User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
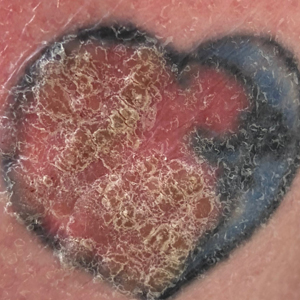
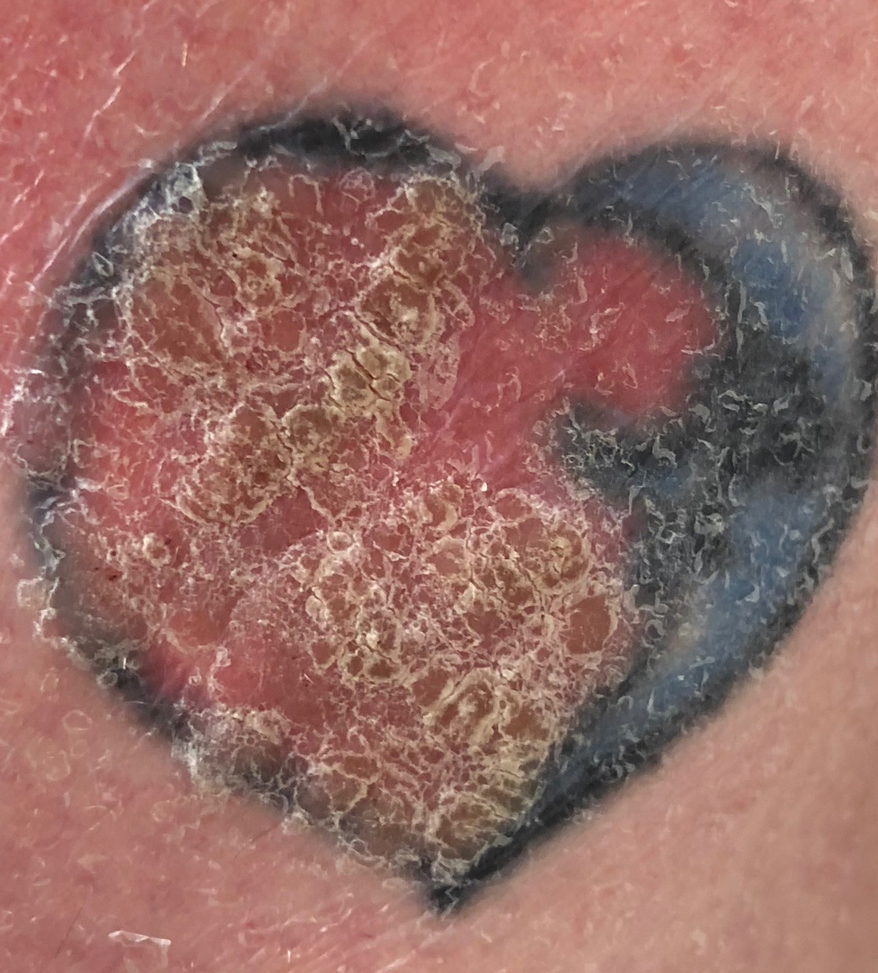
Verruca Vulgaris Arising Within the Red Portion of a Multicolored Tattoo
To the Editor:
The art of tattooing continues to gain popularity in the 21st century, albeit with accompanying hazards.1 Reported adverse reactions to tattoos include infections, tumors, and hypersensitivity and granulomatous reactions.2 Various infectious agents may involve tattoos, including human papillomavirus (HPV), molluscum contagiosum, herpes simplex virus, hepatitis C virus, tuberculoid and nontuberculoid mycobacteria, and Staphylococcus aureus.2 Verruca vulgaris infrequently has been reported to develop in tattoos.3,4 Previously reported cases of verruca in tattoos suggest a predilection for blue or black pigment.1-5 We report a case of verruca vulgaris occurring within the red-inked areas of a tattoo that first appeared approximately 18 years after the initial tattoo placement.
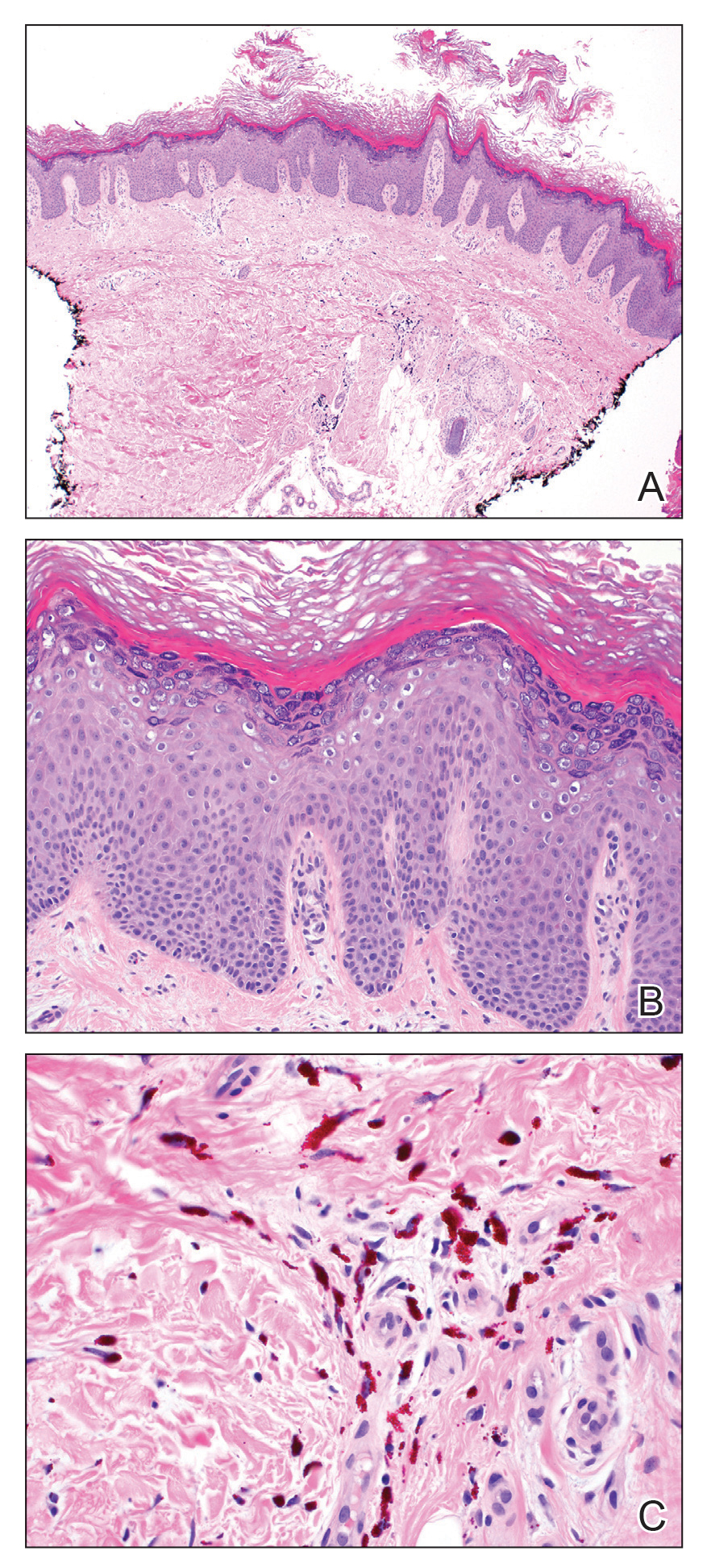
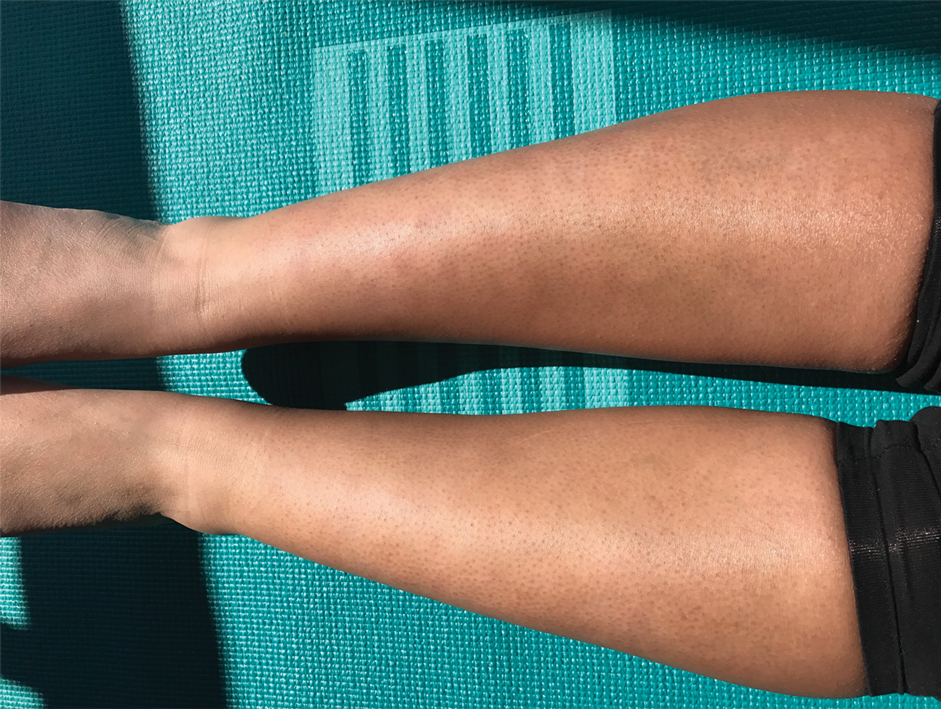
A 44-year-old woman presented with erythema, induration, and irritation of a tattoo on the left leg of 2 years’ duration. The tattoo initially was inscribed more than 20 years prior. The patient had a history of type 2 diabetes mellitus and chronic obstructive pulmonary disease. She reported no prior trauma to the area, prior rash or irritation, or similar changes to her other tattoos, including those with red ink. The affected tattoo was inscribed at a separate time from the other tattoos. Physical examination of the irritated tattoo revealed hyperkeratotic papules with firm scaling in the zone of dermal red pigment (Figure 1). Notable nodularity or deep induration was not present. The clinical differential diagnosis included a hypersensitivity reaction to red tattoo ink, sarcoidosis, and an infectious process, such as an atypical mycobacterial infection. A punch biopsy demonstrated papillomatous epidermal hyperplasia with hyperkeratosis, focal parakeratosis, and frequent vacuolization of keratinocytes with enlarged keratohyalin granules, diagnostic of verruca vulgaris (Figure 2). Of note, the patient did not have clinically apparent viral warts elsewhere on physical examination. The patient was successfully managedwith a combination of 2 treatments of intralesional Candida antigen and 3 treatments of cryotherapy with resolution of most lesions over the course of 8 months. Over the following several months, the patient applied topical salicylic acid, which led to the resolution of the remaining lesions. The verrucae had not recurred 19 months after the initial presentation.
The development of verruca vulgaris within a tattoo may occur secondary to various mechanisms of HPV inoculation, including introduction of the virus through contaminated ink, the tattoo artist’s saliva, autoinoculation, or koebnerization of a pre-existing verruca vulgaris.4 Local immune system dysregulation secondary to tattoo ink also has been proposed as a mechanism for HPV infection in this setting.1,5 The contents of darker tattoo pigments may promote formation of reactive oxygen species inducing local immunocompromise.5
The pathogenic mechanism was elusive in our patient. Although the localization of verruca vulgaris to the zones of red pigment may be merely coincidental, this phenomenon raised suspicion for direct inoculation via contaminated red ink. The patient’s other red ink–containing tattoos that were inscribed separately were spared, compatible with contamination of the red ink used for the affected tattoo. However, the delayed onset of nearly 2 decades was exceptional, given the shorter previously reported latencies ranging from months to 10 years.4 Autoinoculation or koebnerization is plausible, though greater involvement of nonred pigments would be expected as well as a briefer latency. Finally, the possibility of local immune dysregulation seemed feasible, given the slow evolution of the lesions largely restricted to one pigment type.
We report a case of verruca vulgaris within the red area of a multicolored tattoo that occurred approximately 18 years after tattoo placement. This case highlights a rare presentation of an infectious agent that may complicate tattoos. Both predilection for red pigment rather than black or blue pigment and the long latency period raised interesting questions regarding pathogenesis. Confirmatory biopsy enables effective management of this tattoo complication.
- Huynh TN, Jackson JD, Brodell RT. Tattoo and vaccination sites: possible nest for opportunistic infections, tumors, and dysimmune reactions. Clin Dermatol. 2014;32:678-684.
- Wenzel SM, Rittmann I, Landthaler M, et al. Adverse reactions after tattooing: review of the literature and comparison to results of a survey. Dermatology. 2013;226:138-147.
- Trefzer U, Schmollack K, Stockfleth E, et al. Verrucae in a multicolored decorative tattoo. J Am Acad Dermatol. 2004;50:478-479.
- Wanat KA, Tyring S, Rady P, et al. Human papillomavirus type 27 associated with multiple verruca within a tattoo: report of a case and review of the literature. Int J Dermatol. 2014;53:882-884.
- Ramey K, Ibrahim J, Brodell RT. Verruca localization predominately in black tattoo ink: a retrospective case series. J Eur Acad Dermatol Venereol. 2016;30:E34-E36.
To the Editor:
The art of tattooing continues to gain popularity in the 21st century, albeit with accompanying hazards.1 Reported adverse reactions to tattoos include infections, tumors, and hypersensitivity and granulomatous reactions.2 Various infectious agents may involve tattoos, including human papillomavirus (HPV), molluscum contagiosum, herpes simplex virus, hepatitis C virus, tuberculoid and nontuberculoid mycobacteria, and Staphylococcus aureus.2 Verruca vulgaris infrequently has been reported to develop in tattoos.3,4 Previously reported cases of verruca in tattoos suggest a predilection for blue or black pigment.1-5 We report a case of verruca vulgaris occurring within the red-inked areas of a tattoo that first appeared approximately 18 years after the initial tattoo placement.
A 44-year-old woman presented with erythema, induration, and irritation of a tattoo on the left leg of 2 years’ duration. The tattoo initially was inscribed more than 20 years prior. The patient had a history of type 2 diabetes mellitus and chronic obstructive pulmonary disease. She reported no prior trauma to the area, prior rash or irritation, or similar changes to her other tattoos, including those with red ink. The affected tattoo was inscribed at a separate time from the other tattoos. Physical examination of the irritated tattoo revealed hyperkeratotic papules with firm scaling in the zone of dermal red pigment (Figure 1). Notable nodularity or deep induration was not present. The clinical differential diagnosis included a hypersensitivity reaction to red tattoo ink, sarcoidosis, and an infectious process, such as an atypical mycobacterial infection. A punch biopsy demonstrated papillomatous epidermal hyperplasia with hyperkeratosis, focal parakeratosis, and frequent vacuolization of keratinocytes with enlarged keratohyalin granules, diagnostic of verruca vulgaris (Figure 2). Of note, the patient did not have clinically apparent viral warts elsewhere on physical examination. The patient was successfully managedwith a combination of 2 treatments of intralesional Candida antigen and 3 treatments of cryotherapy with resolution of most lesions over the course of 8 months. Over the following several months, the patient applied topical salicylic acid, which led to the resolution of the remaining lesions. The verrucae had not recurred 19 months after the initial presentation.


The development of verruca vulgaris within a tattoo may occur secondary to various mechanisms of HPV inoculation, including introduction of the virus through contaminated ink, the tattoo artist’s saliva, autoinoculation, or koebnerization of a pre-existing verruca vulgaris.4 Local immune system dysregulation secondary to tattoo ink also has been proposed as a mechanism for HPV infection in this setting.1,5 The contents of darker tattoo pigments may promote formation of reactive oxygen species inducing local immunocompromise.5
The pathogenic mechanism was elusive in our patient. Although the localization of verruca vulgaris to the zones of red pigment may be merely coincidental, this phenomenon raised suspicion for direct inoculation via contaminated red ink. The patient’s other red ink–containing tattoos that were inscribed separately were spared, compatible with contamination of the red ink used for the affected tattoo. However, the delayed onset of nearly 2 decades was exceptional, given the shorter previously reported latencies ranging from months to 10 years.4 Autoinoculation or koebnerization is plausible, though greater involvement of nonred pigments would be expected as well as a briefer latency. Finally, the possibility of local immune dysregulation seemed feasible, given the slow evolution of the lesions largely restricted to one pigment type.
We report a case of verruca vulgaris within the red area of a multicolored tattoo that occurred approximately 18 years after tattoo placement. This case highlights a rare presentation of an infectious agent that may complicate tattoos. Both predilection for red pigment rather than black or blue pigment and the long latency period raised interesting questions regarding pathogenesis. Confirmatory biopsy enables effective management of this tattoo complication.
To the Editor:
The art of tattooing continues to gain popularity in the 21st century, albeit with accompanying hazards.1 Reported adverse reactions to tattoos include infections, tumors, and hypersensitivity and granulomatous reactions.2 Various infectious agents may involve tattoos, including human papillomavirus (HPV), molluscum contagiosum, herpes simplex virus, hepatitis C virus, tuberculoid and nontuberculoid mycobacteria, and Staphylococcus aureus.2 Verruca vulgaris infrequently has been reported to develop in tattoos.3,4 Previously reported cases of verruca in tattoos suggest a predilection for blue or black pigment.1-5 We report a case of verruca vulgaris occurring within the red-inked areas of a tattoo that first appeared approximately 18 years after the initial tattoo placement.
A 44-year-old woman presented with erythema, induration, and irritation of a tattoo on the left leg of 2 years’ duration. The tattoo initially was inscribed more than 20 years prior. The patient had a history of type 2 diabetes mellitus and chronic obstructive pulmonary disease. She reported no prior trauma to the area, prior rash or irritation, or similar changes to her other tattoos, including those with red ink. The affected tattoo was inscribed at a separate time from the other tattoos. Physical examination of the irritated tattoo revealed hyperkeratotic papules with firm scaling in the zone of dermal red pigment (Figure 1). Notable nodularity or deep induration was not present. The clinical differential diagnosis included a hypersensitivity reaction to red tattoo ink, sarcoidosis, and an infectious process, such as an atypical mycobacterial infection. A punch biopsy demonstrated papillomatous epidermal hyperplasia with hyperkeratosis, focal parakeratosis, and frequent vacuolization of keratinocytes with enlarged keratohyalin granules, diagnostic of verruca vulgaris (Figure 2). Of note, the patient did not have clinically apparent viral warts elsewhere on physical examination. The patient was successfully managedwith a combination of 2 treatments of intralesional Candida antigen and 3 treatments of cryotherapy with resolution of most lesions over the course of 8 months. Over the following several months, the patient applied topical salicylic acid, which led to the resolution of the remaining lesions. The verrucae had not recurred 19 months after the initial presentation.


The development of verruca vulgaris within a tattoo may occur secondary to various mechanisms of HPV inoculation, including introduction of the virus through contaminated ink, the tattoo artist’s saliva, autoinoculation, or koebnerization of a pre-existing verruca vulgaris.4 Local immune system dysregulation secondary to tattoo ink also has been proposed as a mechanism for HPV infection in this setting.1,5 The contents of darker tattoo pigments may promote formation of reactive oxygen species inducing local immunocompromise.5
The pathogenic mechanism was elusive in our patient. Although the localization of verruca vulgaris to the zones of red pigment may be merely coincidental, this phenomenon raised suspicion for direct inoculation via contaminated red ink. The patient’s other red ink–containing tattoos that were inscribed separately were spared, compatible with contamination of the red ink used for the affected tattoo. However, the delayed onset of nearly 2 decades was exceptional, given the shorter previously reported latencies ranging from months to 10 years.4 Autoinoculation or koebnerization is plausible, though greater involvement of nonred pigments would be expected as well as a briefer latency. Finally, the possibility of local immune dysregulation seemed feasible, given the slow evolution of the lesions largely restricted to one pigment type.
We report a case of verruca vulgaris within the red area of a multicolored tattoo that occurred approximately 18 years after tattoo placement. This case highlights a rare presentation of an infectious agent that may complicate tattoos. Both predilection for red pigment rather than black or blue pigment and the long latency period raised interesting questions regarding pathogenesis. Confirmatory biopsy enables effective management of this tattoo complication.
- Huynh TN, Jackson JD, Brodell RT. Tattoo and vaccination sites: possible nest for opportunistic infections, tumors, and dysimmune reactions. Clin Dermatol. 2014;32:678-684.
- Wenzel SM, Rittmann I, Landthaler M, et al. Adverse reactions after tattooing: review of the literature and comparison to results of a survey. Dermatology. 2013;226:138-147.
- Trefzer U, Schmollack K, Stockfleth E, et al. Verrucae in a multicolored decorative tattoo. J Am Acad Dermatol. 2004;50:478-479.
- Wanat KA, Tyring S, Rady P, et al. Human papillomavirus type 27 associated with multiple verruca within a tattoo: report of a case and review of the literature. Int J Dermatol. 2014;53:882-884.
- Ramey K, Ibrahim J, Brodell RT. Verruca localization predominately in black tattoo ink: a retrospective case series. J Eur Acad Dermatol Venereol. 2016;30:E34-E36.
- Huynh TN, Jackson JD, Brodell RT. Tattoo and vaccination sites: possible nest for opportunistic infections, tumors, and dysimmune reactions. Clin Dermatol. 2014;32:678-684.
- Wenzel SM, Rittmann I, Landthaler M, et al. Adverse reactions after tattooing: review of the literature and comparison to results of a survey. Dermatology. 2013;226:138-147.
- Trefzer U, Schmollack K, Stockfleth E, et al. Verrucae in a multicolored decorative tattoo. J Am Acad Dermatol. 2004;50:478-479.
- Wanat KA, Tyring S, Rady P, et al. Human papillomavirus type 27 associated with multiple verruca within a tattoo: report of a case and review of the literature. Int J Dermatol. 2014;53:882-884.
- Ramey K, Ibrahim J, Brodell RT. Verruca localization predominately in black tattoo ink: a retrospective case series. J Eur Acad Dermatol Venereol. 2016;30:E34-E36.
Practice Points
- Various adverse reactions and infectious agents may involve tattoos.
- Verruca vulgaris may affect tattoos in a color-restricted manner and demonstrate latency of many years after tattoo placement.
- Timely diagnosis of the tattoo-involving process, confirmed by biopsy, allows for appropriate management.
Pediatric-Onset Refractory Lupus Erythematosus Panniculitis Treated With Rituximab
To the Editor:
Lupus erythematosus panniculitis (LEP) is rare in the pediatric population. It can be difficult to manage, as patients may not respond to conventional treatments including hydroxychloroquine and prednisone. We report the use of rituximab in the treatment of a 20-year-old woman with LEP of the face, legs, and arms that was refractory to standard treatments. She also had a history of hemophagocytic lymphohistiocytosis (HLH). Further studies are warranted to determine the role of rituximab in the treatment of pediatric patients with LEP.
A 20-year-old woman with history of LEP and HLH initially presented with migratory violaceous nodules on the face 16 years prior to the current presentation. A skin biopsy 3 years after that initial presentation suggested a diagnosis of cutaneous lupus erythematosus. Six years later, numerous asymptomatic lesions appeared on the legs, predominantly on the calves; she was successfully treated with hydroxychloroquine and high-dose prednisone. Four years prior to the current presentation, a febrile illness prompted discontinuation of hydroxychloroquine and hospitalization, where she was first was diagnosed with HLH; she achieved remission with cyclosporine. At the current presentation, she continued to have persistent violaceous lesions on the face, lower arms, and legs with underlying nodularity (Figure 1). Skin biopsies revealed LEP and were less suggestive of HLH. She was restarted on hydroxychloroquine, which did not adequately control the disease. Rheumatologic workup was only notable for an antinuclear antibody titer of 1:80 (reference range, <1:80) in a speckled pattern.
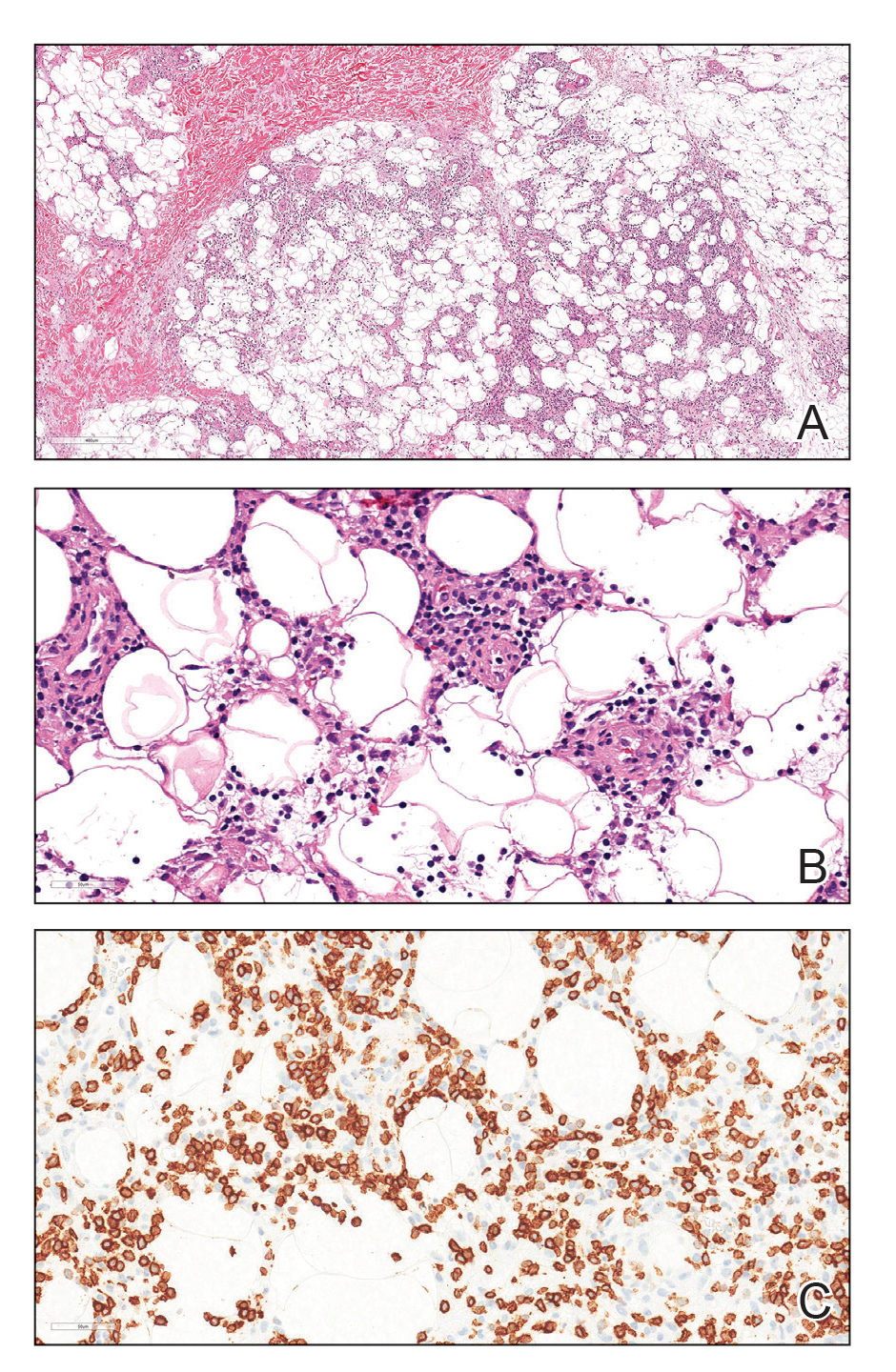
Due to the refractory nature of her condition, continued lesion development despite standard treatment, and concerns of possible scarring, we considered a trial of rituximab. Because HLH and LEP can mimic subcutaneous T-cell lymphoma, another skin biopsy was performed, which revealed a deep dermal and subcutaneous lymphohistiocytic infiltrate composed of predominantly CD3+ T cells with a mixed population of CD4+ and CD8+ cells (Figure 2). There was no evidence of transformation into lymphoma. Pathologic findings were most compatible with LEP rather than an HLH-associated panniculitis due to the lack of definitive phagocytosis. She received rituximab using body surface area–based dosing at 375 mg/m2. CD19 levels decreased to undetectable levels after the first dose. Rituximab was dosed based on clinical response; she tolerated treatment well and experienced considerable improvement in the number of lesions following completion of 4 doses at weeks 0, 1, 5, and 7 (Figure 3). She developed a flare at 7 months and improved again after another dose of rituximab.

Lupus erythematosus panniculitis is a rare variant of lupus erythematosus with an average age of presentation between 30 and 60 years.1 In children, LEP presents as recurrent subcutaneous nodules and plaques, commonly involving the face and upper arms.1,2 Long-term sequelae include local swelling and skin atrophy.3 Conventional treatment options for pediatric patients include hydroxychloroquine and corticosteroids.1 Management can be challenging due to the lack of response to conventional treatments as well as the chronic progressive nature of LEP.2 In refractory cases, cyclosporine, azathioprine, sulfones, thalidomide, mycophenolate mofetil, and cyclophosphamide are alternative treatment options.1-4
Rituximab, a chimeric monoclonal antibody targeting B-cell surface marker CD20, results in depletion of mature B cells. Use of rituximab for LEP has been described in multiple case reports involving an 8-year-old boy, 22-year-old girl, and 2 middle-aged women.2-4 In addition, a recently published case series of 4 patients with childhood-onset refractory LEP described improvement of disease activity with rituximab.5 It is important to rule out subcutaneous T-cell lymphoma before treatment with rituximab, as its histopathology can closely resemble that seen in LEP and HLH-associated cytophagic histiocytic panniculitis.1,6
Rituximab may be an effective treatment option in pediatric patients with refractory LEP. Larger studies on the use of rituximab in the pediatric population are necessary.
- Weingartner JS, Zedek DC, Burkhart CN, et al. Lupus erythematosus panniculitis in children: report of three cases and review of previously reported cases. Pediatr Dermatol. 2011;29:169-176.
- Moreno-Suárez F, Pulpillo-Ruiz Á. Rituximab for the treatment of lupus erythematosus panniculitis. Dermatol Ther. 2013;26:415-418.
- Guissa VR, Trudes G, Jesus AA, et al. Lupus erythematosus panniculitis in children and adolescents. Acta Reumatol Port. 2012;37:82-85.
- Mcardle A, Baker JF. A case of “refractory” lupus erythematosus profundus responsive to rituximab. Clin Rheumatol. 2009;28:745-746.
- Correll CK, Miller DD, Maguiness SM. Treatment of childhood-onset lupus erythematosus panniculitis with rituximab. JAMA Dermatol. 2020;156:566-569.
- Aronson IK, Worobec SM. Cytophagic histiocytic panniculitis and hemophagocytic lymphohistiocytosis: an overview. Dermatol Ther. 2010;23:389-402.
To the Editor:
Lupus erythematosus panniculitis (LEP) is rare in the pediatric population. It can be difficult to manage, as patients may not respond to conventional treatments including hydroxychloroquine and prednisone. We report the use of rituximab in the treatment of a 20-year-old woman with LEP of the face, legs, and arms that was refractory to standard treatments. She also had a history of hemophagocytic lymphohistiocytosis (HLH). Further studies are warranted to determine the role of rituximab in the treatment of pediatric patients with LEP.
A 20-year-old woman with history of LEP and HLH initially presented with migratory violaceous nodules on the face 16 years prior to the current presentation. A skin biopsy 3 years after that initial presentation suggested a diagnosis of cutaneous lupus erythematosus. Six years later, numerous asymptomatic lesions appeared on the legs, predominantly on the calves; she was successfully treated with hydroxychloroquine and high-dose prednisone. Four years prior to the current presentation, a febrile illness prompted discontinuation of hydroxychloroquine and hospitalization, where she was first was diagnosed with HLH; she achieved remission with cyclosporine. At the current presentation, she continued to have persistent violaceous lesions on the face, lower arms, and legs with underlying nodularity (Figure 1). Skin biopsies revealed LEP and were less suggestive of HLH. She was restarted on hydroxychloroquine, which did not adequately control the disease. Rheumatologic workup was only notable for an antinuclear antibody titer of 1:80 (reference range, <1:80) in a speckled pattern.

Due to the refractory nature of her condition, continued lesion development despite standard treatment, and concerns of possible scarring, we considered a trial of rituximab. Because HLH and LEP can mimic subcutaneous T-cell lymphoma, another skin biopsy was performed, which revealed a deep dermal and subcutaneous lymphohistiocytic infiltrate composed of predominantly CD3+ T cells with a mixed population of CD4+ and CD8+ cells (Figure 2). There was no evidence of transformation into lymphoma. Pathologic findings were most compatible with LEP rather than an HLH-associated panniculitis due to the lack of definitive phagocytosis. She received rituximab using body surface area–based dosing at 375 mg/m2. CD19 levels decreased to undetectable levels after the first dose. Rituximab was dosed based on clinical response; she tolerated treatment well and experienced considerable improvement in the number of lesions following completion of 4 doses at weeks 0, 1, 5, and 7 (Figure 3). She developed a flare at 7 months and improved again after another dose of rituximab.


Lupus erythematosus panniculitis is a rare variant of lupus erythematosus with an average age of presentation between 30 and 60 years.1 In children, LEP presents as recurrent subcutaneous nodules and plaques, commonly involving the face and upper arms.1,2 Long-term sequelae include local swelling and skin atrophy.3 Conventional treatment options for pediatric patients include hydroxychloroquine and corticosteroids.1 Management can be challenging due to the lack of response to conventional treatments as well as the chronic progressive nature of LEP.2 In refractory cases, cyclosporine, azathioprine, sulfones, thalidomide, mycophenolate mofetil, and cyclophosphamide are alternative treatment options.1-4
Rituximab, a chimeric monoclonal antibody targeting B-cell surface marker CD20, results in depletion of mature B cells. Use of rituximab for LEP has been described in multiple case reports involving an 8-year-old boy, 22-year-old girl, and 2 middle-aged women.2-4 In addition, a recently published case series of 4 patients with childhood-onset refractory LEP described improvement of disease activity with rituximab.5 It is important to rule out subcutaneous T-cell lymphoma before treatment with rituximab, as its histopathology can closely resemble that seen in LEP and HLH-associated cytophagic histiocytic panniculitis.1,6
Rituximab may be an effective treatment option in pediatric patients with refractory LEP. Larger studies on the use of rituximab in the pediatric population are necessary.
To the Editor:
Lupus erythematosus panniculitis (LEP) is rare in the pediatric population. It can be difficult to manage, as patients may not respond to conventional treatments including hydroxychloroquine and prednisone. We report the use of rituximab in the treatment of a 20-year-old woman with LEP of the face, legs, and arms that was refractory to standard treatments. She also had a history of hemophagocytic lymphohistiocytosis (HLH). Further studies are warranted to determine the role of rituximab in the treatment of pediatric patients with LEP.
A 20-year-old woman with history of LEP and HLH initially presented with migratory violaceous nodules on the face 16 years prior to the current presentation. A skin biopsy 3 years after that initial presentation suggested a diagnosis of cutaneous lupus erythematosus. Six years later, numerous asymptomatic lesions appeared on the legs, predominantly on the calves; she was successfully treated with hydroxychloroquine and high-dose prednisone. Four years prior to the current presentation, a febrile illness prompted discontinuation of hydroxychloroquine and hospitalization, where she was first was diagnosed with HLH; she achieved remission with cyclosporine. At the current presentation, she continued to have persistent violaceous lesions on the face, lower arms, and legs with underlying nodularity (Figure 1). Skin biopsies revealed LEP and were less suggestive of HLH. She was restarted on hydroxychloroquine, which did not adequately control the disease. Rheumatologic workup was only notable for an antinuclear antibody titer of 1:80 (reference range, <1:80) in a speckled pattern.

