User login
Seminal, highly anticipated Alzheimer’s trial falters
DIAN-TU top-line results negative
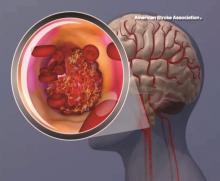
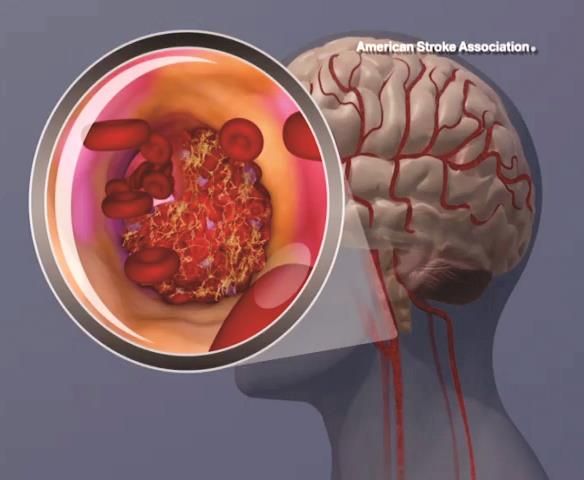
Top-line results from the seminal phase 2/3 Dominantly Inherited Alzheimer’s Network–Trials Unit (DIAN-TU) study show that the novel drugs gantenerumab (Roche) and solanezumab (Lilly) did not meet the primary endpoint in patients with early-stage, dominantly inherited Alzheimer’s disease (AD), investigators have announced.
In the international trial, which included almost 200 participants, the two experimental agents were evaluated separately. However, initial analyses showed that neither significantly slowed cognitive decline, the primary outcome measure, nor memory loss.
Still, the researchers noted that they will continue exploring data from DIAN-TU’s cognitive and clinical outcomes and are awaiting analyses of various biomarkers.
“Although the drugs we evaluated were not successful, the trial will move us forward in understanding Alzheimer’s,” principal investigator Randall J. Bateman, MD, of Washington University, St. Louis, said in a news release.
Funders for the trial included the National Institute on Aging and the Alzheimer’s Association.
“While the top-line data fell short, the Alzheimer’s Association looks forward to a more complete report at upcoming scientific conferences. We learn from every trial,” Maria Carrillo, PhD, chief scientific officer at the Alzheimer’s Association, noted in the organization’s own release.
Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, agreed with Dr. Carrillo that, although the results were disappointing, the data will be beneficial for the field.
“It’s always a difficult day when we get news like this,” Dr. Edelmayer said in an interview. However, “this research is going to absolutely provide valuable information once we can really pick through all of the data.”
Rare condition
Dominantly inherited AD, also known as familial AD or autosomal dominant AD, is rare but can affect memory and cognitive skills in individuals as young as age 30. It is caused by mutations on chromosomes 21, 14, and/or 1 that play a part in the breakdown of amyloid proteins and formation of amyloid plaques.
Both gantenerumab and solanezumab were created to target and neutralize amyloid-beta, albeit through different mechanisms. Both are also being assessed in other trials as treatment for more common forms of AD.
As reported by Medscape Medical News, results of the phase 3 EXPEDITION3 trial of solanezumab in patients with mild AD were negative, as were two other phase 3 trials. The drug is now being evaluated in the ongoing solanezumab Anti-Amyloid Treatment in Asymptomatic Alzheimer’s (A4) study.
Although the phase 3 SCARLET ROAD trial of gantenerumab for mild AD was stopped early for futility in 2014, it was continued as an open-label extension at the high dose for 2 years. During that period, follow-up analyses showed a dramatic decline in amyloid-beta deposition in the participants – leading to the launch of the phase 3 GRADUATE 1 and GRADUATE 2 trials.
Starting in 2012, DIAN-TU was conducted at 24 sites in the United States, the United Kingdom, Canada, Europe, and Australia. It followed 194 adult patients for up to 7 years (average duration, 5 years). Its original estimated completion date was December 2020, as stated on clinicaltrials.gov.
All participants had family members with a genetic mutation that causes early-onset Alzheimer’s disease. They already had very mild symptoms of cognitive decline and memory loss at the start of the trial or were expected to develop symptoms within 15 years of enrollment.
“People who inherit the mutation are all but guaranteed to develop symptoms at about the same age their parents did,” the release noted.
“While devastating for families, such mutations allow researchers to identify people in the early stages of the disease before their behavior and memory begin to change,” it added.
The Alzheimer’s Association noted in its release that a child of a parent with the mutation has a 50/50 chance of inheriting the disease. “This form affects less than 1% of the individuals living with Alzheimer’s disease today,” Dr. Edelmayer noted.
Detailed data coming soon
Trial participants were randomly assigned to receive either solanezumab, gantenerumab, or matching placebo. To act as a comparator group, family members without the AD mutation were also included.
The primary measure was change from baseline in the DIAN-TU cognitive composite score. Secondary measures included changes on the Mini-Mental State Examination, the Functional Assessment Scale, the Neuropsychiatric Inventory Questionnaire, the 12-item International Shopping List Test, the Memory Complaint Questionnaire, and the Wechsler Memory Scale Logical Memory/Paragraph Memory test.
The researchers also conducted imaging scans and collected samples of blood and cerebrospinal fluid.
Along with announcing the negative top-line results for the trial, the investigators noted that “a more detailed analysis of the trial’s data” will be presented at the Advances in Alzheimer’s and Parkinson’s Therapies in Vienna on April 2, 2020, and at the Alzheimer’s Association International Conference in Amsterdam in July.
The researchers will continue to explore all data gathered – but already new insights have been discovered into the development and progression of AD, Dr. Bateman noted.
Included among these discoveries is that brain changes that occur as the disease progresses are similar among those with the inherited, early-onset form of AD and the late-onset form.
“The trial’s innovative design ... will make advances for future Alzheimer’s trials. Ongoing and continued research and trials will bring us closer to our goal to stop Alzheimer’s,” Dr. Bateman said. “We will continue until we are successful.”
“These results reflect the difficult nature of treating [AD] and the great need for continued research,” said Daniel Skovronsky, MD, PhD, chief scientific officer and president of Lilly Research Labs.
“If we have learned one thing after more than 30 years of Alzheimer’s research, it is that even negative results propel the science forward,” he added.
Lilly noted in a statement that the DIAN-TU top-line results will not affect its ongoing A4 study of solanezumab. Roche noted in its own statement that the findings also will not affect the company’s ongoing GRADUATE studies of gantenerumab.
“The work doesn’t stop here”
Richard J. Hodes, MD, director of the National Institute on Aging, said that DIAN-TU will advance the field’s knowledge about a complex disease.
“We look forward to learning more through the published, peer-reviewed data, which will provide a broad range of scientists with crucial information and guidance for future research,” he said.
Howard Fillit, MD, founding executive director and chief science officer at the Alzheimer’s Drug Discovery Foundation (ADDF), agreed.
“While we are disappointed that patients in this study did not see a benefit, we need to keep in mind that Alzheimer’s is a complicated disease due to complex, multifactorial causes,” he said in a statement.
“ADDF has long supported a broader approach that moves past targeting beta-amyloid and advances a diverse pipeline of drugs addressing multiple targets” in AD, Dr. Fillit added. “We need multiple ‘shots on goals’ to discover effective drugs.”
Dr. Edelmayer said the results emphasize that “this story isn’t yet completely told” and that there is still a lot to learn from the data, especially regarding the biomarkers that were tested.
“With that information, we will gain valuable insight into the outcomes that have been released but will also probably better understand where we should be putting our energies and focus moving forward,” she said.
Going forward, “we will continue this fight until we have an effective treatment for all individuals living with Alzheimer’s, whether it’s dominantly inherited [AD] or the more common version, which is the late-onset or sporadic form of the disease,” said Dr. Edelmayer.
“We have to stay optimistic. The work doesn’t stop here.”
The trial was funded by Eli Lilly, Roche, the Alzheimer’s Association, the National Institute on Aging, the GHR Foundation, and FBRI.
This article first appeared on Medscape.com.
DIAN-TU top-line results negative
DIAN-TU top-line results negative
Top-line results from the seminal phase 2/3 Dominantly Inherited Alzheimer’s Network–Trials Unit (DIAN-TU) study show that the novel drugs gantenerumab (Roche) and solanezumab (Lilly) did not meet the primary endpoint in patients with early-stage, dominantly inherited Alzheimer’s disease (AD), investigators have announced.
In the international trial, which included almost 200 participants, the two experimental agents were evaluated separately. However, initial analyses showed that neither significantly slowed cognitive decline, the primary outcome measure, nor memory loss.
Still, the researchers noted that they will continue exploring data from DIAN-TU’s cognitive and clinical outcomes and are awaiting analyses of various biomarkers.
“Although the drugs we evaluated were not successful, the trial will move us forward in understanding Alzheimer’s,” principal investigator Randall J. Bateman, MD, of Washington University, St. Louis, said in a news release.
Funders for the trial included the National Institute on Aging and the Alzheimer’s Association.
“While the top-line data fell short, the Alzheimer’s Association looks forward to a more complete report at upcoming scientific conferences. We learn from every trial,” Maria Carrillo, PhD, chief scientific officer at the Alzheimer’s Association, noted in the organization’s own release.
Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, agreed with Dr. Carrillo that, although the results were disappointing, the data will be beneficial for the field.
“It’s always a difficult day when we get news like this,” Dr. Edelmayer said in an interview. However, “this research is going to absolutely provide valuable information once we can really pick through all of the data.”
Rare condition
Dominantly inherited AD, also known as familial AD or autosomal dominant AD, is rare but can affect memory and cognitive skills in individuals as young as age 30. It is caused by mutations on chromosomes 21, 14, and/or 1 that play a part in the breakdown of amyloid proteins and formation of amyloid plaques.
Both gantenerumab and solanezumab were created to target and neutralize amyloid-beta, albeit through different mechanisms. Both are also being assessed in other trials as treatment for more common forms of AD.
As reported by Medscape Medical News, results of the phase 3 EXPEDITION3 trial of solanezumab in patients with mild AD were negative, as were two other phase 3 trials. The drug is now being evaluated in the ongoing solanezumab Anti-Amyloid Treatment in Asymptomatic Alzheimer’s (A4) study.
Although the phase 3 SCARLET ROAD trial of gantenerumab for mild AD was stopped early for futility in 2014, it was continued as an open-label extension at the high dose for 2 years. During that period, follow-up analyses showed a dramatic decline in amyloid-beta deposition in the participants – leading to the launch of the phase 3 GRADUATE 1 and GRADUATE 2 trials.
Starting in 2012, DIAN-TU was conducted at 24 sites in the United States, the United Kingdom, Canada, Europe, and Australia. It followed 194 adult patients for up to 7 years (average duration, 5 years). Its original estimated completion date was December 2020, as stated on clinicaltrials.gov.
All participants had family members with a genetic mutation that causes early-onset Alzheimer’s disease. They already had very mild symptoms of cognitive decline and memory loss at the start of the trial or were expected to develop symptoms within 15 years of enrollment.
“People who inherit the mutation are all but guaranteed to develop symptoms at about the same age their parents did,” the release noted.
“While devastating for families, such mutations allow researchers to identify people in the early stages of the disease before their behavior and memory begin to change,” it added.
The Alzheimer’s Association noted in its release that a child of a parent with the mutation has a 50/50 chance of inheriting the disease. “This form affects less than 1% of the individuals living with Alzheimer’s disease today,” Dr. Edelmayer noted.
Detailed data coming soon
Trial participants were randomly assigned to receive either solanezumab, gantenerumab, or matching placebo. To act as a comparator group, family members without the AD mutation were also included.
The primary measure was change from baseline in the DIAN-TU cognitive composite score. Secondary measures included changes on the Mini-Mental State Examination, the Functional Assessment Scale, the Neuropsychiatric Inventory Questionnaire, the 12-item International Shopping List Test, the Memory Complaint Questionnaire, and the Wechsler Memory Scale Logical Memory/Paragraph Memory test.
The researchers also conducted imaging scans and collected samples of blood and cerebrospinal fluid.
Along with announcing the negative top-line results for the trial, the investigators noted that “a more detailed analysis of the trial’s data” will be presented at the Advances in Alzheimer’s and Parkinson’s Therapies in Vienna on April 2, 2020, and at the Alzheimer’s Association International Conference in Amsterdam in July.
The researchers will continue to explore all data gathered – but already new insights have been discovered into the development and progression of AD, Dr. Bateman noted.
Included among these discoveries is that brain changes that occur as the disease progresses are similar among those with the inherited, early-onset form of AD and the late-onset form.
“The trial’s innovative design ... will make advances for future Alzheimer’s trials. Ongoing and continued research and trials will bring us closer to our goal to stop Alzheimer’s,” Dr. Bateman said. “We will continue until we are successful.”
“These results reflect the difficult nature of treating [AD] and the great need for continued research,” said Daniel Skovronsky, MD, PhD, chief scientific officer and president of Lilly Research Labs.
“If we have learned one thing after more than 30 years of Alzheimer’s research, it is that even negative results propel the science forward,” he added.
Lilly noted in a statement that the DIAN-TU top-line results will not affect its ongoing A4 study of solanezumab. Roche noted in its own statement that the findings also will not affect the company’s ongoing GRADUATE studies of gantenerumab.
“The work doesn’t stop here”
Richard J. Hodes, MD, director of the National Institute on Aging, said that DIAN-TU will advance the field’s knowledge about a complex disease.
“We look forward to learning more through the published, peer-reviewed data, which will provide a broad range of scientists with crucial information and guidance for future research,” he said.
Howard Fillit, MD, founding executive director and chief science officer at the Alzheimer’s Drug Discovery Foundation (ADDF), agreed.
“While we are disappointed that patients in this study did not see a benefit, we need to keep in mind that Alzheimer’s is a complicated disease due to complex, multifactorial causes,” he said in a statement.
“ADDF has long supported a broader approach that moves past targeting beta-amyloid and advances a diverse pipeline of drugs addressing multiple targets” in AD, Dr. Fillit added. “We need multiple ‘shots on goals’ to discover effective drugs.”
Dr. Edelmayer said the results emphasize that “this story isn’t yet completely told” and that there is still a lot to learn from the data, especially regarding the biomarkers that were tested.
“With that information, we will gain valuable insight into the outcomes that have been released but will also probably better understand where we should be putting our energies and focus moving forward,” she said.
Going forward, “we will continue this fight until we have an effective treatment for all individuals living with Alzheimer’s, whether it’s dominantly inherited [AD] or the more common version, which is the late-onset or sporadic form of the disease,” said Dr. Edelmayer.
“We have to stay optimistic. The work doesn’t stop here.”
The trial was funded by Eli Lilly, Roche, the Alzheimer’s Association, the National Institute on Aging, the GHR Foundation, and FBRI.
This article first appeared on Medscape.com.
Top-line results from the seminal phase 2/3 Dominantly Inherited Alzheimer’s Network–Trials Unit (DIAN-TU) study show that the novel drugs gantenerumab (Roche) and solanezumab (Lilly) did not meet the primary endpoint in patients with early-stage, dominantly inherited Alzheimer’s disease (AD), investigators have announced.
