User login
Teen tanning bed ban would prevent more than 15,000 melanoma cases
and cost less than other, well-established public health interventions, according to a microsimulation of that age group’s virtual life course.
“Even with extensive sensitivity analyses on the costs of inspections, noncompliance with a ban, and the risk of developing melanoma in those who have used tanning beds, a ban can be considered highly cost effective,” Antoine Eskander, MD, ScM, of the University of Toronto, and associates said in Cancer.
Compared with no ban, such an intervention could save over $205 million in lifetime health care costs among the 17.1 million young people (based on the 2010 Census population) who would be affected, they said.
The more than 15,000 melanoma cases and 3,300 recurrences prevented would save $12 per average minor after adjusting for societal costs, such as lost productivity, formal and informal health care, economic losses to the tanning bed industry, and the need for monitoring, the investigators reported.
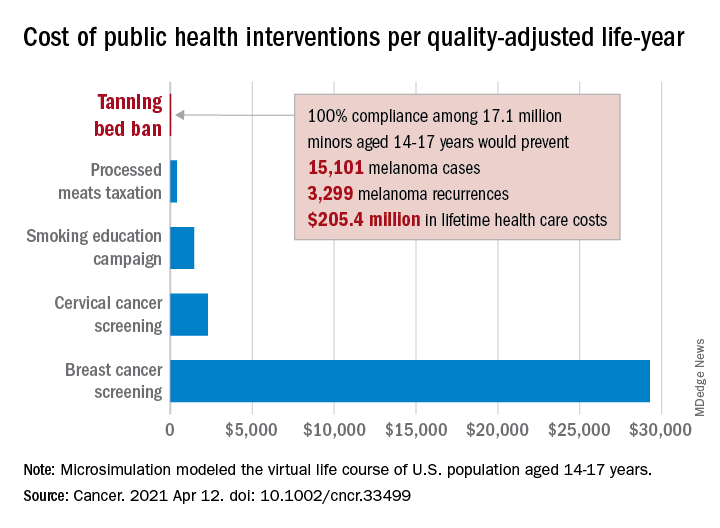
Switching to quality-adjusted life-years shows an improvement of 0.0002 QALYs per child for a ban, based on an overall cost of almost $24.9 per QALY, compared with no ban, they said, which makes it “more cost effective than many well-established public health interventions”:
- Processed meats taxation ($270/QALY).
- Smoking education campaign ($1,337/QALY).
- Cervical cancer screening ($2,166/QALY).
- Breast cancer screening ($29,284/QALY).
- Lung cancer screening ($49,200-$96,700/QALY).
Among the many parameters included in the microsimulation were the odds ratio of developing melanoma from exposure to tanning beds before age 25 (1.35), melanoma stage at presentation, risk of recurrence, and the cost of four annual inspections for each of the nation’s more than 13,000 tanning salons, Dr. Eskander and associates explained.
and cost less than other, well-established public health interventions, according to a microsimulation of that age group’s virtual life course.

“Even with extensive sensitivity analyses on the costs of inspections, noncompliance with a ban, and the risk of developing melanoma in those who have used tanning beds, a ban can be considered highly cost effective,” Antoine Eskander, MD, ScM, of the University of Toronto, and associates said in Cancer.
Compared with no ban, such an intervention could save over $205 million in lifetime health care costs among the 17.1 million young people (based on the 2010 Census population) who would be affected, they said.
The more than 15,000 melanoma cases and 3,300 recurrences prevented would save $12 per average minor after adjusting for societal costs, such as lost productivity, formal and informal health care, economic losses to the tanning bed industry, and the need for monitoring, the investigators reported.
Switching to quality-adjusted life-years shows an improvement of 0.0002 QALYs per child for a ban, based on an overall cost of almost $24.9 per QALY, compared with no ban, they said, which makes it “more cost effective than many well-established public health interventions”:
- Processed meats taxation ($270/QALY).
- Smoking education campaign ($1,337/QALY).
- Cervical cancer screening ($2,166/QALY).
- Breast cancer screening ($29,284/QALY).
- Lung cancer screening ($49,200-$96,700/QALY).
Among the many parameters included in the microsimulation were the odds ratio of developing melanoma from exposure to tanning beds before age 25 (1.35), melanoma stage at presentation, risk of recurrence, and the cost of four annual inspections for each of the nation’s more than 13,000 tanning salons, Dr. Eskander and associates explained.
and cost less than other, well-established public health interventions, according to a microsimulation of that age group’s virtual life course.

“Even with extensive sensitivity analyses on the costs of inspections, noncompliance with a ban, and the risk of developing melanoma in those who have used tanning beds, a ban can be considered highly cost effective,” Antoine Eskander, MD, ScM, of the University of Toronto, and associates said in Cancer.
Compared with no ban, such an intervention could save over $205 million in lifetime health care costs among the 17.1 million young people (based on the 2010 Census population) who would be affected, they said.
The more than 15,000 melanoma cases and 3,300 recurrences prevented would save $12 per average minor after adjusting for societal costs, such as lost productivity, formal and informal health care, economic losses to the tanning bed industry, and the need for monitoring, the investigators reported.
Switching to quality-adjusted life-years shows an improvement of 0.0002 QALYs per child for a ban, based on an overall cost of almost $24.9 per QALY, compared with no ban, they said, which makes it “more cost effective than many well-established public health interventions”:
- Processed meats taxation ($270/QALY).
- Smoking education campaign ($1,337/QALY).
- Cervical cancer screening ($2,166/QALY).
- Breast cancer screening ($29,284/QALY).
- Lung cancer screening ($49,200-$96,700/QALY).
Among the many parameters included in the microsimulation were the odds ratio of developing melanoma from exposure to tanning beds before age 25 (1.35), melanoma stage at presentation, risk of recurrence, and the cost of four annual inspections for each of the nation’s more than 13,000 tanning salons, Dr. Eskander and associates explained.
FROM CANCER
Survey finds Mohs surgeons favor nicotinamide for chemoprevention
, in a survey of members of the American College of Mohs Surgeons.
Although nicotinamide, a vitamin B3 derivative, has been shown to reduce keratinocyte carcinoma (KC) in high-risk patients, it is not approved by the Food and Drug Administration for chemoprevention, and no safe upper limit has been established in clinical trials to date, wrote Sheena Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
The investigators emailed an anonymous 12-question survey to 1,500 members of the American College of Mohs Surgeons. Of the 170 who responded, 10 were excluded for discordant responses, leaving 160 participants whose replies were included in a multiple logistic regression analysis. The respondents were mainly U.S. board-certified dermatologists and Mohs surgeons (99.4% for both); 86.9% were in clinical practice, including 78.8% in private practice, according to the report of the results, published in Dermatologic Surgery.
Overall, 76.9% of the respondents said they recommended nicotinamide for preventing KC, and 20% said they had recommended nicotinamide to more than 100 patients in the past year. In addition, 45% of respondents reported patients who had been taking nicotinamide for 2 years or more. Overall, 63.8% of the respondents expressed no concerns about long-term safety of nicotinamide, compared with 28.1% who said they were uncertain about long-term safety. Those who expressed concern or uncertainty about long-term safety were significantly less likely to recommend nicotinamide for KC prevention in the past year (odds ratio, 0.30; 95% confidence interval [CI] 0.13-0.71). Clinicians with more than 10 years in practice were significantly less likely to recommend nicotinamide for chemoprevention (OR, 0.20; 95% CI 0.05-0.82).
The study findings were limited by several factors, including the low number of responses and the potential lack of generalizability to clinicians other than Mohs surgeons, the researchers noted. “Additional studies on nicotinamide safety and use patterns, including cost-effectiveness analyses, are needed given the widespread use identified in this study,” they concluded.
Limited safety data highlight research gaps
The study is particularly important at this time because nicotinamide has been increasingly used for KC chemoprevention since a randomized, controlled trial published in 2015 in the New England Journal of Medicine showed benefits, corresponding author Rebecca I. Hartman, MD, of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, said in an interview. That study of high-risk patients found that nicotinamide, 500 mg twice a day, was safe and effective in lowering the rates of new nonmelanoma skin cancers and AKs after 12 months .
“However, because this is not a prescription medication, but rather an OTC vitamin supplement, data on its use are not available,” she said.
Dr. Hartman said she was not surprised that nicotinamide is being used frequently by a majority of the survey respondents. “Most are using this if someone has two KCs over 2 years, which is a quite common occurrence,” she noted. However, “I was a bit surprised that nearly two-thirds had no safety concerns with long-term use, even though this has not been well-studied,” she added.
“Like anything we recommend, we must consider the risks and benefits,” Dr. Hartman said of nicotinamide. “Unfortunately, we don’t know the risks well, since this hasn’t been well-characterized with regular long-term use in these doses,” and more research is needed, she said. “The risks are likely low, as this is a vitamin that has been used for years in various OTC supplements,” she added. “However, there are some data showing slightly increased all-cause mortality with similar doses of a related medicine, niacin, in cardiovascular patients. For this reason, I recommend the medication when a patient’s KCs are really becoming burdensome – several KCs in a year or two – or when they are high-risk due to immunosuppression,” she explained.
“We also must consider the individual patient. For a healthy younger patient who has a public-facing job and as a result is very averse to developing any KCs on his or her face and very motivated to try prevention, it may make sense to try nicotinamide,” Dr. Hartman said. But for an older patient with cardiovascular comorbidities who is not bothered by a KC on his or her back or extremities, “this medication may not have a favorable risk-benefit profile.”
To address safety concerns, “researchers need to examine whether there are any harms in long-term regular nicotinamide use for KC prevention,” Dr. Hartman said. “This is something we hope to do in our patients; however, it is challenging to study in a retrospective way since the harm is likely small and there are so many other features that influence mortality as an outcome,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose.
, in a survey of members of the American College of Mohs Surgeons.
Although nicotinamide, a vitamin B3 derivative, has been shown to reduce keratinocyte carcinoma (KC) in high-risk patients, it is not approved by the Food and Drug Administration for chemoprevention, and no safe upper limit has been established in clinical trials to date, wrote Sheena Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
The investigators emailed an anonymous 12-question survey to 1,500 members of the American College of Mohs Surgeons. Of the 170 who responded, 10 were excluded for discordant responses, leaving 160 participants whose replies were included in a multiple logistic regression analysis. The respondents were mainly U.S. board-certified dermatologists and Mohs surgeons (99.4% for both); 86.9% were in clinical practice, including 78.8% in private practice, according to the report of the results, published in Dermatologic Surgery.
Overall, 76.9% of the respondents said they recommended nicotinamide for preventing KC, and 20% said they had recommended nicotinamide to more than 100 patients in the past year. In addition, 45% of respondents reported patients who had been taking nicotinamide for 2 years or more. Overall, 63.8% of the respondents expressed no concerns about long-term safety of nicotinamide, compared with 28.1% who said they were uncertain about long-term safety. Those who expressed concern or uncertainty about long-term safety were significantly less likely to recommend nicotinamide for KC prevention in the past year (odds ratio, 0.30; 95% confidence interval [CI] 0.13-0.71). Clinicians with more than 10 years in practice were significantly less likely to recommend nicotinamide for chemoprevention (OR, 0.20; 95% CI 0.05-0.82).
The study findings were limited by several factors, including the low number of responses and the potential lack of generalizability to clinicians other than Mohs surgeons, the researchers noted. “Additional studies on nicotinamide safety and use patterns, including cost-effectiveness analyses, are needed given the widespread use identified in this study,” they concluded.
Limited safety data highlight research gaps
The study is particularly important at this time because nicotinamide has been increasingly used for KC chemoprevention since a randomized, controlled trial published in 2015 in the New England Journal of Medicine showed benefits, corresponding author Rebecca I. Hartman, MD, of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, said in an interview. That study of high-risk patients found that nicotinamide, 500 mg twice a day, was safe and effective in lowering the rates of new nonmelanoma skin cancers and AKs after 12 months .
“However, because this is not a prescription medication, but rather an OTC vitamin supplement, data on its use are not available,” she said.
Dr. Hartman said she was not surprised that nicotinamide is being used frequently by a majority of the survey respondents. “Most are using this if someone has two KCs over 2 years, which is a quite common occurrence,” she noted. However, “I was a bit surprised that nearly two-thirds had no safety concerns with long-term use, even though this has not been well-studied,” she added.
“Like anything we recommend, we must consider the risks and benefits,” Dr. Hartman said of nicotinamide. “Unfortunately, we don’t know the risks well, since this hasn’t been well-characterized with regular long-term use in these doses,” and more research is needed, she said. “The risks are likely low, as this is a vitamin that has been used for years in various OTC supplements,” she added. “However, there are some data showing slightly increased all-cause mortality with similar doses of a related medicine, niacin, in cardiovascular patients. For this reason, I recommend the medication when a patient’s KCs are really becoming burdensome – several KCs in a year or two – or when they are high-risk due to immunosuppression,” she explained.
“We also must consider the individual patient. For a healthy younger patient who has a public-facing job and as a result is very averse to developing any KCs on his or her face and very motivated to try prevention, it may make sense to try nicotinamide,” Dr. Hartman said. But for an older patient with cardiovascular comorbidities who is not bothered by a KC on his or her back or extremities, “this medication may not have a favorable risk-benefit profile.”
To address safety concerns, “researchers need to examine whether there are any harms in long-term regular nicotinamide use for KC prevention,” Dr. Hartman said. “This is something we hope to do in our patients; however, it is challenging to study in a retrospective way since the harm is likely small and there are so many other features that influence mortality as an outcome,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose.
, in a survey of members of the American College of Mohs Surgeons.
Although nicotinamide, a vitamin B3 derivative, has been shown to reduce keratinocyte carcinoma (KC) in high-risk patients, it is not approved by the Food and Drug Administration for chemoprevention, and no safe upper limit has been established in clinical trials to date, wrote Sheena Desai of Brigham and Women’s Hospital and Harvard Medical School, Boston, and colleagues.
The investigators emailed an anonymous 12-question survey to 1,500 members of the American College of Mohs Surgeons. Of the 170 who responded, 10 were excluded for discordant responses, leaving 160 participants whose replies were included in a multiple logistic regression analysis. The respondents were mainly U.S. board-certified dermatologists and Mohs surgeons (99.4% for both); 86.9% were in clinical practice, including 78.8% in private practice, according to the report of the results, published in Dermatologic Surgery.
Overall, 76.9% of the respondents said they recommended nicotinamide for preventing KC, and 20% said they had recommended nicotinamide to more than 100 patients in the past year. In addition, 45% of respondents reported patients who had been taking nicotinamide for 2 years or more. Overall, 63.8% of the respondents expressed no concerns about long-term safety of nicotinamide, compared with 28.1% who said they were uncertain about long-term safety. Those who expressed concern or uncertainty about long-term safety were significantly less likely to recommend nicotinamide for KC prevention in the past year (odds ratio, 0.30; 95% confidence interval [CI] 0.13-0.71). Clinicians with more than 10 years in practice were significantly less likely to recommend nicotinamide for chemoprevention (OR, 0.20; 95% CI 0.05-0.82).
The study findings were limited by several factors, including the low number of responses and the potential lack of generalizability to clinicians other than Mohs surgeons, the researchers noted. “Additional studies on nicotinamide safety and use patterns, including cost-effectiveness analyses, are needed given the widespread use identified in this study,” they concluded.
Limited safety data highlight research gaps
The study is particularly important at this time because nicotinamide has been increasingly used for KC chemoprevention since a randomized, controlled trial published in 2015 in the New England Journal of Medicine showed benefits, corresponding author Rebecca I. Hartman, MD, of the department of dermatology, Brigham and Women’s Hospital and Harvard University, Boston, said in an interview. That study of high-risk patients found that nicotinamide, 500 mg twice a day, was safe and effective in lowering the rates of new nonmelanoma skin cancers and AKs after 12 months .
“However, because this is not a prescription medication, but rather an OTC vitamin supplement, data on its use are not available,” she said.
Dr. Hartman said she was not surprised that nicotinamide is being used frequently by a majority of the survey respondents. “Most are using this if someone has two KCs over 2 years, which is a quite common occurrence,” she noted. However, “I was a bit surprised that nearly two-thirds had no safety concerns with long-term use, even though this has not been well-studied,” she added.
“Like anything we recommend, we must consider the risks and benefits,” Dr. Hartman said of nicotinamide. “Unfortunately, we don’t know the risks well, since this hasn’t been well-characterized with regular long-term use in these doses,” and more research is needed, she said. “The risks are likely low, as this is a vitamin that has been used for years in various OTC supplements,” she added. “However, there are some data showing slightly increased all-cause mortality with similar doses of a related medicine, niacin, in cardiovascular patients. For this reason, I recommend the medication when a patient’s KCs are really becoming burdensome – several KCs in a year or two – or when they are high-risk due to immunosuppression,” she explained.
“We also must consider the individual patient. For a healthy younger patient who has a public-facing job and as a result is very averse to developing any KCs on his or her face and very motivated to try prevention, it may make sense to try nicotinamide,” Dr. Hartman said. But for an older patient with cardiovascular comorbidities who is not bothered by a KC on his or her back or extremities, “this medication may not have a favorable risk-benefit profile.”
To address safety concerns, “researchers need to examine whether there are any harms in long-term regular nicotinamide use for KC prevention,” Dr. Hartman said. “This is something we hope to do in our patients; however, it is challenging to study in a retrospective way since the harm is likely small and there are so many other features that influence mortality as an outcome,” she noted.
The study received no outside funding. The researchers had no financial conflicts to disclose.
FROM DERMATOLOGIC SURGERY
A Rash Against the Grain
ANSWER
The correct answer is dermatitis herpetiformis (DH; choice “b”).
DISCUSSION
DH is unusual, although not rare, and it is often overlooked in the investigation of itchy rashes—even by dermatologists. As is so often the case, the biopsy was the missing piece of the puzzle, since it clearly showed changes consistent with this condition.
The history of DH has some interesting overlap with that of biopsy. It was Rudolph Virchow, an 18th century Austrian pathologist, who first connected clinical disease to specific arrangements of disordered tissue seen microscopically—at that time, a giant leap for medicine. An American dermatologist, Louis Adolphus Duhring, had the good fortune to study under Virchow and his successors in Vienna and brought this cutting-edge knowledge back to this country, where he began the process of categorizing diseases by their histologic patterns as well as by their presentation and clinical course. This was when he discovered the basis for the condition that became known as Duhring disease, also known as dermatitis herpetiformis.
It was much later (1967) that a connection was made between DH and the ingestion of gluten. Our patient experienced a rapid decline in his itching and rash as soon as he started a gluten-free diet. However, since many patients find such a diet difficult to maintain, there are pharmacologic options as well: dapsone, colchicine, sulfa drugs, drugs from the tetracycline family, and nicotinamide. All have potential adverse effects with which the prescriber needs to become familiar.
With a combination of medication and reduced gluten intake, it’s entirely possible that the patient’s DH will permanently resolve, although he will remain at increased risk for other autoimmune conditions.
ANSWER
The correct answer is dermatitis herpetiformis (DH; choice “b”).
DISCUSSION
DH is unusual, although not rare, and it is often overlooked in the investigation of itchy rashes—even by dermatologists. As is so often the case, the biopsy was the missing piece of the puzzle, since it clearly showed changes consistent with this condition.
The history of DH has some interesting overlap with that of biopsy. It was Rudolph Virchow, an 18th century Austrian pathologist, who first connected clinical disease to specific arrangements of disordered tissue seen microscopically—at that time, a giant leap for medicine. An American dermatologist, Louis Adolphus Duhring, had the good fortune to study under Virchow and his successors in Vienna and brought this cutting-edge knowledge back to this country, where he began the process of categorizing diseases by their histologic patterns as well as by their presentation and clinical course. This was when he discovered the basis for the condition that became known as Duhring disease, also known as dermatitis herpetiformis.
It was much later (1967) that a connection was made between DH and the ingestion of gluten. Our patient experienced a rapid decline in his itching and rash as soon as he started a gluten-free diet. However, since many patients find such a diet difficult to maintain, there are pharmacologic options as well: dapsone, colchicine, sulfa drugs, drugs from the tetracycline family, and nicotinamide. All have potential adverse effects with which the prescriber needs to become familiar.
With a combination of medication and reduced gluten intake, it’s entirely possible that the patient’s DH will permanently resolve, although he will remain at increased risk for other autoimmune conditions.
ANSWER
The correct answer is dermatitis herpetiformis (DH; choice “b”).
DISCUSSION
DH is unusual, although not rare, and it is often overlooked in the investigation of itchy rashes—even by dermatologists. As is so often the case, the biopsy was the missing piece of the puzzle, since it clearly showed changes consistent with this condition.
The history of DH has some interesting overlap with that of biopsy. It was Rudolph Virchow, an 18th century Austrian pathologist, who first connected clinical disease to specific arrangements of disordered tissue seen microscopically—at that time, a giant leap for medicine. An American dermatologist, Louis Adolphus Duhring, had the good fortune to study under Virchow and his successors in Vienna and brought this cutting-edge knowledge back to this country, where he began the process of categorizing diseases by their histologic patterns as well as by their presentation and clinical course. This was when he discovered the basis for the condition that became known as Duhring disease, also known as dermatitis herpetiformis.
It was much later (1967) that a connection was made between DH and the ingestion of gluten. Our patient experienced a rapid decline in his itching and rash as soon as he started a gluten-free diet. However, since many patients find such a diet difficult to maintain, there are pharmacologic options as well: dapsone, colchicine, sulfa drugs, drugs from the tetracycline family, and nicotinamide. All have potential adverse effects with which the prescriber needs to become familiar.
With a combination of medication and reduced gluten intake, it’s entirely possible that the patient’s DH will permanently resolve, although he will remain at increased risk for other autoimmune conditions.
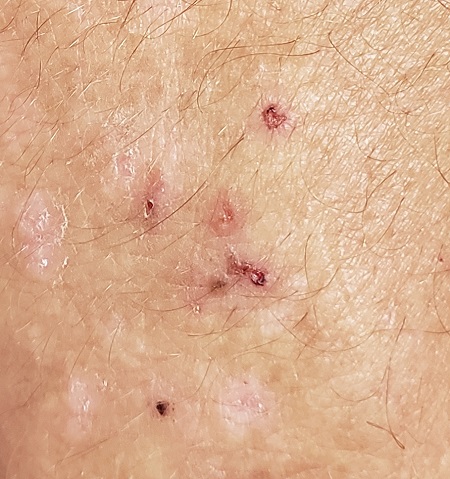
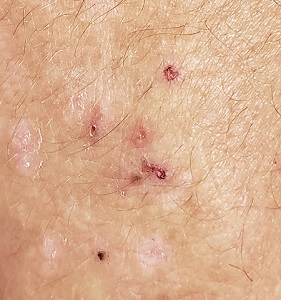
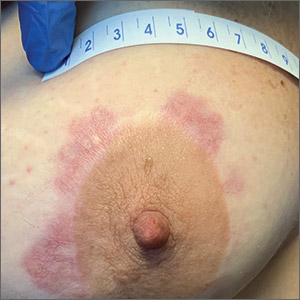
A 56-year-old man began to itch almost 10 years ago, when lesions appeared on his extensor forearms; they later branched out to his knees, scalp, and waistline. He has continued to suffer despite seeing numerous providers, including dermatologists.
An allergist pronounced him free of any significant allergies. Another provider was certain the patient had scabies, although his household was unaffected and the prescribed treatment—permethrin lotion and oral ivermectin—had no impact on the rash or the symptoms. Most of the other consulted providers diagnosed eczema, despite a complete lack of atopy in the patient or his family of origin. Furthermore, treatment with topical, oral, and intramuscular steroids had offered minimal and very short-lived relief. No one took a scraping or performed a biopsy of the rash.
The patient claims to be otherwise healthy, with no gastrointestinal symptoms and no new medications. Examination reveals a slightly overweight man in no distress.
The lesions in question are patches of excoriated papulovesicular lesions. Microscopic examination of a KOH prep shows no scabetic elements. His volar wrists, the sides of his fingers, and his genitals are free of lesions.
A shave biopsy is performed under local anesthetic. The pathology report indicates subepidermal collections of eosinophils.
Study aims to enhance understanding of ‘tremendously understudied’ prurigo nodularis
compared with age-matched controls, as well those with atopic dermatitis and psoriasis.
Those are key findings from a retrospective analysis of claims data that was published online April 3, 2021, in the Journal of Investigative Dermatology.
“Prurigo nodularis is a tremendously understudied inflammatory skin disease,” one of the study’s cosenior authors, Shawn G. Kwatra, MD, of the department of dermatology, Johns Hopkins University, Baltimore, said in an interview. “Prurigo nodularis patients have uncontrolled itch, which leads to reduced quality of life, and the association with many disease comorbidities. We focused on better understanding in this work the unique comorbidities of prurigo nodularis, compared to other inflammatory skin diseases.”
For the study, Dr. Kwatra, cosenior author Yevgeniy R. Semenov, MD, of the department of dermatology, Massachusetts General Hospital, Boston, and colleagues evaluated nationally representative, private insurance claims data from October 2015 to December 2019 to identify prurigo nodularis (PN) patients, who were defined as individuals with two or more medical claims for PN using ICD-10-CM codes. For comparison with patients with inflammatory skin diseases, they used the same claims data to identify patients with atopic dermatitis (AD) and psoriasis as well as to select controls who were age and gender matched to PN patients. Next, they quantified the overall comorbidity burden with the Charlson Comorbidity Index (CCI).
In 2016, the claims database included 2,658 patients with PN, 21,482 patients with AD, 21,073 patients with psoriasis, and 13,290 controls. The number of patients in each category rose each subsequent year, so that by the end of 2019 there were 9,426 patients with PN, 70,298 patients with AD, 59,509 patients with psoriasis, and 47,130 controls. Between 2016 and 2019 the mean age of PN patients increased from 57.5 to 59.8 years and the percent of male patients rose from 44.5% to 46.5%.
Between 2016 and 2019, the overall PN prevalence rates rose from 18 per 100,000 to 58 per 100,000, while the PN prevalence rates among adults increased from 22 per 100,000 to 70 per 100,000, and the rates among children rose grew from 2 per 100,000 to 7 per 100,000. “Our report shows an estimated disease prevalence of around 335,000 cases of PN in the United States,” said Dr. Kwatra, who was among a group of researchers to recently report on systemic Th22-polarized inflammation in PN patients.
