User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Hiccups in patients with cancer often overlooked, undertreated
But even if recognized, hiccups may not be treated effectively, according to a national survey of cancer care clinicians.
When poorly controlled, persistent hiccups can affect a patient’s quality of life, with 40% of survey respondents considering chronic hiccups “much more” or “somewhat more” severe than nausea and vomiting.
Overall, the findings indicate that patients with cancer who develop persistent hiccups are “truly suffering,” the authors wrote.
The survey results were published online recently in the American Journal of Hospice and Palliative Medicine.
Hiccups may simply be a nuisance for most, but these spasms can become problematic for patients with cancer, leading to sleep deprivation, fatigue, aspiration pneumonia, compromised food intake, weight loss, pain, and even death.
Hiccups can develop when the nerve that controls the diaphragm becomes irritated, which can be triggered by certain chemotherapy drugs.
Yet few studies have focused on hiccups in patients with cancer and none, until now, has sought the perspectives of cancer care clinicians.
Aminah Jatoi, MD, medical oncologist with the Mayo Clinic in Rochester, Minn., and two Mayo colleagues developed a survey, alongside MeterHealth, which this news organization distributed to clinicians with an interest in cancer care.
The survey gauged clinicians’ awareness or lack of awareness about clinically significant hiccups as well as treatments for hiccups and whether they consider hiccups an unmet palliative need.
A total of 684 clinicians completed two eligibility screening questions, which required them to have cared for more than 10 patients with cancer in the past 6 months with clinically significant hiccups (defined as hiccups that lasted more than 48 hours or occurred from cancer or cancer care).
Among 113 eligible health care professionals, 90 completed the survey: 42 physicians, 29 nurses, 15 nurse practitioners, and 4 physician assistants.
The survey revealed three key issues.
The first is that hiccups appear to be an underrecognized issue.
Among health care professionals who answered the eligibility screening questions, fewer than 20% reported caring for more than 10 patients with cancer in the past 6 months who had persistent hiccups. Most of these clinicians reported caring for more than 1,000 patients per year.
Given that 15%-40% of patients with cancer report hiccups, this finding suggests that hiccups are not widely recognized by health care professionals.
Second: The survey data showed that hiccups often increase patients’ anxiety, fatigue, and sleep problems and can decrease productivity at work or school.
In fact, when comparing hiccups to nausea and vomiting – sometimes described as one of the most severe side effects of cancer care – 40% of respondents rated hiccups as “much more” or “somewhat more” severe than nausea and vomiting for their patients and 38% rated the severity of the two issues as “about the same.”
Finally, even when hiccups are recognized and treated, about 20% of respondents said that current therapies are not very effective, and more treatment options are needed.
Among the survey respondents, the most frequently prescribed medications for chronic hiccups were the antipsychotic chlorpromazine, the muscle relaxant baclofen (Lioresal), the antiemetic metoclopramide (Metozolv ODT, Reglan), and the anticonvulsants gabapentin (Neurontin) and carbamazepine (Tegretol).
Survey respondents who provided comments about current treatments for hiccups highlighted a range of challenges. One respondent said, “When current therapies do not work, it can be very demoralizing to our patients.” Another said, “I feel like it is a gamble whether treatment for hiccups will work or not.”
Still another felt that while current treatments work “quite well to halt hiccups,” they come with side effects which can be “quite severe.”
These results “clearly point to the unmet needs of hiccups in patients with cancer and should prompt more research aimed at generating more palliative options,” the authors said.
This research had no commercial funding. MeterHealth reviewed the manuscript and provided input on the accuracy of methods and results. Dr. Jatoi reports serving on an advisory board for MeterHealth (honoraria to institution).
A version of this article first appeared on Medscape.com.
But even if recognized, hiccups may not be treated effectively, according to a national survey of cancer care clinicians.
When poorly controlled, persistent hiccups can affect a patient’s quality of life, with 40% of survey respondents considering chronic hiccups “much more” or “somewhat more” severe than nausea and vomiting.
Overall, the findings indicate that patients with cancer who develop persistent hiccups are “truly suffering,” the authors wrote.
The survey results were published online recently in the American Journal of Hospice and Palliative Medicine.
Hiccups may simply be a nuisance for most, but these spasms can become problematic for patients with cancer, leading to sleep deprivation, fatigue, aspiration pneumonia, compromised food intake, weight loss, pain, and even death.
Hiccups can develop when the nerve that controls the diaphragm becomes irritated, which can be triggered by certain chemotherapy drugs.
Yet few studies have focused on hiccups in patients with cancer and none, until now, has sought the perspectives of cancer care clinicians.
Aminah Jatoi, MD, medical oncologist with the Mayo Clinic in Rochester, Minn., and two Mayo colleagues developed a survey, alongside MeterHealth, which this news organization distributed to clinicians with an interest in cancer care.
The survey gauged clinicians’ awareness or lack of awareness about clinically significant hiccups as well as treatments for hiccups and whether they consider hiccups an unmet palliative need.
A total of 684 clinicians completed two eligibility screening questions, which required them to have cared for more than 10 patients with cancer in the past 6 months with clinically significant hiccups (defined as hiccups that lasted more than 48 hours or occurred from cancer or cancer care).
Among 113 eligible health care professionals, 90 completed the survey: 42 physicians, 29 nurses, 15 nurse practitioners, and 4 physician assistants.
The survey revealed three key issues.
The first is that hiccups appear to be an underrecognized issue.
Among health care professionals who answered the eligibility screening questions, fewer than 20% reported caring for more than 10 patients with cancer in the past 6 months who had persistent hiccups. Most of these clinicians reported caring for more than 1,000 patients per year.
Given that 15%-40% of patients with cancer report hiccups, this finding suggests that hiccups are not widely recognized by health care professionals.
Second: The survey data showed that hiccups often increase patients’ anxiety, fatigue, and sleep problems and can decrease productivity at work or school.
In fact, when comparing hiccups to nausea and vomiting – sometimes described as one of the most severe side effects of cancer care – 40% of respondents rated hiccups as “much more” or “somewhat more” severe than nausea and vomiting for their patients and 38% rated the severity of the two issues as “about the same.”
Finally, even when hiccups are recognized and treated, about 20% of respondents said that current therapies are not very effective, and more treatment options are needed.
Among the survey respondents, the most frequently prescribed medications for chronic hiccups were the antipsychotic chlorpromazine, the muscle relaxant baclofen (Lioresal), the antiemetic metoclopramide (Metozolv ODT, Reglan), and the anticonvulsants gabapentin (Neurontin) and carbamazepine (Tegretol).
Survey respondents who provided comments about current treatments for hiccups highlighted a range of challenges. One respondent said, “When current therapies do not work, it can be very demoralizing to our patients.” Another said, “I feel like it is a gamble whether treatment for hiccups will work or not.”
Still another felt that while current treatments work “quite well to halt hiccups,” they come with side effects which can be “quite severe.”
These results “clearly point to the unmet needs of hiccups in patients with cancer and should prompt more research aimed at generating more palliative options,” the authors said.
This research had no commercial funding. MeterHealth reviewed the manuscript and provided input on the accuracy of methods and results. Dr. Jatoi reports serving on an advisory board for MeterHealth (honoraria to institution).
A version of this article first appeared on Medscape.com.
But even if recognized, hiccups may not be treated effectively, according to a national survey of cancer care clinicians.
When poorly controlled, persistent hiccups can affect a patient’s quality of life, with 40% of survey respondents considering chronic hiccups “much more” or “somewhat more” severe than nausea and vomiting.
Overall, the findings indicate that patients with cancer who develop persistent hiccups are “truly suffering,” the authors wrote.
The survey results were published online recently in the American Journal of Hospice and Palliative Medicine.
Hiccups may simply be a nuisance for most, but these spasms can become problematic for patients with cancer, leading to sleep deprivation, fatigue, aspiration pneumonia, compromised food intake, weight loss, pain, and even death.
Hiccups can develop when the nerve that controls the diaphragm becomes irritated, which can be triggered by certain chemotherapy drugs.
Yet few studies have focused on hiccups in patients with cancer and none, until now, has sought the perspectives of cancer care clinicians.
Aminah Jatoi, MD, medical oncologist with the Mayo Clinic in Rochester, Minn., and two Mayo colleagues developed a survey, alongside MeterHealth, which this news organization distributed to clinicians with an interest in cancer care.
The survey gauged clinicians’ awareness or lack of awareness about clinically significant hiccups as well as treatments for hiccups and whether they consider hiccups an unmet palliative need.
A total of 684 clinicians completed two eligibility screening questions, which required them to have cared for more than 10 patients with cancer in the past 6 months with clinically significant hiccups (defined as hiccups that lasted more than 48 hours or occurred from cancer or cancer care).
Among 113 eligible health care professionals, 90 completed the survey: 42 physicians, 29 nurses, 15 nurse practitioners, and 4 physician assistants.
The survey revealed three key issues.
The first is that hiccups appear to be an underrecognized issue.
Among health care professionals who answered the eligibility screening questions, fewer than 20% reported caring for more than 10 patients with cancer in the past 6 months who had persistent hiccups. Most of these clinicians reported caring for more than 1,000 patients per year.
Given that 15%-40% of patients with cancer report hiccups, this finding suggests that hiccups are not widely recognized by health care professionals.
Second: The survey data showed that hiccups often increase patients’ anxiety, fatigue, and sleep problems and can decrease productivity at work or school.
In fact, when comparing hiccups to nausea and vomiting – sometimes described as one of the most severe side effects of cancer care – 40% of respondents rated hiccups as “much more” or “somewhat more” severe than nausea and vomiting for their patients and 38% rated the severity of the two issues as “about the same.”
Finally, even when hiccups are recognized and treated, about 20% of respondents said that current therapies are not very effective, and more treatment options are needed.
Among the survey respondents, the most frequently prescribed medications for chronic hiccups were the antipsychotic chlorpromazine, the muscle relaxant baclofen (Lioresal), the antiemetic metoclopramide (Metozolv ODT, Reglan), and the anticonvulsants gabapentin (Neurontin) and carbamazepine (Tegretol).
Survey respondents who provided comments about current treatments for hiccups highlighted a range of challenges. One respondent said, “When current therapies do not work, it can be very demoralizing to our patients.” Another said, “I feel like it is a gamble whether treatment for hiccups will work or not.”
Still another felt that while current treatments work “quite well to halt hiccups,” they come with side effects which can be “quite severe.”
These results “clearly point to the unmet needs of hiccups in patients with cancer and should prompt more research aimed at generating more palliative options,” the authors said.
This research had no commercial funding. MeterHealth reviewed the manuscript and provided input on the accuracy of methods and results. Dr. Jatoi reports serving on an advisory board for MeterHealth (honoraria to institution).
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF HOSPICE AND PALLIATIVE MEDICINE
Patient harm, not malpractice, top of mind for emergency medicine physicians
according to a study published in JAMA Network Open.
The cross-sectional study was conducted by researchers from Soroka University Medical Center, Israel; the University of Massachusetts, Worcester; Beth Israel Deaconess Medical Center; Harvard Medical School, Boston; and the University of Massachusetts, Amherst.
Online survey responses were collected from 1,222 emergency department attending physicians and advanced practice clinicians (APCs) in acute care hospitals throughout Massachusetts from January to September 2020.
Participants were asked to rank their level of agreement – from “strongly disagree” to “strongly agree” – with two statements: “In my day-to-day practice, I am fearful of making a mistake which results in [1] harm to the patient” (fear of harm) and [2] “being sued” (fear of suit).
The average age of the participants was about 44 years; 54.2% were men, 45.1% were women, and 0.7% were of other gender. Approximately 70% of responses were from MDs or DOs, and the remainder were from nurse practitioners and physician assistants. Participants had between 5 and 19 years of experience (median, 10 years).
The study found that the mean score was greater with regard to fear of harm than to fear of suit, regardless of clinician type, experience, or sex and whether the survey was completed before or after the start of the COVID-19 pandemic. There was no significant difference in mean scores regarding fear of suit before the pandemic and after it.
“Our data show a significantly greater fear of harming a patient than a fear of a malpractice suit,” Linda Isbell, PhD, professor of psychology at the University of Massachusetts, Amherst, who is one of the study’s authors, told this news organization. “There is a genuine concern and fear of harming patients and a desire to provide the best care for the patient’s well-being.”
In general, fear-of-harm and fear-of-suit scores decreased as providers gained experience. Those with less than 5 years of experience reported the highest levels of both.
“Although our data do not specifically provide reasons why age may impact [fear] levels, it is possible that with more practice experience ... providers have a better sense of the likelihood of patient harm and malpractice and how to manage such outcomes should they happen,” says Dr. Isbell. She noted that a longitudinal study is necessary to confirm this hypothesis.
One exception was female APCs, whose fear-of-harm scores remained relatively steady across all experience levels. Among male APCs, fear of causing patient harm decreased among those with 5-14 years of experience but increased slightly at 14-44 years of experience.
While previous research typically focused on fear of malpractice as a significant driver of defensive medicine, such as testing excessively, this study examined providers’ fear of harming patients because of a medical error.
The findings suggest “that fear of harm should be considered with, and may be more consequential than, fear of suit in medical decision-making,” the authors note.
“[F]ear can motivate people to engage in more careful and thorough information processing, which can drive behaviors in systematic ways,” says Dr. Isbell. “It is possible that one’s fear of harming a patient is triggering a high level of vigilance, reflected in the practice of defensive medicine across different types of patients – some of whom may be better off with less testing and referrals.”
Rade B. Vukmir, MD, JD, FACEP, an emergency medicine physician and spokesman for the American College of Emergency Physicians, says defensive medicine is common in the specialty and that it occurs 20%-40% of the time.
“Early in practice, the proverbial worst sin is missing a diagnosis, so that’s where the overtesting mentality comes from,” he says. In addition, “there are cities where you can’t drive a mile without seeing a half dozen legal advertisements. That imposes a cost burden on the system, [adding] roughly 20% to the cost of overall care.”
Emergency medicine providers attempt to minimize testing, but between their role as “America’s safety net” and the difficult circumstances they often face when treating patients, it takes a while to strike a balance, Dr. Vukmir acknowledges.
“There’s a training correlation, which showed up [in this study]; as people got further advanced in training, they felt more comfortable and felt the need to do it less,” says Dr. Vukmir.
The study was funded by a grant from the Agency for Healthcare Research and Quality. Dr. Isbell reports no conflicts of interest. Dr. Vukmir has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a study published in JAMA Network Open.
The cross-sectional study was conducted by researchers from Soroka University Medical Center, Israel; the University of Massachusetts, Worcester; Beth Israel Deaconess Medical Center; Harvard Medical School, Boston; and the University of Massachusetts, Amherst.
Online survey responses were collected from 1,222 emergency department attending physicians and advanced practice clinicians (APCs) in acute care hospitals throughout Massachusetts from January to September 2020.
Participants were asked to rank their level of agreement – from “strongly disagree” to “strongly agree” – with two statements: “In my day-to-day practice, I am fearful of making a mistake which results in [1] harm to the patient” (fear of harm) and [2] “being sued” (fear of suit).
The average age of the participants was about 44 years; 54.2% were men, 45.1% were women, and 0.7% were of other gender. Approximately 70% of responses were from MDs or DOs, and the remainder were from nurse practitioners and physician assistants. Participants had between 5 and 19 years of experience (median, 10 years).
The study found that the mean score was greater with regard to fear of harm than to fear of suit, regardless of clinician type, experience, or sex and whether the survey was completed before or after the start of the COVID-19 pandemic. There was no significant difference in mean scores regarding fear of suit before the pandemic and after it.
“Our data show a significantly greater fear of harming a patient than a fear of a malpractice suit,” Linda Isbell, PhD, professor of psychology at the University of Massachusetts, Amherst, who is one of the study’s authors, told this news organization. “There is a genuine concern and fear of harming patients and a desire to provide the best care for the patient’s well-being.”
In general, fear-of-harm and fear-of-suit scores decreased as providers gained experience. Those with less than 5 years of experience reported the highest levels of both.
“Although our data do not specifically provide reasons why age may impact [fear] levels, it is possible that with more practice experience ... providers have a better sense of the likelihood of patient harm and malpractice and how to manage such outcomes should they happen,” says Dr. Isbell. She noted that a longitudinal study is necessary to confirm this hypothesis.
One exception was female APCs, whose fear-of-harm scores remained relatively steady across all experience levels. Among male APCs, fear of causing patient harm decreased among those with 5-14 years of experience but increased slightly at 14-44 years of experience.
While previous research typically focused on fear of malpractice as a significant driver of defensive medicine, such as testing excessively, this study examined providers’ fear of harming patients because of a medical error.
The findings suggest “that fear of harm should be considered with, and may be more consequential than, fear of suit in medical decision-making,” the authors note.
“[F]ear can motivate people to engage in more careful and thorough information processing, which can drive behaviors in systematic ways,” says Dr. Isbell. “It is possible that one’s fear of harming a patient is triggering a high level of vigilance, reflected in the practice of defensive medicine across different types of patients – some of whom may be better off with less testing and referrals.”
Rade B. Vukmir, MD, JD, FACEP, an emergency medicine physician and spokesman for the American College of Emergency Physicians, says defensive medicine is common in the specialty and that it occurs 20%-40% of the time.
“Early in practice, the proverbial worst sin is missing a diagnosis, so that’s where the overtesting mentality comes from,” he says. In addition, “there are cities where you can’t drive a mile without seeing a half dozen legal advertisements. That imposes a cost burden on the system, [adding] roughly 20% to the cost of overall care.”
Emergency medicine providers attempt to minimize testing, but between their role as “America’s safety net” and the difficult circumstances they often face when treating patients, it takes a while to strike a balance, Dr. Vukmir acknowledges.
“There’s a training correlation, which showed up [in this study]; as people got further advanced in training, they felt more comfortable and felt the need to do it less,” says Dr. Vukmir.
The study was funded by a grant from the Agency for Healthcare Research and Quality. Dr. Isbell reports no conflicts of interest. Dr. Vukmir has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a study published in JAMA Network Open.
The cross-sectional study was conducted by researchers from Soroka University Medical Center, Israel; the University of Massachusetts, Worcester; Beth Israel Deaconess Medical Center; Harvard Medical School, Boston; and the University of Massachusetts, Amherst.
Online survey responses were collected from 1,222 emergency department attending physicians and advanced practice clinicians (APCs) in acute care hospitals throughout Massachusetts from January to September 2020.
Participants were asked to rank their level of agreement – from “strongly disagree” to “strongly agree” – with two statements: “In my day-to-day practice, I am fearful of making a mistake which results in [1] harm to the patient” (fear of harm) and [2] “being sued” (fear of suit).
The average age of the participants was about 44 years; 54.2% were men, 45.1% were women, and 0.7% were of other gender. Approximately 70% of responses were from MDs or DOs, and the remainder were from nurse practitioners and physician assistants. Participants had between 5 and 19 years of experience (median, 10 years).
The study found that the mean score was greater with regard to fear of harm than to fear of suit, regardless of clinician type, experience, or sex and whether the survey was completed before or after the start of the COVID-19 pandemic. There was no significant difference in mean scores regarding fear of suit before the pandemic and after it.
“Our data show a significantly greater fear of harming a patient than a fear of a malpractice suit,” Linda Isbell, PhD, professor of psychology at the University of Massachusetts, Amherst, who is one of the study’s authors, told this news organization. “There is a genuine concern and fear of harming patients and a desire to provide the best care for the patient’s well-being.”
In general, fear-of-harm and fear-of-suit scores decreased as providers gained experience. Those with less than 5 years of experience reported the highest levels of both.
“Although our data do not specifically provide reasons why age may impact [fear] levels, it is possible that with more practice experience ... providers have a better sense of the likelihood of patient harm and malpractice and how to manage such outcomes should they happen,” says Dr. Isbell. She noted that a longitudinal study is necessary to confirm this hypothesis.
One exception was female APCs, whose fear-of-harm scores remained relatively steady across all experience levels. Among male APCs, fear of causing patient harm decreased among those with 5-14 years of experience but increased slightly at 14-44 years of experience.
While previous research typically focused on fear of malpractice as a significant driver of defensive medicine, such as testing excessively, this study examined providers’ fear of harming patients because of a medical error.
The findings suggest “that fear of harm should be considered with, and may be more consequential than, fear of suit in medical decision-making,” the authors note.
“[F]ear can motivate people to engage in more careful and thorough information processing, which can drive behaviors in systematic ways,” says Dr. Isbell. “It is possible that one’s fear of harming a patient is triggering a high level of vigilance, reflected in the practice of defensive medicine across different types of patients – some of whom may be better off with less testing and referrals.”
Rade B. Vukmir, MD, JD, FACEP, an emergency medicine physician and spokesman for the American College of Emergency Physicians, says defensive medicine is common in the specialty and that it occurs 20%-40% of the time.
“Early in practice, the proverbial worst sin is missing a diagnosis, so that’s where the overtesting mentality comes from,” he says. In addition, “there are cities where you can’t drive a mile without seeing a half dozen legal advertisements. That imposes a cost burden on the system, [adding] roughly 20% to the cost of overall care.”
Emergency medicine providers attempt to minimize testing, but between their role as “America’s safety net” and the difficult circumstances they often face when treating patients, it takes a while to strike a balance, Dr. Vukmir acknowledges.
“There’s a training correlation, which showed up [in this study]; as people got further advanced in training, they felt more comfortable and felt the need to do it less,” says Dr. Vukmir.
The study was funded by a grant from the Agency for Healthcare Research and Quality. Dr. Isbell reports no conflicts of interest. Dr. Vukmir has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Is there a doctor on the plane? Tips for providing in-flight assistance
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.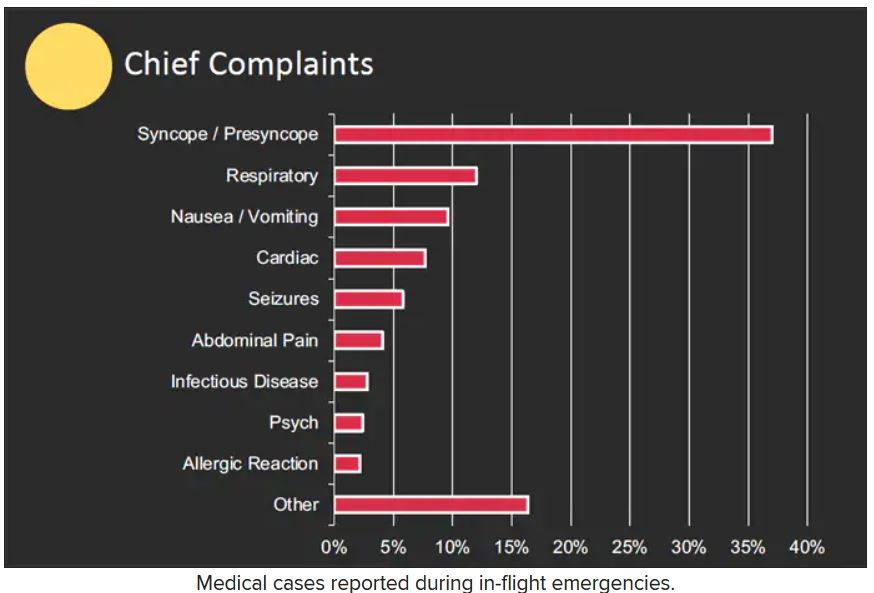
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
In most cases, passengers on an airline flight are representative of the general population, which means that anyone could have an emergency at any time.
as determined on the basis of in-flight medical emergencies that resulted in calls to a physician-directed medical communications center, said Amy Faith Ho, MD, MPH of Integrative Emergency Services, Dallas–Fort Worth, in a presentation at the annual meeting of the American College of Emergency Physicians.
The study authors reviewed records of 11,920 in-flight medical emergencies between Jan. 1, 2008, and Oct. 31, 2010. The data showed that physician passengers provided medical assistance in nearly half of in-flight emergencies (48.1%) and that flights were diverted because of the emergency in 7.3% of cases.
The majority of the in-flight emergencies involved syncope or presyncope (37.4% of cases), followed by respiratory symptoms (12.1%) and nausea or vomiting (9.5%), according to the study.
When a physician is faced with an in-flight emergency, the medical team includes the physician himself, medical ground control, and the flight attendants, said Dr. Ho. Requirements may vary among airlines, but all flight attendants will be trained in cardiopulmonary resuscitation (CPR) or basic life support, as well as use of automated external defibrillators (AEDs).
Physician call centers (medical ground control) can provide additional assistance remotely, she said.
The in-flight medical bag
Tools in a physician’s in-flight toolbox start with the first-aid kit. Airplanes also have an emergency medical kit (EMK), an oxygen tank, and an AED.
The minimum EMK contents are mandated by the Federal Aviation Administration, said Dr. Ho. The standard equipment includes a stethoscope, a sphygmomanometer, and three sizes of oropharyngeal airways. Other items include self-inflating manual resuscitation devices and CPR masks in thee sizes, alcohol sponges, gloves, adhesive tape, scissors, a tourniquet, as well as saline solution, needles, syringes, and an intravenous administration set consisting of tubing and two Y connectors.
An EMK also should contain the following medications: nonnarcotic analgesic tablets, antihistamine tablets, an injectable antihistamine, atropine, aspirin tablets, a bronchodilator, and epinephrine (both 1:1000; 1 injectable cc and 1:10,000; two injectable cc). Nitroglycerin tablets and 5 cc of 20 mg/mL injectable cardiac lidocaine are part of the mandated kit as well, according to Dr. Ho.
Some airlines carry additional supplies on all their flights, said Dr. Ho. Notably, American Airlines and British Airways carry EpiPens for adults and children, as well as opioid reversal medication (naloxone) and glucose for managing low blood sugar. American Airlines and Delta stock antiemetics, and Delta also carries naloxone. British Airways is unique in stocking additional cardiac medications, both oral and injectable.
How to handle an in-flight emergency
Physicians should always carry a copy of their medical license when traveling for documentation by the airline if they assist in a medical emergency during a flight, Dr. Ho emphasized. “Staff” personnel should be used. These include the flight attendants, medical ground control, and other passengers who might have useful skills, such as nursing, the ability to perform CPR, or therapy/counseling to calm a frightened patient. If needed, “crowdsource additional supplies from passengers,” such as a glucometer or pulse oximeter.
Legal lessons
Physicians are not obligated to assist during an in-flight medical emergency, said Dr. Ho. Legal jurisdiction can vary. In the United States, a bystander who assists in an emergency is generally protected by Good Samaritan laws; for international airlines, the laws may vary; those where the airline is based usually apply.
The Aviation Medical Assistance Act, passed in 1998, protects individuals from being sued for negligence while providing medical assistance, “unless the individual, while rendering such assistance, is guilty of gross negligence of willful misconduct,” Dr. Ho noted. The Aviation Medical Assistance Act also protects the airline itself “if the carrier in good faith believes that the passenger is a medically qualified individual.”
Dr. Ho disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACEP 2022
Children and COVID: Weekly cases continue to hold fairly steady
The incidence of new COVID-19 cases in children seems to have stabilized as the national count remained under 30,000 for the fifth consecutive week, but hospitalization data may indicate some possible turbulence.
Just over 28,000 pediatric cases were reported during the week of Nov. 4-10, a drop of 5.4% from the previous week, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report involving data from state and territorial health departments, several of which are no longer updating their websites.
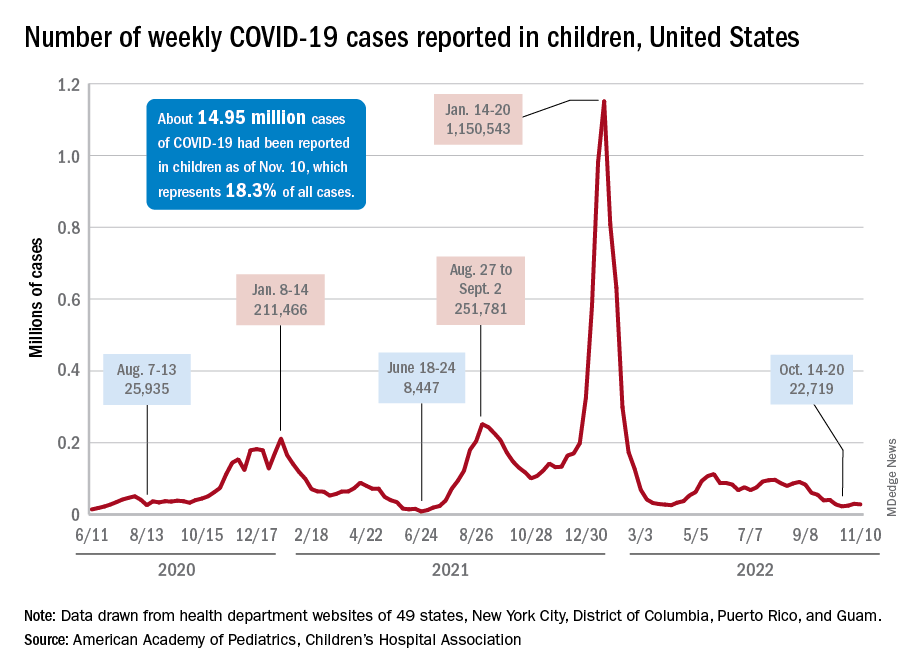
The stability in weekly cases, however, comes in contrast to a very recent and considerable increase in new hospital admissions of children aged 0-17 years with confirmed COVID-19. That rate, which was 0.18 hospitalizations per 100,000 population on Nov. 7 and 0.19 per 100,000 on Nov. 8 and 9, jumped all the way to 0.34 on Nov. 10 and 0.48 on Nov. 11, according to data from the Centers for Disease Control and Prevention. That is the highest rate since the closing days of the Omicron surge in February.
The rate for Nov. 12, the most recent one available, was down slightly to 0.47 admissions per 100,000. There doesn’t seem to be any evidence in the CDC’s data of a similar sudden increase in new hospitalizations among any other age group, and no age group, including children, shows any sign of a recent increase in emergency department visits with diagnosed COVID. (The CDC has not yet responded to our inquiry about this development.)
The two most recent 7-day averages for new admissions in children aged 0-17 show a small increase, but they cover the periods of Oct. 15 to Oct. 31, when there were 126 admissions per day, and Nov. 1 to Nov. 7, when the average went up to 133 per day, the CDC said on its COVID Data Tracker.
The CDC does not publish a weekly count of new COVID cases, but its latest data on the rate of incident cases seem to agree with the AAP/CHA figures: A gradual decline in all age groups, including children, since the beginning of September.
Vaccinations, on the other hand, bucked their recent trend and increased in the last week. About 43,000 children under age 5 years received their initial dose of COVID vaccine during Nov. 3-9, compared with 30,000 and 33,000 the 2 previous weeks, while 5- to 11-year-olds hit their highest weekly mark (31,000) since late August and 12- to 17-year-olds had their biggest week (27,000) since mid-August, the AAP reported based on CDC data.
The incidence of new COVID-19 cases in children seems to have stabilized as the national count remained under 30,000 for the fifth consecutive week, but hospitalization data may indicate some possible turbulence.
Just over 28,000 pediatric cases were reported during the week of Nov. 4-10, a drop of 5.4% from the previous week, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report involving data from state and territorial health departments, several of which are no longer updating their websites.

The stability in weekly cases, however, comes in contrast to a very recent and considerable increase in new hospital admissions of children aged 0-17 years with confirmed COVID-19. That rate, which was 0.18 hospitalizations per 100,000 population on Nov. 7 and 0.19 per 100,000 on Nov. 8 and 9, jumped all the way to 0.34 on Nov. 10 and 0.48 on Nov. 11, according to data from the Centers for Disease Control and Prevention. That is the highest rate since the closing days of the Omicron surge in February.
The rate for Nov. 12, the most recent one available, was down slightly to 0.47 admissions per 100,000. There doesn’t seem to be any evidence in the CDC’s data of a similar sudden increase in new hospitalizations among any other age group, and no age group, including children, shows any sign of a recent increase in emergency department visits with diagnosed COVID. (The CDC has not yet responded to our inquiry about this development.)
The two most recent 7-day averages for new admissions in children aged 0-17 show a small increase, but they cover the periods of Oct. 15 to Oct. 31, when there were 126 admissions per day, and Nov. 1 to Nov. 7, when the average went up to 133 per day, the CDC said on its COVID Data Tracker.
The CDC does not publish a weekly count of new COVID cases, but its latest data on the rate of incident cases seem to agree with the AAP/CHA figures: A gradual decline in all age groups, including children, since the beginning of September.
Vaccinations, on the other hand, bucked their recent trend and increased in the last week. About 43,000 children under age 5 years received their initial dose of COVID vaccine during Nov. 3-9, compared with 30,000 and 33,000 the 2 previous weeks, while 5- to 11-year-olds hit their highest weekly mark (31,000) since late August and 12- to 17-year-olds had their biggest week (27,000) since mid-August, the AAP reported based on CDC data.
The incidence of new COVID-19 cases in children seems to have stabilized as the national count remained under 30,000 for the fifth consecutive week, but hospitalization data may indicate some possible turbulence.
Just over 28,000 pediatric cases were reported during the week of Nov. 4-10, a drop of 5.4% from the previous week, the American Academy of Pediatrics and the Children’s Hospital Association said in their weekly COVID-19 report involving data from state and territorial health departments, several of which are no longer updating their websites.

The stability in weekly cases, however, comes in contrast to a very recent and considerable increase in new hospital admissions of children aged 0-17 years with confirmed COVID-19. That rate, which was 0.18 hospitalizations per 100,000 population on Nov. 7 and 0.19 per 100,000 on Nov. 8 and 9, jumped all the way to 0.34 on Nov. 10 and 0.48 on Nov. 11, according to data from the Centers for Disease Control and Prevention. That is the highest rate since the closing days of the Omicron surge in February.
The rate for Nov. 12, the most recent one available, was down slightly to 0.47 admissions per 100,000. There doesn’t seem to be any evidence in the CDC’s data of a similar sudden increase in new hospitalizations among any other age group, and no age group, including children, shows any sign of a recent increase in emergency department visits with diagnosed COVID. (The CDC has not yet responded to our inquiry about this development.)
The two most recent 7-day averages for new admissions in children aged 0-17 show a small increase, but they cover the periods of Oct. 15 to Oct. 31, when there were 126 admissions per day, and Nov. 1 to Nov. 7, when the average went up to 133 per day, the CDC said on its COVID Data Tracker.
The CDC does not publish a weekly count of new COVID cases, but its latest data on the rate of incident cases seem to agree with the AAP/CHA figures: A gradual decline in all age groups, including children, since the beginning of September.
Vaccinations, on the other hand, bucked their recent trend and increased in the last week. About 43,000 children under age 5 years received their initial dose of COVID vaccine during Nov. 3-9, compared with 30,000 and 33,000 the 2 previous weeks, while 5- to 11-year-olds hit their highest weekly mark (31,000) since late August and 12- to 17-year-olds had their biggest week (27,000) since mid-August, the AAP reported based on CDC data.
Infant anaphylaxis: Study characterizes symptoms, treatment
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
LOUISVILLE, KY. – , research findings indicate.
Given that early administration of epinephrine can be potentially lifesaving for infants with anaphylaxis, the study highlighted the real-world successes in increased uptake of treatment in this vulnerable patient population.
Most infants in the study who presented to the ED and received epinephrine were able to be discharged home after just a few hours, with only 1 out of 10 requiring hospitalization.
The study also reported that most symptoms were in the skin/mucosal, gastrointestinal, respiratory, and cardiovascular (CV) systems, providing improved characterization of anaphylaxis symptoms in the infant population.
Nearly “all episodes were triggered by food – especially egg, peanut, milk, and cashew,” commented Colleen Shannon, MD, a pediatrician at Children’s Hospital of Philadelphia, who presented the research findings at the annual meeting of the American College of Allergy, Asthma, and Immunology.
Dr. Shannon noted that despite previous research demonstrating age-based differences in the presentation of anaphylaxis, the symptomatology of anaphylaxis in infants has not been robustly characterized. Better characterization of anaphylaxis in infants with allergies may help ensure earlier and more accurate diagnosis and management, she said.
For the study, the researchers performed a retrospective chart review of 169 patients between 0 and 24 months of age (mean age, 1.0 years) who presented to the emergency department of a pediatric tertiary referral center between 2019 and 2022.
All patients in the study met diagnostic criteria for anaphylaxis. The investigators used the medical records of patients to evaluate for demographics, as well as presenting symptoms and treatment.
More than half (56.2%) of infants in the study were 12 months of age or younger, and 64.5% were male.
Nearly all (96.5%) anaphylaxis episodes presenting to the ED were triggered by food. The most common foods triggering these episodes were egg (26.6%), peanut (25.4%), milk (13.6%), and cashew (10.1%).
Most symptoms involved the skin/mucosal (97.6%) and GI (74.6%) systems, followed by respiratory (56.8%) and CV (34.3%) systems. Isolated tachycardia was recorded in 84.5% of patients with CV-related symptoms.
Epinephrine was administered to 86.4% of infants who presented to the ED with anaphylaxis. Nearly a third (30.1%) of these infants received epinephrine before arriving to the ED, and 9.5% required more than 1 dose.
The researchers also found that 10.1% of patients required hospital admission, but none had symptoms severe enough to require intensive care.
Jennifer Hoffmann, MD, an emergency medicine physician at the Lurie Children’s Hospital of Chicago, told this news organization that while characterizing anaphylaxis symptoms is relevant for clinicians, it also remains vitally important “to teach parents of infants how to recognize the signs of anaphylaxis, particularly as they begin to introduce new foods,” to ensure timely treatment.
She added that since most infants in the study improved after a single dose of epinephrine, most infants presenting to the ED with anaphylaxis can therefore be safely discharged home after only a brief period of observation. “That is, age alone should not be a reason for admission,” explained Dr. Hoffmann, who wasn’t involved in the research study.
The study was independently supported. Dr. Shannon and Dr. Hoffmann report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM ACAAI
ED visits for kids with suicidal thoughts increasing: Study
A new study sheds light on the escalating youth suicide crisis, revealing that emergency room visits for suicidal thoughts among kids and teens steeply increased even before the start of the COVID-19 pandemic.
Emergency room visits for “suicidal ideation” (or suicidal thoughts) among 5- to 19-year-olds increased 59% from 2016 to 2021, and hospitalizations rose 57% from fall 2019 to the fall of 2020, according to the study published in Pediatrics.
“A lot of people have talked about mental health problems in youth during the pandemic, but it was happening before the pandemic,” said author Audrey Brewer, MD, MPH, in a news release from the Ann and Robert H. Lurie Children’s Hospital of Chicago. “This has been an issue for so long, and it’s getting worse.”
Researchers looked at data for 81,105 emergency room visits across 205 Illinois hospitals from 2016 to 2021 for kids between the ages of 5 and 19.
The researchers found “there was a very sharp spike in fall 2019, followed by a similar spike during the pandemic fall of 2020, with the highest number of monthly visits during October 2020,” the authors said. “Youth aged 14-17 years had the highest frequency of [suicidal ideation emergency room] monthly visits, with visits in this group greater than the other age groups combined.”
Last year, the Centers for Disease Control and Prevention announced that suicide is the second leading cause of death among 10- to 19-year-olds.
The new research is being called a benchmark because it evaluates emergency room data for suicidal thoughts – a critical point of care for serving youths’ mental health needs. The data showed that providers were increasingly likely to list suicidal thoughts as the main diagnosis.
“Suicidal ideation can be thought about as two types: actively thinking about suicide or having thoughts, but not having a plan,” Dr. Brewer said in the news release. “That could be the difference in why someone might get admitted to the hospital.”
The researchers hypothesize that care in 2019 (when the initial spike occurred) was delayed in the early days of the pandemic, and that delay possibly contributed to the increase in providers identifying suicidal ideation as the main diagnosis.
“The early pandemic period coincided with constrained access to pediatric mental health services through schools, pediatric primary care homes, and mental health clinics for many children and their families,” the authors wrote. “The proportion of child mental health visits increased relative to other types as patients avoided ED visits during the early wave of the COVID-19 pandemic. Thus, the increase in hospitalizations during fall 2020 may reflect patients’ deferring care until symptoms became even more severe.”
Other health care scholars agreed the study spurred questions about whether the pandemic was truly the source of the crisis.
“Was it the pandemic that exacerbated the increase or is this a growing trend?” wrote Lisa M. Horowitz, PhD, MPH, and Jeffrey A. Bridge, PhD, in a commentary published along with the study. “These rising rates underscore the worsening mental health crisis for youth, as noted by the 2022 Surgeon General report and several youth mental health organizations.”
A version of this article first appeared on WebMD.com.
A new study sheds light on the escalating youth suicide crisis, revealing that emergency room visits for suicidal thoughts among kids and teens steeply increased even before the start of the COVID-19 pandemic.
Emergency room visits for “suicidal ideation” (or suicidal thoughts) among 5- to 19-year-olds increased 59% from 2016 to 2021, and hospitalizations rose 57% from fall 2019 to the fall of 2020, according to the study published in Pediatrics.
“A lot of people have talked about mental health problems in youth during the pandemic, but it was happening before the pandemic,” said author Audrey Brewer, MD, MPH, in a news release from the Ann and Robert H. Lurie Children’s Hospital of Chicago. “This has been an issue for so long, and it’s getting worse.”
Researchers looked at data for 81,105 emergency room visits across 205 Illinois hospitals from 2016 to 2021 for kids between the ages of 5 and 19.
The researchers found “there was a very sharp spike in fall 2019, followed by a similar spike during the pandemic fall of 2020, with the highest number of monthly visits during October 2020,” the authors said. “Youth aged 14-17 years had the highest frequency of [suicidal ideation emergency room] monthly visits, with visits in this group greater than the other age groups combined.”
Last year, the Centers for Disease Control and Prevention announced that suicide is the second leading cause of death among 10- to 19-year-olds.
The new research is being called a benchmark because it evaluates emergency room data for suicidal thoughts – a critical point of care for serving youths’ mental health needs. The data showed that providers were increasingly likely to list suicidal thoughts as the main diagnosis.
“Suicidal ideation can be thought about as two types: actively thinking about suicide or having thoughts, but not having a plan,” Dr. Brewer said in the news release. “That could be the difference in why someone might get admitted to the hospital.”
The researchers hypothesize that care in 2019 (when the initial spike occurred) was delayed in the early days of the pandemic, and that delay possibly contributed to the increase in providers identifying suicidal ideation as the main diagnosis.
“The early pandemic period coincided with constrained access to pediatric mental health services through schools, pediatric primary care homes, and mental health clinics for many children and their families,” the authors wrote. “The proportion of child mental health visits increased relative to other types as patients avoided ED visits during the early wave of the COVID-19 pandemic. Thus, the increase in hospitalizations during fall 2020 may reflect patients’ deferring care until symptoms became even more severe.”
Other health care scholars agreed the study spurred questions about whether the pandemic was truly the source of the crisis.
“Was it the pandemic that exacerbated the increase or is this a growing trend?” wrote Lisa M. Horowitz, PhD, MPH, and Jeffrey A. Bridge, PhD, in a commentary published along with the study. “These rising rates underscore the worsening mental health crisis for youth, as noted by the 2022 Surgeon General report and several youth mental health organizations.”
A version of this article first appeared on WebMD.com.
A new study sheds light on the escalating youth suicide crisis, revealing that emergency room visits for suicidal thoughts among kids and teens steeply increased even before the start of the COVID-19 pandemic.
Emergency room visits for “suicidal ideation” (or suicidal thoughts) among 5- to 19-year-olds increased 59% from 2016 to 2021, and hospitalizations rose 57% from fall 2019 to the fall of 2020, according to the study published in Pediatrics.
“A lot of people have talked about mental health problems in youth during the pandemic, but it was happening before the pandemic,” said author Audrey Brewer, MD, MPH, in a news release from the Ann and Robert H. Lurie Children’s Hospital of Chicago. “This has been an issue for so long, and it’s getting worse.”
Researchers looked at data for 81,105 emergency room visits across 205 Illinois hospitals from 2016 to 2021 for kids between the ages of 5 and 19.
The researchers found “there was a very sharp spike in fall 2019, followed by a similar spike during the pandemic fall of 2020, with the highest number of monthly visits during October 2020,” the authors said. “Youth aged 14-17 years had the highest frequency of [suicidal ideation emergency room] monthly visits, with visits in this group greater than the other age groups combined.”
Last year, the Centers for Disease Control and Prevention announced that suicide is the second leading cause of death among 10- to 19-year-olds.
The new research is being called a benchmark because it evaluates emergency room data for suicidal thoughts – a critical point of care for serving youths’ mental health needs. The data showed that providers were increasingly likely to list suicidal thoughts as the main diagnosis.
“Suicidal ideation can be thought about as two types: actively thinking about suicide or having thoughts, but not having a plan,” Dr. Brewer said in the news release. “That could be the difference in why someone might get admitted to the hospital.”
The researchers hypothesize that care in 2019 (when the initial spike occurred) was delayed in the early days of the pandemic, and that delay possibly contributed to the increase in providers identifying suicidal ideation as the main diagnosis.
“The early pandemic period coincided with constrained access to pediatric mental health services through schools, pediatric primary care homes, and mental health clinics for many children and their families,” the authors wrote. “The proportion of child mental health visits increased relative to other types as patients avoided ED visits during the early wave of the COVID-19 pandemic. Thus, the increase in hospitalizations during fall 2020 may reflect patients’ deferring care until symptoms became even more severe.”
Other health care scholars agreed the study spurred questions about whether the pandemic was truly the source of the crisis.
“Was it the pandemic that exacerbated the increase or is this a growing trend?” wrote Lisa M. Horowitz, PhD, MPH, and Jeffrey A. Bridge, PhD, in a commentary published along with the study. “These rising rates underscore the worsening mental health crisis for youth, as noted by the 2022 Surgeon General report and several youth mental health organizations.”
A version of this article first appeared on WebMD.com.
FROM PEDIATRICS
Statins boost glycemia slightly, but CVD benefits prevail
CHICAGO – A new, expanded meta-analysis confirmed the long-known effect that statin treatment has on raising blood glucose levels and causing incident diabetes, but it also documented that these effects are small and any risk they pose to statin users is dwarfed by the cholesterol-lowering effect of statins and their ability to reduce risk for atherosclerotic cardiovascular disease (ASCVD).
This meta-analysis of 23 trials with a total of more than 150,000 participants showed that statin therapy significantly increased the risk for new-onset diabetes and worsening glycemia, driven by a “very small but generalized increase in glucose,” with a greater effect from high-intensity statin regimens and a similar but somewhat more muted effect from low- and moderate-intensity statin treatment, David Preiss, MBChB, PhD, reported at the American Heart Association scientific sessions.
Dr. Preiss also stressed that despite this, “the cardiovascular benefits of statin therapy remain substantial and profound” in people regardless of whether they have diabetes, prediabetes, or normoglycemia when they start statin treatment, noting that the impact of even high-intensity statin treatment is “absolutely tiny” increases in hemoglobin A1c and blood glucose.
“This does not detract from the substantial benefit of statin treatment,” declared Dr. Preiss, a metabolic medicine specialist and endocrinologist at Oxford (England) University.
Small glycemia increases ‘nudge’ some into diabetes
The data Dr. Preiss reported showed that high-intensity statin treatment (atorvastatin at a daily dose of at least 40 mg, or rosuvastatin at a daily dose of at least 20 mg) led to an average increase in A1c levels of 0.08 percentage points among people without diabetes when their treatment began and 0.24 percentage points among people already diagnosed with diabetes. Blood glucose levels rose by an average of 0.04 mmol/L (less than 1 mg/d) in those without diabetes, and by an average 0.22 mmol/L (about 4 mg/dL) in those with diabetes. People who received low- or moderate-intensity statin regimens had significant but smaller increases.
“We’re not talking about people going from no diabetes to frank diabetes. We’re talking about [statins] nudging a very small number of people across a diabetes threshold,” an A1c of 6.5% that is set somewhat arbitrarily based on an increased risk for developing retinopathy, Dr. Preiss said. ”A person just needs to lose a [daily] can of Coke’s worth of weight to eliminate any apparent diabetes risk,” he noted.
Benefit outweighs risks by three- to sevenfold
Dr. Preiss presented two other examples of what his findings showed to illustrate the relatively small risk posed by statin therapy compared with its potential benefits. Treating 10,000 people for 5 years with a high-intensity statin regimen in those with established ASCVD (secondary prevention) would result in an increment of 150 extra people developing diabetes because of the hyperglycemic effect of statins, compared with an expected prevention of 1,000 ASCVD events. Among 10,000 people at high ASCVD risk and taking a high-intensity statin regimen for primary prevention 5 years of treatment would result in roughly 130 extra cases of incident diabetes while preventing about 500 ASCVD events.
In addition, applying the new risk estimates to the people included in the UK Biobank database, whose median A1c is 5.5%, showed that a high-intensity statin regimen could be expected to raise the prevalence of those with an A1c of 6.5% or greater from 4.5% to 5.7%.
Several preventive cardiologists who heard the report and were not involved with the analysis agreed with Dr. Preiss that the benefits of statin treatment substantially offset this confirmed hyperglycemic effect.
Risk ‘more than counterbalanced by benefit’
“He clearly showed that the small hyperglycemia risk posed by statin use is more than counterbalanced by its benefit for reducing ASCVD events,” commented Neil J. Stone, MD, a cardiologist and professor of medicine at Northwestern University, Chicago. “I agree that, for those with prediabetes who are on the road to diabetes with or without a statin, the small increase in glucose with a statin should not dissuade statin usage because the benefit is so large. Rather, it should focus efforts to improve diet, increase physical activity, and keep weight controlled.”
Dr. Stone also noted in an interview that in the JUPITER trial, which examined the effects of a daily 20-mg dose of rosuvastatin (Crestor), a high-intensity regimen, study participants with diabetes risk factors who were assigned to rosuvastatin had an onset of diabetes that was earlier than people assigned to placebo by only about 5.4 weeks, yet this group had evidence of significant benefit.
“I agree with Dr. Preiss that the benefits of statins in reducing heart attack, stroke, and cardiovascular death far outweigh their modest effects on glycemia,” commented Brendan M. Everett, MD, a cardiologist and preventive medicine specialist at Brigham and Women’s Hospital in Boston. “This is particularly true for those with preexisting prediabetes or diabetes, who have an elevated risk of atherosclerotic events and thus stand to derive more significant benefit from statins. The benefits of lowering LDL cholesterol with a statin for preventing seriously morbid, and potentially fatal, cardiovascular events far outweigh the extremely modest, or even negligible, increases in the risk of diabetes that could be seen with the extremely small increases in A1c,” Dr. Everett said in an interview.
The new findings “reaffirm that there is a increased risk [from statins] but the most important point is that it is a very, very tiny difference in A1c,” commented Marc S. Sabatine, MD, a cardiologist and professor at Harvard Medical School, Boston. “These data have been known for quite some time, but this analysis was done in a more rigorous way.” The finding of “a small increase in risk for diabetes is really because diabetes has a biochemical threshold and statin treatment nudges some people a little past a line that is semi-arbitrary. It’s important to be cognizant of this, but it in no way dissuades me from treating patients aggressively with statins to reduce their ASCVD risk. I would monitor their A1c levels, and if they go higher and can’t be controlled with lifestyle we have plenty of medications that can control it,” he said in an interview.
No difference by statin type
The meta-analysis used data from 13 placebo-controlled statin trials that together involved 123,940 participants and had an average 4.3 years of follow-up, and four trials that compared one statin with another and collectively involved 30,734 participants with an average 4.9 years of follow-up.
The analyses showed that high-intensity statin treatment increased the rate of incident diabetes by a significant 36% relative to controls and increased the rate of worsening glycemia by a significant 24% compared with controls. Low- or moderate-intensity statin regimens increased incident diabetes by a significant 10% and raised the incidence of worsening glycemia by a significant 10% compared with controls, Dr. Preiss reported.
These effects did not significantly differ by type of statin (the study included people treated with atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin), nor across a variety of subgroups based on age, sex, race, body mass index, diabetes risk, renal function, cholesterol levels, or cardiovascular disease. The effect was also consistent regardless of the duration of treatment.
Dr. Preiss also downplayed the magnitude of the apparent difference in risk posed by high-intensity and less intense statin regimens. “I suspect the apparent heterogeneity is true, but not quite as big as what we see,” he said.
The mechanisms by which statins have this effect remain unclear, but evidence suggests that it may be a direct effect of the main action of statins, inhibition of the HMG-CoA reductase enzyme.
The study received no commercial funding. Dr. Preiss and Dr. Stone had no disclosures. Dr. Everett has been a consultant to Eli Lilly, Gilead, Ipsen, Janssen, and Provention. Dr. Sabatine has been a consultant to Althera, Amgen, Anthos Therapeutics, AstraZeneca, Beren Therapeutics, Bristol-Myers Squibb, DalCor, Dr Reddy’s Laboratories, Fibrogen, Intarcia, Merck, Moderna, Novo Nordisk, and Silence Therapeutics.
CHICAGO – A new, expanded meta-analysis confirmed the long-known effect that statin treatment has on raising blood glucose levels and causing incident diabetes, but it also documented that these effects are small and any risk they pose to statin users is dwarfed by the cholesterol-lowering effect of statins and their ability to reduce risk for atherosclerotic cardiovascular disease (ASCVD).
This meta-analysis of 23 trials with a total of more than 150,000 participants showed that statin therapy significantly increased the risk for new-onset diabetes and worsening glycemia, driven by a “very small but generalized increase in glucose,” with a greater effect from high-intensity statin regimens and a similar but somewhat more muted effect from low- and moderate-intensity statin treatment, David Preiss, MBChB, PhD, reported at the American Heart Association scientific sessions.
Dr. Preiss also stressed that despite this, “the cardiovascular benefits of statin therapy remain substantial and profound” in people regardless of whether they have diabetes, prediabetes, or normoglycemia when they start statin treatment, noting that the impact of even high-intensity statin treatment is “absolutely tiny” increases in hemoglobin A1c and blood glucose.
“This does not detract from the substantial benefit of statin treatment,” declared Dr. Preiss, a metabolic medicine specialist and endocrinologist at Oxford (England) University.
Small glycemia increases ‘nudge’ some into diabetes
The data Dr. Preiss reported showed that high-intensity statin treatment (atorvastatin at a daily dose of at least 40 mg, or rosuvastatin at a daily dose of at least 20 mg) led to an average increase in A1c levels of 0.08 percentage points among people without diabetes when their treatment began and 0.24 percentage points among people already diagnosed with diabetes. Blood glucose levels rose by an average of 0.04 mmol/L (less than 1 mg/d) in those without diabetes, and by an average 0.22 mmol/L (about 4 mg/dL) in those with diabetes. People who received low- or moderate-intensity statin regimens had significant but smaller increases.
“We’re not talking about people going from no diabetes to frank diabetes. We’re talking about [statins] nudging a very small number of people across a diabetes threshold,” an A1c of 6.5% that is set somewhat arbitrarily based on an increased risk for developing retinopathy, Dr. Preiss said. ”A person just needs to lose a [daily] can of Coke’s worth of weight to eliminate any apparent diabetes risk,” he noted.
Benefit outweighs risks by three- to sevenfold
Dr. Preiss presented two other examples of what his findings showed to illustrate the relatively small risk posed by statin therapy compared with its potential benefits. Treating 10,000 people for 5 years with a high-intensity statin regimen in those with established ASCVD (secondary prevention) would result in an increment of 150 extra people developing diabetes because of the hyperglycemic effect of statins, compared with an expected prevention of 1,000 ASCVD events. Among 10,000 people at high ASCVD risk and taking a high-intensity statin regimen for primary prevention 5 years of treatment would result in roughly 130 extra cases of incident diabetes while preventing about 500 ASCVD events.
In addition, applying the new risk estimates to the people included in the UK Biobank database, whose median A1c is 5.5%, showed that a high-intensity statin regimen could be expected to raise the prevalence of those with an A1c of 6.5% or greater from 4.5% to 5.7%.
Several preventive cardiologists who heard the report and were not involved with the analysis agreed with Dr. Preiss that the benefits of statin treatment substantially offset this confirmed hyperglycemic effect.
Risk ‘more than counterbalanced by benefit’
“He clearly showed that the small hyperglycemia risk posed by statin use is more than counterbalanced by its benefit for reducing ASCVD events,” commented Neil J. Stone, MD, a cardiologist and professor of medicine at Northwestern University, Chicago. “I agree that, for those with prediabetes who are on the road to diabetes with or without a statin, the small increase in glucose with a statin should not dissuade statin usage because the benefit is so large. Rather, it should focus efforts to improve diet, increase physical activity, and keep weight controlled.”
Dr. Stone also noted in an interview that in the JUPITER trial, which examined the effects of a daily 20-mg dose of rosuvastatin (Crestor), a high-intensity regimen, study participants with diabetes risk factors who were assigned to rosuvastatin had an onset of diabetes that was earlier than people assigned to placebo by only about 5.4 weeks, yet this group had evidence of significant benefit.
“I agree with Dr. Preiss that the benefits of statins in reducing heart attack, stroke, and cardiovascular death far outweigh their modest effects on glycemia,” commented Brendan M. Everett, MD, a cardiologist and preventive medicine specialist at Brigham and Women’s Hospital in Boston. “This is particularly true for those with preexisting prediabetes or diabetes, who have an elevated risk of atherosclerotic events and thus stand to derive more significant benefit from statins. The benefits of lowering LDL cholesterol with a statin for preventing seriously morbid, and potentially fatal, cardiovascular events far outweigh the extremely modest, or even negligible, increases in the risk of diabetes that could be seen with the extremely small increases in A1c,” Dr. Everett said in an interview.
The new findings “reaffirm that there is a increased risk [from statins] but the most important point is that it is a very, very tiny difference in A1c,” commented Marc S. Sabatine, MD, a cardiologist and professor at Harvard Medical School, Boston. “These data have been known for quite some time, but this analysis was done in a more rigorous way.” The finding of “a small increase in risk for diabetes is really because diabetes has a biochemical threshold and statin treatment nudges some people a little past a line that is semi-arbitrary. It’s important to be cognizant of this, but it in no way dissuades me from treating patients aggressively with statins to reduce their ASCVD risk. I would monitor their A1c levels, and if they go higher and can’t be controlled with lifestyle we have plenty of medications that can control it,” he said in an interview.
No difference by statin type
The meta-analysis used data from 13 placebo-controlled statin trials that together involved 123,940 participants and had an average 4.3 years of follow-up, and four trials that compared one statin with another and collectively involved 30,734 participants with an average 4.9 years of follow-up.
The analyses showed that high-intensity statin treatment increased the rate of incident diabetes by a significant 36% relative to controls and increased the rate of worsening glycemia by a significant 24% compared with controls. Low- or moderate-intensity statin regimens increased incident diabetes by a significant 10% and raised the incidence of worsening glycemia by a significant 10% compared with controls, Dr. Preiss reported.
These effects did not significantly differ by type of statin (the study included people treated with atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin), nor across a variety of subgroups based on age, sex, race, body mass index, diabetes risk, renal function, cholesterol levels, or cardiovascular disease. The effect was also consistent regardless of the duration of treatment.
Dr. Preiss also downplayed the magnitude of the apparent difference in risk posed by high-intensity and less intense statin regimens. “I suspect the apparent heterogeneity is true, but not quite as big as what we see,” he said.
The mechanisms by which statins have this effect remain unclear, but evidence suggests that it may be a direct effect of the main action of statins, inhibition of the HMG-CoA reductase enzyme.
The study received no commercial funding. Dr. Preiss and Dr. Stone had no disclosures. Dr. Everett has been a consultant to Eli Lilly, Gilead, Ipsen, Janssen, and Provention. Dr. Sabatine has been a consultant to Althera, Amgen, Anthos Therapeutics, AstraZeneca, Beren Therapeutics, Bristol-Myers Squibb, DalCor, Dr Reddy’s Laboratories, Fibrogen, Intarcia, Merck, Moderna, Novo Nordisk, and Silence Therapeutics.
CHICAGO – A new, expanded meta-analysis confirmed the long-known effect that statin treatment has on raising blood glucose levels and causing incident diabetes, but it also documented that these effects are small and any risk they pose to statin users is dwarfed by the cholesterol-lowering effect of statins and their ability to reduce risk for atherosclerotic cardiovascular disease (ASCVD).
This meta-analysis of 23 trials with a total of more than 150,000 participants showed that statin therapy significantly increased the risk for new-onset diabetes and worsening glycemia, driven by a “very small but generalized increase in glucose,” with a greater effect from high-intensity statin regimens and a similar but somewhat more muted effect from low- and moderate-intensity statin treatment, David Preiss, MBChB, PhD, reported at the American Heart Association scientific sessions.
Dr. Preiss also stressed that despite this, “the cardiovascular benefits of statin therapy remain substantial and profound” in people regardless of whether they have diabetes, prediabetes, or normoglycemia when they start statin treatment, noting that the impact of even high-intensity statin treatment is “absolutely tiny” increases in hemoglobin A1c and blood glucose.
“This does not detract from the substantial benefit of statin treatment,” declared Dr. Preiss, a metabolic medicine specialist and endocrinologist at Oxford (England) University.
Small glycemia increases ‘nudge’ some into diabetes
The data Dr. Preiss reported showed that high-intensity statin treatment (atorvastatin at a daily dose of at least 40 mg, or rosuvastatin at a daily dose of at least 20 mg) led to an average increase in A1c levels of 0.08 percentage points among people without diabetes when their treatment began and 0.24 percentage points among people already diagnosed with diabetes. Blood glucose levels rose by an average of 0.04 mmol/L (less than 1 mg/d) in those without diabetes, and by an average 0.22 mmol/L (about 4 mg/dL) in those with diabetes. People who received low- or moderate-intensity statin regimens had significant but smaller increases.
“We’re not talking about people going from no diabetes to frank diabetes. We’re talking about [statins] nudging a very small number of people across a diabetes threshold,” an A1c of 6.5% that is set somewhat arbitrarily based on an increased risk for developing retinopathy, Dr. Preiss said. ”A person just needs to lose a [daily] can of Coke’s worth of weight to eliminate any apparent diabetes risk,” he noted.
Benefit outweighs risks by three- to sevenfold
Dr. Preiss presented two other examples of what his findings showed to illustrate the relatively small risk posed by statin therapy compared with its potential benefits. Treating 10,000 people for 5 years with a high-intensity statin regimen in those with established ASCVD (secondary prevention) would result in an increment of 150 extra people developing diabetes because of the hyperglycemic effect of statins, compared with an expected prevention of 1,000 ASCVD events. Among 10,000 people at high ASCVD risk and taking a high-intensity statin regimen for primary prevention 5 years of treatment would result in roughly 130 extra cases of incident diabetes while preventing about 500 ASCVD events.
In addition, applying the new risk estimates to the people included in the UK Biobank database, whose median A1c is 5.5%, showed that a high-intensity statin regimen could be expected to raise the prevalence of those with an A1c of 6.5% or greater from 4.5% to 5.7%.
Several preventive cardiologists who heard the report and were not involved with the analysis agreed with Dr. Preiss that the benefits of statin treatment substantially offset this confirmed hyperglycemic effect.
Risk ‘more than counterbalanced by benefit’
“He clearly showed that the small hyperglycemia risk posed by statin use is more than counterbalanced by its benefit for reducing ASCVD events,” commented Neil J. Stone, MD, a cardiologist and professor of medicine at Northwestern University, Chicago. “I agree that, for those with prediabetes who are on the road to diabetes with or without a statin, the small increase in glucose with a statin should not dissuade statin usage because the benefit is so large. Rather, it should focus efforts to improve diet, increase physical activity, and keep weight controlled.”
Dr. Stone also noted in an interview that in the JUPITER trial, which examined the effects of a daily 20-mg dose of rosuvastatin (Crestor), a high-intensity regimen, study participants with diabetes risk factors who were assigned to rosuvastatin had an onset of diabetes that was earlier than people assigned to placebo by only about 5.4 weeks, yet this group had evidence of significant benefit.
“I agree with Dr. Preiss that the benefits of statins in reducing heart attack, stroke, and cardiovascular death far outweigh their modest effects on glycemia,” commented Brendan M. Everett, MD, a cardiologist and preventive medicine specialist at Brigham and Women’s Hospital in Boston. “This is particularly true for those with preexisting prediabetes or diabetes, who have an elevated risk of atherosclerotic events and thus stand to derive more significant benefit from statins. The benefits of lowering LDL cholesterol with a statin for preventing seriously morbid, and potentially fatal, cardiovascular events far outweigh the extremely modest, or even negligible, increases in the risk of diabetes that could be seen with the extremely small increases in A1c,” Dr. Everett said in an interview.
The new findings “reaffirm that there is a increased risk [from statins] but the most important point is that it is a very, very tiny difference in A1c,” commented Marc S. Sabatine, MD, a cardiologist and professor at Harvard Medical School, Boston. “These data have been known for quite some time, but this analysis was done in a more rigorous way.” The finding of “a small increase in risk for diabetes is really because diabetes has a biochemical threshold and statin treatment nudges some people a little past a line that is semi-arbitrary. It’s important to be cognizant of this, but it in no way dissuades me from treating patients aggressively with statins to reduce their ASCVD risk. I would monitor their A1c levels, and if they go higher and can’t be controlled with lifestyle we have plenty of medications that can control it,” he said in an interview.
No difference by statin type
The meta-analysis used data from 13 placebo-controlled statin trials that together involved 123,940 participants and had an average 4.3 years of follow-up, and four trials that compared one statin with another and collectively involved 30,734 participants with an average 4.9 years of follow-up.
The analyses showed that high-intensity statin treatment increased the rate of incident diabetes by a significant 36% relative to controls and increased the rate of worsening glycemia by a significant 24% compared with controls. Low- or moderate-intensity statin regimens increased incident diabetes by a significant 10% and raised the incidence of worsening glycemia by a significant 10% compared with controls, Dr. Preiss reported.
These effects did not significantly differ by type of statin (the study included people treated with atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, and simvastatin), nor across a variety of subgroups based on age, sex, race, body mass index, diabetes risk, renal function, cholesterol levels, or cardiovascular disease. The effect was also consistent regardless of the duration of treatment.
Dr. Preiss also downplayed the magnitude of the apparent difference in risk posed by high-intensity and less intense statin regimens. “I suspect the apparent heterogeneity is true, but not quite as big as what we see,” he said.
The mechanisms by which statins have this effect remain unclear, but evidence suggests that it may be a direct effect of the main action of statins, inhibition of the HMG-CoA reductase enzyme.
The study received no commercial funding. Dr. Preiss and Dr. Stone had no disclosures. Dr. Everett has been a consultant to Eli Lilly, Gilead, Ipsen, Janssen, and Provention. Dr. Sabatine has been a consultant to Althera, Amgen, Anthos Therapeutics, AstraZeneca, Beren Therapeutics, Bristol-Myers Squibb, DalCor, Dr Reddy’s Laboratories, Fibrogen, Intarcia, Merck, Moderna, Novo Nordisk, and Silence Therapeutics.
AT AHA 2022
No benefit of rivaroxaban in COVID outpatients: PREVENT-HD
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
A new U.S. randomized trial has failed to show benefit of a 35-day course of oral anticoagulation with rivaroxaban for the prevention of thrombotic events in outpatients with symptomatic COVID-19.
The PREVENT-HD trial was presented at the American Heart Association scientific sessions by Gregory Piazza, MD, Brigham and Women’s Hospital, Boston.
“With the caveat that the trial was underpowered to provide a definitive conclusion, these data do not support routine antithrombotic prophylaxis in nonhospitalized patients with symptomatic COVID-19,” Dr. Piazza concluded.
PREVENT-HD is the largest randomized study to look at anticoagulation in nonhospitalized COVID-19 patients and joins a long list of smaller trials that have also shown no benefit with this approach.
However, anticoagulation is recommended in patients who are hospitalized with COVID-19.
Dr. Piazza noted that the issue of anticoagulation in COVID-19 has focused mainly on hospitalized patients, but most COVID-19 cases are treated as outpatients, who are also suspected to be at risk for venous and arterial thrombotic events, especially if they have additional risk factors. Histopathological evidence also suggests that at least part of the deterioration in lung function leading to hospitalization may be attributable to in situ pulmonary artery thrombosis.
The PREVENT-HD trial explored the question of whether early initiation of thromboprophylaxis dosing of rivaroxaban in higher-risk outpatients with COVID-19 may lower the incidence of venous and arterial thrombotic events, reduce in situ pulmonary thrombosis and the worsening of pulmonary function that may lead to hospitalization, and reduce all-cause mortality.
The trial included 1,284 outpatients with a positive test for COVID-19 and who were within 14 days of symptom onset. They also had to have at least one of the following additional risk factors: age over 60 years; prior history of venous thromboembolism (VTE), thrombophilia, coronary artery disease, peripheral artery disease, cardiovascular disease or ischemic stroke, cancer, diabetes, heart failure, obesity (body mass index ≥ 35 kg/m2) or D-dimer > upper limit of normal. Around 35% of the study population had two or more of these risk factors.
Patients were randomized to rivaroxaban 10 mg daily for 35 days or placebo.
The primary efficacy endpoint was time to first occurrence of a composite of symptomatic VTE, myocardial infarction, ischemic stroke, acute limb ischemia, non–central nervous system systemic embolization, all-cause hospitalization, and all-cause mortality up to day 35.
The primary safety endpoint was time to first occurrence of International Society on Thrombosis and Hemostasis (ISTH) critical-site and fatal bleeding.
A modified intention-to-treat analysis (all participants taking at least one dose of study intervention) was also planned.
The trial was stopped early in April this year because of a lower than expected event incidence (3.2%), compared with the planned rate (8.5%), giving a very low likelihood of being able to achieve the required number of events.
Dr. Piazza said reasons contributing to the low event rate included a falling COVID-19 death and hospitalization rate nationwide, and increased use of effective vaccines.
