User login
Angioedema risk jumps when switching HF meds
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New renin-angiotensin-system (RAS) inhibitor therapy using sacubitril-valsartan (Entresto) is no more likely to cause angioedema than starting out with an ACE inhibitor or angiotensin receptor blocker (ARB).
But the risk climbs when such patients start on an ACE inhibitor or ARB and then switch to sacubitril-valsartan, compared with those prescribed the newer drug, the only available angiotensin receptor-neprilysin inhibitor (ARNI), in the first place.
Those findings and others from a large database analysis, by researchers at the Food and Drug Administration and Harvard Medical School, may clarify and help alleviate a residual safety concern about the ARNI – that it might promote angioedema – that persists after the drug’s major HF trials.
The angioedema risk increased the most right after the switch to the ARNI from one of the older RAS inhibitors. For example, the overall risk doubled for patients who started with an ARB then switched to sacubitril-valsartan, compared with those who started on the newer drug. But it went up about 2.5 times during the first 14 days after the switch.
A similar pattern emerged for ACE inhibitors, but the increased angioedema risk reached significance only within 2 weeks of the switch from an ACE inhibitor to sacubitril-valsartan compared to starting on the latter.
The analysis, based on data from the FDA’s Sentinel adverse event reporting system, was published in the Journal of the American College of Cardiology.
A rare complication, but ...
Angioedema was rare overall in the study, with an unadjusted rate of about 6.75 per 1,000 person-years for users of ACE inhibitors, less than half that rate for ARB users, and only one-fifth that rate for sacubitril-valsartan recipients.
But even a rare complication can be a worry for drugs as widely used as RAS inhibitors. And it’s not unusual for patients cautiously started on an ACE inhibitor or ARB to be switched to sacubitril-valsartan, which is only recently a core guideline–recommended therapy for HF with reduced ejection fraction.
Such patients transitioning to the ARNI, the current study suggests, should probably be watched closely for signs of angioedema for 2 weeks but especially during the first few days. Indeed, the study’s event curves show most of the extra risk “popping up” right after the switch to sacubitril-valsartan, lead author Efe Eworuke, PhD, told this news organization.
The ARNI’s labeling, which states the drug should follow ACE inhibitors only after 36-hour washout period, “has done justice to this issue,” she said. But “whether clinicians are adhering to that, we can’t tell.”
Potentially, patients who miss the 36-hour washout between ACE inhibitors or ARBs and sacubitril-valsartan may account for the excess angioedema risk seen in the analysis, said Dr. Eworuke, with the FDA’s Center for Drug Evaluation and Research, Silver Spring, Md.
But the analysis doesn’t nail down the window of excess risk to only 36 hours. It suggests that patients switching to the ARNI – even those pausing for 36 hours in between drugs – should probably be monitored “2 weeks or longer,” she said. “They could still have angioedema after the washout period.”
Indeed, the “timing of the switch may be critical,” according to an editorial accompanying the report. “Perhaps a longer initial exposure period of ACE inhibitor or ARB,” beyond 2 weeks, “should be considered before switching to an ARNI,” contended Robert L. Page II, PharmD, MSPH, University of Colorado Skaggs School of Pharmacy and Pharmaceutical Sciences, Aurora.
Moreover, he wrote, the study suggests that “initiation of an ARNI de novo may be safer compared with trialing an ACE inhibitor or ARB then switching to an ARNI,” and “should be a consideration when beginning guideline-directed medical therapy for patients with HF.”
New RAS inhibition with ARNI ‘protective’
Compared with ARNI “new users” who had not received any RAS inhibitor in the prior 6 months, patients in the study who switched from an ACE inhibitor to ARNI (41,548 matched pairs) showed a hazard ratio (HR) for angioedema of 1.62 (95% confidence interval [CI], 0.91-2.89), that is, only a “trend,” the report states.
But that trend became significant when the analysis considered only angioedema cases in the first 14 days after the drug switch: HR, 1.98 (95% CI, 1.11-3.53).
Those switching from an ARB to ARNI, compared with ARNI new users (37,893 matched pairs), showed a significant HR for angioedema of 2.03 (95% CI, 1.16-3.54). The effect was more pronounced when considering only angioedema arising in the first 2 weeks: HR, 2.45 (95% CI, 1.36-4.43).
Compared with new use of ACE inhibitors, new ARNI use (41,998 matched pairs) was “protective,” the report states, with an HR for angioedema of 0.18 (95% CI, 0.11-0.29). So was a switch from ACE inhibitors to the ARNI (69,639 matched pairs), with an HR of 0.31 (95% CI, 0.23-0.43).
But compared with starting with an ARB, ARNI new use (43,755 matched pairs) had a null effect on angioedema risk, HR, 0.59 (95% CI, 0.35-1.01); as did switching from an ARB to ARNI (49,137 matched pairs), HR, 0.85 (95% CI, 0.58-1.26).
The analysis has limitations, Dr. Eworuke acknowledged. The comparator groups probably differed in unknown ways given the limits of propensity matching, for example, and because the FDA’s Sentinel system data can reflect only cases that are reported, the study probably underestimates the true prevalence of angioedema.
For example, a patient may see a clinician for a milder case that resolves without a significant intervention, she noted. But “those types of angioedema would not have been captured by our study.”
Dr. Eworuke disclosed that her comments reflect her views and are not those of the Food and Drug Administration; she and the other authors, as well as editorialist Dr. Page, report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
FDA approves pirtobrutinib for r/r mantle cell lymphoma
Pirtobrutinib is the first and only noncovalent Bruton’s tyrosine kinase inhibitor approved for use in this MCL setting, manufacturer Eli Lilly noted in a press release.
“The approval of Jaypirca represents an important advance for patients with relapsed or refractory MCL, who currently have limited options and historically have had a poor prognosis following discontinuation of treatment with a covalent Bruton’s tyrosine kinase inhibitor,” senior author Michael Wang, MD, University of Texas MD Anderson Cancer Center, Houston, said in the release.
The approval was based on efficacy demonstrated in the open-label, single-arm, phase 1/2 BRUIN trial – a multicenter study assessing 200 mg once-daily oral pirtobrutinib monotherapy in 120 patients with MCL who had previously received a Bruton’s tyrosine kinase inhibitor, most often ibrutinib (Imbruvica, 67%) acalabrutinib (Calquence, 30%) and zanubrutinib (Brukinsa, 8%). Pirtobrutinib was continued until disease progression or unacceptable toxicity.
Study participants had a median of three prior lines of therapy, and 83% discontinued their last Bruton’s tyrosine kinase inhibitor because of refractory or progressive disease.
The overall response rate in pirtobrutinib-treated patients was 50% with a complete response rate of 13%. Estimated median duration of response was 8.3 months, and the estimated duration of response at 6 months occurred in nearly two-thirds of patients.
Adverse reactions that occurred in at least 15% of patients included fatigue, musculoskeletal pain, diarrhea, edema, dyspnea, pneumonia, and bruising. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included decreased neutrophil counts, lymphocyte counts, and platelet counts.
Prescribing information for pirtobrutinib includes warnings and precautions for infections, hemorrhage, cytopenias, atrial fibrillation and flutter, and second primary malignancies, noted the FDA, which granted priority review, fast track designation, and orphan drug designation for the application submitted by Eli Lilly.
“Jaypirca can reestablish Bruton’s tyrosine kinase inhibition in MCL patients previously treated with a covalent Bruton’s tyrosine kinase inhibitor (ibrutinib, acalabrutinib, or zanubrutinib) and extend the benefit of targeting the Bruton’s tyrosine kinase pathway,” according to Eli Lilly’s release.
Dr. Wang added that the agent “has the potential to meaningfully impact the treatment paradigm for relapsed and refractory MCL patients.”
Meghan Gutierrez, CEO at the Lymphoma Research Foundation, also noted that “the approval of Jaypirca brings a new treatment option and, along with that, new hope for people with relapsed or refractory MCL.”
The drug is expected to be available in the United States in the coming weeks, and the confirmatory phase 3 BRUIN trial is currently enrolling patients, Eli Lilly announced. The company also indicated the list price would be $21,000 for a 30-day supply of the 200-mg dose.
Serious adverse events believed to be associated with the use of pirtobrutinib or any medicine or device should be reported to the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article first appeared on Medscape.com.
Pirtobrutinib is the first and only noncovalent Bruton’s tyrosine kinase inhibitor approved for use in this MCL setting, manufacturer Eli Lilly noted in a press release.
“The approval of Jaypirca represents an important advance for patients with relapsed or refractory MCL, who currently have limited options and historically have had a poor prognosis following discontinuation of treatment with a covalent Bruton’s tyrosine kinase inhibitor,” senior author Michael Wang, MD, University of Texas MD Anderson Cancer Center, Houston, said in the release.
The approval was based on efficacy demonstrated in the open-label, single-arm, phase 1/2 BRUIN trial – a multicenter study assessing 200 mg once-daily oral pirtobrutinib monotherapy in 120 patients with MCL who had previously received a Bruton’s tyrosine kinase inhibitor, most often ibrutinib (Imbruvica, 67%) acalabrutinib (Calquence, 30%) and zanubrutinib (Brukinsa, 8%). Pirtobrutinib was continued until disease progression or unacceptable toxicity.
Study participants had a median of three prior lines of therapy, and 83% discontinued their last Bruton’s tyrosine kinase inhibitor because of refractory or progressive disease.
The overall response rate in pirtobrutinib-treated patients was 50% with a complete response rate of 13%. Estimated median duration of response was 8.3 months, and the estimated duration of response at 6 months occurred in nearly two-thirds of patients.
Adverse reactions that occurred in at least 15% of patients included fatigue, musculoskeletal pain, diarrhea, edema, dyspnea, pneumonia, and bruising. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included decreased neutrophil counts, lymphocyte counts, and platelet counts.
Prescribing information for pirtobrutinib includes warnings and precautions for infections, hemorrhage, cytopenias, atrial fibrillation and flutter, and second primary malignancies, noted the FDA, which granted priority review, fast track designation, and orphan drug designation for the application submitted by Eli Lilly.
“Jaypirca can reestablish Bruton’s tyrosine kinase inhibition in MCL patients previously treated with a covalent Bruton’s tyrosine kinase inhibitor (ibrutinib, acalabrutinib, or zanubrutinib) and extend the benefit of targeting the Bruton’s tyrosine kinase pathway,” according to Eli Lilly’s release.
Dr. Wang added that the agent “has the potential to meaningfully impact the treatment paradigm for relapsed and refractory MCL patients.”
Meghan Gutierrez, CEO at the Lymphoma Research Foundation, also noted that “the approval of Jaypirca brings a new treatment option and, along with that, new hope for people with relapsed or refractory MCL.”
The drug is expected to be available in the United States in the coming weeks, and the confirmatory phase 3 BRUIN trial is currently enrolling patients, Eli Lilly announced. The company also indicated the list price would be $21,000 for a 30-day supply of the 200-mg dose.
Serious adverse events believed to be associated with the use of pirtobrutinib or any medicine or device should be reported to the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article first appeared on Medscape.com.
Pirtobrutinib is the first and only noncovalent Bruton’s tyrosine kinase inhibitor approved for use in this MCL setting, manufacturer Eli Lilly noted in a press release.