In the international trial, which included almost 200 participants, the two experimental agents were evaluated separately. However, initial analyses showed that neither significantly slowed cognitive decline, the primary outcome measure, nor memory loss.
Still, the researchers noted that they will continue exploring data from DIAN-TU’s cognitive and clinical outcomes and are awaiting analyses of various biomarkers.
“Although the drugs we evaluated were not successful, the trial will move us forward in understanding Alzheimer’s,” principal investigator Randall J. Bateman, MD, of Washington University, St. Louis, said in a news release.
Funders for the trial included the National Institute on Aging and the Alzheimer’s Association.
“While the top-line data fell short, the Alzheimer’s Association looks forward to a more complete report at upcoming scientific conferences. We learn from every trial,” Maria Carrillo, PhD, chief scientific officer at the Alzheimer’s Association, noted in the organization’s own release.
Rebecca M. Edelmayer, PhD, director of scientific engagement for the Alzheimer’s Association, agreed with Dr. Carrillo that, although the results were disappointing, the data will be beneficial for the field.
“It’s always a difficult day when we get news like this,” Dr. Edelmayer said in an interview. However, “this research is going to absolutely provide valuable information once we can really pick through all of the data.”
Rare condition
Dominantly inherited AD, also known as familial AD or autosomal dominant AD, is rare but can affect memory and cognitive skills in individuals as young as age 30. It is caused by mutations on chromosomes 21, 14, and/or 1 that play a part in the breakdown of amyloid proteins and formation of amyloid plaques.
Both gantenerumab and solanezumab were created to target and neutralize amyloid-beta, albeit through different mechanisms. Both are also being assessed in other trials as treatment for more common forms of AD.
As reported by Medscape Medical News, results of the phase 3 EXPEDITION3 trial of solanezumab in patients with mild AD were negative, as were two other phase 3 trials. The drug is now being evaluated in the ongoing solanezumab Anti-Amyloid Treatment in Asymptomatic Alzheimer’s (A4) study.
Although the phase 3 SCARLET ROAD trial of gantenerumab for mild AD was stopped early for futility in 2014, it was continued as an open-label extension at the high dose for 2 years. During that period, follow-up analyses showed a dramatic decline in amyloid-beta deposition in the participants – leading to the launch of the phase 3 GRADUATE 1 and GRADUATE 2 trials.
Starting in 2012, DIAN-TU was conducted at 24 sites in the United States, the United Kingdom, Canada, Europe, and Australia. It followed 194 adult patients for up to 7 years (average duration, 5 years). Its original estimated completion date was December 2020, as stated on clinicaltrials.gov.
All participants had family members with a genetic mutation that causes early-onset Alzheimer’s disease. They already had very mild symptoms of cognitive decline and memory loss at the start of the trial or were expected to develop symptoms within 15 years of enrollment.
“People who inherit the mutation are all but guaranteed to develop symptoms at about the same age their parents did,” the release noted.
“While devastating for families, such mutations allow researchers to identify people in the early stages of the disease before their behavior and memory begin to change,” it added.
The Alzheimer’s Association noted in its release that a child of a parent with the mutation has a 50/50 chance of inheriting the disease. “This form affects less than 1% of the individuals living with Alzheimer’s disease today,” Dr. Edelmayer noted.
Detailed data coming soon
Trial participants were randomly assigned to receive either solanezumab, gantenerumab, or matching placebo. To act as a comparator group, family members without the AD mutation were also included.
The primary measure was change from baseline in the DIAN-TU cognitive composite score. Secondary measures included changes on the Mini-Mental State Examination, the Functional Assessment Scale, the Neuropsychiatric Inventory Questionnaire, the 12-item International Shopping List Test, the Memory Complaint Questionnaire, and the Wechsler Memory Scale Logical Memory/Paragraph Memory test.
The researchers also conducted imaging scans and collected samples of blood and cerebrospinal fluid.
Along with announcing the negative top-line results for the trial, the investigators noted that “a more detailed analysis of the trial’s data” will be presented at the Advances in Alzheimer’s and Parkinson’s Therapies in Vienna on April 2, 2020, and at the Alzheimer’s Association International Conference in Amsterdam in July.
The researchers will continue to explore all data gathered – but already new insights have been discovered into the development and progression of AD, Dr. Bateman noted.
Included among these discoveries is that brain changes that occur as the disease progresses are similar among those with the inherited, early-onset form of AD and the late-onset form.
“The trial’s innovative design ... will make advances for future Alzheimer’s trials. Ongoing and continued research and trials will bring us closer to our goal to stop Alzheimer’s,” Dr. Bateman said. “We will continue until we are successful.”
“These results reflect the difficult nature of treating [AD] and the great need for continued research,” said Daniel Skovronsky, MD, PhD, chief scientific officer and president of Lilly Research Labs.
“If we have learned one thing after more than 30 years of Alzheimer’s research, it is that even negative results propel the science forward,” he added.
Lilly noted in a statement that the DIAN-TU top-line results will not affect its ongoing A4 study of solanezumab. Roche noted in its own statement that the findings also will not affect the company’s ongoing GRADUATE studies of gantenerumab.
“The work doesn’t stop here”
Richard J. Hodes, MD, director of the National Institute on Aging, said that DIAN-TU will advance the field’s knowledge about a complex disease.
“We look forward to learning more through the published, peer-reviewed data, which will provide a broad range of scientists with crucial information and guidance for future research,” he said.
Howard Fillit, MD, founding executive director and chief science officer at the Alzheimer’s Drug Discovery Foundation (ADDF), agreed.
“While we are disappointed that patients in this study did not see a benefit, we need to keep in mind that Alzheimer’s is a complicated disease due to complex, multifactorial causes,” he said in a statement.
“ADDF has long supported a broader approach that moves past targeting beta-amyloid and advances a diverse pipeline of drugs addressing multiple targets” in AD, Dr. Fillit added. “We need multiple ‘shots on goals’ to discover effective drugs.”
Dr. Edelmayer said the results emphasize that “this story isn’t yet completely told” and that there is still a lot to learn from the data, especially regarding the biomarkers that were tested.
“With that information, we will gain valuable insight into the outcomes that have been released but will also probably better understand where we should be putting our energies and focus moving forward,” she said.
Going forward, “we will continue this fight until we have an effective treatment for all individuals living with Alzheimer’s, whether it’s dominantly inherited [AD] or the more common version, which is the late-onset or sporadic form of the disease,” said Dr. Edelmayer.
“We have to stay optimistic. The work doesn’t stop here.”
The trial was funded by Eli Lilly, Roche, the Alzheimer’s Association, the National Institute on Aging, the GHR Foundation, and FBRI.
This article first appeared on Medscape.com.
Older NHL survivors show worse cognitive decline
Older long-term survivors of non-Hodgkin lymphoma (NHL) may have worse cognitive outcomes compared with the noncancer aging population, according to a cross-sectional study.
The findings suggest additional research is needed to better understand cognitive decline in older survivors of NHL.
“The aim of the present study was to examine the difference in cognitive status between a group of long-term older survivors of NHL compared with a group of noncancer controls of the same age,” wrote Domenico La Carpia, MD, of Fondazione ANT Italia Onlus, Florence, Italy, and colleagues.
The researchers conducted a multicenter cross-sectional cohort study involving 63 long-term survivors of NHL and 61 age-matched controls. Their report was published in the Journal of Geriatric Oncology.
Eligible survivors and controls were aged 65 years and older. Among both groups, the mean age of study participants was 74 years, and most survivors were women (58.7%).
While cognitive decline was assessed via standardized neuropsychological testing, the team also evaluated polypharmacy, functional status, and level of multimorbidity in the cohort.
Other clinical data, including the time from complete remission, type of treatment received, and histopathological type of tumor, were collected from patient charts and included in the analysis.
After analysis, the researchers found that NHL survivors had a higher mean number of chronic conditions (3.4 vs. 2.3; P = .003), were receiving more medications (3.4 vs. 2.3; P = .03), and had worse functional status compared with controls.
In addition, survivors had impaired executive functioning compared with control subjects (Trail Making Test B-A, 47.9 vs. 32.1; P = .04), but scores on the Mini Mental State Examination (MMSE) did not differ between the groups.
“A small, statistically significant difference was also observed in verbal memory scores between the two groups,” they reported.
The researchers acknowledged that a key limitation was the cross-sectional nature of the study; hence, causality cannot be inferred from the data.
“Comprehensive geriatric assessment for older cancer survivors is advisable to identify those individuals who are at highest risk of developing disability and to implement tailored early interventions,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: La Carpia D et al. J Geriatr Oncol. 2020 Jan 31. doi: 10.1016/j.jgo.2020.01.007.
Older long-term survivors of non-Hodgkin lymphoma (NHL) may have worse cognitive outcomes compared with the noncancer aging population, according to a cross-sectional study.
The findings suggest additional research is needed to better understand cognitive decline in older survivors of NHL.
“The aim of the present study was to examine the difference in cognitive status between a group of long-term older survivors of NHL compared with a group of noncancer controls of the same age,” wrote Domenico La Carpia, MD, of Fondazione ANT Italia Onlus, Florence, Italy, and colleagues.
The researchers conducted a multicenter cross-sectional cohort study involving 63 long-term survivors of NHL and 61 age-matched controls. Their report was published in the Journal of Geriatric Oncology.
Eligible survivors and controls were aged 65 years and older. Among both groups, the mean age of study participants was 74 years, and most survivors were women (58.7%).
While cognitive decline was assessed via standardized neuropsychological testing, the team also evaluated polypharmacy, functional status, and level of multimorbidity in the cohort.
Other clinical data, including the time from complete remission, type of treatment received, and histopathological type of tumor, were collected from patient charts and included in the analysis.
After analysis, the researchers found that NHL survivors had a higher mean number of chronic conditions (3.4 vs. 2.3; P = .003), were receiving more medications (3.4 vs. 2.3; P = .03), and had worse functional status compared with controls.
In addition, survivors had impaired executive functioning compared with control subjects (Trail Making Test B-A, 47.9 vs. 32.1; P = .04), but scores on the Mini Mental State Examination (MMSE) did not differ between the groups.
“A small, statistically significant difference was also observed in verbal memory scores between the two groups,” they reported.
The researchers acknowledged that a key limitation was the cross-sectional nature of the study; hence, causality cannot be inferred from the data.
“Comprehensive geriatric assessment for older cancer survivors is advisable to identify those individuals who are at highest risk of developing disability and to implement tailored early interventions,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: La Carpia D et al. J Geriatr Oncol. 2020 Jan 31. doi: 10.1016/j.jgo.2020.01.007.
Older long-term survivors of non-Hodgkin lymphoma (NHL) may have worse cognitive outcomes compared with the noncancer aging population, according to a cross-sectional study.
The findings suggest additional research is needed to better understand cognitive decline in older survivors of NHL.
“The aim of the present study was to examine the difference in cognitive status between a group of long-term older survivors of NHL compared with a group of noncancer controls of the same age,” wrote Domenico La Carpia, MD, of Fondazione ANT Italia Onlus, Florence, Italy, and colleagues.
The researchers conducted a multicenter cross-sectional cohort study involving 63 long-term survivors of NHL and 61 age-matched controls. Their report was published in the Journal of Geriatric Oncology.
Eligible survivors and controls were aged 65 years and older. Among both groups, the mean age of study participants was 74 years, and most survivors were women (58.7%).
While cognitive decline was assessed via standardized neuropsychological testing, the team also evaluated polypharmacy, functional status, and level of multimorbidity in the cohort.
Other clinical data, including the time from complete remission, type of treatment received, and histopathological type of tumor, were collected from patient charts and included in the analysis.
After analysis, the researchers found that NHL survivors had a higher mean number of chronic conditions (3.4 vs. 2.3; P = .003), were receiving more medications (3.4 vs. 2.3; P = .03), and had worse functional status compared with controls.
In addition, survivors had impaired executive functioning compared with control subjects (Trail Making Test B-A, 47.9 vs. 32.1; P = .04), but scores on the Mini Mental State Examination (MMSE) did not differ between the groups.
“A small, statistically significant difference was also observed in verbal memory scores between the two groups,” they reported.
The researchers acknowledged that a key limitation was the cross-sectional nature of the study; hence, causality cannot be inferred from the data.
“Comprehensive geriatric assessment for older cancer survivors is advisable to identify those individuals who are at highest risk of developing disability and to implement tailored early interventions,” they concluded.
No funding sources were reported. The authors reported having no conflicts of interest.
SOURCE: La Carpia D et al. J Geriatr Oncol. 2020 Jan 31. doi: 10.1016/j.jgo.2020.01.007.
FROM THE JOURNAL OF GERIATRIC ONCOLOGY
Collaborative Dementia Care via Telephone and Internet Improves Quality of Life and Reduces Caregiver Burden
Study Overview
Objective. To examine the effectiveness of a hub site–based care delivery system in delivering a dementia care management program to persons with dementia and their caregivers.
Design. Randomized pragmatic clinical trial enrolling dyads of persons with dementia and their caregiver. Study participants were randomly assigned to the dementia care management program and usual care in a 2:1 ratio.
Setting and participants. The study was conducted from 2 hub sites: the University of California, San Francisco, and the University of Nebraska Medical Center in Omaha. Each hub-site team served persons with dementia and their caregivers in California, Nebraska, and Iowa in both urban and rural areas. Participants were recruited through referral by treating providers or self-referral in response to advertising presented through a community outreach event, in the news, or on the internet. Eligibility requirements included: having a dementia diagnosis made by a treating provider; age older than 45 years; Medicare or Medicaid enrollment or eligibility; presence of a caregiver willing to enroll in the study; fluency in English, Spanish, or Cantonese; and residence in California, Nebraska, or Iowa. Exclusion criteria included residence in a nursing home. Out of 2585 referred dyads of persons with dementia and caregivers, 780 met inclusion criteria and were enrolled. A 2:1 randomization yielded 512 dyads in the intervention group and 268 dyads in the control group.
Intervention. The dementia care management program was implemented through the Care Ecosystem, a telephone- and internet-based supportive care intervention delivered by care team navigators. The navigators were unlicensed but trained dementia care guides working under the supervision of an advanced practice nurse, social worker, and pharmacist. The intervention consisted of telephone calls, monthly or at a frequency determined by needs and preferences, placed by navigators over a 12-month period; the content of the calls included response to immediate needs of persons with dementia and their caregiver, screening for common problems, and provision of support and education using care plan protocols. Caregivers and persons with dementia were encouraged to initiate contact through email, mail, or telephone for dementia-related questions. Additional support was provided by an advanced practice nurse, social worker, or pharmacist, as needed, and these health care professionals conducted further communication with the persons with dementia, caregiver, or outside professionals, such as physicians, for the persons with dementia, as needed. The average number of telephone calls over the 12-month period was 15.3 (standard deviation, 11.3). Participants assigned to usual care were offered contact information on dementia and aging-related organizations, including the Alzheimer’s Association and the Area Agencies on Aging, and also were sent a quarterly newsletter with general information about dementia.
