User login
HRT may prevent Alzheimer’s in high-risk women
new research suggests.
Results from a cohort study of almost 1,200 women showed that use of HRT was associated with higher delayed memory scores and larger entorhinal and hippocampal brain volumes – areas that are affected early by Alzheimer’s disease (AD) pathology.
HRT was also found to be most effective, as seen by larger hippocampal volume, when introduced during early perimenopause.
“Clinicians are very much aware of the susceptibility of women to cognitive disturbances during menopause,” lead author Rasha Saleh, MD, senior research associate, University of East Anglia (England), said in an interview.
“Identifying the at-risk APOE4 women and early HRT introduction can be of benefit. Confirming our findings in a clinical trial would be the next step forward,” Dr. Saleh said.
The findings were published online in Alzheimer’s Research and Therapy.
Personalized approaches
Dr. Saleh noted that estrogen receptors are localized in various areas of the brain, including cognition-related areas. Estrogen regulates such things as neuroinflammatory status, glucose utilization, and lipid metabolism.
“The decline of estrogen during menopause can lead to disturbance in these functions, which can accelerate AD-related pathology,” she said.
HRT during the menopausal transition and afterward is “being considered as a strategy to mitigate cognitive decline,” the investigators wrote. Early observational studies have suggested that oral estrogen “may be protective against dementia,” but results of clinical trials have been inconsistent, and some have even shown “harmful effects.”
The current researchers were “interested in the personalized approaches in the prevention of AD,” Dr. Saleh said. Preclinical and pilot data from her group have shown that women with APOE4 have “better cognitive test scores with nutritional and hormonal interventions.”
This led Dr. Saleh to hypothesize that HRT would be of more cognitive benefit for those with versus without APOE4, particularly when introduced early during the menopausal transition.
To investigate this hypothesis, the researchers analyzed baseline data from participants in the European Prevention of Alzheimer’s Dementia (EPAD) cohort. This project was initiated in 2015 with the aim of developing longitudinal models over the entire course of AD prior to dementia clinical diagnosis.
Participants were recruited from 10 European countries. All were required to be at least 50 years old, to have not been diagnosed with dementia at baseline, and to have no medical or psychiatric illness that could potentially exclude them from further research.
The current study included 1,178 women (mean age, 65.1 years), who were divided by genotype into non-APOE4 and APOE4 groups. HRT treatment for current or previous users included estrogen alone or estrogen plus progestogens via oral or transdermal administration routes, and at different doses.
The four tests used to assess cognition were the Mini-Mental State Examination dot counting to evaluate verbal working memory, the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) total score, the Four Mountain Test, and the supermarket trolley virtual reality test.
Brain MRI data were collected. The researchers focused on the medial temporal lobe as the “main brain region regulating cognition and memory processing.” This lobe includes the hippocampus, the parahippocampus, the entorhinal cortex, and the amygdala.
‘Critical window’
The researchers found a “trend” toward an APOE-HRT interaction (P-interaction = .097) for the total RBANS score. In particular, it was significant for the RBANS delayed memory index, where scores were consistently higher for women with APOE4 who had received HRT, compared with all other groups (P-interaction = .009).
Within-genotype group comparisons showed that HRT users had a higher RBANS total scale score and delayed memory index (P = .045 and P = .002, respectively), but only among APOE4 carriers. Effect size analyses showed a large effect of HRT use on the Four Mountain Test score and the supermarket trolley virtual reality test score (Cohen’s d = 0.988 and 1.2, respectively).
“This large effect was found only in APOE4 carriers,” the investigators noted.
Similarly, a moderate to large effect of HRT on the left entorhinal volume was observed in APOE4 carriers (Cohen’s d = 0.63).
In members of the APOE4 group who received HRT, the left entorhinal and left and right amygdala volumes were larger, compared with both no-APOE4 and non-HRT users (P-interaction = .002, .003, and .005, respectively). Similar trends were observed for the right entorhinal volume (P = .074).
In addition, among HRT users, the left entorhinal volume was larger (P = .03); the right and left anterior cingulate gyrus volumes were smaller (P = .003 and .062, respectively); and the left superior frontal gyrus volume was larger (P = .009) in comparison with women who did not receive HRT, independently of their APOE genotype.
Early use of HRT among APOE4 carriers was associated with larger right and left hippocampal volume (P = .035 and P = .028, respectively) – an association not found in non-APOE4 carriers. The association was also not significant when participants were not stratified by APOE genotype.
“The key important point here is the timing, or the ‘critical window,’ when HRT can be of most benefit,” Dr. Saleh said. “This is most beneficial when introduced early, before the neuropathology becomes irreversible.”
Study limitations include its cross-sectional design, which precludes the establishment of a causal relationship, and the fact that information regarding the type and dose of estrogen was not available for all participants.
HRT is not without risk, Dr. Saleh noted. She recommended that clinicians “carry out various screening tests to make sure that a woman is eligible for HRT and not at risk of hypercoagulability, for instance.”
Risk-benefit ratio
In a comment, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, called the study “exactly the kind of work that needs to be done.”
Dr. Fillit, who was not involved with the current research, is a clinical professor of geriatric medicine, palliative care medicine, and neuroscience at Mount Sinai Hospital, New York.
He compared the process with that of osteoporosis. “We know that if women are treated [with HRT] at the time of the menopause, you can prevent the rapid bone loss that occurs with rapid estrogen loss. But if you wait 5, 10 years out, once the bone loss has occurred, the HRT doesn’t really have any impact on osteoporosis risk because the horse is already out of the barn,” he said.
Although HRT carries risks, “they can clearly be managed; and if it’s proven that estrogen or hormone replacement around the time of the menopause can be protective [against AD], the risk-benefit ratio of HRT could be in favor of treatment,” Dr. Fillit added.
The study was conducted as part of the Medical Research Council NuBrain Consortium. The investigators and Dr. Fillit reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Results from a cohort study of almost 1,200 women showed that use of HRT was associated with higher delayed memory scores and larger entorhinal and hippocampal brain volumes – areas that are affected early by Alzheimer’s disease (AD) pathology.
HRT was also found to be most effective, as seen by larger hippocampal volume, when introduced during early perimenopause.
“Clinicians are very much aware of the susceptibility of women to cognitive disturbances during menopause,” lead author Rasha Saleh, MD, senior research associate, University of East Anglia (England), said in an interview.
“Identifying the at-risk APOE4 women and early HRT introduction can be of benefit. Confirming our findings in a clinical trial would be the next step forward,” Dr. Saleh said.
The findings were published online in Alzheimer’s Research and Therapy.
Personalized approaches
Dr. Saleh noted that estrogen receptors are localized in various areas of the brain, including cognition-related areas. Estrogen regulates such things as neuroinflammatory status, glucose utilization, and lipid metabolism.
“The decline of estrogen during menopause can lead to disturbance in these functions, which can accelerate AD-related pathology,” she said.
HRT during the menopausal transition and afterward is “being considered as a strategy to mitigate cognitive decline,” the investigators wrote. Early observational studies have suggested that oral estrogen “may be protective against dementia,” but results of clinical trials have been inconsistent, and some have even shown “harmful effects.”
The current researchers were “interested in the personalized approaches in the prevention of AD,” Dr. Saleh said. Preclinical and pilot data from her group have shown that women with APOE4 have “better cognitive test scores with nutritional and hormonal interventions.”
This led Dr. Saleh to hypothesize that HRT would be of more cognitive benefit for those with versus without APOE4, particularly when introduced early during the menopausal transition.
To investigate this hypothesis, the researchers analyzed baseline data from participants in the European Prevention of Alzheimer’s Dementia (EPAD) cohort. This project was initiated in 2015 with the aim of developing longitudinal models over the entire course of AD prior to dementia clinical diagnosis.
Participants were recruited from 10 European countries. All were required to be at least 50 years old, to have not been diagnosed with dementia at baseline, and to have no medical or psychiatric illness that could potentially exclude them from further research.
The current study included 1,178 women (mean age, 65.1 years), who were divided by genotype into non-APOE4 and APOE4 groups. HRT treatment for current or previous users included estrogen alone or estrogen plus progestogens via oral or transdermal administration routes, and at different doses.
The four tests used to assess cognition were the Mini-Mental State Examination dot counting to evaluate verbal working memory, the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) total score, the Four Mountain Test, and the supermarket trolley virtual reality test.
Brain MRI data were collected. The researchers focused on the medial temporal lobe as the “main brain region regulating cognition and memory processing.” This lobe includes the hippocampus, the parahippocampus, the entorhinal cortex, and the amygdala.
‘Critical window’
The researchers found a “trend” toward an APOE-HRT interaction (P-interaction = .097) for the total RBANS score. In particular, it was significant for the RBANS delayed memory index, where scores were consistently higher for women with APOE4 who had received HRT, compared with all other groups (P-interaction = .009).
Within-genotype group comparisons showed that HRT users had a higher RBANS total scale score and delayed memory index (P = .045 and P = .002, respectively), but only among APOE4 carriers. Effect size analyses showed a large effect of HRT use on the Four Mountain Test score and the supermarket trolley virtual reality test score (Cohen’s d = 0.988 and 1.2, respectively).
“This large effect was found only in APOE4 carriers,” the investigators noted.
Similarly, a moderate to large effect of HRT on the left entorhinal volume was observed in APOE4 carriers (Cohen’s d = 0.63).
In members of the APOE4 group who received HRT, the left entorhinal and left and right amygdala volumes were larger, compared with both no-APOE4 and non-HRT users (P-interaction = .002, .003, and .005, respectively). Similar trends were observed for the right entorhinal volume (P = .074).
In addition, among HRT users, the left entorhinal volume was larger (P = .03); the right and left anterior cingulate gyrus volumes were smaller (P = .003 and .062, respectively); and the left superior frontal gyrus volume was larger (P = .009) in comparison with women who did not receive HRT, independently of their APOE genotype.
Early use of HRT among APOE4 carriers was associated with larger right and left hippocampal volume (P = .035 and P = .028, respectively) – an association not found in non-APOE4 carriers. The association was also not significant when participants were not stratified by APOE genotype.
“The key important point here is the timing, or the ‘critical window,’ when HRT can be of most benefit,” Dr. Saleh said. “This is most beneficial when introduced early, before the neuropathology becomes irreversible.”
Study limitations include its cross-sectional design, which precludes the establishment of a causal relationship, and the fact that information regarding the type and dose of estrogen was not available for all participants.
HRT is not without risk, Dr. Saleh noted. She recommended that clinicians “carry out various screening tests to make sure that a woman is eligible for HRT and not at risk of hypercoagulability, for instance.”
Risk-benefit ratio
In a comment, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, called the study “exactly the kind of work that needs to be done.”
Dr. Fillit, who was not involved with the current research, is a clinical professor of geriatric medicine, palliative care medicine, and neuroscience at Mount Sinai Hospital, New York.
He compared the process with that of osteoporosis. “We know that if women are treated [with HRT] at the time of the menopause, you can prevent the rapid bone loss that occurs with rapid estrogen loss. But if you wait 5, 10 years out, once the bone loss has occurred, the HRT doesn’t really have any impact on osteoporosis risk because the horse is already out of the barn,” he said.
Although HRT carries risks, “they can clearly be managed; and if it’s proven that estrogen or hormone replacement around the time of the menopause can be protective [against AD], the risk-benefit ratio of HRT could be in favor of treatment,” Dr. Fillit added.
The study was conducted as part of the Medical Research Council NuBrain Consortium. The investigators and Dr. Fillit reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
new research suggests.
Results from a cohort study of almost 1,200 women showed that use of HRT was associated with higher delayed memory scores and larger entorhinal and hippocampal brain volumes – areas that are affected early by Alzheimer’s disease (AD) pathology.
HRT was also found to be most effective, as seen by larger hippocampal volume, when introduced during early perimenopause.
“Clinicians are very much aware of the susceptibility of women to cognitive disturbances during menopause,” lead author Rasha Saleh, MD, senior research associate, University of East Anglia (England), said in an interview.
“Identifying the at-risk APOE4 women and early HRT introduction can be of benefit. Confirming our findings in a clinical trial would be the next step forward,” Dr. Saleh said.
The findings were published online in Alzheimer’s Research and Therapy.
Personalized approaches
Dr. Saleh noted that estrogen receptors are localized in various areas of the brain, including cognition-related areas. Estrogen regulates such things as neuroinflammatory status, glucose utilization, and lipid metabolism.
“The decline of estrogen during menopause can lead to disturbance in these functions, which can accelerate AD-related pathology,” she said.
HRT during the menopausal transition and afterward is “being considered as a strategy to mitigate cognitive decline,” the investigators wrote. Early observational studies have suggested that oral estrogen “may be protective against dementia,” but results of clinical trials have been inconsistent, and some have even shown “harmful effects.”
The current researchers were “interested in the personalized approaches in the prevention of AD,” Dr. Saleh said. Preclinical and pilot data from her group have shown that women with APOE4 have “better cognitive test scores with nutritional and hormonal interventions.”
This led Dr. Saleh to hypothesize that HRT would be of more cognitive benefit for those with versus without APOE4, particularly when introduced early during the menopausal transition.
To investigate this hypothesis, the researchers analyzed baseline data from participants in the European Prevention of Alzheimer’s Dementia (EPAD) cohort. This project was initiated in 2015 with the aim of developing longitudinal models over the entire course of AD prior to dementia clinical diagnosis.
Participants were recruited from 10 European countries. All were required to be at least 50 years old, to have not been diagnosed with dementia at baseline, and to have no medical or psychiatric illness that could potentially exclude them from further research.
The current study included 1,178 women (mean age, 65.1 years), who were divided by genotype into non-APOE4 and APOE4 groups. HRT treatment for current or previous users included estrogen alone or estrogen plus progestogens via oral or transdermal administration routes, and at different doses.
The four tests used to assess cognition were the Mini-Mental State Examination dot counting to evaluate verbal working memory, the Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) total score, the Four Mountain Test, and the supermarket trolley virtual reality test.
Brain MRI data were collected. The researchers focused on the medial temporal lobe as the “main brain region regulating cognition and memory processing.” This lobe includes the hippocampus, the parahippocampus, the entorhinal cortex, and the amygdala.
‘Critical window’
The researchers found a “trend” toward an APOE-HRT interaction (P-interaction = .097) for the total RBANS score. In particular, it was significant for the RBANS delayed memory index, where scores were consistently higher for women with APOE4 who had received HRT, compared with all other groups (P-interaction = .009).
Within-genotype group comparisons showed that HRT users had a higher RBANS total scale score and delayed memory index (P = .045 and P = .002, respectively), but only among APOE4 carriers. Effect size analyses showed a large effect of HRT use on the Four Mountain Test score and the supermarket trolley virtual reality test score (Cohen’s d = 0.988 and 1.2, respectively).
“This large effect was found only in APOE4 carriers,” the investigators noted.
Similarly, a moderate to large effect of HRT on the left entorhinal volume was observed in APOE4 carriers (Cohen’s d = 0.63).
In members of the APOE4 group who received HRT, the left entorhinal and left and right amygdala volumes were larger, compared with both no-APOE4 and non-HRT users (P-interaction = .002, .003, and .005, respectively). Similar trends were observed for the right entorhinal volume (P = .074).
In addition, among HRT users, the left entorhinal volume was larger (P = .03); the right and left anterior cingulate gyrus volumes were smaller (P = .003 and .062, respectively); and the left superior frontal gyrus volume was larger (P = .009) in comparison with women who did not receive HRT, independently of their APOE genotype.
Early use of HRT among APOE4 carriers was associated with larger right and left hippocampal volume (P = .035 and P = .028, respectively) – an association not found in non-APOE4 carriers. The association was also not significant when participants were not stratified by APOE genotype.
“The key important point here is the timing, or the ‘critical window,’ when HRT can be of most benefit,” Dr. Saleh said. “This is most beneficial when introduced early, before the neuropathology becomes irreversible.”
Study limitations include its cross-sectional design, which precludes the establishment of a causal relationship, and the fact that information regarding the type and dose of estrogen was not available for all participants.
HRT is not without risk, Dr. Saleh noted. She recommended that clinicians “carry out various screening tests to make sure that a woman is eligible for HRT and not at risk of hypercoagulability, for instance.”
Risk-benefit ratio
In a comment, Howard Fillit, MD, cofounder and chief science officer at the Alzheimer’s Drug Discovery Foundation, called the study “exactly the kind of work that needs to be done.”
Dr. Fillit, who was not involved with the current research, is a clinical professor of geriatric medicine, palliative care medicine, and neuroscience at Mount Sinai Hospital, New York.
He compared the process with that of osteoporosis. “We know that if women are treated [with HRT] at the time of the menopause, you can prevent the rapid bone loss that occurs with rapid estrogen loss. But if you wait 5, 10 years out, once the bone loss has occurred, the HRT doesn’t really have any impact on osteoporosis risk because the horse is already out of the barn,” he said.
Although HRT carries risks, “they can clearly be managed; and if it’s proven that estrogen or hormone replacement around the time of the menopause can be protective [against AD], the risk-benefit ratio of HRT could be in favor of treatment,” Dr. Fillit added.
The study was conducted as part of the Medical Research Council NuBrain Consortium. The investigators and Dr. Fillit reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ALZHEIMER’S RESEARCH AND THERAPY
Transition to Tenecteplase From t-PA for Acute Ischemic Stroke at Walter Reed National Military Medical Center
Tissue plasminogen activator (t-PA) has been the standard IV thrombolytic used in acute ischemic stroke treatment since its US Food and Drug Administration (FDA) approval in 1995. Trials have established this drug’s efficacy in the treatment of acute ischemic stroke and the appropriate patient population for therapy.1-3 Published guidelines and experiences have made clear that a written protocol with extensive personnel training is important to deliver this care properly.4
Tenecteplase has been available for use in the treatment of acute myocardial infarction (MI) and studied in acute ischemic strokes since 2000. Recent large multicenter trials have suggested tenecteplase may work better than t-PA in the recanalization of large vessel occlusions (LVOs) and have provided guidance on proper dosing in acute ischemic stroke victims.5-8 Compared with t-PA, tenecteplase has a longer half-life, is more fibrin specific (causing less coagulopathy), and is more resistant to endogenous plasminogen activator inhibitor.9,10 Using tenecteplase for acute ischemic stroke is simpler as a single dose bolus rather than a bolus followed by a 1-hour infusion with t-PA. Immediate mechanical thrombectomy for LVO is less complicated without the 1-hour t-PA infusion.5,6 Tenecteplase use also allows for nonthrombectomy hospitals to accelerate transfer times for patients who need thrombectomy following thrombolysis by eliminating the need for critical care nurse–staffed ambulances for interfacility transfer.11 Tenecteplase also is cheaper: Tenecteplase costs $3748 per vial, whereas t-PA costs $5800 per vial equating to roughly a $2000 savings per patient.12,13 Finally, the pharmacy formulary is simplified by using a single thrombolytic agent for both cardiac and neurologic emergencies.