“The approval of Jaypirca represents an important advance for patients with relapsed or refractory MCL, who currently have limited options and historically have had a poor prognosis following discontinuation of treatment with a covalent Bruton’s tyrosine kinase inhibitor,” senior author Michael Wang, MD, University of Texas MD Anderson Cancer Center, Houston, said in the release.
The approval was based on efficacy demonstrated in the open-label, single-arm, phase 1/2 BRUIN trial – a multicenter study assessing 200 mg once-daily oral pirtobrutinib monotherapy in 120 patients with MCL who had previously received a Bruton’s tyrosine kinase inhibitor, most often ibrutinib (Imbruvica, 67%) acalabrutinib (Calquence, 30%) and zanubrutinib (Brukinsa, 8%). Pirtobrutinib was continued until disease progression or unacceptable toxicity.
Study participants had a median of three prior lines of therapy, and 83% discontinued their last Bruton’s tyrosine kinase inhibitor because of refractory or progressive disease.
The overall response rate in pirtobrutinib-treated patients was 50% with a complete response rate of 13%. Estimated median duration of response was 8.3 months, and the estimated duration of response at 6 months occurred in nearly two-thirds of patients.
Adverse reactions that occurred in at least 15% of patients included fatigue, musculoskeletal pain, diarrhea, edema, dyspnea, pneumonia, and bruising. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included decreased neutrophil counts, lymphocyte counts, and platelet counts.
Prescribing information for pirtobrutinib includes warnings and precautions for infections, hemorrhage, cytopenias, atrial fibrillation and flutter, and second primary malignancies, noted the FDA, which granted priority review, fast track designation, and orphan drug designation for the application submitted by Eli Lilly.
“Jaypirca can reestablish Bruton’s tyrosine kinase inhibition in MCL patients previously treated with a covalent Bruton’s tyrosine kinase inhibitor (ibrutinib, acalabrutinib, or zanubrutinib) and extend the benefit of targeting the Bruton’s tyrosine kinase pathway,” according to Eli Lilly’s release.
Dr. Wang added that the agent “has the potential to meaningfully impact the treatment paradigm for relapsed and refractory MCL patients.”
Meghan Gutierrez, CEO at the Lymphoma Research Foundation, also noted that “the approval of Jaypirca brings a new treatment option and, along with that, new hope for people with relapsed or refractory MCL.”
The drug is expected to be available in the United States in the coming weeks, and the confirmatory phase 3 BRUIN trial is currently enrolling patients, Eli Lilly announced. The company also indicated the list price would be $21,000 for a 30-day supply of the 200-mg dose.
Serious adverse events believed to be associated with the use of pirtobrutinib or any medicine or device should be reported to the FDA’s MedWatch Reporting System or by calling 1-800-FDA-1088.
A version of this article first appeared on Medscape.com.
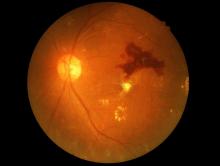
Eye check important before starting semaglutide for diabetes
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
A small potential increased risk of retinopathy worsening at 1 year with injected semaglutide (Ozempic, Novo Nordisk), a glucagon-like peptide 1 (GLP-1) agonist approved for type 2 diabetes, doesn’t outweigh the drug’s cardiovascular benefits but does highlight the need for baseline ophthalmologic evaluation before initiating treatment and ongoing retinal monitoring, researchers say.
That conclusion was based on data from a meta-analysis of the seven major cardiovascular outcomes trials of GLP-1 agonists currently on the market.
The findings were recently published in Diabetes & Metabolic Syndrome: Clinical Research & Reviews, by Stewart G. Albert, MD, and colleagues.
Concerns about retinopathy worsening with the GLP-1 agonist drug class first arose from the SUSTAIN-6 cardiovascular outcomes trial for injectable semaglutide, although a subsequent analysis of data from that trial appeared to suggest the problem is likely due to rapid glucose-lowering in already vulnerable patients rather than a drug-specific effect. This effect had been previously reported, most notably in the landmark Diabetes Control and Complications Trial.
In this new meta-analysis, “we showed that with improvements in A1c there were correlations with decreases in the rate of cardiovascular events but increases in the rate of retinopathy,” Dr. Albert, of St. Louis University, told this news organization.
“As a class of drugs, we did not find an increased rate of retinopathy. The effect of GLP-1 agonists on retinopathy did not appear to be due to an immediate direct toxic effect of the drug. The worsening of the rate of retinopathy was seen with semaglutide after 1 year of therapy and when there was a decrease in A1c of 1%,” he explained.
He noted that because the increased risk was seen primarily among those who already had retinopathy at baseline, “it would seem prudent to know the level of retinopathy either before or plan for close ophthalmologic monitoring around the time of drug initiation ... We routinely evaluate patients with known type 2 diabetes mellitus at yearly intervals for retinopathy. From our data, we saw worsening at 1 year of drug exposure, but we do not know the exact time when the changes occurred during that year.”
The Ozempic label advises that “patients with a history of diabetic retinopathy should be monitored for progression of diabetic retinopathy” but doesn’t specifically mention baseline assessment at the time of drug initiation.
No increase in retinopathy risk for GLP-1 agonist class overall
The seven trials in the meta-analysis comprised 56,004 participants, with baseline retinopathy prevalence ranging from 9% to 31%.
For the GLP-1 agonist class overall, there was no significant increase in the relative rate (RR) of retinopathy (RR, 1.09; P = .36), while there were significant reductions in relative rates of major adverse cardiac events, overall deaths, and cardiovascular deaths (all P < .001 or P = .001).
The increased retinopathy risk was seen only in the subcutaneous semaglutide group (RR, 1.73; P = .02).
The overall number needed to harm was 1,000 and the number to treat was 77. For semaglutide, those values were 77 and 43, respectively.
There was a significant correlation between a decrease in major adverse cardiac events and a decrease in A1c (P = .014), while for retinopathy, the risk increased with improved A1c (P = .076).
Semaglutide subanalysis finds increased retinopathy worsening
Dr. Albert and colleagues conducted a separate subanalysis of 11 studies of semaglutide that enrolled 11,894 patients, of which 6 studies (n = 5,610) were of oral semaglutide (Rybelsus) and 5 studies were of subcutaneous semaglutide (Ozempic; n = 6,284).
In the subanalysis, there was an overall increase in relative rates of new or worsening retinopathy (RR, 1.218; P = .049).
The change in relative rate of retinopathy was predominantly found for subcutaneous semaglutide given for longer than 1 year (RR, 1.559; P = .022) and decreases in A1c of more than 1.0% (RR, 1.590; P = .016). No such differences were seen with oral semaglutide.
A further evaluation of the data without the SUSTAIN 6 trial showed no effect on retinopathy but the analysis lacked power.
Dr. Albert told this news organization: “We did not find an immediate toxic effect of any drug. However, we cannot rule out that there was a cumulative effect of the dose over longer times.”
No disclosures were given.
A version of this article first appeared on Medscape.com.
FROM DIABETES & METABOLIC SYNDROME: CLINICAL RESEARCH & REVIEWS
PCSK9 inhibitors for severe COVID? Pilot trial signals of benefit
PCSK9 inhibitors may best be known for their powerful LDL-lowering effects but are less appreciated as anti-inflammatory agents with potential beyond cardiovascular health.
In a small pilot trial, for example, patients hospitalized with severe COVID-19 who received a single injection of PCSK9 inhibitor became less sick and more likely to survive than those given a placebo. Their 30-day risk of death or intubation fell significantly, as did their levels of the inflammatory cytokine interleukin 6 (IL-6).
Indeed, survival gains in the PCSK9-inhibitor group were greatest among patients with higher baseline concentrations of IL-6. Although the trial wasn’t powered for clinical outcomes, it suggests the drugs’ efficacy in COVID-19 tracks with intensity of inflammation, proposes a report published in the Journal of the American College of Cardiology.
Therefore, “PCSK9 inhibition may represent a novel therapeutic pathway in addition to currently recommended therapeutic approaches for severe COVID-19,” conclude the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
PCSK9 inhibitors as anti-inflammatories
Although the study was small and only hypothesis-generating, the fact that outcomes for actively treated patients were proportional to baseline IL-6 levels “strongly suggests that PCSK9 inhibition can directly modulate inflammation in COVID-19,” argues an editorial accompanying the report.
and likely sheds light on “mechanisms through which PCSK9 inhibition dually modulates lipoprotein metabolism and inflammation,” write Sascha N. Goonewardena, MD, University of Michigan, Ann Arbor, and Robert S. Rosenson, MD, Icahn School of Medicine at Mount Sinai, New York.
The results are consistent with prior evidence that the drugs are anti-inflammatory at least partly because of their interference with inflammatory pathways triggered by PCSK9 and mediated by IL-6, as described by Dr. Navarese and colleagues.
Indeed, they write, PCSK9 inhibitors may improve COVID outcomes mostly through mechanisms unrelated to LDL-receptor expression, “including direct inhibition of PCSK9-triggered inflammation.”
If true, the authors observe, it might explain “why the positive findings of the present study have not been consistently observed in trials involving other lipid-lowering agents, such as statins.” Those drugs are well-known to decrease levels of the inflammatory biomarker C-reactive protein.
In patients with stable coronary disease, in whom inflammation is typically tracked by measuring CRP, “the PCSK9 inhibitors have not been shown to have an anti-inflammatory effect,” Dr. Rosenson further explained.
But the current study’s patients with acute, severe COVID-19, a “profound inflammatory insult” with upregulation of IL-6, were “a good population” for evaluating the drugs’ potential anti-inflammatory effects, Dr. Rosenson said in an interview. The results “are quite enticing but require corroboration in a larger trial.”
A single injection
The IMPACT-SIRIO 5 trial entered 60 adults hospitalized with severe COVID-19 and elevated IL-6 at four centers in Poland. Patients with other known active infections were excluded.
They were randomly assigned double-blind to receive a 140 mg injection of evolocumab (Repatha) or placebo. The 2 groups were similar with respect to demographics, body-mass index, time since symptom onset, and treatments for managing COVID-19 and its complications.
Rates of death or need for intubation at 30 days, the primary endpoint, were 23.3% in the PCSK9-inhibitor group and 53.3% for controls, a risk difference of 30% (95% confidence interval –53.4% to –6.6%). The median durations of oxygen therapy were significantly different at 13 days and 20 days, respectively, the report states.
Serum IL-6 levels fell further over 30 days in the PCSK9-inhibitor group (–56% vs. –21% among controls). A drop by more than 90% was seen in 60% of patients in the PCSK9-inhibitor group and in 27% of controls.
The average hospital stay was shorter for those getting the PCSK9 inhibitor, compared with placebo, 16 days versus 22 days, and their 30-day mortality was numerically lower, 16% versus 33.3%.
Patients’ baseline IL-6 levels above the median, the report states, had a lower mortality on the PCSK9 inhibitor versus placebo (risk difference –37.5%; 95% CI –68.2% to –6.70%).
A larger trial to corroborate these results would potentially enter similar patients hospitalized with COVID-19 with reproducible evidence of an ongoing cytokine storm, such as elevated levels of IL-6, who would be assigned to either a PCSK9 inhibitor or placebo, Dr. Rosenson proposed.
Although the current primary endpoint that combines mortality and intubation was “reasonable” for a small pilot trial, he said, if the researchers embark on a larger study, “they’ll want to look at those events separately.”
