User login
Survey: Acceptance of COVID-19 vaccine dips below 50%
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Less than half of Americans now say that they would get a coronavirus vaccine if one became available, according to a survey conducted Oct. 8-10.
the lowest number since the weekly survey began at the end of February, digital media company Morning Consult reported.
Americans’ willingness to receive such a vaccine reached its high point, 72%, in early April but has been steadily dropping. “Overall willingness has hovered around 50% throughout September, fueled primarily by a sharp drop among Democrats since mid-August, around the time reports of White House interference at the Food and Drug Administration and other federal health agencies began to command more public attention,” Morning Consult noted.
Despite that drop, a majority of Democrats (55%) are still willing to get a COVID-19 vaccine, compared with 48% of Republicans and just 41% of independents. The willingness gap between the two parties was quite a bit wider in the previous poll, conducted Oct. 1-4: 60% of Democrats versus 48% for Republicans, the company said.
“Keeping with longstanding trends, the survey also shows women were less likely to say they’d seek a vaccine than men (42% to 55%), as were people with lower education levels and those who live in rural areas,” the news outlet added.
The latest poll results also show that 33% of respondents (43% of Republicans/25% of Democrats) are socializing in public places. The overall number was just 8% in mid-April but was up to 27% by mid-June. The proportion of all adults who believe in the effectiveness of face masks has been around 80% since April, but there is a significant gap between those who strongly approve of President Trump (66%) and those who strongly disapprove (95%), Morning Consult said.
Proton pump inhibitors tied to COVID-19 risk
In light of this finding, physicians should consider which patients truly need these powerful acid-lowering drugs, said Brennan Spiegel, MD, MSHS, AGAF, professor of medicine and public health at Cedars Sinai Medical Center in Los Angeles, Calif.
“All it means is that we’re going to have a conversation with our patients,” he said in an interview. “We don’t normally have that conversation because we don’t live in an environment with a high risk of enteric infection. But now we’re in a pandemic.”
The study by Dr. Spiegel and his colleagues was published online on July 7 in the American Journal of Gastroenterology.
Use of PPIs has skyrocketed over the past 2 decades. For ambulatory care visits, their use increased from 1.6% in 1998 to 7.6% in 2015. The increase raised questions about overprescription.
Although studies have not borne out many of the other concerns raised about adverse reactions, they have shown that the drugs increase the risk for enteric infections, including infections by SARS-CoV-1, a virus that is related to the COVID-19 virus, SARS-CoV-2, Dr. Spiegel said.
SARS-CoV-2 uses the angiotensin-converting enzyme–2 receptor to invade enterocytes. Dr. Spiegel theorized that an increase in stomach pH above 3 as a result of use of PPIs might allow the virus to enter the GI tract more easily, leading to enteritis, colitis, and systemic spread to other organs, including the lungs. “There is a reason we have acid in our stomachs,” Dr. Spiegel said.
To see how PPI use relates to COVID-19 infections, Dr. Spiegel and his colleagues surveyed online a nationally representative sample of Americans between May 3 and June 24, 2020, as part of a larger survey on gastroenterologic health.
Participants answered questions about gastrointestinal symptoms, current use of PPIs, and COVID-19 test results. They also answered questions about histamine-2 receptor agonists (H2RAs), also known as H2 blockers, which are used to treat some of the same conditions as PPIs but that do not reduce stomach acid as much.
The surveying firm, Cint, contacted 264,058 people. Of the 86,602 eligible participants who completed the survey, 53,130 said they had experienced abdominal discomfort, acid reflux, heartburn, or regurgitation. These survey participants were subsequently asked about PPI and H2RA use.
Of these, 6.4% reported testing positive for SARS-CoV-2. The researchers adjusted for age, sex, race/ethnicity, education, marital status, household income, body mass index, smoking, alcohol consumption, U.S. region, insurance status, and the presence of irritable bowel syndrome, celiac disease, gastroesophageal reflux disease, liver cirrhosis, Crohn’s disease, ulcerative colitis, diabetes, and HIV/AIDS.
After adjusting for these factors, the researchers found that those who took PPIs up to once a day were twice as likely to have had a positive COVID-19 test result than those who did not take the drugs (odds ratio, 2.15; 95% confidence interval, 1.90-2.44).
Those who took PPIs twice a day were almost four times as likely to have tested positive for the disease (OR, 3.67; 95% CI, 2.93-4.60).
By contrast, those taking H2RA drugs once daily were 15% less likely to report a positive COVID-19 test result (OR, 0.85; 95% CI, 0.74-0.99). Research is currently underway to determine whether H2RAs might protect against the disease for reasons unrelated to pH balance.
Dr. Spiegel cautioned that the current data show only an association between PPI use and COVID-19 positivity; it cannot prove cause and effect.
Nevertheless, Dr. Spiegel said the findings should encourage physicians to prescribe PPIs only when clearly indicated. “If somebody is not yet on a PPI and you’re considering whether to start them on a PPI, it’s a good idea to consider H2 blockers,” he said.
People who need a daily dose of a PPI to control a severe condition can safely continue doing so, but such patients should take care to follow standard public health recommendations for avoiding exposure to the virus. These recommendations include wearing a mask, maintaining social distance, and washing hands frequently.
“People who are older, comorbid, or smokers – if they get infected, it could be severe,” he said. “[For] someone like that, it’s reasonable to ask, do we really need to be on twice-daily PPIs? There is good evidence that they are no better off than if they are taking once-daily doses.”
Brian Lacy, MD, PhD, a professor of medicine at the Mayo Clinic in Jacksonville, Fla., agreed that the study should prompt physicians to take a second look at their patients’ PPI prescriptions. “My view is that PPIs are frequently overused, and maybe this is one more piece of data that, if someone is on PPIs, maybe they don’t need to be on this medication.”
On the other hand, the drugs are important for treating conditions such as erosive esophagitis and healing ulcers, he said. The overall risk of contracting COVID-19 is low, so even this finding of a 3.7-fold increased risk should not lead patients or providers to stop taking or prescribing PPIs.
The study also lends support to the idea that the gastrointestinal tract could be involved in SARS-CoV-2 transmission, and it supports warnings about aerosols emitted from flushing toilets and through exhalation, Dr. Spiegel said. There is less evidence of the virus being transmitted through food. “It may not be fecal-oral; it may be fecal-respiratory,” he said.
The study was part of a larger project funded by Ironwood Pharmaceuticals. Dr. Spiegel reported relationships with Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Shire Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals. Dr. Lacy has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In light of this finding, physicians should consider which patients truly need these powerful acid-lowering drugs, said Brennan Spiegel, MD, MSHS, AGAF, professor of medicine and public health at Cedars Sinai Medical Center in Los Angeles, Calif.
“All it means is that we’re going to have a conversation with our patients,” he said in an interview. “We don’t normally have that conversation because we don’t live in an environment with a high risk of enteric infection. But now we’re in a pandemic.”
The study by Dr. Spiegel and his colleagues was published online on July 7 in the American Journal of Gastroenterology.
Use of PPIs has skyrocketed over the past 2 decades. For ambulatory care visits, their use increased from 1.6% in 1998 to 7.6% in 2015. The increase raised questions about overprescription.
Although studies have not borne out many of the other concerns raised about adverse reactions, they have shown that the drugs increase the risk for enteric infections, including infections by SARS-CoV-1, a virus that is related to the COVID-19 virus, SARS-CoV-2, Dr. Spiegel said.
SARS-CoV-2 uses the angiotensin-converting enzyme–2 receptor to invade enterocytes. Dr. Spiegel theorized that an increase in stomach pH above 3 as a result of use of PPIs might allow the virus to enter the GI tract more easily, leading to enteritis, colitis, and systemic spread to other organs, including the lungs. “There is a reason we have acid in our stomachs,” Dr. Spiegel said.
To see how PPI use relates to COVID-19 infections, Dr. Spiegel and his colleagues surveyed online a nationally representative sample of Americans between May 3 and June 24, 2020, as part of a larger survey on gastroenterologic health.
Participants answered questions about gastrointestinal symptoms, current use of PPIs, and COVID-19 test results. They also answered questions about histamine-2 receptor agonists (H2RAs), also known as H2 blockers, which are used to treat some of the same conditions as PPIs but that do not reduce stomach acid as much.
The surveying firm, Cint, contacted 264,058 people. Of the 86,602 eligible participants who completed the survey, 53,130 said they had experienced abdominal discomfort, acid reflux, heartburn, or regurgitation. These survey participants were subsequently asked about PPI and H2RA use.
Of these, 6.4% reported testing positive for SARS-CoV-2. The researchers adjusted for age, sex, race/ethnicity, education, marital status, household income, body mass index, smoking, alcohol consumption, U.S. region, insurance status, and the presence of irritable bowel syndrome, celiac disease, gastroesophageal reflux disease, liver cirrhosis, Crohn’s disease, ulcerative colitis, diabetes, and HIV/AIDS.
After adjusting for these factors, the researchers found that those who took PPIs up to once a day were twice as likely to have had a positive COVID-19 test result than those who did not take the drugs (odds ratio, 2.15; 95% confidence interval, 1.90-2.44).
Those who took PPIs twice a day were almost four times as likely to have tested positive for the disease (OR, 3.67; 95% CI, 2.93-4.60).
By contrast, those taking H2RA drugs once daily were 15% less likely to report a positive COVID-19 test result (OR, 0.85; 95% CI, 0.74-0.99). Research is currently underway to determine whether H2RAs might protect against the disease for reasons unrelated to pH balance.
Dr. Spiegel cautioned that the current data show only an association between PPI use and COVID-19 positivity; it cannot prove cause and effect.
Nevertheless, Dr. Spiegel said the findings should encourage physicians to prescribe PPIs only when clearly indicated. “If somebody is not yet on a PPI and you’re considering whether to start them on a PPI, it’s a good idea to consider H2 blockers,” he said.
People who need a daily dose of a PPI to control a severe condition can safely continue doing so, but such patients should take care to follow standard public health recommendations for avoiding exposure to the virus. These recommendations include wearing a mask, maintaining social distance, and washing hands frequently.
“People who are older, comorbid, or smokers – if they get infected, it could be severe,” he said. “[For] someone like that, it’s reasonable to ask, do we really need to be on twice-daily PPIs? There is good evidence that they are no better off than if they are taking once-daily doses.”
Brian Lacy, MD, PhD, a professor of medicine at the Mayo Clinic in Jacksonville, Fla., agreed that the study should prompt physicians to take a second look at their patients’ PPI prescriptions. “My view is that PPIs are frequently overused, and maybe this is one more piece of data that, if someone is on PPIs, maybe they don’t need to be on this medication.”
On the other hand, the drugs are important for treating conditions such as erosive esophagitis and healing ulcers, he said. The overall risk of contracting COVID-19 is low, so even this finding of a 3.7-fold increased risk should not lead patients or providers to stop taking or prescribing PPIs.
The study also lends support to the idea that the gastrointestinal tract could be involved in SARS-CoV-2 transmission, and it supports warnings about aerosols emitted from flushing toilets and through exhalation, Dr. Spiegel said. There is less evidence of the virus being transmitted through food. “It may not be fecal-oral; it may be fecal-respiratory,” he said.
The study was part of a larger project funded by Ironwood Pharmaceuticals. Dr. Spiegel reported relationships with Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Shire Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals. Dr. Lacy has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
In light of this finding, physicians should consider which patients truly need these powerful acid-lowering drugs, said Brennan Spiegel, MD, MSHS, AGAF, professor of medicine and public health at Cedars Sinai Medical Center in Los Angeles, Calif.
“All it means is that we’re going to have a conversation with our patients,” he said in an interview. “We don’t normally have that conversation because we don’t live in an environment with a high risk of enteric infection. But now we’re in a pandemic.”
The study by Dr. Spiegel and his colleagues was published online on July 7 in the American Journal of Gastroenterology.
Use of PPIs has skyrocketed over the past 2 decades. For ambulatory care visits, their use increased from 1.6% in 1998 to 7.6% in 2015. The increase raised questions about overprescription.
Although studies have not borne out many of the other concerns raised about adverse reactions, they have shown that the drugs increase the risk for enteric infections, including infections by SARS-CoV-1, a virus that is related to the COVID-19 virus, SARS-CoV-2, Dr. Spiegel said.
SARS-CoV-2 uses the angiotensin-converting enzyme–2 receptor to invade enterocytes. Dr. Spiegel theorized that an increase in stomach pH above 3 as a result of use of PPIs might allow the virus to enter the GI tract more easily, leading to enteritis, colitis, and systemic spread to other organs, including the lungs. “There is a reason we have acid in our stomachs,” Dr. Spiegel said.
To see how PPI use relates to COVID-19 infections, Dr. Spiegel and his colleagues surveyed online a nationally representative sample of Americans between May 3 and June 24, 2020, as part of a larger survey on gastroenterologic health.
Participants answered questions about gastrointestinal symptoms, current use of PPIs, and COVID-19 test results. They also answered questions about histamine-2 receptor agonists (H2RAs), also known as H2 blockers, which are used to treat some of the same conditions as PPIs but that do not reduce stomach acid as much.
The surveying firm, Cint, contacted 264,058 people. Of the 86,602 eligible participants who completed the survey, 53,130 said they had experienced abdominal discomfort, acid reflux, heartburn, or regurgitation. These survey participants were subsequently asked about PPI and H2RA use.
Of these, 6.4% reported testing positive for SARS-CoV-2. The researchers adjusted for age, sex, race/ethnicity, education, marital status, household income, body mass index, smoking, alcohol consumption, U.S. region, insurance status, and the presence of irritable bowel syndrome, celiac disease, gastroesophageal reflux disease, liver cirrhosis, Crohn’s disease, ulcerative colitis, diabetes, and HIV/AIDS.
After adjusting for these factors, the researchers found that those who took PPIs up to once a day were twice as likely to have had a positive COVID-19 test result than those who did not take the drugs (odds ratio, 2.15; 95% confidence interval, 1.90-2.44).
Those who took PPIs twice a day were almost four times as likely to have tested positive for the disease (OR, 3.67; 95% CI, 2.93-4.60).
By contrast, those taking H2RA drugs once daily were 15% less likely to report a positive COVID-19 test result (OR, 0.85; 95% CI, 0.74-0.99). Research is currently underway to determine whether H2RAs might protect against the disease for reasons unrelated to pH balance.
Dr. Spiegel cautioned that the current data show only an association between PPI use and COVID-19 positivity; it cannot prove cause and effect.
Nevertheless, Dr. Spiegel said the findings should encourage physicians to prescribe PPIs only when clearly indicated. “If somebody is not yet on a PPI and you’re considering whether to start them on a PPI, it’s a good idea to consider H2 blockers,” he said.
People who need a daily dose of a PPI to control a severe condition can safely continue doing so, but such patients should take care to follow standard public health recommendations for avoiding exposure to the virus. These recommendations include wearing a mask, maintaining social distance, and washing hands frequently.
“People who are older, comorbid, or smokers – if they get infected, it could be severe,” he said. “[For] someone like that, it’s reasonable to ask, do we really need to be on twice-daily PPIs? There is good evidence that they are no better off than if they are taking once-daily doses.”
Brian Lacy, MD, PhD, a professor of medicine at the Mayo Clinic in Jacksonville, Fla., agreed that the study should prompt physicians to take a second look at their patients’ PPI prescriptions. “My view is that PPIs are frequently overused, and maybe this is one more piece of data that, if someone is on PPIs, maybe they don’t need to be on this medication.”
On the other hand, the drugs are important for treating conditions such as erosive esophagitis and healing ulcers, he said. The overall risk of contracting COVID-19 is low, so even this finding of a 3.7-fold increased risk should not lead patients or providers to stop taking or prescribing PPIs.
The study also lends support to the idea that the gastrointestinal tract could be involved in SARS-CoV-2 transmission, and it supports warnings about aerosols emitted from flushing toilets and through exhalation, Dr. Spiegel said. There is less evidence of the virus being transmitted through food. “It may not be fecal-oral; it may be fecal-respiratory,” he said.
The study was part of a larger project funded by Ironwood Pharmaceuticals. Dr. Spiegel reported relationships with Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Shire Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals. Dr. Lacy has disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Infants around the world with bronchiolitis received excess tests despite guidelines
While guidelines for bronchiolitis aim to reduce gratuitous tests and treatments, one-third of infants presenting at EDs with bronchiolitis receive an unnecessary intervention, according to a new global study.

For infants with symptoms of bronchiolitis, viral testing, blood tests, and chest x-rays are discouraged in most cases. Antibiotics are not recommended as treatment.
In a study published in Pediatrics, Amy Zipursky, MD, of the Hospital for Sick Children and the University of Toronto, and colleagues, reviewed records for 2,359 infants aged 2-11 months diagnosed with bronchiolitis during the year 2013. The data came from a network of 38 EDs in the Australia, Canada, Ireland, New Zealand, Portugal, Spain, the United Kingdom, and the United States.
Dr. Zipursky and colleagues found that, while 8% of infants in the cohort had been treated with antibiotics, 33% had received at least one nonrecommended test, with rates ranging widely across regions. In the United Kingdom and Ireland, for example, only 15% received such a test, compared with 50% in Spain and Portugal.
Of the children given antibiotics, two-thirds had suspected bacterial infections, the researchers found. Antibiotic use was highest in the United States, at 11% of infants seen for bronchiolitis, and lowest in the United Kingdom and Ireland at 4%. Administration of chest x-rays – which occurred in nearly a quarter of the cohort – increased the likelihood of antibiotics being administered (odds ratio, 2.29; 95% confidence interval, 1.62-3.24) independent of fever or severe symptoms.
The most common nonrecommended tests performed in the study were:
- Nasopharyngeal viral testing without admission to hospital (n = 591).
- Chest x-ray without ICU admission (n = 507).
- Complete blood counts (n = 222).
- Blood cultures (n = 129).
- Urinalysis in the absence of fever (n = 86).
- Febrile infants 3 months of age or less had blood cultures (n = 49).
In some treatment centers the rate of nonrecommended tests performed was 6%, while others saw rates of 74%.
“Despite the evidence that laboratory testing rarely impacts bronchiolitis management and that bacterial infections in bronchiolitis are uncommon, our study reveals that these tests continue to be performed frequently in many parts of the world,” Dr. Zipursky and colleagues wrote in their analysis.
“Plausible reasons may include ‘automatic’ blood draws with intravenous placement, uncertainty about institutional policies, perceived need for reassurance about the diagnosis, perception of ‘doing something,’ and parental desire for a viral label,” the authors surmised. “Because parental pressure to provide interventions may be a driver of care in infants with bronchiolitis in some countries, ED clinicians need to have higher confidence in the evidence-based bronchiolitis care and convey this trust to families.”
The researchers listed among the weaknesses of their study its retrospective design, and that results from x-rays and lab tests performed were not available.
In an editorial comment accompanying the study, Joseph J. Zorc, MD, of Children’s Hospital of Philadelphia and the University of Pennsylvania in Philadelphia, noted that some of the regional differences seen in the study may be attributable to different clinical criteria used to diagnose bronchiolitis. In the United Kingdom, for example, national guidelines include the presence of crackles, while in North America guidelines focus on wheeze. “Perhaps clinicians in the United Kingdom accept the presence of crackles as an expected finding in infant with bronchiolitis and are less likely to order imaging,” Dr. Zorc said (Pediatrics. 2020 Jul 13;146[2]:e20193684).
He also pointed out that the coronavirus pandemic caused by SARS-CoV-2 (COVID- 19) could have an impact on global testing and treatment practices for bronchiolitis, as coronaviruses are a known cause of bronchiolitis. The Pediatric Emergency Research Network, comprising the 38 EDs from which Dr. Zipursky and colleagues drew their data, is conducting a prospective study looking at pediatric disease caused by SARS-CoV-2.
The “collaboration of international networks of pediatric emergency providers is an encouraging sign of potential opportunities to come ... [providing] an opportunity to evaluate variation that can lead to innovation,” Dr. Zorc concluded.
Dr. Zipursky and colleagues reported no external funding or relevant financial disclosures. Dr. Zorc reported no relevant conflicts of interest.
SOURCE: Zipursky A et al. Pediatrics. 2020 Jul 13;146(2):e2020002311.
While guidelines for bronchiolitis aim to reduce gratuitous tests and treatments, one-third of infants presenting at EDs with bronchiolitis receive an unnecessary intervention, according to a new global study.

For infants with symptoms of bronchiolitis, viral testing, blood tests, and chest x-rays are discouraged in most cases. Antibiotics are not recommended as treatment.
In a study published in Pediatrics, Amy Zipursky, MD, of the Hospital for Sick Children and the University of Toronto, and colleagues, reviewed records for 2,359 infants aged 2-11 months diagnosed with bronchiolitis during the year 2013. The data came from a network of 38 EDs in the Australia, Canada, Ireland, New Zealand, Portugal, Spain, the United Kingdom, and the United States.
Dr. Zipursky and colleagues found that, while 8% of infants in the cohort had been treated with antibiotics, 33% had received at least one nonrecommended test, with rates ranging widely across regions. In the United Kingdom and Ireland, for example, only 15% received such a test, compared with 50% in Spain and Portugal.
Of the children given antibiotics, two-thirds had suspected bacterial infections, the researchers found. Antibiotic use was highest in the United States, at 11% of infants seen for bronchiolitis, and lowest in the United Kingdom and Ireland at 4%. Administration of chest x-rays – which occurred in nearly a quarter of the cohort – increased the likelihood of antibiotics being administered (odds ratio, 2.29; 95% confidence interval, 1.62-3.24) independent of fever or severe symptoms.
The most common nonrecommended tests performed in the study were:
- Nasopharyngeal viral testing without admission to hospital (n = 591).
- Chest x-ray without ICU admission (n = 507).
- Complete blood counts (n = 222).
- Blood cultures (n = 129).
- Urinalysis in the absence of fever (n = 86).
- Febrile infants 3 months of age or less had blood cultures (n = 49).
In some treatment centers the rate of nonrecommended tests performed was 6%, while others saw rates of 74%.
“Despite the evidence that laboratory testing rarely impacts bronchiolitis management and that bacterial infections in bronchiolitis are uncommon, our study reveals that these tests continue to be performed frequently in many parts of the world,” Dr. Zipursky and colleagues wrote in their analysis.
“Plausible reasons may include ‘automatic’ blood draws with intravenous placement, uncertainty about institutional policies, perceived need for reassurance about the diagnosis, perception of ‘doing something,’ and parental desire for a viral label,” the authors surmised. “Because parental pressure to provide interventions may be a driver of care in infants with bronchiolitis in some countries, ED clinicians need to have higher confidence in the evidence-based bronchiolitis care and convey this trust to families.”
The researchers listed among the weaknesses of their study its retrospective design, and that results from x-rays and lab tests performed were not available.
In an editorial comment accompanying the study, Joseph J. Zorc, MD, of Children’s Hospital of Philadelphia and the University of Pennsylvania in Philadelphia, noted that some of the regional differences seen in the study may be attributable to different clinical criteria used to diagnose bronchiolitis. In the United Kingdom, for example, national guidelines include the presence of crackles, while in North America guidelines focus on wheeze. “Perhaps clinicians in the United Kingdom accept the presence of crackles as an expected finding in infant with bronchiolitis and are less likely to order imaging,” Dr. Zorc said (Pediatrics. 2020 Jul 13;146[2]:e20193684).
He also pointed out that the coronavirus pandemic caused by SARS-CoV-2 (COVID- 19) could have an impact on global testing and treatment practices for bronchiolitis, as coronaviruses are a known cause of bronchiolitis. The Pediatric Emergency Research Network, comprising the 38 EDs from which Dr. Zipursky and colleagues drew their data, is conducting a prospective study looking at pediatric disease caused by SARS-CoV-2.
The “collaboration of international networks of pediatric emergency providers is an encouraging sign of potential opportunities to come ... [providing] an opportunity to evaluate variation that can lead to innovation,” Dr. Zorc concluded.
Dr. Zipursky and colleagues reported no external funding or relevant financial disclosures. Dr. Zorc reported no relevant conflicts of interest.
SOURCE: Zipursky A et al. Pediatrics. 2020 Jul 13;146(2):e2020002311.
While guidelines for bronchiolitis aim to reduce gratuitous tests and treatments, one-third of infants presenting at EDs with bronchiolitis receive an unnecessary intervention, according to a new global study.

For infants with symptoms of bronchiolitis, viral testing, blood tests, and chest x-rays are discouraged in most cases. Antibiotics are not recommended as treatment.
In a study published in Pediatrics, Amy Zipursky, MD, of the Hospital for Sick Children and the University of Toronto, and colleagues, reviewed records for 2,359 infants aged 2-11 months diagnosed with bronchiolitis during the year 2013. The data came from a network of 38 EDs in the Australia, Canada, Ireland, New Zealand, Portugal, Spain, the United Kingdom, and the United States.
Dr. Zipursky and colleagues found that, while 8% of infants in the cohort had been treated with antibiotics, 33% had received at least one nonrecommended test, with rates ranging widely across regions. In the United Kingdom and Ireland, for example, only 15% received such a test, compared with 50% in Spain and Portugal.
Of the children given antibiotics, two-thirds had suspected bacterial infections, the researchers found. Antibiotic use was highest in the United States, at 11% of infants seen for bronchiolitis, and lowest in the United Kingdom and Ireland at 4%. Administration of chest x-rays – which occurred in nearly a quarter of the cohort – increased the likelihood of antibiotics being administered (odds ratio, 2.29; 95% confidence interval, 1.62-3.24) independent of fever or severe symptoms.
The most common nonrecommended tests performed in the study were:
- Nasopharyngeal viral testing without admission to hospital (n = 591).
- Chest x-ray without ICU admission (n = 507).
- Complete blood counts (n = 222).
- Blood cultures (n = 129).
- Urinalysis in the absence of fever (n = 86).
- Febrile infants 3 months of age or less had blood cultures (n = 49).
In some treatment centers the rate of nonrecommended tests performed was 6%, while others saw rates of 74%.
“Despite the evidence that laboratory testing rarely impacts bronchiolitis management and that bacterial infections in bronchiolitis are uncommon, our study reveals that these tests continue to be performed frequently in many parts of the world,” Dr. Zipursky and colleagues wrote in their analysis.
“Plausible reasons may include ‘automatic’ blood draws with intravenous placement, uncertainty about institutional policies, perceived need for reassurance about the diagnosis, perception of ‘doing something,’ and parental desire for a viral label,” the authors surmised. “Because parental pressure to provide interventions may be a driver of care in infants with bronchiolitis in some countries, ED clinicians need to have higher confidence in the evidence-based bronchiolitis care and convey this trust to families.”
The researchers listed among the weaknesses of their study its retrospective design, and that results from x-rays and lab tests performed were not available.
In an editorial comment accompanying the study, Joseph J. Zorc, MD, of Children’s Hospital of Philadelphia and the University of Pennsylvania in Philadelphia, noted that some of the regional differences seen in the study may be attributable to different clinical criteria used to diagnose bronchiolitis. In the United Kingdom, for example, national guidelines include the presence of crackles, while in North America guidelines focus on wheeze. “Perhaps clinicians in the United Kingdom accept the presence of crackles as an expected finding in infant with bronchiolitis and are less likely to order imaging,” Dr. Zorc said (Pediatrics. 2020 Jul 13;146[2]:e20193684).
He also pointed out that the coronavirus pandemic caused by SARS-CoV-2 (COVID- 19) could have an impact on global testing and treatment practices for bronchiolitis, as coronaviruses are a known cause of bronchiolitis. The Pediatric Emergency Research Network, comprising the 38 EDs from which Dr. Zipursky and colleagues drew their data, is conducting a prospective study looking at pediatric disease caused by SARS-CoV-2.
The “collaboration of international networks of pediatric emergency providers is an encouraging sign of potential opportunities to come ... [providing] an opportunity to evaluate variation that can lead to innovation,” Dr. Zorc concluded.
Dr. Zipursky and colleagues reported no external funding or relevant financial disclosures. Dr. Zorc reported no relevant conflicts of interest.
SOURCE: Zipursky A et al. Pediatrics. 2020 Jul 13;146(2):e2020002311.
FROM PEDIATRICS
Key clinical point:
Major finding: In a global cohort, 33% of infants received at least one nonrecommended test, most commonly viral tests, chest x-rays, and blood cultures.
Study details: A retrospective cohort of 2,359 infants aged 2-11 months seen in 38 EDs in developed countries.
Disclosures: Dr. Zipursky and colleagues reported no external funding or relevant financial disclosures.
Source: Zipursky A et al. Pediatrics. 2020 Jul 13;146(2):e2020002311.
COVID-19: Haiti is vulnerable, but the international community can help
Doctors Without Borders, other groups urged to mobilize
Do you want to know what keeps us up at night? As 4th-year medical students born, raised, and living in Haiti, we worry about the impact of COVID-19 on our patients.
The pandemic has shaken the world, and Haiti is no exception.
It has taken several months for the disease to spread, and it began with two confirmed cases, one from France and the other from Belgium, on March 19.1 Much of the spread of COVID-19 in Haiti has been tied to workers returning from the Dominican Republic. As of June 29, Haiti had 5,975 confirmed cases and 105 deaths.2 Of course, those numbers sound minuscule, compared with those in the United States, where the number of deaths from COVID-19 surpassed 100,000 several weeks ago. But the population of Haiti is 30 times smaller than that of the United States, and Haiti is the poorest country in the Western Hemisphere. We have watched in horror as the virus has ravaged marginalized groups in the United States and worry that it will do the same in our own country.
Just as the Haitian Ministry of Health worked with various groups to reach the 1-year free of cholera mark in Haiti, groups such as Doctors Without Borders must mobilize to rein in COVID-19.
Community transmission rapid
After the first two cases were confirmed, a state of health emergency was immediately declared. Haitian President Jovenel Moïse and other government officials called for the implementation of several measures aimed at limiting the spread of COVID-19.
Schools, universities, clinical training programs, vocational centers, factories, airports, and ports, except for the transport of goods, were all ordered to close until further notice. Gatherings of larger than 10 people were banned. A curfew from 8 p.m. EST time to 5 a.m. EST was imposed. Measures such as those encouraged by U.S. Centers for Disease Control and Prevention, such as hand washing, physical distancing, and staying at home were also encouraged by the Haitian Ministry of Health. Mask wearing in public places was deemed mandatory.
The latest testing data show that community spread has been occurring among the Haitian population at a rapid rate. According to Jean William Pape, MD, Haiti’s top infectious diseases expert and founder of GHESKIO, an iconic infectious disease center that cares for people with HIV-AIDS and tuberculosis, a COVID-19 simulation from Cornell University in New York shows that about 35% of the Haitian population will be infected by the end of August 2020. A simulation by the University of Oxford (England) paints an even more dire picture. That simulation shows that 86% of the population could be infected, More than 9,000 additional hospital beds would be needed, and 20,000 people would be likely to die from COVID-19, Dr. Pape said in an interview with Haiti’s Nouvelliste newspaper.3
Medical response
We know that there is a global shortage of health care workers,4 and Haiti is no exception. According to a 2018 report from the Haitian Ministry of Health, the country has 11,775 health care professionals, including about 3,354 medical doctors, to care for more than 11 million people. That translates to about 23.4 physicians per 100,000.5
The pandemic has led some members of this already anemic health care workforce to stay home because of a lack of personal protective equipment. Others, because of reduced hospital or clinic budgets, have been furloughed, making the COVID-19 national health emergency even harder to manage.
