User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Health care in America: Let that tapeworm grow
In my most recent column, “ ‘They All Laughed When I Spoke of Greedy Doctors,’ ” I attempted to provide a global understanding of some of the economic forces that have made American medicine what it is, how that happened, and why it is still happening.
I did not propose a fix. I have been proposing fixes for more than 30 years, on the pages of JAMA until 1999 and then for this news organization, most recently in 2019 with “Healthcare for All in a Land of Special Interests.”
Where you stand depends a lot on where you sit.
Is this good news or bad news? When William Hubbard was the dean of the University of Michigan School of Medicine in 1969, he said that “an academic medical center is the most efficient energy and resource trapping device that has ever been created” (personal communication, 1969).
To me as a faculty member of an academic medical center for many years, that was great news. We could grow faculty, erect buildings, take the best care of sick people, churn out research papers, mint new physicians and specialists, and get paid well in the process for doing “the Lord’s work.” What’s not to like? At that time, the proportion of the country’s gross national product expended for medical and health care was about 7%. And the predicted life span of an American at birth was 70.5 years.
Is this good news or bad news? In 2021, the proportion of our annual gross domestic product (GDP) consumed by health care was 18.3%, totaling $4.3 trillion, or $12,914 per person. For perspective, in 2021, the median income per capita was $37,638. Because quite a few Americans have very high incomes, the mean income per capita is much higher: $63,444. Predicted life span in 2021 was 76.4 years.
Thus, in a span of 53 years (1969-2022), only 5.9 years of life were gained per person born, for how many trillions of dollars expended? To me as a tax-paying citizen and payer of medical insurance premiums, that is bad news.
Is this good news or bad news? If we compare developed societies globally, our medical system does a whole lot of things very well indeed. But we spend a great deal more than any other country for health care and objectively achieve poorer outcomes. Thus, we are neither efficient nor effective. We keep a lot of workers very busy doing stuff, and they are generally well paid. As a worker, that’s good news; as a manager who values efficiency, it’s bad news indeed.
Is this good news or bad news? We’re the leader at finding money to pay people to do “health care work.” More Americans work in health care than any other field. In 2019, the United States employed some 21,000,000 people doing “health care and social assistance.” Among others, these occupations include physicians, dentists, dental hygienists and assistants, pharmacists, registered nurses, LVNs/LPNs, nursing aides, technologists and technicians, home health aides, respiratory therapists, occupational and speech therapists, social workers, childcare workers, and personal and home care aides. For a patient, parent, grandparent, and great-grandparent, it is good news to have all those folks available to take care of us when we need it.
So, while I have cringed at the frequent exposés from Roy Poses of what seem to me to be massive societal betrayals by American health care industry giants, it doesn’t have to be that way. Might it still be possible to do well while doing good?
A jobs program
Consider such common medical procedures as coronary artery stents or bypass grafts for stable angina (when optimal medical therapy is as good, or better than, and much less expensive); PSAs on asymptomatic men followed by unnecessary surgery for localized cancer; excess surgery for low back pain; and the jobs created by managing the people caught up in medical complications of the obesity epidemic.
Don’t forget the number of people employed simply to “follow the money” within our byzantine cockamamie medical billing system. In 2009, this prompted me to describe the bloated system as a “health care bubble” not unlike Enron, the submarket real estate financing debacle, or the dot-com boom and bust. I warned of the downside of bursting that bubble, particularly lost jobs.
The Affordable Care Act (ACA) provided health insurance to some 35 million Americans who had been uninsured. It retarded health care inflation. But it did nothing to trim administrative costs or very high pay for nonclinical executives, or shareholder profits in those companies that were for-profit, or drug and device prices. Without the support of all those groups, the ACA would never have passed Congress. The ACA has clearly been a mixed blessing.
If any large American constituency were ever serious about reducing the percentage of our GDP expended on health care, we have excellent ways to do that while improving the health and well-being of the American people. But remember, one person’s liability (unnecessary work) is another person’s asset (needed job).
The MBAization of medicine
Meanwhile, back at Dean Hubbard’s voracious academic medical center, the high intellect and driven nature of those who are attracted to medicine as a career has had other effects. The resulting organizations reflect not only the glorious calling of caring for the sick and the availability of lots of money to recruit and compensate leaders, but also the necessity to develop strong executive types who won’t be “eaten alive” by the high-powered workforce of demanding physicians and the surrounding environment.
Thus, it came as no great surprise that in its 2021 determination of America’s top 25 Best Large Employers, Forbes included five health care organizations and seven universities. Beating out such giants as NASA, Cisco, Microsoft, Netflix, and Google, the University of Alabama Birmingham Hospital was ranked first. Mayo Clinic and Yale University came in third and fifth, respectively, and at the other end of the list were Duke (23), MIT (24), and MD Anderson (25).
My goodness! Well done.
Yet, as a country attempting to be balanced, Warren Buffett’s descriptive entreaty on the 2021 failure of Haven, the Amazon-Chase-Berkshire Hathaway joint initiative, remains troubling. Calling upon Haven to change the U.S. health care system, Buffet said, “We learned a lot about the difficulty of changing around an industry that’s 17% of the GDP. We were fighting a tapeworm in the American economy, and the tapeworm won.” They had failed to tame the American health care cost beast.
I am on record as despising the “MBAization” of American medicine. Unfairly, I blamed a professional and technical discipline for what I considered misuse. I hereby repent and renounce my earlier condemnations.
Take it all over?
Here’s an idea: If you can’t beat them, join them.
Medical care is important, especially for acute illnesses and injuries, early cancer therapy, and many chronic conditions. But the real determinants of health writ large are social: wealth, education, housing, nutritious food, childcare, climate, clean air and water, meaningful employment, safety from violence, exercise schemes, vaccinations, and so on.
Why doesn’t the American medical-industrial complex simply bestow the label of “health care” on all health-related social determinants? Take it all over. Good “health care” jobs for everyone. Medical professionals will still be blamed for the low health quality and poor outcome scores, the main social determinants of health over which we have no control or influence.
Let that tapeworm grow to encompass all social determinants of health, and measure results by length and quality of life, national human happiness, and, of course, jobs. We can do it. Let that bubble glow. Party time.
And that’s the way it is. That’s my opinion.
George Lundberg, MD, is editor-in-chief at Cancer Commons, president of the Lundberg Institute, executive advisor at Cureus, and a clinical professor of pathology at Northwestern University. Previously, he served as editor-in-chief of JAMA (including 10 specialty journals), American Medical News, and Medscape.
A version of this article first appeared on Medscape.com.
In my most recent column, “ ‘They All Laughed When I Spoke of Greedy Doctors,’ ” I attempted to provide a global understanding of some of the economic forces that have made American medicine what it is, how that happened, and why it is still happening.
I did not propose a fix. I have been proposing fixes for more than 30 years, on the pages of JAMA until 1999 and then for this news organization, most recently in 2019 with “Healthcare for All in a Land of Special Interests.”
Where you stand depends a lot on where you sit.
Is this good news or bad news? When William Hubbard was the dean of the University of Michigan School of Medicine in 1969, he said that “an academic medical center is the most efficient energy and resource trapping device that has ever been created” (personal communication, 1969).
To me as a faculty member of an academic medical center for many years, that was great news. We could grow faculty, erect buildings, take the best care of sick people, churn out research papers, mint new physicians and specialists, and get paid well in the process for doing “the Lord’s work.” What’s not to like? At that time, the proportion of the country’s gross national product expended for medical and health care was about 7%. And the predicted life span of an American at birth was 70.5 years.
Is this good news or bad news? In 2021, the proportion of our annual gross domestic product (GDP) consumed by health care was 18.3%, totaling $4.3 trillion, or $12,914 per person. For perspective, in 2021, the median income per capita was $37,638. Because quite a few Americans have very high incomes, the mean income per capita is much higher: $63,444. Predicted life span in 2021 was 76.4 years.
Thus, in a span of 53 years (1969-2022), only 5.9 years of life were gained per person born, for how many trillions of dollars expended? To me as a tax-paying citizen and payer of medical insurance premiums, that is bad news.
Is this good news or bad news? If we compare developed societies globally, our medical system does a whole lot of things very well indeed. But we spend a great deal more than any other country for health care and objectively achieve poorer outcomes. Thus, we are neither efficient nor effective. We keep a lot of workers very busy doing stuff, and they are generally well paid. As a worker, that’s good news; as a manager who values efficiency, it’s bad news indeed.
Is this good news or bad news? We’re the leader at finding money to pay people to do “health care work.” More Americans work in health care than any other field. In 2019, the United States employed some 21,000,000 people doing “health care and social assistance.” Among others, these occupations include physicians, dentists, dental hygienists and assistants, pharmacists, registered nurses, LVNs/LPNs, nursing aides, technologists and technicians, home health aides, respiratory therapists, occupational and speech therapists, social workers, childcare workers, and personal and home care aides. For a patient, parent, grandparent, and great-grandparent, it is good news to have all those folks available to take care of us when we need it.
So, while I have cringed at the frequent exposés from Roy Poses of what seem to me to be massive societal betrayals by American health care industry giants, it doesn’t have to be that way. Might it still be possible to do well while doing good?
A jobs program
Consider such common medical procedures as coronary artery stents or bypass grafts for stable angina (when optimal medical therapy is as good, or better than, and much less expensive); PSAs on asymptomatic men followed by unnecessary surgery for localized cancer; excess surgery for low back pain; and the jobs created by managing the people caught up in medical complications of the obesity epidemic.
Don’t forget the number of people employed simply to “follow the money” within our byzantine cockamamie medical billing system. In 2009, this prompted me to describe the bloated system as a “health care bubble” not unlike Enron, the submarket real estate financing debacle, or the dot-com boom and bust. I warned of the downside of bursting that bubble, particularly lost jobs.
The Affordable Care Act (ACA) provided health insurance to some 35 million Americans who had been uninsured. It retarded health care inflation. But it did nothing to trim administrative costs or very high pay for nonclinical executives, or shareholder profits in those companies that were for-profit, or drug and device prices. Without the support of all those groups, the ACA would never have passed Congress. The ACA has clearly been a mixed blessing.
If any large American constituency were ever serious about reducing the percentage of our GDP expended on health care, we have excellent ways to do that while improving the health and well-being of the American people. But remember, one person’s liability (unnecessary work) is another person’s asset (needed job).
The MBAization of medicine
Meanwhile, back at Dean Hubbard’s voracious academic medical center, the high intellect and driven nature of those who are attracted to medicine as a career has had other effects. The resulting organizations reflect not only the glorious calling of caring for the sick and the availability of lots of money to recruit and compensate leaders, but also the necessity to develop strong executive types who won’t be “eaten alive” by the high-powered workforce of demanding physicians and the surrounding environment.
Thus, it came as no great surprise that in its 2021 determination of America’s top 25 Best Large Employers, Forbes included five health care organizations and seven universities. Beating out such giants as NASA, Cisco, Microsoft, Netflix, and Google, the University of Alabama Birmingham Hospital was ranked first. Mayo Clinic and Yale University came in third and fifth, respectively, and at the other end of the list were Duke (23), MIT (24), and MD Anderson (25).
My goodness! Well done.
Yet, as a country attempting to be balanced, Warren Buffett’s descriptive entreaty on the 2021 failure of Haven, the Amazon-Chase-Berkshire Hathaway joint initiative, remains troubling. Calling upon Haven to change the U.S. health care system, Buffet said, “We learned a lot about the difficulty of changing around an industry that’s 17% of the GDP. We were fighting a tapeworm in the American economy, and the tapeworm won.” They had failed to tame the American health care cost beast.
I am on record as despising the “MBAization” of American medicine. Unfairly, I blamed a professional and technical discipline for what I considered misuse. I hereby repent and renounce my earlier condemnations.
Take it all over?
Here’s an idea: If you can’t beat them, join them.
Medical care is important, especially for acute illnesses and injuries, early cancer therapy, and many chronic conditions. But the real determinants of health writ large are social: wealth, education, housing, nutritious food, childcare, climate, clean air and water, meaningful employment, safety from violence, exercise schemes, vaccinations, and so on.
Why doesn’t the American medical-industrial complex simply bestow the label of “health care” on all health-related social determinants? Take it all over. Good “health care” jobs for everyone. Medical professionals will still be blamed for the low health quality and poor outcome scores, the main social determinants of health over which we have no control or influence.
Let that tapeworm grow to encompass all social determinants of health, and measure results by length and quality of life, national human happiness, and, of course, jobs. We can do it. Let that bubble glow. Party time.
And that’s the way it is. That’s my opinion.
George Lundberg, MD, is editor-in-chief at Cancer Commons, president of the Lundberg Institute, executive advisor at Cureus, and a clinical professor of pathology at Northwestern University. Previously, he served as editor-in-chief of JAMA (including 10 specialty journals), American Medical News, and Medscape.
A version of this article first appeared on Medscape.com.
In my most recent column, “ ‘They All Laughed When I Spoke of Greedy Doctors,’ ” I attempted to provide a global understanding of some of the economic forces that have made American medicine what it is, how that happened, and why it is still happening.
I did not propose a fix. I have been proposing fixes for more than 30 years, on the pages of JAMA until 1999 and then for this news organization, most recently in 2019 with “Healthcare for All in a Land of Special Interests.”
Where you stand depends a lot on where you sit.
Is this good news or bad news? When William Hubbard was the dean of the University of Michigan School of Medicine in 1969, he said that “an academic medical center is the most efficient energy and resource trapping device that has ever been created” (personal communication, 1969).
To me as a faculty member of an academic medical center for many years, that was great news. We could grow faculty, erect buildings, take the best care of sick people, churn out research papers, mint new physicians and specialists, and get paid well in the process for doing “the Lord’s work.” What’s not to like? At that time, the proportion of the country’s gross national product expended for medical and health care was about 7%. And the predicted life span of an American at birth was 70.5 years.
Is this good news or bad news? In 2021, the proportion of our annual gross domestic product (GDP) consumed by health care was 18.3%, totaling $4.3 trillion, or $12,914 per person. For perspective, in 2021, the median income per capita was $37,638. Because quite a few Americans have very high incomes, the mean income per capita is much higher: $63,444. Predicted life span in 2021 was 76.4 years.
Thus, in a span of 53 years (1969-2022), only 5.9 years of life were gained per person born, for how many trillions of dollars expended? To me as a tax-paying citizen and payer of medical insurance premiums, that is bad news.
Is this good news or bad news? If we compare developed societies globally, our medical system does a whole lot of things very well indeed. But we spend a great deal more than any other country for health care and objectively achieve poorer outcomes. Thus, we are neither efficient nor effective. We keep a lot of workers very busy doing stuff, and they are generally well paid. As a worker, that’s good news; as a manager who values efficiency, it’s bad news indeed.
Is this good news or bad news? We’re the leader at finding money to pay people to do “health care work.” More Americans work in health care than any other field. In 2019, the United States employed some 21,000,000 people doing “health care and social assistance.” Among others, these occupations include physicians, dentists, dental hygienists and assistants, pharmacists, registered nurses, LVNs/LPNs, nursing aides, technologists and technicians, home health aides, respiratory therapists, occupational and speech therapists, social workers, childcare workers, and personal and home care aides. For a patient, parent, grandparent, and great-grandparent, it is good news to have all those folks available to take care of us when we need it.
So, while I have cringed at the frequent exposés from Roy Poses of what seem to me to be massive societal betrayals by American health care industry giants, it doesn’t have to be that way. Might it still be possible to do well while doing good?