Dr. Navarese discloses receiving speaker and consultancy fees from Amgen, Sanofi-Regeneron, Bayer; and grants from Abbott. Disclosures for the other authors are in the report. Rosenson discloses receiving research funding to his institution from Amgen, Arrowhead, Eli Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, CRISPR Therapeutics, Eli Lilly, Lipigon, Novartis, Precision Biosciences, Regeneron, Ultragenyx, and Verve; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer; and owning stock in MediMergent. Dr. Goonewardena reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PCSK9 inhibitors may best be known for their powerful LDL-lowering effects but are less appreciated as anti-inflammatory agents with potential beyond cardiovascular health.
In a small pilot trial, for example, patients hospitalized with severe COVID-19 who received a single injection of PCSK9 inhibitor became less sick and more likely to survive than those given a placebo. Their 30-day risk of death or intubation fell significantly, as did their levels of the inflammatory cytokine interleukin 6 (IL-6).
Indeed, survival gains in the PCSK9-inhibitor group were greatest among patients with higher baseline concentrations of IL-6. Although the trial wasn’t powered for clinical outcomes, it suggests the drugs’ efficacy in COVID-19 tracks with intensity of inflammation, proposes a report published in the Journal of the American College of Cardiology.
Therefore, “PCSK9 inhibition may represent a novel therapeutic pathway in addition to currently recommended therapeutic approaches for severe COVID-19,” conclude the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
PCSK9 inhibitors as anti-inflammatories
Although the study was small and only hypothesis-generating, the fact that outcomes for actively treated patients were proportional to baseline IL-6 levels “strongly suggests that PCSK9 inhibition can directly modulate inflammation in COVID-19,” argues an editorial accompanying the report.
and likely sheds light on “mechanisms through which PCSK9 inhibition dually modulates lipoprotein metabolism and inflammation,” write Sascha N. Goonewardena, MD, University of Michigan, Ann Arbor, and Robert S. Rosenson, MD, Icahn School of Medicine at Mount Sinai, New York.
The results are consistent with prior evidence that the drugs are anti-inflammatory at least partly because of their interference with inflammatory pathways triggered by PCSK9 and mediated by IL-6, as described by Dr. Navarese and colleagues.
Indeed, they write, PCSK9 inhibitors may improve COVID outcomes mostly through mechanisms unrelated to LDL-receptor expression, “including direct inhibition of PCSK9-triggered inflammation.”
If true, the authors observe, it might explain “why the positive findings of the present study have not been consistently observed in trials involving other lipid-lowering agents, such as statins.” Those drugs are well-known to decrease levels of the inflammatory biomarker C-reactive protein.
In patients with stable coronary disease, in whom inflammation is typically tracked by measuring CRP, “the PCSK9 inhibitors have not been shown to have an anti-inflammatory effect,” Dr. Rosenson further explained.
But the current study’s patients with acute, severe COVID-19, a “profound inflammatory insult” with upregulation of IL-6, were “a good population” for evaluating the drugs’ potential anti-inflammatory effects, Dr. Rosenson said in an interview. The results “are quite enticing but require corroboration in a larger trial.”
A single injection
The IMPACT-SIRIO 5 trial entered 60 adults hospitalized with severe COVID-19 and elevated IL-6 at four centers in Poland. Patients with other known active infections were excluded.
They were randomly assigned double-blind to receive a 140 mg injection of evolocumab (Repatha) or placebo. The 2 groups were similar with respect to demographics, body-mass index, time since symptom onset, and treatments for managing COVID-19 and its complications.
Rates of death or need for intubation at 30 days, the primary endpoint, were 23.3% in the PCSK9-inhibitor group and 53.3% for controls, a risk difference of 30% (95% confidence interval –53.4% to –6.6%). The median durations of oxygen therapy were significantly different at 13 days and 20 days, respectively, the report states.
Serum IL-6 levels fell further over 30 days in the PCSK9-inhibitor group (–56% vs. –21% among controls). A drop by more than 90% was seen in 60% of patients in the PCSK9-inhibitor group and in 27% of controls.
The average hospital stay was shorter for those getting the PCSK9 inhibitor, compared with placebo, 16 days versus 22 days, and their 30-day mortality was numerically lower, 16% versus 33.3%.
Patients’ baseline IL-6 levels above the median, the report states, had a lower mortality on the PCSK9 inhibitor versus placebo (risk difference –37.5%; 95% CI –68.2% to –6.70%).
A larger trial to corroborate these results would potentially enter similar patients hospitalized with COVID-19 with reproducible evidence of an ongoing cytokine storm, such as elevated levels of IL-6, who would be assigned to either a PCSK9 inhibitor or placebo, Dr. Rosenson proposed.
Although the current primary endpoint that combines mortality and intubation was “reasonable” for a small pilot trial, he said, if the researchers embark on a larger study, “they’ll want to look at those events separately.”
Dr. Navarese discloses receiving speaker and consultancy fees from Amgen, Sanofi-Regeneron, Bayer; and grants from Abbott. Disclosures for the other authors are in the report. Rosenson discloses receiving research funding to his institution from Amgen, Arrowhead, Eli Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, CRISPR Therapeutics, Eli Lilly, Lipigon, Novartis, Precision Biosciences, Regeneron, Ultragenyx, and Verve; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer; and owning stock in MediMergent. Dr. Goonewardena reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
PCSK9 inhibitors may best be known for their powerful LDL-lowering effects but are less appreciated as anti-inflammatory agents with potential beyond cardiovascular health.
In a small pilot trial, for example, patients hospitalized with severe COVID-19 who received a single injection of PCSK9 inhibitor became less sick and more likely to survive than those given a placebo. Their 30-day risk of death or intubation fell significantly, as did their levels of the inflammatory cytokine interleukin 6 (IL-6).
Indeed, survival gains in the PCSK9-inhibitor group were greatest among patients with higher baseline concentrations of IL-6. Although the trial wasn’t powered for clinical outcomes, it suggests the drugs’ efficacy in COVID-19 tracks with intensity of inflammation, proposes a report published in the Journal of the American College of Cardiology.
Therefore, “PCSK9 inhibition may represent a novel therapeutic pathway in addition to currently recommended therapeutic approaches for severe COVID-19,” conclude the authors, led by Eliano P. Navarese, MD, PhD, Nicolaus Copernicus University, Bydgoszcz, Poland.
PCSK9 inhibitors as anti-inflammatories
Although the study was small and only hypothesis-generating, the fact that outcomes for actively treated patients were proportional to baseline IL-6 levels “strongly suggests that PCSK9 inhibition can directly modulate inflammation in COVID-19,” argues an editorial accompanying the report.
and likely sheds light on “mechanisms through which PCSK9 inhibition dually modulates lipoprotein metabolism and inflammation,” write Sascha N. Goonewardena, MD, University of Michigan, Ann Arbor, and Robert S. Rosenson, MD, Icahn School of Medicine at Mount Sinai, New York.
The results are consistent with prior evidence that the drugs are anti-inflammatory at least partly because of their interference with inflammatory pathways triggered by PCSK9 and mediated by IL-6, as described by Dr. Navarese and colleagues.
Indeed, they write, PCSK9 inhibitors may improve COVID outcomes mostly through mechanisms unrelated to LDL-receptor expression, “including direct inhibition of PCSK9-triggered inflammation.”
If true, the authors observe, it might explain “why the positive findings of the present study have not been consistently observed in trials involving other lipid-lowering agents, such as statins.” Those drugs are well-known to decrease levels of the inflammatory biomarker C-reactive protein.
In patients with stable coronary disease, in whom inflammation is typically tracked by measuring CRP, “the PCSK9 inhibitors have not been shown to have an anti-inflammatory effect,” Dr. Rosenson further explained.
But the current study’s patients with acute, severe COVID-19, a “profound inflammatory insult” with upregulation of IL-6, were “a good population” for evaluating the drugs’ potential anti-inflammatory effects, Dr. Rosenson said in an interview. The results “are quite enticing but require corroboration in a larger trial.”
A single injection
The IMPACT-SIRIO 5 trial entered 60 adults hospitalized with severe COVID-19 and elevated IL-6 at four centers in Poland. Patients with other known active infections were excluded.
They were randomly assigned double-blind to receive a 140 mg injection of evolocumab (Repatha) or placebo. The 2 groups were similar with respect to demographics, body-mass index, time since symptom onset, and treatments for managing COVID-19 and its complications.
Rates of death or need for intubation at 30 days, the primary endpoint, were 23.3% in the PCSK9-inhibitor group and 53.3% for controls, a risk difference of 30% (95% confidence interval –53.4% to –6.6%). The median durations of oxygen therapy were significantly different at 13 days and 20 days, respectively, the report states.
Serum IL-6 levels fell further over 30 days in the PCSK9-inhibitor group (–56% vs. –21% among controls). A drop by more than 90% was seen in 60% of patients in the PCSK9-inhibitor group and in 27% of controls.
The average hospital stay was shorter for those getting the PCSK9 inhibitor, compared with placebo, 16 days versus 22 days, and their 30-day mortality was numerically lower, 16% versus 33.3%.
Patients’ baseline IL-6 levels above the median, the report states, had a lower mortality on the PCSK9 inhibitor versus placebo (risk difference –37.5%; 95% CI –68.2% to –6.70%).
A larger trial to corroborate these results would potentially enter similar patients hospitalized with COVID-19 with reproducible evidence of an ongoing cytokine storm, such as elevated levels of IL-6, who would be assigned to either a PCSK9 inhibitor or placebo, Dr. Rosenson proposed.
Although the current primary endpoint that combines mortality and intubation was “reasonable” for a small pilot trial, he said, if the researchers embark on a larger study, “they’ll want to look at those events separately.”
Dr. Navarese discloses receiving speaker and consultancy fees from Amgen, Sanofi-Regeneron, Bayer; and grants from Abbott. Disclosures for the other authors are in the report. Rosenson discloses receiving research funding to his institution from Amgen, Arrowhead, Eli Lilly, Novartis, and Regeneron; consulting fees from Amgen, Arrowhead, CRISPR Therapeutics, Eli Lilly, Lipigon, Novartis, Precision Biosciences, Regeneron, Ultragenyx, and Verve; speaking fees from Amgen, Kowa, and Regeneron; and royalties from Wolters Kluwer; and owning stock in MediMergent. Dr. Goonewardena reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FDA approves new type 2 diabetes drug bexagliflozin
The U.S. Food and Drug Administration has approved bexagliflozin (Brenzavvy, TheracosBio) for the treatment of adults with type 2 diabetes.
The once-daily 20-mg oral sodium-glucose cotransporter 2 (SGLT2) inhibitor is indicated as an adjunct to diet and exercise to improve glycemic control for those with type 2 diabetes, but not type 1 diabetes. It can be used in adults with an estimated glomerular filtration rate (eGFR) > 30 mL/min per 1.73 m2.
Approval was based on results from 23 clinical trials with more than 5,000 participants, including more than 300 patients with stage 3 kidney disease (eGFR < 60 and > 30 mL/min per 1.73 m2).
In the phase 3 studies, bexagliflozin significantly reduced hemoglobin A1c and fasting blood glucose at 24 weeks as monotherapy or as add-on to metformin and other glucose-lowering drugs and combinations. It also produced modest reductions in body weight and systolic blood pressure.
In the phase 3 Bexagliflozin Efficacy and Safety Trial (BEST) cardiovascular outcomes trial, the drug met its efficacy and safety objectives in patients at high cardiovascular risk. Noninferiority was demonstrated for the composite outcome of cardiovascular death, myocardial infarction, stroke, or unstable angina.
