User login
Negotiation: Priceless in good communication
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
The Society of Hospital Medicine held its annual meeting recently in Las Vegas, and Stephen had the opportunity to speak on the topic of "Family Meetings: The Art and the Evidence." As a special edition of Palliatively Speaking, we thought we would highlight one aspect of this subject, with other elements forthcoming in future pieces.
As a hospitalist, I stumbled and stuttered through many family meetings until I eventually found myself on more comfortable ground. Overall, I found them rewarding when they went well but stressful and deflating when they did not. The latter sensation was enough to create some avoidant behavior on my part.
After a few years of practice, my hospitalist group began shadowing one another periodically on rounds to provide feedback to our colleagues in the hope of improving the quality of our communication skills. It was then that I noticed that one of my partners was a master at these meetings. A real Rembrandt. He had the ability to deliver bad or difficult news without the dynamic in the room becoming inflammatory or out of control.
I will never forget watching him mediate a disagreement between a nurse and a patient suspected of using illicit substances while hospitalized. He flipped an antagonistic, heated situation into one where the patient, nurse, and physician all agreed on putting the past to rest and forging ahead with his proposed plan. We all left the room with a genuine sense that we had mutual purpose. In my admiration I realized that some of these skills must be teachable.
While I didn’t act on learning those communication techniques immediately after that encounter, I would eventually be formally exposed to them during my palliative medicine training. As it turns out, I still have some uncomfortable meetings with patients and families, but they come around much less frequently and when they do I now have a variety of tools to deal with challenges.
My appreciation of these tools doesn’t stop when I walk through the hospital doors each evening. I have found them to be invaluable in my personal life. In fact, learning to communicate better has been a source of renewal for me at work and staves off burnout. These techniques include active listening, motivational interviewing, demonstration of empathy, conflict resolution, and also negotiation. For the Society of Hospital Medicine meeting audience, I dissected negotiation, citing how it and the other skills can inject vitality into your interactions.
In any negotiation, it’s all about the other party. You are the smallest person in the room, the least important.
This is counterintuitive. Oftentimes at work we are trying to convince everyone how important we are. The readmissions committee should implement your plan to reduce recurrent hospitalizations. Your fellow hospitalists should recognize your value and make you the leader of the group. Patients show their appreciation for you making the right diagnosis and averting a medical calamity for them. But when you enter a family meeting, the patient and his or her loved ones are the center stage. To be successful you have to listen more and talk less. Get to understand the pictures in their heads and then summarize those thoughts and ideas back to them to show you’ve listened.
Make emotional payments. I don’t get into the meat of the meeting until I’ve done that with the patient and every family member in the room. No one holds family meetings for patients who are thriving and have outstanding outcomes. We have family meetings to figure out goals in the face of terrible diseases, when elder abuse is a possibility, when insurance-funded resources are depleted, and for a host of other difficult reasons.
This means that everyone in the room is suffering, sacrificing, scared, confused, or worried. Acknowledge them. Hold them up. Thank them. Reflect on similar moments in your life and demonstrate empathy. Apologize when things haven’t gone right for them at your hospital. These payments will pay handsome dividends as your relationship evolves.
Not manipulation. The term negotiation might bring up images of used car salespeople. I strongly disagree. In manipulation, one side wins and the other doesn’t. In negotiation, the goal is improved communication and understanding. Manipulation is about one side of the equation having knowledge that the other side is lacking and using that to achieve its means. Negotiators hope everyone at the table has the same knowledge.
This leads to two key principles of negotiations: transparency and genuineness. Patients and families are excellent at taking the temperature of the room when you sit down to meet with them. Share knowledge. Don’t have any hidden agendas. Following this principle builds trust.
Be incremental. Taking patients from comfortable, familiar territory into that which is uncomfortable or unfamiliar should not be done in one giant leap. Let’s use code status (CS) as an example because of the frequency with which it comes up (though I rarely talk about CS without first understanding the patient’s goals and hopes).
Some patients refuse to talk about CS, so I think incrementally. I ask that they consider talking about CS with me in the future. Very few people refuse to consider something. Two or three days later I ask, "Have you considered talking to me about CS?" That by itself opens up the topic for conversation. In the extremely unusual case where they still won’t engage, I then ask them, "What would it take for you to consider talking to me about this?" More incrementalism.
While this is not nearly an exhaustive list of negotiation techniques, we hope it is stimulating enough that you might be curious enough to learn more on your own and try incorporating this into your practice. If you’re motivated to do so, please feel free to contact us for reading suggestions: E-mail [email protected].
Dr. Bekanich and Dr. Fredholm are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
Palliative care consultations improve outcomes, but referrals lag
TAMPA – Outpatient palliative care consultations are associated with decreased symptom burden in women with gynecologic malignancies, but American Society of Clinical Oncology recommendations for referral are often ignored, according to retrospective data and a review of patient records.
In one study, 78 patients seen between June 2007 and March 2013 at an outpatient symptom management clinic for follow-up within 90 days of their initial consultation completed a questionnaire at each visit, including the nine-item Edmonton Symptom Assessment System. The responses, along with information from the patients’ charts, showed significant improvements in almost all symptoms over time, Dr. Rachel Ruskin, a clinical fellow at the University of California, San Francisco, reported at the annual meeting of the Society of Gynecologic Oncology.
For example, mean pain, fatigue, anxiety, depression, nausea, drowsiness, and appetite scores decreased between 0.7 and 1.5 points from the median baseline scores (on a 10 point scale). A decline in shortness of breath score also approached significance.
No difference was seen with respect to symptom improvement between patients with and without disease, although there appeared to be a trend toward a difference in anxiety scores, Dr. Ruskin noted.
Patients who were treated with concurrent cancer-directed therapies had improvements in pain and fatigue, but to a lesser extent than did those who did not receive treatment, she said.
Among the 35 patients who attended at least two follow-up visits, the improvements in nausea and shortness of breath seen at the first visit persisted at the second, and symptoms of depression and drowsiness continued to improve at each visit. Of those 35 patients, 58% had ovarian, fallopian tube, or peritoneal cancer; 20% had uterine cancer; and 15% had cervical cancer. Most (81%) had stage III, IV, or recurrent cancer.
Mean age at study entry was 57 years, 85% of patients had disease present, and 62% were undergoing treatment. The vast majority (87%) had received chemotherapy, 30% received radiation, and 8% had undergone surgery.
In patients for whom relevant data were available, there was evidence of mild hematologic, renal, and nutritional compromise, and nearly 25% of these patients had been hospitalized within the prior month.
"Notably, in our cohort the improvement in symptoms cannot be attributed to antineoplastic therapies, since – if anything – treatment by traditional oncologic modalities was associated with less benefit in some symptoms," Dr. Ruskin said, adding that future research should focus on "which aspects of palliative care are effective and by what mechanisms," as this information would be helpful for determining best practices that can be replicated across settings and for designing prospective concurrent standard oncologic and palliative care trials.
"In the meantime, we hope that these data will encourage providers to consider referral to their outpatient palliative care colleagues," she concluded.
Findings from another study presented at the meeting suggest there is some work to do in that regard.
In that study, Dr. Carolyn Lefkowits of the University of Pittsburgh found that oncologists are falling short when it comes to following the 2012 ASCO recommendation to consider early palliative care integration for "any patient with metastatic cancer and/or high symptom burden."
Of 340 women with a gynecologic malignancy who were admitted to a gynecologic oncology service between February 2012 and August 2012, only 32% were referred to palliative care, Dr. Lefkowits said.
The patients had a median age of 62 years, and an equal number had early- and late-stage disease. Nearly 25% had recurrent disease by the end of the study period.
Multivariate logistic regression identified independent predictors of palliative care consultation, including number of admission (odds ratio, 17.4 for greater than three vs. three or fewer admissions), admission for symptom management (OR, 22.0), and death within 6 months (OR, 15.7).
Notably, only 16% of the patients died within 6 months of the last admission during the study period, and although 25% of patients overall were referred to palliative care, 54% of those who died within 6 months were referred, suggesting that most referrals are not made for patients who are early in their disease course.
Furthermore, only 53% of patients with recurrent disease – all of whom should have been considered for palliative care integration based on the ASCO recommendations – were seen for palliative care, including 59% who had received three or more lines of chemotherapy, Dr. Lefkowits said.
The findings suggest that the group of patients referred for palliative care is characterized by high symptom burden and poor prognosis. In fact, most of those referred were likely already at the point where they would be considered hospice eligible, she said.
An analysis of referrals based on each ASCO recommendation category showed that the highest referral rate was for "symptom admission" (79%), and the lowest was for recurrent disease (52%).
"So, although the predictors of consultation are in keeping, I think, with the spirit of the ASCO recommendations, we’re still not comprehensively capturing these high-risk subgroups," Dr. Lefkowits said.
She added that she hopes the findings will serve as a "conversation starter, spurring us to address questions, including which gynecologic oncology patients are most appropriate to target for consistent palliative care referral."
Other questions to consider include which systems might help improve referral rates among those patients and which patients (and at what rates) should be referred for specialized palliative care.
All of the patients in the ASCO categories should be receiving palliative care, but it remains unclear what percentage need specialized palliative care, she said
As for improving the rate of palliative care consultations, Dr. Lois M. Ramondetta of the University of Texas M.D. Anderson Cancer Center, Houston, said during a "lecturette" following the presentations by Dr. Ruskin and Dr. Lefkowits, that "branding" is important.
One study showed that 70% of Americans don’t even know what palliative care is, and many of those who do – including both patients and health care providers – equate palliative care with end-of-life care.
Simply changing the name of the palliative care clinic at M.D. Anderson to the "supportive care clinic" led to a 40% increase in consultations, she said.
Most patients and physicians reacted favorably to the concept of supportive care, she explained, noting that palliative care should be rebranded as an extra layer of support, and it should be offered throughout the treatment process.
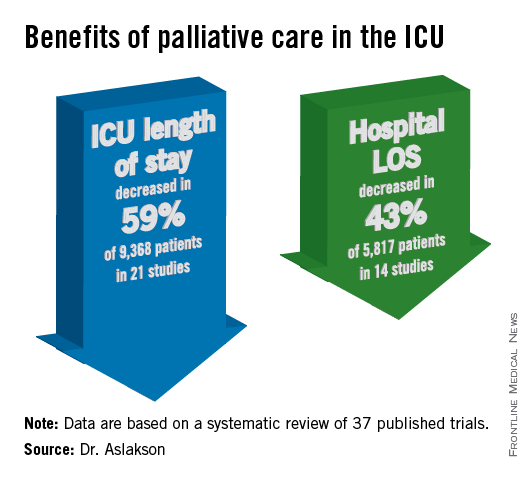
Studies consistently show that palliative care provides multiple benefits, including decreased hospital length of stay, fewer intensive care admissions, and reduced costs and need for potentially harmful procedures. Some studies have suggested palliative care is associated with improved overall survival, she said.
A number of efforts to improve palliative care skills and to increase referral for palliative care consultations are underway through both ASCO and the SGO, including a virtual learning collaborative being developed by ASCO, efforts to incorporate palliative care education into training and recertification programs, and the revival of a palliative care task force to address these issues.
Dr. Ruskin, Dr. Lefkowits, and Dr. Ramondetta each reported having no disclosures.
TAMPA – Outpatient palliative care consultations are associated with decreased symptom burden in women with gynecologic malignancies, but American Society of Clinical Oncology recommendations for referral are often ignored, according to retrospective data and a review of patient records.
In one study, 78 patients seen between June 2007 and March 2013 at an outpatient symptom management clinic for follow-up within 90 days of their initial consultation completed a questionnaire at each visit, including the nine-item Edmonton Symptom Assessment System. The responses, along with information from the patients’ charts, showed significant improvements in almost all symptoms over time, Dr. Rachel Ruskin, a clinical fellow at the University of California, San Francisco, reported at the annual meeting of the Society of Gynecologic Oncology.
For example, mean pain, fatigue, anxiety, depression, nausea, drowsiness, and appetite scores decreased between 0.7 and 1.5 points from the median baseline scores (on a 10 point scale). A decline in shortness of breath score also approached significance.
No difference was seen with respect to symptom improvement between patients with and without disease, although there appeared to be a trend toward a difference in anxiety scores, Dr. Ruskin noted.
Patients who were treated with concurrent cancer-directed therapies had improvements in pain and fatigue, but to a lesser extent than did those who did not receive treatment, she said.
Among the 35 patients who attended at least two follow-up visits, the improvements in nausea and shortness of breath seen at the first visit persisted at the second, and symptoms of depression and drowsiness continued to improve at each visit. Of those 35 patients, 58% had ovarian, fallopian tube, or peritoneal cancer; 20% had uterine cancer; and 15% had cervical cancer. Most (81%) had stage III, IV, or recurrent cancer.
Mean age at study entry was 57 years, 85% of patients had disease present, and 62% were undergoing treatment. The vast majority (87%) had received chemotherapy, 30% received radiation, and 8% had undergone surgery.
In patients for whom relevant data were available, there was evidence of mild hematologic, renal, and nutritional compromise, and nearly 25% of these patients had been hospitalized within the prior month.
"Notably, in our cohort the improvement in symptoms cannot be attributed to antineoplastic therapies, since – if anything – treatment by traditional oncologic modalities was associated with less benefit in some symptoms," Dr. Ruskin said, adding that future research should focus on "which aspects of palliative care are effective and by what mechanisms," as this information would be helpful for determining best practices that can be replicated across settings and for designing prospective concurrent standard oncologic and palliative care trials.
"In the meantime, we hope that these data will encourage providers to consider referral to their outpatient palliative care colleagues," she concluded.
Findings from another study presented at the meeting suggest there is some work to do in that regard.
In that study, Dr. Carolyn Lefkowits of the University of Pittsburgh found that oncologists are falling short when it comes to following the 2012 ASCO recommendation to consider early palliative care integration for "any patient with metastatic cancer and/or high symptom burden."
Of 340 women with a gynecologic malignancy who were admitted to a gynecologic oncology service between February 2012 and August 2012, only 32% were referred to palliative care, Dr. Lefkowits said.
The patients had a median age of 62 years, and an equal number had early- and late-stage disease. Nearly 25% had recurrent disease by the end of the study period.
Multivariate logistic regression identified independent predictors of palliative care consultation, including number of admission (odds ratio, 17.4 for greater than three vs. three or fewer admissions), admission for symptom management (OR, 22.0), and death within 6 months (OR, 15.7).
Notably, only 16% of the patients died within 6 months of the last admission during the study period, and although 25% of patients overall were referred to palliative care, 54% of those who died within 6 months were referred, suggesting that most referrals are not made for patients who are early in their disease course.
Furthermore, only 53% of patients with recurrent disease – all of whom should have been considered for palliative care integration based on the ASCO recommendations – were seen for palliative care, including 59% who had received three or more lines of chemotherapy, Dr. Lefkowits said.
The findings suggest that the group of patients referred for palliative care is characterized by high symptom burden and poor prognosis. In fact, most of those referred were likely already at the point where they would be considered hospice eligible, she said.
An analysis of referrals based on each ASCO recommendation category showed that the highest referral rate was for "symptom admission" (79%), and the lowest was for recurrent disease (52%).
"So, although the predictors of consultation are in keeping, I think, with the spirit of the ASCO recommendations, we’re still not comprehensively capturing these high-risk subgroups," Dr. Lefkowits said.
She added that she hopes the findings will serve as a "conversation starter, spurring us to address questions, including which gynecologic oncology patients are most appropriate to target for consistent palliative care referral."
Other questions to consider include which systems might help improve referral rates among those patients and which patients (and at what rates) should be referred for specialized palliative care.
All of the patients in the ASCO categories should be receiving palliative care, but it remains unclear what percentage need specialized palliative care, she said
As for improving the rate of palliative care consultations, Dr. Lois M. Ramondetta of the University of Texas M.D. Anderson Cancer Center, Houston, said during a "lecturette" following the presentations by Dr. Ruskin and Dr. Lefkowits, that "branding" is important.
One study showed that 70% of Americans don’t even know what palliative care is, and many of those who do – including both patients and health care providers – equate palliative care with end-of-life care.
Simply changing the name of the palliative care clinic at M.D. Anderson to the "supportive care clinic" led to a 40% increase in consultations, she said.
Most patients and physicians reacted favorably to the concept of supportive care, she explained, noting that palliative care should be rebranded as an extra layer of support, and it should be offered throughout the treatment process.
Studies consistently show that palliative care provides multiple benefits, including decreased hospital length of stay, fewer intensive care admissions, and reduced costs and need for potentially harmful procedures. Some studies have suggested palliative care is associated with improved overall survival, she said.
A number of efforts to improve palliative care skills and to increase referral for palliative care consultations are underway through both ASCO and the SGO, including a virtual learning collaborative being developed by ASCO, efforts to incorporate palliative care education into training and recertification programs, and the revival of a palliative care task force to address these issues.
Dr. Ruskin, Dr. Lefkowits, and Dr. Ramondetta each reported having no disclosures.
TAMPA – Outpatient palliative care consultations are associated with decreased symptom burden in women with gynecologic malignancies, but American Society of Clinical Oncology recommendations for referral are often ignored, according to retrospective data and a review of patient records.
In one study, 78 patients seen between June 2007 and March 2013 at an outpatient symptom management clinic for follow-up within 90 days of their initial consultation completed a questionnaire at each visit, including the nine-item Edmonton Symptom Assessment System. The responses, along with information from the patients’ charts, showed significant improvements in almost all symptoms over time, Dr. Rachel Ruskin, a clinical fellow at the University of California, San Francisco, reported at the annual meeting of the Society of Gynecologic Oncology.
For example, mean pain, fatigue, anxiety, depression, nausea, drowsiness, and appetite scores decreased between 0.7 and 1.5 points from the median baseline scores (on a 10 point scale). A decline in shortness of breath score also approached significance.
No difference was seen with respect to symptom improvement between patients with and without disease, although there appeared to be a trend toward a difference in anxiety scores, Dr. Ruskin noted.
Patients who were treated with concurrent cancer-directed therapies had improvements in pain and fatigue, but to a lesser extent than did those who did not receive treatment, she said.
Among the 35 patients who attended at least two follow-up visits, the improvements in nausea and shortness of breath seen at the first visit persisted at the second, and symptoms of depression and drowsiness continued to improve at each visit. Of those 35 patients, 58% had ovarian, fallopian tube, or peritoneal cancer; 20% had uterine cancer; and 15% had cervical cancer. Most (81%) had stage III, IV, or recurrent cancer.
Mean age at study entry was 57 years, 85% of patients had disease present, and 62% were undergoing treatment. The vast majority (87%) had received chemotherapy, 30% received radiation, and 8% had undergone surgery.
In patients for whom relevant data were available, there was evidence of mild hematologic, renal, and nutritional compromise, and nearly 25% of these patients had been hospitalized within the prior month.
"Notably, in our cohort the improvement in symptoms cannot be attributed to antineoplastic therapies, since – if anything – treatment by traditional oncologic modalities was associated with less benefit in some symptoms," Dr. Ruskin said, adding that future research should focus on "which aspects of palliative care are effective and by what mechanisms," as this information would be helpful for determining best practices that can be replicated across settings and for designing prospective concurrent standard oncologic and palliative care trials.
"In the meantime, we hope that these data will encourage providers to consider referral to their outpatient palliative care colleagues," she concluded.
Findings from another study presented at the meeting suggest there is some work to do in that regard.
In that study, Dr. Carolyn Lefkowits of the University of Pittsburgh found that oncologists are falling short when it comes to following the 2012 ASCO recommendation to consider early palliative care integration for "any patient with metastatic cancer and/or high symptom burden."
Of 340 women with a gynecologic malignancy who were admitted to a gynecologic oncology service between February 2012 and August 2012, only 32% were referred to palliative care, Dr. Lefkowits said.
The patients had a median age of 62 years, and an equal number had early- and late-stage disease. Nearly 25% had recurrent disease by the end of the study period.
Multivariate logistic regression identified independent predictors of palliative care consultation, including number of admission (odds ratio, 17.4 for greater than three vs. three or fewer admissions), admission for symptom management (OR, 22.0), and death within 6 months (OR, 15.7).
Notably, only 16% of the patients died within 6 months of the last admission during the study period, and although 25% of patients overall were referred to palliative care, 54% of those who died within 6 months were referred, suggesting that most referrals are not made for patients who are early in their disease course.
Furthermore, only 53% of patients with recurrent disease – all of whom should have been considered for palliative care integration based on the ASCO recommendations – were seen for palliative care, including 59% who had received three or more lines of chemotherapy, Dr. Lefkowits said.
The findings suggest that the group of patients referred for palliative care is characterized by high symptom burden and poor prognosis. In fact, most of those referred were likely already at the point where they would be considered hospice eligible, she said.
An analysis of referrals based on each ASCO recommendation category showed that the highest referral rate was for "symptom admission" (79%), and the lowest was for recurrent disease (52%).
"So, although the predictors of consultation are in keeping, I think, with the spirit of the ASCO recommendations, we’re still not comprehensively capturing these high-risk subgroups," Dr. Lefkowits said.
She added that she hopes the findings will serve as a "conversation starter, spurring us to address questions, including which gynecologic oncology patients are most appropriate to target for consistent palliative care referral."
Other questions to consider include which systems might help improve referral rates among those patients and which patients (and at what rates) should be referred for specialized palliative care.
All of the patients in the ASCO categories should be receiving palliative care, but it remains unclear what percentage need specialized palliative care, she said
As for improving the rate of palliative care consultations, Dr. Lois M. Ramondetta of the University of Texas M.D. Anderson Cancer Center, Houston, said during a "lecturette" following the presentations by Dr. Ruskin and Dr. Lefkowits, that "branding" is important.
One study showed that 70% of Americans don’t even know what palliative care is, and many of those who do – including both patients and health care providers – equate palliative care with end-of-life care.
Simply changing the name of the palliative care clinic at M.D. Anderson to the "supportive care clinic" led to a 40% increase in consultations, she said.
Most patients and physicians reacted favorably to the concept of supportive care, she explained, noting that palliative care should be rebranded as an extra layer of support, and it should be offered throughout the treatment process.
Studies consistently show that palliative care provides multiple benefits, including decreased hospital length of stay, fewer intensive care admissions, and reduced costs and need for potentially harmful procedures. Some studies have suggested palliative care is associated with improved overall survival, she said.
A number of efforts to improve palliative care skills and to increase referral for palliative care consultations are underway through both ASCO and the SGO, including a virtual learning collaborative being developed by ASCO, efforts to incorporate palliative care education into training and recertification programs, and the revival of a palliative care task force to address these issues.
Dr. Ruskin, Dr. Lefkowits, and Dr. Ramondetta each reported having no disclosures.
AT THE ANNUAL MEETING ON WOMEN’S CANCER
Major finding: Palliative care consultation was associated with 12%-57% reductions in mean pain, fatigue, anxiety, depression, nausea, drowsiness, and appetite scores, but less than one-third of patients in another study were referred for consultation.
Data source: Retrospective studies that included 78 patients and 340 patients.
Disclosures: Dr. Ruskin, Dr. Lefkowits, and Dr. Ramondetta each reported having no disclosures.
VIDEO: Feeding elderly patients after hip surgery
LAS VEGAS – Among 100 hip fracture patients at Salem (Ore.) Hospital, those fed within 24 hours left the hospital sooner, and fewer of them died. Other patients went up to a week without being fed.
Doctors might have overlooked nutrition or been put off by the notion that gastronomy feeding tubes don’t help in end-stage dementia. The Salem patients, however, didn’t have end-stage dementia and were being fed by nasogastric tubes. In an interview, Dr. Cynthia Wallace, medical director of Vibra Specialty Hospital in nearby Portland, explained that it’s time to broaden who’s authorized to order nutritional consults, so that patients don’t fall through the cracks.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Among 100 hip fracture patients at Salem (Ore.) Hospital, those fed within 24 hours left the hospital sooner, and fewer of them died. Other patients went up to a week without being fed.
Doctors might have overlooked nutrition or been put off by the notion that gastronomy feeding tubes don’t help in end-stage dementia. The Salem patients, however, didn’t have end-stage dementia and were being fed by nasogastric tubes. In an interview, Dr. Cynthia Wallace, medical director of Vibra Specialty Hospital in nearby Portland, explained that it’s time to broaden who’s authorized to order nutritional consults, so that patients don’t fall through the cracks.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
LAS VEGAS – Among 100 hip fracture patients at Salem (Ore.) Hospital, those fed within 24 hours left the hospital sooner, and fewer of them died. Other patients went up to a week without being fed.
Doctors might have overlooked nutrition or been put off by the notion that gastronomy feeding tubes don’t help in end-stage dementia. The Salem patients, however, didn’t have end-stage dementia and were being fed by nasogastric tubes. In an interview, Dr. Cynthia Wallace, medical director of Vibra Specialty Hospital in nearby Portland, explained that it’s time to broaden who’s authorized to order nutritional consults, so that patients don’t fall through the cracks.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Don't forget non-Alzheimer dementias
Dementia is not always due to Alzheimer disease. An accurate diagnosis is important, as the various causative conditions can differ in their course and treatment.
Dementia refers to cognitive impairment severe enough to interfere with the ability to independently perform activities of daily living. It can occur at any age but is most common after age 60. Some studies estimate that 13.9% of people age 71 and older have some form of dementia.1 The prevalence increases with age, ranging from 5% at age 70 to 79 to 37% at age 90 and older.1
Alzheimer disease accounts for about 60% to 80% of cases,2 or an estimated 4.7 million people age 65 and older in the United States, a number anticipated to climb to 13.8 million by 2050.3
Other types of dementia are less often considered and are challenging to recognize, although many have distinct characteristics. This article summarizes the features and management of the more common non-Alzheimer dementias:
- Vascular dementia
- Dementia with Lewy bodies
- Progressive supranuclear palsy
- Corticobasal degeneration
- Multiple system atrophy
- Parkinson disease dementia
- Frontotemporal dementia
- Primary progressive aphasia
- Normal-pressure hydrocephalus
- Rapidly progressive dementia (ie, Creutzfeld-Jakob disease, autoimmune disease).
VASCULAR DEMENTIA
After Alzheimer disease, vascular dementia is the most common dementia, accounting for about 20% to 30% of cases. Clinical criteria have not been widely accepted, although several have been published, including those in the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition) and the National Institute of Neurological and Communicative Diseases and Stroke-Association Internationale pour la Recherche et l’Enseignement en Neurosciences.
Risk factors for vascular dementia include cerebrovascular disease (hypertension, diabetes, hyperlipidemia) and coexisting conditions related to atherosclerosis (coronary artery disease, peripheral artery disease).
The Hachinski Ischemic Score is a good bedside tool to help differentiate Alzheimer dementia from vascular dementia.5
Sudden onset and stepwise decline
Vascular dementia often presents as a sudden and stepwise progression of cognitive deficits that stabilize and that are caused by vascular insults (Table 1).6–10 Some patients have continuous decline after a vascular event, indicating that Alzheimer dementia may also be present. Dementia is then defined as a mixed type.
