User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'main-prefix')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
The psychiatrist’s role in navigating a toxic news cycle
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m Dr. Gregory Scott Brown, an Austin (Tex.)-based psychiatrist and an affiliate faculty member at the University of Texas Dell Medical School.
Recently, a patient of mine told me that, because of the political environment we find ourselves in, he’s avoiding conversations with some of his longtime friends. Because of this, he’s feeling even more isolated than before.
We’re all coming off the heels of a tough year, and many of us expected that when we entered 2021 we’d quickly turn the page and life would get a whole lot easier. Since the reality is that we’re still dealing with deep divisions, social injustices, and the politicization of evidence-based medicine, emotions are naturally running high.
In listening to my patients over the past few weeks, there’s definitely a sense of optimism about coronavirus vaccines and getting back to life as usual. But there’s also a lingering sense of uncertainty and fear, especially when it comes to the possibility of saying the wrong thing, offending our friends, or just having conversations with people we may disagree with. I’m hearing concerns from my patients that 2020 exposed a dormant hatred that was brewing in the underbelly of our society, in our politics, and in our institutions. Patients are telling me that these concerns are making them anxious and some are avoiding interacting with people they may disagree with altogether because they’re afraid of the difficult conversations that may follow.
Since, like many of my patients as well as many of you, I follow the news, including stories of COVID-related deaths, economic hardships, peaceful protests gone bad, and political vitriol, I’ve had to remind myself about the importance of intentional kindness for effective communication and for supporting mental health.
I was reminded of the paper “Hate in the Counter-Transference” by the well-known pediatrician and psychoanalyst Donald Winnicott. He focuses on how to manage and sort through the strong emotions that may be experienced even during an encounter between a therapist and a patient. Although some of what he has to say doesn’t translate well to modern times, his recommendation that we acknowledge and try to normalize some of our feelings does. And this is how I’ve been starting a conversation with my patients – just by normalizing things a bit.
The past year or so has brought with it a range of intense emotions, including frustration, exhaustion, and some degree of sadness for most of us. When we’re self-aware about our feelings, we can make sense of them early on so that they don’t evolve into maladaptive ones like unhinged anger or hatred. My patients and I actively discuss how our feelings don’t have to get in the way of our ability to live and to interact with each other as we’d like to.
Considering the basic tenets of kindness is a good place to start. I recently spoke with Kelli Harding, MD, a psychiatrist and author of The Rabbit Effect: Live Longer, Happier, and Healthier with the Groundbreaking Science of Kindness. She pointed to research suggesting that kindness can benefit multiple areas of our health, from reducing cardiovascular events to improving mood and anxiety. In her book, she notes that
I tell my patients that we can’t always change our environment, but we can definitely change how we respond to it. This doesn’t mean it’s always an easy process, but there are things we can do. First, we have to acknowledge that some degree of conflict or cordial disagreement is inevitable and it doesn’t have to disrupt our mood.
An interesting study on conflict management pointed out that healthy conflict is actually beneficial in some cases. For instance, in the work environment, conflict can help with team development and better group decision-making. But it’s rigid personality differences, poor communication, emotional stress, and lack of candor that may contribute to so-called high-tension events, and this is where conflict can go awry. These are the areas that we can all focus on improving, not only for performance benefits but for our overall health and well-being as well.
The authors also recommend an active style of engagement as a technique to manage conflict, but in a way that feels both natural and safe. Other authors agree that so-called engaged coping is associated with a higher sense of control and overall improved psychological well-being. What this means is that avoidance may be necessary in the short term, but over time it may lead to more emotional stress and anxiety.
Overcoming the tendency to avoid requires both motivation and self-awareness. We need to know about patterns in our own behavior and how the behavior of others can push our buttons and spiral a healthy disagreement into a heated argument.
I like to recommend the hunger, angry, lonely, tired (HALT) model, which is often used as a self-care gauge in addiction treatment and to reduce medication errors. But I think it’s also a useful way to assess personal readiness for having a difficult conversation. I tell my patients to ask themselves in this moment: “Am I hungry, angry, lonely, or tired?” And if they are, perhaps it’s not the best time for the conversation.
Because 2020 brought with it a new set of challenges, it also forced many of us to focus on things that we just didn’t pay as much attention to before, including checking in on our feelings and the feelings of those around us. It also taught us to pay attention to the way and manner in which we communicate. I think that kindness is much easier to carry into difficult conversations if we approach them with a sense of curiosity before judgment. Ultimately, kindness is one of the best tools for balancing the intense emotions that many of us are feeling right now.
A version of this article first appeared on Medscape.com.
‘Landmark’ schizophrenia drug in the wings?
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A novel therapy that combines a muscarinic receptor agonist with an anticholinergic agent is associated with a greater reduction in psychosis symptoms, compared with placebo, new research shows.
In a randomized, phase 2 trial composed of nearly 200 participants, xanomeline-trospium (KarXT) was generally well tolerated and had none of the common side effects linked to current antipsychotics, including weight gain and extrapyramidal symptoms such as dystonia, parkinsonism, and tardive dyskinesia.
“The results showing robust therapeutic efficacy of a non–dopamine targeting antipsychotic drug is an important milestone in the advance of the therapeutics of schizophrenia and other psychotic disorders,” coinvestigator Jeffrey A. Lieberman, MD, professor and chairman in the department of psychiatry, Columbia University, New York, said in an interview.
If approved, the new agent will be a “landmark” drug, Dr. Lieberman added.
The study was published in the Feb. 25, 2021, issue of the New England Journal of Medicine.
Long journey
The journey to develop an effective schizophrenia drug that reduces psychosis symptoms without onerous side effects has been a long one full of excitement and disappointment.
First-generation antipsychotics, dating back to the 1950s, targeted the postsynaptic dopamine-2 (D2) receptor. At the time, it was a “breakthrough” similar in scope to insulin for diabetes or antibiotics for infections, said Dr. Lieberman.
That was followed by development of numerous “me too” drugs with the same mechanism of action. However, these drugs had significant side effects, especially neurologic adverse events such as parkinsonism.
In 1989, second-generation antipsychotics were introduced, beginning with clozapine. They still targeted the D2 receptor but were “kinder and gentler,” Dr. Lieberman noted. “They didn’t bind to [the receptor] with such affinity that it shut things down completely, so had fewer neurologic side effects.”
However, these agents had other adverse consequences, such as weight gain and other metabolic effects including hyperglycemia and hyperlipidemia.
Many have poor functional status and quality of life despite lifelong treatment with current antipsychotic agents.
“The pharmaceutical industry, biotech industry, and academic psychiatric community have been desperately trying to find novel strategies for antipsychotic drug development and asking, ‘Is D2 the only holy grail or are there other ways of treating psychotic symptoms of schizophrenia?’ ” Dr. Lieberman said.
Enter KarXT – a novel combination of xanomeline with trospium.
An ‘ingenious’ combination
Xanomeline, an oral muscarinic cholinergic receptor agonist, does not have direct effects on the dopamine receptor. Evidence suggests the muscarinic cholinergic system is involved in the pathophysiology of schizophrenia.
However, there may be dose-dependent adverse events with the medication, such as nausea, vomiting, diarrhea, sweating, and hypersalivation from stimulation of peripheral muscarinic cholinergic receptors.
That’s where trospium chloride, an oral panmuscarinic receptor antagonist approved for treating overactive bladder, comes in. It does not reach detectable levels in the cerebrospinal fluid and should avoid adverse central nervous system effects.
Dr. Lieberman said the idea of the drug combination is “ingenious.”
The new phase 2, multisite study included adult patients with a validated diagnosis of schizophrenia who were hospitalized with an acute exacerbation of psychosis, and who were free of antipsychotic medication for at least 2 weeks.
Participants were required to have a baseline Positive and Negative Syndrome Scale (PANSS) total score of 80 points or more.
In addition to seven positive symptom items, including delusions, hallucinations, and conceptual disorganization, the PANSS has seven negative symptom items. These include restricted emotional expression, paucity of speech, and diminished interest, social drive, and activity. Each item is scored from 1 to 7, with higher scores indicating more severe symptoms.
Patients also had to have a score on the Clinical Global Impression–Severity (CGI-S) scale of 4 or higher. Scores on the CGI-S range from 1 to 7, with higher scores indicating greater severity of illness.
The modified intention-to-treat analysis included patients randomly assigned to receive oral xanomeline-trospium (n = 83) or placebo (n = 87).
The dosing schedule was flexible, starting with 50 mg of xanomeline and 20 mg of trospium twice daily. The schedule increased to a maximum of 125 mg of xanomeline and 30 mg of trospium twice daily, with the option of lowering the dose if there were unacceptable side effects.
Mean scores at baseline for the treatment and placebo groups were 97.7 versus 96.6 for the PANSS total score, 26.4 versus 26.3 for the positive subscore, 22.6 versus 22.8 for the negative subscore, and 5.0 versus 4.9 in the CGI-S scale.
‘Impressively robust’ effect size
The primary endpoint was change in the PANSS total score at 5 weeks. Results showed a change of –17.4 points in the treatment group and –5.9 points in the placebo group (least-squares mean difference, –11.6 points; 95% confidence interval, –16.1 to –7.1; P < .001).
The effect size, which was almost 0.8 (0.75), was “impressively robust,” said Dr. Lieberman, adding that a moderate effect size in this patient population might be in the order of 0.4 or 0.5.
“That gives hope that this drug may not just be as effective as other antipsychotics, albeit acting in a novel way and in a way that has a less of side effect burden, but that it may actually have some elements of superior efficacy,” he said.
There were significant benefits on some secondary outcomes, including change in the PANSS positive symptom subscore (–5.6 points in the treatment group vs. –2.4 points in the placebo group; least-squares mean difference, –3.2 points; 95% CI, –4.8 to –1.7; P < .001).
The active treatment also came out on top for CGI-S scores (P < .001), and PANSS negative symptom subscore (P < .001).
Because participants were hospitalized with an acute exacerbation of positive symptoms at time of study, it is difficult to determine “definitive efficacy” for negative symptoms, Dr. Lieberman noted. Negative symptoms may have improved simply because positive symptoms got better, he said.
Although the study included adults only, “there is nothing in the KarXT clinical profile that suggests it would be problematic for younger people,” Dr. Lieberman noted. This could include teenagers with first-episode psychosis.
Safety profile
Adverse events (AEs) were reported in 54% of the treatment group and 43% of the placebo group. AEs that were more common in the active treatment group included constipation (17% vs. 3%), nausea (17% vs. 4%), dry mouth (9% vs. 1%), dyspepsia (9% vs. 4%), and vomiting (9% vs. 4%). All AEs were rated as mild or moderate in severity and none resulted in discontinuation of treatment.
Rates of nausea, vomiting, and dry mouth were highest early in the trial and lower at the end, whereas constipation remained constant throughout the study.
Persistent constipation could affect the drug’s “utility” in elderly patients with cognitive issues but may be more of a “minor nuisance,” compared with other antipsychotics for those with schizophrenia, said Dr. Lieberman. He added that constipation might be mitigated with an over-the-counter treatment such as Metamucil. Importantly, there was no difference between groups in extrapyramidal symptoms.
In addition, participants receiving the active treatment did not have greater weight gain, which was about 3% versus 4% in the placebo group. The mean change in weight was 1.5 kg (3.3 lb) and 1.1 kg (2.4 lb), respectively.
Dr. Lieberman praised the manufacturer for undertaking the study.
“In an era when Big Pharma has retreated to a considerable degree from psychotropic drug development, it’s commendable that some companies have stayed the course and are succeeding in drug development,” he said.
Exciting mechanism
Commenting on the findings in an interview, Thomas Sedlak, MD, PhD, director of the Schizophrenia and Psychosis Consult Clinic and assistant professor of psychiatry and behavioral sciences at Johns Hopkins University, Baltimore, called some aspects of the study “exciting.”
What’s especially important is that the novel agent “acts by a different mechanism than what we have been using for maybe 60 years,” said Dr. Sedlak, who was not involved with the research.
“There has been a lot of research in the last few decades into drugs that might act by alternate mechanisms of action, but nothing’s really gotten to market for schizophrenia,” he noted.
Another interesting aspect of the study is that it examined a combination drug that aims to maximize effects on the brain while minimizing periphery effects, Dr. Sedlak said.
In addition, he called the PANSS results “respectable.”
“It’s interesting that you get a big change in PANSS total score, but it’s not as big in the positive symptom score as you might have expected, and it’s not a huge drop in negative symptoms either,” said Dr. Sedlak.
It’s possible, he said, that the drug is targeting PANSS elements not captured in the main positive or negative symptom items; for example, anxiety, depression, guilt, attention, impulse control, disorientation, and judgment.
Dr. Sedlak noted that, while the new results are exciting, the enthusiasm may not be long-lasting. “When a new drug comes out, there’s often a lot of excitement about it, but once you start using it, expectations may deflate..
“More flexible ratios” of the two components of the drug might be useful to treat individual patients, he added.
