User login
Liberalized European sports cardiology guidelines break new ground
New guidelines on sports cardiology from the European Society of Cardiology break fresh ground by green-lighting participation in vigorous competitive sports by selected patients with stable coronary artery disease, heart failure, or mild arrhythmias.
These liberalized guidelines, released at the virtual annual congress of the European Society of Cardiology, thus move well beyond the standard exercise advice to engage in about 150 minutes per week of moderate physical activity, typically defined as brisk walking or its equivalent.
The guidelines reflect a conviction that exercise is powerful medicine for patients with cardiovascular disease and also affords a means to help curb the epidemics of diabetes and obesity that drive cardiovascular risk, according to Antonio Pelliccia, MD, who cochaired the 24-member task force of European and American experts that developed the guidelines.
In a session highlighting the new sports cardiology guidelines, Mats Borjesson, MD, head of the Center for Health and Performance at Gothenburg (Sweden) University, summarized the section devoted to patients with stable coronary artery disease: “If you have established CAD and a low risk of adverse events during exercise, you are eligible for high-intensity exercise and competitive sports. But if you have persistent ischemia despite medical treatment, or symptoms, then you’re only eligible for leisure-time subthreshold activity.”
Dr. Pelliccia put this new recommendation into context.
“We are not talking anymore in this particular disease just about cardiac rehabilitation or leisure-time activity, but we are also opening the border and talking about competitive sports activity in selected patients where you have the evidence for low risk of exercise-induced adverse events. This is a major achievement now for what is the major disease in our adult population,” said Dr. Pelliccia, chief of cardiology at the Institute of Sports Medicine and Science at the Italian National Olympic Committee and professor of sports cardiology at La Sapienza University of Rome.
The recommendation for individualized consideration of all types of exercise, even including vigorous competitive sports, in low-risk patients with CAD gets a class IIa, level of evidence (LOE) C recommendation in the new guidelines. That’s a big step down from a ringing class Ia endorsement, but since sports cardiology is a relatively young field with little evidence that’s based on randomized trials, the guidelines are rife with many other class IIa, LOE C recommendations as well.
“The level of evidence is rather low, so these guidelines are very much the personal perspective of the expert panel,” explained Martin Halle, MD, professor and head of the department of prevention, rehabilitation, and sports cardiology at Technical University of Munich.
The high-risk features for exercise-induced cardiac adverse events in patients with longstanding stable CAD, as cited in the guidelines, include a critical coronary stenosis, defined as a more than 70% lesion in a major coronary artery or a greater than 50% stenosis in the left main, and/or a fractional flow reserve score of less than 0.8; a left ventricular ejection fraction of 50% or less with wall-motion abnormalities; inducible myocardial ischemia on maximal exercise testing; nonsustained ventricular tachycardia; polymorphic or very frequent ventricular premature beats at rest and during maximum stress; and a recent acute coronary syndrome (ACS). These features call for an exercise prescription tailored to remain below the patient’s angina and ischemia thresholds.
“It’s important for cardiologists out there to understand that we definitely need a maximal exercise test. In somebody who is running and has an ACS and then wants to start running again, 200 watts on an ergometer is too low. We have to push them up to the end, and then if everything is okay – left ventricular function is okay, no ischemia, no arrhythmias under exercise testing – then it’s fine,” Dr. Halle said.
Dr. Pelliccia added that close follow-up is needed, because this is an evolving disease.”
Exercise and heart failure
Massimo F. Piepoli, MD, PhD, noted that the guidelines give a class IIb, LOE C recommendation for consideration of high-intensity recreational endurance and power sports in patients with heart failure with either midrange or preserved ejection fraction, provided they are stable, asymptomatic, on optimal guideline-directed medical therapy, and without abnormalities on a maximal exercise stress test.
However, such intense physical activity is not recommended in patients with heart failure with reduced ejection fraction, regardless of their symptom status, added Dr. Piepoli of Guglielmo da Saliceto Hospital in Placenza, Italy.
“We’re talking here, I think for the first time, about possible competitive sports participation in individuals with heart failure, depending on their clinical condition. We are really opening the barriers to sports participation, even in these patients in whom we never thought of it before,” Dr. Pelliccia observed.
Valvular heart disease and exercise
Guidelines panelist Sabiha Gati, MRCP, PhD, said asymptomatic individuals with mild valvular abnormalities can participate in all recreational and competitive sports; that’s a class I, LOE C recommendation.
“Moderate regurgitant lesions are better tolerated than stenotic lesions, and those with preserved systolic function, good functional capacity, without any exercise-induced arrhythmias or ischemia or abnormal hemodynamic response are considered to be low risk and can participate in all sports,” added Dr. Gati, a cardiologist at Royal Brompton Hospital, London.
The two most common valvular abnormalities encountered in clinical practice are bicuspid aortic valve and mitral valve prolapse. Dr. Gati noted that, while mitral valve prolapse has a benign prognosis in the great majority of affected individuals, the presence of specific features indicative of increased risk for sudden cardiac death precludes participation in strenuous exercise. These include T-wave inversion in the inferior leads on a 12-lead ECG, long QT, bileaflet mitral valve prolapse, basal inferolateral wall fibrosis, severe mitral regurgitation, or a family history of sudden cardiac death.
Bicuspid aortic valve has a prevalence of 1%-2% in the general population. It can be associated with aortic stenosis, aortic regurgitation, and increased risk of ascending aortic aneurysm and dissection. Since it remains unclear whether intensive exercise accelerates aortic dilatation, a cautious approach to sports participation is recommended in patients with an ascending aorta above the normal limit of 40 mm, she said.
The 80-page ESC sports cardiology guidelines, published online simultaneously with their presentation, cover a broad range of additional topics, including exercise recommendations for the general public, for the elderly, as well as for patients with cardiomyopathies, adult congenital heart disease, arrhythmias, and channelopathies. Gaps in evidence are also highlighted.
SOURCE: Pelliccia A. ESC 2020 and Eur Heart J. 2020 Aug 29. doi: 10.1093/eurheartj/ehaa605.
New guidelines on sports cardiology from the European Society of Cardiology break fresh ground by green-lighting participation in vigorous competitive sports by selected patients with stable coronary artery disease, heart failure, or mild arrhythmias.
These liberalized guidelines, released at the virtual annual congress of the European Society of Cardiology, thus move well beyond the standard exercise advice to engage in about 150 minutes per week of moderate physical activity, typically defined as brisk walking or its equivalent.
The guidelines reflect a conviction that exercise is powerful medicine for patients with cardiovascular disease and also affords a means to help curb the epidemics of diabetes and obesity that drive cardiovascular risk, according to Antonio Pelliccia, MD, who cochaired the 24-member task force of European and American experts that developed the guidelines.
In a session highlighting the new sports cardiology guidelines, Mats Borjesson, MD, head of the Center for Health and Performance at Gothenburg (Sweden) University, summarized the section devoted to patients with stable coronary artery disease: “If you have established CAD and a low risk of adverse events during exercise, you are eligible for high-intensity exercise and competitive sports. But if you have persistent ischemia despite medical treatment, or symptoms, then you’re only eligible for leisure-time subthreshold activity.”
Dr. Pelliccia put this new recommendation into context.
“We are not talking anymore in this particular disease just about cardiac rehabilitation or leisure-time activity, but we are also opening the border and talking about competitive sports activity in selected patients where you have the evidence for low risk of exercise-induced adverse events. This is a major achievement now for what is the major disease in our adult population,” said Dr. Pelliccia, chief of cardiology at the Institute of Sports Medicine and Science at the Italian National Olympic Committee and professor of sports cardiology at La Sapienza University of Rome.
The recommendation for individualized consideration of all types of exercise, even including vigorous competitive sports, in low-risk patients with CAD gets a class IIa, level of evidence (LOE) C recommendation in the new guidelines. That’s a big step down from a ringing class Ia endorsement, but since sports cardiology is a relatively young field with little evidence that’s based on randomized trials, the guidelines are rife with many other class IIa, LOE C recommendations as well.
“The level of evidence is rather low, so these guidelines are very much the personal perspective of the expert panel,” explained Martin Halle, MD, professor and head of the department of prevention, rehabilitation, and sports cardiology at Technical University of Munich.
The high-risk features for exercise-induced cardiac adverse events in patients with longstanding stable CAD, as cited in the guidelines, include a critical coronary stenosis, defined as a more than 70% lesion in a major coronary artery or a greater than 50% stenosis in the left main, and/or a fractional flow reserve score of less than 0.8; a left ventricular ejection fraction of 50% or less with wall-motion abnormalities; inducible myocardial ischemia on maximal exercise testing; nonsustained ventricular tachycardia; polymorphic or very frequent ventricular premature beats at rest and during maximum stress; and a recent acute coronary syndrome (ACS). These features call for an exercise prescription tailored to remain below the patient’s angina and ischemia thresholds.
“It’s important for cardiologists out there to understand that we definitely need a maximal exercise test. In somebody who is running and has an ACS and then wants to start running again, 200 watts on an ergometer is too low. We have to push them up to the end, and then if everything is okay – left ventricular function is okay, no ischemia, no arrhythmias under exercise testing – then it’s fine,” Dr. Halle said.
Dr. Pelliccia added that close follow-up is needed, because this is an evolving disease.”
Exercise and heart failure
Massimo F. Piepoli, MD, PhD, noted that the guidelines give a class IIb, LOE C recommendation for consideration of high-intensity recreational endurance and power sports in patients with heart failure with either midrange or preserved ejection fraction, provided they are stable, asymptomatic, on optimal guideline-directed medical therapy, and without abnormalities on a maximal exercise stress test.
However, such intense physical activity is not recommended in patients with heart failure with reduced ejection fraction, regardless of their symptom status, added Dr. Piepoli of Guglielmo da Saliceto Hospital in Placenza, Italy.
“We’re talking here, I think for the first time, about possible competitive sports participation in individuals with heart failure, depending on their clinical condition. We are really opening the barriers to sports participation, even in these patients in whom we never thought of it before,” Dr. Pelliccia observed.
Valvular heart disease and exercise
Guidelines panelist Sabiha Gati, MRCP, PhD, said asymptomatic individuals with mild valvular abnormalities can participate in all recreational and competitive sports; that’s a class I, LOE C recommendation.
“Moderate regurgitant lesions are better tolerated than stenotic lesions, and those with preserved systolic function, good functional capacity, without any exercise-induced arrhythmias or ischemia or abnormal hemodynamic response are considered to be low risk and can participate in all sports,” added Dr. Gati, a cardiologist at Royal Brompton Hospital, London.
The two most common valvular abnormalities encountered in clinical practice are bicuspid aortic valve and mitral valve prolapse. Dr. Gati noted that, while mitral valve prolapse has a benign prognosis in the great majority of affected individuals, the presence of specific features indicative of increased risk for sudden cardiac death precludes participation in strenuous exercise. These include T-wave inversion in the inferior leads on a 12-lead ECG, long QT, bileaflet mitral valve prolapse, basal inferolateral wall fibrosis, severe mitral regurgitation, or a family history of sudden cardiac death.
Bicuspid aortic valve has a prevalence of 1%-2% in the general population. It can be associated with aortic stenosis, aortic regurgitation, and increased risk of ascending aortic aneurysm and dissection. Since it remains unclear whether intensive exercise accelerates aortic dilatation, a cautious approach to sports participation is recommended in patients with an ascending aorta above the normal limit of 40 mm, she said.
The 80-page ESC sports cardiology guidelines, published online simultaneously with their presentation, cover a broad range of additional topics, including exercise recommendations for the general public, for the elderly, as well as for patients with cardiomyopathies, adult congenital heart disease, arrhythmias, and channelopathies. Gaps in evidence are also highlighted.
SOURCE: Pelliccia A. ESC 2020 and Eur Heart J. 2020 Aug 29. doi: 10.1093/eurheartj/ehaa605.
New guidelines on sports cardiology from the European Society of Cardiology break fresh ground by green-lighting participation in vigorous competitive sports by selected patients with stable coronary artery disease, heart failure, or mild arrhythmias.
These liberalized guidelines, released at the virtual annual congress of the European Society of Cardiology, thus move well beyond the standard exercise advice to engage in about 150 minutes per week of moderate physical activity, typically defined as brisk walking or its equivalent.
The guidelines reflect a conviction that exercise is powerful medicine for patients with cardiovascular disease and also affords a means to help curb the epidemics of diabetes and obesity that drive cardiovascular risk, according to Antonio Pelliccia, MD, who cochaired the 24-member task force of European and American experts that developed the guidelines.
In a session highlighting the new sports cardiology guidelines, Mats Borjesson, MD, head of the Center for Health and Performance at Gothenburg (Sweden) University, summarized the section devoted to patients with stable coronary artery disease: “If you have established CAD and a low risk of adverse events during exercise, you are eligible for high-intensity exercise and competitive sports. But if you have persistent ischemia despite medical treatment, or symptoms, then you’re only eligible for leisure-time subthreshold activity.”
Dr. Pelliccia put this new recommendation into context.
“We are not talking anymore in this particular disease just about cardiac rehabilitation or leisure-time activity, but we are also opening the border and talking about competitive sports activity in selected patients where you have the evidence for low risk of exercise-induced adverse events. This is a major achievement now for what is the major disease in our adult population,” said Dr. Pelliccia, chief of cardiology at the Institute of Sports Medicine and Science at the Italian National Olympic Committee and professor of sports cardiology at La Sapienza University of Rome.
The recommendation for individualized consideration of all types of exercise, even including vigorous competitive sports, in low-risk patients with CAD gets a class IIa, level of evidence (LOE) C recommendation in the new guidelines. That’s a big step down from a ringing class Ia endorsement, but since sports cardiology is a relatively young field with little evidence that’s based on randomized trials, the guidelines are rife with many other class IIa, LOE C recommendations as well.
“The level of evidence is rather low, so these guidelines are very much the personal perspective of the expert panel,” explained Martin Halle, MD, professor and head of the department of prevention, rehabilitation, and sports cardiology at Technical University of Munich.
The high-risk features for exercise-induced cardiac adverse events in patients with longstanding stable CAD, as cited in the guidelines, include a critical coronary stenosis, defined as a more than 70% lesion in a major coronary artery or a greater than 50% stenosis in the left main, and/or a fractional flow reserve score of less than 0.8; a left ventricular ejection fraction of 50% or less with wall-motion abnormalities; inducible myocardial ischemia on maximal exercise testing; nonsustained ventricular tachycardia; polymorphic or very frequent ventricular premature beats at rest and during maximum stress; and a recent acute coronary syndrome (ACS). These features call for an exercise prescription tailored to remain below the patient’s angina and ischemia thresholds.
“It’s important for cardiologists out there to understand that we definitely need a maximal exercise test. In somebody who is running and has an ACS and then wants to start running again, 200 watts on an ergometer is too low. We have to push them up to the end, and then if everything is okay – left ventricular function is okay, no ischemia, no arrhythmias under exercise testing – then it’s fine,” Dr. Halle said.
