User login
Bathing now more widely accepted as an eczema treatment strategy
According to Noreen Heer Nicol, PhD, RN, FNP, frustration still exists for patients, families, and health care providers regarding the lack of consensus that routine bathing is good for patients with atopic dermatitis.
During the Revolutionizing Atopic Dermatitis symposium, she said that conflicting and vague guidelines currently exist on the topic.
“This stems from the fact that we just don’t have good studies,” said Dr. Nicol, associate dean and associate professor of nursing at the University of Colorado at Denver, Aurora. “Particularly, we don’t have randomized, controlled trials on the effects of water and bathing. It’s not just parents that are frustrated, but health care providers are as well.”
In an observational analysis, researchers evaluated results from three online surveys of dermatologists, allergists, and immunologists, and primary care physicians regarding routine bathing frequency recommendations for children with AD. It found that PCPs recommended daily bathing less than 50% of the time, while specialists recommended daily bathing more than 50% of the time.
“It seems like the PCPs have embraced that old dermatology notion when bathing was avoided in patients with AD,” Dr. Nicol said. “This lack of consensus on the basic daily care steps in AD management causes a great deal of confusion amongst patients, families, and young health care providers, in particular,” she added.
She believes that this goes back to a century-long debate about the pros and cons of bathing in AD. “We used to say that bathing will dry the skin out if you take a bath or a shower without immediately applying something like a good moisturizer. That’s where the 3-minute rule came along from the National Eczema Association, meaning that bathing hydrates the stratum corneum if you take a bath or a shower and you immediately apply that good moisturizer within 3 minutes to retain that hydration and keep the barrier intact and flexible.”
Dr. Nicol presented a stepwise management model that she has published many times over the years (see Pediatr Nursing 2020;46[2]:92-8 and J Allergy Clin Immunol Pract 2019;7[1]:1-16).
Step 1 consists of basic care, including skin hydration/bathing, application of a daily moisturizer, avoiding irritants, and identifying and addressing specific triggers. “This is the foundation for every step as you go forward,” she explained. Soak and seal has been a mainstay of treatment at National Jewish Health, she noted. “By that, I mean taking a soaking 10-15 minute bath in warm water daily. Gently pat away excess water. Immediately apply skin medications or moisturizer within 3 minutes. Using a gentle fragrance-free, dye-free cleanser to clean skin is also important. Avoid scrubbing.”
A review article on bathing and associated treatments in AD was published in 2017 and includes 144 references to bathing studies. A separate recommendation known as the “AD Yardstick” published by Dr. Nicol’s colleague at National Jewish Health, Mark Boguniewicz, MD, and coauthors, elaborated on the definition of basic skin care for nonlesional AD. Besides recommending the liberal and frequent application of moisturizers, it suggests management with warm baths or showers using nonsoap cleansers, usually once per day, followed by application of a moisturizer, even on clear areas.
“This is now what people are thinking as the basis of skin care in patients with AD,” Dr. Nicol said. “Warm baths and showers don’t look so controversial anymore. This model nicely lays out what we want people to remember. In the past, many times we just skipped that important step of telling people about bathing.”
In a small 2009 study, researchers conducted a quantitative assessment of combination bathing and moisturizing regimens on skin hydration in AD. They found that bathing followed by application of a moisturizer provides modest hydration benefits, though less than that of simply applying moisturizer alone. “That has not been the case for most of us who are bathing advocates,” Dr. Nicol said. “We believe that there is an additional hydration that’s gained from bathing and moisturizers done properly.”
In an earlier retrospective study of 28 patients referred to a tertiary care center for refractory chronic pruritic eruptions, researchers found that a plain-water 20-minute soak followed by smearing of midstrength corticosteroid ointment led to clearing or dramatic improvement of the lesions (Arch Dermatol 2005;14:1556-9). The authors recommended prospective studies to confirm the findings.
In a separate review of medical literature, researchers explored the role of frequent bathing in the treatment of pediatric AD (Ann Allergy Asthma Immunol 2016;117[1]:9-13). They found that the weight of evidence suggests that the frequent soak and smear bathing is preferred to infrequent bathing in the management of AD. Frequent bathing was defined as bathing at least once a day, while infrequent bathing was defined as bathing less than once a day.
“Bleach baths have received much attention in recent years, and have been endorsed by multiple AD guidelines, though not to the same degree as regular bathing,” Dr. Nicol said. “Right now, you can find almost as much literature for this practice as against it. The populations that seem to value from beach baths the most, however, are those with frequent infections, particularly those who are methicillin resistant. Most people recommend a maximum of two to three times per week but only with an active infection. Care must be taken to avoid additional drying or irritation of the skin from bleach.”
Many bleach bath recipes call for adding one-eighth to one-half of a cup of bleach to a tub full or water.
Dr. Nicol disclosed that she has served as an advisory board member for Eli Lilly.
According to Noreen Heer Nicol, PhD, RN, FNP, frustration still exists for patients, families, and health care providers regarding the lack of consensus that routine bathing is good for patients with atopic dermatitis.
During the Revolutionizing Atopic Dermatitis symposium, she said that conflicting and vague guidelines currently exist on the topic.
“This stems from the fact that we just don’t have good studies,” said Dr. Nicol, associate dean and associate professor of nursing at the University of Colorado at Denver, Aurora. “Particularly, we don’t have randomized, controlled trials on the effects of water and bathing. It’s not just parents that are frustrated, but health care providers are as well.”
In an observational analysis, researchers evaluated results from three online surveys of dermatologists, allergists, and immunologists, and primary care physicians regarding routine bathing frequency recommendations for children with AD. It found that PCPs recommended daily bathing less than 50% of the time, while specialists recommended daily bathing more than 50% of the time.
“It seems like the PCPs have embraced that old dermatology notion when bathing was avoided in patients with AD,” Dr. Nicol said. “This lack of consensus on the basic daily care steps in AD management causes a great deal of confusion amongst patients, families, and young health care providers, in particular,” she added.
She believes that this goes back to a century-long debate about the pros and cons of bathing in AD. “We used to say that bathing will dry the skin out if you take a bath or a shower without immediately applying something like a good moisturizer. That’s where the 3-minute rule came along from the National Eczema Association, meaning that bathing hydrates the stratum corneum if you take a bath or a shower and you immediately apply that good moisturizer within 3 minutes to retain that hydration and keep the barrier intact and flexible.”
Dr. Nicol presented a stepwise management model that she has published many times over the years (see Pediatr Nursing 2020;46[2]:92-8 and J Allergy Clin Immunol Pract 2019;7[1]:1-16).
Step 1 consists of basic care, including skin hydration/bathing, application of a daily moisturizer, avoiding irritants, and identifying and addressing specific triggers. “This is the foundation for every step as you go forward,” she explained. Soak and seal has been a mainstay of treatment at National Jewish Health, she noted. “By that, I mean taking a soaking 10-15 minute bath in warm water daily. Gently pat away excess water. Immediately apply skin medications or moisturizer within 3 minutes. Using a gentle fragrance-free, dye-free cleanser to clean skin is also important. Avoid scrubbing.”
A review article on bathing and associated treatments in AD was published in 2017 and includes 144 references to bathing studies. A separate recommendation known as the “AD Yardstick” published by Dr. Nicol’s colleague at National Jewish Health, Mark Boguniewicz, MD, and coauthors, elaborated on the definition of basic skin care for nonlesional AD. Besides recommending the liberal and frequent application of moisturizers, it suggests management with warm baths or showers using nonsoap cleansers, usually once per day, followed by application of a moisturizer, even on clear areas.
“This is now what people are thinking as the basis of skin care in patients with AD,” Dr. Nicol said. “Warm baths and showers don’t look so controversial anymore. This model nicely lays out what we want people to remember. In the past, many times we just skipped that important step of telling people about bathing.”
In a small 2009 study, researchers conducted a quantitative assessment of combination bathing and moisturizing regimens on skin hydration in AD. They found that bathing followed by application of a moisturizer provides modest hydration benefits, though less than that of simply applying moisturizer alone. “That has not been the case for most of us who are bathing advocates,” Dr. Nicol said. “We believe that there is an additional hydration that’s gained from bathing and moisturizers done properly.”
In an earlier retrospective study of 28 patients referred to a tertiary care center for refractory chronic pruritic eruptions, researchers found that a plain-water 20-minute soak followed by smearing of midstrength corticosteroid ointment led to clearing or dramatic improvement of the lesions (Arch Dermatol 2005;14:1556-9). The authors recommended prospective studies to confirm the findings.
In a separate review of medical literature, researchers explored the role of frequent bathing in the treatment of pediatric AD (Ann Allergy Asthma Immunol 2016;117[1]:9-13). They found that the weight of evidence suggests that the frequent soak and smear bathing is preferred to infrequent bathing in the management of AD. Frequent bathing was defined as bathing at least once a day, while infrequent bathing was defined as bathing less than once a day.
“Bleach baths have received much attention in recent years, and have been endorsed by multiple AD guidelines, though not to the same degree as regular bathing,” Dr. Nicol said. “Right now, you can find almost as much literature for this practice as against it. The populations that seem to value from beach baths the most, however, are those with frequent infections, particularly those who are methicillin resistant. Most people recommend a maximum of two to three times per week but only with an active infection. Care must be taken to avoid additional drying or irritation of the skin from bleach.”
Many bleach bath recipes call for adding one-eighth to one-half of a cup of bleach to a tub full or water.
Dr. Nicol disclosed that she has served as an advisory board member for Eli Lilly.
According to Noreen Heer Nicol, PhD, RN, FNP, frustration still exists for patients, families, and health care providers regarding the lack of consensus that routine bathing is good for patients with atopic dermatitis.
During the Revolutionizing Atopic Dermatitis symposium, she said that conflicting and vague guidelines currently exist on the topic.
“This stems from the fact that we just don’t have good studies,” said Dr. Nicol, associate dean and associate professor of nursing at the University of Colorado at Denver, Aurora. “Particularly, we don’t have randomized, controlled trials on the effects of water and bathing. It’s not just parents that are frustrated, but health care providers are as well.”
In an observational analysis, researchers evaluated results from three online surveys of dermatologists, allergists, and immunologists, and primary care physicians regarding routine bathing frequency recommendations for children with AD. It found that PCPs recommended daily bathing less than 50% of the time, while specialists recommended daily bathing more than 50% of the time.
“It seems like the PCPs have embraced that old dermatology notion when bathing was avoided in patients with AD,” Dr. Nicol said. “This lack of consensus on the basic daily care steps in AD management causes a great deal of confusion amongst patients, families, and young health care providers, in particular,” she added.
She believes that this goes back to a century-long debate about the pros and cons of bathing in AD. “We used to say that bathing will dry the skin out if you take a bath or a shower without immediately applying something like a good moisturizer. That’s where the 3-minute rule came along from the National Eczema Association, meaning that bathing hydrates the stratum corneum if you take a bath or a shower and you immediately apply that good moisturizer within 3 minutes to retain that hydration and keep the barrier intact and flexible.”
Dr. Nicol presented a stepwise management model that she has published many times over the years (see Pediatr Nursing 2020;46[2]:92-8 and J Allergy Clin Immunol Pract 2019;7[1]:1-16).
Step 1 consists of basic care, including skin hydration/bathing, application of a daily moisturizer, avoiding irritants, and identifying and addressing specific triggers. “This is the foundation for every step as you go forward,” she explained. Soak and seal has been a mainstay of treatment at National Jewish Health, she noted. “By that, I mean taking a soaking 10-15 minute bath in warm water daily. Gently pat away excess water. Immediately apply skin medications or moisturizer within 3 minutes. Using a gentle fragrance-free, dye-free cleanser to clean skin is also important. Avoid scrubbing.”
A review article on bathing and associated treatments in AD was published in 2017 and includes 144 references to bathing studies. A separate recommendation known as the “AD Yardstick” published by Dr. Nicol’s colleague at National Jewish Health, Mark Boguniewicz, MD, and coauthors, elaborated on the definition of basic skin care for nonlesional AD. Besides recommending the liberal and frequent application of moisturizers, it suggests management with warm baths or showers using nonsoap cleansers, usually once per day, followed by application of a moisturizer, even on clear areas.
“This is now what people are thinking as the basis of skin care in patients with AD,” Dr. Nicol said. “Warm baths and showers don’t look so controversial anymore. This model nicely lays out what we want people to remember. In the past, many times we just skipped that important step of telling people about bathing.”
In a small 2009 study, researchers conducted a quantitative assessment of combination bathing and moisturizing regimens on skin hydration in AD. They found that bathing followed by application of a moisturizer provides modest hydration benefits, though less than that of simply applying moisturizer alone. “That has not been the case for most of us who are bathing advocates,” Dr. Nicol said. “We believe that there is an additional hydration that’s gained from bathing and moisturizers done properly.”
In an earlier retrospective study of 28 patients referred to a tertiary care center for refractory chronic pruritic eruptions, researchers found that a plain-water 20-minute soak followed by smearing of midstrength corticosteroid ointment led to clearing or dramatic improvement of the lesions (Arch Dermatol 2005;14:1556-9). The authors recommended prospective studies to confirm the findings.
In a separate review of medical literature, researchers explored the role of frequent bathing in the treatment of pediatric AD (Ann Allergy Asthma Immunol 2016;117[1]:9-13). They found that the weight of evidence suggests that the frequent soak and smear bathing is preferred to infrequent bathing in the management of AD. Frequent bathing was defined as bathing at least once a day, while infrequent bathing was defined as bathing less than once a day.
“Bleach baths have received much attention in recent years, and have been endorsed by multiple AD guidelines, though not to the same degree as regular bathing,” Dr. Nicol said. “Right now, you can find almost as much literature for this practice as against it. The populations that seem to value from beach baths the most, however, are those with frequent infections, particularly those who are methicillin resistant. Most people recommend a maximum of two to three times per week but only with an active infection. Care must be taken to avoid additional drying or irritation of the skin from bleach.”
Many bleach bath recipes call for adding one-eighth to one-half of a cup of bleach to a tub full or water.
Dr. Nicol disclosed that she has served as an advisory board member for Eli Lilly.
FROM REVOLUTIONIZING AD 2020
Which providers miss metabolic monitoring of children taking antipsychotics?
The number and types of providers involved in a child’s care are associated with the likelihood that the child will receive metabolic monitoring, according to the study, which was published in Pediatrics.
The results suggest that primary care providers and mental health providers should collaborate to monitor children taking antipsychotics, the researchers said.
“Shared care arrangements between primary care physicians and mental health specialists significantly increased the chances that metabolic monitoring would be done, compared with care delivered by one provider,” reported Elizabeth A. Shenkman, PhD, chair of the department of health outcomes and biomedical informatics at the University of Florida, Gainesville, and colleagues. “The results of our study point to the importance of state Medicaid agencies and Medicaid managed care plans in identifying all providers caring for the children taking antipsychotic medication and using this information to engage the providers in quality improvement efforts to improve metabolic monitoring rates.”
Comparing specialties
Children who take antipsychotic medication are at risk for obesity, impaired glucose metabolism, and hyperlipidemia, but less than 40% receive recommended metabolic monitoring with glucose and cholesterol tests.
To examine how health care provider specialty influences the receipt of metabolic monitoring, Dr. Shenkman and colleagues analyzed Medicaid enrollment and health care and pharmacy claims data from Florida and Texas.
They focused on 41,078 children who had an antipsychotic medication dispensed at least twice in 2017 and were eligible for inclusion in the Centers for Medicare & Medicaid Services metabolic monitoring measure. The Metabolic Monitoring for Children and Adolescents on Antipsychotics measure is a “priority nationally and is currently on the CMS Child Core Set, which is used to annually assess state-specific performance on pediatric quality measures,” the authors wrote.
About 65% were boys, and the children had an average age of 12 years. The researchers compared metabolic monitoring rates when children received outpatient care from a primary care provider, a mental health provider with prescribing privileges, or both.
Less than 40% of the children received metabolic monitoring, that is, at least one diabetes test and at least one cholesterol test, during the year.
Most of the children (61%) saw both primary care providers and mental health providers. Approximately one-third had a primary care provider prescribe antipsychotic medication the majority of the time, and 60% had a mental health provider prescribe antipsychotic medication the majority of the time.
Patients who saw both types of providers were significantly more likely to receive metabolic monitoring, relative to those who saw primary care providers only (adjusted odds ratio, 1.42). Those seeing a mental health provider alone had adjusted odds of metabolic monitoring that were 23% lower than those seeing a primary care provider alone.
Children who had a mental health provider prescribe the medication the majority of the time were 25% more likely to receive metabolic monitoring, compared with those who had a primary care provider prescribing the medication the majority of the time.
Slipping through the cracks
Child psychiatrist Fred Volkmar, MD, commented that the results are “sadly” unsurprising and reflect issues that pertain to other psychotropic drugs as well as antipsychotics and to adults as well as children.
The researchers “are quite right to point to it,” and “we really do need to develop better plans for improving” monitoring, said Dr. Volkmar, the Irving B. Harris Professor in the Child Study Center and professor of psychology at Yale University, New Haven, Conn.
“Increasingly we are asking primary care providers ... to take care of folks who have important either developmental or mental health problems,” Dr. Volkmar said. While they can do a good job, they increasingly are underpaid. Monitoring patients takes work, and they may be less familiar with the medications. “Either they prescribe these medications or they are asked to monitor them in place of the specialist provider who may have started them or suggested them,” he said. Metabolic monitoring may not be prioritized and can easily “slip through the cracks.” At the same time, doctors need to be aware of the risk of serious side effects of antipsychotic medications, such as malignant hyperthermia.
These medications can be overused and inappropriately used, which is a further complication. And when patients are taking multiple medications, there may be a need for additional monitoring and awareness of drug interactions.
“These medications are very complicated to use,” and there needs to be a way to connect primary care providers with child psychiatrists who are best trained in their use, said Dr. Volkmar.
A system with reminders can facilitate effective metabolic monitoring, he suggested. Dr. Volkmar has established a routine while providing care for a group home of adults with autism. Every 3 months, he reviews lab results. “You just have to force yourself to do it.”
Shared care arrangements may be another way to promote metabolic monitoring, Dr. Shenkman and colleagues said.
“Attributing care back to the multiple providers is important for care coordination and development of strategies to ensure that the evidence-based care is delivered and there is appropriate follow-up with the family and child to be sure care is received,” the study authors wrote. “Formalized shared care arrangements and adaptation of existing care delivery models to support integrated care, which can vary in degree from external coordination to on-site intervention and collaboration, are effective methods to promote partnership between primary and mental health providers.”
It is possible that clinicians in the study ordered metabolic monitoring but families did not take the children for testing, the investigators noted. In addition, it is not clear how much information providers have about other providers their patients are seeing.
The study authors and Dr. Volkmar had no disclosures.
The number and types of providers involved in a child’s care are associated with the likelihood that the child will receive metabolic monitoring, according to the study, which was published in Pediatrics.
The results suggest that primary care providers and mental health providers should collaborate to monitor children taking antipsychotics, the researchers said.
“Shared care arrangements between primary care physicians and mental health specialists significantly increased the chances that metabolic monitoring would be done, compared with care delivered by one provider,” reported Elizabeth A. Shenkman, PhD, chair of the department of health outcomes and biomedical informatics at the University of Florida, Gainesville, and colleagues. “The results of our study point to the importance of state Medicaid agencies and Medicaid managed care plans in identifying all providers caring for the children taking antipsychotic medication and using this information to engage the providers in quality improvement efforts to improve metabolic monitoring rates.”
Comparing specialties
Children who take antipsychotic medication are at risk for obesity, impaired glucose metabolism, and hyperlipidemia, but less than 40% receive recommended metabolic monitoring with glucose and cholesterol tests.
To examine how health care provider specialty influences the receipt of metabolic monitoring, Dr. Shenkman and colleagues analyzed Medicaid enrollment and health care and pharmacy claims data from Florida and Texas.
They focused on 41,078 children who had an antipsychotic medication dispensed at least twice in 2017 and were eligible for inclusion in the Centers for Medicare & Medicaid Services metabolic monitoring measure. The Metabolic Monitoring for Children and Adolescents on Antipsychotics measure is a “priority nationally and is currently on the CMS Child Core Set, which is used to annually assess state-specific performance on pediatric quality measures,” the authors wrote.
About 65% were boys, and the children had an average age of 12 years. The researchers compared metabolic monitoring rates when children received outpatient care from a primary care provider, a mental health provider with prescribing privileges, or both.
Less than 40% of the children received metabolic monitoring, that is, at least one diabetes test and at least one cholesterol test, during the year.
Most of the children (61%) saw both primary care providers and mental health providers. Approximately one-third had a primary care provider prescribe antipsychotic medication the majority of the time, and 60% had a mental health provider prescribe antipsychotic medication the majority of the time.