Tenecteplase does have some drawbacks to consider. Currently, tenecteplase is not approved by the FDA for the indication of acute ischemic stroke, though the drug is endorsed by the American Heart Association stroke guidelines of 2019 as an alternative to t-PA.14 There is no stroke-specific preparation of the drug, leading to potential dosing errors. Therefore, a systematic process to safely transition from t-PA to tenecteplase for acute ischemic stroke was undertaken at Walter Reed National Military Medical Center (WRNMMC) in Bethesda, Maryland. Here, we report the process required in making a complex switch in thrombolytic medication along with the potential benefits of making this transition.
OBSERVATIONS
The process to implement tenecteplase required extensive training and education for staff physicians, nurses, pharmacists, radiologists, trainees, and the rapid response team. Our institution administered IV thrombolytic drugs up to 25 times annually to acute ischemic stroke victims, meaning we had to train personnel extensively and repeatedly.
In preparation for the transition to tenecteplase, hospital leadership gathered staff for multidisciplinary administrative meetings that included neurology, emergency medicine, intensive care, pharmacy, radiology, and nursing departments. The purpose of these meetings was to establish a standard operating procedure (SOP) to ensure a safe transition. This process began in May 2020 and involved regular meetings to draft and revise our SOP. Additionally, several leadership and training sessions were held over a 6-month period. Stroke boxes were developed that contained the required evaluation tools, consent forms, medications (tenecteplase and treatments for known complications), dosing cards, and instructions. Final approval of the updated acute ischemic stroke hospital policy was obtained in November 2020 and signed by the above departments.
All inclusion and exclusion criteria were determined to be the same for tenecteplase as they were for t-PA with the notable exception that the WAKE-UP trial protocol would not be supported until further evidence became available.9 The results of the WAKE-UP trial had previously been used at WRNMMC to justify administration of t-PA in patients who awoke with symptoms of acute ischemic stroke, the last known well was unclear or > 4.5 hours, and for whom a magnetic resonance imaging (MRI) of the brain could be obtained rapidly. Based on the WAKE-UP trial, if the MRI scan of the brain in these patients demonstrated restricted diffusion without fluid attenuated inversion recovery (FLAIR) signal changes (diffusion-weighted [DWI]-FLAIR mismatch sign), this indicated that the stroke had likely occurred recently, and it was safe to administer t-PA. This allowed for administration of t-PA outside the standard treatment window of 4.5 hours from last known well, especially in the cases of patients who awoke with symptoms.
Since safety data are not yet available for the use of tenecteplase in this fashion, the WAKE-UP trial protocol was not used as an inclusion criterion. The informed consent form was modified, and the following scenarios were outlined: (1) If the patient or surrogate is immediately available to consent, paper consent will be documented with the additional note that tenecteplase is being used off-label; and (2) If the patient cannot consent and a surrogate is not immediately available, the medicine will be used emergently as long as the neurology resident and attending physicians agree.15
Risk mitigation was considered carefully. The stroke box described above is stocked and maintained by the pharmacy as we have transitioned to using designated pharmacists for the storage and preparation of tenecteplase. We highly recommend the use of designated pharmacists or emergency department pharmacists in this manner to avoid dosing errors.7,16 Since the current pharmacy-provided tenecteplase bottle contains twice the maximum dose indicated for ischemic stroke, only a 5 mL syringe is included in the stroke box to ensure a maximum dose of 25 mg is drawn up after reconstitution. Dosing card charts were made like existing dosing card charts for t-PA to quickly calculate the 0.25 mg/kg dose. In training, the difference in dosing in ischemic stroke was emphasized. Finally, pharmacy has taken responsibility for dosing the medication during stroke codes.
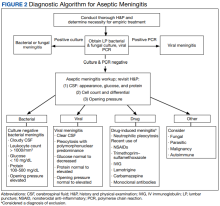
Any medical personnel at WRNMMC can initiate a stroke code by sending a page to the neurology consult service (Figure).
TRANSITION AND RESULTS
From November 2020 to December 2021, 10 patients have been treated in total at WRNMMC (Table).
CONCLUSIONS
The available evidence supports the transition from t-PA to tenecteplase for acute ischemic stroke. The successful transition required months of preparation involving multidisciplinary meetings between neurology, nursing, pharmacy, radiology, rapid response teams, critical care, and emergency medicine departments. Safeguards must be implemented to avoid a tenecteplase dosing error that can lead to potentially life-threatening adverse effects. The results at WRNMMC thus far are promising for safety and efficacy. Several process improvements are planned: a hospital-wide overhead page will accompany the direct page to neurology; other team members, including radiology and pharmacy, will be included on the acute stroke alert; and a stroke-specific paging application will be implemented to better track real-time stroke metrics and improve flow. These measures mirror processes that are occurring in institutions that treat acute stroke patients.
1. Lees KR, Bluhmki E, von Kummer R, et al. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet. 2010;375(9727):1695-1703. doi:10.1016/S0140-6736(10)60491-6
2. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581- 1587. doi:10.1056/NEJM199512143332401
3. Hacke W, Donnan G, Fieschi C, et al. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet. 2004;363(9411):768-774. doi:10.1016/S0140-6736(04)15692-4
4. Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947. doi:10.1161/STR.0b013e318284056a
5. Campbell B, Mitchell P, Churilov L, et al. Tenecteplase versus alteplase before thrombectomy for ischemic stroke. N Engl J Med. 2018;378(17):1573-1582. doi:10.1056/nejmoa1716405
6. Yang P, Zhang Y, Zhang L, et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med. 2020;382(21):1981-1993. doi:10.1056/NEJMoa2001123
7. Menon BK, Buck BH, Singh N, et al. Intravenous tenecteplase compared with alteplase for acute ischaemic stroke in Canada (AcT): a pragmatic, multicentre, open-label, registry-linked, randomised, controlled, noninferiority trial. Lancet. 2022;400(10347):161-169. doi:10.1016/S0140-6736(22)01054-6
8. Campbell BCV, Mitchell PJ, Churilov L, et al. Effect of intravenous tenecteplase dose on cerebral reperfusion before thrombectomy in patients with large vessel occlusion ischemic stroke: the EXTEND-IA TNK part 2 randomized clinical trial. JAMA. 2020;323(13):1257- 1265. doi:10.1001/jama.2020.1511
9. Warach SJ, Dula AN, Milling TJ Jr. Tenecteplase thrombolysis for acute ischemic stroke. Stroke. 2020;51(11):3440- 3451. doi:10.1161/STROKEAHA.120.029749
10. Huang X, Moreton FC, Kalladka D, et al. Coagulation and fibrinolytic activity of tenecteplase and alteplase in acute ischemic stroke. Stroke. 2015;46(12):3543-3546. doi:10.1161/STROKEAHA.115.011290
11. Burgos AM, Saver JL. Evidence that tenecteplase is noninferior to alteplase for acute ischemic stroke: meta-analysis of 5 randomized trials. Stroke. 2019;50(8):2156-2162. doi:10.1161/STROKEAHA.119.025080
12. Potla N, Ganti L. Tenecteplase vs. alteplase for acute ischemic stroke: a systematic review. Int J Emerg Med. 2022;15(1). doi:10.1186/s12245-021-00399-w
13. Warach SJ, Winegar A, Ottenbacher A, Miller C, Gibson D. Abstract WMP52: reduced hospital costs for ischemic stroke treated with tenecteplase. Stroke. 2022;53(suppl 1):AWMP52. doi:10.1161/str.53.suppl_1.WMP52
14. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418. doi:10.1161/str.0000000000000211
15. Faris H, Dewar B, Dowlatshahi D, et al. Ethical justification for deferral of consent in the AcT trial for acute ischemic stroke. Stroke. 2022;53(7):2420-2423. doi:10.1161/strokeaha.122.038760
16. Kvistad CE, Næss H, Helleberg BH, et al. Tenecteplase versus alteplase for the management of acute ischaemic stroke in Norway (NOR-TEST 2, part A): a phase 3, randomised, open-label, blinded endpoint, non-inferiority trial. Lancet Neurol. 2022;21(6):511-519. doi:10.1016/S1474-4422(22)00124-7
Tissue plasminogen activator (t-PA) has been the standard IV thrombolytic used in acute ischemic stroke treatment since its US Food and Drug Administration (FDA) approval in 1995. Trials have established this drug’s efficacy in the treatment of acute ischemic stroke and the appropriate patient population for therapy.1-3 Published guidelines and experiences have made clear that a written protocol with extensive personnel training is important to deliver this care properly.4
Tenecteplase has been available for use in the treatment of acute myocardial infarction (MI) and studied in acute ischemic strokes since 2000. Recent large multicenter trials have suggested tenecteplase may work better than t-PA in the recanalization of large vessel occlusions (LVOs) and have provided guidance on proper dosing in acute ischemic stroke victims.5-8 Compared with t-PA, tenecteplase has a longer half-life, is more fibrin specific (causing less coagulopathy), and is more resistant to endogenous plasminogen activator inhibitor.9,10 Using tenecteplase for acute ischemic stroke is simpler as a single dose bolus rather than a bolus followed by a 1-hour infusion with t-PA. Immediate mechanical thrombectomy for LVO is less complicated without the 1-hour t-PA infusion.5,6 Tenecteplase use also allows for nonthrombectomy hospitals to accelerate transfer times for patients who need thrombectomy following thrombolysis by eliminating the need for critical care nurse–staffed ambulances for interfacility transfer.11 Tenecteplase also is cheaper: Tenecteplase costs $3748 per vial, whereas t-PA costs $5800 per vial equating to roughly a $2000 savings per patient.12,13 Finally, the pharmacy formulary is simplified by using a single thrombolytic agent for both cardiac and neurologic emergencies.
Tenecteplase does have some drawbacks to consider. Currently, tenecteplase is not approved by the FDA for the indication of acute ischemic stroke, though the drug is endorsed by the American Heart Association stroke guidelines of 2019 as an alternative to t-PA.14 There is no stroke-specific preparation of the drug, leading to potential dosing errors. Therefore, a systematic process to safely transition from t-PA to tenecteplase for acute ischemic stroke was undertaken at Walter Reed National Military Medical Center (WRNMMC) in Bethesda, Maryland. Here, we report the process required in making a complex switch in thrombolytic medication along with the potential benefits of making this transition.
OBSERVATIONS
The process to implement tenecteplase required extensive training and education for staff physicians, nurses, pharmacists, radiologists, trainees, and the rapid response team. Our institution administered IV thrombolytic drugs up to 25 times annually to acute ischemic stroke victims, meaning we had to train personnel extensively and repeatedly.
In preparation for the transition to tenecteplase, hospital leadership gathered staff for multidisciplinary administrative meetings that included neurology, emergency medicine, intensive care, pharmacy, radiology, and nursing departments. The purpose of these meetings was to establish a standard operating procedure (SOP) to ensure a safe transition. This process began in May 2020 and involved regular meetings to draft and revise our SOP. Additionally, several leadership and training sessions were held over a 6-month period. Stroke boxes were developed that contained the required evaluation tools, consent forms, medications (tenecteplase and treatments for known complications), dosing cards, and instructions. Final approval of the updated acute ischemic stroke hospital policy was obtained in November 2020 and signed by the above departments.
All inclusion and exclusion criteria were determined to be the same for tenecteplase as they were for t-PA with the notable exception that the WAKE-UP trial protocol would not be supported until further evidence became available.9 The results of the WAKE-UP trial had previously been used at WRNMMC to justify administration of t-PA in patients who awoke with symptoms of acute ischemic stroke, the last known well was unclear or > 4.5 hours, and for whom a magnetic resonance imaging (MRI) of the brain could be obtained rapidly. Based on the WAKE-UP trial, if the MRI scan of the brain in these patients demonstrated restricted diffusion without fluid attenuated inversion recovery (FLAIR) signal changes (diffusion-weighted [DWI]-FLAIR mismatch sign), this indicated that the stroke had likely occurred recently, and it was safe to administer t-PA. This allowed for administration of t-PA outside the standard treatment window of 4.5 hours from last known well, especially in the cases of patients who awoke with symptoms.
Since safety data are not yet available for the use of tenecteplase in this fashion, the WAKE-UP trial protocol was not used as an inclusion criterion. The informed consent form was modified, and the following scenarios were outlined: (1) If the patient or surrogate is immediately available to consent, paper consent will be documented with the additional note that tenecteplase is being used off-label; and (2) If the patient cannot consent and a surrogate is not immediately available, the medicine will be used emergently as long as the neurology resident and attending physicians agree.15
Risk mitigation was considered carefully. The stroke box described above is stocked and maintained by the pharmacy as we have transitioned to using designated pharmacists for the storage and preparation of tenecteplase. We highly recommend the use of designated pharmacists or emergency department pharmacists in this manner to avoid dosing errors.7,16 Since the current pharmacy-provided tenecteplase bottle contains twice the maximum dose indicated for ischemic stroke, only a 5 mL syringe is included in the stroke box to ensure a maximum dose of 25 mg is drawn up after reconstitution. Dosing card charts were made like existing dosing card charts for t-PA to quickly calculate the 0.25 mg/kg dose. In training, the difference in dosing in ischemic stroke was emphasized. Finally, pharmacy has taken responsibility for dosing the medication during stroke codes.
Any medical personnel at WRNMMC can initiate a stroke code by sending a page to the neurology consult service (Figure).
TRANSITION AND RESULTS
From November 2020 to December 2021, 10 patients have been treated in total at WRNMMC (Table).
CONCLUSIONS
The available evidence supports the transition from t-PA to tenecteplase for acute ischemic stroke. The successful transition required months of preparation involving multidisciplinary meetings between neurology, nursing, pharmacy, radiology, rapid response teams, critical care, and emergency medicine departments. Safeguards must be implemented to avoid a tenecteplase dosing error that can lead to potentially life-threatening adverse effects. The results at WRNMMC thus far are promising for safety and efficacy. Several process improvements are planned: a hospital-wide overhead page will accompany the direct page to neurology; other team members, including radiology and pharmacy, will be included on the acute stroke alert; and a stroke-specific paging application will be implemented to better track real-time stroke metrics and improve flow. These measures mirror processes that are occurring in institutions that treat acute stroke patients.
Tissue plasminogen activator (t-PA) has been the standard IV thrombolytic used in acute ischemic stroke treatment since its US Food and Drug Administration (FDA) approval in 1995. Trials have established this drug’s efficacy in the treatment of acute ischemic stroke and the appropriate patient population for therapy.1-3 Published guidelines and experiences have made clear that a written protocol with extensive personnel training is important to deliver this care properly.4
Tenecteplase has been available for use in the treatment of acute myocardial infarction (MI) and studied in acute ischemic strokes since 2000. Recent large multicenter trials have suggested tenecteplase may work better than t-PA in the recanalization of large vessel occlusions (LVOs) and have provided guidance on proper dosing in acute ischemic stroke victims.5-8 Compared with t-PA, tenecteplase has a longer half-life, is more fibrin specific (causing less coagulopathy), and is more resistant to endogenous plasminogen activator inhibitor.9,10 Using tenecteplase for acute ischemic stroke is simpler as a single dose bolus rather than a bolus followed by a 1-hour infusion with t-PA. Immediate mechanical thrombectomy for LVO is less complicated without the 1-hour t-PA infusion.5,6 Tenecteplase use also allows for nonthrombectomy hospitals to accelerate transfer times for patients who need thrombectomy following thrombolysis by eliminating the need for critical care nurse–staffed ambulances for interfacility transfer.11 Tenecteplase also is cheaper: Tenecteplase costs $3748 per vial, whereas t-PA costs $5800 per vial equating to roughly a $2000 savings per patient.12,13 Finally, the pharmacy formulary is simplified by using a single thrombolytic agent for both cardiac and neurologic emergencies.
Tenecteplase does have some drawbacks to consider. Currently, tenecteplase is not approved by the FDA for the indication of acute ischemic stroke, though the drug is endorsed by the American Heart Association stroke guidelines of 2019 as an alternative to t-PA.14 There is no stroke-specific preparation of the drug, leading to potential dosing errors. Therefore, a systematic process to safely transition from t-PA to tenecteplase for acute ischemic stroke was undertaken at Walter Reed National Military Medical Center (WRNMMC) in Bethesda, Maryland. Here, we report the process required in making a complex switch in thrombolytic medication along with the potential benefits of making this transition.
OBSERVATIONS
The process to implement tenecteplase required extensive training and education for staff physicians, nurses, pharmacists, radiologists, trainees, and the rapid response team. Our institution administered IV thrombolytic drugs up to 25 times annually to acute ischemic stroke victims, meaning we had to train personnel extensively and repeatedly.
In preparation for the transition to tenecteplase, hospital leadership gathered staff for multidisciplinary administrative meetings that included neurology, emergency medicine, intensive care, pharmacy, radiology, and nursing departments. The purpose of these meetings was to establish a standard operating procedure (SOP) to ensure a safe transition. This process began in May 2020 and involved regular meetings to draft and revise our SOP. Additionally, several leadership and training sessions were held over a 6-month period. Stroke boxes were developed that contained the required evaluation tools, consent forms, medications (tenecteplase and treatments for known complications), dosing cards, and instructions. Final approval of the updated acute ischemic stroke hospital policy was obtained in November 2020 and signed by the above departments.
All inclusion and exclusion criteria were determined to be the same for tenecteplase as they were for t-PA with the notable exception that the WAKE-UP trial protocol would not be supported until further evidence became available.9 The results of the WAKE-UP trial had previously been used at WRNMMC to justify administration of t-PA in patients who awoke with symptoms of acute ischemic stroke, the last known well was unclear or > 4.5 hours, and for whom a magnetic resonance imaging (MRI) of the brain could be obtained rapidly. Based on the WAKE-UP trial, if the MRI scan of the brain in these patients demonstrated restricted diffusion without fluid attenuated inversion recovery (FLAIR) signal changes (diffusion-weighted [DWI]-FLAIR mismatch sign), this indicated that the stroke had likely occurred recently, and it was safe to administer t-PA. This allowed for administration of t-PA outside the standard treatment window of 4.5 hours from last known well, especially in the cases of patients who awoke with symptoms.
