User login
Prophylaxis and Treatment of Venous Thromboembolism in Cancer Patients
The Case
A 62-year-old woman with a past medical history significant for metastatic adenocarcinoma of the lung presents to the ED with complaints of fever and shortness of breath. She has recently completed her first cycle of carboplatin, pemetrexed, and bevacizumab. Upon admission, she is found to have an absolute neutrophil count of 800 and a platelet count of 48,000. She is admitted for neutropenic fever and placed on IV antimicrobials. Sequential compression devices are initiated for DVT prophylaxis.
Key Clinical Questions
What risk do cancer patients have for VTE?
Patients with cancer have a risk of clinically significant VTE that is four to seven times that of patients without malignancy.1 This is due to a number of reasons:
- Tumor cells produce procoagulant activity inducing thrombin formation;2
- The cancer itself can compress or invade deep veins; and3
- Some cancer therapies such L-asparaginase and thalidomide/lenalidomide, plus high-dose steroids, or anti-estrogen medications such as tamoxifen can also increase patients’ risk of VTE.3,4,5
What inpatients with cancer need VTE prophylaxis?
Much like other hospitalized medical patients, patients with cancer who have reduced mobility and are not on therapeutic anticoagulation should receive pharmacologic prophylaxis unless there is a contraindication.3,6,7,8 Cancer patients with acute medical illnesses should also likely receive prophylaxis if there are no contraindications, because the vast majority of these have factors increasing their VTE risk, including infection, kidney disease, or pulmonary disease.3,6,7,8 Patients undergoing major cancer surgery should also receive pharmacologic prophylaxis prior to surgery and for at least seven to 10 days post-operatively.3,6,7,8
For ambulatory cancer patients who are admitted for short courses of chemotherapy or for minor procedures, however, there is not enough evidence to recommend routine VTE prophylaxis.6,7 An exception to this is patients with multiple myeloma receiving thalidomide-based or lenalidomide-based chemotherapy, who should receive pharmacologic prophylaxis.6,7
What are the options available for VTE in hospitalized cancer patients?
The guidelines for VTE prophylaxis in hospitalized cancer patients recommend either unfractionated heparin (UFH) or low molecular weight heparin (LMWH) for prophylaxis when no contraindications exist.5 The only two LMWH that have been FDA approved for prophylaxis are enoxaparin and dalteparin. When deciding between UFH and LMWH, no evidence shows that one is better than the other in preventing VTE in hospitalized cancer patients.9 There is evidence that the use of LMWH results in a lower incidence of major hemorrhage when compared to UFH.10
What are the contraindications to pharmacologic VTE prophylaxis in cancer patients?
Contraindications for pharmacologic VTE prophylaxis in cancer patients include active major bleeding, thrombocytopenia (platelet count <50,000/µL), severe coagulopathy, inherited bleeding disorder, and at the time of surgery or invasive procedures (including lumbar puncture and epidural or spinal anesthesia).3,6,7 Those with contraindications to pharmacologic VTE prophylaxis should have mechanical prophylaxis instead.
What is the recommended treatment of VTE in cancer patients?
After the diagnosis of pulmonary embolism (PE) or DVT is found, LMWH is the preferred initial anticoagulant instead of UFH unless the patient has severe renal impairment (CrCl of less than 30 ml/min).6,7,8 LMWH is also preferred over warfarin for long-term anticoagulation during the initial six months of therapy.6,7,8 Following the initial six months, continued anticoagulation with either LMWH or warfarin could be considered in patients with active cancer, metastatic disease, or ongoing chemotherapy.6,7,8
When should IVC filters be considered in treating VTE in cancer patients?
IVC filter insertion should be reserved for those patients found to have a DVT or PE who have a contraindication to pharmacologic anticoagulation.3,6 It can be considered in patients who have recurrent VTE despite the appropriate use of optimally dosed LMWH therapy.6,8
What about the new oral anticoagulants?
At this point, because the majority of the major trials looking at the new oral anticoagulants (dabigatran, rivaroxaban, and apixaban) excluded cancer patients or included them only in small numbers, there is not enough evidence to support their use in cancer patients diagnosed with VTE.6,7,8
Back to the Case
On hospital day three, the patient is clinically improved. She is afebrile, her neutropenia has resolved, and her platelet count is up to 80,000. Her only complaint is pain and swelling of her left leg. A lower extremity Doppler is performed. She is found to have an acute left femoral DVT. The patient is then started on enoxaparin 1 mg/kg every 12 hours. Her left leg swelling and pain begin to improve, and she is discharged on enoxaparin and follows up with her oncologist in the next week. TH
Drs. Bell and O’Rourke are assistant professor of medicine in the division of hospital medicine at the University of California San Diego.
References
1. Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013;122(10):1712-1723.
2. Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293(6):715-722.
3. Streiff MB, Bockenstedt PL, Cataland SR, et al. Venous thromboembolic disease. J Natl Compr Canc Netw. 2013;11(11):1402-1429.
4. Payne JH, Vora AJ. Thrombosis and acute lymphoblastic leukaemia. Br J Haematol. 2007;138(4):430-445.
5. Amir E, Seruga B, Niraula S, Carlsson L, Ocaña A. Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst. 2011;103(17):1299-1309.
6. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(17):2189-2204.
7. Lyman GH, Bohlke K, Khorana AA, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: american society of clinical oncology clinical practice guideline update 2014. J Clin Oncol. 2015;33(6):654-656.
8. Farge D, Debourdeau P, Beckers M, et al. International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. J Thromb Haemost. 2013;11(1):56-70.
9. Khorana AA. The NCCN clinical practice guidelines on venous thromboembolic disease: strategies for improving VTE prophylaxis in hospitalized cancer patients. Oncologist. 2007;12(11):1361-1370.
10. Mismetti P, Laporte-Simitisidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomized clinical trials. Thromb Haemost. 2000;83(1):14-19.
The Case
A 62-year-old woman with a past medical history significant for metastatic adenocarcinoma of the lung presents to the ED with complaints of fever and shortness of breath. She has recently completed her first cycle of carboplatin, pemetrexed, and bevacizumab. Upon admission, she is found to have an absolute neutrophil count of 800 and a platelet count of 48,000. She is admitted for neutropenic fever and placed on IV antimicrobials. Sequential compression devices are initiated for DVT prophylaxis.
Key Clinical Questions
What risk do cancer patients have for VTE?
Patients with cancer have a risk of clinically significant VTE that is four to seven times that of patients without malignancy.1 This is due to a number of reasons:
- Tumor cells produce procoagulant activity inducing thrombin formation;2
- The cancer itself can compress or invade deep veins; and3
- Some cancer therapies such L-asparaginase and thalidomide/lenalidomide, plus high-dose steroids, or anti-estrogen medications such as tamoxifen can also increase patients’ risk of VTE.3,4,5
What inpatients with cancer need VTE prophylaxis?
Much like other hospitalized medical patients, patients with cancer who have reduced mobility and are not on therapeutic anticoagulation should receive pharmacologic prophylaxis unless there is a contraindication.3,6,7,8 Cancer patients with acute medical illnesses should also likely receive prophylaxis if there are no contraindications, because the vast majority of these have factors increasing their VTE risk, including infection, kidney disease, or pulmonary disease.3,6,7,8 Patients undergoing major cancer surgery should also receive pharmacologic prophylaxis prior to surgery and for at least seven to 10 days post-operatively.3,6,7,8
For ambulatory cancer patients who are admitted for short courses of chemotherapy or for minor procedures, however, there is not enough evidence to recommend routine VTE prophylaxis.6,7 An exception to this is patients with multiple myeloma receiving thalidomide-based or lenalidomide-based chemotherapy, who should receive pharmacologic prophylaxis.6,7
What are the options available for VTE in hospitalized cancer patients?
The guidelines for VTE prophylaxis in hospitalized cancer patients recommend either unfractionated heparin (UFH) or low molecular weight heparin (LMWH) for prophylaxis when no contraindications exist.5 The only two LMWH that have been FDA approved for prophylaxis are enoxaparin and dalteparin. When deciding between UFH and LMWH, no evidence shows that one is better than the other in preventing VTE in hospitalized cancer patients.9 There is evidence that the use of LMWH results in a lower incidence of major hemorrhage when compared to UFH.10
What are the contraindications to pharmacologic VTE prophylaxis in cancer patients?
Contraindications for pharmacologic VTE prophylaxis in cancer patients include active major bleeding, thrombocytopenia (platelet count <50,000/µL), severe coagulopathy, inherited bleeding disorder, and at the time of surgery or invasive procedures (including lumbar puncture and epidural or spinal anesthesia).3,6,7 Those with contraindications to pharmacologic VTE prophylaxis should have mechanical prophylaxis instead.
What is the recommended treatment of VTE in cancer patients?
After the diagnosis of pulmonary embolism (PE) or DVT is found, LMWH is the preferred initial anticoagulant instead of UFH unless the patient has severe renal impairment (CrCl of less than 30 ml/min).6,7,8 LMWH is also preferred over warfarin for long-term anticoagulation during the initial six months of therapy.6,7,8 Following the initial six months, continued anticoagulation with either LMWH or warfarin could be considered in patients with active cancer, metastatic disease, or ongoing chemotherapy.6,7,8
When should IVC filters be considered in treating VTE in cancer patients?
IVC filter insertion should be reserved for those patients found to have a DVT or PE who have a contraindication to pharmacologic anticoagulation.3,6 It can be considered in patients who have recurrent VTE despite the appropriate use of optimally dosed LMWH therapy.6,8
What about the new oral anticoagulants?
At this point, because the majority of the major trials looking at the new oral anticoagulants (dabigatran, rivaroxaban, and apixaban) excluded cancer patients or included them only in small numbers, there is not enough evidence to support their use in cancer patients diagnosed with VTE.6,7,8
Back to the Case
On hospital day three, the patient is clinically improved. She is afebrile, her neutropenia has resolved, and her platelet count is up to 80,000. Her only complaint is pain and swelling of her left leg. A lower extremity Doppler is performed. She is found to have an acute left femoral DVT. The patient is then started on enoxaparin 1 mg/kg every 12 hours. Her left leg swelling and pain begin to improve, and she is discharged on enoxaparin and follows up with her oncologist in the next week. TH
Drs. Bell and O’Rourke are assistant professor of medicine in the division of hospital medicine at the University of California San Diego.
References
1. Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013;122(10):1712-1723.
2. Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293(6):715-722.
3. Streiff MB, Bockenstedt PL, Cataland SR, et al. Venous thromboembolic disease. J Natl Compr Canc Netw. 2013;11(11):1402-1429.
4. Payne JH, Vora AJ. Thrombosis and acute lymphoblastic leukaemia. Br J Haematol. 2007;138(4):430-445.
5. Amir E, Seruga B, Niraula S, Carlsson L, Ocaña A. Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst. 2011;103(17):1299-1309.
6. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(17):2189-2204.
7. Lyman GH, Bohlke K, Khorana AA, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: american society of clinical oncology clinical practice guideline update 2014. J Clin Oncol. 2015;33(6):654-656.
8. Farge D, Debourdeau P, Beckers M, et al. International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. J Thromb Haemost. 2013;11(1):56-70.
9. Khorana AA. The NCCN clinical practice guidelines on venous thromboembolic disease: strategies for improving VTE prophylaxis in hospitalized cancer patients. Oncologist. 2007;12(11):1361-1370.
10. Mismetti P, Laporte-Simitisidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomized clinical trials. Thromb Haemost. 2000;83(1):14-19.
The Case
A 62-year-old woman with a past medical history significant for metastatic adenocarcinoma of the lung presents to the ED with complaints of fever and shortness of breath. She has recently completed her first cycle of carboplatin, pemetrexed, and bevacizumab. Upon admission, she is found to have an absolute neutrophil count of 800 and a platelet count of 48,000. She is admitted for neutropenic fever and placed on IV antimicrobials. Sequential compression devices are initiated for DVT prophylaxis.
Key Clinical Questions
What risk do cancer patients have for VTE?
Patients with cancer have a risk of clinically significant VTE that is four to seven times that of patients without malignancy.1 This is due to a number of reasons:
- Tumor cells produce procoagulant activity inducing thrombin formation;2
- The cancer itself can compress or invade deep veins; and3
- Some cancer therapies such L-asparaginase and thalidomide/lenalidomide, plus high-dose steroids, or anti-estrogen medications such as tamoxifen can also increase patients’ risk of VTE.3,4,5
What inpatients with cancer need VTE prophylaxis?
Much like other hospitalized medical patients, patients with cancer who have reduced mobility and are not on therapeutic anticoagulation should receive pharmacologic prophylaxis unless there is a contraindication.3,6,7,8 Cancer patients with acute medical illnesses should also likely receive prophylaxis if there are no contraindications, because the vast majority of these have factors increasing their VTE risk, including infection, kidney disease, or pulmonary disease.3,6,7,8 Patients undergoing major cancer surgery should also receive pharmacologic prophylaxis prior to surgery and for at least seven to 10 days post-operatively.3,6,7,8
For ambulatory cancer patients who are admitted for short courses of chemotherapy or for minor procedures, however, there is not enough evidence to recommend routine VTE prophylaxis.6,7 An exception to this is patients with multiple myeloma receiving thalidomide-based or lenalidomide-based chemotherapy, who should receive pharmacologic prophylaxis.6,7
What are the options available for VTE in hospitalized cancer patients?
The guidelines for VTE prophylaxis in hospitalized cancer patients recommend either unfractionated heparin (UFH) or low molecular weight heparin (LMWH) for prophylaxis when no contraindications exist.5 The only two LMWH that have been FDA approved for prophylaxis are enoxaparin and dalteparin. When deciding between UFH and LMWH, no evidence shows that one is better than the other in preventing VTE in hospitalized cancer patients.9 There is evidence that the use of LMWH results in a lower incidence of major hemorrhage when compared to UFH.10
What are the contraindications to pharmacologic VTE prophylaxis in cancer patients?
Contraindications for pharmacologic VTE prophylaxis in cancer patients include active major bleeding, thrombocytopenia (platelet count <50,000/µL), severe coagulopathy, inherited bleeding disorder, and at the time of surgery or invasive procedures (including lumbar puncture and epidural or spinal anesthesia).3,6,7 Those with contraindications to pharmacologic VTE prophylaxis should have mechanical prophylaxis instead.
What is the recommended treatment of VTE in cancer patients?
After the diagnosis of pulmonary embolism (PE) or DVT is found, LMWH is the preferred initial anticoagulant instead of UFH unless the patient has severe renal impairment (CrCl of less than 30 ml/min).6,7,8 LMWH is also preferred over warfarin for long-term anticoagulation during the initial six months of therapy.6,7,8 Following the initial six months, continued anticoagulation with either LMWH or warfarin could be considered in patients with active cancer, metastatic disease, or ongoing chemotherapy.6,7,8
When should IVC filters be considered in treating VTE in cancer patients?
IVC filter insertion should be reserved for those patients found to have a DVT or PE who have a contraindication to pharmacologic anticoagulation.3,6 It can be considered in patients who have recurrent VTE despite the appropriate use of optimally dosed LMWH therapy.6,8
What about the new oral anticoagulants?
At this point, because the majority of the major trials looking at the new oral anticoagulants (dabigatran, rivaroxaban, and apixaban) excluded cancer patients or included them only in small numbers, there is not enough evidence to support their use in cancer patients diagnosed with VTE.6,7,8
Back to the Case
On hospital day three, the patient is clinically improved. She is afebrile, her neutropenia has resolved, and her platelet count is up to 80,000. Her only complaint is pain and swelling of her left leg. A lower extremity Doppler is performed. She is found to have an acute left femoral DVT. The patient is then started on enoxaparin 1 mg/kg every 12 hours. Her left leg swelling and pain begin to improve, and she is discharged on enoxaparin and follows up with her oncologist in the next week. TH
Drs. Bell and O’Rourke are assistant professor of medicine in the division of hospital medicine at the University of California San Diego.
References
1. Timp JF, Braekkan SK, Versteeg HH, Cannegieter SC. Epidemiology of cancer-associated venous thrombosis. Blood. 2013;122(10):1712-1723.
2. Blom JW, Doggen CJ, Osanto S, Rosendaal FR. Malignancies, prothrombotic mutations, and the risk of venous thrombosis. JAMA. 2005;293(6):715-722.
3. Streiff MB, Bockenstedt PL, Cataland SR, et al. Venous thromboembolic disease. J Natl Compr Canc Netw. 2013;11(11):1402-1429.
4. Payne JH, Vora AJ. Thrombosis and acute lymphoblastic leukaemia. Br J Haematol. 2007;138(4):430-445.
5. Amir E, Seruga B, Niraula S, Carlsson L, Ocaña A. Toxicity of adjuvant endocrine therapy in postmenopausal breast cancer patients: a systematic review and meta-analysis. J Natl Cancer Inst. 2011;103(17):1299-1309.
6. Lyman GH, Khorana AA, Kuderer NM, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: American Society of Clinical Oncology clinical practice guideline update. J Clin Oncol. 2013;31(17):2189-2204.
7. Lyman GH, Bohlke K, Khorana AA, et al. Venous thromboembolism prophylaxis and treatment in patients with cancer: american society of clinical oncology clinical practice guideline update 2014. J Clin Oncol. 2015;33(6):654-656.
8. Farge D, Debourdeau P, Beckers M, et al. International clinical practice guidelines for the treatment and prophylaxis of venous thromboembolism in patients with cancer. J Thromb Haemost. 2013;11(1):56-70.
9. Khorana AA. The NCCN clinical practice guidelines on venous thromboembolic disease: strategies for improving VTE prophylaxis in hospitalized cancer patients. Oncologist. 2007;12(11):1361-1370.
10. Mismetti P, Laporte-Simitisidis S, Tardy B, et al. Prevention of venous thromboembolism in internal medicine with unfractionated or low-molecular-weight heparins: a meta-analysis of randomized clinical trials. Thromb Haemost. 2000;83(1):14-19.
I am unworthy
The dream unfolds late in the semester with just a week to go, usually my final semester. My college career has been exemplary … good grades, honor society member, academic behavior any parent would be proud of. But for some reason I realize that I have failed to attend any of the classes of one of my courses, usually a math course. In fact, I’m not sure I have the text or maybe I never purchased it. More frighteningly, I can’t remember in which classroom it meets or even the hour. No one else seems to have noticed my failure to show up for class. Remember, it’s a math course and BSing doesn’t work in math. There is no way I will be able to resurrect myself from this academic disaster. The dream eventually dissolves without resolution, but it will return in some permutation, fortunately less often as I have gotten older. My wife and many of our friends share similar nightmares.
There are many angles from which one can interpret a dream like this, but one explanation is that I finally have been discovered as an impostor. I had studied hard, gotten good grades but at the core of things I was a goof-off and really wasn’t worthy of the adulation I had received. My good works were merely a shell over a life of not doing all the things that other people thought I had been doing.
It turns out that I had fallen into a surprisingly common psychological trap, probably during medical school. Despite accumulating significant amounts of clinical acumen, and in my later years what some might call wisdom, my dream suggests that I still have been unable to free myself of a nagging self-doubt. In 1978, two American psychologists Pauline Clance and Suzanne Imes labeled this phenomenon “the impostor syndrome” (“Learning to Deal with the Impostor Syndrome” by Carl Richards [The New York Times, Oct. 26, 2015]). They characterized it as a feeling “of phoniness in people who believe that they are not intelligent, capable, or creative despite evidence of high achievement.” The victims “are highly motivated to achieve” but “live in fear of being ‘found out’ or exposed as frauds.”
In college, I was in awe of those classmates who could play bridge for hours day after day, write their papers in the wee morning hours on the day they were due, and still get very acceptable grades. I imagined that if these guys had studied a third as much as I did or had simply begun their term papers on the day before they were due, their academic credentials would have blown mine out of the water.
In medical school I always had a sense that I didn’t belong there. I had never heard of anyone else who had gotten into an elite medical school off the waiting list as I had. There must have been a clerical error, and I had been mistaken for the scion of a wealthy benefactor with a similar sounding name. I had been around some smart people before, but my medical school classmates were in a different league altogether.
It turns out I was not alone bobbing in my sea of self-doubt. I learned from a blog entry on KevinMD.com (“The effect of impostor syndrome on medical students” by Aryeh Goldberg, March 1, 2014) of a lecture by Suzanne Poirier at Northwestern’s Feinberg School of Medicine, during which she reported on her analysis of more than forty book-length medical school memoirs. She discovered that a theme of a sense of not belonging ran through most (if not all) of the sources she reviewed. Other observers have wondered how much the impostor syndrome contributes to burnout, depression, and suicidal ideation in medical students.
I suffered from none of those maladies, but my feeling of unworthiness followed me into practice. Even as I acquired more experience during hundreds of thousands of patient encounters, I continued to worry that the next patient through the door would be the end of a decade’s long string of good fortune and my clinical ineptitude would be unmasked.
One of the most effective strategies for dealing with such feelings is sharing them. Unfortunately, most physicians don’t often find themselves in settings in which they can comfortably share these feelings with their peers. And of course it is probably not the best idea to share your self-doubts when you are trying to reassure a patient who is feeling vulnerable herself. Finding the balance between admitting that we don’t know everything and projecting the image that we know more than enough to help our patients is one of the biggest challenges facing us as we struggle to master the art of clinical medicine.
I will leave the question of whether I was an impostor to those who can be more objective. All I know is that I was damn lucky for 40 years.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
The dream unfolds late in the semester with just a week to go, usually my final semester. My college career has been exemplary … good grades, honor society member, academic behavior any parent would be proud of. But for some reason I realize that I have failed to attend any of the classes of one of my courses, usually a math course. In fact, I’m not sure I have the text or maybe I never purchased it. More frighteningly, I can’t remember in which classroom it meets or even the hour. No one else seems to have noticed my failure to show up for class. Remember, it’s a math course and BSing doesn’t work in math. There is no way I will be able to resurrect myself from this academic disaster. The dream eventually dissolves without resolution, but it will return in some permutation, fortunately less often as I have gotten older. My wife and many of our friends share similar nightmares.
There are many angles from which one can interpret a dream like this, but one explanation is that I finally have been discovered as an impostor. I had studied hard, gotten good grades but at the core of things I was a goof-off and really wasn’t worthy of the adulation I had received. My good works were merely a shell over a life of not doing all the things that other people thought I had been doing.
It turns out that I had fallen into a surprisingly common psychological trap, probably during medical school. Despite accumulating significant amounts of clinical acumen, and in my later years what some might call wisdom, my dream suggests that I still have been unable to free myself of a nagging self-doubt. In 1978, two American psychologists Pauline Clance and Suzanne Imes labeled this phenomenon “the impostor syndrome” (“Learning to Deal with the Impostor Syndrome” by Carl Richards [The New York Times, Oct. 26, 2015]). They characterized it as a feeling “of phoniness in people who believe that they are not intelligent, capable, or creative despite evidence of high achievement.” The victims “are highly motivated to achieve” but “live in fear of being ‘found out’ or exposed as frauds.”
In college, I was in awe of those classmates who could play bridge for hours day after day, write their papers in the wee morning hours on the day they were due, and still get very acceptable grades. I imagined that if these guys had studied a third as much as I did or had simply begun their term papers on the day before they were due, their academic credentials would have blown mine out of the water.
In medical school I always had a sense that I didn’t belong there. I had never heard of anyone else who had gotten into an elite medical school off the waiting list as I had. There must have been a clerical error, and I had been mistaken for the scion of a wealthy benefactor with a similar sounding name. I had been around some smart people before, but my medical school classmates were in a different league altogether.
It turns out I was not alone bobbing in my sea of self-doubt. I learned from a blog entry on KevinMD.com (“The effect of impostor syndrome on medical students” by Aryeh Goldberg, March 1, 2014) of a lecture by Suzanne Poirier at Northwestern’s Feinberg School of Medicine, during which she reported on her analysis of more than forty book-length medical school memoirs. She discovered that a theme of a sense of not belonging ran through most (if not all) of the sources she reviewed. Other observers have wondered how much the impostor syndrome contributes to burnout, depression, and suicidal ideation in medical students.
I suffered from none of those maladies, but my feeling of unworthiness followed me into practice. Even as I acquired more experience during hundreds of thousands of patient encounters, I continued to worry that the next patient through the door would be the end of a decade’s long string of good fortune and my clinical ineptitude would be unmasked.
One of the most effective strategies for dealing with such feelings is sharing them. Unfortunately, most physicians don’t often find themselves in settings in which they can comfortably share these feelings with their peers. And of course it is probably not the best idea to share your self-doubts when you are trying to reassure a patient who is feeling vulnerable herself. Finding the balance between admitting that we don’t know everything and projecting the image that we know more than enough to help our patients is one of the biggest challenges facing us as we struggle to master the art of clinical medicine.
I will leave the question of whether I was an impostor to those who can be more objective. All I know is that I was damn lucky for 40 years.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
The dream unfolds late in the semester with just a week to go, usually my final semester. My college career has been exemplary … good grades, honor society member, academic behavior any parent would be proud of. But for some reason I realize that I have failed to attend any of the classes of one of my courses, usually a math course. In fact, I’m not sure I have the text or maybe I never purchased it. More frighteningly, I can’t remember in which classroom it meets or even the hour. No one else seems to have noticed my failure to show up for class. Remember, it’s a math course and BSing doesn’t work in math. There is no way I will be able to resurrect myself from this academic disaster. The dream eventually dissolves without resolution, but it will return in some permutation, fortunately less often as I have gotten older. My wife and many of our friends share similar nightmares.
There are many angles from which one can interpret a dream like this, but one explanation is that I finally have been discovered as an impostor. I had studied hard, gotten good grades but at the core of things I was a goof-off and really wasn’t worthy of the adulation I had received. My good works were merely a shell over a life of not doing all the things that other people thought I had been doing.
It turns out that I had fallen into a surprisingly common psychological trap, probably during medical school. Despite accumulating significant amounts of clinical acumen, and in my later years what some might call wisdom, my dream suggests that I still have been unable to free myself of a nagging self-doubt. In 1978, two American psychologists Pauline Clance and Suzanne Imes labeled this phenomenon “the impostor syndrome” (“Learning to Deal with the Impostor Syndrome” by Carl Richards [The New York Times, Oct. 26, 2015]). They characterized it as a feeling “of phoniness in people who believe that they are not intelligent, capable, or creative despite evidence of high achievement.” The victims “are highly motivated to achieve” but “live in fear of being ‘found out’ or exposed as frauds.”
In college, I was in awe of those classmates who could play bridge for hours day after day, write their papers in the wee morning hours on the day they were due, and still get very acceptable grades. I imagined that if these guys had studied a third as much as I did or had simply begun their term papers on the day before they were due, their academic credentials would have blown mine out of the water.
In medical school I always had a sense that I didn’t belong there. I had never heard of anyone else who had gotten into an elite medical school off the waiting list as I had. There must have been a clerical error, and I had been mistaken for the scion of a wealthy benefactor with a similar sounding name. I had been around some smart people before, but my medical school classmates were in a different league altogether.
It turns out I was not alone bobbing in my sea of self-doubt. I learned from a blog entry on KevinMD.com (“The effect of impostor syndrome on medical students” by Aryeh Goldberg, March 1, 2014) of a lecture by Suzanne Poirier at Northwestern’s Feinberg School of Medicine, during which she reported on her analysis of more than forty book-length medical school memoirs. She discovered that a theme of a sense of not belonging ran through most (if not all) of the sources she reviewed. Other observers have wondered how much the impostor syndrome contributes to burnout, depression, and suicidal ideation in medical students.
I suffered from none of those maladies, but my feeling of unworthiness followed me into practice. Even as I acquired more experience during hundreds of thousands of patient encounters, I continued to worry that the next patient through the door would be the end of a decade’s long string of good fortune and my clinical ineptitude would be unmasked.
One of the most effective strategies for dealing with such feelings is sharing them. Unfortunately, most physicians don’t often find themselves in settings in which they can comfortably share these feelings with their peers. And of course it is probably not the best idea to share your self-doubts when you are trying to reassure a patient who is feeling vulnerable herself. Finding the balance between admitting that we don’t know everything and projecting the image that we know more than enough to help our patients is one of the biggest challenges facing us as we struggle to master the art of clinical medicine.
I will leave the question of whether I was an impostor to those who can be more objective. All I know is that I was damn lucky for 40 years.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
Save your breath
If you haven’t already stopped investing your limited supply of office time in fruitless attempts to convince vaccine-hesitant parents to immunize their children, a small study at the University of North Carolina Women’s Hospital in Chapel Hill might finally convince you it’s time to save your breath for other more achievable goals. In a survey of 171 parents, 72% reported that they already had settled on their vaccine preferences prior to pregnancy.
This was a limited survey and may not reflect the responses of a national sample of parents, but it is concerning in light of several other studies that paint a similar gloomy picture. One such study found that even when vaccine-denying parents were presented with educational materials that they acknowledged seemed valid, they continued to withhold vaccines by falling back on other arguments to support their views (“Effective Messages in Vaccine Promotion: A Randomized Trial” by Nyhan et al. [Pediatrics. 2014 Apr;133(4):e835-42]).
If a larger and more geographically diverse study continues to find that the die is cast well before pediatricians have gotten our chance to discuss vaccines with parents-to-be, we will need to rethink our strategies for dealing with vaccine refusers. For those pediatricians who already ask vaccine decliners to find another practice, this new study suggests that they could save themselves time and trouble by advertising their policy to the obstetricians in their communities. This proactive advertising would require some courage, but in the long run it probably makes economic sense.
However, for most pediatricians it may be better to wait in hopes that future research can determine exactly when and under what circumstances most vaccine decliners arrive at their unfortunate decisions. How often was it a philosophy that they inherited from their parents? How often did it reflect their religious views? How often did it evolve from something they heard in school? Junior high, high school, college? Was it a science class, or history, or philosophy?
Was it the result of some media story? TV? Print? Internet? If they can recall a particular show or website, what was it that made it sound so convincing? If it was an individual, was it a friend, celebrity, or a teacher?
A study this detailed would be time consuming and labor intensive, as it would be best done in face-to-face structured interviews by someone who could project a nonjudgmental aura. It would necessarily be retrospective. But it might yield some surprising and helpful information that could be used to target our attack on the epidemic of vaccine refusal.
We know that outbreaks of certain infectious diseases, smallpox being the prime example, do not respond to media blitzes and immunization campaigns. Epidemics will continue to roll along unchecked until a labor-intensive, boots-on-the-ground, door-to-door case-finding effort is undertaken. Vaccine refusal may be similar to smallpox. It appears to be unresponsive to mass media and educational initiatives. It may continue to plague us until we chase down its roots.
Whatever strategy we try next, it is clear that although most parents report that they consider pediatricians among their most trusted sources of health information for their children, we are failing to reach a segment of our target audience. We are too late, long after the die is cast.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
*This story was updated 1/28/2016.
If you haven’t already stopped investing your limited supply of office time in fruitless attempts to convince vaccine-hesitant parents to immunize their children, a small study at the University of North Carolina Women’s Hospital in Chapel Hill might finally convince you it’s time to save your breath for other more achievable goals. In a survey of 171 parents, 72% reported that they already had settled on their vaccine preferences prior to pregnancy.
This was a limited survey and may not reflect the responses of a national sample of parents, but it is concerning in light of several other studies that paint a similar gloomy picture. One such study found that even when vaccine-denying parents were presented with educational materials that they acknowledged seemed valid, they continued to withhold vaccines by falling back on other arguments to support their views (“Effective Messages in Vaccine Promotion: A Randomized Trial” by Nyhan et al. [Pediatrics. 2014 Apr;133(4):e835-42]).
If a larger and more geographically diverse study continues to find that the die is cast well before pediatricians have gotten our chance to discuss vaccines with parents-to-be, we will need to rethink our strategies for dealing with vaccine refusers. For those pediatricians who already ask vaccine decliners to find another practice, this new study suggests that they could save themselves time and trouble by advertising their policy to the obstetricians in their communities. This proactive advertising would require some courage, but in the long run it probably makes economic sense.
However, for most pediatricians it may be better to wait in hopes that future research can determine exactly when and under what circumstances most vaccine decliners arrive at their unfortunate decisions. How often was it a philosophy that they inherited from their parents? How often did it reflect their religious views? How often did it evolve from something they heard in school? Junior high, high school, college? Was it a science class, or history, or philosophy?
Was it the result of some media story? TV? Print? Internet? If they can recall a particular show or website, what was it that made it sound so convincing? If it was an individual, was it a friend, celebrity, or a teacher?
A study this detailed would be time consuming and labor intensive, as it would be best done in face-to-face structured interviews by someone who could project a nonjudgmental aura. It would necessarily be retrospective. But it might yield some surprising and helpful information that could be used to target our attack on the epidemic of vaccine refusal.
We know that outbreaks of certain infectious diseases, smallpox being the prime example, do not respond to media blitzes and immunization campaigns. Epidemics will continue to roll along unchecked until a labor-intensive, boots-on-the-ground, door-to-door case-finding effort is undertaken. Vaccine refusal may be similar to smallpox. It appears to be unresponsive to mass media and educational initiatives. It may continue to plague us until we chase down its roots.
Whatever strategy we try next, it is clear that although most parents report that they consider pediatricians among their most trusted sources of health information for their children, we are failing to reach a segment of our target audience. We are too late, long after the die is cast.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
*This story was updated 1/28/2016.
If you haven’t already stopped investing your limited supply of office time in fruitless attempts to convince vaccine-hesitant parents to immunize their children, a small study at the University of North Carolina Women’s Hospital in Chapel Hill might finally convince you it’s time to save your breath for other more achievable goals. In a survey of 171 parents, 72% reported that they already had settled on their vaccine preferences prior to pregnancy.
This was a limited survey and may not reflect the responses of a national sample of parents, but it is concerning in light of several other studies that paint a similar gloomy picture. One such study found that even when vaccine-denying parents were presented with educational materials that they acknowledged seemed valid, they continued to withhold vaccines by falling back on other arguments to support their views (“Effective Messages in Vaccine Promotion: A Randomized Trial” by Nyhan et al. [Pediatrics. 2014 Apr;133(4):e835-42]).
If a larger and more geographically diverse study continues to find that the die is cast well before pediatricians have gotten our chance to discuss vaccines with parents-to-be, we will need to rethink our strategies for dealing with vaccine refusers. For those pediatricians who already ask vaccine decliners to find another practice, this new study suggests that they could save themselves time and trouble by advertising their policy to the obstetricians in their communities. This proactive advertising would require some courage, but in the long run it probably makes economic sense.
However, for most pediatricians it may be better to wait in hopes that future research can determine exactly when and under what circumstances most vaccine decliners arrive at their unfortunate decisions. How often was it a philosophy that they inherited from their parents? How often did it reflect their religious views? How often did it evolve from something they heard in school? Junior high, high school, college? Was it a science class, or history, or philosophy?
Was it the result of some media story? TV? Print? Internet? If they can recall a particular show or website, what was it that made it sound so convincing? If it was an individual, was it a friend, celebrity, or a teacher?
A study this detailed would be time consuming and labor intensive, as it would be best done in face-to-face structured interviews by someone who could project a nonjudgmental aura. It would necessarily be retrospective. But it might yield some surprising and helpful information that could be used to target our attack on the epidemic of vaccine refusal.
We know that outbreaks of certain infectious diseases, smallpox being the prime example, do not respond to media blitzes and immunization campaigns. Epidemics will continue to roll along unchecked until a labor-intensive, boots-on-the-ground, door-to-door case-finding effort is undertaken. Vaccine refusal may be similar to smallpox. It appears to be unresponsive to mass media and educational initiatives. It may continue to plague us until we chase down its roots.
Whatever strategy we try next, it is clear that although most parents report that they consider pediatricians among their most trusted sources of health information for their children, we are failing to reach a segment of our target audience. We are too late, long after the die is cast.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics including “How to Say No to Your Toddler.”
*This story was updated 1/28/2016.
Diagnosis and Management of Cold Urticaria
Cold urticaria is a rare condition characterized by a localized or systemic eruption of papules upon exposure of the skin to cold air, liquids, and/or objects. In some cases, angioedema and anaphylaxis can occur. The wheal-and-flare reaction results from a localized or systemic release of histamine, leukotrienes, and various other proinflammatory mast cell mediators. Cold urticaria can be acquired or follow an autosomal-dominant familial transmission pattern. Acquired cold urticaria often presents in young adulthood with a mean duration of 4 to 5 years and remission or improvement of symptoms after 5 years in 50% of cases.1 The familial variant most commonly presents in early childhood and endures throughout the patient’s life.2 Cold urticaria generally is classified as acute or chronic if symptoms persist for more than 6 weeks. Pharmacologic therapies with prophylactic effects that may reduce the intensity of symptoms or inhibit their development include antihistamines, leuko-triene receptor antagonists, biologics, and glucocorticoids. We present the case of a 23-year-old man with cold urticaria that was refractory to initial treatment with H1 antihistamines along with a review of the literature.