“As a class of drugs, SGLT2 inhibitors have shown tremendous benefit in treating adults with type 2 diabetes,” said Mason Freeman, MD, director of the Translational Research Center at Massachusetts General Hospital, Boston, in a press release from TheracosBio.
“Being involved in all of the clinical trials for Brenzavvy, I am greatly impressed with the efficacy of the drug in reducing blood glucose levels and I believe it is an important addition to the SGLT2 inhibitor class of drugs.”
As with other SGLT2 inhibitors, adverse events seen in the trials include ketoacidosis, lower limb amputation, volume depletion, urosepsis, pyelonephritis, Fournier’s gangrene, genital mycotic infections, and hypoglycemia when used with insulin or insulin secretagogues.
Bexagliflozin joins an already crowded field of SGLT2 inhibitors, some of which have been approved for additional cardiovascular and kidney indications.
Of interest, bexagliflozin was approved by the FDA for diabetes in cats in December 2022, as the first oral new animal drug to improve glycemic control in otherwise healthy cats with diabetes not previously treated with insulin.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved bexagliflozin (Brenzavvy, TheracosBio) for the treatment of adults with type 2 diabetes.
The once-daily 20-mg oral sodium-glucose cotransporter 2 (SGLT2) inhibitor is indicated as an adjunct to diet and exercise to improve glycemic control for those with type 2 diabetes, but not type 1 diabetes. It can be used in adults with an estimated glomerular filtration rate (eGFR) > 30 mL/min per 1.73 m2.
Approval was based on results from 23 clinical trials with more than 5,000 participants, including more than 300 patients with stage 3 kidney disease (eGFR < 60 and > 30 mL/min per 1.73 m2).
In the phase 3 studies, bexagliflozin significantly reduced hemoglobin A1c and fasting blood glucose at 24 weeks as monotherapy or as add-on to metformin and other glucose-lowering drugs and combinations. It also produced modest reductions in body weight and systolic blood pressure.
In the phase 3 Bexagliflozin Efficacy and Safety Trial (BEST) cardiovascular outcomes trial, the drug met its efficacy and safety objectives in patients at high cardiovascular risk. Noninferiority was demonstrated for the composite outcome of cardiovascular death, myocardial infarction, stroke, or unstable angina.
“As a class of drugs, SGLT2 inhibitors have shown tremendous benefit in treating adults with type 2 diabetes,” said Mason Freeman, MD, director of the Translational Research Center at Massachusetts General Hospital, Boston, in a press release from TheracosBio.
“Being involved in all of the clinical trials for Brenzavvy, I am greatly impressed with the efficacy of the drug in reducing blood glucose levels and I believe it is an important addition to the SGLT2 inhibitor class of drugs.”
As with other SGLT2 inhibitors, adverse events seen in the trials include ketoacidosis, lower limb amputation, volume depletion, urosepsis, pyelonephritis, Fournier’s gangrene, genital mycotic infections, and hypoglycemia when used with insulin or insulin secretagogues.
Bexagliflozin joins an already crowded field of SGLT2 inhibitors, some of which have been approved for additional cardiovascular and kidney indications.
Of interest, bexagliflozin was approved by the FDA for diabetes in cats in December 2022, as the first oral new animal drug to improve glycemic control in otherwise healthy cats with diabetes not previously treated with insulin.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has approved bexagliflozin (Brenzavvy, TheracosBio) for the treatment of adults with type 2 diabetes.
The once-daily 20-mg oral sodium-glucose cotransporter 2 (SGLT2) inhibitor is indicated as an adjunct to diet and exercise to improve glycemic control for those with type 2 diabetes, but not type 1 diabetes. It can be used in adults with an estimated glomerular filtration rate (eGFR) > 30 mL/min per 1.73 m2.
Approval was based on results from 23 clinical trials with more than 5,000 participants, including more than 300 patients with stage 3 kidney disease (eGFR < 60 and > 30 mL/min per 1.73 m2).
In the phase 3 studies, bexagliflozin significantly reduced hemoglobin A1c and fasting blood glucose at 24 weeks as monotherapy or as add-on to metformin and other glucose-lowering drugs and combinations. It also produced modest reductions in body weight and systolic blood pressure.
In the phase 3 Bexagliflozin Efficacy and Safety Trial (BEST) cardiovascular outcomes trial, the drug met its efficacy and safety objectives in patients at high cardiovascular risk. Noninferiority was demonstrated for the composite outcome of cardiovascular death, myocardial infarction, stroke, or unstable angina.
“As a class of drugs, SGLT2 inhibitors have shown tremendous benefit in treating adults with type 2 diabetes,” said Mason Freeman, MD, director of the Translational Research Center at Massachusetts General Hospital, Boston, in a press release from TheracosBio.
“Being involved in all of the clinical trials for Brenzavvy, I am greatly impressed with the efficacy of the drug in reducing blood glucose levels and I believe it is an important addition to the SGLT2 inhibitor class of drugs.”
As with other SGLT2 inhibitors, adverse events seen in the trials include ketoacidosis, lower limb amputation, volume depletion, urosepsis, pyelonephritis, Fournier’s gangrene, genital mycotic infections, and hypoglycemia when used with insulin or insulin secretagogues.
Bexagliflozin joins an already crowded field of SGLT2 inhibitors, some of which have been approved for additional cardiovascular and kidney indications.
Of interest, bexagliflozin was approved by the FDA for diabetes in cats in December 2022, as the first oral new animal drug to improve glycemic control in otherwise healthy cats with diabetes not previously treated with insulin.
A version of this article first appeared on Medscape.com.
Adding venetoclax improves ibrutinib outcomes in CLL
Investigators led by Philip Thompson, MD, a hematologist/oncologist at the center, explained that CLL patients receiving ibrutinib, a Bruton’s kinase inhibitor, “rarely achieve complete remission with undetectable measurable residual disease,” so they stay on the costly treatment indefinitely or until disease progression or accumulating adverse events force a switch to venetoclax.
Using the two agents together, instead of consecutively, may allow strong responders to stop treatment altogether and suboptimal responders to have longer remissions, they said.
“We would not advocate prolonged Bruton’s kinase inhibitor use prior to starting venetoclax in treatment-naive patients, as the safety and efficacy of commencing venetoclax after a 3-month ibrutinib monotherapy phase has been repeatedly demonstrated,” the team said.
However, the investigators noted that their “study was not intended to directly answer the question of whether combination therapy is superior to the current paradigm of sequential monotherapy.” Randomized trials are looking into the matter. The study was published recently as a preprint on ResearchSquare.com and has not been peer reviewed.
Complete remission in over half
The 45 adult subjects had one or more high-risk features for CLL progression and had received at least 1 year of ibrutinib at 140-420 mg once daily, depending on tolerance. They had bone marrow detectable disease at study entry but did not meet criteria for progression. Median duration of ibrutinib at baseline was 32 months, and about half the subjects were on it as their initial therapy.
Venetoclax, a BCL2 inhibitor with a completely different mechanisms of action, was added to ibrutinib for up to 2 years, escalated up to a target dose of 400 mg once daily.
On intention-to-treat analysis, venetoclax add-on improved ibrutinib response to complete remission in 55% of patients; complete remission was defined as less than 1 CLL cell per 10,000 leukocytes in bone marrow on two consecutive occasions 6 months apart.
The rate of undetectable bone marrow disease was 57% after 1 year of combined treatment and 71% after venetoclax completion, at which point 23 patients with undetectable disease stopped ibrutinib along with venetoclax.
Five patients had disease progression at a median of 41 months after venetoclax initiation, one during combined therapy, three during ibrutinib maintenance afterward, and one with Richter transformation after complete remission and discontinuation of all treatment. No patient had died from CLL.
“There has so far been no significant difference noted in” time to residual disease re-emergence, the team said, based on whether or not patients continued ibrutinib after venetoclax add-on.
There was no significant difference in the rate of bone marrow clearance according to the presence or absence of TP53 abnormalities, complex karyotypes, or prior treatment status.
The most common grade 3/4 adverse event was neutropenia in 20% of patients. Nine patients developed nonmelanoma skin cancer during the trial; six were diagnosed with other solid tumors; three came down with grade 3 infections, and two developed myelodysplastic syndrome, both with a prior history of chemotherapy.
No one stopped venetoclax because of toxicity, but about a third of subjects required dose reductions, most often because of neutropenia.
The study was funded by AbbVie, which is commercializing venetoclax along with Genentech. Investigators disclosed ties to both companies and many others. Dr. Thompson disclosed ties to AbbVie, Pharmacyclics, Lilly, Adaptive Biotechnologies, Janssen, Beigene, and Genentech.
Investigators led by Philip Thompson, MD, a hematologist/oncologist at the center, explained that CLL patients receiving ibrutinib, a Bruton’s kinase inhibitor, “rarely achieve complete remission with undetectable measurable residual disease,” so they stay on the costly treatment indefinitely or until disease progression or accumulating adverse events force a switch to venetoclax.
Using the two agents together, instead of consecutively, may allow strong responders to stop treatment altogether and suboptimal responders to have longer remissions, they said.
“We would not advocate prolonged Bruton’s kinase inhibitor use prior to starting venetoclax in treatment-naive patients, as the safety and efficacy of commencing venetoclax after a 3-month ibrutinib monotherapy phase has been repeatedly demonstrated,” the team said.
However, the investigators noted that their “study was not intended to directly answer the question of whether combination therapy is superior to the current paradigm of sequential monotherapy.” Randomized trials are looking into the matter. The study was published recently as a preprint on ResearchSquare.com and has not been peer reviewed.
Complete remission in over half
The 45 adult subjects had one or more high-risk features for CLL progression and had received at least 1 year of ibrutinib at 140-420 mg once daily, depending on tolerance. They had bone marrow detectable disease at study entry but did not meet criteria for progression. Median duration of ibrutinib at baseline was 32 months, and about half the subjects were on it as their initial therapy.
Venetoclax, a BCL2 inhibitor with a completely different mechanisms of action, was added to ibrutinib for up to 2 years, escalated up to a target dose of 400 mg once daily.
On intention-to-treat analysis, venetoclax add-on improved ibrutinib response to complete remission in 55% of patients; complete remission was defined as less than 1 CLL cell per 10,000 leukocytes in bone marrow on two consecutive occasions 6 months apart.
The rate of undetectable bone marrow disease was 57% after 1 year of combined treatment and 71% after venetoclax completion, at which point 23 patients with undetectable disease stopped ibrutinib along with venetoclax.
Five patients had disease progression at a median of 41 months after venetoclax initiation, one during combined therapy, three during ibrutinib maintenance afterward, and one with Richter transformation after complete remission and discontinuation of all treatment. No patient had died from CLL.
“There has so far been no significant difference noted in” time to residual disease re-emergence, the team said, based on whether or not patients continued ibrutinib after venetoclax add-on.
There was no significant difference in the rate of bone marrow clearance according to the presence or absence of TP53 abnormalities, complex karyotypes, or prior treatment status.
The most common grade 3/4 adverse event was neutropenia in 20% of patients. Nine patients developed nonmelanoma skin cancer during the trial; six were diagnosed with other solid tumors; three came down with grade 3 infections, and two developed myelodysplastic syndrome, both with a prior history of chemotherapy.
No one stopped venetoclax because of toxicity, but about a third of subjects required dose reductions, most often because of neutropenia.
The study was funded by AbbVie, which is commercializing venetoclax along with Genentech. Investigators disclosed ties to both companies and many others. Dr. Thompson disclosed ties to AbbVie, Pharmacyclics, Lilly, Adaptive Biotechnologies, Janssen, Beigene, and Genentech.
Investigators led by Philip Thompson, MD, a hematologist/oncologist at the center, explained that CLL patients receiving ibrutinib, a Bruton’s kinase inhibitor, “rarely achieve complete remission with undetectable measurable residual disease,” so they stay on the costly treatment indefinitely or until disease progression or accumulating adverse events force a switch to venetoclax.
Using the two agents together, instead of consecutively, may allow strong responders to stop treatment altogether and suboptimal responders to have longer remissions, they said.
“We would not advocate prolonged Bruton’s kinase inhibitor use prior to starting venetoclax in treatment-naive patients, as the safety and efficacy of commencing venetoclax after a 3-month ibrutinib monotherapy phase has been repeatedly demonstrated,” the team said.
However, the investigators noted that their “study was not intended to directly answer the question of whether combination therapy is superior to the current paradigm of sequential monotherapy.” Randomized trials are looking into the matter. The study was published recently as a preprint on ResearchSquare.com and has not been peer reviewed.
Complete remission in over half
The 45 adult subjects had one or more high-risk features for CLL progression and had received at least 1 year of ibrutinib at 140-420 mg once daily, depending on tolerance. They had bone marrow detectable disease at study entry but did not meet criteria for progression. Median duration of ibrutinib at baseline was 32 months, and about half the subjects were on it as their initial therapy.
Venetoclax, a BCL2 inhibitor with a completely different mechanisms of action, was added to ibrutinib for up to 2 years, escalated up to a target dose of 400 mg once daily.
On intention-to-treat analysis, venetoclax add-on improved ibrutinib response to complete remission in 55% of patients; complete remission was defined as less than 1 CLL cell per 10,000 leukocytes in bone marrow on two consecutive occasions 6 months apart.
The rate of undetectable bone marrow disease was 57% after 1 year of combined treatment and 71% after venetoclax completion, at which point 23 patients with undetectable disease stopped ibrutinib along with venetoclax.
Five patients had disease progression at a median of 41 months after venetoclax initiation, one during combined therapy, three during ibrutinib maintenance afterward, and one with Richter transformation after complete remission and discontinuation of all treatment. No patient had died from CLL.
“There has so far been no significant difference noted in” time to residual disease re-emergence, the team said, based on whether or not patients continued ibrutinib after venetoclax add-on.
There was no significant difference in the rate of bone marrow clearance according to the presence or absence of TP53 abnormalities, complex karyotypes, or prior treatment status.
The most common grade 3/4 adverse event was neutropenia in 20% of patients. Nine patients developed nonmelanoma skin cancer during the trial; six were diagnosed with other solid tumors; three came down with grade 3 infections, and two developed myelodysplastic syndrome, both with a prior history of chemotherapy.
No one stopped venetoclax because of toxicity, but about a third of subjects required dose reductions, most often because of neutropenia.
The study was funded by AbbVie, which is commercializing venetoclax along with Genentech. Investigators disclosed ties to both companies and many others. Dr. Thompson disclosed ties to AbbVie, Pharmacyclics, Lilly, Adaptive Biotechnologies, Janssen, Beigene, and Genentech.
FROM RESEARCHSQUARE
People with cancer should be wary of taking dietary supplements
Cancer dietitian Lisa Cianciotta often finds herself sitting across from a patient who suddenly fishes a bottle of antioxidant supplements from their bag and says, “My friend told me this works really well,” or “I read on the Internet that this is supposed to be really good for cancer.”
Although taking an antioxidant pill sounds harmless, Ms. Cianciotta, a clinical dietitian who works with cancer patients at New York–Presbyterian Hospital, knows well that this popular dietary supplement can interfere with a patient’s radiation or chemotherapy.
But many patients with cancer believe these over-the-counter vitamins, minerals, or herbal remedies will help them, and most use at least one dietary supplement alongside their cancer treatment.
And that leaves Ms. Cianciotta with a delicate conversation ahead of her.
. Popular dietary supplements may, for instance, cancel the effects of a cancer treatment, making it less effective, or increase serious side effects, such as liver toxicity. But in other cases, supplementation, such as vitamin D for patients who lack the vitamin, may be beneficial, Ms. Cianciotta said.
These drug-supplement interactions can be hard to pinpoint, given that more than two-thirds of doctors don’t know their patients are using supplements.
Here’s what patients need to know about the potential risks of supplement use during treatment, and how oncologists can address this thorny, often poorly understood topic with patients.
The complex drug-supplement landscape
The list of dietary supplements and how they can interact with different treatments and cancer types is long and nuanced.
But certain supplements appear to affect cancer treatments regardless of other things and should be avoided. Any supplement that strongly alters the body’s levels of the protein cytochromes P450 is one example. This group of enzymes plays a key role in metabolizing drugs, including chemotherapy and immunotherapy agents.
Certain supplements – most notably St. John’s wort extract – may decrease or increase the activity of cytochrome P450, which can then affect the concentrations of anticancer drugs in the blood, said William Figg, PharmD, an associate director of the Center for Cancer Research at the National Cancer Institute in Bethesda, Md. Studies show, for instance, that this common herbal supplement can increase the activity of cytochrome P450, resulting in lower levels of cancer drugs.
Outside of drug metabolism, patients with hormone-related cancers, such as breast and prostate cancers, should steer clear of dietary supplements that can alter levels of testosterone or estrogen, Dr. Figg said. The evergreen shrub ashwagandha, for example, is marketed to reduce stress and fatigue, but can also increase testosterone levels – a potential problem for those with prostate cancer receiving androgen deprivation therapy, which lowers testosterone levels.
Many oncologists counsel patients against using antioxidant-based dietary supplements – particularly turmeric and green tea extract – while they have radiation therapy and certain chemotherapies. These therapies work by creating an abundance of highly reactive molecules called free radicals in tumor cells, which increase stress within these cells, ultimately killing them off. Antioxidants, in theory, can neutralize this effect, said Skyler Johnson, MD, a radiation oncologist at Huntsman Cancer Institute at the University of Utah, Salt Lake City. Some studies suggest that antioxidant supplements may lessen the effects of radiation and chemotherapy, although the evidence is mixed.
Some dietary supplements, including high-dose green tea extract and vitamin A, can cause kidney or liver toxicity, and “many cancer patients already have compromised kidney or liver function,” said Jun J. Mao, MD, chief of integrative medicine at Memorial Sloan Kettering Cancer Center in New York. Even herbs that don’t interfere with how well a cancer drug works, such as stevia, can increase treatment-related side effects, such as nausea and vomiting.
Another potential problem with dietary supplements: It’s nearly impossible to know exactly what’s in them. For instance, just last year, the Food and Drug Administration sent nearly 50 warning letters to companies marketing dietary supplements. The issue is that federal regulations governing production are less strict for supplements than for medications. As a result, some supplements contain ingredients not listed on the label.
One historical example was the supplement PC-SPES, a mix of eight herbs, marketed to men with prostate cancer. The supplement was recalled in 2002 after certain batches were found to contain traces of prescription drugs, including diethylstilbestrol, ethinyl estradiol, warfarin, and alprazolam.
To further complicate matters, some dietary supplements can be helpful. Most patients with cancer “are malnourished and missing out on nutrients they could be getting from food,” said Ms. Cianciotta.
Patients are tested routinely for vitamin deficiencies and receive supplements as needed, she said. Vitamin D and folic acid are two of the most common deficiencies in this patient population. Vitamin D supplementation can improve outcomes in patients who have received a stem cell transplant by aiding engraftment and rebuilding the immune system, while folic acid supplementation can help to raise low red blood cell counts and hemoglobin levels.
Although she rarely sees vitamin toxicity, Ms. Cianciotta stressed that more is not always better and supplement use, even when it seems safe or warranted due to a deficiency, should be taken under supervision, and monitored carefully by the patient’s care team.
Bringing supplement use into the light
Too often, providers are unaware of a patient’s supplement use.
A core reason: Dietary supplements are often touted as natural, which many patients equate with safety, said Samantha Heller, a senior clinical nutritionist at New York (N.Y.) University Langone Health.
That means patients may not know a supplement can act like a drug and interfere with their cancer treatment, and thus may not see the importance of telling their doctors.
Still, the promise of herbs, vitamins, and minerals can be alluring, and there are many reasons patients decide to partake. One major appeal: Dietary supplements can help some patients feel empowered.
“Cancer is a disease that takes away a lot of control from the individual. Taking supplements or herbs is a way to regain some sense of control,” said Dr. Mao.
The phenomenon can also be cultural, he said. Some people grow up taking herbs and supplements to stay healthy or combat health woes.
Pressure or advice from family or friends who may think they are helping a loved one with their dietary recommendations may play a role as well. Friends and family “cannot prescribe chemo, but they can buy herbs and supplements,” Dr. Mao said.
Patients seeking greater control over their health or who feel high levels of anxiety may be more likely to take suggestions from friends and family or more likely to believe false or misleading claims about the efficacy or safety of supplements, explained medical oncologist William Dahut, MD, chief scientific officer for the American Cancer Society.
Plus, social media often amplifies and normalizes this misinformation, noted Dr. Johnson. In a 2021 study published in the Journal of the National Cancer Institute, he and colleagues found that one-third of the most popular articles on cancer treatment posted to social media in 2018 and 2019 contained false, inaccurate, or misleading information that was often harmful.
Some of the false claims centered on unproven, potentially unsafe herbal remedies, according to Dr. Johnson. These included “lung cancer can be cured with cannabis oil” and “golden berries cure and prevent cancer.”
Given exaggerated claims of “cures,” some patients may keep their supplement use under the radar out of fear they will be judged or criticized.
“Clinicians should avoid making patients feel judged or telling people not to go online to do their own research,” Dr. Johnson said.
Guiding patients, instead, to accurate sources of online information may be one way to help patients feel empowered, he said. Cancer.gov and the Memorial Sloan Kettering Cancer Center’s About Herbs database provide accessible and accurate information on dietary supplements and cancer treatment for both health care professionals and patients, he noted.
If a particular supplement is not safe during treatment, providers should be able to explain why, said Ms. Cianciotta. In a recent study, 80% of health care providers surveyed believed that interactions between herbals and medications could be problematic, but only 15% could explain why.
“Being able to explain why we are discouraging a particular supplement right now tends to be much better received than just telling a patient not to take something, because it is bad,” she said.
Another key is listening closely to patients to understand why they may be taking a particular supplement. Does the patient feel out of control? Is nausea a problem?
“Allowing patients to tell you why they are using a particular supplement will often reveal unmet needs or psychosocial challenges,” Dr. Mao said. This information can allow providers to suggest an evidence-based alternative, such as mindfulness meditation or acupuncture, to manage stress.
And if a patient has received a dietary supplement from well-meaning family and friends?
“Simply telling a patient that a given supplement is useless or harmful could create family tension,” said Dr. Mao.
Instead, he recommends reframing the issue.
“We want to have a better understanding of how patients are tolerating chemo or immunotherapy before throwing other things on top of it. Let them know that now may just not be the right time to add a supplement to the mix,” Dr. Mao said.
