User login
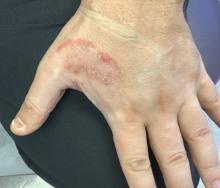
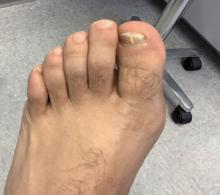
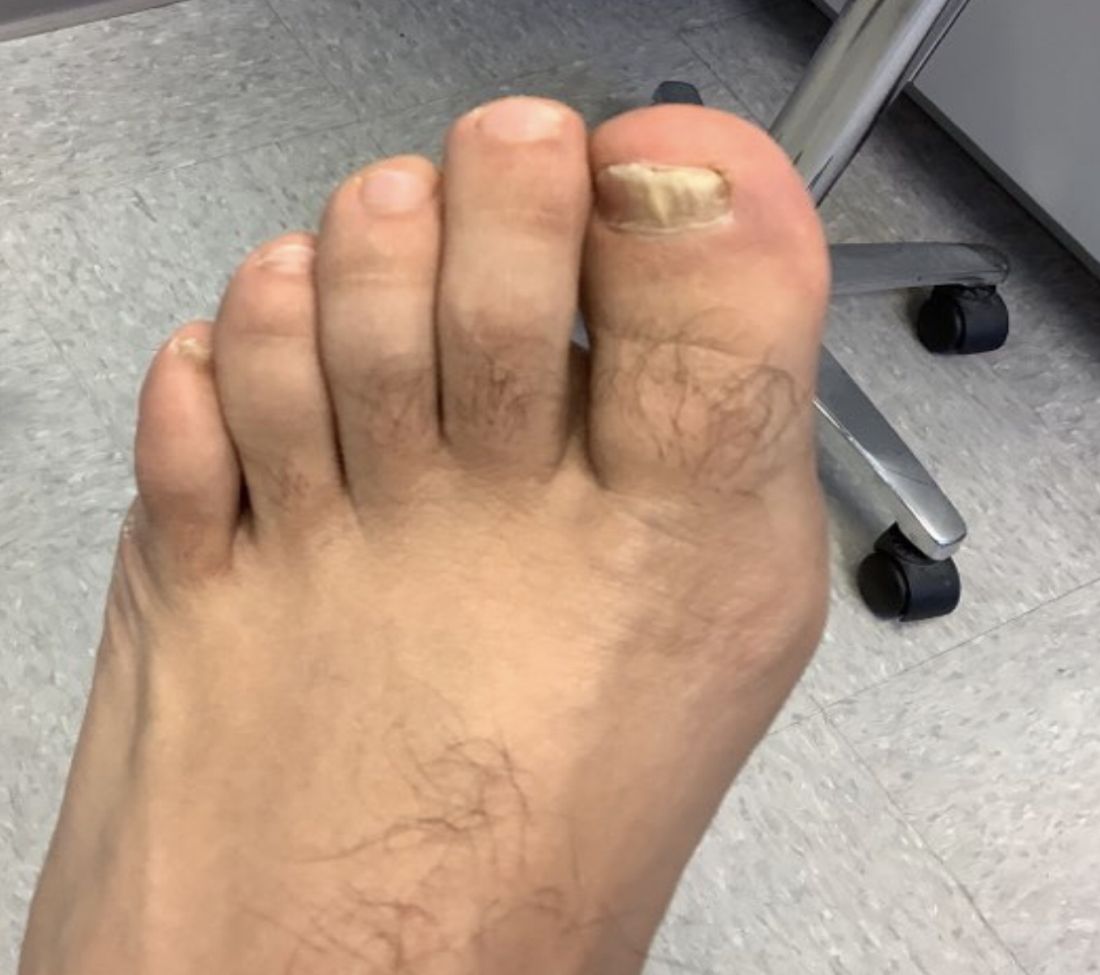
White male presents with pruritic, scaly, erythematous patches on his feet and left hand
Two feet–one hand syndrome
This condition, also known as ringworm, is a fungal infection caused by a dermatophyte, and presents as a superficial annular or circular rash with a raised, scaly border.
Symptoms include dryness and itchiness, and the lesions may appear red-pink on lighter skin and gray-brown on darker skin types. Although these infections can arise in a variety of combinations, two feet–one hand syndrome occurs in about 60% of cases. Trichophyton rubrum is the most common agent.
Diagnosis is made by patient history, dermoscopic visualization, and staining of skin scraping with KOH or fungal culture. Dermatophytes prefer moist, warm environments, so this disease is prevalent in tropical conditions and associated with moist public areas such as locker rooms and showers. As a result, tinea pedis is also nicknamed “athlete’s foot” for its common presentation in athletes. The fungus spreads easily through contact and can survive on infected surfaces, so patients often self-inoculate by touching/scratching the affected area then touching another body part. Cautions that should be taken to avoid transmission include not sharing personal care products, washing the area and keeping it dry, and avoiding close, humid environments.
The syndrome is highly associated with onychomycosis, which can be more difficult to treat and often requires oral antifungals. Tinea manuum is commonly misdiagnosed as hand dermatitis or eczema and treated with topical steroids, which will exacerbate or flare the tinea.
Two feet–one hand syndrome can typically be treated with over-the-counter topical antifungal medications such as miconazole or clotrimazole. Topical ketoconazole may be prescribed, and oral terbinafine or itraconazole are used in more severe cases when a larger body surface area is affected or in immunocompromised patients.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Davie, Fla.; Kiran C. Patel, Tampa Bay Regional Campus; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cleveland Clinic. Tinea manuum: Symptoms, causes & treatment. 2022. https://my.clevelandclinic.org/health/diseases/24063-tinea-manuum.
Ugalde-Trejo NX et al. Curr Fungal Infect Rep. 2022 Nov 17. doi: 10.1007/s12281-022-00447-9.
Mizumoto J. Cureus. 2021 Dec 27;13(12):e20758.
Two feet–one hand syndrome
This condition, also known as ringworm, is a fungal infection caused by a dermatophyte, and presents as a superficial annular or circular rash with a raised, scaly border.
Symptoms include dryness and itchiness, and the lesions may appear red-pink on lighter skin and gray-brown on darker skin types. Although these infections can arise in a variety of combinations, two feet–one hand syndrome occurs in about 60% of cases. Trichophyton rubrum is the most common agent.
Diagnosis is made by patient history, dermoscopic visualization, and staining of skin scraping with KOH or fungal culture. Dermatophytes prefer moist, warm environments, so this disease is prevalent in tropical conditions and associated with moist public areas such as locker rooms and showers. As a result, tinea pedis is also nicknamed “athlete’s foot” for its common presentation in athletes. The fungus spreads easily through contact and can survive on infected surfaces, so patients often self-inoculate by touching/scratching the affected area then touching another body part. Cautions that should be taken to avoid transmission include not sharing personal care products, washing the area and keeping it dry, and avoiding close, humid environments.
The syndrome is highly associated with onychomycosis, which can be more difficult to treat and often requires oral antifungals. Tinea manuum is commonly misdiagnosed as hand dermatitis or eczema and treated with topical steroids, which will exacerbate or flare the tinea.
Two feet–one hand syndrome can typically be treated with over-the-counter topical antifungal medications such as miconazole or clotrimazole. Topical ketoconazole may be prescribed, and oral terbinafine or itraconazole are used in more severe cases when a larger body surface area is affected or in immunocompromised patients.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Davie, Fla.; Kiran C. Patel, Tampa Bay Regional Campus; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cleveland Clinic. Tinea manuum: Symptoms, causes & treatment. 2022. https://my.clevelandclinic.org/health/diseases/24063-tinea-manuum.
Ugalde-Trejo NX et al. Curr Fungal Infect Rep. 2022 Nov 17. doi: 10.1007/s12281-022-00447-9.
Mizumoto J. Cureus. 2021 Dec 27;13(12):e20758.
Two feet–one hand syndrome
This condition, also known as ringworm, is a fungal infection caused by a dermatophyte, and presents as a superficial annular or circular rash with a raised, scaly border.
Symptoms include dryness and itchiness, and the lesions may appear red-pink on lighter skin and gray-brown on darker skin types. Although these infections can arise in a variety of combinations, two feet–one hand syndrome occurs in about 60% of cases. Trichophyton rubrum is the most common agent.
Diagnosis is made by patient history, dermoscopic visualization, and staining of skin scraping with KOH or fungal culture. Dermatophytes prefer moist, warm environments, so this disease is prevalent in tropical conditions and associated with moist public areas such as locker rooms and showers. As a result, tinea pedis is also nicknamed “athlete’s foot” for its common presentation in athletes. The fungus spreads easily through contact and can survive on infected surfaces, so patients often self-inoculate by touching/scratching the affected area then touching another body part. Cautions that should be taken to avoid transmission include not sharing personal care products, washing the area and keeping it dry, and avoiding close, humid environments.
The syndrome is highly associated with onychomycosis, which can be more difficult to treat and often requires oral antifungals. Tinea manuum is commonly misdiagnosed as hand dermatitis or eczema and treated with topical steroids, which will exacerbate or flare the tinea.
Two feet–one hand syndrome can typically be treated with over-the-counter topical antifungal medications such as miconazole or clotrimazole. Topical ketoconazole may be prescribed, and oral terbinafine or itraconazole are used in more severe cases when a larger body surface area is affected or in immunocompromised patients.
This case and photo were submitted by Lucas Shapiro, BS, Nova Southeastern University, Davie, Fla.; Kiran C. Patel, Tampa Bay Regional Campus; and Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cleveland Clinic. Tinea manuum: Symptoms, causes & treatment. 2022. https://my.clevelandclinic.org/health/diseases/24063-tinea-manuum.
Ugalde-Trejo NX et al. Curr Fungal Infect Rep. 2022 Nov 17. doi: 10.1007/s12281-022-00447-9.
Mizumoto J. Cureus. 2021 Dec 27;13(12):e20758.
What’s it like to take Ozempic? A doctor’s own story
With the rising popularity of weight-loss drug injections, I’ve received many questions from patients about the pros, cons, and costs. While Ozempic (semaglutide) is perhaps the best known, it’s technically an agent approved only for type 2 diabetes that has been used off label for obesity. The same substance, semaglutide, is approved for use in obesity, but at a higher dose, under the brand name Wegovy. Alternatives are available, and results will vary depending on the specific agent used and the individual.
Ultimately, I decided to try these new injections for myself. I am not a paid representative for, nor an advocate of, any of these medications; I’m here only to share my personal experience.
In my discussions with patients about weight, I sometimes felt like an imposter. While I was overweight by medical standards, I fortunately had none of the underlying health problems. I wasn’t on medications for blood pressure nor did I have diabetes, but I was counseling people to lose weight and eat better while not always following my own advice.
Since having children and turning 40, my metabolism, like many other women’s, seems to have plummeted. I tried a number of older weight-loss medications, like phentermine and phendimetrazine, under the supervision of medical professionals.
Each time, the efforts worked for a short while, particularly when I followed good portion control and practiced moderate exercise. Once the side effects (that is, tachycardia, palpitations, mood changes, constipation) became intolerable, or I became tired or fearful of being on the medications too long, I’d stop and I would regain some of the weight.
When the newer subcutaneous injectable medications arrived on the scene and I started to talk to my patients about them, I was intrigued by their novel mode of action and seeming benefits.
These medications, glucagonlike peptide–1 (GLP-1) receptor agonists, were first approved for type 2 diabetes, and it soon became apparent that patients were losing significant amounts of weight taking them, so manufacturers conducted further trials in obesity patients without type 2 diabetes.
The first of these, liraglutide, is injected daily and was first approved as Victoza for type 2 diabetes; it later received an additional approval for obesity, in December 2014, as Saxenda.
Semaglutide, another of the new GLP-1 agonists, was first approved for type 2 diabetes as Ozempic but again was found to lead to substantial weight loss, so a subsequent approval of the drug for obesity, as Wegovy, came in June 2021. Semaglutide is injected once a week.
Semaglutide was branded a “game changer” when it was licensed for obesity because the mean weight loss seen in trials was around 15%, more than for any other drug and approaching what could be achieved with bariatric surgery, some doctors said.
These medications work in a different way from the older weight loss drugs, which had focused on the use of amphetamines. The newer medications became very popular because treating obesity helps lower blood glucose, blood pressure, cholesterol, kidney disease risk, and other comorbidities that occur with diabetes. Plus, for most people, there were fewer side effects.
I first tried Saxenda when it arrived on the market, via some samples that our pharmaceutical representative brought, both out of curiosity and to see if it would help me lose the stubborn baby weight. I ended up stopping the daily injections after my second or third week because of nausea and vomiting. I took a break, got a prescription for antinausea medicine, and tried again because it did indeed decrease my appetite. However, when I took my prescription to the pharmacy, my insurance wouldn’t cover it. It happens to doctors, too.
Fast-forward to 2017-2018. The baby weight was still holding on despite lifestyle changes, diet, and exercising. The newer drug classes hit the market, and again we had samples from our reps.
When Ozempic was on backorder, I switched to a low dose of Mounjaro (tirzepatide), a new dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, approved for type 2 diabetes in May 2022, again using it off label as a weekly injection, as it isn’t currently approved for weight loss. However, it does produce significant weight loss and is awaiting approval for obesity.
With these new medications, I noticed that both my patients and I didn’t complain as much about nausea and vomiting, but I did experience stomach upset, constipation, and acid reflux.
The appetite suppression is effective. It slows down the emptying of the gut so I feel full longer. I’ve lost 30 lb with these weekly injections and would like to lose another 20 lb. I follow a routine of reasonable, portion-controlled eating and moderate exercise (30 minutes of cardiovascular activity at least two to three times a week).
Discontinuing the medications may cause rebound weight gain, especially if I’m no longer following a routine of healthy eating and/or moderate exercise. I deal with minimal constipation by taking stool softeners, and I take antacids for acid reflux.
Here’s what I recommend applying when working with patients who have obesity: First, explain how these medications work. Then conduct a health history to make sure these injections are right for them. Patients with a family history of pancreatic cancer can’t take these medications. You also want to monitor use in patients with a history of hypoglycemia so their blood sugar doesn’t drop too low. It’s also important to make sure your patients are able to afford the medication. My husband takes Ozempic for diabetes, and recently we were told that a refill would cost about $1,500 a month, even with insurance. “Covered” doesn’t necessarily mean affordable.
Take a baseline hemoglobin A1c and repeat it after the patient has been on the medication for 2-3 weeks. Also remind them that they can’t rely solely on the medication but need to practice portion control and healthier eating and to exercise more.
For myself, I want to lose those remaining 20 lb or so by eating healthy and being physically active without having to rely on medication for the rest of my life. Research on these medications is still early so we don’t know the long-term effects yet.
As clinicians, I feel it’s okay to be honest with our patients about our own personal struggles to help them understand that they are not alone and that losing weight is a challenge for everyone.
Dr. Swiner is a family physician in Durham, N.C. She reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
With the rising popularity of weight-loss drug injections, I’ve received many questions from patients about the pros, cons, and costs. While Ozempic (semaglutide) is perhaps the best known, it’s technically an agent approved only for type 2 diabetes that has been used off label for obesity. The same substance, semaglutide, is approved for use in obesity, but at a higher dose, under the brand name Wegovy. Alternatives are available, and results will vary depending on the specific agent used and the individual.
Ultimately, I decided to try these new injections for myself. I am not a paid representative for, nor an advocate of, any of these medications; I’m here only to share my personal experience.
In my discussions with patients about weight, I sometimes felt like an imposter. While I was overweight by medical standards, I fortunately had none of the underlying health problems. I wasn’t on medications for blood pressure nor did I have diabetes, but I was counseling people to lose weight and eat better while not always following my own advice.
Since having children and turning 40, my metabolism, like many other women’s, seems to have plummeted. I tried a number of older weight-loss medications, like phentermine and phendimetrazine, under the supervision of medical professionals.
Each time, the efforts worked for a short while, particularly when I followed good portion control and practiced moderate exercise. Once the side effects (that is, tachycardia, palpitations, mood changes, constipation) became intolerable, or I became tired or fearful of being on the medications too long, I’d stop and I would regain some of the weight.
When the newer subcutaneous injectable medications arrived on the scene and I started to talk to my patients about them, I was intrigued by their novel mode of action and seeming benefits.
These medications, glucagonlike peptide–1 (GLP-1) receptor agonists, were first approved for type 2 diabetes, and it soon became apparent that patients were losing significant amounts of weight taking them, so manufacturers conducted further trials in obesity patients without type 2 diabetes.