Case Report
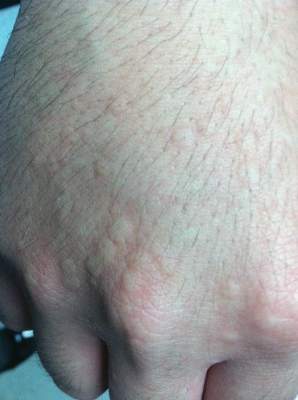
A 23-year-old man presented to the dermatology clinic for evaluation of recurrent burning, itching, and sometimes development of a painful rash on the face, neck, and arms of 2 years’ duration that typically occurred following exposure to cold, wind, and rain. He also developed symptoms in warm weather when exposed to wind while sweating. His medical history was remarkable for asthma, which was not active. He was not taking any medications and had no known drug or environmental allergies. No other members of his household developed similar symptoms. His only successful means of prevention was to stay indoors, which thereby limited his activities.
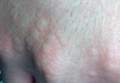
Physical examination of the dorsal hands following an ice cube test revealed numerous 3- to 5-mm urticarial papules with surrounding erythema (Figure).
Following the initial evaluation, the patient was treated unsuccessfully with a mix of first- and second-generation antihistamines in gradually increasing doses to a maximum dose of loratadine 20 mg once daily, cetirizine 20 mg once daily, and hydroxyzine 20 mg once daily. A course of montelukast 10 mg once daily was started in addition to the antihistamines and led to a reduction in the severity of the lesions but not the frequency and did not relieve the burning sensation; the patient subsequently discontinued therapy. Next, a trial of cyclosporine was attempted, but the patient reported that it caused emesis and subsequently discontinued treatment. The patient also did not tolerate prednisone. He eventually decided to treat his symptoms with lifestyle choices only, such as making sure to be well covered in cold temperatures.
Comment
Cold urticaria is a physical urticaria resulting from mast cell degranulation and the subsequent release of histamine and proinflammatory cytokines upon exposure of the skin to cold air, liquid, and/or objects. Symtpoms usually are limited to localized exposed areas of the skin but also can be generalized. Cold urticaria typically manifests as erythematous, pruritic papules and also may be accompanied by deep tissue involvement resulting in angioedema and/or anaphylaxis. Symptoms usually occur within minutes of cold exposure; however, in delayed-type cold urticaria, symptoms may develop 24 to 72 hours later.3 Prevalence is relatively equal in both sexes and is highest among young adults (ie, 18–27 years old), with a greater incidence associated with cold climates.4 In one study, the overall incidence of acquired cold urticaria in Central Europe was estimated to be 0.05%.1
Systemic involvement may occur with extensive cold contact, ranging in severity from generalized urticaria to anaphylaxis and involvement of the cardiovascular, respiratory, and/or gastrointestinal systems.5 Patients who exhibit systemic responses to cold exposure should avoid swimming in cold water, as this may induce anaphylaxis and result in injury or death. In a 2004 study that included 30 children with cold urticaria at a tertiary center in Boston,6 11 (36.7%) participants who underwent cold stimulation testing developed systemic symptoms; 5 (45.5%) participants experienced respiratory distress and 8 (72.7%) experienced a decrease in level of consciousness (eg, faintness, dizziness, hypotension). Aquatic activity was the trigger in all 11 participants except for 1 (9.0%), who experienced systemic symptoms on exposure to cold air. In the same study, 14 (46.7%) participants were diagnosed with asthma and 15 (50%) were diagnosed with allergic rhinitis. Of the 28 participants whose family histories were available for review, 25 (89.3%) had a family history of atopic disease.6 A 2008 Greek study4 of 62 adults with acquired cold urticaria found that 18 (29%) participants had at least 1 serious systemic response resulting in generalized urticaria or angioedema associated with hypotension (eg, dizziness, fainting, disorientation, shock). In both of these studies, a majority of the serious systemic reactions were associated with cold water activities.
Cold urticaria is primarily an idiopathic phenomenon but can be classified as acquired or familial. Acquired cold urticaria may result from primary or secondary causes, which can include cryoglobulinemia, human immunodeficiency virus, syphilis, mononucleosis, rubeola, toxoplasmosis, varicella, hepatitis, and various drugs (eg, penicillin, angiotensin-converting enzyme inhibitors, oral contraceptives).7 Familial causes include cryopyrin-associated periodic syndrome, phospholipase Cγ2 gene–associated antibody deficiency and immune dysregulation, Muckle-Wells syndrome, and neonatal-onset multisystem inflammatory disease.
Typically, cold urticaria is diagnosed using cold stimulation tests such as the ice cube test, in which an ice cube is applied directly to the patient’s skin for 3 to 5 minutes and a response is measured 10 minutes after its removal.8 This test has been shown to have a sensitivity of 83% to 90% and a specificity of 100%.9 Alternatively, cold urticaria may be diagnosed through the use of a Peltier element-based cold-provocation device, which exposes the patient to a variety of temperatures in order for clinicians to determine the threshold upon which there is an observable reaction. With a sensitivity of 93% and specificity of 100%, the accuracy of this test is similar to that of the ice cube test.10 If a patient has a history of serious systemic involvement, any testing that exposes the patient to extensive cold exposure should be used with caution.
Patients should be counseled about potential serious systemic symptoms and the importance of wearing appropriate cold-weather clothing. Avoidance of cold water activities and overexposure to cold weather also should be emphasized. Pharmacologic therapy for prophylaxis typically includes a second-generation H1 antihistamine (eg, cetirizine, loratadine, desloratadine). Since these drugs have been shown to be less sedating than first-generation antihistamines, they are considered a better choice for chronic treatment. At high doses, however, these medications may have a sedative effect; therefore, nighttime use is preferable if possible. The standard dosage is 5 mg to 10 mg daily for oral cetirizine, 10 mg daily for oral loratadine, and 5 mg daily for oral desloratadine; however, up to 4 times the standard dosage of these medications may be required for effective treatment of cold urticaria.11 Given the associated risk of anaphylaxis, patients should be prescribed an epinephrine pen and educated about its appropriate use, including the importance of keeping the pen accessible at all times.
In refractory cases of cold urticaria, an H2 antihistamine (eg, ranitidine) can be used in conjunction with H1 antihistamines.12 Omalizumab, an IgE-mediated treatment, also has been shown to be safe and effective in patients with recalcitrant physical urticaria, including cold urticaria.13,14 One report described the case of a 69-year-old woman with cold urticaria who was unable to leave the house without developing a widespread eruption on the face, trunk, and limbs.15 After undergoing a series of unsuccessful treatments, the patient was started on cyclosporine 125 mg twice daily, which was reduced to 100 mg twice daily after 4 weeks of therapy and then reduced to 75 mg twice daily after 4 months of treatment. One week after therapy was initiated the patient reported that she was able to leave the house, and after 4 weeks of treatment the lesions only developed on the hands and feet. The patient remained in remission with a low-dose therapy of cyclosporine 75 mg twice daily with lesions only occurring on the hands and feet. The low-dose maintenance therapy was associated with minimal adverse effects.15 To our knowledge, there are no known large studies on the efficacy of cyclosporine in the treatment of cold urticaria.
Leukotriene receptor antagonists (eg, montelukast, zafirlukast, zileuton) have been used to treat chronic urticaria. In one report, montelukast was used in a 29-year-old woman with cold urticaria who had initially been treated with cetirizine 30 mg daily, cyproheptadine 4 mg daily, and doxycycline 200 mg daily with minimal to no relief. After treatment with montelukast, she experienced notable and stable improvements in symptoms.16 Hydroxychloroquine also has been shown to be safe and to substantially improve quality of life in patients with idiopathic chronic urticaria.17 Methotrexate (with close patient monitoring for adverse effects) has been reported to benefit some patients whose chronic urticaria was unresponsive to standard treatment.18 Treatment regimens for chronic urticaria have shown variable success in the treatment of cold urticaria and may be considered in cases refractory to treatment with high-dose second-generation H1 antihistamines.
Topical application of capsaicin for 4 to 7 days has been shown to deplete the neuropeptides in sensory fibers that may be involved in cold reactions, although skin irritation may prevent usage.19
Prednisone therapy was used in a small study of 6 patients with acquired cold urticaria.20 Three patients were treated for periods of 3 to 5 days with prednisone 20 mg each morning. Three other patients were given a single dose of prednisone 20 mg or 25 mg in the morning, depending on body weight. Following prednisone therapy, complete or partial pruritus was subjectively improved in all 6 patients. Additionally, significant reductions in venous histamine concentrations at 5 and 10 minutes following cold immersion were noted (P<.05 and P<.025, respectively); however, no significant improvement in either erythema or edema was noted posttreatment following cold immersion.20 Despite these findings, prednisone has not been shown to consistently prevent histamine release. Another report noted the case of a 47-year-old man with cold urticaria who required hypothermic cardiopulmonary bypass. Pretreatment with prednisone 20 mg daily and preoperative hydrocortisone 100 mg intravenously did not prevent histamine release.21
Cold desensitization (ie, exposing progressively larger areas of the patient’s skin to increasingly colder water) may induce tolerance to cold and decrease the temperature threshold at which symptoms develop; however, patients with known serious systemic reactions should be tested with extreme caution and only under the supervision of a clinician.22,23 Tolerance may wane when cold desensitization therapy is stopped.
The prognosis for patients with acquired cold urticaria generally is good. Improvement of symptoms or full remission occurs within 5 to 6 years in 50% of patients.24 Once remission has occurred, patients generally remain symptom free. For other familial variants, symptoms may last a lifetime.
Conclusion
This case report and review of the literature highlights the limitations of cold urticaria and the importance of effective management in improving quality of life in affected patients. Symptoms may limit patients’ ability to work in certain environments, inhibit them from engaging in daily activities, and even prevent them from leaving their homes in colder temperatures. In addition to behavioral modifications, pharmacologic management may provide symptomatic relief. Antihistamines are the first line of treatment in cold urticaria. Second-generation antihistamines, which are more selective for H1 receptors and less sedating, are generally recommended. Up to 4 times the standard dosage of these medications may be required for effective treatment.5 The primary goal of therapy in mild to moderate cases is improvement in quality of life.
- Siebenhaar F, Weller K, Mlynek A, et al. Acquired cold urticaria: clinical picture and update on diagnosis and treatment. Clin Exp Dermatol. 2007;32:241-245.
- Gandhi C, Healy C, Wanderer AA, et al. Familial atypical cold urticaria: description of a new hereditary disease. J Allergy Clin Immunol. 2009;124:1245-1250.
- Bäck O, Larsen A. Delayed cold urticaria. Acta Derm Venereol. 1978;58:369-371.
- Katsarou-Katsari A, Makris M, Lagogianni E, et al. Clinical features and natural history of acquired cold urticaria in a tertiary referral hospital: a 10-year prospective study. J Eur Acad Dermatol Venereol. 2008;22:1405-1411.
- Wanderer AA, Grandel KE, Wasserman SI, et al. Clinical characteristics of cold-induced systemic reactions in acquired cold urticaria syndromes: recommendations for prevention of this complication and a proposal for a diagnostic classification of cold urticaria. J Allergy Clin Immunol. 1986;78(3 Pt 1):417-423.
- Alangari AA, Twarog FJ, Shih MC, et al. Clinical features and anaphylaxis in children with cold urticaria. Pediatrics. 2004;113:e313-e317.
- Wanderer AA, Hoffman HM. The spectrum of acquired and familial cold-induced urticaria/urticaria-like syndromes. Immunol Allergy Clin North Am. 2004;24:259-286.
- Visitsuntorn N, Tuchinda M, Arunyanark N, et al. Ice cube test in children with cold urticaria. Asian Pac J Allergy Immunol. 1992;10:111-115.
- Neittaanmäki H. Cold urticaria. clinical findings in 220 patients. J Am Acad Dermatol. 1985;13:636-644.
- Siebenhaar F, Staubach P, Metz M, et al. Peltier effect-based temperature challenge: an improved method for diagnosing cold urticaria. J Allergy Clin Immunol. 2004;114:1224-1225.
- Siebenhaar F, Degener F, Zuberbier T, et al. High-dose desloratadine decreases wheal volume and improves cold provocation thresholds compared with standard-dose treatment in patients with acquired cold urticaria: a randomized, placebo-controlled, crossover study. J Allergy Clin Immunol. 2009;123:672-679.
- Duc J, Pécoud A. Successful treatment of idiopathic cold urticaria with the association of H1 and H2 antagonists: a case report. Ann Allergy. 1986;56:355-357.
- Metz M, Altrichter S, Ardelean E, et al. Anti-immunoglobulin E treatment of patients with recalcitrant physical urticaria. Int Arch Allergy Immunol. 2011;154:177-180.
- Boyce JA. Successful treatment of cold-induced urticaria/anaphylaxis with anti-IgE. J Allergy Clin Immunol. 2006;117:1415-1418.
- Marsland AM, Beck MH. Cold urticaria responding to systemic cyclosporine. Br J Dermatol. 2003;149:214-215.
- Hani N, Hartmann K, Casper C, et al. Improvement of cold urticaria by treatment with the leukotriene receptor antagonist montelukast. Acta Derm Venereol. 2000;80:229.
- Reeves GE, Boyle MJ, Bonfield J, et al. Impact of hydroxychloroquine therapy on chronic urticaria: chronic autoimmune urticaria study and evaluation. Intern Med J. 2004;34:182-186.
- Perez A, Woods A, Grattan CE. Methotrexate: a useful steroid-sparing agent in recalcitrant chronic urticaria. Br J Dermatol. 2010;162:191-194.
- Tóth-Kása I, Jancsó G, Obál F Jr, et al. Involvement of sensory nerve endings in cold and heat urticaria. J Invest Dermatol. 1983;80:34-36.
- Black AK, Keahey TM, Eady RA, et al. Dissociation of histamine release and clinical improvement following treatment of acquired cold urticaria by prednisone. Br J Clin Pharmacol. 1981;12:327-331.
- Johnston WE, Moss J, Philbin DM, et al. Management of cold urticaria during hypothermic cardiopulmonary bypass. N Engl J Med. 1982;306:219-221.
- Krause K, Zuberbier T, Maurer, M. Modern Approaches to the diagnosis and treatment of cold contact urticaria. Curr Allergy Asthma Rep. 2010;10:273-279.
- von Mackensen YA, Sticherling M. Cold urticaria: tolerance induction with cold baths. Br J Dermatol. 2007;157:835-836.
- Möller A, Henning M, Zuberbier T, et al. Epidemiology and clinical aspects of cold urticaria [article in German]. Hautarzt. 1996;47:510-514.
Cold urticaria is a rare condition characterized by a localized or systemic eruption of papules upon exposure of the skin to cold air, liquids, and/or objects. In some cases, angioedema and anaphylaxis can occur. The wheal-and-flare reaction results from a localized or systemic release of histamine, leukotrienes, and various other proinflammatory mast cell mediators. Cold urticaria can be acquired or follow an autosomal-dominant familial transmission pattern. Acquired cold urticaria often presents in young adulthood with a mean duration of 4 to 5 years and remission or improvement of symptoms after 5 years in 50% of cases.1 The familial variant most commonly presents in early childhood and endures throughout the patient’s life.2 Cold urticaria generally is classified as acute or chronic if symptoms persist for more than 6 weeks. Pharmacologic therapies with prophylactic effects that may reduce the intensity of symptoms or inhibit their development include antihistamines, leuko-triene receptor antagonists, biologics, and glucocorticoids. We present the case of a 23-year-old man with cold urticaria that was refractory to initial treatment with H1 antihistamines along with a review of the literature.
Case Report
A 23-year-old man presented to the dermatology clinic for evaluation of recurrent burning, itching, and sometimes development of a painful rash on the face, neck, and arms of 2 years’ duration that typically occurred following exposure to cold, wind, and rain. He also developed symptoms in warm weather when exposed to wind while sweating. His medical history was remarkable for asthma, which was not active. He was not taking any medications and had no known drug or environmental allergies. No other members of his household developed similar symptoms. His only successful means of prevention was to stay indoors, which thereby limited his activities.
Physical examination of the dorsal hands following an ice cube test revealed numerous 3- to 5-mm urticarial papules with surrounding erythema (Figure).

Following the initial evaluation, the patient was treated unsuccessfully with a mix of first- and second-generation antihistamines in gradually increasing doses to a maximum dose of loratadine 20 mg once daily, cetirizine 20 mg once daily, and hydroxyzine 20 mg once daily. A course of montelukast 10 mg once daily was started in addition to the antihistamines and led to a reduction in the severity of the lesions but not the frequency and did not relieve the burning sensation; the patient subsequently discontinued therapy. Next, a trial of cyclosporine was attempted, but the patient reported that it caused emesis and subsequently discontinued treatment. The patient also did not tolerate prednisone. He eventually decided to treat his symptoms with lifestyle choices only, such as making sure to be well covered in cold temperatures.
Comment
Cold urticaria is a physical urticaria resulting from mast cell degranulation and the subsequent release of histamine and proinflammatory cytokines upon exposure of the skin to cold air, liquid, and/or objects. Symtpoms usually are limited to localized exposed areas of the skin but also can be generalized. Cold urticaria typically manifests as erythematous, pruritic papules and also may be accompanied by deep tissue involvement resulting in angioedema and/or anaphylaxis. Symptoms usually occur within minutes of cold exposure; however, in delayed-type cold urticaria, symptoms may develop 24 to 72 hours later.3 Prevalence is relatively equal in both sexes and is highest among young adults (ie, 18–27 years old), with a greater incidence associated with cold climates.4 In one study, the overall incidence of acquired cold urticaria in Central Europe was estimated to be 0.05%.1
Systemic involvement may occur with extensive cold contact, ranging in severity from generalized urticaria to anaphylaxis and involvement of the cardiovascular, respiratory, and/or gastrointestinal systems.5 Patients who exhibit systemic responses to cold exposure should avoid swimming in cold water, as this may induce anaphylaxis and result in injury or death. In a 2004 study that included 30 children with cold urticaria at a tertiary center in Boston,6 11 (36.7%) participants who underwent cold stimulation testing developed systemic symptoms; 5 (45.5%) participants experienced respiratory distress and 8 (72.7%) experienced a decrease in level of consciousness (eg, faintness, dizziness, hypotension). Aquatic activity was the trigger in all 11 participants except for 1 (9.0%), who experienced systemic symptoms on exposure to cold air. In the same study, 14 (46.7%) participants were diagnosed with asthma and 15 (50%) were diagnosed with allergic rhinitis. Of the 28 participants whose family histories were available for review, 25 (89.3%) had a family history of atopic disease.6 A 2008 Greek study4 of 62 adults with acquired cold urticaria found that 18 (29%) participants had at least 1 serious systemic response resulting in generalized urticaria or angioedema associated with hypotension (eg, dizziness, fainting, disorientation, shock). In both of these studies, a majority of the serious systemic reactions were associated with cold water activities.
Cold urticaria is primarily an idiopathic phenomenon but can be classified as acquired or familial. Acquired cold urticaria may result from primary or secondary causes, which can include cryoglobulinemia, human immunodeficiency virus, syphilis, mononucleosis, rubeola, toxoplasmosis, varicella, hepatitis, and various drugs (eg, penicillin, angiotensin-converting enzyme inhibitors, oral contraceptives).7 Familial causes include cryopyrin-associated periodic syndrome, phospholipase Cγ2 gene–associated antibody deficiency and immune dysregulation, Muckle-Wells syndrome, and neonatal-onset multisystem inflammatory disease.
Typically, cold urticaria is diagnosed using cold stimulation tests such as the ice cube test, in which an ice cube is applied directly to the patient’s skin for 3 to 5 minutes and a response is measured 10 minutes after its removal.8 This test has been shown to have a sensitivity of 83% to 90% and a specificity of 100%.9 Alternatively, cold urticaria may be diagnosed through the use of a Peltier element-based cold-provocation device, which exposes the patient to a variety of temperatures in order for clinicians to determine the threshold upon which there is an observable reaction. With a sensitivity of 93% and specificity of 100%, the accuracy of this test is similar to that of the ice cube test.10 If a patient has a history of serious systemic involvement, any testing that exposes the patient to extensive cold exposure should be used with caution.
Patients should be counseled about potential serious systemic symptoms and the importance of wearing appropriate cold-weather clothing. Avoidance of cold water activities and overexposure to cold weather also should be emphasized. Pharmacologic therapy for prophylaxis typically includes a second-generation H1 antihistamine (eg, cetirizine, loratadine, desloratadine). Since these drugs have been shown to be less sedating than first-generation antihistamines, they are considered a better choice for chronic treatment. At high doses, however, these medications may have a sedative effect; therefore, nighttime use is preferable if possible. The standard dosage is 5 mg to 10 mg daily for oral cetirizine, 10 mg daily for oral loratadine, and 5 mg daily for oral desloratadine; however, up to 4 times the standard dosage of these medications may be required for effective treatment of cold urticaria.11 Given the associated risk of anaphylaxis, patients should be prescribed an epinephrine pen and educated about its appropriate use, including the importance of keeping the pen accessible at all times.
In refractory cases of cold urticaria, an H2 antihistamine (eg, ranitidine) can be used in conjunction with H1 antihistamines.12 Omalizumab, an IgE-mediated treatment, also has been shown to be safe and effective in patients with recalcitrant physical urticaria, including cold urticaria.13,14 One report described the case of a 69-year-old woman with cold urticaria who was unable to leave the house without developing a widespread eruption on the face, trunk, and limbs.15 After undergoing a series of unsuccessful treatments, the patient was started on cyclosporine 125 mg twice daily, which was reduced to 100 mg twice daily after 4 weeks of therapy and then reduced to 75 mg twice daily after 4 months of treatment. One week after therapy was initiated the patient reported that she was able to leave the house, and after 4 weeks of treatment the lesions only developed on the hands and feet. The patient remained in remission with a low-dose therapy of cyclosporine 75 mg twice daily with lesions only occurring on the hands and feet. The low-dose maintenance therapy was associated with minimal adverse effects.15 To our knowledge, there are no known large studies on the efficacy of cyclosporine in the treatment of cold urticaria.
Leukotriene receptor antagonists (eg, montelukast, zafirlukast, zileuton) have been used to treat chronic urticaria. In one report, montelukast was used in a 29-year-old woman with cold urticaria who had initially been treated with cetirizine 30 mg daily, cyproheptadine 4 mg daily, and doxycycline 200 mg daily with minimal to no relief. After treatment with montelukast, she experienced notable and stable improvements in symptoms.16 Hydroxychloroquine also has been shown to be safe and to substantially improve quality of life in patients with idiopathic chronic urticaria.17 Methotrexate (with close patient monitoring for adverse effects) has been reported to benefit some patients whose chronic urticaria was unresponsive to standard treatment.18 Treatment regimens for chronic urticaria have shown variable success in the treatment of cold urticaria and may be considered in cases refractory to treatment with high-dose second-generation H1 antihistamines.
Topical application of capsaicin for 4 to 7 days has been shown to deplete the neuropeptides in sensory fibers that may be involved in cold reactions, although skin irritation may prevent usage.19
Prednisone therapy was used in a small study of 6 patients with acquired cold urticaria.20 Three patients were treated for periods of 3 to 5 days with prednisone 20 mg each morning. Three other patients were given a single dose of prednisone 20 mg or 25 mg in the morning, depending on body weight. Following prednisone therapy, complete or partial pruritus was subjectively improved in all 6 patients. Additionally, significant reductions in venous histamine concentrations at 5 and 10 minutes following cold immersion were noted (P<.05 and P<.025, respectively); however, no significant improvement in either erythema or edema was noted posttreatment following cold immersion.20 Despite these findings, prednisone has not been shown to consistently prevent histamine release. Another report noted the case of a 47-year-old man with cold urticaria who required hypothermic cardiopulmonary bypass. Pretreatment with prednisone 20 mg daily and preoperative hydrocortisone 100 mg intravenously did not prevent histamine release.21
Cold desensitization (ie, exposing progressively larger areas of the patient’s skin to increasingly colder water) may induce tolerance to cold and decrease the temperature threshold at which symptoms develop; however, patients with known serious systemic reactions should be tested with extreme caution and only under the supervision of a clinician.22,23 Tolerance may wane when cold desensitization therapy is stopped.
The prognosis for patients with acquired cold urticaria generally is good. Improvement of symptoms or full remission occurs within 5 to 6 years in 50% of patients.24 Once remission has occurred, patients generally remain symptom free. For other familial variants, symptoms may last a lifetime.
Conclusion
This case report and review of the literature highlights the limitations of cold urticaria and the importance of effective management in improving quality of life in affected patients. Symptoms may limit patients’ ability to work in certain environments, inhibit them from engaging in daily activities, and even prevent them from leaving their homes in colder temperatures. In addition to behavioral modifications, pharmacologic management may provide symptomatic relief. Antihistamines are the first line of treatment in cold urticaria. Second-generation antihistamines, which are more selective for H1 receptors and less sedating, are generally recommended. Up to 4 times the standard dosage of these medications may be required for effective treatment.5 The primary goal of therapy in mild to moderate cases is improvement in quality of life.
Cold urticaria is a rare condition characterized by a localized or systemic eruption of papules upon exposure of the skin to cold air, liquids, and/or objects. In some cases, angioedema and anaphylaxis can occur. The wheal-and-flare reaction results from a localized or systemic release of histamine, leukotrienes, and various other proinflammatory mast cell mediators. Cold urticaria can be acquired or follow an autosomal-dominant familial transmission pattern. Acquired cold urticaria often presents in young adulthood with a mean duration of 4 to 5 years and remission or improvement of symptoms after 5 years in 50% of cases.1 The familial variant most commonly presents in early childhood and endures throughout the patient’s life.2 Cold urticaria generally is classified as acute or chronic if symptoms persist for more than 6 weeks. Pharmacologic therapies with prophylactic effects that may reduce the intensity of symptoms or inhibit their development include antihistamines, leuko-triene receptor antagonists, biologics, and glucocorticoids. We present the case of a 23-year-old man with cold urticaria that was refractory to initial treatment with H1 antihistamines along with a review of the literature.
Case Report
A 23-year-old man presented to the dermatology clinic for evaluation of recurrent burning, itching, and sometimes development of a painful rash on the face, neck, and arms of 2 years’ duration that typically occurred following exposure to cold, wind, and rain. He also developed symptoms in warm weather when exposed to wind while sweating. His medical history was remarkable for asthma, which was not active. He was not taking any medications and had no known drug or environmental allergies. No other members of his household developed similar symptoms. His only successful means of prevention was to stay indoors, which thereby limited his activities.
Physical examination of the dorsal hands following an ice cube test revealed numerous 3- to 5-mm urticarial papules with surrounding erythema (Figure).

Following the initial evaluation, the patient was treated unsuccessfully with a mix of first- and second-generation antihistamines in gradually increasing doses to a maximum dose of loratadine 20 mg once daily, cetirizine 20 mg once daily, and hydroxyzine 20 mg once daily. A course of montelukast 10 mg once daily was started in addition to the antihistamines and led to a reduction in the severity of the lesions but not the frequency and did not relieve the burning sensation; the patient subsequently discontinued therapy. Next, a trial of cyclosporine was attempted, but the patient reported that it caused emesis and subsequently discontinued treatment. The patient also did not tolerate prednisone. He eventually decided to treat his symptoms with lifestyle choices only, such as making sure to be well covered in cold temperatures.
Comment
Cold urticaria is a physical urticaria resulting from mast cell degranulation and the subsequent release of histamine and proinflammatory cytokines upon exposure of the skin to cold air, liquid, and/or objects. Symtpoms usually are limited to localized exposed areas of the skin but also can be generalized. Cold urticaria typically manifests as erythematous, pruritic papules and also may be accompanied by deep tissue involvement resulting in angioedema and/or anaphylaxis. Symptoms usually occur within minutes of cold exposure; however, in delayed-type cold urticaria, symptoms may develop 24 to 72 hours later.3 Prevalence is relatively equal in both sexes and is highest among young adults (ie, 18–27 years old), with a greater incidence associated with cold climates.4 In one study, the overall incidence of acquired cold urticaria in Central Europe was estimated to be 0.05%.1
Systemic involvement may occur with extensive cold contact, ranging in severity from generalized urticaria to anaphylaxis and involvement of the cardiovascular, respiratory, and/or gastrointestinal systems.5 Patients who exhibit systemic responses to cold exposure should avoid swimming in cold water, as this may induce anaphylaxis and result in injury or death. In a 2004 study that included 30 children with cold urticaria at a tertiary center in Boston,6 11 (36.7%) participants who underwent cold stimulation testing developed systemic symptoms; 5 (45.5%) participants experienced respiratory distress and 8 (72.7%) experienced a decrease in level of consciousness (eg, faintness, dizziness, hypotension). Aquatic activity was the trigger in all 11 participants except for 1 (9.0%), who experienced systemic symptoms on exposure to cold air. In the same study, 14 (46.7%) participants were diagnosed with asthma and 15 (50%) were diagnosed with allergic rhinitis. Of the 28 participants whose family histories were available for review, 25 (89.3%) had a family history of atopic disease.6 A 2008 Greek study4 of 62 adults with acquired cold urticaria found that 18 (29%) participants had at least 1 serious systemic response resulting in generalized urticaria or angioedema associated with hypotension (eg, dizziness, fainting, disorientation, shock). In both of these studies, a majority of the serious systemic reactions were associated with cold water activities.
Cold urticaria is primarily an idiopathic phenomenon but can be classified as acquired or familial. Acquired cold urticaria may result from primary or secondary causes, which can include cryoglobulinemia, human immunodeficiency virus, syphilis, mononucleosis, rubeola, toxoplasmosis, varicella, hepatitis, and various drugs (eg, penicillin, angiotensin-converting enzyme inhibitors, oral contraceptives).7 Familial causes include cryopyrin-associated periodic syndrome, phospholipase Cγ2 gene–associated antibody deficiency and immune dysregulation, Muckle-Wells syndrome, and neonatal-onset multisystem inflammatory disease.
Typically, cold urticaria is diagnosed using cold stimulation tests such as the ice cube test, in which an ice cube is applied directly to the patient’s skin for 3 to 5 minutes and a response is measured 10 minutes after its removal.8 This test has been shown to have a sensitivity of 83% to 90% and a specificity of 100%.9 Alternatively, cold urticaria may be diagnosed through the use of a Peltier element-based cold-provocation device, which exposes the patient to a variety of temperatures in order for clinicians to determine the threshold upon which there is an observable reaction. With a sensitivity of 93% and specificity of 100%, the accuracy of this test is similar to that of the ice cube test.10 If a patient has a history of serious systemic involvement, any testing that exposes the patient to extensive cold exposure should be used with caution.
Patients should be counseled about potential serious systemic symptoms and the importance of wearing appropriate cold-weather clothing. Avoidance of cold water activities and overexposure to cold weather also should be emphasized. Pharmacologic therapy for prophylaxis typically includes a second-generation H1 antihistamine (eg, cetirizine, loratadine, desloratadine). Since these drugs have been shown to be less sedating than first-generation antihistamines, they are considered a better choice for chronic treatment. At high doses, however, these medications may have a sedative effect; therefore, nighttime use is preferable if possible. The standard dosage is 5 mg to 10 mg daily for oral cetirizine, 10 mg daily for oral loratadine, and 5 mg daily for oral desloratadine; however, up to 4 times the standard dosage of these medications may be required for effective treatment of cold urticaria.11 Given the associated risk of anaphylaxis, patients should be prescribed an epinephrine pen and educated about its appropriate use, including the importance of keeping the pen accessible at all times.
In refractory cases of cold urticaria, an H2 antihistamine (eg, ranitidine) can be used in conjunction with H1 antihistamines.12 Omalizumab, an IgE-mediated treatment, also has been shown to be safe and effective in patients with recalcitrant physical urticaria, including cold urticaria.13,14 One report described the case of a 69-year-old woman with cold urticaria who was unable to leave the house without developing a widespread eruption on the face, trunk, and limbs.15 After undergoing a series of unsuccessful treatments, the patient was started on cyclosporine 125 mg twice daily, which was reduced to 100 mg twice daily after 4 weeks of therapy and then reduced to 75 mg twice daily after 4 months of treatment. One week after therapy was initiated the patient reported that she was able to leave the house, and after 4 weeks of treatment the lesions only developed on the hands and feet. The patient remained in remission with a low-dose therapy of cyclosporine 75 mg twice daily with lesions only occurring on the hands and feet. The low-dose maintenance therapy was associated with minimal adverse effects.15 To our knowledge, there are no known large studies on the efficacy of cyclosporine in the treatment of cold urticaria.
Leukotriene receptor antagonists (eg, montelukast, zafirlukast, zileuton) have been used to treat chronic urticaria. In one report, montelukast was used in a 29-year-old woman with cold urticaria who had initially been treated with cetirizine 30 mg daily, cyproheptadine 4 mg daily, and doxycycline 200 mg daily with minimal to no relief. After treatment with montelukast, she experienced notable and stable improvements in symptoms.16 Hydroxychloroquine also has been shown to be safe and to substantially improve quality of life in patients with idiopathic chronic urticaria.17 Methotrexate (with close patient monitoring for adverse effects) has been reported to benefit some patients whose chronic urticaria was unresponsive to standard treatment.18 Treatment regimens for chronic urticaria have shown variable success in the treatment of cold urticaria and may be considered in cases refractory to treatment with high-dose second-generation H1 antihistamines.
Topical application of capsaicin for 4 to 7 days has been shown to deplete the neuropeptides in sensory fibers that may be involved in cold reactions, although skin irritation may prevent usage.19
Prednisone therapy was used in a small study of 6 patients with acquired cold urticaria.20 Three patients were treated for periods of 3 to 5 days with prednisone 20 mg each morning. Three other patients were given a single dose of prednisone 20 mg or 25 mg in the morning, depending on body weight. Following prednisone therapy, complete or partial pruritus was subjectively improved in all 6 patients. Additionally, significant reductions in venous histamine concentrations at 5 and 10 minutes following cold immersion were noted (P<.05 and P<.025, respectively); however, no significant improvement in either erythema or edema was noted posttreatment following cold immersion.20 Despite these findings, prednisone has not been shown to consistently prevent histamine release. Another report noted the case of a 47-year-old man with cold urticaria who required hypothermic cardiopulmonary bypass. Pretreatment with prednisone 20 mg daily and preoperative hydrocortisone 100 mg intravenously did not prevent histamine release.21
Cold desensitization (ie, exposing progressively larger areas of the patient’s skin to increasingly colder water) may induce tolerance to cold and decrease the temperature threshold at which symptoms develop; however, patients with known serious systemic reactions should be tested with extreme caution and only under the supervision of a clinician.22,23 Tolerance may wane when cold desensitization therapy is stopped.
The prognosis for patients with acquired cold urticaria generally is good. Improvement of symptoms or full remission occurs within 5 to 6 years in 50% of patients.24 Once remission has occurred, patients generally remain symptom free. For other familial variants, symptoms may last a lifetime.
Conclusion
This case report and review of the literature highlights the limitations of cold urticaria and the importance of effective management in improving quality of life in affected patients. Symptoms may limit patients’ ability to work in certain environments, inhibit them from engaging in daily activities, and even prevent them from leaving their homes in colder temperatures. In addition to behavioral modifications, pharmacologic management may provide symptomatic relief. Antihistamines are the first line of treatment in cold urticaria. Second-generation antihistamines, which are more selective for H1 receptors and less sedating, are generally recommended. Up to 4 times the standard dosage of these medications may be required for effective treatment.5 The primary goal of therapy in mild to moderate cases is improvement in quality of life.
- Siebenhaar F, Weller K, Mlynek A, et al. Acquired cold urticaria: clinical picture and update on diagnosis and treatment. Clin Exp Dermatol. 2007;32:241-245.
- Gandhi C, Healy C, Wanderer AA, et al. Familial atypical cold urticaria: description of a new hereditary disease. J Allergy Clin Immunol. 2009;124:1245-1250.
- Bäck O, Larsen A. Delayed cold urticaria. Acta Derm Venereol. 1978;58:369-371.