The researchers also found that patients with PN had the highest mean CCI in both 2016 and 2019. In 2016, their mean CCI was 1.53, compared with 0.98 among controls, 0.53 among those with AD, and 1.16 among those with psoriasis. In 2019, the mean CCI had increased in all groups of patients, to 2.32 among those with PN, 1.57 among controls, 0.75 among those with AD patients, and 1.71 among those with psoriasis.
The top five medical specialties who cared for PN patients, defined as the estimated number of visits per year per patient, were internal medicine (2.01 visits), dermatology (1.87 visits), family practice (1.60 visits), cardiology or cardiovascular disease (0.85 visits), and orthopedics or orthopedic surgery (0.49 visits).
“If you encounter a patient with prurigo nodularis, it’s important to perform a screening for chronic kidney disease, diabetes, and liver disease,” Dr. Kwatra said. “These comorbidities along with emerging studies on circulating blood biomarkers suggest prurigo nodularis is a systemic inflammatory disorder; thus systemic agents are needed for most patients as part of multimodal therapy in prurigo nodularis.”
The researchers acknowledged certain limitations of the study, including its retrospective design and the identification of patients with PN with the ICD-10-CM code, which require further validation. “Furthermore, the increase in annual prevalence estimates for PN, AD, and psoriasis observed in the study could also be a result of increasing coding of these diagnoses in the claims data along with rising awareness by the medical profession,” they wrote.
Dr. Kwatra disclosed that he is an advisory board member/consultant for AbbVie, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals, and has received grant funding from Galderma, Pfizer, and Kiniksa. He has also received a Dermatology Foundation Medical Dermatology Career Development Award, a research grant from the Skin of Color Society, and is supported by the National Institutes of Health. One coauthor has been funded by NIH grants.
compared with age-matched controls, as well those with atopic dermatitis and psoriasis.
Those are key findings from a retrospective analysis of claims data that was published online April 3, 2021, in the Journal of Investigative Dermatology.
“Prurigo nodularis is a tremendously understudied inflammatory skin disease,” one of the study’s cosenior authors, Shawn G. Kwatra, MD, of the department of dermatology, Johns Hopkins University, Baltimore, said in an interview. “Prurigo nodularis patients have uncontrolled itch, which leads to reduced quality of life, and the association with many disease comorbidities. We focused on better understanding in this work the unique comorbidities of prurigo nodularis, compared to other inflammatory skin diseases.”
For the study, Dr. Kwatra, cosenior author Yevgeniy R. Semenov, MD, of the department of dermatology, Massachusetts General Hospital, Boston, and colleagues evaluated nationally representative, private insurance claims data from October 2015 to December 2019 to identify prurigo nodularis (PN) patients, who were defined as individuals with two or more medical claims for PN using ICD-10-CM codes. For comparison with patients with inflammatory skin diseases, they used the same claims data to identify patients with atopic dermatitis (AD) and psoriasis as well as to select controls who were age and gender matched to PN patients. Next, they quantified the overall comorbidity burden with the Charlson Comorbidity Index (CCI).
In 2016, the claims database included 2,658 patients with PN, 21,482 patients with AD, 21,073 patients with psoriasis, and 13,290 controls. The number of patients in each category rose each subsequent year, so that by the end of 2019 there were 9,426 patients with PN, 70,298 patients with AD, 59,509 patients with psoriasis, and 47,130 controls. Between 2016 and 2019 the mean age of PN patients increased from 57.5 to 59.8 years and the percent of male patients rose from 44.5% to 46.5%.
Between 2016 and 2019, the overall PN prevalence rates rose from 18 per 100,000 to 58 per 100,000, while the PN prevalence rates among adults increased from 22 per 100,000 to 70 per 100,000, and the rates among children rose grew from 2 per 100,000 to 7 per 100,000. “Our report shows an estimated disease prevalence of around 335,000 cases of PN in the United States,” said Dr. Kwatra, who was among a group of researchers to recently report on systemic Th22-polarized inflammation in PN patients.
The researchers also found that patients with PN had the highest mean CCI in both 2016 and 2019. In 2016, their mean CCI was 1.53, compared with 0.98 among controls, 0.53 among those with AD, and 1.16 among those with psoriasis. In 2019, the mean CCI had increased in all groups of patients, to 2.32 among those with PN, 1.57 among controls, 0.75 among those with AD patients, and 1.71 among those with psoriasis.
The top five medical specialties who cared for PN patients, defined as the estimated number of visits per year per patient, were internal medicine (2.01 visits), dermatology (1.87 visits), family practice (1.60 visits), cardiology or cardiovascular disease (0.85 visits), and orthopedics or orthopedic surgery (0.49 visits).
“If you encounter a patient with prurigo nodularis, it’s important to perform a screening for chronic kidney disease, diabetes, and liver disease,” Dr. Kwatra said. “These comorbidities along with emerging studies on circulating blood biomarkers suggest prurigo nodularis is a systemic inflammatory disorder; thus systemic agents are needed for most patients as part of multimodal therapy in prurigo nodularis.”
The researchers acknowledged certain limitations of the study, including its retrospective design and the identification of patients with PN with the ICD-10-CM code, which require further validation. “Furthermore, the increase in annual prevalence estimates for PN, AD, and psoriasis observed in the study could also be a result of increasing coding of these diagnoses in the claims data along with rising awareness by the medical profession,” they wrote.
Dr. Kwatra disclosed that he is an advisory board member/consultant for AbbVie, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals, and has received grant funding from Galderma, Pfizer, and Kiniksa. He has also received a Dermatology Foundation Medical Dermatology Career Development Award, a research grant from the Skin of Color Society, and is supported by the National Institutes of Health. One coauthor has been funded by NIH grants.
compared with age-matched controls, as well those with atopic dermatitis and psoriasis.
Those are key findings from a retrospective analysis of claims data that was published online April 3, 2021, in the Journal of Investigative Dermatology.
“Prurigo nodularis is a tremendously understudied inflammatory skin disease,” one of the study’s cosenior authors, Shawn G. Kwatra, MD, of the department of dermatology, Johns Hopkins University, Baltimore, said in an interview. “Prurigo nodularis patients have uncontrolled itch, which leads to reduced quality of life, and the association with many disease comorbidities. We focused on better understanding in this work the unique comorbidities of prurigo nodularis, compared to other inflammatory skin diseases.”
For the study, Dr. Kwatra, cosenior author Yevgeniy R. Semenov, MD, of the department of dermatology, Massachusetts General Hospital, Boston, and colleagues evaluated nationally representative, private insurance claims data from October 2015 to December 2019 to identify prurigo nodularis (PN) patients, who were defined as individuals with two or more medical claims for PN using ICD-10-CM codes. For comparison with patients with inflammatory skin diseases, they used the same claims data to identify patients with atopic dermatitis (AD) and psoriasis as well as to select controls who were age and gender matched to PN patients. Next, they quantified the overall comorbidity burden with the Charlson Comorbidity Index (CCI).
In 2016, the claims database included 2,658 patients with PN, 21,482 patients with AD, 21,073 patients with psoriasis, and 13,290 controls. The number of patients in each category rose each subsequent year, so that by the end of 2019 there were 9,426 patients with PN, 70,298 patients with AD, 59,509 patients with psoriasis, and 47,130 controls. Between 2016 and 2019 the mean age of PN patients increased from 57.5 to 59.8 years and the percent of male patients rose from 44.5% to 46.5%.
Between 2016 and 2019, the overall PN prevalence rates rose from 18 per 100,000 to 58 per 100,000, while the PN prevalence rates among adults increased from 22 per 100,000 to 70 per 100,000, and the rates among children rose grew from 2 per 100,000 to 7 per 100,000. “Our report shows an estimated disease prevalence of around 335,000 cases of PN in the United States,” said Dr. Kwatra, who was among a group of researchers to recently report on systemic Th22-polarized inflammation in PN patients.
The researchers also found that patients with PN had the highest mean CCI in both 2016 and 2019. In 2016, their mean CCI was 1.53, compared with 0.98 among controls, 0.53 among those with AD, and 1.16 among those with psoriasis. In 2019, the mean CCI had increased in all groups of patients, to 2.32 among those with PN, 1.57 among controls, 0.75 among those with AD patients, and 1.71 among those with psoriasis.
The top five medical specialties who cared for PN patients, defined as the estimated number of visits per year per patient, were internal medicine (2.01 visits), dermatology (1.87 visits), family practice (1.60 visits), cardiology or cardiovascular disease (0.85 visits), and orthopedics or orthopedic surgery (0.49 visits).
“If you encounter a patient with prurigo nodularis, it’s important to perform a screening for chronic kidney disease, diabetes, and liver disease,” Dr. Kwatra said. “These comorbidities along with emerging studies on circulating blood biomarkers suggest prurigo nodularis is a systemic inflammatory disorder; thus systemic agents are needed for most patients as part of multimodal therapy in prurigo nodularis.”
The researchers acknowledged certain limitations of the study, including its retrospective design and the identification of patients with PN with the ICD-10-CM code, which require further validation. “Furthermore, the increase in annual prevalence estimates for PN, AD, and psoriasis observed in the study could also be a result of increasing coding of these diagnoses in the claims data along with rising awareness by the medical profession,” they wrote.
Dr. Kwatra disclosed that he is an advisory board member/consultant for AbbVie, Galderma, Incyte, Pfizer, Regeneron, and Kiniksa Pharmaceuticals, and has received grant funding from Galderma, Pfizer, and Kiniksa. He has also received a Dermatology Foundation Medical Dermatology Career Development Award, a research grant from the Skin of Color Society, and is supported by the National Institutes of Health. One coauthor has been funded by NIH grants.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
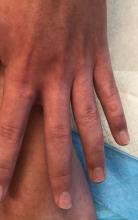
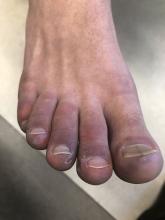
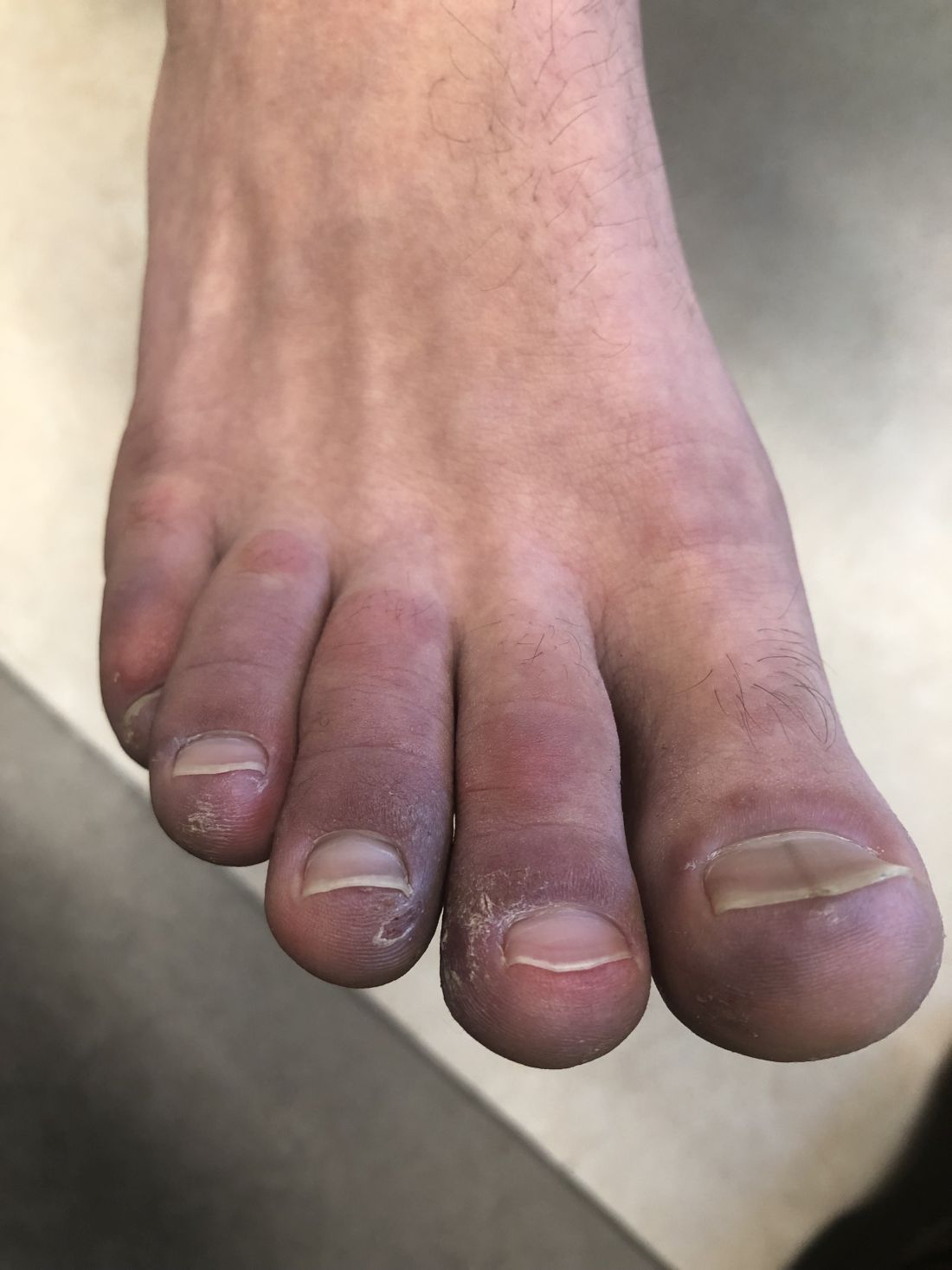
A 12-year-old male has persistent purple toes and new red lesions on his hands
A punch biopsy from one of the lesions on the feet showed subtle basal vacuolar interface inflammation on the epidermis and rare apoptotic keratinocytes. There was an underlying dermal lymphocytic inflammatory infiltrate around the vascular plexus. Dermal mucin appeared slightly increased. The histologic findings are consistent with pernio. He had a negative direct immunofluorescence study.
Laboratory work-up showed an elevated antinuclear antibody (ANA) of 1:620; positive anticardiolipin IgM was at 15.2. A complete blood count showed no anemia or lymphopenia, he had normal complement C3 and C4 levels, normal urinalysis, negative cryoglobulins and cold agglutinins, and a normal protein electrophoresis.
Given the chronicity of his lesions, the lack of improvement with weather changes, the histopathologic findings of a vacuolar interface dermatitis and the positive ANA titer he was diagnosed with chilblain lupus.
Chilblain lupus erythematosus (CLE) is an uncommon form of chronic cutaneous lupus erythematosus that presents with tender pink to violaceous macules, papules, and/or nodules that sometimes can ulcerate and are present on the fingers, toes, and sometimes the nose and ears. The lesions are usually triggered by cold exposure.1 These patients also have clinical and laboratory findings consistent with lupus erythematosus.
Even though more studies are needed to clarify the clinical and histopathologic features of chilblain lupus, compared with idiopathic pernio, some authors suggest several characteristics: CLE lesions tend to persist in summer months, as occurred in our patient, and histopathologic evaluation usually shows vacuolar and interface inflammation on the basal cell layer and may also have a positive lupus band on direct immunofluorescence.2 About 20% of patient with CLE may later develop systemic lupus erythematosus.3
There is also a familial form of CLE which is usually inherited as an autosomal-dominant trait. Mutations in TREX1, SAMHD1, and STING have been described in these patients.4 Affected children present with skin lesions at a young age and those with TREX1 mutations are at a higher risk to develop systemic lupus erythematosus.
The differential diagnosis of chilblain lupus includes idiopathic pernio or pernio secondary to other conditions. Other conditions that are thought to be associated with pernio, besides lupus erythematosus, include infectious causes (hepatitis B, COVID-19 infection),5 autoimmune conditions, malignancy and hematologic disorders (paraproteinemia).6 In histopathology, pernio lesions present with dermal edema and superficial and deep lymphocytic infiltrate.
The pathogenesis of pernio is not fully understood but is thought be related to vasospasm with secondary poor perfusion and ischemia and type I interferon (INF1) immune response. A recent review of the published studies trying to explain the causality between COVID 19 and pernio-like lesions, from January 2020 to December 2020, speculate several possible mechanisms: an increase in the vasoconstrictive, prothrombotic, and proinflammatory effects of the angiotensin II pathway through activation of the ACE2 by the virus; COVID-19 triggers a robust INF1 immune response in predisposed patients; pernio as a sign of mild disease, may be explained by genetic and hormonal differences in the patients affected.7
Another condition that can be confused with CLE is Raynaud phenomenon, were patients present with white to purple to red patches on the fingers and toes after exposure to cold, but in comparison with pernio, the lesions improve within minutes to hours after rewarming. Secondary Raynaud phenomenon can be seen in patients with systemic lupus erythematosus and in patients with other connective tissue disorders. The skin lesions in our patient were persistent and were not triggered by cold exposure, making Raynaud phenomenon less likely. Children with vasculitis can present with painful red, violaceous, or necrotic lesions on the extremities, which can mimic pernio. Vasculitis lesions tend to be more purpuric and angulated, compared with pernio lesions, though in severe cases of pernio with ulceration it may be difficult to distinguish between the two entities and a skin biopsy may be needed.
Sweet syndrome, also known as acute febrile neutrophilic dermatosis, is a rare skin condition in which children present with edematous tender nodules on the hands and with less frequency in other parts of the body with associated fever, malaise, conjunctivitis, or joint pain and it is usually associated with infection or malignancy. Our patient denied any systemic symptoms and had no conjunctivitis nor arthritis.
Most patients with idiopathic pernio do not require a biopsy or further laboratory evaluation unless the lesions are atypical, chronic, or there is a suspected associated condition. The workup for patients with prolonged or atypical pernio-like lesions include a skin biopsy with direct immunofluorescence, ANA, complete blood count, complement levels, antiphospholipid antibodies, cold agglutinins, and cryoglobulins.
Treatment of mild CLE is with moderate- to high-potency topical corticosteroids. In those patients not responding to topical measures and keeping the extremities warm, the use of hydroxychloroquine has been reported to be beneficial in some patients as well as the use of calcium-channel blockers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Su WP et al. Cutis. 1994 Dec;54(6):395-9.
2. Boada A et al. Am J Dermatopathol. 2010 Feb;32(1):19-23.
3. Patel et al. SBMJ Case Rep. 2013;2013:bcr2013201165.
4. Genes Yi et al. BMC. 2020 Apr 15;18(1):32.
5. Battesti G et al. J Am Acad Dermatol. 2020;83(4):1219-22.
6. Cappel JA et al. Mayo Clin Proc. 2014 Feb;89(2):207-15.
7. Cappel MA et al. Mayo Clin Proc. 2021;96(4):989-1005.
A punch biopsy from one of the lesions on the feet showed subtle basal vacuolar interface inflammation on the epidermis and rare apoptotic keratinocytes. There was an underlying dermal lymphocytic inflammatory infiltrate around the vascular plexus. Dermal mucin appeared slightly increased. The histologic findings are consistent with pernio. He had a negative direct immunofluorescence study.
Laboratory work-up showed an elevated antinuclear antibody (ANA) of 1:620; positive anticardiolipin IgM was at 15.2. A complete blood count showed no anemia or lymphopenia, he had normal complement C3 and C4 levels, normal urinalysis, negative cryoglobulins and cold agglutinins, and a normal protein electrophoresis.
Given the chronicity of his lesions, the lack of improvement with weather changes, the histopathologic findings of a vacuolar interface dermatitis and the positive ANA titer he was diagnosed with chilblain lupus.
Chilblain lupus erythematosus (CLE) is an uncommon form of chronic cutaneous lupus erythematosus that presents with tender pink to violaceous macules, papules, and/or nodules that sometimes can ulcerate and are present on the fingers, toes, and sometimes the nose and ears. The lesions are usually triggered by cold exposure.1 These patients also have clinical and laboratory findings consistent with lupus erythematosus.
Even though more studies are needed to clarify the clinical and histopathologic features of chilblain lupus, compared with idiopathic pernio, some authors suggest several characteristics: CLE lesions tend to persist in summer months, as occurred in our patient, and histopathologic evaluation usually shows vacuolar and interface inflammation on the basal cell layer and may also have a positive lupus band on direct immunofluorescence.2 About 20% of patient with CLE may later develop systemic lupus erythematosus.3
There is also a familial form of CLE which is usually inherited as an autosomal-dominant trait. Mutations in TREX1, SAMHD1, and STING have been described in these patients.4 Affected children present with skin lesions at a young age and those with TREX1 mutations are at a higher risk to develop systemic lupus erythematosus.
The differential diagnosis of chilblain lupus includes idiopathic pernio or pernio secondary to other conditions. Other conditions that are thought to be associated with pernio, besides lupus erythematosus, include infectious causes (hepatitis B, COVID-19 infection),5 autoimmune conditions, malignancy and hematologic disorders (paraproteinemia).6 In histopathology, pernio lesions present with dermal edema and superficial and deep lymphocytic infiltrate.
The pathogenesis of pernio is not fully understood but is thought be related to vasospasm with secondary poor perfusion and ischemia and type I interferon (INF1) immune response. A recent review of the published studies trying to explain the causality between COVID 19 and pernio-like lesions, from January 2020 to December 2020, speculate several possible mechanisms: an increase in the vasoconstrictive, prothrombotic, and proinflammatory effects of the angiotensin II pathway through activation of the ACE2 by the virus; COVID-19 triggers a robust INF1 immune response in predisposed patients; pernio as a sign of mild disease, may be explained by genetic and hormonal differences in the patients affected.7
Another condition that can be confused with CLE is Raynaud phenomenon, were patients present with white to purple to red patches on the fingers and toes after exposure to cold, but in comparison with pernio, the lesions improve within minutes to hours after rewarming. Secondary Raynaud phenomenon can be seen in patients with systemic lupus erythematosus and in patients with other connective tissue disorders. The skin lesions in our patient were persistent and were not triggered by cold exposure, making Raynaud phenomenon less likely. Children with vasculitis can present with painful red, violaceous, or necrotic lesions on the extremities, which can mimic pernio. Vasculitis lesions tend to be more purpuric and angulated, compared with pernio lesions, though in severe cases of pernio with ulceration it may be difficult to distinguish between the two entities and a skin biopsy may be needed.
Sweet syndrome, also known as acute febrile neutrophilic dermatosis, is a rare skin condition in which children present with edematous tender nodules on the hands and with less frequency in other parts of the body with associated fever, malaise, conjunctivitis, or joint pain and it is usually associated with infection or malignancy. Our patient denied any systemic symptoms and had no conjunctivitis nor arthritis.
Most patients with idiopathic pernio do not require a biopsy or further laboratory evaluation unless the lesions are atypical, chronic, or there is a suspected associated condition. The workup for patients with prolonged or atypical pernio-like lesions include a skin biopsy with direct immunofluorescence, ANA, complete blood count, complement levels, antiphospholipid antibodies, cold agglutinins, and cryoglobulins.
Treatment of mild CLE is with moderate- to high-potency topical corticosteroids. In those patients not responding to topical measures and keeping the extremities warm, the use of hydroxychloroquine has been reported to be beneficial in some patients as well as the use of calcium-channel blockers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Su WP et al. Cutis. 1994 Dec;54(6):395-9.
2. Boada A et al. Am J Dermatopathol. 2010 Feb;32(1):19-23.
3. Patel et al. SBMJ Case Rep. 2013;2013:bcr2013201165.
4. Genes Yi et al. BMC. 2020 Apr 15;18(1):32.
5. Battesti G et al. J Am Acad Dermatol. 2020;83(4):1219-22.
6. Cappel JA et al. Mayo Clin Proc. 2014 Feb;89(2):207-15.
7. Cappel MA et al. Mayo Clin Proc. 2021;96(4):989-1005.
A punch biopsy from one of the lesions on the feet showed subtle basal vacuolar interface inflammation on the epidermis and rare apoptotic keratinocytes. There was an underlying dermal lymphocytic inflammatory infiltrate around the vascular plexus. Dermal mucin appeared slightly increased. The histologic findings are consistent with pernio. He had a negative direct immunofluorescence study.
Laboratory work-up showed an elevated antinuclear antibody (ANA) of 1:620; positive anticardiolipin IgM was at 15.2. A complete blood count showed no anemia or lymphopenia, he had normal complement C3 and C4 levels, normal urinalysis, negative cryoglobulins and cold agglutinins, and a normal protein electrophoresis.
Given the chronicity of his lesions, the lack of improvement with weather changes, the histopathologic findings of a vacuolar interface dermatitis and the positive ANA titer he was diagnosed with chilblain lupus.
Chilblain lupus erythematosus (CLE) is an uncommon form of chronic cutaneous lupus erythematosus that presents with tender pink to violaceous macules, papules, and/or nodules that sometimes can ulcerate and are present on the fingers, toes, and sometimes the nose and ears. The lesions are usually triggered by cold exposure.1 These patients also have clinical and laboratory findings consistent with lupus erythematosus.
Even though more studies are needed to clarify the clinical and histopathologic features of chilblain lupus, compared with idiopathic pernio, some authors suggest several characteristics: CLE lesions tend to persist in summer months, as occurred in our patient, and histopathologic evaluation usually shows vacuolar and interface inflammation on the basal cell layer and may also have a positive lupus band on direct immunofluorescence.2 About 20% of patient with CLE may later develop systemic lupus erythematosus.3
There is also a familial form of CLE which is usually inherited as an autosomal-dominant trait. Mutations in TREX1, SAMHD1, and STING have been described in these patients.4 Affected children present with skin lesions at a young age and those with TREX1 mutations are at a higher risk to develop systemic lupus erythematosus.
The differential diagnosis of chilblain lupus includes idiopathic pernio or pernio secondary to other conditions. Other conditions that are thought to be associated with pernio, besides lupus erythematosus, include infectious causes (hepatitis B, COVID-19 infection),5 autoimmune conditions, malignancy and hematologic disorders (paraproteinemia).6 In histopathology, pernio lesions present with dermal edema and superficial and deep lymphocytic infiltrate.
The pathogenesis of pernio is not fully understood but is thought be related to vasospasm with secondary poor perfusion and ischemia and type I interferon (INF1) immune response. A recent review of the published studies trying to explain the causality between COVID 19 and pernio-like lesions, from January 2020 to December 2020, speculate several possible mechanisms: an increase in the vasoconstrictive, prothrombotic, and proinflammatory effects of the angiotensin II pathway through activation of the ACE2 by the virus; COVID-19 triggers a robust INF1 immune response in predisposed patients; pernio as a sign of mild disease, may be explained by genetic and hormonal differences in the patients affected.7
Another condition that can be confused with CLE is Raynaud phenomenon, were patients present with white to purple to red patches on the fingers and toes after exposure to cold, but in comparison with pernio, the lesions improve within minutes to hours after rewarming. Secondary Raynaud phenomenon can be seen in patients with systemic lupus erythematosus and in patients with other connective tissue disorders. The skin lesions in our patient were persistent and were not triggered by cold exposure, making Raynaud phenomenon less likely. Children with vasculitis can present with painful red, violaceous, or necrotic lesions on the extremities, which can mimic pernio. Vasculitis lesions tend to be more purpuric and angulated, compared with pernio lesions, though in severe cases of pernio with ulceration it may be difficult to distinguish between the two entities and a skin biopsy may be needed.
Sweet syndrome, also known as acute febrile neutrophilic dermatosis, is a rare skin condition in which children present with edematous tender nodules on the hands and with less frequency in other parts of the body with associated fever, malaise, conjunctivitis, or joint pain and it is usually associated with infection or malignancy. Our patient denied any systemic symptoms and had no conjunctivitis nor arthritis.
Most patients with idiopathic pernio do not require a biopsy or further laboratory evaluation unless the lesions are atypical, chronic, or there is a suspected associated condition. The workup for patients with prolonged or atypical pernio-like lesions include a skin biopsy with direct immunofluorescence, ANA, complete blood count, complement levels, antiphospholipid antibodies, cold agglutinins, and cryoglobulins.
Treatment of mild CLE is with moderate- to high-potency topical corticosteroids. In those patients not responding to topical measures and keeping the extremities warm, the use of hydroxychloroquine has been reported to be beneficial in some patients as well as the use of calcium-channel blockers.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego.
References
1. Su WP et al. Cutis. 1994 Dec;54(6):395-9.
2. Boada A et al. Am J Dermatopathol. 2010 Feb;32(1):19-23.
3. Patel et al. SBMJ Case Rep. 2013;2013:bcr2013201165.
4. Genes Yi et al. BMC. 2020 Apr 15;18(1):32.
5. Battesti G et al. J Am Acad Dermatol. 2020;83(4):1219-22.
6. Cappel JA et al. Mayo Clin Proc. 2014 Feb;89(2):207-15.
7. Cappel MA et al. Mayo Clin Proc. 2021;96(4):989-1005.
He denied any hair loss, mouth sores, sun sensitivity, headaches, gastrointestinal complaints, joint pain, or muscle weakness.
He is not taking any medications.
He has been at home doing virtual school and has not traveled. He likes to play the piano. There is no family history of similar lesions, connective tissue disorder, or autoimmunity.
On physical exam he has purple discoloration on the toes with some violaceous and pink papules. On the fingers he has pink to violaceous papules and macules.
There is no joint edema or pain.
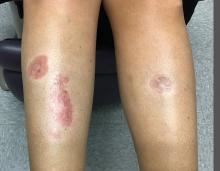
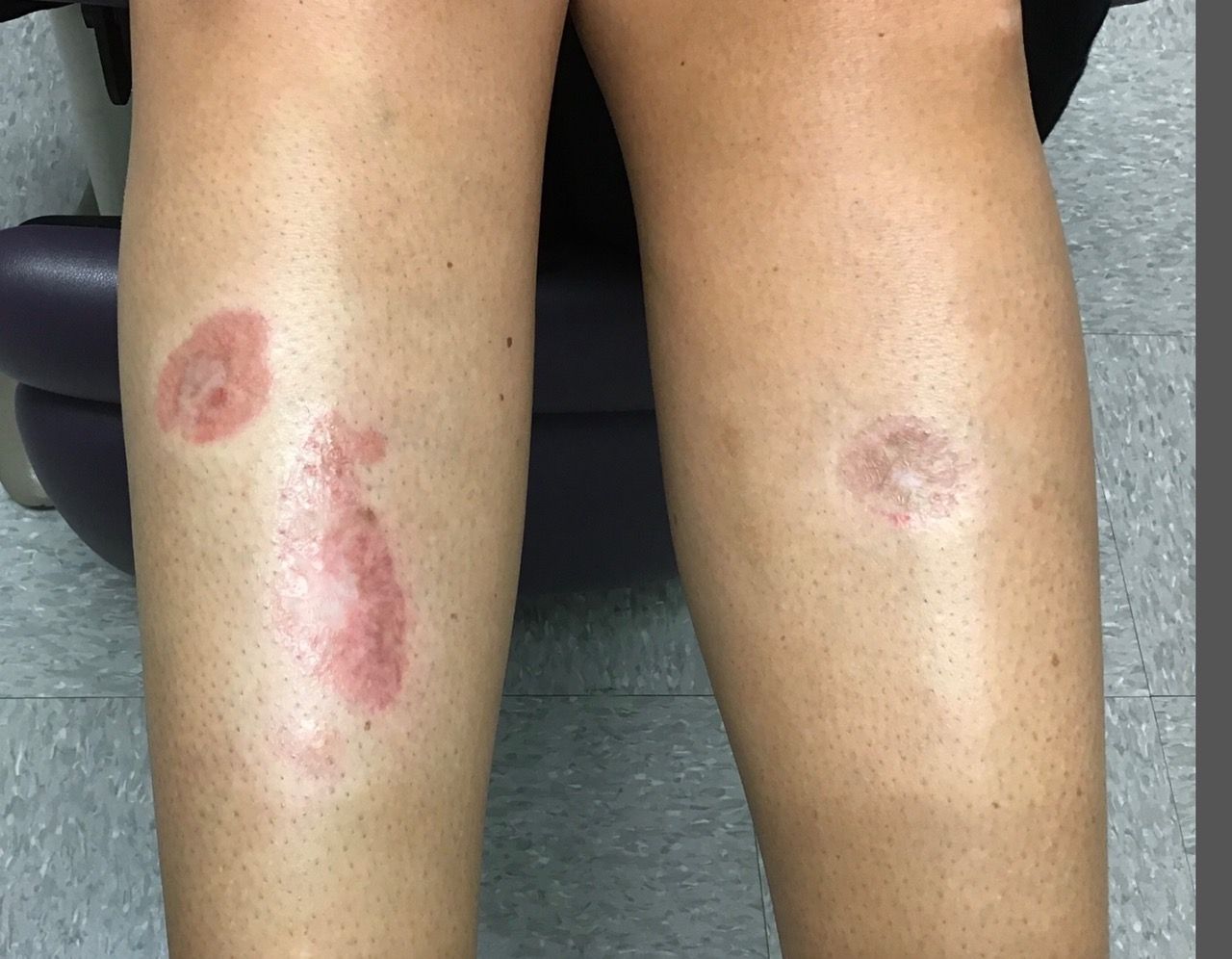
A woman with a history of diabetes, and plaques on both shins
. Women are often more affected than men. Patients often present in their 30s and 40s. The cause of NLD is unknown. Twenty percent of patients with NLD will have glucose intolerance or a family history of diabetes.1 The percentage of patients with NLD who have diabetes varies in reports from 11% to 65%.2 NLD may progress despite the diabetes treatment. Only 0.03% of patient with diabetes will have NLD.3
Lesions most commonly occur on the extremities, with shins being affected in most cases. They vary from asymptomatic to painful. Typically, lesions begin as small, firm erythematous papules that evolve into shiny, well-defined plaques. In older plaques, the center will often appear yellow, depressed, and atrophic, with telangiectasias. The periphery appears pink to violaceous to brown. Ulceration may be present, particularly after trauma, and there may be decreased sensation in the plaques. NLD is clinically distinct from diabetic dermopathy, which appear as brown macules, often in older patients with diabetes.
Ideally, biopsy should be taken at the edge of a lesion. Histologically, the epidermis appears normal or atrophic. A diffuse palisaded and interstitial granulomatous dermatitis consisting of histiocytes, multinucleated giant cells, lymphocytes, and plasma cells is seen in the dermis. Granulomas are often oriented parallel to the epidermis. There is no mucin at the center of the granulomas (as seen in granuloma annulare). Inflammation may extend into the subcutaneous fat. Asteroid bodies (as seen in sarcoid) are absent.
Unfortunately, treatment of NLD is often unsuccessful. Treatment includes potent topical corticosteroids for early lesions and intralesional triamcinolone to the leading edge of lesions. Care should be taken to avoid injecting centrally where atrophy and ulceration may result. Systemic steroids may be helpful in some cases, but can elevate glucose levels. Other reported medical treatments include pentoxifylline, cyclosporine, and niacinamide. Some lesions may spontaneously resolve. Ulcerations may require surgical excision with grafting.
This case and photo are provided by Dr. Bilu Martin, who is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. James WD et al. Andrews’ Diseases of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
2. Hashemi D et al. JAMA Dermatol. 2019 Apr 1;155(4):455-9.
3. Bolognia JL et al. Dermatology. St. Louis, Mo.: Mosby Elsevier, 2008.
. Women are often more affected than men. Patients often present in their 30s and 40s. The cause of NLD is unknown. Twenty percent of patients with NLD will have glucose intolerance or a family history of diabetes.1 The percentage of patients with NLD who have diabetes varies in reports from 11% to 65%.2 NLD may progress despite the diabetes treatment. Only 0.03% of patient with diabetes will have NLD.3
Lesions most commonly occur on the extremities, with shins being affected in most cases. They vary from asymptomatic to painful. Typically, lesions begin as small, firm erythematous papules that evolve into shiny, well-defined plaques. In older plaques, the center will often appear yellow, depressed, and atrophic, with telangiectasias. The periphery appears pink to violaceous to brown. Ulceration may be present, particularly after trauma, and there may be decreased sensation in the plaques. NLD is clinically distinct from diabetic dermopathy, which appear as brown macules, often in older patients with diabetes.
Ideally, biopsy should be taken at the edge of a lesion. Histologically, the epidermis appears normal or atrophic. A diffuse palisaded and interstitial granulomatous dermatitis consisting of histiocytes, multinucleated giant cells, lymphocytes, and plasma cells is seen in the dermis. Granulomas are often oriented parallel to the epidermis. There is no mucin at the center of the granulomas (as seen in granuloma annulare). Inflammation may extend into the subcutaneous fat. Asteroid bodies (as seen in sarcoid) are absent.
Unfortunately, treatment of NLD is often unsuccessful. Treatment includes potent topical corticosteroids for early lesions and intralesional triamcinolone to the leading edge of lesions. Care should be taken to avoid injecting centrally where atrophy and ulceration may result. Systemic steroids may be helpful in some cases, but can elevate glucose levels. Other reported medical treatments include pentoxifylline, cyclosporine, and niacinamide. Some lesions may spontaneously resolve. Ulcerations may require surgical excision with grafting.
This case and photo are provided by Dr. Bilu Martin, who is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. James WD et al. Andrews’ Diseases of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
2. Hashemi D et al. JAMA Dermatol. 2019 Apr 1;155(4):455-9.
3. Bolognia JL et al. Dermatology. St. Louis, Mo.: Mosby Elsevier, 2008.
. Women are often more affected than men. Patients often present in their 30s and 40s. The cause of NLD is unknown. Twenty percent of patients with NLD will have glucose intolerance or a family history of diabetes.1 The percentage of patients with NLD who have diabetes varies in reports from 11% to 65%.2 NLD may progress despite the diabetes treatment. Only 0.03% of patient with diabetes will have NLD.3
Lesions most commonly occur on the extremities, with shins being affected in most cases. They vary from asymptomatic to painful. Typically, lesions begin as small, firm erythematous papules that evolve into shiny, well-defined plaques. In older plaques, the center will often appear yellow, depressed, and atrophic, with telangiectasias. The periphery appears pink to violaceous to brown. Ulceration may be present, particularly after trauma, and there may be decreased sensation in the plaques. NLD is clinically distinct from diabetic dermopathy, which appear as brown macules, often in older patients with diabetes.
Ideally, biopsy should be taken at the edge of a lesion. Histologically, the epidermis appears normal or atrophic. A diffuse palisaded and interstitial granulomatous dermatitis consisting of histiocytes, multinucleated giant cells, lymphocytes, and plasma cells is seen in the dermis. Granulomas are often oriented parallel to the epidermis. There is no mucin at the center of the granulomas (as seen in granuloma annulare). Inflammation may extend into the subcutaneous fat. Asteroid bodies (as seen in sarcoid) are absent.
Unfortunately, treatment of NLD is often unsuccessful. Treatment includes potent topical corticosteroids for early lesions and intralesional triamcinolone to the leading edge of lesions. Care should be taken to avoid injecting centrally where atrophy and ulceration may result. Systemic steroids may be helpful in some cases, but can elevate glucose levels. Other reported medical treatments include pentoxifylline, cyclosporine, and niacinamide. Some lesions may spontaneously resolve. Ulcerations may require surgical excision with grafting.