Since safety data are not yet available for the use of tenecteplase in this fashion, the WAKE-UP trial protocol was not used as an inclusion criterion. The informed consent form was modified, and the following scenarios were outlined: (1) If the patient or surrogate is immediately available to consent, paper consent will be documented with the additional note that tenecteplase is being used off-label; and (2) If the patient cannot consent and a surrogate is not immediately available, the medicine will be used emergently as long as the neurology resident and attending physicians agree.15
Risk mitigation was considered carefully. The stroke box described above is stocked and maintained by the pharmacy as we have transitioned to using designated pharmacists for the storage and preparation of tenecteplase. We highly recommend the use of designated pharmacists or emergency department pharmacists in this manner to avoid dosing errors.7,16 Since the current pharmacy-provided tenecteplase bottle contains twice the maximum dose indicated for ischemic stroke, only a 5 mL syringe is included in the stroke box to ensure a maximum dose of 25 mg is drawn up after reconstitution. Dosing card charts were made like existing dosing card charts for t-PA to quickly calculate the 0.25 mg/kg dose. In training, the difference in dosing in ischemic stroke was emphasized. Finally, pharmacy has taken responsibility for dosing the medication during stroke codes.
Any medical personnel at WRNMMC can initiate a stroke code by sending a page to the neurology consult service (Figure).
TRANSITION AND RESULTS
From November 2020 to December 2021, 10 patients have been treated in total at WRNMMC (Table).
CONCLUSIONS
The available evidence supports the transition from t-PA to tenecteplase for acute ischemic stroke. The successful transition required months of preparation involving multidisciplinary meetings between neurology, nursing, pharmacy, radiology, rapid response teams, critical care, and emergency medicine departments. Safeguards must be implemented to avoid a tenecteplase dosing error that can lead to potentially life-threatening adverse effects. The results at WRNMMC thus far are promising for safety and efficacy. Several process improvements are planned: a hospital-wide overhead page will accompany the direct page to neurology; other team members, including radiology and pharmacy, will be included on the acute stroke alert; and a stroke-specific paging application will be implemented to better track real-time stroke metrics and improve flow. These measures mirror processes that are occurring in institutions that treat acute stroke patients.
1. Lees KR, Bluhmki E, von Kummer R, et al. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet. 2010;375(9727):1695-1703. doi:10.1016/S0140-6736(10)60491-6
2. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581- 1587. doi:10.1056/NEJM199512143332401
3. Hacke W, Donnan G, Fieschi C, et al. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet. 2004;363(9411):768-774. doi:10.1016/S0140-6736(04)15692-4
4. Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947. doi:10.1161/STR.0b013e318284056a
5. Campbell B, Mitchell P, Churilov L, et al. Tenecteplase versus alteplase before thrombectomy for ischemic stroke. N Engl J Med. 2018;378(17):1573-1582. doi:10.1056/nejmoa1716405
6. Yang P, Zhang Y, Zhang L, et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med. 2020;382(21):1981-1993. doi:10.1056/NEJMoa2001123
7. Menon BK, Buck BH, Singh N, et al. Intravenous tenecteplase compared with alteplase for acute ischaemic stroke in Canada (AcT): a pragmatic, multicentre, open-label, registry-linked, randomised, controlled, noninferiority trial. Lancet. 2022;400(10347):161-169. doi:10.1016/S0140-6736(22)01054-6
8. Campbell BCV, Mitchell PJ, Churilov L, et al. Effect of intravenous tenecteplase dose on cerebral reperfusion before thrombectomy in patients with large vessel occlusion ischemic stroke: the EXTEND-IA TNK part 2 randomized clinical trial. JAMA. 2020;323(13):1257- 1265. doi:10.1001/jama.2020.1511
9. Warach SJ, Dula AN, Milling TJ Jr. Tenecteplase thrombolysis for acute ischemic stroke. Stroke. 2020;51(11):3440- 3451. doi:10.1161/STROKEAHA.120.029749
10. Huang X, Moreton FC, Kalladka D, et al. Coagulation and fibrinolytic activity of tenecteplase and alteplase in acute ischemic stroke. Stroke. 2015;46(12):3543-3546. doi:10.1161/STROKEAHA.115.011290
11. Burgos AM, Saver JL. Evidence that tenecteplase is noninferior to alteplase for acute ischemic stroke: meta-analysis of 5 randomized trials. Stroke. 2019;50(8):2156-2162. doi:10.1161/STROKEAHA.119.025080
12. Potla N, Ganti L. Tenecteplase vs. alteplase for acute ischemic stroke: a systematic review. Int J Emerg Med. 2022;15(1). doi:10.1186/s12245-021-00399-w
13. Warach SJ, Winegar A, Ottenbacher A, Miller C, Gibson D. Abstract WMP52: reduced hospital costs for ischemic stroke treated with tenecteplase. Stroke. 2022;53(suppl 1):AWMP52. doi:10.1161/str.53.suppl_1.WMP52
14. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418. doi:10.1161/str.0000000000000211
15. Faris H, Dewar B, Dowlatshahi D, et al. Ethical justification for deferral of consent in the AcT trial for acute ischemic stroke. Stroke. 2022;53(7):2420-2423. doi:10.1161/strokeaha.122.038760
16. Kvistad CE, Næss H, Helleberg BH, et al. Tenecteplase versus alteplase for the management of acute ischaemic stroke in Norway (NOR-TEST 2, part A): a phase 3, randomised, open-label, blinded endpoint, non-inferiority trial. Lancet Neurol. 2022;21(6):511-519. doi:10.1016/S1474-4422(22)00124-7
1. Lees KR, Bluhmki E, von Kummer R, et al. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet. 2010;375(9727):1695-1703. doi:10.1016/S0140-6736(10)60491-6
2. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581- 1587. doi:10.1056/NEJM199512143332401
3. Hacke W, Donnan G, Fieschi C, et al. Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet. 2004;363(9411):768-774. doi:10.1016/S0140-6736(04)15692-4
4. Jauch EC, Saver JL, Adams HP Jr, et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2013;44(3):870-947. doi:10.1161/STR.0b013e318284056a
5. Campbell B, Mitchell P, Churilov L, et al. Tenecteplase versus alteplase before thrombectomy for ischemic stroke. N Engl J Med. 2018;378(17):1573-1582. doi:10.1056/nejmoa1716405
6. Yang P, Zhang Y, Zhang L, et al. Endovascular thrombectomy with or without intravenous alteplase in acute stroke. N Engl J Med. 2020;382(21):1981-1993. doi:10.1056/NEJMoa2001123
7. Menon BK, Buck BH, Singh N, et al. Intravenous tenecteplase compared with alteplase for acute ischaemic stroke in Canada (AcT): a pragmatic, multicentre, open-label, registry-linked, randomised, controlled, noninferiority trial. Lancet. 2022;400(10347):161-169. doi:10.1016/S0140-6736(22)01054-6
8. Campbell BCV, Mitchell PJ, Churilov L, et al. Effect of intravenous tenecteplase dose on cerebral reperfusion before thrombectomy in patients with large vessel occlusion ischemic stroke: the EXTEND-IA TNK part 2 randomized clinical trial. JAMA. 2020;323(13):1257- 1265. doi:10.1001/jama.2020.1511
9. Warach SJ, Dula AN, Milling TJ Jr. Tenecteplase thrombolysis for acute ischemic stroke. Stroke. 2020;51(11):3440- 3451. doi:10.1161/STROKEAHA.120.029749
10. Huang X, Moreton FC, Kalladka D, et al. Coagulation and fibrinolytic activity of tenecteplase and alteplase in acute ischemic stroke. Stroke. 2015;46(12):3543-3546. doi:10.1161/STROKEAHA.115.011290
11. Burgos AM, Saver JL. Evidence that tenecteplase is noninferior to alteplase for acute ischemic stroke: meta-analysis of 5 randomized trials. Stroke. 2019;50(8):2156-2162. doi:10.1161/STROKEAHA.119.025080
12. Potla N, Ganti L. Tenecteplase vs. alteplase for acute ischemic stroke: a systematic review. Int J Emerg Med. 2022;15(1). doi:10.1186/s12245-021-00399-w
13. Warach SJ, Winegar A, Ottenbacher A, Miller C, Gibson D. Abstract WMP52: reduced hospital costs for ischemic stroke treated with tenecteplase. Stroke. 2022;53(suppl 1):AWMP52. doi:10.1161/str.53.suppl_1.WMP52
14. Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines for the Early Management of Acute Ischemic Stroke: A Guideline for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke. 2019;50(12):e344-e418. doi:10.1161/str.0000000000000211
15. Faris H, Dewar B, Dowlatshahi D, et al. Ethical justification for deferral of consent in the AcT trial for acute ischemic stroke. Stroke. 2022;53(7):2420-2423. doi:10.1161/strokeaha.122.038760
16. Kvistad CE, Næss H, Helleberg BH, et al. Tenecteplase versus alteplase for the management of acute ischaemic stroke in Norway (NOR-TEST 2, part A): a phase 3, randomised, open-label, blinded endpoint, non-inferiority trial. Lancet Neurol. 2022;21(6):511-519. doi:10.1016/S1474-4422(22)00124-7
PPI use in type 2 diabetes links with cardiovascular events
Among people with type 2 diabetes who self-reported regularly using a proton pump inhibitor (PPI), the incidence of cardiovascular disease (CVD) events as well as all-cause death was significantly increased in a study of more than 19,000 people with type 2 diabetes in a prospective U.K. database.
During median follow-up of about 11 years, regular use of a PPI by people with type 2 diabetes was significantly linked with a 27% relative increase in the incidence of coronary artery disease, compared with nonuse of a PPI, after full adjustment for potential confounding variables.
The results also show PPI use was significantly linked after full adjustment with a 34% relative increase in MI, a 35% relative increase in heart failure, and a 30% relative increase in all-cause death, say a team of Chinese researchers in a recent report in the Journal of Clinical Endocrinology and Metabolism.
PPIs are a medication class widely used in both over-the-counter and prescription formulations to reduce acid production in the stomach and to treat gastroesophageal reflux disease and other acid-related disorders. The PPI class includes such widely used agents as esomeprazole (Nexium), lansoprazole (Prevacid), and omeprazole (Prilosec).
The analyses in this report, which used data collected in the UK Biobank, are “rigorous,” and the findings of “a modest elevation of CVD risk are consistent with a growing number of observational studies in populations with and without diabetes,” commented Mary R. Rooney, PhD, an epidemiologist at Johns Hopkins University, Baltimore, who focuses on diabetes and cardiovascular diseases.
Prior observational reports
For example, a report from a prospective, observational study of more than 4300 U.S. residents published in 2021 that Dr. Rooney coauthored documented that cumulative PPI exposure for more than 5 years was significantly linked with a twofold increase in the rate of CVD events, compared with people who did not use a PPI. (This analysis did not examine a possible effect of diabetes status.)
And in a separate prospective, observational study of more than 1,000 Australians with type 2 diabetes, initiation of PPI treatment was significantly linked with a 3.6-fold increased incidence of CVD events, compared with PPI nonuse.
However, Dr. Rooney cautioned that the role of PPI use in raising CVD events “is still an unresolved question. It is too soon to tell if PPI use in people with diabetes should trigger additional caution.” Findings are needed from prospective, randomized trials to determine more definitively whether PPIs play a causal role in the incidence of CVD events, she said in an interview.
U.S. practice often results in unwarranted prolongation of PPI treatment, said the authors of an editorial that accompanied the 2021 report by Dr. Rooney and coauthors.
Long-term PPI use threatens harm
“The practice of initiating stress ulcer prophylaxis [by administering a PPI] in critical care is common,” wrote the authors of the 2021 editorial, Nitin Malik, MD, and William S. Weintraub, MD. “Although it is data driven and well intentioned, the possibility of causing harm – if it is continued on a long-term basis after resolution of the acute illness – is palpable.”
The new analyses using UK Biobank data included 19,229 adults with type 2 diabetes and no preexisting coronary artery disease, MI, heart failure, or stroke. The cohort included 15,954 people (83%) who did not report using a PPI and 3,275 who currently used PPIs regularly. Study limitations include self-report as the only verification of PPI use and lack of information on type of PPI, dose size, or use duration.
The findings remained consistent in several sensitivity analyses, including a propensity score–matched analysis and after further adjustment for use of histamine2 receptor antagonists, a drug class with indications similar to those for PPIs.
The authors of the report speculated that mechanisms that might link PPI use and increased CVD and mortality risk could include changes to the gut microbiota and possible interactions between PPIs and antiplatelet agents.
The study received no commercial funding. The authors and Dr. Rooney disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Among people with type 2 diabetes who self-reported regularly using a proton pump inhibitor (PPI), the incidence of cardiovascular disease (CVD) events as well as all-cause death was significantly increased in a study of more than 19,000 people with type 2 diabetes in a prospective U.K. database.
During median follow-up of about 11 years, regular use of a PPI by people with type 2 diabetes was significantly linked with a 27% relative increase in the incidence of coronary artery disease, compared with nonuse of a PPI, after full adjustment for potential confounding variables.
The results also show PPI use was significantly linked after full adjustment with a 34% relative increase in MI, a 35% relative increase in heart failure, and a 30% relative increase in all-cause death, say a team of Chinese researchers in a recent report in the Journal of Clinical Endocrinology and Metabolism.
PPIs are a medication class widely used in both over-the-counter and prescription formulations to reduce acid production in the stomach and to treat gastroesophageal reflux disease and other acid-related disorders. The PPI class includes such widely used agents as esomeprazole (Nexium), lansoprazole (Prevacid), and omeprazole (Prilosec).
The analyses in this report, which used data collected in the UK Biobank, are “rigorous,” and the findings of “a modest elevation of CVD risk are consistent with a growing number of observational studies in populations with and without diabetes,” commented Mary R. Rooney, PhD, an epidemiologist at Johns Hopkins University, Baltimore, who focuses on diabetes and cardiovascular diseases.
Prior observational reports
For example, a report from a prospective, observational study of more than 4300 U.S. residents published in 2021 that Dr. Rooney coauthored documented that cumulative PPI exposure for more than 5 years was significantly linked with a twofold increase in the rate of CVD events, compared with people who did not use a PPI. (This analysis did not examine a possible effect of diabetes status.)
And in a separate prospective, observational study of more than 1,000 Australians with type 2 diabetes, initiation of PPI treatment was significantly linked with a 3.6-fold increased incidence of CVD events, compared with PPI nonuse.
However, Dr. Rooney cautioned that the role of PPI use in raising CVD events “is still an unresolved question. It is too soon to tell if PPI use in people with diabetes should trigger additional caution.” Findings are needed from prospective, randomized trials to determine more definitively whether PPIs play a causal role in the incidence of CVD events, she said in an interview.
U.S. practice often results in unwarranted prolongation of PPI treatment, said the authors of an editorial that accompanied the 2021 report by Dr. Rooney and coauthors.
Long-term PPI use threatens harm
“The practice of initiating stress ulcer prophylaxis [by administering a PPI] in critical care is common,” wrote the authors of the 2021 editorial, Nitin Malik, MD, and William S. Weintraub, MD. “Although it is data driven and well intentioned, the possibility of causing harm – if it is continued on a long-term basis after resolution of the acute illness – is palpable.”
The new analyses using UK Biobank data included 19,229 adults with type 2 diabetes and no preexisting coronary artery disease, MI, heart failure, or stroke. The cohort included 15,954 people (83%) who did not report using a PPI and 3,275 who currently used PPIs regularly. Study limitations include self-report as the only verification of PPI use and lack of information on type of PPI, dose size, or use duration.
The findings remained consistent in several sensitivity analyses, including a propensity score–matched analysis and after further adjustment for use of histamine2 receptor antagonists, a drug class with indications similar to those for PPIs.
The authors of the report speculated that mechanisms that might link PPI use and increased CVD and mortality risk could include changes to the gut microbiota and possible interactions between PPIs and antiplatelet agents.
The study received no commercial funding. The authors and Dr. Rooney disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Among people with type 2 diabetes who self-reported regularly using a proton pump inhibitor (PPI), the incidence of cardiovascular disease (CVD) events as well as all-cause death was significantly increased in a study of more than 19,000 people with type 2 diabetes in a prospective U.K. database.
During median follow-up of about 11 years, regular use of a PPI by people with type 2 diabetes was significantly linked with a 27% relative increase in the incidence of coronary artery disease, compared with nonuse of a PPI, after full adjustment for potential confounding variables.
The results also show PPI use was significantly linked after full adjustment with a 34% relative increase in MI, a 35% relative increase in heart failure, and a 30% relative increase in all-cause death, say a team of Chinese researchers in a recent report in the Journal of Clinical Endocrinology and Metabolism.
PPIs are a medication class widely used in both over-the-counter and prescription formulations to reduce acid production in the stomach and to treat gastroesophageal reflux disease and other acid-related disorders. The PPI class includes such widely used agents as esomeprazole (Nexium), lansoprazole (Prevacid), and omeprazole (Prilosec).
The analyses in this report, which used data collected in the UK Biobank, are “rigorous,” and the findings of “a modest elevation of CVD risk are consistent with a growing number of observational studies in populations with and without diabetes,” commented Mary R. Rooney, PhD, an epidemiologist at Johns Hopkins University, Baltimore, who focuses on diabetes and cardiovascular diseases.
Prior observational reports
For example, a report from a prospective, observational study of more than 4300 U.S. residents published in 2021 that Dr. Rooney coauthored documented that cumulative PPI exposure for more than 5 years was significantly linked with a twofold increase in the rate of CVD events, compared with people who did not use a PPI. (This analysis did not examine a possible effect of diabetes status.)
And in a separate prospective, observational study of more than 1,000 Australians with type 2 diabetes, initiation of PPI treatment was significantly linked with a 3.6-fold increased incidence of CVD events, compared with PPI nonuse.
However, Dr. Rooney cautioned that the role of PPI use in raising CVD events “is still an unresolved question. It is too soon to tell if PPI use in people with diabetes should trigger additional caution.” Findings are needed from prospective, randomized trials to determine more definitively whether PPIs play a causal role in the incidence of CVD events, she said in an interview.
U.S. practice often results in unwarranted prolongation of PPI treatment, said the authors of an editorial that accompanied the 2021 report by Dr. Rooney and coauthors.