- Katsarou-Katsari A, Makris M, Lagogianni E, et al. Clinical features and natural history of acquired cold urticaria in a tertiary referral hospital: a 10-year prospective study. J Eur Acad Dermatol Venereol. 2008;22:1405-1411.
- Wanderer AA, Grandel KE, Wasserman SI, et al. Clinical characteristics of cold-induced systemic reactions in acquired cold urticaria syndromes: recommendations for prevention of this complication and a proposal for a diagnostic classification of cold urticaria. J Allergy Clin Immunol. 1986;78(3 Pt 1):417-423.
- Alangari AA, Twarog FJ, Shih MC, et al. Clinical features and anaphylaxis in children with cold urticaria. Pediatrics. 2004;113:e313-e317.
- Wanderer AA, Hoffman HM. The spectrum of acquired and familial cold-induced urticaria/urticaria-like syndromes. Immunol Allergy Clin North Am. 2004;24:259-286.
- Visitsuntorn N, Tuchinda M, Arunyanark N, et al. Ice cube test in children with cold urticaria. Asian Pac J Allergy Immunol. 1992;10:111-115.
- Neittaanmäki H. Cold urticaria. clinical findings in 220 patients. J Am Acad Dermatol. 1985;13:636-644.
- Siebenhaar F, Staubach P, Metz M, et al. Peltier effect-based temperature challenge: an improved method for diagnosing cold urticaria. J Allergy Clin Immunol. 2004;114:1224-1225.
- Siebenhaar F, Degener F, Zuberbier T, et al. High-dose desloratadine decreases wheal volume and improves cold provocation thresholds compared with standard-dose treatment in patients with acquired cold urticaria: a randomized, placebo-controlled, crossover study. J Allergy Clin Immunol. 2009;123:672-679.
- Duc J, Pécoud A. Successful treatment of idiopathic cold urticaria with the association of H1 and H2 antagonists: a case report. Ann Allergy. 1986;56:355-357.
- Metz M, Altrichter S, Ardelean E, et al. Anti-immunoglobulin E treatment of patients with recalcitrant physical urticaria. Int Arch Allergy Immunol. 2011;154:177-180.
- Boyce JA. Successful treatment of cold-induced urticaria/anaphylaxis with anti-IgE. J Allergy Clin Immunol. 2006;117:1415-1418.
- Marsland AM, Beck MH. Cold urticaria responding to systemic cyclosporine. Br J Dermatol. 2003;149:214-215.
- Hani N, Hartmann K, Casper C, et al. Improvement of cold urticaria by treatment with the leukotriene receptor antagonist montelukast. Acta Derm Venereol. 2000;80:229.
- Reeves GE, Boyle MJ, Bonfield J, et al. Impact of hydroxychloroquine therapy on chronic urticaria: chronic autoimmune urticaria study and evaluation. Intern Med J. 2004;34:182-186.
- Perez A, Woods A, Grattan CE. Methotrexate: a useful steroid-sparing agent in recalcitrant chronic urticaria. Br J Dermatol. 2010;162:191-194.
- Tóth-Kása I, Jancsó G, Obál F Jr, et al. Involvement of sensory nerve endings in cold and heat urticaria. J Invest Dermatol. 1983;80:34-36.
- Black AK, Keahey TM, Eady RA, et al. Dissociation of histamine release and clinical improvement following treatment of acquired cold urticaria by prednisone. Br J Clin Pharmacol. 1981;12:327-331.
- Johnston WE, Moss J, Philbin DM, et al. Management of cold urticaria during hypothermic cardiopulmonary bypass. N Engl J Med. 1982;306:219-221.
- Krause K, Zuberbier T, Maurer, M. Modern Approaches to the diagnosis and treatment of cold contact urticaria. Curr Allergy Asthma Rep. 2010;10:273-279.
- von Mackensen YA, Sticherling M. Cold urticaria: tolerance induction with cold baths. Br J Dermatol. 2007;157:835-836.
- Möller A, Henning M, Zuberbier T, et al. Epidemiology and clinical aspects of cold urticaria [article in German]. Hautarzt. 1996;47:510-514.
- Siebenhaar F, Weller K, Mlynek A, et al. Acquired cold urticaria: clinical picture and update on diagnosis and treatment. Clin Exp Dermatol. 2007;32:241-245.
- Gandhi C, Healy C, Wanderer AA, et al. Familial atypical cold urticaria: description of a new hereditary disease. J Allergy Clin Immunol. 2009;124:1245-1250.
- Bäck O, Larsen A. Delayed cold urticaria. Acta Derm Venereol. 1978;58:369-371.
- Katsarou-Katsari A, Makris M, Lagogianni E, et al. Clinical features and natural history of acquired cold urticaria in a tertiary referral hospital: a 10-year prospective study. J Eur Acad Dermatol Venereol. 2008;22:1405-1411.
- Wanderer AA, Grandel KE, Wasserman SI, et al. Clinical characteristics of cold-induced systemic reactions in acquired cold urticaria syndromes: recommendations for prevention of this complication and a proposal for a diagnostic classification of cold urticaria. J Allergy Clin Immunol. 1986;78(3 Pt 1):417-423.
- Alangari AA, Twarog FJ, Shih MC, et al. Clinical features and anaphylaxis in children with cold urticaria. Pediatrics. 2004;113:e313-e317.
- Wanderer AA, Hoffman HM. The spectrum of acquired and familial cold-induced urticaria/urticaria-like syndromes. Immunol Allergy Clin North Am. 2004;24:259-286.
- Visitsuntorn N, Tuchinda M, Arunyanark N, et al. Ice cube test in children with cold urticaria. Asian Pac J Allergy Immunol. 1992;10:111-115.
- Neittaanmäki H. Cold urticaria. clinical findings in 220 patients. J Am Acad Dermatol. 1985;13:636-644.
- Siebenhaar F, Staubach P, Metz M, et al. Peltier effect-based temperature challenge: an improved method for diagnosing cold urticaria. J Allergy Clin Immunol. 2004;114:1224-1225.
- Siebenhaar F, Degener F, Zuberbier T, et al. High-dose desloratadine decreases wheal volume and improves cold provocation thresholds compared with standard-dose treatment in patients with acquired cold urticaria: a randomized, placebo-controlled, crossover study. J Allergy Clin Immunol. 2009;123:672-679.
- Duc J, Pécoud A. Successful treatment of idiopathic cold urticaria with the association of H1 and H2 antagonists: a case report. Ann Allergy. 1986;56:355-357.
- Metz M, Altrichter S, Ardelean E, et al. Anti-immunoglobulin E treatment of patients with recalcitrant physical urticaria. Int Arch Allergy Immunol. 2011;154:177-180.
- Boyce JA. Successful treatment of cold-induced urticaria/anaphylaxis with anti-IgE. J Allergy Clin Immunol. 2006;117:1415-1418.
- Marsland AM, Beck MH. Cold urticaria responding to systemic cyclosporine. Br J Dermatol. 2003;149:214-215.
- Hani N, Hartmann K, Casper C, et al. Improvement of cold urticaria by treatment with the leukotriene receptor antagonist montelukast. Acta Derm Venereol. 2000;80:229.
- Reeves GE, Boyle MJ, Bonfield J, et al. Impact of hydroxychloroquine therapy on chronic urticaria: chronic autoimmune urticaria study and evaluation. Intern Med J. 2004;34:182-186.
- Perez A, Woods A, Grattan CE. Methotrexate: a useful steroid-sparing agent in recalcitrant chronic urticaria. Br J Dermatol. 2010;162:191-194.
- Tóth-Kása I, Jancsó G, Obál F Jr, et al. Involvement of sensory nerve endings in cold and heat urticaria. J Invest Dermatol. 1983;80:34-36.
- Black AK, Keahey TM, Eady RA, et al. Dissociation of histamine release and clinical improvement following treatment of acquired cold urticaria by prednisone. Br J Clin Pharmacol. 1981;12:327-331.
- Johnston WE, Moss J, Philbin DM, et al. Management of cold urticaria during hypothermic cardiopulmonary bypass. N Engl J Med. 1982;306:219-221.
- Krause K, Zuberbier T, Maurer, M. Modern Approaches to the diagnosis and treatment of cold contact urticaria. Curr Allergy Asthma Rep. 2010;10:273-279.
- von Mackensen YA, Sticherling M. Cold urticaria: tolerance induction with cold baths. Br J Dermatol. 2007;157:835-836.
- Möller A, Henning M, Zuberbier T, et al. Epidemiology and clinical aspects of cold urticaria [article in German]. Hautarzt. 1996;47:510-514.
Practice Points
- Cold urticaria is a physical urticaria characterized by a localized or systemic eruption of papules upon exposure of the skin to cold air, liquids, and/or objects.
- Symptoms of cold urticaria, which range from erythema, pruritus, and hives to angioedema and sometimes anaphylaxis, may be debilitating for patients; therefore, effective treatment is required to improve quality of life.
- First-line treatment for cold urticaria includes second-generation H1 antihistamines at up to 4 times the standard dosage.
Cancer prevention field riding high into the new year
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
The new year has us all looking forward and the cancer prevention community is no exception.
In a special report entitled “Transforming Cancer Prevention through Precision Medicine and Immune-Oncology,” a team of experts offer a brief look at what we can expect in the near future for cancer prevention research, including a Pre-Cancer Genome Atlas (PCGA), and highlight some of the recent advances shaping their optimism.
“Just as precision therapy and immunotherapy are transforming cancer treatment, precision medicine and immunoprevention approaches are being translated to the clinic and showing great promise. We stand at the edge of a new frontier that will include comprehensively characterizing the molecular and cellular events that drive premalignant progression (e.g. PCGA),” Dr. Scott M. Lippman, director of the University of California San Diego Moores Cancer Center, and his coauthors wrote (Cancer Prev Res. 2016;9:2-10).
The report details some of the clinical firsts in 2015 including genomic studies suggesting that clonal hematopoiesis is a premalignant state for blood cancer, the first precision medicine trial in cancer prevention (EPOC) reporting that loss of heterozygosity can predict which patients with premalignant mouth lesions are most likely to develop oral cancer, and the U.S. Preventive Services Task Force recommending low-dose aspirin for colorectal cancer prevention based on age and risk.
Randomized trials have also suggested that a single dose of human papillomavirus vaccine can provide durable protection against HPV infection. Tumor biology studies established new chemoprevention for familial adenomatous polyposis syndrome and universal tumor screening guidelines based on DNA mismatch repair mutations and microsatellite instability for colorectal cancer in patients with Lynch syndrome.
Further, remarkable advances have been made in liquid biopsy technology, high-throughput functional screening, and computational biology methods and algorithms that “provide unprecedented opportunities to interrogate the biology of premalignancy...” they noted.
In an American Association for Cancer Research blog post, Dr. Lippman acknowledges that not everyone is the same page when it comes to the underlying principles of cancer prevention.
A “contentious” paper published at the start of 2015 suggested that variations in cancer risk are due to random mutations or what might otherwise be called bad luck. The new year was heralded in by a second paper, however, that came to roughly the opposite conclusion or that most cancers are preventable.
In February, an AACR Cancer Prevention Summit will bring together various stakeholders to discuss the current state of cancer prevention and to identify top priorities and research directions for the field, he noted.
The authors acknowledged grant support from the National Institutes of Health/National Cancer Institute.
FROM CANCER PREVENTION RESEARCH
Flu activity high in New Jersey, South Carolina
Week 12 of the 2015-2016 U.S. flu season saw two states in the “high” range of the influenza-like illness (ILI) activity scale, the Centers for Disease Control and Prevention reported.
New Jersey was at level 10 on the CDC’s 1-10 scale for the week ending Jan. 2, 2016, and South Carolina was at level 9. The next-most-active state, Texas, was at level 7, putting it in the “moderate” range along with Maryland, which was at level 6, the CDC said.
States in the “low” range were Arizona, Oklahoma, Pennsylvania, and Virginia at level 5 and California, Colorado, and Georgia at level 4. Altogether, there were 24 states at level 2 or higher, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network.
Puerto Rico also moved up to level 10 for the week ending Jan. 2 and, in a case of geographic concentration, New York City was at level 6 even though New York State was still at level 1.
The proportion of outpatient visits for ILI nationally was 2.8% at the end of week 12, which is above the national baseline of 2.1%. For comparison, at this point in the 2014-2015 flu season, the proportion of visits for ILI was about 6%, and in 2013-2014 it was around 4.5%.
There were two flu-related pediatric deaths reported during the week, bringing the total to six for the 2015-2016 season. One death was associated with an influenza A (H3) virus and the other was associated with
an influenza A (H1N1)pdm09 virus. Looking again at previous years, there had been single weeks with at least six pediatric deaths by week 12 in each of the last three seasons, the CDC report showed.
Week 12 of the 2015-2016 U.S. flu season saw two states in the “high” range of the influenza-like illness (ILI) activity scale, the Centers for Disease Control and Prevention reported.
New Jersey was at level 10 on the CDC’s 1-10 scale for the week ending Jan. 2, 2016, and South Carolina was at level 9. The next-most-active state, Texas, was at level 7, putting it in the “moderate” range along with Maryland, which was at level 6, the CDC said.
States in the “low” range were Arizona, Oklahoma, Pennsylvania, and Virginia at level 5 and California, Colorado, and Georgia at level 4. Altogether, there were 24 states at level 2 or higher, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network.
Puerto Rico also moved up to level 10 for the week ending Jan. 2 and, in a case of geographic concentration, New York City was at level 6 even though New York State was still at level 1.
The proportion of outpatient visits for ILI nationally was 2.8% at the end of week 12, which is above the national baseline of 2.1%. For comparison, at this point in the 2014-2015 flu season, the proportion of visits for ILI was about 6%, and in 2013-2014 it was around 4.5%.
There were two flu-related pediatric deaths reported during the week, bringing the total to six for the 2015-2016 season. One death was associated with an influenza A (H3) virus and the other was associated with
an influenza A (H1N1)pdm09 virus. Looking again at previous years, there had been single weeks with at least six pediatric deaths by week 12 in each of the last three seasons, the CDC report showed.
Week 12 of the 2015-2016 U.S. flu season saw two states in the “high” range of the influenza-like illness (ILI) activity scale, the Centers for Disease Control and Prevention reported.
New Jersey was at level 10 on the CDC’s 1-10 scale for the week ending Jan. 2, 2016, and South Carolina was at level 9. The next-most-active state, Texas, was at level 7, putting it in the “moderate” range along with Maryland, which was at level 6, the CDC said.
States in the “low” range were Arizona, Oklahoma, Pennsylvania, and Virginia at level 5 and California, Colorado, and Georgia at level 4. Altogether, there were 24 states at level 2 or higher, according to data from the CDC’s Outpatient Influenza-like Illness Surveillance Network.
Puerto Rico also moved up to level 10 for the week ending Jan. 2 and, in a case of geographic concentration, New York City was at level 6 even though New York State was still at level 1.
The proportion of outpatient visits for ILI nationally was 2.8% at the end of week 12, which is above the national baseline of 2.1%. For comparison, at this point in the 2014-2015 flu season, the proportion of visits for ILI was about 6%, and in 2013-2014 it was around 4.5%.
There were two flu-related pediatric deaths reported during the week, bringing the total to six for the 2015-2016 season. One death was associated with an influenza A (H3) virus and the other was associated with
an influenza A (H1N1)pdm09 virus. Looking again at previous years, there had been single weeks with at least six pediatric deaths by week 12 in each of the last three seasons, the CDC report showed.
Proton Pump Inhibitors Linked to Chronic Kidney Disease
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
See below for "Views on the News," a pro/con discussion on PPIs.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
Pro: When used as indicated, PPIs are good medicine
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
See below for "Views on the News," a pro/con discussion on PPIs.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
Pro: When used as indicated, PPIs are good medicine
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
See below for "Views on the News," a pro/con discussion on PPIs.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
Pro: When used as indicated, PPIs are good medicine
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
FROM JAMA INTERNAL MEDICINE
Proton pump inhibitors linked to chronic kidney disease
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
Available evidence suggests that proton pump inhibitor use is associated with an increased risk of both acute and chronic kidney disease, hypomagnesemia, Clostridium difficile infection, and osteoporotic fractures. Caution in prescribing PPIs should be used in patients at high risk for any of these conditions. Given the association with kidney disease and low magnesium levels, serum creatinine and magnesium levels probably should be monitored in patients using PPIs, especially those using high doses.
Given the evidence that PPI use is linked with a number of adverse outcomes, we recommend that patients and clinicians discuss the potential benefits and risks of PPI treatment, as well as potential alternative regimens such as histamine H2 receptor antagonists or lifestyle changes, before PPIs are prescribed. In patients with symptomatic gastrointestinal reflux, ulcer disease, and severe dyspepsia, the benefits of PPI use likely outweigh its potential harms. For less serious symptoms, however, and for prevention of bleeding in low-risk patients, potential harms may outweigh the benefits. A large number of patients are taking PPIs for no clear reason – often remote symptoms of dyspepsia or heartburn that have since resolved. In these patients, PPIs should be stopped to determine if symptomatic treatment is needed.
Dr. Adam J. Schoenfeld and Dr. Deborah Grady are with the University of California, San Francisco. They had no disclosures. These comments were taken from their editorial (JAMA Intern Med. 2016 Jan 11. doi: 10.1001/jamainternmed.2015.7927).
The bottom line is that PPIs should be used continually for the three specific conditions for which they are known to be beneficial – hypersecretory states, gastroesophageal reflux disease (in all its manifestations), and NSAID/aspirin prophylaxis. As with all drugs, treatment always should be at the lowest effective dose. Although it is quite appropriate to limit chronic PPI use to these groups, given the potential association (no causality identified) with various putative side effects including renal disease, in my opinion, the risks of denying PPIs when indicated are higher than the low risks of renal or other possible side effects.
Dr. David C. Metz is associate chief for clinical affairs, GI division; codirector, esophagology and swallowing program; director, acid-peptic program; codirector, neuroendocrine tumor center; and professor of medicine at the Hospital of the University of Pennsylvania, Philadelphia.
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
The use of proton pump inhibitors increased the risk of chronic kidney disease by 20%-50%, said the authors of two large population-based cohort analyses published online Jan. 11 in JAMA Internal Medicine.
These are the first such studies to link PPI use to chronic kidney disease (CKD), and the association held up after controlling for multiple potential confounders, said Dr. Benjamin Lazarus of Johns Hopkins University, Baltimore, and his associates. “Further research is required to investigate whether PPI use itself causes kidney damage and, if so, the underlying mechanisms of this association,” they wrote.
Proton pump inhibitors have been linked to other adverse health effects but remain among the most frequently prescribed medications in the United States. To further explore the risk of PPI use, the researchers analyzed data for 10,482 adults from the Atherosclerosis Risk in Communities (ARIC) study who were followed for a median of 13.9 years, and a replication cohort of 248,751 patients from a large rural health care system who were followed for a median of 6.2 years.
Incident CKD was defined based on hospital discharge diagnosis codes, reports of end-stage renal disease from the United States Renal Data System Registry, or a glomerular filtration rate of less than 60 mL/min per 1.73 m2 that persisted at follow-up visits (JAMA Intern Med. 2016 Jan 11. doi: 0.1001/jamainternmed.2015.7193).
In the ARIC study, there were 56 cases of CKD among 322 self-reported baseline PPI users, for an incidence of 14.2 cases per 1,000 person-years – significantly higher than the rate of 10.7 cases per 1,000 person-years among self-reported baseline nonusers. The 10-year estimated absolute risk of CKD among baseline users was 11.8% – 3.3% higher than the expected risk had they not used PPIs. Furthermore, PPI users were at significantly higher risk of CKD after demographic, socioeconomic, and clinical variables were accounted for (hazard ratio, 1.50; 95% confidence interval, 1.1-2.0), after modeling varying use of PPIs over time (adjusted HR, 1.3; 95% CI, 1.2-1.5), after directly comparing PPI users with H2 receptor antagonist users (adjusted HR, 1.4; 95% CI, 1.01-1.9), and after comparing baseline PPI users with propensity score–matched nonusers (HR, 1.8; 95% CI, 1.1-2.7).
In the replication cohort, there were 1,921 new cases of CKD among 16,900 patients with an outpatient PPI prescription (incidence of 20.1 cases per 1,000 person-years). The incidence of CKD among the other patients was lower: 18.3 cases per 1,000 person-years. The use of PPIs was significantly associated with incident CKD in all analyses, and the 10-year absolute risk of CKD among baseline PPI users was 15.6% – 1.7% higher than the expected risk had they not used PPIs.
These observational analyses cannot show causality, but a causal relationship between PPIs and CKD “could have a considerable public health impact, give the widespread extent of use,” the researchers emphasized. “More than 15 million Americans used prescription PPIs in 2013, costing more than $10 billion. Study findings suggest that up to 70% of these prescriptions are without indication and that 25% of long-term PPI users could discontinue therapy without developing symptoms. Indeed, there are already calls for the reduction of unnecessary use of PPIs (BMJ. 2008;336:2-3).”
The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
FROM JAMA INTERNAL MEDICINE
Key clinical point: The use of proton pump inhibitors was significantly associated with incident chronic kidney disease (CKD) in two large population-based studies.
Major finding: Baseline PPI use was associated with a 20%-50% increase in the risk of CKD, and the association held up in all sensitivity analyses.
Data source: A prospective, population-based cohort study of 10,482 adults from the Atherosclerosis Risk in Communities study and a separate replication analysis of 248,751 patients from a large health care system.
Disclosures: The study was funded by the National Institute of Diabetes and Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute, both of which are part of the National Institutes of Health. The researchers had no disclosures.
How new dietary guidelines affect health care providers
The U.S. Department of Health and Human Services and the U.S. Department of Agriculture have released the eighth iteration of the Dietary Guidelines for Americans, a set of recommendations for healthy eating habits Americans should adopt to prevent development of hypertension, heart disease, and type 2 diabetes, among other conditions.
The new guidelines, effective through 2020, highlight the importance of eating a wide variety of fruits, vegetables, grains, and dairy products, while staying away from processed foods heavy in saturated fats, sugar, and cholesterol as much as possible. The difference between this and earlier editions of the guidelines, the health agencies say, is to promote the importance of a wider variety of foods Americans should be consuming, rather than focusing on just a few isolated foods that should be integrated into an otherwise inadequate diet.
“Protecting the health of the American public includes empowering them with the tools they need to make healthy choices in their daily lives,” Secretary of Health and Human Services Sylvia M. Burwell said in a statement. “By focusing on small shifts in what we eat and drink, eating healthy becomes more manageable. The Dietary Guidelines provide science-based recommendations on food and nutrition so people can make decisions that may help keep their weight under control, and prevent chronic conditions, like Type 2 diabetes, hypertension, and heart disease.”
The American Medical Association voiced their support of the new guidelines, saying that they are “extremely pleased that the new recommendations call for significantly reducing the amount of added sugars and sugar sweetened beverages from the American diet.” Similarly, the American College of Cardiology issued a statement saying that the existence of “a source of clear science-based information about diet” is more important than ever for Americans in the face of increasingly omnipresent and often confusing information available; the college also lauded the recommendations to limit the intake of added sugars, saturated and trans fats, and sodium. The American Heart Association released a new Authoritative Review of data on the topic of nutritional balance as it related to chronic diseases.
While the recommendations may seem common sense and geared more towards patients and laymen, they are of equal importance to health care providers. Dr. Carolyn Lopez – a Chicago-area family physician and adjunct professor of medicine at Northwestern University – called the guidelines an important resource for physicians, with several elements that will be of particular benefit to physicians and clinicians looking to improve the quality of care given to their patients.
“People [should] understand that while individual food choices are important, the pattern of eating is paramount,” Dr. Lopez explained in an interview. “It’s not impossible to bring vegetables into breakfast – veggie omelettes are great – but it’s hard to imagine bringing a vegetable into the whole grain cereals with skim milk breakfast.”
As a family practitioner, however, Dr. Lopez stressed the difficulty of any doctor having a significant enough amount of time with each patient to really go in-depth into what needs to be done to enact meaningful nutritional and lifestyle changes. “These guidelines can only be effective if the whole team is talking to patients,” she explained. “On its own, it would be extraordinarily difficult [or] impossible to accomplish.
Dr. Nazrat Mirza, a pediatrician who is medical director of the IDEAL Pediatric Weight Management Clinic at Children’s National Health System, Washington, said in an interview that the guidelines are “significant in that they are leading us even closer to healthier dietary living – and from my pediatric perspective, healthier children who will grow into healthier adults. We must keep in mind that these guidelines are not prescriptive – but generalized to relatively healthy people. In particular, the recommendation to reduce sugar intake with specification of an upper limit of 10% of calories from added sugar factored into the suggested daily nutritional intake is an excellent update. Added sugar consumption is linked to diabetes, so any reduction in recommended intake will steer people away from potentially developing diabetes.”
She continued, “In my clinic and day-to-day counseling of patients, being able to point to helpful resources such as these guidelines is crucial. These guidelines will continue to serve as yet another way to reach parents and affect the daily dietary habits they practice at home. I was happy to see resources such as MyPlate.gov referenced in the guidelines; that is a tool I use regularly. When educating families on what a healthy plate looks like, I’m able to point directly to the MyPlate.gov posters hanging in the clinic. Comprehensive resources such as these guidelines, [which] give parents actual examples of foods, and deliver the information in a clear, concise, implementable way, are the best methods to reach parents and kids.”
“As a medical provider, such guidelines provide us with tools and reinforcement to say ‘this is policy,’ when approaching schools about the food and drink options they offer children on a daily basis – because the schools are going to have to follow these guidelines. If these guidelines can enforce public policy for the kids to eat healthy, ... and we can implement changes according to the guidelines, that would help greatly in the prevention of chronic disorders such as obesity, hypertension and diabetes – which is a step in the right direction,” said Dr. Mirza, also of George Washington University, Washington.
“Most physicians are so rushed nowadays by the health care system that they typically do not have adequate time to discuss these very important issues,” Dr. Rodbard said in an interview. “It takes time and perseverance to educate the patient and provide individualized care, assessing their current diet, assessing their willingness or potential for changing their diet or increasing their physical activity.”
On that last point. Dr. Rodbard criticized the new guidelines for not emphasizing the importance of exercise enough. “More clarity and emphasis on physical activity should have been provided,” she said, adding that “people need continual, gentle reminders with repetition and long-term follow-up.” Dr. Rodbard summed up the guidelines as “marginally adequate” but “a step in the right direction.”
An endocrinologist who practices in Winter Park, Fla., Dr. Victor L. Roberts – a Fellow of the ACP and ACE – agreed that time with the patients is critical in order for these guidelines to have any effect, calling the guidelines “just the GPS, not the destination.”
Preaching an ABC strategy – accountability, behavior, and calories, the latter of which he stressed is the most important part of any dietary strategy – Dr. Roberts explained that clinicians and physicians should leave the more specific dietary advice to nutritional experts and focus on telling their patients to watch calories, watch the amount of food they’re eating of any given type, and to exercise more.
“These guidelines crystallize and summarize what we’ve already known, and what we should have been practicing and advocating, for decades,” Dr. Roberts said in an interview, adding that “the difficulty is putting these recommendations into practice and having patients and doctors accept responsibility.”
Ultimately, the three doctors who treat adult patients concurred that the new guidelines are imperfect, and likely won’t result in any automatic widespread change. Dr. Rodbard admitted that she does not anticipate the guidelines affecting the way she treats her patients very much, if at all, while Dr. Lopez said that she’s “not sure it’s going to have a major effect.” Furthermore, the sheer volume of data and information available often leads to patients simply not knowing what to believe, which Dr. Roberts described as a “glazed over” effect of being told so many different things from so many different sources, patients just tune everything out.
Ultimately, while it’s important for health care providers to give advice and recommendations as necessary, these should be done on an individual basis. And, if a patient requires extensive dietary and nutritional intervention, that should be left in the hands of specialist who can accurately determine what the patient is capable of, what their goals should be, and how to tailor a plan specifically for them.
“I don’t know many clinicians, even in my own specialty, who know much about what specifically to eat,” said Dr. Roberts. “That should come from nutritional medicine people, not us.”
Dr. Lopez, Dr. Rodbard, and Dr. Roberts did not report any relevant financial disclosures.
*This article was updated 1/12/2016.
The U.S. Department of Health and Human Services and the U.S. Department of Agriculture have released the eighth iteration of the Dietary Guidelines for Americans, a set of recommendations for healthy eating habits Americans should adopt to prevent development of hypertension, heart disease, and type 2 diabetes, among other conditions.
The new guidelines, effective through 2020, highlight the importance of eating a wide variety of fruits, vegetables, grains, and dairy products, while staying away from processed foods heavy in saturated fats, sugar, and cholesterol as much as possible. The difference between this and earlier editions of the guidelines, the health agencies say, is to promote the importance of a wider variety of foods Americans should be consuming, rather than focusing on just a few isolated foods that should be integrated into an otherwise inadequate diet.
“Protecting the health of the American public includes empowering them with the tools they need to make healthy choices in their daily lives,” Secretary of Health and Human Services Sylvia M. Burwell said in a statement. “By focusing on small shifts in what we eat and drink, eating healthy becomes more manageable. The Dietary Guidelines provide science-based recommendations on food and nutrition so people can make decisions that may help keep their weight under control, and prevent chronic conditions, like Type 2 diabetes, hypertension, and heart disease.”
The American Medical Association voiced their support of the new guidelines, saying that they are “extremely pleased that the new recommendations call for significantly reducing the amount of added sugars and sugar sweetened beverages from the American diet.” Similarly, the American College of Cardiology issued a statement saying that the existence of “a source of clear science-based information about diet” is more important than ever for Americans in the face of increasingly omnipresent and often confusing information available; the college also lauded the recommendations to limit the intake of added sugars, saturated and trans fats, and sodium. The American Heart Association released a new Authoritative Review of data on the topic of nutritional balance as it related to chronic diseases.
While the recommendations may seem common sense and geared more towards patients and laymen, they are of equal importance to health care providers. Dr. Carolyn Lopez – a Chicago-area family physician and adjunct professor of medicine at Northwestern University – called the guidelines an important resource for physicians, with several elements that will be of particular benefit to physicians and clinicians looking to improve the quality of care given to their patients.
“People [should] understand that while individual food choices are important, the pattern of eating is paramount,” Dr. Lopez explained in an interview. “It’s not impossible to bring vegetables into breakfast – veggie omelettes are great – but it’s hard to imagine bringing a vegetable into the whole grain cereals with skim milk breakfast.”
As a family practitioner, however, Dr. Lopez stressed the difficulty of any doctor having a significant enough amount of time with each patient to really go in-depth into what needs to be done to enact meaningful nutritional and lifestyle changes. “These guidelines can only be effective if the whole team is talking to patients,” she explained. “On its own, it would be extraordinarily difficult [or] impossible to accomplish.
Dr. Nazrat Mirza, a pediatrician who is medical director of the IDEAL Pediatric Weight Management Clinic at Children’s National Health System, Washington, said in an interview that the guidelines are “significant in that they are leading us even closer to healthier dietary living – and from my pediatric perspective, healthier children who will grow into healthier adults. We must keep in mind that these guidelines are not prescriptive – but generalized to relatively healthy people. In particular, the recommendation to reduce sugar intake with specification of an upper limit of 10% of calories from added sugar factored into the suggested daily nutritional intake is an excellent update. Added sugar consumption is linked to diabetes, so any reduction in recommended intake will steer people away from potentially developing diabetes.”
She continued, “In my clinic and day-to-day counseling of patients, being able to point to helpful resources such as these guidelines is crucial. These guidelines will continue to serve as yet another way to reach parents and affect the daily dietary habits they practice at home. I was happy to see resources such as MyPlate.gov referenced in the guidelines; that is a tool I use regularly. When educating families on what a healthy plate looks like, I’m able to point directly to the MyPlate.gov posters hanging in the clinic. Comprehensive resources such as these guidelines, [which] give parents actual examples of foods, and deliver the information in a clear, concise, implementable way, are the best methods to reach parents and kids.”
“As a medical provider, such guidelines provide us with tools and reinforcement to say ‘this is policy,’ when approaching schools about the food and drink options they offer children on a daily basis – because the schools are going to have to follow these guidelines. If these guidelines can enforce public policy for the kids to eat healthy, ... and we can implement changes according to the guidelines, that would help greatly in the prevention of chronic disorders such as obesity, hypertension and diabetes – which is a step in the right direction,” said Dr. Mirza, also of George Washington University, Washington.
“Most physicians are so rushed nowadays by the health care system that they typically do not have adequate time to discuss these very important issues,” Dr. Rodbard said in an interview. “It takes time and perseverance to educate the patient and provide individualized care, assessing their current diet, assessing their willingness or potential for changing their diet or increasing their physical activity.”
On that last point. Dr. Rodbard criticized the new guidelines for not emphasizing the importance of exercise enough. “More clarity and emphasis on physical activity should have been provided,” she said, adding that “people need continual, gentle reminders with repetition and long-term follow-up.” Dr. Rodbard summed up the guidelines as “marginally adequate” but “a step in the right direction.”
An endocrinologist who practices in Winter Park, Fla., Dr. Victor L. Roberts – a Fellow of the ACP and ACE – agreed that time with the patients is critical in order for these guidelines to have any effect, calling the guidelines “just the GPS, not the destination.”
Preaching an ABC strategy – accountability, behavior, and calories, the latter of which he stressed is the most important part of any dietary strategy – Dr. Roberts explained that clinicians and physicians should leave the more specific dietary advice to nutritional experts and focus on telling their patients to watch calories, watch the amount of food they’re eating of any given type, and to exercise more.
“These guidelines crystallize and summarize what we’ve already known, and what we should have been practicing and advocating, for decades,” Dr. Roberts said in an interview, adding that “the difficulty is putting these recommendations into practice and having patients and doctors accept responsibility.”
Ultimately, the three doctors who treat adult patients concurred that the new guidelines are imperfect, and likely won’t result in any automatic widespread change. Dr. Rodbard admitted that she does not anticipate the guidelines affecting the way she treats her patients very much, if at all, while Dr. Lopez said that she’s “not sure it’s going to have a major effect.” Furthermore, the sheer volume of data and information available often leads to patients simply not knowing what to believe, which Dr. Roberts described as a “glazed over” effect of being told so many different things from so many different sources, patients just tune everything out.
Ultimately, while it’s important for health care providers to give advice and recommendations as necessary, these should be done on an individual basis. And, if a patient requires extensive dietary and nutritional intervention, that should be left in the hands of specialist who can accurately determine what the patient is capable of, what their goals should be, and how to tailor a plan specifically for them.
“I don’t know many clinicians, even in my own specialty, who know much about what specifically to eat,” said Dr. Roberts. “That should come from nutritional medicine people, not us.”
Dr. Lopez, Dr. Rodbard, and Dr. Roberts did not report any relevant financial disclosures.
*This article was updated 1/12/2016.
The U.S. Department of Health and Human Services and the U.S. Department of Agriculture have released the eighth iteration of the Dietary Guidelines for Americans, a set of recommendations for healthy eating habits Americans should adopt to prevent development of hypertension, heart disease, and type 2 diabetes, among other conditions.
The new guidelines, effective through 2020, highlight the importance of eating a wide variety of fruits, vegetables, grains, and dairy products, while staying away from processed foods heavy in saturated fats, sugar, and cholesterol as much as possible. The difference between this and earlier editions of the guidelines, the health agencies say, is to promote the importance of a wider variety of foods Americans should be consuming, rather than focusing on just a few isolated foods that should be integrated into an otherwise inadequate diet.
“Protecting the health of the American public includes empowering them with the tools they need to make healthy choices in their daily lives,” Secretary of Health and Human Services Sylvia M. Burwell said in a statement. “By focusing on small shifts in what we eat and drink, eating healthy becomes more manageable. The Dietary Guidelines provide science-based recommendations on food and nutrition so people can make decisions that may help keep their weight under control, and prevent chronic conditions, like Type 2 diabetes, hypertension, and heart disease.”
The American Medical Association voiced their support of the new guidelines, saying that they are “extremely pleased that the new recommendations call for significantly reducing the amount of added sugars and sugar sweetened beverages from the American diet.” Similarly, the American College of Cardiology issued a statement saying that the existence of “a source of clear science-based information about diet” is more important than ever for Americans in the face of increasingly omnipresent and often confusing information available; the college also lauded the recommendations to limit the intake of added sugars, saturated and trans fats, and sodium. The American Heart Association released a new Authoritative Review of data on the topic of nutritional balance as it related to chronic diseases.