A jobs program
Consider such common medical procedures as coronary artery stents or bypass grafts for stable angina (when optimal medical therapy is as good, or better than, and much less expensive); PSAs on asymptomatic men followed by unnecessary surgery for localized cancer; excess surgery for low back pain; and the jobs created by managing the people caught up in medical complications of the obesity epidemic.
Don’t forget the number of people employed simply to “follow the money” within our byzantine cockamamie medical billing system. In 2009, this prompted me to describe the bloated system as a “health care bubble” not unlike Enron, the submarket real estate financing debacle, or the dot-com boom and bust. I warned of the downside of bursting that bubble, particularly lost jobs.
The Affordable Care Act (ACA) provided health insurance to some 35 million Americans who had been uninsured. It retarded health care inflation. But it did nothing to trim administrative costs or very high pay for nonclinical executives, or shareholder profits in those companies that were for-profit, or drug and device prices. Without the support of all those groups, the ACA would never have passed Congress. The ACA has clearly been a mixed blessing.
If any large American constituency were ever serious about reducing the percentage of our GDP expended on health care, we have excellent ways to do that while improving the health and well-being of the American people. But remember, one person’s liability (unnecessary work) is another person’s asset (needed job).
The MBAization of medicine
Meanwhile, back at Dean Hubbard’s voracious academic medical center, the high intellect and driven nature of those who are attracted to medicine as a career has had other effects. The resulting organizations reflect not only the glorious calling of caring for the sick and the availability of lots of money to recruit and compensate leaders, but also the necessity to develop strong executive types who won’t be “eaten alive” by the high-powered workforce of demanding physicians and the surrounding environment.
Thus, it came as no great surprise that in its 2021 determination of America’s top 25 Best Large Employers, Forbes included five health care organizations and seven universities. Beating out such giants as NASA, Cisco, Microsoft, Netflix, and Google, the University of Alabama Birmingham Hospital was ranked first. Mayo Clinic and Yale University came in third and fifth, respectively, and at the other end of the list were Duke (23), MIT (24), and MD Anderson (25).
My goodness! Well done.
Yet, as a country attempting to be balanced, Warren Buffett’s descriptive entreaty on the 2021 failure of Haven, the Amazon-Chase-Berkshire Hathaway joint initiative, remains troubling. Calling upon Haven to change the U.S. health care system, Buffet said, “We learned a lot about the difficulty of changing around an industry that’s 17% of the GDP. We were fighting a tapeworm in the American economy, and the tapeworm won.” They had failed to tame the American health care cost beast.
I am on record as despising the “MBAization” of American medicine. Unfairly, I blamed a professional and technical discipline for what I considered misuse. I hereby repent and renounce my earlier condemnations.
Take it all over?
Here’s an idea: If you can’t beat them, join them.
Medical care is important, especially for acute illnesses and injuries, early cancer therapy, and many chronic conditions. But the real determinants of health writ large are social: wealth, education, housing, nutritious food, childcare, climate, clean air and water, meaningful employment, safety from violence, exercise schemes, vaccinations, and so on.
Why doesn’t the American medical-industrial complex simply bestow the label of “health care” on all health-related social determinants? Take it all over. Good “health care” jobs for everyone. Medical professionals will still be blamed for the low health quality and poor outcome scores, the main social determinants of health over which we have no control or influence.
Let that tapeworm grow to encompass all social determinants of health, and measure results by length and quality of life, national human happiness, and, of course, jobs. We can do it. Let that bubble glow. Party time.
And that’s the way it is. That’s my opinion.
George Lundberg, MD, is editor-in-chief at Cancer Commons, president of the Lundberg Institute, executive advisor at Cureus, and a clinical professor of pathology at Northwestern University. Previously, he served as editor-in-chief of JAMA (including 10 specialty journals), American Medical News, and Medscape.
A version of this article first appeared on Medscape.com.
Previously unknown viral families hide in the darnedest places
You and me and baby makes 10,003
If you were a virus hunter, looking for your next big virus discovery, where would you go? The wholesale seafood market in Wuhan? A gathering of unmasked anti-vaxxers in the heartland of America? The frozen snot fields of northwest Siberia?
How about babies? Well, it’s too late now, because that’s what Dennis Sandris Nielsen, PhD, of the University of Copenhagen, and his associates did, and they hit the mother lode. Actually, it was more like the infant load, if we’re being honest here.
“We found an exceptional number of unknown viruses in the faeces of these babies,” Dr. Nielsen said in a written statement from the university. (The study was published in Nature Microbiology, so we get the English spelling of feces.)
The investigators mapped the gut “viromes” of 647 healthy Danish 1-year-old children over the course of 5 years and found 10,000 species of viruses distributed across 248 different viral families, of which only 16 were already known. Incredible stuff, but then things took a turn for the cute. “The researchers named the remaining 232 unknown viral families after the children whose diapers made the study possible. As a result, new viral families include names like Sylvesterviridae, Rigmorviridae and Tristanviridae,” the university said.
About 90% of the viruses found in the feces are bacterial viruses, aka bacteriophages, which have bacteria as their hosts and don’t attack the children’s cells, so they don’t cause disease. The other 10%, however, are eukaryotic: They use human cells as hosts, so they can be either friend or foe. “It is thought-provoking that all children run around with 10-20 of these virus types that infect human cells. So, there is a constant viral infection taking place, which apparently doesn’t make them sick,” Dr. Nielsen said.
Doesn’t make them sick? Riiiight. The thought that this gives rise to now? People love babies. Everyone wants to pick up the baby. Now we know why. Because the viruses want us to! Well, those cute little faces aren’t fooling us anymore. No more babies for us. Everyone should stay away from babies and their evil little eukaryotic viruses. STOP THE BABIES!
[Editor’s note: After a short timeout, we explained to the staff that the human species actually needs babies for its survival. They calmed down, picked up their crayons, and quietly went back to work.]
Fooled them. Stop the babies!
At least someone out there appreciates hospital food
Life in Alaska is not for the meek. It’s dark half the year. Summer is 3 weeks in July. And somehow, there’s a moose in line ahead of you at the doctor’s office. To make matters worse, it’s arguing about insurance. “What do you mean, you’ve heard the Moo Cross Moo Shield joke before?”
One might expect that Providence Alaska Health Park, located near downtown Anchorage, the largest city in Alaska by a massive margin, might be safe from ungulate invasion. Nope. In recent days, a young moose has taken to hanging around Providence campus, and it just could not find anything to eat. Remember, it may be early April, but this is Alaska. It’s still winter there. The ground’s still covered in snow.
Eventually, the gears in our young moose friend’s mind turned and it settled on a course of action: “Hey, those are some nice-looking plants behind that door over there. …” And that’s how Providence Alaska Health ended up with a moose munching on decorative potted plants in the hospital lobby.
Funnily enough, the moose didn’t even make a big scene. It just walked through the automatic doors and started chowing down. Security only found out because a tenant called them. Naturally though, once security made the announcement that a massive wild animal had been spotted in the building, the lobby was evacuated. … What do you mean, half the hospital came around to see it? Apparently, even though Alaskans have to fight moose herds on their daily commute, a lot of people wanted to see our moose friend do its thing.
“That’s crazy,” a woman in scrubs said in a video as she snapped a photo with her phone.
“This is the best. Like, what’s the code for this?” asked another bystander.
Despite security’s best efforts to shoo the moose out with barricades and offers of tasty branches, our furry friend left of its own volition, presumably irritated that his breakfast had become a spectator sport. But it didn’t go far. It hung around the front drive for a while, then went around the back of the building for a nap. What has four hooves and still doesn’t give a crap? Bob Moose-o! How you doing?
That click sounded stressed
How can people tell that you’re stressed? Maybe you get irritable and a little snappy. Some people have an inability to concentrate or focus. Eating that muffin when you weren’t really hungry could be a sign you’re not relaxed.
Did you know that your computer can be an indicator of your stress levels?
We tend to be working when we’re using computers, right? That can be a stressor in itself. Well, some researchers at ETH Zürich decided to have a look at the situation. Surprisingly, at least to us, one in three Swiss employees experience workplace stress, which makes us wonder what the percentage is in this country.
The Swiss researchers developed a model that tells how stressed someone is just by the way they use their computer mouse or type. The results of their study showed that those who were stressed clicked and tapped differently than participants who were more relaxed.
Stressed people click “more often and less precisely and cover longer distances on the screen,” while the relaxed take “shorter, more direct routes to reach their destination and take more time doing so,” study author Mara Nägelin explained in a written statement from ETH (Eidgenössische Technische Hochschule, or Swiss Federal Institute of Technology) Zürich.
Ever find when you’re frustrated and in a rush you end up making more mistakes? Same deal. Coauthor Jasmine Kerr noted that “increased levels of stress negatively impact our brain’s ability to process information.” Which totally is going to affect how we move.
Hopefully, these results can give insight to companies on how stressed their employees are and the effect it has on their work performance, eventually leading to, guess what, more research on how to alleviate workplace stress in general, which can benefit us all.
So if you find yourself in the office working on your computer like it’s a game of Perfection and time is running out, take a beat. Maybe try a stress-relieving breathing technique. Nonstressed people, according to the study, take fewer and longer pauses on their computers. Perfection on the job may mean relaxing first.
You and me and baby makes 10,003
If you were a virus hunter, looking for your next big virus discovery, where would you go? The wholesale seafood market in Wuhan? A gathering of unmasked anti-vaxxers in the heartland of America? The frozen snot fields of northwest Siberia?
How about babies? Well, it’s too late now, because that’s what Dennis Sandris Nielsen, PhD, of the University of Copenhagen, and his associates did, and they hit the mother lode. Actually, it was more like the infant load, if we’re being honest here.
“We found an exceptional number of unknown viruses in the faeces of these babies,” Dr. Nielsen said in a written statement from the university. (The study was published in Nature Microbiology, so we get the English spelling of feces.)
The investigators mapped the gut “viromes” of 647 healthy Danish 1-year-old children over the course of 5 years and found 10,000 species of viruses distributed across 248 different viral families, of which only 16 were already known. Incredible stuff, but then things took a turn for the cute. “The researchers named the remaining 232 unknown viral families after the children whose diapers made the study possible. As a result, new viral families include names like Sylvesterviridae, Rigmorviridae and Tristanviridae,” the university said.
About 90% of the viruses found in the feces are bacterial viruses, aka bacteriophages, which have bacteria as their hosts and don’t attack the children’s cells, so they don’t cause disease. The other 10%, however, are eukaryotic: They use human cells as hosts, so they can be either friend or foe. “It is thought-provoking that all children run around with 10-20 of these virus types that infect human cells. So, there is a constant viral infection taking place, which apparently doesn’t make them sick,” Dr. Nielsen said.
Doesn’t make them sick? Riiiight. The thought that this gives rise to now? People love babies. Everyone wants to pick up the baby. Now we know why. Because the viruses want us to! Well, those cute little faces aren’t fooling us anymore. No more babies for us. Everyone should stay away from babies and their evil little eukaryotic viruses. STOP THE BABIES!
[Editor’s note: After a short timeout, we explained to the staff that the human species actually needs babies for its survival. They calmed down, picked up their crayons, and quietly went back to work.]
Fooled them. Stop the babies!
At least someone out there appreciates hospital food
Life in Alaska is not for the meek. It’s dark half the year. Summer is 3 weeks in July. And somehow, there’s a moose in line ahead of you at the doctor’s office. To make matters worse, it’s arguing about insurance. “What do you mean, you’ve heard the Moo Cross Moo Shield joke before?”
One might expect that Providence Alaska Health Park, located near downtown Anchorage, the largest city in Alaska by a massive margin, might be safe from ungulate invasion. Nope. In recent days, a young moose has taken to hanging around Providence campus, and it just could not find anything to eat. Remember, it may be early April, but this is Alaska. It’s still winter there. The ground’s still covered in snow.
Eventually, the gears in our young moose friend’s mind turned and it settled on a course of action: “Hey, those are some nice-looking plants behind that door over there. …” And that’s how Providence Alaska Health ended up with a moose munching on decorative potted plants in the hospital lobby.
Funnily enough, the moose didn’t even make a big scene. It just walked through the automatic doors and started chowing down. Security only found out because a tenant called them. Naturally though, once security made the announcement that a massive wild animal had been spotted in the building, the lobby was evacuated. … What do you mean, half the hospital came around to see it? Apparently, even though Alaskans have to fight moose herds on their daily commute, a lot of people wanted to see our moose friend do its thing.
“That’s crazy,” a woman in scrubs said in a video as she snapped a photo with her phone.
“This is the best. Like, what’s the code for this?” asked another bystander.
Despite security’s best efforts to shoo the moose out with barricades and offers of tasty branches, our furry friend left of its own volition, presumably irritated that his breakfast had become a spectator sport. But it didn’t go far. It hung around the front drive for a while, then went around the back of the building for a nap. What has four hooves and still doesn’t give a crap? Bob Moose-o! How you doing?
That click sounded stressed
How can people tell that you’re stressed? Maybe you get irritable and a little snappy. Some people have an inability to concentrate or focus. Eating that muffin when you weren’t really hungry could be a sign you’re not relaxed.
Did you know that your computer can be an indicator of your stress levels?
We tend to be working when we’re using computers, right? That can be a stressor in itself. Well, some researchers at ETH Zürich decided to have a look at the situation. Surprisingly, at least to us, one in three Swiss employees experience workplace stress, which makes us wonder what the percentage is in this country.
The Swiss researchers developed a model that tells how stressed someone is just by the way they use their computer mouse or type. The results of their study showed that those who were stressed clicked and tapped differently than participants who were more relaxed.
Stressed people click “more often and less precisely and cover longer distances on the screen,” while the relaxed take “shorter, more direct routes to reach their destination and take more time doing so,” study author Mara Nägelin explained in a written statement from ETH (Eidgenössische Technische Hochschule, or Swiss Federal Institute of Technology) Zürich.
Ever find when you’re frustrated and in a rush you end up making more mistakes? Same deal. Coauthor Jasmine Kerr noted that “increased levels of stress negatively impact our brain’s ability to process information.” Which totally is going to affect how we move.
Hopefully, these results can give insight to companies on how stressed their employees are and the effect it has on their work performance, eventually leading to, guess what, more research on how to alleviate workplace stress in general, which can benefit us all.
So if you find yourself in the office working on your computer like it’s a game of Perfection and time is running out, take a beat. Maybe try a stress-relieving breathing technique. Nonstressed people, according to the study, take fewer and longer pauses on their computers. Perfection on the job may mean relaxing first.
You and me and baby makes 10,003
If you were a virus hunter, looking for your next big virus discovery, where would you go? The wholesale seafood market in Wuhan? A gathering of unmasked anti-vaxxers in the heartland of America? The frozen snot fields of northwest Siberia?
How about babies? Well, it’s too late now, because that’s what Dennis Sandris Nielsen, PhD, of the University of Copenhagen, and his associates did, and they hit the mother lode. Actually, it was more like the infant load, if we’re being honest here.
“We found an exceptional number of unknown viruses in the faeces of these babies,” Dr. Nielsen said in a written statement from the university. (The study was published in Nature Microbiology, so we get the English spelling of feces.)
The investigators mapped the gut “viromes” of 647 healthy Danish 1-year-old children over the course of 5 years and found 10,000 species of viruses distributed across 248 different viral families, of which only 16 were already known. Incredible stuff, but then things took a turn for the cute. “The researchers named the remaining 232 unknown viral families after the children whose diapers made the study possible. As a result, new viral families include names like Sylvesterviridae, Rigmorviridae and Tristanviridae,” the university said.
About 90% of the viruses found in the feces are bacterial viruses, aka bacteriophages, which have bacteria as their hosts and don’t attack the children’s cells, so they don’t cause disease. The other 10%, however, are eukaryotic: They use human cells as hosts, so they can be either friend or foe. “It is thought-provoking that all children run around with 10-20 of these virus types that infect human cells. So, there is a constant viral infection taking place, which apparently doesn’t make them sick,” Dr. Nielsen said.
Doesn’t make them sick? Riiiight. The thought that this gives rise to now? People love babies. Everyone wants to pick up the baby. Now we know why. Because the viruses want us to! Well, those cute little faces aren’t fooling us anymore. No more babies for us. Everyone should stay away from babies and their evil little eukaryotic viruses. STOP THE BABIES!
[Editor’s note: After a short timeout, we explained to the staff that the human species actually needs babies for its survival. They calmed down, picked up their crayons, and quietly went back to work.]
Fooled them. Stop the babies!
At least someone out there appreciates hospital food
Life in Alaska is not for the meek. It’s dark half the year. Summer is 3 weeks in July. And somehow, there’s a moose in line ahead of you at the doctor’s office. To make matters worse, it’s arguing about insurance. “What do you mean, you’ve heard the Moo Cross Moo Shield joke before?”
One might expect that Providence Alaska Health Park, located near downtown Anchorage, the largest city in Alaska by a massive margin, might be safe from ungulate invasion. Nope. In recent days, a young moose has taken to hanging around Providence campus, and it just could not find anything to eat. Remember, it may be early April, but this is Alaska. It’s still winter there. The ground’s still covered in snow.
Eventually, the gears in our young moose friend’s mind turned and it settled on a course of action: “Hey, those are some nice-looking plants behind that door over there. …” And that’s how Providence Alaska Health ended up with a moose munching on decorative potted plants in the hospital lobby.
Funnily enough, the moose didn’t even make a big scene. It just walked through the automatic doors and started chowing down. Security only found out because a tenant called them. Naturally though, once security made the announcement that a massive wild animal had been spotted in the building, the lobby was evacuated. … What do you mean, half the hospital came around to see it? Apparently, even though Alaskans have to fight moose herds on their daily commute, a lot of people wanted to see our moose friend do its thing.
“That’s crazy,” a woman in scrubs said in a video as she snapped a photo with her phone.
“This is the best. Like, what’s the code for this?” asked another bystander.
Despite security’s best efforts to shoo the moose out with barricades and offers of tasty branches, our furry friend left of its own volition, presumably irritated that his breakfast had become a spectator sport. But it didn’t go far. It hung around the front drive for a while, then went around the back of the building for a nap. What has four hooves and still doesn’t give a crap? Bob Moose-o! How you doing?
That click sounded stressed
How can people tell that you’re stressed? Maybe you get irritable and a little snappy. Some people have an inability to concentrate or focus. Eating that muffin when you weren’t really hungry could be a sign you’re not relaxed.
Did you know that your computer can be an indicator of your stress levels?
We tend to be working when we’re using computers, right? That can be a stressor in itself. Well, some researchers at ETH Zürich decided to have a look at the situation. Surprisingly, at least to us, one in three Swiss employees experience workplace stress, which makes us wonder what the percentage is in this country.
The Swiss researchers developed a model that tells how stressed someone is just by the way they use their computer mouse or type. The results of their study showed that those who were stressed clicked and tapped differently than participants who were more relaxed.
Stressed people click “more often and less precisely and cover longer distances on the screen,” while the relaxed take “shorter, more direct routes to reach their destination and take more time doing so,” study author Mara Nägelin explained in a written statement from ETH (Eidgenössische Technische Hochschule, or Swiss Federal Institute of Technology) Zürich.
Ever find when you’re frustrated and in a rush you end up making more mistakes? Same deal. Coauthor Jasmine Kerr noted that “increased levels of stress negatively impact our brain’s ability to process information.” Which totally is going to affect how we move.
Hopefully, these results can give insight to companies on how stressed their employees are and the effect it has on their work performance, eventually leading to, guess what, more research on how to alleviate workplace stress in general, which can benefit us all.
So if you find yourself in the office working on your computer like it’s a game of Perfection and time is running out, take a beat. Maybe try a stress-relieving breathing technique. Nonstressed people, according to the study, take fewer and longer pauses on their computers. Perfection on the job may mean relaxing first.
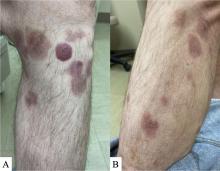
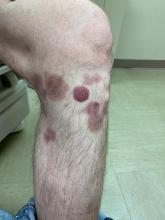
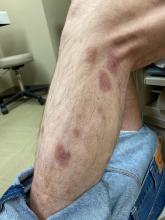
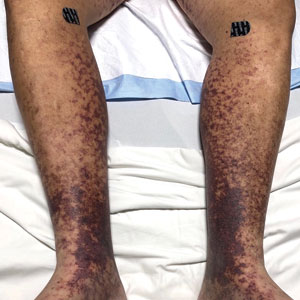
A 50-year-old White male presented with a 4- to 5-year history of progressively growing violaceous lesions on his left lower extremity
with scarce T-cells, classically presenting as rapidly progressive, plum-colored lesions on the lower extremities.1,2 CBCLs, with PCDLBCL-LT accounting for 4%, make up the minority of cutaneous lymphomas in the Western world.1-3 The leg type variant, typically demonstrating a female predominance and median age of onset in the 70s, is clinically aggressive and associated with a poorer prognosis, increased recurrence rate, and 40%-60% 5-year survival rate.1-5
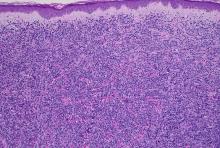
Histologically, this variant demonstrates a diffuse sheet-like growth of enlarged atypical B-cells distinctively separated from the epidermis by a prominent grenz zone. Classic PCDLBCL-LT immunophenotype includes B-cell markers CD20 and IgM; triple expressor phenotype indicating c-MYC, BCL-2, and BCL-6 positivity; as well as CD10 negativity, lack of BCL-2 rearrangement, and presence of a positive MYD-88 molecular result.
Other characteristic histopathological findings include positivity for post-germinal markers IRF4/MUM-1 and FOXP-1, positivity for additional B-cell markers, including CD79 and PAX5, and negativity of t(14;18) (q32;21).1,3-5
This case is of significant interest as it falls within the approximately 10% of PCDLBCL-LT cases demonstrating weak to negative MUM-1 staining, in addition to its presentation in a younger male individual.
While MUM-1 positivity is common in this subtype, its presence, or lack thereof, should not be looked at in isolation when evaluating diagnostic criteria, nor has it been shown to have a statistically significant effect on survival rate – in contrast to factors like lesion location on the leg versus non-leg lesions, multiple lesions at diagnosis, and dissemination to other sites.2,6
PCDLBCL-LT can uncommonly present in non-leg locations and only 10% depict associated B-symptoms, such as fatigue, night sweats, weight loss, or lymphadenopathy.2,6 First-line treatment is with the R-CHOP chemotherapy regimen – consisting of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone – although radiotherapy is sometimes considered in patients with a single small lesion.1,2
Because of possible cutaneous involvement beyond the legs, common lack of systemic symptoms, and variable immunophenotypes, this case of MUM-1 negative PCDLBCL-LT highlights the importance of a clinicopathological approach to differentiate the subtypes of CBCLs, allowing for proper and individualized stratification of risk, prognosis, and treatment.
This case was submitted and written by Marlee Hill, BS, Michael Franzetti, MD, Jeffrey McBride, MD, and Allison Hood, MD, of the University of Oklahoma, Oklahoma City. They also provided the photos. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Willemze R et al. Blood. 2019;133(16):1703-14.
2. Willemze R et al. Blood. 2005;105(10):3768-85.
3. Sukswai N et al. Pathology. 2020;52(1):53-67.
4. Hristov AC. Arch Pathol Lab Med. 2012;136(8):876-81.
5. Sokol L et al. Cancer Control. 2012;19(3):236-44.
6. Grange F et al. Arch Dermatol. 2007;143(9):1144-50.
with scarce T-cells, classically presenting as rapidly progressive, plum-colored lesions on the lower extremities.1,2 CBCLs, with PCDLBCL-LT accounting for 4%, make up the minority of cutaneous lymphomas in the Western world.1-3 The leg type variant, typically demonstrating a female predominance and median age of onset in the 70s, is clinically aggressive and associated with a poorer prognosis, increased recurrence rate, and 40%-60% 5-year survival rate.1-5
Histologically, this variant demonstrates a diffuse sheet-like growth of enlarged atypical B-cells distinctively separated from the epidermis by a prominent grenz zone. Classic PCDLBCL-LT immunophenotype includes B-cell markers CD20 and IgM; triple expressor phenotype indicating c-MYC, BCL-2, and BCL-6 positivity; as well as CD10 negativity, lack of BCL-2 rearrangement, and presence of a positive MYD-88 molecular result.
Other characteristic histopathological findings include positivity for post-germinal markers IRF4/MUM-1 and FOXP-1, positivity for additional B-cell markers, including CD79 and PAX5, and negativity of t(14;18) (q32;21).1,3-5
This case is of significant interest as it falls within the approximately 10% of PCDLBCL-LT cases demonstrating weak to negative MUM-1 staining, in addition to its presentation in a younger male individual.
While MUM-1 positivity is common in this subtype, its presence, or lack thereof, should not be looked at in isolation when evaluating diagnostic criteria, nor has it been shown to have a statistically significant effect on survival rate – in contrast to factors like lesion location on the leg versus non-leg lesions, multiple lesions at diagnosis, and dissemination to other sites.2,6
PCDLBCL-LT can uncommonly present in non-leg locations and only 10% depict associated B-symptoms, such as fatigue, night sweats, weight loss, or lymphadenopathy.2,6 First-line treatment is with the R-CHOP chemotherapy regimen – consisting of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone – although radiotherapy is sometimes considered in patients with a single small lesion.1,2
Because of possible cutaneous involvement beyond the legs, common lack of systemic symptoms, and variable immunophenotypes, this case of MUM-1 negative PCDLBCL-LT highlights the importance of a clinicopathological approach to differentiate the subtypes of CBCLs, allowing for proper and individualized stratification of risk, prognosis, and treatment.
This case was submitted and written by Marlee Hill, BS, Michael Franzetti, MD, Jeffrey McBride, MD, and Allison Hood, MD, of the University of Oklahoma, Oklahoma City. They also provided the photos. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Willemze R et al. Blood. 2019;133(16):1703-14.
2. Willemze R et al. Blood. 2005;105(10):3768-85.
3. Sukswai N et al. Pathology. 2020;52(1):53-67.
4. Hristov AC. Arch Pathol Lab Med. 2012;136(8):876-81.
5. Sokol L et al. Cancer Control. 2012;19(3):236-44.
6. Grange F et al. Arch Dermatol. 2007;143(9):1144-50.
with scarce T-cells, classically presenting as rapidly progressive, plum-colored lesions on the lower extremities.1,2 CBCLs, with PCDLBCL-LT accounting for 4%, make up the minority of cutaneous lymphomas in the Western world.1-3 The leg type variant, typically demonstrating a female predominance and median age of onset in the 70s, is clinically aggressive and associated with a poorer prognosis, increased recurrence rate, and 40%-60% 5-year survival rate.1-5
Histologically, this variant demonstrates a diffuse sheet-like growth of enlarged atypical B-cells distinctively separated from the epidermis by a prominent grenz zone. Classic PCDLBCL-LT immunophenotype includes B-cell markers CD20 and IgM; triple expressor phenotype indicating c-MYC, BCL-2, and BCL-6 positivity; as well as CD10 negativity, lack of BCL-2 rearrangement, and presence of a positive MYD-88 molecular result.
Other characteristic histopathological findings include positivity for post-germinal markers IRF4/MUM-1 and FOXP-1, positivity for additional B-cell markers, including CD79 and PAX5, and negativity of t(14;18) (q32;21).1,3-5
This case is of significant interest as it falls within the approximately 10% of PCDLBCL-LT cases demonstrating weak to negative MUM-1 staining, in addition to its presentation in a younger male individual.
While MUM-1 positivity is common in this subtype, its presence, or lack thereof, should not be looked at in isolation when evaluating diagnostic criteria, nor has it been shown to have a statistically significant effect on survival rate – in contrast to factors like lesion location on the leg versus non-leg lesions, multiple lesions at diagnosis, and dissemination to other sites.2,6
PCDLBCL-LT can uncommonly present in non-leg locations and only 10% depict associated B-symptoms, such as fatigue, night sweats, weight loss, or lymphadenopathy.2,6 First-line treatment is with the R-CHOP chemotherapy regimen – consisting of rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone – although radiotherapy is sometimes considered in patients with a single small lesion.1,2
Because of possible cutaneous involvement beyond the legs, common lack of systemic symptoms, and variable immunophenotypes, this case of MUM-1 negative PCDLBCL-LT highlights the importance of a clinicopathological approach to differentiate the subtypes of CBCLs, allowing for proper and individualized stratification of risk, prognosis, and treatment.
This case was submitted and written by Marlee Hill, BS, Michael Franzetti, MD, Jeffrey McBride, MD, and Allison Hood, MD, of the University of Oklahoma, Oklahoma City. They also provided the photos. Donna Bilu Martin, MD, edited the column.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
1. Willemze R et al. Blood. 2019;133(16):1703-14.
2. Willemze R et al. Blood. 2005;105(10):3768-85.
3. Sukswai N et al. Pathology. 2020;52(1):53-67.
4. Hristov AC. Arch Pathol Lab Med. 2012;136(8):876-81.
5. Sokol L et al. Cancer Control. 2012;19(3):236-44.
6. Grange F et al. Arch Dermatol. 2007;143(9):1144-50.
There was no cervical, axillary, or inguinal lymphadenopathy.
New 46-week PsA data released for IL-17A inhibitor izokibep
out to 46 weeks, according to an announcement reporting some of the long-term data by the drug’s developer, Acelyrin.
Izokibep is an antibody mimetic designed to inhibit IL-17A that the company says has “high potency and the potential for robust tissue penetration due to its small molecular size, about one-tenth the size of a monoclonal antibody.”
“Patients want both rapid and meaningful improvement of their symptoms, as well as lasting – and ideally improving – resolution of disease over time. Building on the 16-week data for izokibep reported at EULAR and ACR [American College of Rheumatology] last year, the 46-week data now show not only continued but marked improvements over time in key areas of psoriatic arthritis including joint pain, skin psoriasis, and enthesitis,” Philip J. Mease, MD, director of rheumatology research at the Swedish Medical Center and clinical professor at the University of Washington, both in Seattle, and an investigator in the izokibep PsA program, said in the announcement.
The phase 2 trial tested two doses of izokibep – 40 mg and 80 mg – given by subcutaneous injection every 2 weeks – against placebo in 135 adult patients with active PsA. For inclusion in the trial, patients had to have at least three swollen and at least three tender joints and an inadequate response to prior therapy including NSAIDs, conventional synthetic disease-modifying antirheumatic drugs, or tumor necrosis factor inhibitors. At week 16, the placebo group transitioned to 80 mg izokibep every 2 weeks and the trial treatment period continued for up to 46 weeks.
The trial’s primary endpoint of a 50% or higher level of improvement in ACR response criteria (ACR 50) was achieved by 48% of those on the 40 mg dose at week 16 and by 50% at week 46. For the 80-mg group, this rate rose from 52% to 79%. In the group that went from placebo to 80 mg, the ACR 50 rose from 13% with placebo to 73% with izokibep at week 46.
Resolution of enthesitis, measured by the Leeds Enthesitis Index, among those on the 40 mg dose, was achieved by 63% at week 16 and 83% at week 46, and among those on the 80 mg dose, 88% at week 16 and 89% at week 46. Those on placebo who switched to 80 mg of izokibep at week 16 had an 80% rate of enthesitis resolution at week 46.
Total resolution of skin involvement – 100% clearance of psoriasis based on the Psoriasis Area Severity Index (PASI) – was observed at 46 weeks in 50% of those on 40 mg, 71% of those on 80 mg, and 67% of those on 80 mg after week 16.
In its announcement, Acelyrin did not report withdrawal rates from the study after 16 weeks and through 46 weeks, although the statement said that izokibep “was generally well tolerated through 46 weeks, which is in line with previous trials of izokibep.” The most common adverse event was localized injection site reactions, with the majority graded mild to moderate in severity. They were generally the size of a quarter to half-dollar, and typically presented within the first few injections, after which they declined in incidence. In the trial, a case of vulvar cancer was determined to be potentially drug related, the company said.
Acelyrin is currently conducting a phase 2b/3 trial in PsA evaluating a range of doses, including significantly higher doses than in the phase 2 trial, that the company said “could potentially result in better ACR, PASI, and enthesitis resolution responses.”
The drug has been tested at doses up to 160 mg, in some cases for up to 3 years, in more than 400 patients with psoriasis, spondyloarthritis, noninfective uveitis, and hidradenitis suppurativa.
The full 46-week data from this trial will be presented at a future scientific meeting, according to the company.
out to 46 weeks, according to an announcement reporting some of the long-term data by the drug’s developer, Acelyrin.
Izokibep is an antibody mimetic designed to inhibit IL-17A that the company says has “high potency and the potential for robust tissue penetration due to its small molecular size, about one-tenth the size of a monoclonal antibody.”
“Patients want both rapid and meaningful improvement of their symptoms, as well as lasting – and ideally improving – resolution of disease over time. Building on the 16-week data for izokibep reported at EULAR and ACR [American College of Rheumatology] last year, the 46-week data now show not only continued but marked improvements over time in key areas of psoriatic arthritis including joint pain, skin psoriasis, and enthesitis,” Philip J. Mease, MD, director of rheumatology research at the Swedish Medical Center and clinical professor at the University of Washington, both in Seattle, and an investigator in the izokibep PsA program, said in the announcement.
The phase 2 trial tested two doses of izokibep – 40 mg and 80 mg – given by subcutaneous injection every 2 weeks – against placebo in 135 adult patients with active PsA. For inclusion in the trial, patients had to have at least three swollen and at least three tender joints and an inadequate response to prior therapy including NSAIDs, conventional synthetic disease-modifying antirheumatic drugs, or tumor necrosis factor inhibitors. At week 16, the placebo group transitioned to 80 mg izokibep every 2 weeks and the trial treatment period continued for up to 46 weeks.
The trial’s primary endpoint of a 50% or higher level of improvement in ACR response criteria (ACR 50) was achieved by 48% of those on the 40 mg dose at week 16 and by 50% at week 46. For the 80-mg group, this rate rose from 52% to 79%. In the group that went from placebo to 80 mg, the ACR 50 rose from 13% with placebo to 73% with izokibep at week 46.
Resolution of enthesitis, measured by the Leeds Enthesitis Index, among those on the 40 mg dose, was achieved by 63% at week 16 and 83% at week 46, and among those on the 80 mg dose, 88% at week 16 and 89% at week 46. Those on placebo who switched to 80 mg of izokibep at week 16 had an 80% rate of enthesitis resolution at week 46.
Total resolution of skin involvement – 100% clearance of psoriasis based on the Psoriasis Area Severity Index (PASI) – was observed at 46 weeks in 50% of those on 40 mg, 71% of those on 80 mg, and 67% of those on 80 mg after week 16.
In its announcement, Acelyrin did not report withdrawal rates from the study after 16 weeks and through 46 weeks, although the statement said that izokibep “was generally well tolerated through 46 weeks, which is in line with previous trials of izokibep.” The most common adverse event was localized injection site reactions, with the majority graded mild to moderate in severity. They were generally the size of a quarter to half-dollar, and typically presented within the first few injections, after which they declined in incidence. In the trial, a case of vulvar cancer was determined to be potentially drug related, the company said.
Acelyrin is currently conducting a phase 2b/3 trial in PsA evaluating a range of doses, including significantly higher doses than in the phase 2 trial, that the company said “could potentially result in better ACR, PASI, and enthesitis resolution responses.”
The drug has been tested at doses up to 160 mg, in some cases for up to 3 years, in more than 400 patients with psoriasis, spondyloarthritis, noninfective uveitis, and hidradenitis suppurativa.
The full 46-week data from this trial will be presented at a future scientific meeting, according to the company.
out to 46 weeks, according to an announcement reporting some of the long-term data by the drug’s developer, Acelyrin.
Izokibep is an antibody mimetic designed to inhibit IL-17A that the company says has “high potency and the potential for robust tissue penetration due to its small molecular size, about one-tenth the size of a monoclonal antibody.”
“Patients want both rapid and meaningful improvement of their symptoms, as well as lasting – and ideally improving – resolution of disease over time. Building on the 16-week data for izokibep reported at EULAR and ACR [American College of Rheumatology] last year, the 46-week data now show not only continued but marked improvements over time in key areas of psoriatic arthritis including joint pain, skin psoriasis, and enthesitis,” Philip J. Mease, MD, director of rheumatology research at the Swedish Medical Center and clinical professor at the University of Washington, both in Seattle, and an investigator in the izokibep PsA program, said in the announcement.
The phase 2 trial tested two doses of izokibep – 40 mg and 80 mg – given by subcutaneous injection every 2 weeks – against placebo in 135 adult patients with active PsA. For inclusion in the trial, patients had to have at least three swollen and at least three tender joints and an inadequate response to prior therapy including NSAIDs, conventional synthetic disease-modifying antirheumatic drugs, or tumor necrosis factor inhibitors. At week 16, the placebo group transitioned to 80 mg izokibep every 2 weeks and the trial treatment period continued for up to 46 weeks.
The trial’s primary endpoint of a 50% or higher level of improvement in ACR response criteria (ACR 50) was achieved by 48% of those on the 40 mg dose at week 16 and by 50% at week 46. For the 80-mg group, this rate rose from 52% to 79%. In the group that went from placebo to 80 mg, the ACR 50 rose from 13% with placebo to 73% with izokibep at week 46.
Resolution of enthesitis, measured by the Leeds Enthesitis Index, among those on the 40 mg dose, was achieved by 63% at week 16 and 83% at week 46, and among those on the 80 mg dose, 88% at week 16 and 89% at week 46. Those on placebo who switched to 80 mg of izokibep at week 16 had an 80% rate of enthesitis resolution at week 46.
Total resolution of skin involvement – 100% clearance of psoriasis based on the Psoriasis Area Severity Index (PASI) – was observed at 46 weeks in 50% of those on 40 mg, 71% of those on 80 mg, and 67% of those on 80 mg after week 16.
In its announcement, Acelyrin did not report withdrawal rates from the study after 16 weeks and through 46 weeks, although the statement said that izokibep “was generally well tolerated through 46 weeks, which is in line with previous trials of izokibep.” The most common adverse event was localized injection site reactions, with the majority graded mild to moderate in severity. They were generally the size of a quarter to half-dollar, and typically presented within the first few injections, after which they declined in incidence. In the trial, a case of vulvar cancer was determined to be potentially drug related, the company said.
Acelyrin is currently conducting a phase 2b/3 trial in PsA evaluating a range of doses, including significantly higher doses than in the phase 2 trial, that the company said “could potentially result in better ACR, PASI, and enthesitis resolution responses.”
The drug has been tested at doses up to 160 mg, in some cases for up to 3 years, in more than 400 patients with psoriasis, spondyloarthritis, noninfective uveitis, and hidradenitis suppurativa.
The full 46-week data from this trial will be presented at a future scientific meeting, according to the company.
Retiform Purpura on the Lower Legs
The Diagnosis: Type I Cryoglobulinemia
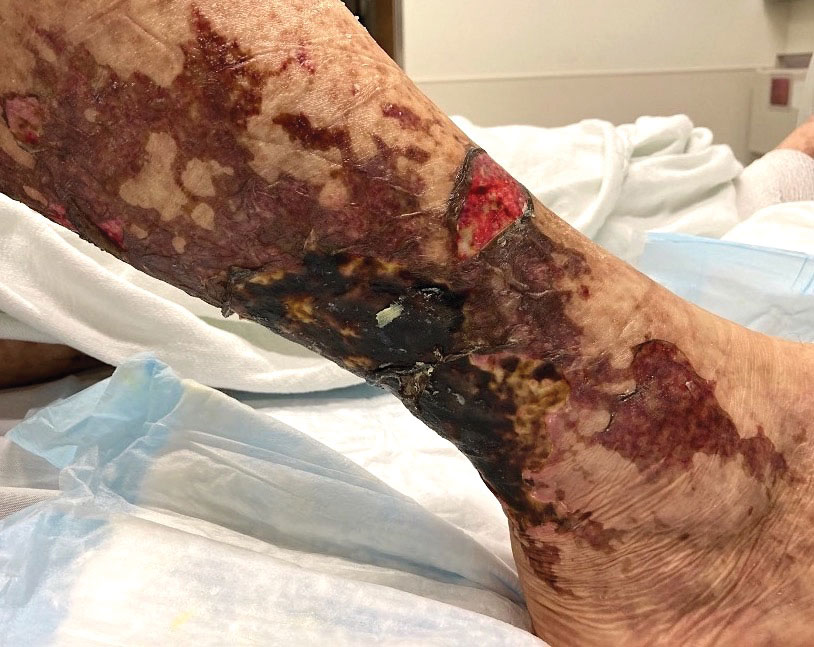
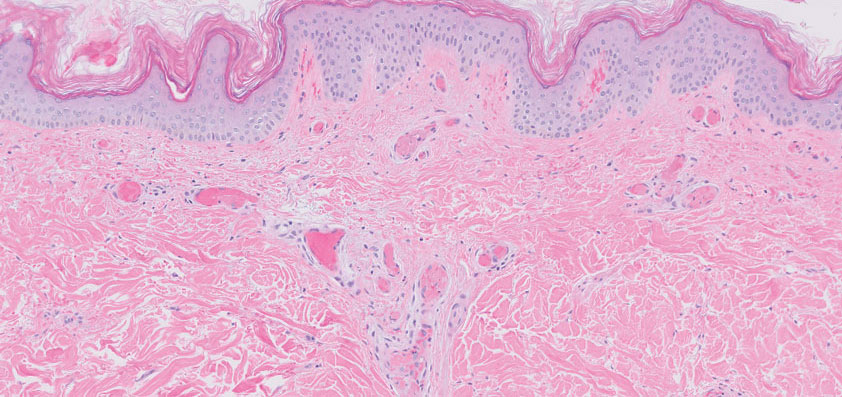
Retiform purpura with overlying necrosis subsequently developed over the course of a week following presentation (Figure 1). A skin biopsy showed fibrin thrombi and congestion of small- and medium-sized blood vessels, consistent with vasculopathy (Figure 2). Urinalysis revealed hematuria and proteinuria. A renal biopsy performed due to a continually elevated serum creatinine level revealed glomerulonephritis with numerous IgG1 lambda–restricted glomerular capillary hyaline thrombi, compatible with a lymphoproliferative disorder–associated type I cryoglobulinemia. A serum cryoglobulin immunofixation test confirmed type I cryoglobulinemia involving monoclonal IgG lambda. The combination of cutaneous, renal, and hematologic findings was consistent with type I cryoglobulinemia. A subsequent bone marrow biopsy demonstrated a CD20+ lambda–restricted plasma cell neoplasm. Initial treatment with high-dose corticosteroids followed by targeted treatment of the underlying hematologic condition with bortezomib, rituximab, and dexamethasone improved the skin disease.
Cryoglobulins are abnormal immunoglobulins that precipitate at temperatures below 37 °C. The persistent presence of cryoglobulins in the serum is termed cryoglobulinemia.1 Type I cryoglobulinemia is distinguished from mixed cryoglobulinemia—types II and III—by the presence of a single monoclonal immunoglobulin, typically IgM or IgG. It is associated with lymphoproliferative disorders, most commonly monoclonal gammopathy of undetermined significance and B-cell malignancies such as Waldenström macroglobulinemia, multiple myeloma, or chronic lymphocytic leukemia. Histopathology shows occlusion of small vessel lumina with homogenous eosinophilic material containing the monoclonal cryoprecipitate.2 Disease manifestations are caused by small vessel occlusion, which leads to ischemia and tissue damage.
Retiform purpura, livedo reticularis/racemosa, and necrosis leading to ulcers are the most common cutaneous clinical findings. Extracutaneous signs include peripheral neuropathy, arthralgia, Raynaud phenomenon, and acrocyanosis. Renal involvement, most commonly glomerulonephritis with associated proteinuria, is noted in 14% to 20% of cases.3,4 An elevated cryocrit can lead to symptoms of hyperviscosity syndrome.2
Treatment is difficult and primarily is focused on addressing the underlying hematologic condition, which is responsible for synthesis of the cryoglobulin. Decreasing cryoglobulin production leads to decreased occlusion of blood vessels, thus alleviating the ischemia and skin damage. Monoclonal gammopathy of undetermined significance–related type I cryoglobulinemia initially is treated with corticosteroids followed by rituximab if a CD20+ B-cell clone is identified.2 Bortezomib is recommended for cases associated with Waldenström macroglobulinemia and cases associated with multiple myeloma with concurrent renal failure. In patients with neuropathy, a lenalidomide-based treatment can be employed. Patients should be instructed to keep extremities warm.2 Diabetic foot care guidelines should be followed to prevent wound complications. The differential diagnosis for type I cryoglobulinemia includes other causes of retiform purpura–like angioinvasive fungal infection, antiphospholipid antibody syndrome, calciphylaxis, and livedoid vasculopathy.5 Angioinvasive fungal infections are caused by Candida, Aspergillus, and Mucorales species, as well as other hyaline molds. They typically occur in immunocompromised patients and invade the blood vessels via direct inoculation or dissemination.6 Patients present with retiform purpura but typically will be acutely ill with fevers and vital sign abnormalities. Histopathology with special stains often will identify the fungal organisms in the dermis or inside blood vessel walls with vessel wall destruction and hemorrhage.7 Accurate diagnosis is essential to selecting appropriate antifungal agents. If angioinvasive fungal infection is clinically suspected, treatment should begin before culture and histopathologic data are available.7
Antiphospholipid antibody syndrome is an autoimmune thrombophilia that can occur as primary disease or in association with other autoimmune conditions, most commonly systemic lupus erythematosus. Diagnosis requires the presence of antiphospholipid antibodies, such as lupus anticoagulant, anticardiolipin antibody, anti–β2-glycoprotein-1 antibody, with arterial or venous thrombosis and/or recurrent pregnancy loss. Paraproteinemia is not seen. The most common cutaneous finding is livedo reticularis, with livedo racemosa being a more distinctive finding.8 Small vessel thrombosis is seen histopathologically. Treatment includes antiplatelet and anticoagulant medications. Patients with refractory disease may benefit from additional therapy with hydroxychloroquine or intravenous immunoglobulins.8
Calciphylaxis is a rare depositional vasculopathy that often occurs in patients with end-stage renal disease on dialysis. Patients present with painful and poor-healing skin lesions including indurated nodules, violaceous plaques, and retiform purpura that typically affect areas of high adiposity such as the thighs, abdomen, and buttocks.9 Ulceration and superimposed infections are common complications. Histopathologically, small dermal and subcutaneous vessels demonstrate calcification, microthrombosis, and fibrointimal hyperplasia.9 Wound management is critically important in patients with calciphylaxis. Treatment with intravenous sodium thiosulfate is typical, but prognosis remains poor. Although livedoid vasculopathy may present with retiform purpura in the ankles, paraproteinemia is not seen and patients frequently present with punched-out ulcerations that tend to heal into atrophie blanche.10 Livedoid vasculopathy has been associated with underlying hypercoagulable states, connective tissue diseases, and chronic venous hypertension. Hypercoagulability and endothelial cell damage contribute to the formation of fibrin thrombi in the superficial dermal blood vessels. Histopathology demonstrates thickening of vessel walls and intraluminal hyaline thrombi. Successful treatment in most cases is achieved with anticoagulation therapy, typically rivaroxaban, especially in patients with underlying hypercoagulability. Antiplatelet therapy also may be considered, while anabolic agents have been shown to be helpful in patients with connective tissue disease.10
- Desbois AC, Cacoub P, Saadoun D. Cryoglobulinemia: an update in 2019. Joint Bone Spine. 2019;86:707-713. doi:10.1016/j .jbspin.2019.01.016
- Muchtar E, Magen H, Gertz MA. How I treat cryoglobulinemia. Blood. 2017;129:289-298. doi:10.1182/blood-2016-09-719773
- Sidana S, Rajkumar SV, Dispenzieri A, et al. Clinical presentation and outcomes of patients with type 1 monoclonal cryoglobulinemia. Am J Hematol. 2017;92:668-673. doi:10.1002/ajh.24745
- Harel S, Mohr M, Jahn I, et al. Clinico-biological characteristics and treatment of type I monoclonal cryoglobulinaemia: a study of 64 cases. Br J Haematol. 2015;168:671-678. doi:10.1111/bjh.13196
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Shields BE, Rosenbach M, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: background, epidemiology, and clinical presentation. J Am Acad Dermatol. 2019;80:869-880.e5. doi:10.1016/j.jaad.2018.04.059
- Berger AP, Ford BA, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: diagnosis, management, and complications. J Am Acad Dermatol. 2019;80:883-898.e2. doi:10.1016/j.jaad.2018.04.058
- Negrini S, Pappalardo F, Murdaca G, et al. The antiphospholipid syndrome: from pathophysiology to treatment. Clin Exp Med. 2017;17:257-267. doi:10.1007/s10238-016-0430-5
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis. 2015;66:133-146. doi:10.1053/j.ajkd.2015.01.034
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816. doi:10.1016/j.jaad.2019.07.113
The Diagnosis: Type I Cryoglobulinemia
Retiform purpura with overlying necrosis subsequently developed over the course of a week following presentation (Figure 1). A skin biopsy showed fibrin thrombi and congestion of small- and medium-sized blood vessels, consistent with vasculopathy (Figure 2). Urinalysis revealed hematuria and proteinuria. A renal biopsy performed due to a continually elevated serum creatinine level revealed glomerulonephritis with numerous IgG1 lambda–restricted glomerular capillary hyaline thrombi, compatible with a lymphoproliferative disorder–associated type I cryoglobulinemia. A serum cryoglobulin immunofixation test confirmed type I cryoglobulinemia involving monoclonal IgG lambda. The combination of cutaneous, renal, and hematologic findings was consistent with type I cryoglobulinemia. A subsequent bone marrow biopsy demonstrated a CD20+ lambda–restricted plasma cell neoplasm. Initial treatment with high-dose corticosteroids followed by targeted treatment of the underlying hematologic condition with bortezomib, rituximab, and dexamethasone improved the skin disease.

Cryoglobulins are abnormal immunoglobulins that precipitate at temperatures below 37 °C. The persistent presence of cryoglobulins in the serum is termed cryoglobulinemia.1 Type I cryoglobulinemia is distinguished from mixed cryoglobulinemia—types II and III—by the presence of a single monoclonal immunoglobulin, typically IgM or IgG. It is associated with lymphoproliferative disorders, most commonly monoclonal gammopathy of undetermined significance and B-cell malignancies such as Waldenström macroglobulinemia, multiple myeloma, or chronic lymphocytic leukemia. Histopathology shows occlusion of small vessel lumina with homogenous eosinophilic material containing the monoclonal cryoprecipitate.2 Disease manifestations are caused by small vessel occlusion, which leads to ischemia and tissue damage.

Retiform purpura, livedo reticularis/racemosa, and necrosis leading to ulcers are the most common cutaneous clinical findings. Extracutaneous signs include peripheral neuropathy, arthralgia, Raynaud phenomenon, and acrocyanosis. Renal involvement, most commonly glomerulonephritis with associated proteinuria, is noted in 14% to 20% of cases.3,4 An elevated cryocrit can lead to symptoms of hyperviscosity syndrome.2
Treatment is difficult and primarily is focused on addressing the underlying hematologic condition, which is responsible for synthesis of the cryoglobulin. Decreasing cryoglobulin production leads to decreased occlusion of blood vessels, thus alleviating the ischemia and skin damage. Monoclonal gammopathy of undetermined significance–related type I cryoglobulinemia initially is treated with corticosteroids followed by rituximab if a CD20+ B-cell clone is identified.2 Bortezomib is recommended for cases associated with Waldenström macroglobulinemia and cases associated with multiple myeloma with concurrent renal failure. In patients with neuropathy, a lenalidomide-based treatment can be employed. Patients should be instructed to keep extremities warm.2 Diabetic foot care guidelines should be followed to prevent wound complications. The differential diagnosis for type I cryoglobulinemia includes other causes of retiform purpura–like angioinvasive fungal infection, antiphospholipid antibody syndrome, calciphylaxis, and livedoid vasculopathy.5 Angioinvasive fungal infections are caused by Candida, Aspergillus, and Mucorales species, as well as other hyaline molds. They typically occur in immunocompromised patients and invade the blood vessels via direct inoculation or dissemination.6 Patients present with retiform purpura but typically will be acutely ill with fevers and vital sign abnormalities. Histopathology with special stains often will identify the fungal organisms in the dermis or inside blood vessel walls with vessel wall destruction and hemorrhage.7 Accurate diagnosis is essential to selecting appropriate antifungal agents. If angioinvasive fungal infection is clinically suspected, treatment should begin before culture and histopathologic data are available.7
Antiphospholipid antibody syndrome is an autoimmune thrombophilia that can occur as primary disease or in association with other autoimmune conditions, most commonly systemic lupus erythematosus. Diagnosis requires the presence of antiphospholipid antibodies, such as lupus anticoagulant, anticardiolipin antibody, anti–β2-glycoprotein-1 antibody, with arterial or venous thrombosis and/or recurrent pregnancy loss. Paraproteinemia is not seen. The most common cutaneous finding is livedo reticularis, with livedo racemosa being a more distinctive finding.8 Small vessel thrombosis is seen histopathologically. Treatment includes antiplatelet and anticoagulant medications. Patients with refractory disease may benefit from additional therapy with hydroxychloroquine or intravenous immunoglobulins.8
Calciphylaxis is a rare depositional vasculopathy that often occurs in patients with end-stage renal disease on dialysis. Patients present with painful and poor-healing skin lesions including indurated nodules, violaceous plaques, and retiform purpura that typically affect areas of high adiposity such as the thighs, abdomen, and buttocks.9 Ulceration and superimposed infections are common complications. Histopathologically, small dermal and subcutaneous vessels demonstrate calcification, microthrombosis, and fibrointimal hyperplasia.9 Wound management is critically important in patients with calciphylaxis. Treatment with intravenous sodium thiosulfate is typical, but prognosis remains poor. Although livedoid vasculopathy may present with retiform purpura in the ankles, paraproteinemia is not seen and patients frequently present with punched-out ulcerations that tend to heal into atrophie blanche.10 Livedoid vasculopathy has been associated with underlying hypercoagulable states, connective tissue diseases, and chronic venous hypertension. Hypercoagulability and endothelial cell damage contribute to the formation of fibrin thrombi in the superficial dermal blood vessels. Histopathology demonstrates thickening of vessel walls and intraluminal hyaline thrombi. Successful treatment in most cases is achieved with anticoagulation therapy, typically rivaroxaban, especially in patients with underlying hypercoagulability. Antiplatelet therapy also may be considered, while anabolic agents have been shown to be helpful in patients with connective tissue disease.10
The Diagnosis: Type I Cryoglobulinemia
Retiform purpura with overlying necrosis subsequently developed over the course of a week following presentation (Figure 1). A skin biopsy showed fibrin thrombi and congestion of small- and medium-sized blood vessels, consistent with vasculopathy (Figure 2). Urinalysis revealed hematuria and proteinuria. A renal biopsy performed due to a continually elevated serum creatinine level revealed glomerulonephritis with numerous IgG1 lambda–restricted glomerular capillary hyaline thrombi, compatible with a lymphoproliferative disorder–associated type I cryoglobulinemia. A serum cryoglobulin immunofixation test confirmed type I cryoglobulinemia involving monoclonal IgG lambda. The combination of cutaneous, renal, and hematologic findings was consistent with type I cryoglobulinemia. A subsequent bone marrow biopsy demonstrated a CD20+ lambda–restricted plasma cell neoplasm. Initial treatment with high-dose corticosteroids followed by targeted treatment of the underlying hematologic condition with bortezomib, rituximab, and dexamethasone improved the skin disease.

Cryoglobulins are abnormal immunoglobulins that precipitate at temperatures below 37 °C. The persistent presence of cryoglobulins in the serum is termed cryoglobulinemia.1 Type I cryoglobulinemia is distinguished from mixed cryoglobulinemia—types II and III—by the presence of a single monoclonal immunoglobulin, typically IgM or IgG. It is associated with lymphoproliferative disorders, most commonly monoclonal gammopathy of undetermined significance and B-cell malignancies such as Waldenström macroglobulinemia, multiple myeloma, or chronic lymphocytic leukemia. Histopathology shows occlusion of small vessel lumina with homogenous eosinophilic material containing the monoclonal cryoprecipitate.2 Disease manifestations are caused by small vessel occlusion, which leads to ischemia and tissue damage.

Retiform purpura, livedo reticularis/racemosa, and necrosis leading to ulcers are the most common cutaneous clinical findings. Extracutaneous signs include peripheral neuropathy, arthralgia, Raynaud phenomenon, and acrocyanosis. Renal involvement, most commonly glomerulonephritis with associated proteinuria, is noted in 14% to 20% of cases.3,4 An elevated cryocrit can lead to symptoms of hyperviscosity syndrome.2
Treatment is difficult and primarily is focused on addressing the underlying hematologic condition, which is responsible for synthesis of the cryoglobulin. Decreasing cryoglobulin production leads to decreased occlusion of blood vessels, thus alleviating the ischemia and skin damage. Monoclonal gammopathy of undetermined significance–related type I cryoglobulinemia initially is treated with corticosteroids followed by rituximab if a CD20+ B-cell clone is identified.2 Bortezomib is recommended for cases associated with Waldenström macroglobulinemia and cases associated with multiple myeloma with concurrent renal failure. In patients with neuropathy, a lenalidomide-based treatment can be employed. Patients should be instructed to keep extremities warm.2 Diabetic foot care guidelines should be followed to prevent wound complications. The differential diagnosis for type I cryoglobulinemia includes other causes of retiform purpura–like angioinvasive fungal infection, antiphospholipid antibody syndrome, calciphylaxis, and livedoid vasculopathy.5 Angioinvasive fungal infections are caused by Candida, Aspergillus, and Mucorales species, as well as other hyaline molds. They typically occur in immunocompromised patients and invade the blood vessels via direct inoculation or dissemination.6 Patients present with retiform purpura but typically will be acutely ill with fevers and vital sign abnormalities. Histopathology with special stains often will identify the fungal organisms in the dermis or inside blood vessel walls with vessel wall destruction and hemorrhage.7 Accurate diagnosis is essential to selecting appropriate antifungal agents. If angioinvasive fungal infection is clinically suspected, treatment should begin before culture and histopathologic data are available.7
Antiphospholipid antibody syndrome is an autoimmune thrombophilia that can occur as primary disease or in association with other autoimmune conditions, most commonly systemic lupus erythematosus. Diagnosis requires the presence of antiphospholipid antibodies, such as lupus anticoagulant, anticardiolipin antibody, anti–β2-glycoprotein-1 antibody, with arterial or venous thrombosis and/or recurrent pregnancy loss. Paraproteinemia is not seen. The most common cutaneous finding is livedo reticularis, with livedo racemosa being a more distinctive finding.8 Small vessel thrombosis is seen histopathologically. Treatment includes antiplatelet and anticoagulant medications. Patients with refractory disease may benefit from additional therapy with hydroxychloroquine or intravenous immunoglobulins.8
Calciphylaxis is a rare depositional vasculopathy that often occurs in patients with end-stage renal disease on dialysis. Patients present with painful and poor-healing skin lesions including indurated nodules, violaceous plaques, and retiform purpura that typically affect areas of high adiposity such as the thighs, abdomen, and buttocks.9 Ulceration and superimposed infections are common complications. Histopathologically, small dermal and subcutaneous vessels demonstrate calcification, microthrombosis, and fibrointimal hyperplasia.9 Wound management is critically important in patients with calciphylaxis. Treatment with intravenous sodium thiosulfate is typical, but prognosis remains poor. Although livedoid vasculopathy may present with retiform purpura in the ankles, paraproteinemia is not seen and patients frequently present with punched-out ulcerations that tend to heal into atrophie blanche.10 Livedoid vasculopathy has been associated with underlying hypercoagulable states, connective tissue diseases, and chronic venous hypertension. Hypercoagulability and endothelial cell damage contribute to the formation of fibrin thrombi in the superficial dermal blood vessels. Histopathology demonstrates thickening of vessel walls and intraluminal hyaline thrombi. Successful treatment in most cases is achieved with anticoagulation therapy, typically rivaroxaban, especially in patients with underlying hypercoagulability. Antiplatelet therapy also may be considered, while anabolic agents have been shown to be helpful in patients with connective tissue disease.10
- Desbois AC, Cacoub P, Saadoun D. Cryoglobulinemia: an update in 2019. Joint Bone Spine. 2019;86:707-713. doi:10.1016/j .jbspin.2019.01.016
- Muchtar E, Magen H, Gertz MA. How I treat cryoglobulinemia. Blood. 2017;129:289-298. doi:10.1182/blood-2016-09-719773
- Sidana S, Rajkumar SV, Dispenzieri A, et al. Clinical presentation and outcomes of patients with type 1 monoclonal cryoglobulinemia. Am J Hematol. 2017;92:668-673. doi:10.1002/ajh.24745
- Harel S, Mohr M, Jahn I, et al. Clinico-biological characteristics and treatment of type I monoclonal cryoglobulinaemia: a study of 64 cases. Br J Haematol. 2015;168:671-678. doi:10.1111/bjh.13196
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Shields BE, Rosenbach M, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: background, epidemiology, and clinical presentation. J Am Acad Dermatol. 2019;80:869-880.e5. doi:10.1016/j.jaad.2018.04.059
- Berger AP, Ford BA, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: diagnosis, management, and complications. J Am Acad Dermatol. 2019;80:883-898.e2. doi:10.1016/j.jaad.2018.04.058
- Negrini S, Pappalardo F, Murdaca G, et al. The antiphospholipid syndrome: from pathophysiology to treatment. Clin Exp Med. 2017;17:257-267. doi:10.1007/s10238-016-0430-5
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis. 2015;66:133-146. doi:10.1053/j.ajkd.2015.01.034
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816. doi:10.1016/j.jaad.2019.07.113
- Desbois AC, Cacoub P, Saadoun D. Cryoglobulinemia: an update in 2019. Joint Bone Spine. 2019;86:707-713. doi:10.1016/j .jbspin.2019.01.016
- Muchtar E, Magen H, Gertz MA. How I treat cryoglobulinemia. Blood. 2017;129:289-298. doi:10.1182/blood-2016-09-719773
- Sidana S, Rajkumar SV, Dispenzieri A, et al. Clinical presentation and outcomes of patients with type 1 monoclonal cryoglobulinemia. Am J Hematol. 2017;92:668-673. doi:10.1002/ajh.24745
- Harel S, Mohr M, Jahn I, et al. Clinico-biological characteristics and treatment of type I monoclonal cryoglobulinaemia: a study of 64 cases. Br J Haematol. 2015;168:671-678. doi:10.1111/bjh.13196
- Georgesen C, Fox LP, Harp J. Retiform purpura: a diagnostic approach. J Am Acad Dermatol. 2020;82:783-796. doi:10.1016/j.jaad.2019.07.112
- Shields BE, Rosenbach M, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: background, epidemiology, and clinical presentation. J Am Acad Dermatol. 2019;80:869-880.e5. doi:10.1016/j.jaad.2018.04.059
- Berger AP, Ford BA, Brown-Joel Z, et al. Angioinvasive fungal infections impacting the skin: diagnosis, management, and complications. J Am Acad Dermatol. 2019;80:883-898.e2. doi:10.1016/j.jaad.2018.04.058
- Negrini S, Pappalardo F, Murdaca G, et al. The antiphospholipid syndrome: from pathophysiology to treatment. Clin Exp Med. 2017;17:257-267. doi:10.1007/s10238-016-0430-5
- Nigwekar SU, Kroshinsky D, Nazarian RM, et al. Calciphylaxis: risk factors, diagnosis, and treatment. Am J Kidney Dis. 2015;66:133-146. doi:10.1053/j.ajkd.2015.01.034
- Georgesen C, Fox LP, Harp J. Retiform purpura: workup and therapeutic considerations in select conditions. J Am Acad Dermatol. 2020;82:799-816. doi:10.1016/j.jaad.2019.07.113
A 58-year-old man presented with a petechial and purpuric rash limited to the lower extremities. He reported that the rash had been present for months but worsened acutely over the last 3 days with new-onset dark urine, joint pain, and edema limiting his ability to walk. Physical examination showed areas of violaceous macules and papules on the legs and dorsal feet in a reticular distribution. Laboratory findings were remarkable for an elevated serum creatinine level of 2.75 mg/dL (reference range, 0.70–1.30 mg/dL), and serum immunofixation revealed the presence of markedly elevated IgG lambda monoclonal proteins. He was afebrile and his vital signs were stable. Dermatology, nephrology, and rheumatology services were consulted.
