User login
Improvement needed for U.S. acute care hospitals implementing ASPs
There is more room for improving U.S. acute care hospitals’ antibiotic stewardship programs (ASPs) and implementing the seven core elements, according to findings from the 2014 National Healthcare Safety Network (NHSN) Annual Hospital Survey.
In univariate analyses, Dr. Lori A. Pollack, of the Division of Cancer Prevention and Control at the Centers for Disease Control and Prevention, and her associates looked at 4,184 acute care hospitals. Of those acute care hospitals, 1,642 (39%) reported implementing all seven CDC-defined core elements – leadership commitment, a single program leader responsible for outcomes, a pharmacy leader, specific interventions to improve prescribing, tracking antibiotic use and resistance, reporting data back to provider, and education – for hospital ASPs. In the hospitals with more than 200 beds, 775 (56%) were more likely to report all seven core elements, compared with 672 (39%) hospitals with 51-200 beds, and 328 (22%) of hospitals with 50 or fewer beds.
The hospitals with 50 or fewer beds were less likely to report leadership support (40%) or antibiotic stewardship education (46%), compared with facilities with greater than 50 beds (69% leadership, 69% education). Also, the major teaching hospitals were more likely to report all seven core elements (54%) than were hospitals that had only undergraduate education or no teaching affiliation (34%).
The study also conducted a final multivariate model and found that the strongest predictor for meeting all seven core elements was support from the facility administration (adjusted relative risk, 7.2; P less than .0001).
“Our findings suggest that many hospitals need to add infrastructure and measurement support to their current actions to improve antibiotic use,” the researchers concluded. “CDC is committed to on-going work with partners to help all hospitals implement effective antibiotic stewardship programs, and future years of this survey will help monitor progress toward that goal.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw323).
There is more room for improving U.S. acute care hospitals’ antibiotic stewardship programs (ASPs) and implementing the seven core elements, according to findings from the 2014 National Healthcare Safety Network (NHSN) Annual Hospital Survey.
In univariate analyses, Dr. Lori A. Pollack, of the Division of Cancer Prevention and Control at the Centers for Disease Control and Prevention, and her associates looked at 4,184 acute care hospitals. Of those acute care hospitals, 1,642 (39%) reported implementing all seven CDC-defined core elements – leadership commitment, a single program leader responsible for outcomes, a pharmacy leader, specific interventions to improve prescribing, tracking antibiotic use and resistance, reporting data back to provider, and education – for hospital ASPs. In the hospitals with more than 200 beds, 775 (56%) were more likely to report all seven core elements, compared with 672 (39%) hospitals with 51-200 beds, and 328 (22%) of hospitals with 50 or fewer beds.
The hospitals with 50 or fewer beds were less likely to report leadership support (40%) or antibiotic stewardship education (46%), compared with facilities with greater than 50 beds (69% leadership, 69% education). Also, the major teaching hospitals were more likely to report all seven core elements (54%) than were hospitals that had only undergraduate education or no teaching affiliation (34%).
The study also conducted a final multivariate model and found that the strongest predictor for meeting all seven core elements was support from the facility administration (adjusted relative risk, 7.2; P less than .0001).
“Our findings suggest that many hospitals need to add infrastructure and measurement support to their current actions to improve antibiotic use,” the researchers concluded. “CDC is committed to on-going work with partners to help all hospitals implement effective antibiotic stewardship programs, and future years of this survey will help monitor progress toward that goal.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw323).
There is more room for improving U.S. acute care hospitals’ antibiotic stewardship programs (ASPs) and implementing the seven core elements, according to findings from the 2014 National Healthcare Safety Network (NHSN) Annual Hospital Survey.
In univariate analyses, Dr. Lori A. Pollack, of the Division of Cancer Prevention and Control at the Centers for Disease Control and Prevention, and her associates looked at 4,184 acute care hospitals. Of those acute care hospitals, 1,642 (39%) reported implementing all seven CDC-defined core elements – leadership commitment, a single program leader responsible for outcomes, a pharmacy leader, specific interventions to improve prescribing, tracking antibiotic use and resistance, reporting data back to provider, and education – for hospital ASPs. In the hospitals with more than 200 beds, 775 (56%) were more likely to report all seven core elements, compared with 672 (39%) hospitals with 51-200 beds, and 328 (22%) of hospitals with 50 or fewer beds.
The hospitals with 50 or fewer beds were less likely to report leadership support (40%) or antibiotic stewardship education (46%), compared with facilities with greater than 50 beds (69% leadership, 69% education). Also, the major teaching hospitals were more likely to report all seven core elements (54%) than were hospitals that had only undergraduate education or no teaching affiliation (34%).
The study also conducted a final multivariate model and found that the strongest predictor for meeting all seven core elements was support from the facility administration (adjusted relative risk, 7.2; P less than .0001).
“Our findings suggest that many hospitals need to add infrastructure and measurement support to their current actions to improve antibiotic use,” the researchers concluded. “CDC is committed to on-going work with partners to help all hospitals implement effective antibiotic stewardship programs, and future years of this survey will help monitor progress toward that goal.”
Read the full study in Clinical Infectious Diseases (doi: 10.1093/cid/ciw323).
FROM CLINICAL INFECTIOUS DISEASES
Peter Kramer returns to his role in defending antidepressants in ‘Ordinarily Well’
With his 1993 landmark book, “Listening to Prozac,” psychiatrist Peter D. Kramer became one of the most famous psychiatrists in America – second perhaps only to the fictional Frasier Crane of primetime TV. Since then, Dr. Kramer has continued to write – including a novel, books on other psychiatric topics, a blog, and articles for mainstream media, including the New York Times. In his latest book, “Ordinarily Well: The Case for Antidepressants,” Dr. Kramer will return to the role he left behind 23 years ago: defending the use of antidepressants.
Dr. Kramer notes that he doesn’t do this easily. In the preface, with a bit of dismay, he talks about how reviewers have called him “Dr. Prozac.” He’s felt stuck on a wave that didn’t reflect his diverse interests, and he didn’t want to be covered by the spreading stain of Big Pharma.
“Against all indications, I remained hopeful that I might walk free by day, alter my obituary,” Dr. Kramer tells the reader.
With “Ordinarily Well,” Peter Kramer is back as Dr. Prozac. The book is written as a response to the “research”– now seen so often in headlines – that antidepressants are as effective as sugar pills for mild to moderate depression, and they should be prescribed only for severe major depression. He gets even more specific: The book is written partly in response to an article by Dr. Marcia Angell, former editor in chief of the New England Journal of Medicine, published in June 2011 in the New York Review of Books, and Dr. Angell’s assertion that psychiatric medications are no more effective than placebo. Since psychotropics come with more side effects than placebos, the next logical conclusion is that they are not only ineffective – they are harmful.
As a clinical psychiatrist, I’ve found this evidence-based stance to be perplexing. People get better on antidepressants, even if you aren’t measuring Hamilton rating scores and even if you aren’t limiting treatment to those with severe major depression. I would estimate that at least some of the people, some of the time, get better, and when you progress to strategies of switching and augmenting, most of the the people, most of the time, get significant relief from their major depression.
Obviously, this is my clinical impression and not research, and Dr. Kramer takes a more ardent stance: Most patients with depression, be it mild, moderate, severe, or long-standing dysthymia, have a good response to antidepressants. It’s the minority who don’t respond.
Dr. Kramer goes through the “science” that would suggest that antidepressants are not effective for milder forms of depression, dysthymia, and neuroticism. He does a systematic and comprehensive review of how pharmaceutical studies are conducted, and what factors might skew results, and there is plenty here to fill the pages. He explains complex issues – such as meta-analyses and numbers needed to treat – such that the lay reader can follow.
As just one example, Dr. Kramer talks about screening research subjects for participation in antidepressant studies: “If raters have a sense of the minimum Hamilton score for admission to a study, and if they are under pressure to fill an enrollment quota, they will be inclined to tack on questionable Hamilton points. The boost will not be uniform. There’s no need to raise rating in the very ill. Scores for least afflicted participants will be most inflated.
“When off-site raters, with no stake in the pace of enrollment, analyze tapes of admission interviews,” he continues, “they find patients to be much healthier than the on-site Hamilton scores suggest. According to off-site assessments, many patients admitted to drug studies simply are not depressed.”
Dr. Kramer methodically marches through problems with finding patients for the studies, shortcomings of the Hamilton rating scale, which gives suicidal ideation the same point as a somatic symptom; the bias some studies have of excluding people with severe depression; substance abuse, or comorbid disorders; and “the floor effect,” which underestimates efficacy in patients with fewer symptoms.
He follows subjects at an unnamed for-profit research center and praises the skills of everyone who comes in contact with the research subjects, including the friendly van driver who fetches patients from their homes.
In a chapter titled “How We’re Doing,” Dr. Kramer goes into detail about specific studies, including the STAR*D trials, where patients were recruited from primary care and psychiatric clinics with the guarantee that they would be provided active medication, and those with comorbid conditions were not excluded.
“Only the sickest came. For nearly 80% of the participants (more than 2,800 were tested), the disorder was chronic. The average length of the current depressive episode was more than 2 years, generally despite attempts at treatment. The average enrollee had lived with depression on and off for more than15 years and was now in a seventh episode. Most patients were alcoholics or had other forms of mental illness.
“In the first phase, patients were put on Celexa [citalopram], managed by their own doctors. About 30% of patients achieved remission within weeks – with virtually no symptoms. Responses (including remissions) ran at just under 50%.
“Commentators considered this outcome disappointing, but is it?”
So this is Dr. Kramer’s strength: He writes an engaging book about a complex topic, arguing throughout that antidepressants work well and have been given a bum rap by flawed research and careless journalism that enjoy the sensationalism of villainizing psychotropics. There are no headlines, he points out, singing the praises of antidepressants for milder forms of depression, even when the evidence is there. Despite the complexity of the topic and the breadth of his research reviews, Dr. Kramer tells the story of antidepressant research in a way that a lay reader can follow. There are no mice or moleculars, no genetic loci, and no explanations of cytochrome P450 metabolism, neurotransmission, or synaptic blockade. The complexities are explained without medical lingo and, in the end, he concludes what psychiatrists see every day: Antidepressants work. They work for the sickest of the sick, and they work for those who are suffering from less-severe forms of depression.
Dr. Kramer ends the book with a discussion of his own clinical experiences, which are not always in tune with what the “science” declares to be true. He cites studies that show that psychotherapy adds nothing to the treatment of depression, yet still, he treats his own patients with psychotherapy. He notes studies that show maximal efficacy when medications are used at high doses and continue for the long haul at these high doses, yet in his own practice, he sometimes uses lower doses and weans patients off medications. He does a wonderful job of pointing out the disconnect of the promise of evidence-based medicine and how its usefulness has limits in clinical practice.
In our communications, Dr. Kramer wrote to me: “There were many reasons not to write this book, and I was reluctant. I took up the topic only after declining a series of opportunities to weigh in. This book is the only one of mine that I wrote primarily out of a sense of duty. The debunking of antidepressants had gone too far and been too widely accepted, and I believed that the underlying research was shaky.”
He worried that I saw his view of antidepressants as more favorable than he intended it to be, and in fact, his perception is correct: While I prescribe antidepressants and see their benefits (as well as their side effects) with many of my patients, I believe Peter Kramer is more enthusiastic than I am about the efficacy of antidepressants for milder forms of depression, dysthymia, and neuroticism.
“My view is the one expressed in the title,” Dr. Kramer countered, “Our medications work ordinarily well, and they bring patients to a state of ordinary wellness.”
“Ordinarily Well” will be available on June 7; it is available for preorder on Amazon now.
Dr. Miller is a coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work” (Baltimore: Johns Hopkins University Press, 2011).
With his 1993 landmark book, “Listening to Prozac,” psychiatrist Peter D. Kramer became one of the most famous psychiatrists in America – second perhaps only to the fictional Frasier Crane of primetime TV. Since then, Dr. Kramer has continued to write – including a novel, books on other psychiatric topics, a blog, and articles for mainstream media, including the New York Times. In his latest book, “Ordinarily Well: The Case for Antidepressants,” Dr. Kramer will return to the role he left behind 23 years ago: defending the use of antidepressants.
Dr. Kramer notes that he doesn’t do this easily. In the preface, with a bit of dismay, he talks about how reviewers have called him “Dr. Prozac.” He’s felt stuck on a wave that didn’t reflect his diverse interests, and he didn’t want to be covered by the spreading stain of Big Pharma.
“Against all indications, I remained hopeful that I might walk free by day, alter my obituary,” Dr. Kramer tells the reader.
With “Ordinarily Well,” Peter Kramer is back as Dr. Prozac. The book is written as a response to the “research”– now seen so often in headlines – that antidepressants are as effective as sugar pills for mild to moderate depression, and they should be prescribed only for severe major depression. He gets even more specific: The book is written partly in response to an article by Dr. Marcia Angell, former editor in chief of the New England Journal of Medicine, published in June 2011 in the New York Review of Books, and Dr. Angell’s assertion that psychiatric medications are no more effective than placebo. Since psychotropics come with more side effects than placebos, the next logical conclusion is that they are not only ineffective – they are harmful.
As a clinical psychiatrist, I’ve found this evidence-based stance to be perplexing. People get better on antidepressants, even if you aren’t measuring Hamilton rating scores and even if you aren’t limiting treatment to those with severe major depression. I would estimate that at least some of the people, some of the time, get better, and when you progress to strategies of switching and augmenting, most of the the people, most of the time, get significant relief from their major depression.
Obviously, this is my clinical impression and not research, and Dr. Kramer takes a more ardent stance: Most patients with depression, be it mild, moderate, severe, or long-standing dysthymia, have a good response to antidepressants. It’s the minority who don’t respond.
Dr. Kramer goes through the “science” that would suggest that antidepressants are not effective for milder forms of depression, dysthymia, and neuroticism. He does a systematic and comprehensive review of how pharmaceutical studies are conducted, and what factors might skew results, and there is plenty here to fill the pages. He explains complex issues – such as meta-analyses and numbers needed to treat – such that the lay reader can follow.
As just one example, Dr. Kramer talks about screening research subjects for participation in antidepressant studies: “If raters have a sense of the minimum Hamilton score for admission to a study, and if they are under pressure to fill an enrollment quota, they will be inclined to tack on questionable Hamilton points. The boost will not be uniform. There’s no need to raise rating in the very ill. Scores for least afflicted participants will be most inflated.
“When off-site raters, with no stake in the pace of enrollment, analyze tapes of admission interviews,” he continues, “they find patients to be much healthier than the on-site Hamilton scores suggest. According to off-site assessments, many patients admitted to drug studies simply are not depressed.”
Dr. Kramer methodically marches through problems with finding patients for the studies, shortcomings of the Hamilton rating scale, which gives suicidal ideation the same point as a somatic symptom; the bias some studies have of excluding people with severe depression; substance abuse, or comorbid disorders; and “the floor effect,” which underestimates efficacy in patients with fewer symptoms.
He follows subjects at an unnamed for-profit research center and praises the skills of everyone who comes in contact with the research subjects, including the friendly van driver who fetches patients from their homes.
In a chapter titled “How We’re Doing,” Dr. Kramer goes into detail about specific studies, including the STAR*D trials, where patients were recruited from primary care and psychiatric clinics with the guarantee that they would be provided active medication, and those with comorbid conditions were not excluded.
“Only the sickest came. For nearly 80% of the participants (more than 2,800 were tested), the disorder was chronic. The average length of the current depressive episode was more than 2 years, generally despite attempts at treatment. The average enrollee had lived with depression on and off for more than15 years and was now in a seventh episode. Most patients were alcoholics or had other forms of mental illness.
“In the first phase, patients were put on Celexa [citalopram], managed by their own doctors. About 30% of patients achieved remission within weeks – with virtually no symptoms. Responses (including remissions) ran at just under 50%.
“Commentators considered this outcome disappointing, but is it?”
So this is Dr. Kramer’s strength: He writes an engaging book about a complex topic, arguing throughout that antidepressants work well and have been given a bum rap by flawed research and careless journalism that enjoy the sensationalism of villainizing psychotropics. There are no headlines, he points out, singing the praises of antidepressants for milder forms of depression, even when the evidence is there. Despite the complexity of the topic and the breadth of his research reviews, Dr. Kramer tells the story of antidepressant research in a way that a lay reader can follow. There are no mice or moleculars, no genetic loci, and no explanations of cytochrome P450 metabolism, neurotransmission, or synaptic blockade. The complexities are explained without medical lingo and, in the end, he concludes what psychiatrists see every day: Antidepressants work. They work for the sickest of the sick, and they work for those who are suffering from less-severe forms of depression.
Dr. Kramer ends the book with a discussion of his own clinical experiences, which are not always in tune with what the “science” declares to be true. He cites studies that show that psychotherapy adds nothing to the treatment of depression, yet still, he treats his own patients with psychotherapy. He notes studies that show maximal efficacy when medications are used at high doses and continue for the long haul at these high doses, yet in his own practice, he sometimes uses lower doses and weans patients off medications. He does a wonderful job of pointing out the disconnect of the promise of evidence-based medicine and how its usefulness has limits in clinical practice.
In our communications, Dr. Kramer wrote to me: “There were many reasons not to write this book, and I was reluctant. I took up the topic only after declining a series of opportunities to weigh in. This book is the only one of mine that I wrote primarily out of a sense of duty. The debunking of antidepressants had gone too far and been too widely accepted, and I believed that the underlying research was shaky.”
He worried that I saw his view of antidepressants as more favorable than he intended it to be, and in fact, his perception is correct: While I prescribe antidepressants and see their benefits (as well as their side effects) with many of my patients, I believe Peter Kramer is more enthusiastic than I am about the efficacy of antidepressants for milder forms of depression, dysthymia, and neuroticism.
“My view is the one expressed in the title,” Dr. Kramer countered, “Our medications work ordinarily well, and they bring patients to a state of ordinary wellness.”
“Ordinarily Well” will be available on June 7; it is available for preorder on Amazon now.
Dr. Miller is a coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work” (Baltimore: Johns Hopkins University Press, 2011).
With his 1993 landmark book, “Listening to Prozac,” psychiatrist Peter D. Kramer became one of the most famous psychiatrists in America – second perhaps only to the fictional Frasier Crane of primetime TV. Since then, Dr. Kramer has continued to write – including a novel, books on other psychiatric topics, a blog, and articles for mainstream media, including the New York Times. In his latest book, “Ordinarily Well: The Case for Antidepressants,” Dr. Kramer will return to the role he left behind 23 years ago: defending the use of antidepressants.
Dr. Kramer notes that he doesn’t do this easily. In the preface, with a bit of dismay, he talks about how reviewers have called him “Dr. Prozac.” He’s felt stuck on a wave that didn’t reflect his diverse interests, and he didn’t want to be covered by the spreading stain of Big Pharma.
“Against all indications, I remained hopeful that I might walk free by day, alter my obituary,” Dr. Kramer tells the reader.
With “Ordinarily Well,” Peter Kramer is back as Dr. Prozac. The book is written as a response to the “research”– now seen so often in headlines – that antidepressants are as effective as sugar pills for mild to moderate depression, and they should be prescribed only for severe major depression. He gets even more specific: The book is written partly in response to an article by Dr. Marcia Angell, former editor in chief of the New England Journal of Medicine, published in June 2011 in the New York Review of Books, and Dr. Angell’s assertion that psychiatric medications are no more effective than placebo. Since psychotropics come with more side effects than placebos, the next logical conclusion is that they are not only ineffective – they are harmful.
As a clinical psychiatrist, I’ve found this evidence-based stance to be perplexing. People get better on antidepressants, even if you aren’t measuring Hamilton rating scores and even if you aren’t limiting treatment to those with severe major depression. I would estimate that at least some of the people, some of the time, get better, and when you progress to strategies of switching and augmenting, most of the the people, most of the time, get significant relief from their major depression.
Obviously, this is my clinical impression and not research, and Dr. Kramer takes a more ardent stance: Most patients with depression, be it mild, moderate, severe, or long-standing dysthymia, have a good response to antidepressants. It’s the minority who don’t respond.
Dr. Kramer goes through the “science” that would suggest that antidepressants are not effective for milder forms of depression, dysthymia, and neuroticism. He does a systematic and comprehensive review of how pharmaceutical studies are conducted, and what factors might skew results, and there is plenty here to fill the pages. He explains complex issues – such as meta-analyses and numbers needed to treat – such that the lay reader can follow.