Dr. Pelliccia added that close follow-up is needed, because this is an evolving disease.”
Exercise and heart failure
Massimo F. Piepoli, MD, PhD, noted that the guidelines give a class IIb, LOE C recommendation for consideration of high-intensity recreational endurance and power sports in patients with heart failure with either midrange or preserved ejection fraction, provided they are stable, asymptomatic, on optimal guideline-directed medical therapy, and without abnormalities on a maximal exercise stress test.
However, such intense physical activity is not recommended in patients with heart failure with reduced ejection fraction, regardless of their symptom status, added Dr. Piepoli of Guglielmo da Saliceto Hospital in Placenza, Italy.
“We’re talking here, I think for the first time, about possible competitive sports participation in individuals with heart failure, depending on their clinical condition. We are really opening the barriers to sports participation, even in these patients in whom we never thought of it before,” Dr. Pelliccia observed.
Valvular heart disease and exercise
Guidelines panelist Sabiha Gati, MRCP, PhD, said asymptomatic individuals with mild valvular abnormalities can participate in all recreational and competitive sports; that’s a class I, LOE C recommendation.
“Moderate regurgitant lesions are better tolerated than stenotic lesions, and those with preserved systolic function, good functional capacity, without any exercise-induced arrhythmias or ischemia or abnormal hemodynamic response are considered to be low risk and can participate in all sports,” added Dr. Gati, a cardiologist at Royal Brompton Hospital, London.
The two most common valvular abnormalities encountered in clinical practice are bicuspid aortic valve and mitral valve prolapse. Dr. Gati noted that, while mitral valve prolapse has a benign prognosis in the great majority of affected individuals, the presence of specific features indicative of increased risk for sudden cardiac death precludes participation in strenuous exercise. These include T-wave inversion in the inferior leads on a 12-lead ECG, long QT, bileaflet mitral valve prolapse, basal inferolateral wall fibrosis, severe mitral regurgitation, or a family history of sudden cardiac death.
Bicuspid aortic valve has a prevalence of 1%-2% in the general population. It can be associated with aortic stenosis, aortic regurgitation, and increased risk of ascending aortic aneurysm and dissection. Since it remains unclear whether intensive exercise accelerates aortic dilatation, a cautious approach to sports participation is recommended in patients with an ascending aorta above the normal limit of 40 mm, she said.
The 80-page ESC sports cardiology guidelines, published online simultaneously with their presentation, cover a broad range of additional topics, including exercise recommendations for the general public, for the elderly, as well as for patients with cardiomyopathies, adult congenital heart disease, arrhythmias, and channelopathies. Gaps in evidence are also highlighted.
SOURCE: Pelliccia A. ESC 2020 and Eur Heart J. 2020 Aug 29. doi: 10.1093/eurheartj/ehaa605.
FROM ESC CONGRESS 2020
Biologics for psoriasis may also reduce coronary plaque
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
Biologics used as treatment for psoriasis may also help reduce lipid-rich necrotic core (LRNC), a high-risk plaque associated with cardiovascular events, recent research from a prospective, observational study suggests.
Cardiac CT scans performed on patients with psoriasis 1 year after starting biologic therapy revealed a reduction in LRNC, compared with patients who were not receiving biologics, according to Harry Choi, MD, of the National Heart, Lung, and Blood Institute at the National Institutes of Health and colleagues. The association with reduction in LRNC and biologic therapy remained significant when adjusted for type of biologic. “These findings demonstrate that LRNC may be modulated by the control of systemic inflammation,” the researchers wrote in their study, published Sept. 15 in Circulation: Cardiovascular Imaging.
Dr. Choi and colleagues evaluated 289 patients with psoriasis within the Psoriasis Atherosclerosis and Cardiometabolic Disease Initiative cohort. The patients had a mean age of 50 years and a mean body mass index of 29.4 kg/m2, as well as a mean Psoriasis Area and Severity Index (PASI) score of 6.0. At baseline, 29% of patients had hypertension, 41% had hyperlipidemia, their mean Framingham risk score was 1.9, and a three-quarters (212 of 289) had mild to moderate psoriasis.
Changes in LRNC were observed at 1 year, compared with baseline prior to and after receiving biologic therapy (124 patients) in comparison with patients who did not undergo biologic therapy (85 patients). Biologic therapies were grouped by type, which included anti–tumor necrosis factor (anti-TNF), anti–interleukin (IL)–12/23, and anti–IL-17 biologics.
There were a significant associations between LRNC and Framingham risk score (standardized beta coefficient, 0.12; 95% confidence interval, 0.00-0.15; P = .045) and severity of psoriasis (beta, 0.13; 95% CI, 0.01-0.26; P = .029) at baseline.
Key findings
The researchers found a significant reduction in LRNC 1 year after patients began biologic therapy (median, 2.97 mm2; interquartile range, 1.99-4.66), compared with baseline (median, 3.12 mm2; IQR, 1.84-4.35) (P = .028), while patients who did not receive biologic therapy had nonsignificantly higher LRNC after 1 year (median, 3.12 mm2; IQR, 1.82-4.60), compared with baseline measurements (median, 3.34 mm2; IQR, 2.04–4.74) (P = .06).
The results remained significant after the researchers adjusted for psoriasis severity, Framingham risk score, BMI, use of statins (beta, −0.09; 95% CI, −0.01 to −0.18; P = .033). Significant reductions in LRNC also remained when analyzing patients receiving anti-TNF, anti–IL-12/23, and anti–IL-17 biologics independently, and there were no significant between-group differences in reduction of LRNC.
The potential of biologics for improving vascular health
Discussing the study results in a press release from the American Heart Association, senior author Nehal N. Mehta, MD, MSCE, FAHA, chief of the Lab of Inflammation and Cardiometabolic Diseases at the NHLBI at NIH, compared the effect biologic therapy had on coronary plaque reduction with that of statins.
“There is approximately 6%-8% reduction in coronary plaque following therapy with statins. Similarly, our treatment with biologic therapy reduced coronary plaque by the same amount after one year. These findings suggest that biologic therapy to treat psoriasis may be just as beneficial as statin therapy on heart arteries,” Dr. Mehta said in the release.
In an interview, Nieca Goldberg, MD, medical director of NYU Women’s Heart Program at NYU Langone Health, echoed Dr. Mehta’s commments and said psoriasis carries the “potential to treat two conditions with the same drug.”
“We know conditions such as psoriatic arthritis and rheumatoid arthritis cause chronic inflammation. Chronic inflammation causes injury to blood vessels and high-risk coronary plaque. Individuals with these inflammatory conditions are at high risk for heart attack,” she said. “This study shows that biologic treatment for psoriatic arthritis can reduce the presence of high-risk plaque. It shows the potential to treat chronic inflammation and high-risk coronary plaque.”
While the results show an association between use of biologics and LRNC reduction, the study design was observational and patients had a short follow-up period. Dr. Goldberg noted more studies are needed to evaluate the effect of biologics on reducing cardiovascular events such as a myocardial infarction.
“We have never before been able to show healing of an inflamed plaque like this in humans. Biologic therapy reduces systemic inflammation and immune activation, and it has a favorable impact on improving overall vascular health,” Dr. Mehta said in the press release. “Imagine if we can treat both psoriasis and coronary heart disease with one therapy – that is the question to be asked in future studies.”
This study was funded with support from the NHLBI Intramural Research Program and the NIH Medical Research Scholars Program at the National Institutes of Health. One investigator reports financial relationships with numerous pharmaceutical companies. The other authors report no relevant conflicts of interest. Dr. Mehta also reports numerous such relationships. Dr. Goldberg reports no relevant conflicts of interest.
SOURCE: Choi H et al. Circ Cardiovasc Imaging. 2020 Sep;13(9):e011199.
FROM CIRCULATION: CARDIOVASCULAR IMAGING
Many providers don’t follow hypertension guidelines
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Many health care professionals are not following current, evidence-based guidelines to screen for and diagnose hypertension, and appear to have substantial gaps in knowledge, beliefs, and use of recommended practices, results from a large survey suggest.
“One surprising finding was that there was so much trust in the stethoscope, because the automated monitors are a better way to take blood pressure,” lead author Beverly Green, MD, of Kaiser Permanente Washington Health Research Institute, Seattle, said in an interview.
The results of the survey were presented Sept. 10 at the virtual joint scientific sessions of the American Heart Association Council on Hypertension, AHA Council on Kidney in Cardiovascular Disease, and American Society of Hypertension.
The U.S. Preventive Services Task Force (USPSTF) and the American Heart Association/American College of Cardiology recommend out-of-office blood pressure measurements – via ambulatory blood pressure monitoring (ABPM) or home BP monitoring – before making a new diagnosis of hypertension.
To gauge provider knowledge, beliefs, and practices related to BP diagnostic tests, the researchers surveyed 282 providers: 102 medical assistants (MA), 28 licensed practical nurses (LPNs), 33 registered nurses (RNs), 86 primary care physicians, and 33 advanced practitioners (APs).
More than three-quarters of providers (79%) felt that BP measured manually with a stethoscope and ABPM were “very or highly” accurate ways to measure BP when making a new diagnosis of hypertension.
Most did not think that automated clinic BPs, home BP, or kiosk BP measurements were very or highly accurate.
Nearly all providers surveyed (96%) reported that they “always or almost always” rely on clinic BP measurements when diagnosing hypertension, but the majority of physicians/APs would prefer using ABPM (61%) if available.
The problem with ABPM, said Dr. Green, is “it’s just not very available or convenient for patients, and a lot of providers think that patients won’t tolerate it.” Yet, without it, there is a risk for misclassification, she said.
Karen A. Griffin, MD, who chairs the AHA Council on Hypertension, said it became “customary to use clinic BP since ABPM was not previously reimbursed for the routine diagnosis of hypertension.
“Now that the payment for ABPM has been expanded, the number of machines at most institutions is not adequate for the need. Consequently, it will take some time to catch up with the current guidelines for diagnosing hypertension,” she said in an interview.
The provider survey by Dr. Green and colleagues also shows slow uptake of updated thresholds for high blood pressure.
Eighty-four percent of physicians/APs and 68% of MA/LPN/RNs said they used a clinic BP threshold of at least 140/90 mm Hg for making a new diagnosis of hypertension.
Only 3.5% and 9.0%, respectively, reported using the updated threshold of at least 130/80 mm Hg put forth in 2017.
Dr. Griffin said part of this stems from the fact that the survey began before the updated guidelines were released in 2017, “not to mention the fact that some societies have opposed the new threshold of 130/80 mm Hg.”
“I think, with time, the data on morbidity and mortality associated with the goal of 130/80 mm Hg will hopefully convince those who have not yet implemented these new guidelines that it is a safe and effective BP goal,” Dr. Griffin said.
This research had no specific funding. Dr. Green and Dr. Griffin have no relevant disclosures.
A version of this article originally appeared on Medscape.com.
Diffuse idiopathic skeletal hyperostosis heart risk higher than expected
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
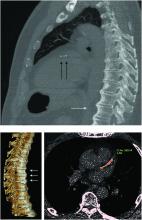
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
More people with diffuse idiopathic skeletal hyperostosis (DISH) develop cardiovascular disease (CVD) than is predicted by the Framingham Risk Score, results of an observational study have shown.
Notably, a higher rate of myocardial infarction (MI) was seen in those with DISH than in those without DISH over the 10-year follow-up period (24.4% vs. 4.3%; P = .0055).
“We propose more scrutiny is warranted in evaluating CV risk in these patients, more demanding treatment target goals should be established, and as a result, earlier and more aggressive preventive medical interventions instituted,” corresponding author Reuven Mader, MD, and associates wrote in Arthritis Research & Therapy.
“What Mader’s study is pointing out is that it’s worth the radiologist reporting [DISH],” Elizabeth A. Regan, MD, PhD, from the National Jewish Health Center in Denver, said in an interview.
DISH on a chest x-ray or CT scan should be another “red flag to be even more attentive to cardiovascular risk,” she added, particularly because studies have shown that people with DISH tend to be obese, have metabolic syndrome, or diabetes – all of which independently increase their risk for cardiovascular disease.
An old condition often found by accident
Physicians have known about DISH for many years, Dr. Mader of Ha’Emek Medical Center in Afula, Israel, observed in an interview. Historical evidence suggests it was present more than a thousand years ago, but it wasn’t until the 1950s that it gained scientific interest. Originally coined Forestier’s disease, it was renamed DISH in the late 1960s following the realization that it was not limited to the spine.
“It is a condition which is characterized by new bone formation,” Dr. Mader explained. This new bone formation has some predilection for the entheses – the tendons, ligaments, or joint capsules, that attach to the bone.
“Diagnosis of the disease is based mainly on radiographs, especially of the thoracic spine, and it requires the formation of bridges that connect at least four contiguous vertebra,” he continued.
“The bridges are usually right-sided and usually the intervertebral spaces are spared. Classically there is no involvement of the sacroiliac joints, although there are some changes that might involve the sacroiliac joints but in a different manner than in inflammatory sacroiliitis.”
DISH was originally thought to be a pain syndrome, which has “not played out,” Dr. Regan noted in her interview. While there may be people who experience pain as a result of DISH, most cases are asymptomatic and usually picked up incidentally on a chest x-ray or CT scan.
“It’s something that’s not obvious,” she said. One of the main problems it can cause is stiffness and lack of mobility in the spine and this can lead to quite severe fractures in some cases, such as during a car accident. Hence spinal surgeons and other orthopedic specialists, such as Dr. Regan, have also taken an interest in the condition.
“Apart from the thoracic spine, DISH may also involve the cervical spine; there have been many reports about difficulty in swallowing, breathing, and in the lumbar spine, spinal stenosis and so forth,” Dr. Mader said. The differential diagnosis includes ankylosing spondylitis, although there is some evidence that the two can coexist.
“The diagnosis depends on the alertness of the examining physician,” he added, noting that rheumatologists and other specialists would be “very aware of this condition” and “sensitive to changes that we see when we examine these patients.”
DISH and heightened cardiovascular risk
Previous work by Dr. Mader and associates has shown that people with DISH are more often affected by the metabolic syndrome than are those without DISH. The cross-sectional study had excluded those with preexisting CVD and found that people with DISH had a significantly higher Framingham Risk Score, compared with a control group of people with osteoarthritis and no DISH (P = .004), which in turn meant they had a significantly (P = .007) higher 10-year risk for developing CVD.
The aim of their most recent study was to compare the actual rate of CV events in 2016 versus those predicted by the Framingham Risk Score in 2006. To do this, they compared the available electronic medical records of 45 individuals with DISH and 47 without it.
The results showed that almost 39% of people with DISH had developed CVD, whereas the Framingham Risk Score had estimated that just under 27% would develop CVD.
