User login
United States up to 764 measles cases for the year
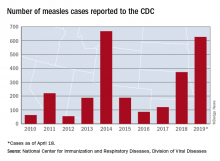
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.
The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.

The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
The number of U.S. measles cases in 2019 rose by 60 during the week ending May 3, which puts the new postelimination high at 764 cases for the year, according to the Centers for Disease Control and Prevention.

The CDC is currently tracking nine measles outbreaks in six states. The largest outbreak this year has been in New York City, mainly Brooklyn, which has almost half (367) of all cases in the country. An outbreak in New York’s Rockland County has resulted in another 109 cases so far this year. California is experiencing outbreaks in three counties – Butte, Los Angeles, and Sacramento – and the state was up to 40 confirmed cases as of May 1. The other states with outbreaks are Michigan, New Jersey, Georgia, and Maryland.
Twenty-three states now have reported measles cases in 2019, as officials in Pittsburgh reported Pennsylvania’s first case on April 30. Four additional cases in the area were reported on May 2 by the Allegheny County Health Department. A measles outbreak is “defined as three or more cases” by the CDC, but the situation in Pennsylvania is not yet being reported as such.
One Pennsylvania legislator in the state, Rep. Daryl Metcalfe (R) of Butler County, has authored a bill that would “bar health care practitioners and facilities and insurance companies from denying care if parents refuse or delay” recommended vaccinations, PennLive.com reported.
Pennsylvania Governor Tom Wolf (D) said that the “bill would put children, pregnant women, and vulnerable patients at risk of being exposed to horrific diseases – at the doctor’s office.”
Arthritis joint pain, inactivity vary greatly across U.S.
Almost 31% of the estimated 54 million adults in the United States with arthritis have severe joint pain, according to the Centers for Disease Control and Prevention.

Nationally, the prevalence of severe joint pain was 30.8% in adults with arthritis in 2017, but state-specific, age-standardized prevalences varied from a low of 20.8% in Colorado to 45.2% in Mississippi. Regionally, prevalences of both severe joint pain and physical inactivity in arthritis patients were highest in the Southeast, noted Dana Guglielmo, MPH, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and associates (MMWR 2019 May 3;68(17):381-7).
The prevalence of arthritis itself was lowest in the District of Columbia at 15.7% and highest in West Virginia at 34.6%. Alabama, at 30.4%, was the only other state above 30%. Colorado had the lowest physical inactivity rate (23.2%), while Kentucky had the highest (44.4%), the investigators said.
The differences among arthritis patients were demographic as well as geographic in 2017. The prevalence of severe joint pain was 33.0% among those aged 18-44 years and 35.6% in those 45-64 but only 25.1% in those aged 65 and older. Whites had a 27.4% prevalence of severe joint pain, compared with 42.0% for Hispanics and 50.9% for blacks. For arthritis patients with a college degree, the age-standardized prevalence of severe joint pain was 15.1%, compared with 35.5% for high school graduates and 54.1% for those with less than a high school degree, based on data from the Behavioral Risk Factor Surveillance System.
“Although persons with arthritis report that pain, or fear of causing or worsening it, is a substantial barrier to exercising, physical activity is an inexpensive intervention that can reduce pain, prevent or delay disability and limitations, and improve mental health, physical functioning, and quality of life with few adverse effects,” wrote Ms. Guglielmo and associates. Adults with severe joint pain “should engage in regular physical activity according to their abilities and avoid physical inactivity [since] even small amounts of physical activity can improve physical functioning in adults with joint conditions.”
SOURCE: Guglielmo D et al. MMWR 2019 May 3;68(17):381-7.
Almost 31% of the estimated 54 million adults in the United States with arthritis have severe joint pain, according to the Centers for Disease Control and Prevention.

Nationally, the prevalence of severe joint pain was 30.8% in adults with arthritis in 2017, but state-specific, age-standardized prevalences varied from a low of 20.8% in Colorado to 45.2% in Mississippi. Regionally, prevalences of both severe joint pain and physical inactivity in arthritis patients were highest in the Southeast, noted Dana Guglielmo, MPH, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and associates (MMWR 2019 May 3;68(17):381-7).
The prevalence of arthritis itself was lowest in the District of Columbia at 15.7% and highest in West Virginia at 34.6%. Alabama, at 30.4%, was the only other state above 30%. Colorado had the lowest physical inactivity rate (23.2%), while Kentucky had the highest (44.4%), the investigators said.
The differences among arthritis patients were demographic as well as geographic in 2017. The prevalence of severe joint pain was 33.0% among those aged 18-44 years and 35.6% in those 45-64 but only 25.1% in those aged 65 and older. Whites had a 27.4% prevalence of severe joint pain, compared with 42.0% for Hispanics and 50.9% for blacks. For arthritis patients with a college degree, the age-standardized prevalence of severe joint pain was 15.1%, compared with 35.5% for high school graduates and 54.1% for those with less than a high school degree, based on data from the Behavioral Risk Factor Surveillance System.
“Although persons with arthritis report that pain, or fear of causing or worsening it, is a substantial barrier to exercising, physical activity is an inexpensive intervention that can reduce pain, prevent or delay disability and limitations, and improve mental health, physical functioning, and quality of life with few adverse effects,” wrote Ms. Guglielmo and associates. Adults with severe joint pain “should engage in regular physical activity according to their abilities and avoid physical inactivity [since] even small amounts of physical activity can improve physical functioning in adults with joint conditions.”
SOURCE: Guglielmo D et al. MMWR 2019 May 3;68(17):381-7.
Almost 31% of the estimated 54 million adults in the United States with arthritis have severe joint pain, according to the Centers for Disease Control and Prevention.

Nationally, the prevalence of severe joint pain was 30.8% in adults with arthritis in 2017, but state-specific, age-standardized prevalences varied from a low of 20.8% in Colorado to 45.2% in Mississippi. Regionally, prevalences of both severe joint pain and physical inactivity in arthritis patients were highest in the Southeast, noted Dana Guglielmo, MPH, of the CDC’s National Center for Chronic Disease Prevention and Health Promotion, Atlanta, and associates (MMWR 2019 May 3;68(17):381-7).
The prevalence of arthritis itself was lowest in the District of Columbia at 15.7% and highest in West Virginia at 34.6%. Alabama, at 30.4%, was the only other state above 30%. Colorado had the lowest physical inactivity rate (23.2%), while Kentucky had the highest (44.4%), the investigators said.
The differences among arthritis patients were demographic as well as geographic in 2017. The prevalence of severe joint pain was 33.0% among those aged 18-44 years and 35.6% in those 45-64 but only 25.1% in those aged 65 and older. Whites had a 27.4% prevalence of severe joint pain, compared with 42.0% for Hispanics and 50.9% for blacks. For arthritis patients with a college degree, the age-standardized prevalence of severe joint pain was 15.1%, compared with 35.5% for high school graduates and 54.1% for those with less than a high school degree, based on data from the Behavioral Risk Factor Surveillance System.
“Although persons with arthritis report that pain, or fear of causing or worsening it, is a substantial barrier to exercising, physical activity is an inexpensive intervention that can reduce pain, prevent or delay disability and limitations, and improve mental health, physical functioning, and quality of life with few adverse effects,” wrote Ms. Guglielmo and associates. Adults with severe joint pain “should engage in regular physical activity according to their abilities and avoid physical inactivity [since] even small amounts of physical activity can improve physical functioning in adults with joint conditions.”
SOURCE: Guglielmo D et al. MMWR 2019 May 3;68(17):381-7.
FROM MMWR
FDA approves ivosidenib frontline for certain AML patients
The Food and Drug Administration has approved ivosidenib (Tibsovo) for newly diagnosed acute myeloid leukemia (AML) with a susceptible IDH1 mutation in patients who are at least 75 years old or have comorbidities preventing the use of intensive induction chemotherapy.
In July 2018, the FDA approved ivosidenib for adults with relapsed or refractory AML with a susceptible IDH1 mutation.
The latest approval was based on results from an open-label, single-arm, multicenter trial of patients with newly diagnosed AML with an IDH1 mutation. Patients were treated with 500 mg ivosidenib daily until disease progression, development of unacceptable toxicity, or hematopoietic stem cell transplantation; the median age of the 28 patients treated with ivosidenib was 77 years.
Of the 28 patients treated, 12 achieved complete remission or complete remission with partial hematologic recovery; 7 of the 17 transfusion-dependent patients achieved transfusion independence for at least 8 weeks.
The most common adverse events were diarrhea, fatigue, edema, decreased appetite, leukocytosis, nausea, arthralgia, abdominal pain, dyspnea, differentiation syndrome, and myalgia. The drug’s prescribing information includes a boxed warning on the risk of differentiation syndrome.