Main outcome measures. The primary outcome measure was the Quality of Life in Alzheimer’s Disease score obtained by caregiver interview. This quality of life measure includes the following aspects, each rated on an ordinal scale of 1 to 4: physical health, energy level, mood, living situation, memory, family, closest relationship, friends, self, ability to do things for fun, finances, and life as a whole. The scores range from 13 to 52, with a higher score indicating better quality of life for persons with dementia. Other outcomes included frequency of emergency room visits, hospital use, and ambulance use; caregiver depression score from the Patient Health Questionnaire scale; caregiver burden score using the 12-item Zarit Burden Interview; caregiver self-efficacy; and caregiver satisfaction.
Main results. The study found that the quality of life for persons with dementia declined more in the usual care group than in the intervention group during the 12-month study period (difference of 0.53; 95% confidence interval, 0.25-1.3; P = 0.04). Persons with dementia also had fewer emergency room visits, with a number needed to treat to prevent 1 emergency room visit of 5. The intervention did not reduce ambulance use or hospital use. Caregivers in the intervention group had a greater decline in depression when compared to usual care; the frequency of moderate to severe depression decreased from 13.4% at baseline to 7.9% at 12 months (P = 0.004). Caregiver burden declined more in the intervention group than in the control group at 12 months (P = 0.046). In terms of caregiver satisfaction, 97% of caregivers surveyed in the intervention group said they would recommend the intervention to another caregiver; 45% indicated they were very satisfied, and 33% that they were satisfied.
Conclusion. Delivering dementia care via telephone and internet through a collaborative program with care navigators can improve caregiver burden and well-being and improve quality of life, emergency room utilization, and depression for persons with dementia. In addition, the program was well received.
Commentary
Dementia, including Alzheimer’s disease, primarily affects older adults and is characterized by declines in memory and cognitive function. It is often accompanied by neuropsychological symptoms such as agitation, wandering, and physical and verbal outbursts, which are debilitating for persons living with dementia and difficult to cope with for caregivers.1 These symptoms are often the source of caregiver stress, potentially leading to caregiver depression and eventual need for long-term institution-based care, such as nursing home placement.2
Prior literature has established the potential effect of support in improving caregiver outcomes, including caregiver stress and burden, through interventions such as enhancing resources for caregivers, teaching coping strategies to caregivers, and teaching caregivers how to manage support for their loved ones.3,4 However, wider adoption of these interventions may be limited if the interventions involve in-person meetings or activities that take caregivers away from caregiving; the scalability of these programs is also limited by their ability to reach persons with dementia and their caregivers. These barriers are particularly important for older adults living in rural areas, where the availability of resources and distance from access to quality care may be particularly limiting.5 Leveraging advances in technology and telecommunication, this study examined the effects of providing dementia care support via telephone and internet using a trained, unlicensed care navigator as the main point of contact. The results showed improved quality of life for persons with dementia, reduced need for emergency room visits, and reduced caregiver burden and depression. The intervention is promising as a scalable intervention that may impact dementia care nationwide.
Despite the promising results, there are several issues regarding the intervention’s applicability and impact that future studies may help to further clarify. Although the improvement in quality of life in persons with dementia is important to document, it is unclear whether this difference is clinically significant. Also, it may be important to examine whether the 12-month program has sustained impact beyond the study period, although the intervention could be conceived as a long-term care solution. If the intervention is sustained beyond 12 months, future studies may look at other clinical outcomes, such as incidence of institutionalization and perhaps time to institutionalization. The study population consisted of persons with dementia of various stages, half of whom had mild disease. Future studies may further clarify at which stage of dementia the intervention is most useful. Other changes that occurred during the study period, such as change in the use of paid home-based support services and referrals to other relevant evaluations and treatment, may provide further clues about how the dementia care intervention achieved its beneficial effects.
Applications for Clinical Practice
From the health systems perspective, dementia care accounts for significant resources, and these costs are expected to grow as the population ages and dementia prevalence increases. Identifying potentially scalable interventions that yield clinical benefits and are sustainable from a cost perspective is an important step forward in improving care for persons with dementia and their caregivers across the nation. The use of centralized hubs to deliver this intervention and the novel use of telecommunications advances make this intervention applicable across large areas. Policy makers should explore how an intervention such as this could be established and sustained in our health care system.
–William W. Hung, MD, MPH
1. Mega MS, Cummings JL, Fiorello T, Gornbein J. The spectrum of behavioral changes in Alzheimer’s disease. Neurology. 1996;46:130-135.
2. Gallagher-Thompson D, Brooks JO 3rd, Bliwise D, et al. The relations among caregiver stress, “sundowning” symptoms, and cognitive decline in Alzheimer’s disease. J Am Geriatr Soc. 1992;40:807-810.
3. Livingston G, Barber J, Rapaport P, et al. Clinical effectiveness of a manual based coping strategy programme (START, STrAtegies for RelaTives) in promoting the mental health of carers of family members with dementia: pragmatic randomised controlled trial. BMJ. 2013;347:f6276.
4. Belle SH, Burgio L, Burns R, et al; Resources for Enhancing Alzheimer’s Caregiver Health (REACH) II Investigators. Enhancing the quality of life of dementia caregivers from different ethnic or racial groups: a randomized, controlled trial. Ann Intern Med. 2006;145:727-738.
5. Goins RT, Williams KA, Carter MW, et al. Perceived barriers to health care access among rural older adults: a qualitative study. J Rural Health. 2005;21:206-213.
Study Overview
Objective. To examine the effectiveness of a hub site–based care delivery system in delivering a dementia care management program to persons with dementia and their caregivers.
Design. Randomized pragmatic clinical trial enrolling dyads of persons with dementia and their caregiver. Study participants were randomly assigned to the dementia care management program and usual care in a 2:1 ratio.
Setting and participants. The study was conducted from 2 hub sites: the University of California, San Francisco, and the University of Nebraska Medical Center in Omaha. Each hub-site team served persons with dementia and their caregivers in California, Nebraska, and Iowa in both urban and rural areas. Participants were recruited through referral by treating providers or self-referral in response to advertising presented through a community outreach event, in the news, or on the internet. Eligibility requirements included: having a dementia diagnosis made by a treating provider; age older than 45 years; Medicare or Medicaid enrollment or eligibility; presence of a caregiver willing to enroll in the study; fluency in English, Spanish, or Cantonese; and residence in California, Nebraska, or Iowa. Exclusion criteria included residence in a nursing home. Out of 2585 referred dyads of persons with dementia and caregivers, 780 met inclusion criteria and were enrolled. A 2:1 randomization yielded 512 dyads in the intervention group and 268 dyads in the control group.
Intervention. The dementia care management program was implemented through the Care Ecosystem, a telephone- and internet-based supportive care intervention delivered by care team navigators. The navigators were unlicensed but trained dementia care guides working under the supervision of an advanced practice nurse, social worker, and pharmacist. The intervention consisted of telephone calls, monthly or at a frequency determined by needs and preferences, placed by navigators over a 12-month period; the content of the calls included response to immediate needs of persons with dementia and their caregiver, screening for common problems, and provision of support and education using care plan protocols. Caregivers and persons with dementia were encouraged to initiate contact through email, mail, or telephone for dementia-related questions. Additional support was provided by an advanced practice nurse, social worker, or pharmacist, as needed, and these health care professionals conducted further communication with the persons with dementia, caregiver, or outside professionals, such as physicians, for the persons with dementia, as needed. The average number of telephone calls over the 12-month period was 15.3 (standard deviation, 11.3). Participants assigned to usual care were offered contact information on dementia and aging-related organizations, including the Alzheimer’s Association and the Area Agencies on Aging, and also were sent a quarterly newsletter with general information about dementia.
Main outcome measures. The primary outcome measure was the Quality of Life in Alzheimer’s Disease score obtained by caregiver interview. This quality of life measure includes the following aspects, each rated on an ordinal scale of 1 to 4: physical health, energy level, mood, living situation, memory, family, closest relationship, friends, self, ability to do things for fun, finances, and life as a whole. The scores range from 13 to 52, with a higher score indicating better quality of life for persons with dementia. Other outcomes included frequency of emergency room visits, hospital use, and ambulance use; caregiver depression score from the Patient Health Questionnaire scale; caregiver burden score using the 12-item Zarit Burden Interview; caregiver self-efficacy; and caregiver satisfaction.
Main results. The study found that the quality of life for persons with dementia declined more in the usual care group than in the intervention group during the 12-month study period (difference of 0.53; 95% confidence interval, 0.25-1.3; P = 0.04). Persons with dementia also had fewer emergency room visits, with a number needed to treat to prevent 1 emergency room visit of 5. The intervention did not reduce ambulance use or hospital use. Caregivers in the intervention group had a greater decline in depression when compared to usual care; the frequency of moderate to severe depression decreased from 13.4% at baseline to 7.9% at 12 months (P = 0.004). Caregiver burden declined more in the intervention group than in the control group at 12 months (P = 0.046). In terms of caregiver satisfaction, 97% of caregivers surveyed in the intervention group said they would recommend the intervention to another caregiver; 45% indicated they were very satisfied, and 33% that they were satisfied.
Conclusion. Delivering dementia care via telephone and internet through a collaborative program with care navigators can improve caregiver burden and well-being and improve quality of life, emergency room utilization, and depression for persons with dementia. In addition, the program was well received.
Commentary
Dementia, including Alzheimer’s disease, primarily affects older adults and is characterized by declines in memory and cognitive function. It is often accompanied by neuropsychological symptoms such as agitation, wandering, and physical and verbal outbursts, which are debilitating for persons living with dementia and difficult to cope with for caregivers.1 These symptoms are often the source of caregiver stress, potentially leading to caregiver depression and eventual need for long-term institution-based care, such as nursing home placement.2
Prior literature has established the potential effect of support in improving caregiver outcomes, including caregiver stress and burden, through interventions such as enhancing resources for caregivers, teaching coping strategies to caregivers, and teaching caregivers how to manage support for their loved ones.3,4 However, wider adoption of these interventions may be limited if the interventions involve in-person meetings or activities that take caregivers away from caregiving; the scalability of these programs is also limited by their ability to reach persons with dementia and their caregivers. These barriers are particularly important for older adults living in rural areas, where the availability of resources and distance from access to quality care may be particularly limiting.5 Leveraging advances in technology and telecommunication, this study examined the effects of providing dementia care support via telephone and internet using a trained, unlicensed care navigator as the main point of contact. The results showed improved quality of life for persons with dementia, reduced need for emergency room visits, and reduced caregiver burden and depression. The intervention is promising as a scalable intervention that may impact dementia care nationwide.
Despite the promising results, there are several issues regarding the intervention’s applicability and impact that future studies may help to further clarify. Although the improvement in quality of life in persons with dementia is important to document, it is unclear whether this difference is clinically significant. Also, it may be important to examine whether the 12-month program has sustained impact beyond the study period, although the intervention could be conceived as a long-term care solution. If the intervention is sustained beyond 12 months, future studies may look at other clinical outcomes, such as incidence of institutionalization and perhaps time to institutionalization. The study population consisted of persons with dementia of various stages, half of whom had mild disease. Future studies may further clarify at which stage of dementia the intervention is most useful. Other changes that occurred during the study period, such as change in the use of paid home-based support services and referrals to other relevant evaluations and treatment, may provide further clues about how the dementia care intervention achieved its beneficial effects.
Applications for Clinical Practice
From the health systems perspective, dementia care accounts for significant resources, and these costs are expected to grow as the population ages and dementia prevalence increases. Identifying potentially scalable interventions that yield clinical benefits and are sustainable from a cost perspective is an important step forward in improving care for persons with dementia and their caregivers across the nation. The use of centralized hubs to deliver this intervention and the novel use of telecommunications advances make this intervention applicable across large areas. Policy makers should explore how an intervention such as this could be established and sustained in our health care system.
–William W. Hung, MD, MPH
Study Overview
Objective. To examine the effectiveness of a hub site–based care delivery system in delivering a dementia care management program to persons with dementia and their caregivers.
Design. Randomized pragmatic clinical trial enrolling dyads of persons with dementia and their caregiver. Study participants were randomly assigned to the dementia care management program and usual care in a 2:1 ratio.
Setting and participants. The study was conducted from 2 hub sites: the University of California, San Francisco, and the University of Nebraska Medical Center in Omaha. Each hub-site team served persons with dementia and their caregivers in California, Nebraska, and Iowa in both urban and rural areas. Participants were recruited through referral by treating providers or self-referral in response to advertising presented through a community outreach event, in the news, or on the internet. Eligibility requirements included: having a dementia diagnosis made by a treating provider; age older than 45 years; Medicare or Medicaid enrollment or eligibility; presence of a caregiver willing to enroll in the study; fluency in English, Spanish, or Cantonese; and residence in California, Nebraska, or Iowa. Exclusion criteria included residence in a nursing home. Out of 2585 referred dyads of persons with dementia and caregivers, 780 met inclusion criteria and were enrolled. A 2:1 randomization yielded 512 dyads in the intervention group and 268 dyads in the control group.
Intervention. The dementia care management program was implemented through the Care Ecosystem, a telephone- and internet-based supportive care intervention delivered by care team navigators. The navigators were unlicensed but trained dementia care guides working under the supervision of an advanced practice nurse, social worker, and pharmacist. The intervention consisted of telephone calls, monthly or at a frequency determined by needs and preferences, placed by navigators over a 12-month period; the content of the calls included response to immediate needs of persons with dementia and their caregiver, screening for common problems, and provision of support and education using care plan protocols. Caregivers and persons with dementia were encouraged to initiate contact through email, mail, or telephone for dementia-related questions. Additional support was provided by an advanced practice nurse, social worker, or pharmacist, as needed, and these health care professionals conducted further communication with the persons with dementia, caregiver, or outside professionals, such as physicians, for the persons with dementia, as needed. The average number of telephone calls over the 12-month period was 15.3 (standard deviation, 11.3). Participants assigned to usual care were offered contact information on dementia and aging-related organizations, including the Alzheimer’s Association and the Area Agencies on Aging, and also were sent a quarterly newsletter with general information about dementia.
Main outcome measures. The primary outcome measure was the Quality of Life in Alzheimer’s Disease score obtained by caregiver interview. This quality of life measure includes the following aspects, each rated on an ordinal scale of 1 to 4: physical health, energy level, mood, living situation, memory, family, closest relationship, friends, self, ability to do things for fun, finances, and life as a whole. The scores range from 13 to 52, with a higher score indicating better quality of life for persons with dementia. Other outcomes included frequency of emergency room visits, hospital use, and ambulance use; caregiver depression score from the Patient Health Questionnaire scale; caregiver burden score using the 12-item Zarit Burden Interview; caregiver self-efficacy; and caregiver satisfaction.
Main results. The study found that the quality of life for persons with dementia declined more in the usual care group than in the intervention group during the 12-month study period (difference of 0.53; 95% confidence interval, 0.25-1.3; P = 0.04). Persons with dementia also had fewer emergency room visits, with a number needed to treat to prevent 1 emergency room visit of 5. The intervention did not reduce ambulance use or hospital use. Caregivers in the intervention group had a greater decline in depression when compared to usual care; the frequency of moderate to severe depression decreased from 13.4% at baseline to 7.9% at 12 months (P = 0.004). Caregiver burden declined more in the intervention group than in the control group at 12 months (P = 0.046). In terms of caregiver satisfaction, 97% of caregivers surveyed in the intervention group said they would recommend the intervention to another caregiver; 45% indicated they were very satisfied, and 33% that they were satisfied.