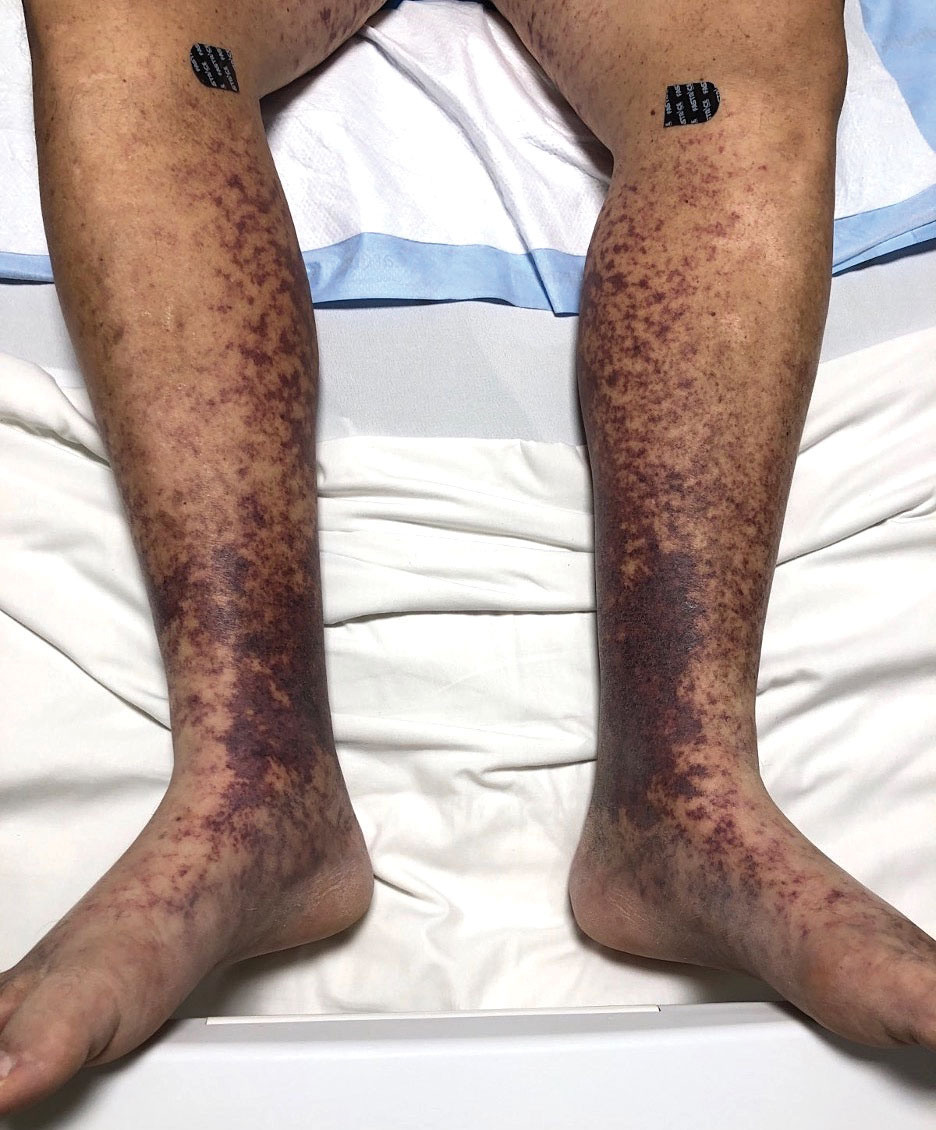
The earlier baricitinib for severe alopecia areata is started, the better
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
NEW ORLEANS – In the nearly 1 year .
“The journey to JAK inhibition in alopecia areata has been incredible,” Raj Chovatiya, MD, PhD, assistant professor of dermatology and director of the center for eczema and itch at Northwestern University, Chicago, said at the annual meeting of the American Academy of Dermatology. “JAK inhibitors are here to stay, and I think baricitinib offers an amazing opportunity for the right patients.”
The efficacy and safety of baricitinib (Olumiant) for AA was studied in two randomized, double-blind, placebo-controlled trials (BRAVE-AA1 and BRAVE-AA2) with patients who had at least 50% scalp hair loss as measured by the Severity of Alopecia Tool (SALT) for more than 6 months. Patients in these trials received either a placebo, 2 mg of baricitinib, or 4 mg of baricitinib every day. The primary measurement of efficacy for both trials was the proportion of patients who achieved a SALT score of 20 or less, or at least 80% scalp hair coverage at week 36. The researchers found that 36%-39% of individuals in the 4-mg arm achieved a SALT score of less than 20, compared with 19%-23% of individuals in the 2 mg arm. Similar outcomes were observed for eyebrow and eyelash hair loss.
Most adverse events observed in BRAVE-AA1 and BRAVE-AA2 were in the mild to moderate range, and the actual number of adverse events leading to permanent discontinuation was extremely low. The most common adverse events were upper respiratory tract infections, headache, nasopharyngitis, acne, urinary tract infections, and an increase in blood creatine kinase.
Baricitinib is not recommended for use in combination with other JAK inhibitors, biologic immunomodulators, or other potent immunosuppressants, Dr. Chovatiya said. Required lab evaluations include baseline testing for tuberculosis and viral hepatitis; CBC, hepatic function, and renal function at baseline and then as clinically indicated; and lipids after 12 weeks of therapy, then as clinically indicated. The recommended starting dose of baricitinib is 2 mg per day, which can be increased to 4 mg per day if the response is not adequate. “However, for patients with nearly complete or complete scalp hair loss, with or without substantial eyelash or eyebrow hair loss, 4 mg once daily is recommended,” he said. “Once an adequate response is achieved, it’s recommended to reduce from 4 to 2 mg daily.”
52-week, 76-week data
According to pooled data from BRAVE-AA1 and BRAVE-AA2 published online March 1, 2023, efficacy continues to increase out to 52 weeks. Specifically, by week 52, 39% of individuals in the 4 mg arm achieved a SALT score of 20 or less, compared with 22.6% of individuals in the 2 mg arm. “You see similar linear growth in the eyebrow and eyelash response loss as well,” Dr. Chovatiya said.
In other findings, patients in the 4 mg treatment arm who achieved a SALT score of 20 or less at week 52 were eligible for randomized down titration, provided that they had stayed on the same dose of baricitinib from initial randomization. According to data from baricitinib manufacturer Eli Lilly, 77.5% of patients who stepped down to the 2 mg dose from the 4 mg dose at week 52 achieved a SALT score of 20 or less at week 76, Dr. Chovatiya said. “If I can keep someone on 4 mg that’s great, but it looks like you can go to a lower dose and do a pretty good job,” he said.
Patients in the baricitinib arms who achieved a SALT score of 20 or less at week 52 were eligible for randomized withdrawal, provided that they had stayed on the same dose of the drug from initial randomization. According to Dr. Chovatiya, 89.4% of individuals who remained on the 4 mg dose to week 76 maintained a SALT score of 20 or less, compared with 33.3% of those who switched from the 4 mg to placebo. “The takeaway here is that clinically, longitudinal treatment looks to be required in this time period” for continued efficacy, he said. “However, what this looks like in the real world remains to be seen.”
A recently published integrated analysis of safety data from BRAVE-AA1 and BRAVE-AA2 reported that no deaths occurred and of the few reported serious infections, nearly half were COVID-19. There was a single case of multidermatomal herpes zoster and no cases of tuberculosis. One patient with risk factors for MI had an MI during a placebo-controlled period, and one study participant with a history of COVID-19 infection developed a pulmonary embolism at day 638. There was one case each of chronic lymphocytic leukemia, B-cell lymphoma, breast cancer, and appendicitis.
Baseline severity and treatment response
“Does treatment response vary with baseline disease status?” Dr. Chovatiya asked. “Yes. People with very severe hair loss [defined as a SALT score of 95 or higher] tended to do worse, while the rest of the study population did even better – an almost twofold difference. This means that you want to treat as early as you possibly can. It’s interesting to note that you don’t see this difference as much in the case of eyebrows and eyelashes. This makes sense, though. Eyebrows and eyelashes probably behave differently in terms of growth than the scalp does.”
Certain baseline characteristics of patients in BRAVE-AA1 and BRAVE-AA2 portended better outcomes. Women tended to fare better than men, but individuals who had longer histories of AA did not respond well. “People who had a shorter duration of their current episode of AA also did better than people who had a longer current episode, so we want to think about treating as soon as we possibly can,” Dr. Chovatiya said.
Dr. Chovatiya disclosed that he is a consultant to, a speaker for, investigator, and/or a member of the advisory board for several pharmaceutical companies, including Eli Lilly.
AT AAD 2023
Why 9 is not too young for the HPV vaccine
For Sonja O’Leary, MD, higher rates of vaccination against human papillomavirus came with the flip of a switch.
Dr. O’Leary, the interim director of service for outpatient pediatric services at Denver Health and Hospital Authority, and her colleagues saw rates of HPV and other childhood immunizations drop during the COVID-19 pandemic and decided to act. Their health system, which includes 28 federally qualified health centers, offers vaccines at any inpatient or outpatient visit based on alerts from their electronic health record.
“It was actually really simple; it was really just changing our best-practice alert,” Dr. O’Leary said. Beginning in May 2021, and after notifying clinic staff of the impending change, DHHA dropped the alert for first dose of HPV from age 11 to 9.
The approach worked. Compared with the first 5 months of 2021, the percentage of children aged 9-13 years with an in-person visit who received at least one dose of HPV vaccine between June 2021 and August 2022 rose from 30.3% to 42.8% – a 41% increase. The share who received two doses by age 13 years more than doubled, from 19.3% to 42.7%, Dr. O’Leary said.
Frustrated efforts
Although those figures might seem to make an iron-clad case for earlier vaccinations against HPV – which is responsible for nearly 35,000 cases of cancer annually – factors beyond statistics have frustrated efforts to increase acceptance of the shots.
Data published in 2022 from the U.S. Centers for Disease Control and Prevention found that 89.6% of teens aged 13-17 years received at least one dose of tetanus, diphtheria, and acellular pertussis vaccine, and 89% got one or more doses of meningococcal conjugate vaccine. However, only 76.9% had received one or more doses of HPV vaccine. The rate of receiving both doses needed for full protection was much lower (61.7%).
Both the American Academy of Pediatrics and the American Cancer Society now endorse the strategy of offering HPV vaccine as early as age 9, which avoids the need for multiple shots at a single visit and results in more kids getting both doses. In a recent study that surveyed primary care professionals who see pediatric patients, 21% were already offering HPV vaccine at age 9, and another 48% were willing to try the approach.
What was the most common objection to the earlier age? Nearly three-quarters of clinicians said they felt that parents weren’t ready to talk about HPV vaccination yet.
Noel Brewer, PhD, one of the authors of the survey study, wondered why clinicians feel the need to bring up sex at all. “Providers should never be talking about sex when they are talking about vaccine, because that’s not the point,” said Dr. Brewer, the distinguished professor in public health at the University of North Carolina at Chapel Hill. He pointed out that providers don’t talk about the route of transmission for any other vaccine.
Dr. Brewer led a randomized controlled trial that trained pediatric clinicians in the “announcement” strategy, in which the clinician announces the vaccines that are due at that visit. If the parent hesitates, the clinician then probes further to identify and address their concerns and provides more information. If the parent is still not convinced, the clinician notes the discussion in the chart and tries again at the next visit.
The strategy was effective: Intervention clinics had a 5.4% higher rate of HPV vaccination coverage than control clinics after six months. Dr. Brewer and his colleagues have trained over 1,700 providers in the technique since 2020.
A cancer – not STI – vaccine
Although DHHA hasn’t participated in Dr. Brewer’s training, Dr. O’Leary and her colleagues take a similar approach of simply stating which vaccines the child should receive that day. And they talk about HPV as a cancer vaccine instead of one to prevent a sexually transmitted infection.
In her experience, this emphasis changes the conversation. Dr. O’Leary described a typical comment from parents as, “Oh, of course I would give my child a vaccine that could prevent cancer.”
Ana Rodriguez, MD, MPH, an obstetrician, became interested in raising rates of vaccination against HPV after watching too many women battle a preventable cancer. She worked for several years in the Rio Grande Valley along the U.S. border with Mexico, an impoverished rural area with poor access to health care and high rates of HPV infection.
“I would treat women very young – not even 30 years of age – already fighting advanced precancerous lesions secondary to HPV,” said Dr. Rodriguez, an associate professor of Obstetrics & Gynecology at the University of Texas Medical Branch at Galveston.
In 2016, when Texas ranked 47th in the nation for rates of up-to-date HPV vaccination, Dr. Rodriguez helped launch a community-based educational campaign in four rural counties in the Rio Grande Valley using social media, radio, and in-person meetings with school PTA members and members of school boards to educate staff and parents about the need for vaccination against the infection.
