User login
Helping patients move forward following traumatic brain injury
THE CASE
Declan M*, a 42-year-old man, presents as a new patient for general medical care. One year ago, he sustained a severe frontal traumatic brain injury (TBI) when he was hit by a car while crossing a street. He developed a subdural hematoma and was in a coma for 6 days. He also had fractured ribs and a fractured left foot. When he regained consciousness, he had posttraumatic amnesia. He also had executive function deficits and memory difficulties, so a guardian was appointed.
Mr. M no longer works as an auto mechanic, a career he once greatly enjoyed. Mr. M’s guardian reports that recently, Mr. M has lost interest in activities he’d previously enjoyed, is frequently irritable, has poor sleep, is socially isolated, and is spending increasing amounts of time at home. When his new primary care physician (PCP) enters the examining room, Mr. M is seated in a chair with his arms folded across his chest. He states that he is “fine” and just needs to “get a doctor.”
●
*This patient is an amalgam of patients for whom the author has provided care.
TBI ranges from mild to severe and can produce a number of profound effects that are a direct—or indirect—result of the physical injury.1 The location and the severity of the injury affect symptoms.2 Even mild TBI can cause impairment, and severe TBI can lead to broad cognitive, behavioral, and physical difficulties. As numbers of TBI cases increase globally, primary care providers need to recognize the symptoms and assess accordingly.1 The Acute Concussion Evaluation (ACE; Physician/Clinician Office Version) facilitates a structured evaluation for patients presenting with possible TBI symptoms. It can easily be accessed on the Centers for Disease Control and Prevention website.3
Direct effects of TBI include impulsivity, depression, reduced frustration tolerance, reduced motivation, low awareness, and insomnia and other sleep difficulties.4,5 Depression may also result indirectly from, or be exacerbated by, new posttraumatic limitations and lifestyle changes as well as loss of career and community.4 Both direct and indirect depression often manifest as feelings of hopelessness and worthlessness and a lack of interest in once enjoyable activities. Depression can worsen other TBI sequelae such as difficulty concentrating, lack of initiation, flat affect, irritability, reduced independence, reduced functional performance, loss of inhibition, and physical pain.6
Nationwide, most mental health concerns continue to be addressed in the primary care setting.7 Individuals with TBI experience major depression at a rate 5 to 6 times higher than those in the general population, with a prevalence rate of 45%.8
Suicide. The subject of suicide must be explored with survivors of TBI; evidence suggests a correlation between TBI, depression, and increased risk for suicide.9 Among those who have TBI, as many as 22% experience suicidal ideation; the risk of suicide in survivors of severe TBI is 3 to 4 times the risk in the general population.10 Additionally, suicidality in this context appears to be a chronic concern; therefore, carefully assess for its presence no matter how long ago the TBI occurred.10
Additional TBI-associated health concerns
Grief and loss. We so often focus on death as the only cause for grief, but grief can occur for other types of loss, as well. Individuals with TBI often experience a radical negative change in self-concept after their injury, which is associated with feelings of grief.11 Helping patients recognize that they are grieving the loss of the person they once were can help set a framework for their experience.
Continue to: Relationship loss
Relationship loss. Many people with TBI lose close relationships.12 This can be due to life changes such as job loss, loss of function or ability to do previously enjoyed activities, or personality changes. These relationship losses can affect a person profoundly.12 Going forward, they may have difficulty trusting others, for example.
Existential issues. Many people with TBI also find that cognitive deficits prevent them from engaging in formerly meaningful work. For example, Mr. M lost his longstanding career as an auto mechanic and therefore part of his identity. Not being able to find purpose and meaning can be a strong contributor to coping difficulties in those with TBI.13
Chronic pain. More than half of people with TBI experience chronic pain. Headaches are the most common pain condition among all TBI survivors.14
Substance use disorders. The directionality of substance use disorders and TBI is not always clear; however, most evidence suggests that substance abuse is highly prevalent, premorbid, and often a contributing factor in TBI (eg, car accidents).15 Alcohol abuse is the most common risk factor, followed by drug abuse.16 Substance abuse may be exacerbated after TBI when it becomes a coping mechanism under worsening stressors; additionally, executive function deficits or other neurologic problems may result in poor decision-making with regard to substance use.15 While substance abuse may decline in the immediate post-TBI period, it can return to pre-injury levels within a year.17
Selective serotonin reuptake inhibitors may help
Few studies have explored the efficacy of antidepressant medication in TBI survivors. In a controlled study of patients with TBI, Fann and colleagues18 found no significant improvement in depression symptoms between sertraline and a placebo. However, they did note some possibilities for this lack of significance: socially isolated TBI survivors in the placebo group may have demonstrated improvement in depression symptoms because of increased social interaction;
Continue to: Other research has found...
Other research has found that sertraline improved both depression and quality of life for men with post-TBI depression.19 In a meta-analysis of 4 studies, Paraschakis and Katsanos20 found that sertraline demonstrated a “trend toward significance” in the treatment of depression among patients with TBI. Silverberg and Panenka21 argue that selective serotonin reuptake inhibitors should be used as first-line treatment for depression in survivors of TBI. They note that in non-randomized studies, treatment effects with antidepressants are significant. Additionally, patients who do not respond to the first antidepressant prescribed will often respond to adjunctive or different medications. Finally, they argue that depression measures can capture symptoms related to the physical brain injury, in addition to symptoms of depression, thus confounding results.
THE CASE
Mr. M’s chart showed that he was not taking any medication and that he had no history of substance abuse or tobacco use. He refused to fill out the Patient Health Questionnaire (PHQ)-2. His guardian said that Mr. M was spending much of his time at home, and that he used to be an avid painter and guitar player but had not engaged in either activity for months. Furthermore, Mr. M used to enjoy working out but did so rarely now.
During the interview, the PCP was careful to make eye contact with Mr. M as well as his guardian, thereby making sure Mr. M was part of the conversation about his care. Pacing of questions was deliberate and unhurried; a return visit would be scheduled to further explore any concerns not covered in this visit. This collaborative, inclusive, patient-centered approach to the clinical interview seemed to place Mr. M at ease. When his guardian said he thought Mr. M was depressed, Mr. M agreed. Although Mr. M still refused to fill out the PHQ-2, he was now willing to answer questions about depression. He acknowledged that he was feeling hopeless and took little pleasure in activities he used to enjoy, thereby indicating a positive screen for depression.
The PCP opted to read the PHQ-9 questions aloud, and Mr. M agreed with most of the items but strongly denied suicidal ideation, citing his religious faith.
The PCP determined that Mr. M’s depression was likely a combination of the direct and indirect effects of his TBI. A quantitative estimate based on Mr. M’s report yielded a PHQ-9 score of 17, indicating moderately severe depression.
Continue to: In addition to building rapport...
In addition to building rapport, careful listening garnered important information about Mr. M. For example, until his accident and subsequent depression, Mr. M had long prioritized his physical health through diet and exercise. He followed a vegetarian diet but recently had little appetite and was eating one microwaveable meal a day. He had an irregular sleep schedule and struggled with insomnia. He lost his closest long-term relationship after his accident due to difficulties with affect regulation. He also lost his job as he could no longer cognitively handle the tasks required.
Hearing Mr. M’s story provided the opportunity to customize education about self-management skills including regular diet, exercise, and sleep hygiene. Due to limited visit time, the PCP elected to use this first visit to focus on sleep and depression. As cognitive behavioral therapy (CBT) for insomnia is first-line treatment for both primary insomnia and insomnia due to a medical condition such as TBI,5 a sleep aid was not prescribed. Fortunately, the clinic psychologist who offered CBT was able to join the interview to meet Mr. M and explain the treatment.
Mr. M expressed some initial reluctance to try an antidepressant. However, acknowledging he “just hasn’t been the same” since his TBI, he agreed to a prescription for sertraline and said he hoped it could make him “more like [he] was.”
RETURN VISIT
Four weeks after Mr. M began taking sertraline and participating in weekly CBT sessions, he returned for a follow-up visit with his PCP. He had a noticeably brighter affect, and his guardian reported that he had been playing the guitar again. Mr. M said that he had more energy as a result of improved sleep and mood, and that he felt like his “thinking was clearer.” Mr. M noted that he never thought he would meet with a psychologist but was finding CBT for insomnia helpful.
The psychologist’s notes proposed a treatment plan that would also include targeted grief and existential therapies to address Mr. M’s sudden life changes. At this visit, Mr. M admitted that his reading comprehension and speed were negatively affected by the accident and said this is why he did not wish to fill out the PHQ-2. But he was again willing to have the PHQ-9 questions read to him with his guardian’s support. Results showed a score of 6, indicating mild depression.
A follow-up appointment with Mr. M was scheduled for 6 weeks later, and the team was confident he was getting the behavioral and mental health support he needed through medication and therapy.
CORRESPONDENCE
Elizabeth Imbesi, PhD, VA Ann Arbor Healthcare System, 2215 Fuller Road, Ann Arbor, MI 48105; [email protected]
1. CDC. Traumatic brain injury & concussion. 2020. Accessed May 19, 2022. www.cdc.gov/traumaticbraininjury/index.html
2. Finset A, Anderson S. Coping strategies in patients with acquired brain injury: relationships between coping, apathy, depression and lesion location. Brain Inj. 2009;14:887-905. doi: 10.1080/026990500445718
3. CDC. Gioia G, Collins M. Acute concussion evaluation. 2006. Accessed May 19, 2022. www.cdc.gov/headsup/pdfs/providers/ace_v2-a.pdf
4. Prigatano GP. Psychotherapy and the process of coping with a brain disorder. Oral presentation at: American Psychological Association annual convention. August 2015; Toronto, Canada.
5. Ouellet M, Beaulieu-Bonneau S, Savard J, Morin C. Insomnia and Fatigue After Traumatic Brain Injury: A CBT Approach to Assessment and Treatment. Elsevier Academic Press: 2019.
6. Lewis FD, Horn GH. Depression following traumatic brain injury: impact on post-hospital residential rehabilitation outcomes. NeuroRehabilitation. 2017;40:401-410. doi: 10.3233/NRE-161427
7. Barkil-Oteo A. Collaborative care for depression in primary care: how psychiatry could “troubleshoot” current treatments and practices. Yale J Bio Med. 2013;86:139-146.
8. Whelan-Goodinson R, Ponsford J, Johnston L, et al. Psychiatric disorders following traumatic brain injury: their nature and frequency. J Head Trauma Rehabil. 2009;24:324-332. doi: 10.1097/HTR.0b013e3181a712aa
9. Reeves RR, Laizer JT. Traumatic brain injury and suicide. J Psychosoc Nurs Ment Health Serv. 2012;50:32-38. doi: 10.3928/02793695-20120207-02
10. Simpson G, Tate R. Suicidality in people surviving a traumatic brain injury: Prevalence, risk factors and implications for clinical management. Brain Inj. 2007;21:1335-1351. doi: 10.1080/02699050701785542
11. Carroll E, Coetzer R. Identity, grief and self-awareness after traumatic brain injury. Neuropsychol Rehabil. 2011;21:289-305. doi: 10.1080/09602011.2011.555972
12. Salas CE, Casassus M, Rowlands L, et al. “Relating through sameness”: a qualitative study of friendship and social isolation in chronic traumatic brain injury. Neuropsychol Rehabil. 2018;28:1161-1178. doi: 10.1080/09602011.2016.1247730
13. Hinkebein JA, Stucky R. Coping with traumatic brain injury: existential challenges and managing hope. In: Martz E, Livneh H, eds. Coping with Chronic Illness and Disability: Theoretical, Empirical, and Clinical Aspects. Springer Science & Business Media; 2007:389-409.
14. Khoury S, Benavides R. Pain with traumatic brain injury and psychological disorders. Prog Neuropsychopharmacol and Biol Psychiatry. 2018;87:224-233. doi: 10.1016/j.pnpbp.2017.06.007
15. Bjork JM, Grant SJ. Does traumatic brain injury increase risk for substance abuse? J Neurotrauma. 2009;26:1077-1082. doi: 10.1089/neu.2008.0849
16. Unsworth DJ, Mathias JL. Traumatic brain injury and alcohol/substance abuse: a Bayesian meta-analysis comparing the outcomes of people with and without a history of abuse. J Clin Exp Neuropsychol. 2017,39:547-562. doi: 10.1080/13803395.2016.1248812
17. Beaulieu-Bonneau S, St-Onge F, Blackburn M, et al. Alcohol and drug use before and during the first year after traumatic brain injury. J Head Trauma Rehabil. 2018;33:E51-E60. doi: 10.1097/HTR.0000000000000341
18. Fann JR, Bombardier CH, Temkin N, et al. Sertraline for major depression during the year following traumatic brain injury: a randomized control trial. J Head Trauma Rehabil. 2017;32:332-342. doi: 10.1097/HTR.0000000000000322
19. Ansari A, Jain A, Sharma A, et al. Role of sertraline in posttraumatic brain injury depression and quality of life in TBI. Asian J Neurosurg. 2014;9:182-188. doi: 10.4103/1793-5482.146597
20. Paraschakis A, Katsanos AH. Antidepressants for depression associated with traumatic brain injury: a meta-analytical study of randomized control trials. East Asian Arch Psychiatry. 2017;27:142-149.
21. Silverberg ND, Panenka WJ. Antidepressants for depression after concussion and traumatic brain injury are still best practice. BMC Psychiatry. 2019;19:100. doi: 10.1186/s12888-019-2076-9
THE CASE
Declan M*, a 42-year-old man, presents as a new patient for general medical care. One year ago, he sustained a severe frontal traumatic brain injury (TBI) when he was hit by a car while crossing a street. He developed a subdural hematoma and was in a coma for 6 days. He also had fractured ribs and a fractured left foot. When he regained consciousness, he had posttraumatic amnesia. He also had executive function deficits and memory difficulties, so a guardian was appointed.
Mr. M no longer works as an auto mechanic, a career he once greatly enjoyed. Mr. M’s guardian reports that recently, Mr. M has lost interest in activities he’d previously enjoyed, is frequently irritable, has poor sleep, is socially isolated, and is spending increasing amounts of time at home. When his new primary care physician (PCP) enters the examining room, Mr. M is seated in a chair with his arms folded across his chest. He states that he is “fine” and just needs to “get a doctor.”
●
*This patient is an amalgam of patients for whom the author has provided care.
TBI ranges from mild to severe and can produce a number of profound effects that are a direct—or indirect—result of the physical injury.1 The location and the severity of the injury affect symptoms.2 Even mild TBI can cause impairment, and severe TBI can lead to broad cognitive, behavioral, and physical difficulties. As numbers of TBI cases increase globally, primary care providers need to recognize the symptoms and assess accordingly.1 The Acute Concussion Evaluation (ACE; Physician/Clinician Office Version) facilitates a structured evaluation for patients presenting with possible TBI symptoms. It can easily be accessed on the Centers for Disease Control and Prevention website.3
Direct effects of TBI include impulsivity, depression, reduced frustration tolerance, reduced motivation, low awareness, and insomnia and other sleep difficulties.4,5 Depression may also result indirectly from, or be exacerbated by, new posttraumatic limitations and lifestyle changes as well as loss of career and community.4 Both direct and indirect depression often manifest as feelings of hopelessness and worthlessness and a lack of interest in once enjoyable activities. Depression can worsen other TBI sequelae such as difficulty concentrating, lack of initiation, flat affect, irritability, reduced independence, reduced functional performance, loss of inhibition, and physical pain.6
Nationwide, most mental health concerns continue to be addressed in the primary care setting.7 Individuals with TBI experience major depression at a rate 5 to 6 times higher than those in the general population, with a prevalence rate of 45%.8
Suicide. The subject of suicide must be explored with survivors of TBI; evidence suggests a correlation between TBI, depression, and increased risk for suicide.9 Among those who have TBI, as many as 22% experience suicidal ideation; the risk of suicide in survivors of severe TBI is 3 to 4 times the risk in the general population.10 Additionally, suicidality in this context appears to be a chronic concern; therefore, carefully assess for its presence no matter how long ago the TBI occurred.10
Additional TBI-associated health concerns
Grief and loss. We so often focus on death as the only cause for grief, but grief can occur for other types of loss, as well. Individuals with TBI often experience a radical negative change in self-concept after their injury, which is associated with feelings of grief.11 Helping patients recognize that they are grieving the loss of the person they once were can help set a framework for their experience.
Continue to: Relationship loss
Relationship loss. Many people with TBI lose close relationships.12 This can be due to life changes such as job loss, loss of function or ability to do previously enjoyed activities, or personality changes. These relationship losses can affect a person profoundly.12 Going forward, they may have difficulty trusting others, for example.
Existential issues. Many people with TBI also find that cognitive deficits prevent them from engaging in formerly meaningful work. For example, Mr. M lost his longstanding career as an auto mechanic and therefore part of his identity. Not being able to find purpose and meaning can be a strong contributor to coping difficulties in those with TBI.13
Chronic pain. More than half of people with TBI experience chronic pain. Headaches are the most common pain condition among all TBI survivors.14
Substance use disorders. The directionality of substance use disorders and TBI is not always clear; however, most evidence suggests that substance abuse is highly prevalent, premorbid, and often a contributing factor in TBI (eg, car accidents).15 Alcohol abuse is the most common risk factor, followed by drug abuse.16 Substance abuse may be exacerbated after TBI when it becomes a coping mechanism under worsening stressors; additionally, executive function deficits or other neurologic problems may result in poor decision-making with regard to substance use.15 While substance abuse may decline in the immediate post-TBI period, it can return to pre-injury levels within a year.17
Selective serotonin reuptake inhibitors may help
Few studies have explored the efficacy of antidepressant medication in TBI survivors. In a controlled study of patients with TBI, Fann and colleagues18 found no significant improvement in depression symptoms between sertraline and a placebo. However, they did note some possibilities for this lack of significance: socially isolated TBI survivors in the placebo group may have demonstrated improvement in depression symptoms because of increased social interaction;
Continue to: Other research has found...
Other research has found that sertraline improved both depression and quality of life for men with post-TBI depression.19 In a meta-analysis of 4 studies, Paraschakis and Katsanos20 found that sertraline demonstrated a “trend toward significance” in the treatment of depression among patients with TBI. Silverberg and Panenka21 argue that selective serotonin reuptake inhibitors should be used as first-line treatment for depression in survivors of TBI. They note that in non-randomized studies, treatment effects with antidepressants are significant. Additionally, patients who do not respond to the first antidepressant prescribed will often respond to adjunctive or different medications. Finally, they argue that depression measures can capture symptoms related to the physical brain injury, in addition to symptoms of depression, thus confounding results.
THE CASE
Mr. M’s chart showed that he was not taking any medication and that he had no history of substance abuse or tobacco use. He refused to fill out the Patient Health Questionnaire (PHQ)-2. His guardian said that Mr. M was spending much of his time at home, and that he used to be an avid painter and guitar player but had not engaged in either activity for months. Furthermore, Mr. M used to enjoy working out but did so rarely now.
During the interview, the PCP was careful to make eye contact with Mr. M as well as his guardian, thereby making sure Mr. M was part of the conversation about his care. Pacing of questions was deliberate and unhurried; a return visit would be scheduled to further explore any concerns not covered in this visit. This collaborative, inclusive, patient-centered approach to the clinical interview seemed to place Mr. M at ease. When his guardian said he thought Mr. M was depressed, Mr. M agreed. Although Mr. M still refused to fill out the PHQ-2, he was now willing to answer questions about depression. He acknowledged that he was feeling hopeless and took little pleasure in activities he used to enjoy, thereby indicating a positive screen for depression.
The PCP opted to read the PHQ-9 questions aloud, and Mr. M agreed with most of the items but strongly denied suicidal ideation, citing his religious faith.
The PCP determined that Mr. M’s depression was likely a combination of the direct and indirect effects of his TBI. A quantitative estimate based on Mr. M’s report yielded a PHQ-9 score of 17, indicating moderately severe depression.
Continue to: In addition to building rapport...
In addition to building rapport, careful listening garnered important information about Mr. M. For example, until his accident and subsequent depression, Mr. M had long prioritized his physical health through diet and exercise. He followed a vegetarian diet but recently had little appetite and was eating one microwaveable meal a day. He had an irregular sleep schedule and struggled with insomnia. He lost his closest long-term relationship after his accident due to difficulties with affect regulation. He also lost his job as he could no longer cognitively handle the tasks required.
Hearing Mr. M’s story provided the opportunity to customize education about self-management skills including regular diet, exercise, and sleep hygiene. Due to limited visit time, the PCP elected to use this first visit to focus on sleep and depression. As cognitive behavioral therapy (CBT) for insomnia is first-line treatment for both primary insomnia and insomnia due to a medical condition such as TBI,5 a sleep aid was not prescribed. Fortunately, the clinic psychologist who offered CBT was able to join the interview to meet Mr. M and explain the treatment.
Mr. M expressed some initial reluctance to try an antidepressant. However, acknowledging he “just hasn’t been the same” since his TBI, he agreed to a prescription for sertraline and said he hoped it could make him “more like [he] was.”
RETURN VISIT
Four weeks after Mr. M began taking sertraline and participating in weekly CBT sessions, he returned for a follow-up visit with his PCP. He had a noticeably brighter affect, and his guardian reported that he had been playing the guitar again. Mr. M said that he had more energy as a result of improved sleep and mood, and that he felt like his “thinking was clearer.” Mr. M noted that he never thought he would meet with a psychologist but was finding CBT for insomnia helpful.
The psychologist’s notes proposed a treatment plan that would also include targeted grief and existential therapies to address Mr. M’s sudden life changes. At this visit, Mr. M admitted that his reading comprehension and speed were negatively affected by the accident and said this is why he did not wish to fill out the PHQ-2. But he was again willing to have the PHQ-9 questions read to him with his guardian’s support. Results showed a score of 6, indicating mild depression.
A follow-up appointment with Mr. M was scheduled for 6 weeks later, and the team was confident he was getting the behavioral and mental health support he needed through medication and therapy.
CORRESPONDENCE
Elizabeth Imbesi, PhD, VA Ann Arbor Healthcare System, 2215 Fuller Road, Ann Arbor, MI 48105; [email protected]
THE CASE
Declan M*, a 42-year-old man, presents as a new patient for general medical care. One year ago, he sustained a severe frontal traumatic brain injury (TBI) when he was hit by a car while crossing a street. He developed a subdural hematoma and was in a coma for 6 days. He also had fractured ribs and a fractured left foot. When he regained consciousness, he had posttraumatic amnesia. He also had executive function deficits and memory difficulties, so a guardian was appointed.
Mr. M no longer works as an auto mechanic, a career he once greatly enjoyed. Mr. M’s guardian reports that recently, Mr. M has lost interest in activities he’d previously enjoyed, is frequently irritable, has poor sleep, is socially isolated, and is spending increasing amounts of time at home. When his new primary care physician (PCP) enters the examining room, Mr. M is seated in a chair with his arms folded across his chest. He states that he is “fine” and just needs to “get a doctor.”
●
*This patient is an amalgam of patients for whom the author has provided care.
TBI ranges from mild to severe and can produce a number of profound effects that are a direct—or indirect—result of the physical injury.1 The location and the severity of the injury affect symptoms.2 Even mild TBI can cause impairment, and severe TBI can lead to broad cognitive, behavioral, and physical difficulties. As numbers of TBI cases increase globally, primary care providers need to recognize the symptoms and assess accordingly.1 The Acute Concussion Evaluation (ACE; Physician/Clinician Office Version) facilitates a structured evaluation for patients presenting with possible TBI symptoms. It can easily be accessed on the Centers for Disease Control and Prevention website.3
Direct effects of TBI include impulsivity, depression, reduced frustration tolerance, reduced motivation, low awareness, and insomnia and other sleep difficulties.4,5 Depression may also result indirectly from, or be exacerbated by, new posttraumatic limitations and lifestyle changes as well as loss of career and community.4 Both direct and indirect depression often manifest as feelings of hopelessness and worthlessness and a lack of interest in once enjoyable activities. Depression can worsen other TBI sequelae such as difficulty concentrating, lack of initiation, flat affect, irritability, reduced independence, reduced functional performance, loss of inhibition, and physical pain.6
Nationwide, most mental health concerns continue to be addressed in the primary care setting.7 Individuals with TBI experience major depression at a rate 5 to 6 times higher than those in the general population, with a prevalence rate of 45%.8
Suicide. The subject of suicide must be explored with survivors of TBI; evidence suggests a correlation between TBI, depression, and increased risk for suicide.9 Among those who have TBI, as many as 22% experience suicidal ideation; the risk of suicide in survivors of severe TBI is 3 to 4 times the risk in the general population.10 Additionally, suicidality in this context appears to be a chronic concern; therefore, carefully assess for its presence no matter how long ago the TBI occurred.10
Additional TBI-associated health concerns
Grief and loss. We so often focus on death as the only cause for grief, but grief can occur for other types of loss, as well. Individuals with TBI often experience a radical negative change in self-concept after their injury, which is associated with feelings of grief.11 Helping patients recognize that they are grieving the loss of the person they once were can help set a framework for their experience.
Continue to: Relationship loss
Relationship loss. Many people with TBI lose close relationships.12 This can be due to life changes such as job loss, loss of function or ability to do previously enjoyed activities, or personality changes. These relationship losses can affect a person profoundly.12 Going forward, they may have difficulty trusting others, for example.
Existential issues. Many people with TBI also find that cognitive deficits prevent them from engaging in formerly meaningful work. For example, Mr. M lost his longstanding career as an auto mechanic and therefore part of his identity. Not being able to find purpose and meaning can be a strong contributor to coping difficulties in those with TBI.13
Chronic pain. More than half of people with TBI experience chronic pain. Headaches are the most common pain condition among all TBI survivors.14
Substance use disorders. The directionality of substance use disorders and TBI is not always clear; however, most evidence suggests that substance abuse is highly prevalent, premorbid, and often a contributing factor in TBI (eg, car accidents).15 Alcohol abuse is the most common risk factor, followed by drug abuse.16 Substance abuse may be exacerbated after TBI when it becomes a coping mechanism under worsening stressors; additionally, executive function deficits or other neurologic problems may result in poor decision-making with regard to substance use.15 While substance abuse may decline in the immediate post-TBI period, it can return to pre-injury levels within a year.17
Selective serotonin reuptake inhibitors may help
Few studies have explored the efficacy of antidepressant medication in TBI survivors. In a controlled study of patients with TBI, Fann and colleagues18 found no significant improvement in depression symptoms between sertraline and a placebo. However, they did note some possibilities for this lack of significance: socially isolated TBI survivors in the placebo group may have demonstrated improvement in depression symptoms because of increased social interaction;
Continue to: Other research has found...
Other research has found that sertraline improved both depression and quality of life for men with post-TBI depression.19 In a meta-analysis of 4 studies, Paraschakis and Katsanos20 found that sertraline demonstrated a “trend toward significance” in the treatment of depression among patients with TBI. Silverberg and Panenka21 argue that selective serotonin reuptake inhibitors should be used as first-line treatment for depression in survivors of TBI. They note that in non-randomized studies, treatment effects with antidepressants are significant. Additionally, patients who do not respond to the first antidepressant prescribed will often respond to adjunctive or different medications. Finally, they argue that depression measures can capture symptoms related to the physical brain injury, in addition to symptoms of depression, thus confounding results.
THE CASE
Mr. M’s chart showed that he was not taking any medication and that he had no history of substance abuse or tobacco use. He refused to fill out the Patient Health Questionnaire (PHQ)-2. His guardian said that Mr. M was spending much of his time at home, and that he used to be an avid painter and guitar player but had not engaged in either activity for months. Furthermore, Mr. M used to enjoy working out but did so rarely now.
During the interview, the PCP was careful to make eye contact with Mr. M as well as his guardian, thereby making sure Mr. M was part of the conversation about his care. Pacing of questions was deliberate and unhurried; a return visit would be scheduled to further explore any concerns not covered in this visit. This collaborative, inclusive, patient-centered approach to the clinical interview seemed to place Mr. M at ease. When his guardian said he thought Mr. M was depressed, Mr. M agreed. Although Mr. M still refused to fill out the PHQ-2, he was now willing to answer questions about depression. He acknowledged that he was feeling hopeless and took little pleasure in activities he used to enjoy, thereby indicating a positive screen for depression.
The PCP opted to read the PHQ-9 questions aloud, and Mr. M agreed with most of the items but strongly denied suicidal ideation, citing his religious faith.
The PCP determined that Mr. M’s depression was likely a combination of the direct and indirect effects of his TBI. A quantitative estimate based on Mr. M’s report yielded a PHQ-9 score of 17, indicating moderately severe depression.
Continue to: In addition to building rapport...
In addition to building rapport, careful listening garnered important information about Mr. M. For example, until his accident and subsequent depression, Mr. M had long prioritized his physical health through diet and exercise. He followed a vegetarian diet but recently had little appetite and was eating one microwaveable meal a day. He had an irregular sleep schedule and struggled with insomnia. He lost his closest long-term relationship after his accident due to difficulties with affect regulation. He also lost his job as he could no longer cognitively handle the tasks required.
Hearing Mr. M’s story provided the opportunity to customize education about self-management skills including regular diet, exercise, and sleep hygiene. Due to limited visit time, the PCP elected to use this first visit to focus on sleep and depression. As cognitive behavioral therapy (CBT) for insomnia is first-line treatment for both primary insomnia and insomnia due to a medical condition such as TBI,5 a sleep aid was not prescribed. Fortunately, the clinic psychologist who offered CBT was able to join the interview to meet Mr. M and explain the treatment.
Mr. M expressed some initial reluctance to try an antidepressant. However, acknowledging he “just hasn’t been the same” since his TBI, he agreed to a prescription for sertraline and said he hoped it could make him “more like [he] was.”
RETURN VISIT
Four weeks after Mr. M began taking sertraline and participating in weekly CBT sessions, he returned for a follow-up visit with his PCP. He had a noticeably brighter affect, and his guardian reported that he had been playing the guitar again. Mr. M said that he had more energy as a result of improved sleep and mood, and that he felt like his “thinking was clearer.” Mr. M noted that he never thought he would meet with a psychologist but was finding CBT for insomnia helpful.
The psychologist’s notes proposed a treatment plan that would also include targeted grief and existential therapies to address Mr. M’s sudden life changes. At this visit, Mr. M admitted that his reading comprehension and speed were negatively affected by the accident and said this is why he did not wish to fill out the PHQ-2. But he was again willing to have the PHQ-9 questions read to him with his guardian’s support. Results showed a score of 6, indicating mild depression.
A follow-up appointment with Mr. M was scheduled for 6 weeks later, and the team was confident he was getting the behavioral and mental health support he needed through medication and therapy.
CORRESPONDENCE
Elizabeth Imbesi, PhD, VA Ann Arbor Healthcare System, 2215 Fuller Road, Ann Arbor, MI 48105; [email protected]
1. CDC. Traumatic brain injury & concussion. 2020. Accessed May 19, 2022. www.cdc.gov/traumaticbraininjury/index.html
2. Finset A, Anderson S. Coping strategies in patients with acquired brain injury: relationships between coping, apathy, depression and lesion location. Brain Inj. 2009;14:887-905. doi: 10.1080/026990500445718
3. CDC. Gioia G, Collins M. Acute concussion evaluation. 2006. Accessed May 19, 2022. www.cdc.gov/headsup/pdfs/providers/ace_v2-a.pdf
4. Prigatano GP. Psychotherapy and the process of coping with a brain disorder. Oral presentation at: American Psychological Association annual convention. August 2015; Toronto, Canada.
5. Ouellet M, Beaulieu-Bonneau S, Savard J, Morin C. Insomnia and Fatigue After Traumatic Brain Injury: A CBT Approach to Assessment and Treatment. Elsevier Academic Press: 2019.
6. Lewis FD, Horn GH. Depression following traumatic brain injury: impact on post-hospital residential rehabilitation outcomes. NeuroRehabilitation. 2017;40:401-410. doi: 10.3233/NRE-161427
7. Barkil-Oteo A. Collaborative care for depression in primary care: how psychiatry could “troubleshoot” current treatments and practices. Yale J Bio Med. 2013;86:139-146.
8. Whelan-Goodinson R, Ponsford J, Johnston L, et al. Psychiatric disorders following traumatic brain injury: their nature and frequency. J Head Trauma Rehabil. 2009;24:324-332. doi: 10.1097/HTR.0b013e3181a712aa
9. Reeves RR, Laizer JT. Traumatic brain injury and suicide. J Psychosoc Nurs Ment Health Serv. 2012;50:32-38. doi: 10.3928/02793695-20120207-02
10. Simpson G, Tate R. Suicidality in people surviving a traumatic brain injury: Prevalence, risk factors and implications for clinical management. Brain Inj. 2007;21:1335-1351. doi: 10.1080/02699050701785542
11. Carroll E, Coetzer R. Identity, grief and self-awareness after traumatic brain injury. Neuropsychol Rehabil. 2011;21:289-305. doi: 10.1080/09602011.2011.555972
12. Salas CE, Casassus M, Rowlands L, et al. “Relating through sameness”: a qualitative study of friendship and social isolation in chronic traumatic brain injury. Neuropsychol Rehabil. 2018;28:1161-1178. doi: 10.1080/09602011.2016.1247730
13. Hinkebein JA, Stucky R. Coping with traumatic brain injury: existential challenges and managing hope. In: Martz E, Livneh H, eds. Coping with Chronic Illness and Disability: Theoretical, Empirical, and Clinical Aspects. Springer Science & Business Media; 2007:389-409.
14. Khoury S, Benavides R. Pain with traumatic brain injury and psychological disorders. Prog Neuropsychopharmacol and Biol Psychiatry. 2018;87:224-233. doi: 10.1016/j.pnpbp.2017.06.007
15. Bjork JM, Grant SJ. Does traumatic brain injury increase risk for substance abuse? J Neurotrauma. 2009;26:1077-1082. doi: 10.1089/neu.2008.0849
16. Unsworth DJ, Mathias JL. Traumatic brain injury and alcohol/substance abuse: a Bayesian meta-analysis comparing the outcomes of people with and without a history of abuse. J Clin Exp Neuropsychol. 2017,39:547-562. doi: 10.1080/13803395.2016.1248812
17. Beaulieu-Bonneau S, St-Onge F, Blackburn M, et al. Alcohol and drug use before and during the first year after traumatic brain injury. J Head Trauma Rehabil. 2018;33:E51-E60. doi: 10.1097/HTR.0000000000000341
18. Fann JR, Bombardier CH, Temkin N, et al. Sertraline for major depression during the year following traumatic brain injury: a randomized control trial. J Head Trauma Rehabil. 2017;32:332-342. doi: 10.1097/HTR.0000000000000322
19. Ansari A, Jain A, Sharma A, et al. Role of sertraline in posttraumatic brain injury depression and quality of life in TBI. Asian J Neurosurg. 2014;9:182-188. doi: 10.4103/1793-5482.146597
20. Paraschakis A, Katsanos AH. Antidepressants for depression associated with traumatic brain injury: a meta-analytical study of randomized control trials. East Asian Arch Psychiatry. 2017;27:142-149.
21. Silverberg ND, Panenka WJ. Antidepressants for depression after concussion and traumatic brain injury are still best practice. BMC Psychiatry. 2019;19:100. doi: 10.1186/s12888-019-2076-9
1. CDC. Traumatic brain injury & concussion. 2020. Accessed May 19, 2022. www.cdc.gov/traumaticbraininjury/index.html
2. Finset A, Anderson S. Coping strategies in patients with acquired brain injury: relationships between coping, apathy, depression and lesion location. Brain Inj. 2009;14:887-905. doi: 10.1080/026990500445718
3. CDC. Gioia G, Collins M. Acute concussion evaluation. 2006. Accessed May 19, 2022. www.cdc.gov/headsup/pdfs/providers/ace_v2-a.pdf
4. Prigatano GP. Psychotherapy and the process of coping with a brain disorder. Oral presentation at: American Psychological Association annual convention. August 2015; Toronto, Canada.
5. Ouellet M, Beaulieu-Bonneau S, Savard J, Morin C. Insomnia and Fatigue After Traumatic Brain Injury: A CBT Approach to Assessment and Treatment. Elsevier Academic Press: 2019.
6. Lewis FD, Horn GH. Depression following traumatic brain injury: impact on post-hospital residential rehabilitation outcomes. NeuroRehabilitation. 2017;40:401-410. doi: 10.3233/NRE-161427
7. Barkil-Oteo A. Collaborative care for depression in primary care: how psychiatry could “troubleshoot” current treatments and practices. Yale J Bio Med. 2013;86:139-146.
8. Whelan-Goodinson R, Ponsford J, Johnston L, et al. Psychiatric disorders following traumatic brain injury: their nature and frequency. J Head Trauma Rehabil. 2009;24:324-332. doi: 10.1097/HTR.0b013e3181a712aa
9. Reeves RR, Laizer JT. Traumatic brain injury and suicide. J Psychosoc Nurs Ment Health Serv. 2012;50:32-38. doi: 10.3928/02793695-20120207-02
10. Simpson G, Tate R. Suicidality in people surviving a traumatic brain injury: Prevalence, risk factors and implications for clinical management. Brain Inj. 2007;21:1335-1351. doi: 10.1080/02699050701785542
11. Carroll E, Coetzer R. Identity, grief and self-awareness after traumatic brain injury. Neuropsychol Rehabil. 2011;21:289-305. doi: 10.1080/09602011.2011.555972
12. Salas CE, Casassus M, Rowlands L, et al. “Relating through sameness”: a qualitative study of friendship and social isolation in chronic traumatic brain injury. Neuropsychol Rehabil. 2018;28:1161-1178. doi: 10.1080/09602011.2016.1247730
13. Hinkebein JA, Stucky R. Coping with traumatic brain injury: existential challenges and managing hope. In: Martz E, Livneh H, eds. Coping with Chronic Illness and Disability: Theoretical, Empirical, and Clinical Aspects. Springer Science & Business Media; 2007:389-409.
14. Khoury S, Benavides R. Pain with traumatic brain injury and psychological disorders. Prog Neuropsychopharmacol and Biol Psychiatry. 2018;87:224-233. doi: 10.1016/j.pnpbp.2017.06.007
15. Bjork JM, Grant SJ. Does traumatic brain injury increase risk for substance abuse? J Neurotrauma. 2009;26:1077-1082. doi: 10.1089/neu.2008.0849
16. Unsworth DJ, Mathias JL. Traumatic brain injury and alcohol/substance abuse: a Bayesian meta-analysis comparing the outcomes of people with and without a history of abuse. J Clin Exp Neuropsychol. 2017,39:547-562. doi: 10.1080/13803395.2016.1248812
17. Beaulieu-Bonneau S, St-Onge F, Blackburn M, et al. Alcohol and drug use before and during the first year after traumatic brain injury. J Head Trauma Rehabil. 2018;33:E51-E60. doi: 10.1097/HTR.0000000000000341
18. Fann JR, Bombardier CH, Temkin N, et al. Sertraline for major depression during the year following traumatic brain injury: a randomized control trial. J Head Trauma Rehabil. 2017;32:332-342. doi: 10.1097/HTR.0000000000000322
19. Ansari A, Jain A, Sharma A, et al. Role of sertraline in posttraumatic brain injury depression and quality of life in TBI. Asian J Neurosurg. 2014;9:182-188. doi: 10.4103/1793-5482.146597
20. Paraschakis A, Katsanos AH. Antidepressants for depression associated with traumatic brain injury: a meta-analytical study of randomized control trials. East Asian Arch Psychiatry. 2017;27:142-149.
21. Silverberg ND, Panenka WJ. Antidepressants for depression after concussion and traumatic brain injury are still best practice. BMC Psychiatry. 2019;19:100. doi: 10.1186/s12888-019-2076-9
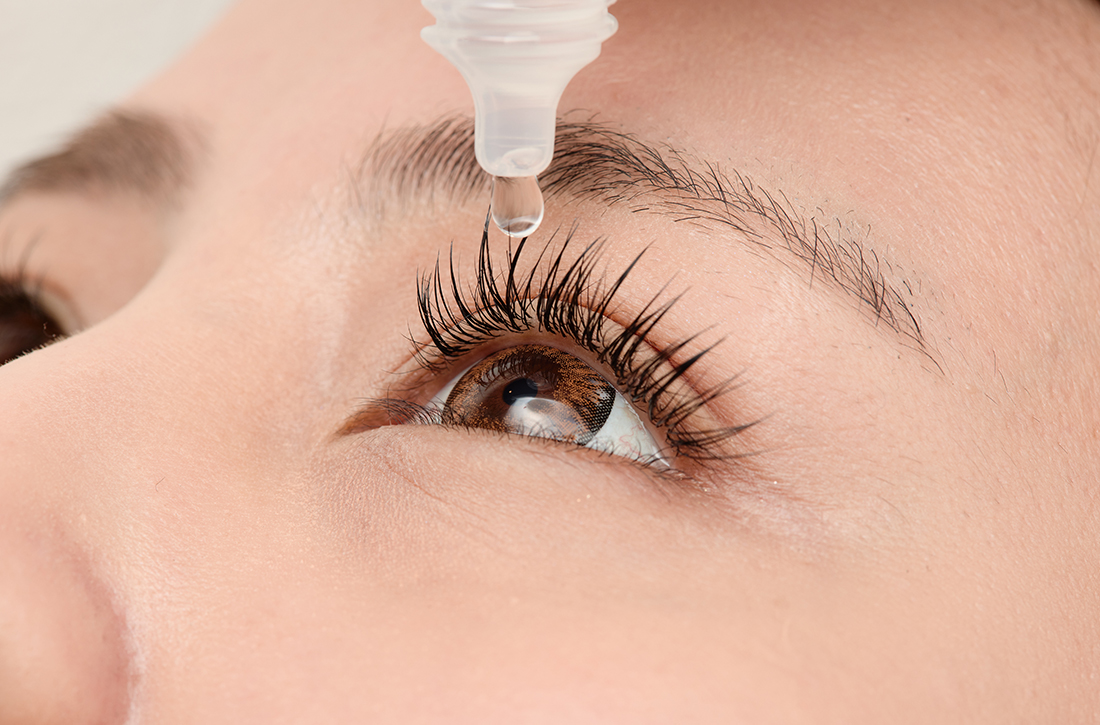
Migraine relief in 20 minutes using eyedrops?
ILLUSTRATIVE CASE
A 35-year-old woman with no significant past medical history presents for follow-up of migraine. At the previous visit, she was prescribed sumatriptan for abortive therapy. However, she has been having significant adverse effect intolerance from the oral formulation, and the nasal formulation is cost prohibitive. What can you recommend as an alternative abortive therapy for this patient’s migraine?
Migraine is among the most common causes of disability worldwide, affecting more than 10% of the global population.2 The prevalence of migraine is between 2.6% and 21.7% across multiple countries.3 On a scale of 0% to 100%, disability caused by migraine is 43.3%, comparable to the first 2 days after an acute myocardial infarction (42.2%) and severe dementia (43.8%).4
Abortive therapy for acute migraine includes nonsteroidal anti-inflammatory drugs (NSAIDs),
Nausea and vomiting, common components of migraine (that are included in International Classification of Headache Disorders, 3rd edition [ICHD-3] criteria for migraine5) present obstacles to effective oral administration if experienced by the patient. In addition, for migraine refractory to first-line treatments, abortive options—including the recently approved
Two oral beta-blockers, propranolol and timolol, are approved by the US Food and Drug Administration for migraine prophylaxis. Unfortunately, oral beta-blockers are ineffective for abortive treatment.7 Ophthalmic timolol is typically used in the treatment of glaucoma, but there have been case reports describing its benefits in acute migraine treatment.8,9 In addition, ophthalmic timolol is far cheaper than medications such as ubrogepant.10 A 2014 case series of 7 patients discussed ophthalmic beta-blockers as an effective and possibly cheaper option for acute migraine treatment.8 A randomized, crossover, placebo-controlled pilot study of 198 migraine attacks in 10 participants using timolol eyedrops for abortive therapy found timolol was not significantly more effective than placebo.9 However, it was an underpowered pilot study, with a lack of masking and an imperfect placebo. The trial discussed here was a controlled, prospective study investigating topical beta-blockers for acute migraine treatment.
STUDY SUMMARY
Crossover study achieved primary endpoint in pain reduction
This randomized, single-center, double-masked, crossover trial compared timolol maleate ophthalmic solution 0.5% with placebo among 43 patients ages 12 or older presenting with a diagnosis of migraine based on ICHD-3 (beta) criteria. Patients were eligible if they had not taken any antimigraine medications for at least 1 month prior to the study and were excluded if they had taken systemic beta-blockers at baseline, or had asthma, bradyarrhythmias, or cardiac dysfunction.
Patients were randomized 1:1 to treatment with timolol maleate 0.5% eyedrops or placebo. At the earliest onset of migraine, patients used 1 drop of timolol maleate 0.5% or placebo in each eye; if they experienced no relief after 10 minutes, they used a second drop or matching placebo. Patients were instructed to score their headache pain on a 10-point scale prior to using the eyedrops and then again 20 minutes after treatment. If a patient had migraine with aura, they were asked to use the eyedrops at the onset of the aura but measure their score at headache onset. If no headaches developed within 20 minutes of the aura, the episode was not included for analysis. All patients were permitted to use their standard oral rescue medication if no relief occurred after 20 minutes of pain onset.
Continue to: The groups were observed...
The groups were observed for 3 months and then followed for a 1-month washout period, during which they received no study medications. The groups were then crossed over to the other treatment and were observed for another 3 months. The primary outcome was a reduction in pain score by 4 or more points, or to 0 on a 10-point pain scale, 20 minutes after treatment. The secondary outcome was nonuse of oral rescue medication.
Forty-three patients were included in a modified intention-to-treat analysis. The primary outcome was achieved in 233 of 284 (82%) timolol-treated migraines, compared to 38 of 271 (14%) placebo-treated migraines (percentage difference = 68 percentage points; 95% CI, 62-74 percentage points; P < .001). The mean pain score at the onset of migraine attacks was 6.01 for those treated with timolol and 5.93 for those treated with placebo. Patients treated with timolol had a reduction in pain of 5.98 points, compared with 0.93 points after using placebo (difference = 5.05; 95% CI, 4.19-5.91). No attacks included in the data required oral rescue medications, and there were no systemic adverse effects from the timolol eyedrops.
WHAT’S NEW
Evidence of benefit as abortive therapy for acute migraine
This randomized controlled trial (RCT) showed evidence to support timolol maleate ophthalmic solution 0.5% vs placebo for treatment of acute migraine by significantly reducing pain when taken at the onset of an acute migraine attack.
CAVEATS
Single-center trial, measuring limited response time
The generalizability of this RCT is limited because it was a single-center trial with a study population from a single region in India. It is unknown whether pain relief, adverse effects, or adherence would differ for the global population. Additionally, only migraines with headache were included in the analysis, limiting non-headache migraine subgroup-directed treatment. Also, this trial evaluated only the response to treatment at 20 minutes, and it is unknown if pain response continued for several hours. Headaches that began more than 20 minutes after the onset of aura were not evaluated.
CHALLENGES TO IMPLEMENTATION
Timolol’s systemic adverse effects require caution
Systemic beta-blocker effects (eg, bradycardia, hypotension, drowsiness, and bronchospasm) from topical timolol have been reported. Caution should be used when prescribing timolol for patients with current cardiovascular and pulmonary conditions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
- Kurian A, Reghunadhan I, Thilak P, et al. Short-term efficacy and safety of topical β-blockers (timolol maleate ophthalmic solution, 0.5%) in acute migraine: a randomized crossover trial. JAMA Ophthalmol. 2020;138:1160-1166. doi: 10.1001/jamaophthalmol.2020.3676
- Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386:743-800. doi: 10.1016/S0140-6736(15)60692-4
- Yeh WZ, Blizzard L, Taylor BV. What is the actual prevalence of migraine? Brain Behav. 2018;8:e00950. doi: 10.1002/brb3.950
- Leonardi M, Raggi A. Burden of migraine: international perspectives. Neurol Sci. 2013;34(suppl 1):S117-S118. doi: 10.1007/s10072-013-1387-8
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33:629-808. doi: 10.1177/0333102413485658
- Ubrogepant. GoodRx. Accessed May 23, 2022. www.goodrx.com/ubrogepant
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache. 2016;56:911-940. doi: 10.1111/head.12835
- 8. Migliazzo CV, Hagan JC III. Beta blocker eye drops for treatment of acute migraine. Mo Med. 2014;111:283-288.
- 9. Cossack M, Nabrinsky E, Turner H, et al. Timolol eyedrops in the treatment of acute migraine attacks: a randomized crossover study. JAMA Neurol. 2018;75:1024-1025. doi: 10.1001/jamaneurol.2018.0970
- 10. Timolol. GoodRx. Accessed May 23, 2022. www.goodrx.com/timolol
ILLUSTRATIVE CASE
A 35-year-old woman with no significant past medical history presents for follow-up of migraine. At the previous visit, she was prescribed sumatriptan for abortive therapy. However, she has been having significant adverse effect intolerance from the oral formulation, and the nasal formulation is cost prohibitive. What can you recommend as an alternative abortive therapy for this patient’s migraine?
Migraine is among the most common causes of disability worldwide, affecting more than 10% of the global population.2 The prevalence of migraine is between 2.6% and 21.7% across multiple countries.3 On a scale of 0% to 100%, disability caused by migraine is 43.3%, comparable to the first 2 days after an acute myocardial infarction (42.2%) and severe dementia (43.8%).4
Abortive therapy for acute migraine includes nonsteroidal anti-inflammatory drugs (NSAIDs),
Nausea and vomiting, common components of migraine (that are included in International Classification of Headache Disorders, 3rd edition [ICHD-3] criteria for migraine5) present obstacles to effective oral administration if experienced by the patient. In addition, for migraine refractory to first-line treatments, abortive options—including the recently approved
Two oral beta-blockers, propranolol and timolol, are approved by the US Food and Drug Administration for migraine prophylaxis. Unfortunately, oral beta-blockers are ineffective for abortive treatment.7 Ophthalmic timolol is typically used in the treatment of glaucoma, but there have been case reports describing its benefits in acute migraine treatment.8,9 In addition, ophthalmic timolol is far cheaper than medications such as ubrogepant.10 A 2014 case series of 7 patients discussed ophthalmic beta-blockers as an effective and possibly cheaper option for acute migraine treatment.8 A randomized, crossover, placebo-controlled pilot study of 198 migraine attacks in 10 participants using timolol eyedrops for abortive therapy found timolol was not significantly more effective than placebo.9 However, it was an underpowered pilot study, with a lack of masking and an imperfect placebo. The trial discussed here was a controlled, prospective study investigating topical beta-blockers for acute migraine treatment.
STUDY SUMMARY
Crossover study achieved primary endpoint in pain reduction
This randomized, single-center, double-masked, crossover trial compared timolol maleate ophthalmic solution 0.5% with placebo among 43 patients ages 12 or older presenting with a diagnosis of migraine based on ICHD-3 (beta) criteria. Patients were eligible if they had not taken any antimigraine medications for at least 1 month prior to the study and were excluded if they had taken systemic beta-blockers at baseline, or had asthma, bradyarrhythmias, or cardiac dysfunction.
Patients were randomized 1:1 to treatment with timolol maleate 0.5% eyedrops or placebo. At the earliest onset of migraine, patients used 1 drop of timolol maleate 0.5% or placebo in each eye; if they experienced no relief after 10 minutes, they used a second drop or matching placebo. Patients were instructed to score their headache pain on a 10-point scale prior to using the eyedrops and then again 20 minutes after treatment. If a patient had migraine with aura, they were asked to use the eyedrops at the onset of the aura but measure their score at headache onset. If no headaches developed within 20 minutes of the aura, the episode was not included for analysis. All patients were permitted to use their standard oral rescue medication if no relief occurred after 20 minutes of pain onset.
Continue to: The groups were observed...
The groups were observed for 3 months and then followed for a 1-month washout period, during which they received no study medications. The groups were then crossed over to the other treatment and were observed for another 3 months. The primary outcome was a reduction in pain score by 4 or more points, or to 0 on a 10-point pain scale, 20 minutes after treatment. The secondary outcome was nonuse of oral rescue medication.
Forty-three patients were included in a modified intention-to-treat analysis. The primary outcome was achieved in 233 of 284 (82%) timolol-treated migraines, compared to 38 of 271 (14%) placebo-treated migraines (percentage difference = 68 percentage points; 95% CI, 62-74 percentage points; P < .001). The mean pain score at the onset of migraine attacks was 6.01 for those treated with timolol and 5.93 for those treated with placebo. Patients treated with timolol had a reduction in pain of 5.98 points, compared with 0.93 points after using placebo (difference = 5.05; 95% CI, 4.19-5.91). No attacks included in the data required oral rescue medications, and there were no systemic adverse effects from the timolol eyedrops.
WHAT’S NEW
Evidence of benefit as abortive therapy for acute migraine
This randomized controlled trial (RCT) showed evidence to support timolol maleate ophthalmic solution 0.5% vs placebo for treatment of acute migraine by significantly reducing pain when taken at the onset of an acute migraine attack.
CAVEATS
Single-center trial, measuring limited response time
The generalizability of this RCT is limited because it was a single-center trial with a study population from a single region in India. It is unknown whether pain relief, adverse effects, or adherence would differ for the global population. Additionally, only migraines with headache were included in the analysis, limiting non-headache migraine subgroup-directed treatment. Also, this trial evaluated only the response to treatment at 20 minutes, and it is unknown if pain response continued for several hours. Headaches that began more than 20 minutes after the onset of aura were not evaluated.
CHALLENGES TO IMPLEMENTATION
Timolol’s systemic adverse effects require caution
Systemic beta-blocker effects (eg, bradycardia, hypotension, drowsiness, and bronchospasm) from topical timolol have been reported. Caution should be used when prescribing timolol for patients with current cardiovascular and pulmonary conditions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
A 35-year-old woman with no significant past medical history presents for follow-up of migraine. At the previous visit, she was prescribed sumatriptan for abortive therapy. However, she has been having significant adverse effect intolerance from the oral formulation, and the nasal formulation is cost prohibitive. What can you recommend as an alternative abortive therapy for this patient’s migraine?
Migraine is among the most common causes of disability worldwide, affecting more than 10% of the global population.2 The prevalence of migraine is between 2.6% and 21.7% across multiple countries.3 On a scale of 0% to 100%, disability caused by migraine is 43.3%, comparable to the first 2 days after an acute myocardial infarction (42.2%) and severe dementia (43.8%).4
Abortive therapy for acute migraine includes nonsteroidal anti-inflammatory drugs (NSAIDs),
Nausea and vomiting, common components of migraine (that are included in International Classification of Headache Disorders, 3rd edition [ICHD-3] criteria for migraine5) present obstacles to effective oral administration if experienced by the patient. In addition, for migraine refractory to first-line treatments, abortive options—including the recently approved
Two oral beta-blockers, propranolol and timolol, are approved by the US Food and Drug Administration for migraine prophylaxis. Unfortunately, oral beta-blockers are ineffective for abortive treatment.7 Ophthalmic timolol is typically used in the treatment of glaucoma, but there have been case reports describing its benefits in acute migraine treatment.8,9 In addition, ophthalmic timolol is far cheaper than medications such as ubrogepant.10 A 2014 case series of 7 patients discussed ophthalmic beta-blockers as an effective and possibly cheaper option for acute migraine treatment.8 A randomized, crossover, placebo-controlled pilot study of 198 migraine attacks in 10 participants using timolol eyedrops for abortive therapy found timolol was not significantly more effective than placebo.9 However, it was an underpowered pilot study, with a lack of masking and an imperfect placebo. The trial discussed here was a controlled, prospective study investigating topical beta-blockers for acute migraine treatment.
STUDY SUMMARY
Crossover study achieved primary endpoint in pain reduction
This randomized, single-center, double-masked, crossover trial compared timolol maleate ophthalmic solution 0.5% with placebo among 43 patients ages 12 or older presenting with a diagnosis of migraine based on ICHD-3 (beta) criteria. Patients were eligible if they had not taken any antimigraine medications for at least 1 month prior to the study and were excluded if they had taken systemic beta-blockers at baseline, or had asthma, bradyarrhythmias, or cardiac dysfunction.
Patients were randomized 1:1 to treatment with timolol maleate 0.5% eyedrops or placebo. At the earliest onset of migraine, patients used 1 drop of timolol maleate 0.5% or placebo in each eye; if they experienced no relief after 10 minutes, they used a second drop or matching placebo. Patients were instructed to score their headache pain on a 10-point scale prior to using the eyedrops and then again 20 minutes after treatment. If a patient had migraine with aura, they were asked to use the eyedrops at the onset of the aura but measure their score at headache onset. If no headaches developed within 20 minutes of the aura, the episode was not included for analysis. All patients were permitted to use their standard oral rescue medication if no relief occurred after 20 minutes of pain onset.
Continue to: The groups were observed...
The groups were observed for 3 months and then followed for a 1-month washout period, during which they received no study medications. The groups were then crossed over to the other treatment and were observed for another 3 months. The primary outcome was a reduction in pain score by 4 or more points, or to 0 on a 10-point pain scale, 20 minutes after treatment. The secondary outcome was nonuse of oral rescue medication.
Forty-three patients were included in a modified intention-to-treat analysis. The primary outcome was achieved in 233 of 284 (82%) timolol-treated migraines, compared to 38 of 271 (14%) placebo-treated migraines (percentage difference = 68 percentage points; 95% CI, 62-74 percentage points; P < .001). The mean pain score at the onset of migraine attacks was 6.01 for those treated with timolol and 5.93 for those treated with placebo. Patients treated with timolol had a reduction in pain of 5.98 points, compared with 0.93 points after using placebo (difference = 5.05; 95% CI, 4.19-5.91). No attacks included in the data required oral rescue medications, and there were no systemic adverse effects from the timolol eyedrops.
WHAT’S NEW
Evidence of benefit as abortive therapy for acute migraine
This randomized controlled trial (RCT) showed evidence to support timolol maleate ophthalmic solution 0.5% vs placebo for treatment of acute migraine by significantly reducing pain when taken at the onset of an acute migraine attack.
CAVEATS
Single-center trial, measuring limited response time
The generalizability of this RCT is limited because it was a single-center trial with a study population from a single region in India. It is unknown whether pain relief, adverse effects, or adherence would differ for the global population. Additionally, only migraines with headache were included in the analysis, limiting non-headache migraine subgroup-directed treatment. Also, this trial evaluated only the response to treatment at 20 minutes, and it is unknown if pain response continued for several hours. Headaches that began more than 20 minutes after the onset of aura were not evaluated.
CHALLENGES TO IMPLEMENTATION
Timolol’s systemic adverse effects require caution
Systemic beta-blocker effects (eg, bradycardia, hypotension, drowsiness, and bronchospasm) from topical timolol have been reported. Caution should be used when prescribing timolol for patients with current cardiovascular and pulmonary conditions.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
- Kurian A, Reghunadhan I, Thilak P, et al. Short-term efficacy and safety of topical β-blockers (timolol maleate ophthalmic solution, 0.5%) in acute migraine: a randomized crossover trial. JAMA Ophthalmol. 2020;138:1160-1166. doi: 10.1001/jamaophthalmol.2020.3676
- Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386:743-800. doi: 10.1016/S0140-6736(15)60692-4
- Yeh WZ, Blizzard L, Taylor BV. What is the actual prevalence of migraine? Brain Behav. 2018;8:e00950. doi: 10.1002/brb3.950
- Leonardi M, Raggi A. Burden of migraine: international perspectives. Neurol Sci. 2013;34(suppl 1):S117-S118. doi: 10.1007/s10072-013-1387-8
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33:629-808. doi: 10.1177/0333102413485658
- Ubrogepant. GoodRx. Accessed May 23, 2022. www.goodrx.com/ubrogepant
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache. 2016;56:911-940. doi: 10.1111/head.12835
- 8. Migliazzo CV, Hagan JC III. Beta blocker eye drops for treatment of acute migraine. Mo Med. 2014;111:283-288.
- 9. Cossack M, Nabrinsky E, Turner H, et al. Timolol eyedrops in the treatment of acute migraine attacks: a randomized crossover study. JAMA Neurol. 2018;75:1024-1025. doi: 10.1001/jamaneurol.2018.0970
- 10. Timolol. GoodRx. Accessed May 23, 2022. www.goodrx.com/timolol
- Kurian A, Reghunadhan I, Thilak P, et al. Short-term efficacy and safety of topical β-blockers (timolol maleate ophthalmic solution, 0.5%) in acute migraine: a randomized crossover trial. JAMA Ophthalmol. 2020;138:1160-1166. doi: 10.1001/jamaophthalmol.2020.3676
- Global Burden of Disease Study 2013 Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 301 acute and chronic diseases and injuries in 188 countries, 1990-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2015;386:743-800. doi: 10.1016/S0140-6736(15)60692-4
- Yeh WZ, Blizzard L, Taylor BV. What is the actual prevalence of migraine? Brain Behav. 2018;8:e00950. doi: 10.1002/brb3.950
- Leonardi M, Raggi A. Burden of migraine: international perspectives. Neurol Sci. 2013;34(suppl 1):S117-S118. doi: 10.1007/s10072-013-1387-8
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia. 2013;33:629-808. doi: 10.1177/0333102413485658
- Ubrogepant. GoodRx. Accessed May 23, 2022. www.goodrx.com/ubrogepant
- Orr SL, Friedman BW, Christie S, et al. Management of adults with acute migraine in the emergency department: the American Headache Society evidence assessment of parenteral pharmacotherapies. Headache. 2016;56:911-940. doi: 10.1111/head.12835
- 8. Migliazzo CV, Hagan JC III. Beta blocker eye drops for treatment of acute migraine. Mo Med. 2014;111:283-288.
- 9. Cossack M, Nabrinsky E, Turner H, et al. Timolol eyedrops in the treatment of acute migraine attacks: a randomized crossover study. JAMA Neurol. 2018;75:1024-1025. doi: 10.1001/jamaneurol.2018.0970
- 10. Timolol. GoodRx. Accessed May 23, 2022. www.goodrx.com/timolol
PRACTICE CHANGER
Consider timolol maleate 0.5% eyedrops as a quick and effective abortive therapy for migraine.1
STRENGTH OF RECOMMENDATION
B: Based on a single randomized controlled trial.1
Kurian A, Reghunadhan I, Thilak P, et al. Short-term efficacy and safety of topical β-blockers (timolol maleate ophthalmic solution, 0.5%) in acute migraine: a randomized crossover trial. JAMA Ophthalmol. 2020;138:1160-1166.
In utero COVID exposure tied to neurodevelopmental disorders at 1 year
Infants exposed to SARS-CoV-2 in utero are at increased risk for neurodevelopmental disorders in the first year of life, new research suggests.
But whether it is exposure to the pandemic or maternal exposure to the virus itself that may harm early childhood neurodevelopment is unclear, caution investigators, led by Roy Perlis, MD, MSc, with Massachusetts General Hospital, Boston.
“In this analysis of 222 offspring of mothers infected with SARS-CoV-2, compared with the offspring of 7,550 mothers in the control group (not infected) delivered during the same period, we observed neurodevelopmental diagnoses to be significantly more common among exposed offspring, particularly those exposed to third-trimester maternal infection,” they write.
The study was published online in JAMA Network Open.
Speech and language disorders
The study included 7,772 mostly singleton live births across six hospitals in Massachusetts between March and September 2020, including 222 (2.9%) births to mothers with SARS-CoV-2 infection confirmed by polymerase chain reaction testing during pregnancy.
In all, 14 of 222 children born to SARS-CoV-2–infected mothers (6.3%) were diagnosed with a neurodevelopmental disorder in the first year of life versus 227 of 7,550 unexposed offspring (3%) (unadjusted odds ratio, 2.17; 95% confidence interval, 1.24-3.79; P = .006).
In models adjusted for preterm delivery, as well as race, ethnicity, insurance status, child sex, and maternal age, COVID-exposed offspring were significantly more likely to receive a neurodevelopmental diagnosis in the first year of life (adjusted OR, 1.86; 95% CI, 1.03-3.36; P = .04).
The magnitude of the association with neurodevelopmental disorders was greater with third-trimester SARS-CoV-2 infection (aOR, 2.34; 95% CI, 1.23-4.44; P = .01).
The majority of these diagnoses reflected developmental disorders of motor function or speech and language.
The researchers noted that the finding of an association between prenatal SARS-CoV-2 exposure and neurodevelopmental diagnoses at 12 months is in line with a “large body of literature” linking maternal viral infection and maternal immune activation with offspring neurodevelopmental disorders later in life.
They cautioned, however, that whether a definitive connection exists between prenatal SARS-CoV-2 exposure and adverse neurodevelopment in offspring is not yet known, in part because children born to women infected in the first wave of the pandemic haven’t reached their second birthday – a time when neurodevelopment disorders such as autism are typically diagnosed.
There is also the risk for ascertainment bias arising from greater concern for offspring of infected mothers who were ill during pregnancy. These parents may be more inclined to seek evaluation, and clinicians may be more inclined to diagnose or refer for evaluation, the researchers noted.
Nonetheless, as reported by this news organization, the study results support those of research released at the European Psychiatric Association 2022 Congress; those results also showed an association between maternal SARS-CoV-2 infection and impaired neurodevelopment in 6-week-old infants.
Hypothesis generating
In an accompanying commentary, Torri D. Metz, MD, MS, with University of Utah Health, Salt Lake City, said the preliminary findings of Dr. Perlis and colleagues are “critically important, yet many questions remain.”
“Essentially all of what we know now about the effects of in utero exposure to maternal SARS-CoV-2 infection is from children who were exposed to the early and Alpha variants of SARS-CoV-2, as those are the only children now old enough to undergo rigorous neurodevelopmental assessments,” Dr. Metz pointed out.
Ultimately, Dr. Metz said it’s not surprising that the pandemic and in utero exposure to maternal SARS-CoV-2 infection may adversely affect neurodevelopmental outcomes in young children.
Yet, as a retrospective cohort study, the study can only demonstrate associations, not causality.
“This type of work is intended to be hypothesis generating, and that goal has been accomplished as these preliminary findings generate numerous additional research questions to explore,” Dr. Metz wrote.
Among them: Are there genetic predispositions to adverse outcomes? Will we observe differential effects by SARS-CoV-2 variant, by severity of infection, and by trimester of infection? Is it the virus itself or all of the societal changes that occurred during this period, including differences in how those changes were experienced among those with and without SARS-CoV-2?
“Perhaps the most important question is how do we intervene to help mitigate the adverse effects of the pandemic on young children,” Dr. Metz noted.
“Prospective studies to validate these findings, tease out some of the nuance, and identify those at highest risk will help health care practitioners appropriately dedicate resources to improve outcomes as we follow the life course of this generation of children born during the COVID-19 pandemic,” she added.
The study was supported by the National Institute of Mental Health and the National Institute of Child Health and Human Development. Dr. Perlis is an associate editor for JAMA Network Open but was not involved in the editorial review or decision for the study. Dr. Metz reported receiving personal fees and grants from Pfizer and grants from GestVision.
A version of this article first appeared on Medscape.com.
Infants exposed to SARS-CoV-2 in utero are at increased risk for neurodevelopmental disorders in the first year of life, new research suggests.
But whether it is exposure to the pandemic or maternal exposure to the virus itself that may harm early childhood neurodevelopment is unclear, caution investigators, led by Roy Perlis, MD, MSc, with Massachusetts General Hospital, Boston.
“In this analysis of 222 offspring of mothers infected with SARS-CoV-2, compared with the offspring of 7,550 mothers in the control group (not infected) delivered during the same period, we observed neurodevelopmental diagnoses to be significantly more common among exposed offspring, particularly those exposed to third-trimester maternal infection,” they write.
The study was published online in JAMA Network Open.
Speech and language disorders
The study included 7,772 mostly singleton live births across six hospitals in Massachusetts between March and September 2020, including 222 (2.9%) births to mothers with SARS-CoV-2 infection confirmed by polymerase chain reaction testing during pregnancy.
In all, 14 of 222 children born to SARS-CoV-2–infected mothers (6.3%) were diagnosed with a neurodevelopmental disorder in the first year of life versus 227 of 7,550 unexposed offspring (3%) (unadjusted odds ratio, 2.17; 95% confidence interval, 1.24-3.79; P = .006).
In models adjusted for preterm delivery, as well as race, ethnicity, insurance status, child sex, and maternal age, COVID-exposed offspring were significantly more likely to receive a neurodevelopmental diagnosis in the first year of life (adjusted OR, 1.86; 95% CI, 1.03-3.36; P = .04).
The magnitude of the association with neurodevelopmental disorders was greater with third-trimester SARS-CoV-2 infection (aOR, 2.34; 95% CI, 1.23-4.44; P = .01).
The majority of these diagnoses reflected developmental disorders of motor function or speech and language.
The researchers noted that the finding of an association between prenatal SARS-CoV-2 exposure and neurodevelopmental diagnoses at 12 months is in line with a “large body of literature” linking maternal viral infection and maternal immune activation with offspring neurodevelopmental disorders later in life.
They cautioned, however, that whether a definitive connection exists between prenatal SARS-CoV-2 exposure and adverse neurodevelopment in offspring is not yet known, in part because children born to women infected in the first wave of the pandemic haven’t reached their second birthday – a time when neurodevelopment disorders such as autism are typically diagnosed.
There is also the risk for ascertainment bias arising from greater concern for offspring of infected mothers who were ill during pregnancy. These parents may be more inclined to seek evaluation, and clinicians may be more inclined to diagnose or refer for evaluation, the researchers noted.
Nonetheless, as reported by this news organization, the study results support those of research released at the European Psychiatric Association 2022 Congress; those results also showed an association between maternal SARS-CoV-2 infection and impaired neurodevelopment in 6-week-old infants.
Hypothesis generating
In an accompanying commentary, Torri D. Metz, MD, MS, with University of Utah Health, Salt Lake City, said the preliminary findings of Dr. Perlis and colleagues are “critically important, yet many questions remain.”
“Essentially all of what we know now about the effects of in utero exposure to maternal SARS-CoV-2 infection is from children who were exposed to the early and Alpha variants of SARS-CoV-2, as those are the only children now old enough to undergo rigorous neurodevelopmental assessments,” Dr. Metz pointed out.
Ultimately, Dr. Metz said it’s not surprising that the pandemic and in utero exposure to maternal SARS-CoV-2 infection may adversely affect neurodevelopmental outcomes in young children.
Yet, as a retrospective cohort study, the study can only demonstrate associations, not causality.
“This type of work is intended to be hypothesis generating, and that goal has been accomplished as these preliminary findings generate numerous additional research questions to explore,” Dr. Metz wrote.
Among them: Are there genetic predispositions to adverse outcomes? Will we observe differential effects by SARS-CoV-2 variant, by severity of infection, and by trimester of infection? Is it the virus itself or all of the societal changes that occurred during this period, including differences in how those changes were experienced among those with and without SARS-CoV-2?
“Perhaps the most important question is how do we intervene to help mitigate the adverse effects of the pandemic on young children,” Dr. Metz noted.
“Prospective studies to validate these findings, tease out some of the nuance, and identify those at highest risk will help health care practitioners appropriately dedicate resources to improve outcomes as we follow the life course of this generation of children born during the COVID-19 pandemic,” she added.
The study was supported by the National Institute of Mental Health and the National Institute of Child Health and Human Development. Dr. Perlis is an associate editor for JAMA Network Open but was not involved in the editorial review or decision for the study. Dr. Metz reported receiving personal fees and grants from Pfizer and grants from GestVision.
A version of this article first appeared on Medscape.com.
Infants exposed to SARS-CoV-2 in utero are at increased risk for neurodevelopmental disorders in the first year of life, new research suggests.
But whether it is exposure to the pandemic or maternal exposure to the virus itself that may harm early childhood neurodevelopment is unclear, caution investigators, led by Roy Perlis, MD, MSc, with Massachusetts General Hospital, Boston.
“In this analysis of 222 offspring of mothers infected with SARS-CoV-2, compared with the offspring of 7,550 mothers in the control group (not infected) delivered during the same period, we observed neurodevelopmental diagnoses to be significantly more common among exposed offspring, particularly those exposed to third-trimester maternal infection,” they write.
The study was published online in JAMA Network Open.
Speech and language disorders
The study included 7,772 mostly singleton live births across six hospitals in Massachusetts between March and September 2020, including 222 (2.9%) births to mothers with SARS-CoV-2 infection confirmed by polymerase chain reaction testing during pregnancy.
In all, 14 of 222 children born to SARS-CoV-2–infected mothers (6.3%) were diagnosed with a neurodevelopmental disorder in the first year of life versus 227 of 7,550 unexposed offspring (3%) (unadjusted odds ratio, 2.17; 95% confidence interval, 1.24-3.79; P = .006).
In models adjusted for preterm delivery, as well as race, ethnicity, insurance status, child sex, and maternal age, COVID-exposed offspring were significantly more likely to receive a neurodevelopmental diagnosis in the first year of life (adjusted OR, 1.86; 95% CI, 1.03-3.36; P = .04).
The magnitude of the association with neurodevelopmental disorders was greater with third-trimester SARS-CoV-2 infection (aOR, 2.34; 95% CI, 1.23-4.44; P = .01).
The majority of these diagnoses reflected developmental disorders of motor function or speech and language.
The researchers noted that the finding of an association between prenatal SARS-CoV-2 exposure and neurodevelopmental diagnoses at 12 months is in line with a “large body of literature” linking maternal viral infection and maternal immune activation with offspring neurodevelopmental disorders later in life.
They cautioned, however, that whether a definitive connection exists between prenatal SARS-CoV-2 exposure and adverse neurodevelopment in offspring is not yet known, in part because children born to women infected in the first wave of the pandemic haven’t reached their second birthday – a time when neurodevelopment disorders such as autism are typically diagnosed.
There is also the risk for ascertainment bias arising from greater concern for offspring of infected mothers who were ill during pregnancy. These parents may be more inclined to seek evaluation, and clinicians may be more inclined to diagnose or refer for evaluation, the researchers noted.
Nonetheless, as reported by this news organization, the study results support those of research released at the European Psychiatric Association 2022 Congress; those results also showed an association between maternal SARS-CoV-2 infection and impaired neurodevelopment in 6-week-old infants.
Hypothesis generating
In an accompanying commentary, Torri D. Metz, MD, MS, with University of Utah Health, Salt Lake City, said the preliminary findings of Dr. Perlis and colleagues are “critically important, yet many questions remain.”
“Essentially all of what we know now about the effects of in utero exposure to maternal SARS-CoV-2 infection is from children who were exposed to the early and Alpha variants of SARS-CoV-2, as those are the only children now old enough to undergo rigorous neurodevelopmental assessments,” Dr. Metz pointed out.
Ultimately, Dr. Metz said it’s not surprising that the pandemic and in utero exposure to maternal SARS-CoV-2 infection may adversely affect neurodevelopmental outcomes in young children.
Yet, as a retrospective cohort study, the study can only demonstrate associations, not causality.
“This type of work is intended to be hypothesis generating, and that goal has been accomplished as these preliminary findings generate numerous additional research questions to explore,” Dr. Metz wrote.
Among them: Are there genetic predispositions to adverse outcomes? Will we observe differential effects by SARS-CoV-2 variant, by severity of infection, and by trimester of infection? Is it the virus itself or all of the societal changes that occurred during this period, including differences in how those changes were experienced among those with and without SARS-CoV-2?
“Perhaps the most important question is how do we intervene to help mitigate the adverse effects of the pandemic on young children,” Dr. Metz noted.
“Prospective studies to validate these findings, tease out some of the nuance, and identify those at highest risk will help health care practitioners appropriately dedicate resources to improve outcomes as we follow the life course of this generation of children born during the COVID-19 pandemic,” she added.
The study was supported by the National Institute of Mental Health and the National Institute of Child Health and Human Development. Dr. Perlis is an associate editor for JAMA Network Open but was not involved in the editorial review or decision for the study. Dr. Metz reported receiving personal fees and grants from Pfizer and grants from GestVision.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Stimulants may not improve academic learning in children with ADHD
Extended-release methylphenidate (Concerta) had no effect on learning academic material taught in a small group of children with attention-deficit/hyperactivity disorder (ADHD), a controlled crossover study found.
As in previous studies, however, the stimulant did improve seat work productivity and classroom behavior, but these benefits did not translate into better learning of individual academic learning units, according to William E. Pelham Jr., PhD, of the department of psychology at Florida International University in Miami, and colleagues.
The results were published online in the Journal of Consulting and Clinical Psychology.
The authors said the finding raises questions about how stimulant medication leads to improved academic achievement over time. “This is important given that many parents and pediatricians believe that medication will improve academic achievement; parents are more likely to pursue medication (vs. other treatment options) when they identify academic achievement as a primary goal for treatment. The current findings suggest this emphasis may be misguided,” they wrote.
In their view, efforts to improve learning in children with ADHD should focus on delivering effective academic instruction and support such as individualized educational plans rather than stimulant therapy.
The study
The study cohort consisted of 173 children aged 7-12 (77% male, 86% Hispanic) who met Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, criteria for ADHD and were participating in a therapeutic summer camp classroom.
The experimental design was a triple-masked, within-subject, AB/BA crossover trial. Children completed two consecutive phases of daily, 25-minute instruction in both subject-area content (science and social studies) and vocabulary. Each phase was a standard instructional unit lasting for 3 weeks and lessons were given by credentialed teachers via small-group, evidence-based instruction.
Each child was randomized to receive daily osmotic-release oral system methylphenidate (OROS-MPH) during either the first or second instructional phase and to receive placebo during the other.
Seat work referred to the amount of work a pupil completed in a fixed duration of independent work time, and classroom behavior referred to the frequency of violating classroom rules. Learning was measured by tests, and multilevel models were fit separately to the subject and vocabulary test scores, with four observations per child: pretest and posttest in the two academic subject areas.
The results showed that medication had large, salutary, statistically significant effects on children’s academic seat work productivity and classroom behavior on every single day of the instructional period.
Pupils completed 37% more arithmetic problems per minute when taking OROS-MPH and committed 53% fewer rule violations per hour. In terms of learning the material taught during instruction, however, tests showed that children learned the same amount of subject-area and vocabulary content whether they were taking OROS-MPH or placebo during the instructional period.
Consistent with previous studies, medication slightly helped to improve test scores when taken on the day of a test, but not enough to boost most children’s grades. For example, medication helped children increase on average 1.7 percentage points out of 100 on science and social studies tests.
“This finding has relevance for parents deciding whether to medicate their child for occasions such as a psychoeducational evaluation or high-stakes academic testing – while the effect size was small, findings suggest being medicated would improve scores,” the investigators wrote.
Sharing his perspective on the study but not involved in it, Herschel R. Lessin, MD, a pediatrician at The Children’s Medical Group in Poughkeepsie, N.Y., and coauthor of the American Academy of Pediatrics (AAP) guidelines on ADHD, said, “If you ignore the sensationalized headlines, this study is an interesting but preliminary first step, and justifies further research on the topic. It also has several potential defects, which the authors in fact address in the supplements.” The cohort size was small, for example, the doses of medication were very low, and the study took place in a controlled therapeutic setting – not the everyday classroom.
Furthermore, Dr. Lessin added, the study’s conclusions “are contrary to my 40 years of experience in treating ADHD. If they had used standard measures of assessment, as in previous studies, they would have found medication did impact learning. More research is clearly needed.”
In other comments, Holly K. Harris, MD, assistant professor of pediatrics-development at Baylor College of Medicine and Texas Children’s Hospital in Houston, said the core symptoms of ADHD are primarily behavioral in nature, not academic learning related.
“Stimulant medications are targeting these core behavioral symptoms of ADHD ... but the goal of treatment is more than just the reduction of symptoms; it is to improve a child’s overall functioning so that they succeed at what is expected of them and avoid developing even more impairments,” Dr. Harris said, adding that symptom improvement can sometimes allow a child to learn better in the classroom and achieve more academically.
Children with ADHD may have diagnosed or undiagnosed comorbid learning disabilities, with one 2013 study suggesting a rate of 31%-45%.
With such learning disabilities being distinct from core behavioral symptoms, stimulant medications would not be expected to address a child’s learning disability. “In fact, best practice is for a child with ADHD who is not responding to stimulant medication (doctors might refer to this as complex ADHD) to undergo full individual evaluations either through the school system or an outside psychological assessment to assess for potential learning disabilities or other comorbid developmental/learning or psychiatric diagnosis,” Dr. Harris said.
Rather than changing prescribing patterns, she continued, pediatricians could consider advising parents to request learning evaluations through the school system if the child continues to struggle academically with no change in learning outcomes despite improvement in some behavioral outcomes.
As a reference, Dr. Harris recommended the Society for Developmental and Behavioral Pediatrics guidelines for complex ADHD.
This study was funded by the National Institute on Mental Health with additional support from the National Institute on Alcohol Abuse and Alcoholism, the National Institute on Drug Abuse, and the Institute of Education Sciences. Coauthor James Waxmonsky, MD, has received research funding from the National Institutes of Health, Supernus, and Pfizer and served on the advisory board for Iron Shore, NLS Pharma, and Purdue Pharma.
Extended-release methylphenidate (Concerta) had no effect on learning academic material taught in a small group of children with attention-deficit/hyperactivity disorder (ADHD), a controlled crossover study found.
As in previous studies, however, the stimulant did improve seat work productivity and classroom behavior, but these benefits did not translate into better learning of individual academic learning units, according to William E. Pelham Jr., PhD, of the department of psychology at Florida International University in Miami, and colleagues.
The results were published online in the Journal of Consulting and Clinical Psychology.
The authors said the finding raises questions about how stimulant medication leads to improved academic achievement over time. “This is important given that many parents and pediatricians believe that medication will improve academic achievement; parents are more likely to pursue medication (vs. other treatment options) when they identify academic achievement as a primary goal for treatment. The current findings suggest this emphasis may be misguided,” they wrote.
In their view, efforts to improve learning in children with ADHD should focus on delivering effective academic instruction and support such as individualized educational plans rather than stimulant therapy.
The study
The study cohort consisted of 173 children aged 7-12 (77% male, 86% Hispanic) who met Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, criteria for ADHD and were participating in a therapeutic summer camp classroom.
The experimental design was a triple-masked, within-subject, AB/BA crossover trial. Children completed two consecutive phases of daily, 25-minute instruction in both subject-area content (science and social studies) and vocabulary. Each phase was a standard instructional unit lasting for 3 weeks and lessons were given by credentialed teachers via small-group, evidence-based instruction.
Each child was randomized to receive daily osmotic-release oral system methylphenidate (OROS-MPH) during either the first or second instructional phase and to receive placebo during the other.
Seat work referred to the amount of work a pupil completed in a fixed duration of independent work time, and classroom behavior referred to the frequency of violating classroom rules. Learning was measured by tests, and multilevel models were fit separately to the subject and vocabulary test scores, with four observations per child: pretest and posttest in the two academic subject areas.
The results showed that medication had large, salutary, statistically significant effects on children’s academic seat work productivity and classroom behavior on every single day of the instructional period.
Pupils completed 37% more arithmetic problems per minute when taking OROS-MPH and committed 53% fewer rule violations per hour. In terms of learning the material taught during instruction, however, tests showed that children learned the same amount of subject-area and vocabulary content whether they were taking OROS-MPH or placebo during the instructional period.
Consistent with previous studies, medication slightly helped to improve test scores when taken on the day of a test, but not enough to boost most children’s grades. For example, medication helped children increase on average 1.7 percentage points out of 100 on science and social studies tests.
“This finding has relevance for parents deciding whether to medicate their child for occasions such as a psychoeducational evaluation or high-stakes academic testing – while the effect size was small, findings suggest being medicated would improve scores,” the investigators wrote.
Sharing his perspective on the study but not involved in it, Herschel R. Lessin, MD, a pediatrician at The Children’s Medical Group in Poughkeepsie, N.Y., and coauthor of the American Academy of Pediatrics (AAP) guidelines on ADHD, said, “If you ignore the sensationalized headlines, this study is an interesting but preliminary first step, and justifies further research on the topic. It also has several potential defects, which the authors in fact address in the supplements.” The cohort size was small, for example, the doses of medication were very low, and the study took place in a controlled therapeutic setting – not the everyday classroom.
Furthermore, Dr. Lessin added, the study’s conclusions “are contrary to my 40 years of experience in treating ADHD. If they had used standard measures of assessment, as in previous studies, they would have found medication did impact learning. More research is clearly needed.”
In other comments, Holly K. Harris, MD, assistant professor of pediatrics-development at Baylor College of Medicine and Texas Children’s Hospital in Houston, said the core symptoms of ADHD are primarily behavioral in nature, not academic learning related.
“Stimulant medications are targeting these core behavioral symptoms of ADHD ... but the goal of treatment is more than just the reduction of symptoms; it is to improve a child’s overall functioning so that they succeed at what is expected of them and avoid developing even more impairments,” Dr. Harris said, adding that symptom improvement can sometimes allow a child to learn better in the classroom and achieve more academically.
Children with ADHD may have diagnosed or undiagnosed comorbid learning disabilities, with one 2013 study suggesting a rate of 31%-45%.
With such learning disabilities being distinct from core behavioral symptoms, stimulant medications would not be expected to address a child’s learning disability. “In fact, best practice is for a child with ADHD who is not responding to stimulant medication (doctors might refer to this as complex ADHD) to undergo full individual evaluations either through the school system or an outside psychological assessment to assess for potential learning disabilities or other comorbid developmental/learning or psychiatric diagnosis,” Dr. Harris said.
Rather than changing prescribing patterns, she continued, pediatricians could consider advising parents to request learning evaluations through the school system if the child continues to struggle academically with no change in learning outcomes despite improvement in some behavioral outcomes.
As a reference, Dr. Harris recommended the Society for Developmental and Behavioral Pediatrics guidelines for complex ADHD.
This study was funded by the National Institute on Mental Health with additional support from the National Institute on Alcohol Abuse and Alcoholism, the National Institute on Drug Abuse, and the Institute of Education Sciences. Coauthor James Waxmonsky, MD, has received research funding from the National Institutes of Health, Supernus, and Pfizer and served on the advisory board for Iron Shore, NLS Pharma, and Purdue Pharma.
Extended-release methylphenidate (Concerta) had no effect on learning academic material taught in a small group of children with attention-deficit/hyperactivity disorder (ADHD), a controlled crossover study found.
As in previous studies, however, the stimulant did improve seat work productivity and classroom behavior, but these benefits did not translate into better learning of individual academic learning units, according to William E. Pelham Jr., PhD, of the department of psychology at Florida International University in Miami, and colleagues.
The results were published online in the Journal of Consulting and Clinical Psychology.
The authors said the finding raises questions about how stimulant medication leads to improved academic achievement over time. “This is important given that many parents and pediatricians believe that medication will improve academic achievement; parents are more likely to pursue medication (vs. other treatment options) when they identify academic achievement as a primary goal for treatment. The current findings suggest this emphasis may be misguided,” they wrote.
In their view, efforts to improve learning in children with ADHD should focus on delivering effective academic instruction and support such as individualized educational plans rather than stimulant therapy.
The study
The study cohort consisted of 173 children aged 7-12 (77% male, 86% Hispanic) who met Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, criteria for ADHD and were participating in a therapeutic summer camp classroom.
The experimental design was a triple-masked, within-subject, AB/BA crossover trial. Children completed two consecutive phases of daily, 25-minute instruction in both subject-area content (science and social studies) and vocabulary. Each phase was a standard instructional unit lasting for 3 weeks and lessons were given by credentialed teachers via small-group, evidence-based instruction.
Each child was randomized to receive daily osmotic-release oral system methylphenidate (OROS-MPH) during either the first or second instructional phase and to receive placebo during the other.
Seat work referred to the amount of work a pupil completed in a fixed duration of independent work time, and classroom behavior referred to the frequency of violating classroom rules. Learning was measured by tests, and multilevel models were fit separately to the subject and vocabulary test scores, with four observations per child: pretest and posttest in the two academic subject areas.
The results showed that medication had large, salutary, statistically significant effects on children’s academic seat work productivity and classroom behavior on every single day of the instructional period.
Pupils completed 37% more arithmetic problems per minute when taking OROS-MPH and committed 53% fewer rule violations per hour. In terms of learning the material taught during instruction, however, tests showed that children learned the same amount of subject-area and vocabulary content whether they were taking OROS-MPH or placebo during the instructional period.
Consistent with previous studies, medication slightly helped to improve test scores when taken on the day of a test, but not enough to boost most children’s grades. For example, medication helped children increase on average 1.7 percentage points out of 100 on science and social studies tests.
“This finding has relevance for parents deciding whether to medicate their child for occasions such as a psychoeducational evaluation or high-stakes academic testing – while the effect size was small, findings suggest being medicated would improve scores,” the investigators wrote.
Sharing his perspective on the study but not involved in it, Herschel R. Lessin, MD, a pediatrician at The Children’s Medical Group in Poughkeepsie, N.Y., and coauthor of the American Academy of Pediatrics (AAP) guidelines on ADHD, said, “If you ignore the sensationalized headlines, this study is an interesting but preliminary first step, and justifies further research on the topic. It also has several potential defects, which the authors in fact address in the supplements.” The cohort size was small, for example, the doses of medication were very low, and the study took place in a controlled therapeutic setting – not the everyday classroom.
Furthermore, Dr. Lessin added, the study’s conclusions “are contrary to my 40 years of experience in treating ADHD. If they had used standard measures of assessment, as in previous studies, they would have found medication did impact learning. More research is clearly needed.”
In other comments, Holly K. Harris, MD, assistant professor of pediatrics-development at Baylor College of Medicine and Texas Children’s Hospital in Houston, said the core symptoms of ADHD are primarily behavioral in nature, not academic learning related.
“Stimulant medications are targeting these core behavioral symptoms of ADHD ... but the goal of treatment is more than just the reduction of symptoms; it is to improve a child’s overall functioning so that they succeed at what is expected of them and avoid developing even more impairments,” Dr. Harris said, adding that symptom improvement can sometimes allow a child to learn better in the classroom and achieve more academically.
Children with ADHD may have diagnosed or undiagnosed comorbid learning disabilities, with one 2013 study suggesting a rate of 31%-45%.
With such learning disabilities being distinct from core behavioral symptoms, stimulant medications would not be expected to address a child’s learning disability. “In fact, best practice is for a child with ADHD who is not responding to stimulant medication (doctors might refer to this as complex ADHD) to undergo full individual evaluations either through the school system or an outside psychological assessment to assess for potential learning disabilities or other comorbid developmental/learning or psychiatric diagnosis,” Dr. Harris said.
Rather than changing prescribing patterns, she continued, pediatricians could consider advising parents to request learning evaluations through the school system if the child continues to struggle academically with no change in learning outcomes despite improvement in some behavioral outcomes.
As a reference, Dr. Harris recommended the Society for Developmental and Behavioral Pediatrics guidelines for complex ADHD.
This study was funded by the National Institute on Mental Health with additional support from the National Institute on Alcohol Abuse and Alcoholism, the National Institute on Drug Abuse, and the Institute of Education Sciences. Coauthor James Waxmonsky, MD, has received research funding from the National Institutes of Health, Supernus, and Pfizer and served on the advisory board for Iron Shore, NLS Pharma, and Purdue Pharma.
FROM JOURNAL OF CONSULTING AND CLINICAL PSYCHOLOGY
ECT may reduce all-cause mortality in major depression
In an analysis of data from a large database of inpatients across the United States, use of ECT for those with resistant MDD was associated with significantly lower in-hospital mortality compared with those who did not receive ECT.
This held true even after the researchers controlled for demographics and loss of function due to comorbid medical conditions.
“I think the risks of ECT are far less than the benefits in this population,” coinvestigator Nagy A. Youssef, MD, PhD, professor of psychiatry and director of clinical research, The Ohio State University, Columbus, told this news organization.
“My hope is that providers will not be afraid to refer appropriate cases for ECT. If meds and other therapeutics are not working, you should start discussing ECT as a second or third line,” he said.
The findings were presented at the American Society of Clinical Psychopharmacology annual meeting.
Lower mortality
Dr. Youssef, a brain stimulation researcher who uses ECT in his clinical practice, said that in his experience, it is a highly effective therapy for resistant depression.
“I see great responses in patients who have tried everything else. Most of the time, it works very well, and results are very rewarding.”
For the study, the investigators used a large, national insurance claims database that included 949,394 adult inpatients with MDD across the United States from 2012 to 2014. The cohort represented over 4,000 hospitals across the country.
The investigators used logistic regression to determine the odds ratio for in-hospital all-cause mortality for the 25,535 MDD patients who were treated with ECT in comparison with 923,859 patients with MDD who were not treated with ECT.
Results showed that ECT use was significantly higher among older patients (mean age, 56.9 years), women (64%), and White patients (86.9%). In addition, patients in the ECT group were physically sicker than were their peers in the non-ECT group.
A higher proportion of patients in the ECT group in comparison with the non-ECT group had experienced major loss of physical function (37% vs. 5%, respectively) and extreme loss of physical function (63% vs. 0.2%).
“By loss of function, I mean the degree of impairment caused by medical disease,” said Dr. Youssef.
He added that patients with MDD are more likely to care less for their health and do things that are not good for their well-being, such as drinking alcohol or using drugs, and are less likely to adhere to prescribed medication regimens or seek medical attention for physical illness.
“Also, there is probably a biological component where depression, by dysregulation of the hypothalamus and pituitary regions of the brain, can increase the likelihood of physical illness or disease,” Dr. Youssef said.
After adjusting for demographics and extreme loss of function because of medical conditions, the investigators found that in-hospital mortality was significantly lower in the ECT group (odds ratio [OR], 0.05; 95% confidence interval [CI], 0.02-0.11; P < .001).
In-hospital mortality was numerically but not statistically significantly lower in the ECT group (OR, 0.7; 95% CI, 0.41-1.50; P < .47) when adjusted for demographics and major loss of function.
“While this was not statistically significant with marked loss of function, it is clinically important and meaningful. With extreme loss of function, the decrease in mortality was statistically significant,” Dr. Youssef noted.
Designations of extreme and major loss of function were derived from ICD codes.
“This is a complex grading system that takes into account how sick the patient is and includes medical disease severity and comorbidities assessed by the clinician,” he said.
A lifesaving treatment
Commenting on the study, Jair C. Soares, MD, PhD, professor and chair, Pat Rutherford Chair in Psychiatry, UT Houston Medical School, Texas, said, “These are interesting results in a very large national sample suggesting some potential benefits of ECT.
“For the most severely ill patients with major depression who do not respond to currently available medications, ECT is still the most efficacious treatment and indeed a lifesaving treatment modality for many patients,” said Dr. Soares, who was not part of the study.
He noted that ECT is not right for everyone, but “as administered these days, with careful patient selection, it is indeed a safe treatment that can save many lives,” Dr. Soares said.
Dr. Youssef reports a financial relationship with Mecta. Dr. Soares reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an analysis of data from a large database of inpatients across the United States, use of ECT for those with resistant MDD was associated with significantly lower in-hospital mortality compared with those who did not receive ECT.
This held true even after the researchers controlled for demographics and loss of function due to comorbid medical conditions.
“I think the risks of ECT are far less than the benefits in this population,” coinvestigator Nagy A. Youssef, MD, PhD, professor of psychiatry and director of clinical research, The Ohio State University, Columbus, told this news organization.
“My hope is that providers will not be afraid to refer appropriate cases for ECT. If meds and other therapeutics are not working, you should start discussing ECT as a second or third line,” he said.
The findings were presented at the American Society of Clinical Psychopharmacology annual meeting.
Lower mortality
Dr. Youssef, a brain stimulation researcher who uses ECT in his clinical practice, said that in his experience, it is a highly effective therapy for resistant depression.
“I see great responses in patients who have tried everything else. Most of the time, it works very well, and results are very rewarding.”
For the study, the investigators used a large, national insurance claims database that included 949,394 adult inpatients with MDD across the United States from 2012 to 2014. The cohort represented over 4,000 hospitals across the country.
The investigators used logistic regression to determine the odds ratio for in-hospital all-cause mortality for the 25,535 MDD patients who were treated with ECT in comparison with 923,859 patients with MDD who were not treated with ECT.
Results showed that ECT use was significantly higher among older patients (mean age, 56.9 years), women (64%), and White patients (86.9%). In addition, patients in the ECT group were physically sicker than were their peers in the non-ECT group.
A higher proportion of patients in the ECT group in comparison with the non-ECT group had experienced major loss of physical function (37% vs. 5%, respectively) and extreme loss of physical function (63% vs. 0.2%).
“By loss of function, I mean the degree of impairment caused by medical disease,” said Dr. Youssef.
He added that patients with MDD are more likely to care less for their health and do things that are not good for their well-being, such as drinking alcohol or using drugs, and are less likely to adhere to prescribed medication regimens or seek medical attention for physical illness.
“Also, there is probably a biological component where depression, by dysregulation of the hypothalamus and pituitary regions of the brain, can increase the likelihood of physical illness or disease,” Dr. Youssef said.
After adjusting for demographics and extreme loss of function because of medical conditions, the investigators found that in-hospital mortality was significantly lower in the ECT group (odds ratio [OR], 0.05; 95% confidence interval [CI], 0.02-0.11; P < .001).
In-hospital mortality was numerically but not statistically significantly lower in the ECT group (OR, 0.7; 95% CI, 0.41-1.50; P < .47) when adjusted for demographics and major loss of function.
“While this was not statistically significant with marked loss of function, it is clinically important and meaningful. With extreme loss of function, the decrease in mortality was statistically significant,” Dr. Youssef noted.
Designations of extreme and major loss of function were derived from ICD codes.
“This is a complex grading system that takes into account how sick the patient is and includes medical disease severity and comorbidities assessed by the clinician,” he said.
A lifesaving treatment
Commenting on the study, Jair C. Soares, MD, PhD, professor and chair, Pat Rutherford Chair in Psychiatry, UT Houston Medical School, Texas, said, “These are interesting results in a very large national sample suggesting some potential benefits of ECT.
“For the most severely ill patients with major depression who do not respond to currently available medications, ECT is still the most efficacious treatment and indeed a lifesaving treatment modality for many patients,” said Dr. Soares, who was not part of the study.
He noted that ECT is not right for everyone, but “as administered these days, with careful patient selection, it is indeed a safe treatment that can save many lives,” Dr. Soares said.
Dr. Youssef reports a financial relationship with Mecta. Dr. Soares reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In an analysis of data from a large database of inpatients across the United States, use of ECT for those with resistant MDD was associated with significantly lower in-hospital mortality compared with those who did not receive ECT.
This held true even after the researchers controlled for demographics and loss of function due to comorbid medical conditions.
“I think the risks of ECT are far less than the benefits in this population,” coinvestigator Nagy A. Youssef, MD, PhD, professor of psychiatry and director of clinical research, The Ohio State University, Columbus, told this news organization.
“My hope is that providers will not be afraid to refer appropriate cases for ECT. If meds and other therapeutics are not working, you should start discussing ECT as a second or third line,” he said.
The findings were presented at the American Society of Clinical Psychopharmacology annual meeting.
Lower mortality
Dr. Youssef, a brain stimulation researcher who uses ECT in his clinical practice, said that in his experience, it is a highly effective therapy for resistant depression.
“I see great responses in patients who have tried everything else. Most of the time, it works very well, and results are very rewarding.”
For the study, the investigators used a large, national insurance claims database that included 949,394 adult inpatients with MDD across the United States from 2012 to 2014. The cohort represented over 4,000 hospitals across the country.
The investigators used logistic regression to determine the odds ratio for in-hospital all-cause mortality for the 25,535 MDD patients who were treated with ECT in comparison with 923,859 patients with MDD who were not treated with ECT.
Results showed that ECT use was significantly higher among older patients (mean age, 56.9 years), women (64%), and White patients (86.9%). In addition, patients in the ECT group were physically sicker than were their peers in the non-ECT group.
A higher proportion of patients in the ECT group in comparison with the non-ECT group had experienced major loss of physical function (37% vs. 5%, respectively) and extreme loss of physical function (63% vs. 0.2%).
“By loss of function, I mean the degree of impairment caused by medical disease,” said Dr. Youssef.
He added that patients with MDD are more likely to care less for their health and do things that are not good for their well-being, such as drinking alcohol or using drugs, and are less likely to adhere to prescribed medication regimens or seek medical attention for physical illness.
“Also, there is probably a biological component where depression, by dysregulation of the hypothalamus and pituitary regions of the brain, can increase the likelihood of physical illness or disease,” Dr. Youssef said.
After adjusting for demographics and extreme loss of function because of medical conditions, the investigators found that in-hospital mortality was significantly lower in the ECT group (odds ratio [OR], 0.05; 95% confidence interval [CI], 0.02-0.11; P < .001).
In-hospital mortality was numerically but not statistically significantly lower in the ECT group (OR, 0.7; 95% CI, 0.41-1.50; P < .47) when adjusted for demographics and major loss of function.
“While this was not statistically significant with marked loss of function, it is clinically important and meaningful. With extreme loss of function, the decrease in mortality was statistically significant,” Dr. Youssef noted.
Designations of extreme and major loss of function were derived from ICD codes.
“This is a complex grading system that takes into account how sick the patient is and includes medical disease severity and comorbidities assessed by the clinician,” he said.
A lifesaving treatment
Commenting on the study, Jair C. Soares, MD, PhD, professor and chair, Pat Rutherford Chair in Psychiatry, UT Houston Medical School, Texas, said, “These are interesting results in a very large national sample suggesting some potential benefits of ECT.
“For the most severely ill patients with major depression who do not respond to currently available medications, ECT is still the most efficacious treatment and indeed a lifesaving treatment modality for many patients,” said Dr. Soares, who was not part of the study.
He noted that ECT is not right for everyone, but “as administered these days, with careful patient selection, it is indeed a safe treatment that can save many lives,” Dr. Soares said.
Dr. Youssef reports a financial relationship with Mecta. Dr. Soares reports no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ASCP 2022
Surprising link between herpes zoster and dementia
Herpes zoster does not appear to increase dementia risk – on the contrary, the viral infection may offer some protection, a large population-based study suggests.
“We were surprised by these results [and] the reasons for the decreased risk are unclear,” study author Sigrun Alba Johannesdottir Schmidt, MD, PhD, with Aarhus (Denmark) University Hospital, said in a news release.
The study was published online in Neurology.
Conflicting findings
Herpes zoster (HZ) is an acute, cutaneous viral infection caused by the reactivation of varicella-zoster virus (VZV). Previous population-based studies have reported both decreased and increased risks of dementia after having HZ.
It’s thought that HZ may contribute to the development of dementia through neuroinflammation, cerebral vasculopathy, or direct neural damage, but epidemiologic evidence is limited.
To investigate further, Dr. Schmidt and colleagues used Danish medical registries to identify 247,305 people who had visited a hospital for HZ or were prescribed antiviral medication for HZ over a 20-year period and matched them to 1,235,890 people who did not have HZ. For both cohorts, the median age was 64 years, and 61% were women.
Dementia was diagnosed in 9.7% of zoster patients and 10.3% of matched control persons during up to 21 years of follow-up.
Contrary to the researchers’ expectation, HZ was associated with a small (7%) decreased relative risk of all-cause dementia during follow-up (hazard ratio, 0.93; 95% confidence interval, 0.90-0.95).
There was no increased long-term risk of dementia in subgroup analyses, except possibly among those with HZ that involved the central nervous system (HR, 1.94; 95% CI, 0.78-4.80), which has been shown before.
However, the population attributable fraction of dementia caused by this rare complication is low (< 1%), suggesting that universal vaccination against VZV in the elderly has limited potential to reduce dementia risk, the investigators noted.
Nonetheless, Dr. Schmidt said shingles vaccination should be encouraged in older people because it can prevent complications from the disease.
The research team admitted that the slightly decreased long-term risk of dementia, including Alzheimer’s disease, was “unexpected.” The reasons for this decreased risk are unclear, they say, and could be explained by missed diagnoses of shingles in people with undiagnosed dementia.
They were not able to examine whether antiviral treatment modifies the association between HZ and dementia and said that this topic merits further research.
The study was supported by the Edel and Wilhelm Daubenmerkls Charitable Foundation. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Herpes zoster does not appear to increase dementia risk – on the contrary, the viral infection may offer some protection, a large population-based study suggests.
“We were surprised by these results [and] the reasons for the decreased risk are unclear,” study author Sigrun Alba Johannesdottir Schmidt, MD, PhD, with Aarhus (Denmark) University Hospital, said in a news release.
The study was published online in Neurology.
Conflicting findings
Herpes zoster (HZ) is an acute, cutaneous viral infection caused by the reactivation of varicella-zoster virus (VZV). Previous population-based studies have reported both decreased and increased risks of dementia after having HZ.
It’s thought that HZ may contribute to the development of dementia through neuroinflammation, cerebral vasculopathy, or direct neural damage, but epidemiologic evidence is limited.
To investigate further, Dr. Schmidt and colleagues used Danish medical registries to identify 247,305 people who had visited a hospital for HZ or were prescribed antiviral medication for HZ over a 20-year period and matched them to 1,235,890 people who did not have HZ. For both cohorts, the median age was 64 years, and 61% were women.
Dementia was diagnosed in 9.7% of zoster patients and 10.3% of matched control persons during up to 21 years of follow-up.
Contrary to the researchers’ expectation, HZ was associated with a small (7%) decreased relative risk of all-cause dementia during follow-up (hazard ratio, 0.93; 95% confidence interval, 0.90-0.95).
There was no increased long-term risk of dementia in subgroup analyses, except possibly among those with HZ that involved the central nervous system (HR, 1.94; 95% CI, 0.78-4.80), which has been shown before.
However, the population attributable fraction of dementia caused by this rare complication is low (< 1%), suggesting that universal vaccination against VZV in the elderly has limited potential to reduce dementia risk, the investigators noted.
Nonetheless, Dr. Schmidt said shingles vaccination should be encouraged in older people because it can prevent complications from the disease.
The research team admitted that the slightly decreased long-term risk of dementia, including Alzheimer’s disease, was “unexpected.” The reasons for this decreased risk are unclear, they say, and could be explained by missed diagnoses of shingles in people with undiagnosed dementia.
They were not able to examine whether antiviral treatment modifies the association between HZ and dementia and said that this topic merits further research.
The study was supported by the Edel and Wilhelm Daubenmerkls Charitable Foundation. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Herpes zoster does not appear to increase dementia risk – on the contrary, the viral infection may offer some protection, a large population-based study suggests.
“We were surprised by these results [and] the reasons for the decreased risk are unclear,” study author Sigrun Alba Johannesdottir Schmidt, MD, PhD, with Aarhus (Denmark) University Hospital, said in a news release.
The study was published online in Neurology.
Conflicting findings
Herpes zoster (HZ) is an acute, cutaneous viral infection caused by the reactivation of varicella-zoster virus (VZV). Previous population-based studies have reported both decreased and increased risks of dementia after having HZ.
It’s thought that HZ may contribute to the development of dementia through neuroinflammation, cerebral vasculopathy, or direct neural damage, but epidemiologic evidence is limited.
To investigate further, Dr. Schmidt and colleagues used Danish medical registries to identify 247,305 people who had visited a hospital for HZ or were prescribed antiviral medication for HZ over a 20-year period and matched them to 1,235,890 people who did not have HZ. For both cohorts, the median age was 64 years, and 61% were women.
Dementia was diagnosed in 9.7% of zoster patients and 10.3% of matched control persons during up to 21 years of follow-up.
Contrary to the researchers’ expectation, HZ was associated with a small (7%) decreased relative risk of all-cause dementia during follow-up (hazard ratio, 0.93; 95% confidence interval, 0.90-0.95).
There was no increased long-term risk of dementia in subgroup analyses, except possibly among those with HZ that involved the central nervous system (HR, 1.94; 95% CI, 0.78-4.80), which has been shown before.
However, the population attributable fraction of dementia caused by this rare complication is low (< 1%), suggesting that universal vaccination against VZV in the elderly has limited potential to reduce dementia risk, the investigators noted.
Nonetheless, Dr. Schmidt said shingles vaccination should be encouraged in older people because it can prevent complications from the disease.
The research team admitted that the slightly decreased long-term risk of dementia, including Alzheimer’s disease, was “unexpected.” The reasons for this decreased risk are unclear, they say, and could be explained by missed diagnoses of shingles in people with undiagnosed dementia.
They were not able to examine whether antiviral treatment modifies the association between HZ and dementia and said that this topic merits further research.
The study was supported by the Edel and Wilhelm Daubenmerkls Charitable Foundation. The authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
New—and surprising—ways to approach migraine pain
Migraine headaches pose a challenge for many patients and their physicians, so new, effective approaches are always welcome. Sometimes new treatments come as total surprises. For example, who would have guessed that timolol eyedrops could be effective for acute migraine?1 Granted, the results (discussed in this issue's PURLs) are from a single randomized trial, but they look very promising.
This is not the only new and innovative treatment for migraine. Everyone knows about the heavily marketed calcium gene-related peptide antagonists, which include monoclonal antibodies and the so-called “gepants.” The monoclonal antibodies and atogepant are approved for migraine prevention, and they do a decent job (although at a high price). In randomized trials, these agents reduced migraine days per month by an average of about 1.5 to 2.5 days compared to placebo.2-5
Ubrogepant and rimegepant are approved for acute migraine treatment. In clinical trials, about 20% of patients taking ubrogepant or rimegepant were pain free at 2 hours post dose, compared to 12% to 14% taking placebo.6,7 Unfortunately, that means 80% of patients still have some pain at 2 hours. By comparison, zolmitriptan performs a bit better, with 34% of patients pain free at 2 hours.8 However, for those who can’t tolerate zolmitriptan, these newer options provide an alternative.
We also now have nonpharmacologic options. The caloric vestibular stimulation device is essentially a headset with ear probes that change temperature, alternating warm and cold. In a randomized controlled trial, it reduced monthly migraine days by 1.1 compared to placebo, from a baseline of 7.7 to 3.9 days.9 It can also be used to treat acute migraine. There is also a vagus nerve–stimulating device that reduced migraine headache severity by 20% on average in 32.2% of patients in 30 minutes. Sham treatment was as effective for 18.5% of patients, giving a number needed to treat of 6 compared to sham.10
And finally, there are complementary and alternative medicine options. Two recent randomized trials demonstrated that ≥ 2000 IU/d of vitamin D reduced monthly migraine days an average of 2 days, which is comparable to the effectiveness of the calcium gene-related peptide antagonists at a fraction of the cost.11,12 In another randomized trial, intranasal 1.5% peppermint oil was as effective as topical 4% lidocaine in providing substantial pain relief for acute migraine; about 42% of patients achieved significant relief with either treatment.13
While we may not have a perfect treatment for our patients with migraine headache, we certainly have many options to choose from.
1. Ge Y, Castelli G. Migraine relief in 20 minutes using eyedrops? J Fam Pract. 2022;71:222-223, 226.
2. Loder E, Renthal W. Calcitonin gene-related peptide monoclonal antibody treatments for migraine. JAMA Intern Med. 2019;179:421-422. doi: 10.1001/jamainternmed.2018.7536
3. Silberstein S, Diamond M, Hindiyeh NA, et al. Eptinezumab for the prevention of chronic migraine: efficacy and safety through 24 weeks of treatment in the phase 3 PROMISE-2 (Prevention of migraine via intravenous ALD403 safety and efficacy-2) study. J Headache Pain. 2020;21:120. doi: 10.1186/s10194-020-01186-3
4. Ament M, Day K, Stauffer VL, et al. Effect of galcanezumab on severity and symptoms of migraine in phase 3 trials in patients with episodic or chronic migraine. J Headache Pain. 2021;22:6. doi: 10.1186/s10194-021-01215-9
5. Goadsby PJ, Dodick DW, Ailani J, et al. Safety, tolerability, and efficacy of orally administered atogepant for the prevention of episodic migraine in adults: a double-blind, randomised phase 2b/3 trial. Lancet Neurol. 2020;19:727-737. doi: 10.1016/S1474-4422(20)30234-9
6. Lipton RB, Croop R, Stock EG, et al. Rimegepant, an oral calcitonin gene-related peptide receptor antagonist, for migraine. N Engl J Med. 2019;381:142-149. doi: 10.1056/NEJMoa1811090
7. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
8. Bird S, Derry S, Moore R. Zolmitriptan for acute migraine attacks in adults. Cochrane Database Syst Rev. 2014;2014:CD008616. doi: 10.1002/14651858.CD008616.pub2
9. Wilkinson D, Ade KK, Rogers LL, et al. Preventing episodic migraine with caloric vestibular stimulation: a randomized controlled trial. Headache. 2017;57:1065-1087. doi: 10.1111/head.13120
10. Grazzi L, Tassorelli C, de Tommaso M, et al; PRESTO Study Group. Practical and clinical utility of non-invasive vagus nerve stimulation (nVNS) for the acute treatment of migraine: a post hoc analysis of the randomized, sham-controlled, double-blind PRESTO trial. J Headache Pain. 2018;19:98. doi: 10.1186/s10194-018-0928-1
11. Gazerani P, Fuglsang R, Pedersen JG, et al. A randomized, double-blinded, placebo-controlled, parallel trial of vitamin D3 supplementation in adult patients with migraine. Curr Med Res Opin. 2019;35:715-723. doi: 10.1080/03007995.2018.1519503
12. Ghorbani Z, Togha M, Rafiee P, et al. Vitamin D3 might improve headache characteristics and protect against inflammation in migraine: a randomized clinical trial. Neurol Sci. 2020;41:1183-1192. doi: 10.1007/s10072-019-04220-8
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
Migraine headaches pose a challenge for many patients and their physicians, so new, effective approaches are always welcome. Sometimes new treatments come as total surprises. For example, who would have guessed that timolol eyedrops could be effective for acute migraine?1 Granted, the results (discussed in this issue's PURLs) are from a single randomized trial, but they look very promising.
This is not the only new and innovative treatment for migraine. Everyone knows about the heavily marketed calcium gene-related peptide antagonists, which include monoclonal antibodies and the so-called “gepants.” The monoclonal antibodies and atogepant are approved for migraine prevention, and they do a decent job (although at a high price). In randomized trials, these agents reduced migraine days per month by an average of about 1.5 to 2.5 days compared to placebo.2-5
Ubrogepant and rimegepant are approved for acute migraine treatment. In clinical trials, about 20% of patients taking ubrogepant or rimegepant were pain free at 2 hours post dose, compared to 12% to 14% taking placebo.6,7 Unfortunately, that means 80% of patients still have some pain at 2 hours. By comparison, zolmitriptan performs a bit better, with 34% of patients pain free at 2 hours.8 However, for those who can’t tolerate zolmitriptan, these newer options provide an alternative.
We also now have nonpharmacologic options. The caloric vestibular stimulation device is essentially a headset with ear probes that change temperature, alternating warm and cold. In a randomized controlled trial, it reduced monthly migraine days by 1.1 compared to placebo, from a baseline of 7.7 to 3.9 days.9 It can also be used to treat acute migraine. There is also a vagus nerve–stimulating device that reduced migraine headache severity by 20% on average in 32.2% of patients in 30 minutes. Sham treatment was as effective for 18.5% of patients, giving a number needed to treat of 6 compared to sham.10
And finally, there are complementary and alternative medicine options. Two recent randomized trials demonstrated that ≥ 2000 IU/d of vitamin D reduced monthly migraine days an average of 2 days, which is comparable to the effectiveness of the calcium gene-related peptide antagonists at a fraction of the cost.11,12 In another randomized trial, intranasal 1.5% peppermint oil was as effective as topical 4% lidocaine in providing substantial pain relief for acute migraine; about 42% of patients achieved significant relief with either treatment.13
While we may not have a perfect treatment for our patients with migraine headache, we certainly have many options to choose from.
Migraine headaches pose a challenge for many patients and their physicians, so new, effective approaches are always welcome. Sometimes new treatments come as total surprises. For example, who would have guessed that timolol eyedrops could be effective for acute migraine?1 Granted, the results (discussed in this issue's PURLs) are from a single randomized trial, but they look very promising.
This is not the only new and innovative treatment for migraine. Everyone knows about the heavily marketed calcium gene-related peptide antagonists, which include monoclonal antibodies and the so-called “gepants.” The monoclonal antibodies and atogepant are approved for migraine prevention, and they do a decent job (although at a high price). In randomized trials, these agents reduced migraine days per month by an average of about 1.5 to 2.5 days compared to placebo.2-5
Ubrogepant and rimegepant are approved for acute migraine treatment. In clinical trials, about 20% of patients taking ubrogepant or rimegepant were pain free at 2 hours post dose, compared to 12% to 14% taking placebo.6,7 Unfortunately, that means 80% of patients still have some pain at 2 hours. By comparison, zolmitriptan performs a bit better, with 34% of patients pain free at 2 hours.8 However, for those who can’t tolerate zolmitriptan, these newer options provide an alternative.
We also now have nonpharmacologic options. The caloric vestibular stimulation device is essentially a headset with ear probes that change temperature, alternating warm and cold. In a randomized controlled trial, it reduced monthly migraine days by 1.1 compared to placebo, from a baseline of 7.7 to 3.9 days.9 It can also be used to treat acute migraine. There is also a vagus nerve–stimulating device that reduced migraine headache severity by 20% on average in 32.2% of patients in 30 minutes. Sham treatment was as effective for 18.5% of patients, giving a number needed to treat of 6 compared to sham.10
And finally, there are complementary and alternative medicine options. Two recent randomized trials demonstrated that ≥ 2000 IU/d of vitamin D reduced monthly migraine days an average of 2 days, which is comparable to the effectiveness of the calcium gene-related peptide antagonists at a fraction of the cost.11,12 In another randomized trial, intranasal 1.5% peppermint oil was as effective as topical 4% lidocaine in providing substantial pain relief for acute migraine; about 42% of patients achieved significant relief with either treatment.13
While we may not have a perfect treatment for our patients with migraine headache, we certainly have many options to choose from.
1. Ge Y, Castelli G. Migraine relief in 20 minutes using eyedrops? J Fam Pract. 2022;71:222-223, 226.
2. Loder E, Renthal W. Calcitonin gene-related peptide monoclonal antibody treatments for migraine. JAMA Intern Med. 2019;179:421-422. doi: 10.1001/jamainternmed.2018.7536
3. Silberstein S, Diamond M, Hindiyeh NA, et al. Eptinezumab for the prevention of chronic migraine: efficacy and safety through 24 weeks of treatment in the phase 3 PROMISE-2 (Prevention of migraine via intravenous ALD403 safety and efficacy-2) study. J Headache Pain. 2020;21:120. doi: 10.1186/s10194-020-01186-3
4. Ament M, Day K, Stauffer VL, et al. Effect of galcanezumab on severity and symptoms of migraine in phase 3 trials in patients with episodic or chronic migraine. J Headache Pain. 2021;22:6. doi: 10.1186/s10194-021-01215-9
5. Goadsby PJ, Dodick DW, Ailani J, et al. Safety, tolerability, and efficacy of orally administered atogepant for the prevention of episodic migraine in adults: a double-blind, randomised phase 2b/3 trial. Lancet Neurol. 2020;19:727-737. doi: 10.1016/S1474-4422(20)30234-9
6. Lipton RB, Croop R, Stock EG, et al. Rimegepant, an oral calcitonin gene-related peptide receptor antagonist, for migraine. N Engl J Med. 2019;381:142-149. doi: 10.1056/NEJMoa1811090
7. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
8. Bird S, Derry S, Moore R. Zolmitriptan for acute migraine attacks in adults. Cochrane Database Syst Rev. 2014;2014:CD008616. doi: 10.1002/14651858.CD008616.pub2
9. Wilkinson D, Ade KK, Rogers LL, et al. Preventing episodic migraine with caloric vestibular stimulation: a randomized controlled trial. Headache. 2017;57:1065-1087. doi: 10.1111/head.13120
10. Grazzi L, Tassorelli C, de Tommaso M, et al; PRESTO Study Group. Practical and clinical utility of non-invasive vagus nerve stimulation (nVNS) for the acute treatment of migraine: a post hoc analysis of the randomized, sham-controlled, double-blind PRESTO trial. J Headache Pain. 2018;19:98. doi: 10.1186/s10194-018-0928-1
11. Gazerani P, Fuglsang R, Pedersen JG, et al. A randomized, double-blinded, placebo-controlled, parallel trial of vitamin D3 supplementation in adult patients with migraine. Curr Med Res Opin. 2019;35:715-723. doi: 10.1080/03007995.2018.1519503
12. Ghorbani Z, Togha M, Rafiee P, et al. Vitamin D3 might improve headache characteristics and protect against inflammation in migraine: a randomized clinical trial. Neurol Sci. 2020;41:1183-1192. doi: 10.1007/s10072-019-04220-8
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
1. Ge Y, Castelli G. Migraine relief in 20 minutes using eyedrops? J Fam Pract. 2022;71:222-223, 226.
2. Loder E, Renthal W. Calcitonin gene-related peptide monoclonal antibody treatments for migraine. JAMA Intern Med. 2019;179:421-422. doi: 10.1001/jamainternmed.2018.7536
3. Silberstein S, Diamond M, Hindiyeh NA, et al. Eptinezumab for the prevention of chronic migraine: efficacy and safety through 24 weeks of treatment in the phase 3 PROMISE-2 (Prevention of migraine via intravenous ALD403 safety and efficacy-2) study. J Headache Pain. 2020;21:120. doi: 10.1186/s10194-020-01186-3
4. Ament M, Day K, Stauffer VL, et al. Effect of galcanezumab on severity and symptoms of migraine in phase 3 trials in patients with episodic or chronic migraine. J Headache Pain. 2021;22:6. doi: 10.1186/s10194-021-01215-9
5. Goadsby PJ, Dodick DW, Ailani J, et al. Safety, tolerability, and efficacy of orally administered atogepant for the prevention of episodic migraine in adults: a double-blind, randomised phase 2b/3 trial. Lancet Neurol. 2020;19:727-737. doi: 10.1016/S1474-4422(20)30234-9
6. Lipton RB, Croop R, Stock EG, et al. Rimegepant, an oral calcitonin gene-related peptide receptor antagonist, for migraine. N Engl J Med. 2019;381:142-149. doi: 10.1056/NEJMoa1811090
7. Lipton RB, Dodick DW, Ailani J, et al. Effect of ubrogepant vs placebo on pain and the most bothersome associated symptom in the acute treatment of migraine: the ACHIEVE II randomized clinical trial. JAMA. 2019;322:1887-1898. doi: 10.1001/jama.2019.16711
8. Bird S, Derry S, Moore R. Zolmitriptan for acute migraine attacks in adults. Cochrane Database Syst Rev. 2014;2014:CD008616. doi: 10.1002/14651858.CD008616.pub2
9. Wilkinson D, Ade KK, Rogers LL, et al. Preventing episodic migraine with caloric vestibular stimulation: a randomized controlled trial. Headache. 2017;57:1065-1087. doi: 10.1111/head.13120
10. Grazzi L, Tassorelli C, de Tommaso M, et al; PRESTO Study Group. Practical and clinical utility of non-invasive vagus nerve stimulation (nVNS) for the acute treatment of migraine: a post hoc analysis of the randomized, sham-controlled, double-blind PRESTO trial. J Headache Pain. 2018;19:98. doi: 10.1186/s10194-018-0928-1
11. Gazerani P, Fuglsang R, Pedersen JG, et al. A randomized, double-blinded, placebo-controlled, parallel trial of vitamin D3 supplementation in adult patients with migraine. Curr Med Res Opin. 2019;35:715-723. doi: 10.1080/03007995.2018.1519503
12. Ghorbani Z, Togha M, Rafiee P, et al. Vitamin D3 might improve headache characteristics and protect against inflammation in migraine: a randomized clinical trial. Neurol Sci. 2020;41:1183-1192. doi: 10.1007/s10072-019-04220-8
13. Rafieian-Kopaei M, Hasanpour-Dehkordi A, Lorigooini Z, et al. Comparing the effect of intranasal lidocaine 4% with peppermint essential oil drop 1.5% on migraine attacks: a double-blind clinical trial. Int J Prev Med. 2019;10:121. doi: 10.4103/ijpvm.IJPVM_530_17
Analysis shows predictive capabilities of sleep EEG
CHARLOTTE, N.C. – , a researcher reported at the annual meeting of the Associated Professional Sleep Societies. “Sleep EEGs contain decodable information about the risk of unfavorable outcomes,” said Haoqi Sun, PhD, an instructor of neurology at Massachusetts General Hospital, Boston, and lead study author. “The results suggest that it’s feasible to use sleep to identify people with high risk of unfavorable outcomes and it strengthens the concept of sleep as a window into brain and general health.”
The researchers performed a quantitative analysis of sleep data collected on 8,673 adults who had diagnostic sleep studies that included polysomnography (PSG). The analysis used ICD codes to consider these 11 health outcomes: dementia, mild cognitive impairment (MCI) or dementia, ischemic stroke, intracranial hemorrhage, atrial fibrillation, myocardial infarction, type 2 diabetes, hypertension, bipolar disorder, depression, and mortality.
Then, Dr. Sun explained, they extracted 86 spectral and time-domain features of REM and non-REM sleep from sleep EEG recordings, and analyzed that data by adjusting for eight covariates including age, sex, body mass index, and use of benzodiazepines, antidepressants, sedatives, antiseizure drugs, and stimulants.
Participants were partitioned into three sleep-quality groups: poor, average, and good. The outcome-wise mean prediction difference in 10-year cumulative incidence was 2.3% for the poor sleep group, 0.5% for the average sleep group, and 1.3% for the good sleep group.
The outcomes with the three greatest poor to average risk ratios were dementia (6.2; 95% confidence interval, 4.5-9.3), mortality (5.7; 95% CI, 5-7.5) and MCI or dementia (4; 95% CI, 3.2-4.9).
Ready for the clinic?
In an interview, Dr. Sun said the results demonstrated the potential of using EEG brain wave data to predict health outcomes on an individual basis, although he acknowledged that most of the 86 sleep features the researchers used are not readily available in the clinic.
He noted the spectral features used in the study can be captured through software compatible with PSG. “From there you can identify the various bands, the different frequency ranges, and then you can easily see within this range whether a person has a higher power or lower power,” he said. However, the spindle and slow-oscillation features that researchers used in the study are beyond the reach of most clinics.
Next steps
This research is in its early stage, Dr. Sun said, but at some point the data collected from sleep studies could be paired with machine learning to make the model workable for evaluating individual patients. “Our goal is to first make this individualized,” he said. “We want to minimize the noise in the recording and minimize the night-to-night variability in the findings. There is some clinical-informed approach and there is also some algorithm-informed approach where you can minimize the variation over time.”
The model also has the potential to predict outcomes, particularly with chronic diseases such as diabetes and dementia, well before a diagnosis is made, he said.
‘Fascinating’ and ‘provocative’
Donald Bliwise, PhD, professor of neurology at Emory Sleep Center in Atlanta, said the study was “fascinating; it’s provocative; it’s exciting and interesting,” but added, “Sleep is vital for health. That’s abundantly clear in a study like that, but trying to push it a little bit further with all of these 86 measurements of the EEG, I think it becomes complicated.”
The study methodology, particularly the use of cumulative incidence of various diseases, was laudable, he said, and the use of simpler EEG-measured sleep features, such as alpha band power, “make intuitive sense.”
But it’s less clear on how the more sophisticated features the study model used – for example, kurtosis of theta frequency or coupling between spindle and slow oscillation – rank on sleep quality, he said, adding that the researchers have most likely done that but couldn’t add that into the format of the presentation.
“Kurtosis of the theta frequency band we don’t get on everyone in the sleep lab,” Dr. Bliwise said. “We might be able to, but I don’t know how to quite plug that into a turnkey model.”
The clinical components of the study were conducted by M. Brandon Westover, MD, PhD, at Massachusetts General Hospital, and Robert J. Thomas, MD, at Beth Israel Deaconess Medical Center, both in Boston. The study received support from the American Academy of Sleep Medicine Foundation. Dr. Sun has no relevant disclosures. Dr. Bliwise has no disclosures.
CHARLOTTE, N.C. – , a researcher reported at the annual meeting of the Associated Professional Sleep Societies. “Sleep EEGs contain decodable information about the risk of unfavorable outcomes,” said Haoqi Sun, PhD, an instructor of neurology at Massachusetts General Hospital, Boston, and lead study author. “The results suggest that it’s feasible to use sleep to identify people with high risk of unfavorable outcomes and it strengthens the concept of sleep as a window into brain and general health.”
The researchers performed a quantitative analysis of sleep data collected on 8,673 adults who had diagnostic sleep studies that included polysomnography (PSG). The analysis used ICD codes to consider these 11 health outcomes: dementia, mild cognitive impairment (MCI) or dementia, ischemic stroke, intracranial hemorrhage, atrial fibrillation, myocardial infarction, type 2 diabetes, hypertension, bipolar disorder, depression, and mortality.
Then, Dr. Sun explained, they extracted 86 spectral and time-domain features of REM and non-REM sleep from sleep EEG recordings, and analyzed that data by adjusting for eight covariates including age, sex, body mass index, and use of benzodiazepines, antidepressants, sedatives, antiseizure drugs, and stimulants.
Participants were partitioned into three sleep-quality groups: poor, average, and good. The outcome-wise mean prediction difference in 10-year cumulative incidence was 2.3% for the poor sleep group, 0.5% for the average sleep group, and 1.3% for the good sleep group.
The outcomes with the three greatest poor to average risk ratios were dementia (6.2; 95% confidence interval, 4.5-9.3), mortality (5.7; 95% CI, 5-7.5) and MCI or dementia (4; 95% CI, 3.2-4.9).
Ready for the clinic?
In an interview, Dr. Sun said the results demonstrated the potential of using EEG brain wave data to predict health outcomes on an individual basis, although he acknowledged that most of the 86 sleep features the researchers used are not readily available in the clinic.
He noted the spectral features used in the study can be captured through software compatible with PSG. “From there you can identify the various bands, the different frequency ranges, and then you can easily see within this range whether a person has a higher power or lower power,” he said. However, the spindle and slow-oscillation features that researchers used in the study are beyond the reach of most clinics.
Next steps
This research is in its early stage, Dr. Sun said, but at some point the data collected from sleep studies could be paired with machine learning to make the model workable for evaluating individual patients. “Our goal is to first make this individualized,” he said. “We want to minimize the noise in the recording and minimize the night-to-night variability in the findings. There is some clinical-informed approach and there is also some algorithm-informed approach where you can minimize the variation over time.”
The model also has the potential to predict outcomes, particularly with chronic diseases such as diabetes and dementia, well before a diagnosis is made, he said.
‘Fascinating’ and ‘provocative’
Donald Bliwise, PhD, professor of neurology at Emory Sleep Center in Atlanta, said the study was “fascinating; it’s provocative; it’s exciting and interesting,” but added, “Sleep is vital for health. That’s abundantly clear in a study like that, but trying to push it a little bit further with all of these 86 measurements of the EEG, I think it becomes complicated.”
The study methodology, particularly the use of cumulative incidence of various diseases, was laudable, he said, and the use of simpler EEG-measured sleep features, such as alpha band power, “make intuitive sense.”
But it’s less clear on how the more sophisticated features the study model used – for example, kurtosis of theta frequency or coupling between spindle and slow oscillation – rank on sleep quality, he said, adding that the researchers have most likely done that but couldn’t add that into the format of the presentation.
“Kurtosis of the theta frequency band we don’t get on everyone in the sleep lab,” Dr. Bliwise said. “We might be able to, but I don’t know how to quite plug that into a turnkey model.”
The clinical components of the study were conducted by M. Brandon Westover, MD, PhD, at Massachusetts General Hospital, and Robert J. Thomas, MD, at Beth Israel Deaconess Medical Center, both in Boston. The study received support from the American Academy of Sleep Medicine Foundation. Dr. Sun has no relevant disclosures. Dr. Bliwise has no disclosures.
CHARLOTTE, N.C. – , a researcher reported at the annual meeting of the Associated Professional Sleep Societies. “Sleep EEGs contain decodable information about the risk of unfavorable outcomes,” said Haoqi Sun, PhD, an instructor of neurology at Massachusetts General Hospital, Boston, and lead study author. “The results suggest that it’s feasible to use sleep to identify people with high risk of unfavorable outcomes and it strengthens the concept of sleep as a window into brain and general health.”
The researchers performed a quantitative analysis of sleep data collected on 8,673 adults who had diagnostic sleep studies that included polysomnography (PSG). The analysis used ICD codes to consider these 11 health outcomes: dementia, mild cognitive impairment (MCI) or dementia, ischemic stroke, intracranial hemorrhage, atrial fibrillation, myocardial infarction, type 2 diabetes, hypertension, bipolar disorder, depression, and mortality.
Then, Dr. Sun explained, they extracted 86 spectral and time-domain features of REM and non-REM sleep from sleep EEG recordings, and analyzed that data by adjusting for eight covariates including age, sex, body mass index, and use of benzodiazepines, antidepressants, sedatives, antiseizure drugs, and stimulants.
Participants were partitioned into three sleep-quality groups: poor, average, and good. The outcome-wise mean prediction difference in 10-year cumulative incidence was 2.3% for the poor sleep group, 0.5% for the average sleep group, and 1.3% for the good sleep group.
The outcomes with the three greatest poor to average risk ratios were dementia (6.2; 95% confidence interval, 4.5-9.3), mortality (5.7; 95% CI, 5-7.5) and MCI or dementia (4; 95% CI, 3.2-4.9).
Ready for the clinic?
In an interview, Dr. Sun said the results demonstrated the potential of using EEG brain wave data to predict health outcomes on an individual basis, although he acknowledged that most of the 86 sleep features the researchers used are not readily available in the clinic.
He noted the spectral features used in the study can be captured through software compatible with PSG. “From there you can identify the various bands, the different frequency ranges, and then you can easily see within this range whether a person has a higher power or lower power,” he said. However, the spindle and slow-oscillation features that researchers used in the study are beyond the reach of most clinics.
Next steps
This research is in its early stage, Dr. Sun said, but at some point the data collected from sleep studies could be paired with machine learning to make the model workable for evaluating individual patients. “Our goal is to first make this individualized,” he said. “We want to minimize the noise in the recording and minimize the night-to-night variability in the findings. There is some clinical-informed approach and there is also some algorithm-informed approach where you can minimize the variation over time.”
The model also has the potential to predict outcomes, particularly with chronic diseases such as diabetes and dementia, well before a diagnosis is made, he said.
‘Fascinating’ and ‘provocative’
Donald Bliwise, PhD, professor of neurology at Emory Sleep Center in Atlanta, said the study was “fascinating; it’s provocative; it’s exciting and interesting,” but added, “Sleep is vital for health. That’s abundantly clear in a study like that, but trying to push it a little bit further with all of these 86 measurements of the EEG, I think it becomes complicated.”
The study methodology, particularly the use of cumulative incidence of various diseases, was laudable, he said, and the use of simpler EEG-measured sleep features, such as alpha band power, “make intuitive sense.”
But it’s less clear on how the more sophisticated features the study model used – for example, kurtosis of theta frequency or coupling between spindle and slow oscillation – rank on sleep quality, he said, adding that the researchers have most likely done that but couldn’t add that into the format of the presentation.
“Kurtosis of the theta frequency band we don’t get on everyone in the sleep lab,” Dr. Bliwise said. “We might be able to, but I don’t know how to quite plug that into a turnkey model.”
The clinical components of the study were conducted by M. Brandon Westover, MD, PhD, at Massachusetts General Hospital, and Robert J. Thomas, MD, at Beth Israel Deaconess Medical Center, both in Boston. The study received support from the American Academy of Sleep Medicine Foundation. Dr. Sun has no relevant disclosures. Dr. Bliwise has no disclosures.
AT SLEEP 2022
Opioid use in the elderly a dementia risk factor?
in new findings that suggest exposure to these drugs may be another modifiable risk factor for dementia.
“Clinicians and others may want to consider that opioid exposure in those aged 75-80 increases dementia risk, and to balance the potential benefits of opioid use in old age with adverse side effects,” said Stephen Z. Levine, PhD, professor, department of community mental health, University of Haifa (Israel).
The study was published online in the American Journal of Geriatric Psychiatry.
Widespread use
Evidence points to a relatively high rate of opioid prescriptions among older adults. A Morbidity and Mortality Weekly Report noted 19.2% of the U.S. adult population filled an opioid prescription in 2018, with the rate in those over 65 double that of adults aged 20-24 years (25% vs. 11.2%).
Disorders and illnesses for which opioids might be prescribed, including cancer and some pain conditions, “are far more prevalent in old age than at a younger age,” said Dr. Levine.
This high rate of opioid use underscores the need to consider the risks of opioid use in old age, said Dr. Levine. “Unfortunately, studies of the association between opioid use and dementia risk in old age are few, and their results are inconsistent.”
The study included 91,307 Israeli citizens aged 60 and over without dementia who were enrolled in the Meuhedet Healthcare Services, a nonprofit health maintenance organization (HMO) serving 14% of the country’s population. Meuhedet has maintained an up-to-date dementia registry since 2002.
The average age of the study sample was 68.29 years at the start of the study (in 2012).
In Israel, opioids are prescribed for a 30-day period. In this study, opioid exposure was defined as opioid medication fills covering 60 days (or two prescriptions) within a 120-day interval.
The primary outcome was incident dementia during follow-up from Jan. 1, 2013 to Oct. 30, 2017. The analysis controlled for a number of factors, including age, sex, smoking status, health conditions such as arthritis, depression, diabetes, osteoporosis, cognitive decline, vitamin deficiencies, cancer, cardiovascular conditions, and hospitalizations for falls.
Researchers also accounted for the competing risk of mortality.
During the study, 3.1% of subjects were exposed to opioids at a mean age of 73.94 years, and 5.8% of subjects developed dementia at an average age of 78.07 years.
Increased dementia risk
The risk of incident dementia was significantly increased in those exposed to opioids versus unexposed individuals in the 75- to 80-year age group (adjusted hazard ratio, 1.39; 95% confidence interval, 1.01-1.92; z statistic = 2.02; P < .05).
The authors noted the effect size for opioid exposure in this elderly age group is like other potentially modifiable risk factors for dementia, including body mass index and smoking.
The current study could not determine the biological explanation for the increased dementia risk among older opioid users. “Causal notions are challenging in observational studies and should be viewed with caution,” Dr. Levine noted.
However, a plausible mechanism highlighted in the literature is that opioids promote apoptosis of microglia and neurons that contribute to neurodegenerative diseases, he said.
The study included 14 sensitivity analyses, including those that looked at females, subjects older than 70, smokers, and groups with and without comorbid health conditions. The only sensitivity analysis that didn’t have similar findings to the primary analysis looked at dementia risk restricted to subjects without a vitamin deficiency.
“It’s reassuring that 13 or 14 sensitivity analyses found a significant association between opioid exposure and dementia risk,” said Dr. Levine.
Some prior studies did not show an association between opioid exposure and dementia risk. One possible reason for the discrepancy with the current findings is that the previous research didn’t account for age-specific opioid use effects, or the competing risk of mortality, said Dr. Levine.
Clinicians have a number of potential alternatives to opioids to treat various conditions including acetaminophen, non-steroidal anti-inflammatory drugs, amine reuptake inhibitors (ARIs), membrane stabilizers, muscle relaxants, topical capsaicin, botulinum toxin, cannabinoids, and steroids.
A limitation of the study was that it didn’t adjust for all possible comorbid health conditions, including vascular conditions, or for use of benzodiazepines, and surgical procedures.
In addition, since up to 50% of dementia cases are undetected, it’s possible some in the unexposed opioid group may actually have undiagnosed dementia, thereby reducing the effect sizes in the results.
Reverse causality is also a possibility as the neuropathological process associated with dementia could have started prior to opioid exposure. In addition, the results are limited to prolonged opioid exposure.
Interpret with caution
Commenting on the study, David Knopman, MD, a neurologist at Mayo Clinic in Rochester, Minn., whose research involves late-life cognitive disorders, was skeptical.
“On the face of it, the fact that an association was seen only in one narrow age range – 75+ to 80 years – ought to raise serious suspicion about the reliability and validity of the claim that opioid use is a risk factor for dementia, he said.
Although the researchers performed several sensitivity analyses, including accounting for mortality, “pharmacoepidemiological studies are terribly sensitive to residual biases” related to physician and patient choices related to medication use, added Dr. Knopman.
The claim that opioids are a dementia risk “should be viewed with great caution” and should not influence use of opioids where they’re truly indicated, he said.
“It would be a great pity if patients with pain requiring opioids avoid them because of fears about dementia based on the dubious relationship between age and opioid use.”
Dr. Levine and Dr. Knopman report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
in new findings that suggest exposure to these drugs may be another modifiable risk factor for dementia.
“Clinicians and others may want to consider that opioid exposure in those aged 75-80 increases dementia risk, and to balance the potential benefits of opioid use in old age with adverse side effects,” said Stephen Z. Levine, PhD, professor, department of community mental health, University of Haifa (Israel).
The study was published online in the American Journal of Geriatric Psychiatry.
Widespread use
Evidence points to a relatively high rate of opioid prescriptions among older adults. A Morbidity and Mortality Weekly Report noted 19.2% of the U.S. adult population filled an opioid prescription in 2018, with the rate in those over 65 double that of adults aged 20-24 years (25% vs. 11.2%).
Disorders and illnesses for which opioids might be prescribed, including cancer and some pain conditions, “are far more prevalent in old age than at a younger age,” said Dr. Levine.
This high rate of opioid use underscores the need to consider the risks of opioid use in old age, said Dr. Levine. “Unfortunately, studies of the association between opioid use and dementia risk in old age are few, and their results are inconsistent.”
The study included 91,307 Israeli citizens aged 60 and over without dementia who were enrolled in the Meuhedet Healthcare Services, a nonprofit health maintenance organization (HMO) serving 14% of the country’s population. Meuhedet has maintained an up-to-date dementia registry since 2002.
The average age of the study sample was 68.29 years at the start of the study (in 2012).
In Israel, opioids are prescribed for a 30-day period. In this study, opioid exposure was defined as opioid medication fills covering 60 days (or two prescriptions) within a 120-day interval.
The primary outcome was incident dementia during follow-up from Jan. 1, 2013 to Oct. 30, 2017. The analysis controlled for a number of factors, including age, sex, smoking status, health conditions such as arthritis, depression, diabetes, osteoporosis, cognitive decline, vitamin deficiencies, cancer, cardiovascular conditions, and hospitalizations for falls.
Researchers also accounted for the competing risk of mortality.
During the study, 3.1% of subjects were exposed to opioids at a mean age of 73.94 years, and 5.8% of subjects developed dementia at an average age of 78.07 years.
Increased dementia risk
The risk of incident dementia was significantly increased in those exposed to opioids versus unexposed individuals in the 75- to 80-year age group (adjusted hazard ratio, 1.39; 95% confidence interval, 1.01-1.92; z statistic = 2.02; P < .05).
The authors noted the effect size for opioid exposure in this elderly age group is like other potentially modifiable risk factors for dementia, including body mass index and smoking.
The current study could not determine the biological explanation for the increased dementia risk among older opioid users. “Causal notions are challenging in observational studies and should be viewed with caution,” Dr. Levine noted.
However, a plausible mechanism highlighted in the literature is that opioids promote apoptosis of microglia and neurons that contribute to neurodegenerative diseases, he said.
The study included 14 sensitivity analyses, including those that looked at females, subjects older than 70, smokers, and groups with and without comorbid health conditions. The only sensitivity analysis that didn’t have similar findings to the primary analysis looked at dementia risk restricted to subjects without a vitamin deficiency.
“It’s reassuring that 13 or 14 sensitivity analyses found a significant association between opioid exposure and dementia risk,” said Dr. Levine.
Some prior studies did not show an association between opioid exposure and dementia risk. One possible reason for the discrepancy with the current findings is that the previous research didn’t account for age-specific opioid use effects, or the competing risk of mortality, said Dr. Levine.
Clinicians have a number of potential alternatives to opioids to treat various conditions including acetaminophen, non-steroidal anti-inflammatory drugs, amine reuptake inhibitors (ARIs), membrane stabilizers, muscle relaxants, topical capsaicin, botulinum toxin, cannabinoids, and steroids.
A limitation of the study was that it didn’t adjust for all possible comorbid health conditions, including vascular conditions, or for use of benzodiazepines, and surgical procedures.
In addition, since up to 50% of dementia cases are undetected, it’s possible some in the unexposed opioid group may actually have undiagnosed dementia, thereby reducing the effect sizes in the results.
Reverse causality is also a possibility as the neuropathological process associated with dementia could have started prior to opioid exposure. In addition, the results are limited to prolonged opioid exposure.
Interpret with caution
Commenting on the study, David Knopman, MD, a neurologist at Mayo Clinic in Rochester, Minn., whose research involves late-life cognitive disorders, was skeptical.
“On the face of it, the fact that an association was seen only in one narrow age range – 75+ to 80 years – ought to raise serious suspicion about the reliability and validity of the claim that opioid use is a risk factor for dementia, he said.
Although the researchers performed several sensitivity analyses, including accounting for mortality, “pharmacoepidemiological studies are terribly sensitive to residual biases” related to physician and patient choices related to medication use, added Dr. Knopman.
The claim that opioids are a dementia risk “should be viewed with great caution” and should not influence use of opioids where they’re truly indicated, he said.
“It would be a great pity if patients with pain requiring opioids avoid them because of fears about dementia based on the dubious relationship between age and opioid use.”
Dr. Levine and Dr. Knopman report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
in new findings that suggest exposure to these drugs may be another modifiable risk factor for dementia.
“Clinicians and others may want to consider that opioid exposure in those aged 75-80 increases dementia risk, and to balance the potential benefits of opioid use in old age with adverse side effects,” said Stephen Z. Levine, PhD, professor, department of community mental health, University of Haifa (Israel).
The study was published online in the American Journal of Geriatric Psychiatry.
Widespread use
Evidence points to a relatively high rate of opioid prescriptions among older adults. A Morbidity and Mortality Weekly Report noted 19.2% of the U.S. adult population filled an opioid prescription in 2018, with the rate in those over 65 double that of adults aged 20-24 years (25% vs. 11.2%).
Disorders and illnesses for which opioids might be prescribed, including cancer and some pain conditions, “are far more prevalent in old age than at a younger age,” said Dr. Levine.
This high rate of opioid use underscores the need to consider the risks of opioid use in old age, said Dr. Levine. “Unfortunately, studies of the association between opioid use and dementia risk in old age are few, and their results are inconsistent.”
The study included 91,307 Israeli citizens aged 60 and over without dementia who were enrolled in the Meuhedet Healthcare Services, a nonprofit health maintenance organization (HMO) serving 14% of the country’s population. Meuhedet has maintained an up-to-date dementia registry since 2002.
The average age of the study sample was 68.29 years at the start of the study (in 2012).
In Israel, opioids are prescribed for a 30-day period. In this study, opioid exposure was defined as opioid medication fills covering 60 days (or two prescriptions) within a 120-day interval.
The primary outcome was incident dementia during follow-up from Jan. 1, 2013 to Oct. 30, 2017. The analysis controlled for a number of factors, including age, sex, smoking status, health conditions such as arthritis, depression, diabetes, osteoporosis, cognitive decline, vitamin deficiencies, cancer, cardiovascular conditions, and hospitalizations for falls.
Researchers also accounted for the competing risk of mortality.
During the study, 3.1% of subjects were exposed to opioids at a mean age of 73.94 years, and 5.8% of subjects developed dementia at an average age of 78.07 years.
Increased dementia risk
The risk of incident dementia was significantly increased in those exposed to opioids versus unexposed individuals in the 75- to 80-year age group (adjusted hazard ratio, 1.39; 95% confidence interval, 1.01-1.92; z statistic = 2.02; P < .05).
The authors noted the effect size for opioid exposure in this elderly age group is like other potentially modifiable risk factors for dementia, including body mass index and smoking.
The current study could not determine the biological explanation for the increased dementia risk among older opioid users. “Causal notions are challenging in observational studies and should be viewed with caution,” Dr. Levine noted.
However, a plausible mechanism highlighted in the literature is that opioids promote apoptosis of microglia and neurons that contribute to neurodegenerative diseases, he said.
The study included 14 sensitivity analyses, including those that looked at females, subjects older than 70, smokers, and groups with and without comorbid health conditions. The only sensitivity analysis that didn’t have similar findings to the primary analysis looked at dementia risk restricted to subjects without a vitamin deficiency.
“It’s reassuring that 13 or 14 sensitivity analyses found a significant association between opioid exposure and dementia risk,” said Dr. Levine.
Some prior studies did not show an association between opioid exposure and dementia risk. One possible reason for the discrepancy with the current findings is that the previous research didn’t account for age-specific opioid use effects, or the competing risk of mortality, said Dr. Levine.
Clinicians have a number of potential alternatives to opioids to treat various conditions including acetaminophen, non-steroidal anti-inflammatory drugs, amine reuptake inhibitors (ARIs), membrane stabilizers, muscle relaxants, topical capsaicin, botulinum toxin, cannabinoids, and steroids.
A limitation of the study was that it didn’t adjust for all possible comorbid health conditions, including vascular conditions, or for use of benzodiazepines, and surgical procedures.
In addition, since up to 50% of dementia cases are undetected, it’s possible some in the unexposed opioid group may actually have undiagnosed dementia, thereby reducing the effect sizes in the results.
Reverse causality is also a possibility as the neuropathological process associated with dementia could have started prior to opioid exposure. In addition, the results are limited to prolonged opioid exposure.
Interpret with caution
Commenting on the study, David Knopman, MD, a neurologist at Mayo Clinic in Rochester, Minn., whose research involves late-life cognitive disorders, was skeptical.
“On the face of it, the fact that an association was seen only in one narrow age range – 75+ to 80 years – ought to raise serious suspicion about the reliability and validity of the claim that opioid use is a risk factor for dementia, he said.
Although the researchers performed several sensitivity analyses, including accounting for mortality, “pharmacoepidemiological studies are terribly sensitive to residual biases” related to physician and patient choices related to medication use, added Dr. Knopman.
The claim that opioids are a dementia risk “should be viewed with great caution” and should not influence use of opioids where they’re truly indicated, he said.
“It would be a great pity if patients with pain requiring opioids avoid them because of fears about dementia based on the dubious relationship between age and opioid use.”
Dr. Levine and Dr. Knopman report no relevant financial disclosures.
A version of this article first appeared on Medscape.com.
FROM AMERICAN JOURNAL OF GERIATRIC PSYCHIATRY
‘Genetic’ height linked to peripheral neuropathy and certain skin and bone infections
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
, according to a study published in PLOS Genetics.
Prior studies have investigated height as a risk factor for chronic diseases, such as a higher risk for atrial fibrillation and a reduced risk of cardiovascular disease. It’s been consistently difficult, however, to eliminate the confounding influences of diet, socioeconomics, lifestyle behaviors, and other environmental factors that may interfere with a person’s reaching their expected height based on their genes.
This study, however, was able to better parse those differences by using Mendelian randomization within the comprehensive clinical and genetic dataset of a national health care system biobank. Mendelian randomization uses “genetic instruments for exposures of interest under the assumption that genotype is less susceptible to confounding than measured exposures,” the authors explained. The findings confirmed previously suspected associations between height and a range of cardiovascular and metabolic conditions as well as revealing new associations with several other conditions.
Prior associations confirmed, new associations uncovered
The results confirmed that being tall is linked to a higher risk of atrial fibrillation and varicose veins, and a lower risk of coronary heart disease, high blood pressure, and high cholesterol. The study also uncovered new associations between greater height and a higher risk of peripheral neuropathy, which is caused by damage to nerves on the extremities, as well as skin and bone infections, such as leg and foot ulcers.
The meta-analysis “identified five additional traits associated with genetically-predicted height,” wrote Sridharan Raghavan, MD, assistant professor of medicine at the University of Colorado Anschutz Medical Campus, and colleagues. “Two were genitourinary conditions – erectile dysfunction and urinary retention – that can be associated with neuropathy, and a third was a phecode for nonspecific skin disorders that may be related to skin infections – consistent with the race/ethnicity stratified results.”
Removing potential confounders
F. Perry Wilson, MD, associate professor of medicine at Yale University, New Haven, Conn., who was not involved in the study, said the findings were not particularly surprising overall, but it’s striking that the researchers had ”such a large cohort with such detailed electronic health records allowing for the comparison of genetic height with a variety of clinical outcomes.” He also noted the study’s strength in using Mendelian randomization so that the exposure is the predicted genetic height instead of a person’s measured height.
“This is key, since lots of things affect actual height – nutrition is an important one that could certainly be linked to disease as well,” Dr. Wilson said. ”By using genetic height, the authors remove these potential confounders. Since genetic height is “assigned” at birth (or conception), there is little opportunity for confounding. Of course, it is possible that some of the gene variants used to predict genetic height actually do something else, such as make you seek out less nutritious meals, but by and large this is how these types of studies need to be done.”
Height may impact over 100 clinical traits
The study relied on data from the U.S. Veteran Affairs Million Veteran Program with 222,300 non-Hispanic White and 58,151 non-Hispanic Black participants. The researchers first estimated the likelihood of participants’ genetic height based on 3,290 genetic variants determined to affect genetic height in a recent European-ancestry genome-wide meta-analysis. Then they compared these estimates with participants’ actual height in the VA medical record, adjusting for age, sex, and other genetic characteristics.
In doing so, the researchers found 345 clinical traits that were associated with the actual measured height in White participants plus another 17 clinical trials linked to actual measured height in Black participants. An overall 127 of these clinical traits were significantly associated with White participants’ genetically predicted height, and two of them were significantly associated with Black participants’ genetically predicted height.
In analyzing all these data together, the researchers were largely able to separate out those associations between genetically predicted height and certain health conditions from those associations between health conditions and a person’s actual measured height. They also determined that including body mass index as a covariate had little impact on the results. The researchers conducted the appropriate statistical correction to ensure the use of so many variables did not result in spurious statistical significance in some associations.
“Using genetic methods applied to the VA Million Veteran Program, we found evidence that adult height may impact over 100 clinical traits, including several conditions associated with poor outcomes and quality of life – peripheral neuropathy, lower extremity ulcers, and chronic venous insufficiency. We conclude that height may be an unrecognized nonmodifiable risk factor for several common conditions in adults.”
Height linked with health conditions
Genetically predicted height predicted a reduced risk of hyperlipidemia and hypertension independent of coronary heart disease, the analysis revealed. Genetically predicted height was also linked to an approximately 51% increased risk of atrial fibrillation in participants without coronary heart disease but, paradoxically, only a 39% increased risk in those with coronary heart disease, despite coronary heart disease being a risk factor for atrial fibrillation. Genetically predicted height was also associated with a greater risk of varicose veins in the legs and deep vein thrombosis.
Another novel association uncovered by the analysis was between women’s genetically predicted height and both asthma and nonspecific peripheral nerve disorders. “Whether these associations reflect differences by sex in disease pathophysiology related to height may warrant exploration in a sample with better balance between men and women,” the authors wrote. “In sum, our results suggest that an individual’s height may warrant consideration as a nonmodifiable predictor for several common conditions, particularly those affecting peripheral/distal extremities that are most physically impacted by tall stature.”
A substantial limitation of the study was its homogeneity of participants, who were 92% male with an average height of 176 cm and an average BMI of 30.1. The Black participants tended to be younger, with an average age of 58 compared with 64 years in the White participants, but the groups were otherwise similar in height and weight.* The database included data from Hispanic participants, but the researchers excluded these data because of the small sample size.
The smaller dataset for Black participants was a limitation as well as the fact that the genome-wide association study the researchers relied on came from a European population, which may not be as accurate in people with other ancestry, Dr. Wilson said. The bigger limitation, however, is what the findings’ clinical relevance is.
What does it all mean?
“Genetic height is in your genes – there is nothing to be done about it – so it is more of academic interest than clinical interest,” Dr. Wilson said. It’s not even clear whether incorporating a person’s height – actual or genetically predicted, if it could be easily determined for each person – into risk calculators. ”To know whether it would be beneficial to use height (or genetic height) as a risk factor, you’d need to examine each condition of interest, adjusting for all known risk factors, to see if height improved the prediction,” Dr. Wilson said. “I suspect for most conditions, the well-known risk factors would swamp height. For example, high genetic height might truly increase risk for neuropathy. But diabetes might increase the risk so much more that height is not particularly relevant.”
On the other hand, the fact that height in general has any potential influence at all on disease risk may inspire physicians to consider other risk factors in especially tall individuals.
”Physicians may find it interesting that we have some confirmation that height does increase the risk of certain conditions,” Dr. Wilson said. “While this is unlikely to dramatically change practice, they may be a bit more diligent in looking for other relevant risk factors for the diseases found in this study in their very tall patients.”
The research was funded by the U.S. Department of Veteran Affairs, the Boettcher Foundation’s Webb-Waring Biomedical Research Program, the National Institutes of Health, and a Linda Pechenik Montague Investigator award. One study coauthor is a full-time employee of Novartis Institutes of Biomedical Research. The other authors and Dr. Wilson had no disclosures.
*Correction, 6/29/22: An earlier version of this article misstated the average age of Black participants.
FROM PLOS GENETICS