As just one example, Dr. Kramer talks about screening research subjects for participation in antidepressant studies: “If raters have a sense of the minimum Hamilton score for admission to a study, and if they are under pressure to fill an enrollment quota, they will be inclined to tack on questionable Hamilton points. The boost will not be uniform. There’s no need to raise rating in the very ill. Scores for least afflicted participants will be most inflated.
“When off-site raters, with no stake in the pace of enrollment, analyze tapes of admission interviews,” he continues, “they find patients to be much healthier than the on-site Hamilton scores suggest. According to off-site assessments, many patients admitted to drug studies simply are not depressed.”
Dr. Kramer methodically marches through problems with finding patients for the studies, shortcomings of the Hamilton rating scale, which gives suicidal ideation the same point as a somatic symptom; the bias some studies have of excluding people with severe depression; substance abuse, or comorbid disorders; and “the floor effect,” which underestimates efficacy in patients with fewer symptoms.
He follows subjects at an unnamed for-profit research center and praises the skills of everyone who comes in contact with the research subjects, including the friendly van driver who fetches patients from their homes.
In a chapter titled “How We’re Doing,” Dr. Kramer goes into detail about specific studies, including the STAR*D trials, where patients were recruited from primary care and psychiatric clinics with the guarantee that they would be provided active medication, and those with comorbid conditions were not excluded.
“Only the sickest came. For nearly 80% of the participants (more than 2,800 were tested), the disorder was chronic. The average length of the current depressive episode was more than 2 years, generally despite attempts at treatment. The average enrollee had lived with depression on and off for more than15 years and was now in a seventh episode. Most patients were alcoholics or had other forms of mental illness.
“In the first phase, patients were put on Celexa [citalopram], managed by their own doctors. About 30% of patients achieved remission within weeks – with virtually no symptoms. Responses (including remissions) ran at just under 50%.
“Commentators considered this outcome disappointing, but is it?”
So this is Dr. Kramer’s strength: He writes an engaging book about a complex topic, arguing throughout that antidepressants work well and have been given a bum rap by flawed research and careless journalism that enjoy the sensationalism of villainizing psychotropics. There are no headlines, he points out, singing the praises of antidepressants for milder forms of depression, even when the evidence is there. Despite the complexity of the topic and the breadth of his research reviews, Dr. Kramer tells the story of antidepressant research in a way that a lay reader can follow. There are no mice or moleculars, no genetic loci, and no explanations of cytochrome P450 metabolism, neurotransmission, or synaptic blockade. The complexities are explained without medical lingo and, in the end, he concludes what psychiatrists see every day: Antidepressants work. They work for the sickest of the sick, and they work for those who are suffering from less-severe forms of depression.
Dr. Kramer ends the book with a discussion of his own clinical experiences, which are not always in tune with what the “science” declares to be true. He cites studies that show that psychotherapy adds nothing to the treatment of depression, yet still, he treats his own patients with psychotherapy. He notes studies that show maximal efficacy when medications are used at high doses and continue for the long haul at these high doses, yet in his own practice, he sometimes uses lower doses and weans patients off medications. He does a wonderful job of pointing out the disconnect of the promise of evidence-based medicine and how its usefulness has limits in clinical practice.
In our communications, Dr. Kramer wrote to me: “There were many reasons not to write this book, and I was reluctant. I took up the topic only after declining a series of opportunities to weigh in. This book is the only one of mine that I wrote primarily out of a sense of duty. The debunking of antidepressants had gone too far and been too widely accepted, and I believed that the underlying research was shaky.”
He worried that I saw his view of antidepressants as more favorable than he intended it to be, and in fact, his perception is correct: While I prescribe antidepressants and see their benefits (as well as their side effects) with many of my patients, I believe Peter Kramer is more enthusiastic than I am about the efficacy of antidepressants for milder forms of depression, dysthymia, and neuroticism.
“My view is the one expressed in the title,” Dr. Kramer countered, “Our medications work ordinarily well, and they bring patients to a state of ordinary wellness.”
“Ordinarily Well” will be available on June 7; it is available for preorder on Amazon now.
Dr. Miller is a coauthor of “Shrink Rap: Three Psychiatrists Explain Their Work” (Baltimore: Johns Hopkins University Press, 2011).
Ozanimod linked to histologic healing of moderate, severe ulcerative colitis
SAN DIEGO – A new analysis of phase II research finds that the experimental drug ozanimod prompted histologic healing in ulcerative colitis patients at 8 and 32 weeks.
“Ozanimod both induces and maintains remissions in a proportion of patients with refractory ulcerative colitis,” said study coauthor Dr. Stephen B. Hanauer, professor of medicine and medical director of the Digestive Health Center at Northwestern University, Chicago. “It’s a safe and effective oral drug for ulcerative colitis and possibly Crohn’s disease.”
Ozanimod, an experimental drug developed by Celgene and its subsidiary Receptos, has been undergoing review as a treatment for both multiple sclerosis and ulcerative colitis.
“Ozanimod impacts lymphocyte trafficking by retaining lymphocytes in lymph nodes and prevents recirculation. The mechanism of action is impairing S1P [sphingosine-1-phosphate] that ‘traps’ lymphocytes in lymph nodes.” Dr. Hanauer said.
“Other mechanisms by which lymphocyte trafficking has been inhibited have been effective in treating multiple sclerosis and Crohn’s disease (natalizumab) and both ulcerative colitis and Crohn’s disease (vedolizumab),” he said. However, he said, natalizumab has been linked to a risk of progressive multifocal leukoencephalopathy, a rare and mostly fatal brain disease.
Vedolizumab, meanwhile, “is a biologic that requires IV infusions.” By contrast, he says, ozanimod is an oral drug.
In a recent issue of the New England Journal of Medicine, researchers published findings from a phase II randomized, double-blind, placebo-controlled trial of ozanimod in 197 patients with moderate to severe ulcerative colitis. Patients were assigned to high dose (n = 67), low dose (n = 65), or placebo (n = 65) (N Engl J Med. 2016 May;374:1754-62).
At 8 weeks, clinical remission (Mayo Clinic score less than or equal to 2, with no subscore over 1) occurred in 16% of patients who received 1-mg doses (P = .048) and 14% of those who received the 0.5-mg doses (P = .14); the remission rate was 6% for placebo.
The new analysis examined histologic improvement from baseline to 8 and 32 weeks. (Those who reached clinical response continued through 32 weeks.) Improvement was greater in the high-dose group than in the placebo group at week 8 (Geboes score [–4.37 vs. –2.20; P = .0345]) and week 32 (Geboes score [–5.50 vs. –2.24; P = .0033]); low-dose improvement was greater than placebo but didn’t reach statistical significance.
Histologic remission (Geboes score less than 2) occurred in 15/67 (22.4%) for high dose (P = .0705, compared with placebo), 9/65 (13.8%) for low dose (P = .6294, compared with placebo) and 7/65 (10.8%) for placebo at week 8. At week 32, remission was 21/67 (31.3%) for high dose (P = 0.0006, compared with placebo), 15/65 (23.1%) for low dose (P = .0164, compared with placebo) and 5/65 (7.7%) for placebo.
Adverse events were “minor, without significant cardiotoxicity or risk of infections,” Dr. Hanauer said. The events affected 26/67 (38.8%) patients on the high dose, 26/65 (40.0%) on the low dose, and 26/65 (40.0%) on placebo; worsening of ulcerative colitis and anemia were most common, especially in the placebo group.
The cost of the drug is unclear, Dr. Hanauer said.
The patients are now in open-label follow-up, he said.
The study is industry funded by Receptos. Dr. Hanauer is on the ozanimod steering committee and consults for Celgene and its subsidiary research division, Receptos.
SAN DIEGO – A new analysis of phase II research finds that the experimental drug ozanimod prompted histologic healing in ulcerative colitis patients at 8 and 32 weeks.
“Ozanimod both induces and maintains remissions in a proportion of patients with refractory ulcerative colitis,” said study coauthor Dr. Stephen B. Hanauer, professor of medicine and medical director of the Digestive Health Center at Northwestern University, Chicago. “It’s a safe and effective oral drug for ulcerative colitis and possibly Crohn’s disease.”
Ozanimod, an experimental drug developed by Celgene and its subsidiary Receptos, has been undergoing review as a treatment for both multiple sclerosis and ulcerative colitis.
“Ozanimod impacts lymphocyte trafficking by retaining lymphocytes in lymph nodes and prevents recirculation. The mechanism of action is impairing S1P [sphingosine-1-phosphate] that ‘traps’ lymphocytes in lymph nodes.” Dr. Hanauer said.
“Other mechanisms by which lymphocyte trafficking has been inhibited have been effective in treating multiple sclerosis and Crohn’s disease (natalizumab) and both ulcerative colitis and Crohn’s disease (vedolizumab),” he said. However, he said, natalizumab has been linked to a risk of progressive multifocal leukoencephalopathy, a rare and mostly fatal brain disease.
Vedolizumab, meanwhile, “is a biologic that requires IV infusions.” By contrast, he says, ozanimod is an oral drug.
In a recent issue of the New England Journal of Medicine, researchers published findings from a phase II randomized, double-blind, placebo-controlled trial of ozanimod in 197 patients with moderate to severe ulcerative colitis. Patients were assigned to high dose (n = 67), low dose (n = 65), or placebo (n = 65) (N Engl J Med. 2016 May;374:1754-62).
At 8 weeks, clinical remission (Mayo Clinic score less than or equal to 2, with no subscore over 1) occurred in 16% of patients who received 1-mg doses (P = .048) and 14% of those who received the 0.5-mg doses (P = .14); the remission rate was 6% for placebo.
The new analysis examined histologic improvement from baseline to 8 and 32 weeks. (Those who reached clinical response continued through 32 weeks.) Improvement was greater in the high-dose group than in the placebo group at week 8 (Geboes score [–4.37 vs. –2.20; P = .0345]) and week 32 (Geboes score [–5.50 vs. –2.24; P = .0033]); low-dose improvement was greater than placebo but didn’t reach statistical significance.
Histologic remission (Geboes score less than 2) occurred in 15/67 (22.4%) for high dose (P = .0705, compared with placebo), 9/65 (13.8%) for low dose (P = .6294, compared with placebo) and 7/65 (10.8%) for placebo at week 8. At week 32, remission was 21/67 (31.3%) for high dose (P = 0.0006, compared with placebo), 15/65 (23.1%) for low dose (P = .0164, compared with placebo) and 5/65 (7.7%) for placebo.
Adverse events were “minor, without significant cardiotoxicity or risk of infections,” Dr. Hanauer said. The events affected 26/67 (38.8%) patients on the high dose, 26/65 (40.0%) on the low dose, and 26/65 (40.0%) on placebo; worsening of ulcerative colitis and anemia were most common, especially in the placebo group.
The cost of the drug is unclear, Dr. Hanauer said.
The patients are now in open-label follow-up, he said.
The study is industry funded by Receptos. Dr. Hanauer is on the ozanimod steering committee and consults for Celgene and its subsidiary research division, Receptos.
SAN DIEGO – A new analysis of phase II research finds that the experimental drug ozanimod prompted histologic healing in ulcerative colitis patients at 8 and 32 weeks.
“Ozanimod both induces and maintains remissions in a proportion of patients with refractory ulcerative colitis,” said study coauthor Dr. Stephen B. Hanauer, professor of medicine and medical director of the Digestive Health Center at Northwestern University, Chicago. “It’s a safe and effective oral drug for ulcerative colitis and possibly Crohn’s disease.”
Ozanimod, an experimental drug developed by Celgene and its subsidiary Receptos, has been undergoing review as a treatment for both multiple sclerosis and ulcerative colitis.
“Ozanimod impacts lymphocyte trafficking by retaining lymphocytes in lymph nodes and prevents recirculation. The mechanism of action is impairing S1P [sphingosine-1-phosphate] that ‘traps’ lymphocytes in lymph nodes.” Dr. Hanauer said.
“Other mechanisms by which lymphocyte trafficking has been inhibited have been effective in treating multiple sclerosis and Crohn’s disease (natalizumab) and both ulcerative colitis and Crohn’s disease (vedolizumab),” he said. However, he said, natalizumab has been linked to a risk of progressive multifocal leukoencephalopathy, a rare and mostly fatal brain disease.
Vedolizumab, meanwhile, “is a biologic that requires IV infusions.” By contrast, he says, ozanimod is an oral drug.
In a recent issue of the New England Journal of Medicine, researchers published findings from a phase II randomized, double-blind, placebo-controlled trial of ozanimod in 197 patients with moderate to severe ulcerative colitis. Patients were assigned to high dose (n = 67), low dose (n = 65), or placebo (n = 65) (N Engl J Med. 2016 May;374:1754-62).
At 8 weeks, clinical remission (Mayo Clinic score less than or equal to 2, with no subscore over 1) occurred in 16% of patients who received 1-mg doses (P = .048) and 14% of those who received the 0.5-mg doses (P = .14); the remission rate was 6% for placebo.
The new analysis examined histologic improvement from baseline to 8 and 32 weeks. (Those who reached clinical response continued through 32 weeks.) Improvement was greater in the high-dose group than in the placebo group at week 8 (Geboes score [–4.37 vs. –2.20; P = .0345]) and week 32 (Geboes score [–5.50 vs. –2.24; P = .0033]); low-dose improvement was greater than placebo but didn’t reach statistical significance.
Histologic remission (Geboes score less than 2) occurred in 15/67 (22.4%) for high dose (P = .0705, compared with placebo), 9/65 (13.8%) for low dose (P = .6294, compared with placebo) and 7/65 (10.8%) for placebo at week 8. At week 32, remission was 21/67 (31.3%) for high dose (P = 0.0006, compared with placebo), 15/65 (23.1%) for low dose (P = .0164, compared with placebo) and 5/65 (7.7%) for placebo.
Adverse events were “minor, without significant cardiotoxicity or risk of infections,” Dr. Hanauer said. The events affected 26/67 (38.8%) patients on the high dose, 26/65 (40.0%) on the low dose, and 26/65 (40.0%) on placebo; worsening of ulcerative colitis and anemia were most common, especially in the placebo group.
The cost of the drug is unclear, Dr. Hanauer said.
The patients are now in open-label follow-up, he said.
The study is industry funded by Receptos. Dr. Hanauer is on the ozanimod steering committee and consults for Celgene and its subsidiary research division, Receptos.
AT DDW® 2016
Key clinical point: In addition to clinical remission, response, and endoscopic mucosal healing, ozanimod appears to offer benefits on the histologic front.
Major finding: Histologic improvement was greater in patients who took higher dose of ozanimod (1 mg) than placebo at week 8 (Geboes score [–4.37 vs. –2.20; P = .0345]) and 32 (Geboes score [–5.50 vs. –2.24; P = .0033]). The lower dose (0.5 mg) showed improvement, but it was not statistically significant.
Data source: Randomized, double-blind, placebo-controlled phase II trial of 197 patients (high dose, 67; low dose, 65; placebo, 65).
Disclosures: The study was industry funded by Receptos. Dr. Hanauer is on the ozanimod steering committee and consults for Celgene and its subsidiary research division, Receptos.
Weight gain and antidepressants
When could use of antenatal corticosteroids in the late preterm birth period be beneficial?
The use of antenatal corticosteroids for preterm deliveries between 24 and 34 weeks has been standard of care in obstetric practice. But approximately 70% of preterm deliveries in the United States occur after 34 weeks, in the so-called late preterm period (34 weeks 0 days to 36 weeks 6 days). Recently, Gyamfi-Bannerman and colleagues at the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network completed a trial that examined the use of antenatal betamethasone in women at risk for delivery in the late preterm period.
Details of the study
The Antenatal Late Preterm Steroids (ALPS) trial was a randomized, double-blind, placebo-controlled study that included women with a singleton gestation between 34 weeks 0 days and 36 weeks 5 days who had a high probability risk of delivery in the late preterm period. The authors defined “high probability of delivery” as spontaneous labor with cervical change (at least 3-cm dilation or 75% effacement), preterm premature rupture of the membranes, or a planned delivery scheduled in the late preterm period for specific obstetric indications, such as oligohydramnios, preeclampsia, gestational hypertension, and intrauterine growth restriction.
Women were excluded from the study if they had previously received a course of steroids or had multiple gestations, pregestational diabetes, chorioamnionitis, or were expected to deliver in less than 12 hours due to advanced labor, vaginal bleeding, or nonreassuring fetal status.
Study participants were randomly assigned to receive 2 doses (12 mg intramuscularly) of betamethasone 24 hours apart (1,429 participants) or identical-appearing placebo (1,402 participants). Tocolysis was not allowed in the protocol.
Positive outcomes for neonates
The use of corticosteroids was associated with a significant reduction in the primary outcome of need for respiratory support in the first 72 hours of life (14.4% in the placebo group vs 11.6% in the betamethasone group; relative risk [RR], 0.80; 95% confidence interval [CI], 0.66–0.97; P = .02). Steroid use also decreased the incidence of severe respiratory complications, the need for resuscitation at birth, the need for surfactant therapy, the incidence of transient tachypnea of the newborn, and the incidence of bronchopulmonary dysplasia. Neonatal hypoglycemia was more frequent among infants exposed to betamethasone (24% vs 15%; RR, 1.6; 95% CI, 1.37–1.87; P<.001).
New guidelines issued
The ALPS study is the largest randomized trial to evaluate the benefit of antenatal steroids during the late preterm period. The study’s findings certainly will change clinical practice. Based on the study’s large sample size, rigorous design and protocol, and a cohort generalizable to the US population, SMFM has issued new recommendations for practitioners on using antenatal steroids in the late preterm period in women at risk for preterm delivery.
What this EVIDENCE means for practice
In light of the new SMFM recommendations, in my practice, I will adhere to the inclusion criteria used in the ALPS study, and be careful not to apply the same approach used before 34 weeks, when delivery is often delayed intentionally in order to achieve steroid benefit. If considering adoption of this same practice, clinicians should not use tocolytics when administering corticosteroids in the late preterm period. When indicated, such as in women with severe preeclampsia or ruptured membranes, delivery should not be delayed. A patient with high probability of delivery in the late preterm period is eligible for treatment as long as the clinician thinks that she is not going to deliver within 12 hours. On the other hand, clinicians should not overtreat women, and should maintain a high suspicion for delivery in patients with preterm labor (a cervix that is at least 3 cm dilated or 75% effaced).
The ALPS trial did not allow the administration of more than one course of steroids. The eligibility criteria for corticosteroid use in the late preterm period should not be extended to include subpopulations that were not studied in the trial (including patients with multiple gestations, pregestational diabetes, or those who already had received a complete course of steroids).
— Luis Pacheco, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The use of antenatal corticosteroids for preterm deliveries between 24 and 34 weeks has been standard of care in obstetric practice. But approximately 70% of preterm deliveries in the United States occur after 34 weeks, in the so-called late preterm period (34 weeks 0 days to 36 weeks 6 days). Recently, Gyamfi-Bannerman and colleagues at the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network completed a trial that examined the use of antenatal betamethasone in women at risk for delivery in the late preterm period.
Details of the study
The Antenatal Late Preterm Steroids (ALPS) trial was a randomized, double-blind, placebo-controlled study that included women with a singleton gestation between 34 weeks 0 days and 36 weeks 5 days who had a high probability risk of delivery in the late preterm period. The authors defined “high probability of delivery” as spontaneous labor with cervical change (at least 3-cm dilation or 75% effacement), preterm premature rupture of the membranes, or a planned delivery scheduled in the late preterm period for specific obstetric indications, such as oligohydramnios, preeclampsia, gestational hypertension, and intrauterine growth restriction.
Women were excluded from the study if they had previously received a course of steroids or had multiple gestations, pregestational diabetes, chorioamnionitis, or were expected to deliver in less than 12 hours due to advanced labor, vaginal bleeding, or nonreassuring fetal status.
Study participants were randomly assigned to receive 2 doses (12 mg intramuscularly) of betamethasone 24 hours apart (1,429 participants) or identical-appearing placebo (1,402 participants). Tocolysis was not allowed in the protocol.
Positive outcomes for neonates
The use of corticosteroids was associated with a significant reduction in the primary outcome of need for respiratory support in the first 72 hours of life (14.4% in the placebo group vs 11.6% in the betamethasone group; relative risk [RR], 0.80; 95% confidence interval [CI], 0.66–0.97; P = .02). Steroid use also decreased the incidence of severe respiratory complications, the need for resuscitation at birth, the need for surfactant therapy, the incidence of transient tachypnea of the newborn, and the incidence of bronchopulmonary dysplasia. Neonatal hypoglycemia was more frequent among infants exposed to betamethasone (24% vs 15%; RR, 1.6; 95% CI, 1.37–1.87; P<.001).