Long-term PPI use threatens harm
“The practice of initiating stress ulcer prophylaxis [by administering a PPI] in critical care is common,” wrote the authors of the 2021 editorial, Nitin Malik, MD, and William S. Weintraub, MD. “Although it is data driven and well intentioned, the possibility of causing harm – if it is continued on a long-term basis after resolution of the acute illness – is palpable.”
The new analyses using UK Biobank data included 19,229 adults with type 2 diabetes and no preexisting coronary artery disease, MI, heart failure, or stroke. The cohort included 15,954 people (83%) who did not report using a PPI and 3,275 who currently used PPIs regularly. Study limitations include self-report as the only verification of PPI use and lack of information on type of PPI, dose size, or use duration.
The findings remained consistent in several sensitivity analyses, including a propensity score–matched analysis and after further adjustment for use of histamine2 receptor antagonists, a drug class with indications similar to those for PPIs.
The authors of the report speculated that mechanisms that might link PPI use and increased CVD and mortality risk could include changes to the gut microbiota and possible interactions between PPIs and antiplatelet agents.
The study received no commercial funding. The authors and Dr. Rooney disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF CLINICAL ENDOCRINOLOGY AND METABOLISM
Some BP meds tied to significantly lower risk for dementia, Alzheimer’s
Antihypertensive medications that stimulate rather than inhibit type 2 and 4 angiotensin II receptors can lower the rate of dementia among new users of these medications, new research suggests.
Results from a cohort study of more than 57,000 older Medicare beneficiaries showed that the initiation of antihypertensives that stimulate the receptors was linked to a 16% lower risk for incident Alzheimer’s disease and related dementia (ADRD) and an 18% lower risk for vascular dementia compared with those that inhibit the receptors.
“Achieving appropriate blood pressure control is essential for maximizing brain health, and this promising research suggests certain antihypertensives could yield brain benefit compared to others,” lead study author Zachary A. Marcum, PharmD, PhD, associate professor, University of Washington School of Pharmacy, Seattle, told this news organization.
The findings were published online in JAMA Network Open.
Medicare beneficiaries
Previous observational studies showed that antihypertensive medications that stimulate type 2 and 4 angiotensin II receptors, in comparison with those that don’t, were associated with lower rates of dementia. However, those studies included individuals with prevalent hypertension and were relatively small.
The new retrospective cohort study included a random sample of 57,773 Medicare beneficiaries aged at least 65 years with new-onset hypertension. The mean age of participants was 73.8 years, 62.9% were women, and 86.9% were White.
Over the course of the study, some participants filled at least one prescription for a stimulating angiotensin II receptor type 2 and 4, such as angiotensin II receptor type 1 blockers, dihydropyridine calcium channel blockers, and thiazide diuretics.
Others participants filled a prescription for an inhibiting type 2 and 4 angiotensin II receptors, including angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and nondihydropyridine calcium channel blockers.
“All these medications lower blood pressure, but they do it in different ways,” said Dr. Marcum.
The researchers were interested in the varying activity of these drugs at the type 2 and 4 angiotensin II receptors.
For each 30-day interval, they categorized beneficiaries into four groups: a stimulating medication group (n = 4,879) consisting of individuals mostly taking stimulating antihypertensives; an inhibiting medication group (n = 10,303) that mostly included individuals prescribed this type of antihypertensive; a mixed group (n = 2,179) that included a combination of the first two classifications; and a nonuser group (n = 40,413) of individuals who were not using either type of drug.
The primary outcome was time to first occurrence of ADRD. The secondary outcome was time to first occurrence of vascular dementia.
Researchers controlled for cardiovascular risk factors and sociodemographic characteristics, such as age, sex, race/ethnicity, and receipt of low-income subsidy.
Unanswered questions
After adjustments, results showed that initiation of an antihypertensive medication regimen that exclusively stimulates, rather than inhibits, type 2 and 4 angiotensin II receptors was associated with a 16% lower risk for incident ADRD over a follow-up of just under 7 years (hazard ratio, 0.84; 95% confidence interval, 0.79-0.90; P < .001).
The mixed regimen was also associated with statistically significant (P = .001) reduced odds of ADRD compared with the inhibiting medications.
As for vascular dementia, use of stimulating vs. inhibiting medications was associated with an 18% lower risk (HR, 0.82; 95% CI, 0.69-0.96; P = .02).
Again, use of the mixed regimen was associated with reduced risk of vascular dementia compared with the inhibiting medications (P = .03).
A variety of potential mechanisms might explain the superiority of stimulating agents when it comes to dementia risk, said Dr. Marcum. These could include, for example, increased blood flow to the brain and reduced amyloid.
“But more mechanistic work is needed as well as evaluation of dose responses, because that’s not something we looked at in this study,” Dr. Marcum said. “There are still a lot of unanswered questions.”
Stimulators instead of inhibitors?
The results of the current analysis come on the heels of some previous work showing the benefits of lowering blood pressure. For example, the Systolic Blood Pressure Intervention Trial (SPRINT) showed that targeting a systolic blood pressure below 120 mm Hg significantly reduces risk for heart disease, stroke, and death from these diseases.
But in contrast to previous research, the current study included only beneficiaries with incident hypertension and new use of antihypertensive medications, and it adjusted for time-varying confounding.
Prescribing stimulating instead of inhibiting treatments could make a difference at the population level, Dr. Marcum noted.
“If we could shift the prescribing a little bit from inhibiting to stimulating, that could possibly reduce dementia risk,” he said.
However, “we’re not suggesting [that all patients] have their regimen switched,” he added.
That’s because inhibiting medications still have an important place in the antihypertensive treatment armamentarium, Dr. Marcum noted. As an example, beta-blockers are used post heart attack.
As well, factors such as cost and side effects should be taken into consideration when prescribing an antihypertensive drug.
The new results could be used to set up a comparison in a future randomized controlled trial that would provide the strongest evidence for estimating causal effects of treatments, said Dr. Marcum.
‘More convincing’
Carlos G. Santos-Gallego, MD, Icahn School of Medicine at Mount Sinai, New York, said the study is “more convincing” than previous related research, as it has a larger sample size and a longer follow-up.
“And the exquisite statistical analysis gives more robustness, more solidity, to the hypothesis that drugs that stimulate type 2 and 4 angiotensin II receptors might be protective for dementia,” said Dr. Santos-Gallego, who was not involved with the research.
However, he noted that the retrospective study had some limitations, including the underdiagnosis of dementia. “The diagnosis of dementia is, honestly, very poorly done in the clinical setting,” he said.
As well, the study could be subject to “confounding by indication,” Dr. Santos-Gallego said. “There could be a third variable, another confounding factor, that’s responsible both for the dementia and for the prescription of these drugs,” he added.
For example, he noted that comorbidities such as atrial fibrillation, myocardial infarction, and heart failure might increase the risk of dementia.
He agreed with the investigators that a randomized clinical trial would address these limitations. “All comorbidities would be equally shared” in the randomized groups, and all participants would be given “a specific test for dementia at the same time,” Dr. Santos-Gallego said.
Still, he noted that the new results are in keeping with hypertension guidelines that recommend stimulating drugs.
“This trial definitely shows that the current hypertension guidelines are good treatment for our patients, not only to control blood pressure and not only to prevent infarction to prevent stroke but also to prevent dementia,” said Dr. Santos-Gallego.
Also commenting for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, said the new data provide “clarity” on why previous research had differing results on the effect of antihypertensives on cognition.
Among the caveats of this new analysis is that “it’s unclear if the demographics in this study are fully representative of Medicare beneficiaries,” said Dr. Snyder.
She, too, said a clinical trial is important “to understand if there is a preventative and/or treatment potential in the medications that stimulate type 2 and 4 angiotensin II receptors.”
The study received funding from the National Institute on Aging. Dr. Marcum and Dr. Santos-Gallego have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antihypertensive medications that stimulate rather than inhibit type 2 and 4 angiotensin II receptors can lower the rate of dementia among new users of these medications, new research suggests.
Results from a cohort study of more than 57,000 older Medicare beneficiaries showed that the initiation of antihypertensives that stimulate the receptors was linked to a 16% lower risk for incident Alzheimer’s disease and related dementia (ADRD) and an 18% lower risk for vascular dementia compared with those that inhibit the receptors.
“Achieving appropriate blood pressure control is essential for maximizing brain health, and this promising research suggests certain antihypertensives could yield brain benefit compared to others,” lead study author Zachary A. Marcum, PharmD, PhD, associate professor, University of Washington School of Pharmacy, Seattle, told this news organization.
The findings were published online in JAMA Network Open.
Medicare beneficiaries
Previous observational studies showed that antihypertensive medications that stimulate type 2 and 4 angiotensin II receptors, in comparison with those that don’t, were associated with lower rates of dementia. However, those studies included individuals with prevalent hypertension and were relatively small.
The new retrospective cohort study included a random sample of 57,773 Medicare beneficiaries aged at least 65 years with new-onset hypertension. The mean age of participants was 73.8 years, 62.9% were women, and 86.9% were White.
Over the course of the study, some participants filled at least one prescription for a stimulating angiotensin II receptor type 2 and 4, such as angiotensin II receptor type 1 blockers, dihydropyridine calcium channel blockers, and thiazide diuretics.
Others participants filled a prescription for an inhibiting type 2 and 4 angiotensin II receptors, including angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and nondihydropyridine calcium channel blockers.
“All these medications lower blood pressure, but they do it in different ways,” said Dr. Marcum.
The researchers were interested in the varying activity of these drugs at the type 2 and 4 angiotensin II receptors.
For each 30-day interval, they categorized beneficiaries into four groups: a stimulating medication group (n = 4,879) consisting of individuals mostly taking stimulating antihypertensives; an inhibiting medication group (n = 10,303) that mostly included individuals prescribed this type of antihypertensive; a mixed group (n = 2,179) that included a combination of the first two classifications; and a nonuser group (n = 40,413) of individuals who were not using either type of drug.
The primary outcome was time to first occurrence of ADRD. The secondary outcome was time to first occurrence of vascular dementia.
Researchers controlled for cardiovascular risk factors and sociodemographic characteristics, such as age, sex, race/ethnicity, and receipt of low-income subsidy.
Unanswered questions
After adjustments, results showed that initiation of an antihypertensive medication regimen that exclusively stimulates, rather than inhibits, type 2 and 4 angiotensin II receptors was associated with a 16% lower risk for incident ADRD over a follow-up of just under 7 years (hazard ratio, 0.84; 95% confidence interval, 0.79-0.90; P < .001).
The mixed regimen was also associated with statistically significant (P = .001) reduced odds of ADRD compared with the inhibiting medications.
As for vascular dementia, use of stimulating vs. inhibiting medications was associated with an 18% lower risk (HR, 0.82; 95% CI, 0.69-0.96; P = .02).
Again, use of the mixed regimen was associated with reduced risk of vascular dementia compared with the inhibiting medications (P = .03).
A variety of potential mechanisms might explain the superiority of stimulating agents when it comes to dementia risk, said Dr. Marcum. These could include, for example, increased blood flow to the brain and reduced amyloid.
“But more mechanistic work is needed as well as evaluation of dose responses, because that’s not something we looked at in this study,” Dr. Marcum said. “There are still a lot of unanswered questions.”
Stimulators instead of inhibitors?
The results of the current analysis come on the heels of some previous work showing the benefits of lowering blood pressure. For example, the Systolic Blood Pressure Intervention Trial (SPRINT) showed that targeting a systolic blood pressure below 120 mm Hg significantly reduces risk for heart disease, stroke, and death from these diseases.
But in contrast to previous research, the current study included only beneficiaries with incident hypertension and new use of antihypertensive medications, and it adjusted for time-varying confounding.
Prescribing stimulating instead of inhibiting treatments could make a difference at the population level, Dr. Marcum noted.
“If we could shift the prescribing a little bit from inhibiting to stimulating, that could possibly reduce dementia risk,” he said.
However, “we’re not suggesting [that all patients] have their regimen switched,” he added.
That’s because inhibiting medications still have an important place in the antihypertensive treatment armamentarium, Dr. Marcum noted. As an example, beta-blockers are used post heart attack.
As well, factors such as cost and side effects should be taken into consideration when prescribing an antihypertensive drug.
The new results could be used to set up a comparison in a future randomized controlled trial that would provide the strongest evidence for estimating causal effects of treatments, said Dr. Marcum.
‘More convincing’
Carlos G. Santos-Gallego, MD, Icahn School of Medicine at Mount Sinai, New York, said the study is “more convincing” than previous related research, as it has a larger sample size and a longer follow-up.
“And the exquisite statistical analysis gives more robustness, more solidity, to the hypothesis that drugs that stimulate type 2 and 4 angiotensin II receptors might be protective for dementia,” said Dr. Santos-Gallego, who was not involved with the research.
However, he noted that the retrospective study had some limitations, including the underdiagnosis of dementia. “The diagnosis of dementia is, honestly, very poorly done in the clinical setting,” he said.
As well, the study could be subject to “confounding by indication,” Dr. Santos-Gallego said. “There could be a third variable, another confounding factor, that’s responsible both for the dementia and for the prescription of these drugs,” he added.
For example, he noted that comorbidities such as atrial fibrillation, myocardial infarction, and heart failure might increase the risk of dementia.
He agreed with the investigators that a randomized clinical trial would address these limitations. “All comorbidities would be equally shared” in the randomized groups, and all participants would be given “a specific test for dementia at the same time,” Dr. Santos-Gallego said.
Still, he noted that the new results are in keeping with hypertension guidelines that recommend stimulating drugs.
“This trial definitely shows that the current hypertension guidelines are good treatment for our patients, not only to control blood pressure and not only to prevent infarction to prevent stroke but also to prevent dementia,” said Dr. Santos-Gallego.
Also commenting for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, said the new data provide “clarity” on why previous research had differing results on the effect of antihypertensives on cognition.
Among the caveats of this new analysis is that “it’s unclear if the demographics in this study are fully representative of Medicare beneficiaries,” said Dr. Snyder.
She, too, said a clinical trial is important “to understand if there is a preventative and/or treatment potential in the medications that stimulate type 2 and 4 angiotensin II receptors.”
The study received funding from the National Institute on Aging. Dr. Marcum and Dr. Santos-Gallego have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Antihypertensive medications that stimulate rather than inhibit type 2 and 4 angiotensin II receptors can lower the rate of dementia among new users of these medications, new research suggests.
Results from a cohort study of more than 57,000 older Medicare beneficiaries showed that the initiation of antihypertensives that stimulate the receptors was linked to a 16% lower risk for incident Alzheimer’s disease and related dementia (ADRD) and an 18% lower risk for vascular dementia compared with those that inhibit the receptors.
“Achieving appropriate blood pressure control is essential for maximizing brain health, and this promising research suggests certain antihypertensives could yield brain benefit compared to others,” lead study author Zachary A. Marcum, PharmD, PhD, associate professor, University of Washington School of Pharmacy, Seattle, told this news organization.
The findings were published online in JAMA Network Open.
Medicare beneficiaries
Previous observational studies showed that antihypertensive medications that stimulate type 2 and 4 angiotensin II receptors, in comparison with those that don’t, were associated with lower rates of dementia. However, those studies included individuals with prevalent hypertension and were relatively small.
The new retrospective cohort study included a random sample of 57,773 Medicare beneficiaries aged at least 65 years with new-onset hypertension. The mean age of participants was 73.8 years, 62.9% were women, and 86.9% were White.
Over the course of the study, some participants filled at least one prescription for a stimulating angiotensin II receptor type 2 and 4, such as angiotensin II receptor type 1 blockers, dihydropyridine calcium channel blockers, and thiazide diuretics.
Others participants filled a prescription for an inhibiting type 2 and 4 angiotensin II receptors, including angiotensin-converting enzyme (ACE) inhibitors, beta-blockers, and nondihydropyridine calcium channel blockers.
“All these medications lower blood pressure, but they do it in different ways,” said Dr. Marcum.
The researchers were interested in the varying activity of these drugs at the type 2 and 4 angiotensin II receptors.
For each 30-day interval, they categorized beneficiaries into four groups: a stimulating medication group (n = 4,879) consisting of individuals mostly taking stimulating antihypertensives; an inhibiting medication group (n = 10,303) that mostly included individuals prescribed this type of antihypertensive; a mixed group (n = 2,179) that included a combination of the first two classifications; and a nonuser group (n = 40,413) of individuals who were not using either type of drug.
The primary outcome was time to first occurrence of ADRD. The secondary outcome was time to first occurrence of vascular dementia.
Researchers controlled for cardiovascular risk factors and sociodemographic characteristics, such as age, sex, race/ethnicity, and receipt of low-income subsidy.
Unanswered questions
After adjustments, results showed that initiation of an antihypertensive medication regimen that exclusively stimulates, rather than inhibits, type 2 and 4 angiotensin II receptors was associated with a 16% lower risk for incident ADRD over a follow-up of just under 7 years (hazard ratio, 0.84; 95% confidence interval, 0.79-0.90; P < .001).
The mixed regimen was also associated with statistically significant (P = .001) reduced odds of ADRD compared with the inhibiting medications.
As for vascular dementia, use of stimulating vs. inhibiting medications was associated with an 18% lower risk (HR, 0.82; 95% CI, 0.69-0.96; P = .02).
Again, use of the mixed regimen was associated with reduced risk of vascular dementia compared with the inhibiting medications (P = .03).
A variety of potential mechanisms might explain the superiority of stimulating agents when it comes to dementia risk, said Dr. Marcum. These could include, for example, increased blood flow to the brain and reduced amyloid.
“But more mechanistic work is needed as well as evaluation of dose responses, because that’s not something we looked at in this study,” Dr. Marcum said. “There are still a lot of unanswered questions.”
Stimulators instead of inhibitors?
The results of the current analysis come on the heels of some previous work showing the benefits of lowering blood pressure. For example, the Systolic Blood Pressure Intervention Trial (SPRINT) showed that targeting a systolic blood pressure below 120 mm Hg significantly reduces risk for heart disease, stroke, and death from these diseases.
But in contrast to previous research, the current study included only beneficiaries with incident hypertension and new use of antihypertensive medications, and it adjusted for time-varying confounding.
Prescribing stimulating instead of inhibiting treatments could make a difference at the population level, Dr. Marcum noted.
“If we could shift the prescribing a little bit from inhibiting to stimulating, that could possibly reduce dementia risk,” he said.
However, “we’re not suggesting [that all patients] have their regimen switched,” he added.