For every 1% increase in the CVD risk calculated by the Framingham Risk Score, the odds of CVD increased by 4% in the DISH group versus the control group (P = .02).
While there was a significant (P < .003) difference in the Framingham Risk Score between the DISH and control groups in 2006 (28.6% vs. 17.8%), there was no overall statistical difference (P = .2) in the composite CVD outcome (38.8% vs. 25.5%) 10 years later, as calculated by the revised Framingham Risk Score, which included MI, cerebrovascular accident, transient ischemic attack, peripheral artery disease, and heart failure with preserved ejection fraction.
“We are dealing with patients who are in their 70s. So, it is expected that this group of patients will be more often affected by cardiovascular disease” than younger individuals, Dr. Mader observed. That said, the study’s findings “confirm the theory that patients with DISH have a high likelihood of developing cardiovascular disease,” he added, acknowledging that it was only the risk for MI that was statistically significantly higher in people with DISH than in the controls.
DISH and coronary artery calcification
“It might be even more interesting to have a different control population that had no osteoarthritis,” Dr. Regan observed.
As the associate director of the COPDGene study, Dr. Regan has access to data collected from a large cohort of people with chronic obstructive pulmonary disease (COPD; n = 2,728), around 13% of whom were identified as having DISH in one recent study.
In that study, the presence of DISH versus no DISH was associated with a 37% higher risk for having coronary artery calcification (CAC) – a marker for atherosclerosis and cardiovascular disease. Two-thirds of people with DISH had CAC, compared with 46.9% of those without DISH (P < .001). The prevalence of DISH was 8.8% in those without CAC, 12.8% in those with a CAC score of 1-100, 20% in those with a CAC score of 100-400, and 24.7% in those with a CAC score of more than 400, which is associated with a very high risk for coronary artery disease.
Dr. Regan observed that information on heart attacks and strokes were collected within the COPDGene study, so it would be possible to look at cardiovascular risk in their patients with DISH and confirm the findings of Mader and colleagues.
“I think the most important thing is recognizing that there are things going on in the spine that are important to people’s general health,” Dr. Regan said.
Dr. Mader noted: “It makes sense that patients with DISH should be more meticulously followed for at least the traditional risk factors and better treated because they are at a higher risk for these events.”
The study received no financial support. Neither Dr. Mader nor Dr. Regan had any conflicts of interest to disclose.
SOURCE: Glick K et al. Arthritis Res Ther. 2020. doi: 10.1186/s13075-020-02278-w.
FROM ARTHRITIS RESEARCH & THERAPY
Novel calculator predicts cancer risk in patients with CVD
Individualized 10-year and lifetime risks of cancer can now for the first time be estimated in patients with established cardiovascular disease, Cilie C. van ’t Klooster, MD, reported at the virtual annual congress of the European Society of Cardiology.
She and her coinvestigators have developed an easy-to-use predictive model that generates individualized risk estimates for total cancer, lung cancer, and colorectal cancer. The tool relies on nine readily available clinical variables: age, sex, smoking, weight, height, alcohol use, diabetes, antiplatelet drug use, and C-reactive protein level. The cancer risk calculator factors in an individual’s competing risk of death because of cardiovascular disease (CVD).
The risk calculator was developed using data on 7,280 patients with established CVD enrolled in the ongoing long-term Dutch UCC-SMART (Utrecht Cardiovascular Cohort – Second Manifestations of Arterial Disease) study, then independently validated in 9,322 patients in the double-blind CANTOS (Canakinumab Anti-Inflammatory Thrombosis Outcomes) trial, explained Dr. van ’t Klooster of Utrecht (the Netherlands) University.
Several other prediction models estimate the risk of a specific type of cancer, most commonly breast cancer or lung cancer. But the new Utrecht prediction tool is the first one to estimate total cancer risk. It’s also the first to apply specifically to patients with known CVD, thus filling an unmet need, because patients with established CVD are known to be on average at 19% increased risk of total cancer and 56% greater risk for lung cancer, compared with the general population. This is thought to be caused mainly by shared risk factors, including smoking, obesity, and low-grade systemic inflammation.
As the Utrecht/CANTOS analysis shows, however, that 19% increased relative risk for cancer in patients with CVD doesn’t tell the whole story. While the median lifetime and 10-year risks of total cancer in CANTOS were 26% and 10%, respectively, the individual patient risks for total cancer estimated using the Dutch prediction model ranged from 1% to 52% for lifetime and from 1% to 31% for 10-year risk. The same was true for lung cancer risk: median 5% lifetime and 2% 10-year risks, with individual patient risks ranging from 0% to 37% and from 0% to 24%. Likewise for colorectal cancer: a median 4% lifetime risk, ranging from 0% to 6%, and a median 2% risk over the next 10 years, with personalized risks ranging as high as 13% for lifetime risk and 6% for 10-year colorectal cancer risk.
The risk calculator performed “reasonably well,” according to Dr. van ’t Klooster. She pointed to a C-statistic of 0.74 for lung cancer, 0.63 for total cancer, and 0.64 for colorectal cancer. It’s possible the risk predictor’s performance could be further enhanced by incorporation of several potentially important factors that weren’t available in the UCC-SMART derivation cohort, including race, education level, and socioeconomic status, she added.
Potential applications for the risk calculator in clinical practice require further study, but include using the lifetime risk prediction for cancer as a motivational aid in conversations with patients about the importance of behavioral change in support of a healthier lifestyle. Also, a high predicted 10-year lung cancer risk could potentially be used to lower the threshold for a screening chest CT, resulting in earlier detection and treatment of lung cancer, Dr. van ’t Klooster noted.
In an interview, Bonnie Ky, MD, MSCE, praised the risk prediction study as rigorously executed, topical, and clinically significant.
“This paper signifies the overlap between our two disciplines of cancer and cardiovascular disease in terms of the risks that we face together when we care for this patient population,” said Dr. Ky, a cardiologist at the University of Pennsylvania, Philadelphia.
“Many of us in medicine believe in the importance of risk prediction: identifying who’s at high risk and doing everything we can to mitigate that risk. This paper speaks to that and moves us one step closer to accomplishing that aim,” added Dr. Ky, who is editor in chief of JACC: CardioOncology, which published the study simultaneously with Dr. van ’t Klooster’s presentation at ESC 2020. The paper provides direct access to the risk calculator.
Dr. van ’t Klooster reported having no financial conflicts regarding her study. UCC-SMART is funded by a Utrecht University grant, and CANTOS was funded by Novartis.
SOURCE: van ’t Klooster CC. ESC 2020 and JACC CardioOncol. 2020 Aug. doi: 10.1016/j.jaccao.2020.07.001.
Individualized 10-year and lifetime risks of cancer can now for the first time be estimated in patients with established cardiovascular disease, Cilie C. van ’t Klooster, MD, reported at the virtual annual congress of the European Society of Cardiology.
She and her coinvestigators have developed an easy-to-use predictive model that generates individualized risk estimates for total cancer, lung cancer, and colorectal cancer. The tool relies on nine readily available clinical variables: age, sex, smoking, weight, height, alcohol use, diabetes, antiplatelet drug use, and C-reactive protein level. The cancer risk calculator factors in an individual’s competing risk of death because of cardiovascular disease (CVD).
The risk calculator was developed using data on 7,280 patients with established CVD enrolled in the ongoing long-term Dutch UCC-SMART (Utrecht Cardiovascular Cohort – Second Manifestations of Arterial Disease) study, then independently validated in 9,322 patients in the double-blind CANTOS (Canakinumab Anti-Inflammatory Thrombosis Outcomes) trial, explained Dr. van ’t Klooster of Utrecht (the Netherlands) University.
Several other prediction models estimate the risk of a specific type of cancer, most commonly breast cancer or lung cancer. But the new Utrecht prediction tool is the first one to estimate total cancer risk. It’s also the first to apply specifically to patients with known CVD, thus filling an unmet need, because patients with established CVD are known to be on average at 19% increased risk of total cancer and 56% greater risk for lung cancer, compared with the general population. This is thought to be caused mainly by shared risk factors, including smoking, obesity, and low-grade systemic inflammation.
As the Utrecht/CANTOS analysis shows, however, that 19% increased relative risk for cancer in patients with CVD doesn’t tell the whole story. While the median lifetime and 10-year risks of total cancer in CANTOS were 26% and 10%, respectively, the individual patient risks for total cancer estimated using the Dutch prediction model ranged from 1% to 52% for lifetime and from 1% to 31% for 10-year risk. The same was true for lung cancer risk: median 5% lifetime and 2% 10-year risks, with individual patient risks ranging from 0% to 37% and from 0% to 24%. Likewise for colorectal cancer: a median 4% lifetime risk, ranging from 0% to 6%, and a median 2% risk over the next 10 years, with personalized risks ranging as high as 13% for lifetime risk and 6% for 10-year colorectal cancer risk.
The risk calculator performed “reasonably well,” according to Dr. van ’t Klooster. She pointed to a C-statistic of 0.74 for lung cancer, 0.63 for total cancer, and 0.64 for colorectal cancer. It’s possible the risk predictor’s performance could be further enhanced by incorporation of several potentially important factors that weren’t available in the UCC-SMART derivation cohort, including race, education level, and socioeconomic status, she added.
Potential applications for the risk calculator in clinical practice require further study, but include using the lifetime risk prediction for cancer as a motivational aid in conversations with patients about the importance of behavioral change in support of a healthier lifestyle. Also, a high predicted 10-year lung cancer risk could potentially be used to lower the threshold for a screening chest CT, resulting in earlier detection and treatment of lung cancer, Dr. van ’t Klooster noted.
In an interview, Bonnie Ky, MD, MSCE, praised the risk prediction study as rigorously executed, topical, and clinically significant.
“This paper signifies the overlap between our two disciplines of cancer and cardiovascular disease in terms of the risks that we face together when we care for this patient population,” said Dr. Ky, a cardiologist at the University of Pennsylvania, Philadelphia.
“Many of us in medicine believe in the importance of risk prediction: identifying who’s at high risk and doing everything we can to mitigate that risk. This paper speaks to that and moves us one step closer to accomplishing that aim,” added Dr. Ky, who is editor in chief of JACC: CardioOncology, which published the study simultaneously with Dr. van ’t Klooster’s presentation at ESC 2020. The paper provides direct access to the risk calculator.
Dr. van ’t Klooster reported having no financial conflicts regarding her study. UCC-SMART is funded by a Utrecht University grant, and CANTOS was funded by Novartis.
SOURCE: van ’t Klooster CC. ESC 2020 and JACC CardioOncol. 2020 Aug. doi: 10.1016/j.jaccao.2020.07.001.
Individualized 10-year and lifetime risks of cancer can now for the first time be estimated in patients with established cardiovascular disease, Cilie C. van ’t Klooster, MD, reported at the virtual annual congress of the European Society of Cardiology.
She and her coinvestigators have developed an easy-to-use predictive model that generates individualized risk estimates for total cancer, lung cancer, and colorectal cancer. The tool relies on nine readily available clinical variables: age, sex, smoking, weight, height, alcohol use, diabetes, antiplatelet drug use, and C-reactive protein level. The cancer risk calculator factors in an individual’s competing risk of death because of cardiovascular disease (CVD).
The risk calculator was developed using data on 7,280 patients with established CVD enrolled in the ongoing long-term Dutch UCC-SMART (Utrecht Cardiovascular Cohort – Second Manifestations of Arterial Disease) study, then independently validated in 9,322 patients in the double-blind CANTOS (Canakinumab Anti-Inflammatory Thrombosis Outcomes) trial, explained Dr. van ’t Klooster of Utrecht (the Netherlands) University.
Several other prediction models estimate the risk of a specific type of cancer, most commonly breast cancer or lung cancer. But the new Utrecht prediction tool is the first one to estimate total cancer risk. It’s also the first to apply specifically to patients with known CVD, thus filling an unmet need, because patients with established CVD are known to be on average at 19% increased risk of total cancer and 56% greater risk for lung cancer, compared with the general population. This is thought to be caused mainly by shared risk factors, including smoking, obesity, and low-grade systemic inflammation.
As the Utrecht/CANTOS analysis shows, however, that 19% increased relative risk for cancer in patients with CVD doesn’t tell the whole story. While the median lifetime and 10-year risks of total cancer in CANTOS were 26% and 10%, respectively, the individual patient risks for total cancer estimated using the Dutch prediction model ranged from 1% to 52% for lifetime and from 1% to 31% for 10-year risk. The same was true for lung cancer risk: median 5% lifetime and 2% 10-year risks, with individual patient risks ranging from 0% to 37% and from 0% to 24%. Likewise for colorectal cancer: a median 4% lifetime risk, ranging from 0% to 6%, and a median 2% risk over the next 10 years, with personalized risks ranging as high as 13% for lifetime risk and 6% for 10-year colorectal cancer risk.
The risk calculator performed “reasonably well,” according to Dr. van ’t Klooster. She pointed to a C-statistic of 0.74 for lung cancer, 0.63 for total cancer, and 0.64 for colorectal cancer. It’s possible the risk predictor’s performance could be further enhanced by incorporation of several potentially important factors that weren’t available in the UCC-SMART derivation cohort, including race, education level, and socioeconomic status, she added.
Potential applications for the risk calculator in clinical practice require further study, but include using the lifetime risk prediction for cancer as a motivational aid in conversations with patients about the importance of behavioral change in support of a healthier lifestyle. Also, a high predicted 10-year lung cancer risk could potentially be used to lower the threshold for a screening chest CT, resulting in earlier detection and treatment of lung cancer, Dr. van ’t Klooster noted.
In an interview, Bonnie Ky, MD, MSCE, praised the risk prediction study as rigorously executed, topical, and clinically significant.
“This paper signifies the overlap between our two disciplines of cancer and cardiovascular disease in terms of the risks that we face together when we care for this patient population,” said Dr. Ky, a cardiologist at the University of Pennsylvania, Philadelphia.
“Many of us in medicine believe in the importance of risk prediction: identifying who’s at high risk and doing everything we can to mitigate that risk. This paper speaks to that and moves us one step closer to accomplishing that aim,” added Dr. Ky, who is editor in chief of JACC: CardioOncology, which published the study simultaneously with Dr. van ’t Klooster’s presentation at ESC 2020. The paper provides direct access to the risk calculator.
Dr. van ’t Klooster reported having no financial conflicts regarding her study. UCC-SMART is funded by a Utrecht University grant, and CANTOS was funded by Novartis.
SOURCE: van ’t Klooster CC. ESC 2020 and JACC CardioOncol. 2020 Aug. doi: 10.1016/j.jaccao.2020.07.001.
FROM ESC CONGRESS 2020
The earlier the better for colchicine post-MI: COLCOT
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
The earlier the anti-inflammatory drug colchicine is initiated after a myocardial infarction (MI) the greater the benefit, a new COLCOT analysis suggests.