The bottom line: “Patients want to play an active role in their own care, and we want to help them do that in a safe way,” he said.
A version of this article first appeared on WebMD.com.
Cancer dietitian Lisa Cianciotta often finds herself sitting across from a patient who suddenly fishes a bottle of antioxidant supplements from their bag and says, “My friend told me this works really well,” or “I read on the Internet that this is supposed to be really good for cancer.”
Although taking an antioxidant pill sounds harmless, Ms. Cianciotta, a clinical dietitian who works with cancer patients at New York–Presbyterian Hospital, knows well that this popular dietary supplement can interfere with a patient’s radiation or chemotherapy.
But many patients with cancer believe these over-the-counter vitamins, minerals, or herbal remedies will help them, and most use at least one dietary supplement alongside their cancer treatment.
And that leaves Ms. Cianciotta with a delicate conversation ahead of her.
. Popular dietary supplements may, for instance, cancel the effects of a cancer treatment, making it less effective, or increase serious side effects, such as liver toxicity. But in other cases, supplementation, such as vitamin D for patients who lack the vitamin, may be beneficial, Ms. Cianciotta said.
These drug-supplement interactions can be hard to pinpoint, given that more than two-thirds of doctors don’t know their patients are using supplements.
Here’s what patients need to know about the potential risks of supplement use during treatment, and how oncologists can address this thorny, often poorly understood topic with patients.
The complex drug-supplement landscape
The list of dietary supplements and how they can interact with different treatments and cancer types is long and nuanced.
But certain supplements appear to affect cancer treatments regardless of other things and should be avoided. Any supplement that strongly alters the body’s levels of the protein cytochromes P450 is one example. This group of enzymes plays a key role in metabolizing drugs, including chemotherapy and immunotherapy agents.
Certain supplements – most notably St. John’s wort extract – may decrease or increase the activity of cytochrome P450, which can then affect the concentrations of anticancer drugs in the blood, said William Figg, PharmD, an associate director of the Center for Cancer Research at the National Cancer Institute in Bethesda, Md. Studies show, for instance, that this common herbal supplement can increase the activity of cytochrome P450, resulting in lower levels of cancer drugs.
Outside of drug metabolism, patients with hormone-related cancers, such as breast and prostate cancers, should steer clear of dietary supplements that can alter levels of testosterone or estrogen, Dr. Figg said. The evergreen shrub ashwagandha, for example, is marketed to reduce stress and fatigue, but can also increase testosterone levels – a potential problem for those with prostate cancer receiving androgen deprivation therapy, which lowers testosterone levels.
Many oncologists counsel patients against using antioxidant-based dietary supplements – particularly turmeric and green tea extract – while they have radiation therapy and certain chemotherapies. These therapies work by creating an abundance of highly reactive molecules called free radicals in tumor cells, which increase stress within these cells, ultimately killing them off. Antioxidants, in theory, can neutralize this effect, said Skyler Johnson, MD, a radiation oncologist at Huntsman Cancer Institute at the University of Utah, Salt Lake City. Some studies suggest that antioxidant supplements may lessen the effects of radiation and chemotherapy, although the evidence is mixed.
Some dietary supplements, including high-dose green tea extract and vitamin A, can cause kidney or liver toxicity, and “many cancer patients already have compromised kidney or liver function,” said Jun J. Mao, MD, chief of integrative medicine at Memorial Sloan Kettering Cancer Center in New York. Even herbs that don’t interfere with how well a cancer drug works, such as stevia, can increase treatment-related side effects, such as nausea and vomiting.
Another potential problem with dietary supplements: It’s nearly impossible to know exactly what’s in them. For instance, just last year, the Food and Drug Administration sent nearly 50 warning letters to companies marketing dietary supplements. The issue is that federal regulations governing production are less strict for supplements than for medications. As a result, some supplements contain ingredients not listed on the label.
One historical example was the supplement PC-SPES, a mix of eight herbs, marketed to men with prostate cancer. The supplement was recalled in 2002 after certain batches were found to contain traces of prescription drugs, including diethylstilbestrol, ethinyl estradiol, warfarin, and alprazolam.
To further complicate matters, some dietary supplements can be helpful. Most patients with cancer “are malnourished and missing out on nutrients they could be getting from food,” said Ms. Cianciotta.
Patients are tested routinely for vitamin deficiencies and receive supplements as needed, she said. Vitamin D and folic acid are two of the most common deficiencies in this patient population. Vitamin D supplementation can improve outcomes in patients who have received a stem cell transplant by aiding engraftment and rebuilding the immune system, while folic acid supplementation can help to raise low red blood cell counts and hemoglobin levels.
Although she rarely sees vitamin toxicity, Ms. Cianciotta stressed that more is not always better and supplement use, even when it seems safe or warranted due to a deficiency, should be taken under supervision, and monitored carefully by the patient’s care team.
Bringing supplement use into the light
Too often, providers are unaware of a patient’s supplement use.
A core reason: Dietary supplements are often touted as natural, which many patients equate with safety, said Samantha Heller, a senior clinical nutritionist at New York (N.Y.) University Langone Health.
That means patients may not know a supplement can act like a drug and interfere with their cancer treatment, and thus may not see the importance of telling their doctors.
Still, the promise of herbs, vitamins, and minerals can be alluring, and there are many reasons patients decide to partake. One major appeal: Dietary supplements can help some patients feel empowered.
“Cancer is a disease that takes away a lot of control from the individual. Taking supplements or herbs is a way to regain some sense of control,” said Dr. Mao.
The phenomenon can also be cultural, he said. Some people grow up taking herbs and supplements to stay healthy or combat health woes.
Pressure or advice from family or friends who may think they are helping a loved one with their dietary recommendations may play a role as well. Friends and family “cannot prescribe chemo, but they can buy herbs and supplements,” Dr. Mao said.
Patients seeking greater control over their health or who feel high levels of anxiety may be more likely to take suggestions from friends and family or more likely to believe false or misleading claims about the efficacy or safety of supplements, explained medical oncologist William Dahut, MD, chief scientific officer for the American Cancer Society.
Plus, social media often amplifies and normalizes this misinformation, noted Dr. Johnson. In a 2021 study published in the Journal of the National Cancer Institute, he and colleagues found that one-third of the most popular articles on cancer treatment posted to social media in 2018 and 2019 contained false, inaccurate, or misleading information that was often harmful.
Some of the false claims centered on unproven, potentially unsafe herbal remedies, according to Dr. Johnson. These included “lung cancer can be cured with cannabis oil” and “golden berries cure and prevent cancer.”
Given exaggerated claims of “cures,” some patients may keep their supplement use under the radar out of fear they will be judged or criticized.
“Clinicians should avoid making patients feel judged or telling people not to go online to do their own research,” Dr. Johnson said.
Guiding patients, instead, to accurate sources of online information may be one way to help patients feel empowered, he said. Cancer.gov and the Memorial Sloan Kettering Cancer Center’s About Herbs database provide accessible and accurate information on dietary supplements and cancer treatment for both health care professionals and patients, he noted.
If a particular supplement is not safe during treatment, providers should be able to explain why, said Ms. Cianciotta. In a recent study, 80% of health care providers surveyed believed that interactions between herbals and medications could be problematic, but only 15% could explain why.
“Being able to explain why we are discouraging a particular supplement right now tends to be much better received than just telling a patient not to take something, because it is bad,” she said.
Another key is listening closely to patients to understand why they may be taking a particular supplement. Does the patient feel out of control? Is nausea a problem?
“Allowing patients to tell you why they are using a particular supplement will often reveal unmet needs or psychosocial challenges,” Dr. Mao said. This information can allow providers to suggest an evidence-based alternative, such as mindfulness meditation or acupuncture, to manage stress.
And if a patient has received a dietary supplement from well-meaning family and friends?
“Simply telling a patient that a given supplement is useless or harmful could create family tension,” said Dr. Mao.
Instead, he recommends reframing the issue.
“We want to have a better understanding of how patients are tolerating chemo or immunotherapy before throwing other things on top of it. Let them know that now may just not be the right time to add a supplement to the mix,” Dr. Mao said.
The bottom line: “Patients want to play an active role in their own care, and we want to help them do that in a safe way,” he said.
A version of this article first appeared on WebMD.com.
Cancer dietitian Lisa Cianciotta often finds herself sitting across from a patient who suddenly fishes a bottle of antioxidant supplements from their bag and says, “My friend told me this works really well,” or “I read on the Internet that this is supposed to be really good for cancer.”
Although taking an antioxidant pill sounds harmless, Ms. Cianciotta, a clinical dietitian who works with cancer patients at New York–Presbyterian Hospital, knows well that this popular dietary supplement can interfere with a patient’s radiation or chemotherapy.
But many patients with cancer believe these over-the-counter vitamins, minerals, or herbal remedies will help them, and most use at least one dietary supplement alongside their cancer treatment.
And that leaves Ms. Cianciotta with a delicate conversation ahead of her.
. Popular dietary supplements may, for instance, cancel the effects of a cancer treatment, making it less effective, or increase serious side effects, such as liver toxicity. But in other cases, supplementation, such as vitamin D for patients who lack the vitamin, may be beneficial, Ms. Cianciotta said.
These drug-supplement interactions can be hard to pinpoint, given that more than two-thirds of doctors don’t know their patients are using supplements.
Here’s what patients need to know about the potential risks of supplement use during treatment, and how oncologists can address this thorny, often poorly understood topic with patients.
The complex drug-supplement landscape
The list of dietary supplements and how they can interact with different treatments and cancer types is long and nuanced.
But certain supplements appear to affect cancer treatments regardless of other things and should be avoided. Any supplement that strongly alters the body’s levels of the protein cytochromes P450 is one example. This group of enzymes plays a key role in metabolizing drugs, including chemotherapy and immunotherapy agents.
Certain supplements – most notably St. John’s wort extract – may decrease or increase the activity of cytochrome P450, which can then affect the concentrations of anticancer drugs in the blood, said William Figg, PharmD, an associate director of the Center for Cancer Research at the National Cancer Institute in Bethesda, Md. Studies show, for instance, that this common herbal supplement can increase the activity of cytochrome P450, resulting in lower levels of cancer drugs.
Outside of drug metabolism, patients with hormone-related cancers, such as breast and prostate cancers, should steer clear of dietary supplements that can alter levels of testosterone or estrogen, Dr. Figg said. The evergreen shrub ashwagandha, for example, is marketed to reduce stress and fatigue, but can also increase testosterone levels – a potential problem for those with prostate cancer receiving androgen deprivation therapy, which lowers testosterone levels.
Many oncologists counsel patients against using antioxidant-based dietary supplements – particularly turmeric and green tea extract – while they have radiation therapy and certain chemotherapies. These therapies work by creating an abundance of highly reactive molecules called free radicals in tumor cells, which increase stress within these cells, ultimately killing them off. Antioxidants, in theory, can neutralize this effect, said Skyler Johnson, MD, a radiation oncologist at Huntsman Cancer Institute at the University of Utah, Salt Lake City. Some studies suggest that antioxidant supplements may lessen the effects of radiation and chemotherapy, although the evidence is mixed.
Some dietary supplements, including high-dose green tea extract and vitamin A, can cause kidney or liver toxicity, and “many cancer patients already have compromised kidney or liver function,” said Jun J. Mao, MD, chief of integrative medicine at Memorial Sloan Kettering Cancer Center in New York. Even herbs that don’t interfere with how well a cancer drug works, such as stevia, can increase treatment-related side effects, such as nausea and vomiting.