That’s because inhibiting medications still have an important place in the antihypertensive treatment armamentarium, Dr. Marcum noted. As an example, beta-blockers are used post heart attack.
As well, factors such as cost and side effects should be taken into consideration when prescribing an antihypertensive drug.
The new results could be used to set up a comparison in a future randomized controlled trial that would provide the strongest evidence for estimating causal effects of treatments, said Dr. Marcum.
‘More convincing’
Carlos G. Santos-Gallego, MD, Icahn School of Medicine at Mount Sinai, New York, said the study is “more convincing” than previous related research, as it has a larger sample size and a longer follow-up.
“And the exquisite statistical analysis gives more robustness, more solidity, to the hypothesis that drugs that stimulate type 2 and 4 angiotensin II receptors might be protective for dementia,” said Dr. Santos-Gallego, who was not involved with the research.
However, he noted that the retrospective study had some limitations, including the underdiagnosis of dementia. “The diagnosis of dementia is, honestly, very poorly done in the clinical setting,” he said.
As well, the study could be subject to “confounding by indication,” Dr. Santos-Gallego said. “There could be a third variable, another confounding factor, that’s responsible both for the dementia and for the prescription of these drugs,” he added.
For example, he noted that comorbidities such as atrial fibrillation, myocardial infarction, and heart failure might increase the risk of dementia.
He agreed with the investigators that a randomized clinical trial would address these limitations. “All comorbidities would be equally shared” in the randomized groups, and all participants would be given “a specific test for dementia at the same time,” Dr. Santos-Gallego said.
Still, he noted that the new results are in keeping with hypertension guidelines that recommend stimulating drugs.
“This trial definitely shows that the current hypertension guidelines are good treatment for our patients, not only to control blood pressure and not only to prevent infarction to prevent stroke but also to prevent dementia,” said Dr. Santos-Gallego.
Also commenting for this news organization, Heather Snyder, PhD, vice president of medical and scientific relations at the Alzheimer’s Association, said the new data provide “clarity” on why previous research had differing results on the effect of antihypertensives on cognition.
Among the caveats of this new analysis is that “it’s unclear if the demographics in this study are fully representative of Medicare beneficiaries,” said Dr. Snyder.
She, too, said a clinical trial is important “to understand if there is a preventative and/or treatment potential in the medications that stimulate type 2 and 4 angiotensin II receptors.”
The study received funding from the National Institute on Aging. Dr. Marcum and Dr. Santos-Gallego have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA approves second antiamyloid for Alzheimer’s disease
Like its controversial cousin aducanumab (Aduhelm, Biogen/Eisai), lecanemab was approved under the FDA’s accelerated approval pathway, which can be used to fast-track a drug that provides a meaningful therapeutic advantage over existing treatments for a serious or life-threatening illness.
Unlike aducanumab, however, there was no formal FDA advisory committee meeting on lecanemab prior to approval.
“Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones,” Billy Dunn, MD, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, said in a press release.
“This treatment option is the latest therapy to target and affect the underlying disease process of Alzheimer’s, instead of only treating the symptoms of the disease,” Dr. Dunn added.
Eisai has reported that lecanemab will cost $26,500 a year.
Modest benefit, adverse events
The FDA noted, “The labeling states that treatment with Leqembi should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was studied in clinical trials.”
The agency approved the treatment on the basis of findings from the CLARITY AD trial, which showed modest cognitive benefit for patients with early AD – but at a cost of increased risk for amyloid-related edema and effusions.
The trial enrolled 1,795 adults with mild cognitive impairment or early Alzheimer’s disease in whom amyloid pathology in the brain had been confirmed. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
After 18 months of treatment, lecanemab slowed cognitive decline by 27%, compared with placebo, as measured by the Clinical Dementia Rating–Sum of Boxes (CDR-SB). This was an absolute difference of 0.45 points (change from baseline, 1.21 for lecanemab vs. 1.66 with placebo; P < .001).
While the results are “welcome news,” a 0.45-point difference on the CDR-SB might not be clinically meaningful, authors of a recent editorial in The Lancet cautioned.
Amyloid-related imaging abnormalities that manifest as edema or microhemorrhages also occurred in one in five patients taking lecanemab.
In addition, a newly published case report in The New England Journal of Medicine describes a patient with Alzheimer’s disease who was taking lecanemab and who died after experiencing numerous intracerebral hemorrhages during treatment with tissue plasminogen activator (tPA) for acute ischemic stroke.
“The findings raise the possibility of cerebral hemorrhages and necrotizing vasculopathy associated with tPA infusion in a patient with cerebrovascular amyloid who had received lecanemab,” the authors wrote.
Alzheimer’s Association reaction
Still, in anticipation of accelerated approval of lecanemab and the antiamyloid drug donanemab (Eli Lilly), which the FDA has also fast-tracked, the Alzheimer’s Association filed a formal request last month with the Centers for Medicare & Medicaid Services asking that it provide full and unrestricted coverage for FDA-approved Alzheimer’s disease treatments.
In a letter addressed to CMS administrator Chiquita Brooks-LaSure, the association asked the agency to remove the requirements for “coverage with evidence development” in its national coverage determination for FDA-approved antiamyloid monoclonal antibodies.
“Each day matters when it comes to slowing the progression of this disease,” Joanne Pike, DrPH, president and CEO for the Alzheimer’s Association, noted in a news release at the time.
“The current CMS policy to severely limit access to these treatments eliminates people’s options, is resulting in continued irreversible disease progression, and contributes to greater health inequities. That’s not acceptable,” Dr. Pike added.
After news of today’s approval was released, Dr. Pike noted in a new release, “The Alzheimer’s Association welcomes and celebrates this action by the FDA. We now have a second approved treatment that changes the course of Alzheimer’s disease in a meaningful way for people in the early stages of the disease.”
Maria C. Carrillo, PhD, chief science officer at the Alzheimer’s Association, called today’s approval “a milestone achievement.”
“The progress we’ve seen in not only this class of treatments but also in the diversification of treatment types and targets over the past few years is exciting and provides real hope to those impacted by this devastating disease,” Dr. Carrillo said.
Critical issues
Commenting on the approval, Alvaro Pascual-Leone, MD, PhD, professor of neurology at Harvard Medical School, Boston, and chief medical officer at Linus Health, said FDA approval of lecanemab and its adoption in the clinic represent a “very exciting development and prospect; but arguably some critical issues need to be considered.”
He noted that the health care system “is not currently prepared to cope with the challenges and demands of lecanemab,” as well as future pharmacologic agents.
“First, we need better workflows to identify suitable patients who can most benefit from this treatment,” said Dr. Pascual-Leone. He added that beyond identification of cognitive difficulties, amyloid status will need to be determined.
“Presently, this requires expensive and invasive tests,” such as positron-emission tomography scans or lumbar punctures for cerebrospinal fluid analysis. However, these are not fully covered by insurance companies and would be challenging to fully scale, he noted.
“In addition to screening, health systems will need to resolve the logistics challenges around the administration of lecanemab with twice-monthly infusions and the need for careful longitudinal evaluations for potential side effects,” said Dr. Pascual-Leone.
“While lecanemab may represent the first disease-modifying therapy widely available for early Alzheimer’s disease, the likely more promising approach is the addition of other therapies to lecanemab as part of a multi-intervention strategy combining pharmacologic and nonpharmacologic interventions,” he added.
Dr. Pascual-Leone has served as a paid member on scientific advisory boards for Neuroelectrics, Magstim, TetraNeuron, Skin2Neuron, MedRhythms, and Hearts Radiant and is a cofounder of TI Solutions and Linus Health.
A version of this article first appeared on Medscape.com.
This article was updated 1/9/23.
Like its controversial cousin aducanumab (Aduhelm, Biogen/Eisai), lecanemab was approved under the FDA’s accelerated approval pathway, which can be used to fast-track a drug that provides a meaningful therapeutic advantage over existing treatments for a serious or life-threatening illness.
Unlike aducanumab, however, there was no formal FDA advisory committee meeting on lecanemab prior to approval.
“Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones,” Billy Dunn, MD, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, said in a press release.
“This treatment option is the latest therapy to target and affect the underlying disease process of Alzheimer’s, instead of only treating the symptoms of the disease,” Dr. Dunn added.
Eisai has reported that lecanemab will cost $26,500 a year.
Modest benefit, adverse events
The FDA noted, “The labeling states that treatment with Leqembi should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was studied in clinical trials.”
The agency approved the treatment on the basis of findings from the CLARITY AD trial, which showed modest cognitive benefit for patients with early AD – but at a cost of increased risk for amyloid-related edema and effusions.
The trial enrolled 1,795 adults with mild cognitive impairment or early Alzheimer’s disease in whom amyloid pathology in the brain had been confirmed. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
After 18 months of treatment, lecanemab slowed cognitive decline by 27%, compared with placebo, as measured by the Clinical Dementia Rating–Sum of Boxes (CDR-SB). This was an absolute difference of 0.45 points (change from baseline, 1.21 for lecanemab vs. 1.66 with placebo; P < .001).
While the results are “welcome news,” a 0.45-point difference on the CDR-SB might not be clinically meaningful, authors of a recent editorial in The Lancet cautioned.
Amyloid-related imaging abnormalities that manifest as edema or microhemorrhages also occurred in one in five patients taking lecanemab.
In addition, a newly published case report in The New England Journal of Medicine describes a patient with Alzheimer’s disease who was taking lecanemab and who died after experiencing numerous intracerebral hemorrhages during treatment with tissue plasminogen activator (tPA) for acute ischemic stroke.
“The findings raise the possibility of cerebral hemorrhages and necrotizing vasculopathy associated with tPA infusion in a patient with cerebrovascular amyloid who had received lecanemab,” the authors wrote.
Alzheimer’s Association reaction
Still, in anticipation of accelerated approval of lecanemab and the antiamyloid drug donanemab (Eli Lilly), which the FDA has also fast-tracked, the Alzheimer’s Association filed a formal request last month with the Centers for Medicare & Medicaid Services asking that it provide full and unrestricted coverage for FDA-approved Alzheimer’s disease treatments.
In a letter addressed to CMS administrator Chiquita Brooks-LaSure, the association asked the agency to remove the requirements for “coverage with evidence development” in its national coverage determination for FDA-approved antiamyloid monoclonal antibodies.
“Each day matters when it comes to slowing the progression of this disease,” Joanne Pike, DrPH, president and CEO for the Alzheimer’s Association, noted in a news release at the time.
“The current CMS policy to severely limit access to these treatments eliminates people’s options, is resulting in continued irreversible disease progression, and contributes to greater health inequities. That’s not acceptable,” Dr. Pike added.
After news of today’s approval was released, Dr. Pike noted in a new release, “The Alzheimer’s Association welcomes and celebrates this action by the FDA. We now have a second approved treatment that changes the course of Alzheimer’s disease in a meaningful way for people in the early stages of the disease.”
Maria C. Carrillo, PhD, chief science officer at the Alzheimer’s Association, called today’s approval “a milestone achievement.”
“The progress we’ve seen in not only this class of treatments but also in the diversification of treatment types and targets over the past few years is exciting and provides real hope to those impacted by this devastating disease,” Dr. Carrillo said.
Critical issues
Commenting on the approval, Alvaro Pascual-Leone, MD, PhD, professor of neurology at Harvard Medical School, Boston, and chief medical officer at Linus Health, said FDA approval of lecanemab and its adoption in the clinic represent a “very exciting development and prospect; but arguably some critical issues need to be considered.”
He noted that the health care system “is not currently prepared to cope with the challenges and demands of lecanemab,” as well as future pharmacologic agents.
“First, we need better workflows to identify suitable patients who can most benefit from this treatment,” said Dr. Pascual-Leone. He added that beyond identification of cognitive difficulties, amyloid status will need to be determined.
“Presently, this requires expensive and invasive tests,” such as positron-emission tomography scans or lumbar punctures for cerebrospinal fluid analysis. However, these are not fully covered by insurance companies and would be challenging to fully scale, he noted.
“In addition to screening, health systems will need to resolve the logistics challenges around the administration of lecanemab with twice-monthly infusions and the need for careful longitudinal evaluations for potential side effects,” said Dr. Pascual-Leone.
“While lecanemab may represent the first disease-modifying therapy widely available for early Alzheimer’s disease, the likely more promising approach is the addition of other therapies to lecanemab as part of a multi-intervention strategy combining pharmacologic and nonpharmacologic interventions,” he added.
Dr. Pascual-Leone has served as a paid member on scientific advisory boards for Neuroelectrics, Magstim, TetraNeuron, Skin2Neuron, MedRhythms, and Hearts Radiant and is a cofounder of TI Solutions and Linus Health.
A version of this article first appeared on Medscape.com.
This article was updated 1/9/23.
Like its controversial cousin aducanumab (Aduhelm, Biogen/Eisai), lecanemab was approved under the FDA’s accelerated approval pathway, which can be used to fast-track a drug that provides a meaningful therapeutic advantage over existing treatments for a serious or life-threatening illness.
Unlike aducanumab, however, there was no formal FDA advisory committee meeting on lecanemab prior to approval.
“Alzheimer’s disease immeasurably incapacitates the lives of those who suffer from it and has devastating effects on their loved ones,” Billy Dunn, MD, director of the Office of Neuroscience in the FDA’s Center for Drug Evaluation and Research, said in a press release.
“This treatment option is the latest therapy to target and affect the underlying disease process of Alzheimer’s, instead of only treating the symptoms of the disease,” Dr. Dunn added.
Eisai has reported that lecanemab will cost $26,500 a year.
Modest benefit, adverse events
The FDA noted, “The labeling states that treatment with Leqembi should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was studied in clinical trials.”
The agency approved the treatment on the basis of findings from the CLARITY AD trial, which showed modest cognitive benefit for patients with early AD – but at a cost of increased risk for amyloid-related edema and effusions.
The trial enrolled 1,795 adults with mild cognitive impairment or early Alzheimer’s disease in whom amyloid pathology in the brain had been confirmed. Treatment consisted of lecanemab 10 mg/kg biweekly or matching placebo.
After 18 months of treatment, lecanemab slowed cognitive decline by 27%, compared with placebo, as measured by the Clinical Dementia Rating–Sum of Boxes (CDR-SB). This was an absolute difference of 0.45 points (change from baseline, 1.21 for lecanemab vs. 1.66 with placebo; P < .001).
While the results are “welcome news,” a 0.45-point difference on the CDR-SB might not be clinically meaningful, authors of a recent editorial in The Lancet cautioned.
Amyloid-related imaging abnormalities that manifest as edema or microhemorrhages also occurred in one in five patients taking lecanemab.
In addition, a newly published case report in The New England Journal of Medicine describes a patient with Alzheimer’s disease who was taking lecanemab and who died after experiencing numerous intracerebral hemorrhages during treatment with tissue plasminogen activator (tPA) for acute ischemic stroke.
“The findings raise the possibility of cerebral hemorrhages and necrotizing vasculopathy associated with tPA infusion in a patient with cerebrovascular amyloid who had received lecanemab,” the authors wrote.
Alzheimer’s Association reaction
Still, in anticipation of accelerated approval of lecanemab and the antiamyloid drug donanemab (Eli Lilly), which the FDA has also fast-tracked, the Alzheimer’s Association filed a formal request last month with the Centers for Medicare & Medicaid Services asking that it provide full and unrestricted coverage for FDA-approved Alzheimer’s disease treatments.
In a letter addressed to CMS administrator Chiquita Brooks-LaSure, the association asked the agency to remove the requirements for “coverage with evidence development” in its national coverage determination for FDA-approved antiamyloid monoclonal antibodies.
“Each day matters when it comes to slowing the progression of this disease,” Joanne Pike, DrPH, president and CEO for the Alzheimer’s Association, noted in a news release at the time.
“The current CMS policy to severely limit access to these treatments eliminates people’s options, is resulting in continued irreversible disease progression, and contributes to greater health inequities. That’s not acceptable,” Dr. Pike added.
After news of today’s approval was released, Dr. Pike noted in a new release, “The Alzheimer’s Association welcomes and celebrates this action by the FDA. We now have a second approved treatment that changes the course of Alzheimer’s disease in a meaningful way for people in the early stages of the disease.”
Maria C. Carrillo, PhD, chief science officer at the Alzheimer’s Association, called today’s approval “a milestone achievement.”
“The progress we’ve seen in not only this class of treatments but also in the diversification of treatment types and targets over the past few years is exciting and provides real hope to those impacted by this devastating disease,” Dr. Carrillo said.
Critical issues
Commenting on the approval, Alvaro Pascual-Leone, MD, PhD, professor of neurology at Harvard Medical School, Boston, and chief medical officer at Linus Health, said FDA approval of lecanemab and its adoption in the clinic represent a “very exciting development and prospect; but arguably some critical issues need to be considered.”
He noted that the health care system “is not currently prepared to cope with the challenges and demands of lecanemab,” as well as future pharmacologic agents.
“First, we need better workflows to identify suitable patients who can most benefit from this treatment,” said Dr. Pascual-Leone. He added that beyond identification of cognitive difficulties, amyloid status will need to be determined.
“Presently, this requires expensive and invasive tests,” such as positron-emission tomography scans or lumbar punctures for cerebrospinal fluid analysis. However, these are not fully covered by insurance companies and would be challenging to fully scale, he noted.
“In addition to screening, health systems will need to resolve the logistics challenges around the administration of lecanemab with twice-monthly infusions and the need for careful longitudinal evaluations for potential side effects,” said Dr. Pascual-Leone.
“While lecanemab may represent the first disease-modifying therapy widely available for early Alzheimer’s disease, the likely more promising approach is the addition of other therapies to lecanemab as part of a multi-intervention strategy combining pharmacologic and nonpharmacologic interventions,” he added.
Dr. Pascual-Leone has served as a paid member on scientific advisory boards for Neuroelectrics, Magstim, TetraNeuron, Skin2Neuron, MedRhythms, and Hearts Radiant and is a cofounder of TI Solutions and Linus Health.
A version of this article first appeared on Medscape.com.
This article was updated 1/9/23.
IV ketamine a promising option for resistant depression in older adults
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
Results showed nearly 50% of participants responded to ketamine and 25% achieved complete remission from TRD, as measured by scores on the Montgomery-Asberg Depression Rating Scale (MADRS).
“Our pilot study suggests that IV ketamine is well-tolerated, safe, and associated with improvement in late-life TRD,” co-investigator Marie Anne Gebara, MD, assistant professor of psychiatry at the University of Pittsburgh, told this news organization.