The parent trial was conducted in patients with a recent MI because of the intense inflammation present at that time, and added colchicine 0.5 mg daily to standard care within 30 days following MI.
As previously reported, colchicine significantly reduced the risk of the primary end point – a composite of cardiovascular (CV) death, resuscitated cardiac arrest, MI, stroke, or urgent hospitalization for angina requiring revascularization – by 23% compared with placebo.
This new analysis shows the risk was reduced by 48% in patients receiving colchicine within 3 days of an MI (4.3% vs. 8.3%; adjusted hazard ratio, 0.52; 95% confidence interval, 0.32-0.84, P = .007).
Risk of a secondary efficacy end point – CV death, resuscitated cardiac arrest, MI, or stroke – was reduced by 45% over an average follow up of 22.7 months (3.3% vs 6.1%; adjusted HR, 0.55; 95% CI, 0.32-0.95, P = .031).
“We believe that our results support an early, in-hospital initiation of adjunctive colchicine for post-MI prevention,” Nadia Bouabdallaoui, MD, Montreal Heart Institute, Quebec, Canada, said during an online session devoted to colchicine at the European Society of Cardiology Congress 2020.
Session moderator Massimo Imazio, MD, professor of cardiology at the University of Turin, Italy, said the improved outcomes suggest that earlier treatment is better – a finding that parallels his own experience using colchicine in patients with pericarditis.
“This substudy is very important because this is probably also the year in cardiovascular applications [that] early use of the drug could improve outcomes,” he said.
Positive data have been accumulating for colchicine from COLCOT, LoDoCo, and, most recently, the LoDoCo2 trial, even as another anti-inflammatory drug, methotrexate, flamed out as secondary prevention in the CIRT trial.
The new COLCOT substudy included 4,661 of the 4,745 original patients and examined treatment initiation using three strata: within 0-3 days (n = 1,193), 4-7 days (n = 720), and 8-30 days (n = 2,748). Patients who received treatment within 3 days were slightly younger, more likely to be smokers, and to have a shorter time from MI to randomization (2.1 days vs 5.1 days vs. 20.8 days, respectively).
In the subset receiving treatment within 3 days, those assigned to colchicine had the same number of cardiac deaths as those given placebo (2 vs. 2) but fewer resuscitated cardiac arrests (1 vs. 3), MIs (17 vs. 29), strokes (1 vs. 5), and urgent hospitalizations for angina requiring revascularization (6 vs. 17).
“A larger trial might have allowed for a better assessment of individual endpoints and subgroups,” observed Bouabdallaoui.
Although there is growing support for colchicine, experts caution that the drug many not be for everyone. In COLCOT, 1 in 10 patients were unable to tolerate the drug, largely because of gastrointestinal (GI) issues.
Pharmacogenomics substudy
A second COLCOT substudy aimed to identify genetic markers predictive of colchicine response and to gain insights into the mechanisms behind this response. It included 767 patients treated with colchicine and another 755 treated with placebo – or about one-third the patients in the original trial.
A genome-wide association study did not find a significant association for the primary CV endpoint, although a prespecified subgroup analysis in men identified an interesting region on chromosome 9 (variant: rs10811106), which just missed reaching genomewide significance, said Marie-Pierre Dubé, PhD, director of the Université de Montréal Beaulieu-Saucier Pharmacogenomics Centre at the Montreal Heart Institute.
In addition, the genomewide analysis found two significant regions for GI events: one on chromosome 6 (variant: rs6916345) and one on chromosome 10 (variant: rs74795203).
For each of the identified regions, the researchers then tested the effect of the allele in the placebo group and the interaction between the genetic variant and treatment with colchicine. For the chromosome 9 region in males, there was no effect in the placebo group and a significant interaction in the colchicine group.
For the significant GI event findings, there was a small effect for the chromosome 6 region in the placebo group and a very significant interaction with colchicine, Dubé said. Similarly, there was no effect for the chromosome 10 region in the placebo group and a significant interaction with colchicine.
Additional analyses in stratified patient populations showed that males with the protective allele (CC) for the chromosome 9 region represented 83% of the population. The primary CV endpoint occurred in 3.2% of these men treated with colchicine and 6.3% treated with placebo (HR, 0.46; 95% CI, 0.24 - 0.86).
For the gastrointestinal events, 25% of patients carried the risk allele (AA) for the chromosome 6 region and 36.9% of these had GI events when treated with colchicine versus 18.6% when treated with placebo (HR, 2.42; 95% CI, 1.57-3.72).
Similarly, 13% of individuals carried one or two copies of the risk allele (AG+GG) for the chromosome 10 region and the risk of GI events in these was nearly four times higher with colchicine (47.1% vs. 18.9%; HR, 3.98; 95% CI 2.24-7.07).
Functional genomic analyses of the identified regions were also performed and showed that the chromosome 9 locus overlaps with the SAXO1 gene, a stabilizer of axonemal microtubules 1.
“The leading variant at this locus (rs10811106 C allele) correlated with the expression of the HAUS6 gene, which is involved in microtubule generation from existing microtubules, and may interact with the effect of colchicine, which is known to inhibit microtubule formation,” observed Dubé.
Also, the chromosome 6 locus associated with gastrointestinal events was colocalizing with the Crohn’s disease locus, adding further support for this region.
“The results support potential personalized approaches to inflammation reduction for cardiovascular prevention,” Dubé said.
This is a post hoc subgroup analysis, however, and replication is necessary, ideally in prospective randomized trials, she noted.
The substudy is important because it provides further insights into the link between colchicine and microtubule polymerization, affecting the activation of the inflammasome, session moderator Imazio said.
“Second, it is important because pharmacogenomics can help us to better understand the optimal responder to colchicine and colchicine resistance,” he said. “So it can be useful for personalized medicine, leading to the proper use of the drug for the proper patient.”
COLCOT was supported by the government of Quebec, the Canadian Institutes of Health Research, and philanthropic foundations. Bouabdallaoui has disclosed no relevant financial relationships. Dubé reported grants from the government of Quebec; personal fees from DalCor and GlaxoSmithKline; research support from AstraZeneca, Pfizer, Servier, Sanofi; and minor equity interest in DalCor. Dubé is also coauthor of patents on pharmacogenomics-guided CETP inhibition, and pharmacogenomics markers of response to colchicine.
This article first appeared on Medscape.com.
Does evidence support the use of supplements to aid in BP control?
EVIDENCE SUMMARY
Cocoa. A 2017 Cochrane review evaluated data from more than 1800 patients (401 in hypertension studies) to determine the effect of cocoa on BP.1 Compared with placebo (in flavanol-free or low-flavanol controls), cocoa lowered systolic BP by 1.8 mm Hg (confidence interval [CI], –3.1 to –0.4) and diastolic BP by 1.8 mm Hg (CI, –2.6 to –0.9). Further analysis of patients with hypertension (only) showed a reduction in systolic BP of 4 mm Hg (CI, –6.7 to –1.3).
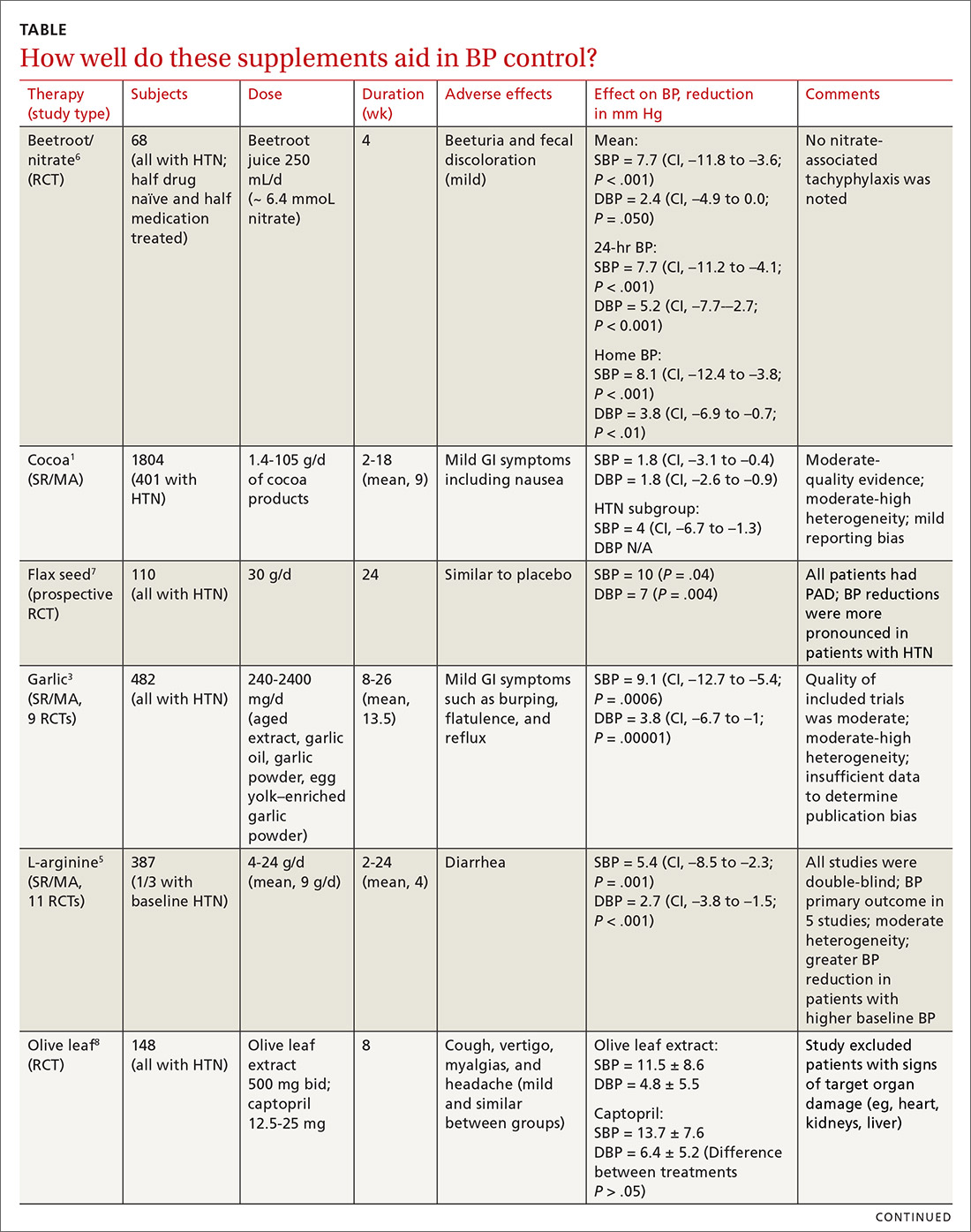
Omega-3 fatty acids. Similarly, a 2014 meta-analysis investigating omega-3 fatty acids (eicosapentaenoic acid [EPA] + docosahexaenoic acid [DHA]) included data from 4489 patients (956 with hypertension) and showed reductions in systolic BP of 1.5 mm Hg (CI, –2.3 to –0.8) and diastolic BP of 1 mm Hg (CI, –1.5 to –0.4), compared with placebo.2 Again, subgroup analysis of patients with hypertension (only) at baseline revealed a greater decrease in systolic and diastolic BP: 4.5 mm Hg (CI, –6.1 to –2.8) and 3.1 mm Hg (CI, –4.4 to –1.8), respectively.
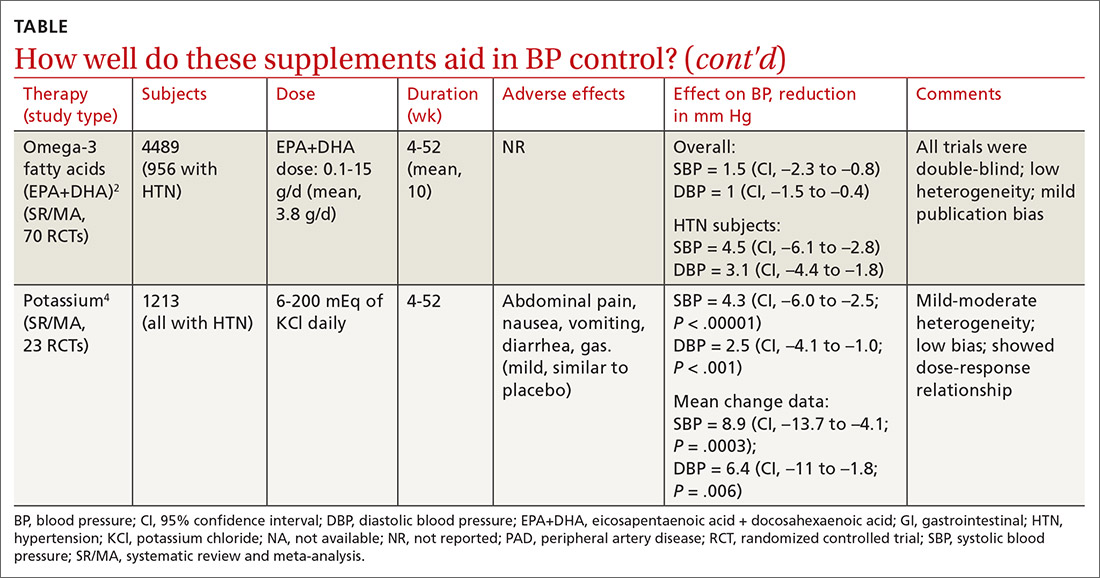
Garlic and potassium chloride. Separate meta-analyses that included only patients with hypertension found that both garlic and potassium significantly lowered BP.3,4 A 2015 meta-analysis comparing a variety of garlic preparations with placebo in patients with hypertension showed decreases in systolic BP of 9.1 mm Hg (CI, –12.7 to –5.4) and in diastolic BP of 3.8 mm Hg (CI, –6.7 to –1).3 Meanwhile, a meta-analysis in 2017 comparing different doses of potassium chloride with placebo demonstrated reductions in systolic BP of 4.3 mm Hg (CI, –6 to –2.5) and diastolic BP of 2.5 mm Hg (CI, –4.1 to –1).4
L-arginine. Another meta-analysis of randomized controlled trials reported evidence that oral L-arginine, compared with placebo, significantly reduced systolic BP by 5.4 mm Hg (CI, –8.5 to –2.3) and diastolic BP by 2.7 mm Hg (CI, –3.8 to –1.5).5 Close to one-third of patients had hypertension at baseline.
Beetroot juice. A double-blind, placebo-controlled study showed that consumption of beetroot juice (with nitrate) once daily reduced BP in 3 different settings (clinic, 24-hour ambulatory, and home readings) when compared with placebo (nitrate-free beetroot juice).6 Study participants were mostly British women, overweight, without significant cardiovascular or renal disease, and with uncontrolled ambulatory BP (> 135/85 mm Hg).