In 2019, the team began offering the vaccine to children ages 9-12 years at back-to-school events, progress report nights, and other school events, pivoting to outdoor events using a mobile vaccine van after COVID-19 struck. They recently published a study showing that 73.6% of students who received their first dose of vaccine at age 11 or younger completed the series, compared with only 45.1% of children who got their first dose at age 12 or older.
Dr. Rodriguez encountered parents who felt 9 or 10 years old was too young because their children were not going to be sexually active anytime soon. Her response was to describe HPV as a tool to prevent cancer, telling parents, “If you vaccinate your kids young enough, they will be protected for life.”
Lifetime protection is another point in favor of giving HPV vaccine prior to Tdap and MenACWY. The response to the two-dose series of HPV in preadolescents is robust and long-lasting, with no downside to giving it a few years earlier. In contrast, immunity to MenACWY wanes after a few years, so the immunization must be given before children enter high school, when their risk for meningitis increases.
The annual toll of deaths in the United States from meningococcus, tetanus, diphtheria, and pertussis typically totals less than 100, whereas cancer deaths attributable to HPV infection number in the thousands each year. And that may be the best reason for attempting new strategies to help HPV vaccination rates catch up to the rest of the preteen vaccines.
Dr. Brewer’s work was supported by the Gillings School of Global Public Health, the Lineberger Comprehensive Cancer Center at the University of North Carolina, and from training grants from the National Cancer Institute. Dr. Brewer has received research funding from Merck, Pfizer, and GSK and served as a paid advisor for Merck. Dr. O’Leary reports no relevant financial relationships. Dr. Rodriguez received a grant from the Cancer Prevention Research Institute of Texas, and the study was supported by the Institute for Translational Sciences at the University of Texas Medical Branch.
A version of this article first appeared on Medscape.com.
For Sonja O’Leary, MD, higher rates of vaccination against human papillomavirus came with the flip of a switch.
Dr. O’Leary, the interim director of service for outpatient pediatric services at Denver Health and Hospital Authority, and her colleagues saw rates of HPV and other childhood immunizations drop during the COVID-19 pandemic and decided to act. Their health system, which includes 28 federally qualified health centers, offers vaccines at any inpatient or outpatient visit based on alerts from their electronic health record.
“It was actually really simple; it was really just changing our best-practice alert,” Dr. O’Leary said. Beginning in May 2021, and after notifying clinic staff of the impending change, DHHA dropped the alert for first dose of HPV from age 11 to 9.
The approach worked. Compared with the first 5 months of 2021, the percentage of children aged 9-13 years with an in-person visit who received at least one dose of HPV vaccine between June 2021 and August 2022 rose from 30.3% to 42.8% – a 41% increase. The share who received two doses by age 13 years more than doubled, from 19.3% to 42.7%, Dr. O’Leary said.
Frustrated efforts
Although those figures might seem to make an iron-clad case for earlier vaccinations against HPV – which is responsible for nearly 35,000 cases of cancer annually – factors beyond statistics have frustrated efforts to increase acceptance of the shots.
Data published in 2022 from the U.S. Centers for Disease Control and Prevention found that 89.6% of teens aged 13-17 years received at least one dose of tetanus, diphtheria, and acellular pertussis vaccine, and 89% got one or more doses of meningococcal conjugate vaccine. However, only 76.9% had received one or more doses of HPV vaccine. The rate of receiving both doses needed for full protection was much lower (61.7%).
Both the American Academy of Pediatrics and the American Cancer Society now endorse the strategy of offering HPV vaccine as early as age 9, which avoids the need for multiple shots at a single visit and results in more kids getting both doses. In a recent study that surveyed primary care professionals who see pediatric patients, 21% were already offering HPV vaccine at age 9, and another 48% were willing to try the approach.
What was the most common objection to the earlier age? Nearly three-quarters of clinicians said they felt that parents weren’t ready to talk about HPV vaccination yet.
Noel Brewer, PhD, one of the authors of the survey study, wondered why clinicians feel the need to bring up sex at all. “Providers should never be talking about sex when they are talking about vaccine, because that’s not the point,” said Dr. Brewer, the distinguished professor in public health at the University of North Carolina at Chapel Hill. He pointed out that providers don’t talk about the route of transmission for any other vaccine.
Dr. Brewer led a randomized controlled trial that trained pediatric clinicians in the “announcement” strategy, in which the clinician announces the vaccines that are due at that visit. If the parent hesitates, the clinician then probes further to identify and address their concerns and provides more information. If the parent is still not convinced, the clinician notes the discussion in the chart and tries again at the next visit.
The strategy was effective: Intervention clinics had a 5.4% higher rate of HPV vaccination coverage than control clinics after six months. Dr. Brewer and his colleagues have trained over 1,700 providers in the technique since 2020.
A cancer – not STI – vaccine
Although DHHA hasn’t participated in Dr. Brewer’s training, Dr. O’Leary and her colleagues take a similar approach of simply stating which vaccines the child should receive that day. And they talk about HPV as a cancer vaccine instead of one to prevent a sexually transmitted infection.
In her experience, this emphasis changes the conversation. Dr. O’Leary described a typical comment from parents as, “Oh, of course I would give my child a vaccine that could prevent cancer.”
Ana Rodriguez, MD, MPH, an obstetrician, became interested in raising rates of vaccination against HPV after watching too many women battle a preventable cancer. She worked for several years in the Rio Grande Valley along the U.S. border with Mexico, an impoverished rural area with poor access to health care and high rates of HPV infection.
“I would treat women very young – not even 30 years of age – already fighting advanced precancerous lesions secondary to HPV,” said Dr. Rodriguez, an associate professor of Obstetrics & Gynecology at the University of Texas Medical Branch at Galveston.
In 2016, when Texas ranked 47th in the nation for rates of up-to-date HPV vaccination, Dr. Rodriguez helped launch a community-based educational campaign in four rural counties in the Rio Grande Valley using social media, radio, and in-person meetings with school PTA members and members of school boards to educate staff and parents about the need for vaccination against the infection.
In 2019, the team began offering the vaccine to children ages 9-12 years at back-to-school events, progress report nights, and other school events, pivoting to outdoor events using a mobile vaccine van after COVID-19 struck. They recently published a study showing that 73.6% of students who received their first dose of vaccine at age 11 or younger completed the series, compared with only 45.1% of children who got their first dose at age 12 or older.
Dr. Rodriguez encountered parents who felt 9 or 10 years old was too young because their children were not going to be sexually active anytime soon. Her response was to describe HPV as a tool to prevent cancer, telling parents, “If you vaccinate your kids young enough, they will be protected for life.”
Lifetime protection is another point in favor of giving HPV vaccine prior to Tdap and MenACWY. The response to the two-dose series of HPV in preadolescents is robust and long-lasting, with no downside to giving it a few years earlier. In contrast, immunity to MenACWY wanes after a few years, so the immunization must be given before children enter high school, when their risk for meningitis increases.
The annual toll of deaths in the United States from meningococcus, tetanus, diphtheria, and pertussis typically totals less than 100, whereas cancer deaths attributable to HPV infection number in the thousands each year. And that may be the best reason for attempting new strategies to help HPV vaccination rates catch up to the rest of the preteen vaccines.
Dr. Brewer’s work was supported by the Gillings School of Global Public Health, the Lineberger Comprehensive Cancer Center at the University of North Carolina, and from training grants from the National Cancer Institute. Dr. Brewer has received research funding from Merck, Pfizer, and GSK and served as a paid advisor for Merck. Dr. O’Leary reports no relevant financial relationships. Dr. Rodriguez received a grant from the Cancer Prevention Research Institute of Texas, and the study was supported by the Institute for Translational Sciences at the University of Texas Medical Branch.
A version of this article first appeared on Medscape.com.
For Sonja O’Leary, MD, higher rates of vaccination against human papillomavirus came with the flip of a switch.
Dr. O’Leary, the interim director of service for outpatient pediatric services at Denver Health and Hospital Authority, and her colleagues saw rates of HPV and other childhood immunizations drop during the COVID-19 pandemic and decided to act. Their health system, which includes 28 federally qualified health centers, offers vaccines at any inpatient or outpatient visit based on alerts from their electronic health record.
“It was actually really simple; it was really just changing our best-practice alert,” Dr. O’Leary said. Beginning in May 2021, and after notifying clinic staff of the impending change, DHHA dropped the alert for first dose of HPV from age 11 to 9.
The approach worked. Compared with the first 5 months of 2021, the percentage of children aged 9-13 years with an in-person visit who received at least one dose of HPV vaccine between June 2021 and August 2022 rose from 30.3% to 42.8% – a 41% increase. The share who received two doses by age 13 years more than doubled, from 19.3% to 42.7%, Dr. O’Leary said.
Frustrated efforts
Although those figures might seem to make an iron-clad case for earlier vaccinations against HPV – which is responsible for nearly 35,000 cases of cancer annually – factors beyond statistics have frustrated efforts to increase acceptance of the shots.
Data published in 2022 from the U.S. Centers for Disease Control and Prevention found that 89.6% of teens aged 13-17 years received at least one dose of tetanus, diphtheria, and acellular pertussis vaccine, and 89% got one or more doses of meningococcal conjugate vaccine. However, only 76.9% had received one or more doses of HPV vaccine. The rate of receiving both doses needed for full protection was much lower (61.7%).
Both the American Academy of Pediatrics and the American Cancer Society now endorse the strategy of offering HPV vaccine as early as age 9, which avoids the need for multiple shots at a single visit and results in more kids getting both doses. In a recent study that surveyed primary care professionals who see pediatric patients, 21% were already offering HPV vaccine at age 9, and another 48% were willing to try the approach.
What was the most common objection to the earlier age? Nearly three-quarters of clinicians said they felt that parents weren’t ready to talk about HPV vaccination yet.
Noel Brewer, PhD, one of the authors of the survey study, wondered why clinicians feel the need to bring up sex at all. “Providers should never be talking about sex when they are talking about vaccine, because that’s not the point,” said Dr. Brewer, the distinguished professor in public health at the University of North Carolina at Chapel Hill. He pointed out that providers don’t talk about the route of transmission for any other vaccine.
Dr. Brewer led a randomized controlled trial that trained pediatric clinicians in the “announcement” strategy, in which the clinician announces the vaccines that are due at that visit. If the parent hesitates, the clinician then probes further to identify and address their concerns and provides more information. If the parent is still not convinced, the clinician notes the discussion in the chart and tries again at the next visit.
The strategy was effective: Intervention clinics had a 5.4% higher rate of HPV vaccination coverage than control clinics after six months. Dr. Brewer and his colleagues have trained over 1,700 providers in the technique since 2020.
A cancer – not STI – vaccine
Although DHHA hasn’t participated in Dr. Brewer’s training, Dr. O’Leary and her colleagues take a similar approach of simply stating which vaccines the child should receive that day. And they talk about HPV as a cancer vaccine instead of one to prevent a sexually transmitted infection.
In her experience, this emphasis changes the conversation. Dr. O’Leary described a typical comment from parents as, “Oh, of course I would give my child a vaccine that could prevent cancer.”
Ana Rodriguez, MD, MPH, an obstetrician, became interested in raising rates of vaccination against HPV after watching too many women battle a preventable cancer. She worked for several years in the Rio Grande Valley along the U.S. border with Mexico, an impoverished rural area with poor access to health care and high rates of HPV infection.
“I would treat women very young – not even 30 years of age – already fighting advanced precancerous lesions secondary to HPV,” said Dr. Rodriguez, an associate professor of Obstetrics & Gynecology at the University of Texas Medical Branch at Galveston.
In 2016, when Texas ranked 47th in the nation for rates of up-to-date HPV vaccination, Dr. Rodriguez helped launch a community-based educational campaign in four rural counties in the Rio Grande Valley using social media, radio, and in-person meetings with school PTA members and members of school boards to educate staff and parents about the need for vaccination against the infection.
In 2019, the team began offering the vaccine to children ages 9-12 years at back-to-school events, progress report nights, and other school events, pivoting to outdoor events using a mobile vaccine van after COVID-19 struck. They recently published a study showing that 73.6% of students who received their first dose of vaccine at age 11 or younger completed the series, compared with only 45.1% of children who got their first dose at age 12 or older.
Dr. Rodriguez encountered parents who felt 9 or 10 years old was too young because their children were not going to be sexually active anytime soon. Her response was to describe HPV as a tool to prevent cancer, telling parents, “If you vaccinate your kids young enough, they will be protected for life.”
Lifetime protection is another point in favor of giving HPV vaccine prior to Tdap and MenACWY. The response to the two-dose series of HPV in preadolescents is robust and long-lasting, with no downside to giving it a few years earlier. In contrast, immunity to MenACWY wanes after a few years, so the immunization must be given before children enter high school, when their risk for meningitis increases.
The annual toll of deaths in the United States from meningococcus, tetanus, diphtheria, and pertussis typically totals less than 100, whereas cancer deaths attributable to HPV infection number in the thousands each year. And that may be the best reason for attempting new strategies to help HPV vaccination rates catch up to the rest of the preteen vaccines.
Dr. Brewer’s work was supported by the Gillings School of Global Public Health, the Lineberger Comprehensive Cancer Center at the University of North Carolina, and from training grants from the National Cancer Institute. Dr. Brewer has received research funding from Merck, Pfizer, and GSK and served as a paid advisor for Merck. Dr. O’Leary reports no relevant financial relationships. Dr. Rodriguez received a grant from the Cancer Prevention Research Institute of Texas, and the study was supported by the Institute for Translational Sciences at the University of Texas Medical Branch.
A version of this article first appeared on Medscape.com.
Antimicrobial resistance requires a manifold response
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
BUENOS AIRES – Antimicrobial resistance (AMR) has become a global concern. And while one issue to be addressed is the deficit in research and development for new antibiotics, efforts to tackle this public health threat also should be directed toward promoting more rational prescription practices and strengthening the ability to identify the microorganisms responsible for infections, according to the World Health Organization. This was the conclusion reached at the fourth meeting of the WHO AMR Surveillance and Quality Assessment Collaborating Centres Network, which was held in Buenos Aires.
“We have to provide assistance to countries to ensure that the drugs are being used responsibly. We can come up with new antibiotics, but the issue at hand is not simply one of innovation: If nothing is done to correct inappropriate prescription practices and to overcome the lack of diagnostic laboratories at the country level, we’re going to miss out on those drugs as soon as they become available,” Kitty van Weezenbeek, MD, PhD, MPH, director of the AMR Surveillance, Prevention, and Control (AMR/SPC) Department at the WHO’s headquarters in Geneva, told this news organization.
Dr. van Weezenbeek pointed out that although there are currently no shortages of antimicrobials, the development and launch of new drugs that fight multidrug-resistant infections – infections for which there are few therapeutic options – has proceeded slowly. “It takes 10 to 15 years to develop a new antibiotic,” she said, adding that “the majority of pharmaceutical companies that had been engaged in the development of antimicrobials have filed for bankruptcy.”
In 2019, more people died – 1.2 million – from AMR than from malaria, tuberculosis, and HIV combined. Why are there so few market incentives when there is such a great need for those drugs? “One reason is that the pharmaceutical industry makes more money with long-term treatments, such as those for cancer and respiratory diseases. The other problem is that people everywhere are told not to use antibiotics,” said Dr. van Weezenbeek.
“A course of antibiotics lasts a few days, especially because we’re promoting rational use. Therefore, the trend is for the total amount of antimicrobials being used to be lower. So, it’s not as profitable,” added Carmem Lucia Pessoa-Silva, MD, PhD, head of the Surveillance, Evidence, and Laboratory Strengthening Unit of the WHO’s AMR/SPC Department.
On that note, Dr. van Weezenbeek mentioned that member countries are working with pharmaceutical companies and universities to address this problem. The WHO, for its part, has responded by implementing a global mechanism with a public health approach to create a “healthy” and equitable market for these medicines.
AMR is one of the top 10 global threats to human health. But it also has an impact on animal production, agricultural production, and the environment. Strategies to tackle AMR based on the One Health approach should involve all actors, social sectors, and citizens, according to Eva Jané Llopis, PhD, the representative of the Pan American Health Organization/WHO in Argentina.
At the root of the AMR problem is the widespread use of these drugs as growth promoters in animal production – for which several countries have enacted regulations – as well as “misunderstandings” between patients and physicians when there is not sufficient, timely access to laboratory diagnostics, especially in low- and middle-income countries.
“People think that if they’re given broad-spectrum antibiotics, they’re being prescribed the best antibiotics; and doctors, because there are no laboratory services, prescribe broad-spectrum antibiotics because they want to help patients. But that ends up causing more resistance to drugs, and thus, those antibiotics aren’t good for the patients,” said Dr. van Weezenbeek.
The WHO Global AMR and Use Surveillance System (GLASS) was launched in 2015. Its 2022 report, which marked the end of the system’s early implementation period, noted that the reported AMR rates are often lower in countries, territories, and areas with better testing coverage for most pathogen-drug-infection site combinations. However, as Dr. Pessoa-Silva acknowledged, monitoring “has not yet generated representative data,” because in many cases, countries either do not have surveillance systems or have only recently started implementing them.
Even so, the indicators that are available paint an increasingly worrisome picture. “For example, in many countries, resistance rates to first-line antibiotics were around 10%-20% with respect to Escherichia coli urinary tract infections and bloodstream bacteriologically confirmed infections. So, the risk of treatment failure is very high,” explained Dr. Pessoa-Silva.
The latest estimates indicate that every 2 or 3 minutes, somewhere in the world, a child dies from AMR. And the situation is particularly “dramatic” in neonatal intensive care units, where outbreaks of multidrug-resistant infections have a mortality rate of 50%, said Pilar Ramón-Pardo, MD, PhD, lead of the Special Program on AMR at the Pan American Health Organization, the WHO Regional Office for the Americas.
AMR rates also got worse during the pandemic because of the inappropriate prescription of massive amounts of antibiotics to hospitalized patients – something that was not in compliance with guidelines or protocols. Silvia Bertagnolio, MD, is an infectious disease specialist and the head of the Control and Response Strategies Unit in the WHO’s AMR Division. She spoke about the global clinical platform data pertaining to more than 1,500,000 patients who were hospitalized for COVID-19. Since 2020, 85% received antimicrobial treatment, despite the fact that only 5% had a concomitant infection at admission. “It’s easier to give antibiotics than to make a proper diagnosis,” said Dr. Bertagnolio.
This article was translated from Medscape’s Spanish edition and a version appeared on Medscape.com.
New Medicare rule streamlines prior authorization in Medicare Advantage plans
A new federal rule seeks to reduce Medicare Advantage insurance plans’ prior authorization burdens on physicians while also ensuring that enrollees have the same access to necessary care that they would receive under traditional fee-for-service Medicare.
The prior authorization changes, announced this week, are part of the Centers for Medicare & Medicaid Services’ 2024 update of policy changes for Medicare Advantage and Part D pharmacy plans
Medicare Advantage plans’ business practices have raised significant concerns in recent years. More than 28 million Americans were enrolled in a Medicare Advantage plan in 2022, which is nearly half of all Medicare enrollees, according to the Kaiser Family Foundation.
Medicare pays a fixed amount per enrollee per year to these privately run managed care plans, in contrast to traditional fee-for-service Medicare. Medicare Advantage plans have been criticized for aggressive marketing, for overbilling the federal government for care, and for using prior authorization to inappropriately deny needed care to patients.
About 13% of prior authorization requests that are denied by Medicare Advantage plans actually met Medicare coverage rules and should have been approved, the Office of the Inspector General at the U.S. Department of Health & Human Services reported in 2022.
The newly finalized rule now requires Medicare Advantage plans to do the following.
- Ensure that a prior authorization approval, once granted, remains valid for as long as medically necessary to avoid disruptions in care.
- Conduct an annual review of utilization management policies.
- Ensure that coverage denials based on medical necessity be reviewed by health care professionals with relevant expertise before a denial can be issued.
Physician groups welcomed the changes. In a statement, the American Medical Association said that an initial reading of the rule suggested CMS had “taken important steps toward right-sizing the prior authorization process.”
The Medical Group Management Association praised CMS in a statement for having limited “dangerous disruptions and delays to necessary patient care” resulting from the cumbersome processes of prior approval. With the new rules, CMS will provide greater consistency across Advantage plans as well as traditional Medicare, said Anders Gilberg, MGMA’s senior vice president of government affairs, in a statement.
Peer consideration
The final rule did disappoint physician groups in one key way. CMS rebuffed requests to have CMS require Advantage plans to use reviewers of the same specialty as treating physicians in handling disputes about prior authorization. CMS said it expects plans to exercise judgment in finding reviewers with “sufficient expertise to make an informed and supportable decision.”
“In some instances, we expect that plans will use a physician or other health care professional of the same specialty or subspecialty as the treating physician,” CMS said. “In other instances, we expect that plans will utilize a reviewer with specialized training, certification, or clinical experience in the applicable field of medicine.”
Medicare Advantage marketing ‘sowing confusion’
With this final rule, CMS also sought to protect consumers from “potentially misleading marketing practices” used in promoting Medicare Advantage and Part D prescription drug plans.
The agency said it had received complaints about people who have received official-looking promotional materials for Medicare that directed them not to government sources of information but to Medicare Advantage and Part D plans or their agents and brokers.
Ads now must mention a specific plan name, and they cannot use the Medicare name, CMS logo, Medicare card, or other government information in a misleading way, CMS said.
“CMS can see no value or purpose in a non-governmental entity’s use of the Medicare logo or HHS logo except for the express purpose of sowing confusion and misrepresenting itself as the government,” the agency said.
A version of this article first appeared on Medscape.com.
A new federal rule seeks to reduce Medicare Advantage insurance plans’ prior authorization burdens on physicians while also ensuring that enrollees have the same access to necessary care that they would receive under traditional fee-for-service Medicare.
The prior authorization changes, announced this week, are part of the Centers for Medicare & Medicaid Services’ 2024 update of policy changes for Medicare Advantage and Part D pharmacy plans
Medicare Advantage plans’ business practices have raised significant concerns in recent years. More than 28 million Americans were enrolled in a Medicare Advantage plan in 2022, which is nearly half of all Medicare enrollees, according to the Kaiser Family Foundation.
Medicare pays a fixed amount per enrollee per year to these privately run managed care plans, in contrast to traditional fee-for-service Medicare. Medicare Advantage plans have been criticized for aggressive marketing, for overbilling the federal government for care, and for using prior authorization to inappropriately deny needed care to patients.
About 13% of prior authorization requests that are denied by Medicare Advantage plans actually met Medicare coverage rules and should have been approved, the Office of the Inspector General at the U.S. Department of Health & Human Services reported in 2022.
The newly finalized rule now requires Medicare Advantage plans to do the following.
- Ensure that a prior authorization approval, once granted, remains valid for as long as medically necessary to avoid disruptions in care.
- Conduct an annual review of utilization management policies.
- Ensure that coverage denials based on medical necessity be reviewed by health care professionals with relevant expertise before a denial can be issued.
Physician groups welcomed the changes. In a statement, the American Medical Association said that an initial reading of the rule suggested CMS had “taken important steps toward right-sizing the prior authorization process.”
The Medical Group Management Association praised CMS in a statement for having limited “dangerous disruptions and delays to necessary patient care” resulting from the cumbersome processes of prior approval. With the new rules, CMS will provide greater consistency across Advantage plans as well as traditional Medicare, said Anders Gilberg, MGMA’s senior vice president of government affairs, in a statement.
Peer consideration
The final rule did disappoint physician groups in one key way. CMS rebuffed requests to have CMS require Advantage plans to use reviewers of the same specialty as treating physicians in handling disputes about prior authorization. CMS said it expects plans to exercise judgment in finding reviewers with “sufficient expertise to make an informed and supportable decision.”
“In some instances, we expect that plans will use a physician or other health care professional of the same specialty or subspecialty as the treating physician,” CMS said. “In other instances, we expect that plans will utilize a reviewer with specialized training, certification, or clinical experience in the applicable field of medicine.”
Medicare Advantage marketing ‘sowing confusion’
With this final rule, CMS also sought to protect consumers from “potentially misleading marketing practices” used in promoting Medicare Advantage and Part D prescription drug plans.
The agency said it had received complaints about people who have received official-looking promotional materials for Medicare that directed them not to government sources of information but to Medicare Advantage and Part D plans or their agents and brokers.
Ads now must mention a specific plan name, and they cannot use the Medicare name, CMS logo, Medicare card, or other government information in a misleading way, CMS said.
“CMS can see no value or purpose in a non-governmental entity’s use of the Medicare logo or HHS logo except for the express purpose of sowing confusion and misrepresenting itself as the government,” the agency said.
A version of this article first appeared on Medscape.com.
A new federal rule seeks to reduce Medicare Advantage insurance plans’ prior authorization burdens on physicians while also ensuring that enrollees have the same access to necessary care that they would receive under traditional fee-for-service Medicare.
The prior authorization changes, announced this week, are part of the Centers for Medicare & Medicaid Services’ 2024 update of policy changes for Medicare Advantage and Part D pharmacy plans
Medicare Advantage plans’ business practices have raised significant concerns in recent years. More than 28 million Americans were enrolled in a Medicare Advantage plan in 2022, which is nearly half of all Medicare enrollees, according to the Kaiser Family Foundation.
Medicare pays a fixed amount per enrollee per year to these privately run managed care plans, in contrast to traditional fee-for-service Medicare. Medicare Advantage plans have been criticized for aggressive marketing, for overbilling the federal government for care, and for using prior authorization to inappropriately deny needed care to patients.
About 13% of prior authorization requests that are denied by Medicare Advantage plans actually met Medicare coverage rules and should have been approved, the Office of the Inspector General at the U.S. Department of Health & Human Services reported in 2022.
The newly finalized rule now requires Medicare Advantage plans to do the following.
- Ensure that a prior authorization approval, once granted, remains valid for as long as medically necessary to avoid disruptions in care.
- Conduct an annual review of utilization management policies.
- Ensure that coverage denials based on medical necessity be reviewed by health care professionals with relevant expertise before a denial can be issued.
Physician groups welcomed the changes. In a statement, the American Medical Association said that an initial reading of the rule suggested CMS had “taken important steps toward right-sizing the prior authorization process.”
The Medical Group Management Association praised CMS in a statement for having limited “dangerous disruptions and delays to necessary patient care” resulting from the cumbersome processes of prior approval. With the new rules, CMS will provide greater consistency across Advantage plans as well as traditional Medicare, said Anders Gilberg, MGMA’s senior vice president of government affairs, in a statement.
Peer consideration
The final rule did disappoint physician groups in one key way. CMS rebuffed requests to have CMS require Advantage plans to use reviewers of the same specialty as treating physicians in handling disputes about prior authorization. CMS said it expects plans to exercise judgment in finding reviewers with “sufficient expertise to make an informed and supportable decision.”
“In some instances, we expect that plans will use a physician or other health care professional of the same specialty or subspecialty as the treating physician,” CMS said. “In other instances, we expect that plans will utilize a reviewer with specialized training, certification, or clinical experience in the applicable field of medicine.”
Medicare Advantage marketing ‘sowing confusion’
With this final rule, CMS also sought to protect consumers from “potentially misleading marketing practices” used in promoting Medicare Advantage and Part D prescription drug plans.
The agency said it had received complaints about people who have received official-looking promotional materials for Medicare that directed them not to government sources of information but to Medicare Advantage and Part D plans or their agents and brokers.
Ads now must mention a specific plan name, and they cannot use the Medicare name, CMS logo, Medicare card, or other government information in a misleading way, CMS said.
“CMS can see no value or purpose in a non-governmental entity’s use of the Medicare logo or HHS logo except for the express purpose of sowing confusion and misrepresenting itself as the government,” the agency said.
A version of this article first appeared on Medscape.com.
Study highlights potential skin cancer risk of UV nail polish dryers
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Results of a study recently published in Nature Communications suggests that According to two experts, these findings raise concerns regarding the safety of frequent use of these nail dryers.
In the study, human and mouse cells were exposed to radiation from UV nail dryers. Exposing human and mice skin cells to UVA light for 20 minutes resulted in the death of 20%-30% of cells; three consecutive 20-minute sessions resulted in the death of 65%-70% of cells. Additionally, surviving cells suffered oxidative damage to their DNA and mitochondria, with mutational patterns similar to those seen in skin cancer, study investigator Maria Zhivagui, PhD, of the University of California, San Diego, and associates reported.
“This study showed that irradiation of human and mouse cell lines using UV nail polish dryers resulted in DNA damage and genome mutations,” Shari Lipner, MD, PhD, director of the nail division at New York–Presbyterian Hospital/Weill Cornell Medicine, New York, said in an interview. The study “ties together exposure to UV light from nail polish dryers and genetic mutations that are associated with skin cancers,” added Dr. Lipner, who was not involved with the study.
UV nail lamps are commonly used to dry and harden gel nail polish formulas. Often referred to as “mini tanning beds,” these devices emit UVA radiation, classified as a Group 1 Carcinogen by the International Agency for Research on Cancer.
“Both UVA and UVB are main drivers of both melanoma and keratinocyte carcinomas (basal cell carcinoma and squamous cell carcinoma),” said Anthony Rossi, MD, a dermatologic surgeon at Memorial Sloan Kettering Cancer Center, New York, who was also not a study investigator. UV irradiance “produces DNA mutations that are specific to forming types of skin cancer,” he said in an interview.
UVA wavelengths commonly used in nail dryers can penetrate all layers of the epidermis, the top layer of the skin, potentially affecting stem cells in the skin, according to the study.
Dr. Lipner noted that “there have been several case reports of patients with histories of gel manicures using UV nail polish dryers who later developed squamous cell carcinomas on the dorsal hands, fingers, and nails, and articles describing high UV emissions from nail polish dryers, but the direct connection between UV dryers and skin cancer development was tenuous.” The first of its kind, the new study investigated the impact of UV nail drying devices at a cellular level.
The results of this study, in combination with previous case reports suggesting the development of skin cancers following UVA dryer use, raise concern regarding the safety of these commonly used devices. The study, the authors wrote, “does not provide direct evidence for an increased cancer risk in human beings,” but their findings and “prior evidence strongly suggest that radiation emitted by UV nail polish dryers may cause cancers of the hand and that UV nail polish dryers, similar to tanning beds, may increase the risk of early onset skin cancer.”
Dr. Rossi said that, “while this study shows that the UV exposure does affect human cells and causes mutations, the study was not done in vivo in human beings, so further studies are needed to know at what dose and frequency gel manicures would be needed to cause detrimental effects.” However, for people who regularly receive gel manicures involving UV nail dryers, both Dr. Lipner and Dr. Rossi recommend applying a broad-spectrum sunscreen to protect the dorsal hands, fingertips, and skin surrounding the nails, or wearing UV-protective gloves.
The study was supported by an Alfred B. Sloan Research Fellowship to one of the authors and grants from the National Institutes of Health to two authors. One author reported being a compensated consultant and having an equity interest in io9. Dr. Lipner and Dr. Rossi reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM NATURE COMMUNICATIONS