The first of these, liraglutide, is injected daily and was first approved as Victoza for type 2 diabetes; it later received an additional approval for obesity, in December 2014, as Saxenda.
Semaglutide, another of the new GLP-1 agonists, was first approved for type 2 diabetes as Ozempic but again was found to lead to substantial weight loss, so a subsequent approval of the drug for obesity, as Wegovy, came in June 2021. Semaglutide is injected once a week.
Semaglutide was branded a “game changer” when it was licensed for obesity because the mean weight loss seen in trials was around 15%, more than for any other drug and approaching what could be achieved with bariatric surgery, some doctors said.
These medications work in a different way from the older weight loss drugs, which had focused on the use of amphetamines. The newer medications became very popular because treating obesity helps lower blood glucose, blood pressure, cholesterol, kidney disease risk, and other comorbidities that occur with diabetes. Plus, for most people, there were fewer side effects.
I first tried Saxenda when it arrived on the market, via some samples that our pharmaceutical representative brought, both out of curiosity and to see if it would help me lose the stubborn baby weight. I ended up stopping the daily injections after my second or third week because of nausea and vomiting. I took a break, got a prescription for antinausea medicine, and tried again because it did indeed decrease my appetite. However, when I took my prescription to the pharmacy, my insurance wouldn’t cover it. It happens to doctors, too.
Fast-forward to 2017-2018. The baby weight was still holding on despite lifestyle changes, diet, and exercising. The newer drug classes hit the market, and again we had samples from our reps.
When Ozempic was on backorder, I switched to a low dose of Mounjaro (tirzepatide), a new dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, approved for type 2 diabetes in May 2022, again using it off label as a weekly injection, as it isn’t currently approved for weight loss. However, it does produce significant weight loss and is awaiting approval for obesity.
With these new medications, I noticed that both my patients and I didn’t complain as much about nausea and vomiting, but I did experience stomach upset, constipation, and acid reflux.
The appetite suppression is effective. It slows down the emptying of the gut so I feel full longer. I’ve lost 30 lb with these weekly injections and would like to lose another 20 lb. I follow a routine of reasonable, portion-controlled eating and moderate exercise (30 minutes of cardiovascular activity at least two to three times a week).
Discontinuing the medications may cause rebound weight gain, especially if I’m no longer following a routine of healthy eating and/or moderate exercise. I deal with minimal constipation by taking stool softeners, and I take antacids for acid reflux.
Here’s what I recommend applying when working with patients who have obesity: First, explain how these medications work. Then conduct a health history to make sure these injections are right for them. Patients with a family history of pancreatic cancer can’t take these medications. You also want to monitor use in patients with a history of hypoglycemia so their blood sugar doesn’t drop too low. It’s also important to make sure your patients are able to afford the medication. My husband takes Ozempic for diabetes, and recently we were told that a refill would cost about $1,500 a month, even with insurance. “Covered” doesn’t necessarily mean affordable.
Take a baseline hemoglobin A1c and repeat it after the patient has been on the medication for 2-3 weeks. Also remind them that they can’t rely solely on the medication but need to practice portion control and healthier eating and to exercise more.
For myself, I want to lose those remaining 20 lb or so by eating healthy and being physically active without having to rely on medication for the rest of my life. Research on these medications is still early so we don’t know the long-term effects yet.
As clinicians, I feel it’s okay to be honest with our patients about our own personal struggles to help them understand that they are not alone and that losing weight is a challenge for everyone.
Dr. Swiner is a family physician in Durham, N.C. She reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
With the rising popularity of weight-loss drug injections, I’ve received many questions from patients about the pros, cons, and costs. While Ozempic (semaglutide) is perhaps the best known, it’s technically an agent approved only for type 2 diabetes that has been used off label for obesity. The same substance, semaglutide, is approved for use in obesity, but at a higher dose, under the brand name Wegovy. Alternatives are available, and results will vary depending on the specific agent used and the individual.
Ultimately, I decided to try these new injections for myself. I am not a paid representative for, nor an advocate of, any of these medications; I’m here only to share my personal experience.
In my discussions with patients about weight, I sometimes felt like an imposter. While I was overweight by medical standards, I fortunately had none of the underlying health problems. I wasn’t on medications for blood pressure nor did I have diabetes, but I was counseling people to lose weight and eat better while not always following my own advice.
Since having children and turning 40, my metabolism, like many other women’s, seems to have plummeted. I tried a number of older weight-loss medications, like phentermine and phendimetrazine, under the supervision of medical professionals.
Each time, the efforts worked for a short while, particularly when I followed good portion control and practiced moderate exercise. Once the side effects (that is, tachycardia, palpitations, mood changes, constipation) became intolerable, or I became tired or fearful of being on the medications too long, I’d stop and I would regain some of the weight.
When the newer subcutaneous injectable medications arrived on the scene and I started to talk to my patients about them, I was intrigued by their novel mode of action and seeming benefits.
These medications, glucagonlike peptide–1 (GLP-1) receptor agonists, were first approved for type 2 diabetes, and it soon became apparent that patients were losing significant amounts of weight taking them, so manufacturers conducted further trials in obesity patients without type 2 diabetes.
The first of these, liraglutide, is injected daily and was first approved as Victoza for type 2 diabetes; it later received an additional approval for obesity, in December 2014, as Saxenda.
Semaglutide, another of the new GLP-1 agonists, was first approved for type 2 diabetes as Ozempic but again was found to lead to substantial weight loss, so a subsequent approval of the drug for obesity, as Wegovy, came in June 2021. Semaglutide is injected once a week.
Semaglutide was branded a “game changer” when it was licensed for obesity because the mean weight loss seen in trials was around 15%, more than for any other drug and approaching what could be achieved with bariatric surgery, some doctors said.
These medications work in a different way from the older weight loss drugs, which had focused on the use of amphetamines. The newer medications became very popular because treating obesity helps lower blood glucose, blood pressure, cholesterol, kidney disease risk, and other comorbidities that occur with diabetes. Plus, for most people, there were fewer side effects.
I first tried Saxenda when it arrived on the market, via some samples that our pharmaceutical representative brought, both out of curiosity and to see if it would help me lose the stubborn baby weight. I ended up stopping the daily injections after my second or third week because of nausea and vomiting. I took a break, got a prescription for antinausea medicine, and tried again because it did indeed decrease my appetite. However, when I took my prescription to the pharmacy, my insurance wouldn’t cover it. It happens to doctors, too.
Fast-forward to 2017-2018. The baby weight was still holding on despite lifestyle changes, diet, and exercising. The newer drug classes hit the market, and again we had samples from our reps.
When Ozempic was on backorder, I switched to a low dose of Mounjaro (tirzepatide), a new dual GLP-1 and glucose-dependent insulinotropic polypeptide agonist, approved for type 2 diabetes in May 2022, again using it off label as a weekly injection, as it isn’t currently approved for weight loss. However, it does produce significant weight loss and is awaiting approval for obesity.
With these new medications, I noticed that both my patients and I didn’t complain as much about nausea and vomiting, but I did experience stomach upset, constipation, and acid reflux.
The appetite suppression is effective. It slows down the emptying of the gut so I feel full longer. I’ve lost 30 lb with these weekly injections and would like to lose another 20 lb. I follow a routine of reasonable, portion-controlled eating and moderate exercise (30 minutes of cardiovascular activity at least two to three times a week).
Discontinuing the medications may cause rebound weight gain, especially if I’m no longer following a routine of healthy eating and/or moderate exercise. I deal with minimal constipation by taking stool softeners, and I take antacids for acid reflux.
Here’s what I recommend applying when working with patients who have obesity: First, explain how these medications work. Then conduct a health history to make sure these injections are right for them. Patients with a family history of pancreatic cancer can’t take these medications. You also want to monitor use in patients with a history of hypoglycemia so their blood sugar doesn’t drop too low. It’s also important to make sure your patients are able to afford the medication. My husband takes Ozempic for diabetes, and recently we were told that a refill would cost about $1,500 a month, even with insurance. “Covered” doesn’t necessarily mean affordable.
Take a baseline hemoglobin A1c and repeat it after the patient has been on the medication for 2-3 weeks. Also remind them that they can’t rely solely on the medication but need to practice portion control and healthier eating and to exercise more.
For myself, I want to lose those remaining 20 lb or so by eating healthy and being physically active without having to rely on medication for the rest of my life. Research on these medications is still early so we don’t know the long-term effects yet.
As clinicians, I feel it’s okay to be honest with our patients about our own personal struggles to help them understand that they are not alone and that losing weight is a challenge for everyone.
Dr. Swiner is a family physician in Durham, N.C. She reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
What do high BUN/Cr ratios mean?
He has been in good health with the only medical problem in his history being depression.
He is taking sertraline. On exam, his blood pressure is 100/60, and his pulse is 100, both while lying down. His blood pressure while standing is 90/60 and his pulse while standing is 130. The rest of his exam is normal. His lab values include hemoglobin of 10, hematocrit of 30, white blood cell of 4.6, platelet count of 175,000, sodium of 142, chloride of 100, bicarbonate of 24, potassium of 3.8, blood urea nitrogen (BUN) of 38, and creatinine clearance (Cr) of 1.1.
What is the most likely source of his bleeding?
A. Gastric ulcer
B. Meckel’s diverticulum
C. Arteriovenous malformation
D. Diverticulosis
E. Hemorrhoids
What makes the most sense
The most likely cause of this patient’s maroon stool is an upper gastrointestinal bleed, so it would make the most sense for a gastric ulcer to be the source of his bleeding. The clue here is the very high BUN/Cr ratio.
We were all taught early in our training that a high BUN/Cr ratio represented volume depletion. This is certainly the most common cause, but very high BUN/Cr ratios (over 30) can represent causes beyond volume depletion.
Witting and colleagues studied factors that predicted upper GI bleeding in patients presenting without hematemesis. They found that the three strongest predictors were black stool (odds ratio, 16.6), BUN/Cr ratio greater than 30 (OR, 10), and age greater than 50 (OR, 8.4).1
Srygley and colleagues reviewed high-quality studies of factors associated with upper GI bleeding.2 Factors that were found to increase the likelihood of an upper gastrointestinal bleed were Melenic stool on exam (likelihood ratio, 25), blood or coffee grounds on nasogastric aspiration (LR, 9.6), and BUN/Cr ratio greater than 30 (LR, 7.5).
Very high BUN/Cr ratios can indicate problems other than UGI bleeding and volume depletion. High BUN/Cr ratios are seen in patients with heart failure.
Zhang and colleagues studied if a high BUN/Cr ratio helped distinguish heart failure from asthma and chronic obstructive pulmonary disease (COPD).3 They found that, compared with those in the asthma group, the BUN/Cr ratios were significantly increased in the heart failure group (P < .05), whereas no significant differences in BUN/Cr ratios were found between the asthma and COPD groups.
Cheang and colleagues conducted their own study, as well as a meta-analysis, looking to see if high BUN/Cr ratios predicted increased mortality in patients with acute heart failure.4 In the meta-analysis of 8 studies (including their own), they found that the highest BUN/Cr ratio category was associated with an 77% higher all-cause mortality than the lowest category (hazard ratio, 1.77; 95% confidence interval, 1.52-2.07).
High dose corticosteroids can raise BUN levels, especially in patients with chronic kidney disease, and cause unexpectedly high BUN/Cr ratios.
Pearl
Very high BUN/Cr ratios (greater than 30) can signify upper GI bleeding, heart failure, or high-dose corticosteroid use.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Am J Emerg Med. 2006 May;24(3):280-5.
2. JAMA. 2012;307(10):1072-9.
3. Comput Math Methods Med. 2022 Jul 21. doi: 10.1155/2022/4586458.
4. Cardiorenal Med. 2020;10:415-28.
He has been in good health with the only medical problem in his history being depression.
He is taking sertraline. On exam, his blood pressure is 100/60, and his pulse is 100, both while lying down. His blood pressure while standing is 90/60 and his pulse while standing is 130. The rest of his exam is normal. His lab values include hemoglobin of 10, hematocrit of 30, white blood cell of 4.6, platelet count of 175,000, sodium of 142, chloride of 100, bicarbonate of 24, potassium of 3.8, blood urea nitrogen (BUN) of 38, and creatinine clearance (Cr) of 1.1.
What is the most likely source of his bleeding?
A. Gastric ulcer
B. Meckel’s diverticulum
C. Arteriovenous malformation
D. Diverticulosis
E. Hemorrhoids
What makes the most sense
The most likely cause of this patient’s maroon stool is an upper gastrointestinal bleed, so it would make the most sense for a gastric ulcer to be the source of his bleeding. The clue here is the very high BUN/Cr ratio.
We were all taught early in our training that a high BUN/Cr ratio represented volume depletion. This is certainly the most common cause, but very high BUN/Cr ratios (over 30) can represent causes beyond volume depletion.
Witting and colleagues studied factors that predicted upper GI bleeding in patients presenting without hematemesis. They found that the three strongest predictors were black stool (odds ratio, 16.6), BUN/Cr ratio greater than 30 (OR, 10), and age greater than 50 (OR, 8.4).1
Srygley and colleagues reviewed high-quality studies of factors associated with upper GI bleeding.2 Factors that were found to increase the likelihood of an upper gastrointestinal bleed were Melenic stool on exam (likelihood ratio, 25), blood or coffee grounds on nasogastric aspiration (LR, 9.6), and BUN/Cr ratio greater than 30 (LR, 7.5).
Very high BUN/Cr ratios can indicate problems other than UGI bleeding and volume depletion. High BUN/Cr ratios are seen in patients with heart failure.
Zhang and colleagues studied if a high BUN/Cr ratio helped distinguish heart failure from asthma and chronic obstructive pulmonary disease (COPD).3 They found that, compared with those in the asthma group, the BUN/Cr ratios were significantly increased in the heart failure group (P < .05), whereas no significant differences in BUN/Cr ratios were found between the asthma and COPD groups.
Cheang and colleagues conducted their own study, as well as a meta-analysis, looking to see if high BUN/Cr ratios predicted increased mortality in patients with acute heart failure.4 In the meta-analysis of 8 studies (including their own), they found that the highest BUN/Cr ratio category was associated with an 77% higher all-cause mortality than the lowest category (hazard ratio, 1.77; 95% confidence interval, 1.52-2.07).
High dose corticosteroids can raise BUN levels, especially in patients with chronic kidney disease, and cause unexpectedly high BUN/Cr ratios.
Pearl
Very high BUN/Cr ratios (greater than 30) can signify upper GI bleeding, heart failure, or high-dose corticosteroid use.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Am J Emerg Med. 2006 May;24(3):280-5.
2. JAMA. 2012;307(10):1072-9.
3. Comput Math Methods Med. 2022 Jul 21. doi: 10.1155/2022/4586458.
4. Cardiorenal Med. 2020;10:415-28.
He has been in good health with the only medical problem in his history being depression.
He is taking sertraline. On exam, his blood pressure is 100/60, and his pulse is 100, both while lying down. His blood pressure while standing is 90/60 and his pulse while standing is 130. The rest of his exam is normal. His lab values include hemoglobin of 10, hematocrit of 30, white blood cell of 4.6, platelet count of 175,000, sodium of 142, chloride of 100, bicarbonate of 24, potassium of 3.8, blood urea nitrogen (BUN) of 38, and creatinine clearance (Cr) of 1.1.
What is the most likely source of his bleeding?
A. Gastric ulcer
B. Meckel’s diverticulum
C. Arteriovenous malformation
D. Diverticulosis
E. Hemorrhoids
What makes the most sense
The most likely cause of this patient’s maroon stool is an upper gastrointestinal bleed, so it would make the most sense for a gastric ulcer to be the source of his bleeding. The clue here is the very high BUN/Cr ratio.
We were all taught early in our training that a high BUN/Cr ratio represented volume depletion. This is certainly the most common cause, but very high BUN/Cr ratios (over 30) can represent causes beyond volume depletion.
Witting and colleagues studied factors that predicted upper GI bleeding in patients presenting without hematemesis. They found that the three strongest predictors were black stool (odds ratio, 16.6), BUN/Cr ratio greater than 30 (OR, 10), and age greater than 50 (OR, 8.4).1
Srygley and colleagues reviewed high-quality studies of factors associated with upper GI bleeding.2 Factors that were found to increase the likelihood of an upper gastrointestinal bleed were Melenic stool on exam (likelihood ratio, 25), blood or coffee grounds on nasogastric aspiration (LR, 9.6), and BUN/Cr ratio greater than 30 (LR, 7.5).
