger than age 45.5,6 Earlier BSO also has been associated with diabetes, accelerated bone density loss, sexual dysfunction, mood disorders, and decreased cognitive function.7
BSO at hysterectomy: How many procedures are not indicated?
To evaluate the prevalence and predictors of unnecessary BSO at the time of hysterectomy, Wong and colleagues conducted a multicenter retrospective review of hysterectomy procedures completed at 6 Canadian hospitals.8 Criteria for unnecessary BSO included age younger than 51 years; benign preoperative diagnosis (other than endometriosis, premenstrual dysphoric disorder, and gender dysphoria); and absence of endometriosis and pelvic adhesions.
A total of 2,656 hysterectomies were performed by 75 surgeons (28 fellowship trained and 47 generalists) across 3 community and 3 tertiary care hospitals between 2016 and 2018. At the time of hysterectomy, 749 patients (28%) underwent BSO. Of these, 509 women (68%) had at least 1 indication for concurrent BSO based on preoperative diagnosis.
Key study findings. Concurrent BSO procedures performed at academic hospitals were more likely to have a preoperative indication compared with BSO performed at community sites (70% vs 63%; OR, 1.42; 95% CI, 1.02–1.97; P = .04). BSO was more likely to be indicated when performed by fellowship-trained surgeons compared with surgeries performed by generalist surgeons (75% vs 63%; OR, 1.76; 95% CI, 1.26–2.44, P = .001). BSO procedures performed with vaginal hysterectomy were less likely to be indicated (3 of 20, 15%) when compared with open hysterectomy (74 of 154, 48%) and laparoscopic hysterectomy (432 of 575, 75%).
Of the patients who lacked a preoperative indication for concomitant BSO, 105 of 239 (43.9%) were younger than age 51. Overall, 8% (59 of 749) of patients in the study cohort had an unnecessary BSO based on a combination of preoperative diagnosis, age younger than age 51, and intraoperative factors including absence of endometriosis and adhesions.
The retrospective study by Wong and colleagues provides the first assessment of Canadian practice patterns with respect to concurrent BSO at the time of hysterectomy. The authors found that, overall, more than two-thirds of BSO procedures were indicated. However, the proportion of BSO that was indicated was higher in teaching hospitals and in surgeries performed by fellowship-trained gynecologists. These important observations underscore the role of clinician education in reducing nonindicated BSO in pre- and perimenopausal women undergoing hysterectomy for benign disease.
Continue to: HT for menopausal depression: Which patients may benefit?
HT for menopausal depression: Which patients may benefit?
Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686- 705. doi:10.1176/appi.ajp.2020.19080848.
The cumulative lifetime prevalence of major depression in US women is 21%.9 An increased risk of mood symptoms and major depressive disorder occurs with the cessation of ovarian hormone production during menopause. In a review of both physiology and clinical studies, an American Psychiatric Association task force found support for several hormone-related strategies for treating depression and highlighted the rapidly advancing, but mixed, findings in this field.10
Clinical trials that examined mood in peri- and postmenopausal women treated with HT have produced mixed results for a variety of reasons, including differences in psychiatric symptomatology across studies and differences in treatment timing in relation to menopause onset.
HT effectiveness for depression depends on menopausal status
Five studies included in a meta-analysis by Rubinow and colleagues examined the use of ET and EPT as antidepressant monotherapy in peri- or postmenopausal women with major depression.11 Of the 3 higher-quality studies, 2 conducted in perimenopausal women demonstrated the antidepressant efficacy of transdermal estrogen patches compared with placebo. The third study included a mixed population of both peri- and postmenopausal women, and it found that increased estradiol levels (spontaneously occurring or due to ET) were associated with improvement in depression in perimenopausal women but not in postmenopausal women.11
ET also has been investigated as a potential adjunctive treatment to selective serotonin reuptake inhibitors (SSRIs). In a retrospective analysis of a multicenter randomized controlled trial of fluoxetine in patients with depression, women who received ET and fluoxetine demonstrated a greater improvement than those who received fluoxetine monotherapy.12 One small study that prospectively assessed ET in combination with an antidepressant in postmenopausal women demonstrated no benefit of ET in treating depression.13 Another small trial found that while combining transdermal ET with an SSRI accelerated symptom improvement, by the end of the 10-week study, treatment efficacy in the HT plus SSRI group was no greater than that observed in the SSRI-only group.14
Nineteen studies included in the metaanalysis by Rubinow and colleagues, which examined mood after ET or EPT treatment in nondepressed women, found little evidence of benefit, particularly in women without other physical symptoms of menopause.11
The Kronos Early Estrogen Prevention Study (KEEPS) followed 661 women who received either oral estrogen plus progesterone, transdermal estrogen plus progesterone, or placebo over 4 years.15 Women with clinical depression were excluded from the study; however, women with mild to moderate mood symptoms who were being treated with an antidepressant were included. Improvements in depressive symptoms and anxiety were observed only in the oral estrogen plus progesterone group compared with the placebo group.15
In a study of 172 euthymic peri- and postmenopausal women treated for 12 months with transdermal estrogen plus oral progesterone, investigators found that, unlike postmenopausal women and those in the late perimenopausal transition, only women in the early perimenopausal transition had a lower risk of developing depressive symptoms.16
Bottom line
This complex literature suggests that ET/HT interventions are most likely to be successful when implemented early in the menopausal transition. The clearest indication for the use of HT is for perimenopausal women experiencing depression who are also experiencing menopausal symptoms (for example, bothersome hot flashes). There is little evidence that the use of ET/HT in late perimenopausal or postmenopausal women effectively treats depression; accordingly, HT is not recommended for the treatment of mood disorders in this population. The more ambiguous cases are those of perimenopausal women who are depressed but do not have classic vasomotor symptoms; some evidence supports the antidepressant efficacy of HT in this setting.11 Although some studies suggest that HT can be effective in preventing depression in perimenopausal women, more evidence is needed.16
A trial of ET/EPT is reasonable in perimenopausal women with depression and classic menopausal symptoms. Use of HT also can be considered either alone or in combination with an SSRI in perimenopausal women with depression who do not have significant classic menopausal symptoms. However, HT is not recommended as prophylaxis against depression in euthymic perimenopausal women. Finally, keep in mind that the use of HT to address mood issues constitutes off-label use.
The menopause transition: A key period for strategizing CVD risk factor reduction

Chrisandra L. Shufelt, MD, MS, NCMP
Dr. Shufelt is Associate Director of the Barbra
Streisand Women’s Heart Center, Smidt
Heart Institute, Cedars-Sinai Medical Center,
Los Angeles, California.

