User login
She Can’t Turn the Other Cheek on the Lesion
ANSWER
The correct answer is seborrheic keratosis (choice “a”).
DISCUSSION
Seborrheic keratosis could not be in the differential because it is, by definition, an epidermal lesion—that is, “stuck on” the surface of the skin. It creates a rough surface that can be easily scraped off. The lesion could have been an actual scar, but other factors (its continuous growth) and the history of excessive ultraviolet exposure pushed us away from including this condition in the differential.
The differential for this patient included sun-caused skin cancers: basal cell carcinoma (BCC; choice “b”), squamous cell carcinoma (SCC; choice “d”), and amelanotic melanoma (choice “c”). These conditions can have a colorless and scar-like appearance, and they also destroy surface adnexae. Therefore, the lack of hairs, pores, or skin lines in a circumscribed area should raise concern for possible skin cancer, especially in at-risk patients such as this one.
BCC (otherwise known as cicatricial basal cell carcinoma) is by far the most common of all sun-caused skin cancers, but it usually presents as an obvious papule or nodule, often with telltale features such as pearly, rolled borders and focal erosion or ulceration. But there are exceptions, and the scar-like BCC is one.
SCC can also occasionally present in this manner, as can amelanotic melanoma, which is a colorless melanoma and very easy to miss. This case perfectly illustrates the point I often make to the students and residents I teach: When skin cancer is suspected, pay at least as much attention to the owner as to the lesion. Also, when in doubt, biopsy will settle the matter.
TREATMENT
For the patient, shave biopsy confirmed the presence of BCC. She was then referred for Mohs micrographic surgery because of the lesion’s size, location, and uncertain visible margins.
ANSWER
The correct answer is seborrheic keratosis (choice “a”).
DISCUSSION
Seborrheic keratosis could not be in the differential because it is, by definition, an epidermal lesion—that is, “stuck on” the surface of the skin. It creates a rough surface that can be easily scraped off. The lesion could have been an actual scar, but other factors (its continuous growth) and the history of excessive ultraviolet exposure pushed us away from including this condition in the differential.
The differential for this patient included sun-caused skin cancers: basal cell carcinoma (BCC; choice “b”), squamous cell carcinoma (SCC; choice “d”), and amelanotic melanoma (choice “c”). These conditions can have a colorless and scar-like appearance, and they also destroy surface adnexae. Therefore, the lack of hairs, pores, or skin lines in a circumscribed area should raise concern for possible skin cancer, especially in at-risk patients such as this one.
BCC (otherwise known as cicatricial basal cell carcinoma) is by far the most common of all sun-caused skin cancers, but it usually presents as an obvious papule or nodule, often with telltale features such as pearly, rolled borders and focal erosion or ulceration. But there are exceptions, and the scar-like BCC is one.
SCC can also occasionally present in this manner, as can amelanotic melanoma, which is a colorless melanoma and very easy to miss. This case perfectly illustrates the point I often make to the students and residents I teach: When skin cancer is suspected, pay at least as much attention to the owner as to the lesion. Also, when in doubt, biopsy will settle the matter.
TREATMENT
For the patient, shave biopsy confirmed the presence of BCC. She was then referred for Mohs micrographic surgery because of the lesion’s size, location, and uncertain visible margins.
ANSWER
The correct answer is seborrheic keratosis (choice “a”).
DISCUSSION
Seborrheic keratosis could not be in the differential because it is, by definition, an epidermal lesion—that is, “stuck on” the surface of the skin. It creates a rough surface that can be easily scraped off. The lesion could have been an actual scar, but other factors (its continuous growth) and the history of excessive ultraviolet exposure pushed us away from including this condition in the differential.
The differential for this patient included sun-caused skin cancers: basal cell carcinoma (BCC; choice “b”), squamous cell carcinoma (SCC; choice “d”), and amelanotic melanoma (choice “c”). These conditions can have a colorless and scar-like appearance, and they also destroy surface adnexae. Therefore, the lack of hairs, pores, or skin lines in a circumscribed area should raise concern for possible skin cancer, especially in at-risk patients such as this one.
BCC (otherwise known as cicatricial basal cell carcinoma) is by far the most common of all sun-caused skin cancers, but it usually presents as an obvious papule or nodule, often with telltale features such as pearly, rolled borders and focal erosion or ulceration. But there are exceptions, and the scar-like BCC is one.
SCC can also occasionally present in this manner, as can amelanotic melanoma, which is a colorless melanoma and very easy to miss. This case perfectly illustrates the point I often make to the students and residents I teach: When skin cancer is suspected, pay at least as much attention to the owner as to the lesion. Also, when in doubt, biopsy will settle the matter.
TREATMENT
For the patient, shave biopsy confirmed the presence of BCC. She was then referred for Mohs micrographic surgery because of the lesion’s size, location, and uncertain visible margins.
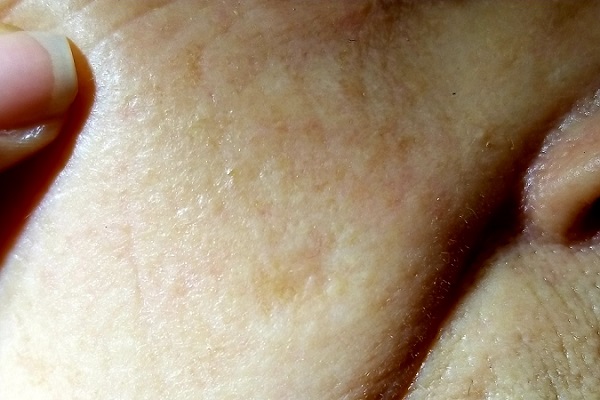
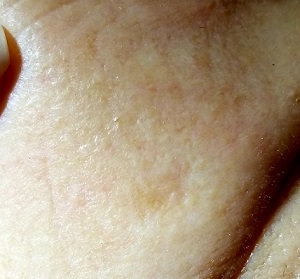
For several years, a 70-year-old woman has had an asymptomatic lesion on her cheek that has been growing slowly and steadily. Her primary care provider has reassured her at multiple visits that it should not cause her worry. Still, because of the lesion’s continued growth and her history of excessive sun exposure when she was young, she self-refers to dermatology for evaluation.
The patient has no history of skin cancer but her 2 sisters do, including a recent diagnosis of melanoma for one of them. During the 1950s, the 3 sisters were often outdoors—all burning easily and often and tanning only with difficulty. Since then, the sisters’ sun-drenched days have ended. All are in otherwise excellent health.
Examination reveals a patient with quite fair (type 2) skin and blue eyes. There is abundant evidence of past overexposure to ultraviolet light, including a weathered effect, scattered telangiectasias, and patches of white mottled skin (otherwise known as solar elastosis).
The lesion in question is quite faint and difficult to see. Magnification shows a 2-cm round patch that is slightly lighter than the surrounding skin and completely macular. No induration is felt on palpation, and no nodes are detected in the region.
An even closer and meticulous examination reveals that the surface adnexal structures—such as pores, skin lines, and even tiny hairs—that should be inside the lesion are completely missing. Slightly yellowish discoloration can be seen in the center of the patch. The rest of her skin shows no other worrisome features or lesions.
Upadacitinib looks effective for psoriatic arthritis
Upadacitinib (Rinvoq) improves joint and skin symptoms in patients with psoriatic arthritis for whom at least one other disease-modifying antirheumatic drug (DMARD) didn’t work or wasn’t well tolerated, a pair of phase 3 trials suggests.
“In psoriatic arthritis patients, there’s still a high proportion of patients who do not respond to traditional, nonbiologic DMARDs, so there’s room for improvement,” said Marina Magrey, MD, from the MetroHealth Medical Center, Case Western Reserve University School of Medicine, in Cleveland.
She and her colleagues evaluated the JAK inhibitor, already approved for rheumatoid arthritis in the United States, in the SELECT-PsA 1 and SELECT-PsA 2 trials, which followed more than 2,300 patients with psoriatic arthritis for an average of 6-10 years.
No safety signals emerged for upadacitinib in either trial that weren’t already seen in patients with rheumatoid arthritis, the investigators report, although a lower dose appeared to prompt fewer adverse events.
The research adds upadacitinib “to the armamentarium of medications we have against psoriatic arthritis,” said Dr. Magrey, who is a SELECT-PsA 1 investigator.
“The advantage of this medication is it’s available orally, so the convenience is there. It will enable both patients and physicians to choose from efficacious medications,” she told Medscape Medical News.
The team was “pleasantly surprised by the magnitude and rapidity of effect” of upadacitinib in study participants, said Philip Mease, MD, from the Swedish Medical Center and the University of Washington in Seattle, who is lead investigator for SELECT-PsA 2.
“It’s important to be able to understand if there’s adequate effectiveness in patients who’ve already been around the block several times with other treatments,” Dr. Mease told Medscape Medical News. “This trial demonstrated there was a high degree of effectiveness in each of the clinical domains” of psoriatic arthritis.
Results from both studies were presented at the virtual European League Against Rheumatism 2020 Congress.
SELECT-PsA 1
In SELECT-PsA 1, upadacitinib was compared with adalimumab and placebo in 1705 patients who previously had an inadequate response or intolerance to at least one nonbiologic DMARD. Participants were randomized to receive upadacitinib – 15 mg or 30 mg once daily – adalimumab 40 mg every other week, or placebo.
The primary endpoint was an improvement of at least 20% (ACR20) at week 12.
Secondary endpoints included change in Health Assessment Questionnaire Disability Index (HAQ-DI) score and change in patient assessment of pain on a numeric rating scale from baseline to week 12, achievement of ACR50 and ACR70 at week 12, and achievement of ACR20 at week 2.
Treatment-related adverse events were reported out to week 24 for patients who received at least one dose of upadacitinib.
Improvement in musculoskeletal symptoms, psoriasis, pain, physical function, and fatigue were seen by week 2 in both upadacitinib groups. At week 12, both doses of upadacitinib were noninferior to adalimumab for the achievement of ACR20 (P < .001), and the 30-mg dose was superior to adalimumab (P < .001).
More patients in the upadacitinib groups than in the placebo group met the stringent criteria for disease control, which included the achievement of minimal disease activity, ACR50, and ACR70.
The difference in effectiveness between the two doses of upadacitinib was small, but “there were relatively more adverse events,” such as infections, in the 30-mg group, Dr. Magrey reported, “so 15 mg seems like it will be the dose to go toward FDA approval.”