In addition, using the components by themselves might be preferable for some clinicians. “I think a lot of physicians prefer using two separate drugs so they can alter the dose of each one independently and not be stuck with the ratio the manufacturer is providing,” Dr. Sedlak said.
However, he acknowledged that studying different choices of ratios would require much larger trials.
The study was supported by Karuna Therapeutics and the Wellcome Trust. Dr. Lieberman reported serving on an advisory board for Karuna Therapeutics and Intra-Cellular Therapies. Dr. Sedlak disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Severe atopic dermatitis often puts a dent in quality of life
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
In his role as head of the division of pediatric behavioral health at National Jewish Health, Denver, Bruce G. Bender, PhD, helps children and adults navigate the adverse effects of severe atopic dermatitis (AD) on their quality of life.
“There have been many surveys of adults with AD who report impairment of their sleep, reduced activity level, increased work absence, financial burden, emotional distress, and social avoidance,” he said at the Revolutionizing Atopic Dermatitis virtual symposium. “Similarly, children with AD or their parents report emotional distress, reduced activity, and increased school absence, social avoidance, and sleep disturbance. Families report financial burdens, conflict, particularly among the adults, social avoidance, sleep disturbance in the parents, and reduction of well-being in the siblings.”
In an effort to objectively measure sleep change in this population, Dr. Bender and colleagues recruited 14 adults with AD and 14 healthy controls who wore an ActiGraph for 1 week and completed questionnaires about sleep, itch, and quality of life. Patients with AD were awake almost twice as many minutes each night as the healthy controls (a mean of 57.3 vs. 32.3 minutes, respectively; P = .0480). Consequently, their sleep efficiency was significantly reduced based on the Pittsburgh sleep quality index (a mean of 90.6 vs. 95; P = .0305).
In another study, Dr. Bender and colleagues enrolled 20 adults with AD who underwent 2 nights of polysomnography and actigraphy. The lab was set up to measure a scratching event, which was recorded when a burst of electromyographic activity of at least 3 seconds was accompanied by a visible scratching motion. “We learned that sleep efficiency as measured by both PSG and actigraphy correlated with total body surface area and scratching index,” he said. “As we might assume, the more skin involved, the more patients scratch, the less well they sleep.”
Behavioral, neurocognitive effects
In a separate study of AD, sleep, and behavior, the researchers studied 1,041 children with asthma who were enrolled in the Childhood Asthma Management Program at eight North American sites. They used baseline parent ratings on standardized sleep and behavior rating scales and found that increased awakenings were associated with increased school absence and daytime behavior problems. “So, not only do children with AD sleep less well, but this shows up to impair their functioning during the day,” said Dr. Bender, professor of psychiatry at the University of Colorado, Denver.
In a report from Australia, researchers set out to explore the association between sleep and neurocognitive function in 21 children with eczema and 20 healthy controls. Participants underwent cognitive testing and polysomnography. The authors found that the children with eczema demonstrated lower test scores. Reduced scores were correlated with parental reports of sleep problems but not polysomnography.
In a much larger study funded by the Agency for Healthcare Research and Quality, investigators analyzed data on 354,416 children and 34,613 adults from 19 U.S. population surveys including the National Health Interview Survey 1997-2013 and the National Survey of Children’s Health 2003/4 and 2007/8. They found that AD was associated with ADHD in children (adjusted odds ratio, 1.14) and adults (aOR, 1.61). Higher odds of ADHD were found in children who had significant sleep disturbance (aOR, 16.83) and other allergic disease and asthma (aOR, 1.61).
“All of these findings show that AD can impact quality of life, especially sleep, with the result of poorer daytime functioning,” Dr. Bender said. “But those studies don’t answer this question: Are patients with AD at increased risk for psychological disorders such as depression and anxiety?”
Impact on depression, anxiety
Two systematic reviews on the topic suggest that patients with AD are twice as likely to experience depression. One was published in 2018 and the other in 2019. The 2018 review reported a little more than a twofold increase (OR, 2.19), the 2019 review a little bit less (OR, 1.71).
“At the more severe end of the depression continuum, we sometimes see suicidal ideation and suicide attempts,” Dr. Bender said. “A number of studies have asked whether these are increased in patients with AD. Quite a few studies collectively show an increased incidence of suicidal ideation. The question of suicide attempts is reflected in fewer studies. And while the result is small, it is significant. There is a significant increase reported of suicide attempts in AD patients.”
The 2018 review also found an increased incidence of anxiety in AD patients: a little more than twofold in adults (OR, 2.19) and a little less than twofold in children (OR, 1.81).
“It’s a two-way relationship between AD and psychological factors,” Dr. Bender said. “We generally think about AD – the stress that it brings, the burden that it puts on children, adults, and families. But it can work the other way around,” he said, referring to patients who have psychological problems, experience a great deal of stress, have trouble being adherent to their treatment regimen, and find it difficult to resist scratching. “The behavioral/psychological characteristics of the patient also drive the AD. It is well established that acute and chronic stress can result in a worsening of skin conditions in AD patients.”
Behavioral health interventions that have been described in the literature include cognitive therapy, stress management, biofeedback, hypnotherapy, relaxation training, mindfulness, habit reversal, and patient education – some of which have been tested in randomized trials. “All of them report a decrease in scratching as a consequence of the behavioral intervention,” Dr. Bender said.
“Other studies have been reported that look at the impact of behavioral interventions on the severity of the skin condition. Most report an improvement in the skin condition from these behavioral interventions but it’s not a perfect literature.” Critiques of these studies include the fact that there is often not enough detail about the intervention or the framework for the intervention that would allow a clinician to test an intervention in another study or actually pull that intervention into clinical practice (Cochrane Database Syst Rev. 2014 Jan 7;2014[1]:CD004054), (Int Arch Allergy Immunol.2007;144[1]:1-9).
“Some of the studies lack rigorous designs, some have sampling bias, and some have inadequate outcome measurements,” he said. “We really need additional, high-quality studies to look at what is helpful for patients with AD.”
Dr. Bender reported having no financial disclosures.
FROM REVOLUTIONIZING AD 2020
When should a patient’s violent thoughts trigger your action?
When patients relay their fantasies during psychotherapy sessions, those visions are often rooted in frustration or wish fulfillment, according to Jessica Ferranti, MD.
“[Sigmund] Freud talked about how our fantasy life is invested with large amounts of energy and interest and conveys a true essence of our personality – a truth about what we’re thinking and who we are,” Dr. Ferranti, a forensic psychiatrist in the division of psychiatry and the law at the University of California, Davis, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
“Fantasy life is one of the most important conveyances of information that we can get from our patients, whether in the general office or in the forensic realm – if we can access it, which is difficult, because fantasies are often intensely personal. They fall into the category of very high resistance topics with many patients.”
Psychiatrists routinely ask about violent thoughts and homicidal ideation, but violent fantasies – especially those that are sexually violent in nature – can be a warning sign of future danger. Dr. Ferranti defined violent fantasies as those depicting the use of physical force with the intent to injure another person or destroy property.
“This would be an individual who fantasizes about sadistically raping a woman, for instance,” said Dr. Ferranti, who directs the UC Davis Workplace Safety and Psychiatric Assessment Clinic. “That is an ominous and psychopathological sign in terms of the preoccupation with that kind of violent crime.”
Aggression, on the other hand, “is a very broad spectrum, with actions like assertion, interpersonal confrontation, or verbal expressions that are angry or hostile, but that do not necessarily lead to violence.”
Dr. Ferranti acknowledged that today’s rushed clinical environment makes it challenging for psychiatrists and psychologists to get patients to share detailed fantasies they may be harboring.
“It’s very difficult to get to deeper material with patients, unless potentially you have more intensive therapy going on, like a psychotherapeutic relationship where you see the patient frequently, an intensive treatment, [or] perhaps an inpatient hospitalization or a partial day program.” The key is that “the patient gets comfortable with relaying more of the truth about what they’re experiencing,” she said. “In some cases, this occurs during the forensic evaluation, because we have the luxury to do very lengthy evaluations. Under the stress of being with another person in the room for many hours, oftentimes the patient will disclose things eventually.
“I’ve been a forensic psychiatrist for the better part of 12 years, and I can tell you after hundreds of evaluations I’ve never had a person not speak. That’s a good thing, because a principle of the work we do, or talk therapy even, is that When we lose symbolism, the ability to represent things in our mind and speak about them, we are at greater risk of collapsing into the real and acting on the things we think about.”
Statutory reporting duties vary from state to state. In California, mandatory reporting duties include child abuse, elder abuse, abuse or neglect of developmentally disabled individuals, domestic violence, and victims of a gunshot wound. “Failing to report any of these crimes is a misdemeanor in California,” she said. “With all these statutory reporting duties, we have no legal obligation to inform the patient of the report. Under California law, patients do not have the right to refuse the report. These are reports we make in our best judgment, whether the patient is happy about that or not.”
What happens if your patient confesses to a past crime? “There’s no legal duty to report this,” Dr. Ferranti said. “The general rule is, unless there’s a current person who’s at risk, it would be violating confidentiality to report. This includes murder, bank robbery, and sexual assault. In addition, you cannot admit a patient to an inpatient setting to help them avoid arrest, even if you think the act in question was due to symptoms of a mental disorder, disease, or defect. You can actually be charged with aiding and abetting a criminal.”
In the 1976 landmark case Tarasoff v. the Regents of the University of California, the California Supreme Court ruled that psychiatrists and other therapists have a duty to do what is reasonably necessary to protect third parties if a patient presents a serious risk of violence to another person.
“Reasonable steps may include warning the third party, notifying police, detaining and hospitalizing the patient, intensifying the treatment to a higher level of care or more frequent outpatient appointments, removing weapons, and changing the medication therapy,” Dr. Ferranti said. “The more you can do of these, the better.”
She also discussed the concept of foreseeability, which she defined as the reasonable anticipation that harm or injury is likely to result from an act or omission to act.
“This is the malpractice standard for negligence,” she said. “In other words, was it foreseeable by a reasonable psychiatrist that this person was going to hurt someone else or themselves?” Another landmark case, Jablonski Pahls v. the United States broadened the reporting obligations of psychiatrists. In this 1983 case, the U.S. Court of Appeals 9th Circuit ruled that mental health professionals have to do more than warn foreseeable victims of an imminent danger of potential harm; they must involuntarily hospitalize the dangerous individual and consult that person’s prior records.
There is no sure-fire way to predict when an individual’s underlying violent fantasies are likely to be acted on, but Dr. Ferranti mentioned several behaviors that should raise alarm. One is a heightened physiological arousal when the person discusses the fantasy, such as rapid heartbeat, sweating; or physical posturing, such as clenching their fists or pounding their hands on an object as they tell you about it. You also want to determine the persistence of the fantasy.
“Can the patient think about it?” she asked. “Can they retain the ability to symbolize and separate themselves from necessarily doing whatever it is they think about?” You also want to determine the individual’s propensity for externalizing behaviors. “Here we’re talking about cluster B personality group patients – antisocial, narcissistic, and borderline patients who by virtue of their aggressivity titer and difficulties with anger, have a higher propensity for acting out and acting violently.”
Then there’s the concept of foreseeability. “Ask yourself, how likely is it that this could actually happen, based on the known risk factors and what you know about the patient?” Dr. Ferranti said. “Past history of violence is also very important. What people have done once before, they’re likely to do again.”
A good violence risk assessment can help you mitigate the potential for one of your patients to carry out harm to self or to others. Key risk factors include psychopathy, past violence, substance abuse, specific person/entity threatened, a history of impulsivity, unemployment, military history, gun possession, and the presence of paranoid and/or persecutory ideation or delusions.
“Know your specific state statutes and case law,” Dr. Ferranti concluded. “Delaying Tarasoff notification may indicate no need to violate confidentiality. If you think it’s warranted, do it without delay. Documentation is important when you’re consulting with therapists back and forth. You also want to attempt to obtain prior records and release only information that is required in a case of violence toward others. The details of the therapy or diagnosis are likely not relevant.”
Dr. Ferranti reported having no disclosures.
When patients relay their fantasies during psychotherapy sessions, those visions are often rooted in frustration or wish fulfillment, according to Jessica Ferranti, MD.
“[Sigmund] Freud talked about how our fantasy life is invested with large amounts of energy and interest and conveys a true essence of our personality – a truth about what we’re thinking and who we are,” Dr. Ferranti, a forensic psychiatrist in the division of psychiatry and the law at the University of California, Davis, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
“Fantasy life is one of the most important conveyances of information that we can get from our patients, whether in the general office or in the forensic realm – if we can access it, which is difficult, because fantasies are often intensely personal. They fall into the category of very high resistance topics with many patients.”
Psychiatrists routinely ask about violent thoughts and homicidal ideation, but violent fantasies – especially those that are sexually violent in nature – can be a warning sign of future danger. Dr. Ferranti defined violent fantasies as those depicting the use of physical force with the intent to injure another person or destroy property.
“This would be an individual who fantasizes about sadistically raping a woman, for instance,” said Dr. Ferranti, who directs the UC Davis Workplace Safety and Psychiatric Assessment Clinic. “That is an ominous and psychopathological sign in terms of the preoccupation with that kind of violent crime.”
