User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
All’s Well That Ends Swell(ing)
ANSWER
The correct answer is elephantiasis nostras verrucosa (ENV; choice “d”).
DISCUSSION
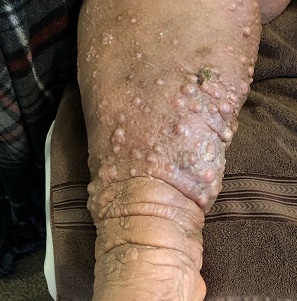
ENV is a rare condition of advanced cutaneous hypertrophy secondary to a combination of contributing factors including: a sedentary lifestyle, obesity, chronic venous stasis, repeated bouts of lymphangitis, cellulitis, and congestive heart failure (CHF). Most commonly affecting the lower extremities, it is occasionally seen in other dependent areas such as the scrotum and the abdominal pannus. It is, essentially, an exaggerated form of cutaneous lymphedema that causes the skin to become increasingly thick and fibrotic, changes which also reduce blood flow to or from the area.
Despite its name, ENV is not associated with elephantiasis, more commonly known as lymphatic filariasis (choice “b”). Although that condition manifests with similar skin changes, it is typically seen only in those who live in tropical areas where these organisms are endemic—places this patient has never visited.
There was no reason to believe that these skin changes were attributable to warts (choice “a”). Biopsy would have settled that question but also would have run the risk of creating a nonhealing wound, which could easily turn into an ulcer.
Lymphedema (choice “c”) was clearly present, but it was quite advanced—far beyond what is usually seen in venous insufficiency. This diagnosis would not, by itself, explain the nodules or extreme fibrosis.
Other potential causes for these skin changes include postradiation and pretibial myxedema, which had been ruled out prior to the dermatology consult.
TREATMENT
As with simple venous insufficiency, treatment of ENV consists of compression, elevation, and weight loss. For this patient, the diuretics prescribed as part of her CHF treatment might help a bit, but her prognosis is guarded at best.
ANSWER
The correct answer is elephantiasis nostras verrucosa (ENV; choice “d”).
DISCUSSION
ENV is a rare condition of advanced cutaneous hypertrophy secondary to a combination of contributing factors including: a sedentary lifestyle, obesity, chronic venous stasis, repeated bouts of lymphangitis, cellulitis, and congestive heart failure (CHF). Most commonly affecting the lower extremities, it is occasionally seen in other dependent areas such as the scrotum and the abdominal pannus. It is, essentially, an exaggerated form of cutaneous lymphedema that causes the skin to become increasingly thick and fibrotic, changes which also reduce blood flow to or from the area.
Despite its name, ENV is not associated with elephantiasis, more commonly known as lymphatic filariasis (choice “b”). Although that condition manifests with similar skin changes, it is typically seen only in those who live in tropical areas where these organisms are endemic—places this patient has never visited.
There was no reason to believe that these skin changes were attributable to warts (choice “a”). Biopsy would have settled that question but also would have run the risk of creating a nonhealing wound, which could easily turn into an ulcer.
Lymphedema (choice “c”) was clearly present, but it was quite advanced—far beyond what is usually seen in venous insufficiency. This diagnosis would not, by itself, explain the nodules or extreme fibrosis.
Other potential causes for these skin changes include postradiation and pretibial myxedema, which had been ruled out prior to the dermatology consult.
TREATMENT
As with simple venous insufficiency, treatment of ENV consists of compression, elevation, and weight loss. For this patient, the diuretics prescribed as part of her CHF treatment might help a bit, but her prognosis is guarded at best.
ANSWER
The correct answer is elephantiasis nostras verrucosa (ENV; choice “d”).
DISCUSSION
ENV is a rare condition of advanced cutaneous hypertrophy secondary to a combination of contributing factors including: a sedentary lifestyle, obesity, chronic venous stasis, repeated bouts of lymphangitis, cellulitis, and congestive heart failure (CHF). Most commonly affecting the lower extremities, it is occasionally seen in other dependent areas such as the scrotum and the abdominal pannus. It is, essentially, an exaggerated form of cutaneous lymphedema that causes the skin to become increasingly thick and fibrotic, changes which also reduce blood flow to or from the area.
Despite its name, ENV is not associated with elephantiasis, more commonly known as lymphatic filariasis (choice “b”). Although that condition manifests with similar skin changes, it is typically seen only in those who live in tropical areas where these organisms are endemic—places this patient has never visited.
There was no reason to believe that these skin changes were attributable to warts (choice “a”). Biopsy would have settled that question but also would have run the risk of creating a nonhealing wound, which could easily turn into an ulcer.
Lymphedema (choice “c”) was clearly present, but it was quite advanced—far beyond what is usually seen in venous insufficiency. This diagnosis would not, by itself, explain the nodules or extreme fibrosis.
Other potential causes for these skin changes include postradiation and pretibial myxedema, which had been ruled out prior to the dermatology consult.
TREATMENT
As with simple venous insufficiency, treatment of ENV consists of compression, elevation, and weight loss. For this patient, the diuretics prescribed as part of her CHF treatment might help a bit, but her prognosis is guarded at best.
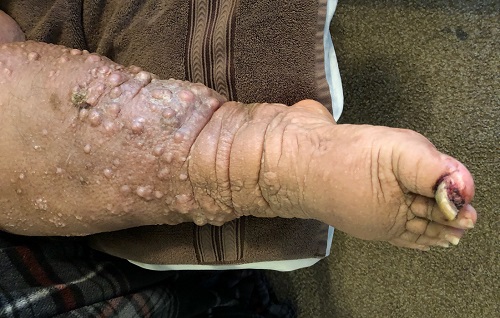
A 61-year-old black woman presents for worrisome skin changes on her lower extremities. She reports that the condition is generally uncomfortable, but during occasional flares, it causes serious pain. She’s been affected for “many years” without diagnosis or resolution. It was her new primary care provider who, after seeing the lesions, sent her to dermatology.
The patient’s medical history includes diabetes, congestive heart failure, and obesity. All are being managed reasonably well.
Examination, performed while she is in a recumbent position, reveals legs swollen out of proportion to the rest of her body. Little or no erythema is noted. Both legs are affected equally, but only from just below the knees down to and including the feet. These areas, including her feet, are quite swollen, though no pitting edema can be provoked. The skin is quite firm and studded with multiple discrete and confluent 1-2 cm firm nodules. The skin around her ankles feels almost “woody” to the touch. There is no tenderness or increased warmth on palpation, nor is any drainage noted. (She also has a dystrophic great toenail that was partially avulsed by recent trauma.)
‘Twincretin’ meets primary endpoints in five pivotal diabetes trials
The investigational, novel, injected once-weekly “twincretin” tirzepatide met its primary efficacy endpoint of significantly cutting hemoglobin A1c as well as its secondary weight-loss endpoint in patients with type 2 diabetes when compared with control patients in top-line results from each of five discrete pivotal trials.
The company developing tirzepatide, Lilly, announced these results in a series of four press releases issued during December 2020–May 2021. Scientific reports on the outcomes from four of these trials are scheduled during the American Diabetes Association’s Scientific Sessions being held virtually in late June 2021, with results from the fifth on track for a report during the annual meeting of the European Association for the Study of Diabetes in September 2021.
Tirzepatide is a “twincretin” because it combines in a single molecule two different gut-hormone activities. It works as both a glucagonlike peptide–1 receptor agonist (GLP-1 RA) and as an agent that mimics the glucose-dependent insulinotropic polypeptide (GIP).
While diabetologists qualified their comments on these results because of the limited scope and format of the five reports to date, they also expressed enthusiasm over what the press releases said.
Results give hope
“It’s quite exciting, but of course we would like to go by the data that’s presented” at upcoming meetings, commented Robert A. Gabbay, MD, PhD, chief science and medical officer of the American Diabetes Association in Arlington, Va. “The idea of GLP-1 and GIP activities working together has been out there for a while, but without any therapeutic options that leverage this,” he said in an interview.
“The preliminary results give us hope that tirzepatide will be a very effective glucose-lowering agent, perhaps the most effective among all options currently available, including insulin,” commented Ildiko Lingvay, MD, a diabetologist and professor at the University of Texas Southwestern Medical Center, Dallas. “Tirzepatide might have the added benefit of clinically meaningful weight loss,” and “the adverse event profile seems to be in line with what we are accustomed to with the GLP-1 RA class. I look forward to seeing the full results. Tirzepatide promises to be a great addition for type 2 diabetes,” Dr. Lingvay said in an interview.
A rare head-to-head against semaglutide
The five phase 3, randomized controlled trials described by Lilly in its four press releases all belong to the SURPASS series of studies for this agent. Perhaps the most intriguing of the five were results from SURPASS-2, announced in a release on March 4. This trial randomized 1,879 patients from the United States or any of seven other countries to 40 weeks of open-label treatment with one of three different dosages of tirzepatide administered by injection once weekly, or to the control group that received a weekly 1-mg injection of semaglutide (Ozempic), the highest dosage approved for controlling glycemia in patients with type 2 diabetes at the time the study launched.
In SURPASS-2 all three tested dosages of tirzepatide led to a significantly larger reduction, from baseline in A1c, compared with semaglutide, after 40 weeks, according to the Lilly release. Each of the three tirzepatide dosages also led to significantly greater weight loss from baseline, compared with semaglutide, and significantly greater percentages of patients who achieved an A1c of less than 7%, compared with semaglutide.
As an example, the highest tested tirzepatide dosage of 15 mg weekly led to an average A1c reduction from baseline of 2.46% and an average weight loss from baseline of 12.4 kg; 92% of patients achieved an A1c of less than 7%, and 51% had their A1c fall below 5.7% which indicates completely normalization of glycemic control. By comparison, the patients randomized to treatment with semaglutide had an average 1.86% reduction in their A1c level from baseline and a 6.2-kg average cut in body weight from baseline; 81% achieved an A1c of less than 7%, and 20% reached an A1c of less than 5.7%.
There are caveats
While these findings are notable as a rare example of an industry-sponsored head-to-head comparison of two new agents, the study comes with a few important asterisks.
First, it was open label, a curious limitation given that both agents are delivered by the same delivery method and schedule. “I cannot conclude based on this study that tirzepatide is superior because it was open label,” commented Anastassia Amaro, MD, medical director of Penn Metabolic Medicine at the University of Pennsylvania, Philadelphia.
“The gold standard is the double-blind study. An open-label design is a limitation,” agreed Dr. Gabbay.
A second caveat is that the Food and Drug Administration recently approved a higher dosage of semaglutide (2.4 g once/week) for treating overweight or obesity in patients with type 2 diabetes and in those without diabetes but a different weight-related condition such as hypertension of hypercholesterolemia. This means that the tested comparator dosage of 1 mg/week is no longer the maximum that most patients treated with semaglutide for glycemic control can receive.
“The inevitable question” about this comparison study is “what about a higher semaglutide dose,” and how might tirzepatide perform relative to that, said Dr. Gabbay. The recently approved higher dosage of semaglutide “adds an interesting wrinkle.”
Lilly has launched a series of studies testing tirzepatide as a treatment for overweight or obesity in people without diabetes, but the results are not expected until sometime in 2022 or 2023.
And there’s a third caveat: Semaglutide has already shown its value for cardiovascular risk reduction in patients with type 2 diabetes in the SUSTAIN 6 trial with nearly 3,300 randomized patients followed for 2 years and reported in 2016. The cardiovascular outcomes trial for tirzepatide, SURPASS-CVOT with more than 12,000 patients with type 2 diabetes, is underway but its results are not expected until 2024.
Despite these important limitations, a blinded comparison of tirzepatide and higher-dose semaglutide is unlikely, Dr. Amaro predicted. “It’s not worth the expense,” she said in an interview. A more likely scenario will be that, if tirzepatide enters the U.S. market, decisions on whether to treat patients with it or semaglutide will pivot on factors like the cost for treatment to individual patients based on their insurance coverage and tolerability, suggested both Dr. Amaro and Dr. Gabbay. “Physicians will need to develop a sense for tirzepatide: Do patients tolerate it and are they happy using it?” Dr. Amaro said.
Tirzepatide versus insulin, or on top of insulin
The other four trials in patients with type 2 diabetes reported by Lilly in releases included SURPASS-1, which randomized 478 patients to treatment with tirzepatide or placebo as monotherapy; SURPASS-3, which randomized 1,444 patients to tirzepatide or insulin degludec (Tresiba) on top of background treatment with metformin; SURPASS-4, which randomized 2,002 patients with high cardiovascular disease risk to treatment with tirzepatide or insulin glargine (Lantus) on top of background treatment with one to three different oral drugs; and SURPASS-5, which randomized 475 patients to treatment with tirzepatide or placebo on top of background treatment with insulin glargine and optional addition of metformin. Altogether, the five trials randomized nearly 6,300 patients.
The studies that compared tirzepatide against two different types of insulin, and the third that tested tirzepatide on top of insulin glargine, are especially notable. “It’s good to see that the combination [of tirzepatide and insulin glargine] works without causing major adverse events,” said Dr. Amaro.
“These are fair and helpful comparisons. I applaud Lilly for doing the right kind of comparisons,” said Dr. Gabbay.
In total, the five studies “provide evidence that tirzepatide will be effective at all stages of type 2 diabetes and can safely be used in combination with other glucose-lowering agents, including insulin,” said Dr. Lingvay. The studies with active comparator agents “allow us to compare tirzepatide’s efficacy against established therapies.”
The SURPASS trials were sponsored by Lilly, which is developing tirzepatide. Dr. Gabbay had no relevant disclosures. Dr. Lingvay has received research funds, consulting and advisory fees, or other support from Lilly as well as from several other companies including Novo Nordisk, which markets semaglutide (Ozempic) and insulin degludec (Tresiba), and Sanofi, which markets insulin glargine (Lantus). Dr. Amaro has received research funding from Lilly and from Fractyl, and has been a consultant to and received research funding from Novo Nordisk.
The investigational, novel, injected once-weekly “twincretin” tirzepatide met its primary efficacy endpoint of significantly cutting hemoglobin A1c as well as its secondary weight-loss endpoint in patients with type 2 diabetes when compared with control patients in top-line results from each of five discrete pivotal trials.
The company developing tirzepatide, Lilly, announced these results in a series of four press releases issued during December 2020–May 2021. Scientific reports on the outcomes from four of these trials are scheduled during the American Diabetes Association’s Scientific Sessions being held virtually in late June 2021, with results from the fifth on track for a report during the annual meeting of the European Association for the Study of Diabetes in September 2021.
Tirzepatide is a “twincretin” because it combines in a single molecule two different gut-hormone activities. It works as both a glucagonlike peptide–1 receptor agonist (GLP-1 RA) and as an agent that mimics the glucose-dependent insulinotropic polypeptide (GIP).
While diabetologists qualified their comments on these results because of the limited scope and format of the five reports to date, they also expressed enthusiasm over what the press releases said.
Results give hope
“It’s quite exciting, but of course we would like to go by the data that’s presented” at upcoming meetings, commented Robert A. Gabbay, MD, PhD, chief science and medical officer of the American Diabetes Association in Arlington, Va. “The idea of GLP-1 and GIP activities working together has been out there for a while, but without any therapeutic options that leverage this,” he said in an interview.
“The preliminary results give us hope that tirzepatide will be a very effective glucose-lowering agent, perhaps the most effective among all options currently available, including insulin,” commented Ildiko Lingvay, MD, a diabetologist and professor at the University of Texas Southwestern Medical Center, Dallas. “Tirzepatide might have the added benefit of clinically meaningful weight loss,” and “the adverse event profile seems to be in line with what we are accustomed to with the GLP-1 RA class. I look forward to seeing the full results. Tirzepatide promises to be a great addition for type 2 diabetes,” Dr. Lingvay said in an interview.
A rare head-to-head against semaglutide
The five phase 3, randomized controlled trials described by Lilly in its four press releases all belong to the SURPASS series of studies for this agent. Perhaps the most intriguing of the five were results from SURPASS-2, announced in a release on March 4. This trial randomized 1,879 patients from the United States or any of seven other countries to 40 weeks of open-label treatment with one of three different dosages of tirzepatide administered by injection once weekly, or to the control group that received a weekly 1-mg injection of semaglutide (Ozempic), the highest dosage approved for controlling glycemia in patients with type 2 diabetes at the time the study launched.
In SURPASS-2 all three tested dosages of tirzepatide led to a significantly larger reduction, from baseline in A1c, compared with semaglutide, after 40 weeks, according to the Lilly release. Each of the three tirzepatide dosages also led to significantly greater weight loss from baseline, compared with semaglutide, and significantly greater percentages of patients who achieved an A1c of less than 7%, compared with semaglutide.
As an example, the highest tested tirzepatide dosage of 15 mg weekly led to an average A1c reduction from baseline of 2.46% and an average weight loss from baseline of 12.4 kg; 92% of patients achieved an A1c of less than 7%, and 51% had their A1c fall below 5.7% which indicates completely normalization of glycemic control. By comparison, the patients randomized to treatment with semaglutide had an average 1.86% reduction in their A1c level from baseline and a 6.2-kg average cut in body weight from baseline; 81% achieved an A1c of less than 7%, and 20% reached an A1c of less than 5.7%.
There are caveats
While these findings are notable as a rare example of an industry-sponsored head-to-head comparison of two new agents, the study comes with a few important asterisks.
First, it was open label, a curious limitation given that both agents are delivered by the same delivery method and schedule. “I cannot conclude based on this study that tirzepatide is superior because it was open label,” commented Anastassia Amaro, MD, medical director of Penn Metabolic Medicine at the University of Pennsylvania, Philadelphia.
“The gold standard is the double-blind study. An open-label design is a limitation,” agreed Dr. Gabbay.
A second caveat is that the Food and Drug Administration recently approved a higher dosage of semaglutide (2.4 g once/week) for treating overweight or obesity in patients with type 2 diabetes and in those without diabetes but a different weight-related condition such as hypertension of hypercholesterolemia. This means that the tested comparator dosage of 1 mg/week is no longer the maximum that most patients treated with semaglutide for glycemic control can receive.
“The inevitable question” about this comparison study is “what about a higher semaglutide dose,” and how might tirzepatide perform relative to that, said Dr. Gabbay. The recently approved higher dosage of semaglutide “adds an interesting wrinkle.”
Lilly has launched a series of studies testing tirzepatide as a treatment for overweight or obesity in people without diabetes, but the results are not expected until sometime in 2022 or 2023.
And there’s a third caveat: Semaglutide has already shown its value for cardiovascular risk reduction in patients with type 2 diabetes in the SUSTAIN 6 trial with nearly 3,300 randomized patients followed for 2 years and reported in 2016. The cardiovascular outcomes trial for tirzepatide, SURPASS-CVOT with more than 12,000 patients with type 2 diabetes, is underway but its results are not expected until 2024.
Despite these important limitations, a blinded comparison of tirzepatide and higher-dose semaglutide is unlikely, Dr. Amaro predicted. “It’s not worth the expense,” she said in an interview. A more likely scenario will be that, if tirzepatide enters the U.S. market, decisions on whether to treat patients with it or semaglutide will pivot on factors like the cost for treatment to individual patients based on their insurance coverage and tolerability, suggested both Dr. Amaro and Dr. Gabbay. “Physicians will need to develop a sense for tirzepatide: Do patients tolerate it and are they happy using it?” Dr. Amaro said.
Tirzepatide versus insulin, or on top of insulin
The other four trials in patients with type 2 diabetes reported by Lilly in releases included SURPASS-1, which randomized 478 patients to treatment with tirzepatide or placebo as monotherapy; SURPASS-3, which randomized 1,444 patients to tirzepatide or insulin degludec (Tresiba) on top of background treatment with metformin; SURPASS-4, which randomized 2,002 patients with high cardiovascular disease risk to treatment with tirzepatide or insulin glargine (Lantus) on top of background treatment with one to three different oral drugs; and SURPASS-5, which randomized 475 patients to treatment with tirzepatide or placebo on top of background treatment with insulin glargine and optional addition of metformin. Altogether, the five trials randomized nearly 6,300 patients.
The studies that compared tirzepatide against two different types of insulin, and the third that tested tirzepatide on top of insulin glargine, are especially notable. “It’s good to see that the combination [of tirzepatide and insulin glargine] works without causing major adverse events,” said Dr. Amaro.
“These are fair and helpful comparisons. I applaud Lilly for doing the right kind of comparisons,” said Dr. Gabbay.
In total, the five studies “provide evidence that tirzepatide will be effective at all stages of type 2 diabetes and can safely be used in combination with other glucose-lowering agents, including insulin,” said Dr. Lingvay. The studies with active comparator agents “allow us to compare tirzepatide’s efficacy against established therapies.”
The SURPASS trials were sponsored by Lilly, which is developing tirzepatide. Dr. Gabbay had no relevant disclosures. Dr. Lingvay has received research funds, consulting and advisory fees, or other support from Lilly as well as from several other companies including Novo Nordisk, which markets semaglutide (Ozempic) and insulin degludec (Tresiba), and Sanofi, which markets insulin glargine (Lantus). Dr. Amaro has received research funding from Lilly and from Fractyl, and has been a consultant to and received research funding from Novo Nordisk.
The investigational, novel, injected once-weekly “twincretin” tirzepatide met its primary efficacy endpoint of significantly cutting hemoglobin A1c as well as its secondary weight-loss endpoint in patients with type 2 diabetes when compared with control patients in top-line results from each of five discrete pivotal trials.
The company developing tirzepatide, Lilly, announced these results in a series of four press releases issued during December 2020–May 2021. Scientific reports on the outcomes from four of these trials are scheduled during the American Diabetes Association’s Scientific Sessions being held virtually in late June 2021, with results from the fifth on track for a report during the annual meeting of the European Association for the Study of Diabetes in September 2021.
Tirzepatide is a “twincretin” because it combines in a single molecule two different gut-hormone activities. It works as both a glucagonlike peptide–1 receptor agonist (GLP-1 RA) and as an agent that mimics the glucose-dependent insulinotropic polypeptide (GIP).
While diabetologists qualified their comments on these results because of the limited scope and format of the five reports to date, they also expressed enthusiasm over what the press releases said.
Results give hope
“It’s quite exciting, but of course we would like to go by the data that’s presented” at upcoming meetings, commented Robert A. Gabbay, MD, PhD, chief science and medical officer of the American Diabetes Association in Arlington, Va. “The idea of GLP-1 and GIP activities working together has been out there for a while, but without any therapeutic options that leverage this,” he said in an interview.
“The preliminary results give us hope that tirzepatide will be a very effective glucose-lowering agent, perhaps the most effective among all options currently available, including insulin,” commented Ildiko Lingvay, MD, a diabetologist and professor at the University of Texas Southwestern Medical Center, Dallas. “Tirzepatide might have the added benefit of clinically meaningful weight loss,” and “the adverse event profile seems to be in line with what we are accustomed to with the GLP-1 RA class. I look forward to seeing the full results. Tirzepatide promises to be a great addition for type 2 diabetes,” Dr. Lingvay said in an interview.
A rare head-to-head against semaglutide
The five phase 3, randomized controlled trials described by Lilly in its four press releases all belong to the SURPASS series of studies for this agent. Perhaps the most intriguing of the five were results from SURPASS-2, announced in a release on March 4. This trial randomized 1,879 patients from the United States or any of seven other countries to 40 weeks of open-label treatment with one of three different dosages of tirzepatide administered by injection once weekly, or to the control group that received a weekly 1-mg injection of semaglutide (Ozempic), the highest dosage approved for controlling glycemia in patients with type 2 diabetes at the time the study launched.
In SURPASS-2 all three tested dosages of tirzepatide led to a significantly larger reduction, from baseline in A1c, compared with semaglutide, after 40 weeks, according to the Lilly release. Each of the three tirzepatide dosages also led to significantly greater weight loss from baseline, compared with semaglutide, and significantly greater percentages of patients who achieved an A1c of less than 7%, compared with semaglutide.
As an example, the highest tested tirzepatide dosage of 15 mg weekly led to an average A1c reduction from baseline of 2.46% and an average weight loss from baseline of 12.4 kg; 92% of patients achieved an A1c of less than 7%, and 51% had their A1c fall below 5.7% which indicates completely normalization of glycemic control. By comparison, the patients randomized to treatment with semaglutide had an average 1.86% reduction in their A1c level from baseline and a 6.2-kg average cut in body weight from baseline; 81% achieved an A1c of less than 7%, and 20% reached an A1c of less than 5.7%.
There are caveats
While these findings are notable as a rare example of an industry-sponsored head-to-head comparison of two new agents, the study comes with a few important asterisks.
First, it was open label, a curious limitation given that both agents are delivered by the same delivery method and schedule. “I cannot conclude based on this study that tirzepatide is superior because it was open label,” commented Anastassia Amaro, MD, medical director of Penn Metabolic Medicine at the University of Pennsylvania, Philadelphia.
“The gold standard is the double-blind study. An open-label design is a limitation,” agreed Dr. Gabbay.
A second caveat is that the Food and Drug Administration recently approved a higher dosage of semaglutide (2.4 g once/week) for treating overweight or obesity in patients with type 2 diabetes and in those without diabetes but a different weight-related condition such as hypertension of hypercholesterolemia. This means that the tested comparator dosage of 1 mg/week is no longer the maximum that most patients treated with semaglutide for glycemic control can receive.
“The inevitable question” about this comparison study is “what about a higher semaglutide dose,” and how might tirzepatide perform relative to that, said Dr. Gabbay. The recently approved higher dosage of semaglutide “adds an interesting wrinkle.”
Lilly has launched a series of studies testing tirzepatide as a treatment for overweight or obesity in people without diabetes, but the results are not expected until sometime in 2022 or 2023.
And there’s a third caveat: Semaglutide has already shown its value for cardiovascular risk reduction in patients with type 2 diabetes in the SUSTAIN 6 trial with nearly 3,300 randomized patients followed for 2 years and reported in 2016. The cardiovascular outcomes trial for tirzepatide, SURPASS-CVOT with more than 12,000 patients with type 2 diabetes, is underway but its results are not expected until 2024.
Despite these important limitations, a blinded comparison of tirzepatide and higher-dose semaglutide is unlikely, Dr. Amaro predicted. “It’s not worth the expense,” she said in an interview. A more likely scenario will be that, if tirzepatide enters the U.S. market, decisions on whether to treat patients with it or semaglutide will pivot on factors like the cost for treatment to individual patients based on their insurance coverage and tolerability, suggested both Dr. Amaro and Dr. Gabbay. “Physicians will need to develop a sense for tirzepatide: Do patients tolerate it and are they happy using it?” Dr. Amaro said.
Tirzepatide versus insulin, or on top of insulin
The other four trials in patients with type 2 diabetes reported by Lilly in releases included SURPASS-1, which randomized 478 patients to treatment with tirzepatide or placebo as monotherapy; SURPASS-3, which randomized 1,444 patients to tirzepatide or insulin degludec (Tresiba) on top of background treatment with metformin; SURPASS-4, which randomized 2,002 patients with high cardiovascular disease risk to treatment with tirzepatide or insulin glargine (Lantus) on top of background treatment with one to three different oral drugs; and SURPASS-5, which randomized 475 patients to treatment with tirzepatide or placebo on top of background treatment with insulin glargine and optional addition of metformin. Altogether, the five trials randomized nearly 6,300 patients.
The studies that compared tirzepatide against two different types of insulin, and the third that tested tirzepatide on top of insulin glargine, are especially notable. “It’s good to see that the combination [of tirzepatide and insulin glargine] works without causing major adverse events,” said Dr. Amaro.
“These are fair and helpful comparisons. I applaud Lilly for doing the right kind of comparisons,” said Dr. Gabbay.
In total, the five studies “provide evidence that tirzepatide will be effective at all stages of type 2 diabetes and can safely be used in combination with other glucose-lowering agents, including insulin,” said Dr. Lingvay. The studies with active comparator agents “allow us to compare tirzepatide’s efficacy against established therapies.”
The SURPASS trials were sponsored by Lilly, which is developing tirzepatide. Dr. Gabbay had no relevant disclosures. Dr. Lingvay has received research funds, consulting and advisory fees, or other support from Lilly as well as from several other companies including Novo Nordisk, which markets semaglutide (Ozempic) and insulin degludec (Tresiba), and Sanofi, which markets insulin glargine (Lantus). Dr. Amaro has received research funding from Lilly and from Fractyl, and has been a consultant to and received research funding from Novo Nordisk.
Oral Verrucous Plaques in a Patient With Urothelial Cancer
The Diagnosis: Paraneoplastic Acanthosis Nigricans
Histopathologic examination demonstrated verrucous epidermal hyperplasia (Figure, A). Fungal organisms were identified with an Alcian blue and periodic acid-Schiff stain (Figure, B). The organisms demonstrated a vertical orientation in relation to the mucosal surface, which was consistent with candidal organisms.
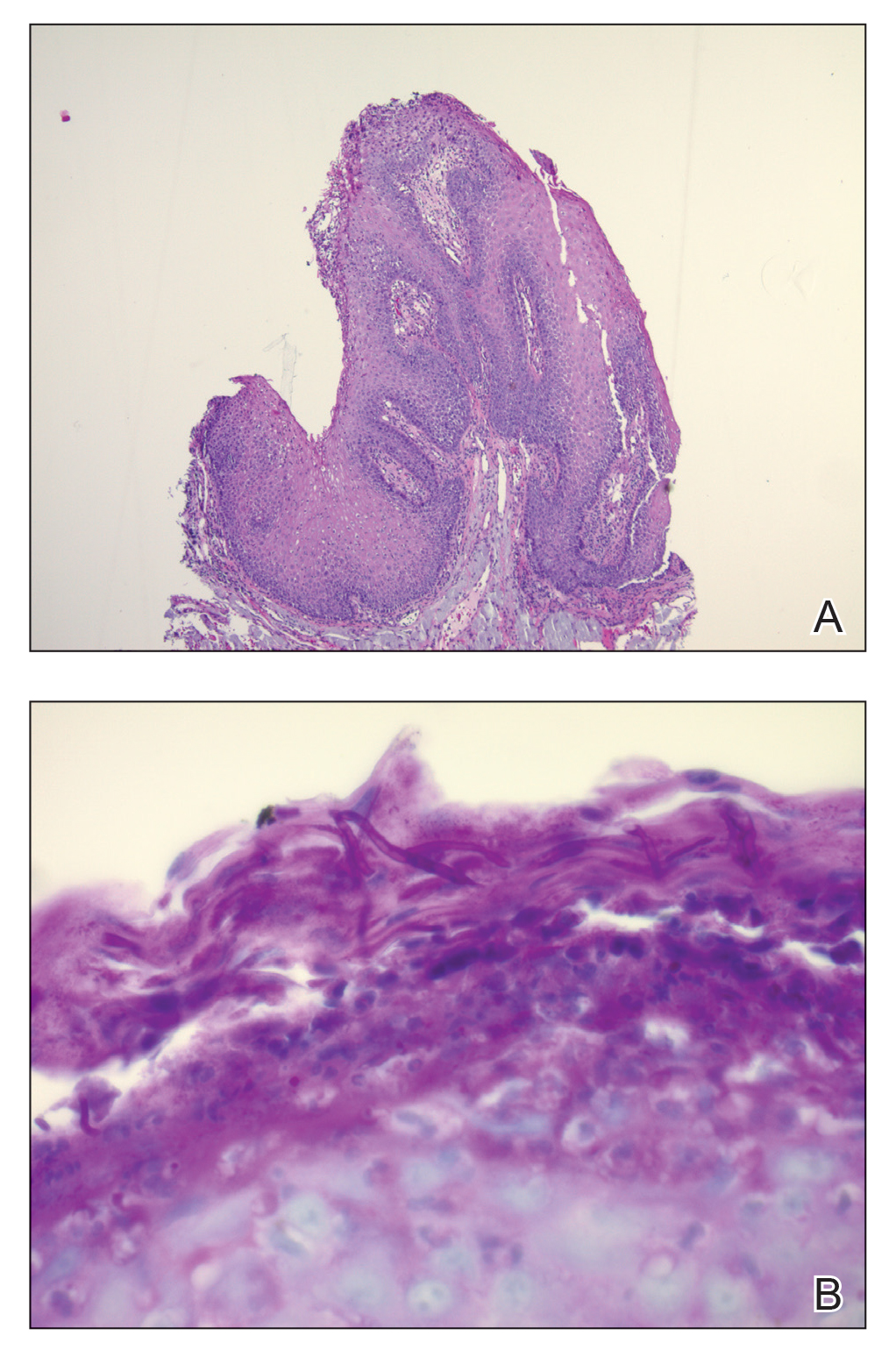
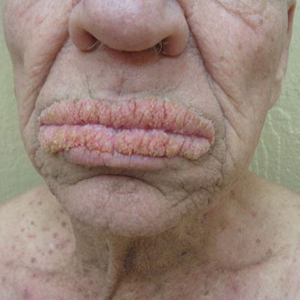
Given the rapid eruption of these plaques, the distribution on the oral and palmar surfaces (tripe palms), and the minimal improvement with both systemic steroids and antifungal treatment, a diagnosis of paraneoplastic acanthosis nigricans with secondary candidal infection was made. Drug-induced cheilitis was considered; however, improvement with discontinuation of the suspected offending drug would have been expected. Although chronic mucocutaneous candidiasis was possible, more prompt improvement upon initiation of systemic antifungal therapy would have been observed. Oral Crohn disease should be included in the differential, but it was unlikely given the lack of granulomas on pathology and absence of history of gastrointestinal tract symptoms. Melkersson-Rosenthal syndrome also was unlikely given the lack of facial nerve palsy as well as the lack of granulomas on pathology. Furthermore, none of these options would be associated with tripe palms, as seen in our patient.
Acanthosis nigricans is a localized skin disorder characterized by hyperpigmented velvety plaques arising in flexural and intertriginous regions. Although most cases (80%) are associated with idiopathic or benign conditions, the link between acanthosis nigricans and an underlying malignancy has been well documented.1-3 Most commonly associated with an underlying intra-abdominal malignancy (often gastric carcinoma), the lesions of paraneoplastic acanthosis nigricans are indistinguishable from their benign counterparts.1,4 When the condition presents abruptly and extensively in a nonobese patient, prompt workup for malignancy should be initiated. Rapid onset and atypical distribution (ie, palmar, perioral, or mucosal) more commonly is associated with a paraneoplastic etiology.5,6
Histopathology for acanthosis nigricans shows hyperkeratosis and epidermal papillomatosis. Horn pseudocyst formation is possible, but usually no hyperpigmentation is observed. The findings typically are indistinguishable from seborrheic keratoses, epidermal nevi, or lesions of confluent and reticulated papillomatosis of Gougerot and Carteaud.2
The underlying pathogenesis of acanthosis nigricans is poorly understood. In the benign subtype, insulin resistance commonly has been described. In the paraneoplastic subtype, it is proposed that the tumor produces a transforming growth factor that mimics epidermal growth factor and leads to keratinocyte proliferation.7,8 Paraneoplastic acanthosis nigricans has the potential to arise at any point of tumor development, further contributing to the diagnostic challenge. Treatment of the skin lesions involves management of the underlying malignancy. Unfortunately, many such malignancies often are at an advanced stage, and subsequent prognosis is poor.2
- Shah A, Jack A, Liu H, et al. Neoplastic/paraneoplastic dermatitis, fasciitis, and panniculitis. Rheum Dis Clin North Am. 2011;37:573-592.
- Chairatchaneeboon M, Kim EJ. Cutaneous paraneoplastic syndromes. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick's Dermatology. 9th ed. McGraw-Hill Education; 2019:2441-2464.
- Lee HC, Ker KJ, Chong WS. Oral malignant acanthosis nigricans and tripe palms associated with renal urothelial carcinoma. JAMA Dermatol. 2015;151:1381-1383.
