User login
Study urges better communication between surgeons, elderly patients
Clinicians would better serve their seriously ill elderly patients by improving communication to avoid emergency surgery that could prove to be too intense and potentially harmful, according to a study published online in Annals of Surgery (Ann. Surg. 2014 May 23 [doi: 10.1097/SLA.0000000000000721]).
Dr. Zara Cooper and her colleagues from Brigham and Women’s Hospital, Boston, conducted a wide-ranging literature review to ascertain factors that lead to communication challenges and nonbeneficial surgery at the end of life.
"Elderly patients with serious illness often receive poor-quality end-of-life care, leading to high rates of emergency department use, hospital and intensive care unit admissions, and in-hospital death, and low rates of hospice referral. High-intensity treatments near the end of life often are burdensome and prolong suffering without meeting patients’ personal goals," the researchers wrote.
If surgeons assume that patients favor life-prolonging treatment, they may opt to provide interventions that are immediately lifesaving but add little to the patient’s quality of life and may prolong, or even accelerate, the dying process, the researchers said. Studies have shown that nearly 1 in 10 Medicare patients who die have surgery in the last week of their lives (Lancet 2011;378:1408-13).
"Unable to quickly elucidate the gravity of the underlying illness and assess the patients’ understanding of their disease, surgeons may hesitate to engage in conversations about patient preferences and aggressive palliation as an alternative to invasive treatment," the authors wrote.
To avoid such communication pitfalls, the study recommends:
• Advance care planning around surgery: Even in an emergency setting, clinicians can set treatment goals early and revisit them if the patient’s condition deteriorates. Emergency surgeons should engage the patient’s primary and specialty providers whenever possible to participate in clinical decisions.
• Education and training: Surgeons routinely encountering seriously ill elderly patients should consider learning evidence-based communication strategies, including how to handle intense emotions, discuss prognosis, and deliver basic palliative-care interventions in a structured format with expert guidance.
• Structured communication: The goal is not to divert elderly patients with limited life expectancy from potentially life-prolonging surgery, but instead to determine patients’ priorities up front to better enable surgeons to recommend treatments. Such communication also should determine health states unacceptable to the patient, and affirm the clinician’s commitment to the patient’s well-being.
• Further research: More information is needed on patient-centered surgical outcomes for elderly patients with serious illness, including survival beyond 30 days, functional outcomes, health-related quality of life, transitions of care, and quality of death.
According to the study, physicians likely opt for invasive treatment of this vulnerable population out of concern about possible legal repercussions if they withhold it. But even when the patient’s prognosis is clear, legal concerns are minimal, and the patient is willing to discuss death and dying, many surgeons lack training in how to discuss death or provide a palliative approach to care, the researchers said.
Thus, surgeons may fail to respond to social cues relevant to end-of-life care or provide support, and instead "frequently employ defensive strategies" such as focusing on medical details, the study found.
Patients in distress often rely on surrogate decision-makers, the researchers noted. Given the disconnect between surrogates’ expectations and what medical treatment realistically can offer, surrogates may consent to treatments that they believe will prolong and restore quality of life when in fact such interventions are associated with physical or psychological suffering and scant, if any, benefit.
The authors did not receive funding support for this work and declared no conflicts of interest.
Clinicians would better serve their seriously ill elderly patients by improving communication to avoid emergency surgery that could prove to be too intense and potentially harmful, according to a study published online in Annals of Surgery (Ann. Surg. 2014 May 23 [doi: 10.1097/SLA.0000000000000721]).
Dr. Zara Cooper and her colleagues from Brigham and Women’s Hospital, Boston, conducted a wide-ranging literature review to ascertain factors that lead to communication challenges and nonbeneficial surgery at the end of life.
"Elderly patients with serious illness often receive poor-quality end-of-life care, leading to high rates of emergency department use, hospital and intensive care unit admissions, and in-hospital death, and low rates of hospice referral. High-intensity treatments near the end of life often are burdensome and prolong suffering without meeting patients’ personal goals," the researchers wrote.
If surgeons assume that patients favor life-prolonging treatment, they may opt to provide interventions that are immediately lifesaving but add little to the patient’s quality of life and may prolong, or even accelerate, the dying process, the researchers said. Studies have shown that nearly 1 in 10 Medicare patients who die have surgery in the last week of their lives (Lancet 2011;378:1408-13).
"Unable to quickly elucidate the gravity of the underlying illness and assess the patients’ understanding of their disease, surgeons may hesitate to engage in conversations about patient preferences and aggressive palliation as an alternative to invasive treatment," the authors wrote.
To avoid such communication pitfalls, the study recommends:
• Advance care planning around surgery: Even in an emergency setting, clinicians can set treatment goals early and revisit them if the patient’s condition deteriorates. Emergency surgeons should engage the patient’s primary and specialty providers whenever possible to participate in clinical decisions.
• Education and training: Surgeons routinely encountering seriously ill elderly patients should consider learning evidence-based communication strategies, including how to handle intense emotions, discuss prognosis, and deliver basic palliative-care interventions in a structured format with expert guidance.
• Structured communication: The goal is not to divert elderly patients with limited life expectancy from potentially life-prolonging surgery, but instead to determine patients’ priorities up front to better enable surgeons to recommend treatments. Such communication also should determine health states unacceptable to the patient, and affirm the clinician’s commitment to the patient’s well-being.
• Further research: More information is needed on patient-centered surgical outcomes for elderly patients with serious illness, including survival beyond 30 days, functional outcomes, health-related quality of life, transitions of care, and quality of death.
According to the study, physicians likely opt for invasive treatment of this vulnerable population out of concern about possible legal repercussions if they withhold it. But even when the patient’s prognosis is clear, legal concerns are minimal, and the patient is willing to discuss death and dying, many surgeons lack training in how to discuss death or provide a palliative approach to care, the researchers said.
Thus, surgeons may fail to respond to social cues relevant to end-of-life care or provide support, and instead "frequently employ defensive strategies" such as focusing on medical details, the study found.
Patients in distress often rely on surrogate decision-makers, the researchers noted. Given the disconnect between surrogates’ expectations and what medical treatment realistically can offer, surrogates may consent to treatments that they believe will prolong and restore quality of life when in fact such interventions are associated with physical or psychological suffering and scant, if any, benefit.
The authors did not receive funding support for this work and declared no conflicts of interest.
Clinicians would better serve their seriously ill elderly patients by improving communication to avoid emergency surgery that could prove to be too intense and potentially harmful, according to a study published online in Annals of Surgery (Ann. Surg. 2014 May 23 [doi: 10.1097/SLA.0000000000000721]).
Dr. Zara Cooper and her colleagues from Brigham and Women’s Hospital, Boston, conducted a wide-ranging literature review to ascertain factors that lead to communication challenges and nonbeneficial surgery at the end of life.
"Elderly patients with serious illness often receive poor-quality end-of-life care, leading to high rates of emergency department use, hospital and intensive care unit admissions, and in-hospital death, and low rates of hospice referral. High-intensity treatments near the end of life often are burdensome and prolong suffering without meeting patients’ personal goals," the researchers wrote.
If surgeons assume that patients favor life-prolonging treatment, they may opt to provide interventions that are immediately lifesaving but add little to the patient’s quality of life and may prolong, or even accelerate, the dying process, the researchers said. Studies have shown that nearly 1 in 10 Medicare patients who die have surgery in the last week of their lives (Lancet 2011;378:1408-13).
"Unable to quickly elucidate the gravity of the underlying illness and assess the patients’ understanding of their disease, surgeons may hesitate to engage in conversations about patient preferences and aggressive palliation as an alternative to invasive treatment," the authors wrote.
To avoid such communication pitfalls, the study recommends:
• Advance care planning around surgery: Even in an emergency setting, clinicians can set treatment goals early and revisit them if the patient’s condition deteriorates. Emergency surgeons should engage the patient’s primary and specialty providers whenever possible to participate in clinical decisions.
• Education and training: Surgeons routinely encountering seriously ill elderly patients should consider learning evidence-based communication strategies, including how to handle intense emotions, discuss prognosis, and deliver basic palliative-care interventions in a structured format with expert guidance.
• Structured communication: The goal is not to divert elderly patients with limited life expectancy from potentially life-prolonging surgery, but instead to determine patients’ priorities up front to better enable surgeons to recommend treatments. Such communication also should determine health states unacceptable to the patient, and affirm the clinician’s commitment to the patient’s well-being.
• Further research: More information is needed on patient-centered surgical outcomes for elderly patients with serious illness, including survival beyond 30 days, functional outcomes, health-related quality of life, transitions of care, and quality of death.
According to the study, physicians likely opt for invasive treatment of this vulnerable population out of concern about possible legal repercussions if they withhold it. But even when the patient’s prognosis is clear, legal concerns are minimal, and the patient is willing to discuss death and dying, many surgeons lack training in how to discuss death or provide a palliative approach to care, the researchers said.
Thus, surgeons may fail to respond to social cues relevant to end-of-life care or provide support, and instead "frequently employ defensive strategies" such as focusing on medical details, the study found.
Patients in distress often rely on surrogate decision-makers, the researchers noted. Given the disconnect between surrogates’ expectations and what medical treatment realistically can offer, surrogates may consent to treatments that they believe will prolong and restore quality of life when in fact such interventions are associated with physical or psychological suffering and scant, if any, benefit.
The authors did not receive funding support for this work and declared no conflicts of interest.
FROM ANNALS OF SURGERY
Palliative Chemotherapy May Be Harmful
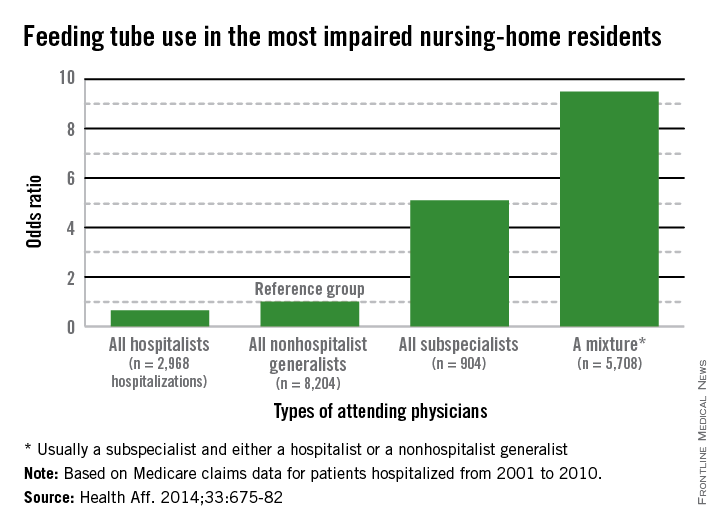
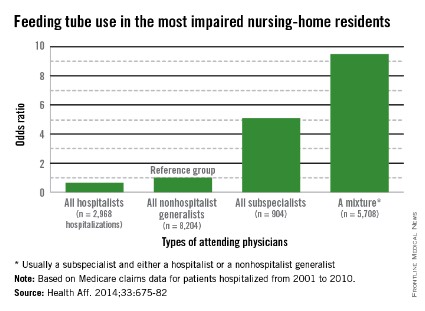
It is a natural inclination to want to prolong life, for however short a time. That’s why terminally ill cancer patients will often choose chemotherapy even though it may reduce their quality of life (QOL), say researchers from Harvard Medical School in Boston, Massachusetts, and Weill Cornell Medical College in New York, New York. And those wishes are honored, the researchers say: As many as 50% of patients with incurable cancers receive chemotherapy within 30 days of death, “despite growing concerns that it may not be effective.”