New guidelines issued
The ALPS study is the largest randomized trial to evaluate the benefit of antenatal steroids during the late preterm period. The study’s findings certainly will change clinical practice. Based on the study’s large sample size, rigorous design and protocol, and a cohort generalizable to the US population, SMFM has issued new recommendations for practitioners on using antenatal steroids in the late preterm period in women at risk for preterm delivery.
What this EVIDENCE means for practice
In light of the new SMFM recommendations, in my practice, I will adhere to the inclusion criteria used in the ALPS study, and be careful not to apply the same approach used before 34 weeks, when delivery is often delayed intentionally in order to achieve steroid benefit. If considering adoption of this same practice, clinicians should not use tocolytics when administering corticosteroids in the late preterm period. When indicated, such as in women with severe preeclampsia or ruptured membranes, delivery should not be delayed. A patient with high probability of delivery in the late preterm period is eligible for treatment as long as the clinician thinks that she is not going to deliver within 12 hours. On the other hand, clinicians should not overtreat women, and should maintain a high suspicion for delivery in patients with preterm labor (a cervix that is at least 3 cm dilated or 75% effaced).
The ALPS trial did not allow the administration of more than one course of steroids. The eligibility criteria for corticosteroid use in the late preterm period should not be extended to include subpopulations that were not studied in the trial (including patients with multiple gestations, pregestational diabetes, or those who already had received a complete course of steroids).
— Luis Pacheco, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
The use of antenatal corticosteroids for preterm deliveries between 24 and 34 weeks has been standard of care in obstetric practice. But approximately 70% of preterm deliveries in the United States occur after 34 weeks, in the so-called late preterm period (34 weeks 0 days to 36 weeks 6 days). Recently, Gyamfi-Bannerman and colleagues at the Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units Network completed a trial that examined the use of antenatal betamethasone in women at risk for delivery in the late preterm period.
Details of the study
The Antenatal Late Preterm Steroids (ALPS) trial was a randomized, double-blind, placebo-controlled study that included women with a singleton gestation between 34 weeks 0 days and 36 weeks 5 days who had a high probability risk of delivery in the late preterm period. The authors defined “high probability of delivery” as spontaneous labor with cervical change (at least 3-cm dilation or 75% effacement), preterm premature rupture of the membranes, or a planned delivery scheduled in the late preterm period for specific obstetric indications, such as oligohydramnios, preeclampsia, gestational hypertension, and intrauterine growth restriction.
Women were excluded from the study if they had previously received a course of steroids or had multiple gestations, pregestational diabetes, chorioamnionitis, or were expected to deliver in less than 12 hours due to advanced labor, vaginal bleeding, or nonreassuring fetal status.
Study participants were randomly assigned to receive 2 doses (12 mg intramuscularly) of betamethasone 24 hours apart (1,429 participants) or identical-appearing placebo (1,402 participants). Tocolysis was not allowed in the protocol.
Positive outcomes for neonates
The use of corticosteroids was associated with a significant reduction in the primary outcome of need for respiratory support in the first 72 hours of life (14.4% in the placebo group vs 11.6% in the betamethasone group; relative risk [RR], 0.80; 95% confidence interval [CI], 0.66–0.97; P = .02). Steroid use also decreased the incidence of severe respiratory complications, the need for resuscitation at birth, the need for surfactant therapy, the incidence of transient tachypnea of the newborn, and the incidence of bronchopulmonary dysplasia. Neonatal hypoglycemia was more frequent among infants exposed to betamethasone (24% vs 15%; RR, 1.6; 95% CI, 1.37–1.87; P<.001).
New guidelines issued
The ALPS study is the largest randomized trial to evaluate the benefit of antenatal steroids during the late preterm period. The study’s findings certainly will change clinical practice. Based on the study’s large sample size, rigorous design and protocol, and a cohort generalizable to the US population, SMFM has issued new recommendations for practitioners on using antenatal steroids in the late preterm period in women at risk for preterm delivery.
What this EVIDENCE means for practice
In light of the new SMFM recommendations, in my practice, I will adhere to the inclusion criteria used in the ALPS study, and be careful not to apply the same approach used before 34 weeks, when delivery is often delayed intentionally in order to achieve steroid benefit. If considering adoption of this same practice, clinicians should not use tocolytics when administering corticosteroids in the late preterm period. When indicated, such as in women with severe preeclampsia or ruptured membranes, delivery should not be delayed. A patient with high probability of delivery in the late preterm period is eligible for treatment as long as the clinician thinks that she is not going to deliver within 12 hours. On the other hand, clinicians should not overtreat women, and should maintain a high suspicion for delivery in patients with preterm labor (a cervix that is at least 3 cm dilated or 75% effaced).
The ALPS trial did not allow the administration of more than one course of steroids. The eligibility criteria for corticosteroid use in the late preterm period should not be extended to include subpopulations that were not studied in the trial (including patients with multiple gestations, pregestational diabetes, or those who already had received a complete course of steroids).
— Luis Pacheco, MD
Share your thoughts on this article! Send your Letter to the Editor to [email protected]. Please include your name and the city and state in which you practice.
Liraglutide prevents ketogenesis in type 1 diabetes
ORLANDO – A single injection of liraglutide can prevent ketogenesis in fasting patients with type 1 diabetes who were on basal insulin, findings from a small study have shown.
Husam Ghanim, Ph.D., research associate professor at the State University of New York at Buffalo, presented the results in a late-breaking oral presentation session at the annual meeting of the American Association of Clinical Endocrinologists.
In a previous trial (Diabetes Care. 2016;39:1027-35) of patients with type 1 diabetes who took liraglutide, which does not have Food and Drug Administration approval for use in type 1 diabetes, for 12 weeks, investigators observed decreases in blood glucose levels compared with placebo and decreases in glucagon concentrations following a meal compared with before starting liraglutide. When patients already taking liraglutide and insulin were put on dapagliflozin for 12 weeks, glucagon levels rose more with dapagliflozin compared to placebo, and urinary acetoacetate and beta-hydroxybutyrate (adjusted to creatinine) rose over baseline levels.
Some researchers have hypothesized that liraglutide might stimulate residual beta cells (or beta cell stem cells) in patients with type 1 diabetes to produce insulin, thereby reducing the need for exogenous insulin. Promising data from animal studies suggesting that the drug stimulated residual beta cells were not duplicated in human studies. But some evidence shows it may reduce insulin doses anyway, even in cases of patients with no C-peptide, which means they are not producing any insulin on their own (Diabetes Care 2011. 34:1463-8).
In their study, Dr. Ghanim and his associates therefore wanted to test the effect on glucagon, free fatty acid, and ketone levels of acute administration of liraglutide to patients with type 1 diabetes in an insulinopenic condition. They randomly assigned patients with type 1 diabetes, aged 18-75 years, with undetectable C-peptide and hemoglobin A1c less than 8.5%, to receive an injection of 1.8 mg of liraglutide (n = 8) or placebo (n = 8) the morning after an overnight fast, which continued for the 5 hours of the study.
Patients had their basal insulin dose from the night before but no further insulin unless they were on an infusion pump, which they continued. Subjects were excluded if they were taking a glucagon-like peptide-1 (GLP-1) receptor agonist or a sodium/glucose cotransporter-2 (SGLT2) inhibitor, if they had renal impairment, had type 1 diabetes for less than 1 year, or had various other comorbidities.
The liraglutide group was slightly older than the placebo group (46 vs. 43 years), had a higher HbA1c (7.7% vs. 7.6%), and higher systolic but lower diastolic blood pressure (130/73 vs. 121/78 mm Hg). Body mass index was around 30 kg/m2 for both groups.
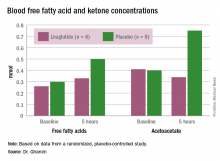
In the placebo group, there was no change in the blood glucose concentrations during the study period, whereas the liraglutide group showed a decrease from a baseline of 175 mg/dL to 135 mg/dL at 5 hours (P less than .05). Glucagon levels were maintained in the placebo group but showed significant suppression from 82 ng/L to 65 ng/L in the liraglutide arm (P less than .05).
“Free fatty acid increased in both groups, but the increase in the placebo arm was significantly higher than that in the liraglutide group,” Dr. Ghanim said. Ketones increased in the placebo group but actually dropped in the liraglutide arm. Ghrelin levels rose by 20% in the placebo group and fell by 10% with liraglutide. Hormone-sensitive lipase decreased about 10% in both arms over the study period.
Dr. Ghanim proposed that since ghrelin is a mediator of lipolysis, possibly the suppression of ghrelin, as well as glucagon, by liraglutide “could contribute to the lower free fatty acid levels, which therefore leads to a lower ketogenic process and reduced ketone bodies.
“With the significant risk of DKA [diabetic ketoacidosis] in type 1 diabetics, especially when you have a drug like an SGLT2 inhibitor, which has been shown to be ketogenic, it is very important to know that liraglutide actually attenuates that response and reduces ketogenesis and therefore reduces the risk of DKA,” he said.
He suggested that these study results should lead to larger randomized trials of GLP-1 receptor agonists and SGLT2 inhibitors, also not approved for use in type 1 diabetes, for use in this population because most of them are not presently well controlled and need additional agents.
Dr. John Miles, professor of both medicine and endocrinology, diabetes, and metabolism at the University of Kansas Medical Center in Kansas City, Kansas, asked Dr. Ghanim why the study subjects did not vomit when receiving the dose of liraglutide. Dr. Ghanim responded that the subjects were not naive to it and had been on it previously.
Session moderator Dr. David Lieb, associate professor of medicine at Eastern Virginia Medical School in Norfolk, said that liraglutide may be a good option for type 1 diabetes patients who are obese and want to lose weight. “I think if there is a drug that can potentially help with glucose control, because liraglutide is not all about causing insulin secretion by the pancreas – it also affects glucagon levels, and it affects appetite and satiety – [so] it may also help with weight loss. I think there’s a role for those sorts of medications in type 1 diabetics on a case-by-case, individual basis,” he said.
However, he wondered if there are any negative effects of suppressing glucagon because patients with type 1 diabetes may be at increased risk for hypoglycemia because of their insulin use, their activities, and their sensitivity to insulin. “Glucagon … allows glucose to be released by the liver,” he said, so (hypothetically) suppressing glucose release may exacerbate hypoglycemia. He said he looks forward to further studies of these drugs for type 1 diabetes and seeing the rate of occurrence of hypoglycemic episodes and how patients respond to them.
There was no funding for the study. Dr. Ghanim and Dr. Lieb reported having no financial disclosures.
ORLANDO – A single injection of liraglutide can prevent ketogenesis in fasting patients with type 1 diabetes who were on basal insulin, findings from a small study have shown.
Husam Ghanim, Ph.D., research associate professor at the State University of New York at Buffalo, presented the results in a late-breaking oral presentation session at the annual meeting of the American Association of Clinical Endocrinologists.
In a previous trial (Diabetes Care. 2016;39:1027-35) of patients with type 1 diabetes who took liraglutide, which does not have Food and Drug Administration approval for use in type 1 diabetes, for 12 weeks, investigators observed decreases in blood glucose levels compared with placebo and decreases in glucagon concentrations following a meal compared with before starting liraglutide. When patients already taking liraglutide and insulin were put on dapagliflozin for 12 weeks, glucagon levels rose more with dapagliflozin compared to placebo, and urinary acetoacetate and beta-hydroxybutyrate (adjusted to creatinine) rose over baseline levels.
Some researchers have hypothesized that liraglutide might stimulate residual beta cells (or beta cell stem cells) in patients with type 1 diabetes to produce insulin, thereby reducing the need for exogenous insulin. Promising data from animal studies suggesting that the drug stimulated residual beta cells were not duplicated in human studies. But some evidence shows it may reduce insulin doses anyway, even in cases of patients with no C-peptide, which means they are not producing any insulin on their own (Diabetes Care 2011. 34:1463-8).
In their study, Dr. Ghanim and his associates therefore wanted to test the effect on glucagon, free fatty acid, and ketone levels of acute administration of liraglutide to patients with type 1 diabetes in an insulinopenic condition. They randomly assigned patients with type 1 diabetes, aged 18-75 years, with undetectable C-peptide and hemoglobin A1c less than 8.5%, to receive an injection of 1.8 mg of liraglutide (n = 8) or placebo (n = 8) the morning after an overnight fast, which continued for the 5 hours of the study.
Patients had their basal insulin dose from the night before but no further insulin unless they were on an infusion pump, which they continued. Subjects were excluded if they were taking a glucagon-like peptide-1 (GLP-1) receptor agonist or a sodium/glucose cotransporter-2 (SGLT2) inhibitor, if they had renal impairment, had type 1 diabetes for less than 1 year, or had various other comorbidities.
The liraglutide group was slightly older than the placebo group (46 vs. 43 years), had a higher HbA1c (7.7% vs. 7.6%), and higher systolic but lower diastolic blood pressure (130/73 vs. 121/78 mm Hg). Body mass index was around 30 kg/m2 for both groups.
In the placebo group, there was no change in the blood glucose concentrations during the study period, whereas the liraglutide group showed a decrease from a baseline of 175 mg/dL to 135 mg/dL at 5 hours (P less than .05). Glucagon levels were maintained in the placebo group but showed significant suppression from 82 ng/L to 65 ng/L in the liraglutide arm (P less than .05).
“Free fatty acid increased in both groups, but the increase in the placebo arm was significantly higher than that in the liraglutide group,” Dr. Ghanim said. Ketones increased in the placebo group but actually dropped in the liraglutide arm. Ghrelin levels rose by 20% in the placebo group and fell by 10% with liraglutide. Hormone-sensitive lipase decreased about 10% in both arms over the study period.
Dr. Ghanim proposed that since ghrelin is a mediator of lipolysis, possibly the suppression of ghrelin, as well as glucagon, by liraglutide “could contribute to the lower free fatty acid levels, which therefore leads to a lower ketogenic process and reduced ketone bodies.
“With the significant risk of DKA [diabetic ketoacidosis] in type 1 diabetics, especially when you have a drug like an SGLT2 inhibitor, which has been shown to be ketogenic, it is very important to know that liraglutide actually attenuates that response and reduces ketogenesis and therefore reduces the risk of DKA,” he said.
He suggested that these study results should lead to larger randomized trials of GLP-1 receptor agonists and SGLT2 inhibitors, also not approved for use in type 1 diabetes, for use in this population because most of them are not presently well controlled and need additional agents.
Dr. John Miles, professor of both medicine and endocrinology, diabetes, and metabolism at the University of Kansas Medical Center in Kansas City, Kansas, asked Dr. Ghanim why the study subjects did not vomit when receiving the dose of liraglutide. Dr. Ghanim responded that the subjects were not naive to it and had been on it previously.
Session moderator Dr. David Lieb, associate professor of medicine at Eastern Virginia Medical School in Norfolk, said that liraglutide may be a good option for type 1 diabetes patients who are obese and want to lose weight. “I think if there is a drug that can potentially help with glucose control, because liraglutide is not all about causing insulin secretion by the pancreas – it also affects glucagon levels, and it affects appetite and satiety – [so] it may also help with weight loss. I think there’s a role for those sorts of medications in type 1 diabetics on a case-by-case, individual basis,” he said.
However, he wondered if there are any negative effects of suppressing glucagon because patients with type 1 diabetes may be at increased risk for hypoglycemia because of their insulin use, their activities, and their sensitivity to insulin. “Glucagon … allows glucose to be released by the liver,” he said, so (hypothetically) suppressing glucose release may exacerbate hypoglycemia. He said he looks forward to further studies of these drugs for type 1 diabetes and seeing the rate of occurrence of hypoglycemic episodes and how patients respond to them.
There was no funding for the study. Dr. Ghanim and Dr. Lieb reported having no financial disclosures.
ORLANDO – A single injection of liraglutide can prevent ketogenesis in fasting patients with type 1 diabetes who were on basal insulin, findings from a small study have shown.
Husam Ghanim, Ph.D., research associate professor at the State University of New York at Buffalo, presented the results in a late-breaking oral presentation session at the annual meeting of the American Association of Clinical Endocrinologists.
In a previous trial (Diabetes Care. 2016;39:1027-35) of patients with type 1 diabetes who took liraglutide, which does not have Food and Drug Administration approval for use in type 1 diabetes, for 12 weeks, investigators observed decreases in blood glucose levels compared with placebo and decreases in glucagon concentrations following a meal compared with before starting liraglutide. When patients already taking liraglutide and insulin were put on dapagliflozin for 12 weeks, glucagon levels rose more with dapagliflozin compared to placebo, and urinary acetoacetate and beta-hydroxybutyrate (adjusted to creatinine) rose over baseline levels.
Some researchers have hypothesized that liraglutide might stimulate residual beta cells (or beta cell stem cells) in patients with type 1 diabetes to produce insulin, thereby reducing the need for exogenous insulin. Promising data from animal studies suggesting that the drug stimulated residual beta cells were not duplicated in human studies. But some evidence shows it may reduce insulin doses anyway, even in cases of patients with no C-peptide, which means they are not producing any insulin on their own (Diabetes Care 2011. 34:1463-8).
In their study, Dr. Ghanim and his associates therefore wanted to test the effect on glucagon, free fatty acid, and ketone levels of acute administration of liraglutide to patients with type 1 diabetes in an insulinopenic condition. They randomly assigned patients with type 1 diabetes, aged 18-75 years, with undetectable C-peptide and hemoglobin A1c less than 8.5%, to receive an injection of 1.8 mg of liraglutide (n = 8) or placebo (n = 8) the morning after an overnight fast, which continued for the 5 hours of the study.
Patients had their basal insulin dose from the night before but no further insulin unless they were on an infusion pump, which they continued. Subjects were excluded if they were taking a glucagon-like peptide-1 (GLP-1) receptor agonist or a sodium/glucose cotransporter-2 (SGLT2) inhibitor, if they had renal impairment, had type 1 diabetes for less than 1 year, or had various other comorbidities.
The liraglutide group was slightly older than the placebo group (46 vs. 43 years), had a higher HbA1c (7.7% vs. 7.6%), and higher systolic but lower diastolic blood pressure (130/73 vs. 121/78 mm Hg). Body mass index was around 30 kg/m2 for both groups.
In the placebo group, there was no change in the blood glucose concentrations during the study period, whereas the liraglutide group showed a decrease from a baseline of 175 mg/dL to 135 mg/dL at 5 hours (P less than .05). Glucagon levels were maintained in the placebo group but showed significant suppression from 82 ng/L to 65 ng/L in the liraglutide arm (P less than .05).
“Free fatty acid increased in both groups, but the increase in the placebo arm was significantly higher than that in the liraglutide group,” Dr. Ghanim said. Ketones increased in the placebo group but actually dropped in the liraglutide arm. Ghrelin levels rose by 20% in the placebo group and fell by 10% with liraglutide. Hormone-sensitive lipase decreased about 10% in both arms over the study period.
Dr. Ghanim proposed that since ghrelin is a mediator of lipolysis, possibly the suppression of ghrelin, as well as glucagon, by liraglutide “could contribute to the lower free fatty acid levels, which therefore leads to a lower ketogenic process and reduced ketone bodies.
“With the significant risk of DKA [diabetic ketoacidosis] in type 1 diabetics, especially when you have a drug like an SGLT2 inhibitor, which has been shown to be ketogenic, it is very important to know that liraglutide actually attenuates that response and reduces ketogenesis and therefore reduces the risk of DKA,” he said.
He suggested that these study results should lead to larger randomized trials of GLP-1 receptor agonists and SGLT2 inhibitors, also not approved for use in type 1 diabetes, for use in this population because most of them are not presently well controlled and need additional agents.
Dr. John Miles, professor of both medicine and endocrinology, diabetes, and metabolism at the University of Kansas Medical Center in Kansas City, Kansas, asked Dr. Ghanim why the study subjects did not vomit when receiving the dose of liraglutide. Dr. Ghanim responded that the subjects were not naive to it and had been on it previously.
Session moderator Dr. David Lieb, associate professor of medicine at Eastern Virginia Medical School in Norfolk, said that liraglutide may be a good option for type 1 diabetes patients who are obese and want to lose weight. “I think if there is a drug that can potentially help with glucose control, because liraglutide is not all about causing insulin secretion by the pancreas – it also affects glucagon levels, and it affects appetite and satiety – [so] it may also help with weight loss. I think there’s a role for those sorts of medications in type 1 diabetics on a case-by-case, individual basis,” he said.