Dr. Gebara pointed out the treatment “may not be appropriate for all patients with TRD,” such as those with a history of psychotic symptoms or uncontrolled hypertension; but “it appears to be a promising option.”
The findings were published online in the American Journal of Geriatric Psychiatry.
Lack of data in seniors
Although ketamine has been shown in prior research to rapidly reduce suicidal ideation in adults, there has been a lack of data on its efficacy and safety in older adults, the current investigators note.
“Almost 50% of older adults suffering from depression have TRD, which is a leading cause of disability, excess mortality from suicide, and dementia,” Dr. Gebara said.
She added that after two failed trials of antidepressants, “older adults have few evidence-based choices: aripiprazole or bupropion augmentation, transcranial magnetic stimulation, or electroconvulsive therapy. Novel treatments with rapid benefit are needed as long-term outcomes are poor and recurrence rates are high.”
Dr. Gebara and colleagues at five sites (Columbia University, New York State Psychiatric Institute, University of Toronto, University of Pittsburgh, and Washington University in St. Louis) each enrolled five participants aged 60 and older into the pilot study between October 2020 and November 2021, for a total of 25 participants (mean age, 71 years).
Each participant was recruited from patient registries or referred by behavioral health or primary care providers and diagnosed with TRD, which was defined as an episode of major depressive disorder without psychotic features that persisted despite two or more trials of antidepressants including at least one evidence-based second-line treatment.
Participants had to take an oral antidepressant dosage for at least 1 month prior to the start of the IV ketamine infusions, and continue their antidepressant for the length of the trial.
They received IV ketamine twice weekly for 4 weeks. The dosage was weight-dependent.
At the end of the 4 weeks, participants who achieved a MADRS total score of less than 10 or had a 30% or greater reduction from their baseline MADRS score entered another 4-week phase of the trial. This phase consisted of once-weekly administration of IV ketamine.
Larger plans
Results showed 15 of the 25 participants (60%) experienced a 30% or higher reduction in MADRS scores in the first phase of the study. The mean change in MADRS total score from the beginning to the end of the first phase was a decrease of 9.4 points (P < .01).
At the end of the continuation phase, half (48%) met criteria for response and 27% met criteria for remission.
After ketamine administration, the researchers also found an improvement in Fluid Cognition Composite Score (Cohen’s d value = .61), indicating a medium to large effect size, and in three measures of executive function.
Overall, adverse events were rare and did not keep patients from participating in the study, the investigators note. Five of the 25 participants reported infusion-induced hypertension that was transient.
Study limitations cited include the small sample size and the absence of randomization and placebo control or comparison treatment.
“We were very pleased with these findings because they establish the safety of this novel intervention in older adults,” Dr. Gebara said.
“After establishing safety and tolerability, we can plan for larger, randomized controlled trials that will allow us to determine the effectiveness of IV ketamine for older adults with TRD,” she added.
Multiple mechanisms
In a comment, Gerard Sanacora, MD, PhD, professor of psychiatry at Yale University and director of the Yale Depression Research Program, New Haven, Conn., noted multiple mechanisms likely contribute to the antidepressant effects of ketamine.
Dr. Sanacora has independently researched the effects of ketamine but was not involved with the current study.
“Much of the work to date has focused on the drug’s proximal effects on the glutamatergic neurotransmitter system and the resulting enhancement of adaptive neuroplasticity in several brain regions,” he said.
“However, there is also evidence to suggest other neurotransmitter systems and possibly even neuroinflammatory regulators are also contributing to the effect,” Dr. Sanacora added.
He noted that these mechanisms are also likely amplified by the “hope, optimism, expectations, and improved medical management overall that are known to be associated with treatments that require close monitoring and follow-up with health care providers.”
Dr. Gebara noted that “internal/department funds at each site” were used to support the study. She also reported receiving support from Otsuka US. Disclosures for the other investigators are listed in the original article. Dr. Sanacora has reported having “no major direct conflicts” with the study.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
Small study finds high dose vitamin D relieved toxic erythema of chemotherapy
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
seen on an inpatient dermatology consultative service.
Currently, chemotherapy cessation, delay, or dose modification are the “only reliable methods of resolving TEC,” and supportive agents such as topical corticosteroids, topical keratolytics, and pain control are associated with variable and “relatively slow improvement involving 2 to 4 weeks of recovery after chemotherapy interruption,” Cuong V. Nguyen, MD, of the department of dermatology at Northwestern University, Chicago, and colleagues, wrote in a research letter.
Onset of TEC in the six patients occurred a mean of 8.5 days after chemotherapy. Vitamin D – 50,000 IU for one patient and 100,000 IU for the others – was administered a mean of 4.3 days from rash onset and again in 7 days. Triamcinolone, 0.1%, or clobetasol, 0.05%, ointments were also prescribed.
All patients experienced symptomatic improvement in pain, pruritus, or swelling within a day of the first vitamin D treatment, and improvement in redness within 1 to 4 days, the authors said. The second treatment was administered for residual symptoms.
Adam Friedman, MD, professor and chair of dermatology and director of the supportive oncodermatology clinic at George Washington University, Washington, said that supporting patients through the “expected, disabling and often treatment-limiting side effects of oncologic therapies” is an area that is “in its infancy” and is characterized by limited evidence-based approaches.
“Creativity is therefore a must,” he said, commenting on the research letter. “Practice starts with anecdote, and this is certainly an exciting finding ... I look forward to trialing this with our patients at GW.”
Five of the six patients had a hematologic condition that required induction chemotherapy before hematopoietic stem cell transplant, and one was receiving regorafenib for treatment of glioblastoma multiforme. Diagnosis of TEC was established by clinical presentation, and five of the six patients underwent a biopsy. Biopsy findings were consistent with a TEC diagnosis in three patients, and showed nonspecific perivascular dermatitis in two, the investigators reported.
Further research is needed to determine optimal dosing, “delineate safety concerns and potential role in cancer treatment, and establish whether a durable response in patients with continuous chemotherapy, such as in an outpatient setting, is possible,” they said.
Dr. Nguyen and his coauthors reported no conflict of interest disclosures.
FROM JAMA DERMATOLOGY
New osteoporosis guideline says start with a bisphosphonate
This is the first update for 5 years since the previous guidance was published in 2017.
It strongly recommends initial therapy with bisphosphonates for postmenopausal women with osteoporosis, as well as men with osteoporosis, among other recommendations.
However, the author of an accompanying editorial, Susan M. Ott, MD, says: “The decision to start a bisphosphonate is actually not that easy.”
She also queries some of the other recommendations in the guidance.
Her editorial, along with the guideline by Amir Qaseem, MD, PhD, MPH, and colleagues, and systematic review by Chelsea Ayers, MPH, and colleagues, were published in the Annals of Internal Medicine.
Ryan D. Mire, MD, MACP, president of the ACP, gave a brief overview of the new guidance in a video.
Systematic review
The ACP commissioned a review of the evidence because it says new data have emerged on the efficacy of newer medications for osteoporosis and low bone mass, as well as treatment comparisons, and treatment in men.
The review authors identified 34 randomized controlled trials (in 100 publications) and 36 observational studies, which evaluated the following pharmacologic interventions:
- Antiresorptive drugs: four bisphosphonates (alendronate, ibandronate, risedronate, zoledronate) and a RANK ligand inhibitor (denosumab).
- Anabolic drugs: an analog of human parathyroid hormone (PTH)–related protein (abaloparatide), recombinant human PTH (teriparatide), and a sclerostin inhibitor (romosozumab).
- Estrogen agonists: selective estrogen receptor modulators (bazedoxifene, raloxifene).
The authors focused on effectiveness and harms of active drugs compared with placebo or bisphosphonates.
Major changes from 2017 guidelines, some questions
“Though there are many nuanced changes in this [2023 guideline] version, perhaps the major change is the explicit hierarchy of pharmacologic recommendations: bisphosphonates first, then denosumab,” Thomas G. Cooney, MD, senior author of the clinical guideline, explained in an interview.
“Bisphosphonates had the most favorable balance among benefits, harms, patient values and preferences, and cost among the examined drugs in postmenopausal females with primary osteoporosis,” Dr. Cooney, professor of medicine, Oregon Health & Science University, Portland, noted, as is stated in the guideline.
“Denosumab also had a favorable long-term net benefit, but bisphosphonates are much cheaper than other pharmacologic treatments and available in generic formulations,” the document states.
The new guideline suggests use of denosumab as second-line pharmacotherapy in adults who have contraindications to or experience adverse effects with bisphosphonates.
The choice among bisphosphonates (alendronate, risedronate, zoledronic acid) would be based on a patient-centered discussion between physician and patient, addressing costs (often related to insurance), delivery-mode preferences (oral versus intravenous), and “values,” which includes the patient’s priorities, concerns, and expectations regarding their health care, Dr. Cooney explained.
Another update in the new guideline is, “We also clarify the specific, albeit more limited, role of sclerostin inhibitors and recombinant PTH ‘to reduce the risk of fractures only in females with primary osteoporosis with very high-risk of fracture’,” Dr. Cooney noted.
In addition, the guideline now states, “treatment to reduce the risk of fractures in males rather than limiting it to ‘vertebral fracture’ in men,” as in the 2017 guideline.
It also explicitly includes denosumab as second-line therapy for men, Dr. Cooney noted, but as in 2017, the strength of evidence in men remains low.
“Finally, we also clarified that in females over the age of 65 with low bone mass or osteopenia that an individualized approach be taken to treatment (similar to last guideline), but if treatment is initiated, that a bisphosphonate be used (new content),” he said.
The use of estrogen, treatment duration, drug discontinuation, and serial bone mineral density monitoring were not addressed in this guideline, but will likely be evaluated within 2 to 3 years.
‘Osteoporosis treatment: Not easy’ – editorial
In her editorial, Dr. Ott writes: “The data about bisphosphonates may seem overwhelmingly positive, leading to strong recommendations for their use to treat osteoporosis, but the decision to start a bisphosphonate is actually not that easy.”
“A strong recommendation should be given only when future studies are unlikely to change it,” continues Dr. Ott, professor of medicine, University of Washington, Seattle.
“Yet, data already suggest that, in patients with serious osteoporosis, treatment should start with anabolic medications because previous treatment with either bisphosphonates or denosumab will prevent the anabolic response of newer medications.”
“Starting with bisphosphonate will change the bone so it will not respond to the newer medicines, and then a patient will lose the chance for getting the best improvement,” Dr. Ott clarified in an email to this news organization.
But, in fact, the new guidance does suggest that, to reduce the risk of fractures in females with primary osteoporosis at very high risk of fracture, one should consider use of the sclerostin inhibitor romosozumab (moderate-certainty evidence) or recombinant human parathyroid hormone (teriparatide) (low-certainty evidence) followed by a bisphosphonate (conditional recommendation).
Dr. Ott said: “If the [fracture] risk is high, then we should start with an anabolic medication for 1-2 years. If the risk is medium, then use a bisphosphonate for up to 5 years, and then stop and monitor the patient for signs that the medicine is wearing off,” based on blood and urine tests.
‘We need medicines that will stop bone aging’
Osteopenia is defined by an arbitrary bone density measurement, Dr. Ott explained. “About half of women over 65 will have osteopenia, and by age 85 there are hardly any ‘normal’ women left.”
“We need medicines that will stop bone aging, which might sound impossible, but we should still try,” she continued.
“In the meantime, while waiting on new discoveries,” Dr. Ott said, “I would not use bisphosphonates in patients who did not already have a fracture or whose bone density T-score was better than –2.5 because, in the major study, alendronate did not prevent fractures in this group.”
Many people are worried about bisphosphonates because of problems with the jaw or femur. These are real, but they are very rare during the first 5 years of treatment, Dr. Ott noted. Then the risk starts to rise, up to more than 1 in 1,000 after 8 years. So people can get the benefits of these drugs with very low risk for 5 years.
“An immediate [guideline] update is necessary to address the severity of bone loss and the high risk for vertebral fractures after discontinuation of denosumab,” Dr. Ott urged.
“I don’t agree with using denosumab for osteoporosis as a second-line treatment,” she said. “I would use it only in patients who have cancer or unusually high bone resorption. You have to get a dose strictly every 6 months, and if you need to stop, it is recommended to treat with bisphosphonates. Denosumab is a poor choice for somebody who does not want to take a bisphosphonate. Many patients and even too many doctors do not realize how serious it can be to skip a dose.”
“I also think that men could be treated with anabolic medications,” Dr. Ott said. “Clinical trials show they respond the same as women. Many men have osteoporosis as a consequence of low testosterone, and then they can usually be treated with testosterone. Osteoporosis in men is a serious problem that is too often ignored – almost reverse discrimination.”
It is also unfortunate that the review and recommendations do not address estrogen, one of the most effective medications to prevent osteoporotic fractures, according to Dr. Ott.
Clinical considerations in addition to drug types
The new guideline also advises:
- Clinicians treating adults with osteoporosis should encourage adherence to recommended treatments and healthy lifestyle habits, including exercise, and counseling to evaluate and prevent falls.
- All adults with osteopenia or osteoporosis should have adequate calcium and vitamin D intake, as part of fracture prevention.
- Clinicians should assess baseline fracture risk based on bone density, fracture history, fracture risk factors, and response to prior osteoporosis treatments.
- Current evidence suggests that more than 3-5 years of bisphosphonate therapy reduces risk for new vertebral but not other fractures; however, it also increases risk for long-term harms. Therefore, clinicians should consider stopping bisphosphonate treatment after 5 years unless the patient has a strong indication for treatment continuation.
- The decision for a bisphosphonate holiday (temporary discontinuation) and its duration should be based on baseline fracture risk, medication half-life in bone, and benefits and harms.
- Women treated with an anabolic agent who discontinue it should be offered an antiresorptive agent to preserve gains and because of serious risk for rebound and multiple vertebral fractures.
- Adults older than 65 years with osteoporosis may be at increased risk for falls or other adverse events because of drug interactions.
- Transgender persons have variable risk for low bone mass.
The review and guideline were funded by the ACP. Dr. Ott has reported no relevant disclosures. Relevant financial disclosures for other authors are listed with the guideline and review.
A version of this article first appeared on Medscape.com.
This is the first update for 5 years since the previous guidance was published in 2017.
It strongly recommends initial therapy with bisphosphonates for postmenopausal women with osteoporosis, as well as men with osteoporosis, among other recommendations.
However, the author of an accompanying editorial, Susan M. Ott, MD, says: “The decision to start a bisphosphonate is actually not that easy.”
She also queries some of the other recommendations in the guidance.
Her editorial, along with the guideline by Amir Qaseem, MD, PhD, MPH, and colleagues, and systematic review by Chelsea Ayers, MPH, and colleagues, were published in the Annals of Internal Medicine.
Ryan D. Mire, MD, MACP, president of the ACP, gave a brief overview of the new guidance in a video.
Systematic review
The ACP commissioned a review of the evidence because it says new data have emerged on the efficacy of newer medications for osteoporosis and low bone mass, as well as treatment comparisons, and treatment in men.
The review authors identified 34 randomized controlled trials (in 100 publications) and 36 observational studies, which evaluated the following pharmacologic interventions:
- Antiresorptive drugs: four bisphosphonates (alendronate, ibandronate, risedronate, zoledronate) and a RANK ligand inhibitor (denosumab).
- Anabolic drugs: an analog of human parathyroid hormone (PTH)–related protein (abaloparatide), recombinant human PTH (teriparatide), and a sclerostin inhibitor (romosozumab).
- Estrogen agonists: selective estrogen receptor modulators (bazedoxifene, raloxifene).
The authors focused on effectiveness and harms of active drugs compared with placebo or bisphosphonates.
Major changes from 2017 guidelines, some questions
“Though there are many nuanced changes in this [2023 guideline] version, perhaps the major change is the explicit hierarchy of pharmacologic recommendations: bisphosphonates first, then denosumab,” Thomas G. Cooney, MD, senior author of the clinical guideline, explained in an interview.
“Bisphosphonates had the most favorable balance among benefits, harms, patient values and preferences, and cost among the examined drugs in postmenopausal females with primary osteoporosis,” Dr. Cooney, professor of medicine, Oregon Health & Science University, Portland, noted, as is stated in the guideline.
“Denosumab also had a favorable long-term net benefit, but bisphosphonates are much cheaper than other pharmacologic treatments and available in generic formulations,” the document states.
The new guideline suggests use of denosumab as second-line pharmacotherapy in adults who have contraindications to or experience adverse effects with bisphosphonates.
The choice among bisphosphonates (alendronate, risedronate, zoledronic acid) would be based on a patient-centered discussion between physician and patient, addressing costs (often related to insurance), delivery-mode preferences (oral versus intravenous), and “values,” which includes the patient’s priorities, concerns, and expectations regarding their health care, Dr. Cooney explained.
Another update in the new guideline is, “We also clarify the specific, albeit more limited, role of sclerostin inhibitors and recombinant PTH ‘to reduce the risk of fractures only in females with primary osteoporosis with very high-risk of fracture’,” Dr. Cooney noted.
In addition, the guideline now states, “treatment to reduce the risk of fractures in males rather than limiting it to ‘vertebral fracture’ in men,” as in the 2017 guideline.
It also explicitly includes denosumab as second-line therapy for men, Dr. Cooney noted, but as in 2017, the strength of evidence in men remains low.
“Finally, we also clarified that in females over the age of 65 with low bone mass or osteopenia that an individualized approach be taken to treatment (similar to last guideline), but if treatment is initiated, that a bisphosphonate be used (new content),” he said.
The use of estrogen, treatment duration, drug discontinuation, and serial bone mineral density monitoring were not addressed in this guideline, but will likely be evaluated within 2 to 3 years.
‘Osteoporosis treatment: Not easy’ – editorial
In her editorial, Dr. Ott writes: “The data about bisphosphonates may seem overwhelmingly positive, leading to strong recommendations for their use to treat osteoporosis, but the decision to start a bisphosphonate is actually not that easy.”
“A strong recommendation should be given only when future studies are unlikely to change it,” continues Dr. Ott, professor of medicine, University of Washington, Seattle.
“Yet, data already suggest that, in patients with serious osteoporosis, treatment should start with anabolic medications because previous treatment with either bisphosphonates or denosumab will prevent the anabolic response of newer medications.”
“Starting with bisphosphonate will change the bone so it will not respond to the newer medicines, and then a patient will lose the chance for getting the best improvement,” Dr. Ott clarified in an email to this news organization.
But, in fact, the new guidance does suggest that, to reduce the risk of fractures in females with primary osteoporosis at very high risk of fracture, one should consider use of the sclerostin inhibitor romosozumab (moderate-certainty evidence) or recombinant human parathyroid hormone (teriparatide) (low-certainty evidence) followed by a bisphosphonate (conditional recommendation).
Dr. Ott said: “If the [fracture] risk is high, then we should start with an anabolic medication for 1-2 years. If the risk is medium, then use a bisphosphonate for up to 5 years, and then stop and monitor the patient for signs that the medicine is wearing off,” based on blood and urine tests.
‘We need medicines that will stop bone aging’
Osteopenia is defined by an arbitrary bone density measurement, Dr. Ott explained. “About half of women over 65 will have osteopenia, and by age 85 there are hardly any ‘normal’ women left.”
“We need medicines that will stop bone aging, which might sound impossible, but we should still try,” she continued.
“In the meantime, while waiting on new discoveries,” Dr. Ott said, “I would not use bisphosphonates in patients who did not already have a fracture or whose bone density T-score was better than –2.5 because, in the major study, alendronate did not prevent fractures in this group.”
Many people are worried about bisphosphonates because of problems with the jaw or femur. These are real, but they are very rare during the first 5 years of treatment, Dr. Ott noted. Then the risk starts to rise, up to more than 1 in 1,000 after 8 years. So people can get the benefits of these drugs with very low risk for 5 years.
“An immediate [guideline] update is necessary to address the severity of bone loss and the high risk for vertebral fractures after discontinuation of denosumab,” Dr. Ott urged.
“I don’t agree with using denosumab for osteoporosis as a second-line treatment,” she said. “I would use it only in patients who have cancer or unusually high bone resorption. You have to get a dose strictly every 6 months, and if you need to stop, it is recommended to treat with bisphosphonates. Denosumab is a poor choice for somebody who does not want to take a bisphosphonate. Many patients and even too many doctors do not realize how serious it can be to skip a dose.”
“I also think that men could be treated with anabolic medications,” Dr. Ott said. “Clinical trials show they respond the same as women. Many men have osteoporosis as a consequence of low testosterone, and then they can usually be treated with testosterone. Osteoporosis in men is a serious problem that is too often ignored – almost reverse discrimination.”
It is also unfortunate that the review and recommendations do not address estrogen, one of the most effective medications to prevent osteoporotic fractures, according to Dr. Ott.
Clinical considerations in addition to drug types
The new guideline also advises:
- Clinicians treating adults with osteoporosis should encourage adherence to recommended treatments and healthy lifestyle habits, including exercise, and counseling to evaluate and prevent falls.
- All adults with osteopenia or osteoporosis should have adequate calcium and vitamin D intake, as part of fracture prevention.
- Clinicians should assess baseline fracture risk based on bone density, fracture history, fracture risk factors, and response to prior osteoporosis treatments.
- Current evidence suggests that more than 3-5 years of bisphosphonate therapy reduces risk for new vertebral but not other fractures; however, it also increases risk for long-term harms. Therefore, clinicians should consider stopping bisphosphonate treatment after 5 years unless the patient has a strong indication for treatment continuation.
- The decision for a bisphosphonate holiday (temporary discontinuation) and its duration should be based on baseline fracture risk, medication half-life in bone, and benefits and harms.
- Women treated with an anabolic agent who discontinue it should be offered an antiresorptive agent to preserve gains and because of serious risk for rebound and multiple vertebral fractures.
- Adults older than 65 years with osteoporosis may be at increased risk for falls or other adverse events because of drug interactions.
- Transgender persons have variable risk for low bone mass.
The review and guideline were funded by the ACP. Dr. Ott has reported no relevant disclosures. Relevant financial disclosures for other authors are listed with the guideline and review.
A version of this article first appeared on Medscape.com.
This is the first update for 5 years since the previous guidance was published in 2017.
It strongly recommends initial therapy with bisphosphonates for postmenopausal women with osteoporosis, as well as men with osteoporosis, among other recommendations.
However, the author of an accompanying editorial, Susan M. Ott, MD, says: “The decision to start a bisphosphonate is actually not that easy.”
She also queries some of the other recommendations in the guidance.
Her editorial, along with the guideline by Amir Qaseem, MD, PhD, MPH, and colleagues, and systematic review by Chelsea Ayers, MPH, and colleagues, were published in the Annals of Internal Medicine.
Ryan D. Mire, MD, MACP, president of the ACP, gave a brief overview of the new guidance in a video.
Systematic review
The ACP commissioned a review of the evidence because it says new data have emerged on the efficacy of newer medications for osteoporosis and low bone mass, as well as treatment comparisons, and treatment in men.
The review authors identified 34 randomized controlled trials (in 100 publications) and 36 observational studies, which evaluated the following pharmacologic interventions:
- Antiresorptive drugs: four bisphosphonates (alendronate, ibandronate, risedronate, zoledronate) and a RANK ligand inhibitor (denosumab).
- Anabolic drugs: an analog of human parathyroid hormone (PTH)–related protein (abaloparatide), recombinant human PTH (teriparatide), and a sclerostin inhibitor (romosozumab).
- Estrogen agonists: selective estrogen receptor modulators (bazedoxifene, raloxifene).
The authors focused on effectiveness and harms of active drugs compared with placebo or bisphosphonates.
Major changes from 2017 guidelines, some questions
“Though there are many nuanced changes in this [2023 guideline] version, perhaps the major change is the explicit hierarchy of pharmacologic recommendations: bisphosphonates first, then denosumab,” Thomas G. Cooney, MD, senior author of the clinical guideline, explained in an interview.
“Bisphosphonates had the most favorable balance among benefits, harms, patient values and preferences, and cost among the examined drugs in postmenopausal females with primary osteoporosis,” Dr. Cooney, professor of medicine, Oregon Health & Science University, Portland, noted, as is stated in the guideline.
“Denosumab also had a favorable long-term net benefit, but bisphosphonates are much cheaper than other pharmacologic treatments and available in generic formulations,” the document states.
The new guideline suggests use of denosumab as second-line pharmacotherapy in adults who have contraindications to or experience adverse effects with bisphosphonates.
The choice among bisphosphonates (alendronate, risedronate, zoledronic acid) would be based on a patient-centered discussion between physician and patient, addressing costs (often related to insurance), delivery-mode preferences (oral versus intravenous), and “values,” which includes the patient’s priorities, concerns, and expectations regarding their health care, Dr. Cooney explained.
Another update in the new guideline is, “We also clarify the specific, albeit more limited, role of sclerostin inhibitors and recombinant PTH ‘to reduce the risk of fractures only in females with primary osteoporosis with very high-risk of fracture’,” Dr. Cooney noted.
In addition, the guideline now states, “treatment to reduce the risk of fractures in males rather than limiting it to ‘vertebral fracture’ in men,” as in the 2017 guideline.
It also explicitly includes denosumab as second-line therapy for men, Dr. Cooney noted, but as in 2017, the strength of evidence in men remains low.
“Finally, we also clarified that in females over the age of 65 with low bone mass or osteopenia that an individualized approach be taken to treatment (similar to last guideline), but if treatment is initiated, that a bisphosphonate be used (new content),” he said.
The use of estrogen, treatment duration, drug discontinuation, and serial bone mineral density monitoring were not addressed in this guideline, but will likely be evaluated within 2 to 3 years.
‘Osteoporosis treatment: Not easy’ – editorial
In her editorial, Dr. Ott writes: “The data about bisphosphonates may seem overwhelmingly positive, leading to strong recommendations for their use to treat osteoporosis, but the decision to start a bisphosphonate is actually not that easy.”
“A strong recommendation should be given only when future studies are unlikely to change it,” continues Dr. Ott, professor of medicine, University of Washington, Seattle.
“Yet, data already suggest that, in patients with serious osteoporosis, treatment should start with anabolic medications because previous treatment with either bisphosphonates or denosumab will prevent the anabolic response of newer medications.”
“Starting with bisphosphonate will change the bone so it will not respond to the newer medicines, and then a patient will lose the chance for getting the best improvement,” Dr. Ott clarified in an email to this news organization.
But, in fact, the new guidance does suggest that, to reduce the risk of fractures in females with primary osteoporosis at very high risk of fracture, one should consider use of the sclerostin inhibitor romosozumab (moderate-certainty evidence) or recombinant human parathyroid hormone (teriparatide) (low-certainty evidence) followed by a bisphosphonate (conditional recommendation).
Dr. Ott said: “If the [fracture] risk is high, then we should start with an anabolic medication for 1-2 years. If the risk is medium, then use a bisphosphonate for up to 5 years, and then stop and monitor the patient for signs that the medicine is wearing off,” based on blood and urine tests.
‘We need medicines that will stop bone aging’
Osteopenia is defined by an arbitrary bone density measurement, Dr. Ott explained. “About half of women over 65 will have osteopenia, and by age 85 there are hardly any ‘normal’ women left.”
“We need medicines that will stop bone aging, which might sound impossible, but we should still try,” she continued.
“In the meantime, while waiting on new discoveries,” Dr. Ott said, “I would not use bisphosphonates in patients who did not already have a fracture or whose bone density T-score was better than –2.5 because, in the major study, alendronate did not prevent fractures in this group.”
Many people are worried about bisphosphonates because of problems with the jaw or femur. These are real, but they are very rare during the first 5 years of treatment, Dr. Ott noted. Then the risk starts to rise, up to more than 1 in 1,000 after 8 years. So people can get the benefits of these drugs with very low risk for 5 years.
“An immediate [guideline] update is necessary to address the severity of bone loss and the high risk for vertebral fractures after discontinuation of denosumab,” Dr. Ott urged.
“I don’t agree with using denosumab for osteoporosis as a second-line treatment,” she said. “I would use it only in patients who have cancer or unusually high bone resorption. You have to get a dose strictly every 6 months, and if you need to stop, it is recommended to treat with bisphosphonates. Denosumab is a poor choice for somebody who does not want to take a bisphosphonate. Many patients and even too many doctors do not realize how serious it can be to skip a dose.”
“I also think that men could be treated with anabolic medications,” Dr. Ott said. “Clinical trials show they respond the same as women. Many men have osteoporosis as a consequence of low testosterone, and then they can usually be treated with testosterone. Osteoporosis in men is a serious problem that is too often ignored – almost reverse discrimination.”
It is also unfortunate that the review and recommendations do not address estrogen, one of the most effective medications to prevent osteoporotic fractures, according to Dr. Ott.
Clinical considerations in addition to drug types
The new guideline also advises:
- Clinicians treating adults with osteoporosis should encourage adherence to recommended treatments and healthy lifestyle habits, including exercise, and counseling to evaluate and prevent falls.
- All adults with osteopenia or osteoporosis should have adequate calcium and vitamin D intake, as part of fracture prevention.
- Clinicians should assess baseline fracture risk based on bone density, fracture history, fracture risk factors, and response to prior osteoporosis treatments.
- Current evidence suggests that more than 3-5 years of bisphosphonate therapy reduces risk for new vertebral but not other fractures; however, it also increases risk for long-term harms. Therefore, clinicians should consider stopping bisphosphonate treatment after 5 years unless the patient has a strong indication for treatment continuation.
- The decision for a bisphosphonate holiday (temporary discontinuation) and its duration should be based on baseline fracture risk, medication half-life in bone, and benefits and harms.
- Women treated with an anabolic agent who discontinue it should be offered an antiresorptive agent to preserve gains and because of serious risk for rebound and multiple vertebral fractures.
- Adults older than 65 years with osteoporosis may be at increased risk for falls or other adverse events because of drug interactions.
- Transgender persons have variable risk for low bone mass.
The review and guideline were funded by the ACP. Dr. Ott has reported no relevant disclosures. Relevant financial disclosures for other authors are listed with the guideline and review.
A version of this article first appeared on Medscape.com.
FROM THE ANNALS OF INTERNAL MEDICINE
FDA considers regulating CBD products
The products can have drug-like effects on the body and contain CBD (cannabidiol) and THC (tetrahydrocannabinol). Both CBD and THC can be derived from hemp, which was legalized by Congress in 2018.
“Given what we know about the safety of CBD so far, it raises concerns for FDA about whether these existing regulatory pathways for food and dietary supplements are appropriate for this substance,” FDA Principal Deputy Commissioner Janet Woodcock, MD, told The Wall Street Journal.
A 2021 FDA report valued the CBD market at $4.6 billion and projected it to quadruple by 2026. The only FDA-approved CBD product is an oil called Epidiolex, which can be prescribed for the seizure-associated disease epilepsy. Research on CBD to treat other diseases is ongoing.
Food, beverage, and beauty products containing CBD are sold in stores and online in many forms, including oils, vaporized liquids, and oil-based capsules, but “research supporting the drug’s benefits is still limited,” the Mayo Clinic said.
Recently, investigations have found that many CBD products also contain THC, which can be derived from legal hemp in a form that is referred to as Delta 8 and produces a psychoactive high. The CDC warned in 2022 that people “mistook” THC products for CBD products, which are often sold at the same stores, and experienced “adverse events.”
The Centers for Disease Control and Prevention and FDA warn that much is unknown about CBD and delta-8 products. The CDC says known CBD risks include liver damage; interference with other drugs you are taking, which may lead to injury or serious side effects; drowsiness or sleepiness; diarrhea or changes in appetite; changes in mood, such as crankiness; potential negative effects on fetuses during pregnancy or on babies during breastfeeding; or unintentional poisoning of children when mistaking THC products for CBD products or due to containing other ingredients such as THC or pesticides.
“I don’t think that we can have the perfect be the enemy of the good when we’re looking at such a vast market that is so available and utilized,” Norman Birenbaum, a senior FDA adviser who is working on the regulatory issue, told the Journal. “You’ve got a widely unregulated market.”
A version of this article first appeared on WebMD.com.
The products can have drug-like effects on the body and contain CBD (cannabidiol) and THC (tetrahydrocannabinol). Both CBD and THC can be derived from hemp, which was legalized by Congress in 2018.
“Given what we know about the safety of CBD so far, it raises concerns for FDA about whether these existing regulatory pathways for food and dietary supplements are appropriate for this substance,” FDA Principal Deputy Commissioner Janet Woodcock, MD, told The Wall Street Journal.
A 2021 FDA report valued the CBD market at $4.6 billion and projected it to quadruple by 2026. The only FDA-approved CBD product is an oil called Epidiolex, which can be prescribed for the seizure-associated disease epilepsy. Research on CBD to treat other diseases is ongoing.
Food, beverage, and beauty products containing CBD are sold in stores and online in many forms, including oils, vaporized liquids, and oil-based capsules, but “research supporting the drug’s benefits is still limited,” the Mayo Clinic said.
Recently, investigations have found that many CBD products also contain THC, which can be derived from legal hemp in a form that is referred to as Delta 8 and produces a psychoactive high. The CDC warned in 2022 that people “mistook” THC products for CBD products, which are often sold at the same stores, and experienced “adverse events.”
The Centers for Disease Control and Prevention and FDA warn that much is unknown about CBD and delta-8 products. The CDC says known CBD risks include liver damage; interference with other drugs you are taking, which may lead to injury or serious side effects; drowsiness or sleepiness; diarrhea or changes in appetite; changes in mood, such as crankiness; potential negative effects on fetuses during pregnancy or on babies during breastfeeding; or unintentional poisoning of children when mistaking THC products for CBD products or due to containing other ingredients such as THC or pesticides.
“I don’t think that we can have the perfect be the enemy of the good when we’re looking at such a vast market that is so available and utilized,” Norman Birenbaum, a senior FDA adviser who is working on the regulatory issue, told the Journal. “You’ve got a widely unregulated market.”
A version of this article first appeared on WebMD.com.
The products can have drug-like effects on the body and contain CBD (cannabidiol) and THC (tetrahydrocannabinol). Both CBD and THC can be derived from hemp, which was legalized by Congress in 2018.
“Given what we know about the safety of CBD so far, it raises concerns for FDA about whether these existing regulatory pathways for food and dietary supplements are appropriate for this substance,” FDA Principal Deputy Commissioner Janet Woodcock, MD, told The Wall Street Journal.
A 2021 FDA report valued the CBD market at $4.6 billion and projected it to quadruple by 2026. The only FDA-approved CBD product is an oil called Epidiolex, which can be prescribed for the seizure-associated disease epilepsy. Research on CBD to treat other diseases is ongoing.
Food, beverage, and beauty products containing CBD are sold in stores and online in many forms, including oils, vaporized liquids, and oil-based capsules, but “research supporting the drug’s benefits is still limited,” the Mayo Clinic said.
Recently, investigations have found that many CBD products also contain THC, which can be derived from legal hemp in a form that is referred to as Delta 8 and produces a psychoactive high. The CDC warned in 2022 that people “mistook” THC products for CBD products, which are often sold at the same stores, and experienced “adverse events.”
The Centers for Disease Control and Prevention and FDA warn that much is unknown about CBD and delta-8 products. The CDC says known CBD risks include liver damage; interference with other drugs you are taking, which may lead to injury or serious side effects; drowsiness or sleepiness; diarrhea or changes in appetite; changes in mood, such as crankiness; potential negative effects on fetuses during pregnancy or on babies during breastfeeding; or unintentional poisoning of children when mistaking THC products for CBD products or due to containing other ingredients such as THC or pesticides.
“I don’t think that we can have the perfect be the enemy of the good when we’re looking at such a vast market that is so available and utilized,” Norman Birenbaum, a senior FDA adviser who is working on the regulatory issue, told the Journal. “You’ve got a widely unregulated market.”
A version of this article first appeared on WebMD.com.
Patient With Severe Headache After IV Immunoglobulin
A 35-year-old woman with a history of hypothyroidism and idiopathic small fiber autonomic and sensory neuropathy presented to the emergency department (ED) 48 hours after IV immunoglobulin (IG) infusion with a severe headache, nausea, neck stiffness, photophobia, and episodes of intense positional eye pressure. The patient reported previous episodes of headaches post-IVIG infusion but not nearly as severe. On ED arrival, the patient was afebrile with vital signs within normal limits. Initial laboratory results were notable for levels within reference range parameters: 5.9 × 109/L white blood cell (WBC) count, 13.3 g/dL hemoglobin, 38.7% hematocrit, and 279 × 109/L platelet count; there were no abnormal urinalysis findings, and she was negative for human chorionic gonadotropin.