Due to the refractory nature of her condition, continued lesion development despite standard treatment, and concerns of possible scarring, we considered a trial of rituximab. Because HLH and LEP can mimic subcutaneous T-cell lymphoma, another skin biopsy was performed, which revealed a deep dermal and subcutaneous lymphohistiocytic infiltrate composed of predominantly CD3+ T cells with a mixed population of CD4+ and CD8+ cells (Figure 2). There was no evidence of transformation into lymphoma. Pathologic findings were most compatible with LEP rather than an HLH-associated panniculitis due to the lack of definitive phagocytosis. She received rituximab using body surface area–based dosing at 375 mg/m2. CD19 levels decreased to undetectable levels after the first dose. Rituximab was dosed based on clinical response; she tolerated treatment well and experienced considerable improvement in the number of lesions following completion of 4 doses at weeks 0, 1, 5, and 7 (Figure 3). She developed a flare at 7 months and improved again after another dose of rituximab.


Lupus erythematosus panniculitis is a rare variant of lupus erythematosus with an average age of presentation between 30 and 60 years.1 In children, LEP presents as recurrent subcutaneous nodules and plaques, commonly involving the face and upper arms.1,2 Long-term sequelae include local swelling and skin atrophy.3 Conventional treatment options for pediatric patients include hydroxychloroquine and corticosteroids.1 Management can be challenging due to the lack of response to conventional treatments as well as the chronic progressive nature of LEP.2 In refractory cases, cyclosporine, azathioprine, sulfones, thalidomide, mycophenolate mofetil, and cyclophosphamide are alternative treatment options.1-4
Rituximab, a chimeric monoclonal antibody targeting B-cell surface marker CD20, results in depletion of mature B cells. Use of rituximab for LEP has been described in multiple case reports involving an 8-year-old boy, 22-year-old girl, and 2 middle-aged women.2-4 In addition, a recently published case series of 4 patients with childhood-onset refractory LEP described improvement of disease activity with rituximab.5 It is important to rule out subcutaneous T-cell lymphoma before treatment with rituximab, as its histopathology can closely resemble that seen in LEP and HLH-associated cytophagic histiocytic panniculitis.1,6
Rituximab may be an effective treatment option in pediatric patients with refractory LEP. Larger studies on the use of rituximab in the pediatric population are necessary.
- Weingartner JS, Zedek DC, Burkhart CN, et al. Lupus erythematosus panniculitis in children: report of three cases and review of previously reported cases. Pediatr Dermatol. 2011;29:169-176.
- Moreno-Suárez F, Pulpillo-Ruiz Á. Rituximab for the treatment of lupus erythematosus panniculitis. Dermatol Ther. 2013;26:415-418.
- Guissa VR, Trudes G, Jesus AA, et al. Lupus erythematosus panniculitis in children and adolescents. Acta Reumatol Port. 2012;37:82-85.
- Mcardle A, Baker JF. A case of “refractory” lupus erythematosus profundus responsive to rituximab. Clin Rheumatol. 2009;28:745-746.
- Correll CK, Miller DD, Maguiness SM. Treatment of childhood-onset lupus erythematosus panniculitis with rituximab. JAMA Dermatol. 2020;156:566-569.
- Aronson IK, Worobec SM. Cytophagic histiocytic panniculitis and hemophagocytic lymphohistiocytosis: an overview. Dermatol Ther. 2010;23:389-402.
- Weingartner JS, Zedek DC, Burkhart CN, et al. Lupus erythematosus panniculitis in children: report of three cases and review of previously reported cases. Pediatr Dermatol. 2011;29:169-176.
- Moreno-Suárez F, Pulpillo-Ruiz Á. Rituximab for the treatment of lupus erythematosus panniculitis. Dermatol Ther. 2013;26:415-418.
- Guissa VR, Trudes G, Jesus AA, et al. Lupus erythematosus panniculitis in children and adolescents. Acta Reumatol Port. 2012;37:82-85.
- Mcardle A, Baker JF. A case of “refractory” lupus erythematosus profundus responsive to rituximab. Clin Rheumatol. 2009;28:745-746.
- Correll CK, Miller DD, Maguiness SM. Treatment of childhood-onset lupus erythematosus panniculitis with rituximab. JAMA Dermatol. 2020;156:566-569.
- Aronson IK, Worobec SM. Cytophagic histiocytic panniculitis and hemophagocytic lymphohistiocytosis: an overview. Dermatol Ther. 2010;23:389-402.
Practice Points
- Lupus erythematosus panniculitis (LEP) is rare in the pediatric population and often is difficult to treat.
- Rituximab can be an effective treatment option for refractory LEP.
- Before the initiation of rituximab, a biopsy is warranted to rule out subcutaneous T-cell lymphoma, which can mimic LEP and hemophagocytic lymphohistiocytosis–associated panniculitis.
Volunteer Opportunities Within Dermatology: More than Skin Deep
The adage “so much to do, so little time” aptly describes the daily challenges facing dermatologists and dermatology residents. The time and attention required by direct patient care, writing notes, navigating electronic health records, and engaging in education and research as well as family commitments can drain even the most tireless clinician. In addition, dermatologists are expected to play a critical role in clinic and practice management to successfully curate an online presence and adapt their skills to successfully manage a teledermatology practice. Coupled with the time spent socializing with friends or colleagues and time for personal hobbies or exercise, it’s easy to see how sleep deprivation is common in many of our colleagues.
What’s being left out of these jam-packed schedules? Increasingly, it is the time and expertise dedicated to volunteering in our local communities. Two recent research letters highlighted how a dramatic increase in the number of research projects and publications is not mirrored by a similar increase in volunteer experiences as dermatology residency selection becomes more competitive.1,2
Although the rate of volunteerism among practicing dermatologists has yet to be studied, a brief review suggests a component of unmet dermatology need within our communities. It’s estimated that approximately 5% to 10% of all emergency department visits are for dermatologic concerns.3-5 In many cases, the reason for the visit is nonurgent and instead reflects a lack of other options for care. However, the need for dermatologists extends beyond the emergency department setting. A review of the prevalence of patients presenting for care to a group of regional free clinics found that 8% (N=5553) of all visitors sought care for dermatologic concerns.6 The benefit is not just for those seated on the examination table; research has shown that while many of the underlying factors resulting in physician burnout stem from systemic issues, participating in volunteer opportunities helps combat burnout in ourselves and our colleagues.7-9 Herein, opportunities that exist for dermatologists to reconnect with their communities, advocate for causes distinctive to the specialty, and care for neighbors most in need are highlighted.
Camp Wonder
Every year, children from across the United States living with chronic and debilitating skin conditions get the opportunity to join fellow campers and spend a week just being kids without the constant focus on being a patient. Camp Wonder’s founder and director, Francesca Tenconi, describes the camp as a place where kids “can form a community and can feel free to be themselves, without judgment, without stares. They get the chance to forget about their skin disease and be themselves” (oral communication, June 18, 2021). Tenconi and the camp’s cofounders and medical directors, Drs. Jenny Kim and Stefani Takahashi, envisioned the camp as a place for all campers regardless of their skin condition to feel safe and welcome. This overall mission guides camp leadership and staff every year over the course of the camp week where campers participate in a mix of traditional and nontraditional summer activities that are safe and accessible for all, from spending time in the pool to arts and crafts and a ropes course.
Camp Wonder is in its 21st year of hosting children and adolescents from across North America at its camp in Livermore, California. This year, Tenconi expects about 100 campers during the last week in July. Camp Wonder relies on medical staff volunteers to make the camp setting safe, inclusive, and fun. “Our dermatology residents and dermatology volunteers are a huge part of why we’re able to have camp,” said Tenconi. “A lot of our kids require very specific medical care throughout the week. We are able to provide this camp experience for them because we have this medical support system available, this specialized dermatology knowledge.” She also noted the benefit to the volunteers themselves, saying,“The feedback we get a lot from residents and dermatologists is that camp gave them a chance to understand the true-life impact of some of the skin diseases these kids and families are living with. Kids will open up to them and tell them how their disease has impacted them personally” (oral communication, June 18, 2021).
Volunteer medical providers help manage the medical needs of the campers beginning at check-in and work shifts in the infirmary as well as help with dispensing and administering medications, changing dressings, and applying ointments or other topical medications. When not assisting with medical care, medical staff can get to know the campers; help out with arts and crafts, games, sports, and other camp activities; and put on skits and plays for campers at nightly camp hangouts (Figure 1).

How to Get Involved
Visit the website (https://www.csdf.org/camp-wonder) for information on becoming a medical volunteer for 2022. Donations to help keep the camp running also are greatly appreciated, as attendance, including travel costs, is free for families through the Children’s Skin Disease Foundation. Finally, dermatologists can help by keeping their young patients with skin disease in mind as future campers. The camp welcomes kids from across the United States and Canada and invites questions from dermatologists and families on how to become a camper and what the experience is like.
Native American Health Services Rotation
Located in the southwestern United States, the Navajo Nation is North America’s largest Native American tribe by enrollment and resides on the largest reservation in the United States.10 Comprised of 27,000 square miles within portions of Arizona, New Mexico, and Utah, the reservation’s total area is greater than that of Massachusetts, Vermont, and New Hampshire combined.11 The reservation is home to an estimated 180,000 Navajo people, a population roughly the size of Salt Lake City, Utah. Yet, many homes on the reservation are without electricity, running water, telephones, or broadband access, and many roads on the reservation remain unpaved. Prior to the COVID-19 pandemic, 4 dermatology residents were selected each year to travel to this unique and remote location to work with the staff of the Chinle Comprehensive Health Care Facility (Chinle, Arizona), an Indian Health Service facility, as part of the American Academy of Dermatology (AAD)–sponsored Native American Health Services Resident Rotation (NAHSRR).
Dr. Lucinda Kohn, Assistant Professor of Dermatology at the University of Colorado and the director of the NAHSRR program discovered the value of this rotation firsthand as a dermatology resident. In 2017, she traveled to the area to spend 2 weeks serving within the community. “I went because of a personal connection. My husband is Native American, although not Navajo. I wanted to experience what it was like to provide dermatologic care for Native Americans. I found the Navajo people to be so friendly and so grateful for our care. The clinicians we worked with at Chinle were excited to have us share our expertise and to pass on their knowledge to us,” said Dr. Kohn (personal communication, June 24, 2021).
Rotating residents provide dermatologic care for the Navajo people and share their unique medical skill set to local primary care clinicians serving as preceptors. They also may have an opportunity to learn from Native healers about traditional Navajo beliefs and ceremonies used as part of a holistic approach to healing.
The program, similar to volunteer programs across the country, was put on hold during the height of the COVID-19 pandemic. “The Navajo nation witnessed a really tragic surge of COVID cases that required that limited medical resources be diverted to help cope with the pandemic,” says Dr. Kohn. “It really wasn’t safe for residents to travel to the reservation either, so the rotation had to be put on hold.” However, in April 2021, the health care staff of the Chinle Comprehensive Care Facility reached out to revive the program, which is now pending the green light from the AAD. It is unclear if or when AAD leadership will allow this rotation to restart. Dr. Kohn hopes to be able to start accepting new applications soon. “This rotation provides a wealth of benefits to all those involved, from the residents who get the chance to work with a unique population in need to the clinicians who gain a diverse understanding of dermatology treatment techniques. And of course, for the patients, who are so appreciative of the care they receive from our volunteers” (personal communication, June 25, 2021).
How to Get Involved
Dr. Kohn is happy to field questions regarding the rotation and requests for more information via email ([email protected]). Residents interested in this program also may reach out to the AAD’s Education and Volunteers Abroad Committee to express interest in the NAHSRR program’s reinstatement.
Destination Healthy Skin
Since 2017, the Skin Cancer Foundation’s Destination Healthy Skin (DHS) RV has been the setting for more than 3800 free skin cancer screenings provided by volunteers within underserved populations across the United States (Figure 2). After a year hiatus due to the pandemic, DHS hit the road again, starting in New York City on August 1 to 3, 2021. From there, the DHS RV will traverse the country in one large loop, starting with visits to large and small cities in the Midwest and the West Coast. Following a visit to San Diego, California, in early October, the RV will turn east, with stops in Arizona, Texas, and several southern states before ending in Philadelphia, Pennsylvania. Dr. Elizabeth Hale, Senior Vice President of the Skin Cancer Foundation, feels that increasing awareness of the importance of regular skin cancer screening for those at risk is more important than ever. “We know that many people in the past year put routine cancer screening on the back burner, but we’re beginning to appreciate that this has led to significant delays in skin cancer diagnosis and potentially more significant disease when cases are diagnosed.” Dr. Hale noted that as the country continues to return to a degree of normalcy, the backlog of patients now seeking their routine screening has led to longer wait times. She expects DHS may offer some relief. “There are no appointments necessary. If the RV is close to their hometown, patients have an advantage in being able to be seen first come, first served, without having to wait for an appointment or make sure their insurance is accepted. It’s a free screening that can increase access to dermatologists” (personal communication, June 21, 2021).

The program’s organizers acknowledge that DHS is not a long-term solution for improving dermatology access in the United States and recognize that more needs to be done to raise awareness, both of the value that screenings can provide and the importance of sun-protective behavior. “This is an important first step,” says Dr. Hale. “It’s important that we disseminate that no one is immune to skin cancer. It’s about education, and this is a tool to educate patients that everyone should have a skin check once a year, regardless of where you live or what your skin type is” (personal communication, June 21, 2021).
Volunteer dermatologists are needed to assist with screenings when the DHS RV arrives in their community. Providers complete a screening form identifying any concerning lesions and can document specific lesions using the patient’s cell phone. Following the screenings, participating dermatologists are welcome to invite participants to make appointments at their practices or suggest local clinics for follow-up care.
How to Get Involved
The schedule for this year’s screening events can be found online (https://www.skincancer.org/early-detection/destination-healthy-skin/). Consider volunteering (https://www.skincancer.org/early-detection/destination-healthy-skin/physician-volunteers/) or helping to raise awareness by reaching out to local dermatology societies or free clinics in your area. Residents and physician’s assistants are welcome to volunteer as well, as long as they are under the on-site supervision of a board-certified dermatologist.
Final Thoughts
As medical professionals, we all recognize there are valuable contributions we can make to groups and organizations that need our help. The stresses and pressure of work and everyday life can make finding the time to offer that help seem impossible. Although it may seem counterintuitive, volunteering our time to help others can help us better navigate the professional burnout that many medical professionals experience today.
- Ezekor M, Pona A, Cline A, et al. An increasing trend in the number of publications and research projects among dermatology residency applicants. J Am Acad Dermatol. 2020;83:214-216.
- Atluri S, Seivright JR, Shi VY, et al. Volunteer and work experiences among dermatology residency applicants. J Am Acad Dermatol. 2021;84:E97-E98.
- Abokwidir M, Davis SA, Fleischer AB, et al. Use of the emergency department for dermatologic care in the United States by ethnic group. J Dermatolog Treat. 2015;26:392-394.
- Uscher-Pines L, Pines J, Kellermann A, et al. Emergency department visits for nonurgent conditions: systematic literature review. Am J Manag Care. 2013;19:47-59.
- Jack AR, Spence AA, Nichols BJ, et al. Cutaneous conditions leading to dermatology consultations in the emergency department. West J Emerg Med. 2011;12:551-555.
- Ayoubi N, Mirza A-S, Swanson J, et al. Dermatologic care of uninsured patients managed at free clinics. J Am Acad Dermatol. 2019;81:433-437.
- Wright AA, Katz IT. Beyond burnout—redesigning care to restore meaning and sanity for physicians. N Engl J Med. 2018;378:309-311.
- Bull C, Aucoin JB. Voluntary association participation and life satisfaction: a replication note. J Gerontol. 1975;30:73-76.
- Iserson KV. Burnout syndrome: global medicine volunteering as a possible treatment strategy. J Emerg Med. 2018;54:516-521.
- Romero S. Navajo Nation becomes largest tribe in U.S. after pandemic enrollment surge. New York Times. May 21, 2021. Accessed August 19, 2021. https://www.nytimes.com/2021/05/21/us/navajo-cherokee-population.html
- Moore GR, Benally J, Tuttle S. The Navajo Nation: quick facts. University of Arizona website. Accessed August 19, 2021. https://extension.arizona.edu/sites/extension.arizona.edu/files/pubs/az1471.pdf
The adage “so much to do, so little time” aptly describes the daily challenges facing dermatologists and dermatology residents. The time and attention required by direct patient care, writing notes, navigating electronic health records, and engaging in education and research as well as family commitments can drain even the most tireless clinician. In addition, dermatologists are expected to play a critical role in clinic and practice management to successfully curate an online presence and adapt their skills to successfully manage a teledermatology practice. Coupled with the time spent socializing with friends or colleagues and time for personal hobbies or exercise, it’s easy to see how sleep deprivation is common in many of our colleagues.
What’s being left out of these jam-packed schedules? Increasingly, it is the time and expertise dedicated to volunteering in our local communities. Two recent research letters highlighted how a dramatic increase in the number of research projects and publications is not mirrored by a similar increase in volunteer experiences as dermatology residency selection becomes more competitive.1,2
Although the rate of volunteerism among practicing dermatologists has yet to be studied, a brief review suggests a component of unmet dermatology need within our communities. It’s estimated that approximately 5% to 10% of all emergency department visits are for dermatologic concerns.3-5 In many cases, the reason for the visit is nonurgent and instead reflects a lack of other options for care. However, the need for dermatologists extends beyond the emergency department setting. A review of the prevalence of patients presenting for care to a group of regional free clinics found that 8% (N=5553) of all visitors sought care for dermatologic concerns.6 The benefit is not just for those seated on the examination table; research has shown that while many of the underlying factors resulting in physician burnout stem from systemic issues, participating in volunteer opportunities helps combat burnout in ourselves and our colleagues.7-9 Herein, opportunities that exist for dermatologists to reconnect with their communities, advocate for causes distinctive to the specialty, and care for neighbors most in need are highlighted.
Camp Wonder
Every year, children from across the United States living with chronic and debilitating skin conditions get the opportunity to join fellow campers and spend a week just being kids without the constant focus on being a patient. Camp Wonder’s founder and director, Francesca Tenconi, describes the camp as a place where kids “can form a community and can feel free to be themselves, without judgment, without stares. They get the chance to forget about their skin disease and be themselves” (oral communication, June 18, 2021). Tenconi and the camp’s cofounders and medical directors, Drs. Jenny Kim and Stefani Takahashi, envisioned the camp as a place for all campers regardless of their skin condition to feel safe and welcome. This overall mission guides camp leadership and staff every year over the course of the camp week where campers participate in a mix of traditional and nontraditional summer activities that are safe and accessible for all, from spending time in the pool to arts and crafts and a ropes course.
Camp Wonder is in its 21st year of hosting children and adolescents from across North America at its camp in Livermore, California. This year, Tenconi expects about 100 campers during the last week in July. Camp Wonder relies on medical staff volunteers to make the camp setting safe, inclusive, and fun. “Our dermatology residents and dermatology volunteers are a huge part of why we’re able to have camp,” said Tenconi. “A lot of our kids require very specific medical care throughout the week. We are able to provide this camp experience for them because we have this medical support system available, this specialized dermatology knowledge.” She also noted the benefit to the volunteers themselves, saying,“The feedback we get a lot from residents and dermatologists is that camp gave them a chance to understand the true-life impact of some of the skin diseases these kids and families are living with. Kids will open up to them and tell them how their disease has impacted them personally” (oral communication, June 18, 2021).
Volunteer medical providers help manage the medical needs of the campers beginning at check-in and work shifts in the infirmary as well as help with dispensing and administering medications, changing dressings, and applying ointments or other topical medications. When not assisting with medical care, medical staff can get to know the campers; help out with arts and crafts, games, sports, and other camp activities; and put on skits and plays for campers at nightly camp hangouts (Figure 1).