SELECT-PsA 2
SELECT-PsA 2 compared upadacitinib – 15 mg or 30 mg once daily – with placebo in 641 patients who previously had an inadequate response or intolerance to one or more biologic DMARDs.
The primary endpoint was the achievement of ACR20 at week 12.
Among the many secondary endpoints were a 75% improvement in Psoriasis Area and Severity Index score (PASI 75) at week 16, change in Self-Assessment of Psoriasis Symptoms (SAPS) score from baseline to week 16, the achievement of minimal disease activity at week 24, the achievement of ACR50 and ACR70 at week 12, and the achievement of ACR20 at week 2.
Adverse events were reported for patients who received at least one dose of upadacitinib.
At week 12, ACR20 was achieved by significantly more patients in the 15 mg and 30 mg upadacitinib groups than in the placebo group (56.9% vs. 63.8% vs. 24.1%; P < .0001), as was ACR50 (31.8% vs. 37.6% vs. 4.1%; P < .0001) and ACR70 (8.5% vs. 16.5% vs. 0.5%; P < .0001). In addition, all secondary endpoints were significantly better with upadacitinib than with placebo.
Rates of adverse events were similar in the 15 mg upadacitinib and placebo groups, but the rate was higher in the 30 mg upadacitinib group, including for herpes zoster.
“I was pleasantly surprised by the overall safety profile,” Dr. Mease said. “Yes, you need to pay attention to the potential for infection, but rates of serious infection were very low.”
“We didn’t see opportunistic infections occurring, and the overall adverse-events profile was one where we could be pretty reassuring with patients when introducing the medication and mechanism of action,” he added.
Upadacitinib appears to have significantly improved PASI scores in both trials, which is surprising, said Christopher Ritchlin, MD, from the University of Rochester Medical Center in New York.
“I think the data indicate that upadacitinib is a viable drug for treatment of psoriatic arthritis,” he told Medscape Medical News. “I don’t think it’s going to be tested in psoriasis, but for those with psoriatic arthritis and those whose burden of psoriasis is not particularly elevated, this drug looks like it might be very helpful to practicing physicians and their patients.”
Dr. Ritchlin added that he hopes future research will address whether upadacitinib is effective for axial disease in psoriatic arthritis, which wasn’t measured in these trials.
“I don’t see this as a weakness” of the current research, he said, but “having some spinal measures would be helpful. It’s something additional we’d like to know.”
Both trials were funded by AbbVie. Dr. Magrey reports financial relationships with Amgen, AbbVie, UCB Pharma, Novartis, Eli Lilly, Pfizer, and Janssen. Dr. Mease reports financial relationships with Abbott, Amgen, Biogen, BMS, Celgene Corporation, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, UCB, Genentech, and Janssen. Dr. Ritchlin has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Upadacitinib (Rinvoq) improves joint and skin symptoms in patients with psoriatic arthritis for whom at least one other disease-modifying antirheumatic drug (DMARD) didn’t work or wasn’t well tolerated, a pair of phase 3 trials suggests.
“In psoriatic arthritis patients, there’s still a high proportion of patients who do not respond to traditional, nonbiologic DMARDs, so there’s room for improvement,” said Marina Magrey, MD, from the MetroHealth Medical Center, Case Western Reserve University School of Medicine, in Cleveland.
She and her colleagues evaluated the JAK inhibitor, already approved for rheumatoid arthritis in the United States, in the SELECT-PsA 1 and SELECT-PsA 2 trials, which followed more than 2,300 patients with psoriatic arthritis for an average of 6-10 years.
No safety signals emerged for upadacitinib in either trial that weren’t already seen in patients with rheumatoid arthritis, the investigators report, although a lower dose appeared to prompt fewer adverse events.
The research adds upadacitinib “to the armamentarium of medications we have against psoriatic arthritis,” said Dr. Magrey, who is a SELECT-PsA 1 investigator.
“The advantage of this medication is it’s available orally, so the convenience is there. It will enable both patients and physicians to choose from efficacious medications,” she told Medscape Medical News.
The team was “pleasantly surprised by the magnitude and rapidity of effect” of upadacitinib in study participants, said Philip Mease, MD, from the Swedish Medical Center and the University of Washington in Seattle, who is lead investigator for SELECT-PsA 2.
“It’s important to be able to understand if there’s adequate effectiveness in patients who’ve already been around the block several times with other treatments,” Dr. Mease told Medscape Medical News. “This trial demonstrated there was a high degree of effectiveness in each of the clinical domains” of psoriatic arthritis.
Results from both studies were presented at the virtual European League Against Rheumatism 2020 Congress.
SELECT-PsA 1
In SELECT-PsA 1, upadacitinib was compared with adalimumab and placebo in 1705 patients who previously had an inadequate response or intolerance to at least one nonbiologic DMARD. Participants were randomized to receive upadacitinib – 15 mg or 30 mg once daily – adalimumab 40 mg every other week, or placebo.
The primary endpoint was an improvement of at least 20% (ACR20) at week 12.
Secondary endpoints included change in Health Assessment Questionnaire Disability Index (HAQ-DI) score and change in patient assessment of pain on a numeric rating scale from baseline to week 12, achievement of ACR50 and ACR70 at week 12, and achievement of ACR20 at week 2.
Treatment-related adverse events were reported out to week 24 for patients who received at least one dose of upadacitinib.
Improvement in musculoskeletal symptoms, psoriasis, pain, physical function, and fatigue were seen by week 2 in both upadacitinib groups. At week 12, both doses of upadacitinib were noninferior to adalimumab for the achievement of ACR20 (P < .001), and the 30-mg dose was superior to adalimumab (P < .001).
More patients in the upadacitinib groups than in the placebo group met the stringent criteria for disease control, which included the achievement of minimal disease activity, ACR50, and ACR70.
The difference in effectiveness between the two doses of upadacitinib was small, but “there were relatively more adverse events,” such as infections, in the 30-mg group, Dr. Magrey reported, “so 15 mg seems like it will be the dose to go toward FDA approval.”
SELECT-PsA 2
SELECT-PsA 2 compared upadacitinib – 15 mg or 30 mg once daily – with placebo in 641 patients who previously had an inadequate response or intolerance to one or more biologic DMARDs.
The primary endpoint was the achievement of ACR20 at week 12.
Among the many secondary endpoints were a 75% improvement in Psoriasis Area and Severity Index score (PASI 75) at week 16, change in Self-Assessment of Psoriasis Symptoms (SAPS) score from baseline to week 16, the achievement of minimal disease activity at week 24, the achievement of ACR50 and ACR70 at week 12, and the achievement of ACR20 at week 2.
Adverse events were reported for patients who received at least one dose of upadacitinib.
At week 12, ACR20 was achieved by significantly more patients in the 15 mg and 30 mg upadacitinib groups than in the placebo group (56.9% vs. 63.8% vs. 24.1%; P < .0001), as was ACR50 (31.8% vs. 37.6% vs. 4.1%; P < .0001) and ACR70 (8.5% vs. 16.5% vs. 0.5%; P < .0001). In addition, all secondary endpoints were significantly better with upadacitinib than with placebo.
Rates of adverse events were similar in the 15 mg upadacitinib and placebo groups, but the rate was higher in the 30 mg upadacitinib group, including for herpes zoster.
“I was pleasantly surprised by the overall safety profile,” Dr. Mease said. “Yes, you need to pay attention to the potential for infection, but rates of serious infection were very low.”
“We didn’t see opportunistic infections occurring, and the overall adverse-events profile was one where we could be pretty reassuring with patients when introducing the medication and mechanism of action,” he added.
Upadacitinib appears to have significantly improved PASI scores in both trials, which is surprising, said Christopher Ritchlin, MD, from the University of Rochester Medical Center in New York.
“I think the data indicate that upadacitinib is a viable drug for treatment of psoriatic arthritis,” he told Medscape Medical News. “I don’t think it’s going to be tested in psoriasis, but for those with psoriatic arthritis and those whose burden of psoriasis is not particularly elevated, this drug looks like it might be very helpful to practicing physicians and their patients.”
Dr. Ritchlin added that he hopes future research will address whether upadacitinib is effective for axial disease in psoriatic arthritis, which wasn’t measured in these trials.
“I don’t see this as a weakness” of the current research, he said, but “having some spinal measures would be helpful. It’s something additional we’d like to know.”
Both trials were funded by AbbVie. Dr. Magrey reports financial relationships with Amgen, AbbVie, UCB Pharma, Novartis, Eli Lilly, Pfizer, and Janssen. Dr. Mease reports financial relationships with Abbott, Amgen, Biogen, BMS, Celgene Corporation, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, UCB, Genentech, and Janssen. Dr. Ritchlin has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Upadacitinib (Rinvoq) improves joint and skin symptoms in patients with psoriatic arthritis for whom at least one other disease-modifying antirheumatic drug (DMARD) didn’t work or wasn’t well tolerated, a pair of phase 3 trials suggests.
“In psoriatic arthritis patients, there’s still a high proportion of patients who do not respond to traditional, nonbiologic DMARDs, so there’s room for improvement,” said Marina Magrey, MD, from the MetroHealth Medical Center, Case Western Reserve University School of Medicine, in Cleveland.
She and her colleagues evaluated the JAK inhibitor, already approved for rheumatoid arthritis in the United States, in the SELECT-PsA 1 and SELECT-PsA 2 trials, which followed more than 2,300 patients with psoriatic arthritis for an average of 6-10 years.
No safety signals emerged for upadacitinib in either trial that weren’t already seen in patients with rheumatoid arthritis, the investigators report, although a lower dose appeared to prompt fewer adverse events.
The research adds upadacitinib “to the armamentarium of medications we have against psoriatic arthritis,” said Dr. Magrey, who is a SELECT-PsA 1 investigator.
“The advantage of this medication is it’s available orally, so the convenience is there. It will enable both patients and physicians to choose from efficacious medications,” she told Medscape Medical News.
The team was “pleasantly surprised by the magnitude and rapidity of effect” of upadacitinib in study participants, said Philip Mease, MD, from the Swedish Medical Center and the University of Washington in Seattle, who is lead investigator for SELECT-PsA 2.
“It’s important to be able to understand if there’s adequate effectiveness in patients who’ve already been around the block several times with other treatments,” Dr. Mease told Medscape Medical News. “This trial demonstrated there was a high degree of effectiveness in each of the clinical domains” of psoriatic arthritis.
Results from both studies were presented at the virtual European League Against Rheumatism 2020 Congress.
