User login
COVID-19 maternal antibodies transferred to fetus, newborn from pregnant and lactating vaccine recipients
, according to a prospective cohort study published March 25 in the American Journal of Obstetrics and Gynecology.
The findings revealed that the antibody response to vaccination in this cohort was greater than that from a COVID-19 infection during pregnancy. Though the researchers detected SARS-CoV-2 antibodies in umbilical cord blood and breast milk, it’s not yet known how much protection these antibodies might provide to newborns.
“The presence of neutralizing antibody transfer in nearly all cords, and improved transfer with increased time from vaccination, points to the promise of mRNA vaccine–induced delivery of immunity to neonates,” wrote Kathryn J. Gray, MD, PhD, of Harvard Medical School and Brigham and Women’s Hospital’s department of obstetrics and gynecology, and colleagues. “Transfer would perhaps be optimized if vaccination is administered earlier during gestation, though this needs to be directly examined in future studies.”
The researchers tracked 84 pregnant women, 31 lactating women, and 16 nonpregnant women who received the COVID-19 vaccine. The titers of IgG, IgA, and IgM antibodies against the SARS-CoV-2 spike, receptor binding domain (RBD), and S1 and S2 components of the spike were measured in the 131 participants’ blood and in the lactating women’s breast milk four times: at baseline, when they received their second vaccine dose, at 2-6 weeks after their second dose, and at delivery for the 13 women who delivered during the study period.
The study population included health care workers and was predominantly White and non-Hispanic. In addition, two pregnant women, two lactating women, and one nonpregnant woman in the study had a previous SARS-CoV-2 infection.
Most of the pregnant women received the vaccine in their second (46%) or third (40%) trimester. The women across all three groups – pregnant, lactating, and nonpregnant – experienced similar side effects from the each dose of the vaccine, including fever/chills in 32% of the pregnant women and half the nonpregnant women after the second dose.
Titers induced by the vaccine were similar across the pregnant, lactating, and nonpregnant women, and titers did not differ based on the trimester when women received the vaccine. The researchers then compared the titers from the vaccine recipients to titers of 37 pregnant women drawn 4-12 weeks after a natural SARS-CoV-2 infection. Vaccine-induced titers were significantly greater than those measured in the women who had a natural infection during pregnancy (P < .001).
The researchers identified IgG, IgA, and IgM antibodies in the breast milk samples, including a boost in IgG antibodies after the second vaccine dose from baseline. “However, whether these antibodies were transferred efficiently to infants remained unclear,” the authors noted.
The researchers found vaccine-induced antibodies in all 10 umbilical cord blood samples tested, all but one of which had been exposed to two doses of the vaccine.
“The cord with the lowest spike- and RBD-specific IgG belonged to a mother who delivered between the first and second vaccine doses and had received her first vaccine dose 17 days prior to delivery, suggesting that 2 doses may be essential to optimize humoral immune transfer to the neonate,” the authors wrote. “Based on what is known about other vaccines, the amount of maternal IgG transferred across the placenta to the cord is likely to differ by trimester of vaccination.”
Although umbilical cord sera had lower titers of neutralizing antibodies than found in maternal sera, the difference was not significant (median interquartile range 52.3 vs. 104.7, P = .05). The two cord blood samples without neutralizing antibodies came from a woman who had not had the second dose and a woman who received the second dose 1 week before delivery.
“These data provide a compelling argument that COVID-19 mRNA vaccines induce similar humoral immunity in pregnant and lactating women as in the nonpregnant population,” the authors wrote. “These data do not elucidate potential risks to the fetus.”
While the study provides evidence about the immune response induced by the COVID-19 mRNA vaccines during pregnant, it leaves other questions unanswered, said Kevin A. Ault, MD, professor of ob.gyn. at The University of Kansas Medical Center in Kansas City.
“The important thing about these findings is that the COVID vaccines are immunogenic in pregnant women. There may be a benefit to the newborns because antibodies are passed on through the placenta,” Dr. Ault said in an interview. “The main questions that remain are safety of the vaccine during pregnancy and effectiveness of the vaccine during pregnancy.”
He said he expects to see more studies on the safety and effectiveness of COVID-19 vaccines during pregnancy. Despite more than 73,600 infections and 80 deaths from COVID-19 in people who were pregnant, none of the initial COVID-19 vaccine trials included pregnant or lactating participants.
“This is an important initial study to confirm the antibody generation from mRNA vaccination in pregnant women, and the passage of antibody via cord blood and breast milk,” said Linda Eckert, MD, a professor of ob.gyn. at The University of Washington, Seattle, who specializes in maternal immunization. “Further studies are important to look at the timing of vaccination in pregnancy and whether it influences the level of antibody passed to the fetus.”
Though this study is not a safety study, it “does not show increased expected vaccine reactions, such as aches, pains, and fever, in pregnant versus nonpregnant patients,” Dr. Eckert said in an interview. “It is not able to evaluate pregnancy outcome data, but it does allow pregnant women being vaccinated with the mRNA vaccines to know that the vaccine is generating protection for them, and the protection is being passed to the fetus in utero via cordblood and to the infant via breast milk.”
The research was funded by the National Institutes of Health along with the Gates Foundation, the Massachusetts Consortium on Pathogen Readiness (MassCPR), the Musk Foundation, the Ragon Institute of MGH and MIT, and Massachusetts General Hospital and Brigham and Women’s Hospital.
Lead author Dr. Gray has consulted for Illumina, BillionToOne, and Aetion, and three other authors have financial or scientific/medical advising connections to Alba Therapeutics, NextCure, Viome, Systems Seromyx, and Mirvie. Dr. Ault and Dr. Eckert had no disclosures.
, according to a prospective cohort study published March 25 in the American Journal of Obstetrics and Gynecology.
The findings revealed that the antibody response to vaccination in this cohort was greater than that from a COVID-19 infection during pregnancy. Though the researchers detected SARS-CoV-2 antibodies in umbilical cord blood and breast milk, it’s not yet known how much protection these antibodies might provide to newborns.
“The presence of neutralizing antibody transfer in nearly all cords, and improved transfer with increased time from vaccination, points to the promise of mRNA vaccine–induced delivery of immunity to neonates,” wrote Kathryn J. Gray, MD, PhD, of Harvard Medical School and Brigham and Women’s Hospital’s department of obstetrics and gynecology, and colleagues. “Transfer would perhaps be optimized if vaccination is administered earlier during gestation, though this needs to be directly examined in future studies.”
The researchers tracked 84 pregnant women, 31 lactating women, and 16 nonpregnant women who received the COVID-19 vaccine. The titers of IgG, IgA, and IgM antibodies against the SARS-CoV-2 spike, receptor binding domain (RBD), and S1 and S2 components of the spike were measured in the 131 participants’ blood and in the lactating women’s breast milk four times: at baseline, when they received their second vaccine dose, at 2-6 weeks after their second dose, and at delivery for the 13 women who delivered during the study period.
The study population included health care workers and was predominantly White and non-Hispanic. In addition, two pregnant women, two lactating women, and one nonpregnant woman in the study had a previous SARS-CoV-2 infection.
Most of the pregnant women received the vaccine in their second (46%) or third (40%) trimester. The women across all three groups – pregnant, lactating, and nonpregnant – experienced similar side effects from the each dose of the vaccine, including fever/chills in 32% of the pregnant women and half the nonpregnant women after the second dose.
Titers induced by the vaccine were similar across the pregnant, lactating, and nonpregnant women, and titers did not differ based on the trimester when women received the vaccine. The researchers then compared the titers from the vaccine recipients to titers of 37 pregnant women drawn 4-12 weeks after a natural SARS-CoV-2 infection. Vaccine-induced titers were significantly greater than those measured in the women who had a natural infection during pregnancy (P < .001).
The researchers identified IgG, IgA, and IgM antibodies in the breast milk samples, including a boost in IgG antibodies after the second vaccine dose from baseline. “However, whether these antibodies were transferred efficiently to infants remained unclear,” the authors noted.
The researchers found vaccine-induced antibodies in all 10 umbilical cord blood samples tested, all but one of which had been exposed to two doses of the vaccine.
“The cord with the lowest spike- and RBD-specific IgG belonged to a mother who delivered between the first and second vaccine doses and had received her first vaccine dose 17 days prior to delivery, suggesting that 2 doses may be essential to optimize humoral immune transfer to the neonate,” the authors wrote. “Based on what is known about other vaccines, the amount of maternal IgG transferred across the placenta to the cord is likely to differ by trimester of vaccination.”
Although umbilical cord sera had lower titers of neutralizing antibodies than found in maternal sera, the difference was not significant (median interquartile range 52.3 vs. 104.7, P = .05). The two cord blood samples without neutralizing antibodies came from a woman who had not had the second dose and a woman who received the second dose 1 week before delivery.
“These data provide a compelling argument that COVID-19 mRNA vaccines induce similar humoral immunity in pregnant and lactating women as in the nonpregnant population,” the authors wrote. “These data do not elucidate potential risks to the fetus.”
While the study provides evidence about the immune response induced by the COVID-19 mRNA vaccines during pregnant, it leaves other questions unanswered, said Kevin A. Ault, MD, professor of ob.gyn. at The University of Kansas Medical Center in Kansas City.
“The important thing about these findings is that the COVID vaccines are immunogenic in pregnant women. There may be a benefit to the newborns because antibodies are passed on through the placenta,” Dr. Ault said in an interview. “The main questions that remain are safety of the vaccine during pregnancy and effectiveness of the vaccine during pregnancy.”
He said he expects to see more studies on the safety and effectiveness of COVID-19 vaccines during pregnancy. Despite more than 73,600 infections and 80 deaths from COVID-19 in people who were pregnant, none of the initial COVID-19 vaccine trials included pregnant or lactating participants.
“This is an important initial study to confirm the antibody generation from mRNA vaccination in pregnant women, and the passage of antibody via cord blood and breast milk,” said Linda Eckert, MD, a professor of ob.gyn. at The University of Washington, Seattle, who specializes in maternal immunization. “Further studies are important to look at the timing of vaccination in pregnancy and whether it influences the level of antibody passed to the fetus.”
Though this study is not a safety study, it “does not show increased expected vaccine reactions, such as aches, pains, and fever, in pregnant versus nonpregnant patients,” Dr. Eckert said in an interview. “It is not able to evaluate pregnancy outcome data, but it does allow pregnant women being vaccinated with the mRNA vaccines to know that the vaccine is generating protection for them, and the protection is being passed to the fetus in utero via cordblood and to the infant via breast milk.”
The research was funded by the National Institutes of Health along with the Gates Foundation, the Massachusetts Consortium on Pathogen Readiness (MassCPR), the Musk Foundation, the Ragon Institute of MGH and MIT, and Massachusetts General Hospital and Brigham and Women’s Hospital.
Lead author Dr. Gray has consulted for Illumina, BillionToOne, and Aetion, and three other authors have financial or scientific/medical advising connections to Alba Therapeutics, NextCure, Viome, Systems Seromyx, and Mirvie. Dr. Ault and Dr. Eckert had no disclosures.
, according to a prospective cohort study published March 25 in the American Journal of Obstetrics and Gynecology.
The findings revealed that the antibody response to vaccination in this cohort was greater than that from a COVID-19 infection during pregnancy. Though the researchers detected SARS-CoV-2 antibodies in umbilical cord blood and breast milk, it’s not yet known how much protection these antibodies might provide to newborns.
“The presence of neutralizing antibody transfer in nearly all cords, and improved transfer with increased time from vaccination, points to the promise of mRNA vaccine–induced delivery of immunity to neonates,” wrote Kathryn J. Gray, MD, PhD, of Harvard Medical School and Brigham and Women’s Hospital’s department of obstetrics and gynecology, and colleagues. “Transfer would perhaps be optimized if vaccination is administered earlier during gestation, though this needs to be directly examined in future studies.”
The researchers tracked 84 pregnant women, 31 lactating women, and 16 nonpregnant women who received the COVID-19 vaccine. The titers of IgG, IgA, and IgM antibodies against the SARS-CoV-2 spike, receptor binding domain (RBD), and S1 and S2 components of the spike were measured in the 131 participants’ blood and in the lactating women’s breast milk four times: at baseline, when they received their second vaccine dose, at 2-6 weeks after their second dose, and at delivery for the 13 women who delivered during the study period.
The study population included health care workers and was predominantly White and non-Hispanic. In addition, two pregnant women, two lactating women, and one nonpregnant woman in the study had a previous SARS-CoV-2 infection.