JoAnn E. Manson, MD, DrPH, NCMP
Dr. Manson is Professor of Medicine and the
Michael and Lee Bell Professor of Women’s
Health at Harvard Medical School; Professor
in the Department of Epidemiology, Harvard
T.H. Chan School of Public Health; and Chief
of the Division of Preventive Medicine
at Brigham and Women’s Hospital, Boston,
Massachusetts.
The authors report no financial relationships relevant to this article. Dr. Manson is a coauthor of the AHA Scientific Statement discussed in this article.
In the United States, nearly one-half of a woman’s life, on average, will be lived after menopause. For women with natural menopause, the menopause transition (MT) can begin 2 to 7 years before and may extend 1 year past the final menstrual period, which occurs at an average age of 51 years. For women with surgical menopause, the MT occurs abruptly with the sudden loss of endogenous ovarian hormones. Both types of transitions mark a critical time period when reproduction and endogenous sex hormone levels diminish and when cardiovascular disease (CVD) risk factors begin to rise.
The 2020 American Heart Association (AHA) scientific statement, “Menopause transition and cardiovascular disease risk: Implications for timing of early prevention,” highlights the MT as a window of opportunity for CVD prevention.1
CVD risk factors associated with ovarian aging
In the AHA scientific statement, data from several longitudinal women’s health studies were used to identify which CVD risk factor changes during the MT are related to ovarian aging as opposed to chronologic aging. Independent of aging, those associated with reproductive or ovarian aging included an increase in serum total cholesterol, low-density lipoprotein cholesterol (LDL-C), and apolipoprotein B. Changes in high-density lipoprotein cholesterol (HDL-C) particles and function also occur during the MT, which may explain why higher HDL-C levels during the MT and the postmenopausal years are not as cardioprotective as during the premenopausal period.
Changes in body composition and adipose tissue distribution also are associated with ovarian aging, with reduction in muscle mass and lean body mass and an increase in abdominal/visceral fat and subcutaneous adipose tissue. Although these body composition changes reflect ovarian aging, midlife weight gain is more closely related to chronologic aging.
The risk of the metabolic syndrome constellation of risk factors was found to be more closely associated with ovarian aging, whereas changes in blood pressure, insulin, and glucose individually tracked more closely with chronologic aging. Additionally, the AHA statement notes the research that identified several symptoms during the MT—including vasomotor symptoms, sleep disturbance, and depression—as being associated with more adverse CVD risk factor status and with subclinical measures of atherosclerosis. Additional research on the mechanistic basis for these associations is needed.
Chronologic age and type of menopause
Notably, a woman’s age and type of menopause matter with respect to CVD risk. Higher CVD risk is seen in women with premature onset (age < 40 years) or early onset (age < 45 years) of menopause and in women undergoing surgical menopause (bilateral oophorectomy) before age 45. In general, menopausal hormone therapy (HT) is recommended for women with premature or early menopause, whether natural or surgical, with continuation through at least the average age of natural menopause. In other women, although not recommended for the express purpose of CVD prevention, menopausal HT is appropriate for the treatment of bothersome vasomotor or other menopausal symptoms, especially when therapy is started before age 60 or within 10 years of menopause among women who are not at elevated risk of CVD.
While the AHA statement suggests that some women who begin estrogen early in menopause may experience reduced coronary heart disease risk, major research gaps remain with regard to HT dose, formulation, route of delivery, and recommended duration of treatment.
An opportunity to promote healthy lifestyle behaviors
Translating the AHA’s first-of-its-kind scientific statement into clinical practice requires recognition and awareness of the MT as a unique phase in a woman’s life associated with myriad changes in CVD risk factors. The statement underscores that the MT is an important time to target behavioral changes to promote CVD risk reduction, including lifestyle modifications in the AHA’s Life’s Simple 7 components (increased physical activity, smoking cessation, healthy diet, avoidance of weight gain) as well as vigilant control of blood pressure, cholesterol, and glucose levels. The MT is truly a window of opportunity for reinvigorated efforts to lower women’s CVD risk. ●
Reference
1. El Khoudary SR, Aggarwal B, Beckie TM, et al; American Heart Association Prevention Science Committee of the Council on Epidemiology and Prevention; and Council on Cardiovascular and Stroke Nursing. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation. 2020;142:e506-e532. doi: 10.1161/CIR.000000000000912.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310:1353- 1368. doi: 10.1001/jama.2013.278040.
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Minami CA, Freedman RA. Menopausal hormone therapy and long-term breast cancer risk: further data from the Women’s Health Initiative trials. JAMA. 2020;324:347-349. doi: 10.1001/jama.2020.9620.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
- Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279. doi: 10.1016/j.ajog.2017.07.037.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15-23. doi: 10.1097/gme.0b013e31818888f7.
- Karp NE, Fenner DE, Burgunder-Zdravkovski L, et al. Removal of normal ovaries in women under age 51 at the time of hysterectomy. Am J Obstetr Gynecol. 2015;213:716.e1-6. doi: 10.1016/j.ajog.2015.05.062.
- Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2021;28:8-11. doi: 10.1097/GME.0000000000001652.
- Kessler RC, McGonagle KA, Swartz M, et al. Sex and depression in the National Comorbidity Survey. I: lifetime prevalence, chronicity, and recurrence. J Affect Disord. 1993;29:85- 96. doi: 10.1016/0165-0327(93)00026-g.
- Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686-705. doi:10.1176/appi. ajp.2020.19080848.
- Rubinow DR, Johnson SL, Schmidt PJ, et al. Efficacy of estradiol in perimenopausal depression: so much promise and so few answers. Depress Anxiety. 2015;32:539-549. doi: 10.1002/ da.22391.
- Schneider LS, Small GW, Hamilton SH, et al. Estrogen replacement and response to fluoxetine in a multicenter geriatric depression trial. Fluoxetine Collaborative Study Group. Am J Geriatr Psychiatry. 1997;5:97-106.
- Dias RS, Kerr-Corrêa F, Moreno RA, et al. Efficacy of hormone therapy with and without methyltestosterone augmentation of venlafaxine in the treatment of postmenopausal depression: a double-blind controlled pilot study. Menopause. 2006;13:202-211. doi:10.1097/01.gme.0000198491.34371.9c.
- Rasgon NL, Dunkin J, Fairbanks L, et al. Estrogen and response to sertraline in postmenopausal women with major depressive disorder: a pilot study. J Psychiatr Res. 2007;41:338- 343. doi: 10.1016/j.jpsychires.2006.03.009.
- Gleason CE, Dowling NM, Wharton W, et al. Effects of hormone therapy on cognition and mood in recently postmenopausal women: findings from the randomized, controlled KEEPS–cognitive and affective study. PLoS Med. 2015;12:e1001833. doi: 10.1371/journal.pmed.1001833.
- Gordon JL, Rubinow DR, Eisenlohr-Moul TA, et al. Efficacy of transdermal estradiol and micronized progesterone in the prevention of depressive symptoms in the menopause transition: a randomized clinical trial. JAMA Psychiatry. 2018;75:149–157. doi:10.1001/jamapsychiatry.2017.3998.