Conclusion. Delivering dementia care via telephone and internet through a collaborative program with care navigators can improve caregiver burden and well-being and improve quality of life, emergency room utilization, and depression for persons with dementia. In addition, the program was well received.
Commentary
Dementia, including Alzheimer’s disease, primarily affects older adults and is characterized by declines in memory and cognitive function. It is often accompanied by neuropsychological symptoms such as agitation, wandering, and physical and verbal outbursts, which are debilitating for persons living with dementia and difficult to cope with for caregivers.1 These symptoms are often the source of caregiver stress, potentially leading to caregiver depression and eventual need for long-term institution-based care, such as nursing home placement.2
Prior literature has established the potential effect of support in improving caregiver outcomes, including caregiver stress and burden, through interventions such as enhancing resources for caregivers, teaching coping strategies to caregivers, and teaching caregivers how to manage support for their loved ones.3,4 However, wider adoption of these interventions may be limited if the interventions involve in-person meetings or activities that take caregivers away from caregiving; the scalability of these programs is also limited by their ability to reach persons with dementia and their caregivers. These barriers are particularly important for older adults living in rural areas, where the availability of resources and distance from access to quality care may be particularly limiting.5 Leveraging advances in technology and telecommunication, this study examined the effects of providing dementia care support via telephone and internet using a trained, unlicensed care navigator as the main point of contact. The results showed improved quality of life for persons with dementia, reduced need for emergency room visits, and reduced caregiver burden and depression. The intervention is promising as a scalable intervention that may impact dementia care nationwide.
Despite the promising results, there are several issues regarding the intervention’s applicability and impact that future studies may help to further clarify. Although the improvement in quality of life in persons with dementia is important to document, it is unclear whether this difference is clinically significant. Also, it may be important to examine whether the 12-month program has sustained impact beyond the study period, although the intervention could be conceived as a long-term care solution. If the intervention is sustained beyond 12 months, future studies may look at other clinical outcomes, such as incidence of institutionalization and perhaps time to institutionalization. The study population consisted of persons with dementia of various stages, half of whom had mild disease. Future studies may further clarify at which stage of dementia the intervention is most useful. Other changes that occurred during the study period, such as change in the use of paid home-based support services and referrals to other relevant evaluations and treatment, may provide further clues about how the dementia care intervention achieved its beneficial effects.
Applications for Clinical Practice
From the health systems perspective, dementia care accounts for significant resources, and these costs are expected to grow as the population ages and dementia prevalence increases. Identifying potentially scalable interventions that yield clinical benefits and are sustainable from a cost perspective is an important step forward in improving care for persons with dementia and their caregivers across the nation. The use of centralized hubs to deliver this intervention and the novel use of telecommunications advances make this intervention applicable across large areas. Policy makers should explore how an intervention such as this could be established and sustained in our health care system.
–William W. Hung, MD, MPH
1. Mega MS, Cummings JL, Fiorello T, Gornbein J. The spectrum of behavioral changes in Alzheimer’s disease. Neurology. 1996;46:130-135.
2. Gallagher-Thompson D, Brooks JO 3rd, Bliwise D, et al. The relations among caregiver stress, “sundowning” symptoms, and cognitive decline in Alzheimer’s disease. J Am Geriatr Soc. 1992;40:807-810.
3. Livingston G, Barber J, Rapaport P, et al. Clinical effectiveness of a manual based coping strategy programme (START, STrAtegies for RelaTives) in promoting the mental health of carers of family members with dementia: pragmatic randomised controlled trial. BMJ. 2013;347:f6276.
4. Belle SH, Burgio L, Burns R, et al; Resources for Enhancing Alzheimer’s Caregiver Health (REACH) II Investigators. Enhancing the quality of life of dementia caregivers from different ethnic or racial groups: a randomized, controlled trial. Ann Intern Med. 2006;145:727-738.
5. Goins RT, Williams KA, Carter MW, et al. Perceived barriers to health care access among rural older adults: a qualitative study. J Rural Health. 2005;21:206-213.
1. Mega MS, Cummings JL, Fiorello T, Gornbein J. The spectrum of behavioral changes in Alzheimer’s disease. Neurology. 1996;46:130-135.
2. Gallagher-Thompson D, Brooks JO 3rd, Bliwise D, et al. The relations among caregiver stress, “sundowning” symptoms, and cognitive decline in Alzheimer’s disease. J Am Geriatr Soc. 1992;40:807-810.
3. Livingston G, Barber J, Rapaport P, et al. Clinical effectiveness of a manual based coping strategy programme (START, STrAtegies for RelaTives) in promoting the mental health of carers of family members with dementia: pragmatic randomised controlled trial. BMJ. 2013;347:f6276.
4. Belle SH, Burgio L, Burns R, et al; Resources for Enhancing Alzheimer’s Caregiver Health (REACH) II Investigators. Enhancing the quality of life of dementia caregivers from different ethnic or racial groups: a randomized, controlled trial. Ann Intern Med. 2006;145:727-738.
5. Goins RT, Williams KA, Carter MW, et al. Perceived barriers to health care access among rural older adults: a qualitative study. J Rural Health. 2005;21:206-213.
Switching from TDF- to TAF-Containing Antiretroviral Therapy: Impact on Bone Mineral Density in Older Patients Living With HIV
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
Study Overview
Objective. To evaluate the effect of changing from tenofovir disoproxil fumarate (TDF) –containing antiretroviral therapy (ART) to tenofovir alafenamide (TAF) –containing ART in patients ages 60 years and older living with HIV.
Design. Prospective, open-label, multicenter, randomized controlled trial.
Setting and participants. The study was completed across 36 European centers over 48 weeks. Patients were enrolled from December 12, 2015, to March 21, 2018, and were eligible to participate if they were diagnosed with HIV-1; virologically suppressed to < 50 copies/mL; on a TDF-containing ART regimen; and ≥ 60 years of age.
Intervention. Participants (n = 167) were randomly assigned in a 2:1 ratio to ART with TAF (10 mg), elvitegravir (EVG; 150 mg), cobicistat (COB; 150 mg), and emtricitabine (FTC; 200 mg) or to continued therapy with a TDF-containing ART regimen (300 mg TDF).
Main outcome measures. Primary outcome measures were the change in spine and hip bone mineral density from baseline at week 48. Secondary outcome measures included bone mineral density changes from baseline at week 24, HIV viral suppression and change in CD4 count at weeks 24 and 48, and the assessment of safety and tolerability of each ART regimen until week 48.
Main results. At 48 weeks, patients (n = 111) in the TAF+EVG+COB+FTC group had a mean 2.24% (SD, 3.27) increase in spine bone mineral density, while those in the TDF-containing group (n = 56) had a mean 0.10% decrease (SD, 3.39), a difference of 2.43% (95% confidence interval [CI], 1.34-3.52; P < 0.0001). In addition, at 48 weeks patients in the TAF+EVG+COB+FTC group had a mean 1.33% increase (SD, 2.20) in hip bone mineral density, as compared with a mean 0.73% decrease (SD, 3.21) in the TDF-containing group, a difference of 2.04% (95% CI, 1.17-2.90; P < 0.0001).
Similar results were seen in spine and hip bone mineral density in the TAF+EVG+COB+FTC group at week 24, with increases of 1.75% (P = 0.00080) and 1.35% (P = 0.00040), respectively. Both treatment groups maintained high virologic suppression. The TAF+EVG+COB+FTC group maintained 94.5% virologic suppression at week 24 and 93.6% at week 48, as compared with virologic suppression of 100% and 94.5% at weeks 24 and 48, respectively, in the TDF-containing group. However, the TAF+EVG+COB+FTC group had an increase in CD4 count from baseline (56 cells/µL), with no real change in the TDF-containing group (–1 cell/µL). Patients in the TAF+EVG+COB+FTC group had a mean 27.8 mg/g decrease in urine albumin-to-creatinine ratio (UACR) versus a 7.7 mg/g decrease in the TDF-containing group (P = 0.0042). In addition, patients in the TAF+EVG+COB+FTC group had a mean 49.8 mg/g decrease in urine protein-to-creatinine ratio (UPCR) versus a 3.8 mg/g decrease in the TDF-containing group (P = 0.0042).
Conclusion. Patients 60 years of age or older living with virologically suppressed HIV may benefit from improved bone mineral density by switching from a TDF-containing ART regimen to a TAF-containing regimen after 48 weeks, which, in turn, may help to reduce the risk for osteoporosis. Patients who were switched to a TAF-containing regimen also had favorable improvements in UACR and UPCR, which could indicate better renal function.
The Centers for Disease Control and Prevention estimated that in 2018 nearly half of those living with HIV in the United States were older than 50 years.1 Today, the life expectancy of patients living with HIV on ART in developed countries is similar to that of patients not living with HIV. A meta-analysis published in 2017 estimated that patients diagnosed with HIV at age 20 beginning ART have a life expectancy of 63 years, and another study estimated that life expectancy in such patients is 89.1% of that of the general population in Canada.2,3 Overall, most people living with HIV infection are aging and at risk for medical conditions similar to persons without HIV disease. However, rates of osteoporosis in elderly patients with HIV are estimated to be 3 times greater than rates in persons without HIV.4 As a result, it is becoming increasingly important to find ways to decrease the risk of osteoporosis in these patients.
ART typically includes a nucleoside reverse transcriptase inhibitor (NRTI) combination and a third agent, such as an integrase strand inhibitor. Tenofovir is a commonly used backbone NRTI that comes in 2 forms, TDF (tenofovir disoproxil fumarate) and TAF (tenofovir alafenamide). Both are prodrugs that are converted to tenofovir diphosphate. TDF specifically is associated with an increased risk of bone loss and nephrotoxicity. The loss in bone mineral density is most similar to the bone loss seen with oral glucocorticoids.5 TDF has been shown to increase plasma levels of RANKL and tumor necrosis factor-α, leading to increased bone resorption.6 The long-term effects of TDF- versus TAF-containing ART on bone mineral density have, to our knowledge, not been compared previously in a randomized control study. The significance of demonstrating an increase in bone mineral density in the prevention of osteoporotic bone fracture in people living with HIV is less clear. A long-term cohort study completed in Japan looking at patients on TDF showed an increased risk of bone fractures in both older postmenopausal women and younger men.7 However, a retrospective cohort study looking at 1981 patients with HIV found no association between bone fractures and TDF.8
This randomized controlled trial used appropriate methods to measure the reported primary and secondary endpoints; however, it would be of benefit to continue following these patients to measure their true long-term risk of osteoporosis-related complications. In terms of the study’s secondary endpoints, it is notable that the patients maintained HIV viral suppression after the switch and CD4 counts remained stable (with a slight increase observed in the TAF-containing ART cohort).
In regard to the patient’s renal function, patients in the TAF group had significantly improved UACR and UPCR, which likely reflects improved glomerular filtration. Improved renal function is also increasingly important for patients with HIV, as up to 48.5% have some form of chronic kidney disease.9
Applications for Clinical Practice
This study shows that making the switch from TDF- to TAF-containing ART can lead to improved bone mineral density. We can extrapolate that switching may lead to a decreased risk of osteoporosis and osteoporosis-related complications, such as bone fracture, but this needs to be investigated in more detail. As demonstrated in this study, switching from a TDF- to a TAF-containing regimen can also lead to improved renal function while maintaining HIV viral suppression and CD4 counts.
Unfortunately, the regimen selected with TAF in this study (elvitegravir, cobicistat, and emtricitabine) includes cobicistat, which is no longer recommended as initial therapy due to its risk of drug-drug interactions, and elvitegravir, which has a lower barrier to resistance than other integrase strand inhibitors.10,11 The United States Department of Health and Human Services guidelines and the International Antiviral Society-USA Panel suggest using several other TAF-containing regimens for beginning or even switching therapy in older patients.10,11
When choosing between either a TAF- or a TDF-containing regimen to treat HIV infection in older patients, increasing evidence shows that using a TAF-containing ART regimen may be more beneficial for people living and aging with virologically suppressed HIV infection.
–Sean P. Bliven, and Norman L. Beatty, MD, University of Florida College of Medicine, Division of Infectious Diseases and Global Medicine, Gainesville, FL
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
1. Centers for Disease Control and Prevention. HIV among people aged 50 and over. 2018. https://www.cdc.gov/hiv/group/age/olderamericans/index.html. Accessed on November 22, 2019.
2. Teeraananchai S, Kerr S, Amin J, et al. Life expectancy of HIV-positive people after starting combination antiretroviral therapy: a meta-analysis. HIV Medicine. 2016;18:256-266.
3. Wandeler G, Johnson LF, Egger M. Trends in life expectancy of HIV-positive adults on antiretroviral therapy across the globe. Curr Opin HIV AIDS. 2016;11:492-500.
4. Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165-2174.
5. Bolland MJ, Grey A, Reid IR. Skeletal health in adults with HIV infection. Lancet Diabetes Endocrinol. 2015;3:63-74.
6. Ofotokun I, Titanji K, Vunnava A, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405-414.
7. Komatsu A, Ikeda A, Kikuchi A, et al. Osteoporosis-related fractures in HIV-infected patients receiving long-term tenofovir disoproxil fumarate: an observational cohort study. Drug Saf. 2018;41:843-848.
8. Gediminas L, Wright EA, Dong Y, et al. Factors associated with fractures in HIV-infected persons: which factors matter? Osteoporos Int. 201728:239-244.
9. Naicker S, Rahmania, Kopp JB. HIV and chronic kidney disease. Clin Nephrol. 2015; 83(Suppl 1):S32-S38.
10. United States Department of Health and Human Services. Guidelines for the use of antiretroviral agents in adults and adolescents living with HIV. https://aidsinfo.nih.gov/guidelines/html/1/adult-and-adolescent-arv/0. Accessed December 10, 2019.
11. Saag MS, Benson CA, Gandhi RT, et al. Antiretroviral drugs for treatment and prevention of HIV infection in adults: 2018 recommendations of the International Antiviral Society-USA Panel. JAMA. 2018;320:379-396.
Dietary flavonol intake linked to reduced risk of Alzheimer’s
Onset of Alzheimer’s disease (AD) was inversely associated with intake of flavonols, a subclass of flavonoids with antioxidant and anti-inflammatory properties, according to the study authors.
The rate of developing AD was reduced by 50% among individuals reporting high intake of kaempferol, a flavonol plentiful in leafy green vegetables, and by 38% for high intake of the flavonols myricetin and isorhamnetin, researchers said in a report published in Neurology.
The findings are from the Rush Memory and Aging Project (MAP), a large, prospective study of older individuals in retirement communities and public housing in the Chicago area that has been ongoing since 1997.
“Although there is more work to be done, the associations that we observed are promising and deserve further study,” said Thomas M. Holland, MD, of the Rush Institute for Healthy Aging in Chicago, and coauthors.
Those associations between flavonol intake and AD help set the stage for U.S. POINTER and other randomized, controlled trials that seek to evaluate the effects of dietary interventions in a more rigorous way, according to Laura D. Baker, PhD, associate professor of internal medicine at Wake Forest University, Winston-Salem, N.C.
“This kind of data helps us feel like we are looking in the right direction in the randomized, controlled trials,” Dr. Baker said in an interview.
Dr. Baker is an investigator in the U.S. POINTER study, which will in part evaluate the impact of the MIND diet, which has been shown to slow cognitive decline with age in a previously published MAP study.