Most of the pregnant women received the vaccine in their second (46%) or third (40%) trimester. The women across all three groups – pregnant, lactating, and nonpregnant – experienced similar side effects from the each dose of the vaccine, including fever/chills in 32% of the pregnant women and half the nonpregnant women after the second dose.
Titers induced by the vaccine were similar across the pregnant, lactating, and nonpregnant women, and titers did not differ based on the trimester when women received the vaccine. The researchers then compared the titers from the vaccine recipients to titers of 37 pregnant women drawn 4-12 weeks after a natural SARS-CoV-2 infection. Vaccine-induced titers were significantly greater than those measured in the women who had a natural infection during pregnancy (P < .001).
The researchers identified IgG, IgA, and IgM antibodies in the breast milk samples, including a boost in IgG antibodies after the second vaccine dose from baseline. “However, whether these antibodies were transferred efficiently to infants remained unclear,” the authors noted.
The researchers found vaccine-induced antibodies in all 10 umbilical cord blood samples tested, all but one of which had been exposed to two doses of the vaccine.
“The cord with the lowest spike- and RBD-specific IgG belonged to a mother who delivered between the first and second vaccine doses and had received her first vaccine dose 17 days prior to delivery, suggesting that 2 doses may be essential to optimize humoral immune transfer to the neonate,” the authors wrote. “Based on what is known about other vaccines, the amount of maternal IgG transferred across the placenta to the cord is likely to differ by trimester of vaccination.”
Although umbilical cord sera had lower titers of neutralizing antibodies than found in maternal sera, the difference was not significant (median interquartile range 52.3 vs. 104.7, P = .05). The two cord blood samples without neutralizing antibodies came from a woman who had not had the second dose and a woman who received the second dose 1 week before delivery.
“These data provide a compelling argument that COVID-19 mRNA vaccines induce similar humoral immunity in pregnant and lactating women as in the nonpregnant population,” the authors wrote. “These data do not elucidate potential risks to the fetus.”
While the study provides evidence about the immune response induced by the COVID-19 mRNA vaccines during pregnant, it leaves other questions unanswered, said Kevin A. Ault, MD, professor of ob.gyn. at The University of Kansas Medical Center in Kansas City.
“The important thing about these findings is that the COVID vaccines are immunogenic in pregnant women. There may be a benefit to the newborns because antibodies are passed on through the placenta,” Dr. Ault said in an interview. “The main questions that remain are safety of the vaccine during pregnancy and effectiveness of the vaccine during pregnancy.”
He said he expects to see more studies on the safety and effectiveness of COVID-19 vaccines during pregnancy. Despite more than 73,600 infections and 80 deaths from COVID-19 in people who were pregnant, none of the initial COVID-19 vaccine trials included pregnant or lactating participants.
“This is an important initial study to confirm the antibody generation from mRNA vaccination in pregnant women, and the passage of antibody via cord blood and breast milk,” said Linda Eckert, MD, a professor of ob.gyn. at The University of Washington, Seattle, who specializes in maternal immunization. “Further studies are important to look at the timing of vaccination in pregnancy and whether it influences the level of antibody passed to the fetus.”
Though this study is not a safety study, it “does not show increased expected vaccine reactions, such as aches, pains, and fever, in pregnant versus nonpregnant patients,” Dr. Eckert said in an interview. “It is not able to evaluate pregnancy outcome data, but it does allow pregnant women being vaccinated with the mRNA vaccines to know that the vaccine is generating protection for them, and the protection is being passed to the fetus in utero via cordblood and to the infant via breast milk.”
The research was funded by the National Institutes of Health along with the Gates Foundation, the Massachusetts Consortium on Pathogen Readiness (MassCPR), the Musk Foundation, the Ragon Institute of MGH and MIT, and Massachusetts General Hospital and Brigham and Women’s Hospital.
Lead author Dr. Gray has consulted for Illumina, BillionToOne, and Aetion, and three other authors have financial or scientific/medical advising connections to Alba Therapeutics, NextCure, Viome, Systems Seromyx, and Mirvie. Dr. Ault and Dr. Eckert had no disclosures.
FROM AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY
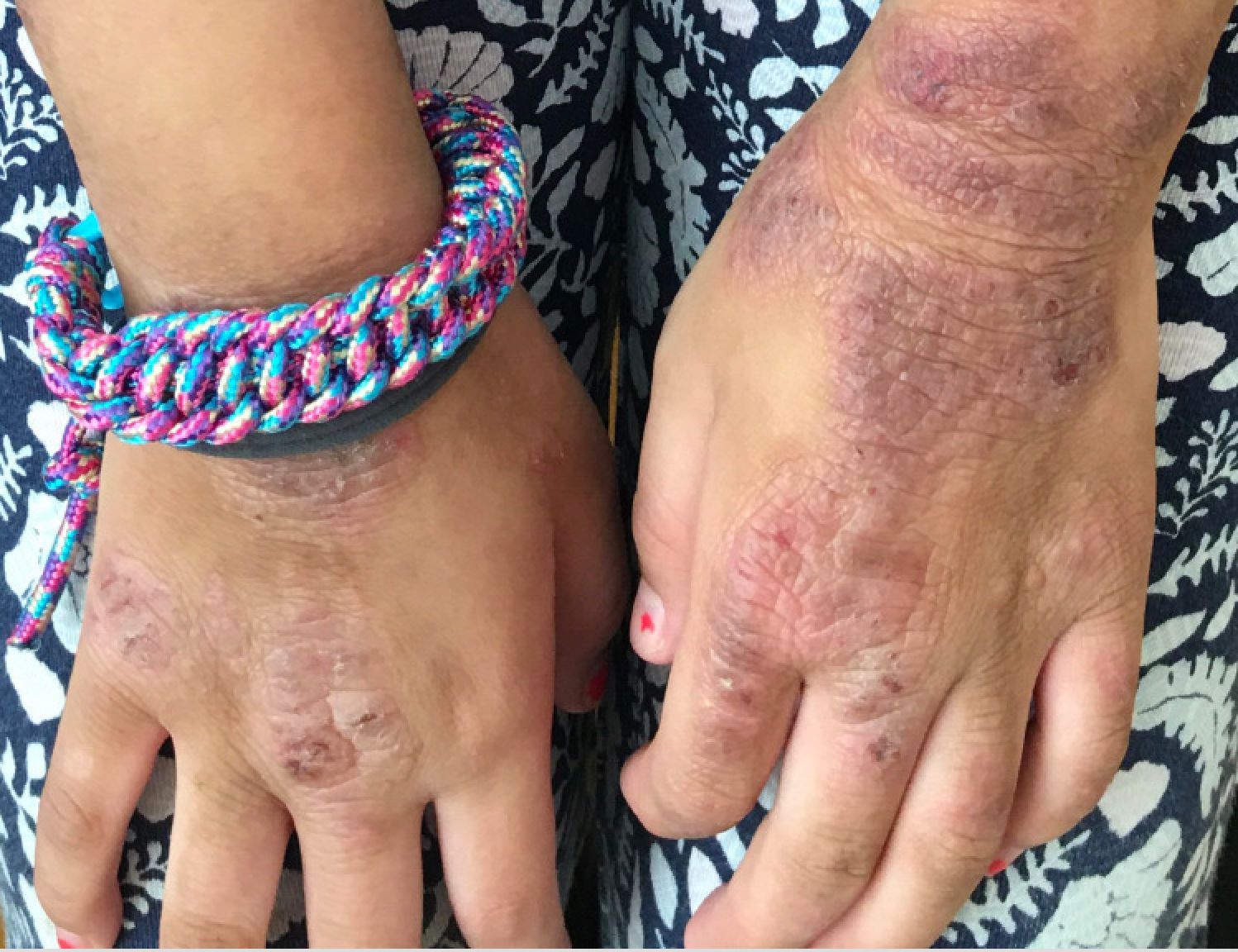
Contact allergen of the year found in foam in shin guards, footwear
.
The announcement was made by Donald V. Belsito, MD, professor of dermatology, Columbia University, New York, during a presentation at the annual meeting of the American Contact Dermatitis Society, held virtually this year. In his opinion, he said, the most exciting selections occur when international cooperation results in the identification of a new allergen that could become problematic, and acetophenone azine falls into this category.
The chemical formula of acetophenone azine is C16H16N2.
Acetophenone azine was highlighted as a contact allergen in a recent report in Dermatitis. The authors, Nadia Raison-Peyron, MD, from the department of dermatology at the University of Montpelier (France), and Denis Sasseville, MD, from the division of dermatology at McGill University Health Center, Quebec, described publications and reports of about 12 cases of severe allergic contact dermatitis secondary to shin pads or footwear, mainly in children and teens in Europe (one case was in Canada).
A common feature of these cases was the presence of a foam used for cushioning, made of ethyl vinyl acetate (EVA) used in the relevant products.
In one case, a 13-year-old boy who wore shin pads for soccer developed contact dermatitis on both shins that spread, and was described as severe. Patch testing revealed the EVA foam in the shin pads as the only positive reaction. Similar cases have been reported after exposure to EVA-containing products, including shin pads, sneakers, flip-flops, ski boots, insoles, swimming goggles, and bicycle seats, according to the authors.
In some reports, cases related to footwear presented as dyshidrosiform, vesiculobullous eczema, with or without palmar lesions, or presented as plantar hyperkeratotic dermatitis, they wrote. In other cases, patients experienced scarring and postinflammatory hypopigmentation.
The compound is likely not added to EVA intentionally, they added, but instead is thought to result from reactions between additives during the manufacturing process. The presence of acetophenone azine is not well explained, but the current theory is that it results from a combination of “the degradation of the initiator dicumylperoxide and hydrazine from the foaming agent azodicarbonamide,” the authors said.
In the paper, Dr. Raison-Peyron and Dr. Sasseville recommended a patch testing concentration of 0.1% in acetone or petrolatum, as acetophenone azine is not currently available from path test suppliers, although it can be obtained from chemical product distributors.
“Given the recent discovery of this allergen, it is presumed that cases of allergic contact dermatitis would have been missed and labeled irritant contact dermatitis or dyshidrosis,” they noted. To avoid missing more cases, acetophenone azine should be added to the patch testing shoe series, as well as plastics and glues series, they emphasized.
Although no cases of allergic reactions to acetophenone azine have been reported in the United States to date, it is an emerging allergen that should be on the radar for U.S. dermatologists, Amber Atwater, MD, outgoing ACDS president, said in an interview. The lack of reported cases may be in part attributed to the fact that acetophenone azine is not yet available to purchase for testing in the United States, and the allergen could be present in shin guards and other products identified in reported cases, added Dr. Atwater, associate professor of dermatology, Duke University, Durham, N.C.
.
The announcement was made by Donald V. Belsito, MD, professor of dermatology, Columbia University, New York, during a presentation at the annual meeting of the American Contact Dermatitis Society, held virtually this year. In his opinion, he said, the most exciting selections occur when international cooperation results in the identification of a new allergen that could become problematic, and acetophenone azine falls into this category.
The chemical formula of acetophenone azine is C16H16N2.
Acetophenone azine was highlighted as a contact allergen in a recent report in Dermatitis. The authors, Nadia Raison-Peyron, MD, from the department of dermatology at the University of Montpelier (France), and Denis Sasseville, MD, from the division of dermatology at McGill University Health Center, Quebec, described publications and reports of about 12 cases of severe allergic contact dermatitis secondary to shin pads or footwear, mainly in children and teens in Europe (one case was in Canada).
A common feature of these cases was the presence of a foam used for cushioning, made of ethyl vinyl acetate (EVA) used in the relevant products.
In one case, a 13-year-old boy who wore shin pads for soccer developed contact dermatitis on both shins that spread, and was described as severe. Patch testing revealed the EVA foam in the shin pads as the only positive reaction. Similar cases have been reported after exposure to EVA-containing products, including shin pads, sneakers, flip-flops, ski boots, insoles, swimming goggles, and bicycle seats, according to the authors.
In some reports, cases related to footwear presented as dyshidrosiform, vesiculobullous eczema, with or without palmar lesions, or presented as plantar hyperkeratotic dermatitis, they wrote. In other cases, patients experienced scarring and postinflammatory hypopigmentation.
The compound is likely not added to EVA intentionally, they added, but instead is thought to result from reactions between additives during the manufacturing process. The presence of acetophenone azine is not well explained, but the current theory is that it results from a combination of “the degradation of the initiator dicumylperoxide and hydrazine from the foaming agent azodicarbonamide,” the authors said.
In the paper, Dr. Raison-Peyron and Dr. Sasseville recommended a patch testing concentration of 0.1% in acetone or petrolatum, as acetophenone azine is not currently available from path test suppliers, although it can be obtained from chemical product distributors.
“Given the recent discovery of this allergen, it is presumed that cases of allergic contact dermatitis would have been missed and labeled irritant contact dermatitis or dyshidrosis,” they noted. To avoid missing more cases, acetophenone azine should be added to the patch testing shoe series, as well as plastics and glues series, they emphasized.
Although no cases of allergic reactions to acetophenone azine have been reported in the United States to date, it is an emerging allergen that should be on the radar for U.S. dermatologists, Amber Atwater, MD, outgoing ACDS president, said in an interview. The lack of reported cases may be in part attributed to the fact that acetophenone azine is not yet available to purchase for testing in the United States, and the allergen could be present in shin guards and other products identified in reported cases, added Dr. Atwater, associate professor of dermatology, Duke University, Durham, N.C.
.
The announcement was made by Donald V. Belsito, MD, professor of dermatology, Columbia University, New York, during a presentation at the annual meeting of the American Contact Dermatitis Society, held virtually this year. In his opinion, he said, the most exciting selections occur when international cooperation results in the identification of a new allergen that could become problematic, and acetophenone azine falls into this category.
The chemical formula of acetophenone azine is C16H16N2.
Acetophenone azine was highlighted as a contact allergen in a recent report in Dermatitis. The authors, Nadia Raison-Peyron, MD, from the department of dermatology at the University of Montpelier (France), and Denis Sasseville, MD, from the division of dermatology at McGill University Health Center, Quebec, described publications and reports of about 12 cases of severe allergic contact dermatitis secondary to shin pads or footwear, mainly in children and teens in Europe (one case was in Canada).
A common feature of these cases was the presence of a foam used for cushioning, made of ethyl vinyl acetate (EVA) used in the relevant products.
In one case, a 13-year-old boy who wore shin pads for soccer developed contact dermatitis on both shins that spread, and was described as severe. Patch testing revealed the EVA foam in the shin pads as the only positive reaction. Similar cases have been reported after exposure to EVA-containing products, including shin pads, sneakers, flip-flops, ski boots, insoles, swimming goggles, and bicycle seats, according to the authors.
In some reports, cases related to footwear presented as dyshidrosiform, vesiculobullous eczema, with or without palmar lesions, or presented as plantar hyperkeratotic dermatitis, they wrote. In other cases, patients experienced scarring and postinflammatory hypopigmentation.
The compound is likely not added to EVA intentionally, they added, but instead is thought to result from reactions between additives during the manufacturing process. The presence of acetophenone azine is not well explained, but the current theory is that it results from a combination of “the degradation of the initiator dicumylperoxide and hydrazine from the foaming agent azodicarbonamide,” the authors said.
In the paper, Dr. Raison-Peyron and Dr. Sasseville recommended a patch testing concentration of 0.1% in acetone or petrolatum, as acetophenone azine is not currently available from path test suppliers, although it can be obtained from chemical product distributors.
“Given the recent discovery of this allergen, it is presumed that cases of allergic contact dermatitis would have been missed and labeled irritant contact dermatitis or dyshidrosis,” they noted. To avoid missing more cases, acetophenone azine should be added to the patch testing shoe series, as well as plastics and glues series, they emphasized.
Although no cases of allergic reactions to acetophenone azine have been reported in the United States to date, it is an emerging allergen that should be on the radar for U.S. dermatologists, Amber Atwater, MD, outgoing ACDS president, said in an interview. The lack of reported cases may be in part attributed to the fact that acetophenone azine is not yet available to purchase for testing in the United States, and the allergen could be present in shin guards and other products identified in reported cases, added Dr. Atwater, associate professor of dermatology, Duke University, Durham, N.C.
FROM ACDS 2021
Here we go again? Rate of COVID-19 in children takes a turn for the worse
After declining for 8 consecutive weeks, new cases of COVID-19 rose among children in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.

, ending a streak of declines going back to mid-January, the AAP and CHA said in their weekly COVID-19 report.
Also up for the week was the proportion of all cases occurring in children. The 57,000-plus cases represented 18.7% of the total (304,610) for all ages, and that is the largest share of the new-case burden for the entire pandemic. The previous high, 18.0%, came just 2 weeks earlier, based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Speaking of the entire pandemic, the total number of COVID-19 cases in children is over 3.34 million, and that represents 13.3% of cases among all ages in the United States. The cumulative rate of infection as of March 18 was 4,440 cases per 100,000 children, up from 4,364 per 100,000 a week earlier, the AAP and CHA said.
At the state level, Vermont has now passed the 20% mark (20.1%, to be exact) for children’s proportion of cases and is higher in that measure than any other state. The highest rate of infection (8,763 cases per 100,000) can be found in North Dakota, the AAP/CHA data show.
There were only two new coronavirus-related deaths during the week of March 12-18 after Kansas revised its mortality data, bringing the total to 268 in the 46 jurisdictions (43 states, New York City, Puerto Rico, and Guam) that are reporting deaths by age, the AAP and CHA said.
After declining for 8 consecutive weeks, new cases of COVID-19 rose among children in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.

, ending a streak of declines going back to mid-January, the AAP and CHA said in their weekly COVID-19 report.
Also up for the week was the proportion of all cases occurring in children. The 57,000-plus cases represented 18.7% of the total (304,610) for all ages, and that is the largest share of the new-case burden for the entire pandemic. The previous high, 18.0%, came just 2 weeks earlier, based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Speaking of the entire pandemic, the total number of COVID-19 cases in children is over 3.34 million, and that represents 13.3% of cases among all ages in the United States. The cumulative rate of infection as of March 18 was 4,440 cases per 100,000 children, up from 4,364 per 100,000 a week earlier, the AAP and CHA said.
At the state level, Vermont has now passed the 20% mark (20.1%, to be exact) for children’s proportion of cases and is higher in that measure than any other state. The highest rate of infection (8,763 cases per 100,000) can be found in North Dakota, the AAP/CHA data show.
There were only two new coronavirus-related deaths during the week of March 12-18 after Kansas revised its mortality data, bringing the total to 268 in the 46 jurisdictions (43 states, New York City, Puerto Rico, and Guam) that are reporting deaths by age, the AAP and CHA said.
After declining for 8 consecutive weeks, new cases of COVID-19 rose among children in the United States, according to the American Academy of Pediatrics and the Children’s Hospital Association.

, ending a streak of declines going back to mid-January, the AAP and CHA said in their weekly COVID-19 report.
Also up for the week was the proportion of all cases occurring in children. The 57,000-plus cases represented 18.7% of the total (304,610) for all ages, and that is the largest share of the new-case burden for the entire pandemic. The previous high, 18.0%, came just 2 weeks earlier, based on data collected from 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam.
Speaking of the entire pandemic, the total number of COVID-19 cases in children is over 3.34 million, and that represents 13.3% of cases among all ages in the United States. The cumulative rate of infection as of March 18 was 4,440 cases per 100,000 children, up from 4,364 per 100,000 a week earlier, the AAP and CHA said.
At the state level, Vermont has now passed the 20% mark (20.1%, to be exact) for children’s proportion of cases and is higher in that measure than any other state. The highest rate of infection (8,763 cases per 100,000) can be found in North Dakota, the AAP/CHA data show.
There were only two new coronavirus-related deaths during the week of March 12-18 after Kansas revised its mortality data, bringing the total to 268 in the 46 jurisdictions (43 states, New York City, Puerto Rico, and Guam) that are reporting deaths by age, the AAP and CHA said.
Ruxolitinib cream for atopic dermatitis is in regulatory home stretch
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
, demonstrated a dual mechanism of action in two pivotal phase 3 trials: antipruritic and anti-inflammatory, Kim A. Papp, MD, PhD, said at Innovations in Dermatology: Virtual Spring Conference 2021. He presented a pooled analysis of the TRuE-AD1 and TRuE-AD2 trials, in which 1,249 patients with AD affecting 3%-20% of the body surface area were randomized 2:2:1 double-blind to ruxolitinib cream 0.75%, 1.5%, or vehicle twice daily for 8 weeks.
Striking evidence of the drug’s antipruritic effect comes from the finding that patient-reported itch scores separated significantly from the vehicle controls within just 12 hours after the first application. The margin of difference grew over time such that at 4 weeks, 48.5% of patients on ruxolitinib 1.5% experienced a clinically meaningful reduction in itch – defined by at least a 4-point improvement on the itch numeric rating scale – as did 30.1% of those on ruxolitinib 0.75% and 6.1% of controls. By week 8, these figures had further improved to 51.5%, 41.5%, and 15.8%, respectively, noted Dr. Papp, a dermatologist and president of Probity Medical Research in Waterloo, Ont.
Ruxolitinib’s anti-inflammatory mechanism of action was on display in the primary study endpoint, which was the proportion of patients achieving an Investigator Global Assessment score of 0 or 1 with at least a 2-grade improvement from baseline at week 8. The rates were 52.6% with ruxolitinib 1.5% and 44.7% at the lower dose, both significantly better than the 11.5% rate with vehicle.
For the secondary endpoint of at least a 75% improvement in Eczema Area and Severity Index score at week 8, the rates were 62% with ruxolitinib 1.5% and 53.8% at the 0.75% concentration, compared with 19.7% with vehicle.
The topical JAK inhibitor also showed superior efficacy in terms of improvement on the Patient-Reported Outcomes Measurement Information System Sleep Disturbance Score, with a clinically meaningful 6-point or greater improvement in 23.9% and 20.9% of patients in the high- and low-dose ruxolitinib groups, versus 14.2% in controls.
Plasma drug levels remained consistently low and near-flat throughout the study.
Session comoderator Lawrence F. Eichenfield, MD, was struck by what he termed the “incredibly low” rates of irritancy, burning, and stinging in the ruxolitinib-treated patients: 7 cases of application-site burning in 999 treated patients, compared with 11 cases in 250 vehicle-treated patients, and 4 cases of application-site pruritus in nearly 1,000 patients on ruxolitinib, versus 6 cases in one-fourth as many controls.
“If that’s really true in clinical practice, it would be tremendous to have a nonsteroid that doesn’t have stinging and burning and may have that efficacy,” said Dr. Eichenfield, professor of dermatology and pediatrics and vice-chair of dermatology at the University of California, San Diego.
“I think the fast action is an exciting aspect of this,” said comoderator Jonathan I. Silverberg, MD, PhD, MBA, director of clinical research and contact dermatitis in the department of dermatology at George Washington University in Washington.
He noted that in an earlier phase 2 study, ruxolitinib cream was at least as efficacious as 0.1% triamcinolone cream, providing dermatologists with a rough yardstick as to where the topical JAK inhibitor lies on the potency spectrum for AD treatment.
The FDA is expected to issue a decision on the application for approval of ruxolitinib cream in June. Dr. Eichenfield expects the drug to easily win approval. The big unanswered question is whether the regulatory agency will require boxed safety warnings, as it does for the oral JAK inhibitors approved for various indications, even though safety issues haven’t arisen with the topical agent in the clinical trials.
Dr. Papp reported receiving research grants from and serving as a consultant to Incyte Corp., which funded the ruxolitinib studies, as well as numerous other pharmaceutical companies. MedscapeLive and this news organization are owned by the same parent company.
FROM INNOVATIONS IN DERMATOLOGY
THC persists in breast milk 6 weeks after quitting cannabis
Delta-9-Tetrahydrocannabinol (THC), the main psychoactive component of cannabis, remains detectable in breast milk even after weeks of abstinence, new data show. The estimated half-life of THC in breast milk is 17 days, according to the study results, with a projected time to elimination of more than 6 weeks. The clinical importance of the remaining THC is up for debate, according to some experts.
“To limit THC effects on fetal brain development and promote safe breastfeeding, it is critical to emphasize marijuana abstention both early in pregnancy and post partum,” Erica M. Wymore, MD, MPH, an assistant professor of pediatrics and neonatology at the University of Colorado at Denver, Aurora, and colleagues wrote. The group published their results online March 8, 2021, in JAMA Pediatrics.
And while the study was a pharmacokinetic analysis rather than a safety investigation, Dr. Wymore said in an interview that the detectable levels of THC suggest any use is of concern and no safety thresholds have been established. “We wish we had more data on the potential effects on the neurocognitive development of children, but for now we must discourage any use in prepregnancy, pregnancy, and breastfeeding, as our national guidelines recommend.”