But a severe health care shortage is not the only challenge facing Haiti. It spends about $131 U.S. per capita, which makes Haiti one of most vulnerable among low- and middle-income countries in the world. As a poor country,7 its health care infrastructure is among the most inadequate and weakest. Prior to COVID-19, medical advocacy groups already had started movements and strikes demanding that the government improve the health care system. The country’s precarious health care infrastructure includes a lack of hospital beds, and basic medical supplies and equipment, such as oxygen and ventilators.8 The emergence of COVID-19 has only exacerbated the situation.
Clinical training programs have been suspended, many doctors and nurses are on quarantine, and some hospitals and clinics are closing. We have witnessed makeshift voodoo clinics built by Haitian voodoo leaders to receive, hospitalize, and treat COVID-19 patients through rituals and herbal remedies. In some areas of the country, residents have protested against the opening of several COVID-19 treatment and management centers.
Unique cultural challenges
Public health officials around the world are facing challenges persuading citizens to engage in behaviors that could protect them from the virus.
Just as in America, where many people opt to not wear face coverings9,10 despite the public health risks, deep distrust of the Haitian government has undermined the messages of President Moïse and public health officials about the role of masks in limiting the spread of COVID. We see large numbers of unmasked people on the streets in the informal markets every day. Crammed tap-taps and overloaded motorcycles are moving everywhere. This also could be tied to cultural attitudes about COVID that persist among some Haitians. For example, many people with signs and symptoms of COVID-19 are afraid of going to the hospital to get tested and receive care, and resort to going to the voodoo clinics. Along with rituals, voodoo priests have been serving up teas with ingredients, including moringa, eucalyptus, ginger, and honey to those seeking COVID-19 care in the centers. The voodoo priests claim that the teas they serve strengthen the immune system.
In addition, it is difficult for poor people who live in small quarters with several other people to adhere to physical distancing.11
Stigma and violence
Other barriers in the fight against COVID-19 in Haiti are stigma and violence. If widespread testing were available, some Haitians would opt not to do so – despite clear signs and symptoms of the infection. Some people who would get tested if they could are afraid to do so because of fears tied to being attacked by neighbors.
When Haitian University professor Bellamy Nelson and his girlfriend returned to Haiti from the United States in March and began experiencing some pain and fever, he experienced attacks from neighbors, he said in an interview. He said neighbors threatened to burn down his house. When an ambulance arrived at his house to transport him to a hospital, it had to drive through back roads to avoid people armed with rocks, fire, and machetes, he told us. No hospital wanted to admit him. Eventually, Professor Nelson self-quarantined at home, he said.
In another incident, a national ambulance center in Gonaïves, a town toward the northern region of Haiti, reportedly was vandalized, because COVID-19 equipment and supplies used to treat people had been stored there. Hospital Bernard Mevs, along with many other hospitals, was forced by the area’s residents to suspend the plan to open a center for COVID-19 management. Threats to burn down the hospitals caused the leaders of the hospitals to back down and give up a plan to build a 20-bed COVID-19 response center.
Maternal health
Another concern we have about the pandemic is the risk it could be to pregnant women. On average, 94,000 deaths occur annually in Haiti. Out of this number, maternal mortality accounts for 1,000. In 2017, for every 100,000 live births for women of reproductive age from 15 to 49 years old, 480 women died. In contrast, in the Dominican Republic, 95 women died per 100,000 that same year. In the United States, 19 died, and in Norway, no more than 2 died that year.12
Some of the primary factors contributing to the crisis are limited accessibility, inadequate health care facilities, and an inadequate number of trained health care practitioners; low percentages of skilled attendants at deliveries and of prenatal and postnatal visits; and high numbers of high-risk deliveries in nonqualified health facilities.
During the COVID-19 national health emergency, with most hospitals reducing their health care personnel either because of budget-related reasons or because they are on quarantine, this maternal-fetal health crisis has escalated.
One of the biggest hospitals in Jacmel, a town in the southern region of Haiti, has stopped its prenatal care program. In Delmas, the city with the highest incidence and prevalence of COVID-19, Hôpital Universitaire de la Paix has reduced this program to 50% of its capacity and gynecologic care has been completely suspended. Hôpital St. Luc, one of the first hospitals in the western region of Haiti to open its doors to care for COVID-19 patients, has recently shut down the entire maternal-fetal department.
So, access to prenatal and postnatal care, including the ability to deliver babies in health care institutions, is significantly reduced because of COVID-19. This leaves thousands of already vulnerable pregnant women at risk and having to deliver domestically with little to no health care professional assistance. We worry that, in light of the data, more women and babies will die because of the COVID-19 pandemic.
A call to action
Despite these conditions, there are reasons for hope. Various groups, both from the international community and locally have mobilized to respond to the pandemic.
International health care organizations such as Doctors Without Borders and Partners in Health, and local groups such as GHESKIO, the St. Luke Foundation for Haiti, and others have been collaborating with the Haitian Ministry of Health to devise and strategic plans and deploy valuable resources with the common goal of saving lives from COVID-19.
GHESKIO, for example, under Dr. Pape’s leadership, currently has one of the three COVID-19 testing centers in the country. It also has two COVID-19 treatment centers in full operation, in Port-au-Prince, the capital city, managing and treating 520 patients with confirmed COVID-19. GHESKIO, which has been in the front lines of previous major infectious disease outbreaks,13 has trained about 200 clinicians from both public and private health care institutions to care for COVID-19 patients.
Doctors Without Borders has been investing in efforts to support the Ministry of Health by converting and renovating its Burn Center in Drouillard, a small section of the city of Cité Soleil, one of the country’s biggest slums. In May, as part of its COVID-19 response, it launched a 20-bed capacity center that can accommodate up to 45 beds to care for patients who have tested positive for COVID-19.
Partners in Health, the Boston-based nonprofit health care organization cofounded in 1987 by American anthropologist and infectious disease specialist, Paul Farmer, MD, and the largest nonprofit health care provider in Haiti, also joined the Ministry of Health through its national and public health efforts to tackle COVID-19 in Haiti. Partners in Health, through its sister organization, Zanmi Lasante, has pioneered the movement of diagnosing and treating people with HIV-AIDS and TB. Since the late 1990s, its efforts against both infectious diseases have helped 15,000 HIV-positive patients begin and remain on treatment. And every year, 1,500 TB patients have started treatment on the path to a cure.
Early in the pandemic in Haiti, Partners in Health, through its state-of-the-art 300-bed university hospital (Hôpital Universitaire de Mirebalais de Mirebalais), was the first to open a COVID-19 center with a 20-bed capacity and has been caring for COVID-19 patients since then. In June, Partners in Health supported and inaugurated the renovation of the internal medicine department at one of its affiliated community hospitals, Hôpital Saint-Nicolas de Saint Marc. That department will have a 24-bed capacity that can extend up to 36 beds to manage and treat COVID-19 patients.
In total, currently, 26 COVID-19 centers with a capacity of 1,011 beds are available to serve, manage, and treat Haitian patients affected with COVID-19. But are those efforts enough? No.
Haiti, as a weak state even before COVID-19, continues to need funding from the international community so it can strengthen its health care infrastructure to be effective and strong in fighting against COVID-19.
In addition, we would like to see preventive initiatives implemented on the local level. Our family has taken on a role that, we think, could help conquer COVID-19 if others followed suit on a large scale.
As part of our contribution in tackling COVID-19, the two of us have launched a small-scale community experiment. We have educated our family in Delmas about COVID-19 and subsequently launched an awareness campaign in the community. We dispatched small groups that go door to door in the community to educate neighbors about the disease in an effort to help them understand that COVID-19 is real and it is normal for people that feel they may have the disease to seek medical care. This approach helps suppress the transmission of the virus. This pilot project could be reproduced in several other communities. It is easy to operate, rapid, effective, and cost-free. The community has been very receptive to and grateful for our efforts.
Like other countries across the world, Haiti was not ready for COVID-19. But we are confident that, with help from the international community, organizations such as GHESKIO,14 and with due diligence on the local level, we are strong and resilient enough to beat COVID. We must act together – quickly.
References
1. Sénat JD. Coronavirus: 2 cas confirmés en Haïti, Jovenel Moïse décrète l’état d’ur-gence sanitaire. 2020 Le Nouvelliste.
2. Haitian Ministry of Health.
3. “Entre appel a la solidarite et de sombres previsions, le Dr William Pape fait le point.” Le Nouvelliste.
4. Darzi A and Evans T. Lancet. 2016 Nov-Dec 26. 388;10060:2576-7.
5. Rapport Statistique 2018. 2019 Republic of Haiti.
6. Sentlinger K. “Water Crisis in Haiti.” The Water Project.
7. The World Bank in Haiti. worldbank.org.
8. Cenat JM. Travel Med Infect Dis. 2020 Mar 28. doi: 10.1016/jtmaid.2020.101684.
9. Block D. “Why some Americans resist wearing face masks.” voanews.com. 2020 May 31.
10. Panceski B and Douglas J. “Masks could help stop coronavirus. So why are they still controversial?” wsj.com. Updated 29 Jun 2020.
11. Bojarski S. “Social distancing: A luxury Haiti’s poor cannot afford. The Haitian Times. 2020 Apr.
12. World Health Organization, UNICEF, World Bank Group, and the U.N. Population Division. Maternal mortality ratio, Haiti.
13. Feliciano I and Kargbo C. “As COVID cases surge, Haiti’s Dr. Pape is on the front line again.” PBS NewsHour Weekend. 2020 Jun 13.
14. Liautaud B and Deschamps MM. New Engl J Med. 2020 Jun 16.
Mr. Dorcela is a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince, Haiti. He also is a medical intern at Unité de Médecine Familiale Hôpital Saint Nicolas in Saint-Marc. Mr. Dorcela has no disclosures. Mr. St. Jean, who is Mr. Dorcela’s brother, is also a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince. He has no disclosures.
Doctors Without Borders, other groups urged to mobilize
Doctors Without Borders, other groups urged to mobilize
Do you want to know what keeps us up at night? As 4th-year medical students born, raised, and living in Haiti, we worry about the impact of COVID-19 on our patients.
The pandemic has shaken the world, and Haiti is no exception.
It has taken several months for the disease to spread, and it began with two confirmed cases, one from France and the other from Belgium, on March 19.1 Much of the spread of COVID-19 in Haiti has been tied to workers returning from the Dominican Republic. As of June 29, Haiti had 5,975 confirmed cases and 105 deaths.2 Of course, those numbers sound minuscule, compared with those in the United States, where the number of deaths from COVID-19 surpassed 100,000 several weeks ago. But the population of Haiti is 30 times smaller than that of the United States, and Haiti is the poorest country in the Western Hemisphere. We have watched in horror as the virus has ravaged marginalized groups in the United States and worry that it will do the same in our own country.
Just as the Haitian Ministry of Health worked with various groups to reach the 1-year free of cholera mark in Haiti, groups such as Doctors Without Borders must mobilize to rein in COVID-19.
Community transmission rapid
After the first two cases were confirmed, a state of health emergency was immediately declared. Haitian President Jovenel Moïse and other government officials called for the implementation of several measures aimed at limiting the spread of COVID-19.
Schools, universities, clinical training programs, vocational centers, factories, airports, and ports, except for the transport of goods, were all ordered to close until further notice. Gatherings of larger than 10 people were banned. A curfew from 8 p.m. EST time to 5 a.m. EST was imposed. Measures such as those encouraged by U.S. Centers for Disease Control and Prevention, such as hand washing, physical distancing, and staying at home were also encouraged by the Haitian Ministry of Health. Mask wearing in public places was deemed mandatory.
The latest testing data show that community spread has been occurring among the Haitian population at a rapid rate. According to Jean William Pape, MD, Haiti’s top infectious diseases expert and founder of GHESKIO, an iconic infectious disease center that cares for people with HIV-AIDS and tuberculosis, a COVID-19 simulation from Cornell University in New York shows that about 35% of the Haitian population will be infected by the end of August 2020. A simulation by the University of Oxford (England) paints an even more dire picture. That simulation shows that 86% of the population could be infected, More than 9,000 additional hospital beds would be needed, and 20,000 people would be likely to die from COVID-19, Dr. Pape said in an interview with Haiti’s Nouvelliste newspaper.3
Medical response
We know that there is a global shortage of health care workers,4 and Haiti is no exception. According to a 2018 report from the Haitian Ministry of Health, the country has 11,775 health care professionals, including about 3,354 medical doctors, to care for more than 11 million people. That translates to about 23.4 physicians per 100,000.5
The pandemic has led some members of this already anemic health care workforce to stay home because of a lack of personal protective equipment. Others, because of reduced hospital or clinic budgets, have been furloughed, making the COVID-19 national health emergency even harder to manage.
But a severe health care shortage is not the only challenge facing Haiti. It spends about $131 U.S. per capita, which makes Haiti one of most vulnerable among low- and middle-income countries in the world. As a poor country,7 its health care infrastructure is among the most inadequate and weakest. Prior to COVID-19, medical advocacy groups already had started movements and strikes demanding that the government improve the health care system. The country’s precarious health care infrastructure includes a lack of hospital beds, and basic medical supplies and equipment, such as oxygen and ventilators.8 The emergence of COVID-19 has only exacerbated the situation.
Clinical training programs have been suspended, many doctors and nurses are on quarantine, and some hospitals and clinics are closing. We have witnessed makeshift voodoo clinics built by Haitian voodoo leaders to receive, hospitalize, and treat COVID-19 patients through rituals and herbal remedies. In some areas of the country, residents have protested against the opening of several COVID-19 treatment and management centers.
Unique cultural challenges
Public health officials around the world are facing challenges persuading citizens to engage in behaviors that could protect them from the virus.
Just as in America, where many people opt to not wear face coverings9,10 despite the public health risks, deep distrust of the Haitian government has undermined the messages of President Moïse and public health officials about the role of masks in limiting the spread of COVID. We see large numbers of unmasked people on the streets in the informal markets every day. Crammed tap-taps and overloaded motorcycles are moving everywhere. This also could be tied to cultural attitudes about COVID that persist among some Haitians. For example, many people with signs and symptoms of COVID-19 are afraid of going to the hospital to get tested and receive care, and resort to going to the voodoo clinics. Along with rituals, voodoo priests have been serving up teas with ingredients, including moringa, eucalyptus, ginger, and honey to those seeking COVID-19 care in the centers. The voodoo priests claim that the teas they serve strengthen the immune system.
In addition, it is difficult for poor people who live in small quarters with several other people to adhere to physical distancing.11
Stigma and violence
Other barriers in the fight against COVID-19 in Haiti are stigma and violence. If widespread testing were available, some Haitians would opt not to do so – despite clear signs and symptoms of the infection. Some people who would get tested if they could are afraid to do so because of fears tied to being attacked by neighbors.
When Haitian University professor Bellamy Nelson and his girlfriend returned to Haiti from the United States in March and began experiencing some pain and fever, he experienced attacks from neighbors, he said in an interview. He said neighbors threatened to burn down his house. When an ambulance arrived at his house to transport him to a hospital, it had to drive through back roads to avoid people armed with rocks, fire, and machetes, he told us. No hospital wanted to admit him. Eventually, Professor Nelson self-quarantined at home, he said.
In another incident, a national ambulance center in Gonaïves, a town toward the northern region of Haiti, reportedly was vandalized, because COVID-19 equipment and supplies used to treat people had been stored there. Hospital Bernard Mevs, along with many other hospitals, was forced by the area’s residents to suspend the plan to open a center for COVID-19 management. Threats to burn down the hospitals caused the leaders of the hospitals to back down and give up a plan to build a 20-bed COVID-19 response center.
Maternal health
Another concern we have about the pandemic is the risk it could be to pregnant women. On average, 94,000 deaths occur annually in Haiti. Out of this number, maternal mortality accounts for 1,000. In 2017, for every 100,000 live births for women of reproductive age from 15 to 49 years old, 480 women died. In contrast, in the Dominican Republic, 95 women died per 100,000 that same year. In the United States, 19 died, and in Norway, no more than 2 died that year.12
Some of the primary factors contributing to the crisis are limited accessibility, inadequate health care facilities, and an inadequate number of trained health care practitioners; low percentages of skilled attendants at deliveries and of prenatal and postnatal visits; and high numbers of high-risk deliveries in nonqualified health facilities.
During the COVID-19 national health emergency, with most hospitals reducing their health care personnel either because of budget-related reasons or because they are on quarantine, this maternal-fetal health crisis has escalated.
One of the biggest hospitals in Jacmel, a town in the southern region of Haiti, has stopped its prenatal care program. In Delmas, the city with the highest incidence and prevalence of COVID-19, Hôpital Universitaire de la Paix has reduced this program to 50% of its capacity and gynecologic care has been completely suspended. Hôpital St. Luc, one of the first hospitals in the western region of Haiti to open its doors to care for COVID-19 patients, has recently shut down the entire maternal-fetal department.
So, access to prenatal and postnatal care, including the ability to deliver babies in health care institutions, is significantly reduced because of COVID-19. This leaves thousands of already vulnerable pregnant women at risk and having to deliver domestically with little to no health care professional assistance. We worry that, in light of the data, more women and babies will die because of the COVID-19 pandemic.
A call to action
Despite these conditions, there are reasons for hope. Various groups, both from the international community and locally have mobilized to respond to the pandemic.
International health care organizations such as Doctors Without Borders and Partners in Health, and local groups such as GHESKIO, the St. Luke Foundation for Haiti, and others have been collaborating with the Haitian Ministry of Health to devise and strategic plans and deploy valuable resources with the common goal of saving lives from COVID-19.
GHESKIO, for example, under Dr. Pape’s leadership, currently has one of the three COVID-19 testing centers in the country. It also has two COVID-19 treatment centers in full operation, in Port-au-Prince, the capital city, managing and treating 520 patients with confirmed COVID-19. GHESKIO, which has been in the front lines of previous major infectious disease outbreaks,13 has trained about 200 clinicians from both public and private health care institutions to care for COVID-19 patients.
Doctors Without Borders has been investing in efforts to support the Ministry of Health by converting and renovating its Burn Center in Drouillard, a small section of the city of Cité Soleil, one of the country’s biggest slums. In May, as part of its COVID-19 response, it launched a 20-bed capacity center that can accommodate up to 45 beds to care for patients who have tested positive for COVID-19.
Partners in Health, the Boston-based nonprofit health care organization cofounded in 1987 by American anthropologist and infectious disease specialist, Paul Farmer, MD, and the largest nonprofit health care provider in Haiti, also joined the Ministry of Health through its national and public health efforts to tackle COVID-19 in Haiti. Partners in Health, through its sister organization, Zanmi Lasante, has pioneered the movement of diagnosing and treating people with HIV-AIDS and TB. Since the late 1990s, its efforts against both infectious diseases have helped 15,000 HIV-positive patients begin and remain on treatment. And every year, 1,500 TB patients have started treatment on the path to a cure.
Early in the pandemic in Haiti, Partners in Health, through its state-of-the-art 300-bed university hospital (Hôpital Universitaire de Mirebalais de Mirebalais), was the first to open a COVID-19 center with a 20-bed capacity and has been caring for COVID-19 patients since then. In June, Partners in Health supported and inaugurated the renovation of the internal medicine department at one of its affiliated community hospitals, Hôpital Saint-Nicolas de Saint Marc. That department will have a 24-bed capacity that can extend up to 36 beds to manage and treat COVID-19 patients.
In total, currently, 26 COVID-19 centers with a capacity of 1,011 beds are available to serve, manage, and treat Haitian patients affected with COVID-19. But are those efforts enough? No.
Haiti, as a weak state even before COVID-19, continues to need funding from the international community so it can strengthen its health care infrastructure to be effective and strong in fighting against COVID-19.
In addition, we would like to see preventive initiatives implemented on the local level. Our family has taken on a role that, we think, could help conquer COVID-19 if others followed suit on a large scale.
As part of our contribution in tackling COVID-19, the two of us have launched a small-scale community experiment. We have educated our family in Delmas about COVID-19 and subsequently launched an awareness campaign in the community. We dispatched small groups that go door to door in the community to educate neighbors about the disease in an effort to help them understand that COVID-19 is real and it is normal for people that feel they may have the disease to seek medical care. This approach helps suppress the transmission of the virus. This pilot project could be reproduced in several other communities. It is easy to operate, rapid, effective, and cost-free. The community has been very receptive to and grateful for our efforts.
Like other countries across the world, Haiti was not ready for COVID-19. But we are confident that, with help from the international community, organizations such as GHESKIO,14 and with due diligence on the local level, we are strong and resilient enough to beat COVID. We must act together – quickly.
References
1. Sénat JD. Coronavirus: 2 cas confirmés en Haïti, Jovenel Moïse décrète l’état d’ur-gence sanitaire. 2020 Le Nouvelliste.
2. Haitian Ministry of Health.
3. “Entre appel a la solidarite et de sombres previsions, le Dr William Pape fait le point.” Le Nouvelliste.
4. Darzi A and Evans T. Lancet. 2016 Nov-Dec 26. 388;10060:2576-7.
5. Rapport Statistique 2018. 2019 Republic of Haiti.
6. Sentlinger K. “Water Crisis in Haiti.” The Water Project.
7. The World Bank in Haiti. worldbank.org.
8. Cenat JM. Travel Med Infect Dis. 2020 Mar 28. doi: 10.1016/jtmaid.2020.101684.
9. Block D. “Why some Americans resist wearing face masks.” voanews.com. 2020 May 31.
10. Panceski B and Douglas J. “Masks could help stop coronavirus. So why are they still controversial?” wsj.com. Updated 29 Jun 2020.
11. Bojarski S. “Social distancing: A luxury Haiti’s poor cannot afford. The Haitian Times. 2020 Apr.
12. World Health Organization, UNICEF, World Bank Group, and the U.N. Population Division. Maternal mortality ratio, Haiti.
13. Feliciano I and Kargbo C. “As COVID cases surge, Haiti’s Dr. Pape is on the front line again.” PBS NewsHour Weekend. 2020 Jun 13.
14. Liautaud B and Deschamps MM. New Engl J Med. 2020 Jun 16.
Mr. Dorcela is a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince, Haiti. He also is a medical intern at Unité de Médecine Familiale Hôpital Saint Nicolas in Saint-Marc. Mr. Dorcela has no disclosures. Mr. St. Jean, who is Mr. Dorcela’s brother, is also a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince. He has no disclosures.
Do you want to know what keeps us up at night? As 4th-year medical students born, raised, and living in Haiti, we worry about the impact of COVID-19 on our patients.
The pandemic has shaken the world, and Haiti is no exception.
It has taken several months for the disease to spread, and it began with two confirmed cases, one from France and the other from Belgium, on March 19.1 Much of the spread of COVID-19 in Haiti has been tied to workers returning from the Dominican Republic. As of June 29, Haiti had 5,975 confirmed cases and 105 deaths.2 Of course, those numbers sound minuscule, compared with those in the United States, where the number of deaths from COVID-19 surpassed 100,000 several weeks ago. But the population of Haiti is 30 times smaller than that of the United States, and Haiti is the poorest country in the Western Hemisphere. We have watched in horror as the virus has ravaged marginalized groups in the United States and worry that it will do the same in our own country.
Just as the Haitian Ministry of Health worked with various groups to reach the 1-year free of cholera mark in Haiti, groups such as Doctors Without Borders must mobilize to rein in COVID-19.
Community transmission rapid
After the first two cases were confirmed, a state of health emergency was immediately declared. Haitian President Jovenel Moïse and other government officials called for the implementation of several measures aimed at limiting the spread of COVID-19.
Schools, universities, clinical training programs, vocational centers, factories, airports, and ports, except for the transport of goods, were all ordered to close until further notice. Gatherings of larger than 10 people were banned. A curfew from 8 p.m. EST time to 5 a.m. EST was imposed. Measures such as those encouraged by U.S. Centers for Disease Control and Prevention, such as hand washing, physical distancing, and staying at home were also encouraged by the Haitian Ministry of Health. Mask wearing in public places was deemed mandatory.
The latest testing data show that community spread has been occurring among the Haitian population at a rapid rate. According to Jean William Pape, MD, Haiti’s top infectious diseases expert and founder of GHESKIO, an iconic infectious disease center that cares for people with HIV-AIDS and tuberculosis, a COVID-19 simulation from Cornell University in New York shows that about 35% of the Haitian population will be infected by the end of August 2020. A simulation by the University of Oxford (England) paints an even more dire picture. That simulation shows that 86% of the population could be infected, More than 9,000 additional hospital beds would be needed, and 20,000 people would be likely to die from COVID-19, Dr. Pape said in an interview with Haiti’s Nouvelliste newspaper.3
Medical response
We know that there is a global shortage of health care workers,4 and Haiti is no exception. According to a 2018 report from the Haitian Ministry of Health, the country has 11,775 health care professionals, including about 3,354 medical doctors, to care for more than 11 million people. That translates to about 23.4 physicians per 100,000.5
The pandemic has led some members of this already anemic health care workforce to stay home because of a lack of personal protective equipment. Others, because of reduced hospital or clinic budgets, have been furloughed, making the COVID-19 national health emergency even harder to manage.
But a severe health care shortage is not the only challenge facing Haiti. It spends about $131 U.S. per capita, which makes Haiti one of most vulnerable among low- and middle-income countries in the world. As a poor country,7 its health care infrastructure is among the most inadequate and weakest. Prior to COVID-19, medical advocacy groups already had started movements and strikes demanding that the government improve the health care system. The country’s precarious health care infrastructure includes a lack of hospital beds, and basic medical supplies and equipment, such as oxygen and ventilators.8 The emergence of COVID-19 has only exacerbated the situation.
Clinical training programs have been suspended, many doctors and nurses are on quarantine, and some hospitals and clinics are closing. We have witnessed makeshift voodoo clinics built by Haitian voodoo leaders to receive, hospitalize, and treat COVID-19 patients through rituals and herbal remedies. In some areas of the country, residents have protested against the opening of several COVID-19 treatment and management centers.
Unique cultural challenges
Public health officials around the world are facing challenges persuading citizens to engage in behaviors that could protect them from the virus.
Just as in America, where many people opt to not wear face coverings9,10 despite the public health risks, deep distrust of the Haitian government has undermined the messages of President Moïse and public health officials about the role of masks in limiting the spread of COVID. We see large numbers of unmasked people on the streets in the informal markets every day. Crammed tap-taps and overloaded motorcycles are moving everywhere. This also could be tied to cultural attitudes about COVID that persist among some Haitians. For example, many people with signs and symptoms of COVID-19 are afraid of going to the hospital to get tested and receive care, and resort to going to the voodoo clinics. Along with rituals, voodoo priests have been serving up teas with ingredients, including moringa, eucalyptus, ginger, and honey to those seeking COVID-19 care in the centers. The voodoo priests claim that the teas they serve strengthen the immune system.
In addition, it is difficult for poor people who live in small quarters with several other people to adhere to physical distancing.11
Stigma and violence
Other barriers in the fight against COVID-19 in Haiti are stigma and violence. If widespread testing were available, some Haitians would opt not to do so – despite clear signs and symptoms of the infection. Some people who would get tested if they could are afraid to do so because of fears tied to being attacked by neighbors.
When Haitian University professor Bellamy Nelson and his girlfriend returned to Haiti from the United States in March and began experiencing some pain and fever, he experienced attacks from neighbors, he said in an interview. He said neighbors threatened to burn down his house. When an ambulance arrived at his house to transport him to a hospital, it had to drive through back roads to avoid people armed with rocks, fire, and machetes, he told us. No hospital wanted to admit him. Eventually, Professor Nelson self-quarantined at home, he said.
In another incident, a national ambulance center in Gonaïves, a town toward the northern region of Haiti, reportedly was vandalized, because COVID-19 equipment and supplies used to treat people had been stored there. Hospital Bernard Mevs, along with many other hospitals, was forced by the area’s residents to suspend the plan to open a center for COVID-19 management. Threats to burn down the hospitals caused the leaders of the hospitals to back down and give up a plan to build a 20-bed COVID-19 response center.
Maternal health
Another concern we have about the pandemic is the risk it could be to pregnant women. On average, 94,000 deaths occur annually in Haiti. Out of this number, maternal mortality accounts for 1,000. In 2017, for every 100,000 live births for women of reproductive age from 15 to 49 years old, 480 women died. In contrast, in the Dominican Republic, 95 women died per 100,000 that same year. In the United States, 19 died, and in Norway, no more than 2 died that year.12
Some of the primary factors contributing to the crisis are limited accessibility, inadequate health care facilities, and an inadequate number of trained health care practitioners; low percentages of skilled attendants at deliveries and of prenatal and postnatal visits; and high numbers of high-risk deliveries in nonqualified health facilities.
During the COVID-19 national health emergency, with most hospitals reducing their health care personnel either because of budget-related reasons or because they are on quarantine, this maternal-fetal health crisis has escalated.
One of the biggest hospitals in Jacmel, a town in the southern region of Haiti, has stopped its prenatal care program. In Delmas, the city with the highest incidence and prevalence of COVID-19, Hôpital Universitaire de la Paix has reduced this program to 50% of its capacity and gynecologic care has been completely suspended. Hôpital St. Luc, one of the first hospitals in the western region of Haiti to open its doors to care for COVID-19 patients, has recently shut down the entire maternal-fetal department.
So, access to prenatal and postnatal care, including the ability to deliver babies in health care institutions, is significantly reduced because of COVID-19. This leaves thousands of already vulnerable pregnant women at risk and having to deliver domestically with little to no health care professional assistance. We worry that, in light of the data, more women and babies will die because of the COVID-19 pandemic.
A call to action
Despite these conditions, there are reasons for hope. Various groups, both from the international community and locally have mobilized to respond to the pandemic.
International health care organizations such as Doctors Without Borders and Partners in Health, and local groups such as GHESKIO, the St. Luke Foundation for Haiti, and others have been collaborating with the Haitian Ministry of Health to devise and strategic plans and deploy valuable resources with the common goal of saving lives from COVID-19.
GHESKIO, for example, under Dr. Pape’s leadership, currently has one of the three COVID-19 testing centers in the country. It also has two COVID-19 treatment centers in full operation, in Port-au-Prince, the capital city, managing and treating 520 patients with confirmed COVID-19. GHESKIO, which has been in the front lines of previous major infectious disease outbreaks,13 has trained about 200 clinicians from both public and private health care institutions to care for COVID-19 patients.
Doctors Without Borders has been investing in efforts to support the Ministry of Health by converting and renovating its Burn Center in Drouillard, a small section of the city of Cité Soleil, one of the country’s biggest slums. In May, as part of its COVID-19 response, it launched a 20-bed capacity center that can accommodate up to 45 beds to care for patients who have tested positive for COVID-19.
Partners in Health, the Boston-based nonprofit health care organization cofounded in 1987 by American anthropologist and infectious disease specialist, Paul Farmer, MD, and the largest nonprofit health care provider in Haiti, also joined the Ministry of Health through its national and public health efforts to tackle COVID-19 in Haiti. Partners in Health, through its sister organization, Zanmi Lasante, has pioneered the movement of diagnosing and treating people with HIV-AIDS and TB. Since the late 1990s, its efforts against both infectious diseases have helped 15,000 HIV-positive patients begin and remain on treatment. And every year, 1,500 TB patients have started treatment on the path to a cure.
Early in the pandemic in Haiti, Partners in Health, through its state-of-the-art 300-bed university hospital (Hôpital Universitaire de Mirebalais de Mirebalais), was the first to open a COVID-19 center with a 20-bed capacity and has been caring for COVID-19 patients since then. In June, Partners in Health supported and inaugurated the renovation of the internal medicine department at one of its affiliated community hospitals, Hôpital Saint-Nicolas de Saint Marc. That department will have a 24-bed capacity that can extend up to 36 beds to manage and treat COVID-19 patients.
In total, currently, 26 COVID-19 centers with a capacity of 1,011 beds are available to serve, manage, and treat Haitian patients affected with COVID-19. But are those efforts enough? No.
Haiti, as a weak state even before COVID-19, continues to need funding from the international community so it can strengthen its health care infrastructure to be effective and strong in fighting against COVID-19.
In addition, we would like to see preventive initiatives implemented on the local level. Our family has taken on a role that, we think, could help conquer COVID-19 if others followed suit on a large scale.
As part of our contribution in tackling COVID-19, the two of us have launched a small-scale community experiment. We have educated our family in Delmas about COVID-19 and subsequently launched an awareness campaign in the community. We dispatched small groups that go door to door in the community to educate neighbors about the disease in an effort to help them understand that COVID-19 is real and it is normal for people that feel they may have the disease to seek medical care. This approach helps suppress the transmission of the virus. This pilot project could be reproduced in several other communities. It is easy to operate, rapid, effective, and cost-free. The community has been very receptive to and grateful for our efforts.
Like other countries across the world, Haiti was not ready for COVID-19. But we are confident that, with help from the international community, organizations such as GHESKIO,14 and with due diligence on the local level, we are strong and resilient enough to beat COVID. We must act together – quickly.
References
1. Sénat JD. Coronavirus: 2 cas confirmés en Haïti, Jovenel Moïse décrète l’état d’ur-gence sanitaire. 2020 Le Nouvelliste.
2. Haitian Ministry of Health.
3. “Entre appel a la solidarite et de sombres previsions, le Dr William Pape fait le point.” Le Nouvelliste.
4. Darzi A and Evans T. Lancet. 2016 Nov-Dec 26. 388;10060:2576-7.
5. Rapport Statistique 2018. 2019 Republic of Haiti.
6. Sentlinger K. “Water Crisis in Haiti.” The Water Project.
7. The World Bank in Haiti. worldbank.org.
8. Cenat JM. Travel Med Infect Dis. 2020 Mar 28. doi: 10.1016/jtmaid.2020.101684.
9. Block D. “Why some Americans resist wearing face masks.” voanews.com. 2020 May 31.
10. Panceski B and Douglas J. “Masks could help stop coronavirus. So why are they still controversial?” wsj.com. Updated 29 Jun 2020.
11. Bojarski S. “Social distancing: A luxury Haiti’s poor cannot afford. The Haitian Times. 2020 Apr.
12. World Health Organization, UNICEF, World Bank Group, and the U.N. Population Division. Maternal mortality ratio, Haiti.
13. Feliciano I and Kargbo C. “As COVID cases surge, Haiti’s Dr. Pape is on the front line again.” PBS NewsHour Weekend. 2020 Jun 13.
14. Liautaud B and Deschamps MM. New Engl J Med. 2020 Jun 16.
Mr. Dorcela is a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince, Haiti. He also is a medical intern at Unité de Médecine Familiale Hôpital Saint Nicolas in Saint-Marc. Mr. Dorcela has no disclosures. Mr. St. Jean, who is Mr. Dorcela’s brother, is also a senior medical student at Faculté des Sciences de la Santé Université Quisqueya in Port-au-Prince. He has no disclosures.
FLU/SAL inhalers for COPD carry greater pneumonia risk
For well over a decade the elevated risk of pneumonia from inhaled corticosteroids for moderate to very severe COPD has been well documented, although the pneumonia risks from different types of ICSs have not been well understood.
Researchers from Taiwan have taken a step in to investigate this question with a nationwide cohort study that reported inhalers with budesonide and beclomethasone may have a lower pneumonia risk than that of fluticasone propionate/salmeterol inhalers (CHEST. 2020;157:117-29).
The study is the first to include beclomethasone-containing inhalers in a comparison of ICS/long-acting beta2-agonist (LABA) fixed combinations to evaluate pneumonia risk, along with dose and drug properties, wrote Ting-Yu Chang, MS, of the Graduate Institute of Clinical Pharmacology at the College of Medicine, National Taiwan University in Taipei, and colleagues.
The study evaluated 42,393 people with COPD in the National Health Insurance Research Database who got at least two continuous prescriptions for three different types of inhalers:
- Budesonide/formoterol (BUD/FOR).
- Beclomethasone/formoterol (BEC/FOR).
- Fluticasone propionate/salmeterol (FLU/SAL).
The study included patients aged 40 years and older who used a metered-dose inhaler (MDI) or dry-powder inhaler (DPI) between January 2011 and June 2015.
Patient experience with adverse events (AEs) was a factor in risk stratification, Mr. Chang and colleagues noted. “For the comparison between the BEC/FOR MDI and FLU/SAL MDI, the lower risk associated with the BEC/FOR MDI was more prominent in patients without severe AE in the past year,” they wrote.
The study found that BUD/FOR DPI users had a 17% lower risk of severe pneumonia and a 12% lower risk of severe AEs than that of FLU/SAL DPI users. The risk difference in pneumonia remained significant after adjustment for the ICS-equivalent daily dose, but the spread for AEs didn’t.
BEC/FOR MDI users were 31% less likely to get severe pneumonia and 18% less likely to have severe AEs than were FLU/SAL MDI users, but that difference declined and became nonsignificant after adjustment for the ICS-equivalent daily dose.
The study also found that a high average daily dose (> 500 mcg/d) of FLU/SAL MDI carried a 66% greater risk of severe pneumonia, compared with that of low-dose users. Also, medium-dose BEC/FOR MDI users (FLU equivalent 299-499 mcg/d) had a 38% greater risk of severe pneumonia than low-dose (< 200 mcg/d) users.
The variable pneumonia risks may be linked to each ICS’s pharmacokinetics, specifically their distinct lipophilic properties, Mr. Chang and colleagues wrote. Fluticasone propionate is known to be more lipophilic than budesonide, and while beclomethasone is more lipophilic than both, as a prodrug it rapidly converts to lower lipophilicity upon contact with bronchial secretions. “In general, a lipophilic ICS has a longer retention time within the airway or lung tissue to exert local immunosuppression and reduce inflammation,” Mr. Chang and colleagues stated.
The Taiwan Ministry of Science and Technology provided partial support for the study. Mr. Chang and colleagues have no relationships to disclose.
SOURCE: Chang TY et al. CHEST. 2020;157:117-29.
For well over a decade the elevated risk of pneumonia from inhaled corticosteroids for moderate to very severe COPD has been well documented, although the pneumonia risks from different types of ICSs have not been well understood.
Researchers from Taiwan have taken a step in to investigate this question with a nationwide cohort study that reported inhalers with budesonide and beclomethasone may have a lower pneumonia risk than that of fluticasone propionate/salmeterol inhalers (CHEST. 2020;157:117-29).
The study is the first to include beclomethasone-containing inhalers in a comparison of ICS/long-acting beta2-agonist (LABA) fixed combinations to evaluate pneumonia risk, along with dose and drug properties, wrote Ting-Yu Chang, MS, of the Graduate Institute of Clinical Pharmacology at the College of Medicine, National Taiwan University in Taipei, and colleagues.
The study evaluated 42,393 people with COPD in the National Health Insurance Research Database who got at least two continuous prescriptions for three different types of inhalers:
- Budesonide/formoterol (BUD/FOR).
- Beclomethasone/formoterol (BEC/FOR).
- Fluticasone propionate/salmeterol (FLU/SAL).
The study included patients aged 40 years and older who used a metered-dose inhaler (MDI) or dry-powder inhaler (DPI) between January 2011 and June 2015.
Patient experience with adverse events (AEs) was a factor in risk stratification, Mr. Chang and colleagues noted. “For the comparison between the BEC/FOR MDI and FLU/SAL MDI, the lower risk associated with the BEC/FOR MDI was more prominent in patients without severe AE in the past year,” they wrote.
The study found that BUD/FOR DPI users had a 17% lower risk of severe pneumonia and a 12% lower risk of severe AEs than that of FLU/SAL DPI users. The risk difference in pneumonia remained significant after adjustment for the ICS-equivalent daily dose, but the spread for AEs didn’t.
BEC/FOR MDI users were 31% less likely to get severe pneumonia and 18% less likely to have severe AEs than were FLU/SAL MDI users, but that difference declined and became nonsignificant after adjustment for the ICS-equivalent daily dose.
The study also found that a high average daily dose (> 500 mcg/d) of FLU/SAL MDI carried a 66% greater risk of severe pneumonia, compared with that of low-dose users. Also, medium-dose BEC/FOR MDI users (FLU equivalent 299-499 mcg/d) had a 38% greater risk of severe pneumonia than low-dose (< 200 mcg/d) users.
The variable pneumonia risks may be linked to each ICS’s pharmacokinetics, specifically their distinct lipophilic properties, Mr. Chang and colleagues wrote. Fluticasone propionate is known to be more lipophilic than budesonide, and while beclomethasone is more lipophilic than both, as a prodrug it rapidly converts to lower lipophilicity upon contact with bronchial secretions. “In general, a lipophilic ICS has a longer retention time within the airway or lung tissue to exert local immunosuppression and reduce inflammation,” Mr. Chang and colleagues stated.
The Taiwan Ministry of Science and Technology provided partial support for the study. Mr. Chang and colleagues have no relationships to disclose.
SOURCE: Chang TY et al. CHEST. 2020;157:117-29.
For well over a decade the elevated risk of pneumonia from inhaled corticosteroids for moderate to very severe COPD has been well documented, although the pneumonia risks from different types of ICSs have not been well understood.
Researchers from Taiwan have taken a step in to investigate this question with a nationwide cohort study that reported inhalers with budesonide and beclomethasone may have a lower pneumonia risk than that of fluticasone propionate/salmeterol inhalers (CHEST. 2020;157:117-29).
The study is the first to include beclomethasone-containing inhalers in a comparison of ICS/long-acting beta2-agonist (LABA) fixed combinations to evaluate pneumonia risk, along with dose and drug properties, wrote Ting-Yu Chang, MS, of the Graduate Institute of Clinical Pharmacology at the College of Medicine, National Taiwan University in Taipei, and colleagues.
The study evaluated 42,393 people with COPD in the National Health Insurance Research Database who got at least two continuous prescriptions for three different types of inhalers:
- Budesonide/formoterol (BUD/FOR).
- Beclomethasone/formoterol (BEC/FOR).
- Fluticasone propionate/salmeterol (FLU/SAL).
The study included patients aged 40 years and older who used a metered-dose inhaler (MDI) or dry-powder inhaler (DPI) between January 2011 and June 2015.
Patient experience with adverse events (AEs) was a factor in risk stratification, Mr. Chang and colleagues noted. “For the comparison between the BEC/FOR MDI and FLU/SAL MDI, the lower risk associated with the BEC/FOR MDI was more prominent in patients without severe AE in the past year,” they wrote.
The study found that BUD/FOR DPI users had a 17% lower risk of severe pneumonia and a 12% lower risk of severe AEs than that of FLU/SAL DPI users. The risk difference in pneumonia remained significant after adjustment for the ICS-equivalent daily dose, but the spread for AEs didn’t.
BEC/FOR MDI users were 31% less likely to get severe pneumonia and 18% less likely to have severe AEs than were FLU/SAL MDI users, but that difference declined and became nonsignificant after adjustment for the ICS-equivalent daily dose.
The study also found that a high average daily dose (> 500 mcg/d) of FLU/SAL MDI carried a 66% greater risk of severe pneumonia, compared with that of low-dose users. Also, medium-dose BEC/FOR MDI users (FLU equivalent 299-499 mcg/d) had a 38% greater risk of severe pneumonia than low-dose (< 200 mcg/d) users.
The variable pneumonia risks may be linked to each ICS’s pharmacokinetics, specifically their distinct lipophilic properties, Mr. Chang and colleagues wrote. Fluticasone propionate is known to be more lipophilic than budesonide, and while beclomethasone is more lipophilic than both, as a prodrug it rapidly converts to lower lipophilicity upon contact with bronchial secretions. “In general, a lipophilic ICS has a longer retention time within the airway or lung tissue to exert local immunosuppression and reduce inflammation,” Mr. Chang and colleagues stated.
The Taiwan Ministry of Science and Technology provided partial support for the study. Mr. Chang and colleagues have no relationships to disclose.
SOURCE: Chang TY et al. CHEST. 2020;157:117-29.
FROM CHEST
Americans avoided emergency departments early in the pandemic
compared with the corresponding period in 2019, according to a report from the Centers for Disease Control and Prevention.
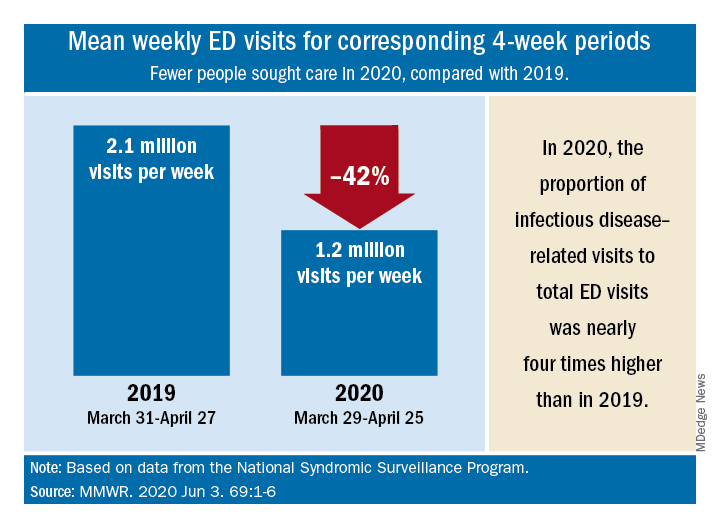
“The striking decline in ED visits nationwide … suggests that the pandemic has altered the use of the ED by the public,” Kathleen P. Hartnett, PhD, and associates at the CDC said June 3 in the Mortality and Morbidity Weekly Report.
The weekly mean was just over 1.2 million ED visits for the 4 weeks from March 29 to April 25, 2020, compared with the nearly 2.2 million visits per week recorded from March 31 to April 27, 2019 – a drop of 42%, based on an analysis of data from the National Syndromic Surveillance Program.
Despite that drop, ED visits for infectious disease–related reasons, taken as a proportion of all 1.2 ED visits during the early pandemic period, were 3.8 times higher than the comparison period in 2019, the investigators reported.
ED visits also were higher in 2020 for specified and unspecified lower respiratory disease not including influenza, pneumonia, asthma, or bronchitis (prevalence ratio of 1.99, compared with 2019), cardiac arrest and ventricular fibrillation (PR, 1.98), and pneumonia not caused by tuberculosis (PR, 1.91), Dr. Hartnett and associates said.
Prevalence ratios for the early pandemic period were down for most other conditions, with some of the largest decreases seen for influenza (PR, 0.16), otitis media (PR, 0.35), and neoplasm-related encounters (PR, 0.40), they said.
Visits have increased each week since reaching their lowest point during April 12-18, but the number for the most recent full week, May 24-30, which was not included in the analysis, was still 26% lower than the corresponding week in 2019, the CDC team pointed out.
“Some persons could be delaying care for conditions that might result in additional mortality if left untreated,” the investigators noted, and those “who use the ED as a safety net because they lack access to primary care and telemedicine might be disproportionately affected if they avoid seeking care because of concerns about the infection risk in the ED.”
SOURCE: Hartnett KP et al. MMWR. 2020 Jun 3. 69:1-6.
compared with the corresponding period in 2019, according to a report from the Centers for Disease Control and Prevention.

“The striking decline in ED visits nationwide … suggests that the pandemic has altered the use of the ED by the public,” Kathleen P. Hartnett, PhD, and associates at the CDC said June 3 in the Mortality and Morbidity Weekly Report.
The weekly mean was just over 1.2 million ED visits for the 4 weeks from March 29 to April 25, 2020, compared with the nearly 2.2 million visits per week recorded from March 31 to April 27, 2019 – a drop of 42%, based on an analysis of data from the National Syndromic Surveillance Program.
Despite that drop, ED visits for infectious disease–related reasons, taken as a proportion of all 1.2 ED visits during the early pandemic period, were 3.8 times higher than the comparison period in 2019, the investigators reported.
ED visits also were higher in 2020 for specified and unspecified lower respiratory disease not including influenza, pneumonia, asthma, or bronchitis (prevalence ratio of 1.99, compared with 2019), cardiac arrest and ventricular fibrillation (PR, 1.98), and pneumonia not caused by tuberculosis (PR, 1.91), Dr. Hartnett and associates said.
Prevalence ratios for the early pandemic period were down for most other conditions, with some of the largest decreases seen for influenza (PR, 0.16), otitis media (PR, 0.35), and neoplasm-related encounters (PR, 0.40), they said.
Visits have increased each week since reaching their lowest point during April 12-18, but the number for the most recent full week, May 24-30, which was not included in the analysis, was still 26% lower than the corresponding week in 2019, the CDC team pointed out.
“Some persons could be delaying care for conditions that might result in additional mortality if left untreated,” the investigators noted, and those “who use the ED as a safety net because they lack access to primary care and telemedicine might be disproportionately affected if they avoid seeking care because of concerns about the infection risk in the ED.”
SOURCE: Hartnett KP et al. MMWR. 2020 Jun 3. 69:1-6.
compared with the corresponding period in 2019, according to a report from the Centers for Disease Control and Prevention.

“The striking decline in ED visits nationwide … suggests that the pandemic has altered the use of the ED by the public,” Kathleen P. Hartnett, PhD, and associates at the CDC said June 3 in the Mortality and Morbidity Weekly Report.
The weekly mean was just over 1.2 million ED visits for the 4 weeks from March 29 to April 25, 2020, compared with the nearly 2.2 million visits per week recorded from March 31 to April 27, 2019 – a drop of 42%, based on an analysis of data from the National Syndromic Surveillance Program.
Despite that drop, ED visits for infectious disease–related reasons, taken as a proportion of all 1.2 ED visits during the early pandemic period, were 3.8 times higher than the comparison period in 2019, the investigators reported.
ED visits also were higher in 2020 for specified and unspecified lower respiratory disease not including influenza, pneumonia, asthma, or bronchitis (prevalence ratio of 1.99, compared with 2019), cardiac arrest and ventricular fibrillation (PR, 1.98), and pneumonia not caused by tuberculosis (PR, 1.91), Dr. Hartnett and associates said.
Prevalence ratios for the early pandemic period were down for most other conditions, with some of the largest decreases seen for influenza (PR, 0.16), otitis media (PR, 0.35), and neoplasm-related encounters (PR, 0.40), they said.
Visits have increased each week since reaching their lowest point during April 12-18, but the number for the most recent full week, May 24-30, which was not included in the analysis, was still 26% lower than the corresponding week in 2019, the CDC team pointed out.
“Some persons could be delaying care for conditions that might result in additional mortality if left untreated,” the investigators noted, and those “who use the ED as a safety net because they lack access to primary care and telemedicine might be disproportionately affected if they avoid seeking care because of concerns about the infection risk in the ED.”
SOURCE: Hartnett KP et al. MMWR. 2020 Jun 3. 69:1-6.
FROM MMWR
Antibody testing suggests COVID-19 cases are being missed
The number of COVID-19 infections in the community may be “substantially greater” than totals confirmed by authorities, based on SARS-CoV-2 antibody testing among a random sample of adults in Los Angeles County, Calif.
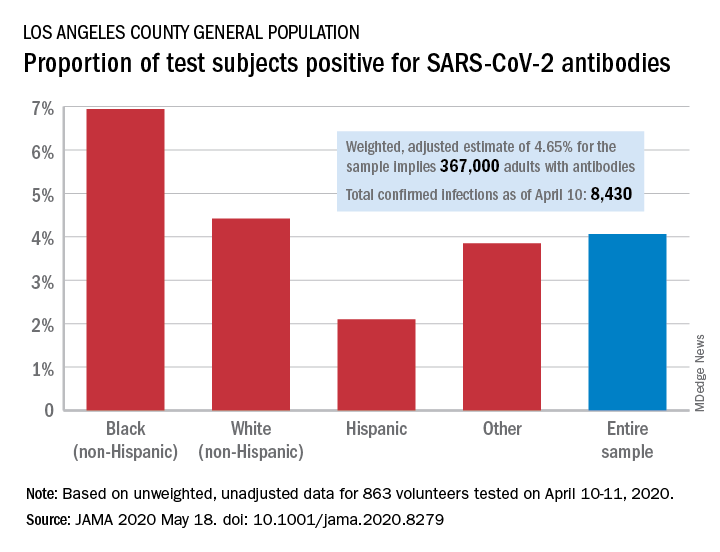
Testing of 863 people on April 10-11 revealed that 35 (4.06%) were positive for SARS-CoV-2–specific antibodies (IgM or IgG), and after adjustment for test sensitivity and specificity, the weighted prevalence for the entire sample was 4.65%, Neeraj Sood, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
The estimate of 4.65% “implies that approximately 367,000 adults [in Los Angeles County] had SARS-CoV-2 antibodies, which is substantially greater than the 8,430 cumulative number of confirmed infections in the county on April 10,” they wrote.
It also suggests that fatality rates based on the larger number of infections may be lower than rates based on confirmed cases. “In addition, contact tracing methods to limit the spread of infection will face considerable challenges,” Dr. Sood and associates said.
Test positivity varied by race/ethnicity, sex, and income. The proportion of non-Hispanic blacks with a positive result was 6.94%, compared with 4.42% for non-Hispanic whites, 2.10% for Hispanics, and 3.85% for others. Men were much more likely than women to be positive for SARS-CoV-2: 5.18% vs. 3.31%, the investigators said.
Household income favored the middle ground. Those individuals making less than $50,000 a year had a positivity rate of 5.14% and those with an income of $100,000 or more had a rate of 4.90%, but only 1.58% of those making $50,000-$99,999 tested positive, they reported.
The authors reported numerous sources of nonprofit organization support.
SOURCE: Sood N et al. JAMA 2020 May 18. doi: 10.1001/jama.2020.8279.
The number of COVID-19 infections in the community may be “substantially greater” than totals confirmed by authorities, based on SARS-CoV-2 antibody testing among a random sample of adults in Los Angeles County, Calif.

Testing of 863 people on April 10-11 revealed that 35 (4.06%) were positive for SARS-CoV-2–specific antibodies (IgM or IgG), and after adjustment for test sensitivity and specificity, the weighted prevalence for the entire sample was 4.65%, Neeraj Sood, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
The estimate of 4.65% “implies that approximately 367,000 adults [in Los Angeles County] had SARS-CoV-2 antibodies, which is substantially greater than the 8,430 cumulative number of confirmed infections in the county on April 10,” they wrote.
It also suggests that fatality rates based on the larger number of infections may be lower than rates based on confirmed cases. “In addition, contact tracing methods to limit the spread of infection will face considerable challenges,” Dr. Sood and associates said.
Test positivity varied by race/ethnicity, sex, and income. The proportion of non-Hispanic blacks with a positive result was 6.94%, compared with 4.42% for non-Hispanic whites, 2.10% for Hispanics, and 3.85% for others. Men were much more likely than women to be positive for SARS-CoV-2: 5.18% vs. 3.31%, the investigators said.
Household income favored the middle ground. Those individuals making less than $50,000 a year had a positivity rate of 5.14% and those with an income of $100,000 or more had a rate of 4.90%, but only 1.58% of those making $50,000-$99,999 tested positive, they reported.
The authors reported numerous sources of nonprofit organization support.
SOURCE: Sood N et al. JAMA 2020 May 18. doi: 10.1001/jama.2020.8279.
The number of COVID-19 infections in the community may be “substantially greater” than totals confirmed by authorities, based on SARS-CoV-2 antibody testing among a random sample of adults in Los Angeles County, Calif.

Testing of 863 people on April 10-11 revealed that 35 (4.06%) were positive for SARS-CoV-2–specific antibodies (IgM or IgG), and after adjustment for test sensitivity and specificity, the weighted prevalence for the entire sample was 4.65%, Neeraj Sood, PhD, of the University of Southern California, Los Angeles, and associates wrote in JAMA.
The estimate of 4.65% “implies that approximately 367,000 adults [in Los Angeles County] had SARS-CoV-2 antibodies, which is substantially greater than the 8,430 cumulative number of confirmed infections in the county on April 10,” they wrote.
It also suggests that fatality rates based on the larger number of infections may be lower than rates based on confirmed cases. “In addition, contact tracing methods to limit the spread of infection will face considerable challenges,” Dr. Sood and associates said.
Test positivity varied by race/ethnicity, sex, and income. The proportion of non-Hispanic blacks with a positive result was 6.94%, compared with 4.42% for non-Hispanic whites, 2.10% for Hispanics, and 3.85% for others. Men were much more likely than women to be positive for SARS-CoV-2: 5.18% vs. 3.31%, the investigators said.
Household income favored the middle ground. Those individuals making less than $50,000 a year had a positivity rate of 5.14% and those with an income of $100,000 or more had a rate of 4.90%, but only 1.58% of those making $50,000-$99,999 tested positive, they reported.
The authors reported numerous sources of nonprofit organization support.
SOURCE: Sood N et al. JAMA 2020 May 18. doi: 10.1001/jama.2020.8279.
FROM JAMA
A surge in PTSD may be the ‘new normal’
The prolonged and unique stresses imparted by the COVID-19 pandemic has many predicting a significant rise in mental health issues in the weeks, months, and years ahead.
To understand how health care workers can best get ahead of this emerging crisis within a crisis, Medscape Psychiatry editorial director Bret Stetka, MD, spoke with Sheila Rauch, PhD, who’s with the Department of Psychiatry and Behavioral Sciences at the Emory University, Atlanta. The director of Mental Health Research and Program Evaluation at the Atlanta VA Medical Center, Dr. Rauch has studied the effects of and best treatments for posttraumatic stress disorder (PTSD) and anxiety disorders over the past 20 years.
Are we going to see a PTSD or anxiety epidemic as a result of the pandemic?
First, I think it’s really important that we prepare for the worst but hope for the best. But I would expect that, given the high levels of stress, the impact on resources, and other factors, we are going to see a pretty significant mental health impact over time. This could be the new normal for a while. Some of that will be PTSD, but there will also be other things. I would suspect that the resulting increase in rates of depression, traumatic grief, and loss is probably going to be a significant issue for years to come.
What will the anxiety we see as a result of COVID-19 look like compared with that seen in past disasters, like 9/11?
Most disasters in recent history, like 9/11, are single incidents. Something horrible happened, it impacted people at different levels, and we were able to start putting the pieces back together right away. The prolonged nature of this pandemic makes it even more variable given that the impact is going to be extended over time.
We’re also going to see a lot more people with compound impact – people who’ve lost their jobs, loved ones, maybe even their homes. All of those financial and resource losses put people in a higher risk category for negative mental health outcomes.
Is this analogous to the prolonged trauma that can occur with military service during war?
There is some similarity there. Combat is kind of an overarching context in which people experience trauma and, much like this pandemic, may or may not have traumatic exposures during it.
We’re asking health care workers to actually be in a role similar to what we ask of our military: going into danger, sometimes even without proper protective equipment, in order to save the lives of others. That’s also something we need to be factoring in as we plan to support those people and their families.
This is an ongoing incident, but is there a time window we need to be particularly worried about for seeing spikes in anxiety and PTSD?
I think we’re going to see variability on that. PTSD is a disorder that’s related to a specific incident or a couple of incidents that are similar. It’s a memory that’s haunting you.
For instance, typically if you have a combat veteran who has PTSD, they’ve been exposed to the overarching context of combat but then they have specific memories that are stuck. If they don’t have PTSD about 3-6 months after those incidents happen, then we would expect that they will not develop it, or it’s much less common that they would.
Depression has a very different course. It’s more prolonged and tends to grow with time.
Are you already seeing increased symptoms in your patients?
This is pretty similar to what we see in combat veterans. They’ll often be unhappy with the leadership decisions that were made as they were being deployed.
We’re also seeing lots more anger, sadness, and isolation now. Especially over the past couple of weeks, we’ve seen a rise in things like people reaching out for help in our intakes because we’re still open and doing phone assessments and telehealth with veterans and the veterans program.
In terms of interventions for this, what should psychiatrists, psychologists, and other clinicians be thinking about?
Right now, the best thing that we can do as mental health providers for people affected by the trauma is provide crisis intervention for those saying they are a danger to themselves and others. That means providing coping strategies and support. It also means making sure people are taking breaks and taking care of themselves, taking that little bit of time off so that they can go back, fully recharged, to their jobs and really stay there.
As we move forward, it will be clearer whether people are going to naturally recover, which most people will. For those who are going to have ongoing problems with time, we need to be getting ready as a system and as a country for those long-term mental health issues that are going to be coming up. And when I say long-term, it means the next 1-3 months. We want to be providing preventive interventions, versions of prolonged exposure, and other things that have shown some help in preventing PTSD. Psychological first aid is helpful.
There’s also an app called COVID Coach that the National Center for PTSD has created. That features a lot of positive coping resources together in one source.
Then when we get to the middle of that point and beyond it, we need to be ready to provide those evidence-based interventions for PTSD, depression, panic disorder, and other issues that are going to come out of this current situation.
But we were already short-staffed as far as mental health resources in general across the country, and especially in rural areas. So that means finding ways to efficiently use what we have through potentially briefer versions of interventions, through primary care, mental health, and other staff.
In what ways can primary care providers help?
There are versions of prolonged exposure therapy for primary care. That’s one of my big areas of research – increasing access. That would be something that we need to be building, by training and embedding mental health providers in primary care settings so that they can help to accommodate the increased need for access that’s going to be showing up for the next, I would suspect, several years with the pandemic.
Is there evidence that a prior episode of PTSD or traumatic experience like combat influences a subsequent reaction to a trauma like this?
It depends on how they manage. Research suggests that veterans or other people who have experienced trauma and naturally recovered, or who have gotten good treatment and remitted from that issue, are probably at no higher risk. But people who have subsyndromal PTSD or depression, or who are still experiencing symptoms from a history of trauma exposure, are maybe at a higher risk of having problems over time.
Do you have any guidance for healthcare providers on how to approach the pandemic with their patients, and also on how they can look after their own mental health?
In talking to patients, make sure that they have what they need. Ask if they’ve thought through how they’re going to cope if things get harder for them.
For people who have preexisting mental health issues, I’m talking with them about whether things have gotten worse. If they’re at high risk for suicide, I’m checking in to make sure that they’ve got new plans and ways to connect with people to reduce isolation, keeping in mind the social distancing that we’re asked to engage in so that they can do that safely.
It’s important to check and see if they have had any losses, whether it’s a financial loss or a personal loss of people that they care about. Also have them think through ways to stay entertained, which tends to help manage their own anxiety.
Every coping strategy we outline for patients also applies to mental health professionals. However, you would add to it the real need to take time to recharge, to take breaks, time off. It can feel overwhelming and like you need to just keep going. But the more that you get stuck in that mode of overdoing it, the less effective you’re going to be in helping people and also the more likely that you’ll be at risk of perhaps being one of the people that needs help.
It’s also important to make sure you’re staying connected with family and friends virtually, in whatever ways you can safely do that with social distancing.
So take a break to watch some Netflix now and then?
Yes!
A version of this article originally appeared on Medscape.com.
The prolonged and unique stresses imparted by the COVID-19 pandemic has many predicting a significant rise in mental health issues in the weeks, months, and years ahead.
To understand how health care workers can best get ahead of this emerging crisis within a crisis, Medscape Psychiatry editorial director Bret Stetka, MD, spoke with Sheila Rauch, PhD, who’s with the Department of Psychiatry and Behavioral Sciences at the Emory University, Atlanta. The director of Mental Health Research and Program Evaluation at the Atlanta VA Medical Center, Dr. Rauch has studied the effects of and best treatments for posttraumatic stress disorder (PTSD) and anxiety disorders over the past 20 years.
Are we going to see a PTSD or anxiety epidemic as a result of the pandemic?
First, I think it’s really important that we prepare for the worst but hope for the best. But I would expect that, given the high levels of stress, the impact on resources, and other factors, we are going to see a pretty significant mental health impact over time. This could be the new normal for a while. Some of that will be PTSD, but there will also be other things. I would suspect that the resulting increase in rates of depression, traumatic grief, and loss is probably going to be a significant issue for years to come.
What will the anxiety we see as a result of COVID-19 look like compared with that seen in past disasters, like 9/11?
Most disasters in recent history, like 9/11, are single incidents. Something horrible happened, it impacted people at different levels, and we were able to start putting the pieces back together right away. The prolonged nature of this pandemic makes it even more variable given that the impact is going to be extended over time.
We’re also going to see a lot more people with compound impact – people who’ve lost their jobs, loved ones, maybe even their homes. All of those financial and resource losses put people in a higher risk category for negative mental health outcomes.
Is this analogous to the prolonged trauma that can occur with military service during war?
There is some similarity there. Combat is kind of an overarching context in which people experience trauma and, much like this pandemic, may or may not have traumatic exposures during it.
We’re asking health care workers to actually be in a role similar to what we ask of our military: going into danger, sometimes even without proper protective equipment, in order to save the lives of others. That’s also something we need to be factoring in as we plan to support those people and their families.
This is an ongoing incident, but is there a time window we need to be particularly worried about for seeing spikes in anxiety and PTSD?
I think we’re going to see variability on that. PTSD is a disorder that’s related to a specific incident or a couple of incidents that are similar. It’s a memory that’s haunting you.
For instance, typically if you have a combat veteran who has PTSD, they’ve been exposed to the overarching context of combat but then they have specific memories that are stuck. If they don’t have PTSD about 3-6 months after those incidents happen, then we would expect that they will not develop it, or it’s much less common that they would.
Depression has a very different course. It’s more prolonged and tends to grow with time.
Are you already seeing increased symptoms in your patients?
This is pretty similar to what we see in combat veterans. They’ll often be unhappy with the leadership decisions that were made as they were being deployed.
We’re also seeing lots more anger, sadness, and isolation now. Especially over the past couple of weeks, we’ve seen a rise in things like people reaching out for help in our intakes because we’re still open and doing phone assessments and telehealth with veterans and the veterans program.
In terms of interventions for this, what should psychiatrists, psychologists, and other clinicians be thinking about?
Right now, the best thing that we can do as mental health providers for people affected by the trauma is provide crisis intervention for those saying they are a danger to themselves and others. That means providing coping strategies and support. It also means making sure people are taking breaks and taking care of themselves, taking that little bit of time off so that they can go back, fully recharged, to their jobs and really stay there.
As we move forward, it will be clearer whether people are going to naturally recover, which most people will. For those who are going to have ongoing problems with time, we need to be getting ready as a system and as a country for those long-term mental health issues that are going to be coming up. And when I say long-term, it means the next 1-3 months. We want to be providing preventive interventions, versions of prolonged exposure, and other things that have shown some help in preventing PTSD. Psychological first aid is helpful.
There’s also an app called COVID Coach that the National Center for PTSD has created. That features a lot of positive coping resources together in one source.
Then when we get to the middle of that point and beyond it, we need to be ready to provide those evidence-based interventions for PTSD, depression, panic disorder, and other issues that are going to come out of this current situation.
But we were already short-staffed as far as mental health resources in general across the country, and especially in rural areas. So that means finding ways to efficiently use what we have through potentially briefer versions of interventions, through primary care, mental health, and other staff.
In what ways can primary care providers help?
There are versions of prolonged exposure therapy for primary care. That’s one of my big areas of research – increasing access. That would be something that we need to be building, by training and embedding mental health providers in primary care settings so that they can help to accommodate the increased need for access that’s going to be showing up for the next, I would suspect, several years with the pandemic.
Is there evidence that a prior episode of PTSD or traumatic experience like combat influences a subsequent reaction to a trauma like this?
It depends on how they manage. Research suggests that veterans or other people who have experienced trauma and naturally recovered, or who have gotten good treatment and remitted from that issue, are probably at no higher risk. But people who have subsyndromal PTSD or depression, or who are still experiencing symptoms from a history of trauma exposure, are maybe at a higher risk of having problems over time.
Do you have any guidance for healthcare providers on how to approach the pandemic with their patients, and also on how they can look after their own mental health?
In talking to patients, make sure that they have what they need. Ask if they’ve thought through how they’re going to cope if things get harder for them.
For people who have preexisting mental health issues, I’m talking with them about whether things have gotten worse. If they’re at high risk for suicide, I’m checking in to make sure that they’ve got new plans and ways to connect with people to reduce isolation, keeping in mind the social distancing that we’re asked to engage in so that they can do that safely.
It’s important to check and see if they have had any losses, whether it’s a financial loss or a personal loss of people that they care about. Also have them think through ways to stay entertained, which tends to help manage their own anxiety.
Every coping strategy we outline for patients also applies to mental health professionals. However, you would add to it the real need to take time to recharge, to take breaks, time off. It can feel overwhelming and like you need to just keep going. But the more that you get stuck in that mode of overdoing it, the less effective you’re going to be in helping people and also the more likely that you’ll be at risk of perhaps being one of the people that needs help.
It’s also important to make sure you’re staying connected with family and friends virtually, in whatever ways you can safely do that with social distancing.
So take a break to watch some Netflix now and then?
Yes!
A version of this article originally appeared on Medscape.com.
The prolonged and unique stresses imparted by the COVID-19 pandemic has many predicting a significant rise in mental health issues in the weeks, months, and years ahead.
To understand how health care workers can best get ahead of this emerging crisis within a crisis, Medscape Psychiatry editorial director Bret Stetka, MD, spoke with Sheila Rauch, PhD, who’s with the Department of Psychiatry and Behavioral Sciences at the Emory University, Atlanta. The director of Mental Health Research and Program Evaluation at the Atlanta VA Medical Center, Dr. Rauch has studied the effects of and best treatments for posttraumatic stress disorder (PTSD) and anxiety disorders over the past 20 years.
Are we going to see a PTSD or anxiety epidemic as a result of the pandemic?
First, I think it’s really important that we prepare for the worst but hope for the best. But I would expect that, given the high levels of stress, the impact on resources, and other factors, we are going to see a pretty significant mental health impact over time. This could be the new normal for a while. Some of that will be PTSD, but there will also be other things. I would suspect that the resulting increase in rates of depression, traumatic grief, and loss is probably going to be a significant issue for years to come.
What will the anxiety we see as a result of COVID-19 look like compared with that seen in past disasters, like 9/11?
Most disasters in recent history, like 9/11, are single incidents. Something horrible happened, it impacted people at different levels, and we were able to start putting the pieces back together right away. The prolonged nature of this pandemic makes it even more variable given that the impact is going to be extended over time.
We’re also going to see a lot more people with compound impact – people who’ve lost their jobs, loved ones, maybe even their homes. All of those financial and resource losses put people in a higher risk category for negative mental health outcomes.
Is this analogous to the prolonged trauma that can occur with military service during war?
There is some similarity there. Combat is kind of an overarching context in which people experience trauma and, much like this pandemic, may or may not have traumatic exposures during it.
We’re asking health care workers to actually be in a role similar to what we ask of our military: going into danger, sometimes even without proper protective equipment, in order to save the lives of others. That’s also something we need to be factoring in as we plan to support those people and their families.
This is an ongoing incident, but is there a time window we need to be particularly worried about for seeing spikes in anxiety and PTSD?
I think we’re going to see variability on that. PTSD is a disorder that’s related to a specific incident or a couple of incidents that are similar. It’s a memory that’s haunting you.
For instance, typically if you have a combat veteran who has PTSD, they’ve been exposed to the overarching context of combat but then they have specific memories that are stuck. If they don’t have PTSD about 3-6 months after those incidents happen, then we would expect that they will not develop it, or it’s much less common that they would.
Depression has a very different course. It’s more prolonged and tends to grow with time.
Are you already seeing increased symptoms in your patients?
This is pretty similar to what we see in combat veterans. They’ll often be unhappy with the leadership decisions that were made as they were being deployed.
We’re also seeing lots more anger, sadness, and isolation now. Especially over the past couple of weeks, we’ve seen a rise in things like people reaching out for help in our intakes because we’re still open and doing phone assessments and telehealth with veterans and the veterans program.
In terms of interventions for this, what should psychiatrists, psychologists, and other clinicians be thinking about?
Right now, the best thing that we can do as mental health providers for people affected by the trauma is provide crisis intervention for those saying they are a danger to themselves and others. That means providing coping strategies and support. It also means making sure people are taking breaks and taking care of themselves, taking that little bit of time off so that they can go back, fully recharged, to their jobs and really stay there.
As we move forward, it will be clearer whether people are going to naturally recover, which most people will. For those who are going to have ongoing problems with time, we need to be getting ready as a system and as a country for those long-term mental health issues that are going to be coming up. And when I say long-term, it means the next 1-3 months. We want to be providing preventive interventions, versions of prolonged exposure, and other things that have shown some help in preventing PTSD. Psychological first aid is helpful.
There’s also an app called COVID Coach that the National Center for PTSD has created. That features a lot of positive coping resources together in one source.
Then when we get to the middle of that point and beyond it, we need to be ready to provide those evidence-based interventions for PTSD, depression, panic disorder, and other issues that are going to come out of this current situation.
But we were already short-staffed as far as mental health resources in general across the country, and especially in rural areas. So that means finding ways to efficiently use what we have through potentially briefer versions of interventions, through primary care, mental health, and other staff.
In what ways can primary care providers help?
There are versions of prolonged exposure therapy for primary care. That’s one of my big areas of research – increasing access. That would be something that we need to be building, by training and embedding mental health providers in primary care settings so that they can help to accommodate the increased need for access that’s going to be showing up for the next, I would suspect, several years with the pandemic.
Is there evidence that a prior episode of PTSD or traumatic experience like combat influences a subsequent reaction to a trauma like this?
It depends on how they manage. Research suggests that veterans or other people who have experienced trauma and naturally recovered, or who have gotten good treatment and remitted from that issue, are probably at no higher risk. But people who have subsyndromal PTSD or depression, or who are still experiencing symptoms from a history of trauma exposure, are maybe at a higher risk of having problems over time.
Do you have any guidance for healthcare providers on how to approach the pandemic with their patients, and also on how they can look after their own mental health?
In talking to patients, make sure that they have what they need. Ask if they’ve thought through how they’re going to cope if things get harder for them.
For people who have preexisting mental health issues, I’m talking with them about whether things have gotten worse. If they’re at high risk for suicide, I’m checking in to make sure that they’ve got new plans and ways to connect with people to reduce isolation, keeping in mind the social distancing that we’re asked to engage in so that they can do that safely.
It’s important to check and see if they have had any losses, whether it’s a financial loss or a personal loss of people that they care about. Also have them think through ways to stay entertained, which tends to help manage their own anxiety.
Every coping strategy we outline for patients also applies to mental health professionals. However, you would add to it the real need to take time to recharge, to take breaks, time off. It can feel overwhelming and like you need to just keep going. But the more that you get stuck in that mode of overdoing it, the less effective you’re going to be in helping people and also the more likely that you’ll be at risk of perhaps being one of the people that needs help.
It’s also important to make sure you’re staying connected with family and friends virtually, in whatever ways you can safely do that with social distancing.
So take a break to watch some Netflix now and then?
Yes!
A version of this article originally appeared on Medscape.com.
Plan now to address the COVID-19 mental health fallout
COVID-19 affects the physical, psychological, and social health of people around the world. In the United States, newly reported cases are rising at alarming rates.
As of early May, more than 1.3 million people were confirmed to be COVID-19 infected in the United States and more than 4 million cases were reported globally.1
According to new internal projections from the Centers for Disease Control and Prevention, by June 1, the number of daily deaths could reach about 3,000. By the end of June, a draft CDC report projects that the United States will see 200,000 new cases each day.2
COVID-19 undeniably harms mental health. It gravely instills uncertainty and anxiety, sometimes compounded by the grief of losing loved ones and not being able to mourn those losses in traditional ways. The pandemic also has led to occupational and/or financial losses. Physical distancing and shelter-in-place practices make it even harder to cope with those stresses, although those practices mitigate the dangers. The fears tied to those practices are thought to be keeping some patients with health problems from seeking needed care from hospital EDs.3 In light of the mental health crisis emerging because of the profound impact of this pandemic on all aspects of life, clinicians should start working with public health and political leaders to develop plans to address these issues now.
Known impact of previous outbreaks
Previous disease outbreaks evidence a similar pattern of heightened anxiety as the patterns seen with COVID-19. For example, during the 2009 swine flu outbreak, 36 surveys of more than 3,000 participants in the United Kingdom found that 9.6%-32.9% of the participants were “very” or “fairly” worried about the possibility of contracting swine flu.4 The 1995 Ebola outbreak in the Democratic Republic of the Congo produced stigmatization tied to the illness. That outbreak provided many lessons for physicians.5
The metaphors ascribed to different diseases affect communities’ responses to it. The SARS virus has been particularly insidious and has been thought of as a “plague.”6 Epidemics of all kinds cause fears, not only of contracting the disease and dying, but also of social exclusion.7 The emotional responses to COVID-19 can precipitate anxiety, depression, insomnia, and somatic symptoms.
Repeated exposure to news media about the disease adds to theses stresss.10 Constant news consumption can result in panicky hoarding of resources, such as masks; gloves; first-aid kits; alcohol hand rubs; and daily necessities such as food, water, and toilet paper.
Who is most affected by outbreaks?
Those most affected after a disease outbreak are patients, their families, and medical personnel. In one study, researchers who conducted an online survey of 1,210 respondents in 194 cities in China during the early phase of the outbreak found that the psychological effects were worst among women, students, and vulnerable populations.11
Meanwhile, a 2003 cross-sectional survey of 1,115 ethnic Chinese adults in Hong Kong who responded to the SARS outbreak found that the respondents most likely to heed precautionary measures against the infection were “older, female, more educated people as well as those with a positive contact history and SARS-like symptoms.”12
Negative mental health consequences of a disease outbreak might persist long after the infection has dissipated. An increased association has been found between people with mental illness and posttraumatic stress following many disasters.13,14,15
Political and health care leaders should develop plans aimed at helping people copewith pandemics.16 Such strategies should include prioritizing treatment of the physical and mental health needs of patients infected with COVID-19 and of the general population. Screening for anxiety, depression, and suicidal thoughts ought to be implemented, and specialized psychiatric care teams should be assigned.17 We know that psychiatrists and other physicians turned to telemedicine to provide support, psychotherapy, and medical attention to patients soon after physical distancing measures were put into place. Those kinds of quick responses are important for our patients.
Fear of contagious diseases often creates social divisions. Governments should offer accurate information to reduce the detrimental effect of rumors and false propaganda.18 “Social distancing” is a misleading term; these practices should be referred to as “physical distancing.” We should encourage patients to maintain interpersonal contacts – albeit at a distance – to reach out to those in need, and to support one another during these troubled times.19
References
1. World Health Organization. Situation Report–107. 2020 May 6.
2. Centers for Disease Control and Prevention. Situation Update. 2020 Apr 30.
3. O’Brien M. “Are Americans in medical crisis avoiding the ER due to coronavirus?” PBS Newshour. 2020 May 6.
4. Rubin G et al. Health Technol Assess. 2010 Jul;14(340):183-266.
5. Hall R et al. Gen Hosp Psychiatry. 2008 Sep-Oct;30(5):466-52.
6. Verghese A. Clin Infect Dis. 2004;38:932-3.
7. Interagency Standing Committee. Briefing note on addressing health and psychosocial aspects of COVID-19 Outbreak – Version 11. 2020 Feb.
8. Sim K et al. J Psychosom Res. 2010;68:195-202.
9. Shigemura J et al. Psychiatry Clin Neurosci. 2020;74:281-2.
10. Garfin DR et al. Health Psychol. 2020 May;39(5):355-7.
11. Wang C et al. Int J Environ Res Public Health. 2020 Mar 6. doi: 10.3390/ijerph1751729.
12. Leung GM et al. J Epidemiol Community Health. 2003 Nov;57(1):857-63.
13. Xiang Y et al. Int J Biol Sci. 2020;16:1741-4.
14. Alvarez J, Hunt M. J Trauma Stress. 2005 Oct 18(5);18:497-505.
15. Cukor J et al. Depress Anxiety. 2011 Mar;28(3):210-7.
16. Horton R. Lancet. 2020 Feb;395(10222):400.
17. Xiang Y-T et al. Lancet Psychiatry. 2020 Feb 4;7:228-9.
18. World Health Organization. “Rational use of personal protective equipment (PPE) for coronavirus (COVID-19).” Interim Guidance. 2020 Mar.
19. Brooks S et al. Lancet 2020 Mar 14;395:912-20.
Dr. Doppalapudi is affiliated with Griffin Memorial Hospital in Norman, Okla. Dr. Lippmann is emeritus professor of psychiatry and also in family medicine at the University of Louisville (Ky.) Dr. Doppalapudi and Dr. Lippmann disclosed no conflicts of interest.
COVID-19 affects the physical, psychological, and social health of people around the world. In the United States, newly reported cases are rising at alarming rates.
As of early May, more than 1.3 million people were confirmed to be COVID-19 infected in the United States and more than 4 million cases were reported globally.1
According to new internal projections from the Centers for Disease Control and Prevention, by June 1, the number of daily deaths could reach about 3,000. By the end of June, a draft CDC report projects that the United States will see 200,000 new cases each day.2
COVID-19 undeniably harms mental health. It gravely instills uncertainty and anxiety, sometimes compounded by the grief of losing loved ones and not being able to mourn those losses in traditional ways. The pandemic also has led to occupational and/or financial losses. Physical distancing and shelter-in-place practices make it even harder to cope with those stresses, although those practices mitigate the dangers. The fears tied to those practices are thought to be keeping some patients with health problems from seeking needed care from hospital EDs.3 In light of the mental health crisis emerging because of the profound impact of this pandemic on all aspects of life, clinicians should start working with public health and political leaders to develop plans to address these issues now.
Known impact of previous outbreaks
Previous disease outbreaks evidence a similar pattern of heightened anxiety as the patterns seen with COVID-19. For example, during the 2009 swine flu outbreak, 36 surveys of more than 3,000 participants in the United Kingdom found that 9.6%-32.9% of the participants were “very” or “fairly” worried about the possibility of contracting swine flu.4 The 1995 Ebola outbreak in the Democratic Republic of the Congo produced stigmatization tied to the illness. That outbreak provided many lessons for physicians.5
The metaphors ascribed to different diseases affect communities’ responses to it. The SARS virus has been particularly insidious and has been thought of as a “plague.”6 Epidemics of all kinds cause fears, not only of contracting the disease and dying, but also of social exclusion.7 The emotional responses to COVID-19 can precipitate anxiety, depression, insomnia, and somatic symptoms.
Repeated exposure to news media about the disease adds to theses stresss.10 Constant news consumption can result in panicky hoarding of resources, such as masks; gloves; first-aid kits; alcohol hand rubs; and daily necessities such as food, water, and toilet paper.
Who is most affected by outbreaks?
Those most affected after a disease outbreak are patients, their families, and medical personnel. In one study, researchers who conducted an online survey of 1,210 respondents in 194 cities in China during the early phase of the outbreak found that the psychological effects were worst among women, students, and vulnerable populations.11
Meanwhile, a 2003 cross-sectional survey of 1,115 ethnic Chinese adults in Hong Kong who responded to the SARS outbreak found that the respondents most likely to heed precautionary measures against the infection were “older, female, more educated people as well as those with a positive contact history and SARS-like symptoms.”12
Negative mental health consequences of a disease outbreak might persist long after the infection has dissipated. An increased association has been found between people with mental illness and posttraumatic stress following many disasters.13,14,15
Political and health care leaders should develop plans aimed at helping people copewith pandemics.16 Such strategies should include prioritizing treatment of the physical and mental health needs of patients infected with COVID-19 and of the general population. Screening for anxiety, depression, and suicidal thoughts ought to be implemented, and specialized psychiatric care teams should be assigned.17 We know that psychiatrists and other physicians turned to telemedicine to provide support, psychotherapy, and medical attention to patients soon after physical distancing measures were put into place. Those kinds of quick responses are important for our patients.
Fear of contagious diseases often creates social divisions. Governments should offer accurate information to reduce the detrimental effect of rumors and false propaganda.18 “Social distancing” is a misleading term; these practices should be referred to as “physical distancing.” We should encourage patients to maintain interpersonal contacts – albeit at a distance – to reach out to those in need, and to support one another during these troubled times.19
References
1. World Health Organization. Situation Report–107. 2020 May 6.
2. Centers for Disease Control and Prevention. Situation Update. 2020 Apr 30.
3. O’Brien M. “Are Americans in medical crisis avoiding the ER due to coronavirus?” PBS Newshour. 2020 May 6.
4. Rubin G et al. Health Technol Assess. 2010 Jul;14(340):183-266.
5. Hall R et al. Gen Hosp Psychiatry. 2008 Sep-Oct;30(5):466-52.
6. Verghese A. Clin Infect Dis. 2004;38:932-3.
7. Interagency Standing Committee. Briefing note on addressing health and psychosocial aspects of COVID-19 Outbreak – Version 11. 2020 Feb.
8. Sim K et al. J Psychosom Res. 2010;68:195-202.
9. Shigemura J et al. Psychiatry Clin Neurosci. 2020;74:281-2.
10. Garfin DR et al. Health Psychol. 2020 May;39(5):355-7.
11. Wang C et al. Int J Environ Res Public Health. 2020 Mar 6. doi: 10.3390/ijerph1751729.
12. Leung GM et al. J Epidemiol Community Health. 2003 Nov;57(1):857-63.
13. Xiang Y et al. Int J Biol Sci. 2020;16:1741-4.
14. Alvarez J, Hunt M. J Trauma Stress. 2005 Oct 18(5);18:497-505.
15. Cukor J et al. Depress Anxiety. 2011 Mar;28(3):210-7.
16. Horton R. Lancet. 2020 Feb;395(10222):400.
17. Xiang Y-T et al. Lancet Psychiatry. 2020 Feb 4;7:228-9.
18. World Health Organization. “Rational use of personal protective equipment (PPE) for coronavirus (COVID-19).” Interim Guidance. 2020 Mar.
19. Brooks S et al. Lancet 2020 Mar 14;395:912-20.
Dr. Doppalapudi is affiliated with Griffin Memorial Hospital in Norman, Okla. Dr. Lippmann is emeritus professor of psychiatry and also in family medicine at the University of Louisville (Ky.) Dr. Doppalapudi and Dr. Lippmann disclosed no conflicts of interest.
COVID-19 affects the physical, psychological, and social health of people around the world. In the United States, newly reported cases are rising at alarming rates.
As of early May, more than 1.3 million people were confirmed to be COVID-19 infected in the United States and more than 4 million cases were reported globally.1
According to new internal projections from the Centers for Disease Control and Prevention, by June 1, the number of daily deaths could reach about 3,000. By the end of June, a draft CDC report projects that the United States will see 200,000 new cases each day.2
COVID-19 undeniably harms mental health. It gravely instills uncertainty and anxiety, sometimes compounded by the grief of losing loved ones and not being able to mourn those losses in traditional ways. The pandemic also has led to occupational and/or financial losses. Physical distancing and shelter-in-place practices make it even harder to cope with those stresses, although those practices mitigate the dangers. The fears tied to those practices are thought to be keeping some patients with health problems from seeking needed care from hospital EDs.3 In light of the mental health crisis emerging because of the profound impact of this pandemic on all aspects of life, clinicians should start working with public health and political leaders to develop plans to address these issues now.
Known impact of previous outbreaks
Previous disease outbreaks evidence a similar pattern of heightened anxiety as the patterns seen with COVID-19. For example, during the 2009 swine flu outbreak, 36 surveys of more than 3,000 participants in the United Kingdom found that 9.6%-32.9% of the participants were “very” or “fairly” worried about the possibility of contracting swine flu.4 The 1995 Ebola outbreak in the Democratic Republic of the Congo produced stigmatization tied to the illness. That outbreak provided many lessons for physicians.5
The metaphors ascribed to different diseases affect communities’ responses to it. The SARS virus has been particularly insidious and has been thought of as a “plague.”6 Epidemics of all kinds cause fears, not only of contracting the disease and dying, but also of social exclusion.7 The emotional responses to COVID-19 can precipitate anxiety, depression, insomnia, and somatic symptoms.
Repeated exposure to news media about the disease adds to theses stresss.10 Constant news consumption can result in panicky hoarding of resources, such as masks; gloves; first-aid kits; alcohol hand rubs; and daily necessities such as food, water, and toilet paper.
Who is most affected by outbreaks?
Those most affected after a disease outbreak are patients, their families, and medical personnel. In one study, researchers who conducted an online survey of 1,210 respondents in 194 cities in China during the early phase of the outbreak found that the psychological effects were worst among women, students, and vulnerable populations.11
Meanwhile, a 2003 cross-sectional survey of 1,115 ethnic Chinese adults in Hong Kong who responded to the SARS outbreak found that the respondents most likely to heed precautionary measures against the infection were “older, female, more educated people as well as those with a positive contact history and SARS-like symptoms.”12
Negative mental health consequences of a disease outbreak might persist long after the infection has dissipated. An increased association has been found between people with mental illness and posttraumatic stress following many disasters.13,14,15
Political and health care leaders should develop plans aimed at helping people copewith pandemics.16 Such strategies should include prioritizing treatment of the physical and mental health needs of patients infected with COVID-19 and of the general population. Screening for anxiety, depression, and suicidal thoughts ought to be implemented, and specialized psychiatric care teams should be assigned.17 We know that psychiatrists and other physicians turned to telemedicine to provide support, psychotherapy, and medical attention to patients soon after physical distancing measures were put into place. Those kinds of quick responses are important for our patients.
Fear of contagious diseases often creates social divisions. Governments should offer accurate information to reduce the detrimental effect of rumors and false propaganda.18 “Social distancing” is a misleading term; these practices should be referred to as “physical distancing.” We should encourage patients to maintain interpersonal contacts – albeit at a distance – to reach out to those in need, and to support one another during these troubled times.19
References
1. World Health Organization. Situation Report–107. 2020 May 6.
2. Centers for Disease Control and Prevention. Situation Update. 2020 Apr 30.
3. O’Brien M. “Are Americans in medical crisis avoiding the ER due to coronavirus?” PBS Newshour. 2020 May 6.
4. Rubin G et al. Health Technol Assess. 2010 Jul;14(340):183-266.
5. Hall R et al. Gen Hosp Psychiatry. 2008 Sep-Oct;30(5):466-52.
6. Verghese A. Clin Infect Dis. 2004;38:932-3.
7. Interagency Standing Committee. Briefing note on addressing health and psychosocial aspects of COVID-19 Outbreak – Version 11. 2020 Feb.
8. Sim K et al. J Psychosom Res. 2010;68:195-202.
9. Shigemura J et al. Psychiatry Clin Neurosci. 2020;74:281-2.
10. Garfin DR et al. Health Psychol. 2020 May;39(5):355-7.
11. Wang C et al. Int J Environ Res Public Health. 2020 Mar 6. doi: 10.3390/ijerph1751729.
12. Leung GM et al. J Epidemiol Community Health. 2003 Nov;57(1):857-63.
13. Xiang Y et al. Int J Biol Sci. 2020;16:1741-4.
14. Alvarez J, Hunt M. J Trauma Stress. 2005 Oct 18(5);18:497-505.
15. Cukor J et al. Depress Anxiety. 2011 Mar;28(3):210-7.
16. Horton R. Lancet. 2020 Feb;395(10222):400.
17. Xiang Y-T et al. Lancet Psychiatry. 2020 Feb 4;7:228-9.
18. World Health Organization. “Rational use of personal protective equipment (PPE) for coronavirus (COVID-19).” Interim Guidance. 2020 Mar.
19. Brooks S et al. Lancet 2020 Mar 14;395:912-20.
Dr. Doppalapudi is affiliated with Griffin Memorial Hospital in Norman, Okla. Dr. Lippmann is emeritus professor of psychiatry and also in family medicine at the University of Louisville (Ky.) Dr. Doppalapudi and Dr. Lippmann disclosed no conflicts of interest.
COVID-19: A ‘marathon, not a sprint’ for psychiatry
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The tragic death by suicide of an emergency department physician who had been caring for COVID-19 patients in New York City underscores the huge psychological impact of the pandemic – which will linger long after the virus is gone, experts say.
“For frontline responders, the trauma of witnessing so much illness and death will have lasting effects for many,” Bruce Schwartz, MD, president of the American Psychiatric Association (APA), said during the opening session of the annual meeting of the American Psychiatric Association, which was held as a virtual live event, replacing the organization’s canceled annual meeting.
“We will need the full workforce to cope with the psychiatric effects” of the pandemic, added Dr. Schwartz, deputy chairman and professor, department of psychiatry and behavioral sciences, Montefiore Medical Center and Albert Einstein College of Medicine, New York.
Joshua Morganstein, MD, chair of the APA’s Committee on the Psychiatric Dimensions of Disaster, led an afternoon session at the virtual meeting on “healthcare worker and organizational sustainment during COVID-19.”
The crisis is shaping up to be “a marathon, not a sprint; and self-care will remain a critical and ongoing issue. We are in this together,” he said.
Once the pandemic passes, Dr. Morganstein added.
Psychological first aid
It is important to realize that the psychological and behavioral effects of disasters are experienced by “more people, over a greater geography, across a much longer period of time than all other medical effects combined. This is important for disaster resource planning,” Dr. Morganstein told meeting attendees.
At times of crisis, many people will experience distress reactions and engage in behaviors that put their health at risk. Insomnia, increased alcohol and substance use, and family conflict are common and have a negative impact on functioning, he said.
In addition, pandemics result in unique responses. Protracted fear and uncertainty, elements of isolation, anger, misinformation, and faltering confidence in government/institutions may alter perceptions of risk.
“It’s the perception of risk, not the actual risk, that will ultimately determine how people behave,” Dr. Morganstein said.
“The ability to influence risk perception will alter the degree to which any group, community, or population ultimately chooses to engage in or reject recommended health behaviors,” he added.
In times of crisis, it’s also helpful to keep in mind and act upon the five essential elements of “psychological first aid,” he noted. These are safety, calming, self/community efficacy, social connectedness, and hope/optimism.
Psychological first aid is an evidence-based framework of supporting resilience in individuals, communities, and organizations, Dr. Morganstein said.
Individuals have a wide range of needs during times of crisis, and support should be tailored accordingly, he noted. As with many crises, instrumental support needs are significant and may be the primary need for many people. These include the need for food, clothing, rent/mortgage, financial relief, and child care.
Providing emotional support – empathy, validation, self-actualization, encouragement, and insight – will help individuals engage with instrumental supports.
“The reality is that it’s often difficult to talk about being sad when you feel hungry or worried you can’t pay the rent,” said Dr. Morganstein.
He also emphasized the importance of appropriate messaging and language during a crisis. These can have a profound impact on community well-being and the willingness of the public to engage in recommended health behaviors.
“As psychiatrists, we understand [that] the words we choose when we discuss this pandemic will have power. Communication is not only a means by which we deliver interventions, but it is, in and of itself, a behavioral health intervention. Good communication can serve to normalize experiences and function as an antidote to distress during times of uncertainty,” Dr. Morganstein said.
Importantly, “we need to remind people that eventually this will end and the vast majority of people, including those who have difficulties along the way, will ultimately be okay.”
The APA has provided a COVID-19 resource page on its website.
Dr. Morganstein and Dr. Schwartz have reported no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FROM APA 2020