Among the studies we review in this Update are a follow-up of the US Women’s Health Initiative clinical trials and a large observational study from the United Kingdom, which exlore the impact of different hormone therapies (HTs) on breast cancer risk. We look at the interesting patterns found by authors of a study in Canada that analyzed predictors of unnecessary bilateral salpingo-oophorectomy. In addition, we review a study that investigates whether hormone therapy can be effective, alone or adjunctively, in peri- and postmenopausal women with depression. Finally, Dr. Chrisandra Shufelt and Dr. JoAnn Manson summarize highlights from the recent American Heart Association’s scientific statement on the menopause transition and increasing risk factors for cardiovascular disease, and how this period can be viewed as an opportunity to encourage healthy, cardiovascular risk–reducing behaviors.
Studies clarify menopausal HT’s impact on breast cancer risk
Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
For many menopausal women, the most worrisome concern related to the use of HT is that it might increase breast cancer risk. In the summer and fall of 2020, 2 important articles were published that addressed how the use of menopausal HT impacts the risk of breast cancer.
The Women’s Health Initiative (WHI) represents the largest and longest-term randomized trial assessing the health impacts of systemic HT. A 2013 WHI report found that with a median of 13 years’ cumulative follow-up, estrogen-only HT (ET) reduced the risk for breast cancer while estrogen-progestin therapy (EPT) increased the risk.1 In a July 2020 issue of JAMA, WHI investigators analyzed longer-term data (cumulative median follow-up >20 years), which allowed assessment of whether these trends (breast cancer incidence) persisted and if they led to changes in mortality from breast cancer.2
WHI data on breast cancer risk trends in ET vs EPT users
In the ET trial, in which Chlebowski and colleagues studied 10,739 women with prior hysterectomy, 238 versus 296 new cases of breast cancer were diagnosed in women in the ET versus placebo groups, respectively (annualized incidence, 0.30% [ET] vs 0.37% [placebo]; hazard ratio [HR], 0.78; P = .005). ET also was associated with significantly lower mortality from breast cancer: 30 versus 46 deaths (annualized mortality, 0.031% [ET] vs 0.046% [placebo]; HR, 0.60; P = 0.04).
In the EPT trial, which included 16,608 participants with an intact uterus, EPT compared with placebo was associated with significantly elevated risk for incident breast cancer: 584 versus 447 new cases, respectively (annualized incidence, 0.45% [EPT] vs 0.36% [placebo]; HR, 1.28; P<.001). However, mortality from breast cancer was similar in the EPT and placebo groups: 71 and 53 deaths (annualized mortality, 0.045% [EPT] and 0.035% [placebo]; HR, 1.35; P = .11).2
For women with previous hysterectomy who are considering initiating or continuing ET for treatment of bothersome menopausal symptoms, the breast cancer mortality benefit documented in this long-term WHI analysis could, as editorialists point out, “tip the scales” in favor of ET.3 Furthermore, the mortality benefit raises the possibility that ET could be evaluated as a risk-reduction strategy for selected high-risk menopausal women who have undergone hysterectomy. Although tamoxifen and aromatase inhibitors are approved for breast cancer chemoprophylaxis in high-risk menopausal women, these agents have not been found to lower breast cancer mortality.2
UK data analysis and risk for breast cancer in HT users
In an October 2020 issue of BMJ, Vinogradova and colleagues described their analysis of 2 primary care databases in the United Kingdom that in aggregate included roughly 99,000 women with breast cancer diagnosed between 1998 and 2018 (age range, 50–79; mean age at diagnosis, 63; >95% White); these were matched with more than 450,000 women without breast cancer (controls).4 Analyses were adjusted for smoking, body mass index (BMI), ethnicity, and mammography.
In this study, ever-use of EPT was associated with an adjusted odds ratio (OR) for breast cancer of 1.26 (95% confidence interval [CI], 1.24–1.29), while ET had an OR of 1.06 (95% CI, 1.03–1.10). In women aged 50 to 59 who used EPT for 5 years or more, 15 additional breast cancers were diagnosed per 10,000 woman-years; for ET users, the attributable risk was 3. Although risk rose with longer HT duration, this trend was less evident with ET than EPT.
In addition, the increased risk associated with ET use was less pronounced in women with a BMI greater than 30 kg/m2. Among EPT users, risks were similar with the progestins medroxyprogesterone acetate (MPA), norethindrone (NET), and levonorgestrel (LNG). Likewise, risks were similar regardless of estrogen dose and route of administration (that is, oral vs transdermal). Vaginal estrogen was not associated with a higher or lower risk for breast cancer. Among past users of ET or EPT (with MPA), no increased risk was noted 5 years or more after stopping HT. For users of EPT (with NET or LNG), risks diminished 5 years or more after stopping HT but remained modestly elevated compared with risk in never-users.4
In this large observational UK study, ET was associated with minimally elevated risk for breast cancer, while in the WHI study, ET reduced the risk for breast cancer. For EPT, the excess risk in both studies was identical. As the authors note, mean BMI in the UK study participants was slightly lower than that in the WHI participants, a distinction that might explain the differing findings with ET use.
In our practice, for women with an intact uterus who are considering the use of EPT for treatment of bothersome menopausal symptoms, we counsel that long-term use of HT slightly elevates the risk for breast cancer. By contrast, we advise posthysterectomy women with bothersome menopausal symptoms that ET does not appear to increase the risk for breast cancer.
Continue to: Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women...
Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women
Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2020;28:8-11. doi: 10.1097/GME.0000000000001652.
While prevention of ovarian cancer is an important benefit of bilateral salpingo-oophorectomy (BSO), performing a BSO at the time of hysterectomy in pre- or perimenopausal patients not only will induce surgical menopause but also is associated with significantly increased overall mortality and an increased risk of mortality due to cardiovascular disease in patients younger than age 45.5,6 Earlier BSO also has been associated with diabetes, accelerated bone density loss, sexual dysfunction, mood disorders, and decreased cognitive function.7
BSO at hysterectomy: How many procedures are not indicated?
To evaluate the prevalence and predictors of unnecessary BSO at the time of hysterectomy, Wong and colleagues conducted a multicenter retrospective review of hysterectomy procedures completed at 6 Canadian hospitals.8 Criteria for unnecessary BSO included age younger than 51 years; benign preoperative diagnosis (other than endometriosis, premenstrual dysphoric disorder, and gender dysphoria); and absence of endometriosis and pelvic adhesions.
A total of 2,656 hysterectomies were performed by 75 surgeons (28 fellowship trained and 47 generalists) across 3 community and 3 tertiary care hospitals between 2016 and 2018. At the time of hysterectomy, 749 patients (28%) underwent BSO. Of these, 509 women (68%) had at least 1 indication for concurrent BSO based on preoperative diagnosis.