While the recommendations may seem common sense and geared more towards patients and laymen, they are of equal importance to health care providers. Dr. Carolyn Lopez – a Chicago-area family physician and adjunct professor of medicine at Northwestern University – called the guidelines an important resource for physicians, with several elements that will be of particular benefit to physicians and clinicians looking to improve the quality of care given to their patients.
“People [should] understand that while individual food choices are important, the pattern of eating is paramount,” Dr. Lopez explained in an interview. “It’s not impossible to bring vegetables into breakfast – veggie omelettes are great – but it’s hard to imagine bringing a vegetable into the whole grain cereals with skim milk breakfast.”
As a family practitioner, however, Dr. Lopez stressed the difficulty of any doctor having a significant enough amount of time with each patient to really go in-depth into what needs to be done to enact meaningful nutritional and lifestyle changes. “These guidelines can only be effective if the whole team is talking to patients,” she explained. “On its own, it would be extraordinarily difficult [or] impossible to accomplish.
Dr. Nazrat Mirza, a pediatrician who is medical director of the IDEAL Pediatric Weight Management Clinic at Children’s National Health System, Washington, said in an interview that the guidelines are “significant in that they are leading us even closer to healthier dietary living – and from my pediatric perspective, healthier children who will grow into healthier adults. We must keep in mind that these guidelines are not prescriptive – but generalized to relatively healthy people. In particular, the recommendation to reduce sugar intake with specification of an upper limit of 10% of calories from added sugar factored into the suggested daily nutritional intake is an excellent update. Added sugar consumption is linked to diabetes, so any reduction in recommended intake will steer people away from potentially developing diabetes.”
She continued, “In my clinic and day-to-day counseling of patients, being able to point to helpful resources such as these guidelines is crucial. These guidelines will continue to serve as yet another way to reach parents and affect the daily dietary habits they practice at home. I was happy to see resources such as MyPlate.gov referenced in the guidelines; that is a tool I use regularly. When educating families on what a healthy plate looks like, I’m able to point directly to the MyPlate.gov posters hanging in the clinic. Comprehensive resources such as these guidelines, [which] give parents actual examples of foods, and deliver the information in a clear, concise, implementable way, are the best methods to reach parents and kids.”
“As a medical provider, such guidelines provide us with tools and reinforcement to say ‘this is policy,’ when approaching schools about the food and drink options they offer children on a daily basis – because the schools are going to have to follow these guidelines. If these guidelines can enforce public policy for the kids to eat healthy, ... and we can implement changes according to the guidelines, that would help greatly in the prevention of chronic disorders such as obesity, hypertension and diabetes – which is a step in the right direction,” said Dr. Mirza, also of George Washington University, Washington.
“Most physicians are so rushed nowadays by the health care system that they typically do not have adequate time to discuss these very important issues,” Dr. Rodbard said in an interview. “It takes time and perseverance to educate the patient and provide individualized care, assessing their current diet, assessing their willingness or potential for changing their diet or increasing their physical activity.”
On that last point. Dr. Rodbard criticized the new guidelines for not emphasizing the importance of exercise enough. “More clarity and emphasis on physical activity should have been provided,” she said, adding that “people need continual, gentle reminders with repetition and long-term follow-up.” Dr. Rodbard summed up the guidelines as “marginally adequate” but “a step in the right direction.”
An endocrinologist who practices in Winter Park, Fla., Dr. Victor L. Roberts – a Fellow of the ACP and ACE – agreed that time with the patients is critical in order for these guidelines to have any effect, calling the guidelines “just the GPS, not the destination.”
Preaching an ABC strategy – accountability, behavior, and calories, the latter of which he stressed is the most important part of any dietary strategy – Dr. Roberts explained that clinicians and physicians should leave the more specific dietary advice to nutritional experts and focus on telling their patients to watch calories, watch the amount of food they’re eating of any given type, and to exercise more.
“These guidelines crystallize and summarize what we’ve already known, and what we should have been practicing and advocating, for decades,” Dr. Roberts said in an interview, adding that “the difficulty is putting these recommendations into practice and having patients and doctors accept responsibility.”
Ultimately, the three doctors who treat adult patients concurred that the new guidelines are imperfect, and likely won’t result in any automatic widespread change. Dr. Rodbard admitted that she does not anticipate the guidelines affecting the way she treats her patients very much, if at all, while Dr. Lopez said that she’s “not sure it’s going to have a major effect.” Furthermore, the sheer volume of data and information available often leads to patients simply not knowing what to believe, which Dr. Roberts described as a “glazed over” effect of being told so many different things from so many different sources, patients just tune everything out.
Ultimately, while it’s important for health care providers to give advice and recommendations as necessary, these should be done on an individual basis. And, if a patient requires extensive dietary and nutritional intervention, that should be left in the hands of specialist who can accurately determine what the patient is capable of, what their goals should be, and how to tailor a plan specifically for them.
“I don’t know many clinicians, even in my own specialty, who know much about what specifically to eat,” said Dr. Roberts. “That should come from nutritional medicine people, not us.”
Dr. Lopez, Dr. Rodbard, and Dr. Roberts did not report any relevant financial disclosures.
*This article was updated 1/12/2016.
FROM THE USDA AND HHS
Treating Migraine in Teenagers
From the Cincinnati Children’s Hospital Medical Center, Cincinnati, OH.
Abstract
- Objective: To review the management of migraine in adolescent patients.
- Methods: Literature review in the context of 2 clinical cases.
- Results: Migraine is common in adolescents and can affect school and social functioning. Management options include lifestyle modifications and acute and preventative therapies. First-line medications for migraine in the adolescent population are over-the-counter medications, including ibuprofen, acetaminophen, and naproxen. Studies of efficacy of triptans in the treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. Several classes of medications are commonly used for migraine prevention, including antidepressants and antiepileptics. Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older. Biobehavioral treatments such as relaxation training, biofeedback, and cognitive behavioral therapy have been evaluated in randomized controlled trials and found to be efficacious.
- Conclusion: Approach to management of migraine in adolescents should be multifactorial with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Case Study 1
Initial Presentation
A 13-year-old left-handed boy with allergic rhinitis has been referred by his pediatrician for evaluation of headaches.
History
The headaches have been occurring since he was 7 years old. He describes a bilateral frontal and periorbital pain, which is either aching or throbbing in nature. There is associated photophobia and phonophobia, and the pain worsens with activity. When he was younger the headaches were frequently associated with emesis. He occasionally gets tenderness of his face during the headaches, mostly in the areas adjacent to his nose and above his eyes and occasionally associated rhinorrhea.
Initially the headaches were mild and occurring infrequently, but for the past 6 months they have been more severe and occurring 1 to 3 times per week, sometimes on consecutive days. They typically begin in the afternoon but at times occur soon after waking in the morning. If they do occur in the morning, they worsen after getting out of bed. He denies any type of warning symptoms indicating the headache will occur, and has no associated focal neurologic symptoms associated with the headaches. His mother tries to minimize medication intake so will typically wait to see if the headache is severe before giving him medication, typically 2 chewable children’s ibuprofen, which helps some of the time; however, his headache often does not completely abate unless he naps. He has also been taking loratadine daily due to concerns that his headaches may be secondary to chronic sinusitis.
He is a good student and enjoys school, but notes stress surrounding tests. He reports missing 4 days of school in the past 3 months as well as coming late twice. He thinks that there were 9 additional days in which he was unable to function at his full ability at school and at least 5 times he was unable to concentrate on his homework. He plays soccer 4 to 5 times per week. He says twice he had to skip soccer practice to due to headache and at least 3 other times he needed to take breaks during soccer due to headache. He is unsure how much he drinks on an average day but he drinks mostly with meals. He does not drink caffeine. He gets into bed at 10 pm and sometimes does not fall asleep until after 11 pm, often due to worry about tests. He often plays with his tablet when trying to fall asleep. He wakes up at 6 am for school. He typically eats 3 meals per day but occasionally misses breakfast if he is rushed in the morning.
Physical Examination
On examination, the patient is a well-developed, well-nourished male in no apparent distress. His weight was 50.9 kg (68th percentile), height 163.7 cm (65th percentile), BMI 19.9 (69th percentile), blood pressure 118/62 mm Hg, and heart rate 79 bpm. His general physical examination, including skin, HEENT, extremities, lung, cardiac, and abdominal examination was normal, with lungs clear to auscultation, heart with a regular rate and rhythm, and abdomen soft and tender without organomegaly.
On neurologic examination he was alert and attentive with normal mental status. Speech was fluent. Skull, spine, and meninges were normocephalic and atraumatic with a supple neck. Cranial nerves II through XII were normal with a normal fundoscopic examination including sharp disk, no papilledema, and normal fundus bilaterally. Motor examination was normal for tone and bulk with full strength throughout. Sensory examination was normal for light touch, temperature, vibration, and joint position sense. Finger-nose-finger fine finger movement and heel-knee-shin were normal. Deep tendon reflexes were symmetric and 2+ throughout with toes downgoing. Station and gait were normal including toe walking, heel walking, tandem walking, running, skipping, one-legged standing, Fog gate procedure, and there was no Romberg’s sign.
He had a normal comprehensive headache examination. There was no tenderness at typical migraine trigger points (nuchal line and mandibular process) and no tenderness at supraorbital notch. Neck was supple with normal rotation and normal bilateral trapezius muscle tightness. No bruits or palpitations were heard over carotid or jugular veins. There was a negative Muller sign with no pain on neck bending with pressure, and no signs of allergy or sinus symptoms.
What is the initial evaluation of a teen with recurrent headaches?
The overwhelming majority of children and adolescents presenting to medical attention with recurrent headaches will have a primary headache disorder, most commonly migraine [1].However, ruling out a secondary cause is typically of concern, both for providers and parents, and the decision regarding whether to image is often a daunting one. In a review of 6 large studies in which imaging was done in pediatric patients who were examined by a neurologist, only 14 patients were found to have CNS lesions requiring intervention, and all of these patients had abnormalities on physical exam [2]. An American Academy of Neurology (AAN) practice parameter published in 2002 recommended considering neuroimaging only in patients with abnormal neurologic examination, co-existing seizures, concerning associated neurologic features, or recent onset of severe headache or change in headache type [2].There are additional factors which may prompt one to consider imaging as well including atypical auras or very short (< 5 minutes) or protracted ( > 60 minutes) auras, trigeminal autonomic cephalalgia, brief headaches precipitated by cough, frequent early morning headaches or which wake the patient from sleep, headaches with primarily occipital location, headaches in a patient less than 6 years old or in a child who cannot describe the headaches well, or migrainous headaches in a child without any family history of migraine or migraine equivalents. However, recent studies continue to show that the imaging yield in patients with headaches is low and further studies are needed to elucidate which patients truly require imaging [3].
Given this, a thorough history, family history, physical exam, and detailed neurologic exam including fundoscopy are imperative. In addition to the general neurologic exam, a focused headache exam should be performed, including Mueller’s maneuver, auscultation for cranial bruits, evaluation of the temporomandibular joint, palpation of possible trigger points, and maneuvers to assess for cervical spine disease [4].
Classification of Symptoms
Once establishing the likelihood of a primary headache disorder, the International Classification of Headache Disorders, 3rd edition, beta version (ICHD IIIβ) [5]should be used to classify the headache diagnosis. Note that while this classification system was established for adults, most criteria are similar for children and adolescents, and the typical differences are noted in the comments of the ICHD IIIβ [5].Migraine is the most common type of primary headache brought to medical attention [1]. Migraine without aura is described as a recurrent headache disorder (at least 5 lifetime attacks) with attacks lasting 4 to 72 hours, typically unilateral, throbbing in nature, moderate to severe in intensity, aggravated by routine physical activity, and associated with nausea and/or photophobia and phonophobia. In young children, migraine is more frequently bilateral, the gastrointestinal symptoms often more pronounced than photophobia and phonophobia, and migraines may be shorter, lasting at least 2 hours without treatment [5].As patients reach adolescence, migraine features typically start to evolve into patterns described in adults. Differentiating migraine from other primary headaches, such as tension-type, can be challenging, especially in children in whom migraines are more likely to be shorter and bilateral. Tension-type headaches are bilateral or diffuse, pressing or tightening pain that is non-pulsatile lasting at least 30 minutes. The pain is described as mild to moderate in intensity and not aggravated by activity [5].They may be associated with photophobia or phonophobia (not both) and they may not be associated with nausea or vomiting [5].
Is any further workup indicated?
The patient has recurrent headaches which have been present for 5 years. While they have increased in frequency, there has not been any change in their quality. His headache description and history have no “red flag” features, and a thorough examination is normal. Therefore, neuroimaging and further workup would not be indicated in this case.
What is the diagnosis?
The patient’s headaches are throbbing in nature, exacerbated by activity, associated with nausea, photophobia, and phonophobia, and are moderate to severe. Using the ICHD IIIβ, he meets criteria for migraine without aura. While his headaches are frequent, he is having less than 15 headache days per month, so this is episodic migraine. Note that he complains of forehead and periorbital pain and occasional rhinorrhea with his headaches, leading him to have been placed on loratadine for treatment of presumed allergic sinusitis. Children meeting criteria for migraine are very frequently misdiagnosed as having sinusitis [6]due to the overlap in location of migraine pain and proximity to the frontal and maxillary sinuses, as well as the presence of autonomic features frequently present in migraine, reportedly present in 70% of pediatric migraine patients [7].The negative headache examination, including Muller’s maneuver, points against sinus disease as well.
What is the approach to management of the adolescent with migraine?
Approach to management of migraine in adolescents should be multifactorial, with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Acute Treatment
General Approach
In counseling patients with migraine, and in particular adolescents, it should be stressed that achieving a normal level of functioning as soon as possible is the goal of therapy. Common missteps in treatment include failure to take acute medication early into the headache, incorrect dosing, incompletely treating the headache, and avoidance of participating in daily activities when headaches occur.
Adolescent patients may frequently wait to take an acute medication, typically due participation in another activity, not having medication with them, or discomfort with taking medication in front of peers or at school. Additionally, patients who have headaches beginning in early childhood, with pronounced gastrointestinal (GI) features, may be aware that their headaches resolve after vomiting, and therefore get used to not treating with medications. When these patients reach adolescence, when GI symptoms tend to become less pronounced, they will need to be educated that taking medication early is imperative. Acute medications are typically more effective when taken earlier in the course of a migraine, and the importance of pausing to take medication at the onset of the headache should be stressed to all patients and parents. With that in mind, however, care should be taken to counsel patients regarding the potential for development of medication overuse headache. When headaches are frequent, a more detailed and intricate plan may need to be devised so that adolescents and parents know which headaches to treat with medication.
Given that first-line medications for treatment of migraines are over-the-counter, underdosing occurs commonly, as dosing listed on packaging is typically age-based, not weight-based. At initial visits, young adolescent patients will frequently report that a particular medication is not effective, but this is often because they are still receiving low/less optimal dosing. Clinicians should remember to follow weights and recommend dosing changes at the initial visit and follow-up visits as well.
Treatment goals, ie, complete resolution of pain and migrainous features with ability to return to normal functioning, should be made clear to patients and families at the onset of treatment. Patients frequently fall into a pattern of continuing to treat with a medication that may lessen but not completely ablate the pain of a headache, and then sleep and avoid activity. Upon awakening, the headache may be gone, however, given incomplete initial treatment, the headache may be more likely to recur within 24 hours. At that point, the headache may be more difficult to treat and therefore cause further decrease in functioning. If this cycle perpetuates, disability can become extremely burdensome in adolescents, significantly affecting school and social functioning. Therefore, a detailed plan for initial steps in management as well as steps to take should initial therapy fail to fully break the headache should be given to every patient.
Of note, given that dehydration is now recognized as a common trigger for migraine [8,9], it is generally our recommendation to drink 16 to 32 oz (depending on weight) of a hydrating fluid together with whichever acute medication is chosen, as rehydration likely assists in breaking migraines as well [8].
Medications
Over-the-Counter Agents
The first-line medications for migraine in the pediatric and adolescent populations are over-the counter medications, including ibuprofen, acetaminophen, and naproxen sodium. Ibuprofen has been well studied in pediatrics and was found to be safe and effective in 2 studies [10,11]at doses of 7.5–10 mg/kg. Again, care should be taken to ensure timely administration of ibuprofen at onset of headache, or aura if present, with appropriate weight-based dosing. Naproxen sodium has not been studied in pediatrics for treatment of migraine. However in practice, it is often used in similar doses of 10 mg/kg, with good efficacy. Although ibuprofen and naproxen sodium are both nonsteroidal anti-inflammatory medications (NSAIDs), anecdotally many patients report successful treatment with one NSAID when another has failed. Aspirin has shown efficacy in adults for treatment of acute migraine [12].It is likely effective in the pediatric population as well, but it is generally avoided due to long-standing concerns for precipitation of Reye syndrome in children. In adolescents over 16 years old, however, it is a reasonable option if there are no contraindications.
In one study, acetaminophen was compared to ibuprofen and placebo for treatment of migraine in children and adolescents and found to be effective more frequently than placebo but not as frequently as ibuprofen [10],likely due to its only minimal anti-inflammatory effects. It is a reasonable option for children and adolescents, particularly in those who have contraindications to NSAIDs.
While these over-the-counter medications are generally safe and well tolerated, clinicians should not overlook the potential for toxicity as well as medication overuse headache, and patients should be counseled to avoid use of any of these medications for more than 2 to 3 headache days per week.
Triptans
Studies of efficacy of triptans in treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. However, some have shown efficacy over placebo, and in the past few years have received FDA approval for use in the pediatric and adolescent populations. Clinicians should remember that these medications are vasoconstrictors, so they should not be used in patients with vascular disease or in patients with migraine with brainstem aura or hemiplegic migraine. Additionally, due to the risk of serotonin syndrome, they should not be used in patients on monoamine oxidase inhibitors. It is also important to educate patients to limit use of these medications to 4 to 6 times per month to avoid precipitation of medication overuse headache.
Almotriptan was approved by the FDA in 2009 for use in patients 12 years and older, based on a large randomized controlled trial comparing doses of 6.25, 12.5, and 25 mg with placebo in patients ages 12 to 17 years old. All doses resulted in statistically significant pain relief as compared to placebo, and interestingly, the 12.5-mg dose seemed to be the most effective [13].
Rizatriptan received FDA approval in 2012 for use in patients 6 years and older. In 2 randomized controlled trials in patients 6 to 17 years old, rizatriptan (5 mg for patients < 40 kg, 10 mg for patients ≥ 40 kg) was more effective than placebo in providing pain freedom at 2 hours [14,15].One earlier trial found efficacy only on some measures (weekend treatment, decrease in nausea and functional disability) but no statistically significant difference than placebo in terms of overall efficacy in achieving pain freedom at 2 hours [16].However, this trial had a higher placebo response rate than typically seen in adult triptan trials. In a recent long-term open-label study in patients 12 to 17 years old, rizatriptan was found to be generally safe and well-tolerated with consistent efficacy of 46% to 51% pain freedom at 2 hours over time [17].
A combination pill consisting of sumatriptan and naproxen (Treximet) received FDA approval in May 2015 for patients 12 years and older. This was based on a randomized controlled parallel-group trial in patients 12 to 17 years old using the sumatriptan/naproxen combination in various dose combinations: 10/60 mg, 30/180 mg, or 85/500 [18].All doses were found to be equally effective in providing pain relief at 2 hours as compared to placebo, with a higher chance of sustained pain relief at 24 hours in the group receiving the 85/500 dose. In this trial, and in a long-term open-label safety trial [19],all doses were generally well tolerated with minimal adverse reactions.
Most recently, in June 2015, zolmitriptan nasal spray was approved for patients 12 years of age and older. In the large “Double-Diamond” study, which used a novel design to attempt to minimize placebo effect, zolmitriptan nasal spray (5 mg) was found to have a significantly higher headache response rate at 1 hour than placebo, and was significantly superior to placebo with regard to multiple secondary end-points [20].Additionally, a long-term open-label trial in patients 12 to 17 years old using oral zolmitriptan (2.5 mg or 5 mg) found it to be generally effective and well tolerated [21].
No other triptans have been approved for children or adolescents, however, most are widely used in clinical practice. There is good evidence for the efficacy and tolerability of sumatriptan nasal spray [22–26]in adolescents, and it is approved for use in adolescents with migraine in Europe. Although oral sumatriptan was the first triptan available clinically in the United States and is very widely used, there is surprisingly little published evidence for its use in the pediatric and adolescent populations. In fact, 2 randomized controlled trials in adolescents failed to show efficacy of oral sumatriptan as compared to placebo [27,28].Despite this, given its availability it is a reasonable choice for adolescent patients and is often one of the first tried. There has been 1 randomized controlled trial in adolescents for eletriptan without significant differences in efficacy at 2 hours as compared to placebo, although similar to other trials the placebo rate was high and there were some differences seen in secondary outcomes measures [29].Similarly, one randomized controlled trial of naratriptan 12.5-mg tablets failed to show efficacy as compared to placebo in pain relief [30].At present time, for frovatriptan there has only been a study looking at the pharmacokinetics and safety in adolescents, which found that it was generally well tolerated and recommended adolescent dosing similar to adult dosing [31].
In choosing a triptan, clinicians should keep in mind availability of alternate forms of administration, absence or presence of significant emesis, and the age of the patient. For patients who are unable to swallow pills or who have significant emesis associated with their migraines, nasal sprays and oral dissolving forms (melts) are good options. The nasal sprays (zolmitriptan and sumatriptan) additionally have the benefit of a quicker onset (~15 minutes) in general than the oral formulations. The downside to these nasal formulations is bad taste, which is frequently reported by patients. Patients should be counseled in proper administration of these nasal sprays (ie, avoiding inhalation, which causes the medication to enter the mouth) to minimize the bad taste and maximize absorption through the nasal mucosa. Other alternatives are the oral dissolving forms (rizatriptan and zolmitriptan). Given the FDA approval, the rizatriptan melt tablets are often the first-line triptan for children under age 12, but zolmitriptan melts are an option as well.
Preventive Treatment
General Approach
The decision to place an adolescent on a daily preventive medication should be based on a combination of headache frequency, severity, ease of breaking headaches, and overall disability as established by a disability scale (such as the PedMIDAS). Any patient with headaches occurring one or more days per week, those whose headaches are not easily treated or tend to be prolonged, and those with a PedMIDAS score of 30 or more should be considered candidate for a daily preventive. It is particularly important to consider starting a preventive early in adolescent patients given the possibility of impacting overall disease progression at a young age. While the natural history of headaches that start in the young is still being investigated, a known risk factor for transformation of episodic to chronic migraine is frequent headaches [32].It is therefore imperative to attempt to intervene early to improve quality of life in the present and also to prevent a downward cycle into chronicity, potential medication overuse, and worsening disability.
At present, there are several classes of medications that are commonly used for migraine prevention, including antidepressants, antiepileptics, antihistamines, and antihypertensives. Patients should be educated regarding the medication’s typical use, the specific way in which it is used in migraine, potential side effects, and overall expectations of efficacy. Most preventives need to be titrated up slowly to maximize tolerability, and patients need to understand that it may take time before they start to see results. In general, we recommend titrating up over the course of 4 to 12 weeks (depending on medication and dose goal), and then a substantial trial of 4 to 6 weeks on a full dose of medication before determining efficacy. If a medication shows a trend towards improvement but the patient has not met treatment goals, medication can be titrated up further at that point as tolerated. Patients and families should also understand that these medications are not intended as a “cure” for migraine but rather as a tool for improvement, which should be used in conjunction with the rest of a detailed plan. Generally, if a patient is well controlled for 4 to 6 months (ie, 3 or less headaches/month that are easily broken with medications, and a PedMIDAS < 30), attempts should be made to wean off of medication.
Medications
First-line Therapies
Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older (approved in March 2014). Three randomized double-blind placebo-controlled trials in children and adolescents have found topiramate to be superior to placebo in multiple endpoints [33–35].Doses in these trials were 2–3 mg/kg/day, 100 mg/day, and 50 or 100 mg/day, respectively. Notably, the 50 mg/day dose was not found to be superior to placebo. Multiple retrospective, open-label, and drug comparison trials have shown effectiveness and tolerability as well [36–42].We typically use a goal dose of 2 mg/kg/day, which is reached after titrating over 8 to 12 weeks, which minimizes side effects (most commonly paresthesias, memory/language dysfunction, appetite suppression, and drowsiness), but higher doses can be used if needed.
Amitriptyline has consistently shown efficacy in adult migraine trials and is therefore one of the most commonly used medications worldwide for prevention of migraine in children and adolescents. Surprisingly, there have been no published randomized controlled trials using amitriptyline in the pediatric population, although a trial comparing amitriptyline, topiramate, and placebo is currently underway (Childhood and Adolescent Migraine Prevention Study) [43].In 2 retrospective pediatric studies improvement with amitriptyline was reported in 84.2% and 89% of patients, respectively [44,45].We typically use a goal dose of 1 mg/kg/day, also with an 8- to 12-week titration. The most common side effects with amitriptyline are somnolence, dry mouth, and weight gain, but it is generally well tolerated in children and adolescents. There is also a risk of worsening depression and suicidal thoughts, so it is recommended to use caution if considering prescribing to a patient with underlying depression. It is typically administered in once daily dosing a few hours before bed to minimize morning drowsiness. There is also a concern for precipitation of arrhythmias, and while there are no guidelines recommending screening ECGs, this should be considered in patients with a family history of heart disease. Of note, many practitioners use nortriptyline in place of amitriptyline as it can be less sedating. It should be noted, however, that evidence for its efficacy is lacking. Additionally, it may carry a higher risk of arrhythmia [44].
Second-line Therapies
The second-line therapies typically considered are other antiepileptics, valproic acid, levetiracetam, and zonisamide, for which there is some evidence, although mostly in the form of open-label or retrospective studies [46–51]. Of note, despite the many promising retrospective and open-label studies for valproic acid [46–48], one randomized double-blind placebo control trial comparing various doses of extended release divalproex sodium with placebo in adolescent patients failed to show a statistically significant treatment difference between any dose and placebo [52].It is, however, frequently prescribed and anecdotally quite efficacious. Given concerns about potential for teratogenicity, and possible effects on ovarian function, as well as potential for weight gain and hair loss, it should be used with caution in adolescent females.
Antihypertensives (beta blockers and calcium channel blockers) have long been used for prevention of migraine in both the adult and pediatric population, but evidence for their use in the pediatric population is conflicting. An early double-blind crossover study of propranolol in patients 7 to 16 years old showed significant efficacy as compared with placebo [53].However, in 2 subsequent studies it failed to show efficacy as compared with placebo and self-hypnosis, respectively [54,55].Given the conflicting evidence and the potential for hypotension, depression, and exercise-induced asthma, use of propranolol has fallen out of favor by experts for use in pediatric and adolescent migraine prevention. Flunarizine, a nonselective calcium channel blocker, has demonstrated effectiveness in pediatric migraine prevention [56,57],and is actually approved in Europe for this indication, but it is not available in the United States.
Cyproheptadine is an antihistamine with antiserotonergic properties which is used frequently for migraine prevention in young children who are unable to swallow tablets, although evidence is limited to 1 retrospective study [45].However, given the propensity for weight gain with this medication, it is generally not recommended for use in adolescents.
In recent years, there has been a growing interest in nutraceuticals given their general tolerability and minimal side effects, as well as a comfort in using a more “natural” approach, particularly in pediatrics. Evidence is limited but some of the frequently used substances may be beneficial. Butterbur (petasites hybridus) has strong evidence in adults and is recommended by the AAN for migraine prevention in adults [58].There is one small pediatric RCT showing efficacy for butterbur as compared to music therapy [59],as well as one promising open-label trial [60]. Typical dosing ranges from 50–150 mg daily. However, butterbur contains pyrrolizidine alkaloids, which are hepatotoxic, and due to the concerns for inadequate monitoring of removal of these substances in the manufacturing of commercial butterbur, it has been generally avoided in the pediatric population. Coenzyme Q10 (CoQ10) is considered possibly effective for adult migraine prevention by the AAN [58].Pediatric evidence is limited to an open-label study showing improvement with supplementation of CoQ10 in deficient patients [61],and a subsequent double-blind placebo-controlled add-on study, which showed improvement in both the CoQ10 and placebo groups but faster improvement in the CoQ10 group [62].Typical pediatric dosing is 1–2 mg/kg/day. Magnesium is considered by the AAN to be a good option for migraine prevention in adults [58].One randomized controlled trial in children however had equivocal results [63],while one small prospective open-label study had positive results [64]. Magnesium supplementation may be more effective in patients who have low ionized magnesium levels, but this is difficult to measure reliably in the clinical setting. Doses of 9 mg/kg/day can be used with the most common side effects reported being gastrointestinal upset and diarrhea, generally dose dependent. Riboflavin is also considered by the AAN to be probably effective for prevention in adults [58],but again the evidence in children is limited to one positive retrospective study [65]and 2 equivocal randomized controlled trials [66,67],one of which had an unusually high placebo rate [66]. Appropriate dosing is also something of debate, as riboflavin is minimally absorbed and has a short half-life, so while studies were done using 200–400 mg daily, smaller more frequent dosing may be needed.
A potential approach to treatment with vitamins is to check for deficiencies, but currently only the study mentioned above in which CoQ10 levels were checked showed improvement with normalizing of low levels [61,62].Further research into this topic is needed to elucidate whether checking and repleting levels of specific vitamins would be beneficial in prevention of migraines in certain patients.
Healthy Habits
The importance of maintaining healthy lifestyle habits and modifying potentially detrimental ones should be stressed to patients and families, and counseling regarding these issues should be provided at every visit, as repetition is often key to patients understanding their importance. Skipping meals is a commonly reported migraine trigger [68,69].This is not an uncommon occurrence even in the most well-meaning of families; adolescents often report not feeling hungry in the morning, and in the rush to get to school will often skip breakfast. Many patients report not liking school lunches leading them to go most of the day without food. Adolescent girls may skip meals due to weight concerns. Patients need to be reminded that well-balanced meals throughout the day is imperative, and they often may need specific counseling on how to achieve this practically. Of note, unless a specific migraine trigger has been identified in a given patient, we do not generally recommend restricting any specific food lists.
Maintaining good sleep hygiene and a consistent sleep schedule is also often difficult for adolescent patients, with after-school activities, homework, and screen use (eg, television, electronic devices) often contributing to late bedtimes with then forced early morning waking for school. However, improvement in sleep hygiene has been shown to be effective in improving migraines in children and adolescents [70],and realistic plans for improvement in sleep should be discussed with patients.
Dehydration is also a common migraine trigger [8,9],so the importance of staying well hydrated should be stressed as well. Again, specific recommendations for how this can be achieved are often needed, especially given adolescents’ busy schedules. Additionally, many schools do not allow water bottles to be carried, and often a school note is needed so patients may be allowed to carry a water bottle at school and also be provided extra bathroom breaks as needed.
Also important is stressing maintenance of daily functioning throughout migraine treatment. By the time adolescents seek medical care, they may already be in a cycle of missing school due to headaches, and some may even be receiving home-schooling. The goals of staying in school and learning to function with headaches should be stressed, often with the help of coping skills, which will be discussed below, as it has been shown that functional disability generally improves before pain [71].
Behavioral Treatments
Psychological treatments are an important aspect of migraine management. Biobehavioral treatments such as relaxation training, biofeedback, and multimodal cognitive behavioral therapy (CBT) have been evaluated in randomized controlled trials and reviewed in meta-analyses in pediatric migraine populations and found to be efficacious [72–75].These treatments have been shown to reduce pain intensity and disability for children and adolescents with headaches and therapeutic gains appear to be maintained [72].
Relaxation training typically includes instruction in techniques such as diaphragmatic breathing, progressive muscle relaxation, and guided imagery. Relaxation training is most effective with children 7 years of age or older [76].Biofeedback is often used in conjunction with relaxation training to provide audio or visual feedback about normally unconscious physiological body responses associated with increased relaxation. Effective biofeedback parameters used with children and adolescents with migraine headache include electromyographic (EMG) activity and peripheral skin temperature [77].Biofeedback techniques can help children and adolescents become more aware of physical responses, better control these responses and generalize physical responses outside of therapy sessions to better cope with pain [76].CBT involves instruction in skills such as biofeedback-assisted relaxation training, activity pacing, distraction, and cognitive strategies for coping with pain. CBT was shown to be effective in a recent study comparing adolescent patients with chronic migraine receiving amitriptyline and CBT with patients receiving amitriptyline and standard headache education [78]. Patients who received CBT plus amitriptyline had greater reductions in days with headache and migraine-related disability compared with patients who received headache education plus amitriptyline [78].Hypnosis and acceptance and commitment therapy (ACT) are also psychological treatments used with children and adolescents with migraine headaches. ACT prioritizes the outcome of improved functioning above headache reduction and has broadly demonstrated efficacy for chronic pain [79]. Both treatments have shown promising benefit but there has been less evidence supporting the use of these therapies in pediatric headache than other well-established behavioral treatments.
The presence of comorbid psychiatric issues such as anxiety, depression, or ADHD can make the treatment of patients with migraine headaches more complex. A recent study found that approximately 30% of a sample of children and adolescents with chronic daily headache had a lifetime psychiatric diagnoses and having a lifetime psychiatric diagnosis was associated with poorer headache-related disability and quality of life [80].As a result, more intensive behavioral treatment for children and adolescents with a psychiatric comorbidity may be needed to focus on emotional and behavioral issues.
Pediatric psychologists can assess pain-related disability and coping difficulties and treat children and adolescents with migraine headaches. Children are often referred to a psychologist or other mental health professional if headaches are severe or impairing functioning. Unfortunately, access to this therapy is sometimes limited, but when available, should be offered to any migraine patients with significant disability requiring prevention, and specifically to patients with chronic migraine.
How should the patient be treated?
Acute Treatment
The patient reports only minimal response to ibuprofen. However, he is only taking 200 mg and does not take it until well after the headache has started. He should be instructed to take ibuprofen 600 mg (~10 mg/kg) as soon as the headache starts, along with 32 oz of a sports drink, and told that he should repeat this dose in 4 hours if his headache has not completely resolved with the first dose. As his headaches are occurring less than 3 times per week on average, he can do this with every headache. He should be given a note for school allowing him to receive the medication at school, and noting the importance of allowing him to get the medication as soon as he reports a headache. Given that some of his headaches occur on consecutive days, we suspect some may be continuations of a prior headache, so again the importance of obtaining complete pain relief from his acute medication should be stressed. If at his next follow-up visit he reports not always having complete relief with ibuprofen, we would consider trying naproxen instead, and/or adding a triptan, most likely sumatriptan 100 mg given its availability. He would need to understand that he may only use the triptan for 4 to 6 headache days/month. Depending on his response to sumatriptan, it could be used alone or in combination with his NSAID. He also may be instructed to use an NSAID alone for more mild headaches and his NSAID together with sumatriptan for more severe headaches.
Preventive Treatment
PedMIDAS score is a 25, indicating mild disability, however his migraines are frequent, occurring 1–3 times per week and clearly having a significant impact on his functioning. We would therefore recommend starting a prophylactic medication. He notes difficulty remembering to take medications as well as difficulty falling asleep, so amitriptyline would be a good choice for him. For his weight of 50.9 kg, we would start with at 12.5 mg at night and increase by 12.5 mg every 2 weeks to a dose of 50 mg (~1mg/kg/day). He and Mom should be counseled that it will likely take at least a few weeks on his goal dose before they start to see results. He should be counseled also to report to Mom immediately if he has any depressive or suicidal thoughts. Topiramate is another option, especially as it is FDA-approved for migraine prevention in children older than 12 years, although twice daily dosing may be a factor in maintaining compliance. Careful discussion with the patient and family in regards to the potential risks and benefits is important prior to starting any preventative medication.
Biobehavioral Management
The patient should be drinking 8–10 cups of non-caffeinated fluid per day and additional cups on days he plays soccer. He should be given a school note to this effect so he may carry a water bottle at school. He eats well, but the importance of not skipping breakfast should be stressed, and ideas for fitting this in should be given. The importance of a consistent bedtime routine should be stressed, with good sleep hygiene to assist with easier sleep onset. It should be made clear that he should not be using screens within an hour prior to bed. He already exercises frequently and should be commended for being active. Given his report of frequent headaches which are impairing school attendance, functioning at school, and participation in soccer activities, screening for potential co-morbid psychiatric issues and making a referral for further treatment with a pediatric pain psychologist focused on coping with chronic pain is appropriate.