Flax seed. A prospective, double-blind trial of patients with peripheral artery disease compared the antihypertensive effects of flax seed with placebo in patients with and without hypertension and found marked decreases in systolic and diastolic BP.7 Study participants were all older than 40 years without other major cardiac or renal disease, and the majority of enrolled patients with hypertension were concurrently taking medications to treat hypertension during the study.
Olive leaf extract. A double-blind, parallel, and active-control clinical trial in Indonesia compared the BP-lowering effect of olive leaf extract (Olea europaea) to captopril as monotherapies in patients with stage 1 hypertension.8 After a 4-week period of dietary intervention, individuals who were still hypertensive (range, 140/90 to 159/99 mm Hg) were treated with either olive leaf extract or captopril. After 8 weeks of treatment, both groups saw comparable reductions in BP.
Continue to: Editor's takeaway
Editor’s takeaway
Many studies have demonstrated BP benefits from a variety of natural supplements. Although the studies’ durations are short, the effects sometimes modest, and the outcomes disease-oriented rather than patient-oriented, the findings can provide a useful complement to our efforts to manage this most common chronic disease.
1. Ried K, Fakler P, Stocks NP. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2017;(4):CD008893.
2. Miller PE, Van Elswyk M, Alexander DD. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: a meta-analysis of randomized controlled trials. Am J Hypertens. 2014;27:885-896.
3. Rohner A, Ried K, Sobenin IA, et al. A systematic review and meta-analysis on the effects of garlic preparations on blood pressure in individuals with hypertension. Am J Hypertens. 2015;28:414-423.
4. Poorolajal J, Zeraati F, Soltanian AR, et al. Oral potassium supplementation for management of essential hypertension: a meta-analysis of randomized controlled trials. PLoS One. 2017;12:e0174967.
5. Dong JY, Qin LQ, Zhang Z, et al. Effect of oral L-arginine supplementation on blood pressure: a meta-analysis of randomized, double-blind, placebo-controlled trials. Am Heart J. 2011;162:959-965.
6. Kapil V, Khambata RS, Robertson A, et al. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: a randomized, phase 2, double-blind, placebo-controlled study. Hypertension. 2015;65:320-327.
7. Rodriguez-Leyva D, Weighell W, Edel AL, et al. Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension. 2013;62:1081-1089.
8. Susalit E, Agus N, Effendi I, et al. Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: comparison with captopril. Phytomedicine. 2011;18:251-258.
EVIDENCE SUMMARY
Cocoa. A 2017 Cochrane review evaluated data from more than 1800 patients (401 in hypertension studies) to determine the effect of cocoa on BP.1 Compared with placebo (in flavanol-free or low-flavanol controls), cocoa lowered systolic BP by 1.8 mm Hg (confidence interval [CI], –3.1 to –0.4) and diastolic BP by 1.8 mm Hg (CI, –2.6 to –0.9). Further analysis of patients with hypertension (only) showed a reduction in systolic BP of 4 mm Hg (CI, –6.7 to –1.3).

Omega-3 fatty acids. Similarly, a 2014 meta-analysis investigating omega-3 fatty acids (eicosapentaenoic acid [EPA] + docosahexaenoic acid [DHA]) included data from 4489 patients (956 with hypertension) and showed reductions in systolic BP of 1.5 mm Hg (CI, –2.3 to –0.8) and diastolic BP of 1 mm Hg (CI, –1.5 to –0.4), compared with placebo.2 Again, subgroup analysis of patients with hypertension (only) at baseline revealed a greater decrease in systolic and diastolic BP: 4.5 mm Hg (CI, –6.1 to –2.8) and 3.1 mm Hg (CI, –4.4 to –1.8), respectively.

Garlic and potassium chloride. Separate meta-analyses that included only patients with hypertension found that both garlic and potassium significantly lowered BP.3,4 A 2015 meta-analysis comparing a variety of garlic preparations with placebo in patients with hypertension showed decreases in systolic BP of 9.1 mm Hg (CI, –12.7 to –5.4) and in diastolic BP of 3.8 mm Hg (CI, –6.7 to –1).3 Meanwhile, a meta-analysis in 2017 comparing different doses of potassium chloride with placebo demonstrated reductions in systolic BP of 4.3 mm Hg (CI, –6 to –2.5) and diastolic BP of 2.5 mm Hg (CI, –4.1 to –1).4
L-arginine. Another meta-analysis of randomized controlled trials reported evidence that oral L-arginine, compared with placebo, significantly reduced systolic BP by 5.4 mm Hg (CI, –8.5 to –2.3) and diastolic BP by 2.7 mm Hg (CI, –3.8 to –1.5).5 Close to one-third of patients had hypertension at baseline.
Beetroot juice. A double-blind, placebo-controlled study showed that consumption of beetroot juice (with nitrate) once daily reduced BP in 3 different settings (clinic, 24-hour ambulatory, and home readings) when compared with placebo (nitrate-free beetroot juice).6 Study participants were mostly British women, overweight, without significant cardiovascular or renal disease, and with uncontrolled ambulatory BP (> 135/85 mm Hg).
Flax seed. A prospective, double-blind trial of patients with peripheral artery disease compared the antihypertensive effects of flax seed with placebo in patients with and without hypertension and found marked decreases in systolic and diastolic BP.7 Study participants were all older than 40 years without other major cardiac or renal disease, and the majority of enrolled patients with hypertension were concurrently taking medications to treat hypertension during the study.
Olive leaf extract. A double-blind, parallel, and active-control clinical trial in Indonesia compared the BP-lowering effect of olive leaf extract (Olea europaea) to captopril as monotherapies in patients with stage 1 hypertension.8 After a 4-week period of dietary intervention, individuals who were still hypertensive (range, 140/90 to 159/99 mm Hg) were treated with either olive leaf extract or captopril. After 8 weeks of treatment, both groups saw comparable reductions in BP.
Continue to: Editor's takeaway
Editor’s takeaway
Many studies have demonstrated BP benefits from a variety of natural supplements. Although the studies’ durations are short, the effects sometimes modest, and the outcomes disease-oriented rather than patient-oriented, the findings can provide a useful complement to our efforts to manage this most common chronic disease.
EVIDENCE SUMMARY
Cocoa. A 2017 Cochrane review evaluated data from more than 1800 patients (401 in hypertension studies) to determine the effect of cocoa on BP.1 Compared with placebo (in flavanol-free or low-flavanol controls), cocoa lowered systolic BP by 1.8 mm Hg (confidence interval [CI], –3.1 to –0.4) and diastolic BP by 1.8 mm Hg (CI, –2.6 to –0.9). Further analysis of patients with hypertension (only) showed a reduction in systolic BP of 4 mm Hg (CI, –6.7 to –1.3).

Omega-3 fatty acids. Similarly, a 2014 meta-analysis investigating omega-3 fatty acids (eicosapentaenoic acid [EPA] + docosahexaenoic acid [DHA]) included data from 4489 patients (956 with hypertension) and showed reductions in systolic BP of 1.5 mm Hg (CI, –2.3 to –0.8) and diastolic BP of 1 mm Hg (CI, –1.5 to –0.4), compared with placebo.2 Again, subgroup analysis of patients with hypertension (only) at baseline revealed a greater decrease in systolic and diastolic BP: 4.5 mm Hg (CI, –6.1 to –2.8) and 3.1 mm Hg (CI, –4.4 to –1.8), respectively.

Garlic and potassium chloride. Separate meta-analyses that included only patients with hypertension found that both garlic and potassium significantly lowered BP.3,4 A 2015 meta-analysis comparing a variety of garlic preparations with placebo in patients with hypertension showed decreases in systolic BP of 9.1 mm Hg (CI, –12.7 to –5.4) and in diastolic BP of 3.8 mm Hg (CI, –6.7 to –1).3 Meanwhile, a meta-analysis in 2017 comparing different doses of potassium chloride with placebo demonstrated reductions in systolic BP of 4.3 mm Hg (CI, –6 to –2.5) and diastolic BP of 2.5 mm Hg (CI, –4.1 to –1).4
L-arginine. Another meta-analysis of randomized controlled trials reported evidence that oral L-arginine, compared with placebo, significantly reduced systolic BP by 5.4 mm Hg (CI, –8.5 to –2.3) and diastolic BP by 2.7 mm Hg (CI, –3.8 to –1.5).5 Close to one-third of patients had hypertension at baseline.
Beetroot juice. A double-blind, placebo-controlled study showed that consumption of beetroot juice (with nitrate) once daily reduced BP in 3 different settings (clinic, 24-hour ambulatory, and home readings) when compared with placebo (nitrate-free beetroot juice).6 Study participants were mostly British women, overweight, without significant cardiovascular or renal disease, and with uncontrolled ambulatory BP (> 135/85 mm Hg).
Flax seed. A prospective, double-blind trial of patients with peripheral artery disease compared the antihypertensive effects of flax seed with placebo in patients with and without hypertension and found marked decreases in systolic and diastolic BP.7 Study participants were all older than 40 years without other major cardiac or renal disease, and the majority of enrolled patients with hypertension were concurrently taking medications to treat hypertension during the study.
Olive leaf extract. A double-blind, parallel, and active-control clinical trial in Indonesia compared the BP-lowering effect of olive leaf extract (Olea europaea) to captopril as monotherapies in patients with stage 1 hypertension.8 After a 4-week period of dietary intervention, individuals who were still hypertensive (range, 140/90 to 159/99 mm Hg) were treated with either olive leaf extract or captopril. After 8 weeks of treatment, both groups saw comparable reductions in BP.
Continue to: Editor's takeaway
Editor’s takeaway
Many studies have demonstrated BP benefits from a variety of natural supplements. Although the studies’ durations are short, the effects sometimes modest, and the outcomes disease-oriented rather than patient-oriented, the findings can provide a useful complement to our efforts to manage this most common chronic disease.
1. Ried K, Fakler P, Stocks NP. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2017;(4):CD008893.
2. Miller PE, Van Elswyk M, Alexander DD. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: a meta-analysis of randomized controlled trials. Am J Hypertens. 2014;27:885-896.
3. Rohner A, Ried K, Sobenin IA, et al. A systematic review and meta-analysis on the effects of garlic preparations on blood pressure in individuals with hypertension. Am J Hypertens. 2015;28:414-423.
4. Poorolajal J, Zeraati F, Soltanian AR, et al. Oral potassium supplementation for management of essential hypertension: a meta-analysis of randomized controlled trials. PLoS One. 2017;12:e0174967.
5. Dong JY, Qin LQ, Zhang Z, et al. Effect of oral L-arginine supplementation on blood pressure: a meta-analysis of randomized, double-blind, placebo-controlled trials. Am Heart J. 2011;162:959-965.
6. Kapil V, Khambata RS, Robertson A, et al. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: a randomized, phase 2, double-blind, placebo-controlled study. Hypertension. 2015;65:320-327.
7. Rodriguez-Leyva D, Weighell W, Edel AL, et al. Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension. 2013;62:1081-1089.
8. Susalit E, Agus N, Effendi I, et al. Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: comparison with captopril. Phytomedicine. 2011;18:251-258.
1. Ried K, Fakler P, Stocks NP. Effect of cocoa on blood pressure. Cochrane Database Syst Rev. 2017;(4):CD008893.
2. Miller PE, Van Elswyk M, Alexander DD. Long-chain omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and blood pressure: a meta-analysis of randomized controlled trials. Am J Hypertens. 2014;27:885-896.
3. Rohner A, Ried K, Sobenin IA, et al. A systematic review and meta-analysis on the effects of garlic preparations on blood pressure in individuals with hypertension. Am J Hypertens. 2015;28:414-423.
4. Poorolajal J, Zeraati F, Soltanian AR, et al. Oral potassium supplementation for management of essential hypertension: a meta-analysis of randomized controlled trials. PLoS One. 2017;12:e0174967.
5. Dong JY, Qin LQ, Zhang Z, et al. Effect of oral L-arginine supplementation on blood pressure: a meta-analysis of randomized, double-blind, placebo-controlled trials. Am Heart J. 2011;162:959-965.
6. Kapil V, Khambata RS, Robertson A, et al. Dietary nitrate provides sustained blood pressure lowering in hypertensive patients: a randomized, phase 2, double-blind, placebo-controlled study. Hypertension. 2015;65:320-327.
7. Rodriguez-Leyva D, Weighell W, Edel AL, et al. Potent antihypertensive action of dietary flaxseed in hypertensive patients. Hypertension. 2013;62:1081-1089.
8. Susalit E, Agus N, Effendi I, et al. Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: comparison with captopril. Phytomedicine. 2011;18:251-258.
EVIDENCE-BASED ANSWER:
Yes. A number of well-tolerated natural therapies have been shown to reduce systolic and diastolic blood pressure (BP). (See Table1-8 for summary.) However, the studies don’t provide direct evidence of whether the decrease in BP is linked to patient-oriented outcomes. Nor do they allow definitive conclusions concerning the lasting nature of the reductions, because most studies were fewer than 6 months in duration (strength of recommendation: C, disease-oriented evidence).
It's time to change when BP meds are taken
In this issue of JFP, there is an extraordinarily valuable PURL (Priority Updates from the Research Literature) for you. PURLs® are written by academic family physicians who comb through volumes of research to identify and then summarize for JFP important studies we believe should change your practice. After reading a PURL, you may find that you have already implemented this new evidence into your practice. In that case, the PURL confirms that you are doing the right thing.
Here is the good news from this month’s PURL: Having patients take their blood pressure (BP) medication in the evening, rather than in the morning, leads not only to better BP control but also to a reduction in cardiovascular events.1 How large is this effect? This simple intervention nearly cut in half the number of major cardiovascular events—including myocardial infarction, stroke, and congestive heart failure—and the risk for death from a cardiovascular event was reduced 56%. The number needed to treat to prevent 1 major cardiovascular event over the course of 6.3 years was 20. That means this intervention is more powerful than taking a statin!
The investigators, who call this intervention “chronotherapy” since it takes into account the body’s circadian rhythms, have been studying the effect of this simple intervention for many years, and this large randomized trial provides very strong evidence that it’s effective. Despite the excellent trial design and execution, some cardiovascular researchers have questioned the integrity of the trial and believe patients should continue to take their antihypertensives in the morning.2 The main investigator of the study, however, has provided a very strong rebuttal in print.3
I am delighted to see the positive results of this definitive trial of chronotherapy for hypertension. When these investigators published their first randomized trial of chronotherapy in 2010,4 which demonstrated a significant BP reduction with evening dosing of antihypertensives, I began telling all of my patients to take at least 1 of their antihypertensive meds in the evening. Maybe I was jumping the gun at that time, but we should all tell our patients to take their BP medication in the evening from now on. What could be an easier way to reduce cardiovascular morbidity and mortality?