Another potential problem with dietary supplements: It’s nearly impossible to know exactly what’s in them. For instance, just last year, the Food and Drug Administration sent nearly 50 warning letters to companies marketing dietary supplements. The issue is that federal regulations governing production are less strict for supplements than for medications. As a result, some supplements contain ingredients not listed on the label.
One historical example was the supplement PC-SPES, a mix of eight herbs, marketed to men with prostate cancer. The supplement was recalled in 2002 after certain batches were found to contain traces of prescription drugs, including diethylstilbestrol, ethinyl estradiol, warfarin, and alprazolam.
To further complicate matters, some dietary supplements can be helpful. Most patients with cancer “are malnourished and missing out on nutrients they could be getting from food,” said Ms. Cianciotta.
Patients are tested routinely for vitamin deficiencies and receive supplements as needed, she said. Vitamin D and folic acid are two of the most common deficiencies in this patient population. Vitamin D supplementation can improve outcomes in patients who have received a stem cell transplant by aiding engraftment and rebuilding the immune system, while folic acid supplementation can help to raise low red blood cell counts and hemoglobin levels.
Although she rarely sees vitamin toxicity, Ms. Cianciotta stressed that more is not always better and supplement use, even when it seems safe or warranted due to a deficiency, should be taken under supervision, and monitored carefully by the patient’s care team.
Bringing supplement use into the light
Too often, providers are unaware of a patient’s supplement use.
A core reason: Dietary supplements are often touted as natural, which many patients equate with safety, said Samantha Heller, a senior clinical nutritionist at New York (N.Y.) University Langone Health.
That means patients may not know a supplement can act like a drug and interfere with their cancer treatment, and thus may not see the importance of telling their doctors.
Still, the promise of herbs, vitamins, and minerals can be alluring, and there are many reasons patients decide to partake. One major appeal: Dietary supplements can help some patients feel empowered.
“Cancer is a disease that takes away a lot of control from the individual. Taking supplements or herbs is a way to regain some sense of control,” said Dr. Mao.
The phenomenon can also be cultural, he said. Some people grow up taking herbs and supplements to stay healthy or combat health woes.
Pressure or advice from family or friends who may think they are helping a loved one with their dietary recommendations may play a role as well. Friends and family “cannot prescribe chemo, but they can buy herbs and supplements,” Dr. Mao said.
Patients seeking greater control over their health or who feel high levels of anxiety may be more likely to take suggestions from friends and family or more likely to believe false or misleading claims about the efficacy or safety of supplements, explained medical oncologist William Dahut, MD, chief scientific officer for the American Cancer Society.
Plus, social media often amplifies and normalizes this misinformation, noted Dr. Johnson. In a 2021 study published in the Journal of the National Cancer Institute, he and colleagues found that one-third of the most popular articles on cancer treatment posted to social media in 2018 and 2019 contained false, inaccurate, or misleading information that was often harmful.
Some of the false claims centered on unproven, potentially unsafe herbal remedies, according to Dr. Johnson. These included “lung cancer can be cured with cannabis oil” and “golden berries cure and prevent cancer.”
Given exaggerated claims of “cures,” some patients may keep their supplement use under the radar out of fear they will be judged or criticized.
“Clinicians should avoid making patients feel judged or telling people not to go online to do their own research,” Dr. Johnson said.
Guiding patients, instead, to accurate sources of online information may be one way to help patients feel empowered, he said. Cancer.gov and the Memorial Sloan Kettering Cancer Center’s About Herbs database provide accessible and accurate information on dietary supplements and cancer treatment for both health care professionals and patients, he noted.
If a particular supplement is not safe during treatment, providers should be able to explain why, said Ms. Cianciotta. In a recent study, 80% of health care providers surveyed believed that interactions between herbals and medications could be problematic, but only 15% could explain why.
“Being able to explain why we are discouraging a particular supplement right now tends to be much better received than just telling a patient not to take something, because it is bad,” she said.
Another key is listening closely to patients to understand why they may be taking a particular supplement. Does the patient feel out of control? Is nausea a problem?
“Allowing patients to tell you why they are using a particular supplement will often reveal unmet needs or psychosocial challenges,” Dr. Mao said. This information can allow providers to suggest an evidence-based alternative, such as mindfulness meditation or acupuncture, to manage stress.
And if a patient has received a dietary supplement from well-meaning family and friends?
“Simply telling a patient that a given supplement is useless or harmful could create family tension,” said Dr. Mao.
Instead, he recommends reframing the issue.
“We want to have a better understanding of how patients are tolerating chemo or immunotherapy before throwing other things on top of it. Let them know that now may just not be the right time to add a supplement to the mix,” Dr. Mao said.
The bottom line: “Patients want to play an active role in their own care, and we want to help them do that in a safe way,” he said.
A version of this article first appeared on WebMD.com.
Tucatinib plus trastuzumab approved for HER2+ colorectal cancer
The U.S. Food and Drug Administration has granted accelerated approval to tucatinib (Tukysa) in combination with trastuzumab for use in RAS wild-type, HER2-positive unresectable or metastatic colorectal cancer that has progressed after fluoropyrimidine, oxaliplatin, and irinotecan-based chemotherapy.
This is the first FDA-approved treatment for HER2-positive metastatic colorectal cancer, maker Seagen said in a Jan. 19 press release.
“Historically, patients with HER2-positive metastatic colorectal cancer who have progressed following frontline therapy have had poor outcomes. The FDA approval of a chemotherapy-free combination regimen that specifically targets HER2 is great news for these patients,” John Strickler, MD, associate professor of medicine at Duke University Medical Center, Durham, N.C., said in the press release.
Dr. Strickler was the lead investigator on the approval trial, dubbed MOUNTAINEER, which involved 84 patients who met the treatment criteria and who had also been treated with an anti-VEGF antibody. Participants whose tumors were deficient in mismatch repair proteins or were microsatellite instability–high must also have received a PD-1 inhibitor. Patients who received prior anti-HER2 therapy were excluded, the FDA explained in its own press release.
Participants were treated with tucatinib 300 mg orally twice daily– the recommended dose in product labeling – with trastuzumab administered at a loading dose of 8 mg/kg intravenously on day 1 of cycle 1 followed by a maintenance dose of trastuzumab 6 mg/kg on day 1 of each subsequent 21-day cycle.
Overall response rate was 38%, and median duration of response was 12.4 months.
The most common adverse events, occurring in at least 20% of study participants, were diarrhea, fatigue, rash, nausea, abdominal pain, infusion related reactions, and pyrexia. The most common laboratory abnormalities were increased creatinine, decreased lymphocytes, increased alanine aminotransferase, and decreased hemoglobin, among others.
Serious adverse reactions occurred in 22% of patients. The most common (occurring in ≥ 2% of patients) were intestinal obstruction (7%); urinary tract infection (3.5%); and pneumonia, abdominal pain, and rectal perforation (2.3% each). Adverse reactions leading to permanent discontinuation occurred in 6% of patients, including increased alanine aminotransferase in 2.3%.
Continued approval for the indication may be contingent upon verification and description of clinical benefit in confirmatory trials, the company said.
A global, randomized phase 3 clinical trial (MOUNTAINEER-03) is ongoing and is comparing tucatinib in combination with trastuzumab and mFOLFOX6 with standard of care and is intended to serve as a confirmatory trial, the company said.
Tucatinib is already approved in combination with trastuzumab and capecitabine for use in the treatment of advanced unresectable or metastatic HER2-positive breast cancer.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted accelerated approval to tucatinib (Tukysa) in combination with trastuzumab for use in RAS wild-type, HER2-positive unresectable or metastatic colorectal cancer that has progressed after fluoropyrimidine, oxaliplatin, and irinotecan-based chemotherapy.
This is the first FDA-approved treatment for HER2-positive metastatic colorectal cancer, maker Seagen said in a Jan. 19 press release.
“Historically, patients with HER2-positive metastatic colorectal cancer who have progressed following frontline therapy have had poor outcomes. The FDA approval of a chemotherapy-free combination regimen that specifically targets HER2 is great news for these patients,” John Strickler, MD, associate professor of medicine at Duke University Medical Center, Durham, N.C., said in the press release.
Dr. Strickler was the lead investigator on the approval trial, dubbed MOUNTAINEER, which involved 84 patients who met the treatment criteria and who had also been treated with an anti-VEGF antibody. Participants whose tumors were deficient in mismatch repair proteins or were microsatellite instability–high must also have received a PD-1 inhibitor. Patients who received prior anti-HER2 therapy were excluded, the FDA explained in its own press release.
Participants were treated with tucatinib 300 mg orally twice daily– the recommended dose in product labeling – with trastuzumab administered at a loading dose of 8 mg/kg intravenously on day 1 of cycle 1 followed by a maintenance dose of trastuzumab 6 mg/kg on day 1 of each subsequent 21-day cycle.
Overall response rate was 38%, and median duration of response was 12.4 months.
The most common adverse events, occurring in at least 20% of study participants, were diarrhea, fatigue, rash, nausea, abdominal pain, infusion related reactions, and pyrexia. The most common laboratory abnormalities were increased creatinine, decreased lymphocytes, increased alanine aminotransferase, and decreased hemoglobin, among others.
Serious adverse reactions occurred in 22% of patients. The most common (occurring in ≥ 2% of patients) were intestinal obstruction (7%); urinary tract infection (3.5%); and pneumonia, abdominal pain, and rectal perforation (2.3% each). Adverse reactions leading to permanent discontinuation occurred in 6% of patients, including increased alanine aminotransferase in 2.3%.
Continued approval for the indication may be contingent upon verification and description of clinical benefit in confirmatory trials, the company said.
A global, randomized phase 3 clinical trial (MOUNTAINEER-03) is ongoing and is comparing tucatinib in combination with trastuzumab and mFOLFOX6 with standard of care and is intended to serve as a confirmatory trial, the company said.
Tucatinib is already approved in combination with trastuzumab and capecitabine for use in the treatment of advanced unresectable or metastatic HER2-positive breast cancer.
A version of this article first appeared on Medscape.com.
The U.S. Food and Drug Administration has granted accelerated approval to tucatinib (Tukysa) in combination with trastuzumab for use in RAS wild-type, HER2-positive unresectable or metastatic colorectal cancer that has progressed after fluoropyrimidine, oxaliplatin, and irinotecan-based chemotherapy.
This is the first FDA-approved treatment for HER2-positive metastatic colorectal cancer, maker Seagen said in a Jan. 19 press release.
“Historically, patients with HER2-positive metastatic colorectal cancer who have progressed following frontline therapy have had poor outcomes. The FDA approval of a chemotherapy-free combination regimen that specifically targets HER2 is great news for these patients,” John Strickler, MD, associate professor of medicine at Duke University Medical Center, Durham, N.C., said in the press release.
Dr. Strickler was the lead investigator on the approval trial, dubbed MOUNTAINEER, which involved 84 patients who met the treatment criteria and who had also been treated with an anti-VEGF antibody. Participants whose tumors were deficient in mismatch repair proteins or were microsatellite instability–high must also have received a PD-1 inhibitor. Patients who received prior anti-HER2 therapy were excluded, the FDA explained in its own press release.