Very high BUN/Cr ratios can indicate problems other than UGI bleeding and volume depletion. High BUN/Cr ratios are seen in patients with heart failure.
Zhang and colleagues studied if a high BUN/Cr ratio helped distinguish heart failure from asthma and chronic obstructive pulmonary disease (COPD).3 They found that, compared with those in the asthma group, the BUN/Cr ratios were significantly increased in the heart failure group (P < .05), whereas no significant differences in BUN/Cr ratios were found between the asthma and COPD groups.
Cheang and colleagues conducted their own study, as well as a meta-analysis, looking to see if high BUN/Cr ratios predicted increased mortality in patients with acute heart failure.4 In the meta-analysis of 8 studies (including their own), they found that the highest BUN/Cr ratio category was associated with an 77% higher all-cause mortality than the lowest category (hazard ratio, 1.77; 95% confidence interval, 1.52-2.07).
High dose corticosteroids can raise BUN levels, especially in patients with chronic kidney disease, and cause unexpectedly high BUN/Cr ratios.
Pearl
Very high BUN/Cr ratios (greater than 30) can signify upper GI bleeding, heart failure, or high-dose corticosteroid use.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. Contact Dr. Paauw at [email protected].
References
1. Am J Emerg Med. 2006 May;24(3):280-5.
2. JAMA. 2012;307(10):1072-9.
3. Comput Math Methods Med. 2022 Jul 21. doi: 10.1155/2022/4586458.
4. Cardiorenal Med. 2020;10:415-28.
What impact do carbs have on bone health?
I am often asked about the impact of dietary nutrients on bone health, particularly as many patients with low bone density, many with a history of multiple fractures, are referred to me. Many factors affect bone density, an important predictor of fracture risk, including genetics, body weight and muscle mass, bone loading exercise, menstrual status, other hormonal factors, nutritional status, optimal absorption of dietary nutrients, and medication use.
Dietary nutrients include macronutrients (carbohydrates, proteins, fat, and fiber) and micronutrients (such as dietary minerals and vitamins). The importance of micronutrients such as calcium, phosphorus, magnesium, and vitamins C, D, and K in optimizing bone mineralization and bone formation has been well documented.
The impact of protein intake on bone health is slightly more controversial, with some studies suggesting that increased protein intake may be deleterious to bone by increasing acid load, which in turn, increases calcium loss in urine. Overall data analysis from multiple studies support the finding that a higher protein intake is modestly beneficial for bone at certain sites, such as the spine.
Though data regarding the impact of dietary carbohydrates on bone are not as robust, it’s important to understand these effects given the increasing knowledge of the deleterious impact of processed carbohydrates on weight and cardiometabolic outcomes. This leads to the growing recommendations to limit carbohydrates in diet.
Quality and quantity of carbs affect bone health
Available studies suggest that both the quality and quantity of carbohydrates that are in a diet as well as the glycemic index of food may affect bone outcomes. Glycemic index refers to the extent of blood glucose elevation that occurs after the intake of any specific food. Foods with a higher glycemic index cause a rapid increase in blood glucose, whereas those with a low glycemic index result in a slower and more gradual increase. Examples of high–glycemic index food include processed and baked foods (such as breakfast cereals [unless whole grain], pretzels, cookies, doughnuts, pastries, cake, white bread, bagels, croissants, and corn chips), sugar-sweetened beverages, white rice, fast food (such as pizza and burgers), and potatoes. Examples of low glycemic index foods include vegetables, fruits, legumes, dairy and dairy products (without added sugar), whole-grain foods (such as oat porridge), and nuts.
A high–glycemic index diet has been associated with a greater risk for obesity and cardiovascular disease, and with lower bone density, an increased risk for fracture. This has been attributed to acute increases in glucose and insulin levels after consumption of high–glycemic index food, which causes increased oxidative stress and secretion of inflammatory cytokines, such as interleukin 6 and tumor necrosis factor alpha, that activate cells in bone that increase bone loss.
Higher blood glucose concentrations induced by a higher dietary glycemic index can have deleterious effects on osteoblasts, the cells important for bone formation, and increase bone loss through production of advanced glycation end products that affect the cross linking of collagen in bone (important for bone strength), as well as calcium loss in urine. This was recently reported in a study by Garcia-Gavilan and others, in which the authors showed that high dietary glycemic index and dietary glucose load are associated with a higher risk for osteoporosis-related fractures in an older Mediterranean population who are at high risk for cardiovascular events. Similar data were reported by Nouri and coauthors in a study from Iran.
The quantity and quality of dietary carbohydrates may also have an impact on bone. The quality of carbohydrates has been assessed using the carbohydrate quality index (CQI) and the low carbohydrate diet score (LCDS). The CQI takes into account dietary fiber intake, glycemic index, intake of processed vs. whole grain, and solid vs. total carbohydrates in diet. A higher CQI diet is associated with reduced cardiovascular risk. Higher LCDS reflects lower carbohydrate and higher fat and protein intake.
Diets that are rich in refined or processed carbohydrates with added sugar are proinflammatory and increase oxidative stress, which may lead to increased bone loss, low bone density, and increased fracture risk. These foods also have a high glycemic index.
In contrast, diets that are rich in whole grains, legumes, fruits, vegetables, nuts, and olive oil have a lower glycemic index and are beneficial to bone. These diets have a higher CQI and LCDS (as reported by Nouri and coauthors) and provide a rich source of antioxidants, vitamins, minerals, and other nutrients (such as calcium, magnesium, and vitamins B, C, and K), which are all beneficial to bone. Gao and others have reported that implementing a low glycemic index pulse-based diet (lentils, peas, beans) is superior to a regular hospital diet in preventing the increase in bone loss that typically occurs during hospitalization with enforced bed rest.
Most reports of the impact of carbohydrates on bone health are from observational studies. In an interventional study, Dalskov and coauthors randomly assigned children aged 5-18 years who had parents with overweight to one of five diets (high protein/low glycemic index, high protein/high glycemic index, low protein/low glycemic index, low protein/high glycemic index, or regular) for 6 months.
Contrasting with our understanding that protein intake is overall good for bone, this study found that among patients receiving a high–glycemic index diet, those who were on a high-protein diet had greater reductions in a bone formation marker than did those on a low-protein diet, with no major changes observed with the other diets. This suggests the influence of associated dietary nutrients on bone outcomes and that protein intake may modify the effects of dietary carbohydrates on bone formation. Similarly, the fat content of food can alter the glycemic index and thus may modify the impact of dietary carbohydrates on bone.
In summary, available data suggest that the quantity and quality of carbohydrates, including the glycemic index of food, may affect bone health and that it is important to exercise moderation in the consumption of such foods. However, there are only a few studies that have examined these associations, and more studies are necessary to further clarify the impact of dietary carbohydrates on bone as well as any modifications of these effects by other associated food groups. These studies will allow us to refine our recommendations to our patients as we advance our understanding of the impact of the combined effects of various dietary nutrients on bone.
Madhusmita Misra, MD, MPH, is chief of the division of pediatric endocrinology, Mass General for Children, Boston, and serves or has served as a director, officer, partner, employee, advisor, consultant, or trustee for AbbVie, Sanofi, and Ipsen.
A version of this article first appeared on Medscape.com.
I am often asked about the impact of dietary nutrients on bone health, particularly as many patients with low bone density, many with a history of multiple fractures, are referred to me. Many factors affect bone density, an important predictor of fracture risk, including genetics, body weight and muscle mass, bone loading exercise, menstrual status, other hormonal factors, nutritional status, optimal absorption of dietary nutrients, and medication use.
Dietary nutrients include macronutrients (carbohydrates, proteins, fat, and fiber) and micronutrients (such as dietary minerals and vitamins). The importance of micronutrients such as calcium, phosphorus, magnesium, and vitamins C, D, and K in optimizing bone mineralization and bone formation has been well documented.
The impact of protein intake on bone health is slightly more controversial, with some studies suggesting that increased protein intake may be deleterious to bone by increasing acid load, which in turn, increases calcium loss in urine. Overall data analysis from multiple studies support the finding that a higher protein intake is modestly beneficial for bone at certain sites, such as the spine.
Though data regarding the impact of dietary carbohydrates on bone are not as robust, it’s important to understand these effects given the increasing knowledge of the deleterious impact of processed carbohydrates on weight and cardiometabolic outcomes. This leads to the growing recommendations to limit carbohydrates in diet.
Quality and quantity of carbs affect bone health
Available studies suggest that both the quality and quantity of carbohydrates that are in a diet as well as the glycemic index of food may affect bone outcomes. Glycemic index refers to the extent of blood glucose elevation that occurs after the intake of any specific food. Foods with a higher glycemic index cause a rapid increase in blood glucose, whereas those with a low glycemic index result in a slower and more gradual increase. Examples of high–glycemic index food include processed and baked foods (such as breakfast cereals [unless whole grain], pretzels, cookies, doughnuts, pastries, cake, white bread, bagels, croissants, and corn chips), sugar-sweetened beverages, white rice, fast food (such as pizza and burgers), and potatoes. Examples of low glycemic index foods include vegetables, fruits, legumes, dairy and dairy products (without added sugar), whole-grain foods (such as oat porridge), and nuts.
A high–glycemic index diet has been associated with a greater risk for obesity and cardiovascular disease, and with lower bone density, an increased risk for fracture. This has been attributed to acute increases in glucose and insulin levels after consumption of high–glycemic index food, which causes increased oxidative stress and secretion of inflammatory cytokines, such as interleukin 6 and tumor necrosis factor alpha, that activate cells in bone that increase bone loss.
Higher blood glucose concentrations induced by a higher dietary glycemic index can have deleterious effects on osteoblasts, the cells important for bone formation, and increase bone loss through production of advanced glycation end products that affect the cross linking of collagen in bone (important for bone strength), as well as calcium loss in urine. This was recently reported in a study by Garcia-Gavilan and others, in which the authors showed that high dietary glycemic index and dietary glucose load are associated with a higher risk for osteoporosis-related fractures in an older Mediterranean population who are at high risk for cardiovascular events. Similar data were reported by Nouri and coauthors in a study from Iran.
The quantity and quality of dietary carbohydrates may also have an impact on bone. The quality of carbohydrates has been assessed using the carbohydrate quality index (CQI) and the low carbohydrate diet score (LCDS). The CQI takes into account dietary fiber intake, glycemic index, intake of processed vs. whole grain, and solid vs. total carbohydrates in diet. A higher CQI diet is associated with reduced cardiovascular risk. Higher LCDS reflects lower carbohydrate and higher fat and protein intake.
Diets that are rich in refined or processed carbohydrates with added sugar are proinflammatory and increase oxidative stress, which may lead to increased bone loss, low bone density, and increased fracture risk. These foods also have a high glycemic index.
In contrast, diets that are rich in whole grains, legumes, fruits, vegetables, nuts, and olive oil have a lower glycemic index and are beneficial to bone. These diets have a higher CQI and LCDS (as reported by Nouri and coauthors) and provide a rich source of antioxidants, vitamins, minerals, and other nutrients (such as calcium, magnesium, and vitamins B, C, and K), which are all beneficial to bone. Gao and others have reported that implementing a low glycemic index pulse-based diet (lentils, peas, beans) is superior to a regular hospital diet in preventing the increase in bone loss that typically occurs during hospitalization with enforced bed rest.
Most reports of the impact of carbohydrates on bone health are from observational studies. In an interventional study, Dalskov and coauthors randomly assigned children aged 5-18 years who had parents with overweight to one of five diets (high protein/low glycemic index, high protein/high glycemic index, low protein/low glycemic index, low protein/high glycemic index, or regular) for 6 months.
Contrasting with our understanding that protein intake is overall good for bone, this study found that among patients receiving a high–glycemic index diet, those who were on a high-protein diet had greater reductions in a bone formation marker than did those on a low-protein diet, with no major changes observed with the other diets. This suggests the influence of associated dietary nutrients on bone outcomes and that protein intake may modify the effects of dietary carbohydrates on bone formation. Similarly, the fat content of food can alter the glycemic index and thus may modify the impact of dietary carbohydrates on bone.
In summary, available data suggest that the quantity and quality of carbohydrates, including the glycemic index of food, may affect bone health and that it is important to exercise moderation in the consumption of such foods. However, there are only a few studies that have examined these associations, and more studies are necessary to further clarify the impact of dietary carbohydrates on bone as well as any modifications of these effects by other associated food groups. These studies will allow us to refine our recommendations to our patients as we advance our understanding of the impact of the combined effects of various dietary nutrients on bone.
Madhusmita Misra, MD, MPH, is chief of the division of pediatric endocrinology, Mass General for Children, Boston, and serves or has served as a director, officer, partner, employee, advisor, consultant, or trustee for AbbVie, Sanofi, and Ipsen.
A version of this article first appeared on Medscape.com.
I am often asked about the impact of dietary nutrients on bone health, particularly as many patients with low bone density, many with a history of multiple fractures, are referred to me. Many factors affect bone density, an important predictor of fracture risk, including genetics, body weight and muscle mass, bone loading exercise, menstrual status, other hormonal factors, nutritional status, optimal absorption of dietary nutrients, and medication use.
Dietary nutrients include macronutrients (carbohydrates, proteins, fat, and fiber) and micronutrients (such as dietary minerals and vitamins). The importance of micronutrients such as calcium, phosphorus, magnesium, and vitamins C, D, and K in optimizing bone mineralization and bone formation has been well documented.
The impact of protein intake on bone health is slightly more controversial, with some studies suggesting that increased protein intake may be deleterious to bone by increasing acid load, which in turn, increases calcium loss in urine. Overall data analysis from multiple studies support the finding that a higher protein intake is modestly beneficial for bone at certain sites, such as the spine.
Though data regarding the impact of dietary carbohydrates on bone are not as robust, it’s important to understand these effects given the increasing knowledge of the deleterious impact of processed carbohydrates on weight and cardiometabolic outcomes. This leads to the growing recommendations to limit carbohydrates in diet.
Quality and quantity of carbs affect bone health
Available studies suggest that both the quality and quantity of carbohydrates that are in a diet as well as the glycemic index of food may affect bone outcomes. Glycemic index refers to the extent of blood glucose elevation that occurs after the intake of any specific food. Foods with a higher glycemic index cause a rapid increase in blood glucose, whereas those with a low glycemic index result in a slower and more gradual increase. Examples of high–glycemic index food include processed and baked foods (such as breakfast cereals [unless whole grain], pretzels, cookies, doughnuts, pastries, cake, white bread, bagels, croissants, and corn chips), sugar-sweetened beverages, white rice, fast food (such as pizza and burgers), and potatoes. Examples of low glycemic index foods include vegetables, fruits, legumes, dairy and dairy products (without added sugar), whole-grain foods (such as oat porridge), and nuts.
A high–glycemic index diet has been associated with a greater risk for obesity and cardiovascular disease, and with lower bone density, an increased risk for fracture. This has been attributed to acute increases in glucose and insulin levels after consumption of high–glycemic index food, which causes increased oxidative stress and secretion of inflammatory cytokines, such as interleukin 6 and tumor necrosis factor alpha, that activate cells in bone that increase bone loss.
Higher blood glucose concentrations induced by a higher dietary glycemic index can have deleterious effects on osteoblasts, the cells important for bone formation, and increase bone loss through production of advanced glycation end products that affect the cross linking of collagen in bone (important for bone strength), as well as calcium loss in urine. This was recently reported in a study by Garcia-Gavilan and others, in which the authors showed that high dietary glycemic index and dietary glucose load are associated with a higher risk for osteoporosis-related fractures in an older Mediterranean population who are at high risk for cardiovascular events. Similar data were reported by Nouri and coauthors in a study from Iran.
The quantity and quality of dietary carbohydrates may also have an impact on bone. The quality of carbohydrates has been assessed using the carbohydrate quality index (CQI) and the low carbohydrate diet score (LCDS). The CQI takes into account dietary fiber intake, glycemic index, intake of processed vs. whole grain, and solid vs. total carbohydrates in diet. A higher CQI diet is associated with reduced cardiovascular risk. Higher LCDS reflects lower carbohydrate and higher fat and protein intake.