Key study findings. Concurrent BSO procedures performed at academic hospitals were more likely to have a preoperative indication compared with BSO performed at community sites (70% vs 63%; OR, 1.42; 95% CI, 1.02–1.97; P = .04). BSO was more likely to be indicated when performed by fellowship-trained surgeons compared with surgeries performed by generalist surgeons (75% vs 63%; OR, 1.76; 95% CI, 1.26–2.44, P = .001). BSO procedures performed with vaginal hysterectomy were less likely to be indicated (3 of 20, 15%) when compared with open hysterectomy (74 of 154, 48%) and laparoscopic hysterectomy (432 of 575, 75%).
Of the patients who lacked a preoperative indication for concomitant BSO, 105 of 239 (43.9%) were younger than age 51. Overall, 8% (59 of 749) of patients in the study cohort had an unnecessary BSO based on a combination of preoperative diagnosis, age younger than age 51, and intraoperative factors including absence of endometriosis and adhesions.
The retrospective study by Wong and colleagues provides the first assessment of Canadian practice patterns with respect to concurrent BSO at the time of hysterectomy. The authors found that, overall, more than two-thirds of BSO procedures were indicated. However, the proportion of BSO that was indicated was higher in teaching hospitals and in surgeries performed by fellowship-trained gynecologists. These important observations underscore the role of clinician education in reducing nonindicated BSO in pre- and perimenopausal women undergoing hysterectomy for benign disease.
Continue to: HT for menopausal depression: Which patients may benefit?
HT for menopausal depression: Which patients may benefit?
Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686- 705. doi:10.1176/appi.ajp.2020.19080848.
The cumulative lifetime prevalence of major depression in US women is 21%.9 An increased risk of mood symptoms and major depressive disorder occurs with the cessation of ovarian hormone production during menopause. In a review of both physiology and clinical studies, an American Psychiatric Association task force found support for several hormone-related strategies for treating depression and highlighted the rapidly advancing, but mixed, findings in this field.10
Clinical trials that examined mood in peri- and postmenopausal women treated with HT have produced mixed results for a variety of reasons, including differences in psychiatric symptomatology across studies and differences in treatment timing in relation to menopause onset.
HT effectiveness for depression depends on menopausal status
Five studies included in a meta-analysis by Rubinow and colleagues examined the use of ET and EPT as antidepressant monotherapy in peri- or postmenopausal women with major depression.11 Of the 3 higher-quality studies, 2 conducted in perimenopausal women demonstrated the antidepressant efficacy of transdermal estrogen patches compared with placebo. The third study included a mixed population of both peri- and postmenopausal women, and it found that increased estradiol levels (spontaneously occurring or due to ET) were associated with improvement in depression in perimenopausal women but not in postmenopausal women.11
ET also has been investigated as a potential adjunctive treatment to selective serotonin reuptake inhibitors (SSRIs). In a retrospective analysis of a multicenter randomized controlled trial of fluoxetine in patients with depression, women who received ET and fluoxetine demonstrated a greater improvement than those who received fluoxetine monotherapy.12 One small study that prospectively assessed ET in combination with an antidepressant in postmenopausal women demonstrated no benefit of ET in treating depression.13 Another small trial found that while combining transdermal ET with an SSRI accelerated symptom improvement, by the end of the 10-week study, treatment efficacy in the HT plus SSRI group was no greater than that observed in the SSRI-only group.14
Nineteen studies included in the metaanalysis by Rubinow and colleagues, which examined mood after ET or EPT treatment in nondepressed women, found little evidence of benefit, particularly in women without other physical symptoms of menopause.11
The Kronos Early Estrogen Prevention Study (KEEPS) followed 661 women who received either oral estrogen plus progesterone, transdermal estrogen plus progesterone, or placebo over 4 years.15 Women with clinical depression were excluded from the study; however, women with mild to moderate mood symptoms who were being treated with an antidepressant were included. Improvements in depressive symptoms and anxiety were observed only in the oral estrogen plus progesterone group compared with the placebo group.15
In a study of 172 euthymic peri- and postmenopausal women treated for 12 months with transdermal estrogen plus oral progesterone, investigators found that, unlike postmenopausal women and those in the late perimenopausal transition, only women in the early perimenopausal transition had a lower risk of developing depressive symptoms.16
Bottom line
This complex literature suggests that ET/HT interventions are most likely to be successful when implemented early in the menopausal transition. The clearest indication for the use of HT is for perimenopausal women experiencing depression who are also experiencing menopausal symptoms (for example, bothersome hot flashes). There is little evidence that the use of ET/HT in late perimenopausal or postmenopausal women effectively treats depression; accordingly, HT is not recommended for the treatment of mood disorders in this population. The more ambiguous cases are those of perimenopausal women who are depressed but do not have classic vasomotor symptoms; some evidence supports the antidepressant efficacy of HT in this setting.11 Although some studies suggest that HT can be effective in preventing depression in perimenopausal women, more evidence is needed.16
A trial of ET/EPT is reasonable in perimenopausal women with depression and classic menopausal symptoms. Use of HT also can be considered either alone or in combination with an SSRI in perimenopausal women with depression who do not have significant classic menopausal symptoms. However, HT is not recommended as prophylaxis against depression in euthymic perimenopausal women. Finally, keep in mind that the use of HT to address mood issues constitutes off-label use.
The menopause transition: A key period for strategizing CVD risk factor reduction

Chrisandra L. Shufelt, MD, MS, NCMP
Dr. Shufelt is Associate Director of the Barbra
Streisand Women’s Heart Center, Smidt
Heart Institute, Cedars-Sinai Medical Center,
Los Angeles, California.

JoAnn E. Manson, MD, DrPH, NCMP
Dr. Manson is Professor of Medicine and the
Michael and Lee Bell Professor of Women’s
Health at Harvard Medical School; Professor
in the Department of Epidemiology, Harvard
T.H. Chan School of Public Health; and Chief
of the Division of Preventive Medicine
at Brigham and Women’s Hospital, Boston,
Massachusetts.
The authors report no financial relationships relevant to this article. Dr. Manson is a coauthor of the AHA Scientific Statement discussed in this article.
In the United States, nearly one-half of a woman’s life, on average, will be lived after menopause. For women with natural menopause, the menopause transition (MT) can begin 2 to 7 years before and may extend 1 year past the final menstrual period, which occurs at an average age of 51 years. For women with surgical menopause, the MT occurs abruptly with the sudden loss of endogenous ovarian hormones. Both types of transitions mark a critical time period when reproduction and endogenous sex hormone levels diminish and when cardiovascular disease (CVD) risk factors begin to rise.
The 2020 American Heart Association (AHA) scientific statement, “Menopause transition and cardiovascular disease risk: Implications for timing of early prevention,” highlights the MT as a window of opportunity for CVD prevention.1
CVD risk factors associated with ovarian aging
In the AHA scientific statement, data from several longitudinal women’s health studies were used to identify which CVD risk factor changes during the MT are related to ovarian aging as opposed to chronologic aging. Independent of aging, those associated with reproductive or ovarian aging included an increase in serum total cholesterol, low-density lipoprotein cholesterol (LDL-C), and apolipoprotein B. Changes in high-density lipoprotein cholesterol (HDL-C) particles and function also occur during the MT, which may explain why higher HDL-C levels during the MT and the postmenopausal years are not as cardioprotective as during the premenopausal period.
Changes in body composition and adipose tissue distribution also are associated with ovarian aging, with reduction in muscle mass and lean body mass and an increase in abdominal/visceral fat and subcutaneous adipose tissue. Although these body composition changes reflect ovarian aging, midlife weight gain is more closely related to chronologic aging.
The risk of the metabolic syndrome constellation of risk factors was found to be more closely associated with ovarian aging, whereas changes in blood pressure, insulin, and glucose individually tracked more closely with chronologic aging. Additionally, the AHA statement notes the research that identified several symptoms during the MT—including vasomotor symptoms, sleep disturbance, and depression—as being associated with more adverse CVD risk factor status and with subclinical measures of atherosclerosis. Additional research on the mechanistic basis for these associations is needed.
Chronologic age and type of menopause
Notably, a woman’s age and type of menopause matter with respect to CVD risk. Higher CVD risk is seen in women with premature onset (age < 40 years) or early onset (age < 45 years) of menopause and in women undergoing surgical menopause (bilateral oophorectomy) before age 45. In general, menopausal hormone therapy (HT) is recommended for women with premature or early menopause, whether natural or surgical, with continuation through at least the average age of natural menopause. In other women, although not recommended for the express purpose of CVD prevention, menopausal HT is appropriate for the treatment of bothersome vasomotor or other menopausal symptoms, especially when therapy is started before age 60 or within 10 years of menopause among women who are not at elevated risk of CVD.
While the AHA statement suggests that some women who begin estrogen early in menopause may experience reduced coronary heart disease risk, major research gaps remain with regard to HT dose, formulation, route of delivery, and recommended duration of treatment.
An opportunity to promote healthy lifestyle behaviors
Translating the AHA’s first-of-its-kind scientific statement into clinical practice requires recognition and awareness of the MT as a unique phase in a woman’s life associated with myriad changes in CVD risk factors. The statement underscores that the MT is an important time to target behavioral changes to promote CVD risk reduction, including lifestyle modifications in the AHA’s Life’s Simple 7 components (increased physical activity, smoking cessation, healthy diet, avoidance of weight gain) as well as vigilant control of blood pressure, cholesterol, and glucose levels. The MT is truly a window of opportunity for reinvigorated efforts to lower women’s CVD risk. ●
Reference
1. El Khoudary SR, Aggarwal B, Beckie TM, et al; American Heart Association Prevention Science Committee of the Council on Epidemiology and Prevention; and Council on Cardiovascular and Stroke Nursing. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation. 2020;142:e506-e532. doi: 10.1161/CIR.000000000000912.
Among the studies we review in this Update are a follow-up of the US Women’s Health Initiative clinical trials and a large observational study from the United Kingdom, which exlore the impact of different hormone therapies (HTs) on breast cancer risk. We look at the interesting patterns found by authors of a study in Canada that analyzed predictors of unnecessary bilateral salpingo-oophorectomy. In addition, we review a study that investigates whether hormone therapy can be effective, alone or adjunctively, in peri- and postmenopausal women with depression. Finally, Dr. Chrisandra Shufelt and Dr. JoAnn Manson summarize highlights from the recent American Heart Association’s scientific statement on the menopause transition and increasing risk factors for cardiovascular disease, and how this period can be viewed as an opportunity to encourage healthy, cardiovascular risk–reducing behaviors.
Studies clarify menopausal HT’s impact on breast cancer risk
Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
For many menopausal women, the most worrisome concern related to the use of HT is that it might increase breast cancer risk. In the summer and fall of 2020, 2 important articles were published that addressed how the use of menopausal HT impacts the risk of breast cancer.
The Women’s Health Initiative (WHI) represents the largest and longest-term randomized trial assessing the health impacts of systemic HT. A 2013 WHI report found that with a median of 13 years’ cumulative follow-up, estrogen-only HT (ET) reduced the risk for breast cancer while estrogen-progestin therapy (EPT) increased the risk.1 In a July 2020 issue of JAMA, WHI investigators analyzed longer-term data (cumulative median follow-up >20 years), which allowed assessment of whether these trends (breast cancer incidence) persisted and if they led to changes in mortality from breast cancer.2
WHI data on breast cancer risk trends in ET vs EPT users
In the ET trial, in which Chlebowski and colleagues studied 10,739 women with prior hysterectomy, 238 versus 296 new cases of breast cancer were diagnosed in women in the ET versus placebo groups, respectively (annualized incidence, 0.30% [ET] vs 0.37% [placebo]; hazard ratio [HR], 0.78; P = .005). ET also was associated with significantly lower mortality from breast cancer: 30 versus 46 deaths (annualized mortality, 0.031% [ET] vs 0.046% [placebo]; HR, 0.60; P = 0.04).
In the EPT trial, which included 16,608 participants with an intact uterus, EPT compared with placebo was associated with significantly elevated risk for incident breast cancer: 584 versus 447 new cases, respectively (annualized incidence, 0.45% [EPT] vs 0.36% [placebo]; HR, 1.28; P<.001). However, mortality from breast cancer was similar in the EPT and placebo groups: 71 and 53 deaths (annualized mortality, 0.045% [EPT] and 0.035% [placebo]; HR, 1.35; P = .11).2
For women with previous hysterectomy who are considering initiating or continuing ET for treatment of bothersome menopausal symptoms, the breast cancer mortality benefit documented in this long-term WHI analysis could, as editorialists point out, “tip the scales” in favor of ET.3 Furthermore, the mortality benefit raises the possibility that ET could be evaluated as a risk-reduction strategy for selected high-risk menopausal women who have undergone hysterectomy. Although tamoxifen and aromatase inhibitors are approved for breast cancer chemoprophylaxis in high-risk menopausal women, these agents have not been found to lower breast cancer mortality.2
UK data analysis and risk for breast cancer in HT users
In an October 2020 issue of BMJ, Vinogradova and colleagues described their analysis of 2 primary care databases in the United Kingdom that in aggregate included roughly 99,000 women with breast cancer diagnosed between 1998 and 2018 (age range, 50–79; mean age at diagnosis, 63; >95% White); these were matched with more than 450,000 women without breast cancer (controls).4 Analyses were adjusted for smoking, body mass index (BMI), ethnicity, and mammography.