Patients who saw both types of providers were significantly more likely to receive metabolic monitoring, relative to those who saw primary care providers only (adjusted odds ratio, 1.42). Those seeing a mental health provider alone had adjusted odds of metabolic monitoring that were 23% lower than those seeing a primary care provider alone.
Children who had a mental health provider prescribe the medication the majority of the time were 25% more likely to receive metabolic monitoring, compared with those who had a primary care provider prescribing the medication the majority of the time.
Slipping through the cracks
Child psychiatrist Fred Volkmar, MD, commented that the results are “sadly” unsurprising and reflect issues that pertain to other psychotropic drugs as well as antipsychotics and to adults as well as children.
The researchers “are quite right to point to it,” and “we really do need to develop better plans for improving” monitoring, said Dr. Volkmar, the Irving B. Harris Professor in the Child Study Center and professor of psychology at Yale University, New Haven, Conn.
“Increasingly we are asking primary care providers ... to take care of folks who have important either developmental or mental health problems,” Dr. Volkmar said. While they can do a good job, they increasingly are underpaid. Monitoring patients takes work, and they may be less familiar with the medications. “Either they prescribe these medications or they are asked to monitor them in place of the specialist provider who may have started them or suggested them,” he said. Metabolic monitoring may not be prioritized and can easily “slip through the cracks.” At the same time, doctors need to be aware of the risk of serious side effects of antipsychotic medications, such as malignant hyperthermia.
These medications can be overused and inappropriately used, which is a further complication. And when patients are taking multiple medications, there may be a need for additional monitoring and awareness of drug interactions.
“These medications are very complicated to use,” and there needs to be a way to connect primary care providers with child psychiatrists who are best trained in their use, said Dr. Volkmar.
A system with reminders can facilitate effective metabolic monitoring, he suggested. Dr. Volkmar has established a routine while providing care for a group home of adults with autism. Every 3 months, he reviews lab results. “You just have to force yourself to do it.”
Shared care arrangements may be another way to promote metabolic monitoring, Dr. Shenkman and colleagues said.
“Attributing care back to the multiple providers is important for care coordination and development of strategies to ensure that the evidence-based care is delivered and there is appropriate follow-up with the family and child to be sure care is received,” the study authors wrote. “Formalized shared care arrangements and adaptation of existing care delivery models to support integrated care, which can vary in degree from external coordination to on-site intervention and collaboration, are effective methods to promote partnership between primary and mental health providers.”
It is possible that clinicians in the study ordered metabolic monitoring but families did not take the children for testing, the investigators noted. In addition, it is not clear how much information providers have about other providers their patients are seeing.
The study authors and Dr. Volkmar had no disclosures.
The number and types of providers involved in a child’s care are associated with the likelihood that the child will receive metabolic monitoring, according to the study, which was published in Pediatrics.
The results suggest that primary care providers and mental health providers should collaborate to monitor children taking antipsychotics, the researchers said.
“Shared care arrangements between primary care physicians and mental health specialists significantly increased the chances that metabolic monitoring would be done, compared with care delivered by one provider,” reported Elizabeth A. Shenkman, PhD, chair of the department of health outcomes and biomedical informatics at the University of Florida, Gainesville, and colleagues. “The results of our study point to the importance of state Medicaid agencies and Medicaid managed care plans in identifying all providers caring for the children taking antipsychotic medication and using this information to engage the providers in quality improvement efforts to improve metabolic monitoring rates.”
Comparing specialties
Children who take antipsychotic medication are at risk for obesity, impaired glucose metabolism, and hyperlipidemia, but less than 40% receive recommended metabolic monitoring with glucose and cholesterol tests.
To examine how health care provider specialty influences the receipt of metabolic monitoring, Dr. Shenkman and colleagues analyzed Medicaid enrollment and health care and pharmacy claims data from Florida and Texas.
They focused on 41,078 children who had an antipsychotic medication dispensed at least twice in 2017 and were eligible for inclusion in the Centers for Medicare & Medicaid Services metabolic monitoring measure. The Metabolic Monitoring for Children and Adolescents on Antipsychotics measure is a “priority nationally and is currently on the CMS Child Core Set, which is used to annually assess state-specific performance on pediatric quality measures,” the authors wrote.
About 65% were boys, and the children had an average age of 12 years. The researchers compared metabolic monitoring rates when children received outpatient care from a primary care provider, a mental health provider with prescribing privileges, or both.
Less than 40% of the children received metabolic monitoring, that is, at least one diabetes test and at least one cholesterol test, during the year.
Most of the children (61%) saw both primary care providers and mental health providers. Approximately one-third had a primary care provider prescribe antipsychotic medication the majority of the time, and 60% had a mental health provider prescribe antipsychotic medication the majority of the time.
Patients who saw both types of providers were significantly more likely to receive metabolic monitoring, relative to those who saw primary care providers only (adjusted odds ratio, 1.42). Those seeing a mental health provider alone had adjusted odds of metabolic monitoring that were 23% lower than those seeing a primary care provider alone.
Children who had a mental health provider prescribe the medication the majority of the time were 25% more likely to receive metabolic monitoring, compared with those who had a primary care provider prescribing the medication the majority of the time.
Slipping through the cracks
Child psychiatrist Fred Volkmar, MD, commented that the results are “sadly” unsurprising and reflect issues that pertain to other psychotropic drugs as well as antipsychotics and to adults as well as children.
The researchers “are quite right to point to it,” and “we really do need to develop better plans for improving” monitoring, said Dr. Volkmar, the Irving B. Harris Professor in the Child Study Center and professor of psychology at Yale University, New Haven, Conn.
“Increasingly we are asking primary care providers ... to take care of folks who have important either developmental or mental health problems,” Dr. Volkmar said. While they can do a good job, they increasingly are underpaid. Monitoring patients takes work, and they may be less familiar with the medications. “Either they prescribe these medications or they are asked to monitor them in place of the specialist provider who may have started them or suggested them,” he said. Metabolic monitoring may not be prioritized and can easily “slip through the cracks.” At the same time, doctors need to be aware of the risk of serious side effects of antipsychotic medications, such as malignant hyperthermia.
These medications can be overused and inappropriately used, which is a further complication. And when patients are taking multiple medications, there may be a need for additional monitoring and awareness of drug interactions.
“These medications are very complicated to use,” and there needs to be a way to connect primary care providers with child psychiatrists who are best trained in their use, said Dr. Volkmar.
A system with reminders can facilitate effective metabolic monitoring, he suggested. Dr. Volkmar has established a routine while providing care for a group home of adults with autism. Every 3 months, he reviews lab results. “You just have to force yourself to do it.”
Shared care arrangements may be another way to promote metabolic monitoring, Dr. Shenkman and colleagues said.
“Attributing care back to the multiple providers is important for care coordination and development of strategies to ensure that the evidence-based care is delivered and there is appropriate follow-up with the family and child to be sure care is received,” the study authors wrote. “Formalized shared care arrangements and adaptation of existing care delivery models to support integrated care, which can vary in degree from external coordination to on-site intervention and collaboration, are effective methods to promote partnership between primary and mental health providers.”
It is possible that clinicians in the study ordered metabolic monitoring but families did not take the children for testing, the investigators noted. In addition, it is not clear how much information providers have about other providers their patients are seeing.
The study authors and Dr. Volkmar had no disclosures.
FROM PEDIATRICS
Pediatric HM highlights from the 2020 State of Hospital Medicine Report
To improve the pediatric data in the State of Hospital Medicine (SoHM) Report, the Practice Analysis Committee (PAC) developed a pediatric task force to recommend content specific to pediatric practice and garner support for survey participation. The pediatric hospital medicine (PHM) community responded with its usual enthusiasm, resulting in a threefold increase in PHM participation (99 groups), making the data from 2020 SoHM Report the most meaningful ever for pediatric practices.
However, data collection for the 2020 SoHM Report concluded in February, just before the face of medical practice and hospital care changed dramatically. A recent report at the virtual Pediatric Hospital Medicine meeting stated that pre–COVID-19 hospital operating margins had already taken a significant decline (from 5% to 2%-3%), putting pressure on pediatric programs in community settings that typically do not generate much revenue. After COVID-19, hospital revenues took an even greater downturn, affecting many hospital-based pediatric programs. While the future direction of many PHM programs remains unclear, the robust nature of the pediatric data in the 2020 SoHM Report defines where we were and where we once again hope to be. In addition, the PAC conducted a supplemental survey designed to assess the impact of COVID-19 on the practice of hospital medicine. Here’s a quick review of PHM highlights from the 2020 SoHM Report, with preliminary findings from the supplemental survey.
Diversity of service and scope of practice: pediatric hospitalist programs continue to provide a wide variety of services beyond care on inpatient wards, with the most common being procedure performance (56.6%), care of healthy newborns (51.5%), and rapid response team (38.4%) coverage. In addition, most PHM programs have a role in comanagement of a wide variety of patient populations, with the greatest presence among the surgical specialties. Approximately 90% of programs report some role in the care of patients admitted to general surgery, orthopedic surgery, and other surgical subspecialties. The role for comanagement with medical specialties remains diverse, with PHM programs routinely having some role in caring for patients hospitalized for neurologic, gastroenterological, cardiac concerns, and others. With the recent decline in hospital revenues affecting PHM practices, one way to ensure program value is to continue to diversify. Based on data from the 2020 SoHM report, broadening of clinical coverage will not require a significant change in practice for most PHM programs.
PHM board certification: With the first certifying exam for PHM taking place just months before SoHM data collection, the survey sought to establish a baseline percentage of providers board certified in PHM. With 98 groups responding, an average of 26.4% of PHM practitioners per group were reported to be board certified. While no difference was seen based on academic status, practitioners in PHM programs employed by a hospital, health system, or integrated delivery system were much more likely to be board certified than those employed by a university or medical school (31% vs. 20%). Regional differences were noted as well, with the East region reporting a much higher median proportion of PHM-certified physicians. It will be interesting to watch the trend in board certification status evolve over the upcoming years.
Anticipated change of budgeted full-time equivalents in the next year/post–COVID-19 analysis: Of the PHM programs responding to the SoHM Survey, 46.5% predicted an increase in budgeted full-time equivalents in the next year, while only 5.1% anticipated a decrease. Expecting this to change in response to COVID-19, the supplemental survey sought to update this information. Of the 30 PHM respondents to the supplemental survey, 41% instituted a temporary hiring freeze because of COVID-19, while 8.3% instituted a hiring freeze felt likely to be permanent. As PHM programs gear up for the next viral season, we wait to see whether the impact of COVID-19 will continue to be reflected in the volume and variety of patients admitted. It is clear that PHM programs will need to remain nimble to stay ahead of the changing landscape of practice in the days ahead. View all data by obtaining access to the 2020 SoHM Report at hospitalmedicine.org/sohm.
Many thanks to pediatric task force members Jack Percelay, MD; Vivien Kon-Ea Sun, MD; Marcos Mestre, MD; Ann Allen, MD; Dimple Khona, MD; Jeff Grill, MD; and Michelle Marks, MD.
Dr. Gage is director of faculty development, pediatric hospital medicine, at Phoenix Children’s Hospital, and associate professor of pediatrics at the University of Arizona, Phoenix.
To improve the pediatric data in the State of Hospital Medicine (SoHM) Report, the Practice Analysis Committee (PAC) developed a pediatric task force to recommend content specific to pediatric practice and garner support for survey participation. The pediatric hospital medicine (PHM) community responded with its usual enthusiasm, resulting in a threefold increase in PHM participation (99 groups), making the data from 2020 SoHM Report the most meaningful ever for pediatric practices.
However, data collection for the 2020 SoHM Report concluded in February, just before the face of medical practice and hospital care changed dramatically. A recent report at the virtual Pediatric Hospital Medicine meeting stated that pre–COVID-19 hospital operating margins had already taken a significant decline (from 5% to 2%-3%), putting pressure on pediatric programs in community settings that typically do not generate much revenue. After COVID-19, hospital revenues took an even greater downturn, affecting many hospital-based pediatric programs. While the future direction of many PHM programs remains unclear, the robust nature of the pediatric data in the 2020 SoHM Report defines where we were and where we once again hope to be. In addition, the PAC conducted a supplemental survey designed to assess the impact of COVID-19 on the practice of hospital medicine. Here’s a quick review of PHM highlights from the 2020 SoHM Report, with preliminary findings from the supplemental survey.
Diversity of service and scope of practice: pediatric hospitalist programs continue to provide a wide variety of services beyond care on inpatient wards, with the most common being procedure performance (56.6%), care of healthy newborns (51.5%), and rapid response team (38.4%) coverage. In addition, most PHM programs have a role in comanagement of a wide variety of patient populations, with the greatest presence among the surgical specialties. Approximately 90% of programs report some role in the care of patients admitted to general surgery, orthopedic surgery, and other surgical subspecialties. The role for comanagement with medical specialties remains diverse, with PHM programs routinely having some role in caring for patients hospitalized for neurologic, gastroenterological, cardiac concerns, and others. With the recent decline in hospital revenues affecting PHM practices, one way to ensure program value is to continue to diversify. Based on data from the 2020 SoHM report, broadening of clinical coverage will not require a significant change in practice for most PHM programs.
PHM board certification: With the first certifying exam for PHM taking place just months before SoHM data collection, the survey sought to establish a baseline percentage of providers board certified in PHM. With 98 groups responding, an average of 26.4% of PHM practitioners per group were reported to be board certified. While no difference was seen based on academic status, practitioners in PHM programs employed by a hospital, health system, or integrated delivery system were much more likely to be board certified than those employed by a university or medical school (31% vs. 20%). Regional differences were noted as well, with the East region reporting a much higher median proportion of PHM-certified physicians. It will be interesting to watch the trend in board certification status evolve over the upcoming years.
Anticipated change of budgeted full-time equivalents in the next year/post–COVID-19 analysis: Of the PHM programs responding to the SoHM Survey, 46.5% predicted an increase in budgeted full-time equivalents in the next year, while only 5.1% anticipated a decrease. Expecting this to change in response to COVID-19, the supplemental survey sought to update this information. Of the 30 PHM respondents to the supplemental survey, 41% instituted a temporary hiring freeze because of COVID-19, while 8.3% instituted a hiring freeze felt likely to be permanent. As PHM programs gear up for the next viral season, we wait to see whether the impact of COVID-19 will continue to be reflected in the volume and variety of patients admitted. It is clear that PHM programs will need to remain nimble to stay ahead of the changing landscape of practice in the days ahead. View all data by obtaining access to the 2020 SoHM Report at hospitalmedicine.org/sohm.
Many thanks to pediatric task force members Jack Percelay, MD; Vivien Kon-Ea Sun, MD; Marcos Mestre, MD; Ann Allen, MD; Dimple Khona, MD; Jeff Grill, MD; and Michelle Marks, MD.
Dr. Gage is director of faculty development, pediatric hospital medicine, at Phoenix Children’s Hospital, and associate professor of pediatrics at the University of Arizona, Phoenix.
To improve the pediatric data in the State of Hospital Medicine (SoHM) Report, the Practice Analysis Committee (PAC) developed a pediatric task force to recommend content specific to pediatric practice and garner support for survey participation. The pediatric hospital medicine (PHM) community responded with its usual enthusiasm, resulting in a threefold increase in PHM participation (99 groups), making the data from 2020 SoHM Report the most meaningful ever for pediatric practices.
However, data collection for the 2020 SoHM Report concluded in February, just before the face of medical practice and hospital care changed dramatically. A recent report at the virtual Pediatric Hospital Medicine meeting stated that pre–COVID-19 hospital operating margins had already taken a significant decline (from 5% to 2%-3%), putting pressure on pediatric programs in community settings that typically do not generate much revenue. After COVID-19, hospital revenues took an even greater downturn, affecting many hospital-based pediatric programs. While the future direction of many PHM programs remains unclear, the robust nature of the pediatric data in the 2020 SoHM Report defines where we were and where we once again hope to be. In addition, the PAC conducted a supplemental survey designed to assess the impact of COVID-19 on the practice of hospital medicine. Here’s a quick review of PHM highlights from the 2020 SoHM Report, with preliminary findings from the supplemental survey.
Diversity of service and scope of practice: pediatric hospitalist programs continue to provide a wide variety of services beyond care on inpatient wards, with the most common being procedure performance (56.6%), care of healthy newborns (51.5%), and rapid response team (38.4%) coverage. In addition, most PHM programs have a role in comanagement of a wide variety of patient populations, with the greatest presence among the surgical specialties. Approximately 90% of programs report some role in the care of patients admitted to general surgery, orthopedic surgery, and other surgical subspecialties. The role for comanagement with medical specialties remains diverse, with PHM programs routinely having some role in caring for patients hospitalized for neurologic, gastroenterological, cardiac concerns, and others. With the recent decline in hospital revenues affecting PHM practices, one way to ensure program value is to continue to diversify. Based on data from the 2020 SoHM report, broadening of clinical coverage will not require a significant change in practice for most PHM programs.
PHM board certification: With the first certifying exam for PHM taking place just months before SoHM data collection, the survey sought to establish a baseline percentage of providers board certified in PHM. With 98 groups responding, an average of 26.4% of PHM practitioners per group were reported to be board certified. While no difference was seen based on academic status, practitioners in PHM programs employed by a hospital, health system, or integrated delivery system were much more likely to be board certified than those employed by a university or medical school (31% vs. 20%). Regional differences were noted as well, with the East region reporting a much higher median proportion of PHM-certified physicians. It will be interesting to watch the trend in board certification status evolve over the upcoming years.
Anticipated change of budgeted full-time equivalents in the next year/post–COVID-19 analysis: Of the PHM programs responding to the SoHM Survey, 46.5% predicted an increase in budgeted full-time equivalents in the next year, while only 5.1% anticipated a decrease. Expecting this to change in response to COVID-19, the supplemental survey sought to update this information. Of the 30 PHM respondents to the supplemental survey, 41% instituted a temporary hiring freeze because of COVID-19, while 8.3% instituted a hiring freeze felt likely to be permanent. As PHM programs gear up for the next viral season, we wait to see whether the impact of COVID-19 will continue to be reflected in the volume and variety of patients admitted. It is clear that PHM programs will need to remain nimble to stay ahead of the changing landscape of practice in the days ahead. View all data by obtaining access to the 2020 SoHM Report at hospitalmedicine.org/sohm.
Many thanks to pediatric task force members Jack Percelay, MD; Vivien Kon-Ea Sun, MD; Marcos Mestre, MD; Ann Allen, MD; Dimple Khona, MD; Jeff Grill, MD; and Michelle Marks, MD.
Dr. Gage is director of faculty development, pediatric hospital medicine, at Phoenix Children’s Hospital, and associate professor of pediatrics at the University of Arizona, Phoenix.
Recognizing and intervening in child sex trafficking
THE CASE
Emily T.* is a 15-year-old, cisgender, homeless runaway. While on the streets, she was lured to a hotel where a “pimp” informed her she was going to work for him. She repeatedly tried to leave, but he would strike her, so she eventually succumbed. She was forced to have sex with several men and rarely allowed to use condoms.
On 1 occasion, when she went to a hospital with her pimp to visit a patient, her aunt (a nurse on duty at that facility) saw Ms. T and called the police. The pimp was arrested. Ms. T was interviewed by the police and gave a statement but refused a forensic exam.
Because of her involvement with the pimp, she was incarcerated. In prison, she was seen by a physician. On evaluation, she reported difficulty sleeping, flashbacks, and feelings of shame and guilt.1
●
* The patient’s name has been changed to protect her identity.
Child and adolescent sex trafficking is defined as the sexual exploitation of minors through force, fraud, or coercion. Specifically, it includes the recruitment, harboring, transportation, or advertising of a minor, and includes the exchange of anything of value in return for sexual activity. Commercial sexual exploitation and sex trafficking against minors include crimes such as prostitution; survival sex (exchanging sex/sexual acts for money or something of value, such as shelter, food, or drugs); pornography; sex tourism, mail-order-bride trade, and early marriage; or sexual performances (peep shows or strip clubs).2
Providing optimal care for children and adolescents exploited by sex trafficking depends on knowing risk factors, having an awareness of recommended screening and assessment tools, and employing a trauma-informed approach to interviews, examination, and support.2
Continue to: Recognize clues to trafficking
Recognize clues to trafficking
The Centers for Disease Control and Prevention (CDC) offers a framework for trafficking prevention. Health care providers are encouraged to use the CDC Social-Ecological Model which describes targeted prevention strategies at the individual, relationship, community, and societal levels.3
Risk factors for entering trafficking. Younger age increases a child’s vulnerability to exploitation, due to a lack of maturity, limited cognitive development, and ease of deception. The mean age of trafficking survivors is 15 years.4 History of child abuse and other traumatic experiences can lead children to run away from home. It is estimated that, once on the streets, most teens will be recruited by a trafficker within 48 hours.5 Poor self-esteem, depression, substance abuse, history of truancy, and early sexual maturation also increase the risk of becoming involved in trafficking (TABLE 1).2,6-8

Clinical findings suggestive of trafficking. Many physicians grasp the critical role they play in confronting trafficking, but they lack specific training, experience, and assessment tools.3 Notably, in 1 retrospective study, 46% of victims had been seen by a provider within the previous 2 months.6 One of the major challenges to identifying survivors of trafficking is to recognize critical signs, of which there are many (TABLE 12,6-8). Often these include a history of sexual assault, multiple pregnancies, requests for contraception at an early age, or evidence of physical injury.

Screen to identify trafficking
Universal, validated screening tools to accurately identify trafficked youth is an area of growing research. Some tools have been validated, but only for specific populations such as homeless or incarcerated youth or in emergency department (ED) patients. Tools for sex trafficking identification that may be useful in primary care include the Child Sex Trafficking (CST) screen validated in ED settings (TABLE 2),9 the Commercial Sexual Exploitation-Identification Tool (CSE-IT) in multiple settings (TABLE 3),10,11 and the Quick Youth Indicators for Trafficking (QYIT) in homeless youth (TABLE 4).11 The QYIT is the first validated labor and sex trafficking screening tool in homeless young adults. Children who screen positive for sex trafficking should be further assessed using a comprehensive tool.

Barriers to effective recognition of trafficked individuals. Financial factors limit access to health care. Also, survivors have cited multiple barriers for health professionals that prevent identification of survivors’ trafficked status.12,13 Once in the medical setting, disclosure is impacted by time constraints, fear of judgment by clinicians, and the risk of re-traumatization. Survivors have also cited lack of privacy, control strategies by their traffickers, lack of provider empathy, and fear of police as barriers to disclosure.14

Continue to: The medical impact of trafficking
The medical impact of trafficking
Sexual exploitation is a traumatic experience that is known to cause harm across multiple domains including serious physical injuries related to violence, as well as reproductive and mental health consequences.15,16
Acute and chronic illnesses. In an initial evaluation, assess the survivor’s acute medical conditions (TABLE 5).15,16 Common acute issues include physical injuries due to assault, infections, and reproductive complications.17 Health concerns can also result from stressors such as deprivation of food and sleep, hazardous living conditions, and limited access to care.17

As part of caring for sex-trafficking survivors, assess for, and treat, chronic health issues such as pain, gastrointestinal complaints, poor dental care, malnutrition, and fatigue.16,18 Substance use, as well as chronic mental health concerns (eg, anxiety, depression, posttraumatic stress disorder [PTSD]) may also influence the clinical presentation.
Physical injuries. A cross-sectional study of female survivors of sex trafficking in the United States found that 89% sustained physical injury resulting from violence, including fractures, open wounds, head injury, dental problems, burns, and anogenital trauma.16,17 In many cases, acute injury may not be present in the clinical setting since care is often delayed, but a full examination can reveal signs of prior trauma.16
Reproductive health concerns. Significant, long-term impact on reproductive health can result due to forced penetration by multiple perpetrators, sodomy, and sex without protection or lubricants. Survivors are therefore at high risk for unplanned and unwanted pregnancies, sexually transmitted infections (STIs), pelvic pain, and infertility.16
Continue to: Psychological effects and trauma exposure
Psychological effects and trauma exposure. Survivors often have experienced abuse and neglect prior to commercial exploitation, and they may exhibit long-term sequelae. Survivors may present with major depression, anxiety, panic attacks, suicidality, addiction, PTSD, or aggression.16 Long-term sequelae for patients can include dysfunctional relationships due to an inability to trust, self-destructive behaviors, and significant shame.2,18
Care and treatment of trafficked youth
Initial presentation may occur in a variety of health care settings. Use a trauma-informed approach emphasizing physical and emotional safety and positive relationships, to reduce risk to the survivor, staff, and providers.19 Establishing trust and rapport may provide better short-term safety, as well as help build stronger long-term relationships that can lead to better health outcomes.
Clinical examination. Provide traumatized patients with a sense of safety, control, and autonomy in the health care setting. During the physical exam, be aware of the impact of re-traumatization as patients are asked to undress, endure sensitive examinations, and undergo invasive procedures. Explain the examination, ask for permission at each step, and use slow movements. Allow the patient to guide certain sensitive exams.15 Adopt an approach that recognizes the impact of trauma, avoids revictimization, and acknowledges the resilience of survivors.20
Treatment plan. After the initial exam, treat acute physical injuries and determine if any further testing is needed. Offer emergency contraception, STI prophylaxis, pre-exposure prophylaxis, and vaccines.15 After assessing patient readiness, offer local resources, identify safe methods for communication, identify individuals who could intervene in a crisis, and consider a safety plan (TABLE 6).

Coordination of care. Consider referrals to behavioral health services, substance recovery centers, food programs, housing resources, and a primary care clinic.15 ED clinicians may be asked to complete a sexual or physical assault forensic examination. After obtaining informed consent, one needs to19
- document skin signs such as scars, bites, strangulation marks, and tattoos. Note the size, shape, color, location, and other characteristics of each lesion.
- perform an oral, genital, and rectal examination and use a sexual assault evidence kit as indicated.
- use body diagrams and take photographs of all injuries/physical findings.
- order diagnostic testing as appropriate (eg, imaging to assess fractures).
Continue to: THE CASE
THE CASE
During Ms. T’s incarceration, she was tested for STIs and treated for gonorrhea, trichomonas, and bacterial vaginosis. She was educated about sexual health, was counseled on contraception, and accepted condoms. She was referred to a therapist and given information on additional community resources she could contact upon her release.
A year after her release, she was incarcerated again. She also had an unplanned pregnancy. With the support she received from community programs, social workers, and her primary care provider, she moved in with her family, where she is currently living. She denies any ongoing trafficking activity.
CORRESPONDENCE
Piali Basu, DO, MPH, UCSF Primary Care, 185 Berry Street, Lobby 1, Suite 1000, San Francisco, CA 94107; [email protected]
1. Samko Tracey (Department of Pediatrics, LA County + University of Southern California, Los Angeles, CA). Conversation with: Vidhi Doshi (Department of Internal Medicine, University of California San Francisco, San Francisco, CA). July 8, 2019.
2. Institute of Medicine; National Research Council. Confronting Commercial Sexual Exploitation and Sex Trafficking of Minors in the United States. Washington, DC: The National Academies Press; 2013.
3. Greenbaum VJ, Titchen K, Walker-Descartes I, et al. Multi-level prevention of human trafficking: the role of health care professionals. Prev Med. 2018;114:164-167.
4. Smith LA, Vardman SH, Snow MA. The national report on domestic minor sex trafficking: America’s prostituted children. 2009. Accessed January 11, 2021. http://sharedhope.org/wp-content/uploads/2012/09/SHI_National_Report_on_DMST_2009.pdf
5. Know the facts: commercial sexual exploitation of children. In: Connections. Summer 2011. Washington Coalition of Sexual Assault Programs. Accessed January 11, 2021. www.wcsap.org/sites/default/files/uploads/resources_publications/connections/Commercial_Sexual_Exploitation_of_Youth_2011.pdf
6. Varma S, Gillespie S, McCracken C, et al. Characteristics of child commercial sexual exploitation and sex trafficking victims presenting for medical care in the United States. Child Abuse Negl. 2015;44:98-105.
7. Walker K, California Child Welfare Council. Ending the commercial sexual exploitation of children: a call for multi-system collaboration in California. 2013. Accessed January 11, 2021. https://youthlaw.org/wp-content/uploads/2015/01/Ending-CSEC-A-Call-for-Multi-System_Collaboration-in-CA.pdf
8. Landers M, McGrath K, Johnson MH, et al. Baseline characteristics of dependent youth who have been commercially sexually exploited: findings from a specialized treatment program. J Child Sex Abus. 2017;26:692-709.
9. Greenbaum VJ, Dodd M, McCracken C. A short screening tool to identify victims of child sex trafficking in the health care setting. Pediatr Emerg Care. 2018;34:33-37.
10. Sy E, Quach T, Lee J, et al. Responding to commercially sexually exploited children (CSEC): a community health center‘s journey towards creating a primary care clinical CSEC screening tool in the United States. Inter J Soc Sci Stud. 2016;4:45-51.
11. Chisolm-Straker M, Sze J, Einbond J, et al. Screening for human trafficking among homeless young adults. Childr Youth Serv Rev. 2019;98:72-79.
12. Richie-Zavaleta AC, Villanueva A, Martinez-Donate A, et al. Sex trafficking victims at their junction with the healthcare setting—a mixed-methods inquiry. J Hum Traffick. 2020;6:1-29.
13. Chisolm-Straker M, Miller CL, Duke G, et al. A framework for the development of healthcare provider education programs on human trafficking part two: survivors. J Hum Traffick. 2019;5:410-424.
14. Heilemann T, Santhiveeran J. How do female adolescents cope and survive the hardships of prostitution? A content analysis of existing literature. J Ethn Cult Divers Soc Work. 2011;20:57-76.
15. Greenbaum J, Crawford-Jakubiak JE, Committee on Child Abuse and Neglect. Child sex trafficking and commercial sexual exploitation: health care needs of victims. Pediatrics. 2015;135:566-574.
16. Barnert E, Iqbal Z, Bruce J, et al. Commercial sexual exploitation and sex trafficking of children and adolescents: a narrative review. Acad Pediatr. 2017;17:825-829.
17. Dovydaitis T. Human trafficking: the role of the health care provider. J Midwifery Womens Health. 2010;55:462-467.
18. English A, Kivlahan C. Human rights and human trafficking of adolescents: legal and clinical perspectives. In: Titchen K, Miller E, Eds. Medical Perspectives on Human Trafficking in Adolescents. Springer Nature; 2020:21-41.
19. Price K, Nelson BD, Macias-Konstantopoulos WL. Understanding health care access disparities among human trafficking survivors: profiles of health care experiences, access, and engagement. J Interpers Violence. 2019; doi: 10.1177/0886260519889934.
20. Chambers R, Ravi A, Paulus S. Human trafficking: how family physicians can recognize and assist victims. Am Fam Physician. 2019;100:202-204.
THE CASE
Emily T.* is a 15-year-old, cisgender, homeless runaway. While on the streets, she was lured to a hotel where a “pimp” informed her she was going to work for him. She repeatedly tried to leave, but he would strike her, so she eventually succumbed. She was forced to have sex with several men and rarely allowed to use condoms.
On 1 occasion, when she went to a hospital with her pimp to visit a patient, her aunt (a nurse on duty at that facility) saw Ms. T and called the police. The pimp was arrested. Ms. T was interviewed by the police and gave a statement but refused a forensic exam.
Because of her involvement with the pimp, she was incarcerated. In prison, she was seen by a physician. On evaluation, she reported difficulty sleeping, flashbacks, and feelings of shame and guilt.1
●
* The patient’s name has been changed to protect her identity.
Child and adolescent sex trafficking is defined as the sexual exploitation of minors through force, fraud, or coercion. Specifically, it includes the recruitment, harboring, transportation, or advertising of a minor, and includes the exchange of anything of value in return for sexual activity. Commercial sexual exploitation and sex trafficking against minors include crimes such as prostitution; survival sex (exchanging sex/sexual acts for money or something of value, such as shelter, food, or drugs); pornography; sex tourism, mail-order-bride trade, and early marriage; or sexual performances (peep shows or strip clubs).2
Providing optimal care for children and adolescents exploited by sex trafficking depends on knowing risk factors, having an awareness of recommended screening and assessment tools, and employing a trauma-informed approach to interviews, examination, and support.2
Continue to: Recognize clues to trafficking
Recognize clues to trafficking
The Centers for Disease Control and Prevention (CDC) offers a framework for trafficking prevention. Health care providers are encouraged to use the CDC Social-Ecological Model which describes targeted prevention strategies at the individual, relationship, community, and societal levels.3
Risk factors for entering trafficking. Younger age increases a child’s vulnerability to exploitation, due to a lack of maturity, limited cognitive development, and ease of deception. The mean age of trafficking survivors is 15 years.4 History of child abuse and other traumatic experiences can lead children to run away from home. It is estimated that, once on the streets, most teens will be recruited by a trafficker within 48 hours.5 Poor self-esteem, depression, substance abuse, history of truancy, and early sexual maturation also increase the risk of becoming involved in trafficking (TABLE 1).2,6-8

Clinical findings suggestive of trafficking. Many physicians grasp the critical role they play in confronting trafficking, but they lack specific training, experience, and assessment tools.3 Notably, in 1 retrospective study, 46% of victims had been seen by a provider within the previous 2 months.6 One of the major challenges to identifying survivors of trafficking is to recognize critical signs, of which there are many (TABLE 12,6-8). Often these include a history of sexual assault, multiple pregnancies, requests for contraception at an early age, or evidence of physical injury.

Screen to identify trafficking
Universal, validated screening tools to accurately identify trafficked youth is an area of growing research. Some tools have been validated, but only for specific populations such as homeless or incarcerated youth or in emergency department (ED) patients. Tools for sex trafficking identification that may be useful in primary care include the Child Sex Trafficking (CST) screen validated in ED settings (TABLE 2),9 the Commercial Sexual Exploitation-Identification Tool (CSE-IT) in multiple settings (TABLE 3),10,11 and the Quick Youth Indicators for Trafficking (QYIT) in homeless youth (TABLE 4).11 The QYIT is the first validated labor and sex trafficking screening tool in homeless young adults. Children who screen positive for sex trafficking should be further assessed using a comprehensive tool.

Barriers to effective recognition of trafficked individuals. Financial factors limit access to health care. Also, survivors have cited multiple barriers for health professionals that prevent identification of survivors’ trafficked status.12,13 Once in the medical setting, disclosure is impacted by time constraints, fear of judgment by clinicians, and the risk of re-traumatization. Survivors have also cited lack of privacy, control strategies by their traffickers, lack of provider empathy, and fear of police as barriers to disclosure.14

Continue to: The medical impact of trafficking
The medical impact of trafficking
Sexual exploitation is a traumatic experience that is known to cause harm across multiple domains including serious physical injuries related to violence, as well as reproductive and mental health consequences.15,16
Acute and chronic illnesses. In an initial evaluation, assess the survivor’s acute medical conditions (TABLE 5).15,16 Common acute issues include physical injuries due to assault, infections, and reproductive complications.17 Health concerns can also result from stressors such as deprivation of food and sleep, hazardous living conditions, and limited access to care.17

As part of caring for sex-trafficking survivors, assess for, and treat, chronic health issues such as pain, gastrointestinal complaints, poor dental care, malnutrition, and fatigue.16,18 Substance use, as well as chronic mental health concerns (eg, anxiety, depression, posttraumatic stress disorder [PTSD]) may also influence the clinical presentation.
Physical injuries. A cross-sectional study of female survivors of sex trafficking in the United States found that 89% sustained physical injury resulting from violence, including fractures, open wounds, head injury, dental problems, burns, and anogenital trauma.16,17 In many cases, acute injury may not be present in the clinical setting since care is often delayed, but a full examination can reveal signs of prior trauma.16
Reproductive health concerns. Significant, long-term impact on reproductive health can result due to forced penetration by multiple perpetrators, sodomy, and sex without protection or lubricants. Survivors are therefore at high risk for unplanned and unwanted pregnancies, sexually transmitted infections (STIs), pelvic pain, and infertility.16
Continue to: Psychological effects and trauma exposure
Psychological effects and trauma exposure. Survivors often have experienced abuse and neglect prior to commercial exploitation, and they may exhibit long-term sequelae. Survivors may present with major depression, anxiety, panic attacks, suicidality, addiction, PTSD, or aggression.16 Long-term sequelae for patients can include dysfunctional relationships due to an inability to trust, self-destructive behaviors, and significant shame.2,18
Care and treatment of trafficked youth
Initial presentation may occur in a variety of health care settings. Use a trauma-informed approach emphasizing physical and emotional safety and positive relationships, to reduce risk to the survivor, staff, and providers.19 Establishing trust and rapport may provide better short-term safety, as well as help build stronger long-term relationships that can lead to better health outcomes.
Clinical examination. Provide traumatized patients with a sense of safety, control, and autonomy in the health care setting. During the physical exam, be aware of the impact of re-traumatization as patients are asked to undress, endure sensitive examinations, and undergo invasive procedures. Explain the examination, ask for permission at each step, and use slow movements. Allow the patient to guide certain sensitive exams.15 Adopt an approach that recognizes the impact of trauma, avoids revictimization, and acknowledges the resilience of survivors.20
Treatment plan. After the initial exam, treat acute physical injuries and determine if any further testing is needed. Offer emergency contraception, STI prophylaxis, pre-exposure prophylaxis, and vaccines.15 After assessing patient readiness, offer local resources, identify safe methods for communication, identify individuals who could intervene in a crisis, and consider a safety plan (TABLE 6).

Coordination of care. Consider referrals to behavioral health services, substance recovery centers, food programs, housing resources, and a primary care clinic.15 ED clinicians may be asked to complete a sexual or physical assault forensic examination. After obtaining informed consent, one needs to19
- document skin signs such as scars, bites, strangulation marks, and tattoos. Note the size, shape, color, location, and other characteristics of each lesion.
- perform an oral, genital, and rectal examination and use a sexual assault evidence kit as indicated.
- use body diagrams and take photographs of all injuries/physical findings.
- order diagnostic testing as appropriate (eg, imaging to assess fractures).
Continue to: THE CASE
THE CASE
During Ms. T’s incarceration, she was tested for STIs and treated for gonorrhea, trichomonas, and bacterial vaginosis. She was educated about sexual health, was counseled on contraception, and accepted condoms. She was referred to a therapist and given information on additional community resources she could contact upon her release.
A year after her release, she was incarcerated again. She also had an unplanned pregnancy. With the support she received from community programs, social workers, and her primary care provider, she moved in with her family, where she is currently living. She denies any ongoing trafficking activity.
CORRESPONDENCE
Piali Basu, DO, MPH, UCSF Primary Care, 185 Berry Street, Lobby 1, Suite 1000, San Francisco, CA 94107; [email protected]
THE CASE
Emily T.* is a 15-year-old, cisgender, homeless runaway. While on the streets, she was lured to a hotel where a “pimp” informed her she was going to work for him. She repeatedly tried to leave, but he would strike her, so she eventually succumbed. She was forced to have sex with several men and rarely allowed to use condoms.
On 1 occasion, when she went to a hospital with her pimp to visit a patient, her aunt (a nurse on duty at that facility) saw Ms. T and called the police. The pimp was arrested. Ms. T was interviewed by the police and gave a statement but refused a forensic exam.
Because of her involvement with the pimp, she was incarcerated. In prison, she was seen by a physician. On evaluation, she reported difficulty sleeping, flashbacks, and feelings of shame and guilt.1
●
* The patient’s name has been changed to protect her identity.
Child and adolescent sex trafficking is defined as the sexual exploitation of minors through force, fraud, or coercion. Specifically, it includes the recruitment, harboring, transportation, or advertising of a minor, and includes the exchange of anything of value in return for sexual activity. Commercial sexual exploitation and sex trafficking against minors include crimes such as prostitution; survival sex (exchanging sex/sexual acts for money or something of value, such as shelter, food, or drugs); pornography; sex tourism, mail-order-bride trade, and early marriage; or sexual performances (peep shows or strip clubs).2
Providing optimal care for children and adolescents exploited by sex trafficking depends on knowing risk factors, having an awareness of recommended screening and assessment tools, and employing a trauma-informed approach to interviews, examination, and support.2
Continue to: Recognize clues to trafficking
Recognize clues to trafficking
The Centers for Disease Control and Prevention (CDC) offers a framework for trafficking prevention. Health care providers are encouraged to use the CDC Social-Ecological Model which describes targeted prevention strategies at the individual, relationship, community, and societal levels.3
Risk factors for entering trafficking. Younger age increases a child’s vulnerability to exploitation, due to a lack of maturity, limited cognitive development, and ease of deception. The mean age of trafficking survivors is 15 years.4 History of child abuse and other traumatic experiences can lead children to run away from home. It is estimated that, once on the streets, most teens will be recruited by a trafficker within 48 hours.5 Poor self-esteem, depression, substance abuse, history of truancy, and early sexual maturation also increase the risk of becoming involved in trafficking (TABLE 1).2,6-8

Clinical findings suggestive of trafficking. Many physicians grasp the critical role they play in confronting trafficking, but they lack specific training, experience, and assessment tools.3 Notably, in 1 retrospective study, 46% of victims had been seen by a provider within the previous 2 months.6 One of the major challenges to identifying survivors of trafficking is to recognize critical signs, of which there are many (TABLE 12,6-8). Often these include a history of sexual assault, multiple pregnancies, requests for contraception at an early age, or evidence of physical injury.

Screen to identify trafficking
Universal, validated screening tools to accurately identify trafficked youth is an area of growing research. Some tools have been validated, but only for specific populations such as homeless or incarcerated youth or in emergency department (ED) patients. Tools for sex trafficking identification that may be useful in primary care include the Child Sex Trafficking (CST) screen validated in ED settings (TABLE 2),9 the Commercial Sexual Exploitation-Identification Tool (CSE-IT) in multiple settings (TABLE 3),10,11 and the Quick Youth Indicators for Trafficking (QYIT) in homeless youth (TABLE 4).11 The QYIT is the first validated labor and sex trafficking screening tool in homeless young adults. Children who screen positive for sex trafficking should be further assessed using a comprehensive tool.

Barriers to effective recognition of trafficked individuals. Financial factors limit access to health care. Also, survivors have cited multiple barriers for health professionals that prevent identification of survivors’ trafficked status.12,13 Once in the medical setting, disclosure is impacted by time constraints, fear of judgment by clinicians, and the risk of re-traumatization. Survivors have also cited lack of privacy, control strategies by their traffickers, lack of provider empathy, and fear of police as barriers to disclosure.14

Continue to: The medical impact of trafficking
The medical impact of trafficking
Sexual exploitation is a traumatic experience that is known to cause harm across multiple domains including serious physical injuries related to violence, as well as reproductive and mental health consequences.15,16
Acute and chronic illnesses. In an initial evaluation, assess the survivor’s acute medical conditions (TABLE 5).15,16 Common acute issues include physical injuries due to assault, infections, and reproductive complications.17 Health concerns can also result from stressors such as deprivation of food and sleep, hazardous living conditions, and limited access to care.17

As part of caring for sex-trafficking survivors, assess for, and treat, chronic health issues such as pain, gastrointestinal complaints, poor dental care, malnutrition, and fatigue.16,18 Substance use, as well as chronic mental health concerns (eg, anxiety, depression, posttraumatic stress disorder [PTSD]) may also influence the clinical presentation.
Physical injuries. A cross-sectional study of female survivors of sex trafficking in the United States found that 89% sustained physical injury resulting from violence, including fractures, open wounds, head injury, dental problems, burns, and anogenital trauma.16,17 In many cases, acute injury may not be present in the clinical setting since care is often delayed, but a full examination can reveal signs of prior trauma.16
Reproductive health concerns. Significant, long-term impact on reproductive health can result due to forced penetration by multiple perpetrators, sodomy, and sex without protection or lubricants. Survivors are therefore at high risk for unplanned and unwanted pregnancies, sexually transmitted infections (STIs), pelvic pain, and infertility.16
Continue to: Psychological effects and trauma exposure
Psychological effects and trauma exposure. Survivors often have experienced abuse and neglect prior to commercial exploitation, and they may exhibit long-term sequelae. Survivors may present with major depression, anxiety, panic attacks, suicidality, addiction, PTSD, or aggression.16 Long-term sequelae for patients can include dysfunctional relationships due to an inability to trust, self-destructive behaviors, and significant shame.2,18
Care and treatment of trafficked youth
Initial presentation may occur in a variety of health care settings. Use a trauma-informed approach emphasizing physical and emotional safety and positive relationships, to reduce risk to the survivor, staff, and providers.19 Establishing trust and rapport may provide better short-term safety, as well as help build stronger long-term relationships that can lead to better health outcomes.
Clinical examination. Provide traumatized patients with a sense of safety, control, and autonomy in the health care setting. During the physical exam, be aware of the impact of re-traumatization as patients are asked to undress, endure sensitive examinations, and undergo invasive procedures. Explain the examination, ask for permission at each step, and use slow movements. Allow the patient to guide certain sensitive exams.15 Adopt an approach that recognizes the impact of trauma, avoids revictimization, and acknowledges the resilience of survivors.20
Treatment plan. After the initial exam, treat acute physical injuries and determine if any further testing is needed. Offer emergency contraception, STI prophylaxis, pre-exposure prophylaxis, and vaccines.15 After assessing patient readiness, offer local resources, identify safe methods for communication, identify individuals who could intervene in a crisis, and consider a safety plan (TABLE 6).

Coordination of care. Consider referrals to behavioral health services, substance recovery centers, food programs, housing resources, and a primary care clinic.15 ED clinicians may be asked to complete a sexual or physical assault forensic examination. After obtaining informed consent, one needs to19
- document skin signs such as scars, bites, strangulation marks, and tattoos. Note the size, shape, color, location, and other characteristics of each lesion.
- perform an oral, genital, and rectal examination and use a sexual assault evidence kit as indicated.
- use body diagrams and take photographs of all injuries/physical findings.
- order diagnostic testing as appropriate (eg, imaging to assess fractures).
Continue to: THE CASE
THE CASE
During Ms. T’s incarceration, she was tested for STIs and treated for gonorrhea, trichomonas, and bacterial vaginosis. She was educated about sexual health, was counseled on contraception, and accepted condoms. She was referred to a therapist and given information on additional community resources she could contact upon her release.
A year after her release, she was incarcerated again. She also had an unplanned pregnancy. With the support she received from community programs, social workers, and her primary care provider, she moved in with her family, where she is currently living. She denies any ongoing trafficking activity.
CORRESPONDENCE
Piali Basu, DO, MPH, UCSF Primary Care, 185 Berry Street, Lobby 1, Suite 1000, San Francisco, CA 94107; [email protected]
1. Samko Tracey (Department of Pediatrics, LA County + University of Southern California, Los Angeles, CA). Conversation with: Vidhi Doshi (Department of Internal Medicine, University of California San Francisco, San Francisco, CA). July 8, 2019.
2. Institute of Medicine; National Research Council. Confronting Commercial Sexual Exploitation and Sex Trafficking of Minors in the United States. Washington, DC: The National Academies Press; 2013.
3. Greenbaum VJ, Titchen K, Walker-Descartes I, et al. Multi-level prevention of human trafficking: the role of health care professionals. Prev Med. 2018;114:164-167.
4. Smith LA, Vardman SH, Snow MA. The national report on domestic minor sex trafficking: America’s prostituted children. 2009. Accessed January 11, 2021. http://sharedhope.org/wp-content/uploads/2012/09/SHI_National_Report_on_DMST_2009.pdf
5. Know the facts: commercial sexual exploitation of children. In: Connections. Summer 2011. Washington Coalition of Sexual Assault Programs. Accessed January 11, 2021. www.wcsap.org/sites/default/files/uploads/resources_publications/connections/Commercial_Sexual_Exploitation_of_Youth_2011.pdf
6. Varma S, Gillespie S, McCracken C, et al. Characteristics of child commercial sexual exploitation and sex trafficking victims presenting for medical care in the United States. Child Abuse Negl. 2015;44:98-105.
7. Walker K, California Child Welfare Council. Ending the commercial sexual exploitation of children: a call for multi-system collaboration in California. 2013. Accessed January 11, 2021. https://youthlaw.org/wp-content/uploads/2015/01/Ending-CSEC-A-Call-for-Multi-System_Collaboration-in-CA.pdf
8. Landers M, McGrath K, Johnson MH, et al. Baseline characteristics of dependent youth who have been commercially sexually exploited: findings from a specialized treatment program. J Child Sex Abus. 2017;26:692-709.
9. Greenbaum VJ, Dodd M, McCracken C. A short screening tool to identify victims of child sex trafficking in the health care setting. Pediatr Emerg Care. 2018;34:33-37.
10. Sy E, Quach T, Lee J, et al. Responding to commercially sexually exploited children (CSEC): a community health center‘s journey towards creating a primary care clinical CSEC screening tool in the United States. Inter J Soc Sci Stud. 2016;4:45-51.
11. Chisolm-Straker M, Sze J, Einbond J, et al. Screening for human trafficking among homeless young adults. Childr Youth Serv Rev. 2019;98:72-79.
12. Richie-Zavaleta AC, Villanueva A, Martinez-Donate A, et al. Sex trafficking victims at their junction with the healthcare setting—a mixed-methods inquiry. J Hum Traffick. 2020;6:1-29.
13. Chisolm-Straker M, Miller CL, Duke G, et al. A framework for the development of healthcare provider education programs on human trafficking part two: survivors. J Hum Traffick. 2019;5:410-424.
14. Heilemann T, Santhiveeran J. How do female adolescents cope and survive the hardships of prostitution? A content analysis of existing literature. J Ethn Cult Divers Soc Work. 2011;20:57-76.
15. Greenbaum J, Crawford-Jakubiak JE, Committee on Child Abuse and Neglect. Child sex trafficking and commercial sexual exploitation: health care needs of victims. Pediatrics. 2015;135:566-574.
16. Barnert E, Iqbal Z, Bruce J, et al. Commercial sexual exploitation and sex trafficking of children and adolescents: a narrative review. Acad Pediatr. 2017;17:825-829.
17. Dovydaitis T. Human trafficking: the role of the health care provider. J Midwifery Womens Health. 2010;55:462-467.
18. English A, Kivlahan C. Human rights and human trafficking of adolescents: legal and clinical perspectives. In: Titchen K, Miller E, Eds. Medical Perspectives on Human Trafficking in Adolescents. Springer Nature; 2020:21-41.
19. Price K, Nelson BD, Macias-Konstantopoulos WL. Understanding health care access disparities among human trafficking survivors: profiles of health care experiences, access, and engagement. J Interpers Violence. 2019; doi: 10.1177/0886260519889934.
20. Chambers R, Ravi A, Paulus S. Human trafficking: how family physicians can recognize and assist victims. Am Fam Physician. 2019;100:202-204.
1. Samko Tracey (Department of Pediatrics, LA County + University of Southern California, Los Angeles, CA). Conversation with: Vidhi Doshi (Department of Internal Medicine, University of California San Francisco, San Francisco, CA). July 8, 2019.
2. Institute of Medicine; National Research Council. Confronting Commercial Sexual Exploitation and Sex Trafficking of Minors in the United States. Washington, DC: The National Academies Press; 2013.
3. Greenbaum VJ, Titchen K, Walker-Descartes I, et al. Multi-level prevention of human trafficking: the role of health care professionals. Prev Med. 2018;114:164-167.
4. Smith LA, Vardman SH, Snow MA. The national report on domestic minor sex trafficking: America’s prostituted children. 2009. Accessed January 11, 2021. http://sharedhope.org/wp-content/uploads/2012/09/SHI_National_Report_on_DMST_2009.pdf
5. Know the facts: commercial sexual exploitation of children. In: Connections. Summer 2011. Washington Coalition of Sexual Assault Programs. Accessed January 11, 2021. www.wcsap.org/sites/default/files/uploads/resources_publications/connections/Commercial_Sexual_Exploitation_of_Youth_2011.pdf
6. Varma S, Gillespie S, McCracken C, et al. Characteristics of child commercial sexual exploitation and sex trafficking victims presenting for medical care in the United States. Child Abuse Negl. 2015;44:98-105.
7. Walker K, California Child Welfare Council. Ending the commercial sexual exploitation of children: a call for multi-system collaboration in California. 2013. Accessed January 11, 2021. https://youthlaw.org/wp-content/uploads/2015/01/Ending-CSEC-A-Call-for-Multi-System_Collaboration-in-CA.pdf
8. Landers M, McGrath K, Johnson MH, et al. Baseline characteristics of dependent youth who have been commercially sexually exploited: findings from a specialized treatment program. J Child Sex Abus. 2017;26:692-709.
9. Greenbaum VJ, Dodd M, McCracken C. A short screening tool to identify victims of child sex trafficking in the health care setting. Pediatr Emerg Care. 2018;34:33-37.
10. Sy E, Quach T, Lee J, et al. Responding to commercially sexually exploited children (CSEC): a community health center‘s journey towards creating a primary care clinical CSEC screening tool in the United States. Inter J Soc Sci Stud. 2016;4:45-51.
11. Chisolm-Straker M, Sze J, Einbond J, et al. Screening for human trafficking among homeless young adults. Childr Youth Serv Rev. 2019;98:72-79.
12. Richie-Zavaleta AC, Villanueva A, Martinez-Donate A, et al. Sex trafficking victims at their junction with the healthcare setting—a mixed-methods inquiry. J Hum Traffick. 2020;6:1-29.
13. Chisolm-Straker M, Miller CL, Duke G, et al. A framework for the development of healthcare provider education programs on human trafficking part two: survivors. J Hum Traffick. 2019;5:410-424.
14. Heilemann T, Santhiveeran J. How do female adolescents cope and survive the hardships of prostitution? A content analysis of existing literature. J Ethn Cult Divers Soc Work. 2011;20:57-76.
15. Greenbaum J, Crawford-Jakubiak JE, Committee on Child Abuse and Neglect. Child sex trafficking and commercial sexual exploitation: health care needs of victims. Pediatrics. 2015;135:566-574.
16. Barnert E, Iqbal Z, Bruce J, et al. Commercial sexual exploitation and sex trafficking of children and adolescents: a narrative review. Acad Pediatr. 2017;17:825-829.
17. Dovydaitis T. Human trafficking: the role of the health care provider. J Midwifery Womens Health. 2010;55:462-467.
18. English A, Kivlahan C. Human rights and human trafficking of adolescents: legal and clinical perspectives. In: Titchen K, Miller E, Eds. Medical Perspectives on Human Trafficking in Adolescents. Springer Nature; 2020:21-41.
19. Price K, Nelson BD, Macias-Konstantopoulos WL. Understanding health care access disparities among human trafficking survivors: profiles of health care experiences, access, and engagement. J Interpers Violence. 2019; doi: 10.1177/0886260519889934.
20. Chambers R, Ravi A, Paulus S. Human trafficking: how family physicians can recognize and assist victims. Am Fam Physician. 2019;100:202-204.
Atopic dermatitis: More than just a rash
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.
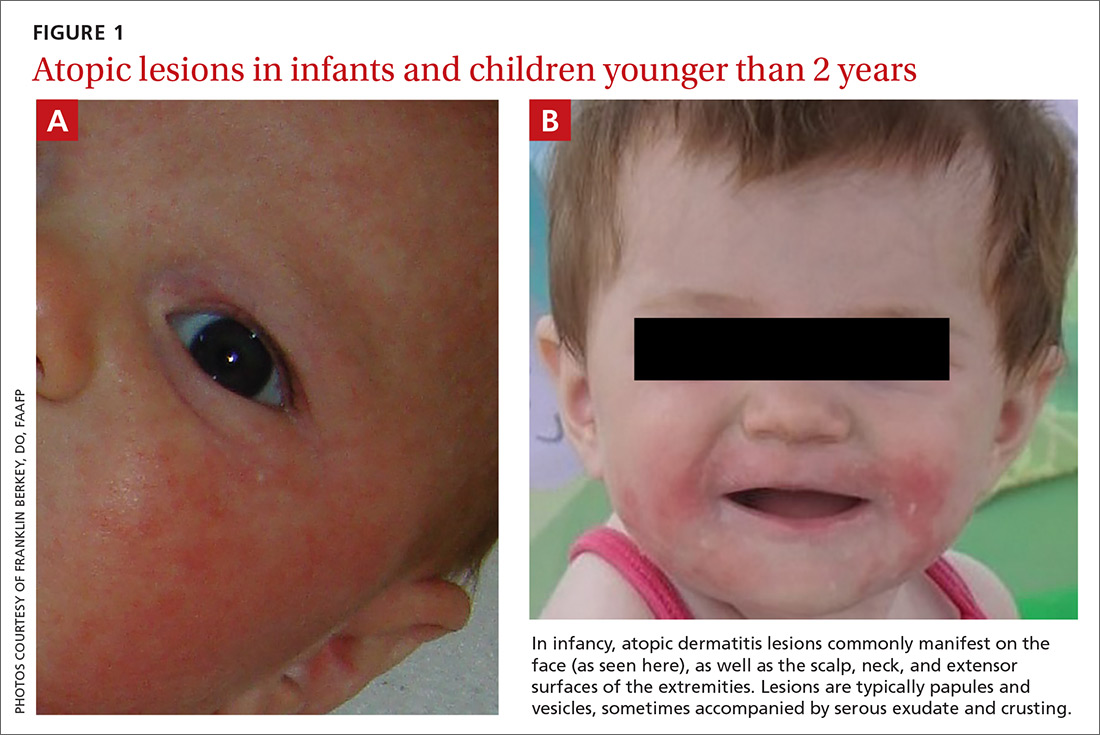
Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2
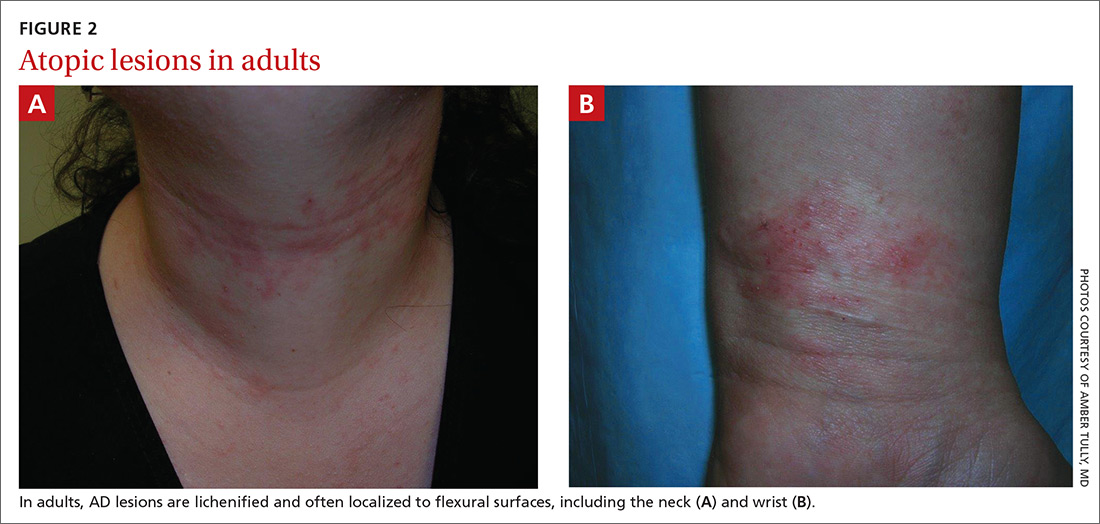
Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of
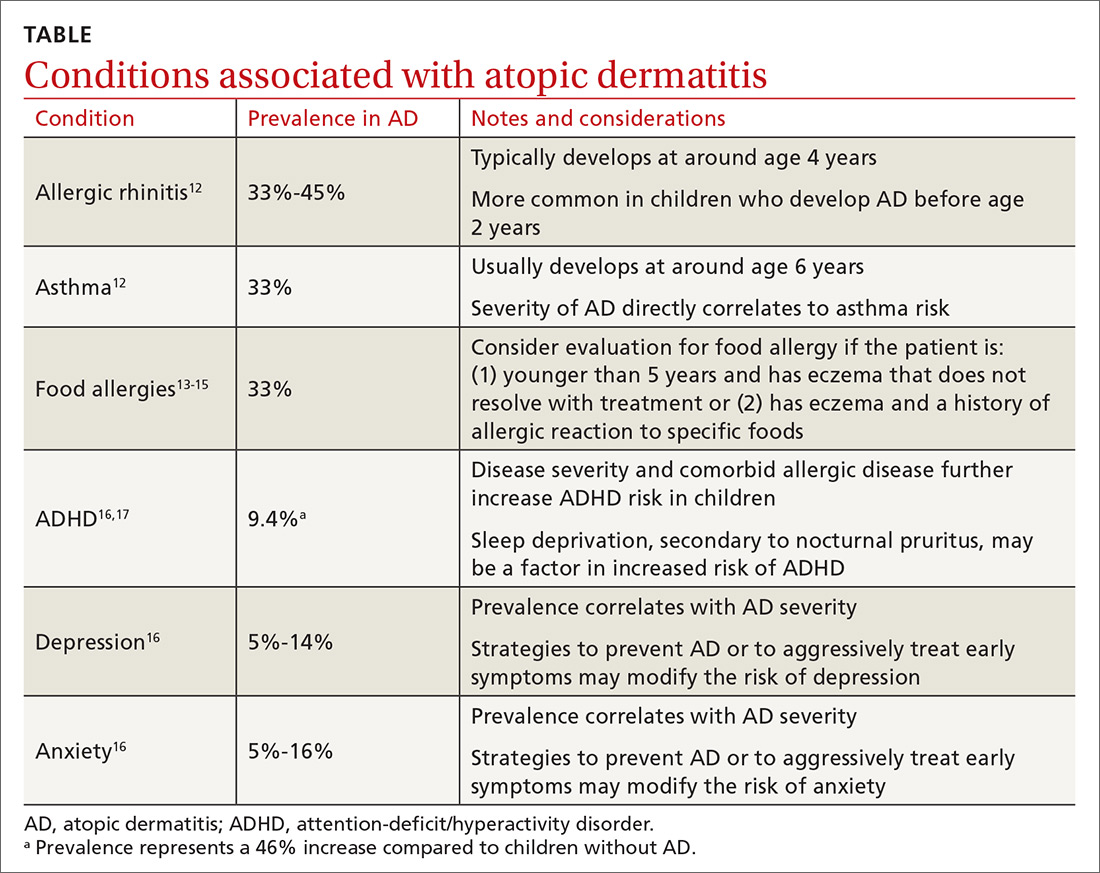
Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.

Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2

Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of

Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
Atopic dermatitis (AD), also known as eczema, is a chronic inflammatory skin condition that is well known for its relapsing, pruritic rash in children and adults. Less recognized are its associated conditions—allergic rhinitis, asthma, food allergies, attention-deficit/hyperactivity disorder (ADHD), depression, and anxiety—and its burden on patients and their families. In fact, families that have children with AD report lower overall quality of life than those with otherwise healthy children.1 Given AD’s prevalence across age groups and its effect on the family, family physicians are uniquely positioned to diagnose, care for, and counsel patients with AD and its associated maladies.
The prevalence and pathogenesis of AD
AD affects up to 20% of children and 5% of adults in the United States.2 AD typically manifests before a child reaches age 5 (often in the first 6 months of life), and it is slightly more common in females (1.3:1). A family history of atopy (eczema, asthma, allergic rhinitis) is common. In fact, children with one atopic parent have a 2- to 3-fold increased risk of atopic dermatitis; those with 2 atopic parents have a 3- to 5-fold increased risk.3
The pathophysiology of AD is complex, culminating in impaired barrier function of the skin and transepidermal water loss resulting in dry and inflamed skin. Additionally, alterations in a cell-mediated immune response leading to an immunoglobulin (Ig) E-mediated hypersensitivity is also theorized to play a role in the development of AD.
Signs and symptoms
Signs at birth. Physical signs of atopic dermatitis typically appear between birth and 6 months. In infancy, lesions generally occur on the scalp, face (FIGURES 1A and 1B), neck, and extensor surfaces of the extremities. Lesions are typically papules and vesicles, sometimes accompanied by serous exudate and crusting. Eczematous lesions typically spare the groin and diaper area, and their presence in this area should raise suspicion for an alternative diagnosis.

Beginning at age 2 years, eczematous lesions are more commonly limited to the folds of the flexor surfaces. Instead of the weeping and crusting lesions seen in infancy, eczema in older children manifests as dry, lichenified papules and plaques in areas that are typically affected in adults: the wrist, hands, ankles, and popliteal and antecubital fossa.2
Although lesions in adults are similar to those of childhood, they may manifest in a more localized area (hand or eyelid, for example). As is the case in childhood, the lesions are dry, sometimes lichenified, and found on the flexural surfaces (FIGURES 2A and 2B).2

Symptom triggers are unproven
While anecdotal reports cite various triggers for AD flares, a systematic review found little scientific evidence to substantiate identifiable triggers.4 Triggers often cited and studied are foods, dust mite exposure, airborne allergens, detergents, sunlight, fabrics, bacterial infections, and stress. While as many as one-third of people with AD who also have confirmed dust mite allergy report worsening of symptoms when exposed to dust, a Cochrane review of 7 randomized controlled trials totaling 324 adults and children with eczema found that efforts at dust mite mitigation (laundering of bed covers, increased vacuuming, spraying for mites) were not effective in reducing symptoms.5
Continue to: How quality of life diminishes with AD
How quality of life diminishes with AD
AD substantially lessens quality of life. For children, the most distressing physical symptoms include itching that inhibits sleep and provokes scratching, pain, and bleeding. Emotional distress can cause irritability, crying, and uncooperativeness with treatments. Parents also report that they frequently restrict their children from activities, such as playing in the heat or swimming, that may lead to worsening of their eczema.6
The loss of sleep associated with AD is not completely understood but is likely multifactorial. Pruritus and scratching leading to sleeplessness is the most obvious culprit, but an altered circadian rhythm, immune system response, and changes in skin physiology are also likely factors.7 Whatever the cause, sleep disturbance is reported in as many as 60% of patients with AD, and the degree of sleep disturbance is proportional to increases in disease severity and worsening of quality-of-life scores.8 Lost sleep is not limited to patients; parents of children with AD also report significant loss of sleep and subsequent decreased work productivity and quality of life.9
Children with AD are often the target of bullying.10 A 2015 survey by the National Eczema Association indicates that 1 in 5 children reported being bullied due to their AD.11
Associated conditions and comorbidities
AD increases patients’ risks for other illnesses, due either to their underlying atopy or to the effects of

Atopic march
Atopic march—the clinical succession of AD, allergic rhinitis, and asthma—is a well-established clinical progression. The presence of all 3 conditions appears to be more common in children diagnosed with AD before 2 years of age.12 Typically, allergic rhinitis manifests at around age 4, and asthma develops between ages 6 and 8. The severity of AD predicts progression. Compared with an 8% chance of asthma developing among the general population, children with mild AD have a 20% to 30% chance of developing asthma, and those with severe AD have about a 70% chance.12
Continue to: Food allergies
Food allergies
Patients with AD are at higher risk for food-induced anaphylaxis, with up to one-third of AD patients having an IgE-mediated food allergy.13 While it is theorized that the impaired skin barrier of an atopic child may allow for early sensitization and allergy development, a landmark 2015 study demonstrated that early allergen introduction (specifically, peanuts) may serve as a preventive strategy in those at high risk of food allergies.14 Current guidelines recommend that physicians be aware of the increased possibility of food allergies in those with AD, and consider evaluating a child for milk, egg, peanut, wheat, and soy allergy if the child is younger than 5 years and has eczema that does not resolve with treatment, or has eczema and a history of an allergic reaction to a specific food.15
Interestingly, despite the strong association between AD and food allergies, it is not clear that food allergies trigger atopic flares; as such, elimination diets are not universally recommended in those without a proven food allergy.
Psychiatric diagnoses
Children with AD have an increased prevalence of several psychiatric conditions, including ADHD, depression, anxiety, conduct disorder, and autism when compared with peers who do not have AD, and the probability correlates with the severity of AD
What we do know is that one of the strongest associations between AD and a psychiatric condition is with ADHD, with a recent pooled meta-analysis showing a 46% increase in risk.17 The incidence of depression among children with AD appears to correlate with the severity of AD symptoms: estimated at 5% with mild AD, 7% with moderate disease, and 14% with severe disease (compared with 3% without AD). Similar incremental increases are seen when correlating AD and anxiety.16
Nonpharmacologic care
Bathing
Bathing habits are critical to controlling AD. While bathing serves to both hydrate the skin and remove allergens, the water’s evaporation off the skin surface can lead to increased transepidermal water loss. Combining bathing and immediate application of a moisturizer improves skin hydration in patients with AD vs bathing alone.18 Thus, consensus guidelines recommend once-daily bathing (bath or shower) to remove scale and crust, followed by immediate application of a moisturizing emollient.19
Continue to: Emollients
Emollients
Application of moisturizing emollients is the mainstay of nonpharmacologic care of AD, and there is strong evidence that their regimented use reduces disease burden and the need for prescription treatment.19 Emollient creams and ointments help retain moisture and improve the skin’s barrier. While ointments may provide a better barrier, patients tend to prefer creams as they are less greasy than ointments.
Emollient therapy may also help prevent development of AD, especially in those infants thought to be at high risk with a family history of atopy. In a multinational randomized controlled trial, infants who received daily full-body application of emollient beginning at 3 weeks of life were significantly less likely than controls to develop AD by 6 months.20 While the mechanism of action is not clearly understood, it is believed that early emollient use prevents skin dehydration and maintains the skin’s barrier integrity, thus decreasing allergen epidermal penetration and subsequent inflammation.
Bleach bath
A bleach bath, prepared by adding 1/2 cup of unconcentrated bleach (5.25% sodium hypochlorite) to a standard 40-gallon bathtub, produces a chlorine mixture equivalent to an average swimming pool. Soaking in a bleach bath for 10 minutes once or twice weekly is thought to reduce inflammation and bacteria on the skin, but studies of its efficacy in improving atopic symptoms are mixed.
In a pooled analysis of 5 studies evaluating bleach baths vs standard baths, there was no significant difference in disease severity at 4 weeks.21 Thus, while bleach baths were effective in decreasing disease severity, they appeared to be no more effective than a standard water bath.21 Bleach baths may be helpful, however, in cases of moderate-to-severe disease with frequent bacterial infections.19
Pharmacologic therapy
Steroids
For symptoms refractory to nonpharmacologic skin care, topical steroids are the initial pharmacologic treatment for AD.19 Choose steroid potency based on symptom severity and disease location. Low- to medium-potency is appropriate for mild disease, and medium- to high-potency is useful for moderate-to-severe symptoms. High-potency steroids are generally avoided on the face and skin folds; however, they can be used for short periods in these areas to induce remission. They must then be quickly tapered and discontinued.
Continue to: Frequency
Frequency. Topical corticosteroids are typically applied twice daily, although recent studies indicate that once-daily application is just as efficacious.22 In addition to treatment of an acute flare, topical steroids are useful as maintenance therapy for patients with recurrent outbreaks in the same anatomical site. Guidelines suggest once- or twice-weekly application of a medium-potency steroid to prolong time between flares.19
For children, a practical guide is for caregivers to apply the amount of steroid covering 1 adult fingertip to an area of the child’s skin equal to that of 2 adult palms.23 Topical steroids are generally well tolerated and have a good safety profile. Adverse effects are proportional to the amount and duration of use and include purpura, telangiectasias, striae, and skin atrophy. The risk of skin atrophy increases with higher potency steroids, occlusion (covering affected area after steroid application), use on thin-skinned areas, and older patient age.24
Reassure patients/parents about the safety of topical steroids, as fears regarding the potential adverse effects can limit compliance. In one study of 200 patients with AD, 72.5% of respondents expressed fear of using steroids on their own skin or that of their child, and 24% admitted being noncompliant with therapy based on these concerns.25
Treating flares. Oral steroids are sometimes needed to abort or control an AD flare in older children and adults. A tapering course of prednisone over 5 to 7 days, transitioning to medium- to high-dose topical steroids, may be needed to achieve symptom control.
Topical calcineurin inhibitors
Topical calcineurin inhibitors, including tacrolimus and pimecrolimus, are generally second-line therapy to topical corticosteroids. However, as nonsteroidal agents, topical calcineurin inhibitors do not cause skin atrophy and can be a first-line option in areas where atrophy is more common (face, eyelids, neck, and skin folds).26
Continue to: A Cochrane review found...
A Cochrane review found tacrolimus 0.1% to be better than low‐potency topical corticosteroids on the face and neck areas, while results were equivocal when compared with moderate‐potency topical corticosteroids on the trunk and extremities (no difference based on physician assessment, but marginal benefit favoring tacrolimus based on participant scoring).27 When compared head-to-head, tacrolimus was more effective than pimecrolimus, although tacrolimus has a higher rate of local irritation. The most common adverse effects are stinging and burning at the application site, although these adverse effects generally improve with repeated application.
There have been long-term safety concerns with topical calcineurin inhibitors—chiefly a 2006 Food and Drug Administration (FDA) black box warning regarding a possible link between topical calcineurin inhibitors and cancer. However, while there may be a slight increased risk of lymphoma in AD patients, a recent meta-analysis did not find an association between topical calcineurin inhibitors use and lymphoma.28 Given the initial concern—and pending additional data—the FDA currently recommends reserving topical calcineurin inhibitors for second-line therapy and only for the minimum amount of time to induce improvement. It also recommends avoiding their use in patients younger than 2 years and in those with compromised immune systems.
Cisaborole
Cisaborole, a topical phosphodiesterase 4 (PDE4) inhibitor, received FDA approval in 2016 for mild-to-moderate AD. By inhibiting PDE4, the drug limits inflammation. In a multicenter randomized trial, patients applying cisaborole 2% twice a day noted reductions in pruritus, inflammation, excoriation, and lichenification.29 Adverse effects are minimal and limited to application site irritation.
Systemic treatments
While beyond the care of a family physician, symptoms refractory to conservative, nonpharmacologic measures and combinations of topical pharmaceuticals can be treated with systemic immunomodulators such as cyclosporine, azathioprine, and methotrexate. Phototherapy is also effective in patients with more widespread skin involvement. Dupilumab, an injectable monoclonal antibody that binds to interleukin-4 receptor and inhibits inflammation, is approved to treat moderate-to-severe AD in adults.30
Ineffective therapies: Oral montelukast and probiotics
While oral antihistamines are frequently prescribed and used, there are no studies evaluating the use of antihistamines (H1) as monotherapy for AD.31 Nonetheless, while not altering the disease process, the sedative effect of antihistamines may palliate the nocturnal pruritus frequently associated with AD. Although nonsedating antihistamines may still have a role for atopic patients with concurrent seasonal and environmental allergies, there is no evidence to support their use in the treatment of AD.
Continue to: Data are limited...
Data are limited on the effectiveness of leukotriene receptor antagonists for AD, and all studies meeting inclusion for a Cochrane review assessed oral montelukast. The review found no benefit with the use of montelukast 10 mg in terms of severity of disease, pruritus, or need for topical steroids.32
A systematic review investigating the benefit of probiotics for the treatment of AD found no improvement in patient-rated eczema scores for quality of life.33 Additionally, a review of 11 randomized controlled trials including 596 participants found no evidence to suggest efficacy of fish oil, zinc, selenium, vitamin D, vitamin E, pyridoxine, sea buckthorn oil, hempseed oil, or sunflower oil in the treatment of AD.34
Education can reduce AD severity
Family physicians can be a source of education and support for patients and families of patients with AD. Support programs for adults with AD—including education, relaxation techniques, and cognitive behavioral therapy—have been shown to decrease disease severity.35 Comparable improvement in disease severity has been demonstrated in children with AD when similar education is provided to them and their families.
CORRESPONDENCE
Franklin Berkey, DO, Penn State Health, 1850 East Park Avenue, Suite 207, State College, PA 16803; fberkey@ pennstatehealth.psu.edu.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
1. Carroll CL, Balkrishnan R, Feldman SR, et al. The burden of atopic dermatitis: impact on the patient, family, and society. Pediatr Dermatol. 2005;22:192-199.
2. Ahn C, Huang W. Clinical presentation of atopic dermatitis. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:38-46.
3. Eichenfield LF, Tom WL, Chamblin SL, et al. Guidelines of care for the management of atopic dermatitis. Part 1: diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338-351.
4. Langan SM, Williams HC. What causes worsening of eczema? A systematic review. Br J Dermatol. 2006;155:504-514.
5. Nankervis H, Pynn EV, Boyle RJ, et al. House dust mite reduction and avoidance measures for treating eczema. Cochrane Database Syst Rev. 2015:CD008426.
6. Chamlin SL, Frieden IJ, Williams ML, et al. Effects of atopic dermatitis on young American children and their families. Pediatrics. 2004;114:607-611.
7. Chang Y-S, Chiang B-L. Mechanism of sleep disturbance in children with atopic dermatitis and the role of the circadian rhythm and melatonin. Int J Mol Sci. 2016;17:462.
8. Camfferman D, Kennedy JD, Gold M, et al. Eczema and sleep and its relationship to daytime functioning in children. Sleep Med Rev. 2010;14:359-369.
9. Chamlin SL, Mattson CL, Frieden IJ, et al. The price of pruritus: sleep disturbance and cosleeping in atopic dermatitis. Arch Pediatr Adolesc Med. 2005;159:745-750.
10. Drucker AM, Wang AR, Li W-Q, et al. The burden of atopic dermatitis: summary of a report for the National Eczema Association. J Invest Dermatol. 2017;137:P26-P30.
11. National Eczema Association. Tools for school: addressing school bullying for kids with eczema. Accessed January 5, 2021. https://nationaleczema.org/children-with-eczema-experience-bullying/
12. Bantz SK, Zhu Z, Zhen T. The atopic march: progression from atopic dermatitis to allergic rhinitis and asthma. J Clin Cell Immunol. 2014;5:202
13. Laird M, Sicco KL. Defining and measuring the scope of atopic dermatitis. Adv Exp Med Biol. 2017;1027:93-104.
14. Du Toit G, Roberts G, Sayre PH, et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N Engl J Med. 2015;372:803-813.
15. Boyce JA, Assa’ad A, Burks AW, et al. Guidelines for the diagnosis and management of food allergy in the United States: report of the NIAID-sponsored expert panel. J Allergy Clin Immunol. 2010;126:S1–S58.
16. Yaghmaie P, Koudelka CW, Simpson EL. Mental health comorbidity in patients with atopic dermatitis. J Allergy Clin Immunol. 2013;131:428-433.
17. Strom MA, Fishbein AB, Paller AS, et al. Association between atopic dermatitis and attention deficit hyperactivity disorder in U.S. children and adults. Br J Dermatol. 2016;175:920-929.
18. Chiang C, Eichenfield LF. Quantitative assessment of combination bathing and moisturizing regimens on skin hydration in atopic dermatitis. Pediatr Dermatol. 2009;26:273-278.
19. Eichenfield LF, Tom WL, Berger TG, et al. Guidelines of care for the management of atopic dermatitis: section 2. Management and treatment of atopic dermatitis with topical therapies. J Am Acad Dermatol. 2014;71:116-132.
20. Simpson EL, Chalmers JR, Hanifin JM, et al. Emollient enhancement of the skin barrier from birth offers effective atopic dermatitis prevention. J Allergy Clin Immunol. 2014;134:818-823.
21. Chopra R, Vakharia PP, Sacotte R, et al. Efficacy of bleach baths in reducing severity of atopic dermatitis: a systematic review and meta-analysis. Ann Allergy Asthma Immunol. 2017;119:435-440.
22. Williams HC. Established corticosteroid creams should be applied only once daily in patients with atopic eczema. BMJ. 2007;334:1272.
23. Long CC, Mills CM, Finlay AY. A practical guide to topical therapy in children. Br J Dermatol. 1998;138:293-296.
24. Callen J, Chamlin S, Eichenfield LF, et al. A systematic review of the safety of topical therapies for atopic dermatitis. Br J Dermatol. 2007;156:203-221.
25. Charman CR, Morris AD, Williams HC. Topical corticosteroid phobia in patients with atopic eczema. Br J Dermatol. 2000;142:931-936.
26. Ashcroft DM, Dimmock P, Garside R, et al. Efficacy and tolerability of topical pimecrolimus and tacrolimus in the treatment of atopic dermatitis: a meta-analysis of randomised controlled trials. BMJ. 2005;330:516.
27. Cury Martins J, Martins C, Aoki V, et al. Topical tacrolimus for atopic dermatitis. Cochrane Database Syst Rev. 2015:CD009864.
28. Legendre L, Barnetche T, Mazereeuw-Hautier J, et al. Risk of lymphoma in patients with atopic dermatitis and the role of topical treatment: a systematic review and meta-analysis. J Am Acad Dermatol. 2015;72:992-1002.
29. Paller AS, Tom WL, Lebwohl MG, et al. Efficacy and safety of crisaborole ointment, a novel, nonsteroidal phosphodiesterase 4 (PDE4) inhibitor for the topical treatment of atopic dermatitis (AD) in children and adults. J Am Acad Dermatol. 2016;75:494-503.
30. Dupilumab [package insert]. Tarrytown, NY: Regeneron Pharmaceuticals Inc; 2017.
31. van Zuuren EJ, Apfelbacher CJ, Fedorowicz Z, et al. No high level evidence to support the use of oral H1 antihistamines as monotherapy for eczema: a summary of a Cochrane systematic review. Syst Rev. 2014;3:25.
32. Ferguson L, Futamura M, Vakirlis E, et al. Leukotriene receptor antagonists for eczema. Cochrane Database Syst Rev. 2018:CD011224.
33. Makrgeorgou A, Leonardi-Bee J, Bath-Hextall FJ, et al. Probiotics for treating eczema. Cochrane Database Syst Rev. 2018:CD006135.
34. Bath-Hextall FJ, Jenkinson C, Humphreys R, et al. Dietary supplements for established atopic eczema. Cochrane Database Syst Rev. 2012:CD005205.
35. Sy W, Lamb AJ. Atopic dermatitis disease education. In: Fortson E, Feldman SR, Stroud LC, eds. Management of Atopic Dermatitis: Methods and Challenges. Springer International Publishing; 2017:179-184.
PRACTICE RECOMMENDATIONS
› Advise patients to regularly apply moisturizers, which reduces atopic dermatitis (AD) severity and may avert the need for pharmacologic intervention. A
› Assure patients that a topical corticosteroid is safe and effective as first-line treatment for AD symptoms refractory to nonpharmacologic recommendations. A
› Consider topical calcineurin inhibitors for both acute and chronic AD in adults and children, especially in areas more prone to topical corticosteroid adverse effects. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Peripartum maternal oxygen supplementation shows little benefit
Peripartum maternal oxygen supplementation does not yield a clinically relevant improvement in umbilical artery gas pH or other neonatal outcomes, reported Nandini Raghuraman, MD, MS, of the Washington University School of Medicine, St. Louis, MO, and her associates.
In a meta-analysis of 16 studies identified between Feb. 18 and April 3, 2020, the investigators sought to determine whether maternal oxygen supplementation during delivery leads to improved measures in umbilical artery (UA) gas and neonatal outcomes. Using data from randomized clinical trials, they compared peripartum oxygen supplementation with room air and examined the link between oxygen delivery during regular labor or planned cesarean delivery (CD) with UA gas measures and other neonatal outcomes.
Altogether, 1,078 patients were randomized to the oxygen group or the room air group. UA pH remained similar between the two groups even after the researchers factored in risk of bias, use of low-flow devices, or FIO2 below 60%, noted the authors. Oxygen supplementation also appeared to reduce rates of UA pH that were less than 7.2 and increase UA PaO2 relative to room air during scheduled cesarean deliveries, they added.
Considerable interstudy heterogeneity was found
Although marginally lower one-minute Apgar scores were observed in infants whose mothers received oxygen during cesarean delivery, the mean difference between oxygen and room air was less than a point and there were no other statistically significant differences in any secondary outcomes, the authors said. Considerable interstudy heterogeneity was noted across most of the study outcomes.
It is important to note that results pooled from all the studies reviewed indicated an increase in UA PaO2 but no notable differences in UA pH when oxygen was used. Citing multiple studies included in the review, the authors observed that UA PaO2 is a “poor estimator of neonatal morbidity” because, when evaluated in cord blood gas, it represents dissolved oxygen and is not an accurate indication of how much oxygen is bound to hemoglobin. For this reason, dissolved oxygen content by itself is not an indication of hypoxia or subpar tissue oxygenation.
“Prolonged tissue hypoxia leads to anaerobic metabolism, resulting in decreased pH, which is why UA pH ultimately serves as a better marker for prediction of neonatal morbidity. An intervention that increases the PaO2 without concomitantly increasing the pH has limited clinical benefit, particularly because hyperoxemia is associated with production of free radicals and oxidative cell damage in adults and neonates,” they explained.
With unproven benefits and potential for risk of harm, prolonged oxygen use should be limited
“A large, adequately powered trial is needed to investigate the effect of maternal oxygen supplementation in response to fetal heart rate tracings on short- and long-term neonatal morbidity,” the authors suggested. For the time being, they cautioned limiting prolonged oxygen use since the benefits are unproven and there is a potential risk of harm.
In a separate interview, Iris Krishna, MD, MPH, FACOG, Emory University, Atlanta, noted, “The use of maternal supplemental oxygen with the intent of improving fetal oxygenation is a common clinical practice. Previous studies on maternal oxygen supplementation during labor have yielded conflicting results; however, there is growing literature suggesting that maternal intrapartum supplemental oxygenation may not provide clinically significant benefit and there may even be potential harm to mother and baby.
“Unique to this meta-analysis is evaluation of maternal oxygen supplementation in the presence or absence of labor, hypothesizing that placental oxygen transfer may be affected by regular uterine contractions. The pooled results suggest that the use of maternal supplemental oxygenation does not result in clinically relevant fetal oxygenation in the presence or absence of labor when compared to room air. A limitation of this meta-analysis is that the use of oxygen in response to nonreassuring fetal tracing was not assessed, the most common clinical indication for maternal oxygen supplementation.
“This study further challenges the practice of maternal intrapartum supplemental oxygen and highlights that we have much to learn about the impact of this practice. More research is needed to assess optimal duration of oxygen supplementation, safety and efficacy of oxygen supplementation, appropriate clinical indications for oxygen supplementation, as well as the long-term neonatal outcomes of in utero hyperoxygenation.“
Dr. Raghuraman reported receiving multiple grants and acknowledged multiple funding sources. Her colleagues and Dr. Krishna had no conflicts of interest to report.
Peripartum maternal oxygen supplementation does not yield a clinically relevant improvement in umbilical artery gas pH or other neonatal outcomes, reported Nandini Raghuraman, MD, MS, of the Washington University School of Medicine, St. Louis, MO, and her associates.
In a meta-analysis of 16 studies identified between Feb. 18 and April 3, 2020, the investigators sought to determine whether maternal oxygen supplementation during delivery leads to improved measures in umbilical artery (UA) gas and neonatal outcomes. Using data from randomized clinical trials, they compared peripartum oxygen supplementation with room air and examined the link between oxygen delivery during regular labor or planned cesarean delivery (CD) with UA gas measures and other neonatal outcomes.
Altogether, 1,078 patients were randomized to the oxygen group or the room air group. UA pH remained similar between the two groups even after the researchers factored in risk of bias, use of low-flow devices, or FIO2 below 60%, noted the authors. Oxygen supplementation also appeared to reduce rates of UA pH that were less than 7.2 and increase UA PaO2 relative to room air during scheduled cesarean deliveries, they added.
Considerable interstudy heterogeneity was found
Although marginally lower one-minute Apgar scores were observed in infants whose mothers received oxygen during cesarean delivery, the mean difference between oxygen and room air was less than a point and there were no other statistically significant differences in any secondary outcomes, the authors said. Considerable interstudy heterogeneity was noted across most of the study outcomes.
It is important to note that results pooled from all the studies reviewed indicated an increase in UA PaO2 but no notable differences in UA pH when oxygen was used. Citing multiple studies included in the review, the authors observed that UA PaO2 is a “poor estimator of neonatal morbidity” because, when evaluated in cord blood gas, it represents dissolved oxygen and is not an accurate indication of how much oxygen is bound to hemoglobin. For this reason, dissolved oxygen content by itself is not an indication of hypoxia or subpar tissue oxygenation.
“Prolonged tissue hypoxia leads to anaerobic metabolism, resulting in decreased pH, which is why UA pH ultimately serves as a better marker for prediction of neonatal morbidity. An intervention that increases the PaO2 without concomitantly increasing the pH has limited clinical benefit, particularly because hyperoxemia is associated with production of free radicals and oxidative cell damage in adults and neonates,” they explained.
With unproven benefits and potential for risk of harm, prolonged oxygen use should be limited
“A large, adequately powered trial is needed to investigate the effect of maternal oxygen supplementation in response to fetal heart rate tracings on short- and long-term neonatal morbidity,” the authors suggested. For the time being, they cautioned limiting prolonged oxygen use since the benefits are unproven and there is a potential risk of harm.
In a separate interview, Iris Krishna, MD, MPH, FACOG, Emory University, Atlanta, noted, “The use of maternal supplemental oxygen with the intent of improving fetal oxygenation is a common clinical practice. Previous studies on maternal oxygen supplementation during labor have yielded conflicting results; however, there is growing literature suggesting that maternal intrapartum supplemental oxygenation may not provide clinically significant benefit and there may even be potential harm to mother and baby.
“Unique to this meta-analysis is evaluation of maternal oxygen supplementation in the presence or absence of labor, hypothesizing that placental oxygen transfer may be affected by regular uterine contractions. The pooled results suggest that the use of maternal supplemental oxygenation does not result in clinically relevant fetal oxygenation in the presence or absence of labor when compared to room air. A limitation of this meta-analysis is that the use of oxygen in response to nonreassuring fetal tracing was not assessed, the most common clinical indication for maternal oxygen supplementation.
“This study further challenges the practice of maternal intrapartum supplemental oxygen and highlights that we have much to learn about the impact of this practice. More research is needed to assess optimal duration of oxygen supplementation, safety and efficacy of oxygen supplementation, appropriate clinical indications for oxygen supplementation, as well as the long-term neonatal outcomes of in utero hyperoxygenation.“
Dr. Raghuraman reported receiving multiple grants and acknowledged multiple funding sources. Her colleagues and Dr. Krishna had no conflicts of interest to report.
Peripartum maternal oxygen supplementation does not yield a clinically relevant improvement in umbilical artery gas pH or other neonatal outcomes, reported Nandini Raghuraman, MD, MS, of the Washington University School of Medicine, St. Louis, MO, and her associates.
In a meta-analysis of 16 studies identified between Feb. 18 and April 3, 2020, the investigators sought to determine whether maternal oxygen supplementation during delivery leads to improved measures in umbilical artery (UA) gas and neonatal outcomes. Using data from randomized clinical trials, they compared peripartum oxygen supplementation with room air and examined the link between oxygen delivery during regular labor or planned cesarean delivery (CD) with UA gas measures and other neonatal outcomes.
Altogether, 1,078 patients were randomized to the oxygen group or the room air group. UA pH remained similar between the two groups even after the researchers factored in risk of bias, use of low-flow devices, or FIO2 below 60%, noted the authors. Oxygen supplementation also appeared to reduce rates of UA pH that were less than 7.2 and increase UA PaO2 relative to room air during scheduled cesarean deliveries, they added.
Considerable interstudy heterogeneity was found
Although marginally lower one-minute Apgar scores were observed in infants whose mothers received oxygen during cesarean delivery, the mean difference between oxygen and room air was less than a point and there were no other statistically significant differences in any secondary outcomes, the authors said. Considerable interstudy heterogeneity was noted across most of the study outcomes.
It is important to note that results pooled from all the studies reviewed indicated an increase in UA PaO2 but no notable differences in UA pH when oxygen was used. Citing multiple studies included in the review, the authors observed that UA PaO2 is a “poor estimator of neonatal morbidity” because, when evaluated in cord blood gas, it represents dissolved oxygen and is not an accurate indication of how much oxygen is bound to hemoglobin. For this reason, dissolved oxygen content by itself is not an indication of hypoxia or subpar tissue oxygenation.
“Prolonged tissue hypoxia leads to anaerobic metabolism, resulting in decreased pH, which is why UA pH ultimately serves as a better marker for prediction of neonatal morbidity. An intervention that increases the PaO2 without concomitantly increasing the pH has limited clinical benefit, particularly because hyperoxemia is associated with production of free radicals and oxidative cell damage in adults and neonates,” they explained.
With unproven benefits and potential for risk of harm, prolonged oxygen use should be limited
“A large, adequately powered trial is needed to investigate the effect of maternal oxygen supplementation in response to fetal heart rate tracings on short- and long-term neonatal morbidity,” the authors suggested. For the time being, they cautioned limiting prolonged oxygen use since the benefits are unproven and there is a potential risk of harm.
In a separate interview, Iris Krishna, MD, MPH, FACOG, Emory University, Atlanta, noted, “The use of maternal supplemental oxygen with the intent of improving fetal oxygenation is a common clinical practice. Previous studies on maternal oxygen supplementation during labor have yielded conflicting results; however, there is growing literature suggesting that maternal intrapartum supplemental oxygenation may not provide clinically significant benefit and there may even be potential harm to mother and baby.
“Unique to this meta-analysis is evaluation of maternal oxygen supplementation in the presence or absence of labor, hypothesizing that placental oxygen transfer may be affected by regular uterine contractions. The pooled results suggest that the use of maternal supplemental oxygenation does not result in clinically relevant fetal oxygenation in the presence or absence of labor when compared to room air. A limitation of this meta-analysis is that the use of oxygen in response to nonreassuring fetal tracing was not assessed, the most common clinical indication for maternal oxygen supplementation.
“This study further challenges the practice of maternal intrapartum supplemental oxygen and highlights that we have much to learn about the impact of this practice. More research is needed to assess optimal duration of oxygen supplementation, safety and efficacy of oxygen supplementation, appropriate clinical indications for oxygen supplementation, as well as the long-term neonatal outcomes of in utero hyperoxygenation.“
Dr. Raghuraman reported receiving multiple grants and acknowledged multiple funding sources. Her colleagues and Dr. Krishna had no conflicts of interest to report.
FROM JAMA PEDIATRICS
How COVID-19 will continue to alter patient visits
Finding the current domestic and global situations too disheartening to write about, I have decided for the moment to take the long view in hopes of finding something to stimulate your imaginations. It appears that we have several vaccines effective against SARS-CoV-2 if not in your hands at the moment at least in someone’s freezer or at the very least somewhere near beginning of their journey in the production pipeline. It may be a year of more but thanks to the vaccines and herd immunity there will be a time when parents may feel more comfortable about bringing their children into your office. How are you going to dial back your office routine to something even vaguely familiar?
To keep your office afloat financially you have probably been forced to adopt and adapt telemedicine strategies to your practice style. Prior to the pandemic you may have been among the few who were actively experimenting with practicing remotely. But, it is more likely that you had given little serious thought to how you would manage your patients without them being physically present.
You probably carried in your mind a list of symptoms and complaints which you had promised yourself that you would never treat without first laying eyes and hands on the patient. You may have even codified this list into a set of guidelines that you included in the office manual for your nurses, assistants, and receptionists. You may have looked askance at some of your colleagues whom you felt too often treated their patients (and yours when they were covering) based on what seemed to be scanty information gleaned from a phone call. The impropriety of this kind of clinical behavior may have even come up at staff meetings or at least been the topic of hallway discussions.
How did your list of complaints that demanded an in-person visit evolve? I suspect that in large part it was formed as you modeled the behavior of your mentors and teachers. In some cases you may have heard of tragic cases in which a child had died or suffered serious consequences of being treated without an in-person evaluation. In many cases you were following a tradition or ethic that said treating in certain circumstances without an exam just wasn’t done.
Have the realities of the pandemic forced you to alter your list of must-see-before-I’ll-treat complaints? Have you found yourself calling in antibiotic prescriptions for children with ear pain who 1 year ago you would have told to come in for an office visit? Are you treating “strep throats” without a rapid strep test or culture? How many stimulant prescriptions have you refilled for children who haven’t been reevaluated in the office in over a year? How are you going to manage the tsunami of requests for sports physicals once the junior high and high school teams are allowed to return to action? You probably won’t have the time to examine all of the sports candidates who show up in your office with crumpled forms recently retrieved from crumb-filled backpacks.
Where are you going to reset the bar as the pandemic lifts and the barriers that have prevented patients from coming to your office over the last year or year and a half recede? Have you realized that many of your office visits in prepandemic times were unnecessary? How many children with otitis really needed to be followed up with an ear recheck visit? Which children with sore throats and a fever needed to be examined? Was a yearly exam really necessary for a high school sophomore who wanted to play basketball? Has your comfort zone widened to include more patient complaints that can be managed without a face to face encounter? Where will telemedicine fit into the mix?
At some time in the next 12 months you will have to recalibrate and reset the bar. It will probably be a gradual process that in large part can be molded by the responses of the families who may have also come to realize that seeing you in the office isn’t quite as necessary as you both may have thought it was.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected]
Finding the current domestic and global situations too disheartening to write about, I have decided for the moment to take the long view in hopes of finding something to stimulate your imaginations. It appears that we have several vaccines effective against SARS-CoV-2 if not in your hands at the moment at least in someone’s freezer or at the very least somewhere near beginning of their journey in the production pipeline. It may be a year of more but thanks to the vaccines and herd immunity there will be a time when parents may feel more comfortable about bringing their children into your office. How are you going to dial back your office routine to something even vaguely familiar?
To keep your office afloat financially you have probably been forced to adopt and adapt telemedicine strategies to your practice style. Prior to the pandemic you may have been among the few who were actively experimenting with practicing remotely. But, it is more likely that you had given little serious thought to how you would manage your patients without them being physically present.
You probably carried in your mind a list of symptoms and complaints which you had promised yourself that you would never treat without first laying eyes and hands on the patient. You may have even codified this list into a set of guidelines that you included in the office manual for your nurses, assistants, and receptionists. You may have looked askance at some of your colleagues whom you felt too often treated their patients (and yours when they were covering) based on what seemed to be scanty information gleaned from a phone call. The impropriety of this kind of clinical behavior may have even come up at staff meetings or at least been the topic of hallway discussions.
How did your list of complaints that demanded an in-person visit evolve? I suspect that in large part it was formed as you modeled the behavior of your mentors and teachers. In some cases you may have heard of tragic cases in which a child had died or suffered serious consequences of being treated without an in-person evaluation. In many cases you were following a tradition or ethic that said treating in certain circumstances without an exam just wasn’t done.
Have the realities of the pandemic forced you to alter your list of must-see-before-I’ll-treat complaints? Have you found yourself calling in antibiotic prescriptions for children with ear pain who 1 year ago you would have told to come in for an office visit? Are you treating “strep throats” without a rapid strep test or culture? How many stimulant prescriptions have you refilled for children who haven’t been reevaluated in the office in over a year? How are you going to manage the tsunami of requests for sports physicals once the junior high and high school teams are allowed to return to action? You probably won’t have the time to examine all of the sports candidates who show up in your office with crumpled forms recently retrieved from crumb-filled backpacks.
Where are you going to reset the bar as the pandemic lifts and the barriers that have prevented patients from coming to your office over the last year or year and a half recede? Have you realized that many of your office visits in prepandemic times were unnecessary? How many children with otitis really needed to be followed up with an ear recheck visit? Which children with sore throats and a fever needed to be examined? Was a yearly exam really necessary for a high school sophomore who wanted to play basketball? Has your comfort zone widened to include more patient complaints that can be managed without a face to face encounter? Where will telemedicine fit into the mix?
At some time in the next 12 months you will have to recalibrate and reset the bar. It will probably be a gradual process that in large part can be molded by the responses of the families who may have also come to realize that seeing you in the office isn’t quite as necessary as you both may have thought it was.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected]
Finding the current domestic and global situations too disheartening to write about, I have decided for the moment to take the long view in hopes of finding something to stimulate your imaginations. It appears that we have several vaccines effective against SARS-CoV-2 if not in your hands at the moment at least in someone’s freezer or at the very least somewhere near beginning of their journey in the production pipeline. It may be a year of more but thanks to the vaccines and herd immunity there will be a time when parents may feel more comfortable about bringing their children into your office. How are you going to dial back your office routine to something even vaguely familiar?
To keep your office afloat financially you have probably been forced to adopt and adapt telemedicine strategies to your practice style. Prior to the pandemic you may have been among the few who were actively experimenting with practicing remotely. But, it is more likely that you had given little serious thought to how you would manage your patients without them being physically present.
You probably carried in your mind a list of symptoms and complaints which you had promised yourself that you would never treat without first laying eyes and hands on the patient. You may have even codified this list into a set of guidelines that you included in the office manual for your nurses, assistants, and receptionists. You may have looked askance at some of your colleagues whom you felt too often treated their patients (and yours when they were covering) based on what seemed to be scanty information gleaned from a phone call. The impropriety of this kind of clinical behavior may have even come up at staff meetings or at least been the topic of hallway discussions.
How did your list of complaints that demanded an in-person visit evolve? I suspect that in large part it was formed as you modeled the behavior of your mentors and teachers. In some cases you may have heard of tragic cases in which a child had died or suffered serious consequences of being treated without an in-person evaluation. In many cases you were following a tradition or ethic that said treating in certain circumstances without an exam just wasn’t done.
Have the realities of the pandemic forced you to alter your list of must-see-before-I’ll-treat complaints? Have you found yourself calling in antibiotic prescriptions for children with ear pain who 1 year ago you would have told to come in for an office visit? Are you treating “strep throats” without a rapid strep test or culture? How many stimulant prescriptions have you refilled for children who haven’t been reevaluated in the office in over a year? How are you going to manage the tsunami of requests for sports physicals once the junior high and high school teams are allowed to return to action? You probably won’t have the time to examine all of the sports candidates who show up in your office with crumpled forms recently retrieved from crumb-filled backpacks.
Where are you going to reset the bar as the pandemic lifts and the barriers that have prevented patients from coming to your office over the last year or year and a half recede? Have you realized that many of your office visits in prepandemic times were unnecessary? How many children with otitis really needed to be followed up with an ear recheck visit? Which children with sore throats and a fever needed to be examined? Was a yearly exam really necessary for a high school sophomore who wanted to play basketball? Has your comfort zone widened to include more patient complaints that can be managed without a face to face encounter? Where will telemedicine fit into the mix?
At some time in the next 12 months you will have to recalibrate and reset the bar. It will probably be a gradual process that in large part can be molded by the responses of the families who may have also come to realize that seeing you in the office isn’t quite as necessary as you both may have thought it was.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “How to Say No to Your Toddler.” Other than a Littman stethoscope he accepted as a first-year medical student in 1966, Dr. Wilkoff reports having nothing to disclose. Email him at [email protected]
Early Head Start program boosts healthy eating, self-regulation
Home-based preventive interventions not only improve healthy eating habits and self-regulation in toddlers but also guide their parents toward better food presentation and response to picky behaviors, reported Robert L. Nix, PhD, of the University of Wisconsin, Madison, and his associates.
In a small, randomized controlled trial of 73 families with toddlers aged 18-36 months enrolled in home-based Early Head Start (EHS), the researchers evaluated four protective factors, including toddlers’ healthy eating habits, toddlers’ self-regulation, parents’ responsive feeding practices, and parents’ sensitive scaffolding. The study, conducted from April to October 2013, is the first clinical trial of Recipe 4 Success, a preschool-focused intervention created by administrators and home visitors of EHS that promotes healthy eating and self-regulation in toddlers living in poverty who may otherwise face weight challenges and obesity later in life. Integrating the intervention into EHS allowed the researchers to take full advantage of its national infrastructure and to make dissemination more efficient.
Of the families selected to participate, all of whom were living below the Federal poverty threshold, 66 were retained through post treatment. Most participating parents were biological mothers; 61% were single; 29% were not high school graduates; just 11% were employed full time. The toddlers averaged 30.72 months; 44% were female. Roughly 48% of families were non-Hispanic White; 29% were Black; and 23% were Hispanic or Latinx, the investigators reported in Pediatrics. More than three-quarters of participants were enrolled in the Special Supplemental Nutrition Program for Women, Infants, and Children or the Supplemental Nutrition Assistance Program.
The program allowed parents to transform toddler eating habits quickly
The study, which was designed to evaluate for posttreatment differences in the primary outcomes, involved 10 consecutive weekly lessons implemented by regularly assigned EHS home visitors. Parents were required to adhere to feeding practices carefully targeted with sensitive, structured scaffolding designed to keep toddlers from becoming overwhelmed. Parents were guided to understand, for instance, that toddlers frequently need to be exposed to a new food 10-20 times before eating it, and that poor sleep can have a detrimental influence on emotional and behavioral controls that can progress to weight gain.
Parent recall of what food and drinks their toddlers consumed in the previous 24-hour period was collected by interviewers. The percentage of all meals that included a fruit and/or vegetable, a protein source, and the absence of sweets and junk food were noted. Toddler self-regulation was assessed in accordance with delay of gratification, task orientation, and emotional/behavioral control. Parents were asked to rate toddler ability to cease desired activities to comply with parental requests. Parental responsive feeding practices were also recorded to observe how they introduce unique healthy foods and how they responded to their toddlers’ reactions. Parental sensitive scaffolding was similarly observed for their ability to structure activities in a developmentally appropriate manner promoting self-regulation.
The researchers noted no statistically significant differences between families in the treatment and control groups, nor were there differences in outcome measures or covariates. Study findings showed that, compared with toddlers who continued to receive just EHS support, the toddlers randomly assigned to Recipe 4 Success were more likely to consume snacks and meals that contained fruits, vegetables, protein, and no sweets or junk food.
As the results of this study and others have shown, early food preferences offer the strongest indication of later diet and healthy eating habits throughout life. The program targeted in this study is significant in its ability to accelerate the adoption of better toddler eating habits in just a 10-week period.
Recipe 4 Success along with other successful preventive interventions for young children are most effective when parents drive the change. “In the present trial, the quality of parenting was most highly related to healthy eating habits and self-regulation at baseline,” the researchers noted.
Specifically, the authors attributed the success of the program to “targeting specific interrelated outcomes with an integrated, theoretically driven intervention model,” which allowed Recipe 4 Success to boost the effectiveness of EHS substantially “in just 10 weeks with a minimal increase in funding,” the authors added.
The authors noted several weaknesses as well as strengths of the study. Its primary weakness was a baseline-posttreatment design, which made it impossible to assert that intervention effects can be sustained. The study was also limited to English-speaking families. Given that most home visitors attended to families in both Recipe 4 Success and EHS, the researchers noted the possibility for contamination across conditions, but they added that this would have actually reduced the intervention effects. The study’s primary strength was the evidenced-based nature of the randomized control. That Recipe 4 Success was operated as an intervention only strengthen the benefits of normal EHS visits.
Patient parents who promote self-regulation have the best chance of success
“This small study emphasizes the importance of parent education and support in setting the toddlers’ palate for lifelong eating habits and self-regulation,” observed Silver Spring, MD, private practice pediatrician and associate clinical professor of pediatrics at George Washington University, Washington, Lillian M. Beard, MD, in a separate interview.
“With the goal of promoting eating habits and self-regulation, I try to guide parents’ choices of what they offer to their toddler. I applaud parents’ patience as I encourage them not to give in and quickly resort to offering salty or sweet snacks. I suggest that if during the course of a day, a palette of colorful healthy choices is offered, most toddlers will graze independently as they go about their play. The challenge is to really support the parent through this quirky stage of their child’s development,” she explained.
“The ultimate challenge today with so much food insecurity, COVID-19 related job losses, and shrinking dollars to feed families is that too many families are feeling a food crisis! A program such as Recipe 4 Success can provide invaluable education for families on how to best stretch their few dollars, with knowledge of which items to seek from their community food pantries, how to best utilize items from the State WIC programs and still seek nutrition tips from their pediatricians while avoiding expensive fast foods that only offer immediate satiety and gratification. The Recipe 4 Success educator, pediatrician, or any community educator can give recommendations about which fresh produce may be inexpensive, but nutritional,” Dr. Beard suggested.
Dr. Nix and colleagues as well as Dr. Beard had no conflicts of interest and no relevant financial disclosures.
Home-based preventive interventions not only improve healthy eating habits and self-regulation in toddlers but also guide their parents toward better food presentation and response to picky behaviors, reported Robert L. Nix, PhD, of the University of Wisconsin, Madison, and his associates.
In a small, randomized controlled trial of 73 families with toddlers aged 18-36 months enrolled in home-based Early Head Start (EHS), the researchers evaluated four protective factors, including toddlers’ healthy eating habits, toddlers’ self-regulation, parents’ responsive feeding practices, and parents’ sensitive scaffolding. The study, conducted from April to October 2013, is the first clinical trial of Recipe 4 Success, a preschool-focused intervention created by administrators and home visitors of EHS that promotes healthy eating and self-regulation in toddlers living in poverty who may otherwise face weight challenges and obesity later in life. Integrating the intervention into EHS allowed the researchers to take full advantage of its national infrastructure and to make dissemination more efficient.
Of the families selected to participate, all of whom were living below the Federal poverty threshold, 66 were retained through post treatment. Most participating parents were biological mothers; 61% were single; 29% were not high school graduates; just 11% were employed full time. The toddlers averaged 30.72 months; 44% were female. Roughly 48% of families were non-Hispanic White; 29% were Black; and 23% were Hispanic or Latinx, the investigators reported in Pediatrics. More than three-quarters of participants were enrolled in the Special Supplemental Nutrition Program for Women, Infants, and Children or the Supplemental Nutrition Assistance Program.
The program allowed parents to transform toddler eating habits quickly
The study, which was designed to evaluate for posttreatment differences in the primary outcomes, involved 10 consecutive weekly lessons implemented by regularly assigned EHS home visitors. Parents were required to adhere to feeding practices carefully targeted with sensitive, structured scaffolding designed to keep toddlers from becoming overwhelmed. Parents were guided to understand, for instance, that toddlers frequently need to be exposed to a new food 10-20 times before eating it, and that poor sleep can have a detrimental influence on emotional and behavioral controls that can progress to weight gain.
Parent recall of what food and drinks their toddlers consumed in the previous 24-hour period was collected by interviewers. The percentage of all meals that included a fruit and/or vegetable, a protein source, and the absence of sweets and junk food were noted. Toddler self-regulation was assessed in accordance with delay of gratification, task orientation, and emotional/behavioral control. Parents were asked to rate toddler ability to cease desired activities to comply with parental requests. Parental responsive feeding practices were also recorded to observe how they introduce unique healthy foods and how they responded to their toddlers’ reactions. Parental sensitive scaffolding was similarly observed for their ability to structure activities in a developmentally appropriate manner promoting self-regulation.
The researchers noted no statistically significant differences between families in the treatment and control groups, nor were there differences in outcome measures or covariates. Study findings showed that, compared with toddlers who continued to receive just EHS support, the toddlers randomly assigned to Recipe 4 Success were more likely to consume snacks and meals that contained fruits, vegetables, protein, and no sweets or junk food.
As the results of this study and others have shown, early food preferences offer the strongest indication of later diet and healthy eating habits throughout life. The program targeted in this study is significant in its ability to accelerate the adoption of better toddler eating habits in just a 10-week period.
Recipe 4 Success along with other successful preventive interventions for young children are most effective when parents drive the change. “In the present trial, the quality of parenting was most highly related to healthy eating habits and self-regulation at baseline,” the researchers noted.
Specifically, the authors attributed the success of the program to “targeting specific interrelated outcomes with an integrated, theoretically driven intervention model,” which allowed Recipe 4 Success to boost the effectiveness of EHS substantially “in just 10 weeks with a minimal increase in funding,” the authors added.
The authors noted several weaknesses as well as strengths of the study. Its primary weakness was a baseline-posttreatment design, which made it impossible to assert that intervention effects can be sustained. The study was also limited to English-speaking families. Given that most home visitors attended to families in both Recipe 4 Success and EHS, the researchers noted the possibility for contamination across conditions, but they added that this would have actually reduced the intervention effects. The study’s primary strength was the evidenced-based nature of the randomized control. That Recipe 4 Success was operated as an intervention only strengthen the benefits of normal EHS visits.
Patient parents who promote self-regulation have the best chance of success
“This small study emphasizes the importance of parent education and support in setting the toddlers’ palate for lifelong eating habits and self-regulation,” observed Silver Spring, MD, private practice pediatrician and associate clinical professor of pediatrics at George Washington University, Washington, Lillian M. Beard, MD, in a separate interview.
“With the goal of promoting eating habits and self-regulation, I try to guide parents’ choices of what they offer to their toddler. I applaud parents’ patience as I encourage them not to give in and quickly resort to offering salty or sweet snacks. I suggest that if during the course of a day, a palette of colorful healthy choices is offered, most toddlers will graze independently as they go about their play. The challenge is to really support the parent through this quirky stage of their child’s development,” she explained.
“The ultimate challenge today with so much food insecurity, COVID-19 related job losses, and shrinking dollars to feed families is that too many families are feeling a food crisis! A program such as Recipe 4 Success can provide invaluable education for families on how to best stretch their few dollars, with knowledge of which items to seek from their community food pantries, how to best utilize items from the State WIC programs and still seek nutrition tips from their pediatricians while avoiding expensive fast foods that only offer immediate satiety and gratification. The Recipe 4 Success educator, pediatrician, or any community educator can give recommendations about which fresh produce may be inexpensive, but nutritional,” Dr. Beard suggested.
Dr. Nix and colleagues as well as Dr. Beard had no conflicts of interest and no relevant financial disclosures.
Home-based preventive interventions not only improve healthy eating habits and self-regulation in toddlers but also guide their parents toward better food presentation and response to picky behaviors, reported Robert L. Nix, PhD, of the University of Wisconsin, Madison, and his associates.
In a small, randomized controlled trial of 73 families with toddlers aged 18-36 months enrolled in home-based Early Head Start (EHS), the researchers evaluated four protective factors, including toddlers’ healthy eating habits, toddlers’ self-regulation, parents’ responsive feeding practices, and parents’ sensitive scaffolding. The study, conducted from April to October 2013, is the first clinical trial of Recipe 4 Success, a preschool-focused intervention created by administrators and home visitors of EHS that promotes healthy eating and self-regulation in toddlers living in poverty who may otherwise face weight challenges and obesity later in life. Integrating the intervention into EHS allowed the researchers to take full advantage of its national infrastructure and to make dissemination more efficient.
Of the families selected to participate, all of whom were living below the Federal poverty threshold, 66 were retained through post treatment. Most participating parents were biological mothers; 61% were single; 29% were not high school graduates; just 11% were employed full time. The toddlers averaged 30.72 months; 44% were female. Roughly 48% of families were non-Hispanic White; 29% were Black; and 23% were Hispanic or Latinx, the investigators reported in Pediatrics. More than three-quarters of participants were enrolled in the Special Supplemental Nutrition Program for Women, Infants, and Children or the Supplemental Nutrition Assistance Program.
The program allowed parents to transform toddler eating habits quickly
The study, which was designed to evaluate for posttreatment differences in the primary outcomes, involved 10 consecutive weekly lessons implemented by regularly assigned EHS home visitors. Parents were required to adhere to feeding practices carefully targeted with sensitive, structured scaffolding designed to keep toddlers from becoming overwhelmed. Parents were guided to understand, for instance, that toddlers frequently need to be exposed to a new food 10-20 times before eating it, and that poor sleep can have a detrimental influence on emotional and behavioral controls that can progress to weight gain.
Parent recall of what food and drinks their toddlers consumed in the previous 24-hour period was collected by interviewers. The percentage of all meals that included a fruit and/or vegetable, a protein source, and the absence of sweets and junk food were noted. Toddler self-regulation was assessed in accordance with delay of gratification, task orientation, and emotional/behavioral control. Parents were asked to rate toddler ability to cease desired activities to comply with parental requests. Parental responsive feeding practices were also recorded to observe how they introduce unique healthy foods and how they responded to their toddlers’ reactions. Parental sensitive scaffolding was similarly observed for their ability to structure activities in a developmentally appropriate manner promoting self-regulation.
The researchers noted no statistically significant differences between families in the treatment and control groups, nor were there differences in outcome measures or covariates. Study findings showed that, compared with toddlers who continued to receive just EHS support, the toddlers randomly assigned to Recipe 4 Success were more likely to consume snacks and meals that contained fruits, vegetables, protein, and no sweets or junk food.
As the results of this study and others have shown, early food preferences offer the strongest indication of later diet and healthy eating habits throughout life. The program targeted in this study is significant in its ability to accelerate the adoption of better toddler eating habits in just a 10-week period.
Recipe 4 Success along with other successful preventive interventions for young children are most effective when parents drive the change. “In the present trial, the quality of parenting was most highly related to healthy eating habits and self-regulation at baseline,” the researchers noted.
Specifically, the authors attributed the success of the program to “targeting specific interrelated outcomes with an integrated, theoretically driven intervention model,” which allowed Recipe 4 Success to boost the effectiveness of EHS substantially “in just 10 weeks with a minimal increase in funding,” the authors added.
The authors noted several weaknesses as well as strengths of the study. Its primary weakness was a baseline-posttreatment design, which made it impossible to assert that intervention effects can be sustained. The study was also limited to English-speaking families. Given that most home visitors attended to families in both Recipe 4 Success and EHS, the researchers noted the possibility for contamination across conditions, but they added that this would have actually reduced the intervention effects. The study’s primary strength was the evidenced-based nature of the randomized control. That Recipe 4 Success was operated as an intervention only strengthen the benefits of normal EHS visits.
Patient parents who promote self-regulation have the best chance of success
“This small study emphasizes the importance of parent education and support in setting the toddlers’ palate for lifelong eating habits and self-regulation,” observed Silver Spring, MD, private practice pediatrician and associate clinical professor of pediatrics at George Washington University, Washington, Lillian M. Beard, MD, in a separate interview.
“With the goal of promoting eating habits and self-regulation, I try to guide parents’ choices of what they offer to their toddler. I applaud parents’ patience as I encourage them not to give in and quickly resort to offering salty or sweet snacks. I suggest that if during the course of a day, a palette of colorful healthy choices is offered, most toddlers will graze independently as they go about their play. The challenge is to really support the parent through this quirky stage of their child’s development,” she explained.
“The ultimate challenge today with so much food insecurity, COVID-19 related job losses, and shrinking dollars to feed families is that too many families are feeling a food crisis! A program such as Recipe 4 Success can provide invaluable education for families on how to best stretch their few dollars, with knowledge of which items to seek from their community food pantries, how to best utilize items from the State WIC programs and still seek nutrition tips from their pediatricians while avoiding expensive fast foods that only offer immediate satiety and gratification. The Recipe 4 Success educator, pediatrician, or any community educator can give recommendations about which fresh produce may be inexpensive, but nutritional,” Dr. Beard suggested.
Dr. Nix and colleagues as well as Dr. Beard had no conflicts of interest and no relevant financial disclosures.
FROM PEDIATRICS
COVID-19 in children: Latest weekly increase is largest yet
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
There were 211,466 new cases reported in children during the week of Jan. 8-14, topping the previous high (Dec. 11-17) by almost 30,000. Those new cases bring the total for the pandemic to over 2.5 million children infected with the coronavirus, which represents 12.6% of all reported cases, the AAP and the CHA said Jan. 19 in their weekly COVID-19 report.
The rise in cases also brought an increase in the proportion reported among children. The week before (Jan. 1-7), cases in children were 12.9% of all cases reported, but the most recent week saw that number rise to 14.5% of all cases, the highest it’s been since early October, based on data collected from the health department websites of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rio, and Guam.
The corresponding figures for severe illness continue to be low: Children represent 1.8% of all hospitalizations from COVID-19 in 24 states and New York City and 0.06% of all deaths in 43 states and New York City. Three deaths were reported for the week of Jan. 8-14, making for a total of 191 since the pandemic started, the AAP and CHA said in their report.
Among the states, California has the most overall cases at just over 350,000, Wyoming has the highest proportion of cases in children (20.3%), and North Dakota has the highest rate of infection (over 8,100 per 100,000 children). The infection rate for the nation is now above 3,300 per 100,000 children, and 11 states reported rates over 5,000, according to the AAP and the CHA.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

There were 211,466 new cases reported in children during the week of Jan. 8-14, topping the previous high (Dec. 11-17) by almost 30,000. Those new cases bring the total for the pandemic to over 2.5 million children infected with the coronavirus, which represents 12.6% of all reported cases, the AAP and the CHA said Jan. 19 in their weekly COVID-19 report.
The rise in cases also brought an increase in the proportion reported among children. The week before (Jan. 1-7), cases in children were 12.9% of all cases reported, but the most recent week saw that number rise to 14.5% of all cases, the highest it’s been since early October, based on data collected from the health department websites of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rio, and Guam.
The corresponding figures for severe illness continue to be low: Children represent 1.8% of all hospitalizations from COVID-19 in 24 states and New York City and 0.06% of all deaths in 43 states and New York City. Three deaths were reported for the week of Jan. 8-14, making for a total of 191 since the pandemic started, the AAP and CHA said in their report.
Among the states, California has the most overall cases at just over 350,000, Wyoming has the highest proportion of cases in children (20.3%), and North Dakota has the highest rate of infection (over 8,100 per 100,000 children). The infection rate for the nation is now above 3,300 per 100,000 children, and 11 states reported rates over 5,000, according to the AAP and the CHA.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

There were 211,466 new cases reported in children during the week of Jan. 8-14, topping the previous high (Dec. 11-17) by almost 30,000. Those new cases bring the total for the pandemic to over 2.5 million children infected with the coronavirus, which represents 12.6% of all reported cases, the AAP and the CHA said Jan. 19 in their weekly COVID-19 report.
The rise in cases also brought an increase in the proportion reported among children. The week before (Jan. 1-7), cases in children were 12.9% of all cases reported, but the most recent week saw that number rise to 14.5% of all cases, the highest it’s been since early October, based on data collected from the health department websites of 49 states (excluding New York), the District of Columbia, New York City, Puerto Rio, and Guam.
The corresponding figures for severe illness continue to be low: Children represent 1.8% of all hospitalizations from COVID-19 in 24 states and New York City and 0.06% of all deaths in 43 states and New York City. Three deaths were reported for the week of Jan. 8-14, making for a total of 191 since the pandemic started, the AAP and CHA said in their report.
Among the states, California has the most overall cases at just over 350,000, Wyoming has the highest proportion of cases in children (20.3%), and North Dakota has the highest rate of infection (over 8,100 per 100,000 children). The infection rate for the nation is now above 3,300 per 100,000 children, and 11 states reported rates over 5,000, according to the AAP and the CHA.
Schools, COVID-19, and Jan. 6, 2021
The first weeks of 2021 have us considering how best to face compound challenges and we expect parents will be looking to their pediatricians for guidance. There are daily stories of the COVID-19 death toll, an abstraction made real by tragic stories of shattered families. Most families are approaching the first anniversary of their children being in virtual school, with growing concerns about the quality of virtual education, loss of socialization and group activities, and additional risks facing poor and vulnerable children. There are real concerns about the future impact of children spending so much time every day on their screens for school, extracurricular activities, social time, and relaxation. While the COVID-19 vaccines promise a return to “normal” sometime in 2021, in-person school may not return until late in the spring or next fall.
After the events of Jan. 6, families face an additional challenge: Discussing the violent invasion of the U.S. Capitol by the president’s supporters. This event was shocking, frightening, and confusing for most, and continues to be heavily covered in the news and online. There is a light in all this darkness. We have the opportunity to talk with our children – and to share explanations, perspectives, values, and even the discomfort of the unknowns – about COVID-19, use of the Internet, and the violence of Jan 6. We will consider how parents can approach this challenge for three age groups. With each group, parents will need to be calm and curious and will need time to give their children their full attention. We are all living through history. When parents can be fully present with their children, even for short periods at meals or at bedtime, it will help all to get their balance back and start to make sense of the extraordinary events we have been facing.
The youngest children (aged 3-6 years), those who were in preschool or kindergarten before the pandemic, need the most from their parents during this time. If they are attending school virtually, their online school days are likely short and challenging. Children at this age are mastering behavior rather than cognitive tasks. They are learning how to manage their bodies in space (stay in their seats!), how to be patient and kind (take turns!), and how to manage frustration (math is hard, try again!). Without the physical presence of their teacher and classmates, these lessons are tougher to internalize. Given their age-appropriate short attention spans, they often walk away from a screen, even if it’s class time. They are more likely to be paying attention to their parents, responding to the emotional climate at home. Even if they are not watching news websites themselves, they are likely to have overheard or noticed the news about recent events. Parents of young children should take care to turn off the television or their own computer, as repeated frightening videos of the insurrection can cause their children to worry that these events continue to unfold. These children need their parents’ undivided attention, even just for a little while. Play a board game with them (good chance to stay in their seats, take turns, and manage losing). Or get them outside for some physical play. While playing, parents can ask what they have seen, heard, or understand about what happened in the Capitol. Then they can correct misperceptions that might be frightening and offer reasonable reassurances in language these young children can understand. They might tell their children that sometimes people get angry when they have lost, and even adults can behave badly and make mistakes. They can focus on who the helpers are, and what they could do to help also. They could write letters of appreciation to their elected officials or to the Capitol police who were so brave in protecting others. If their children are curious, parents can find books or videos that are age appropriate about the Constitution and how elections work in a democracy. Parents don’t need to be able to answer every question, watching “Schoolhouse Rock” videos on YouTube together is a wonderful way to complement their online school and support their healthy development.
School-aged children (7-12 years) are developmentally focused on mastery experiences, whether they are social, academic, or athletic. They may be better equipped to pay attention and do homework than their younger siblings, but they will miss building friendships and having a real audience for their efforts as they build emotional maturity. They are prone to worry and distress about the big events that they can understand, at least in concrete terms, but have never faced before. These children usually have been able to use social media and online games to stay connected to friends, but they are less likely than their older siblings to independently exercise or explore new interests without a parent or teacher to guide and support them. These children are likely to be spending a lot of their time online on websites their parents don’t know about, and most likely to be curious about the events of Jan. 6. Parents should close their own device and invite their school-age children to show them what they are working on in school. Be curious about all of it, even how they are doing gym or music class. Then ask about what they have seen or heard about the election and its aftermath at school, from friends, or on their own. Let them be the teachers about what happened and how they learned about it. Parents can correct misinformation or offer reliable sources of information they can investigate together. What they will need is validation of the difficult feelings that events like these can cause; that is, acknowledgment, acceptance, and understanding of big feelings, without trying to just make those feelings go away. Parents might help them to be curious about what can make people get angry, break laws, and even hurt others, and how we protest injustices in a democracy. These children may be ready to take a deeper dive into history, via a good film or documentary, with their parents’ company for discussion afterward. Be their audience and model curiosity and patience, all the while validating the feelings that might arise.
Teenagers are developmentally focused on building their own identities, cultivating independence, and deeper relationships beyond their family. While they may be well equipped to manage online learning and to stay connected to their friends and teachers through electronic means, they are also facing considerable challenge, as their ability to explore new interests, build new relationships, and be meaningfully independent has been profoundly restrained over the past year. And they are facing other losses, as milestones like proms, performances, and competitions have been altered or missed. Parents still know when their teenager is most likely to talk, and they should check in with them during those times. They can ask them about what classes are working online and which ones aren’t, and what extracurriculars are still possible. They should not be discouraged if their teenager only offers cursory responses, it matters that they are showing up and showing interest. The election and its aftermath provide a meaningful matter to discuss; parents can find out if it is being discussed by any teachers or friends. What do they think triggered the events of Jan. 6? Who should be held responsible? How to reasonably protest injustice? What does a society do when citizens can’t agree on facts? More than offering reassurance, parents should be curious about their adolescent’s developing identity and their values, how they are thinking about complex issues around free speech and justice. It is a wonderful opportunity for parents to learn about their adolescent’s emerging identity, to be tolerant of their autonomy, and an opportunity to offer their perspective and values.
At every age, parents need to be present by listening and drawing their children out without distraction. Now is a time to build relationships and to use the difficult events of the day to shed light on deeper issues and values. This is hard, but far better than having children deal with these issues in darkness or alone.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics at Harvard Medical School, Boston. Email them at [email protected].
The first weeks of 2021 have us considering how best to face compound challenges and we expect parents will be looking to their pediatricians for guidance. There are daily stories of the COVID-19 death toll, an abstraction made real by tragic stories of shattered families. Most families are approaching the first anniversary of their children being in virtual school, with growing concerns about the quality of virtual education, loss of socialization and group activities, and additional risks facing poor and vulnerable children. There are real concerns about the future impact of children spending so much time every day on their screens for school, extracurricular activities, social time, and relaxation. While the COVID-19 vaccines promise a return to “normal” sometime in 2021, in-person school may not return until late in the spring or next fall.
After the events of Jan. 6, families face an additional challenge: Discussing the violent invasion of the U.S. Capitol by the president’s supporters. This event was shocking, frightening, and confusing for most, and continues to be heavily covered in the news and online. There is a light in all this darkness. We have the opportunity to talk with our children – and to share explanations, perspectives, values, and even the discomfort of the unknowns – about COVID-19, use of the Internet, and the violence of Jan 6. We will consider how parents can approach this challenge for three age groups. With each group, parents will need to be calm and curious and will need time to give their children their full attention. We are all living through history. When parents can be fully present with their children, even for short periods at meals or at bedtime, it will help all to get their balance back and start to make sense of the extraordinary events we have been facing.
The youngest children (aged 3-6 years), those who were in preschool or kindergarten before the pandemic, need the most from their parents during this time. If they are attending school virtually, their online school days are likely short and challenging. Children at this age are mastering behavior rather than cognitive tasks. They are learning how to manage their bodies in space (stay in their seats!), how to be patient and kind (take turns!), and how to manage frustration (math is hard, try again!). Without the physical presence of their teacher and classmates, these lessons are tougher to internalize. Given their age-appropriate short attention spans, they often walk away from a screen, even if it’s class time. They are more likely to be paying attention to their parents, responding to the emotional climate at home. Even if they are not watching news websites themselves, they are likely to have overheard or noticed the news about recent events. Parents of young children should take care to turn off the television or their own computer, as repeated frightening videos of the insurrection can cause their children to worry that these events continue to unfold. These children need their parents’ undivided attention, even just for a little while. Play a board game with them (good chance to stay in their seats, take turns, and manage losing). Or get them outside for some physical play. While playing, parents can ask what they have seen, heard, or understand about what happened in the Capitol. Then they can correct misperceptions that might be frightening and offer reasonable reassurances in language these young children can understand. They might tell their children that sometimes people get angry when they have lost, and even adults can behave badly and make mistakes. They can focus on who the helpers are, and what they could do to help also. They could write letters of appreciation to their elected officials or to the Capitol police who were so brave in protecting others. If their children are curious, parents can find books or videos that are age appropriate about the Constitution and how elections work in a democracy. Parents don’t need to be able to answer every question, watching “Schoolhouse Rock” videos on YouTube together is a wonderful way to complement their online school and support their healthy development.
School-aged children (7-12 years) are developmentally focused on mastery experiences, whether they are social, academic, or athletic. They may be better equipped to pay attention and do homework than their younger siblings, but they will miss building friendships and having a real audience for their efforts as they build emotional maturity. They are prone to worry and distress about the big events that they can understand, at least in concrete terms, but have never faced before. These children usually have been able to use social media and online games to stay connected to friends, but they are less likely than their older siblings to independently exercise or explore new interests without a parent or teacher to guide and support them. These children are likely to be spending a lot of their time online on websites their parents don’t know about, and most likely to be curious about the events of Jan. 6. Parents should close their own device and invite their school-age children to show them what they are working on in school. Be curious about all of it, even how they are doing gym or music class. Then ask about what they have seen or heard about the election and its aftermath at school, from friends, or on their own. Let them be the teachers about what happened and how they learned about it. Parents can correct misinformation or offer reliable sources of information they can investigate together. What they will need is validation of the difficult feelings that events like these can cause; that is, acknowledgment, acceptance, and understanding of big feelings, without trying to just make those feelings go away. Parents might help them to be curious about what can make people get angry, break laws, and even hurt others, and how we protest injustices in a democracy. These children may be ready to take a deeper dive into history, via a good film or documentary, with their parents’ company for discussion afterward. Be their audience and model curiosity and patience, all the while validating the feelings that might arise.
Teenagers are developmentally focused on building their own identities, cultivating independence, and deeper relationships beyond their family. While they may be well equipped to manage online learning and to stay connected to their friends and teachers through electronic means, they are also facing considerable challenge, as their ability to explore new interests, build new relationships, and be meaningfully independent has been profoundly restrained over the past year. And they are facing other losses, as milestones like proms, performances, and competitions have been altered or missed. Parents still know when their teenager is most likely to talk, and they should check in with them during those times. They can ask them about what classes are working online and which ones aren’t, and what extracurriculars are still possible. They should not be discouraged if their teenager only offers cursory responses, it matters that they are showing up and showing interest. The election and its aftermath provide a meaningful matter to discuss; parents can find out if it is being discussed by any teachers or friends. What do they think triggered the events of Jan. 6? Who should be held responsible? How to reasonably protest injustice? What does a society do when citizens can’t agree on facts? More than offering reassurance, parents should be curious about their adolescent’s developing identity and their values, how they are thinking about complex issues around free speech and justice. It is a wonderful opportunity for parents to learn about their adolescent’s emerging identity, to be tolerant of their autonomy, and an opportunity to offer their perspective and values.
At every age, parents need to be present by listening and drawing their children out without distraction. Now is a time to build relationships and to use the difficult events of the day to shed light on deeper issues and values. This is hard, but far better than having children deal with these issues in darkness or alone.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics at Harvard Medical School, Boston. Email them at [email protected].
The first weeks of 2021 have us considering how best to face compound challenges and we expect parents will be looking to their pediatricians for guidance. There are daily stories of the COVID-19 death toll, an abstraction made real by tragic stories of shattered families. Most families are approaching the first anniversary of their children being in virtual school, with growing concerns about the quality of virtual education, loss of socialization and group activities, and additional risks facing poor and vulnerable children. There are real concerns about the future impact of children spending so much time every day on their screens for school, extracurricular activities, social time, and relaxation. While the COVID-19 vaccines promise a return to “normal” sometime in 2021, in-person school may not return until late in the spring or next fall.
After the events of Jan. 6, families face an additional challenge: Discussing the violent invasion of the U.S. Capitol by the president’s supporters. This event was shocking, frightening, and confusing for most, and continues to be heavily covered in the news and online. There is a light in all this darkness. We have the opportunity to talk with our children – and to share explanations, perspectives, values, and even the discomfort of the unknowns – about COVID-19, use of the Internet, and the violence of Jan 6. We will consider how parents can approach this challenge for three age groups. With each group, parents will need to be calm and curious and will need time to give their children their full attention. We are all living through history. When parents can be fully present with their children, even for short periods at meals or at bedtime, it will help all to get their balance back and start to make sense of the extraordinary events we have been facing.
The youngest children (aged 3-6 years), those who were in preschool or kindergarten before the pandemic, need the most from their parents during this time. If they are attending school virtually, their online school days are likely short and challenging. Children at this age are mastering behavior rather than cognitive tasks. They are learning how to manage their bodies in space (stay in their seats!), how to be patient and kind (take turns!), and how to manage frustration (math is hard, try again!). Without the physical presence of their teacher and classmates, these lessons are tougher to internalize. Given their age-appropriate short attention spans, they often walk away from a screen, even if it’s class time. They are more likely to be paying attention to their parents, responding to the emotional climate at home. Even if they are not watching news websites themselves, they are likely to have overheard or noticed the news about recent events. Parents of young children should take care to turn off the television or their own computer, as repeated frightening videos of the insurrection can cause their children to worry that these events continue to unfold. These children need their parents’ undivided attention, even just for a little while. Play a board game with them (good chance to stay in their seats, take turns, and manage losing). Or get them outside for some physical play. While playing, parents can ask what they have seen, heard, or understand about what happened in the Capitol. Then they can correct misperceptions that might be frightening and offer reasonable reassurances in language these young children can understand. They might tell their children that sometimes people get angry when they have lost, and even adults can behave badly and make mistakes. They can focus on who the helpers are, and what they could do to help also. They could write letters of appreciation to their elected officials or to the Capitol police who were so brave in protecting others. If their children are curious, parents can find books or videos that are age appropriate about the Constitution and how elections work in a democracy. Parents don’t need to be able to answer every question, watching “Schoolhouse Rock” videos on YouTube together is a wonderful way to complement their online school and support their healthy development.
School-aged children (7-12 years) are developmentally focused on mastery experiences, whether they are social, academic, or athletic. They may be better equipped to pay attention and do homework than their younger siblings, but they will miss building friendships and having a real audience for their efforts as they build emotional maturity. They are prone to worry and distress about the big events that they can understand, at least in concrete terms, but have never faced before. These children usually have been able to use social media and online games to stay connected to friends, but they are less likely than their older siblings to independently exercise or explore new interests without a parent or teacher to guide and support them. These children are likely to be spending a lot of their time online on websites their parents don’t know about, and most likely to be curious about the events of Jan. 6. Parents should close their own device and invite their school-age children to show them what they are working on in school. Be curious about all of it, even how they are doing gym or music class. Then ask about what they have seen or heard about the election and its aftermath at school, from friends, or on their own. Let them be the teachers about what happened and how they learned about it. Parents can correct misinformation or offer reliable sources of information they can investigate together. What they will need is validation of the difficult feelings that events like these can cause; that is, acknowledgment, acceptance, and understanding of big feelings, without trying to just make those feelings go away. Parents might help them to be curious about what can make people get angry, break laws, and even hurt others, and how we protest injustices in a democracy. These children may be ready to take a deeper dive into history, via a good film or documentary, with their parents’ company for discussion afterward. Be their audience and model curiosity and patience, all the while validating the feelings that might arise.
Teenagers are developmentally focused on building their own identities, cultivating independence, and deeper relationships beyond their family. While they may be well equipped to manage online learning and to stay connected to their friends and teachers through electronic means, they are also facing considerable challenge, as their ability to explore new interests, build new relationships, and be meaningfully independent has been profoundly restrained over the past year. And they are facing other losses, as milestones like proms, performances, and competitions have been altered or missed. Parents still know when their teenager is most likely to talk, and they should check in with them during those times. They can ask them about what classes are working online and which ones aren’t, and what extracurriculars are still possible. They should not be discouraged if their teenager only offers cursory responses, it matters that they are showing up and showing interest. The election and its aftermath provide a meaningful matter to discuss; parents can find out if it is being discussed by any teachers or friends. What do they think triggered the events of Jan. 6? Who should be held responsible? How to reasonably protest injustice? What does a society do when citizens can’t agree on facts? More than offering reassurance, parents should be curious about their adolescent’s developing identity and their values, how they are thinking about complex issues around free speech and justice. It is a wonderful opportunity for parents to learn about their adolescent’s emerging identity, to be tolerant of their autonomy, and an opportunity to offer their perspective and values.
At every age, parents need to be present by listening and drawing their children out without distraction. Now is a time to build relationships and to use the difficult events of the day to shed light on deeper issues and values. This is hard, but far better than having children deal with these issues in darkness or alone.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics at Harvard Medical School, Boston. Email them at [email protected].