Diets that are rich in refined or processed carbohydrates with added sugar are proinflammatory and increase oxidative stress, which may lead to increased bone loss, low bone density, and increased fracture risk. These foods also have a high glycemic index.
In contrast, diets that are rich in whole grains, legumes, fruits, vegetables, nuts, and olive oil have a lower glycemic index and are beneficial to bone. These diets have a higher CQI and LCDS (as reported by Nouri and coauthors) and provide a rich source of antioxidants, vitamins, minerals, and other nutrients (such as calcium, magnesium, and vitamins B, C, and K), which are all beneficial to bone. Gao and others have reported that implementing a low glycemic index pulse-based diet (lentils, peas, beans) is superior to a regular hospital diet in preventing the increase in bone loss that typically occurs during hospitalization with enforced bed rest.
Most reports of the impact of carbohydrates on bone health are from observational studies. In an interventional study, Dalskov and coauthors randomly assigned children aged 5-18 years who had parents with overweight to one of five diets (high protein/low glycemic index, high protein/high glycemic index, low protein/low glycemic index, low protein/high glycemic index, or regular) for 6 months.
Contrasting with our understanding that protein intake is overall good for bone, this study found that among patients receiving a high–glycemic index diet, those who were on a high-protein diet had greater reductions in a bone formation marker than did those on a low-protein diet, with no major changes observed with the other diets. This suggests the influence of associated dietary nutrients on bone outcomes and that protein intake may modify the effects of dietary carbohydrates on bone formation. Similarly, the fat content of food can alter the glycemic index and thus may modify the impact of dietary carbohydrates on bone.
In summary, available data suggest that the quantity and quality of carbohydrates, including the glycemic index of food, may affect bone health and that it is important to exercise moderation in the consumption of such foods. However, there are only a few studies that have examined these associations, and more studies are necessary to further clarify the impact of dietary carbohydrates on bone as well as any modifications of these effects by other associated food groups. These studies will allow us to refine our recommendations to our patients as we advance our understanding of the impact of the combined effects of various dietary nutrients on bone.
Madhusmita Misra, MD, MPH, is chief of the division of pediatric endocrinology, Mass General for Children, Boston, and serves or has served as a director, officer, partner, employee, advisor, consultant, or trustee for AbbVie, Sanofi, and Ipsen.
A version of this article first appeared on Medscape.com.
Meaningful improvement for patients like Tante Ilse
Last year, after a long delay due to COVID, my father’s ashes were finally laid to rest at Arlington National Cemetery. Among the loved ones who came was my favorite aunt, Tante Ilse, who was suffering from dementia. While she wasn’t “following” everything that was going on, she did perk up when she heard my father’s name and would comment on how she liked him and how wonderful he had been to her.
After the ceremony, our family of about 30 gathered at a restaurant where we shared stories and old pictures. Tante Ilse seemed to relish the photos and the time with family. She was doing so well that when we went back to my mom’s home after the reception, my cousins decided to bring Tante Ilse there, too. She had a great time, as evidenced by her famous total-body laugh. In the months before her death, we all commented about that day and how happy she seemed.
My aunt’s decline comes to mind as I reflect on media reports of 2 Alzheimer drugs— aducanumab and lecanemab—that have been billed by some as “gamechangers.” These new drugs are monoclonal antibodies directed at amyloid, one of several agents thought to cause Alzheimer disease. The details of aducanumab’s approval by the US Food and Drug Administration (FDA) generated a great deal of criticism—with good reason.
Two manufacturer-sponsored studies of aducanumab were halted due to futility of finding a benefit.1 The FDA’s scientific advisory panel recommended against approval due to a lack of evidence that it did anything more than remove amyloid plaque from the brain. And yet aducanumab received accelerated approval from the FDA. (This author collaborated on an additional analysis using data presented to the FDA, after its approval, which also reported no clinically meaningful effects.2) The other agent, lecanemab, also reduces markers of amyloid and was shown to be only moderately better than placebo in decreasing the rate of decline on various measures of cognition.3 Quite notably, both aducanumab and lecanemab, which are administered parenterally, cost more than $25,000 per year4,5 and cause amyloid-related imaging abnormalities (brain edema or hemorrhage).
Expensive agents without meaningful benefit. So far, neither of these agents has shown a reduction in things that are truly important to our patients and their families/caregivers: a reduction in caregiver burden and a reduction in the need for placement in long-term care facilities.
This is in contrast to cholinesterase inhibitors, which also slow the rate of cognitive decline.6 Among the differences that exist between these agents: Cholinesterase inhibitors are taken orally and are available as generics, which cost less than a thousand dollars per year.7 Limited data also suggest that they are associated with a lower risk for nursing home placement.8,9 (A February 2023 search of clinicaltrials.gov did not reveal any completed or planned head-to-head comparisons of monoclonal antibodies and anticholinergic agents.)
Our patients, their families, and caregivers hold out hope for something that will improve the patient’s cognition and extend the meaningful time they have with their loved ones. So far, the best we have to offer falls far short of these goals. I certainly would have hoped for something better than merely clearing amyloid for my aunt.
It’s time that the FDA adopt more rigorous standards requiring new drugs to, among other things, demonstrate meaningful clinical benefits, provide real cost savings, and be safer than currently available therapies. Other nations seem to be able to do this.10,11 It is bad enough to provide “hope in a bottle”; it is worse when what is offered is false hope.
1. Budd Haeberlein S, Aisen PS, Barkhof F, et al. Two randomized phase 3 studies of aducanumab in early Alzheimer’s disease. J Prev Alzheimers Dis. 2022;9:197-210. doi: 10.14283/jpad.2022.30
2. Ebell MH, Barry HC. Why physicians should not prescribe aducanumab for Alzheimer disease. Am Fam Physician. 2022;105:353-354.
3. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388:9-21. doi: 10.1056/NEJMoa2212948
4. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023; 613:227-228. doi: 10.1038/d41586-023-00030-3
5. Biogen announces reduced price for Aduhelm to improve access for patients with early Alzheimer’s disease. December 20, 2021. Accessed February 20, 2023. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-reduced-price-aduhelmr-improve-access-patients
6. Takramah WK, Asem L. The efficacy of pharmacological interventions to improve cognitive and behavior symptoms in people with dementia: A systematic review and meta-analysis. Health Sci Rep. 2022;5:e913. doi: 10.1002/hsr2.913
7. GoodRx. Donepezil generic Aricept. Accessed February 20, 2023. www.goodrx.com/donepezil
8. Howard R, McShane R, Lindesay J, et al. Nursing home placement in the donepezil and memantine in moderate to severe Alzheimer’s disease (DOMINO-AD) trial: secondary and post-hoc analyses. Lancet Neurol. 2015;14:1171-1181. doi: 10.1016/S1474-4422(15)00258-6
9. Geldmacher DS, Provenzano G, McRae T, et al. Donepezil is associated with delayed nursing home placement in patients with Alzheimer’s disease. J Am Geriatr Soc. 2003;51:937-944. doi: 10.1046/j.1365-2389.2003.51306.x
10. Pham C, Le K, Draves M, et al. Assessment of FDA-approved drugs not recommended for use or reimbursement in other countries, 2017-2020. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6787
11. Johnston JL, Ross JS, Ramachandran R. US Food and Drug Administration approval of drugs not meeting pivotal trial primary end points, 2018-2021. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6444
Last year, after a long delay due to COVID, my father’s ashes were finally laid to rest at Arlington National Cemetery. Among the loved ones who came was my favorite aunt, Tante Ilse, who was suffering from dementia. While she wasn’t “following” everything that was going on, she did perk up when she heard my father’s name and would comment on how she liked him and how wonderful he had been to her.
After the ceremony, our family of about 30 gathered at a restaurant where we shared stories and old pictures. Tante Ilse seemed to relish the photos and the time with family. She was doing so well that when we went back to my mom’s home after the reception, my cousins decided to bring Tante Ilse there, too. She had a great time, as evidenced by her famous total-body laugh. In the months before her death, we all commented about that day and how happy she seemed.
My aunt’s decline comes to mind as I reflect on media reports of 2 Alzheimer drugs— aducanumab and lecanemab—that have been billed by some as “gamechangers.” These new drugs are monoclonal antibodies directed at amyloid, one of several agents thought to cause Alzheimer disease. The details of aducanumab’s approval by the US Food and Drug Administration (FDA) generated a great deal of criticism—with good reason.
Two manufacturer-sponsored studies of aducanumab were halted due to futility of finding a benefit.1 The FDA’s scientific advisory panel recommended against approval due to a lack of evidence that it did anything more than remove amyloid plaque from the brain. And yet aducanumab received accelerated approval from the FDA. (This author collaborated on an additional analysis using data presented to the FDA, after its approval, which also reported no clinically meaningful effects.2) The other agent, lecanemab, also reduces markers of amyloid and was shown to be only moderately better than placebo in decreasing the rate of decline on various measures of cognition.3 Quite notably, both aducanumab and lecanemab, which are administered parenterally, cost more than $25,000 per year4,5 and cause amyloid-related imaging abnormalities (brain edema or hemorrhage).
Expensive agents without meaningful benefit. So far, neither of these agents has shown a reduction in things that are truly important to our patients and their families/caregivers: a reduction in caregiver burden and a reduction in the need for placement in long-term care facilities.
This is in contrast to cholinesterase inhibitors, which also slow the rate of cognitive decline.6 Among the differences that exist between these agents: Cholinesterase inhibitors are taken orally and are available as generics, which cost less than a thousand dollars per year.7 Limited data also suggest that they are associated with a lower risk for nursing home placement.8,9 (A February 2023 search of clinicaltrials.gov did not reveal any completed or planned head-to-head comparisons of monoclonal antibodies and anticholinergic agents.)
Our patients, their families, and caregivers hold out hope for something that will improve the patient’s cognition and extend the meaningful time they have with their loved ones. So far, the best we have to offer falls far short of these goals. I certainly would have hoped for something better than merely clearing amyloid for my aunt.
It’s time that the FDA adopt more rigorous standards requiring new drugs to, among other things, demonstrate meaningful clinical benefits, provide real cost savings, and be safer than currently available therapies. Other nations seem to be able to do this.10,11 It is bad enough to provide “hope in a bottle”; it is worse when what is offered is false hope.
Last year, after a long delay due to COVID, my father’s ashes were finally laid to rest at Arlington National Cemetery. Among the loved ones who came was my favorite aunt, Tante Ilse, who was suffering from dementia. While she wasn’t “following” everything that was going on, she did perk up when she heard my father’s name and would comment on how she liked him and how wonderful he had been to her.
After the ceremony, our family of about 30 gathered at a restaurant where we shared stories and old pictures. Tante Ilse seemed to relish the photos and the time with family. She was doing so well that when we went back to my mom’s home after the reception, my cousins decided to bring Tante Ilse there, too. She had a great time, as evidenced by her famous total-body laugh. In the months before her death, we all commented about that day and how happy she seemed.
My aunt’s decline comes to mind as I reflect on media reports of 2 Alzheimer drugs— aducanumab and lecanemab—that have been billed by some as “gamechangers.” These new drugs are monoclonal antibodies directed at amyloid, one of several agents thought to cause Alzheimer disease. The details of aducanumab’s approval by the US Food and Drug Administration (FDA) generated a great deal of criticism—with good reason.
Two manufacturer-sponsored studies of aducanumab were halted due to futility of finding a benefit.1 The FDA’s scientific advisory panel recommended against approval due to a lack of evidence that it did anything more than remove amyloid plaque from the brain. And yet aducanumab received accelerated approval from the FDA. (This author collaborated on an additional analysis using data presented to the FDA, after its approval, which also reported no clinically meaningful effects.2) The other agent, lecanemab, also reduces markers of amyloid and was shown to be only moderately better than placebo in decreasing the rate of decline on various measures of cognition.3 Quite notably, both aducanumab and lecanemab, which are administered parenterally, cost more than $25,000 per year4,5 and cause amyloid-related imaging abnormalities (brain edema or hemorrhage).
Expensive agents without meaningful benefit. So far, neither of these agents has shown a reduction in things that are truly important to our patients and their families/caregivers: a reduction in caregiver burden and a reduction in the need for placement in long-term care facilities.
This is in contrast to cholinesterase inhibitors, which also slow the rate of cognitive decline.6 Among the differences that exist between these agents: Cholinesterase inhibitors are taken orally and are available as generics, which cost less than a thousand dollars per year.7 Limited data also suggest that they are associated with a lower risk for nursing home placement.8,9 (A February 2023 search of clinicaltrials.gov did not reveal any completed or planned head-to-head comparisons of monoclonal antibodies and anticholinergic agents.)
Our patients, their families, and caregivers hold out hope for something that will improve the patient’s cognition and extend the meaningful time they have with their loved ones. So far, the best we have to offer falls far short of these goals. I certainly would have hoped for something better than merely clearing amyloid for my aunt.
It’s time that the FDA adopt more rigorous standards requiring new drugs to, among other things, demonstrate meaningful clinical benefits, provide real cost savings, and be safer than currently available therapies. Other nations seem to be able to do this.10,11 It is bad enough to provide “hope in a bottle”; it is worse when what is offered is false hope.
1. Budd Haeberlein S, Aisen PS, Barkhof F, et al. Two randomized phase 3 studies of aducanumab in early Alzheimer’s disease. J Prev Alzheimers Dis. 2022;9:197-210. doi: 10.14283/jpad.2022.30
2. Ebell MH, Barry HC. Why physicians should not prescribe aducanumab for Alzheimer disease. Am Fam Physician. 2022;105:353-354.
3. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388:9-21. doi: 10.1056/NEJMoa2212948
4. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023; 613:227-228. doi: 10.1038/d41586-023-00030-3
5. Biogen announces reduced price for Aduhelm to improve access for patients with early Alzheimer’s disease. December 20, 2021. Accessed February 20, 2023. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-reduced-price-aduhelmr-improve-access-patients
6. Takramah WK, Asem L. The efficacy of pharmacological interventions to improve cognitive and behavior symptoms in people with dementia: A systematic review and meta-analysis. Health Sci Rep. 2022;5:e913. doi: 10.1002/hsr2.913
7. GoodRx. Donepezil generic Aricept. Accessed February 20, 2023. www.goodrx.com/donepezil
8. Howard R, McShane R, Lindesay J, et al. Nursing home placement in the donepezil and memantine in moderate to severe Alzheimer’s disease (DOMINO-AD) trial: secondary and post-hoc analyses. Lancet Neurol. 2015;14:1171-1181. doi: 10.1016/S1474-4422(15)00258-6
9. Geldmacher DS, Provenzano G, McRae T, et al. Donepezil is associated with delayed nursing home placement in patients with Alzheimer’s disease. J Am Geriatr Soc. 2003;51:937-944. doi: 10.1046/j.1365-2389.2003.51306.x
10. Pham C, Le K, Draves M, et al. Assessment of FDA-approved drugs not recommended for use or reimbursement in other countries, 2017-2020. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6787
11. Johnston JL, Ross JS, Ramachandran R. US Food and Drug Administration approval of drugs not meeting pivotal trial primary end points, 2018-2021. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6444
1. Budd Haeberlein S, Aisen PS, Barkhof F, et al. Two randomized phase 3 studies of aducanumab in early Alzheimer’s disease. J Prev Alzheimers Dis. 2022;9:197-210. doi: 10.14283/jpad.2022.30
2. Ebell MH, Barry HC. Why physicians should not prescribe aducanumab for Alzheimer disease. Am Fam Physician. 2022;105:353-354.
3. van Dyck CH, Swanson CJ, Aisen P, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388:9-21. doi: 10.1056/NEJMoa2212948
4. Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023; 613:227-228. doi: 10.1038/d41586-023-00030-3
5. Biogen announces reduced price for Aduhelm to improve access for patients with early Alzheimer’s disease. December 20, 2021. Accessed February 20, 2023. https://investors.biogen.com/news-releases/news-release-details/biogen-announces-reduced-price-aduhelmr-improve-access-patients
6. Takramah WK, Asem L. The efficacy of pharmacological interventions to improve cognitive and behavior symptoms in people with dementia: A systematic review and meta-analysis. Health Sci Rep. 2022;5:e913. doi: 10.1002/hsr2.913
7. GoodRx. Donepezil generic Aricept. Accessed February 20, 2023. www.goodrx.com/donepezil
8. Howard R, McShane R, Lindesay J, et al. Nursing home placement in the donepezil and memantine in moderate to severe Alzheimer’s disease (DOMINO-AD) trial: secondary and post-hoc analyses. Lancet Neurol. 2015;14:1171-1181. doi: 10.1016/S1474-4422(15)00258-6
9. Geldmacher DS, Provenzano G, McRae T, et al. Donepezil is associated with delayed nursing home placement in patients with Alzheimer’s disease. J Am Geriatr Soc. 2003;51:937-944. doi: 10.1046/j.1365-2389.2003.51306.x
10. Pham C, Le K, Draves M, et al. Assessment of FDA-approved drugs not recommended for use or reimbursement in other countries, 2017-2020. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6787
11. Johnston JL, Ross JS, Ramachandran R. US Food and Drug Administration approval of drugs not meeting pivotal trial primary end points, 2018-2021. JAMA Intern Med. Published online February 13, 2023. doi: 10.1001/jamainternmed.2022.6444
The SHOW UP Act Threatens VA Telehealth
In February, the US House of Representatives hurriedly passed the Stopping Home Office Work’s Unproductive Problems (SHOW UP) Act, H.R. 139, a bill that calls into question the contributions of federal employees allowed to work from home and resets telework policies to those in place in 2019. Its author, House Oversight Committee Chairman James Comer (R, Kentucky) claimed that this change was necessary because the expansion of federal telework during the COVID-19 pandemic “has crippled the ability of agencies to get their jobs done and created backlogs.” His targets included the US Department of Veterans Affairs (VA), where, he charged, “veterans have been unable…to obtain care they have earned.” He added, “it’s hard to argue that teleworking has helped the VA.”
While oversight of government programs is an authority of Congress, the SHOW UP Act is based on unsubstantiated assumptions of dereliction. It also disregards the devastating impact the proposed changes will have on veterans’ ability to receive care and inaccurately implies improving it. As the Senate considers the bill, they should take heed of these and other facts involving this often misunderstood form of labor.
COVID-19 irrevocably transformed the use of virtual care within the VA and across the world. Even as the pandemic subsides, public and private health care systems have continued to use telework-centered telehealth far above prepandemic levels, especially for mental health and primary care. Employers, including the VA, capitalize on telework for its benefits to both consumers and the workforce. For consumers, research supports the clinical effectiveness of telemental health service, as well as its cost-effectiveness and consumer satisfaction. On the workforce side, research has documented heightened productivity, lower distractibility, and higher job satisfaction among counselors who shifted to remote work.
Remote work also serves as a key tool in attracting and retaining a qualified workforce. As one VA service chief explained, “I am having enough trouble competing with the private sector, where extensive telework is now the norm. If telework options were rolled back, the private sector will have a field day picking off my best staff.” These comments are consistent with the data. McKinsey’s American Opportunity Survey shows that Americans have embraced remote work and want more of it. Recent data from Gallup show that 6 of 10 currently exclusively remote employees would be extremely likely to change companies if they lost their remote flexibility. Further, Gallup data show that when an employee’s location preference does not match their current work location, burnout rises, and engagement drops.
Between 2019 and 2023, the VA’s telework expansion is what has enabled it to meet the growing demand for mental health services. VA is keeping pace by having 2 or more clinicians rotate between home and a shared VA office. Forcing these hybrid practitioners to work full time at VA facilities would drastically reduce the number of patients they can care for. There simply are not enough offices on crammed VA grounds to house staff who telework today. The net result would be that fewer appointments would be available, creating longer wait times. And that is just for existing patients. It does not factor in the expected influx due to new veteran eligibility made possible by the toxic exposures PACT Act.
Here is another good example of crucial VA telework: With the advent of the 988 Suicide & Crisis Lifeline, VA is adding more than 1000 new Veterans Crisis Line responders. All these new positions are remote. The SHOW UP Act would inhibit this expansion of lifesaving programs.
Veterans want more, not fewer, telehealth options. At a House Committee on Veterans’ Affairs hearing this past September, the VA reported that most veterans would prefer to receive mental health services virtually than to have to commute to a VA medical center or clinic. Telehealth benefits veterans in meaningful ways, including that it reduces their travel time, travel expense, depletion of sick leave, and need for childcare. Veterans with posttraumatic stress disorder, military sexual trauma, those with mobility issues, or those who struggle with the stigma of mental health treatment may prefer the familiarity of their own homes for care. Virtual options also relieve a patient’s need to enter a hospital and be unnecessarily exposed to contagious viruses. That’s safer not only for veterans but also for VA staff.
Finally, virtual care improves treatment. Research has revealed that the likelihood of missing telehealth appointments is lower than for in-person appointments. When patients miss appointments, continuity of care is disrupted, and health care outcomes are diminished.
The pandemic is receding, but the advantages of telework-centered virtual care are greater than ever. Political representatives who want to show up for veterans should do everything in their power to expand—not cut—VA’s ability to authorize working from home.
In February, the US House of Representatives hurriedly passed the Stopping Home Office Work’s Unproductive Problems (SHOW UP) Act, H.R. 139, a bill that calls into question the contributions of federal employees allowed to work from home and resets telework policies to those in place in 2019. Its author, House Oversight Committee Chairman James Comer (R, Kentucky) claimed that this change was necessary because the expansion of federal telework during the COVID-19 pandemic “has crippled the ability of agencies to get their jobs done and created backlogs.” His targets included the US Department of Veterans Affairs (VA), where, he charged, “veterans have been unable…to obtain care they have earned.” He added, “it’s hard to argue that teleworking has helped the VA.”
While oversight of government programs is an authority of Congress, the SHOW UP Act is based on unsubstantiated assumptions of dereliction. It also disregards the devastating impact the proposed changes will have on veterans’ ability to receive care and inaccurately implies improving it. As the Senate considers the bill, they should take heed of these and other facts involving this often misunderstood form of labor.
COVID-19 irrevocably transformed the use of virtual care within the VA and across the world. Even as the pandemic subsides, public and private health care systems have continued to use telework-centered telehealth far above prepandemic levels, especially for mental health and primary care. Employers, including the VA, capitalize on telework for its benefits to both consumers and the workforce. For consumers, research supports the clinical effectiveness of telemental health service, as well as its cost-effectiveness and consumer satisfaction. On the workforce side, research has documented heightened productivity, lower distractibility, and higher job satisfaction among counselors who shifted to remote work.
Remote work also serves as a key tool in attracting and retaining a qualified workforce. As one VA service chief explained, “I am having enough trouble competing with the private sector, where extensive telework is now the norm. If telework options were rolled back, the private sector will have a field day picking off my best staff.” These comments are consistent with the data. McKinsey’s American Opportunity Survey shows that Americans have embraced remote work and want more of it. Recent data from Gallup show that 6 of 10 currently exclusively remote employees would be extremely likely to change companies if they lost their remote flexibility. Further, Gallup data show that when an employee’s location preference does not match their current work location, burnout rises, and engagement drops.
Between 2019 and 2023, the VA’s telework expansion is what has enabled it to meet the growing demand for mental health services. VA is keeping pace by having 2 or more clinicians rotate between home and a shared VA office. Forcing these hybrid practitioners to work full time at VA facilities would drastically reduce the number of patients they can care for. There simply are not enough offices on crammed VA grounds to house staff who telework today. The net result would be that fewer appointments would be available, creating longer wait times. And that is just for existing patients. It does not factor in the expected influx due to new veteran eligibility made possible by the toxic exposures PACT Act.
Here is another good example of crucial VA telework: With the advent of the 988 Suicide & Crisis Lifeline, VA is adding more than 1000 new Veterans Crisis Line responders. All these new positions are remote. The SHOW UP Act would inhibit this expansion of lifesaving programs.
Veterans want more, not fewer, telehealth options. At a House Committee on Veterans’ Affairs hearing this past September, the VA reported that most veterans would prefer to receive mental health services virtually than to have to commute to a VA medical center or clinic. Telehealth benefits veterans in meaningful ways, including that it reduces their travel time, travel expense, depletion of sick leave, and need for childcare. Veterans with posttraumatic stress disorder, military sexual trauma, those with mobility issues, or those who struggle with the stigma of mental health treatment may prefer the familiarity of their own homes for care. Virtual options also relieve a patient’s need to enter a hospital and be unnecessarily exposed to contagious viruses. That’s safer not only for veterans but also for VA staff.
Finally, virtual care improves treatment. Research has revealed that the likelihood of missing telehealth appointments is lower than for in-person appointments. When patients miss appointments, continuity of care is disrupted, and health care outcomes are diminished.
The pandemic is receding, but the advantages of telework-centered virtual care are greater than ever. Political representatives who want to show up for veterans should do everything in their power to expand—not cut—VA’s ability to authorize working from home.
In February, the US House of Representatives hurriedly passed the Stopping Home Office Work’s Unproductive Problems (SHOW UP) Act, H.R. 139, a bill that calls into question the contributions of federal employees allowed to work from home and resets telework policies to those in place in 2019. Its author, House Oversight Committee Chairman James Comer (R, Kentucky) claimed that this change was necessary because the expansion of federal telework during the COVID-19 pandemic “has crippled the ability of agencies to get their jobs done and created backlogs.” His targets included the US Department of Veterans Affairs (VA), where, he charged, “veterans have been unable…to obtain care they have earned.” He added, “it’s hard to argue that teleworking has helped the VA.”
While oversight of government programs is an authority of Congress, the SHOW UP Act is based on unsubstantiated assumptions of dereliction. It also disregards the devastating impact the proposed changes will have on veterans’ ability to receive care and inaccurately implies improving it. As the Senate considers the bill, they should take heed of these and other facts involving this often misunderstood form of labor.
COVID-19 irrevocably transformed the use of virtual care within the VA and across the world. Even as the pandemic subsides, public and private health care systems have continued to use telework-centered telehealth far above prepandemic levels, especially for mental health and primary care. Employers, including the VA, capitalize on telework for its benefits to both consumers and the workforce. For consumers, research supports the clinical effectiveness of telemental health service, as well as its cost-effectiveness and consumer satisfaction. On the workforce side, research has documented heightened productivity, lower distractibility, and higher job satisfaction among counselors who shifted to remote work.
Remote work also serves as a key tool in attracting and retaining a qualified workforce. As one VA service chief explained, “I am having enough trouble competing with the private sector, where extensive telework is now the norm. If telework options were rolled back, the private sector will have a field day picking off my best staff.” These comments are consistent with the data. McKinsey’s American Opportunity Survey shows that Americans have embraced remote work and want more of it. Recent data from Gallup show that 6 of 10 currently exclusively remote employees would be extremely likely to change companies if they lost their remote flexibility. Further, Gallup data show that when an employee’s location preference does not match their current work location, burnout rises, and engagement drops.
Between 2019 and 2023, the VA’s telework expansion is what has enabled it to meet the growing demand for mental health services. VA is keeping pace by having 2 or more clinicians rotate between home and a shared VA office. Forcing these hybrid practitioners to work full time at VA facilities would drastically reduce the number of patients they can care for. There simply are not enough offices on crammed VA grounds to house staff who telework today. The net result would be that fewer appointments would be available, creating longer wait times. And that is just for existing patients. It does not factor in the expected influx due to new veteran eligibility made possible by the toxic exposures PACT Act.
Here is another good example of crucial VA telework: With the advent of the 988 Suicide & Crisis Lifeline, VA is adding more than 1000 new Veterans Crisis Line responders. All these new positions are remote. The SHOW UP Act would inhibit this expansion of lifesaving programs.
Veterans want more, not fewer, telehealth options. At a House Committee on Veterans’ Affairs hearing this past September, the VA reported that most veterans would prefer to receive mental health services virtually than to have to commute to a VA medical center or clinic. Telehealth benefits veterans in meaningful ways, including that it reduces their travel time, travel expense, depletion of sick leave, and need for childcare. Veterans with posttraumatic stress disorder, military sexual trauma, those with mobility issues, or those who struggle with the stigma of mental health treatment may prefer the familiarity of their own homes for care. Virtual options also relieve a patient’s need to enter a hospital and be unnecessarily exposed to contagious viruses. That’s safer not only for veterans but also for VA staff.
Finally, virtual care improves treatment. Research has revealed that the likelihood of missing telehealth appointments is lower than for in-person appointments. When patients miss appointments, continuity of care is disrupted, and health care outcomes are diminished.
The pandemic is receding, but the advantages of telework-centered virtual care are greater than ever. Political representatives who want to show up for veterans should do everything in their power to expand—not cut—VA’s ability to authorize working from home.
Inequity, Bias, Racism, and Physician Burnout: Staying Connected to Purpose and Identity as an Antidote
“Where are you really from?”
When I tell patients I am from Casper, Wyoming—wh ere I have lived the majority of my life—it’smet with disbelief. The subtext: YOU can’t be from THERE.
I didn’t used to think much of comments like this, but as I have continued to hear them, I find myself feeling tired—tired of explaining myself, tired of being treated differently than my colleagues, and tired of justifying myself. My experiences as a woman of color sadly are not uncommon in medicine.
Sara Martinez-Garcia, BA
Racial bias and racism are steeped in the culture of medicine—from the medical school admissions process1,2 to the medical training itself.3 More than half of medical students who identify as underrepresented in medicine (UIM) experience microaggressions.4 Experiencing racism and sexism in the learning environment can lead to burnout, and microaggressions promote feelings of self-doubt and isolation. Medical students who experience microaggressions are more likely to report feelings of burnout and impaired learning.4 These experiences can leave one feeling as if “You do not belong” and “You are unworthy of being in this position.”
Addressing physician burnout already is complex, and addressing burnout caused by inequity, bias, and racism is even more so. In an ideal world, we would eliminate inequity, bias, and racism in medicine through institutional and individual actions. There has been movement to do so. For example, the Accreditation Council for Graduate Medical Education (ACGME), which oversees standards for US resident and fellow training, launched ACGME Equity Matters (https://www.acgme.org/what-we-do/diversity-equity-and-inclusion/ACGME-Equity-Matters/), an initiative aimed to improve diversity, equity, and antiracism practices within graduate medical eduation. However, we know that education alone isn’t enough to fix this monumental problem. Traditional diversity training as we have known it has never been demonstrated to contribute to lasting changes in behavior; it takes much more extensive and complex interventions to meaningfully reduce bias.5 In the meantime, we need action. As a medical community, we need to be better about not turning the other way when we see these things happening in our classrooms and in our hospitals. As individuals, we must self-reflect on the role that we each play in contributing to or combatting injustices and seek out bystander training to empower us to speak out against acts of bias such as sexism or racism. Whether it is supporting a fellow colleague or speaking out against an inappropriate interaction, we can all do our part. A very brief list of actions and resources to support our UIM students and colleagues are listed in the Table; those interested in more in-depth resources are encouraged to explore the Association of American Medical Colleges Diversity and Inclusion Toolkit (https://www.aamc.org/professional-development/affinity-groups/cfas/diversity-inclusion-toolkit/resources).

We can’t change the culture of medicine quickly or even in our lifetime. In the meantime, those who are UIM will continue to experience these events that erode our well-being. They will continue to need support. Discussing mental health has long been stigmatized, and physicians are no exception. Many physicians are hesitant to discuss mental health issues out of fear of judgement and perceived or even real repercussions on their careers.10 However, times are changing and evolving with the current generation of medical students. It’s no secret that medicine is stressful. Most medical schools provide free counseling services, which lowers the barrier for discussions of mental health from the beginning. Making talk about mental health just as normal as talking about other aspects of health takes away the fear that “something is wrong with me” if someone seeks out counseling and mental health services. Faculty should actively check in and maintain open lines of communication, which can be invaluable for UIM students and their training experience. Creating an environment where trainees can be real and honest about the struggles they face in and out of the classroom can make everyone feel like they are not alone.
Addressing burnout in medicine is going to require an all-hands-on-deck approach. At an institutional level, there is a lot of room for improvement—improving systems for physicians so they are able to operate at their highest level (eg, addressing the burdens of prior authorizations and the electronic medical record), setting reasonable expectations around productivity, and creating work structures that respect work-life balance.11 But what can we do for ourselves? We believe that one of the most important ways to protect ourselves from burnout is to remember why. As a medical student, there is enormous pressure—pressure to learn an enormous volume of information, pass examinations, get involved in extracurricular activities, make connections, and seek research opportunities, while also cooking healthy food, grocery shopping, maintaining relationships with loved ones, and generally taking care of oneself. At times it can feel as if our lives outside of medical school are not important enough or valuable enough to make time for, but the pieces of our identity outside of medicine are what shape us into who we are today and are the roots of our purpose in medicine. Sometimes you can feel the most motivated, valued, and supported when you make time to have dinner with friends, call a family member, or simply spend time alone in the outdoors. Who you are and how you got to this point in your life are your identity. Reminding yourself of that can help when experiencing microaggressions or when that voice tries to tell you that you are not worthy. As you progress further in your career, maintaining that relationship with who you are outside of medicine can be your armor against burnout.