How to Get Involved
Visit the website (https://www.csdf.org/camp-wonder) for information on becoming a medical volunteer for 2022. Donations to help keep the camp running also are greatly appreciated, as attendance, including travel costs, is free for families through the Children’s Skin Disease Foundation. Finally, dermatologists can help by keeping their young patients with skin disease in mind as future campers. The camp welcomes kids from across the United States and Canada and invites questions from dermatologists and families on how to become a camper and what the experience is like.
Native American Health Services Rotation
Located in the southwestern United States, the Navajo Nation is North America’s largest Native American tribe by enrollment and resides on the largest reservation in the United States.10 Comprised of 27,000 square miles within portions of Arizona, New Mexico, and Utah, the reservation’s total area is greater than that of Massachusetts, Vermont, and New Hampshire combined.11 The reservation is home to an estimated 180,000 Navajo people, a population roughly the size of Salt Lake City, Utah. Yet, many homes on the reservation are without electricity, running water, telephones, or broadband access, and many roads on the reservation remain unpaved. Prior to the COVID-19 pandemic, 4 dermatology residents were selected each year to travel to this unique and remote location to work with the staff of the Chinle Comprehensive Health Care Facility (Chinle, Arizona), an Indian Health Service facility, as part of the American Academy of Dermatology (AAD)–sponsored Native American Health Services Resident Rotation (NAHSRR).
Dr. Lucinda Kohn, Assistant Professor of Dermatology at the University of Colorado and the director of the NAHSRR program discovered the value of this rotation firsthand as a dermatology resident. In 2017, she traveled to the area to spend 2 weeks serving within the community. “I went because of a personal connection. My husband is Native American, although not Navajo. I wanted to experience what it was like to provide dermatologic care for Native Americans. I found the Navajo people to be so friendly and so grateful for our care. The clinicians we worked with at Chinle were excited to have us share our expertise and to pass on their knowledge to us,” said Dr. Kohn (personal communication, June 24, 2021).
Rotating residents provide dermatologic care for the Navajo people and share their unique medical skill set to local primary care clinicians serving as preceptors. They also may have an opportunity to learn from Native healers about traditional Navajo beliefs and ceremonies used as part of a holistic approach to healing.
The program, similar to volunteer programs across the country, was put on hold during the height of the COVID-19 pandemic. “The Navajo nation witnessed a really tragic surge of COVID cases that required that limited medical resources be diverted to help cope with the pandemic,” says Dr. Kohn. “It really wasn’t safe for residents to travel to the reservation either, so the rotation had to be put on hold.” However, in April 2021, the health care staff of the Chinle Comprehensive Care Facility reached out to revive the program, which is now pending the green light from the AAD. It is unclear if or when AAD leadership will allow this rotation to restart. Dr. Kohn hopes to be able to start accepting new applications soon. “This rotation provides a wealth of benefits to all those involved, from the residents who get the chance to work with a unique population in need to the clinicians who gain a diverse understanding of dermatology treatment techniques. And of course, for the patients, who are so appreciative of the care they receive from our volunteers” (personal communication, June 25, 2021).
How to Get Involved
Dr. Kohn is happy to field questions regarding the rotation and requests for more information via email ([email protected]). Residents interested in this program also may reach out to the AAD’s Education and Volunteers Abroad Committee to express interest in the NAHSRR program’s reinstatement.
Destination Healthy Skin
Since 2017, the Skin Cancer Foundation’s Destination Healthy Skin (DHS) RV has been the setting for more than 3800 free skin cancer screenings provided by volunteers within underserved populations across the United States (Figure 2). After a year hiatus due to the pandemic, DHS hit the road again, starting in New York City on August 1 to 3, 2021. From there, the DHS RV will traverse the country in one large loop, starting with visits to large and small cities in the Midwest and the West Coast. Following a visit to San Diego, California, in early October, the RV will turn east, with stops in Arizona, Texas, and several southern states before ending in Philadelphia, Pennsylvania. Dr. Elizabeth Hale, Senior Vice President of the Skin Cancer Foundation, feels that increasing awareness of the importance of regular skin cancer screening for those at risk is more important than ever. “We know that many people in the past year put routine cancer screening on the back burner, but we’re beginning to appreciate that this has led to significant delays in skin cancer diagnosis and potentially more significant disease when cases are diagnosed.” Dr. Hale noted that as the country continues to return to a degree of normalcy, the backlog of patients now seeking their routine screening has led to longer wait times. She expects DHS may offer some relief. “There are no appointments necessary. If the RV is close to their hometown, patients have an advantage in being able to be seen first come, first served, without having to wait for an appointment or make sure their insurance is accepted. It’s a free screening that can increase access to dermatologists” (personal communication, June 21, 2021).

The program’s organizers acknowledge that DHS is not a long-term solution for improving dermatology access in the United States and recognize that more needs to be done to raise awareness, both of the value that screenings can provide and the importance of sun-protective behavior. “This is an important first step,” says Dr. Hale. “It’s important that we disseminate that no one is immune to skin cancer. It’s about education, and this is a tool to educate patients that everyone should have a skin check once a year, regardless of where you live or what your skin type is” (personal communication, June 21, 2021).
Volunteer dermatologists are needed to assist with screenings when the DHS RV arrives in their community. Providers complete a screening form identifying any concerning lesions and can document specific lesions using the patient’s cell phone. Following the screenings, participating dermatologists are welcome to invite participants to make appointments at their practices or suggest local clinics for follow-up care.
How to Get Involved
The schedule for this year’s screening events can be found online (https://www.skincancer.org/early-detection/destination-healthy-skin/). Consider volunteering (https://www.skincancer.org/early-detection/destination-healthy-skin/physician-volunteers/) or helping to raise awareness by reaching out to local dermatology societies or free clinics in your area. Residents and physician’s assistants are welcome to volunteer as well, as long as they are under the on-site supervision of a board-certified dermatologist.
Final Thoughts
As medical professionals, we all recognize there are valuable contributions we can make to groups and organizations that need our help. The stresses and pressure of work and everyday life can make finding the time to offer that help seem impossible. Although it may seem counterintuitive, volunteering our time to help others can help us better navigate the professional burnout that many medical professionals experience today.
The adage “so much to do, so little time” aptly describes the daily challenges facing dermatologists and dermatology residents. The time and attention required by direct patient care, writing notes, navigating electronic health records, and engaging in education and research as well as family commitments can drain even the most tireless clinician. In addition, dermatologists are expected to play a critical role in clinic and practice management to successfully curate an online presence and adapt their skills to successfully manage a teledermatology practice. Coupled with the time spent socializing with friends or colleagues and time for personal hobbies or exercise, it’s easy to see how sleep deprivation is common in many of our colleagues.
What’s being left out of these jam-packed schedules? Increasingly, it is the time and expertise dedicated to volunteering in our local communities. Two recent research letters highlighted how a dramatic increase in the number of research projects and publications is not mirrored by a similar increase in volunteer experiences as dermatology residency selection becomes more competitive.1,2
Although the rate of volunteerism among practicing dermatologists has yet to be studied, a brief review suggests a component of unmet dermatology need within our communities. It’s estimated that approximately 5% to 10% of all emergency department visits are for dermatologic concerns.3-5 In many cases, the reason for the visit is nonurgent and instead reflects a lack of other options for care. However, the need for dermatologists extends beyond the emergency department setting. A review of the prevalence of patients presenting for care to a group of regional free clinics found that 8% (N=5553) of all visitors sought care for dermatologic concerns.6 The benefit is not just for those seated on the examination table; research has shown that while many of the underlying factors resulting in physician burnout stem from systemic issues, participating in volunteer opportunities helps combat burnout in ourselves and our colleagues.7-9 Herein, opportunities that exist for dermatologists to reconnect with their communities, advocate for causes distinctive to the specialty, and care for neighbors most in need are highlighted.
Camp Wonder
Every year, children from across the United States living with chronic and debilitating skin conditions get the opportunity to join fellow campers and spend a week just being kids without the constant focus on being a patient. Camp Wonder’s founder and director, Francesca Tenconi, describes the camp as a place where kids “can form a community and can feel free to be themselves, without judgment, without stares. They get the chance to forget about their skin disease and be themselves” (oral communication, June 18, 2021). Tenconi and the camp’s cofounders and medical directors, Drs. Jenny Kim and Stefani Takahashi, envisioned the camp as a place for all campers regardless of their skin condition to feel safe and welcome. This overall mission guides camp leadership and staff every year over the course of the camp week where campers participate in a mix of traditional and nontraditional summer activities that are safe and accessible for all, from spending time in the pool to arts and crafts and a ropes course.
Camp Wonder is in its 21st year of hosting children and adolescents from across North America at its camp in Livermore, California. This year, Tenconi expects about 100 campers during the last week in July. Camp Wonder relies on medical staff volunteers to make the camp setting safe, inclusive, and fun. “Our dermatology residents and dermatology volunteers are a huge part of why we’re able to have camp,” said Tenconi. “A lot of our kids require very specific medical care throughout the week. We are able to provide this camp experience for them because we have this medical support system available, this specialized dermatology knowledge.” She also noted the benefit to the volunteers themselves, saying,“The feedback we get a lot from residents and dermatologists is that camp gave them a chance to understand the true-life impact of some of the skin diseases these kids and families are living with. Kids will open up to them and tell them how their disease has impacted them personally” (oral communication, June 18, 2021).
Volunteer medical providers help manage the medical needs of the campers beginning at check-in and work shifts in the infirmary as well as help with dispensing and administering medications, changing dressings, and applying ointments or other topical medications. When not assisting with medical care, medical staff can get to know the campers; help out with arts and crafts, games, sports, and other camp activities; and put on skits and plays for campers at nightly camp hangouts (Figure 1).

How to Get Involved
Visit the website (https://www.csdf.org/camp-wonder) for information on becoming a medical volunteer for 2022. Donations to help keep the camp running also are greatly appreciated, as attendance, including travel costs, is free for families through the Children’s Skin Disease Foundation. Finally, dermatologists can help by keeping their young patients with skin disease in mind as future campers. The camp welcomes kids from across the United States and Canada and invites questions from dermatologists and families on how to become a camper and what the experience is like.
Native American Health Services Rotation
Located in the southwestern United States, the Navajo Nation is North America’s largest Native American tribe by enrollment and resides on the largest reservation in the United States.10 Comprised of 27,000 square miles within portions of Arizona, New Mexico, and Utah, the reservation’s total area is greater than that of Massachusetts, Vermont, and New Hampshire combined.11 The reservation is home to an estimated 180,000 Navajo people, a population roughly the size of Salt Lake City, Utah. Yet, many homes on the reservation are without electricity, running water, telephones, or broadband access, and many roads on the reservation remain unpaved. Prior to the COVID-19 pandemic, 4 dermatology residents were selected each year to travel to this unique and remote location to work with the staff of the Chinle Comprehensive Health Care Facility (Chinle, Arizona), an Indian Health Service facility, as part of the American Academy of Dermatology (AAD)–sponsored Native American Health Services Resident Rotation (NAHSRR).
Dr. Lucinda Kohn, Assistant Professor of Dermatology at the University of Colorado and the director of the NAHSRR program discovered the value of this rotation firsthand as a dermatology resident. In 2017, she traveled to the area to spend 2 weeks serving within the community. “I went because of a personal connection. My husband is Native American, although not Navajo. I wanted to experience what it was like to provide dermatologic care for Native Americans. I found the Navajo people to be so friendly and so grateful for our care. The clinicians we worked with at Chinle were excited to have us share our expertise and to pass on their knowledge to us,” said Dr. Kohn (personal communication, June 24, 2021).
Rotating residents provide dermatologic care for the Navajo people and share their unique medical skill set to local primary care clinicians serving as preceptors. They also may have an opportunity to learn from Native healers about traditional Navajo beliefs and ceremonies used as part of a holistic approach to healing.
The program, similar to volunteer programs across the country, was put on hold during the height of the COVID-19 pandemic. “The Navajo nation witnessed a really tragic surge of COVID cases that required that limited medical resources be diverted to help cope with the pandemic,” says Dr. Kohn. “It really wasn’t safe for residents to travel to the reservation either, so the rotation had to be put on hold.” However, in April 2021, the health care staff of the Chinle Comprehensive Care Facility reached out to revive the program, which is now pending the green light from the AAD. It is unclear if or when AAD leadership will allow this rotation to restart. Dr. Kohn hopes to be able to start accepting new applications soon. “This rotation provides a wealth of benefits to all those involved, from the residents who get the chance to work with a unique population in need to the clinicians who gain a diverse understanding of dermatology treatment techniques. And of course, for the patients, who are so appreciative of the care they receive from our volunteers” (personal communication, June 25, 2021).
How to Get Involved
Dr. Kohn is happy to field questions regarding the rotation and requests for more information via email ([email protected]). Residents interested in this program also may reach out to the AAD’s Education and Volunteers Abroad Committee to express interest in the NAHSRR program’s reinstatement.
Destination Healthy Skin
Since 2017, the Skin Cancer Foundation’s Destination Healthy Skin (DHS) RV has been the setting for more than 3800 free skin cancer screenings provided by volunteers within underserved populations across the United States (Figure 2). After a year hiatus due to the pandemic, DHS hit the road again, starting in New York City on August 1 to 3, 2021. From there, the DHS RV will traverse the country in one large loop, starting with visits to large and small cities in the Midwest and the West Coast. Following a visit to San Diego, California, in early October, the RV will turn east, with stops in Arizona, Texas, and several southern states before ending in Philadelphia, Pennsylvania. Dr. Elizabeth Hale, Senior Vice President of the Skin Cancer Foundation, feels that increasing awareness of the importance of regular skin cancer screening for those at risk is more important than ever. “We know that many people in the past year put routine cancer screening on the back burner, but we’re beginning to appreciate that this has led to significant delays in skin cancer diagnosis and potentially more significant disease when cases are diagnosed.” Dr. Hale noted that as the country continues to return to a degree of normalcy, the backlog of patients now seeking their routine screening has led to longer wait times. She expects DHS may offer some relief. “There are no appointments necessary. If the RV is close to their hometown, patients have an advantage in being able to be seen first come, first served, without having to wait for an appointment or make sure their insurance is accepted. It’s a free screening that can increase access to dermatologists” (personal communication, June 21, 2021).

The program’s organizers acknowledge that DHS is not a long-term solution for improving dermatology access in the United States and recognize that more needs to be done to raise awareness, both of the value that screenings can provide and the importance of sun-protective behavior. “This is an important first step,” says Dr. Hale. “It’s important that we disseminate that no one is immune to skin cancer. It’s about education, and this is a tool to educate patients that everyone should have a skin check once a year, regardless of where you live or what your skin type is” (personal communication, June 21, 2021).
Volunteer dermatologists are needed to assist with screenings when the DHS RV arrives in their community. Providers complete a screening form identifying any concerning lesions and can document specific lesions using the patient’s cell phone. Following the screenings, participating dermatologists are welcome to invite participants to make appointments at their practices or suggest local clinics for follow-up care.
How to Get Involved
The schedule for this year’s screening events can be found online (https://www.skincancer.org/early-detection/destination-healthy-skin/). Consider volunteering (https://www.skincancer.org/early-detection/destination-healthy-skin/physician-volunteers/) or helping to raise awareness by reaching out to local dermatology societies or free clinics in your area. Residents and physician’s assistants are welcome to volunteer as well, as long as they are under the on-site supervision of a board-certified dermatologist.
Final Thoughts
As medical professionals, we all recognize there are valuable contributions we can make to groups and organizations that need our help. The stresses and pressure of work and everyday life can make finding the time to offer that help seem impossible. Although it may seem counterintuitive, volunteering our time to help others can help us better navigate the professional burnout that many medical professionals experience today.
- Ezekor M, Pona A, Cline A, et al. An increasing trend in the number of publications and research projects among dermatology residency applicants. J Am Acad Dermatol. 2020;83:214-216.
- Atluri S, Seivright JR, Shi VY, et al. Volunteer and work experiences among dermatology residency applicants. J Am Acad Dermatol. 2021;84:E97-E98.
- Abokwidir M, Davis SA, Fleischer AB, et al. Use of the emergency department for dermatologic care in the United States by ethnic group. J Dermatolog Treat. 2015;26:392-394.
- Uscher-Pines L, Pines J, Kellermann A, et al. Emergency department visits for nonurgent conditions: systematic literature review. Am J Manag Care. 2013;19:47-59.
- Jack AR, Spence AA, Nichols BJ, et al. Cutaneous conditions leading to dermatology consultations in the emergency department. West J Emerg Med. 2011;12:551-555.
- Ayoubi N, Mirza A-S, Swanson J, et al. Dermatologic care of uninsured patients managed at free clinics. J Am Acad Dermatol. 2019;81:433-437.
- Wright AA, Katz IT. Beyond burnout—redesigning care to restore meaning and sanity for physicians. N Engl J Med. 2018;378:309-311.
- Bull C, Aucoin JB. Voluntary association participation and life satisfaction: a replication note. J Gerontol. 1975;30:73-76.
- Iserson KV. Burnout syndrome: global medicine volunteering as a possible treatment strategy. J Emerg Med. 2018;54:516-521.
- Romero S. Navajo Nation becomes largest tribe in U.S. after pandemic enrollment surge. New York Times. May 21, 2021. Accessed August 19, 2021. https://www.nytimes.com/2021/05/21/us/navajo-cherokee-population.html
- Moore GR, Benally J, Tuttle S. The Navajo Nation: quick facts. University of Arizona website. Accessed August 19, 2021. https://extension.arizona.edu/sites/extension.arizona.edu/files/pubs/az1471.pdf
- Ezekor M, Pona A, Cline A, et al. An increasing trend in the number of publications and research projects among dermatology residency applicants. J Am Acad Dermatol. 2020;83:214-216.
- Atluri S, Seivright JR, Shi VY, et al. Volunteer and work experiences among dermatology residency applicants. J Am Acad Dermatol. 2021;84:E97-E98.
- Abokwidir M, Davis SA, Fleischer AB, et al. Use of the emergency department for dermatologic care in the United States by ethnic group. J Dermatolog Treat. 2015;26:392-394.
- Uscher-Pines L, Pines J, Kellermann A, et al. Emergency department visits for nonurgent conditions: systematic literature review. Am J Manag Care. 2013;19:47-59.
- Jack AR, Spence AA, Nichols BJ, et al. Cutaneous conditions leading to dermatology consultations in the emergency department. West J Emerg Med. 2011;12:551-555.
- Ayoubi N, Mirza A-S, Swanson J, et al. Dermatologic care of uninsured patients managed at free clinics. J Am Acad Dermatol. 2019;81:433-437.
- Wright AA, Katz IT. Beyond burnout—redesigning care to restore meaning and sanity for physicians. N Engl J Med. 2018;378:309-311.
- Bull C, Aucoin JB. Voluntary association participation and life satisfaction: a replication note. J Gerontol. 1975;30:73-76.
- Iserson KV. Burnout syndrome: global medicine volunteering as a possible treatment strategy. J Emerg Med. 2018;54:516-521.
- Romero S. Navajo Nation becomes largest tribe in U.S. after pandemic enrollment surge. New York Times. May 21, 2021. Accessed August 19, 2021. https://www.nytimes.com/2021/05/21/us/navajo-cherokee-population.html
- Moore GR, Benally J, Tuttle S. The Navajo Nation: quick facts. University of Arizona website. Accessed August 19, 2021. https://extension.arizona.edu/sites/extension.arizona.edu/files/pubs/az1471.pdf
Resident Pearl
- Volunteerism rates among dermatology residents seem to be decreasing. We should work to combat this trend by finding ways to give back to our communities and spur our colleagues to do the same.
Clinical Edge Journal Scan Commentary: Psoriasis September 2021
Several recent studies have evaluated that association between psoriasis and known comorbidities including cardiovascular disease, non-alcoholic fatty liver disease (NAFLD), and malignancy. Several recent studies have added to our understanding of the relationship between these conditions.
Using the largest database of hospitalized patients in the United States, Edgen et al found that hospitalization rates for patients with psoriasis are increasing. While the proportion of patients with psoriasis hospitalized with psoriasis as a primary diagnosis decreased about four-fold over a 20-year period (1999-2018), incidence of hospitalizations with any diagnosis of psoriasis has increased. Hospitalized psoriasis patients are increasingly more likely to have other comorbid conditions as during the study period the proportion of hospitalized psoriasis patients with a Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9%. Psoriasis severity, medication use, and reasons for hospitalization were not reported. The authors suggest that screening and management of comorbidities in the outpatient setting may help reduce preventable psoriasis hospitalizations.
Both NAFLD and cardiovascular disease are well-known psoriasis comorbidities, Gonzalez-Cantaro et al studied two cohorts of patients to better define the relationship between these two conditions. In a European cohort of 76 psoriasis patients and 76 control patients, psoriasis patients with NAFLD had a higher prevalence of subclinical atherosclerosis than both psoriasis patients without NAFLD (61% vs 23%) and age, sex, and BMI-matched controls with NAFLD (61% vs 32%). Psoriasis patients were also more likely that control patients to have insulin resistance, higher weight circumference, and dysplipidemia. Among 162 psoriasis patients who underwent PET and coronary CT angiography, higher hepatic FDG uptake (indicating NAFLD) was associated higher atherosclerotic disease burden. Importantly, both the NAFLD and CAD were subclinical in these patients. While the cross-sectional study design precludes any conclusions about causality, physicians should be aware that these two comorbidities are related. Lower waist circumference and greater physical activity were both associated with lower rates of NAFLD among patients with psoriasis, providing some guidance for counseling patients.
Several recent studies have found that cancer rates among patients with psoriasis are higher than what is observed in the general population. The association of psoriasis with lymphohematologic malignancies (LHM) has been controversial. A systematic review and meta-analysis of 25 observational studies including over 2.5 million subjects (Bellinato et al.) found a 1.55-fold increased risk of LHM in patients with moderate to severe psoriasis. Strikingly, the risk of cutaneous T cell lymphoma (CTCL) was increased 6.22-fold, with more severe psoriasis being associated with the highest risk of CTCL. A causal relationship cannot be established from this type of studies, but the authors hypothesize that drugs used to treat psoriasis or the chronic T cell activation caused by active disease may contribute to the risk of LMH. Additionally, psoriasis and CTCL can share clinical features and some cases may be due to misdiagnosis. Interestingly, two psoriasis comorbities, diabetes and obesity, are also associated with an increased risk of LHM.
Early identification and management of comorbidities can help in reducing morbidity and mortality. With so many psoriasis treatments available, understanding how different therapies may impact comorbid conditions is important in helping dermatologists to choose the best therapy for each individual patient.
Several recent studies have evaluated that association between psoriasis and known comorbidities including cardiovascular disease, non-alcoholic fatty liver disease (NAFLD), and malignancy. Several recent studies have added to our understanding of the relationship between these conditions.
Using the largest database of hospitalized patients in the United States, Edgen et al found that hospitalization rates for patients with psoriasis are increasing. While the proportion of patients with psoriasis hospitalized with psoriasis as a primary diagnosis decreased about four-fold over a 20-year period (1999-2018), incidence of hospitalizations with any diagnosis of psoriasis has increased. Hospitalized psoriasis patients are increasingly more likely to have other comorbid conditions as during the study period the proportion of hospitalized psoriasis patients with a Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9%. Psoriasis severity, medication use, and reasons for hospitalization were not reported. The authors suggest that screening and management of comorbidities in the outpatient setting may help reduce preventable psoriasis hospitalizations.
Both NAFLD and cardiovascular disease are well-known psoriasis comorbidities, Gonzalez-Cantaro et al studied two cohorts of patients to better define the relationship between these two conditions. In a European cohort of 76 psoriasis patients and 76 control patients, psoriasis patients with NAFLD had a higher prevalence of subclinical atherosclerosis than both psoriasis patients without NAFLD (61% vs 23%) and age, sex, and BMI-matched controls with NAFLD (61% vs 32%). Psoriasis patients were also more likely that control patients to have insulin resistance, higher weight circumference, and dysplipidemia. Among 162 psoriasis patients who underwent PET and coronary CT angiography, higher hepatic FDG uptake (indicating NAFLD) was associated higher atherosclerotic disease burden. Importantly, both the NAFLD and CAD were subclinical in these patients. While the cross-sectional study design precludes any conclusions about causality, physicians should be aware that these two comorbidities are related. Lower waist circumference and greater physical activity were both associated with lower rates of NAFLD among patients with psoriasis, providing some guidance for counseling patients.
Several recent studies have found that cancer rates among patients with psoriasis are higher than what is observed in the general population. The association of psoriasis with lymphohematologic malignancies (LHM) has been controversial. A systematic review and meta-analysis of 25 observational studies including over 2.5 million subjects (Bellinato et al.) found a 1.55-fold increased risk of LHM in patients with moderate to severe psoriasis. Strikingly, the risk of cutaneous T cell lymphoma (CTCL) was increased 6.22-fold, with more severe psoriasis being associated with the highest risk of CTCL. A causal relationship cannot be established from this type of studies, but the authors hypothesize that drugs used to treat psoriasis or the chronic T cell activation caused by active disease may contribute to the risk of LMH. Additionally, psoriasis and CTCL can share clinical features and some cases may be due to misdiagnosis. Interestingly, two psoriasis comorbities, diabetes and obesity, are also associated with an increased risk of LHM.
Early identification and management of comorbidities can help in reducing morbidity and mortality. With so many psoriasis treatments available, understanding how different therapies may impact comorbid conditions is important in helping dermatologists to choose the best therapy for each individual patient.
Several recent studies have evaluated that association between psoriasis and known comorbidities including cardiovascular disease, non-alcoholic fatty liver disease (NAFLD), and malignancy. Several recent studies have added to our understanding of the relationship between these conditions.
Using the largest database of hospitalized patients in the United States, Edgen et al found that hospitalization rates for patients with psoriasis are increasing. While the proportion of patients with psoriasis hospitalized with psoriasis as a primary diagnosis decreased about four-fold over a 20-year period (1999-2018), incidence of hospitalizations with any diagnosis of psoriasis has increased. Hospitalized psoriasis patients are increasingly more likely to have other comorbid conditions as during the study period the proportion of hospitalized psoriasis patients with a Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9%. Psoriasis severity, medication use, and reasons for hospitalization were not reported. The authors suggest that screening and management of comorbidities in the outpatient setting may help reduce preventable psoriasis hospitalizations.
Both NAFLD and cardiovascular disease are well-known psoriasis comorbidities, Gonzalez-Cantaro et al studied two cohorts of patients to better define the relationship between these two conditions. In a European cohort of 76 psoriasis patients and 76 control patients, psoriasis patients with NAFLD had a higher prevalence of subclinical atherosclerosis than both psoriasis patients without NAFLD (61% vs 23%) and age, sex, and BMI-matched controls with NAFLD (61% vs 32%). Psoriasis patients were also more likely that control patients to have insulin resistance, higher weight circumference, and dysplipidemia. Among 162 psoriasis patients who underwent PET and coronary CT angiography, higher hepatic FDG uptake (indicating NAFLD) was associated higher atherosclerotic disease burden. Importantly, both the NAFLD and CAD were subclinical in these patients. While the cross-sectional study design precludes any conclusions about causality, physicians should be aware that these two comorbidities are related. Lower waist circumference and greater physical activity were both associated with lower rates of NAFLD among patients with psoriasis, providing some guidance for counseling patients.
Several recent studies have found that cancer rates among patients with psoriasis are higher than what is observed in the general population. The association of psoriasis with lymphohematologic malignancies (LHM) has been controversial. A systematic review and meta-analysis of 25 observational studies including over 2.5 million subjects (Bellinato et al.) found a 1.55-fold increased risk of LHM in patients with moderate to severe psoriasis. Strikingly, the risk of cutaneous T cell lymphoma (CTCL) was increased 6.22-fold, with more severe psoriasis being associated with the highest risk of CTCL. A causal relationship cannot be established from this type of studies, but the authors hypothesize that drugs used to treat psoriasis or the chronic T cell activation caused by active disease may contribute to the risk of LMH. Additionally, psoriasis and CTCL can share clinical features and some cases may be due to misdiagnosis. Interestingly, two psoriasis comorbities, diabetes and obesity, are also associated with an increased risk of LHM.
Early identification and management of comorbidities can help in reducing morbidity and mortality. With so many psoriasis treatments available, understanding how different therapies may impact comorbid conditions is important in helping dermatologists to choose the best therapy for each individual patient.
Report urges complete residency overhaul
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
from the Graduate Medical Education Review Committee (UGRC) of the Coalition for Physician Accountability.
The 275-page report presents preliminary findings that were released in April 2021 and a long list of stakeholder comments. According to the report, the coalition will meet soon to discuss the final recommendations and consider next steps toward implementation.