This case and photo are provided by Dr. Bilu Martin, who is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. James WD et al. Andrews’ Diseases of the Skin: Clinical Dermatology. Philadelphia: Saunders Elsevier, 2006.
2. Hashemi D et al. JAMA Dermatol. 2019 Apr 1;155(4):455-9.
3. Bolognia JL et al. Dermatology. St. Louis, Mo.: Mosby Elsevier, 2008.
Black patients with cutaneous sarcoidosis may have more systemic and CV disease
according to a retrospective chart review of patients seen at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Black patients were also significantly more likely to have two or more organs involved and have higher rates of cardiac involvement, the latter of which is associated with worse prognosis. “Our data suggest there may be substantial variations in organ involvement between racial groups of patients presenting with cutaneous sarcoidosis,” said medical student Kylee Kus, a medical student at Oakland University, Auburn Hills, Mich., who presented the findings with Bina Kassamali, a medical student at Harvard University, Boston, at the annual Skin of Color Society scientific symposium.
Sotonye Imadojemu, MD, MBE; Avery LeChance, MD, MPH; and Ruth Anne Vleugels, MD, MPH, MBA; of Brigham and Women’s Hospital, are cosenior authors of the abstract.
The researchers identified 111 patients who were diagnosed with cutaneous sarcoidosis over a 20-year period (January 2000–December 2019), 50 of whom presented without established extracutaneous disease. They examined the charts of these 50 patients for whether subsequent work-up revealed systemic disease.
Of the 50 patients, 9 were Black. Seven of these nine patients (77.8%), were found to have systemic involvement, compared with 14 of 41 (46.3%) non-Black patients – a 31.5% higher probability (P < .05). One-third of the nine Black patients were found to have disease in one organ, and 44.4% in two or more organs. In non-Black patients, these rates were 12.2% and 34.1%, respectively.
Cardiovascular involvement was not found in any of the non-Black patients who had extracutaneous disease, but was found in 29% of the Black patients with extracutaneous disease, a statistically significant difference.
Black patients are known to be at higher risk for sarcoidosis than non-Black patients, and because “there is an association between cardiac sarcoid involvement and poor prognosis largely due to manifestations such as heart block, arrhythmias, and heart failure ... the study helps demonstrate how this organ involvement can disproportionately affect the Black population,” Ms. Kassamali said in an interview after the meeting.
A separate, recently published analysis of data from the same patient population examined the work-ups that patients received after a dermatologist’s diagnosis of sarcoidosis and found that patients with no previous systemic work-up were subsequently assessed for cardiac involvement in only 58.3% of cases. Assessment for pulmonary and ocular disease was completed more than 90% of the time.
“Crucial testing for cardiac involvement fell short,” Dr. Imadojemu, of the department of dermatology, Brigham and Women’s Hospital, and coinvestigators wrote in the research letter.
“Because the cutaneous manifestations of sarcoidosis often present at disease onset, dermatologists may be the first physicians to diagnose a patient with sarcoidosis,” they wrote. “As such, dermatologists are often responsible for initiating the appropriate evaluation of patients with sarcoidosis.”
Pulmonary involvement occurs in nearly all cases of sarcoidosis, while ocular and cardiac disease develop in approximately 25% and 10% of patients, respectively. Cardiac sarcoidosis is usually asymptomatic and accounts for 13%-25% of sarcoidosis-related deaths in the United States, they wrote.
An electrocardiogram is the appropriate initial screening tool and “is warranted in all patients with sarcoidosis,” they advised.
according to a retrospective chart review of patients seen at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Black patients were also significantly more likely to have two or more organs involved and have higher rates of cardiac involvement, the latter of which is associated with worse prognosis. “Our data suggest there may be substantial variations in organ involvement between racial groups of patients presenting with cutaneous sarcoidosis,” said medical student Kylee Kus, a medical student at Oakland University, Auburn Hills, Mich., who presented the findings with Bina Kassamali, a medical student at Harvard University, Boston, at the annual Skin of Color Society scientific symposium.
Sotonye Imadojemu, MD, MBE; Avery LeChance, MD, MPH; and Ruth Anne Vleugels, MD, MPH, MBA; of Brigham and Women’s Hospital, are cosenior authors of the abstract.
The researchers identified 111 patients who were diagnosed with cutaneous sarcoidosis over a 20-year period (January 2000–December 2019), 50 of whom presented without established extracutaneous disease. They examined the charts of these 50 patients for whether subsequent work-up revealed systemic disease.
Of the 50 patients, 9 were Black. Seven of these nine patients (77.8%), were found to have systemic involvement, compared with 14 of 41 (46.3%) non-Black patients – a 31.5% higher probability (P < .05). One-third of the nine Black patients were found to have disease in one organ, and 44.4% in two or more organs. In non-Black patients, these rates were 12.2% and 34.1%, respectively.
Cardiovascular involvement was not found in any of the non-Black patients who had extracutaneous disease, but was found in 29% of the Black patients with extracutaneous disease, a statistically significant difference.
Black patients are known to be at higher risk for sarcoidosis than non-Black patients, and because “there is an association between cardiac sarcoid involvement and poor prognosis largely due to manifestations such as heart block, arrhythmias, and heart failure ... the study helps demonstrate how this organ involvement can disproportionately affect the Black population,” Ms. Kassamali said in an interview after the meeting.
A separate, recently published analysis of data from the same patient population examined the work-ups that patients received after a dermatologist’s diagnosis of sarcoidosis and found that patients with no previous systemic work-up were subsequently assessed for cardiac involvement in only 58.3% of cases. Assessment for pulmonary and ocular disease was completed more than 90% of the time.
“Crucial testing for cardiac involvement fell short,” Dr. Imadojemu, of the department of dermatology, Brigham and Women’s Hospital, and coinvestigators wrote in the research letter.
“Because the cutaneous manifestations of sarcoidosis often present at disease onset, dermatologists may be the first physicians to diagnose a patient with sarcoidosis,” they wrote. “As such, dermatologists are often responsible for initiating the appropriate evaluation of patients with sarcoidosis.”
Pulmonary involvement occurs in nearly all cases of sarcoidosis, while ocular and cardiac disease develop in approximately 25% and 10% of patients, respectively. Cardiac sarcoidosis is usually asymptomatic and accounts for 13%-25% of sarcoidosis-related deaths in the United States, they wrote.
An electrocardiogram is the appropriate initial screening tool and “is warranted in all patients with sarcoidosis,” they advised.
according to a retrospective chart review of patients seen at Massachusetts General Hospital and Brigham and Women’s Hospital, both in Boston.
Black patients were also significantly more likely to have two or more organs involved and have higher rates of cardiac involvement, the latter of which is associated with worse prognosis. “Our data suggest there may be substantial variations in organ involvement between racial groups of patients presenting with cutaneous sarcoidosis,” said medical student Kylee Kus, a medical student at Oakland University, Auburn Hills, Mich., who presented the findings with Bina Kassamali, a medical student at Harvard University, Boston, at the annual Skin of Color Society scientific symposium.
Sotonye Imadojemu, MD, MBE; Avery LeChance, MD, MPH; and Ruth Anne Vleugels, MD, MPH, MBA; of Brigham and Women’s Hospital, are cosenior authors of the abstract.
The researchers identified 111 patients who were diagnosed with cutaneous sarcoidosis over a 20-year period (January 2000–December 2019), 50 of whom presented without established extracutaneous disease. They examined the charts of these 50 patients for whether subsequent work-up revealed systemic disease.
Of the 50 patients, 9 were Black. Seven of these nine patients (77.8%), were found to have systemic involvement, compared with 14 of 41 (46.3%) non-Black patients – a 31.5% higher probability (P < .05). One-third of the nine Black patients were found to have disease in one organ, and 44.4% in two or more organs. In non-Black patients, these rates were 12.2% and 34.1%, respectively.
Cardiovascular involvement was not found in any of the non-Black patients who had extracutaneous disease, but was found in 29% of the Black patients with extracutaneous disease, a statistically significant difference.
Black patients are known to be at higher risk for sarcoidosis than non-Black patients, and because “there is an association between cardiac sarcoid involvement and poor prognosis largely due to manifestations such as heart block, arrhythmias, and heart failure ... the study helps demonstrate how this organ involvement can disproportionately affect the Black population,” Ms. Kassamali said in an interview after the meeting.
A separate, recently published analysis of data from the same patient population examined the work-ups that patients received after a dermatologist’s diagnosis of sarcoidosis and found that patients with no previous systemic work-up were subsequently assessed for cardiac involvement in only 58.3% of cases. Assessment for pulmonary and ocular disease was completed more than 90% of the time.
“Crucial testing for cardiac involvement fell short,” Dr. Imadojemu, of the department of dermatology, Brigham and Women’s Hospital, and coinvestigators wrote in the research letter.
“Because the cutaneous manifestations of sarcoidosis often present at disease onset, dermatologists may be the first physicians to diagnose a patient with sarcoidosis,” they wrote. “As such, dermatologists are often responsible for initiating the appropriate evaluation of patients with sarcoidosis.”
Pulmonary involvement occurs in nearly all cases of sarcoidosis, while ocular and cardiac disease develop in approximately 25% and 10% of patients, respectively. Cardiac sarcoidosis is usually asymptomatic and accounts for 13%-25% of sarcoidosis-related deaths in the United States, they wrote.
An electrocardiogram is the appropriate initial screening tool and “is warranted in all patients with sarcoidosis,” they advised.
FROM SOC SOCIETY 2021
Blacks and Hispanics have higher inpatient use for mycosis fungoides
according to an analysis of the 2012-2017 National Inpatient Sample (NIS).
The findings are consistent with prior studies implicating earlier and more severe disease in Black and Hispanic patients, and reinforce the importance of accurate diagnosis and early treatment.
Dermatologists should maintain “a higher index of suspicion for MF in patients with skin of color, as early diagnosis may help mitigate the downstream costs of management,” Justin Choi, BA, a medical student at the University of Illinois at Chicago, said at the annual Skin of Color Society symposium.
Mr. Choi and coinvestigators, led by Shawn Kwatra, MD, of Johns Hopkins University, Baltimore, identified hospital admissions for MF in the NIS for 10,790 White patients, 4,020 Black patients, and 1,615 Hispanic patients over the 5-year period. The inpatient prevalence of MF – the most common variant of primary cutaneous T-cell lymphoma – was highest in these groups.
Black and Hispanic patients who were hospitalized for MF were significantly younger than White patients, with a mean age of 51.7 years and 48.5 years, respectively, compared with 59.9 years (P < .001 in each case). They also had longer lengths of stay: 8.34 days on average for Black patients and 8.88 for Hispanic patients, compared with 6.66 days for White patients (P < .001 and P = .001, respectively).
Hispanic patients accrued the highest costs of care (a mean of $107,242 vs. $64,049, P =.003) and underwent more procedures (a mean of 2.43 vs. 1.93, P = .004) than White patients. Black patients similarly had higher costs associated with their hospital stay (a mean of $75,053 vs. $64,049, P =.042).
In a multivariate linear regression adjusted for age, sex and insurance type, Black race remained significantly associated with a longer LOS than White race, and Hispanic ethnicity with a longer LOS, increased costs, and more procedures than White race.
The NIS is a publicly available, all-payer inpatient care database developed for the Agency for Healthcare Research and Quality’s Healthcare Cost and Utilization Project.
Mr. Choi is a dermatology research fellow working under the guidance of Dr. Kwatra.
according to an analysis of the 2012-2017 National Inpatient Sample (NIS).
The findings are consistent with prior studies implicating earlier and more severe disease in Black and Hispanic patients, and reinforce the importance of accurate diagnosis and early treatment.
Dermatologists should maintain “a higher index of suspicion for MF in patients with skin of color, as early diagnosis may help mitigate the downstream costs of management,” Justin Choi, BA, a medical student at the University of Illinois at Chicago, said at the annual Skin of Color Society symposium.
Mr. Choi and coinvestigators, led by Shawn Kwatra, MD, of Johns Hopkins University, Baltimore, identified hospital admissions for MF in the NIS for 10,790 White patients, 4,020 Black patients, and 1,615 Hispanic patients over the 5-year period. The inpatient prevalence of MF – the most common variant of primary cutaneous T-cell lymphoma – was highest in these groups.
Black and Hispanic patients who were hospitalized for MF were significantly younger than White patients, with a mean age of 51.7 years and 48.5 years, respectively, compared with 59.9 years (P < .001 in each case). They also had longer lengths of stay: 8.34 days on average for Black patients and 8.88 for Hispanic patients, compared with 6.66 days for White patients (P < .001 and P = .001, respectively).
Hispanic patients accrued the highest costs of care (a mean of $107,242 vs. $64,049, P =.003) and underwent more procedures (a mean of 2.43 vs. 1.93, P = .004) than White patients. Black patients similarly had higher costs associated with their hospital stay (a mean of $75,053 vs. $64,049, P =.042).
In a multivariate linear regression adjusted for age, sex and insurance type, Black race remained significantly associated with a longer LOS than White race, and Hispanic ethnicity with a longer LOS, increased costs, and more procedures than White race.
The NIS is a publicly available, all-payer inpatient care database developed for the Agency for Healthcare Research and Quality’s Healthcare Cost and Utilization Project.