“The recommended ivosidenib dose is 500 mg orally once daily with or without food until disease progression or unacceptable toxicity. For patients without disease progression or unacceptable toxicity, treatment is recommended for a minimum of 6 months to allow time for clinical response,” the FDA noted.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ivosidenib (Tibsovo) for newly diagnosed acute myeloid leukemia (AML) with a susceptible IDH1 mutation in patients who are at least 75 years old or have comorbidities preventing the use of intensive induction chemotherapy.
In July 2018, the FDA approved ivosidenib for adults with relapsed or refractory AML with a susceptible IDH1 mutation.
The latest approval was based on results from an open-label, single-arm, multicenter trial of patients with newly diagnosed AML with an IDH1 mutation. Patients were treated with 500 mg ivosidenib daily until disease progression, development of unacceptable toxicity, or hematopoietic stem cell transplantation; the median age of the 28 patients treated with ivosidenib was 77 years.
Of the 28 patients treated, 12 achieved complete remission or complete remission with partial hematologic recovery; 7 of the 17 transfusion-dependent patients achieved transfusion independence for at least 8 weeks.
The most common adverse events were diarrhea, fatigue, edema, decreased appetite, leukocytosis, nausea, arthralgia, abdominal pain, dyspnea, differentiation syndrome, and myalgia. The drug’s prescribing information includes a boxed warning on the risk of differentiation syndrome.
“The recommended ivosidenib dose is 500 mg orally once daily with or without food until disease progression or unacceptable toxicity. For patients without disease progression or unacceptable toxicity, treatment is recommended for a minimum of 6 months to allow time for clinical response,” the FDA noted.
Find the full press release on the FDA website.
The Food and Drug Administration has approved ivosidenib (Tibsovo) for newly diagnosed acute myeloid leukemia (AML) with a susceptible IDH1 mutation in patients who are at least 75 years old or have comorbidities preventing the use of intensive induction chemotherapy.
In July 2018, the FDA approved ivosidenib for adults with relapsed or refractory AML with a susceptible IDH1 mutation.
The latest approval was based on results from an open-label, single-arm, multicenter trial of patients with newly diagnosed AML with an IDH1 mutation. Patients were treated with 500 mg ivosidenib daily until disease progression, development of unacceptable toxicity, or hematopoietic stem cell transplantation; the median age of the 28 patients treated with ivosidenib was 77 years.
Of the 28 patients treated, 12 achieved complete remission or complete remission with partial hematologic recovery; 7 of the 17 transfusion-dependent patients achieved transfusion independence for at least 8 weeks.
The most common adverse events were diarrhea, fatigue, edema, decreased appetite, leukocytosis, nausea, arthralgia, abdominal pain, dyspnea, differentiation syndrome, and myalgia. The drug’s prescribing information includes a boxed warning on the risk of differentiation syndrome.
“The recommended ivosidenib dose is 500 mg orally once daily with or without food until disease progression or unacceptable toxicity. For patients without disease progression or unacceptable toxicity, treatment is recommended for a minimum of 6 months to allow time for clinical response,” the FDA noted.
Find the full press release on the FDA website.
FDA approves new etanercept biosimilar, Eticovo
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
The Food and Drug Administration has approved Eticovo (etanercept-ykro), a biosimilar of Enbrel (etanercept), for the treatment of several different rheumatologic and dermatologic conditions.
FDA approval was based in part on the results of a phase 3 trial in which 596 patients with moderate to severe rheumatoid arthritis uncontrolled by methotrexate received either Eticovo or Enbrel. The American College of Rheumatology 20% response rate after 24 weeks was 78.1% for Eticovo and 80.3% for Enbrel; the two drugs were statistically equivalent. Both groups had statistically equivalent rates of treatment-emergent adverse events (55.2% vs. 58.2%).
According to the label, Eticovo is a tumor necrosis factor blocker approved for the treatment of rheumatoid arthritis, polyarticular juvenile idiopathic arthritis, psoriatic arthritis, ankylosing spondylitis, and plaque psoriasis in patients aged 4 years or older. The most common adverse events associated with the drug include infections and injection site reactions.
Eticovo is the second etanercept biosimilar approved by the FDA. The first FDA-approved etanercept biosimilar, etanercept-szzs (Erelzi), is currently facing a legal challenge from Amgen, the manufacturer of Enbrel.
Measles cases for 2019 now at postelimination high
according to the Centers for Disease Control and Prevention.
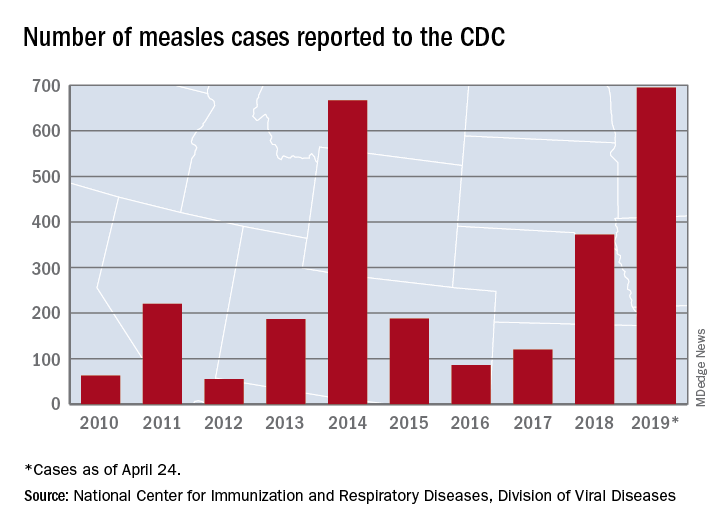
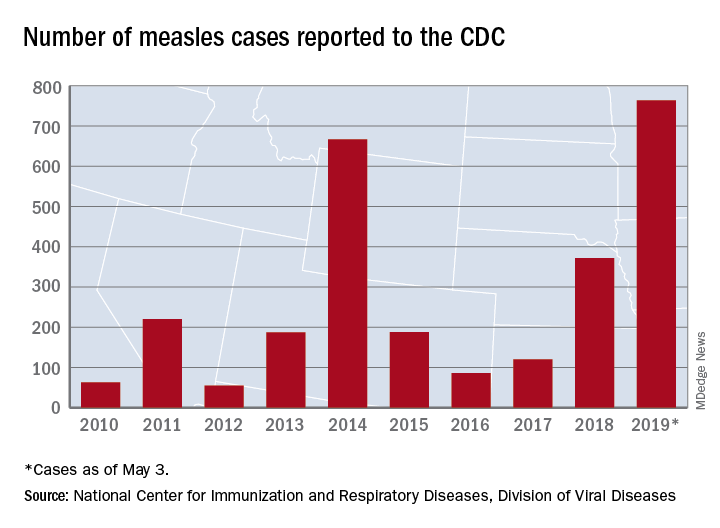
As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”
according to the Centers for Disease Control and Prevention.

As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”
according to the Centers for Disease Control and Prevention.

As of Wednesday, April 24, the case count for measles is 695, which eclipses the mark of 667 cases that had been the highest since the disease was declared to be eliminated from this country in 2000, the CDC reported.
“The high number of cases in 2019 is primarily the result of a few large outbreaks – one in Washington State and two large outbreaks in New York that started in late 2018. The outbreaks in New York City and New York State are among the largest and longest lasting since measles elimination in 2000. The longer these outbreaks continue, the greater the chance measles will again get a sustained foothold in the United States,” according to a written statement by the CDC.
Although these outbreaks began when the virus was brought into this country by unvaccinated travelers from other countries where there is widespread transmission, “a significant factor contributing to the outbreaks in New York is misinformation in the communities about the safety of the measles/mumps/rubella vaccine. Some organizations are deliberately targeting these communities with inaccurate and misleading information about vaccines,” according to the statement.
“Measles is not a harmless childhood illness, but a highly contagious, potentially life-threatening disease,” Health and Human Services Secretary Alex Azar said in a separate statement. “We have the ability to safely protect our children and our communities. Vaccines are a safe, highly effective public health solution that can prevent this disease. The measles vaccines are among the most extensively studied medical products we have, and their safety has been firmly established over many years in some of the largest vaccine studies ever undertaken. With a safe and effective vaccine that protects against measles, the suffering we are seeing is avoidable.”
CDC warns against misuse of opioid-prescribing guideline
Officials at the Centers for Disease Control and Prevention are warning against the misapplication of the agency’s 2016 guidelines on opioid prescribing, as well as clarifying dosage recommendations for patients starting or stopping pain medications.