The researchers analyzed data from Coping With Cancer, a prospective, longitudinal study of patients with advanced cancer at 8 outpatient oncology clinics in the U.S. Of 386 patients, 56% were receiving palliative chemotherapy when they enrolled in the study. Those patients were more likely to choose life-extending care over comfort care, including chemotherapy, if it might add as little as 1 week to their life. Patients receiving palliative chemotherapy were less likely to acknowledge that their illness was terminal, compared with patients who were not receiving chemotherapy (35% vs 49%). They were also less likely to report that they had discussed their end-of-life wishes with a physician or to have completed a do not resuscitate order than were the patients who were not receiving chemotherapy (36% vs 49%).
However, the patients who received palliative chemotherapy were also much more likely to receive cardiopulmonary resuscitation, mechanical ventilation, or both in the last week of life (14% vs 2%) and twice as likely to be fed through a feeding tube in the last week of life (11% vs 5%), compared with patients who were not receiving chemotherapy. In addition, patients who received palliative chemotherapy were more likely to be referred late to hospice: 54% were enrolled within 1 week of death, compared with 37% of patients not on palliative chemotherapy.
Further, the choice of palliative chemotherapy meant that patients were more likely to die in an intensive care unit than at home or another preferred place (11% vs 2%, respectively). This is the first study, the researchers believe, to examine the associations between use of palliative chemotherapy and patients’ location of death. All those factors have been associated with worse QOL for patients at the end of life, more distress for their caregivers, and higher costs, the researchers say.
In this study, although only 40% of terminally ill patients receiving palliative chemotherapy preferred life-extending medical care to care that focused on relieving pain and discomfort, 86% preferred chemotherapy if it would give them 1 more week—but so did 60% of patients not receiving chemotherapy.
Many patients choose chemotherapy, though, because they have not been told about other options. This can be a difficult conversation, especially because both patients and physicians, the researchers note, may equate stopping treatment with “giving up” or “doing nothing.” But this study found no difference in survival between patients who received palliative chemotherapy and those who did not. That finding is a reminder, the researchers say, “that palliative chemotherapy does not necessarily extend life….[C]ontinuing treatment should not come at the cost of engaging in advance care planning” to ensure that “patients’ end-of-life experiences are congruent with their values.”
Source
Wright AA, Zhang B, Keating NL, Weeks JC, Prigerson HG. BMJ. 2014;348:g1219.
doi: 10.1136/bmj.g1219.
It is a natural inclination to want to prolong life, for however short a time. That’s why terminally ill cancer patients will often choose chemotherapy even though it may reduce their quality of life (QOL), say researchers from Harvard Medical School in Boston, Massachusetts, and Weill Cornell Medical College in New York, New York. And those wishes are honored, the researchers say: As many as 50% of patients with incurable cancers receive chemotherapy within 30 days of death, “despite growing concerns that it may not be effective.”
The researchers analyzed data from Coping With Cancer, a prospective, longitudinal study of patients with advanced cancer at 8 outpatient oncology clinics in the U.S. Of 386 patients, 56% were receiving palliative chemotherapy when they enrolled in the study. Those patients were more likely to choose life-extending care over comfort care, including chemotherapy, if it might add as little as 1 week to their life. Patients receiving palliative chemotherapy were less likely to acknowledge that their illness was terminal, compared with patients who were not receiving chemotherapy (35% vs 49%). They were also less likely to report that they had discussed their end-of-life wishes with a physician or to have completed a do not resuscitate order than were the patients who were not receiving chemotherapy (36% vs 49%).
However, the patients who received palliative chemotherapy were also much more likely to receive cardiopulmonary resuscitation, mechanical ventilation, or both in the last week of life (14% vs 2%) and twice as likely to be fed through a feeding tube in the last week of life (11% vs 5%), compared with patients who were not receiving chemotherapy. In addition, patients who received palliative chemotherapy were more likely to be referred late to hospice: 54% were enrolled within 1 week of death, compared with 37% of patients not on palliative chemotherapy.
Further, the choice of palliative chemotherapy meant that patients were more likely to die in an intensive care unit than at home or another preferred place (11% vs 2%, respectively). This is the first study, the researchers believe, to examine the associations between use of palliative chemotherapy and patients’ location of death. All those factors have been associated with worse QOL for patients at the end of life, more distress for their caregivers, and higher costs, the researchers say.
In this study, although only 40% of terminally ill patients receiving palliative chemotherapy preferred life-extending medical care to care that focused on relieving pain and discomfort, 86% preferred chemotherapy if it would give them 1 more week—but so did 60% of patients not receiving chemotherapy.
Many patients choose chemotherapy, though, because they have not been told about other options. This can be a difficult conversation, especially because both patients and physicians, the researchers note, may equate stopping treatment with “giving up” or “doing nothing.” But this study found no difference in survival between patients who received palliative chemotherapy and those who did not. That finding is a reminder, the researchers say, “that palliative chemotherapy does not necessarily extend life….[C]ontinuing treatment should not come at the cost of engaging in advance care planning” to ensure that “patients’ end-of-life experiences are congruent with their values.”
Source
Wright AA, Zhang B, Keating NL, Weeks JC, Prigerson HG. BMJ. 2014;348:g1219.
doi: 10.1136/bmj.g1219.
It is a natural inclination to want to prolong life, for however short a time. That’s why terminally ill cancer patients will often choose chemotherapy even though it may reduce their quality of life (QOL), say researchers from Harvard Medical School in Boston, Massachusetts, and Weill Cornell Medical College in New York, New York. And those wishes are honored, the researchers say: As many as 50% of patients with incurable cancers receive chemotherapy within 30 days of death, “despite growing concerns that it may not be effective.”
The researchers analyzed data from Coping With Cancer, a prospective, longitudinal study of patients with advanced cancer at 8 outpatient oncology clinics in the U.S. Of 386 patients, 56% were receiving palliative chemotherapy when they enrolled in the study. Those patients were more likely to choose life-extending care over comfort care, including chemotherapy, if it might add as little as 1 week to their life. Patients receiving palliative chemotherapy were less likely to acknowledge that their illness was terminal, compared with patients who were not receiving chemotherapy (35% vs 49%). They were also less likely to report that they had discussed their end-of-life wishes with a physician or to have completed a do not resuscitate order than were the patients who were not receiving chemotherapy (36% vs 49%).
However, the patients who received palliative chemotherapy were also much more likely to receive cardiopulmonary resuscitation, mechanical ventilation, or both in the last week of life (14% vs 2%) and twice as likely to be fed through a feeding tube in the last week of life (11% vs 5%), compared with patients who were not receiving chemotherapy. In addition, patients who received palliative chemotherapy were more likely to be referred late to hospice: 54% were enrolled within 1 week of death, compared with 37% of patients not on palliative chemotherapy.
Further, the choice of palliative chemotherapy meant that patients were more likely to die in an intensive care unit than at home or another preferred place (11% vs 2%, respectively). This is the first study, the researchers believe, to examine the associations between use of palliative chemotherapy and patients’ location of death. All those factors have been associated with worse QOL for patients at the end of life, more distress for their caregivers, and higher costs, the researchers say.
In this study, although only 40% of terminally ill patients receiving palliative chemotherapy preferred life-extending medical care to care that focused on relieving pain and discomfort, 86% preferred chemotherapy if it would give them 1 more week—but so did 60% of patients not receiving chemotherapy.
Many patients choose chemotherapy, though, because they have not been told about other options. This can be a difficult conversation, especially because both patients and physicians, the researchers note, may equate stopping treatment with “giving up” or “doing nothing.” But this study found no difference in survival between patients who received palliative chemotherapy and those who did not. That finding is a reminder, the researchers say, “that palliative chemotherapy does not necessarily extend life….[C]ontinuing treatment should not come at the cost of engaging in advance care planning” to ensure that “patients’ end-of-life experiences are congruent with their values.”
Source
Wright AA, Zhang B, Keating NL, Weeks JC, Prigerson HG. BMJ. 2014;348:g1219.
doi: 10.1136/bmj.g1219.
Naloxegol cut opioid-associated constipation without impairing pain relief
Treatment with opioid receptor-antagonist naloxegol significantly improved opioid-associated constipation, compared with placebo, without affecting pain scores or daily opioid requirements, according to data from two identical double-blind studies.
Outpatients with noncancer pain who were given 25 mg of naloxegol showed a significantly shorter time to first spontaneous bowel movement after treatment, compared with those given placebo – a median time of 5.9 hours and 12 hours in the two studies, compared with 35.8 hours and 37.2 hours with placebo, according to a study published online June 4 in the New England Journal of Medicine.
Treatment with naloxegol was also associated with a significantly greater number of spontaneous bowel movements over the course of the 12-week study period, compared with placebo, and an increase in the mean number of days per week with one or more spontaneous bowel movements.
The Food and Drug Administration is currently considering whether to approve naloxegol; the agency is expected to decide by Sept. 16, 2014.
The two phase III randomized, controlled studies were nearly identical in size – one including 652 individuals with opioid-induced constipation, and the other including 700 – and entirely identical in design: Participants were randomized to receive either 25 mg or 12.5 mg of naloxegol daily, or placebo.
"In both studies, naloxegol at a dose of 25 mg was associated with an increased rate of response (10-15 percentage points higher than the response with placebo) over a period of 12 weeks," wrote Dr. William D. Chey of the University of Michigan Health System, Ann Arbor, and his colleagues.
The higher dose of naloxegol also was associated with more significant improvements in severity of straining, stool consistency, and the frequency of days with complete, spontaneous bowel movements.
Naloxegol’s benefits were even greater among individuals who had previously failed to respond to laxatives before study enrollment, which accounted for 71% of participants, a prespecified subgroup analysis showed (N. Engl. J. Med. 2014 June 4 [doi:10.1056/NEJMoa1310246]).
"In clinical practice, osmotic and stimulant laxatives are likely to be used before more expensive prescription medications," the researchers wrote. "Thus, the finding that naloxegol proved beneficial in patients who had persistent symptoms of opioid-induced constipation despite using standard laxatives is of potential importance."
There were some dose-related side effects observed in the naloxegol group, including abdominal pain, nausea, diarrhea, and vomiting, occurring soon after initiation of treatment. But most of these effects were mild to moderate.
There had been concern about potential cardiovascular side effects, which had been observed previously with alvimopan, another peripherally-acting mu-opioid antagonist. However the incidence of major cardiovascular events was rare and similar across both active and placebo groups.
Researchers found no significant interaction between naloxegol treatment and daily opioid dose, and there also were no significant differences in the mean change from baseline in pain scores.
Around half of the patients enrolled were taking opioids for back pain, while other reasons included arthritis, joint pain, or fibromyalgia. On average, participants had been taking opioids for 3.65 years.
AstraZeneca supported the study, and the authors declared a range of grants, consultancies, and other financial relationships with several pharmaceutical companies, including AstraZeneca.
Treatment with opioid receptor-antagonist naloxegol significantly improved opioid-associated constipation, compared with placebo, without affecting pain scores or daily opioid requirements, according to data from two identical double-blind studies.
Outpatients with noncancer pain who were given 25 mg of naloxegol showed a significantly shorter time to first spontaneous bowel movement after treatment, compared with those given placebo – a median time of 5.9 hours and 12 hours in the two studies, compared with 35.8 hours and 37.2 hours with placebo, according to a study published online June 4 in the New England Journal of Medicine.
Treatment with naloxegol was also associated with a significantly greater number of spontaneous bowel movements over the course of the 12-week study period, compared with placebo, and an increase in the mean number of days per week with one or more spontaneous bowel movements.
The Food and Drug Administration is currently considering whether to approve naloxegol; the agency is expected to decide by Sept. 16, 2014.
The two phase III randomized, controlled studies were nearly identical in size – one including 652 individuals with opioid-induced constipation, and the other including 700 – and entirely identical in design: Participants were randomized to receive either 25 mg or 12.5 mg of naloxegol daily, or placebo.