Mr. Choi is a dermatology research fellow working under the guidance of Dr. Kwatra.
according to an analysis of the 2012-2017 National Inpatient Sample (NIS).
The findings are consistent with prior studies implicating earlier and more severe disease in Black and Hispanic patients, and reinforce the importance of accurate diagnosis and early treatment.
Dermatologists should maintain “a higher index of suspicion for MF in patients with skin of color, as early diagnosis may help mitigate the downstream costs of management,” Justin Choi, BA, a medical student at the University of Illinois at Chicago, said at the annual Skin of Color Society symposium.
Mr. Choi and coinvestigators, led by Shawn Kwatra, MD, of Johns Hopkins University, Baltimore, identified hospital admissions for MF in the NIS for 10,790 White patients, 4,020 Black patients, and 1,615 Hispanic patients over the 5-year period. The inpatient prevalence of MF – the most common variant of primary cutaneous T-cell lymphoma – was highest in these groups.
Black and Hispanic patients who were hospitalized for MF were significantly younger than White patients, with a mean age of 51.7 years and 48.5 years, respectively, compared with 59.9 years (P < .001 in each case). They also had longer lengths of stay: 8.34 days on average for Black patients and 8.88 for Hispanic patients, compared with 6.66 days for White patients (P < .001 and P = .001, respectively).
Hispanic patients accrued the highest costs of care (a mean of $107,242 vs. $64,049, P =.003) and underwent more procedures (a mean of 2.43 vs. 1.93, P = .004) than White patients. Black patients similarly had higher costs associated with their hospital stay (a mean of $75,053 vs. $64,049, P =.042).
In a multivariate linear regression adjusted for age, sex and insurance type, Black race remained significantly associated with a longer LOS than White race, and Hispanic ethnicity with a longer LOS, increased costs, and more procedures than White race.
The NIS is a publicly available, all-payer inpatient care database developed for the Agency for Healthcare Research and Quality’s Healthcare Cost and Utilization Project.
Mr. Choi is a dermatology research fellow working under the guidance of Dr. Kwatra.
FROM SOC SOCIETY 2021
Chronic breast rash
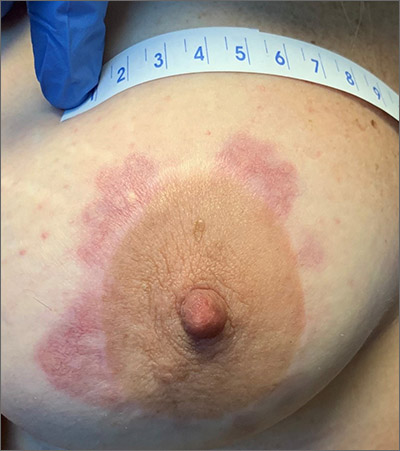
A punch biopsy revealed that the patient had granuloma annulare (GA).
GA is usually a self-limiting disorder that manifests as a single or, less commonly, multiple nonscaly, red, annular lesions that are typically found on the extremities. It frequently starts as a papule or cluster of papules before coalescing into its classic annular pattern. Biopsy is not usually needed to make the diagnosis when annular lesions are present. In this case, the lesions displayed the Koebner phenomenon, occurring along her areolar scar, making diagnosis more difficult and necessitating the biopsy. While the cause of GA is unknown, it has been found more often in women than men, but has no predilection for race, ethnicity, or geographic areas.1
GA is typically asymptomatic and can resolve spontaneously. Treatment is often performed for cosmetic reasons. First-line therapies include topical corticosteroids, topical tacrolimus, imiquimod cream, intralesional injections into the elevated border with 2.5 to 5 mg/mL triamcinolone acetonide, or destructive methods such as cryosurgery or pulsed dye laser therapy.1
After a discussion of treatment options, this patient chose watchful waiting.
Image courtesy of Kamini Geer, MD, and text courtesy of Kamini Geer, MD, AdventHealth East Orlando Osteopathic Family Medicine Residency and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Habif TP. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Saunders; 2015.

A punch biopsy revealed that the patient had granuloma annulare (GA).
GA is usually a self-limiting disorder that manifests as a single or, less commonly, multiple nonscaly, red, annular lesions that are typically found on the extremities. It frequently starts as a papule or cluster of papules before coalescing into its classic annular pattern. Biopsy is not usually needed to make the diagnosis when annular lesions are present. In this case, the lesions displayed the Koebner phenomenon, occurring along her areolar scar, making diagnosis more difficult and necessitating the biopsy. While the cause of GA is unknown, it has been found more often in women than men, but has no predilection for race, ethnicity, or geographic areas.1
GA is typically asymptomatic and can resolve spontaneously. Treatment is often performed for cosmetic reasons. First-line therapies include topical corticosteroids, topical tacrolimus, imiquimod cream, intralesional injections into the elevated border with 2.5 to 5 mg/mL triamcinolone acetonide, or destructive methods such as cryosurgery or pulsed dye laser therapy.1
After a discussion of treatment options, this patient chose watchful waiting.
Image courtesy of Kamini Geer, MD, and text courtesy of Kamini Geer, MD, AdventHealth East Orlando Osteopathic Family Medicine Residency and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

A punch biopsy revealed that the patient had granuloma annulare (GA).
GA is usually a self-limiting disorder that manifests as a single or, less commonly, multiple nonscaly, red, annular lesions that are typically found on the extremities. It frequently starts as a papule or cluster of papules before coalescing into its classic annular pattern. Biopsy is not usually needed to make the diagnosis when annular lesions are present. In this case, the lesions displayed the Koebner phenomenon, occurring along her areolar scar, making diagnosis more difficult and necessitating the biopsy. While the cause of GA is unknown, it has been found more often in women than men, but has no predilection for race, ethnicity, or geographic areas.1
GA is typically asymptomatic and can resolve spontaneously. Treatment is often performed for cosmetic reasons. First-line therapies include topical corticosteroids, topical tacrolimus, imiquimod cream, intralesional injections into the elevated border with 2.5 to 5 mg/mL triamcinolone acetonide, or destructive methods such as cryosurgery or pulsed dye laser therapy.1
After a discussion of treatment options, this patient chose watchful waiting.
Image courtesy of Kamini Geer, MD, and text courtesy of Kamini Geer, MD, AdventHealth East Orlando Osteopathic Family Medicine Residency and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
1. Habif TP. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Saunders; 2015.
1. Habif TP. Clinical Dermatology: A Color Guide to Diagnosis and Therapy. 6th ed. Saunders; 2015.
Data about COVID-19-related skin manifestations in children continue to emerge
Two and stratifying children at risk for serious, systemic illness due to the virus.
In a single-center descriptive study carried out over a 9-month period, researchers in Madrid found that of 50 hospitalized children infected with COVID-19, 21 (42%) had mucocutaneous symptoms, most commonly exanthem, followed by conjunctival hyperemia without secretion and red cracked lips or strawberry tongue. In addition, 18 (36%) fulfilled criteria for Multisystem Inflammatory Syndrome in Children (MIS-C).
“Based on findings in adult patients, the skin manifestations of COVID-19 have been classified under five categories: acral pseudo-chilblain, vesicular eruptions, urticarial lesions, maculopapular eruptions, and livedo or necrosis,” David Andina-Martinez, MD, of Hospital Infantil Universitario Niño Jesús, Madrid, and colleagues wrote in the study, which was published online on April 2 in the Journal of the American Academy of Dermatology.
“Chilblain lesions in healthy children and adolescents have received much attention; these lesions resolve without complications after a few weeks,” they added. “Besides, other cutaneous manifestations of COVID-19 in children have been the matter of case reports or small case series. Nevertheless, the mucocutaneous manifestations in hospitalized children infected with SARS-CoV-2 and their implications on the clinical course have not yet been extensively described.”
In an effort to describe the mucocutaneous manifestations in children hospitalized for COVID-19, the researchers evaluated 50 children up to 18 years of age who were admitted between March 1 and Nov. 30, 2020, to Hospital Infantil Universitario Niño Jesús, which was designated as a pediatric reference center during the peak of the pandemic. The main reasons for admission were respiratory illness (40%) and MIS-C (40%).
Of the 50 patients, 44 (88%) had a positive RT-PCR for SARS-CoV-2 and 6 (12%) met clinical suspicion criteria and had a negative RT-PCR with a positive IgG serology. In 34 patients (68%), a close contact with a suspected or confirmed case of COVID-19 was referred, while the source of the infection remained unknown in the remaining 16 patients (32%).
The researchers reported that 21 patients (42%) had mucocutaneous symptoms, most commonly maculopapular exanthem (86%), conjunctival hyperemia (81%), and red cracked lips or strawberry tongue (43%). In addition, 18 of the 21 patients (86%) fulfilled criteria for MIS-C.
“A tricky thing about MIS-C is that it often manifests 4-5 weeks after a child had COVID-19,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was asked to comment on the study. “MIS-C is associated with characteristic bright red lips and a red tongue that might resemble a strawberry. Such oral findings should prompt rapid evaluation for other signs and symptoms. There can be redness of the eyes or other more nonspecific skin findings (large or small areas of redness on the trunk or limbs, sometimes with surface change), but more importantly, fever, a rapid heartbeat, diarrhea, or breathing issues. The risk with MIS-C is a rapid decline in a child’s health, with admission to an intensive care unit.”
Dr. Andina-Martinez and his colleagues also contrast the skin findings of MIS-C, which are not generally on the hands or feet, with the so-called “COVID toe” or finger phenomenon, which has also been associated with SARS-CoV-2, particularly in children. “Only one of the patients in this series had skin involvement of a finger, and it only appeared after recovery from MIS-C,” Dr. Ko noted. “Distinguishing COVID toes from MIS-C is important, as COVID toes has a very good outcome, while MIS-C can have severe consequences, including protracted heart disease.”
In other findings, patients who presented with mucocutaneous signs tended to be older than those without skin signs and they presented at the emergency department with poor general status and extreme tachycardia. They also had higher C-reactive protein and D-dimer levels and lower lymphocyte counts and faced a more than a 10-fold increased risk of being admitted to the PICU, compared with patients who did not have skin signs (OR, 10.24; P = .003).
In a separate study published online on April 7 in JAMA Dermatology, Zachary E. Holcomb, MD, of the combined dermatology residency program at Massachusetts General Hospital, Boston, and colleagues presented what is believed to be the first case report of reactive infectious mucocutaneous eruption (RIME) triggered by SARS-CoV-2. RIME is the preferred term for pediatric patients who present with mucositis and rash (often a scant or even absent skin eruption) triggered by various infectious agents.
The patient, a 17-year-old male, presented to the emergency department with 3 days of mouth pain and nonpainful penile erosions. “One week prior, he experienced transient anosmia and ageusia that had since spontaneously resolved,” the researchers wrote. “At that time, he was tested for SARS-CoV-2 infection via nasopharyngeal polymerase chain reaction (PCR), the results of which were positive.”
At presentation, the patient had no fever, his vital signs were normal, and the physical exam revealed shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities. Serum analysis revealed a normal white blood cell count with mild absolute lymphopenia, slightly elevated creatinine level, normal liver function, slightly elevated C-reactive protein level, and normal ferritin level.
Dr. Holcomb and colleagues made a diagnosis of SARS-CoV-2–associated RIME based on microbiological results, which revealed positive repeated SARS-CoV-2 nasopharyngeal PCR and negative nasopharyngeal PCR testing for Mycoplasma pneumoniae, adenovirus, Chlamydophila pneumoniae, human metapneumovirus, influenza A/B, parainfluenza 1 to 4, rhinovirus, and respiratory syncytial virus. In addition, titers of Mycoplasma pneumoniae IgM levels were negative, but Mycoplasma pneumoniae IgG levels were elevated.
The lesions resolved with 60 mg of oral prednisone taken daily for 4 days. A recurrence of oral mucositis 3 months later responded to 80 mg oral prednisone taken daily for 6 days.
“It’s not surprising that SARS-CoV-2 is yet another trigger for RIME,” said Anna Yasmine Kirkorian, MD, chief of the division of dermatology at Children’s National Hospital, Washington, who was asked to comment about the case report.
“The take-home message is for clinicians to be aware of this association and distinguish these patients from those with MIS-C, because patients with MIS-C require monitoring and urgent systemic treatment. RIME and MIS-C may potentially be distinguished clinically based on the nature of the mucositis (hemorrhagic and erosive in RIME, dry, cracked lips with ‘strawberry tongue’ in MIS-C) but more importantly patients with RIME lack laboratory evidence of severe systemic inflammation,” such as ESR, CRP, or ferritin, she said.
“A final interesting point in this article was the recurrence of mucositis in this patient, which could mean that recurrent mucositis/recurrent RIME might be yet another manifestation of ‘long-COVID’ (now called post-Acute Sequelae of SARS-CoV-2 infection) in some patients,” Dr. Kirkorian added. She noted that the American Academy of Dermatology–International League of Dermatologic Societies COVID-19 Dermatology Registry and articles like these “provide invaluable ‘hot off the presses’ information for clinicians who are facing the protean manifestations of a novel viral epidemic.”
The researchers reported having no financial disclosures.
Two and stratifying children at risk for serious, systemic illness due to the virus.