- Capers Q IV, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369.
- Lucey CR, Saguil A. The consequences of structural racism on MCAT scores and medical school admissions: the past is prologue. Acad Med. 2020;95:351-356.
- Nguemeni Tiako MJ, South EC, Ray V. Medical schools as racialized organizations: a primer. Ann Intern Med. 2021;174:1143-1144.
- Chisholm LP, Jackson KR, Davidson HA, et al. Evaluation of racial microaggressions experienced during medical school training and the effect on medical student education and burnout: a validation study. J Natl Med Assoc. 2021;113:310-314.
- Dobbin F, Kalev A. Why doesn’t diversity training work? the challenge for industry and academia. Anthropology Now. 2018;10:48-55.
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Int J Womens Dermatol. 2020;6:57-60.
- Hackworth JM, Kotagal M, Bignall ONR, et al. Microaggressions: privileged observers’ duty to act and what they can do [published online December 1, 2021]. Pediatrics. doi:10.1542/peds.2021-052758.
- Wheeler DJ, Zapata J, Davis D, et al. Twelve tips for responding to microaggressions and overt discrimination: when the patient offends the learner. Med Teach. 2019;41:1112-1117.
- Scott K. Just Work: How to Root Out Bias, Prejudice, and Bullying to Build a Kick-Ass Culture of Inclusivity. St. Martin’s Press; 2021.
- Center C, Davis M, Detre T, et al. Confronting depression and suicide in physicians: a consensus statement. JAMA. 2003;289:3161-3166.
- West CP, Dyrbye LN, Shanafelt TD. Physician burnout: contributors, consequences and solutions. J Intern Med. 2018;283:516-529.
“Where are you really from?”
When I tell patients I am from Casper, Wyoming—wh ere I have lived the majority of my life—it’smet with disbelief. The subtext: YOU can’t be from THERE.
I didn’t used to think much of comments like this, but as I have continued to hear them, I find myself feeling tired—tired of explaining myself, tired of being treated differently than my colleagues, and tired of justifying myself. My experiences as a woman of color sadly are not uncommon in medicine.
Sara Martinez-Garcia, BA
Racial bias and racism are steeped in the culture of medicine—from the medical school admissions process1,2 to the medical training itself.3 More than half of medical students who identify as underrepresented in medicine (UIM) experience microaggressions.4 Experiencing racism and sexism in the learning environment can lead to burnout, and microaggressions promote feelings of self-doubt and isolation. Medical students who experience microaggressions are more likely to report feelings of burnout and impaired learning.4 These experiences can leave one feeling as if “You do not belong” and “You are unworthy of being in this position.”
Addressing physician burnout already is complex, and addressing burnout caused by inequity, bias, and racism is even more so. In an ideal world, we would eliminate inequity, bias, and racism in medicine through institutional and individual actions. There has been movement to do so. For example, the Accreditation Council for Graduate Medical Education (ACGME), which oversees standards for US resident and fellow training, launched ACGME Equity Matters (https://www.acgme.org/what-we-do/diversity-equity-and-inclusion/ACGME-Equity-Matters/), an initiative aimed to improve diversity, equity, and antiracism practices within graduate medical eduation. However, we know that education alone isn’t enough to fix this monumental problem. Traditional diversity training as we have known it has never been demonstrated to contribute to lasting changes in behavior; it takes much more extensive and complex interventions to meaningfully reduce bias.5 In the meantime, we need action. As a medical community, we need to be better about not turning the other way when we see these things happening in our classrooms and in our hospitals. As individuals, we must self-reflect on the role that we each play in contributing to or combatting injustices and seek out bystander training to empower us to speak out against acts of bias such as sexism or racism. Whether it is supporting a fellow colleague or speaking out against an inappropriate interaction, we can all do our part. A very brief list of actions and resources to support our UIM students and colleagues are listed in the Table; those interested in more in-depth resources are encouraged to explore the Association of American Medical Colleges Diversity and Inclusion Toolkit (https://www.aamc.org/professional-development/affinity-groups/cfas/diversity-inclusion-toolkit/resources).

We can’t change the culture of medicine quickly or even in our lifetime. In the meantime, those who are UIM will continue to experience these events that erode our well-being. They will continue to need support. Discussing mental health has long been stigmatized, and physicians are no exception. Many physicians are hesitant to discuss mental health issues out of fear of judgement and perceived or even real repercussions on their careers.10 However, times are changing and evolving with the current generation of medical students. It’s no secret that medicine is stressful. Most medical schools provide free counseling services, which lowers the barrier for discussions of mental health from the beginning. Making talk about mental health just as normal as talking about other aspects of health takes away the fear that “something is wrong with me” if someone seeks out counseling and mental health services. Faculty should actively check in and maintain open lines of communication, which can be invaluable for UIM students and their training experience. Creating an environment where trainees can be real and honest about the struggles they face in and out of the classroom can make everyone feel like they are not alone.
Addressing burnout in medicine is going to require an all-hands-on-deck approach. At an institutional level, there is a lot of room for improvement—improving systems for physicians so they are able to operate at their highest level (eg, addressing the burdens of prior authorizations and the electronic medical record), setting reasonable expectations around productivity, and creating work structures that respect work-life balance.11 But what can we do for ourselves? We believe that one of the most important ways to protect ourselves from burnout is to remember why. As a medical student, there is enormous pressure—pressure to learn an enormous volume of information, pass examinations, get involved in extracurricular activities, make connections, and seek research opportunities, while also cooking healthy food, grocery shopping, maintaining relationships with loved ones, and generally taking care of oneself. At times it can feel as if our lives outside of medical school are not important enough or valuable enough to make time for, but the pieces of our identity outside of medicine are what shape us into who we are today and are the roots of our purpose in medicine. Sometimes you can feel the most motivated, valued, and supported when you make time to have dinner with friends, call a family member, or simply spend time alone in the outdoors. Who you are and how you got to this point in your life are your identity. Reminding yourself of that can help when experiencing microaggressions or when that voice tries to tell you that you are not worthy. As you progress further in your career, maintaining that relationship with who you are outside of medicine can be your armor against burnout.
“Where are you really from?”
When I tell patients I am from Casper, Wyoming—wh ere I have lived the majority of my life—it’smet with disbelief. The subtext: YOU can’t be from THERE.
I didn’t used to think much of comments like this, but as I have continued to hear them, I find myself feeling tired—tired of explaining myself, tired of being treated differently than my colleagues, and tired of justifying myself. My experiences as a woman of color sadly are not uncommon in medicine.
Sara Martinez-Garcia, BA
Racial bias and racism are steeped in the culture of medicine—from the medical school admissions process1,2 to the medical training itself.3 More than half of medical students who identify as underrepresented in medicine (UIM) experience microaggressions.4 Experiencing racism and sexism in the learning environment can lead to burnout, and microaggressions promote feelings of self-doubt and isolation. Medical students who experience microaggressions are more likely to report feelings of burnout and impaired learning.4 These experiences can leave one feeling as if “You do not belong” and “You are unworthy of being in this position.”
Addressing physician burnout already is complex, and addressing burnout caused by inequity, bias, and racism is even more so. In an ideal world, we would eliminate inequity, bias, and racism in medicine through institutional and individual actions. There has been movement to do so. For example, the Accreditation Council for Graduate Medical Education (ACGME), which oversees standards for US resident and fellow training, launched ACGME Equity Matters (https://www.acgme.org/what-we-do/diversity-equity-and-inclusion/ACGME-Equity-Matters/), an initiative aimed to improve diversity, equity, and antiracism practices within graduate medical eduation. However, we know that education alone isn’t enough to fix this monumental problem. Traditional diversity training as we have known it has never been demonstrated to contribute to lasting changes in behavior; it takes much more extensive and complex interventions to meaningfully reduce bias.5 In the meantime, we need action. As a medical community, we need to be better about not turning the other way when we see these things happening in our classrooms and in our hospitals. As individuals, we must self-reflect on the role that we each play in contributing to or combatting injustices and seek out bystander training to empower us to speak out against acts of bias such as sexism or racism. Whether it is supporting a fellow colleague or speaking out against an inappropriate interaction, we can all do our part. A very brief list of actions and resources to support our UIM students and colleagues are listed in the Table; those interested in more in-depth resources are encouraged to explore the Association of American Medical Colleges Diversity and Inclusion Toolkit (https://www.aamc.org/professional-development/affinity-groups/cfas/diversity-inclusion-toolkit/resources).

We can’t change the culture of medicine quickly or even in our lifetime. In the meantime, those who are UIM will continue to experience these events that erode our well-being. They will continue to need support. Discussing mental health has long been stigmatized, and physicians are no exception. Many physicians are hesitant to discuss mental health issues out of fear of judgement and perceived or even real repercussions on their careers.10 However, times are changing and evolving with the current generation of medical students. It’s no secret that medicine is stressful. Most medical schools provide free counseling services, which lowers the barrier for discussions of mental health from the beginning. Making talk about mental health just as normal as talking about other aspects of health takes away the fear that “something is wrong with me” if someone seeks out counseling and mental health services. Faculty should actively check in and maintain open lines of communication, which can be invaluable for UIM students and their training experience. Creating an environment where trainees can be real and honest about the struggles they face in and out of the classroom can make everyone feel like they are not alone.
Addressing burnout in medicine is going to require an all-hands-on-deck approach. At an institutional level, there is a lot of room for improvement—improving systems for physicians so they are able to operate at their highest level (eg, addressing the burdens of prior authorizations and the electronic medical record), setting reasonable expectations around productivity, and creating work structures that respect work-life balance.11 But what can we do for ourselves? We believe that one of the most important ways to protect ourselves from burnout is to remember why. As a medical student, there is enormous pressure—pressure to learn an enormous volume of information, pass examinations, get involved in extracurricular activities, make connections, and seek research opportunities, while also cooking healthy food, grocery shopping, maintaining relationships with loved ones, and generally taking care of oneself. At times it can feel as if our lives outside of medical school are not important enough or valuable enough to make time for, but the pieces of our identity outside of medicine are what shape us into who we are today and are the roots of our purpose in medicine. Sometimes you can feel the most motivated, valued, and supported when you make time to have dinner with friends, call a family member, or simply spend time alone in the outdoors. Who you are and how you got to this point in your life are your identity. Reminding yourself of that can help when experiencing microaggressions or when that voice tries to tell you that you are not worthy. As you progress further in your career, maintaining that relationship with who you are outside of medicine can be your armor against burnout.
- Capers Q IV, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369.
- Lucey CR, Saguil A. The consequences of structural racism on MCAT scores and medical school admissions: the past is prologue. Acad Med. 2020;95:351-356.
- Nguemeni Tiako MJ, South EC, Ray V. Medical schools as racialized organizations: a primer. Ann Intern Med. 2021;174:1143-1144.
- Chisholm LP, Jackson KR, Davidson HA, et al. Evaluation of racial microaggressions experienced during medical school training and the effect on medical student education and burnout: a validation study. J Natl Med Assoc. 2021;113:310-314.
- Dobbin F, Kalev A. Why doesn’t diversity training work? the challenge for industry and academia. Anthropology Now. 2018;10:48-55.
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Int J Womens Dermatol. 2020;6:57-60.
- Hackworth JM, Kotagal M, Bignall ONR, et al. Microaggressions: privileged observers’ duty to act and what they can do [published online December 1, 2021]. Pediatrics. doi:10.1542/peds.2021-052758.
- Wheeler DJ, Zapata J, Davis D, et al. Twelve tips for responding to microaggressions and overt discrimination: when the patient offends the learner. Med Teach. 2019;41:1112-1117.
- Scott K. Just Work: How to Root Out Bias, Prejudice, and Bullying to Build a Kick-Ass Culture of Inclusivity. St. Martin’s Press; 2021.
- Center C, Davis M, Detre T, et al. Confronting depression and suicide in physicians: a consensus statement. JAMA. 2003;289:3161-3166.
- West CP, Dyrbye LN, Shanafelt TD. Physician burnout: contributors, consequences and solutions. J Intern Med. 2018;283:516-529.
- Capers Q IV, Clinchot D, McDougle L, et al. Implicit racial bias in medical school admissions. Acad Med. 2017;92:365-369.
- Lucey CR, Saguil A. The consequences of structural racism on MCAT scores and medical school admissions: the past is prologue. Acad Med. 2020;95:351-356.
- Nguemeni Tiako MJ, South EC, Ray V. Medical schools as racialized organizations: a primer. Ann Intern Med. 2021;174:1143-1144.
- Chisholm LP, Jackson KR, Davidson HA, et al. Evaluation of racial microaggressions experienced during medical school training and the effect on medical student education and burnout: a validation study. J Natl Med Assoc. 2021;113:310-314.
- Dobbin F, Kalev A. Why doesn’t diversity training work? the challenge for industry and academia. Anthropology Now. 2018;10:48-55.
- Okoye GA. Supporting underrepresented minority women in academic dermatology. Int J Womens Dermatol. 2020;6:57-60.
- Hackworth JM, Kotagal M, Bignall ONR, et al. Microaggressions: privileged observers’ duty to act and what they can do [published online December 1, 2021]. Pediatrics. doi:10.1542/peds.2021-052758.
- Wheeler DJ, Zapata J, Davis D, et al. Twelve tips for responding to microaggressions and overt discrimination: when the patient offends the learner. Med Teach. 2019;41:1112-1117.
- Scott K. Just Work: How to Root Out Bias, Prejudice, and Bullying to Build a Kick-Ass Culture of Inclusivity. St. Martin’s Press; 2021.
- Center C, Davis M, Detre T, et al. Confronting depression and suicide in physicians: a consensus statement. JAMA. 2003;289:3161-3166.
- West CP, Dyrbye LN, Shanafelt TD. Physician burnout: contributors, consequences and solutions. J Intern Med. 2018;283:516-529.
Robotic peritoneal vaginoplasty
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
When the Food and Drug Administration first approved the da Vinci Surgical System (Intuitive Surgical, Sunnyvale, Calif.) for adult use in 2000, it altered the face of minimally invasive surgery across a multitude of specialties. Improved three-dimensional visualization and enhanced instrument articulation facilitates complex dissections and intracorporeal suturing. While the standard of care for gender-affirming vaginoplasty remains the single-stage penile inversion vaginoplasty, robotic procedures are quickly emerging as alternative options for both primary and revisional surgeries.
The single-stage penile inversion vaginoplasty requires an adequate amount of penoscrotal tissue not only to line a neovaginal canal that measures 12-15 cm, but also to create external vulvar structures. While this is often sufficient in most candidates, there is an increasing number of patients who are receiving puberty blockers, resulting in penoscrotal hypoplasia.
Alternatively, there are patients who experience loss of vaginal depth and vaginal stenosis who seek revisional surgeries. Additional donor sites for skin grafting are available and include the lower abdomen and thighs, although patients may not want these donor site scars. With these donor sites, there is also concern about graft contracture, which could lead to recurrent vaginal stenosis.1 Robotic peritoneal vaginoplasty and robotic enteric vaginoplasty can serve as additional options for patients seeking revisional surgery or who have insufficient genital skin. One benefit of using peritoneal flaps is that they are hairless and are well vascularized with minimal donor site morbidity.1 Currently, there are two predominant techniques that utilize peritoneal flaps: the modified Davydov procedure and the tubularized urachus-peritoneal hinge flap.
The modified Davydov technique, which originated in the treatment of congenital vaginal agenesis in cisgender women, involves the creation of anterior and posterior peritoneal flaps. This type of peritoneal vaginoplasty is more commonly utilized for primary cases.
Ideally, there is a robotic surgeon (typically a urologist) working in tandem with the perineal surgeon. The robotic surgeon makes a horizontal incision along the peritoneal ridge at the rectovesical junction and continues the dissection within Denonvilliers fascia, between the prostate and rectum, to the pelvic floor. This dissection is like that performed in a robot-assisted laparoscopic prostatectomy.
