User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Tape strips detect hidradenitis suppurativa biomarkers, novel study shows
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
, results from a novel study showed.
“Tape strips can provide important clues to when and which drugs to use in HS in patients with both early and late disease, which can change clinical practice,” corresponding study author Emma Guttman-Yassky, MD, PhD, professor and chair of dermatology at the Icahn School of Medicine at Mount Sinai in New York City, said in an interview. “It is noninvasive and nonscarring,” she added.
Tape stripping has been validated in atopic dermatitis, psoriasis, and other dermatologic conditions in recent years. For the current study, which was published online in the Journal of the American Academy of Dermatology, and is believed to be the first of its kind, Dr. Guttman-Yassky and colleagues performed RNA sequencing from large D-Squame tape strips collected from lesional and nonlesional skin of 22 patients with HS and from 21 age- and sex-matched healthy controls. They correlated the expression of skin biomarkers between tape strips and a previously published gene-signature of HS biopsies. The mean age of patients with HS was 43 years, while the mean age of healthy controls was 35. The average International Hidradenitis Suppurativa Severity Score System (IHS4) score of the HS cohort was 36.
Consistent with published studies, the researchers found that tape strips identified an overall higher inflammatory burden in HS. Specifically, they observed an upregulation of known cytokines within the following pathways: Th1 (such as IFNG, CXCL9/10/11, and CCR5); Th17 (such as interleukin [IL]-17A/F, IL12B, IL23A, CAMP, and CCL20); Th2 (such as IL4R, IL13/IL31/IL10, CCR4, CCL7/CCL13/CCL24, TNFSF4/OX40L, and TNFRSF4/OX40); and Th22 (such as IL22 and IL32).
The researchers also found that the expression of Th17 and tumor necrosis factor (TNF)–alpha pathways were highly correlated between tape strips and biopsies and that HS clinical severity was significantly associated with expression of biomarkers, such as TNF-alpha, IL17A/F, OX40, JAK1-3, and IL4R in HS lesional and/or nonlesional skin.
“It was quite unexpected that we are able to identify, using a minimally invasive approach that samples only the upper layers of the epidermis, products and processes that are considered to be deeper-situated, such as IL-17, and other immune markers,” Dr. Guttman-Yassky said in the interview. “We were also surprised to see how well the tape-stripped–derived skin molecular profile correlated with that of biopsies, as well as how well it correlated with the clinical disease severity of HS.”
Also surprising, she added, was that the biomarkers in nonlesional tape-stripped skin, such as IL-17 and TNF alpha, “show high correlations with disease severity and provide clues to early disease.”
If using tape strips in HS is validated in larger cohort studies, the potential cost implications of using this approach in practice remain unclear, Dr. Guttman-Yassky said. “It is currently not cheap, but we are hoping that one day, we can provide a means to diagnose the disease and treat it early, and appropriately, utilizing this approach,” she commented. “We are excited about the applicability of this study to the early treatment and longitudinal follow up of HS with drugs that are targeting specific immune molecules and pathways,” she said, adding that it will also be useful for helping determine which drug should be used for which patient.
She and her co-authors acknowledged certain limitations of the study, including its small sample size and the fact that tape stripping is limited to the epidermis.
Asked to comment on the study, Jennifer L. Hsiao, MD, a dermatologist who directs the HS clinic at the University of Southern California, Los Angeles, said the findings “have important potential implications for our ability to one day personalize treatments for a patient with early HS in a minimally invasive way.”
As the study authors point out, she added, “tape strips only allow sampling of the epidermis, which is limiting in a disease like HS where much of the disruption is in the dermis with deep nodules and dermal tunnels. However, our overall goal should be to catch patients in the early stages of their disease before the occurrence of irreversible tissue damage such as dermal tunnels. Thus, the ongoing campaign for early diagnosis and early intervention by various stakeholders in the field of HS can help mitigate the impact of this inherent limitation of tape strips. It will be exciting to see larger studies that investigate tape strip results in relation to clinical phenotypes, disease progression, and therapeutic responses.”
The study was funded by an International Dermatology Outcome Measures Hidradenitis Suppurativa Grant. Dr. Guttman-Yassky disclosed that she has been a consultant to, an adviser for, and has received research grants from many pharmaceutical companies. Of the remaining authors, 2 also had multiple disclosures and 11 had no disclosures. Dr. Hsiao disclosed that she is a member of the board of directors for the Hidradenitis Suppurativa Foundation. She has also served as a consultant for AbbVie, Aclaris, Boehringer Ingelheim, Incyte, Novartis, and UCB; as a speaker for AbbVie; and as an investigator for Amgen, Boehringer Ingelheim, and Incyte.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
New COVID variant JN.1 could disrupt holiday plans
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
No one planning holiday gatherings or travel wants to hear this, but the rise of a new COVID-19 variant, JN.1, is concerning experts, who say it may threaten those good times.
The good news is recent research suggests the 2023-2024 COVID-19 vaccine appears to work against this newest variant. But so few people have gotten the latest vaccine — less than 16% of U.S. adults — that some experts suggest it’s time for the CDC to urge the public who haven’t it to do so now, so the antibodies can kick in before the festivities.
“A significant wave [of JN.1] has started here and could be blunted with a high booster rate and mitigation measures,” said Eric Topol, MD, professor and executive vice president of Scripps Research in La Jolla, CA, and editor-in-chief of Medscape, a sister site of this news organization.
COVID metrics, meanwhile, have started to climb again. Nearly 10,000 people were hospitalized for COVID in the U.S. for the week ending Nov. 25, the CDC said, a 10% increase over the previous week.
Who’s Who in the Family Tree
JN.1, an Omicron subvariant, was first detected in the U.S. in September and is termed “a notable descendent lineage” of Omicron subvariant BA.2.86 by the World Health Organization. When BA.2.86, also known as Pirola, was first identified in August, it appeared very different from other variants, the CDC said. That triggered concerns it might be more infectious than previous ones, even for people with immunity from vaccination and previous infections.
“JN.1 is Pirola’s kid,” said Rajendram Rajnarayanan, PhD, assistant dean of research and associate professor at the New York Institute of Technology at Arkansas State University, who maintains a COVID-19 variant database. The variant BA.2.86 and offspring are worrisome due to the mutations, he said.
How Widespread Is JN.1?
As of Nov. 27, the CDC says, BA.2.86 is projected to comprise 5%-15% of circulating variants in the U.S. “The expected public health risk of this variant, including its offshoot JN.1, is low,” the agency said.
Currently, JN.1 is reported more often in Europe, Dr. Rajnarayanan said, but some countries have better reporting data than others. “It has probably spread to every country tracking COVID,’’ he said, due to the mutations in the spike protein that make it easier for it to bind and infect.
Wastewater data suggest the variant’s rise is helping to fuel a wave, Dr. Topol said.
Vaccine Effectiveness Against JN.1, Other New Variants
The new XBB.1.5 monovalent vaccine, protects against XBB.1.5, another Omicron subvariant, but also JN.1 and other “emergent” viruses, a team of researchers reported Nov. 26 in a study on bioRxiv that has not yet been certified by peer review.
The updated vaccine, when given to uninfected people, boosted antibodies about 27-fold against XBB.1.5 and about 13- to 27-fold against JN.1 and other emergent viruses, the researchers reported.
While even primary doses of the COVID vaccine will likely help protect against the new JN.1 subvariant, “if you got the XBB.1.5 booster, it is going to be protecting you better against this new variant,” Dr. Rajnarayanan said.
2023-2024 Vaccine Uptake Low
In November, the CDC posted the first detailed estimates of who did. As of Nov. 18, less than 16% of U.S. adults had, with nearly 15% saying they planned to get it.
Coverage among children is lower, with just 6.3% of children up to date on the newest vaccine and 19% of parents saying they planned to get the 2023-2024 vaccine for their children.
Predictions, Mitigation
While some experts say a peak due to JN.1 is expected in the weeks ahead, Dr. Topol said it’s impossible to predict exactly how JN.1 will play out.
“It’s not going to be a repeat of November 2021,” when Omicron surfaced, Dr. Rajnarayanan predicted. Within 4 weeks of the World Health Organization declaring Omicron as a virus of concern, it spread around the world.
Mitigation measures can help, Dr. Rajnarayanan said. He suggested:
Get the new vaccine, and especially encourage vulnerable family and friends to do so.
If you are gathering inside for holiday festivities, improve circulation in the house, if possible.
Wear masks in airports and on planes and other public transportation.
A version of this article appeared on WebMD.com.
FDA approves first 2 gene-editing therapies for sickle cell
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.