The UGRC includes representatives of national medical organizations, medical schools, and residency programs. Among the organizations that participated in the report’s creation are the American Medical Association, the National Board of Medical Examiners, the American Osteopathic Association, the National Board of Osteopathic Medical Examiners, the Educational Commission for Foreign Medical Graduates, and the Association of American Medical Colleges.
The report identifies a list of challenges that affect the transition of medical students into residency programs and beyond. They include:
- Too much focus on finding and filling residency positions instead of “assuring learner competence and readiness for residency training”
- Inattention to assuring congruence between applicant goals and program missions
- Overreliance on licensure exam scores rather than “valid, trustworthy measures of students’ competence and clinical abilities”
- Increasing financial costs to students
- Individual and systemic biases in the UME-GME transition, as well as inequities related to international medical graduates
Seeking a common framework for competence
Overall, the report calls for increased standardization of how students are evaluated in medical school and how residency programs evaluate students. Less reliance should be placed on the numerical scores of the U.S. Medical Licensing Examination (USMLE), the report says, and more attention should be paid to the direct observation of student performance in clinical situations. In addition, the various organizations involved in the UME-GME transition process are asked to work better together.
To develop better methods of evaluating medical students and residents, UME and GME educators should jointly define and implement a common framework and set of competencies to apply to learners across the UME-GME transition, the report suggests.
While emphasizing the need for a broader student assessment framework, the report says, USMLE scores should also continue to be used in judging residency applicants. “Assessment information should be shared in residency applications and a postmatch learner handover. Licensing examinations should be used for their intended purpose to ensure requisite competence.”
Among the committee’s three dozen recommendations are the following:
- The Centers for Medicare & Medicaid Services should change the GME funding structure so that the initial residency period is calculated starting with the second year of postgraduate training. This change would allow residents to reconsider their career choices. Currently, if a resident decides to switch to another program or specialty after beginning training, the hospital may not receive full GME funding, so may be less likely to approve the change.
- Residency programs should improve recruitment practices to increase specialty-specific diversity of residents. Medical educators should also receive additional training regarding antiracism, avoiding bias, and ensuring equity.
- The self-reported demographic information of applicants to residency programs should be measured and shared with stakeholders, including the programs and medical schools, to promote equity. “A residency program that finds bias in its selection process could go back in real time to find qualified applicants who may have been missed, potentially improving outcomes,” the report notes.
- An interactive database of GME program and specialty track information should be created and made available to all applicants, medical schools, and residency programs at no cost to applicants. “Applicants and their advisors should be able to sort the information according to demographic and educational features that may significantly impact the likelihood of matching at a program.”
Less than half of applicants get in-depth reviews
The 2020 National Resident Matching Program Program Director Survey found that only 49% of applications received in-depth review. In light of this, the report suggests that the application system be updated to use modern information technology, including discrete fields for key data to expedite application reviews.
Many applications have been discarded because of various filters used to block consideration of certain applications. The report suggests that new filters be designed to ensure that each detects meaningful differences among applicants and promotes review based on mission alignment and likelihood of success in a program. Filters should be improved to decrease the likelihood of random exclusions of qualified applicants.
Specialty-specific, just-in-time training for all incoming first-year residents is also suggested to support the transition from the role of student to a physician ready to assume increased responsibility for patient care. In addition, the report urges adequate time be allowed between medical school graduation and residency to enable new residents to relocate and find homes.
The report also calls for a standardized process in the United States for initial licensing of doctors at entrance to residency in order to streamline the process of credentialing for both residency training and continuing practice.
Osteopathic students’ dilemma
To promote equitable treatment of applicants regardless of licensure examination requirements, comparable exams with different scales (COMLEX-USA and USMLE) should be reported within the electronic application system in a single field, the report said.
Osteopathic students, who make up 25% of U.S. medical students, must take the COMLEX-USA exam, but residency programs may filter them out if they don’t also take the USMLE exam. Thus, many osteopathic students take both exams, incurring extra time, cost, and stress.
The UGRC recommends creating a combined field in the electronic residency application service that normalizes the scores between the two exams. Residency programs could then filter applications based only on the single normalized score.
This approach makes sense from the viewpoint that it would reduce the pressure on osteopathic students to take the USMLE, Bryan Carmody, MD, an outspoken critic of various current training policies, said in an interview. But it could also have serious disadvantages.
For one thing, only osteopathic students can take the COMLEX-USA exam, he noted. If they don’t like their score, they can then take the USMLE test to get a higher score – an option that allopathic students don’t have. It’s not clear that they’d be prevented from doing this under the UGRC recommendation.
Second, he said, osteopathic students, on average, don’t do as well as allopathic students on the UMSLE exam. If they only take the COMLEX-USA test, they’re competing against other students who don’t do as well on tests as allopathic students do. If their scores were normalized with those of the USMLE test takers, they’d gain an unfair advantage against students who can only take the USMLE, including international medical graduates.
Although Dr. Carmody admitted that osteopathic students face a harder challenge than allopathic students in matching to residency programs, he said that the UGRC approach to the licensing exams might actually penalize them further. As a result of the scores of the two exams being averaged, residency program directors might discount the scores of all osteopathic students.
A version of this article first appeared on Medscape.com.
Verrucous Scalp Plaque and Widespread Eruption
The Diagnosis: Pemphigus Foliaceous
Laboratory workup including a complete blood cell count with differential, comprehensive metabolic panel, antinuclear antibodies, Sjögren syndrome A and B antibodies, hepatitis profile, rapid plasma reagin, HIV screen, aldolase, anti–Jo-1, T-Spot TB test (Quest Diagnostics), and tissue cultures was unremarkable. Two 4-mm punch biopsies were obtained from the left cheek and upper back, both of which demonstrated intragranular acantholysis suggestive of pemphigus foliaceous (Figure 1A). A subsequent punch biopsy from the right lower abdomen sent for direct immunofluorescence demonstrated netlike positivity of IgG and C3 in the upper epidermis (Figure 1B), and serum sent for indirect immunofluorescence demonstrated intercellular IgG antibodies to desmoglein (Dsg) 1 on monkey esophagus and positive Dsg-1 antibodies on enzyme-linked immunosorbent assay, confirming the diagnosis.
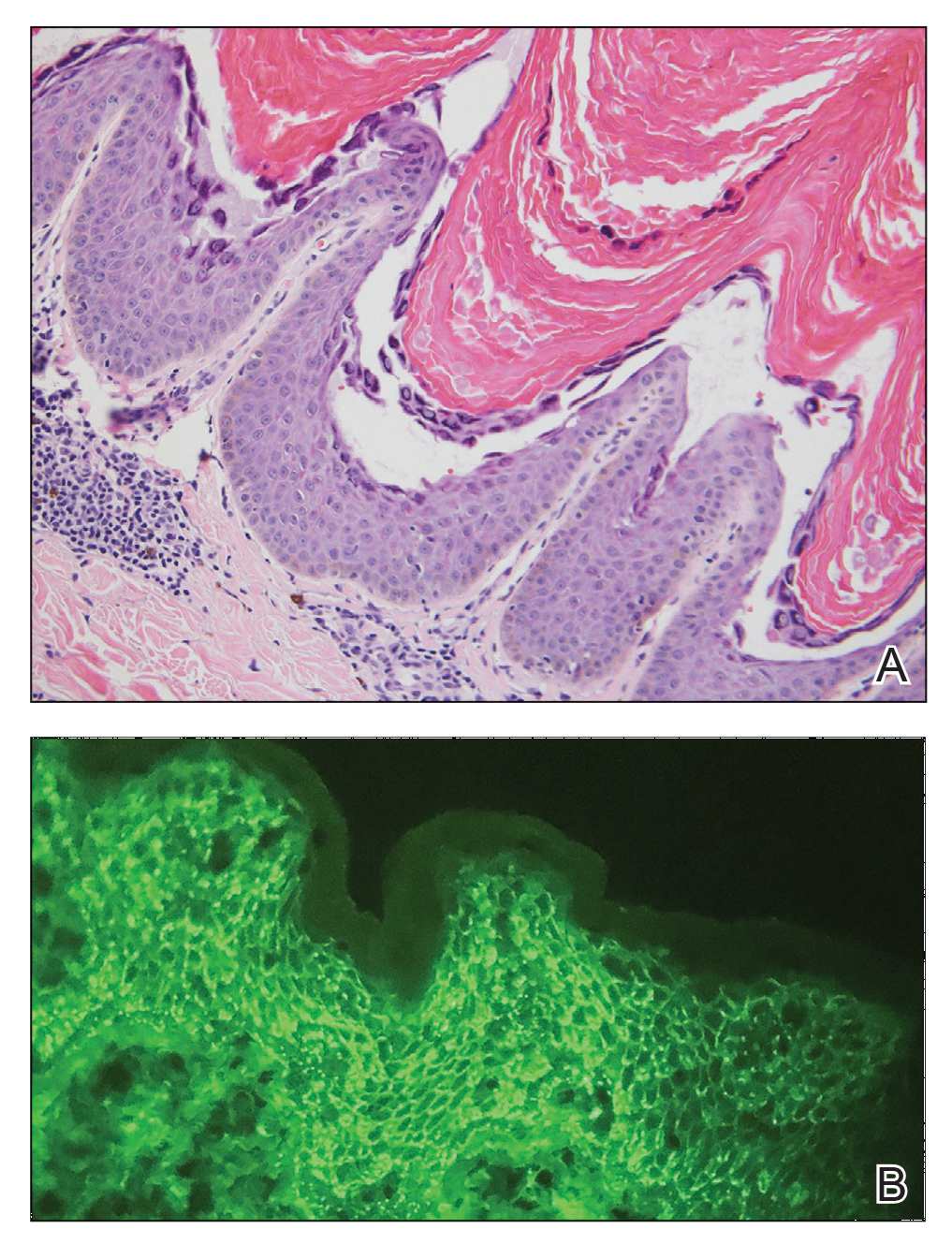
The patient was started on a 60-mg prednisone taper as well as dapsone 50 mg daily; the dapsone was titrated up to 100 mg daily. After tapering down to 10 mg daily of prednisone over 2 months and continuing dapsone with minimal improvement, he was given 2 infusions of rituximab 1000 mg 2 weeks apart. The scalp plaque was dramatically improved at 3-month follow-up (Figure 2), with partial improvement of the cheek plaques (Figure 3). Dapsone was increased to 150 mg daily, and he was encouraged to use triamcinolone acetonide ointment 0.1% twice daily, which led to further improvement.
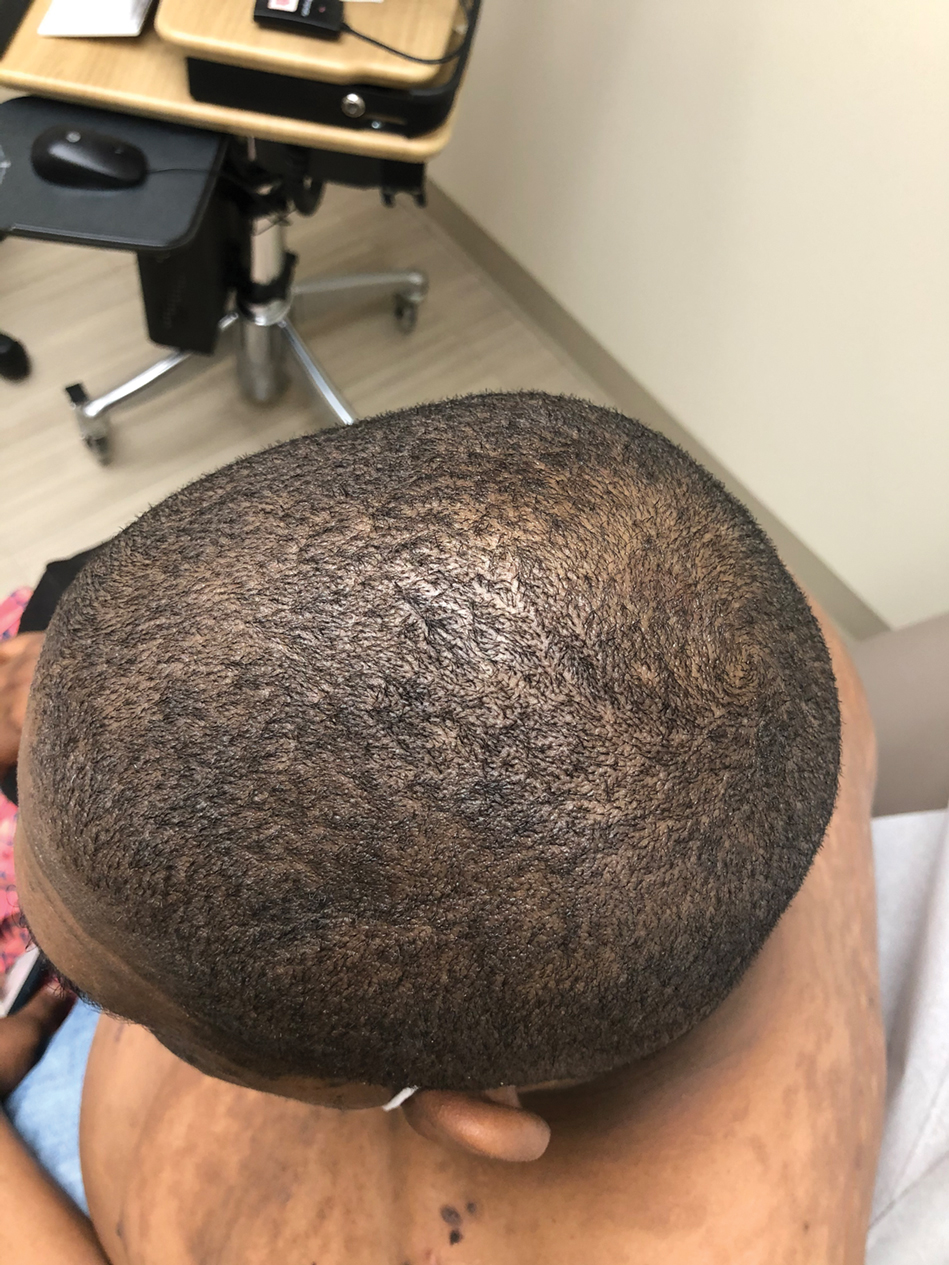
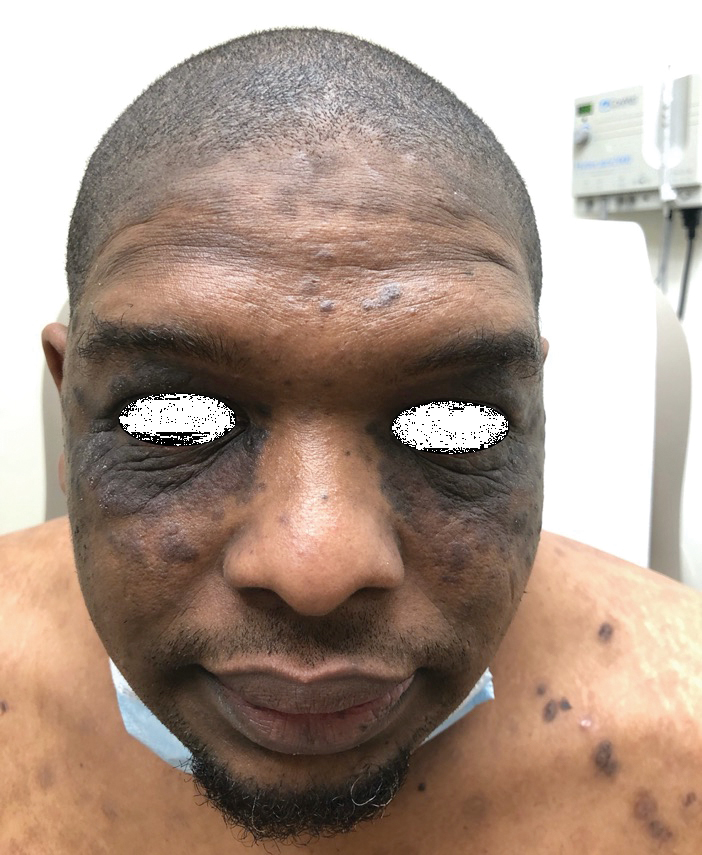
Pemphigus foliaceus is an autoimmune blistering disease that most commonly occurs in middle-aged adults. It generally is less common than pemphigus vulgaris, except in Finland, Tunisia, and Brazil, where there is an endemic condition with an identical clinical and histological presentation known as fogo selvagem.1
The pathogenesis of pemphigus foliaceous is characterized by IgG autoantibodies against Dsg-1, a transmembrane glycoprotein involved in the cellular adhesion of keratinocytes, which is preferentially expressed in the superficial epidermis.2-7 Dysfunction of Dsg-1 results in the separation of superficial epidermal cells, resulting in intraepidermal blisters.2,7 In contrast to pemphigus vulgaris, there typically is a lack of oral mucosal involvement due to compensation by Dsg-3 in the mucosa.4 Potential triggers for pemphigus foliaceous include exposure to UV radiation; radiotherapy; pregnancy; physiologic stress; and drugs, most commonly captopril, penicillamine, and thiols.8
Pemphigus foliaceous lesions clinically appear as eroded and crusted lesions on an erythematous base, commonly in a seborrheic distribution on the face, scalp, and trunk with sparing of the oral mucosa,2,6 but lesions can progress to a widespread and more severe exfoliative dermatitis.7 Lesions also can appear as psoriasiform plaques and often are initially misdiagnosed as psoriasis, particularly in patients with skin of color.9,10
Diagnosis of pemphigus foliaceous typically is made using a combination of histology as well as both direct and indirect immunofluorescence. Histologically, pemphigus foliaceus presents with subcorneal acantholysis, which is most prominent in the granular layer and occasionally the presence of neutrophils and eosinophils in the blister cavity.7 Direct immunofluorescence demonstrates netlike intercellular IgG and C3 in the upper portion of the epidermis.11 Indirect immunofluorescence can help detect circulating IgG antibodies to Dsg-1, with guinea pig esophagus being the ideal substrate.11,12
First-line treatment of pemphigus foliaceus consists of systemic glucocorticoid therapy, often administered with azathioprine, methotrexate, or mycophenolate mofetil.2,6,13 Although first-line treatment is effective in 60% to 80% of patients,2 relapsing cases can be treated with cyclophosphamide, intravenous immunoglobulin, immunoadsorption, plasmapheresis, or rituximab.2
Rituximab is a chimeric monoclonal antibody targeting CD20+ B cells, leading to decreased antibody production, which has been shown to be effective in treating severe and refractory cases of pemphigus foliaceus.6,13Rituximab with short-course prednisone has been found to be more effective in achieving complete remission at 24 months than prednisone alone.14 In patients with contraindications to systemic glucocorticoid therapy, rituximab has been shown as an effective first-line therapy.15 One-quarter of patients treated with rituximab relapsed within 2 years of treatment6 (average time to relapse, 6–26 months).16 High-dose rituximab regimens, along with a higher number of rituximab treatment cycles, have been shown to prolong time to relapse.6 Further, higher baseline levels of Dsg-1 antibody have been correlated to earlier relapse and can be used following rituximab therapy to monitor disease progression.6,16
The differential diagnosis for pemphigus foliaceous includes disseminated blastomycosis, hypertrophic lupus erythematosus, sebopsoriasis, and secondary syphilis. Disseminated blastomycosis presents with cutaneous manifestations such as nodules, papules, or pustules evolving over weeks to months into ulcers with subsequent scarring.17 Hypertrophic lupus erythematosus presents with papules and nodules with associated keratotic scaling on the face, palms, and extensor surfaces of the limbs.18 Sebopsoriasis is characterized by well-defined lesions with an overlying scale distributed on the scalp, face, and chest.19 Secondary syphilis presents as early hyperpigmented macules transitioning to acral papulosquamous lesions involving the palms and soles.20
- Hans-Filho G, Aoki V, Hans Bittner NR, et al. Fogo selvagem: endemic pemphigus foliaceus. An Bras Dermatol. 2018;93:638-650.
- Jenson KK, Burr DM, Edwards BC. Case report: reatment of refractory pemphigus foliaceus with rituximab. Practical Dermatology. February 2016:33-36. Accessed August 27, 2021. https://practicaldermatology.com/articles/2016-feb/case-report -treatment-of-refractory-pemphigus-foliaceus-with-rituximab -financial-matters-aad-asds-resources
- Amagai M, Hashimoto T, Green KJ, et al. Antigen-specific immunoadsorption of pathogenic autoantibodies in pemphigus foliaceus. J Invest Dermatol. 1995;104:895-901.
- Mahoney MG, Wang Z, Rothenberger K, et al. Explanations for the clinical and microscopic localization of lesions in pemphigus foliaceus and vulgaris. J Clin Invest. 1999;103:461-468.
- Oktarina DAM, Sokol E, Kramer D, et al. Endocytosis of IgG, desmoglein 1, and plakoglobin in pemphigus foliaceus patient skin. Front Immunol. 2019;10:1-12.
- Kraft M, Worm M. Pemphigus foliaceus-repeated treatment with rituximab 7 years after initial response: a case report. Front Med. 2018;5:315.
- Hale EK. Pemphigus foliaceous. Dermatol Online J. 2002;8:9.
- Tavakolpour S. Pemphigus trigger factors: special focus on pemphigus vulgaris and pemphigus foliaceus. Arch Dermatol Res. 2018;310:95-106.
- A boobaker J, Morar N, Ramdial PK, et al. Pemphigus in South Africa. Int J Dermatol. 2001;40:115-119.
- Austin E, Millsop JW, Ely H, et al. Psoriasiform pemphigus foliaceus in an African American female: an important clinical manifestation. J Drugs Dermatol. 2018;17:471.
- Arbache ST, Nogueira TG, Delgado L, et al. Immunofluorescence testing in the diagnosis of autoimmune blistering diseases: overview of 10-year experience. An Bras Dermatol. 2014;89:885-889.
- Sabolinski ML, Beutner EH, Krasny S, et al. Substrate specificity of antiepithelial antibodies of pemphigus vulgaris and pemphigus foliaceus sera in immunofluorescence tests on monkey and guinea pig esophagus sections. J Invest Dermatol. 1987;88:545-549.
- Palacios-Álvarez I, Riquelme-McLoughlin C, Curto-Barredo L, et al. Rituximab treatment of pemphigus foliaceus: a retrospective study of 12 patients. J Am Acad Dermatol. 2021;85:484-486.
- Murrell DF, Sprecher E. Rituximab and short-course prednisone as the new gold standard for new-onset pemphigus vulgaris and pemphigus foliaceus. Br J Dermatol. 2017;177:1143-1144.
- Gregoriou S, Efthymiou O, Stefanaki C, et al. Management of pemphigus vulgaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2015;8:521-527.
- Saleh MA. A prospective study comparing patients with early and late relapsing pemphigus treated with rituximab. J Am Acad Dermatol. 2018;79:97-103.
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264.
- Herzum A, Gasparini G, Emanuele C, et al. Atypical and rare forms of cutaneous lupus erythematosus: the importance of the diagnosis for the best management of patients. Dermatology. 2013;1-10.
- Tull TJ, Noy M, Bunker CB, et al. Sebopsoriasis in patients with HIV: a case series of 20 patients. Br J Dermatol. 2016; 173:813-815.
- Balagula Y, Mattei P, Wisco OJ, et al. The great imitator revised: the spectrum of atypical cutaneous manifestations of secondary syphilis. Int J Dermatol. 2014;53:1434-1441.
The Diagnosis: Pemphigus Foliaceous
Laboratory workup including a complete blood cell count with differential, comprehensive metabolic panel, antinuclear antibodies, Sjögren syndrome A and B antibodies, hepatitis profile, rapid plasma reagin, HIV screen, aldolase, anti–Jo-1, T-Spot TB test (Quest Diagnostics), and tissue cultures was unremarkable. Two 4-mm punch biopsies were obtained from the left cheek and upper back, both of which demonstrated intragranular acantholysis suggestive of pemphigus foliaceous (Figure 1A). A subsequent punch biopsy from the right lower abdomen sent for direct immunofluorescence demonstrated netlike positivity of IgG and C3 in the upper epidermis (Figure 1B), and serum sent for indirect immunofluorescence demonstrated intercellular IgG antibodies to desmoglein (Dsg) 1 on monkey esophagus and positive Dsg-1 antibodies on enzyme-linked immunosorbent assay, confirming the diagnosis.

The patient was started on a 60-mg prednisone taper as well as dapsone 50 mg daily; the dapsone was titrated up to 100 mg daily. After tapering down to 10 mg daily of prednisone over 2 months and continuing dapsone with minimal improvement, he was given 2 infusions of rituximab 1000 mg 2 weeks apart. The scalp plaque was dramatically improved at 3-month follow-up (Figure 2), with partial improvement of the cheek plaques (Figure 3). Dapsone was increased to 150 mg daily, and he was encouraged to use triamcinolone acetonide ointment 0.1% twice daily, which led to further improvement.


Pemphigus foliaceus is an autoimmune blistering disease that most commonly occurs in middle-aged adults. It generally is less common than pemphigus vulgaris, except in Finland, Tunisia, and Brazil, where there is an endemic condition with an identical clinical and histological presentation known as fogo selvagem.1
The pathogenesis of pemphigus foliaceous is characterized by IgG autoantibodies against Dsg-1, a transmembrane glycoprotein involved in the cellular adhesion of keratinocytes, which is preferentially expressed in the superficial epidermis.2-7 Dysfunction of Dsg-1 results in the separation of superficial epidermal cells, resulting in intraepidermal blisters.2,7 In contrast to pemphigus vulgaris, there typically is a lack of oral mucosal involvement due to compensation by Dsg-3 in the mucosa.4 Potential triggers for pemphigus foliaceous include exposure to UV radiation; radiotherapy; pregnancy; physiologic stress; and drugs, most commonly captopril, penicillamine, and thiols.8
Pemphigus foliaceous lesions clinically appear as eroded and crusted lesions on an erythematous base, commonly in a seborrheic distribution on the face, scalp, and trunk with sparing of the oral mucosa,2,6 but lesions can progress to a widespread and more severe exfoliative dermatitis.7 Lesions also can appear as psoriasiform plaques and often are initially misdiagnosed as psoriasis, particularly in patients with skin of color.9,10
Diagnosis of pemphigus foliaceous typically is made using a combination of histology as well as both direct and indirect immunofluorescence. Histologically, pemphigus foliaceus presents with subcorneal acantholysis, which is most prominent in the granular layer and occasionally the presence of neutrophils and eosinophils in the blister cavity.7 Direct immunofluorescence demonstrates netlike intercellular IgG and C3 in the upper portion of the epidermis.11 Indirect immunofluorescence can help detect circulating IgG antibodies to Dsg-1, with guinea pig esophagus being the ideal substrate.11,12
First-line treatment of pemphigus foliaceus consists of systemic glucocorticoid therapy, often administered with azathioprine, methotrexate, or mycophenolate mofetil.2,6,13 Although first-line treatment is effective in 60% to 80% of patients,2 relapsing cases can be treated with cyclophosphamide, intravenous immunoglobulin, immunoadsorption, plasmapheresis, or rituximab.2
Rituximab is a chimeric monoclonal antibody targeting CD20+ B cells, leading to decreased antibody production, which has been shown to be effective in treating severe and refractory cases of pemphigus foliaceus.6,13Rituximab with short-course prednisone has been found to be more effective in achieving complete remission at 24 months than prednisone alone.14 In patients with contraindications to systemic glucocorticoid therapy, rituximab has been shown as an effective first-line therapy.15 One-quarter of patients treated with rituximab relapsed within 2 years of treatment6 (average time to relapse, 6–26 months).16 High-dose rituximab regimens, along with a higher number of rituximab treatment cycles, have been shown to prolong time to relapse.6 Further, higher baseline levels of Dsg-1 antibody have been correlated to earlier relapse and can be used following rituximab therapy to monitor disease progression.6,16
The differential diagnosis for pemphigus foliaceous includes disseminated blastomycosis, hypertrophic lupus erythematosus, sebopsoriasis, and secondary syphilis. Disseminated blastomycosis presents with cutaneous manifestations such as nodules, papules, or pustules evolving over weeks to months into ulcers with subsequent scarring.17 Hypertrophic lupus erythematosus presents with papules and nodules with associated keratotic scaling on the face, palms, and extensor surfaces of the limbs.18 Sebopsoriasis is characterized by well-defined lesions with an overlying scale distributed on the scalp, face, and chest.19 Secondary syphilis presents as early hyperpigmented macules transitioning to acral papulosquamous lesions involving the palms and soles.20
The Diagnosis: Pemphigus Foliaceous
Laboratory workup including a complete blood cell count with differential, comprehensive metabolic panel, antinuclear antibodies, Sjögren syndrome A and B antibodies, hepatitis profile, rapid plasma reagin, HIV screen, aldolase, anti–Jo-1, T-Spot TB test (Quest Diagnostics), and tissue cultures was unremarkable. Two 4-mm punch biopsies were obtained from the left cheek and upper back, both of which demonstrated intragranular acantholysis suggestive of pemphigus foliaceous (Figure 1A). A subsequent punch biopsy from the right lower abdomen sent for direct immunofluorescence demonstrated netlike positivity of IgG and C3 in the upper epidermis (Figure 1B), and serum sent for indirect immunofluorescence demonstrated intercellular IgG antibodies to desmoglein (Dsg) 1 on monkey esophagus and positive Dsg-1 antibodies on enzyme-linked immunosorbent assay, confirming the diagnosis.

The patient was started on a 60-mg prednisone taper as well as dapsone 50 mg daily; the dapsone was titrated up to 100 mg daily. After tapering down to 10 mg daily of prednisone over 2 months and continuing dapsone with minimal improvement, he was given 2 infusions of rituximab 1000 mg 2 weeks apart. The scalp plaque was dramatically improved at 3-month follow-up (Figure 2), with partial improvement of the cheek plaques (Figure 3). Dapsone was increased to 150 mg daily, and he was encouraged to use triamcinolone acetonide ointment 0.1% twice daily, which led to further improvement.