However, in the absence of randomized, controlled trial data, Dr. Baker cautioned against “prematurely advocating” for specific dietary approaches when speaking to patients and caregivers now.
“What I say is, we know for sure that the standard American Heart Association diet has been shown in clinical trials to reduce the risk of heart disease, and in terms of brain health, if you can reduce risk of heart disease, you are protecting your brain,” she said in the interview.
The present MAP study linking a reduced rate of AD to flavonol consumption is believed to be the first of its kind, though two previous studies from the early 2000s did find inverse associations between incident AD and intake of flavonoids, of which flavonoids are just one subclass, said Dr. Holland and coinvestigators in their report.
Moreover, in a MAP study published in 2018, Martha Clare Morris, ScD, and coauthors concluded that consuming about a serving per day of green leafy vegetables and foods rich in kaempferol, among other nutrients and bioactive compounds, may help slow cognitive decline associated with aging.
To more specifically study the relationship between kaempferol and other flavonols and the development of AD, Dr. Holland and colleagues evaluated data for MAP participants who had completed a comprehensive food frequency questionnaire and underwent at least two evaluations to assess incidence of disease.
The mean age of the 921 individuals in the present analysis was 81 years, three-quarters were female, and over approximately 6 years of follow-up, 220 developed AD.
The rate of developing AD was 48% lower among participants reporting the highest total dietary intake of flavonols, compared with those reporting the lowest intake, Dr. Holland and coauthors reported.
Intake of the specific flavonols kaempferol, myricetin, and isorhamnetin were associated with incident AD reductions of 50%, 38%, and 38%, respectively. Another flavonol, quercetin, was by contrast not inversely associated with incident AD, according to the report.
Kaempferol was independently associated with AD in subsequent analyses, while there was no such independent association for myricetin, isorhamnetin, or quercetin, according to Dr. Holland and coinvestigators.
Further analyses of the data suggested the linkages between flavonols and AD were independent of lifestyle factors, dietary intakes, or cardiovascular conditions, they said in their report.
“Confirmation of these findings is warranted through other longitudinal epidemiologic studies and clinical trials, in addition to further elucidation of the biologic mechanisms,” they concluded.
The study was funded by grants from the National Institutes of Health and the USDA Agricultural Research Service. Dr. Holland and coauthors said that they had no disclosures relevant to their report.
SOURCE: Holland TM et al. Neurology. 2020 Jan 29. doi: 10.1212/WNL.0000000000008981.
Onset of Alzheimer’s disease (AD) was inversely associated with intake of flavonols, a subclass of flavonoids with antioxidant and anti-inflammatory properties, according to the study authors.
The rate of developing AD was reduced by 50% among individuals reporting high intake of kaempferol, a flavonol plentiful in leafy green vegetables, and by 38% for high intake of the flavonols myricetin and isorhamnetin, researchers said in a report published in Neurology.
The findings are from the Rush Memory and Aging Project (MAP), a large, prospective study of older individuals in retirement communities and public housing in the Chicago area that has been ongoing since 1997.
“Although there is more work to be done, the associations that we observed are promising and deserve further study,” said Thomas M. Holland, MD, of the Rush Institute for Healthy Aging in Chicago, and coauthors.
Those associations between flavonol intake and AD help set the stage for U.S. POINTER and other randomized, controlled trials that seek to evaluate the effects of dietary interventions in a more rigorous way, according to Laura D. Baker, PhD, associate professor of internal medicine at Wake Forest University, Winston-Salem, N.C.
“This kind of data helps us feel like we are looking in the right direction in the randomized, controlled trials,” Dr. Baker said in an interview.
Dr. Baker is an investigator in the U.S. POINTER study, which will in part evaluate the impact of the MIND diet, which has been shown to slow cognitive decline with age in a previously published MAP study.
However, in the absence of randomized, controlled trial data, Dr. Baker cautioned against “prematurely advocating” for specific dietary approaches when speaking to patients and caregivers now.
“What I say is, we know for sure that the standard American Heart Association diet has been shown in clinical trials to reduce the risk of heart disease, and in terms of brain health, if you can reduce risk of heart disease, you are protecting your brain,” she said in the interview.
The present MAP study linking a reduced rate of AD to flavonol consumption is believed to be the first of its kind, though two previous studies from the early 2000s did find inverse associations between incident AD and intake of flavonoids, of which flavonoids are just one subclass, said Dr. Holland and coinvestigators in their report.
Moreover, in a MAP study published in 2018, Martha Clare Morris, ScD, and coauthors concluded that consuming about a serving per day of green leafy vegetables and foods rich in kaempferol, among other nutrients and bioactive compounds, may help slow cognitive decline associated with aging.
To more specifically study the relationship between kaempferol and other flavonols and the development of AD, Dr. Holland and colleagues evaluated data for MAP participants who had completed a comprehensive food frequency questionnaire and underwent at least two evaluations to assess incidence of disease.
The mean age of the 921 individuals in the present analysis was 81 years, three-quarters were female, and over approximately 6 years of follow-up, 220 developed AD.
The rate of developing AD was 48% lower among participants reporting the highest total dietary intake of flavonols, compared with those reporting the lowest intake, Dr. Holland and coauthors reported.
Intake of the specific flavonols kaempferol, myricetin, and isorhamnetin were associated with incident AD reductions of 50%, 38%, and 38%, respectively. Another flavonol, quercetin, was by contrast not inversely associated with incident AD, according to the report.
Kaempferol was independently associated with AD in subsequent analyses, while there was no such independent association for myricetin, isorhamnetin, or quercetin, according to Dr. Holland and coinvestigators.
Further analyses of the data suggested the linkages between flavonols and AD were independent of lifestyle factors, dietary intakes, or cardiovascular conditions, they said in their report.
“Confirmation of these findings is warranted through other longitudinal epidemiologic studies and clinical trials, in addition to further elucidation of the biologic mechanisms,” they concluded.
The study was funded by grants from the National Institutes of Health and the USDA Agricultural Research Service. Dr. Holland and coauthors said that they had no disclosures relevant to their report.
SOURCE: Holland TM et al. Neurology. 2020 Jan 29. doi: 10.1212/WNL.0000000000008981.
Onset of Alzheimer’s disease (AD) was inversely associated with intake of flavonols, a subclass of flavonoids with antioxidant and anti-inflammatory properties, according to the study authors.
The rate of developing AD was reduced by 50% among individuals reporting high intake of kaempferol, a flavonol plentiful in leafy green vegetables, and by 38% for high intake of the flavonols myricetin and isorhamnetin, researchers said in a report published in Neurology.
The findings are from the Rush Memory and Aging Project (MAP), a large, prospective study of older individuals in retirement communities and public housing in the Chicago area that has been ongoing since 1997.
“Although there is more work to be done, the associations that we observed are promising and deserve further study,” said Thomas M. Holland, MD, of the Rush Institute for Healthy Aging in Chicago, and coauthors.
Those associations between flavonol intake and AD help set the stage for U.S. POINTER and other randomized, controlled trials that seek to evaluate the effects of dietary interventions in a more rigorous way, according to Laura D. Baker, PhD, associate professor of internal medicine at Wake Forest University, Winston-Salem, N.C.
“This kind of data helps us feel like we are looking in the right direction in the randomized, controlled trials,” Dr. Baker said in an interview.
Dr. Baker is an investigator in the U.S. POINTER study, which will in part evaluate the impact of the MIND diet, which has been shown to slow cognitive decline with age in a previously published MAP study.
However, in the absence of randomized, controlled trial data, Dr. Baker cautioned against “prematurely advocating” for specific dietary approaches when speaking to patients and caregivers now.
“What I say is, we know for sure that the standard American Heart Association diet has been shown in clinical trials to reduce the risk of heart disease, and in terms of brain health, if you can reduce risk of heart disease, you are protecting your brain,” she said in the interview.
The present MAP study linking a reduced rate of AD to flavonol consumption is believed to be the first of its kind, though two previous studies from the early 2000s did find inverse associations between incident AD and intake of flavonoids, of which flavonoids are just one subclass, said Dr. Holland and coinvestigators in their report.
Moreover, in a MAP study published in 2018, Martha Clare Morris, ScD, and coauthors concluded that consuming about a serving per day of green leafy vegetables and foods rich in kaempferol, among other nutrients and bioactive compounds, may help slow cognitive decline associated with aging.
To more specifically study the relationship between kaempferol and other flavonols and the development of AD, Dr. Holland and colleagues evaluated data for MAP participants who had completed a comprehensive food frequency questionnaire and underwent at least two evaluations to assess incidence of disease.
The mean age of the 921 individuals in the present analysis was 81 years, three-quarters were female, and over approximately 6 years of follow-up, 220 developed AD.
The rate of developing AD was 48% lower among participants reporting the highest total dietary intake of flavonols, compared with those reporting the lowest intake, Dr. Holland and coauthors reported.
Intake of the specific flavonols kaempferol, myricetin, and isorhamnetin were associated with incident AD reductions of 50%, 38%, and 38%, respectively. Another flavonol, quercetin, was by contrast not inversely associated with incident AD, according to the report.
Kaempferol was independently associated with AD in subsequent analyses, while there was no such independent association for myricetin, isorhamnetin, or quercetin, according to Dr. Holland and coinvestigators.
Further analyses of the data suggested the linkages between flavonols and AD were independent of lifestyle factors, dietary intakes, or cardiovascular conditions, they said in their report.
“Confirmation of these findings is warranted through other longitudinal epidemiologic studies and clinical trials, in addition to further elucidation of the biologic mechanisms,” they concluded.
The study was funded by grants from the National Institutes of Health and the USDA Agricultural Research Service. Dr. Holland and coauthors said that they had no disclosures relevant to their report.
SOURCE: Holland TM et al. Neurology. 2020 Jan 29. doi: 10.1212/WNL.0000000000008981.
FROM NEUROLOGY
BP levels during endovascular stroke therapy affect neurologic outcomes
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
For patients with acute ischemic stroke, prolonged durations of blood pressure above or below certain thresholds during endovascular therapy may be linked to poor functional outcome, results of a retrospective study suggest.
Mean arterial blood pressure (MABP) lower than 70 mm Hg for 10 minutes or more, or higher than 90 mm Hg for 45 minutes or more, represented “critical thresholds” associated with worse neurologic outcomes, the study authors wrote in JAMA Neurology.
“These results suggest MABP may be a modifiable therapeutic target to prevent or reduce poor functional outcome in patients undergoing endovascular therapy for acute ischemic stroke, and that MABP should possibly be maintained within such narrow limits, wrote the authors, led by Mads Rasmussen, MD, PhD, of the department of anesthesia at Aarhus (Denmark) University Hospital.
The findings come from an analysis of BP data from 365 patients with acute ischemic stroke enrolled in three randomized trials evaluating different strategies for anesthesia. Among those patients, the mean age was approximately 71 years, and about 45% were women.
The investigators looked at a variety of BP-related variables during endovascular therapy to assess their impact on functional outcome, based on modified Rankin Scale (mRS) scores at 90 days.
Having an MABP below 70 mm Hg for a cumulative time of at least 10 minutes substantially increased odds of higher 90-day mRS scores (odds ratio, 1.51; 95% confidence interval, 1.02-2.22), according to Dr. Rasmussen and colleagues. The number needed to harm (NNH) at this threshold was 10; in other words, to harm 1 patient, 10 patients are needed with procedural MABP below 70 mm Hg for at least 10 minutes.
Likewise, having an MABP above 90 mm Hg for a cumulated time of at least 45 minutes significantly increased odds of higher 90-day mRS scores, with an OR of 1.49 (95% CI, 1.11-2.02) and a number needed to harm of 10.
Odds of shifting toward a worse neurologic outcome increased by 62% for every continuous 10 minutes of MABP below 70 mm Hg, and by 8% for every continuous 10 minutes above 90 mm Hg.
The maximum MABP during the procedure was significantly associated with neurologic outcomes in the study, while by contrast, maximum procedural systolic BP was not, according to the investigators.
In general, the study findings suggest that MABP is “more sensitive” than systolic BP when assessing hypotension and hypertension in these patients. However, these findings are subject to a number of limitations, the investigators wrote, including the retrospective nature of the analysis and the selected group of patients enrolled in studies designed to evaluate anesthesia strategies, not hemodynamic management.
“Randomized studies are needed to determine the optimal blood pressure management strategy during endovascular therapy,” the investigators wrote.
Dr. Rasmussen reported grant support from the Health Research Foundation of Central Denmark Region and the National Helicopter Emergency Medical Service Foundation. Coauthors reported receiving grant support from the Novo Nordisk Foundation; a research award from the Patient-Centered Outcomes Research Institute; and personal fees from Abbott Medical Sweden, I4L Innovation for Life, Boehringer Ingelheim, Medtronic, and Zoll.
SOURCE: Rasmussen M et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4838.
FROM JAMA NEUROLOGY
Cognitive screening of older physicians: What’s fair?
Cognitive screening of 141 clinicians 70 years or older at Yale New Haven (Conn.) Hospital identified 18 with cognitive deficits likely to impair their ability to practice medicine. Six retired and 12 agreed to limit their practice to closely proctored environments, according to a report in JAMA.
It was part of a program to screen all practitioners 70 years or older who apply for reappointment to the medical staff, and every 2 years thereafter, due to “concerns about the potentially compromised ability of older clinicians,” said the authors, Yale rheumatologist and geriatrician Leo M. Cooney Jr., MD, and Thomas Balcezak, MD, Yale New Haven’s chief medical officer.
Yale is not alone. Intermountain Healthcare, Stanford Hospitals and Clinics, Scripps Health Care, Penn Medicine, and the University of California, San Diego, are among the institutions with similar programs.
The move is being driven by the aging of the medical community. About 15% of U.S. physicians are over 65 years old, a tripling from 23,000 in 1980 to 73,000 in 2012-2016, and the number is growing, according to an editorial by Jeffrey L. Saver, MD, professor of neurology and senior associate vice president of neurology at the University of California, Los Angeles.
Given the trend, “it is not surprising that the issue of screening aging physicians for cognitive deficits has gained attention over the last decade,” Katrina Armstrong, MD, chair of the department of medicine at Massachusetts General Hospital, Boston, and Eileen E. Reynolds, MD, associate professor of medicine at Beth Israel Deaconess Medical Center, Boston, noted in a second editorial.
“Cognitive decline often accompanies aging, and the prevalence of dementia increases rapidly after age 70 years,” they said.
The data on whether older clinicians pose a risk to patients is limited and somewhat mixed. An analysis of 736,537 Medicare hospitalizations found no association between physician age and 30-day patient mortality among physicians 60 years or older with more than 201 admissions per year, but higher mortality among older physicians with lower volumes.
A meta-analysis of 62 studies showed that “older physicians have less factual knowledge, are less likely to adhere to appropriate standards of care, and may also have poorer patient outcomes.”
The new Yale data, meanwhile, suggests that “approximately 13% [18 of 141] of physicians and other clinicians older than 70 years should not be practicing independently,” Dr. Armstrong and Dr. Reynolds said in their editorial.
There is support for screening efforts. “As a profession that deals with human life, medical practitioners must obviously have the cognitive capacity to safely practice medicine. I applaud the approach taken by Yale New Haven Hospital in that cognitive abilities themselves, and not simply funds of knowledge, are assessed,” said Richard J. Caselli, MD, professor of neurology at the Mayo Clinic Arizona, Scottsdale, and a leader of the Alzheimer’s disease program there.
However, it’s not hard to imagine highly competent but older physicians taking umbrage at cognitive screening, and there’s been pushback. Stanford was considering a Yale-like approach but opted instead for peer review after opposition. Objections from the Utah Medical Association led Utah to enact a law banning age-based physician screening. In 2015, the American Medical Association issued a report calling for the development of guidelines and standards for assessing competency in aging physicians, but the AMA House of Delegates shelved it pending further study.
There are concerns about age discrimination, discounting the accumulated wisdom of long-practicing physicians, and misclassifying competent physicians, particularly those who provide quality care in rural and other underserved areas. Indeed, 8 of 14 clinicians who screened positive at Yale and underwent more extensive testing were allowed to recredential, “suggesting that the false-positive screening rate could be as high as 57%,” Dr. Armstrong and Dr. Reynolds noted.
The consensus seems to be that there probably is a need for some sort of screening, but it must be both sound and fair. Rather than a piecemeal institutional approach, perhaps there is “an important opportunity for other groups, including specialty boards and state licensing boards” to standardize the process, they said.
Among other things, assessments could focus less on test scores and more on the practice of medicine. For instance, fine motor skill/motor planning assessments for surgeons, and intermediate results could trigger a more extensive assessment of actual clinical performance, perhaps even direct observation, Dr. Saver said in his editorial.
As far as clinical performance goes, none of the 18 clinicians at Yale had previous performance problems. “Was this a failure of the system to report impaired physicians or were these physicians compensating sufficiently to avoid detection?” In either case, “cognitive testing should be a red flag that triggers other clinical assessments,” said Carl I. Cohen, MD, professor and director of the division of geriatric psychiatry at the State University of New York, Brooklyn.
The original plan at Yale was for neurologic and ophthalmologic examinations beginning at age 70, but ultimately it was decided to go with a battery of 16 tests to assess visual scanning and psychomotor efficiency, processing speed under pressure, concentration, and working memory, among other things. Testing takes about 50-90 minutes, and is graded by single neuropsychologist to ensure consistency. Results were compared with normative scores from both older and younger clinicians.
To prevent clinicians from preparing for it, Yale isn’t releasing its test battery.
Suboptimal performance triggered additional evaluations, including in-depth assessment of intellectual, memory, and executive function. Final reviews and recommendations were made by a committee that included a geriatrician, the clinician’s section or department chair, and current and past chief medical officers.
Among the 18 providers who demonstrated deficits impairing their ability to practice medicine, 5 were 70-74 years old; 4 were 75-79; and 9 were 80 years or older. Minor abnormalities were found in 34 other candidates (24.1%); they were allowed to recredential but were scheduled for rescreening at 1-year intervals, instead of every 2 years.
The mean age among the 141 screened clinicians was 74.3 years and ranged from 69 to 92 years; 86% were men. Applicants included 125 physicians (88.7%) as well as 5 advanced practice registered nurses; 4 dentists; 3 psychologists; 2 podiatrists; 1 physician associate; and 1 midwife.
The authors had no relevant disclosures.
SOURCE: Cooney L et al. JAMA. 2020 Jan 14;323(2):179-80.
Cognitive screening of 141 clinicians 70 years or older at Yale New Haven (Conn.) Hospital identified 18 with cognitive deficits likely to impair their ability to practice medicine. Six retired and 12 agreed to limit their practice to closely proctored environments, according to a report in JAMA.
It was part of a program to screen all practitioners 70 years or older who apply for reappointment to the medical staff, and every 2 years thereafter, due to “concerns about the potentially compromised ability of older clinicians,” said the authors, Yale rheumatologist and geriatrician Leo M. Cooney Jr., MD, and Thomas Balcezak, MD, Yale New Haven’s chief medical officer.
Yale is not alone. Intermountain Healthcare, Stanford Hospitals and Clinics, Scripps Health Care, Penn Medicine, and the University of California, San Diego, are among the institutions with similar programs.
The move is being driven by the aging of the medical community. About 15% of U.S. physicians are over 65 years old, a tripling from 23,000 in 1980 to 73,000 in 2012-2016, and the number is growing, according to an editorial by Jeffrey L. Saver, MD, professor of neurology and senior associate vice president of neurology at the University of California, Los Angeles.
Given the trend, “it is not surprising that the issue of screening aging physicians for cognitive deficits has gained attention over the last decade,” Katrina Armstrong, MD, chair of the department of medicine at Massachusetts General Hospital, Boston, and Eileen E. Reynolds, MD, associate professor of medicine at Beth Israel Deaconess Medical Center, Boston, noted in a second editorial.
“Cognitive decline often accompanies aging, and the prevalence of dementia increases rapidly after age 70 years,” they said.
The data on whether older clinicians pose a risk to patients is limited and somewhat mixed. An analysis of 736,537 Medicare hospitalizations found no association between physician age and 30-day patient mortality among physicians 60 years or older with more than 201 admissions per year, but higher mortality among older physicians with lower volumes.
A meta-analysis of 62 studies showed that “older physicians have less factual knowledge, are less likely to adhere to appropriate standards of care, and may also have poorer patient outcomes.”
The new Yale data, meanwhile, suggests that “approximately 13% [18 of 141] of physicians and other clinicians older than 70 years should not be practicing independently,” Dr. Armstrong and Dr. Reynolds said in their editorial.
There is support for screening efforts. “As a profession that deals with human life, medical practitioners must obviously have the cognitive capacity to safely practice medicine. I applaud the approach taken by Yale New Haven Hospital in that cognitive abilities themselves, and not simply funds of knowledge, are assessed,” said Richard J. Caselli, MD, professor of neurology at the Mayo Clinic Arizona, Scottsdale, and a leader of the Alzheimer’s disease program there.
However, it’s not hard to imagine highly competent but older physicians taking umbrage at cognitive screening, and there’s been pushback. Stanford was considering a Yale-like approach but opted instead for peer review after opposition. Objections from the Utah Medical Association led Utah to enact a law banning age-based physician screening. In 2015, the American Medical Association issued a report calling for the development of guidelines and standards for assessing competency in aging physicians, but the AMA House of Delegates shelved it pending further study.
There are concerns about age discrimination, discounting the accumulated wisdom of long-practicing physicians, and misclassifying competent physicians, particularly those who provide quality care in rural and other underserved areas. Indeed, 8 of 14 clinicians who screened positive at Yale and underwent more extensive testing were allowed to recredential, “suggesting that the false-positive screening rate could be as high as 57%,” Dr. Armstrong and Dr. Reynolds noted.
The consensus seems to be that there probably is a need for some sort of screening, but it must be both sound and fair. Rather than a piecemeal institutional approach, perhaps there is “an important opportunity for other groups, including specialty boards and state licensing boards” to standardize the process, they said.
Among other things, assessments could focus less on test scores and more on the practice of medicine. For instance, fine motor skill/motor planning assessments for surgeons, and intermediate results could trigger a more extensive assessment of actual clinical performance, perhaps even direct observation, Dr. Saver said in his editorial.
As far as clinical performance goes, none of the 18 clinicians at Yale had previous performance problems. “Was this a failure of the system to report impaired physicians or were these physicians compensating sufficiently to avoid detection?” In either case, “cognitive testing should be a red flag that triggers other clinical assessments,” said Carl I. Cohen, MD, professor and director of the division of geriatric psychiatry at the State University of New York, Brooklyn.
The original plan at Yale was for neurologic and ophthalmologic examinations beginning at age 70, but ultimately it was decided to go with a battery of 16 tests to assess visual scanning and psychomotor efficiency, processing speed under pressure, concentration, and working memory, among other things. Testing takes about 50-90 minutes, and is graded by single neuropsychologist to ensure consistency. Results were compared with normative scores from both older and younger clinicians.
To prevent clinicians from preparing for it, Yale isn’t releasing its test battery.
Suboptimal performance triggered additional evaluations, including in-depth assessment of intellectual, memory, and executive function. Final reviews and recommendations were made by a committee that included a geriatrician, the clinician’s section or department chair, and current and past chief medical officers.
Among the 18 providers who demonstrated deficits impairing their ability to practice medicine, 5 were 70-74 years old; 4 were 75-79; and 9 were 80 years or older. Minor abnormalities were found in 34 other candidates (24.1%); they were allowed to recredential but were scheduled for rescreening at 1-year intervals, instead of every 2 years.
The mean age among the 141 screened clinicians was 74.3 years and ranged from 69 to 92 years; 86% were men. Applicants included 125 physicians (88.7%) as well as 5 advanced practice registered nurses; 4 dentists; 3 psychologists; 2 podiatrists; 1 physician associate; and 1 midwife.
The authors had no relevant disclosures.
SOURCE: Cooney L et al. JAMA. 2020 Jan 14;323(2):179-80.
Cognitive screening of 141 clinicians 70 years or older at Yale New Haven (Conn.) Hospital identified 18 with cognitive deficits likely to impair their ability to practice medicine. Six retired and 12 agreed to limit their practice to closely proctored environments, according to a report in JAMA.
It was part of a program to screen all practitioners 70 years or older who apply for reappointment to the medical staff, and every 2 years thereafter, due to “concerns about the potentially compromised ability of older clinicians,” said the authors, Yale rheumatologist and geriatrician Leo M. Cooney Jr., MD, and Thomas Balcezak, MD, Yale New Haven’s chief medical officer.
Yale is not alone. Intermountain Healthcare, Stanford Hospitals and Clinics, Scripps Health Care, Penn Medicine, and the University of California, San Diego, are among the institutions with similar programs.
The move is being driven by the aging of the medical community. About 15% of U.S. physicians are over 65 years old, a tripling from 23,000 in 1980 to 73,000 in 2012-2016, and the number is growing, according to an editorial by Jeffrey L. Saver, MD, professor of neurology and senior associate vice president of neurology at the University of California, Los Angeles.
Given the trend, “it is not surprising that the issue of screening aging physicians for cognitive deficits has gained attention over the last decade,” Katrina Armstrong, MD, chair of the department of medicine at Massachusetts General Hospital, Boston, and Eileen E. Reynolds, MD, associate professor of medicine at Beth Israel Deaconess Medical Center, Boston, noted in a second editorial.
“Cognitive decline often accompanies aging, and the prevalence of dementia increases rapidly after age 70 years,” they said.
The data on whether older clinicians pose a risk to patients is limited and somewhat mixed. An analysis of 736,537 Medicare hospitalizations found no association between physician age and 30-day patient mortality among physicians 60 years or older with more than 201 admissions per year, but higher mortality among older physicians with lower volumes.
A meta-analysis of 62 studies showed that “older physicians have less factual knowledge, are less likely to adhere to appropriate standards of care, and may also have poorer patient outcomes.”
The new Yale data, meanwhile, suggests that “approximately 13% [18 of 141] of physicians and other clinicians older than 70 years should not be practicing independently,” Dr. Armstrong and Dr. Reynolds said in their editorial.
There is support for screening efforts. “As a profession that deals with human life, medical practitioners must obviously have the cognitive capacity to safely practice medicine. I applaud the approach taken by Yale New Haven Hospital in that cognitive abilities themselves, and not simply funds of knowledge, are assessed,” said Richard J. Caselli, MD, professor of neurology at the Mayo Clinic Arizona, Scottsdale, and a leader of the Alzheimer’s disease program there.
However, it’s not hard to imagine highly competent but older physicians taking umbrage at cognitive screening, and there’s been pushback. Stanford was considering a Yale-like approach but opted instead for peer review after opposition. Objections from the Utah Medical Association led Utah to enact a law banning age-based physician screening. In 2015, the American Medical Association issued a report calling for the development of guidelines and standards for assessing competency in aging physicians, but the AMA House of Delegates shelved it pending further study.
There are concerns about age discrimination, discounting the accumulated wisdom of long-practicing physicians, and misclassifying competent physicians, particularly those who provide quality care in rural and other underserved areas. Indeed, 8 of 14 clinicians who screened positive at Yale and underwent more extensive testing were allowed to recredential, “suggesting that the false-positive screening rate could be as high as 57%,” Dr. Armstrong and Dr. Reynolds noted.
The consensus seems to be that there probably is a need for some sort of screening, but it must be both sound and fair. Rather than a piecemeal institutional approach, perhaps there is “an important opportunity for other groups, including specialty boards and state licensing boards” to standardize the process, they said.
Among other things, assessments could focus less on test scores and more on the practice of medicine. For instance, fine motor skill/motor planning assessments for surgeons, and intermediate results could trigger a more extensive assessment of actual clinical performance, perhaps even direct observation, Dr. Saver said in his editorial.
As far as clinical performance goes, none of the 18 clinicians at Yale had previous performance problems. “Was this a failure of the system to report impaired physicians or were these physicians compensating sufficiently to avoid detection?” In either case, “cognitive testing should be a red flag that triggers other clinical assessments,” said Carl I. Cohen, MD, professor and director of the division of geriatric psychiatry at the State University of New York, Brooklyn.
The original plan at Yale was for neurologic and ophthalmologic examinations beginning at age 70, but ultimately it was decided to go with a battery of 16 tests to assess visual scanning and psychomotor efficiency, processing speed under pressure, concentration, and working memory, among other things. Testing takes about 50-90 minutes, and is graded by single neuropsychologist to ensure consistency. Results were compared with normative scores from both older and younger clinicians.
To prevent clinicians from preparing for it, Yale isn’t releasing its test battery.
Suboptimal performance triggered additional evaluations, including in-depth assessment of intellectual, memory, and executive function. Final reviews and recommendations were made by a committee that included a geriatrician, the clinician’s section or department chair, and current and past chief medical officers.
Among the 18 providers who demonstrated deficits impairing their ability to practice medicine, 5 were 70-74 years old; 4 were 75-79; and 9 were 80 years or older. Minor abnormalities were found in 34 other candidates (24.1%); they were allowed to recredential but were scheduled for rescreening at 1-year intervals, instead of every 2 years.
The mean age among the 141 screened clinicians was 74.3 years and ranged from 69 to 92 years; 86% were men. Applicants included 125 physicians (88.7%) as well as 5 advanced practice registered nurses; 4 dentists; 3 psychologists; 2 podiatrists; 1 physician associate; and 1 midwife.
The authors had no relevant disclosures.
SOURCE: Cooney L et al. JAMA. 2020 Jan 14;323(2):179-80.
FROM JAMA
Sarcopenia associated with increased cardiometabolic risk
LOS ANGELES –
“Loss of lean body mass and function with aging decreases the amount of metabolically active tissue, which can lead to insulin resistance,” Elena Volpi, MD, said at the World Congress on Insulin Resistance, Diabetes and Cardiovascular Disease. “Insulin resistance reduces muscle protein anabolism and accelerates sarcopenia, perpetuating a vicious cycle.”
Sarcopenia, the involuntary loss of muscle mass and function that occurs with aging, is an ICD-10 codable condition that can be diagnosed by measuring muscle strength and quality, said Dr. Volpi, director of the Sealy Center on Aging at the University of Texas Medical Branch at Galveston. In the Health, Aging and Body Composition Study (Health ABC), researchers followed 2,292 relatively healthy adults aged 70-79 years for an average of 4.9 years (J Gerontol A Biol Sci Med. 2006;61[1]:72-7). The researchers used isokinetic dynamometry to measure knee extension strength, isometric dynamometry to measure grip strength, CT scan to measure thigh muscle area, and dual X-ray absorptiometry to determine leg and arm lean soft-tissue mass. “Those individuals who started with the highest levels of muscle strength had the greatest survival, while those who had the lowest levels of muscle strength died earlier,” said Dr. Volpi, who was not affiliated with the study. “That was true for both men and women.”
More recently, researchers conducted a pooled analysis of nine cohort studies involving 34,485 community-dwelling older individuals who were tested with gait speed and followed for 6-21 years (JAMA. 2011;305[1]:50-8). They found that a higher gait speed was associated with higher survival at 5 and 10 years (P less than .001). “Muscle mass also appears to be associated in part with mortality and survival, although the association is not as strong as measures of strength and gait speed,” Dr. Volpi said.
Data from the 2009 Korea National Health and Nutrition Examination Survey of 1,537 participants, aged 65 years and older, found that sarcopenia is independently associated with cardiovascular disease (PLoS One. 2013 Mar 22. doi: 10.1371/journal.pone.0060119). Most of the risk factors for cardiovascular disease – such as age, waist circumference, body mass index, fasting plasma glucose, and total cholesterol – showed significant negative correlations with the ratio between appendicular skeletal muscle mass and body weight. Multiple logistic regression analysis demonstrated that sarcopenia was associated with cardiovascular disease, independent of other well-documented risk factors, renal function, and medications (odds ratio, 1.77; P = .025).
In addition, data from the British Regional Heart Study, which followed 4,252 older men for a mean of 11.3 years, found an association of sarcopenia and adiposity with cardiovascular mortality and all-cause mortality (J Am Geriatr Soc. 2014;62[2]:253-60). Specifically, all-cause mortality risk was significantly greater in men in the sarcopenic and obese groups (HRs, 1.41 and 1.21, respectively), compared with those in the optimal reference group, with the highest risk in sarcopenic obese individuals (HR, 1.72) after adjustment for lifestyle characteristics.
“Diabetes also accelerates loss of lean body mass in older adults,” added Dr. Volpi. “Data from the Health ABC study showed that individuals who did not have diabetes at the beginning of the 6-year observation period ... lost the least amount of muscle, compared with those who had undiagnosed or already diagnosed diabetes.”
The precise way in which sarcopenia is linked to metabolic disease remains elusive, she continued, but current evidence suggests that sarcopenia is characterized by a reduction in the protein synthetic response to metabolic stimulation by amino acids, exercise, and insulin in skeletal muscle. “This reduction in the anabolic response to protein synthesis is called anabolic resistance of aging, and it is mediated by reduced acute activation of mTORC1 [mTOR complex 1] signaling,” Dr. Volpi said. “There’s another step upstream of the mTORC1, in which the amino acids and insulin have to cross the blood-muscle barrier. Amino acids need to be transported into the muscle actively, like glucose. That is an important unexplored area that may contribute to sarcopenia.”
Dr. Volpi went on to note that endothelial dysfunction underlies muscle anabolic resistance and cardiovascular risk and is likely to be a fundamental cause of both problems. Recent studies have shown that increased levels of physical activity improve endothelial function, enhance insulin sensitivity and anabolic sensitivity to nutrients, and reduce cardiovascular risk.
For example, in a cohort of 45 nonfrail older adults with a mean age of 72 years, Dr. Volpi and colleagues carried out a phase 1, double-blind, placebo-controlled, randomized clinical trial to determine if chronic essential amino acid supplementation, aerobic exercise training, or a combination of the two interventions could improve muscle mass and function by stimulating muscle protein synthesis over the course of 24 weeks (J Gerontol A Biol Sci Med Sci. 2019;74[10]:1598-604). “We found that exercise supervised three times per week on a treadmill for 6 months improved physical function in both groups randomized to exercise,” Dr. Volpi said. “Disappointingly, there was no change in total lean mass with any of the interventions. There was a decrease in fat mass with exercise alone, and no change with exercise and amino acids. [Of note is that] the individuals who were randomized to the amino acids plus exercise group had a significant increase in leg strength, whereas the others did not.”
Preliminary findings from ongoing work by Dr. Volpi and colleagues suggest that, in diabetes, muscle protein synthesis and blood flow really “are not different in response to insulin in healthy older adults and diabetic older adults because they don’t change at all. However, we did find alterations in amino acid trafficking in diabetes. We found that older individuals with type 2 diabetes had a reduction of amino acid transport and a higher intracellular amino acid concentration, compared with age-matched, healthier individuals. The intracellular amino acid clearance improved in the healthy, nondiabetic older adults with hyperinsulinemia, whereas it did not change in diabetic older adults. As a result, the net muscle protein balance improved a little in the nondiabetic patients, but did not change in the diabetic patients.”
The researchers are evaluating older patients with type 2 diabetes to see whether there are alterations in vascular reactivity and protein synthesis and whether those can be overcome by resistance-exercise training. “Preliminary results show that flow-mediated dilation can actually increase in an older diabetic patient with resistance exercise training three times a week for 3 months,” she said. “Exercise can improve both endothelial dysfunction and sarcopenia and therefore improve physical function and reduce cardiovascular risk.”
Dr. Volpi reported having no relevant disclosures.
LOS ANGELES –
“Loss of lean body mass and function with aging decreases the amount of metabolically active tissue, which can lead to insulin resistance,” Elena Volpi, MD, said at the World Congress on Insulin Resistance, Diabetes and Cardiovascular Disease. “Insulin resistance reduces muscle protein anabolism and accelerates sarcopenia, perpetuating a vicious cycle.”
Sarcopenia, the involuntary loss of muscle mass and function that occurs with aging, is an ICD-10 codable condition that can be diagnosed by measuring muscle strength and quality, said Dr. Volpi, director of the Sealy Center on Aging at the University of Texas Medical Branch at Galveston. In the Health, Aging and Body Composition Study (Health ABC), researchers followed 2,292 relatively healthy adults aged 70-79 years for an average of 4.9 years (J Gerontol A Biol Sci Med. 2006;61[1]:72-7). The researchers used isokinetic dynamometry to measure knee extension strength, isometric dynamometry to measure grip strength, CT scan to measure thigh muscle area, and dual X-ray absorptiometry to determine leg and arm lean soft-tissue mass. “Those individuals who started with the highest levels of muscle strength had the greatest survival, while those who had the lowest levels of muscle strength died earlier,” said Dr. Volpi, who was not affiliated with the study. “That was true for both men and women.”
More recently, researchers conducted a pooled analysis of nine cohort studies involving 34,485 community-dwelling older individuals who were tested with gait speed and followed for 6-21 years (JAMA. 2011;305[1]:50-8). They found that a higher gait speed was associated with higher survival at 5 and 10 years (P less than .001). “Muscle mass also appears to be associated in part with mortality and survival, although the association is not as strong as measures of strength and gait speed,” Dr. Volpi said.
Data from the 2009 Korea National Health and Nutrition Examination Survey of 1,537 participants, aged 65 years and older, found that sarcopenia is independently associated with cardiovascular disease (PLoS One. 2013 Mar 22. doi: 10.1371/journal.pone.0060119). Most of the risk factors for cardiovascular disease – such as age, waist circumference, body mass index, fasting plasma glucose, and total cholesterol – showed significant negative correlations with the ratio between appendicular skeletal muscle mass and body weight. Multiple logistic regression analysis demonstrated that sarcopenia was associated with cardiovascular disease, independent of other well-documented risk factors, renal function, and medications (odds ratio, 1.77; P = .025).
In addition, data from the British Regional Heart Study, which followed 4,252 older men for a mean of 11.3 years, found an association of sarcopenia and adiposity with cardiovascular mortality and all-cause mortality (J Am Geriatr Soc. 2014;62[2]:253-60). Specifically, all-cause mortality risk was significantly greater in men in the sarcopenic and obese groups (HRs, 1.41 and 1.21, respectively), compared with those in the optimal reference group, with the highest risk in sarcopenic obese individuals (HR, 1.72) after adjustment for lifestyle characteristics.
“Diabetes also accelerates loss of lean body mass in older adults,” added Dr. Volpi. “Data from the Health ABC study showed that individuals who did not have diabetes at the beginning of the 6-year observation period ... lost the least amount of muscle, compared with those who had undiagnosed or already diagnosed diabetes.”
The precise way in which sarcopenia is linked to metabolic disease remains elusive, she continued, but current evidence suggests that sarcopenia is characterized by a reduction in the protein synthetic response to metabolic stimulation by amino acids, exercise, and insulin in skeletal muscle. “This reduction in the anabolic response to protein synthesis is called anabolic resistance of aging, and it is mediated by reduced acute activation of mTORC1 [mTOR complex 1] signaling,” Dr. Volpi said. “There’s another step upstream of the mTORC1, in which the amino acids and insulin have to cross the blood-muscle barrier. Amino acids need to be transported into the muscle actively, like glucose. That is an important unexplored area that may contribute to sarcopenia.”
Dr. Volpi went on to note that endothelial dysfunction underlies muscle anabolic resistance and cardiovascular risk and is likely to be a fundamental cause of both problems. Recent studies have shown that increased levels of physical activity improve endothelial function, enhance insulin sensitivity and anabolic sensitivity to nutrients, and reduce cardiovascular risk.
For example, in a cohort of 45 nonfrail older adults with a mean age of 72 years, Dr. Volpi and colleagues carried out a phase 1, double-blind, placebo-controlled, randomized clinical trial to determine if chronic essential amino acid supplementation, aerobic exercise training, or a combination of the two interventions could improve muscle mass and function by stimulating muscle protein synthesis over the course of 24 weeks (J Gerontol A Biol Sci Med Sci. 2019;74[10]:1598-604). “We found that exercise supervised three times per week on a treadmill for 6 months improved physical function in both groups randomized to exercise,” Dr. Volpi said. “Disappointingly, there was no change in total lean mass with any of the interventions. There was a decrease in fat mass with exercise alone, and no change with exercise and amino acids. [Of note is that] the individuals who were randomized to the amino acids plus exercise group had a significant increase in leg strength, whereas the others did not.”
Preliminary findings from ongoing work by Dr. Volpi and colleagues suggest that, in diabetes, muscle protein synthesis and blood flow really “are not different in response to insulin in healthy older adults and diabetic older adults because they don’t change at all. However, we did find alterations in amino acid trafficking in diabetes. We found that older individuals with type 2 diabetes had a reduction of amino acid transport and a higher intracellular amino acid concentration, compared with age-matched, healthier individuals. The intracellular amino acid clearance improved in the healthy, nondiabetic older adults with hyperinsulinemia, whereas it did not change in diabetic older adults. As a result, the net muscle protein balance improved a little in the nondiabetic patients, but did not change in the diabetic patients.”
The researchers are evaluating older patients with type 2 diabetes to see whether there are alterations in vascular reactivity and protein synthesis and whether those can be overcome by resistance-exercise training. “Preliminary results show that flow-mediated dilation can actually increase in an older diabetic patient with resistance exercise training three times a week for 3 months,” she said. “Exercise can improve both endothelial dysfunction and sarcopenia and therefore improve physical function and reduce cardiovascular risk.”
Dr. Volpi reported having no relevant disclosures.
LOS ANGELES –
“Loss of lean body mass and function with aging decreases the amount of metabolically active tissue, which can lead to insulin resistance,” Elena Volpi, MD, said at the World Congress on Insulin Resistance, Diabetes and Cardiovascular Disease. “Insulin resistance reduces muscle protein anabolism and accelerates sarcopenia, perpetuating a vicious cycle.”
Sarcopenia, the involuntary loss of muscle mass and function that occurs with aging, is an ICD-10 codable condition that can be diagnosed by measuring muscle strength and quality, said Dr. Volpi, director of the Sealy Center on Aging at the University of Texas Medical Branch at Galveston. In the Health, Aging and Body Composition Study (Health ABC), researchers followed 2,292 relatively healthy adults aged 70-79 years for an average of 4.9 years (J Gerontol A Biol Sci Med. 2006;61[1]:72-7). The researchers used isokinetic dynamometry to measure knee extension strength, isometric dynamometry to measure grip strength, CT scan to measure thigh muscle area, and dual X-ray absorptiometry to determine leg and arm lean soft-tissue mass. “Those individuals who started with the highest levels of muscle strength had the greatest survival, while those who had the lowest levels of muscle strength died earlier,” said Dr. Volpi, who was not affiliated with the study. “That was true for both men and women.”
More recently, researchers conducted a pooled analysis of nine cohort studies involving 34,485 community-dwelling older individuals who were tested with gait speed and followed for 6-21 years (JAMA. 2011;305[1]:50-8). They found that a higher gait speed was associated with higher survival at 5 and 10 years (P less than .001). “Muscle mass also appears to be associated in part with mortality and survival, although the association is not as strong as measures of strength and gait speed,” Dr. Volpi said.
Data from the 2009 Korea National Health and Nutrition Examination Survey of 1,537 participants, aged 65 years and older, found that sarcopenia is independently associated with cardiovascular disease (PLoS One. 2013 Mar 22. doi: 10.1371/journal.pone.0060119). Most of the risk factors for cardiovascular disease – such as age, waist circumference, body mass index, fasting plasma glucose, and total cholesterol – showed significant negative correlations with the ratio between appendicular skeletal muscle mass and body weight. Multiple logistic regression analysis demonstrated that sarcopenia was associated with cardiovascular disease, independent of other well-documented risk factors, renal function, and medications (odds ratio, 1.77; P = .025).
In addition, data from the British Regional Heart Study, which followed 4,252 older men for a mean of 11.3 years, found an association of sarcopenia and adiposity with cardiovascular mortality and all-cause mortality (J Am Geriatr Soc. 2014;62[2]:253-60). Specifically, all-cause mortality risk was significantly greater in men in the sarcopenic and obese groups (HRs, 1.41 and 1.21, respectively), compared with those in the optimal reference group, with the highest risk in sarcopenic obese individuals (HR, 1.72) after adjustment for lifestyle characteristics.
“Diabetes also accelerates loss of lean body mass in older adults,” added Dr. Volpi. “Data from the Health ABC study showed that individuals who did not have diabetes at the beginning of the 6-year observation period ... lost the least amount of muscle, compared with those who had undiagnosed or already diagnosed diabetes.”
The precise way in which sarcopenia is linked to metabolic disease remains elusive, she continued, but current evidence suggests that sarcopenia is characterized by a reduction in the protein synthetic response to metabolic stimulation by amino acids, exercise, and insulin in skeletal muscle. “This reduction in the anabolic response to protein synthesis is called anabolic resistance of aging, and it is mediated by reduced acute activation of mTORC1 [mTOR complex 1] signaling,” Dr. Volpi said. “There’s another step upstream of the mTORC1, in which the amino acids and insulin have to cross the blood-muscle barrier. Amino acids need to be transported into the muscle actively, like glucose. That is an important unexplored area that may contribute to sarcopenia.”
Dr. Volpi went on to note that endothelial dysfunction underlies muscle anabolic resistance and cardiovascular risk and is likely to be a fundamental cause of both problems. Recent studies have shown that increased levels of physical activity improve endothelial function, enhance insulin sensitivity and anabolic sensitivity to nutrients, and reduce cardiovascular risk.
For example, in a cohort of 45 nonfrail older adults with a mean age of 72 years, Dr. Volpi and colleagues carried out a phase 1, double-blind, placebo-controlled, randomized clinical trial to determine if chronic essential amino acid supplementation, aerobic exercise training, or a combination of the two interventions could improve muscle mass and function by stimulating muscle protein synthesis over the course of 24 weeks (J Gerontol A Biol Sci Med Sci. 2019;74[10]:1598-604). “We found that exercise supervised three times per week on a treadmill for 6 months improved physical function in both groups randomized to exercise,” Dr. Volpi said. “Disappointingly, there was no change in total lean mass with any of the interventions. There was a decrease in fat mass with exercise alone, and no change with exercise and amino acids. [Of note is that] the individuals who were randomized to the amino acids plus exercise group had a significant increase in leg strength, whereas the others did not.”
Preliminary findings from ongoing work by Dr. Volpi and colleagues suggest that, in diabetes, muscle protein synthesis and blood flow really “are not different in response to insulin in healthy older adults and diabetic older adults because they don’t change at all. However, we did find alterations in amino acid trafficking in diabetes. We found that older individuals with type 2 diabetes had a reduction of amino acid transport and a higher intracellular amino acid concentration, compared with age-matched, healthier individuals. The intracellular amino acid clearance improved in the healthy, nondiabetic older adults with hyperinsulinemia, whereas it did not change in diabetic older adults. As a result, the net muscle protein balance improved a little in the nondiabetic patients, but did not change in the diabetic patients.”
The researchers are evaluating older patients with type 2 diabetes to see whether there are alterations in vascular reactivity and protein synthesis and whether those can be overcome by resistance-exercise training. “Preliminary results show that flow-mediated dilation can actually increase in an older diabetic patient with resistance exercise training three times a week for 3 months,” she said. “Exercise can improve both endothelial dysfunction and sarcopenia and therefore improve physical function and reduce cardiovascular risk.”
Dr. Volpi reported having no relevant disclosures.
EXPERT ANALYSIS FROM WCIRDC 2019
Hip fracture patients with dementia benefit from increased rehab intensity
according to a recent Japanese study in the Archives of Physical Medicine and Rehabilitation.
Looking at 43,506 patients cared for at 1,053 hospitals, Kazuaki Uda, MPH, and colleagues of the University of Tokyo found that scores on the Barthel Index, a measure of functional status, climbed significantly as the frequency and duration of postoperative rehabilitation increased. There was also a statistically significant, but small, association with improved functional status and early initiation of rehabilitation.
“Our results suggest that additional days of rehabilitation or an additional 20 minutes for each daily rehabilitation session in acute-care hospitals may provide better functional outcomes for patients with dementia,” concluded Mr. Uda and coinvestigators.
The Barthel Index (BI) measures independence in performing 10 activities of daily living (ADLs), including feeding, bathing, grooming, and dressing; bowel, bladder, and toileting; and transfers, mobility, and stair use. Each ADL is rate 0, 5, 10, or, for some, 15 points, and higher scores indicate more independence.
Compared with patients who received 3 days or fewer of rehabilitation weekly, patients receiving 3-4 days of rehabilitation saw an improvement of 2.62 on the BI. For those receiving 4-5 days, 5-6 days, and 6 or more days of rehabilitation, BI scores were higher by 5.83, 7.56, and 9.16, respectively. The results were statistically significant for all but the 3-4 day rehabilitation group.
Similarly, patients who received longer periods of rehabilitation saw more improvement in functional status. Compared with those who received 20-39 minutes per day of rehabilitation, those who received 40-59 minutes of therapy saw an increase of 4.37 on the BI, and those receiving an hour or more of therapy saw BI scores rise by 6.60 – both significant increases.
These results included a multivariable analysis that accounted for a number of patient characteristics such as comorbidities and body mass index, as well as fracture, fixation, and anesthesia type, and the interval from injury to surgery.
Representing the data in another way, the investigators found that “each increase in the average units of rehabilitation (units per day) was associated with a 5.46 increase in the BI.”
This retrospective cohort study, when placed in the context of previous work, suggests that “patients with cognitive impairment may benefit from rehabilitation for functional gains after hip fracture surgery in both acute and postacute settings,” the investigators wrote. They noted, however, that patients with dementia have often been excluded from larger outcome studies of hip fracture rehabilitation.
Patients in this study had a median 21-day inpatient stay after admission for their hip fracture, so much of the rehabilitation included as inpatient care in the Japanese schema would be delivered in the outpatient setting in the United States, where the mean inpatient length of stay after hip fracture is about 5 days.
Patients aged 65 years and older were included in the study if they had a prefracture diagnosis of dementia and sustained a hip fracture that was surgically repaired. Patients with multiple fracture sites, those with incomplete data, and those who didn’t undergo surgery or died in the hospital were excluded from the study. Almost two-thirds of patients (65.7%) were aged 85 years or older, and about a third (36.6%) were living in nursing facilities at the time of fracture. About 60% of patients were assessed as having mild dementia – a classification requiring little assistance with ADLs – before admission.
The authors noted that their study broke out timing, duration, and frequency of rehabilitation separately, unlike some previous work. They posited that longer or more frequent rounds of rehabilitation may be particularly effective in patients with dementia, who may face some communication barriers and require reteaching.
The study was unrandomized by design, and unmeasured confounders may have affected the results, they noted. Also, the study wasn’t designed to detect whether patient factors such as premorbid functional status, level of dementia, or living situation affected the timing, duration, and intensity of rehabilitation they were provided. The investigators recommended randomized studies to validate the effect of early, intensive rehabilitation for hip fracture surgery in patients with dementia.
The study was funded by the Japanese Ministry of Health, Labor, and Welfare. The authors reported that they have no relevant conflicts of interest.
SOURCE: Uda K et al. Arch Phys Med Rehabil. 2019;100:2301-7.
according to a recent Japanese study in the Archives of Physical Medicine and Rehabilitation.
Looking at 43,506 patients cared for at 1,053 hospitals, Kazuaki Uda, MPH, and colleagues of the University of Tokyo found that scores on the Barthel Index, a measure of functional status, climbed significantly as the frequency and duration of postoperative rehabilitation increased. There was also a statistically significant, but small, association with improved functional status and early initiation of rehabilitation.
“Our results suggest that additional days of rehabilitation or an additional 20 minutes for each daily rehabilitation session in acute-care hospitals may provide better functional outcomes for patients with dementia,” concluded Mr. Uda and coinvestigators.
The Barthel Index (BI) measures independence in performing 10 activities of daily living (ADLs), including feeding, bathing, grooming, and dressing; bowel, bladder, and toileting; and transfers, mobility, and stair use. Each ADL is rate 0, 5, 10, or, for some, 15 points, and higher scores indicate more independence.
Compared with patients who received 3 days or fewer of rehabilitation weekly, patients receiving 3-4 days of rehabilitation saw an improvement of 2.62 on the BI. For those receiving 4-5 days, 5-6 days, and 6 or more days of rehabilitation, BI scores were higher by 5.83, 7.56, and 9.16, respectively. The results were statistically significant for all but the 3-4 day rehabilitation group.
Similarly, patients who received longer periods of rehabilitation saw more improvement in functional status. Compared with those who received 20-39 minutes per day of rehabilitation, those who received 40-59 minutes of therapy saw an increase of 4.37 on the BI, and those receiving an hour or more of therapy saw BI scores rise by 6.60 – both significant increases.
These results included a multivariable analysis that accounted for a number of patient characteristics such as comorbidities and body mass index, as well as fracture, fixation, and anesthesia type, and the interval from injury to surgery.
Representing the data in another way, the investigators found that “each increase in the average units of rehabilitation (units per day) was associated with a 5.46 increase in the BI.”
This retrospective cohort study, when placed in the context of previous work, suggests that “patients with cognitive impairment may benefit from rehabilitation for functional gains after hip fracture surgery in both acute and postacute settings,” the investigators wrote. They noted, however, that patients with dementia have often been excluded from larger outcome studies of hip fracture rehabilitation.
Patients in this study had a median 21-day inpatient stay after admission for their hip fracture, so much of the rehabilitation included as inpatient care in the Japanese schema would be delivered in the outpatient setting in the United States, where the mean inpatient length of stay after hip fracture is about 5 days.
Patients aged 65 years and older were included in the study if they had a prefracture diagnosis of dementia and sustained a hip fracture that was surgically repaired. Patients with multiple fracture sites, those with incomplete data, and those who didn’t undergo surgery or died in the hospital were excluded from the study. Almost two-thirds of patients (65.7%) were aged 85 years or older, and about a third (36.6%) were living in nursing facilities at the time of fracture. About 60% of patients were assessed as having mild dementia – a classification requiring little assistance with ADLs – before admission.
The authors noted that their study broke out timing, duration, and frequency of rehabilitation separately, unlike some previous work. They posited that longer or more frequent rounds of rehabilitation may be particularly effective in patients with dementia, who may face some communication barriers and require reteaching.
The study was unrandomized by design, and unmeasured confounders may have affected the results, they noted. Also, the study wasn’t designed to detect whether patient factors such as premorbid functional status, level of dementia, or living situation affected the timing, duration, and intensity of rehabilitation they were provided. The investigators recommended randomized studies to validate the effect of early, intensive rehabilitation for hip fracture surgery in patients with dementia.
The study was funded by the Japanese Ministry of Health, Labor, and Welfare. The authors reported that they have no relevant conflicts of interest.
SOURCE: Uda K et al. Arch Phys Med Rehabil. 2019;100:2301-7.
according to a recent Japanese study in the Archives of Physical Medicine and Rehabilitation.
Looking at 43,506 patients cared for at 1,053 hospitals, Kazuaki Uda, MPH, and colleagues of the University of Tokyo found that scores on the Barthel Index, a measure of functional status, climbed significantly as the frequency and duration of postoperative rehabilitation increased. There was also a statistically significant, but small, association with improved functional status and early initiation of rehabilitation.
“Our results suggest that additional days of rehabilitation or an additional 20 minutes for each daily rehabilitation session in acute-care hospitals may provide better functional outcomes for patients with dementia,” concluded Mr. Uda and coinvestigators.
The Barthel Index (BI) measures independence in performing 10 activities of daily living (ADLs), including feeding, bathing, grooming, and dressing; bowel, bladder, and toileting; and transfers, mobility, and stair use. Each ADL is rate 0, 5, 10, or, for some, 15 points, and higher scores indicate more independence.
Compared with patients who received 3 days or fewer of rehabilitation weekly, patients receiving 3-4 days of rehabilitation saw an improvement of 2.62 on the BI. For those receiving 4-5 days, 5-6 days, and 6 or more days of rehabilitation, BI scores were higher by 5.83, 7.56, and 9.16, respectively. The results were statistically significant for all but the 3-4 day rehabilitation group.
Similarly, patients who received longer periods of rehabilitation saw more improvement in functional status. Compared with those who received 20-39 minutes per day of rehabilitation, those who received 40-59 minutes of therapy saw an increase of 4.37 on the BI, and those receiving an hour or more of therapy saw BI scores rise by 6.60 – both significant increases.
These results included a multivariable analysis that accounted for a number of patient characteristics such as comorbidities and body mass index, as well as fracture, fixation, and anesthesia type, and the interval from injury to surgery.
Representing the data in another way, the investigators found that “each increase in the average units of rehabilitation (units per day) was associated with a 5.46 increase in the BI.”
This retrospective cohort study, when placed in the context of previous work, suggests that “patients with cognitive impairment may benefit from rehabilitation for functional gains after hip fracture surgery in both acute and postacute settings,” the investigators wrote. They noted, however, that patients with dementia have often been excluded from larger outcome studies of hip fracture rehabilitation.
Patients in this study had a median 21-day inpatient stay after admission for their hip fracture, so much of the rehabilitation included as inpatient care in the Japanese schema would be delivered in the outpatient setting in the United States, where the mean inpatient length of stay after hip fracture is about 5 days.
Patients aged 65 years and older were included in the study if they had a prefracture diagnosis of dementia and sustained a hip fracture that was surgically repaired. Patients with multiple fracture sites, those with incomplete data, and those who didn’t undergo surgery or died in the hospital were excluded from the study. Almost two-thirds of patients (65.7%) were aged 85 years or older, and about a third (36.6%) were living in nursing facilities at the time of fracture. About 60% of patients were assessed as having mild dementia – a classification requiring little assistance with ADLs – before admission.
The authors noted that their study broke out timing, duration, and frequency of rehabilitation separately, unlike some previous work. They posited that longer or more frequent rounds of rehabilitation may be particularly effective in patients with dementia, who may face some communication barriers and require reteaching.
The study was unrandomized by design, and unmeasured confounders may have affected the results, they noted. Also, the study wasn’t designed to detect whether patient factors such as premorbid functional status, level of dementia, or living situation affected the timing, duration, and intensity of rehabilitation they were provided. The investigators recommended randomized studies to validate the effect of early, intensive rehabilitation for hip fracture surgery in patients with dementia.
The study was funded by the Japanese Ministry of Health, Labor, and Welfare. The authors reported that they have no relevant conflicts of interest.
SOURCE: Uda K et al. Arch Phys Med Rehabil. 2019;100:2301-7.
REPORTING FROM THE ARCHIVES OF PHYSICAL MEDICINE AND REHABILITATION
Delayed hospital admission after hip fracture raises mortality risk
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
a retrospective, observational study suggests.
Among 867 elderly patients who underwent hip fracture surgery at a university hospital in China and who were available for follow-up, the proportion hospitalized on the day of injury was 25.4%, and the proportion hospitalized on days 1, 2, and 7 after injury were 54.7%, 66.3%, and 12.6%, respectively, reported Wei He, MD, of the Second Affiliated Hospital of Zhejiang University, Hangzhou, China, and colleagues in the World Journal of Emergency Medicine.
The mean time from admission to surgery was 5.2 days. Mortality rates at 1 year, 3 months, and 1 month after surgery were 10.5%, 5.4%, and 3.3%, respectively. Hospitalization at 7 or more days after injury was an independent risk factor for 1-year mortality (odds ratio, 1.76), the authors found.
Although the influence of surgical delay on mortality and morbidity among hip fracture patients has been widely studied, most data focus on surgery timing among hospitalized patients and fail to consider preadmission waiting time, they noted.
The current study aimed to assess outcomes based on “actual preadmission waiting time” through an analysis of data and surgical outcomes from a hospital electronic medical record system and from postoperative telephone interviews. Study subjects were patients aged over 65 years who underwent hip fracture surgery between Jan. 1, 2014, and Dec. 31, 2017. The mean age was 81.4 years, 74.7% of the patients were women, 67.1% had femoral neck fracture, and 56.1% had hip replacement surgery.
The findings, though limited by the retrospective nature of the study and the single-center design, suggest that, under the current conditions in China, admission delay may increase 1-year mortality, they wrote, concluding that “[i]n addition to early surgery highlighted in the guidelines, we also advocate early admission.”
The authors reported having no disclosures.
SOURCE: He W et al. World J Emerg Med. 2020;11(1):27-32.
FROM THE WORLD JOURNAL OF EMERGENCY MEDICINE