However, he wondered if there are any negative effects of suppressing glucagon because patients with type 1 diabetes may be at increased risk for hypoglycemia because of their insulin use, their activities, and their sensitivity to insulin. “Glucagon … allows glucose to be released by the liver,” he said, so (hypothetically) suppressing glucose release may exacerbate hypoglycemia. He said he looks forward to further studies of these drugs for type 1 diabetes and seeing the rate of occurrence of hypoglycemic episodes and how patients respond to them.
There was no funding for the study. Dr. Ghanim and Dr. Lieb reported having no financial disclosures.
AT AACE 2016
Key clinical point: Liraglutide suppresses glucagon and ketogenesis in fasting patients with type 1 diabetes.
Major finding: FFA increase was 60% lower on liraglutide than on placebo.
Data source: Randomized, placebo controlled study involving 16 patients.
Disclosures: There was no funding for the study. Dr. Ghanim and Dr. Lieb reported having no financial disclosures.
Emergency Imaging: Right hallux pain
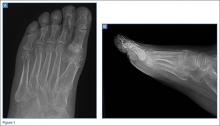
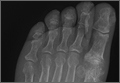
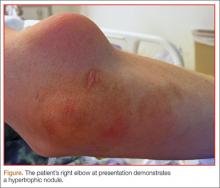
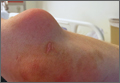
A 55-year-old man presented with a 2-day history of acute first toe pain in his right foot after banging the affected toe on a door. Physical examination demonstrated a swollen first toe with marked tenderness to palpation. Radiographs were obtained (Figures 1a and 1b).
What is the diagnosis? What additional imaging tests may be useful to confirm the diagnosis?
Answer
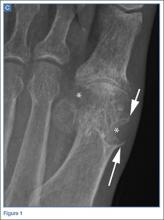
The radiographs of the right foot excluded fracture as the underlying etiology of the patient’s pain. The findings included soft tissue swelling and periarticular (ie, near but not involving the joint) erosions involving the first metatarsal head (white asterisks, Figure 1c). The erosion on the medial aspect of the metatarsal head had remodeling of bone at the periphery of the erosion, which created the appearance of “overhanging edges” (white arrows, Figure 1c). The radiographic appearance suggests the diagnosis of gouty arthritis.
Gouty arthritis, which is caused by the deposition of monosodium urate crystals in the soft tissues surrounding joints, continues to increase in prevalence—likely due to the growing aging population and risk factors such as obesity and diabetes. Gouty arthritis typically presents as painful episodes of arthritis affecting a single joint that can be extremely tender to touch. Acute attacks typically subside within 5 to 7 days. Acute gout may result in fever and elevated white blood cell counts, making it difficult to distinguish from septic arthritis.1 While more common in males in the younger population, gout affects men and women equally in patients older than age 60 years.2
While patients with gouty arthritis have hyperuricemia, only approximately 10% develop gout. The American College of Rheumatology’s preliminary criteria2 for the diagnosis of gout include the presence of characteristic urate crystals in the joint fluid of the affected joint during the attack, the presence of a tophus (soft tissue mass containing urate crystals), or at least six of the following:
- More than one attack of acute arthritis
- Maximum joint inflammation developed within 1 day
- Monoarticular arthritis
- Redness of the joint
- First metatarsophalangeal (MTP) joint pain/swelling
- Unilateral first MTP joint attack
- Unilateral tarsal joint attack
- Suspected tophus
- Hyperuricemia
- Asymmetrical swelling of the joint on radiography
- Subcortical cysts without erosions on radiography
- Joint fluid culture negative during an attack.
As highlighted by the criteria, the first MTP joint is a common location for gouty arthritis, and is referred to as podagra. A meta-analysis published in 2016 reports that an estimated 73% of patients with gout will have involvement of the first MTP.3
Regarding imaging studies, radiography is often the first imaging test performed to evaluate for gout, and can reveal characteristic findings such as periarticular erosions with sclerotic margins, overhanging edges of remodeling bone, and adjacent soft tissue tophi. These findings, however, occur late in the disease. Ultrasound may be useful for earlier diagnosis with the “double contour sign,” which is a specific finding representing the appearance of urate crystals deposited on the hyaline cartilage of the joint. Dual-energy computed tomography (CT) has been shown to not only demonstrate early erosions and soft tissue tophi, but also to characterize the crystals, making CT a highly sensitive and specific test for the detection of gouty arthritis.4
Treatment of acute episodes of gout includes nonsteroidal anti-inflammatory agents, colchicine, and corticosteroids. Early diagnosis and treatment can prevent progression to advanced arthritis and chronic impairment.
1. Eggebeen AT. Gout: an update. Am Fam Physician. 2007;76(6):801-808.
2. Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, Yü TF. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum. 1977;20(3):895-900.
3. Stewart S, Dalbeth N, Vandal AC, Rome K. The first metatarsophalangeal joint in gout: a systematic review and meta-analyis. BMC Musculoskelet Disord. 2016;17(1):69.
4. Girish G, Glazebrook KN, Jacobson JA. Advanced imaging in gout. AJR Am J Roentgenol. 2013;201(3):515-525.
A 55-year-old man presented with a 2-day history of acute first toe pain in his right foot after banging the affected toe on a door. Physical examination demonstrated a swollen first toe with marked tenderness to palpation. Radiographs were obtained (Figures 1a and 1b).
What is the diagnosis? What additional imaging tests may be useful to confirm the diagnosis?
Answer
The radiographs of the right foot excluded fracture as the underlying etiology of the patient’s pain. The findings included soft tissue swelling and periarticular (ie, near but not involving the joint) erosions involving the first metatarsal head (white asterisks, Figure 1c). The erosion on the medial aspect of the metatarsal head had remodeling of bone at the periphery of the erosion, which created the appearance of “overhanging edges” (white arrows, Figure 1c). The radiographic appearance suggests the diagnosis of gouty arthritis.
Gouty arthritis, which is caused by the deposition of monosodium urate crystals in the soft tissues surrounding joints, continues to increase in prevalence—likely due to the growing aging population and risk factors such as obesity and diabetes. Gouty arthritis typically presents as painful episodes of arthritis affecting a single joint that can be extremely tender to touch. Acute attacks typically subside within 5 to 7 days. Acute gout may result in fever and elevated white blood cell counts, making it difficult to distinguish from septic arthritis.1 While more common in males in the younger population, gout affects men and women equally in patients older than age 60 years.2
While patients with gouty arthritis have hyperuricemia, only approximately 10% develop gout. The American College of Rheumatology’s preliminary criteria2 for the diagnosis of gout include the presence of characteristic urate crystals in the joint fluid of the affected joint during the attack, the presence of a tophus (soft tissue mass containing urate crystals), or at least six of the following:
- More than one attack of acute arthritis
- Maximum joint inflammation developed within 1 day
- Monoarticular arthritis
- Redness of the joint
- First metatarsophalangeal (MTP) joint pain/swelling
- Unilateral first MTP joint attack
- Unilateral tarsal joint attack
- Suspected tophus
- Hyperuricemia
- Asymmetrical swelling of the joint on radiography
- Subcortical cysts without erosions on radiography
- Joint fluid culture negative during an attack.
As highlighted by the criteria, the first MTP joint is a common location for gouty arthritis, and is referred to as podagra. A meta-analysis published in 2016 reports that an estimated 73% of patients with gout will have involvement of the first MTP.3
Regarding imaging studies, radiography is often the first imaging test performed to evaluate for gout, and can reveal characteristic findings such as periarticular erosions with sclerotic margins, overhanging edges of remodeling bone, and adjacent soft tissue tophi. These findings, however, occur late in the disease. Ultrasound may be useful for earlier diagnosis with the “double contour sign,” which is a specific finding representing the appearance of urate crystals deposited on the hyaline cartilage of the joint. Dual-energy computed tomography (CT) has been shown to not only demonstrate early erosions and soft tissue tophi, but also to characterize the crystals, making CT a highly sensitive and specific test for the detection of gouty arthritis.4
Treatment of acute episodes of gout includes nonsteroidal anti-inflammatory agents, colchicine, and corticosteroids. Early diagnosis and treatment can prevent progression to advanced arthritis and chronic impairment.
A 55-year-old man presented with a 2-day history of acute first toe pain in his right foot after banging the affected toe on a door. Physical examination demonstrated a swollen first toe with marked tenderness to palpation. Radiographs were obtained (Figures 1a and 1b).
What is the diagnosis? What additional imaging tests may be useful to confirm the diagnosis?
Answer
The radiographs of the right foot excluded fracture as the underlying etiology of the patient’s pain. The findings included soft tissue swelling and periarticular (ie, near but not involving the joint) erosions involving the first metatarsal head (white asterisks, Figure 1c). The erosion on the medial aspect of the metatarsal head had remodeling of bone at the periphery of the erosion, which created the appearance of “overhanging edges” (white arrows, Figure 1c). The radiographic appearance suggests the diagnosis of gouty arthritis.
Gouty arthritis, which is caused by the deposition of monosodium urate crystals in the soft tissues surrounding joints, continues to increase in prevalence—likely due to the growing aging population and risk factors such as obesity and diabetes. Gouty arthritis typically presents as painful episodes of arthritis affecting a single joint that can be extremely tender to touch. Acute attacks typically subside within 5 to 7 days. Acute gout may result in fever and elevated white blood cell counts, making it difficult to distinguish from septic arthritis.1 While more common in males in the younger population, gout affects men and women equally in patients older than age 60 years.2
While patients with gouty arthritis have hyperuricemia, only approximately 10% develop gout. The American College of Rheumatology’s preliminary criteria2 for the diagnosis of gout include the presence of characteristic urate crystals in the joint fluid of the affected joint during the attack, the presence of a tophus (soft tissue mass containing urate crystals), or at least six of the following:
- More than one attack of acute arthritis
- Maximum joint inflammation developed within 1 day
- Monoarticular arthritis
- Redness of the joint
- First metatarsophalangeal (MTP) joint pain/swelling
- Unilateral first MTP joint attack
- Unilateral tarsal joint attack
- Suspected tophus
- Hyperuricemia
- Asymmetrical swelling of the joint on radiography
- Subcortical cysts without erosions on radiography
- Joint fluid culture negative during an attack.
As highlighted by the criteria, the first MTP joint is a common location for gouty arthritis, and is referred to as podagra. A meta-analysis published in 2016 reports that an estimated 73% of patients with gout will have involvement of the first MTP.3
Regarding imaging studies, radiography is often the first imaging test performed to evaluate for gout, and can reveal characteristic findings such as periarticular erosions with sclerotic margins, overhanging edges of remodeling bone, and adjacent soft tissue tophi. These findings, however, occur late in the disease. Ultrasound may be useful for earlier diagnosis with the “double contour sign,” which is a specific finding representing the appearance of urate crystals deposited on the hyaline cartilage of the joint. Dual-energy computed tomography (CT) has been shown to not only demonstrate early erosions and soft tissue tophi, but also to characterize the crystals, making CT a highly sensitive and specific test for the detection of gouty arthritis.4
Treatment of acute episodes of gout includes nonsteroidal anti-inflammatory agents, colchicine, and corticosteroids. Early diagnosis and treatment can prevent progression to advanced arthritis and chronic impairment.
1. Eggebeen AT. Gout: an update. Am Fam Physician. 2007;76(6):801-808.
2. Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, Yü TF. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum. 1977;20(3):895-900.
3. Stewart S, Dalbeth N, Vandal AC, Rome K. The first metatarsophalangeal joint in gout: a systematic review and meta-analyis. BMC Musculoskelet Disord. 2016;17(1):69.
4. Girish G, Glazebrook KN, Jacobson JA. Advanced imaging in gout. AJR Am J Roentgenol. 2013;201(3):515-525.
1. Eggebeen AT. Gout: an update. Am Fam Physician. 2007;76(6):801-808.
2. Wallace SL, Robinson H, Masi AT, Decker JL, McCarty DJ, Yü TF. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum. 1977;20(3):895-900.
3. Stewart S, Dalbeth N, Vandal AC, Rome K. The first metatarsophalangeal joint in gout: a systematic review and meta-analyis. BMC Musculoskelet Disord. 2016;17(1):69.
4. Girish G, Glazebrook KN, Jacobson JA. Advanced imaging in gout. AJR Am J Roentgenol. 2013;201(3):515-525.
Learn More About the Benefits of Belonging
It’s a chance to:
• Meet the man who will be president (of SVS, of course)!
• Learn ways to become involved with SVS, and reap the benefits of that involvement.
• Hear about benefits you weren’t aware of before.
Learn all that and more at Wednesday’s SVS New Member Session, from 4 to 5 p.m. in National Harbor 12/13. The session is intended for physicians who have been members three years or less, as well as those considering becoming members.
Membership Committee Chair Dr. Dawn Coleman is a relatively new member herself. She and committee member Dr. Eric Endean will lead the session, brand new this year.
A chance to meet, mingle and talk informally with SVS leadership will follow, at a reception from 5 to 5:30 p.m.
“Dr. Endean and I want to show our new members the full scope of what SVS can do for them, plus how to get involved in the Society,” Dr. Coleman said. “We want new members to come away with a sense of what the Society has to offer them from a practice perspective and, also, the bigger picture of what such a group can accomplish.”
The session includes:
• Introduction of Society leadership
• A welcome from President-Elect Dr. Ronald M. Fairman
• The SVS branding campaign and how SVS can help new members differentiate themselves as vascular surgeons from other specialties, Dr. George H. Meier, Clinical Practice Council chair
•The SVS Political Action Committee and the importance of a unified voice in advocacy, Dr. Randall R. DeMartino (PAC chair)
• An overview of the Vascular Quality Initiative and Patient Safety Organization, Dr. Larry W. Kraiss, PSO chair
• The SVS Foundation, with a review of basic and clinical research award opportunities, Dr. Peter F. Lawrence, Foundation chair
• Overview of additional membership benefits, Dr. Eric Endean
The New Member Reception will follow at 5 to 5:30 p.m. in National Harbor 7.
The casual setting includes cocktails, wine and small plates and is the perfect chance for the newer members to talk informally with and ask questions of leaders of various branches of the Society.
It’s a chance to:
• Meet the man who will be president (of SVS, of course)!
• Learn ways to become involved with SVS, and reap the benefits of that involvement.
• Hear about benefits you weren’t aware of before.
Learn all that and more at Wednesday’s SVS New Member Session, from 4 to 5 p.m. in National Harbor 12/13. The session is intended for physicians who have been members three years or less, as well as those considering becoming members.
Membership Committee Chair Dr. Dawn Coleman is a relatively new member herself. She and committee member Dr. Eric Endean will lead the session, brand new this year.
A chance to meet, mingle and talk informally with SVS leadership will follow, at a reception from 5 to 5:30 p.m.
“Dr. Endean and I want to show our new members the full scope of what SVS can do for them, plus how to get involved in the Society,” Dr. Coleman said. “We want new members to come away with a sense of what the Society has to offer them from a practice perspective and, also, the bigger picture of what such a group can accomplish.”
The session includes:
• Introduction of Society leadership
• A welcome from President-Elect Dr. Ronald M. Fairman
• The SVS branding campaign and how SVS can help new members differentiate themselves as vascular surgeons from other specialties, Dr. George H. Meier, Clinical Practice Council chair
•The SVS Political Action Committee and the importance of a unified voice in advocacy, Dr. Randall R. DeMartino (PAC chair)
• An overview of the Vascular Quality Initiative and Patient Safety Organization, Dr. Larry W. Kraiss, PSO chair
• The SVS Foundation, with a review of basic and clinical research award opportunities, Dr. Peter F. Lawrence, Foundation chair
• Overview of additional membership benefits, Dr. Eric Endean
The New Member Reception will follow at 5 to 5:30 p.m. in National Harbor 7.
The casual setting includes cocktails, wine and small plates and is the perfect chance for the newer members to talk informally with and ask questions of leaders of various branches of the Society.
It’s a chance to:
• Meet the man who will be president (of SVS, of course)!
• Learn ways to become involved with SVS, and reap the benefits of that involvement.
• Hear about benefits you weren’t aware of before.
Learn all that and more at Wednesday’s SVS New Member Session, from 4 to 5 p.m. in National Harbor 12/13. The session is intended for physicians who have been members three years or less, as well as those considering becoming members.
Membership Committee Chair Dr. Dawn Coleman is a relatively new member herself. She and committee member Dr. Eric Endean will lead the session, brand new this year.
A chance to meet, mingle and talk informally with SVS leadership will follow, at a reception from 5 to 5:30 p.m.
“Dr. Endean and I want to show our new members the full scope of what SVS can do for them, plus how to get involved in the Society,” Dr. Coleman said. “We want new members to come away with a sense of what the Society has to offer them from a practice perspective and, also, the bigger picture of what such a group can accomplish.”
The session includes:
• Introduction of Society leadership
• A welcome from President-Elect Dr. Ronald M. Fairman
• The SVS branding campaign and how SVS can help new members differentiate themselves as vascular surgeons from other specialties, Dr. George H. Meier, Clinical Practice Council chair
•The SVS Political Action Committee and the importance of a unified voice in advocacy, Dr. Randall R. DeMartino (PAC chair)
• An overview of the Vascular Quality Initiative and Patient Safety Organization, Dr. Larry W. Kraiss, PSO chair
• The SVS Foundation, with a review of basic and clinical research award opportunities, Dr. Peter F. Lawrence, Foundation chair
• Overview of additional membership benefits, Dr. Eric Endean
The New Member Reception will follow at 5 to 5:30 p.m. in National Harbor 7.
The casual setting includes cocktails, wine and small plates and is the perfect chance for the newer members to talk informally with and ask questions of leaders of various branches of the Society.
He Huffed and He Puffed and He Got Frostbite
Case
A 27-year-old man presented to an ED after experiencing a syncopal episode. His vital signs at presentation were normal. Physical examination was generally normal except that there were blisters on the patient’s abdomen, left hand, and right arm, as well as a hypertrophic nodule on the right elbow (Figure) and hard growths on the digits of the right hand. The patient stated the growths started 5 months ago and had been increasing in size. On further questioning, the patient admitted to “huffing” (ie, inhaling) at least six cans of pressurized dust-removal keyboard cleaning spray daily for the past 11 months.
Why do patients abuse keyboard cleaning spray?
The propellant used in certain liquefied compressed gas products is 1,1-difluoroethane (1,1-DFE), a fluorinated hydrocarbon. It is a member of a broad class of related compounds that are present in spray paints, glues, nail polish removers, fuels, hair sprays, and air-freshening products. These 1,1-DFE-containing products are abused for their rapid and short-acting central nervous system (CNS) depressant effects—not unlike that of ethanol. Typically, the vapor of a volatile hydrocarbon is inhaled directly from the open container (“sniffing”), from a bag (“bagging”), or from a soaked rag (huffing). Not only are such hydrocarbon-containing products easy to conceal, they are also highly accessible and inexpensive. Moreover, there are generally no direct legal consequences resulting from abuse of these substances.
All of the aforementioned factors make hydrocarbons a popular drug of abuse among adolescents. Approximately 75% of the population abusing hydrocarbons is younger than age 18 years, half of whom reported first use prior to age 13 years.1,2 Though inhalant abuse rarely continues into adulthood, 0.1% of individuals between the ages of 18 and 30 years report having an inhalant-use disorder.
Hydrocarbons and their halogenated derivatives are lipophilic compounds that are rapidly absorbed after inhalation and rapidly distributed to CNS and cardiac tissue. The brain concentration of 1,1-DFE likely peaks higher than concentrations in other organs and is cleared more rapidly.3 Hydrocarbons produce CNS depression secondary to multiple mechanisms, including gamma-aminobutyric acid agonism, dopamine modulation, and N-methyl-D-aspartate-receptor antagonism.4,5
What causes skin lesions on the abdomen and arms?
The lesions on the patient’s abdomen and extremities were consistent with frostbite. The liquefied compressed gas in computer-cleaning and related products is housed in a pressurized canister. The pressure is released when the spray nozzle is depressed; this causes the liquid to rapidly expand to a gas as it is released, resulting in a quick decrease in the temperature of the metal canister. This process, referred to as adiabatic cooling, demonstrates the first law of thermodynamics. The cold temperature of both the liquid and the canister can cause frostbite in the digits and other parts of the body with which the canister or liquid comes into contact.6
Why did the patient have syncope?