Simultaneously, the perineal surgeon will break through the pelvic floor with assistance of the robotic view. Peritoneal flaps are raised from the anterior rectum and posterior bladder.2,3 In primary cases, the penoscrotal flap is introduced into the abdomen from the perineum and sutured to the anterior and posterior peritoneum to create a circumferential canal. At the apex of the neovagina, these anterior and posterior flaps are then sutured together.2,3
The tubularized urachus-peritoneal hinge flap technique is predominantly used for revision cases in patients who experienced neovaginal shortening and desire increased neovaginal depth. As peritoneal reach is limited, candidates for this procedure must have both adequate width and neovaginal canal depth.4 Once intra-abdominal access is achieved, an anterior peritoneal flap is mobilized to the level of the bladder and rotated 180 degrees inferiorly.4 The superior aspect of the flap is flipped is mobilized and is sutured to the peritoneum at the apex of the neovaginal canal.
The main benefit of these procedures, compared with traditional techniques, is increased neovaginal depth. The average vaginal length in patients undergoing peritoneal vaginoplasties is 14.2 cm, compared with 11.6 cm achieved in those using skin grafts.1,3 However, many surgeons report achieving 14-15 cm of depth with the traditional vaginoplasty. There are insufficient short- and long-term data for the peritoneal technique to recommend this as a first-line procedure.
Complications for peritoneal vaginoplasty procedures are similar to those of single-stage penile inversion vaginoplasty cases but with additional operative risks associated with laparoscopic/robotic surgery. These risks include injury to viscera and major vessels during initial intra-abdominal access, intra-abdominal adhesions, port site hernias, need to convert to an open procedure, and equipment malfunction.2 Additional postoperative risks include pelvic abscess formation, dehiscence of the peritoneal-vaginal incision, and peritoneal perforation during dilation.2,3 Surgeons and institutions must also weigh the cost of using the robot versus the cost of additional revisional surgical procedures. While initial studies evaluating robotic peritoneal vaginoplasty procedures have yielded promising preliminary results, additional studies are warranted.
Dr. Brandt is an ob.gyn. and fellowship-trained gender-affirming surgeon in West Reading, Pa.
References
1. Salibian AA et al. Plast Reconstr Surg. 2021;147(4):634e-43e.
2. Dy GW et al. In: Nikolavsky D and Blakely SA, eds. Urological care for the transgender patient: A comprehensive guide. Switzerland: Springer, 2021:237-48.
3. Jacoby A et al. J Urol. 2019;201(6):1171-5.
4. Smith SM et al. J Sex Med. 2022;10(6):100572.
U.S. vs. French guidelines for osteoporosis treatment
Lille, France – Bernard Cortet, MD, PhD, chairperson of the Osteoporosis Research and Information Group and head of the rheumatology department at Lille (France) University Hospital, has agreed to compare the new U.S. guidelines to the 2018 French recommendations written under the aegis of the French Society for Rheumatology and GRIO. Dr. Cortet participated in drafting the French recommendations.
Question: The ACP “strongly” recommends initial pharmacologic treatment with bisphosphonate antiresorptive drugs (alendronate, ibandronate, risedronate, zoledronate) in postmenopausal females diagnosed with primary osteoporosis. Isn’t this what the SFR–GRIO have been recommending for many years?
Answer: The ACP reinforces its stance by arguing that in postmenopausal females with primary osteoporosis, bisphosphonates have the most favorable balance between benefits, harms, patient values and preferences, and cost among the drug classes that were evaluated. In addition to net clinical benefits, bisphosphonates are much cheaper than other pharmacologic treatments and are available in generic oral and injectable formulations.
Our French recommendations specify the choice of drug based on the type of fracture in women and on their bone mineral density (BMD). However, bisphosphonates are definitely given pride of place. When treatment for osteoporosis needs to be started, most of the time, a bisphosphonate is the treatment of choice.
Nevertheless, as also highlighted by the ACP, a more “aggressive” approach must be considered for more severe cases.
In the case of a severe fracture, the French recommendations indicate that all treatments can be prescribed. However, zoledronic acid should be favored as first-line treatment for a hip fracture. In other cases – with or without a nonsevere fracture – the therapeutic indication depends on the BMD values, and in difficult cases, on tools such as FRAX [the Fracture Risk Assessment Tool].
Our guidance strongly recommends opting for an injection in other contexts, such as significant decrease in bone density, presence of comorbidities, poor treatment compliance, brain function disorders, and polymedication.
Q. But it’s not really as simple as prescribing a bisphosphonate, is it?
A. You’re right, many people find the idea of taking bisphosphonates worrying because of associated jaw problems – osteonecrosis of the jaw – or atypical femoral fractures, based on what they’ve read on the Internet, where these serious adverse events are on display front and center with no mention of how often they actually happen and, often, failing to mention how effective bisphosphonates truly are.
These complications are real, but fortunately rare, especially during the first 5 years of treatment. To put this into context, for bisphosphonates, there’s one case of osteonecrosis of the jaw for every 10,000. And for denosumab, there are five cases for every 10,000. For atypical fractures, there’s one case for every 30,000 to 50,000.
Q. The U.S. guidelines also recommend that clinicians use a RANK ligand inhibitor – denosumab, also an antiresorptive drug – as second-line medical treatment. This is to reduce the risk of fractures in postmenopausal women diagnosed with primary osteoporosis and presenting with contraindications or side effects of bisphosphonates. Do you support the use of denosumab as second-line treatment?
A. French legislation classifies it as a second-line treatment, after bisphosphonates. However, there are arguments in favor of prescribing it as first-line treatment in some contexts. If denosumab is to be prescribed – via a twice-yearly subcutaneous injection – full compliance must be observed. If a patient is to stop taking denosumab, an opinion from a medical professional is required before treatment can be discontinued, and then treatment with bisphosphonates must be prescribed.
Q. The ACP recommends that clinicians use either a sclerostin inhibitor – romosozumab – or recombinant human parathyroid hormone – teriparatide – two anabolic agents, followed by a bisphosphonate, with the aim of reducing the risk of fractures. This is only used in women with primary osteoporosis who are at a very high risk of fracture. As romosozumab is not available in France, it’s not really worth discussing its use. Does this strategy seem advisable to you, though?
A. The main issue is what is understood by “women at a very high risk of fracture.” There’s no consensus on the definition of what constitutes a woman at a very high risk of fracture, but we can assume that it involves the combination of low BMD and at least one severe fracture.
The role of anabolic bone treatment, as [the ACP] has defined it, seems logical to me, because in cases of severe osteoporosis with fracture, the risk of recurrence is very high in the next 2-3 years. In a study comparing risedronate and teriparatide in cases of severe osteoporosis, teriparatide was more effective in reducing the recurrence of vertebral fractures.
The favorable opinion of the French National Authority for Health in relation to medical coverage for romosozumab in the treatment of severe postmenopausal osteoporosis in women under the age of 75 years with a history of severe fractures, a T-score less than –2.5, and no previous history of coronary artery disease dates to 2021. This is because medical coverage for this specific group was not listed in the marketing authorization (MA) description for this drug.
But the review by the Economic Committee for Health Products failed to reach a consensus regarding the price. Today, in theory, romosozumab can be dispensed in France by hospital pharmacies, because it is approved for use in public hospitals. Romosozumab is a very interesting drug for relatively young women, especially those with multiple vertebral fractures. This injectable treatment is more effective than teriparatide in increasing BMD values and more effective than alendronate in preventing the recurrence of fractures.
Regarding medical coverage, as it stands, in cases where patients have a T-score less than or equal to –3, the 2018 SFR–GRIO recommends starting treatment even if the patient has no fractures. In cases with severe fractures combined with very low BMD (T-score ≤ –3), injectable treatments may be used to reach a bone density target (T-score > –2.5 to –2 for the hip) at the end of the treatment plan. [These treatments include] zoledronic acid, denosumab (in case of bisphosphonate failure or intolerance), or a treatment plan with teriparatide (covered by medical insurance if the patient has at least two vertebral fractures) followed by an antiresorptive drug (bisphosphonate or denosumab).
Romosozumab is a humanized monoclonal antibody (IgG2) that binds to sclerostin and acts as an inhibitor. This increases bone formation because of the activation of [bone lining cells], the production of bone matrix by osteoblasts, and the recruitment of osteochondroprogenitor cells. Moreover, romosozumab causes changes in the expression of osteoclast mediators, which decreases bone resorption. Together, these two effects that increase bone formation and decrease bone resorption lead to the rapid increase of trabecular and cortical bone mass, as well as improvements in bone structure and strength.
Women treated with a bone anabolic agent must take an antiresorptive agent at the end of their treatment so that the benefits from the treatment remain in the long term. The French and U.S. guidelines line up on this point.
In patients with two prevalent vertebral fractures, the U.S. guidelines state that teriparatide can be prescribed as first-line treatment at diagnosis in the absence of any contraindications. We agree on this point as well.
Moreover, in women under the age of 70 years with osteoporosis requiring treatment, French experts recommend prescribing raloxifene, a selective estrogen-receptor modulator. This is if the risk of nonvertebral fracture is low, as defined by the absence of the following criteria: low hip T-score, risk of falling, and history of nonvertebral fracture. Opportunities for its use are limited, and it doesn’t even figure among the U.S. recommendations.
Q. The ACP recommends that clinicians adopt an individualized approach regarding whether to start medical treatment with a bisphosphonate in women over age 65 years with low bone mass (osteopenia) to reduce the risk of fractures. If treatment is started, they›re of the opinion that a bisphosphonate must be used. What are the recommendations in France?
A. It should be noted that this recommendation by the ACP is conditional because of the low-certainty evidence.
Here’s a brief reminder of important things to note: a T-score between –2.5 and –1 indicates osteopenia; a T-score less than or equal to –2.5 indicates osteoporosis; a T-score less than or equal to –2.5 with one or several fractures indicates severe osteoporosis. The French recommendations state that treatment is not justified if a patient’s T-score is higher than –2 and there’s no presence of fractures, even with risk factors (and/or multiple falls). For T-scores less than or equal to –2 and higher than –3, the decision to prescribe depends on the specialist.
Q. The ACP recommends that clinicians use bisphosphonates for the initial medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis.
A. The ACP recommends that clinicians use a RANK ligand inhibitor – denosumab – as second-line medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis who present with contraindications or who are experiencing side effects of bisphosphonates. This treatment is not covered by health insurance for men in France.
Between 20% and 25% of clinical osteoporotic fractures occur in men. After age 50 years, men are roughly 20% more likely to experience an osteoporotic fracture in their lifetime. The French recommendations regarding the management and treatment of osteoporosis in men were published in 2021.
In the case of severe fractures (vertebrae, pelvis, upper end of the femur, distal femur, proximal humerus) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –1.
In the case of nonsevere fractures (particularly wrist and ankle) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –2. If there are no fractures, osteoporosis treatment is recommended in men at risk of bone fragility or of falling and if one of the T-scores is less than or equal to –3. In patients who had a fracture of the upper end of the femur attributable to bone fragility, zoledronic acid is recommended as first-line treatment.
For men with a severe nonvertebral fracture, single vertebral fracture, or nonsevere fracture, two treatments are indicated and covered by health insurance in France: zoledronic acid and risedronate. In men with at least two vertebral fractures, the following treatments are indicated and covered by health insurance in France: teriparatide and risedronate. In this case, teriparatide is prescribed for a period of 18 months. It must be followed by a prescription of oral or intravenous bisphosphonates.
Q. What is your take on the HAS update to the proper use of osteoporosis medication that’s just been published?
A. Like in the 2018 SFR–GRIO guidelines, no update has been made to the section on postmenopausal osteoporosis, except for the HAS introduction to the proper use of romosozumab, even though it’s not covered by health insurance in France.
In accordance with the MA, it doesn’t make sense to include this drug on the list of treatment options available for women with and without fractures, as it’s not included in the HAS-selected list of drugs covered by health insurance in France.
But I’m glad that the HAS has adopted the GRIO and SFR recommendations regarding corticosteroid-induced osteoporosis. Preventive treatment for corticosteroid-induced osteoporosis must be considered as soon as the daily dose of corticosteroids reaches or exceeds the equivalent of 7.5 mg of prednisone and when the estimated duration of corticosteroid therapy exceeds 3 months.
In summary, in women and men over the age of 50 years, the intake of the equivalent of 7.5 mg/day or more of prednisone or a history of a low-trauma fracture or being age 70 years or older, even with a T-score less than or equal to –2.5 for one of the two sites, indicates prescribing a bisphosphonate. Teriparatide is indicated if the patient has two vertebral fractures.
This article was translated from Medscape’s French edition.
A version of this article first appeared on Medscape.com.
Lille, France – Bernard Cortet, MD, PhD, chairperson of the Osteoporosis Research and Information Group and head of the rheumatology department at Lille (France) University Hospital, has agreed to compare the new U.S. guidelines to the 2018 French recommendations written under the aegis of the French Society for Rheumatology and GRIO. Dr. Cortet participated in drafting the French recommendations.
Question: The ACP “strongly” recommends initial pharmacologic treatment with bisphosphonate antiresorptive drugs (alendronate, ibandronate, risedronate, zoledronate) in postmenopausal females diagnosed with primary osteoporosis. Isn’t this what the SFR–GRIO have been recommending for many years?
Answer: The ACP reinforces its stance by arguing that in postmenopausal females with primary osteoporosis, bisphosphonates have the most favorable balance between benefits, harms, patient values and preferences, and cost among the drug classes that were evaluated. In addition to net clinical benefits, bisphosphonates are much cheaper than other pharmacologic treatments and are available in generic oral and injectable formulations.
Our French recommendations specify the choice of drug based on the type of fracture in women and on their bone mineral density (BMD). However, bisphosphonates are definitely given pride of place. When treatment for osteoporosis needs to be started, most of the time, a bisphosphonate is the treatment of choice.
Nevertheless, as also highlighted by the ACP, a more “aggressive” approach must be considered for more severe cases.
In the case of a severe fracture, the French recommendations indicate that all treatments can be prescribed. However, zoledronic acid should be favored as first-line treatment for a hip fracture. In other cases – with or without a nonsevere fracture – the therapeutic indication depends on the BMD values, and in difficult cases, on tools such as FRAX [the Fracture Risk Assessment Tool].
Our guidance strongly recommends opting for an injection in other contexts, such as significant decrease in bone density, presence of comorbidities, poor treatment compliance, brain function disorders, and polymedication.
Q. But it’s not really as simple as prescribing a bisphosphonate, is it?
A. You’re right, many people find the idea of taking bisphosphonates worrying because of associated jaw problems – osteonecrosis of the jaw – or atypical femoral fractures, based on what they’ve read on the Internet, where these serious adverse events are on display front and center with no mention of how often they actually happen and, often, failing to mention how effective bisphosphonates truly are.
These complications are real, but fortunately rare, especially during the first 5 years of treatment. To put this into context, for bisphosphonates, there’s one case of osteonecrosis of the jaw for every 10,000. And for denosumab, there are five cases for every 10,000. For atypical fractures, there’s one case for every 30,000 to 50,000.
Q. The U.S. guidelines also recommend that clinicians use a RANK ligand inhibitor – denosumab, also an antiresorptive drug – as second-line medical treatment. This is to reduce the risk of fractures in postmenopausal women diagnosed with primary osteoporosis and presenting with contraindications or side effects of bisphosphonates. Do you support the use of denosumab as second-line treatment?
A. French legislation classifies it as a second-line treatment, after bisphosphonates. However, there are arguments in favor of prescribing it as first-line treatment in some contexts. If denosumab is to be prescribed – via a twice-yearly subcutaneous injection – full compliance must be observed. If a patient is to stop taking denosumab, an opinion from a medical professional is required before treatment can be discontinued, and then treatment with bisphosphonates must be prescribed.
Q. The ACP recommends that clinicians use either a sclerostin inhibitor – romosozumab – or recombinant human parathyroid hormone – teriparatide – two anabolic agents, followed by a bisphosphonate, with the aim of reducing the risk of fractures. This is only used in women with primary osteoporosis who are at a very high risk of fracture. As romosozumab is not available in France, it’s not really worth discussing its use. Does this strategy seem advisable to you, though?
A. The main issue is what is understood by “women at a very high risk of fracture.” There’s no consensus on the definition of what constitutes a woman at a very high risk of fracture, but we can assume that it involves the combination of low BMD and at least one severe fracture.