Aggression, on the other hand, “is a very broad spectrum, with actions like assertion, interpersonal confrontation, or verbal expressions that are angry or hostile, but that do not necessarily lead to violence.”
Dr. Ferranti acknowledged that today’s rushed clinical environment makes it challenging for psychiatrists and psychologists to get patients to share detailed fantasies they may be harboring.
“It’s very difficult to get to deeper material with patients, unless potentially you have more intensive therapy going on, like a psychotherapeutic relationship where you see the patient frequently, an intensive treatment, [or] perhaps an inpatient hospitalization or a partial day program.” The key is that “the patient gets comfortable with relaying more of the truth about what they’re experiencing,” she said. “In some cases, this occurs during the forensic evaluation, because we have the luxury to do very lengthy evaluations. Under the stress of being with another person in the room for many hours, oftentimes the patient will disclose things eventually.
“I’ve been a forensic psychiatrist for the better part of 12 years, and I can tell you after hundreds of evaluations I’ve never had a person not speak. That’s a good thing, because a principle of the work we do, or talk therapy even, is that When we lose symbolism, the ability to represent things in our mind and speak about them, we are at greater risk of collapsing into the real and acting on the things we think about.”
Statutory reporting duties vary from state to state. In California, mandatory reporting duties include child abuse, elder abuse, abuse or neglect of developmentally disabled individuals, domestic violence, and victims of a gunshot wound. “Failing to report any of these crimes is a misdemeanor in California,” she said. “With all these statutory reporting duties, we have no legal obligation to inform the patient of the report. Under California law, patients do not have the right to refuse the report. These are reports we make in our best judgment, whether the patient is happy about that or not.”
What happens if your patient confesses to a past crime? “There’s no legal duty to report this,” Dr. Ferranti said. “The general rule is, unless there’s a current person who’s at risk, it would be violating confidentiality to report. This includes murder, bank robbery, and sexual assault. In addition, you cannot admit a patient to an inpatient setting to help them avoid arrest, even if you think the act in question was due to symptoms of a mental disorder, disease, or defect. You can actually be charged with aiding and abetting a criminal.”
In the 1976 landmark case Tarasoff v. the Regents of the University of California, the California Supreme Court ruled that psychiatrists and other therapists have a duty to do what is reasonably necessary to protect third parties if a patient presents a serious risk of violence to another person.
“Reasonable steps may include warning the third party, notifying police, detaining and hospitalizing the patient, intensifying the treatment to a higher level of care or more frequent outpatient appointments, removing weapons, and changing the medication therapy,” Dr. Ferranti said. “The more you can do of these, the better.”
She also discussed the concept of foreseeability, which she defined as the reasonable anticipation that harm or injury is likely to result from an act or omission to act.
“This is the malpractice standard for negligence,” she said. “In other words, was it foreseeable by a reasonable psychiatrist that this person was going to hurt someone else or themselves?” Another landmark case, Jablonski Pahls v. the United States broadened the reporting obligations of psychiatrists. In this 1983 case, the U.S. Court of Appeals 9th Circuit ruled that mental health professionals have to do more than warn foreseeable victims of an imminent danger of potential harm; they must involuntarily hospitalize the dangerous individual and consult that person’s prior records.
There is no sure-fire way to predict when an individual’s underlying violent fantasies are likely to be acted on, but Dr. Ferranti mentioned several behaviors that should raise alarm. One is a heightened physiological arousal when the person discusses the fantasy, such as rapid heartbeat, sweating; or physical posturing, such as clenching their fists or pounding their hands on an object as they tell you about it. You also want to determine the persistence of the fantasy.
“Can the patient think about it?” she asked. “Can they retain the ability to symbolize and separate themselves from necessarily doing whatever it is they think about?” You also want to determine the individual’s propensity for externalizing behaviors. “Here we’re talking about cluster B personality group patients – antisocial, narcissistic, and borderline patients who by virtue of their aggressivity titer and difficulties with anger, have a higher propensity for acting out and acting violently.”
Then there’s the concept of foreseeability. “Ask yourself, how likely is it that this could actually happen, based on the known risk factors and what you know about the patient?” Dr. Ferranti said. “Past history of violence is also very important. What people have done once before, they’re likely to do again.”
A good violence risk assessment can help you mitigate the potential for one of your patients to carry out harm to self or to others. Key risk factors include psychopathy, past violence, substance abuse, specific person/entity threatened, a history of impulsivity, unemployment, military history, gun possession, and the presence of paranoid and/or persecutory ideation or delusions.
“Know your specific state statutes and case law,” Dr. Ferranti concluded. “Delaying Tarasoff notification may indicate no need to violate confidentiality. If you think it’s warranted, do it without delay. Documentation is important when you’re consulting with therapists back and forth. You also want to attempt to obtain prior records and release only information that is required in a case of violence toward others. The details of the therapy or diagnosis are likely not relevant.”
Dr. Ferranti reported having no disclosures.
When patients relay their fantasies during psychotherapy sessions, those visions are often rooted in frustration or wish fulfillment, according to Jessica Ferranti, MD.
“[Sigmund] Freud talked about how our fantasy life is invested with large amounts of energy and interest and conveys a true essence of our personality – a truth about what we’re thinking and who we are,” Dr. Ferranti, a forensic psychiatrist in the division of psychiatry and the law at the University of California, Davis, said during an annual psychopharmacology update held by the Nevada Psychiatric Association.
“Fantasy life is one of the most important conveyances of information that we can get from our patients, whether in the general office or in the forensic realm – if we can access it, which is difficult, because fantasies are often intensely personal. They fall into the category of very high resistance topics with many patients.”
Psychiatrists routinely ask about violent thoughts and homicidal ideation, but violent fantasies – especially those that are sexually violent in nature – can be a warning sign of future danger. Dr. Ferranti defined violent fantasies as those depicting the use of physical force with the intent to injure another person or destroy property.
“This would be an individual who fantasizes about sadistically raping a woman, for instance,” said Dr. Ferranti, who directs the UC Davis Workplace Safety and Psychiatric Assessment Clinic. “That is an ominous and psychopathological sign in terms of the preoccupation with that kind of violent crime.”
Aggression, on the other hand, “is a very broad spectrum, with actions like assertion, interpersonal confrontation, or verbal expressions that are angry or hostile, but that do not necessarily lead to violence.”
Dr. Ferranti acknowledged that today’s rushed clinical environment makes it challenging for psychiatrists and psychologists to get patients to share detailed fantasies they may be harboring.
“It’s very difficult to get to deeper material with patients, unless potentially you have more intensive therapy going on, like a psychotherapeutic relationship where you see the patient frequently, an intensive treatment, [or] perhaps an inpatient hospitalization or a partial day program.” The key is that “the patient gets comfortable with relaying more of the truth about what they’re experiencing,” she said. “In some cases, this occurs during the forensic evaluation, because we have the luxury to do very lengthy evaluations. Under the stress of being with another person in the room for many hours, oftentimes the patient will disclose things eventually.
“I’ve been a forensic psychiatrist for the better part of 12 years, and I can tell you after hundreds of evaluations I’ve never had a person not speak. That’s a good thing, because a principle of the work we do, or talk therapy even, is that When we lose symbolism, the ability to represent things in our mind and speak about them, we are at greater risk of collapsing into the real and acting on the things we think about.”
Statutory reporting duties vary from state to state. In California, mandatory reporting duties include child abuse, elder abuse, abuse or neglect of developmentally disabled individuals, domestic violence, and victims of a gunshot wound. “Failing to report any of these crimes is a misdemeanor in California,” she said. “With all these statutory reporting duties, we have no legal obligation to inform the patient of the report. Under California law, patients do not have the right to refuse the report. These are reports we make in our best judgment, whether the patient is happy about that or not.”
What happens if your patient confesses to a past crime? “There’s no legal duty to report this,” Dr. Ferranti said. “The general rule is, unless there’s a current person who’s at risk, it would be violating confidentiality to report. This includes murder, bank robbery, and sexual assault. In addition, you cannot admit a patient to an inpatient setting to help them avoid arrest, even if you think the act in question was due to symptoms of a mental disorder, disease, or defect. You can actually be charged with aiding and abetting a criminal.”
In the 1976 landmark case Tarasoff v. the Regents of the University of California, the California Supreme Court ruled that psychiatrists and other therapists have a duty to do what is reasonably necessary to protect third parties if a patient presents a serious risk of violence to another person.
“Reasonable steps may include warning the third party, notifying police, detaining and hospitalizing the patient, intensifying the treatment to a higher level of care or more frequent outpatient appointments, removing weapons, and changing the medication therapy,” Dr. Ferranti said. “The more you can do of these, the better.”
She also discussed the concept of foreseeability, which she defined as the reasonable anticipation that harm or injury is likely to result from an act or omission to act.
“This is the malpractice standard for negligence,” she said. “In other words, was it foreseeable by a reasonable psychiatrist that this person was going to hurt someone else or themselves?” Another landmark case, Jablonski Pahls v. the United States broadened the reporting obligations of psychiatrists. In this 1983 case, the U.S. Court of Appeals 9th Circuit ruled that mental health professionals have to do more than warn foreseeable victims of an imminent danger of potential harm; they must involuntarily hospitalize the dangerous individual and consult that person’s prior records.
There is no sure-fire way to predict when an individual’s underlying violent fantasies are likely to be acted on, but Dr. Ferranti mentioned several behaviors that should raise alarm. One is a heightened physiological arousal when the person discusses the fantasy, such as rapid heartbeat, sweating; or physical posturing, such as clenching their fists or pounding their hands on an object as they tell you about it. You also want to determine the persistence of the fantasy.
“Can the patient think about it?” she asked. “Can they retain the ability to symbolize and separate themselves from necessarily doing whatever it is they think about?” You also want to determine the individual’s propensity for externalizing behaviors. “Here we’re talking about cluster B personality group patients – antisocial, narcissistic, and borderline patients who by virtue of their aggressivity titer and difficulties with anger, have a higher propensity for acting out and acting violently.”
Then there’s the concept of foreseeability. “Ask yourself, how likely is it that this could actually happen, based on the known risk factors and what you know about the patient?” Dr. Ferranti said. “Past history of violence is also very important. What people have done once before, they’re likely to do again.”
A good violence risk assessment can help you mitigate the potential for one of your patients to carry out harm to self or to others. Key risk factors include psychopathy, past violence, substance abuse, specific person/entity threatened, a history of impulsivity, unemployment, military history, gun possession, and the presence of paranoid and/or persecutory ideation or delusions.
“Know your specific state statutes and case law,” Dr. Ferranti concluded. “Delaying Tarasoff notification may indicate no need to violate confidentiality. If you think it’s warranted, do it without delay. Documentation is important when you’re consulting with therapists back and forth. You also want to attempt to obtain prior records and release only information that is required in a case of violence toward others. The details of the therapy or diagnosis are likely not relevant.”
Dr. Ferranti reported having no disclosures.
FROM NPA 2021
Mepolizumab reduced exacerbations in patients with asthma and atopy, depression comorbidities
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
, according to research from the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
“Mepolizumab has clearly been shown to improve severe asthma control in many clinical trials, but atopy, obesity, and depression/anxiety affect patients with asthma at an increased rate,” Thomas B. Casale, MD, former AAAAI president and professor of medicine and pediatrics at the University of South Florida in Tampa, said in a presentation at the meeting. “Yet, few studies have examined whether asthma therapy with these comorbidities works.”
Dr. Casale and colleagues performed a retrospective analysis of patients in the United States from the MarketScan Commercial and Medicare Supplemental Database between November 2014 and December 2018 who had atopy, obesity, or depression/anxiety in addition to asthma and were receiving mepolizumab. Atopy in the study was defined as allergic rhinitis, anaphylaxis, atopic dermatitis, conjunctivitis, eosinophilic esophagitis, and food allergies. Patients were at least age 12 years, had at least one diagnosis for asthma, at least one diagnosis code for atopic disease, obesity, or depression/anxiety at baseline, and at least two administrations of mepolizumab within 180 days.
The researchers examined the number of exacerbations, oral corticosteroid (OCS) claims, and OCS bursts per year at 12-month follow-up, compared with baseline. They identified exacerbations by examining patients who had an emergency department or outpatient claim related to their asthma, and a claim for systemic corticosteroids made in the 4 days prior to or 5 days after a visit, or if their inpatient hospital admission contained a primary asthma diagnosis. Dr. Casale and colleagues measured OCS bursts as a pharmacy claim of at least 20 mg of prednisone per day for between 3 and 28 days plus a claim for an emergency department visit related to asthma in the 7 days prior or 6 days after the claim.
At baseline, patients across all groups were mean age 50.5-52.4 years with a Charleson Comorbidity Index score between 1.1 and 1.4, a majority were women (59.0%-72.0%) and nearly all were commercially insured (88.0%-90.0%). Patients who used biologics at baseline and/or used a biologic that wasn’t mepolizumab during the follow-up period were excluded.
Medication claims in the groups included inhaled corticosteroids (ICS) (36.8%-48.6%), ICS/long-acting beta-agonist (LABA) (60.2%-63.0%), LABA/ long-acting muscarinic antagonist (LAMA) (1.2%-3.5%), ICS/LABA/LAMA (21.2%-25.1%), short-acting beta-agonist (SABA) (83.2%-87.7%), LAMA alone (33.5%-42.1%), or leukotriene receptor antagonist (LTRA).
In the non–mutually exclusive group of patients with atopy (468 patients), 28.0% had comorbid obesity and 26.0% had comorbid depression/anxiety. For patients with obesity categorized in a non–mutually exclusive subgroup (171 patients), 79.0% had comorbid atopy and 32.0% had comorbid depression/anxiety. Among patients with non–mutually exclusive depression/anxiety (173 patients), 70.0% had comorbid atopy, while 32.0% had comorbid obesity.
The results showed the mean number of overall exacerbations decreased by 48% at 12 months in the atopic group (2.3 vs. 1.2; P < .001), 52% in the group with obesity (2.5 vs. 1.2; P < .001), and 38% in the depression/anxiety group (2.4 vs. 1.5; P < .001). The mean number of exacerbations leading to hospitalizations decreased by 64% in the atopic group (0.11 vs. 0.04; P < .001), 65% in the group with obesity (0.20 vs. 0.07; P < .001), and 68% in the group with depression/anxiety (0.22 vs. 0.07; P < .001).
The researchers also found the mean number of OCS claims and OCS bursts also significantly decreased over the 12-month follow-up period. Mean OCS claims decreased by 33% for patients in the atopic group (5.5 vs. 3.7; P < .001), by 38% in the group with obesity (6.1 vs. 3.8; P < .001), and by 31% in the group with depression/anxiety (6.2 vs. 4.3; P < .001).
The mean number of OCS bursts also significantly decreased by 40% in the atopic group (2.0 vs. 2.1; P < .001), 48% in the group with obesity (2.3 vs. 1.2; P < .001), and by 37% in the group with depression/anxiety (1.9 vs. 1.2; P < .001). In total, 69% of patients with comorbid atopy, 70.8% of patients with comorbid obesity, and 68.2% of patients with comorbid depression/anxiety experienced a mean decrease in their OCS dose over 12 months.
“These data demonstrate that patients with asthma and atopy, obesity, or depression and anxiety have significantly fewer exacerbations and reduced OCS use in a real-world setting with treatment of mepolizumab,” Dr. Casale said. “Thus, holistic patient care for severe asthma is critical, and mepolizumab provides tangible clinical benefit despite the complexities of medical comorbidities.”
This study was funded by GlaxoSmithKline, and the company also funded graphic design support of the poster. Dr. Casale reports he has received research funds from GlaxoSmithKline. Four authors report being current or former GlaxoSmithKline employees; three authors report holding stock and/or shares of GlaxoSmithKline. Three authors are IBM Watson Health employees, a company GlaxoSmithKline has provided research funding.
FROM AAAAI 2021
Neurologic disorders ubiquitous and rising in the U.S.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
, according to new findings derived from the 2017 Global Burden of Disease study.
The authors of the analysis, led by Valery Feigin, MD, PhD, of New Zealand’s National Institute for Stroke and Applied Neurosciences, and published in the February 2021 issue of JAMA Neurology, looked at prevalence, incidence, mortality, and disability-adjusted life years for 14 neurological disorders across 50 states between 1990 and 2017. The diseases included in the analysis were stroke, Alzheimer’s disease and other dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, headaches, traumatic brain injury, spinal cord injuries, brain and other nervous system cancers, meningitis, encephalitis, and tetanus.
Tracking the burden of neurologic diseases
Dr. Feigin and colleagues estimated that a full 60% of the U.S. population lives with one or more of these disorders, a figure much greater than previous estimates for neurological disease burden nationwide. Tension-type headache and migraine were the most prevalent in the analysis by Dr. Feigin and colleagues. During the study period, they found, prevalence, incidence, and disability burden of nearly all the included disorders increased, with the exception of brain and spinal cord injuries, meningitis, and encephalitis.
The researchers attributed most of the rise in noncommunicable neurological diseases to population aging. An age-standardized analysis found trends for stroke and Alzheimer’s disease and other dementias to be declining or flat. Age-standardized stroke incidence dropped by 16% from 1990 to 2017, while stroke mortality declined by nearly a third, and stroke disability by a quarter. Age-standardized incidence of Alzheimer’s disease and other dementias dropped by 12%, and their prevalence by 13%, during the study period, though dementia mortality and disability were seen increasing.
The authors surmised that the age-standardized declines in stroke and dementias could reflect that “primary prevention of these disorders are beginning to show an influence.” With dementia, which is linked to cognitive reserve and education, “improving educational levels of cohort reaching the age groups at greatest risk of disease may also be contributing to a modest decline over time,” Dr. Feigin and his colleagues wrote.
Parkinson’s disease and multiple sclerosis, meanwhile, were both seen rising in incidence, prevalence, and disability adjusted life years (DALYs) even with age-standardized figures. The United States saw comparatively more disability in 2017 from dementias, Parkinson’s disease, epilepsy, multiple sclerosis, motor neuron disease, and headache disorders, which together comprised 6.7% of DALYs, compared with 4.4% globally; these also accounted for a higher share of mortality in the U.S. than worldwide. The authors attributed at least some of the difference to better case ascertainment in the U.S.
Regional variations
The researchers also reported variations in disease burden by state and region. While previous studies have identified a “stroke belt” concentrated in North Carolina, South Carolina, and Georgia, the new findings point to stroke disability highest in Alabama, Arkansas, and Mississippi, and mortality highest in Alabama, Mississippi, and South Carolina. The researchers noted increases in dementia mortality in these states, “likely attributable to the reciprocal association between stroke and dementia.”
Northern states saw higher burdens of multiple sclerosis compared with the rest of the country, while eastern states had higher rates of Parkinson’s disease.
Such regional and state-by state variations, Dr. Feigin and colleagues wrote in their analysis, “may be associated with differences in the case ascertainment, as well as access to health care; racial/ethnic, genetic, and socioeconomic diversity; quality and comprehensiveness of preventive strategies; and risk factor distribution.”
The researchers noted as a limitation of their study that the 14 diseases captured were not an exhaustive list of neurological conditions; chronic lower back pain, a condition included in a previous major study of the burden of neurological disease in the United States, was omitted, as were restless legs syndrome and peripheral neuropathy. The researchers cited changes to coding practice in the U.S. and accuracy of medical claims data as potential limitations of their analysis. The Global Burden of Disease study is funded by the Bill and Melinda Gates Foundation, and several of Dr. Feigin’s coauthors reported financial relationships with industry.
Time to adjust the stroke belt?
Amelia Boehme, PhD, a stroke epidemiologist at Columbia University Mailman School of Public Health in New York, said in an interview that the current study added to recent findings showing surprising local variability in stroke prevalence, incidence, and mortality. “What we had always conceptually thought of as the ‘stroke belt’ isn’t necessarily the case,” Dr. Boehme said, but is rather subject to local, county-by-county variations. “Looking at the data here in conjunction with what previous authors have found, it raises some questions as to whether or not state-level data is giving a completely accurate picture, and whether we need to start looking at the county level and adjust for populations and age.” Importantly, Dr. Boehme said, data collected in the Global Burden of Disease study tends to be exceptionally rigorous and systematic, adding weight to Dr. Feigin and colleagues’ suggestions that prevention efforts may be making a dent in stroke and dementia.
“More data is always needed before we start to say we’re seeing things change,” Dr. Boehme noted. “But any glimmer of optimism is welcome, especially with regard to interventions that have been put in place, to allow us to build on those interventions.”
Dr. Boehme disclosed no financial conflicts of interest.
FROM JAMA NEUROLOGY
Fired for good judgment a sign of physicians’ lost respect
What happened to Hasan Gokal, MD, should stick painfully in the craws of all physicians. It should serve as a call to action, because Dr. Gokal is sitting at home today without a job and under threat of further legal action while we continue about our day.
Dr. Gokal’s “crime” is that he vaccinated 10 strangers and acquaintances with soon-to-expire doses of the Moderna COVID-19 vaccine. He drove to the homes of some in the dark of night and injected others on his Sugar Land, Texas, lawn. He spent hours in a frantic search for willing recipients to beat the expiration clock. With minutes to spare, he gave the last dose to his at-risk wife, who has symptomatic pulmonary sarcoidosis, but whose age meant she did not fall into a vaccine priority tier.
According to the New York Times, Dr. Gokal’s wife was hesitant, afraid he might get into trouble. But why would she be hesitant? He wasn’t doing anything immoral. Perhaps she knew how far physicians have fallen and how bitterly they both could suffer.
In Barren County, Ky., where I live, a state of emergency was declared by our judge executive because of inclement weather. This directive allows our emergency management to “waive procedures and formalities otherwise required by the law.” It’s too bad that the same courtesy was not afforded to Dr. Gokal in Texas. It’s a shame that ice and snow didn’t drive his actions. Perhaps that would have protected him against the harsh criticism. Rather, it was his oath to patients and dedication to his fellow humans that motivated him, and for that, he was made to suffer.
Dr. Gokal was right to think that pouring the last 10 vaccine doses down the toilet would be an egregious act. But he was wrong in thinking his decision to find takers for the vaccine would be viewed as expedient. Instead, he was accused of graft and even nepotism. And there is the rub. That he was fired and charged with the theft of $137 worth of vaccines says everything about how physicians are treated in the year 2021. Dr. Gokal’s lawyer says the charge carried a maximum penalty of 1 year in prison and a fine of nearly $4,000.
Thank God a sage judge threw out the case and “rebuked” the office of District Attorney Kim Ogg. That hasn’t stopped her from threatening to bring the case to a grand jury. That threat invites anyone faced with the same scenario to flush the extra vaccine doses into the septic system. It encourages us to choose the toilet handle to avoid a mug shot.
And we can’t ignore the racial slant to this story. The Times reported that Dr. Gokal asked the officials, “Are you suggesting that there were too many Indian names in this group?”
“Exactly” was the answer. Let that sink in.
None of this would have happened 20 years ago. Back then, no one would have questioned the wisdom a physician gains from all our years of training and residency. In an age when anyone who conducts an office visit is now called “doctor,” respect for the letters “MD” has been leveled. We physicians have lost our autonomy and been cowed into submission.
But whatever his profession, Hasan Gokal was fired for being a good human. Today, the sun rose on 10 individuals who now enjoy better protection against a deadly pandemic. They include a bed-bound nonagenarian. A woman in her 80s with dementia. A mother with a child who uses a ventilator. All now have antibodies against SARS-CoV2 because of the tireless actions of Dr. Gokal.
Yet Dr. Gokal’s future is uncertain. Will we help him, or will we leave him to the wolves? In an email exchange with his lawyer’s office, I learned that Dr. Gokal has received offers of employment but is unable to entertain them because the actions by the Harris County District Attorney triggered an automatic review by the Texas Medical Board. A GoFundMe page was launched, but an appreciative Dr. Gokal stated publicly that he’d rather the money go to a needy charity.
In the last paragraph of the Times article, Dr. Gokal asks, “How can I take it back?” referencing stories about “the Pakistani doctor in Houston who stole all those vaccines.”
Let’s help him take back his story. In helping him, perhaps we can take back a little control. We could start with letters of support that could be mailed to his lawyer, Paul Doyle, Esq., of Houston, or tweet, respectfully of course, to the district attorney @Kimoggforda.
We can also let the Harris County Public Health Department in Houston know what we think of their actions.
On Martin Luther King Day, Kim Ogg, the district attorney who charged Dr. Gokal, tweeted MLK’s famous quote: “Injustice anywhere is a threat to justice everywhere.”
Let that motivate us to action.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. In addition to opinion writing, she enjoys spending time with her husband, daughters and parents, and sidelines as a backing vocalist for local rock bands. A version of this article first appeared on Medscape.com.
What happened to Hasan Gokal, MD, should stick painfully in the craws of all physicians. It should serve as a call to action, because Dr. Gokal is sitting at home today without a job and under threat of further legal action while we continue about our day.
Dr. Gokal’s “crime” is that he vaccinated 10 strangers and acquaintances with soon-to-expire doses of the Moderna COVID-19 vaccine. He drove to the homes of some in the dark of night and injected others on his Sugar Land, Texas, lawn. He spent hours in a frantic search for willing recipients to beat the expiration clock. With minutes to spare, he gave the last dose to his at-risk wife, who has symptomatic pulmonary sarcoidosis, but whose age meant she did not fall into a vaccine priority tier.
According to the New York Times, Dr. Gokal’s wife was hesitant, afraid he might get into trouble. But why would she be hesitant? He wasn’t doing anything immoral. Perhaps she knew how far physicians have fallen and how bitterly they both could suffer.
In Barren County, Ky., where I live, a state of emergency was declared by our judge executive because of inclement weather. This directive allows our emergency management to “waive procedures and formalities otherwise required by the law.” It’s too bad that the same courtesy was not afforded to Dr. Gokal in Texas. It’s a shame that ice and snow didn’t drive his actions. Perhaps that would have protected him against the harsh criticism. Rather, it was his oath to patients and dedication to his fellow humans that motivated him, and for that, he was made to suffer.
Dr. Gokal was right to think that pouring the last 10 vaccine doses down the toilet would be an egregious act. But he was wrong in thinking his decision to find takers for the vaccine would be viewed as expedient. Instead, he was accused of graft and even nepotism. And there is the rub. That he was fired and charged with the theft of $137 worth of vaccines says everything about how physicians are treated in the year 2021. Dr. Gokal’s lawyer says the charge carried a maximum penalty of 1 year in prison and a fine of nearly $4,000.
Thank God a sage judge threw out the case and “rebuked” the office of District Attorney Kim Ogg. That hasn’t stopped her from threatening to bring the case to a grand jury. That threat invites anyone faced with the same scenario to flush the extra vaccine doses into the septic system. It encourages us to choose the toilet handle to avoid a mug shot.
And we can’t ignore the racial slant to this story. The Times reported that Dr. Gokal asked the officials, “Are you suggesting that there were too many Indian names in this group?”
“Exactly” was the answer. Let that sink in.
None of this would have happened 20 years ago. Back then, no one would have questioned the wisdom a physician gains from all our years of training and residency. In an age when anyone who conducts an office visit is now called “doctor,” respect for the letters “MD” has been leveled. We physicians have lost our autonomy and been cowed into submission.
But whatever his profession, Hasan Gokal was fired for being a good human. Today, the sun rose on 10 individuals who now enjoy better protection against a deadly pandemic. They include a bed-bound nonagenarian. A woman in her 80s with dementia. A mother with a child who uses a ventilator. All now have antibodies against SARS-CoV2 because of the tireless actions of Dr. Gokal.
Yet Dr. Gokal’s future is uncertain. Will we help him, or will we leave him to the wolves? In an email exchange with his lawyer’s office, I learned that Dr. Gokal has received offers of employment but is unable to entertain them because the actions by the Harris County District Attorney triggered an automatic review by the Texas Medical Board. A GoFundMe page was launched, but an appreciative Dr. Gokal stated publicly that he’d rather the money go to a needy charity.
In the last paragraph of the Times article, Dr. Gokal asks, “How can I take it back?” referencing stories about “the Pakistani doctor in Houston who stole all those vaccines.”
Let’s help him take back his story. In helping him, perhaps we can take back a little control. We could start with letters of support that could be mailed to his lawyer, Paul Doyle, Esq., of Houston, or tweet, respectfully of course, to the district attorney @Kimoggforda.
We can also let the Harris County Public Health Department in Houston know what we think of their actions.
On Martin Luther King Day, Kim Ogg, the district attorney who charged Dr. Gokal, tweeted MLK’s famous quote: “Injustice anywhere is a threat to justice everywhere.”
Let that motivate us to action.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. In addition to opinion writing, she enjoys spending time with her husband, daughters and parents, and sidelines as a backing vocalist for local rock bands. A version of this article first appeared on Medscape.com.
What happened to Hasan Gokal, MD, should stick painfully in the craws of all physicians. It should serve as a call to action, because Dr. Gokal is sitting at home today without a job and under threat of further legal action while we continue about our day.
Dr. Gokal’s “crime” is that he vaccinated 10 strangers and acquaintances with soon-to-expire doses of the Moderna COVID-19 vaccine. He drove to the homes of some in the dark of night and injected others on his Sugar Land, Texas, lawn. He spent hours in a frantic search for willing recipients to beat the expiration clock. With minutes to spare, he gave the last dose to his at-risk wife, who has symptomatic pulmonary sarcoidosis, but whose age meant she did not fall into a vaccine priority tier.
According to the New York Times, Dr. Gokal’s wife was hesitant, afraid he might get into trouble. But why would she be hesitant? He wasn’t doing anything immoral. Perhaps she knew how far physicians have fallen and how bitterly they both could suffer.
In Barren County, Ky., where I live, a state of emergency was declared by our judge executive because of inclement weather. This directive allows our emergency management to “waive procedures and formalities otherwise required by the law.” It’s too bad that the same courtesy was not afforded to Dr. Gokal in Texas. It’s a shame that ice and snow didn’t drive his actions. Perhaps that would have protected him against the harsh criticism. Rather, it was his oath to patients and dedication to his fellow humans that motivated him, and for that, he was made to suffer.
Dr. Gokal was right to think that pouring the last 10 vaccine doses down the toilet would be an egregious act. But he was wrong in thinking his decision to find takers for the vaccine would be viewed as expedient. Instead, he was accused of graft and even nepotism. And there is the rub. That he was fired and charged with the theft of $137 worth of vaccines says everything about how physicians are treated in the year 2021. Dr. Gokal’s lawyer says the charge carried a maximum penalty of 1 year in prison and a fine of nearly $4,000.
Thank God a sage judge threw out the case and “rebuked” the office of District Attorney Kim Ogg. That hasn’t stopped her from threatening to bring the case to a grand jury. That threat invites anyone faced with the same scenario to flush the extra vaccine doses into the septic system. It encourages us to choose the toilet handle to avoid a mug shot.
And we can’t ignore the racial slant to this story. The Times reported that Dr. Gokal asked the officials, “Are you suggesting that there were too many Indian names in this group?”
“Exactly” was the answer. Let that sink in.
None of this would have happened 20 years ago. Back then, no one would have questioned the wisdom a physician gains from all our years of training and residency. In an age when anyone who conducts an office visit is now called “doctor,” respect for the letters “MD” has been leveled. We physicians have lost our autonomy and been cowed into submission.
But whatever his profession, Hasan Gokal was fired for being a good human. Today, the sun rose on 10 individuals who now enjoy better protection against a deadly pandemic. They include a bed-bound nonagenarian. A woman in her 80s with dementia. A mother with a child who uses a ventilator. All now have antibodies against SARS-CoV2 because of the tireless actions of Dr. Gokal.
Yet Dr. Gokal’s future is uncertain. Will we help him, or will we leave him to the wolves? In an email exchange with his lawyer’s office, I learned that Dr. Gokal has received offers of employment but is unable to entertain them because the actions by the Harris County District Attorney triggered an automatic review by the Texas Medical Board. A GoFundMe page was launched, but an appreciative Dr. Gokal stated publicly that he’d rather the money go to a needy charity.
In the last paragraph of the Times article, Dr. Gokal asks, “How can I take it back?” referencing stories about “the Pakistani doctor in Houston who stole all those vaccines.”
Let’s help him take back his story. In helping him, perhaps we can take back a little control. We could start with letters of support that could be mailed to his lawyer, Paul Doyle, Esq., of Houston, or tweet, respectfully of course, to the district attorney @Kimoggforda.
We can also let the Harris County Public Health Department in Houston know what we think of their actions.
On Martin Luther King Day, Kim Ogg, the district attorney who charged Dr. Gokal, tweeted MLK’s famous quote: “Injustice anywhere is a threat to justice everywhere.”
Let that motivate us to action.
Melissa Walton-Shirley, MD, is a native Kentuckian who retired from full-time invasive cardiology. She enjoys locums work in Montana and is a champion of physician rights and patient safety. In addition to opinion writing, she enjoys spending time with her husband, daughters and parents, and sidelines as a backing vocalist for local rock bands. A version of this article first appeared on Medscape.com.
Mindfulness can help patients manage ‘good’ change – and relief
Two themes have emerged recently in my psychotherapy practice, and in the mirror: relief and exhaustion. Some peace in the public discourse, or at least a pause in the ominous discord, has had the effect of a lightening, an unburdening. Some release from a contracted sense of tension around the specifics of violence and a broader sense of civil fracture has been palpable like a big, deep breath, exhaled. No sensible person would mistake this for being out of the metaphoric woods. A virus menaces and mutates, economic woes follow, and lots of us don’t get along. But, yes, there is some relief, some good change.
But even good change, even a downshift into relief, can pose some challenges to look for and overcome.
Consider for a moment the notion that every change represents a loss, a metaphoric “death” of the prior state of things. This is true of big, painful losses, like the death of a loved one, and small ones, like finding an empty cookie jar. It’s also true in changes we associate with benefit or relief: a refund check, a job promotion, a resolving migraine, or the breaking out of some civility.
In changes of all sorts, the world outside of one’s mind has shifted – at odds, momentarily, with our inner, now obsolete understanding of that changed world. The inside of the head does not match the outside. How we make that adjustment, so “inside = outside,” is a clinically familiar process: it’s grieving, with a sequence famously elaborated upon by Elisabeth Kübler-Ross, MD,1 and others.
We all likely know the steps: shock/denial, anger, “bargaining,” depression, and acceptance. A quick review: Our initial anxious/threat reaction leads to grievous judgment, to rationalizing “woulda/coulda/shoulda’s,” then to truly landing in the disappointment of a loss or change, and the accepting of a new steady state. Inside proceeds to match outside.
So, what then of relief? How do we process “good” change? I think we still must move from “in ≠ out” to “in = out,” navigating some pitfalls along the way.
Initial threat often remains; apprehension of the “new” still can generate energy, and even a sense of threat, regardless of a kiss or a shove. Our brainstems run roughshod over this first phase.
Step two is about judgment. We can move past the threat to, “How do I feel about it?” Here’s where grievous feeling gets swapped out for something more peak-positive – joy, or relief if the change represents an ending of a state of suffering, tension, or uncertainty.
The “bargaining” step still happens, but often around a kind of testing regimen: Is this too good to be true? Is it really different? We run scenarios.
The thud of disappointment also gets a makeover. It’s a settling into the beneficial change and its associations: gratitude, a sense of energy shifting.
The bookend “OK” seems anodyne here – why would anyone not accept relief, some good change?2 But it can nevertheless represent a challenge for many. The receding tension of the last year could open into a burst of energy, but I’m finding that exhaustion is just as or more common. That’s not illness, but a weary exhaling from the longest of held breaths.
One other twist: What happens when one of those steps is an individual obstacle, trigger, or hard-to-hold state? Especially for those with deep experience in disappointment or even trauma, buying into acceptance of a new normal can feel like a fool’s game. This is an especially complex spot for individuals who won’t quite allow for joyful acceptance to break out, lest it reveals itself as a humiliating trick or a too-brief respite from the “usual.”
Mindfulness practices, such as meditation, are helpful in managing this process. Committed time and optimal conditions to witness and adapt to the various inner states that ebb and flow generate a clear therapeutic benefit. Patients improve their identification of somatic manifestations, emotional reactions, and cycling ruminations of thought. What generates distraction and loss of mindful attention becomes better recognized. Contemplative work in between sessions becomes more productive.
What else do I advise?3 Patience, and some compassion for ourselves in this unusual time. Grief, and relief, are complex but truly human processes that generate not just one state of experience, but a cascade of them. While that cascade can hurt, it’s actually normal, not illness. But it can be exhausting.
Dr. Sazima is a Northern California psychiatrist, educator, and author. He is senior behavioral faculty at the Stanford-O’Connor Family Medicine Residency Program in San José, Calif. His latest book is “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021. Dr. Sazima disclosed no relevant financial relationships.
References
1. Kübler-Ross E. “On Death And Dying,” New York: Simon & Schuster, 1969.
2. Selye H. “Stress Without Distress,” New York: Lippincott, Williams & Wilkins, 1974.
3. Sazima G. “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021.
Two themes have emerged recently in my psychotherapy practice, and in the mirror: relief and exhaustion. Some peace in the public discourse, or at least a pause in the ominous discord, has had the effect of a lightening, an unburdening. Some release from a contracted sense of tension around the specifics of violence and a broader sense of civil fracture has been palpable like a big, deep breath, exhaled. No sensible person would mistake this for being out of the metaphoric woods. A virus menaces and mutates, economic woes follow, and lots of us don’t get along. But, yes, there is some relief, some good change.
But even good change, even a downshift into relief, can pose some challenges to look for and overcome.
Consider for a moment the notion that every change represents a loss, a metaphoric “death” of the prior state of things. This is true of big, painful losses, like the death of a loved one, and small ones, like finding an empty cookie jar. It’s also true in changes we associate with benefit or relief: a refund check, a job promotion, a resolving migraine, or the breaking out of some civility.
In changes of all sorts, the world outside of one’s mind has shifted – at odds, momentarily, with our inner, now obsolete understanding of that changed world. The inside of the head does not match the outside. How we make that adjustment, so “inside = outside,” is a clinically familiar process: it’s grieving, with a sequence famously elaborated upon by Elisabeth Kübler-Ross, MD,1 and others.
We all likely know the steps: shock/denial, anger, “bargaining,” depression, and acceptance. A quick review: Our initial anxious/threat reaction leads to grievous judgment, to rationalizing “woulda/coulda/shoulda’s,” then to truly landing in the disappointment of a loss or change, and the accepting of a new steady state. Inside proceeds to match outside.
So, what then of relief? How do we process “good” change? I think we still must move from “in ≠ out” to “in = out,” navigating some pitfalls along the way.
Initial threat often remains; apprehension of the “new” still can generate energy, and even a sense of threat, regardless of a kiss or a shove. Our brainstems run roughshod over this first phase.
Step two is about judgment. We can move past the threat to, “How do I feel about it?” Here’s where grievous feeling gets swapped out for something more peak-positive – joy, or relief if the change represents an ending of a state of suffering, tension, or uncertainty.
The “bargaining” step still happens, but often around a kind of testing regimen: Is this too good to be true? Is it really different? We run scenarios.
The thud of disappointment also gets a makeover. It’s a settling into the beneficial change and its associations: gratitude, a sense of energy shifting.
The bookend “OK” seems anodyne here – why would anyone not accept relief, some good change?2 But it can nevertheless represent a challenge for many. The receding tension of the last year could open into a burst of energy, but I’m finding that exhaustion is just as or more common. That’s not illness, but a weary exhaling from the longest of held breaths.
One other twist: What happens when one of those steps is an individual obstacle, trigger, or hard-to-hold state? Especially for those with deep experience in disappointment or even trauma, buying into acceptance of a new normal can feel like a fool’s game. This is an especially complex spot for individuals who won’t quite allow for joyful acceptance to break out, lest it reveals itself as a humiliating trick or a too-brief respite from the “usual.”
Mindfulness practices, such as meditation, are helpful in managing this process. Committed time and optimal conditions to witness and adapt to the various inner states that ebb and flow generate a clear therapeutic benefit. Patients improve their identification of somatic manifestations, emotional reactions, and cycling ruminations of thought. What generates distraction and loss of mindful attention becomes better recognized. Contemplative work in between sessions becomes more productive.
What else do I advise?3 Patience, and some compassion for ourselves in this unusual time. Grief, and relief, are complex but truly human processes that generate not just one state of experience, but a cascade of them. While that cascade can hurt, it’s actually normal, not illness. But it can be exhausting.
Dr. Sazima is a Northern California psychiatrist, educator, and author. He is senior behavioral faculty at the Stanford-O’Connor Family Medicine Residency Program in San José, Calif. His latest book is “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021. Dr. Sazima disclosed no relevant financial relationships.
References
1. Kübler-Ross E. “On Death And Dying,” New York: Simon & Schuster, 1969.
2. Selye H. “Stress Without Distress,” New York: Lippincott, Williams & Wilkins, 1974.
3. Sazima G. “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021.
Two themes have emerged recently in my psychotherapy practice, and in the mirror: relief and exhaustion. Some peace in the public discourse, or at least a pause in the ominous discord, has had the effect of a lightening, an unburdening. Some release from a contracted sense of tension around the specifics of violence and a broader sense of civil fracture has been palpable like a big, deep breath, exhaled. No sensible person would mistake this for being out of the metaphoric woods. A virus menaces and mutates, economic woes follow, and lots of us don’t get along. But, yes, there is some relief, some good change.
But even good change, even a downshift into relief, can pose some challenges to look for and overcome.
Consider for a moment the notion that every change represents a loss, a metaphoric “death” of the prior state of things. This is true of big, painful losses, like the death of a loved one, and small ones, like finding an empty cookie jar. It’s also true in changes we associate with benefit or relief: a refund check, a job promotion, a resolving migraine, or the breaking out of some civility.
In changes of all sorts, the world outside of one’s mind has shifted – at odds, momentarily, with our inner, now obsolete understanding of that changed world. The inside of the head does not match the outside. How we make that adjustment, so “inside = outside,” is a clinically familiar process: it’s grieving, with a sequence famously elaborated upon by Elisabeth Kübler-Ross, MD,1 and others.
We all likely know the steps: shock/denial, anger, “bargaining,” depression, and acceptance. A quick review: Our initial anxious/threat reaction leads to grievous judgment, to rationalizing “woulda/coulda/shoulda’s,” then to truly landing in the disappointment of a loss or change, and the accepting of a new steady state. Inside proceeds to match outside.
So, what then of relief? How do we process “good” change? I think we still must move from “in ≠ out” to “in = out,” navigating some pitfalls along the way.
Initial threat often remains; apprehension of the “new” still can generate energy, and even a sense of threat, regardless of a kiss or a shove. Our brainstems run roughshod over this first phase.
Step two is about judgment. We can move past the threat to, “How do I feel about it?” Here’s where grievous feeling gets swapped out for something more peak-positive – joy, or relief if the change represents an ending of a state of suffering, tension, or uncertainty.
The “bargaining” step still happens, but often around a kind of testing regimen: Is this too good to be true? Is it really different? We run scenarios.
The thud of disappointment also gets a makeover. It’s a settling into the beneficial change and its associations: gratitude, a sense of energy shifting.
The bookend “OK” seems anodyne here – why would anyone not accept relief, some good change?2 But it can nevertheless represent a challenge for many. The receding tension of the last year could open into a burst of energy, but I’m finding that exhaustion is just as or more common. That’s not illness, but a weary exhaling from the longest of held breaths.
One other twist: What happens when one of those steps is an individual obstacle, trigger, or hard-to-hold state? Especially for those with deep experience in disappointment or even trauma, buying into acceptance of a new normal can feel like a fool’s game. This is an especially complex spot for individuals who won’t quite allow for joyful acceptance to break out, lest it reveals itself as a humiliating trick or a too-brief respite from the “usual.”
Mindfulness practices, such as meditation, are helpful in managing this process. Committed time and optimal conditions to witness and adapt to the various inner states that ebb and flow generate a clear therapeutic benefit. Patients improve their identification of somatic manifestations, emotional reactions, and cycling ruminations of thought. What generates distraction and loss of mindful attention becomes better recognized. Contemplative work in between sessions becomes more productive.
What else do I advise?3 Patience, and some compassion for ourselves in this unusual time. Grief, and relief, are complex but truly human processes that generate not just one state of experience, but a cascade of them. While that cascade can hurt, it’s actually normal, not illness. But it can be exhausting.
Dr. Sazima is a Northern California psychiatrist, educator, and author. He is senior behavioral faculty at the Stanford-O’Connor Family Medicine Residency Program in San José, Calif. His latest book is “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021. Dr. Sazima disclosed no relevant financial relationships.
References
1. Kübler-Ross E. “On Death And Dying,” New York: Simon & Schuster, 1969.
2. Selye H. “Stress Without Distress,” New York: Lippincott, Williams & Wilkins, 1974.
3. Sazima G. “Practical Mindfulness: A Physician’s No-Nonsense Guide to Meditation for Beginners,” Miami: Mango Publishing, 2021.
Journal to retract psych paper after plagiarism allegations, editor steps down
A medical journal is retracting a paper after a psychiatrist alleged that the managing editor closely copied and published her withdrawn work and claimed it for his own.
In addition, the managing editor, Gary VandenBos, PhD, has resigned at the journal’s request, according to an email sent to the paper’s original author, psychiatrist Amy Barnhorst, MD, vice chair for community mental health at the University of California, Davis, and coauthor and UC Davis colleague Rocco Pallin, MPH.
Dr. Barnhorst shared emails – from the journal’s publisher, Springer Publishing Company, and from the editor in chief, Morgan Sammons, PhD – with this news organization.
The retraction is the end of a saga that began when Dr. Barnhorst and Dr. Pallin submitted a paper, at Dr. VandenBos’s request, to the Journal of Health Service Psychology, published by Springer.
As previously reported by this news organization, Dr. Barnhorst and Dr. Pallin eventually decided to withdraw the paper and were later notified by Dr. VandenBos that he’d published a similar article under his own authorship. Michael O. Miller, a retired judge who trained as a psychologist, was listed as a coauthor.
The two women – acknowledged experts in the article’s subject matter on how physicians can talk to patients about gun violence – immediately notified Dr. Sammons and Springer Publishing when they saw the published piece, saying they believed it plagiarized their original submission.
According to the e-mail Springer sent to Dr. Barnhorst, the publisher investigated and said that it would “be retracting the article shortly.”
The retraction notice will state: “The Editor in Chief and the authors, Dr. VandenBos and Mr. Miller, have retracted this article, as it significantly overlaps with an unpublished manuscript by Amy Barnhorst and Rocco Pallin.” It also states that “[Dr.] VandenBos accepts full responsibility for the overlap.”
The original article will still be available, but it will be marked as “retracted” and feature a link to the retraction notice.
Dr. Barnhorst, who garnered at least 40,000 likes when she tweeted about the alleged theft of her work, said in an interview that she and Dr. Pallin are “glad to see that the investigation is complete and the retraction has been issued.”
“At least we can now submit it to a new journal in a version that appropriately represents our work and expertise,” said Dr. Barnhorst. “I still have no idea how or why this happened, nor how much of it was intentional and on whose part, but I guess I never will!”
Editor removed
When contacted by this news organization to comment on the retraction and removal of Dr. VandenBos as managing editor, Dr. Sammons said it was not possible because, “I treat such correspondence as confidential.”
Dr. Sammons said he could “confirm that our investigation is reaching its conclusion and my colleagues at Springer Nature would be happy to contact you when we can provide a further update.”
Springer spokesperson Anne Korn also would not comment beyond saying, “The conclusion of our investigation is still in progress and may take a little additional time.”
In the letter sent to Dr. Barnhorst and Dr. Pallin, Dr. Sammons said he had “asked for and accepted the resignation of Dr. VandenBos,” and that the resignation will be announced “in the upcoming print issue of our journal.”
Dr. Sammons said he also notified the dean of the University of Arizona College of Law that Mr. Miller, who held a position at the school, was not aware of the original submission by the two women. Even so, the school suspended Mr. Miller’s academic appointment, according to Dr. Sammons’ letter.
The editor in chief also said that, while “[Dr.] VandenBos’ errors were substantial and had substantial consequences, my investigation did not find any intent to plagiarize your work.”
He apologized again to Dr. Barnhorst and Dr. Pallin, though, adding, “I and my associate editors have initiated a revision of our publications processes to ensure that errors such as the above do not occur in the future, and I apologize again that this lapse affected you and your scholarly work.”
A version of this article first appeared on Medscape.com.
A medical journal is retracting a paper after a psychiatrist alleged that the managing editor closely copied and published her withdrawn work and claimed it for his own.
In addition, the managing editor, Gary VandenBos, PhD, has resigned at the journal’s request, according to an email sent to the paper’s original author, psychiatrist Amy Barnhorst, MD, vice chair for community mental health at the University of California, Davis, and coauthor and UC Davis colleague Rocco Pallin, MPH.
Dr. Barnhorst shared emails – from the journal’s publisher, Springer Publishing Company, and from the editor in chief, Morgan Sammons, PhD – with this news organization.
The retraction is the end of a saga that began when Dr. Barnhorst and Dr. Pallin submitted a paper, at Dr. VandenBos’s request, to the Journal of Health Service Psychology, published by Springer.
As previously reported by this news organization, Dr. Barnhorst and Dr. Pallin eventually decided to withdraw the paper and were later notified by Dr. VandenBos that he’d published a similar article under his own authorship. Michael O. Miller, a retired judge who trained as a psychologist, was listed as a coauthor.
The two women – acknowledged experts in the article’s subject matter on how physicians can talk to patients about gun violence – immediately notified Dr. Sammons and Springer Publishing when they saw the published piece, saying they believed it plagiarized their original submission.
According to the e-mail Springer sent to Dr. Barnhorst, the publisher investigated and said that it would “be retracting the article shortly.”
The retraction notice will state: “The Editor in Chief and the authors, Dr. VandenBos and Mr. Miller, have retracted this article, as it significantly overlaps with an unpublished manuscript by Amy Barnhorst and Rocco Pallin.” It also states that “[Dr.] VandenBos accepts full responsibility for the overlap.”
The original article will still be available, but it will be marked as “retracted” and feature a link to the retraction notice.
Dr. Barnhorst, who garnered at least 40,000 likes when she tweeted about the alleged theft of her work, said in an interview that she and Dr. Pallin are “glad to see that the investigation is complete and the retraction has been issued.”
“At least we can now submit it to a new journal in a version that appropriately represents our work and expertise,” said Dr. Barnhorst. “I still have no idea how or why this happened, nor how much of it was intentional and on whose part, but I guess I never will!”
Editor removed
When contacted by this news organization to comment on the retraction and removal of Dr. VandenBos as managing editor, Dr. Sammons said it was not possible because, “I treat such correspondence as confidential.”
Dr. Sammons said he could “confirm that our investigation is reaching its conclusion and my colleagues at Springer Nature would be happy to contact you when we can provide a further update.”
Springer spokesperson Anne Korn also would not comment beyond saying, “The conclusion of our investigation is still in progress and may take a little additional time.”
In the letter sent to Dr. Barnhorst and Dr. Pallin, Dr. Sammons said he had “asked for and accepted the resignation of Dr. VandenBos,” and that the resignation will be announced “in the upcoming print issue of our journal.”
Dr. Sammons said he also notified the dean of the University of Arizona College of Law that Mr. Miller, who held a position at the school, was not aware of the original submission by the two women. Even so, the school suspended Mr. Miller’s academic appointment, according to Dr. Sammons’ letter.
The editor in chief also said that, while “[Dr.] VandenBos’ errors were substantial and had substantial consequences, my investigation did not find any intent to plagiarize your work.”
He apologized again to Dr. Barnhorst and Dr. Pallin, though, adding, “I and my associate editors have initiated a revision of our publications processes to ensure that errors such as the above do not occur in the future, and I apologize again that this lapse affected you and your scholarly work.”
A version of this article first appeared on Medscape.com.
A medical journal is retracting a paper after a psychiatrist alleged that the managing editor closely copied and published her withdrawn work and claimed it for his own.
In addition, the managing editor, Gary VandenBos, PhD, has resigned at the journal’s request, according to an email sent to the paper’s original author, psychiatrist Amy Barnhorst, MD, vice chair for community mental health at the University of California, Davis, and coauthor and UC Davis colleague Rocco Pallin, MPH.
Dr. Barnhorst shared emails – from the journal’s publisher, Springer Publishing Company, and from the editor in chief, Morgan Sammons, PhD – with this news organization.
The retraction is the end of a saga that began when Dr. Barnhorst and Dr. Pallin submitted a paper, at Dr. VandenBos’s request, to the Journal of Health Service Psychology, published by Springer.
As previously reported by this news organization, Dr. Barnhorst and Dr. Pallin eventually decided to withdraw the paper and were later notified by Dr. VandenBos that he’d published a similar article under his own authorship. Michael O. Miller, a retired judge who trained as a psychologist, was listed as a coauthor.
The two women – acknowledged experts in the article’s subject matter on how physicians can talk to patients about gun violence – immediately notified Dr. Sammons and Springer Publishing when they saw the published piece, saying they believed it plagiarized their original submission.
According to the e-mail Springer sent to Dr. Barnhorst, the publisher investigated and said that it would “be retracting the article shortly.”
The retraction notice will state: “The Editor in Chief and the authors, Dr. VandenBos and Mr. Miller, have retracted this article, as it significantly overlaps with an unpublished manuscript by Amy Barnhorst and Rocco Pallin.” It also states that “[Dr.] VandenBos accepts full responsibility for the overlap.”
The original article will still be available, but it will be marked as “retracted” and feature a link to the retraction notice.
Dr. Barnhorst, who garnered at least 40,000 likes when she tweeted about the alleged theft of her work, said in an interview that she and Dr. Pallin are “glad to see that the investigation is complete and the retraction has been issued.”
“At least we can now submit it to a new journal in a version that appropriately represents our work and expertise,” said Dr. Barnhorst. “I still have no idea how or why this happened, nor how much of it was intentional and on whose part, but I guess I never will!”
Editor removed
When contacted by this news organization to comment on the retraction and removal of Dr. VandenBos as managing editor, Dr. Sammons said it was not possible because, “I treat such correspondence as confidential.”
Dr. Sammons said he could “confirm that our investigation is reaching its conclusion and my colleagues at Springer Nature would be happy to contact you when we can provide a further update.”
Springer spokesperson Anne Korn also would not comment beyond saying, “The conclusion of our investigation is still in progress and may take a little additional time.”
In the letter sent to Dr. Barnhorst and Dr. Pallin, Dr. Sammons said he had “asked for and accepted the resignation of Dr. VandenBos,” and that the resignation will be announced “in the upcoming print issue of our journal.”
Dr. Sammons said he also notified the dean of the University of Arizona College of Law that Mr. Miller, who held a position at the school, was not aware of the original submission by the two women. Even so, the school suspended Mr. Miller’s academic appointment, according to Dr. Sammons’ letter.
The editor in chief also said that, while “[Dr.] VandenBos’ errors were substantial and had substantial consequences, my investigation did not find any intent to plagiarize your work.”
He apologized again to Dr. Barnhorst and Dr. Pallin, though, adding, “I and my associate editors have initiated a revision of our publications processes to ensure that errors such as the above do not occur in the future, and I apologize again that this lapse affected you and your scholarly work.”
A version of this article first appeared on Medscape.com.
The lasting effects of childhood trauma
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21
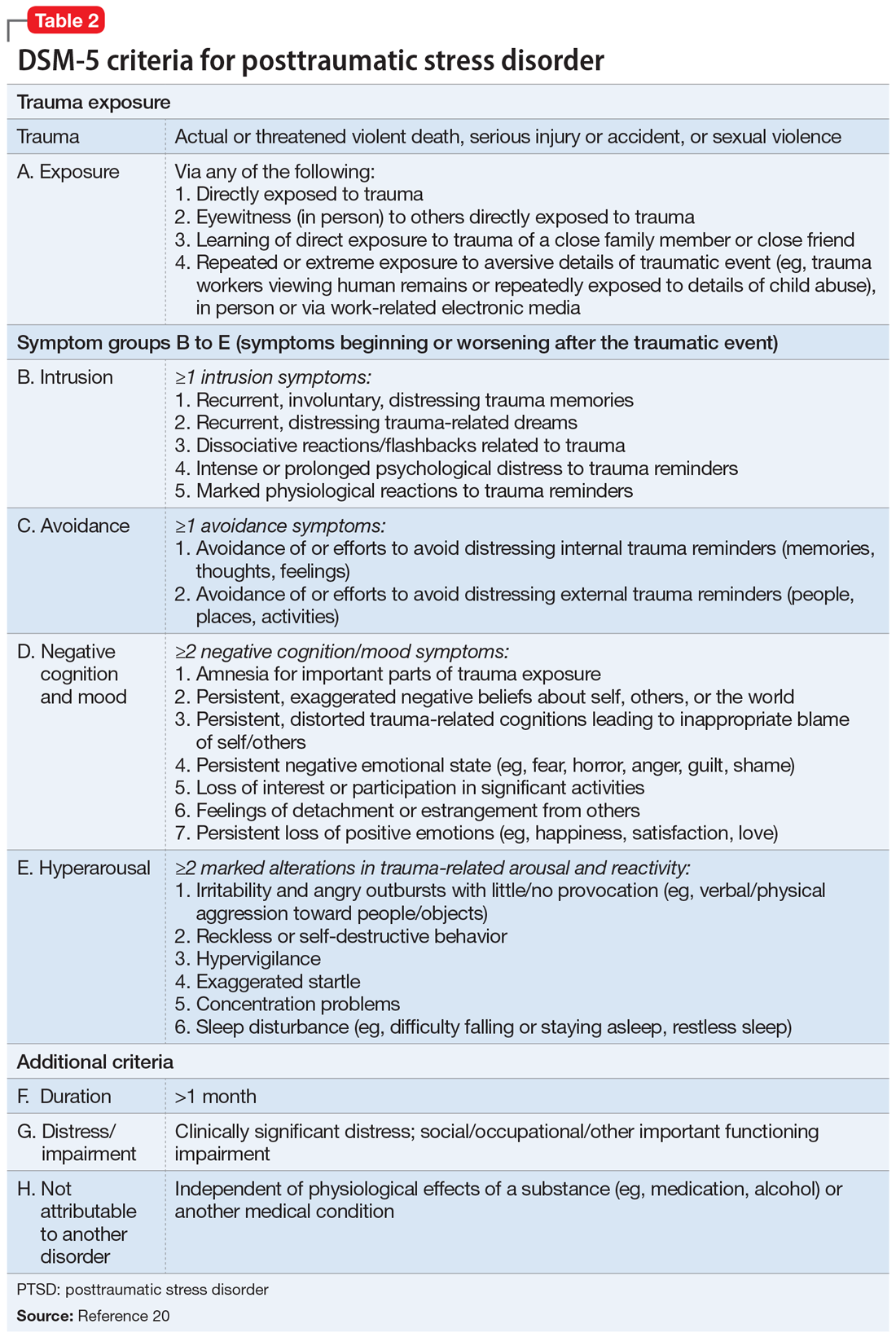
Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21

Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
Childhood trauma, which is also called adverse childhood experiences (ACEs), can have lasting detrimental effects on individuals as they grow and mature into adulthood. ACEs may occur in children age ≤18 years if they experience abuse or neglect, violence, or other traumatic losses. More than 60% of people experience at least 1 ACE, and 1 in 6 individuals reported that they had experienced ≥4 ACEs.1 Subsequent additional ACEs have a cumulative deteriorating impact on the brain. This predisposes individuals to mental health disorders, substance use disorders, and other psychosocial problems. The efficacy of current therapeutic approaches provides only partial symptom resolution. For such individuals, the illness load and health care costs typically remain high across the lifespan.1,2
In this article, we discuss types of ACEs, protective factors and risk factors that influence the development of posttraumatic stress disorder (PTSD) in individuals who experience ACEs, how ACEs can negatively impact mental health in adulthood, and approaches to prevent or treat PTSD and other symptoms.
Types of trauma and correlation with PTSD
ACEs can be indexed as neglect or emotional, physical, or sexual abuse. Physical and sexual abuse strongly correlate with an increased risk of PTSD.3 Although neglect and emotional abuse do not directly predict the development of PTSD, these experiences foretell high rates of lifelong trauma exposure and are indirectly related to late PTSD symptoms.4,5 ACEs can impede an individual’s cognitive, social, and emotional development, diminish quality of life, and lead to an early death.6 The lifetime prevalence of PTSD is 6.1% to 9.2%.7 Compared with men, women are 4 times more likely to develop PTSD following a traumatic event.7
The development of PTSD is influenced by the nature, duration, and degree of trauma, and age at the time of exposure to trauma. Children who survive complex trauma (≥2 types of trauma) have a higher likelihood of developing PTSD.8 Prolonged trauma exposure has a more substantial negative impact than a one-time occurrence. However, it is an erroneous oversimplification to assume that each type of ACE has an equally traumatic effect.6
Factors that protect against PTSD
Factors that can protect against developing PTSD are listed in Table 1.7 Two of these are resilience and hope.
Resilience is defined as an individual’s strength to cope with difficulties in life.9 Resilience has internal psychological characteristics and external factors that aid in protecting against childhood adversities.10,11 The Brief Resilience Scale is a self-assessment that measures innate abilities to cope, including optimism, self-efficacy, patience, faith, and humor.12,13 External factors associated with resilience are family, friends, and community support.11,13
Hope can help in surmounting ACEs. The Adult Hope Scale has been used in many studies to assess this construct in individuals who have survived trauma.13 Some studies have found decreased hope in individuals who sustained early trauma and were diagnosed with PTSD in adulthood.14 A study examining children exposed to domestic violence found that children who showed high hope, endurance, and curiosity were better able to cope with adversities.15
Continue to: PTSD risk factors
PTSD risk factors
Many individual and societal risk factors can influence the likelihood of developing PTSD. Some of these factors are outlined in Table 1.7
Pathophysiology of PTSD
Multiple brain regions, pathways, and neurotransmitters are involved in the development of PTSD. Neuroimaging has identified volume and activity changes of the hippocampus, prefrontal cortex, and amygdala in patients with early trauma and PTSD. Some researchers have suggested a gross reduction in locus coeruleus neuronal volume in war veterans with a likely diagnosis of PTSD compared with controls.16,17 In other studies, chronic stress exposure has been found to cause neuronal cell death and affect neuronal plasticity in the limbic area of the brain.18
Diagnosing PTSD
More than 30% of individuals who experience ACEs develop PTSD.19 The DSM-5 diagnostic criteria for PTSD are outlined in Table 2.20 Several instruments are used to determine the diagnosis and assess the severity of PTSD. These include the Clinician-Administered PTSD Scale for DSM-5,21 which is a 30-item structured interview that can be administered in 45 to 60 minutes; the PTSD Symptom Scale Self-Report Version, which is a 17-item, Likert scale, self-report questionnaire; and the Structured Clinical Interview: PTSD Module, which is a semi-structured interview that can take up to several hours to administer.21

Other disorders. In addition to PTSD, individuals with ACEs are at high risk for other mental health issues throughout their lifetime. Individuals with ACE often experience depressive symptoms (approximately 40%); anxiety (approximately 30%); anger; guilt or shame; negative self-cognition; interpersonal difficulties; rumination; and thoughts of self-harm and suicide.22 Epidemiological studies suggest that patients who experience childhood sexual abuse are more likely to develop mood, anxiety, and substance use disorders in adulthood.23,24
Psychotherapeutic treatments for PTSD
Cognitive-behavioral therapy (CBT) addresses the relationship between an individual’s thoughts, emotions, and behaviors. CBT can be used to treat adults and children with PTSD. Before starting CBT, assess the patient’s current safety to ensure that they have the coping skills to manage distress related to their ACEs, and address any coexisting substance use.25
Continue to: According to the American Psychological Association...
According to the American Psychological Association, several CBT-based psychotherapies are recommended for treating PTSD26:
Trauma-focused–CBT includes psychoeducation, trauma narrative, processing, exposure, and relaxation skills training. It consists of approximately 12 to 16 sessions and incorporates elements of family therapy.
Cognitive processing therapy (CPT) focuses on helping patients develop adaptive cognitive domains about the self, the people around them, and the world. CPT therapists assist in information processing by accessing the traumatic memory and trying to eliminate emotions tied to it.25,27 CPT consists of 12 to 16 structured individual, group, or combined sessions.
Prolonged exposure (PE) targets fear-related emotions and works on the principles of habituation to extinguish trauma and fear response to the trigger. This increases self-reliance and competence and decreases the generalization of anxiety to innocuous triggers. PE typically consists of 9 to 12 sessions. PE alone or in combination with cognitive restructuring is successful in treating patients with PTSD, but cognitive restructuring has limited utility in young children.25,27
Cognitive exposure can be individual or group therapy delivered over 3 months, where negative self-evaluation and traumatic memories are challenged with the goal of interrupting maladaptive behaviors and thoughts.27
Continue to: Stress inoculation training
Stress inoculation training (SIT) provides psychoeducation, skills training, role-playing, deep muscle relaxation, paced breathing, and thought stopping. Emphasis is on coaching skills to alleviate anxiety, fear, and symptoms of depression associated with trauma. In SIT, exposures to traumatic memories are indirect (eg, role play), compared with PE, where the exposures are direct.25
The American Psychological Association conditionally recommended several other forms for psychotherapy for treating patients with PTSD26:
Brief eclectic psychotherapy uses CBT and psychodynamic approaches to target feelings of guilt and shame in 16 sessions.27
Narrative exposure therapy consists of 4 to 10 group sessions in which individuals provide detailed narration of the events; the focus is on self-respect and personal rights.27
Eye movement desensitization and reprocessing (EMDR) is a 6- to 12-session, 8-phase treatment that uses principles of accelerated information processing to target nonverbal expression of trauma and dissociative experiences. Patients with PTSD are suggested to have disrupted rapid eye movements. In EMDR, patients follow rhythmic movements of the therapist’s hands or flashed light. This is designed to decrease stress associated with accessing trauma memories, the emotional/physiologic response from the memories, and negative cognitive distortions about self, and to replace negative cognition distortions with positive thoughts about self.25,27
Continue to: Accelerated resolution therapy
Accelerated resolution therapy is a derivative of EMDR. It helps to reconsolidate the emotional and physical experiences associated with distressing memories by replacing them with positive ones or decreasing physiological arousal and anxiety related to the recall of traumatic memories.28
Pharmacologic treatments
Selective serotonin reuptake inhibitors (SSRIs). Multiple studies using different scales have found that paroxetine, sertraline, and fluoxetine can decrease PTSD symptoms. Approximately 60% of patients treated with SSRIs experience partial remission of symptoms, and 20% to 30% experience complete symptom resolution.29 Davidson et al30 found that 22% of patients with PTSD who received fluoxetine had a relapse of symptoms, compared with 50% of patients who received placebo.
Serotonin-norepinephrine reuptake inhibitors (SNRIs) and other antidepressants. The SNRIs venlafaxine and duloxetine can help reduce hyperarousal symptoms and improve mood, anxiety, and sleep.26 Mirtazapine, an alpha 2A/2C adrenoceptor antagonist/5-HT 2A/2C/3 antagonist, can address PTSD symptoms from both serotonergic pathways and increase norepinephrine release by blocking autoreceptors and enhancing alpha-1 receptor activity. This alleviates hyperarousal symptoms and promotes sleep.29 In addition to having monoaminergic effects, antidepressant medications also regulate the hypothalamic–pituitary–adrenal (HPA) axis response to stress and promote neurogenesis in the hippocampal region.29
Adrenergic agents
Adrenergic receptor antagonists. Prazosin, an alpha-1 adrenoceptor antagonist, decreases hyperarousal symptoms, improves sleep, and decreases nightmares related to PTSD by decreasing noradrenergic hyperactivity.29
Beta-blockers such as propranolol can decrease physiological response to trauma but have mixed results in the prevention or improvement of PTSD symptoms.29,31
Continue to: Glucocorticoid receptor agonists
Glucocorticoid receptor agonists. In a very small study, low-dose cortisol decreased the severity of traumatic memory (consolidation phase).32 Glucocorticoid receptor agonists can also diminish memory retrieval (reconsolidation phase) through intrusive thoughts and flashbacks.29
Anticonvulsants, benzodiazepines, and antipsychotics
These medications have had a limited role in the treatment of PTSD.26,29
Future directions: Preventive treatments
Because PTSD has a profound impact on an individual’s quality of life and the development of other illnesses, there is strong interest in finding treatments that can prevent PTSD. Based on limited evidence primarily from animal studies, some researchers have suggested that certain agents may someday be helpful for PTSD prevention29:
Glucocorticoid antagonists such as corticotropin-releasing factor 1 (CRF1) antagonists or cholecystokinin 2 (CCK2) receptor antagonists might promote resilience to stress by inhibiting the HPA axis and influencing the amygdala by decreasing fear conditioning, as observed in animal models. Similarly, in animal models, CRF1 and CCK2 are predicted to decrease memory consolidation in response to exposure to stress.
Adrenoceptor antagonists and agonists also might have a role in preventive treatment, but the evidence is scarce. Prazosin, an alpha-1 adrenoceptor antagonist, was ineffective in animal models.29,31 Propranolol, a beta-adrenoceptor blocker, has had mixed results but can decrease trauma-induced physiological arousal when administered soon after exposure.29
Continue to: N-methyl-
N-methyl-
Bottom Line
Adverse childhood experiences (ACEs) are strong predictors for the development of posttraumatic stress disorder (PTSD) and other mental health or medical issues in late adolescence and adulthood. Experiencing a higher number of ACEs increases the risk of developing PTSD as an adult. Timely psychotherapeutic and pharmacologic interventions can help limit symptoms and reduce the severity of PTSD.
Related Resources
- Smith P, Dalglesih T, Meiser-Stedman R. Practitioner review: posttraumatic stress disorder and its treatment in children and adolescents. J Child Psychol Psychiatry. 2019;60(5):500-515.
- North CS, Hong BA, Downs DL. PTSD: a systematic approach to diagnosis and treatment. Current Psychiatry 2018;17(4):35-43.
Drug Brand Names
Duloxetine • Cymbalta
Fluoxetine • Prozac
Mirtazapine • Remeron
Paroxetine • Paxil
Prazosin • Minipress
Propranolol • Inderal, Pronol
Sertraline • Zoloft
Venlafaxine • Effexor
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
1. Centers for Disease Control and Prevention. Preventing adverse childhood experiences. Published April 3, 2020. Accessed January 26, 2021. https://www.cdc.gov/violenceprevention/childabuseandneglect/aces/fastfact.html
2. Kessler RC, McLaughlin KA, Green JG, et al. Childhood adversities and adult psychopathology in the WHO world mental health surveys. Br J Psychiatry. 2010;197:378-385.
3. Norman RE, Byambaa M, De R, et al. The long-term health consequences of child physical abuse, emotional abuse, and neglect: a systematic review and meta-analysis. PLoS Medicine. 2012;9(11):e1001349. doi: 10.1371/journal.pmed.1001349
4. Spertus IL, Yehuda R, Wong CM, et al. Childhood emotional abuse and neglect as predictors of psychological and physical symptoms in women presenting to a primary care practice. Child Abuse Negl. 2003;27(11):1247-1258.
5. Glück TM, Knefel M, Lueger-Schuster B. A network analysis of anger, shame, proposed ICD-11 post-traumatic stress disorder, and different types of childhood trauma in foster care settings in a sample of adult survivors. Eur J Psychotraumatol. 2017;8(suppl 3):1372543. doi: 10.1080/20008198.2017.1372543
6. Edwards VJ, Holden GW, Felitti VJ, et al. Relationship between multiple forms of childhood maltreatment and adult mental health in community respondents: results from the adverse childhood experiences study. Am J Psychiatry. 2003;160:1453-1460.
7. Sareen J. Posttraumatic stress disorder in adults: epidemiology, pathophysiology, clinical manifestations, course, assessment, and diagnosis. UpToDate. Updated December 3, 2020. Accessed January 26, 2021. https://www.uptodate.com/contents/posttraumatic-stress-disorder-in-adults-epidemiology-pathophysiology-clinical-manifestations-course-assessment-and-diagnosis
8. Widom CS. Posttraumatic stress disorder in abused and neglected children grown up. Am J Psychiatry. 1999:156;1223-1229.
9. Rutter M. Psychosocial resilience and protective mechanisms. Am J Orthopsychiatry. 1987;57(3):316-331.
10. Ahern NR, Kiehl EM, Sole ML, et al. A review of instruments measuring resilience. Issues Compr Pediatr Nurs. 2006;29(2):103-125.
11. Zimmerman MA. Resiliency theory: a strengths-based approach to research and practice for adolescent health. Health Educ Behav. 2013;40(4):381-383.
12. Connor KM, Davidson JR. Development of a new resilience scale: the Connor-Davidson Resilience Scale (CD-RISC). Depress Anxiety. 2003;18(2):76-82.
13. Munoz RT, Hanks H, Hellman CM. Hope and resilience as distinct contributors to psychological flourishing among childhood trauma survivors. Traumatology. 2020;26(2):177-184.
14. Baxter MA, Hemming EJ, McIntosh HC, et al. Exploring the relationship between adverse childhood experiences and hope. J Child Sex Abus. 2017;26(8):948-956.
15. Hellman CM, Gwinn C. Camp HOPE as an intervention for children exposed to domestic violence: a program evaluation of hope, and strength of character. Child Adolesc Soc Work J. 2017;34:269-276.
16. Bracha HS, Garcia-Rill E, Mrak RE, et al. Postmortem locus coeruleus neuron count in three American veterans with probable or possible war-related PTSD. J Neuropsychiatry Clin Neurosci. 2005;17(4):503-9.
17. de Lange GM. Understanding the cellular and molecular alterations in PTSD brains: the necessity of post-mortem brain tissue. Eur J Psychotraumatol. 2017;8(1):1341824. doi: 10.1080/20008198.2017.1341824
18. Zunszain PA, Anacker C, Cattaneo A, et al. Glucocorticoids, cytokines and brain abnormalities in depression. Prog Neuropsychopharmacol Biol Psychiatry. 2011;35(3):722-729.
19. Greeson JKP, Briggs EC, Kisiel CL, et al. Complex trauma and mental health in children and adolescents placed in foster care: findings from the national child traumatic stress network. Child Welfare. 2011;90(6):91-108.
20. Diagnostic and statistical manual of mental disorders, 5th ed. American Psychiatric Association; 2013.
21. American Psychological Association. PTSD assessment instruments. Updated September 26, 2018. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/assessment/
22. Bellis MA, Hughes K, Ford K, et al. Life course health consequences and associated annual costs of adverse childhood experiences across Europe and North America: a systematic review and meta-analysis. Lancet Public Health. 2019;4(10):e517-e528. doi: 10.1016/S2468-2667(19)30145-8
23. Mullen PE, Martin JL, Anderson JC, et al. Childhood sexual abuse and mental health in adult life. Br J Psychiatry. 1993;163:721-732.
24. Kendler KS, Bulik CM, Silberg J, et al. Childhood sexual abuse and adult psychiatric and substance use disorders in women. An epidemiological and cotwin control analysis. Arch Gen Psychiatry. 2000;57(10):953-959.
25. Chard KM, Gilman R. Counseling trauma victims: 4 brief therapies meet the test. Current Psychiatry. 2005;4(8):50,55-58,61-62.
26. Guideline Development Panel for the Treatment of PTSD in Adults, American Psychological Association. Summary of the clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. American Psychol. 2019;74(5):596-607.
27. American Psychological Association. Clinical practice guideline for the treatment of posttraumatic stress disorder. PTSD treatments. Updated June 2020. Accessed January 27, 2021. https://www.apa.org/ptsd-guideline/treatments/
28. Kip KE, Elk CA, Sullivan KL, et al. Brief treatment of symptoms of post-traumatic stress disorder (PTSD) by use of accelerated resolution therapy (ART(®)). Behav Sci (Basel). 2012;2(2):115-134.
29. Steckler T, Risbrough V. Pharmacological treatment of PTSD - established and new approaches. Neuropharmacology. 2012;62(2):617-627.
30. Davidson JR, Connor KM, Hertzberg MA, et al. Maintenance therapy with fluoxetine in posttraumatic stress disorder: a placebo-controlled discontinuation study. J Clin Psychopharmacol. 2005;25(2):166-169.
31. Benedek DM, Friedman MJ, Zatzick D, et al. Guideline watch (March 2009): Practice guideline for the treatment of patients with acute stress disorder and posttraumatic stress disorder. Focus. 2009;7(2):204-213.
32. Aerni A, Traber R, Hock C, et al. Low-dose cortisol for symptoms of posttraumatic stress disorder. Am J Psychiat. 2004;161(8):1488-1490.
33. McGhee LL, Maani CV, Garza TH, et al. The correlation between ketamine and posttraumatic stress disorder in burned service members. J Trauma. 2008;64(2 suppl):S195-S198. doi: 10.1097/TA.0b013e318160ba1d
