Halogenated hydrocarbons inhibit the cardiac delayed rectifier potassium channels involved in the repolarization of cardiac myocytes, causing a delay in repolarization that is manifested as prolongation of the QT interval on an electrocardiogram. This condition places patients at an increased risk of developing torsades de pointes (TdP).7 In most cases, TdP is self-terminating; however, if TdP persists, degeneration to ventricular fibrillation will result. Deaths caused in this fashion have been referred to as “sudden sniffing death syndrome,” and account for half of all hydrocarbon-related deaths.6,8 In addition to the cardiac effects, hydrocarbons are simple asphyxiants that act by displacing oxygen from inspired air, which also contributes to syncope.
It is important to note that epinephrine and other catecholamines increase the risk for dysrhythmias such as TdP in the setting of hydrocarbon abuse.9 For this reason, epinephrine should be used with caution in the setting of a hydrocarbon-induced arrhythmia. Beta-adrenergic antagonists such as esmolol and propranolol are preferable because they reduce the incidence of ectopia that may trigger TdP.10
What is the significance of the masses noted on the examination and radiograph?
Fluorosis is associated with abnormalities of skeletal and dental tissue. Skeletal fluorosis causes osteosclerosis of the axial skeleton, periosteal new bone formation, ligamentous and tendinous ossification, and osteophyte formation. Dental fluorosis causes a yellow/brown discoloration of the teeth with horizontal streaking (mottling), pitting, and chipping.11 Fluorosis is well-described in regions where water fluoride concentrations are high due to industrial exposure; from consumption of fluorinated wine or chronic overconsumption of tea (especially green or black tea); or from fluoridated toothpaste.12-14 More recently, fluorosis has been described in patients treated for an extended duration of time with voriconazole, a fluorinated antifungal agent.15 Unlike other hydrocarbon products, fluorinated hydrocarbons such as 1,1-DFE can significantly increase systemic fluoride concentrations with excessive use. Rapid skeletal fluorosis is not well described, but has been reported after chronic abuse of fluorinated hydrocarbons.16
How is fluorosis diagnosed and managed?
The lack of rapid laboratory testing available for serum, urine, and bone fluoride concentrations makes the initial diagnosis of fluorosis a clinical one. Imaging studies are generally highly suggestive of fluorosis and can be used to support the diagnosis. A dual energy X-ray absorptiometry scan of the spine, hip, femur, and distal portions of the radii can reveal elevated T-scores consistent with osteosclerosis.14 These findings, in conjunction with bone or joint pain, reduced range of motion, or kyphosis, should prompt clinicians to conduct further testing—even without a confirmed fluoride source. A serum fluoride (reference range, 0.2-3.2 mg/L) and 24-hour urine fluoride (reference range, 0.2-3.2 mg/dL) and creatinine evaluation can be used to diagnose fluorosis. However, a bone biopsy with quantitative bone ash fluoride analysis remains the gold standard for the diagnosis of skeletal fluorosis.16 Laboratory evaluation should also include an assessment of electrolytes, specifically calcium, 25-hydroxyvitamin D, and alkaline phosphatase. The differential diagnosis should include hemoglobinopathies, renal osteodystrophy, Paget disease, hypothyroidism, and skeletal metastases.16
Treatment of fluorosis is largely symptomatic and supportive, with identification and discontinuation of the fluoride source. Patients should be referred to an orthopedist for evaluation and management as needed. Evaluation by an endocrinologist should also be considered because patients may have chronic vitamin D and calcium deficiencies as a result of systemic fluorosis.
Case Conclusion
The patient’s laboratory assessment was notable for the following: alkaline phosphatase, 624 U/L (reference range, 44-147 IU/L); vitamin D, 10 ng/mL (reference range, 20-40 ng/mL); serum fluoride, 0.3 mg/L (reference range, 0.2-3.2 mg/L); urine fluoride, 52 mg/dL (0.2-3.2 mg/dL); and urine creatinine, 1 g/L (reference range, 0.3-3 g/L). Imaging studies noted periosteal bone formation on the lateral epicondyle of the distal right humerus, as well as similar osseous abnormalities in other locations. A bone biopsy was scheduled. The patient was treated with oral vitamin D and educated about the importance of discontinuing the huffing of all hydrocarbons.
1. Williams JF, Storck M; American Academy of Pediatrics Committee on Substance Abuse; American Academy of Pediatrics Committee on Native American Child Health. Inhalant abuse. Pediatrics. 2007;119(5):1009-1017.
2. Wu LT, Pilowsky DJ, Schlenger WE. Inhalant abuse and dependence among adolescents in the United States. J Am Acad Child Adolesc Psychiatry. 2004;43(10):1206-1214.
3. Avella J, Kunaparaju N, Kumar S, Lehrer M, Zito SW, Barletta M. Uptake and distribution of the abused inhalant 1,1-difluoroethane in the rat. J Anal Toxicol. 2010;34(7):381-388.
4. Tormoehlen LM, Tekulve KJ, Nañagas KA. Hydrocarbon toxicity: A review. Clin Toxicol (Phila). 2014;52(5):479-489.
5. Duncan JR, Lawrence AJ. Conventional concepts and new perspectives for understanding the addictive properties of inhalants. J Pharmacol Sci. 2013;122(4):237-243.
6. Sakai K, Maruyama-Maebashi K, Takatsu A, et al. Sudden death involving inhalation of 1,1-difluoroethane (HFC-152a) with spray cleaner: three case reports. Forensic Sci Int. 2011;206(1-3):e58-e61.
7. Himmel HM. Mechanisms involved in cardiac sensitization by volatile anesthetics: general applicability to halogenated hydrocarbons? Crit Rev Toxicol. 2008;38(9):773-803.
8. Avella J, Wilson JC, Lehrer M. Fatal cardiac arrhythmia after repeated exposure to 1,1-difluoroethane (DFE). Am J Forensic Med Pathol. 2006;27(1):58-60.
9. Nelson LS. Toxicologic myocardial sensitization. J Toxicol Clin Toxicol. 2002;40(7):867-879.
10. Mortiz F, de La Chapelle A, Bauer F, Leroy JP, Goullé JP, Bonmarchand G. Esmolol in the treatment of severe arrhythmia after acute trichloroethylene poisoning. Intensive Care Med. 2000;26(2):256.
11. Majumdar KK. Health impact of supplying safe drinking water containing fluoride below permissible level on flourosis patients in a fluoride-endemic rural area of West Bengal. Indian J Public Health. 2011;55(4):303-308.
12. Kakumanu N, Rao SD. Images in clinical medicine. Skeletal fluorosis due to excessive tea drinking. N Engl J Med 2013;368(12):1140.
13. Soriano M, Manchón F. Radiological aspects of a new type of bone fluorosis, periostitis deformans. Radiology 1966;87(6):1089-1094.
14. Tamer MN, Kale Köroğlu B, Arslan C, et al. Osteosclerosis due to endemic fluorosis. Sci Total Environ. 2007;373(1):43-48.
15. Bucknor MD, Gross AJ, Link TM. Voriconazole-induced periostitis in two post-transplant patients. J Radiol Case Rep. 2013;7(8):10-17.
16. Cohen E, Hsu RY, Evangelista P, Aaron R, Rubin LE. Rapid-onset diffuse skeletal fluorosis from inhalant abuse: a case report. JBJS Case Connector. 2014;4(4):e108.
Case
A 27-year-old man presented to an ED after experiencing a syncopal episode. His vital signs at presentation were normal. Physical examination was generally normal except that there were blisters on the patient’s abdomen, left hand, and right arm, as well as a hypertrophic nodule on the right elbow (Figure) and hard growths on the digits of the right hand. The patient stated the growths started 5 months ago and had been increasing in size. On further questioning, the patient admitted to “huffing” (ie, inhaling) at least six cans of pressurized dust-removal keyboard cleaning spray daily for the past 11 months.
Why do patients abuse keyboard cleaning spray?
The propellant used in certain liquefied compressed gas products is 1,1-difluoroethane (1,1-DFE), a fluorinated hydrocarbon. It is a member of a broad class of related compounds that are present in spray paints, glues, nail polish removers, fuels, hair sprays, and air-freshening products. These 1,1-DFE-containing products are abused for their rapid and short-acting central nervous system (CNS) depressant effects—not unlike that of ethanol. Typically, the vapor of a volatile hydrocarbon is inhaled directly from the open container (“sniffing”), from a bag (“bagging”), or from a soaked rag (huffing). Not only are such hydrocarbon-containing products easy to conceal, they are also highly accessible and inexpensive. Moreover, there are generally no direct legal consequences resulting from abuse of these substances.
All of the aforementioned factors make hydrocarbons a popular drug of abuse among adolescents. Approximately 75% of the population abusing hydrocarbons is younger than age 18 years, half of whom reported first use prior to age 13 years.1,2 Though inhalant abuse rarely continues into adulthood, 0.1% of individuals between the ages of 18 and 30 years report having an inhalant-use disorder.
Hydrocarbons and their halogenated derivatives are lipophilic compounds that are rapidly absorbed after inhalation and rapidly distributed to CNS and cardiac tissue. The brain concentration of 1,1-DFE likely peaks higher than concentrations in other organs and is cleared more rapidly.3 Hydrocarbons produce CNS depression secondary to multiple mechanisms, including gamma-aminobutyric acid agonism, dopamine modulation, and N-methyl-D-aspartate-receptor antagonism.4,5
What causes skin lesions on the abdomen and arms?
The lesions on the patient’s abdomen and extremities were consistent with frostbite. The liquefied compressed gas in computer-cleaning and related products is housed in a pressurized canister. The pressure is released when the spray nozzle is depressed; this causes the liquid to rapidly expand to a gas as it is released, resulting in a quick decrease in the temperature of the metal canister. This process, referred to as adiabatic cooling, demonstrates the first law of thermodynamics. The cold temperature of both the liquid and the canister can cause frostbite in the digits and other parts of the body with which the canister or liquid comes into contact.6
Why did the patient have syncope?
Halogenated hydrocarbons inhibit the cardiac delayed rectifier potassium channels involved in the repolarization of cardiac myocytes, causing a delay in repolarization that is manifested as prolongation of the QT interval on an electrocardiogram. This condition places patients at an increased risk of developing torsades de pointes (TdP).7 In most cases, TdP is self-terminating; however, if TdP persists, degeneration to ventricular fibrillation will result. Deaths caused in this fashion have been referred to as “sudden sniffing death syndrome,” and account for half of all hydrocarbon-related deaths.6,8 In addition to the cardiac effects, hydrocarbons are simple asphyxiants that act by displacing oxygen from inspired air, which also contributes to syncope.
It is important to note that epinephrine and other catecholamines increase the risk for dysrhythmias such as TdP in the setting of hydrocarbon abuse.9 For this reason, epinephrine should be used with caution in the setting of a hydrocarbon-induced arrhythmia. Beta-adrenergic antagonists such as esmolol and propranolol are preferable because they reduce the incidence of ectopia that may trigger TdP.10
What is the significance of the masses noted on the examination and radiograph?
Fluorosis is associated with abnormalities of skeletal and dental tissue. Skeletal fluorosis causes osteosclerosis of the axial skeleton, periosteal new bone formation, ligamentous and tendinous ossification, and osteophyte formation. Dental fluorosis causes a yellow/brown discoloration of the teeth with horizontal streaking (mottling), pitting, and chipping.11 Fluorosis is well-described in regions where water fluoride concentrations are high due to industrial exposure; from consumption of fluorinated wine or chronic overconsumption of tea (especially green or black tea); or from fluoridated toothpaste.12-14 More recently, fluorosis has been described in patients treated for an extended duration of time with voriconazole, a fluorinated antifungal agent.15 Unlike other hydrocarbon products, fluorinated hydrocarbons such as 1,1-DFE can significantly increase systemic fluoride concentrations with excessive use. Rapid skeletal fluorosis is not well described, but has been reported after chronic abuse of fluorinated hydrocarbons.16
How is fluorosis diagnosed and managed?
The lack of rapid laboratory testing available for serum, urine, and bone fluoride concentrations makes the initial diagnosis of fluorosis a clinical one. Imaging studies are generally highly suggestive of fluorosis and can be used to support the diagnosis. A dual energy X-ray absorptiometry scan of the spine, hip, femur, and distal portions of the radii can reveal elevated T-scores consistent with osteosclerosis.14 These findings, in conjunction with bone or joint pain, reduced range of motion, or kyphosis, should prompt clinicians to conduct further testing—even without a confirmed fluoride source. A serum fluoride (reference range, 0.2-3.2 mg/L) and 24-hour urine fluoride (reference range, 0.2-3.2 mg/dL) and creatinine evaluation can be used to diagnose fluorosis. However, a bone biopsy with quantitative bone ash fluoride analysis remains the gold standard for the diagnosis of skeletal fluorosis.16 Laboratory evaluation should also include an assessment of electrolytes, specifically calcium, 25-hydroxyvitamin D, and alkaline phosphatase. The differential diagnosis should include hemoglobinopathies, renal osteodystrophy, Paget disease, hypothyroidism, and skeletal metastases.16
Treatment of fluorosis is largely symptomatic and supportive, with identification and discontinuation of the fluoride source. Patients should be referred to an orthopedist for evaluation and management as needed. Evaluation by an endocrinologist should also be considered because patients may have chronic vitamin D and calcium deficiencies as a result of systemic fluorosis.
Case Conclusion
The patient’s laboratory assessment was notable for the following: alkaline phosphatase, 624 U/L (reference range, 44-147 IU/L); vitamin D, 10 ng/mL (reference range, 20-40 ng/mL); serum fluoride, 0.3 mg/L (reference range, 0.2-3.2 mg/L); urine fluoride, 52 mg/dL (0.2-3.2 mg/dL); and urine creatinine, 1 g/L (reference range, 0.3-3 g/L). Imaging studies noted periosteal bone formation on the lateral epicondyle of the distal right humerus, as well as similar osseous abnormalities in other locations. A bone biopsy was scheduled. The patient was treated with oral vitamin D and educated about the importance of discontinuing the huffing of all hydrocarbons.
Case
A 27-year-old man presented to an ED after experiencing a syncopal episode. His vital signs at presentation were normal. Physical examination was generally normal except that there were blisters on the patient’s abdomen, left hand, and right arm, as well as a hypertrophic nodule on the right elbow (Figure) and hard growths on the digits of the right hand. The patient stated the growths started 5 months ago and had been increasing in size. On further questioning, the patient admitted to “huffing” (ie, inhaling) at least six cans of pressurized dust-removal keyboard cleaning spray daily for the past 11 months.
Why do patients abuse keyboard cleaning spray?
The propellant used in certain liquefied compressed gas products is 1,1-difluoroethane (1,1-DFE), a fluorinated hydrocarbon. It is a member of a broad class of related compounds that are present in spray paints, glues, nail polish removers, fuels, hair sprays, and air-freshening products. These 1,1-DFE-containing products are abused for their rapid and short-acting central nervous system (CNS) depressant effects—not unlike that of ethanol. Typically, the vapor of a volatile hydrocarbon is inhaled directly from the open container (“sniffing”), from a bag (“bagging”), or from a soaked rag (huffing). Not only are such hydrocarbon-containing products easy to conceal, they are also highly accessible and inexpensive. Moreover, there are generally no direct legal consequences resulting from abuse of these substances.
All of the aforementioned factors make hydrocarbons a popular drug of abuse among adolescents. Approximately 75% of the population abusing hydrocarbons is younger than age 18 years, half of whom reported first use prior to age 13 years.1,2 Though inhalant abuse rarely continues into adulthood, 0.1% of individuals between the ages of 18 and 30 years report having an inhalant-use disorder.
Hydrocarbons and their halogenated derivatives are lipophilic compounds that are rapidly absorbed after inhalation and rapidly distributed to CNS and cardiac tissue. The brain concentration of 1,1-DFE likely peaks higher than concentrations in other organs and is cleared more rapidly.3 Hydrocarbons produce CNS depression secondary to multiple mechanisms, including gamma-aminobutyric acid agonism, dopamine modulation, and N-methyl-D-aspartate-receptor antagonism.4,5
What causes skin lesions on the abdomen and arms?
The lesions on the patient’s abdomen and extremities were consistent with frostbite. The liquefied compressed gas in computer-cleaning and related products is housed in a pressurized canister. The pressure is released when the spray nozzle is depressed; this causes the liquid to rapidly expand to a gas as it is released, resulting in a quick decrease in the temperature of the metal canister. This process, referred to as adiabatic cooling, demonstrates the first law of thermodynamics. The cold temperature of both the liquid and the canister can cause frostbite in the digits and other parts of the body with which the canister or liquid comes into contact.6
Why did the patient have syncope?
Halogenated hydrocarbons inhibit the cardiac delayed rectifier potassium channels involved in the repolarization of cardiac myocytes, causing a delay in repolarization that is manifested as prolongation of the QT interval on an electrocardiogram. This condition places patients at an increased risk of developing torsades de pointes (TdP).7 In most cases, TdP is self-terminating; however, if TdP persists, degeneration to ventricular fibrillation will result. Deaths caused in this fashion have been referred to as “sudden sniffing death syndrome,” and account for half of all hydrocarbon-related deaths.6,8 In addition to the cardiac effects, hydrocarbons are simple asphyxiants that act by displacing oxygen from inspired air, which also contributes to syncope.
It is important to note that epinephrine and other catecholamines increase the risk for dysrhythmias such as TdP in the setting of hydrocarbon abuse.9 For this reason, epinephrine should be used with caution in the setting of a hydrocarbon-induced arrhythmia. Beta-adrenergic antagonists such as esmolol and propranolol are preferable because they reduce the incidence of ectopia that may trigger TdP.10
What is the significance of the masses noted on the examination and radiograph?
Fluorosis is associated with abnormalities of skeletal and dental tissue. Skeletal fluorosis causes osteosclerosis of the axial skeleton, periosteal new bone formation, ligamentous and tendinous ossification, and osteophyte formation. Dental fluorosis causes a yellow/brown discoloration of the teeth with horizontal streaking (mottling), pitting, and chipping.11 Fluorosis is well-described in regions where water fluoride concentrations are high due to industrial exposure; from consumption of fluorinated wine or chronic overconsumption of tea (especially green or black tea); or from fluoridated toothpaste.12-14 More recently, fluorosis has been described in patients treated for an extended duration of time with voriconazole, a fluorinated antifungal agent.15 Unlike other hydrocarbon products, fluorinated hydrocarbons such as 1,1-DFE can significantly increase systemic fluoride concentrations with excessive use. Rapid skeletal fluorosis is not well described, but has been reported after chronic abuse of fluorinated hydrocarbons.16
How is fluorosis diagnosed and managed?
The lack of rapid laboratory testing available for serum, urine, and bone fluoride concentrations makes the initial diagnosis of fluorosis a clinical one. Imaging studies are generally highly suggestive of fluorosis and can be used to support the diagnosis. A dual energy X-ray absorptiometry scan of the spine, hip, femur, and distal portions of the radii can reveal elevated T-scores consistent with osteosclerosis.14 These findings, in conjunction with bone or joint pain, reduced range of motion, or kyphosis, should prompt clinicians to conduct further testing—even without a confirmed fluoride source. A serum fluoride (reference range, 0.2-3.2 mg/L) and 24-hour urine fluoride (reference range, 0.2-3.2 mg/dL) and creatinine evaluation can be used to diagnose fluorosis. However, a bone biopsy with quantitative bone ash fluoride analysis remains the gold standard for the diagnosis of skeletal fluorosis.16 Laboratory evaluation should also include an assessment of electrolytes, specifically calcium, 25-hydroxyvitamin D, and alkaline phosphatase. The differential diagnosis should include hemoglobinopathies, renal osteodystrophy, Paget disease, hypothyroidism, and skeletal metastases.16
Treatment of fluorosis is largely symptomatic and supportive, with identification and discontinuation of the fluoride source. Patients should be referred to an orthopedist for evaluation and management as needed. Evaluation by an endocrinologist should also be considered because patients may have chronic vitamin D and calcium deficiencies as a result of systemic fluorosis.
Case Conclusion
The patient’s laboratory assessment was notable for the following: alkaline phosphatase, 624 U/L (reference range, 44-147 IU/L); vitamin D, 10 ng/mL (reference range, 20-40 ng/mL); serum fluoride, 0.3 mg/L (reference range, 0.2-3.2 mg/L); urine fluoride, 52 mg/dL (0.2-3.2 mg/dL); and urine creatinine, 1 g/L (reference range, 0.3-3 g/L). Imaging studies noted periosteal bone formation on the lateral epicondyle of the distal right humerus, as well as similar osseous abnormalities in other locations. A bone biopsy was scheduled. The patient was treated with oral vitamin D and educated about the importance of discontinuing the huffing of all hydrocarbons.
1. Williams JF, Storck M; American Academy of Pediatrics Committee on Substance Abuse; American Academy of Pediatrics Committee on Native American Child Health. Inhalant abuse. Pediatrics. 2007;119(5):1009-1017.
2. Wu LT, Pilowsky DJ, Schlenger WE. Inhalant abuse and dependence among adolescents in the United States. J Am Acad Child Adolesc Psychiatry. 2004;43(10):1206-1214.
3. Avella J, Kunaparaju N, Kumar S, Lehrer M, Zito SW, Barletta M. Uptake and distribution of the abused inhalant 1,1-difluoroethane in the rat. J Anal Toxicol. 2010;34(7):381-388.
4. Tormoehlen LM, Tekulve KJ, Nañagas KA. Hydrocarbon toxicity: A review. Clin Toxicol (Phila). 2014;52(5):479-489.
5. Duncan JR, Lawrence AJ. Conventional concepts and new perspectives for understanding the addictive properties of inhalants. J Pharmacol Sci. 2013;122(4):237-243.
6. Sakai K, Maruyama-Maebashi K, Takatsu A, et al. Sudden death involving inhalation of 1,1-difluoroethane (HFC-152a) with spray cleaner: three case reports. Forensic Sci Int. 2011;206(1-3):e58-e61.
7. Himmel HM. Mechanisms involved in cardiac sensitization by volatile anesthetics: general applicability to halogenated hydrocarbons? Crit Rev Toxicol. 2008;38(9):773-803.
8. Avella J, Wilson JC, Lehrer M. Fatal cardiac arrhythmia after repeated exposure to 1,1-difluoroethane (DFE). Am J Forensic Med Pathol. 2006;27(1):58-60.
9. Nelson LS. Toxicologic myocardial sensitization. J Toxicol Clin Toxicol. 2002;40(7):867-879.
10. Mortiz F, de La Chapelle A, Bauer F, Leroy JP, Goullé JP, Bonmarchand G. Esmolol in the treatment of severe arrhythmia after acute trichloroethylene poisoning. Intensive Care Med. 2000;26(2):256.
11. Majumdar KK. Health impact of supplying safe drinking water containing fluoride below permissible level on flourosis patients in a fluoride-endemic rural area of West Bengal. Indian J Public Health. 2011;55(4):303-308.
12. Kakumanu N, Rao SD. Images in clinical medicine. Skeletal fluorosis due to excessive tea drinking. N Engl J Med 2013;368(12):1140.
13. Soriano M, Manchón F. Radiological aspects of a new type of bone fluorosis, periostitis deformans. Radiology 1966;87(6):1089-1094.
14. Tamer MN, Kale Köroğlu B, Arslan C, et al. Osteosclerosis due to endemic fluorosis. Sci Total Environ. 2007;373(1):43-48.
15. Bucknor MD, Gross AJ, Link TM. Voriconazole-induced periostitis in two post-transplant patients. J Radiol Case Rep. 2013;7(8):10-17.
16. Cohen E, Hsu RY, Evangelista P, Aaron R, Rubin LE. Rapid-onset diffuse skeletal fluorosis from inhalant abuse: a case report. JBJS Case Connector. 2014;4(4):e108.
1. Williams JF, Storck M; American Academy of Pediatrics Committee on Substance Abuse; American Academy of Pediatrics Committee on Native American Child Health. Inhalant abuse. Pediatrics. 2007;119(5):1009-1017.
2. Wu LT, Pilowsky DJ, Schlenger WE. Inhalant abuse and dependence among adolescents in the United States. J Am Acad Child Adolesc Psychiatry. 2004;43(10):1206-1214.
3. Avella J, Kunaparaju N, Kumar S, Lehrer M, Zito SW, Barletta M. Uptake and distribution of the abused inhalant 1,1-difluoroethane in the rat. J Anal Toxicol. 2010;34(7):381-388.
4. Tormoehlen LM, Tekulve KJ, Nañagas KA. Hydrocarbon toxicity: A review. Clin Toxicol (Phila). 2014;52(5):479-489.
5. Duncan JR, Lawrence AJ. Conventional concepts and new perspectives for understanding the addictive properties of inhalants. J Pharmacol Sci. 2013;122(4):237-243.
6. Sakai K, Maruyama-Maebashi K, Takatsu A, et al. Sudden death involving inhalation of 1,1-difluoroethane (HFC-152a) with spray cleaner: three case reports. Forensic Sci Int. 2011;206(1-3):e58-e61.
7. Himmel HM. Mechanisms involved in cardiac sensitization by volatile anesthetics: general applicability to halogenated hydrocarbons? Crit Rev Toxicol. 2008;38(9):773-803.
8. Avella J, Wilson JC, Lehrer M. Fatal cardiac arrhythmia after repeated exposure to 1,1-difluoroethane (DFE). Am J Forensic Med Pathol. 2006;27(1):58-60.
9. Nelson LS. Toxicologic myocardial sensitization. J Toxicol Clin Toxicol. 2002;40(7):867-879.
10. Mortiz F, de La Chapelle A, Bauer F, Leroy JP, Goullé JP, Bonmarchand G. Esmolol in the treatment of severe arrhythmia after acute trichloroethylene poisoning. Intensive Care Med. 2000;26(2):256.
11. Majumdar KK. Health impact of supplying safe drinking water containing fluoride below permissible level on flourosis patients in a fluoride-endemic rural area of West Bengal. Indian J Public Health. 2011;55(4):303-308.
12. Kakumanu N, Rao SD. Images in clinical medicine. Skeletal fluorosis due to excessive tea drinking. N Engl J Med 2013;368(12):1140.
13. Soriano M, Manchón F. Radiological aspects of a new type of bone fluorosis, periostitis deformans. Radiology 1966;87(6):1089-1094.
14. Tamer MN, Kale Köroğlu B, Arslan C, et al. Osteosclerosis due to endemic fluorosis. Sci Total Environ. 2007;373(1):43-48.
15. Bucknor MD, Gross AJ, Link TM. Voriconazole-induced periostitis in two post-transplant patients. J Radiol Case Rep. 2013;7(8):10-17.
16. Cohen E, Hsu RY, Evangelista P, Aaron R, Rubin LE. Rapid-onset diffuse skeletal fluorosis from inhalant abuse: a case report. JBJS Case Connector. 2014;4(4):e108.
Medical Mimics of Psychiatric Conditions, Part 2
Although the emergency physician (EP) typically encounters common conditions such as chest pain, urinary tract infection, and gastroenteritis, many other clinical presentations can confound diagnosis of the true underlying condition. This may be the case with a patient who presents with apparent psychiatric symptoms that are actually masking an acute medical condition. For example, a patient who appears to be depressed may actually be exhibiting early signs of dementia. Likewise, a manic patient may not have a true underlying psychiatric disorder but rather rhabdomyolysis and hyperthermia from ingesting an illicit substance such as synthetic cathinones (“bath salts”).
Part 1 of this series reviewed psychiatric presentations caused by underlying infectious, pharmacological withdrawal, metabolic, autoimmune, traumatic, and central nervous system etiologies (Emerg Med. 2016;48[5]:202-211). Part 2 covers psychiatric presentations related to dementia, cancer, cardiac disease, nutritional deficiencies, endocrine disorders, or toxins (Table 1).
Case Scenarios
Case 1
A 62-year-old man with a history of hypertension, hyperlipidemia, and past alcohol abuse presented to the ED with reported mental status changes after he was pulled over by police for driving the wrong way down the highway. On presentation, the patient’s vital signs were normal. When questioned, the patient was alert and fully oriented and believed the officers were mistaken about what was reported. He denied any recent illness and had a normal physical examination, including neurological examination.
A brief work-up was ordered and the patient passed the time by politely flirting with the nurses. When his wife arrived at the ED, she was relieved that her husband seemed to be all right. She confirmed that the patient had not consumed any alcohol in years. The patient, meanwhile, playfully minimized his wife’s concern at his presence in the ED. A full toxicology screen, laboratory evaluation, and head computed tomography (CT) scan were ordered.
Case 2
A 48-year-old woman with a history of anxiety disorder, depression, and diabetes mellitus presented to the ED with a 2-hour history of chest pain. She stated that the pain had started toward the end of a heated argument with her son. The patient was escorted into the examination room by hospital security because she was still agitated and kept yelling at her son. On examination the patient was tachycardic (110 beats/minute), diaphoretic, and crying. During the examination, she asked the EP for a “Xanax”; her son further noted that this would help his mother’s condition.
The patient repeatedly claimed she could not breathe and could not lie flat on the stretcher. After verbal de-escalation, she cooperated with the electrocardiography (ECG) technician and phlebotomist. Her ECG showed nonspecific ST changes with no prior study for comparison. While glaring at her son, she maintained that she had constant chest pain.
Dementia
Alzheimer’s Disease
Alzheimer’s disease (AD), the most common cause of dementia, is a chronic neurodegenerative disease characterized by an insidiously progressive cognitive decline and loss of function. There is considerable apparent variability in the early signs of the disease, and recent literature has suggested that the manifestation of initial symptoms may be age-dependent. Younger patients tend to present with non-memory cognitive changes such as problem-solving difficulties, as well as personality changes and behavioral symptoms of depression, apathy, and withdrawal.1
Lewy Body Dementia
Lewy body dementia (LBD) is a chronic neurodegenerative disease with a presentation that overlaps substantially with AD. However, LBD is associated with a significantly more rapid course than AD and presents more frequently with visual hallucinations or illusions due to specific visuospatial dysfunction.2
Frontotemporal Dementia
Frontotemporal dementia is a comparatively rare chronic neurodegenerative disease characterized by early-onset memory impairment with cognitive decline, as well as behavioral changes such as disinhibition, emotional blunting, and language difficulty. Initial presentations can also include atypical features such as paranoia or delusion, and misdiagnosis as a primary psychiatric problem is common.3
Cancer
Brain Tumor
Primary and metastatic brain tumors classically present with either focal neurological signs or less specific symptoms such as headaches, seizures, or syncope. Additionally, central nervous system (CNS) tumors can also initially present with primary psychiatric complaints (eg, personality changes, depression, mania, panic attacks, auditory or visual hallucinations). Patients with a brain neoplasm who are initially misdiagnosed with a primary psychiatric disorder face significant delays in proper diagnosis and treatment, leading to increased morbidity. To correctly diagnose the true cause as soon as possible, early imaging is recommended for patients who present with psychiatric symptoms that are abrupt in onset, atypical in presentation, resistant to conventional treatments, or associated with a change in headache pattern.4
Paraneoplastic Limbic Encephalitis
Paraneoplastic limbic encephalitis (PLE) is a rare neurological consequence of certain cancers. Although PLE most commonly occurs in patients with small cell lung cancer, the condition has also been reported (though less frequently) in cases of esophageal adenocarcinoma, ovarian teratoma, metastatic breast cancer, and germ cell testicular cancer.5 This disease overlaps substantially with anti-N-methyl-D-aspartate (anti-NMDA) receptor encephalitis. Moreover, PLE can present initially with prominent neuropsychiatric symptoms such as confusion, cognitive problems, behavioral changes, irritability, depression, or frank psychosis with hallucinations. Paraneoplastic limbic encephalitis can occur early in the course of cancer—often before other systemic signs appear—and its significance is often only recognized in retrospect or postmortem. A higher index of suspicion for the disorder may lead to earlier detection of treatable cancers.
Malignant Meningitis
Malignant meningitis is the metastatic spread of a primary solid tumor to the leptomeninges. It can present as a wide variety of neuropsychiatric complaints, including depression, anxiety, disorientation, and paranoia. Diagnosis can often be made through lumbar puncture. Malignant meningitis should be considered in the differential diagnosis of new psychiatric symptoms in a patient with a history of cancer—even in the absence of focal neurological deficits or meningeal signs.6
Pancreatic Insulinoma
Pancreatic insulinoma is a rare, potentially curable endocrine tumor that can present initially with vague psychiatric complaints such as irrational behavior, confusion, depression, or anxiety. In up to 64% of patients, insulinomas are misdiagnosed as primary neurological or psychiatric disease, which can delay potentially curative surgery—sometimes for years.7 The EP should suspect pancreatic insulinoma in any patient who presents with psychiatric symptoms and unexplained episodes of hypoglycemia.7
Cardiac Disease
Transient Left Ventricular Apical Ballooning Syndrome
Transient left ventricular apical ballooning syndrome (TLVABS), first identified in Japan as Takotsubo syndrome, has more recently been recognized worldwide as overlapping with the classic broken heart syndrome. In postmenopausal women, TLVABS appears to follow a catecholamine surge triggered by extreme emotional stress, resulting in an acute coronary artery spasm. Researchers have hypothesized that there may be a link between TLVABS and dissociative amnesia, which is also thought to result from a catecholamine surge in response to emotional stress.8
Nutritional Deficiencies
Wernicke/Korsakoff Syndrome and Thiamine Deficiency
Wernicke encephalopathy and Korsakoff syndrome (WKS) represent a spectrum of neurodegenerative disorders caused by thiamine deficiency. The condition typically occurs in malnourished alcoholic patients, manifesting as a triad of mental status changes, ophthalmoplegia, and ataxia. Recent research has suggested that WKS is more common than previously thought, is not confined exclusively to alcoholic patients, is unlikely to present with the full classic triad, and is typically only diagnosed postmortem.9
Nonalcoholic WKS tends to occur in younger female patients with a wide array of conditions that affect nutrition (eg, gastrointestinal malignancy, bariatric surgery, hyperemesis gravidarum, anorexia nervosa).9 In a patient with chronic alcoholism, application of the Caine criteria (any two of the following findings: ophthalmoplegia, ataxia, even mild memory impairment or confusion without another cause, evidence of malnutrition) has been shown to be more sensitive and specific than the classic triad.10
Subacute Combined Degeneration
Patients with subacute combined degeneration and extrapyramidal symptoms due to B12 (cobalamin) deficiency are well documented. However, patients with B12 deficiency can also present with mood disorders, acute psychosis, psychotic depression, or paranoid hallucinations. The EP should always consider vitamin B12 deficiency as an important, reversible cause of altered mental status—even in the absence of megaloblastic anemia—especially in patients with celiac disease or anorexia nervosa, and in teenagers and those who are vegans/vegetarians.11
Zinc/Vitamin D Deficiency
Zinc and vitamin D deficiency are both highly prevalent in geriatric patients and have been associated with a range of psychiatric complaints, including depressive disorders, bipolar disorder, and psychotic episodes. Though the neurodevelopmental effects of long-term deficiency of these nutrients are well documented in pediatric patients, the role and relationship to acute psychiatric complaints in elderly patients remain unclear.12,13
Endocrine Disorders
Hypothyroidism
Hypothyroidism is a commonly encountered endocrine disruption that classically presents with fatigue, cold insensitivity, weight gain, and thinning hair. Thyroid dysfunction can result in various neuropsychiatric presentations, including mood disorders, cognitive impairment, and exacerbation of underlying psychiatric disorders. Though rare, primary hypothyroidism can present as mania, psychosis, and auditory or visual hallucinations, a phenomenon termed “myxedema madness.” Myxedema madness typically occurs in older women, but has also been described in adolescents and as a postoperative complication of thyroidectomy.14
Hyperthyroidism
Hyperthyroidism classically presents with tachycardia, nervousness or anxiety, heat insensitivity, and weight loss despite increased appetite. Involvement of the CNS in thyrotoxicosis is rare, but when present, it is a significant predictor of mortality. Neuropsychiatric presentations of hyperthyroidism or thyroid storm vary widely, and have been reported to include psychosis, catatonia, auditory hallucinations, delusional parasitosis, new-onset sleepwalking, dissociative disorder, and suicide attempts.15
Steroid Dysregulation
Steroid dysregulation, either endogenous or iatrogenic in nature, has been reported to cause neuropsychiatric symptoms. Major depression with psychotic features can be an initial presentation of Cushing disease, especially in the presence of other systemic signs.16 Adrenal insufficiency has also been shown to cause severe psychotic disorder.17
Chronic treatment with exogenous corticosteroids can cause a recurrent steroid psychosis, primarily manifesting as subacute mania with psychotic features. Treatment of acute adrenal crisis can also cause an acute steroid psychosis with hallucinations, delusions, and dangerous behavior.17
Parathyroid Dysregulation
Elevated calcium levels caused by primary hyperparathyroidism can present as cognitive slowing, reductions in psychomotor speed, memory impairment, and depression. While the disorder is most prevalent in older women, it has been reported in adolescents, and often remains undiagnosed in younger patients until end-organ damage occurs.18 Hypoparathyroidism has also been reported to cause mood disorders, which can occur with or without the classic symptoms of hypocalcemia (eg, tetany, seizures, dementia, and parkinsonism).18
Pheochromocytoma
Pheochromocytoma is a neuroendocrine tumor of the adrenal medulla that causes sympathetic hyperactivity by the release of large amounts of catecholamines. Pheochromocytoma is well-reported to present with nervousness, anxiety, panic attacks, or depression.19
Gonadal Hormone Dysregulation
Gonadal hormone dysregulation can be either congenital or acquired and is typically caused by a pituitary tumor or traumatic brain injury. Thought to be a result of dopaminergic hyperactivity, acute psychosis can develop in cases of hypogonadotropic hypogonadism, hypopituitarism, and/or hyperprolactinemia.20 There is a high incidence of psychotic manifestations in hypogonadal disorders such as Klinefelter syndrome and Prader-Willi syndrome.
Toxins
Many toxins can cause altered mental status and psychiatric manifestations. The administration of these toxins can be iatrogenic, related to prescribed use, or overdose—whether accidental, recreational, or intentional (eg, suicide attempt). Table 2 lists common drugs and toxins associated with psychiatric symptoms.21
Synthetic Drugs
The use of numerous unregulated, synthetic analogues of popular recreational drugs has greatly increased over the last several years. Synthetic cannabinoids are available under a variety of names (eg, “Spice,” “K2”) and can cause prominent psychiatric symptoms, including new-onset psychosis, paranoid delusions, hallucinations, and suicide ideation or attempt. While most clinical symptoms are self-limited and require only supportive care, more serious complications have been reported, including myocardial infarction, ischemic stroke, and acute kidney injury.22 Synthetic cathinones (bath salts) can also cause autonomic instability and prominent acute psychosis, sometimes creating a clinical picture indistinguishable from excited delirium syndrome.23
Heavy Metals
Chronic toxicity of many heavy metals is implicated in abnormal neurodevelopment, behavioral disturbances, and progression of neurodegenerative diseases. Recent literature has also implicated acute metal overload in new-onset impaired emotional behavior, though the mechanism is not currently well understood.24
Case Scenarios Continued
Case 1
[The 62-year-old man with altered mental status.]
The patient’s laboratory evaluation and toxicology screen were negative, including a screen for alcohol. He remained jovial but otherwise in no distress. Since the noncontrast head CT scan showed a subtle asymmetry in the frontal lobes, a magnetic resonance imaging (MRI) study was recommended. The brain MRI showed a 5-cm mass in the right frontal lobe with surrounding edema, findings consistent with glioblastoma multiforme. A neurosurgeon was consulted, and the patient was admitted to the intensive care unit.
Case Scenarios Continued
Case 2
[The 48-year-old woman with chest pain.]
The patient received a dose of oral lorazepam, after which she began to feel less anxious, and her chest pain and shortness of breath also improved slightly. The repeat ECG showed worsening of the ST segment changes. The laboratory evaluation was negative. The patient’s son asked if he could take his mother home for what he felt was much needed rest. The EP, however, ordered a stat two-dimensional echocardiogram (ECHO) and repeat troponin level test. The repeat troponin test was positive, and the ECHO was remarkable for a decreased left ventricular ejection fraction of 15%, with apical ballooning. These findings were consistent with stress cardiomyopathy (Takotsubo syndrome). The patient was admitted to the cardiology service and given a beta blocker and an angiotensin-converting enzyme inhibitor.
After a normal coronary angiogram, the patient developed cardiogenic shock and was intubated. Seven days later, she was extubated and transferred to inpatient rehabilitation services where she also received an assessment and treatment for her underlying depression. Eight weeks postdiagnosis, the patient’s ejection fraction had returned to 50%, and she was close to her baseline exercise tolerance.
1. Barnes J, Dickerson BC, Frost C, Jiskoot LC, Wolk D, van der Flier WM. Alzheimer’s disease first symptoms are age dependent: Evidence from the NACC dataset. Alzheimers Dement. 2015;11(11):1349-1357.
2. Yoshizawa H, Vonsattel JP, Honig LS. Early neuropsychological discriminants for Lewy body disease: an autopsy series. J Neurol Neurosurg Psychiatry. 2013;84(12):1326-1330.
3. Iroka N, Jehangir W, Ii JL, Pattan V, Yousif A, Mishra AK. Paranoid personality masking an atypical case of frontotemporal dementia. J Clin Med Res. 2015;7(5):364-366.
4. Filley CM, Kleinschmidt-DeMasters BK. Neurobehavioral presentations of brain neoplasms. West J Med. 1995;163(1):19-25.
5. Said S, Cooper CJ, Reyna E, Alkhateeb H, Diaz J, Nahleh Z. Paraneoplastic limbic encephalitis, an uncommon presentation of a common cancer: Case report and discussion. Am J Case Rep. 2013;14:391-394.
6. Weitzner MA, Olofsson SM, Forman AD. Patients with malignant meningitis presenting with neuropsychiatric manifestations. Cancer. 1995;76(10):1804-1808.
7. Ding Y, Wang S, Liu J. Neuropsychiatric profiles of patients with insulinomas. Eur Neurol. 2010;63(1):48-51.
8. Toussi A, Bryk J, Alam A. Forgetting heart break: a fascinating case of transient left ventricular apical ballooning syndrome associated with dissociative amnesia. Gen Hosp Psychiatry. 2014;36(2):225-227.
9. Scalzo SJ, Bowden SC, Ambrose ML, Whelan G, Cook MJ. Wernicke-Korsakoff syndrome not related to alcohol use: a systematic review. J Neurol Neurosurg Psychiatry. 2015;86(12):1362-1368.
10. Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: under-recognized and under-treated. Psychosomatics. 2012;53(6):507-516.
11. Issac TG, Soundarya S, Christopher R, Chandra SR. Vitamin B12 deficiency: an important reversible co-morbidity in neuropsychiatric manifestations. Indian J Psychol Med. 2015;37(1):26-29.
12. Grønli O, Kvamme JM, Friborg O, Wynn R. Zinc deficiency is common in several psychiatric disorders. PLoS One. 2013;8(12):e82793.
13. Grønli O, Kvamme JM, Jorde R, Wynn R. Vitamin D deficiency is common in psychogeriatric patients, independent of diagnosis. BMC Psychiatry. 2014;14:134.
14. Heinrich TW, Grahm G. Hypothyroidism presenting as psychosis: myxedema madness revisited. Prim Care Companion J Clin Psychiatry. 2003;5(6):260-266.
15. Swee du S, Chng CL, Lim A. Clinical characteristics and outcome of thyroid storm: a case series and review of neuropsychiatric derangements in thyrotoxicosis. Endocr Pract. 2015;21(2):182-189.
16. Tang A, O’Sullivan AJ, Diamond T, Gerard A, Campbell P. Psychiatric symptoms as a clinical presentation of Cushing’s syndrome. Ann Gen Psychiatry. 2013;12(1):23.
17. Farah Jde L, Lauand CV, Chequi L, et al. Severe psychotic disorder as the main manifestation of adrenal insufficiency. Case Rep Psychiatry. 2015;2015:512430.
18. Rice T, Azova S, Coffey BJ. Negative symptoms in a depressed teen? Primary hyperparathyroidism and its psychiatric manifestations. J Child Adolesc Psychopharmacol. 2015;25(8):653-655.
19. Zardawi IM. Phaeochromocytoma masquerading as anxiety and depression. Am J Case Rep. 2013;14:161-163.
20. Kate S, Dhanwal DK, Kumar S, Bharti P. Acute psychosis as a presentation of hypopituitarism. BMJ Case Rep. 2013;2013.
21. Abramowicz M. Drugs that may cause psychiatric symptoms. Med Lett Drugs Ther. 2008;50(1301-1302):100-103.
22. Tait RJ, Caldicott D, Mountain D, Hill SL, Lenton S. A systematic review of adverse events arising from the use of synthetic cannabinoids and their associated treatment. Clin Toxicol (Phila). 2016;54(1):1-13.
23. Karch SB. Cathinone neurotoxicity (“The “3Ms”). Curr Neuropharmacol. 2015;13(1): 21-25.
24. Menon AV, Chang J, Kim J. Mechanisms of divalent metal toxicity in affective disorders. Toxicology. 2016;339:58-72.
Although the emergency physician (EP) typically encounters common conditions such as chest pain, urinary tract infection, and gastroenteritis, many other clinical presentations can confound diagnosis of the true underlying condition. This may be the case with a patient who presents with apparent psychiatric symptoms that are actually masking an acute medical condition. For example, a patient who appears to be depressed may actually be exhibiting early signs of dementia. Likewise, a manic patient may not have a true underlying psychiatric disorder but rather rhabdomyolysis and hyperthermia from ingesting an illicit substance such as synthetic cathinones (“bath salts”).
Part 1 of this series reviewed psychiatric presentations caused by underlying infectious, pharmacological withdrawal, metabolic, autoimmune, traumatic, and central nervous system etiologies (Emerg Med. 2016;48[5]:202-211). Part 2 covers psychiatric presentations related to dementia, cancer, cardiac disease, nutritional deficiencies, endocrine disorders, or toxins (Table 1).
Case Scenarios
Case 1
A 62-year-old man with a history of hypertension, hyperlipidemia, and past alcohol abuse presented to the ED with reported mental status changes after he was pulled over by police for driving the wrong way down the highway. On presentation, the patient’s vital signs were normal. When questioned, the patient was alert and fully oriented and believed the officers were mistaken about what was reported. He denied any recent illness and had a normal physical examination, including neurological examination.
A brief work-up was ordered and the patient passed the time by politely flirting with the nurses. When his wife arrived at the ED, she was relieved that her husband seemed to be all right. She confirmed that the patient had not consumed any alcohol in years. The patient, meanwhile, playfully minimized his wife’s concern at his presence in the ED. A full toxicology screen, laboratory evaluation, and head computed tomography (CT) scan were ordered.
Case 2
A 48-year-old woman with a history of anxiety disorder, depression, and diabetes mellitus presented to the ED with a 2-hour history of chest pain. She stated that the pain had started toward the end of a heated argument with her son. The patient was escorted into the examination room by hospital security because she was still agitated and kept yelling at her son. On examination the patient was tachycardic (110 beats/minute), diaphoretic, and crying. During the examination, she asked the EP for a “Xanax”; her son further noted that this would help his mother’s condition.
The patient repeatedly claimed she could not breathe and could not lie flat on the stretcher. After verbal de-escalation, she cooperated with the electrocardiography (ECG) technician and phlebotomist. Her ECG showed nonspecific ST changes with no prior study for comparison. While glaring at her son, she maintained that she had constant chest pain.
Dementia
Alzheimer’s Disease
Alzheimer’s disease (AD), the most common cause of dementia, is a chronic neurodegenerative disease characterized by an insidiously progressive cognitive decline and loss of function. There is considerable apparent variability in the early signs of the disease, and recent literature has suggested that the manifestation of initial symptoms may be age-dependent. Younger patients tend to present with non-memory cognitive changes such as problem-solving difficulties, as well as personality changes and behavioral symptoms of depression, apathy, and withdrawal.1
Lewy Body Dementia
Lewy body dementia (LBD) is a chronic neurodegenerative disease with a presentation that overlaps substantially with AD. However, LBD is associated with a significantly more rapid course than AD and presents more frequently with visual hallucinations or illusions due to specific visuospatial dysfunction.2
Frontotemporal Dementia
Frontotemporal dementia is a comparatively rare chronic neurodegenerative disease characterized by early-onset memory impairment with cognitive decline, as well as behavioral changes such as disinhibition, emotional blunting, and language difficulty. Initial presentations can also include atypical features such as paranoia or delusion, and misdiagnosis as a primary psychiatric problem is common.3
Cancer
Brain Tumor
Primary and metastatic brain tumors classically present with either focal neurological signs or less specific symptoms such as headaches, seizures, or syncope. Additionally, central nervous system (CNS) tumors can also initially present with primary psychiatric complaints (eg, personality changes, depression, mania, panic attacks, auditory or visual hallucinations). Patients with a brain neoplasm who are initially misdiagnosed with a primary psychiatric disorder face significant delays in proper diagnosis and treatment, leading to increased morbidity. To correctly diagnose the true cause as soon as possible, early imaging is recommended for patients who present with psychiatric symptoms that are abrupt in onset, atypical in presentation, resistant to conventional treatments, or associated with a change in headache pattern.4
Paraneoplastic Limbic Encephalitis
Paraneoplastic limbic encephalitis (PLE) is a rare neurological consequence of certain cancers. Although PLE most commonly occurs in patients with small cell lung cancer, the condition has also been reported (though less frequently) in cases of esophageal adenocarcinoma, ovarian teratoma, metastatic breast cancer, and germ cell testicular cancer.5 This disease overlaps substantially with anti-N-methyl-D-aspartate (anti-NMDA) receptor encephalitis. Moreover, PLE can present initially with prominent neuropsychiatric symptoms such as confusion, cognitive problems, behavioral changes, irritability, depression, or frank psychosis with hallucinations. Paraneoplastic limbic encephalitis can occur early in the course of cancer—often before other systemic signs appear—and its significance is often only recognized in retrospect or postmortem. A higher index of suspicion for the disorder may lead to earlier detection of treatable cancers.
Malignant Meningitis
Malignant meningitis is the metastatic spread of a primary solid tumor to the leptomeninges. It can present as a wide variety of neuropsychiatric complaints, including depression, anxiety, disorientation, and paranoia. Diagnosis can often be made through lumbar puncture. Malignant meningitis should be considered in the differential diagnosis of new psychiatric symptoms in a patient with a history of cancer—even in the absence of focal neurological deficits or meningeal signs.6
Pancreatic Insulinoma
Pancreatic insulinoma is a rare, potentially curable endocrine tumor that can present initially with vague psychiatric complaints such as irrational behavior, confusion, depression, or anxiety. In up to 64% of patients, insulinomas are misdiagnosed as primary neurological or psychiatric disease, which can delay potentially curative surgery—sometimes for years.7 The EP should suspect pancreatic insulinoma in any patient who presents with psychiatric symptoms and unexplained episodes of hypoglycemia.7
Cardiac Disease
Transient Left Ventricular Apical Ballooning Syndrome
Transient left ventricular apical ballooning syndrome (TLVABS), first identified in Japan as Takotsubo syndrome, has more recently been recognized worldwide as overlapping with the classic broken heart syndrome. In postmenopausal women, TLVABS appears to follow a catecholamine surge triggered by extreme emotional stress, resulting in an acute coronary artery spasm. Researchers have hypothesized that there may be a link between TLVABS and dissociative amnesia, which is also thought to result from a catecholamine surge in response to emotional stress.8
Nutritional Deficiencies
Wernicke/Korsakoff Syndrome and Thiamine Deficiency
Wernicke encephalopathy and Korsakoff syndrome (WKS) represent a spectrum of neurodegenerative disorders caused by thiamine deficiency. The condition typically occurs in malnourished alcoholic patients, manifesting as a triad of mental status changes, ophthalmoplegia, and ataxia. Recent research has suggested that WKS is more common than previously thought, is not confined exclusively to alcoholic patients, is unlikely to present with the full classic triad, and is typically only diagnosed postmortem.9
Nonalcoholic WKS tends to occur in younger female patients with a wide array of conditions that affect nutrition (eg, gastrointestinal malignancy, bariatric surgery, hyperemesis gravidarum, anorexia nervosa).9 In a patient with chronic alcoholism, application of the Caine criteria (any two of the following findings: ophthalmoplegia, ataxia, even mild memory impairment or confusion without another cause, evidence of malnutrition) has been shown to be more sensitive and specific than the classic triad.10
Subacute Combined Degeneration
Patients with subacute combined degeneration and extrapyramidal symptoms due to B12 (cobalamin) deficiency are well documented. However, patients with B12 deficiency can also present with mood disorders, acute psychosis, psychotic depression, or paranoid hallucinations. The EP should always consider vitamin B12 deficiency as an important, reversible cause of altered mental status—even in the absence of megaloblastic anemia—especially in patients with celiac disease or anorexia nervosa, and in teenagers and those who are vegans/vegetarians.11
Zinc/Vitamin D Deficiency
Zinc and vitamin D deficiency are both highly prevalent in geriatric patients and have been associated with a range of psychiatric complaints, including depressive disorders, bipolar disorder, and psychotic episodes. Though the neurodevelopmental effects of long-term deficiency of these nutrients are well documented in pediatric patients, the role and relationship to acute psychiatric complaints in elderly patients remain unclear.12,13
Endocrine Disorders
Hypothyroidism
Hypothyroidism is a commonly encountered endocrine disruption that classically presents with fatigue, cold insensitivity, weight gain, and thinning hair. Thyroid dysfunction can result in various neuropsychiatric presentations, including mood disorders, cognitive impairment, and exacerbation of underlying psychiatric disorders. Though rare, primary hypothyroidism can present as mania, psychosis, and auditory or visual hallucinations, a phenomenon termed “myxedema madness.” Myxedema madness typically occurs in older women, but has also been described in adolescents and as a postoperative complication of thyroidectomy.14
Hyperthyroidism
Hyperthyroidism classically presents with tachycardia, nervousness or anxiety, heat insensitivity, and weight loss despite increased appetite. Involvement of the CNS in thyrotoxicosis is rare, but when present, it is a significant predictor of mortality. Neuropsychiatric presentations of hyperthyroidism or thyroid storm vary widely, and have been reported to include psychosis, catatonia, auditory hallucinations, delusional parasitosis, new-onset sleepwalking, dissociative disorder, and suicide attempts.15
Steroid Dysregulation
Steroid dysregulation, either endogenous or iatrogenic in nature, has been reported to cause neuropsychiatric symptoms. Major depression with psychotic features can be an initial presentation of Cushing disease, especially in the presence of other systemic signs.16 Adrenal insufficiency has also been shown to cause severe psychotic disorder.17
Chronic treatment with exogenous corticosteroids can cause a recurrent steroid psychosis, primarily manifesting as subacute mania with psychotic features. Treatment of acute adrenal crisis can also cause an acute steroid psychosis with hallucinations, delusions, and dangerous behavior.17
Parathyroid Dysregulation
Elevated calcium levels caused by primary hyperparathyroidism can present as cognitive slowing, reductions in psychomotor speed, memory impairment, and depression. While the disorder is most prevalent in older women, it has been reported in adolescents, and often remains undiagnosed in younger patients until end-organ damage occurs.18 Hypoparathyroidism has also been reported to cause mood disorders, which can occur with or without the classic symptoms of hypocalcemia (eg, tetany, seizures, dementia, and parkinsonism).18
Pheochromocytoma
Pheochromocytoma is a neuroendocrine tumor of the adrenal medulla that causes sympathetic hyperactivity by the release of large amounts of catecholamines. Pheochromocytoma is well-reported to present with nervousness, anxiety, panic attacks, or depression.19
Gonadal Hormone Dysregulation
Gonadal hormone dysregulation can be either congenital or acquired and is typically caused by a pituitary tumor or traumatic brain injury. Thought to be a result of dopaminergic hyperactivity, acute psychosis can develop in cases of hypogonadotropic hypogonadism, hypopituitarism, and/or hyperprolactinemia.20 There is a high incidence of psychotic manifestations in hypogonadal disorders such as Klinefelter syndrome and Prader-Willi syndrome.
Toxins
Many toxins can cause altered mental status and psychiatric manifestations. The administration of these toxins can be iatrogenic, related to prescribed use, or overdose—whether accidental, recreational, or intentional (eg, suicide attempt). Table 2 lists common drugs and toxins associated with psychiatric symptoms.21
Synthetic Drugs
The use of numerous unregulated, synthetic analogues of popular recreational drugs has greatly increased over the last several years. Synthetic cannabinoids are available under a variety of names (eg, “Spice,” “K2”) and can cause prominent psychiatric symptoms, including new-onset psychosis, paranoid delusions, hallucinations, and suicide ideation or attempt. While most clinical symptoms are self-limited and require only supportive care, more serious complications have been reported, including myocardial infarction, ischemic stroke, and acute kidney injury.22 Synthetic cathinones (bath salts) can also cause autonomic instability and prominent acute psychosis, sometimes creating a clinical picture indistinguishable from excited delirium syndrome.23
Heavy Metals
Chronic toxicity of many heavy metals is implicated in abnormal neurodevelopment, behavioral disturbances, and progression of neurodegenerative diseases. Recent literature has also implicated acute metal overload in new-onset impaired emotional behavior, though the mechanism is not currently well understood.24
Case Scenarios Continued
Case 1
[The 62-year-old man with altered mental status.]
The patient’s laboratory evaluation and toxicology screen were negative, including a screen for alcohol. He remained jovial but otherwise in no distress. Since the noncontrast head CT scan showed a subtle asymmetry in the frontal lobes, a magnetic resonance imaging (MRI) study was recommended. The brain MRI showed a 5-cm mass in the right frontal lobe with surrounding edema, findings consistent with glioblastoma multiforme. A neurosurgeon was consulted, and the patient was admitted to the intensive care unit.
Case Scenarios Continued
Case 2
[The 48-year-old woman with chest pain.]
The patient received a dose of oral lorazepam, after which she began to feel less anxious, and her chest pain and shortness of breath also improved slightly. The repeat ECG showed worsening of the ST segment changes. The laboratory evaluation was negative. The patient’s son asked if he could take his mother home for what he felt was much needed rest. The EP, however, ordered a stat two-dimensional echocardiogram (ECHO) and repeat troponin level test. The repeat troponin test was positive, and the ECHO was remarkable for a decreased left ventricular ejection fraction of 15%, with apical ballooning. These findings were consistent with stress cardiomyopathy (Takotsubo syndrome). The patient was admitted to the cardiology service and given a beta blocker and an angiotensin-converting enzyme inhibitor.
After a normal coronary angiogram, the patient developed cardiogenic shock and was intubated. Seven days later, she was extubated and transferred to inpatient rehabilitation services where she also received an assessment and treatment for her underlying depression. Eight weeks postdiagnosis, the patient’s ejection fraction had returned to 50%, and she was close to her baseline exercise tolerance.
Although the emergency physician (EP) typically encounters common conditions such as chest pain, urinary tract infection, and gastroenteritis, many other clinical presentations can confound diagnosis of the true underlying condition. This may be the case with a patient who presents with apparent psychiatric symptoms that are actually masking an acute medical condition. For example, a patient who appears to be depressed may actually be exhibiting early signs of dementia. Likewise, a manic patient may not have a true underlying psychiatric disorder but rather rhabdomyolysis and hyperthermia from ingesting an illicit substance such as synthetic cathinones (“bath salts”).
Part 1 of this series reviewed psychiatric presentations caused by underlying infectious, pharmacological withdrawal, metabolic, autoimmune, traumatic, and central nervous system etiologies (Emerg Med. 2016;48[5]:202-211). Part 2 covers psychiatric presentations related to dementia, cancer, cardiac disease, nutritional deficiencies, endocrine disorders, or toxins (Table 1).
Case Scenarios
Case 1
A 62-year-old man with a history of hypertension, hyperlipidemia, and past alcohol abuse presented to the ED with reported mental status changes after he was pulled over by police for driving the wrong way down the highway. On presentation, the patient’s vital signs were normal. When questioned, the patient was alert and fully oriented and believed the officers were mistaken about what was reported. He denied any recent illness and had a normal physical examination, including neurological examination.
A brief work-up was ordered and the patient passed the time by politely flirting with the nurses. When his wife arrived at the ED, she was relieved that her husband seemed to be all right. She confirmed that the patient had not consumed any alcohol in years. The patient, meanwhile, playfully minimized his wife’s concern at his presence in the ED. A full toxicology screen, laboratory evaluation, and head computed tomography (CT) scan were ordered.
Case 2
A 48-year-old woman with a history of anxiety disorder, depression, and diabetes mellitus presented to the ED with a 2-hour history of chest pain. She stated that the pain had started toward the end of a heated argument with her son. The patient was escorted into the examination room by hospital security because she was still agitated and kept yelling at her son. On examination the patient was tachycardic (110 beats/minute), diaphoretic, and crying. During the examination, she asked the EP for a “Xanax”; her son further noted that this would help his mother’s condition.
The patient repeatedly claimed she could not breathe and could not lie flat on the stretcher. After verbal de-escalation, she cooperated with the electrocardiography (ECG) technician and phlebotomist. Her ECG showed nonspecific ST changes with no prior study for comparison. While glaring at her son, she maintained that she had constant chest pain.
Dementia
Alzheimer’s Disease
Alzheimer’s disease (AD), the most common cause of dementia, is a chronic neurodegenerative disease characterized by an insidiously progressive cognitive decline and loss of function. There is considerable apparent variability in the early signs of the disease, and recent literature has suggested that the manifestation of initial symptoms may be age-dependent. Younger patients tend to present with non-memory cognitive changes such as problem-solving difficulties, as well as personality changes and behavioral symptoms of depression, apathy, and withdrawal.1
Lewy Body Dementia
Lewy body dementia (LBD) is a chronic neurodegenerative disease with a presentation that overlaps substantially with AD. However, LBD is associated with a significantly more rapid course than AD and presents more frequently with visual hallucinations or illusions due to specific visuospatial dysfunction.2
Frontotemporal Dementia
Frontotemporal dementia is a comparatively rare chronic neurodegenerative disease characterized by early-onset memory impairment with cognitive decline, as well as behavioral changes such as disinhibition, emotional blunting, and language difficulty. Initial presentations can also include atypical features such as paranoia or delusion, and misdiagnosis as a primary psychiatric problem is common.3
Cancer
Brain Tumor
Primary and metastatic brain tumors classically present with either focal neurological signs or less specific symptoms such as headaches, seizures, or syncope. Additionally, central nervous system (CNS) tumors can also initially present with primary psychiatric complaints (eg, personality changes, depression, mania, panic attacks, auditory or visual hallucinations). Patients with a brain neoplasm who are initially misdiagnosed with a primary psychiatric disorder face significant delays in proper diagnosis and treatment, leading to increased morbidity. To correctly diagnose the true cause as soon as possible, early imaging is recommended for patients who present with psychiatric symptoms that are abrupt in onset, atypical in presentation, resistant to conventional treatments, or associated with a change in headache pattern.4
Paraneoplastic Limbic Encephalitis
Paraneoplastic limbic encephalitis (PLE) is a rare neurological consequence of certain cancers. Although PLE most commonly occurs in patients with small cell lung cancer, the condition has also been reported (though less frequently) in cases of esophageal adenocarcinoma, ovarian teratoma, metastatic breast cancer, and germ cell testicular cancer.5 This disease overlaps substantially with anti-N-methyl-D-aspartate (anti-NMDA) receptor encephalitis. Moreover, PLE can present initially with prominent neuropsychiatric symptoms such as confusion, cognitive problems, behavioral changes, irritability, depression, or frank psychosis with hallucinations. Paraneoplastic limbic encephalitis can occur early in the course of cancer—often before other systemic signs appear—and its significance is often only recognized in retrospect or postmortem. A higher index of suspicion for the disorder may lead to earlier detection of treatable cancers.
Malignant Meningitis
Malignant meningitis is the metastatic spread of a primary solid tumor to the leptomeninges. It can present as a wide variety of neuropsychiatric complaints, including depression, anxiety, disorientation, and paranoia. Diagnosis can often be made through lumbar puncture. Malignant meningitis should be considered in the differential diagnosis of new psychiatric symptoms in a patient with a history of cancer—even in the absence of focal neurological deficits or meningeal signs.6
Pancreatic Insulinoma
Pancreatic insulinoma is a rare, potentially curable endocrine tumor that can present initially with vague psychiatric complaints such as irrational behavior, confusion, depression, or anxiety. In up to 64% of patients, insulinomas are misdiagnosed as primary neurological or psychiatric disease, which can delay potentially curative surgery—sometimes for years.7 The EP should suspect pancreatic insulinoma in any patient who presents with psychiatric symptoms and unexplained episodes of hypoglycemia.7
Cardiac Disease
Transient Left Ventricular Apical Ballooning Syndrome
Transient left ventricular apical ballooning syndrome (TLVABS), first identified in Japan as Takotsubo syndrome, has more recently been recognized worldwide as overlapping with the classic broken heart syndrome. In postmenopausal women, TLVABS appears to follow a catecholamine surge triggered by extreme emotional stress, resulting in an acute coronary artery spasm. Researchers have hypothesized that there may be a link between TLVABS and dissociative amnesia, which is also thought to result from a catecholamine surge in response to emotional stress.8
Nutritional Deficiencies
Wernicke/Korsakoff Syndrome and Thiamine Deficiency
Wernicke encephalopathy and Korsakoff syndrome (WKS) represent a spectrum of neurodegenerative disorders caused by thiamine deficiency. The condition typically occurs in malnourished alcoholic patients, manifesting as a triad of mental status changes, ophthalmoplegia, and ataxia. Recent research has suggested that WKS is more common than previously thought, is not confined exclusively to alcoholic patients, is unlikely to present with the full classic triad, and is typically only diagnosed postmortem.9
Nonalcoholic WKS tends to occur in younger female patients with a wide array of conditions that affect nutrition (eg, gastrointestinal malignancy, bariatric surgery, hyperemesis gravidarum, anorexia nervosa).9 In a patient with chronic alcoholism, application of the Caine criteria (any two of the following findings: ophthalmoplegia, ataxia, even mild memory impairment or confusion without another cause, evidence of malnutrition) has been shown to be more sensitive and specific than the classic triad.10
Subacute Combined Degeneration
Patients with subacute combined degeneration and extrapyramidal symptoms due to B12 (cobalamin) deficiency are well documented. However, patients with B12 deficiency can also present with mood disorders, acute psychosis, psychotic depression, or paranoid hallucinations. The EP should always consider vitamin B12 deficiency as an important, reversible cause of altered mental status—even in the absence of megaloblastic anemia—especially in patients with celiac disease or anorexia nervosa, and in teenagers and those who are vegans/vegetarians.11
Zinc/Vitamin D Deficiency
Zinc and vitamin D deficiency are both highly prevalent in geriatric patients and have been associated with a range of psychiatric complaints, including depressive disorders, bipolar disorder, and psychotic episodes. Though the neurodevelopmental effects of long-term deficiency of these nutrients are well documented in pediatric patients, the role and relationship to acute psychiatric complaints in elderly patients remain unclear.12,13
Endocrine Disorders
Hypothyroidism
Hypothyroidism is a commonly encountered endocrine disruption that classically presents with fatigue, cold insensitivity, weight gain, and thinning hair. Thyroid dysfunction can result in various neuropsychiatric presentations, including mood disorders, cognitive impairment, and exacerbation of underlying psychiatric disorders. Though rare, primary hypothyroidism can present as mania, psychosis, and auditory or visual hallucinations, a phenomenon termed “myxedema madness.” Myxedema madness typically occurs in older women, but has also been described in adolescents and as a postoperative complication of thyroidectomy.14
Hyperthyroidism
Hyperthyroidism classically presents with tachycardia, nervousness or anxiety, heat insensitivity, and weight loss despite increased appetite. Involvement of the CNS in thyrotoxicosis is rare, but when present, it is a significant predictor of mortality. Neuropsychiatric presentations of hyperthyroidism or thyroid storm vary widely, and have been reported to include psychosis, catatonia, auditory hallucinations, delusional parasitosis, new-onset sleepwalking, dissociative disorder, and suicide attempts.15
Steroid Dysregulation
Steroid dysregulation, either endogenous or iatrogenic in nature, has been reported to cause neuropsychiatric symptoms. Major depression with psychotic features can be an initial presentation of Cushing disease, especially in the presence of other systemic signs.16 Adrenal insufficiency has also been shown to cause severe psychotic disorder.17
Chronic treatment with exogenous corticosteroids can cause a recurrent steroid psychosis, primarily manifesting as subacute mania with psychotic features. Treatment of acute adrenal crisis can also cause an acute steroid psychosis with hallucinations, delusions, and dangerous behavior.17
Parathyroid Dysregulation
Elevated calcium levels caused by primary hyperparathyroidism can present as cognitive slowing, reductions in psychomotor speed, memory impairment, and depression. While the disorder is most prevalent in older women, it has been reported in adolescents, and often remains undiagnosed in younger patients until end-organ damage occurs.18 Hypoparathyroidism has also been reported to cause mood disorders, which can occur with or without the classic symptoms of hypocalcemia (eg, tetany, seizures, dementia, and parkinsonism).18
Pheochromocytoma
Pheochromocytoma is a neuroendocrine tumor of the adrenal medulla that causes sympathetic hyperactivity by the release of large amounts of catecholamines. Pheochromocytoma is well-reported to present with nervousness, anxiety, panic attacks, or depression.19
Gonadal Hormone Dysregulation
Gonadal hormone dysregulation can be either congenital or acquired and is typically caused by a pituitary tumor or traumatic brain injury. Thought to be a result of dopaminergic hyperactivity, acute psychosis can develop in cases of hypogonadotropic hypogonadism, hypopituitarism, and/or hyperprolactinemia.20 There is a high incidence of psychotic manifestations in hypogonadal disorders such as Klinefelter syndrome and Prader-Willi syndrome.
Toxins
Many toxins can cause altered mental status and psychiatric manifestations. The administration of these toxins can be iatrogenic, related to prescribed use, or overdose—whether accidental, recreational, or intentional (eg, suicide attempt). Table 2 lists common drugs and toxins associated with psychiatric symptoms.21
Synthetic Drugs
The use of numerous unregulated, synthetic analogues of popular recreational drugs has greatly increased over the last several years. Synthetic cannabinoids are available under a variety of names (eg, “Spice,” “K2”) and can cause prominent psychiatric symptoms, including new-onset psychosis, paranoid delusions, hallucinations, and suicide ideation or attempt. While most clinical symptoms are self-limited and require only supportive care, more serious complications have been reported, including myocardial infarction, ischemic stroke, and acute kidney injury.22 Synthetic cathinones (bath salts) can also cause autonomic instability and prominent acute psychosis, sometimes creating a clinical picture indistinguishable from excited delirium syndrome.23
Heavy Metals
Chronic toxicity of many heavy metals is implicated in abnormal neurodevelopment, behavioral disturbances, and progression of neurodegenerative diseases. Recent literature has also implicated acute metal overload in new-onset impaired emotional behavior, though the mechanism is not currently well understood.24
Case Scenarios Continued
Case 1
[The 62-year-old man with altered mental status.]
The patient’s laboratory evaluation and toxicology screen were negative, including a screen for alcohol. He remained jovial but otherwise in no distress. Since the noncontrast head CT scan showed a subtle asymmetry in the frontal lobes, a magnetic resonance imaging (MRI) study was recommended. The brain MRI showed a 5-cm mass in the right frontal lobe with surrounding edema, findings consistent with glioblastoma multiforme. A neurosurgeon was consulted, and the patient was admitted to the intensive care unit.
Case Scenarios Continued
Case 2
[The 48-year-old woman with chest pain.]
The patient received a dose of oral lorazepam, after which she began to feel less anxious, and her chest pain and shortness of breath also improved slightly. The repeat ECG showed worsening of the ST segment changes. The laboratory evaluation was negative. The patient’s son asked if he could take his mother home for what he felt was much needed rest. The EP, however, ordered a stat two-dimensional echocardiogram (ECHO) and repeat troponin level test. The repeat troponin test was positive, and the ECHO was remarkable for a decreased left ventricular ejection fraction of 15%, with apical ballooning. These findings were consistent with stress cardiomyopathy (Takotsubo syndrome). The patient was admitted to the cardiology service and given a beta blocker and an angiotensin-converting enzyme inhibitor.
After a normal coronary angiogram, the patient developed cardiogenic shock and was intubated. Seven days later, she was extubated and transferred to inpatient rehabilitation services where she also received an assessment and treatment for her underlying depression. Eight weeks postdiagnosis, the patient’s ejection fraction had returned to 50%, and she was close to her baseline exercise tolerance.
1. Barnes J, Dickerson BC, Frost C, Jiskoot LC, Wolk D, van der Flier WM. Alzheimer’s disease first symptoms are age dependent: Evidence from the NACC dataset. Alzheimers Dement. 2015;11(11):1349-1357.
2. Yoshizawa H, Vonsattel JP, Honig LS. Early neuropsychological discriminants for Lewy body disease: an autopsy series. J Neurol Neurosurg Psychiatry. 2013;84(12):1326-1330.
3. Iroka N, Jehangir W, Ii JL, Pattan V, Yousif A, Mishra AK. Paranoid personality masking an atypical case of frontotemporal dementia. J Clin Med Res. 2015;7(5):364-366.
4. Filley CM, Kleinschmidt-DeMasters BK. Neurobehavioral presentations of brain neoplasms. West J Med. 1995;163(1):19-25.
5. Said S, Cooper CJ, Reyna E, Alkhateeb H, Diaz J, Nahleh Z. Paraneoplastic limbic encephalitis, an uncommon presentation of a common cancer: Case report and discussion. Am J Case Rep. 2013;14:391-394.
6. Weitzner MA, Olofsson SM, Forman AD. Patients with malignant meningitis presenting with neuropsychiatric manifestations. Cancer. 1995;76(10):1804-1808.
7. Ding Y, Wang S, Liu J. Neuropsychiatric profiles of patients with insulinomas. Eur Neurol. 2010;63(1):48-51.
8. Toussi A, Bryk J, Alam A. Forgetting heart break: a fascinating case of transient left ventricular apical ballooning syndrome associated with dissociative amnesia. Gen Hosp Psychiatry. 2014;36(2):225-227.
9. Scalzo SJ, Bowden SC, Ambrose ML, Whelan G, Cook MJ. Wernicke-Korsakoff syndrome not related to alcohol use: a systematic review. J Neurol Neurosurg Psychiatry. 2015;86(12):1362-1368.
10. Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: under-recognized and under-treated. Psychosomatics. 2012;53(6):507-516.
11. Issac TG, Soundarya S, Christopher R, Chandra SR. Vitamin B12 deficiency: an important reversible co-morbidity in neuropsychiatric manifestations. Indian J Psychol Med. 2015;37(1):26-29.
12. Grønli O, Kvamme JM, Friborg O, Wynn R. Zinc deficiency is common in several psychiatric disorders. PLoS One. 2013;8(12):e82793.
13. Grønli O, Kvamme JM, Jorde R, Wynn R. Vitamin D deficiency is common in psychogeriatric patients, independent of diagnosis. BMC Psychiatry. 2014;14:134.
14. Heinrich TW, Grahm G. Hypothyroidism presenting as psychosis: myxedema madness revisited. Prim Care Companion J Clin Psychiatry. 2003;5(6):260-266.
15. Swee du S, Chng CL, Lim A. Clinical characteristics and outcome of thyroid storm: a case series and review of neuropsychiatric derangements in thyrotoxicosis. Endocr Pract. 2015;21(2):182-189.
16. Tang A, O’Sullivan AJ, Diamond T, Gerard A, Campbell P. Psychiatric symptoms as a clinical presentation of Cushing’s syndrome. Ann Gen Psychiatry. 2013;12(1):23.
17. Farah Jde L, Lauand CV, Chequi L, et al. Severe psychotic disorder as the main manifestation of adrenal insufficiency. Case Rep Psychiatry. 2015;2015:512430.
18. Rice T, Azova S, Coffey BJ. Negative symptoms in a depressed teen? Primary hyperparathyroidism and its psychiatric manifestations. J Child Adolesc Psychopharmacol. 2015;25(8):653-655.
19. Zardawi IM. Phaeochromocytoma masquerading as anxiety and depression. Am J Case Rep. 2013;14:161-163.
20. Kate S, Dhanwal DK, Kumar S, Bharti P. Acute psychosis as a presentation of hypopituitarism. BMJ Case Rep. 2013;2013.
21. Abramowicz M. Drugs that may cause psychiatric symptoms. Med Lett Drugs Ther. 2008;50(1301-1302):100-103.
22. Tait RJ, Caldicott D, Mountain D, Hill SL, Lenton S. A systematic review of adverse events arising from the use of synthetic cannabinoids and their associated treatment. Clin Toxicol (Phila). 2016;54(1):1-13.
23. Karch SB. Cathinone neurotoxicity (“The “3Ms”). Curr Neuropharmacol. 2015;13(1): 21-25.
24. Menon AV, Chang J, Kim J. Mechanisms of divalent metal toxicity in affective disorders. Toxicology. 2016;339:58-72.
1. Barnes J, Dickerson BC, Frost C, Jiskoot LC, Wolk D, van der Flier WM. Alzheimer’s disease first symptoms are age dependent: Evidence from the NACC dataset. Alzheimers Dement. 2015;11(11):1349-1357.
2. Yoshizawa H, Vonsattel JP, Honig LS. Early neuropsychological discriminants for Lewy body disease: an autopsy series. J Neurol Neurosurg Psychiatry. 2013;84(12):1326-1330.
3. Iroka N, Jehangir W, Ii JL, Pattan V, Yousif A, Mishra AK. Paranoid personality masking an atypical case of frontotemporal dementia. J Clin Med Res. 2015;7(5):364-366.
4. Filley CM, Kleinschmidt-DeMasters BK. Neurobehavioral presentations of brain neoplasms. West J Med. 1995;163(1):19-25.
5. Said S, Cooper CJ, Reyna E, Alkhateeb H, Diaz J, Nahleh Z. Paraneoplastic limbic encephalitis, an uncommon presentation of a common cancer: Case report and discussion. Am J Case Rep. 2013;14:391-394.
6. Weitzner MA, Olofsson SM, Forman AD. Patients with malignant meningitis presenting with neuropsychiatric manifestations. Cancer. 1995;76(10):1804-1808.
7. Ding Y, Wang S, Liu J. Neuropsychiatric profiles of patients with insulinomas. Eur Neurol. 2010;63(1):48-51.
8. Toussi A, Bryk J, Alam A. Forgetting heart break: a fascinating case of transient left ventricular apical ballooning syndrome associated with dissociative amnesia. Gen Hosp Psychiatry. 2014;36(2):225-227.
9. Scalzo SJ, Bowden SC, Ambrose ML, Whelan G, Cook MJ. Wernicke-Korsakoff syndrome not related to alcohol use: a systematic review. J Neurol Neurosurg Psychiatry. 2015;86(12):1362-1368.
10. Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: under-recognized and under-treated. Psychosomatics. 2012;53(6):507-516.
11. Issac TG, Soundarya S, Christopher R, Chandra SR. Vitamin B12 deficiency: an important reversible co-morbidity in neuropsychiatric manifestations. Indian J Psychol Med. 2015;37(1):26-29.
12. Grønli O, Kvamme JM, Friborg O, Wynn R. Zinc deficiency is common in several psychiatric disorders. PLoS One. 2013;8(12):e82793.
13. Grønli O, Kvamme JM, Jorde R, Wynn R. Vitamin D deficiency is common in psychogeriatric patients, independent of diagnosis. BMC Psychiatry. 2014;14:134.
14. Heinrich TW, Grahm G. Hypothyroidism presenting as psychosis: myxedema madness revisited. Prim Care Companion J Clin Psychiatry. 2003;5(6):260-266.
15. Swee du S, Chng CL, Lim A. Clinical characteristics and outcome of thyroid storm: a case series and review of neuropsychiatric derangements in thyrotoxicosis. Endocr Pract. 2015;21(2):182-189.
16. Tang A, O’Sullivan AJ, Diamond T, Gerard A, Campbell P. Psychiatric symptoms as a clinical presentation of Cushing’s syndrome. Ann Gen Psychiatry. 2013;12(1):23.
17. Farah Jde L, Lauand CV, Chequi L, et al. Severe psychotic disorder as the main manifestation of adrenal insufficiency. Case Rep Psychiatry. 2015;2015:512430.
18. Rice T, Azova S, Coffey BJ. Negative symptoms in a depressed teen? Primary hyperparathyroidism and its psychiatric manifestations. J Child Adolesc Psychopharmacol. 2015;25(8):653-655.
19. Zardawi IM. Phaeochromocytoma masquerading as anxiety and depression. Am J Case Rep. 2013;14:161-163.
20. Kate S, Dhanwal DK, Kumar S, Bharti P. Acute psychosis as a presentation of hypopituitarism. BMJ Case Rep. 2013;2013.
21. Abramowicz M. Drugs that may cause psychiatric symptoms. Med Lett Drugs Ther. 2008;50(1301-1302):100-103.
22. Tait RJ, Caldicott D, Mountain D, Hill SL, Lenton S. A systematic review of adverse events arising from the use of synthetic cannabinoids and their associated treatment. Clin Toxicol (Phila). 2016;54(1):1-13.
23. Karch SB. Cathinone neurotoxicity (“The “3Ms”). Curr Neuropharmacol. 2015;13(1): 21-25.
24. Menon AV, Chang J, Kim J. Mechanisms of divalent metal toxicity in affective disorders. Toxicology. 2016;339:58-72.