- Yu Q, Li XL, Ji G, et al. Malignant acanthosis nigricans: an early diagnostic clue for gastric adenocarcinoma. World J Surg Oncol. 2017;15:208.
- Mohrenschlager M, Vocks E, Wessner DB, et al. Tripe palms and malignant acanthosis nigricans: cutaneous signs of imminent metastasis in bladder cancer? J Urol. 2001;165:1629-1630.
- Cohen PR, Grossman ME, Almeida L, et al. Tripe palms and malignancy. J Clin Oncol. 1989;7:669-678.
- Higgins SP, Freemark M, Prose NS. Acanthosis nigricans: a practical approach to evaluation and management. Dermatol Online J. 2008;14:2.
- Torley D, Bellus GA, Munro CS. Genes, growth factors and acanthosis nigricans. Br J Dermatol. 2002;147:1096-1101.
The Diagnosis: Paraneoplastic Acanthosis Nigricans
Histopathologic examination demonstrated verrucous epidermal hyperplasia (Figure, A). Fungal organisms were identified with an Alcian blue and periodic acid-Schiff stain (Figure, B). The organisms demonstrated a vertical orientation in relation to the mucosal surface, which was consistent with candidal organisms.

Given the rapid eruption of these plaques, the distribution on the oral and palmar surfaces (tripe palms), and the minimal improvement with both systemic steroids and antifungal treatment, a diagnosis of paraneoplastic acanthosis nigricans with secondary candidal infection was made. Drug-induced cheilitis was considered; however, improvement with discontinuation of the suspected offending drug would have been expected. Although chronic mucocutaneous candidiasis was possible, more prompt improvement upon initiation of systemic antifungal therapy would have been observed. Oral Crohn disease should be included in the differential, but it was unlikely given the lack of granulomas on pathology and absence of history of gastrointestinal tract symptoms. Melkersson-Rosenthal syndrome also was unlikely given the lack of facial nerve palsy as well as the lack of granulomas on pathology. Furthermore, none of these options would be associated with tripe palms, as seen in our patient.
Acanthosis nigricans is a localized skin disorder characterized by hyperpigmented velvety plaques arising in flexural and intertriginous regions. Although most cases (80%) are associated with idiopathic or benign conditions, the link between acanthosis nigricans and an underlying malignancy has been well documented.1-3 Most commonly associated with an underlying intra-abdominal malignancy (often gastric carcinoma), the lesions of paraneoplastic acanthosis nigricans are indistinguishable from their benign counterparts.1,4 When the condition presents abruptly and extensively in a nonobese patient, prompt workup for malignancy should be initiated. Rapid onset and atypical distribution (ie, palmar, perioral, or mucosal) more commonly is associated with a paraneoplastic etiology.5,6
Histopathology for acanthosis nigricans shows hyperkeratosis and epidermal papillomatosis. Horn pseudocyst formation is possible, but usually no hyperpigmentation is observed. The findings typically are indistinguishable from seborrheic keratoses, epidermal nevi, or lesions of confluent and reticulated papillomatosis of Gougerot and Carteaud.2
The underlying pathogenesis of acanthosis nigricans is poorly understood. In the benign subtype, insulin resistance commonly has been described. In the paraneoplastic subtype, it is proposed that the tumor produces a transforming growth factor that mimics epidermal growth factor and leads to keratinocyte proliferation.7,8 Paraneoplastic acanthosis nigricans has the potential to arise at any point of tumor development, further contributing to the diagnostic challenge. Treatment of the skin lesions involves management of the underlying malignancy. Unfortunately, many such malignancies often are at an advanced stage, and subsequent prognosis is poor.2
The Diagnosis: Paraneoplastic Acanthosis Nigricans
Histopathologic examination demonstrated verrucous epidermal hyperplasia (Figure, A). Fungal organisms were identified with an Alcian blue and periodic acid-Schiff stain (Figure, B). The organisms demonstrated a vertical orientation in relation to the mucosal surface, which was consistent with candidal organisms.

Given the rapid eruption of these plaques, the distribution on the oral and palmar surfaces (tripe palms), and the minimal improvement with both systemic steroids and antifungal treatment, a diagnosis of paraneoplastic acanthosis nigricans with secondary candidal infection was made. Drug-induced cheilitis was considered; however, improvement with discontinuation of the suspected offending drug would have been expected. Although chronic mucocutaneous candidiasis was possible, more prompt improvement upon initiation of systemic antifungal therapy would have been observed. Oral Crohn disease should be included in the differential, but it was unlikely given the lack of granulomas on pathology and absence of history of gastrointestinal tract symptoms. Melkersson-Rosenthal syndrome also was unlikely given the lack of facial nerve palsy as well as the lack of granulomas on pathology. Furthermore, none of these options would be associated with tripe palms, as seen in our patient.
Acanthosis nigricans is a localized skin disorder characterized by hyperpigmented velvety plaques arising in flexural and intertriginous regions. Although most cases (80%) are associated with idiopathic or benign conditions, the link between acanthosis nigricans and an underlying malignancy has been well documented.1-3 Most commonly associated with an underlying intra-abdominal malignancy (often gastric carcinoma), the lesions of paraneoplastic acanthosis nigricans are indistinguishable from their benign counterparts.1,4 When the condition presents abruptly and extensively in a nonobese patient, prompt workup for malignancy should be initiated. Rapid onset and atypical distribution (ie, palmar, perioral, or mucosal) more commonly is associated with a paraneoplastic etiology.5,6
Histopathology for acanthosis nigricans shows hyperkeratosis and epidermal papillomatosis. Horn pseudocyst formation is possible, but usually no hyperpigmentation is observed. The findings typically are indistinguishable from seborrheic keratoses, epidermal nevi, or lesions of confluent and reticulated papillomatosis of Gougerot and Carteaud.2
The underlying pathogenesis of acanthosis nigricans is poorly understood. In the benign subtype, insulin resistance commonly has been described. In the paraneoplastic subtype, it is proposed that the tumor produces a transforming growth factor that mimics epidermal growth factor and leads to keratinocyte proliferation.7,8 Paraneoplastic acanthosis nigricans has the potential to arise at any point of tumor development, further contributing to the diagnostic challenge. Treatment of the skin lesions involves management of the underlying malignancy. Unfortunately, many such malignancies often are at an advanced stage, and subsequent prognosis is poor.2
- Shah A, Jack A, Liu H, et al. Neoplastic/paraneoplastic dermatitis, fasciitis, and panniculitis. Rheum Dis Clin North Am. 2011;37:573-592.
- Chairatchaneeboon M, Kim EJ. Cutaneous paraneoplastic syndromes. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick's Dermatology. 9th ed. McGraw-Hill Education; 2019:2441-2464.
- Lee HC, Ker KJ, Chong WS. Oral malignant acanthosis nigricans and tripe palms associated with renal urothelial carcinoma. JAMA Dermatol. 2015;151:1381-1383.
- Yu Q, Li XL, Ji G, et al. Malignant acanthosis nigricans: an early diagnostic clue for gastric adenocarcinoma. World J Surg Oncol. 2017;15:208.
- Mohrenschlager M, Vocks E, Wessner DB, et al. Tripe palms and malignant acanthosis nigricans: cutaneous signs of imminent metastasis in bladder cancer? J Urol. 2001;165:1629-1630.
- Cohen PR, Grossman ME, Almeida L, et al. Tripe palms and malignancy. J Clin Oncol. 1989;7:669-678.
- Higgins SP, Freemark M, Prose NS. Acanthosis nigricans: a practical approach to evaluation and management. Dermatol Online J. 2008;14:2.
- Torley D, Bellus GA, Munro CS. Genes, growth factors and acanthosis nigricans. Br J Dermatol. 2002;147:1096-1101.
- Shah A, Jack A, Liu H, et al. Neoplastic/paraneoplastic dermatitis, fasciitis, and panniculitis. Rheum Dis Clin North Am. 2011;37:573-592.
- Chairatchaneeboon M, Kim EJ. Cutaneous paraneoplastic syndromes. In: Kang S, Amagai M, Bruckner AL, et al, eds. Fitzpatrick's Dermatology. 9th ed. McGraw-Hill Education; 2019:2441-2464.
- Lee HC, Ker KJ, Chong WS. Oral malignant acanthosis nigricans and tripe palms associated with renal urothelial carcinoma. JAMA Dermatol. 2015;151:1381-1383.
- Yu Q, Li XL, Ji G, et al. Malignant acanthosis nigricans: an early diagnostic clue for gastric adenocarcinoma. World J Surg Oncol. 2017;15:208.
- Mohrenschlager M, Vocks E, Wessner DB, et al. Tripe palms and malignant acanthosis nigricans: cutaneous signs of imminent metastasis in bladder cancer? J Urol. 2001;165:1629-1630.
- Cohen PR, Grossman ME, Almeida L, et al. Tripe palms and malignancy. J Clin Oncol. 1989;7:669-678.
- Higgins SP, Freemark M, Prose NS. Acanthosis nigricans: a practical approach to evaluation and management. Dermatol Online J. 2008;14:2.
- Torley D, Bellus GA, Munro CS. Genes, growth factors and acanthosis nigricans. Br J Dermatol. 2002;147:1096-1101.
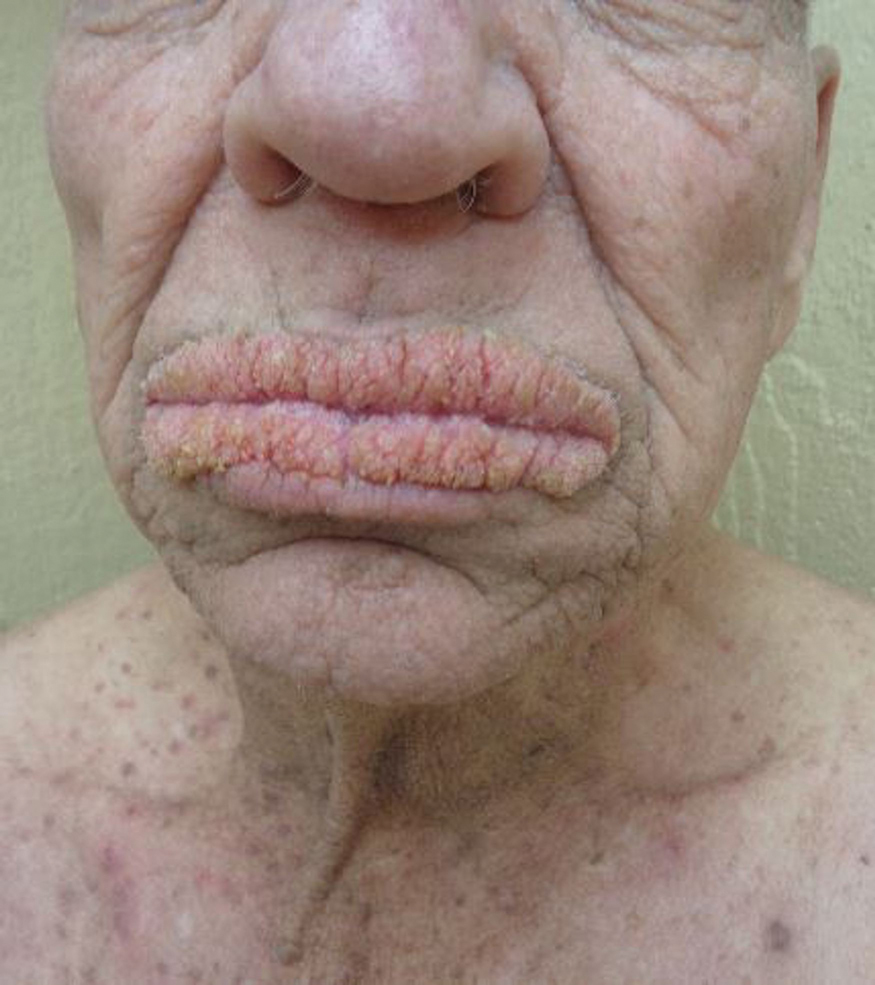
A 75-year-old nonobese man with metastatic urothelial carcinoma presented for evaluation and treatment of swollen lips. The patient stated that his lips began to swell and crack shortly after beginning pembrolizumab approximately 5 months prior. The swelling had progressively worsened, prompting discontinuation of the pembrolizumab by oncology about 2 months prior to presentation to our dermatology clinic. He reported slight improvement after the discontinuation of pembrolizumab, and he had since been started on carboplatin and gemcitabine. He previously was treated with oral corticosteroids without improvement. His oncologist started him on oral fluconazole for treatment of oral thrush on the day of presentation to our clinic. Physical examination revealed diffuse papillomatous and verrucous plaques of the upper and lower lips with involvement of the buccal mucosa. He also had deep fissures and white plaques on the tongue. Velvety hyperpigmented plaques were noted in the axillae, and he had confluent thickening of the palms. A 3-mm punch biopsy from the lower lip was performed. The patient subsequently was evaluated 2 weeks after the initial appointment, and minor improvement in the oral verrucous hyperplasia was noted following antifungal therapy, with resolution of the candidiasis.
Mavrilimumab may aid severe COVID-19 recovery
Inhibiting granulocyte/macrophage–colony stimulating factor (GM-CSF) with mavrilimumab prevented some patients with severe COVID-19 pneumonia and hyperinflammation from needing mechanical ventilation and reduced their risk of dying versus placebo in a phase 2 study.
There was no difference in outcomes between the two doses of mavrilimumab used in the trial (6 mg/kg or 10 mg/kg) and combined data showed a higher percentage of patients achieving the primary endpoint of being alive and free of mechanical ventilation at 29 days, at 87%, versus placebo, at 74%.
The P value was 0.12, “which achieved the prespecified evidentiary standard of 0.2,” according to Lara Pupim, MD, vice president of clinical research and development at Kiniksa Pharmaceuticals in Lexington, Mass.
Importantly, there was a 61% reduction in the risk of dying if patients had received mavrilimumab rather than placebo, she reported at the annual European Congress of Rheumatology. Mortality at day 29 was 21% in the placebo arm but just 8% in the combined mavrilimumab arms (P = .07).
Hendrik Schulze-Koops, MD, called it a “surprising study” and that “the outcome is very spectacular” in his short appraisal of the study during the Clinical Highlights session on the final day of the congress.
Mavrilimumab was “a compound that we would not have thought that would have such an impact on the outcome of COVID-19 infected patients,” Dr. Schulze-Koops of Ludwig Maximilian University of Munich added.
In this small study, “there was a consistent suggestion of a biological effect across key endpoints,” Richard Conway, MBChB, PhD, a consultant rheumatologist at St. James’s Hospital in Dublin, pointed out in an interview.
“Similar to tocilizumab, the benefits with mavrilimumab appear to be in addition to those seen with glucocorticoids, as 96% of patients received dexamethasone,” Dr. Conway observed. Furthermore, nearly one-third received antiviral or remdesivir treatment.
“This study was likely underpowered to assess a clinically meaningful benefit,” he said, adding that “there is insufficient evidence at present to begin using mavrilimumab as an alternative to currently available agents.” That said, “these results are promising for future studies.”
Rationale for GM-CSF inhibition with mavrilimumab in COVID-19 pneumonia
“The cytokine GM-CSF is vital to both lung homeostasis and regulation of inflammation in autoimmunity,” Dr. Pupim explained.
She added that “GM-CSF is implicated in the mechanism of aberrant immune cell infiltration and activation in the lungs, and it may contribute to respiratory failure and death in patients with severe COVID-19 pneumonia and systemic hyperinflammation.”
The efficacy and safety of blocking GM-CSF with mavrilimumab have been shown previously in phase 2 studies in other diseases, Dr. Pupim noted. This includes patients with rheumatoid arthritis and those with giant cell arteritis.
“It was hypothesized that GM-CSF receptor–alpha blockade may reduce infiltration of pathogenic cells into the lung and may suppress inflammation in COVID-19 pneumonia in hyperinflammation,” she explained.
Study details and other outcome results
The study presented by Dr. Pupim was a phase 2/3 double-blind, placebo-controlled trial predominantly conducted in Brazil, the United States, and South Africa, with some participation in Peru and Chile.
Patients were eligible for inclusion if they had had a positive COVID-19 test within 14 days of randomization and had been hospitalized but not ventilated. Evidence of bilateral pneumonia on chest x-ray or CT scan and clinical laboratory evidence indicative of hyperinflammation were also prerequisites for study enrollment.
The ongoing study comprised two cohorts, Dr. Pupim explained: patients who have not been ventilated and those who have recently been ventilated. Dr. Pupim presented the data on the nonventilated cohort, noting that there was a total of 116 patients aged a mean of 57 years.
Patients were randomized to one of three treatment arms: two groups received a single intravenous infusion of mavrilimumab, either 6 mg/kg or 10 mg/kg, and the third group got a placebo.
“Using a time-to-event approach, looking at mechanical ventilation-free survival, mavrilimumab recipients experienced a 65% reduction in the risk of mechanical ventilation or death,” Dr. Pupim said (P = .0175).
“Separation in the Kaplan-Meier curves was evident very early after study drug administration,” she added.
There were trends toward a faster benefit with mavrilimumab than placebo in two other key secondary endpoints: the median time to achieving a two-point clinical improvement (7 vs. 11 days) and the median time to room air (7 vs. 9 days).
Timing of mavrilimumab administration and safety
Study coauthor and chief clinical development officer at Kiniksa, Arian Pano, MD, answered questions on the presentation. When asked about the timing of giving mavrilimumab, he said: “Based on these data it is before they go to ventilation, as soon as you have symptoms of hyperinflammation and a need for oxygen.”
Mavrilimumab is given as a single infusion “and has been well tolerated; virtually no interruptions occurred in this study.”
No serious adverse events related to mavrilimumab were seen, and adverse events, including secondary infections, which are known complications of COVID-19, occurred less frequently in mavrilimumab recipients, compared with placebo.
Dr. Pupim reported that there was a case of tuberculosis in one patient treated with mavrilimumab (10 mg/kg). That case had occurred in an “endemic area for tuberculosis,” and the patient had been screened before entry but only via a sputum sample.
“Prior to these events, the patient received high-dose corticosteroids, a known risk factor for reactivation of TB, and thus the potential additive contribution of mavrilimumab, if any, is uncertain.” Dr. Pupim said.
“Thrombotic events, another known complication of COVID-19, occurred in the placebo arm only,” she added.
Dr. Pano commented that the study has now “seamlessly continued to phase 3. So, basically, we did not stop the study. At the end of phase 2, we just locked the database and collected the data.” Both the 6 mg/kg and 10 mg/kg are being studied, but it’s “very likely [that] 6 mg/kg could be the dose that we may bring forward to the clinic in terms of registration, but that’s at this point in time. We will need to wait for the phase 3 data,” he observed. Those findings will hopefully be available later this year.
Kiniksa funded the study. Dr. Pupim, Dr. Pano, and multiple study coinvestigators are employees of the company.
Dr. Schulze-Koops was not involved in the study and had no specific disclosures. Dr. Conway had no financial disclosures to make in relation to his comments.
Inhibiting granulocyte/macrophage–colony stimulating factor (GM-CSF) with mavrilimumab prevented some patients with severe COVID-19 pneumonia and hyperinflammation from needing mechanical ventilation and reduced their risk of dying versus placebo in a phase 2 study.
There was no difference in outcomes between the two doses of mavrilimumab used in the trial (6 mg/kg or 10 mg/kg) and combined data showed a higher percentage of patients achieving the primary endpoint of being alive and free of mechanical ventilation at 29 days, at 87%, versus placebo, at 74%.
The P value was 0.12, “which achieved the prespecified evidentiary standard of 0.2,” according to Lara Pupim, MD, vice president of clinical research and development at Kiniksa Pharmaceuticals in Lexington, Mass.
Importantly, there was a 61% reduction in the risk of dying if patients had received mavrilimumab rather than placebo, she reported at the annual European Congress of Rheumatology. Mortality at day 29 was 21% in the placebo arm but just 8% in the combined mavrilimumab arms (P = .07).
Hendrik Schulze-Koops, MD, called it a “surprising study” and that “the outcome is very spectacular” in his short appraisal of the study during the Clinical Highlights session on the final day of the congress.
Mavrilimumab was “a compound that we would not have thought that would have such an impact on the outcome of COVID-19 infected patients,” Dr. Schulze-Koops of Ludwig Maximilian University of Munich added.
In this small study, “there was a consistent suggestion of a biological effect across key endpoints,” Richard Conway, MBChB, PhD, a consultant rheumatologist at St. James’s Hospital in Dublin, pointed out in an interview.
“Similar to tocilizumab, the benefits with mavrilimumab appear to be in addition to those seen with glucocorticoids, as 96% of patients received dexamethasone,” Dr. Conway observed. Furthermore, nearly one-third received antiviral or remdesivir treatment.
“This study was likely underpowered to assess a clinically meaningful benefit,” he said, adding that “there is insufficient evidence at present to begin using mavrilimumab as an alternative to currently available agents.” That said, “these results are promising for future studies.”
Rationale for GM-CSF inhibition with mavrilimumab in COVID-19 pneumonia
“The cytokine GM-CSF is vital to both lung homeostasis and regulation of inflammation in autoimmunity,” Dr. Pupim explained.
She added that “GM-CSF is implicated in the mechanism of aberrant immune cell infiltration and activation in the lungs, and it may contribute to respiratory failure and death in patients with severe COVID-19 pneumonia and systemic hyperinflammation.”
The efficacy and safety of blocking GM-CSF with mavrilimumab have been shown previously in phase 2 studies in other diseases, Dr. Pupim noted. This includes patients with rheumatoid arthritis and those with giant cell arteritis.
“It was hypothesized that GM-CSF receptor–alpha blockade may reduce infiltration of pathogenic cells into the lung and may suppress inflammation in COVID-19 pneumonia in hyperinflammation,” she explained.
Study details and other outcome results
The study presented by Dr. Pupim was a phase 2/3 double-blind, placebo-controlled trial predominantly conducted in Brazil, the United States, and South Africa, with some participation in Peru and Chile.
Patients were eligible for inclusion if they had had a positive COVID-19 test within 14 days of randomization and had been hospitalized but not ventilated. Evidence of bilateral pneumonia on chest x-ray or CT scan and clinical laboratory evidence indicative of hyperinflammation were also prerequisites for study enrollment.
The ongoing study comprised two cohorts, Dr. Pupim explained: patients who have not been ventilated and those who have recently been ventilated. Dr. Pupim presented the data on the nonventilated cohort, noting that there was a total of 116 patients aged a mean of 57 years.
Patients were randomized to one of three treatment arms: two groups received a single intravenous infusion of mavrilimumab, either 6 mg/kg or 10 mg/kg, and the third group got a placebo.
“Using a time-to-event approach, looking at mechanical ventilation-free survival, mavrilimumab recipients experienced a 65% reduction in the risk of mechanical ventilation or death,” Dr. Pupim said (P = .0175).
“Separation in the Kaplan-Meier curves was evident very early after study drug administration,” she added.
There were trends toward a faster benefit with mavrilimumab than placebo in two other key secondary endpoints: the median time to achieving a two-point clinical improvement (7 vs. 11 days) and the median time to room air (7 vs. 9 days).
Timing of mavrilimumab administration and safety
Study coauthor and chief clinical development officer at Kiniksa, Arian Pano, MD, answered questions on the presentation. When asked about the timing of giving mavrilimumab, he said: “Based on these data it is before they go to ventilation, as soon as you have symptoms of hyperinflammation and a need for oxygen.”
Mavrilimumab is given as a single infusion “and has been well tolerated; virtually no interruptions occurred in this study.”
No serious adverse events related to mavrilimumab were seen, and adverse events, including secondary infections, which are known complications of COVID-19, occurred less frequently in mavrilimumab recipients, compared with placebo.
Dr. Pupim reported that there was a case of tuberculosis in one patient treated with mavrilimumab (10 mg/kg). That case had occurred in an “endemic area for tuberculosis,” and the patient had been screened before entry but only via a sputum sample.
“Prior to these events, the patient received high-dose corticosteroids, a known risk factor for reactivation of TB, and thus the potential additive contribution of mavrilimumab, if any, is uncertain.” Dr. Pupim said.
“Thrombotic events, another known complication of COVID-19, occurred in the placebo arm only,” she added.
Dr. Pano commented that the study has now “seamlessly continued to phase 3. So, basically, we did not stop the study. At the end of phase 2, we just locked the database and collected the data.” Both the 6 mg/kg and 10 mg/kg are being studied, but it’s “very likely [that] 6 mg/kg could be the dose that we may bring forward to the clinic in terms of registration, but that’s at this point in time. We will need to wait for the phase 3 data,” he observed. Those findings will hopefully be available later this year.
Kiniksa funded the study. Dr. Pupim, Dr. Pano, and multiple study coinvestigators are employees of the company.
Dr. Schulze-Koops was not involved in the study and had no specific disclosures. Dr. Conway had no financial disclosures to make in relation to his comments.
Inhibiting granulocyte/macrophage–colony stimulating factor (GM-CSF) with mavrilimumab prevented some patients with severe COVID-19 pneumonia and hyperinflammation from needing mechanical ventilation and reduced their risk of dying versus placebo in a phase 2 study.
There was no difference in outcomes between the two doses of mavrilimumab used in the trial (6 mg/kg or 10 mg/kg) and combined data showed a higher percentage of patients achieving the primary endpoint of being alive and free of mechanical ventilation at 29 days, at 87%, versus placebo, at 74%.
The P value was 0.12, “which achieved the prespecified evidentiary standard of 0.2,” according to Lara Pupim, MD, vice president of clinical research and development at Kiniksa Pharmaceuticals in Lexington, Mass.
Importantly, there was a 61% reduction in the risk of dying if patients had received mavrilimumab rather than placebo, she reported at the annual European Congress of Rheumatology. Mortality at day 29 was 21% in the placebo arm but just 8% in the combined mavrilimumab arms (P = .07).
Hendrik Schulze-Koops, MD, called it a “surprising study” and that “the outcome is very spectacular” in his short appraisal of the study during the Clinical Highlights session on the final day of the congress.
Mavrilimumab was “a compound that we would not have thought that would have such an impact on the outcome of COVID-19 infected patients,” Dr. Schulze-Koops of Ludwig Maximilian University of Munich added.
In this small study, “there was a consistent suggestion of a biological effect across key endpoints,” Richard Conway, MBChB, PhD, a consultant rheumatologist at St. James’s Hospital in Dublin, pointed out in an interview.
“Similar to tocilizumab, the benefits with mavrilimumab appear to be in addition to those seen with glucocorticoids, as 96% of patients received dexamethasone,” Dr. Conway observed. Furthermore, nearly one-third received antiviral or remdesivir treatment.
“This study was likely underpowered to assess a clinically meaningful benefit,” he said, adding that “there is insufficient evidence at present to begin using mavrilimumab as an alternative to currently available agents.” That said, “these results are promising for future studies.”
Rationale for GM-CSF inhibition with mavrilimumab in COVID-19 pneumonia
“The cytokine GM-CSF is vital to both lung homeostasis and regulation of inflammation in autoimmunity,” Dr. Pupim explained.
She added that “GM-CSF is implicated in the mechanism of aberrant immune cell infiltration and activation in the lungs, and it may contribute to respiratory failure and death in patients with severe COVID-19 pneumonia and systemic hyperinflammation.”
The efficacy and safety of blocking GM-CSF with mavrilimumab have been shown previously in phase 2 studies in other diseases, Dr. Pupim noted. This includes patients with rheumatoid arthritis and those with giant cell arteritis.
“It was hypothesized that GM-CSF receptor–alpha blockade may reduce infiltration of pathogenic cells into the lung and may suppress inflammation in COVID-19 pneumonia in hyperinflammation,” she explained.
Study details and other outcome results
The study presented by Dr. Pupim was a phase 2/3 double-blind, placebo-controlled trial predominantly conducted in Brazil, the United States, and South Africa, with some participation in Peru and Chile.
Patients were eligible for inclusion if they had had a positive COVID-19 test within 14 days of randomization and had been hospitalized but not ventilated. Evidence of bilateral pneumonia on chest x-ray or CT scan and clinical laboratory evidence indicative of hyperinflammation were also prerequisites for study enrollment.
The ongoing study comprised two cohorts, Dr. Pupim explained: patients who have not been ventilated and those who have recently been ventilated. Dr. Pupim presented the data on the nonventilated cohort, noting that there was a total of 116 patients aged a mean of 57 years.
Patients were randomized to one of three treatment arms: two groups received a single intravenous infusion of mavrilimumab, either 6 mg/kg or 10 mg/kg, and the third group got a placebo.
“Using a time-to-event approach, looking at mechanical ventilation-free survival, mavrilimumab recipients experienced a 65% reduction in the risk of mechanical ventilation or death,” Dr. Pupim said (P = .0175).
“Separation in the Kaplan-Meier curves was evident very early after study drug administration,” she added.
There were trends toward a faster benefit with mavrilimumab than placebo in two other key secondary endpoints: the median time to achieving a two-point clinical improvement (7 vs. 11 days) and the median time to room air (7 vs. 9 days).
Timing of mavrilimumab administration and safety
Study coauthor and chief clinical development officer at Kiniksa, Arian Pano, MD, answered questions on the presentation. When asked about the timing of giving mavrilimumab, he said: “Based on these data it is before they go to ventilation, as soon as you have symptoms of hyperinflammation and a need for oxygen.”
Mavrilimumab is given as a single infusion “and has been well tolerated; virtually no interruptions occurred in this study.”
No serious adverse events related to mavrilimumab were seen, and adverse events, including secondary infections, which are known complications of COVID-19, occurred less frequently in mavrilimumab recipients, compared with placebo.
Dr. Pupim reported that there was a case of tuberculosis in one patient treated with mavrilimumab (10 mg/kg). That case had occurred in an “endemic area for tuberculosis,” and the patient had been screened before entry but only via a sputum sample.
“Prior to these events, the patient received high-dose corticosteroids, a known risk factor for reactivation of TB, and thus the potential additive contribution of mavrilimumab, if any, is uncertain.” Dr. Pupim said.
“Thrombotic events, another known complication of COVID-19, occurred in the placebo arm only,” she added.
Dr. Pano commented that the study has now “seamlessly continued to phase 3. So, basically, we did not stop the study. At the end of phase 2, we just locked the database and collected the data.” Both the 6 mg/kg and 10 mg/kg are being studied, but it’s “very likely [that] 6 mg/kg could be the dose that we may bring forward to the clinic in terms of registration, but that’s at this point in time. We will need to wait for the phase 3 data,” he observed. Those findings will hopefully be available later this year.
Kiniksa funded the study. Dr. Pupim, Dr. Pano, and multiple study coinvestigators are employees of the company.
Dr. Schulze-Koops was not involved in the study and had no specific disclosures. Dr. Conway had no financial disclosures to make in relation to his comments.
FROM EULAR 2021 CONGRESS
Red meat intake tied to higher coronary heart disease risk
Increased intake of meat was linked to the risk of coronary heart disease, and substituting plant protein for red or processed meat appeared to reduce that risk, in a study from pooled cohorts totaling more than a million persons.
“We know that red and processed meat intake has been associated with higher risks of fatal coronary heart disease,” said Laila Al-Shaar, PhD, of Penn State University, Hershey. However, very few studies have evaluated substitution of alternative protein sources for red and processed meat in relation to fatal CHD risk, she said.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, Dr. Al-Shaar and colleagues reviewed individual-level data from the Pooling Project of Prospective Studies of Diet and Cancer, which included 16 prospective cohorts totaling 1,364,211 participants. The average age of the participants was 57 years, and 40% were men. Individuals with a history of cancer or cardiovascular disease were excluded. The participants were followed for 7-32 years. Diet was assessed in each cohort using baselines questionnaires, and cases were identified through medical records.
Total red meat included processed meat and unprocessed red meat; animal protein sources included seafood, poultry, eggs, and low- and high-fat dairy products; and plant protein sources included nuts and beans.
The researchers identified 51,176 fatal CHD cases during the study period. After controlling for dietary and nondietary factors, they found that an increase of 100 g per day of total red meat intake was associated with a 7% increased risk of fatal coronary heart disease (relative risk, 1.07).
However, substituting 200 calories (kcal) per day from nuts, low- and high-fat dairy products, and poultry for 200 calories per day from total red meat was associated with a 6%-14% lower risk of fatal CHD, Dr. Al-Shaar added at the meeting sponsored by the American Heart Association.
These associations were stronger when substituting the alternative protein sources for processed meat, especially among women; risk was reduced by 17%-24%, on the basis of 14,888 cases.
The researchers also found that substituting 200 calories per day from eggs for 200 calories per day for total red meat and unprocessed red meat was associated with 8% and 14% higher risk of fatal CHD, respectively; but this substitution of eggs for processed meat was not significant (4%).
“When we did the association by gender, the results were even stronger in women,” said Dr. Al-Shaar. However, “these are very preliminary results” that should be interpreted with caution, and more analysis is needed, she said. “We are planning to include other cohorts with other protein sources such as soy protein,” she noted. However, the results provide additional evidence that consumption of red and processed meat contributes to an increased risk of coronary heart disease, and that substituting some red and processed meat with nuts, dairy products, or poultry may reduce this risk, she concluded.
Women especially benefit from red meat reduction
The study is important because of the continuing interest in various sources of dietary protein intake, Linda Van Horn, PhD, RD, of Northwestern University, Chicago, said in an interview.
“The investigators studied associations of substituting other animal and plant protein sources for total red meat, unprocessed red meat, and processed meat in relation to risk of fatal CHD,” she said.
The researchers found that swapping as little as 200 calories per day of total red meat for nuts, low- or high-fat dairy products, or poultry were associated with a 6%-14% reduced risk of fatal CHD, said Dr. Van Horn. “Alternatively, if those 200 calories per day for red meat were substituted with eggs, they saw as much as 14% higher risk of fatal CHD,” she noted.
The message for both consumers and clinicians is that the findings from this large study support recommendations for plant-based and lean animal sources of protein instead of red and processed meat or eggs, as these sources “offer significantly lower risk for CHD mortality,” Dr. Van Horn said. “This may be especially true for women, but the total population is likely to benefit from this approach,” she said.
Additional research is needed, Dr. Van Horn emphasized. “Prospective lifetime data, starting in utero and over the life course, are needed to better establish recommended dietary patterns at every age and among all ethnicities and diverse socioeconomic groups,” she said.
Dr. Al-Shaar had no financial conflicts to disclose. Dr. Van Horn had no financial conflicts to disclose.
Increased intake of meat was linked to the risk of coronary heart disease, and substituting plant protein for red or processed meat appeared to reduce that risk, in a study from pooled cohorts totaling more than a million persons.
“We know that red and processed meat intake has been associated with higher risks of fatal coronary heart disease,” said Laila Al-Shaar, PhD, of Penn State University, Hershey. However, very few studies have evaluated substitution of alternative protein sources for red and processed meat in relation to fatal CHD risk, she said.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, Dr. Al-Shaar and colleagues reviewed individual-level data from the Pooling Project of Prospective Studies of Diet and Cancer, which included 16 prospective cohorts totaling 1,364,211 participants. The average age of the participants was 57 years, and 40% were men. Individuals with a history of cancer or cardiovascular disease were excluded. The participants were followed for 7-32 years. Diet was assessed in each cohort using baselines questionnaires, and cases were identified through medical records.
Total red meat included processed meat and unprocessed red meat; animal protein sources included seafood, poultry, eggs, and low- and high-fat dairy products; and plant protein sources included nuts and beans.
The researchers identified 51,176 fatal CHD cases during the study period. After controlling for dietary and nondietary factors, they found that an increase of 100 g per day of total red meat intake was associated with a 7% increased risk of fatal coronary heart disease (relative risk, 1.07).