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754
2. Kreutz R, Kjeldsen SE, Burnier M, et al. Blood pressure medication should not be routinely dosed at bedtime. We must disregard the data from the HYGIA project [editorial]. Blood Press. 2020;29:135-136.
3. Crespo JJ, Domínguez-Sardiña M, Otero A, et. al. Bedtime hypertension chronotherapy best reduces cardiovascular disease risk as corroborated by the Hygia Chronotherapy Trial. Rebuttal to European Society of Hypertension officials. Chronobiol Int. 2020;37:771-780.
4. Hermida RC, Ayala DE, Mojón A, Fernández JR. Influence of circadian time of hypertension treatment on cardiovascular risk: results of the MAPEC study. Chronobiol Int. 2010;27:1629-1651.
In this issue of JFP, there is an extraordinarily valuable PURL (Priority Updates from the Research Literature) for you. PURLs® are written by academic family physicians who comb through volumes of research to identify and then summarize for JFP important studies we believe should change your practice. After reading a PURL, you may find that you have already implemented this new evidence into your practice. In that case, the PURL confirms that you are doing the right thing.
Here is the good news from this month’s PURL: Having patients take their blood pressure (BP) medication in the evening, rather than in the morning, leads not only to better BP control but also to a reduction in cardiovascular events.1 How large is this effect? This simple intervention nearly cut in half the number of major cardiovascular events—including myocardial infarction, stroke, and congestive heart failure—and the risk for death from a cardiovascular event was reduced 56%. The number needed to treat to prevent 1 major cardiovascular event over the course of 6.3 years was 20. That means this intervention is more powerful than taking a statin!
The investigators, who call this intervention “chronotherapy” since it takes into account the body’s circadian rhythms, have been studying the effect of this simple intervention for many years, and this large randomized trial provides very strong evidence that it’s effective. Despite the excellent trial design and execution, some cardiovascular researchers have questioned the integrity of the trial and believe patients should continue to take their antihypertensives in the morning.2 The main investigator of the study, however, has provided a very strong rebuttal in print.3
I am delighted to see the positive results of this definitive trial of chronotherapy for hypertension. When these investigators published their first randomized trial of chronotherapy in 2010,4 which demonstrated a significant BP reduction with evening dosing of antihypertensives, I began telling all of my patients to take at least 1 of their antihypertensive meds in the evening. Maybe I was jumping the gun at that time, but we should all tell our patients to take their BP medication in the evening from now on. What could be an easier way to reduce cardiovascular morbidity and mortality?
In this issue of JFP, there is an extraordinarily valuable PURL (Priority Updates from the Research Literature) for you. PURLs® are written by academic family physicians who comb through volumes of research to identify and then summarize for JFP important studies we believe should change your practice. After reading a PURL, you may find that you have already implemented this new evidence into your practice. In that case, the PURL confirms that you are doing the right thing.
Here is the good news from this month’s PURL: Having patients take their blood pressure (BP) medication in the evening, rather than in the morning, leads not only to better BP control but also to a reduction in cardiovascular events.1 How large is this effect? This simple intervention nearly cut in half the number of major cardiovascular events—including myocardial infarction, stroke, and congestive heart failure—and the risk for death from a cardiovascular event was reduced 56%. The number needed to treat to prevent 1 major cardiovascular event over the course of 6.3 years was 20. That means this intervention is more powerful than taking a statin!
The investigators, who call this intervention “chronotherapy” since it takes into account the body’s circadian rhythms, have been studying the effect of this simple intervention for many years, and this large randomized trial provides very strong evidence that it’s effective. Despite the excellent trial design and execution, some cardiovascular researchers have questioned the integrity of the trial and believe patients should continue to take their antihypertensives in the morning.2 The main investigator of the study, however, has provided a very strong rebuttal in print.3
I am delighted to see the positive results of this definitive trial of chronotherapy for hypertension. When these investigators published their first randomized trial of chronotherapy in 2010,4 which demonstrated a significant BP reduction with evening dosing of antihypertensives, I began telling all of my patients to take at least 1 of their antihypertensive meds in the evening. Maybe I was jumping the gun at that time, but we should all tell our patients to take their BP medication in the evening from now on. What could be an easier way to reduce cardiovascular morbidity and mortality?
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754
2. Kreutz R, Kjeldsen SE, Burnier M, et al. Blood pressure medication should not be routinely dosed at bedtime. We must disregard the data from the HYGIA project [editorial]. Blood Press. 2020;29:135-136.
3. Crespo JJ, Domínguez-Sardiña M, Otero A, et. al. Bedtime hypertension chronotherapy best reduces cardiovascular disease risk as corroborated by the Hygia Chronotherapy Trial. Rebuttal to European Society of Hypertension officials. Chronobiol Int. 2020;37:771-780.
4. Hermida RC, Ayala DE, Mojón A, Fernández JR. Influence of circadian time of hypertension treatment on cardiovascular risk: results of the MAPEC study. Chronobiol Int. 2010;27:1629-1651.
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754
2. Kreutz R, Kjeldsen SE, Burnier M, et al. Blood pressure medication should not be routinely dosed at bedtime. We must disregard the data from the HYGIA project [editorial]. Blood Press. 2020;29:135-136.
3. Crespo JJ, Domínguez-Sardiña M, Otero A, et. al. Bedtime hypertension chronotherapy best reduces cardiovascular disease risk as corroborated by the Hygia Chronotherapy Trial. Rebuttal to European Society of Hypertension officials. Chronobiol Int. 2020;37:771-780.
4. Hermida RC, Ayala DE, Mojón A, Fernández JR. Influence of circadian time of hypertension treatment on cardiovascular risk: results of the MAPEC study. Chronobiol Int. 2010;27:1629-1651.
Is it better to take that antihypertensive at night?
ILLUSTRATIVE CASE
A 54-year-old White woman presents to your office with new-onset hypertension. As you are discussing options for treatment, she mentions she would prefer once-daily dosing to help her remember to take her medication. She also wants to know what the best time of day is to take her medication to reduce her risk of cardiovascular disease (CVD). What do you advise?
The burden of hypertension is significant and growing in the United States. The 2017 American College of Cardiology/American Heart Association (ACC/AHA) guidelines reported that more than 108 million people were affected in 2015-2016—up from 87 million in 1999-2000.2 Yet control of hypertension is improving among those receiving antihypertension pharmacotherapy. As reported in the ACC/AHA guidelines, data from the 2016 National Health and Nutrition Examination Survey (NHANES) indicate an increase of controlled hypertension among those receiving treatment from 25.6% (1999-2000) to 43.5% (2015-2016).2
Chronotherapy involves the administration of medication in coordination with the body’s circadian rhythms to maximize therapeutic effectiveness and/or minimize adverse effects. It is not a new concept as it applies to hypertension. Circadian rhythm–dependent mechanisms influence the natural rise and fall of blood pressure (BP).1 The renin-angiotensin-aldosterone system, known to be most active at night, is a target mechanism for BP control.1 Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) are more effective (alone or in combination with other agents) at reducing BP during sleep and wakefulness when they are taken at night.3,4 Additional prospective clinical trials and systematic reviews have documented improved BP during sleep and on 24-hour ambulatory monitoring when antihypertensives are taken at bedtime.3-5
However, there have been few long-term studies assessing the effects of bedtime administration of antihypertensive medication on CVD risk reduction with patient-oriented outcomes.6,7 Additionally, no studies have evaluated morning vs bedtime administration of antihypertensive medication for CVD risk reduction in a primary care setting. The 2019 ACC/AHA guideline on the primary prevention of CVD offers no recommendation regarding when to take antihypertensive medication.8 Timing of medication administration also is not addressed in the NHANES study of hypertension awareness, treatment, and control in US adults.9
This study sought to determine in a primary care setting whether taking antihypertensives at bedtime, as opposed to upon waking, more effectively reduces CVD risk.
STUDY SUMMARY
PM vs AM antihypertensive dosing reduces CV events
This prospective, randomized, open-label, blinded endpoint trial of antihypertensive medication administration timing was part of a large, multicenter Spanish study investigating ambulatory BP monitoring (ABPM) as a routine diagnostic tool.
Study participants were randomly assigned in a 1:1 ratio to 2 treatment arms; participants either took all of their BP medications in the morning upon waking (n = 9532) or right before bedtime (n = 9552). The study was conducted in a primary care clinical setting. It included adult participants (age ≥ 18 years) with hypertension (defined as having at least 1 of the following benchmarks: awake systolic BP [SBP] mean ≥ 135 mm Hg, awake diastolic BP (DBP) mean ≥ 85 mm Hg, asleep SBP mean ≥ 120 mm Hg, asleep DBP mean ≥ 70 mm Hg as corroborated by 48-hour ABPM) who were taking at least 1 antihypertensive medication.
Continue to: Any antihypertension medication...
Any antihypertension medication included in the Spanish national formulary was allowed (exact agents were not delineated, but the following classes were included: ARB, ACE inhibitor, calcium channel blocker [CCB], beta-blocker, and/or diuretic). All BP medications had to be dosed once daily for inclusion. Exclusion criteria included pregnancy, night or rotating-shift work, alcohol or other substance dependence, acquired immunodeficiency syndrome, preexisting CVD (unstable angina, heart failure, arrhythmia, kidney failure, and retinopathy), inability to tolerate ABPM, and inability to comply with required 1-year follow-up.
Upon enrollment and at every subsequent clinic visit (scheduled at least annually), participants underwent 48-hour ABPM. Those with uncontrolled BP or elevated CVD risk had scheduled follow-up and ABPM more frequently. The primary outcome was a composite of CVD events including new-onset myocardial infarction, coronary revascularization, heart failure, ischemic stroke, hemorrhagic stroke, and CVD death. Secondary endpoints were individually analyzed primary outcomes of CVD events. The typical patient at baseline was 60.5 years of age with a body mass index of 29.7, an almost 9-year duration of hypertension, and a baseline office BP of 149/86 mm Hg. The patient break-out by antihypertensive class (awakening vs bedtime groups) was as follows: ARB (53% vs 53%), ACE inhibitor (25% vs 23%), CCB (33% vs 37%), beta-blocker (22% vs 18%), and diuretic (47% vs 40%).
During the median 6.3-year patient follow-up period, 1752 participants experienced a total of 2454 CVD events. Patients in the bedtime administration group, compared with those in the morning group, showed significantly lower risk for a CVD event (hazard ratio [HR] = 0.55; 95% confidence interval [CI], 0.50-0.61; P < .001). Also, there was a lower risk for individual CVD events in the bedtime administration group: CVD death (HR = 0.44; 95% CI, 0.34-0.56), myocardial infarction (HR = 0.66; 95% CI, 0.52-0.84), coronary revascularization (HR = 0.60; 95% CI, 0.47-0.75), heart failure (HR = 0.58; 95% CI, 0.49-0.70), and stroke (HR = 0.51; 95% CI, 0.41-0.63). This difference remained after correction for multiple potential confounders. There were no differences in adverse events, such as sleep-time hypotension, between groups.
WHAT’S NEW
First RCT in primary care to show dosing time change reduces CV risk
This is the first randomized controlled trial (RCT) performed in a primary care setting to compare before-bedtime to upon-waking administration of antihypertensive medications using clinically significant endpoints. The study demonstrates that a simple change in administration time has the potential to significantly improve the lives of our patients by reducing the risk for cardiovascular events and their medication burden.
CAVEATS
Homogenous population and exclusions limit generalizability
Because the study population consisted of white Spanish men and women, the results may not be generalizable beyond that ethnic group. In addition, the study exclusions limit interpretation in night/rotating-shift employees, patients with secondary hypertension, and those with CVD, chronic kidney disease, or severe retinopathy looking to reduce their risk.
Continue to: CHALLENGES TO IMPLEMENTATION
CHALLENGES TO IMPLEMENTATION
Nighttime urination could lead to nonadherence
Taking diuretics at bedtime may result in unwanted nighttime awakenings for visits to the bathroom, which could lead to nonadherence in some patients.
ACKNOWLEDGMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754.
2. Dorans KS, Mills KT, Liu Y, et al. Trends in prevalence and control of hypertension according to the 2017 American College of Cardiology/American Heart Association (ACC/AHA) guideline. J Am Heart Assoc. 2018;7:e008888.
3. Hermida RC, Ayala DE, Smolensky MH, et al. Chronotherapy with conventional blood pressure medications improves management of hypertension and reduces cardiovascular and stroke risks. Hypertens Res. 2016;39:277-292.
4. Bowles NP, Thosar SS, Herzig MX, et al. Chronotherapy for hypertension. Curr Hypertens Rep. 2018;20:97.
5. Zhao P, Xu P, Wan C, et al. Evening versus morning dosing regimen drug therapy for hypertension. Cochrane Database Syst Rev. 2011:CD004184.
6. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients: the Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
7. Black HR, Elliott WJ, Grandits G, et al. Principal results of the Controlled Onset Verapamil Investigation of Cardiovascular End Points (CONVINCE) trial. JAMA. 2003;289:2073-2082.
8. Arnette DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;74:e177-e232.
9. Foti K, Wang D, Appel LJ, et al. Hypertension awareness, treatment, and control in US adults: trends in the hypertensive control cascade by population subgroup (National Health and Nutrition Examination Survey, 1999-2016). Am J Epidemiol. 2019;188:2165-2174.
ILLUSTRATIVE CASE
A 54-year-old White woman presents to your office with new-onset hypertension. As you are discussing options for treatment, she mentions she would prefer once-daily dosing to help her remember to take her medication. She also wants to know what the best time of day is to take her medication to reduce her risk of cardiovascular disease (CVD). What do you advise?
The burden of hypertension is significant and growing in the United States. The 2017 American College of Cardiology/American Heart Association (ACC/AHA) guidelines reported that more than 108 million people were affected in 2015-2016—up from 87 million in 1999-2000.2 Yet control of hypertension is improving among those receiving antihypertension pharmacotherapy. As reported in the ACC/AHA guidelines, data from the 2016 National Health and Nutrition Examination Survey (NHANES) indicate an increase of controlled hypertension among those receiving treatment from 25.6% (1999-2000) to 43.5% (2015-2016).2
Chronotherapy involves the administration of medication in coordination with the body’s circadian rhythms to maximize therapeutic effectiveness and/or minimize adverse effects. It is not a new concept as it applies to hypertension. Circadian rhythm–dependent mechanisms influence the natural rise and fall of blood pressure (BP).1 The renin-angiotensin-aldosterone system, known to be most active at night, is a target mechanism for BP control.1 Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) are more effective (alone or in combination with other agents) at reducing BP during sleep and wakefulness when they are taken at night.3,4 Additional prospective clinical trials and systematic reviews have documented improved BP during sleep and on 24-hour ambulatory monitoring when antihypertensives are taken at bedtime.3-5
However, there have been few long-term studies assessing the effects of bedtime administration of antihypertensive medication on CVD risk reduction with patient-oriented outcomes.6,7 Additionally, no studies have evaluated morning vs bedtime administration of antihypertensive medication for CVD risk reduction in a primary care setting. The 2019 ACC/AHA guideline on the primary prevention of CVD offers no recommendation regarding when to take antihypertensive medication.8 Timing of medication administration also is not addressed in the NHANES study of hypertension awareness, treatment, and control in US adults.9
This study sought to determine in a primary care setting whether taking antihypertensives at bedtime, as opposed to upon waking, more effectively reduces CVD risk.