Participants were treated with tucatinib 300 mg orally twice daily– the recommended dose in product labeling – with trastuzumab administered at a loading dose of 8 mg/kg intravenously on day 1 of cycle 1 followed by a maintenance dose of trastuzumab 6 mg/kg on day 1 of each subsequent 21-day cycle.
Overall response rate was 38%, and median duration of response was 12.4 months.
The most common adverse events, occurring in at least 20% of study participants, were diarrhea, fatigue, rash, nausea, abdominal pain, infusion related reactions, and pyrexia. The most common laboratory abnormalities were increased creatinine, decreased lymphocytes, increased alanine aminotransferase, and decreased hemoglobin, among others.
Serious adverse reactions occurred in 22% of patients. The most common (occurring in ≥ 2% of patients) were intestinal obstruction (7%); urinary tract infection (3.5%); and pneumonia, abdominal pain, and rectal perforation (2.3% each). Adverse reactions leading to permanent discontinuation occurred in 6% of patients, including increased alanine aminotransferase in 2.3%.
Continued approval for the indication may be contingent upon verification and description of clinical benefit in confirmatory trials, the company said.
A global, randomized phase 3 clinical trial (MOUNTAINEER-03) is ongoing and is comparing tucatinib in combination with trastuzumab and mFOLFOX6 with standard of care and is intended to serve as a confirmatory trial, the company said.
Tucatinib is already approved in combination with trastuzumab and capecitabine for use in the treatment of advanced unresectable or metastatic HER2-positive breast cancer.
A version of this article first appeared on Medscape.com.
Highly anticipated HIV vaccine fails in large trial
officials announced Wednesday.
The vaccine had been in development since 2019 and was given to 3,900 study participants through October 2022, but data shows it does not protect against HIV compared with a placebo, according to developer Janssen Pharmaceutical.
Experts estimate the failure means there won’t be another potential vaccine on the horizon for 3 to 5 years, the New York Times reported.
“It’s obviously disappointing,” Anthony Fauci, MD, former head of the National Institute of Allergy and Infectious Diseases, told MSNBC, noting that other areas of HIV treatment research are promising. “I don’t think that people should give up on the field of the HIV vaccine.”
No safety issues had been identified with the vaccine during the trial, which studied the experimental treatment in men who have sex with men or with transgender people.
There is no cure for HIV, but disease progression can be managed with existing treatments. HIV attacks the body’s immune system and destroys white blood cells, increasing the risk of other infections. More than 1.5 million people worldwide were infected with HIV in 2021 and 38.4 million people are living with the virus, according to UNAIDS.
A version of this article first appeared on WebMD.com.
officials announced Wednesday.
The vaccine had been in development since 2019 and was given to 3,900 study participants through October 2022, but data shows it does not protect against HIV compared with a placebo, according to developer Janssen Pharmaceutical.
Experts estimate the failure means there won’t be another potential vaccine on the horizon for 3 to 5 years, the New York Times reported.
“It’s obviously disappointing,” Anthony Fauci, MD, former head of the National Institute of Allergy and Infectious Diseases, told MSNBC, noting that other areas of HIV treatment research are promising. “I don’t think that people should give up on the field of the HIV vaccine.”
No safety issues had been identified with the vaccine during the trial, which studied the experimental treatment in men who have sex with men or with transgender people.
There is no cure for HIV, but disease progression can be managed with existing treatments. HIV attacks the body’s immune system and destroys white blood cells, increasing the risk of other infections. More than 1.5 million people worldwide were infected with HIV in 2021 and 38.4 million people are living with the virus, according to UNAIDS.
A version of this article first appeared on WebMD.com.
officials announced Wednesday.
The vaccine had been in development since 2019 and was given to 3,900 study participants through October 2022, but data shows it does not protect against HIV compared with a placebo, according to developer Janssen Pharmaceutical.
Experts estimate the failure means there won’t be another potential vaccine on the horizon for 3 to 5 years, the New York Times reported.
“It’s obviously disappointing,” Anthony Fauci, MD, former head of the National Institute of Allergy and Infectious Diseases, told MSNBC, noting that other areas of HIV treatment research are promising. “I don’t think that people should give up on the field of the HIV vaccine.”
No safety issues had been identified with the vaccine during the trial, which studied the experimental treatment in men who have sex with men or with transgender people.
There is no cure for HIV, but disease progression can be managed with existing treatments. HIV attacks the body’s immune system and destroys white blood cells, increasing the risk of other infections. More than 1.5 million people worldwide were infected with HIV in 2021 and 38.4 million people are living with the virus, according to UNAIDS.
A version of this article first appeared on WebMD.com.
Best estimates made for hydroxychloroquine retinopathy risk
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
A new study likely makes the best estimate yet of the degree of retinopathy risk that patients who take the antimalarial drug hydroxychloroquine (HCQ) can expect, deriving mainly from the cumulative dose taken during the first 5 years of use, according to a study published in Annals of Internal Medicine.
HCQ works to decrease activity in a patient’s immune system, which is effective in many cases of systemic lupus erythematosus, one of the most common indications for the drug. However, an adverse outcome of treatment can be HCQ retinopathy, a progressive form of vision loss in patients taking HCQ over an extended period (mostly for longer than 5 years). The disease is often asymptomatic, although some patients do present a paracentral scotoma and a decrease in color vision. Patients may also notice flashing shapes in their vision and find that they have difficulty reading. Eventually, HCQ retinopathy can lead to loss of visual acuity, loss of peripheral vision, and loss of night vision.
Researchers from Kaiser Permanente Northern California and Harvard Medical School analyzed 3,325 persons who received HCQ for 5 or more years between 2004 and 2020. Their goal was to both characterize the long-term risk for incident HCQ retinopathy and examine the degree to which average HCQ dose within the first 5 years of treatment serves as a prediction of the risk.
The researchers then estimated the risk for developing retinopathy after 15 years, according to patients’ average dosing levels during the first 5 years of therapy. Overall, 81 participants developed HCQ retinopathy with overall cumulative incidences of 2.5% after 10 years and 8.6% after 15 years; the risk was greater for those given a higher dose during the first 5 years of treatment.
The mechanism of how HCQ toxicity may occur is still not completely known. There is evidence that toxicity happens because HCQ binds to melanin in both the retinal pigment epithelium and uvea in high concentrations. HCQ can interfere with lysosomal function, leading to oxidation and accumulation of lysosomes, which can cause dysfunction of the retinal pigment epithelium.
Progressive retinopathy can continue even after the drug is stopped. “It’s thought to be a very mild but important risk,” said Nilanjana Bose, MD, MBA, a rheumatologist with Memorial Hermann Health System in Houston. “Patients taking HCQ must be screened for retinal issues, most certainly elderly patients and patients with any kind of comorbidities.”
A 2021 joint position statement from the American College of Rheumatology, American Academy of Dermatology, the Rheumatologic Dermatology Society, and the American Academy of Ophthalmology recommends a baseline eye exam within a few months after starting therapy, then additional screening at 5 years on HCQ and annually thereafter.
“Early detection of retinopathy is important in overall visual prognosis, because toxicity can continue even after discontinuation of the medication,” said Rukhsana G. Mirza, MD, professor of ophthalmology and medical education at Northwestern University in Chicago.
“Examination alone is not sufficient to evaluate early changes, and specialized testing must be done. These include color photos, visual field tests, optical coherence tomography, fundus autofluorescence and in some cases, multifocal electroretinogram. Also, the AAO [American Academy of Ophthalmology] has specific recommendations related to Asian patients as they may have a different pattern of retinopathy that must also be considered.”
More accurate risk measurements
This news organization asked study coauthor April Jorge, MD, assistant professor of medicine in the division of rheumatology, allergy, and immunology at Massachusetts General Hospital and Harvard Medical School, Boston, to discuss the study, how it correlates to past research, and what it adds that’s new and useful to rheumatologists and ophthalmologists:
Question: Your research found that a higher dose of HCQ in the first 5 years of treatment led to a greater risk of retinopathy. Is there any indication that a lower dose given more frequently, either within that 5-year period or longer, would pose a similar risk?
Answer: In our study, we assessed the HCQ dose in the first 5 years of use but followed patients who continued the medication longer than 5 years, through up to 15 years of use. Therefore, we compared the risk of HCQ retinopathy associated with different HCQ dosages but for the same duration of use. We found that for any dose of HCQ, the risk of retinopathy increases the longer the medication is used. However, patients who used a higher dose of HCQ had a higher risk of developing retinopathy over time.
Although current guidelines recommend avoiding any HCQ dose over 5 mg/kg per day to reduce the risk of retinopathy, we found a higher risk of retinopathy associated with dosing over 6 mg/kg per day than between 5 and 6 mg/kg per day and the lowest risk with dosing under 5 mg/kg per day.
Q: How does your study align with and/or expand upon previous research regarding HCQ risk?
A: An important prior study of hydroxychloroquine retinopathy was the 2014 study by Ronald B. Melles, MD, and Michael F. Marmor, MD, published in JAMA Ophthalmology. Prior to our present study, that was the largest study to use the modern screening method (optical coherence tomography) to detect HCQ retinopathy. That screening tool is more sensitive than older methods, so it can detect early/mild cases of retinopathy that are typically asymptomatic. Compared to older studies, that 2014 study found a much higher risk of HCQ retinopathy than was previously appreciated.
However, that 2014 study did have some key limitations that could affect the risk estimates, such as using prevalent cases. A key feature of our present study is that we took several important steps to generate more accurate risk estimates. This included using an incident user cohort and detecting incident retinopathy cases through serial review of optical coherence tomography (screening) studies.
To achieve a high degree of methodologic rigor in correctly identifying retinopathy outcomes, we had expert ophthalmologists perform masked adjudication of all screening studies, and we assessed the intra-rater reliability of these study interpretations. Therefore, our study adds to the literature more accurate estimates of retinopathy risk. We found a lower cumulative incidence of retinopathy than was identified in the 2014 study, but the risk is still noteworthy.
Also unique to our study, we graded the severity of HCQ retinopathy outcomes. This was important, as we found that the majority of retinopathy cases detected through routine screening are mild and presumed to be asymptomatic. This will likely be reassuring news for patients that we can screen for this adverse event to detect it early and prevent vision loss.
Another important difference was that we assessed the risk of retinopathy associated with using over 6 mg/kg per day, between 5 and 6 mg/kg per day, and less than 5 mg/kg per day, whereas the highest dosing group assessed in the 2014 study included all patients using over 5 mg/kg per day. The risk was considerably higher in the > 6 mg/kg per day group than in the 5-6 mg/kg per day group.
Q: How can rheumatologists and ophthalmologists use this new information specifically to better treat their patients?
A: Our study provides more accurate estimates of the risk of HCQ retinopathy than in prior studies. These risk estimates can be used when rheumatologists (and other clinicians who prescribe HCQ) consider the risks and benefits of this otherwise important and well-tolerated medication. The risk associated with different dose ranges could also inform dosing decisions, since dosing over 6 mg/kg per day may be more of a concern than using doses in the 5-6 mg/kg range. Ophthalmologists can also use these new risk estimates to counsel patients of the importance of HCQ retinopathy screening and can also hopefully provide some reassurance to patients that the risk of severe retinopathy is low as long as they are being monitored.
The study authors were supported by grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and the Rheumatology Research Foundation. The authors report no relevant financial relationships. Dr. Bose and Dr. Mirza had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE