User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
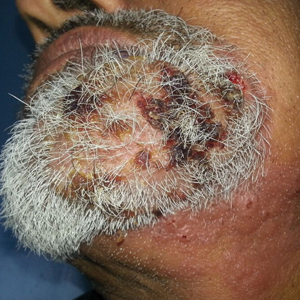
Multiple Crusted Swellings on the Chin
The Diagnosis: Cutaneous Cryptococcosis
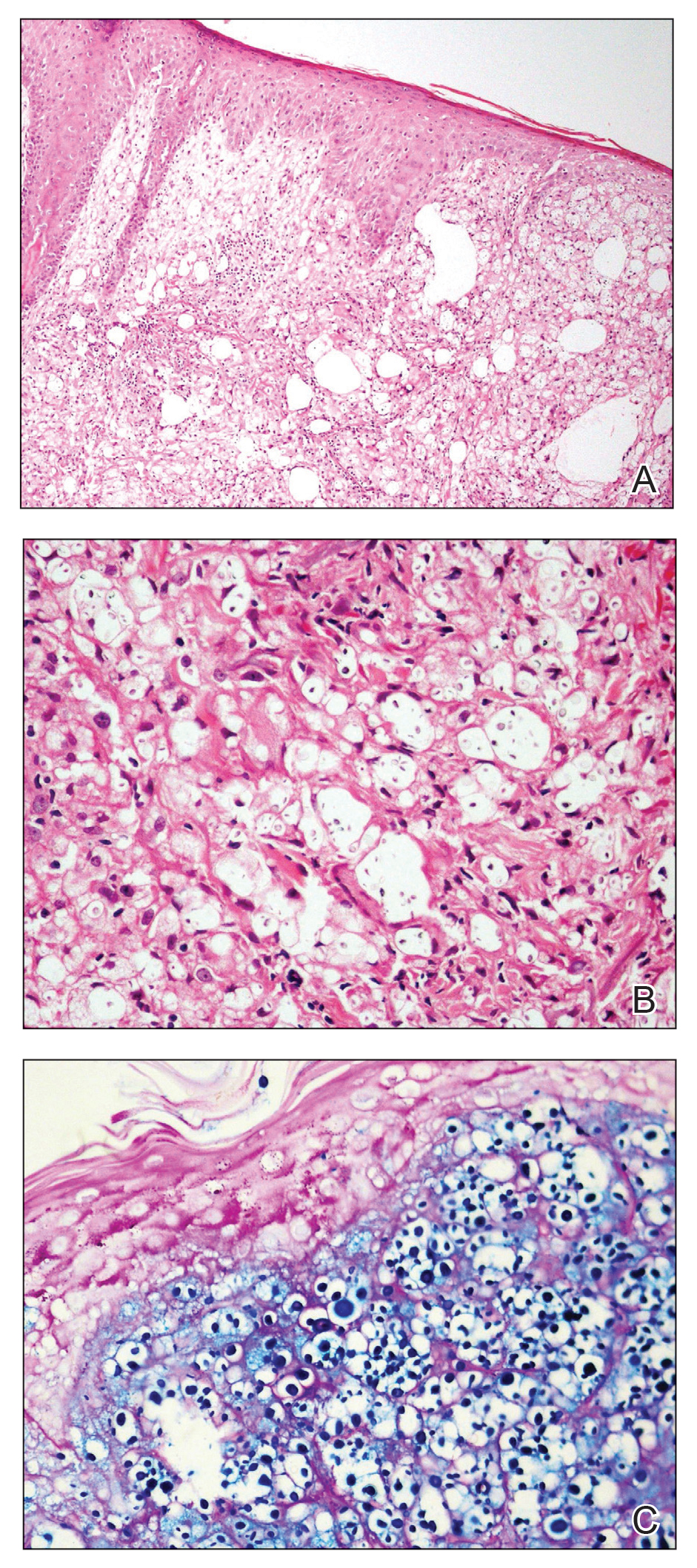
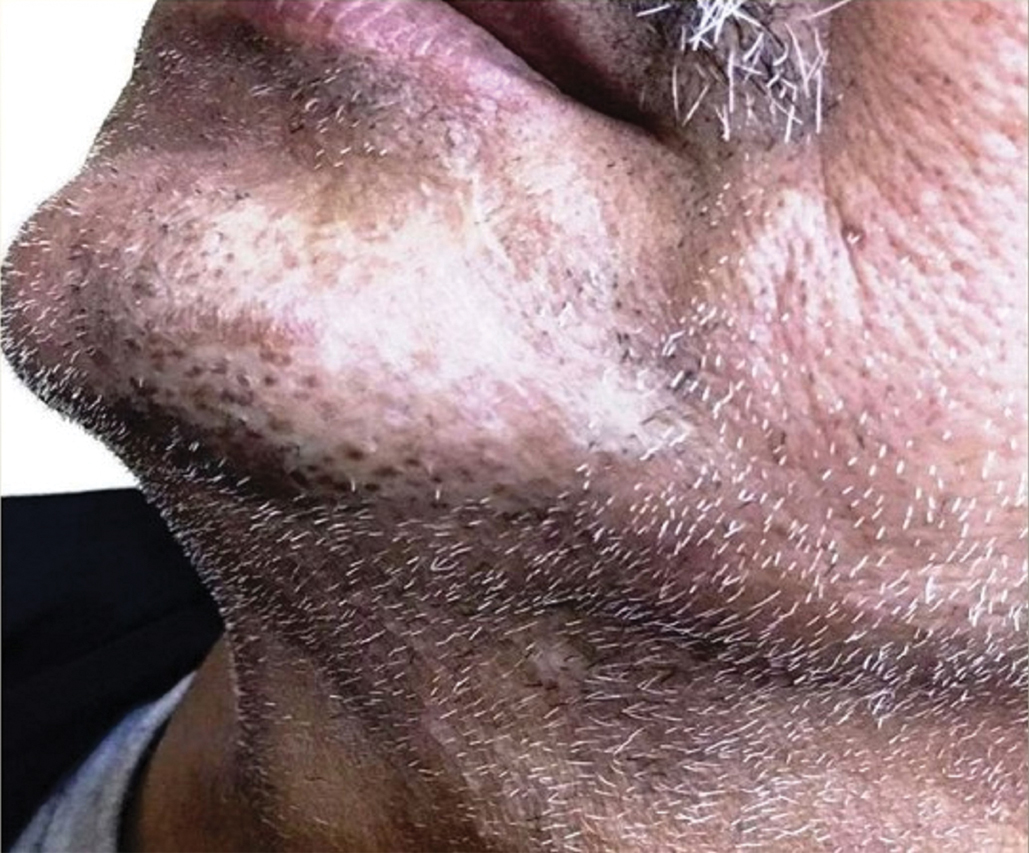
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).
Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
The Diagnosis: Cutaneous Cryptococcosis
Histologic examination revealed infiltration of the dermis and subcutaneous tissue with rounded basophilic cells on low magnification (Figure 1A). On higher magnification, encapsulated yeast cells (cryptococci) of varying size accompanied by chronic granulomatous inflammatory infiltration with occasional giant cells were seen (Figure 1B). Alcian blue stain showed mucinous capsular material (Figure 1C). There was no history of diabetes mellitus, tuberculosis, steroid therapy, or immunosuppression. Moreover, systemic involvement or systemic focus of infection was ruled out after computed tomography of the head, chest, and abdomen. Therefore, the diagnosis of primary cutaneous cryptococcosis (PCC) was established. The patient was started on oral itraconazole 100 mg twice daily along with 5 drops of a saturated solution of potassium iodide 3 times daily that later was increased to 20 drops 3 times daily at a weekly interval. The lesions started improving after 1 month and healed completely after 9 months of treatment (Figure 2).


Primary cutaneous cryptococcosis is the identification of Cryptococcus neoformans in a skin lesion without evidence of simultaneous disseminated disease. Neuville et al1 observed that skin lesions resemble cellulitis, ulcerations, or whitlows and were located on unclothed areas. In contrast, lesions from disseminated disease presented as scattered umbilicated papules resembling molluscum contagiosum. Diagnosis of PCC is based on the observation of encapsulated yeasts by direct microscopic examination, isolation of C neoformans or Cryptococcus gattii in culture, and by the demonstration of capsular antigen in various fluids, including serum and cerebrospinal fluid by latex particle agglutination or enzyme-linked immunosorbent assay. Histologically, Cryptococcus species produce a proliferative inflammatory reaction in immunocompetent hosts with the formation of compact epithelioid granulomas, with giant cells and a peripheral layer of lymphocytes. Treatment options for PCC infection range from antifungal medications and surgical debridement to observation.
The differential diagnosis may include cutaneous leishmaniasis, cutaneous tuberculosis, cutaneous histoplasmosis, and basal cell carcinoma. These entities may have similar presentations and can only be confidently differentiated on direct microscopy and histopathologic examination. The characteristic Leishmania donovani bodies on microscopy in cutaneous leishmaniasis and tubercular granuloma with central necrosis on histology in cutaneous tuberculosis can differentiate these conditions from cryptococcosis. In some patients with cryptococcosis, the yeast may produce a less characteristic polysaccharide capsule and thus may be confused with histoplasmosis. Fontana-Masson staining may show melanin-producing yeast, which is characteristic of cryptococci.2 Ulcerated basal cell carcinoma may present similar clinically; however, histopathology will rule it out.
Cutaneous cryptococcal infection should be presumed to be disseminated until proven otherwise, and a search for other sites of involvement must immediately be undertaken. Cutaneous signs may be the first indication of infection, preceding the diagnosis of disseminated disease by 2 to 8 months, making its recognition crucial to early treatment. It is not possible to diagnose PCC on a specific clinical manifestation because a diverse range of skin lesions may be present. Therefore, culture and histology are the gold standards for diagnosis of cryptococcosis.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
- Neuville S, Dromer F, Morin O, et al. Primary cutaneous cryptococcosis: a distinct clinical entity. Clin Infect Dis. 2003;36:337-347.
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280.
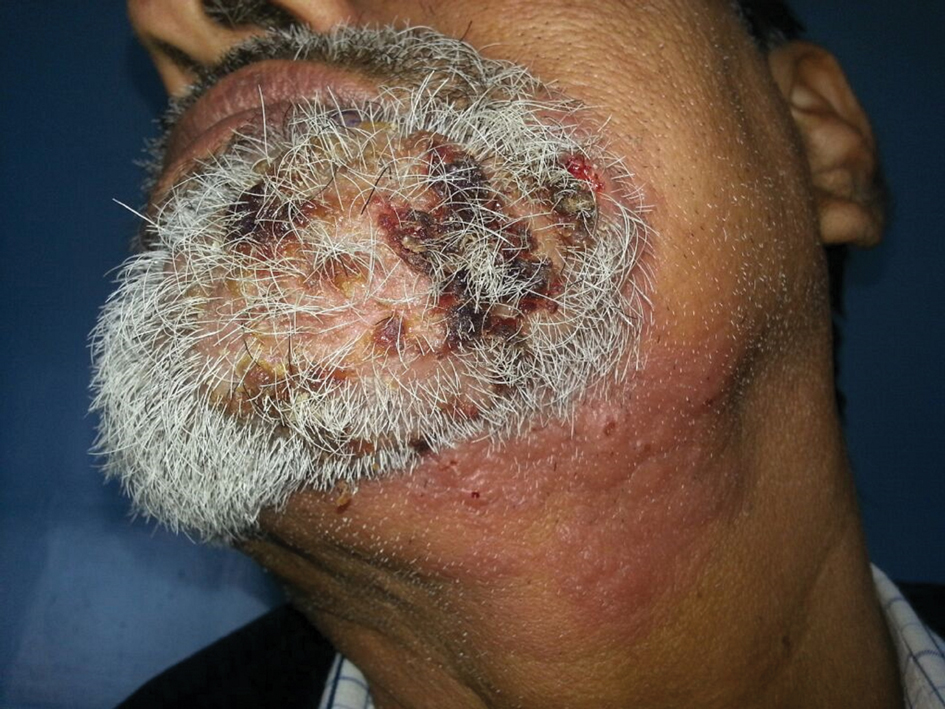
A 54-year-old man with no comorbidities presented with multiple painless swellings on the left side of the chin of 1 month’s duration that progressively were increasing, both in size and number. He denied any discharge of pus or grains from the lesion, facial trauma, insect bites, or dental procedures. The patient was treated with oral antibiotics for 15 days with no relief at an outside hospital. All routine blood and serologic investigations including viral markers and chest radiography were normal. Bacterial and fungal cultures as well as an acid-fast bacilli culture were negative. Systemic examination was normal, and vitals were within reference range. Mucocutaneous examination revealed multiple nontender small nodules and plaques with yellow-brown to dark brown hemorrhagic crusts with mild perilesional erythema on the left side of the chin extending to the adjacent neck. All mucosal sites were normal, and a biopsy was performed.
In Zambia, PCR tracks pertussis
In the periurban slum of Lusaka, Zambia, asymptomatic pertussis infections were common among both mothers and infants, a surprising finding since asymptomatic infections are assumed to be rare in infants. The findings suggested that pertussis should be considered in cases of chronic cough, and that current standards of treating pertussis infections in low-resource settings may need to be reexamined.
The results come from testing of 1,320 infant-mother pairs who were first enrolled at a public health clinic, then followed over at least four visits. The researchers tracked pertussis infection using quantitative PCR (qPCR) on nasopharyngeal swabs. Over the course of the study, 8.9% tested positive, although only one infant developed clinical pertussis during the study.
The study was presented by Christian Gunning, a postdoctoral researcher at the University of Georgia, at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year. The group also included researchers at Boston University and the University of Zambia, where PCR tests were conducted.
“That was amazing,” said session moderator Vana Spoulou, MD, PhD, professor of pediatric infectious diseases at National and Kapodistrian University of Athens, who is associated with Aghia Sofia Children’s Hospital of Athens. She noted that the study found that many physicians misdiagnosed coughs, believing them to be caused by another agent. “It was very interesting that there was so much pertussis spreading around in that community, and that nobody knew that it was around,” said Dr. Spoulou.
It’s important that physicians provide appropriate treatment, since ampicillin, which is typically prescribed for childhood upper respiratory illnesses, is believed to be ineffective against pertussis, while macrolides are effective and can prevent transmission.
Dr. Spoulou also noted that Zambia uses a whole cell vaccine, which is contraindicated in pregnant women because of potential side effects. “The good thing, despite that there was [a lot of] infection, there were no deaths, which means that maybe because the mother was infected, maybe some antibodies of the mother had passed to the child and could help the child to develop milder symptoms. So these are the pros and cons of natural infection,” said Dr. Spoulou.
The study took place in 2015, and participants were seen at the Chawama Public Health Clinic from about age 1 week to 4 months (with a target of seven clinic visits). Researchers recorded respiratory symptoms and antibiotics use at each visit, and collected a nasopharyngeal swab that was tested retrospectively using qPCR for Bordetella pertussis.
Real-time PCR analysis of the samples yields the CT value, which represents the number of amplification cycles that the PCR test must complete before Bordetella pertussis is detectable. The fewer the cycles (and the lower the CT value), the more infectious particles must have been present in the sample. For pertussis testing, a value below 35 is considered a clinically positive result. Tests that come back with higher CT values are increasingly likely to be false positives.
The researchers plotted a value called evidence for infection (EFI), which combined a range of CT values with the number of positive tests over the seven clinic visits to group patients into none, weak, or strong EFI. Among infants with no symptoms, 77% were in the no EFI category, 16% were in the weak category, and 7% were in the strong EFI group. Of infants with minimal respiratory symptoms, 18% were in the strong group, and 20% with moderate to severe symptoms were in the strong EFI group. Among mothers, 13% with no symptoms were in the strong group. 19% in the minimal symptom group were categorized as strong EFI, as were 11% in the moderate to severe symptom group.
The study used a full range of CT, not just positive test results (for pertussis, CT ≤ 35). Beyond contributing to composite measures such as EFI, CT values can serve as leading indicators of infectious disease outbreaks in a population, according to Dr. Gunning. That’s because weaker qPCR signals (CT > 35) can provide additional information within a large sample population. Higher CT values are successively more prone to false positives, but that’s less important for disease surveillance where sensitivity is of the highest importance. The false positive “noise” tends to cancel out over time. “It may be the case that you don’t make that call (correctly) 100% of the time for 100% of the people, but if you get it right in 80 out of 100 people, that’s sufficient to say we see this pathogen circulating in the population,” said Dr. Gunning.
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Gunning and Dr. Spoulou have no relevant financial disclosures.
In the periurban slum of Lusaka, Zambia, asymptomatic pertussis infections were common among both mothers and infants, a surprising finding since asymptomatic infections are assumed to be rare in infants. The findings suggested that pertussis should be considered in cases of chronic cough, and that current standards of treating pertussis infections in low-resource settings may need to be reexamined.
The results come from testing of 1,320 infant-mother pairs who were first enrolled at a public health clinic, then followed over at least four visits. The researchers tracked pertussis infection using quantitative PCR (qPCR) on nasopharyngeal swabs. Over the course of the study, 8.9% tested positive, although only one infant developed clinical pertussis during the study.
The study was presented by Christian Gunning, a postdoctoral researcher at the University of Georgia, at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year. The group also included researchers at Boston University and the University of Zambia, where PCR tests were conducted.
“That was amazing,” said session moderator Vana Spoulou, MD, PhD, professor of pediatric infectious diseases at National and Kapodistrian University of Athens, who is associated with Aghia Sofia Children’s Hospital of Athens. She noted that the study found that many physicians misdiagnosed coughs, believing them to be caused by another agent. “It was very interesting that there was so much pertussis spreading around in that community, and that nobody knew that it was around,” said Dr. Spoulou.
It’s important that physicians provide appropriate treatment, since ampicillin, which is typically prescribed for childhood upper respiratory illnesses, is believed to be ineffective against pertussis, while macrolides are effective and can prevent transmission.
Dr. Spoulou also noted that Zambia uses a whole cell vaccine, which is contraindicated in pregnant women because of potential side effects. “The good thing, despite that there was [a lot of] infection, there were no deaths, which means that maybe because the mother was infected, maybe some antibodies of the mother had passed to the child and could help the child to develop milder symptoms. So these are the pros and cons of natural infection,” said Dr. Spoulou.
The study took place in 2015, and participants were seen at the Chawama Public Health Clinic from about age 1 week to 4 months (with a target of seven clinic visits). Researchers recorded respiratory symptoms and antibiotics use at each visit, and collected a nasopharyngeal swab that was tested retrospectively using qPCR for Bordetella pertussis.
Real-time PCR analysis of the samples yields the CT value, which represents the number of amplification cycles that the PCR test must complete before Bordetella pertussis is detectable. The fewer the cycles (and the lower the CT value), the more infectious particles must have been present in the sample. For pertussis testing, a value below 35 is considered a clinically positive result. Tests that come back with higher CT values are increasingly likely to be false positives.
The researchers plotted a value called evidence for infection (EFI), which combined a range of CT values with the number of positive tests over the seven clinic visits to group patients into none, weak, or strong EFI. Among infants with no symptoms, 77% were in the no EFI category, 16% were in the weak category, and 7% were in the strong EFI group. Of infants with minimal respiratory symptoms, 18% were in the strong group, and 20% with moderate to severe symptoms were in the strong EFI group. Among mothers, 13% with no symptoms were in the strong group. 19% in the minimal symptom group were categorized as strong EFI, as were 11% in the moderate to severe symptom group.
The study used a full range of CT, not just positive test results (for pertussis, CT ≤ 35). Beyond contributing to composite measures such as EFI, CT values can serve as leading indicators of infectious disease outbreaks in a population, according to Dr. Gunning. That’s because weaker qPCR signals (CT > 35) can provide additional information within a large sample population. Higher CT values are successively more prone to false positives, but that’s less important for disease surveillance where sensitivity is of the highest importance. The false positive “noise” tends to cancel out over time. “It may be the case that you don’t make that call (correctly) 100% of the time for 100% of the people, but if you get it right in 80 out of 100 people, that’s sufficient to say we see this pathogen circulating in the population,” said Dr. Gunning.
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Gunning and Dr. Spoulou have no relevant financial disclosures.
In the periurban slum of Lusaka, Zambia, asymptomatic pertussis infections were common among both mothers and infants, a surprising finding since asymptomatic infections are assumed to be rare in infants. The findings suggested that pertussis should be considered in cases of chronic cough, and that current standards of treating pertussis infections in low-resource settings may need to be reexamined.
The results come from testing of 1,320 infant-mother pairs who were first enrolled at a public health clinic, then followed over at least four visits. The researchers tracked pertussis infection using quantitative PCR (qPCR) on nasopharyngeal swabs. Over the course of the study, 8.9% tested positive, although only one infant developed clinical pertussis during the study.
The study was presented by Christian Gunning, a postdoctoral researcher at the University of Georgia, at the annual meeting of the European Society for Paediatric Infectious Diseases, held virtually this year. The group also included researchers at Boston University and the University of Zambia, where PCR tests were conducted.
“That was amazing,” said session moderator Vana Spoulou, MD, PhD, professor of pediatric infectious diseases at National and Kapodistrian University of Athens, who is associated with Aghia Sofia Children’s Hospital of Athens. She noted that the study found that many physicians misdiagnosed coughs, believing them to be caused by another agent. “It was very interesting that there was so much pertussis spreading around in that community, and that nobody knew that it was around,” said Dr. Spoulou.
It’s important that physicians provide appropriate treatment, since ampicillin, which is typically prescribed for childhood upper respiratory illnesses, is believed to be ineffective against pertussis, while macrolides are effective and can prevent transmission.
Dr. Spoulou also noted that Zambia uses a whole cell vaccine, which is contraindicated in pregnant women because of potential side effects. “The good thing, despite that there was [a lot of] infection, there were no deaths, which means that maybe because the mother was infected, maybe some antibodies of the mother had passed to the child and could help the child to develop milder symptoms. So these are the pros and cons of natural infection,” said Dr. Spoulou.
The study took place in 2015, and participants were seen at the Chawama Public Health Clinic from about age 1 week to 4 months (with a target of seven clinic visits). Researchers recorded respiratory symptoms and antibiotics use at each visit, and collected a nasopharyngeal swab that was tested retrospectively using qPCR for Bordetella pertussis.
Real-time PCR analysis of the samples yields the CT value, which represents the number of amplification cycles that the PCR test must complete before Bordetella pertussis is detectable. The fewer the cycles (and the lower the CT value), the more infectious particles must have been present in the sample. For pertussis testing, a value below 35 is considered a clinically positive result. Tests that come back with higher CT values are increasingly likely to be false positives.
The researchers plotted a value called evidence for infection (EFI), which combined a range of CT values with the number of positive tests over the seven clinic visits to group patients into none, weak, or strong EFI. Among infants with no symptoms, 77% were in the no EFI category, 16% were in the weak category, and 7% were in the strong EFI group. Of infants with minimal respiratory symptoms, 18% were in the strong group, and 20% with moderate to severe symptoms were in the strong EFI group. Among mothers, 13% with no symptoms were in the strong group. 19% in the minimal symptom group were categorized as strong EFI, as were 11% in the moderate to severe symptom group.
The study used a full range of CT, not just positive test results (for pertussis, CT ≤ 35). Beyond contributing to composite measures such as EFI, CT values can serve as leading indicators of infectious disease outbreaks in a population, according to Dr. Gunning. That’s because weaker qPCR signals (CT > 35) can provide additional information within a large sample population. Higher CT values are successively more prone to false positives, but that’s less important for disease surveillance where sensitivity is of the highest importance. The false positive “noise” tends to cancel out over time. “It may be the case that you don’t make that call (correctly) 100% of the time for 100% of the people, but if you get it right in 80 out of 100 people, that’s sufficient to say we see this pathogen circulating in the population,” said Dr. Gunning.
The study was funded by the National Institute of Allergy and Infectious Diseases. Dr. Gunning and Dr. Spoulou have no relevant financial disclosures.
FROM ESPID 2021
Children aged 12-15 years continue to close COVID-19 vaccination gap
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.
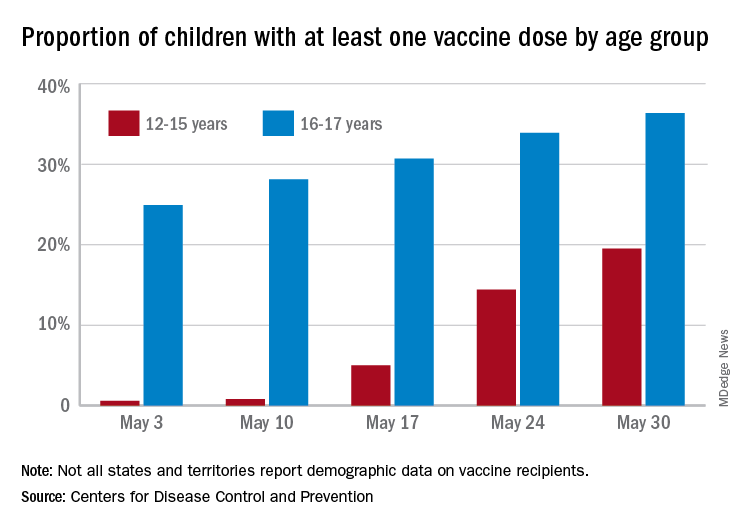
with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.

with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
More children aged 12-15 years already have received at least one dose of a COVID-19 vaccine than have 16- and 17-year-olds, based on data from the Centers for Disease Control and Prevention.

with those figures representing increases of 31.6% and 6.6% in the past week, respectively. Since the overall size of the 12-15 population is much larger, however, the proportion vaccinated is still smaller: 19.5% to 36.4%, according to the CDC’s COVID Data Tracker.
A look at full vaccination status shows that only 0.7% of those aged 12-15 years have received both doses of a two-dose vaccine or one dose of the single-shot variety, compared with 24% of those aged 16-17. For the country as a whole, 50.5% of all ages have received at least one dose and 40.7% are fully vaccinated, the CDC said.
Children aged 12-15 represent the largest share of the U.S. population (23.4%) initiating vaccination in the 14 days ending May 30, while children aged 16-17 made up just 4.5% of those getting their first dose. The younger group’s later entry into the vaccination pool shows up again when looking at completion rates, though, representing just 0.4% of all Americans who reached full vaccination during that same 14-day period, compared with 4.6% of the older children, the CDC data show.
Not all states are reporting data such as age for vaccine recipients, the CDC noted, and there are other variables that affect data collection. “Demographic data ... might differ by populations prioritized within each state or jurisdiction’s vaccination phase. Every geographic area has a different racial and ethnic composition, and not all are in the same vaccination phase,” the CDC said.
Semaglutide boosts weight loss following endoscopic gastroplasty
Combining minimally invasive endoscopic sleeve gastroplasty with a weekly injection of the glucagonlike peptide–1 agonist semaglutide (Ozempic, Novo Nordisk) leads to significantly greater weight loss than ESG alone in patients with diabetes and excess weight who are not candidates for bariatric surgery, new research shows.
During minimally invasive ESG, a flexible endoscope equipped with an endoscopic suturing device is inserted down the esophagus and into the stomach. The endoscopist then applies the sutures to the upper portion of the stomach, minimizing its size to restrict the amount of food a patient can ingest.
“Our stomachs can stretch back a bit, but we can use the suturing device again,” explained the lead investigator of the research Anna Carolina Hoff, MD, founder and clinical director of Angioskope Brazil in São José dos Campos.
“It’s important that patients with diabetes lose as much weight as possible because, if they lose about 10% of their total body weight, they have a great improvement in their glycemic levels, and some patients can even stop taking their [antidiabetic] medications,” Dr. Hoff said in an interview.
“And we found that by adding the GLP-1 agonist [semaglutide], we could increase weight loss from, on average, about 16%-18% of total body weight with ESG alone to up to 27%, so it’s a great metabolic combination,” she noted.
Dr. Hoff presented the findings at the annual Digestive Disease Week® (DDW).
Asked to comment, Scott Kahan, MD, MPH, director, National Center for Weight and Wellness, George Washington University, Washington, cautioned that it’s still early days for minimally invasive ESG.
“It is reasonable to assume that the long-term outcomes [with ESG] won’t be as good or durable over time as with bariatric surgery, but ... we will have to see.”
However, “we know that, typically, combinations of therapeutic options work better than a one-off option, so I think the real benefit of this study – outside the specific procedure and this specific medication – is that it is a very valuable proof-of-principle study showing that combinations do work better,” Dr. Kahan said in an interview.
Minimally invasive endoscopic sleeve gastroplasty
ESG is a surrogate for laparoscopic sleeve gastrectomy that can offer the benefits of such a procedure to those who don’t qualify for, or don’t wish to pursue, bariatric surgery. It can be performed at an earlier stage of disease, in those with a body mass index of 30 mg/kg2, whereas generally people are not offered bariatric procedures unless they have a BMI of at least 35 with comorbidities or a BMI of at least 40 if they do not have comorbidities.
Subcutaneous semaglutide is already approved for the treatment of type 2 diabetes in adults at doses of up to 1 mg/week; higher doses are needed for weight loss. Novo Nordisk has been investigating higher doses for weight loss in the STEP trial program, which is now complete, and the company has submitted the data to the Food and Drug Administration and European Medicines Agency for an additional indication of adults with obesity (BMI ≥30) or who are overweight (BMI ≥27) and who have at least one weight-related comorbidity, as an adjunct to a reduced-calorie diet and increased physical activity, with a decision expected soon.
Novo Nordisk has also developed an oral form of semaglutide, which has been approved as a once-daily agent for type 2 diabetes (Rybelsus) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Patients lost fat mass as well as excess weight
The Brazilian study involved 58 patients with obesity or overweight who also had diabetes and were undergoing minimally invasive ESG; they were further randomized to receive semaglutide or placebo.
The GLP-1 agonist (or sham placebo) was initiated 1 month after participants had undergone the procedure and patients were monitored each month for weight loss and type of fat loss achieved with the combination versus ESG alone. The initial dose of semaglutide used was 0.25 mg subcutaneous a week but could be titrated up to a maximum dose of 1.5 mg.
At the end of 11 months of active treatment versus placebo (12 months after ESG), patients who received additional semaglutide lost 86.3% of their excess body weight – the amount of weight patients needed to lose to reach normal BMI – compared with only 60.4% for ESG controls.
Specifically, the mean percentage total body weight loss at the end of 12 months was 25.2% for those in the combination group, compared with 18.6% for those treated with ESG alone (P < .001).
More importantly, patients in the combination group lost 12.6% of their body fat mass, compared with 9% for ESG controls, while mean A1c levels fell more in those treated with additional semaglutide compared with controls (P = .0394).
Indeed, five patients in the combination group reverted to a nondiabetic state and were able to discontinue antidiabetic medications altogether, Dr. Hoff noted.
“Our main goal is not just to lose weight but to lose body mass fat, which is very different from just losing weight,” she explained.
If patients lose weight but still maintain a high percentage of body fat mass, they have what she refers to as “sarcopenic obesity” because in this state patients have lost a lot of muscle mass but still have high levels of metabolically active visceral fat. Among many other inflammatory complexes, metabolically active visceral fat contains a large number of inflammasomes, and it is the latter that have been associated with obesity-related cancers.
“Obesity is a progressive disease, so what we are trying to do here is buy time for patients so they do not progress to [bariatric] surgery, and this approach gives patients a chance to act earlier before obesity takes over and more metabolic consequences occur,” Dr. Hoff emphasized.
So, when combined with semaglutide, “we now have a minimally invasive procedure that can be just as successful [as surgery] and which can be made available to even more people looking to lose a significant amount of weight,” she concluded.
Dr. Hoff and Dr. Kahan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Combining minimally invasive endoscopic sleeve gastroplasty with a weekly injection of the glucagonlike peptide–1 agonist semaglutide (Ozempic, Novo Nordisk) leads to significantly greater weight loss than ESG alone in patients with diabetes and excess weight who are not candidates for bariatric surgery, new research shows.
During minimally invasive ESG, a flexible endoscope equipped with an endoscopic suturing device is inserted down the esophagus and into the stomach. The endoscopist then applies the sutures to the upper portion of the stomach, minimizing its size to restrict the amount of food a patient can ingest.
“Our stomachs can stretch back a bit, but we can use the suturing device again,” explained the lead investigator of the research Anna Carolina Hoff, MD, founder and clinical director of Angioskope Brazil in São José dos Campos.
“It’s important that patients with diabetes lose as much weight as possible because, if they lose about 10% of their total body weight, they have a great improvement in their glycemic levels, and some patients can even stop taking their [antidiabetic] medications,” Dr. Hoff said in an interview.
“And we found that by adding the GLP-1 agonist [semaglutide], we could increase weight loss from, on average, about 16%-18% of total body weight with ESG alone to up to 27%, so it’s a great metabolic combination,” she noted.
Dr. Hoff presented the findings at the annual Digestive Disease Week® (DDW).
Asked to comment, Scott Kahan, MD, MPH, director, National Center for Weight and Wellness, George Washington University, Washington, cautioned that it’s still early days for minimally invasive ESG.
“It is reasonable to assume that the long-term outcomes [with ESG] won’t be as good or durable over time as with bariatric surgery, but ... we will have to see.”
However, “we know that, typically, combinations of therapeutic options work better than a one-off option, so I think the real benefit of this study – outside the specific procedure and this specific medication – is that it is a very valuable proof-of-principle study showing that combinations do work better,” Dr. Kahan said in an interview.
Minimally invasive endoscopic sleeve gastroplasty
ESG is a surrogate for laparoscopic sleeve gastrectomy that can offer the benefits of such a procedure to those who don’t qualify for, or don’t wish to pursue, bariatric surgery. It can be performed at an earlier stage of disease, in those with a body mass index of 30 mg/kg2, whereas generally people are not offered bariatric procedures unless they have a BMI of at least 35 with comorbidities or a BMI of at least 40 if they do not have comorbidities.
Subcutaneous semaglutide is already approved for the treatment of type 2 diabetes in adults at doses of up to 1 mg/week; higher doses are needed for weight loss. Novo Nordisk has been investigating higher doses for weight loss in the STEP trial program, which is now complete, and the company has submitted the data to the Food and Drug Administration and European Medicines Agency for an additional indication of adults with obesity (BMI ≥30) or who are overweight (BMI ≥27) and who have at least one weight-related comorbidity, as an adjunct to a reduced-calorie diet and increased physical activity, with a decision expected soon.
Novo Nordisk has also developed an oral form of semaglutide, which has been approved as a once-daily agent for type 2 diabetes (Rybelsus) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Patients lost fat mass as well as excess weight
The Brazilian study involved 58 patients with obesity or overweight who also had diabetes and were undergoing minimally invasive ESG; they were further randomized to receive semaglutide or placebo.
The GLP-1 agonist (or sham placebo) was initiated 1 month after participants had undergone the procedure and patients were monitored each month for weight loss and type of fat loss achieved with the combination versus ESG alone. The initial dose of semaglutide used was 0.25 mg subcutaneous a week but could be titrated up to a maximum dose of 1.5 mg.
At the end of 11 months of active treatment versus placebo (12 months after ESG), patients who received additional semaglutide lost 86.3% of their excess body weight – the amount of weight patients needed to lose to reach normal BMI – compared with only 60.4% for ESG controls.
Specifically, the mean percentage total body weight loss at the end of 12 months was 25.2% for those in the combination group, compared with 18.6% for those treated with ESG alone (P < .001).
More importantly, patients in the combination group lost 12.6% of their body fat mass, compared with 9% for ESG controls, while mean A1c levels fell more in those treated with additional semaglutide compared with controls (P = .0394).
Indeed, five patients in the combination group reverted to a nondiabetic state and were able to discontinue antidiabetic medications altogether, Dr. Hoff noted.
“Our main goal is not just to lose weight but to lose body mass fat, which is very different from just losing weight,” she explained.
If patients lose weight but still maintain a high percentage of body fat mass, they have what she refers to as “sarcopenic obesity” because in this state patients have lost a lot of muscle mass but still have high levels of metabolically active visceral fat. Among many other inflammatory complexes, metabolically active visceral fat contains a large number of inflammasomes, and it is the latter that have been associated with obesity-related cancers.
“Obesity is a progressive disease, so what we are trying to do here is buy time for patients so they do not progress to [bariatric] surgery, and this approach gives patients a chance to act earlier before obesity takes over and more metabolic consequences occur,” Dr. Hoff emphasized.
So, when combined with semaglutide, “we now have a minimally invasive procedure that can be just as successful [as surgery] and which can be made available to even more people looking to lose a significant amount of weight,” she concluded.
Dr. Hoff and Dr. Kahan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Combining minimally invasive endoscopic sleeve gastroplasty with a weekly injection of the glucagonlike peptide–1 agonist semaglutide (Ozempic, Novo Nordisk) leads to significantly greater weight loss than ESG alone in patients with diabetes and excess weight who are not candidates for bariatric surgery, new research shows.
During minimally invasive ESG, a flexible endoscope equipped with an endoscopic suturing device is inserted down the esophagus and into the stomach. The endoscopist then applies the sutures to the upper portion of the stomach, minimizing its size to restrict the amount of food a patient can ingest.
“Our stomachs can stretch back a bit, but we can use the suturing device again,” explained the lead investigator of the research Anna Carolina Hoff, MD, founder and clinical director of Angioskope Brazil in São José dos Campos.
“It’s important that patients with diabetes lose as much weight as possible because, if they lose about 10% of their total body weight, they have a great improvement in their glycemic levels, and some patients can even stop taking their [antidiabetic] medications,” Dr. Hoff said in an interview.
“And we found that by adding the GLP-1 agonist [semaglutide], we could increase weight loss from, on average, about 16%-18% of total body weight with ESG alone to up to 27%, so it’s a great metabolic combination,” she noted.
Dr. Hoff presented the findings at the annual Digestive Disease Week® (DDW).
Asked to comment, Scott Kahan, MD, MPH, director, National Center for Weight and Wellness, George Washington University, Washington, cautioned that it’s still early days for minimally invasive ESG.
“It is reasonable to assume that the long-term outcomes [with ESG] won’t be as good or durable over time as with bariatric surgery, but ... we will have to see.”
However, “we know that, typically, combinations of therapeutic options work better than a one-off option, so I think the real benefit of this study – outside the specific procedure and this specific medication – is that it is a very valuable proof-of-principle study showing that combinations do work better,” Dr. Kahan said in an interview.
Minimally invasive endoscopic sleeve gastroplasty
ESG is a surrogate for laparoscopic sleeve gastrectomy that can offer the benefits of such a procedure to those who don’t qualify for, or don’t wish to pursue, bariatric surgery. It can be performed at an earlier stage of disease, in those with a body mass index of 30 mg/kg2, whereas generally people are not offered bariatric procedures unless they have a BMI of at least 35 with comorbidities or a BMI of at least 40 if they do not have comorbidities.
Subcutaneous semaglutide is already approved for the treatment of type 2 diabetes in adults at doses of up to 1 mg/week; higher doses are needed for weight loss. Novo Nordisk has been investigating higher doses for weight loss in the STEP trial program, which is now complete, and the company has submitted the data to the Food and Drug Administration and European Medicines Agency for an additional indication of adults with obesity (BMI ≥30) or who are overweight (BMI ≥27) and who have at least one weight-related comorbidity, as an adjunct to a reduced-calorie diet and increased physical activity, with a decision expected soon.
Novo Nordisk has also developed an oral form of semaglutide, which has been approved as a once-daily agent for type 2 diabetes (Rybelsus) in doses of 7 mg and 14 mg to improve glycemic control along with diet and exercise. It is the first GLP-1 agonist available in tablet form.
Patients lost fat mass as well as excess weight
The Brazilian study involved 58 patients with obesity or overweight who also had diabetes and were undergoing minimally invasive ESG; they were further randomized to receive semaglutide or placebo.
The GLP-1 agonist (or sham placebo) was initiated 1 month after participants had undergone the procedure and patients were monitored each month for weight loss and type of fat loss achieved with the combination versus ESG alone. The initial dose of semaglutide used was 0.25 mg subcutaneous a week but could be titrated up to a maximum dose of 1.5 mg.
At the end of 11 months of active treatment versus placebo (12 months after ESG), patients who received additional semaglutide lost 86.3% of their excess body weight – the amount of weight patients needed to lose to reach normal BMI – compared with only 60.4% for ESG controls.
Specifically, the mean percentage total body weight loss at the end of 12 months was 25.2% for those in the combination group, compared with 18.6% for those treated with ESG alone (P < .001).
More importantly, patients in the combination group lost 12.6% of their body fat mass, compared with 9% for ESG controls, while mean A1c levels fell more in those treated with additional semaglutide compared with controls (P = .0394).
Indeed, five patients in the combination group reverted to a nondiabetic state and were able to discontinue antidiabetic medications altogether, Dr. Hoff noted.
“Our main goal is not just to lose weight but to lose body mass fat, which is very different from just losing weight,” she explained.
If patients lose weight but still maintain a high percentage of body fat mass, they have what she refers to as “sarcopenic obesity” because in this state patients have lost a lot of muscle mass but still have high levels of metabolically active visceral fat. Among many other inflammatory complexes, metabolically active visceral fat contains a large number of inflammasomes, and it is the latter that have been associated with obesity-related cancers.
“Obesity is a progressive disease, so what we are trying to do here is buy time for patients so they do not progress to [bariatric] surgery, and this approach gives patients a chance to act earlier before obesity takes over and more metabolic consequences occur,” Dr. Hoff emphasized.
So, when combined with semaglutide, “we now have a minimally invasive procedure that can be just as successful [as surgery] and which can be made available to even more people looking to lose a significant amount of weight,” she concluded.
Dr. Hoff and Dr. Kahan have reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Liver transplant outcomes improving for U.S. patients with HIV/HCV
While liver transplant outcomes were historically poor in people coinfected with HIV and hepatitis C virus (HCV), they have improved significantly in the era of direct-acting antiviral (DAA) therapy, a recent analysis of U.S. organ transplant data showed.
The availability of highly potent DAA therapy should change how transplant specialists view patients coinfected with HIV/HCV who need a liver transplant, according to researcher Jennifer Wang, MD, chief gastroenterology fellow at the University of Chicago, who presented the results of the analysis at the annual Digestive Disease Week® (DDW). Cumulative graft survival rates since the introduction of DAAs are comparable between transplant recipients with HIV/HCV coinfection and recipients who are both HIV and HCV negative, according to the study.
“Having hepatitis C no longer confers worse patient survival in the DAA era, and this is the main takeaway from our study,” Dr. Wang said.
The study also showed that the number of liver transplants among HIV-infected patients has increased over the past 4-5 years. However, the absolute number remains low at 64 cases in 2019, or less than 1% of all liver transplants that year, and only about one-third of those HIV-positive recipients had HCV coinfection, according to Dr. Wang.
Moreover, relatively few centers are performing liver transplants for patients who are HIV/HCV coinfected, and there is significant geographic variation in where the procedures are done, she said in her presentation.
Reassuring data that should prompt referral
Taken together, these results should offer reassurance to transplant centers that patients coinfected with HIV/HCV are no longer at increased risk for poor outcomes after transplantation, said Christine M. Durand, MD, associate professor of medicine at Johns Hopkins University, Baltimore.
“The additional call for action should be beyond the transplantation community to ensure that referrals for liver transplant are where they should be,” Dr. Durand said in an interview.
“With a number of only 64 transplants a year, we’re not doing enough, and there are more patients that could benefit from liver transplants,” added Dr. Durand, who is principal investigator of HOPE in Action, a prospective, multicenter, clinical trial evaluating the safety and survival outcomes of HIV-positive deceased donor liver transplants in HIV-positive recipients.
Impact of the HOPE Act
Liver transplantation for HIV-positive patients has increased since the signing of the HIV Organ Policy Equity (HOPE) Act in 2013, according to Dr. Wang.
The HOPE act expanded the donor pool to include HIV-positive deceased donors, which not only increased the donor supply overall, but specifically helped HIV-positive individuals, who experience a higher rate of waiting-list mortality, according to a review on the topic authored by Dr. Durand and coauthors.
However, some transplant centers may be reluctant to do liver transplants in HIV-positive patients coinfected with HCV. That’s because, in previous studies that were conducted before the DAA era, outcomes after liver transplant in HIV/HCV-coinfected patients were inferior to those in patients with HIV but no HCV infection, Dr. Wang said.
Accordingly, Dr. Wang and colleagues analyzed Organ Procurement and Transplantation Network (OPTN) data on adult patients who underwent liver transplants between 2008 and 2019 to see if the introduction of DAAs had leveled the playing field for those with HCV coinfection.
Progress in a still-underserved population
The practice of liver transplant in the HIV population has been increasing since the HOPE Act, according to Dr. Wang.
Overall, out of 70,125 liver transplant recipients over the 2008-2019 period, 416 (0.6%) were HIV infected, the data show.
In 2014, 28 liver transplants (0.5%) were performed in HIV-infected individuals, which increased to 64 transplants (0.8%) in 2019, data show. Of those 64 HIV-positive liver transplant recipients in 2019, 23 (35.9%) were coinfected with HCV.
Graft survival has greatly improved, from a 3-year survival of only 58% in patients transplanted before the availability of DAAs to 82% in the DAA era, a difference that was statistically significant, Dr. Wang said.
In the DAA era, there was no significant difference in graft failure outcomes when comparing HIV/HCV-coinfected recipients with uninfected recipients, she added.
The largest proportion of liver transplantations in HIV/HCV-coinfected recipients have been done in OPTN Region 9 (New York), both in the pre- and post-DAA eras, according to Dr. Wang. Several regions have very low numbers or have performed no liver transplants in HIV/HCV-coinfected patients in either era.
“The number of transplant centers participating in liver transplant for coinfected patients is still quite low, so this is a very underserved patient population,” Dr. Wang said.
Dr. Wang provided no financial disclosures related to the research. Dr. Durand receives grants to the institution from Abbvie and GlaxoSmithKline and she receives honoraria from Gilead Sciences for serving on a grant review committee.
While liver transplant outcomes were historically poor in people coinfected with HIV and hepatitis C virus (HCV), they have improved significantly in the era of direct-acting antiviral (DAA) therapy, a recent analysis of U.S. organ transplant data showed.
The availability of highly potent DAA therapy should change how transplant specialists view patients coinfected with HIV/HCV who need a liver transplant, according to researcher Jennifer Wang, MD, chief gastroenterology fellow at the University of Chicago, who presented the results of the analysis at the annual Digestive Disease Week® (DDW). Cumulative graft survival rates since the introduction of DAAs are comparable between transplant recipients with HIV/HCV coinfection and recipients who are both HIV and HCV negative, according to the study.
“Having hepatitis C no longer confers worse patient survival in the DAA era, and this is the main takeaway from our study,” Dr. Wang said.
The study also showed that the number of liver transplants among HIV-infected patients has increased over the past 4-5 years. However, the absolute number remains low at 64 cases in 2019, or less than 1% of all liver transplants that year, and only about one-third of those HIV-positive recipients had HCV coinfection, according to Dr. Wang.
Moreover, relatively few centers are performing liver transplants for patients who are HIV/HCV coinfected, and there is significant geographic variation in where the procedures are done, she said in her presentation.
Reassuring data that should prompt referral
Taken together, these results should offer reassurance to transplant centers that patients coinfected with HIV/HCV are no longer at increased risk for poor outcomes after transplantation, said Christine M. Durand, MD, associate professor of medicine at Johns Hopkins University, Baltimore.
“The additional call for action should be beyond the transplantation community to ensure that referrals for liver transplant are where they should be,” Dr. Durand said in an interview.
“With a number of only 64 transplants a year, we’re not doing enough, and there are more patients that could benefit from liver transplants,” added Dr. Durand, who is principal investigator of HOPE in Action, a prospective, multicenter, clinical trial evaluating the safety and survival outcomes of HIV-positive deceased donor liver transplants in HIV-positive recipients.
Impact of the HOPE Act
Liver transplantation for HIV-positive patients has increased since the signing of the HIV Organ Policy Equity (HOPE) Act in 2013, according to Dr. Wang.
The HOPE act expanded the donor pool to include HIV-positive deceased donors, which not only increased the donor supply overall, but specifically helped HIV-positive individuals, who experience a higher rate of waiting-list mortality, according to a review on the topic authored by Dr. Durand and coauthors.
However, some transplant centers may be reluctant to do liver transplants in HIV-positive patients coinfected with HCV. That’s because, in previous studies that were conducted before the DAA era, outcomes after liver transplant in HIV/HCV-coinfected patients were inferior to those in patients with HIV but no HCV infection, Dr. Wang said.
Accordingly, Dr. Wang and colleagues analyzed Organ Procurement and Transplantation Network (OPTN) data on adult patients who underwent liver transplants between 2008 and 2019 to see if the introduction of DAAs had leveled the playing field for those with HCV coinfection.
Progress in a still-underserved population
The practice of liver transplant in the HIV population has been increasing since the HOPE Act, according to Dr. Wang.
Overall, out of 70,125 liver transplant recipients over the 2008-2019 period, 416 (0.6%) were HIV infected, the data show.
In 2014, 28 liver transplants (0.5%) were performed in HIV-infected individuals, which increased to 64 transplants (0.8%) in 2019, data show. Of those 64 HIV-positive liver transplant recipients in 2019, 23 (35.9%) were coinfected with HCV.
Graft survival has greatly improved, from a 3-year survival of only 58% in patients transplanted before the availability of DAAs to 82% in the DAA era, a difference that was statistically significant, Dr. Wang said.
In the DAA era, there was no significant difference in graft failure outcomes when comparing HIV/HCV-coinfected recipients with uninfected recipients, she added.
The largest proportion of liver transplantations in HIV/HCV-coinfected recipients have been done in OPTN Region 9 (New York), both in the pre- and post-DAA eras, according to Dr. Wang. Several regions have very low numbers or have performed no liver transplants in HIV/HCV-coinfected patients in either era.
“The number of transplant centers participating in liver transplant for coinfected patients is still quite low, so this is a very underserved patient population,” Dr. Wang said.
Dr. Wang provided no financial disclosures related to the research. Dr. Durand receives grants to the institution from Abbvie and GlaxoSmithKline and she receives honoraria from Gilead Sciences for serving on a grant review committee.
While liver transplant outcomes were historically poor in people coinfected with HIV and hepatitis C virus (HCV), they have improved significantly in the era of direct-acting antiviral (DAA) therapy, a recent analysis of U.S. organ transplant data showed.
The availability of highly potent DAA therapy should change how transplant specialists view patients coinfected with HIV/HCV who need a liver transplant, according to researcher Jennifer Wang, MD, chief gastroenterology fellow at the University of Chicago, who presented the results of the analysis at the annual Digestive Disease Week® (DDW). Cumulative graft survival rates since the introduction of DAAs are comparable between transplant recipients with HIV/HCV coinfection and recipients who are both HIV and HCV negative, according to the study.
“Having hepatitis C no longer confers worse patient survival in the DAA era, and this is the main takeaway from our study,” Dr. Wang said.
The study also showed that the number of liver transplants among HIV-infected patients has increased over the past 4-5 years. However, the absolute number remains low at 64 cases in 2019, or less than 1% of all liver transplants that year, and only about one-third of those HIV-positive recipients had HCV coinfection, according to Dr. Wang.
Moreover, relatively few centers are performing liver transplants for patients who are HIV/HCV coinfected, and there is significant geographic variation in where the procedures are done, she said in her presentation.
Reassuring data that should prompt referral
Taken together, these results should offer reassurance to transplant centers that patients coinfected with HIV/HCV are no longer at increased risk for poor outcomes after transplantation, said Christine M. Durand, MD, associate professor of medicine at Johns Hopkins University, Baltimore.
“The additional call for action should be beyond the transplantation community to ensure that referrals for liver transplant are where they should be,” Dr. Durand said in an interview.
“With a number of only 64 transplants a year, we’re not doing enough, and there are more patients that could benefit from liver transplants,” added Dr. Durand, who is principal investigator of HOPE in Action, a prospective, multicenter, clinical trial evaluating the safety and survival outcomes of HIV-positive deceased donor liver transplants in HIV-positive recipients.
Impact of the HOPE Act
Liver transplantation for HIV-positive patients has increased since the signing of the HIV Organ Policy Equity (HOPE) Act in 2013, according to Dr. Wang.
The HOPE act expanded the donor pool to include HIV-positive deceased donors, which not only increased the donor supply overall, but specifically helped HIV-positive individuals, who experience a higher rate of waiting-list mortality, according to a review on the topic authored by Dr. Durand and coauthors.
However, some transplant centers may be reluctant to do liver transplants in HIV-positive patients coinfected with HCV. That’s because, in previous studies that were conducted before the DAA era, outcomes after liver transplant in HIV/HCV-coinfected patients were inferior to those in patients with HIV but no HCV infection, Dr. Wang said.
Accordingly, Dr. Wang and colleagues analyzed Organ Procurement and Transplantation Network (OPTN) data on adult patients who underwent liver transplants between 2008 and 2019 to see if the introduction of DAAs had leveled the playing field for those with HCV coinfection.
Progress in a still-underserved population
The practice of liver transplant in the HIV population has been increasing since the HOPE Act, according to Dr. Wang.
Overall, out of 70,125 liver transplant recipients over the 2008-2019 period, 416 (0.6%) were HIV infected, the data show.
In 2014, 28 liver transplants (0.5%) were performed in HIV-infected individuals, which increased to 64 transplants (0.8%) in 2019, data show. Of those 64 HIV-positive liver transplant recipients in 2019, 23 (35.9%) were coinfected with HCV.
Graft survival has greatly improved, from a 3-year survival of only 58% in patients transplanted before the availability of DAAs to 82% in the DAA era, a difference that was statistically significant, Dr. Wang said.
In the DAA era, there was no significant difference in graft failure outcomes when comparing HIV/HCV-coinfected recipients with uninfected recipients, she added.
The largest proportion of liver transplantations in HIV/HCV-coinfected recipients have been done in OPTN Region 9 (New York), both in the pre- and post-DAA eras, according to Dr. Wang. Several regions have very low numbers or have performed no liver transplants in HIV/HCV-coinfected patients in either era.
“The number of transplant centers participating in liver transplant for coinfected patients is still quite low, so this is a very underserved patient population,” Dr. Wang said.
Dr. Wang provided no financial disclosures related to the research. Dr. Durand receives grants to the institution from Abbvie and GlaxoSmithKline and she receives honoraria from Gilead Sciences for serving on a grant review committee.
FROM DDW 2021
Obstructive sleep apnea linked to COVID-19 risk
Greater severity of obstructive sleep apnea (OSA) is associated with a higher risk of contracting COVID-19, and positive airway pressure (PAP) treatment may counter that risk, according to a retrospective analysis from the records of Kaiser Permanente Southern California.
OSA patients often worry that PAP therapy might increase risk of severe COVID-19, said Dennis Hwang, MD, who presented the study at the American Thoracic Society’s virtual international conference (Abstract A1108). But the findings should be reassuring. “If you have obstructive sleep apnea, and you’re supposed to be using PAP, we recommend that you continue using PAP. It’s good for your overall wellness and reducing the risk of cardiovascular disease, but as it relates to COVID-19, it’s possible that it could protect. And there doesn’t appear to be any risk of increased severity of illness (with use of PAP),” Dr. Hwang said in an interview. He is medical director of sleep medicine for Kaiser Permanente San Bernardino County and cochair of sleep medicine for Kaiser Southern California.
He noted that the retrospective nature of the study makes it difficult to pin down whether PAP therapy is truly protective, “but I think there’s enough that we’ve been able conceptually to understand, to suggest that a direct causative relationship is possible,” said Dr. Hwang.
The results may imply that OSA patients should pay special attention to their OSA when there’s concern about exposure to an infectious agent like SARS-CoV-2. “The intermittent hypoxia at night, which can linger over to the day as increased sympathetic activity, increased heart rate. All of these are stresses to the body. So if you’re going to get infected, you want to start at a healthier level. You want to eliminate your sleep apnea to help reduce your risk of morbidity,” said Esra Tasali, MD, who was asked to comment on the study. Dr. Tasali is associate professor of medicine at the University of Chicago, and director of the Sleep Research Center there.
During the Q&A session after the talk, audience members asked about the timing of PAP use during COVID-19 infection, for example how often it was used during the asymptomatic phase of infection and if PAP has a positive effect. The data were not available, but “I think that the way to go is to understand this chronology,” said Dr. Tasali.
The researchers examined records between 2015 and 2020, using sleep study data, remotely collected daily PAP data, and electronic health records, all from Kaiser Permanente Southern California. Included subjects were adults who had enrolled before Feb. 1, 2020, and had sleep diagnostic or PAP data on record by March 1, 2020. The researchers analyzed PAP adherence between March 1, 2020, and the time of COVID-19 diagnosis, or until the study ended on July 31, 2020.
Patients were defined as being untreated (< 2 hours/night PAP), moderately treated (2-3.9 hours/night), or well treated (4 or more hours/night). Apnea hypopnea index (AHI) was used to determine severity. The analysis included 81,932 patients (39.8% were women, mean age was 54.0 years, 9.9% were Black, and 34.5% were Hispanic). A total of 1.7% of subjects without OSA experienced COVID-19 infection, compared to 1.8% with OSA; 0.3% with OSA were hospitalized and 0.07% underwent intensive care or died.
There were some differences between the two groups. The non-USA population was younger (mean age 47.0 vs. 54.5 years), was less likely to be men (44% vs. 60.3%), had a lower mean body mass index (30.4 vs. 34.3), had fewer comorbidities according to the Charleston Comorbidity Index (1.3 vs. 2.0), and were less likely to have hypertension (5.6% vs. 12.4%; P < .0001 for all).
Infection rates were higher in patients with more severe OSA. The rates in untreated mild, moderate, and severe OSA were 2%, 2%, and 2.4% respectively. The rate among all treated patients was 1.4% (P < .0001). Infection rates also dropped among patients with better treatment: untreated, 2.1%; moderately treated, 1.7%; and well treated, 1.3% (P < .0001).
Not having OSA was associated with a lower infection risk than was having OSA (odds ratio [OR], 0.82; 95% confidence interval, 0.70-0.96). Compared to untreated patients, there was lower infection risk in the moderately treated (OR, 0.82; 95% CI, 0.65-1.03) and well treated (OR, 0.68; 95% CI, 0.59-0.79) groups. Higher infection rates were associated with obesity, higher Charlson Comorbidity score (> 2; OR, 1.29; 95% CI, 1.09-1.53), Black (OR, 1.51; 95% CI, 1.24-1.84) and Hispanic ethnicities (OR, 2.23; 95% CI, 1.96-2.54), and Medicaid enrollment. Increasing age was associated with lower risk of infection, with each 5-year increment linked to reduced risk (OR, 0.88; 95% CI, 0.86-0.90). Dr. Hwang suggested that the age association may be because older individuals were more likely to follow social distancing and other precautions.
A multivariate analysis found that OSA was associated with infection risk according to OSA severity, including mild (OR, 1.21; 95% CI, 1.01-1.44), and moderate to severe (OR, 1.27; 95% CI, 1.07-1.51). There was no association between hospitalization rate or ICU admission/death and presence of OSA or PAP adherence in the data presented, but Dr. Hwang said that an updated analysis suggests that OSA may be associated with a risk of greater COVID-19 severity.
The control group was composed of individuals who had undergone sleep testing, but found to not have OSA. Still, they aren’t necessarily representative of the general population, since symptoms likely drove them to testing. A high percentage were also obese, and the average BMI was 30. “It’s certainly not a ‘normal population,’ but the advantage of what we did in terms of using this control group is that they underwent sleep testing, so they were proven to have no obstructive sleep apnea, whereas if we used a general population, we just don’t know,” said Dr. Hwang.
The study received technical and data support from Somnoware, and was funded by Kaiser Permanente. Dr. Tasali has no relevant financial disclosures.
Greater severity of obstructive sleep apnea (OSA) is associated with a higher risk of contracting COVID-19, and positive airway pressure (PAP) treatment may counter that risk, according to a retrospective analysis from the records of Kaiser Permanente Southern California.
OSA patients often worry that PAP therapy might increase risk of severe COVID-19, said Dennis Hwang, MD, who presented the study at the American Thoracic Society’s virtual international conference (Abstract A1108). But the findings should be reassuring. “If you have obstructive sleep apnea, and you’re supposed to be using PAP, we recommend that you continue using PAP. It’s good for your overall wellness and reducing the risk of cardiovascular disease, but as it relates to COVID-19, it’s possible that it could protect. And there doesn’t appear to be any risk of increased severity of illness (with use of PAP),” Dr. Hwang said in an interview. He is medical director of sleep medicine for Kaiser Permanente San Bernardino County and cochair of sleep medicine for Kaiser Southern California.
He noted that the retrospective nature of the study makes it difficult to pin down whether PAP therapy is truly protective, “but I think there’s enough that we’ve been able conceptually to understand, to suggest that a direct causative relationship is possible,” said Dr. Hwang.
The results may imply that OSA patients should pay special attention to their OSA when there’s concern about exposure to an infectious agent like SARS-CoV-2. “The intermittent hypoxia at night, which can linger over to the day as increased sympathetic activity, increased heart rate. All of these are stresses to the body. So if you’re going to get infected, you want to start at a healthier level. You want to eliminate your sleep apnea to help reduce your risk of morbidity,” said Esra Tasali, MD, who was asked to comment on the study. Dr. Tasali is associate professor of medicine at the University of Chicago, and director of the Sleep Research Center there.
During the Q&A session after the talk, audience members asked about the timing of PAP use during COVID-19 infection, for example how often it was used during the asymptomatic phase of infection and if PAP has a positive effect. The data were not available, but “I think that the way to go is to understand this chronology,” said Dr. Tasali.
The researchers examined records between 2015 and 2020, using sleep study data, remotely collected daily PAP data, and electronic health records, all from Kaiser Permanente Southern California. Included subjects were adults who had enrolled before Feb. 1, 2020, and had sleep diagnostic or PAP data on record by March 1, 2020. The researchers analyzed PAP adherence between March 1, 2020, and the time of COVID-19 diagnosis, or until the study ended on July 31, 2020.
Patients were defined as being untreated (< 2 hours/night PAP), moderately treated (2-3.9 hours/night), or well treated (4 or more hours/night). Apnea hypopnea index (AHI) was used to determine severity. The analysis included 81,932 patients (39.8% were women, mean age was 54.0 years, 9.9% were Black, and 34.5% were Hispanic). A total of 1.7% of subjects without OSA experienced COVID-19 infection, compared to 1.8% with OSA; 0.3% with OSA were hospitalized and 0.07% underwent intensive care or died.
There were some differences between the two groups. The non-USA population was younger (mean age 47.0 vs. 54.5 years), was less likely to be men (44% vs. 60.3%), had a lower mean body mass index (30.4 vs. 34.3), had fewer comorbidities according to the Charleston Comorbidity Index (1.3 vs. 2.0), and were less likely to have hypertension (5.6% vs. 12.4%; P < .0001 for all).
Infection rates were higher in patients with more severe OSA. The rates in untreated mild, moderate, and severe OSA were 2%, 2%, and 2.4% respectively. The rate among all treated patients was 1.4% (P < .0001). Infection rates also dropped among patients with better treatment: untreated, 2.1%; moderately treated, 1.7%; and well treated, 1.3% (P < .0001).
Not having OSA was associated with a lower infection risk than was having OSA (odds ratio [OR], 0.82; 95% confidence interval, 0.70-0.96). Compared to untreated patients, there was lower infection risk in the moderately treated (OR, 0.82; 95% CI, 0.65-1.03) and well treated (OR, 0.68; 95% CI, 0.59-0.79) groups. Higher infection rates were associated with obesity, higher Charlson Comorbidity score (> 2; OR, 1.29; 95% CI, 1.09-1.53), Black (OR, 1.51; 95% CI, 1.24-1.84) and Hispanic ethnicities (OR, 2.23; 95% CI, 1.96-2.54), and Medicaid enrollment. Increasing age was associated with lower risk of infection, with each 5-year increment linked to reduced risk (OR, 0.88; 95% CI, 0.86-0.90). Dr. Hwang suggested that the age association may be because older individuals were more likely to follow social distancing and other precautions.
A multivariate analysis found that OSA was associated with infection risk according to OSA severity, including mild (OR, 1.21; 95% CI, 1.01-1.44), and moderate to severe (OR, 1.27; 95% CI, 1.07-1.51). There was no association between hospitalization rate or ICU admission/death and presence of OSA or PAP adherence in the data presented, but Dr. Hwang said that an updated analysis suggests that OSA may be associated with a risk of greater COVID-19 severity.
The control group was composed of individuals who had undergone sleep testing, but found to not have OSA. Still, they aren’t necessarily representative of the general population, since symptoms likely drove them to testing. A high percentage were also obese, and the average BMI was 30. “It’s certainly not a ‘normal population,’ but the advantage of what we did in terms of using this control group is that they underwent sleep testing, so they were proven to have no obstructive sleep apnea, whereas if we used a general population, we just don’t know,” said Dr. Hwang.
The study received technical and data support from Somnoware, and was funded by Kaiser Permanente. Dr. Tasali has no relevant financial disclosures.
Greater severity of obstructive sleep apnea (OSA) is associated with a higher risk of contracting COVID-19, and positive airway pressure (PAP) treatment may counter that risk, according to a retrospective analysis from the records of Kaiser Permanente Southern California.
OSA patients often worry that PAP therapy might increase risk of severe COVID-19, said Dennis Hwang, MD, who presented the study at the American Thoracic Society’s virtual international conference (Abstract A1108). But the findings should be reassuring. “If you have obstructive sleep apnea, and you’re supposed to be using PAP, we recommend that you continue using PAP. It’s good for your overall wellness and reducing the risk of cardiovascular disease, but as it relates to COVID-19, it’s possible that it could protect. And there doesn’t appear to be any risk of increased severity of illness (with use of PAP),” Dr. Hwang said in an interview. He is medical director of sleep medicine for Kaiser Permanente San Bernardino County and cochair of sleep medicine for Kaiser Southern California.
He noted that the retrospective nature of the study makes it difficult to pin down whether PAP therapy is truly protective, “but I think there’s enough that we’ve been able conceptually to understand, to suggest that a direct causative relationship is possible,” said Dr. Hwang.
The results may imply that OSA patients should pay special attention to their OSA when there’s concern about exposure to an infectious agent like SARS-CoV-2. “The intermittent hypoxia at night, which can linger over to the day as increased sympathetic activity, increased heart rate. All of these are stresses to the body. So if you’re going to get infected, you want to start at a healthier level. You want to eliminate your sleep apnea to help reduce your risk of morbidity,” said Esra Tasali, MD, who was asked to comment on the study. Dr. Tasali is associate professor of medicine at the University of Chicago, and director of the Sleep Research Center there.
During the Q&A session after the talk, audience members asked about the timing of PAP use during COVID-19 infection, for example how often it was used during the asymptomatic phase of infection and if PAP has a positive effect. The data were not available, but “I think that the way to go is to understand this chronology,” said Dr. Tasali.
The researchers examined records between 2015 and 2020, using sleep study data, remotely collected daily PAP data, and electronic health records, all from Kaiser Permanente Southern California. Included subjects were adults who had enrolled before Feb. 1, 2020, and had sleep diagnostic or PAP data on record by March 1, 2020. The researchers analyzed PAP adherence between March 1, 2020, and the time of COVID-19 diagnosis, or until the study ended on July 31, 2020.
Patients were defined as being untreated (< 2 hours/night PAP), moderately treated (2-3.9 hours/night), or well treated (4 or more hours/night). Apnea hypopnea index (AHI) was used to determine severity. The analysis included 81,932 patients (39.8% were women, mean age was 54.0 years, 9.9% were Black, and 34.5% were Hispanic). A total of 1.7% of subjects without OSA experienced COVID-19 infection, compared to 1.8% with OSA; 0.3% with OSA were hospitalized and 0.07% underwent intensive care or died.
There were some differences between the two groups. The non-USA population was younger (mean age 47.0 vs. 54.5 years), was less likely to be men (44% vs. 60.3%), had a lower mean body mass index (30.4 vs. 34.3), had fewer comorbidities according to the Charleston Comorbidity Index (1.3 vs. 2.0), and were less likely to have hypertension (5.6% vs. 12.4%; P < .0001 for all).
Infection rates were higher in patients with more severe OSA. The rates in untreated mild, moderate, and severe OSA were 2%, 2%, and 2.4% respectively. The rate among all treated patients was 1.4% (P < .0001). Infection rates also dropped among patients with better treatment: untreated, 2.1%; moderately treated, 1.7%; and well treated, 1.3% (P < .0001).
Not having OSA was associated with a lower infection risk than was having OSA (odds ratio [OR], 0.82; 95% confidence interval, 0.70-0.96). Compared to untreated patients, there was lower infection risk in the moderately treated (OR, 0.82; 95% CI, 0.65-1.03) and well treated (OR, 0.68; 95% CI, 0.59-0.79) groups. Higher infection rates were associated with obesity, higher Charlson Comorbidity score (> 2; OR, 1.29; 95% CI, 1.09-1.53), Black (OR, 1.51; 95% CI, 1.24-1.84) and Hispanic ethnicities (OR, 2.23; 95% CI, 1.96-2.54), and Medicaid enrollment. Increasing age was associated with lower risk of infection, with each 5-year increment linked to reduced risk (OR, 0.88; 95% CI, 0.86-0.90). Dr. Hwang suggested that the age association may be because older individuals were more likely to follow social distancing and other precautions.
A multivariate analysis found that OSA was associated with infection risk according to OSA severity, including mild (OR, 1.21; 95% CI, 1.01-1.44), and moderate to severe (OR, 1.27; 95% CI, 1.07-1.51). There was no association between hospitalization rate or ICU admission/death and presence of OSA or PAP adherence in the data presented, but Dr. Hwang said that an updated analysis suggests that OSA may be associated with a risk of greater COVID-19 severity.
The control group was composed of individuals who had undergone sleep testing, but found to not have OSA. Still, they aren’t necessarily representative of the general population, since symptoms likely drove them to testing. A high percentage were also obese, and the average BMI was 30. “It’s certainly not a ‘normal population,’ but the advantage of what we did in terms of using this control group is that they underwent sleep testing, so they were proven to have no obstructive sleep apnea, whereas if we used a general population, we just don’t know,” said Dr. Hwang.
The study received technical and data support from Somnoware, and was funded by Kaiser Permanente. Dr. Tasali has no relevant financial disclosures.
FROM ATS 2021
New AHA/ASA guideline on secondary stroke prevention
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
When possible, diagnostic tests to determine the cause of a first stroke or transient ischemic attack (TIA) should be completed within 48 hours after symptom onset, the American Heart Association/American Stroke Association said in an updated clinical practice guideline.
“It is critically important to understand the best ways to prevent another stroke once someone has had a stroke or a TIA,” Dawn O. Kleindorfer, MD, chair of the guideline writing group, said in a news release.
“If we can pinpoint the cause of the first stroke or TIA, we can tailor strategies to prevent a second stroke,” said Dr. Kleindorfer, professor and chair, department of neurology, University of Michigan, Ann Arbor.
The updated guideline was published online May 24, 2021, in Stroke.
“The secondary prevention of stroke guideline is one of the ASA’s ‘flagship’ guidelines, last updated in 2014,” Dr. Kleindorfer said.
The update includes “a number of changes to the writing and formatting of this guideline to make it easier for professionals to understand and locate information more quickly, ultimately greatly improving patient care and preventing more strokes in our patients,” she noted.
Let pathogenic subtype guide prevention
For patients who have survived a stroke or TIA, management of vascular risk factors, particularly hypertension, diabetes, cholesterol/triglyceride levels, and smoking cessation, are key secondary prevention tactics, the guideline said.
Limiting salt intake and/or following a heart-healthy Mediterranean diet is also advised, as is engaging in at least moderate-intensity aerobic activity for at least 10 minutes four times a week or vigorous-intensity aerobic activity for at least 20 minutes twice a week.
“Approximately 80% of strokes can be prevented by controlling blood pressure, eating a healthy diet, engaging in regular physical activity, not smoking and maintaining a healthy weight,” Amytis Towfighi, MD, vice chair of the guideline writing group and director of neurologic services, Los Angeles County Department of Health Services, noted in the release.
For health care professionals, the guideline said specific recommendations for secondary prevention often depend on the ischemic stroke/TIA subtype. “Therefore, new in this guideline is a section describing recommendations for the diagnostic workup after ischemic stroke, to define ischemic stroke pathogenesis (when possible), and to identify targets for treatment to reduce the risk of recurrent ischemic stroke. Recommendations are now segregated by pathogenetic subtype,” the guideline stated.
Among the recommendations:
- Use multidisciplinary care teams to personalize care for patients and employ shared decision-making with the patient to develop care plans that incorporate a patient’s wishes, goals, and concerns.
- Screen for and initiate anticoagulant drug therapy to reduce recurrent events.
- Prescribe antithrombotic therapy, including antiplatelets or anticoagulants, in the absence of contraindications. The guideline noted that the combination of antiplatelets and anticoagulation is typically not recommended for preventing second strokes and that dual antiplatelet therapy (DAPT) – taking along with a second medication to prevent blood clotting – is recommended in the short term and only for specific patients: those with early arriving minor stroke and high-risk TIA or severe symptomatic stenosis.
- Consider or carotid artery stenting for select patients with narrowing of carotid arteries.
- Aggressive medical management of risk factors and short-term DAPT are preferred for patients with severe intracranial stenosis thought to be the cause of first stroke or TIA.
- In some patients, it’s reasonable to consider percutaneous closure of .
The guideline is accompanied by a systematic review and meta-analysis regarding the benefits and risks of dual antiplatelet versus single antiplatelet therapy for secondary stroke prevention. The authors conclude that DAPT may be appropriate for select patients.
“Additional research is needed to determine: the optimal timing of starting treatment relative to the clinical event; the optimal duration of DAPT to maximize the risk-benefit ratio; whether additional populations excluded from POINT and CHANCE [two of the trials examined], such as those with major stroke, may also benefit from early DAPT; and whether certain genetic profiles eliminate the benefit of early DAPT,” concluded the reviewers, led by Devin Brown, MD, University of Michigan.
The guideline was prepared on behalf of and approved by the AHA Stroke Council’s Scientific Statements Oversight Committee on Clinical Practice Guidelines. The writing group included representatives from the AHA/ASA and the American Academy of Neurology. The guideline has been endorsed by the American Association of Neurological Surgeons/Congress of Neurological Surgeons and the Society of Vascular and Interventional Neurology. It has also been affirmed by the AAN as an educational tool for neurologists.
The research had no commercial funding.
A version of this article first appeared on Medscape.com.
FDA restricts obeticholic acid (Ocaliva) over serious liver injury risk
The risk for serious liver injury with obeticholic acid (Ocaliva, Intercept Pharmaceuticals) has prompted the U.S. Food and Drug Administration to restrict its use in patients with primary biliary cholangitis (PBC) and advanced cirrhosis.
The agency has added a new contraindication to the obeticholic acid prescribing information and patient medication guide stating that the drug should not be used in patients with PBC and advanced cirrhosis.
The boxed warning on the label has also been revised to include this information.
For patients with PBC who do not have advanced cirrhosis, the FDA believes the benefits of Ocaliva outweigh the risks, based on the original clinical trials.
Five years ago, the FDA granted accelerated approval to obeticholic acid in combination with ursodeoxycholic acid (UDCA) in adults who fail to respond adequately to UDCA, or as a monotherapy in adults who cannot tolerate UDCA.
Since then, the FDA has identified 25 cases of serious liver injury leading to liver decompensation or liver failure in patients with PBC and cirrhosis who were taking obeticholic acid at recommended doses.
According to the FDA, 18 of the cases happened in patients with PBC and compensated cirrhosis who experienced liver injury that led to decompensation. Ten of these patients had evidence or suspicion of portal hypertension at baseline; in the other eight patients, it was unclear whether portal hypertension was present.
PBC was not expected to progress rapidly in these patients, yet they experienced accelerated clinical deterioration within months of starting obeticholic acid, the FDA said.
The median time to liver decompensation after initiating treatment was 4 months (range, 2 weeks to 10 months). Four patients with PBC and compensated cirrhosis needed a liver transplant within 1.3 years after starting obeticholic acid, and one patient died from liver failure.
The other seven cases of serious liver injury occurred in patients with PBC and decompensated cirrhosis, two of whom died.
Although there was a temporal relationship between starting obeticholic acid and liver injury, it is difficult to distinguish a drug-induced effect from disease progression in the patients with advanced baseline liver disease, the FDA cautioned.
The median time to a new decompensation event after starting the drug was 2.5 months (range, 10 days to 8 months).
Before starting obeticholic acid, clinicians should determine whether a patient with PBC has advanced cirrhosis as the drug is now contraindicated in these patients, the FDA said.
During obeticholic acid treatment, patients should be routinely monitored for progression of PBC with laboratory and clinical assessments to determine whether the drug needs to be discontinued.
The medication should be permanently discontinued in patients with cirrhosis who progress to advanced cirrhosis.
Patients should also be monitored for clinically significant liver-related adverse reactions that may manifest as development of acute-on-chronic liver disease with nausea, vomiting, diarrhea, jaundice, scleral icterus, and/or dark urine.
Obeticholic acid should be stopped permanently in any patient who develops these symptoms, the FDA advised.
Health care professionals are encouraged to report adverse events or side effects related to the use of obeticholic acid to MedWatch, FDA’s adverse event reporting site.
A version of this article first appeared on Medscape.com.
The risk for serious liver injury with obeticholic acid (Ocaliva, Intercept Pharmaceuticals) has prompted the U.S. Food and Drug Administration to restrict its use in patients with primary biliary cholangitis (PBC) and advanced cirrhosis.
The agency has added a new contraindication to the obeticholic acid prescribing information and patient medication guide stating that the drug should not be used in patients with PBC and advanced cirrhosis.
The boxed warning on the label has also been revised to include this information.
For patients with PBC who do not have advanced cirrhosis, the FDA believes the benefits of Ocaliva outweigh the risks, based on the original clinical trials.
Five years ago, the FDA granted accelerated approval to obeticholic acid in combination with ursodeoxycholic acid (UDCA) in adults who fail to respond adequately to UDCA, or as a monotherapy in adults who cannot tolerate UDCA.
Since then, the FDA has identified 25 cases of serious liver injury leading to liver decompensation or liver failure in patients with PBC and cirrhosis who were taking obeticholic acid at recommended doses.
According to the FDA, 18 of the cases happened in patients with PBC and compensated cirrhosis who experienced liver injury that led to decompensation. Ten of these patients had evidence or suspicion of portal hypertension at baseline; in the other eight patients, it was unclear whether portal hypertension was present.
PBC was not expected to progress rapidly in these patients, yet they experienced accelerated clinical deterioration within months of starting obeticholic acid, the FDA said.
The median time to liver decompensation after initiating treatment was 4 months (range, 2 weeks to 10 months). Four patients with PBC and compensated cirrhosis needed a liver transplant within 1.3 years after starting obeticholic acid, and one patient died from liver failure.
The other seven cases of serious liver injury occurred in patients with PBC and decompensated cirrhosis, two of whom died.
Although there was a temporal relationship between starting obeticholic acid and liver injury, it is difficult to distinguish a drug-induced effect from disease progression in the patients with advanced baseline liver disease, the FDA cautioned.
The median time to a new decompensation event after starting the drug was 2.5 months (range, 10 days to 8 months).
Before starting obeticholic acid, clinicians should determine whether a patient with PBC has advanced cirrhosis as the drug is now contraindicated in these patients, the FDA said.
During obeticholic acid treatment, patients should be routinely monitored for progression of PBC with laboratory and clinical assessments to determine whether the drug needs to be discontinued.
The medication should be permanently discontinued in patients with cirrhosis who progress to advanced cirrhosis.
Patients should also be monitored for clinically significant liver-related adverse reactions that may manifest as development of acute-on-chronic liver disease with nausea, vomiting, diarrhea, jaundice, scleral icterus, and/or dark urine.
Obeticholic acid should be stopped permanently in any patient who develops these symptoms, the FDA advised.
Health care professionals are encouraged to report adverse events or side effects related to the use of obeticholic acid to MedWatch, FDA’s adverse event reporting site.
A version of this article first appeared on Medscape.com.
The risk for serious liver injury with obeticholic acid (Ocaliva, Intercept Pharmaceuticals) has prompted the U.S. Food and Drug Administration to restrict its use in patients with primary biliary cholangitis (PBC) and advanced cirrhosis.
The agency has added a new contraindication to the obeticholic acid prescribing information and patient medication guide stating that the drug should not be used in patients with PBC and advanced cirrhosis.
The boxed warning on the label has also been revised to include this information.
For patients with PBC who do not have advanced cirrhosis, the FDA believes the benefits of Ocaliva outweigh the risks, based on the original clinical trials.
Five years ago, the FDA granted accelerated approval to obeticholic acid in combination with ursodeoxycholic acid (UDCA) in adults who fail to respond adequately to UDCA, or as a monotherapy in adults who cannot tolerate UDCA.
Since then, the FDA has identified 25 cases of serious liver injury leading to liver decompensation or liver failure in patients with PBC and cirrhosis who were taking obeticholic acid at recommended doses.
According to the FDA, 18 of the cases happened in patients with PBC and compensated cirrhosis who experienced liver injury that led to decompensation. Ten of these patients had evidence or suspicion of portal hypertension at baseline; in the other eight patients, it was unclear whether portal hypertension was present.
PBC was not expected to progress rapidly in these patients, yet they experienced accelerated clinical deterioration within months of starting obeticholic acid, the FDA said.
The median time to liver decompensation after initiating treatment was 4 months (range, 2 weeks to 10 months). Four patients with PBC and compensated cirrhosis needed a liver transplant within 1.3 years after starting obeticholic acid, and one patient died from liver failure.
The other seven cases of serious liver injury occurred in patients with PBC and decompensated cirrhosis, two of whom died.
Although there was a temporal relationship between starting obeticholic acid and liver injury, it is difficult to distinguish a drug-induced effect from disease progression in the patients with advanced baseline liver disease, the FDA cautioned.
The median time to a new decompensation event after starting the drug was 2.5 months (range, 10 days to 8 months).
Before starting obeticholic acid, clinicians should determine whether a patient with PBC has advanced cirrhosis as the drug is now contraindicated in these patients, the FDA said.
During obeticholic acid treatment, patients should be routinely monitored for progression of PBC with laboratory and clinical assessments to determine whether the drug needs to be discontinued.
The medication should be permanently discontinued in patients with cirrhosis who progress to advanced cirrhosis.
Patients should also be monitored for clinically significant liver-related adverse reactions that may manifest as development of acute-on-chronic liver disease with nausea, vomiting, diarrhea, jaundice, scleral icterus, and/or dark urine.
Obeticholic acid should be stopped permanently in any patient who develops these symptoms, the FDA advised.
Health care professionals are encouraged to report adverse events or side effects related to the use of obeticholic acid to MedWatch, FDA’s adverse event reporting site.
A version of this article first appeared on Medscape.com.
‘Overbasalization’ common in type 2 diabetes management
that impedes achievement of optimal glycemic control, new research suggests.
Such ‘overbasalization,’ defined as a hemoglobin A1c of greater than 8% despite use of more than 0.5 units/kg per day of basal insulin, was identified in about 40% of patients seen in a Florida primary care clinic during 2015-2018. The findings were published in the April 2021 issue of Clinical Diabetes by Kevin Cowart, PharmD, a diabetes care and education specialist at the University of South Florida, Tampa, and colleagues.
The literature suggests that once people with type 2 diabetes start basal insulin, the chance that they’ll achieve a given hemoglobin A1c target, i.e., less than 7%, diminishes significantly if that goal isn’t achieved within the first year of starting insulin, Dr. Cowart said in an interview.
“Our analysis suggests that overbasalization plays a role in patients with type 2 diabetes on basal insulin not achieving optimal glycemic control. Basal insulin is not designed to address postprandial hyperglycemia. I think there’s a clear need to address hesitancy in therapeutic progression beyond basal insulin. A lot of factors underlie the delays, with therapeutic inertia being one of them. It’s complex,” he said.
Overbasalization seen in large proportion of patients
The study comprised 655 adults diagnosed with type 2 diabetes for at least a year who received a prescription for a basal insulin (glargine U-100, glargine U-300, detemir, degludec U-100, degludec U-200, regular U-500, or NPH insulin).
The patients had a mean hemoglobin A1c of 8.4% and a mean basal insulin dose 0.4 units/kg per day. The prevalence of overbasalization was 38.1% for those with hemoglobin A1c above 8%, 42.7% for those with A1c of 9% or above, and 42% with A1c of 10% or greater.
Patient characteristics independently associated with overbasalization were age 35-54 years (odds ratio 1.89), age 65-80 years (0.44), A1c 9% or greater (13.97), and A1c 10% or greater (6.04). Having a prescription for insulin glargine U-100 was associated with a lower overbasalization risk (0.62). In multivariate analysis, only an A1c of 9% or greater remained significant.
Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic, Rochester, Minn., said in an interview that she sees [overbasalization] frequently in patients who are referred to her. “It’s kind of that wall that patients with type 2 diabetes hit because their A1c is high but their fasting blood sugars are normal. Sometimes it’s assumed that there’s a discrepancy, because people don’t always think about postprandial hyperglycemia.”
She also noted that there has been a push in recent years to simplify regimens, particularly in older patients.
“We really want to avoid rapid-acting insulin in older patients because we’re afraid of hypoglycemia, so we start them on basal and keep the noninsulins like metformin and sulfonylureas around. Initially those control the postprandial blood sugar but over time they’re no longer enough.”
Options exist for addressing postmeal blood sugar highs while minimizing lows
While in the past adding premeal insulin was the only option, today there are alternatives for addressing postmeal hyperglycemia, at least in the short term.
Dr. Cowart advised that the first step is to have patients self-monitor their blood glucose and titrate their basal insulin to address fasting hyperglycemia first. Once that appropriate dose is reached, if the patient’s hemoglobin A1c is still above target, the next step is to evaluate the need for postmeal control.
For patients who are at high cardiovascular risk, the next step might involve adding a sodium-glucose cotransporter 2 inhibitor (SGLT2i) or a glucagon-like peptide 1 receptor agonist (GLP-1RA) instead of premeal insulin. But for patients in whom overbasalization is the main concern, a GLP-1RA might be the better choice since it will have a greater impact on postprandial glucose levels, while an SGLT2i will have more effect on fasting blood sugar, he said.
Another option is to use a fixed-dose combination of basal insulin and a glucagon-like peptide 1 receptor agonist (GLP-1RA), provided there aren’t cost or formulary barriers. “We want to use the right combination of drugs and not use too much of one to lead to hypoglycemia,” Dr. Cowart said.
Dr. McCoy doesn’t use fixed-dose combinations because they don’t allow as much flexibility in dosing. To correct overbasalization, she also recommends adding either a GLP-1RA or SGLT2i instead of premeal insulin. However, she cautions, “you still have to monitor those patients because after a few years it still won’t be enough and you’ll have to add mealtime insulin.”
If cost or lack of coverage prevents a patient’s use of SLGT2i/GLP-1RAs, Dr. McCoy said that adding just one premeal injection of rapid-acting insulin before the largest meal of the day is one option. Another is to use twice-daily NPH insulin instead of analog basal insulin, since that does offer some postprandial coverage.
Dr. Cowart said his approach in cost barrier situations is to try to use patient assistance programs and to look into the patient’s formulary to see if there is step therapy or tier considerations, and maybe have a discussion with the insurance company. “We often have to navigate that, and it does take a significant amount of time and could potentially delay patients getting the right therapy when it’s warranted. That is an area where there is a particular role for pharmacists in helping to overcome that and get patients on the right drugs,” he explained.
Problem may be even more common; testing is key
Dr. McCoy said that the A1c cutoff of 8% used to define overbasalization in the study probably resulted in an underestimation of the problem, since many patients are experiencing nighttime hypoglycemia from the basal insulin. The lows bring down their A1c level, but they’re still experiencing postmeal highs.
“I think they’re missing a lot of people, to be honest. I see a lot of patients with A1cs that aren’t that bad, say 7.5%, and their fasting blood sugars are okay, but if you were to put a [continuous glucose monitor] on those patients, invariably there’s hypoglycemia at night that no one knew about.”
Of course, for insurance reasons, most people with type 2 diabetes don’t currently have access to continuous glucose monitors. And often those who are not taking multiple daily injections are limited to one fingerstick test strip a day.
Dr. McCoy says that if hypoglycemia is a concern she will write a prior authorization justifying more test strips.
“I state explicitly in my notes why I recommend frequent monitoring. If they’re on a sulfonylurea, they should be able to check more frequently because they can have hypoglycemia. Same thing with basal insulin.”
Dr. McCoy advises that patients test their blood sugar 2 hours after the largest meal on one day, and at other times on different days. “Blood glucose after a meal shouldn’t be more than 200 [mg/dL]. If it is, that’s not a failure of basal insulin. It’s doing its job. You just need a different agent.”
Dr. Cowart has no disclosures. Dr. McCoy receives funding from the National Institutes of Health.
that impedes achievement of optimal glycemic control, new research suggests.
Such ‘overbasalization,’ defined as a hemoglobin A1c of greater than 8% despite use of more than 0.5 units/kg per day of basal insulin, was identified in about 40% of patients seen in a Florida primary care clinic during 2015-2018. The findings were published in the April 2021 issue of Clinical Diabetes by Kevin Cowart, PharmD, a diabetes care and education specialist at the University of South Florida, Tampa, and colleagues.
The literature suggests that once people with type 2 diabetes start basal insulin, the chance that they’ll achieve a given hemoglobin A1c target, i.e., less than 7%, diminishes significantly if that goal isn’t achieved within the first year of starting insulin, Dr. Cowart said in an interview.
“Our analysis suggests that overbasalization plays a role in patients with type 2 diabetes on basal insulin not achieving optimal glycemic control. Basal insulin is not designed to address postprandial hyperglycemia. I think there’s a clear need to address hesitancy in therapeutic progression beyond basal insulin. A lot of factors underlie the delays, with therapeutic inertia being one of them. It’s complex,” he said.
Overbasalization seen in large proportion of patients
The study comprised 655 adults diagnosed with type 2 diabetes for at least a year who received a prescription for a basal insulin (glargine U-100, glargine U-300, detemir, degludec U-100, degludec U-200, regular U-500, or NPH insulin).
The patients had a mean hemoglobin A1c of 8.4% and a mean basal insulin dose 0.4 units/kg per day. The prevalence of overbasalization was 38.1% for those with hemoglobin A1c above 8%, 42.7% for those with A1c of 9% or above, and 42% with A1c of 10% or greater.
Patient characteristics independently associated with overbasalization were age 35-54 years (odds ratio 1.89), age 65-80 years (0.44), A1c 9% or greater (13.97), and A1c 10% or greater (6.04). Having a prescription for insulin glargine U-100 was associated with a lower overbasalization risk (0.62). In multivariate analysis, only an A1c of 9% or greater remained significant.
Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic, Rochester, Minn., said in an interview that she sees [overbasalization] frequently in patients who are referred to her. “It’s kind of that wall that patients with type 2 diabetes hit because their A1c is high but their fasting blood sugars are normal. Sometimes it’s assumed that there’s a discrepancy, because people don’t always think about postprandial hyperglycemia.”
She also noted that there has been a push in recent years to simplify regimens, particularly in older patients.
“We really want to avoid rapid-acting insulin in older patients because we’re afraid of hypoglycemia, so we start them on basal and keep the noninsulins like metformin and sulfonylureas around. Initially those control the postprandial blood sugar but over time they’re no longer enough.”
Options exist for addressing postmeal blood sugar highs while minimizing lows
While in the past adding premeal insulin was the only option, today there are alternatives for addressing postmeal hyperglycemia, at least in the short term.
Dr. Cowart advised that the first step is to have patients self-monitor their blood glucose and titrate their basal insulin to address fasting hyperglycemia first. Once that appropriate dose is reached, if the patient’s hemoglobin A1c is still above target, the next step is to evaluate the need for postmeal control.
For patients who are at high cardiovascular risk, the next step might involve adding a sodium-glucose cotransporter 2 inhibitor (SGLT2i) or a glucagon-like peptide 1 receptor agonist (GLP-1RA) instead of premeal insulin. But for patients in whom overbasalization is the main concern, a GLP-1RA might be the better choice since it will have a greater impact on postprandial glucose levels, while an SGLT2i will have more effect on fasting blood sugar, he said.
Another option is to use a fixed-dose combination of basal insulin and a glucagon-like peptide 1 receptor agonist (GLP-1RA), provided there aren’t cost or formulary barriers. “We want to use the right combination of drugs and not use too much of one to lead to hypoglycemia,” Dr. Cowart said.
Dr. McCoy doesn’t use fixed-dose combinations because they don’t allow as much flexibility in dosing. To correct overbasalization, she also recommends adding either a GLP-1RA or SGLT2i instead of premeal insulin. However, she cautions, “you still have to monitor those patients because after a few years it still won’t be enough and you’ll have to add mealtime insulin.”
If cost or lack of coverage prevents a patient’s use of SLGT2i/GLP-1RAs, Dr. McCoy said that adding just one premeal injection of rapid-acting insulin before the largest meal of the day is one option. Another is to use twice-daily NPH insulin instead of analog basal insulin, since that does offer some postprandial coverage.
Dr. Cowart said his approach in cost barrier situations is to try to use patient assistance programs and to look into the patient’s formulary to see if there is step therapy or tier considerations, and maybe have a discussion with the insurance company. “We often have to navigate that, and it does take a significant amount of time and could potentially delay patients getting the right therapy when it’s warranted. That is an area where there is a particular role for pharmacists in helping to overcome that and get patients on the right drugs,” he explained.
Problem may be even more common; testing is key
Dr. McCoy said that the A1c cutoff of 8% used to define overbasalization in the study probably resulted in an underestimation of the problem, since many patients are experiencing nighttime hypoglycemia from the basal insulin. The lows bring down their A1c level, but they’re still experiencing postmeal highs.
“I think they’re missing a lot of people, to be honest. I see a lot of patients with A1cs that aren’t that bad, say 7.5%, and their fasting blood sugars are okay, but if you were to put a [continuous glucose monitor] on those patients, invariably there’s hypoglycemia at night that no one knew about.”
Of course, for insurance reasons, most people with type 2 diabetes don’t currently have access to continuous glucose monitors. And often those who are not taking multiple daily injections are limited to one fingerstick test strip a day.
Dr. McCoy says that if hypoglycemia is a concern she will write a prior authorization justifying more test strips.
“I state explicitly in my notes why I recommend frequent monitoring. If they’re on a sulfonylurea, they should be able to check more frequently because they can have hypoglycemia. Same thing with basal insulin.”
Dr. McCoy advises that patients test their blood sugar 2 hours after the largest meal on one day, and at other times on different days. “Blood glucose after a meal shouldn’t be more than 200 [mg/dL]. If it is, that’s not a failure of basal insulin. It’s doing its job. You just need a different agent.”
Dr. Cowart has no disclosures. Dr. McCoy receives funding from the National Institutes of Health.
that impedes achievement of optimal glycemic control, new research suggests.
Such ‘overbasalization,’ defined as a hemoglobin A1c of greater than 8% despite use of more than 0.5 units/kg per day of basal insulin, was identified in about 40% of patients seen in a Florida primary care clinic during 2015-2018. The findings were published in the April 2021 issue of Clinical Diabetes by Kevin Cowart, PharmD, a diabetes care and education specialist at the University of South Florida, Tampa, and colleagues.
The literature suggests that once people with type 2 diabetes start basal insulin, the chance that they’ll achieve a given hemoglobin A1c target, i.e., less than 7%, diminishes significantly if that goal isn’t achieved within the first year of starting insulin, Dr. Cowart said in an interview.
“Our analysis suggests that overbasalization plays a role in patients with type 2 diabetes on basal insulin not achieving optimal glycemic control. Basal insulin is not designed to address postprandial hyperglycemia. I think there’s a clear need to address hesitancy in therapeutic progression beyond basal insulin. A lot of factors underlie the delays, with therapeutic inertia being one of them. It’s complex,” he said.
Overbasalization seen in large proportion of patients
The study comprised 655 adults diagnosed with type 2 diabetes for at least a year who received a prescription for a basal insulin (glargine U-100, glargine U-300, detemir, degludec U-100, degludec U-200, regular U-500, or NPH insulin).
The patients had a mean hemoglobin A1c of 8.4% and a mean basal insulin dose 0.4 units/kg per day. The prevalence of overbasalization was 38.1% for those with hemoglobin A1c above 8%, 42.7% for those with A1c of 9% or above, and 42% with A1c of 10% or greater.
Patient characteristics independently associated with overbasalization were age 35-54 years (odds ratio 1.89), age 65-80 years (0.44), A1c 9% or greater (13.97), and A1c 10% or greater (6.04). Having a prescription for insulin glargine U-100 was associated with a lower overbasalization risk (0.62). In multivariate analysis, only an A1c of 9% or greater remained significant.
Rozalina G. McCoy, MD, an endocrinologist and primary care clinician at the Mayo Clinic, Rochester, Minn., said in an interview that she sees [overbasalization] frequently in patients who are referred to her. “It’s kind of that wall that patients with type 2 diabetes hit because their A1c is high but their fasting blood sugars are normal. Sometimes it’s assumed that there’s a discrepancy, because people don’t always think about postprandial hyperglycemia.”
She also noted that there has been a push in recent years to simplify regimens, particularly in older patients.
“We really want to avoid rapid-acting insulin in older patients because we’re afraid of hypoglycemia, so we start them on basal and keep the noninsulins like metformin and sulfonylureas around. Initially those control the postprandial blood sugar but over time they’re no longer enough.”
Options exist for addressing postmeal blood sugar highs while minimizing lows
While in the past adding premeal insulin was the only option, today there are alternatives for addressing postmeal hyperglycemia, at least in the short term.
Dr. Cowart advised that the first step is to have patients self-monitor their blood glucose and titrate their basal insulin to address fasting hyperglycemia first. Once that appropriate dose is reached, if the patient’s hemoglobin A1c is still above target, the next step is to evaluate the need for postmeal control.
For patients who are at high cardiovascular risk, the next step might involve adding a sodium-glucose cotransporter 2 inhibitor (SGLT2i) or a glucagon-like peptide 1 receptor agonist (GLP-1RA) instead of premeal insulin. But for patients in whom overbasalization is the main concern, a GLP-1RA might be the better choice since it will have a greater impact on postprandial glucose levels, while an SGLT2i will have more effect on fasting blood sugar, he said.
Another option is to use a fixed-dose combination of basal insulin and a glucagon-like peptide 1 receptor agonist (GLP-1RA), provided there aren’t cost or formulary barriers. “We want to use the right combination of drugs and not use too much of one to lead to hypoglycemia,” Dr. Cowart said.
Dr. McCoy doesn’t use fixed-dose combinations because they don’t allow as much flexibility in dosing. To correct overbasalization, she also recommends adding either a GLP-1RA or SGLT2i instead of premeal insulin. However, she cautions, “you still have to monitor those patients because after a few years it still won’t be enough and you’ll have to add mealtime insulin.”
If cost or lack of coverage prevents a patient’s use of SLGT2i/GLP-1RAs, Dr. McCoy said that adding just one premeal injection of rapid-acting insulin before the largest meal of the day is one option. Another is to use twice-daily NPH insulin instead of analog basal insulin, since that does offer some postprandial coverage.
Dr. Cowart said his approach in cost barrier situations is to try to use patient assistance programs and to look into the patient’s formulary to see if there is step therapy or tier considerations, and maybe have a discussion with the insurance company. “We often have to navigate that, and it does take a significant amount of time and could potentially delay patients getting the right therapy when it’s warranted. That is an area where there is a particular role for pharmacists in helping to overcome that and get patients on the right drugs,” he explained.
Problem may be even more common; testing is key
Dr. McCoy said that the A1c cutoff of 8% used to define overbasalization in the study probably resulted in an underestimation of the problem, since many patients are experiencing nighttime hypoglycemia from the basal insulin. The lows bring down their A1c level, but they’re still experiencing postmeal highs.
“I think they’re missing a lot of people, to be honest. I see a lot of patients with A1cs that aren’t that bad, say 7.5%, and their fasting blood sugars are okay, but if you were to put a [continuous glucose monitor] on those patients, invariably there’s hypoglycemia at night that no one knew about.”
Of course, for insurance reasons, most people with type 2 diabetes don’t currently have access to continuous glucose monitors. And often those who are not taking multiple daily injections are limited to one fingerstick test strip a day.
Dr. McCoy says that if hypoglycemia is a concern she will write a prior authorization justifying more test strips.
“I state explicitly in my notes why I recommend frequent monitoring. If they’re on a sulfonylurea, they should be able to check more frequently because they can have hypoglycemia. Same thing with basal insulin.”
Dr. McCoy advises that patients test their blood sugar 2 hours after the largest meal on one day, and at other times on different days. “Blood glucose after a meal shouldn’t be more than 200 [mg/dL]. If it is, that’s not a failure of basal insulin. It’s doing its job. You just need a different agent.”
Dr. Cowart has no disclosures. Dr. McCoy receives funding from the National Institutes of Health.
FROM CLINICAL DIABETES
Scaly patches on hand and feet
A potassium hydroxide (KOH) mount of skin scrapings from the patient’s feet and hand confirmed a diagnosis of unilateral tinea manuum and bilateral tinea pedis—the so-called 2-foot, 1-hand syndrome. Additionally, nail clippings from the patient’s right hand confirmed onychomycosis.
Tinea is common and caused by various dermatophytes that are ubiquitous in soil. Often there is a history of atopic dermatitis or xerosis leading to skin barrier dysfunction. Immunosuppression and diabetes mellitus are also predisposing factors. Trichophyton rubrum is a commonly isolated cause.
On the hands, tinea may be challenging to distinguish from irritant dermatitis, atopic dermatitis, and contact dermatitis. A KOH prep test should be considered for any red, scaly rash, especially on the hands and feet. Curiously, the incidence of unilateral tinea manuum and bilateral tinea pedis occurs relatively frequently and can affect either the dominant or nondominant hand. The cause of this asymmetry is speculative.
Topical therapy with various antifungals—terbinafine, clotrimazole, ketoconazole, ciclopirox—can be effective, but challenging to apply to affected areas. For the treatment of nail disease, oral therapy with terbinafine or itraconazole is usually indicated (6 weeks for fingernails and 12 weeks for toenails). Terbinafine is generally tolerated very well for both 6- and 12-week courses. Some clinicians consider lab monitoring unnecessary because the risk of hepatic injury from terbinafine is uncertain; others consider it worthwhile to check liver function test results prior to initiation of terbinafine and after 6 weeks of therapy, with either a 6- or 12-week course.1
Since the patient in this case had skin and nail disease, oral therapy with terbinafine 250 mg/d for 6 weeks was prescribed. His skin cleared within 3 weeks and his nails cleared after 6 months. It is important to counsel patients with nail disease that treatment will end before they see a clinical improvement. Fingernails typically require 6 months to see clearance and toenails require 18 months.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
1. Stolmeier DA, Stratman HB, McIntee TJ, et al. Utility of laboratory test result monitoring in patients taking oral terbinafine or griseofulvin for dermatophyte infections. JAMA Dermatol. 2018;154:1409-1416. doi:10.1001/jamadermatol.2018.3578

A potassium hydroxide (KOH) mount of skin scrapings from the patient’s feet and hand confirmed a diagnosis of unilateral tinea manuum and bilateral tinea pedis—the so-called 2-foot, 1-hand syndrome. Additionally, nail clippings from the patient’s right hand confirmed onychomycosis.
Tinea is common and caused by various dermatophytes that are ubiquitous in soil. Often there is a history of atopic dermatitis or xerosis leading to skin barrier dysfunction. Immunosuppression and diabetes mellitus are also predisposing factors. Trichophyton rubrum is a commonly isolated cause.
On the hands, tinea may be challenging to distinguish from irritant dermatitis, atopic dermatitis, and contact dermatitis. A KOH prep test should be considered for any red, scaly rash, especially on the hands and feet. Curiously, the incidence of unilateral tinea manuum and bilateral tinea pedis occurs relatively frequently and can affect either the dominant or nondominant hand. The cause of this asymmetry is speculative.
Topical therapy with various antifungals—terbinafine, clotrimazole, ketoconazole, ciclopirox—can be effective, but challenging to apply to affected areas. For the treatment of nail disease, oral therapy with terbinafine or itraconazole is usually indicated (6 weeks for fingernails and 12 weeks for toenails). Terbinafine is generally tolerated very well for both 6- and 12-week courses. Some clinicians consider lab monitoring unnecessary because the risk of hepatic injury from terbinafine is uncertain; others consider it worthwhile to check liver function test results prior to initiation of terbinafine and after 6 weeks of therapy, with either a 6- or 12-week course.1
Since the patient in this case had skin and nail disease, oral therapy with terbinafine 250 mg/d for 6 weeks was prescribed. His skin cleared within 3 weeks and his nails cleared after 6 months. It is important to counsel patients with nail disease that treatment will end before they see a clinical improvement. Fingernails typically require 6 months to see clearance and toenails require 18 months.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)

A potassium hydroxide (KOH) mount of skin scrapings from the patient’s feet and hand confirmed a diagnosis of unilateral tinea manuum and bilateral tinea pedis—the so-called 2-foot, 1-hand syndrome. Additionally, nail clippings from the patient’s right hand confirmed onychomycosis.
Tinea is common and caused by various dermatophytes that are ubiquitous in soil. Often there is a history of atopic dermatitis or xerosis leading to skin barrier dysfunction. Immunosuppression and diabetes mellitus are also predisposing factors. Trichophyton rubrum is a commonly isolated cause.
On the hands, tinea may be challenging to distinguish from irritant dermatitis, atopic dermatitis, and contact dermatitis. A KOH prep test should be considered for any red, scaly rash, especially on the hands and feet. Curiously, the incidence of unilateral tinea manuum and bilateral tinea pedis occurs relatively frequently and can affect either the dominant or nondominant hand. The cause of this asymmetry is speculative.
Topical therapy with various antifungals—terbinafine, clotrimazole, ketoconazole, ciclopirox—can be effective, but challenging to apply to affected areas. For the treatment of nail disease, oral therapy with terbinafine or itraconazole is usually indicated (6 weeks for fingernails and 12 weeks for toenails). Terbinafine is generally tolerated very well for both 6- and 12-week courses. Some clinicians consider lab monitoring unnecessary because the risk of hepatic injury from terbinafine is uncertain; others consider it worthwhile to check liver function test results prior to initiation of terbinafine and after 6 weeks of therapy, with either a 6- or 12-week course.1
Since the patient in this case had skin and nail disease, oral therapy with terbinafine 250 mg/d for 6 weeks was prescribed. His skin cleared within 3 weeks and his nails cleared after 6 months. It is important to counsel patients with nail disease that treatment will end before they see a clinical improvement. Fingernails typically require 6 months to see clearance and toenails require 18 months.
Text and photos courtesy of Jonathan Karnes, MD, medical director, MDFMR Dermatology Services, Augusta, ME. (Photo copyright retained.)
1. Stolmeier DA, Stratman HB, McIntee TJ, et al. Utility of laboratory test result monitoring in patients taking oral terbinafine or griseofulvin for dermatophyte infections. JAMA Dermatol. 2018;154:1409-1416. doi:10.1001/jamadermatol.2018.3578
1. Stolmeier DA, Stratman HB, McIntee TJ, et al. Utility of laboratory test result monitoring in patients taking oral terbinafine or griseofulvin for dermatophyte infections. JAMA Dermatol. 2018;154:1409-1416. doi:10.1001/jamadermatol.2018.3578