STUDY SUMMARY
PM vs AM antihypertensive dosing reduces CV events
This prospective, randomized, open-label, blinded endpoint trial of antihypertensive medication administration timing was part of a large, multicenter Spanish study investigating ambulatory BP monitoring (ABPM) as a routine diagnostic tool.
Study participants were randomly assigned in a 1:1 ratio to 2 treatment arms; participants either took all of their BP medications in the morning upon waking (n = 9532) or right before bedtime (n = 9552). The study was conducted in a primary care clinical setting. It included adult participants (age ≥ 18 years) with hypertension (defined as having at least 1 of the following benchmarks: awake systolic BP [SBP] mean ≥ 135 mm Hg, awake diastolic BP (DBP) mean ≥ 85 mm Hg, asleep SBP mean ≥ 120 mm Hg, asleep DBP mean ≥ 70 mm Hg as corroborated by 48-hour ABPM) who were taking at least 1 antihypertensive medication.
Continue to: Any antihypertension medication...
Any antihypertension medication included in the Spanish national formulary was allowed (exact agents were not delineated, but the following classes were included: ARB, ACE inhibitor, calcium channel blocker [CCB], beta-blocker, and/or diuretic). All BP medications had to be dosed once daily for inclusion. Exclusion criteria included pregnancy, night or rotating-shift work, alcohol or other substance dependence, acquired immunodeficiency syndrome, preexisting CVD (unstable angina, heart failure, arrhythmia, kidney failure, and retinopathy), inability to tolerate ABPM, and inability to comply with required 1-year follow-up.
Upon enrollment and at every subsequent clinic visit (scheduled at least annually), participants underwent 48-hour ABPM. Those with uncontrolled BP or elevated CVD risk had scheduled follow-up and ABPM more frequently. The primary outcome was a composite of CVD events including new-onset myocardial infarction, coronary revascularization, heart failure, ischemic stroke, hemorrhagic stroke, and CVD death. Secondary endpoints were individually analyzed primary outcomes of CVD events. The typical patient at baseline was 60.5 years of age with a body mass index of 29.7, an almost 9-year duration of hypertension, and a baseline office BP of 149/86 mm Hg. The patient break-out by antihypertensive class (awakening vs bedtime groups) was as follows: ARB (53% vs 53%), ACE inhibitor (25% vs 23%), CCB (33% vs 37%), beta-blocker (22% vs 18%), and diuretic (47% vs 40%).
During the median 6.3-year patient follow-up period, 1752 participants experienced a total of 2454 CVD events. Patients in the bedtime administration group, compared with those in the morning group, showed significantly lower risk for a CVD event (hazard ratio [HR] = 0.55; 95% confidence interval [CI], 0.50-0.61; P < .001). Also, there was a lower risk for individual CVD events in the bedtime administration group: CVD death (HR = 0.44; 95% CI, 0.34-0.56), myocardial infarction (HR = 0.66; 95% CI, 0.52-0.84), coronary revascularization (HR = 0.60; 95% CI, 0.47-0.75), heart failure (HR = 0.58; 95% CI, 0.49-0.70), and stroke (HR = 0.51; 95% CI, 0.41-0.63). This difference remained after correction for multiple potential confounders. There were no differences in adverse events, such as sleep-time hypotension, between groups.
WHAT’S NEW
First RCT in primary care to show dosing time change reduces CV risk
This is the first randomized controlled trial (RCT) performed in a primary care setting to compare before-bedtime to upon-waking administration of antihypertensive medications using clinically significant endpoints. The study demonstrates that a simple change in administration time has the potential to significantly improve the lives of our patients by reducing the risk for cardiovascular events and their medication burden.
CAVEATS
Homogenous population and exclusions limit generalizability
Because the study population consisted of white Spanish men and women, the results may not be generalizable beyond that ethnic group. In addition, the study exclusions limit interpretation in night/rotating-shift employees, patients with secondary hypertension, and those with CVD, chronic kidney disease, or severe retinopathy looking to reduce their risk.
Continue to: CHALLENGES TO IMPLEMENTATION
CHALLENGES TO IMPLEMENTATION
Nighttime urination could lead to nonadherence
Taking diuretics at bedtime may result in unwanted nighttime awakenings for visits to the bathroom, which could lead to nonadherence in some patients.
ACKNOWLEDGMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 54-year-old White woman presents to your office with new-onset hypertension. As you are discussing options for treatment, she mentions she would prefer once-daily dosing to help her remember to take her medication. She also wants to know what the best time of day is to take her medication to reduce her risk of cardiovascular disease (CVD). What do you advise?
The burden of hypertension is significant and growing in the United States. The 2017 American College of Cardiology/American Heart Association (ACC/AHA) guidelines reported that more than 108 million people were affected in 2015-2016—up from 87 million in 1999-2000.2 Yet control of hypertension is improving among those receiving antihypertension pharmacotherapy. As reported in the ACC/AHA guidelines, data from the 2016 National Health and Nutrition Examination Survey (NHANES) indicate an increase of controlled hypertension among those receiving treatment from 25.6% (1999-2000) to 43.5% (2015-2016).2
Chronotherapy involves the administration of medication in coordination with the body’s circadian rhythms to maximize therapeutic effectiveness and/or minimize adverse effects. It is not a new concept as it applies to hypertension. Circadian rhythm–dependent mechanisms influence the natural rise and fall of blood pressure (BP).1 The renin-angiotensin-aldosterone system, known to be most active at night, is a target mechanism for BP control.1 Angiotensin-converting enzyme (ACE) inhibitors and angiotensin II receptor blockers (ARBs) are more effective (alone or in combination with other agents) at reducing BP during sleep and wakefulness when they are taken at night.3,4 Additional prospective clinical trials and systematic reviews have documented improved BP during sleep and on 24-hour ambulatory monitoring when antihypertensives are taken at bedtime.3-5
However, there have been few long-term studies assessing the effects of bedtime administration of antihypertensive medication on CVD risk reduction with patient-oriented outcomes.6,7 Additionally, no studies have evaluated morning vs bedtime administration of antihypertensive medication for CVD risk reduction in a primary care setting. The 2019 ACC/AHA guideline on the primary prevention of CVD offers no recommendation regarding when to take antihypertensive medication.8 Timing of medication administration also is not addressed in the NHANES study of hypertension awareness, treatment, and control in US adults.9
This study sought to determine in a primary care setting whether taking antihypertensives at bedtime, as opposed to upon waking, more effectively reduces CVD risk.
STUDY SUMMARY
PM vs AM antihypertensive dosing reduces CV events
This prospective, randomized, open-label, blinded endpoint trial of antihypertensive medication administration timing was part of a large, multicenter Spanish study investigating ambulatory BP monitoring (ABPM) as a routine diagnostic tool.
Study participants were randomly assigned in a 1:1 ratio to 2 treatment arms; participants either took all of their BP medications in the morning upon waking (n = 9532) or right before bedtime (n = 9552). The study was conducted in a primary care clinical setting. It included adult participants (age ≥ 18 years) with hypertension (defined as having at least 1 of the following benchmarks: awake systolic BP [SBP] mean ≥ 135 mm Hg, awake diastolic BP (DBP) mean ≥ 85 mm Hg, asleep SBP mean ≥ 120 mm Hg, asleep DBP mean ≥ 70 mm Hg as corroborated by 48-hour ABPM) who were taking at least 1 antihypertensive medication.
Continue to: Any antihypertension medication...
Any antihypertension medication included in the Spanish national formulary was allowed (exact agents were not delineated, but the following classes were included: ARB, ACE inhibitor, calcium channel blocker [CCB], beta-blocker, and/or diuretic). All BP medications had to be dosed once daily for inclusion. Exclusion criteria included pregnancy, night or rotating-shift work, alcohol or other substance dependence, acquired immunodeficiency syndrome, preexisting CVD (unstable angina, heart failure, arrhythmia, kidney failure, and retinopathy), inability to tolerate ABPM, and inability to comply with required 1-year follow-up.
Upon enrollment and at every subsequent clinic visit (scheduled at least annually), participants underwent 48-hour ABPM. Those with uncontrolled BP or elevated CVD risk had scheduled follow-up and ABPM more frequently. The primary outcome was a composite of CVD events including new-onset myocardial infarction, coronary revascularization, heart failure, ischemic stroke, hemorrhagic stroke, and CVD death. Secondary endpoints were individually analyzed primary outcomes of CVD events. The typical patient at baseline was 60.5 years of age with a body mass index of 29.7, an almost 9-year duration of hypertension, and a baseline office BP of 149/86 mm Hg. The patient break-out by antihypertensive class (awakening vs bedtime groups) was as follows: ARB (53% vs 53%), ACE inhibitor (25% vs 23%), CCB (33% vs 37%), beta-blocker (22% vs 18%), and diuretic (47% vs 40%).
During the median 6.3-year patient follow-up period, 1752 participants experienced a total of 2454 CVD events. Patients in the bedtime administration group, compared with those in the morning group, showed significantly lower risk for a CVD event (hazard ratio [HR] = 0.55; 95% confidence interval [CI], 0.50-0.61; P < .001). Also, there was a lower risk for individual CVD events in the bedtime administration group: CVD death (HR = 0.44; 95% CI, 0.34-0.56), myocardial infarction (HR = 0.66; 95% CI, 0.52-0.84), coronary revascularization (HR = 0.60; 95% CI, 0.47-0.75), heart failure (HR = 0.58; 95% CI, 0.49-0.70), and stroke (HR = 0.51; 95% CI, 0.41-0.63). This difference remained after correction for multiple potential confounders. There were no differences in adverse events, such as sleep-time hypotension, between groups.
WHAT’S NEW
First RCT in primary care to show dosing time change reduces CV risk
This is the first randomized controlled trial (RCT) performed in a primary care setting to compare before-bedtime to upon-waking administration of antihypertensive medications using clinically significant endpoints. The study demonstrates that a simple change in administration time has the potential to significantly improve the lives of our patients by reducing the risk for cardiovascular events and their medication burden.
CAVEATS
Homogenous population and exclusions limit generalizability
Because the study population consisted of white Spanish men and women, the results may not be generalizable beyond that ethnic group. In addition, the study exclusions limit interpretation in night/rotating-shift employees, patients with secondary hypertension, and those with CVD, chronic kidney disease, or severe retinopathy looking to reduce their risk.
Continue to: CHALLENGES TO IMPLEMENTATION
CHALLENGES TO IMPLEMENTATION
Nighttime urination could lead to nonadherence
Taking diuretics at bedtime may result in unwanted nighttime awakenings for visits to the bathroom, which could lead to nonadherence in some patients.
ACKNOWLEDGMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754.
2. Dorans KS, Mills KT, Liu Y, et al. Trends in prevalence and control of hypertension according to the 2017 American College of Cardiology/American Heart Association (ACC/AHA) guideline. J Am Heart Assoc. 2018;7:e008888.
3. Hermida RC, Ayala DE, Smolensky MH, et al. Chronotherapy with conventional blood pressure medications improves management of hypertension and reduces cardiovascular and stroke risks. Hypertens Res. 2016;39:277-292.
4. Bowles NP, Thosar SS, Herzig MX, et al. Chronotherapy for hypertension. Curr Hypertens Rep. 2018;20:97.
5. Zhao P, Xu P, Wan C, et al. Evening versus morning dosing regimen drug therapy for hypertension. Cochrane Database Syst Rev. 2011:CD004184.
6. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients: the Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
7. Black HR, Elliott WJ, Grandits G, et al. Principal results of the Controlled Onset Verapamil Investigation of Cardiovascular End Points (CONVINCE) trial. JAMA. 2003;289:2073-2082.
8. Arnette DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;74:e177-e232.
9. Foti K, Wang D, Appel LJ, et al. Hypertension awareness, treatment, and control in US adults: trends in the hypertensive control cascade by population subgroup (National Health and Nutrition Examination Survey, 1999-2016). Am J Epidemiol. 2019;188:2165-2174.
1. Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754.
2. Dorans KS, Mills KT, Liu Y, et al. Trends in prevalence and control of hypertension according to the 2017 American College of Cardiology/American Heart Association (ACC/AHA) guideline. J Am Heart Assoc. 2018;7:e008888.
3. Hermida RC, Ayala DE, Smolensky MH, et al. Chronotherapy with conventional blood pressure medications improves management of hypertension and reduces cardiovascular and stroke risks. Hypertens Res. 2016;39:277-292.
4. Bowles NP, Thosar SS, Herzig MX, et al. Chronotherapy for hypertension. Curr Hypertens Rep. 2018;20:97.
5. Zhao P, Xu P, Wan C, et al. Evening versus morning dosing regimen drug therapy for hypertension. Cochrane Database Syst Rev. 2011:CD004184.
6. Yusuf S, Sleight P, Pogue J, et al. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients: the Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med. 2000;342:145-153.
7. Black HR, Elliott WJ, Grandits G, et al. Principal results of the Controlled Onset Verapamil Investigation of Cardiovascular End Points (CONVINCE) trial. JAMA. 2003;289:2073-2082.
8. Arnette DK, Blumenthal RS, Albert MA, et al. 2019 ACC/AHA guideline on the primary prevention of cardiovascular disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol. 2019;74:e177-e232.
9. Foti K, Wang D, Appel LJ, et al. Hypertension awareness, treatment, and control in US adults: trends in the hypertensive control cascade by population subgroup (National Health and Nutrition Examination Survey, 1999-2016). Am J Epidemiol. 2019;188:2165-2174.
PRACTICE CHANGER
Advise patients to take blood pressure (BP) medication at bedtime rather than upon waking because it results in a decrease in major cardiovascular disease events.
STRENGTH OF RECOMMENDATION
B: Based on a single, good-quality, multicenter trial.
Hermida RC, Crespo JJ, Domínguez-Sardiña M, et al. Bedtime hypertension treatment improves cardiovascular risk reduction: the Hygia Chronotherapy Trial [published online ahead of print October 22, 2019]. Eur Heart J. 2019;ehz754. doi:10.1093/eurheartj/ehz754.1
MIS-C cardiac evaluation requires more than EF
Patients with multisystem inflammatory syndrome caused by COVID-19 typically seem to avoid coronary artery dilation early on, but they may be prone to cardiac injury and dysfunction longer term that requires a more discerning diagnostic approach to sort out.