However, substituting 200 calories (kcal) per day from nuts, low- and high-fat dairy products, and poultry for 200 calories per day from total red meat was associated with a 6%-14% lower risk of fatal CHD, Dr. Al-Shaar added at the meeting sponsored by the American Heart Association.
These associations were stronger when substituting the alternative protein sources for processed meat, especially among women; risk was reduced by 17%-24%, on the basis of 14,888 cases.
The researchers also found that substituting 200 calories per day from eggs for 200 calories per day for total red meat and unprocessed red meat was associated with 8% and 14% higher risk of fatal CHD, respectively; but this substitution of eggs for processed meat was not significant (4%).
“When we did the association by gender, the results were even stronger in women,” said Dr. Al-Shaar. However, “these are very preliminary results” that should be interpreted with caution, and more analysis is needed, she said. “We are planning to include other cohorts with other protein sources such as soy protein,” she noted. However, the results provide additional evidence that consumption of red and processed meat contributes to an increased risk of coronary heart disease, and that substituting some red and processed meat with nuts, dairy products, or poultry may reduce this risk, she concluded.
Women especially benefit from red meat reduction
The study is important because of the continuing interest in various sources of dietary protein intake, Linda Van Horn, PhD, RD, of Northwestern University, Chicago, said in an interview.
“The investigators studied associations of substituting other animal and plant protein sources for total red meat, unprocessed red meat, and processed meat in relation to risk of fatal CHD,” she said.
The researchers found that swapping as little as 200 calories per day of total red meat for nuts, low- or high-fat dairy products, or poultry were associated with a 6%-14% reduced risk of fatal CHD, said Dr. Van Horn. “Alternatively, if those 200 calories per day for red meat were substituted with eggs, they saw as much as 14% higher risk of fatal CHD,” she noted.
The message for both consumers and clinicians is that the findings from this large study support recommendations for plant-based and lean animal sources of protein instead of red and processed meat or eggs, as these sources “offer significantly lower risk for CHD mortality,” Dr. Van Horn said. “This may be especially true for women, but the total population is likely to benefit from this approach,” she said.
Additional research is needed, Dr. Van Horn emphasized. “Prospective lifetime data, starting in utero and over the life course, are needed to better establish recommended dietary patterns at every age and among all ethnicities and diverse socioeconomic groups,” she said.
Dr. Al-Shaar had no financial conflicts to disclose. Dr. Van Horn had no financial conflicts to disclose.
Increased intake of meat was linked to the risk of coronary heart disease, and substituting plant protein for red or processed meat appeared to reduce that risk, in a study from pooled cohorts totaling more than a million persons.
“We know that red and processed meat intake has been associated with higher risks of fatal coronary heart disease,” said Laila Al-Shaar, PhD, of Penn State University, Hershey. However, very few studies have evaluated substitution of alternative protein sources for red and processed meat in relation to fatal CHD risk, she said.
In a study presented at the Epidemiology and Prevention/Lifestyle and Cardiometabolic Health meeting, Dr. Al-Shaar and colleagues reviewed individual-level data from the Pooling Project of Prospective Studies of Diet and Cancer, which included 16 prospective cohorts totaling 1,364,211 participants. The average age of the participants was 57 years, and 40% were men. Individuals with a history of cancer or cardiovascular disease were excluded. The participants were followed for 7-32 years. Diet was assessed in each cohort using baselines questionnaires, and cases were identified through medical records.
Total red meat included processed meat and unprocessed red meat; animal protein sources included seafood, poultry, eggs, and low- and high-fat dairy products; and plant protein sources included nuts and beans.
The researchers identified 51,176 fatal CHD cases during the study period. After controlling for dietary and nondietary factors, they found that an increase of 100 g per day of total red meat intake was associated with a 7% increased risk of fatal coronary heart disease (relative risk, 1.07).
However, substituting 200 calories (kcal) per day from nuts, low- and high-fat dairy products, and poultry for 200 calories per day from total red meat was associated with a 6%-14% lower risk of fatal CHD, Dr. Al-Shaar added at the meeting sponsored by the American Heart Association.
These associations were stronger when substituting the alternative protein sources for processed meat, especially among women; risk was reduced by 17%-24%, on the basis of 14,888 cases.
The researchers also found that substituting 200 calories per day from eggs for 200 calories per day for total red meat and unprocessed red meat was associated with 8% and 14% higher risk of fatal CHD, respectively; but this substitution of eggs for processed meat was not significant (4%).
“When we did the association by gender, the results were even stronger in women,” said Dr. Al-Shaar. However, “these are very preliminary results” that should be interpreted with caution, and more analysis is needed, she said. “We are planning to include other cohorts with other protein sources such as soy protein,” she noted. However, the results provide additional evidence that consumption of red and processed meat contributes to an increased risk of coronary heart disease, and that substituting some red and processed meat with nuts, dairy products, or poultry may reduce this risk, she concluded.
Women especially benefit from red meat reduction
The study is important because of the continuing interest in various sources of dietary protein intake, Linda Van Horn, PhD, RD, of Northwestern University, Chicago, said in an interview.
“The investigators studied associations of substituting other animal and plant protein sources for total red meat, unprocessed red meat, and processed meat in relation to risk of fatal CHD,” she said.
The researchers found that swapping as little as 200 calories per day of total red meat for nuts, low- or high-fat dairy products, or poultry were associated with a 6%-14% reduced risk of fatal CHD, said Dr. Van Horn. “Alternatively, if those 200 calories per day for red meat were substituted with eggs, they saw as much as 14% higher risk of fatal CHD,” she noted.
The message for both consumers and clinicians is that the findings from this large study support recommendations for plant-based and lean animal sources of protein instead of red and processed meat or eggs, as these sources “offer significantly lower risk for CHD mortality,” Dr. Van Horn said. “This may be especially true for women, but the total population is likely to benefit from this approach,” she said.
Additional research is needed, Dr. Van Horn emphasized. “Prospective lifetime data, starting in utero and over the life course, are needed to better establish recommended dietary patterns at every age and among all ethnicities and diverse socioeconomic groups,” she said.
Dr. Al-Shaar had no financial conflicts to disclose. Dr. Van Horn had no financial conflicts to disclose.
FROM EPI/LIFESTYLE 2021
Obesity amplifies harmful effects of alcohol on the liver
Being overweight or having obesity significantly increases the risk for liver disease and the likelihood of dying from it compared with being of normal weight, regardless of level of alcohol consumption, new research shows.
"People in the overweight or obese range who drank were found to be at greater risk of liver diseases compared with participants within a healthy weight range who consumed alcohol at the same level," senior author Emmanuel Stamatakis, PhD, of the Charles Perkins Centre and the Faculty of Medicine and Health, Sydney, said in a press statement.
"Even for people who drank within alcohol guidelines, participants classified as obese were at over 50% greater risk of liver disease," he said.
"Obesity is an independent risk factor for steatosis, acute alcoholic hepatitis, and cirrhosis in alcoholic liver disease (ALD), which may increase the risk of mortality in ALD patients," the study's first author, Elif Inan-Eroglu, PhD, a postdoctoral research fellow at the Charles Perkins Centre, said in an interview.
Further prospective studies are needed to better understand the underlying mechanisms behind the association between alcohol consumption and liver disease across different adiposity levels, the authors say.
Meanwhile, the take-home message from the findings should be that "clinicians should consider the presence of overweight and obesity when they discuss defining safe alcohol levels for their patients, keeping in mind that there is no 'safe' level of alcohol," Dr. Inan-Eroglu said.
"Alcohol drinking guidelines need to acknowledge that two-thirds of the adult population are overweight or obese and consider specific recommendations for this majority population group," he said.
First and largest study of its kind
Obesity, well-known to be an independent risk factor for nonalcoholic fatty liver disease (NAFLD), is also known to worsen outcomes in ALD. And likewise, alcohol consumption, the cause of ALD, can promote obesity and therefore increase the risk of NAFLD.
Dr. Stamatakis and colleagues sought to evaluate the roles of the combined factors in terms of incidence and mortality in both ALD and NAFLD.
For the study, published online May 31 in the European Journal of Clinical Nutrition, they evaluated data from 465,437 participants in the U.K. Biobank. The study is said to be the first and largest of its kind.
In the cohort, a total of 1,090 liver disease deaths were recorded, including 230 deaths from ALD and 192 from NAFLD over an average follow-up of 10.5 years.
After a multivariate adjustment, the overall risk of ALD, NAFLD, and liver disease incidence and mortality were significantly higher in participants who were overweight or had obesity, compared with those of normal weight, at all levels of alcohol consumption.
For instance, among those with alcohol use exceeding guidelines, the risk of ALD was significantly increased in normal weight individuals versus never-drinkers (hazard ratio [HR], 5.38), and the risk was even higher among those who were also overweight or had obesity (HR, 8.58).
In terms of the risk of death related to ALD, among those reporting alcohol consumption above guidelines, the risk was nearly double among those who were overweight or had obesity (HR, 10.29) versus those with normal weight (HR, 5.84), when each group was compared to those drinking within guidelines.
Regarding NAFLD, consistent with evidence that low to moderate alcohol consumption is, in fact, linked to a reduced risk, those in the study who reported alcohol consumption within guidelines and normal weight did show a reduced risk of NAFLD compared with an index group of never-drinkers (HR, 0.85).
However, being overweight or having obesity increased the risk of NAFLD in those participants (HR, 1.51).
Furthermore, even those reporting alcohol consumption above guidelines who were of normal weight had a reduced risk of NAFLD compared with never drinkers of normal weight (HR, 0.89).
Regarding the risk of liver disease among those reporting alcohol consumption above guidelines compared with never-drinkers, the risk was again lower among those of normal weight versus those who were overweight or had obesity (HR, 0.95 vs. 1.52), as were the risks of mortality (HR, 1.24 vs. 2.20).
Overall, "we found evidence that being overweight/[having obesity] amplified the harmful effect of alcohol on the liver disease incidence and mortality," the authors conclude.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
Being overweight or having obesity significantly increases the risk for liver disease and the likelihood of dying from it compared with being of normal weight, regardless of level of alcohol consumption, new research shows.
"People in the overweight or obese range who drank were found to be at greater risk of liver diseases compared with participants within a healthy weight range who consumed alcohol at the same level," senior author Emmanuel Stamatakis, PhD, of the Charles Perkins Centre and the Faculty of Medicine and Health, Sydney, said in a press statement.
"Even for people who drank within alcohol guidelines, participants classified as obese were at over 50% greater risk of liver disease," he said.
"Obesity is an independent risk factor for steatosis, acute alcoholic hepatitis, and cirrhosis in alcoholic liver disease (ALD), which may increase the risk of mortality in ALD patients," the study's first author, Elif Inan-Eroglu, PhD, a postdoctoral research fellow at the Charles Perkins Centre, said in an interview.
Further prospective studies are needed to better understand the underlying mechanisms behind the association between alcohol consumption and liver disease across different adiposity levels, the authors say.
Meanwhile, the take-home message from the findings should be that "clinicians should consider the presence of overweight and obesity when they discuss defining safe alcohol levels for their patients, keeping in mind that there is no 'safe' level of alcohol," Dr. Inan-Eroglu said.
"Alcohol drinking guidelines need to acknowledge that two-thirds of the adult population are overweight or obese and consider specific recommendations for this majority population group," he said.
First and largest study of its kind
Obesity, well-known to be an independent risk factor for nonalcoholic fatty liver disease (NAFLD), is also known to worsen outcomes in ALD. And likewise, alcohol consumption, the cause of ALD, can promote obesity and therefore increase the risk of NAFLD.
Dr. Stamatakis and colleagues sought to evaluate the roles of the combined factors in terms of incidence and mortality in both ALD and NAFLD.
For the study, published online May 31 in the European Journal of Clinical Nutrition, they evaluated data from 465,437 participants in the U.K. Biobank. The study is said to be the first and largest of its kind.
In the cohort, a total of 1,090 liver disease deaths were recorded, including 230 deaths from ALD and 192 from NAFLD over an average follow-up of 10.5 years.
After a multivariate adjustment, the overall risk of ALD, NAFLD, and liver disease incidence and mortality were significantly higher in participants who were overweight or had obesity, compared with those of normal weight, at all levels of alcohol consumption.
For instance, among those with alcohol use exceeding guidelines, the risk of ALD was significantly increased in normal weight individuals versus never-drinkers (hazard ratio [HR], 5.38), and the risk was even higher among those who were also overweight or had obesity (HR, 8.58).
In terms of the risk of death related to ALD, among those reporting alcohol consumption above guidelines, the risk was nearly double among those who were overweight or had obesity (HR, 10.29) versus those with normal weight (HR, 5.84), when each group was compared to those drinking within guidelines.
Regarding NAFLD, consistent with evidence that low to moderate alcohol consumption is, in fact, linked to a reduced risk, those in the study who reported alcohol consumption within guidelines and normal weight did show a reduced risk of NAFLD compared with an index group of never-drinkers (HR, 0.85).
However, being overweight or having obesity increased the risk of NAFLD in those participants (HR, 1.51).
Furthermore, even those reporting alcohol consumption above guidelines who were of normal weight had a reduced risk of NAFLD compared with never drinkers of normal weight (HR, 0.89).
Regarding the risk of liver disease among those reporting alcohol consumption above guidelines compared with never-drinkers, the risk was again lower among those of normal weight versus those who were overweight or had obesity (HR, 0.95 vs. 1.52), as were the risks of mortality (HR, 1.24 vs. 2.20).
Overall, "we found evidence that being overweight/[having obesity] amplified the harmful effect of alcohol on the liver disease incidence and mortality," the authors conclude.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
Being overweight or having obesity significantly increases the risk for liver disease and the likelihood of dying from it compared with being of normal weight, regardless of level of alcohol consumption, new research shows.
"People in the overweight or obese range who drank were found to be at greater risk of liver diseases compared with participants within a healthy weight range who consumed alcohol at the same level," senior author Emmanuel Stamatakis, PhD, of the Charles Perkins Centre and the Faculty of Medicine and Health, Sydney, said in a press statement.
"Even for people who drank within alcohol guidelines, participants classified as obese were at over 50% greater risk of liver disease," he said.
"Obesity is an independent risk factor for steatosis, acute alcoholic hepatitis, and cirrhosis in alcoholic liver disease (ALD), which may increase the risk of mortality in ALD patients," the study's first author, Elif Inan-Eroglu, PhD, a postdoctoral research fellow at the Charles Perkins Centre, said in an interview.
Further prospective studies are needed to better understand the underlying mechanisms behind the association between alcohol consumption and liver disease across different adiposity levels, the authors say.
Meanwhile, the take-home message from the findings should be that "clinicians should consider the presence of overweight and obesity when they discuss defining safe alcohol levels for their patients, keeping in mind that there is no 'safe' level of alcohol," Dr. Inan-Eroglu said.
"Alcohol drinking guidelines need to acknowledge that two-thirds of the adult population are overweight or obese and consider specific recommendations for this majority population group," he said.
First and largest study of its kind
Obesity, well-known to be an independent risk factor for nonalcoholic fatty liver disease (NAFLD), is also known to worsen outcomes in ALD. And likewise, alcohol consumption, the cause of ALD, can promote obesity and therefore increase the risk of NAFLD.
Dr. Stamatakis and colleagues sought to evaluate the roles of the combined factors in terms of incidence and mortality in both ALD and NAFLD.
For the study, published online May 31 in the European Journal of Clinical Nutrition, they evaluated data from 465,437 participants in the U.K. Biobank. The study is said to be the first and largest of its kind.
In the cohort, a total of 1,090 liver disease deaths were recorded, including 230 deaths from ALD and 192 from NAFLD over an average follow-up of 10.5 years.
After a multivariate adjustment, the overall risk of ALD, NAFLD, and liver disease incidence and mortality were significantly higher in participants who were overweight or had obesity, compared with those of normal weight, at all levels of alcohol consumption.
For instance, among those with alcohol use exceeding guidelines, the risk of ALD was significantly increased in normal weight individuals versus never-drinkers (hazard ratio [HR], 5.38), and the risk was even higher among those who were also overweight or had obesity (HR, 8.58).
In terms of the risk of death related to ALD, among those reporting alcohol consumption above guidelines, the risk was nearly double among those who were overweight or had obesity (HR, 10.29) versus those with normal weight (HR, 5.84), when each group was compared to those drinking within guidelines.
Regarding NAFLD, consistent with evidence that low to moderate alcohol consumption is, in fact, linked to a reduced risk, those in the study who reported alcohol consumption within guidelines and normal weight did show a reduced risk of NAFLD compared with an index group of never-drinkers (HR, 0.85).
However, being overweight or having obesity increased the risk of NAFLD in those participants (HR, 1.51).
Furthermore, even those reporting alcohol consumption above guidelines who were of normal weight had a reduced risk of NAFLD compared with never drinkers of normal weight (HR, 0.89).
Regarding the risk of liver disease among those reporting alcohol consumption above guidelines compared with never-drinkers, the risk was again lower among those of normal weight versus those who were overweight or had obesity (HR, 0.95 vs. 1.52), as were the risks of mortality (HR, 1.24 vs. 2.20).
Overall, "we found evidence that being overweight/[having obesity] amplified the harmful effect of alcohol on the liver disease incidence and mortality," the authors conclude.
The authors have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com
A large proportion of migraine patients are not offered preventive treatment
, new research suggests. Investigators found that among patients with migraine who are eligible for preventive therapy, more than a third were not offered this option. In addition, fewer than 10% were currently taking preventive medication, and an additional 10% had discontinued preventive therapy.
“We confirmed that as of 2012 to 2013 – the years these data were collected from a large, comprehensive survey – gaps in care remained,” said study investigator Stephanie J. Nahas, MD, director of the headache medicine fellowship program, Thomas Jefferson University, Philadelphia. “In this preventive-eligible population, 35% reported never even being offered preventive medication.”
Furthermore, only 28% of patients taking preventive medication experienced a reduction in headache frequency to less than 4 days per month, which is a primary goal of treatment, said Dr. Nahas. Disease burden, as measured with scales of disability and affective comorbidities, remained substantial.
The findings were presented at the American Headache Society’s 2021 annual meeting.
Lack of efficacy?
In 2019, the American Headache Society published a position statement recommending that preventive treatment be considered for patients who have migraine and four or more monthly headache days (MHDs), regardless of their level of associated disability. However, previous data suggest few patients who are eligible for preventive treatment receive it. In addition, many who have used preventive medications do not adhere to their regimens because of problems with tolerability, efficacy, or both.
To identify treatment gaps and characterize self-reported use of preventive medications for migraine, the investigators examined data from the Chronic Migraine Epidemiology and Outcomes (CaMEO) study, a web-based survey conducted in a representative U.S. sample from September 2012 through November 2013.
The survey identified and characterized patients who met modified criteria for migraine consistent with those in ICHD-3. The researchers classified respondents who had migraine and four or more MHDs as potentially eligible for migraine preventive treatment.
The investigators assessed the study population’s use of oral preventive medications, migraine-related disability and burden, willingness to take preventive treatment, and reasons for discontinuation.
Assessments included the Migraine Disability Assessment Questionnaire, the Patient Health Questionnaire–9 for depression, the Generalized Anxiety Disorder 7-Item Scale, the Migraine Specific Quality of Life questionnaire, and the Migraine Symptom Severity Scale.
In all, 16,789 respondents met criteria for migraine, and 6,579 (39.2%) reported having at least four MHDs. The median age of this subgroup that was eligible for preventive treatment was 40.3 years, and approximately 79% were women.
Only 9.8% of respondents who were eligible for preventive medications were currently using an oral preventive medication. Among those who had ever tried an oral preventive medication, 53.6% discontinued it. Efficacy for patients who used medications appeared to be inadequate. Among all current users of preventive treatment, 68.4% continued to have at least four MHDs.
The researchers assessed treatment eligibility among patients not taking preventive medication. Among respondents who had never used a preventive treatment, 35.7% were eligible to receive it. Among all users who had discontinued preventive medication, 61.0% were still eligible to receive it.
Attitudes toward injectables
Among respondents who had never used a preventive treatment, 64.3% had zero to three MHDs. The remaining 35.7% had 4-7, 8-14, or 15 or more MHDs. Among current users of preventive treatments, 68.4% had four or more MHDs. Among those who had discontinued preventive treatment, 61.0% had four or more MHDs.
Patients who have never used preventive medication “have substantial management gaps,” said Dr. Nahas. High proportions of these patients have moderate or severe disability (64.7%), depression (43%), and anxiety (39%). The rates of these outcomes are higher in users who discontinued treatment, likely because of confounding by indication, she added.
The prevalence of anxiety was similar between those who currently used, formerly used, or never used preventive medications. However, there were differences between never-users and current or former users with respect to moderate to severe depression (never-users, 43%; current users, 49.4%; discontinued users, 46.5%) and moderate to severe disability (never-users, 64.7%; current users, 80.4%; discontinued users, 78.9%).
In all, 44.6% of those who discontinued preventive therapy reported safety and tolerability problems as reasons for stopping treatment. In addition, 39.7% reported that these medications did not prevent enough headaches. Some patients reported partial or temporary efficacy as a reason for discontinuation. Other reasons were related to health care costs and access and personal preferences. Only 9.2% of patients who discontinued treatment said that their headaches improved enough to stop medication.
The investigators also analyzed respondents’ interest in preventive therapies. Among respondents who had never used preventive therapies, 61.8% of those who were eligible to use them were somewhat or very interested in trying an oral prescription medication for migraine prevention. However, 59.1% of never-users who were eligible for preventive medications were not at all interested, not sure, or needed more information about trying an injectable preventive medication. About 40% were not at all interested in injectables. In general, current users and those who had discontinued medication were more interested in preventive medication, including injectables.
‘Disheartening’ discontinuation rates
There are likely multiple reasons for the low rate of migraine prevention treatment, said Dr. Nahas. Many people with migraine never consult a clinician, owing to factors such as stigma, cost, lack of access, and lack of awareness. In addition, patients with migraine are frequently misdiagnosed, she added.
“Other data suggest that only about a quarter of people with episodic migraine and under 5% of people with chronic migraine consult a clinician, receive an accurate diagnosis, and are prescribed appropriate therapy,” said Dr. Nahas.
When the data in this analysis were gathered, public awareness of migraine was much lower than it is today, and injectable migraine therapies had not gained broad acceptance, she noted. Dr. Nahas added it is possible that attitudes toward injectable preventive medications have changed.
“Would people still prefer daily oral medications? We can’t know for sure until we start asking,” she said. In addition, scientific advances and educational outreach have increased clinicians’ awareness, interest, and skill regarding injectable medications, she said.
“I would certainly hope to see that a much greater proportion of preventive-eligible persons with migraine were at least offered, if not currently taking, preventive medication,” said Dr. Nahas. “But there’s no pleasing everyone, so I think we would still see somewhat disheartening discontinuation rates. The reasons for discontinuation, however, might be less typified by concerns about safety and tolerability.”
Still relevant
Commenting on the study, Mia Tova Minen, MD, chief of headache research and associate professor of neurology and population health at NYU Langone Health, New York, noted that although CaMEO is an older study, its results are still highly relevant.
“Unfortunately, primary care providers are still uncomfortable prescribing migraine preventive medications, and this accounts for the large percentage [of patients] with migraine who, while eligible for migraine preventive therapy, are not offered it,” she said.
Although the public and primary care physicians are now more aware of preventive treatments for migraine, “the number of people offered migraine preventive medication still needs to increase dramatically,” said Dr. Minen.
The American Academy of Neurology’s guidelines for migraine prevention were published in 2012 and are currently being updated. The updated guidelines may include new evidence for candesartan and emerging treatments, such as melatonin and aerobic exercise.
“It is my hope that primary care providers will become more comfortable prescribing migraine preventive medications sooner,” said Dr. Minen.
The current findings suggest a need for additional ways of educating patients with migraine who are eligible for preventive therapies so that they can advocate for themselves, she added. They also suggest the idea of demanding more insurance coverage of behavioral therapies for migraine, because data indicate that these treatments have long-term efficacy and good safety profiles, said Dr. Minen.
An ‘invisible’ disorder
Also commenting on the study, Barbara L. Nye, MD, director of the headache fellowship and codirector of the headache clinic at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said the CaMEO cohort likely is representative of the general population of patients with migraine.
She noted that a significant weakness of the current study is that it examined data collected before the Food and Drug Administration approved monoclonal antibodies and therefore does not reflect patients’ current experience with medications.
“I believe that the attitudes and fears surrounding the use of injectable medication are now likely far less than previously reported, given the positive track record the new generation of once-a-month injectable medications has,” said Dr. Nye.
The findings reinforce the idea that either patients are not talking to their primary care physicians about their headaches and disability or that clinicians are not asking about them, she added. “Both issues are likely linked to the stigma that this disease state has surrounding it. This is an invisible neurological disorder to most,” Dr. Nye said.
The study was sponsored by Allergan before it was acquired by AbbVie. Dr. Nahas has served as a consultant, advisory board member, or speaker for AbbVie/Allergan, Alder/Lundbeck, Amgen/Novartis, Biohaven, Eli Lilly, Impel, Nesos Corp, Supernus, Teva, Theranica, and Zosano. She has not received and will not receive monetary compensation for this research. Dr. Minen has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. Investigators found that among patients with migraine who are eligible for preventive therapy, more than a third were not offered this option. In addition, fewer than 10% were currently taking preventive medication, and an additional 10% had discontinued preventive therapy.
“We confirmed that as of 2012 to 2013 – the years these data were collected from a large, comprehensive survey – gaps in care remained,” said study investigator Stephanie J. Nahas, MD, director of the headache medicine fellowship program, Thomas Jefferson University, Philadelphia. “In this preventive-eligible population, 35% reported never even being offered preventive medication.”
Furthermore, only 28% of patients taking preventive medication experienced a reduction in headache frequency to less than 4 days per month, which is a primary goal of treatment, said Dr. Nahas. Disease burden, as measured with scales of disability and affective comorbidities, remained substantial.
The findings were presented at the American Headache Society’s 2021 annual meeting.
Lack of efficacy?
In 2019, the American Headache Society published a position statement recommending that preventive treatment be considered for patients who have migraine and four or more monthly headache days (MHDs), regardless of their level of associated disability. However, previous data suggest few patients who are eligible for preventive treatment receive it. In addition, many who have used preventive medications do not adhere to their regimens because of problems with tolerability, efficacy, or both.
To identify treatment gaps and characterize self-reported use of preventive medications for migraine, the investigators examined data from the Chronic Migraine Epidemiology and Outcomes (CaMEO) study, a web-based survey conducted in a representative U.S. sample from September 2012 through November 2013.
The survey identified and characterized patients who met modified criteria for migraine consistent with those in ICHD-3. The researchers classified respondents who had migraine and four or more MHDs as potentially eligible for migraine preventive treatment.
The investigators assessed the study population’s use of oral preventive medications, migraine-related disability and burden, willingness to take preventive treatment, and reasons for discontinuation.
Assessments included the Migraine Disability Assessment Questionnaire, the Patient Health Questionnaire–9 for depression, the Generalized Anxiety Disorder 7-Item Scale, the Migraine Specific Quality of Life questionnaire, and the Migraine Symptom Severity Scale.
In all, 16,789 respondents met criteria for migraine, and 6,579 (39.2%) reported having at least four MHDs. The median age of this subgroup that was eligible for preventive treatment was 40.3 years, and approximately 79% were women.
Only 9.8% of respondents who were eligible for preventive medications were currently using an oral preventive medication. Among those who had ever tried an oral preventive medication, 53.6% discontinued it. Efficacy for patients who used medications appeared to be inadequate. Among all current users of preventive treatment, 68.4% continued to have at least four MHDs.
The researchers assessed treatment eligibility among patients not taking preventive medication. Among respondents who had never used a preventive treatment, 35.7% were eligible to receive it. Among all users who had discontinued preventive medication, 61.0% were still eligible to receive it.
Attitudes toward injectables
Among respondents who had never used a preventive treatment, 64.3% had zero to three MHDs. The remaining 35.7% had 4-7, 8-14, or 15 or more MHDs. Among current users of preventive treatments, 68.4% had four or more MHDs. Among those who had discontinued preventive treatment, 61.0% had four or more MHDs.
Patients who have never used preventive medication “have substantial management gaps,” said Dr. Nahas. High proportions of these patients have moderate or severe disability (64.7%), depression (43%), and anxiety (39%). The rates of these outcomes are higher in users who discontinued treatment, likely because of confounding by indication, she added.
The prevalence of anxiety was similar between those who currently used, formerly used, or never used preventive medications. However, there were differences between never-users and current or former users with respect to moderate to severe depression (never-users, 43%; current users, 49.4%; discontinued users, 46.5%) and moderate to severe disability (never-users, 64.7%; current users, 80.4%; discontinued users, 78.9%).
In all, 44.6% of those who discontinued preventive therapy reported safety and tolerability problems as reasons for stopping treatment. In addition, 39.7% reported that these medications did not prevent enough headaches. Some patients reported partial or temporary efficacy as a reason for discontinuation. Other reasons were related to health care costs and access and personal preferences. Only 9.2% of patients who discontinued treatment said that their headaches improved enough to stop medication.
The investigators also analyzed respondents’ interest in preventive therapies. Among respondents who had never used preventive therapies, 61.8% of those who were eligible to use them were somewhat or very interested in trying an oral prescription medication for migraine prevention. However, 59.1% of never-users who were eligible for preventive medications were not at all interested, not sure, or needed more information about trying an injectable preventive medication. About 40% were not at all interested in injectables. In general, current users and those who had discontinued medication were more interested in preventive medication, including injectables.
‘Disheartening’ discontinuation rates
There are likely multiple reasons for the low rate of migraine prevention treatment, said Dr. Nahas. Many people with migraine never consult a clinician, owing to factors such as stigma, cost, lack of access, and lack of awareness. In addition, patients with migraine are frequently misdiagnosed, she added.
“Other data suggest that only about a quarter of people with episodic migraine and under 5% of people with chronic migraine consult a clinician, receive an accurate diagnosis, and are prescribed appropriate therapy,” said Dr. Nahas.
When the data in this analysis were gathered, public awareness of migraine was much lower than it is today, and injectable migraine therapies had not gained broad acceptance, she noted. Dr. Nahas added it is possible that attitudes toward injectable preventive medications have changed.
“Would people still prefer daily oral medications? We can’t know for sure until we start asking,” she said. In addition, scientific advances and educational outreach have increased clinicians’ awareness, interest, and skill regarding injectable medications, she said.
“I would certainly hope to see that a much greater proportion of preventive-eligible persons with migraine were at least offered, if not currently taking, preventive medication,” said Dr. Nahas. “But there’s no pleasing everyone, so I think we would still see somewhat disheartening discontinuation rates. The reasons for discontinuation, however, might be less typified by concerns about safety and tolerability.”
Still relevant
Commenting on the study, Mia Tova Minen, MD, chief of headache research and associate professor of neurology and population health at NYU Langone Health, New York, noted that although CaMEO is an older study, its results are still highly relevant.
“Unfortunately, primary care providers are still uncomfortable prescribing migraine preventive medications, and this accounts for the large percentage [of patients] with migraine who, while eligible for migraine preventive therapy, are not offered it,” she said.
Although the public and primary care physicians are now more aware of preventive treatments for migraine, “the number of people offered migraine preventive medication still needs to increase dramatically,” said Dr. Minen.
The American Academy of Neurology’s guidelines for migraine prevention were published in 2012 and are currently being updated. The updated guidelines may include new evidence for candesartan and emerging treatments, such as melatonin and aerobic exercise.
“It is my hope that primary care providers will become more comfortable prescribing migraine preventive medications sooner,” said Dr. Minen.
The current findings suggest a need for additional ways of educating patients with migraine who are eligible for preventive therapies so that they can advocate for themselves, she added. They also suggest the idea of demanding more insurance coverage of behavioral therapies for migraine, because data indicate that these treatments have long-term efficacy and good safety profiles, said Dr. Minen.
An ‘invisible’ disorder
Also commenting on the study, Barbara L. Nye, MD, director of the headache fellowship and codirector of the headache clinic at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said the CaMEO cohort likely is representative of the general population of patients with migraine.
She noted that a significant weakness of the current study is that it examined data collected before the Food and Drug Administration approved monoclonal antibodies and therefore does not reflect patients’ current experience with medications.
“I believe that the attitudes and fears surrounding the use of injectable medication are now likely far less than previously reported, given the positive track record the new generation of once-a-month injectable medications has,” said Dr. Nye.
The findings reinforce the idea that either patients are not talking to their primary care physicians about their headaches and disability or that clinicians are not asking about them, she added. “Both issues are likely linked to the stigma that this disease state has surrounding it. This is an invisible neurological disorder to most,” Dr. Nye said.
The study was sponsored by Allergan before it was acquired by AbbVie. Dr. Nahas has served as a consultant, advisory board member, or speaker for AbbVie/Allergan, Alder/Lundbeck, Amgen/Novartis, Biohaven, Eli Lilly, Impel, Nesos Corp, Supernus, Teva, Theranica, and Zosano. She has not received and will not receive monetary compensation for this research. Dr. Minen has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, new research suggests. Investigators found that among patients with migraine who are eligible for preventive therapy, more than a third were not offered this option. In addition, fewer than 10% were currently taking preventive medication, and an additional 10% had discontinued preventive therapy.
“We confirmed that as of 2012 to 2013 – the years these data were collected from a large, comprehensive survey – gaps in care remained,” said study investigator Stephanie J. Nahas, MD, director of the headache medicine fellowship program, Thomas Jefferson University, Philadelphia. “In this preventive-eligible population, 35% reported never even being offered preventive medication.”
Furthermore, only 28% of patients taking preventive medication experienced a reduction in headache frequency to less than 4 days per month, which is a primary goal of treatment, said Dr. Nahas. Disease burden, as measured with scales of disability and affective comorbidities, remained substantial.
The findings were presented at the American Headache Society’s 2021 annual meeting.
Lack of efficacy?
In 2019, the American Headache Society published a position statement recommending that preventive treatment be considered for patients who have migraine and four or more monthly headache days (MHDs), regardless of their level of associated disability. However, previous data suggest few patients who are eligible for preventive treatment receive it. In addition, many who have used preventive medications do not adhere to their regimens because of problems with tolerability, efficacy, or both.
To identify treatment gaps and characterize self-reported use of preventive medications for migraine, the investigators examined data from the Chronic Migraine Epidemiology and Outcomes (CaMEO) study, a web-based survey conducted in a representative U.S. sample from September 2012 through November 2013.
The survey identified and characterized patients who met modified criteria for migraine consistent with those in ICHD-3. The researchers classified respondents who had migraine and four or more MHDs as potentially eligible for migraine preventive treatment.
The investigators assessed the study population’s use of oral preventive medications, migraine-related disability and burden, willingness to take preventive treatment, and reasons for discontinuation.
Assessments included the Migraine Disability Assessment Questionnaire, the Patient Health Questionnaire–9 for depression, the Generalized Anxiety Disorder 7-Item Scale, the Migraine Specific Quality of Life questionnaire, and the Migraine Symptom Severity Scale.
In all, 16,789 respondents met criteria for migraine, and 6,579 (39.2%) reported having at least four MHDs. The median age of this subgroup that was eligible for preventive treatment was 40.3 years, and approximately 79% were women.
Only 9.8% of respondents who were eligible for preventive medications were currently using an oral preventive medication. Among those who had ever tried an oral preventive medication, 53.6% discontinued it. Efficacy for patients who used medications appeared to be inadequate. Among all current users of preventive treatment, 68.4% continued to have at least four MHDs.
The researchers assessed treatment eligibility among patients not taking preventive medication. Among respondents who had never used a preventive treatment, 35.7% were eligible to receive it. Among all users who had discontinued preventive medication, 61.0% were still eligible to receive it.
Attitudes toward injectables
Among respondents who had never used a preventive treatment, 64.3% had zero to three MHDs. The remaining 35.7% had 4-7, 8-14, or 15 or more MHDs. Among current users of preventive treatments, 68.4% had four or more MHDs. Among those who had discontinued preventive treatment, 61.0% had four or more MHDs.
Patients who have never used preventive medication “have substantial management gaps,” said Dr. Nahas. High proportions of these patients have moderate or severe disability (64.7%), depression (43%), and anxiety (39%). The rates of these outcomes are higher in users who discontinued treatment, likely because of confounding by indication, she added.
The prevalence of anxiety was similar between those who currently used, formerly used, or never used preventive medications. However, there were differences between never-users and current or former users with respect to moderate to severe depression (never-users, 43%; current users, 49.4%; discontinued users, 46.5%) and moderate to severe disability (never-users, 64.7%; current users, 80.4%; discontinued users, 78.9%).
In all, 44.6% of those who discontinued preventive therapy reported safety and tolerability problems as reasons for stopping treatment. In addition, 39.7% reported that these medications did not prevent enough headaches. Some patients reported partial or temporary efficacy as a reason for discontinuation. Other reasons were related to health care costs and access and personal preferences. Only 9.2% of patients who discontinued treatment said that their headaches improved enough to stop medication.
The investigators also analyzed respondents’ interest in preventive therapies. Among respondents who had never used preventive therapies, 61.8% of those who were eligible to use them were somewhat or very interested in trying an oral prescription medication for migraine prevention. However, 59.1% of never-users who were eligible for preventive medications were not at all interested, not sure, or needed more information about trying an injectable preventive medication. About 40% were not at all interested in injectables. In general, current users and those who had discontinued medication were more interested in preventive medication, including injectables.
‘Disheartening’ discontinuation rates
There are likely multiple reasons for the low rate of migraine prevention treatment, said Dr. Nahas. Many people with migraine never consult a clinician, owing to factors such as stigma, cost, lack of access, and lack of awareness. In addition, patients with migraine are frequently misdiagnosed, she added.
“Other data suggest that only about a quarter of people with episodic migraine and under 5% of people with chronic migraine consult a clinician, receive an accurate diagnosis, and are prescribed appropriate therapy,” said Dr. Nahas.
When the data in this analysis were gathered, public awareness of migraine was much lower than it is today, and injectable migraine therapies had not gained broad acceptance, she noted. Dr. Nahas added it is possible that attitudes toward injectable preventive medications have changed.
“Would people still prefer daily oral medications? We can’t know for sure until we start asking,” she said. In addition, scientific advances and educational outreach have increased clinicians’ awareness, interest, and skill regarding injectable medications, she said.
“I would certainly hope to see that a much greater proportion of preventive-eligible persons with migraine were at least offered, if not currently taking, preventive medication,” said Dr. Nahas. “But there’s no pleasing everyone, so I think we would still see somewhat disheartening discontinuation rates. The reasons for discontinuation, however, might be less typified by concerns about safety and tolerability.”
Still relevant
Commenting on the study, Mia Tova Minen, MD, chief of headache research and associate professor of neurology and population health at NYU Langone Health, New York, noted that although CaMEO is an older study, its results are still highly relevant.
“Unfortunately, primary care providers are still uncomfortable prescribing migraine preventive medications, and this accounts for the large percentage [of patients] with migraine who, while eligible for migraine preventive therapy, are not offered it,” she said.
Although the public and primary care physicians are now more aware of preventive treatments for migraine, “the number of people offered migraine preventive medication still needs to increase dramatically,” said Dr. Minen.
The American Academy of Neurology’s guidelines for migraine prevention were published in 2012 and are currently being updated. The updated guidelines may include new evidence for candesartan and emerging treatments, such as melatonin and aerobic exercise.
“It is my hope that primary care providers will become more comfortable prescribing migraine preventive medications sooner,” said Dr. Minen.
The current findings suggest a need for additional ways of educating patients with migraine who are eligible for preventive therapies so that they can advocate for themselves, she added. They also suggest the idea of demanding more insurance coverage of behavioral therapies for migraine, because data indicate that these treatments have long-term efficacy and good safety profiles, said Dr. Minen.
An ‘invisible’ disorder
Also commenting on the study, Barbara L. Nye, MD, director of the headache fellowship and codirector of the headache clinic at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said the CaMEO cohort likely is representative of the general population of patients with migraine.
She noted that a significant weakness of the current study is that it examined data collected before the Food and Drug Administration approved monoclonal antibodies and therefore does not reflect patients’ current experience with medications.
“I believe that the attitudes and fears surrounding the use of injectable medication are now likely far less than previously reported, given the positive track record the new generation of once-a-month injectable medications has,” said Dr. Nye.
The findings reinforce the idea that either patients are not talking to their primary care physicians about their headaches and disability or that clinicians are not asking about them, she added. “Both issues are likely linked to the stigma that this disease state has surrounding it. This is an invisible neurological disorder to most,” Dr. Nye said.
The study was sponsored by Allergan before it was acquired by AbbVie. Dr. Nahas has served as a consultant, advisory board member, or speaker for AbbVie/Allergan, Alder/Lundbeck, Amgen/Novartis, Biohaven, Eli Lilly, Impel, Nesos Corp, Supernus, Teva, Theranica, and Zosano. She has not received and will not receive monetary compensation for this research. Dr. Minen has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AHS 2021
By the numbers: Children and COVID-19 prevention
Over 6.3 million doses of COVID-19 vaccine have been administered to children aged 12-17 years as of June 7, according to data from the Centers for Disease Control and Prevention.
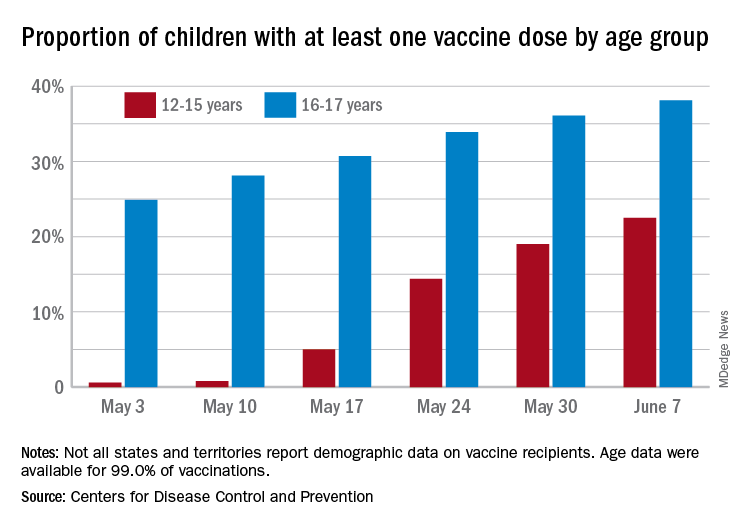
The latest results from the CDC’s COVID Data Tracker show that , with the corresponding figures for vaccine completion coming in at 4.1% and 26.4%. Compared with a week earlier, those numbers are up by 15.4% (one dose) and 486% (completion) for the younger group and by 4.7% and 8.6%, respectively, for the older children.
Children aged 12-15 represented 17.9% of all persons who initiated vaccination in the last 14 days up to June 7, while children aged 16-17 made up 4.8% of vaccine initiation over that period. The 25- to 39-year-olds, at 23.7% of all vaccine initiators, were the only group ahead of those aged 12-15, and the 50- to 64-year-olds were just behind at 17.7%, the CDC data show.
Both groups of children were on the low side, however, when it came to vaccine completion in the last 14 days, with those aged 12-15 at 6.7% of the total and those aged 16-17 years at 4.3%. The only age groups lower than that were ≥75 at 3.5% and <12 at 0.2%, and the highest share of vaccine completion was 26.0% for those aged 25-39, which also happens to be the group with the largest share of the U.S. population (20.5%), the CDC said.
People considered fully vaccinated are those who have received the second dose of a two-dose series or one dose of a single-shot vaccine, but children under age 18 years are eligible only for the Pfizer-BioNTech version, the CDC noted.
Meanwhile, back on the incidence side of the COVID-19 pandemic, the number of new cases in U.S. children for the week ending June 3 was at its lowest point (16,281) since mid-June of 2020, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
Cases among children now total 3.99 million, which represents 14.1% of cases among all ages, a proportion that hasn’t increased since mid-May, which hasn’t happened since the two groups started keeping track in mid-April of 2020 in the 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam that report such data by age.
Less encouraging was the CDC’s report that “COVID-19-associated hospitalization rates among adolescents ages 12-17 years increased during March and April, following declines in January and February 2021.”
Children have been experiencing much lower rates of severe disease than those of adults throughout the pandemic, the CDC pointed out, but “recent increases in COVID-19-associated hospitalization rates and the potential for severe disease in adolescents reinforce the importance of continued prevention strategies, including vaccination and the correct and consistent use of masks in those who are not yet fully vaccinated.”
Over 6.3 million doses of COVID-19 vaccine have been administered to children aged 12-17 years as of June 7, according to data from the Centers for Disease Control and Prevention.

The latest results from the CDC’s COVID Data Tracker show that , with the corresponding figures for vaccine completion coming in at 4.1% and 26.4%. Compared with a week earlier, those numbers are up by 15.4% (one dose) and 486% (completion) for the younger group and by 4.7% and 8.6%, respectively, for the older children.
Children aged 12-15 represented 17.9% of all persons who initiated vaccination in the last 14 days up to June 7, while children aged 16-17 made up 4.8% of vaccine initiation over that period. The 25- to 39-year-olds, at 23.7% of all vaccine initiators, were the only group ahead of those aged 12-15, and the 50- to 64-year-olds were just behind at 17.7%, the CDC data show.
Both groups of children were on the low side, however, when it came to vaccine completion in the last 14 days, with those aged 12-15 at 6.7% of the total and those aged 16-17 years at 4.3%. The only age groups lower than that were ≥75 at 3.5% and <12 at 0.2%, and the highest share of vaccine completion was 26.0% for those aged 25-39, which also happens to be the group with the largest share of the U.S. population (20.5%), the CDC said.
People considered fully vaccinated are those who have received the second dose of a two-dose series or one dose of a single-shot vaccine, but children under age 18 years are eligible only for the Pfizer-BioNTech version, the CDC noted.
Meanwhile, back on the incidence side of the COVID-19 pandemic, the number of new cases in U.S. children for the week ending June 3 was at its lowest point (16,281) since mid-June of 2020, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
Cases among children now total 3.99 million, which represents 14.1% of cases among all ages, a proportion that hasn’t increased since mid-May, which hasn’t happened since the two groups started keeping track in mid-April of 2020 in the 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam that report such data by age.
Less encouraging was the CDC’s report that “COVID-19-associated hospitalization rates among adolescents ages 12-17 years increased during March and April, following declines in January and February 2021.”
Children have been experiencing much lower rates of severe disease than those of adults throughout the pandemic, the CDC pointed out, but “recent increases in COVID-19-associated hospitalization rates and the potential for severe disease in adolescents reinforce the importance of continued prevention strategies, including vaccination and the correct and consistent use of masks in those who are not yet fully vaccinated.”
Over 6.3 million doses of COVID-19 vaccine have been administered to children aged 12-17 years as of June 7, according to data from the Centers for Disease Control and Prevention.

The latest results from the CDC’s COVID Data Tracker show that , with the corresponding figures for vaccine completion coming in at 4.1% and 26.4%. Compared with a week earlier, those numbers are up by 15.4% (one dose) and 486% (completion) for the younger group and by 4.7% and 8.6%, respectively, for the older children.
Children aged 12-15 represented 17.9% of all persons who initiated vaccination in the last 14 days up to June 7, while children aged 16-17 made up 4.8% of vaccine initiation over that period. The 25- to 39-year-olds, at 23.7% of all vaccine initiators, were the only group ahead of those aged 12-15, and the 50- to 64-year-olds were just behind at 17.7%, the CDC data show.
Both groups of children were on the low side, however, when it came to vaccine completion in the last 14 days, with those aged 12-15 at 6.7% of the total and those aged 16-17 years at 4.3%. The only age groups lower than that were ≥75 at 3.5% and <12 at 0.2%, and the highest share of vaccine completion was 26.0% for those aged 25-39, which also happens to be the group with the largest share of the U.S. population (20.5%), the CDC said.
People considered fully vaccinated are those who have received the second dose of a two-dose series or one dose of a single-shot vaccine, but children under age 18 years are eligible only for the Pfizer-BioNTech version, the CDC noted.
Meanwhile, back on the incidence side of the COVID-19 pandemic, the number of new cases in U.S. children for the week ending June 3 was at its lowest point (16,281) since mid-June of 2020, according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
Cases among children now total 3.99 million, which represents 14.1% of cases among all ages, a proportion that hasn’t increased since mid-May, which hasn’t happened since the two groups started keeping track in mid-April of 2020 in the 49 states (excluding New York), the District of Columbia, New York City, Puerto Rico, and Guam that report such data by age.
Less encouraging was the CDC’s report that “COVID-19-associated hospitalization rates among adolescents ages 12-17 years increased during March and April, following declines in January and February 2021.”
Children have been experiencing much lower rates of severe disease than those of adults throughout the pandemic, the CDC pointed out, but “recent increases in COVID-19-associated hospitalization rates and the potential for severe disease in adolescents reinforce the importance of continued prevention strategies, including vaccination and the correct and consistent use of masks in those who are not yet fully vaccinated.”
Air pollution linked to increased fibroid risk in Black women
Black women exposed to ozone air pollution have an increased risk of developing fibroids, according to new research published in Human Production.
Uterine fibroids are a common type of pelvic growth, affecting up to 80% of women by the time they reach age 50, according to the U.S. Department of Health and Human Services. Black women are hit hardest by fibroids; they are diagnosed two to three times the rate of White women and tend to have more severe symptoms.
Researchers are unclear on why exposure to ozone air pollution increases the risk of developing fibroids. However, they believe that when it comes to identifying causes of fibroids and explanations for racial disparities in fibroids, more research that focuses on environmental and neighborhood-level risk factors could help inform policy and interventions to improve gynecologic health.
“A large body of literature from the environmental justice field has documented that people of color, and Black people specifically, are inequitably exposed to air pollution,” study author Amelia K. Wesselink, PhD, assistant professor at Boston University School of Public Health, said in an interview. “And there is growing evidence that air pollution can influence gynecologic health and therefore may contribute to racial disparities in gynecologic outcomes.”
Dr. Wesselink and colleagues wanted to know the extent to which three air pollutants – particulate matter (PM2.5), nitrogen dioxide (NO2) and ozone (03) – were linked to the development of fibroids. To figure this out, they analyzed data on nearly 22,000 premenopausal Black women who lived in 56 metropolitan areas in the United States between 2007 and 2011. They assigned air pollution exposures to participants’ residential addresses collected at baseline and over follow-up and tried to capture long-term exposure to air pollutants.
During the study, nearly 30% of participants reported that they were diagnosed with fibroids. Researchers observed that the exposure to PM2.5 and NO2 was not associated with an increased risk of developing these fibroids.
Dr. Wesselink said the findings may have underestimated fibroid incidence, so they “need to be replicated in a prospective, ultrasound-based study that can identify all fibroid cases.”
“There has not been a lot of research on how air pollution influences fibroid risk, but the two studies that are out there show some evidence of an association,” said Dr. Wesselink. “The fact that our results were consistent with this is interesting. The surprising part of our findings was that we observed an association for ozone, but not for PM2.5 or NO2.”
Nathaniel DeNicola, MD, MSHP, FACOG, a Washington-based obstetrics and gynecology physician affiliated with John Hopkins Health System, applauded the methodology of the study and said the findings prove that patients and doctors should be talking about the environment and exposures to air pollutants.
“[Air pollution] has numerous components to it. And we should try to figure out exactly what components are most dangerous to human health and what doses and what times of life,” said Dr. DeNicola, an environmental health expert.
The increased risk of developing fibroids is a “historical observation” and air pollution may be part of a multifactorial cause of that, Dr. DeNicola said. He said he wouldn’t be surprised if future studies show that “higher exposure [to air pollution] – due to how city planning works, often communities of color are in the areas with the most dense air pollution – exacerbates some other mechanism already in place.
Although it’s unclear how ozone exposure increases fibroid risk, Dr. Wesselink said it may be through a mechanism that is unique to ozone.
“In other words, it might be that there is a factor related to ozone that we did not account for that explains our findings. Vitamin D is a factor that we were not able to account for in this study,” Dr. Wesselink said. “Future work on this topic should consider the role of vitamin D [exposure or deficiency].”
Dr. DeNicola said ozone’s impact may also be tied to its “known association” with hypertension. A 2017 study by Drew B. Day, PhD, of Duke University, Durham, N.C., and colleagues, found that ozone exposure has been linked to hypertension. Meanwhile, a 2015 study has found an association between hypertension and fibroids.
“[This study] does raise an important message. It shines a light where more research needs to be done,” Dr. DeNicola said. “The ozone connection to hypertension was probably most compelling as a true risk factor for uterine fibroids.”
Dr. Wesselink said future work on fibroid etiology should focus on environmental and neighborhood-level exposures to pollutants.
Black women exposed to ozone air pollution have an increased risk of developing fibroids, according to new research published in Human Production.
Uterine fibroids are a common type of pelvic growth, affecting up to 80% of women by the time they reach age 50, according to the U.S. Department of Health and Human Services. Black women are hit hardest by fibroids; they are diagnosed two to three times the rate of White women and tend to have more severe symptoms.
Researchers are unclear on why exposure to ozone air pollution increases the risk of developing fibroids. However, they believe that when it comes to identifying causes of fibroids and explanations for racial disparities in fibroids, more research that focuses on environmental and neighborhood-level risk factors could help inform policy and interventions to improve gynecologic health.
“A large body of literature from the environmental justice field has documented that people of color, and Black people specifically, are inequitably exposed to air pollution,” study author Amelia K. Wesselink, PhD, assistant professor at Boston University School of Public Health, said in an interview. “And there is growing evidence that air pollution can influence gynecologic health and therefore may contribute to racial disparities in gynecologic outcomes.”
Dr. Wesselink and colleagues wanted to know the extent to which three air pollutants – particulate matter (PM2.5), nitrogen dioxide (NO2) and ozone (03) – were linked to the development of fibroids. To figure this out, they analyzed data on nearly 22,000 premenopausal Black women who lived in 56 metropolitan areas in the United States between 2007 and 2011. They assigned air pollution exposures to participants’ residential addresses collected at baseline and over follow-up and tried to capture long-term exposure to air pollutants.
During the study, nearly 30% of participants reported that they were diagnosed with fibroids. Researchers observed that the exposure to PM2.5 and NO2 was not associated with an increased risk of developing these fibroids.
Dr. Wesselink said the findings may have underestimated fibroid incidence, so they “need to be replicated in a prospective, ultrasound-based study that can identify all fibroid cases.”
“There has not been a lot of research on how air pollution influences fibroid risk, but the two studies that are out there show some evidence of an association,” said Dr. Wesselink. “The fact that our results were consistent with this is interesting. The surprising part of our findings was that we observed an association for ozone, but not for PM2.5 or NO2.”
Nathaniel DeNicola, MD, MSHP, FACOG, a Washington-based obstetrics and gynecology physician affiliated with John Hopkins Health System, applauded the methodology of the study and said the findings prove that patients and doctors should be talking about the environment and exposures to air pollutants.
“[Air pollution] has numerous components to it. And we should try to figure out exactly what components are most dangerous to human health and what doses and what times of life,” said Dr. DeNicola, an environmental health expert.
The increased risk of developing fibroids is a “historical observation” and air pollution may be part of a multifactorial cause of that, Dr. DeNicola said. He said he wouldn’t be surprised if future studies show that “higher exposure [to air pollution] – due to how city planning works, often communities of color are in the areas with the most dense air pollution – exacerbates some other mechanism already in place.
Although it’s unclear how ozone exposure increases fibroid risk, Dr. Wesselink said it may be through a mechanism that is unique to ozone.
“In other words, it might be that there is a factor related to ozone that we did not account for that explains our findings. Vitamin D is a factor that we were not able to account for in this study,” Dr. Wesselink said. “Future work on this topic should consider the role of vitamin D [exposure or deficiency].”
Dr. DeNicola said ozone’s impact may also be tied to its “known association” with hypertension. A 2017 study by Drew B. Day, PhD, of Duke University, Durham, N.C., and colleagues, found that ozone exposure has been linked to hypertension. Meanwhile, a 2015 study has found an association between hypertension and fibroids.
“[This study] does raise an important message. It shines a light where more research needs to be done,” Dr. DeNicola said. “The ozone connection to hypertension was probably most compelling as a true risk factor for uterine fibroids.”
Dr. Wesselink said future work on fibroid etiology should focus on environmental and neighborhood-level exposures to pollutants.
Black women exposed to ozone air pollution have an increased risk of developing fibroids, according to new research published in Human Production.
Uterine fibroids are a common type of pelvic growth, affecting up to 80% of women by the time they reach age 50, according to the U.S. Department of Health and Human Services. Black women are hit hardest by fibroids; they are diagnosed two to three times the rate of White women and tend to have more severe symptoms.
Researchers are unclear on why exposure to ozone air pollution increases the risk of developing fibroids. However, they believe that when it comes to identifying causes of fibroids and explanations for racial disparities in fibroids, more research that focuses on environmental and neighborhood-level risk factors could help inform policy and interventions to improve gynecologic health.
“A large body of literature from the environmental justice field has documented that people of color, and Black people specifically, are inequitably exposed to air pollution,” study author Amelia K. Wesselink, PhD, assistant professor at Boston University School of Public Health, said in an interview. “And there is growing evidence that air pollution can influence gynecologic health and therefore may contribute to racial disparities in gynecologic outcomes.”
Dr. Wesselink and colleagues wanted to know the extent to which three air pollutants – particulate matter (PM2.5), nitrogen dioxide (NO2) and ozone (03) – were linked to the development of fibroids. To figure this out, they analyzed data on nearly 22,000 premenopausal Black women who lived in 56 metropolitan areas in the United States between 2007 and 2011. They assigned air pollution exposures to participants’ residential addresses collected at baseline and over follow-up and tried to capture long-term exposure to air pollutants.
During the study, nearly 30% of participants reported that they were diagnosed with fibroids. Researchers observed that the exposure to PM2.5 and NO2 was not associated with an increased risk of developing these fibroids.
Dr. Wesselink said the findings may have underestimated fibroid incidence, so they “need to be replicated in a prospective, ultrasound-based study that can identify all fibroid cases.”
“There has not been a lot of research on how air pollution influences fibroid risk, but the two studies that are out there show some evidence of an association,” said Dr. Wesselink. “The fact that our results were consistent with this is interesting. The surprising part of our findings was that we observed an association for ozone, but not for PM2.5 or NO2.”
Nathaniel DeNicola, MD, MSHP, FACOG, a Washington-based obstetrics and gynecology physician affiliated with John Hopkins Health System, applauded the methodology of the study and said the findings prove that patients and doctors should be talking about the environment and exposures to air pollutants.
“[Air pollution] has numerous components to it. And we should try to figure out exactly what components are most dangerous to human health and what doses and what times of life,” said Dr. DeNicola, an environmental health expert.
The increased risk of developing fibroids is a “historical observation” and air pollution may be part of a multifactorial cause of that, Dr. DeNicola said. He said he wouldn’t be surprised if future studies show that “higher exposure [to air pollution] – due to how city planning works, often communities of color are in the areas with the most dense air pollution – exacerbates some other mechanism already in place.
Although it’s unclear how ozone exposure increases fibroid risk, Dr. Wesselink said it may be through a mechanism that is unique to ozone.
“In other words, it might be that there is a factor related to ozone that we did not account for that explains our findings. Vitamin D is a factor that we were not able to account for in this study,” Dr. Wesselink said. “Future work on this topic should consider the role of vitamin D [exposure or deficiency].”
Dr. DeNicola said ozone’s impact may also be tied to its “known association” with hypertension. A 2017 study by Drew B. Day, PhD, of Duke University, Durham, N.C., and colleagues, found that ozone exposure has been linked to hypertension. Meanwhile, a 2015 study has found an association between hypertension and fibroids.
“[This study] does raise an important message. It shines a light where more research needs to be done,” Dr. DeNicola said. “The ozone connection to hypertension was probably most compelling as a true risk factor for uterine fibroids.”
Dr. Wesselink said future work on fibroid etiology should focus on environmental and neighborhood-level exposures to pollutants.
NIAID advances universal flu vaccine candidate into phase 1 trial
Last month, U.S. government researchers began a test of an experimental influenza vaccine that they hope will provide long-lasting immunity against multiple strains of the virus. Their project adds to the many approaches that have been tried in the decades-long quest for a universal flu shot.
For the first time, the National Institute of Allergy and Infectious Diseases (NIAID) is testing an investigational flu vaccine, known as FluMos-v1, on people. Researchers in recent years have targeted the stalk or stem of an influenza surface protein called hemagglutinin (HA) in trying to develop better flu vaccines. NIAID said FluMos-v1 is designed to spark production of antibodies against the HA protein from different virus strains, which could make it superior to vaccines now available, NIAID said.
“It could be longer lasting than the traditional flu vaccine and give us what we call super seasonal protection that might go beyond just one flu season to next year’s or the year after, or offer additional protection in a pandemic setting,” Alicia T. Widge, MD, of NIAID’s Vaccine Research Center, who is the principal investigator of the trial, said in an interview.
The phase 1 study (NCT04896086) aims to enroll 35 participants, 15 of whom will receive a single intramuscular injection of a comparator treatment, Flucelvax, which has already been approved by the U.S. Food and Drug Administration. The FluMos-v1 group will start with five participants who will receive one 20-μg dose. If no safety problems emerge at that dosage, another 15 volunteers will receive one 60-μg dose of the investigational vaccine.
The incorporation of a comparator group in the phase 1 study may help investigators get an early idea of how well FluMos-v1 compares to a marketed product, Dr. Widge said. The test will be carried out through the National Institutes of Health Clinical Center.
‘Renaissance’ of flu-vaccine research?
Currently, flu vaccines are reformulated each year in an attempt to match the dominant strain for the upcoming season, an effort that often falls notably short. The estimated vaccine effectiveness rate in the United States has ranged from a low of 19% to a high of 60% in recent years, according to the Centers for Disease Control and Prevention.
Scientists have been working for decades on a universal flu vaccine that would offer better results but haven’t yet identified the right strategy to outwit mutations in the virus. Recent setbacks include BiondVax Pharmaceuticals’ October 2020 announcement of a failed phase 3 trial of its experimental M-001 universal flu vaccine candidate.
But advances in understanding the immune system may set the stage for a “renaissance” in efforts to develop a universal flu vaccine, Michael Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, said in an interview.
The COVID-19 pandemic has spurred greater interest in the need to develop a universal flu vaccine, he said. Dr. Osterholm said he is “more optimistic now than ever” about the chances for developing vaccines that can fend off multiple strains over longer periods, although the goal of a shot that can ward off influenza in all cases may remain elusive.
“How good can we make them? Will they ever be really universal? Will they have long periods of protection? I don’t think any of us know that yet,” Dr. Osterholm said. “But this is not the influenza vaccine world of 5 or 7 years ago.”
The mRNA technology used to develop the world’s first approved COVID-19 vaccines, for example, may be applied against influenza, Dr. Osterholm said.
In January 2021, Moderna announced plans to test three development candidates for a seasonal influenza vaccine and aims to start a phase 1 study this year. In an April interview on CNBC’s Squawk Box program, Moderna’s chief executive, Stephané Bancel, spoke about the company’s plans to eventually create a combination vaccine for SARS-Cov-2 and flu viruses.
SARS-CoV-2 “is not going away.” Like flu, this virus will persist and change forms, Ms. Bancel said. Creating a flu shot that outperforms the existing ones would boost confidence in influenza vaccines, which many people now skip, Ms. Bancel said. People might someday be able to get a combination of this more effective flu shot with a COVID-19 vaccine booster in their local pharmacies.
“You can take one dose and then have a nice winter,” Ms. Bancel said of Moderna’s goal for a combination vaccine.
A version of this article first appeared on Medscape.com.
Last month, U.S. government researchers began a test of an experimental influenza vaccine that they hope will provide long-lasting immunity against multiple strains of the virus. Their project adds to the many approaches that have been tried in the decades-long quest for a universal flu shot.
For the first time, the National Institute of Allergy and Infectious Diseases (NIAID) is testing an investigational flu vaccine, known as FluMos-v1, on people. Researchers in recent years have targeted the stalk or stem of an influenza surface protein called hemagglutinin (HA) in trying to develop better flu vaccines. NIAID said FluMos-v1 is designed to spark production of antibodies against the HA protein from different virus strains, which could make it superior to vaccines now available, NIAID said.
“It could be longer lasting than the traditional flu vaccine and give us what we call super seasonal protection that might go beyond just one flu season to next year’s or the year after, or offer additional protection in a pandemic setting,” Alicia T. Widge, MD, of NIAID’s Vaccine Research Center, who is the principal investigator of the trial, said in an interview.
The phase 1 study (NCT04896086) aims to enroll 35 participants, 15 of whom will receive a single intramuscular injection of a comparator treatment, Flucelvax, which has already been approved by the U.S. Food and Drug Administration. The FluMos-v1 group will start with five participants who will receive one 20-μg dose. If no safety problems emerge at that dosage, another 15 volunteers will receive one 60-μg dose of the investigational vaccine.
The incorporation of a comparator group in the phase 1 study may help investigators get an early idea of how well FluMos-v1 compares to a marketed product, Dr. Widge said. The test will be carried out through the National Institutes of Health Clinical Center.
‘Renaissance’ of flu-vaccine research?
Currently, flu vaccines are reformulated each year in an attempt to match the dominant strain for the upcoming season, an effort that often falls notably short. The estimated vaccine effectiveness rate in the United States has ranged from a low of 19% to a high of 60% in recent years, according to the Centers for Disease Control and Prevention.
Scientists have been working for decades on a universal flu vaccine that would offer better results but haven’t yet identified the right strategy to outwit mutations in the virus. Recent setbacks include BiondVax Pharmaceuticals’ October 2020 announcement of a failed phase 3 trial of its experimental M-001 universal flu vaccine candidate.
But advances in understanding the immune system may set the stage for a “renaissance” in efforts to develop a universal flu vaccine, Michael Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, said in an interview.
The COVID-19 pandemic has spurred greater interest in the need to develop a universal flu vaccine, he said. Dr. Osterholm said he is “more optimistic now than ever” about the chances for developing vaccines that can fend off multiple strains over longer periods, although the goal of a shot that can ward off influenza in all cases may remain elusive.
“How good can we make them? Will they ever be really universal? Will they have long periods of protection? I don’t think any of us know that yet,” Dr. Osterholm said. “But this is not the influenza vaccine world of 5 or 7 years ago.”
The mRNA technology used to develop the world’s first approved COVID-19 vaccines, for example, may be applied against influenza, Dr. Osterholm said.
In January 2021, Moderna announced plans to test three development candidates for a seasonal influenza vaccine and aims to start a phase 1 study this year. In an April interview on CNBC’s Squawk Box program, Moderna’s chief executive, Stephané Bancel, spoke about the company’s plans to eventually create a combination vaccine for SARS-Cov-2 and flu viruses.
SARS-CoV-2 “is not going away.” Like flu, this virus will persist and change forms, Ms. Bancel said. Creating a flu shot that outperforms the existing ones would boost confidence in influenza vaccines, which many people now skip, Ms. Bancel said. People might someday be able to get a combination of this more effective flu shot with a COVID-19 vaccine booster in their local pharmacies.
“You can take one dose and then have a nice winter,” Ms. Bancel said of Moderna’s goal for a combination vaccine.
A version of this article first appeared on Medscape.com.
Last month, U.S. government researchers began a test of an experimental influenza vaccine that they hope will provide long-lasting immunity against multiple strains of the virus. Their project adds to the many approaches that have been tried in the decades-long quest for a universal flu shot.
For the first time, the National Institute of Allergy and Infectious Diseases (NIAID) is testing an investigational flu vaccine, known as FluMos-v1, on people. Researchers in recent years have targeted the stalk or stem of an influenza surface protein called hemagglutinin (HA) in trying to develop better flu vaccines. NIAID said FluMos-v1 is designed to spark production of antibodies against the HA protein from different virus strains, which could make it superior to vaccines now available, NIAID said.
“It could be longer lasting than the traditional flu vaccine and give us what we call super seasonal protection that might go beyond just one flu season to next year’s or the year after, or offer additional protection in a pandemic setting,” Alicia T. Widge, MD, of NIAID’s Vaccine Research Center, who is the principal investigator of the trial, said in an interview.
The phase 1 study (NCT04896086) aims to enroll 35 participants, 15 of whom will receive a single intramuscular injection of a comparator treatment, Flucelvax, which has already been approved by the U.S. Food and Drug Administration. The FluMos-v1 group will start with five participants who will receive one 20-μg dose. If no safety problems emerge at that dosage, another 15 volunteers will receive one 60-μg dose of the investigational vaccine.
The incorporation of a comparator group in the phase 1 study may help investigators get an early idea of how well FluMos-v1 compares to a marketed product, Dr. Widge said. The test will be carried out through the National Institutes of Health Clinical Center.
‘Renaissance’ of flu-vaccine research?
Currently, flu vaccines are reformulated each year in an attempt to match the dominant strain for the upcoming season, an effort that often falls notably short. The estimated vaccine effectiveness rate in the United States has ranged from a low of 19% to a high of 60% in recent years, according to the Centers for Disease Control and Prevention.
Scientists have been working for decades on a universal flu vaccine that would offer better results but haven’t yet identified the right strategy to outwit mutations in the virus. Recent setbacks include BiondVax Pharmaceuticals’ October 2020 announcement of a failed phase 3 trial of its experimental M-001 universal flu vaccine candidate.
But advances in understanding the immune system may set the stage for a “renaissance” in efforts to develop a universal flu vaccine, Michael Osterholm, PhD, MPH, director of the Center for Infectious Disease Research and Policy at the University of Minnesota, said in an interview.
The COVID-19 pandemic has spurred greater interest in the need to develop a universal flu vaccine, he said. Dr. Osterholm said he is “more optimistic now than ever” about the chances for developing vaccines that can fend off multiple strains over longer periods, although the goal of a shot that can ward off influenza in all cases may remain elusive.
“How good can we make them? Will they ever be really universal? Will they have long periods of protection? I don’t think any of us know that yet,” Dr. Osterholm said. “But this is not the influenza vaccine world of 5 or 7 years ago.”
The mRNA technology used to develop the world’s first approved COVID-19 vaccines, for example, may be applied against influenza, Dr. Osterholm said.
In January 2021, Moderna announced plans to test three development candidates for a seasonal influenza vaccine and aims to start a phase 1 study this year. In an April interview on CNBC’s Squawk Box program, Moderna’s chief executive, Stephané Bancel, spoke about the company’s plans to eventually create a combination vaccine for SARS-Cov-2 and flu viruses.
SARS-CoV-2 “is not going away.” Like flu, this virus will persist and change forms, Ms. Bancel said. Creating a flu shot that outperforms the existing ones would boost confidence in influenza vaccines, which many people now skip, Ms. Bancel said. People might someday be able to get a combination of this more effective flu shot with a COVID-19 vaccine booster in their local pharmacies.
“You can take one dose and then have a nice winter,” Ms. Bancel said of Moderna’s goal for a combination vaccine.
A version of this article first appeared on Medscape.com.