These “milestone treatments” mark the first cell-based gene therapies for this debilitating and potentially life-threatening blood disorder that affects about 100,000 people in the United States.
The two therapies are exagamglogene autotemcel, or exa-cel (Casgevy; Vertex Pharmaceuticals and Crispr Therapeutics), and lovotibeglogene autotemcel, or lovo-cel (Lyfgenia; bluebird bio).
“The approval of the first gene therapies for [sickle cell disease] represents a tremendous step forward for the [sickle cell] community, which has been historically overlooked and underfunded,” said Robert A. Brodsky, MD, of Johns Hopkins University School of Medicine, in a statement from the American Society of Hematology, following the approval.
“We are excited to advance the field, especially for individuals whose lives have been severely disrupted by the disease, by approving two cell-based gene therapies today,” Nicole Verdun, MD, of the FDA’s Center for Biologics Evaluation and Research, added in an agency press release.
Sickle cell disease involves a mutation in hemoglobin, a protein in red blood cells that provides oxygen to tissues. The mutation leads red blood cells to develop a crescent or sickle shape, which can restrict blood flow and cause severe pain and organ damage, known as vaso-occlusive events or crises.
Treatment options prior to these approvals primarily included red blood transfusions and hydroxyurea alongside pain management. The only potential curative option has been allogeneic hematopoietic stem cell transplantation, but that comes with significant risks and most patients don’t have an appropriate donor.
Exa-cel
Exa-cel uses CRISPR gene-editing technology. Before the infusion, patients undergo myeloablative conditioning, which removes cells from the bone marrow. These cells are genetically modified to produce fetal hemoglobin. Patients then receive an infusion of the edited cells, which can help restore normal hemoglobin production.
The FDA approval was based on data from the pivotal CLIMB SCD-121 trial. In an October advisory committee meeting, the FDA highlighted trial data demonstrating that 29 of 31 patients reached the trial’s primary endpoint: freedom from severe vaso-occlusive crises over a 12-month period. In addition, 28 of these patients remained free of vaso-occlusive crises for almost 2 years.
The committee noted that one of the 31 patients died about 9 months after receiving an exa-cel infusion.
The cell-based gene therapy also increased both fetal and total hemoglobin, with total hemoglobin levels increasing to > 11 g/dL by month 3 and remaining at that level afterward. No patients experienced graft failure or rejection.
The most common side effects included low platelets and white blood cell counts, mouth sores, nausea, musculoskeletal pain, vomiting, and febrile neutropenia.
Exa-cel could “provide a one-time functional cure” for patients with severe sickle cell disease, according to Franco Locatelli, MD, of Sapienza University of Rome, who presented initial findings last year.
While the current approval is for patients with infusion-dependent sickle cell disease, exa-cel is also being evaluated in patients with another blood disorder, beta-thalassemia.
Lovo-cel
Lovo-cel, a cell-based gene therapy, uses a different technology — a lentiviral vector, or gene delivery vehicle — that can also genetically modify a patient’s blood stem cells.
Like exa-cel, lovo-cel is a one-time, single-dose infusion that contains the patient’s modified cells. Before the infusion, patients undergo myeloablative conditioning. The patient’s stem cells are then genetically modified to allow them to produce the most common form of hemoglobin, HbA
This approval was based on data from a single-arm, 24-month study in patients aged 12-50 years who had sickle cell disease and a history of vaso-occlusive events.
Overall, 88% of patients (28 of 32) achieved complete resolution of vaso-occlusive events 6-18 months after the infusion.
The most common side effects included stomatitis; febrile neutropenia; and low platelet, white blood cell, and red blood cell counts.
The FDA noted that hematologic cancer has occurred in patients treated with lovo-cel, and the label includes a black-box warning about the risk.
Dr. Brodsky noted, however, that “while these new gene therapies are potentially life-changing for individuals living with [sickle cell disease], they must be accessible to be effective.”
Access is a potential concern. Exa-cel and lovo-cel could cost about $2 million.
A version of this article appeared on Medscape.com.
Just gas? New study on colic suggests some longer-term implications
Pediatricians commonly are asked to see infants presenting with symptoms of colic. The frequent and intense crying associated with colic is understandably quite distressing to parents, who often worry about a serious underlying medical cause. There also is the stress of trying to soothe an irritable infant who often does not seem to respond to the typical interventions.
Conventional wisdom about colic has been that the behaviors are the result of some gastrointestinal problem that, while not perfectly understood, tends to be mercifully self-limited and not predictive of future medical or mental health problems. This perspective then leads to pediatricians typically offering mainly sympathy and reassurance at these visits.
A new study,1 however, challenges some of this traditional thinking. The data come from a remarkable longitudinal study called the Generation R Study (R being Rotterdam in the Netherlands) that has prospectively studied a group of nearly 5,000 children from before birth into adolescence. Colic symptoms were briefly assessed when infants were about 3 months old and emotional-behavioral problems have been prospectively measured at multiple time points subsequently using well-validated rating scales.
The main finding of the study was and, to a lesser extent, in adolescence. This held for both internalizing problems (like anxiety and depressive symptoms) and externalizing problems (like defiance and aggressive behavior). At age 10, participants also underwent an MRI scan and those who were excessive criers as infants were found to have a smaller amygdala, a region known for being important in regulating emotions.
The authors concluded that colicky behavior in infancy may reflect some underlying temperamental vulnerabilities and may have more predictive value than previously thought. The connection between excessive crying and a measurable brain region difference later in life is also interesting, although these kinds of brain imaging findings have been notoriously difficult to interpret clinically.
Overall, this is a solid study that deserves to be considered. Colic may reflect a bit more than most of us have been taught and shouldn’t necessarily be “shrugged off,” as the authors state in their discussion.
At the same time, however, it is important not to overinterpret the findings. The magnitude of the effects were on the small side (about 0.2 of a standard deviation) and most children with excessive crying in early infancy did not manifest high levels of mental health problems later in life. The mothers of high crying infants also had slightly higher levels of mental health problems themselves so there could be other mechanisms at work here, such as genetic differences between the two groups.
So how could a pediatrician best use this new information without taking things too far? Regardless of the question of whether the excessive crying infancy is a true risk factor for later behavior problems (in the causal sense) or whether it represents more of a marker for something else, its presence so early in life offers an opportunity. Primary care clinicians would still likely want to provide the reassurance that has typically been given in these visits but perhaps with the caveat that some of these kids go on to struggle a bit more with mental health and that they might benefit from some additional support. We are not talking about prophylactic medications here, but something like additional parenting skills. Especially if you, as the pediatrician, suspect that the parents might benefit from expanding their parenting toolkit already, here is a nice opportunity to invite them to learn some new approaches and skills — framed in a way that focuses on the temperament of the child rather than any “deficits” you perceive in the parents. Some parents may be more receptive and less defensive to the idea of participating in parent training under the framework that they are doing this because they have a temperamentally more challenging child (rather than feeling that they are deficient in basic parenting skills).
It’s always a good idea to know about what resources are available in the community when it comes to teaching parenting skills. In addition to scientifically supported books and podcasts, there has been a steady increase in reliable websites, apps, and other digital platforms related to parenting, as well as standard in-person groups and classes. This could also be a great use of an integrated behavioral health professional for practices fortunate enough to have one.
In summary, there is some new evidence that colic can represent a little more than “just gas,” and while we shouldn’t take this one study to the extreme, there may be some good opportunities here to discuss and support good parenting practices in general.
Dr. Rettew is a child and adolescent psychiatrist with Lane County Behavioral Health in Eugene, Ore., and Oregon Health & Science University, Portland. His latest book is “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
1. Sammallahti S et al. Excessive crying, behavior problems, and amygdala volume: A study from infancy to adolescence. J Am Acad Child Adolesc Psychiatry. 2023 Jun;62(6):675-83. doi: 10.1016/j.jaac.2023.01.014.
Pediatricians commonly are asked to see infants presenting with symptoms of colic. The frequent and intense crying associated with colic is understandably quite distressing to parents, who often worry about a serious underlying medical cause. There also is the stress of trying to soothe an irritable infant who often does not seem to respond to the typical interventions.
Conventional wisdom about colic has been that the behaviors are the result of some gastrointestinal problem that, while not perfectly understood, tends to be mercifully self-limited and not predictive of future medical or mental health problems. This perspective then leads to pediatricians typically offering mainly sympathy and reassurance at these visits.
A new study,1 however, challenges some of this traditional thinking. The data come from a remarkable longitudinal study called the Generation R Study (R being Rotterdam in the Netherlands) that has prospectively studied a group of nearly 5,000 children from before birth into adolescence. Colic symptoms were briefly assessed when infants were about 3 months old and emotional-behavioral problems have been prospectively measured at multiple time points subsequently using well-validated rating scales.
The main finding of the study was and, to a lesser extent, in adolescence. This held for both internalizing problems (like anxiety and depressive symptoms) and externalizing problems (like defiance and aggressive behavior). At age 10, participants also underwent an MRI scan and those who were excessive criers as infants were found to have a smaller amygdala, a region known for being important in regulating emotions.
The authors concluded that colicky behavior in infancy may reflect some underlying temperamental vulnerabilities and may have more predictive value than previously thought. The connection between excessive crying and a measurable brain region difference later in life is also interesting, although these kinds of brain imaging findings have been notoriously difficult to interpret clinically.
Overall, this is a solid study that deserves to be considered. Colic may reflect a bit more than most of us have been taught and shouldn’t necessarily be “shrugged off,” as the authors state in their discussion.
At the same time, however, it is important not to overinterpret the findings. The magnitude of the effects were on the small side (about 0.2 of a standard deviation) and most children with excessive crying in early infancy did not manifest high levels of mental health problems later in life. The mothers of high crying infants also had slightly higher levels of mental health problems themselves so there could be other mechanisms at work here, such as genetic differences between the two groups.
So how could a pediatrician best use this new information without taking things too far? Regardless of the question of whether the excessive crying infancy is a true risk factor for later behavior problems (in the causal sense) or whether it represents more of a marker for something else, its presence so early in life offers an opportunity. Primary care clinicians would still likely want to provide the reassurance that has typically been given in these visits but perhaps with the caveat that some of these kids go on to struggle a bit more with mental health and that they might benefit from some additional support. We are not talking about prophylactic medications here, but something like additional parenting skills. Especially if you, as the pediatrician, suspect that the parents might benefit from expanding their parenting toolkit already, here is a nice opportunity to invite them to learn some new approaches and skills — framed in a way that focuses on the temperament of the child rather than any “deficits” you perceive in the parents. Some parents may be more receptive and less defensive to the idea of participating in parent training under the framework that they are doing this because they have a temperamentally more challenging child (rather than feeling that they are deficient in basic parenting skills).
It’s always a good idea to know about what resources are available in the community when it comes to teaching parenting skills. In addition to scientifically supported books and podcasts, there has been a steady increase in reliable websites, apps, and other digital platforms related to parenting, as well as standard in-person groups and classes. This could also be a great use of an integrated behavioral health professional for practices fortunate enough to have one.
In summary, there is some new evidence that colic can represent a little more than “just gas,” and while we shouldn’t take this one study to the extreme, there may be some good opportunities here to discuss and support good parenting practices in general.
Dr. Rettew is a child and adolescent psychiatrist with Lane County Behavioral Health in Eugene, Ore., and Oregon Health & Science University, Portland. His latest book is “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
1. Sammallahti S et al. Excessive crying, behavior problems, and amygdala volume: A study from infancy to adolescence. J Am Acad Child Adolesc Psychiatry. 2023 Jun;62(6):675-83. doi: 10.1016/j.jaac.2023.01.014.
Pediatricians commonly are asked to see infants presenting with symptoms of colic. The frequent and intense crying associated with colic is understandably quite distressing to parents, who often worry about a serious underlying medical cause. There also is the stress of trying to soothe an irritable infant who often does not seem to respond to the typical interventions.
Conventional wisdom about colic has been that the behaviors are the result of some gastrointestinal problem that, while not perfectly understood, tends to be mercifully self-limited and not predictive of future medical or mental health problems. This perspective then leads to pediatricians typically offering mainly sympathy and reassurance at these visits.
A new study,1 however, challenges some of this traditional thinking. The data come from a remarkable longitudinal study called the Generation R Study (R being Rotterdam in the Netherlands) that has prospectively studied a group of nearly 5,000 children from before birth into adolescence. Colic symptoms were briefly assessed when infants were about 3 months old and emotional-behavioral problems have been prospectively measured at multiple time points subsequently using well-validated rating scales.
The main finding of the study was and, to a lesser extent, in adolescence. This held for both internalizing problems (like anxiety and depressive symptoms) and externalizing problems (like defiance and aggressive behavior). At age 10, participants also underwent an MRI scan and those who were excessive criers as infants were found to have a smaller amygdala, a region known for being important in regulating emotions.
The authors concluded that colicky behavior in infancy may reflect some underlying temperamental vulnerabilities and may have more predictive value than previously thought. The connection between excessive crying and a measurable brain region difference later in life is also interesting, although these kinds of brain imaging findings have been notoriously difficult to interpret clinically.
Overall, this is a solid study that deserves to be considered. Colic may reflect a bit more than most of us have been taught and shouldn’t necessarily be “shrugged off,” as the authors state in their discussion.
At the same time, however, it is important not to overinterpret the findings. The magnitude of the effects were on the small side (about 0.2 of a standard deviation) and most children with excessive crying in early infancy did not manifest high levels of mental health problems later in life. The mothers of high crying infants also had slightly higher levels of mental health problems themselves so there could be other mechanisms at work here, such as genetic differences between the two groups.
So how could a pediatrician best use this new information without taking things too far? Regardless of the question of whether the excessive crying infancy is a true risk factor for later behavior problems (in the causal sense) or whether it represents more of a marker for something else, its presence so early in life offers an opportunity. Primary care clinicians would still likely want to provide the reassurance that has typically been given in these visits but perhaps with the caveat that some of these kids go on to struggle a bit more with mental health and that they might benefit from some additional support. We are not talking about prophylactic medications here, but something like additional parenting skills. Especially if you, as the pediatrician, suspect that the parents might benefit from expanding their parenting toolkit already, here is a nice opportunity to invite them to learn some new approaches and skills — framed in a way that focuses on the temperament of the child rather than any “deficits” you perceive in the parents. Some parents may be more receptive and less defensive to the idea of participating in parent training under the framework that they are doing this because they have a temperamentally more challenging child (rather than feeling that they are deficient in basic parenting skills).
It’s always a good idea to know about what resources are available in the community when it comes to teaching parenting skills. In addition to scientifically supported books and podcasts, there has been a steady increase in reliable websites, apps, and other digital platforms related to parenting, as well as standard in-person groups and classes. This could also be a great use of an integrated behavioral health professional for practices fortunate enough to have one.
In summary, there is some new evidence that colic can represent a little more than “just gas,” and while we shouldn’t take this one study to the extreme, there may be some good opportunities here to discuss and support good parenting practices in general.
Dr. Rettew is a child and adolescent psychiatrist with Lane County Behavioral Health in Eugene, Ore., and Oregon Health & Science University, Portland. His latest book is “Parenting Made Complicated: What Science Really Knows about the Greatest Debates of Early Childhood.” You can follow him on Twitter and Facebook @PediPsych.
Reference
1. Sammallahti S et al. Excessive crying, behavior problems, and amygdala volume: A study from infancy to adolescence. J Am Acad Child Adolesc Psychiatry. 2023 Jun;62(6):675-83. doi: 10.1016/j.jaac.2023.01.014.
Some reasons to get off the fence about COVID booster
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Though many people remain on the fence about getting the latest COVID vaccine booster, new research suggests a strong argument for getting the shot this winter: It sharply reduces the risk for COVID.
The risk reduction was 37% for those who received two doses. Experts say the research provides a strong argument for getting the vaccine, noting that about 10% of people infected with COVID go on to have long COVID, which can be debilitating for one quarter of those with long-lasting symptoms.
The data come from a systematic literature review and meta-analysis published in October in Antimicrobial Stewardship & Epidemiology. Researchers examined 32 studies published between December 2019 and June 2023, involving 775,931 adults. Twenty-four studies, encompassing 620,221 individuals, were included in the meta-analysis.
“The body of evidence from all these different studies converge on one single reality — that vaccines reduce the risk of long COVID, and people who keep up to date on their vaccinations also fared better than people who got it once or twice and didn’t follow up,” said Ziyad Al-Aly, MD, a clinical epidemiologist at Washington University in St Louis.
Researchers have reported similar results for children. The National Institutes of Health RECOVER Initiative team found that vaccines are up to 42% effective in preventing long COVID in children, said Dr. Carlos Oliveira, MD, a pediatric infectious diseases specialist and Yale researcher who contributed to the study, which is in preprint.
Vaccines also protect children from multisystem inflammatory syndrome, a condition that can happen after COVID, as well as protect against other COVID-related problems, such as missed school days, Oliveira said. “Even if the vaccine doesn’t completely stop long COVID, it’s still good for kids to get vaccinated for all these other reasons.”
However, uptake for the latest boosters has been slow: the Centers for Disease Control and Prevention reported that by mid-November, less than 16% of people aged 18 years or older had received a shot. For children, the number was closer to 6%. A recent Kaiser Family Foundation survey found that booster rates for adults are similar to what it was 1 year ago.
The survey results suggest that people are no longer as worried about COVID, which is why there is less concerned about keeping up with boosters. Though the current mutation of the virus is not as debilitating as its predecessors, long COVID continues to be a problem: as of January 2023, 28% of people who had contracted the virus had experienced long-COVID symptoms. And though the mechanisms are still not fully understood, and researchers have yet to agree on a definition of long COVID, they are certain about this much: The best way to avoid it is to avoid getting infected to begin with.
The lack of a diagnostic test for long COVID and the fact that the symptoms mimic those of other diseases lead to inconsistency that can make studies hard to replicate. In the papers reviewed for the Antimicrobial Stewardship & Epidemiology study, long COVID was defined as having symptoms lasting from more than 4 weeks to more than 6 months. Alexandre Marra, MD, the lead author and a researcher at the Hospital Israelita Albert Einstein, in São Paulo, Brazil, and at the University of Iowa, said that a clear standard definition is needed to better understand the actual prevalence and evaluate vaccine effectiveness.
Al-Aly noted that there is a logical explanation for one finding in the paper: The percentage of individuals who had COVID and reported that long-COVID symptoms declined from 19% in June 2022 to 11% in January 2023.
Because a pandemic is a dynamic event, constantly producing different variants with different phenotypes, the prevalence of disease is naturally going to be affected. “People who got infected early in the pandemic may have a different long COVID profile and long COVID risk than people who got infected in the second or third year of the pandemic,” Al-Aly said.
Most of the studies reported data from before the Omicron-variant era. Only eight reported data during that era. Omicron was not as lethal as previous variants, and consequently, fewer patients developed long COVID during that time.
One of those who did is Yeng Chang, age 40 years, a family doctor who lives in Sherwood Park, Alberta, Canada. Chang developed long COVID during fall 2022 after getting the virus in June. By then, she’d been vaccinated three times, but she isn’t surprised that she got sick because each vaccine she had was developed before Omicron.
“When I had COVID I was really sick, but I was well enough to stay home,” she said. “I think if I didn’t have my immunizations, I might have been hospitalized, and I don’t know what would have happened.”
Long COVID has left Chang with brain fog, fatigue, and a lack of physical stamina that forced her to pause her medical practice. For the past year and a half, she’s spent more time as a patient than a physician.
Chang had her fifth COVID vaccination in the fall and recommends that others do the same. “The booster you got however many years ago was effective for the COVID of that time but there is a new COVID now. You can’t just say, ‘I had one and I’m fine forever.’”
A version of this article appeared on Medscape.com.
Global measles deaths increased by 43% in 2022
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
The number of total reported cases rose by 18% over the same period, accounting for approximately 9 million cases and 136,000 deaths globally, mostly among children. This information comes from a new report by the World Health Organization (WHO), published in partnership with the US Centers for Disease Control and Prevention (CDC).
More Measles Outbreaks
The report also notes an increase in the number of countries experiencing significant measles outbreaks. There were 37 such countries in 2022, compared with 22 the previous year. The most affected continents were Africa and Asia.
“The rise in measles outbreaks and deaths is impressive but, unfortunately, not surprising, given the decline in vaccination rates in recent years,” said John Vertefeuille, PhD, director of the CDC’s Global Immunization Division.
Vertefeuille emphasized that measles cases anywhere in the world pose a risk to “countries and communities where people are undervaccinated.” In recent years, several regions have fallen short of their immunization targets.
Vaccination Trends
In 2022, there was a slight increase in measles vaccination after a decline exacerbated by the COVID-19 pandemic and its impact on global healthcare systems. However, 33 million children did not receive at least one dose of the vaccine last year: 22 million missed the first dose, and 11 million missed the second.
For communities to be considered protected against outbreaks, immunization coverage with the full vaccine cycle should be at least 95%. The global coverage rate for the first dose was 83%, and for the second, it was 74%.
Nevertheless, immunization recovery has not reached the poorest countries, where the immunization rate stands at 66%. Brazil is among the top 10 countries where more children missed the first dose in 2022. These nations account for over half of the 22 million unadministered vaccines. According to the report, half a million children did not receive the vaccine in Brazil.
Measles in Brazil
Brazil’s results highlight setbacks in vaccination efforts. In 2016, the country was certified to have eliminated measles, but after experiencing outbreaks in 2018, the certification was lost in 2019. In 2018, Brazil confirmed 9325 cases. The situation worsened in 2019 with 20,901 diagnoses. Since then, numbers have been decreasing: 8100 in 2020, 676 in 2021, and 44 in 2022.
Last year, four Brazilian states reported confirmed virus cases: Rio de Janeiro, Pará, São Paulo, and Amapá. Ministry of Health data indicated no confirmed measles cases in Brazil as of June 15, 2023.
Vaccination in Brazil
Vaccination coverage in Brazil, which once reached 95%, has sharply declined in recent years. The rate of patients receiving the full immunization scheme was 59% in 2021.
Globally, although the COVID-19 pandemic affected measles vaccination, measures like social isolation and mask use potentially contributed to reducing measles cases. The incidence of the disease decreased in 2020 and 2021 but is now rising again.
“From 2021 to 2022, reported measles cases increased by 67% worldwide, and the number of countries experiencing large or disruptive outbreaks increased by 68%,” the report stated.
Because of these data, the WHO and the CDC urge increased efforts for vaccination, along with improvements in epidemiological surveillance systems, especially in developing nations. “Children everywhere have the right to be protected by the lifesaving measles vaccine, no matter where they live,” said Kate O’Brien, MD, director of immunization, vaccines, and biologicals at the WHO.
“Measles is called the virus of inequality for a good reason. It is the disease that will find and attack those who are not protected.”
This article was translated from the Medscape Portuguese edition.
Patients with hypermobile Ehlers-Danlos syndrome report skin laxity, scarring
.
The genetic cause of hEDS, a common inherited connective tissue disorder, remains unknown, wrote Alan Snyder, MD, of the department of dermatology and dermatologic surgery at Medical University of South Carolina, Charleston, and colleagues.
Previous research suggests that changes in dermal mechanics predispose these patients to a range of skin conditions including mast cell activation disorder (MCAD) spectrum and chronic spontaneous urticaria, abnormal scars or wound healing, piezogenic papules, dyshidrosis, skin laxity or softness, easy bruising, local anesthesia resistance, keratosis pilaris, striae, and hidradenitis suppurativa, the researchers wrote.
However, data on these and other dermatologic manifestations of hEDS are limited, they said.
The diagnosis of hEDS will continue to be made more frequently and carefully, as the condition becomes more recognized and understood in the medical community, especially with anticipated capabilities of genetic testing, Dr. Snyder said in an interview.
“Being able to be aware of disease-specific comorbidities, such as those discovered in this study, allows providers to better stratify phenotypes and improve patient disease co-management,” he said.
In the study, published in the Journal of the American Academy of Dermatology, the researchers reviewed data on 1,364 patients with ICD-10 or ICD-9 codes for hEDS or EDS unspecified who were seen at a single institution between June 2005 and May 2022. Most of the patients were White (95.4%) and female (86.7%); the average age was 29.2 years.
Of the 1,364 patients included in the chart review, 497 (36.4%) had documented skin manifestations. Of these, 118 (24.2%) had disorders of follicular occlusion (12 had hidradenitis suppurativa, 32 had folliculitis, and 74 had acne); 112 (23%) had eczema or atopic dermatitis, 98 (19.7%) had mast cell disorder, 32 (6.4%) had psoriasis, and 32 (6.4%) had wound healing issues (16 had hypertrophic keloids/scarring, 5 had abscesses, 3 had abnormal bruising, and 8 had other would healing issues).
The study also included results of a multiple-choice patient survey from 1,354 individuals. In the survey, approximately two-thirds of patients reported abnormal scarring, abnormal wound healing, and cutaneous laxity (61.7%, 69.0%, and 71.0%, respectively).
The findings were limited by several factors including the retrospective study design, lack of testing to confirm hEDS diagnosis, and the potential interdisciplinary selection bias for diagnoses, the authors noted.
However, the results support previous studies showing increased rates of occlusive conditions in hEDS and higher rates of acne, folliculitis, and psoriasis, and highlight the need for clinician education to manage patients and promote better outcomes, the researchers concluded.
Data Enhance Clinical Awareness
“Given the increasingly understood relation between TH2-directed and mast-cell mediated diseases and hEDS, it was not necessarily a surprise to find the increased prevalence of atopy and mast cell disease, but rather an interesting confirmation, within the limitations that exist with retrospective chart review,” Dr. Snyder told this news organization. “While it may make some intuitive sense that certain cohorts with higher risk of HS may have a higher risk of acne, this had not been reported in the literature to date,” he noted. “Given the high levels of patient reported issues with scarring and wound healing, I was surprised that so few analogous diagnoses were physician-reported in the medical records.”
In clinical practice, “health care professionals and patients need to be aware hEDS is associated with high rates of eczematous, mast-cell mediated and follicular occlusive cutaneous disorders,” Dr. Snyder said in an interview. “There seems to be a discrepancy between patients and physician awareness of scarring or wound healing issues in this patient population,” he added.
Looking ahead, “we need to better research and characterize the various hEDS phenotypes to understand who is at highest risk for various TH2-mediated or follicular occlusive disorders,” said Dr. Snyder. “Moreover, a greater understanding is needed of the wound healing inadequacies that predispose these patients to poor outcomes during dermatologic surgery,” he said.
The study was supported by the Ehlers-Danlos Society and the Milton and Tamar Maltz Family Foundation. The researchers had no financial conflicts to disclose.
.
The genetic cause of hEDS, a common inherited connective tissue disorder, remains unknown, wrote Alan Snyder, MD, of the department of dermatology and dermatologic surgery at Medical University of South Carolina, Charleston, and colleagues.
Previous research suggests that changes in dermal mechanics predispose these patients to a range of skin conditions including mast cell activation disorder (MCAD) spectrum and chronic spontaneous urticaria, abnormal scars or wound healing, piezogenic papules, dyshidrosis, skin laxity or softness, easy bruising, local anesthesia resistance, keratosis pilaris, striae, and hidradenitis suppurativa, the researchers wrote.
However, data on these and other dermatologic manifestations of hEDS are limited, they said.
The diagnosis of hEDS will continue to be made more frequently and carefully, as the condition becomes more recognized and understood in the medical community, especially with anticipated capabilities of genetic testing, Dr. Snyder said in an interview.
“Being able to be aware of disease-specific comorbidities, such as those discovered in this study, allows providers to better stratify phenotypes and improve patient disease co-management,” he said.
In the study, published in the Journal of the American Academy of Dermatology, the researchers reviewed data on 1,364 patients with ICD-10 or ICD-9 codes for hEDS or EDS unspecified who were seen at a single institution between June 2005 and May 2022. Most of the patients were White (95.4%) and female (86.7%); the average age was 29.2 years.
Of the 1,364 patients included in the chart review, 497 (36.4%) had documented skin manifestations. Of these, 118 (24.2%) had disorders of follicular occlusion (12 had hidradenitis suppurativa, 32 had folliculitis, and 74 had acne); 112 (23%) had eczema or atopic dermatitis, 98 (19.7%) had mast cell disorder, 32 (6.4%) had psoriasis, and 32 (6.4%) had wound healing issues (16 had hypertrophic keloids/scarring, 5 had abscesses, 3 had abnormal bruising, and 8 had other would healing issues).
The study also included results of a multiple-choice patient survey from 1,354 individuals. In the survey, approximately two-thirds of patients reported abnormal scarring, abnormal wound healing, and cutaneous laxity (61.7%, 69.0%, and 71.0%, respectively).
The findings were limited by several factors including the retrospective study design, lack of testing to confirm hEDS diagnosis, and the potential interdisciplinary selection bias for diagnoses, the authors noted.
However, the results support previous studies showing increased rates of occlusive conditions in hEDS and higher rates of acne, folliculitis, and psoriasis, and highlight the need for clinician education to manage patients and promote better outcomes, the researchers concluded.
Data Enhance Clinical Awareness
“Given the increasingly understood relation between TH2-directed and mast-cell mediated diseases and hEDS, it was not necessarily a surprise to find the increased prevalence of atopy and mast cell disease, but rather an interesting confirmation, within the limitations that exist with retrospective chart review,” Dr. Snyder told this news organization. “While it may make some intuitive sense that certain cohorts with higher risk of HS may have a higher risk of acne, this had not been reported in the literature to date,” he noted. “Given the high levels of patient reported issues with scarring and wound healing, I was surprised that so few analogous diagnoses were physician-reported in the medical records.”
In clinical practice, “health care professionals and patients need to be aware hEDS is associated with high rates of eczematous, mast-cell mediated and follicular occlusive cutaneous disorders,” Dr. Snyder said in an interview. “There seems to be a discrepancy between patients and physician awareness of scarring or wound healing issues in this patient population,” he added.
Looking ahead, “we need to better research and characterize the various hEDS phenotypes to understand who is at highest risk for various TH2-mediated or follicular occlusive disorders,” said Dr. Snyder. “Moreover, a greater understanding is needed of the wound healing inadequacies that predispose these patients to poor outcomes during dermatologic surgery,” he said.
The study was supported by the Ehlers-Danlos Society and the Milton and Tamar Maltz Family Foundation. The researchers had no financial conflicts to disclose.
.
The genetic cause of hEDS, a common inherited connective tissue disorder, remains unknown, wrote Alan Snyder, MD, of the department of dermatology and dermatologic surgery at Medical University of South Carolina, Charleston, and colleagues.
Previous research suggests that changes in dermal mechanics predispose these patients to a range of skin conditions including mast cell activation disorder (MCAD) spectrum and chronic spontaneous urticaria, abnormal scars or wound healing, piezogenic papules, dyshidrosis, skin laxity or softness, easy bruising, local anesthesia resistance, keratosis pilaris, striae, and hidradenitis suppurativa, the researchers wrote.
However, data on these and other dermatologic manifestations of hEDS are limited, they said.
The diagnosis of hEDS will continue to be made more frequently and carefully, as the condition becomes more recognized and understood in the medical community, especially with anticipated capabilities of genetic testing, Dr. Snyder said in an interview.
“Being able to be aware of disease-specific comorbidities, such as those discovered in this study, allows providers to better stratify phenotypes and improve patient disease co-management,” he said.
In the study, published in the Journal of the American Academy of Dermatology, the researchers reviewed data on 1,364 patients with ICD-10 or ICD-9 codes for hEDS or EDS unspecified who were seen at a single institution between June 2005 and May 2022. Most of the patients were White (95.4%) and female (86.7%); the average age was 29.2 years.
Of the 1,364 patients included in the chart review, 497 (36.4%) had documented skin manifestations. Of these, 118 (24.2%) had disorders of follicular occlusion (12 had hidradenitis suppurativa, 32 had folliculitis, and 74 had acne); 112 (23%) had eczema or atopic dermatitis, 98 (19.7%) had mast cell disorder, 32 (6.4%) had psoriasis, and 32 (6.4%) had wound healing issues (16 had hypertrophic keloids/scarring, 5 had abscesses, 3 had abnormal bruising, and 8 had other would healing issues).
The study also included results of a multiple-choice patient survey from 1,354 individuals. In the survey, approximately two-thirds of patients reported abnormal scarring, abnormal wound healing, and cutaneous laxity (61.7%, 69.0%, and 71.0%, respectively).
The findings were limited by several factors including the retrospective study design, lack of testing to confirm hEDS diagnosis, and the potential interdisciplinary selection bias for diagnoses, the authors noted.
However, the results support previous studies showing increased rates of occlusive conditions in hEDS and higher rates of acne, folliculitis, and psoriasis, and highlight the need for clinician education to manage patients and promote better outcomes, the researchers concluded.
Data Enhance Clinical Awareness
“Given the increasingly understood relation between TH2-directed and mast-cell mediated diseases and hEDS, it was not necessarily a surprise to find the increased prevalence of atopy and mast cell disease, but rather an interesting confirmation, within the limitations that exist with retrospective chart review,” Dr. Snyder told this news organization. “While it may make some intuitive sense that certain cohorts with higher risk of HS may have a higher risk of acne, this had not been reported in the literature to date,” he noted. “Given the high levels of patient reported issues with scarring and wound healing, I was surprised that so few analogous diagnoses were physician-reported in the medical records.”
In clinical practice, “health care professionals and patients need to be aware hEDS is associated with high rates of eczematous, mast-cell mediated and follicular occlusive cutaneous disorders,” Dr. Snyder said in an interview. “There seems to be a discrepancy between patients and physician awareness of scarring or wound healing issues in this patient population,” he added.
Looking ahead, “we need to better research and characterize the various hEDS phenotypes to understand who is at highest risk for various TH2-mediated or follicular occlusive disorders,” said Dr. Snyder. “Moreover, a greater understanding is needed of the wound healing inadequacies that predispose these patients to poor outcomes during dermatologic surgery,” he said.
The study was supported by the Ehlers-Danlos Society and the Milton and Tamar Maltz Family Foundation. The researchers had no financial conflicts to disclose.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
An alternative to walking out
Organized labor seems to be experiencing a rebirth of sorts. In October 2022 a strike by railroad workers was averted when a tentative agreement about wages, working conditions, health insurance, and medical leave was hammered out. This past fall, strikes by auto workers that threatened to paralyze the big three manufacturers have now been resolved with agreements that meet many of the workers’ demands. The President even made an appearance on a picket line. Baristas at coffee shops, screenwriters, and actors have all been involved in work actions around the country.
While the health care industry has been relatively immune to threatened work stoppages, there are a growing number of hospitals and clinics where nurses and physicians are exploring the possibility of organizing to give themselves a stronger voice in how health care is being delivered. The realities that come when you transition from owner to employee are finally beginning to sink in for physicians, whether they are specialists or primary care providers.
One of the most significant efforts toward unionization recently occurred in Minnesota and Wisconsin. About 400 physicians and 150 physician’s assistants and nurse practitioners employed at Allina Health System voted to unionize and join the Doctors Council.
In an interview with Jacobin, a publication that offers a socialist perspective, three of the providers involved in the process that led to the vote shared their observations. The physicians claim that the first steps toward unionization came after multiple efforts to work with the Allina’s administration were rebuffed. As primary care physicians, their initial demands focused on getting help with hiring staffing and getting support with paperwork and administrative obligations.
The organizers complained that while Medicare hoped to bolster primary care by paying the providers more, the funds went to the companies, who then distributed them in a way that often did little to help the overworked providers. In addition to achieving a more equitable distribution of the monies, one of the organizers sees unionization as a way to provide a layer of protection when providers feel they must speak out about situations which clearly put quality of care at risk.
The organizers say the idea of unionization has been particularly appealing to the younger providers who are feeling threatened by burnout. When these new physicians look to their older coworkers for advice, they often find that the seasoned employees are as stressed as they are. Realizing that things aren’t going to improve with time, acting now to strengthen their voices sounds appealing.
With the vote for unionization behind them, the organizers are now ready to formulate a prioritized list of demands. Those of you who are regular readers of Letters from Maine know that I have been urging primary care physicians to find their voices. Unfortunately, unionization seems to be becoming a more common fall-back strategy when other avenues have failed to reach a sympathetic ear in the corporate boardrooms.
As more unions form, it will be interesting to see how the organizers structure their demands and job actions. While walkouts and strikes can certainly be effective in gaining attention, that attention can carry a risk of counter productivity sometimes by alienating patients, who should become allies.
Since an unsustainable burden of paperwork and administrative demands seems to be at the top of everyone’s priority list, it might make sense to adopt this message as a scaffolding on which to built a work action. Instead of walking off the job or marching on a picket line, why not stay in the hospital and continue to see patients but only for part of the work day. The remainder of the day would be spent doing all the clerical work that has become so onerous.
Providers would agree to see patients in the mornings, saving up the clerical work and administrative obligations for the afternoon. The definition of “morning” could vary depending on local conditions.
The important message to the public and the patients would be that the providers were not abandoning them by walking out. The patients’ access to face-to-face care was being limited not because the doctors didn’t want to see them but because the providers were being forced to accept other responsibilities by the administration. The physicians would always be on site in case of a crisis, but until reasonable demands for support from the company were met, a certain portion of the providers’ day would be spent doing things not directly related to face-to-face patient care. This burden of meaningless work is the reality as it stands already. Why not organize it in a way that makes it startlingly visible to the patients and the public.
There would be no video clips of physicians walking the picket lines carrying signs. Any images released to the media would be of empty waiting rooms while providers sat hunched over their computers or talking on the phone to insurance companies.
The strategy needs a catchy phrase like “a paperwork-in” but I’m still struggling with a name. Let me know if you have a better one or even a better strategy.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Organized labor seems to be experiencing a rebirth of sorts. In October 2022 a strike by railroad workers was averted when a tentative agreement about wages, working conditions, health insurance, and medical leave was hammered out. This past fall, strikes by auto workers that threatened to paralyze the big three manufacturers have now been resolved with agreements that meet many of the workers’ demands. The President even made an appearance on a picket line. Baristas at coffee shops, screenwriters, and actors have all been involved in work actions around the country.
While the health care industry has been relatively immune to threatened work stoppages, there are a growing number of hospitals and clinics where nurses and physicians are exploring the possibility of organizing to give themselves a stronger voice in how health care is being delivered. The realities that come when you transition from owner to employee are finally beginning to sink in for physicians, whether they are specialists or primary care providers.
One of the most significant efforts toward unionization recently occurred in Minnesota and Wisconsin. About 400 physicians and 150 physician’s assistants and nurse practitioners employed at Allina Health System voted to unionize and join the Doctors Council.
In an interview with Jacobin, a publication that offers a socialist perspective, three of the providers involved in the process that led to the vote shared their observations. The physicians claim that the first steps toward unionization came after multiple efforts to work with the Allina’s administration were rebuffed. As primary care physicians, their initial demands focused on getting help with hiring staffing and getting support with paperwork and administrative obligations.
The organizers complained that while Medicare hoped to bolster primary care by paying the providers more, the funds went to the companies, who then distributed them in a way that often did little to help the overworked providers. In addition to achieving a more equitable distribution of the monies, one of the organizers sees unionization as a way to provide a layer of protection when providers feel they must speak out about situations which clearly put quality of care at risk.
The organizers say the idea of unionization has been particularly appealing to the younger providers who are feeling threatened by burnout. When these new physicians look to their older coworkers for advice, they often find that the seasoned employees are as stressed as they are. Realizing that things aren’t going to improve with time, acting now to strengthen their voices sounds appealing.
With the vote for unionization behind them, the organizers are now ready to formulate a prioritized list of demands. Those of you who are regular readers of Letters from Maine know that I have been urging primary care physicians to find their voices. Unfortunately, unionization seems to be becoming a more common fall-back strategy when other avenues have failed to reach a sympathetic ear in the corporate boardrooms.
As more unions form, it will be interesting to see how the organizers structure their demands and job actions. While walkouts and strikes can certainly be effective in gaining attention, that attention can carry a risk of counter productivity sometimes by alienating patients, who should become allies.
Since an unsustainable burden of paperwork and administrative demands seems to be at the top of everyone’s priority list, it might make sense to adopt this message as a scaffolding on which to built a work action. Instead of walking off the job or marching on a picket line, why not stay in the hospital and continue to see patients but only for part of the work day. The remainder of the day would be spent doing all the clerical work that has become so onerous.
Providers would agree to see patients in the mornings, saving up the clerical work and administrative obligations for the afternoon. The definition of “morning” could vary depending on local conditions.
The important message to the public and the patients would be that the providers were not abandoning them by walking out. The patients’ access to face-to-face care was being limited not because the doctors didn’t want to see them but because the providers were being forced to accept other responsibilities by the administration. The physicians would always be on site in case of a crisis, but until reasonable demands for support from the company were met, a certain portion of the providers’ day would be spent doing things not directly related to face-to-face patient care. This burden of meaningless work is the reality as it stands already. Why not organize it in a way that makes it startlingly visible to the patients and the public.
There would be no video clips of physicians walking the picket lines carrying signs. Any images released to the media would be of empty waiting rooms while providers sat hunched over their computers or talking on the phone to insurance companies.
The strategy needs a catchy phrase like “a paperwork-in” but I’m still struggling with a name. Let me know if you have a better one or even a better strategy.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
Organized labor seems to be experiencing a rebirth of sorts. In October 2022 a strike by railroad workers was averted when a tentative agreement about wages, working conditions, health insurance, and medical leave was hammered out. This past fall, strikes by auto workers that threatened to paralyze the big three manufacturers have now been resolved with agreements that meet many of the workers’ demands. The President even made an appearance on a picket line. Baristas at coffee shops, screenwriters, and actors have all been involved in work actions around the country.
While the health care industry has been relatively immune to threatened work stoppages, there are a growing number of hospitals and clinics where nurses and physicians are exploring the possibility of organizing to give themselves a stronger voice in how health care is being delivered. The realities that come when you transition from owner to employee are finally beginning to sink in for physicians, whether they are specialists or primary care providers.
One of the most significant efforts toward unionization recently occurred in Minnesota and Wisconsin. About 400 physicians and 150 physician’s assistants and nurse practitioners employed at Allina Health System voted to unionize and join the Doctors Council.
In an interview with Jacobin, a publication that offers a socialist perspective, three of the providers involved in the process that led to the vote shared their observations. The physicians claim that the first steps toward unionization came after multiple efforts to work with the Allina’s administration were rebuffed. As primary care physicians, their initial demands focused on getting help with hiring staffing and getting support with paperwork and administrative obligations.
The organizers complained that while Medicare hoped to bolster primary care by paying the providers more, the funds went to the companies, who then distributed them in a way that often did little to help the overworked providers. In addition to achieving a more equitable distribution of the monies, one of the organizers sees unionization as a way to provide a layer of protection when providers feel they must speak out about situations which clearly put quality of care at risk.
The organizers say the idea of unionization has been particularly appealing to the younger providers who are feeling threatened by burnout. When these new physicians look to their older coworkers for advice, they often find that the seasoned employees are as stressed as they are. Realizing that things aren’t going to improve with time, acting now to strengthen their voices sounds appealing.
With the vote for unionization behind them, the organizers are now ready to formulate a prioritized list of demands. Those of you who are regular readers of Letters from Maine know that I have been urging primary care physicians to find their voices. Unfortunately, unionization seems to be becoming a more common fall-back strategy when other avenues have failed to reach a sympathetic ear in the corporate boardrooms.
As more unions form, it will be interesting to see how the organizers structure their demands and job actions. While walkouts and strikes can certainly be effective in gaining attention, that attention can carry a risk of counter productivity sometimes by alienating patients, who should become allies.
Since an unsustainable burden of paperwork and administrative demands seems to be at the top of everyone’s priority list, it might make sense to adopt this message as a scaffolding on which to built a work action. Instead of walking off the job or marching on a picket line, why not stay in the hospital and continue to see patients but only for part of the work day. The remainder of the day would be spent doing all the clerical work that has become so onerous.
Providers would agree to see patients in the mornings, saving up the clerical work and administrative obligations for the afternoon. The definition of “morning” could vary depending on local conditions.
The important message to the public and the patients would be that the providers were not abandoning them by walking out. The patients’ access to face-to-face care was being limited not because the doctors didn’t want to see them but because the providers were being forced to accept other responsibilities by the administration. The physicians would always be on site in case of a crisis, but until reasonable demands for support from the company were met, a certain portion of the providers’ day would be spent doing things not directly related to face-to-face patient care. This burden of meaningless work is the reality as it stands already. Why not organize it in a way that makes it startlingly visible to the patients and the public.
There would be no video clips of physicians walking the picket lines carrying signs. Any images released to the media would be of empty waiting rooms while providers sat hunched over their computers or talking on the phone to insurance companies.
The strategy needs a catchy phrase like “a paperwork-in” but I’m still struggling with a name. Let me know if you have a better one or even a better strategy.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine, for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected].
FDA mandates five changes to iPLEDGE program for isotretinoin
In a letter dated Nov. 30, 2023, the .
The development follows a March 2023 joint meeting of the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee about iPLEDGE REMS requirements, which included feedback from patients and dermatologists and recommendations for changes to the REMS program, aimed at minimizing the burden of the program on patients, pharmacies, and prescribers while continuing to maintain safe use of the highly teratogenic drug for patients.
The five changes include the following:
- Remove the requirement that pregnancy tests must be performed in a specially certified (i.e., Clinical Laboratory Improvement Amendments [CLIA]) laboratory. In the opinion of John S. Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, this change “may make it easier to perform pregnancy tests in a clinic setting without needing to send the patient to a separate lab,” he said in an interview.
- Allow prescribers the option of using home pregnancy testing for their patients during and after isotretinoin treatment. Prescribers who rely on the patient to perform a home pregnancy test need to take steps to minimize patients falsifying the results of these tests. According to Dr. Barbieri, this means that two pregnancy tests prior to starting isotretinoin must be done in a lab or office setting. “However, all the pregnancy tests on therapy can be either in a medical setting or using a home pregnancy test,” he told this news organization. “This option facilitates the use of telemedicine so that patients would not need to come in; they can just share a pregnancy test with their name and date with their dermatologist.”
- Remove the waiting period requirement — also known as the “19-day lockout” — for patients if they do not obtain isotretinoin within the first 7-day prescription window. According to Dr. Barbieri, this change helps to ensure that patients can begin isotretinoin in a timely manner. “Insurance and pharmacy delays that are no fault of the patient can commonly cause missed initial window periods,” he said. “Allowing for immediate repeat of a pregnancy test to start a new window period, rather than requiring the patient to wait 19 more days, can ensure patient safety and pregnancy prevention without negatively impacting access.”
- Revise the pregnancy registry requirement to remove the objective to document the pregnancy and fetal outcomes for each pregnancy.
- Revise the requirement for prescribers to document patient counseling in patients who cannot become pregnant from monthly to only at enrollment. Dr. Barbieri characterized this change as “major” and said that it could eliminate the need for monthly visits for persons of non–childbearing potential. “This could substantially reduce logistical burdens for patients and reduce wait times to see a dermatologist,” he said.
Future changes to iPLEDGE that Dr. Barbieri would like to see include allowing for home pregnancy tests prior to starting therapy — particularly the test after the 30-day window period. “In addition, it would be good to be able to reduce the 30-day waiting period prior to therapy to something shorter,” such as 14 days, which would still “reliably exclude pregnancy, particularly for those on stable long-acting reversible contraception,” he said. There are also opportunities to improve the iPLEDGE website functionality and to ensure that the website is accessible to patients with limited English proficiency, he added.
He also recommended greater transparency by the Isotretinoin Products Manufacturers Group and inclusion of input from diverse stakeholders such as dermatologists, patients, and pharmacists.
Dr. Barbieri reported personal fees from Dexcel Pharma.
In a letter dated Nov. 30, 2023, the .
The development follows a March 2023 joint meeting of the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee about iPLEDGE REMS requirements, which included feedback from patients and dermatologists and recommendations for changes to the REMS program, aimed at minimizing the burden of the program on patients, pharmacies, and prescribers while continuing to maintain safe use of the highly teratogenic drug for patients.
The five changes include the following:
- Remove the requirement that pregnancy tests must be performed in a specially certified (i.e., Clinical Laboratory Improvement Amendments [CLIA]) laboratory. In the opinion of John S. Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, this change “may make it easier to perform pregnancy tests in a clinic setting without needing to send the patient to a separate lab,” he said in an interview.
- Allow prescribers the option of using home pregnancy testing for their patients during and after isotretinoin treatment. Prescribers who rely on the patient to perform a home pregnancy test need to take steps to minimize patients falsifying the results of these tests. According to Dr. Barbieri, this means that two pregnancy tests prior to starting isotretinoin must be done in a lab or office setting. “However, all the pregnancy tests on therapy can be either in a medical setting or using a home pregnancy test,” he told this news organization. “This option facilitates the use of telemedicine so that patients would not need to come in; they can just share a pregnancy test with their name and date with their dermatologist.”
- Remove the waiting period requirement — also known as the “19-day lockout” — for patients if they do not obtain isotretinoin within the first 7-day prescription window. According to Dr. Barbieri, this change helps to ensure that patients can begin isotretinoin in a timely manner. “Insurance and pharmacy delays that are no fault of the patient can commonly cause missed initial window periods,” he said. “Allowing for immediate repeat of a pregnancy test to start a new window period, rather than requiring the patient to wait 19 more days, can ensure patient safety and pregnancy prevention without negatively impacting access.”
- Revise the pregnancy registry requirement to remove the objective to document the pregnancy and fetal outcomes for each pregnancy.
- Revise the requirement for prescribers to document patient counseling in patients who cannot become pregnant from monthly to only at enrollment. Dr. Barbieri characterized this change as “major” and said that it could eliminate the need for monthly visits for persons of non–childbearing potential. “This could substantially reduce logistical burdens for patients and reduce wait times to see a dermatologist,” he said.
Future changes to iPLEDGE that Dr. Barbieri would like to see include allowing for home pregnancy tests prior to starting therapy — particularly the test after the 30-day window period. “In addition, it would be good to be able to reduce the 30-day waiting period prior to therapy to something shorter,” such as 14 days, which would still “reliably exclude pregnancy, particularly for those on stable long-acting reversible contraception,” he said. There are also opportunities to improve the iPLEDGE website functionality and to ensure that the website is accessible to patients with limited English proficiency, he added.
He also recommended greater transparency by the Isotretinoin Products Manufacturers Group and inclusion of input from diverse stakeholders such as dermatologists, patients, and pharmacists.
Dr. Barbieri reported personal fees from Dexcel Pharma.
In a letter dated Nov. 30, 2023, the .
The development follows a March 2023 joint meeting of the FDA’s Drug Safety and Risk Management Advisory Committee and the Dermatologic and Ophthalmic Drugs Advisory Committee about iPLEDGE REMS requirements, which included feedback from patients and dermatologists and recommendations for changes to the REMS program, aimed at minimizing the burden of the program on patients, pharmacies, and prescribers while continuing to maintain safe use of the highly teratogenic drug for patients.
The five changes include the following:
- Remove the requirement that pregnancy tests must be performed in a specially certified (i.e., Clinical Laboratory Improvement Amendments [CLIA]) laboratory. In the opinion of John S. Barbieri, MD, MBA, director of the Advanced Acne Therapeutics Clinic at Brigham and Women’s Hospital, Boston, this change “may make it easier to perform pregnancy tests in a clinic setting without needing to send the patient to a separate lab,” he said in an interview.
- Allow prescribers the option of using home pregnancy testing for their patients during and after isotretinoin treatment. Prescribers who rely on the patient to perform a home pregnancy test need to take steps to minimize patients falsifying the results of these tests. According to Dr. Barbieri, this means that two pregnancy tests prior to starting isotretinoin must be done in a lab or office setting. “However, all the pregnancy tests on therapy can be either in a medical setting or using a home pregnancy test,” he told this news organization. “This option facilitates the use of telemedicine so that patients would not need to come in; they can just share a pregnancy test with their name and date with their dermatologist.”
- Remove the waiting period requirement — also known as the “19-day lockout” — for patients if they do not obtain isotretinoin within the first 7-day prescription window. According to Dr. Barbieri, this change helps to ensure that patients can begin isotretinoin in a timely manner. “Insurance and pharmacy delays that are no fault of the patient can commonly cause missed initial window periods,” he said. “Allowing for immediate repeat of a pregnancy test to start a new window period, rather than requiring the patient to wait 19 more days, can ensure patient safety and pregnancy prevention without negatively impacting access.”
- Revise the pregnancy registry requirement to remove the objective to document the pregnancy and fetal outcomes for each pregnancy.
- Revise the requirement for prescribers to document patient counseling in patients who cannot become pregnant from monthly to only at enrollment. Dr. Barbieri characterized this change as “major” and said that it could eliminate the need for monthly visits for persons of non–childbearing potential. “This could substantially reduce logistical burdens for patients and reduce wait times to see a dermatologist,” he said.
Future changes to iPLEDGE that Dr. Barbieri would like to see include allowing for home pregnancy tests prior to starting therapy — particularly the test after the 30-day window period. “In addition, it would be good to be able to reduce the 30-day waiting period prior to therapy to something shorter,” such as 14 days, which would still “reliably exclude pregnancy, particularly for those on stable long-acting reversible contraception,” he said. There are also opportunities to improve the iPLEDGE website functionality and to ensure that the website is accessible to patients with limited English proficiency, he added.
He also recommended greater transparency by the Isotretinoin Products Manufacturers Group and inclusion of input from diverse stakeholders such as dermatologists, patients, and pharmacists.
Dr. Barbieri reported personal fees from Dexcel Pharma.
Are you sure your patient is alive?
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.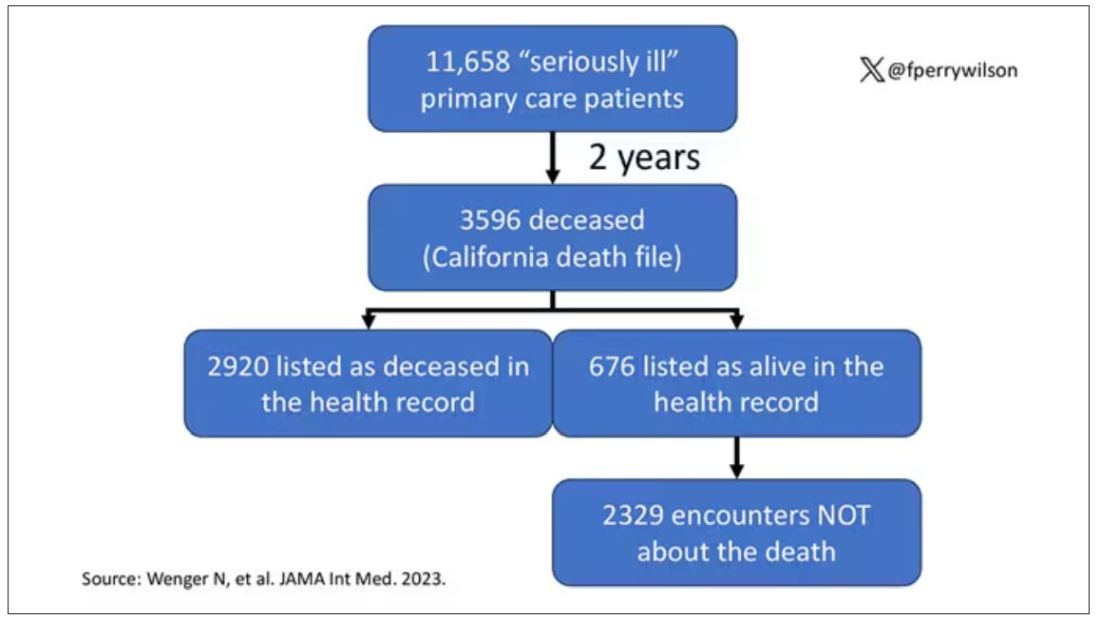
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .
This transcript has been edited for clarity.
Much of my research focuses on what is known as clinical decision support — prompts and messages to providers to help them make good decisions for their patients. I know that these things can be annoying, which is exactly why I study them — to figure out which ones actually help.
When I got started on this about 10 years ago, we were learning a lot about how best to message providers about their patients. My team had developed a simple alert for acute kidney injury (AKI). We knew that providers often missed the diagnosis, so maybe letting them know would improve patient outcomes.
As we tested the alert, we got feedback, and I have kept an email from an ICU doctor from those early days. It read:
Dear Dr. Wilson: Thank you for the automated alert informing me that my patient had AKI. Regrettably, the alert fired about an hour after the patient had died. I feel that the information is less than actionable at this time.
Our early system had neglected to add a conditional flag ensuring that the patient was still alive at the time it sent the alert message. A small oversight, but one that had very large implications. Future studies would show that “false positive” alerts like this seriously degrade physician confidence in the system. And why wouldn’t they?
Not knowing the vital status of a patient can have major consequences.
Health systems send messages to their patients all the time: reminders of appointments, reminders for preventive care, reminders for vaccinations, and so on.
But what if the patient being reminded has died? It’s a waste of resources, of course, but more than that, it can be painful for their families and reflects poorly on the health care system. Of all the people who should know whether someone is alive or dead, shouldn’t their doctor be at the top of the list?
A new study in JAMA Internal Medicine quantifies this very phenomenon.
Researchers examined 11,658 primary care patients in their health system who met the criteria of being “seriously ill” and followed them for 2 years. During that period of time, 25% were recorded as deceased in the electronic health record. But 30.8% had died. That left 676 patients who had died, but were not known to have died, left in the system.
And those 676 were not left to rest in peace. They received 221 telephone and 338 health portal messages not related to death, and 920 letters reminding them about unmet primary care metrics like flu shots and cancer screening. Orders were entered into the health record for things like vaccines and routine screenings for 158 patients, and 310 future appointments — destined to be no-shows — were still on the books. One can only imagine the frustration of families checking their mail and finding yet another letter reminding their deceased loved one to get a mammogram.
How did the researchers figure out who had died? It turns out it’s not that hard. California keeps a record of all deaths in the state; they simply had to search it. Like all state death records, they tend to lag a bit so it’s not clinically terribly useful, but it works. California and most other states also have a very accurate and up-to-date death file which can only be used by law enforcement to investigate criminal activity and fraud; health care is left in the lurch.
Nationwide, there is the real-time fact of death service, supported by the National Association for Public Health Statistics and Information Systems. This allows employers to verify, in real time, whether the person applying for a job is alive. Healthcare systems are not allowed to use it.
Let’s also remember that very few people die in this country without some health care agency knowing about it and recording it. But sharing of medical information is so poor in the United States that your patient could die in a hospital one city away from you and you might not find out until you’re calling them to see why they missed a scheduled follow-up appointment.
These events — the embarrassing lack of knowledge about the very vital status of our patients — highlight a huge problem with health care in our country. The fragmented health care system is terrible at data sharing, in part because of poor protocols, in part because of unfounded concerns about patient privacy, and in part because of a tendency to hoard data that might be valuable in the future. It has to stop. We need to know how our patients are doing even when they are not sitting in front of us. When it comes to life and death, the knowledge is out there; we just can’t access it. Seems like a pretty easy fix.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Connecticut. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com .