Therefore, the findings support current guidelines discouraging any cannabis use in mothers-to-be and breast-feeding mothers issued by national organizations, including those from the American Academy of Pediatrics, the American College of Obstetricians and Gynecologists, and the Academy of Breastfeeding Medicine.
Furthermore, the difficulties many mothers face in abstaining from marijuana, a commonly used drug in pregnancy, and the persistence of THC in maternal milk led the authors to question the feasibility of having women who use marijuana simply discard their breast milk until THC is cleared.
“We report challenges in abstention and prolonged excretion of THC in breast milk greater than 6 weeks among women with prenatal marijuana use,” they wrote. “These findings make the recommendations for mothers to discard breast milk until THC is undetectable unrealistic for mothers committed to breastfeeding.”
However, not all experts are equally concerned about low THC concentrations in breast milk. Neonatal pharmacologist Thomas R. Hale, PhD, a professor of pediatrics at Texas Tech University, Lubbock, said a previous study by his group showed that THC levels in maternal milk peaked within 60 minutes of a moderate dose of inhaled marijuana and fell to quite low levels over the next 4 hours. The highest concentration in maternal milk occurred shortly after the peak in plasma.
“So you can see that, just because a mom is drug screen positive, the clinical dose transferred to the infant is probably exceedingly low,” he said in an interview.
Dr. Hale also stressed that judgments about drugs in this context should weigh the risk of the drug against the risk of not breastfeeding. “All of us caution women not to use cannabis when pregnant or breastfeeding,” Dr. Hale said. “But when the decision has to be made as to whether a mom breastfeeds or not if she is drug screen positive, a lot of other factors must be analyzed to make such a decision.”
Study cohort
For the study, Dr. Wymore and colleagues screened 394 women who gave birth between Nov. 1, 2016, and June 30, 2019. Of those, 25 women, with a median age of 26 years, were eligible and enrolled. Inclusion criteria included known prenatal marijuana use, intention to breastfeed, and self-reported abstinence. Prenatal use primarily involved inhaling cannabis more than twice a week.
Of the 25 enrolled mothers, 12 who self-reported marijuana abstinence were in fact found to be abstinent according to the results of plasma analysis. Those who continued to use the substance were younger than the overall sample, with a median age of 21, and were less likely to have attended college (23%) than abstainers (58%).
The researchers prospectively collected data on self-reported marijuana usage and paired maternal plasma and breast milk samples several times a week. All participants had detectable THC in breast milk throughout the study. Initial median THC concentrations were 3.2 ng/mL (interquartile range, 1.2-6.8) within the first week after delivery. These increased to 5.5 ng/mL (IQR, 4.4-16.0) at 2 weeks and declined to 1.9 ng/mL (IQR, 1.1-4.3) at 6 weeks. In terms of ratio, the milk:plasma partition coefficient for THC was approximately 6:1 (IQR, 3.8:1-8.1:1).
Dr. Hale noted that, although THC was detectable in milk, the levels were exceedingly low. “This is where the risk assessment comes in. There’s a lot of hysteria in the cannabis field right now, and we’re going to need time and a lot more studies to really be able to predict any untoward complications.”
Dr. Wymore, however, countered that THC levels were low only in those who abstained and that her concerns relate not just to postpartum breast milk levels but the health effects on children of mothers’ cannabis use over the course of prepregnancy, pregnancy, and lactation. “[Dr. Hale’s] message makes it difficult for clinicians to counsel mothers since it goes against national guidelines,” she said. “We need to be consistent.”
But Dr. Wymore and other experts acknowledge the dilemma faced in that breast milk clearly offers substantial benefits for infant and child health. “The risks of an infant’s exposure to marijuana versus the benefit of breast milk must be considered,” said Amy B. Hair, MD, assistant professor of pediatrics and neonatal medicine at Baylor College of Medicine, Houston, who was not involved in the Colorado study. “And it’s unrealistic, as the study suggests, for mothers to discard breast milk for 6 weeks.”
Nevertheless, calling the findings of THC persistence after abstinence “troublesome,” Dr. Hair said the legalization of marijuana in some states gives the public the impression it’s safe to use marijuana even during pregnancy and lactation. “Research studies, however, are concerning for potential detrimental effects on brain growth and development in infants whose mothers use marijuana during pregnancy and breastfeeding,” she added.
Dr. Wymore stressed that more U.S. cannabis dispensaries must engage in rigorous point-of-sale counseling to women on the potential harms during pregnancy. This is the case in Canada, she noted, where recreational and medicinal cannabis has been legal since 2018 and more than 90% of outlets (vs. two thirds of their U.S. counterparts) advise women not to use cannabis during pregnancy or lactation, even for nausea.
“This is where many women are getting their information on cannabis,” she said. “We learned the hard way with alcohol and we don’t want to make the same mistake with marijuana.”
The study was funded by the Colorado Department of Public Health and Environment, the Children’s Hospital Colorado Research Institute, the Colorado Fetal Care Center, the Colorado Perinatal Clinical and Translational Research Center, and the Children’s Colorado Research Institute. Two study coauthors disclosed relationships with the private sector outside the submitted work. Dr. Hale and Dr. Hair have disclosed no competing interests with regard to their comments.
A version of this article first appeared on Medscape.com.
Delta-9-Tetrahydrocannabinol (THC), the main psychoactive component of cannabis, remains detectable in breast milk even after weeks of abstinence, new data show. The estimated half-life of THC in breast milk is 17 days, according to the study results, with a projected time to elimination of more than 6 weeks. The clinical importance of the remaining THC is up for debate, according to some experts.
“To limit THC effects on fetal brain development and promote safe breastfeeding, it is critical to emphasize marijuana abstention both early in pregnancy and post partum,” Erica M. Wymore, MD, MPH, an assistant professor of pediatrics and neonatology at the University of Colorado at Denver, Aurora, and colleagues wrote. The group published their results online March 8, 2021, in JAMA Pediatrics.
And while the study was a pharmacokinetic analysis rather than a safety investigation, Dr. Wymore said in an interview that the detectable levels of THC suggest any use is of concern and no safety thresholds have been established. “We wish we had more data on the potential effects on the neurocognitive development of children, but for now we must discourage any use in prepregnancy, pregnancy, and breastfeeding, as our national guidelines recommend.”
Therefore, the findings support current guidelines discouraging any cannabis use in mothers-to-be and breast-feeding mothers issued by national organizations, including those from the American Academy of Pediatrics, the American College of Obstetricians and Gynecologists, and the Academy of Breastfeeding Medicine.
Furthermore, the difficulties many mothers face in abstaining from marijuana, a commonly used drug in pregnancy, and the persistence of THC in maternal milk led the authors to question the feasibility of having women who use marijuana simply discard their breast milk until THC is cleared.
“We report challenges in abstention and prolonged excretion of THC in breast milk greater than 6 weeks among women with prenatal marijuana use,” they wrote. “These findings make the recommendations for mothers to discard breast milk until THC is undetectable unrealistic for mothers committed to breastfeeding.”
However, not all experts are equally concerned about low THC concentrations in breast milk. Neonatal pharmacologist Thomas R. Hale, PhD, a professor of pediatrics at Texas Tech University, Lubbock, said a previous study by his group showed that THC levels in maternal milk peaked within 60 minutes of a moderate dose of inhaled marijuana and fell to quite low levels over the next 4 hours. The highest concentration in maternal milk occurred shortly after the peak in plasma.
“So you can see that, just because a mom is drug screen positive, the clinical dose transferred to the infant is probably exceedingly low,” he said in an interview.
Dr. Hale also stressed that judgments about drugs in this context should weigh the risk of the drug against the risk of not breastfeeding. “All of us caution women not to use cannabis when pregnant or breastfeeding,” Dr. Hale said. “But when the decision has to be made as to whether a mom breastfeeds or not if she is drug screen positive, a lot of other factors must be analyzed to make such a decision.”
Study cohort
For the study, Dr. Wymore and colleagues screened 394 women who gave birth between Nov. 1, 2016, and June 30, 2019. Of those, 25 women, with a median age of 26 years, were eligible and enrolled. Inclusion criteria included known prenatal marijuana use, intention to breastfeed, and self-reported abstinence. Prenatal use primarily involved inhaling cannabis more than twice a week.
Of the 25 enrolled mothers, 12 who self-reported marijuana abstinence were in fact found to be abstinent according to the results of plasma analysis. Those who continued to use the substance were younger than the overall sample, with a median age of 21, and were less likely to have attended college (23%) than abstainers (58%).
The researchers prospectively collected data on self-reported marijuana usage and paired maternal plasma and breast milk samples several times a week. All participants had detectable THC in breast milk throughout the study. Initial median THC concentrations were 3.2 ng/mL (interquartile range, 1.2-6.8) within the first week after delivery. These increased to 5.5 ng/mL (IQR, 4.4-16.0) at 2 weeks and declined to 1.9 ng/mL (IQR, 1.1-4.3) at 6 weeks. In terms of ratio, the milk:plasma partition coefficient for THC was approximately 6:1 (IQR, 3.8:1-8.1:1).
Dr. Hale noted that, although THC was detectable in milk, the levels were exceedingly low. “This is where the risk assessment comes in. There’s a lot of hysteria in the cannabis field right now, and we’re going to need time and a lot more studies to really be able to predict any untoward complications.”
Dr. Wymore, however, countered that THC levels were low only in those who abstained and that her concerns relate not just to postpartum breast milk levels but the health effects on children of mothers’ cannabis use over the course of prepregnancy, pregnancy, and lactation. “[Dr. Hale’s] message makes it difficult for clinicians to counsel mothers since it goes against national guidelines,” she said. “We need to be consistent.”
But Dr. Wymore and other experts acknowledge the dilemma faced in that breast milk clearly offers substantial benefits for infant and child health. “The risks of an infant’s exposure to marijuana versus the benefit of breast milk must be considered,” said Amy B. Hair, MD, assistant professor of pediatrics and neonatal medicine at Baylor College of Medicine, Houston, who was not involved in the Colorado study. “And it’s unrealistic, as the study suggests, for mothers to discard breast milk for 6 weeks.”
Nevertheless, calling the findings of THC persistence after abstinence “troublesome,” Dr. Hair said the legalization of marijuana in some states gives the public the impression it’s safe to use marijuana even during pregnancy and lactation. “Research studies, however, are concerning for potential detrimental effects on brain growth and development in infants whose mothers use marijuana during pregnancy and breastfeeding,” she added.
Dr. Wymore stressed that more U.S. cannabis dispensaries must engage in rigorous point-of-sale counseling to women on the potential harms during pregnancy. This is the case in Canada, she noted, where recreational and medicinal cannabis has been legal since 2018 and more than 90% of outlets (vs. two thirds of their U.S. counterparts) advise women not to use cannabis during pregnancy or lactation, even for nausea.
“This is where many women are getting their information on cannabis,” she said. “We learned the hard way with alcohol and we don’t want to make the same mistake with marijuana.”
The study was funded by the Colorado Department of Public Health and Environment, the Children’s Hospital Colorado Research Institute, the Colorado Fetal Care Center, the Colorado Perinatal Clinical and Translational Research Center, and the Children’s Colorado Research Institute. Two study coauthors disclosed relationships with the private sector outside the submitted work. Dr. Hale and Dr. Hair have disclosed no competing interests with regard to their comments.
A version of this article first appeared on Medscape.com.
Delta-9-Tetrahydrocannabinol (THC), the main psychoactive component of cannabis, remains detectable in breast milk even after weeks of abstinence, new data show. The estimated half-life of THC in breast milk is 17 days, according to the study results, with a projected time to elimination of more than 6 weeks. The clinical importance of the remaining THC is up for debate, according to some experts.
“To limit THC effects on fetal brain development and promote safe breastfeeding, it is critical to emphasize marijuana abstention both early in pregnancy and post partum,” Erica M. Wymore, MD, MPH, an assistant professor of pediatrics and neonatology at the University of Colorado at Denver, Aurora, and colleagues wrote. The group published their results online March 8, 2021, in JAMA Pediatrics.
And while the study was a pharmacokinetic analysis rather than a safety investigation, Dr. Wymore said in an interview that the detectable levels of THC suggest any use is of concern and no safety thresholds have been established. “We wish we had more data on the potential effects on the neurocognitive development of children, but for now we must discourage any use in prepregnancy, pregnancy, and breastfeeding, as our national guidelines recommend.”
Therefore, the findings support current guidelines discouraging any cannabis use in mothers-to-be and breast-feeding mothers issued by national organizations, including those from the American Academy of Pediatrics, the American College of Obstetricians and Gynecologists, and the Academy of Breastfeeding Medicine.
Furthermore, the difficulties many mothers face in abstaining from marijuana, a commonly used drug in pregnancy, and the persistence of THC in maternal milk led the authors to question the feasibility of having women who use marijuana simply discard their breast milk until THC is cleared.
“We report challenges in abstention and prolonged excretion of THC in breast milk greater than 6 weeks among women with prenatal marijuana use,” they wrote. “These findings make the recommendations for mothers to discard breast milk until THC is undetectable unrealistic for mothers committed to breastfeeding.”
However, not all experts are equally concerned about low THC concentrations in breast milk. Neonatal pharmacologist Thomas R. Hale, PhD, a professor of pediatrics at Texas Tech University, Lubbock, said a previous study by his group showed that THC levels in maternal milk peaked within 60 minutes of a moderate dose of inhaled marijuana and fell to quite low levels over the next 4 hours. The highest concentration in maternal milk occurred shortly after the peak in plasma.
“So you can see that, just because a mom is drug screen positive, the clinical dose transferred to the infant is probably exceedingly low,” he said in an interview.
Dr. Hale also stressed that judgments about drugs in this context should weigh the risk of the drug against the risk of not breastfeeding. “All of us caution women not to use cannabis when pregnant or breastfeeding,” Dr. Hale said. “But when the decision has to be made as to whether a mom breastfeeds or not if she is drug screen positive, a lot of other factors must be analyzed to make such a decision.”
Study cohort
For the study, Dr. Wymore and colleagues screened 394 women who gave birth between Nov. 1, 2016, and June 30, 2019. Of those, 25 women, with a median age of 26 years, were eligible and enrolled. Inclusion criteria included known prenatal marijuana use, intention to breastfeed, and self-reported abstinence. Prenatal use primarily involved inhaling cannabis more than twice a week.
Of the 25 enrolled mothers, 12 who self-reported marijuana abstinence were in fact found to be abstinent according to the results of plasma analysis. Those who continued to use the substance were younger than the overall sample, with a median age of 21, and were less likely to have attended college (23%) than abstainers (58%).
The researchers prospectively collected data on self-reported marijuana usage and paired maternal plasma and breast milk samples several times a week. All participants had detectable THC in breast milk throughout the study. Initial median THC concentrations were 3.2 ng/mL (interquartile range, 1.2-6.8) within the first week after delivery. These increased to 5.5 ng/mL (IQR, 4.4-16.0) at 2 weeks and declined to 1.9 ng/mL (IQR, 1.1-4.3) at 6 weeks. In terms of ratio, the milk:plasma partition coefficient for THC was approximately 6:1 (IQR, 3.8:1-8.1:1).
Dr. Hale noted that, although THC was detectable in milk, the levels were exceedingly low. “This is where the risk assessment comes in. There’s a lot of hysteria in the cannabis field right now, and we’re going to need time and a lot more studies to really be able to predict any untoward complications.”
Dr. Wymore, however, countered that THC levels were low only in those who abstained and that her concerns relate not just to postpartum breast milk levels but the health effects on children of mothers’ cannabis use over the course of prepregnancy, pregnancy, and lactation. “[Dr. Hale’s] message makes it difficult for clinicians to counsel mothers since it goes against national guidelines,” she said. “We need to be consistent.”
But Dr. Wymore and other experts acknowledge the dilemma faced in that breast milk clearly offers substantial benefits for infant and child health. “The risks of an infant’s exposure to marijuana versus the benefit of breast milk must be considered,” said Amy B. Hair, MD, assistant professor of pediatrics and neonatal medicine at Baylor College of Medicine, Houston, who was not involved in the Colorado study. “And it’s unrealistic, as the study suggests, for mothers to discard breast milk for 6 weeks.”
Nevertheless, calling the findings of THC persistence after abstinence “troublesome,” Dr. Hair said the legalization of marijuana in some states gives the public the impression it’s safe to use marijuana even during pregnancy and lactation. “Research studies, however, are concerning for potential detrimental effects on brain growth and development in infants whose mothers use marijuana during pregnancy and breastfeeding,” she added.
Dr. Wymore stressed that more U.S. cannabis dispensaries must engage in rigorous point-of-sale counseling to women on the potential harms during pregnancy. This is the case in Canada, she noted, where recreational and medicinal cannabis has been legal since 2018 and more than 90% of outlets (vs. two thirds of their U.S. counterparts) advise women not to use cannabis during pregnancy or lactation, even for nausea.
“This is where many women are getting their information on cannabis,” she said. “We learned the hard way with alcohol and we don’t want to make the same mistake with marijuana.”
The study was funded by the Colorado Department of Public Health and Environment, the Children’s Hospital Colorado Research Institute, the Colorado Fetal Care Center, the Colorado Perinatal Clinical and Translational Research Center, and the Children’s Colorado Research Institute. Two study coauthors disclosed relationships with the private sector outside the submitted work. Dr. Hale and Dr. Hair have disclosed no competing interests with regard to their comments.
A version of this article first appeared on Medscape.com.
Comic books help explain type 1 diabetes to all ages
Overcoming the challenges in managing type 1 diabetes can sometimes feel like an unappreciated “superpower.” That was part of the thinking behind the creation of a comic book trilogy that aims to educate people of all ages – including health care providers – about the realities of living with this condition.
The series was initially launched by a team from Portsmouth (England) Hospitals University National Health Service Trust and University Hospital Southampton NHS Foundation Trust. It is now officially backed by the NHS. The first book in the trilogy, published in 2016, visually illustrates the challenges faced by a teenage boy who had recently been diagnosed with type 1 diabetes. The second volume, released in 2018, follows a young girl who is hospitalized with diabetic ketoacidosis. The third, published in December 2020, explores the stigma associated with diabetes and delves into hypoglycemia.
Available for free online, the three comic books are meant for adults, children, health care professionals, and laypeople. This news organization spoke with series cocreator Partha Kar, MBBS, MD, national specialty adviser, Diabetes for NHS England, about the series. This interview has been edited for length and clarity.
How did the idea for a comic book series about type 1 diabetes come about?Dr. Kar: My Southampton colleague Mayank Patel, BM, DM, FRCP, and I were discussing ways of reaching different audiences to raise awareness about type 1 diabetes, and we had the idea of comic books. After all, comic book movies are among the biggest blockbusters if one looks at popular culture, because it’s not just kids watching them.
One of our patients made an interesting observation that really resonated. He said having type 1 diabetes was like the Marvel Comics superhero Hulk.
Several scenes in the first publication, Type 1: Origins, were based on the Hulk, a scientist who gets a radioactive dose by accident. He doesn’t like turning green when he’s angry, even though he also becomes very strong. He basically spends the rest of his life trying to find the cure for himself, but he eventually makes the best of his two worlds – Professor and Hulk – rather than constantly fighting his situation.
The story line was primarily written by a group of patients with type 1 diabetes based on their own experiences. Mayank and I were mostly just supervising and financing the project. The graphics and layout were done by Revolve Comics, a publisher specializing in health education via the comic book medium.
Our aim was to bring awareness of type 1 diabetes to people who don’t have diabetes, including teachers, family members, and friends. At the end of Origins, we provide a list of online resources for more information and for social connection.
Since it launched in October 2016, Origins has been downloaded nearly 10,000 times. Lots of local charities and schools have picked it up. Parents and kids have come to us asking for more and giving us ideas. That’s what prompted the next one.
The second volume, Type 1: Attack of the Ketones, is more technical and somewhat surprising in that it portrays some hospital staff members as not well-informed about type 1 diabetes. Are they part of the intended audience?
Yes, this one was directed a little bit more towards professionals, hospitals, and staff. It’s also informed by patient feedback, and dovetails with my efforts to improve hospital care for people with type 1 diabetes. But of course, patients and interested laypeople can also learn from it.
A theme in volume 2 comes from another Marvel Comics superhero, Iron Man. In the movie, when Tony Stark’s heart is severely damaged with shrapnel, he acquires an arc reactor that keeps him alive and also powers the suit that gives him superpowers. After the reactor is taken away, he devises a way to replace the missing part and reassemble the suit.
Similarly, in type 1 diabetes, the ability to produce insulin has been taken away without permission. But what is missing can thankfully be replaced, albeit imperfectly. As we illustrate, things don’t always go to plan despite best efforts to administer insulin in the right dose at the right time.
At the end of Attack of the Ketones, we provide two pages of text about recognizing and managing hyperglycemia and preventing diabetic ketoacidosis. This volume was funded by NHS England and then backed by JDRF and Diabetes UK, and many hospitals picked it up. It has had about 8,000 downloads.
In Volume 3, you explore stigma and the issue of language regarding type 1 diabetes. How did those topics come about?
Kar: Type 1 Mission 3: S.T.I.G.M.A. was also based on patient feedback, with input from some Indian diabetes groups I’ve worked with. Here, the protagonist is a young man with type 1 diabetes who goes on holiday to India, where diabetes stigma is widespread. The characters address language problems such as use of the word “diabetic” to label a person, and they counter misconceptions such as that diabetes is contagious. There’s an Indian comic book version of this volume out now.
The main character of this volume experiences severe hypoglycemia and is saved by a glucagon injection from a colleague, one of several presented as superheroes who help in the fight to end diabetes stigma. They are referred to as Guardians of the Glucose, a take on yet another Marvel franchise, Guardians of the Galaxy.
At the end of this volume, we provide two pages of text about recognizing, managing, and preventing hypoglycemia. Again, we hope to educate as wide an audience as possible.
At the end of volume 3, you also briefly mention the COVID-19 pandemic. Will there be a fourth volume dealing with that, or other topics, such as diabetes technology?
We’ve left it open. We want to see how volume 3 lands. Depending on that, we might take it forward. There are certainly plenty of topics to tackle. We’ve also discussed moving into gaming or virtual reality. Overall, we hope to educate people by engaging them in different ways.
Dr. Kar has been a consultant diabetologist/endocrinologist within the NHS since 2008. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Overcoming the challenges in managing type 1 diabetes can sometimes feel like an unappreciated “superpower.” That was part of the thinking behind the creation of a comic book trilogy that aims to educate people of all ages – including health care providers – about the realities of living with this condition.
The series was initially launched by a team from Portsmouth (England) Hospitals University National Health Service Trust and University Hospital Southampton NHS Foundation Trust. It is now officially backed by the NHS. The first book in the trilogy, published in 2016, visually illustrates the challenges faced by a teenage boy who had recently been diagnosed with type 1 diabetes. The second volume, released in 2018, follows a young girl who is hospitalized with diabetic ketoacidosis. The third, published in December 2020, explores the stigma associated with diabetes and delves into hypoglycemia.
Available for free online, the three comic books are meant for adults, children, health care professionals, and laypeople. This news organization spoke with series cocreator Partha Kar, MBBS, MD, national specialty adviser, Diabetes for NHS England, about the series. This interview has been edited for length and clarity.
How did the idea for a comic book series about type 1 diabetes come about?Dr. Kar: My Southampton colleague Mayank Patel, BM, DM, FRCP, and I were discussing ways of reaching different audiences to raise awareness about type 1 diabetes, and we had the idea of comic books. After all, comic book movies are among the biggest blockbusters if one looks at popular culture, because it’s not just kids watching them.
One of our patients made an interesting observation that really resonated. He said having type 1 diabetes was like the Marvel Comics superhero Hulk.
Several scenes in the first publication, Type 1: Origins, were based on the Hulk, a scientist who gets a radioactive dose by accident. He doesn’t like turning green when he’s angry, even though he also becomes very strong. He basically spends the rest of his life trying to find the cure for himself, but he eventually makes the best of his two worlds – Professor and Hulk – rather than constantly fighting his situation.
The story line was primarily written by a group of patients with type 1 diabetes based on their own experiences. Mayank and I were mostly just supervising and financing the project. The graphics and layout were done by Revolve Comics, a publisher specializing in health education via the comic book medium.
Our aim was to bring awareness of type 1 diabetes to people who don’t have diabetes, including teachers, family members, and friends. At the end of Origins, we provide a list of online resources for more information and for social connection.
Since it launched in October 2016, Origins has been downloaded nearly 10,000 times. Lots of local charities and schools have picked it up. Parents and kids have come to us asking for more and giving us ideas. That’s what prompted the next one.
The second volume, Type 1: Attack of the Ketones, is more technical and somewhat surprising in that it portrays some hospital staff members as not well-informed about type 1 diabetes. Are they part of the intended audience?
Yes, this one was directed a little bit more towards professionals, hospitals, and staff. It’s also informed by patient feedback, and dovetails with my efforts to improve hospital care for people with type 1 diabetes. But of course, patients and interested laypeople can also learn from it.
A theme in volume 2 comes from another Marvel Comics superhero, Iron Man. In the movie, when Tony Stark’s heart is severely damaged with shrapnel, he acquires an arc reactor that keeps him alive and also powers the suit that gives him superpowers. After the reactor is taken away, he devises a way to replace the missing part and reassemble the suit.
Similarly, in type 1 diabetes, the ability to produce insulin has been taken away without permission. But what is missing can thankfully be replaced, albeit imperfectly. As we illustrate, things don’t always go to plan despite best efforts to administer insulin in the right dose at the right time.
At the end of Attack of the Ketones, we provide two pages of text about recognizing and managing hyperglycemia and preventing diabetic ketoacidosis. This volume was funded by NHS England and then backed by JDRF and Diabetes UK, and many hospitals picked it up. It has had about 8,000 downloads.
In Volume 3, you explore stigma and the issue of language regarding type 1 diabetes. How did those topics come about?
Kar: Type 1 Mission 3: S.T.I.G.M.A. was also based on patient feedback, with input from some Indian diabetes groups I’ve worked with. Here, the protagonist is a young man with type 1 diabetes who goes on holiday to India, where diabetes stigma is widespread. The characters address language problems such as use of the word “diabetic” to label a person, and they counter misconceptions such as that diabetes is contagious. There’s an Indian comic book version of this volume out now.
The main character of this volume experiences severe hypoglycemia and is saved by a glucagon injection from a colleague, one of several presented as superheroes who help in the fight to end diabetes stigma. They are referred to as Guardians of the Glucose, a take on yet another Marvel franchise, Guardians of the Galaxy.
At the end of this volume, we provide two pages of text about recognizing, managing, and preventing hypoglycemia. Again, we hope to educate as wide an audience as possible.
At the end of volume 3, you also briefly mention the COVID-19 pandemic. Will there be a fourth volume dealing with that, or other topics, such as diabetes technology?
We’ve left it open. We want to see how volume 3 lands. Depending on that, we might take it forward. There are certainly plenty of topics to tackle. We’ve also discussed moving into gaming or virtual reality. Overall, we hope to educate people by engaging them in different ways.
Dr. Kar has been a consultant diabetologist/endocrinologist within the NHS since 2008. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Overcoming the challenges in managing type 1 diabetes can sometimes feel like an unappreciated “superpower.” That was part of the thinking behind the creation of a comic book trilogy that aims to educate people of all ages – including health care providers – about the realities of living with this condition.
The series was initially launched by a team from Portsmouth (England) Hospitals University National Health Service Trust and University Hospital Southampton NHS Foundation Trust. It is now officially backed by the NHS. The first book in the trilogy, published in 2016, visually illustrates the challenges faced by a teenage boy who had recently been diagnosed with type 1 diabetes. The second volume, released in 2018, follows a young girl who is hospitalized with diabetic ketoacidosis. The third, published in December 2020, explores the stigma associated with diabetes and delves into hypoglycemia.
Available for free online, the three comic books are meant for adults, children, health care professionals, and laypeople. This news organization spoke with series cocreator Partha Kar, MBBS, MD, national specialty adviser, Diabetes for NHS England, about the series. This interview has been edited for length and clarity.
How did the idea for a comic book series about type 1 diabetes come about?Dr. Kar: My Southampton colleague Mayank Patel, BM, DM, FRCP, and I were discussing ways of reaching different audiences to raise awareness about type 1 diabetes, and we had the idea of comic books. After all, comic book movies are among the biggest blockbusters if one looks at popular culture, because it’s not just kids watching them.
One of our patients made an interesting observation that really resonated. He said having type 1 diabetes was like the Marvel Comics superhero Hulk.
Several scenes in the first publication, Type 1: Origins, were based on the Hulk, a scientist who gets a radioactive dose by accident. He doesn’t like turning green when he’s angry, even though he also becomes very strong. He basically spends the rest of his life trying to find the cure for himself, but he eventually makes the best of his two worlds – Professor and Hulk – rather than constantly fighting his situation.
The story line was primarily written by a group of patients with type 1 diabetes based on their own experiences. Mayank and I were mostly just supervising and financing the project. The graphics and layout were done by Revolve Comics, a publisher specializing in health education via the comic book medium.
Our aim was to bring awareness of type 1 diabetes to people who don’t have diabetes, including teachers, family members, and friends. At the end of Origins, we provide a list of online resources for more information and for social connection.
Since it launched in October 2016, Origins has been downloaded nearly 10,000 times. Lots of local charities and schools have picked it up. Parents and kids have come to us asking for more and giving us ideas. That’s what prompted the next one.
The second volume, Type 1: Attack of the Ketones, is more technical and somewhat surprising in that it portrays some hospital staff members as not well-informed about type 1 diabetes. Are they part of the intended audience?
Yes, this one was directed a little bit more towards professionals, hospitals, and staff. It’s also informed by patient feedback, and dovetails with my efforts to improve hospital care for people with type 1 diabetes. But of course, patients and interested laypeople can also learn from it.
A theme in volume 2 comes from another Marvel Comics superhero, Iron Man. In the movie, when Tony Stark’s heart is severely damaged with shrapnel, he acquires an arc reactor that keeps him alive and also powers the suit that gives him superpowers. After the reactor is taken away, he devises a way to replace the missing part and reassemble the suit.
Similarly, in type 1 diabetes, the ability to produce insulin has been taken away without permission. But what is missing can thankfully be replaced, albeit imperfectly. As we illustrate, things don’t always go to plan despite best efforts to administer insulin in the right dose at the right time.
At the end of Attack of the Ketones, we provide two pages of text about recognizing and managing hyperglycemia and preventing diabetic ketoacidosis. This volume was funded by NHS England and then backed by JDRF and Diabetes UK, and many hospitals picked it up. It has had about 8,000 downloads.
In Volume 3, you explore stigma and the issue of language regarding type 1 diabetes. How did those topics come about?
Kar: Type 1 Mission 3: S.T.I.G.M.A. was also based on patient feedback, with input from some Indian diabetes groups I’ve worked with. Here, the protagonist is a young man with type 1 diabetes who goes on holiday to India, where diabetes stigma is widespread. The characters address language problems such as use of the word “diabetic” to label a person, and they counter misconceptions such as that diabetes is contagious. There’s an Indian comic book version of this volume out now.
The main character of this volume experiences severe hypoglycemia and is saved by a glucagon injection from a colleague, one of several presented as superheroes who help in the fight to end diabetes stigma. They are referred to as Guardians of the Glucose, a take on yet another Marvel franchise, Guardians of the Galaxy.
At the end of this volume, we provide two pages of text about recognizing, managing, and preventing hypoglycemia. Again, we hope to educate as wide an audience as possible.
At the end of volume 3, you also briefly mention the COVID-19 pandemic. Will there be a fourth volume dealing with that, or other topics, such as diabetes technology?
We’ve left it open. We want to see how volume 3 lands. Depending on that, we might take it forward. There are certainly plenty of topics to tackle. We’ve also discussed moving into gaming or virtual reality. Overall, we hope to educate people by engaging them in different ways.
Dr. Kar has been a consultant diabetologist/endocrinologist within the NHS since 2008. He disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A young girl presents with ‘itchy, rashy’ hands
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Given the presence of erythema, lichenification, fissuring, and scale of the hands over the course of more than 3 months with the absence of nail findings is most consistent with a diagnosis of chronic hand eczema.
Chronic hand eczema (CHE) is an inflammatory dermatitis of the hands or wrists that persists for longer than 3 months or recurs twice or more in a 12-month timespan.1,2 Hand eczema can be a manifestation of atopic dermatitis, allergic contact dermatitis, or irritant contact dermatitis. Its multifactorial pathogenesis includes epidermal injury and disturbed epidermal barrier function from exogenous factors such as irritants or contact allergens, as well as endogenous factors including atopic dermatitis.3 In pediatrics, it often presents after an acute phase of hand dermatitis with chronic pruritus, erythema, and dry skin with scale.4 Examination findings vary widely with erythema, vesicles, scale, fissures, crusting, hyperkeratosis, and/or lichenification.3,5 Diagnosis is often achieved with careful history, asking about potential exposures that may induce lesions, and physical exam of the entire skin, including the feet. Based upon clinical history or persistent dermatitis, allergic contact dermatitis patch testing should be considered.2
What’s the treatment plan?
Given that CHE is an inflammatory disease process, the goal of treatment is to reduce inflammation and allow for skin barrier repair. Unfortunately, only one study has investigated therapeutics for pediatric CHE,6 with the remainder of the literature based on adult CHE. Current CHE guidelines recommend avoidance of allergens, irritants, or other triggers of the disease as well as liberal and regular use of emollients. Because of the relative thickness of hand skin, higher-potency topical corticosteroids are often used as first-line therapy, with lower-strength topical steroids, calcineurin inhibitors, or crisaborole used as maintenance therapy. Other treatment options include phototherapy, and rarely, systemic therapies are utilized for atopic dermatitis.
What’s the differential diagnosis?
The differential diagnosis of CHE includes other scaling or hyperkeratotic skin conditions including psoriasis and tinea manuum. Other skin conditions that localize to extremities including scabies and hand-foot-and-mouth disease are discussed below.
Psoriasis can present on the hands with erythematous, well-demarcated, silver scaling plaques. However, additional plaques may be found on the elbows, knees, scalp, umbilicus, and sacrum. Nails can demonstrate pitting, oil drops, splinter hemorrhages, or onycholysis. First-line treatment includes a combination of topical steroids, topical vitamin D analogues, and keratolytics.
Tinea mannum is a dermatophyte infection of the skin of the hands. Typically, only one hand is affected with concomitant bilateral tinea pedis. It results in a white scaly plaque with dorsal hand involvement demonstrating an annular appearance, elevated edge, and central clearing. KOH prep will demonstrate septate hyphae, and cultures will grow dermatophyte colonies. Treatment includes topical antifungals or systemic antifungals for recalcitrant disease.
Scabies presents with short linear hypopigmented lesions with a black dot on one end as well as erythematous pruritic papules. These appear on the interdigital web spaces, wrists, axilla, buttocks, and genital region. Skin scraping prep with mineral oil can show mites and eggs. All individuals in an affected household should be treated with either topical permethrin or oral ivermectin to avoid reinfection or parasitic spread. All contacted linens must be cleaned with hot water and dried on high heat.
Hand-foot-and-mouth disease, classically caused by coxsackievirus, is an acute viral illness that results in an eruption of erythematous macules, papules, and vesicles on the ventral hands, soles of the feet, and oral mucosa. Diagnosis is achieved clinically and treatment is symptomatic as the lesions are self-limited.
Our patient underwent patch testing but did not return positive to any allergens. She was started on potent topical corticosteroids, educated on trigger avoidance, and gradually achieved good disease control.
Neither Mr. Haft nor Dr. Eichenfield have any relevant financial disclosures.
Michael Haft is a pediatric dermatology research associate in the division of pediatric and adolescent dermatology at the University of California, San Diego, and Rady Children’s Hospital, San Diego. He is a 4th year medical student at the University of Rochester (N.Y.). Dr. Eichenfield is vice chair of the department of dermatology and professor of dermatology and pediatrics at the University of California, San Diego, and Rady Children’s Hospital.
References
1. Diepgen TL et al. Br J Dermatol. 2009;160(2):353-8.
2. Diepgen TL et al. J Dtsch Dermatol Ges. 2015;13(1):e1-22.
3. Agner T and Elsner P. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:4-12.
4. Mortz CG et al. Br J Dermatol. 2001;144(3):523-32.
5. Silvestre Salvador JF et al. Actas Dermosifiliogr. 2020;111(1):26-40.
6. Luchsinger I et al. J Eur Acad Dermatol Venereol. 2020;34(5):1037-42.
7. English J et al. Clin Exp Dermatol. 2009;34(7):761-9.
8. Elsner P and Agner T. J Eur Acad Dermatol Venereol. 2020;34 Suppl 1:13-21.
Examination findings of the bilateral hands and wrists demonstrate plaques of erythema, lichenification, and scale of the dorsal surfaces of the hands and digits. Closer inspection reveals fissuring and erythematous crust of the affected skin but normal nails. The rest of the skin exam is unremarkable.
Success in achondroplasia spurs testing vosoritide in more growth disorders
On the basis of the quality of sustained bone growth achieved with vosoritide in dwarfism, studies are underway or being considered for more diseases that impair bone growth, according to discussion that followed the presentation of a phase 3 trial extension study at the annual meeting of the Endocrine Society.
After 1 year on active therapy in the randomized trial and another year in the extension study, patients in the vosoritide group had sustained growth velocity while placebo group patients who crossed over to active therapy caught up, reported Ravi Savarirayan, MD, Murdoch Children’s Research Institute, University of Melbourne, Australia.
Moreover, the quality and type of bone growth, such as the improvement in body segment ratios over the second year of the study, support a durable benefit. Dr. Savarirayan said that improvements in activities of daily living are expected from this improvement in upper-to-lower body segment ratios, as well as the growth seen in the limbs.
Currently there is no approved pharmacologic therapy for achondroplasia in the United States. Growth hormone has been approved in Japan, but Dr. Savarirayan said its effects have been limited. Surgery such as limb lengthening is another option, but this approach is not uniformly effective and carries risks.
The 52-week results from the multinational phase 3 trial with vosoritide, which stimulates bone growth, were published last year in The Lancet. In that trial, 121 patients between the ages of 5 and 18 years with achondroplasia were randomized to vosoritide at a dose of 15 μg/kg once daily or placebo.
Relative to those in the placebo arm, which did not experience any change in growth, the median growth at the end of 52 weeks was 1.75 cm/year greater (6.71 vs. 3.99 cm).
After crossover, placebo patients catch up
In the extension study, the placebo patients were crossed over to the active therapy and both groups were followed for an additional 52 weeks. Over this period, velocity declined modestly in those in the group initially randomized to vosoritide but climbed steeply in the placebo group so that rates after 1 year were nearly identical (5.57 vs. 5.65 cm, respectively).
“The results suggest this medication may well have a durable effect,” said Dr. Savarirayan, who believes that the benefit is derived from stimulation of the growth plates. Based on the very similar efficacy observed in the placebo group once switched to active therapy, the response to vosoritide appears to be predictable.
Of the 60 patients initially randomized to vosoritide, 58 entered the extension. Of the patients who did not remain in the study, two left due to discomfort from injection-site reactions. All 61 patients initially assigned to placebo crossed over.
“We did not see any evidence of tachyphylaxis in the randomized study or in the extension,” Dr. Savarirayan said.
Although two more patients initiated on vosoritide discontinued treatment before the end of 2 years, there were no new adverse events observed. Rather, injection-site pain, which self-resolved in all patients, appears to be the most significant side effect.
“In children, the daily subcutaneous injections can be an issue,” Dr. Savarirayan acknowledged.
Injection site reactions most common adverse event
In a detailed evaluation of safety in a previously published dose-finding phase 2 study, injection-site reactions were also the most common of treatment-related adverse events, but there were no episodes of anaphylaxis or other grade 3 or higher hypersensitivity reactions (N Engl J Med. 2019 Jul 4;381:25-35).
Prior to clinical trials, continuous infusion of endogenous C-type natriuretic peptide demonstrated an ability to stimulate long-bone growth in experimental studies. Vosoritide, a recombinant analogue of C-type natriuretic peptide, appears to provide the same activity but offers a longer half-life.
Based on the benefits observed in achondroplasia, other applications are now being explored.
“When you evaluate the quality of the bone growth associated with vosoritide, it is normal,” said Melita Irving, MD, a consultant in clinical genetics at the Guy’s and St .Thomas’ NHS Trust, London. Dr. Irving has been involved in other research initiatives with this therapy and she cited a variety of evidence that has supported healthy bone development, including favorable changes in markers of bone growth such as type 10 collagen.
As a result, vosoritide, which is now under review by the U.S. Food and Drug Administration for treatment of dwarfism, is being pursued for several other diseases that result in abnormal bone growth, such as hypochondroplasia. Not least, clinical studies in idiopathic short stature have reached “early stages,” Dr. Irving said.
Dr. Savarirayan and Dr. Irving report no relevant conflicts of interest.
On the basis of the quality of sustained bone growth achieved with vosoritide in dwarfism, studies are underway or being considered for more diseases that impair bone growth, according to discussion that followed the presentation of a phase 3 trial extension study at the annual meeting of the Endocrine Society.
After 1 year on active therapy in the randomized trial and another year in the extension study, patients in the vosoritide group had sustained growth velocity while placebo group patients who crossed over to active therapy caught up, reported Ravi Savarirayan, MD, Murdoch Children’s Research Institute, University of Melbourne, Australia.
Moreover, the quality and type of bone growth, such as the improvement in body segment ratios over the second year of the study, support a durable benefit. Dr. Savarirayan said that improvements in activities of daily living are expected from this improvement in upper-to-lower body segment ratios, as well as the growth seen in the limbs.
Currently there is no approved pharmacologic therapy for achondroplasia in the United States. Growth hormone has been approved in Japan, but Dr. Savarirayan said its effects have been limited. Surgery such as limb lengthening is another option, but this approach is not uniformly effective and carries risks.
The 52-week results from the multinational phase 3 trial with vosoritide, which stimulates bone growth, were published last year in The Lancet. In that trial, 121 patients between the ages of 5 and 18 years with achondroplasia were randomized to vosoritide at a dose of 15 μg/kg once daily or placebo.
Relative to those in the placebo arm, which did not experience any change in growth, the median growth at the end of 52 weeks was 1.75 cm/year greater (6.71 vs. 3.99 cm).
After crossover, placebo patients catch up
In the extension study, the placebo patients were crossed over to the active therapy and both groups were followed for an additional 52 weeks. Over this period, velocity declined modestly in those in the group initially randomized to vosoritide but climbed steeply in the placebo group so that rates after 1 year were nearly identical (5.57 vs. 5.65 cm, respectively).
“The results suggest this medication may well have a durable effect,” said Dr. Savarirayan, who believes that the benefit is derived from stimulation of the growth plates. Based on the very similar efficacy observed in the placebo group once switched to active therapy, the response to vosoritide appears to be predictable.
Of the 60 patients initially randomized to vosoritide, 58 entered the extension. Of the patients who did not remain in the study, two left due to discomfort from injection-site reactions. All 61 patients initially assigned to placebo crossed over.
“We did not see any evidence of tachyphylaxis in the randomized study or in the extension,” Dr. Savarirayan said.
Although two more patients initiated on vosoritide discontinued treatment before the end of 2 years, there were no new adverse events observed. Rather, injection-site pain, which self-resolved in all patients, appears to be the most significant side effect.
“In children, the daily subcutaneous injections can be an issue,” Dr. Savarirayan acknowledged.
Injection site reactions most common adverse event
In a detailed evaluation of safety in a previously published dose-finding phase 2 study, injection-site reactions were also the most common of treatment-related adverse events, but there were no episodes of anaphylaxis or other grade 3 or higher hypersensitivity reactions (N Engl J Med. 2019 Jul 4;381:25-35).
Prior to clinical trials, continuous infusion of endogenous C-type natriuretic peptide demonstrated an ability to stimulate long-bone growth in experimental studies. Vosoritide, a recombinant analogue of C-type natriuretic peptide, appears to provide the same activity but offers a longer half-life.
Based on the benefits observed in achondroplasia, other applications are now being explored.
“When you evaluate the quality of the bone growth associated with vosoritide, it is normal,” said Melita Irving, MD, a consultant in clinical genetics at the Guy’s and St .Thomas’ NHS Trust, London. Dr. Irving has been involved in other research initiatives with this therapy and she cited a variety of evidence that has supported healthy bone development, including favorable changes in markers of bone growth such as type 10 collagen.
As a result, vosoritide, which is now under review by the U.S. Food and Drug Administration for treatment of dwarfism, is being pursued for several other diseases that result in abnormal bone growth, such as hypochondroplasia. Not least, clinical studies in idiopathic short stature have reached “early stages,” Dr. Irving said.
Dr. Savarirayan and Dr. Irving report no relevant conflicts of interest.
On the basis of the quality of sustained bone growth achieved with vosoritide in dwarfism, studies are underway or being considered for more diseases that impair bone growth, according to discussion that followed the presentation of a phase 3 trial extension study at the annual meeting of the Endocrine Society.
After 1 year on active therapy in the randomized trial and another year in the extension study, patients in the vosoritide group had sustained growth velocity while placebo group patients who crossed over to active therapy caught up, reported Ravi Savarirayan, MD, Murdoch Children’s Research Institute, University of Melbourne, Australia.
Moreover, the quality and type of bone growth, such as the improvement in body segment ratios over the second year of the study, support a durable benefit. Dr. Savarirayan said that improvements in activities of daily living are expected from this improvement in upper-to-lower body segment ratios, as well as the growth seen in the limbs.
Currently there is no approved pharmacologic therapy for achondroplasia in the United States. Growth hormone has been approved in Japan, but Dr. Savarirayan said its effects have been limited. Surgery such as limb lengthening is another option, but this approach is not uniformly effective and carries risks.
The 52-week results from the multinational phase 3 trial with vosoritide, which stimulates bone growth, were published last year in The Lancet. In that trial, 121 patients between the ages of 5 and 18 years with achondroplasia were randomized to vosoritide at a dose of 15 μg/kg once daily or placebo.
Relative to those in the placebo arm, which did not experience any change in growth, the median growth at the end of 52 weeks was 1.75 cm/year greater (6.71 vs. 3.99 cm).
After crossover, placebo patients catch up
In the extension study, the placebo patients were crossed over to the active therapy and both groups were followed for an additional 52 weeks. Over this period, velocity declined modestly in those in the group initially randomized to vosoritide but climbed steeply in the placebo group so that rates after 1 year were nearly identical (5.57 vs. 5.65 cm, respectively).
“The results suggest this medication may well have a durable effect,” said Dr. Savarirayan, who believes that the benefit is derived from stimulation of the growth plates. Based on the very similar efficacy observed in the placebo group once switched to active therapy, the response to vosoritide appears to be predictable.
Of the 60 patients initially randomized to vosoritide, 58 entered the extension. Of the patients who did not remain in the study, two left due to discomfort from injection-site reactions. All 61 patients initially assigned to placebo crossed over.
“We did not see any evidence of tachyphylaxis in the randomized study or in the extension,” Dr. Savarirayan said.
Although two more patients initiated on vosoritide discontinued treatment before the end of 2 years, there were no new adverse events observed. Rather, injection-site pain, which self-resolved in all patients, appears to be the most significant side effect.
“In children, the daily subcutaneous injections can be an issue,” Dr. Savarirayan acknowledged.
Injection site reactions most common adverse event
In a detailed evaluation of safety in a previously published dose-finding phase 2 study, injection-site reactions were also the most common of treatment-related adverse events, but there were no episodes of anaphylaxis or other grade 3 or higher hypersensitivity reactions (N Engl J Med. 2019 Jul 4;381:25-35).
Prior to clinical trials, continuous infusion of endogenous C-type natriuretic peptide demonstrated an ability to stimulate long-bone growth in experimental studies. Vosoritide, a recombinant analogue of C-type natriuretic peptide, appears to provide the same activity but offers a longer half-life.
Based on the benefits observed in achondroplasia, other applications are now being explored.
“When you evaluate the quality of the bone growth associated with vosoritide, it is normal,” said Melita Irving, MD, a consultant in clinical genetics at the Guy’s and St .Thomas’ NHS Trust, London. Dr. Irving has been involved in other research initiatives with this therapy and she cited a variety of evidence that has supported healthy bone development, including favorable changes in markers of bone growth such as type 10 collagen.
As a result, vosoritide, which is now under review by the U.S. Food and Drug Administration for treatment of dwarfism, is being pursued for several other diseases that result in abnormal bone growth, such as hypochondroplasia. Not least, clinical studies in idiopathic short stature have reached “early stages,” Dr. Irving said.
Dr. Savarirayan and Dr. Irving report no relevant conflicts of interest.
FROM ENDO 2021
Children with increased suicide risk are falling through the cracks
Children in the welfare system who died by suicide were twice as likely to receive mental health services within the 6 months before their death, according to a recent study published in Pediatrics.
“Health care settings that provide more robust mental health screening and suicide risk assessment are needed for youth with child welfare system involvement,” study author Donna Ruch, PhD, a research scientist at the Nationwide Children’s Hospital, said in an interview.
Researchers noted that integrating suicide prevention strategies in primary care and providing access to effective health services for this vulnerable group could be beneficial.
At-risk kids are falling through the cracks
Suicide is the second leading cause of death in children, adolescents, and young adults between the ages of 15 and 24 years old. Children in the welfare system are four times more likely to have attempted suicide; however, research on suicide rates in this population is minimal.
“Kids in the child welfare system are so understudied and yet at such a high risk for suicide,” said Lisa Horowitz, PhD, clinical psychologist at the National Institutes of Health, who was not involved in the study. “A lot of kids pass through the health care system undetected.”
In an attempt to understand and prevent suicide in this group, Dr. Ruch and her team examined health service utilization patterns of children in the welfare system who committed suicide, compared with those in the system who did not die by suicide.
Researchers collected data on 120 deceased youth between the ages of 5 and 21 years old who had an open case in Ohio’s Statewide Automated Child Welfare Information System between 2010 and 2017. For the purpose of the study, open cases were defined as investigated child maltreatment where the family received services or the child was removed from the home.
Researchers matched each child who died by suicide with 10 controls – children in the welfare system who did not commit suicide – based on sex, race, and ethnicity.
The findings revealed that 59.2% of suicide decedents had a diagnosed mental health condition, compared with 31.3% of the control group. Researchers also found that the suicide decedent group was more likely to have multiple mental health diagnoses, with a quarter of them having at least three diagnosed conditions.
Children who died by suicide were also more likely to have a history of self-harm and to have been placed in foster or kinship care.
“Existing research also suggests that known risk factors for youth suicide are more common in youth involved with the child welfare system. This includes mental health conditions, developmental delays, problematic family-related issues, and trauma,” said Dr. Ruch. “All of these factors may be compounded for youth who are removed from their homes.”
Dr. Ruch said it is likely that children who are removed from their homes and placed in foster care may not have consistent access to necessary health services, such as therapy, which may place them at an increased risk for suicidal behavior.
Robust prevention strategies needed
Researchers also found that 90% of children who died by suicide had a health care visit within 6 months of their deaths, compared with 69.4% of controls; 48% of those visits occurred 1 month before they died.
The frequency of health care services used by suicide decedents suggests that prevention strategies for children in the welfare system should be embedded in routine medical and mental health care.
“If we as mental health counselors allow these kids to pass through the health care system, it’s really further neglect,” said Dr. Horowitz, who wrote an accompanying commentary. “And these children already deal with abuse and neglect – we don’t need to further neglect them.”
Dr. Horowitz said health care providers could go over coping strategies and discuss how children deal with hard times and make sure they have access to suicide prevention resources, such as the suicide hotline.
Additionally, better coordination with health care systems and the child welfare system is necessary to make sure there are follow-ups and screenings for suicide and other mental health conditions.
It’s not one size fits all: There may be tailored suicide prevention strategies that work better,” Dr. Horowitz explained.
Dr. Ruch and her team also believe suicide prevention strategies such as the Zero Suicide approach – an initiative that aims to embed suicide prevention health and behavioral health care systems – as well as interventions focused on family preservation to reduce the chance of a child being removed from their home could also benefit children in the welfare system.
Dr. Ruch, the other authors of the study, and Dr. Horowitz disclosed no relevant financial conflicts,
Children in the welfare system who died by suicide were twice as likely to receive mental health services within the 6 months before their death, according to a recent study published in Pediatrics.
“Health care settings that provide more robust mental health screening and suicide risk assessment are needed for youth with child welfare system involvement,” study author Donna Ruch, PhD, a research scientist at the Nationwide Children’s Hospital, said in an interview.
Researchers noted that integrating suicide prevention strategies in primary care and providing access to effective health services for this vulnerable group could be beneficial.
At-risk kids are falling through the cracks
Suicide is the second leading cause of death in children, adolescents, and young adults between the ages of 15 and 24 years old. Children in the welfare system are four times more likely to have attempted suicide; however, research on suicide rates in this population is minimal.
“Kids in the child welfare system are so understudied and yet at such a high risk for suicide,” said Lisa Horowitz, PhD, clinical psychologist at the National Institutes of Health, who was not involved in the study. “A lot of kids pass through the health care system undetected.”
In an attempt to understand and prevent suicide in this group, Dr. Ruch and her team examined health service utilization patterns of children in the welfare system who committed suicide, compared with those in the system who did not die by suicide.
Researchers collected data on 120 deceased youth between the ages of 5 and 21 years old who had an open case in Ohio’s Statewide Automated Child Welfare Information System between 2010 and 2017. For the purpose of the study, open cases were defined as investigated child maltreatment where the family received services or the child was removed from the home.
Researchers matched each child who died by suicide with 10 controls – children in the welfare system who did not commit suicide – based on sex, race, and ethnicity.
The findings revealed that 59.2% of suicide decedents had a diagnosed mental health condition, compared with 31.3% of the control group. Researchers also found that the suicide decedent group was more likely to have multiple mental health diagnoses, with a quarter of them having at least three diagnosed conditions.
Children who died by suicide were also more likely to have a history of self-harm and to have been placed in foster or kinship care.
“Existing research also suggests that known risk factors for youth suicide are more common in youth involved with the child welfare system. This includes mental health conditions, developmental delays, problematic family-related issues, and trauma,” said Dr. Ruch. “All of these factors may be compounded for youth who are removed from their homes.”
Dr. Ruch said it is likely that children who are removed from their homes and placed in foster care may not have consistent access to necessary health services, such as therapy, which may place them at an increased risk for suicidal behavior.
Robust prevention strategies needed
Researchers also found that 90% of children who died by suicide had a health care visit within 6 months of their deaths, compared with 69.4% of controls; 48% of those visits occurred 1 month before they died.
The frequency of health care services used by suicide decedents suggests that prevention strategies for children in the welfare system should be embedded in routine medical and mental health care.
“If we as mental health counselors allow these kids to pass through the health care system, it’s really further neglect,” said Dr. Horowitz, who wrote an accompanying commentary. “And these children already deal with abuse and neglect – we don’t need to further neglect them.”
Dr. Horowitz said health care providers could go over coping strategies and discuss how children deal with hard times and make sure they have access to suicide prevention resources, such as the suicide hotline.
Additionally, better coordination with health care systems and the child welfare system is necessary to make sure there are follow-ups and screenings for suicide and other mental health conditions.
It’s not one size fits all: There may be tailored suicide prevention strategies that work better,” Dr. Horowitz explained.
Dr. Ruch and her team also believe suicide prevention strategies such as the Zero Suicide approach – an initiative that aims to embed suicide prevention health and behavioral health care systems – as well as interventions focused on family preservation to reduce the chance of a child being removed from their home could also benefit children in the welfare system.
Dr. Ruch, the other authors of the study, and Dr. Horowitz disclosed no relevant financial conflicts,
Children in the welfare system who died by suicide were twice as likely to receive mental health services within the 6 months before their death, according to a recent study published in Pediatrics.
“Health care settings that provide more robust mental health screening and suicide risk assessment are needed for youth with child welfare system involvement,” study author Donna Ruch, PhD, a research scientist at the Nationwide Children’s Hospital, said in an interview.
Researchers noted that integrating suicide prevention strategies in primary care and providing access to effective health services for this vulnerable group could be beneficial.
At-risk kids are falling through the cracks
Suicide is the second leading cause of death in children, adolescents, and young adults between the ages of 15 and 24 years old. Children in the welfare system are four times more likely to have attempted suicide; however, research on suicide rates in this population is minimal.
“Kids in the child welfare system are so understudied and yet at such a high risk for suicide,” said Lisa Horowitz, PhD, clinical psychologist at the National Institutes of Health, who was not involved in the study. “A lot of kids pass through the health care system undetected.”
In an attempt to understand and prevent suicide in this group, Dr. Ruch and her team examined health service utilization patterns of children in the welfare system who committed suicide, compared with those in the system who did not die by suicide.
Researchers collected data on 120 deceased youth between the ages of 5 and 21 years old who had an open case in Ohio’s Statewide Automated Child Welfare Information System between 2010 and 2017. For the purpose of the study, open cases were defined as investigated child maltreatment where the family received services or the child was removed from the home.
Researchers matched each child who died by suicide with 10 controls – children in the welfare system who did not commit suicide – based on sex, race, and ethnicity.
The findings revealed that 59.2% of suicide decedents had a diagnosed mental health condition, compared with 31.3% of the control group. Researchers also found that the suicide decedent group was more likely to have multiple mental health diagnoses, with a quarter of them having at least three diagnosed conditions.
Children who died by suicide were also more likely to have a history of self-harm and to have been placed in foster or kinship care.
“Existing research also suggests that known risk factors for youth suicide are more common in youth involved with the child welfare system. This includes mental health conditions, developmental delays, problematic family-related issues, and trauma,” said Dr. Ruch. “All of these factors may be compounded for youth who are removed from their homes.”
Dr. Ruch said it is likely that children who are removed from their homes and placed in foster care may not have consistent access to necessary health services, such as therapy, which may place them at an increased risk for suicidal behavior.
Robust prevention strategies needed
Researchers also found that 90% of children who died by suicide had a health care visit within 6 months of their deaths, compared with 69.4% of controls; 48% of those visits occurred 1 month before they died.
The frequency of health care services used by suicide decedents suggests that prevention strategies for children in the welfare system should be embedded in routine medical and mental health care.
“If we as mental health counselors allow these kids to pass through the health care system, it’s really further neglect,” said Dr. Horowitz, who wrote an accompanying commentary. “And these children already deal with abuse and neglect – we don’t need to further neglect them.”
Dr. Horowitz said health care providers could go over coping strategies and discuss how children deal with hard times and make sure they have access to suicide prevention resources, such as the suicide hotline.
Additionally, better coordination with health care systems and the child welfare system is necessary to make sure there are follow-ups and screenings for suicide and other mental health conditions.
It’s not one size fits all: There may be tailored suicide prevention strategies that work better,” Dr. Horowitz explained.
Dr. Ruch and her team also believe suicide prevention strategies such as the Zero Suicide approach – an initiative that aims to embed suicide prevention health and behavioral health care systems – as well as interventions focused on family preservation to reduce the chance of a child being removed from their home could also benefit children in the welfare system.
Dr. Ruch, the other authors of the study, and Dr. Horowitz disclosed no relevant financial conflicts,
Baby born to partially vaccinated mom has COVID-19 antibodies
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.
A baby girl who was born 3 weeks after her mom got the first dose of the Moderna COVID-19 vaccine has antibodies against the coronavirus, according to a preprint paper published on the medRxiv server Feb. 5. The paper hasn’t yet been peer reviewed.
The mom, a health care worker in Florida, developed COVID-19 antibodies after she received the shot. Testing showed that the antibodies passed through the placenta to the baby.
“Maternal vaccination for influenza and TDaP have been well studied in terms of safety and efficacy for protection of the newborn by placental passage of antibodies,” Paul Gilbert, MD, and Chad Rudnick, MD, pediatricians and researchers at Florida Atlantic University, wrote in the paper.
Previous research has indicated that moms who have recovered from COVID-19 can deliver babies with antibodies, according to Insider, but this may be the first report that shows how vaccination during pregnancy can provide antibodies as well.
Dr. Gilbert and Dr. Rudnick said they were fortunate to connect with the mom in Boca Raton. She hadn’t contracted COVID-19 and was able to get the vaccine at the end of her pregnancy in January. When the baby was born, they were able to test the cord blood to look for antibodies specifically from the vaccine.
“We were very excited to see, once the test result came back, that the antibodies from the mom’s vaccine did in fact pass through the placenta to the newborn,” Dr. Rudnick told WPTV, an NBC affiliate in West Palm Beach.
“We knew that we were going to be potentially one of the first in the world to report it, and that opportunity probably only comes once in a career,” Dr. Gilbert told WPTV.
In the preprint, Dr. Gilbert and Dr. Rudnick said a “vigorous, healthy, full-term” baby was born, and the mom received the second dose of the Moderna vaccine during the postpartum period. The newborn received a normal “well-infant” evaluation and was breastfeeding.
The two doctors called for a “significant and urgent need” to research the safety and efficacy of COVID-19 vaccines during pregnancy. They also encouraged other researchers to create pregnancy and breastfeeding registries to study COVID-19 vaccines in pregnant and breastfeeding moms and newborns.
Dr. Gilbert and Dr. Rudnick are now preparing their research for publication and hope future studies will investigate the amount and length of antibody response in newborns.
“Total antibody measurements may be used to determine how long protection is expected, which may help to determine when the best time would be to begin vaccination,” they wrote.
A version of this article first appeared on Medscape.com.