"In both studies, naloxegol at a dose of 25 mg was associated with an increased rate of response (10-15 percentage points higher than the response with placebo) over a period of 12 weeks," wrote Dr. William D. Chey of the University of Michigan Health System, Ann Arbor, and his colleagues.
The higher dose of naloxegol also was associated with more significant improvements in severity of straining, stool consistency, and the frequency of days with complete, spontaneous bowel movements.
Naloxegol’s benefits were even greater among individuals who had previously failed to respond to laxatives before study enrollment, which accounted for 71% of participants, a prespecified subgroup analysis showed (N. Engl. J. Med. 2014 June 4 [doi:10.1056/NEJMoa1310246]).
"In clinical practice, osmotic and stimulant laxatives are likely to be used before more expensive prescription medications," the researchers wrote. "Thus, the finding that naloxegol proved beneficial in patients who had persistent symptoms of opioid-induced constipation despite using standard laxatives is of potential importance."
There were some dose-related side effects observed in the naloxegol group, including abdominal pain, nausea, diarrhea, and vomiting, occurring soon after initiation of treatment. But most of these effects were mild to moderate.
There had been concern about potential cardiovascular side effects, which had been observed previously with alvimopan, another peripherally-acting mu-opioid antagonist. However the incidence of major cardiovascular events was rare and similar across both active and placebo groups.
Researchers found no significant interaction between naloxegol treatment and daily opioid dose, and there also were no significant differences in the mean change from baseline in pain scores.
Around half of the patients enrolled were taking opioids for back pain, while other reasons included arthritis, joint pain, or fibromyalgia. On average, participants had been taking opioids for 3.65 years.
AstraZeneca supported the study, and the authors declared a range of grants, consultancies, and other financial relationships with several pharmaceutical companies, including AstraZeneca.
Treatment with opioid receptor-antagonist naloxegol significantly improved opioid-associated constipation, compared with placebo, without affecting pain scores or daily opioid requirements, according to data from two identical double-blind studies.
Outpatients with noncancer pain who were given 25 mg of naloxegol showed a significantly shorter time to first spontaneous bowel movement after treatment, compared with those given placebo – a median time of 5.9 hours and 12 hours in the two studies, compared with 35.8 hours and 37.2 hours with placebo, according to a study published online June 4 in the New England Journal of Medicine.
Treatment with naloxegol was also associated with a significantly greater number of spontaneous bowel movements over the course of the 12-week study period, compared with placebo, and an increase in the mean number of days per week with one or more spontaneous bowel movements.
The Food and Drug Administration is currently considering whether to approve naloxegol; the agency is expected to decide by Sept. 16, 2014.
The two phase III randomized, controlled studies were nearly identical in size – one including 652 individuals with opioid-induced constipation, and the other including 700 – and entirely identical in design: Participants were randomized to receive either 25 mg or 12.5 mg of naloxegol daily, or placebo.
"In both studies, naloxegol at a dose of 25 mg was associated with an increased rate of response (10-15 percentage points higher than the response with placebo) over a period of 12 weeks," wrote Dr. William D. Chey of the University of Michigan Health System, Ann Arbor, and his colleagues.
The higher dose of naloxegol also was associated with more significant improvements in severity of straining, stool consistency, and the frequency of days with complete, spontaneous bowel movements.
Naloxegol’s benefits were even greater among individuals who had previously failed to respond to laxatives before study enrollment, which accounted for 71% of participants, a prespecified subgroup analysis showed (N. Engl. J. Med. 2014 June 4 [doi:10.1056/NEJMoa1310246]).
"In clinical practice, osmotic and stimulant laxatives are likely to be used before more expensive prescription medications," the researchers wrote. "Thus, the finding that naloxegol proved beneficial in patients who had persistent symptoms of opioid-induced constipation despite using standard laxatives is of potential importance."
There were some dose-related side effects observed in the naloxegol group, including abdominal pain, nausea, diarrhea, and vomiting, occurring soon after initiation of treatment. But most of these effects were mild to moderate.
There had been concern about potential cardiovascular side effects, which had been observed previously with alvimopan, another peripherally-acting mu-opioid antagonist. However the incidence of major cardiovascular events was rare and similar across both active and placebo groups.
Researchers found no significant interaction between naloxegol treatment and daily opioid dose, and there also were no significant differences in the mean change from baseline in pain scores.
Around half of the patients enrolled were taking opioids for back pain, while other reasons included arthritis, joint pain, or fibromyalgia. On average, participants had been taking opioids for 3.65 years.
AstraZeneca supported the study, and the authors declared a range of grants, consultancies, and other financial relationships with several pharmaceutical companies, including AstraZeneca.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Naloxegol improves opioid-induced constipation without affecting pain relief.
Major finding: Treatment with naloxegol was associated with a significantly shorter time to first spontaneous bowel movement, a significantly greater number of spontaneous bowel movements, and an increase in the mean number of days per week with one or more spontaneous bowel movements, compared with placebo.
Data source: Two identical 12-week, phase III, double-blind, randomized, controlled trials in a total of 1,352 patients with opioid-associated constipation.
Disclosures: AstraZeneca supported the study, and the authors declared a range of grants, consultancies, and other financial relationships with several pharmaceutical companies including AstraZeneca.
Finally, a Way to Relieve Cancer-related Fatigue
PRACTICE CHANGER
Recommend American ginseng (1,000 mg bid) for four weeks to improve cancer-related fatigue in patients who are undergoing radiation or chemotherapy; no other treatment has been shown to be effective.1
STRENGTH OF RECOMMENDATION
B: Based on a single well-done randomized controlled trial (RCT).1
ILLUSTRATIVE CASE
A 54-year-old woman is receiving chemotherapy for adenocarcinoma of the right breast (T2N1M0) and has persistent, disabling fatigue. She has been unable to work or care for her family since starting chemotherapy. She says she gets enough sleep and denies being depressed or in pain. Lab testing for anemia and thyroid dysfunction is negative. Is there a safe and effective intervention?
On the next page: Study summary >>
Cancer-related fatigue is a common, distressing symptom that occurs in more than half of all patients undergoing chemotherapy and more than two-thirds of those receiving radiation therapy.2 For many cancer survivors, fatigue can persist for five to 10 years after treatment.3
Because no treatments have proven effective, many clinicians and patients accept fatigue as inevitable. In RCTs, psychostimulants (eg, methylphenidate) and antidepressants (eg, donepezil and paroxetine) have not been found effective.4-6 Dietary supplements, such as coenzyme Q10 and l-carnitine, also have not been found effective in placebo-controlled trials.7,8
The double-blind RCT reported on here looked at whether American ginseng might be effective in relieving cancer-related fatigue.
STUDY SUMMARY
Ginseng reduced fatigue after eight weeks
There are two major species of ginseng—Asian and American—and they have varying amounts, strengths, and varieties of ginsenosides, which are the active ingredients. In this eight-week, double-blind RCT, Barton et al1 randomly assigned more than 300 patients from 40 US cancer facilities to receive either 1,000 mg of American ginseng twice daily (in the morning and at noon) or matched placebo capsules.
Patients were either currently receiving treatment for cancer or were posttreatment but within two years of receiving a cancer diagnosis. All participants had experienced fatigue of at least a month’s duration that they rated as 4 or higher on a scale of 0 to 10. Patients with other causes of fatigue were excluded, as were those who had pain or insomnia rated 4 or higher, those with brain cancer or central nervous system (CNS) lymphoma, those taking systemic steroids or opioids, and those who were using, or had used, ginseng or other agents for fatigue.
Of the 364 randomized participants, 300 (147 ginseng patients, 153 placebo patients) remained in the study through the primary endpoint at four weeks, and 261 completed the entire eight-week study. There were no baseline differences between groups in demographic characteristics, time since cancer diagnosis, cancer type, current or prior treatment, and fatigue at baseline.
The primary outcome was a change in score on the Multidimensional Fatigue Symptom Inventory–Short Form (MFSI–SF) at four weeks. Secondary outcomes included a change in MFSI–SF score at eight weeks. The authors also conducted a subset analysis comparing ginseng and placebo in only those patients currently undergoing cancer treatment versus those who had completed treatment. To make it easier to compare results, all scores were converted to a 100-point scale; higher scores indicated less fatigue. Adverse events were documented by patient self-report questionnaires and also by researchers who called or visited patients every other week.
While ginseng did not appear to significantly impact fatigue scores versus placebo at four weeks (14.4 vs 8.2), fatigue scores at eight weeks were significantly improved (20 vs 10.3). Interestingly, though, there was a significant improvement in fatigue scores with ginseng at both four weeks and eight weeks when researchers looked at only those patients who were currently receiving cancer treatment. On the other hand, those patients who were not currently undergoing treatment did not show a significant improvement at either time cutoff.
There was no statistically significant difference in adverse events between the ginseng and placebo groups over the eight-week study.
On the next page: What's new >>
WHAT’S NEW
First evidence-based therapy
We now have good evidence that American ginseng (1,000 mg bid) is safe and effective for ameliorating cancer-related fatigue. Before this study, no other effective treatment had been identified.
CAVEATS
Ginseng may not help posttreatment
In this study, ginseng did not improve fatigue at four weeks, which was the primary outcome, although benefits were noted after eight weeks of treatment. Interestingly, though, participants who were receiving radiation and/or chemotherapy during the study experienced significant improvements at four and eight weeks, while those with previous (but not current) treatment did not significantly improve at either time point.
It may be that ginseng works best to ameliorate cancer-related fatigue in patients simultaneously receiving cancer treatment but not in those who have completed treatment. The findings also suggest that patients who have completed treatment may wish to try ginseng for longer than eight weeks to see if it offers any benefit.
Because this study excluded patients with brain cancer, CNS lymphoma, moderate to severe pain, or insomnia and those taking steroids, it is not known if ginseng would help them.
In one study, a low-dose methanolic extract of American ginseng caused a breast cancer cell line to proliferate; however, it was later discovered that this extract had been contaminated with Fusarium fungi containing zearalenone, which has strong estrogenic activity.9,10 However, higher doses of a similar methanolic extract, as well as other water-based extracts, have reduced proliferation of breast cancer cells.11
Proceed carefully if a patient is taking warfarin. Coadministration of ginseng and warfarin may reduce both warfarin concentrations and a patient’s international normalized ratio (INR).12 Therefore, carefully monitor INR in patients concurrently taking ginseng and warfarin. Furthermore, ginseng may lower blood glucose in patients with diabetes, so carefully monitor blood glucose in these patients when initiating or discontinuing ginseng.13
CHALLENGES TO IMPLEMENTATION
It’s hard to know exactly what you’re getting
Regulating dietary supplements, especially verifying ingredients and potency, has been a challenge for the FDA. Although ginseng commonly is adulterated, this is more common with the Asian species (Panax ginseng) than with the American species (Panax quinquefolius) used in this study.10 Clinicians who want to recommend ginseng for cancer-related fatigue should advise patients to use American ginseng root products produced in the US. Additionally, the products should contain at least 3% ginsenosides to match the dose used in this study.
REFERENCES
1. Barton DL, Liu H, Dakhil SR, et al. Wisconsin Ginseng (Panax quinquefolius) to improve cancer-related fatigue: a randomized, double-blind trial, N07C2. J Natl Cancer Inst. 2013;105(16):1230-1238.
2. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 (suppl 1):4-10.
3. Bower JE, Ganz PA, Desmond KA, et al. Fatigue in long-term breast carcinoma survivors: a longitudinal investigation. Cancer. 2006;106(4):751-758.