Due to the patient’s symptoms concerning for an acute intracranial process, a brain computed tomography (CT) without contrast was ordered. The CT demonstrated no intracranial abnormalities, but the patient’s symptoms continued to worsen. The patient was started on IV fluids and 1 g IV acetaminophen and underwent a lumbar puncture (LP). Her opening pressure was elevated at 29 cm H2O (reference range, 6-20 cm), and the fluid was notably clear. During the LP, 25 mL of cerebrospinal fluid (CSF) was collected for laboratory analysis to include a polymerase chain reaction (PCR) panel and cultures, and a closing pressure of 12 cm H2O was recorded at the end of the procedure with the patient reporting some relief of pressure. The patient was admitted to the medicine ward for further workup and observations.The patient’s meningitis/encephalitis PCR panel detected no pathogens in the CSF, but her WBC count was 84 × 109/L (reference range, 4-11) with 30 segmented neutrophils (reference range, 0-6) and red blood cell count of 24 (reference range, 0-1); her normal glucose at 60 mg/dL (reference range, 40-70) and protein of 33 mg/dL (reference range, 15-45) were within normal parameters. Brain magnetic resonance images with and without contrast was inconsistent with any acute intracranial pathology to include subarachnoid hemorrhage or central nervous system neoplasm (Figure 1). Bacterial and fungal cultures were negative.
- What is your diagnosis?
- How would you treat this patient?
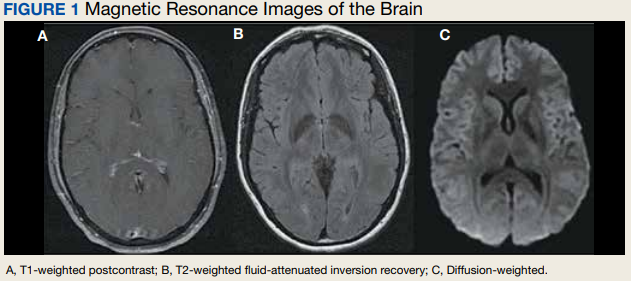
Discussion
Aseptic meningitis presents with a typical clinical picture of meningitis to include headache, stiffened neck, and photophobia. In the event of negative CSF bacterial and fungal cultures and negative viral PCR, a diagnosis of aseptic meningitis is considered.1 Though the differential for aseptic meningitis is broad, in the immunocompetent patient, the most common etiology of aseptic meningitis in the United States is by far viral, and specifically, enterovirus (50.9%). It is less commonly caused by herpes simplex virus (8.3%), varicella zoster virus, and finally, the mosquito-borne St. Louis encephalitis and West Nile viruses typically acquired in the summer or early fall months. Other infectious agents that can present with aseptic meningitis are spirochetes (Lyme disease and syphilis), tuberculous meningitis, fungal infections (cryptococcal meningitis), and other bacterial infections that have a negative culture.
The patient’s history, physical examination, vital signs, imaging, and lumbar puncture findings were most concerning for drug-induced aseptic meningitis (DIAM) secondary to her recent IVIG infusion. An algorithm can be used to work through the diagnostic approach (Figure 2).3,4
Immediate and delayed adverse reactions to IVIG are known risks for IVIG therapy. About 1% to 15% of patients who receive IVIG will experience mild immediate reactions to the infusion.6 These immediate reactions include fever (78.6%), acrocyanosis (71.4%), rash (64.3%), headache (57.1%), shortness of breath (42.8%), hypotension (35.7%), and chest pain (21.4%).
IVIG is an increasingly used biologic pharmacologic agent used for a variety of medical conditions. This can be attributed to its multifaceted properties and ability to fight infection when given as replacement therapy and provide immunomodulation in conjunction with its more well-known anti-inflammatory properties.8 The number of conditions that can potentially benefit from IVIG is so vast that the American Academy of Allergy, Asthma and Immunology had to divide the indication for IVIG therapy into definitely beneficial, probably beneficial, may provide benefit, and unlikely to provide benefit categories.8
Conclusions
We encourage heightened clinical suspicion of DIAM in patients who have recently undergone IVIG infusion and present with meningeal signs (stiff neck, headache, photophobia, and ear/eye pressure) without any evidence of infection on physical examination or laboratory results. With such, we hope to improve clinician suspicion, detection, as well as patient education and outcomes in cases of DIAM.
1. Kareva L, Mironska K, Stavric K, Hasani A. Adverse reactions to intravenous immunoglobulins—our experience. Open Access Maced J Med Sci. 2018;6(12):2359-2362. doi:10.3889/oamjms.2018.513
2. Mount HR, Boyle SD. Aseptic and bacterial meningitis: evaluation, treatment, and prevention. Am Fam Physician. 2017;96(5):314-322.
3. Seehusen DA, Reeves MM, Fomin DA. Cerebrospinal fluid analysis. Am Fam Physician. 2003;68(6):1103-1108.
4. Connolly KJ, Hammer SM. The acute aseptic meningitis syndrome. Infect Dis Clin North Am. 1990;4(4):599-622.
5. Jolles S, Sewell WA, Leighton C. Drug-induced aseptic meningitis: diagnosis and management. Drug Saf. 2000;22(3):215-226. doi:10.2165/00002018-200022030-00005
6. Yelehe-Okouma M, Czmil-Garon J, Pape E, Petitpain N, Gillet P. Drug-induced aseptic meningitis: a mini-review. Fundam Clin Pharmacol. 2018;32(3):252-260. doi:10.1111/fcp.12349
7. Kepa L, Oczko-Grzesik B, Stolarz W, Sobala-Szczygiel B. Drug-induced aseptic meningitis in suspected central nervous system infections. J Clin Neurosci. 2005;12(5):562-564. doi:10.1016/j.jocn.2004.08.024
8. Perez EE, Orange JS, Bonilla F, et al. Update on the use of immunoglobulin in human disease: a review of evidence. J Allergy Clin Immunol. 2017;139(3S):S1-S46. doi:10.1016/j.jaci.2016.09.023
9. Kaarthigeyan K, Burli VV. Aseptic meningitis following intravenous immunoglobulin therapy of common variable immunodeficiency. J Pediatr Neurosci. 2011;6(2):160-161. doi:10.4103/1817-1745.92858
A 35-year-old woman with a history of hypothyroidism and idiopathic small fiber autonomic and sensory neuropathy presented to the emergency department (ED) 48 hours after IV immunoglobulin (IG) infusion with a severe headache, nausea, neck stiffness, photophobia, and episodes of intense positional eye pressure. The patient reported previous episodes of headaches post-IVIG infusion but not nearly as severe. On ED arrival, the patient was afebrile with vital signs within normal limits. Initial laboratory results were notable for levels within reference range parameters: 5.9 × 109/L white blood cell (WBC) count, 13.3 g/dL hemoglobin, 38.7% hematocrit, and 279 × 109/L platelet count; there were no abnormal urinalysis findings, and she was negative for human chorionic gonadotropin.
Due to the patient’s symptoms concerning for an acute intracranial process, a brain computed tomography (CT) without contrast was ordered. The CT demonstrated no intracranial abnormalities, but the patient’s symptoms continued to worsen. The patient was started on IV fluids and 1 g IV acetaminophen and underwent a lumbar puncture (LP). Her opening pressure was elevated at 29 cm H2O (reference range, 6-20 cm), and the fluid was notably clear. During the LP, 25 mL of cerebrospinal fluid (CSF) was collected for laboratory analysis to include a polymerase chain reaction (PCR) panel and cultures, and a closing pressure of 12 cm H2O was recorded at the end of the procedure with the patient reporting some relief of pressure. The patient was admitted to the medicine ward for further workup and observations.The patient’s meningitis/encephalitis PCR panel detected no pathogens in the CSF, but her WBC count was 84 × 109/L (reference range, 4-11) with 30 segmented neutrophils (reference range, 0-6) and red blood cell count of 24 (reference range, 0-1); her normal glucose at 60 mg/dL (reference range, 40-70) and protein of 33 mg/dL (reference range, 15-45) were within normal parameters. Brain magnetic resonance images with and without contrast was inconsistent with any acute intracranial pathology to include subarachnoid hemorrhage or central nervous system neoplasm (Figure 1). Bacterial and fungal cultures were negative.
- What is your diagnosis?
- How would you treat this patient?

Discussion
Aseptic meningitis presents with a typical clinical picture of meningitis to include headache, stiffened neck, and photophobia. In the event of negative CSF bacterial and fungal cultures and negative viral PCR, a diagnosis of aseptic meningitis is considered.1 Though the differential for aseptic meningitis is broad, in the immunocompetent patient, the most common etiology of aseptic meningitis in the United States is by far viral, and specifically, enterovirus (50.9%). It is less commonly caused by herpes simplex virus (8.3%), varicella zoster virus, and finally, the mosquito-borne St. Louis encephalitis and West Nile viruses typically acquired in the summer or early fall months. Other infectious agents that can present with aseptic meningitis are spirochetes (Lyme disease and syphilis), tuberculous meningitis, fungal infections (cryptococcal meningitis), and other bacterial infections that have a negative culture.
The patient’s history, physical examination, vital signs, imaging, and lumbar puncture findings were most concerning for drug-induced aseptic meningitis (DIAM) secondary to her recent IVIG infusion. An algorithm can be used to work through the diagnostic approach (Figure 2).3,4
Immediate and delayed adverse reactions to IVIG are known risks for IVIG therapy. About 1% to 15% of patients who receive IVIG will experience mild immediate reactions to the infusion.6 These immediate reactions include fever (78.6%), acrocyanosis (71.4%), rash (64.3%), headache (57.1%), shortness of breath (42.8%), hypotension (35.7%), and chest pain (21.4%).
IVIG is an increasingly used biologic pharmacologic agent used for a variety of medical conditions. This can be attributed to its multifaceted properties and ability to fight infection when given as replacement therapy and provide immunomodulation in conjunction with its more well-known anti-inflammatory properties.8 The number of conditions that can potentially benefit from IVIG is so vast that the American Academy of Allergy, Asthma and Immunology had to divide the indication for IVIG therapy into definitely beneficial, probably beneficial, may provide benefit, and unlikely to provide benefit categories.8
Conclusions
We encourage heightened clinical suspicion of DIAM in patients who have recently undergone IVIG infusion and present with meningeal signs (stiff neck, headache, photophobia, and ear/eye pressure) without any evidence of infection on physical examination or laboratory results. With such, we hope to improve clinician suspicion, detection, as well as patient education and outcomes in cases of DIAM.
A 35-year-old woman with a history of hypothyroidism and idiopathic small fiber autonomic and sensory neuropathy presented to the emergency department (ED) 48 hours after IV immunoglobulin (IG) infusion with a severe headache, nausea, neck stiffness, photophobia, and episodes of intense positional eye pressure. The patient reported previous episodes of headaches post-IVIG infusion but not nearly as severe. On ED arrival, the patient was afebrile with vital signs within normal limits. Initial laboratory results were notable for levels within reference range parameters: 5.9 × 109/L white blood cell (WBC) count, 13.3 g/dL hemoglobin, 38.7% hematocrit, and 279 × 109/L platelet count; there were no abnormal urinalysis findings, and she was negative for human chorionic gonadotropin.
Due to the patient’s symptoms concerning for an acute intracranial process, a brain computed tomography (CT) without contrast was ordered. The CT demonstrated no intracranial abnormalities, but the patient’s symptoms continued to worsen. The patient was started on IV fluids and 1 g IV acetaminophen and underwent a lumbar puncture (LP). Her opening pressure was elevated at 29 cm H2O (reference range, 6-20 cm), and the fluid was notably clear. During the LP, 25 mL of cerebrospinal fluid (CSF) was collected for laboratory analysis to include a polymerase chain reaction (PCR) panel and cultures, and a closing pressure of 12 cm H2O was recorded at the end of the procedure with the patient reporting some relief of pressure. The patient was admitted to the medicine ward for further workup and observations.The patient’s meningitis/encephalitis PCR panel detected no pathogens in the CSF, but her WBC count was 84 × 109/L (reference range, 4-11) with 30 segmented neutrophils (reference range, 0-6) and red blood cell count of 24 (reference range, 0-1); her normal glucose at 60 mg/dL (reference range, 40-70) and protein of 33 mg/dL (reference range, 15-45) were within normal parameters. Brain magnetic resonance images with and without contrast was inconsistent with any acute intracranial pathology to include subarachnoid hemorrhage or central nervous system neoplasm (Figure 1). Bacterial and fungal cultures were negative.
- What is your diagnosis?
- How would you treat this patient?

Discussion
Aseptic meningitis presents with a typical clinical picture of meningitis to include headache, stiffened neck, and photophobia. In the event of negative CSF bacterial and fungal cultures and negative viral PCR, a diagnosis of aseptic meningitis is considered.1 Though the differential for aseptic meningitis is broad, in the immunocompetent patient, the most common etiology of aseptic meningitis in the United States is by far viral, and specifically, enterovirus (50.9%). It is less commonly caused by herpes simplex virus (8.3%), varicella zoster virus, and finally, the mosquito-borne St. Louis encephalitis and West Nile viruses typically acquired in the summer or early fall months. Other infectious agents that can present with aseptic meningitis are spirochetes (Lyme disease and syphilis), tuberculous meningitis, fungal infections (cryptococcal meningitis), and other bacterial infections that have a negative culture.
The patient’s history, physical examination, vital signs, imaging, and lumbar puncture findings were most concerning for drug-induced aseptic meningitis (DIAM) secondary to her recent IVIG infusion. An algorithm can be used to work through the diagnostic approach (Figure 2).3,4
Immediate and delayed adverse reactions to IVIG are known risks for IVIG therapy. About 1% to 15% of patients who receive IVIG will experience mild immediate reactions to the infusion.6 These immediate reactions include fever (78.6%), acrocyanosis (71.4%), rash (64.3%), headache (57.1%), shortness of breath (42.8%), hypotension (35.7%), and chest pain (21.4%).
IVIG is an increasingly used biologic pharmacologic agent used for a variety of medical conditions. This can be attributed to its multifaceted properties and ability to fight infection when given as replacement therapy and provide immunomodulation in conjunction with its more well-known anti-inflammatory properties.8 The number of conditions that can potentially benefit from IVIG is so vast that the American Academy of Allergy, Asthma and Immunology had to divide the indication for IVIG therapy into definitely beneficial, probably beneficial, may provide benefit, and unlikely to provide benefit categories.8
Conclusions
We encourage heightened clinical suspicion of DIAM in patients who have recently undergone IVIG infusion and present with meningeal signs (stiff neck, headache, photophobia, and ear/eye pressure) without any evidence of infection on physical examination or laboratory results. With such, we hope to improve clinician suspicion, detection, as well as patient education and outcomes in cases of DIAM.
1. Kareva L, Mironska K, Stavric K, Hasani A. Adverse reactions to intravenous immunoglobulins—our experience. Open Access Maced J Med Sci. 2018;6(12):2359-2362. doi:10.3889/oamjms.2018.513
2. Mount HR, Boyle SD. Aseptic and bacterial meningitis: evaluation, treatment, and prevention. Am Fam Physician. 2017;96(5):314-322.
3. Seehusen DA, Reeves MM, Fomin DA. Cerebrospinal fluid analysis. Am Fam Physician. 2003;68(6):1103-1108.
4. Connolly KJ, Hammer SM. The acute aseptic meningitis syndrome. Infect Dis Clin North Am. 1990;4(4):599-622.
5. Jolles S, Sewell WA, Leighton C. Drug-induced aseptic meningitis: diagnosis and management. Drug Saf. 2000;22(3):215-226. doi:10.2165/00002018-200022030-00005
6. Yelehe-Okouma M, Czmil-Garon J, Pape E, Petitpain N, Gillet P. Drug-induced aseptic meningitis: a mini-review. Fundam Clin Pharmacol. 2018;32(3):252-260. doi:10.1111/fcp.12349
7. Kepa L, Oczko-Grzesik B, Stolarz W, Sobala-Szczygiel B. Drug-induced aseptic meningitis in suspected central nervous system infections. J Clin Neurosci. 2005;12(5):562-564. doi:10.1016/j.jocn.2004.08.024
8. Perez EE, Orange JS, Bonilla F, et al. Update on the use of immunoglobulin in human disease: a review of evidence. J Allergy Clin Immunol. 2017;139(3S):S1-S46. doi:10.1016/j.jaci.2016.09.023
9. Kaarthigeyan K, Burli VV. Aseptic meningitis following intravenous immunoglobulin therapy of common variable immunodeficiency. J Pediatr Neurosci. 2011;6(2):160-161. doi:10.4103/1817-1745.92858
1. Kareva L, Mironska K, Stavric K, Hasani A. Adverse reactions to intravenous immunoglobulins—our experience. Open Access Maced J Med Sci. 2018;6(12):2359-2362. doi:10.3889/oamjms.2018.513
2. Mount HR, Boyle SD. Aseptic and bacterial meningitis: evaluation, treatment, and prevention. Am Fam Physician. 2017;96(5):314-322.
3. Seehusen DA, Reeves MM, Fomin DA. Cerebrospinal fluid analysis. Am Fam Physician. 2003;68(6):1103-1108.
4. Connolly KJ, Hammer SM. The acute aseptic meningitis syndrome. Infect Dis Clin North Am. 1990;4(4):599-622.
5. Jolles S, Sewell WA, Leighton C. Drug-induced aseptic meningitis: diagnosis and management. Drug Saf. 2000;22(3):215-226. doi:10.2165/00002018-200022030-00005
6. Yelehe-Okouma M, Czmil-Garon J, Pape E, Petitpain N, Gillet P. Drug-induced aseptic meningitis: a mini-review. Fundam Clin Pharmacol. 2018;32(3):252-260. doi:10.1111/fcp.12349
7. Kepa L, Oczko-Grzesik B, Stolarz W, Sobala-Szczygiel B. Drug-induced aseptic meningitis in suspected central nervous system infections. J Clin Neurosci. 2005;12(5):562-564. doi:10.1016/j.jocn.2004.08.024
8. Perez EE, Orange JS, Bonilla F, et al. Update on the use of immunoglobulin in human disease: a review of evidence. J Allergy Clin Immunol. 2017;139(3S):S1-S46. doi:10.1016/j.jaci.2016.09.023
9. Kaarthigeyan K, Burli VV. Aseptic meningitis following intravenous immunoglobulin therapy of common variable immunodeficiency. J Pediatr Neurosci. 2011;6(2):160-161. doi:10.4103/1817-1745.92858