In this study, ever-use of EPT was associated with an adjusted odds ratio (OR) for breast cancer of 1.26 (95% confidence interval [CI], 1.24–1.29), while ET had an OR of 1.06 (95% CI, 1.03–1.10). In women aged 50 to 59 who used EPT for 5 years or more, 15 additional breast cancers were diagnosed per 10,000 woman-years; for ET users, the attributable risk was 3. Although risk rose with longer HT duration, this trend was less evident with ET than EPT.
In addition, the increased risk associated with ET use was less pronounced in women with a BMI greater than 30 kg/m2. Among EPT users, risks were similar with the progestins medroxyprogesterone acetate (MPA), norethindrone (NET), and levonorgestrel (LNG). Likewise, risks were similar regardless of estrogen dose and route of administration (that is, oral vs transdermal). Vaginal estrogen was not associated with a higher or lower risk for breast cancer. Among past users of ET or EPT (with MPA), no increased risk was noted 5 years or more after stopping HT. For users of EPT (with NET or LNG), risks diminished 5 years or more after stopping HT but remained modestly elevated compared with risk in never-users.4
In this large observational UK study, ET was associated with minimally elevated risk for breast cancer, while in the WHI study, ET reduced the risk for breast cancer. For EPT, the excess risk in both studies was identical. As the authors note, mean BMI in the UK study participants was slightly lower than that in the WHI participants, a distinction that might explain the differing findings with ET use.
In our practice, for women with an intact uterus who are considering the use of EPT for treatment of bothersome menopausal symptoms, we counsel that long-term use of HT slightly elevates the risk for breast cancer. By contrast, we advise posthysterectomy women with bothersome menopausal symptoms that ET does not appear to increase the risk for breast cancer.
Continue to: Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women...
Frequency of nonindicated BSO at the time of hysterectomy in pre- and perimenopausal women
Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2020;28:8-11. doi: 10.1097/GME.0000000000001652.
While prevention of ovarian cancer is an important benefit of bilateral salpingo-oophorectomy (BSO), performing a BSO at the time of hysterectomy in pre- or perimenopausal patients not only will induce surgical menopause but also is associated with significantly increased overall mortality and an increased risk of mortality due to cardiovascular disease in patients younger than age 45.5,6 Earlier BSO also has been associated with diabetes, accelerated bone density loss, sexual dysfunction, mood disorders, and decreased cognitive function.7
BSO at hysterectomy: How many procedures are not indicated?
To evaluate the prevalence and predictors of unnecessary BSO at the time of hysterectomy, Wong and colleagues conducted a multicenter retrospective review of hysterectomy procedures completed at 6 Canadian hospitals.8 Criteria for unnecessary BSO included age younger than 51 years; benign preoperative diagnosis (other than endometriosis, premenstrual dysphoric disorder, and gender dysphoria); and absence of endometriosis and pelvic adhesions.
A total of 2,656 hysterectomies were performed by 75 surgeons (28 fellowship trained and 47 generalists) across 3 community and 3 tertiary care hospitals between 2016 and 2018. At the time of hysterectomy, 749 patients (28%) underwent BSO. Of these, 509 women (68%) had at least 1 indication for concurrent BSO based on preoperative diagnosis.
Key study findings. Concurrent BSO procedures performed at academic hospitals were more likely to have a preoperative indication compared with BSO performed at community sites (70% vs 63%; OR, 1.42; 95% CI, 1.02–1.97; P = .04). BSO was more likely to be indicated when performed by fellowship-trained surgeons compared with surgeries performed by generalist surgeons (75% vs 63%; OR, 1.76; 95% CI, 1.26–2.44, P = .001). BSO procedures performed with vaginal hysterectomy were less likely to be indicated (3 of 20, 15%) when compared with open hysterectomy (74 of 154, 48%) and laparoscopic hysterectomy (432 of 575, 75%).
Of the patients who lacked a preoperative indication for concomitant BSO, 105 of 239 (43.9%) were younger than age 51. Overall, 8% (59 of 749) of patients in the study cohort had an unnecessary BSO based on a combination of preoperative diagnosis, age younger than age 51, and intraoperative factors including absence of endometriosis and adhesions.
The retrospective study by Wong and colleagues provides the first assessment of Canadian practice patterns with respect to concurrent BSO at the time of hysterectomy. The authors found that, overall, more than two-thirds of BSO procedures were indicated. However, the proportion of BSO that was indicated was higher in teaching hospitals and in surgeries performed by fellowship-trained gynecologists. These important observations underscore the role of clinician education in reducing nonindicated BSO in pre- and perimenopausal women undergoing hysterectomy for benign disease.
Continue to: HT for menopausal depression: Which patients may benefit?
HT for menopausal depression: Which patients may benefit?
Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686- 705. doi:10.1176/appi.ajp.2020.19080848.
The cumulative lifetime prevalence of major depression in US women is 21%.9 An increased risk of mood symptoms and major depressive disorder occurs with the cessation of ovarian hormone production during menopause. In a review of both physiology and clinical studies, an American Psychiatric Association task force found support for several hormone-related strategies for treating depression and highlighted the rapidly advancing, but mixed, findings in this field.10
Clinical trials that examined mood in peri- and postmenopausal women treated with HT have produced mixed results for a variety of reasons, including differences in psychiatric symptomatology across studies and differences in treatment timing in relation to menopause onset.
HT effectiveness for depression depends on menopausal status
Five studies included in a meta-analysis by Rubinow and colleagues examined the use of ET and EPT as antidepressant monotherapy in peri- or postmenopausal women with major depression.11 Of the 3 higher-quality studies, 2 conducted in perimenopausal women demonstrated the antidepressant efficacy of transdermal estrogen patches compared with placebo. The third study included a mixed population of both peri- and postmenopausal women, and it found that increased estradiol levels (spontaneously occurring or due to ET) were associated with improvement in depression in perimenopausal women but not in postmenopausal women.11
ET also has been investigated as a potential adjunctive treatment to selective serotonin reuptake inhibitors (SSRIs). In a retrospective analysis of a multicenter randomized controlled trial of fluoxetine in patients with depression, women who received ET and fluoxetine demonstrated a greater improvement than those who received fluoxetine monotherapy.12 One small study that prospectively assessed ET in combination with an antidepressant in postmenopausal women demonstrated no benefit of ET in treating depression.13 Another small trial found that while combining transdermal ET with an SSRI accelerated symptom improvement, by the end of the 10-week study, treatment efficacy in the HT plus SSRI group was no greater than that observed in the SSRI-only group.14
Nineteen studies included in the metaanalysis by Rubinow and colleagues, which examined mood after ET or EPT treatment in nondepressed women, found little evidence of benefit, particularly in women without other physical symptoms of menopause.11
The Kronos Early Estrogen Prevention Study (KEEPS) followed 661 women who received either oral estrogen plus progesterone, transdermal estrogen plus progesterone, or placebo over 4 years.15 Women with clinical depression were excluded from the study; however, women with mild to moderate mood symptoms who were being treated with an antidepressant were included. Improvements in depressive symptoms and anxiety were observed only in the oral estrogen plus progesterone group compared with the placebo group.15
In a study of 172 euthymic peri- and postmenopausal women treated for 12 months with transdermal estrogen plus oral progesterone, investigators found that, unlike postmenopausal women and those in the late perimenopausal transition, only women in the early perimenopausal transition had a lower risk of developing depressive symptoms.16
Bottom line
This complex literature suggests that ET/HT interventions are most likely to be successful when implemented early in the menopausal transition. The clearest indication for the use of HT is for perimenopausal women experiencing depression who are also experiencing menopausal symptoms (for example, bothersome hot flashes). There is little evidence that the use of ET/HT in late perimenopausal or postmenopausal women effectively treats depression; accordingly, HT is not recommended for the treatment of mood disorders in this population. The more ambiguous cases are those of perimenopausal women who are depressed but do not have classic vasomotor symptoms; some evidence supports the antidepressant efficacy of HT in this setting.11 Although some studies suggest that HT can be effective in preventing depression in perimenopausal women, more evidence is needed.16
A trial of ET/EPT is reasonable in perimenopausal women with depression and classic menopausal symptoms. Use of HT also can be considered either alone or in combination with an SSRI in perimenopausal women with depression who do not have significant classic menopausal symptoms. However, HT is not recommended as prophylaxis against depression in euthymic perimenopausal women. Finally, keep in mind that the use of HT to address mood issues constitutes off-label use.
The menopause transition: A key period for strategizing CVD risk factor reduction

Chrisandra L. Shufelt, MD, MS, NCMP
Dr. Shufelt is Associate Director of the Barbra
Streisand Women’s Heart Center, Smidt
Heart Institute, Cedars-Sinai Medical Center,
Los Angeles, California.

JoAnn E. Manson, MD, DrPH, NCMP
Dr. Manson is Professor of Medicine and the
Michael and Lee Bell Professor of Women’s
Health at Harvard Medical School; Professor
in the Department of Epidemiology, Harvard
T.H. Chan School of Public Health; and Chief
of the Division of Preventive Medicine
at Brigham and Women’s Hospital, Boston,
Massachusetts.
The authors report no financial relationships relevant to this article. Dr. Manson is a coauthor of the AHA Scientific Statement discussed in this article.
In the United States, nearly one-half of a woman’s life, on average, will be lived after menopause. For women with natural menopause, the menopause transition (MT) can begin 2 to 7 years before and may extend 1 year past the final menstrual period, which occurs at an average age of 51 years. For women with surgical menopause, the MT occurs abruptly with the sudden loss of endogenous ovarian hormones. Both types of transitions mark a critical time period when reproduction and endogenous sex hormone levels diminish and when cardiovascular disease (CVD) risk factors begin to rise.
The 2020 American Heart Association (AHA) scientific statement, “Menopause transition and cardiovascular disease risk: Implications for timing of early prevention,” highlights the MT as a window of opportunity for CVD prevention.1
CVD risk factors associated with ovarian aging
In the AHA scientific statement, data from several longitudinal women’s health studies were used to identify which CVD risk factor changes during the MT are related to ovarian aging as opposed to chronologic aging. Independent of aging, those associated with reproductive or ovarian aging included an increase in serum total cholesterol, low-density lipoprotein cholesterol (LDL-C), and apolipoprotein B. Changes in high-density lipoprotein cholesterol (HDL-C) particles and function also occur during the MT, which may explain why higher HDL-C levels during the MT and the postmenopausal years are not as cardioprotective as during the premenopausal period.
Changes in body composition and adipose tissue distribution also are associated with ovarian aging, with reduction in muscle mass and lean body mass and an increase in abdominal/visceral fat and subcutaneous adipose tissue. Although these body composition changes reflect ovarian aging, midlife weight gain is more closely related to chronologic aging.
The risk of the metabolic syndrome constellation of risk factors was found to be more closely associated with ovarian aging, whereas changes in blood pressure, insulin, and glucose individually tracked more closely with chronologic aging. Additionally, the AHA statement notes the research that identified several symptoms during the MT—including vasomotor symptoms, sleep disturbance, and depression—as being associated with more adverse CVD risk factor status and with subclinical measures of atherosclerosis. Additional research on the mechanistic basis for these associations is needed.
Chronologic age and type of menopause
Notably, a woman’s age and type of menopause matter with respect to CVD risk. Higher CVD risk is seen in women with premature onset (age < 40 years) or early onset (age < 45 years) of menopause and in women undergoing surgical menopause (bilateral oophorectomy) before age 45. In general, menopausal hormone therapy (HT) is recommended for women with premature or early menopause, whether natural or surgical, with continuation through at least the average age of natural menopause. In other women, although not recommended for the express purpose of CVD prevention, menopausal HT is appropriate for the treatment of bothersome vasomotor or other menopausal symptoms, especially when therapy is started before age 60 or within 10 years of menopause among women who are not at elevated risk of CVD.
While the AHA statement suggests that some women who begin estrogen early in menopause may experience reduced coronary heart disease risk, major research gaps remain with regard to HT dose, formulation, route of delivery, and recommended duration of treatment.
An opportunity to promote healthy lifestyle behaviors
Translating the AHA’s first-of-its-kind scientific statement into clinical practice requires recognition and awareness of the MT as a unique phase in a woman’s life associated with myriad changes in CVD risk factors. The statement underscores that the MT is an important time to target behavioral changes to promote CVD risk reduction, including lifestyle modifications in the AHA’s Life’s Simple 7 components (increased physical activity, smoking cessation, healthy diet, avoidance of weight gain) as well as vigilant control of blood pressure, cholesterol, and glucose levels. The MT is truly a window of opportunity for reinvigorated efforts to lower women’s CVD risk. ●
Reference
1. El Khoudary SR, Aggarwal B, Beckie TM, et al; American Heart Association Prevention Science Committee of the Council on Epidemiology and Prevention; and Council on Cardiovascular and Stroke Nursing. Menopause transition and cardiovascular disease risk: implications for timing of early prevention: a scientific statement from the American Heart Association. Circulation. 2020;142:e506-e532. doi: 10.1161/CIR.000000000000912.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310:1353- 1368. doi: 10.1001/jama.2013.278040.