The role of anabolic bone treatment, as [the ACP] has defined it, seems logical to me, because in cases of severe osteoporosis with fracture, the risk of recurrence is very high in the next 2-3 years. In a study comparing risedronate and teriparatide in cases of severe osteoporosis, teriparatide was more effective in reducing the recurrence of vertebral fractures.
The favorable opinion of the French National Authority for Health in relation to medical coverage for romosozumab in the treatment of severe postmenopausal osteoporosis in women under the age of 75 years with a history of severe fractures, a T-score less than –2.5, and no previous history of coronary artery disease dates to 2021. This is because medical coverage for this specific group was not listed in the marketing authorization (MA) description for this drug.
But the review by the Economic Committee for Health Products failed to reach a consensus regarding the price. Today, in theory, romosozumab can be dispensed in France by hospital pharmacies, because it is approved for use in public hospitals. Romosozumab is a very interesting drug for relatively young women, especially those with multiple vertebral fractures. This injectable treatment is more effective than teriparatide in increasing BMD values and more effective than alendronate in preventing the recurrence of fractures.
Regarding medical coverage, as it stands, in cases where patients have a T-score less than or equal to –3, the 2018 SFR–GRIO recommends starting treatment even if the patient has no fractures. In cases with severe fractures combined with very low BMD (T-score ≤ –3), injectable treatments may be used to reach a bone density target (T-score > –2.5 to –2 for the hip) at the end of the treatment plan. [These treatments include] zoledronic acid, denosumab (in case of bisphosphonate failure or intolerance), or a treatment plan with teriparatide (covered by medical insurance if the patient has at least two vertebral fractures) followed by an antiresorptive drug (bisphosphonate or denosumab).
Romosozumab is a humanized monoclonal antibody (IgG2) that binds to sclerostin and acts as an inhibitor. This increases bone formation because of the activation of [bone lining cells], the production of bone matrix by osteoblasts, and the recruitment of osteochondroprogenitor cells. Moreover, romosozumab causes changes in the expression of osteoclast mediators, which decreases bone resorption. Together, these two effects that increase bone formation and decrease bone resorption lead to the rapid increase of trabecular and cortical bone mass, as well as improvements in bone structure and strength.
Women treated with a bone anabolic agent must take an antiresorptive agent at the end of their treatment so that the benefits from the treatment remain in the long term. The French and U.S. guidelines line up on this point.
In patients with two prevalent vertebral fractures, the U.S. guidelines state that teriparatide can be prescribed as first-line treatment at diagnosis in the absence of any contraindications. We agree on this point as well.
Moreover, in women under the age of 70 years with osteoporosis requiring treatment, French experts recommend prescribing raloxifene, a selective estrogen-receptor modulator. This is if the risk of nonvertebral fracture is low, as defined by the absence of the following criteria: low hip T-score, risk of falling, and history of nonvertebral fracture. Opportunities for its use are limited, and it doesn’t even figure among the U.S. recommendations.
Q. The ACP recommends that clinicians adopt an individualized approach regarding whether to start medical treatment with a bisphosphonate in women over age 65 years with low bone mass (osteopenia) to reduce the risk of fractures. If treatment is started, they›re of the opinion that a bisphosphonate must be used. What are the recommendations in France?
A. It should be noted that this recommendation by the ACP is conditional because of the low-certainty evidence.
Here’s a brief reminder of important things to note: a T-score between –2.5 and –1 indicates osteopenia; a T-score less than or equal to –2.5 indicates osteoporosis; a T-score less than or equal to –2.5 with one or several fractures indicates severe osteoporosis. The French recommendations state that treatment is not justified if a patient’s T-score is higher than –2 and there’s no presence of fractures, even with risk factors (and/or multiple falls). For T-scores less than or equal to –2 and higher than –3, the decision to prescribe depends on the specialist.
Q. The ACP recommends that clinicians use bisphosphonates for the initial medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis.
A. The ACP recommends that clinicians use a RANK ligand inhibitor – denosumab – as second-line medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis who present with contraindications or who are experiencing side effects of bisphosphonates. This treatment is not covered by health insurance for men in France.
Between 20% and 25% of clinical osteoporotic fractures occur in men. After age 50 years, men are roughly 20% more likely to experience an osteoporotic fracture in their lifetime. The French recommendations regarding the management and treatment of osteoporosis in men were published in 2021.
In the case of severe fractures (vertebrae, pelvis, upper end of the femur, distal femur, proximal humerus) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –1.
In the case of nonsevere fractures (particularly wrist and ankle) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –2. If there are no fractures, osteoporosis treatment is recommended in men at risk of bone fragility or of falling and if one of the T-scores is less than or equal to –3. In patients who had a fracture of the upper end of the femur attributable to bone fragility, zoledronic acid is recommended as first-line treatment.
For men with a severe nonvertebral fracture, single vertebral fracture, or nonsevere fracture, two treatments are indicated and covered by health insurance in France: zoledronic acid and risedronate. In men with at least two vertebral fractures, the following treatments are indicated and covered by health insurance in France: teriparatide and risedronate. In this case, teriparatide is prescribed for a period of 18 months. It must be followed by a prescription of oral or intravenous bisphosphonates.
Q. What is your take on the HAS update to the proper use of osteoporosis medication that’s just been published?
A. Like in the 2018 SFR–GRIO guidelines, no update has been made to the section on postmenopausal osteoporosis, except for the HAS introduction to the proper use of romosozumab, even though it’s not covered by health insurance in France.
In accordance with the MA, it doesn’t make sense to include this drug on the list of treatment options available for women with and without fractures, as it’s not included in the HAS-selected list of drugs covered by health insurance in France.
But I’m glad that the HAS has adopted the GRIO and SFR recommendations regarding corticosteroid-induced osteoporosis. Preventive treatment for corticosteroid-induced osteoporosis must be considered as soon as the daily dose of corticosteroids reaches or exceeds the equivalent of 7.5 mg of prednisone and when the estimated duration of corticosteroid therapy exceeds 3 months.
In summary, in women and men over the age of 50 years, the intake of the equivalent of 7.5 mg/day or more of prednisone or a history of a low-trauma fracture or being age 70 years or older, even with a T-score less than or equal to –2.5 for one of the two sites, indicates prescribing a bisphosphonate. Teriparatide is indicated if the patient has two vertebral fractures.
This article was translated from Medscape’s French edition.
A version of this article first appeared on Medscape.com.
Lille, France – Bernard Cortet, MD, PhD, chairperson of the Osteoporosis Research and Information Group and head of the rheumatology department at Lille (France) University Hospital, has agreed to compare the new U.S. guidelines to the 2018 French recommendations written under the aegis of the French Society for Rheumatology and GRIO. Dr. Cortet participated in drafting the French recommendations.
Question: The ACP “strongly” recommends initial pharmacologic treatment with bisphosphonate antiresorptive drugs (alendronate, ibandronate, risedronate, zoledronate) in postmenopausal females diagnosed with primary osteoporosis. Isn’t this what the SFR–GRIO have been recommending for many years?
Answer: The ACP reinforces its stance by arguing that in postmenopausal females with primary osteoporosis, bisphosphonates have the most favorable balance between benefits, harms, patient values and preferences, and cost among the drug classes that were evaluated. In addition to net clinical benefits, bisphosphonates are much cheaper than other pharmacologic treatments and are available in generic oral and injectable formulations.
Our French recommendations specify the choice of drug based on the type of fracture in women and on their bone mineral density (BMD). However, bisphosphonates are definitely given pride of place. When treatment for osteoporosis needs to be started, most of the time, a bisphosphonate is the treatment of choice.
Nevertheless, as also highlighted by the ACP, a more “aggressive” approach must be considered for more severe cases.
In the case of a severe fracture, the French recommendations indicate that all treatments can be prescribed. However, zoledronic acid should be favored as first-line treatment for a hip fracture. In other cases – with or without a nonsevere fracture – the therapeutic indication depends on the BMD values, and in difficult cases, on tools such as FRAX [the Fracture Risk Assessment Tool].
Our guidance strongly recommends opting for an injection in other contexts, such as significant decrease in bone density, presence of comorbidities, poor treatment compliance, brain function disorders, and polymedication.
Q. But it’s not really as simple as prescribing a bisphosphonate, is it?
A. You’re right, many people find the idea of taking bisphosphonates worrying because of associated jaw problems – osteonecrosis of the jaw – or atypical femoral fractures, based on what they’ve read on the Internet, where these serious adverse events are on display front and center with no mention of how often they actually happen and, often, failing to mention how effective bisphosphonates truly are.
These complications are real, but fortunately rare, especially during the first 5 years of treatment. To put this into context, for bisphosphonates, there’s one case of osteonecrosis of the jaw for every 10,000. And for denosumab, there are five cases for every 10,000. For atypical fractures, there’s one case for every 30,000 to 50,000.
Q. The U.S. guidelines also recommend that clinicians use a RANK ligand inhibitor – denosumab, also an antiresorptive drug – as second-line medical treatment. This is to reduce the risk of fractures in postmenopausal women diagnosed with primary osteoporosis and presenting with contraindications or side effects of bisphosphonates. Do you support the use of denosumab as second-line treatment?
A. French legislation classifies it as a second-line treatment, after bisphosphonates. However, there are arguments in favor of prescribing it as first-line treatment in some contexts. If denosumab is to be prescribed – via a twice-yearly subcutaneous injection – full compliance must be observed. If a patient is to stop taking denosumab, an opinion from a medical professional is required before treatment can be discontinued, and then treatment with bisphosphonates must be prescribed.
Q. The ACP recommends that clinicians use either a sclerostin inhibitor – romosozumab – or recombinant human parathyroid hormone – teriparatide – two anabolic agents, followed by a bisphosphonate, with the aim of reducing the risk of fractures. This is only used in women with primary osteoporosis who are at a very high risk of fracture. As romosozumab is not available in France, it’s not really worth discussing its use. Does this strategy seem advisable to you, though?
A. The main issue is what is understood by “women at a very high risk of fracture.” There’s no consensus on the definition of what constitutes a woman at a very high risk of fracture, but we can assume that it involves the combination of low BMD and at least one severe fracture.
The role of anabolic bone treatment, as [the ACP] has defined it, seems logical to me, because in cases of severe osteoporosis with fracture, the risk of recurrence is very high in the next 2-3 years. In a study comparing risedronate and teriparatide in cases of severe osteoporosis, teriparatide was more effective in reducing the recurrence of vertebral fractures.
The favorable opinion of the French National Authority for Health in relation to medical coverage for romosozumab in the treatment of severe postmenopausal osteoporosis in women under the age of 75 years with a history of severe fractures, a T-score less than –2.5, and no previous history of coronary artery disease dates to 2021. This is because medical coverage for this specific group was not listed in the marketing authorization (MA) description for this drug.
But the review by the Economic Committee for Health Products failed to reach a consensus regarding the price. Today, in theory, romosozumab can be dispensed in France by hospital pharmacies, because it is approved for use in public hospitals. Romosozumab is a very interesting drug for relatively young women, especially those with multiple vertebral fractures. This injectable treatment is more effective than teriparatide in increasing BMD values and more effective than alendronate in preventing the recurrence of fractures.
Regarding medical coverage, as it stands, in cases where patients have a T-score less than or equal to –3, the 2018 SFR–GRIO recommends starting treatment even if the patient has no fractures. In cases with severe fractures combined with very low BMD (T-score ≤ –3), injectable treatments may be used to reach a bone density target (T-score > –2.5 to –2 for the hip) at the end of the treatment plan. [These treatments include] zoledronic acid, denosumab (in case of bisphosphonate failure or intolerance), or a treatment plan with teriparatide (covered by medical insurance if the patient has at least two vertebral fractures) followed by an antiresorptive drug (bisphosphonate or denosumab).
Romosozumab is a humanized monoclonal antibody (IgG2) that binds to sclerostin and acts as an inhibitor. This increases bone formation because of the activation of [bone lining cells], the production of bone matrix by osteoblasts, and the recruitment of osteochondroprogenitor cells. Moreover, romosozumab causes changes in the expression of osteoclast mediators, which decreases bone resorption. Together, these two effects that increase bone formation and decrease bone resorption lead to the rapid increase of trabecular and cortical bone mass, as well as improvements in bone structure and strength.
Women treated with a bone anabolic agent must take an antiresorptive agent at the end of their treatment so that the benefits from the treatment remain in the long term. The French and U.S. guidelines line up on this point.
In patients with two prevalent vertebral fractures, the U.S. guidelines state that teriparatide can be prescribed as first-line treatment at diagnosis in the absence of any contraindications. We agree on this point as well.
Moreover, in women under the age of 70 years with osteoporosis requiring treatment, French experts recommend prescribing raloxifene, a selective estrogen-receptor modulator. This is if the risk of nonvertebral fracture is low, as defined by the absence of the following criteria: low hip T-score, risk of falling, and history of nonvertebral fracture. Opportunities for its use are limited, and it doesn’t even figure among the U.S. recommendations.
Q. The ACP recommends that clinicians adopt an individualized approach regarding whether to start medical treatment with a bisphosphonate in women over age 65 years with low bone mass (osteopenia) to reduce the risk of fractures. If treatment is started, they›re of the opinion that a bisphosphonate must be used. What are the recommendations in France?
A. It should be noted that this recommendation by the ACP is conditional because of the low-certainty evidence.
Here’s a brief reminder of important things to note: a T-score between –2.5 and –1 indicates osteopenia; a T-score less than or equal to –2.5 indicates osteoporosis; a T-score less than or equal to –2.5 with one or several fractures indicates severe osteoporosis. The French recommendations state that treatment is not justified if a patient’s T-score is higher than –2 and there’s no presence of fractures, even with risk factors (and/or multiple falls). For T-scores less than or equal to –2 and higher than –3, the decision to prescribe depends on the specialist.
Q. The ACP recommends that clinicians use bisphosphonates for the initial medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis.
A. The ACP recommends that clinicians use a RANK ligand inhibitor – denosumab – as second-line medical treatment to reduce the risk of fractures in men diagnosed with primary osteoporosis who present with contraindications or who are experiencing side effects of bisphosphonates. This treatment is not covered by health insurance for men in France.
Between 20% and 25% of clinical osteoporotic fractures occur in men. After age 50 years, men are roughly 20% more likely to experience an osteoporotic fracture in their lifetime. The French recommendations regarding the management and treatment of osteoporosis in men were published in 2021.
In the case of severe fractures (vertebrae, pelvis, upper end of the femur, distal femur, proximal humerus) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –1.
In the case of nonsevere fractures (particularly wrist and ankle) attributable to bone fragility, osteoporosis treatment is recommended if one of the T-scores is less than or equal to –2. If there are no fractures, osteoporosis treatment is recommended in men at risk of bone fragility or of falling and if one of the T-scores is less than or equal to –3. In patients who had a fracture of the upper end of the femur attributable to bone fragility, zoledronic acid is recommended as first-line treatment.
For men with a severe nonvertebral fracture, single vertebral fracture, or nonsevere fracture, two treatments are indicated and covered by health insurance in France: zoledronic acid and risedronate. In men with at least two vertebral fractures, the following treatments are indicated and covered by health insurance in France: teriparatide and risedronate. In this case, teriparatide is prescribed for a period of 18 months. It must be followed by a prescription of oral or intravenous bisphosphonates.
Q. What is your take on the HAS update to the proper use of osteoporosis medication that’s just been published?
A. Like in the 2018 SFR–GRIO guidelines, no update has been made to the section on postmenopausal osteoporosis, except for the HAS introduction to the proper use of romosozumab, even though it’s not covered by health insurance in France.
In accordance with the MA, it doesn’t make sense to include this drug on the list of treatment options available for women with and without fractures, as it’s not included in the HAS-selected list of drugs covered by health insurance in France.
But I’m glad that the HAS has adopted the GRIO and SFR recommendations regarding corticosteroid-induced osteoporosis. Preventive treatment for corticosteroid-induced osteoporosis must be considered as soon as the daily dose of corticosteroids reaches or exceeds the equivalent of 7.5 mg of prednisone and when the estimated duration of corticosteroid therapy exceeds 3 months.
In summary, in women and men over the age of 50 years, the intake of the equivalent of 7.5 mg/day or more of prednisone or a history of a low-trauma fracture or being age 70 years or older, even with a T-score less than or equal to –2.5 for one of the two sites, indicates prescribing a bisphosphonate. Teriparatide is indicated if the patient has two vertebral fractures.
This article was translated from Medscape’s French edition.
A version of this article first appeared on Medscape.com.