Pemphigus foliaceus is an autoimmune blistering disease that most commonly occurs in middle-aged adults. It generally is less common than pemphigus vulgaris, except in Finland, Tunisia, and Brazil, where there is an endemic condition with an identical clinical and histological presentation known as fogo selvagem.1
The pathogenesis of pemphigus foliaceous is characterized by IgG autoantibodies against Dsg-1, a transmembrane glycoprotein involved in the cellular adhesion of keratinocytes, which is preferentially expressed in the superficial epidermis.2-7 Dysfunction of Dsg-1 results in the separation of superficial epidermal cells, resulting in intraepidermal blisters.2,7 In contrast to pemphigus vulgaris, there typically is a lack of oral mucosal involvement due to compensation by Dsg-3 in the mucosa.4 Potential triggers for pemphigus foliaceous include exposure to UV radiation; radiotherapy; pregnancy; physiologic stress; and drugs, most commonly captopril, penicillamine, and thiols.8
Pemphigus foliaceous lesions clinically appear as eroded and crusted lesions on an erythematous base, commonly in a seborrheic distribution on the face, scalp, and trunk with sparing of the oral mucosa,2,6 but lesions can progress to a widespread and more severe exfoliative dermatitis.7 Lesions also can appear as psoriasiform plaques and often are initially misdiagnosed as psoriasis, particularly in patients with skin of color.9,10
Diagnosis of pemphigus foliaceous typically is made using a combination of histology as well as both direct and indirect immunofluorescence. Histologically, pemphigus foliaceus presents with subcorneal acantholysis, which is most prominent in the granular layer and occasionally the presence of neutrophils and eosinophils in the blister cavity.7 Direct immunofluorescence demonstrates netlike intercellular IgG and C3 in the upper portion of the epidermis.11 Indirect immunofluorescence can help detect circulating IgG antibodies to Dsg-1, with guinea pig esophagus being the ideal substrate.11,12
First-line treatment of pemphigus foliaceus consists of systemic glucocorticoid therapy, often administered with azathioprine, methotrexate, or mycophenolate mofetil.2,6,13 Although first-line treatment is effective in 60% to 80% of patients,2 relapsing cases can be treated with cyclophosphamide, intravenous immunoglobulin, immunoadsorption, plasmapheresis, or rituximab.2
Rituximab is a chimeric monoclonal antibody targeting CD20+ B cells, leading to decreased antibody production, which has been shown to be effective in treating severe and refractory cases of pemphigus foliaceus.6,13Rituximab with short-course prednisone has been found to be more effective in achieving complete remission at 24 months than prednisone alone.14 In patients with contraindications to systemic glucocorticoid therapy, rituximab has been shown as an effective first-line therapy.15 One-quarter of patients treated with rituximab relapsed within 2 years of treatment6 (average time to relapse, 6–26 months).16 High-dose rituximab regimens, along with a higher number of rituximab treatment cycles, have been shown to prolong time to relapse.6 Further, higher baseline levels of Dsg-1 antibody have been correlated to earlier relapse and can be used following rituximab therapy to monitor disease progression.6,16
The differential diagnosis for pemphigus foliaceous includes disseminated blastomycosis, hypertrophic lupus erythematosus, sebopsoriasis, and secondary syphilis. Disseminated blastomycosis presents with cutaneous manifestations such as nodules, papules, or pustules evolving over weeks to months into ulcers with subsequent scarring.17 Hypertrophic lupus erythematosus presents with papules and nodules with associated keratotic scaling on the face, palms, and extensor surfaces of the limbs.18 Sebopsoriasis is characterized by well-defined lesions with an overlying scale distributed on the scalp, face, and chest.19 Secondary syphilis presents as early hyperpigmented macules transitioning to acral papulosquamous lesions involving the palms and soles.20
- Hans-Filho G, Aoki V, Hans Bittner NR, et al. Fogo selvagem: endemic pemphigus foliaceus. An Bras Dermatol. 2018;93:638-650.
- Jenson KK, Burr DM, Edwards BC. Case report: reatment of refractory pemphigus foliaceus with rituximab. Practical Dermatology. February 2016:33-36. Accessed August 27, 2021. https://practicaldermatology.com/articles/2016-feb/case-report -treatment-of-refractory-pemphigus-foliaceus-with-rituximab -financial-matters-aad-asds-resources
- Amagai M, Hashimoto T, Green KJ, et al. Antigen-specific immunoadsorption of pathogenic autoantibodies in pemphigus foliaceus. J Invest Dermatol. 1995;104:895-901.
- Mahoney MG, Wang Z, Rothenberger K, et al. Explanations for the clinical and microscopic localization of lesions in pemphigus foliaceus and vulgaris. J Clin Invest. 1999;103:461-468.
- Oktarina DAM, Sokol E, Kramer D, et al. Endocytosis of IgG, desmoglein 1, and plakoglobin in pemphigus foliaceus patient skin. Front Immunol. 2019;10:1-12.
- Kraft M, Worm M. Pemphigus foliaceus-repeated treatment with rituximab 7 years after initial response: a case report. Front Med. 2018;5:315.
- Hale EK. Pemphigus foliaceous. Dermatol Online J. 2002;8:9.
- Tavakolpour S. Pemphigus trigger factors: special focus on pemphigus vulgaris and pemphigus foliaceus. Arch Dermatol Res. 2018;310:95-106.
- A boobaker J, Morar N, Ramdial PK, et al. Pemphigus in South Africa. Int J Dermatol. 2001;40:115-119.
- Austin E, Millsop JW, Ely H, et al. Psoriasiform pemphigus foliaceus in an African American female: an important clinical manifestation. J Drugs Dermatol. 2018;17:471.
- Arbache ST, Nogueira TG, Delgado L, et al. Immunofluorescence testing in the diagnosis of autoimmune blistering diseases: overview of 10-year experience. An Bras Dermatol. 2014;89:885-889.
- Sabolinski ML, Beutner EH, Krasny S, et al. Substrate specificity of antiepithelial antibodies of pemphigus vulgaris and pemphigus foliaceus sera in immunofluorescence tests on monkey and guinea pig esophagus sections. J Invest Dermatol. 1987;88:545-549.
- Palacios-Álvarez I, Riquelme-McLoughlin C, Curto-Barredo L, et al. Rituximab treatment of pemphigus foliaceus: a retrospective study of 12 patients. J Am Acad Dermatol. 2021;85:484-486.
- Murrell DF, Sprecher E. Rituximab and short-course prednisone as the new gold standard for new-onset pemphigus vulgaris and pemphigus foliaceus. Br J Dermatol. 2017;177:1143-1144.
- Gregoriou S, Efthymiou O, Stefanaki C, et al. Management of pemphigus vulgaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2015;8:521-527.
- Saleh MA. A prospective study comparing patients with early and late relapsing pemphigus treated with rituximab. J Am Acad Dermatol. 2018;79:97-103.
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264.
- Herzum A, Gasparini G, Emanuele C, et al. Atypical and rare forms of cutaneous lupus erythematosus: the importance of the diagnosis for the best management of patients. Dermatology. 2013;1-10.
- Tull TJ, Noy M, Bunker CB, et al. Sebopsoriasis in patients with HIV: a case series of 20 patients. Br J Dermatol. 2016; 173:813-815.
- Balagula Y, Mattei P, Wisco OJ, et al. The great imitator revised: the spectrum of atypical cutaneous manifestations of secondary syphilis. Int J Dermatol. 2014;53:1434-1441.
- Hans-Filho G, Aoki V, Hans Bittner NR, et al. Fogo selvagem: endemic pemphigus foliaceus. An Bras Dermatol. 2018;93:638-650.
- Jenson KK, Burr DM, Edwards BC. Case report: reatment of refractory pemphigus foliaceus with rituximab. Practical Dermatology. February 2016:33-36. Accessed August 27, 2021. https://practicaldermatology.com/articles/2016-feb/case-report -treatment-of-refractory-pemphigus-foliaceus-with-rituximab -financial-matters-aad-asds-resources
- Amagai M, Hashimoto T, Green KJ, et al. Antigen-specific immunoadsorption of pathogenic autoantibodies in pemphigus foliaceus. J Invest Dermatol. 1995;104:895-901.
- Mahoney MG, Wang Z, Rothenberger K, et al. Explanations for the clinical and microscopic localization of lesions in pemphigus foliaceus and vulgaris. J Clin Invest. 1999;103:461-468.
- Oktarina DAM, Sokol E, Kramer D, et al. Endocytosis of IgG, desmoglein 1, and plakoglobin in pemphigus foliaceus patient skin. Front Immunol. 2019;10:1-12.
- Kraft M, Worm M. Pemphigus foliaceus-repeated treatment with rituximab 7 years after initial response: a case report. Front Med. 2018;5:315.
- Hale EK. Pemphigus foliaceous. Dermatol Online J. 2002;8:9.
- Tavakolpour S. Pemphigus trigger factors: special focus on pemphigus vulgaris and pemphigus foliaceus. Arch Dermatol Res. 2018;310:95-106.
- A boobaker J, Morar N, Ramdial PK, et al. Pemphigus in South Africa. Int J Dermatol. 2001;40:115-119.
- Austin E, Millsop JW, Ely H, et al. Psoriasiform pemphigus foliaceus in an African American female: an important clinical manifestation. J Drugs Dermatol. 2018;17:471.
- Arbache ST, Nogueira TG, Delgado L, et al. Immunofluorescence testing in the diagnosis of autoimmune blistering diseases: overview of 10-year experience. An Bras Dermatol. 2014;89:885-889.
- Sabolinski ML, Beutner EH, Krasny S, et al. Substrate specificity of antiepithelial antibodies of pemphigus vulgaris and pemphigus foliaceus sera in immunofluorescence tests on monkey and guinea pig esophagus sections. J Invest Dermatol. 1987;88:545-549.
- Palacios-Álvarez I, Riquelme-McLoughlin C, Curto-Barredo L, et al. Rituximab treatment of pemphigus foliaceus: a retrospective study of 12 patients. J Am Acad Dermatol. 2021;85:484-486.
- Murrell DF, Sprecher E. Rituximab and short-course prednisone as the new gold standard for new-onset pemphigus vulgaris and pemphigus foliaceus. Br J Dermatol. 2017;177:1143-1144.
- Gregoriou S, Efthymiou O, Stefanaki C, et al. Management of pemphigus vulgaris: challenges and solutions. Clin Cosmet Investig Dermatol. 2015;8:521-527.
- Saleh MA. A prospective study comparing patients with early and late relapsing pemphigus treated with rituximab. J Am Acad Dermatol. 2018;79:97-103.
- Castillo CG, Kauffman CA, Miceli MH. Blastomycosis. Infect Dis Clin North Am. 2016;30:247-264.
- Herzum A, Gasparini G, Emanuele C, et al. Atypical and rare forms of cutaneous lupus erythematosus: the importance of the diagnosis for the best management of patients. Dermatology. 2013;1-10.
- Tull TJ, Noy M, Bunker CB, et al. Sebopsoriasis in patients with HIV: a case series of 20 patients. Br J Dermatol. 2016; 173:813-815.
- Balagula Y, Mattei P, Wisco OJ, et al. The great imitator revised: the spectrum of atypical cutaneous manifestations of secondary syphilis. Int J Dermatol. 2014;53:1434-1441.
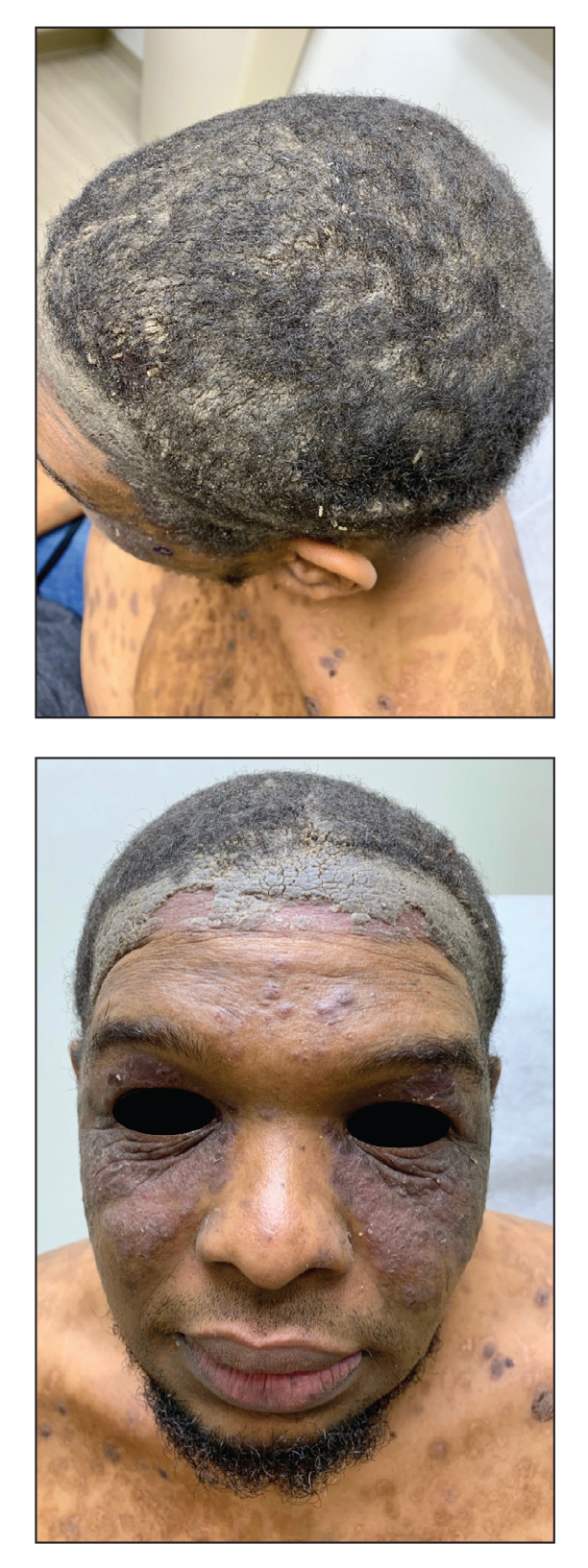
A 40-year-old Black man presented for evaluation of a thick plaque throughout the scalp (top), scaly plaques on the cheeks (bottom), and a spreading rash on the trunk that had progressed over the last few months. He had no relevant medical history, took no medications, and was in a monogamous relationship with a female partner. He previously saw an outside dermatologist who gave him triamcinolone cream, which was mildly helpful. Physical examination revealed a thick verrucous plaque throughout the scalp extending onto the forehead; thick plaques on the cheeks; and numerous, thinly eroded lesions on the trunk. Biopsies and a laboratory workup were performed.
CDC panel unanimously backs Pfizer vax, fortifying FDA approval
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
An independent expert panel within the Centers for Disease Control and Prevention (CDC) has studied the potential benefits and risks of the Pfizer-BioNTech COVID-19 vaccine and voted unanimously to recommend the shots for all Americans ages 16 and older.
The vaccine was fully approved by the U.S. Food and Drug Administration (FDA) last week.
The inoculation is still available to teens ages 12 to 15 under an emergency use authorization from the FDA.
ACIP now sends its recommendation to the CDC Director Rochelle Walensky, MD, for her sign off.
After reviewing the evidence behind the vaccine, panel member Sarah Long, MD, a professor of pediatrics at Drexel University College of Medicine, Philadelphia, said she couldn’t recall another instance where panelists had so much data on which to base their recommendation.
“This vaccine is worthy of the trust of the American people,” she said.
Doctors across the country use vaccines in line with the recommendations made by the ACIP. Their approval typically means that private and government insurers will cover the cost of the shots. In the case of the COVID-19 vaccines, the government is already picking up the tab.
Few surprises
The panel’s independent review of the vaccine’s effectiveness from nine studies held few surprises.
They found the Pfizer vaccine prevented a COVID infection with symptoms about 90%–92% of the time, at least for the first 4 months after the second shot. Protection against hospitalization and death was even higher.
The vaccine was about 89% effective at preventing a COVID infection without symptoms, according to a pooled estimate of five studies.
The data included in the review was updated only through March 13 of this year, however, and does not reflect the impact of further waning of immunity or the impact of the Delta variant.
In making their recommendation, the panel got an update on the safety of the vaccines, which have now been used in the United States for about 9 months.
The rate of anaphylaxis has settled at around five cases for every million shots given, according to the ACIP’s review of the evidence.
Cases of myocarditis and pericarditis were more common after getting a Pfizer-BioNTech vaccine than would be expected to happen naturally in the general population, but the risk was still very rare, and elevated primarily for men younger than age 30.
Out of 17 million second doses of Pfizer-BioNTech vaccines in the United States, there have been 327 confirmed cases of myocarditis reported to the Vaccine Adverse Event Reporting System in people who are younger than age 30. The average hospital stay for a myocarditis cases is 1 to 2 days.
So far, no one in the United States diagnosed with myocarditis after vaccination has died.
What’s more, the risk of myocarditis after vaccination was dwarfed by the risk of myocarditis after a COVID infection. The risk of myocarditis after a COVID infection was 6 to 34 times higher than the risk after receiving an mRNA vaccine.
About 11% of people who get the vaccine experience a serious reaction to the shot, compared with about 3% in the placebo group. Serious reactions were defined as pain; swelling or redness at the injection site that interferes with activity; needing to visit the hospital or ER for pain; tissue necrosis, or having skin slough off; high fever; vomiting that requires hydration; persistent diarrhea; severe headache; or muscle pain/severe joint pain.
“Safe and effective”
After hearing a presentation on the state of the pandemic in the US, some panel members were struck and shaken that 38% of Americans who are eligible are still not fully vaccinated.
Pablo Sanchez, MD, a pediatrician at Nationwide Children’s Hospital in Columbus, Ohio, said, “We’re doing an abysmal job vaccinating the American people. The message has to go out that the vaccines are safe and effective.”
A version of this story first appeared on Medscape.com.
Biologic benefit in psoriasis might extend to arthritis prevention
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
Receiving treatment with a biologic medication, compared with no biologic treatment, appeared to be associated with a lower risk for developing psoriatic arthritis (PsA) in patients with psoriasis.
That’s according to the results of a nested case-control study involving electronic medical record data from an Israeli health maintenance organization in Arthritis & Rheumatology. Compared with no biologic treatment, the risk for developing PsA among PsO patients was reduced by 39%.
This study shows “a statistically and clinically significant lower risk for developing PsA among patients receiving biologic medications for psoriasis treatment,” wrote Yael Shalev Rosenthal, MPH, of the Sackler Faculty of Medicine, Tel Aviv University and colleagues. “The results suggest considering treatment with biologic medications in patients [who] present with significant risk factors for PsA at an earlier stage of treatment.”
“It would be nice to believe this story, but I don’t think we can based on the evidence we’ve got so far,” commented Philip Helliwell, PhD, DM, in an interview.
Dr. Helliwell, who is professor of clinical rheumatology at the University of Leeds (England) and an Honorary Consultant Rheumatologist for the Leeds Teaching Hospitals and Bradford Teaching Hospitals NHS Trust, noted that there were several issues with the current evidence.
Aside from their often retrospective or nonrandomized nature, prior analyses, including the current one, were based on EMR data.
“There’s actually no face-to-face patient contact going on here. It’s all done on coding, and coding can be unreliable,” Dr. Helliwell said.
While the study’s findings are “in line with other studies that have looked at this, and suggest that if you get a biologic, you’re less likely to get PsA with your psoriasis, there could be lots of reasons why.”
The big problem here is confounding by indication. “You don’t get on a biologic unless you’ve got bad psoriasis,” Dr. Helliwell explained. The Israeli criteria for starting a biologic are much higher than in the United Kingdom, he added, requiring more than 50% of patients’ body surface area to be affected, or a Psoriasis Area and Severity Index score of more than 50. Moreover, people with bad psoriasis are more likely to get PsA. This, however, makes the results more impressive.
Confounding by indication is an issue with this study, agreed consultant rheumatologist Adewale Adebajo, PhD, in a separate interview. He acknowledged, however, that the study’s authors did try to account for this by limiting the timescale of their analysis to the first 10 years of biologic therapy. They also used the usual methods of propensity score matching and multivariate Cox regression analysis to hopefully iron out any differences between the two groups of patients.
Study details and results
Ms. Rosenthal and coauthors analyzed EMR data on patients with psoriasis but not PsA that were logged in the Maccabi Healthcare Services (MHS) database. The MHS is the second-largest health maintenance organization in Israel, insuring over 2 million members, the researchers said.
In all, 663 patients with psoriasis but not PsA before or at initiation of biologic treatment were included in their analysis and matched to a control group of 663 patients with psoriasis who had not received biologic treatment. Propensity score matching was used to iron out some differences in baseline characteristics that had been seen between the groups, such as older age at diagnosis, higher body mass index, and a longer time between diagnosis and treatment seen in patients treated without biologics.
After adjusting for multiple risk factors and confounders, “the control group still had a significantly higher risk for PsA, compared to the biological treatment group,” the researchers wrote. Indeed, the adjusted hazard ratio was 1.39, with a 95% confidence interval between 1.03 and 1.87.
An ‘intriguing study’
“This is a retrospective study, and it has all the faults of a retrospective study,” said Dr. Adebajo, associate medical director for research and development at Barnsley (England) NHS Foundation Trust. But “these were patients who hopefully hadn’t yet developed psoriatic arthritis, although it is difficult to exclude subclinical psoriatic arthritis.”
The ideal would of course be to look at patients prospectively, but a randomized clinical trial would be unlikely to ever be conducted, Dr. Helliwell noted. “It would be unfair to randomize people who have got bad psoriasis and need a biologic to placebo just to prove the point really,” he said. “Getting control groups in this arena is very difficult.”
That doesn’t mean that prospective evaluation is not possible. Dr. Adebajo noted that there were already cohorts of newly diagnosed patients who were being prospectively followed up and those could perhaps be used to look at the question again in the future.
“You’re then looking at the natural history, the natural outcome, and you don’t need to worry about confounding because you’re just collecting all of the information as you go along.”
The idea that biologics might slow or even prevent the onset of PsA is “an interesting and enchanting hypothesis,” Dr. Adebajo said. “The study doesn’t prove the hypothesis, but it’s an intriguing study because it doesn’t disprove the hypothesis either.
“It gives us food for thought and a basis for further studies,” as well as some “encouragement to perhaps use biologics earlier because there may be additional benefits of doing so.”
That’s still to be proven of course, as it has been reported that patients with psoriasis can develop PsA while taking biologics.
“Clinically, that’s what we see in the combined clinic. We get people referred with psoriasis [who are] already on a biologic who developed musculoskeletal problems,” Dr. Helliwell said.
“It would be nice to believe” that biologics prevent or slow PsA in patients with psoriasis, Dr. Helliwell added, but I’m not sure these data are conclusive. From this study we know nothing about the phenotype of psoriasis, which is important in the development of PsA. In addition, we know that of the 30% of people with psoriasis who develop PsA, about half of these are undiagnosed at the time of such studies. In that case, what the biologic is doing is just treating preexisting PsA. If you count those numbers up, some of the differences between the two groups seen in this study are accounted for. From registry data there is no way of checking this.”
No external funding was used for the study. One author acknowledged acting as an investigator, adviser, or consultant to several pharmaceutical companies including AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Coherus, Dexcel Pharma, Eli Lilly, Janssen, Novartis, and Pfizer. All other authors had nothing to disclose.
Dr. Helliwell and Dr. Adebajo had no conflicts of interest.
FROM ARTHRITIS & RHEUMATOLOGY
Screening and management of comorbidities could help reduce preventable psoriasis hospitalizations
Key clinical point: Although rates of principal psoriasis hospitalizations have decreased among patients with psoriasis in the last 2 decades in the United States, hospitalization for non-psoriatic reasons has increased, which may be attributed to increased comorbidity burden driving admissions because of comorbidities.
Major finding: Between 1998 and 2018, the incidence of hospitalizations with either principal or secondary diagnosis of psoriasis increased from 17.9 to 52.0 per 100,000 persons, the proportion of patients with psoriasis hospitalized with psoriasis as principal diagnosis reduced from 4.1% to 1.0%, and those with Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9% (all adjusted P-trend < .0001).
Study details: This was a 21-year longitudinal trend analysis of the National Inpatient Sample database between 1998 and 2018 including adults with a principal or secondary diagnosis of psoriasis.
Disclosures: The study did not receive any funding. The authors declared no conflict of interests.
Source: Edigin E et al. J Eur Acad Dermatol Venereol. 2021 Aug 9. doi: 10.1111/jdv.17590.
Key clinical point: Although rates of principal psoriasis hospitalizations have decreased among patients with psoriasis in the last 2 decades in the United States, hospitalization for non-psoriatic reasons has increased, which may be attributed to increased comorbidity burden driving admissions because of comorbidities.
Major finding: Between 1998 and 2018, the incidence of hospitalizations with either principal or secondary diagnosis of psoriasis increased from 17.9 to 52.0 per 100,000 persons, the proportion of patients with psoriasis hospitalized with psoriasis as principal diagnosis reduced from 4.1% to 1.0%, and those with Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9% (all adjusted P-trend < .0001).
Study details: This was a 21-year longitudinal trend analysis of the National Inpatient Sample database between 1998 and 2018 including adults with a principal or secondary diagnosis of psoriasis.
Disclosures: The study did not receive any funding. The authors declared no conflict of interests.
Source: Edigin E et al. J Eur Acad Dermatol Venereol. 2021 Aug 9. doi: 10.1111/jdv.17590.
Key clinical point: Although rates of principal psoriasis hospitalizations have decreased among patients with psoriasis in the last 2 decades in the United States, hospitalization for non-psoriatic reasons has increased, which may be attributed to increased comorbidity burden driving admissions because of comorbidities.
Major finding: Between 1998 and 2018, the incidence of hospitalizations with either principal or secondary diagnosis of psoriasis increased from 17.9 to 52.0 per 100,000 persons, the proportion of patients with psoriasis hospitalized with psoriasis as principal diagnosis reduced from 4.1% to 1.0%, and those with Charlson Comorbidity Index score of 3 or higher increased from 13.9% to 30.9% (all adjusted P-trend < .0001).
Study details: This was a 21-year longitudinal trend analysis of the National Inpatient Sample database between 1998 and 2018 including adults with a principal or secondary diagnosis of psoriasis.
Disclosures: The study did not receive any funding. The authors declared no conflict of interests.
Source: Edigin E et al. J Eur Acad Dermatol Venereol. 2021 Aug 9. doi: 10.1111/jdv.17590.
Low hemoglobin elevates psoriasis risk in patients with chronic kidney disease
Key clinical point: A significant relation was observed between low hemoglobin levels and increased risk for psoriasis in patients with chronic kidney disease (CKD), indicating that proactive treatment for inflammation might help manage both anemia and psoriasis in patients with CKD.
Major finding: During a mean follow-up period of 6.16±1.02 years, 2.39% of patients with CKD developed psoriasis with cumulative incidence higher in patients with vs without anemia (P less than .0001). The risk for psoriasis was significantly higher in patients with vs without anemia (adjusted hazard ratio, 1.109; P < .0001).
Study details: Findings are from a retrospective cohort study of 576,461 patients with CKD.
Disclosures: This study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declare no competing interests.
Source: Lee SH et al. Sci Rep. 2021 Jul 20. doi: 10.1038/s41598-021-94165-w.
Key clinical point: A significant relation was observed between low hemoglobin levels and increased risk for psoriasis in patients with chronic kidney disease (CKD), indicating that proactive treatment for inflammation might help manage both anemia and psoriasis in patients with CKD.
Major finding: During a mean follow-up period of 6.16±1.02 years, 2.39% of patients with CKD developed psoriasis with cumulative incidence higher in patients with vs without anemia (P less than .0001). The risk for psoriasis was significantly higher in patients with vs without anemia (adjusted hazard ratio, 1.109; P < .0001).
Study details: Findings are from a retrospective cohort study of 576,461 patients with CKD.
Disclosures: This study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declare no competing interests.
Source: Lee SH et al. Sci Rep. 2021 Jul 20. doi: 10.1038/s41598-021-94165-w.
Key clinical point: A significant relation was observed between low hemoglobin levels and increased risk for psoriasis in patients with chronic kidney disease (CKD), indicating that proactive treatment for inflammation might help manage both anemia and psoriasis in patients with CKD.
Major finding: During a mean follow-up period of 6.16±1.02 years, 2.39% of patients with CKD developed psoriasis with cumulative incidence higher in patients with vs without anemia (P less than .0001). The risk for psoriasis was significantly higher in patients with vs without anemia (adjusted hazard ratio, 1.109; P < .0001).
Study details: Findings are from a retrospective cohort study of 576,461 patients with CKD.
Disclosures: This study was supported by a National Research Foundation of Korea grant funded by the Korean government. The authors declare no competing interests.
Source: Lee SH et al. Sci Rep. 2021 Jul 20. doi: 10.1038/s41598-021-94165-w.