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Minami CA, Freedman RA. Menopausal hormone therapy and long-term breast cancer risk: further data from the Women’s Health Initiative trials. JAMA. 2020;324:347-349. doi: 10.1001/jama.2020.9620.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
- Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279. doi: 10.1016/j.ajog.2017.07.037.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15-23. doi: 10.1097/gme.0b013e31818888f7.
- Karp NE, Fenner DE, Burgunder-Zdravkovski L, et al. Removal of normal ovaries in women under age 51 at the time of hysterectomy. Am J Obstetr Gynecol. 2015;213:716.e1-6. doi: 10.1016/j.ajog.2015.05.062.
- Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2021;28:8-11. doi: 10.1097/GME.0000000000001652.
- Kessler RC, McGonagle KA, Swartz M, et al. Sex and depression in the National Comorbidity Survey. I: lifetime prevalence, chronicity, and recurrence. J Affect Disord. 1993;29:85- 96. doi: 10.1016/0165-0327(93)00026-g.
- Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686-705. doi:10.1176/appi. ajp.2020.19080848.
- Rubinow DR, Johnson SL, Schmidt PJ, et al. Efficacy of estradiol in perimenopausal depression: so much promise and so few answers. Depress Anxiety. 2015;32:539-549. doi: 10.1002/ da.22391.
- Schneider LS, Small GW, Hamilton SH, et al. Estrogen replacement and response to fluoxetine in a multicenter geriatric depression trial. Fluoxetine Collaborative Study Group. Am J Geriatr Psychiatry. 1997;5:97-106.
- Dias RS, Kerr-Corrêa F, Moreno RA, et al. Efficacy of hormone therapy with and without methyltestosterone augmentation of venlafaxine in the treatment of postmenopausal depression: a double-blind controlled pilot study. Menopause. 2006;13:202-211. doi:10.1097/01.gme.0000198491.34371.9c.
- Rasgon NL, Dunkin J, Fairbanks L, et al. Estrogen and response to sertraline in postmenopausal women with major depressive disorder: a pilot study. J Psychiatr Res. 2007;41:338- 343. doi: 10.1016/j.jpsychires.2006.03.009.
- Gleason CE, Dowling NM, Wharton W, et al. Effects of hormone therapy on cognition and mood in recently postmenopausal women: findings from the randomized, controlled KEEPS–cognitive and affective study. PLoS Med. 2015;12:e1001833. doi: 10.1371/journal.pmed.1001833.
- Gordon JL, Rubinow DR, Eisenlohr-Moul TA, et al. Efficacy of transdermal estradiol and micronized progesterone in the prevention of depressive symptoms in the menopause transition: a randomized clinical trial. JAMA Psychiatry. 2018;75:149–157. doi:10.1001/jamapsychiatry.2017.3998.
- Manson JE, Chlebowski RT, Stefanick ML, et al. Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the Women’s Health Initiative randomized trials. JAMA. 2013;310:1353- 1368. doi: 10.1001/jama.2013.278040.
- Chlebowski RT, Anderson GL, Aragaki AK, et al. Association of menopausal hormone therapy with breast cancer incidence and mortality during long-term follow-up of the Women’s Health Initiative randomized clinical trials. JAMA. 2020;324:369-380. doi: 10.1001/jama.2020.9482.
- Minami CA, Freedman RA. Menopausal hormone therapy and long-term breast cancer risk: further data from the Women’s Health Initiative trials. JAMA. 2020;324:347-349. doi: 10.1001/jama.2020.9620.
- Vinogradova Y, Coupland C, Hippisley-Cox J. Use of hormone replacement therapy and risk of breast cancer: nested case-control studies using the QResearch and CPRD databases. BMJ. 2020;371:m3873. doi: 10.1136/bmj.m3873.
- Adelman MR, Sharp HT. Ovarian conservation vs removal at the time of benign hysterectomy. Am J Obstet Gynecol. 2018;218:269-279. doi: 10.1016/j.ajog.2017.07.037.
- Rivera CM, Grossardt BR, Rhodes DJ, et al. Increased cardiovascular mortality after early bilateral oophorectomy. Menopause. 2009;16:15-23. doi: 10.1097/gme.0b013e31818888f7.
- Karp NE, Fenner DE, Burgunder-Zdravkovski L, et al. Removal of normal ovaries in women under age 51 at the time of hysterectomy. Am J Obstetr Gynecol. 2015;213:716.e1-6. doi: 10.1016/j.ajog.2015.05.062.
- Wong J, Murji A, Sunderji Z, et al. Unnecessary bilateral salpingo-oophorectomy at the time of hysterectomy and potential for ovarian preservation. Menopause. 2021;28:8-11. doi: 10.1097/GME.0000000000001652.
- Kessler RC, McGonagle KA, Swartz M, et al. Sex and depression in the National Comorbidity Survey. I: lifetime prevalence, chronicity, and recurrence. J Affect Disord. 1993;29:85- 96. doi: 10.1016/0165-0327(93)00026-g.
- Dwyer JB, Aftab A, Radhakrishnan R, et al; APA Council of Research Task Force on Novel Biomarkers and Treatments. Hormonal treatments for major depressive disorder: state of the art. Am J Psychiatry. 2020;177:686-705. doi:10.1176/appi. ajp.2020.19080848.
- Rubinow DR, Johnson SL, Schmidt PJ, et al. Efficacy of estradiol in perimenopausal depression: so much promise and so few answers. Depress Anxiety. 2015;32:539-549. doi: 10.1002/ da.22391.
- Schneider LS, Small GW, Hamilton SH, et al. Estrogen replacement and response to fluoxetine in a multicenter geriatric depression trial. Fluoxetine Collaborative Study Group. Am J Geriatr Psychiatry. 1997;5:97-106.
- Dias RS, Kerr-Corrêa F, Moreno RA, et al. Efficacy of hormone therapy with and without methyltestosterone augmentation of venlafaxine in the treatment of postmenopausal depression: a double-blind controlled pilot study. Menopause. 2006;13:202-211. doi:10.1097/01.gme.0000198491.34371.9c.
- Rasgon NL, Dunkin J, Fairbanks L, et al. Estrogen and response to sertraline in postmenopausal women with major depressive disorder: a pilot study. J Psychiatr Res. 2007;41:338- 343. doi: 10.1016/j.jpsychires.2006.03.009.
- Gleason CE, Dowling NM, Wharton W, et al. Effects of hormone therapy on cognition and mood in recently postmenopausal women: findings from the randomized, controlled KEEPS–cognitive and affective study. PLoS Med. 2015;12:e1001833. doi: 10.1371/journal.pmed.1001833.
- Gordon JL, Rubinow DR, Eisenlohr-Moul TA, et al. Efficacy of transdermal estradiol and micronized progesterone in the prevention of depressive symptoms in the menopause transition: a randomized clinical trial. JAMA Psychiatry. 2018;75:149–157. doi:10.1001/jamapsychiatry.2017.3998.