SELECT-PsA 1
In SELECT-PsA 1, upadacitinib was compared with adalimumab and placebo in 1705 patients who previously had an inadequate response or intolerance to at least one nonbiologic DMARD. Participants were randomized to receive upadacitinib – 15 mg or 30 mg once daily – adalimumab 40 mg every other week, or placebo.
The primary endpoint was an improvement of at least 20% (ACR20) at week 12.
Secondary endpoints included change in Health Assessment Questionnaire Disability Index (HAQ-DI) score and change in patient assessment of pain on a numeric rating scale from baseline to week 12, achievement of ACR50 and ACR70 at week 12, and achievement of ACR20 at week 2.
Treatment-related adverse events were reported out to week 24 for patients who received at least one dose of upadacitinib.
Improvement in musculoskeletal symptoms, psoriasis, pain, physical function, and fatigue were seen by week 2 in both upadacitinib groups. At week 12, both doses of upadacitinib were noninferior to adalimumab for the achievement of ACR20 (P < .001), and the 30-mg dose was superior to adalimumab (P < .001).
More patients in the upadacitinib groups than in the placebo group met the stringent criteria for disease control, which included the achievement of minimal disease activity, ACR50, and ACR70.
The difference in effectiveness between the two doses of upadacitinib was small, but “there were relatively more adverse events,” such as infections, in the 30-mg group, Dr. Magrey reported, “so 15 mg seems like it will be the dose to go toward FDA approval.”
SELECT-PsA 2
SELECT-PsA 2 compared upadacitinib – 15 mg or 30 mg once daily – with placebo in 641 patients who previously had an inadequate response or intolerance to one or more biologic DMARDs.
The primary endpoint was the achievement of ACR20 at week 12.
Among the many secondary endpoints were a 75% improvement in Psoriasis Area and Severity Index score (PASI 75) at week 16, change in Self-Assessment of Psoriasis Symptoms (SAPS) score from baseline to week 16, the achievement of minimal disease activity at week 24, the achievement of ACR50 and ACR70 at week 12, and the achievement of ACR20 at week 2.
Adverse events were reported for patients who received at least one dose of upadacitinib.
At week 12, ACR20 was achieved by significantly more patients in the 15 mg and 30 mg upadacitinib groups than in the placebo group (56.9% vs. 63.8% vs. 24.1%; P < .0001), as was ACR50 (31.8% vs. 37.6% vs. 4.1%; P < .0001) and ACR70 (8.5% vs. 16.5% vs. 0.5%; P < .0001). In addition, all secondary endpoints were significantly better with upadacitinib than with placebo.
Rates of adverse events were similar in the 15 mg upadacitinib and placebo groups, but the rate was higher in the 30 mg upadacitinib group, including for herpes zoster.
“I was pleasantly surprised by the overall safety profile,” Dr. Mease said. “Yes, you need to pay attention to the potential for infection, but rates of serious infection were very low.”
“We didn’t see opportunistic infections occurring, and the overall adverse-events profile was one where we could be pretty reassuring with patients when introducing the medication and mechanism of action,” he added.
Upadacitinib appears to have significantly improved PASI scores in both trials, which is surprising, said Christopher Ritchlin, MD, from the University of Rochester Medical Center in New York.
“I think the data indicate that upadacitinib is a viable drug for treatment of psoriatic arthritis,” he told Medscape Medical News. “I don’t think it’s going to be tested in psoriasis, but for those with psoriatic arthritis and those whose burden of psoriasis is not particularly elevated, this drug looks like it might be very helpful to practicing physicians and their patients.”
Dr. Ritchlin added that he hopes future research will address whether upadacitinib is effective for axial disease in psoriatic arthritis, which wasn’t measured in these trials.
“I don’t see this as a weakness” of the current research, he said, but “having some spinal measures would be helpful. It’s something additional we’d like to know.”
Both trials were funded by AbbVie. Dr. Magrey reports financial relationships with Amgen, AbbVie, UCB Pharma, Novartis, Eli Lilly, Pfizer, and Janssen. Dr. Mease reports financial relationships with Abbott, Amgen, Biogen, BMS, Celgene Corporation, Eli Lilly, Novartis, Pfizer, Sun Pharmaceutical, UCB, Genentech, and Janssen. Dr. Ritchlin has disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
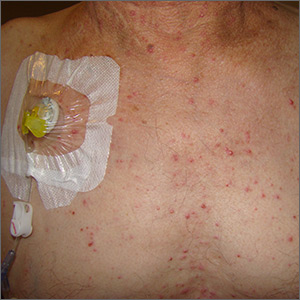
Diffuse pustules
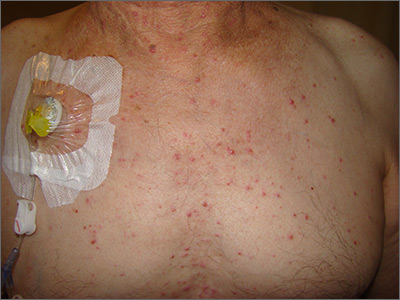
The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.

The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The presence of these sterile pustules with an erythematous base led to a diagnosis of acute generalized exanthematous pustulosis (AGEP), also known as a pustular drug eruption. Although pustules are present, AGEP is an allergic response to medications and not an infection.
AGEP can be associated with fever and leukocytosis. Interestingly, antibiotics are a frequent cause—not a treatment—since the pustules are sterile. It also is worth noting, in light of the COVID-19 pandemic, that hydroxychloroquine use has been linked to AGEP, although the number of cases cited in the literature is small.
Treatment is avoidance of the offending medication and symptomatic care. AGEP typically will resolve approximately 2 weeks after discontinuing the medication causing the reaction. Systemic steroids also may be used for treatment in severe cases.
This patient had a history of repeated episodes with his chemotherapy regimen, so he was treated symptomatically with diphenhydramine for the itching. Since chemotherapy was a priority to treat his colon cancer, avoidance of the offending agent was not an option. The Family Medicine Service recommended pretreatment with diphenhydramine 25 to 50 mg orally or intravenously for future rounds of chemotherapy to blunt future responses.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.
Mercogliano C, Khan M, Lin C, et al. AGEP overlap induced by hydroxychloroquine: a case report and literature review. J Community Hosp Intern Med Perspect. 2018;8:360-362.
24-year-old man • prednisone therapy for nephrotic syndrome • diffuse maculopapular rash • pruritis
THE CASE
A 24-year-old man with no past medical history was referred to a nephrologist for a 5-month history of leg swelling and weight gain. His only medication was furosemide 40 mg/d, prescribed by his primary care physician. His physical examination was unremarkable except for lower extremity and scrotal edema.
Laboratory values included a creatinine of 0.8 mg/dL (reference range, 0.6 to 1.2 mg/dL); hemoglobin concentration, 14.4 g/dL (reference range, 14 to 18 g/dL); albumin, 1.9 g/dL (reference range, 3.5 to 5.5 g/dL); and glucose, 80 mg/dL (reference range, 74 to 106 mg/dL). Electrolyte levels were normal. Urinalysis revealed 3+ blood and 4+ protein on dipstick, as well as the presence of granular and lipid casts on microscopic exam. A 24-hour urine collection contained 10.5 g of protein. Antinuclear antibody titers, complement levels, hepatitis serologies, and antineutrophil cytoplasmic antibody titers were all normal.
A renal biopsy revealed idiopathic focal segmental glomerulosclerosis. The patient was started on oral prednisone 40 mg twice daily.
Two days later, he developed a diffuse pruritic maculopapular rash. He stopped taking the prednisone, and the rash resolved over the next 3 to 5 days. He was then instructed to restart the prednisone for his nephrotic syndrome. When he developed a new but similar rash, the prednisone was discontinued. The rash again resolved.
THE DIAGNOSIS
Since the patient had already been taking furosemide for 6 weeks without an adverse reaction, it was presumed that the prednisone tablet was causing his rash. It would be unusual for prednisone itself to cause a drug eruption, so an additive or coloring agent in the tablet was thought to be responsible for the reaction.
We noted that the patient had been taking a 20-mg orange tablet of prednisone. So we opted to “tweak” the prescription and prescribe the same daily dose but in the form of 10-mg white tablets. The patient tolerated this new regimen without any adverse effects and completed a full 9 months of prednisone therapy without any recurrence of skin lesions. His glomerular disease went into remission.
DISCUSSION
Excipients are inert substances that are added to a food or drug to provide the desired consistency, appearance, or form. They are also used as a preservative for substance stabilization.
Continue to: There are many reports in the literature...
There are many reports in the literature of adverse reactions to excipients.1-3 These include skin rashes induced by the coloring agent in the capsule shell of rifampicin2 and a rash that developed from a coloring agent in oral iron.3 Other reports have noted dyes in foods and even toothpaste as triggers.4,5
Hypersensitivity. Although a specific reaction to prednisone was considered unlikely in this case, type IV delayed hypersensitivity reactions to corticosteroids have been reported. The most common type of corticosteroid-related allergy is contact dermatitis associated with topical corticosteroid use.6 Many cases of delayed maculopapular reactions are thought to be T-cell–mediated type IV reactions.6
Type I immediate hypersensitivity reactions to corticosteroids are also well documented. In a literature review of 120 immediate hypersensitivity reactions to corticosteroids, anaphylactic symptoms were more commonly reported than urticaria or angioedema.7 Intravenous exposure was most frequently associated with reactions, followed by the intra-articular and oral routes of administration.7
Causative agents. The same literature review identified methylprednisolone as the most common steroid to cause a reaction; dexamethasone and prednisone were the least frequently associated with reactions.7 Pharmacologically inactive ingredients were implicated in 28% of the corticosteroid hypersensitivity reactions.7
Additives suspected to be triggers include succinate and phosphate esters, carboxymethylcellulose, polyethylene glycol, and lactose. Interestingly, there have been reports of acute allergic reactions to methylprednisolone sodium succinate 40 mg/mL intravenous preparation in children with milk allergy, due to lactose contaminated with milk protein.8,9
Continue to: Yellow dye was to blame
Yellow dye was to blame. In our case, the 20-mg tablet that the patient had been taking contained the coloring agent FD&C yellow #6, an azo dye also known as sunset yellow or E-110 in Europe. Several reports have described adverse reactions to this coloring agent.1,3 There were other additives in the 20-mg tablet, but a comparison revealed that the 10-mg tablet contained identical substances—but no dye. Thus, it was most likely that the coloring agent was the cause of the patient’s probable type IV exanthematous drug reaction.