Behavioral problems such as physical aggression, hallucinations, paranoia, and mood fluctuations are common.11
Deficits depend on vascular areas affected
Cognitive deficits are heterogeneous and are often related to the location of the vascular insult. Involvement of subcortical areas may result in executive dysfunction, slowed processing speed, and behavioral changes.12
Executive dysfunction may be identified using the Trail Making Test (Part B) or the Executive Interview (EXIT25). Office-based tools such as the Folstein Mini-Mental State Examination, the Montreal Cognitive Assessment, or the St. Louis University Mental Status Examination may also uncover these deficits.
Focal neurologic deficits may be found on clinical examination.
Structural neuroimaging may identify small strokes in areas of the brain affecting cognitive function or occlusion of a larger vessel associated with more profound neurologic deficits. Neuroimaging findings may not correlate with any significant decline noted by the patient, suggesting “silent” strokes.
Treat symptoms and manage risk factors
Although the US Food and Drug Administration (FDA) has not approved any pharmacotherapy for vascular dementia, commonly prescribed cognitive enhancers have demonstrated some benefit.13
Behavioral problems such as aggression can be disturbing to the patient and the caregiver. Nonpharmacologic methods (eg, redirection, rescheduling care activities to avoid conflict, avoiding issues that lead to agitation) should be tried first to address these problems.
Drug therapy may be used off-label for neuropsychiatric symptoms such as hallucinations, delusions, and combativeness, but clinical trials of these agents for this purpose have shown mixed results,14 and their use is often associated with significant risk.15 Antipsychotic drugs are associated with a risk of death and pneumonia when prescribed for dementia. Many also carry a risk of QT prolongation, which is particularly concerning for patients with coronary artery disease or rhythm disturbances.
The key to reducing further decline is to optimize management of vascular risk factors to reduce stroke risk.
DEMENTIA WITH LEWY BODIES
Dementia with Lewy bodies, the next most common neurodegenerative dementia in the elderly, is characterized by progressive loss of cognitive function, prominent visual hallucinations, and parkinsonism (Table 1).6 Disease progression usually occurs over years but can be more rapid than in Alzheimer disease.
Alpha-synucleinopathy results in dysfunction of synaptic vesicles in presynaptic terminals. Lewy bodies may be diffusely spread in cortical and subcortical areas (appearing as spherical masses).
Visual hallucinations are typical
The McKeith criteria16 are the gold standard for diagnosing probable Lewy body dementia, based on clinical and imaging features (Table 2).
Visual hallucinations are usually well formed and detailed. They may initially be pleasant (eg, seeing children and little people) but may evolve to be accompanied by persecutory delusions.
Parkinsonism develops with or after dementia with Lewy bodies
Dementia with Lewy bodies and Parkinson disease dementia share many clinical and pathologic features; Parkinson dementia also is associated with cortical Lewy bodies.
Parkinsonian features include bradykinesia, masked facies, and rigidity. Resting tremor is less common.
The third report of the Dementia With Lewy Bodies Consortium recommends that the condition be diagnosed if dementia occurs before or concurrently with parkinsonism, and dementia with Parkinson disease should be diagnosed if dementia occurs in the context of well-established Parkinson disease.16 The development of dementia within 12 months of extrapyramidal signs suggests dementia with Lewy bodies.
Cognitive deficits fluctuate
Cognitive impairment in Lewy body dementia is characterized by progressive dementia with fluctuations in cognitive performance. Family members or caregivers may report that the patient can carry on a conversation one day and the next day be confused and inattentive. Compared with those with Alzheimer dementia, patients with Lewy body dementia have better delayed recall but more problems with executive functioning (planning) and visuospatial skills (following an unfamiliar route, copying a figure).
Specialized imaging provides clues
Dementia with Lewy bodies is associated with diffuse brain atrophy, with no established characteristic pattern on structural neuroimaging with computed tomography (CT) or magnetic resonance imaging (MRI).17 The contrast agent ioflupane iodine-123 injection (DaTscan) used with single-photon emission CT (SPECT) detects dopamine transporters, which are reduced in parkinsonian syndromes. The scan can also help differentiate between Alzheimer dementia and Lewy body dementia by detecting the loss of functional dopaminergic terminals in the striatum in Lewy body dementia. Alpha-synuclein imaging may become another useful diagnostic tool in the future.
Alzheimer medications may help in dementia with Lewy bodies
Medications with anticholinergic effects and dopamine agonists should be discontinued because of possible effects on cognitive function and parkinsonism. In one clinical trial,18 rivastigmine (Exelon) was found to help cognitive functioning as well as reduce psychotic symptoms in dementia with Lewy bodies, although a recent Cochrane review could not support the evidence for use of all cholinesterase inhibitors in Lewy body dementia.19 In another trial,20 memantine (Namenda) was found to improve global clinical status and behavioral symptoms of Lewy body dementia.
Treating hallucinations of dementia with Lewy bodies
Patients with dementia with Lewy bodies are extremely sensitive to the extrapyramidal side effects of neuroleptic drugs. Some evidence indicates that the atypical antipsychotic drug quetiapine (Seroquel) helps with prominent and disturbing psychotic features and is less likely to worsen parkinsonism than other antipsychotics.21 The best evidence is for clozapine (Clozaril) as a treatment for hallucinations in Parkinson dementia, but the possible side effect of agranulocytosis limits its clinical use. Other atypical antipsychotics such as risperidone (Risperdal) and olanzapine (Zyprexa) are not recommended.22
PROGRESSIVE SUPRANUCLEAR PALSY
Progressive supranuclear palsy is a sporadic atypical parkinsonian disorder with onset between age 50 and 70. Familial cases are infrequent.
Progressive supranuclear palsy presents as early postural instability, vertical supranuclear gaze palsy, and axial muscle rigidity in the first few years. Disease progression is gradual: one study of 50 patients found that the median time from onset to the first key motor impairment (unintelligible speech, no independent walking, inability to stand unassisted, wheelchair-bound, or recommendation for feeding tube placement) was 4 years.23
Histologically, progressive supranuclear palsy is characterized by accumulation of tau protein aggregates in the basal ganglia, brainstem, and cerebral cortex. The degenerative process involves dopaminergic, cholinergic, and gamma-aminobutyric acid (GABA)-ergic neurons.24
Gait and balance problems predominate early in progressive supranuclear palsy
The most commonly used diagnostic criteria are from the National Institute of Neurological Disorders and Stroke. The diagnosis of probable progressive supranuclear palsy requires vertical gaze palsy and falls or the tendency to fall within the first year of disease onset and exclusion of other causes.
The earliest symptom is usually gait and balance impairment.25 Falls (usually backward) and postural instability occur during the first year in 58% of patients.26 Instead of turning en bloc as in Parkinson disease, patients with progressive supranuclear palsy tend to pivot quickly. Patients may also have a coarse groaning voice and moaning. Insomnia has been reported, but rapid-eye-movement sleep behavior disorders are infrequent (unlike in Parkinson disease, multiple system atrophy, and Lewy body dementia).27
Apathy and extreme mood swings
Cognitive impairment is seen in 50% of patients in the early stage of progressive supranuclear palsy. It mostly involves the frontal lobe, including frontal behavioral disturbances (eg, apathy in 91% of patients26 or pseudobulbar affect and extreme emotional lability) and deficits in abstract thoughts or verbal fluency (to test this, patients are asked to say as many words as possible from a category in a given time). Ideomotor apraxia (inability to correctly imitate hand gestures and voluntarily pantomime tool use, such as pretending to brush hair) is rare, despite corticobasal degeneration.28
Vertical gaze palsy
The hallmark of progressive supranuclear palsy is vertical gaze palsy. Initially, this involves slowing of vertical saccades, followed by diminished vertical gaze and more characteristic downward gaze palsy. These findings may develop over 3 to 4 years. Vertical gaze palsy leads to spilling food and tripping while walking.
The gaze abnormality combined with rare blinking and facial dystonia form the classic facial expression of astonishment called “leonine facies.” The face is stiff and deeply furrowed, with a look of surprise.
Axial (especially neck) rigidity is more prominent than limb rigidity. Retrocollis (the head is drawn back) occurs in less than 25% of patients. Parkinsonian features such as bradykinesia affect nearly half of patients by the time of diagnosis.
Instead of the classic symptoms of progressive supranuclear palsy, about one-third of patients present with progressive supranuclear palsy-parkinsonism, which involves asymmetric parkinsonism that initially responds to levodopa.29
MRI shows ‘hummingbird sign’
Brain MRI shows atrophy of the brainstem, particularly the midbrain. Thinning of the superior part of the midbrain and dilation of the third ventricle (“hummingbird sign” on sagittal sections or “morning glory flower” on axial sections) support a diagnosis of progressive supranuclear palsy and differentiate it from Parkinson disease and other atypical parkinsonian disorders.30,31
Levodopa ineffective for supranuclear palsy
There is no treatment to slow progressive supranuclear palsy. Even in high doses, levodopa rarely alleviates parkinsonian features in a clinically meaningful way.26 Successful experimental biologic therapies have been studied in animal models.32 Davunetide is thought to help with neuronal integrity and cell survival through the stabilization of microtubules in preclinical studies, but it has not been used in clinical practice.33
CORTICOBASAL DEGENERATION
Corticobasal degeneration is a progressive, asymmetric movement disorder often manifesting initially with cognitive or behavioral impairment. It is associated with abnormality of the cytoskeleton protein tau. Onset is usually after age 60.
Asymmetric movement disorder with cognitive dysfunction
This diagnosis is clinical. Diagnostic criteria proposed in 2003 include the following core features34:
- Insidious onset and progressive course
- No identifiable cause
- Cortical dysfunction with at least one of the following: apraxia, alien limb phenomenon (one limb moves involuntarily with complex movements, eg, grabbing the other hand), cortical sensory loss, visual hemineglect, nonfluent aphasia
- Extrapyramidal dysfunction: focal rigidity unresponsive to levodopa, asymmetric dystonia.
An international consortium has developed more specific clinical research criteria for probable and possible corticobasal degeneration.35 In a series of 147 patients, the following clinical features were found: parkinsonism (100%), higher cortical dysfunction (93%), dyspraxia (82%), gait disorder (80%), unilateral limb dystonia (71%), tremor (55%), and dementia (25%).36
Behavioral problems commonly include depression; apathy, irritability, and agitation are also reported.37
Cognitive testing may reveal deficits in frontal-parietal cognitive domains including attention and concentration, executive function, verbal fluency, and visuospatial skills.38 Learning disabilities may be improved with verbal cueing (in contrast to Alzheimer disease). Patients may also have impaired graphesthesia (the ability to recognize writing on the skin only by the sensation of touch).39,40
Motor examination may reveal marked asymmetry. Hand, limb, speech, and gait apraxias are common. Gait is typically slow, with short steps and shuffling, and a wide-based or freezing gait. Arm swing may be absent on one side.
Asymmetric cortical atrophy
Early on, MRI may be normal. As the disease progresses, asymmetric cortical atrophy may be seen, especially in the posterior frontal and parietal lobes.
Levodopa ineffective in corticobasal degeneration
Corticobasal degeneration responds poorly to levodopa. Botulinum toxin has been used to help with dystonia and limb pain.
MULTIPLE SYSTEM ATROPHY
Multiple system atrophy is another atypical parkinsonian disorder, most often diagnosed in men over age 60. It is characterized by sporadic parkinsonism, cerebellar signs (involving balance and coordination), pyramidal tract dysfunction, and autonomic insufficiency in varying combinations. Two major subtypes are recognized, depending on whether the predominating presenting features are cerebellar signs or parkinsonism. In contrast to dementia with Lewy bodies, psychiatric symptoms are not a major feature, except possibly depression.41
Diagnosis requires a sporadic progressive disorder that has features of autonomic failure and poor response of parkinsonism or cerebellar ataxia to levodopa.42
Multiple system atrophy is usually not associated with dementia in the early stages, but patients develop deficits in learning, recognition, memory, and verbal fluency as the disease progresses.43 Rapid-eye-movement sleep behavior disorder has been reported in more than half of patients.44
A neurologic examination provides clues
Parkinsonian features are usually symmetric, in contrast to idiopathic Parkinson disease. These signs may include akinesia with rigidity, postural instability, hypokinetic speech, and tremor.
Cerebellar signs include nystagmus and dysarthria (speech disturbance), and gait and limb ataxia.
Pyramidal features include extensor plantar responses and hyperreflexia.
Autonomic dysfunction includes orthostatic hypotension, bladder and rectal atony, loss of sweating, urinary or fecal incontinence, and erectile dysfunction.
Electromyography may demonstrate decreased anal sphincter tone.
MRI shows atrophy of putamen and pons
Brain MRI may show atrophy of the putamen (hypointensity of the putamen with a hyperintense rim). Pons atrophy may also be present, revealing a “hot cross bun” sign in axial images. These combined findings have specificity above 90% but limited sensitivity. These signs are useful to distinguish multiple system atrophy from Parkinson dementia, but their absence does not exclude the diagnosis of multiple system atrophy.45,46
Multiple system atrophy typically responds poorly to levodopa
Levodopa may improve movement and rigidity, but many respond poorly to treatment or lose response after a few years. Fludrocortisone (Florinef) or vasoconstrictors such as midodrine (Orvaten, Proamatine) may help with orthostatic hypotension.47,48
PARKINSON DISEASE DEMENTIA
Dementia eventually develops in most patients with Parkinson disease. Older age and the akinetic rigid form of the disease are associated with higher risk. Diagnosis of idiopathic Parkinson disease before the development of dementia is essential for the diagnosis.
The Movement Disorder Society Task Force has developed new diagnostic criteria.49 Deficits must be present in at least two of the four core cognitive domains (attention, memory, executive, and visuospatial functions) and must be severe enough to affect daily functioning.
Behavioral symptoms such as affective changes, hallucinations, and apathy are common.
MRI shows characteristic brain atrophy in Parkinson disease dementia
MRI shows reduced gray matter volume in the frontal lobe in patients with Parkinson disease without dementia compared with controls. In Parkinson disease dementia, reduced volume extends to temporal, occipital, and subcortical areas. No significant volumetric differences have been observed in Parkinson dementia compared with dementia with Lewy bodies.50 A greater decrease of glucose metabolism has been found in the inferior parietal and occipital lobes in Parkinson disease dementia than in Parkinson disease without dementia.51
Rivastigmine effective for dementia
A Cochrane review supports the use of acetylcholinesterase inhibitors in patients with Parkinson disease dementia, with a positive impact on global assessment, cognitive function, behavioral disturbance, and activities of daily living rating scales.19 At this time, rivastigmine is the only FDA-approved cholinesterase inhibitor for treating Parkinson disease dementia. In clinical trials, memantine did not improve global clinical status or behavioral symptoms of dementia of Parkinson disease.51
FRONTOTEMPORAL DEMENTIA
Frontotemporal dementia frequently starts before age 65 and accounts for 20% to 50% of dementias in this age group.52 Recognition of the condition in older patients is also growing.53 Frontotemporal dementia encompasses a spectrum of dementias, including behavioral variant frontotemporal dementia, semantic dementia, and progressive nonfluent aphasia.54
Gradual onset of uncharacteristic behaviors
Accepted diagnostic criteria include core features of gradual onset, early decline in social and interpersonal conduct, early impairment of self-regulation, emotional blunting, and loss of insight. Many patients are diagnosed with psychiatric conditions. Changes reported by family and caregivers typically deviate substantially from the person’s usual behavior, such as impulsive and inappropriate behaviors or complete withdrawal and apathy.
Language sometimes affected in frontotemporal dementia
Language impairment may be present in some variants. Behavioral and language changes often accompany other forms of dementia (Alzheimer disease, vascular dementia, primary progressive aphasia), making diagnosis more challenging. Office-based testing often does not reveal any deficits, although the Frontal Behavioral Inventory may help.55 A referral to a clinical neuropsychologist may help identify and quantify cognitive impairments.
MRI shows frontotemporal lobes affected
Structural neuroimaging may not reveal abnormalities initially, but with progression, atrophy may be seen in the frontal and temporal lobes. Functional neuroimaging (positron emission tomography, brain SPECT, functional MRI) show hypometabolism in the same areas.
Treat symptoms
There are no specific FDA-approved therapies for frontotemporal dementia. Acetylcholinesterase inhibitors can help progressive nonfluent aphasia in some cases. Selective serotonin reuptake inhibitors may alleviate depressive symptoms, and low doses of atypical antipsychotic medications may help with impulsivity, disinhibition, and aggressive or disruptive behaviors.56
PRIMARY PROGRESSIVE APHASIA
Language impairment predominates
Primary progressive aphasia is a rare form of dementia in which symptoms typically develop around age 60. Pathology is varied. In a study of 60 patients with initial clinical symptoms of primary progressive aphasia, postmortem histology of brain tissue revealed various findings, including those consistent with Alzheimer pathology and motor neuron diseasetype inclusions.57
Patients typically present with expressive language problems as the primary deficit for the first 2 years of the disease, with preservation in other cognitive areas such as memory, visuospatial skills, and executive function.58 Office-based testing may overstate the severity of the dementia, given the dependence of performance on intact language.
It is important to distinguish primary progressive aphasia from other dementias that also affect language. In the frontal variant of frontotemporal dementia, the primary language problem is anomia (inability to name objects) or diminished speech output, which may be accompanied by behavioral problems. Semantic dementia affects word recognition as well as comprehension. In Alzheimer disease, language may be affected along with memory and other areas of cognitive function.
Imaging shows focal degeneration in the left hemisphere
Structural neuroimaging does not initially reveal any deficits, but later it may reveal atrophy in the frontal, perisylvian complex, and temporal areas of the left hemisphere, reflecting the focal nature of the degeneration.59 Functional neuroimaging (positron emission tomography, SPECT) may reveal hypometabolism or diminished blood flow in these areas prior to changes in structural neuroimaging.60
Other communication methods may help
There are no FDA-approved therapies for primary progressive aphasia. Off-label use of some agents (eg, selective serotonin reuptake inhibitors and small doses of antipsychotic medications) has been found useful in small trials.56 Patients may benefit from learning other forms of communication, such as using sign language, laminated cards with printed words or pictures, or artificial voice synthesizers, to express their needs.
NORMAL-PRESSURE HYDROCEPHALUS
Classic triad: Gait, cognition, incontinence
With the onset of symptoms in the sixth or seventh decade, normal-pressure hydrocephalus affects less than 1% of people age 65 and older. It represents up to 5% of dementias, although estimates are influenced by the varied criteria for diagnosis.61 It is characterized by the classic triad of gait impairment, cognitive impairment, and urinary frequency or incontinence.62
Symptoms progress over a period of years, with gait impairment often predominating. As this triad is common in the geriatric population, identifying other explanations is important. Gait impairment caused by spinal stenosis, peripheral neuropathy, or parkinsonism should be explored. Cognitive impairment could be due to depression, Alzheimer disease, or other forms of dementia. Urinary symptoms may be related to detrusor instability or an enlarged prostate.
Gait impairment initially manifests as slowing of gait, but progresses to difficulty with gait initiation. Gait tends to be wide-based (stance more than 1 foot wide).
Cognitive impairment is typically subcortical, manifested as slowed processing speed and impaired executive function. Recall and working memory may be impaired.
Enlarged ventricles seen on imaging in normal-pressure hydrocephalus
Structural neuroimaging reveals enlarged ventricles (Evan’s ratio > 0.358). This can be difficult to distinguish from ventriculomegaly due to cerebral atrophy; assessing the callosal angle on MRI may distinguish the two.63,64 Diagnosis of normal-pressure hydrocephalus can be confirmed using a cerebrospinal fluid infusion test to assess resistance of fluid to resorption.65
Treat with cerebrospinal fluid drainage
Specific tests should be performed to determine candidacy for surgery. These include a high-volume lumbar puncture (40 to 50 mL) or a trial of external lumbar drainage (10 mL per hour for 48 to 72 hours).65 Definitive treatment is surgical placement of a shunt to allow cerebrospinal fluid to drain into the atria or peritoneal cavity.
Surgery may improve gait, but cognitive symptoms often remain,66 and clinical decline may occur after the shunt is placed. Once gait dysfunction is resolved, other explanations for cognitive impairment or residual gait impairment should be considered. An underlying reason for progression of normal-pressure hydrocephalus symptoms after surgical intervention should be identified.67
RAPIDLY PROGRESSIVE DEMENTIAS
Rapidly progressive dementias are among the most challenging of dementing illnesses. They are characterized by a subacute course and an accelerated rate of decline, developing in less than 2 years. Evaluation should typically be more comprehensive than for other types of dementia. The main goal is to diagnose potentially treatable conditions, such as Hashimoto encephalopathy or paraneoplastic limbic encephalitis, and to distinguish these conditions from diseases with a very poor prognosis, such as Creutzfeldt-Jakob disease.
Creutzfeldt-Jakob disease
Creutzfeldt-Jakob disease is a fatal prion-related neurodegenerative illness. Sporadic disease is most common, but variant, familial, and iatrogenic types have been reported. The most common initial symptoms in sporadic disease are cognitive (39%), cerebellar (21%), behavioral (20%), constitutional (20%), sensory (11%), motor (9%), and visual (7%).68
Chronic neurodegenerative diseases can be misdiagnosed as Creutzfeldt-Jakob disease because of an atypical time course and multi-system neurologic findings.
The US Centers for Disease Control and Prevention has adopted criteria for diagnosing probable Creutzfeldt-Jakob disease (Table 3). Routine investigations should also not suggest an alternative diagnosis.69
Autoimmune diseases
Autoimmune conditions may present as a rapidly progressive dementia, including Hashimoto encephalopathy and antibody-mediated limbic encephalitis, either associated with cancer (paraneoplastic) or without cancer (nonparaneoplastic).
Paraneoplastic limbic encephalitis is a group of inflammatory conditions involving antibodies produced within the cerebrospinal fluid and serum resulting in neurologic symptoms. These antibodies react against proteins expressed mostly by a tumor somewhere else in the body.70
Hashimoto encephalitis is a subacute to chronic encephalopathy that may present as dementia with abnormally high levels of thyroid antibodies. The symptoms can vary from confusion to psychosis. There are two main presentations: one involves a relapsing-remitting course with stroke-like episodes (27% of patients) and the second consists of insidious onset of seizures (66% of patients).
Diagnosis involves testing for elevated anti-thyroid peroxidase and thyroglobulin antibodies. MRI findings are nonspecific. Hashimoto encephalitis responds to treatment with corticosteroids, plasmapheresis, or immunosuppressive therapy.71
- Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology 2007; 29:125–132.
- Alzheimer’s Association. 2013 Alzheimer’s disease facts and figures. http://www.alz.org/downloads/facts_figures_2013.pdf. Accessed February 3, 2014.
- Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology 2013; 80:1778–1783.
- Hou CE, Carlin D, Miller BL. Non-Alzheimer’s disease dementias: anatomic, clinical, and molecular correlates. Can J Psychiatry 2004; 49:164–171.
- Hachinski VC, Iliff LD, Zilhka E, et al. Cerebral blood flow in dementia. Arch Neurol 1975; 32:632–637.
- Fereshtehnejad SM, Religa D, Westman E, Aarsland D, Lökk J, Eriksdotter M. Demography, diagnostics, and medication in dementia with Lewy bodies and Parkinson’s disease with dementia: data from the Swedish Dementia Quality Registry (SveDem). Neuropsychiatr Dis Treat 2013; 9:927–935.
- Nomura T, Inoue Y, Takigawa H, Nakashima K. Comparison of REM sleep behaviour disorder variables between patients with progressive supranuclear palsy and those with Parkinson’s disease. Parkinsonism Relat Disord 2012; 18:394–396.
- Davis PH, Golbe LI, Duvoisin RC, Schoenberg BS. Risk factors for progrssive supranuclear palsy. Neurology 1988; 38:1546–1552.
- Tousi B, Schuele SU, Subramanian T. A 46-year-old woman with rigidity and frequent falls. Cleve Clin J Med 2005; 72:57–63.
- Cooper AD, Josephs KA. Photophobia, visual hallucinations, and REM sleep behavior disorder in progressive supranuclear palsy and corticobasal degeneration: a prospective study. Parkinsonism Relat Disord 2009; 15:59–61.
- Lyketsos CG, Lopez O, Jones B, Fitzpatrick AL, Breitner J, DeKosky S. Prevalence of neuropsychiatric symptoms in dementia and mild cognitive impairment: results from the cardiovascular health study0. JAMA 2002; 288:1475–1483.
- Knopman DS. Dementia and cerebrovascular disease. Mayo Clin Proc 2006; 81:223–230.
- Erkinjuntti T, Kurz A, Gauthier S, Bullock R, Lilienfeld S, Damaraju CV. Efficacy of galantamine in probable vascular dementia and Alzheimer’s disease combined with cerebrovascular disease: a randomised trial. Lancet 2002; 359:1283–1290.
- Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA 2005; 293:596–608.
- Schneider LS, Dagerman K, Insel PS. Efficacy and adverse effects of atypical antipsychotics for dementia: meta-analysis of randomized, placebo-controlled trials. Am J Geriatr Psychiatry 2006; 14:191–210.
- McKeith IG, Dickson DW, Lowe J, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 2005; 65:1863–1872.
- Tartaglia MC, Rosen HJ, Miller BL. Neuroimaging in dementia. Neurotherapeutics 2011; 8:82–92.
- McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet 2000; 356:2031–2036.
- Rolinski M, Fox C, Maidment I, McShane R. Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson’s disease dementia and cognitive impairment in Parkinson’s disease. Cochrane Database Syst Rev 2012; 3:CD006504.
- Emre M, Tsolaki M, Bonucelli U, et al; on behalf of the 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol 2010; 9:969–977.
- Kurlan R, Cummings J, Raman R, Thal L; Alzheimer’s Disease Cooperative Study Group. Quetiapine for agitation or psychosis in patients with dementia and parkinsonism. Neurology 2007; 68:1356–1363.
- Ballard C, Aarsland D, Francis P, Corbett A. Neuropsychiatric symptoms in patients with dementias associated with cortical Lewy bodies: pathophysiology, clinical features, and pharmacological management. Drugs Aging 2013; 30:603–611.
- Goetz CG, Leurgans S, Lang AE, Litvan I. Progression of gait, speech and swallowing deficits in progressive supranuclear palsy. Neurology 2003; 60:917–922.
- Kasashima S, Oda Y. Cholinergic neuronal loss in the basal forebrain and mesopontine tegmentum of progressive supranuclear palsy and corticobasal degeneration. Acta Neuropathol 2003; 105:117–124.
- Fahn S, Jankovic J, Hallett M, editors. Principles and Practice of Movement Disorders. 2nd ed. New York, NY: Elsevier/Saunders; 2011.
- Litvan I, Mangone CA, McKee A, et al. Natural history of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome) and clinical predictors of survival: a clinicopathological study. J Neurol Neurosurg Psychiatry 1996; 60:615–620.
- Boeve BF, Silber MH, Parisi JE, et al. Synucleinopathy pathology and REM sleep behavior disorder plus dementia or parkinsonism. Neurology 2003; 61:40–45.
- Pharr V, Uttl B, Stark M, Litvan I, Fantie B, Grafman J. Comparison of apraxia in corticobasal degeneration and progressive supranuclear palsy. Neurology 2001; 56:957–963.
- Williams DR, Lees AJ. What features improve the accuracy of the clinical diagnosis of progressive supranuclear palsy-parkinsonism (PSP-P)? Mov Disord 2010; 25:357–362.
- Wenning GK, Litvan I, Tolosa E. Milestones in atypical and secondary parkinsonisms. Mov Disord 2011; 26:1083–1095.
- Gallucci M, Limbucci N, Catalucci A, Caulo M. Neurodegenerative diseases. Radiol Clin North Am 2008; 46:799–817.
- Stamelou M, de Silva R, Arias-Carrión O, et al. Rational therapeutic approaches to progressive supranuclear palsy. Brain 2010; 133:1578–1590.
- Gold M, Lorenzl S, Stewart AJ, Morimoto BH, Williams DR, Gozes I. Critical appraisal of the role of davunetide in the treatment of progressive supranuclear palsy. Neuropsychiatr Dis Treat 2012; 8:85–93.
- Boeve BF, Lang AE, Litvan I. Corticobasal degeneration and its relationship to progressive supranuclear palsy and frontotemporal dementia. Ann Neurol 2003; 54(suppl 5):S15–S19.
- Armstrong MJ, Litvan I, Lang AE, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013; 80:496–503.
- Kompoliti K, Goetz CG, Boeve BF, et al. Clinical presentation and pharmacological therapy in corticobasal degeneration. Arch Neurol 1998; 55:957–961.
- Litvan I, Cummings JL, Mega M. Neuropsychiatric features of corticobasal degeneration. J Neurol Neurosurg Psychiatry 1998; 65:717–721.
- Pillon B, Blin J, Vidailhet M, et al. The neuropsychological pattern of corticobasal degeneration: comparison with progressive supranuclear palsy and Alzheimer’s disease. Neurology 1995; 45:1477–1483.
- Tang-Wai DF, Josephs KA, Boeve BF, Petersen RC, Parisi JE, Dickson DW. Coexistent Lewy body disease in a case of “visual variant of Alzheimer’s disease.” J Neurol Neurosurg Psychiatry 2003; 74:389.
- Tang-Wai DF, Josephs KA, Boeve BF, Dickson DW, Parisi JE, Petersen RC. Pathologically confirmed corticobasal degeneration presenting with visuospatial dysfunction. Neurology 2003; 61:1134–1135.
- Goto K, Ueki A, Shimode H, Shinjo H, Miwa C, Morita Y. Depression in multiple system atrophy: a case report. Psychiatry Clin Neurosci 2000; 54:507–511.
- Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008; 71:670–676.
- Berent S, Giordani B, Gilman S, et al. Patterns of neuropsychological performance in multiple system atrophy compared to sporadic and hereditary olivopontocerebellar atrophy. Brain Cogn 2002; 50:194–206.
- Ghorayeb I, Yekhlef F, Chrysostome V, Balestre E, Bioulac B, Tison F. Sleep disorders and their determinants in multiple system atrophy. J Neurol Neurosurg Psychiatry 2002; 72:798–800.
- Schrag A, Kingsley D, Phatouros C, et al. Clinical usefulness of magnetic resonance imaging in multiple system atrophy. J Neurol Neurosurg Psychiatry 1998; 65:65–71.
- Massey LA, Micallef C, Paviour DC, et al. Conventional magnetic resonance imaging in confirmed progressive supranuclear palsy and multiple system atrophy. Mov Disord 2012; 27:1754–1762.
- Low PA, Gilden JL, Freeman R, Sheng KN, McElligott MA. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA 1997; 277:1046–1051.
- Mathias CJ, Kimber JR. Postural hypotension: causes, clinical features, investigation, and management. Annu Rev Med 1999; 50:317–336.
- Emre M, Aarsland D, Brown R, et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 2007; 22:1689–1707.
- Burton EJ, McKeith IG, Burn DJ, Williams ED, O’Brien JT. Cerebral atrophy in Parkinson’s disease with and without dementia: a comparison with Alzheimer’s disease, dementia with Lewy bodies and controls. Brain 2004; 127:791–800.
- Piert M, Koeppe RA, Giordani B, Minoshima S, Kuhl DE. Determination of regional rate constants from dynamic FDG-PET studies in Parkinson’s disease. J Nucl Med 1996; 37:1115–1122.
- Ratnavalli E, Brayne C, Dawson K, Hodges JR. The prevalence of frontotemporal dementia. Neurology 2002; 58:1615–1621.
- Baborie A, Griffiths TD, Jaros E, et al. Frontotemporal dementia in elderly individuals. Arch Neurol 2012; 69:1052–1960.
- Neary D, Snowden JS, Gustafson L, et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 1998; 51:1546–1554.
- Kertesz A, Nadkarni N, Davidson W, Thomas AW. The Frontal Behavioral Inventory in the differential diagnosis of frontotemporal dementia. J Int Neuropsychol Soc 2000; 6:460–468.
- Mendez MF, Lauterbach EC, Sampson SM; ANPA Committee on Research. An evidence-based review of the psychopathology of frontotemporal dementia: a report of the ANPA Committee on Research. J Neuropsychiatry Clin Neurosci 2008; 20:130–149.
- Kertesz A, McMonagle P, Blair M, Davidson W, Munoz DG. The evolution and pathology of frontotemporal dementia. Brain 2005; 128:1996–2005.
- Mesulam MM. Primary progressive aphasia—a language-based dementia. N Engl J Med 2003; 349:1535–1542.
- Turner RS, Kenyon LC, Trojanowski JQ, Gonatas N, Grossman M. Clinical, neuroimaging, and pathologic features of progressive nonfluent aphasia. Ann Neurol 1996; 39:166–173.
- Abe K, Ukita H, Yanagihara T. Imaging in primary progressive aphasia. Neuroradiology 1997; 39:556–559.
- Trenkwalder C, Schwarz J, Gebhard J, et al. Starnberg trial on epidemiology of parkinsonism and hypertension in the elderly. Prevalence of Parkinson’s disease and related disorders assessed by a door-to-door survey of inhabitants older than 65 years. Arch Neurol 1995; 52:1017–1022.
- Hakim S, Adams RD. The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965; 2:307–327.
- Vanneste JA. Diagnosis and management of normal-pressure hydrocephalus. J Neurol 2000; 247:5–14.
- Ishii K, Kanda T, Harada A, et al. Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol 2008; 18:2678–2683.
- Marmarou A, Bergsneider M, Klinge P, Relkin N, Black PM. The value of supplemental prognostic tests for the preoperative assessment of idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57(suppl 3):S17–S28.
- Bergsneider M, Miller C, Vespa PM, Hu X. Surgical management of adult hydrocephalus. Neurosurgery 2008; 62(suppl 2):643–659.
- Malm J, Graff-Radford NR, Ishikawa M, et al. Influence of comorbidities in idiopathic normal pressure hydrocephalus—research and clinical care. A report of the ISHCSF task force on comorbidities in INPH. Fluids Barriers CNS 2013; 10:22.
- Rabinovici GD, Wang PN, Levin J, et al. First symptom in sporadic Creutzfeldt-Jakob disease. Neurology 2006; 66:286–287.
- Zerr I, Kallenberg K, Summers DM, et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain 2009; 132:2659–2668.
- Dalmau J, Rosenfeld MR. Paraneoplastic syndromes of the CNS. Lancet Neurol 2008; 7:327–340.
- Chong JY, Rowland LP, Utiger RD. Hashimoto encephalopathy: syndrome or myth? Arch Neurol 2003; 60:164–171.
Dementia is not always due to Alzheimer disease. An accurate diagnosis is important, as the various causative conditions can differ in their course and treatment.
Dementia refers to cognitive impairment severe enough to interfere with the ability to independently perform activities of daily living. It can occur at any age but is most common after age 60. Some studies estimate that 13.9% of people age 71 and older have some form of dementia.1 The prevalence increases with age, ranging from 5% at age 70 to 79 to 37% at age 90 and older.1
Alzheimer disease accounts for about 60% to 80% of cases,2 or an estimated 4.7 million people age 65 and older in the United States, a number anticipated to climb to 13.8 million by 2050.3
Other types of dementia are less often considered and are challenging to recognize, although many have distinct characteristics. This article summarizes the features and management of the more common non-Alzheimer dementias:
- Vascular dementia
- Dementia with Lewy bodies
- Progressive supranuclear palsy
- Corticobasal degeneration
- Multiple system atrophy
- Parkinson disease dementia
- Frontotemporal dementia
- Primary progressive aphasia
- Normal-pressure hydrocephalus
- Rapidly progressive dementia (ie, Creutzfeld-Jakob disease, autoimmune disease).
VASCULAR DEMENTIA
After Alzheimer disease, vascular dementia is the most common dementia, accounting for about 20% to 30% of cases. Clinical criteria have not been widely accepted, although several have been published, including those in the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition) and the National Institute of Neurological and Communicative Diseases and Stroke-Association Internationale pour la Recherche et l’Enseignement en Neurosciences.
Risk factors for vascular dementia include cerebrovascular disease (hypertension, diabetes, hyperlipidemia) and coexisting conditions related to atherosclerosis (coronary artery disease, peripheral artery disease).
The Hachinski Ischemic Score is a good bedside tool to help differentiate Alzheimer dementia from vascular dementia.5
Sudden onset and stepwise decline
Vascular dementia often presents as a sudden and stepwise progression of cognitive deficits that stabilize and that are caused by vascular insults (Table 1).6–10 Some patients have continuous decline after a vascular event, indicating that Alzheimer dementia may also be present. Dementia is then defined as a mixed type.
Behavioral problems such as physical aggression, hallucinations, paranoia, and mood fluctuations are common.11
Deficits depend on vascular areas affected
Cognitive deficits are heterogeneous and are often related to the location of the vascular insult. Involvement of subcortical areas may result in executive dysfunction, slowed processing speed, and behavioral changes.12
Executive dysfunction may be identified using the Trail Making Test (Part B) or the Executive Interview (EXIT25). Office-based tools such as the Folstein Mini-Mental State Examination, the Montreal Cognitive Assessment, or the St. Louis University Mental Status Examination may also uncover these deficits.
Focal neurologic deficits may be found on clinical examination.
Structural neuroimaging may identify small strokes in areas of the brain affecting cognitive function or occlusion of a larger vessel associated with more profound neurologic deficits. Neuroimaging findings may not correlate with any significant decline noted by the patient, suggesting “silent” strokes.
Treat symptoms and manage risk factors
Although the US Food and Drug Administration (FDA) has not approved any pharmacotherapy for vascular dementia, commonly prescribed cognitive enhancers have demonstrated some benefit.13
Behavioral problems such as aggression can be disturbing to the patient and the caregiver. Nonpharmacologic methods (eg, redirection, rescheduling care activities to avoid conflict, avoiding issues that lead to agitation) should be tried first to address these problems.
Drug therapy may be used off-label for neuropsychiatric symptoms such as hallucinations, delusions, and combativeness, but clinical trials of these agents for this purpose have shown mixed results,14 and their use is often associated with significant risk.15 Antipsychotic drugs are associated with a risk of death and pneumonia when prescribed for dementia. Many also carry a risk of QT prolongation, which is particularly concerning for patients with coronary artery disease or rhythm disturbances.
The key to reducing further decline is to optimize management of vascular risk factors to reduce stroke risk.
DEMENTIA WITH LEWY BODIES
Dementia with Lewy bodies, the next most common neurodegenerative dementia in the elderly, is characterized by progressive loss of cognitive function, prominent visual hallucinations, and parkinsonism (Table 1).6 Disease progression usually occurs over years but can be more rapid than in Alzheimer disease.
Alpha-synucleinopathy results in dysfunction of synaptic vesicles in presynaptic terminals. Lewy bodies may be diffusely spread in cortical and subcortical areas (appearing as spherical masses).
Visual hallucinations are typical
The McKeith criteria16 are the gold standard for diagnosing probable Lewy body dementia, based on clinical and imaging features (Table 2).
Visual hallucinations are usually well formed and detailed. They may initially be pleasant (eg, seeing children and little people) but may evolve to be accompanied by persecutory delusions.
Parkinsonism develops with or after dementia with Lewy bodies
Dementia with Lewy bodies and Parkinson disease dementia share many clinical and pathologic features; Parkinson dementia also is associated with cortical Lewy bodies.
Parkinsonian features include bradykinesia, masked facies, and rigidity. Resting tremor is less common.
The third report of the Dementia With Lewy Bodies Consortium recommends that the condition be diagnosed if dementia occurs before or concurrently with parkinsonism, and dementia with Parkinson disease should be diagnosed if dementia occurs in the context of well-established Parkinson disease.16 The development of dementia within 12 months of extrapyramidal signs suggests dementia with Lewy bodies.
Cognitive deficits fluctuate
Cognitive impairment in Lewy body dementia is characterized by progressive dementia with fluctuations in cognitive performance. Family members or caregivers may report that the patient can carry on a conversation one day and the next day be confused and inattentive. Compared with those with Alzheimer dementia, patients with Lewy body dementia have better delayed recall but more problems with executive functioning (planning) and visuospatial skills (following an unfamiliar route, copying a figure).
Specialized imaging provides clues
Dementia with Lewy bodies is associated with diffuse brain atrophy, with no established characteristic pattern on structural neuroimaging with computed tomography (CT) or magnetic resonance imaging (MRI).17 The contrast agent ioflupane iodine-123 injection (DaTscan) used with single-photon emission CT (SPECT) detects dopamine transporters, which are reduced in parkinsonian syndromes. The scan can also help differentiate between Alzheimer dementia and Lewy body dementia by detecting the loss of functional dopaminergic terminals in the striatum in Lewy body dementia. Alpha-synuclein imaging may become another useful diagnostic tool in the future.
Alzheimer medications may help in dementia with Lewy bodies
Medications with anticholinergic effects and dopamine agonists should be discontinued because of possible effects on cognitive function and parkinsonism. In one clinical trial,18 rivastigmine (Exelon) was found to help cognitive functioning as well as reduce psychotic symptoms in dementia with Lewy bodies, although a recent Cochrane review could not support the evidence for use of all cholinesterase inhibitors in Lewy body dementia.19 In another trial,20 memantine (Namenda) was found to improve global clinical status and behavioral symptoms of Lewy body dementia.
Treating hallucinations of dementia with Lewy bodies
Patients with dementia with Lewy bodies are extremely sensitive to the extrapyramidal side effects of neuroleptic drugs. Some evidence indicates that the atypical antipsychotic drug quetiapine (Seroquel) helps with prominent and disturbing psychotic features and is less likely to worsen parkinsonism than other antipsychotics.21 The best evidence is for clozapine (Clozaril) as a treatment for hallucinations in Parkinson dementia, but the possible side effect of agranulocytosis limits its clinical use. Other atypical antipsychotics such as risperidone (Risperdal) and olanzapine (Zyprexa) are not recommended.22
PROGRESSIVE SUPRANUCLEAR PALSY
Progressive supranuclear palsy is a sporadic atypical parkinsonian disorder with onset between age 50 and 70. Familial cases are infrequent.
Progressive supranuclear palsy presents as early postural instability, vertical supranuclear gaze palsy, and axial muscle rigidity in the first few years. Disease progression is gradual: one study of 50 patients found that the median time from onset to the first key motor impairment (unintelligible speech, no independent walking, inability to stand unassisted, wheelchair-bound, or recommendation for feeding tube placement) was 4 years.23
Histologically, progressive supranuclear palsy is characterized by accumulation of tau protein aggregates in the basal ganglia, brainstem, and cerebral cortex. The degenerative process involves dopaminergic, cholinergic, and gamma-aminobutyric acid (GABA)-ergic neurons.24
Gait and balance problems predominate early in progressive supranuclear palsy
The most commonly used diagnostic criteria are from the National Institute of Neurological Disorders and Stroke. The diagnosis of probable progressive supranuclear palsy requires vertical gaze palsy and falls or the tendency to fall within the first year of disease onset and exclusion of other causes.
The earliest symptom is usually gait and balance impairment.25 Falls (usually backward) and postural instability occur during the first year in 58% of patients.26 Instead of turning en bloc as in Parkinson disease, patients with progressive supranuclear palsy tend to pivot quickly. Patients may also have a coarse groaning voice and moaning. Insomnia has been reported, but rapid-eye-movement sleep behavior disorders are infrequent (unlike in Parkinson disease, multiple system atrophy, and Lewy body dementia).27
Apathy and extreme mood swings
Cognitive impairment is seen in 50% of patients in the early stage of progressive supranuclear palsy. It mostly involves the frontal lobe, including frontal behavioral disturbances (eg, apathy in 91% of patients26 or pseudobulbar affect and extreme emotional lability) and deficits in abstract thoughts or verbal fluency (to test this, patients are asked to say as many words as possible from a category in a given time). Ideomotor apraxia (inability to correctly imitate hand gestures and voluntarily pantomime tool use, such as pretending to brush hair) is rare, despite corticobasal degeneration.28
Vertical gaze palsy
The hallmark of progressive supranuclear palsy is vertical gaze palsy. Initially, this involves slowing of vertical saccades, followed by diminished vertical gaze and more characteristic downward gaze palsy. These findings may develop over 3 to 4 years. Vertical gaze palsy leads to spilling food and tripping while walking.
The gaze abnormality combined with rare blinking and facial dystonia form the classic facial expression of astonishment called “leonine facies.” The face is stiff and deeply furrowed, with a look of surprise.
Axial (especially neck) rigidity is more prominent than limb rigidity. Retrocollis (the head is drawn back) occurs in less than 25% of patients. Parkinsonian features such as bradykinesia affect nearly half of patients by the time of diagnosis.
Instead of the classic symptoms of progressive supranuclear palsy, about one-third of patients present with progressive supranuclear palsy-parkinsonism, which involves asymmetric parkinsonism that initially responds to levodopa.29
MRI shows ‘hummingbird sign’
Brain MRI shows atrophy of the brainstem, particularly the midbrain. Thinning of the superior part of the midbrain and dilation of the third ventricle (“hummingbird sign” on sagittal sections or “morning glory flower” on axial sections) support a diagnosis of progressive supranuclear palsy and differentiate it from Parkinson disease and other atypical parkinsonian disorders.30,31
Levodopa ineffective for supranuclear palsy
There is no treatment to slow progressive supranuclear palsy. Even in high doses, levodopa rarely alleviates parkinsonian features in a clinically meaningful way.26 Successful experimental biologic therapies have been studied in animal models.32 Davunetide is thought to help with neuronal integrity and cell survival through the stabilization of microtubules in preclinical studies, but it has not been used in clinical practice.33
CORTICOBASAL DEGENERATION
Corticobasal degeneration is a progressive, asymmetric movement disorder often manifesting initially with cognitive or behavioral impairment. It is associated with abnormality of the cytoskeleton protein tau. Onset is usually after age 60.
Asymmetric movement disorder with cognitive dysfunction
This diagnosis is clinical. Diagnostic criteria proposed in 2003 include the following core features34:
- Insidious onset and progressive course
- No identifiable cause
- Cortical dysfunction with at least one of the following: apraxia, alien limb phenomenon (one limb moves involuntarily with complex movements, eg, grabbing the other hand), cortical sensory loss, visual hemineglect, nonfluent aphasia
- Extrapyramidal dysfunction: focal rigidity unresponsive to levodopa, asymmetric dystonia.
An international consortium has developed more specific clinical research criteria for probable and possible corticobasal degeneration.35 In a series of 147 patients, the following clinical features were found: parkinsonism (100%), higher cortical dysfunction (93%), dyspraxia (82%), gait disorder (80%), unilateral limb dystonia (71%), tremor (55%), and dementia (25%).36
Behavioral problems commonly include depression; apathy, irritability, and agitation are also reported.37
Cognitive testing may reveal deficits in frontal-parietal cognitive domains including attention and concentration, executive function, verbal fluency, and visuospatial skills.38 Learning disabilities may be improved with verbal cueing (in contrast to Alzheimer disease). Patients may also have impaired graphesthesia (the ability to recognize writing on the skin only by the sensation of touch).39,40
Motor examination may reveal marked asymmetry. Hand, limb, speech, and gait apraxias are common. Gait is typically slow, with short steps and shuffling, and a wide-based or freezing gait. Arm swing may be absent on one side.
Asymmetric cortical atrophy
Early on, MRI may be normal. As the disease progresses, asymmetric cortical atrophy may be seen, especially in the posterior frontal and parietal lobes.
Levodopa ineffective in corticobasal degeneration
Corticobasal degeneration responds poorly to levodopa. Botulinum toxin has been used to help with dystonia and limb pain.
MULTIPLE SYSTEM ATROPHY
Multiple system atrophy is another atypical parkinsonian disorder, most often diagnosed in men over age 60. It is characterized by sporadic parkinsonism, cerebellar signs (involving balance and coordination), pyramidal tract dysfunction, and autonomic insufficiency in varying combinations. Two major subtypes are recognized, depending on whether the predominating presenting features are cerebellar signs or parkinsonism. In contrast to dementia with Lewy bodies, psychiatric symptoms are not a major feature, except possibly depression.41
Diagnosis requires a sporadic progressive disorder that has features of autonomic failure and poor response of parkinsonism or cerebellar ataxia to levodopa.42
Multiple system atrophy is usually not associated with dementia in the early stages, but patients develop deficits in learning, recognition, memory, and verbal fluency as the disease progresses.43 Rapid-eye-movement sleep behavior disorder has been reported in more than half of patients.44
A neurologic examination provides clues
Parkinsonian features are usually symmetric, in contrast to idiopathic Parkinson disease. These signs may include akinesia with rigidity, postural instability, hypokinetic speech, and tremor.
Cerebellar signs include nystagmus and dysarthria (speech disturbance), and gait and limb ataxia.
Pyramidal features include extensor plantar responses and hyperreflexia.
Autonomic dysfunction includes orthostatic hypotension, bladder and rectal atony, loss of sweating, urinary or fecal incontinence, and erectile dysfunction.
Electromyography may demonstrate decreased anal sphincter tone.
MRI shows atrophy of putamen and pons
Brain MRI may show atrophy of the putamen (hypointensity of the putamen with a hyperintense rim). Pons atrophy may also be present, revealing a “hot cross bun” sign in axial images. These combined findings have specificity above 90% but limited sensitivity. These signs are useful to distinguish multiple system atrophy from Parkinson dementia, but their absence does not exclude the diagnosis of multiple system atrophy.45,46
Multiple system atrophy typically responds poorly to levodopa
Levodopa may improve movement and rigidity, but many respond poorly to treatment or lose response after a few years. Fludrocortisone (Florinef) or vasoconstrictors such as midodrine (Orvaten, Proamatine) may help with orthostatic hypotension.47,48
PARKINSON DISEASE DEMENTIA
Dementia eventually develops in most patients with Parkinson disease. Older age and the akinetic rigid form of the disease are associated with higher risk. Diagnosis of idiopathic Parkinson disease before the development of dementia is essential for the diagnosis.
The Movement Disorder Society Task Force has developed new diagnostic criteria.49 Deficits must be present in at least two of the four core cognitive domains (attention, memory, executive, and visuospatial functions) and must be severe enough to affect daily functioning.
Behavioral symptoms such as affective changes, hallucinations, and apathy are common.
MRI shows characteristic brain atrophy in Parkinson disease dementia
MRI shows reduced gray matter volume in the frontal lobe in patients with Parkinson disease without dementia compared with controls. In Parkinson disease dementia, reduced volume extends to temporal, occipital, and subcortical areas. No significant volumetric differences have been observed in Parkinson dementia compared with dementia with Lewy bodies.50 A greater decrease of glucose metabolism has been found in the inferior parietal and occipital lobes in Parkinson disease dementia than in Parkinson disease without dementia.51
Rivastigmine effective for dementia
A Cochrane review supports the use of acetylcholinesterase inhibitors in patients with Parkinson disease dementia, with a positive impact on global assessment, cognitive function, behavioral disturbance, and activities of daily living rating scales.19 At this time, rivastigmine is the only FDA-approved cholinesterase inhibitor for treating Parkinson disease dementia. In clinical trials, memantine did not improve global clinical status or behavioral symptoms of dementia of Parkinson disease.51
FRONTOTEMPORAL DEMENTIA
Frontotemporal dementia frequently starts before age 65 and accounts for 20% to 50% of dementias in this age group.52 Recognition of the condition in older patients is also growing.53 Frontotemporal dementia encompasses a spectrum of dementias, including behavioral variant frontotemporal dementia, semantic dementia, and progressive nonfluent aphasia.54
Gradual onset of uncharacteristic behaviors
Accepted diagnostic criteria include core features of gradual onset, early decline in social and interpersonal conduct, early impairment of self-regulation, emotional blunting, and loss of insight. Many patients are diagnosed with psychiatric conditions. Changes reported by family and caregivers typically deviate substantially from the person’s usual behavior, such as impulsive and inappropriate behaviors or complete withdrawal and apathy.
Language sometimes affected in frontotemporal dementia
Language impairment may be present in some variants. Behavioral and language changes often accompany other forms of dementia (Alzheimer disease, vascular dementia, primary progressive aphasia), making diagnosis more challenging. Office-based testing often does not reveal any deficits, although the Frontal Behavioral Inventory may help.55 A referral to a clinical neuropsychologist may help identify and quantify cognitive impairments.
MRI shows frontotemporal lobes affected
Structural neuroimaging may not reveal abnormalities initially, but with progression, atrophy may be seen in the frontal and temporal lobes. Functional neuroimaging (positron emission tomography, brain SPECT, functional MRI) show hypometabolism in the same areas.
Treat symptoms
There are no specific FDA-approved therapies for frontotemporal dementia. Acetylcholinesterase inhibitors can help progressive nonfluent aphasia in some cases. Selective serotonin reuptake inhibitors may alleviate depressive symptoms, and low doses of atypical antipsychotic medications may help with impulsivity, disinhibition, and aggressive or disruptive behaviors.56
PRIMARY PROGRESSIVE APHASIA
Language impairment predominates
Primary progressive aphasia is a rare form of dementia in which symptoms typically develop around age 60. Pathology is varied. In a study of 60 patients with initial clinical symptoms of primary progressive aphasia, postmortem histology of brain tissue revealed various findings, including those consistent with Alzheimer pathology and motor neuron diseasetype inclusions.57
Patients typically present with expressive language problems as the primary deficit for the first 2 years of the disease, with preservation in other cognitive areas such as memory, visuospatial skills, and executive function.58 Office-based testing may overstate the severity of the dementia, given the dependence of performance on intact language.
It is important to distinguish primary progressive aphasia from other dementias that also affect language. In the frontal variant of frontotemporal dementia, the primary language problem is anomia (inability to name objects) or diminished speech output, which may be accompanied by behavioral problems. Semantic dementia affects word recognition as well as comprehension. In Alzheimer disease, language may be affected along with memory and other areas of cognitive function.
Imaging shows focal degeneration in the left hemisphere
Structural neuroimaging does not initially reveal any deficits, but later it may reveal atrophy in the frontal, perisylvian complex, and temporal areas of the left hemisphere, reflecting the focal nature of the degeneration.59 Functional neuroimaging (positron emission tomography, SPECT) may reveal hypometabolism or diminished blood flow in these areas prior to changes in structural neuroimaging.60
Other communication methods may help
There are no FDA-approved therapies for primary progressive aphasia. Off-label use of some agents (eg, selective serotonin reuptake inhibitors and small doses of antipsychotic medications) has been found useful in small trials.56 Patients may benefit from learning other forms of communication, such as using sign language, laminated cards with printed words or pictures, or artificial voice synthesizers, to express their needs.
NORMAL-PRESSURE HYDROCEPHALUS
Classic triad: Gait, cognition, incontinence
With the onset of symptoms in the sixth or seventh decade, normal-pressure hydrocephalus affects less than 1% of people age 65 and older. It represents up to 5% of dementias, although estimates are influenced by the varied criteria for diagnosis.61 It is characterized by the classic triad of gait impairment, cognitive impairment, and urinary frequency or incontinence.62
Symptoms progress over a period of years, with gait impairment often predominating. As this triad is common in the geriatric population, identifying other explanations is important. Gait impairment caused by spinal stenosis, peripheral neuropathy, or parkinsonism should be explored. Cognitive impairment could be due to depression, Alzheimer disease, or other forms of dementia. Urinary symptoms may be related to detrusor instability or an enlarged prostate.
Gait impairment initially manifests as slowing of gait, but progresses to difficulty with gait initiation. Gait tends to be wide-based (stance more than 1 foot wide).
Cognitive impairment is typically subcortical, manifested as slowed processing speed and impaired executive function. Recall and working memory may be impaired.
Enlarged ventricles seen on imaging in normal-pressure hydrocephalus
Structural neuroimaging reveals enlarged ventricles (Evan’s ratio > 0.358). This can be difficult to distinguish from ventriculomegaly due to cerebral atrophy; assessing the callosal angle on MRI may distinguish the two.63,64 Diagnosis of normal-pressure hydrocephalus can be confirmed using a cerebrospinal fluid infusion test to assess resistance of fluid to resorption.65
Treat with cerebrospinal fluid drainage
Specific tests should be performed to determine candidacy for surgery. These include a high-volume lumbar puncture (40 to 50 mL) or a trial of external lumbar drainage (10 mL per hour for 48 to 72 hours).65 Definitive treatment is surgical placement of a shunt to allow cerebrospinal fluid to drain into the atria or peritoneal cavity.
Surgery may improve gait, but cognitive symptoms often remain,66 and clinical decline may occur after the shunt is placed. Once gait dysfunction is resolved, other explanations for cognitive impairment or residual gait impairment should be considered. An underlying reason for progression of normal-pressure hydrocephalus symptoms after surgical intervention should be identified.67
RAPIDLY PROGRESSIVE DEMENTIAS
Rapidly progressive dementias are among the most challenging of dementing illnesses. They are characterized by a subacute course and an accelerated rate of decline, developing in less than 2 years. Evaluation should typically be more comprehensive than for other types of dementia. The main goal is to diagnose potentially treatable conditions, such as Hashimoto encephalopathy or paraneoplastic limbic encephalitis, and to distinguish these conditions from diseases with a very poor prognosis, such as Creutzfeldt-Jakob disease.
Creutzfeldt-Jakob disease
Creutzfeldt-Jakob disease is a fatal prion-related neurodegenerative illness. Sporadic disease is most common, but variant, familial, and iatrogenic types have been reported. The most common initial symptoms in sporadic disease are cognitive (39%), cerebellar (21%), behavioral (20%), constitutional (20%), sensory (11%), motor (9%), and visual (7%).68
Chronic neurodegenerative diseases can be misdiagnosed as Creutzfeldt-Jakob disease because of an atypical time course and multi-system neurologic findings.
The US Centers for Disease Control and Prevention has adopted criteria for diagnosing probable Creutzfeldt-Jakob disease (Table 3). Routine investigations should also not suggest an alternative diagnosis.69
Autoimmune diseases
Autoimmune conditions may present as a rapidly progressive dementia, including Hashimoto encephalopathy and antibody-mediated limbic encephalitis, either associated with cancer (paraneoplastic) or without cancer (nonparaneoplastic).
Paraneoplastic limbic encephalitis is a group of inflammatory conditions involving antibodies produced within the cerebrospinal fluid and serum resulting in neurologic symptoms. These antibodies react against proteins expressed mostly by a tumor somewhere else in the body.70
Hashimoto encephalitis is a subacute to chronic encephalopathy that may present as dementia with abnormally high levels of thyroid antibodies. The symptoms can vary from confusion to psychosis. There are two main presentations: one involves a relapsing-remitting course with stroke-like episodes (27% of patients) and the second consists of insidious onset of seizures (66% of patients).
Diagnosis involves testing for elevated anti-thyroid peroxidase and thyroglobulin antibodies. MRI findings are nonspecific. Hashimoto encephalitis responds to treatment with corticosteroids, plasmapheresis, or immunosuppressive therapy.71
Dementia is not always due to Alzheimer disease. An accurate diagnosis is important, as the various causative conditions can differ in their course and treatment.
Dementia refers to cognitive impairment severe enough to interfere with the ability to independently perform activities of daily living. It can occur at any age but is most common after age 60. Some studies estimate that 13.9% of people age 71 and older have some form of dementia.1 The prevalence increases with age, ranging from 5% at age 70 to 79 to 37% at age 90 and older.1
Alzheimer disease accounts for about 60% to 80% of cases,2 or an estimated 4.7 million people age 65 and older in the United States, a number anticipated to climb to 13.8 million by 2050.3
Other types of dementia are less often considered and are challenging to recognize, although many have distinct characteristics. This article summarizes the features and management of the more common non-Alzheimer dementias:
- Vascular dementia
- Dementia with Lewy bodies
- Progressive supranuclear palsy
- Corticobasal degeneration
- Multiple system atrophy
- Parkinson disease dementia
- Frontotemporal dementia
- Primary progressive aphasia
- Normal-pressure hydrocephalus
- Rapidly progressive dementia (ie, Creutzfeld-Jakob disease, autoimmune disease).
VASCULAR DEMENTIA
After Alzheimer disease, vascular dementia is the most common dementia, accounting for about 20% to 30% of cases. Clinical criteria have not been widely accepted, although several have been published, including those in the Diagnostic and Statistical Manual of Mental Disorders (Fourth Edition) and the National Institute of Neurological and Communicative Diseases and Stroke-Association Internationale pour la Recherche et l’Enseignement en Neurosciences.
Risk factors for vascular dementia include cerebrovascular disease (hypertension, diabetes, hyperlipidemia) and coexisting conditions related to atherosclerosis (coronary artery disease, peripheral artery disease).
The Hachinski Ischemic Score is a good bedside tool to help differentiate Alzheimer dementia from vascular dementia.5
Sudden onset and stepwise decline
Vascular dementia often presents as a sudden and stepwise progression of cognitive deficits that stabilize and that are caused by vascular insults (Table 1).6–10 Some patients have continuous decline after a vascular event, indicating that Alzheimer dementia may also be present. Dementia is then defined as a mixed type.
Behavioral problems such as physical aggression, hallucinations, paranoia, and mood fluctuations are common.11
Deficits depend on vascular areas affected
Cognitive deficits are heterogeneous and are often related to the location of the vascular insult. Involvement of subcortical areas may result in executive dysfunction, slowed processing speed, and behavioral changes.12
Executive dysfunction may be identified using the Trail Making Test (Part B) or the Executive Interview (EXIT25). Office-based tools such as the Folstein Mini-Mental State Examination, the Montreal Cognitive Assessment, or the St. Louis University Mental Status Examination may also uncover these deficits.
Focal neurologic deficits may be found on clinical examination.
Structural neuroimaging may identify small strokes in areas of the brain affecting cognitive function or occlusion of a larger vessel associated with more profound neurologic deficits. Neuroimaging findings may not correlate with any significant decline noted by the patient, suggesting “silent” strokes.
Treat symptoms and manage risk factors
Although the US Food and Drug Administration (FDA) has not approved any pharmacotherapy for vascular dementia, commonly prescribed cognitive enhancers have demonstrated some benefit.13
Behavioral problems such as aggression can be disturbing to the patient and the caregiver. Nonpharmacologic methods (eg, redirection, rescheduling care activities to avoid conflict, avoiding issues that lead to agitation) should be tried first to address these problems.
Drug therapy may be used off-label for neuropsychiatric symptoms such as hallucinations, delusions, and combativeness, but clinical trials of these agents for this purpose have shown mixed results,14 and their use is often associated with significant risk.15 Antipsychotic drugs are associated with a risk of death and pneumonia when prescribed for dementia. Many also carry a risk of QT prolongation, which is particularly concerning for patients with coronary artery disease or rhythm disturbances.
The key to reducing further decline is to optimize management of vascular risk factors to reduce stroke risk.
DEMENTIA WITH LEWY BODIES
Dementia with Lewy bodies, the next most common neurodegenerative dementia in the elderly, is characterized by progressive loss of cognitive function, prominent visual hallucinations, and parkinsonism (Table 1).6 Disease progression usually occurs over years but can be more rapid than in Alzheimer disease.
Alpha-synucleinopathy results in dysfunction of synaptic vesicles in presynaptic terminals. Lewy bodies may be diffusely spread in cortical and subcortical areas (appearing as spherical masses).
Visual hallucinations are typical
The McKeith criteria16 are the gold standard for diagnosing probable Lewy body dementia, based on clinical and imaging features (Table 2).
Visual hallucinations are usually well formed and detailed. They may initially be pleasant (eg, seeing children and little people) but may evolve to be accompanied by persecutory delusions.
Parkinsonism develops with or after dementia with Lewy bodies
Dementia with Lewy bodies and Parkinson disease dementia share many clinical and pathologic features; Parkinson dementia also is associated with cortical Lewy bodies.
Parkinsonian features include bradykinesia, masked facies, and rigidity. Resting tremor is less common.
The third report of the Dementia With Lewy Bodies Consortium recommends that the condition be diagnosed if dementia occurs before or concurrently with parkinsonism, and dementia with Parkinson disease should be diagnosed if dementia occurs in the context of well-established Parkinson disease.16 The development of dementia within 12 months of extrapyramidal signs suggests dementia with Lewy bodies.
Cognitive deficits fluctuate
Cognitive impairment in Lewy body dementia is characterized by progressive dementia with fluctuations in cognitive performance. Family members or caregivers may report that the patient can carry on a conversation one day and the next day be confused and inattentive. Compared with those with Alzheimer dementia, patients with Lewy body dementia have better delayed recall but more problems with executive functioning (planning) and visuospatial skills (following an unfamiliar route, copying a figure).
Specialized imaging provides clues
Dementia with Lewy bodies is associated with diffuse brain atrophy, with no established characteristic pattern on structural neuroimaging with computed tomography (CT) or magnetic resonance imaging (MRI).17 The contrast agent ioflupane iodine-123 injection (DaTscan) used with single-photon emission CT (SPECT) detects dopamine transporters, which are reduced in parkinsonian syndromes. The scan can also help differentiate between Alzheimer dementia and Lewy body dementia by detecting the loss of functional dopaminergic terminals in the striatum in Lewy body dementia. Alpha-synuclein imaging may become another useful diagnostic tool in the future.
Alzheimer medications may help in dementia with Lewy bodies
Medications with anticholinergic effects and dopamine agonists should be discontinued because of possible effects on cognitive function and parkinsonism. In one clinical trial,18 rivastigmine (Exelon) was found to help cognitive functioning as well as reduce psychotic symptoms in dementia with Lewy bodies, although a recent Cochrane review could not support the evidence for use of all cholinesterase inhibitors in Lewy body dementia.19 In another trial,20 memantine (Namenda) was found to improve global clinical status and behavioral symptoms of Lewy body dementia.
Treating hallucinations of dementia with Lewy bodies
Patients with dementia with Lewy bodies are extremely sensitive to the extrapyramidal side effects of neuroleptic drugs. Some evidence indicates that the atypical antipsychotic drug quetiapine (Seroquel) helps with prominent and disturbing psychotic features and is less likely to worsen parkinsonism than other antipsychotics.21 The best evidence is for clozapine (Clozaril) as a treatment for hallucinations in Parkinson dementia, but the possible side effect of agranulocytosis limits its clinical use. Other atypical antipsychotics such as risperidone (Risperdal) and olanzapine (Zyprexa) are not recommended.22
PROGRESSIVE SUPRANUCLEAR PALSY
Progressive supranuclear palsy is a sporadic atypical parkinsonian disorder with onset between age 50 and 70. Familial cases are infrequent.
Progressive supranuclear palsy presents as early postural instability, vertical supranuclear gaze palsy, and axial muscle rigidity in the first few years. Disease progression is gradual: one study of 50 patients found that the median time from onset to the first key motor impairment (unintelligible speech, no independent walking, inability to stand unassisted, wheelchair-bound, or recommendation for feeding tube placement) was 4 years.23
Histologically, progressive supranuclear palsy is characterized by accumulation of tau protein aggregates in the basal ganglia, brainstem, and cerebral cortex. The degenerative process involves dopaminergic, cholinergic, and gamma-aminobutyric acid (GABA)-ergic neurons.24
Gait and balance problems predominate early in progressive supranuclear palsy
The most commonly used diagnostic criteria are from the National Institute of Neurological Disorders and Stroke. The diagnosis of probable progressive supranuclear palsy requires vertical gaze palsy and falls or the tendency to fall within the first year of disease onset and exclusion of other causes.
The earliest symptom is usually gait and balance impairment.25 Falls (usually backward) and postural instability occur during the first year in 58% of patients.26 Instead of turning en bloc as in Parkinson disease, patients with progressive supranuclear palsy tend to pivot quickly. Patients may also have a coarse groaning voice and moaning. Insomnia has been reported, but rapid-eye-movement sleep behavior disorders are infrequent (unlike in Parkinson disease, multiple system atrophy, and Lewy body dementia).27
Apathy and extreme mood swings
Cognitive impairment is seen in 50% of patients in the early stage of progressive supranuclear palsy. It mostly involves the frontal lobe, including frontal behavioral disturbances (eg, apathy in 91% of patients26 or pseudobulbar affect and extreme emotional lability) and deficits in abstract thoughts or verbal fluency (to test this, patients are asked to say as many words as possible from a category in a given time). Ideomotor apraxia (inability to correctly imitate hand gestures and voluntarily pantomime tool use, such as pretending to brush hair) is rare, despite corticobasal degeneration.28
Vertical gaze palsy
The hallmark of progressive supranuclear palsy is vertical gaze palsy. Initially, this involves slowing of vertical saccades, followed by diminished vertical gaze and more characteristic downward gaze palsy. These findings may develop over 3 to 4 years. Vertical gaze palsy leads to spilling food and tripping while walking.
The gaze abnormality combined with rare blinking and facial dystonia form the classic facial expression of astonishment called “leonine facies.” The face is stiff and deeply furrowed, with a look of surprise.
Axial (especially neck) rigidity is more prominent than limb rigidity. Retrocollis (the head is drawn back) occurs in less than 25% of patients. Parkinsonian features such as bradykinesia affect nearly half of patients by the time of diagnosis.
Instead of the classic symptoms of progressive supranuclear palsy, about one-third of patients present with progressive supranuclear palsy-parkinsonism, which involves asymmetric parkinsonism that initially responds to levodopa.29
MRI shows ‘hummingbird sign’
Brain MRI shows atrophy of the brainstem, particularly the midbrain. Thinning of the superior part of the midbrain and dilation of the third ventricle (“hummingbird sign” on sagittal sections or “morning glory flower” on axial sections) support a diagnosis of progressive supranuclear palsy and differentiate it from Parkinson disease and other atypical parkinsonian disorders.30,31
Levodopa ineffective for supranuclear palsy
There is no treatment to slow progressive supranuclear palsy. Even in high doses, levodopa rarely alleviates parkinsonian features in a clinically meaningful way.26 Successful experimental biologic therapies have been studied in animal models.32 Davunetide is thought to help with neuronal integrity and cell survival through the stabilization of microtubules in preclinical studies, but it has not been used in clinical practice.33
CORTICOBASAL DEGENERATION
Corticobasal degeneration is a progressive, asymmetric movement disorder often manifesting initially with cognitive or behavioral impairment. It is associated with abnormality of the cytoskeleton protein tau. Onset is usually after age 60.
Asymmetric movement disorder with cognitive dysfunction
This diagnosis is clinical. Diagnostic criteria proposed in 2003 include the following core features34:
- Insidious onset and progressive course
- No identifiable cause
- Cortical dysfunction with at least one of the following: apraxia, alien limb phenomenon (one limb moves involuntarily with complex movements, eg, grabbing the other hand), cortical sensory loss, visual hemineglect, nonfluent aphasia
- Extrapyramidal dysfunction: focal rigidity unresponsive to levodopa, asymmetric dystonia.
An international consortium has developed more specific clinical research criteria for probable and possible corticobasal degeneration.35 In a series of 147 patients, the following clinical features were found: parkinsonism (100%), higher cortical dysfunction (93%), dyspraxia (82%), gait disorder (80%), unilateral limb dystonia (71%), tremor (55%), and dementia (25%).36
Behavioral problems commonly include depression; apathy, irritability, and agitation are also reported.37
Cognitive testing may reveal deficits in frontal-parietal cognitive domains including attention and concentration, executive function, verbal fluency, and visuospatial skills.38 Learning disabilities may be improved with verbal cueing (in contrast to Alzheimer disease). Patients may also have impaired graphesthesia (the ability to recognize writing on the skin only by the sensation of touch).39,40
Motor examination may reveal marked asymmetry. Hand, limb, speech, and gait apraxias are common. Gait is typically slow, with short steps and shuffling, and a wide-based or freezing gait. Arm swing may be absent on one side.
Asymmetric cortical atrophy
Early on, MRI may be normal. As the disease progresses, asymmetric cortical atrophy may be seen, especially in the posterior frontal and parietal lobes.
Levodopa ineffective in corticobasal degeneration
Corticobasal degeneration responds poorly to levodopa. Botulinum toxin has been used to help with dystonia and limb pain.
MULTIPLE SYSTEM ATROPHY
Multiple system atrophy is another atypical parkinsonian disorder, most often diagnosed in men over age 60. It is characterized by sporadic parkinsonism, cerebellar signs (involving balance and coordination), pyramidal tract dysfunction, and autonomic insufficiency in varying combinations. Two major subtypes are recognized, depending on whether the predominating presenting features are cerebellar signs or parkinsonism. In contrast to dementia with Lewy bodies, psychiatric symptoms are not a major feature, except possibly depression.41
Diagnosis requires a sporadic progressive disorder that has features of autonomic failure and poor response of parkinsonism or cerebellar ataxia to levodopa.42
Multiple system atrophy is usually not associated with dementia in the early stages, but patients develop deficits in learning, recognition, memory, and verbal fluency as the disease progresses.43 Rapid-eye-movement sleep behavior disorder has been reported in more than half of patients.44
A neurologic examination provides clues
Parkinsonian features are usually symmetric, in contrast to idiopathic Parkinson disease. These signs may include akinesia with rigidity, postural instability, hypokinetic speech, and tremor.
Cerebellar signs include nystagmus and dysarthria (speech disturbance), and gait and limb ataxia.
Pyramidal features include extensor plantar responses and hyperreflexia.
Autonomic dysfunction includes orthostatic hypotension, bladder and rectal atony, loss of sweating, urinary or fecal incontinence, and erectile dysfunction.
Electromyography may demonstrate decreased anal sphincter tone.
MRI shows atrophy of putamen and pons
Brain MRI may show atrophy of the putamen (hypointensity of the putamen with a hyperintense rim). Pons atrophy may also be present, revealing a “hot cross bun” sign in axial images. These combined findings have specificity above 90% but limited sensitivity. These signs are useful to distinguish multiple system atrophy from Parkinson dementia, but their absence does not exclude the diagnosis of multiple system atrophy.45,46
Multiple system atrophy typically responds poorly to levodopa
Levodopa may improve movement and rigidity, but many respond poorly to treatment or lose response after a few years. Fludrocortisone (Florinef) or vasoconstrictors such as midodrine (Orvaten, Proamatine) may help with orthostatic hypotension.47,48
PARKINSON DISEASE DEMENTIA
Dementia eventually develops in most patients with Parkinson disease. Older age and the akinetic rigid form of the disease are associated with higher risk. Diagnosis of idiopathic Parkinson disease before the development of dementia is essential for the diagnosis.
The Movement Disorder Society Task Force has developed new diagnostic criteria.49 Deficits must be present in at least two of the four core cognitive domains (attention, memory, executive, and visuospatial functions) and must be severe enough to affect daily functioning.
Behavioral symptoms such as affective changes, hallucinations, and apathy are common.
MRI shows characteristic brain atrophy in Parkinson disease dementia
MRI shows reduced gray matter volume in the frontal lobe in patients with Parkinson disease without dementia compared with controls. In Parkinson disease dementia, reduced volume extends to temporal, occipital, and subcortical areas. No significant volumetric differences have been observed in Parkinson dementia compared with dementia with Lewy bodies.50 A greater decrease of glucose metabolism has been found in the inferior parietal and occipital lobes in Parkinson disease dementia than in Parkinson disease without dementia.51
Rivastigmine effective for dementia
A Cochrane review supports the use of acetylcholinesterase inhibitors in patients with Parkinson disease dementia, with a positive impact on global assessment, cognitive function, behavioral disturbance, and activities of daily living rating scales.19 At this time, rivastigmine is the only FDA-approved cholinesterase inhibitor for treating Parkinson disease dementia. In clinical trials, memantine did not improve global clinical status or behavioral symptoms of dementia of Parkinson disease.51
FRONTOTEMPORAL DEMENTIA
Frontotemporal dementia frequently starts before age 65 and accounts for 20% to 50% of dementias in this age group.52 Recognition of the condition in older patients is also growing.53 Frontotemporal dementia encompasses a spectrum of dementias, including behavioral variant frontotemporal dementia, semantic dementia, and progressive nonfluent aphasia.54
Gradual onset of uncharacteristic behaviors
Accepted diagnostic criteria include core features of gradual onset, early decline in social and interpersonal conduct, early impairment of self-regulation, emotional blunting, and loss of insight. Many patients are diagnosed with psychiatric conditions. Changes reported by family and caregivers typically deviate substantially from the person’s usual behavior, such as impulsive and inappropriate behaviors or complete withdrawal and apathy.
Language sometimes affected in frontotemporal dementia
Language impairment may be present in some variants. Behavioral and language changes often accompany other forms of dementia (Alzheimer disease, vascular dementia, primary progressive aphasia), making diagnosis more challenging. Office-based testing often does not reveal any deficits, although the Frontal Behavioral Inventory may help.55 A referral to a clinical neuropsychologist may help identify and quantify cognitive impairments.
MRI shows frontotemporal lobes affected
Structural neuroimaging may not reveal abnormalities initially, but with progression, atrophy may be seen in the frontal and temporal lobes. Functional neuroimaging (positron emission tomography, brain SPECT, functional MRI) show hypometabolism in the same areas.
Treat symptoms
There are no specific FDA-approved therapies for frontotemporal dementia. Acetylcholinesterase inhibitors can help progressive nonfluent aphasia in some cases. Selective serotonin reuptake inhibitors may alleviate depressive symptoms, and low doses of atypical antipsychotic medications may help with impulsivity, disinhibition, and aggressive or disruptive behaviors.56
PRIMARY PROGRESSIVE APHASIA
Language impairment predominates
Primary progressive aphasia is a rare form of dementia in which symptoms typically develop around age 60. Pathology is varied. In a study of 60 patients with initial clinical symptoms of primary progressive aphasia, postmortem histology of brain tissue revealed various findings, including those consistent with Alzheimer pathology and motor neuron diseasetype inclusions.57
Patients typically present with expressive language problems as the primary deficit for the first 2 years of the disease, with preservation in other cognitive areas such as memory, visuospatial skills, and executive function.58 Office-based testing may overstate the severity of the dementia, given the dependence of performance on intact language.
It is important to distinguish primary progressive aphasia from other dementias that also affect language. In the frontal variant of frontotemporal dementia, the primary language problem is anomia (inability to name objects) or diminished speech output, which may be accompanied by behavioral problems. Semantic dementia affects word recognition as well as comprehension. In Alzheimer disease, language may be affected along with memory and other areas of cognitive function.
Imaging shows focal degeneration in the left hemisphere
Structural neuroimaging does not initially reveal any deficits, but later it may reveal atrophy in the frontal, perisylvian complex, and temporal areas of the left hemisphere, reflecting the focal nature of the degeneration.59 Functional neuroimaging (positron emission tomography, SPECT) may reveal hypometabolism or diminished blood flow in these areas prior to changes in structural neuroimaging.60
Other communication methods may help
There are no FDA-approved therapies for primary progressive aphasia. Off-label use of some agents (eg, selective serotonin reuptake inhibitors and small doses of antipsychotic medications) has been found useful in small trials.56 Patients may benefit from learning other forms of communication, such as using sign language, laminated cards with printed words or pictures, or artificial voice synthesizers, to express their needs.
NORMAL-PRESSURE HYDROCEPHALUS
Classic triad: Gait, cognition, incontinence
With the onset of symptoms in the sixth or seventh decade, normal-pressure hydrocephalus affects less than 1% of people age 65 and older. It represents up to 5% of dementias, although estimates are influenced by the varied criteria for diagnosis.61 It is characterized by the classic triad of gait impairment, cognitive impairment, and urinary frequency or incontinence.62
Symptoms progress over a period of years, with gait impairment often predominating. As this triad is common in the geriatric population, identifying other explanations is important. Gait impairment caused by spinal stenosis, peripheral neuropathy, or parkinsonism should be explored. Cognitive impairment could be due to depression, Alzheimer disease, or other forms of dementia. Urinary symptoms may be related to detrusor instability or an enlarged prostate.
Gait impairment initially manifests as slowing of gait, but progresses to difficulty with gait initiation. Gait tends to be wide-based (stance more than 1 foot wide).
Cognitive impairment is typically subcortical, manifested as slowed processing speed and impaired executive function. Recall and working memory may be impaired.
Enlarged ventricles seen on imaging in normal-pressure hydrocephalus
Structural neuroimaging reveals enlarged ventricles (Evan’s ratio > 0.358). This can be difficult to distinguish from ventriculomegaly due to cerebral atrophy; assessing the callosal angle on MRI may distinguish the two.63,64 Diagnosis of normal-pressure hydrocephalus can be confirmed using a cerebrospinal fluid infusion test to assess resistance of fluid to resorption.65
Treat with cerebrospinal fluid drainage
Specific tests should be performed to determine candidacy for surgery. These include a high-volume lumbar puncture (40 to 50 mL) or a trial of external lumbar drainage (10 mL per hour for 48 to 72 hours).65 Definitive treatment is surgical placement of a shunt to allow cerebrospinal fluid to drain into the atria or peritoneal cavity.
Surgery may improve gait, but cognitive symptoms often remain,66 and clinical decline may occur after the shunt is placed. Once gait dysfunction is resolved, other explanations for cognitive impairment or residual gait impairment should be considered. An underlying reason for progression of normal-pressure hydrocephalus symptoms after surgical intervention should be identified.67
RAPIDLY PROGRESSIVE DEMENTIAS
Rapidly progressive dementias are among the most challenging of dementing illnesses. They are characterized by a subacute course and an accelerated rate of decline, developing in less than 2 years. Evaluation should typically be more comprehensive than for other types of dementia. The main goal is to diagnose potentially treatable conditions, such as Hashimoto encephalopathy or paraneoplastic limbic encephalitis, and to distinguish these conditions from diseases with a very poor prognosis, such as Creutzfeldt-Jakob disease.
Creutzfeldt-Jakob disease
Creutzfeldt-Jakob disease is a fatal prion-related neurodegenerative illness. Sporadic disease is most common, but variant, familial, and iatrogenic types have been reported. The most common initial symptoms in sporadic disease are cognitive (39%), cerebellar (21%), behavioral (20%), constitutional (20%), sensory (11%), motor (9%), and visual (7%).68
Chronic neurodegenerative diseases can be misdiagnosed as Creutzfeldt-Jakob disease because of an atypical time course and multi-system neurologic findings.
The US Centers for Disease Control and Prevention has adopted criteria for diagnosing probable Creutzfeldt-Jakob disease (Table 3). Routine investigations should also not suggest an alternative diagnosis.69
Autoimmune diseases
Autoimmune conditions may present as a rapidly progressive dementia, including Hashimoto encephalopathy and antibody-mediated limbic encephalitis, either associated with cancer (paraneoplastic) or without cancer (nonparaneoplastic).
Paraneoplastic limbic encephalitis is a group of inflammatory conditions involving antibodies produced within the cerebrospinal fluid and serum resulting in neurologic symptoms. These antibodies react against proteins expressed mostly by a tumor somewhere else in the body.70
Hashimoto encephalitis is a subacute to chronic encephalopathy that may present as dementia with abnormally high levels of thyroid antibodies. The symptoms can vary from confusion to psychosis. There are two main presentations: one involves a relapsing-remitting course with stroke-like episodes (27% of patients) and the second consists of insidious onset of seizures (66% of patients).
Diagnosis involves testing for elevated anti-thyroid peroxidase and thyroglobulin antibodies. MRI findings are nonspecific. Hashimoto encephalitis responds to treatment with corticosteroids, plasmapheresis, or immunosuppressive therapy.71
- Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology 2007; 29:125–132.
- Alzheimer’s Association. 2013 Alzheimer’s disease facts and figures. http://www.alz.org/downloads/facts_figures_2013.pdf. Accessed February 3, 2014.
- Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology 2013; 80:1778–1783.
- Hou CE, Carlin D, Miller BL. Non-Alzheimer’s disease dementias: anatomic, clinical, and molecular correlates. Can J Psychiatry 2004; 49:164–171.
- Hachinski VC, Iliff LD, Zilhka E, et al. Cerebral blood flow in dementia. Arch Neurol 1975; 32:632–637.
- Fereshtehnejad SM, Religa D, Westman E, Aarsland D, Lökk J, Eriksdotter M. Demography, diagnostics, and medication in dementia with Lewy bodies and Parkinson’s disease with dementia: data from the Swedish Dementia Quality Registry (SveDem). Neuropsychiatr Dis Treat 2013; 9:927–935.
- Nomura T, Inoue Y, Takigawa H, Nakashima K. Comparison of REM sleep behaviour disorder variables between patients with progressive supranuclear palsy and those with Parkinson’s disease. Parkinsonism Relat Disord 2012; 18:394–396.
- Davis PH, Golbe LI, Duvoisin RC, Schoenberg BS. Risk factors for progrssive supranuclear palsy. Neurology 1988; 38:1546–1552.
- Tousi B, Schuele SU, Subramanian T. A 46-year-old woman with rigidity and frequent falls. Cleve Clin J Med 2005; 72:57–63.
- Cooper AD, Josephs KA. Photophobia, visual hallucinations, and REM sleep behavior disorder in progressive supranuclear palsy and corticobasal degeneration: a prospective study. Parkinsonism Relat Disord 2009; 15:59–61.
- Lyketsos CG, Lopez O, Jones B, Fitzpatrick AL, Breitner J, DeKosky S. Prevalence of neuropsychiatric symptoms in dementia and mild cognitive impairment: results from the cardiovascular health study0. JAMA 2002; 288:1475–1483.
- Knopman DS. Dementia and cerebrovascular disease. Mayo Clin Proc 2006; 81:223–230.
- Erkinjuntti T, Kurz A, Gauthier S, Bullock R, Lilienfeld S, Damaraju CV. Efficacy of galantamine in probable vascular dementia and Alzheimer’s disease combined with cerebrovascular disease: a randomised trial. Lancet 2002; 359:1283–1290.
- Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA 2005; 293:596–608.
- Schneider LS, Dagerman K, Insel PS. Efficacy and adverse effects of atypical antipsychotics for dementia: meta-analysis of randomized, placebo-controlled trials. Am J Geriatr Psychiatry 2006; 14:191–210.
- McKeith IG, Dickson DW, Lowe J, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 2005; 65:1863–1872.
- Tartaglia MC, Rosen HJ, Miller BL. Neuroimaging in dementia. Neurotherapeutics 2011; 8:82–92.
- McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet 2000; 356:2031–2036.
- Rolinski M, Fox C, Maidment I, McShane R. Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson’s disease dementia and cognitive impairment in Parkinson’s disease. Cochrane Database Syst Rev 2012; 3:CD006504.
- Emre M, Tsolaki M, Bonucelli U, et al; on behalf of the 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol 2010; 9:969–977.
- Kurlan R, Cummings J, Raman R, Thal L; Alzheimer’s Disease Cooperative Study Group. Quetiapine for agitation or psychosis in patients with dementia and parkinsonism. Neurology 2007; 68:1356–1363.
- Ballard C, Aarsland D, Francis P, Corbett A. Neuropsychiatric symptoms in patients with dementias associated with cortical Lewy bodies: pathophysiology, clinical features, and pharmacological management. Drugs Aging 2013; 30:603–611.
- Goetz CG, Leurgans S, Lang AE, Litvan I. Progression of gait, speech and swallowing deficits in progressive supranuclear palsy. Neurology 2003; 60:917–922.
- Kasashima S, Oda Y. Cholinergic neuronal loss in the basal forebrain and mesopontine tegmentum of progressive supranuclear palsy and corticobasal degeneration. Acta Neuropathol 2003; 105:117–124.
- Fahn S, Jankovic J, Hallett M, editors. Principles and Practice of Movement Disorders. 2nd ed. New York, NY: Elsevier/Saunders; 2011.
- Litvan I, Mangone CA, McKee A, et al. Natural history of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome) and clinical predictors of survival: a clinicopathological study. J Neurol Neurosurg Psychiatry 1996; 60:615–620.
- Boeve BF, Silber MH, Parisi JE, et al. Synucleinopathy pathology and REM sleep behavior disorder plus dementia or parkinsonism. Neurology 2003; 61:40–45.
- Pharr V, Uttl B, Stark M, Litvan I, Fantie B, Grafman J. Comparison of apraxia in corticobasal degeneration and progressive supranuclear palsy. Neurology 2001; 56:957–963.
- Williams DR, Lees AJ. What features improve the accuracy of the clinical diagnosis of progressive supranuclear palsy-parkinsonism (PSP-P)? Mov Disord 2010; 25:357–362.
- Wenning GK, Litvan I, Tolosa E. Milestones in atypical and secondary parkinsonisms. Mov Disord 2011; 26:1083–1095.
- Gallucci M, Limbucci N, Catalucci A, Caulo M. Neurodegenerative diseases. Radiol Clin North Am 2008; 46:799–817.
- Stamelou M, de Silva R, Arias-Carrión O, et al. Rational therapeutic approaches to progressive supranuclear palsy. Brain 2010; 133:1578–1590.
- Gold M, Lorenzl S, Stewart AJ, Morimoto BH, Williams DR, Gozes I. Critical appraisal of the role of davunetide in the treatment of progressive supranuclear palsy. Neuropsychiatr Dis Treat 2012; 8:85–93.
- Boeve BF, Lang AE, Litvan I. Corticobasal degeneration and its relationship to progressive supranuclear palsy and frontotemporal dementia. Ann Neurol 2003; 54(suppl 5):S15–S19.
- Armstrong MJ, Litvan I, Lang AE, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013; 80:496–503.
- Kompoliti K, Goetz CG, Boeve BF, et al. Clinical presentation and pharmacological therapy in corticobasal degeneration. Arch Neurol 1998; 55:957–961.
- Litvan I, Cummings JL, Mega M. Neuropsychiatric features of corticobasal degeneration. J Neurol Neurosurg Psychiatry 1998; 65:717–721.
- Pillon B, Blin J, Vidailhet M, et al. The neuropsychological pattern of corticobasal degeneration: comparison with progressive supranuclear palsy and Alzheimer’s disease. Neurology 1995; 45:1477–1483.
- Tang-Wai DF, Josephs KA, Boeve BF, Petersen RC, Parisi JE, Dickson DW. Coexistent Lewy body disease in a case of “visual variant of Alzheimer’s disease.” J Neurol Neurosurg Psychiatry 2003; 74:389.
- Tang-Wai DF, Josephs KA, Boeve BF, Dickson DW, Parisi JE, Petersen RC. Pathologically confirmed corticobasal degeneration presenting with visuospatial dysfunction. Neurology 2003; 61:1134–1135.
- Goto K, Ueki A, Shimode H, Shinjo H, Miwa C, Morita Y. Depression in multiple system atrophy: a case report. Psychiatry Clin Neurosci 2000; 54:507–511.
- Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008; 71:670–676.
- Berent S, Giordani B, Gilman S, et al. Patterns of neuropsychological performance in multiple system atrophy compared to sporadic and hereditary olivopontocerebellar atrophy. Brain Cogn 2002; 50:194–206.
- Ghorayeb I, Yekhlef F, Chrysostome V, Balestre E, Bioulac B, Tison F. Sleep disorders and their determinants in multiple system atrophy. J Neurol Neurosurg Psychiatry 2002; 72:798–800.
- Schrag A, Kingsley D, Phatouros C, et al. Clinical usefulness of magnetic resonance imaging in multiple system atrophy. J Neurol Neurosurg Psychiatry 1998; 65:65–71.
- Massey LA, Micallef C, Paviour DC, et al. Conventional magnetic resonance imaging in confirmed progressive supranuclear palsy and multiple system atrophy. Mov Disord 2012; 27:1754–1762.
- Low PA, Gilden JL, Freeman R, Sheng KN, McElligott MA. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA 1997; 277:1046–1051.
- Mathias CJ, Kimber JR. Postural hypotension: causes, clinical features, investigation, and management. Annu Rev Med 1999; 50:317–336.
- Emre M, Aarsland D, Brown R, et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 2007; 22:1689–1707.
- Burton EJ, McKeith IG, Burn DJ, Williams ED, O’Brien JT. Cerebral atrophy in Parkinson’s disease with and without dementia: a comparison with Alzheimer’s disease, dementia with Lewy bodies and controls. Brain 2004; 127:791–800.
- Piert M, Koeppe RA, Giordani B, Minoshima S, Kuhl DE. Determination of regional rate constants from dynamic FDG-PET studies in Parkinson’s disease. J Nucl Med 1996; 37:1115–1122.
- Ratnavalli E, Brayne C, Dawson K, Hodges JR. The prevalence of frontotemporal dementia. Neurology 2002; 58:1615–1621.
- Baborie A, Griffiths TD, Jaros E, et al. Frontotemporal dementia in elderly individuals. Arch Neurol 2012; 69:1052–1960.
- Neary D, Snowden JS, Gustafson L, et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 1998; 51:1546–1554.
- Kertesz A, Nadkarni N, Davidson W, Thomas AW. The Frontal Behavioral Inventory in the differential diagnosis of frontotemporal dementia. J Int Neuropsychol Soc 2000; 6:460–468.
- Mendez MF, Lauterbach EC, Sampson SM; ANPA Committee on Research. An evidence-based review of the psychopathology of frontotemporal dementia: a report of the ANPA Committee on Research. J Neuropsychiatry Clin Neurosci 2008; 20:130–149.
- Kertesz A, McMonagle P, Blair M, Davidson W, Munoz DG. The evolution and pathology of frontotemporal dementia. Brain 2005; 128:1996–2005.
- Mesulam MM. Primary progressive aphasia—a language-based dementia. N Engl J Med 2003; 349:1535–1542.
- Turner RS, Kenyon LC, Trojanowski JQ, Gonatas N, Grossman M. Clinical, neuroimaging, and pathologic features of progressive nonfluent aphasia. Ann Neurol 1996; 39:166–173.
- Abe K, Ukita H, Yanagihara T. Imaging in primary progressive aphasia. Neuroradiology 1997; 39:556–559.
- Trenkwalder C, Schwarz J, Gebhard J, et al. Starnberg trial on epidemiology of parkinsonism and hypertension in the elderly. Prevalence of Parkinson’s disease and related disorders assessed by a door-to-door survey of inhabitants older than 65 years. Arch Neurol 1995; 52:1017–1022.
- Hakim S, Adams RD. The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965; 2:307–327.
- Vanneste JA. Diagnosis and management of normal-pressure hydrocephalus. J Neurol 2000; 247:5–14.
- Ishii K, Kanda T, Harada A, et al. Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol 2008; 18:2678–2683.
- Marmarou A, Bergsneider M, Klinge P, Relkin N, Black PM. The value of supplemental prognostic tests for the preoperative assessment of idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57(suppl 3):S17–S28.
- Bergsneider M, Miller C, Vespa PM, Hu X. Surgical management of adult hydrocephalus. Neurosurgery 2008; 62(suppl 2):643–659.
- Malm J, Graff-Radford NR, Ishikawa M, et al. Influence of comorbidities in idiopathic normal pressure hydrocephalus—research and clinical care. A report of the ISHCSF task force on comorbidities in INPH. Fluids Barriers CNS 2013; 10:22.
- Rabinovici GD, Wang PN, Levin J, et al. First symptom in sporadic Creutzfeldt-Jakob disease. Neurology 2006; 66:286–287.
- Zerr I, Kallenberg K, Summers DM, et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain 2009; 132:2659–2668.
- Dalmau J, Rosenfeld MR. Paraneoplastic syndromes of the CNS. Lancet Neurol 2008; 7:327–340.
- Chong JY, Rowland LP, Utiger RD. Hashimoto encephalopathy: syndrome or myth? Arch Neurol 2003; 60:164–171.
- Plassman BL, Langa KM, Fisher GG, et al. Prevalence of dementia in the United States: the aging, demographics, and memory study. Neuroepidemiology 2007; 29:125–132.
- Alzheimer’s Association. 2013 Alzheimer’s disease facts and figures. http://www.alz.org/downloads/facts_figures_2013.pdf. Accessed February 3, 2014.
- Hebert LE, Weuve J, Scherr PA, Evans DA. Alzheimer disease in the United States (2010-2050) estimated using the 2010 census. Neurology 2013; 80:1778–1783.
- Hou CE, Carlin D, Miller BL. Non-Alzheimer’s disease dementias: anatomic, clinical, and molecular correlates. Can J Psychiatry 2004; 49:164–171.
- Hachinski VC, Iliff LD, Zilhka E, et al. Cerebral blood flow in dementia. Arch Neurol 1975; 32:632–637.
- Fereshtehnejad SM, Religa D, Westman E, Aarsland D, Lökk J, Eriksdotter M. Demography, diagnostics, and medication in dementia with Lewy bodies and Parkinson’s disease with dementia: data from the Swedish Dementia Quality Registry (SveDem). Neuropsychiatr Dis Treat 2013; 9:927–935.
- Nomura T, Inoue Y, Takigawa H, Nakashima K. Comparison of REM sleep behaviour disorder variables between patients with progressive supranuclear palsy and those with Parkinson’s disease. Parkinsonism Relat Disord 2012; 18:394–396.
- Davis PH, Golbe LI, Duvoisin RC, Schoenberg BS. Risk factors for progrssive supranuclear palsy. Neurology 1988; 38:1546–1552.
- Tousi B, Schuele SU, Subramanian T. A 46-year-old woman with rigidity and frequent falls. Cleve Clin J Med 2005; 72:57–63.
- Cooper AD, Josephs KA. Photophobia, visual hallucinations, and REM sleep behavior disorder in progressive supranuclear palsy and corticobasal degeneration: a prospective study. Parkinsonism Relat Disord 2009; 15:59–61.
- Lyketsos CG, Lopez O, Jones B, Fitzpatrick AL, Breitner J, DeKosky S. Prevalence of neuropsychiatric symptoms in dementia and mild cognitive impairment: results from the cardiovascular health study0. JAMA 2002; 288:1475–1483.
- Knopman DS. Dementia and cerebrovascular disease. Mayo Clin Proc 2006; 81:223–230.
- Erkinjuntti T, Kurz A, Gauthier S, Bullock R, Lilienfeld S, Damaraju CV. Efficacy of galantamine in probable vascular dementia and Alzheimer’s disease combined with cerebrovascular disease: a randomised trial. Lancet 2002; 359:1283–1290.
- Sink KM, Holden KF, Yaffe K. Pharmacological treatment of neuropsychiatric symptoms of dementia: a review of the evidence. JAMA 2005; 293:596–608.
- Schneider LS, Dagerman K, Insel PS. Efficacy and adverse effects of atypical antipsychotics for dementia: meta-analysis of randomized, placebo-controlled trials. Am J Geriatr Psychiatry 2006; 14:191–210.
- McKeith IG, Dickson DW, Lowe J, et al; Consortium on DLB. Diagnosis and management of dementia with Lewy bodies: third report of the DLB Consortium. Neurology 2005; 65:1863–1872.
- Tartaglia MC, Rosen HJ, Miller BL. Neuroimaging in dementia. Neurotherapeutics 2011; 8:82–92.
- McKeith I, Del Ser T, Spano P, et al. Efficacy of rivastigmine in dementia with Lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet 2000; 356:2031–2036.
- Rolinski M, Fox C, Maidment I, McShane R. Cholinesterase inhibitors for dementia with Lewy bodies, Parkinson’s disease dementia and cognitive impairment in Parkinson’s disease. Cochrane Database Syst Rev 2012; 3:CD006504.
- Emre M, Tsolaki M, Bonucelli U, et al; on behalf of the 11018 Study Investigators. Memantine for patients with Parkinson’s disease dementia or dementia with Lewy bodies: a randomised, double-blind, placebo-controlled trial. Lancet Neurol 2010; 9:969–977.
- Kurlan R, Cummings J, Raman R, Thal L; Alzheimer’s Disease Cooperative Study Group. Quetiapine for agitation or psychosis in patients with dementia and parkinsonism. Neurology 2007; 68:1356–1363.
- Ballard C, Aarsland D, Francis P, Corbett A. Neuropsychiatric symptoms in patients with dementias associated with cortical Lewy bodies: pathophysiology, clinical features, and pharmacological management. Drugs Aging 2013; 30:603–611.
- Goetz CG, Leurgans S, Lang AE, Litvan I. Progression of gait, speech and swallowing deficits in progressive supranuclear palsy. Neurology 2003; 60:917–922.
- Kasashima S, Oda Y. Cholinergic neuronal loss in the basal forebrain and mesopontine tegmentum of progressive supranuclear palsy and corticobasal degeneration. Acta Neuropathol 2003; 105:117–124.
- Fahn S, Jankovic J, Hallett M, editors. Principles and Practice of Movement Disorders. 2nd ed. New York, NY: Elsevier/Saunders; 2011.
- Litvan I, Mangone CA, McKee A, et al. Natural history of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome) and clinical predictors of survival: a clinicopathological study. J Neurol Neurosurg Psychiatry 1996; 60:615–620.
- Boeve BF, Silber MH, Parisi JE, et al. Synucleinopathy pathology and REM sleep behavior disorder plus dementia or parkinsonism. Neurology 2003; 61:40–45.
- Pharr V, Uttl B, Stark M, Litvan I, Fantie B, Grafman J. Comparison of apraxia in corticobasal degeneration and progressive supranuclear palsy. Neurology 2001; 56:957–963.
- Williams DR, Lees AJ. What features improve the accuracy of the clinical diagnosis of progressive supranuclear palsy-parkinsonism (PSP-P)? Mov Disord 2010; 25:357–362.
- Wenning GK, Litvan I, Tolosa E. Milestones in atypical and secondary parkinsonisms. Mov Disord 2011; 26:1083–1095.
- Gallucci M, Limbucci N, Catalucci A, Caulo M. Neurodegenerative diseases. Radiol Clin North Am 2008; 46:799–817.
- Stamelou M, de Silva R, Arias-Carrión O, et al. Rational therapeutic approaches to progressive supranuclear palsy. Brain 2010; 133:1578–1590.
- Gold M, Lorenzl S, Stewart AJ, Morimoto BH, Williams DR, Gozes I. Critical appraisal of the role of davunetide in the treatment of progressive supranuclear palsy. Neuropsychiatr Dis Treat 2012; 8:85–93.
- Boeve BF, Lang AE, Litvan I. Corticobasal degeneration and its relationship to progressive supranuclear palsy and frontotemporal dementia. Ann Neurol 2003; 54(suppl 5):S15–S19.
- Armstrong MJ, Litvan I, Lang AE, et al. Criteria for the diagnosis of corticobasal degeneration. Neurology 2013; 80:496–503.
- Kompoliti K, Goetz CG, Boeve BF, et al. Clinical presentation and pharmacological therapy in corticobasal degeneration. Arch Neurol 1998; 55:957–961.
- Litvan I, Cummings JL, Mega M. Neuropsychiatric features of corticobasal degeneration. J Neurol Neurosurg Psychiatry 1998; 65:717–721.
- Pillon B, Blin J, Vidailhet M, et al. The neuropsychological pattern of corticobasal degeneration: comparison with progressive supranuclear palsy and Alzheimer’s disease. Neurology 1995; 45:1477–1483.
- Tang-Wai DF, Josephs KA, Boeve BF, Petersen RC, Parisi JE, Dickson DW. Coexistent Lewy body disease in a case of “visual variant of Alzheimer’s disease.” J Neurol Neurosurg Psychiatry 2003; 74:389.
- Tang-Wai DF, Josephs KA, Boeve BF, Dickson DW, Parisi JE, Petersen RC. Pathologically confirmed corticobasal degeneration presenting with visuospatial dysfunction. Neurology 2003; 61:1134–1135.
- Goto K, Ueki A, Shimode H, Shinjo H, Miwa C, Morita Y. Depression in multiple system atrophy: a case report. Psychiatry Clin Neurosci 2000; 54:507–511.
- Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology 2008; 71:670–676.
- Berent S, Giordani B, Gilman S, et al. Patterns of neuropsychological performance in multiple system atrophy compared to sporadic and hereditary olivopontocerebellar atrophy. Brain Cogn 2002; 50:194–206.
- Ghorayeb I, Yekhlef F, Chrysostome V, Balestre E, Bioulac B, Tison F. Sleep disorders and their determinants in multiple system atrophy. J Neurol Neurosurg Psychiatry 2002; 72:798–800.
- Schrag A, Kingsley D, Phatouros C, et al. Clinical usefulness of magnetic resonance imaging in multiple system atrophy. J Neurol Neurosurg Psychiatry 1998; 65:65–71.
- Massey LA, Micallef C, Paviour DC, et al. Conventional magnetic resonance imaging in confirmed progressive supranuclear palsy and multiple system atrophy. Mov Disord 2012; 27:1754–1762.
- Low PA, Gilden JL, Freeman R, Sheng KN, McElligott MA. Efficacy of midodrine vs placebo in neurogenic orthostatic hypotension. A randomized, double-blind multicenter study. Midodrine Study Group. JAMA 1997; 277:1046–1051.
- Mathias CJ, Kimber JR. Postural hypotension: causes, clinical features, investigation, and management. Annu Rev Med 1999; 50:317–336.
- Emre M, Aarsland D, Brown R, et al. Clinical diagnostic criteria for dementia associated with Parkinson’s disease. Mov Disord 2007; 22:1689–1707.
- Burton EJ, McKeith IG, Burn DJ, Williams ED, O’Brien JT. Cerebral atrophy in Parkinson’s disease with and without dementia: a comparison with Alzheimer’s disease, dementia with Lewy bodies and controls. Brain 2004; 127:791–800.
- Piert M, Koeppe RA, Giordani B, Minoshima S, Kuhl DE. Determination of regional rate constants from dynamic FDG-PET studies in Parkinson’s disease. J Nucl Med 1996; 37:1115–1122.
- Ratnavalli E, Brayne C, Dawson K, Hodges JR. The prevalence of frontotemporal dementia. Neurology 2002; 58:1615–1621.
- Baborie A, Griffiths TD, Jaros E, et al. Frontotemporal dementia in elderly individuals. Arch Neurol 2012; 69:1052–1960.
- Neary D, Snowden JS, Gustafson L, et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 1998; 51:1546–1554.
- Kertesz A, Nadkarni N, Davidson W, Thomas AW. The Frontal Behavioral Inventory in the differential diagnosis of frontotemporal dementia. J Int Neuropsychol Soc 2000; 6:460–468.
- Mendez MF, Lauterbach EC, Sampson SM; ANPA Committee on Research. An evidence-based review of the psychopathology of frontotemporal dementia: a report of the ANPA Committee on Research. J Neuropsychiatry Clin Neurosci 2008; 20:130–149.
- Kertesz A, McMonagle P, Blair M, Davidson W, Munoz DG. The evolution and pathology of frontotemporal dementia. Brain 2005; 128:1996–2005.
- Mesulam MM. Primary progressive aphasia—a language-based dementia. N Engl J Med 2003; 349:1535–1542.
- Turner RS, Kenyon LC, Trojanowski JQ, Gonatas N, Grossman M. Clinical, neuroimaging, and pathologic features of progressive nonfluent aphasia. Ann Neurol 1996; 39:166–173.
- Abe K, Ukita H, Yanagihara T. Imaging in primary progressive aphasia. Neuroradiology 1997; 39:556–559.
- Trenkwalder C, Schwarz J, Gebhard J, et al. Starnberg trial on epidemiology of parkinsonism and hypertension in the elderly. Prevalence of Parkinson’s disease and related disorders assessed by a door-to-door survey of inhabitants older than 65 years. Arch Neurol 1995; 52:1017–1022.
- Hakim S, Adams RD. The special clinical problem of symptomatic hydrocephalus with normal cerebrospinal fluid pressure. Observations on cerebrospinal fluid hydrodynamics. J Neurol Sci 1965; 2:307–327.
- Vanneste JA. Diagnosis and management of normal-pressure hydrocephalus. J Neurol 2000; 247:5–14.
- Ishii K, Kanda T, Harada A, et al. Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol 2008; 18:2678–2683.
- Marmarou A, Bergsneider M, Klinge P, Relkin N, Black PM. The value of supplemental prognostic tests for the preoperative assessment of idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57(suppl 3):S17–S28.
- Bergsneider M, Miller C, Vespa PM, Hu X. Surgical management of adult hydrocephalus. Neurosurgery 2008; 62(suppl 2):643–659.
- Malm J, Graff-Radford NR, Ishikawa M, et al. Influence of comorbidities in idiopathic normal pressure hydrocephalus—research and clinical care. A report of the ISHCSF task force on comorbidities in INPH. Fluids Barriers CNS 2013; 10:22.
- Rabinovici GD, Wang PN, Levin J, et al. First symptom in sporadic Creutzfeldt-Jakob disease. Neurology 2006; 66:286–287.
- Zerr I, Kallenberg K, Summers DM, et al. Updated clinical diagnostic criteria for sporadic Creutzfeldt-Jakob disease. Brain 2009; 132:2659–2668.
- Dalmau J, Rosenfeld MR. Paraneoplastic syndromes of the CNS. Lancet Neurol 2008; 7:327–340.
- Chong JY, Rowland LP, Utiger RD. Hashimoto encephalopathy: syndrome or myth? Arch Neurol 2003; 60:164–171.
KEY POINTS
- Vascular dementia presents as a sudden, stepwise progression of cognitive deficits.
- Lewy body dementia often involves prominent visual hallucinations.
- Progressive supranuclear palsy starts with gait and balance problems caused by downward-gaze palsy.
- Many neurodegenerative conditions involve parkinsonism, but unlike Parkinson disease, they do not tend to respond well to levodopa, and dementia develops early.
- Corticobasal degeneration involves markedly asymmetric parkinsonism.
- Frontotemporal dementia involves dramatic behavior changes, including inappropriate impulsivity and complete apathy.
- Patients with rapidly progressive dementia should be evaluated for a treatable condition such as antibody-mediated encephalitis.
Nanotherapies make inroads in wound regeneration
DENVER – Move over, gauze, bandages, and moist wound-healing techniques. Nanomaterials are making significant inroads in wound regeneration.
Dr. Adam Friedman, director of dermatologic research at the Montefiore-Einstein College of Medicine, New York, highlighted four nanotherapies in the field of wound care.
• Antimicrobial nano-based dressings. The antibacterial properties of silver have been documented since 1000 B.C., said Dr. Friedman of the departments of dermatology and of physiology and biophysics at the medical school. Silver ions are believed to directly disrupt pathogen cell walls/membranes and suppress respiratory enzymes and electron transport components. Silver has been used commercially for decades, with demonstrated antimicrobial effects (Crede’s 1% silver nitrate eyedrops were used to prevent mother to child transmission of gonococcal eye infection); anti-inflammatory properties (silver nitrate is used in pleurodesis); and infection protection (silver nanoparticle–impregnated wound dressings prevent infection and enhance wound healing).
Silver nanoparticles have an "increased likelihood of directly interacting with the target bacteria or virus," Dr. Friedman said in an interview in advance of the annual meeting of the American Academy of Dermatology. "They bind to and/or disturb bacterial cell membrane activity as well as release silver ions much more readily then their bulk counterparts." Silver dressings currently on the market include Silvercel, Aquacel, and Acticoat.
Similarly to nanometals, the biological activity of curcumin (a water-insoluble polyphenolic compound derived from tumeric) can be used in the nanoform and has been shown to both effectively clear methicillin-resistant Staphylococcus aureus in burn wound infections and accelerate the healing of thermal burn wounds.
• Immunomodulating antimicrobial nanoparticles. One of the most promising immunomodulators in wound healing is the gaseous molecule nitric oxide (NO). This potential has yet to be realized, Dr. Friedman said, as NO is highly unstable and its site of action is often microns from its source of generation. Nanotechnology can allow for controlled and sustained release of this evasive biomolecule and make therapeutic translation a reality. "At Einstein, we are utilizing NO-generating nanoparticles to accelerate wound healing and eradicate multidrug resistant pathogens," he said. "But, because NO is integral to so many biological processes, it can do so much more. This technology is also being studied for the treatment of cardiovascular disease and even the topical treatment of erectile dysfunction."
• Gene modifying/silencing technologies. RNA interference is an endogenous mechanism to control gene expression in a variety of organisms. "We can take advantage of this process using siRNA (small interfering RNA) to manipulate limitless biological processes," Dr. Friedman explained. "While a hot area, translation to the bedside has been difficult as siRNA are very unstable and have a difficult time reaching their targets inside cells. Nanoparticles have been shown to overcome these limitations." Dr. Friedman noted how siRNA encapsulate nanoparticles targeting fidgetin-like 2 (an ATPase that cleaves microtubules), can knock down this gene in vivo, resulting in accelerated epithelial cell migration and hastened wound closure in both excisional and burn wound mouse models.
• Growth factor–releasing nanoparticles. Growth factors have been found to speed the healing of acute and chronic wounds in humans, including Regranex, a analogue of platelet-derived growth factor that is FDA-approved for treating leg and foot ulcers in diabetic patients, epidermal growth factor (donor-site wounds), fibroblast growth factor (burn wounds), and growth hormone (donor sites in burned children). "Nanomaterials offer many advantages here, from allowing for temporal release depending on the wound environment to delivering multiple factors at the same or different times, to even serving as a structural foundation for wound healing while releasing factors simultaneously," Dr. Friedman said. "The possibilities are limitless."
Dr. Friedman characterized nanomedicine as "a newborn branch of science. These promising and innovative tools should help us overcome limitations of traditional wound care, improve direct intervention on phases of wound healing, and provide better solutions for wound dressing that induce favorable wound healing environments. More work – and funding – is needed."
Dr. Friedman disclosed that he is a consultant and/or a member of the scientific advisory board for SanovaWorks, Prodigy, Oakstone Institute, Liquidia Technologies, L’Oréal, Amgen, Onset, Aveeno, GSK, MicroCures, and Nano Bio-Med. He is also a speaker for Onset and Amgen.
This article was updated March 24, 2014.
DENVER – Move over, gauze, bandages, and moist wound-healing techniques. Nanomaterials are making significant inroads in wound regeneration.
Dr. Adam Friedman, director of dermatologic research at the Montefiore-Einstein College of Medicine, New York, highlighted four nanotherapies in the field of wound care.
• Antimicrobial nano-based dressings. The antibacterial properties of silver have been documented since 1000 B.C., said Dr. Friedman of the departments of dermatology and of physiology and biophysics at the medical school. Silver ions are believed to directly disrupt pathogen cell walls/membranes and suppress respiratory enzymes and electron transport components. Silver has been used commercially for decades, with demonstrated antimicrobial effects (Crede’s 1% silver nitrate eyedrops were used to prevent mother to child transmission of gonococcal eye infection); anti-inflammatory properties (silver nitrate is used in pleurodesis); and infection protection (silver nanoparticle–impregnated wound dressings prevent infection and enhance wound healing).
Silver nanoparticles have an "increased likelihood of directly interacting with the target bacteria or virus," Dr. Friedman said in an interview in advance of the annual meeting of the American Academy of Dermatology. "They bind to and/or disturb bacterial cell membrane activity as well as release silver ions much more readily then their bulk counterparts." Silver dressings currently on the market include Silvercel, Aquacel, and Acticoat.
Similarly to nanometals, the biological activity of curcumin (a water-insoluble polyphenolic compound derived from tumeric) can be used in the nanoform and has been shown to both effectively clear methicillin-resistant Staphylococcus aureus in burn wound infections and accelerate the healing of thermal burn wounds.
• Immunomodulating antimicrobial nanoparticles. One of the most promising immunomodulators in wound healing is the gaseous molecule nitric oxide (NO). This potential has yet to be realized, Dr. Friedman said, as NO is highly unstable and its site of action is often microns from its source of generation. Nanotechnology can allow for controlled and sustained release of this evasive biomolecule and make therapeutic translation a reality. "At Einstein, we are utilizing NO-generating nanoparticles to accelerate wound healing and eradicate multidrug resistant pathogens," he said. "But, because NO is integral to so many biological processes, it can do so much more. This technology is also being studied for the treatment of cardiovascular disease and even the topical treatment of erectile dysfunction."
• Gene modifying/silencing technologies. RNA interference is an endogenous mechanism to control gene expression in a variety of organisms. "We can take advantage of this process using siRNA (small interfering RNA) to manipulate limitless biological processes," Dr. Friedman explained. "While a hot area, translation to the bedside has been difficult as siRNA are very unstable and have a difficult time reaching their targets inside cells. Nanoparticles have been shown to overcome these limitations." Dr. Friedman noted how siRNA encapsulate nanoparticles targeting fidgetin-like 2 (an ATPase that cleaves microtubules), can knock down this gene in vivo, resulting in accelerated epithelial cell migration and hastened wound closure in both excisional and burn wound mouse models.
• Growth factor–releasing nanoparticles. Growth factors have been found to speed the healing of acute and chronic wounds in humans, including Regranex, a analogue of platelet-derived growth factor that is FDA-approved for treating leg and foot ulcers in diabetic patients, epidermal growth factor (donor-site wounds), fibroblast growth factor (burn wounds), and growth hormone (donor sites in burned children). "Nanomaterials offer many advantages here, from allowing for temporal release depending on the wound environment to delivering multiple factors at the same or different times, to even serving as a structural foundation for wound healing while releasing factors simultaneously," Dr. Friedman said. "The possibilities are limitless."
Dr. Friedman characterized nanomedicine as "a newborn branch of science. These promising and innovative tools should help us overcome limitations of traditional wound care, improve direct intervention on phases of wound healing, and provide better solutions for wound dressing that induce favorable wound healing environments. More work – and funding – is needed."
Dr. Friedman disclosed that he is a consultant and/or a member of the scientific advisory board for SanovaWorks, Prodigy, Oakstone Institute, Liquidia Technologies, L’Oréal, Amgen, Onset, Aveeno, GSK, MicroCures, and Nano Bio-Med. He is also a speaker for Onset and Amgen.
This article was updated March 24, 2014.
DENVER – Move over, gauze, bandages, and moist wound-healing techniques. Nanomaterials are making significant inroads in wound regeneration.
Dr. Adam Friedman, director of dermatologic research at the Montefiore-Einstein College of Medicine, New York, highlighted four nanotherapies in the field of wound care.
• Antimicrobial nano-based dressings. The antibacterial properties of silver have been documented since 1000 B.C., said Dr. Friedman of the departments of dermatology and of physiology and biophysics at the medical school. Silver ions are believed to directly disrupt pathogen cell walls/membranes and suppress respiratory enzymes and electron transport components. Silver has been used commercially for decades, with demonstrated antimicrobial effects (Crede’s 1% silver nitrate eyedrops were used to prevent mother to child transmission of gonococcal eye infection); anti-inflammatory properties (silver nitrate is used in pleurodesis); and infection protection (silver nanoparticle–impregnated wound dressings prevent infection and enhance wound healing).
Silver nanoparticles have an "increased likelihood of directly interacting with the target bacteria or virus," Dr. Friedman said in an interview in advance of the annual meeting of the American Academy of Dermatology. "They bind to and/or disturb bacterial cell membrane activity as well as release silver ions much more readily then their bulk counterparts." Silver dressings currently on the market include Silvercel, Aquacel, and Acticoat.
Similarly to nanometals, the biological activity of curcumin (a water-insoluble polyphenolic compound derived from tumeric) can be used in the nanoform and has been shown to both effectively clear methicillin-resistant Staphylococcus aureus in burn wound infections and accelerate the healing of thermal burn wounds.
• Immunomodulating antimicrobial nanoparticles. One of the most promising immunomodulators in wound healing is the gaseous molecule nitric oxide (NO). This potential has yet to be realized, Dr. Friedman said, as NO is highly unstable and its site of action is often microns from its source of generation. Nanotechnology can allow for controlled and sustained release of this evasive biomolecule and make therapeutic translation a reality. "At Einstein, we are utilizing NO-generating nanoparticles to accelerate wound healing and eradicate multidrug resistant pathogens," he said. "But, because NO is integral to so many biological processes, it can do so much more. This technology is also being studied for the treatment of cardiovascular disease and even the topical treatment of erectile dysfunction."
• Gene modifying/silencing technologies. RNA interference is an endogenous mechanism to control gene expression in a variety of organisms. "We can take advantage of this process using siRNA (small interfering RNA) to manipulate limitless biological processes," Dr. Friedman explained. "While a hot area, translation to the bedside has been difficult as siRNA are very unstable and have a difficult time reaching their targets inside cells. Nanoparticles have been shown to overcome these limitations." Dr. Friedman noted how siRNA encapsulate nanoparticles targeting fidgetin-like 2 (an ATPase that cleaves microtubules), can knock down this gene in vivo, resulting in accelerated epithelial cell migration and hastened wound closure in both excisional and burn wound mouse models.
• Growth factor–releasing nanoparticles. Growth factors have been found to speed the healing of acute and chronic wounds in humans, including Regranex, a analogue of platelet-derived growth factor that is FDA-approved for treating leg and foot ulcers in diabetic patients, epidermal growth factor (donor-site wounds), fibroblast growth factor (burn wounds), and growth hormone (donor sites in burned children). "Nanomaterials offer many advantages here, from allowing for temporal release depending on the wound environment to delivering multiple factors at the same or different times, to even serving as a structural foundation for wound healing while releasing factors simultaneously," Dr. Friedman said. "The possibilities are limitless."
Dr. Friedman characterized nanomedicine as "a newborn branch of science. These promising and innovative tools should help us overcome limitations of traditional wound care, improve direct intervention on phases of wound healing, and provide better solutions for wound dressing that induce favorable wound healing environments. More work – and funding – is needed."
Dr. Friedman disclosed that he is a consultant and/or a member of the scientific advisory board for SanovaWorks, Prodigy, Oakstone Institute, Liquidia Technologies, L’Oréal, Amgen, Onset, Aveeno, GSK, MicroCures, and Nano Bio-Med. He is also a speaker for Onset and Amgen.
This article was updated March 24, 2014.
AT THE AAD ANNUAL MEETING
DNR orders and medical futility
Question: A 71-year-old woman with heart disease and breast cancer was hospitalized for uncontrolled diabetes and a hip fracture. There, she suffered two grand mal seizures that could not be controlled with anticonvulsants, and the patient lapsed into coma. Her daughter became the surrogate decision maker, and she made it clear that her mother always said she wanted everything done.
After several weeks, the physicians decided that further care would be futile. The chair of the ethics committee took the view that the family’s opinion was not relevant, because cardiopulmonary resuscitation (CPR) was not a genuine therapeutic option and would be "medically contraindicated, inhumane, and unethical." Accordingly, the attending physician entered a do-not-resuscitate (DNR) order despite strong protest from the daughter. The patient died shortly thereafter without receiving CPR.
In this actual case where the daughter filed a negligence lawsuit against the hospital, which of the following statements is incorrect?
A. The defendant’s expert relied upon the position paper of the American Thoracic Society, which states that life support "can be limited without the consent of patient or surrogate when the intervention is judged to be futile."
B. Futile intervention may be defined as treatment that would be highly unlikely to result in a meaningful survival of the patient.
C. The jury found that if competent, the patient would have wanted CPR and would have wanted ventilation until death.
D. The jury found such treatment would be futile.
E. The jury entered a verdict of negligence.
Answer: E. The above narrative is based on Gilgunn v. Massachusetts General Hospital (verdict issued April 21, 1995) and adapted from an article by the prominent ethicist Alexander M. Capron.1 All of the options listed were evident at trial, except that it was a defense verdict, i.e., no negligence. The case remains the best-known litigated example of medical futility. In earlier cases such as Wanglie2 and Baby K,3 the courts had avoided addressing the issue directly.
Gilgunn supports the notion that futility of CPR can trump a patient’s family insistence on having such intervention. But being a trial court verdict, it lacks the precedential authority that an appellate decision would confer.
The verdict also was not without its critics. As Mr. Capron wrote:
"But to allow Mrs. Gilgunn’s physicians to impose this view, however widely held, on their patient is the equivalent of allowing them to abandon the patient. We still need means ... to reach a social consensus on whether health professionals should have authority to decide, among the interventions patients (or surrogates) will accept, which will actually be provided and which they may withhold based upon their evaluation of the worth of the outcome. When we come to adopt such policies, we would do well to ponder long and hard before adopting a utilitarian measure that affords the waning lives of the most vulnerable in our society less protection from unilateral decisions by powerful professionals."1
In 1999, the District of Columbia Court of Appeals agreed with a trial court’s order to issue a DNR order for a neglected 2-year-old child who was born prematurely with serious medical problems and virtually nonexistent cognition, but who could still experience pain.4 The biological mother and putative father appealed, but the Court of Appeals affirmed. The court held, among other things, that the standard of proof required for issuance of a DNR order is clear and convincing evidence, and the applicable standard is the best interests of the child test rather than a substituted judgment standard. However, this case pitted the biological parents against a court-appointed guardian, rather than the medical providers.
The American Medical Association’s current Code of Ethics urges that when neither a patient nor surrogate is able or available to make a decision regarding CPR, an attending physician contemplating a DNR order should consult another physician or a hospital ethics committee if one is available.5 If the physician determines that a request for resuscitation would not be medically effective, "the physician should seek to resolve the conflict through a fair decision-making process, when time permits." In an earlier version,6 the AMA stated: "CPR may be withheld if, in the judgment of the treating physician, an attempt to resuscitate the patient would be futile."
DNR orders are at the heart of the futility conundrum, especially because CPR is a highly invasive, low-success procedure (notable exceptions exist, however). Medical futility denotes treatment that cannot confer an overall benefit on the whole person even if it can restore some physiologic variable.7 The Latin word "futilis" means leaky, and in Greek mythology, the daughters of Danaus were condemned in the underworld to draw water in leaky sieves, conveying the full meaning of futility.
The debate is over whether an intervention, however hopeless and ineffective, can ever be characterized as completely futile. Moreover, without absolute certainty, will this suffice to override a patient’s insistence on having that treatment?
Others have countered that absoluteness is an unrealistic standard, and that inhumane suffering and costs are relevant factors to ponder. The AMA has clearly stated that "physicians are not ethically obligated to deliver care that, in their best professional judgment, will not have a reasonable chance of benefiting their patients," and "patients should not be given treatments simply because they demand them."8
One legal consequence of discontinuing medical treatment that ends with a patient’s demise is the specter of criminal prosecution, although charges of homicide are unlikely to prevail.
In a landmark California case, Barber v. Superior Court of Los Angeles County,9 the court recognized that "a physician is authorized under the standards of medical practice to discontinue a form of therapy which in his medical judgment is useless. ... If the treating physicians have determined that continued use of a respirator is useless, then they may decide to discontinue it without fear of civil or criminal liability."
In Kansas v. Naramore,10 the state appeals court reversed and entered a verdict of acquittal despite a jury finding Dr. Naramore guilty over his provision of palliative treatment. The court noted that the burden of proof to establish the criminal guilt of a physician for acts arising out of providing medical treatment is higher than that necessary to find medical malpractice or to impose medical licensure discipline. It held that "with no direct evidence of criminal intent, it is highly disturbing that testimony by such an impressive array of apparently objective medical experts, who found the defendant’s actions to be not only noncriminal, but medically appropriate, can be dismissed as ‘unbelievable’ and not even capable of generating reasonable doubt."
Notwithstanding widely accepted ethical notions of medical futility, there are state and federal statutes touching on DNR orders that warrant careful attention.
For example, New York Public Health Law Section 2962, paragraph 1, states: "Every person admitted to a hospital shall be presumed to consent to the administration of cardiopulmonary resuscitation in the event of cardiac or respiratory arrest, unless there is consent to the issuance of an order not to resuscitate as provided in this article." This raises the question as to whether it is ever legally permissible to enter a unilateral DNR order against the wishes of the patient.
On the other hand, Hawaii Revised Statutes 327E-13(d) notes: "This chapter shall not authorize or require a health care provider or institution to provide health care contrary to generally accepted health care standards." This has been interpreted as allowing the prevailing standard of care to serve as the yardstick of propriety.
Finally, the federal "antidumping" law governing emergency treatment in hospitals, widely known as EMTALA (Emergency Medical Treatment & Labor Act), requires all emergency departments to provide treatment necessary to prevent the material deterioration of the individual’s condition. This would always include the use of CPR unless specifically rejected by the patient or surrogate, because the law does not contain a "standard of care" or futility exception.
References
1. Capron, A.M., Abandoning a Waning Life. Hastings Center Report 1995;25:24-6.
2. In re the conservatorship of Helga M. Wanglie, No. PX-91-283, District Probate Division, 4th Judicial District of the County of Hennepin, State of Minnesota.
3. In the Matter of Baby "K", 16 F.3d F. Supp. 590 (E.D. VA 1993). WL 38674 (4th Cir. 1994).
4. In re K.I., 735 A.2d 448 (D.C. Ct. App. 1999).
5. Code of Ethics of the AMA, section 2.22, 2012-2013 edition.
7. Ann. Int. Med. 1990;112:949-54.
8. Code of Ethics of the AMA, section 2.035, 2012-2013 edition.
9. Barber v. Superior Court of Los Angeles County, 147 Cal. App. 3d 1006 (1983).
10. Kansas v. Naramore, 965 P.2d 211 (Kan. 1998).
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006). For additional information, readers may contact the author at [email protected].
Question: A 71-year-old woman with heart disease and breast cancer was hospitalized for uncontrolled diabetes and a hip fracture. There, she suffered two grand mal seizures that could not be controlled with anticonvulsants, and the patient lapsed into coma. Her daughter became the surrogate decision maker, and she made it clear that her mother always said she wanted everything done.
After several weeks, the physicians decided that further care would be futile. The chair of the ethics committee took the view that the family’s opinion was not relevant, because cardiopulmonary resuscitation (CPR) was not a genuine therapeutic option and would be "medically contraindicated, inhumane, and unethical." Accordingly, the attending physician entered a do-not-resuscitate (DNR) order despite strong protest from the daughter. The patient died shortly thereafter without receiving CPR.
In this actual case where the daughter filed a negligence lawsuit against the hospital, which of the following statements is incorrect?
A. The defendant’s expert relied upon the position paper of the American Thoracic Society, which states that life support "can be limited without the consent of patient or surrogate when the intervention is judged to be futile."
B. Futile intervention may be defined as treatment that would be highly unlikely to result in a meaningful survival of the patient.
C. The jury found that if competent, the patient would have wanted CPR and would have wanted ventilation until death.
D. The jury found such treatment would be futile.
E. The jury entered a verdict of negligence.
Answer: E. The above narrative is based on Gilgunn v. Massachusetts General Hospital (verdict issued April 21, 1995) and adapted from an article by the prominent ethicist Alexander M. Capron.1 All of the options listed were evident at trial, except that it was a defense verdict, i.e., no negligence. The case remains the best-known litigated example of medical futility. In earlier cases such as Wanglie2 and Baby K,3 the courts had avoided addressing the issue directly.
Gilgunn supports the notion that futility of CPR can trump a patient’s family insistence on having such intervention. But being a trial court verdict, it lacks the precedential authority that an appellate decision would confer.
The verdict also was not without its critics. As Mr. Capron wrote:
"But to allow Mrs. Gilgunn’s physicians to impose this view, however widely held, on their patient is the equivalent of allowing them to abandon the patient. We still need means ... to reach a social consensus on whether health professionals should have authority to decide, among the interventions patients (or surrogates) will accept, which will actually be provided and which they may withhold based upon their evaluation of the worth of the outcome. When we come to adopt such policies, we would do well to ponder long and hard before adopting a utilitarian measure that affords the waning lives of the most vulnerable in our society less protection from unilateral decisions by powerful professionals."1
In 1999, the District of Columbia Court of Appeals agreed with a trial court’s order to issue a DNR order for a neglected 2-year-old child who was born prematurely with serious medical problems and virtually nonexistent cognition, but who could still experience pain.4 The biological mother and putative father appealed, but the Court of Appeals affirmed. The court held, among other things, that the standard of proof required for issuance of a DNR order is clear and convincing evidence, and the applicable standard is the best interests of the child test rather than a substituted judgment standard. However, this case pitted the biological parents against a court-appointed guardian, rather than the medical providers.
The American Medical Association’s current Code of Ethics urges that when neither a patient nor surrogate is able or available to make a decision regarding CPR, an attending physician contemplating a DNR order should consult another physician or a hospital ethics committee if one is available.5 If the physician determines that a request for resuscitation would not be medically effective, "the physician should seek to resolve the conflict through a fair decision-making process, when time permits." In an earlier version,6 the AMA stated: "CPR may be withheld if, in the judgment of the treating physician, an attempt to resuscitate the patient would be futile."
DNR orders are at the heart of the futility conundrum, especially because CPR is a highly invasive, low-success procedure (notable exceptions exist, however). Medical futility denotes treatment that cannot confer an overall benefit on the whole person even if it can restore some physiologic variable.7 The Latin word "futilis" means leaky, and in Greek mythology, the daughters of Danaus were condemned in the underworld to draw water in leaky sieves, conveying the full meaning of futility.
The debate is over whether an intervention, however hopeless and ineffective, can ever be characterized as completely futile. Moreover, without absolute certainty, will this suffice to override a patient’s insistence on having that treatment?
Others have countered that absoluteness is an unrealistic standard, and that inhumane suffering and costs are relevant factors to ponder. The AMA has clearly stated that "physicians are not ethically obligated to deliver care that, in their best professional judgment, will not have a reasonable chance of benefiting their patients," and "patients should not be given treatments simply because they demand them."8
One legal consequence of discontinuing medical treatment that ends with a patient’s demise is the specter of criminal prosecution, although charges of homicide are unlikely to prevail.
In a landmark California case, Barber v. Superior Court of Los Angeles County,9 the court recognized that "a physician is authorized under the standards of medical practice to discontinue a form of therapy which in his medical judgment is useless. ... If the treating physicians have determined that continued use of a respirator is useless, then they may decide to discontinue it without fear of civil or criminal liability."
In Kansas v. Naramore,10 the state appeals court reversed and entered a verdict of acquittal despite a jury finding Dr. Naramore guilty over his provision of palliative treatment. The court noted that the burden of proof to establish the criminal guilt of a physician for acts arising out of providing medical treatment is higher than that necessary to find medical malpractice or to impose medical licensure discipline. It held that "with no direct evidence of criminal intent, it is highly disturbing that testimony by such an impressive array of apparently objective medical experts, who found the defendant’s actions to be not only noncriminal, but medically appropriate, can be dismissed as ‘unbelievable’ and not even capable of generating reasonable doubt."
Notwithstanding widely accepted ethical notions of medical futility, there are state and federal statutes touching on DNR orders that warrant careful attention.
For example, New York Public Health Law Section 2962, paragraph 1, states: "Every person admitted to a hospital shall be presumed to consent to the administration of cardiopulmonary resuscitation in the event of cardiac or respiratory arrest, unless there is consent to the issuance of an order not to resuscitate as provided in this article." This raises the question as to whether it is ever legally permissible to enter a unilateral DNR order against the wishes of the patient.
On the other hand, Hawaii Revised Statutes 327E-13(d) notes: "This chapter shall not authorize or require a health care provider or institution to provide health care contrary to generally accepted health care standards." This has been interpreted as allowing the prevailing standard of care to serve as the yardstick of propriety.
Finally, the federal "antidumping" law governing emergency treatment in hospitals, widely known as EMTALA (Emergency Medical Treatment & Labor Act), requires all emergency departments to provide treatment necessary to prevent the material deterioration of the individual’s condition. This would always include the use of CPR unless specifically rejected by the patient or surrogate, because the law does not contain a "standard of care" or futility exception.
References
1. Capron, A.M., Abandoning a Waning Life. Hastings Center Report 1995;25:24-6.
2. In re the conservatorship of Helga M. Wanglie, No. PX-91-283, District Probate Division, 4th Judicial District of the County of Hennepin, State of Minnesota.
3. In the Matter of Baby "K", 16 F.3d F. Supp. 590 (E.D. VA 1993). WL 38674 (4th Cir. 1994).
4. In re K.I., 735 A.2d 448 (D.C. Ct. App. 1999).
5. Code of Ethics of the AMA, section 2.22, 2012-2013 edition.
7. Ann. Int. Med. 1990;112:949-54.
8. Code of Ethics of the AMA, section 2.035, 2012-2013 edition.
9. Barber v. Superior Court of Los Angeles County, 147 Cal. App. 3d 1006 (1983).
10. Kansas v. Naramore, 965 P.2d 211 (Kan. 1998).
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006). For additional information, readers may contact the author at [email protected].
Question: A 71-year-old woman with heart disease and breast cancer was hospitalized for uncontrolled diabetes and a hip fracture. There, she suffered two grand mal seizures that could not be controlled with anticonvulsants, and the patient lapsed into coma. Her daughter became the surrogate decision maker, and she made it clear that her mother always said she wanted everything done.
After several weeks, the physicians decided that further care would be futile. The chair of the ethics committee took the view that the family’s opinion was not relevant, because cardiopulmonary resuscitation (CPR) was not a genuine therapeutic option and would be "medically contraindicated, inhumane, and unethical." Accordingly, the attending physician entered a do-not-resuscitate (DNR) order despite strong protest from the daughter. The patient died shortly thereafter without receiving CPR.
In this actual case where the daughter filed a negligence lawsuit against the hospital, which of the following statements is incorrect?
A. The defendant’s expert relied upon the position paper of the American Thoracic Society, which states that life support "can be limited without the consent of patient or surrogate when the intervention is judged to be futile."
B. Futile intervention may be defined as treatment that would be highly unlikely to result in a meaningful survival of the patient.
C. The jury found that if competent, the patient would have wanted CPR and would have wanted ventilation until death.
D. The jury found such treatment would be futile.
E. The jury entered a verdict of negligence.
Answer: E. The above narrative is based on Gilgunn v. Massachusetts General Hospital (verdict issued April 21, 1995) and adapted from an article by the prominent ethicist Alexander M. Capron.1 All of the options listed were evident at trial, except that it was a defense verdict, i.e., no negligence. The case remains the best-known litigated example of medical futility. In earlier cases such as Wanglie2 and Baby K,3 the courts had avoided addressing the issue directly.
Gilgunn supports the notion that futility of CPR can trump a patient’s family insistence on having such intervention. But being a trial court verdict, it lacks the precedential authority that an appellate decision would confer.
The verdict also was not without its critics. As Mr. Capron wrote:
"But to allow Mrs. Gilgunn’s physicians to impose this view, however widely held, on their patient is the equivalent of allowing them to abandon the patient. We still need means ... to reach a social consensus on whether health professionals should have authority to decide, among the interventions patients (or surrogates) will accept, which will actually be provided and which they may withhold based upon their evaluation of the worth of the outcome. When we come to adopt such policies, we would do well to ponder long and hard before adopting a utilitarian measure that affords the waning lives of the most vulnerable in our society less protection from unilateral decisions by powerful professionals."1
In 1999, the District of Columbia Court of Appeals agreed with a trial court’s order to issue a DNR order for a neglected 2-year-old child who was born prematurely with serious medical problems and virtually nonexistent cognition, but who could still experience pain.4 The biological mother and putative father appealed, but the Court of Appeals affirmed. The court held, among other things, that the standard of proof required for issuance of a DNR order is clear and convincing evidence, and the applicable standard is the best interests of the child test rather than a substituted judgment standard. However, this case pitted the biological parents against a court-appointed guardian, rather than the medical providers.
The American Medical Association’s current Code of Ethics urges that when neither a patient nor surrogate is able or available to make a decision regarding CPR, an attending physician contemplating a DNR order should consult another physician or a hospital ethics committee if one is available.5 If the physician determines that a request for resuscitation would not be medically effective, "the physician should seek to resolve the conflict through a fair decision-making process, when time permits." In an earlier version,6 the AMA stated: "CPR may be withheld if, in the judgment of the treating physician, an attempt to resuscitate the patient would be futile."
DNR orders are at the heart of the futility conundrum, especially because CPR is a highly invasive, low-success procedure (notable exceptions exist, however). Medical futility denotes treatment that cannot confer an overall benefit on the whole person even if it can restore some physiologic variable.7 The Latin word "futilis" means leaky, and in Greek mythology, the daughters of Danaus were condemned in the underworld to draw water in leaky sieves, conveying the full meaning of futility.
The debate is over whether an intervention, however hopeless and ineffective, can ever be characterized as completely futile. Moreover, without absolute certainty, will this suffice to override a patient’s insistence on having that treatment?
Others have countered that absoluteness is an unrealistic standard, and that inhumane suffering and costs are relevant factors to ponder. The AMA has clearly stated that "physicians are not ethically obligated to deliver care that, in their best professional judgment, will not have a reasonable chance of benefiting their patients," and "patients should not be given treatments simply because they demand them."8
One legal consequence of discontinuing medical treatment that ends with a patient’s demise is the specter of criminal prosecution, although charges of homicide are unlikely to prevail.
In a landmark California case, Barber v. Superior Court of Los Angeles County,9 the court recognized that "a physician is authorized under the standards of medical practice to discontinue a form of therapy which in his medical judgment is useless. ... If the treating physicians have determined that continued use of a respirator is useless, then they may decide to discontinue it without fear of civil or criminal liability."
In Kansas v. Naramore,10 the state appeals court reversed and entered a verdict of acquittal despite a jury finding Dr. Naramore guilty over his provision of palliative treatment. The court noted that the burden of proof to establish the criminal guilt of a physician for acts arising out of providing medical treatment is higher than that necessary to find medical malpractice or to impose medical licensure discipline. It held that "with no direct evidence of criminal intent, it is highly disturbing that testimony by such an impressive array of apparently objective medical experts, who found the defendant’s actions to be not only noncriminal, but medically appropriate, can be dismissed as ‘unbelievable’ and not even capable of generating reasonable doubt."
Notwithstanding widely accepted ethical notions of medical futility, there are state and federal statutes touching on DNR orders that warrant careful attention.
For example, New York Public Health Law Section 2962, paragraph 1, states: "Every person admitted to a hospital shall be presumed to consent to the administration of cardiopulmonary resuscitation in the event of cardiac or respiratory arrest, unless there is consent to the issuance of an order not to resuscitate as provided in this article." This raises the question as to whether it is ever legally permissible to enter a unilateral DNR order against the wishes of the patient.
On the other hand, Hawaii Revised Statutes 327E-13(d) notes: "This chapter shall not authorize or require a health care provider or institution to provide health care contrary to generally accepted health care standards." This has been interpreted as allowing the prevailing standard of care to serve as the yardstick of propriety.
Finally, the federal "antidumping" law governing emergency treatment in hospitals, widely known as EMTALA (Emergency Medical Treatment & Labor Act), requires all emergency departments to provide treatment necessary to prevent the material deterioration of the individual’s condition. This would always include the use of CPR unless specifically rejected by the patient or surrogate, because the law does not contain a "standard of care" or futility exception.
References
1. Capron, A.M., Abandoning a Waning Life. Hastings Center Report 1995;25:24-6.
2. In re the conservatorship of Helga M. Wanglie, No. PX-91-283, District Probate Division, 4th Judicial District of the County of Hennepin, State of Minnesota.
3. In the Matter of Baby "K", 16 F.3d F. Supp. 590 (E.D. VA 1993). WL 38674 (4th Cir. 1994).
4. In re K.I., 735 A.2d 448 (D.C. Ct. App. 1999).
5. Code of Ethics of the AMA, section 2.22, 2012-2013 edition.
7. Ann. Int. Med. 1990;112:949-54.
8. Code of Ethics of the AMA, section 2.035, 2012-2013 edition.
9. Barber v. Superior Court of Los Angeles County, 147 Cal. App. 3d 1006 (1983).
10. Kansas v. Naramore, 965 P.2d 211 (Kan. 1998).
Dr. Tan is professor emeritus of medicine and former adjunct professor of law at the University of Hawaii. This article is meant to be educational and does not constitute medical, ethical, or legal advice. It is adapted from the author’s book, "Medical Malpractice: Understanding the Law, Managing the Risk" (2006). For additional information, readers may contact the author at [email protected].
Even when curative-intent surgery fails, patients retain hope
PHOENIX – Hope remains for patients with advanced cancers, even after curative-intent surgery fails them, investigators reported at the annual Society of Surgical Oncology Cancer Symposium.
A study of the effects of an online decision aid for advance care planning showed that patients whose cancers had advanced despite initial curative-intent surgery had degrees of hope and desires for the aggressiveness of additional therapy similar to those of patients who had undergone palliative therapy only, said Dr. Niraj J. Gusani, FACS, of Penn State College of Medicine, Hershey, Pa.
"As we do more advance care planning, stratify patients, and get them into these programs to help them figure out what their wishes really are, we should treat everybody the same , and they should all have a durable advance care planning process, maybe using a decision aid such as this," he said.
The results show that outcomes beyond whether a patient lives or dies also matter, commented Dr. Steven Chen, FACS, from City of Hope Medical Center, Duarte, Calif.
"The thing that patients care about at least as much as how long they’re going to live is how well they’re going to live until they die," he said in an interview. Dr. Chen was not involved in the study, but comoderated the poster discussion session where it was presented.
"Almost all of my patients have very advanced cancers with poor prognosis, and it’s all about their quality of life and how they’re going to feel," Dr. Gusani agreed. "They intuitively get that they have a poor prognosis, but if we can get them through with a better experience with their time and they are able to maintain their hope, then it makes a world of difference for the patient and the family as well."
Multimedia advance care planning
Dr. Gusani and his colleagues looked at data from a clinical trial evaluating the effects of an online multimedia decision aid tool for patients with advanced-stage disease. The program, called Making Your Wishes Known: Planning Your Medical Future, was developed by two of the authors.
The program "provides tailored education, values-clarification exercises, and a decision-making tool that translates an individual’s values and preferences into a specific medical plan than can be implemented by a health care team," they wrote.
The investigators hypothesized that patients who went into surgery hoping for or expecting a cure might opt for more aggressive treatment at the end of life and have more hope and/or less hopelessness than patients who underwent treatments that they knew would not be curative.
The researchers administered the advance care planning program to 159 patients from the parent clinical trial, in which 200 patients with stage IV cancers who were judged to have less than 12 months to live were randomly assigned to receive either standard advance care planning or the decision aid. All of the patients had non–central nervous system and nonhematologic malignancies and were classified as having had initial curative-intent surgery or treatment without curative intent (e.g., palliative therapy and tumor debulking).
Making patients’ wishes known
Both at baseline and after the intervention, the patients were rated for hope on the 12-item Herth Hope Index, with scores from 12 (low hope) to 48 (high hope), and for hopelessness on the 7-item Beck Hopelessness Scale, in which 0 is no hopelessness and 7 is the highest level of despair.
The patients were also asked to rate their desires for aggressiveness of additional therapy by choosing one of six general wishes statements. For this scale, questions 1-4 were deemed to be more aggressive, and 5-6 less aggressive.
Additionally, patients who used the decision aid were rated on a treatment aggressiveness scale scored from 0 (least aggressive) to 40 (most) based on their responses to questions within the decision aid that asked about their wishes for eight different treatments (such as mechanical ventilation or cardiopulmonary resuscitation) under five clinical scenarios (such as stroke or dementia).
In all, 46 of 95 patients (48%) who had originally had curative-intent surgery received the control intervention, and 49 (52%) received the decision aid. Of the 64 patients who had non–curative-intent therapy, 35 (52%) received standard advance care planning and 31 (48%) received the Making Your Wishes Known aid.
After either intervention, patients who had curative-intent surgery had a slight but not significant increase in hope, and patients in the non–curative-intent group had a slight but not significant decrease. There was no significant between-group difference.
Similarly, each group had a slight decrease in hopelessness scores, with no significant differences between the groups.
There were also no significant variations in the distribution of the general wishes statements between the curative and noncurative groups or between the decision-aid and control groups, nor were there differences in the median treatment aggressiveness score of patients who used the decision aid, regardless of whether they had initially had curative-intent surgery.
"Regardless of what they choose in terms of their level of aggressiveness, the patients are more satisfied with their decision, they feel more informed, and caregiver satisfaction has improved with the decision aid compared with traditional materials," Dr. Gusani said.
"This is really important work," commented Dr. Sandra L. Wong of the University of Michigan Medical Center, Ann Arbor, who comoderated the poster discussion session.
The study was supported by Penn State College of Medicine. Dr. Gusani, Dr. Chen, and Dr. Wong reported having no financial conflicts of interest.
PHOENIX – Hope remains for patients with advanced cancers, even after curative-intent surgery fails them, investigators reported at the annual Society of Surgical Oncology Cancer Symposium.
A study of the effects of an online decision aid for advance care planning showed that patients whose cancers had advanced despite initial curative-intent surgery had degrees of hope and desires for the aggressiveness of additional therapy similar to those of patients who had undergone palliative therapy only, said Dr. Niraj J. Gusani, FACS, of Penn State College of Medicine, Hershey, Pa.
"As we do more advance care planning, stratify patients, and get them into these programs to help them figure out what their wishes really are, we should treat everybody the same , and they should all have a durable advance care planning process, maybe using a decision aid such as this," he said.
The results show that outcomes beyond whether a patient lives or dies also matter, commented Dr. Steven Chen, FACS, from City of Hope Medical Center, Duarte, Calif.
"The thing that patients care about at least as much as how long they’re going to live is how well they’re going to live until they die," he said in an interview. Dr. Chen was not involved in the study, but comoderated the poster discussion session where it was presented.
"Almost all of my patients have very advanced cancers with poor prognosis, and it’s all about their quality of life and how they’re going to feel," Dr. Gusani agreed. "They intuitively get that they have a poor prognosis, but if we can get them through with a better experience with their time and they are able to maintain their hope, then it makes a world of difference for the patient and the family as well."
Multimedia advance care planning
Dr. Gusani and his colleagues looked at data from a clinical trial evaluating the effects of an online multimedia decision aid tool for patients with advanced-stage disease. The program, called Making Your Wishes Known: Planning Your Medical Future, was developed by two of the authors.
The program "provides tailored education, values-clarification exercises, and a decision-making tool that translates an individual’s values and preferences into a specific medical plan than can be implemented by a health care team," they wrote.
The investigators hypothesized that patients who went into surgery hoping for or expecting a cure might opt for more aggressive treatment at the end of life and have more hope and/or less hopelessness than patients who underwent treatments that they knew would not be curative.
The researchers administered the advance care planning program to 159 patients from the parent clinical trial, in which 200 patients with stage IV cancers who were judged to have less than 12 months to live were randomly assigned to receive either standard advance care planning or the decision aid. All of the patients had non–central nervous system and nonhematologic malignancies and were classified as having had initial curative-intent surgery or treatment without curative intent (e.g., palliative therapy and tumor debulking).
Making patients’ wishes known
Both at baseline and after the intervention, the patients were rated for hope on the 12-item Herth Hope Index, with scores from 12 (low hope) to 48 (high hope), and for hopelessness on the 7-item Beck Hopelessness Scale, in which 0 is no hopelessness and 7 is the highest level of despair.
The patients were also asked to rate their desires for aggressiveness of additional therapy by choosing one of six general wishes statements. For this scale, questions 1-4 were deemed to be more aggressive, and 5-6 less aggressive.
Additionally, patients who used the decision aid were rated on a treatment aggressiveness scale scored from 0 (least aggressive) to 40 (most) based on their responses to questions within the decision aid that asked about their wishes for eight different treatments (such as mechanical ventilation or cardiopulmonary resuscitation) under five clinical scenarios (such as stroke or dementia).
In all, 46 of 95 patients (48%) who had originally had curative-intent surgery received the control intervention, and 49 (52%) received the decision aid. Of the 64 patients who had non–curative-intent therapy, 35 (52%) received standard advance care planning and 31 (48%) received the Making Your Wishes Known aid.
After either intervention, patients who had curative-intent surgery had a slight but not significant increase in hope, and patients in the non–curative-intent group had a slight but not significant decrease. There was no significant between-group difference.
Similarly, each group had a slight decrease in hopelessness scores, with no significant differences between the groups.
There were also no significant variations in the distribution of the general wishes statements between the curative and noncurative groups or between the decision-aid and control groups, nor were there differences in the median treatment aggressiveness score of patients who used the decision aid, regardless of whether they had initially had curative-intent surgery.
"Regardless of what they choose in terms of their level of aggressiveness, the patients are more satisfied with their decision, they feel more informed, and caregiver satisfaction has improved with the decision aid compared with traditional materials," Dr. Gusani said.
"This is really important work," commented Dr. Sandra L. Wong of the University of Michigan Medical Center, Ann Arbor, who comoderated the poster discussion session.
The study was supported by Penn State College of Medicine. Dr. Gusani, Dr. Chen, and Dr. Wong reported having no financial conflicts of interest.
PHOENIX – Hope remains for patients with advanced cancers, even after curative-intent surgery fails them, investigators reported at the annual Society of Surgical Oncology Cancer Symposium.
A study of the effects of an online decision aid for advance care planning showed that patients whose cancers had advanced despite initial curative-intent surgery had degrees of hope and desires for the aggressiveness of additional therapy similar to those of patients who had undergone palliative therapy only, said Dr. Niraj J. Gusani, FACS, of Penn State College of Medicine, Hershey, Pa.
"As we do more advance care planning, stratify patients, and get them into these programs to help them figure out what their wishes really are, we should treat everybody the same , and they should all have a durable advance care planning process, maybe using a decision aid such as this," he said.
The results show that outcomes beyond whether a patient lives or dies also matter, commented Dr. Steven Chen, FACS, from City of Hope Medical Center, Duarte, Calif.
"The thing that patients care about at least as much as how long they’re going to live is how well they’re going to live until they die," he said in an interview. Dr. Chen was not involved in the study, but comoderated the poster discussion session where it was presented.
"Almost all of my patients have very advanced cancers with poor prognosis, and it’s all about their quality of life and how they’re going to feel," Dr. Gusani agreed. "They intuitively get that they have a poor prognosis, but if we can get them through with a better experience with their time and they are able to maintain their hope, then it makes a world of difference for the patient and the family as well."
Multimedia advance care planning
Dr. Gusani and his colleagues looked at data from a clinical trial evaluating the effects of an online multimedia decision aid tool for patients with advanced-stage disease. The program, called Making Your Wishes Known: Planning Your Medical Future, was developed by two of the authors.
The program "provides tailored education, values-clarification exercises, and a decision-making tool that translates an individual’s values and preferences into a specific medical plan than can be implemented by a health care team," they wrote.
The investigators hypothesized that patients who went into surgery hoping for or expecting a cure might opt for more aggressive treatment at the end of life and have more hope and/or less hopelessness than patients who underwent treatments that they knew would not be curative.
The researchers administered the advance care planning program to 159 patients from the parent clinical trial, in which 200 patients with stage IV cancers who were judged to have less than 12 months to live were randomly assigned to receive either standard advance care planning or the decision aid. All of the patients had non–central nervous system and nonhematologic malignancies and were classified as having had initial curative-intent surgery or treatment without curative intent (e.g., palliative therapy and tumor debulking).
Making patients’ wishes known
Both at baseline and after the intervention, the patients were rated for hope on the 12-item Herth Hope Index, with scores from 12 (low hope) to 48 (high hope), and for hopelessness on the 7-item Beck Hopelessness Scale, in which 0 is no hopelessness and 7 is the highest level of despair.
The patients were also asked to rate their desires for aggressiveness of additional therapy by choosing one of six general wishes statements. For this scale, questions 1-4 were deemed to be more aggressive, and 5-6 less aggressive.
Additionally, patients who used the decision aid were rated on a treatment aggressiveness scale scored from 0 (least aggressive) to 40 (most) based on their responses to questions within the decision aid that asked about their wishes for eight different treatments (such as mechanical ventilation or cardiopulmonary resuscitation) under five clinical scenarios (such as stroke or dementia).
In all, 46 of 95 patients (48%) who had originally had curative-intent surgery received the control intervention, and 49 (52%) received the decision aid. Of the 64 patients who had non–curative-intent therapy, 35 (52%) received standard advance care planning and 31 (48%) received the Making Your Wishes Known aid.
After either intervention, patients who had curative-intent surgery had a slight but not significant increase in hope, and patients in the non–curative-intent group had a slight but not significant decrease. There was no significant between-group difference.
Similarly, each group had a slight decrease in hopelessness scores, with no significant differences between the groups.
There were also no significant variations in the distribution of the general wishes statements between the curative and noncurative groups or between the decision-aid and control groups, nor were there differences in the median treatment aggressiveness score of patients who used the decision aid, regardless of whether they had initially had curative-intent surgery.
"Regardless of what they choose in terms of their level of aggressiveness, the patients are more satisfied with their decision, they feel more informed, and caregiver satisfaction has improved with the decision aid compared with traditional materials," Dr. Gusani said.
"This is really important work," commented Dr. Sandra L. Wong of the University of Michigan Medical Center, Ann Arbor, who comoderated the poster discussion session.
The study was supported by Penn State College of Medicine. Dr. Gusani, Dr. Chen, and Dr. Wong reported having no financial conflicts of interest.
AT SSO 2014
Major finding: Patients with advanced-stage cancers who had initially undergone curative-intent surgery had levels of hope and desires for aggressiveness of therapy similar to those of patients who had undergone noncurative treatments.
Data source: Substudy of a randomized clinical trial comparing a computerized decision aid with standard advance care planning counseling.
Disclosures: The study was supported by Penn State College of Medicine. Dr. Gusani, Dr. Chen, and Dr. Wong reported having no financial conflicts of interest.
Palliative care shortens ICU, hospital stays, review shows
SAN FRANCISCO – Palliative care in the intensive care unit reduces the length of stay in the ICU and the hospital without changing mortality rates or family satisfaction, according to a review of the literature.
Although measurements of family satisfaction overall didn’t change much from palliative care of a loved one in the ICU, some measures of components of satisfaction increased with palliative care, such as improved communication with the physician, better consensus around the goals of care, and decreased anxiety and depression in family members, reported Dr. Rebecca A. Aslakson of Johns Hopkins University, Baltimore, and her colleagues.
Dr. Aslakson presented the findings at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
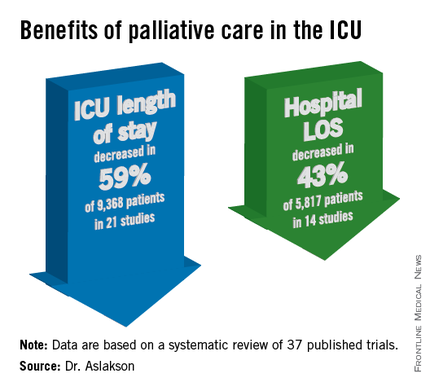
Dr. Aslakson and her associates were unable to perform a formal meta-analysis of the 37 published trials of palliative care in the ICU because of the heterogeneity of the studies, which looked at more than 40 different outcomes.
Instead, their systematic review grouped results under four outcomes that commonly were measured, and assessed those either by the number of studies or by the number of patients studied.
ICU length of stay decreased with palliative care in 13 of 21 studies (62%) that used this outcome and in 59% of 9,368 patients in those studies.
Hospital length of stay decreased with palliative care in 8 of 14 studies (57%) and in 43% of 5,817 patients. Family satisfaction did not decrease in any studies or families and increased in only 1 of 14 studies (7%) and in 2% of families of 4,927 patients, Dr. Aslakson and her colleagues reported (J. Palliat. Med. 2014;17:219-35).
Mortality rates did not change with palliative care in 14 of 16 studies (88%) that assessed mortality and in 57% of 5,969 patients in those studies. Mortality increased in one small study (6%) and decreased in one larger study (6%).
"Talking about big-picture issues and goals of care doesn’t lead to people dying," Dr. Aslakson said.
"No harm came in any of these studies." Some separate studies of palliative care outside of ICUs reported that this increases hope, "because people feel that they have more control over their choices and what’s happening to their loved ones," she added.
Integrative vs. consultative model
Dr. Aslakson and her associates also reviewed studies based on whether the interventions used integrative or consultative models of palliative care.
Generally, consultative models bring outsiders into the ICU to help provide palliative care, and integrative models train the ICU team to be the palliative care providers. In reality, the two models may overlap. For this review, the investigators applied mutually exclusive definitions to 36 of the studies.
In 18 studies of integrative interventions, members of the ICU team were the only caregivers in face-to-face interactions with the patient and families. In 18 studies of consultative interventions, palliative care providers included others besides the ICU team.
In the studies of integrative palliative care, ICU length of stay decreased with palliative care in four of nine studies (44%) that measured this outcome and in 52% of 6,963 patients in those studies, she reported. Hospital length of stay decreased in two of five studies (40%) and in 24% of 3,812 patients. Family satisfaction changed in none of 15 studies, and mortality decreased in 1 of 5 studies (20%) and in 34% of 3,807 patients.
In the studies of consultative care, ICU length of stay decreased with palliative care in 9 of 12 studies (75%) that measured this outcome and in 79% of 2,405 patients in those studies. Hospital length of stay decreased in six of nine studies (67%) and in 79% of 2,005 patients. Family satisfaction increased in one of four studies (25%) and in 21% of 429 patients. Mortality increased in 1 of 11 studies (9%) and in 5% of 2,162 patients.
One model isn’t necessarily better than the other, Dr. Aslakson said. Integrative palliative care may work best in a closed ICU with perhaps four or five intensivists in a relatively small unit. An integrative approach can be much more difficult in open or semiopen ICUs that have "40 different doctors floating around," she said. "We tried that in my unit, and it didn’t work that well."
Different ICUs need palliative care models that fit them. "Look at your unit, the way it works, and who the providers are, then look at the literature and see what matches that and what might work for your unit," she said.
Outcomes of better communication
A previous, separate review of the medical literature identified 21 controlled trials of 16 interventions to improve communication in ICUs between families and care providers. Overall, the interventions improved emotional outcomes for families and reduced ICU length of stay and treatment intensity (Chest 2011;139:543-54), she noted.
Yet another prior review of the literature reported that interventions to promote family meetings, use empathetic communication skills, and employ palliative care consultations improved family satisfaction and reduced ICU length of stay and the adverse effects of family bereavement (Curr. Opin. Crit. Care 2009;15:569-77).
Dr. Aslakson reported having no financial disclosures.
Dr. Jennifer Cox, FCCP, comments: Dr. Aslakson and colleagues’ systematic review adds to the body of literature that demonstrates no mortality increase when palliative care measures are initiated in the ICU. Shorter lengths of stay both in the ICU and hospital were other positive outcomes noted without a significant change in patient or family satisfaction.
These findings were independent of whether an integrative or consultative approach to palliative care was undertaken. This should encourage physicians to examine their practice setting and determine which approach meets the needs of their ICU and begin to utilize palliative care earlier and more aggressively without fear of increasing mortality.
[email protected]
On Twitter @sherryboschert
SAN FRANCISCO – Palliative care in the intensive care unit reduces the length of stay in the ICU and the hospital without changing mortality rates or family satisfaction, according to a review of the literature.
Although measurements of family satisfaction overall didn’t change much from palliative care of a loved one in the ICU, some measures of components of satisfaction increased with palliative care, such as improved communication with the physician, better consensus around the goals of care, and decreased anxiety and depression in family members, reported Dr. Rebecca A. Aslakson of Johns Hopkins University, Baltimore, and her colleagues.
Dr. Aslakson presented the findings at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.

Dr. Aslakson and her associates were unable to perform a formal meta-analysis of the 37 published trials of palliative care in the ICU because of the heterogeneity of the studies, which looked at more than 40 different outcomes.
Instead, their systematic review grouped results under four outcomes that commonly were measured, and assessed those either by the number of studies or by the number of patients studied.
ICU length of stay decreased with palliative care in 13 of 21 studies (62%) that used this outcome and in 59% of 9,368 patients in those studies.
Hospital length of stay decreased with palliative care in 8 of 14 studies (57%) and in 43% of 5,817 patients. Family satisfaction did not decrease in any studies or families and increased in only 1 of 14 studies (7%) and in 2% of families of 4,927 patients, Dr. Aslakson and her colleagues reported (J. Palliat. Med. 2014;17:219-35).
Mortality rates did not change with palliative care in 14 of 16 studies (88%) that assessed mortality and in 57% of 5,969 patients in those studies. Mortality increased in one small study (6%) and decreased in one larger study (6%).
"Talking about big-picture issues and goals of care doesn’t lead to people dying," Dr. Aslakson said.
"No harm came in any of these studies." Some separate studies of palliative care outside of ICUs reported that this increases hope, "because people feel that they have more control over their choices and what’s happening to their loved ones," she added.
Integrative vs. consultative model
Dr. Aslakson and her associates also reviewed studies based on whether the interventions used integrative or consultative models of palliative care.
Generally, consultative models bring outsiders into the ICU to help provide palliative care, and integrative models train the ICU team to be the palliative care providers. In reality, the two models may overlap. For this review, the investigators applied mutually exclusive definitions to 36 of the studies.
In 18 studies of integrative interventions, members of the ICU team were the only caregivers in face-to-face interactions with the patient and families. In 18 studies of consultative interventions, palliative care providers included others besides the ICU team.
In the studies of integrative palliative care, ICU length of stay decreased with palliative care in four of nine studies (44%) that measured this outcome and in 52% of 6,963 patients in those studies, she reported. Hospital length of stay decreased in two of five studies (40%) and in 24% of 3,812 patients. Family satisfaction changed in none of 15 studies, and mortality decreased in 1 of 5 studies (20%) and in 34% of 3,807 patients.
In the studies of consultative care, ICU length of stay decreased with palliative care in 9 of 12 studies (75%) that measured this outcome and in 79% of 2,405 patients in those studies. Hospital length of stay decreased in six of nine studies (67%) and in 79% of 2,005 patients. Family satisfaction increased in one of four studies (25%) and in 21% of 429 patients. Mortality increased in 1 of 11 studies (9%) and in 5% of 2,162 patients.
One model isn’t necessarily better than the other, Dr. Aslakson said. Integrative palliative care may work best in a closed ICU with perhaps four or five intensivists in a relatively small unit. An integrative approach can be much more difficult in open or semiopen ICUs that have "40 different doctors floating around," she said. "We tried that in my unit, and it didn’t work that well."
Different ICUs need palliative care models that fit them. "Look at your unit, the way it works, and who the providers are, then look at the literature and see what matches that and what might work for your unit," she said.
Outcomes of better communication
A previous, separate review of the medical literature identified 21 controlled trials of 16 interventions to improve communication in ICUs between families and care providers. Overall, the interventions improved emotional outcomes for families and reduced ICU length of stay and treatment intensity (Chest 2011;139:543-54), she noted.
Yet another prior review of the literature reported that interventions to promote family meetings, use empathetic communication skills, and employ palliative care consultations improved family satisfaction and reduced ICU length of stay and the adverse effects of family bereavement (Curr. Opin. Crit. Care 2009;15:569-77).
Dr. Aslakson reported having no financial disclosures.
Dr. Jennifer Cox, FCCP, comments: Dr. Aslakson and colleagues’ systematic review adds to the body of literature that demonstrates no mortality increase when palliative care measures are initiated in the ICU. Shorter lengths of stay both in the ICU and hospital were other positive outcomes noted without a significant change in patient or family satisfaction.
These findings were independent of whether an integrative or consultative approach to palliative care was undertaken. This should encourage physicians to examine their practice setting and determine which approach meets the needs of their ICU and begin to utilize palliative care earlier and more aggressively without fear of increasing mortality.
[email protected]
On Twitter @sherryboschert
SAN FRANCISCO – Palliative care in the intensive care unit reduces the length of stay in the ICU and the hospital without changing mortality rates or family satisfaction, according to a review of the literature.
Although measurements of family satisfaction overall didn’t change much from palliative care of a loved one in the ICU, some measures of components of satisfaction increased with palliative care, such as improved communication with the physician, better consensus around the goals of care, and decreased anxiety and depression in family members, reported Dr. Rebecca A. Aslakson of Johns Hopkins University, Baltimore, and her colleagues.
Dr. Aslakson presented the findings at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.

Dr. Aslakson and her associates were unable to perform a formal meta-analysis of the 37 published trials of palliative care in the ICU because of the heterogeneity of the studies, which looked at more than 40 different outcomes.
Instead, their systematic review grouped results under four outcomes that commonly were measured, and assessed those either by the number of studies or by the number of patients studied.
ICU length of stay decreased with palliative care in 13 of 21 studies (62%) that used this outcome and in 59% of 9,368 patients in those studies.
Hospital length of stay decreased with palliative care in 8 of 14 studies (57%) and in 43% of 5,817 patients. Family satisfaction did not decrease in any studies or families and increased in only 1 of 14 studies (7%) and in 2% of families of 4,927 patients, Dr. Aslakson and her colleagues reported (J. Palliat. Med. 2014;17:219-35).
Mortality rates did not change with palliative care in 14 of 16 studies (88%) that assessed mortality and in 57% of 5,969 patients in those studies. Mortality increased in one small study (6%) and decreased in one larger study (6%).
"Talking about big-picture issues and goals of care doesn’t lead to people dying," Dr. Aslakson said.
"No harm came in any of these studies." Some separate studies of palliative care outside of ICUs reported that this increases hope, "because people feel that they have more control over their choices and what’s happening to their loved ones," she added.
Integrative vs. consultative model
Dr. Aslakson and her associates also reviewed studies based on whether the interventions used integrative or consultative models of palliative care.
Generally, consultative models bring outsiders into the ICU to help provide palliative care, and integrative models train the ICU team to be the palliative care providers. In reality, the two models may overlap. For this review, the investigators applied mutually exclusive definitions to 36 of the studies.
In 18 studies of integrative interventions, members of the ICU team were the only caregivers in face-to-face interactions with the patient and families. In 18 studies of consultative interventions, palliative care providers included others besides the ICU team.
In the studies of integrative palliative care, ICU length of stay decreased with palliative care in four of nine studies (44%) that measured this outcome and in 52% of 6,963 patients in those studies, she reported. Hospital length of stay decreased in two of five studies (40%) and in 24% of 3,812 patients. Family satisfaction changed in none of 15 studies, and mortality decreased in 1 of 5 studies (20%) and in 34% of 3,807 patients.
In the studies of consultative care, ICU length of stay decreased with palliative care in 9 of 12 studies (75%) that measured this outcome and in 79% of 2,405 patients in those studies. Hospital length of stay decreased in six of nine studies (67%) and in 79% of 2,005 patients. Family satisfaction increased in one of four studies (25%) and in 21% of 429 patients. Mortality increased in 1 of 11 studies (9%) and in 5% of 2,162 patients.
One model isn’t necessarily better than the other, Dr. Aslakson said. Integrative palliative care may work best in a closed ICU with perhaps four or five intensivists in a relatively small unit. An integrative approach can be much more difficult in open or semiopen ICUs that have "40 different doctors floating around," she said. "We tried that in my unit, and it didn’t work that well."
Different ICUs need palliative care models that fit them. "Look at your unit, the way it works, and who the providers are, then look at the literature and see what matches that and what might work for your unit," she said.
Outcomes of better communication
A previous, separate review of the medical literature identified 21 controlled trials of 16 interventions to improve communication in ICUs between families and care providers. Overall, the interventions improved emotional outcomes for families and reduced ICU length of stay and treatment intensity (Chest 2011;139:543-54), she noted.
Yet another prior review of the literature reported that interventions to promote family meetings, use empathetic communication skills, and employ palliative care consultations improved family satisfaction and reduced ICU length of stay and the adverse effects of family bereavement (Curr. Opin. Crit. Care 2009;15:569-77).
Dr. Aslakson reported having no financial disclosures.
Dr. Jennifer Cox, FCCP, comments: Dr. Aslakson and colleagues’ systematic review adds to the body of literature that demonstrates no mortality increase when palliative care measures are initiated in the ICU. Shorter lengths of stay both in the ICU and hospital were other positive outcomes noted without a significant change in patient or family satisfaction.
These findings were independent of whether an integrative or consultative approach to palliative care was undertaken. This should encourage physicians to examine their practice setting and determine which approach meets the needs of their ICU and begin to utilize palliative care earlier and more aggressively without fear of increasing mortality.
[email protected]
On Twitter @sherryboschert
Relatively few in ICUs get end-of-life dialogue training
SAN FRANCISCO – Despite training recommendations, half of physicians and less than a third of nurses surveyed in adult intensive care units at 56 California hospitals reported receiving formal training in talking with patients and families about the end of life.
A 2008 consensus statement by the American College of Critical Care Medicine included a recommendation for end-of-life communication skills training for clinicians to improve the care of patients dying in ICUs ((Crit. Care Med. 2008;36:953-63).
Dr. Matthew H.R. Anstey and his associates approached 149 California hospitals to gauge the extent of implementation of this recommendation. At 56 hospitals, doctors and nurses who work in adult ICUs voluntarily completed an anonymous web-based survey. Eighty-four percent of the 1,363 respondents were nurses, he reported in a poster presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
Overall, 32% of the respondents said they had received formal training in communication skills. A significantly higher percentage of doctors had undergone training (50%) compared with nurses (29%), said Dr. Anstey, who is currently a lecturer in anesthesia at Harvard Medical School, Boston.
Sixty-six percent of all respondents agreed that "nurses are present during the communication of end-of-life information to the family" at their institution. Nurses were significantly more likely to agree with this statement (69%) than were doctors (52%).
Both doctors and nurses were very supportive of the idea of formal communication training for ICU providers. When asked about possible strategies to reduce inappropriate care for ICU patients, 91% of respondents said communication training would have a positive effect, Dr. Anstey reported.
This could be accomplished by requiring ICU physicians to complete a communication training module for ongoing credentialing, he said in an e-mail interview. Either individual hospitals could require this as part of credentialing for privileges to work in the ICU, or state medical boards could require it, similar to the California Medical Board’s requirement that physicians obtain some continuing medical education in pain management, he suggested.
The characteristics of participating hospitals were similar to those of nonparticipating hospitals in the sizes of the hospitals and ICUs, their regional location in California, and the proportions of hospitals that are teaching facilities.
The 93 nonparticipating hospitals were significantly more likely to be for-profit hospitals (59%) compared with participating hospitals (7%), and significantly less likely to be part of a hospital system containing more than three hospitals (54%) compared with participating hospitals (75%).
Dr. Anstey reported having no financial disclosures. His research was in conjunction with a Commonwealth Fund Harkness Fellowship in Health Care Policy and Practice for which he was placed at Kaiser Permanente in California.
Dr. Paul A. Selecky, FCCP, comments: Physicians are notorious about not doing a good job of communicating with patients in general, and when you focus on a vital subject as end-of-life care, it is of even greater importance. The findings in this study are not surprising. The unanswered question is how to fix it.
On Twitter @sherryboschert
SAN FRANCISCO – Despite training recommendations, half of physicians and less than a third of nurses surveyed in adult intensive care units at 56 California hospitals reported receiving formal training in talking with patients and families about the end of life.
A 2008 consensus statement by the American College of Critical Care Medicine included a recommendation for end-of-life communication skills training for clinicians to improve the care of patients dying in ICUs ((Crit. Care Med. 2008;36:953-63).
Dr. Matthew H.R. Anstey and his associates approached 149 California hospitals to gauge the extent of implementation of this recommendation. At 56 hospitals, doctors and nurses who work in adult ICUs voluntarily completed an anonymous web-based survey. Eighty-four percent of the 1,363 respondents were nurses, he reported in a poster presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
Overall, 32% of the respondents said they had received formal training in communication skills. A significantly higher percentage of doctors had undergone training (50%) compared with nurses (29%), said Dr. Anstey, who is currently a lecturer in anesthesia at Harvard Medical School, Boston.
Sixty-six percent of all respondents agreed that "nurses are present during the communication of end-of-life information to the family" at their institution. Nurses were significantly more likely to agree with this statement (69%) than were doctors (52%).
Both doctors and nurses were very supportive of the idea of formal communication training for ICU providers. When asked about possible strategies to reduce inappropriate care for ICU patients, 91% of respondents said communication training would have a positive effect, Dr. Anstey reported.
This could be accomplished by requiring ICU physicians to complete a communication training module for ongoing credentialing, he said in an e-mail interview. Either individual hospitals could require this as part of credentialing for privileges to work in the ICU, or state medical boards could require it, similar to the California Medical Board’s requirement that physicians obtain some continuing medical education in pain management, he suggested.
The characteristics of participating hospitals were similar to those of nonparticipating hospitals in the sizes of the hospitals and ICUs, their regional location in California, and the proportions of hospitals that are teaching facilities.
The 93 nonparticipating hospitals were significantly more likely to be for-profit hospitals (59%) compared with participating hospitals (7%), and significantly less likely to be part of a hospital system containing more than three hospitals (54%) compared with participating hospitals (75%).
Dr. Anstey reported having no financial disclosures. His research was in conjunction with a Commonwealth Fund Harkness Fellowship in Health Care Policy and Practice for which he was placed at Kaiser Permanente in California.
Dr. Paul A. Selecky, FCCP, comments: Physicians are notorious about not doing a good job of communicating with patients in general, and when you focus on a vital subject as end-of-life care, it is of even greater importance. The findings in this study are not surprising. The unanswered question is how to fix it.
On Twitter @sherryboschert
SAN FRANCISCO – Despite training recommendations, half of physicians and less than a third of nurses surveyed in adult intensive care units at 56 California hospitals reported receiving formal training in talking with patients and families about the end of life.
A 2008 consensus statement by the American College of Critical Care Medicine included a recommendation for end-of-life communication skills training for clinicians to improve the care of patients dying in ICUs ((Crit. Care Med. 2008;36:953-63).
Dr. Matthew H.R. Anstey and his associates approached 149 California hospitals to gauge the extent of implementation of this recommendation. At 56 hospitals, doctors and nurses who work in adult ICUs voluntarily completed an anonymous web-based survey. Eighty-four percent of the 1,363 respondents were nurses, he reported in a poster presentation at the Critical Care Congress, sponsored by the Society for Critical Care Medicine.
Overall, 32% of the respondents said they had received formal training in communication skills. A significantly higher percentage of doctors had undergone training (50%) compared with nurses (29%), said Dr. Anstey, who is currently a lecturer in anesthesia at Harvard Medical School, Boston.
Sixty-six percent of all respondents agreed that "nurses are present during the communication of end-of-life information to the family" at their institution. Nurses were significantly more likely to agree with this statement (69%) than were doctors (52%).
Both doctors and nurses were very supportive of the idea of formal communication training for ICU providers. When asked about possible strategies to reduce inappropriate care for ICU patients, 91% of respondents said communication training would have a positive effect, Dr. Anstey reported.
This could be accomplished by requiring ICU physicians to complete a communication training module for ongoing credentialing, he said in an e-mail interview. Either individual hospitals could require this as part of credentialing for privileges to work in the ICU, or state medical boards could require it, similar to the California Medical Board’s requirement that physicians obtain some continuing medical education in pain management, he suggested.
The characteristics of participating hospitals were similar to those of nonparticipating hospitals in the sizes of the hospitals and ICUs, their regional location in California, and the proportions of hospitals that are teaching facilities.
The 93 nonparticipating hospitals were significantly more likely to be for-profit hospitals (59%) compared with participating hospitals (7%), and significantly less likely to be part of a hospital system containing more than three hospitals (54%) compared with participating hospitals (75%).
Dr. Anstey reported having no financial disclosures. His research was in conjunction with a Commonwealth Fund Harkness Fellowship in Health Care Policy and Practice for which he was placed at Kaiser Permanente in California.
Dr. Paul A. Selecky, FCCP, comments: Physicians are notorious about not doing a good job of communicating with patients in general, and when you focus on a vital subject as end-of-life care, it is of even greater importance. The findings in this study are not surprising. The unanswered question is how to fix it.
On Twitter @sherryboschert
Major finding: Half of doctors and 29% of nurses in ICUs said they had received formal training in end-of-life communications.
Data source: A voluntary web-based survey of 1,363 doctors and nurses working in adult ICUs in 56 California hospitals.
Disclosures: Dr. Anstey reported having no financial disclosures. His research was in conjunction with a Commonwealth Fund Harkness Fellowship in Health Care Policy and Practice, for which he was placed at Kaiser Permanente in California.
Reconsidering comfort care
Recently, members of our palliative care team participated in the care of a man approaching the end of his life. The patient had suffered an in-hospital cardiac arrest 4 weeks earlier, and though he had survived the immediate event, it resulted in anoxic encephalopathy, which rendered him incapable of making decisions.
When it became clear that the patient was declining despite full support, the hospital’s ethics committee was convened to determine goals of care and next steps, as the patient had no family or surrogate decision maker. After determination that the hospital staff had exercised due diligence in attempting to locate a surrogate, the physicians involved reviewed the patient’s case and recommended a change in goals to comfort care. More than one member of the committee expressed confusion as to what interventions are and are not included in comfort care, including medically administered nutrition and hydration (MANH).
Comfort care has traditionally included medications for distressing symptoms (pain, dyspnea, nausea), personal care for hygiene, and choice of place of death (home, hospital, nursing facility), usually with the assistance of a hospice agency.
As the number and complexity of interventions used near the end of life expand, clinicians and hospital staff report confusion about whether these interventions, generally considered to be life-sustaining treatments, can also be considered comfort care. We generally find that when interventions are considered in the context of the patient’s goals of care, the dilemma is clarified. Often the situation is made more complicated by considering the interventions before settling on goals. Broadly speaking, goals of care are derived from a careful consideration (by patient, physician, and family) of the natural history of the illness, expected course and prognosis, and patient preferences.
In the case of the above-referenced patient, we were unable to ascertain his goals because of neurological impairment. We did know, however, that the patient had steadfastly avoided hospitals and medical care of any kind. The attending hospitalist, pulmonologist, and palliative care physician agreed that the patient’s clinical status was declining despite all available interventions, and that his constellation of medical problems constituted a terminal condition. The physicians agreed that future ICU admission, resuscitation, and other new interventions would only prolong his dying process, but not permit him to live outside the hospital. At that time, the patient was receiving nutrition and hydration via a Dobhoff tube, and was tolerating enteral nutrition without excessive residuals or pulmonary secretions.
As with other interventions, whether or not to consider MANH a part of comfort care is individualized. In this patient’s case, in the absence of evidence that he would not want MANH, it was continued. Other patients have expressed the wish that they would under no circumstances accept MANH while receiving comfort care. Both are correct as long as they reflect that patient’s wishes.
With respect to other interventions – including but not limited to BiPAP, inotrope infusion, chemotherapy, radiation therapy, and transfusions – whether or not they provide comfort is a decision to be made jointly by the patient and physician(s). As advances in medicine allow patients to live longer with serious illness, the definition of comfort care must also expand.
Dr. Steven Q. Simpson, FCCP commented: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact [email protected].
Dr. Fredholm and Dr. Bekanich are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
"Palliatively Speaking," appears monthly at ehospitalistnews.com.
Dr. Steven Q. Simpson, FCCP: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
Dr. Steven Q. Simpson, FCCP: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
Dr. Steven Q. Simpson, FCCP: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
Recently, members of our palliative care team participated in the care of a man approaching the end of his life. The patient had suffered an in-hospital cardiac arrest 4 weeks earlier, and though he had survived the immediate event, it resulted in anoxic encephalopathy, which rendered him incapable of making decisions.
When it became clear that the patient was declining despite full support, the hospital’s ethics committee was convened to determine goals of care and next steps, as the patient had no family or surrogate decision maker. After determination that the hospital staff had exercised due diligence in attempting to locate a surrogate, the physicians involved reviewed the patient’s case and recommended a change in goals to comfort care. More than one member of the committee expressed confusion as to what interventions are and are not included in comfort care, including medically administered nutrition and hydration (MANH).
Comfort care has traditionally included medications for distressing symptoms (pain, dyspnea, nausea), personal care for hygiene, and choice of place of death (home, hospital, nursing facility), usually with the assistance of a hospice agency.
As the number and complexity of interventions used near the end of life expand, clinicians and hospital staff report confusion about whether these interventions, generally considered to be life-sustaining treatments, can also be considered comfort care. We generally find that when interventions are considered in the context of the patient’s goals of care, the dilemma is clarified. Often the situation is made more complicated by considering the interventions before settling on goals. Broadly speaking, goals of care are derived from a careful consideration (by patient, physician, and family) of the natural history of the illness, expected course and prognosis, and patient preferences.
In the case of the above-referenced patient, we were unable to ascertain his goals because of neurological impairment. We did know, however, that the patient had steadfastly avoided hospitals and medical care of any kind. The attending hospitalist, pulmonologist, and palliative care physician agreed that the patient’s clinical status was declining despite all available interventions, and that his constellation of medical problems constituted a terminal condition. The physicians agreed that future ICU admission, resuscitation, and other new interventions would only prolong his dying process, but not permit him to live outside the hospital. At that time, the patient was receiving nutrition and hydration via a Dobhoff tube, and was tolerating enteral nutrition without excessive residuals or pulmonary secretions.
As with other interventions, whether or not to consider MANH a part of comfort care is individualized. In this patient’s case, in the absence of evidence that he would not want MANH, it was continued. Other patients have expressed the wish that they would under no circumstances accept MANH while receiving comfort care. Both are correct as long as they reflect that patient’s wishes.
With respect to other interventions – including but not limited to BiPAP, inotrope infusion, chemotherapy, radiation therapy, and transfusions – whether or not they provide comfort is a decision to be made jointly by the patient and physician(s). As advances in medicine allow patients to live longer with serious illness, the definition of comfort care must also expand.
Dr. Steven Q. Simpson, FCCP commented: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact [email protected].
Dr. Fredholm and Dr. Bekanich are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
"Palliatively Speaking," appears monthly at ehospitalistnews.com.
Recently, members of our palliative care team participated in the care of a man approaching the end of his life. The patient had suffered an in-hospital cardiac arrest 4 weeks earlier, and though he had survived the immediate event, it resulted in anoxic encephalopathy, which rendered him incapable of making decisions.
When it became clear that the patient was declining despite full support, the hospital’s ethics committee was convened to determine goals of care and next steps, as the patient had no family or surrogate decision maker. After determination that the hospital staff had exercised due diligence in attempting to locate a surrogate, the physicians involved reviewed the patient’s case and recommended a change in goals to comfort care. More than one member of the committee expressed confusion as to what interventions are and are not included in comfort care, including medically administered nutrition and hydration (MANH).
Comfort care has traditionally included medications for distressing symptoms (pain, dyspnea, nausea), personal care for hygiene, and choice of place of death (home, hospital, nursing facility), usually with the assistance of a hospice agency.
As the number and complexity of interventions used near the end of life expand, clinicians and hospital staff report confusion about whether these interventions, generally considered to be life-sustaining treatments, can also be considered comfort care. We generally find that when interventions are considered in the context of the patient’s goals of care, the dilemma is clarified. Often the situation is made more complicated by considering the interventions before settling on goals. Broadly speaking, goals of care are derived from a careful consideration (by patient, physician, and family) of the natural history of the illness, expected course and prognosis, and patient preferences.
In the case of the above-referenced patient, we were unable to ascertain his goals because of neurological impairment. We did know, however, that the patient had steadfastly avoided hospitals and medical care of any kind. The attending hospitalist, pulmonologist, and palliative care physician agreed that the patient’s clinical status was declining despite all available interventions, and that his constellation of medical problems constituted a terminal condition. The physicians agreed that future ICU admission, resuscitation, and other new interventions would only prolong his dying process, but not permit him to live outside the hospital. At that time, the patient was receiving nutrition and hydration via a Dobhoff tube, and was tolerating enteral nutrition without excessive residuals or pulmonary secretions.
As with other interventions, whether or not to consider MANH a part of comfort care is individualized. In this patient’s case, in the absence of evidence that he would not want MANH, it was continued. Other patients have expressed the wish that they would under no circumstances accept MANH while receiving comfort care. Both are correct as long as they reflect that patient’s wishes.
With respect to other interventions – including but not limited to BiPAP, inotrope infusion, chemotherapy, radiation therapy, and transfusions – whether or not they provide comfort is a decision to be made jointly by the patient and physician(s). As advances in medicine allow patients to live longer with serious illness, the definition of comfort care must also expand.
Dr. Steven Q. Simpson, FCCP commented: Dr. Fredholm and Dr. Beckanich adeptly discuss how to weigh possible life-sustaining measures in terms of whether they are providing comfort for patients and how to ensure that such treatments are discussed with patients in that context.
Additionally, they provide a very nice example of how to proceed when patients cannot communicate for themselves and have no family or other surrogates to speak for them.
Join us in the Critical Care NetWork’s eCommunity.
If you’re interested in more about these topics, you can join a discussion on this topic within the Critical Care e-Community. Simply log in to ecommunity.chestnet.org and find the Critical Care group If you’re not part of the Critical Care NetWork, log in to chestnet.org and add the Critical Care NetWork to your profile.
Questions? Contact [email protected].
Dr. Fredholm and Dr. Bekanich are codirectors of Seton Palliative Care, part of the University of Texas Southwestern Residency Programs in Austin.
"Palliatively Speaking," appears monthly at ehospitalistnews.com.