In a perspective published in the New England Journal of Medicine on April 24, lead author Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control, conveyed concern that some policies and practices derived from the 2016 CDC Guideline for Prescribing Opioids for Chronic Pain are inconsistent with the recommendations and often go beyond their scope.
Misapplication examples include inappropriately applying the guideline to patients in active cancer treatment, patients experiencing acute sickle cell crises, or patients experiencing postsurgical pain, Dr. Dowell wrote.
The guideline offers guidance to clinicians treating chronic pain in adults who are already receiving opioids long-term at high dosages, she noted. It includes advice on maximizing nonopioid treatment, reviewing risks associated with continuing high-dose opioids, and collaborating with patients who agree to taper dosage, among other guidance.
Any application of the guideline’s dosage recommendation that results in hard limits or “cutting off” opioids is also an incorrect use of the recommendations, according to Dr. Dowell.
While the guideline advises clinicians to start opioids at the lowest effective dosage and avoid increasing dosage to 90 morphine milligram equivalents per day or more, that statement does not suggest discontinuation of opioids already prescribed at high dosages, according to the CDC’s clarification.
The guidance also does not apply to patients receiving or starting medication-assisted treatment for opioid use disorder.
The commentary comes after a trio of organizations raised concerns that insurers are inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology, raised the issue in a letter to the CDC in February. In response, Dr. Dowell clarified that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
In the perspective, Dr. Dowell wrote that the CDC is evaluating the intended and unintended impact of the 2016 opioid-prescribing guideline on clinician and patient outcomes and that the agency is committed to updating the recommendations when new evidence is available.
Officials at the Centers for Disease Control and Prevention are warning against the misapplication of the agency’s 2016 guidelines on opioid prescribing, as well as clarifying dosage recommendations for patients starting or stopping pain medications.
In a perspective published in the New England Journal of Medicine on April 24, lead author Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control, conveyed concern that some policies and practices derived from the 2016 CDC Guideline for Prescribing Opioids for Chronic Pain are inconsistent with the recommendations and often go beyond their scope.
Misapplication examples include inappropriately applying the guideline to patients in active cancer treatment, patients experiencing acute sickle cell crises, or patients experiencing postsurgical pain, Dr. Dowell wrote.
The guideline offers guidance to clinicians treating chronic pain in adults who are already receiving opioids long-term at high dosages, she noted. It includes advice on maximizing nonopioid treatment, reviewing risks associated with continuing high-dose opioids, and collaborating with patients who agree to taper dosage, among other guidance.
Any application of the guideline’s dosage recommendation that results in hard limits or “cutting off” opioids is also an incorrect use of the recommendations, according to Dr. Dowell.
While the guideline advises clinicians to start opioids at the lowest effective dosage and avoid increasing dosage to 90 morphine milligram equivalents per day or more, that statement does not suggest discontinuation of opioids already prescribed at high dosages, according to the CDC’s clarification.
The guidance also does not apply to patients receiving or starting medication-assisted treatment for opioid use disorder.
The commentary comes after a trio of organizations raised concerns that insurers are inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology, raised the issue in a letter to the CDC in February. In response, Dr. Dowell clarified that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
In the perspective, Dr. Dowell wrote that the CDC is evaluating the intended and unintended impact of the 2016 opioid-prescribing guideline on clinician and patient outcomes and that the agency is committed to updating the recommendations when new evidence is available.
Officials at the Centers for Disease Control and Prevention are warning against the misapplication of the agency’s 2016 guidelines on opioid prescribing, as well as clarifying dosage recommendations for patients starting or stopping pain medications.
In a perspective published in the New England Journal of Medicine on April 24, lead author Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control, conveyed concern that some policies and practices derived from the 2016 CDC Guideline for Prescribing Opioids for Chronic Pain are inconsistent with the recommendations and often go beyond their scope.
Misapplication examples include inappropriately applying the guideline to patients in active cancer treatment, patients experiencing acute sickle cell crises, or patients experiencing postsurgical pain, Dr. Dowell wrote.
The guideline offers guidance to clinicians treating chronic pain in adults who are already receiving opioids long-term at high dosages, she noted. It includes advice on maximizing nonopioid treatment, reviewing risks associated with continuing high-dose opioids, and collaborating with patients who agree to taper dosage, among other guidance.
Any application of the guideline’s dosage recommendation that results in hard limits or “cutting off” opioids is also an incorrect use of the recommendations, according to Dr. Dowell.
While the guideline advises clinicians to start opioids at the lowest effective dosage and avoid increasing dosage to 90 morphine milligram equivalents per day or more, that statement does not suggest discontinuation of opioids already prescribed at high dosages, according to the CDC’s clarification.
The guidance also does not apply to patients receiving or starting medication-assisted treatment for opioid use disorder.
The commentary comes after a trio of organizations raised concerns that insurers are inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology, raised the issue in a letter to the CDC in February. In response, Dr. Dowell clarified that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
In the perspective, Dr. Dowell wrote that the CDC is evaluating the intended and unintended impact of the 2016 opioid-prescribing guideline on clinician and patient outcomes and that the agency is committed to updating the recommendations when new evidence is available.
U.S. measles cases nearing postelimination-era high
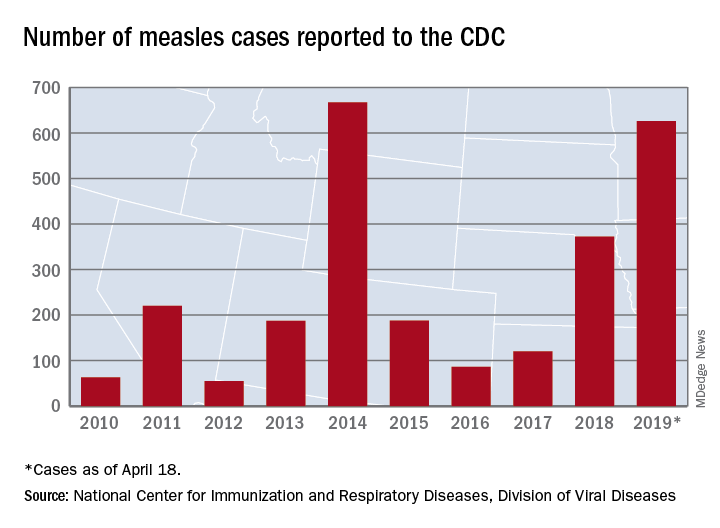
The United States has topped 600 cases of measles for 2019 and is likely to pass the postelimination high set in 2014 “in the coming weeks,” according to the Centers for Disease Control and Prevention.
The 71 new measles cases reported during the week ending April 18 bring the total for the year to 626 in 22 states, the CDC reported April 22. Two states, Iowa and Tennessee, reported their first cases last week.
Outbreaks continue in five states: one in California (Butte County), one in Michigan (Oakland County/Wayne County/Detroit), one in New Jersey (Ocean County/Monmouth County), two in New York (New York City and Rockland County), and one in Washington (Clark County/King County), the CDC said.
The most active outbreak since mid-February has been the one occurring in New York City, mainly in Brooklyn, and last week was no exception as 50 of the 71 new U.S. cases were reported in the borough.
On April 18, a judge in Brooklyn “ruled against a group of parents who challenged New York City’s recently imposed mandatory measles vaccination order,” Reuters reported. That same day, the city issued a summons, subject to a fine of $1,000 each, to three people in Brooklyn who were still unvaccinated, according to NYC Health, which also said that four additional schools would be closed for not complying with an order to exclude unvaccinated students.
On April 15, the Iowa Department of Public Health confirmed the state’s first case of measles since 2011. The individual from Northeastern Iowa had not been vaccinated and had recently returned from Israel. The state’s second case of the year, a household contact of the first individual, was confirmed on April 18.
Also on April 18, the Tennessee Department of Health confirmed its first case of the year in a resident of the eastern part of the state. Meanwhile, media are reporting that state health officials in Mississippi are investigating possible exposures on April 9 and 10 in the Hattiesburg area by the infected Tennessee man.
Outside the United States, “many countries are in the midst of sizeable measles outbreaks, with all regions of the world experiencing sustained rises in cases,” the World Health Organization said. Current outbreaks include the Democratic Republic of the Congo, Ethiopia, Georgia, Kazakhstan, Kyrgyzstan, Madagascar, Myanmar, Philippines, Sudan, Thailand, and Ukraine.
Preliminary data for the first 3 months of 2019 show that cases worldwide were up by 300% over the first 3 months of 2018: 112,163 cases vs. 28,124. The actual numbers for 2019 are expected to be considerably higher than those reported so far, and WHO estimates that, globally, less than 1 in 10 cases are actually reported.
The United States has topped 600 cases of measles for 2019 and is likely to pass the postelimination high set in 2014 “in the coming weeks,” according to the Centers for Disease Control and Prevention.
The 71 new measles cases reported during the week ending April 18 bring the total for the year to 626 in 22 states, the CDC reported April 22. Two states, Iowa and Tennessee, reported their first cases last week.
Outbreaks continue in five states: one in California (Butte County), one in Michigan (Oakland County/Wayne County/Detroit), one in New Jersey (Ocean County/Monmouth County), two in New York (New York City and Rockland County), and one in Washington (Clark County/King County), the CDC said.
The most active outbreak since mid-February has been the one occurring in New York City, mainly in Brooklyn, and last week was no exception as 50 of the 71 new U.S. cases were reported in the borough.
On April 18, a judge in Brooklyn “ruled against a group of parents who challenged New York City’s recently imposed mandatory measles vaccination order,” Reuters reported. That same day, the city issued a summons, subject to a fine of $1,000 each, to three people in Brooklyn who were still unvaccinated, according to NYC Health, which also said that four additional schools would be closed for not complying with an order to exclude unvaccinated students.
On April 15, the Iowa Department of Public Health confirmed the state’s first case of measles since 2011. The individual from Northeastern Iowa had not been vaccinated and had recently returned from Israel. The state’s second case of the year, a household contact of the first individual, was confirmed on April 18.
Also on April 18, the Tennessee Department of Health confirmed its first case of the year in a resident of the eastern part of the state. Meanwhile, media are reporting that state health officials in Mississippi are investigating possible exposures on April 9 and 10 in the Hattiesburg area by the infected Tennessee man.
Outside the United States, “many countries are in the midst of sizeable measles outbreaks, with all regions of the world experiencing sustained rises in cases,” the World Health Organization said. Current outbreaks include the Democratic Republic of the Congo, Ethiopia, Georgia, Kazakhstan, Kyrgyzstan, Madagascar, Myanmar, Philippines, Sudan, Thailand, and Ukraine.
Preliminary data for the first 3 months of 2019 show that cases worldwide were up by 300% over the first 3 months of 2018: 112,163 cases vs. 28,124. The actual numbers for 2019 are expected to be considerably higher than those reported so far, and WHO estimates that, globally, less than 1 in 10 cases are actually reported.
The United States has topped 600 cases of measles for 2019 and is likely to pass the postelimination high set in 2014 “in the coming weeks,” according to the Centers for Disease Control and Prevention.
The 71 new measles cases reported during the week ending April 18 bring the total for the year to 626 in 22 states, the CDC reported April 22. Two states, Iowa and Tennessee, reported their first cases last week.
Outbreaks continue in five states: one in California (Butte County), one in Michigan (Oakland County/Wayne County/Detroit), one in New Jersey (Ocean County/Monmouth County), two in New York (New York City and Rockland County), and one in Washington (Clark County/King County), the CDC said.
The most active outbreak since mid-February has been the one occurring in New York City, mainly in Brooklyn, and last week was no exception as 50 of the 71 new U.S. cases were reported in the borough.
On April 18, a judge in Brooklyn “ruled against a group of parents who challenged New York City’s recently imposed mandatory measles vaccination order,” Reuters reported. That same day, the city issued a summons, subject to a fine of $1,000 each, to three people in Brooklyn who were still unvaccinated, according to NYC Health, which also said that four additional schools would be closed for not complying with an order to exclude unvaccinated students.
On April 15, the Iowa Department of Public Health confirmed the state’s first case of measles since 2011. The individual from Northeastern Iowa had not been vaccinated and had recently returned from Israel. The state’s second case of the year, a household contact of the first individual, was confirmed on April 18.
Also on April 18, the Tennessee Department of Health confirmed its first case of the year in a resident of the eastern part of the state. Meanwhile, media are reporting that state health officials in Mississippi are investigating possible exposures on April 9 and 10 in the Hattiesburg area by the infected Tennessee man.
Outside the United States, “many countries are in the midst of sizeable measles outbreaks, with all regions of the world experiencing sustained rises in cases,” the World Health Organization said. Current outbreaks include the Democratic Republic of the Congo, Ethiopia, Georgia, Kazakhstan, Kyrgyzstan, Madagascar, Myanmar, Philippines, Sudan, Thailand, and Ukraine.
Preliminary data for the first 3 months of 2019 show that cases worldwide were up by 300% over the first 3 months of 2018: 112,163 cases vs. 28,124. The actual numbers for 2019 are expected to be considerably higher than those reported so far, and WHO estimates that, globally, less than 1 in 10 cases are actually reported.
2018 at a glance: Recently approved therapies in oncology
Advances in genomics and technology perpetually change and improve therapies in oncology. Enhanced comprehension of cellular signaling, division, and replication has created a platform to selectively restrict neoplastic growth while preserving the integrity of benign cells.
This article reviews therapies that were newly approved in 2018, as well as those previously approved whose indications were expanded this past year. The list highlights the most clinically important approvals, as well as adverse events that are unique or especially severe.
Click on the PDF above to download the full article and charts in an easy-to-print format.
Apalutamide (Erleada)
Class: Androgen receptor inhibitor.
Disease: Nonmetastatic castration-resistant prostate cancer.
Dose: 240 mg orally, once daily.
Adverse Events (AEs): Hyperkalemia and increased risks of seizures, falls, and fractures.
Phase 3 SPARTAN trial (NCT01946204): 40.5-month metastasis-free survival rate, compared with 16.2 months in the placebo group.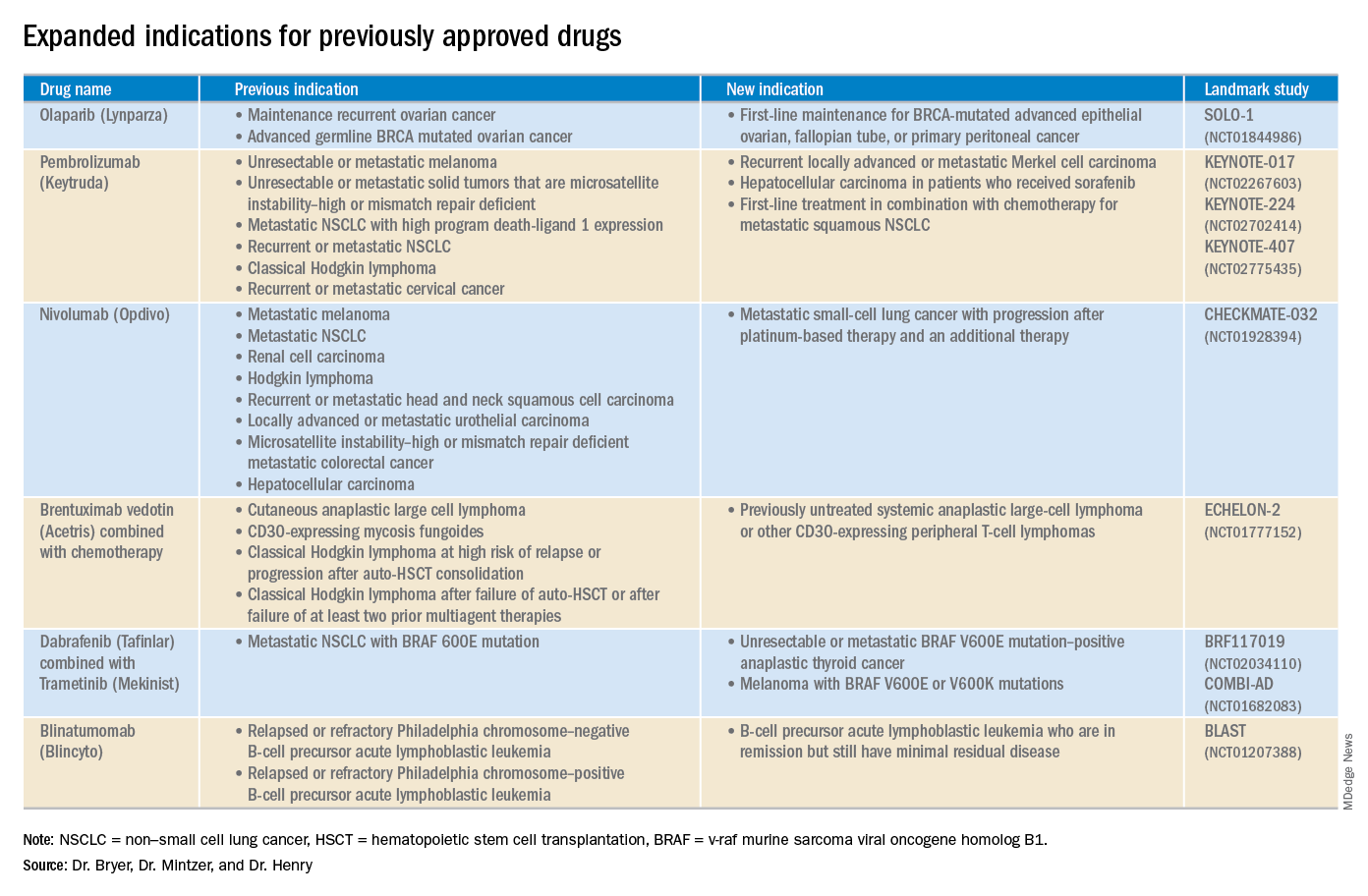
Cemiplimab (Libtayo)
Class: Antibody against programmed cell death protein-1 (PD-1).
Disease: Metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC that is ineligible for curative surgery/radiation.
Dose: 350 mg intravenous infusion every 3 weeks.
AEs: Pneumonitis, autoimmune myocarditis, hepatitis, and aseptic meningitis.
1423 and 1540 trials (NCT02383212 and NCT02760498): 47.2% of patients who received cemiplimab had complete disappearance of the tumor or a decrease in tumor size.
Dacomitinib (Vizimpro)
Class: Second-generation tyrosine kinase inhibitor.
Disease: Metastatic non–small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 19 deletion or exon 21 L858R substitution mutation.
Dose: 45 mg orally once daily.
AEs: Dermatotoxicity and diarrhea.
ARCHER1050 trial (NCT01774721): Patients who received dacomitinib demonstrated an improved overall survival, with a median of 34.1 months, compared with 26.8 months with gefitinib.
Duvelisib (Copiktra)
Class: Dual inhibitor of phosphatidylinositol 3-kinase delta and gamma.
Disease: Relapsed or refractory chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, or relapsed or refractory follicular lymphoma after at least two prior systemic therapies.
Dose: 25 mg orally twice daily.
AEs: Infection, diarrhea or colitis, and pneumonia.
Phase 3 DUO trial (NCT02004522): Progression-free survival in the duvelisib arm was 7.3 months longer than that in the ofatumumab arm. The overall response rate for patients receiving duvelisib was 78%, compared with 39% for those receiving ofatumumab.
Gilteritinib (Xospata)
Class: Inhibits the FLT3 internal tandem duplication (ITD) and FLT3 tyrosine kinase domain (TKD).
Disease: Relapsed or refractory acute myeloid leukemia (AML) with an FLT3 mutation.
Dose: 120 mg orally daily.
ADMIRAL trial (NCT02421939): 21% of the patients who received gilteritinib exhibited complete remission or complete remission with partial hematologic recovery.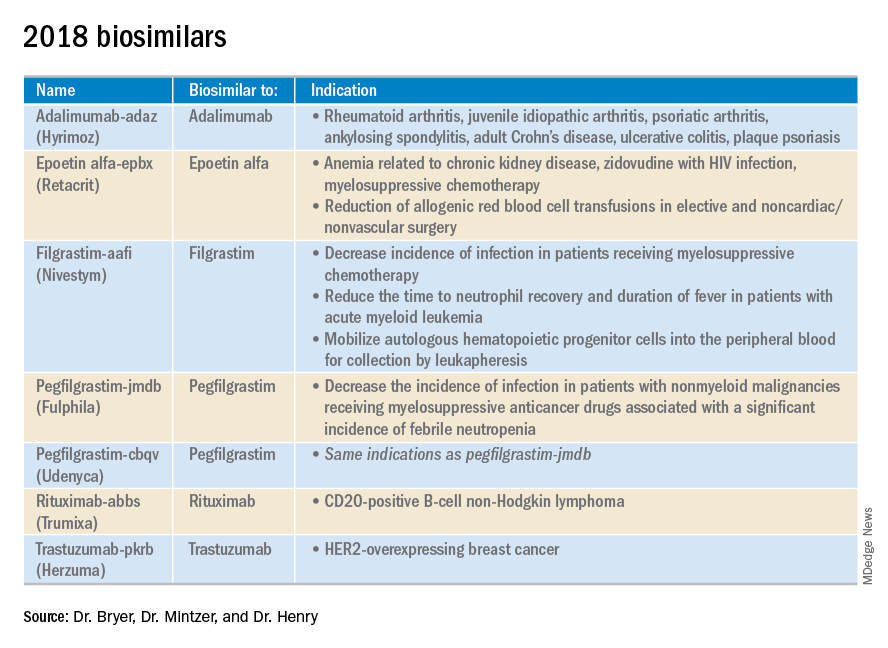
Glasdegib (Daurismo)
Class: Hedgehog pathway inhibitor.
Disease: Adults over age 75 years with newly diagnosed AML and other medical comorbidities that preclude them from intensive chemotherapy.
Dose: The recommended dose is 100 mg orally continuously in 28-day cycles.
AE: QT prolongation and embryo-fetal toxicity
Phase 2 BRIGHT 1003 trial (NCT01546038): 3.9-month overall survival advantage for glasdegib plus cytarabine, compared with cytarabine alone. Overall, 15% of the glasdegib plus low dose cytarabine arm achieved complete remission, compared with the 1% complete remission rate in patients who received cytarabine alone.
Iobenguane I 131 (Azedra)
Class: Radiopharmaceutical agent; induces cell death within the noradrenaline transporter.
Disease: Iobenguane scan–positive, unresectable, locally advanced or metastatic pheochromocytoma or paraganglioma
Dose: Initial intravenous dosimetric dose, followed by two therapeutic doses.
AE: Pancytopenia and elevated international normalized ratio (INR).
IB12B trial (NCT00874614): One-quarter of patients receiving this therapy had at least a 50% reduction in the dose and number of antihypertensives for at least 6 months; almost all patients had a tumor response.
Ivosidenib (Tibsovo)
Class: Small-molecule inhibitor of mutant isocitrate dehydrogenase (IDH1).
Disease: Refractory AML and an IDH1 mutation
Dose: 500 mg orally daily.
AG120-C-001 trial (NCT02074839): Overall response rate of 41.6% in patients who received ivosidenib, with a 30.4% rate of complete remission or complete remission with partial hematologic recovery.
Larotrectinib (Vitrakvi)
Class: Oral tyrosine kinase inhibitor.
Disease: Advanced solid tumors harboring a neurotrophic tyrosine receptor kinase (NTRK) gene fusion.
Dose: 100 mg orally twice daily.
LOXO-TRK-14001, SCOUT, and NAVIGATE trials (NCT02122913, NCT02637687, and NCT02576431): Patients who received larotrectinib had durable responses regardless of patient age, tumor type, and fusion status.
Lutetium Lu 177 dotatate (Lutathera)
Class: Radiolabeled somatostatin analogue.
Disease: Somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs).
Dose: Intravenous infusion 7.4 GBq (200 mCi) every 8 weeks for a total of four doses.
NETTER-1 trial (NCT01578239): 65% of adults who received lutetium Lu 177 showed improved progression-free survival at 20 months, compared with just 10.8% in the control group.
Mogamulizumab (Poteligeo)
Class: Monoclonal antibody that binds to a protein (CC chemokine receptor type 4).
Disease: Relapsed or refractory mycosis fungoides or Sézary syndrome.
Dose: Intravenous infusion 1 mg/kg.
AE: Dermatologic toxicity.
MAVORIC trial (NCT01728805): Patients who received mogamulizumab had improved progression-free survival (median 7.7 months), compared with those taking vorinostat (median 3.1 months).
Moxetumomab pasudotox-tdfk (Lumoxiti)
Class: CD22-directed cytotoxin fused with a fragment of Pseudomonas exotoxin A.
Disease: Relapsed or refractory hairy cell leukemia previously treated with at least two prior systemic therapies, including a purine nucleoside analogue.
Dose: Intravenously as 0.04 mg/kg.
AE: Hemolytic uremic syndrome.
1053 trial (NCT01829711): 30% of the patients who received moxetumomab pasudotox-tdfk had a durable complete response confirmed by maintenance hematologic remission.
Talazoparib (Talzenna)
Class: Poly (ADP-ribose) polymerase (PARP) inhibitor.
Disease: gBRCAm HER2-negative locally advanced or metastatic breast cancer.
Dose: 1 mg orally per day.
EMBRACA trial (NCT01945775): Patients who received talazoparib demonstrated significantly longer progression-free survival, with a median of 8.6 months versis 5.6 months in the control arm.
Dr. Bryer is a resident in the department of internal medicine at the University of Pennsylvania, Philadelphia. Dr. Mentzer is chief of hematology-oncology at Pennsylvania Hospital and professor of medicine at the University of Pennsylvania. Dr. Henry is a hematologist-oncologist at Pennsylvania Hospital and a professor of medicine at the University of Pennsylvania.
Advances in genomics and technology perpetually change and improve therapies in oncology. Enhanced comprehension of cellular signaling, division, and replication has created a platform to selectively restrict neoplastic growth while preserving the integrity of benign cells.
This article reviews therapies that were newly approved in 2018, as well as those previously approved whose indications were expanded this past year. The list highlights the most clinically important approvals, as well as adverse events that are unique or especially severe.
Click on the PDF above to download the full article and charts in an easy-to-print format.
Apalutamide (Erleada)
Class: Androgen receptor inhibitor.
Disease: Nonmetastatic castration-resistant prostate cancer.
Dose: 240 mg orally, once daily.
Adverse Events (AEs): Hyperkalemia and increased risks of seizures, falls, and fractures.
Phase 3 SPARTAN trial (NCT01946204): 40.5-month metastasis-free survival rate, compared with 16.2 months in the placebo group.
Cemiplimab (Libtayo)
Class: Antibody against programmed cell death protein-1 (PD-1).
Disease: Metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC that is ineligible for curative surgery/radiation.
Dose: 350 mg intravenous infusion every 3 weeks.
AEs: Pneumonitis, autoimmune myocarditis, hepatitis, and aseptic meningitis.
1423 and 1540 trials (NCT02383212 and NCT02760498): 47.2% of patients who received cemiplimab had complete disappearance of the tumor or a decrease in tumor size.
Dacomitinib (Vizimpro)
Class: Second-generation tyrosine kinase inhibitor.
Disease: Metastatic non–small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 19 deletion or exon 21 L858R substitution mutation.
Dose: 45 mg orally once daily.
AEs: Dermatotoxicity and diarrhea.
ARCHER1050 trial (NCT01774721): Patients who received dacomitinib demonstrated an improved overall survival, with a median of 34.1 months, compared with 26.8 months with gefitinib.
Duvelisib (Copiktra)
Class: Dual inhibitor of phosphatidylinositol 3-kinase delta and gamma.
Disease: Relapsed or refractory chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, or relapsed or refractory follicular lymphoma after at least two prior systemic therapies.
Dose: 25 mg orally twice daily.
AEs: Infection, diarrhea or colitis, and pneumonia.
Phase 3 DUO trial (NCT02004522): Progression-free survival in the duvelisib arm was 7.3 months longer than that in the ofatumumab arm. The overall response rate for patients receiving duvelisib was 78%, compared with 39% for those receiving ofatumumab.
Gilteritinib (Xospata)
Class: Inhibits the FLT3 internal tandem duplication (ITD) and FLT3 tyrosine kinase domain (TKD).
Disease: Relapsed or refractory acute myeloid leukemia (AML) with an FLT3 mutation.
Dose: 120 mg orally daily.
ADMIRAL trial (NCT02421939): 21% of the patients who received gilteritinib exhibited complete remission or complete remission with partial hematologic recovery.
Glasdegib (Daurismo)
Class: Hedgehog pathway inhibitor.
Disease: Adults over age 75 years with newly diagnosed AML and other medical comorbidities that preclude them from intensive chemotherapy.
Dose: The recommended dose is 100 mg orally continuously in 28-day cycles.
AE: QT prolongation and embryo-fetal toxicity
Phase 2 BRIGHT 1003 trial (NCT01546038): 3.9-month overall survival advantage for glasdegib plus cytarabine, compared with cytarabine alone. Overall, 15% of the glasdegib plus low dose cytarabine arm achieved complete remission, compared with the 1% complete remission rate in patients who received cytarabine alone.
Iobenguane I 131 (Azedra)
Class: Radiopharmaceutical agent; induces cell death within the noradrenaline transporter.
Disease: Iobenguane scan–positive, unresectable, locally advanced or metastatic pheochromocytoma or paraganglioma
Dose: Initial intravenous dosimetric dose, followed by two therapeutic doses.
AE: Pancytopenia and elevated international normalized ratio (INR).
IB12B trial (NCT00874614): One-quarter of patients receiving this therapy had at least a 50% reduction in the dose and number of antihypertensives for at least 6 months; almost all patients had a tumor response.
Ivosidenib (Tibsovo)
Class: Small-molecule inhibitor of mutant isocitrate dehydrogenase (IDH1).
Disease: Refractory AML and an IDH1 mutation
Dose: 500 mg orally daily.
AG120-C-001 trial (NCT02074839): Overall response rate of 41.6% in patients who received ivosidenib, with a 30.4% rate of complete remission or complete remission with partial hematologic recovery.
Larotrectinib (Vitrakvi)
Class: Oral tyrosine kinase inhibitor.
Disease: Advanced solid tumors harboring a neurotrophic tyrosine receptor kinase (NTRK) gene fusion.
Dose: 100 mg orally twice daily.
LOXO-TRK-14001, SCOUT, and NAVIGATE trials (NCT02122913, NCT02637687, and NCT02576431): Patients who received larotrectinib had durable responses regardless of patient age, tumor type, and fusion status.
Lutetium Lu 177 dotatate (Lutathera)
Class: Radiolabeled somatostatin analogue.
Disease: Somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs).
Dose: Intravenous infusion 7.4 GBq (200 mCi) every 8 weeks for a total of four doses.
NETTER-1 trial (NCT01578239): 65% of adults who received lutetium Lu 177 showed improved progression-free survival at 20 months, compared with just 10.8% in the control group.
Mogamulizumab (Poteligeo)
Class: Monoclonal antibody that binds to a protein (CC chemokine receptor type 4).
Disease: Relapsed or refractory mycosis fungoides or Sézary syndrome.
Dose: Intravenous infusion 1 mg/kg.
AE: Dermatologic toxicity.
MAVORIC trial (NCT01728805): Patients who received mogamulizumab had improved progression-free survival (median 7.7 months), compared with those taking vorinostat (median 3.1 months).
Moxetumomab pasudotox-tdfk (Lumoxiti)
Class: CD22-directed cytotoxin fused with a fragment of Pseudomonas exotoxin A.
Disease: Relapsed or refractory hairy cell leukemia previously treated with at least two prior systemic therapies, including a purine nucleoside analogue.
Dose: Intravenously as 0.04 mg/kg.
AE: Hemolytic uremic syndrome.
1053 trial (NCT01829711): 30% of the patients who received moxetumomab pasudotox-tdfk had a durable complete response confirmed by maintenance hematologic remission.
Talazoparib (Talzenna)
Class: Poly (ADP-ribose) polymerase (PARP) inhibitor.
Disease: gBRCAm HER2-negative locally advanced or metastatic breast cancer.
Dose: 1 mg orally per day.
EMBRACA trial (NCT01945775): Patients who received talazoparib demonstrated significantly longer progression-free survival, with a median of 8.6 months versis 5.6 months in the control arm.
Dr. Bryer is a resident in the department of internal medicine at the University of Pennsylvania, Philadelphia. Dr. Mentzer is chief of hematology-oncology at Pennsylvania Hospital and professor of medicine at the University of Pennsylvania. Dr. Henry is a hematologist-oncologist at Pennsylvania Hospital and a professor of medicine at the University of Pennsylvania.
Advances in genomics and technology perpetually change and improve therapies in oncology. Enhanced comprehension of cellular signaling, division, and replication has created a platform to selectively restrict neoplastic growth while preserving the integrity of benign cells.
This article reviews therapies that were newly approved in 2018, as well as those previously approved whose indications were expanded this past year. The list highlights the most clinically important approvals, as well as adverse events that are unique or especially severe.
Click on the PDF above to download the full article and charts in an easy-to-print format.
Apalutamide (Erleada)
Class: Androgen receptor inhibitor.
Disease: Nonmetastatic castration-resistant prostate cancer.
Dose: 240 mg orally, once daily.
Adverse Events (AEs): Hyperkalemia and increased risks of seizures, falls, and fractures.
Phase 3 SPARTAN trial (NCT01946204): 40.5-month metastasis-free survival rate, compared with 16.2 months in the placebo group.
Cemiplimab (Libtayo)
Class: Antibody against programmed cell death protein-1 (PD-1).
Disease: Metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC that is ineligible for curative surgery/radiation.
Dose: 350 mg intravenous infusion every 3 weeks.
AEs: Pneumonitis, autoimmune myocarditis, hepatitis, and aseptic meningitis.
1423 and 1540 trials (NCT02383212 and NCT02760498): 47.2% of patients who received cemiplimab had complete disappearance of the tumor or a decrease in tumor size.
Dacomitinib (Vizimpro)
Class: Second-generation tyrosine kinase inhibitor.
Disease: Metastatic non–small cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) exon 19 deletion or exon 21 L858R substitution mutation.
Dose: 45 mg orally once daily.
AEs: Dermatotoxicity and diarrhea.
ARCHER1050 trial (NCT01774721): Patients who received dacomitinib demonstrated an improved overall survival, with a median of 34.1 months, compared with 26.8 months with gefitinib.
Duvelisib (Copiktra)
Class: Dual inhibitor of phosphatidylinositol 3-kinase delta and gamma.
Disease: Relapsed or refractory chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, or relapsed or refractory follicular lymphoma after at least two prior systemic therapies.
Dose: 25 mg orally twice daily.
AEs: Infection, diarrhea or colitis, and pneumonia.
Phase 3 DUO trial (NCT02004522): Progression-free survival in the duvelisib arm was 7.3 months longer than that in the ofatumumab arm. The overall response rate for patients receiving duvelisib was 78%, compared with 39% for those receiving ofatumumab.
Gilteritinib (Xospata)
Class: Inhibits the FLT3 internal tandem duplication (ITD) and FLT3 tyrosine kinase domain (TKD).
Disease: Relapsed or refractory acute myeloid leukemia (AML) with an FLT3 mutation.
Dose: 120 mg orally daily.
ADMIRAL trial (NCT02421939): 21% of the patients who received gilteritinib exhibited complete remission or complete remission with partial hematologic recovery.
Glasdegib (Daurismo)
Class: Hedgehog pathway inhibitor.
Disease: Adults over age 75 years with newly diagnosed AML and other medical comorbidities that preclude them from intensive chemotherapy.
Dose: The recommended dose is 100 mg orally continuously in 28-day cycles.
AE: QT prolongation and embryo-fetal toxicity
Phase 2 BRIGHT 1003 trial (NCT01546038): 3.9-month overall survival advantage for glasdegib plus cytarabine, compared with cytarabine alone. Overall, 15% of the glasdegib plus low dose cytarabine arm achieved complete remission, compared with the 1% complete remission rate in patients who received cytarabine alone.
Iobenguane I 131 (Azedra)
Class: Radiopharmaceutical agent; induces cell death within the noradrenaline transporter.
Disease: Iobenguane scan–positive, unresectable, locally advanced or metastatic pheochromocytoma or paraganglioma
Dose: Initial intravenous dosimetric dose, followed by two therapeutic doses.
AE: Pancytopenia and elevated international normalized ratio (INR).
IB12B trial (NCT00874614): One-quarter of patients receiving this therapy had at least a 50% reduction in the dose and number of antihypertensives for at least 6 months; almost all patients had a tumor response.
Ivosidenib (Tibsovo)
Class: Small-molecule inhibitor of mutant isocitrate dehydrogenase (IDH1).
Disease: Refractory AML and an IDH1 mutation
Dose: 500 mg orally daily.
AG120-C-001 trial (NCT02074839): Overall response rate of 41.6% in patients who received ivosidenib, with a 30.4% rate of complete remission or complete remission with partial hematologic recovery.
Larotrectinib (Vitrakvi)
Class: Oral tyrosine kinase inhibitor.
Disease: Advanced solid tumors harboring a neurotrophic tyrosine receptor kinase (NTRK) gene fusion.
Dose: 100 mg orally twice daily.
LOXO-TRK-14001, SCOUT, and NAVIGATE trials (NCT02122913, NCT02637687, and NCT02576431): Patients who received larotrectinib had durable responses regardless of patient age, tumor type, and fusion status.
Lutetium Lu 177 dotatate (Lutathera)
Class: Radiolabeled somatostatin analogue.
Disease: Somatostatin receptor–positive gastroenteropancreatic neuroendocrine tumors (GEP-NETs).
Dose: Intravenous infusion 7.4 GBq (200 mCi) every 8 weeks for a total of four doses.
NETTER-1 trial (NCT01578239): 65% of adults who received lutetium Lu 177 showed improved progression-free survival at 20 months, compared with just 10.8% in the control group.
Mogamulizumab (Poteligeo)
Class: Monoclonal antibody that binds to a protein (CC chemokine receptor type 4).
Disease: Relapsed or refractory mycosis fungoides or Sézary syndrome.
Dose: Intravenous infusion 1 mg/kg.
AE: Dermatologic toxicity.
MAVORIC trial (NCT01728805): Patients who received mogamulizumab had improved progression-free survival (median 7.7 months), compared with those taking vorinostat (median 3.1 months).
Moxetumomab pasudotox-tdfk (Lumoxiti)
Class: CD22-directed cytotoxin fused with a fragment of Pseudomonas exotoxin A.
Disease: Relapsed or refractory hairy cell leukemia previously treated with at least two prior systemic therapies, including a purine nucleoside analogue.
Dose: Intravenously as 0.04 mg/kg.
AE: Hemolytic uremic syndrome.
1053 trial (NCT01829711): 30% of the patients who received moxetumomab pasudotox-tdfk had a durable complete response confirmed by maintenance hematologic remission.
Talazoparib (Talzenna)
Class: Poly (ADP-ribose) polymerase (PARP) inhibitor.
Disease: gBRCAm HER2-negative locally advanced or metastatic breast cancer.
Dose: 1 mg orally per day.
EMBRACA trial (NCT01945775): Patients who received talazoparib demonstrated significantly longer progression-free survival, with a median of 8.6 months versis 5.6 months in the control arm.
Dr. Bryer is a resident in the department of internal medicine at the University of Pennsylvania, Philadelphia. Dr. Mentzer is chief of hematology-oncology at Pennsylvania Hospital and professor of medicine at the University of Pennsylvania. Dr. Henry is a hematologist-oncologist at Pennsylvania Hospital and a professor of medicine at the University of Pennsylvania.
Busiest week yet brings 2019 measles total to 555 cases
according to the Centers for Disease Control and Prevention.
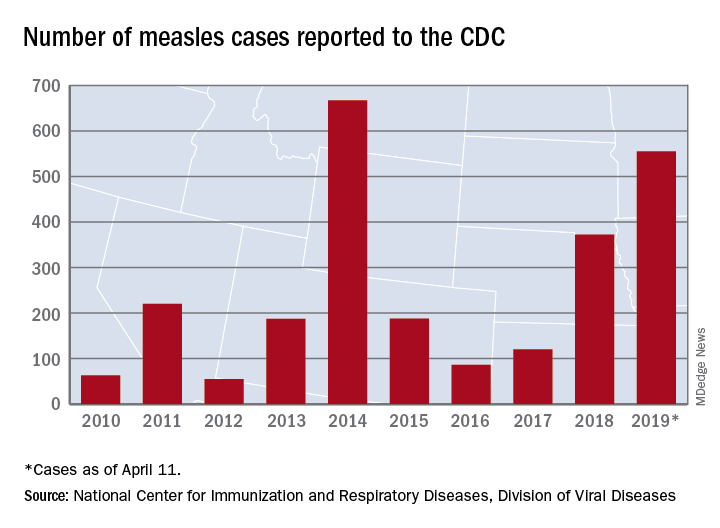
The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
according to the Centers for Disease Control and Prevention.

The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
according to the Centers for Disease Control and Prevention.

The 90 measles cases reported during the week ending April 11 mark the third consecutive weekly high for 2019, topping the 78 recorded during the week of April 4 and the 73 reported during the week of March 28. Meanwhile, this year’s total trails only the 667 cases reported in 2014 for the highest in the postelimination era, the CDC said April 15.
New York reported 26 new cases in Brooklyn’s Williamsburg neighborhood last week, which puts the borough at 227 for the year, with another two occurring in the Flushing section of Queens. A public health emergency declared on April 9 covers several zip codes in Williamsburg and requires unvaccinated individuals who may have been exposed to measles to receive “the measles-mumps-rubella vaccine in order to protect others in the community and help curtail the ongoing outbreak,” the city’s health department said in a written statement.
Maryland became the 20th state to report a measles case this year, and the state’s department of health said it was notifying those in the vicinity of a medical office building in Pikesville about possible exposure on April 2.
The recent outbreak in Michigan’s Oakland County did not result in any new patients over the last week and remains at 38 cases, with the state reporting one additional case in Wayne County. More recent reports of a case in Washtenaw County and another in Oakland County were reversed after additional testing, the state health department reported.
CDC clarifies opioid prescribing guidelines in cancer, sickle cell disease
Officials at the Centers for Disease Control and Prevention have clarified the agency’s guidelines on opioid prescribing after a trio of organizations raised concerns that insurers were inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The CDC guidelines, released in March 2016, address when to initiate or continue opioids for chronic pain, opioid selection, dosage, duration, follow-up, and discontinuation, and assess risk and harms of opioid use. Although the guidelines clearly state they are intended for clinicians prescribing opioids outside of active cancer treatment, insurance companies are still applying the guidelines to opioid coverage decisions for patients with active cancer, according to a Feb. 13, 2019, letter sent to the CDC from leaders at the American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology.
Additionally, the associations wrote that the CDC’s recommendations pose coverage problems for sickle cell patients and select groups of cancer survivors who may benefit from opioids for pain management. The groups asked the CDC to issue a clarification to ensure appropriate implementation of the opioid recommendations.
In a Feb. 28, 2019, letter to ASCO, NCCN, and ASH, Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control took note of the concerns, clarifying that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
The CDC guidance may apply to cancer survivors in certain conditions, Dr. Dowell wrote, namely when survivors experience chronic pain after cancer treatment completion, are in clinical remission, and are under cancer surveillance only. However, she agreed that, for select groups of cancer survivors with persistent pain caused by past cancer, the ratio of opioid benefits to risks for chronic pain is unique. She referred health providers to guidelines by ASCO on chronic pain management for adult cancer survivors and NCCN guidance on managing adult cancer pain when considering opioids for pain control in such populations.
Special considerations in sickle cell disease may also change the balance of opioid risks to benefits for pain management, Dr. Dowell wrote, referring providers and insurers to additional guidance on sickle cell disease from the National Institute of Health when making treatment and reimbursement decisions.
“Clinical decision making should be based on the relationship between the clinician and patient, with an understanding of the patient’s clinical situation, functioning, and life context, as well as careful consideration of the benefits and risk of all treatment options, including opioid therapy,” Dr. Dowell wrote. “CDC encourages physicians to continue using their clinical judgment and base treatment on what they know about their patients, including the use of opioids if determined to be the best course of treatment.”
Clifford A. Hudis, MD, CEO of ASCO, praised the clarification, calling the letter necessary to clear up confusion and prevent inappropriate coverage decisions.
“This clarification from CDC is critically important because, while the agency’s guideline clearly states that it is not intended to apply to patients during active cancer and sickle cell disease treatment, many payers have been inappropriately using it to make opioid coverage determinations for those exact populations,” Dr. Hudis said in a statement.
Sickle cell patients suffer from severe, chronic pain, which is debilitating on its own without the added burden of having to constantly appeal coverage denials, added ASH President Roy Silverstein, MD.
“We appreciate CDC’s acknowledgment that the challenges of managing severe and chronic pain in conditions, such as sickle cell disease, require special consideration, and we hope payers will take the CDC’s clarification into account to ensure that patients’ pain management needs are covered,” he said in the same statement.
Officials at the Centers for Disease Control and Prevention have clarified the agency’s guidelines on opioid prescribing after a trio of organizations raised concerns that insurers were inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The CDC guidelines, released in March 2016, address when to initiate or continue opioids for chronic pain, opioid selection, dosage, duration, follow-up, and discontinuation, and assess risk and harms of opioid use. Although the guidelines clearly state they are intended for clinicians prescribing opioids outside of active cancer treatment, insurance companies are still applying the guidelines to opioid coverage decisions for patients with active cancer, according to a Feb. 13, 2019, letter sent to the CDC from leaders at the American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology.
Additionally, the associations wrote that the CDC’s recommendations pose coverage problems for sickle cell patients and select groups of cancer survivors who may benefit from opioids for pain management. The groups asked the CDC to issue a clarification to ensure appropriate implementation of the opioid recommendations.
In a Feb. 28, 2019, letter to ASCO, NCCN, and ASH, Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control took note of the concerns, clarifying that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
The CDC guidance may apply to cancer survivors in certain conditions, Dr. Dowell wrote, namely when survivors experience chronic pain after cancer treatment completion, are in clinical remission, and are under cancer surveillance only. However, she agreed that, for select groups of cancer survivors with persistent pain caused by past cancer, the ratio of opioid benefits to risks for chronic pain is unique. She referred health providers to guidelines by ASCO on chronic pain management for adult cancer survivors and NCCN guidance on managing adult cancer pain when considering opioids for pain control in such populations.
Special considerations in sickle cell disease may also change the balance of opioid risks to benefits for pain management, Dr. Dowell wrote, referring providers and insurers to additional guidance on sickle cell disease from the National Institute of Health when making treatment and reimbursement decisions.
“Clinical decision making should be based on the relationship between the clinician and patient, with an understanding of the patient’s clinical situation, functioning, and life context, as well as careful consideration of the benefits and risk of all treatment options, including opioid therapy,” Dr. Dowell wrote. “CDC encourages physicians to continue using their clinical judgment and base treatment on what they know about their patients, including the use of opioids if determined to be the best course of treatment.”
Clifford A. Hudis, MD, CEO of ASCO, praised the clarification, calling the letter necessary to clear up confusion and prevent inappropriate coverage decisions.
“This clarification from CDC is critically important because, while the agency’s guideline clearly states that it is not intended to apply to patients during active cancer and sickle cell disease treatment, many payers have been inappropriately using it to make opioid coverage determinations for those exact populations,” Dr. Hudis said in a statement.
Sickle cell patients suffer from severe, chronic pain, which is debilitating on its own without the added burden of having to constantly appeal coverage denials, added ASH President Roy Silverstein, MD.
“We appreciate CDC’s acknowledgment that the challenges of managing severe and chronic pain in conditions, such as sickle cell disease, require special consideration, and we hope payers will take the CDC’s clarification into account to ensure that patients’ pain management needs are covered,” he said in the same statement.
Officials at the Centers for Disease Control and Prevention have clarified the agency’s guidelines on opioid prescribing after a trio of organizations raised concerns that insurers were inappropriately applying the recommendations to active cancer patients when making coverage determinations.
The CDC guidelines, released in March 2016, address when to initiate or continue opioids for chronic pain, opioid selection, dosage, duration, follow-up, and discontinuation, and assess risk and harms of opioid use. Although the guidelines clearly state they are intended for clinicians prescribing opioids outside of active cancer treatment, insurance companies are still applying the guidelines to opioid coverage decisions for patients with active cancer, according to a Feb. 13, 2019, letter sent to the CDC from leaders at the American Society of Clinical Oncology, the National Comprehensive Cancer Network, and the American Society of Hematology.
Additionally, the associations wrote that the CDC’s recommendations pose coverage problems for sickle cell patients and select groups of cancer survivors who may benefit from opioids for pain management. The groups asked the CDC to issue a clarification to ensure appropriate implementation of the opioid recommendations.
In a Feb. 28, 2019, letter to ASCO, NCCN, and ASH, Deborah Dowell, MD, chief medical officer for the CDC’s National Center for Injury Prevention and Control took note of the concerns, clarifying that the recommendations are not intended to deny clinically appropriate opioid therapy to any patients who suffer chronic pain, but rather to ensure that physicians and patients consider all safe and effective treatment options.
The CDC guidance may apply to cancer survivors in certain conditions, Dr. Dowell wrote, namely when survivors experience chronic pain after cancer treatment completion, are in clinical remission, and are under cancer surveillance only. However, she agreed that, for select groups of cancer survivors with persistent pain caused by past cancer, the ratio of opioid benefits to risks for chronic pain is unique. She referred health providers to guidelines by ASCO on chronic pain management for adult cancer survivors and NCCN guidance on managing adult cancer pain when considering opioids for pain control in such populations.
Special considerations in sickle cell disease may also change the balance of opioid risks to benefits for pain management, Dr. Dowell wrote, referring providers and insurers to additional guidance on sickle cell disease from the National Institute of Health when making treatment and reimbursement decisions.
“Clinical decision making should be based on the relationship between the clinician and patient, with an understanding of the patient’s clinical situation, functioning, and life context, as well as careful consideration of the benefits and risk of all treatment options, including opioid therapy,” Dr. Dowell wrote. “CDC encourages physicians to continue using their clinical judgment and base treatment on what they know about their patients, including the use of opioids if determined to be the best course of treatment.”
Clifford A. Hudis, MD, CEO of ASCO, praised the clarification, calling the letter necessary to clear up confusion and prevent inappropriate coverage decisions.
“This clarification from CDC is critically important because, while the agency’s guideline clearly states that it is not intended to apply to patients during active cancer and sickle cell disease treatment, many payers have been inappropriately using it to make opioid coverage determinations for those exact populations,” Dr. Hudis said in a statement.
Sickle cell patients suffer from severe, chronic pain, which is debilitating on its own without the added burden of having to constantly appeal coverage denials, added ASH President Roy Silverstein, MD.
“We appreciate CDC’s acknowledgment that the challenges of managing severe and chronic pain in conditions, such as sickle cell disease, require special consideration, and we hope payers will take the CDC’s clarification into account to ensure that patients’ pain management needs are covered,” he said in the same statement.