Case Study 2
Initial Presentation and History
A 16-year-old right-handed girl who has been having headaches since she was 10 years old presents for evaluation. She reports unilateral mostly right-sided headaches, which are throbbing and associated with photophobia, phonophobia, osmophobia, nausea, and occasionally vomiting. She has no premonitory or aura symptoms. Her headaches typically occur once a week but she has noted that they tend to be more frequent around the time of her menses and that often these headaches do not fully respond to naproxen, which typically works to break her migraines during other times in the month. Upon further review, she believes these headaches typically start on the day or two prior to her menses, and that this happens almost every month. She often misses school due to these headaches. Her mother would like to know whether the patient should see a gynecologist to potentially be placed on birth control to control her headaches.
Physical Examination
On examination, weight was 61.5 kg (75.8 percentile), height 157.1 cm (18.4 percentile), BMI 24.91 (84 percentile), blood pressure 112/55 mm Hg, and heart rate 90 bpm. Her general physical and neurologic exam results were normal. She had tightness over the left trapezius muscles, otherwise the remainder of headache examination was unremarkable.
What is the probable diagnosis?
According to the ICHD-IIIβ, the patient meets criteria for episodic migraine without aura. Based on the history, she likely also has menstrually related migraine without aura [5].Officially, however, 3 months of prospective documentation is needed to make this diagnosis. Menstrually related migraine is included in the appendix of the ICHD-IIIβ, meaning there is ongoing debate about how it should be classified. According to the current appendix criteria, headaches should meet criteria for migraine without aura and also have documented and prospectively recorded evidence over at least 3 consecutive cycles with headaches confirmed on day 1+/– 2 (ie, 2 days prior to onset of menstruation or within the first 3 days of menstruation) in at least 2 out of 3 menstrual cycles, with migraines occurring during other times of the month as well [5]. This is distinguished from pure menstrual migraine without aura, in which migraines occur only during days 1+/–2 of menstruation but not during other times of the month [5].While we therefore cannot say that the patient meets ICHD IIIβ criteria for menstrually related migraine due to the lack of prospective documentation, her history suggests this.
Menstrually Related Migraine
An association of migraine with menses is well described in adults, occurring in up to 60% of adult female migraine patients [81].In adults, it has been observed that the migraines associated with the menstrual cycle tend to be more severe [82],associated with more nausea and vomiting [82],longer and less responsive to acute medications [83],and associated with more work-related disability [83]. Recently, analysis of data from the American Migraine Prevalence and Prevention (AMPP) Study found that women with pure menstrual migraine and menstrually associated migraine have on average higher MIDAS scores than those with non-menstrual migraine [81].
Only one study has explored menstrually related migraines in adolescents [84].It was a clinic-based study that found a similar prevalence—50% of adolescent patients who had reached menarch noted an association of migraine with their menses. Overall worse disability was not demonstrated in this study, but individual menstrual attacks were not compared with nonmenstrual attacks [84].No other studies have addressed this population, but it is clear that the pattern of menstrually related migraine exists in adolescents and it is important to recognize this pattern as these patients may require additional focused care in addition to standard migraine management.
What are treatment options?
In adults, options for more specific management of pure menstrual migraine and menstrually related migraine include intermittent prophylaxis with NSAIDs or triptans or use of hormonal contraceptives. There are no studies addressing any of these treatments in adolescents, so at this point management decisions must be based on evidence extrapolated from adult studies as well as attention to specific concerns in adolescent patients.
Generally, intermittent prophylaxis (using a medication a few days prior to and during the first few days of menses) with various medications has shown efficacy in adult studies. This approach may be more appropriate for patients with pure menstrual migraine, as there is less likelihood of precipitating medication overuse in this population. It can be considered in patients with menstrually related migraine as well if typical daily preventives have not been effective. One small open-label trial using naproxen 550 mg daily prior to and during menses (at differing schedules depending on the month) showed slightly decreased frequency and severity of headaches during that time [85].However, triptans are what are most commonly used.
Triptans
Frovatriptan is the longest-acting triptan and the one used most commonly for intermittent prophylaxis. One double-blind randomized controlled crossover study showed improvements in headache frequency, severity, and duration using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses [86].Frovatriptan 2.5 mg daily showed some efficacy as well but was not as effective as 2.5 mg BID [86].One prospective randomized placebo-controlled trial demonstrated more headache-free cycles using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses, as compared to placebo [87].
Naratriptan, also a longer-acting triptan, was studied in 3 prospective double-blind trials at doses of 1 mg BID used for 5 days total starting 2 days prior to menses, and all 3 showed a decrease in headache frequency as compared to placebo [88,89].Of note no efficacy was shown using 2.5 mg daily [88].
Less commonly used for intermittent prophylaxis are the shorter-acting triptans, but they have shown some efficacy as well. In one prospective study using oral sumatriptan 25 mg TID for 5 days each cycle starting 2 to 3 days before onset of menses, there seemed to be at least some improvement with sumatriptan in most cycles treated [90].Zolmitriptan was studied in one prospective double-blind placebo-controlled study using 2.5 mg BID or 2.5 mg TID for 7 days total with each cycle and was found to be associated with a significant reduction in headache frequency as compared with placebo [91].There has been one prospective trial using eletriptan 20 mg TID for 6 days total starting 2 days prior to menses, which also showed improvement in headache activity in 55% of patients [92].
Hormonal Contraceptives
In general for adult patients, hormonal contraceptives are not considered first-line treatment for menstrually related migraine. However, in patients who do not respond to other modes of treatment, or who plan to use hormonal contraception for contraceptive purposes, published expert opinions have recommended considering extended or continuous hormonal regimens [93,94].
Estrogen withdrawal during the luteal phase of the monthly cycle has long been speculated to be of importance in the pathophysiology of menstrually related migraine [93]and the relationship between hormonal contraceptives and migraine is complicated. Headache is a commonly reported side effect of combined hormonal contraceptives [95]; however, this effect seems to occur mostly during the hormone-free week [96]. It has been shown that headache occurs less frequently in women using an extended cycle regimen (84 or 168 days) as compared to those using a traditional monthly cyclic regimen [97,98].Studies addressing the use of combined hormonal contraceptives for women specifically with menstrually related migraine are limited. One small prospective randomized study showed improvement in menstrually related migraine in patients treated with a low dose (20 mcg ethinyl E[2]) oral hormonal contraceptive in both a 21/7 cycle and a 24/4 cycle, with more improvement in the group using a 24/4 cycle [99].Another showed improvement in menstrually related migraine in all study patients treated with a low-dose hormonal contraceptive (20 mcg ethinyl estradiol) on a 21/7 regimen, but with additional 0.9 mg conjugated equine estrogen during the placebo week [100].There has been one randomized placebo-controlled double-blind trial in patients with menstrually related migraine using an extended 168 hormonal regimen along with frovatriptan vs. placebo for 5 days during the hormone-free interval. Overall daily headache scores were decreased from pre-study cycles, and the increase in headaches during the hormone-free interval was lower in the frovatriptan group [101].A recent study showed that contraceptive-induced amenorrhea can be beneficial for decreasing migraine frequency in patients with menstrual migraine [102].There have been no studies addressing the use of hormonal contraceptives for migraine management in adolescents.
In adolescents, the decision regarding use of hormonal contraceptives is more complicated. There is less of a chance that adolescent patients, especially younger ones, would be planning to use hormones for contraception so their use may be solely for the purpose of migraine management. However, hormonal contraceptives are commonly used in adolescents for management of other menstrual-related disorders, such as menorrhagia, dysmenorrhea, and endometriosis, and extended cycle and continuous regimens have become more popular with adolescent providers in general [103].The general concern with using hormonal contraceptives in adolescents is that they have potential for longer-term use than adult patients, and the effects of using hormonal contraceptives long term are unknown. The major concerns are for potential interference with expected increase in bone mineral density in adolescents, effects on fertility, and risk for cancer and cardiovascular disease [103].Additionally, specifically in migraine patients, is the concern for increased stroke risk. The increased risk for ischemic stroke in patients with migraine, although more specifically with migraine with aura, is well known [104–106]and migraine has recently been shown to be a risk factor for ischemic stroke in adolescents as well [107]. In adults, the use of hormonal contraceptives in patients with migraine with aura is known to increase the risk of ischemic stroke [106],and this has not been studied in adolescents. Given the various unknowns in the use of hormonal contraceptives in patients with menstrually related migraines, we would not recommend this as first-line treatment. However, similarly to adults, in patients who do not respond to other methods of migraine management, or who seek to use hormonal contraceptives for contraception or for other menstrually related disorders, extended or continuous cycle hormonal contraceptives may be a reasonable option, at the lowest possible estrogen dose. However, migraine with aura should be screened for, and its presence should prompt reconsideration of combined hormonal contraceptive use.
How should this patient be managed?
The patient is having frequent and disabling migraines, so starting a preventive medication would be appropriate. She has migraines throughout the month in addition to during her menses, so a daily prophylactic would be more appropriate than intermittent prophylaxis surrounding her menstrual cycle only. At this point, our recommendation would be to start a daily preventive with either amitriptyline or topiramate. Given that naproxen is not breaking some of her migraines, she should be given a prescription for a triptan. Sumatriptan 100 mg would be an appropriate first choice, and she can be instructed to use it along with her naproxen at the onset of her menstrual migraines. She can use it for other migraines as well but she should be instructed not to use it more than 4 to 6 times per month. She should keep a diary for the next 3 months noting most importantly headache days as well as days of menstruation, so that a more definitive pattern can be confirmed and an official diagnosis based on ICHD-IIIβ criteria can be made. If her migraines do not improve with daily preventives, at that point discussion regarding potential for intermittent prophylaxis or trial of extended cycle hormonal contraception may be considered, although with caution and discussion of risks and benefits.
Corresponding author: Hope O’Brien, MD, Cincinnati Children’s Hospital Medical Center, 3333 Burnet Ave., MLC 2015 Cincinnati, OH 45229.
Financial disclosures: None.
1. Tepper SJ, Dahlöf CG, Dowson A, et al. Prevalence and diagnosis of migraine in patients consulting their physician with a complaint of headache: data from the Landmark Study. Headache 2004;44:856–64.
2. Lewis DW et al. Practice parameter: evaluation of children and adolescents with recurrent headaches: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2002;59:490–8.
3. Martens D, Oster I, Gottschlling S, et al. Cerebral MRI and EEG studies in the initial management of pediatric headaches. Swiss Med Wkly 2012;142:w13625.
4. Linder SL. Understanding the comprehensive pediatric headache examination. Pediatr Ann 2005;34:442–6.
5. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia 2013;33:629–808.
6. Senbil N, Gürer YK, Uner C, et al. Sinusitis in children and adolescents with chronic or recurrent headache: a case-control study. J Headache Pain 2008;9:33–6.
7. Gelfand AA, Reider AC, Goadsby PJ. Cranial autonomic symptoms in pediatric migraine are the rule, not the exception. Neurology 2013;81:431–6.
8. Blau JN. Water deprivation: a new migraine precipitant. Headache 2005;45:757–9.
9. Martins IP, Gouveia RG. More on water and migraine. Cephalalgia 2007;27:372–4.
10. Hämäläinen ML, Hoppu K, Valkeila E, et al. Ibuprofen or acetaminophen for the acute treatment of migraine in children: a double-blind, randomized, placebo-controlled, crossover study. Neurology 1997;48:103–7.
11. Lewis DW, Kellsein D, Sahl G, et al. Children’s ibuprofen suspension for the acute treatment of pediatric migraine. Headache 2002;42:780–6.
12. Lipton RB, Golstein J, Baggish JS, et al. Aspirin is efficacious for the treatment of acute migraine. Headache 2005;45:283–92.
13. Linder SL, Mathew NT, Cady RK, et al. Efficacy and tolerability of almotriptan in adolescents: a randomized, double-blind, placebo-controlled trial. Headache 2008;48:1326–36.
14. Ahonen K, Hämäläinen ML, Eerola M, et al. A randomized trial of rizatriptan in migraine attacks in children. Neurology 2006;67:1135–40.
15. Ho TW, Pearlman E, Lewis D, et al. Efficacy and tolerability of rizatriptan in pediatric migraineurs: results from a randomized, double-blind, placebo-controlled trial using a novel adaptive enrichment design. Cephalalgia 2012;32:750–65.
16. Winner P, Lewis D, Visser WH, et al. Rizatriptan 5 mg for the acute treatment of migraine in adolescents: a randomized, double-blind, placebo-controlled study. Headache 2002;42:49–55.
17. Hewitt DJ, Pearlman E, Hämäläinen M, et al. Long-term open-label safety study of rizatriptan acute treatment in pediatric migraineurs. Headache 2013;53:104–17.
18. Derosier FJ, Lewis D, Hershey AD, et al. Randomized trial of sumatriptan and naproxen sodium combination in adolescent migraine. Pediatrics 2012;129:e1411–20.
19. McDonald SA, Hershey AD, Pearlman E, et al. Long-term evaluation of sumatriptan and naproxen sodium for the acute treatment of migraine in adolescents. Headache 2011;51:1374-87.
20. Lewis DW, Winner P, Hershey AD, et al. Efficacy of zolmitriptan nasal spray in adolescent migraine. Pediatrics 2007;
120:390–6.
21. Linder SL, Dowson AJ. Zolmitriptan provides effective migraine relief in adolescents. Int J Clin Pract 2000;54:466–9.
22. Winner P, Rothner AD, Saper J, et al. A randomized, double-blind, placebo-controlled study of sumatriptan nasal spray in the treatment of acute migraine in adolescents. Pediatrics 2000;106:989–97.
23. Rothner AD, Winner P, Nett R, et al. One-year tolerability and efficacy of sumatriptan nasal spray in adolescents with migraine: results of a multicenter, open-label study. Clin Ther 2000;22:1533–46.
24. Ahonen K, Hämäläinen ML, Rantala H, et al. Nasal sumatriptan is effective in treatment of migraine attacks in children: A randomized trial. Neurology 2004;62:883–7.
25. Natarajan S, Jabbour JT, Webster CJ, et al. Long-term tolerability of sumatriptan nasal spray in adolescent patients with migraine. Headache 2004;44:969–77.
26. Winner P, Rothner AD, Wooten JD, et al. Sumatriptan nasal spray in adolescent migraineurs: a randomized, double-blind, placebo-controlled, acute study. Headache 2006;46:212–22.
27. Hämäläinen ML, Hoppu K, Santavuori P. Sumatriptan for migraine attacks in children: a randomized placebo-controlled study. Do children with migraine respond to oral sumatriptan differently from adults? Neurology 1997;48:1100–3.
28. Fujita M, Sato K, Nishioka H, et al. Oral sumatriptan for migraine in children and adolescents: a randomized, multicenter, placebo-controlled, parallel group study. Cephalalgia 2014;34:365–75.
29. Winner P, Linder SL, Lipton RB, et al. Eletriptan for the acute treatment of migraine in adolescents: results of a double-blind, placebo-controlled trial. Headache 2007;47:511–8.
30. Rothner A. Efficacy and safety of naratriptan tablets in adolescent migraine [abstract]. J Neurol Sci 1997;150:S106.
31. Elkind AH, Wade A, Ishkanian G. Pharmacokinetics of frovatriptan in adolescent migraineurs. J Clin Pharmacol 2004;44:1158–65.
32. Manack AN, Buse DC, Lipton RB. Chronic migraine: epidemiology and disease burden. Curr Pain Headache Rep 2011;15:70–8.
33. Winner P, Pearlman EM, Linder SL, et al. Topiramate for migraine prevention in children: a randomized, double-blind, placebo-controlled trial. Headache 2005;45:1304–12.
34. Lakshmi CV, Singhi P, Malhi P, et al. Topiramate in the prophylaxis of pediatric migraine: a double-blind placebo-controlled trial. J Child Neurol 2007;22:829–35.
35. Lewis D, Winner P, Saper J, et al.Randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of topiramate for migraine prevention in pediatric subjects 12 to 17 years of age. Pediatrics 2009;123:924–34.
36. Hershey AD, Powers SW, Vockell AL, et al. Effectiveness of topiramate in the prevention of childhood headaches. Headache 2002;42:810–8.
37. Campistol J, Campos J, Casas C, et al. Topiramate in the prophylactic treatment of migraine in children. J Child Neurol 2005;20:251–3.
38. Unalp A, Uran N, Oztürk A. Comparison of the effectiveness of topiramate and sodium valproate in pediatric migraine. J Child Neurol 2008;23:1377–81.
39. Cruz MJ, Valencia I, Legido A, et al. Efficacy and tolerability of topiramate in pediatric migraine. Pediatr Neurol 2009;41:167–70.
40. Kim H, Byun SH, Kim JS, et al. Comparison of flunarizine and topiramate for the prophylaxis of pediatric migraines. Eur J Paediatr Neurol 2013;17:45–9.
41. Fallah R, Divanizadeh MS, Karimi M, et al. Topiramate and propranolol for prophylaxis of migraine. Indian J Pediatr 2013;80:920–4.
42. Tonekaboni SH, Ghazavi A, Fayyazi A, et al. Prophylaxis of childhood migraine: topiramate versus propranolol. Iran J Child Neurol 2013;7:9–14.
43. Hershey AD, Powers SW, Coffey CS, et al. Childhood and Adolescent Migraine Prevention (CHAMP) study: a double blinded, placebo controlled, comparative effectiveness study of amitriptyline, topiramate, and placebo in prevention of childhood and adolescent migraine. Headache 2013;53:799–816.
44. Hershey AD, Powers SW, Bentti AL, et al. Effectiveness of amitriptyline in the prophylactic management of childhood headaches. Headache 2000;40:539–49.
45. Lewis DW, Diamond S, Scott D, et al. Prophylactic treatment of pediatric migraine. Headache 2004;44:230–7.
46. Caruso JM, Brown WD, Exil G, et al. The efficacy of divalproex sodium in the prophylactic treatment of children with migraine. Headache 2000 Sep;40:672–6.
47. Pakalnis A, Greenberg G, Drake ME Jr, et al. Pediatric migraine prophylaxis with divalproex. J Child Neurol 2001;16:731–4.
48. Serdaroglu G, Erhan E, Tekgul H, et al. Sodium valproate prophylaxis in childhood migraine. Headache 2002;42:819–22.
49. Miller GS. Efficacy and safety of levetiracetem in pediatric migraine. Headache 2004;44:238–43.
50. Pakalnis A, Kring D, Meier L. Levetiracetam prophylaxis in pediatric migraine--an open label study. Headache 2007;43:427–30.
51. Pakalnis A, Kring D. Zonisamide prophylaxis in refractory pediatric headache. Headache 2006;46:804–7.
52. Apostol G, Cady RK, Laforet GA, et al. Divalproex extended-release in adolescent migraine prophylaxis: results of a randomized, double-blind, placebo-controlled study. Headache 2008;48:1012–25.
53. Ludvigsson J. Propranolol used in prophylaxis of migraine in children. Acta Neurol 1974;50:109–15.
54. Forsythe WI, Gillies D, Sills MA. Propanolol (‘Inderal’) in the treatment of childhood migraine. Dev Med Child Neurol 1984;26:737–41.
55. Olness K, MacDonald JT, Uden DL. Comparison of self-hypnosis and propranolol in the treatment of juvenile classic migraine. Pediatrics 1987;79:593–7.
56. Sorge F, DeSimone R, Marano E, et al. Flunarizine in prophylaxis of childhood migraine. A double-blind, placebo-controlled crossover study. Cephalalgia 1988;8:1–6.
57. Guidetti V, Moscato D, Ottaviano S, et al. Flunarizine and migraine in childhood: an evaluation of endocrine function. Cephalalgia 1987;7:263–6.
58. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and complementary treatments for episodic migraine treatment in adults: Report of the quality standards subcommittee of the American Academy of Neurology and American Headache Society. Neurology 2012; 78:1346–53.
59. Oelkers-Ax R, Leins A, Parzer P, et al. Butterbur root extract and music therapy in the prevention of childhood migraine: an explorative study. Eur J Pain 2008;12:301–13.
60. Pothmann R, Danesch U. Migraine prevention in children and adolescents: results of an open study with a special butterbur root extract. Headache 2005;45:196–203.
61. Hershey AD, Powers SW, Vockell AL, et al. Coenzyme Q10 deficiency and response to supplementation in pediatric and adolescent migraine. Headache 2007;47:73–80.
62. Slater SK, Nelson TD, Kabbouche MA, et al. A randomized, double-blind, crossover, add-on study of coenzyme Q10 in the prevention of pediatric and adolescent migraine. Cephalalgia 2011; 31: 897–905.
63. Wang F, Van Den Eeden SK, Ackerson LM, et al. Oral magnesium oxide prophylaxis of frequent migrainous headache in children: a randomized, double-blind, placebo-controlled trial. Headache 2003;43:601–10.
64. Castelli S, Meossi C, Domenici R, et al. [Magnesium in the prophylaxis of primary headache and other periodic disorders in children]. Pediatr Med Chir 1993;15:481–8. Italian.
65. Condò M, Posar A, Arbizzani A, et al. Riboflavin prophylaxis in pediatric and adolescent migraine. J Headache Pain 2009;10:361–5.
66. MacLennan SC, Wade FM, Forrest KM, et al. High-dose riboflavin for migraine prophylaxis in children: a double-blind, randomized, placebo-controlled trial. J Child Neurol 2008;23:1300–4.
67. Bruijn J, Duivenvoorden H, Passchier J, et al. Medium-dose riboflavin as a prophylactic agent in children with migraine: a preliminary placebo-controlled, randomised, double-blind, cross-over trial. Cephalalgia 2010;30:1426–34.
68. Robbins L. Precipitating factors in migraine: a retrospective review of 494 patients. Headache 1994;34:214–6.
69. Leviton A, Slack WV, Masek B, et al. A computerized behavioral assessment for children with headaches. Headache 1984;24:182–5.
70. Bruni O, Galli F, Guidetti V. Sleep hygiene and migraine in children and adolescents. Cephalalgia 1999;19 Suppl 25:57–9.
71. Lynch-Jordan AM, Sil S, Peugh J, et al. Differential changes in functional disability and pain intensity over the course of psychological treatment for children with chronic pain. Pain 2014;155:1955–61.
72. Eccleston C, Palermo TM, Williams AC, et al. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database Syst Rev 2014;5:CD003968.
73. Huguet A, McGrath PJ, Stinson J, et al. Efficacy of psychological treatment for headaches: an overview of systematic reviews and analysis of potential modifiers of treatment efficacy. Clin J Pain 2014;30:353–69.
74. Kropp P, Meyer B, Landgraf M, et al. Headache in children: update on biobehavioral treatments. Neuropediatrics 2013;44:20–4.
75. Kröner-Herwig B. Psychological treatments for pediatric headache. Expert Rev Neurother 2011;11:403–10.
76. Powers SW, Andrasik F. Biobehavioral treatment, disability, and psychological effects of pediatric headache. Pediatr Ann 2005;34:461–5.
77. Hermann C, Blanchard EB. Biofeedback in the treatment of headache and other childhood pain. Appl Psychophysiol Biofeedback 2002;27:143–62.
78. Powers SW, Kashikar-Zuck SM, Hershey AD, et al. Cognitive behavioral therapy plus amitriptyline for chronic migraine in children and adolescents: a randomized clinical trial. JAMA 2013;310:2622–30.
79. Smitherman TA, Wells RE, Ford SG. Emerging behavioral treatments for migraine. Curr Pain Headache Rep 2015;19:13.
80. Slater SK, O’Brien HL, Hershey AD, et al. Psychiatric comorbidity in pediatric chronic daily headache. Cephalalgia 2012;32: 1116–22.
81. Pavlovic JM, Stewart WF, Bruce CA, et al. Burden of migraine related to menses: results from the AMPP study. J Headache Pain 2015;16:24.
82. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 2004;63:351–3.
83. Granella F, Sances G, Allais G. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia 2004;24:707–16.
84. Crawford MJ, Lehman L, Slater S, et al. Menstrual migraine in adolescents. Headache 2009;49:341–7.
85. Allais G, Bussone G, De Lorenzo C, et al. Naproxen sodium in short-term prophylaxis of pure menstrual migraine: pathophysiological and clinical considerations. Neurol Sci 2007;28(Suppl 2):S225–8.
86. Silberstein S, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology 2004;63:261–9.
87. Brandes JL, Poole A, Kallela M, et al. Short-term frovatriptan for the prevention of difficult-to-treat menstrual migraine attacks. Cephalalgia 2009;29:1133–48.
88. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache 2001;41:248–56.
89. Mannix LK, Savani N, Landy S, et al. Efficacy and tolerability of naratriptan for short-term prevention of menstrually related migraine: data from two randomized, double-blind, placebo-controlled studies. Headache 2007;47:1037–49.
90. Newman LC, Lipton RB, Lay CL, et al. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology 1998;51:307–9.
91. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs 2008;22:877–86.
92. Marcus DA, Bernstein CD, Sullivan EA, et al. Perimenstrual eletriptan prevents menstrual migraine: an open-label study. Headache 2010;50:551–6.
93. Silberstein S, Patel S. Menstrual migraine: an updated review on hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother 2014;15:2063–70.
94. MacGregor EA. Migraine management during menstruation and menopause. Continuum (Minneap Minn) 2015;21(4 Headache):990–1003.
95. Aegidius K, Zwart JA, Hagen K, et al. Oral contraceptives and increased headache prevalence: The Head-HUNT Study. Neurology 2006;66:349–53.
96. Sulak PJ, Scow RD, Preece C, et al. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol 2000;95:261–6.
97. LaGuardia KD, Fisher AC, Bainbridge JD, et al. Suppression of estrogen-withdrawal headache with extended transdermal contraception. Fertil Steril 2005;83:1875–7.
98. Sulak P,Willis S, Kuehl T, Coffee A, Clark J. Headaches and oral contraceptives: Impact of eliminating the standard 7-day placebo interval. Headache 2007;47:27–37.
99. De Leo V, Scolaro V, Musacchio MC, et al. Combined oral contraceptives in women with menstrual migraine without aura. Fertil Steril 2011;96:917–20.
100. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J 2004;97:819–22.
101. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health (Larchmt) 2014;23:310–7.
102. Vetvik KG, MacGregor EA, Lundqvist C, et al. Contraceptive-induced amenorrhea leads to reduced migraine frequency in women with menstrual migraine without aura. J Headache Pain 2014;15:30.
103. Sucato GS, Gerschultz KL. Extended cycle hormonal contraception in adolescents. Curr Opin Obstet Gynecol 2005;17:461–5.
104. Spector JT, Kahn SR, Jones MR, et al. Migraine headache and ischemic stroke risk: an updated meta-analysis. Am J Med 2010;123:612–24.
105. Bigal ME, Kurth T, Santanello N, et al. Migraine and cardiovascular disease: a population-based study. Neurology 2010;74:628–35.
106. Schurks M, Rist PM, Bigal ME, et al. Migraine and cardiovascular disease: systematic review and meta-analysis. BMJ 2009;339:b3914.
107. Gelfand AA, Fullerton HJ, Jacobson A, et al. Is migraine a risk factor for pediatric stroke? Cephalalgia 2015;35:1252–60.
From the Cincinnati Children’s Hospital Medical Center, Cincinnati, OH.
Abstract
- Objective: To review the management of migraine in adolescent patients.
- Methods: Literature review in the context of 2 clinical cases.
- Results: Migraine is common in adolescents and can affect school and social functioning. Management options include lifestyle modifications and acute and preventative therapies. First-line medications for migraine in the adolescent population are over-the-counter medications, including ibuprofen, acetaminophen, and naproxen. Studies of efficacy of triptans in the treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. Several classes of medications are commonly used for migraine prevention, including antidepressants and antiepileptics. Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older. Biobehavioral treatments such as relaxation training, biofeedback, and cognitive behavioral therapy have been evaluated in randomized controlled trials and found to be efficacious.
- Conclusion: Approach to management of migraine in adolescents should be multifactorial with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Case Study 1
Initial Presentation
A 13-year-old left-handed boy with allergic rhinitis has been referred by his pediatrician for evaluation of headaches.
History
The headaches have been occurring since he was 7 years old. He describes a bilateral frontal and periorbital pain, which is either aching or throbbing in nature. There is associated photophobia and phonophobia, and the pain worsens with activity. When he was younger the headaches were frequently associated with emesis. He occasionally gets tenderness of his face during the headaches, mostly in the areas adjacent to his nose and above his eyes and occasionally associated rhinorrhea.
Initially the headaches were mild and occurring infrequently, but for the past 6 months they have been more severe and occurring 1 to 3 times per week, sometimes on consecutive days. They typically begin in the afternoon but at times occur soon after waking in the morning. If they do occur in the morning, they worsen after getting out of bed. He denies any type of warning symptoms indicating the headache will occur, and has no associated focal neurologic symptoms associated with the headaches. His mother tries to minimize medication intake so will typically wait to see if the headache is severe before giving him medication, typically 2 chewable children’s ibuprofen, which helps some of the time; however, his headache often does not completely abate unless he naps. He has also been taking loratadine daily due to concerns that his headaches may be secondary to chronic sinusitis.
He is a good student and enjoys school, but notes stress surrounding tests. He reports missing 4 days of school in the past 3 months as well as coming late twice. He thinks that there were 9 additional days in which he was unable to function at his full ability at school and at least 5 times he was unable to concentrate on his homework. He plays soccer 4 to 5 times per week. He says twice he had to skip soccer practice to due to headache and at least 3 other times he needed to take breaks during soccer due to headache. He is unsure how much he drinks on an average day but he drinks mostly with meals. He does not drink caffeine. He gets into bed at 10 pm and sometimes does not fall asleep until after 11 pm, often due to worry about tests. He often plays with his tablet when trying to fall asleep. He wakes up at 6 am for school. He typically eats 3 meals per day but occasionally misses breakfast if he is rushed in the morning.
Physical Examination
On examination, the patient is a well-developed, well-nourished male in no apparent distress. His weight was 50.9 kg (68th percentile), height 163.7 cm (65th percentile), BMI 19.9 (69th percentile), blood pressure 118/62 mm Hg, and heart rate 79 bpm. His general physical examination, including skin, HEENT, extremities, lung, cardiac, and abdominal examination was normal, with lungs clear to auscultation, heart with a regular rate and rhythm, and abdomen soft and tender without organomegaly.
On neurologic examination he was alert and attentive with normal mental status. Speech was fluent. Skull, spine, and meninges were normocephalic and atraumatic with a supple neck. Cranial nerves II through XII were normal with a normal fundoscopic examination including sharp disk, no papilledema, and normal fundus bilaterally. Motor examination was normal for tone and bulk with full strength throughout. Sensory examination was normal for light touch, temperature, vibration, and joint position sense. Finger-nose-finger fine finger movement and heel-knee-shin were normal. Deep tendon reflexes were symmetric and 2+ throughout with toes downgoing. Station and gait were normal including toe walking, heel walking, tandem walking, running, skipping, one-legged standing, Fog gate procedure, and there was no Romberg’s sign.
He had a normal comprehensive headache examination. There was no tenderness at typical migraine trigger points (nuchal line and mandibular process) and no tenderness at supraorbital notch. Neck was supple with normal rotation and normal bilateral trapezius muscle tightness. No bruits or palpitations were heard over carotid or jugular veins. There was a negative Muller sign with no pain on neck bending with pressure, and no signs of allergy or sinus symptoms.
What is the initial evaluation of a teen with recurrent headaches?
The overwhelming majority of children and adolescents presenting to medical attention with recurrent headaches will have a primary headache disorder, most commonly migraine [1].However, ruling out a secondary cause is typically of concern, both for providers and parents, and the decision regarding whether to image is often a daunting one. In a review of 6 large studies in which imaging was done in pediatric patients who were examined by a neurologist, only 14 patients were found to have CNS lesions requiring intervention, and all of these patients had abnormalities on physical exam [2]. An American Academy of Neurology (AAN) practice parameter published in 2002 recommended considering neuroimaging only in patients with abnormal neurologic examination, co-existing seizures, concerning associated neurologic features, or recent onset of severe headache or change in headache type [2].There are additional factors which may prompt one to consider imaging as well including atypical auras or very short (< 5 minutes) or protracted ( > 60 minutes) auras, trigeminal autonomic cephalalgia, brief headaches precipitated by cough, frequent early morning headaches or which wake the patient from sleep, headaches with primarily occipital location, headaches in a patient less than 6 years old or in a child who cannot describe the headaches well, or migrainous headaches in a child without any family history of migraine or migraine equivalents. However, recent studies continue to show that the imaging yield in patients with headaches is low and further studies are needed to elucidate which patients truly require imaging [3].
Given this, a thorough history, family history, physical exam, and detailed neurologic exam including fundoscopy are imperative. In addition to the general neurologic exam, a focused headache exam should be performed, including Mueller’s maneuver, auscultation for cranial bruits, evaluation of the temporomandibular joint, palpation of possible trigger points, and maneuvers to assess for cervical spine disease [4].
Classification of Symptoms
Once establishing the likelihood of a primary headache disorder, the International Classification of Headache Disorders, 3rd edition, beta version (ICHD IIIβ) [5]should be used to classify the headache diagnosis. Note that while this classification system was established for adults, most criteria are similar for children and adolescents, and the typical differences are noted in the comments of the ICHD IIIβ [5].Migraine is the most common type of primary headache brought to medical attention [1]. Migraine without aura is described as a recurrent headache disorder (at least 5 lifetime attacks) with attacks lasting 4 to 72 hours, typically unilateral, throbbing in nature, moderate to severe in intensity, aggravated by routine physical activity, and associated with nausea and/or photophobia and phonophobia. In young children, migraine is more frequently bilateral, the gastrointestinal symptoms often more pronounced than photophobia and phonophobia, and migraines may be shorter, lasting at least 2 hours without treatment [5].As patients reach adolescence, migraine features typically start to evolve into patterns described in adults. Differentiating migraine from other primary headaches, such as tension-type, can be challenging, especially in children in whom migraines are more likely to be shorter and bilateral. Tension-type headaches are bilateral or diffuse, pressing or tightening pain that is non-pulsatile lasting at least 30 minutes. The pain is described as mild to moderate in intensity and not aggravated by activity [5].They may be associated with photophobia or phonophobia (not both) and they may not be associated with nausea or vomiting [5].
Is any further workup indicated?
The patient has recurrent headaches which have been present for 5 years. While they have increased in frequency, there has not been any change in their quality. His headache description and history have no “red flag” features, and a thorough examination is normal. Therefore, neuroimaging and further workup would not be indicated in this case.
What is the diagnosis?
The patient’s headaches are throbbing in nature, exacerbated by activity, associated with nausea, photophobia, and phonophobia, and are moderate to severe. Using the ICHD IIIβ, he meets criteria for migraine without aura. While his headaches are frequent, he is having less than 15 headache days per month, so this is episodic migraine. Note that he complains of forehead and periorbital pain and occasional rhinorrhea with his headaches, leading him to have been placed on loratadine for treatment of presumed allergic sinusitis. Children meeting criteria for migraine are very frequently misdiagnosed as having sinusitis [6]due to the overlap in location of migraine pain and proximity to the frontal and maxillary sinuses, as well as the presence of autonomic features frequently present in migraine, reportedly present in 70% of pediatric migraine patients [7].The negative headache examination, including Muller’s maneuver, points against sinus disease as well.
What is the approach to management of the adolescent with migraine?
Approach to management of migraine in adolescents should be multifactorial, with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Acute Treatment
General Approach
In counseling patients with migraine, and in particular adolescents, it should be stressed that achieving a normal level of functioning as soon as possible is the goal of therapy. Common missteps in treatment include failure to take acute medication early into the headache, incorrect dosing, incompletely treating the headache, and avoidance of participating in daily activities when headaches occur.
Adolescent patients may frequently wait to take an acute medication, typically due participation in another activity, not having medication with them, or discomfort with taking medication in front of peers or at school. Additionally, patients who have headaches beginning in early childhood, with pronounced gastrointestinal (GI) features, may be aware that their headaches resolve after vomiting, and therefore get used to not treating with medications. When these patients reach adolescence, when GI symptoms tend to become less pronounced, they will need to be educated that taking medication early is imperative. Acute medications are typically more effective when taken earlier in the course of a migraine, and the importance of pausing to take medication at the onset of the headache should be stressed to all patients and parents. With that in mind, however, care should be taken to counsel patients regarding the potential for development of medication overuse headache. When headaches are frequent, a more detailed and intricate plan may need to be devised so that adolescents and parents know which headaches to treat with medication.