Our patient
The patient was instructed to avoid all medications and food containing FD&C yellow #6. No formal allergy testing or re-challenge was performed, since the patient did well under the care of his nephrologist.
THE TAKEAWAY
It’s important to recognize that adverse drug reactions can occur from any medication—not only from the drug itself, but also from excipients contained within. This case reminds us that when a patient complains of an adverse effect to a medication, dyes and inactive ingredients need to be considered as possible inciting agents.
CORRESPONDENCE
Neil E. Soifer, MD, Lakeside Nephrology, 2277 West Howard, Chicago, IL 60645; [email protected]
1. Swerlick RA, Campbell CF. Medication dyes as a source of drug allergy. J Drugs Dermatol. 2013;12:99-102.
2. Calişkaner Z, Oztürk S, Karaayvaz M. Not all adverse drug reactions originate from active component: coloring agent-induced skin eruption in a patient treated with rifampicin. Allergy. 2003;58:1077-1079.
3. Rogkakou A, Guerra L, Scordamaglia A, et al. Severe skin reaction to excipients of an oral iron treatment. Allergy. 2007;62:334-335.
4. Zaknun D, Schroecksnadel S, Kurz K, et al. Potential role of antioxidant food supplements, preservatives and colorants in the pathogenesis of allergy and asthma. Int Arch Allergy Immunol. 2012;157:113-124.
5. Barbaud A. Place of excipients in systemic drug allergy. Immunol Allergy Clin N Am. 2014;34:671-679.
6. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273.
7. Patel A, Bahna S. Immediate hypersensitivity reactions to corticosteroids. Ann Allergy Asthma Immunol. 2015;115:178-182.
8. Eda A, Sugai K, Shioya H, et al. Acute allergic reaction due to milk proteins contaminating lactose added to corticosteroid for injection. Allergol Int. 2009;58:137-139.
9. Levy Y, Segal N, Nahum A, et al. Hypersensitivity to methylprednisolone sodium succinate in children with milk allergy. J Allergy Clin Immunol Pract. 2014;2:471-474.
THE CASE
A 24-year-old man with no past medical history was referred to a nephrologist for a 5-month history of leg swelling and weight gain. His only medication was furosemide 40 mg/d, prescribed by his primary care physician. His physical examination was unremarkable except for lower extremity and scrotal edema.
Laboratory values included a creatinine of 0.8 mg/dL (reference range, 0.6 to 1.2 mg/dL); hemoglobin concentration, 14.4 g/dL (reference range, 14 to 18 g/dL); albumin, 1.9 g/dL (reference range, 3.5 to 5.5 g/dL); and glucose, 80 mg/dL (reference range, 74 to 106 mg/dL). Electrolyte levels were normal. Urinalysis revealed 3+ blood and 4+ protein on dipstick, as well as the presence of granular and lipid casts on microscopic exam. A 24-hour urine collection contained 10.5 g of protein. Antinuclear antibody titers, complement levels, hepatitis serologies, and antineutrophil cytoplasmic antibody titers were all normal.
A renal biopsy revealed idiopathic focal segmental glomerulosclerosis. The patient was started on oral prednisone 40 mg twice daily.
Two days later, he developed a diffuse pruritic maculopapular rash. He stopped taking the prednisone, and the rash resolved over the next 3 to 5 days. He was then instructed to restart the prednisone for his nephrotic syndrome. When he developed a new but similar rash, the prednisone was discontinued. The rash again resolved.
THE DIAGNOSIS
Since the patient had already been taking furosemide for 6 weeks without an adverse reaction, it was presumed that the prednisone tablet was causing his rash. It would be unusual for prednisone itself to cause a drug eruption, so an additive or coloring agent in the tablet was thought to be responsible for the reaction.
We noted that the patient had been taking a 20-mg orange tablet of prednisone. So we opted to “tweak” the prescription and prescribe the same daily dose but in the form of 10-mg white tablets. The patient tolerated this new regimen without any adverse effects and completed a full 9 months of prednisone therapy without any recurrence of skin lesions. His glomerular disease went into remission.
DISCUSSION
Excipients are inert substances that are added to a food or drug to provide the desired consistency, appearance, or form. They are also used as a preservative for substance stabilization.
Continue to: There are many reports in the literature...
There are many reports in the literature of adverse reactions to excipients.1-3 These include skin rashes induced by the coloring agent in the capsule shell of rifampicin2 and a rash that developed from a coloring agent in oral iron.3 Other reports have noted dyes in foods and even toothpaste as triggers.4,5
Hypersensitivity. Although a specific reaction to prednisone was considered unlikely in this case, type IV delayed hypersensitivity reactions to corticosteroids have been reported. The most common type of corticosteroid-related allergy is contact dermatitis associated with topical corticosteroid use.6 Many cases of delayed maculopapular reactions are thought to be T-cell–mediated type IV reactions.6
Type I immediate hypersensitivity reactions to corticosteroids are also well documented. In a literature review of 120 immediate hypersensitivity reactions to corticosteroids, anaphylactic symptoms were more commonly reported than urticaria or angioedema.7 Intravenous exposure was most frequently associated with reactions, followed by the intra-articular and oral routes of administration.7
Causative agents. The same literature review identified methylprednisolone as the most common steroid to cause a reaction; dexamethasone and prednisone were the least frequently associated with reactions.7 Pharmacologically inactive ingredients were implicated in 28% of the corticosteroid hypersensitivity reactions.7
Additives suspected to be triggers include succinate and phosphate esters, carboxymethylcellulose, polyethylene glycol, and lactose. Interestingly, there have been reports of acute allergic reactions to methylprednisolone sodium succinate 40 mg/mL intravenous preparation in children with milk allergy, due to lactose contaminated with milk protein.8,9
Continue to: Yellow dye was to blame
Yellow dye was to blame. In our case, the 20-mg tablet that the patient had been taking contained the coloring agent FD&C yellow #6, an azo dye also known as sunset yellow or E-110 in Europe. Several reports have described adverse reactions to this coloring agent.1,3 There were other additives in the 20-mg tablet, but a comparison revealed that the 10-mg tablet contained identical substances—but no dye. Thus, it was most likely that the coloring agent was the cause of the patient’s probable type IV exanthematous drug reaction.
Our patient
The patient was instructed to avoid all medications and food containing FD&C yellow #6. No formal allergy testing or re-challenge was performed, since the patient did well under the care of his nephrologist.
THE TAKEAWAY
It’s important to recognize that adverse drug reactions can occur from any medication—not only from the drug itself, but also from excipients contained within. This case reminds us that when a patient complains of an adverse effect to a medication, dyes and inactive ingredients need to be considered as possible inciting agents.
CORRESPONDENCE
Neil E. Soifer, MD, Lakeside Nephrology, 2277 West Howard, Chicago, IL 60645; [email protected]
THE CASE
A 24-year-old man with no past medical history was referred to a nephrologist for a 5-month history of leg swelling and weight gain. His only medication was furosemide 40 mg/d, prescribed by his primary care physician. His physical examination was unremarkable except for lower extremity and scrotal edema.
Laboratory values included a creatinine of 0.8 mg/dL (reference range, 0.6 to 1.2 mg/dL); hemoglobin concentration, 14.4 g/dL (reference range, 14 to 18 g/dL); albumin, 1.9 g/dL (reference range, 3.5 to 5.5 g/dL); and glucose, 80 mg/dL (reference range, 74 to 106 mg/dL). Electrolyte levels were normal. Urinalysis revealed 3+ blood and 4+ protein on dipstick, as well as the presence of granular and lipid casts on microscopic exam. A 24-hour urine collection contained 10.5 g of protein. Antinuclear antibody titers, complement levels, hepatitis serologies, and antineutrophil cytoplasmic antibody titers were all normal.
A renal biopsy revealed idiopathic focal segmental glomerulosclerosis. The patient was started on oral prednisone 40 mg twice daily.
Two days later, he developed a diffuse pruritic maculopapular rash. He stopped taking the prednisone, and the rash resolved over the next 3 to 5 days. He was then instructed to restart the prednisone for his nephrotic syndrome. When he developed a new but similar rash, the prednisone was discontinued. The rash again resolved.
THE DIAGNOSIS
Since the patient had already been taking furosemide for 6 weeks without an adverse reaction, it was presumed that the prednisone tablet was causing his rash. It would be unusual for prednisone itself to cause a drug eruption, so an additive or coloring agent in the tablet was thought to be responsible for the reaction.
We noted that the patient had been taking a 20-mg orange tablet of prednisone. So we opted to “tweak” the prescription and prescribe the same daily dose but in the form of 10-mg white tablets. The patient tolerated this new regimen without any adverse effects and completed a full 9 months of prednisone therapy without any recurrence of skin lesions. His glomerular disease went into remission.
DISCUSSION
Excipients are inert substances that are added to a food or drug to provide the desired consistency, appearance, or form. They are also used as a preservative for substance stabilization.
Continue to: There are many reports in the literature...
There are many reports in the literature of adverse reactions to excipients.1-3 These include skin rashes induced by the coloring agent in the capsule shell of rifampicin2 and a rash that developed from a coloring agent in oral iron.3 Other reports have noted dyes in foods and even toothpaste as triggers.4,5
Hypersensitivity. Although a specific reaction to prednisone was considered unlikely in this case, type IV delayed hypersensitivity reactions to corticosteroids have been reported. The most common type of corticosteroid-related allergy is contact dermatitis associated with topical corticosteroid use.6 Many cases of delayed maculopapular reactions are thought to be T-cell–mediated type IV reactions.6
Type I immediate hypersensitivity reactions to corticosteroids are also well documented. In a literature review of 120 immediate hypersensitivity reactions to corticosteroids, anaphylactic symptoms were more commonly reported than urticaria or angioedema.7 Intravenous exposure was most frequently associated with reactions, followed by the intra-articular and oral routes of administration.7
Causative agents. The same literature review identified methylprednisolone as the most common steroid to cause a reaction; dexamethasone and prednisone were the least frequently associated with reactions.7 Pharmacologically inactive ingredients were implicated in 28% of the corticosteroid hypersensitivity reactions.7
Additives suspected to be triggers include succinate and phosphate esters, carboxymethylcellulose, polyethylene glycol, and lactose. Interestingly, there have been reports of acute allergic reactions to methylprednisolone sodium succinate 40 mg/mL intravenous preparation in children with milk allergy, due to lactose contaminated with milk protein.8,9
Continue to: Yellow dye was to blame
Yellow dye was to blame. In our case, the 20-mg tablet that the patient had been taking contained the coloring agent FD&C yellow #6, an azo dye also known as sunset yellow or E-110 in Europe. Several reports have described adverse reactions to this coloring agent.1,3 There were other additives in the 20-mg tablet, but a comparison revealed that the 10-mg tablet contained identical substances—but no dye. Thus, it was most likely that the coloring agent was the cause of the patient’s probable type IV exanthematous drug reaction.
