User login
There’s hope: Curbing food wastage at the school cafeteria
It’s hard to find good news these days, but I thought I had stumbled on a nice feel-good story in the Portland Press Herald. It turns out a group of students at the King Middle School in Portland, Maine, has formed a team of “compost guardians,” who by coaxing their fellow students into sorting their uneaten lunch food into five reusable or recyclable categories have reduced the cafeteria’s daily waste production from 12 40-gallon trash bags to 2. (“Each year, Maine’s K-12 schools waste about 7 million pounds of food,” by Rachel Ohm, Portland Press Herald, Feb. 2, 2020). That seems like a heroic accomplishment and good news by any standard.
However, as I read on in the newspaper article it became clear that these students’ efforts represent a tiny speck of light in the middle of a very dark tunnel. In developing their system, the students learned that 34% of the food was not being consumed, which is part of the 30%-40% of food wasted across the country. In Maine, this represents about 7 million pounds of food wasted annually. Not surprisingly, the students found that 60% of the fruit and 28% of the vegetables go uneaten.
But current federal guidelines dictate that students must take a vegetable and a fruit on their trays. While well-intentioned, this is a mandate destined to generate waste.
King Middle School and many other schools around the country offer a program that is not in the federal guidelines: a “share table” where students can place unwanted (but safe to eat) food, and from which other students may serve themselves freely. On the surface, this may seem like a good idea because it legitimizes what children have been doing for years on their own. This shared food should consist of “healthy choices” because it is cafeteria fare dictated by the United States Department of Agriculture. But you know as well as I do that a child can become obese overeating a diet that in moderate amounts would be considered healthy. I suspect that many, if not most, students taking food from the share table don’t need any extra calories.
The USDA recently announced that it will be rolling out reforms for school and summer meal programs (USDA Release # USDA 0129.20). One of the goals of these reforms is to reduce food wastage by giving schools more flexibility in creating menus and offering more à la carte options. In the wake this rollout, there has been some concern voiced that schools will begin to offer less nutritional options. Unfortunately, this concern may be true in some districts, but it is pretty clear that the current guidelines are a significant contributor to food wastage without offering much of an upside. It may be time to lessen the record-keeping burden on local food services, and allow them some leeway in creating more appealing options while taking advantage of local food sources.
With or without the new guidelines, we are asking public schools to cater to multiple cohorts of students whose parents have put them on the bus in the morning as mismanaged picky eaters.
What would have worked at home could work at school. That strategy is to offer a child a balanced diet presented in an appealing manner in a pleasant setting. Also it is not allowing any sweetened beverages or milk in excess. Children may grumble temporarily but if the strategy is applied consistently, they will take it from there. That doesn’t mean that the children have to put the food on their trays if they don’t want it. But they shouldn’t be offered a second run through the cafeteria line or a chance to pick from the share table. Sadly, the success of this strategy relies on two shaky premises: That parents will begin to apply it at home and that school lunch programs will offer only healthy choices.
The bottom line is that schools can’t be expected to cure picky eaters who were enabled at home. On the other hand, it is not unreasonable to ask schools to play a role in curbing the national scourge of food wastage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping with a Picky Eater: A Guide for the Perplexed Parent.” Email him at [email protected].
It’s hard to find good news these days, but I thought I had stumbled on a nice feel-good story in the Portland Press Herald. It turns out a group of students at the King Middle School in Portland, Maine, has formed a team of “compost guardians,” who by coaxing their fellow students into sorting their uneaten lunch food into five reusable or recyclable categories have reduced the cafeteria’s daily waste production from 12 40-gallon trash bags to 2. (“Each year, Maine’s K-12 schools waste about 7 million pounds of food,” by Rachel Ohm, Portland Press Herald, Feb. 2, 2020). That seems like a heroic accomplishment and good news by any standard.
However, as I read on in the newspaper article it became clear that these students’ efforts represent a tiny speck of light in the middle of a very dark tunnel. In developing their system, the students learned that 34% of the food was not being consumed, which is part of the 30%-40% of food wasted across the country. In Maine, this represents about 7 million pounds of food wasted annually. Not surprisingly, the students found that 60% of the fruit and 28% of the vegetables go uneaten.
But current federal guidelines dictate that students must take a vegetable and a fruit on their trays. While well-intentioned, this is a mandate destined to generate waste.
King Middle School and many other schools around the country offer a program that is not in the federal guidelines: a “share table” where students can place unwanted (but safe to eat) food, and from which other students may serve themselves freely. On the surface, this may seem like a good idea because it legitimizes what children have been doing for years on their own. This shared food should consist of “healthy choices” because it is cafeteria fare dictated by the United States Department of Agriculture. But you know as well as I do that a child can become obese overeating a diet that in moderate amounts would be considered healthy. I suspect that many, if not most, students taking food from the share table don’t need any extra calories.
The USDA recently announced that it will be rolling out reforms for school and summer meal programs (USDA Release # USDA 0129.20). One of the goals of these reforms is to reduce food wastage by giving schools more flexibility in creating menus and offering more à la carte options. In the wake this rollout, there has been some concern voiced that schools will begin to offer less nutritional options. Unfortunately, this concern may be true in some districts, but it is pretty clear that the current guidelines are a significant contributor to food wastage without offering much of an upside. It may be time to lessen the record-keeping burden on local food services, and allow them some leeway in creating more appealing options while taking advantage of local food sources.
With or without the new guidelines, we are asking public schools to cater to multiple cohorts of students whose parents have put them on the bus in the morning as mismanaged picky eaters.
What would have worked at home could work at school. That strategy is to offer a child a balanced diet presented in an appealing manner in a pleasant setting. Also it is not allowing any sweetened beverages or milk in excess. Children may grumble temporarily but if the strategy is applied consistently, they will take it from there. That doesn’t mean that the children have to put the food on their trays if they don’t want it. But they shouldn’t be offered a second run through the cafeteria line or a chance to pick from the share table. Sadly, the success of this strategy relies on two shaky premises: That parents will begin to apply it at home and that school lunch programs will offer only healthy choices.
The bottom line is that schools can’t be expected to cure picky eaters who were enabled at home. On the other hand, it is not unreasonable to ask schools to play a role in curbing the national scourge of food wastage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping with a Picky Eater: A Guide for the Perplexed Parent.” Email him at [email protected].
It’s hard to find good news these days, but I thought I had stumbled on a nice feel-good story in the Portland Press Herald. It turns out a group of students at the King Middle School in Portland, Maine, has formed a team of “compost guardians,” who by coaxing their fellow students into sorting their uneaten lunch food into five reusable or recyclable categories have reduced the cafeteria’s daily waste production from 12 40-gallon trash bags to 2. (“Each year, Maine’s K-12 schools waste about 7 million pounds of food,” by Rachel Ohm, Portland Press Herald, Feb. 2, 2020). That seems like a heroic accomplishment and good news by any standard.
However, as I read on in the newspaper article it became clear that these students’ efforts represent a tiny speck of light in the middle of a very dark tunnel. In developing their system, the students learned that 34% of the food was not being consumed, which is part of the 30%-40% of food wasted across the country. In Maine, this represents about 7 million pounds of food wasted annually. Not surprisingly, the students found that 60% of the fruit and 28% of the vegetables go uneaten.
But current federal guidelines dictate that students must take a vegetable and a fruit on their trays. While well-intentioned, this is a mandate destined to generate waste.
King Middle School and many other schools around the country offer a program that is not in the federal guidelines: a “share table” where students can place unwanted (but safe to eat) food, and from which other students may serve themselves freely. On the surface, this may seem like a good idea because it legitimizes what children have been doing for years on their own. This shared food should consist of “healthy choices” because it is cafeteria fare dictated by the United States Department of Agriculture. But you know as well as I do that a child can become obese overeating a diet that in moderate amounts would be considered healthy. I suspect that many, if not most, students taking food from the share table don’t need any extra calories.
The USDA recently announced that it will be rolling out reforms for school and summer meal programs (USDA Release # USDA 0129.20). One of the goals of these reforms is to reduce food wastage by giving schools more flexibility in creating menus and offering more à la carte options. In the wake this rollout, there has been some concern voiced that schools will begin to offer less nutritional options. Unfortunately, this concern may be true in some districts, but it is pretty clear that the current guidelines are a significant contributor to food wastage without offering much of an upside. It may be time to lessen the record-keeping burden on local food services, and allow them some leeway in creating more appealing options while taking advantage of local food sources.
With or without the new guidelines, we are asking public schools to cater to multiple cohorts of students whose parents have put them on the bus in the morning as mismanaged picky eaters.
What would have worked at home could work at school. That strategy is to offer a child a balanced diet presented in an appealing manner in a pleasant setting. Also it is not allowing any sweetened beverages or milk in excess. Children may grumble temporarily but if the strategy is applied consistently, they will take it from there. That doesn’t mean that the children have to put the food on their trays if they don’t want it. But they shouldn’t be offered a second run through the cafeteria line or a chance to pick from the share table. Sadly, the success of this strategy relies on two shaky premises: That parents will begin to apply it at home and that school lunch programs will offer only healthy choices.
The bottom line is that schools can’t be expected to cure picky eaters who were enabled at home. On the other hand, it is not unreasonable to ask schools to play a role in curbing the national scourge of food wastage.
Dr. Wilkoff practiced primary care pediatrics in Brunswick, Maine for nearly 40 years. He has authored several books on behavioral pediatrics, including “Coping with a Picky Eater: A Guide for the Perplexed Parent.” Email him at [email protected].
Gender pronouns in EMR preferred by many gender nonconforming teens
Most transgender and gender nonconforming youth would like their preferred name and pronouns be recorded throughout their EMRs, but very few are ever asked for that identity information outside of gender specialty clinic settings, according to a recent research letter in JAMA Pediatrics.
The findings are not surprising, said Cora Breuner, MD, a professor of pediatrics in adolescent medicine at Seattle Children’s Hospital in Washington, because “we know that use of gender-affirming language when accessing health care is extremely important to transgender youth.”
“Use of gender-affirming language in the health care system is associated with improved mental health outcomes in this population,” Dr Breuner said in an interview.
But the authors of the study noted that EMRs often lack the functions needed to provide gender-affirming care.
“To better support this vulnerable group of youths, health systems and EMRs should allow for EMR-wide name and pronoun documentation, even when a patient has not legally changed their name,” Gina M. Sequeira, MD, of UPMC Children’s Hospital of Pittsburgh and associates wrote.
Although many providers have begun routinely asking patients for both their gender identity and their sex assigned at birth, these questions leave out a patient’s preferred name and pronouns – crucial components of respectful and affirming care, the authors explained.
At a specialty gender clinic, the authors surveyed 204 transgender youths, aged 12-26 years, regarding how their name and pronouns are recorded in their EMR files. Just over half the respondents were under age 18 years (56%), and most were white (86%). Most were transmasculine (59%), with 21% transfeminine and 20% nonbinary.
Most respondents (69%) went by a name other than their legal one, yet only 9% said they were frequently or always asked in clinical settings outside specialty gender centers whether they wanted their preferred name and pronouns noted in the EMR.
A majority (79%), however, said they wanted their name and pronouns noted throughout their EMR. The youths’ preferences varied according to their gender identity and how many people were aware of their gender identity, but not by age, race/ethnicity, or perceived amount of parental support.
Only two-thirds (67%) of 42 transfeminine patients wished their EMR to include their preferred name, compared with most (85%) of 121 transmasculine patients and nearly all (92%) of 37 nonbinary respondents (P = .007). Pronouns preferences were similar: All but one nonbinary respondent wanted their pronouns in the EMR, compared with 84% of transmasculine and 64% of transfeminine respondents (P=.0003).
“It may be that transfeminine patients have more pressure to ‘stay’ their assigned gender,” Dr Breuner said regarding these findings. “ ‘Outness’ may be challenging, and thus they remain in their traditional gender norms, but further research on this theory is warranted.”
Among those who were “out to everyone,” most (88%) wanted their preferred name and pronouns recorded in the EMR, and the proportion was similar for those “out to most.” But only 65% of those “out to few or no one” preferred their name and pronouns be noted in the EMR, a similar proportion for those “out to some.”
Of 7 youths who did not wish to include their name and pronouns throughout their EMR, all but one said they didn’t think it was necessary because they believed they already “passed” well enough as their gender. Just one person said they did not want name and pronouns recorded for confidentiality reasons.
However, confidentiality is still an important consideration particularly for minors, the authors and Dr. Breuner pointed out.
“It is essential to discuss confidentiality with the youth as parents may have access to the medical records younger than 18 years of age,” Dr. Breuner said.
The authors noted the study’s limitation in using a convenience sample but they and Dr. Breuner said that the findings still demonstrate transgender youths’ desire for EMRs to include their name and pronouns.
The research was funded by grants from the National Institutes of Health. The authors had no industry disclosures.
SOURCE: Sequeira GM et al. JAMA Pediatrics. 2020 Feb 23. doi: 10.1001/jamapediatrics.2019.6071.
Most transgender and gender nonconforming youth would like their preferred name and pronouns be recorded throughout their EMRs, but very few are ever asked for that identity information outside of gender specialty clinic settings, according to a recent research letter in JAMA Pediatrics.
The findings are not surprising, said Cora Breuner, MD, a professor of pediatrics in adolescent medicine at Seattle Children’s Hospital in Washington, because “we know that use of gender-affirming language when accessing health care is extremely important to transgender youth.”
“Use of gender-affirming language in the health care system is associated with improved mental health outcomes in this population,” Dr Breuner said in an interview.
But the authors of the study noted that EMRs often lack the functions needed to provide gender-affirming care.
“To better support this vulnerable group of youths, health systems and EMRs should allow for EMR-wide name and pronoun documentation, even when a patient has not legally changed their name,” Gina M. Sequeira, MD, of UPMC Children’s Hospital of Pittsburgh and associates wrote.
Although many providers have begun routinely asking patients for both their gender identity and their sex assigned at birth, these questions leave out a patient’s preferred name and pronouns – crucial components of respectful and affirming care, the authors explained.
At a specialty gender clinic, the authors surveyed 204 transgender youths, aged 12-26 years, regarding how their name and pronouns are recorded in their EMR files. Just over half the respondents were under age 18 years (56%), and most were white (86%). Most were transmasculine (59%), with 21% transfeminine and 20% nonbinary.
Most respondents (69%) went by a name other than their legal one, yet only 9% said they were frequently or always asked in clinical settings outside specialty gender centers whether they wanted their preferred name and pronouns noted in the EMR.
A majority (79%), however, said they wanted their name and pronouns noted throughout their EMR. The youths’ preferences varied according to their gender identity and how many people were aware of their gender identity, but not by age, race/ethnicity, or perceived amount of parental support.
Only two-thirds (67%) of 42 transfeminine patients wished their EMR to include their preferred name, compared with most (85%) of 121 transmasculine patients and nearly all (92%) of 37 nonbinary respondents (P = .007). Pronouns preferences were similar: All but one nonbinary respondent wanted their pronouns in the EMR, compared with 84% of transmasculine and 64% of transfeminine respondents (P=.0003).
“It may be that transfeminine patients have more pressure to ‘stay’ their assigned gender,” Dr Breuner said regarding these findings. “ ‘Outness’ may be challenging, and thus they remain in their traditional gender norms, but further research on this theory is warranted.”
Among those who were “out to everyone,” most (88%) wanted their preferred name and pronouns recorded in the EMR, and the proportion was similar for those “out to most.” But only 65% of those “out to few or no one” preferred their name and pronouns be noted in the EMR, a similar proportion for those “out to some.”
Of 7 youths who did not wish to include their name and pronouns throughout their EMR, all but one said they didn’t think it was necessary because they believed they already “passed” well enough as their gender. Just one person said they did not want name and pronouns recorded for confidentiality reasons.
However, confidentiality is still an important consideration particularly for minors, the authors and Dr. Breuner pointed out.
“It is essential to discuss confidentiality with the youth as parents may have access to the medical records younger than 18 years of age,” Dr. Breuner said.
The authors noted the study’s limitation in using a convenience sample but they and Dr. Breuner said that the findings still demonstrate transgender youths’ desire for EMRs to include their name and pronouns.
The research was funded by grants from the National Institutes of Health. The authors had no industry disclosures.
SOURCE: Sequeira GM et al. JAMA Pediatrics. 2020 Feb 23. doi: 10.1001/jamapediatrics.2019.6071.
Most transgender and gender nonconforming youth would like their preferred name and pronouns be recorded throughout their EMRs, but very few are ever asked for that identity information outside of gender specialty clinic settings, according to a recent research letter in JAMA Pediatrics.
The findings are not surprising, said Cora Breuner, MD, a professor of pediatrics in adolescent medicine at Seattle Children’s Hospital in Washington, because “we know that use of gender-affirming language when accessing health care is extremely important to transgender youth.”
“Use of gender-affirming language in the health care system is associated with improved mental health outcomes in this population,” Dr Breuner said in an interview.
But the authors of the study noted that EMRs often lack the functions needed to provide gender-affirming care.
“To better support this vulnerable group of youths, health systems and EMRs should allow for EMR-wide name and pronoun documentation, even when a patient has not legally changed their name,” Gina M. Sequeira, MD, of UPMC Children’s Hospital of Pittsburgh and associates wrote.
Although many providers have begun routinely asking patients for both their gender identity and their sex assigned at birth, these questions leave out a patient’s preferred name and pronouns – crucial components of respectful and affirming care, the authors explained.
At a specialty gender clinic, the authors surveyed 204 transgender youths, aged 12-26 years, regarding how their name and pronouns are recorded in their EMR files. Just over half the respondents were under age 18 years (56%), and most were white (86%). Most were transmasculine (59%), with 21% transfeminine and 20% nonbinary.
Most respondents (69%) went by a name other than their legal one, yet only 9% said they were frequently or always asked in clinical settings outside specialty gender centers whether they wanted their preferred name and pronouns noted in the EMR.
A majority (79%), however, said they wanted their name and pronouns noted throughout their EMR. The youths’ preferences varied according to their gender identity and how many people were aware of their gender identity, but not by age, race/ethnicity, or perceived amount of parental support.
Only two-thirds (67%) of 42 transfeminine patients wished their EMR to include their preferred name, compared with most (85%) of 121 transmasculine patients and nearly all (92%) of 37 nonbinary respondents (P = .007). Pronouns preferences were similar: All but one nonbinary respondent wanted their pronouns in the EMR, compared with 84% of transmasculine and 64% of transfeminine respondents (P=.0003).
“It may be that transfeminine patients have more pressure to ‘stay’ their assigned gender,” Dr Breuner said regarding these findings. “ ‘Outness’ may be challenging, and thus they remain in their traditional gender norms, but further research on this theory is warranted.”
Among those who were “out to everyone,” most (88%) wanted their preferred name and pronouns recorded in the EMR, and the proportion was similar for those “out to most.” But only 65% of those “out to few or no one” preferred their name and pronouns be noted in the EMR, a similar proportion for those “out to some.”
Of 7 youths who did not wish to include their name and pronouns throughout their EMR, all but one said they didn’t think it was necessary because they believed they already “passed” well enough as their gender. Just one person said they did not want name and pronouns recorded for confidentiality reasons.
However, confidentiality is still an important consideration particularly for minors, the authors and Dr. Breuner pointed out.
“It is essential to discuss confidentiality with the youth as parents may have access to the medical records younger than 18 years of age,” Dr. Breuner said.
The authors noted the study’s limitation in using a convenience sample but they and Dr. Breuner said that the findings still demonstrate transgender youths’ desire for EMRs to include their name and pronouns.
The research was funded by grants from the National Institutes of Health. The authors had no industry disclosures.
SOURCE: Sequeira GM et al. JAMA Pediatrics. 2020 Feb 23. doi: 10.1001/jamapediatrics.2019.6071.
FROM JAMA PEDIATRICS
Pediatric Dermatology Emergencies
Many pediatric skin conditions can be safely monitored with minimal intervention, but certain skin conditions are emergent and require immediate attention and proper assessment of the neonate, infant, or child. The skin may provide the first presentation of a potentially fatal disease with serious sequelae. Cutaneous findings may indicate the need for further evaluation. Therefore, it is important to differentiate skin conditions with benign etiologies from those that require immediate diagnosis and treatment, as early intervention of some of these conditions can be lifesaving. Herein, we discuss pertinent pediatric dermatology emergencies that dermatologists should keep in mind so that these diagnoses are never missed.
Staphylococcal Scalded Skin Syndrome
Presentation
Staphylococcal scalded skin syndrome (SSSS), or Ritter disease, is a potentially fatal pediatric emergency, especially in newborns.1 The mortality rate for SSSS in the United States is 3.6% to 11% in children.2 It typically presents with a prodrome of tenderness, fever, and confluent erythematous patches on the folds of the skin such as the groin, axillae, nose, and ears, with eventual spread to the legs and trunk.1,2 Within 24 to 48 hours of symptom onset, blistering and fluid accumulation will appear diffusely. Bullae are flaccid, and tangential and gentle pressure on involved unblistered skin may lead to shearing of the epithelium, which is a positive Nikolsky sign.1,2
Causes
Staphylococcal scalded skin syndrome is caused by exfoliative toxins A and B, toxigenic strains of Staphylococcus aureus. Exfoliative toxins A and B are serine proteases that target and cleave desmoglein 1, which binds keratinocytes in the stratum granulosum.1,3 Exfoliative toxins disrupt the adhesion of keratinocytes, resulting in bullae formation and subsequently diffuse sheetlike desquamation.1,4,5 Although up to 30% of the human population are asymptomatically and permanently colonized with nasal S aureus,6 the exfoliative toxins are produced by only 5% of species.1
In neonates, the immune and renal systems are underdeveloped; therefore, patients are susceptible to SSSS due to lack of neutralizing antibodies and decreased renal toxin excretion.4 Potential complications of SSSS are deeper soft-tissue infection, septicemia (blood-borne infection), and fluid and electrolyte imbalance.1,4
Diagnosis and Treatment
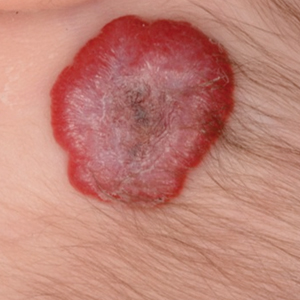
The condition is diagnosed clinically based on the findings of tender erythroderma, bullae, and desquamation with a scalded appearance, especially in friction zones; periorificial crusting; positive Nikolsky sign; and lack of mucosal involvement (Figure 1).1 Histopathology can aid in complicated clinical scenarios as well as culture from affected areas, including the upper respiratory tract, diaper region, and umbilicus.1,4 Hospitalization is required for SSSS for intravenous antibiotics, fluids, and electrolyte repletion.
Differential Diagnosis
There are multiple diagnoses to consider in the setting of flaccid bullae in the pediatric population. Stevens-Johnson syndrome or toxic epidermal necrolysis also can present with fever and superficial desquamation or bullae; however, exposure to medications and mucosal involvement often are absent in SSSS (Figure 2).2 Pemphigus, particularly paraneoplastic pemphigus, also often includes mucosal involvement and scalding thermal burns that are often geometric or focal. Epidermolysis bullosa and toxic shock syndrome also should be considered.1
Impetigo
Presentation
Impetigo is the most common bacterial skin infection in children caused by S aureus or Streptococcus pyogenes.7-9 It begins as erythematous papules transitioning to thin-walled vesicles that rapidly rupture and result in honey-crusted papules.7,9,10 Individuals of any age can be affected by nonbullous impetigo, but it is the most common skin infection in children aged 2 to 5 years.7
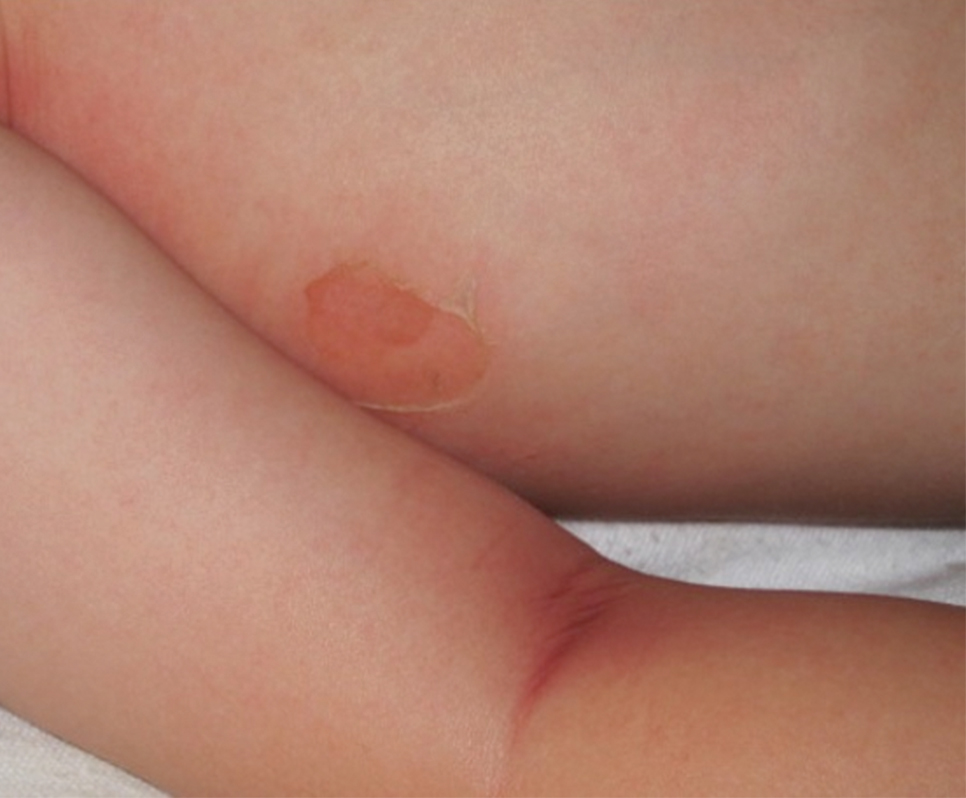
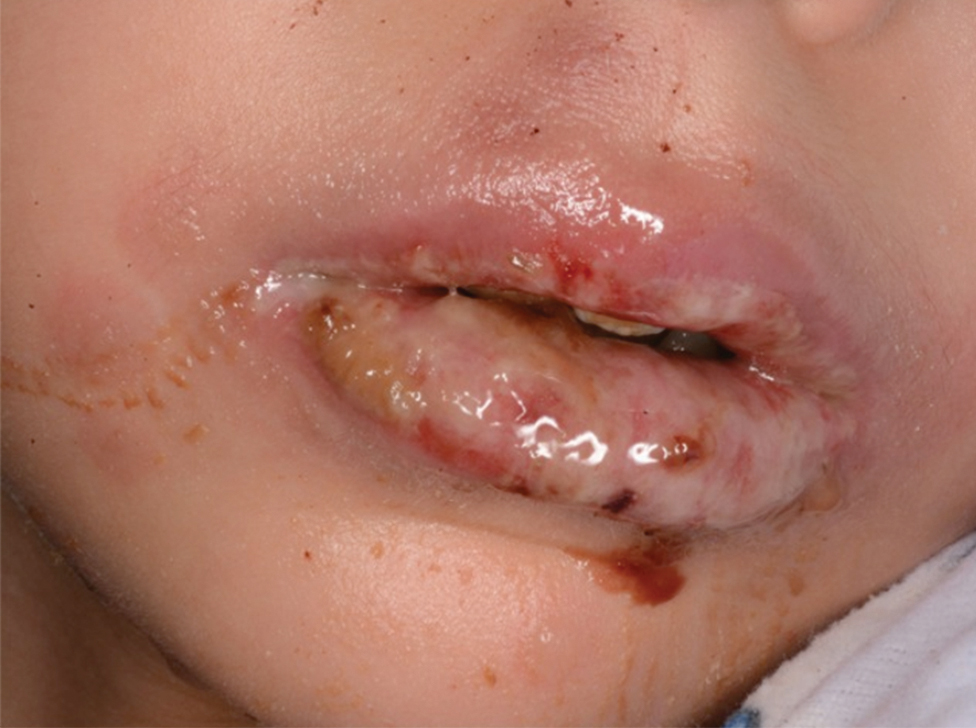
Bullous impetigo primarily is seen in children, especially infants, and rarely can occur in teenagers or adults.7 It most commonly is caused by the exfoliative toxins of S aureus. Bullous impetigo presents as small vesicles that may converge into larger flaccid bullae or pustules.7-10 Once the bullae rupture, an erythematous base with a collarette of scale remains without the formation of a honey-colored crust.8 Bullous impetigo usually affects moist intertriginous areas such as the axillae, neck, and diaper area8,10 (Figure 3). Complications may result in cellulitis, septicemia, osteomyelitis, poststreptococcal glomerulonephritis associated with S pyogenes, and S aureus–induced SSSS.7-9
Diagnosis
Nonbullous and bullous impetigo are largely clinical diagnoses that can be confirmed by culture of a vesicle or pustular fluid.10 Treatment of impetigo includes topical or systemic antibiotics.7,10 Patients should be advised to keep lesions covered and avoid contact with others until all lesions resolve, as lesions are contagious.9
Eczema Herpeticum
Presentation
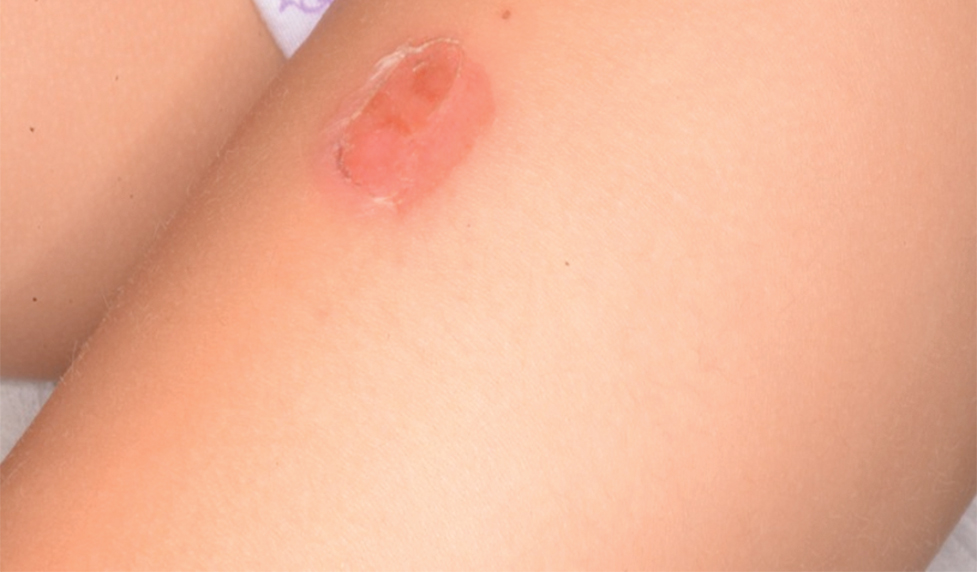
Eczema herpeticum (EH), also known as Kaposi varicelliform eruption, is a disseminated herpes simplex virus infection of impaired skin, most commonly in patients with atopic dermatitis (AD).11 Eczema herpeticum presents as a widespread eruption of erythematous monomorphic vesicles that progress to punched-out erosions with hemorrhagic crusting (Figure 4). Patients may have associated fever or lymphadenopathy.12,13
Causes
The number of children hospitalized annually for EH in the United States is approximately 4 to 7 cases per million children. Less than 3% of pediatric AD patients are affected, with a particularly increased risk in patients with severe and earlier-onset AD.12-15 Patients with AD have skin barrier defects, and decreased IFN-γ expression and cathelicidins predispose patients with AD to developing EH.12,16,17
Diagnosis
Viral polymerase chain reaction for herpes simplex virus types 1 and 2 is the standard for confirmatory diagnosis. Herpes simplex virus cultures from cutaneous scrapings, direct fluorescent antibody testing, or Tzanck test revealing multinucleated giant cells also may help establish the diagnosis.11,12,17
Management
Individuals with severe AD and other dermatologic conditions with cutaneous barrier compromise are at risk for developing EH, which is a medical emergency requiring hospitalization and prompt treatment with antiviral therapy such as acyclovir, often intravenously, as death can result if left untreated.11,17 Topical or systemic antibiotic therapy should be initiated if there is suspicion for secondary bacterial superinfection. Patients should be evaluated for multiorgan involvement such as keratoconjunctivitis, meningitis, encephalitis, and systemic viremia due to increased mortality, especially in infants.12,15,16
Langerhans Cell Histiocytosis
Presentation
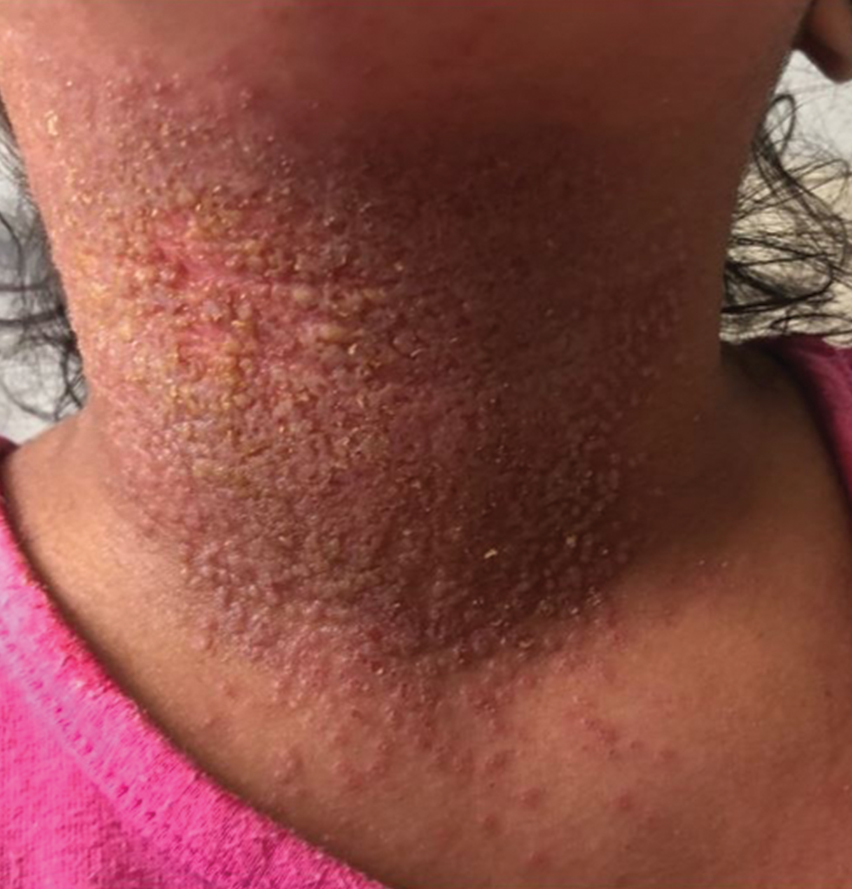
Langerhans cell histiocytosis (LCH) has a variable clinical presentation and can involve a single or multiple organ systems, including the bones and skin. Cutaneous LCH can present as violaceous papules, nodules, or ulcerations and crusted erosions (Figure 5). The lymph nodes, liver, spleen, oral mucosa, and respiratory and central nervous systems also may be involved.
Langerhans cell histiocytosis affects individuals of any age group but more often is seen in pediatric patients. The incidence of LCH is approximately 4.6 cases per million children.18 The pathogenesis is secondary to pathologic Langerhans cells, characterized as a clonal myeloid malignancy and dysregulation of the immune system.18,19
Diagnosis
A thorough physical examination is essential in patients with suspected LCH. Additionally, diagnosis of LCH is heavily based on histopathology of tissue from the involved organ system(s) with features of positive S-100 protein, CD1a, and CD207, and identification of Birbeck granules.20 Imaging and laboratory studies also are indicated and can include a skeletal survey (to assess osteolytic and organ involvement), a complete hematologic panel, coagulation studies, and liver function tests.18,21
Management
Management of LCH varies based on the organ system(s) involved along with the extent of the disease. Dermatology referral may be indicated in patients presenting with nonresolving cutaneous lesions as well as in severe cases. Single-organ and multisystem disease may require one treatment modality or a combination of chemotherapy, surgery, radiation, and/or immunotherapy.21
Infantile Hemangioma
Presentation
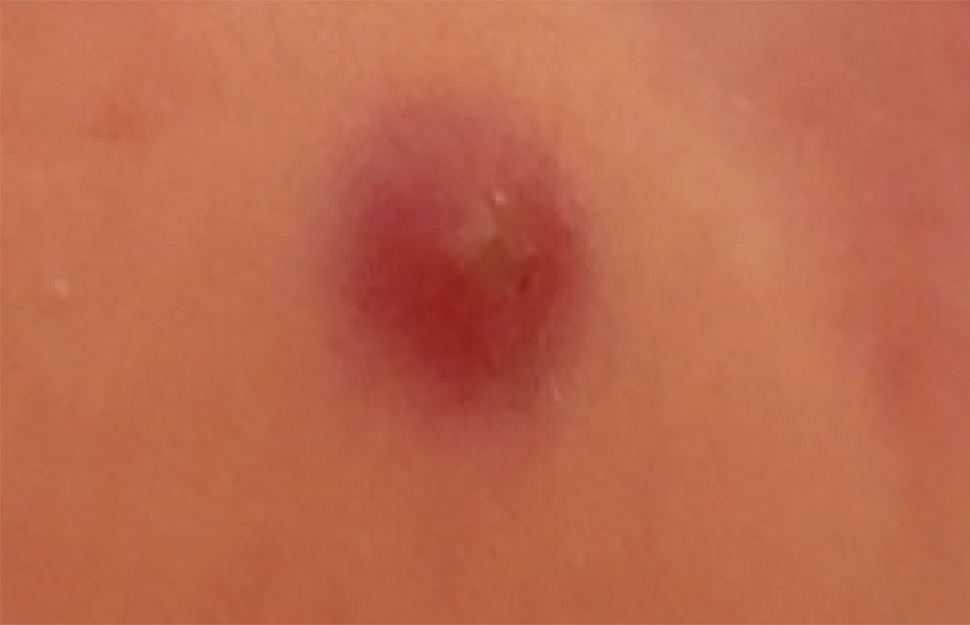
Infantile hemangioma (IH) is the most common benign tumor of infancy and usually is apparent a few weeks after birth. Lesions appear as bright red papules, nodules, or plaques. Deep or subcutaneous lesions present as raised, flesh-colored nodules with a blue hue and bruiselike appearance with or without a central patch of telangiectasia22-24 (Figure 6). Although all IHs eventually resolve, residual skin changes such as scarring, atrophy, and fibrosis can persist.24

The incidence of IH has been reported to occur in up to 4% to 5% of infants in the United States.23,25 Infantile hemangiomas also have been found to be more common among white, preterm, and multiple-gestation infants.25 The proposed pathogenesis of IHs includes angiogenic and vasogenic factors that cause rapid proliferation of blood vessels, likely driven by tissue hypoxia.23,26,27
Diagnosis
Infantile hemangioma is diagnosed clinically; however, immunohistochemical staining showing positivity for glucose transporter 1 also is helpful.26,27 Imaging modalities such as ultrasonography and magnetic resonance imaging also can be utilized to visualize the extent of lesions if necessary.25
Management
Around 15% to 25% of IHs are considered complicated and require intervention.25,27 Infantile hemangiomas can interfere with function depending on location or have potentially fatal complications. Based on the location and extent of involvement, these findings can include ulceration; hemorrhage; impairment of feeding, hearing, and/or vision; facial deformities; airway obstruction; hypothyroidism; and congestive heart failure.25,28 Early treatment with topical or oral beta-blockers is imperative for potentially life-threatening IHs, which can be seen due to large size or dangerous location.28,29 Because the rapid proliferative phase of IHs is thought to begin around 6 weeks of life, treatment should be initiated as early as possible. Initiation of beta-blocker therapy in the first few months of life can prevent functional impairment, ulceration, and permanent cosmetic changes. Additionally, surgery or pulsed dye laser treatment have been found to be effective for skin changes found after involution of IH.25,29
Differential Diagnosis
The differential diagnosis for IH includes vascular malformations, which are present at birth and do not undergo rapid proliferation; sarcoma; and kaposiform hemangioendothelioma, which causes the Kasabach-Merritt phenomenon secondary to platelet trapping. Careful attention to the history of the skin lesion provides good support for diagnosis of IH in most cases.
IgA Vasculitis
Presentation
IgA vasculitis, or Henoch-Schönlein purpura, classically presents as a tetrad of palpable purpura, acute-onset arthritis or arthralgia, abdominal pain, and renal disease with proteinuria or hematuria.30 Skin involvement is seen in almost all cases and is essential for diagnosis of IgA vasculitis. The initial dermatosis may be pruritic and present as an erythematous macular or urticarial wheal that evolves into petechiae, along with palpable purpura that is most frequently located on the legs or buttocks (Figure 7).30-34
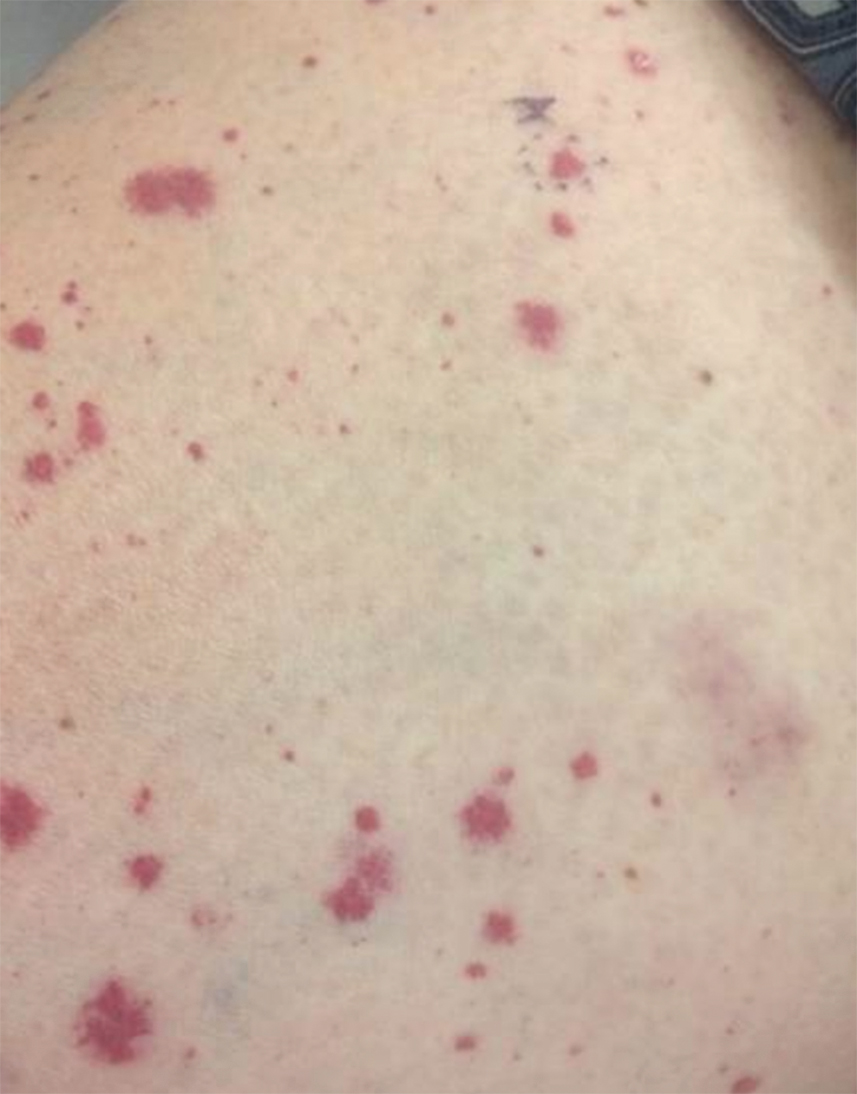
IgA vasculitis is an immune-mediated small vessel vasculitis with deposition of IgA in the small vessels. The underlying cause remains unknown, though infection, dietary allergens, drugs, vaccinations, and chemical triggers have been recognized in literature.32,35,36 IgA vasculitis is largely a pediatric diagnosis, with 90% of affected individuals younger than 10 years worldwide.37 In the pediatric population, the incidence has been reported to be 3 to 26.7 cases per 100,000 children.32
Diagnosis
Diagnosis is based on the clinical presentation and histopathology.30 On direct immunofluorescence, IgA deposition is seen in the vessel walls.35 Laboratory testing is not diagnostic, but urinalysis is mandatory to identify involvement of renal vasculature. Imaging studies may be used in patients with abdominal symptoms, as an ultrasound can be used to visualize bowel structure and abnormalities such as intussusception.33
Management
The majority of cases of IgA vasculitis recover spontaneously, with patients requiring hospital admission based on severity of symptoms.30 The primary approach to management involves providing supportive care including hydration, adequate rest, and symptomatic pain relief of the joints and abdomen with oral analgesics. Systemic corticosteroids or steroid-sparing agents such as dapsone or colchicine can be used to treat cutaneous manifestations in addition to severe pain symptoms.30,31 Patients with IgA vasculitis must be monitored for proteinuria or hematuria to assess the extent of renal involvement. Although much more common in adults, long-term renal impairment can result from childhood cases of IgA vasculitis.34
Final Thoughts
Pediatric dermatology emergencies can be difficult to detect and accurately diagnose. Many of these diseases are potential emergencies that that may result in delayed treatment and considerable morbidity and mortality if missed. Clinicians should be aware that timely recognition and diagnosis, along with possible referral to pediatric dermatology, are essential to avoid complications.
- Leung AKC, Barankin B, Leong KF. Staphylococcal-scalded skin syndrome: evaluation, diagnosis, and management. World J Pediatr. 2018;14:116-120.
- Handler MZ, Schwartz RA. Staphylococcal scalded skin syndrome: diagnosis and management in children and adults. J Eur Acad Dermatol Venereol. 2014;28:1418-1423.
- Davidson J, Polly S, Hayes P, et al. Recurrent staphylococcal scalded skin syndrome in an extremely low-birth-weight neonate. AJP Rep. 2017;7:E134-E137.
- Mishra AK, Yadav P, Mishra A. A systemic review on staphylococcal scalded skin syndrome (SSSS): a rare and critical disease of neonates. Open Microbiol J. 2016;10:150-159.
- Berk D. Staphylococcal scalded skin syndrome. Cancer Therapy Advisor website. https://www.cancertherapyadvisor.com/home/decision-support-in-medicine/pediatrics/staphylococcal-scalded-skin-syndrome/. Published 2017. Accessed February 19, 2020.
- Sakr A, Brégeon F, Mège JL, et al. Staphylococcus aureus nasal colonization: an update on mechanisms, epidemiology, risk factors, and subsequent infections [published online October 8, 2018]. Front Microbiol. 2018;9:2419.
- Pereira LB. Impetigo review. An Bras Dermatol. 2014;89:293-299.
- Nardi NM, Schaefer TJ. Impetigo. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2019. https://www.ncbi.nlm.nih.gov/books/NBK430974/. Accessed February 21, 2020.
- Koning S, van der Sande R, Verhagen AP, et al. Interventions for impetigo. Cochrane Database Syst Rev. 2012;1:CD003261.
- Sommer LL, Reboli AC, Heymann WR. Bacterial diseases. In: Bolognia, JL Schaffer, JV Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2018:1259-1295.
- Micali G, Lacarrubba F. Eczema herpeticum. N Engl J Med. 2017;377:e9.
- Leung DY. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum—a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients [published online November 16, 2019]. J Eur Acad Dermatology Venereol. doi:10.1111/jdv.16090.
- Sun D, Ong PY. Infectious complications in atopic dermatitis. Immunol Allergy Clin North Am. 2017;37:75-93.
- Hsu DY, Shinkai K, Silverberg JI. Epidemiology of eczema herpeticum in hospitalized U.S. children: analysis of a nationwide cohort [published online September 17, 2018]. J Invest Dermatol. 2018;138:265-272.
- Leung DY, Gao PS, Grigoryev DN, et al. Human atopic dermatitis complicated by eczema herpeticum is associated with abnormalities in IFN-γ response. J Allergy Clin Immunol. 2011;127:965-73.e1-5.
- Darji K, Frisch S, Adjei Boakye E, et al. Characterization of children with recurrent eczema herpeticum and response to treatment with interferon-gamma. Pediatr Dermatol. 2017;34:686-689.
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Abla O, Weitzman S. Treatment of Langerhans cell histiocytosis: role of BRAF/MAPK inhibition. Hematology Am Soc Hematol Educ Program. 2015;2015:565-570.
- Allen CE, Li L, Peters TL, et al. Cell-specific gene expression in Langerhans cell histiocytosis lesions reveals a distinct profile compared with epidermal Langerhans cells. J Immunol. 2010;184:4557-4567.
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184.
- Holland KE, Drolet BA. Infantile hemangioma [published online August 21, 2010]. Pediatr Clin North Am. 2010;57:1069-1083.
- Chen TS, Eichenfield LF, Friedlander SF. Infantile hemangiomas: an update on pathogenesis and therapy. Pediatrics. 2013;131:99-108.
- George A, Mani V, Noufal A. Update on the classification of hemangioma. J Oral Maxillofac Pathol. 2014;18(suppl 1):S117-S120.
- Darrow DH, Greene AK, Mancini AJ, et al. Diagnosis and management of infantile hemangioma. Pediatrics. 2015;136:786-791.
- Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170:907-913.
- de Jong S, Itinteang T, Withers AH, et al. Does hypoxia play a role in infantile hemangioma? Arch Dermatol Res. 2016;308:219-227.
- Hogeling M, Adams S, Wargon O. A randomized controlled trial of propranolol for infantile hemangiomas. Pediatrics. 2011;128:E259-E266.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas [published online January 2019]. Pediatrics. doi:10.1542/peds.2018-3475.
- Sohagia AB, Gunturu SG, Tong TR, et al. Henoch-Schönlein purpura—a case report and review of the literature [published online May 23, 2010]. Gastroenterol Res Pract. doi:10.1155/2010/597648.
- Rigante D, Castellazzi L, Bosco A, et al. Is there a crossroad between infections, genetics, and Henoch-Schönlein purpura? Autoimmun Rev. 2013;12:1016-1021.
- Piram M, Mahr A. Epidemiology of immunoglobulin A vasculitis (Henoch–Schönlein): current state of knowledge. Curr Opin Rheumatol. 2013;25:171-178.
- Carlson JA. The histological assessment of cutaneous vasculitis. Histopathology. 2010;56:3-23.
- Eleftheriou D, Batu ED, Ozen S, et al. Vasculitis in children. Nephrol Dial Transplant. 2014;30:I94-I103.
- van Timmeren MM, Heeringa P, Kallenberg CG. Infectious triggers for vasculitis. Curr Opin Rheumatol. 2014;26:416-423.
- Scott DGI, Watts RA. Epidemiology and clinical features of systemic vasculitis [published online July 11, 2013]. Clin Exp Nephrol. 2013;17:607-610.
- He X, Yu C, Zhao P, et al. The genetics of Henoch-Schönlein purpura: a systematic review and meta-analysis. Rheumatol Int. 2013;33:1387-1395.
Many pediatric skin conditions can be safely monitored with minimal intervention, but certain skin conditions are emergent and require immediate attention and proper assessment of the neonate, infant, or child. The skin may provide the first presentation of a potentially fatal disease with serious sequelae. Cutaneous findings may indicate the need for further evaluation. Therefore, it is important to differentiate skin conditions with benign etiologies from those that require immediate diagnosis and treatment, as early intervention of some of these conditions can be lifesaving. Herein, we discuss pertinent pediatric dermatology emergencies that dermatologists should keep in mind so that these diagnoses are never missed.
Staphylococcal Scalded Skin Syndrome
Presentation
Staphylococcal scalded skin syndrome (SSSS), or Ritter disease, is a potentially fatal pediatric emergency, especially in newborns.1 The mortality rate for SSSS in the United States is 3.6% to 11% in children.2 It typically presents with a prodrome of tenderness, fever, and confluent erythematous patches on the folds of the skin such as the groin, axillae, nose, and ears, with eventual spread to the legs and trunk.1,2 Within 24 to 48 hours of symptom onset, blistering and fluid accumulation will appear diffusely. Bullae are flaccid, and tangential and gentle pressure on involved unblistered skin may lead to shearing of the epithelium, which is a positive Nikolsky sign.1,2
Causes
Staphylococcal scalded skin syndrome is caused by exfoliative toxins A and B, toxigenic strains of Staphylococcus aureus. Exfoliative toxins A and B are serine proteases that target and cleave desmoglein 1, which binds keratinocytes in the stratum granulosum.1,3 Exfoliative toxins disrupt the adhesion of keratinocytes, resulting in bullae formation and subsequently diffuse sheetlike desquamation.1,4,5 Although up to 30% of the human population are asymptomatically and permanently colonized with nasal S aureus,6 the exfoliative toxins are produced by only 5% of species.1
In neonates, the immune and renal systems are underdeveloped; therefore, patients are susceptible to SSSS due to lack of neutralizing antibodies and decreased renal toxin excretion.4 Potential complications of SSSS are deeper soft-tissue infection, septicemia (blood-borne infection), and fluid and electrolyte imbalance.1,4
Diagnosis and Treatment
The condition is diagnosed clinically based on the findings of tender erythroderma, bullae, and desquamation with a scalded appearance, especially in friction zones; periorificial crusting; positive Nikolsky sign; and lack of mucosal involvement (Figure 1).1 Histopathology can aid in complicated clinical scenarios as well as culture from affected areas, including the upper respiratory tract, diaper region, and umbilicus.1,4 Hospitalization is required for SSSS for intravenous antibiotics, fluids, and electrolyte repletion.

Differential Diagnosis
There are multiple diagnoses to consider in the setting of flaccid bullae in the pediatric population. Stevens-Johnson syndrome or toxic epidermal necrolysis also can present with fever and superficial desquamation or bullae; however, exposure to medications and mucosal involvement often are absent in SSSS (Figure 2).2 Pemphigus, particularly paraneoplastic pemphigus, also often includes mucosal involvement and scalding thermal burns that are often geometric or focal. Epidermolysis bullosa and toxic shock syndrome also should be considered.1

Impetigo
Presentation
Impetigo is the most common bacterial skin infection in children caused by S aureus or Streptococcus pyogenes.7-9 It begins as erythematous papules transitioning to thin-walled vesicles that rapidly rupture and result in honey-crusted papules.7,9,10 Individuals of any age can be affected by nonbullous impetigo, but it is the most common skin infection in children aged 2 to 5 years.7
Bullous impetigo primarily is seen in children, especially infants, and rarely can occur in teenagers or adults.7 It most commonly is caused by the exfoliative toxins of S aureus. Bullous impetigo presents as small vesicles that may converge into larger flaccid bullae or pustules.7-10 Once the bullae rupture, an erythematous base with a collarette of scale remains without the formation of a honey-colored crust.8 Bullous impetigo usually affects moist intertriginous areas such as the axillae, neck, and diaper area8,10 (Figure 3). Complications may result in cellulitis, septicemia, osteomyelitis, poststreptococcal glomerulonephritis associated with S pyogenes, and S aureus–induced SSSS.7-9

Diagnosis
Nonbullous and bullous impetigo are largely clinical diagnoses that can be confirmed by culture of a vesicle or pustular fluid.10 Treatment of impetigo includes topical or systemic antibiotics.7,10 Patients should be advised to keep lesions covered and avoid contact with others until all lesions resolve, as lesions are contagious.9
Eczema Herpeticum
Presentation
Eczema herpeticum (EH), also known as Kaposi varicelliform eruption, is a disseminated herpes simplex virus infection of impaired skin, most commonly in patients with atopic dermatitis (AD).11 Eczema herpeticum presents as a widespread eruption of erythematous monomorphic vesicles that progress to punched-out erosions with hemorrhagic crusting (Figure 4). Patients may have associated fever or lymphadenopathy.12,13

Causes
The number of children hospitalized annually for EH in the United States is approximately 4 to 7 cases per million children. Less than 3% of pediatric AD patients are affected, with a particularly increased risk in patients with severe and earlier-onset AD.12-15 Patients with AD have skin barrier defects, and decreased IFN-γ expression and cathelicidins predispose patients with AD to developing EH.12,16,17
Diagnosis
Viral polymerase chain reaction for herpes simplex virus types 1 and 2 is the standard for confirmatory diagnosis. Herpes simplex virus cultures from cutaneous scrapings, direct fluorescent antibody testing, or Tzanck test revealing multinucleated giant cells also may help establish the diagnosis.11,12,17
Management
Individuals with severe AD and other dermatologic conditions with cutaneous barrier compromise are at risk for developing EH, which is a medical emergency requiring hospitalization and prompt treatment with antiviral therapy such as acyclovir, often intravenously, as death can result if left untreated.11,17 Topical or systemic antibiotic therapy should be initiated if there is suspicion for secondary bacterial superinfection. Patients should be evaluated for multiorgan involvement such as keratoconjunctivitis, meningitis, encephalitis, and systemic viremia due to increased mortality, especially in infants.12,15,16
Langerhans Cell Histiocytosis
Presentation
Langerhans cell histiocytosis (LCH) has a variable clinical presentation and can involve a single or multiple organ systems, including the bones and skin. Cutaneous LCH can present as violaceous papules, nodules, or ulcerations and crusted erosions (Figure 5). The lymph nodes, liver, spleen, oral mucosa, and respiratory and central nervous systems also may be involved.

Langerhans cell histiocytosis affects individuals of any age group but more often is seen in pediatric patients. The incidence of LCH is approximately 4.6 cases per million children.18 The pathogenesis is secondary to pathologic Langerhans cells, characterized as a clonal myeloid malignancy and dysregulation of the immune system.18,19
Diagnosis
A thorough physical examination is essential in patients with suspected LCH. Additionally, diagnosis of LCH is heavily based on histopathology of tissue from the involved organ system(s) with features of positive S-100 protein, CD1a, and CD207, and identification of Birbeck granules.20 Imaging and laboratory studies also are indicated and can include a skeletal survey (to assess osteolytic and organ involvement), a complete hematologic panel, coagulation studies, and liver function tests.18,21
Management
Management of LCH varies based on the organ system(s) involved along with the extent of the disease. Dermatology referral may be indicated in patients presenting with nonresolving cutaneous lesions as well as in severe cases. Single-organ and multisystem disease may require one treatment modality or a combination of chemotherapy, surgery, radiation, and/or immunotherapy.21
Infantile Hemangioma
Presentation
Infantile hemangioma (IH) is the most common benign tumor of infancy and usually is apparent a few weeks after birth. Lesions appear as bright red papules, nodules, or plaques. Deep or subcutaneous lesions present as raised, flesh-colored nodules with a blue hue and bruiselike appearance with or without a central patch of telangiectasia22-24 (Figure 6). Although all IHs eventually resolve, residual skin changes such as scarring, atrophy, and fibrosis can persist.24

The incidence of IH has been reported to occur in up to 4% to 5% of infants in the United States.23,25 Infantile hemangiomas also have been found to be more common among white, preterm, and multiple-gestation infants.25 The proposed pathogenesis of IHs includes angiogenic and vasogenic factors that cause rapid proliferation of blood vessels, likely driven by tissue hypoxia.23,26,27
Diagnosis
Infantile hemangioma is diagnosed clinically; however, immunohistochemical staining showing positivity for glucose transporter 1 also is helpful.26,27 Imaging modalities such as ultrasonography and magnetic resonance imaging also can be utilized to visualize the extent of lesions if necessary.25
Management
Around 15% to 25% of IHs are considered complicated and require intervention.25,27 Infantile hemangiomas can interfere with function depending on location or have potentially fatal complications. Based on the location and extent of involvement, these findings can include ulceration; hemorrhage; impairment of feeding, hearing, and/or vision; facial deformities; airway obstruction; hypothyroidism; and congestive heart failure.25,28 Early treatment with topical or oral beta-blockers is imperative for potentially life-threatening IHs, which can be seen due to large size or dangerous location.28,29 Because the rapid proliferative phase of IHs is thought to begin around 6 weeks of life, treatment should be initiated as early as possible. Initiation of beta-blocker therapy in the first few months of life can prevent functional impairment, ulceration, and permanent cosmetic changes. Additionally, surgery or pulsed dye laser treatment have been found to be effective for skin changes found after involution of IH.25,29
Differential Diagnosis
The differential diagnosis for IH includes vascular malformations, which are present at birth and do not undergo rapid proliferation; sarcoma; and kaposiform hemangioendothelioma, which causes the Kasabach-Merritt phenomenon secondary to platelet trapping. Careful attention to the history of the skin lesion provides good support for diagnosis of IH in most cases.
IgA Vasculitis
Presentation
IgA vasculitis, or Henoch-Schönlein purpura, classically presents as a tetrad of palpable purpura, acute-onset arthritis or arthralgia, abdominal pain, and renal disease with proteinuria or hematuria.30 Skin involvement is seen in almost all cases and is essential for diagnosis of IgA vasculitis. The initial dermatosis may be pruritic and present as an erythematous macular or urticarial wheal that evolves into petechiae, along with palpable purpura that is most frequently located on the legs or buttocks (Figure 7).30-34

IgA vasculitis is an immune-mediated small vessel vasculitis with deposition of IgA in the small vessels. The underlying cause remains unknown, though infection, dietary allergens, drugs, vaccinations, and chemical triggers have been recognized in literature.32,35,36 IgA vasculitis is largely a pediatric diagnosis, with 90% of affected individuals younger than 10 years worldwide.37 In the pediatric population, the incidence has been reported to be 3 to 26.7 cases per 100,000 children.32
Diagnosis
Diagnosis is based on the clinical presentation and histopathology.30 On direct immunofluorescence, IgA deposition is seen in the vessel walls.35 Laboratory testing is not diagnostic, but urinalysis is mandatory to identify involvement of renal vasculature. Imaging studies may be used in patients with abdominal symptoms, as an ultrasound can be used to visualize bowel structure and abnormalities such as intussusception.33
Management
The majority of cases of IgA vasculitis recover spontaneously, with patients requiring hospital admission based on severity of symptoms.30 The primary approach to management involves providing supportive care including hydration, adequate rest, and symptomatic pain relief of the joints and abdomen with oral analgesics. Systemic corticosteroids or steroid-sparing agents such as dapsone or colchicine can be used to treat cutaneous manifestations in addition to severe pain symptoms.30,31 Patients with IgA vasculitis must be monitored for proteinuria or hematuria to assess the extent of renal involvement. Although much more common in adults, long-term renal impairment can result from childhood cases of IgA vasculitis.34
Final Thoughts
Pediatric dermatology emergencies can be difficult to detect and accurately diagnose. Many of these diseases are potential emergencies that that may result in delayed treatment and considerable morbidity and mortality if missed. Clinicians should be aware that timely recognition and diagnosis, along with possible referral to pediatric dermatology, are essential to avoid complications.
Many pediatric skin conditions can be safely monitored with minimal intervention, but certain skin conditions are emergent and require immediate attention and proper assessment of the neonate, infant, or child. The skin may provide the first presentation of a potentially fatal disease with serious sequelae. Cutaneous findings may indicate the need for further evaluation. Therefore, it is important to differentiate skin conditions with benign etiologies from those that require immediate diagnosis and treatment, as early intervention of some of these conditions can be lifesaving. Herein, we discuss pertinent pediatric dermatology emergencies that dermatologists should keep in mind so that these diagnoses are never missed.
Staphylococcal Scalded Skin Syndrome
Presentation
Staphylococcal scalded skin syndrome (SSSS), or Ritter disease, is a potentially fatal pediatric emergency, especially in newborns.1 The mortality rate for SSSS in the United States is 3.6% to 11% in children.2 It typically presents with a prodrome of tenderness, fever, and confluent erythematous patches on the folds of the skin such as the groin, axillae, nose, and ears, with eventual spread to the legs and trunk.1,2 Within 24 to 48 hours of symptom onset, blistering and fluid accumulation will appear diffusely. Bullae are flaccid, and tangential and gentle pressure on involved unblistered skin may lead to shearing of the epithelium, which is a positive Nikolsky sign.1,2
Causes
Staphylococcal scalded skin syndrome is caused by exfoliative toxins A and B, toxigenic strains of Staphylococcus aureus. Exfoliative toxins A and B are serine proteases that target and cleave desmoglein 1, which binds keratinocytes in the stratum granulosum.1,3 Exfoliative toxins disrupt the adhesion of keratinocytes, resulting in bullae formation and subsequently diffuse sheetlike desquamation.1,4,5 Although up to 30% of the human population are asymptomatically and permanently colonized with nasal S aureus,6 the exfoliative toxins are produced by only 5% of species.1
In neonates, the immune and renal systems are underdeveloped; therefore, patients are susceptible to SSSS due to lack of neutralizing antibodies and decreased renal toxin excretion.4 Potential complications of SSSS are deeper soft-tissue infection, septicemia (blood-borne infection), and fluid and electrolyte imbalance.1,4
Diagnosis and Treatment
The condition is diagnosed clinically based on the findings of tender erythroderma, bullae, and desquamation with a scalded appearance, especially in friction zones; periorificial crusting; positive Nikolsky sign; and lack of mucosal involvement (Figure 1).1 Histopathology can aid in complicated clinical scenarios as well as culture from affected areas, including the upper respiratory tract, diaper region, and umbilicus.1,4 Hospitalization is required for SSSS for intravenous antibiotics, fluids, and electrolyte repletion.

Differential Diagnosis
There are multiple diagnoses to consider in the setting of flaccid bullae in the pediatric population. Stevens-Johnson syndrome or toxic epidermal necrolysis also can present with fever and superficial desquamation or bullae; however, exposure to medications and mucosal involvement often are absent in SSSS (Figure 2).2 Pemphigus, particularly paraneoplastic pemphigus, also often includes mucosal involvement and scalding thermal burns that are often geometric or focal. Epidermolysis bullosa and toxic shock syndrome also should be considered.1

Impetigo
Presentation
Impetigo is the most common bacterial skin infection in children caused by S aureus or Streptococcus pyogenes.7-9 It begins as erythematous papules transitioning to thin-walled vesicles that rapidly rupture and result in honey-crusted papules.7,9,10 Individuals of any age can be affected by nonbullous impetigo, but it is the most common skin infection in children aged 2 to 5 years.7
Bullous impetigo primarily is seen in children, especially infants, and rarely can occur in teenagers or adults.7 It most commonly is caused by the exfoliative toxins of S aureus. Bullous impetigo presents as small vesicles that may converge into larger flaccid bullae or pustules.7-10 Once the bullae rupture, an erythematous base with a collarette of scale remains without the formation of a honey-colored crust.8 Bullous impetigo usually affects moist intertriginous areas such as the axillae, neck, and diaper area8,10 (Figure 3). Complications may result in cellulitis, septicemia, osteomyelitis, poststreptococcal glomerulonephritis associated with S pyogenes, and S aureus–induced SSSS.7-9

Diagnosis
Nonbullous and bullous impetigo are largely clinical diagnoses that can be confirmed by culture of a vesicle or pustular fluid.10 Treatment of impetigo includes topical or systemic antibiotics.7,10 Patients should be advised to keep lesions covered and avoid contact with others until all lesions resolve, as lesions are contagious.9
Eczema Herpeticum
Presentation
Eczema herpeticum (EH), also known as Kaposi varicelliform eruption, is a disseminated herpes simplex virus infection of impaired skin, most commonly in patients with atopic dermatitis (AD).11 Eczema herpeticum presents as a widespread eruption of erythematous monomorphic vesicles that progress to punched-out erosions with hemorrhagic crusting (Figure 4). Patients may have associated fever or lymphadenopathy.12,13

Causes
The number of children hospitalized annually for EH in the United States is approximately 4 to 7 cases per million children. Less than 3% of pediatric AD patients are affected, with a particularly increased risk in patients with severe and earlier-onset AD.12-15 Patients with AD have skin barrier defects, and decreased IFN-γ expression and cathelicidins predispose patients with AD to developing EH.12,16,17
Diagnosis
Viral polymerase chain reaction for herpes simplex virus types 1 and 2 is the standard for confirmatory diagnosis. Herpes simplex virus cultures from cutaneous scrapings, direct fluorescent antibody testing, or Tzanck test revealing multinucleated giant cells also may help establish the diagnosis.11,12,17
Management
Individuals with severe AD and other dermatologic conditions with cutaneous barrier compromise are at risk for developing EH, which is a medical emergency requiring hospitalization and prompt treatment with antiviral therapy such as acyclovir, often intravenously, as death can result if left untreated.11,17 Topical or systemic antibiotic therapy should be initiated if there is suspicion for secondary bacterial superinfection. Patients should be evaluated for multiorgan involvement such as keratoconjunctivitis, meningitis, encephalitis, and systemic viremia due to increased mortality, especially in infants.12,15,16
Langerhans Cell Histiocytosis
Presentation
Langerhans cell histiocytosis (LCH) has a variable clinical presentation and can involve a single or multiple organ systems, including the bones and skin. Cutaneous LCH can present as violaceous papules, nodules, or ulcerations and crusted erosions (Figure 5). The lymph nodes, liver, spleen, oral mucosa, and respiratory and central nervous systems also may be involved.

Langerhans cell histiocytosis affects individuals of any age group but more often is seen in pediatric patients. The incidence of LCH is approximately 4.6 cases per million children.18 The pathogenesis is secondary to pathologic Langerhans cells, characterized as a clonal myeloid malignancy and dysregulation of the immune system.18,19
Diagnosis
A thorough physical examination is essential in patients with suspected LCH. Additionally, diagnosis of LCH is heavily based on histopathology of tissue from the involved organ system(s) with features of positive S-100 protein, CD1a, and CD207, and identification of Birbeck granules.20 Imaging and laboratory studies also are indicated and can include a skeletal survey (to assess osteolytic and organ involvement), a complete hematologic panel, coagulation studies, and liver function tests.18,21
Management
Management of LCH varies based on the organ system(s) involved along with the extent of the disease. Dermatology referral may be indicated in patients presenting with nonresolving cutaneous lesions as well as in severe cases. Single-organ and multisystem disease may require one treatment modality or a combination of chemotherapy, surgery, radiation, and/or immunotherapy.21
Infantile Hemangioma
Presentation
Infantile hemangioma (IH) is the most common benign tumor of infancy and usually is apparent a few weeks after birth. Lesions appear as bright red papules, nodules, or plaques. Deep or subcutaneous lesions present as raised, flesh-colored nodules with a blue hue and bruiselike appearance with or without a central patch of telangiectasia22-24 (Figure 6). Although all IHs eventually resolve, residual skin changes such as scarring, atrophy, and fibrosis can persist.24

The incidence of IH has been reported to occur in up to 4% to 5% of infants in the United States.23,25 Infantile hemangiomas also have been found to be more common among white, preterm, and multiple-gestation infants.25 The proposed pathogenesis of IHs includes angiogenic and vasogenic factors that cause rapid proliferation of blood vessels, likely driven by tissue hypoxia.23,26,27
Diagnosis
Infantile hemangioma is diagnosed clinically; however, immunohistochemical staining showing positivity for glucose transporter 1 also is helpful.26,27 Imaging modalities such as ultrasonography and magnetic resonance imaging also can be utilized to visualize the extent of lesions if necessary.25
Management
Around 15% to 25% of IHs are considered complicated and require intervention.25,27 Infantile hemangiomas can interfere with function depending on location or have potentially fatal complications. Based on the location and extent of involvement, these findings can include ulceration; hemorrhage; impairment of feeding, hearing, and/or vision; facial deformities; airway obstruction; hypothyroidism; and congestive heart failure.25,28 Early treatment with topical or oral beta-blockers is imperative for potentially life-threatening IHs, which can be seen due to large size or dangerous location.28,29 Because the rapid proliferative phase of IHs is thought to begin around 6 weeks of life, treatment should be initiated as early as possible. Initiation of beta-blocker therapy in the first few months of life can prevent functional impairment, ulceration, and permanent cosmetic changes. Additionally, surgery or pulsed dye laser treatment have been found to be effective for skin changes found after involution of IH.25,29
Differential Diagnosis
The differential diagnosis for IH includes vascular malformations, which are present at birth and do not undergo rapid proliferation; sarcoma; and kaposiform hemangioendothelioma, which causes the Kasabach-Merritt phenomenon secondary to platelet trapping. Careful attention to the history of the skin lesion provides good support for diagnosis of IH in most cases.
IgA Vasculitis
Presentation
IgA vasculitis, or Henoch-Schönlein purpura, classically presents as a tetrad of palpable purpura, acute-onset arthritis or arthralgia, abdominal pain, and renal disease with proteinuria or hematuria.30 Skin involvement is seen in almost all cases and is essential for diagnosis of IgA vasculitis. The initial dermatosis may be pruritic and present as an erythematous macular or urticarial wheal that evolves into petechiae, along with palpable purpura that is most frequently located on the legs or buttocks (Figure 7).30-34

IgA vasculitis is an immune-mediated small vessel vasculitis with deposition of IgA in the small vessels. The underlying cause remains unknown, though infection, dietary allergens, drugs, vaccinations, and chemical triggers have been recognized in literature.32,35,36 IgA vasculitis is largely a pediatric diagnosis, with 90% of affected individuals younger than 10 years worldwide.37 In the pediatric population, the incidence has been reported to be 3 to 26.7 cases per 100,000 children.32
Diagnosis
Diagnosis is based on the clinical presentation and histopathology.30 On direct immunofluorescence, IgA deposition is seen in the vessel walls.35 Laboratory testing is not diagnostic, but urinalysis is mandatory to identify involvement of renal vasculature. Imaging studies may be used in patients with abdominal symptoms, as an ultrasound can be used to visualize bowel structure and abnormalities such as intussusception.33
Management
The majority of cases of IgA vasculitis recover spontaneously, with patients requiring hospital admission based on severity of symptoms.30 The primary approach to management involves providing supportive care including hydration, adequate rest, and symptomatic pain relief of the joints and abdomen with oral analgesics. Systemic corticosteroids or steroid-sparing agents such as dapsone or colchicine can be used to treat cutaneous manifestations in addition to severe pain symptoms.30,31 Patients with IgA vasculitis must be monitored for proteinuria or hematuria to assess the extent of renal involvement. Although much more common in adults, long-term renal impairment can result from childhood cases of IgA vasculitis.34
Final Thoughts
Pediatric dermatology emergencies can be difficult to detect and accurately diagnose. Many of these diseases are potential emergencies that that may result in delayed treatment and considerable morbidity and mortality if missed. Clinicians should be aware that timely recognition and diagnosis, along with possible referral to pediatric dermatology, are essential to avoid complications.
- Leung AKC, Barankin B, Leong KF. Staphylococcal-scalded skin syndrome: evaluation, diagnosis, and management. World J Pediatr. 2018;14:116-120.
- Handler MZ, Schwartz RA. Staphylococcal scalded skin syndrome: diagnosis and management in children and adults. J Eur Acad Dermatol Venereol. 2014;28:1418-1423.
- Davidson J, Polly S, Hayes P, et al. Recurrent staphylococcal scalded skin syndrome in an extremely low-birth-weight neonate. AJP Rep. 2017;7:E134-E137.
- Mishra AK, Yadav P, Mishra A. A systemic review on staphylococcal scalded skin syndrome (SSSS): a rare and critical disease of neonates. Open Microbiol J. 2016;10:150-159.
- Berk D. Staphylococcal scalded skin syndrome. Cancer Therapy Advisor website. https://www.cancertherapyadvisor.com/home/decision-support-in-medicine/pediatrics/staphylococcal-scalded-skin-syndrome/. Published 2017. Accessed February 19, 2020.
- Sakr A, Brégeon F, Mège JL, et al. Staphylococcus aureus nasal colonization: an update on mechanisms, epidemiology, risk factors, and subsequent infections [published online October 8, 2018]. Front Microbiol. 2018;9:2419.
- Pereira LB. Impetigo review. An Bras Dermatol. 2014;89:293-299.
- Nardi NM, Schaefer TJ. Impetigo. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2019. https://www.ncbi.nlm.nih.gov/books/NBK430974/. Accessed February 21, 2020.
- Koning S, van der Sande R, Verhagen AP, et al. Interventions for impetigo. Cochrane Database Syst Rev. 2012;1:CD003261.
- Sommer LL, Reboli AC, Heymann WR. Bacterial diseases. In: Bolognia, JL Schaffer, JV Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2018:1259-1295.
- Micali G, Lacarrubba F. Eczema herpeticum. N Engl J Med. 2017;377:e9.
- Leung DY. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum—a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients [published online November 16, 2019]. J Eur Acad Dermatology Venereol. doi:10.1111/jdv.16090.
- Sun D, Ong PY. Infectious complications in atopic dermatitis. Immunol Allergy Clin North Am. 2017;37:75-93.
- Hsu DY, Shinkai K, Silverberg JI. Epidemiology of eczema herpeticum in hospitalized U.S. children: analysis of a nationwide cohort [published online September 17, 2018]. J Invest Dermatol. 2018;138:265-272.
- Leung DY, Gao PS, Grigoryev DN, et al. Human atopic dermatitis complicated by eczema herpeticum is associated with abnormalities in IFN-γ response. J Allergy Clin Immunol. 2011;127:965-73.e1-5.
- Darji K, Frisch S, Adjei Boakye E, et al. Characterization of children with recurrent eczema herpeticum and response to treatment with interferon-gamma. Pediatr Dermatol. 2017;34:686-689.
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Abla O, Weitzman S. Treatment of Langerhans cell histiocytosis: role of BRAF/MAPK inhibition. Hematology Am Soc Hematol Educ Program. 2015;2015:565-570.
- Allen CE, Li L, Peters TL, et al. Cell-specific gene expression in Langerhans cell histiocytosis lesions reveals a distinct profile compared with epidermal Langerhans cells. J Immunol. 2010;184:4557-4567.
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184.
- Holland KE, Drolet BA. Infantile hemangioma [published online August 21, 2010]. Pediatr Clin North Am. 2010;57:1069-1083.
- Chen TS, Eichenfield LF, Friedlander SF. Infantile hemangiomas: an update on pathogenesis and therapy. Pediatrics. 2013;131:99-108.
- George A, Mani V, Noufal A. Update on the classification of hemangioma. J Oral Maxillofac Pathol. 2014;18(suppl 1):S117-S120.
- Darrow DH, Greene AK, Mancini AJ, et al. Diagnosis and management of infantile hemangioma. Pediatrics. 2015;136:786-791.
- Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170:907-913.
- de Jong S, Itinteang T, Withers AH, et al. Does hypoxia play a role in infantile hemangioma? Arch Dermatol Res. 2016;308:219-227.
- Hogeling M, Adams S, Wargon O. A randomized controlled trial of propranolol for infantile hemangiomas. Pediatrics. 2011;128:E259-E266.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas [published online January 2019]. Pediatrics. doi:10.1542/peds.2018-3475.
- Sohagia AB, Gunturu SG, Tong TR, et al. Henoch-Schönlein purpura—a case report and review of the literature [published online May 23, 2010]. Gastroenterol Res Pract. doi:10.1155/2010/597648.
- Rigante D, Castellazzi L, Bosco A, et al. Is there a crossroad between infections, genetics, and Henoch-Schönlein purpura? Autoimmun Rev. 2013;12:1016-1021.
- Piram M, Mahr A. Epidemiology of immunoglobulin A vasculitis (Henoch–Schönlein): current state of knowledge. Curr Opin Rheumatol. 2013;25:171-178.
- Carlson JA. The histological assessment of cutaneous vasculitis. Histopathology. 2010;56:3-23.
- Eleftheriou D, Batu ED, Ozen S, et al. Vasculitis in children. Nephrol Dial Transplant. 2014;30:I94-I103.
- van Timmeren MM, Heeringa P, Kallenberg CG. Infectious triggers for vasculitis. Curr Opin Rheumatol. 2014;26:416-423.
- Scott DGI, Watts RA. Epidemiology and clinical features of systemic vasculitis [published online July 11, 2013]. Clin Exp Nephrol. 2013;17:607-610.
- He X, Yu C, Zhao P, et al. The genetics of Henoch-Schönlein purpura: a systematic review and meta-analysis. Rheumatol Int. 2013;33:1387-1395.
- Leung AKC, Barankin B, Leong KF. Staphylococcal-scalded skin syndrome: evaluation, diagnosis, and management. World J Pediatr. 2018;14:116-120.
- Handler MZ, Schwartz RA. Staphylococcal scalded skin syndrome: diagnosis and management in children and adults. J Eur Acad Dermatol Venereol. 2014;28:1418-1423.
- Davidson J, Polly S, Hayes P, et al. Recurrent staphylococcal scalded skin syndrome in an extremely low-birth-weight neonate. AJP Rep. 2017;7:E134-E137.
- Mishra AK, Yadav P, Mishra A. A systemic review on staphylococcal scalded skin syndrome (SSSS): a rare and critical disease of neonates. Open Microbiol J. 2016;10:150-159.
- Berk D. Staphylococcal scalded skin syndrome. Cancer Therapy Advisor website. https://www.cancertherapyadvisor.com/home/decision-support-in-medicine/pediatrics/staphylococcal-scalded-skin-syndrome/. Published 2017. Accessed February 19, 2020.
- Sakr A, Brégeon F, Mège JL, et al. Staphylococcus aureus nasal colonization: an update on mechanisms, epidemiology, risk factors, and subsequent infections [published online October 8, 2018]. Front Microbiol. 2018;9:2419.
- Pereira LB. Impetigo review. An Bras Dermatol. 2014;89:293-299.
- Nardi NM, Schaefer TJ. Impetigo. In: StatPearls. Treasure Island, FL: StatPearls Publishing; 2019. https://www.ncbi.nlm.nih.gov/books/NBK430974/. Accessed February 21, 2020.
- Koning S, van der Sande R, Verhagen AP, et al. Interventions for impetigo. Cochrane Database Syst Rev. 2012;1:CD003261.
- Sommer LL, Reboli AC, Heymann WR. Bacterial diseases. In: Bolognia, JL Schaffer, JV Cerroni L, eds. Dermatology. 4th ed. Philadelphia, PA: Elsevier; 2018:1259-1295.
- Micali G, Lacarrubba F. Eczema herpeticum. N Engl J Med. 2017;377:e9.
- Leung DY. Why is eczema herpeticum unexpectedly rare? Antiviral Res. 2013;98:153-157.
- Seegräber M, Worm M, Werfel T, et al. Recurrent eczema herpeticum—a retrospective European multicenter study evaluating the clinical characteristics of eczema herpeticum cases in atopic dermatitis patients [published online November 16, 2019]. J Eur Acad Dermatology Venereol. doi:10.1111/jdv.16090.
- Sun D, Ong PY. Infectious complications in atopic dermatitis. Immunol Allergy Clin North Am. 2017;37:75-93.
- Hsu DY, Shinkai K, Silverberg JI. Epidemiology of eczema herpeticum in hospitalized U.S. children: analysis of a nationwide cohort [published online September 17, 2018]. J Invest Dermatol. 2018;138:265-272.
- Leung DY, Gao PS, Grigoryev DN, et al. Human atopic dermatitis complicated by eczema herpeticum is associated with abnormalities in IFN-γ response. J Allergy Clin Immunol. 2011;127:965-73.e1-5.
- Darji K, Frisch S, Adjei Boakye E, et al. Characterization of children with recurrent eczema herpeticum and response to treatment with interferon-gamma. Pediatr Dermatol. 2017;34:686-689.
- Allen CE, Merad M, McClain KL. Langerhans-cell histiocytosis. N Engl J Med. 2018;379:856-868.
- Abla O, Weitzman S. Treatment of Langerhans cell histiocytosis: role of BRAF/MAPK inhibition. Hematology Am Soc Hematol Educ Program. 2015;2015:565-570.
- Allen CE, Li L, Peters TL, et al. Cell-specific gene expression in Langerhans cell histiocytosis lesions reveals a distinct profile compared with epidermal Langerhans cells. J Immunol. 2010;184:4557-4567.
- Haupt R, Minkov M, Astigarraga I, et al. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184.
- Holland KE, Drolet BA. Infantile hemangioma [published online August 21, 2010]. Pediatr Clin North Am. 2010;57:1069-1083.
- Chen TS, Eichenfield LF, Friedlander SF. Infantile hemangiomas: an update on pathogenesis and therapy. Pediatrics. 2013;131:99-108.
- George A, Mani V, Noufal A. Update on the classification of hemangioma. J Oral Maxillofac Pathol. 2014;18(suppl 1):S117-S120.
- Darrow DH, Greene AK, Mancini AJ, et al. Diagnosis and management of infantile hemangioma. Pediatrics. 2015;136:786-791.
- Munden A, Butschek R, Tom WL, et al. Prospective study of infantile haemangiomas: incidence, clinical characteristics and association with placental anomalies. Br J Dermatol. 2014;170:907-913.
- de Jong S, Itinteang T, Withers AH, et al. Does hypoxia play a role in infantile hemangioma? Arch Dermatol Res. 2016;308:219-227.
- Hogeling M, Adams S, Wargon O. A randomized controlled trial of propranolol for infantile hemangiomas. Pediatrics. 2011;128:E259-E266.
- Krowchuk DP, Frieden IJ, Mancini AJ, et al. Clinical practice guideline for the management of infantile hemangiomas [published online January 2019]. Pediatrics. doi:10.1542/peds.2018-3475.
- Sohagia AB, Gunturu SG, Tong TR, et al. Henoch-Schönlein purpura—a case report and review of the literature [published online May 23, 2010]. Gastroenterol Res Pract. doi:10.1155/2010/597648.
- Rigante D, Castellazzi L, Bosco A, et al. Is there a crossroad between infections, genetics, and Henoch-Schönlein purpura? Autoimmun Rev. 2013;12:1016-1021.
- Piram M, Mahr A. Epidemiology of immunoglobulin A vasculitis (Henoch–Schönlein): current state of knowledge. Curr Opin Rheumatol. 2013;25:171-178.
- Carlson JA. The histological assessment of cutaneous vasculitis. Histopathology. 2010;56:3-23.
- Eleftheriou D, Batu ED, Ozen S, et al. Vasculitis in children. Nephrol Dial Transplant. 2014;30:I94-I103.
- van Timmeren MM, Heeringa P, Kallenberg CG. Infectious triggers for vasculitis. Curr Opin Rheumatol. 2014;26:416-423.
- Scott DGI, Watts RA. Epidemiology and clinical features of systemic vasculitis [published online July 11, 2013]. Clin Exp Nephrol. 2013;17:607-610.
- He X, Yu C, Zhao P, et al. The genetics of Henoch-Schönlein purpura: a systematic review and meta-analysis. Rheumatol Int. 2013;33:1387-1395.
Practice Points
- Staphylococcal scalded skin syndrome, impetigo, eczema herpeticum, Langerhans cell histiocytosis, infantile hemangiomas, and IgA vasculitis all present potential emergencies in pediatric patients in dermatologic settings.
- Early and accurate identification and management of these entities is critical to avoid short-term and long-term negative sequalae.
Emollients didn’t prevent atopic dermatitis in high-risk infants
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The “null findings” of these two studies were “unexpected,” Kirsten P. Perrett, MBBS, Phd, and Rachel L. Peters, PhD, of the department of population allergy at Murdoch Children’s Research Institute, Parkville, Australia, wrote in an accompanying editorial. They noted that emollients are used regularly in the management of atopic dermatitis, where they help maintain the skin barrier and reduce the need for anti-inflammatory therapies.
These two large prevention studies were “prompted” by the results of small, proof-of-concept pilot studies, which “provided strong efficacy signals for the hypothesis that daily emollient use could prevent atopic dermatitis,” they wrote. But the two studies “found no evidence that daily emollient use in either a population-based or high-risk cohort of infants during the first year of life could delay, suppress, or prevent atopic dermatitis.” The lower incidence of atopic dermatitis among those in the dietary and emollient combination, compared with controls (5% vs. 8%) in PreventADALL, could be a chance finding.
The large, randomized Prevention of Eczema by a Barrier Lipid Equilibrium Strategy (PEBBLES) trial is ongoing to confirm results from a small study suggesting the efficacy of a ceramide-dominant emollient. But the PreventADALL study showed low compliance, suggesting that this intervention, if effective, a twice-daily emollient regimen may be tough to implement. “At this stage, emollients should not be recommended for the primary prevention of atopic dermatitis in infants,” they concluded.
Dr. Perrett and Dr. Peters declared no competing interests. Their comments appeared in the Lancet (2020 Feb 19. doi: 10.1016/S0140-6736[19]33174-5).
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
The use of including those at high risk, in two new clinical trials.
The BEEP (Barrier Enhancement for Eczema Prevention) study compared the rates of AD among infants identified as at risk of AD because of family history who had daily applications of emollients (Diprobase cream or Doublebase gel) for the first year of life, compared with a standard skin care group. PreventADALL (Preventing Atopic Dermatitis and Allergies in Children) is a randomized, primary-prevention study conducted in Norway and Sweden that randomized infants into one of four groups: controls whose parents followed regular skin care advice and nutrition guidelines; those who received skin emollients (the addition of emulsified oil to their bath and application of facial cream on at least 4 days a week from age 2 weeks to 8 months); those who received early complementary feeding of peanut, cow’s milk, wheat, and egg introduced between aged 12 and 16 weeks; and a group that combined both the emollient and diet interventions.
Neither of the studies, published in the Lancet, found statistically significant differences in AD rates between the intervention and control groups.
The results put a damper on hopes raised by previous studies that included two small pilot studies, which found that daily use of leave-on emollients in infants considered at high risk of AD prevented the development of AD (J Allergy Clin Immunol 2014 Oct;134:824-30.e6; J Allergy Clin Immunol Oct 2014;134:818-23).
“It was maybe a little bit overly hopeful to think that we could just moisturize and prevent such a complex disorder,” Robert Sidbury, MD, chief of dermatology at Seattle Children’s Hospital, said in an interview. He emphasized that the studies only addressed emollients as a preventative, and that “there’s no question that emollients are still critical for the therapy of eczema.”
Bruce Brod, MD, clinical professor of dermatology at the University of Pennsylvania, Philadelphia, suggested that homogeneous patient populations or insufficient numbers might explain the negative findings. PreventADALL drew patients from Norway and Sweden, while BEEP recruited from the United Kingdom. “They’re important studies, but I think they still lend themselves to further studies with different patient populations and larger groups of patients,” Dr. Brod said in an interview.
BEEP was headed by Joanne Chalmers, PhD, and Hywel Williams, DSc, of the Centre of Evidence-Based Dermatology at the University of Nottingham (England). Håvard Ove Skjerven, PhD, and Karin C Lødrup Carlsen, PhD, of Oslo University Hospital led the PreventADALL study.
The BEEP study randomized 1,394 newborns at 16 sites in the United Kingdom to daily emollient treatment with standard skin care, or standard skin care alone. At one year, compliance was 74% in the intervention group. At age 2, 23% of the intervention group had AD, compared with 25% of controls (hazard ratio, 0.95; P =.61). Skin infections were also higher in the treatment arm (mean, 0.23 per year vs. 0.15 per year; adjusted incidence ratio, 1.55; 95% confidence interval, 1.15-2.09).
“Our study does not support the use of emollients for preventing eczema in high-risk infants, a finding supported by PreventADALL, another large trial using a skin barrier enhancing intervention,” they concluded. Their data “relate only to prevention of eczema and do not directly challenge the practice of using emollients as first-line treatment for eczema.”
In the PreventADALL study, 2,397 newborn infants born between 2015 and 2017 were randomized to one of the four groups. Use of facial cream and emollients during bathing began at 2 weeks, and early complementary feeding of peanut, cow’s milk, wheat, and egg at 3-4 months. The frequency of AD at aged 12 months in the control group was 8%, compared with 11% in the skin-intervention group, 9% in the food-intervention group, and 5% in the combined-intervention group.
These differences were not statistically significant, and “the primary hypothesis that either skin intervention or food intervention reduced atopic dermatitis were not confirmed,” the authors wrote. Parental atopy did not influence the effects of the interventions. Their results were in line with the BEEP results, and the authors “cannot recommend these interventions as primary prevention strategies.”
The researchers will continue to follow children until age 3 years to evaluate the food allergy rates, if the combined-treatment group experiences a long-term benefit. Adherence to the protocol was poor, with 44% compliance with the facial cream application and 27% compliance with bathing emollients; 32% fully adhered to the diet protocols.
The studies were funded by the National Institute for Health Research Health Technology Assessment (BEEP); and a range of public and private funders (PreventADALL). One author of the PreventADALL study disclosed receiving honoraria for presentations from several pharmaceutical companies, and one author received honoraria for presentations from Thermo Fisher Scientific; the rest had no disclosures. Dr. Sidbury has been an investigator for Regeneron. Dr. Brod had no relevant financial disclosures.
SOURCES: Chalmers JR et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32984-8; Skjerven HO et al. Lancet. 2020 Feb 19. doi: 10.1016/S0140-6736(19)32983-6.
FROM THE LANCET
Intervention improves antibiotics use in UTIs
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
FROM PEDIATRICS
Key clinical point: After an educational intervention, approximately 62% of clinicians prescribed first-line antibiotics, up from 43% before the intervention.
Major finding: Cephalexin use increased from 29% before the intervention to 53% after the intervention.
Study details: The data come from a review of 2,142 incident outpatient cases of urinary tract infection in patients aged older than 60 days up to 18 years.
Disclosures: The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
Source: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
Esophageal stricture signals urgent treatment in kids with butterfly skin
LONDON – A quarter of urgent contacts in 20 children with generalized severe recessive dystrophic epidermolysis bullosa (GS-RDEB) were tied to esophageal narrowing, according data from a 12-month review of electronic health records.
Urgent advice was sought 102 times outside of regular or scheduled appointments by the parents of 20 children with GS-RDEB, Christine Prodinger, MD, of the University Clinic of Dermatology at Paracelsus Medical University, Salzburg, Austria, and colleagues reported in a poster presentation at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). The researchers looked at the records from the EB clinic at Great Ormond Street Hospital for Children NHS Foundation Trust, London, during April 2018–April 2019.
The mean number of urgent contacts with the specialist unit was 5.1 per patient per year, the researchers reported, with 24 of the 102 contacts (23.5%) resulting in the child being admitted to a hospital. Most of the contacts were made via email or telephone to EB nurses (94%), by contacts during home visits (3%), or in an appointment with the palliative or symptom care team (3%).
“The most common reason [for the urgent contact] was acute dysphagia,” which was experienced as choking, throat pain, difficulty eating, reflux, and vomiting, the researchers observed. Dysphagia affected children in 27 of the contacts (26.5%), and resulted in esophageal dilatation in 90% of the cases. Other reasons for urgent contact were skin infection (15.7% of contacts), uncontrolled pain (15.7% of the contacts), and eye problems (11.8%).
Esophageal dilatation
Strictures are just one of the esophageal manifestations of the disease, noted Anna Bruckner, MD, associate professor of dermatology and pediatrics in the department of dermatology at the University of Colorado at Denver, Aurora, during an oral presentation. Other possible manifestations include blisters and erosions, the formation of webs – a thin extension of esophageal tissue, perforations, and rupture. “These are primarily problems with dystrophic EB” but can occur with other EB subtypes, she noted.
“We don’t have great evidence” on whether the onset of esophageal strictures can be delayed or prevented, Dr. Bruckner observed. As for management, “fluoroscopy-guided balloon dilatation is probably best” for most patients, but the best procedural approach needs to be discussed on a patient-by-patient basis.
Citing a paper that documents her own experience on the use of esophageal dilatation in 24 children who underwent 231 fluoroscopy-guided balloon dilatation procedures, Dr. Bruckner noted that strictures were most commonly located in the proximal part of the esophagus, with a median distance of 13 cm down from the lips (J Pediatr Gastroenterol Nutr. 2018;67[6]:701-5).
The retrospective chart review reported by Dr. Bruckner showed that there were a median of seven dilatation procedures per patient, and 20 patients had repeated procedures at a median interval of 164 days. About 10% of procedures resulted in adverse events – mostly vomiting, pain, and fever – but there were no perforations or other serious effects, and the rate of subsequent hospitalization was 6.9%.
Dysphagia
Dysphagia was the predominant symptom caused by esophageal stricture in another dataset reported in a poster by Elena Pope, MD, MSc, of the Hospital for Sick Children at the University of Toronto, and colleagues.
Of 125 EB patients who had experienced at least 1 esophageal stricture episode, 497 esophageal stricture events were reported, and 85.5% of patients had difficulty swallowing at presentation, with 29.8% unable to swallow solids and 7.2% unable to swallow liquids. Other symptoms at presentation were painful swallowing (11%), food being stuck in the esophagus (8%), regurgitation (5%), coughing (4.8%), and dyspepsia (2.8%).
The aim of the retrospective, multicenter cohort study was to determine the prevalence of, and predisposing factors for, restenosis of esophageal strictures and factors that may predispose to restenosis. The study population consisted of 66 men and 59 women who had experienced their esophageal stricture at around ages 12-13 years. The majority (98.4%) had dystrophic EB, of which almost half (46.5%) had GS-RDEB.
The researchers found that the location of the esophageal stricture was important for restenosis, and that strictures occurring in the lower esophagus were 67.5% less likely to result in restenosis than if they occurred in the upper esophagus (P = .057; hazard ratio, 0.675).
A higher number of strictures was associated with a higher rate of restenosis, they reported. Indeed, patients who had two esophageal strictures had a 29.4% increased risk of restenosis, compared with those who had just one stricture (P = .038; HR, 1.294), and those with three or more strictures had an increased risk of 78.5%, compared with those having one stricture (P = .005; HR, 1.785).
Strictures longer than 1 cm also were associated with a greater (34.7%) risk of restenosis, compared with shorter strictures (P = .032; HR, 1.347). Various methods of resolving the stricture were used, from fluoroscopy-guided balloon dilatation to retro- or antegrade endoscopy. “Irrespective of method, dilatations are successful,” Dr. Pope and colleagues reported. The overall success of dilatation was 99.3%, with full dilatation achieved in almost all of the patients (96%). Of note is that there was a low risk (2.6%) of complications, they observed.
Medications were used in 46.8% of the patients, with the most popular choice being corticosteroids (90.3%), but the researchers noted that the “potential benefit of periprocedural corticosteroids use in decreasing the risk of restenosis needs further exploration.”
Dr. Bruckner had noted in her presentation that her group did not favor the use of periprocedural corticosteroids, but that antifibrotic therapy “could be attractive” for preventing future strictures.
Dr. Prodinger, Dr. Pope, and their colleagues did not provide disclosure information. Dr. Bruckner is the principal investigator for the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. She disclosed the receipt of grants or research funding, honoraria, or consultation fees from a number of drug companies, as well as other support from the EB Research Partnership and the EB Medical Research Foundation.
SOURCES: Prodinger et al. EB 2020. Poster 3; Bruckner A et al. Pediatr Gastroenterol Nutr. 2018;67(6):701-5; Pope et al. EB 2020. Poster 8.
LONDON – A quarter of urgent contacts in 20 children with generalized severe recessive dystrophic epidermolysis bullosa (GS-RDEB) were tied to esophageal narrowing, according data from a 12-month review of electronic health records.
Urgent advice was sought 102 times outside of regular or scheduled appointments by the parents of 20 children with GS-RDEB, Christine Prodinger, MD, of the University Clinic of Dermatology at Paracelsus Medical University, Salzburg, Austria, and colleagues reported in a poster presentation at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). The researchers looked at the records from the EB clinic at Great Ormond Street Hospital for Children NHS Foundation Trust, London, during April 2018–April 2019.
The mean number of urgent contacts with the specialist unit was 5.1 per patient per year, the researchers reported, with 24 of the 102 contacts (23.5%) resulting in the child being admitted to a hospital. Most of the contacts were made via email or telephone to EB nurses (94%), by contacts during home visits (3%), or in an appointment with the palliative or symptom care team (3%).
“The most common reason [for the urgent contact] was acute dysphagia,” which was experienced as choking, throat pain, difficulty eating, reflux, and vomiting, the researchers observed. Dysphagia affected children in 27 of the contacts (26.5%), and resulted in esophageal dilatation in 90% of the cases. Other reasons for urgent contact were skin infection (15.7% of contacts), uncontrolled pain (15.7% of the contacts), and eye problems (11.8%).
Esophageal dilatation
Strictures are just one of the esophageal manifestations of the disease, noted Anna Bruckner, MD, associate professor of dermatology and pediatrics in the department of dermatology at the University of Colorado at Denver, Aurora, during an oral presentation. Other possible manifestations include blisters and erosions, the formation of webs – a thin extension of esophageal tissue, perforations, and rupture. “These are primarily problems with dystrophic EB” but can occur with other EB subtypes, she noted.
“We don’t have great evidence” on whether the onset of esophageal strictures can be delayed or prevented, Dr. Bruckner observed. As for management, “fluoroscopy-guided balloon dilatation is probably best” for most patients, but the best procedural approach needs to be discussed on a patient-by-patient basis.
Citing a paper that documents her own experience on the use of esophageal dilatation in 24 children who underwent 231 fluoroscopy-guided balloon dilatation procedures, Dr. Bruckner noted that strictures were most commonly located in the proximal part of the esophagus, with a median distance of 13 cm down from the lips (J Pediatr Gastroenterol Nutr. 2018;67[6]:701-5).
The retrospective chart review reported by Dr. Bruckner showed that there were a median of seven dilatation procedures per patient, and 20 patients had repeated procedures at a median interval of 164 days. About 10% of procedures resulted in adverse events – mostly vomiting, pain, and fever – but there were no perforations or other serious effects, and the rate of subsequent hospitalization was 6.9%.
Dysphagia
Dysphagia was the predominant symptom caused by esophageal stricture in another dataset reported in a poster by Elena Pope, MD, MSc, of the Hospital for Sick Children at the University of Toronto, and colleagues.
Of 125 EB patients who had experienced at least 1 esophageal stricture episode, 497 esophageal stricture events were reported, and 85.5% of patients had difficulty swallowing at presentation, with 29.8% unable to swallow solids and 7.2% unable to swallow liquids. Other symptoms at presentation were painful swallowing (11%), food being stuck in the esophagus (8%), regurgitation (5%), coughing (4.8%), and dyspepsia (2.8%).
The aim of the retrospective, multicenter cohort study was to determine the prevalence of, and predisposing factors for, restenosis of esophageal strictures and factors that may predispose to restenosis. The study population consisted of 66 men and 59 women who had experienced their esophageal stricture at around ages 12-13 years. The majority (98.4%) had dystrophic EB, of which almost half (46.5%) had GS-RDEB.
The researchers found that the location of the esophageal stricture was important for restenosis, and that strictures occurring in the lower esophagus were 67.5% less likely to result in restenosis than if they occurred in the upper esophagus (P = .057; hazard ratio, 0.675).
A higher number of strictures was associated with a higher rate of restenosis, they reported. Indeed, patients who had two esophageal strictures had a 29.4% increased risk of restenosis, compared with those who had just one stricture (P = .038; HR, 1.294), and those with three or more strictures had an increased risk of 78.5%, compared with those having one stricture (P = .005; HR, 1.785).
Strictures longer than 1 cm also were associated with a greater (34.7%) risk of restenosis, compared with shorter strictures (P = .032; HR, 1.347). Various methods of resolving the stricture were used, from fluoroscopy-guided balloon dilatation to retro- or antegrade endoscopy. “Irrespective of method, dilatations are successful,” Dr. Pope and colleagues reported. The overall success of dilatation was 99.3%, with full dilatation achieved in almost all of the patients (96%). Of note is that there was a low risk (2.6%) of complications, they observed.
Medications were used in 46.8% of the patients, with the most popular choice being corticosteroids (90.3%), but the researchers noted that the “potential benefit of periprocedural corticosteroids use in decreasing the risk of restenosis needs further exploration.”
Dr. Bruckner had noted in her presentation that her group did not favor the use of periprocedural corticosteroids, but that antifibrotic therapy “could be attractive” for preventing future strictures.
Dr. Prodinger, Dr. Pope, and their colleagues did not provide disclosure information. Dr. Bruckner is the principal investigator for the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. She disclosed the receipt of grants or research funding, honoraria, or consultation fees from a number of drug companies, as well as other support from the EB Research Partnership and the EB Medical Research Foundation.
SOURCES: Prodinger et al. EB 2020. Poster 3; Bruckner A et al. Pediatr Gastroenterol Nutr. 2018;67(6):701-5; Pope et al. EB 2020. Poster 8.
LONDON – A quarter of urgent contacts in 20 children with generalized severe recessive dystrophic epidermolysis bullosa (GS-RDEB) were tied to esophageal narrowing, according data from a 12-month review of electronic health records.
Urgent advice was sought 102 times outside of regular or scheduled appointments by the parents of 20 children with GS-RDEB, Christine Prodinger, MD, of the University Clinic of Dermatology at Paracelsus Medical University, Salzburg, Austria, and colleagues reported in a poster presentation at the EB World Congress, organized by the Dystrophic Epidermolysis Bullosa Association (DEBRA). The researchers looked at the records from the EB clinic at Great Ormond Street Hospital for Children NHS Foundation Trust, London, during April 2018–April 2019.
The mean number of urgent contacts with the specialist unit was 5.1 per patient per year, the researchers reported, with 24 of the 102 contacts (23.5%) resulting in the child being admitted to a hospital. Most of the contacts were made via email or telephone to EB nurses (94%), by contacts during home visits (3%), or in an appointment with the palliative or symptom care team (3%).
“The most common reason [for the urgent contact] was acute dysphagia,” which was experienced as choking, throat pain, difficulty eating, reflux, and vomiting, the researchers observed. Dysphagia affected children in 27 of the contacts (26.5%), and resulted in esophageal dilatation in 90% of the cases. Other reasons for urgent contact were skin infection (15.7% of contacts), uncontrolled pain (15.7% of the contacts), and eye problems (11.8%).
Esophageal dilatation
Strictures are just one of the esophageal manifestations of the disease, noted Anna Bruckner, MD, associate professor of dermatology and pediatrics in the department of dermatology at the University of Colorado at Denver, Aurora, during an oral presentation. Other possible manifestations include blisters and erosions, the formation of webs – a thin extension of esophageal tissue, perforations, and rupture. “These are primarily problems with dystrophic EB” but can occur with other EB subtypes, she noted.
“We don’t have great evidence” on whether the onset of esophageal strictures can be delayed or prevented, Dr. Bruckner observed. As for management, “fluoroscopy-guided balloon dilatation is probably best” for most patients, but the best procedural approach needs to be discussed on a patient-by-patient basis.
Citing a paper that documents her own experience on the use of esophageal dilatation in 24 children who underwent 231 fluoroscopy-guided balloon dilatation procedures, Dr. Bruckner noted that strictures were most commonly located in the proximal part of the esophagus, with a median distance of 13 cm down from the lips (J Pediatr Gastroenterol Nutr. 2018;67[6]:701-5).
The retrospective chart review reported by Dr. Bruckner showed that there were a median of seven dilatation procedures per patient, and 20 patients had repeated procedures at a median interval of 164 days. About 10% of procedures resulted in adverse events – mostly vomiting, pain, and fever – but there were no perforations or other serious effects, and the rate of subsequent hospitalization was 6.9%.
Dysphagia
Dysphagia was the predominant symptom caused by esophageal stricture in another dataset reported in a poster by Elena Pope, MD, MSc, of the Hospital for Sick Children at the University of Toronto, and colleagues.
Of 125 EB patients who had experienced at least 1 esophageal stricture episode, 497 esophageal stricture events were reported, and 85.5% of patients had difficulty swallowing at presentation, with 29.8% unable to swallow solids and 7.2% unable to swallow liquids. Other symptoms at presentation were painful swallowing (11%), food being stuck in the esophagus (8%), regurgitation (5%), coughing (4.8%), and dyspepsia (2.8%).
The aim of the retrospective, multicenter cohort study was to determine the prevalence of, and predisposing factors for, restenosis of esophageal strictures and factors that may predispose to restenosis. The study population consisted of 66 men and 59 women who had experienced their esophageal stricture at around ages 12-13 years. The majority (98.4%) had dystrophic EB, of which almost half (46.5%) had GS-RDEB.
The researchers found that the location of the esophageal stricture was important for restenosis, and that strictures occurring in the lower esophagus were 67.5% less likely to result in restenosis than if they occurred in the upper esophagus (P = .057; hazard ratio, 0.675).
A higher number of strictures was associated with a higher rate of restenosis, they reported. Indeed, patients who had two esophageal strictures had a 29.4% increased risk of restenosis, compared with those who had just one stricture (P = .038; HR, 1.294), and those with three or more strictures had an increased risk of 78.5%, compared with those having one stricture (P = .005; HR, 1.785).
Strictures longer than 1 cm also were associated with a greater (34.7%) risk of restenosis, compared with shorter strictures (P = .032; HR, 1.347). Various methods of resolving the stricture were used, from fluoroscopy-guided balloon dilatation to retro- or antegrade endoscopy. “Irrespective of method, dilatations are successful,” Dr. Pope and colleagues reported. The overall success of dilatation was 99.3%, with full dilatation achieved in almost all of the patients (96%). Of note is that there was a low risk (2.6%) of complications, they observed.
Medications were used in 46.8% of the patients, with the most popular choice being corticosteroids (90.3%), but the researchers noted that the “potential benefit of periprocedural corticosteroids use in decreasing the risk of restenosis needs further exploration.”
Dr. Bruckner had noted in her presentation that her group did not favor the use of periprocedural corticosteroids, but that antifibrotic therapy “could be attractive” for preventing future strictures.
Dr. Prodinger, Dr. Pope, and their colleagues did not provide disclosure information. Dr. Bruckner is the principal investigator for the Epidermolysis Bullosa Clinical Characterization and Outcomes Database. She disclosed the receipt of grants or research funding, honoraria, or consultation fees from a number of drug companies, as well as other support from the EB Research Partnership and the EB Medical Research Foundation.
SOURCES: Prodinger et al. EB 2020. Poster 3; Bruckner A et al. Pediatr Gastroenterol Nutr. 2018;67(6):701-5; Pope et al. EB 2020. Poster 8.
REPORTING FROM EB 2020
Meta-analysis highlights safety concerns with interleukin inhibition
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
MAUI, HAWAII – The use of interleukin inhibitors for treatment of rheumatologic diseases doubles a patient’s risk of serious infections, according to a comprehensive systematic review and meta-analysis of 74 randomized, placebo-controlled trials presented by Jawad Bilal, MBBS, at the 2020 Rheumatology Winter Clinical Symposium.
The meta-analysis, which incorporated 29,214 patients with a variety of rheumatic diseases, demonstrated that patients receiving interleukin (IL) inhibitors had a 1.97-fold increased risk of serious infections, a finding accompanied by a high degree of statistical certainty. The number-needed-to-harm was 67 patients treated for a median of 24 weeks in order to generate one additional serious infection.
“That number-needed-to-harm is a significant finding because having a serious infection means by definition you’re getting admitted to the hospital and receiving IV antibiotics,” Dr. Bilal observed in an interview.
The meta-analysis also found that IL inhibition was associated with a 2.35-fold increased risk of opportunistic infections and a 1.52-fold higher risk of developing cancer, both findings with statistical significance (P =.03) but only moderate certainty because fewer of those events were captured in the trials compared to the numbers of serious infections, explained Dr. Bilal of the University of Arizona, Tucson.
For opportunistic infections, the number-needed-to-harm was 250 patients treated with an IL inhibitor for a median of 54 weeks in order to result in one additional opportunistic infection. For cancer, the number-needed-to-harm was 250 for a median of 24 weeks.
Dr. Bilal noted that while the IL inhibitors are drugs of established efficacy in rheumatologic diseases, their safety has not previously undergone anything approaching the comprehensive scrutiny carried out in this meta-analysis. The meta-analysis, which included all published placebo-controlled randomized trials and their extension studies, employed rigorous methodology in accord with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement and the GRADE approach to data analysis. Studies of IL inhibitors in patients with dermatologic and GI diseases were excluded from the meta-analysis.
He offered a caveat regarding the cancer risk findings: “Our analysis showed that the cancer risk is increased, but the results are not conclusive because we only had a few years of data. With cancer, you really need at least 8-10 years of data. So the real-world experience with the interleukin inhibitors in the large registries is what’s going to tell if the cancer risk is really increased or not. In the meantime, we all have to be cautious.”
The number of serious infections collected in the meta-analysis afforded sufficient statistical power for the investigators to break down differential risks based on individual drugs and indications. Among the drugs associated with significantly increased risk of serious infections were anakinra, with an odds ratio of 2.67, compared with placebo; secukinumab with an OR of 2.43; and tocilizumab with an OR or 1.76. Ustekinumab and ixekizumab were associated with 2.57- and 3.89-fold increased risks, respectively, but the number of rheumatology patients treated with those two biologics wasn’t large enough for those findings to achieve statistical significance.
Rheumatoid arthritis patients who received an IL inhibitor rather than placebo had a 1.98-fold increased risk of serious infection, while those with psoriatic arthritis had a 2.21-fold increased risk. Patients treated for SLE had a 6.44-fold increased risk, and those with juvenile idiopathic arthritis had a 5.37-fold higher risk, but the margins for error were such that those results weren’t statistically significant.
“I think this study is going to help clinicians and patients when they’re trying to weigh the risks and benefits of IL inhibitors, especially if they already have risk factors, like a recent history of serious infection or a history of cancer or of opportunistic infection,” Dr. Bilal commented.
A study limitation was that he and his coinvestigators had to lump together the various IL inhibitors in order to gain statistical power, even though the drugs work differently, he noted.
Dr. Bilal reported having no financial conflicts regarding his study, the full details of which have been published (JAMA Netw Open. 2019 Oct 2. doi: 10.1001/jamanetworkopen.2019.13102).
REPORTING FROM RWCS 2020
Consider toys as culprits in children with contact allergies
A variety of according to the results of a review of 25 published articles.
“In recent years the products have become a reflection of the compounds used frequently in manufacturing, including metals and plastic compounds,” wrote Justine Fenner, MD, and coauthors, from the departments of dermatology and pediatrics at the Icahn School of Medicine at Mount Sinai, New York,
In a study published in Contact Dermatitis, the researchers identified 25 articles describing dermatitis, rash, or eczema associated with a range of toy and play product terms including Nintendo, PlayStation, putty, glue, doll, game, car, bicycle, slime, iPad, and iPhone.
Overall, nickel was the most common allergen. Cases of nickel dermatitis were associated with laptops, videogame controllers, iPads, and cell phones. Cell phones were the most common electronics associated with contact dermatitis, which was observed on the cheek, periauricular area, and hand, as well as the breast in one case of a patient who kept her phone in her bra.
Other sources of metal allergens were identified in toy cars and costume jewelry, the researchers noted.
In addition, temporary tattoos have been associated with contact dermatitis in children, as have homemade “slime” products, which often contain not only borax or other household detergents, but also glue, shaving cream, or coloring.
However, identification of true allergic contact dermatitis from toys “requires both identification of the chemical contents of toys, which are proprietary in nature, and then epicutaneous allergy testing of these ingredients,” the researchers said.
The study findings were limited by several factors including the consideration only of English-language articles and of cases in children, which thus eliminates other potential cases, the researchers noted. However, the results suggest that dermatologists consider toys as a source of contact dermatitis in children, especially if the time to diagnosis is months to years, they said. “Additionally, it may be useful, as it was in several of the above cases, to have the patient bring in his or her favorite toys for the dermatologist to examine and help further understand the etiology of patient’s rash,” they noted. Moreover, “there is an unmet need for corporations to reveal the chemical ingredients of their toys when allergic contact dermatitis is suspected in order to properly evaluate the patient,” they added.
“Contact dermatitis has been underreported in children and constitutes an ongoing concern,” senior author Nanette Silverberg, MD, chief of pediatric dermatology for the Mount Sinai Health System, said in an interview.
“In particular, toy-related allergy is concerning due to the rise in allergen inclusion in common play items,” she commented. The current analysis identified many case reports of allergens that pediatric dermatologists are frequently seeing in their offices, notably metals such as nickel, she pointed out. “The allergen that always stands out ahead of others is nickel,” Dr. Silverberg said. “Nickel allergy affects about 25% of Americans, often starting in early childhood,” she said. “In the European Union, legislation has been passed to reduce nickel release from metals, which has resulted in less sensitization to nickel. We lack such legislation in the United States,” she added.
Other trending allergens include methylchloroisothiazolinone/methylisothiazolinone, which may be components of glue or other ingredients in some “slime” products, Dr. Silverberg said.
She advised clinicians to consider patch testing when addressing localized or persistent dermatitis in children. “Furthermore, consider toys as potential relevant allergens that should be modified in order to achieve skin improvement,” she said.
“Greater reporting of pediatric allergic contact dermatitis is needed,” Dr. Silverberg emphasized. “Additionally, surveillance and monitoring for trends in allergen exposures in toys and personal care items is required to analyze this ongoing concern of childhood,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Fenner J et al. Contact Dermatitis. 2020 Feb 22. doi: 10.1111/cod.13500.
A variety of according to the results of a review of 25 published articles.
“In recent years the products have become a reflection of the compounds used frequently in manufacturing, including metals and plastic compounds,” wrote Justine Fenner, MD, and coauthors, from the departments of dermatology and pediatrics at the Icahn School of Medicine at Mount Sinai, New York,
In a study published in Contact Dermatitis, the researchers identified 25 articles describing dermatitis, rash, or eczema associated with a range of toy and play product terms including Nintendo, PlayStation, putty, glue, doll, game, car, bicycle, slime, iPad, and iPhone.
Overall, nickel was the most common allergen. Cases of nickel dermatitis were associated with laptops, videogame controllers, iPads, and cell phones. Cell phones were the most common electronics associated with contact dermatitis, which was observed on the cheek, periauricular area, and hand, as well as the breast in one case of a patient who kept her phone in her bra.
Other sources of metal allergens were identified in toy cars and costume jewelry, the researchers noted.
In addition, temporary tattoos have been associated with contact dermatitis in children, as have homemade “slime” products, which often contain not only borax or other household detergents, but also glue, shaving cream, or coloring.
However, identification of true allergic contact dermatitis from toys “requires both identification of the chemical contents of toys, which are proprietary in nature, and then epicutaneous allergy testing of these ingredients,” the researchers said.
The study findings were limited by several factors including the consideration only of English-language articles and of cases in children, which thus eliminates other potential cases, the researchers noted. However, the results suggest that dermatologists consider toys as a source of contact dermatitis in children, especially if the time to diagnosis is months to years, they said. “Additionally, it may be useful, as it was in several of the above cases, to have the patient bring in his or her favorite toys for the dermatologist to examine and help further understand the etiology of patient’s rash,” they noted. Moreover, “there is an unmet need for corporations to reveal the chemical ingredients of their toys when allergic contact dermatitis is suspected in order to properly evaluate the patient,” they added.
“Contact dermatitis has been underreported in children and constitutes an ongoing concern,” senior author Nanette Silverberg, MD, chief of pediatric dermatology for the Mount Sinai Health System, said in an interview.
“In particular, toy-related allergy is concerning due to the rise in allergen inclusion in common play items,” she commented. The current analysis identified many case reports of allergens that pediatric dermatologists are frequently seeing in their offices, notably metals such as nickel, she pointed out. “The allergen that always stands out ahead of others is nickel,” Dr. Silverberg said. “Nickel allergy affects about 25% of Americans, often starting in early childhood,” she said. “In the European Union, legislation has been passed to reduce nickel release from metals, which has resulted in less sensitization to nickel. We lack such legislation in the United States,” she added.
Other trending allergens include methylchloroisothiazolinone/methylisothiazolinone, which may be components of glue or other ingredients in some “slime” products, Dr. Silverberg said.
She advised clinicians to consider patch testing when addressing localized or persistent dermatitis in children. “Furthermore, consider toys as potential relevant allergens that should be modified in order to achieve skin improvement,” she said.
“Greater reporting of pediatric allergic contact dermatitis is needed,” Dr. Silverberg emphasized. “Additionally, surveillance and monitoring for trends in allergen exposures in toys and personal care items is required to analyze this ongoing concern of childhood,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Fenner J et al. Contact Dermatitis. 2020 Feb 22. doi: 10.1111/cod.13500.
A variety of according to the results of a review of 25 published articles.
“In recent years the products have become a reflection of the compounds used frequently in manufacturing, including metals and plastic compounds,” wrote Justine Fenner, MD, and coauthors, from the departments of dermatology and pediatrics at the Icahn School of Medicine at Mount Sinai, New York,
In a study published in Contact Dermatitis, the researchers identified 25 articles describing dermatitis, rash, or eczema associated with a range of toy and play product terms including Nintendo, PlayStation, putty, glue, doll, game, car, bicycle, slime, iPad, and iPhone.
Overall, nickel was the most common allergen. Cases of nickel dermatitis were associated with laptops, videogame controllers, iPads, and cell phones. Cell phones were the most common electronics associated with contact dermatitis, which was observed on the cheek, periauricular area, and hand, as well as the breast in one case of a patient who kept her phone in her bra.
Other sources of metal allergens were identified in toy cars and costume jewelry, the researchers noted.
In addition, temporary tattoos have been associated with contact dermatitis in children, as have homemade “slime” products, which often contain not only borax or other household detergents, but also glue, shaving cream, or coloring.
However, identification of true allergic contact dermatitis from toys “requires both identification of the chemical contents of toys, which are proprietary in nature, and then epicutaneous allergy testing of these ingredients,” the researchers said.
The study findings were limited by several factors including the consideration only of English-language articles and of cases in children, which thus eliminates other potential cases, the researchers noted. However, the results suggest that dermatologists consider toys as a source of contact dermatitis in children, especially if the time to diagnosis is months to years, they said. “Additionally, it may be useful, as it was in several of the above cases, to have the patient bring in his or her favorite toys for the dermatologist to examine and help further understand the etiology of patient’s rash,” they noted. Moreover, “there is an unmet need for corporations to reveal the chemical ingredients of their toys when allergic contact dermatitis is suspected in order to properly evaluate the patient,” they added.
“Contact dermatitis has been underreported in children and constitutes an ongoing concern,” senior author Nanette Silverberg, MD, chief of pediatric dermatology for the Mount Sinai Health System, said in an interview.
“In particular, toy-related allergy is concerning due to the rise in allergen inclusion in common play items,” she commented. The current analysis identified many case reports of allergens that pediatric dermatologists are frequently seeing in their offices, notably metals such as nickel, she pointed out. “The allergen that always stands out ahead of others is nickel,” Dr. Silverberg said. “Nickel allergy affects about 25% of Americans, often starting in early childhood,” she said. “In the European Union, legislation has been passed to reduce nickel release from metals, which has resulted in less sensitization to nickel. We lack such legislation in the United States,” she added.
Other trending allergens include methylchloroisothiazolinone/methylisothiazolinone, which may be components of glue or other ingredients in some “slime” products, Dr. Silverberg said.
She advised clinicians to consider patch testing when addressing localized or persistent dermatitis in children. “Furthermore, consider toys as potential relevant allergens that should be modified in order to achieve skin improvement,” she said.
“Greater reporting of pediatric allergic contact dermatitis is needed,” Dr. Silverberg emphasized. “Additionally, surveillance and monitoring for trends in allergen exposures in toys and personal care items is required to analyze this ongoing concern of childhood,” she said.
The study received no outside funding. The researchers had no financial conflicts to disclose.
SOURCE: Fenner J et al. Contact Dermatitis. 2020 Feb 22. doi: 10.1111/cod.13500.
FROM CONTACT DERMATITIS
Antibiotic resistance rises among pneumococcus strains in kids
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Dr. Kaur and colleagues report their analysis of pneumococcal resistance among nasopharyngeal and middle ear isolates (90% nasopharyngeal and 10% middle ear) collected between 2008 and 2016. They demonstrate the dominant role that nonvaccine serotypes play in carriage and acute otitis media (AOM) in children, and by extension potentially the entire spectrum of pneumococcal disease in the 13-valent pneumococcal conjugate vaccine (PCV13) era. Nonsusceptibility to beta-lactams was reported for one-third of isolates with the increase in the most recent reported years (2013-2016).
What are the implications for treatment of pneumococcal infections? For AOM, amoxicillin minimum inhibitory concentrations (MIC) were all less than 4 mcg/mL, which is the pharmacodynamic breakpoint for high-dose (90 mg/kg per day) AOM regimens; these data support continued use of high-dose amoxicillin for children with AOM that requires antimicrobial treatment. Resistance to macrolides (erythromycin and likely azithromycin) occurred in approximately one-third of isolates; however, in contrast to beta-lactams (amoxicillin), higher macrolide doses do not overcome resistance. Thus macrolide use for AOM appears limited to those with beta-lactam allergy and no better alternative drug, i.e., expect failure in one-third of AOM patients if macrolides are used. For ceftriaxone, no 2013-2016 isolate had a MIC over 0.5 mcg/mL, implying that ceftriaxone remains appropriate first-line therapy for serious pneumococcal disease and effective for pneumococcal AOM when oral drugs have failed or are not an option because of repeated emesis. Interestingly, trimethoprim/sulfamethoxazole (T/S) had lower resistance rates against the nonvaccine “bad boy” serogroup 35 (8%-15%), compared with cephalosporins (32%-57%). Perhaps we are back to the future and T/S will again have a role against pneumococcal AOM. Of note, no isolate was resistant to levofloxacin or linezolid. Linezolid or macrolide use alone must be considered with the caveat that nontypeable Haemophilus influenzae now likely surpasses pneumococcus as an AOM pathogen, and neither drug class is active against nontypeable H. influenzae.
What are the implications for prevention? This is one of many studies in the post-PCV era reporting serotype replacement with nonvaccine serotypes. But most prior studies reported reduced overall disease burden; in other words, the absolute number of pneumococcal infections was reduced, but residual AOM nonvaccine types dominated as the etiology. The current study, however, suggests that the overall number of AOM episodes may not be less because increases in AOM caused by nonvaccine serotypes may be offsetting declines in AOM caused by vaccine serotypes. This concept contrasts to multiple large epidemiologic studies demonstrating a decline in overall incidence of AOM office visits/episodes and several Israeli studies reporting a decline in pneumococcal AOM in children who warrant tympanocentesis. These new data are food for thought, but antibiotic resistance can vary regionally, so confirmation based on data from other regions seems warranted.
Next-generation vaccines will need to consider which serotypes are prevalent in pneumococcal disease, including AOM, as we continue into the PCV13 era. However, serotypes causing invasive pneumococcal disease and pneumonia would be higher priorities than AOM. Indeed, several candidate PCV vaccines are currently in clinical trials adding up to seven serotypes, including most of the newly emerging invasive disease serotypes. One downside to the newer PCVs is lack of serogroup 35, a prominent culprit in AOM resistance in the current report.
Stephen I. Pelton, MD, is professor of pediatrics and epidemiology at Boston University and senior attending physician at Boston Medical Center. Christopher J. Harrison, MD, is professor of pediatrics and pediatric infectious diseases at Children’s Mercy Hospital–Kansas City, Mo. Dr. Pelton has received honorarium from Merck Vaccines, Pfizer, and Sanofi for participation in advisory board meeting on pneumococcal vaccine and/or membership on the Data and Safety Monitoring Board. Boston Medical Center has received investigator-initiated research grants from Merck Vaccines and Pfizer.
Children’s Mercy Hospital – Kansas City Boston Medical Center has received funding from GlaxoSmithKline, Merck, and Pfizer for research vaccine studies, and from Pfizer and Merck for investigator-initiated research grants for in vitro pneumococcal investigations on which Dr. Harrison is an investigator.
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
Antibiotic resistance in strains of Streptococcus pneumoniae has been rising since 2013 because of changing susceptibility profiles, based on data from 1,201 isolates collected from 448 children in primary care settings.
“New strains expressing capsular serotypes not included in the 13-valent pneumococcal conjugate vaccine are emerging to cause disease, and strains that acquire antibiotic resistance are increasing in frequency due to their survival of the fittest advantage,” wrote Ravinder Kaur, PhD, of Rochester (N.Y.) General Hospital Research Institute, and colleagues.
Similar Darwinian principles occurred after the introduction of PCV-7, the study authors added.
In a prospective cohort study published in Clinical Infectious Diseases, the researchers reviewed 1,201 isolates collected from the nasopharynx during healthy periods, and from the nasopharynx and middle ear fluid (MEF) during episodes of acute otitis media, in children aged 6-36 months who were seen in primary care settings.
The isolates were collected during 2006-2016 to reflect the pre- and post-PCV13 era. Children received PCV-7 from 2006 until April 2010, and received PCV-13 after April 2010.
Overall, the number of acute otitis media (AOM) cases caused by S. pneumoniae was not significantly different between the PCV-7 and PCV-13 eras, nor was the frequency of pneumococci identified in the nasopharynx during healthy visits and visits at the start of an AOM infection.
The researchers examined susceptibility using minimum inhibitory concentrations (MIC). During healthy visits, the MIC50 of isolated pneumococci was low (no greater than 0.06 mcg/mL) for all four beta-lactam drugs tested. And it didn’t change significantly over the study years.
In contrast, among the nasopharyngeal and MEF isolates during AOM, the MIC50 to penicillin, amoxicillin, ceftriaxone, and meropenem during 2013-2016 rose significantly, the investigators said.
A change in antibiotic susceptibility within a subtype also contributed to the development of PCV-13 resistance.
The study authors identified three serotypes that affected the changes in susceptibility in their study population. Serotypes 35B and 35F increased their beta-lactam resistance during 2013-2016, and serotype 11A had a higher MIC to quinolones and became more prevalent during 2013-2016. Those three serotypes accounted for most of the change in antibiotic susceptibility, the researchers said.
In addition, “the frequency of strains resistant to penicillin and amoxicillin decreased with the introduction of PCV-13, but rebounded to levels similar to those before PCV-13 introduction by 2015-2016,” the investigators noted.
The study findings were limited by several factors, including the homogeneous study population and potential lack of generalizability to other settings. In addition, the researchers did not study antibiotic consumption or antibiotic treatment failure, and they could not account for potential AOM cases that may have been treated in settings other than primary care.
However, the investigators said the results support the need for additional studies and attention to the development of the next generation of PCVs, the PCV-15 and PCV-20. Both include serotypes 22F and 33F, but neither includes 35B or 35F. The PCV-20 also includes 11A and 15B.
The study was supported in part by the National Institutes of Health and Sanofi Pasteur. Some isolates collected during the 2010-2013 time period were part of a study supported by Pfizer. The researchers had no relevant financial conflicts to disclose.
SOURCE: Kaur R et al. Clin Inf Dis. 2020 Feb 18. doi: 10.1093/cid/ciaa157.
FROM CLINICAL INFECTIOUS DISEASES
Children bearing the brunt of declining flu activity
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”
National flu activity decreased for the second consecutive week, but pediatric mortality is heading in the opposite direction, according to the Centers for Disease Control and Prevention.
Influenza-like illness (ILI) represented 5.5% of all visits to outpatient health care providers during the week ending Feb. 22, compared with 6.1% the previous week, the CDC’s influenza division reported Feb. 28. The ILI visit rate had reached 6.6% in early February after dropping to 5.0% in mid-January, following a rise to a season-high 7.1% in the last week of December.
Another measure of ILI activity, the percentage of laboratory specimens testing positive, also declined for the second week in a row. The rate was 26.4% for the week ending Feb. 22, which is down from the season high of 30.3% reached 2 weeks before, the influenza division said.
ILI-related deaths among children, however, are not dropping. The total for 2019-2020 is now up to 125, and that “number is higher for the same time period than in every season since reporting began in 2004-05, except for the 2009 pandemic,” the CDC noted.
Hospitalization rates, which have been fairly typical in the general population, also are elevated for young adults and school-aged children, the agency said, and “rates among children 0-4 years old are now the highest CDC has on record at this point in the season, surpassing rates reported during the second wave of the 2009 H1N1 pandemic.”