Given that first-line medications for treatment of migraines are over-the-counter, underdosing occurs commonly, as dosing listed on packaging is typically age-based, not weight-based. At initial visits, young adolescent patients will frequently report that a particular medication is not effective, but this is often because they are still receiving low/less optimal dosing. Clinicians should remember to follow weights and recommend dosing changes at the initial visit and follow-up visits as well.
Treatment goals, ie, complete resolution of pain and migrainous features with ability to return to normal functioning, should be made clear to patients and families at the onset of treatment. Patients frequently fall into a pattern of continuing to treat with a medication that may lessen but not completely ablate the pain of a headache, and then sleep and avoid activity. Upon awakening, the headache may be gone, however, given incomplete initial treatment, the headache may be more likely to recur within 24 hours. At that point, the headache may be more difficult to treat and therefore cause further decrease in functioning. If this cycle perpetuates, disability can become extremely burdensome in adolescents, significantly affecting school and social functioning. Therefore, a detailed plan for initial steps in management as well as steps to take should initial therapy fail to fully break the headache should be given to every patient.
Of note, given that dehydration is now recognized as a common trigger for migraine [8,9], it is generally our recommendation to drink 16 to 32 oz (depending on weight) of a hydrating fluid together with whichever acute medication is chosen, as rehydration likely assists in breaking migraines as well [8].
Medications
Over-the-Counter Agents
The first-line medications for migraine in the pediatric and adolescent populations are over-the counter medications, including ibuprofen, acetaminophen, and naproxen sodium. Ibuprofen has been well studied in pediatrics and was found to be safe and effective in 2 studies [10,11]at doses of 7.5–10 mg/kg. Again, care should be taken to ensure timely administration of ibuprofen at onset of headache, or aura if present, with appropriate weight-based dosing. Naproxen sodium has not been studied in pediatrics for treatment of migraine. However in practice, it is often used in similar doses of 10 mg/kg, with good efficacy. Although ibuprofen and naproxen sodium are both nonsteroidal anti-inflammatory medications (NSAIDs), anecdotally many patients report successful treatment with one NSAID when another has failed. Aspirin has shown efficacy in adults for treatment of acute migraine [12].It is likely effective in the pediatric population as well, but it is generally avoided due to long-standing concerns for precipitation of Reye syndrome in children. In adolescents over 16 years old, however, it is a reasonable option if there are no contraindications.
In one study, acetaminophen was compared to ibuprofen and placebo for treatment of migraine in children and adolescents and found to be effective more frequently than placebo but not as frequently as ibuprofen [10],likely due to its only minimal anti-inflammatory effects. It is a reasonable option for children and adolescents, particularly in those who have contraindications to NSAIDs.
While these over-the-counter medications are generally safe and well tolerated, clinicians should not overlook the potential for toxicity as well as medication overuse headache, and patients should be counseled to avoid use of any of these medications for more than 2 to 3 headache days per week.
Triptans
Studies of efficacy of triptans in treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. However, some have shown efficacy over placebo, and in the past few years have received FDA approval for use in the pediatric and adolescent populations. Clinicians should remember that these medications are vasoconstrictors, so they should not be used in patients with vascular disease or in patients with migraine with brainstem aura or hemiplegic migraine. Additionally, due to the risk of serotonin syndrome, they should not be used in patients on monoamine oxidase inhibitors. It is also important to educate patients to limit use of these medications to 4 to 6 times per month to avoid precipitation of medication overuse headache.
Almotriptan was approved by the FDA in 2009 for use in patients 12 years and older, based on a large randomized controlled trial comparing doses of 6.25, 12.5, and 25 mg with placebo in patients ages 12 to 17 years old. All doses resulted in statistically significant pain relief as compared to placebo, and interestingly, the 12.5-mg dose seemed to be the most effective [13].
Rizatriptan received FDA approval in 2012 for use in patients 6 years and older. In 2 randomized controlled trials in patients 6 to 17 years old, rizatriptan (5 mg for patients < 40 kg, 10 mg for patients ≥ 40 kg) was more effective than placebo in providing pain freedom at 2 hours [14,15].One earlier trial found efficacy only on some measures (weekend treatment, decrease in nausea and functional disability) but no statistically significant difference than placebo in terms of overall efficacy in achieving pain freedom at 2 hours [16].However, this trial had a higher placebo response rate than typically seen in adult triptan trials. In a recent long-term open-label study in patients 12 to 17 years old, rizatriptan was found to be generally safe and well-tolerated with consistent efficacy of 46% to 51% pain freedom at 2 hours over time [17].
A combination pill consisting of sumatriptan and naproxen (Treximet) received FDA approval in May 2015 for patients 12 years and older. This was based on a randomized controlled parallel-group trial in patients 12 to 17 years old using the sumatriptan/naproxen combination in various dose combinations: 10/60 mg, 30/180 mg, or 85/500 [18].All doses were found to be equally effective in providing pain relief at 2 hours as compared to placebo, with a higher chance of sustained pain relief at 24 hours in the group receiving the 85/500 dose. In this trial, and in a long-term open-label safety trial [19],all doses were generally well tolerated with minimal adverse reactions.
Most recently, in June 2015, zolmitriptan nasal spray was approved for patients 12 years of age and older. In the large “Double-Diamond” study, which used a novel design to attempt to minimize placebo effect, zolmitriptan nasal spray (5 mg) was found to have a significantly higher headache response rate at 1 hour than placebo, and was significantly superior to placebo with regard to multiple secondary end-points [20].Additionally, a long-term open-label trial in patients 12 to 17 years old using oral zolmitriptan (2.5 mg or 5 mg) found it to be generally effective and well tolerated [21].
No other triptans have been approved for children or adolescents, however, most are widely used in clinical practice. There is good evidence for the efficacy and tolerability of sumatriptan nasal spray [22–26]in adolescents, and it is approved for use in adolescents with migraine in Europe. Although oral sumatriptan was the first triptan available clinically in the United States and is very widely used, there is surprisingly little published evidence for its use in the pediatric and adolescent populations. In fact, 2 randomized controlled trials in adolescents failed to show efficacy of oral sumatriptan as compared to placebo [27,28].Despite this, given its availability it is a reasonable choice for adolescent patients and is often one of the first tried. There has been 1 randomized controlled trial in adolescents for eletriptan without significant differences in efficacy at 2 hours as compared to placebo, although similar to other trials the placebo rate was high and there were some differences seen in secondary outcomes measures [29].Similarly, one randomized controlled trial of naratriptan 12.5-mg tablets failed to show efficacy as compared to placebo in pain relief [30].At present time, for frovatriptan there has only been a study looking at the pharmacokinetics and safety in adolescents, which found that it was generally well tolerated and recommended adolescent dosing similar to adult dosing [31].
In choosing a triptan, clinicians should keep in mind availability of alternate forms of administration, absence or presence of significant emesis, and the age of the patient. For patients who are unable to swallow pills or who have significant emesis associated with their migraines, nasal sprays and oral dissolving forms (melts) are good options. The nasal sprays (zolmitriptan and sumatriptan) additionally have the benefit of a quicker onset (~15 minutes) in general than the oral formulations. The downside to these nasal formulations is bad taste, which is frequently reported by patients. Patients should be counseled in proper administration of these nasal sprays (ie, avoiding inhalation, which causes the medication to enter the mouth) to minimize the bad taste and maximize absorption through the nasal mucosa. Other alternatives are the oral dissolving forms (rizatriptan and zolmitriptan). Given the FDA approval, the rizatriptan melt tablets are often the first-line triptan for children under age 12, but zolmitriptan melts are an option as well.
Preventive Treatment
General Approach
The decision to place an adolescent on a daily preventive medication should be based on a combination of headache frequency, severity, ease of breaking headaches, and overall disability as established by a disability scale (such as the PedMIDAS). Any patient with headaches occurring one or more days per week, those whose headaches are not easily treated or tend to be prolonged, and those with a PedMIDAS score of 30 or more should be considered candidate for a daily preventive. It is particularly important to consider starting a preventive early in adolescent patients given the possibility of impacting overall disease progression at a young age. While the natural history of headaches that start in the young is still being investigated, a known risk factor for transformation of episodic to chronic migraine is frequent headaches [32].It is therefore imperative to attempt to intervene early to improve quality of life in the present and also to prevent a downward cycle into chronicity, potential medication overuse, and worsening disability.
At present, there are several classes of medications that are commonly used for migraine prevention, including antidepressants, antiepileptics, antihistamines, and antihypertensives. Patients should be educated regarding the medication’s typical use, the specific way in which it is used in migraine, potential side effects, and overall expectations of efficacy. Most preventives need to be titrated up slowly to maximize tolerability, and patients need to understand that it may take time before they start to see results. In general, we recommend titrating up over the course of 4 to 12 weeks (depending on medication and dose goal), and then a substantial trial of 4 to 6 weeks on a full dose of medication before determining efficacy. If a medication shows a trend towards improvement but the patient has not met treatment goals, medication can be titrated up further at that point as tolerated. Patients and families should also understand that these medications are not intended as a “cure” for migraine but rather as a tool for improvement, which should be used in conjunction with the rest of a detailed plan. Generally, if a patient is well controlled for 4 to 6 months (ie, 3 or less headaches/month that are easily broken with medications, and a PedMIDAS < 30), attempts should be made to wean off of medication.
Medications
First-line Therapies
Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older (approved in March 2014). Three randomized double-blind placebo-controlled trials in children and adolescents have found topiramate to be superior to placebo in multiple endpoints [33–35].Doses in these trials were 2–3 mg/kg/day, 100 mg/day, and 50 or 100 mg/day, respectively. Notably, the 50 mg/day dose was not found to be superior to placebo. Multiple retrospective, open-label, and drug comparison trials have shown effectiveness and tolerability as well [36–42].We typically use a goal dose of 2 mg/kg/day, which is reached after titrating over 8 to 12 weeks, which minimizes side effects (most commonly paresthesias, memory/language dysfunction, appetite suppression, and drowsiness), but higher doses can be used if needed.
Amitriptyline has consistently shown efficacy in adult migraine trials and is therefore one of the most commonly used medications worldwide for prevention of migraine in children and adolescents. Surprisingly, there have been no published randomized controlled trials using amitriptyline in the pediatric population, although a trial comparing amitriptyline, topiramate, and placebo is currently underway (Childhood and Adolescent Migraine Prevention Study) [43].In 2 retrospective pediatric studies improvement with amitriptyline was reported in 84.2% and 89% of patients, respectively [44,45].We typically use a goal dose of 1 mg/kg/day, also with an 8- to 12-week titration. The most common side effects with amitriptyline are somnolence, dry mouth, and weight gain, but it is generally well tolerated in children and adolescents. There is also a risk of worsening depression and suicidal thoughts, so it is recommended to use caution if considering prescribing to a patient with underlying depression. It is typically administered in once daily dosing a few hours before bed to minimize morning drowsiness. There is also a concern for precipitation of arrhythmias, and while there are no guidelines recommending screening ECGs, this should be considered in patients with a family history of heart disease. Of note, many practitioners use nortriptyline in place of amitriptyline as it can be less sedating. It should be noted, however, that evidence for its efficacy is lacking. Additionally, it may carry a higher risk of arrhythmia [44].
Second-line Therapies
The second-line therapies typically considered are other antiepileptics, valproic acid, levetiracetam, and zonisamide, for which there is some evidence, although mostly in the form of open-label or retrospective studies [46–51]. Of note, despite the many promising retrospective and open-label studies for valproic acid [46–48], one randomized double-blind placebo control trial comparing various doses of extended release divalproex sodium with placebo in adolescent patients failed to show a statistically significant treatment difference between any dose and placebo [52].It is, however, frequently prescribed and anecdotally quite efficacious. Given concerns about potential for teratogenicity, and possible effects on ovarian function, as well as potential for weight gain and hair loss, it should be used with caution in adolescent females.
Antihypertensives (beta blockers and calcium channel blockers) have long been used for prevention of migraine in both the adult and pediatric population, but evidence for their use in the pediatric population is conflicting. An early double-blind crossover study of propranolol in patients 7 to 16 years old showed significant efficacy as compared with placebo [53].However, in 2 subsequent studies it failed to show efficacy as compared with placebo and self-hypnosis, respectively [54,55].Given the conflicting evidence and the potential for hypotension, depression, and exercise-induced asthma, use of propranolol has fallen out of favor by experts for use in pediatric and adolescent migraine prevention. Flunarizine, a nonselective calcium channel blocker, has demonstrated effectiveness in pediatric migraine prevention [56,57],and is actually approved in Europe for this indication, but it is not available in the United States.
Cyproheptadine is an antihistamine with antiserotonergic properties which is used frequently for migraine prevention in young children who are unable to swallow tablets, although evidence is limited to 1 retrospective study [45].However, given the propensity for weight gain with this medication, it is generally not recommended for use in adolescents.
In recent years, there has been a growing interest in nutraceuticals given their general tolerability and minimal side effects, as well as a comfort in using a more “natural” approach, particularly in pediatrics. Evidence is limited but some of the frequently used substances may be beneficial. Butterbur (petasites hybridus) has strong evidence in adults and is recommended by the AAN for migraine prevention in adults [58].There is one small pediatric RCT showing efficacy for butterbur as compared to music therapy [59],as well as one promising open-label trial [60]. Typical dosing ranges from 50–150 mg daily. However, butterbur contains pyrrolizidine alkaloids, which are hepatotoxic, and due to the concerns for inadequate monitoring of removal of these substances in the manufacturing of commercial butterbur, it has been generally avoided in the pediatric population. Coenzyme Q10 (CoQ10) is considered possibly effective for adult migraine prevention by the AAN [58].Pediatric evidence is limited to an open-label study showing improvement with supplementation of CoQ10 in deficient patients [61],and a subsequent double-blind placebo-controlled add-on study, which showed improvement in both the CoQ10 and placebo groups but faster improvement in the CoQ10 group [62].Typical pediatric dosing is 1–2 mg/kg/day. Magnesium is considered by the AAN to be a good option for migraine prevention in adults [58].One randomized controlled trial in children however had equivocal results [63],while one small prospective open-label study had positive results [64]. Magnesium supplementation may be more effective in patients who have low ionized magnesium levels, but this is difficult to measure reliably in the clinical setting. Doses of 9 mg/kg/day can be used with the most common side effects reported being gastrointestinal upset and diarrhea, generally dose dependent. Riboflavin is also considered by the AAN to be probably effective for prevention in adults [58],but again the evidence in children is limited to one positive retrospective study [65]and 2 equivocal randomized controlled trials [66,67],one of which had an unusually high placebo rate [66]. Appropriate dosing is also something of debate, as riboflavin is minimally absorbed and has a short half-life, so while studies were done using 200–400 mg daily, smaller more frequent dosing may be needed.
A potential approach to treatment with vitamins is to check for deficiencies, but currently only the study mentioned above in which CoQ10 levels were checked showed improvement with normalizing of low levels [61,62].Further research into this topic is needed to elucidate whether checking and repleting levels of specific vitamins would be beneficial in prevention of migraines in certain patients.
Healthy Habits
The importance of maintaining healthy lifestyle habits and modifying potentially detrimental ones should be stressed to patients and families, and counseling regarding these issues should be provided at every visit, as repetition is often key to patients understanding their importance. Skipping meals is a commonly reported migraine trigger [68,69].This is not an uncommon occurrence even in the most well-meaning of families; adolescents often report not feeling hungry in the morning, and in the rush to get to school will often skip breakfast. Many patients report not liking school lunches leading them to go most of the day without food. Adolescent girls may skip meals due to weight concerns. Patients need to be reminded that well-balanced meals throughout the day is imperative, and they often may need specific counseling on how to achieve this practically. Of note, unless a specific migraine trigger has been identified in a given patient, we do not generally recommend restricting any specific food lists.
Maintaining good sleep hygiene and a consistent sleep schedule is also often difficult for adolescent patients, with after-school activities, homework, and screen use (eg, television, electronic devices) often contributing to late bedtimes with then forced early morning waking for school. However, improvement in sleep hygiene has been shown to be effective in improving migraines in children and adolescents [70],and realistic plans for improvement in sleep should be discussed with patients.
Dehydration is also a common migraine trigger [8,9],so the importance of staying well hydrated should be stressed as well. Again, specific recommendations for how this can be achieved are often needed, especially given adolescents’ busy schedules. Additionally, many schools do not allow water bottles to be carried, and often a school note is needed so patients may be allowed to carry a water bottle at school and also be provided extra bathroom breaks as needed.
Also important is stressing maintenance of daily functioning throughout migraine treatment. By the time adolescents seek medical care, they may already be in a cycle of missing school due to headaches, and some may even be receiving home-schooling. The goals of staying in school and learning to function with headaches should be stressed, often with the help of coping skills, which will be discussed below, as it has been shown that functional disability generally improves before pain [71].
Behavioral Treatments
Psychological treatments are an important aspect of migraine management. Biobehavioral treatments such as relaxation training, biofeedback, and multimodal cognitive behavioral therapy (CBT) have been evaluated in randomized controlled trials and reviewed in meta-analyses in pediatric migraine populations and found to be efficacious [72–75].These treatments have been shown to reduce pain intensity and disability for children and adolescents with headaches and therapeutic gains appear to be maintained [72].
Relaxation training typically includes instruction in techniques such as diaphragmatic breathing, progressive muscle relaxation, and guided imagery. Relaxation training is most effective with children 7 years of age or older [76].Biofeedback is often used in conjunction with relaxation training to provide audio or visual feedback about normally unconscious physiological body responses associated with increased relaxation. Effective biofeedback parameters used with children and adolescents with migraine headache include electromyographic (EMG) activity and peripheral skin temperature [77].Biofeedback techniques can help children and adolescents become more aware of physical responses, better control these responses and generalize physical responses outside of therapy sessions to better cope with pain [76].CBT involves instruction in skills such as biofeedback-assisted relaxation training, activity pacing, distraction, and cognitive strategies for coping with pain. CBT was shown to be effective in a recent study comparing adolescent patients with chronic migraine receiving amitriptyline and CBT with patients receiving amitriptyline and standard headache education [78]. Patients who received CBT plus amitriptyline had greater reductions in days with headache and migraine-related disability compared with patients who received headache education plus amitriptyline [78].Hypnosis and acceptance and commitment therapy (ACT) are also psychological treatments used with children and adolescents with migraine headaches. ACT prioritizes the outcome of improved functioning above headache reduction and has broadly demonstrated efficacy for chronic pain [79]. Both treatments have shown promising benefit but there has been less evidence supporting the use of these therapies in pediatric headache than other well-established behavioral treatments.
The presence of comorbid psychiatric issues such as anxiety, depression, or ADHD can make the treatment of patients with migraine headaches more complex. A recent study found that approximately 30% of a sample of children and adolescents with chronic daily headache had a lifetime psychiatric diagnoses and having a lifetime psychiatric diagnosis was associated with poorer headache-related disability and quality of life [80].As a result, more intensive behavioral treatment for children and adolescents with a psychiatric comorbidity may be needed to focus on emotional and behavioral issues.
Pediatric psychologists can assess pain-related disability and coping difficulties and treat children and adolescents with migraine headaches. Children are often referred to a psychologist or other mental health professional if headaches are severe or impairing functioning. Unfortunately, access to this therapy is sometimes limited, but when available, should be offered to any migraine patients with significant disability requiring prevention, and specifically to patients with chronic migraine.
How should the patient be treated?
Acute Treatment
The patient reports only minimal response to ibuprofen. However, he is only taking 200 mg and does not take it until well after the headache has started. He should be instructed to take ibuprofen 600 mg (~10 mg/kg) as soon as the headache starts, along with 32 oz of a sports drink, and told that he should repeat this dose in 4 hours if his headache has not completely resolved with the first dose. As his headaches are occurring less than 3 times per week on average, he can do this with every headache. He should be given a note for school allowing him to receive the medication at school, and noting the importance of allowing him to get the medication as soon as he reports a headache. Given that some of his headaches occur on consecutive days, we suspect some may be continuations of a prior headache, so again the importance of obtaining complete pain relief from his acute medication should be stressed. If at his next follow-up visit he reports not always having complete relief with ibuprofen, we would consider trying naproxen instead, and/or adding a triptan, most likely sumatriptan 100 mg given its availability. He would need to understand that he may only use the triptan for 4 to 6 headache days/month. Depending on his response to sumatriptan, it could be used alone or in combination with his NSAID. He also may be instructed to use an NSAID alone for more mild headaches and his NSAID together with sumatriptan for more severe headaches.
Preventive Treatment
PedMIDAS score is a 25, indicating mild disability, however his migraines are frequent, occurring 1–3 times per week and clearly having a significant impact on his functioning. We would therefore recommend starting a prophylactic medication. He notes difficulty remembering to take medications as well as difficulty falling asleep, so amitriptyline would be a good choice for him. For his weight of 50.9 kg, we would start with at 12.5 mg at night and increase by 12.5 mg every 2 weeks to a dose of 50 mg (~1mg/kg/day). He and Mom should be counseled that it will likely take at least a few weeks on his goal dose before they start to see results. He should be counseled also to report to Mom immediately if he has any depressive or suicidal thoughts. Topiramate is another option, especially as it is FDA-approved for migraine prevention in children older than 12 years, although twice daily dosing may be a factor in maintaining compliance. Careful discussion with the patient and family in regards to the potential risks and benefits is important prior to starting any preventative medication.
Biobehavioral Management
The patient should be drinking 8–10 cups of non-caffeinated fluid per day and additional cups on days he plays soccer. He should be given a school note to this effect so he may carry a water bottle at school. He eats well, but the importance of not skipping breakfast should be stressed, and ideas for fitting this in should be given. The importance of a consistent bedtime routine should be stressed, with good sleep hygiene to assist with easier sleep onset. It should be made clear that he should not be using screens within an hour prior to bed. He already exercises frequently and should be commended for being active. Given his report of frequent headaches which are impairing school attendance, functioning at school, and participation in soccer activities, screening for potential co-morbid psychiatric issues and making a referral for further treatment with a pediatric pain psychologist focused on coping with chronic pain is appropriate.
Case Study 2
Initial Presentation and History
A 16-year-old right-handed girl who has been having headaches since she was 10 years old presents for evaluation. She reports unilateral mostly right-sided headaches, which are throbbing and associated with photophobia, phonophobia, osmophobia, nausea, and occasionally vomiting. She has no premonitory or aura symptoms. Her headaches typically occur once a week but she has noted that they tend to be more frequent around the time of her menses and that often these headaches do not fully respond to naproxen, which typically works to break her migraines during other times in the month. Upon further review, she believes these headaches typically start on the day or two prior to her menses, and that this happens almost every month. She often misses school due to these headaches. Her mother would like to know whether the patient should see a gynecologist to potentially be placed on birth control to control her headaches.
Physical Examination
On examination, weight was 61.5 kg (75.8 percentile), height 157.1 cm (18.4 percentile), BMI 24.91 (84 percentile), blood pressure 112/55 mm Hg, and heart rate 90 bpm. Her general physical and neurologic exam results were normal. She had tightness over the left trapezius muscles, otherwise the remainder of headache examination was unremarkable.
What is the probable diagnosis?
According to the ICHD-IIIβ, the patient meets criteria for episodic migraine without aura. Based on the history, she likely also has menstrually related migraine without aura [5].Officially, however, 3 months of prospective documentation is needed to make this diagnosis. Menstrually related migraine is included in the appendix of the ICHD-IIIβ, meaning there is ongoing debate about how it should be classified. According to the current appendix criteria, headaches should meet criteria for migraine without aura and also have documented and prospectively recorded evidence over at least 3 consecutive cycles with headaches confirmed on day 1+/– 2 (ie, 2 days prior to onset of menstruation or within the first 3 days of menstruation) in at least 2 out of 3 menstrual cycles, with migraines occurring during other times of the month as well [5]. This is distinguished from pure menstrual migraine without aura, in which migraines occur only during days 1+/–2 of menstruation but not during other times of the month [5].While we therefore cannot say that the patient meets ICHD IIIβ criteria for menstrually related migraine due to the lack of prospective documentation, her history suggests this.
Menstrually Related Migraine
An association of migraine with menses is well described in adults, occurring in up to 60% of adult female migraine patients [81].In adults, it has been observed that the migraines associated with the menstrual cycle tend to be more severe [82],associated with more nausea and vomiting [82],longer and less responsive to acute medications [83],and associated with more work-related disability [83]. Recently, analysis of data from the American Migraine Prevalence and Prevention (AMPP) Study found that women with pure menstrual migraine and menstrually associated migraine have on average higher MIDAS scores than those with non-menstrual migraine [81].
Only one study has explored menstrually related migraines in adolescents [84].It was a clinic-based study that found a similar prevalence—50% of adolescent patients who had reached menarch noted an association of migraine with their menses. Overall worse disability was not demonstrated in this study, but individual menstrual attacks were not compared with nonmenstrual attacks [84].No other studies have addressed this population, but it is clear that the pattern of menstrually related migraine exists in adolescents and it is important to recognize this pattern as these patients may require additional focused care in addition to standard migraine management.
What are treatment options?
In adults, options for more specific management of pure menstrual migraine and menstrually related migraine include intermittent prophylaxis with NSAIDs or triptans or use of hormonal contraceptives. There are no studies addressing any of these treatments in adolescents, so at this point management decisions must be based on evidence extrapolated from adult studies as well as attention to specific concerns in adolescent patients.
Generally, intermittent prophylaxis (using a medication a few days prior to and during the first few days of menses) with various medications has shown efficacy in adult studies. This approach may be more appropriate for patients with pure menstrual migraine, as there is less likelihood of precipitating medication overuse in this population. It can be considered in patients with menstrually related migraine as well if typical daily preventives have not been effective. One small open-label trial using naproxen 550 mg daily prior to and during menses (at differing schedules depending on the month) showed slightly decreased frequency and severity of headaches during that time [85].However, triptans are what are most commonly used.
Triptans
Frovatriptan is the longest-acting triptan and the one used most commonly for intermittent prophylaxis. One double-blind randomized controlled crossover study showed improvements in headache frequency, severity, and duration using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses [86].Frovatriptan 2.5 mg daily showed some efficacy as well but was not as effective as 2.5 mg BID [86].One prospective randomized placebo-controlled trial demonstrated more headache-free cycles using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses, as compared to placebo [87].
Naratriptan, also a longer-acting triptan, was studied in 3 prospective double-blind trials at doses of 1 mg BID used for 5 days total starting 2 days prior to menses, and all 3 showed a decrease in headache frequency as compared to placebo [88,89].Of note no efficacy was shown using 2.5 mg daily [88].
Less commonly used for intermittent prophylaxis are the shorter-acting triptans, but they have shown some efficacy as well. In one prospective study using oral sumatriptan 25 mg TID for 5 days each cycle starting 2 to 3 days before onset of menses, there seemed to be at least some improvement with sumatriptan in most cycles treated [90].Zolmitriptan was studied in one prospective double-blind placebo-controlled study using 2.5 mg BID or 2.5 mg TID for 7 days total with each cycle and was found to be associated with a significant reduction in headache frequency as compared with placebo [91].There has been one prospective trial using eletriptan 20 mg TID for 6 days total starting 2 days prior to menses, which also showed improvement in headache activity in 55% of patients [92].
Hormonal Contraceptives
In general for adult patients, hormonal contraceptives are not considered first-line treatment for menstrually related migraine. However, in patients who do not respond to other modes of treatment, or who plan to use hormonal contraception for contraceptive purposes, published expert opinions have recommended considering extended or continuous hormonal regimens [93,94].
Estrogen withdrawal during the luteal phase of the monthly cycle has long been speculated to be of importance in the pathophysiology of menstrually related migraine [93]and the relationship between hormonal contraceptives and migraine is complicated. Headache is a commonly reported side effect of combined hormonal contraceptives [95]; however, this effect seems to occur mostly during the hormone-free week [96]. It has been shown that headache occurs less frequently in women using an extended cycle regimen (84 or 168 days) as compared to those using a traditional monthly cyclic regimen [97,98].Studies addressing the use of combined hormonal contraceptives for women specifically with menstrually related migraine are limited. One small prospective randomized study showed improvement in menstrually related migraine in patients treated with a low dose (20 mcg ethinyl E[2]) oral hormonal contraceptive in both a 21/7 cycle and a 24/4 cycle, with more improvement in the group using a 24/4 cycle [99].Another showed improvement in menstrually related migraine in all study patients treated with a low-dose hormonal contraceptive (20 mcg ethinyl estradiol) on a 21/7 regimen, but with additional 0.9 mg conjugated equine estrogen during the placebo week [100].There has been one randomized placebo-controlled double-blind trial in patients with menstrually related migraine using an extended 168 hormonal regimen along with frovatriptan vs. placebo for 5 days during the hormone-free interval. Overall daily headache scores were decreased from pre-study cycles, and the increase in headaches during the hormone-free interval was lower in the frovatriptan group [101].A recent study showed that contraceptive-induced amenorrhea can be beneficial for decreasing migraine frequency in patients with menstrual migraine [102].There have been no studies addressing the use of hormonal contraceptives for migraine management in adolescents.
In adolescents, the decision regarding use of hormonal contraceptives is more complicated. There is less of a chance that adolescent patients, especially younger ones, would be planning to use hormones for contraception so their use may be solely for the purpose of migraine management. However, hormonal contraceptives are commonly used in adolescents for management of other menstrual-related disorders, such as menorrhagia, dysmenorrhea, and endometriosis, and extended cycle and continuous regimens have become more popular with adolescent providers in general [103].The general concern with using hormonal contraceptives in adolescents is that they have potential for longer-term use than adult patients, and the effects of using hormonal contraceptives long term are unknown. The major concerns are for potential interference with expected increase in bone mineral density in adolescents, effects on fertility, and risk for cancer and cardiovascular disease [103].Additionally, specifically in migraine patients, is the concern for increased stroke risk. The increased risk for ischemic stroke in patients with migraine, although more specifically with migraine with aura, is well known [104–106]and migraine has recently been shown to be a risk factor for ischemic stroke in adolescents as well [107]. In adults, the use of hormonal contraceptives in patients with migraine with aura is known to increase the risk of ischemic stroke [106],and this has not been studied in adolescents. Given the various unknowns in the use of hormonal contraceptives in patients with menstrually related migraines, we would not recommend this as first-line treatment. However, similarly to adults, in patients who do not respond to other methods of migraine management, or who seek to use hormonal contraceptives for contraception or for other menstrually related disorders, extended or continuous cycle hormonal contraceptives may be a reasonable option, at the lowest possible estrogen dose. However, migraine with aura should be screened for, and its presence should prompt reconsideration of combined hormonal contraceptive use.
How should this patient be managed?
The patient is having frequent and disabling migraines, so starting a preventive medication would be appropriate. She has migraines throughout the month in addition to during her menses, so a daily prophylactic would be more appropriate than intermittent prophylaxis surrounding her menstrual cycle only. At this point, our recommendation would be to start a daily preventive with either amitriptyline or topiramate. Given that naproxen is not breaking some of her migraines, she should be given a prescription for a triptan. Sumatriptan 100 mg would be an appropriate first choice, and she can be instructed to use it along with her naproxen at the onset of her menstrual migraines. She can use it for other migraines as well but she should be instructed not to use it more than 4 to 6 times per month. She should keep a diary for the next 3 months noting most importantly headache days as well as days of menstruation, so that a more definitive pattern can be confirmed and an official diagnosis based on ICHD-IIIβ criteria can be made. If her migraines do not improve with daily preventives, at that point discussion regarding potential for intermittent prophylaxis or trial of extended cycle hormonal contraception may be considered, although with caution and discussion of risks and benefits.
Corresponding author: Hope O’Brien, MD, Cincinnati Children’s Hospital Medical Center, 3333 Burnet Ave., MLC 2015 Cincinnati, OH 45229.
Financial disclosures: None.
From the Cincinnati Children’s Hospital Medical Center, Cincinnati, OH.
Abstract
- Objective: To review the management of migraine in adolescent patients.
- Methods: Literature review in the context of 2 clinical cases.
- Results: Migraine is common in adolescents and can affect school and social functioning. Management options include lifestyle modifications and acute and preventative therapies. First-line medications for migraine in the adolescent population are over-the-counter medications, including ibuprofen, acetaminophen, and naproxen. Studies of efficacy of triptans in the treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. Several classes of medications are commonly used for migraine prevention, including antidepressants and antiepileptics. Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older. Biobehavioral treatments such as relaxation training, biofeedback, and cognitive behavioral therapy have been evaluated in randomized controlled trials and found to be efficacious.
- Conclusion: Approach to management of migraine in adolescents should be multifactorial with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Case Study 1
Initial Presentation
A 13-year-old left-handed boy with allergic rhinitis has been referred by his pediatrician for evaluation of headaches.
History
The headaches have been occurring since he was 7 years old. He describes a bilateral frontal and periorbital pain, which is either aching or throbbing in nature. There is associated photophobia and phonophobia, and the pain worsens with activity. When he was younger the headaches were frequently associated with emesis. He occasionally gets tenderness of his face during the headaches, mostly in the areas adjacent to his nose and above his eyes and occasionally associated rhinorrhea.
Initially the headaches were mild and occurring infrequently, but for the past 6 months they have been more severe and occurring 1 to 3 times per week, sometimes on consecutive days. They typically begin in the afternoon but at times occur soon after waking in the morning. If they do occur in the morning, they worsen after getting out of bed. He denies any type of warning symptoms indicating the headache will occur, and has no associated focal neurologic symptoms associated with the headaches. His mother tries to minimize medication intake so will typically wait to see if the headache is severe before giving him medication, typically 2 chewable children’s ibuprofen, which helps some of the time; however, his headache often does not completely abate unless he naps. He has also been taking loratadine daily due to concerns that his headaches may be secondary to chronic sinusitis.
He is a good student and enjoys school, but notes stress surrounding tests. He reports missing 4 days of school in the past 3 months as well as coming late twice. He thinks that there were 9 additional days in which he was unable to function at his full ability at school and at least 5 times he was unable to concentrate on his homework. He plays soccer 4 to 5 times per week. He says twice he had to skip soccer practice to due to headache and at least 3 other times he needed to take breaks during soccer due to headache. He is unsure how much he drinks on an average day but he drinks mostly with meals. He does not drink caffeine. He gets into bed at 10 pm and sometimes does not fall asleep until after 11 pm, often due to worry about tests. He often plays with his tablet when trying to fall asleep. He wakes up at 6 am for school. He typically eats 3 meals per day but occasionally misses breakfast if he is rushed in the morning.
Physical Examination
On examination, the patient is a well-developed, well-nourished male in no apparent distress. His weight was 50.9 kg (68th percentile), height 163.7 cm (65th percentile), BMI 19.9 (69th percentile), blood pressure 118/62 mm Hg, and heart rate 79 bpm. His general physical examination, including skin, HEENT, extremities, lung, cardiac, and abdominal examination was normal, with lungs clear to auscultation, heart with a regular rate and rhythm, and abdomen soft and tender without organomegaly.
On neurologic examination he was alert and attentive with normal mental status. Speech was fluent. Skull, spine, and meninges were normocephalic and atraumatic with a supple neck. Cranial nerves II through XII were normal with a normal fundoscopic examination including sharp disk, no papilledema, and normal fundus bilaterally. Motor examination was normal for tone and bulk with full strength throughout. Sensory examination was normal for light touch, temperature, vibration, and joint position sense. Finger-nose-finger fine finger movement and heel-knee-shin were normal. Deep tendon reflexes were symmetric and 2+ throughout with toes downgoing. Station and gait were normal including toe walking, heel walking, tandem walking, running, skipping, one-legged standing, Fog gate procedure, and there was no Romberg’s sign.
He had a normal comprehensive headache examination. There was no tenderness at typical migraine trigger points (nuchal line and mandibular process) and no tenderness at supraorbital notch. Neck was supple with normal rotation and normal bilateral trapezius muscle tightness. No bruits or palpitations were heard over carotid or jugular veins. There was a negative Muller sign with no pain on neck bending with pressure, and no signs of allergy or sinus symptoms.
What is the initial evaluation of a teen with recurrent headaches?
The overwhelming majority of children and adolescents presenting to medical attention with recurrent headaches will have a primary headache disorder, most commonly migraine [1].However, ruling out a secondary cause is typically of concern, both for providers and parents, and the decision regarding whether to image is often a daunting one. In a review of 6 large studies in which imaging was done in pediatric patients who were examined by a neurologist, only 14 patients were found to have CNS lesions requiring intervention, and all of these patients had abnormalities on physical exam [2]. An American Academy of Neurology (AAN) practice parameter published in 2002 recommended considering neuroimaging only in patients with abnormal neurologic examination, co-existing seizures, concerning associated neurologic features, or recent onset of severe headache or change in headache type [2].There are additional factors which may prompt one to consider imaging as well including atypical auras or very short (< 5 minutes) or protracted ( > 60 minutes) auras, trigeminal autonomic cephalalgia, brief headaches precipitated by cough, frequent early morning headaches or which wake the patient from sleep, headaches with primarily occipital location, headaches in a patient less than 6 years old or in a child who cannot describe the headaches well, or migrainous headaches in a child without any family history of migraine or migraine equivalents. However, recent studies continue to show that the imaging yield in patients with headaches is low and further studies are needed to elucidate which patients truly require imaging [3].
Given this, a thorough history, family history, physical exam, and detailed neurologic exam including fundoscopy are imperative. In addition to the general neurologic exam, a focused headache exam should be performed, including Mueller’s maneuver, auscultation for cranial bruits, evaluation of the temporomandibular joint, palpation of possible trigger points, and maneuvers to assess for cervical spine disease [4].
Classification of Symptoms
Once establishing the likelihood of a primary headache disorder, the International Classification of Headache Disorders, 3rd edition, beta version (ICHD IIIβ) [5]should be used to classify the headache diagnosis. Note that while this classification system was established for adults, most criteria are similar for children and adolescents, and the typical differences are noted in the comments of the ICHD IIIβ [5].Migraine is the most common type of primary headache brought to medical attention [1]. Migraine without aura is described as a recurrent headache disorder (at least 5 lifetime attacks) with attacks lasting 4 to 72 hours, typically unilateral, throbbing in nature, moderate to severe in intensity, aggravated by routine physical activity, and associated with nausea and/or photophobia and phonophobia. In young children, migraine is more frequently bilateral, the gastrointestinal symptoms often more pronounced than photophobia and phonophobia, and migraines may be shorter, lasting at least 2 hours without treatment [5].As patients reach adolescence, migraine features typically start to evolve into patterns described in adults. Differentiating migraine from other primary headaches, such as tension-type, can be challenging, especially in children in whom migraines are more likely to be shorter and bilateral. Tension-type headaches are bilateral or diffuse, pressing or tightening pain that is non-pulsatile lasting at least 30 minutes. The pain is described as mild to moderate in intensity and not aggravated by activity [5].They may be associated with photophobia or phonophobia (not both) and they may not be associated with nausea or vomiting [5].
Is any further workup indicated?
The patient has recurrent headaches which have been present for 5 years. While they have increased in frequency, there has not been any change in their quality. His headache description and history have no “red flag” features, and a thorough examination is normal. Therefore, neuroimaging and further workup would not be indicated in this case.
What is the diagnosis?
The patient’s headaches are throbbing in nature, exacerbated by activity, associated with nausea, photophobia, and phonophobia, and are moderate to severe. Using the ICHD IIIβ, he meets criteria for migraine without aura. While his headaches are frequent, he is having less than 15 headache days per month, so this is episodic migraine. Note that he complains of forehead and periorbital pain and occasional rhinorrhea with his headaches, leading him to have been placed on loratadine for treatment of presumed allergic sinusitis. Children meeting criteria for migraine are very frequently misdiagnosed as having sinusitis [6]due to the overlap in location of migraine pain and proximity to the frontal and maxillary sinuses, as well as the presence of autonomic features frequently present in migraine, reportedly present in 70% of pediatric migraine patients [7].The negative headache examination, including Muller’s maneuver, points against sinus disease as well.
What is the approach to management of the adolescent with migraine?
Approach to management of migraine in adolescents should be multifactorial, with attention to an aggressive acute treatment regimen, preventive medications when indicated, and biobehavioral management.
Acute Treatment
General Approach
In counseling patients with migraine, and in particular adolescents, it should be stressed that achieving a normal level of functioning as soon as possible is the goal of therapy. Common missteps in treatment include failure to take acute medication early into the headache, incorrect dosing, incompletely treating the headache, and avoidance of participating in daily activities when headaches occur.
Adolescent patients may frequently wait to take an acute medication, typically due participation in another activity, not having medication with them, or discomfort with taking medication in front of peers or at school. Additionally, patients who have headaches beginning in early childhood, with pronounced gastrointestinal (GI) features, may be aware that their headaches resolve after vomiting, and therefore get used to not treating with medications. When these patients reach adolescence, when GI symptoms tend to become less pronounced, they will need to be educated that taking medication early is imperative. Acute medications are typically more effective when taken earlier in the course of a migraine, and the importance of pausing to take medication at the onset of the headache should be stressed to all patients and parents. With that in mind, however, care should be taken to counsel patients regarding the potential for development of medication overuse headache. When headaches are frequent, a more detailed and intricate plan may need to be devised so that adolescents and parents know which headaches to treat with medication.
Given that first-line medications for treatment of migraines are over-the-counter, underdosing occurs commonly, as dosing listed on packaging is typically age-based, not weight-based. At initial visits, young adolescent patients will frequently report that a particular medication is not effective, but this is often because they are still receiving low/less optimal dosing. Clinicians should remember to follow weights and recommend dosing changes at the initial visit and follow-up visits as well.
Treatment goals, ie, complete resolution of pain and migrainous features with ability to return to normal functioning, should be made clear to patients and families at the onset of treatment. Patients frequently fall into a pattern of continuing to treat with a medication that may lessen but not completely ablate the pain of a headache, and then sleep and avoid activity. Upon awakening, the headache may be gone, however, given incomplete initial treatment, the headache may be more likely to recur within 24 hours. At that point, the headache may be more difficult to treat and therefore cause further decrease in functioning. If this cycle perpetuates, disability can become extremely burdensome in adolescents, significantly affecting school and social functioning. Therefore, a detailed plan for initial steps in management as well as steps to take should initial therapy fail to fully break the headache should be given to every patient.
Of note, given that dehydration is now recognized as a common trigger for migraine [8,9], it is generally our recommendation to drink 16 to 32 oz (depending on weight) of a hydrating fluid together with whichever acute medication is chosen, as rehydration likely assists in breaking migraines as well [8].
Medications
Over-the-Counter Agents
The first-line medications for migraine in the pediatric and adolescent populations are over-the counter medications, including ibuprofen, acetaminophen, and naproxen sodium. Ibuprofen has been well studied in pediatrics and was found to be safe and effective in 2 studies [10,11]at doses of 7.5–10 mg/kg. Again, care should be taken to ensure timely administration of ibuprofen at onset of headache, or aura if present, with appropriate weight-based dosing. Naproxen sodium has not been studied in pediatrics for treatment of migraine. However in practice, it is often used in similar doses of 10 mg/kg, with good efficacy. Although ibuprofen and naproxen sodium are both nonsteroidal anti-inflammatory medications (NSAIDs), anecdotally many patients report successful treatment with one NSAID when another has failed. Aspirin has shown efficacy in adults for treatment of acute migraine [12].It is likely effective in the pediatric population as well, but it is generally avoided due to long-standing concerns for precipitation of Reye syndrome in children. In adolescents over 16 years old, however, it is a reasonable option if there are no contraindications.
In one study, acetaminophen was compared to ibuprofen and placebo for treatment of migraine in children and adolescents and found to be effective more frequently than placebo but not as frequently as ibuprofen [10],likely due to its only minimal anti-inflammatory effects. It is a reasonable option for children and adolescents, particularly in those who have contraindications to NSAIDs.
While these over-the-counter medications are generally safe and well tolerated, clinicians should not overlook the potential for toxicity as well as medication overuse headache, and patients should be counseled to avoid use of any of these medications for more than 2 to 3 headache days per week.
Triptans
Studies of efficacy of triptans in treatment of pediatric migraine have been limited and results conflicting, largely due to high placebo response rates. However, some have shown efficacy over placebo, and in the past few years have received FDA approval for use in the pediatric and adolescent populations. Clinicians should remember that these medications are vasoconstrictors, so they should not be used in patients with vascular disease or in patients with migraine with brainstem aura or hemiplegic migraine. Additionally, due to the risk of serotonin syndrome, they should not be used in patients on monoamine oxidase inhibitors. It is also important to educate patients to limit use of these medications to 4 to 6 times per month to avoid precipitation of medication overuse headache.
Almotriptan was approved by the FDA in 2009 for use in patients 12 years and older, based on a large randomized controlled trial comparing doses of 6.25, 12.5, and 25 mg with placebo in patients ages 12 to 17 years old. All doses resulted in statistically significant pain relief as compared to placebo, and interestingly, the 12.5-mg dose seemed to be the most effective [13].
Rizatriptan received FDA approval in 2012 for use in patients 6 years and older. In 2 randomized controlled trials in patients 6 to 17 years old, rizatriptan (5 mg for patients < 40 kg, 10 mg for patients ≥ 40 kg) was more effective than placebo in providing pain freedom at 2 hours [14,15].One earlier trial found efficacy only on some measures (weekend treatment, decrease in nausea and functional disability) but no statistically significant difference than placebo in terms of overall efficacy in achieving pain freedom at 2 hours [16].However, this trial had a higher placebo response rate than typically seen in adult triptan trials. In a recent long-term open-label study in patients 12 to 17 years old, rizatriptan was found to be generally safe and well-tolerated with consistent efficacy of 46% to 51% pain freedom at 2 hours over time [17].
A combination pill consisting of sumatriptan and naproxen (Treximet) received FDA approval in May 2015 for patients 12 years and older. This was based on a randomized controlled parallel-group trial in patients 12 to 17 years old using the sumatriptan/naproxen combination in various dose combinations: 10/60 mg, 30/180 mg, or 85/500 [18].All doses were found to be equally effective in providing pain relief at 2 hours as compared to placebo, with a higher chance of sustained pain relief at 24 hours in the group receiving the 85/500 dose. In this trial, and in a long-term open-label safety trial [19],all doses were generally well tolerated with minimal adverse reactions.
Most recently, in June 2015, zolmitriptan nasal spray was approved for patients 12 years of age and older. In the large “Double-Diamond” study, which used a novel design to attempt to minimize placebo effect, zolmitriptan nasal spray (5 mg) was found to have a significantly higher headache response rate at 1 hour than placebo, and was significantly superior to placebo with regard to multiple secondary end-points [20].Additionally, a long-term open-label trial in patients 12 to 17 years old using oral zolmitriptan (2.5 mg or 5 mg) found it to be generally effective and well tolerated [21].
No other triptans have been approved for children or adolescents, however, most are widely used in clinical practice. There is good evidence for the efficacy and tolerability of sumatriptan nasal spray [22–26]in adolescents, and it is approved for use in adolescents with migraine in Europe. Although oral sumatriptan was the first triptan available clinically in the United States and is very widely used, there is surprisingly little published evidence for its use in the pediatric and adolescent populations. In fact, 2 randomized controlled trials in adolescents failed to show efficacy of oral sumatriptan as compared to placebo [27,28].Despite this, given its availability it is a reasonable choice for adolescent patients and is often one of the first tried. There has been 1 randomized controlled trial in adolescents for eletriptan without significant differences in efficacy at 2 hours as compared to placebo, although similar to other trials the placebo rate was high and there were some differences seen in secondary outcomes measures [29].Similarly, one randomized controlled trial of naratriptan 12.5-mg tablets failed to show efficacy as compared to placebo in pain relief [30].At present time, for frovatriptan there has only been a study looking at the pharmacokinetics and safety in adolescents, which found that it was generally well tolerated and recommended adolescent dosing similar to adult dosing [31].
In choosing a triptan, clinicians should keep in mind availability of alternate forms of administration, absence or presence of significant emesis, and the age of the patient. For patients who are unable to swallow pills or who have significant emesis associated with their migraines, nasal sprays and oral dissolving forms (melts) are good options. The nasal sprays (zolmitriptan and sumatriptan) additionally have the benefit of a quicker onset (~15 minutes) in general than the oral formulations. The downside to these nasal formulations is bad taste, which is frequently reported by patients. Patients should be counseled in proper administration of these nasal sprays (ie, avoiding inhalation, which causes the medication to enter the mouth) to minimize the bad taste and maximize absorption through the nasal mucosa. Other alternatives are the oral dissolving forms (rizatriptan and zolmitriptan). Given the FDA approval, the rizatriptan melt tablets are often the first-line triptan for children under age 12, but zolmitriptan melts are an option as well.
Preventive Treatment
General Approach
The decision to place an adolescent on a daily preventive medication should be based on a combination of headache frequency, severity, ease of breaking headaches, and overall disability as established by a disability scale (such as the PedMIDAS). Any patient with headaches occurring one or more days per week, those whose headaches are not easily treated or tend to be prolonged, and those with a PedMIDAS score of 30 or more should be considered candidate for a daily preventive. It is particularly important to consider starting a preventive early in adolescent patients given the possibility of impacting overall disease progression at a young age. While the natural history of headaches that start in the young is still being investigated, a known risk factor for transformation of episodic to chronic migraine is frequent headaches [32].It is therefore imperative to attempt to intervene early to improve quality of life in the present and also to prevent a downward cycle into chronicity, potential medication overuse, and worsening disability.
At present, there are several classes of medications that are commonly used for migraine prevention, including antidepressants, antiepileptics, antihistamines, and antihypertensives. Patients should be educated regarding the medication’s typical use, the specific way in which it is used in migraine, potential side effects, and overall expectations of efficacy. Most preventives need to be titrated up slowly to maximize tolerability, and patients need to understand that it may take time before they start to see results. In general, we recommend titrating up over the course of 4 to 12 weeks (depending on medication and dose goal), and then a substantial trial of 4 to 6 weeks on a full dose of medication before determining efficacy. If a medication shows a trend towards improvement but the patient has not met treatment goals, medication can be titrated up further at that point as tolerated. Patients and families should also understand that these medications are not intended as a “cure” for migraine but rather as a tool for improvement, which should be used in conjunction with the rest of a detailed plan. Generally, if a patient is well controlled for 4 to 6 months (ie, 3 or less headaches/month that are easily broken with medications, and a PedMIDAS < 30), attempts should be made to wean off of medication.
Medications
First-line Therapies
Currently, topiramate is the only medication approved for prevention of migraine in patients 12 years and older (approved in March 2014). Three randomized double-blind placebo-controlled trials in children and adolescents have found topiramate to be superior to placebo in multiple endpoints [33–35].Doses in these trials were 2–3 mg/kg/day, 100 mg/day, and 50 or 100 mg/day, respectively. Notably, the 50 mg/day dose was not found to be superior to placebo. Multiple retrospective, open-label, and drug comparison trials have shown effectiveness and tolerability as well [36–42].We typically use a goal dose of 2 mg/kg/day, which is reached after titrating over 8 to 12 weeks, which minimizes side effects (most commonly paresthesias, memory/language dysfunction, appetite suppression, and drowsiness), but higher doses can be used if needed.
Amitriptyline has consistently shown efficacy in adult migraine trials and is therefore one of the most commonly used medications worldwide for prevention of migraine in children and adolescents. Surprisingly, there have been no published randomized controlled trials using amitriptyline in the pediatric population, although a trial comparing amitriptyline, topiramate, and placebo is currently underway (Childhood and Adolescent Migraine Prevention Study) [43].In 2 retrospective pediatric studies improvement with amitriptyline was reported in 84.2% and 89% of patients, respectively [44,45].We typically use a goal dose of 1 mg/kg/day, also with an 8- to 12-week titration. The most common side effects with amitriptyline are somnolence, dry mouth, and weight gain, but it is generally well tolerated in children and adolescents. There is also a risk of worsening depression and suicidal thoughts, so it is recommended to use caution if considering prescribing to a patient with underlying depression. It is typically administered in once daily dosing a few hours before bed to minimize morning drowsiness. There is also a concern for precipitation of arrhythmias, and while there are no guidelines recommending screening ECGs, this should be considered in patients with a family history of heart disease. Of note, many practitioners use nortriptyline in place of amitriptyline as it can be less sedating. It should be noted, however, that evidence for its efficacy is lacking. Additionally, it may carry a higher risk of arrhythmia [44].
Second-line Therapies
The second-line therapies typically considered are other antiepileptics, valproic acid, levetiracetam, and zonisamide, for which there is some evidence, although mostly in the form of open-label or retrospective studies [46–51]. Of note, despite the many promising retrospective and open-label studies for valproic acid [46–48], one randomized double-blind placebo control trial comparing various doses of extended release divalproex sodium with placebo in adolescent patients failed to show a statistically significant treatment difference between any dose and placebo [52].It is, however, frequently prescribed and anecdotally quite efficacious. Given concerns about potential for teratogenicity, and possible effects on ovarian function, as well as potential for weight gain and hair loss, it should be used with caution in adolescent females.
Antihypertensives (beta blockers and calcium channel blockers) have long been used for prevention of migraine in both the adult and pediatric population, but evidence for their use in the pediatric population is conflicting. An early double-blind crossover study of propranolol in patients 7 to 16 years old showed significant efficacy as compared with placebo [53].However, in 2 subsequent studies it failed to show efficacy as compared with placebo and self-hypnosis, respectively [54,55].Given the conflicting evidence and the potential for hypotension, depression, and exercise-induced asthma, use of propranolol has fallen out of favor by experts for use in pediatric and adolescent migraine prevention. Flunarizine, a nonselective calcium channel blocker, has demonstrated effectiveness in pediatric migraine prevention [56,57],and is actually approved in Europe for this indication, but it is not available in the United States.
Cyproheptadine is an antihistamine with antiserotonergic properties which is used frequently for migraine prevention in young children who are unable to swallow tablets, although evidence is limited to 1 retrospective study [45].However, given the propensity for weight gain with this medication, it is generally not recommended for use in adolescents.
In recent years, there has been a growing interest in nutraceuticals given their general tolerability and minimal side effects, as well as a comfort in using a more “natural” approach, particularly in pediatrics. Evidence is limited but some of the frequently used substances may be beneficial. Butterbur (petasites hybridus) has strong evidence in adults and is recommended by the AAN for migraine prevention in adults [58].There is one small pediatric RCT showing efficacy for butterbur as compared to music therapy [59],as well as one promising open-label trial [60]. Typical dosing ranges from 50–150 mg daily. However, butterbur contains pyrrolizidine alkaloids, which are hepatotoxic, and due to the concerns for inadequate monitoring of removal of these substances in the manufacturing of commercial butterbur, it has been generally avoided in the pediatric population. Coenzyme Q10 (CoQ10) is considered possibly effective for adult migraine prevention by the AAN [58].Pediatric evidence is limited to an open-label study showing improvement with supplementation of CoQ10 in deficient patients [61],and a subsequent double-blind placebo-controlled add-on study, which showed improvement in both the CoQ10 and placebo groups but faster improvement in the CoQ10 group [62].Typical pediatric dosing is 1–2 mg/kg/day. Magnesium is considered by the AAN to be a good option for migraine prevention in adults [58].One randomized controlled trial in children however had equivocal results [63],while one small prospective open-label study had positive results [64]. Magnesium supplementation may be more effective in patients who have low ionized magnesium levels, but this is difficult to measure reliably in the clinical setting. Doses of 9 mg/kg/day can be used with the most common side effects reported being gastrointestinal upset and diarrhea, generally dose dependent. Riboflavin is also considered by the AAN to be probably effective for prevention in adults [58],but again the evidence in children is limited to one positive retrospective study [65]and 2 equivocal randomized controlled trials [66,67],one of which had an unusually high placebo rate [66]. Appropriate dosing is also something of debate, as riboflavin is minimally absorbed and has a short half-life, so while studies were done using 200–400 mg daily, smaller more frequent dosing may be needed.
A potential approach to treatment with vitamins is to check for deficiencies, but currently only the study mentioned above in which CoQ10 levels were checked showed improvement with normalizing of low levels [61,62].Further research into this topic is needed to elucidate whether checking and repleting levels of specific vitamins would be beneficial in prevention of migraines in certain patients.
Healthy Habits
The importance of maintaining healthy lifestyle habits and modifying potentially detrimental ones should be stressed to patients and families, and counseling regarding these issues should be provided at every visit, as repetition is often key to patients understanding their importance. Skipping meals is a commonly reported migraine trigger [68,69].This is not an uncommon occurrence even in the most well-meaning of families; adolescents often report not feeling hungry in the morning, and in the rush to get to school will often skip breakfast. Many patients report not liking school lunches leading them to go most of the day without food. Adolescent girls may skip meals due to weight concerns. Patients need to be reminded that well-balanced meals throughout the day is imperative, and they often may need specific counseling on how to achieve this practically. Of note, unless a specific migraine trigger has been identified in a given patient, we do not generally recommend restricting any specific food lists.
Maintaining good sleep hygiene and a consistent sleep schedule is also often difficult for adolescent patients, with after-school activities, homework, and screen use (eg, television, electronic devices) often contributing to late bedtimes with then forced early morning waking for school. However, improvement in sleep hygiene has been shown to be effective in improving migraines in children and adolescents [70],and realistic plans for improvement in sleep should be discussed with patients.
Dehydration is also a common migraine trigger [8,9],so the importance of staying well hydrated should be stressed as well. Again, specific recommendations for how this can be achieved are often needed, especially given adolescents’ busy schedules. Additionally, many schools do not allow water bottles to be carried, and often a school note is needed so patients may be allowed to carry a water bottle at school and also be provided extra bathroom breaks as needed.
Also important is stressing maintenance of daily functioning throughout migraine treatment. By the time adolescents seek medical care, they may already be in a cycle of missing school due to headaches, and some may even be receiving home-schooling. The goals of staying in school and learning to function with headaches should be stressed, often with the help of coping skills, which will be discussed below, as it has been shown that functional disability generally improves before pain [71].
Behavioral Treatments
Psychological treatments are an important aspect of migraine management. Biobehavioral treatments such as relaxation training, biofeedback, and multimodal cognitive behavioral therapy (CBT) have been evaluated in randomized controlled trials and reviewed in meta-analyses in pediatric migraine populations and found to be efficacious [72–75].These treatments have been shown to reduce pain intensity and disability for children and adolescents with headaches and therapeutic gains appear to be maintained [72].
Relaxation training typically includes instruction in techniques such as diaphragmatic breathing, progressive muscle relaxation, and guided imagery. Relaxation training is most effective with children 7 years of age or older [76].Biofeedback is often used in conjunction with relaxation training to provide audio or visual feedback about normally unconscious physiological body responses associated with increased relaxation. Effective biofeedback parameters used with children and adolescents with migraine headache include electromyographic (EMG) activity and peripheral skin temperature [77].Biofeedback techniques can help children and adolescents become more aware of physical responses, better control these responses and generalize physical responses outside of therapy sessions to better cope with pain [76].CBT involves instruction in skills such as biofeedback-assisted relaxation training, activity pacing, distraction, and cognitive strategies for coping with pain. CBT was shown to be effective in a recent study comparing adolescent patients with chronic migraine receiving amitriptyline and CBT with patients receiving amitriptyline and standard headache education [78]. Patients who received CBT plus amitriptyline had greater reductions in days with headache and migraine-related disability compared with patients who received headache education plus amitriptyline [78].Hypnosis and acceptance and commitment therapy (ACT) are also psychological treatments used with children and adolescents with migraine headaches. ACT prioritizes the outcome of improved functioning above headache reduction and has broadly demonstrated efficacy for chronic pain [79]. Both treatments have shown promising benefit but there has been less evidence supporting the use of these therapies in pediatric headache than other well-established behavioral treatments.
The presence of comorbid psychiatric issues such as anxiety, depression, or ADHD can make the treatment of patients with migraine headaches more complex. A recent study found that approximately 30% of a sample of children and adolescents with chronic daily headache had a lifetime psychiatric diagnoses and having a lifetime psychiatric diagnosis was associated with poorer headache-related disability and quality of life [80].As a result, more intensive behavioral treatment for children and adolescents with a psychiatric comorbidity may be needed to focus on emotional and behavioral issues.
Pediatric psychologists can assess pain-related disability and coping difficulties and treat children and adolescents with migraine headaches. Children are often referred to a psychologist or other mental health professional if headaches are severe or impairing functioning. Unfortunately, access to this therapy is sometimes limited, but when available, should be offered to any migraine patients with significant disability requiring prevention, and specifically to patients with chronic migraine.
How should the patient be treated?
Acute Treatment
The patient reports only minimal response to ibuprofen. However, he is only taking 200 mg and does not take it until well after the headache has started. He should be instructed to take ibuprofen 600 mg (~10 mg/kg) as soon as the headache starts, along with 32 oz of a sports drink, and told that he should repeat this dose in 4 hours if his headache has not completely resolved with the first dose. As his headaches are occurring less than 3 times per week on average, he can do this with every headache. He should be given a note for school allowing him to receive the medication at school, and noting the importance of allowing him to get the medication as soon as he reports a headache. Given that some of his headaches occur on consecutive days, we suspect some may be continuations of a prior headache, so again the importance of obtaining complete pain relief from his acute medication should be stressed. If at his next follow-up visit he reports not always having complete relief with ibuprofen, we would consider trying naproxen instead, and/or adding a triptan, most likely sumatriptan 100 mg given its availability. He would need to understand that he may only use the triptan for 4 to 6 headache days/month. Depending on his response to sumatriptan, it could be used alone or in combination with his NSAID. He also may be instructed to use an NSAID alone for more mild headaches and his NSAID together with sumatriptan for more severe headaches.
Preventive Treatment
PedMIDAS score is a 25, indicating mild disability, however his migraines are frequent, occurring 1–3 times per week and clearly having a significant impact on his functioning. We would therefore recommend starting a prophylactic medication. He notes difficulty remembering to take medications as well as difficulty falling asleep, so amitriptyline would be a good choice for him. For his weight of 50.9 kg, we would start with at 12.5 mg at night and increase by 12.5 mg every 2 weeks to a dose of 50 mg (~1mg/kg/day). He and Mom should be counseled that it will likely take at least a few weeks on his goal dose before they start to see results. He should be counseled also to report to Mom immediately if he has any depressive or suicidal thoughts. Topiramate is another option, especially as it is FDA-approved for migraine prevention in children older than 12 years, although twice daily dosing may be a factor in maintaining compliance. Careful discussion with the patient and family in regards to the potential risks and benefits is important prior to starting any preventative medication.
Biobehavioral Management
The patient should be drinking 8–10 cups of non-caffeinated fluid per day and additional cups on days he plays soccer. He should be given a school note to this effect so he may carry a water bottle at school. He eats well, but the importance of not skipping breakfast should be stressed, and ideas for fitting this in should be given. The importance of a consistent bedtime routine should be stressed, with good sleep hygiene to assist with easier sleep onset. It should be made clear that he should not be using screens within an hour prior to bed. He already exercises frequently and should be commended for being active. Given his report of frequent headaches which are impairing school attendance, functioning at school, and participation in soccer activities, screening for potential co-morbid psychiatric issues and making a referral for further treatment with a pediatric pain psychologist focused on coping with chronic pain is appropriate.
Case Study 2
Initial Presentation and History
A 16-year-old right-handed girl who has been having headaches since she was 10 years old presents for evaluation. She reports unilateral mostly right-sided headaches, which are throbbing and associated with photophobia, phonophobia, osmophobia, nausea, and occasionally vomiting. She has no premonitory or aura symptoms. Her headaches typically occur once a week but she has noted that they tend to be more frequent around the time of her menses and that often these headaches do not fully respond to naproxen, which typically works to break her migraines during other times in the month. Upon further review, she believes these headaches typically start on the day or two prior to her menses, and that this happens almost every month. She often misses school due to these headaches. Her mother would like to know whether the patient should see a gynecologist to potentially be placed on birth control to control her headaches.
Physical Examination
On examination, weight was 61.5 kg (75.8 percentile), height 157.1 cm (18.4 percentile), BMI 24.91 (84 percentile), blood pressure 112/55 mm Hg, and heart rate 90 bpm. Her general physical and neurologic exam results were normal. She had tightness over the left trapezius muscles, otherwise the remainder of headache examination was unremarkable.
What is the probable diagnosis?
According to the ICHD-IIIβ, the patient meets criteria for episodic migraine without aura. Based on the history, she likely also has menstrually related migraine without aura [5].Officially, however, 3 months of prospective documentation is needed to make this diagnosis. Menstrually related migraine is included in the appendix of the ICHD-IIIβ, meaning there is ongoing debate about how it should be classified. According to the current appendix criteria, headaches should meet criteria for migraine without aura and also have documented and prospectively recorded evidence over at least 3 consecutive cycles with headaches confirmed on day 1+/– 2 (ie, 2 days prior to onset of menstruation or within the first 3 days of menstruation) in at least 2 out of 3 menstrual cycles, with migraines occurring during other times of the month as well [5]. This is distinguished from pure menstrual migraine without aura, in which migraines occur only during days 1+/–2 of menstruation but not during other times of the month [5].While we therefore cannot say that the patient meets ICHD IIIβ criteria for menstrually related migraine due to the lack of prospective documentation, her history suggests this.
Menstrually Related Migraine
An association of migraine with menses is well described in adults, occurring in up to 60% of adult female migraine patients [81].In adults, it has been observed that the migraines associated with the menstrual cycle tend to be more severe [82],associated with more nausea and vomiting [82],longer and less responsive to acute medications [83],and associated with more work-related disability [83]. Recently, analysis of data from the American Migraine Prevalence and Prevention (AMPP) Study found that women with pure menstrual migraine and menstrually associated migraine have on average higher MIDAS scores than those with non-menstrual migraine [81].
Only one study has explored menstrually related migraines in adolescents [84].It was a clinic-based study that found a similar prevalence—50% of adolescent patients who had reached menarch noted an association of migraine with their menses. Overall worse disability was not demonstrated in this study, but individual menstrual attacks were not compared with nonmenstrual attacks [84].No other studies have addressed this population, but it is clear that the pattern of menstrually related migraine exists in adolescents and it is important to recognize this pattern as these patients may require additional focused care in addition to standard migraine management.
What are treatment options?
In adults, options for more specific management of pure menstrual migraine and menstrually related migraine include intermittent prophylaxis with NSAIDs or triptans or use of hormonal contraceptives. There are no studies addressing any of these treatments in adolescents, so at this point management decisions must be based on evidence extrapolated from adult studies as well as attention to specific concerns in adolescent patients.
Generally, intermittent prophylaxis (using a medication a few days prior to and during the first few days of menses) with various medications has shown efficacy in adult studies. This approach may be more appropriate for patients with pure menstrual migraine, as there is less likelihood of precipitating medication overuse in this population. It can be considered in patients with menstrually related migraine as well if typical daily preventives have not been effective. One small open-label trial using naproxen 550 mg daily prior to and during menses (at differing schedules depending on the month) showed slightly decreased frequency and severity of headaches during that time [85].However, triptans are what are most commonly used.
Triptans
Frovatriptan is the longest-acting triptan and the one used most commonly for intermittent prophylaxis. One double-blind randomized controlled crossover study showed improvements in headache frequency, severity, and duration using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses [86].Frovatriptan 2.5 mg daily showed some efficacy as well but was not as effective as 2.5 mg BID [86].One prospective randomized placebo-controlled trial demonstrated more headache-free cycles using frovatriptan 2.5 mg BID for 6 days total starting 2 days prior to menses, as compared to placebo [87].
Naratriptan, also a longer-acting triptan, was studied in 3 prospective double-blind trials at doses of 1 mg BID used for 5 days total starting 2 days prior to menses, and all 3 showed a decrease in headache frequency as compared to placebo [88,89].Of note no efficacy was shown using 2.5 mg daily [88].
Less commonly used for intermittent prophylaxis are the shorter-acting triptans, but they have shown some efficacy as well. In one prospective study using oral sumatriptan 25 mg TID for 5 days each cycle starting 2 to 3 days before onset of menses, there seemed to be at least some improvement with sumatriptan in most cycles treated [90].Zolmitriptan was studied in one prospective double-blind placebo-controlled study using 2.5 mg BID or 2.5 mg TID for 7 days total with each cycle and was found to be associated with a significant reduction in headache frequency as compared with placebo [91].There has been one prospective trial using eletriptan 20 mg TID for 6 days total starting 2 days prior to menses, which also showed improvement in headache activity in 55% of patients [92].
Hormonal Contraceptives
In general for adult patients, hormonal contraceptives are not considered first-line treatment for menstrually related migraine. However, in patients who do not respond to other modes of treatment, or who plan to use hormonal contraception for contraceptive purposes, published expert opinions have recommended considering extended or continuous hormonal regimens [93,94].
Estrogen withdrawal during the luteal phase of the monthly cycle has long been speculated to be of importance in the pathophysiology of menstrually related migraine [93]and the relationship between hormonal contraceptives and migraine is complicated. Headache is a commonly reported side effect of combined hormonal contraceptives [95]; however, this effect seems to occur mostly during the hormone-free week [96]. It has been shown that headache occurs less frequently in women using an extended cycle regimen (84 or 168 days) as compared to those using a traditional monthly cyclic regimen [97,98].Studies addressing the use of combined hormonal contraceptives for women specifically with menstrually related migraine are limited. One small prospective randomized study showed improvement in menstrually related migraine in patients treated with a low dose (20 mcg ethinyl E[2]) oral hormonal contraceptive in both a 21/7 cycle and a 24/4 cycle, with more improvement in the group using a 24/4 cycle [99].Another showed improvement in menstrually related migraine in all study patients treated with a low-dose hormonal contraceptive (20 mcg ethinyl estradiol) on a 21/7 regimen, but with additional 0.9 mg conjugated equine estrogen during the placebo week [100].There has been one randomized placebo-controlled double-blind trial in patients with menstrually related migraine using an extended 168 hormonal regimen along with frovatriptan vs. placebo for 5 days during the hormone-free interval. Overall daily headache scores were decreased from pre-study cycles, and the increase in headaches during the hormone-free interval was lower in the frovatriptan group [101].A recent study showed that contraceptive-induced amenorrhea can be beneficial for decreasing migraine frequency in patients with menstrual migraine [102].There have been no studies addressing the use of hormonal contraceptives for migraine management in adolescents.
In adolescents, the decision regarding use of hormonal contraceptives is more complicated. There is less of a chance that adolescent patients, especially younger ones, would be planning to use hormones for contraception so their use may be solely for the purpose of migraine management. However, hormonal contraceptives are commonly used in adolescents for management of other menstrual-related disorders, such as menorrhagia, dysmenorrhea, and endometriosis, and extended cycle and continuous regimens have become more popular with adolescent providers in general [103].The general concern with using hormonal contraceptives in adolescents is that they have potential for longer-term use than adult patients, and the effects of using hormonal contraceptives long term are unknown. The major concerns are for potential interference with expected increase in bone mineral density in adolescents, effects on fertility, and risk for cancer and cardiovascular disease [103].Additionally, specifically in migraine patients, is the concern for increased stroke risk. The increased risk for ischemic stroke in patients with migraine, although more specifically with migraine with aura, is well known [104–106]and migraine has recently been shown to be a risk factor for ischemic stroke in adolescents as well [107]. In adults, the use of hormonal contraceptives in patients with migraine with aura is known to increase the risk of ischemic stroke [106],and this has not been studied in adolescents. Given the various unknowns in the use of hormonal contraceptives in patients with menstrually related migraines, we would not recommend this as first-line treatment. However, similarly to adults, in patients who do not respond to other methods of migraine management, or who seek to use hormonal contraceptives for contraception or for other menstrually related disorders, extended or continuous cycle hormonal contraceptives may be a reasonable option, at the lowest possible estrogen dose. However, migraine with aura should be screened for, and its presence should prompt reconsideration of combined hormonal contraceptive use.
How should this patient be managed?
The patient is having frequent and disabling migraines, so starting a preventive medication would be appropriate. She has migraines throughout the month in addition to during her menses, so a daily prophylactic would be more appropriate than intermittent prophylaxis surrounding her menstrual cycle only. At this point, our recommendation would be to start a daily preventive with either amitriptyline or topiramate. Given that naproxen is not breaking some of her migraines, she should be given a prescription for a triptan. Sumatriptan 100 mg would be an appropriate first choice, and she can be instructed to use it along with her naproxen at the onset of her menstrual migraines. She can use it for other migraines as well but she should be instructed not to use it more than 4 to 6 times per month. She should keep a diary for the next 3 months noting most importantly headache days as well as days of menstruation, so that a more definitive pattern can be confirmed and an official diagnosis based on ICHD-IIIβ criteria can be made. If her migraines do not improve with daily preventives, at that point discussion regarding potential for intermittent prophylaxis or trial of extended cycle hormonal contraception may be considered, although with caution and discussion of risks and benefits.
Corresponding author: Hope O’Brien, MD, Cincinnati Children’s Hospital Medical Center, 3333 Burnet Ave., MLC 2015 Cincinnati, OH 45229.
Financial disclosures: None.
1. Tepper SJ, Dahlöf CG, Dowson A, et al. Prevalence and diagnosis of migraine in patients consulting their physician with a complaint of headache: data from the Landmark Study. Headache 2004;44:856–64.
2. Lewis DW et al. Practice parameter: evaluation of children and adolescents with recurrent headaches: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2002;59:490–8.
3. Martens D, Oster I, Gottschlling S, et al. Cerebral MRI and EEG studies in the initial management of pediatric headaches. Swiss Med Wkly 2012;142:w13625.
4. Linder SL. Understanding the comprehensive pediatric headache examination. Pediatr Ann 2005;34:442–6.
5. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia 2013;33:629–808.
6. Senbil N, Gürer YK, Uner C, et al. Sinusitis in children and adolescents with chronic or recurrent headache: a case-control study. J Headache Pain 2008;9:33–6.
7. Gelfand AA, Reider AC, Goadsby PJ. Cranial autonomic symptoms in pediatric migraine are the rule, not the exception. Neurology 2013;81:431–6.
8. Blau JN. Water deprivation: a new migraine precipitant. Headache 2005;45:757–9.
9. Martins IP, Gouveia RG. More on water and migraine. Cephalalgia 2007;27:372–4.
10. Hämäläinen ML, Hoppu K, Valkeila E, et al. Ibuprofen or acetaminophen for the acute treatment of migraine in children: a double-blind, randomized, placebo-controlled, crossover study. Neurology 1997;48:103–7.
11. Lewis DW, Kellsein D, Sahl G, et al. Children’s ibuprofen suspension for the acute treatment of pediatric migraine. Headache 2002;42:780–6.
12. Lipton RB, Golstein J, Baggish JS, et al. Aspirin is efficacious for the treatment of acute migraine. Headache 2005;45:283–92.
13. Linder SL, Mathew NT, Cady RK, et al. Efficacy and tolerability of almotriptan in adolescents: a randomized, double-blind, placebo-controlled trial. Headache 2008;48:1326–36.
14. Ahonen K, Hämäläinen ML, Eerola M, et al. A randomized trial of rizatriptan in migraine attacks in children. Neurology 2006;67:1135–40.
15. Ho TW, Pearlman E, Lewis D, et al. Efficacy and tolerability of rizatriptan in pediatric migraineurs: results from a randomized, double-blind, placebo-controlled trial using a novel adaptive enrichment design. Cephalalgia 2012;32:750–65.
16. Winner P, Lewis D, Visser WH, et al. Rizatriptan 5 mg for the acute treatment of migraine in adolescents: a randomized, double-blind, placebo-controlled study. Headache 2002;42:49–55.
17. Hewitt DJ, Pearlman E, Hämäläinen M, et al. Long-term open-label safety study of rizatriptan acute treatment in pediatric migraineurs. Headache 2013;53:104–17.
18. Derosier FJ, Lewis D, Hershey AD, et al. Randomized trial of sumatriptan and naproxen sodium combination in adolescent migraine. Pediatrics 2012;129:e1411–20.
19. McDonald SA, Hershey AD, Pearlman E, et al. Long-term evaluation of sumatriptan and naproxen sodium for the acute treatment of migraine in adolescents. Headache 2011;51:1374-87.
20. Lewis DW, Winner P, Hershey AD, et al. Efficacy of zolmitriptan nasal spray in adolescent migraine. Pediatrics 2007;
120:390–6.
21. Linder SL, Dowson AJ. Zolmitriptan provides effective migraine relief in adolescents. Int J Clin Pract 2000;54:466–9.
22. Winner P, Rothner AD, Saper J, et al. A randomized, double-blind, placebo-controlled study of sumatriptan nasal spray in the treatment of acute migraine in adolescents. Pediatrics 2000;106:989–97.
23. Rothner AD, Winner P, Nett R, et al. One-year tolerability and efficacy of sumatriptan nasal spray in adolescents with migraine: results of a multicenter, open-label study. Clin Ther 2000;22:1533–46.
24. Ahonen K, Hämäläinen ML, Rantala H, et al. Nasal sumatriptan is effective in treatment of migraine attacks in children: A randomized trial. Neurology 2004;62:883–7.
25. Natarajan S, Jabbour JT, Webster CJ, et al. Long-term tolerability of sumatriptan nasal spray in adolescent patients with migraine. Headache 2004;44:969–77.
26. Winner P, Rothner AD, Wooten JD, et al. Sumatriptan nasal spray in adolescent migraineurs: a randomized, double-blind, placebo-controlled, acute study. Headache 2006;46:212–22.
27. Hämäläinen ML, Hoppu K, Santavuori P. Sumatriptan for migraine attacks in children: a randomized placebo-controlled study. Do children with migraine respond to oral sumatriptan differently from adults? Neurology 1997;48:1100–3.
28. Fujita M, Sato K, Nishioka H, et al. Oral sumatriptan for migraine in children and adolescents: a randomized, multicenter, placebo-controlled, parallel group study. Cephalalgia 2014;34:365–75.
29. Winner P, Linder SL, Lipton RB, et al. Eletriptan for the acute treatment of migraine in adolescents: results of a double-blind, placebo-controlled trial. Headache 2007;47:511–8.
30. Rothner A. Efficacy and safety of naratriptan tablets in adolescent migraine [abstract]. J Neurol Sci 1997;150:S106.
31. Elkind AH, Wade A, Ishkanian G. Pharmacokinetics of frovatriptan in adolescent migraineurs. J Clin Pharmacol 2004;44:1158–65.
32. Manack AN, Buse DC, Lipton RB. Chronic migraine: epidemiology and disease burden. Curr Pain Headache Rep 2011;15:70–8.
33. Winner P, Pearlman EM, Linder SL, et al. Topiramate for migraine prevention in children: a randomized, double-blind, placebo-controlled trial. Headache 2005;45:1304–12.
34. Lakshmi CV, Singhi P, Malhi P, et al. Topiramate in the prophylaxis of pediatric migraine: a double-blind placebo-controlled trial. J Child Neurol 2007;22:829–35.
35. Lewis D, Winner P, Saper J, et al.Randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of topiramate for migraine prevention in pediatric subjects 12 to 17 years of age. Pediatrics 2009;123:924–34.
36. Hershey AD, Powers SW, Vockell AL, et al. Effectiveness of topiramate in the prevention of childhood headaches. Headache 2002;42:810–8.
37. Campistol J, Campos J, Casas C, et al. Topiramate in the prophylactic treatment of migraine in children. J Child Neurol 2005;20:251–3.
38. Unalp A, Uran N, Oztürk A. Comparison of the effectiveness of topiramate and sodium valproate in pediatric migraine. J Child Neurol 2008;23:1377–81.
39. Cruz MJ, Valencia I, Legido A, et al. Efficacy and tolerability of topiramate in pediatric migraine. Pediatr Neurol 2009;41:167–70.
40. Kim H, Byun SH, Kim JS, et al. Comparison of flunarizine and topiramate for the prophylaxis of pediatric migraines. Eur J Paediatr Neurol 2013;17:45–9.
41. Fallah R, Divanizadeh MS, Karimi M, et al. Topiramate and propranolol for prophylaxis of migraine. Indian J Pediatr 2013;80:920–4.
42. Tonekaboni SH, Ghazavi A, Fayyazi A, et al. Prophylaxis of childhood migraine: topiramate versus propranolol. Iran J Child Neurol 2013;7:9–14.
43. Hershey AD, Powers SW, Coffey CS, et al. Childhood and Adolescent Migraine Prevention (CHAMP) study: a double blinded, placebo controlled, comparative effectiveness study of amitriptyline, topiramate, and placebo in prevention of childhood and adolescent migraine. Headache 2013;53:799–816.
44. Hershey AD, Powers SW, Bentti AL, et al. Effectiveness of amitriptyline in the prophylactic management of childhood headaches. Headache 2000;40:539–49.
45. Lewis DW, Diamond S, Scott D, et al. Prophylactic treatment of pediatric migraine. Headache 2004;44:230–7.
46. Caruso JM, Brown WD, Exil G, et al. The efficacy of divalproex sodium in the prophylactic treatment of children with migraine. Headache 2000 Sep;40:672–6.
47. Pakalnis A, Greenberg G, Drake ME Jr, et al. Pediatric migraine prophylaxis with divalproex. J Child Neurol 2001;16:731–4.
48. Serdaroglu G, Erhan E, Tekgul H, et al. Sodium valproate prophylaxis in childhood migraine. Headache 2002;42:819–22.
49. Miller GS. Efficacy and safety of levetiracetem in pediatric migraine. Headache 2004;44:238–43.
50. Pakalnis A, Kring D, Meier L. Levetiracetam prophylaxis in pediatric migraine--an open label study. Headache 2007;43:427–30.
51. Pakalnis A, Kring D. Zonisamide prophylaxis in refractory pediatric headache. Headache 2006;46:804–7.
52. Apostol G, Cady RK, Laforet GA, et al. Divalproex extended-release in adolescent migraine prophylaxis: results of a randomized, double-blind, placebo-controlled study. Headache 2008;48:1012–25.
53. Ludvigsson J. Propranolol used in prophylaxis of migraine in children. Acta Neurol 1974;50:109–15.
54. Forsythe WI, Gillies D, Sills MA. Propanolol (‘Inderal’) in the treatment of childhood migraine. Dev Med Child Neurol 1984;26:737–41.
55. Olness K, MacDonald JT, Uden DL. Comparison of self-hypnosis and propranolol in the treatment of juvenile classic migraine. Pediatrics 1987;79:593–7.
56. Sorge F, DeSimone R, Marano E, et al. Flunarizine in prophylaxis of childhood migraine. A double-blind, placebo-controlled crossover study. Cephalalgia 1988;8:1–6.
57. Guidetti V, Moscato D, Ottaviano S, et al. Flunarizine and migraine in childhood: an evaluation of endocrine function. Cephalalgia 1987;7:263–6.
58. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and complementary treatments for episodic migraine treatment in adults: Report of the quality standards subcommittee of the American Academy of Neurology and American Headache Society. Neurology 2012; 78:1346–53.
59. Oelkers-Ax R, Leins A, Parzer P, et al. Butterbur root extract and music therapy in the prevention of childhood migraine: an explorative study. Eur J Pain 2008;12:301–13.
60. Pothmann R, Danesch U. Migraine prevention in children and adolescents: results of an open study with a special butterbur root extract. Headache 2005;45:196–203.
61. Hershey AD, Powers SW, Vockell AL, et al. Coenzyme Q10 deficiency and response to supplementation in pediatric and adolescent migraine. Headache 2007;47:73–80.
62. Slater SK, Nelson TD, Kabbouche MA, et al. A randomized, double-blind, crossover, add-on study of coenzyme Q10 in the prevention of pediatric and adolescent migraine. Cephalalgia 2011; 31: 897–905.
63. Wang F, Van Den Eeden SK, Ackerson LM, et al. Oral magnesium oxide prophylaxis of frequent migrainous headache in children: a randomized, double-blind, placebo-controlled trial. Headache 2003;43:601–10.
64. Castelli S, Meossi C, Domenici R, et al. [Magnesium in the prophylaxis of primary headache and other periodic disorders in children]. Pediatr Med Chir 1993;15:481–8. Italian.
65. Condò M, Posar A, Arbizzani A, et al. Riboflavin prophylaxis in pediatric and adolescent migraine. J Headache Pain 2009;10:361–5.
66. MacLennan SC, Wade FM, Forrest KM, et al. High-dose riboflavin for migraine prophylaxis in children: a double-blind, randomized, placebo-controlled trial. J Child Neurol 2008;23:1300–4.
67. Bruijn J, Duivenvoorden H, Passchier J, et al. Medium-dose riboflavin as a prophylactic agent in children with migraine: a preliminary placebo-controlled, randomised, double-blind, cross-over trial. Cephalalgia 2010;30:1426–34.
68. Robbins L. Precipitating factors in migraine: a retrospective review of 494 patients. Headache 1994;34:214–6.
69. Leviton A, Slack WV, Masek B, et al. A computerized behavioral assessment for children with headaches. Headache 1984;24:182–5.
70. Bruni O, Galli F, Guidetti V. Sleep hygiene and migraine in children and adolescents. Cephalalgia 1999;19 Suppl 25:57–9.
71. Lynch-Jordan AM, Sil S, Peugh J, et al. Differential changes in functional disability and pain intensity over the course of psychological treatment for children with chronic pain. Pain 2014;155:1955–61.
72. Eccleston C, Palermo TM, Williams AC, et al. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database Syst Rev 2014;5:CD003968.
73. Huguet A, McGrath PJ, Stinson J, et al. Efficacy of psychological treatment for headaches: an overview of systematic reviews and analysis of potential modifiers of treatment efficacy. Clin J Pain 2014;30:353–69.
74. Kropp P, Meyer B, Landgraf M, et al. Headache in children: update on biobehavioral treatments. Neuropediatrics 2013;44:20–4.
75. Kröner-Herwig B. Psychological treatments for pediatric headache. Expert Rev Neurother 2011;11:403–10.
76. Powers SW, Andrasik F. Biobehavioral treatment, disability, and psychological effects of pediatric headache. Pediatr Ann 2005;34:461–5.
77. Hermann C, Blanchard EB. Biofeedback in the treatment of headache and other childhood pain. Appl Psychophysiol Biofeedback 2002;27:143–62.
78. Powers SW, Kashikar-Zuck SM, Hershey AD, et al. Cognitive behavioral therapy plus amitriptyline for chronic migraine in children and adolescents: a randomized clinical trial. JAMA 2013;310:2622–30.
79. Smitherman TA, Wells RE, Ford SG. Emerging behavioral treatments for migraine. Curr Pain Headache Rep 2015;19:13.
80. Slater SK, O’Brien HL, Hershey AD, et al. Psychiatric comorbidity in pediatric chronic daily headache. Cephalalgia 2012;32: 1116–22.
81. Pavlovic JM, Stewart WF, Bruce CA, et al. Burden of migraine related to menses: results from the AMPP study. J Headache Pain 2015;16:24.
82. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 2004;63:351–3.
83. Granella F, Sances G, Allais G. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia 2004;24:707–16.
84. Crawford MJ, Lehman L, Slater S, et al. Menstrual migraine in adolescents. Headache 2009;49:341–7.
85. Allais G, Bussone G, De Lorenzo C, et al. Naproxen sodium in short-term prophylaxis of pure menstrual migraine: pathophysiological and clinical considerations. Neurol Sci 2007;28(Suppl 2):S225–8.
86. Silberstein S, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology 2004;63:261–9.
87. Brandes JL, Poole A, Kallela M, et al. Short-term frovatriptan for the prevention of difficult-to-treat menstrual migraine attacks. Cephalalgia 2009;29:1133–48.
88. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache 2001;41:248–56.
89. Mannix LK, Savani N, Landy S, et al. Efficacy and tolerability of naratriptan for short-term prevention of menstrually related migraine: data from two randomized, double-blind, placebo-controlled studies. Headache 2007;47:1037–49.
90. Newman LC, Lipton RB, Lay CL, et al. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology 1998;51:307–9.
91. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs 2008;22:877–86.
92. Marcus DA, Bernstein CD, Sullivan EA, et al. Perimenstrual eletriptan prevents menstrual migraine: an open-label study. Headache 2010;50:551–6.
93. Silberstein S, Patel S. Menstrual migraine: an updated review on hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother 2014;15:2063–70.
94. MacGregor EA. Migraine management during menstruation and menopause. Continuum (Minneap Minn) 2015;21(4 Headache):990–1003.
95. Aegidius K, Zwart JA, Hagen K, et al. Oral contraceptives and increased headache prevalence: The Head-HUNT Study. Neurology 2006;66:349–53.
96. Sulak PJ, Scow RD, Preece C, et al. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol 2000;95:261–6.
97. LaGuardia KD, Fisher AC, Bainbridge JD, et al. Suppression of estrogen-withdrawal headache with extended transdermal contraception. Fertil Steril 2005;83:1875–7.
98. Sulak P,Willis S, Kuehl T, Coffee A, Clark J. Headaches and oral contraceptives: Impact of eliminating the standard 7-day placebo interval. Headache 2007;47:27–37.
99. De Leo V, Scolaro V, Musacchio MC, et al. Combined oral contraceptives in women with menstrual migraine without aura. Fertil Steril 2011;96:917–20.
100. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J 2004;97:819–22.
101. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health (Larchmt) 2014;23:310–7.
102. Vetvik KG, MacGregor EA, Lundqvist C, et al. Contraceptive-induced amenorrhea leads to reduced migraine frequency in women with menstrual migraine without aura. J Headache Pain 2014;15:30.
103. Sucato GS, Gerschultz KL. Extended cycle hormonal contraception in adolescents. Curr Opin Obstet Gynecol 2005;17:461–5.
104. Spector JT, Kahn SR, Jones MR, et al. Migraine headache and ischemic stroke risk: an updated meta-analysis. Am J Med 2010;123:612–24.
105. Bigal ME, Kurth T, Santanello N, et al. Migraine and cardiovascular disease: a population-based study. Neurology 2010;74:628–35.
106. Schurks M, Rist PM, Bigal ME, et al. Migraine and cardiovascular disease: systematic review and meta-analysis. BMJ 2009;339:b3914.
107. Gelfand AA, Fullerton HJ, Jacobson A, et al. Is migraine a risk factor for pediatric stroke? Cephalalgia 2015;35:1252–60.
1. Tepper SJ, Dahlöf CG, Dowson A, et al. Prevalence and diagnosis of migraine in patients consulting their physician with a complaint of headache: data from the Landmark Study. Headache 2004;44:856–64.
2. Lewis DW et al. Practice parameter: evaluation of children and adolescents with recurrent headaches: report of the Quality Standards Subcommittee of the American Academy of Neurology and the Practice Committee of the Child Neurology Society. Neurology 2002;59:490–8.
3. Martens D, Oster I, Gottschlling S, et al. Cerebral MRI and EEG studies in the initial management of pediatric headaches. Swiss Med Wkly 2012;142:w13625.
4. Linder SL. Understanding the comprehensive pediatric headache examination. Pediatr Ann 2005;34:442–6.
5. Headache Classification Committee of the International Headache Society (IHS). The international classification of headache disorders, 3rd edition (beta version). Cephalalgia 2013;33:629–808.
6. Senbil N, Gürer YK, Uner C, et al. Sinusitis in children and adolescents with chronic or recurrent headache: a case-control study. J Headache Pain 2008;9:33–6.
7. Gelfand AA, Reider AC, Goadsby PJ. Cranial autonomic symptoms in pediatric migraine are the rule, not the exception. Neurology 2013;81:431–6.
8. Blau JN. Water deprivation: a new migraine precipitant. Headache 2005;45:757–9.
9. Martins IP, Gouveia RG. More on water and migraine. Cephalalgia 2007;27:372–4.
10. Hämäläinen ML, Hoppu K, Valkeila E, et al. Ibuprofen or acetaminophen for the acute treatment of migraine in children: a double-blind, randomized, placebo-controlled, crossover study. Neurology 1997;48:103–7.
11. Lewis DW, Kellsein D, Sahl G, et al. Children’s ibuprofen suspension for the acute treatment of pediatric migraine. Headache 2002;42:780–6.
12. Lipton RB, Golstein J, Baggish JS, et al. Aspirin is efficacious for the treatment of acute migraine. Headache 2005;45:283–92.
13. Linder SL, Mathew NT, Cady RK, et al. Efficacy and tolerability of almotriptan in adolescents: a randomized, double-blind, placebo-controlled trial. Headache 2008;48:1326–36.
14. Ahonen K, Hämäläinen ML, Eerola M, et al. A randomized trial of rizatriptan in migraine attacks in children. Neurology 2006;67:1135–40.
15. Ho TW, Pearlman E, Lewis D, et al. Efficacy and tolerability of rizatriptan in pediatric migraineurs: results from a randomized, double-blind, placebo-controlled trial using a novel adaptive enrichment design. Cephalalgia 2012;32:750–65.
16. Winner P, Lewis D, Visser WH, et al. Rizatriptan 5 mg for the acute treatment of migraine in adolescents: a randomized, double-blind, placebo-controlled study. Headache 2002;42:49–55.
17. Hewitt DJ, Pearlman E, Hämäläinen M, et al. Long-term open-label safety study of rizatriptan acute treatment in pediatric migraineurs. Headache 2013;53:104–17.
18. Derosier FJ, Lewis D, Hershey AD, et al. Randomized trial of sumatriptan and naproxen sodium combination in adolescent migraine. Pediatrics 2012;129:e1411–20.
19. McDonald SA, Hershey AD, Pearlman E, et al. Long-term evaluation of sumatriptan and naproxen sodium for the acute treatment of migraine in adolescents. Headache 2011;51:1374-87.
20. Lewis DW, Winner P, Hershey AD, et al. Efficacy of zolmitriptan nasal spray in adolescent migraine. Pediatrics 2007;
120:390–6.
21. Linder SL, Dowson AJ. Zolmitriptan provides effective migraine relief in adolescents. Int J Clin Pract 2000;54:466–9.
22. Winner P, Rothner AD, Saper J, et al. A randomized, double-blind, placebo-controlled study of sumatriptan nasal spray in the treatment of acute migraine in adolescents. Pediatrics 2000;106:989–97.
23. Rothner AD, Winner P, Nett R, et al. One-year tolerability and efficacy of sumatriptan nasal spray in adolescents with migraine: results of a multicenter, open-label study. Clin Ther 2000;22:1533–46.
24. Ahonen K, Hämäläinen ML, Rantala H, et al. Nasal sumatriptan is effective in treatment of migraine attacks in children: A randomized trial. Neurology 2004;62:883–7.
25. Natarajan S, Jabbour JT, Webster CJ, et al. Long-term tolerability of sumatriptan nasal spray in adolescent patients with migraine. Headache 2004;44:969–77.
26. Winner P, Rothner AD, Wooten JD, et al. Sumatriptan nasal spray in adolescent migraineurs: a randomized, double-blind, placebo-controlled, acute study. Headache 2006;46:212–22.
27. Hämäläinen ML, Hoppu K, Santavuori P. Sumatriptan for migraine attacks in children: a randomized placebo-controlled study. Do children with migraine respond to oral sumatriptan differently from adults? Neurology 1997;48:1100–3.
28. Fujita M, Sato K, Nishioka H, et al. Oral sumatriptan for migraine in children and adolescents: a randomized, multicenter, placebo-controlled, parallel group study. Cephalalgia 2014;34:365–75.
29. Winner P, Linder SL, Lipton RB, et al. Eletriptan for the acute treatment of migraine in adolescents: results of a double-blind, placebo-controlled trial. Headache 2007;47:511–8.
30. Rothner A. Efficacy and safety of naratriptan tablets in adolescent migraine [abstract]. J Neurol Sci 1997;150:S106.
31. Elkind AH, Wade A, Ishkanian G. Pharmacokinetics of frovatriptan in adolescent migraineurs. J Clin Pharmacol 2004;44:1158–65.
32. Manack AN, Buse DC, Lipton RB. Chronic migraine: epidemiology and disease burden. Curr Pain Headache Rep 2011;15:70–8.
33. Winner P, Pearlman EM, Linder SL, et al. Topiramate for migraine prevention in children: a randomized, double-blind, placebo-controlled trial. Headache 2005;45:1304–12.
34. Lakshmi CV, Singhi P, Malhi P, et al. Topiramate in the prophylaxis of pediatric migraine: a double-blind placebo-controlled trial. J Child Neurol 2007;22:829–35.
35. Lewis D, Winner P, Saper J, et al.Randomized, double-blind, placebo-controlled study to evaluate the efficacy and safety of topiramate for migraine prevention in pediatric subjects 12 to 17 years of age. Pediatrics 2009;123:924–34.
36. Hershey AD, Powers SW, Vockell AL, et al. Effectiveness of topiramate in the prevention of childhood headaches. Headache 2002;42:810–8.
37. Campistol J, Campos J, Casas C, et al. Topiramate in the prophylactic treatment of migraine in children. J Child Neurol 2005;20:251–3.
38. Unalp A, Uran N, Oztürk A. Comparison of the effectiveness of topiramate and sodium valproate in pediatric migraine. J Child Neurol 2008;23:1377–81.
39. Cruz MJ, Valencia I, Legido A, et al. Efficacy and tolerability of topiramate in pediatric migraine. Pediatr Neurol 2009;41:167–70.
40. Kim H, Byun SH, Kim JS, et al. Comparison of flunarizine and topiramate for the prophylaxis of pediatric migraines. Eur J Paediatr Neurol 2013;17:45–9.
41. Fallah R, Divanizadeh MS, Karimi M, et al. Topiramate and propranolol for prophylaxis of migraine. Indian J Pediatr 2013;80:920–4.
42. Tonekaboni SH, Ghazavi A, Fayyazi A, et al. Prophylaxis of childhood migraine: topiramate versus propranolol. Iran J Child Neurol 2013;7:9–14.
43. Hershey AD, Powers SW, Coffey CS, et al. Childhood and Adolescent Migraine Prevention (CHAMP) study: a double blinded, placebo controlled, comparative effectiveness study of amitriptyline, topiramate, and placebo in prevention of childhood and adolescent migraine. Headache 2013;53:799–816.
44. Hershey AD, Powers SW, Bentti AL, et al. Effectiveness of amitriptyline in the prophylactic management of childhood headaches. Headache 2000;40:539–49.
45. Lewis DW, Diamond S, Scott D, et al. Prophylactic treatment of pediatric migraine. Headache 2004;44:230–7.
46. Caruso JM, Brown WD, Exil G, et al. The efficacy of divalproex sodium in the prophylactic treatment of children with migraine. Headache 2000 Sep;40:672–6.
47. Pakalnis A, Greenberg G, Drake ME Jr, et al. Pediatric migraine prophylaxis with divalproex. J Child Neurol 2001;16:731–4.
48. Serdaroglu G, Erhan E, Tekgul H, et al. Sodium valproate prophylaxis in childhood migraine. Headache 2002;42:819–22.
49. Miller GS. Efficacy and safety of levetiracetem in pediatric migraine. Headache 2004;44:238–43.
50. Pakalnis A, Kring D, Meier L. Levetiracetam prophylaxis in pediatric migraine--an open label study. Headache 2007;43:427–30.
51. Pakalnis A, Kring D. Zonisamide prophylaxis in refractory pediatric headache. Headache 2006;46:804–7.
52. Apostol G, Cady RK, Laforet GA, et al. Divalproex extended-release in adolescent migraine prophylaxis: results of a randomized, double-blind, placebo-controlled study. Headache 2008;48:1012–25.
53. Ludvigsson J. Propranolol used in prophylaxis of migraine in children. Acta Neurol 1974;50:109–15.
54. Forsythe WI, Gillies D, Sills MA. Propanolol (‘Inderal’) in the treatment of childhood migraine. Dev Med Child Neurol 1984;26:737–41.
55. Olness K, MacDonald JT, Uden DL. Comparison of self-hypnosis and propranolol in the treatment of juvenile classic migraine. Pediatrics 1987;79:593–7.
56. Sorge F, DeSimone R, Marano E, et al. Flunarizine in prophylaxis of childhood migraine. A double-blind, placebo-controlled crossover study. Cephalalgia 1988;8:1–6.
57. Guidetti V, Moscato D, Ottaviano S, et al. Flunarizine and migraine in childhood: an evaluation of endocrine function. Cephalalgia 1987;7:263–6.
58. Holland S, Silberstein SD, Freitag F, et al. Evidence-based guideline update: NSAIDs and complementary treatments for episodic migraine treatment in adults: Report of the quality standards subcommittee of the American Academy of Neurology and American Headache Society. Neurology 2012; 78:1346–53.
59. Oelkers-Ax R, Leins A, Parzer P, et al. Butterbur root extract and music therapy in the prevention of childhood migraine: an explorative study. Eur J Pain 2008;12:301–13.
60. Pothmann R, Danesch U. Migraine prevention in children and adolescents: results of an open study with a special butterbur root extract. Headache 2005;45:196–203.
61. Hershey AD, Powers SW, Vockell AL, et al. Coenzyme Q10 deficiency and response to supplementation in pediatric and adolescent migraine. Headache 2007;47:73–80.
62. Slater SK, Nelson TD, Kabbouche MA, et al. A randomized, double-blind, crossover, add-on study of coenzyme Q10 in the prevention of pediatric and adolescent migraine. Cephalalgia 2011; 31: 897–905.
63. Wang F, Van Den Eeden SK, Ackerson LM, et al. Oral magnesium oxide prophylaxis of frequent migrainous headache in children: a randomized, double-blind, placebo-controlled trial. Headache 2003;43:601–10.
64. Castelli S, Meossi C, Domenici R, et al. [Magnesium in the prophylaxis of primary headache and other periodic disorders in children]. Pediatr Med Chir 1993;15:481–8. Italian.
65. Condò M, Posar A, Arbizzani A, et al. Riboflavin prophylaxis in pediatric and adolescent migraine. J Headache Pain 2009;10:361–5.
66. MacLennan SC, Wade FM, Forrest KM, et al. High-dose riboflavin for migraine prophylaxis in children: a double-blind, randomized, placebo-controlled trial. J Child Neurol 2008;23:1300–4.
67. Bruijn J, Duivenvoorden H, Passchier J, et al. Medium-dose riboflavin as a prophylactic agent in children with migraine: a preliminary placebo-controlled, randomised, double-blind, cross-over trial. Cephalalgia 2010;30:1426–34.
68. Robbins L. Precipitating factors in migraine: a retrospective review of 494 patients. Headache 1994;34:214–6.
69. Leviton A, Slack WV, Masek B, et al. A computerized behavioral assessment for children with headaches. Headache 1984;24:182–5.
70. Bruni O, Galli F, Guidetti V. Sleep hygiene and migraine in children and adolescents. Cephalalgia 1999;19 Suppl 25:57–9.
71. Lynch-Jordan AM, Sil S, Peugh J, et al. Differential changes in functional disability and pain intensity over the course of psychological treatment for children with chronic pain. Pain 2014;155:1955–61.
72. Eccleston C, Palermo TM, Williams AC, et al. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database Syst Rev 2014;5:CD003968.
73. Huguet A, McGrath PJ, Stinson J, et al. Efficacy of psychological treatment for headaches: an overview of systematic reviews and analysis of potential modifiers of treatment efficacy. Clin J Pain 2014;30:353–69.
74. Kropp P, Meyer B, Landgraf M, et al. Headache in children: update on biobehavioral treatments. Neuropediatrics 2013;44:20–4.
75. Kröner-Herwig B. Psychological treatments for pediatric headache. Expert Rev Neurother 2011;11:403–10.
76. Powers SW, Andrasik F. Biobehavioral treatment, disability, and psychological effects of pediatric headache. Pediatr Ann 2005;34:461–5.
77. Hermann C, Blanchard EB. Biofeedback in the treatment of headache and other childhood pain. Appl Psychophysiol Biofeedback 2002;27:143–62.
78. Powers SW, Kashikar-Zuck SM, Hershey AD, et al. Cognitive behavioral therapy plus amitriptyline for chronic migraine in children and adolescents: a randomized clinical trial. JAMA 2013;310:2622–30.
79. Smitherman TA, Wells RE, Ford SG. Emerging behavioral treatments for migraine. Curr Pain Headache Rep 2015;19:13.
80. Slater SK, O’Brien HL, Hershey AD, et al. Psychiatric comorbidity in pediatric chronic daily headache. Cephalalgia 2012;32: 1116–22.
81. Pavlovic JM, Stewart WF, Bruce CA, et al. Burden of migraine related to menses: results from the AMPP study. J Headache Pain 2015;16:24.
82. MacGregor EA, Hackshaw A. Prevalence of migraine on each day of the natural menstrual cycle. Neurology 2004;63:351–3.
83. Granella F, Sances G, Allais G. Characteristics of menstrual and nonmenstrual attacks in women with menstrually related migraine referred to headache centres. Cephalalgia 2004;24:707–16.
84. Crawford MJ, Lehman L, Slater S, et al. Menstrual migraine in adolescents. Headache 2009;49:341–7.
85. Allais G, Bussone G, De Lorenzo C, et al. Naproxen sodium in short-term prophylaxis of pure menstrual migraine: pathophysiological and clinical considerations. Neurol Sci 2007;28(Suppl 2):S225–8.
86. Silberstein S, Elkind AH, Schreiber C, et al. A randomized trial of frovatriptan for the intermittent prevention of menstrual migraine. Neurology 2004;63:261–9.
87. Brandes JL, Poole A, Kallela M, et al. Short-term frovatriptan for the prevention of difficult-to-treat menstrual migraine attacks. Cephalalgia 2009;29:1133–48.
88. Newman L, Mannix LK, Landy S, et al. Naratriptan as short-term prophylaxis of menstrually associated migraine: a randomized, double-blind, placebo-controlled study. Headache 2001;41:248–56.
89. Mannix LK, Savani N, Landy S, et al. Efficacy and tolerability of naratriptan for short-term prevention of menstrually related migraine: data from two randomized, double-blind, placebo-controlled studies. Headache 2007;47:1037–49.
90. Newman LC, Lipton RB, Lay CL, et al. A pilot study of oral sumatriptan as intermittent prophylaxis of menstruation-related migraine. Neurology 1998;51:307–9.
91. Tuchman MM, Hee A, Emeribe U, et al. Oral zolmitriptan in the short-term prevention of menstrual migraine: a randomized, placebo-controlled study. CNS Drugs 2008;22:877–86.
92. Marcus DA, Bernstein CD, Sullivan EA, et al. Perimenstrual eletriptan prevents menstrual migraine: an open-label study. Headache 2010;50:551–6.
93. Silberstein S, Patel S. Menstrual migraine: an updated review on hormonal causes, prophylaxis and treatment. Expert Opin Pharmacother 2014;15:2063–70.
94. MacGregor EA. Migraine management during menstruation and menopause. Continuum (Minneap Minn) 2015;21(4 Headache):990–1003.
95. Aegidius K, Zwart JA, Hagen K, et al. Oral contraceptives and increased headache prevalence: The Head-HUNT Study. Neurology 2006;66:349–53.
96. Sulak PJ, Scow RD, Preece C, et al. Hormone withdrawal symptoms in oral contraceptive users. Obstet Gynecol 2000;95:261–6.
97. LaGuardia KD, Fisher AC, Bainbridge JD, et al. Suppression of estrogen-withdrawal headache with extended transdermal contraception. Fertil Steril 2005;83:1875–7.
98. Sulak P,Willis S, Kuehl T, Coffee A, Clark J. Headaches and oral contraceptives: Impact of eliminating the standard 7-day placebo interval. Headache 2007;47:27–37.
99. De Leo V, Scolaro V, Musacchio MC, et al. Combined oral contraceptives in women with menstrual migraine without aura. Fertil Steril 2011;96:917–20.
100. Calhoun AH. A novel specific prophylaxis for menstrual-associated migraine. South Med J 2004;97:819–22.
101. Coffee AL, Sulak PJ, Hill AJ, et al. Extended cycle combined oral contraceptives and prophylactic frovatriptan during the hormone-free interval in women with menstrual-related migraines. J Womens Health (Larchmt) 2014;23:310–7.
102. Vetvik KG, MacGregor EA, Lundqvist C, et al. Contraceptive-induced amenorrhea leads to reduced migraine frequency in women with menstrual migraine without aura. J Headache Pain 2014;15:30.
103. Sucato GS, Gerschultz KL. Extended cycle hormonal contraception in adolescents. Curr Opin Obstet Gynecol 2005;17:461–5.
104. Spector JT, Kahn SR, Jones MR, et al. Migraine headache and ischemic stroke risk: an updated meta-analysis. Am J Med 2010;123:612–24.
105. Bigal ME, Kurth T, Santanello N, et al. Migraine and cardiovascular disease: a population-based study. Neurology 2010;74:628–35.
106. Schurks M, Rist PM, Bigal ME, et al. Migraine and cardiovascular disease: systematic review and meta-analysis. BMJ 2009;339:b3914.
107. Gelfand AA, Fullerton HJ, Jacobson A, et al. Is migraine a risk factor for pediatric stroke? Cephalalgia 2015;35:1252–60.