Our patient
The patient was instructed to avoid all medications and food containing FD&C yellow #6. No formal allergy testing or re-challenge was performed, since the patient did well under the care of his nephrologist.
THE TAKEAWAY
It’s important to recognize that adverse drug reactions can occur from any medication—not only from the drug itself, but also from excipients contained within. This case reminds us that when a patient complains of an adverse effect to a medication, dyes and inactive ingredients need to be considered as possible inciting agents.
CORRESPONDENCE
Neil E. Soifer, MD, Lakeside Nephrology, 2277 West Howard, Chicago, IL 60645; [email protected]
1. Swerlick RA, Campbell CF. Medication dyes as a source of drug allergy. J Drugs Dermatol. 2013;12:99-102.
2. Calişkaner Z, Oztürk S, Karaayvaz M. Not all adverse drug reactions originate from active component: coloring agent-induced skin eruption in a patient treated with rifampicin. Allergy. 2003;58:1077-1079.
3. Rogkakou A, Guerra L, Scordamaglia A, et al. Severe skin reaction to excipients of an oral iron treatment. Allergy. 2007;62:334-335.
4. Zaknun D, Schroecksnadel S, Kurz K, et al. Potential role of antioxidant food supplements, preservatives and colorants in the pathogenesis of allergy and asthma. Int Arch Allergy Immunol. 2012;157:113-124.
5. Barbaud A. Place of excipients in systemic drug allergy. Immunol Allergy Clin N Am. 2014;34:671-679.
6. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273.
7. Patel A, Bahna S. Immediate hypersensitivity reactions to corticosteroids. Ann Allergy Asthma Immunol. 2015;115:178-182.
8. Eda A, Sugai K, Shioya H, et al. Acute allergic reaction due to milk proteins contaminating lactose added to corticosteroid for injection. Allergol Int. 2009;58:137-139.
9. Levy Y, Segal N, Nahum A, et al. Hypersensitivity to methylprednisolone sodium succinate in children with milk allergy. J Allergy Clin Immunol Pract. 2014;2:471-474.
1. Swerlick RA, Campbell CF. Medication dyes as a source of drug allergy. J Drugs Dermatol. 2013;12:99-102.
2. Calişkaner Z, Oztürk S, Karaayvaz M. Not all adverse drug reactions originate from active component: coloring agent-induced skin eruption in a patient treated with rifampicin. Allergy. 2003;58:1077-1079.
3. Rogkakou A, Guerra L, Scordamaglia A, et al. Severe skin reaction to excipients of an oral iron treatment. Allergy. 2007;62:334-335.
4. Zaknun D, Schroecksnadel S, Kurz K, et al. Potential role of antioxidant food supplements, preservatives and colorants in the pathogenesis of allergy and asthma. Int Arch Allergy Immunol. 2012;157:113-124.
5. Barbaud A. Place of excipients in systemic drug allergy. Immunol Allergy Clin N Am. 2014;34:671-679.
6. Joint Task Force on Practice Parameters; American Academy of Allergy, Asthma and Immunology; American College of Allergy, Asthma and Immunology; Joint Council of Allergy, Asthma and Immunology. Drug allergy: an updated practice parameter. Ann Allergy Asthma Immunol. 2010;105:259-273.
7. Patel A, Bahna S. Immediate hypersensitivity reactions to corticosteroids. Ann Allergy Asthma Immunol. 2015;115:178-182.
8. Eda A, Sugai K, Shioya H, et al. Acute allergic reaction due to milk proteins contaminating lactose added to corticosteroid for injection. Allergol Int. 2009;58:137-139.
9. Levy Y, Segal N, Nahum A, et al. Hypersensitivity to methylprednisolone sodium succinate in children with milk allergy. J Allergy Clin Immunol Pract. 2014;2:471-474.
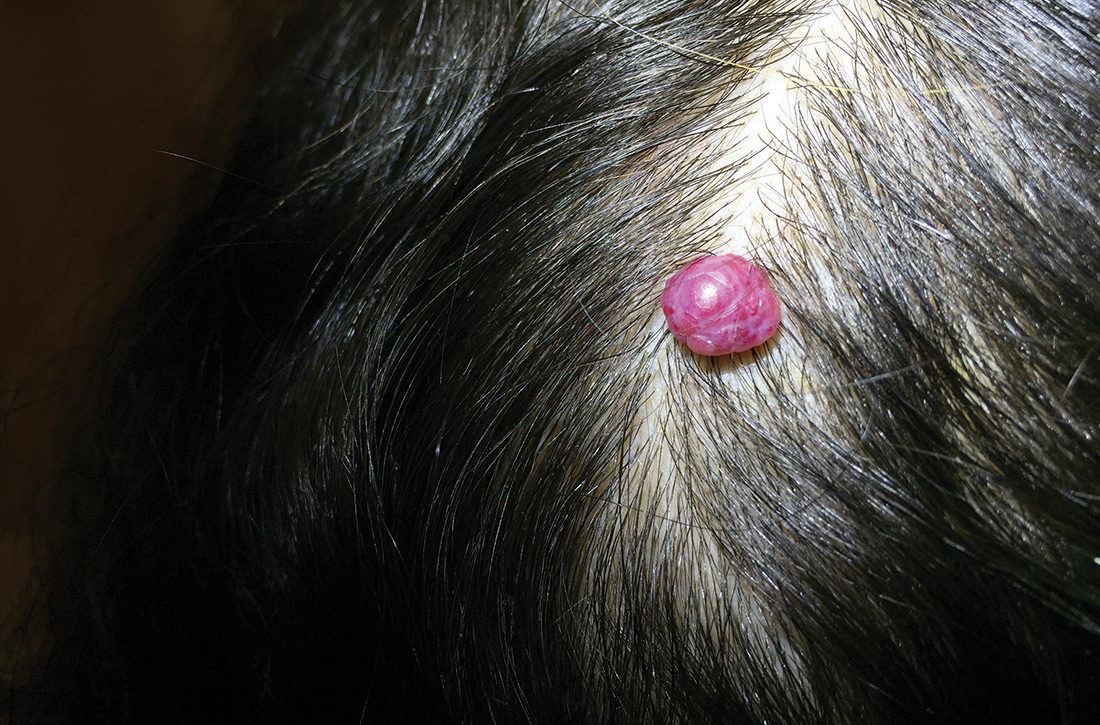
Growing scalp nodule
A 38-year-old woman presented to the primary care clinic with a growing nodule on her head (FIGURE) of 4 to 6 months’ duration. The nodule was painless but was getting caught on her hairbrush.

Physical exam revealed a firm 8 × 10-mm lobulated pink nodule near the vertex of her scalp. It did not bleed with manipulation or appear friable. There were no other lesions on the scalp or the rest of her body. A shave excision was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cherry hemangioma
A benign hemangioma was suspected; however, given its unusually large size and uncharacteristic location, other entities such as amelanotic melanoma and lobular capillary hemangioma (pyogenic granuloma) needed to be ruled out. Pathology following a shave excision (with electrocautery) confirmed that this was a cherry hemangioma.
Cherry hemangiomas, also known as senile hemangiomas or Campbell de Morgan spots, are a nearly ubiquitous benign vascular proliferation that increase in frequency and number with age.1,2 They also have been associated with pregnancy and some chemical exposures.3,4 In general, they are of no clinical consequence. Typically, they are 1- to 5-mm bright pink or bright to dark red papules located on the arms and trunk, a description that has persisted since at least 1947.1 Scalp involvement is considered rare.5
Differential includes malignant entities
The large size of the lesion in addition to its unusual location on the scalp prompted consideration of a malignant entity despite many features of a benign process.
Amelanotic melanomas classically are described as flesh-colored, but up to 70% of amelanotic melanomas may actually be red. Red amelanotic melanomas may account for nearly 4% of all melanomas and frequently are underrecognized.6 Pathology ruled out melanoma for this patient.
Lobular capillary hemangiomas (also known as pyogenic granulomas) typically manifest as rapidly growing, painless, friable papules or nodules in young adults and adolescents. Cutaneous lobular capillary hemangiomas are most often located on the head and neck, nose, face, extremities, and upper trunk. These benign lesions may grow to several centimeters in diameter and are prone to bleeding and ulceration, which this patient notably did not have.7
Continue to: Treatment often isn't required
Treatment often isn’t required
Most cherry hemangiomas are asymptomatic and small enough that they don’t catch on clothing or jewelry. For larger lesions, shave excision with or without electrocautery of the base may be performed. Curettage and laser therapy also have been used with success.5
The patient in this case had no recurrence or development of new cherry hemangiomas 2 years after her scalp lesion was removed.
CORRESPONDENCE
J. Lane Wilson, MD, East Carolina University Family Medicine, 101 Heart Drive, Greenville, NC 27834; [email protected]
1. Murison AR, Sutherland JW, Williamson AM. De Morgan spots. Br Med J. 1947;1:634-636.
2. Plunkett A, Merlin K, Gill D, et al. The frequency of common nonmalignant skin conditions in adults in central Victoria, Australia. Int J Dermatol. 1999;38:901-908.
3. Firooz A, Komeili A, Dowlati Y. Eruptive melanocytic nevi and cherry angiomas secondary to exposure to sulfur mustard gas. J Am Acad Dermatol. 1999;40:646-647.
4. Raymond LW, Williford LS, Burke WA. Eruptive cherry angiomas and irritant symptoms after one acute exposure to the glycol ether solvent 2-butoxyethanol. J Occup Environ Med. 1998;40:1059-1064.
5. Kim JH, Park H, Ahn SK. Cherry angiomas on the scalp. Case Rep Dermatol. 2009;1:82-86.
6. McClain SE, Mayo KB, Shada AL, et al. Amelanotic melanomas presenting as red skin lesions: a diagnostic challenge with potentially lethal consequences. Int J Dermatol. 2012;51:420-426.
7. Usatine R. Pyogenic granuloma. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill Medical; 2009:666-669.
A 38-year-old woman presented to the primary care clinic with a growing nodule on her head (FIGURE) of 4 to 6 months’ duration. The nodule was painless but was getting caught on her hairbrush.