4. Moraska AR, Sood A, Dakhil SR, et al. Phase III, randomized, double-blind, placebo-controlled study of long-acting methylphenidate for cancer-related fatigue: North Central Cancer Treatment Group NCCTG-N05C7 trial. J Clin Oncol. 2010;28(23):3673-3679.
5. Bruera E, El Osta B, Valero V, et al. Donepezil for cancer fatigue: a double-blind, randomized, placebo-controlled trial. J Clin Oncol. 2007;25(23):3475-3481.
6. Morrow GR, Hickok JT, Roscoe JA, et al; University of Rochester Cancer Center Community Clinical Oncology Program. Differential effects of paroxetine on fatigue and depression: a randomized, double-blind trial from the University of Rochester Cancer Center Community Clinical Oncology Program. J Clin Oncol. 2003;21(24):4635-4641.
7. Lesser GJ, Case D, Stark N, et al; Wake Forest University Community Clinical Oncology Program Research Base. A randomized, double-blind, placebo-controlled study of oral coenzyme Q10 to relieve self-reported treatment-related fatigue in newly diagnosed patients with breast cancer. J Support Oncol. 2013;11(1):31-42.
8. Cruciani RA, Zhang JJ, Manola J, et al. L-carnitine supplementation for the management of fatigue in patients with cancer: an Eastern cooperative oncology group phase III, randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2012;30(31):3864-3869.
9. Duda RB, Zhong Y, Navas V, et al. American ginseng and breast cancer therapeutic agents synergistically inhibit MCF-7
breast cancer cell growth. J Surg Oncol. 1999;72(4):230-239.
10. Upton R, ed. American ginseng root Panax quinquefolius, standards of analysis, quality control, and therapeutics. Scotts Valley, CA: American Herbal Pharmacopoeia; 2012.
11. King ML, Adler SR, Murphy LL. Extraction-dependent effects of American ginseng (Panax quinquefolium) on human breast cancer cell proliferation and estrogen receptor activation. Integr Cancer Ther. 2006;5(3): 236-243.
12. Yuan CS, Wei G, Dey L, et al. Brief communication: American ginseng reduces warfarin’s effect in healthy patients: a randomized, controlled trial. Ann Intern Med. 2004;141(1):23-27.
13. Vuksan V, Stavro MP, Sievenpiper JL, et al. Similar postprandial glycemic reductions with escalation of dose and administration time of American ginseng in type 2 diabetes. Diabetes Care. 2000;23(9):1221-1226.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(5):270-272.
PRACTICE CHANGER
Recommend American ginseng (1,000 mg bid) for four weeks to improve cancer-related fatigue in patients who are undergoing radiation or chemotherapy; no other treatment has been shown to be effective.1
STRENGTH OF RECOMMENDATION
B: Based on a single well-done randomized controlled trial (RCT).1
ILLUSTRATIVE CASE
A 54-year-old woman is receiving chemotherapy for adenocarcinoma of the right breast (T2N1M0) and has persistent, disabling fatigue. She has been unable to work or care for her family since starting chemotherapy. She says she gets enough sleep and denies being depressed or in pain. Lab testing for anemia and thyroid dysfunction is negative. Is there a safe and effective intervention?
On the next page: Study summary >>
Cancer-related fatigue is a common, distressing symptom that occurs in more than half of all patients undergoing chemotherapy and more than two-thirds of those receiving radiation therapy.2 For many cancer survivors, fatigue can persist for five to 10 years after treatment.3
Because no treatments have proven effective, many clinicians and patients accept fatigue as inevitable. In RCTs, psychostimulants (eg, methylphenidate) and antidepressants (eg, donepezil and paroxetine) have not been found effective.4-6 Dietary supplements, such as coenzyme Q10 and l-carnitine, also have not been found effective in placebo-controlled trials.7,8
The double-blind RCT reported on here looked at whether American ginseng might be effective in relieving cancer-related fatigue.
STUDY SUMMARY
Ginseng reduced fatigue after eight weeks
There are two major species of ginseng—Asian and American—and they have varying amounts, strengths, and varieties of ginsenosides, which are the active ingredients. In this eight-week, double-blind RCT, Barton et al1 randomly assigned more than 300 patients from 40 US cancer facilities to receive either 1,000 mg of American ginseng twice daily (in the morning and at noon) or matched placebo capsules.
Patients were either currently receiving treatment for cancer or were posttreatment but within two years of receiving a cancer diagnosis. All participants had experienced fatigue of at least a month’s duration that they rated as 4 or higher on a scale of 0 to 10. Patients with other causes of fatigue were excluded, as were those who had pain or insomnia rated 4 or higher, those with brain cancer or central nervous system (CNS) lymphoma, those taking systemic steroids or opioids, and those who were using, or had used, ginseng or other agents for fatigue.
Of the 364 randomized participants, 300 (147 ginseng patients, 153 placebo patients) remained in the study through the primary endpoint at four weeks, and 261 completed the entire eight-week study. There were no baseline differences between groups in demographic characteristics, time since cancer diagnosis, cancer type, current or prior treatment, and fatigue at baseline.
The primary outcome was a change in score on the Multidimensional Fatigue Symptom Inventory–Short Form (MFSI–SF) at four weeks. Secondary outcomes included a change in MFSI–SF score at eight weeks. The authors also conducted a subset analysis comparing ginseng and placebo in only those patients currently undergoing cancer treatment versus those who had completed treatment. To make it easier to compare results, all scores were converted to a 100-point scale; higher scores indicated less fatigue. Adverse events were documented by patient self-report questionnaires and also by researchers who called or visited patients every other week.
While ginseng did not appear to significantly impact fatigue scores versus placebo at four weeks (14.4 vs 8.2), fatigue scores at eight weeks were significantly improved (20 vs 10.3). Interestingly, though, there was a significant improvement in fatigue scores with ginseng at both four weeks and eight weeks when researchers looked at only those patients who were currently receiving cancer treatment. On the other hand, those patients who were not currently undergoing treatment did not show a significant improvement at either time cutoff.
There was no statistically significant difference in adverse events between the ginseng and placebo groups over the eight-week study.
On the next page: What's new >>
WHAT’S NEW
First evidence-based therapy
We now have good evidence that American ginseng (1,000 mg bid) is safe and effective for ameliorating cancer-related fatigue. Before this study, no other effective treatment had been identified.
CAVEATS
Ginseng may not help posttreatment
In this study, ginseng did not improve fatigue at four weeks, which was the primary outcome, although benefits were noted after eight weeks of treatment. Interestingly, though, participants who were receiving radiation and/or chemotherapy during the study experienced significant improvements at four and eight weeks, while those with previous (but not current) treatment did not significantly improve at either time point.
It may be that ginseng works best to ameliorate cancer-related fatigue in patients simultaneously receiving cancer treatment but not in those who have completed treatment. The findings also suggest that patients who have completed treatment may wish to try ginseng for longer than eight weeks to see if it offers any benefit.
Because this study excluded patients with brain cancer, CNS lymphoma, moderate to severe pain, or insomnia and those taking steroids, it is not known if ginseng would help them.
In one study, a low-dose methanolic extract of American ginseng caused a breast cancer cell line to proliferate; however, it was later discovered that this extract had been contaminated with Fusarium fungi containing zearalenone, which has strong estrogenic activity.9,10 However, higher doses of a similar methanolic extract, as well as other water-based extracts, have reduced proliferation of breast cancer cells.11
Proceed carefully if a patient is taking warfarin. Coadministration of ginseng and warfarin may reduce both warfarin concentrations and a patient’s international normalized ratio (INR).12 Therefore, carefully monitor INR in patients concurrently taking ginseng and warfarin. Furthermore, ginseng may lower blood glucose in patients with diabetes, so carefully monitor blood glucose in these patients when initiating or discontinuing ginseng.13
CHALLENGES TO IMPLEMENTATION
It’s hard to know exactly what you’re getting
Regulating dietary supplements, especially verifying ingredients and potency, has been a challenge for the FDA. Although ginseng commonly is adulterated, this is more common with the Asian species (Panax ginseng) than with the American species (Panax quinquefolius) used in this study.10 Clinicians who want to recommend ginseng for cancer-related fatigue should advise patients to use American ginseng root products produced in the US. Additionally, the products should contain at least 3% ginsenosides to match the dose used in this study.
REFERENCES
1. Barton DL, Liu H, Dakhil SR, et al. Wisconsin Ginseng (Panax quinquefolius) to improve cancer-related fatigue: a randomized, double-blind trial, N07C2. J Natl Cancer Inst. 2013;105(16):1230-1238.
2. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 (suppl 1):4-10.
3. Bower JE, Ganz PA, Desmond KA, et al. Fatigue in long-term breast carcinoma survivors: a longitudinal investigation. Cancer. 2006;106(4):751-758.
4. Moraska AR, Sood A, Dakhil SR, et al. Phase III, randomized, double-blind, placebo-controlled study of long-acting methylphenidate for cancer-related fatigue: North Central Cancer Treatment Group NCCTG-N05C7 trial. J Clin Oncol. 2010;28(23):3673-3679.
5. Bruera E, El Osta B, Valero V, et al. Donepezil for cancer fatigue: a double-blind, randomized, placebo-controlled trial. J Clin Oncol. 2007;25(23):3475-3481.
6. Morrow GR, Hickok JT, Roscoe JA, et al; University of Rochester Cancer Center Community Clinical Oncology Program. Differential effects of paroxetine on fatigue and depression: a randomized, double-blind trial from the University of Rochester Cancer Center Community Clinical Oncology Program. J Clin Oncol. 2003;21(24):4635-4641.
7. Lesser GJ, Case D, Stark N, et al; Wake Forest University Community Clinical Oncology Program Research Base. A randomized, double-blind, placebo-controlled study of oral coenzyme Q10 to relieve self-reported treatment-related fatigue in newly diagnosed patients with breast cancer. J Support Oncol. 2013;11(1):31-42.
8. Cruciani RA, Zhang JJ, Manola J, et al. L-carnitine supplementation for the management of fatigue in patients with cancer: an Eastern cooperative oncology group phase III, randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2012;30(31):3864-3869.
9. Duda RB, Zhong Y, Navas V, et al. American ginseng and breast cancer therapeutic agents synergistically inhibit MCF-7
breast cancer cell growth. J Surg Oncol. 1999;72(4):230-239.
10. Upton R, ed. American ginseng root Panax quinquefolius, standards of analysis, quality control, and therapeutics. Scotts Valley, CA: American Herbal Pharmacopoeia; 2012.
11. King ML, Adler SR, Murphy LL. Extraction-dependent effects of American ginseng (Panax quinquefolium) on human breast cancer cell proliferation and estrogen receptor activation. Integr Cancer Ther. 2006;5(3): 236-243.
12. Yuan CS, Wei G, Dey L, et al. Brief communication: American ginseng reduces warfarin’s effect in healthy patients: a randomized, controlled trial. Ann Intern Med. 2004;141(1):23-27.
13. Vuksan V, Stavro MP, Sievenpiper JL, et al. Similar postprandial glycemic reductions with escalation of dose and administration time of American ginseng in type 2 diabetes. Diabetes Care. 2000;23(9):1221-1226.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(5):270-272.
PRACTICE CHANGER
Recommend American ginseng (1,000 mg bid) for four weeks to improve cancer-related fatigue in patients who are undergoing radiation or chemotherapy; no other treatment has been shown to be effective.1
STRENGTH OF RECOMMENDATION
B: Based on a single well-done randomized controlled trial (RCT).1
ILLUSTRATIVE CASE
A 54-year-old woman is receiving chemotherapy for adenocarcinoma of the right breast (T2N1M0) and has persistent, disabling fatigue. She has been unable to work or care for her family since starting chemotherapy. She says she gets enough sleep and denies being depressed or in pain. Lab testing for anemia and thyroid dysfunction is negative. Is there a safe and effective intervention?
On the next page: Study summary >>
Cancer-related fatigue is a common, distressing symptom that occurs in more than half of all patients undergoing chemotherapy and more than two-thirds of those receiving radiation therapy.2 For many cancer survivors, fatigue can persist for five to 10 years after treatment.3
Because no treatments have proven effective, many clinicians and patients accept fatigue as inevitable. In RCTs, psychostimulants (eg, methylphenidate) and antidepressants (eg, donepezil and paroxetine) have not been found effective.4-6 Dietary supplements, such as coenzyme Q10 and l-carnitine, also have not been found effective in placebo-controlled trials.7,8
The double-blind RCT reported on here looked at whether American ginseng might be effective in relieving cancer-related fatigue.
STUDY SUMMARY
Ginseng reduced fatigue after eight weeks
There are two major species of ginseng—Asian and American—and they have varying amounts, strengths, and varieties of ginsenosides, which are the active ingredients. In this eight-week, double-blind RCT, Barton et al1 randomly assigned more than 300 patients from 40 US cancer facilities to receive either 1,000 mg of American ginseng twice daily (in the morning and at noon) or matched placebo capsules.
Patients were either currently receiving treatment for cancer or were posttreatment but within two years of receiving a cancer diagnosis. All participants had experienced fatigue of at least a month’s duration that they rated as 4 or higher on a scale of 0 to 10. Patients with other causes of fatigue were excluded, as were those who had pain or insomnia rated 4 or higher, those with brain cancer or central nervous system (CNS) lymphoma, those taking systemic steroids or opioids, and those who were using, or had used, ginseng or other agents for fatigue.
Of the 364 randomized participants, 300 (147 ginseng patients, 153 placebo patients) remained in the study through the primary endpoint at four weeks, and 261 completed the entire eight-week study. There were no baseline differences between groups in demographic characteristics, time since cancer diagnosis, cancer type, current or prior treatment, and fatigue at baseline.
The primary outcome was a change in score on the Multidimensional Fatigue Symptom Inventory–Short Form (MFSI–SF) at four weeks. Secondary outcomes included a change in MFSI–SF score at eight weeks. The authors also conducted a subset analysis comparing ginseng and placebo in only those patients currently undergoing cancer treatment versus those who had completed treatment. To make it easier to compare results, all scores were converted to a 100-point scale; higher scores indicated less fatigue. Adverse events were documented by patient self-report questionnaires and also by researchers who called or visited patients every other week.
While ginseng did not appear to significantly impact fatigue scores versus placebo at four weeks (14.4 vs 8.2), fatigue scores at eight weeks were significantly improved (20 vs 10.3). Interestingly, though, there was a significant improvement in fatigue scores with ginseng at both four weeks and eight weeks when researchers looked at only those patients who were currently receiving cancer treatment. On the other hand, those patients who were not currently undergoing treatment did not show a significant improvement at either time cutoff.
There was no statistically significant difference in adverse events between the ginseng and placebo groups over the eight-week study.
On the next page: What's new >>
WHAT’S NEW
First evidence-based therapy
We now have good evidence that American ginseng (1,000 mg bid) is safe and effective for ameliorating cancer-related fatigue. Before this study, no other effective treatment had been identified.
CAVEATS
Ginseng may not help posttreatment
In this study, ginseng did not improve fatigue at four weeks, which was the primary outcome, although benefits were noted after eight weeks of treatment. Interestingly, though, participants who were receiving radiation and/or chemotherapy during the study experienced significant improvements at four and eight weeks, while those with previous (but not current) treatment did not significantly improve at either time point.
It may be that ginseng works best to ameliorate cancer-related fatigue in patients simultaneously receiving cancer treatment but not in those who have completed treatment. The findings also suggest that patients who have completed treatment may wish to try ginseng for longer than eight weeks to see if it offers any benefit.
Because this study excluded patients with brain cancer, CNS lymphoma, moderate to severe pain, or insomnia and those taking steroids, it is not known if ginseng would help them.
In one study, a low-dose methanolic extract of American ginseng caused a breast cancer cell line to proliferate; however, it was later discovered that this extract had been contaminated with Fusarium fungi containing zearalenone, which has strong estrogenic activity.9,10 However, higher doses of a similar methanolic extract, as well as other water-based extracts, have reduced proliferation of breast cancer cells.11
Proceed carefully if a patient is taking warfarin. Coadministration of ginseng and warfarin may reduce both warfarin concentrations and a patient’s international normalized ratio (INR).12 Therefore, carefully monitor INR in patients concurrently taking ginseng and warfarin. Furthermore, ginseng may lower blood glucose in patients with diabetes, so carefully monitor blood glucose in these patients when initiating or discontinuing ginseng.13
CHALLENGES TO IMPLEMENTATION
It’s hard to know exactly what you’re getting
Regulating dietary supplements, especially verifying ingredients and potency, has been a challenge for the FDA. Although ginseng commonly is adulterated, this is more common with the Asian species (Panax ginseng) than with the American species (Panax quinquefolius) used in this study.10 Clinicians who want to recommend ginseng for cancer-related fatigue should advise patients to use American ginseng root products produced in the US. Additionally, the products should contain at least 3% ginsenosides to match the dose used in this study.
REFERENCES
1. Barton DL, Liu H, Dakhil SR, et al. Wisconsin Ginseng (Panax quinquefolius) to improve cancer-related fatigue: a randomized, double-blind trial, N07C2. J Natl Cancer Inst. 2013;105(16):1230-1238.
2. Hofman M, Ryan JL, Figueroa-Moseley CD, et al. Cancer-related fatigue: the scale of the problem. Oncologist. 2007;12 (suppl 1):4-10.
3. Bower JE, Ganz PA, Desmond KA, et al. Fatigue in long-term breast carcinoma survivors: a longitudinal investigation. Cancer. 2006;106(4):751-758.
4. Moraska AR, Sood A, Dakhil SR, et al. Phase III, randomized, double-blind, placebo-controlled study of long-acting methylphenidate for cancer-related fatigue: North Central Cancer Treatment Group NCCTG-N05C7 trial. J Clin Oncol. 2010;28(23):3673-3679.
5. Bruera E, El Osta B, Valero V, et al. Donepezil for cancer fatigue: a double-blind, randomized, placebo-controlled trial. J Clin Oncol. 2007;25(23):3475-3481.
6. Morrow GR, Hickok JT, Roscoe JA, et al; University of Rochester Cancer Center Community Clinical Oncology Program. Differential effects of paroxetine on fatigue and depression: a randomized, double-blind trial from the University of Rochester Cancer Center Community Clinical Oncology Program. J Clin Oncol. 2003;21(24):4635-4641.
7. Lesser GJ, Case D, Stark N, et al; Wake Forest University Community Clinical Oncology Program Research Base. A randomized, double-blind, placebo-controlled study of oral coenzyme Q10 to relieve self-reported treatment-related fatigue in newly diagnosed patients with breast cancer. J Support Oncol. 2013;11(1):31-42.
8. Cruciani RA, Zhang JJ, Manola J, et al. L-carnitine supplementation for the management of fatigue in patients with cancer: an Eastern cooperative oncology group phase III, randomized, double-blind, placebo-controlled trial. J Clin Oncol. 2012;30(31):3864-3869.
9. Duda RB, Zhong Y, Navas V, et al. American ginseng and breast cancer therapeutic agents synergistically inhibit MCF-7
breast cancer cell growth. J Surg Oncol. 1999;72(4):230-239.
10. Upton R, ed. American ginseng root Panax quinquefolius, standards of analysis, quality control, and therapeutics. Scotts Valley, CA: American Herbal Pharmacopoeia; 2012.
11. King ML, Adler SR, Murphy LL. Extraction-dependent effects of American ginseng (Panax quinquefolium) on human breast cancer cell proliferation and estrogen receptor activation. Integr Cancer Ther. 2006;5(3): 236-243.
12. Yuan CS, Wei G, Dey L, et al. Brief communication: American ginseng reduces warfarin’s effect in healthy patients: a randomized, controlled trial. Ann Intern Med. 2004;141(1):23-27.
13. Vuksan V, Stavro MP, Sievenpiper JL, et al. Similar postprandial glycemic reductions with escalation of dose and administration time of American ginseng in type 2 diabetes. Diabetes Care. 2000;23(9):1221-1226.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Copyright © 2014. The Family Physicians Inquiries Network. All rights reserved.
Reprinted with permission from the Family Physicians Inquiries Network and The Journal of Family Practice. 2014;63(5):270-272.
FDA requiring lower starting dose for another sleep drug, eszopiclone
The recommended starting dose for the insomnia drug eszopiclone has been lowered from 2 mg to 1 mg, to address the risk of next-morning impairment associated with the drug, the Food and Drug Administration announced on May 15.
The manufacturer of the drug, marketed as Lunesta, was required to change the starting dose recommendation because of evidence that levels of the drug "in some patients may be high enough the morning after use to impair activities that require alertness, including driving, even if they feel fully awake," the FDA statement said. The recommendation applies to men and women.
If necessary, the 1-mg dose can be increased to 2 mg or 3 mg, "but the higher doses are more likely to result in next-day impairment of driving and other activities that require full alertness," the statement added. The FDA also is advising patients who are on the 2-mg or 3-mg doses to contact their health care providers for advice on how they can continue the drug safely, and is cautioning patients on the 3-mg dose not to drive or engage in other activities "that require complete mental alertness the day after use."
The recommendations are based partly on the results of a study of 91 healthy adults aged 24-40 years, which compared the effects of the 3-mg dose of eszopiclone and placebo, according to the FDA. The use of the 3-mg dose was associated with severe psychomotor and memory impairments in men and women the next morning, 7.5 hours after taking the medication. At recommended doses, impairments in driving skills, memory, and coordination were evident for up to 11 hours after the drug was taken, and "despite these long-lasting effects, patients were often unaware they were impaired," the statement said.
In January 2013, the FDA recommended reducing the dose for sleep medications containing zolpidem, which include Ambien and Ambien (CR), for the same reason.
The safety communication regarding this drug is available here. Serious adverse events associated with this and other insomnia drugs should be reported to the FDA at 800-332-1088 or at the MedWatch website.
The recommended starting dose for the insomnia drug eszopiclone has been lowered from 2 mg to 1 mg, to address the risk of next-morning impairment associated with the drug, the Food and Drug Administration announced on May 15.
The manufacturer of the drug, marketed as Lunesta, was required to change the starting dose recommendation because of evidence that levels of the drug "in some patients may be high enough the morning after use to impair activities that require alertness, including driving, even if they feel fully awake," the FDA statement said. The recommendation applies to men and women.
If necessary, the 1-mg dose can be increased to 2 mg or 3 mg, "but the higher doses are more likely to result in next-day impairment of driving and other activities that require full alertness," the statement added. The FDA also is advising patients who are on the 2-mg or 3-mg doses to contact their health care providers for advice on how they can continue the drug safely, and is cautioning patients on the 3-mg dose not to drive or engage in other activities "that require complete mental alertness the day after use."
The recommendations are based partly on the results of a study of 91 healthy adults aged 24-40 years, which compared the effects of the 3-mg dose of eszopiclone and placebo, according to the FDA. The use of the 3-mg dose was associated with severe psychomotor and memory impairments in men and women the next morning, 7.5 hours after taking the medication. At recommended doses, impairments in driving skills, memory, and coordination were evident for up to 11 hours after the drug was taken, and "despite these long-lasting effects, patients were often unaware they were impaired," the statement said.
In January 2013, the FDA recommended reducing the dose for sleep medications containing zolpidem, which include Ambien and Ambien (CR), for the same reason.
The safety communication regarding this drug is available here. Serious adverse events associated with this and other insomnia drugs should be reported to the FDA at 800-332-1088 or at the MedWatch website.
The recommended starting dose for the insomnia drug eszopiclone has been lowered from 2 mg to 1 mg, to address the risk of next-morning impairment associated with the drug, the Food and Drug Administration announced on May 15.
The manufacturer of the drug, marketed as Lunesta, was required to change the starting dose recommendation because of evidence that levels of the drug "in some patients may be high enough the morning after use to impair activities that require alertness, including driving, even if they feel fully awake," the FDA statement said. The recommendation applies to men and women.
If necessary, the 1-mg dose can be increased to 2 mg or 3 mg, "but the higher doses are more likely to result in next-day impairment of driving and other activities that require full alertness," the statement added. The FDA also is advising patients who are on the 2-mg or 3-mg doses to contact their health care providers for advice on how they can continue the drug safely, and is cautioning patients on the 3-mg dose not to drive or engage in other activities "that require complete mental alertness the day after use."
The recommendations are based partly on the results of a study of 91 healthy adults aged 24-40 years, which compared the effects of the 3-mg dose of eszopiclone and placebo, according to the FDA. The use of the 3-mg dose was associated with severe psychomotor and memory impairments in men and women the next morning, 7.5 hours after taking the medication. At recommended doses, impairments in driving skills, memory, and coordination were evident for up to 11 hours after the drug was taken, and "despite these long-lasting effects, patients were often unaware they were impaired," the statement said.
In January 2013, the FDA recommended reducing the dose for sleep medications containing zolpidem, which include Ambien and Ambien (CR), for the same reason.
The safety communication regarding this drug is available here. Serious adverse events associated with this and other insomnia drugs should be reported to the FDA at 800-332-1088 or at the MedWatch website.
Commentary: Preemptive planning is key to palliative care
For decades, surgeons have been at the forefront of the palliative care movement. From the historic utilization of palliative operations to relieve suffering to creation of the American College of Surgeons Palliative Care Task Force, surgeons are often first-line palliative care providers in the management of patients with advanced malignancy.
Palliative care involves paying attention to symptom distress, communicating with patients and families about goals of care in relation to prognosis and patient preferences, planning transitions, and engaging family support. Yet, despite a clear and established role, many surgeons are not prepared to effectively provide palliative care, and they are often resistant to the use of specialty palliative care services.
In my own training, I have witnessed apprehension and failure to use palliative care services. In one instance, I was managing an older patient after a complicated sarcoma resection. The patient suffered complications, which kept him hospitalized and returning to the hospital after short periods at a long-term acute care facility. I suggested that we call palliative care for assistance in management of the patient. He had pain, nausea, and poor oral intake, and he was depressed and anxious about his future. I was told "we don’t want the patient believing we were giving up on him."
After 3 weeks of minimal change, my attending relented. In a short time, the patient’s pain and nausea were better controlled, and we were able to have discussions with the patient and his family to clarify goals of care. Some of the symptom management techniques used methods that I had not yet encountered in my training and that seemed foreign and curious. But seeing the response left no question as to their utility. The patient was able to leave the hospital 2 weeks later with the palliative care service coordinating his management with the primary care provider and surgical team. This was a positive resolution to a significant problem. But what really hindered palliative care use?
Roadblocks include the term palliative having a negative connotation, being equated with "failure." Increased use of quality metrics may deincentivize palliative operations. Also, there is poor training and support for surgeons to provide primary palliative care services themselves. Yet, despite these barriers, there are opportunities for surgeons to improve care of patients with advanced malignancy by improving surgeon-patient communication and giving greater emphasis to advanced care planning prior to operative interventions.
As front-line providers for these patients, surgeons are an ideal conduit for delivery and improved use of early palliative care. My practice includes a discussion of advanced care planning with all of my cancer patients. I introduce this as a normal part of every discussion and refer the patient to the primary care provider or our Quality of Life service to facilitate further conversations and documentation. By destigmatizing the discussion for patients and families, a door is opened to an important part of comprehensive quality care. We must understand that diseases progress and complications occur. Failing to provide preemptive support to patients and families is true failure. With a preemptive approach, patients, families, and caregivers have a better understanding of the medical situation, and the latter can more effectively support the patient.
To standardize the role that surgeons routinely play in management of patients with advanced malignancy, efforts must focus on education and research. The role of education is twofold. First, surgical trainees need adequate tools to perform routine palliative care and an understanding of the appropriate timing to refer for specialized services. There have been multiple national efforts focused on teaching palliative care to varied practitioners. But given the aging population and the paucity of specialist palliative care providers, a renewed effort is needed. Second, surgeons must understand the role that palliative care plays and the benefits their patients can derive from it. Surgeons are routinely involved throughout the course of care of patients with malignancy from diagnosis to the end of life. Recognition of palliative care as a skill along the continuum of care already provided will improve outcomes.
Finally, research must focus on both models of use of palliative care and the quality of current practice. Palliative care as it pertains to surgeons is understudied: What teaching formats are most likely to affect clinical practice? What components of palliative care have an impact in surgical practice? What is the optimal timing and venue for providing palliative care in surgical practice? What health care system changes are needed to support surgeons to provide primary palliative care services?
We are uniquely aware of the complexities of care needed for management of patients with advanced malignancies. We are routinely called on to aid in the management of these patients. Surgical educators and researchers should focus their efforts on what is needed to fully integrate palliative care into patient-centered care already provided by trainees and surgeons.
Dr. Johnston is an assistant professor of surgery in the division of surgical oncology at the Medical College of Wisconsin, Milwaukee. He disclosed no conflicts.
For decades, surgeons have been at the forefront of the palliative care movement. From the historic utilization of palliative operations to relieve suffering to creation of the American College of Surgeons Palliative Care Task Force, surgeons are often first-line palliative care providers in the management of patients with advanced malignancy.
Palliative care involves paying attention to symptom distress, communicating with patients and families about goals of care in relation to prognosis and patient preferences, planning transitions, and engaging family support. Yet, despite a clear and established role, many surgeons are not prepared to effectively provide palliative care, and they are often resistant to the use of specialty palliative care services.
In my own training, I have witnessed apprehension and failure to use palliative care services. In one instance, I was managing an older patient after a complicated sarcoma resection. The patient suffered complications, which kept him hospitalized and returning to the hospital after short periods at a long-term acute care facility. I suggested that we call palliative care for assistance in management of the patient. He had pain, nausea, and poor oral intake, and he was depressed and anxious about his future. I was told "we don’t want the patient believing we were giving up on him."
After 3 weeks of minimal change, my attending relented. In a short time, the patient’s pain and nausea were better controlled, and we were able to have discussions with the patient and his family to clarify goals of care. Some of the symptom management techniques used methods that I had not yet encountered in my training and that seemed foreign and curious. But seeing the response left no question as to their utility. The patient was able to leave the hospital 2 weeks later with the palliative care service coordinating his management with the primary care provider and surgical team. This was a positive resolution to a significant problem. But what really hindered palliative care use?
Roadblocks include the term palliative having a negative connotation, being equated with "failure." Increased use of quality metrics may deincentivize palliative operations. Also, there is poor training and support for surgeons to provide primary palliative care services themselves. Yet, despite these barriers, there are opportunities for surgeons to improve care of patients with advanced malignancy by improving surgeon-patient communication and giving greater emphasis to advanced care planning prior to operative interventions.
As front-line providers for these patients, surgeons are an ideal conduit for delivery and improved use of early palliative care. My practice includes a discussion of advanced care planning with all of my cancer patients. I introduce this as a normal part of every discussion and refer the patient to the primary care provider or our Quality of Life service to facilitate further conversations and documentation. By destigmatizing the discussion for patients and families, a door is opened to an important part of comprehensive quality care. We must understand that diseases progress and complications occur. Failing to provide preemptive support to patients and families is true failure. With a preemptive approach, patients, families, and caregivers have a better understanding of the medical situation, and the latter can more effectively support the patient.
To standardize the role that surgeons routinely play in management of patients with advanced malignancy, efforts must focus on education and research. The role of education is twofold. First, surgical trainees need adequate tools to perform routine palliative care and an understanding of the appropriate timing to refer for specialized services. There have been multiple national efforts focused on teaching palliative care to varied practitioners. But given the aging population and the paucity of specialist palliative care providers, a renewed effort is needed. Second, surgeons must understand the role that palliative care plays and the benefits their patients can derive from it. Surgeons are routinely involved throughout the course of care of patients with malignancy from diagnosis to the end of life. Recognition of palliative care as a skill along the continuum of care already provided will improve outcomes.
Finally, research must focus on both models of use of palliative care and the quality of current practice. Palliative care as it pertains to surgeons is understudied: What teaching formats are most likely to affect clinical practice? What components of palliative care have an impact in surgical practice? What is the optimal timing and venue for providing palliative care in surgical practice? What health care system changes are needed to support surgeons to provide primary palliative care services?
We are uniquely aware of the complexities of care needed for management of patients with advanced malignancies. We are routinely called on to aid in the management of these patients. Surgical educators and researchers should focus their efforts on what is needed to fully integrate palliative care into patient-centered care already provided by trainees and surgeons.
Dr. Johnston is an assistant professor of surgery in the division of surgical oncology at the Medical College of Wisconsin, Milwaukee. He disclosed no conflicts.
For decades, surgeons have been at the forefront of the palliative care movement. From the historic utilization of palliative operations to relieve suffering to creation of the American College of Surgeons Palliative Care Task Force, surgeons are often first-line palliative care providers in the management of patients with advanced malignancy.
Palliative care involves paying attention to symptom distress, communicating with patients and families about goals of care in relation to prognosis and patient preferences, planning transitions, and engaging family support. Yet, despite a clear and established role, many surgeons are not prepared to effectively provide palliative care, and they are often resistant to the use of specialty palliative care services.
In my own training, I have witnessed apprehension and failure to use palliative care services. In one instance, I was managing an older patient after a complicated sarcoma resection. The patient suffered complications, which kept him hospitalized and returning to the hospital after short periods at a long-term acute care facility. I suggested that we call palliative care for assistance in management of the patient. He had pain, nausea, and poor oral intake, and he was depressed and anxious about his future. I was told "we don’t want the patient believing we were giving up on him."
After 3 weeks of minimal change, my attending relented. In a short time, the patient’s pain and nausea were better controlled, and we were able to have discussions with the patient and his family to clarify goals of care. Some of the symptom management techniques used methods that I had not yet encountered in my training and that seemed foreign and curious. But seeing the response left no question as to their utility. The patient was able to leave the hospital 2 weeks later with the palliative care service coordinating his management with the primary care provider and surgical team. This was a positive resolution to a significant problem. But what really hindered palliative care use?
Roadblocks include the term palliative having a negative connotation, being equated with "failure." Increased use of quality metrics may deincentivize palliative operations. Also, there is poor training and support for surgeons to provide primary palliative care services themselves. Yet, despite these barriers, there are opportunities for surgeons to improve care of patients with advanced malignancy by improving surgeon-patient communication and giving greater emphasis to advanced care planning prior to operative interventions.
As front-line providers for these patients, surgeons are an ideal conduit for delivery and improved use of early palliative care. My practice includes a discussion of advanced care planning with all of my cancer patients. I introduce this as a normal part of every discussion and refer the patient to the primary care provider or our Quality of Life service to facilitate further conversations and documentation. By destigmatizing the discussion for patients and families, a door is opened to an important part of comprehensive quality care. We must understand that diseases progress and complications occur. Failing to provide preemptive support to patients and families is true failure. With a preemptive approach, patients, families, and caregivers have a better understanding of the medical situation, and the latter can more effectively support the patient.
To standardize the role that surgeons routinely play in management of patients with advanced malignancy, efforts must focus on education and research. The role of education is twofold. First, surgical trainees need adequate tools to perform routine palliative care and an understanding of the appropriate timing to refer for specialized services. There have been multiple national efforts focused on teaching palliative care to varied practitioners. But given the aging population and the paucity of specialist palliative care providers, a renewed effort is needed. Second, surgeons must understand the role that palliative care plays and the benefits their patients can derive from it. Surgeons are routinely involved throughout the course of care of patients with malignancy from diagnosis to the end of life. Recognition of palliative care as a skill along the continuum of care already provided will improve outcomes.
Finally, research must focus on both models of use of palliative care and the quality of current practice. Palliative care as it pertains to surgeons is understudied: What teaching formats are most likely to affect clinical practice? What components of palliative care have an impact in surgical practice? What is the optimal timing and venue for providing palliative care in surgical practice? What health care system changes are needed to support surgeons to provide primary palliative care services?
We are uniquely aware of the complexities of care needed for management of patients with advanced malignancies. We are routinely called on to aid in the management of these patients. Surgical educators and researchers should focus their efforts on what is needed to fully integrate palliative care into patient-centered care already provided by trainees and surgeons.
Dr. Johnston is an assistant professor of surgery in the division of surgical oncology at the Medical College of Wisconsin, Milwaukee. He disclosed no conflicts.
Some providers quicker to tube feed end-of-life elderly
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).
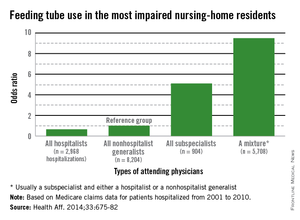
"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Dr. Daniel Ouellette, FCCP, comments: The Choosing Wisely campaign sponsored by Consumer Reports and the American Board of Internal Medicine Foundation provides recommendations to help patients and providers make wise decisions about appropriate care. Both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society target the inappropriate use of feeding tubes in persons at the end of life, suggesting that such treatments do not improve patient outcomes. The news that subspecialists and primary care physicians order feeding tubes much more frequently than hospitalists is sobering information for pulmonary and critical care physicians taking care of such patients. However, future investigations focused on nutritional support and involving the patient population that pulmonologists frequently manage is likely to be complicated by the facts that such patients are or have been critically ill, and are likely to implicate a broader array of end-of-life decision making than just the provision of feeding.
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).

"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Subspecialists who care for dementia patients near the end of life are far more likely to introduce a feeding tube than hospitalists who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube feed hospitalized nursing-home residents - and even less likely to tube feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
The findings clearly illustrate that many physicians could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (2014;33:675-82).

"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Dr. Eric G. Tangalos agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Who's the attending?
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients' mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient's care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding - almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The fragmention factor
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion.
"One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person.
"Organ- and specialty-specific decision making leads to bad practices - including trying to 'solve' a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure - a terminal illness that cannot be fixed with a feeding tube."
The study questions not only the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life - especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing-home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing-home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing-home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
FROM HEALTH AFFAIRS
Major finding: Primary care doctors and subspecialists were up
to 9 times more likely to initiate tube feeding in hospitalized
patients with severe dementia.
Data source: Data are from a review of more than 53,000 Medicare fee-for-service nursing home patients.
Disclosures: The National Institute on Aging sponsored the study. Dr. Teno had no financial disclosures.
Mergers and acquisitions pose legal challenges for physician-sellers
Practice mergers with physician groups and larger health systems are becoming commonplace as more doctors trade in their shingles for fewer business burdens and more stability. But as with all transactions, selling or merging a medical practice comes with legal risks.
"There are numerous complex issues, legal and otherwise, that the seller of a medical practice needs to consider, both in preparations for the sale and during the transaction," wrote health law attorney David N. Vozza in a recent article for Kern Augustine Conroy & Schoppmann, a health care litigation firm with offices in the East Coast.
One of the most significant legal considerations pertains to the correct transfer of medical records during sales and mergers, Mr. Vozza said in an interview. To meet privacy requirements, all current patients must be advised that the practice is being transferred and they must have the opportunity to obtain their original records if they desire a new physician, he said. Records also must not be released to third parties without the patient’s express authorization. In addition, physicians run the risk of an "abandonment" lawsuit, if patient care is compromised because of the sale or merger.
"If you know a patient has a pressing care or treatment need, that can’t be delayed because you’re in the middle of a sale," Mr. Vozza said. "It can’t get lost in the shuffle of the transaction."
Another key consideration when merging practices is the assessment of liability cases, said Mathew J. Levy, a health law attorney and principal at Kern Augustine Conroy & Schoppmann and a coauthor of the risk management article. Physicians combining practices should be aware of any pending malpractice cases or audits of their potential partners.
"When merging with a larger group, you should be concerned with how they are doing internally," Mr. Levy said. "Are they under any audits or investigations? Have they been disciplined by the state licensure board? You have to make sure you’re not accountable for" their debts or malpractice issues.
Physicians should also consider the effect their merger may have on their malpractice insurance, Mr. Levy adds. Coverage for prior actions can become challenging in the event that a physician has a claims-made policy, which offers protection only while the policy is in effect. If a claims-made policy is discontinued, the doctor must obtain "tail" coverage to cover past actions. While the physician likely will have new coverage from the hospital or health system, many hospitals are self-insured and do not provide incoming physicians with prior act or so-called nose coverage.
Along with discussing tail coverage during a merger, doctors should also be wary of "anti-compete" clauses in contracts with larger hospitals and health systems. Such clauses prohibit physicians from working for a competitor and/or in close proximity to the hospital. Mr. Levy encourages physicians to include an exception to the non-compete clause in their contracts and discuss options in case the relationship fails.
"If not, you would be stuck and be prohibited from working in a nearby location and generating patients for your career," he said. "That’s a significant issue."
Antitrust requirements should also be high on physicians’ radar long before a transaction proceeds.
The Federal Trade Commission in January revised the thresholds that determine whether health care providers must notify federal antitrust authorities about pending transactions under the Hart-Scott-Rodino (HSR) Antitrust Improvements Act. The HSR Act requires companies to notify government agencies if the size of the parties at issue and the value of a transaction exceed the filing thresholds. The 2014 FTC revision raised the threshold for reporting proposed mergers and acquisitions from $70.9 million to $75.9 million.
"If notification is required and "you proceed to complete a transaction without filing the required notification, than you’re in violation of the antitrust law," said Christine White, chair of the American Health Lawyers Association’s Antitrust Practice Group and a staff attorney in the FTC’s Northeast Regional Office.
Potential antitrust violations can result in government investigations, fines, legal settlements, or other discipline. Ms. White suggests that physicians review guidance on the FTC’s website for more information about the HSR Act and other antitrust requirements, such as when it’s acceptable to share confidential information with competitors.
"Not every sale of a physician practice will raise significant antitrust concerns. In fact, the vast majority of physician practice group consolidations do not raise major antitrust concerns. But, in certain circumstances, such as if a practice group is selling to a direct competitor or a potential competitor, the antitrust concerns may merit serious consideration," she said.
Practice mergers with physician groups and larger health systems are becoming commonplace as more doctors trade in their shingles for fewer business burdens and more stability. But as with all transactions, selling or merging a medical practice comes with legal risks.
"There are numerous complex issues, legal and otherwise, that the seller of a medical practice needs to consider, both in preparations for the sale and during the transaction," wrote health law attorney David N. Vozza in a recent article for Kern Augustine Conroy & Schoppmann, a health care litigation firm with offices in the East Coast.
One of the most significant legal considerations pertains to the correct transfer of medical records during sales and mergers, Mr. Vozza said in an interview. To meet privacy requirements, all current patients must be advised that the practice is being transferred and they must have the opportunity to obtain their original records if they desire a new physician, he said. Records also must not be released to third parties without the patient’s express authorization. In addition, physicians run the risk of an "abandonment" lawsuit, if patient care is compromised because of the sale or merger.
"If you know a patient has a pressing care or treatment need, that can’t be delayed because you’re in the middle of a sale," Mr. Vozza said. "It can’t get lost in the shuffle of the transaction."
Another key consideration when merging practices is the assessment of liability cases, said Mathew J. Levy, a health law attorney and principal at Kern Augustine Conroy & Schoppmann and a coauthor of the risk management article. Physicians combining practices should be aware of any pending malpractice cases or audits of their potential partners.
"When merging with a larger group, you should be concerned with how they are doing internally," Mr. Levy said. "Are they under any audits or investigations? Have they been disciplined by the state licensure board? You have to make sure you’re not accountable for" their debts or malpractice issues.
Physicians should also consider the effect their merger may have on their malpractice insurance, Mr. Levy adds. Coverage for prior actions can become challenging in the event that a physician has a claims-made policy, which offers protection only while the policy is in effect. If a claims-made policy is discontinued, the doctor must obtain "tail" coverage to cover past actions. While the physician likely will have new coverage from the hospital or health system, many hospitals are self-insured and do not provide incoming physicians with prior act or so-called nose coverage.
Along with discussing tail coverage during a merger, doctors should also be wary of "anti-compete" clauses in contracts with larger hospitals and health systems. Such clauses prohibit physicians from working for a competitor and/or in close proximity to the hospital. Mr. Levy encourages physicians to include an exception to the non-compete clause in their contracts and discuss options in case the relationship fails.
"If not, you would be stuck and be prohibited from working in a nearby location and generating patients for your career," he said. "That’s a significant issue."
Antitrust requirements should also be high on physicians’ radar long before a transaction proceeds.
The Federal Trade Commission in January revised the thresholds that determine whether health care providers must notify federal antitrust authorities about pending transactions under the Hart-Scott-Rodino (HSR) Antitrust Improvements Act. The HSR Act requires companies to notify government agencies if the size of the parties at issue and the value of a transaction exceed the filing thresholds. The 2014 FTC revision raised the threshold for reporting proposed mergers and acquisitions from $70.9 million to $75.9 million.
"If notification is required and "you proceed to complete a transaction without filing the required notification, than you’re in violation of the antitrust law," said Christine White, chair of the American Health Lawyers Association’s Antitrust Practice Group and a staff attorney in the FTC’s Northeast Regional Office.
Potential antitrust violations can result in government investigations, fines, legal settlements, or other discipline. Ms. White suggests that physicians review guidance on the FTC’s website for more information about the HSR Act and other antitrust requirements, such as when it’s acceptable to share confidential information with competitors.
"Not every sale of a physician practice will raise significant antitrust concerns. In fact, the vast majority of physician practice group consolidations do not raise major antitrust concerns. But, in certain circumstances, such as if a practice group is selling to a direct competitor or a potential competitor, the antitrust concerns may merit serious consideration," she said.
Practice mergers with physician groups and larger health systems are becoming commonplace as more doctors trade in their shingles for fewer business burdens and more stability. But as with all transactions, selling or merging a medical practice comes with legal risks.
"There are numerous complex issues, legal and otherwise, that the seller of a medical practice needs to consider, both in preparations for the sale and during the transaction," wrote health law attorney David N. Vozza in a recent article for Kern Augustine Conroy & Schoppmann, a health care litigation firm with offices in the East Coast.
One of the most significant legal considerations pertains to the correct transfer of medical records during sales and mergers, Mr. Vozza said in an interview. To meet privacy requirements, all current patients must be advised that the practice is being transferred and they must have the opportunity to obtain their original records if they desire a new physician, he said. Records also must not be released to third parties without the patient’s express authorization. In addition, physicians run the risk of an "abandonment" lawsuit, if patient care is compromised because of the sale or merger.
"If you know a patient has a pressing care or treatment need, that can’t be delayed because you’re in the middle of a sale," Mr. Vozza said. "It can’t get lost in the shuffle of the transaction."
Another key consideration when merging practices is the assessment of liability cases, said Mathew J. Levy, a health law attorney and principal at Kern Augustine Conroy & Schoppmann and a coauthor of the risk management article. Physicians combining practices should be aware of any pending malpractice cases or audits of their potential partners.
"When merging with a larger group, you should be concerned with how they are doing internally," Mr. Levy said. "Are they under any audits or investigations? Have they been disciplined by the state licensure board? You have to make sure you’re not accountable for" their debts or malpractice issues.
Physicians should also consider the effect their merger may have on their malpractice insurance, Mr. Levy adds. Coverage for prior actions can become challenging in the event that a physician has a claims-made policy, which offers protection only while the policy is in effect. If a claims-made policy is discontinued, the doctor must obtain "tail" coverage to cover past actions. While the physician likely will have new coverage from the hospital or health system, many hospitals are self-insured and do not provide incoming physicians with prior act or so-called nose coverage.
Along with discussing tail coverage during a merger, doctors should also be wary of "anti-compete" clauses in contracts with larger hospitals and health systems. Such clauses prohibit physicians from working for a competitor and/or in close proximity to the hospital. Mr. Levy encourages physicians to include an exception to the non-compete clause in their contracts and discuss options in case the relationship fails.
"If not, you would be stuck and be prohibited from working in a nearby location and generating patients for your career," he said. "That’s a significant issue."
Antitrust requirements should also be high on physicians’ radar long before a transaction proceeds.
The Federal Trade Commission in January revised the thresholds that determine whether health care providers must notify federal antitrust authorities about pending transactions under the Hart-Scott-Rodino (HSR) Antitrust Improvements Act. The HSR Act requires companies to notify government agencies if the size of the parties at issue and the value of a transaction exceed the filing thresholds. The 2014 FTC revision raised the threshold for reporting proposed mergers and acquisitions from $70.9 million to $75.9 million.
"If notification is required and "you proceed to complete a transaction without filing the required notification, than you’re in violation of the antitrust law," said Christine White, chair of the American Health Lawyers Association’s Antitrust Practice Group and a staff attorney in the FTC’s Northeast Regional Office.
Potential antitrust violations can result in government investigations, fines, legal settlements, or other discipline. Ms. White suggests that physicians review guidance on the FTC’s website for more information about the HSR Act and other antitrust requirements, such as when it’s acceptable to share confidential information with competitors.
"Not every sale of a physician practice will raise significant antitrust concerns. In fact, the vast majority of physician practice group consolidations do not raise major antitrust concerns. But, in certain circumstances, such as if a practice group is selling to a direct competitor or a potential competitor, the antitrust concerns may merit serious consideration," she said.
Some providers quicker to tube feed end-of-life elderly
Hospitalists who care for dementia patients near the end of life are much less likely to introduce a feeding tube than other physicians who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube-feed hospitalized nursing home residents – and even less likely to tube-feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.
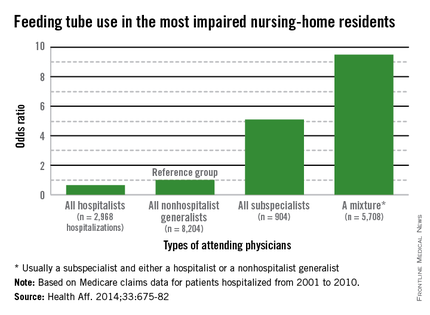
The findings clearly illustrate that nonhospitalists could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (Health Aff. April 2014;33:675-82).
"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."
Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Internal medicine physician Eric G. Tangalos of the Alzheimer’s Disease Research Center at Mayo Clinic, Rochester, Minn., works closely with hospitalists. He agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients’ mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient’s care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding – almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion. "One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person. Organ- and specialty-specific decision making leads to bad practices – including trying to ‘solve’ a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure – a terminal illness that cannot be fixed with a feeding tube."
The study not only questions the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life – especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Hospitalists who care for dementia patients near the end of life are much less likely to introduce a feeding tube than other physicians who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube-feed hospitalized nursing home residents – and even less likely to tube-feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.

The findings clearly illustrate that nonhospitalists could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (Health Aff. April 2014;33:675-82).
"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."
Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Internal medicine physician Eric G. Tangalos of the Alzheimer’s Disease Research Center at Mayo Clinic, Rochester, Minn., works closely with hospitalists. He agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients’ mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient’s care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding – almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion. "One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person. Organ- and specialty-specific decision making leads to bad practices – including trying to ‘solve’ a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure – a terminal illness that cannot be fixed with a feeding tube."
The study not only questions the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life – especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
Hospitalists who care for dementia patients near the end of life are much less likely to introduce a feeding tube than other physicians who follow such patients.
Compared with nonhospital generalists, hospitalists were 22% less likely to tube-feed hospitalized nursing home residents – and even less likely to tube-feed patients who were the most severely impaired (35%). In contrast, subspecialists were five times more likely to insert a tube. When a mixed group of physicians was on the case, rates were even higher, with a 9-fold increase overall and a 9.5-fold increase for severely demented patients.

The findings clearly illustrate that nonhospitalists could benefit from some education about the most appropriate interventions when patients near the end of life enter a hospital, Dr. Joan Teno and her associates reported in the April issue of Health Affairs (Health Aff. April 2014;33:675-82).
"It may be that subspecialists do not have adequate knowledge about the risks and benefits of using feeding tubes in people with advanced dementia," said Dr. Teno of Brown University, Providence, R.I., and her coauthors. "Hospitals should educate physicians about the lack of efficacy of PEG [percutaneous endoscopic gastrostomy] feeding tubes, compared with hand feeding, in prolonging survival and preventing aspiration pneumonias and pressure ulcers in people with advanced dementia. In addition, hospitals should examine how they staff the role of attending physician and ensure coordination of care when patient hand-offs are made between different types of attending physicians."
Such education would bring all physicians up to speed with position statements against tube feeding for this group of patients. The issue sits atop the Choosing Wisely lists of both the American Academy of Hospice and Palliative Medicine and the American Geriatrics Society. The American Academy of Hospice and Palliative Medicine states that "feeding tubes do not result in improved survival, prevention of aspiration pneumonia, or improved healing of pressure ulcers. Feeding tube use in such patients has actually been associated with pressure ulcer development, use of physical and pharmacological restraints, and patient distress about the tube itself."
Internal medicine physician Eric G. Tangalos of the Alzheimer’s Disease Research Center at Mayo Clinic, Rochester, Minn., works closely with hospitalists. He agrees with the concept that tube feeding can impose even more distress on both these patients and their families. "As a medical profession and a society, we have yet to accept some of the futility of our actions and continue to ignore the burdens tube feedings place on patients, families, and the health care system once a hospitalization has come to its conclusion," he said in an interview.
Dr. Teno and her team looked at the rate of feeding tube insertion in fee-for-service Medicare patients with advanced dementia who were within 90 days of death and hospitalized with a diagnosis of urinary tract infection, sepsis, pneumonia, or dehydration. The study examined decisions made by four groups of physicians who cared for these patients: hospitalists, nonhospitalist generalists (geriatricians, general practitioners, internists, and family physicians), subspecialists, and mixed groups that included a subspecialist and either a hospitalist or nonhospitalist generalist.
The cohort comprised 53,492 patients hospitalized from 2001 to 2010. The patients’ mean age was 85 years. About 60% had a do not resuscitate order, and 10% had an order against tube feeding.
The rate of hospitalists as attending physicians increased from 11% in 2001 to 28% in 2010. The portion of patients seen by a mixture of attending physicians increased from 29% in 2001 to 38% in 2010.
The rates of tube feeding were lowest when a hospitalist or nonhospitalist generalist was the attending physician (1.6% and 2.2%, respectively). Subspecialists had significantly higher rates (11%). The highest rate occurred when there were mixed groups of physicians involved in the patient’s care (15.6%).
Using the nonhospitalist generalists as a reference group, the researchers found that hospitalists were 22% less likely to insert a tube overall and 35% less likely to do so when the patient had very severe cognitive and physical impairment.
Conversely, subspecialists were five times more likely to commence tube feeding for all patients and for very severely impaired patients. The mixed groups were the most likely to begin tube feeding – almost 9 times more likely overall and 9.5 times more likely for the most severely impaired patients.
"Our finding that subspecialists had a higher rate of insertions of PEG feeding tubes might reflect their lack of experience in providing care for people with advanced dementia," the authors wrote.
The mixed-physician group could be seen as a proxy for discontinuity of care among the attending physicians, they noted. Prior studies have found that such discontinuity was associated with longer hospital stays.
"There may be a lack of care coordination during patient hand offs between attending physicians that begins a cascade of events, ending with the insertion of a PEG feeding tube."
Dr. Diane E. Meier, professor of geriatrics and palliative medicine at Icahn School of Medicine at Mount Sinai, New York, and director of the Center to Advance Palliative Care, agreed that group care without a leader creates confusion. "One of the hallmarks of modern medicine in the U.S. is fragmentation. It is typical for a person with dementia to have a different specialist for every organ system, a problem compounded in the hospital when a completely new group of specialists is brought into the care team. The problem with this abundance of doctors is that no one is really in charge of the whole patient and what makes the most sense for the patient as a person. Organ- and specialty-specific decision making leads to bad practices – including trying to ‘solve’ a feeding difficulty as if it is an isolated problem when the real issue is progressive brain failure – a terminal illness that cannot be fixed with a feeding tube."
The study not only questions the feeding tube issue, but also the wisdom of repeatedly hospitalizing elderly patients with severe dementia who could be in the last phase of life – especially for conditions that are expected complications of severe dementia. The authors suggested that there may be financial motives to admit fee-for-service patients.
"The fee-for-service system provides incentives to hospitalize nursing home residents with severe dementia because such hospitalizations qualify the patients for skilled nursing home services. Bundling of payments and institutional special needs plans that reverse these financial incentives may reduce health care expenditures and improve the quality of care for nursing home residents with advanced dementia by avoiding burdensome transitions between facilities and the stress of relocation."
The National Institute on Aging funded the study. Dr. Teno made no financial declarations.
FROM HEALTH AFFAIRS
Major finding: Primary care doctors and subspecialists were up to 9 times more likely to initiate tube feeding in hospitalized patients with severe dementia.
Data source: Data are from a review of more than 53,000 Medicare fee-for-service nursing home patients.
Disclosures: The National Institute on Aging sponsored the study. Dr. Teno had no financial disclosures.
Negotiation: Priceless in good communication
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
Dr. Eric Gartman, FCCP, comments: We all have seen these discussions go well and been impressed by those who lead them. However, too often such conversations and family meetings are not actively pursued simply because they are "hard" - they take time and an investment of one's emotional energy. We should follow the example of many medical schools and training programs in recognizing the immense importance of gaining these skills, and foster the desire to be the one that others aim to emulate.
Dr. Eric Gartman, FCCP, comments: We all have seen these discussions go well and been impressed by those who lead them. However, too often such conversations and family meetings are not actively pursued simply because they are "hard" - they take time and an investment of one's emotional energy. We should follow the example of many medical schools and training programs in recognizing the immense importance of gaining these skills, and foster the desire to be the one that others aim to emulate.
Dr. Eric Gartman, FCCP, comments: We all have seen these discussions go well and been impressed by those who lead them. However, too often such conversations and family meetings are not actively pursued simply because they are "hard" - they take time and an investment of one's emotional energy. We should follow the example of many medical schools and training programs in recognizing the immense importance of gaining these skills, and foster the desire to be the one that others aim to emulate.
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.