In a single-center descriptive study carried out over a 9-month period, researchers in Madrid found that of 50 hospitalized children infected with COVID-19, 21 (42%) had mucocutaneous symptoms, most commonly exanthem, followed by conjunctival hyperemia without secretion and red cracked lips or strawberry tongue. In addition, 18 (36%) fulfilled criteria for Multisystem Inflammatory Syndrome in Children (MIS-C).
“Based on findings in adult patients, the skin manifestations of COVID-19 have been classified under five categories: acral pseudo-chilblain, vesicular eruptions, urticarial lesions, maculopapular eruptions, and livedo or necrosis,” David Andina-Martinez, MD, of Hospital Infantil Universitario Niño Jesús, Madrid, and colleagues wrote in the study, which was published online on April 2 in the Journal of the American Academy of Dermatology.
“Chilblain lesions in healthy children and adolescents have received much attention; these lesions resolve without complications after a few weeks,” they added. “Besides, other cutaneous manifestations of COVID-19 in children have been the matter of case reports or small case series. Nevertheless, the mucocutaneous manifestations in hospitalized children infected with SARS-CoV-2 and their implications on the clinical course have not yet been extensively described.”
In an effort to describe the mucocutaneous manifestations in children hospitalized for COVID-19, the researchers evaluated 50 children up to 18 years of age who were admitted between March 1 and Nov. 30, 2020, to Hospital Infantil Universitario Niño Jesús, which was designated as a pediatric reference center during the peak of the pandemic. The main reasons for admission were respiratory illness (40%) and MIS-C (40%).
Of the 50 patients, 44 (88%) had a positive RT-PCR for SARS-CoV-2 and 6 (12%) met clinical suspicion criteria and had a negative RT-PCR with a positive IgG serology. In 34 patients (68%), a close contact with a suspected or confirmed case of COVID-19 was referred, while the source of the infection remained unknown in the remaining 16 patients (32%).
The researchers reported that 21 patients (42%) had mucocutaneous symptoms, most commonly maculopapular exanthem (86%), conjunctival hyperemia (81%), and red cracked lips or strawberry tongue (43%). In addition, 18 of the 21 patients (86%) fulfilled criteria for MIS-C.
“A tricky thing about MIS-C is that it often manifests 4-5 weeks after a child had COVID-19,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was asked to comment on the study. “MIS-C is associated with characteristic bright red lips and a red tongue that might resemble a strawberry. Such oral findings should prompt rapid evaluation for other signs and symptoms. There can be redness of the eyes or other more nonspecific skin findings (large or small areas of redness on the trunk or limbs, sometimes with surface change), but more importantly, fever, a rapid heartbeat, diarrhea, or breathing issues. The risk with MIS-C is a rapid decline in a child’s health, with admission to an intensive care unit.”
Dr. Andina-Martinez and his colleagues also contrast the skin findings of MIS-C, which are not generally on the hands or feet, with the so-called “COVID toe” or finger phenomenon, which has also been associated with SARS-CoV-2, particularly in children. “Only one of the patients in this series had skin involvement of a finger, and it only appeared after recovery from MIS-C,” Dr. Ko noted. “Distinguishing COVID toes from MIS-C is important, as COVID toes has a very good outcome, while MIS-C can have severe consequences, including protracted heart disease.”
In other findings, patients who presented with mucocutaneous signs tended to be older than those without skin signs and they presented at the emergency department with poor general status and extreme tachycardia. They also had higher C-reactive protein and D-dimer levels and lower lymphocyte counts and faced a more than a 10-fold increased risk of being admitted to the PICU, compared with patients who did not have skin signs (OR, 10.24; P = .003).
In a separate study published online on April 7 in JAMA Dermatology, Zachary E. Holcomb, MD, of the combined dermatology residency program at Massachusetts General Hospital, Boston, and colleagues presented what is believed to be the first case report of reactive infectious mucocutaneous eruption (RIME) triggered by SARS-CoV-2. RIME is the preferred term for pediatric patients who present with mucositis and rash (often a scant or even absent skin eruption) triggered by various infectious agents.
The patient, a 17-year-old male, presented to the emergency department with 3 days of mouth pain and nonpainful penile erosions. “One week prior, he experienced transient anosmia and ageusia that had since spontaneously resolved,” the researchers wrote. “At that time, he was tested for SARS-CoV-2 infection via nasopharyngeal polymerase chain reaction (PCR), the results of which were positive.”
At presentation, the patient had no fever, his vital signs were normal, and the physical exam revealed shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities. Serum analysis revealed a normal white blood cell count with mild absolute lymphopenia, slightly elevated creatinine level, normal liver function, slightly elevated C-reactive protein level, and normal ferritin level.
Dr. Holcomb and colleagues made a diagnosis of SARS-CoV-2–associated RIME based on microbiological results, which revealed positive repeated SARS-CoV-2 nasopharyngeal PCR and negative nasopharyngeal PCR testing for Mycoplasma pneumoniae, adenovirus, Chlamydophila pneumoniae, human metapneumovirus, influenza A/B, parainfluenza 1 to 4, rhinovirus, and respiratory syncytial virus. In addition, titers of Mycoplasma pneumoniae IgM levels were negative, but Mycoplasma pneumoniae IgG levels were elevated.
The lesions resolved with 60 mg of oral prednisone taken daily for 4 days. A recurrence of oral mucositis 3 months later responded to 80 mg oral prednisone taken daily for 6 days.
“It’s not surprising that SARS-CoV-2 is yet another trigger for RIME,” said Anna Yasmine Kirkorian, MD, chief of the division of dermatology at Children’s National Hospital, Washington, who was asked to comment about the case report.
“The take-home message is for clinicians to be aware of this association and distinguish these patients from those with MIS-C, because patients with MIS-C require monitoring and urgent systemic treatment. RIME and MIS-C may potentially be distinguished clinically based on the nature of the mucositis (hemorrhagic and erosive in RIME, dry, cracked lips with ‘strawberry tongue’ in MIS-C) but more importantly patients with RIME lack laboratory evidence of severe systemic inflammation,” such as ESR, CRP, or ferritin, she said.
“A final interesting point in this article was the recurrence of mucositis in this patient, which could mean that recurrent mucositis/recurrent RIME might be yet another manifestation of ‘long-COVID’ (now called post-Acute Sequelae of SARS-CoV-2 infection) in some patients,” Dr. Kirkorian added. She noted that the American Academy of Dermatology–International League of Dermatologic Societies COVID-19 Dermatology Registry and articles like these “provide invaluable ‘hot off the presses’ information for clinicians who are facing the protean manifestations of a novel viral epidemic.”
The researchers reported having no financial disclosures.
Two and stratifying children at risk for serious, systemic illness due to the virus.
In a single-center descriptive study carried out over a 9-month period, researchers in Madrid found that of 50 hospitalized children infected with COVID-19, 21 (42%) had mucocutaneous symptoms, most commonly exanthem, followed by conjunctival hyperemia without secretion and red cracked lips or strawberry tongue. In addition, 18 (36%) fulfilled criteria for Multisystem Inflammatory Syndrome in Children (MIS-C).
“Based on findings in adult patients, the skin manifestations of COVID-19 have been classified under five categories: acral pseudo-chilblain, vesicular eruptions, urticarial lesions, maculopapular eruptions, and livedo or necrosis,” David Andina-Martinez, MD, of Hospital Infantil Universitario Niño Jesús, Madrid, and colleagues wrote in the study, which was published online on April 2 in the Journal of the American Academy of Dermatology.
“Chilblain lesions in healthy children and adolescents have received much attention; these lesions resolve without complications after a few weeks,” they added. “Besides, other cutaneous manifestations of COVID-19 in children have been the matter of case reports or small case series. Nevertheless, the mucocutaneous manifestations in hospitalized children infected with SARS-CoV-2 and their implications on the clinical course have not yet been extensively described.”
In an effort to describe the mucocutaneous manifestations in children hospitalized for COVID-19, the researchers evaluated 50 children up to 18 years of age who were admitted between March 1 and Nov. 30, 2020, to Hospital Infantil Universitario Niño Jesús, which was designated as a pediatric reference center during the peak of the pandemic. The main reasons for admission were respiratory illness (40%) and MIS-C (40%).
Of the 50 patients, 44 (88%) had a positive RT-PCR for SARS-CoV-2 and 6 (12%) met clinical suspicion criteria and had a negative RT-PCR with a positive IgG serology. In 34 patients (68%), a close contact with a suspected or confirmed case of COVID-19 was referred, while the source of the infection remained unknown in the remaining 16 patients (32%).
The researchers reported that 21 patients (42%) had mucocutaneous symptoms, most commonly maculopapular exanthem (86%), conjunctival hyperemia (81%), and red cracked lips or strawberry tongue (43%). In addition, 18 of the 21 patients (86%) fulfilled criteria for MIS-C.
“A tricky thing about MIS-C is that it often manifests 4-5 weeks after a child had COVID-19,” said Christine Ko, MD, professor of dermatology and pathology at Yale University, New Haven, Conn., who was asked to comment on the study. “MIS-C is associated with characteristic bright red lips and a red tongue that might resemble a strawberry. Such oral findings should prompt rapid evaluation for other signs and symptoms. There can be redness of the eyes or other more nonspecific skin findings (large or small areas of redness on the trunk or limbs, sometimes with surface change), but more importantly, fever, a rapid heartbeat, diarrhea, or breathing issues. The risk with MIS-C is a rapid decline in a child’s health, with admission to an intensive care unit.”
Dr. Andina-Martinez and his colleagues also contrast the skin findings of MIS-C, which are not generally on the hands or feet, with the so-called “COVID toe” or finger phenomenon, which has also been associated with SARS-CoV-2, particularly in children. “Only one of the patients in this series had skin involvement of a finger, and it only appeared after recovery from MIS-C,” Dr. Ko noted. “Distinguishing COVID toes from MIS-C is important, as COVID toes has a very good outcome, while MIS-C can have severe consequences, including protracted heart disease.”
In other findings, patients who presented with mucocutaneous signs tended to be older than those without skin signs and they presented at the emergency department with poor general status and extreme tachycardia. They also had higher C-reactive protein and D-dimer levels and lower lymphocyte counts and faced a more than a 10-fold increased risk of being admitted to the PICU, compared with patients who did not have skin signs (OR, 10.24; P = .003).
In a separate study published online on April 7 in JAMA Dermatology, Zachary E. Holcomb, MD, of the combined dermatology residency program at Massachusetts General Hospital, Boston, and colleagues presented what is believed to be the first case report of reactive infectious mucocutaneous eruption (RIME) triggered by SARS-CoV-2. RIME is the preferred term for pediatric patients who present with mucositis and rash (often a scant or even absent skin eruption) triggered by various infectious agents.
The patient, a 17-year-old male, presented to the emergency department with 3 days of mouth pain and nonpainful penile erosions. “One week prior, he experienced transient anosmia and ageusia that had since spontaneously resolved,” the researchers wrote. “At that time, he was tested for SARS-CoV-2 infection via nasopharyngeal polymerase chain reaction (PCR), the results of which were positive.”
At presentation, the patient had no fever, his vital signs were normal, and the physical exam revealed shallow erosions of the vermilion lips and hard palate, circumferential erythematous erosions of the periurethral glans penis, and five small vesicles on the trunk and upper extremities. Serum analysis revealed a normal white blood cell count with mild absolute lymphopenia, slightly elevated creatinine level, normal liver function, slightly elevated C-reactive protein level, and normal ferritin level.
Dr. Holcomb and colleagues made a diagnosis of SARS-CoV-2–associated RIME based on microbiological results, which revealed positive repeated SARS-CoV-2 nasopharyngeal PCR and negative nasopharyngeal PCR testing for Mycoplasma pneumoniae, adenovirus, Chlamydophila pneumoniae, human metapneumovirus, influenza A/B, parainfluenza 1 to 4, rhinovirus, and respiratory syncytial virus. In addition, titers of Mycoplasma pneumoniae IgM levels were negative, but Mycoplasma pneumoniae IgG levels were elevated.
The lesions resolved with 60 mg of oral prednisone taken daily for 4 days. A recurrence of oral mucositis 3 months later responded to 80 mg oral prednisone taken daily for 6 days.
“It’s not surprising that SARS-CoV-2 is yet another trigger for RIME,” said Anna Yasmine Kirkorian, MD, chief of the division of dermatology at Children’s National Hospital, Washington, who was asked to comment about the case report.
“The take-home message is for clinicians to be aware of this association and distinguish these patients from those with MIS-C, because patients with MIS-C require monitoring and urgent systemic treatment. RIME and MIS-C may potentially be distinguished clinically based on the nature of the mucositis (hemorrhagic and erosive in RIME, dry, cracked lips with ‘strawberry tongue’ in MIS-C) but more importantly patients with RIME lack laboratory evidence of severe systemic inflammation,” such as ESR, CRP, or ferritin, she said.
“A final interesting point in this article was the recurrence of mucositis in this patient, which could mean that recurrent mucositis/recurrent RIME might be yet another manifestation of ‘long-COVID’ (now called post-Acute Sequelae of SARS-CoV-2 infection) in some patients,” Dr. Kirkorian added. She noted that the American Academy of Dermatology–International League of Dermatologic Societies COVID-19 Dermatology Registry and articles like these “provide invaluable ‘hot off the presses’ information for clinicians who are facing the protean manifestations of a novel viral epidemic.”
The researchers reported having no financial disclosures.