Physical exam revealed a firm 8 × 10-mm lobulated pink nodule near the vertex of her scalp. It did not bleed with manipulation or appear friable. There were no other lesions on the scalp or the rest of her body. A shave excision was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cherry hemangioma
A benign hemangioma was suspected; however, given its unusually large size and uncharacteristic location, other entities such as amelanotic melanoma and lobular capillary hemangioma (pyogenic granuloma) needed to be ruled out. Pathology following a shave excision (with electrocautery) confirmed that this was a cherry hemangioma.
Cherry hemangiomas, also known as senile hemangiomas or Campbell de Morgan spots, are a nearly ubiquitous benign vascular proliferation that increase in frequency and number with age.1,2 They also have been associated with pregnancy and some chemical exposures.3,4 In general, they are of no clinical consequence. Typically, they are 1- to 5-mm bright pink or bright to dark red papules located on the arms and trunk, a description that has persisted since at least 1947.1 Scalp involvement is considered rare.5
Differential includes malignant entities
The large size of the lesion in addition to its unusual location on the scalp prompted consideration of a malignant entity despite many features of a benign process.
Amelanotic melanomas classically are described as flesh-colored, but up to 70% of amelanotic melanomas may actually be red. Red amelanotic melanomas may account for nearly 4% of all melanomas and frequently are underrecognized.6 Pathology ruled out melanoma for this patient.
Lobular capillary hemangiomas (also known as pyogenic granulomas) typically manifest as rapidly growing, painless, friable papules or nodules in young adults and adolescents. Cutaneous lobular capillary hemangiomas are most often located on the head and neck, nose, face, extremities, and upper trunk. These benign lesions may grow to several centimeters in diameter and are prone to bleeding and ulceration, which this patient notably did not have.7
Continue to: Treatment often isn't required
Treatment often isn’t required
Most cherry hemangiomas are asymptomatic and small enough that they don’t catch on clothing or jewelry. For larger lesions, shave excision with or without electrocautery of the base may be performed. Curettage and laser therapy also have been used with success.5
The patient in this case had no recurrence or development of new cherry hemangiomas 2 years after her scalp lesion was removed.
CORRESPONDENCE
J. Lane Wilson, MD, East Carolina University Family Medicine, 101 Heart Drive, Greenville, NC 27834; [email protected]
A 38-year-old woman presented to the primary care clinic with a growing nodule on her head (FIGURE) of 4 to 6 months’ duration. The nodule was painless but was getting caught on her hairbrush.

Physical exam revealed a firm 8 × 10-mm lobulated pink nodule near the vertex of her scalp. It did not bleed with manipulation or appear friable. There were no other lesions on the scalp or the rest of her body. A shave excision was performed.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Cherry hemangioma
A benign hemangioma was suspected; however, given its unusually large size and uncharacteristic location, other entities such as amelanotic melanoma and lobular capillary hemangioma (pyogenic granuloma) needed to be ruled out. Pathology following a shave excision (with electrocautery) confirmed that this was a cherry hemangioma.
Cherry hemangiomas, also known as senile hemangiomas or Campbell de Morgan spots, are a nearly ubiquitous benign vascular proliferation that increase in frequency and number with age.1,2 They also have been associated with pregnancy and some chemical exposures.3,4 In general, they are of no clinical consequence. Typically, they are 1- to 5-mm bright pink or bright to dark red papules located on the arms and trunk, a description that has persisted since at least 1947.1 Scalp involvement is considered rare.5
Differential includes malignant entities
The large size of the lesion in addition to its unusual location on the scalp prompted consideration of a malignant entity despite many features of a benign process.
Amelanotic melanomas classically are described as flesh-colored, but up to 70% of amelanotic melanomas may actually be red. Red amelanotic melanomas may account for nearly 4% of all melanomas and frequently are underrecognized.6 Pathology ruled out melanoma for this patient.
Lobular capillary hemangiomas (also known as pyogenic granulomas) typically manifest as rapidly growing, painless, friable papules or nodules in young adults and adolescents. Cutaneous lobular capillary hemangiomas are most often located on the head and neck, nose, face, extremities, and upper trunk. These benign lesions may grow to several centimeters in diameter and are prone to bleeding and ulceration, which this patient notably did not have.7
Continue to: Treatment often isn't required
Treatment often isn’t required
Most cherry hemangiomas are asymptomatic and small enough that they don’t catch on clothing or jewelry. For larger lesions, shave excision with or without electrocautery of the base may be performed. Curettage and laser therapy also have been used with success.5
The patient in this case had no recurrence or development of new cherry hemangiomas 2 years after her scalp lesion was removed.
CORRESPONDENCE
J. Lane Wilson, MD, East Carolina University Family Medicine, 101 Heart Drive, Greenville, NC 27834; [email protected]
1. Murison AR, Sutherland JW, Williamson AM. De Morgan spots. Br Med J. 1947;1:634-636.
2. Plunkett A, Merlin K, Gill D, et al. The frequency of common nonmalignant skin conditions in adults in central Victoria, Australia. Int J Dermatol. 1999;38:901-908.
3. Firooz A, Komeili A, Dowlati Y. Eruptive melanocytic nevi and cherry angiomas secondary to exposure to sulfur mustard gas. J Am Acad Dermatol. 1999;40:646-647.
4. Raymond LW, Williford LS, Burke WA. Eruptive cherry angiomas and irritant symptoms after one acute exposure to the glycol ether solvent 2-butoxyethanol. J Occup Environ Med. 1998;40:1059-1064.
5. Kim JH, Park H, Ahn SK. Cherry angiomas on the scalp. Case Rep Dermatol. 2009;1:82-86.
6. McClain SE, Mayo KB, Shada AL, et al. Amelanotic melanomas presenting as red skin lesions: a diagnostic challenge with potentially lethal consequences. Int J Dermatol. 2012;51:420-426.
7. Usatine R. Pyogenic granuloma. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill Medical; 2009:666-669.
1. Murison AR, Sutherland JW, Williamson AM. De Morgan spots. Br Med J. 1947;1:634-636.
2. Plunkett A, Merlin K, Gill D, et al. The frequency of common nonmalignant skin conditions in adults in central Victoria, Australia. Int J Dermatol. 1999;38:901-908.
3. Firooz A, Komeili A, Dowlati Y. Eruptive melanocytic nevi and cherry angiomas secondary to exposure to sulfur mustard gas. J Am Acad Dermatol. 1999;40:646-647.
4. Raymond LW, Williford LS, Burke WA. Eruptive cherry angiomas and irritant symptoms after one acute exposure to the glycol ether solvent 2-butoxyethanol. J Occup Environ Med. 1998;40:1059-1064.
5. Kim JH, Park H, Ahn SK. Cherry angiomas on the scalp. Case Rep Dermatol. 2009;1:82-86.
6. McClain SE, Mayo KB, Shada AL, et al. Amelanotic melanomas presenting as red skin lesions: a diagnostic challenge with potentially lethal consequences. Int J Dermatol. 2012;51:420-426.
7. Usatine R. Pyogenic granuloma. The Color Atlas of Family Medicine. New York, NY: McGraw-Hill Medical; 2009:666-669.
Biologics may carry melanoma risk for patients with immune-mediated inflammatory diseases
The in a systematic review and meta-analysis published in JAMA Dermatology.
The studies included in the analysis, however, had limitations, including a lack of those comparing biologic and conventional systemic therapy in psoriasis and inflammatory bowel disease (IBD), according to Shamarke Esse, MRes, of the division of musculoskeletal and dermatological sciences at the University of Manchester (England) and colleagues. “We advocate for more large, well-designed studies of this issue to be performed to help improve certainty” regarding this association, they wrote.
Previous studies that have found an increased risk of melanoma in patients on biologics for psoriasis, rheumatoid arthritis, and IBD have “typically used the general population as the comparator,” they noted. There is a large amount of evidence that has established short-term efficacy and safety of biologics, compared with conventional systemic treatments, but concerns about longer-term cancer risk associated with biologics remains a concern. Moreover, they added, “melanoma is a highly immunogenic skin cancer and therefore of concern to patients treated with TNFIs [tumor necrosis factor inhibitors] because melanoma risk increases with suppression of the immune system and TNF-alpha plays an important role in the immune surveillance of tumors.12,13
In their review, the researchers identified seven cohort studies from MEDLINE, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) databases published between January 1995 and February 2019 that evaluated melanoma risk in about 34,000 patients receiving biologics and 135,370 patients who had never been treated with biologics, and were receiving conventional systemic therapy for psoriasis, RA, or IBD. Of these, four studies were in patients with RA, two studies were in patients with IBD, and a single study was in patients with psoriasis. Six studies examined patients taking TNF inhibitors, but only one of six studies had information on specific TNF inhibitors (adalimumab, etanercept, and infliximab) in patients with RA. One study evaluated abatacept and rituximab in RA patients.
The researchers analyzed the pooled relative risk across all studies. Compared with patients who received conventional systemic therapy, there was a nonsignificant association with risk of melanoma in patients with psoriasis (hazard ratio, 1.57; 95% confidence interval, 0.61-4.09), RA (pooled relative risk, 1.20; 95% CI, 0.83-1.74), and IBD (pRR, 1.20; 95% CI, 0.60-2.40).
Among RA patients who received TNF inhibitors only, there was a slightly elevated nonsignificant risk of melanoma (pRR, 1.08; 95% CI, 0.81-1.43). Patients receiving rituximab had a pRR of 0.73 (95% CI, 0.38-1.39), and patients taking abatacept had a pRR of 1.43 (95% CI, 0.66-3.09), compared with RA patients receiving conventional systemic therapy. When excluding two major studies in the RA subgroup of patients in a sensitivity analysis, pooled risk estimates varied from 0.91 (95% CI, 0.69-1.18) to 1.95 (95% CI, 1.16- 3.30). There were no significant between-study heterogeneity or publication bias among the IBD and RA studies.
Mr. Esse and colleagues acknowledged the small number of IBD and psoriasis studies in the meta-analysis, which could affect pooled risk estimates. “Any future update of our study through the inclusion of newly published studies may produce significantly different pooled risk estimates than those reported in our meta-analysis,” they said. In addition, the use of health insurance databases, lack of risk factors for melanoma, and inconsistent information about treatment duration for patients receiving conventional systemic therapy were also limitations.