The findings were revealed in a study of 28 children with COVID-19–related multisystem inflammatory syndrome (MIS-C) at Children’s Hospital of Philadelphia. The study reported that cardiac injury and dysfunction are common in these patients – even those who have preserved ejection fraction – and that diastolic dysfunction is persistent. For comparison, the study also included 20 healthy controls and 20 patients with classic Kawasaki disease (KD).
The study analyzed echocardiography findings in the patients, reporting left ventricular (LV) systolic and diastolic function were worse than in classic Kawasaki disease (KD), which MIS-C mimics. Lead author Daisuke Matsubara, MD, PhD, and colleagues reported that four markers – LV global longitudinal strain, LV circumferential strain rate, right ventricular strain, and left atrial strain – were the strongest predictors of myocardial injury in these patients. After the acute phase, systolic function tended to recover, but diastolic dysfunction persisted.
‘Strain’ measurement boosts accuracy
While echocardiography has been reported to be valuable in evaluating coronary artery function in MIS-C patients, Dr. Matsubara of the division of cardiology at CHOP, said in an interview that study is the first to use the newer echocardiography indexes, known as “strain,” to assess heart function.
“Strain is a more sensitive tool than more conventional indexes and can detect subtle decrease in heart function, even when ejection fraction is preserved,” he said. “Numerous publications have reached conclusions that strain improves the prognostic and diagnostic accuracy of echocardiography in a wide variety of cardiac pathologies causing LV dysfunction.”
Dr. Matsubara noted that the coronary arteries were mostly unaffected in the acute stage of MIS-C, as only one patient in their MIS-C cohort had coronary artery involvement, which normalized during early follow-up. “On the other hand, 20% of our classic KD patients had coronary abnormalities, including two with aneurysms.”
By using positive troponin I or elevated brain natriuretic peptide (BNP) to assess cardiac injury, they found a “high” (60%) incidence of myocardial injury in their MIS-C cohort. During early follow-up, most of the MIS-C patients showed normalization of systolic function, although diastolic dysfunction persisted.
When compared with the classic KD group, MIS-C patients had higher rates of mitral regurgitation (46% vs. 15%, P = .06), more pericardial effusion (32% vs. 15%, P = 0.46), and more pleural effusion (39% vs. 0%, P = .004). MIS-C patients with suspected myocardial injury show these findings more frequently than those with actual myocardial injury.
Compared with the healthy controls, the MIS-C patients showed both LV systolic and diastolic dysfunction as well as significantly lower left atrium (LA) strain and peak right ventricle (RV) free-wall longitudinal strain.
“In addition to the left ventricle, two other chambers of the heart, the LA and the RV that are often labeled as the ‘forgotten chambers’ of the heart, were also affected by MIS-C,” Dr. Matsubara said. “Both LA and RV strains were markedly reduced in MIS-C patients, compared to normal and KD patients.”
The study also indicates that elevated troponin I levels may not be as dire in children as they are in adults. Dr. Matsubara cited a study of more than 2,700 adult COVID-19 patients that found that even mild increases in troponin I level were associated with increased death during hospitalization (J Am Coll Cardiol. 2020;76:533-46).
However, most of the patients in the CHOP study, even those with elevated troponin I levels, recovered systolic function quickly. “We speculate that the elevation in cardiac troponins may have less dire implications in children, likely due to a more transient type of cardiac injury and less comorbidities in children,” he said. “Clearly further studies are needed before a definitive statement can be made.”
Dr. Matsubara added that recovered COVID-19 patients may be able to participate in sports as some schools reopen. “We are not saying restrict sport participation, but we are merely urging caution.”
Comprehensive LV evaluation needed
The findings reinforce that myocardial involvement is more frequent and sometimes more severe in MIS-C than previously thought, said Kevin G. Friedman, MD, a pediatrician at Harvard Medical School, Boston, and an attending physician in the department of cardiology at Boston Children’s Hospital. “We are underestimating it by using just traditional measures like ejection fraction. It requires a comprehensive evaluation of left ventricular function; it really affects all aspects of the ventricle, both the systolic function and the diastolic function.”
This study supports that MIS-C patients should have a more detailed analysis than EF on echocardiography, including strain imaging. “Probably these patients should all be followed at centers where they can evaluate a more detailed analysis of the LV and RV function,” he said. Patients with ongoing CA enlargement and LV dysfunction should have follow-up cardiac care indefinitely. Patients who have no cardiac symptoms during the acute phase probably don’t need long-term follow-up.
“We’re just trying to learn more about this disease, and it’s certainly concerning that so many kids are having cardiac involvement,” Dr. Friedman said. “Fortunately they’re getting better; we’re just trying to find out what this means for the long term.”
Dr. Matsubara and Dr. Friedman have no relevant financial disclosures.
SOURCE: Matsubara D et al. J Am Coll Cardiol. 2020 Sep 2. doi: 10.1016/j.jacc.2020.08.056.
Patients with multisystem inflammatory syndrome caused by COVID-19 typically seem to avoid coronary artery dilation early on, but they may be prone to cardiac injury and dysfunction longer term that requires a more discerning diagnostic approach to sort out.
The findings were revealed in a study of 28 children with COVID-19–related multisystem inflammatory syndrome (MIS-C) at Children’s Hospital of Philadelphia. The study reported that cardiac injury and dysfunction are common in these patients – even those who have preserved ejection fraction – and that diastolic dysfunction is persistent. For comparison, the study also included 20 healthy controls and 20 patients with classic Kawasaki disease (KD).
The study analyzed echocardiography findings in the patients, reporting left ventricular (LV) systolic and diastolic function were worse than in classic Kawasaki disease (KD), which MIS-C mimics. Lead author Daisuke Matsubara, MD, PhD, and colleagues reported that four markers – LV global longitudinal strain, LV circumferential strain rate, right ventricular strain, and left atrial strain – were the strongest predictors of myocardial injury in these patients. After the acute phase, systolic function tended to recover, but diastolic dysfunction persisted.
‘Strain’ measurement boosts accuracy
While echocardiography has been reported to be valuable in evaluating coronary artery function in MIS-C patients, Dr. Matsubara of the division of cardiology at CHOP, said in an interview that study is the first to use the newer echocardiography indexes, known as “strain,” to assess heart function.
“Strain is a more sensitive tool than more conventional indexes and can detect subtle decrease in heart function, even when ejection fraction is preserved,” he said. “Numerous publications have reached conclusions that strain improves the prognostic and diagnostic accuracy of echocardiography in a wide variety of cardiac pathologies causing LV dysfunction.”
Dr. Matsubara noted that the coronary arteries were mostly unaffected in the acute stage of MIS-C, as only one patient in their MIS-C cohort had coronary artery involvement, which normalized during early follow-up. “On the other hand, 20% of our classic KD patients had coronary abnormalities, including two with aneurysms.”
By using positive troponin I or elevated brain natriuretic peptide (BNP) to assess cardiac injury, they found a “high” (60%) incidence of myocardial injury in their MIS-C cohort. During early follow-up, most of the MIS-C patients showed normalization of systolic function, although diastolic dysfunction persisted.
When compared with the classic KD group, MIS-C patients had higher rates of mitral regurgitation (46% vs. 15%, P = .06), more pericardial effusion (32% vs. 15%, P = 0.46), and more pleural effusion (39% vs. 0%, P = .004). MIS-C patients with suspected myocardial injury show these findings more frequently than those with actual myocardial injury.
Compared with the healthy controls, the MIS-C patients showed both LV systolic and diastolic dysfunction as well as significantly lower left atrium (LA) strain and peak right ventricle (RV) free-wall longitudinal strain.
“In addition to the left ventricle, two other chambers of the heart, the LA and the RV that are often labeled as the ‘forgotten chambers’ of the heart, were also affected by MIS-C,” Dr. Matsubara said. “Both LA and RV strains were markedly reduced in MIS-C patients, compared to normal and KD patients.”
The study also indicates that elevated troponin I levels may not be as dire in children as they are in adults. Dr. Matsubara cited a study of more than 2,700 adult COVID-19 patients that found that even mild increases in troponin I level were associated with increased death during hospitalization (J Am Coll Cardiol. 2020;76:533-46).
However, most of the patients in the CHOP study, even those with elevated troponin I levels, recovered systolic function quickly. “We speculate that the elevation in cardiac troponins may have less dire implications in children, likely due to a more transient type of cardiac injury and less comorbidities in children,” he said. “Clearly further studies are needed before a definitive statement can be made.”
Dr. Matsubara added that recovered COVID-19 patients may be able to participate in sports as some schools reopen. “We are not saying restrict sport participation, but we are merely urging caution.”
Comprehensive LV evaluation needed
The findings reinforce that myocardial involvement is more frequent and sometimes more severe in MIS-C than previously thought, said Kevin G. Friedman, MD, a pediatrician at Harvard Medical School, Boston, and an attending physician in the department of cardiology at Boston Children’s Hospital. “We are underestimating it by using just traditional measures like ejection fraction. It requires a comprehensive evaluation of left ventricular function; it really affects all aspects of the ventricle, both the systolic function and the diastolic function.”
This study supports that MIS-C patients should have a more detailed analysis than EF on echocardiography, including strain imaging. “Probably these patients should all be followed at centers where they can evaluate a more detailed analysis of the LV and RV function,” he said. Patients with ongoing CA enlargement and LV dysfunction should have follow-up cardiac care indefinitely. Patients who have no cardiac symptoms during the acute phase probably don’t need long-term follow-up.
“We’re just trying to learn more about this disease, and it’s certainly concerning that so many kids are having cardiac involvement,” Dr. Friedman said. “Fortunately they’re getting better; we’re just trying to find out what this means for the long term.”
Dr. Matsubara and Dr. Friedman have no relevant financial disclosures.
SOURCE: Matsubara D et al. J Am Coll Cardiol. 2020 Sep 2. doi: 10.1016/j.jacc.2020.08.056.
Patients with multisystem inflammatory syndrome caused by COVID-19 typically seem to avoid coronary artery dilation early on, but they may be prone to cardiac injury and dysfunction longer term that requires a more discerning diagnostic approach to sort out.
The findings were revealed in a study of 28 children with COVID-19–related multisystem inflammatory syndrome (MIS-C) at Children’s Hospital of Philadelphia. The study reported that cardiac injury and dysfunction are common in these patients – even those who have preserved ejection fraction – and that diastolic dysfunction is persistent. For comparison, the study also included 20 healthy controls and 20 patients with classic Kawasaki disease (KD).
The study analyzed echocardiography findings in the patients, reporting left ventricular (LV) systolic and diastolic function were worse than in classic Kawasaki disease (KD), which MIS-C mimics. Lead author Daisuke Matsubara, MD, PhD, and colleagues reported that four markers – LV global longitudinal strain, LV circumferential strain rate, right ventricular strain, and left atrial strain – were the strongest predictors of myocardial injury in these patients. After the acute phase, systolic function tended to recover, but diastolic dysfunction persisted.
‘Strain’ measurement boosts accuracy
While echocardiography has been reported to be valuable in evaluating coronary artery function in MIS-C patients, Dr. Matsubara of the division of cardiology at CHOP, said in an interview that study is the first to use the newer echocardiography indexes, known as “strain,” to assess heart function.
“Strain is a more sensitive tool than more conventional indexes and can detect subtle decrease in heart function, even when ejection fraction is preserved,” he said. “Numerous publications have reached conclusions that strain improves the prognostic and diagnostic accuracy of echocardiography in a wide variety of cardiac pathologies causing LV dysfunction.”
Dr. Matsubara noted that the coronary arteries were mostly unaffected in the acute stage of MIS-C, as only one patient in their MIS-C cohort had coronary artery involvement, which normalized during early follow-up. “On the other hand, 20% of our classic KD patients had coronary abnormalities, including two with aneurysms.”
By using positive troponin I or elevated brain natriuretic peptide (BNP) to assess cardiac injury, they found a “high” (60%) incidence of myocardial injury in their MIS-C cohort. During early follow-up, most of the MIS-C patients showed normalization of systolic function, although diastolic dysfunction persisted.
When compared with the classic KD group, MIS-C patients had higher rates of mitral regurgitation (46% vs. 15%, P = .06), more pericardial effusion (32% vs. 15%, P = 0.46), and more pleural effusion (39% vs. 0%, P = .004). MIS-C patients with suspected myocardial injury show these findings more frequently than those with actual myocardial injury.
Compared with the healthy controls, the MIS-C patients showed both LV systolic and diastolic dysfunction as well as significantly lower left atrium (LA) strain and peak right ventricle (RV) free-wall longitudinal strain.
“In addition to the left ventricle, two other chambers of the heart, the LA and the RV that are often labeled as the ‘forgotten chambers’ of the heart, were also affected by MIS-C,” Dr. Matsubara said. “Both LA and RV strains were markedly reduced in MIS-C patients, compared to normal and KD patients.”
The study also indicates that elevated troponin I levels may not be as dire in children as they are in adults. Dr. Matsubara cited a study of more than 2,700 adult COVID-19 patients that found that even mild increases in troponin I level were associated with increased death during hospitalization (J Am Coll Cardiol. 2020;76:533-46).
However, most of the patients in the CHOP study, even those with elevated troponin I levels, recovered systolic function quickly. “We speculate that the elevation in cardiac troponins may have less dire implications in children, likely due to a more transient type of cardiac injury and less comorbidities in children,” he said. “Clearly further studies are needed before a definitive statement can be made.”
Dr. Matsubara added that recovered COVID-19 patients may be able to participate in sports as some schools reopen. “We are not saying restrict sport participation, but we are merely urging caution.”
Comprehensive LV evaluation needed
The findings reinforce that myocardial involvement is more frequent and sometimes more severe in MIS-C than previously thought, said Kevin G. Friedman, MD, a pediatrician at Harvard Medical School, Boston, and an attending physician in the department of cardiology at Boston Children’s Hospital. “We are underestimating it by using just traditional measures like ejection fraction. It requires a comprehensive evaluation of left ventricular function; it really affects all aspects of the ventricle, both the systolic function and the diastolic function.”
This study supports that MIS-C patients should have a more detailed analysis than EF on echocardiography, including strain imaging. “Probably these patients should all be followed at centers where they can evaluate a more detailed analysis of the LV and RV function,” he said. Patients with ongoing CA enlargement and LV dysfunction should have follow-up cardiac care indefinitely. Patients who have no cardiac symptoms during the acute phase probably don’t need long-term follow-up.
“We’re just trying to learn more about this disease, and it’s certainly concerning that so many kids are having cardiac involvement,” Dr. Friedman said. “Fortunately they’re getting better; we’re just trying to find out what this means for the long term.”
Dr. Matsubara and Dr. Friedman have no relevant financial disclosures.
SOURCE: Matsubara D et al. J Am Coll Cardiol. 2020 Sep 2. doi: 10.1016/j.jacc.2020.08.056.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY