User login
Autism Acceptance Month: Raising awareness and closing the diagnosis gap
April is National Autism Awareness Month, and April 2 is World Autism Awareness Day. In the United States, there appears to be a heightened level of awareness of this condition over the past 10-15 years that has helped reduced its stigma, improve early identification, and (most importantly) increase access to early interventions for children and families.
The most recent prevalence estimates of autism in children in the United States is 1 in 54. This is a 10% increase since 2014 (1 in 59). Those most recent Centers for Disease Control and Prevention surveillance reports also point to a reduction in the racial gap between Black and White children when it comes to diagnosis.1 Across the globe, there are more than 100 autism societies, and research designed to improve prevalence data in lower- to middle-income countries has also increased.2
Even with these recent encouraging numbers regarding identification of autism in historically underrepresented groups in the United States, there are still differences among those groups, compared with children who are socioeconomically well-off, White, or live in large urban areas.3 Specifically, Latinx children were documented to be identified with autism at lower rates, compared with Whites and Blacks. In addition, Black and Latinx children are still diagnosed at a later age, compared with White children. This is important to note because historically, Black and Latinx children have been diagnosed with severe forms of autism or co-occurring intellectual disability at a higher rate, compared with their White counterparts.4 Thus, it would not be inappropriate to infer that Black and Latinx children with “milder” presenting autism symptoms or without co-occurring ID are not identified at the same rates, compared with their White peers. Furthermore, when peering into the international data, epidemiologic studies regarding prevalence, clinical course, and outcomes is skewed heavily toward a few Western industrialized nations, Japan, and South Korea.5
In all, when observing Autism Awareness Month, we should continue to recognize that these aforementioned epidemiologic disparities still exist – both locally and globally. because both clinical and research work have consequences that are not yet fully known. As long as these trends remain, racial and socioeconomic differences in access to treatment in the United States will remain. From an international perspective, we may never appreciate the true extent of the cultural variability within autism symptoms and so may never appreciate the full spectrum of ways the condition can present.
References
1. MMWR Surveill Summ. 2020 Mar 27;69(4):1-12. Erratum in: MMWR Morb Mortal Wkly Rep. 2020 Apr 24;69(16):503.
2. Lancet Glob Health. 2018 Oct;6(10):e1100-21.
3. Am J Public Health. 2009;99(3):493-8.
4. J Dev Behav Pediatr. 2011 Apr;32(3):179-87 and MMWR Surveill Summ. 2019;68(2):1-19.
5. Brain Sci. 2020;10(5):274. doi: 10.3390/brainsci10050274.
Dr. Emejuru is a child and adolescent psychiatrist with Community Hospital of Monterey Peninsula (CHOMP) and its Ohana Center for Child and Adolescent Behavioral Health in Monterey, Calif. His expertise is specific to conducting evaluations for autism spectrum disorder and evaluating, diagnosing, and treating co-occurring psychiatric disorders after training at the Johns Hopkins Hospital/Kennedy Krieger Institute’s Center for Autism and Related Disorders in Baltimore. He has no conflicts of interest.
April is National Autism Awareness Month, and April 2 is World Autism Awareness Day. In the United States, there appears to be a heightened level of awareness of this condition over the past 10-15 years that has helped reduced its stigma, improve early identification, and (most importantly) increase access to early interventions for children and families.
The most recent prevalence estimates of autism in children in the United States is 1 in 54. This is a 10% increase since 2014 (1 in 59). Those most recent Centers for Disease Control and Prevention surveillance reports also point to a reduction in the racial gap between Black and White children when it comes to diagnosis.1 Across the globe, there are more than 100 autism societies, and research designed to improve prevalence data in lower- to middle-income countries has also increased.2
Even with these recent encouraging numbers regarding identification of autism in historically underrepresented groups in the United States, there are still differences among those groups, compared with children who are socioeconomically well-off, White, or live in large urban areas.3 Specifically, Latinx children were documented to be identified with autism at lower rates, compared with Whites and Blacks. In addition, Black and Latinx children are still diagnosed at a later age, compared with White children. This is important to note because historically, Black and Latinx children have been diagnosed with severe forms of autism or co-occurring intellectual disability at a higher rate, compared with their White counterparts.4 Thus, it would not be inappropriate to infer that Black and Latinx children with “milder” presenting autism symptoms or without co-occurring ID are not identified at the same rates, compared with their White peers. Furthermore, when peering into the international data, epidemiologic studies regarding prevalence, clinical course, and outcomes is skewed heavily toward a few Western industrialized nations, Japan, and South Korea.5
In all, when observing Autism Awareness Month, we should continue to recognize that these aforementioned epidemiologic disparities still exist – both locally and globally. because both clinical and research work have consequences that are not yet fully known. As long as these trends remain, racial and socioeconomic differences in access to treatment in the United States will remain. From an international perspective, we may never appreciate the true extent of the cultural variability within autism symptoms and so may never appreciate the full spectrum of ways the condition can present.
References
1. MMWR Surveill Summ. 2020 Mar 27;69(4):1-12. Erratum in: MMWR Morb Mortal Wkly Rep. 2020 Apr 24;69(16):503.
2. Lancet Glob Health. 2018 Oct;6(10):e1100-21.
3. Am J Public Health. 2009;99(3):493-8.
4. J Dev Behav Pediatr. 2011 Apr;32(3):179-87 and MMWR Surveill Summ. 2019;68(2):1-19.
5. Brain Sci. 2020;10(5):274. doi: 10.3390/brainsci10050274.
Dr. Emejuru is a child and adolescent psychiatrist with Community Hospital of Monterey Peninsula (CHOMP) and its Ohana Center for Child and Adolescent Behavioral Health in Monterey, Calif. His expertise is specific to conducting evaluations for autism spectrum disorder and evaluating, diagnosing, and treating co-occurring psychiatric disorders after training at the Johns Hopkins Hospital/Kennedy Krieger Institute’s Center for Autism and Related Disorders in Baltimore. He has no conflicts of interest.
April is National Autism Awareness Month, and April 2 is World Autism Awareness Day. In the United States, there appears to be a heightened level of awareness of this condition over the past 10-15 years that has helped reduced its stigma, improve early identification, and (most importantly) increase access to early interventions for children and families.
The most recent prevalence estimates of autism in children in the United States is 1 in 54. This is a 10% increase since 2014 (1 in 59). Those most recent Centers for Disease Control and Prevention surveillance reports also point to a reduction in the racial gap between Black and White children when it comes to diagnosis.1 Across the globe, there are more than 100 autism societies, and research designed to improve prevalence data in lower- to middle-income countries has also increased.2
Even with these recent encouraging numbers regarding identification of autism in historically underrepresented groups in the United States, there are still differences among those groups, compared with children who are socioeconomically well-off, White, or live in large urban areas.3 Specifically, Latinx children were documented to be identified with autism at lower rates, compared with Whites and Blacks. In addition, Black and Latinx children are still diagnosed at a later age, compared with White children. This is important to note because historically, Black and Latinx children have been diagnosed with severe forms of autism or co-occurring intellectual disability at a higher rate, compared with their White counterparts.4 Thus, it would not be inappropriate to infer that Black and Latinx children with “milder” presenting autism symptoms or without co-occurring ID are not identified at the same rates, compared with their White peers. Furthermore, when peering into the international data, epidemiologic studies regarding prevalence, clinical course, and outcomes is skewed heavily toward a few Western industrialized nations, Japan, and South Korea.5
In all, when observing Autism Awareness Month, we should continue to recognize that these aforementioned epidemiologic disparities still exist – both locally and globally. because both clinical and research work have consequences that are not yet fully known. As long as these trends remain, racial and socioeconomic differences in access to treatment in the United States will remain. From an international perspective, we may never appreciate the true extent of the cultural variability within autism symptoms and so may never appreciate the full spectrum of ways the condition can present.
References
1. MMWR Surveill Summ. 2020 Mar 27;69(4):1-12. Erratum in: MMWR Morb Mortal Wkly Rep. 2020 Apr 24;69(16):503.
2. Lancet Glob Health. 2018 Oct;6(10):e1100-21.
3. Am J Public Health. 2009;99(3):493-8.
4. J Dev Behav Pediatr. 2011 Apr;32(3):179-87 and MMWR Surveill Summ. 2019;68(2):1-19.
5. Brain Sci. 2020;10(5):274. doi: 10.3390/brainsci10050274.
Dr. Emejuru is a child and adolescent psychiatrist with Community Hospital of Monterey Peninsula (CHOMP) and its Ohana Center for Child and Adolescent Behavioral Health in Monterey, Calif. His expertise is specific to conducting evaluations for autism spectrum disorder and evaluating, diagnosing, and treating co-occurring psychiatric disorders after training at the Johns Hopkins Hospital/Kennedy Krieger Institute’s Center for Autism and Related Disorders in Baltimore. He has no conflicts of interest.
Incontinentia Pigmenti: Initial Presentation of Encephalopathy and Seizures
To the Editor:
A 7-day-old full-term infant presented to the neonatal intensive care unit with poor feeding and altered consciousness. She was born at 39 weeks and 3 days to a gravida 1 mother with a pregnancy history complicated by maternal chorioamnionitis and gestational diabetes. During labor, nonreassuring fetal heart tones and arrest of labor prompted an uncomplicated cesarean delivery with normal Apgar scores at birth. The infant’s family history revealed only beta thalassemia minor in her father. At 5 to 7 days of life, the mother noted difficulty with feeding and poor latch along with lethargy and depressed consciousness in the infant.
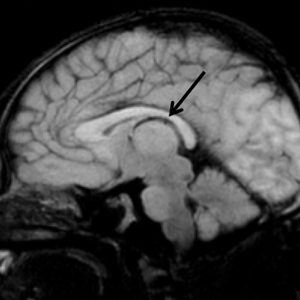
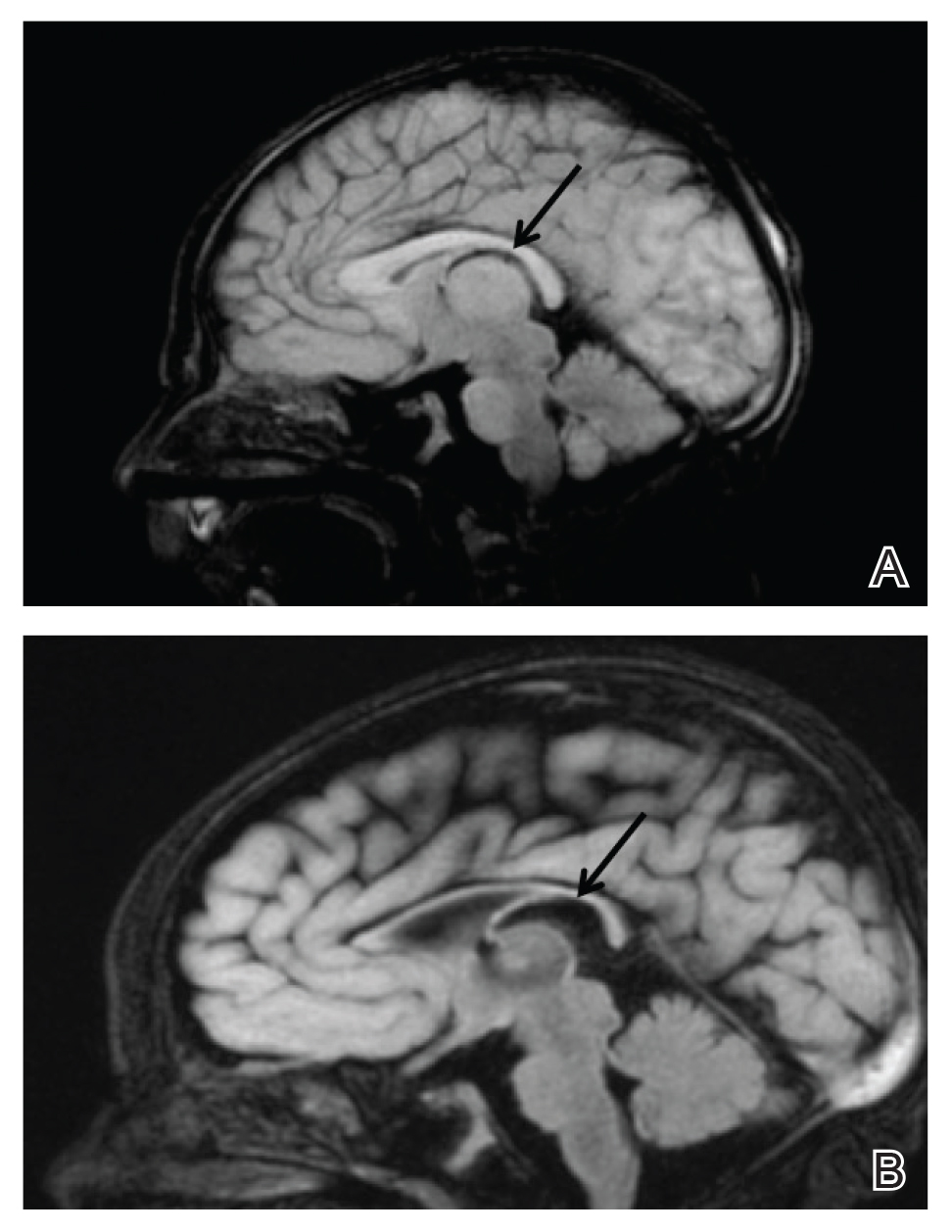
Upon arrival to the neonatal intensive care unit, the infant was noted to have rhythmic lip-smacking behavior, intermittent nystagmus, mild hypotonia, and clonic movements of the left upper extremity. An electroencephalogram was markedly abnormal, capturing multiple seizures in the bilateral cortical hemispheres. She was loaded with phenobarbital with no further seizure activity. Brain magnetic resonance imaging revealed innumerable punctate foci of restricted diffusion with corresponding punctate hemorrhage within the frontal and parietal white matter, as well as cortical diffusion restriction within the occipital lobe, inferior temporal lobe, bilateral thalami, and corpus callosum (Figure 1). An exhaustive infectious workup also was completed and was unremarkable, though she was treated with broad-spectrum antimicrobials, including intravenous acyclovir.
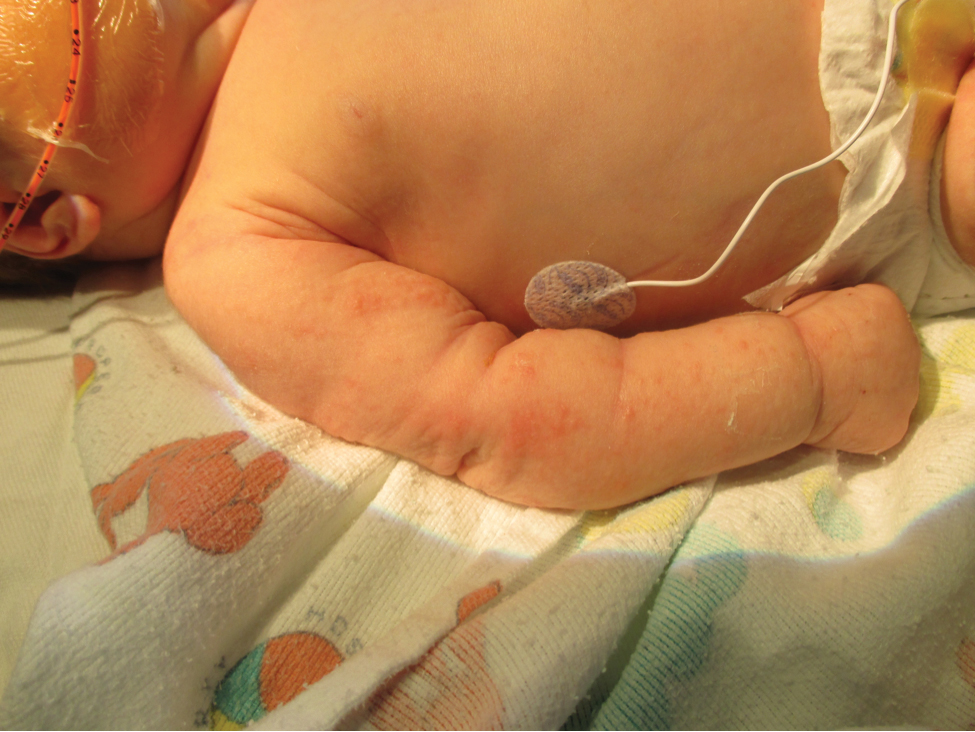
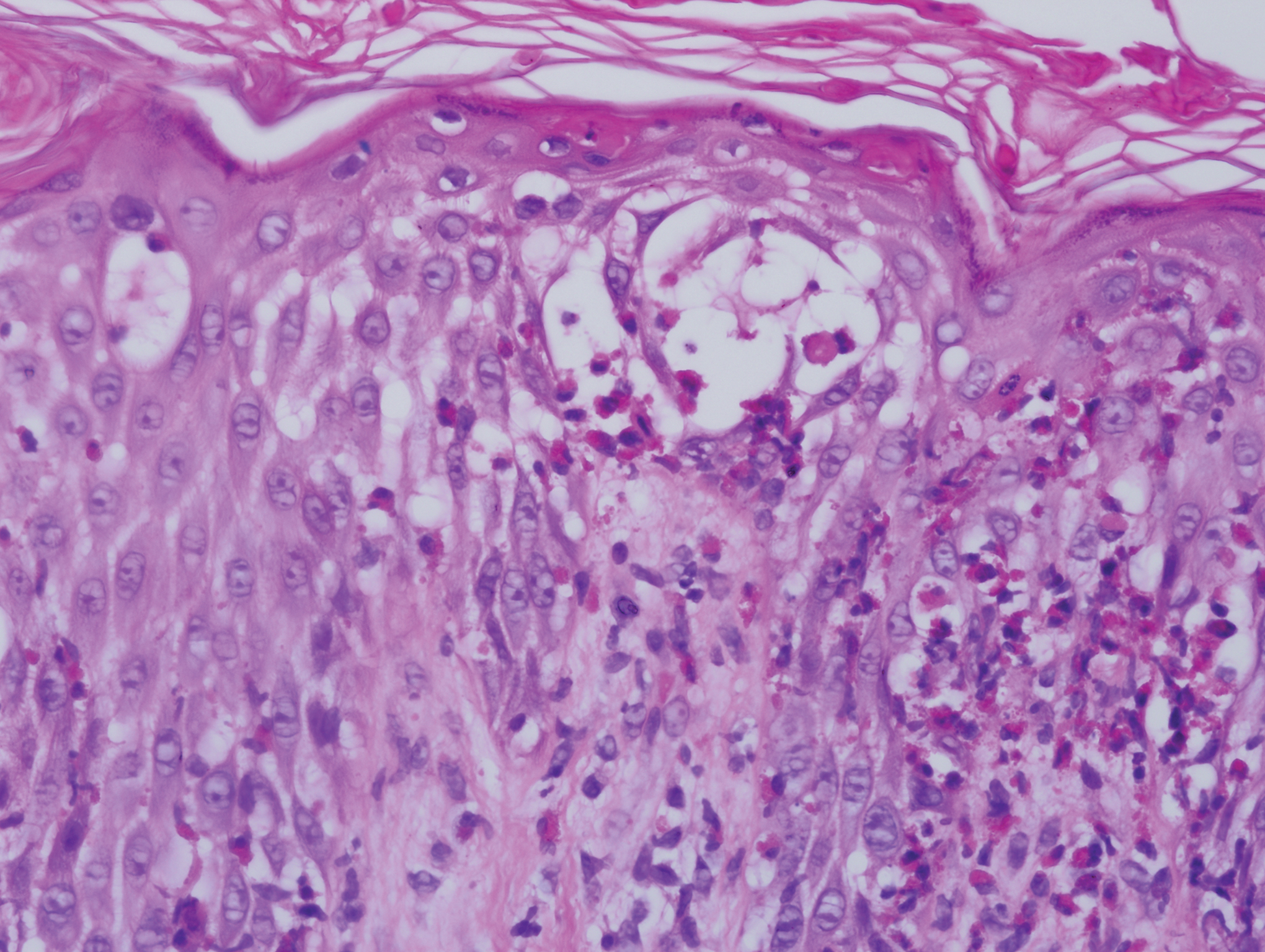
Five days after being hospitalized (day 10 of life), a vesicular rash was noted on the arms and legs (Figure 2). Discussion with the patient’s mother revealed that the first signs of unusual skin lesions occurred as early as several days prior. There were no oral mucosal lesions or gross ocular abnormalities. No nail changes were appreciated. A bedside Tzanck preparation was negative for viral cytopathic changes. A skin biopsy was performed that demonstrated eosinophilic spongiosis with necrotic keratinocytes, typical of the vesicular stage of incontinentia pigmenti (IP)(Figure 3). An ophthalmology examination showed an arteriovenous malformation of the right eye with subtle neovascularization at the infratemporal periphery, consistent with known ocular manifestations of IP. The infant’s mother reported no history of notable dental abnormalities, hair loss, skin rashes, or nail changes. Genetic testing demonstrated the common IKBKG (inhibitor of κ light polypeptide gene enhancer in B cells, kinase gamma [formerly known as NEMO]) gene deletion on the X chromosome, consistent with IP.
She successfully underwent retinal laser ablative therapy for the ocular manifestations without further evidence of neovascularization. She developed a mild cataract that was not visually significant and required no intervention. Her brain abnormalities were thought to represent foci of necrosis with superimposed hemorrhagic transformation due to spontaneous degeneration of brain cells in which the mutated X chromosome was activated. No further treatment was indicated beyond suppression of the consequent seizures. There was no notable cortical edema or other medical indication for systemic glucocorticoid therapy. Phenobarbital was continued without further seizure events.
Several months after the initial presentation, a follow-up electroencephalogram was normal. Phenobarbital was slowly weaned and finally discontinued approximately 6 months after the initial event with no other reported seizures. She currently is achieving normal developmental milestones with the exception of slight motor delay and expected residual hypotonia.
Incontinentia pigmenti, also known as Bloch-Sulzberger syndrome, is a rare multisystem neuroectodermal disorder, primarily affecting the skin, central nervous system (CNS), and retinas. The disorder can be inherited in an X-linked dominant fashion and appears almost exclusively in women with typical in utero lethality seen in males. Most affected individuals have a sporadic, or de novo, mutation, which was likely the case in our patient given that her mother demonstrated no signs or symptoms.1 The pathogenesis of disease is a defect at chromosome Xq28 that is a region encoding the nuclear factor–κB essential modulator, IKBKG. Absence or mutation of IKBKG in IP results in failure to activate nuclear factor–κB and leaves cells vulnerable to cytokine-mediated apoptosis, especially after exposure to tumor necrosis factor α.2
Clinical manifestations of IP are present at or soon after birth. The cutaneous findings of this disorder are classically described as a step-wise progression through 4 distinct stages: (1) a linear and/or whorled vesicular eruption predominantly on the extremities at birth or within the first few weeks of life; (2) thickened linear or whorled verrucous plaques; (3) hyperpigmented streaks and whorls that may or may not correspond with prior affected areas that may resolve by adolescence; and (4) hypopigmented, possibly atrophic plaques on the extremities that may persist lifelong. Importantly, not every patient will experience each of these stages. Overlap can occur, and the time course of each stage is highly variable. Other ectodermal manifestations include dental abnormalities such as small, misshaped, or missing teeth; alopecia; and nail abnormalities. Ocular abnormalities associated with IP primarily occur in the retina, including vascular occlusion, neovascularization, hemorrhages, foveal abnormalities, as well as exudative and tractional detachments.3,4
It is crucial to recognize CNS anomalies in association with the cutaneous findings of IP, as CNS pathology can be severe with profound developmental implications. Central nervous system findings have been noted to correlate with the appearance of the vesicular stage of IP. A high index of suspicion is needed, as the disease can demonstrate progression within a short time.5-8 The most frequent anomalies include seizures, motor impairment, intellectual disability, and microcephaly.9,10 Some of the most commonly identified CNS lesions on imaging include necrosis or brain infarcts, atrophy, and lesions of the corpus callosum.7
The pathogenesis of observed CNS changes in IP is not well understood. There have been numerous proposals of a vascular mechanism, and a microangiopathic process appears to be most plausible. Mutations in IKBKG may result in interruption of signaling via vascular endothelial growth factor receptor 3 with a consequent impact on angiogenesis, supporting a vascular mechanism. Additionally, mutations in IKBKG lead to activation of eotaxin, an eosinophil-selective chemokine.9 Eotaxin activation results in eosinophilic degranulation that mediates the classic eosinophilic infiltrate seen in the classic skin histology of IP. Additionally, it has been shown that eotaxin is strongly expressed by endothelial cells in IP, and more abundant eosinophil degranulation may play a role in mediating vaso-occlusion.7 Other studies have found that the highest expression level of the IKBKG gene is in the CNS, potentially explaining the extensive imaging findings of hemorrhage and diffusion restriction in our patient. These features likely are attributable to apoptosis of cells possessing the mutated IKBKG gene.9-11
- Ehrenreich M, Tarlow MM, Godlewska-Janusz E, et al. Incontinentia pigmenti (Bloch-Sulzberger syndrome): a systemic disorder. Cutis. 2007;79:355-362.
- Smahi A, Courtois G, Rabia SH, et al. The NF-kappaB signaling pathway in human diseases: from incontinentia pigmenti to ectodermal dysplasias and immune-deficiency syndromes. Hum Mol Genet. 2002;11:2371-2375.
- O’Doherty M, McCreery K, Green AJ, et al. Incontinentia pigmenti—ophthalmological observation of a series of cases and review of the literature. Br J Ophthalmol. 2011;95:11-16.
- Swinney CC, Han DP, Karth PA. Incontinentia pigmenti: a comprehensive review and update. Ophthalmic Surg Lasers Imaging Retina. 2015;46:650-657.
- Hennel SJ, Ekert PG, Volpe JJ, et al. Insights into the pathogenesis of cerebral lesions in incontinentia pigmenti. Pediatr Neurol. 2003;29:148-150.
- Maingay-de Groof F, Lequin MH, Roofthooft DW, et al. Extensive cerebral infarction in the newborn due to incontinentia pigmenti. Eur J Paediatr Neurol. 2008;12:284-289.
- Minic´ S, Trpinac D, Obradovic´ M. Systematic review of central nervous system anomalies in incontinentia pigmenti. Orphanet J Rare Dis. 2013;8:25-35.
- Wolf NI, Kramer N, Harting I, et al. Diffuse cortical necrosis in a neonate with incontinentia pigmenti and an encephalitis-like presentation. AJNR Am J Neuroradiol. 2005;26:1580-1582.
- Phan TA, Wargon O, Turner AM. Incontinentia pigmenti case series: clinical spectrum of incontinentia pigmenti in 53 female patients and their relatives. Clin Exp Dermatol. 2005;30:474-480.
- Volpe J. Neurobiology of periventricular leukomalacia in the premature infant. Pediatr Res. 2001;50:553-562.
- Pascual-Castroviejo I, Pascual-Pascual SI, Velazquez-Fragua R, et al. Incontinentia pigmenti: clinical and neuroimaging findings in a series of 12 patients. Neurologia. 2006;21:239-248.
To the Editor:
A 7-day-old full-term infant presented to the neonatal intensive care unit with poor feeding and altered consciousness. She was born at 39 weeks and 3 days to a gravida 1 mother with a pregnancy history complicated by maternal chorioamnionitis and gestational diabetes. During labor, nonreassuring fetal heart tones and arrest of labor prompted an uncomplicated cesarean delivery with normal Apgar scores at birth. The infant’s family history revealed only beta thalassemia minor in her father. At 5 to 7 days of life, the mother noted difficulty with feeding and poor latch along with lethargy and depressed consciousness in the infant.
Upon arrival to the neonatal intensive care unit, the infant was noted to have rhythmic lip-smacking behavior, intermittent nystagmus, mild hypotonia, and clonic movements of the left upper extremity. An electroencephalogram was markedly abnormal, capturing multiple seizures in the bilateral cortical hemispheres. She was loaded with phenobarbital with no further seizure activity. Brain magnetic resonance imaging revealed innumerable punctate foci of restricted diffusion with corresponding punctate hemorrhage within the frontal and parietal white matter, as well as cortical diffusion restriction within the occipital lobe, inferior temporal lobe, bilateral thalami, and corpus callosum (Figure 1). An exhaustive infectious workup also was completed and was unremarkable, though she was treated with broad-spectrum antimicrobials, including intravenous acyclovir.

Five days after being hospitalized (day 10 of life), a vesicular rash was noted on the arms and legs (Figure 2). Discussion with the patient’s mother revealed that the first signs of unusual skin lesions occurred as early as several days prior. There were no oral mucosal lesions or gross ocular abnormalities. No nail changes were appreciated. A bedside Tzanck preparation was negative for viral cytopathic changes. A skin biopsy was performed that demonstrated eosinophilic spongiosis with necrotic keratinocytes, typical of the vesicular stage of incontinentia pigmenti (IP)(Figure 3). An ophthalmology examination showed an arteriovenous malformation of the right eye with subtle neovascularization at the infratemporal periphery, consistent with known ocular manifestations of IP. The infant’s mother reported no history of notable dental abnormalities, hair loss, skin rashes, or nail changes. Genetic testing demonstrated the common IKBKG (inhibitor of κ light polypeptide gene enhancer in B cells, kinase gamma [formerly known as NEMO]) gene deletion on the X chromosome, consistent with IP.


She successfully underwent retinal laser ablative therapy for the ocular manifestations without further evidence of neovascularization. She developed a mild cataract that was not visually significant and required no intervention. Her brain abnormalities were thought to represent foci of necrosis with superimposed hemorrhagic transformation due to spontaneous degeneration of brain cells in which the mutated X chromosome was activated. No further treatment was indicated beyond suppression of the consequent seizures. There was no notable cortical edema or other medical indication for systemic glucocorticoid therapy. Phenobarbital was continued without further seizure events.
Several months after the initial presentation, a follow-up electroencephalogram was normal. Phenobarbital was slowly weaned and finally discontinued approximately 6 months after the initial event with no other reported seizures. She currently is achieving normal developmental milestones with the exception of slight motor delay and expected residual hypotonia.
Incontinentia pigmenti, also known as Bloch-Sulzberger syndrome, is a rare multisystem neuroectodermal disorder, primarily affecting the skin, central nervous system (CNS), and retinas. The disorder can be inherited in an X-linked dominant fashion and appears almost exclusively in women with typical in utero lethality seen in males. Most affected individuals have a sporadic, or de novo, mutation, which was likely the case in our patient given that her mother demonstrated no signs or symptoms.1 The pathogenesis of disease is a defect at chromosome Xq28 that is a region encoding the nuclear factor–κB essential modulator, IKBKG. Absence or mutation of IKBKG in IP results in failure to activate nuclear factor–κB and leaves cells vulnerable to cytokine-mediated apoptosis, especially after exposure to tumor necrosis factor α.2
Clinical manifestations of IP are present at or soon after birth. The cutaneous findings of this disorder are classically described as a step-wise progression through 4 distinct stages: (1) a linear and/or whorled vesicular eruption predominantly on the extremities at birth or within the first few weeks of life; (2) thickened linear or whorled verrucous plaques; (3) hyperpigmented streaks and whorls that may or may not correspond with prior affected areas that may resolve by adolescence; and (4) hypopigmented, possibly atrophic plaques on the extremities that may persist lifelong. Importantly, not every patient will experience each of these stages. Overlap can occur, and the time course of each stage is highly variable. Other ectodermal manifestations include dental abnormalities such as small, misshaped, or missing teeth; alopecia; and nail abnormalities. Ocular abnormalities associated with IP primarily occur in the retina, including vascular occlusion, neovascularization, hemorrhages, foveal abnormalities, as well as exudative and tractional detachments.3,4
It is crucial to recognize CNS anomalies in association with the cutaneous findings of IP, as CNS pathology can be severe with profound developmental implications. Central nervous system findings have been noted to correlate with the appearance of the vesicular stage of IP. A high index of suspicion is needed, as the disease can demonstrate progression within a short time.5-8 The most frequent anomalies include seizures, motor impairment, intellectual disability, and microcephaly.9,10 Some of the most commonly identified CNS lesions on imaging include necrosis or brain infarcts, atrophy, and lesions of the corpus callosum.7
The pathogenesis of observed CNS changes in IP is not well understood. There have been numerous proposals of a vascular mechanism, and a microangiopathic process appears to be most plausible. Mutations in IKBKG may result in interruption of signaling via vascular endothelial growth factor receptor 3 with a consequent impact on angiogenesis, supporting a vascular mechanism. Additionally, mutations in IKBKG lead to activation of eotaxin, an eosinophil-selective chemokine.9 Eotaxin activation results in eosinophilic degranulation that mediates the classic eosinophilic infiltrate seen in the classic skin histology of IP. Additionally, it has been shown that eotaxin is strongly expressed by endothelial cells in IP, and more abundant eosinophil degranulation may play a role in mediating vaso-occlusion.7 Other studies have found that the highest expression level of the IKBKG gene is in the CNS, potentially explaining the extensive imaging findings of hemorrhage and diffusion restriction in our patient. These features likely are attributable to apoptosis of cells possessing the mutated IKBKG gene.9-11
To the Editor:
A 7-day-old full-term infant presented to the neonatal intensive care unit with poor feeding and altered consciousness. She was born at 39 weeks and 3 days to a gravida 1 mother with a pregnancy history complicated by maternal chorioamnionitis and gestational diabetes. During labor, nonreassuring fetal heart tones and arrest of labor prompted an uncomplicated cesarean delivery with normal Apgar scores at birth. The infant’s family history revealed only beta thalassemia minor in her father. At 5 to 7 days of life, the mother noted difficulty with feeding and poor latch along with lethargy and depressed consciousness in the infant.
Upon arrival to the neonatal intensive care unit, the infant was noted to have rhythmic lip-smacking behavior, intermittent nystagmus, mild hypotonia, and clonic movements of the left upper extremity. An electroencephalogram was markedly abnormal, capturing multiple seizures in the bilateral cortical hemispheres. She was loaded with phenobarbital with no further seizure activity. Brain magnetic resonance imaging revealed innumerable punctate foci of restricted diffusion with corresponding punctate hemorrhage within the frontal and parietal white matter, as well as cortical diffusion restriction within the occipital lobe, inferior temporal lobe, bilateral thalami, and corpus callosum (Figure 1). An exhaustive infectious workup also was completed and was unremarkable, though she was treated with broad-spectrum antimicrobials, including intravenous acyclovir.

Five days after being hospitalized (day 10 of life), a vesicular rash was noted on the arms and legs (Figure 2). Discussion with the patient’s mother revealed that the first signs of unusual skin lesions occurred as early as several days prior. There were no oral mucosal lesions or gross ocular abnormalities. No nail changes were appreciated. A bedside Tzanck preparation was negative for viral cytopathic changes. A skin biopsy was performed that demonstrated eosinophilic spongiosis with necrotic keratinocytes, typical of the vesicular stage of incontinentia pigmenti (IP)(Figure 3). An ophthalmology examination showed an arteriovenous malformation of the right eye with subtle neovascularization at the infratemporal periphery, consistent with known ocular manifestations of IP. The infant’s mother reported no history of notable dental abnormalities, hair loss, skin rashes, or nail changes. Genetic testing demonstrated the common IKBKG (inhibitor of κ light polypeptide gene enhancer in B cells, kinase gamma [formerly known as NEMO]) gene deletion on the X chromosome, consistent with IP.


She successfully underwent retinal laser ablative therapy for the ocular manifestations without further evidence of neovascularization. She developed a mild cataract that was not visually significant and required no intervention. Her brain abnormalities were thought to represent foci of necrosis with superimposed hemorrhagic transformation due to spontaneous degeneration of brain cells in which the mutated X chromosome was activated. No further treatment was indicated beyond suppression of the consequent seizures. There was no notable cortical edema or other medical indication for systemic glucocorticoid therapy. Phenobarbital was continued without further seizure events.
Several months after the initial presentation, a follow-up electroencephalogram was normal. Phenobarbital was slowly weaned and finally discontinued approximately 6 months after the initial event with no other reported seizures. She currently is achieving normal developmental milestones with the exception of slight motor delay and expected residual hypotonia.
Incontinentia pigmenti, also known as Bloch-Sulzberger syndrome, is a rare multisystem neuroectodermal disorder, primarily affecting the skin, central nervous system (CNS), and retinas. The disorder can be inherited in an X-linked dominant fashion and appears almost exclusively in women with typical in utero lethality seen in males. Most affected individuals have a sporadic, or de novo, mutation, which was likely the case in our patient given that her mother demonstrated no signs or symptoms.1 The pathogenesis of disease is a defect at chromosome Xq28 that is a region encoding the nuclear factor–κB essential modulator, IKBKG. Absence or mutation of IKBKG in IP results in failure to activate nuclear factor–κB and leaves cells vulnerable to cytokine-mediated apoptosis, especially after exposure to tumor necrosis factor α.2
Clinical manifestations of IP are present at or soon after birth. The cutaneous findings of this disorder are classically described as a step-wise progression through 4 distinct stages: (1) a linear and/or whorled vesicular eruption predominantly on the extremities at birth or within the first few weeks of life; (2) thickened linear or whorled verrucous plaques; (3) hyperpigmented streaks and whorls that may or may not correspond with prior affected areas that may resolve by adolescence; and (4) hypopigmented, possibly atrophic plaques on the extremities that may persist lifelong. Importantly, not every patient will experience each of these stages. Overlap can occur, and the time course of each stage is highly variable. Other ectodermal manifestations include dental abnormalities such as small, misshaped, or missing teeth; alopecia; and nail abnormalities. Ocular abnormalities associated with IP primarily occur in the retina, including vascular occlusion, neovascularization, hemorrhages, foveal abnormalities, as well as exudative and tractional detachments.3,4
It is crucial to recognize CNS anomalies in association with the cutaneous findings of IP, as CNS pathology can be severe with profound developmental implications. Central nervous system findings have been noted to correlate with the appearance of the vesicular stage of IP. A high index of suspicion is needed, as the disease can demonstrate progression within a short time.5-8 The most frequent anomalies include seizures, motor impairment, intellectual disability, and microcephaly.9,10 Some of the most commonly identified CNS lesions on imaging include necrosis or brain infarcts, atrophy, and lesions of the corpus callosum.7
The pathogenesis of observed CNS changes in IP is not well understood. There have been numerous proposals of a vascular mechanism, and a microangiopathic process appears to be most plausible. Mutations in IKBKG may result in interruption of signaling via vascular endothelial growth factor receptor 3 with a consequent impact on angiogenesis, supporting a vascular mechanism. Additionally, mutations in IKBKG lead to activation of eotaxin, an eosinophil-selective chemokine.9 Eotaxin activation results in eosinophilic degranulation that mediates the classic eosinophilic infiltrate seen in the classic skin histology of IP. Additionally, it has been shown that eotaxin is strongly expressed by endothelial cells in IP, and more abundant eosinophil degranulation may play a role in mediating vaso-occlusion.7 Other studies have found that the highest expression level of the IKBKG gene is in the CNS, potentially explaining the extensive imaging findings of hemorrhage and diffusion restriction in our patient. These features likely are attributable to apoptosis of cells possessing the mutated IKBKG gene.9-11
- Ehrenreich M, Tarlow MM, Godlewska-Janusz E, et al. Incontinentia pigmenti (Bloch-Sulzberger syndrome): a systemic disorder. Cutis. 2007;79:355-362.
- Smahi A, Courtois G, Rabia SH, et al. The NF-kappaB signaling pathway in human diseases: from incontinentia pigmenti to ectodermal dysplasias and immune-deficiency syndromes. Hum Mol Genet. 2002;11:2371-2375.
- O’Doherty M, McCreery K, Green AJ, et al. Incontinentia pigmenti—ophthalmological observation of a series of cases and review of the literature. Br J Ophthalmol. 2011;95:11-16.
- Swinney CC, Han DP, Karth PA. Incontinentia pigmenti: a comprehensive review and update. Ophthalmic Surg Lasers Imaging Retina. 2015;46:650-657.
- Hennel SJ, Ekert PG, Volpe JJ, et al. Insights into the pathogenesis of cerebral lesions in incontinentia pigmenti. Pediatr Neurol. 2003;29:148-150.
- Maingay-de Groof F, Lequin MH, Roofthooft DW, et al. Extensive cerebral infarction in the newborn due to incontinentia pigmenti. Eur J Paediatr Neurol. 2008;12:284-289.
- Minic´ S, Trpinac D, Obradovic´ M. Systematic review of central nervous system anomalies in incontinentia pigmenti. Orphanet J Rare Dis. 2013;8:25-35.
- Wolf NI, Kramer N, Harting I, et al. Diffuse cortical necrosis in a neonate with incontinentia pigmenti and an encephalitis-like presentation. AJNR Am J Neuroradiol. 2005;26:1580-1582.
- Phan TA, Wargon O, Turner AM. Incontinentia pigmenti case series: clinical spectrum of incontinentia pigmenti in 53 female patients and their relatives. Clin Exp Dermatol. 2005;30:474-480.
- Volpe J. Neurobiology of periventricular leukomalacia in the premature infant. Pediatr Res. 2001;50:553-562.
- Pascual-Castroviejo I, Pascual-Pascual SI, Velazquez-Fragua R, et al. Incontinentia pigmenti: clinical and neuroimaging findings in a series of 12 patients. Neurologia. 2006;21:239-248.
- Ehrenreich M, Tarlow MM, Godlewska-Janusz E, et al. Incontinentia pigmenti (Bloch-Sulzberger syndrome): a systemic disorder. Cutis. 2007;79:355-362.
- Smahi A, Courtois G, Rabia SH, et al. The NF-kappaB signaling pathway in human diseases: from incontinentia pigmenti to ectodermal dysplasias and immune-deficiency syndromes. Hum Mol Genet. 2002;11:2371-2375.
- O’Doherty M, McCreery K, Green AJ, et al. Incontinentia pigmenti—ophthalmological observation of a series of cases and review of the literature. Br J Ophthalmol. 2011;95:11-16.
- Swinney CC, Han DP, Karth PA. Incontinentia pigmenti: a comprehensive review and update. Ophthalmic Surg Lasers Imaging Retina. 2015;46:650-657.
- Hennel SJ, Ekert PG, Volpe JJ, et al. Insights into the pathogenesis of cerebral lesions in incontinentia pigmenti. Pediatr Neurol. 2003;29:148-150.
- Maingay-de Groof F, Lequin MH, Roofthooft DW, et al. Extensive cerebral infarction in the newborn due to incontinentia pigmenti. Eur J Paediatr Neurol. 2008;12:284-289.
- Minic´ S, Trpinac D, Obradovic´ M. Systematic review of central nervous system anomalies in incontinentia pigmenti. Orphanet J Rare Dis. 2013;8:25-35.
- Wolf NI, Kramer N, Harting I, et al. Diffuse cortical necrosis in a neonate with incontinentia pigmenti and an encephalitis-like presentation. AJNR Am J Neuroradiol. 2005;26:1580-1582.
- Phan TA, Wargon O, Turner AM. Incontinentia pigmenti case series: clinical spectrum of incontinentia pigmenti in 53 female patients and their relatives. Clin Exp Dermatol. 2005;30:474-480.
- Volpe J. Neurobiology of periventricular leukomalacia in the premature infant. Pediatr Res. 2001;50:553-562.
- Pascual-Castroviejo I, Pascual-Pascual SI, Velazquez-Fragua R, et al. Incontinentia pigmenti: clinical and neuroimaging findings in a series of 12 patients. Neurologia. 2006;21:239-248.
Practice Points
- Central nervous system involvement in incontinentia pigmenti (IP) may be profound and can present prior to the classic cutaneous findings.
- A high index of suspicion for IP should be maintained in neonatal vesicular eruptions of unclear etiology, especially in the setting of unexplained seizures and/or abnormal brain imaging.
‘Striking’ increase in childhood obesity during pandemic
Obesity rates among children jumped substantially in the first months of the COVID-19 pandemic, according to a study published online in Pediatrics. Experts worry the excess weight will be a continuing problem for these children.
“Across the board in the span of a year, there has been a 2% increase in obesity, which is really striking,” lead author Brian P. Jenssen, MD, said in an interview.
The prevalence of obesity in a large pediatric primary care network increased from 13.7% to 15.4%.
Preexisting disparities by race or ethnicity and socioeconomic status worsened, noted Dr. Jenssen, a primary care pediatrician affiliated with Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania, Philadelphia.
Dr. Jenssen and colleagues compared the average obesity rate from June to December 2020 with the rate from June to December 2019 among patients in the CHOP Care Network, which includes 29 urban, suburban, and semirural clinics in the Philadelphia region. In June 2020, the volume of patient visits “returned to near-normal” after a dramatic decline in March 2020, the study authors wrote.
The investigators examined body mass index at all visits for patients aged 2-17 years for whom height and weight were documented. Patients with a BMI at or above the 95th percentile were classified as obese. The analysis included approximately 169,000 visits in 2019 and about 145,000 in 2020.
The average age of the patients was 9.2 years, and 48.9% were girls. In all, 21.4% were non-Hispanic Black, and about 30% were publicly insured.
Increases in obesity rates were more pronounced among patients aged 5-9 years and among patients who were Hispanic/Latino, non-Hispanic Black, publicly insured, or from lower-income neighborhoods.
Whereas the obesity rate increased 1% for patients aged 13-17 years, the rate increased 2.6% for patients aged 5-9 years.
Nearly 25% of Hispanic/Latino or non-Hispanic Black patients seen during the pandemic were obese, compared with 11.3% of non-Hispanic White patients. Before the pandemic, differences by race or ethnicity had been about 10%-11%.
Limiting the analysis to preventive visits did not meaningfully change the results, wrote Dr. Jenssen and colleagues.
“Having any increase in the obesity rates is alarming,” said Sandra Hassink, MD, medical director for the American Academy of Pediatrics’ (AAP’s) Institute for Healthy Childhood Weight. “I think what we’re seeing is what we feared.”
Before the pandemic, children received appropriately portioned breakfasts and lunches at school, but during the pandemic, they had less access to such meals, the academy noted. Disruptions to schooling, easier access to unhealthy snacks, increased screen time, and economic issues such as parents’ job losses were further factors, Hassink said.
Tackling the weight gain
In December 2020, the AAP issued two clinical guidance documents to highlight the importance of addressing obesity during the pandemic. Recommendations included physician counseling of families about maintaining healthy nutrition, minimizing sedentary time, and getting enough sleep and physical activity, as well as the assessment of all patients for onset of obesity and the maintenance of obesity treatment for patients with obesity.
In addition to clinical assessments and guidance, Dr. Jenssen emphasized that a return to routines may be crucial. Prepandemic studies have shown that many children, especially those insured by Medicaid, gain more weight during the summer when they are out of school, he noted. Many of the same factors are present during the pandemic, he said.
“One solution, and probably the most important solution, is getting kids back in school,” Dr. Jenssen said. School disruptions also have affected children’s learning and mental health, but those effects may be harder to quantify than BMI, he said.
Dr. Jenssen suggests that parents do their best to model good routines and habits. For example, they might decide that they and their children will stop drinking soda as a family, or opt for an apple instead of a bag of chips. They can walk around the house or up and down stairs when talking. “Those sorts of little things can make a big difference in the long run,” Dr. Jenssen said.
Clinicians should address obesity in a compassionate and caring way, be aware of community resources to help families adopt healthy lifestyles, and “look for the comorbidities of obesity,” such as type 2 diabetes, liver disease, sleep apnea, knee problems, and hypertension, Dr. Hassink said.
Policies that address other factors, such as the cost of healthy foods and the marketing of unhealthy foods, may also be needed, Dr. Hassink said.
“I’ve always thought of obesity as kind of the canary in the coal mine,” Dr. Hassink said. “It is important to keep our minds on the fact that it is a chronic disease. But it also indicates a lot of things about how we are able to support a healthy population.”
Potato chips, red meat, and sugary drinks
Other researchers have assessed how healthy behaviors tended to take a turn for the worse when routines were disrupted during the pandemic. Steven B. Heymsfield, MD, a professor in the metabolism and body composition laboratory at Pennington Biomedical Research Center, Louisiana State University System, Baton Rouge, and collaborators documented how diet and activity changed for children during the pandemic.
Dr. Heymsfield worked with researchers in Italy to examine changes in behavior among 41 children and adolescents with obesity in Verona, Italy, during an early lockdown.
As part of a longitudinal observational study, they had baseline data about diet and physical activity from interviews conducted from May to July 2019. They repeated the interviews 3 weeks after a mandatory quarantine.
Intake of potato chips, red meat, and sugary drinks had increased, time spent in sports activities had decreased by more than 2 hours per week, and screen time had increased by more than 4 hours per day, the researchers found. Their study was published in Obesity.
Unpublished follow-up data indicate that “there was further deterioration in the diets and activity patterns” for some but not all of the participants, Dr. Heymsfield said.
He said he was hopeful that children who experienced the onset of obesity during the pandemic may lose weight when routines return to normal, but added that it is unclear whether that will happen.
“My impression from the limited written literature on this question is that for some kids who gain weight during the lockdown or, by analogy, the summer months, the weight doesn’t go back down again. It is not universal, but it is a known phenomenon that it is a bit of a ratchet,” he said. “They just sort of slowly ratchet their weights up, up to adulthood.”
Recognizing weight gain during the pandemic may be an important first step.
“The first thing is not to ignore it,” Dr. Heymsfield said. “Anything that can be done to prevent excess weight gain during childhood – not to promote anorexia or anything like that, but just being careful – is very important, because these behaviors are formed early in life, and they persist.”
CHOP supported the research. Dr. Jenssen and Dr. Hassink have disclosed no relevant financial relationships. Dr. Heymsfield is a medical adviser for Medifast, a weight loss company.
A version of this article first appeared on Medscape.com.
Obesity rates among children jumped substantially in the first months of the COVID-19 pandemic, according to a study published online in Pediatrics. Experts worry the excess weight will be a continuing problem for these children.
“Across the board in the span of a year, there has been a 2% increase in obesity, which is really striking,” lead author Brian P. Jenssen, MD, said in an interview.
The prevalence of obesity in a large pediatric primary care network increased from 13.7% to 15.4%.
Preexisting disparities by race or ethnicity and socioeconomic status worsened, noted Dr. Jenssen, a primary care pediatrician affiliated with Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania, Philadelphia.
Dr. Jenssen and colleagues compared the average obesity rate from June to December 2020 with the rate from June to December 2019 among patients in the CHOP Care Network, which includes 29 urban, suburban, and semirural clinics in the Philadelphia region. In June 2020, the volume of patient visits “returned to near-normal” after a dramatic decline in March 2020, the study authors wrote.
The investigators examined body mass index at all visits for patients aged 2-17 years for whom height and weight were documented. Patients with a BMI at or above the 95th percentile were classified as obese. The analysis included approximately 169,000 visits in 2019 and about 145,000 in 2020.
The average age of the patients was 9.2 years, and 48.9% were girls. In all, 21.4% were non-Hispanic Black, and about 30% were publicly insured.
Increases in obesity rates were more pronounced among patients aged 5-9 years and among patients who were Hispanic/Latino, non-Hispanic Black, publicly insured, or from lower-income neighborhoods.
Whereas the obesity rate increased 1% for patients aged 13-17 years, the rate increased 2.6% for patients aged 5-9 years.
Nearly 25% of Hispanic/Latino or non-Hispanic Black patients seen during the pandemic were obese, compared with 11.3% of non-Hispanic White patients. Before the pandemic, differences by race or ethnicity had been about 10%-11%.
Limiting the analysis to preventive visits did not meaningfully change the results, wrote Dr. Jenssen and colleagues.
“Having any increase in the obesity rates is alarming,” said Sandra Hassink, MD, medical director for the American Academy of Pediatrics’ (AAP’s) Institute for Healthy Childhood Weight. “I think what we’re seeing is what we feared.”
Before the pandemic, children received appropriately portioned breakfasts and lunches at school, but during the pandemic, they had less access to such meals, the academy noted. Disruptions to schooling, easier access to unhealthy snacks, increased screen time, and economic issues such as parents’ job losses were further factors, Hassink said.
Tackling the weight gain
In December 2020, the AAP issued two clinical guidance documents to highlight the importance of addressing obesity during the pandemic. Recommendations included physician counseling of families about maintaining healthy nutrition, minimizing sedentary time, and getting enough sleep and physical activity, as well as the assessment of all patients for onset of obesity and the maintenance of obesity treatment for patients with obesity.
In addition to clinical assessments and guidance, Dr. Jenssen emphasized that a return to routines may be crucial. Prepandemic studies have shown that many children, especially those insured by Medicaid, gain more weight during the summer when they are out of school, he noted. Many of the same factors are present during the pandemic, he said.
“One solution, and probably the most important solution, is getting kids back in school,” Dr. Jenssen said. School disruptions also have affected children’s learning and mental health, but those effects may be harder to quantify than BMI, he said.
Dr. Jenssen suggests that parents do their best to model good routines and habits. For example, they might decide that they and their children will stop drinking soda as a family, or opt for an apple instead of a bag of chips. They can walk around the house or up and down stairs when talking. “Those sorts of little things can make a big difference in the long run,” Dr. Jenssen said.
Clinicians should address obesity in a compassionate and caring way, be aware of community resources to help families adopt healthy lifestyles, and “look for the comorbidities of obesity,” such as type 2 diabetes, liver disease, sleep apnea, knee problems, and hypertension, Dr. Hassink said.
Policies that address other factors, such as the cost of healthy foods and the marketing of unhealthy foods, may also be needed, Dr. Hassink said.
“I’ve always thought of obesity as kind of the canary in the coal mine,” Dr. Hassink said. “It is important to keep our minds on the fact that it is a chronic disease. But it also indicates a lot of things about how we are able to support a healthy population.”
Potato chips, red meat, and sugary drinks
Other researchers have assessed how healthy behaviors tended to take a turn for the worse when routines were disrupted during the pandemic. Steven B. Heymsfield, MD, a professor in the metabolism and body composition laboratory at Pennington Biomedical Research Center, Louisiana State University System, Baton Rouge, and collaborators documented how diet and activity changed for children during the pandemic.
Dr. Heymsfield worked with researchers in Italy to examine changes in behavior among 41 children and adolescents with obesity in Verona, Italy, during an early lockdown.
As part of a longitudinal observational study, they had baseline data about diet and physical activity from interviews conducted from May to July 2019. They repeated the interviews 3 weeks after a mandatory quarantine.
Intake of potato chips, red meat, and sugary drinks had increased, time spent in sports activities had decreased by more than 2 hours per week, and screen time had increased by more than 4 hours per day, the researchers found. Their study was published in Obesity.
Unpublished follow-up data indicate that “there was further deterioration in the diets and activity patterns” for some but not all of the participants, Dr. Heymsfield said.
He said he was hopeful that children who experienced the onset of obesity during the pandemic may lose weight when routines return to normal, but added that it is unclear whether that will happen.
“My impression from the limited written literature on this question is that for some kids who gain weight during the lockdown or, by analogy, the summer months, the weight doesn’t go back down again. It is not universal, but it is a known phenomenon that it is a bit of a ratchet,” he said. “They just sort of slowly ratchet their weights up, up to adulthood.”
Recognizing weight gain during the pandemic may be an important first step.
“The first thing is not to ignore it,” Dr. Heymsfield said. “Anything that can be done to prevent excess weight gain during childhood – not to promote anorexia or anything like that, but just being careful – is very important, because these behaviors are formed early in life, and they persist.”
CHOP supported the research. Dr. Jenssen and Dr. Hassink have disclosed no relevant financial relationships. Dr. Heymsfield is a medical adviser for Medifast, a weight loss company.
A version of this article first appeared on Medscape.com.
Obesity rates among children jumped substantially in the first months of the COVID-19 pandemic, according to a study published online in Pediatrics. Experts worry the excess weight will be a continuing problem for these children.
“Across the board in the span of a year, there has been a 2% increase in obesity, which is really striking,” lead author Brian P. Jenssen, MD, said in an interview.
The prevalence of obesity in a large pediatric primary care network increased from 13.7% to 15.4%.
Preexisting disparities by race or ethnicity and socioeconomic status worsened, noted Dr. Jenssen, a primary care pediatrician affiliated with Children’s Hospital of Philadelphia (CHOP) and the University of Pennsylvania, Philadelphia.
Dr. Jenssen and colleagues compared the average obesity rate from June to December 2020 with the rate from June to December 2019 among patients in the CHOP Care Network, which includes 29 urban, suburban, and semirural clinics in the Philadelphia region. In June 2020, the volume of patient visits “returned to near-normal” after a dramatic decline in March 2020, the study authors wrote.
The investigators examined body mass index at all visits for patients aged 2-17 years for whom height and weight were documented. Patients with a BMI at or above the 95th percentile were classified as obese. The analysis included approximately 169,000 visits in 2019 and about 145,000 in 2020.
The average age of the patients was 9.2 years, and 48.9% were girls. In all, 21.4% were non-Hispanic Black, and about 30% were publicly insured.
Increases in obesity rates were more pronounced among patients aged 5-9 years and among patients who were Hispanic/Latino, non-Hispanic Black, publicly insured, or from lower-income neighborhoods.
Whereas the obesity rate increased 1% for patients aged 13-17 years, the rate increased 2.6% for patients aged 5-9 years.
Nearly 25% of Hispanic/Latino or non-Hispanic Black patients seen during the pandemic were obese, compared with 11.3% of non-Hispanic White patients. Before the pandemic, differences by race or ethnicity had been about 10%-11%.
Limiting the analysis to preventive visits did not meaningfully change the results, wrote Dr. Jenssen and colleagues.
“Having any increase in the obesity rates is alarming,” said Sandra Hassink, MD, medical director for the American Academy of Pediatrics’ (AAP’s) Institute for Healthy Childhood Weight. “I think what we’re seeing is what we feared.”
Before the pandemic, children received appropriately portioned breakfasts and lunches at school, but during the pandemic, they had less access to such meals, the academy noted. Disruptions to schooling, easier access to unhealthy snacks, increased screen time, and economic issues such as parents’ job losses were further factors, Hassink said.
Tackling the weight gain
In December 2020, the AAP issued two clinical guidance documents to highlight the importance of addressing obesity during the pandemic. Recommendations included physician counseling of families about maintaining healthy nutrition, minimizing sedentary time, and getting enough sleep and physical activity, as well as the assessment of all patients for onset of obesity and the maintenance of obesity treatment for patients with obesity.
In addition to clinical assessments and guidance, Dr. Jenssen emphasized that a return to routines may be crucial. Prepandemic studies have shown that many children, especially those insured by Medicaid, gain more weight during the summer when they are out of school, he noted. Many of the same factors are present during the pandemic, he said.
“One solution, and probably the most important solution, is getting kids back in school,” Dr. Jenssen said. School disruptions also have affected children’s learning and mental health, but those effects may be harder to quantify than BMI, he said.
Dr. Jenssen suggests that parents do their best to model good routines and habits. For example, they might decide that they and their children will stop drinking soda as a family, or opt for an apple instead of a bag of chips. They can walk around the house or up and down stairs when talking. “Those sorts of little things can make a big difference in the long run,” Dr. Jenssen said.
Clinicians should address obesity in a compassionate and caring way, be aware of community resources to help families adopt healthy lifestyles, and “look for the comorbidities of obesity,” such as type 2 diabetes, liver disease, sleep apnea, knee problems, and hypertension, Dr. Hassink said.
Policies that address other factors, such as the cost of healthy foods and the marketing of unhealthy foods, may also be needed, Dr. Hassink said.
“I’ve always thought of obesity as kind of the canary in the coal mine,” Dr. Hassink said. “It is important to keep our minds on the fact that it is a chronic disease. But it also indicates a lot of things about how we are able to support a healthy population.”
Potato chips, red meat, and sugary drinks
Other researchers have assessed how healthy behaviors tended to take a turn for the worse when routines were disrupted during the pandemic. Steven B. Heymsfield, MD, a professor in the metabolism and body composition laboratory at Pennington Biomedical Research Center, Louisiana State University System, Baton Rouge, and collaborators documented how diet and activity changed for children during the pandemic.
Dr. Heymsfield worked with researchers in Italy to examine changes in behavior among 41 children and adolescents with obesity in Verona, Italy, during an early lockdown.
As part of a longitudinal observational study, they had baseline data about diet and physical activity from interviews conducted from May to July 2019. They repeated the interviews 3 weeks after a mandatory quarantine.
Intake of potato chips, red meat, and sugary drinks had increased, time spent in sports activities had decreased by more than 2 hours per week, and screen time had increased by more than 4 hours per day, the researchers found. Their study was published in Obesity.
Unpublished follow-up data indicate that “there was further deterioration in the diets and activity patterns” for some but not all of the participants, Dr. Heymsfield said.
He said he was hopeful that children who experienced the onset of obesity during the pandemic may lose weight when routines return to normal, but added that it is unclear whether that will happen.
“My impression from the limited written literature on this question is that for some kids who gain weight during the lockdown or, by analogy, the summer months, the weight doesn’t go back down again. It is not universal, but it is a known phenomenon that it is a bit of a ratchet,” he said. “They just sort of slowly ratchet their weights up, up to adulthood.”
Recognizing weight gain during the pandemic may be an important first step.
“The first thing is not to ignore it,” Dr. Heymsfield said. “Anything that can be done to prevent excess weight gain during childhood – not to promote anorexia or anything like that, but just being careful – is very important, because these behaviors are formed early in life, and they persist.”
CHOP supported the research. Dr. Jenssen and Dr. Hassink have disclosed no relevant financial relationships. Dr. Heymsfield is a medical adviser for Medifast, a weight loss company.
A version of this article first appeared on Medscape.com.
New COVID-19 cases rise again in children
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
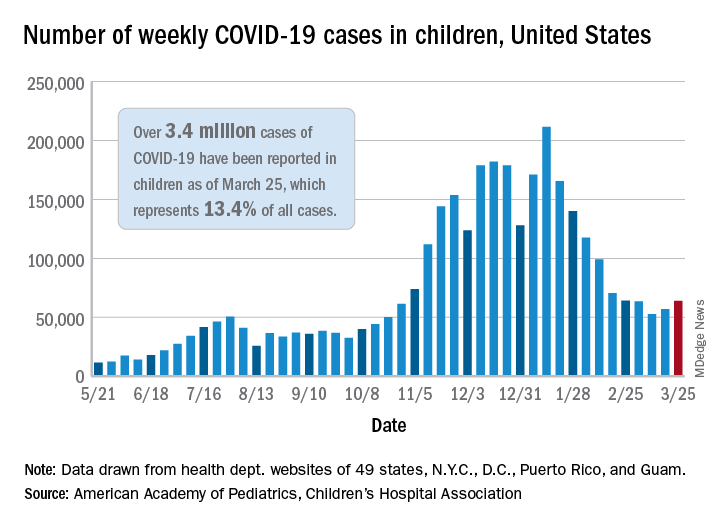
That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
The number of new COVID-19 cases in children increased for the second consecutive week in the United States, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

That brings the number of children infected with the coronavirus to over 3.4 million since the beginning of the pandemic, or 13.4% of all reported cases, the AAP and CHA said in their weekly COVID-19 report.
For just the week of March 19-25, however, the proportion of all cases occurring in children was quite a bit higher, 19.1%. That’s higher than at any other point during the pandemic, passing the previous high of 18.7% set just a week earlier, based on the data collected by AAP/CHA from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
The national infection rate was 4,525 cases per 100,000 children for the week of March 19-25, compared with 4,440 per 100,000 the previous week. States falling the farthest from that national mark were Hawaii at 1,101 per 100,000 and North Dakota at 8,848, the AAP and CHA said.
There was double-digit increase, 11, in the number of child deaths, as the total went from 268 to 279 despite Virginia’s revising its mortality data downward. The mortality rate for children remains 0.01%, and children represent only 0.06% of all COVID-19–related deaths in the 43 states, along with New York City, Puerto Rico, and Guam, that are reporting deaths by age, the report shows.
The state/local-level data show that Texas has the highest number of child deaths (48), followed by Arizona (26), New York City (22), California (16), and Illinois (16), while nine states and the District of Columbia have not yet reported a death, the AAP and CHA said.
FDA approves mirabegron to treat pediatric NDO
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has expanded the indication for mirabegron (Myrbetriq/Myrbetriq Granules) to treat neurogenic detrusor overactivity (NDO), a bladder dysfunction related to neurologic impairment, in children aged 3 years and older.
This comes 1 year after the FDA approved solifenacin succinate, the first treatment of NDO in pediatric patients aged 2 years and older.
The approval of the drug for these new indications is a “positive step” for the treatment of NDO in young patients, Christine P. Nguyen, MD, director of the FDA’s Division of Urology, Obstetrics, and Gynecology, said in an FDA statement.
“Mirabegron, the active ingredient in Myrbetriq and Myrbetriq Granules, works by a different mechanism of action from the currently approved treatments, providing a new treatment option for these young patients. We remain committed to facilitating the development and approval of safe and effective therapies for pediatric NDO patients,” Dr. Nguyen said.
NDO is a bladder dysfunction that frequently occurs in patients with congenital conditions, such as spina bifida. It also occurs in people who suffer from other diseases or injuries of the nervous system, such as multiple sclerosis and spinal cord injury. Symptoms of the condition include urinary frequency and incontinence.
The condition is characterized by the overactivity of the bladder wall muscle, which is normally relaxed to allow storage of urine. Irregular bladder muscle contraction increases storage pressure and decreases the amount of urine the bladder can hold. This can also put the upper urinary tract at risk for deterioration and cause permanent damage to the kidneys.
The effectiveness of Myrbetriq and Myrbetriq Granules for pediatric NDO was determined in a study of 86 children and adolescents aged 3-17 years. The researchers found that after 24 weeks of treatment, the drug improved the patients’ bladder capacity, reduced the number of bladder wall muscle contractions, and improved the volume of urine that could be held. It also reduced the daily number of episodes of leakage.
Side effects of Myrbetriq and Myrbetriq Granules include urinary tract infection, cold symptoms, angioedema, constipation, and headache. The FDA said the drug may also increase blood pressure and may worsen blood pressure in patients who have a history of hypertension.
The FDA approved mirabegron in 2012 to treat overactive bladder in adults.
A version of this article first appeared on Medscape.com.
Child abuse tied to persistent inflammation in later life
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People who suffer abuse as children continue to have higher levels of inflammatory biomarkers as adults, likely placing them at increased risk for chronic health problems, new research shows.
In a study assessing trajectories of inflammation over a 3-year period in healthy adults, those who reported higher rates of physical, sexual, or emotional abuse had higher levels of bioinflammatory blood markers versus those who reported lower rates of abuse. These individuals also had significantly higher rates of loneliness and depression.
“These adverse experiences that people can have in childhood and adolescence really can continue to influence our health at a biological level well into adulthood,” investigator Megan E. Renna, PhD, postdoctoral fellow at Ohio State University, Columbus, said in an interview.
“There may be a somewhat invisible biological effect of that abuse later on in life, in addition to all of the psychological distress that could go along with those experiences,” she added.
The findings were presented at the virtual Anxiety and Depression Association of America Conference 2021.
Need for intervention
Ages ranged from 47 to 67 years (mean age, 57 years), 81% were women, all were relatively healthy, and there were low rates of medical comorbidities.
Participant data on physical, emotional, and sexual abuse prior to age 18 were ascertained using the Childhood Experiences Questionnaire. Blood samples were assayed for cytokines interleukin-6, IL-8, IL-1 beta and tumor necrosis factor–alpha at each visit.
After controlling for age, sex, body mass index, and medical comorbidities, results showed markers of inflammation increased at a greater rate over time in participants with higher rates of physical (P = .05) and sexual abuse (P = .02), compared with those with no history of childhood abuse.
“So, inflammation was increasing at a faster rate across those three visits for people with versus without an abuse history. And this was abuse experienced before age 18, but the mean age of our participants was about 57,” said Dr. Renna.
“It is likely that emotional abuse plays a role in inflammation but we did not have a big enough sample to show significance,” she added.
Participants who had reported childhood abuse also demonstrated significantly higher rates of loneliness and depression across all visits than those without a history of abuse.
“One of the things this work really highlights is the need for intervention for these children and adolescents who are experiencing abuse. This may have a helpful impact on their psychological health as they age, as well as their physical health,” Dr. Renna said.
‘Considerable interest’
In a comment, ADAA President Luana Marques, PhD, Harvard Medical School, Boston, said research is “consistently indicating that childhood adversity puts individuals at risk for a host of problems, including inflammatory concerns, which are precursors for other physical illnesses.”
Such results “demonstrate the importance of early identification and intervention of possible traumatic experiences for youth, and how early intervention at the parent level might also be helpful,” said Dr. Marques, who was not involved with the research.
Also commenting on the study, Charles B. Nemeroff, MD, PhD, professor and chair in the department of psychiatry and behavioral science at the University of Texas at Austin, and president-elect of the ADAA, said in an interview that the findings are pertinent for the field.
“The investigators demonstrated that a history of childhood physical or sexual abuse was associated with a greater inflammatory response, and this is of considerable interest because this increased inflammatory response very likely contributes to the well-documented increased prevalence of serious medical disorders such as heart disease, diabetes, and cancer in victims of child abuse and neglect,” said Dr. Nemeroff, who was not associated with the research.
Dr. Renna, Dr. Marques, and Dr. Nemeroff disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
‘Politics has no place here,’ AAP says about transgender care
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
The American Academy of Pediatrics (AAP) released a statement condemning state legislation introduced across the country that would prohibit the medical treatment of children who identify as transgender.
“Politics has no place here. Transgender children, like all children, just want to belong. We will fight state by state, in the courts, and on the national stage to make sure they know they do,” said the American Academy of Pediatrics, an organization that recommends that youth who identify as transgender have access to comprehensive, gender-affirming and “developmentally appropriate” health care.
There has been a lot of controversy among physicians surrounding gender-affirming care for children. In 2019, three separate groups of physicians questioned the hormonal treatment of children and adolescents with gender dysphoria, saying more research is needed to support gender-affirming care recommendations from groups such as the AAP. In addition, California-based endocrinologist Michael K. Laidlaw, MD, called the use of puberty blockers – medication used to delay or prevent the onset of puberty – an “experiment” and “public health problem.”
Some of the bills circulating would prohibit gender-affirming care for children and penalize pediatricians and other physicians for providing such care. Other bills would prevent transgender youth from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Jason Rafferty, MD, a pediatrician who authored AAP’s 2018 policy statement encouraging pediatricians to provide gender-affirmative care to children and adolescents, said in an interview that these proposed state bills are targeting a vulnerable population at a vulnerable time.
“You have this marginalized group and right in the midst of a pandemic – and [social isolation] – and you have these policies coming out that are really targeting the most vulnerable,” said Dr. Rafferty, who practices at the Adolescent Healthcare Center at Hasbro Children’s Hospital, Providence, R.I. “Some of the proposed policies would ensure that some of these transgender kids wouldn’t have access to basic medical care [or access to areas] where pediatricians are affirming these kids and creating a safe environment for them.”
M. Brett Cooper, MD, of pediatrics at UT Southwestern Medical Center, Dallas, said in an interview that the proposed state legislations are based on personal beliefs, not evidence-based care.
He said the AAP is speaking out because many of these proposed state legislatures are advancing bills that would affect gender-diverse children.
“Many of these bills are making it close to passing both chambers of a state legislature and thus sent to the governor for approval,” Dr. Cooper said. “In past legislative sessions, bills such as these have never it made out of committee in either a state House or state Senate.”
Early in March, South Dakota Governor Kristi Noem tweeted that she was “excited to sign” a bill that would prevent children who are transgender from participating in sports or on athletic teams according to their gender identity, requiring them to join teams aligned with the sex they were assigned at birth.
Meanwhile, previous studies have shown that teens whose gender identity does not align with their sex assigned at birth were more likely to report a suicide attempt, compared with cisgender youth. A 2015 study has attributed this increased risk of suicide to marginalization, social exclusion, and trauma. The bills being proposed may exacerbate this, according to Dr. Rafferty.
These bills point in the direction of approaches and models that research shows may be harmful to transgender children, he said.
“Beyond that, policies that are out there affecting participation in sports and other activities really just draw attention to these youth in unnecessary ways and really puts their physical and mental health at risk in a school environment,” he added.
“Whether it’s health care, school, community, or home [these policies] just really create a very hostile and dangerous environment toward a population that already is at significant risk,” Dr. Rafferty said.
The AAP’s clinical care guidelines for children who identify as transgender state that pediatricians who provide gender-affirmative care and speak with children and their families about gender issues help foster an environment of nurturance and support.
“Many people think that the care provided to gender diverse young people is rogue,” Dr. Cooper said. “Pediatric gender clinics are often either multidisciplinary or have a readily accessible team, including social work, mental health, and the physician. Medical care for gender diverse youth follows well-established standards of care.”
Dr. Rafferty said it is important for care providers to support children and teenagers who identify as transgender by creating a safe space and helping them identify what they need.
“There’s a lot out there that really tries to unite gender and biological sex together, saying that gender can be reduced to your anatomy,” Dr. Rafferty said. “But I think what we’re coming to appreciate is that our sense of identity is much more complex than simply our genes or biology.”
Dr. Rafferty is a member of the American Academy of Pediatrics, the Society of Adolescent Health and Medicine, and the American Academy of Child and Adolescent Psychiatry. Dr. Cooper is a columnist for Pediatric News and is on the board of trustees for the Texas Medical Association.
COVID-19 vaccination in RMD patients: Safety data “reassuring”
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
Two reports support the safety and immunogenicity of SARS-CoV-2 mRNA vaccines in patients with rheumatic and musculoskeletal diseases (RMDs) and represent the first available data on such patients.
In an observational cohort study published in Annals of the Rheumatic Diseases, Caoilfhionn M. Connolly, MD, of Johns Hopkins University, Baltimore, and colleagues reviewed data from 325 adults with RMDs who received the first dose of SARS-CoV-2 mRNA vaccine during the period of Dec. 17, 2020, to Feb. 11, 2021. Of these, 51% received the Pfizer/BioNTech vaccine and 49% received the Moderna vaccine.
The patients, who were invited to participate on social media, were aged 34-54 years, 96% were women, and 89% were White. Inflammatory arthritis was the most common RMD condition (38%), followed by systemic lupus erythematosus (28%) and overlap connective tissue disease (19%). The patients were using a range of immunomodulatory treatment regimens, including nonbiologic disease modifying antirheumatic drugs (DMARDs) in 44%, biologics in 19%, and combination therapy in 37%.
Overall, 89% of patients reported localized symptoms of pain, swelling, and erythema, and 69% reported systemic symptoms. Fatigue was the most common systemic symptom, and 7.4% reported severe fatigue.
None of the patients experienced allergic reactions requiring epinephrine, and 3% reported new infections that required treatment.
“These early, reassuring results may ameliorate concern among patients and provide guidance for rheumatology providers in critical discussions regarding vaccine hesitancy or refusal,” they concluded.
Antibody responses
In another study published in Annals of the Rheumatic Diseases by the same group of researchers, antibody responses against the receptor binding domain of the SARS-CoV-2 spike protein were seen in 74% of 123 adults with an RMD at 18-26 days after receiving a first dose of SARS-CoV-2 mRNA vaccine (52% Pfizer vaccine and 48% Moderna) between Jan. 8, 2021, and Feb. 12, 2021.
The most common diagnoses in these patients were inflammatory arthritis (28%), systemic lupus erythematosus (20%), and Sjögren’s syndrome (13%). A total of 28% of participants reported taking no immunomodulatory agents, 19% reported nonbiologic DMARDs, 14% reported biologic DMARDs, and 19% reported combination therapy.
Although no differences appeared based on disease groups or overall categories of immunomodulatory therapies, patients whose treatment included mycophenolate or rituximab were significantly less likely to develop antibody responses than were patients not taking these medications (P = .001 and P = .04, respectively). Although rituximab and methotrexate have been associated with reduced responses to vaccines such as the flu vaccine, methotrexate was not associated with reduced vaccine response in this study. A total of 94% of patients taking a tumor necrosis factor inhibitor had detectable antibodies.
The studies’ findings were limited by several factors including a lack of longer-term safety data; the small, nonrandomized sample of mainly white women; limited information on immunomodulatory drug dosage and timing; lack of serial antibody measurements; use of an enzyme immunoassay designed to detect antibody response after natural infection; and the inclusion of data only on the first dose of a two-dose vaccine series, the researchers noted. However, the data should provide additional reassurance to RMD patients and their health care teams about vaccination against COVID-19, they said.
Both studies were supported by the Ben-Dov family. In addition, the studies were supported by grants to various study authors from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and the Transplantation and Immunology Research Network of the American Society of Transplantation. One author disclosed financial relationships with Sanofi, Novartis, CSL Behring, Jazz Pharmaceuticals, Veloxis, Mallinckrodt, and Thermo Fisher Scientific. The other researchers had no financial conflicts to disclose.
FROM ANNALS OF THE RHEUMATIC DISEASES
Maternal caffeine consumption, even small amounts, may reduce neonatal size
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
For pregnant women, just half a cup of coffee a day may reduce neonatal birth size and body weight, according to a prospective study involving more than 2,500 women.
That’s only 50 mg of a caffeine day, which falls below the upper threshold of 200 mg set by the American College of Obstetricians and Gynecologists, lead author Jessica Gleason, PhD, MPH, of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Bethesda, Md, and colleagues reported.
“Systematic reviews and meta-analyses have reported that maternal caffeine consumption, even in doses lower than 200 mg, is associated with a higher risk for low birth weight, small for gestational age (SGA), and fetal growth restriction, suggesting there may be no safe amount of caffeine during pregnancy,” the investigators wrote in JAMA Network Open.
Findings to date have been inconsistent, with a 2014 meta-analysis reporting contrary or null results in four out of nine studies.
Dr. Gleason and colleagues suggested that such discrepancies may be caused by uncontrolled confounding factors in some of the studies, such as smoking, as well as the inadequacy of self-reporting, which fails to incorporate variations in caffeine content between beverages, or differences in rates of metabolism between individuals.
“To our knowledge, no studies have examined the association between caffeine intake and neonatal anthropometric measures beyond weight, length, and head circumference, and few have analyzed plasma concentrations of caffeine and its metabolites or genetic variations in the rate of metabolism associated with neonatal size,” the investigators wrote.
Dr. Gleason and colleagues set out to address this knowledge gap with a prospective cohort study, including 2,055 nonsmoking women with low risk of birth defects who presented at 12 centers between 2009 and 2013. Mean participant age was 28.3 years and mean body mass index was 23.6. Races and ethnicities were represented almost evenly even across four groups: Hispanic (28.2%), White (27.4%), Black (25.2%), and Asian/Pacific Islander (19.2%). Rate of caffeine metabolism was defined by the single-nucleotide variant rs762551 (CYP1A2*1F), according to which, slightly more women had slow metabolism (52.7%) than fast metabolism (47.3%).
Women were enrolled at 8-13 weeks’ gestational age, at which time they underwent interviews and blood draws, allowing for measurement of caffeine and paraxanthine plasma levels, as well as self-reported caffeine consumption during the preceding week.
Over the course of six visits, fetal growth was observed via ultrasound. Medical records were used to determine birth weights and neonatal anthropometric measures, including fat and skin fold mass, body length, and circumferences of the thigh, arm, abdomen, and head.
Neonatal measurements were compared with plasma levels of caffeine and paraxanthine, both continuously and as quartiles (Q1, ≤ 28.3 ng/mL; Q2, 28.4-157.1 ng/mL; Q3, 157.2-658.8 ng/mL; Q4, > 658.8 ng/mL). Comparisons were also made with self-reported caffeine intake.
Women who reported drinking 1-50 mg of caffeine per day had neonates with smaller subscapular skin folds (beta = –0.14 mm; 95% confidence interval, –0.27 to -–0.01 mm), while those who reported more than 50 mg per day had newborns with lower birth weight (beta = –66 g; 95% CI, –121 to –10 g), and smaller circumferences of mid-upper thigh (beta = –0.32 cm; 95% CI, –0.55 to –0.09 cm), anterior thigh skin fold (beta = –0.24 mm; 95% CI, –0.47 to -.01 mm), and mid-upper arm (beta = –0.17 cm; 95% CI, –0.31 to –0.02 cm).
Caffeine plasma concentrations supported these findings.
Compared with women who had caffeine plasma concentrations in the lowest quartile, those in the highest quartile gave birth to neonates with shorter length (beta = –0.44 cm; P = .04 for trend) and lower body weight (beta = –84.3 g; P = .04 for trend), as well as smaller mid-upper arm circumference (beta = -0.25 cm; P = .02 for trend), mid-upper thigh circumference (beta = –0.29 cm; P = .07 for trend), and head circumference (beta = –0.28 cm; P < .001 for trend). A comparison of lower and upper paraxanthine quartiles revealed the similar trends, as did analyses of continuous measures.
“Our results suggest that caffeine consumption during pregnancy, even at levels much lower than the recommended 200 mg per day of caffeine may be associated with decreased fetal growth,” the investigators concluded.
Sarah W. Prager, MD, of the University of Washington, Seattle, suggested that the findings “do not demonstrate that caffeine has a clinically meaningful negative clinical impact on newborn size and weight.”
She noted that there was no difference in the rate of SGA between plasma caffeine quartiles, and that most patients were thin, which may not accurately represent the U.S. population.
“Based on these new data, my take home message to patients would be that increasing amounts of caffeine can have a small but real impact on the size of their baby at birth, though it is unlikely to result in a diagnosis of SGA,” she said. “Pregnant patients may want to limit caffeine intake even more than the ACOG recommendation of 200 mg per day.”
According to Robert M. Silver, MD, of the University of Utah Health Sciences Center, Salt Lake City, “data from this study are of high quality, owing to the prospective cohort design, large numbers, assessment of biomarkers, and sophisticated analyses.”
Still, he urged a cautious interpretation from a clinical perspective.
“It is important to not overreact to these data,” he said. “The decrease in fetal growth associated with caffeine is small and may prove to be clinically meaningless. Accordingly, clinical recommendations regarding caffeine intake during pregnancy should not be modified solely based on this study.”
Dr. Silver suggested that the findings deserve additional investigation.
“These observations warrant further research about the effects of caffeine exposure during pregnancy,” he said. “Ideally, studies should assess the effect of caffeine exposure on fetal growth in various pregnancy epochs as well as on neonatal and childhood growth.”
The study was funded by the Intramural Research Program of the NICHD. Dr. Gerlanc is an employee of The Prospective Group, which was contracted to provide statistical support.
FROM JAMA NETWORK OPEN
Preterm infant supine sleep positioning becoming more common, but racial/ethnic disparities remain
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
Although supine sleep positioning of preterm infants is becoming more common, racial disparities remain, according to a retrospective analysis involving more than 66,000 mothers.
Non-Hispanic Black preterm infants were 39%-56% less likely to sleep on their backs than were non-Hispanic White preterm infants, reported lead author Sunah S. Hwang, MD, MPH, of the University Colorado, Aurora, and colleagues.
According to the investigators, these findings may explain, in part, why the risk of sudden unexpected infant death (SUID) is more than twofold higher among non-Hispanic Black preterm infants than non-Hispanic White preterm infants.
“During the first year of life, one of the most effective and modifiable parental behaviors that may reduce the risk for SUID is adhering to safe infant sleep practices, including supine sleep positioning or back-sleeping,” wrote Dr. Hwang and colleagues. The report is in the Journal of Pediatrics. “For the healthy-term population, research on the racial/ethnic disparity in adherence to safe sleep practices is robust, but for preterm infants who are at much higher risk for SUID, less is known.”
To address this knowledge gap, the investigators conducted a retrospective study using data from the Pregnancy Risk Assessment Monitoring System (PRAMS), a population-based perinatal surveillance system. The final dataset involved 66,131 mothers who gave birth to preterm infants in 16 states between 2000 and 2015. The sample size was weighted to 1,020,986 mothers.
The investigators evaluated annual marginal prevalence of supine sleep positioning among two cohorts: early preterm infants (gestational age less than 34 weeks) and late preterm infants (gestational age 34-36 weeks). The primary outcome was rate of supine sleep positioning, a practice that must have been followed consistently, excluding other positions (i.e. prone or side). Mothers were grouped by race/ethnicity into four categories: non-Hispanic Black, non-Hispanic White, Hispanic, and other. Several other maternal and infant characteristics were recorded, including marital status, maternal age, education, insurance prior to birth, history of previous live birth, insurance, method of delivery, birth weight, and sex.
From 2000 to 2015, the overall adjusted odds of supine sleep positioning increased by 8.5% in the early preterm group and 5.2% in the late preterm group. This intergroup difference may be due to disparate levels of in-hospital education, the investigators suggested.
“Perhaps the longer NICU hospitalization for early preterm infants compared with late preterm infants affords greater opportunities for parental education and engagement about safe sleep practices,” they wrote.
Among early preterm infants, odds percentages increased by 7.3%, 7.7%, and 10.0% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers, respectively. For late preterm infants, respective rates increased by 5.9%, 4.8%, and 5.8% for non-Hispanic Black, Hispanic, and non-Hispanic White mothers.
Despite these improvements, racial disparities were still observed. Non-Hispanic Black mothers reported lower rates of supine sleep positioning for both early preterm infants (odds ratio [OR], 0.61; P less than .0001) and late preterm infants (OR, 0.44; P less than .0001) compared with non-Hispanic White mothers.
These disparities seem “to be in line with racial/ethnic disparity trends in infant mortality and in SUID rates that have persisted for decades among infants,” the investigators wrote.
To a lesser degree, and lacking statistical significance, Hispanic mothers reported lower odds of supine sleep positioning than the odds of White mothers for both early preterm infants (OR, 0.80; P = .1670) and late preterm infants (OR, 0.81; P = .1054).
According to Dr. Hwang and colleagues, more specific demographic data are needed to accurately describe supine sleep positioning rates among Hispanic mothers, partly because of the heterogeneity of this cohort.
“A large body of literature has shown significant variability by immigrant status and country of origin in several infant health outcomes among the Hispanic population,” the investigators wrote. “This study was unable to stratify the Hispanic cohort by these characteristics and thus the distribution of supine sleep positioning prevalence across different Hispanic subgroups could not be demonstrated in this study.”
The investigators also suggested that interventional studies are needed.
“Additional efforts to understand the barriers and facilitators to SSP [supine sleep positioning] adherence among all preterm infant caregivers, particularly non-Hispanic Black and Hispanic parents, are needed so that novel interventions can then be developed,” they wrote.
According to Denice Cora-Bramble, MD, MBA, chief diversity officer at Children’s National Hospital and professor of pediatrics at George Washington University, Washington, the observed improvements in supine sleep positioning may predict lower rates of infant mortality, but more work in the area is needed.
“In spite of improvement in infants’ supine sleep positioning during the study period, racial/ethnic disparities persisted among non-Hispanic Blacks and Hispanics,” Dr. Cora-Bramble said. “That there was improvement among the populations included in the study is significant because of the associated and expected decrease in infant mortality. However, the study results need to be evaluated within the context of [the study’s] limitations, such as the inclusion of only sixteen states in the data analysis. More research is needed to understand and effectively address the disparities highlighted in the study.”
The investigators and Dr. Cora-Bramble reported no conflicts of interest.
FROM JOURNAL OF PEDIATRICS