“Prospective cohort studies using an active comparator, new-user study design providing detailed information on treatment history, concomitant treatments, biologic and conventional systemic treatment duration, recreational and treatment-related UV exposure, skin color, and date of melanoma diagnosis are required to help improve certainty. These studies would also need to account for key risk factors and the latency period of melanoma,” the researchers said.
Mr. Esse disclosed being funded by a PhD studentship from the Psoriasis Association. One author disclosed receiving personal fees from Janssen, LEO Pharma, Lilly, and Novartis outside the study; another disclosed receiving grants and personal fees from those and several other pharmaceutical companies during the study, and personal fees from several pharmaceutical companies outside of the submitted work; the fourth author had no disclosures.
SOURCE: Esse S et al. JAMA Dermatol. 2020 May 20;e201300.
The in a systematic review and meta-analysis published in JAMA Dermatology.
The studies included in the analysis, however, had limitations, including a lack of those comparing biologic and conventional systemic therapy in psoriasis and inflammatory bowel disease (IBD), according to Shamarke Esse, MRes, of the division of musculoskeletal and dermatological sciences at the University of Manchester (England) and colleagues. “We advocate for more large, well-designed studies of this issue to be performed to help improve certainty” regarding this association, they wrote.
Previous studies that have found an increased risk of melanoma in patients on biologics for psoriasis, rheumatoid arthritis, and IBD have “typically used the general population as the comparator,” they noted. There is a large amount of evidence that has established short-term efficacy and safety of biologics, compared with conventional systemic treatments, but concerns about longer-term cancer risk associated with biologics remains a concern. Moreover, they added, “melanoma is a highly immunogenic skin cancer and therefore of concern to patients treated with TNFIs [tumor necrosis factor inhibitors] because melanoma risk increases with suppression of the immune system and TNF-alpha plays an important role in the immune surveillance of tumors.12,13
In their review, the researchers identified seven cohort studies from MEDLINE, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) databases published between January 1995 and February 2019 that evaluated melanoma risk in about 34,000 patients receiving biologics and 135,370 patients who had never been treated with biologics, and were receiving conventional systemic therapy for psoriasis, RA, or IBD. Of these, four studies were in patients with RA, two studies were in patients with IBD, and a single study was in patients with psoriasis. Six studies examined patients taking TNF inhibitors, but only one of six studies had information on specific TNF inhibitors (adalimumab, etanercept, and infliximab) in patients with RA. One study evaluated abatacept and rituximab in RA patients.
The researchers analyzed the pooled relative risk across all studies. Compared with patients who received conventional systemic therapy, there was a nonsignificant association with risk of melanoma in patients with psoriasis (hazard ratio, 1.57; 95% confidence interval, 0.61-4.09), RA (pooled relative risk, 1.20; 95% CI, 0.83-1.74), and IBD (pRR, 1.20; 95% CI, 0.60-2.40).
Among RA patients who received TNF inhibitors only, there was a slightly elevated nonsignificant risk of melanoma (pRR, 1.08; 95% CI, 0.81-1.43). Patients receiving rituximab had a pRR of 0.73 (95% CI, 0.38-1.39), and patients taking abatacept had a pRR of 1.43 (95% CI, 0.66-3.09), compared with RA patients receiving conventional systemic therapy. When excluding two major studies in the RA subgroup of patients in a sensitivity analysis, pooled risk estimates varied from 0.91 (95% CI, 0.69-1.18) to 1.95 (95% CI, 1.16- 3.30). There were no significant between-study heterogeneity or publication bias among the IBD and RA studies.
Mr. Esse and colleagues acknowledged the small number of IBD and psoriasis studies in the meta-analysis, which could affect pooled risk estimates. “Any future update of our study through the inclusion of newly published studies may produce significantly different pooled risk estimates than those reported in our meta-analysis,” they said. In addition, the use of health insurance databases, lack of risk factors for melanoma, and inconsistent information about treatment duration for patients receiving conventional systemic therapy were also limitations.
“Prospective cohort studies using an active comparator, new-user study design providing detailed information on treatment history, concomitant treatments, biologic and conventional systemic treatment duration, recreational and treatment-related UV exposure, skin color, and date of melanoma diagnosis are required to help improve certainty. These studies would also need to account for key risk factors and the latency period of melanoma,” the researchers said.
Mr. Esse disclosed being funded by a PhD studentship from the Psoriasis Association. One author disclosed receiving personal fees from Janssen, LEO Pharma, Lilly, and Novartis outside the study; another disclosed receiving grants and personal fees from those and several other pharmaceutical companies during the study, and personal fees from several pharmaceutical companies outside of the submitted work; the fourth author had no disclosures.
SOURCE: Esse S et al. JAMA Dermatol. 2020 May 20;e201300.
The in a systematic review and meta-analysis published in JAMA Dermatology.
The studies included in the analysis, however, had limitations, including a lack of those comparing biologic and conventional systemic therapy in psoriasis and inflammatory bowel disease (IBD), according to Shamarke Esse, MRes, of the division of musculoskeletal and dermatological sciences at the University of Manchester (England) and colleagues. “We advocate for more large, well-designed studies of this issue to be performed to help improve certainty” regarding this association, they wrote.
Previous studies that have found an increased risk of melanoma in patients on biologics for psoriasis, rheumatoid arthritis, and IBD have “typically used the general population as the comparator,” they noted. There is a large amount of evidence that has established short-term efficacy and safety of biologics, compared with conventional systemic treatments, but concerns about longer-term cancer risk associated with biologics remains a concern. Moreover, they added, “melanoma is a highly immunogenic skin cancer and therefore of concern to patients treated with TNFIs [tumor necrosis factor inhibitors] because melanoma risk increases with suppression of the immune system and TNF-alpha plays an important role in the immune surveillance of tumors.12,13
In their review, the researchers identified seven cohort studies from MEDLINE, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) databases published between January 1995 and February 2019 that evaluated melanoma risk in about 34,000 patients receiving biologics and 135,370 patients who had never been treated with biologics, and were receiving conventional systemic therapy for psoriasis, RA, or IBD. Of these, four studies were in patients with RA, two studies were in patients with IBD, and a single study was in patients with psoriasis. Six studies examined patients taking TNF inhibitors, but only one of six studies had information on specific TNF inhibitors (adalimumab, etanercept, and infliximab) in patients with RA. One study evaluated abatacept and rituximab in RA patients.
The researchers analyzed the pooled relative risk across all studies. Compared with patients who received conventional systemic therapy, there was a nonsignificant association with risk of melanoma in patients with psoriasis (hazard ratio, 1.57; 95% confidence interval, 0.61-4.09), RA (pooled relative risk, 1.20; 95% CI, 0.83-1.74), and IBD (pRR, 1.20; 95% CI, 0.60-2.40).
Among RA patients who received TNF inhibitors only, there was a slightly elevated nonsignificant risk of melanoma (pRR, 1.08; 95% CI, 0.81-1.43). Patients receiving rituximab had a pRR of 0.73 (95% CI, 0.38-1.39), and patients taking abatacept had a pRR of 1.43 (95% CI, 0.66-3.09), compared with RA patients receiving conventional systemic therapy. When excluding two major studies in the RA subgroup of patients in a sensitivity analysis, pooled risk estimates varied from 0.91 (95% CI, 0.69-1.18) to 1.95 (95% CI, 1.16- 3.30). There were no significant between-study heterogeneity or publication bias among the IBD and RA studies.
Mr. Esse and colleagues acknowledged the small number of IBD and psoriasis studies in the meta-analysis, which could affect pooled risk estimates. “Any future update of our study through the inclusion of newly published studies may produce significantly different pooled risk estimates than those reported in our meta-analysis,” they said. In addition, the use of health insurance databases, lack of risk factors for melanoma, and inconsistent information about treatment duration for patients receiving conventional systemic therapy were also limitations.
“Prospective cohort studies using an active comparator, new-user study design providing detailed information on treatment history, concomitant treatments, biologic and conventional systemic treatment duration, recreational and treatment-related UV exposure, skin color, and date of melanoma diagnosis are required to help improve certainty. These studies would also need to account for key risk factors and the latency period of melanoma,” the researchers said.
Mr. Esse disclosed being funded by a PhD studentship from the Psoriasis Association. One author disclosed receiving personal fees from Janssen, LEO Pharma, Lilly, and Novartis outside the study; another disclosed receiving grants and personal fees from those and several other pharmaceutical companies during the study, and personal fees from several pharmaceutical companies outside of the submitted work; the fourth author had no disclosures.
SOURCE: Esse S et al. JAMA Dermatol. 2020 May 20;e201300.
FROM JAMA DERMATOLOGY
Painful ear lesion
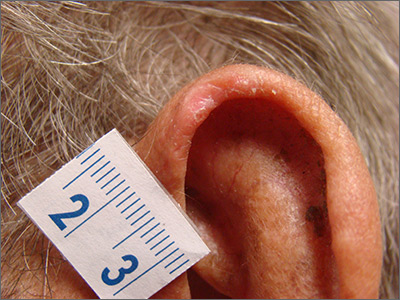
The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.

The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.

The patient was given a diagnosis of chondrodermatitis nodularis helicis (CNH), an inflammation of the cartilage and overlying skin causing a painful nodule of the helix. These lesions typically have a more prominent nodular component and a central ulceration or firm scale. They are thought to be due to chronic pressure on the ear.
There is a slight male predominance of CNH and onset is usually gradual. Patients often experience pain when sleeping on the affected side. The tenderness usually can be reproduced clinically by pressing on the lesion. The ears also are a high-risk area for actinic keratoses (AK) and nonmelanoma skin cancer (NMSC); if there is doubt about the diagnosis, a biopsy may be warranted to rule out AK or NMSC. In this patient, a shave biopsy was performed.
Various treatment regimens are available for CNH. The least invasive treatment approach is to use a “cut out” foam or a special donut-shaped pillow to protect the area from further pressure. By protecting from pressure and irritation, the lesion resolves in 57% to 92% of cases in clinical studies. Intralesional injection with 0.2 mL of 10 mg/mL triamcinolone acetonide is a simple in-office procedure that frequently helps the pain and may be curative; although, repeat injections may be necessary. Excision of the overlying skin and the affected cartilage is a more aggressive treatment with high success rates. More recently, treatment with topical nitroglycerin patches or photodynamic therapy have been described in small trials.
After confirming by a shave biopsy that the lesion was not cancerous, the patient returned for an elliptical excision of the lesion. A dermal curette was used to remove the rough abnormal inflammation of the underlying cartilage, and the elliptical wound was sutured with a linear closure.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, University of New Mexico School of Medicine, Albuquerque.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.
Juul Nielsen L, Holkmann Olsen C, Lock-Andersen J. Therapeutic options of chondrodermatitis nodularis helicis [published online January 27, 2016]. Plast Surg Int. 2016;2016:4340168.
Today’s top news highlights: COVID-19 could worsen gambling problems, food allergies less common than thought
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Could COVID-19 worsen gambling problems?
Take isolation, add excess available time and anxiety about illness or finances and you get the potential to increase problem gambling behaviors during the COVID-19 pandemic. A call to action, recently published in the Journal of Addiction Medicine, says it’s essential to gather data and supply guidance on this issue. “People are likely to be experiencing stress at levels they haven’t experienced previously,” said coauthor Marc N. Potenza, MD, PhD, of Yale University, New Haven, Conn. While multiple factors can contribute to addictive behaviors, “with respect to the pandemic, one concern is that so-called negative reinforcement motivations – engaging in an addictive behavior to escape from depressed or negative mood states – may be a driving motivation for a significant number of people during this time,” he said. Read more.
Food allergies in children are less frequent than expected
Food allergies appear to be less common than previously reported among 6- to 10-year-olds in Europe, according to a recent study. Prevalance ranged from a low of 1.4% to a high of 3.8%, both of which are “considerably lower” than the 16% rate based on parental reports of symptoms such as rash, itching, or diarrhea, Linus Grabenhenrich, MD, MPH, and colleagues reported in Allergy. The most commonly reported allergies were to peanuts and hazelnuts, with a prevalence of just over 5% for both. Previous research on pediatric food allergy prevalence has largely consisted of single-center studies with heterogeneous designs, the researchers noted. Read more.
The grocery store hug
William G. Wilkoff, MD, grew up in a family that didn’t embrace hugging, but as a small-town pediatrician he warmed up to the concept so much that he would frequently hug a passing acquaintance at the grocery store. That’s something he misses in the current environment and that he doesn’t expect will return. “[N]early every week I encounter one or two people with whom I have a long and sometimes emotionally charged relationship,” Dr. Wilkoff wrote in a column on MDedge. “Nurses with whom I sweated over difficult delivery room resuscitations. Parents for whom their anxiety was getting in the way of their ability to parent. Parents and caregivers of complex multiply disabled children who are now adults. Peers who have lost a spouse or a child. I’m sure you have your own list of people who send off that we-need-to-hug spark.” Read more.
Identifying structural lesions of axial spondyloarthritis
What constitutes a structural lesion of the sacroiliac joints on MRI that’s indicative of axial spondyloarthritis (axSpA) has long been a matter of conjecture, but the Assessment of SpondyloArthritis International Society (ASAS) MRI Working Group has developed new definitions that show a high degree of specificity in identifying such lesions in the disease. “Previous studies have described structural lesions in different ways, precluding meaningful comparisons between studies,” Walter P. Maksymowych, MD, said at the annual European Congress of Rheumatology, held online this year due to COVID-19. “The ASAS MRI group has generated updated consensus lesion definitions that describe each of the MRI lesions in the sacroiliac joint. These definitions have been validated by seven expert readers from the ASAS MRI group on MRI images from the ASAS classification cohort.” Read more.
Making the world’s skin crawl
Clinicians should be aware of the skin manifestations of COVID-19, especially when triaging patients. In a commentary published on MDedge, Kathleen M. Coerdt and Amor Khachemoune, MD, describe the dermatologic implications of COVID-19, including the clinical manifestations of the disease, risk reduction techniques for patients and providers, personal protective equipment-associated adverse reactions, and the financial impact on dermatologists. Read more.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Atopic dermatitis in adults, children linked to neuropsychiatric disorders
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
according to a study presented at the annual meeting of the Society for Investigative Dermatology, held virtually.
“The risk increase ranges from as low as 5% up to 59%, depending on the outcome, with generally greater effects observed among the adults,” Joy Wan, MD, a postdoctoral dermatology fellow at the University of Pennsylvania, Philadelphia, said in her presentation. The risk was independent of other atopic disease, gender, age, and socioeconomic status.
Dr. Wan and colleagues conducted a cohort study of patients with AD in the United Kingdom using data from the Health Improvement Network (THIN) electronic records database, matching AD patients in THIN with up to five patients without AD, similar in age and also registered to general practices. The researchers validated AD disease status using an algorithm that identified patients with a diagnostic code and two therapy codes related to AD. Outcomes of interest included anxiety, depression, bipolar disorder, obsessive-compulsive disorder, ADHD, schizophrenia, and autism. Patients entered into the cohort when they were diagnosed with AD, registered by a practice, or when data from a practice was reported to THIN. The researchers stopped following patients when they developed a neuropsychiatric outcome of interest, left a practice, died, or when the study ended.
“Previous studies have found associations between atopic dermatitis and anxiety, depression, and attention-deficit/hyperactivity disorder. However, many previous studies had been cross-sectional and they were unable to evaluate the directionality of association between atopic dermatitis and neuropsychiatric outcomes, while other previous studies have relied on the self-report of atopic dermatitis and outcomes as well,” Dr. Wan said. “Thus, longitudinal studies, using validated measures of atopic dermatitis, and those that include the entire age span, are really needed.”
Overall, 434,859 children and adolescents under aged 18 with AD in the THIN database were matched to 1,983,589 controls, and 644,802 adults with AD were matched to almost 2,900,000 adults without AD. In the pediatric group, demographics were mostly balanced between children with and without AD: the average age ranged between about 5 and almost 6 years. In pediatric patients with AD, there was a higher rate of allergic rhinitis (6.2% vs. 4%) and asthma (13.5% vs. 9.3%) than in the control group.
For adults, the average age was about 48 years in both groups. Compared with patients who did not have AD, adults with AD also had higher rates of allergic rhinitis (15.2% vs. 9.6%) and asthma (19.9% vs. 12.6%).
After adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis, Dr. Wan and colleagues found greater rates of bipolar disorder (hazard ratio, 1.34; 95% confidence interval, 1.09-1.65), obsessive-compulsive disorder (HR, 1.30; 95% CI, 1.21-1.41), anxiety (HR, 1.09; 95% CI, 1.07-1.11), and depression (HR, 1.06; 95% CI, 1.04-1.08) among children and adolescents with AD, compared with controls.
In the adult cohort, a diagnosis of AD was associated with an increased risk of autism (HR, 1.53; 95% CI, 1.30-1.80), obsessive-compulsive disorder (HR, 1.49; 95% CI, 1.40-1.59), ADHD (HR, 1.31; 95% CI, 1.13-1.53), anxiety (HR, 1.17; 95% CI, 1.15-1.18), depression (HR, 1.15; 95% CI, 1.14-1.16), and bipolar disorder (HR, 1.12; 95% CI, 1.04-1.21), after adjusting for age, gender, socioeconomic status, asthma, and allergic rhinitis.
One reason for the increased associations among the adults, even for ADHD and autism, which are more characteristically diagnosed in childhood, Dr. Wan said, is that, since they looked at incident outcomes, “many children may already have had these prevalent comorbidities at the time of the entry in the cohort.”
She noted that the study may have observation bias or unknown confounders, but she hopes these results raise awareness of the association between AD and neuropsychiatric disorders, although more research is needed to determine how AD severity affects neuropsychiatric outcomes. “Additional work is needed to really understand the mechanisms that drive these associations, whether it’s mediated through symptoms of atopic dermatitis such as itch and poor sleep, or potentially the stigma of having a chronic skin disease, or perhaps shared pathophysiology between atopic dermatitis and these neuropsychiatric diseases,” she said.
The study was funded by a grant from Pfizer. Dr. Wan reports receiving research funding from Pfizer paid to the University of Pennsylvania.
FROM SID 2020
Cancer risk elevated in hidradenitis suppurativa patients
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
.
HS is associated with severe comorbidities, and previous studies have suggested a link between HS and cancer development, wrote Joon Min Jung, MD, of the University of Ulsan College of Medicine, Seoul, Korea, and colleagues.
“The aberrant immune response and chronic inflammation in HS and genetic and environmental factors associated with the disease may all be factors in the development of cancer,” but large, population-based studies of cancer in HS patients are limited, they noted.
In a study published in JAMA Dermatology, the researchers reviewed data from 22,468 adults with HS and 179,734 matched controls, in the Korean National Health Insurance System, seen by physicians between January 2009 and December 2017. The average age of the participants was 34 years, and 64% were male.
Overall, HS patients had a significantly higher risk of cancer compared with controls, with an adjusted hazard ratio (aHR) of 1.28.
As for specific cancers, HS patients had a significantly higher risk for Hodgkin lymphoma (aHR 5.08), oral cavity and pharyngeal cancer (aHR 3.10), central nervous system cancer (aHR 2.40), nonmelanoma skin cancer (aHR 2.06), prostate cancer (aHR 2.05), and colorectal cancer (aHR 1.45).
The risk of any cancer was not significantly different between women with HS and female controls (after adjustment for comorbidities), but was significantly higher among men with HS compared with male controls, also after adjustment for comorbidities (aHR, 1.37). In addition, HS patients in both younger (less than 40 years) and older (aged 40 years and older) age groups had increased cancer risk compared with age-matched controls. Overall cancer risk and the risk of most cancer types were higher among HS patients with moderate to severe disease than in those with mild disease, with the exception of nonmelanoma skin cancer, prostate cancer, lymphoma, and leukemia.
“Overall cancer risk showed a tendency to increase with worsening HS severity, reinforcing the possibility of an association between HS and cancer development,” the researchers noted. “However, we could not identify tendencies in some specific cancers, such as nonmelanoma skin cancer, CNS cancer, and prostate cancer, because the number of occurrences of those cancers was too small in the group with moderate to severe HS.”
The study findings were limited by several factors including the potential underestimate of HS cases in the population and the inability of the study design to adjust for factors including smoking status, alcohol use, and obesity, the researchers noted. However, the results support an increased cancer risk in HS patients and suggest the need to promote lifestyle modifications to reduce risk, and to increase cancer surveillance in these patients, they said. “For early detection of skin cancer, more aggressive histologic examination and a high level of suspicion are required,” they added.
The study was supported by the National Research Foundation of Korea and the Korea Health Technology R&D Project. The researchers had no financial conflicts to disclose.
SOURCE: Jung JM et al. JAMA Dermatol. 2020 May 27. doi: 10.1001/jamadermatol.2020.1422.
FROM JAMA DERMATOLOGY