Results of the main intention-to-treat analysis (in 1,284 patients) showed no significant difference in the primary efficacy composite endpoint, which occurred in 3.4% of the rivaroxaban group versus 3.0% of the placebo group.
In the modified intention-to-treat analysis (which included 1,197 patients who actually took at least one dose of the study medication) there was shift in the directionality of the point estimate (rivaroxaban 2.0% vs. placebo 2.7%), which Dr. Piazza said was related to a higher number of patients hospitalized before receiving study drug in the rivaroxaban group. However, the difference was still nonsignificant.
The first major secondary outcome of symptomatic VTE, arterial thrombotic events, and all-cause mortality occurred in 0.3% of rivaroxaban patients versus 1.1% of placebo patients, but this difference did not reach statistical significance.
However, a post hoc exploratory analysis did show a significant reduction in the outcome of symptomatic VTE and arterial thrombotic events.
In terms of safety, there were no fatal critical-site bleeding events, and there was no difference in ISTH major bleeding, which occurred in one patient in the rivaroxaban group versus no patients in the placebo group.
There was, however, a significant increase in nonmajor clinically relevant bleeding with rivaroxaban, which occurred in nine patients (1.5%) versus one patient (0.2%) in the placebo group.
Trivial bleeding was also increased in the rivaroxaban group, occurring in 17 patients (2.8%) versus 5 patients (0.8%) in the placebo group.
Discussant for the study, Renato Lopes, MD, Duke University Medical Center, Durham, N.C., noted that the relationship between COVID-19 and thrombosis has been an important issue since the beginning of the pandemic, with many proposed mechanisms to explain the COVID-19–associated coagulopathy, which is a major cause of death and disability.
While observational data at the beginning of the pandemic suggested patients with COVID-19 might benefit from anticoagulation, looking at all the different randomized trials that have tested anticoagulation in COVID-19 outpatients, there is no treatment effect on the various different primary outcomes in those studies and also no effect on all-cause mortality, Dr. Lopes said.
He pointed out that PREVENT-HD was stopped prematurely with only about one-third of the planned number of patients enrolled, “just like every other outpatient COVID-19 trial.”
He also drew attention to the low rates of vaccination in the trial population, which does not reflect the current vaccination rate in the United States, and said the different direction of the results between the main intention-to-treat and modified intention-to-treat analyses deserve further investigation.
However, Dr. Lopes concluded, “The results of this trial, in line with the body of evidence in this field, do not support the routine use of any antithrombotic therapy for outpatients with COVID-19.”
The PREVENT-HD trial was sponsored by Janssen. Dr. Piazza has reported receiving research support from Bristol-Myers Squibb/Pfizer Alliance, Bayer, Janssen, Alexion, Amgen, and Boston Scientific, and consulting fees from Bristol-Myers Squibb/Pfizer Alliance, Boston Scientific, Janssen, NAMSA, Prairie Education and Research Cooperative, Boston Clinical Research Institute, and Amgen.
A version of this article first appeared on Medscape.com.
FROM AHA 2022
Test strips ID fetal tissue in vaginal blood
A rapid test strip can accurately identify embryonic or fetal tissue in vaginal blood at the bedside, say authors of a paper published in Obstetrics and Gynecology.
The strip, called the ROM Plus test, can detect alpha-fetoprotein (AFP) and insulin-like growth factor–binding protein 1 (IGFBP-1) to identify the presence of the tissue, researchers say.
A positive test could help diagnose miscarriage and rule out ectopic pregnancy.
The ROM Plus test was originally created for diagnosing rupture of amniotic membranes and has been approved by the Food and Drug Administration. This study describes an off-label use of the test.
Lead author Michelle Volovsky, MD, of the department of obstetrics and gynecology at Maimonides Medical Center in Brooklyn, N.Y., said that in the current legal climate for abortion care, the test could have an additional use for women having vaginal bleeding.
“This test could be used as evidence to confirm that a miscarriage has occurred, and hence, a D&C (dilation and curettage) procedure in that case is not an induced abortion of a viable pregnancy,” Dr. Volovsky said.
Women in study
Three groups of reproductive-age women (totaling 90) were included in the study.
One was a negative control group consisting of nonpregnant women undergoing D&C or experiencing vaginal bleeding (n = 23). The positive control group of women had confirmed intrauterine pregnancy undergoing D&C (n = 31), and the third group was a study group of pregnant women with first-trimester bleeding (n = 36). Twelve women in the study group had confirmed ectopic pregnancies.
High sensitivity and specificity
Overall, 47 women had confirmed embryonic or fetal tissue in vaginal or uterine blood samples. The test strip was accurately positive in 45 of those 47 cases for a test sensitivity of 95.7%. The other 43 had confirmed absence of embryonic or fetal tissue in their vaginal or uterine blood samples.
The test had high specificity as well. “In the absence of embryonic or fetal tissue, such as vaginal blood sampled in cases of ectopic pregnancy, threatened or complete miscarriage, or nonpregnant individuals, the test strip had a specificity of 97.7% for obtaining a negative result,” the authors wrote.
The researchers noted that the high sensitivity and specificity were seen as all tests were performed in real-time, common clinical scenarios encountered in a high-volume, urban ob.gyn. unit.
First-trimester bleeding can be common
First-trimester bleeding occurs in 20%-40% of pregnancies and results in almost 500,000 emergency department visits in the United States every year, the authors wrote.
The most common causes include threatened miscarriage, but more serious etiologies include ectopic pregnancy.
Because criteria often are not met for ectopic pregnancy, many women are told they have a pregnancy of unknown location, which can lead to extensive and expensive follow-up.
“The current study was designed to offer a simple diagnostic alternative that does not require the use of an automated laboratory analyzer,” the authors wrote.
The test could be used by patients with confirmed intrauterine pregnancies at home – a highly desirable feature for people hesitant to come into medical offices or who live in remote areas, they noted.
Questions about test’s use
Lauren Thaxton, MD, assistant professor in the department of women’s health at the University of Texas at Austin, told this publication she sees the ease of use by patients at home as the biggest benefit of the test strips.
She said she’s not sure they would add much benefit otherwise because “we already have a pretty great way of identifying pregnancy tissue by floating the products of conception.”
She said the traditional “floating products” method is very inexpensive, involving a pan and strainer, and may be more comprehensive in that it can determine whether a miscarriage is finished.
She also wonders whether in the current legal climate of abortion laws, the test could be used not only to prove that an abortion didn’t happen but used as evidence the opposite way to criminalize abortion.
It’s unfortunate that we are evaluating new technology as ‘could this cause more harm than good?’ But I think it would be wrong not to recognize the long history of criminalization of abortion as well as the current reproductive health and policy climate,” Dr. Thaxton said.
Coauthors Amir Mor, MD, PhD, and Hugh Taylor, MD, hold U.S. patent rights related to the methods described in this article. Coauthor David Seifer, MD, received payment from the Women’s Integrated Network and Rutgers Medical School. In this article the authors describe off-label use of the ROM Plus test kit, produced by Laborie USA. This device was used to test vaginal blood for the presence of embryonic or fetal products. The other authors did not report any potential conflicts of interest. Dr. Thaxton reports no relevant financial relationships.
A rapid test strip can accurately identify embryonic or fetal tissue in vaginal blood at the bedside, say authors of a paper published in Obstetrics and Gynecology.
The strip, called the ROM Plus test, can detect alpha-fetoprotein (AFP) and insulin-like growth factor–binding protein 1 (IGFBP-1) to identify the presence of the tissue, researchers say.
A positive test could help diagnose miscarriage and rule out ectopic pregnancy.
The ROM Plus test was originally created for diagnosing rupture of amniotic membranes and has been approved by the Food and Drug Administration. This study describes an off-label use of the test.
Lead author Michelle Volovsky, MD, of the department of obstetrics and gynecology at Maimonides Medical Center in Brooklyn, N.Y., said that in the current legal climate for abortion care, the test could have an additional use for women having vaginal bleeding.
“This test could be used as evidence to confirm that a miscarriage has occurred, and hence, a D&C (dilation and curettage) procedure in that case is not an induced abortion of a viable pregnancy,” Dr. Volovsky said.
Women in study
Three groups of reproductive-age women (totaling 90) were included in the study.
One was a negative control group consisting of nonpregnant women undergoing D&C or experiencing vaginal bleeding (n = 23). The positive control group of women had confirmed intrauterine pregnancy undergoing D&C (n = 31), and the third group was a study group of pregnant women with first-trimester bleeding (n = 36). Twelve women in the study group had confirmed ectopic pregnancies.
High sensitivity and specificity
Overall, 47 women had confirmed embryonic or fetal tissue in vaginal or uterine blood samples. The test strip was accurately positive in 45 of those 47 cases for a test sensitivity of 95.7%. The other 43 had confirmed absence of embryonic or fetal tissue in their vaginal or uterine blood samples.
The test had high specificity as well. “In the absence of embryonic or fetal tissue, such as vaginal blood sampled in cases of ectopic pregnancy, threatened or complete miscarriage, or nonpregnant individuals, the test strip had a specificity of 97.7% for obtaining a negative result,” the authors wrote.
The researchers noted that the high sensitivity and specificity were seen as all tests were performed in real-time, common clinical scenarios encountered in a high-volume, urban ob.gyn. unit.
First-trimester bleeding can be common
First-trimester bleeding occurs in 20%-40% of pregnancies and results in almost 500,000 emergency department visits in the United States every year, the authors wrote.
The most common causes include threatened miscarriage, but more serious etiologies include ectopic pregnancy.
Because criteria often are not met for ectopic pregnancy, many women are told they have a pregnancy of unknown location, which can lead to extensive and expensive follow-up.
“The current study was designed to offer a simple diagnostic alternative that does not require the use of an automated laboratory analyzer,” the authors wrote.
The test could be used by patients with confirmed intrauterine pregnancies at home – a highly desirable feature for people hesitant to come into medical offices or who live in remote areas, they noted.
Questions about test’s use
Lauren Thaxton, MD, assistant professor in the department of women’s health at the University of Texas at Austin, told this publication she sees the ease of use by patients at home as the biggest benefit of the test strips.
She said she’s not sure they would add much benefit otherwise because “we already have a pretty great way of identifying pregnancy tissue by floating the products of conception.”
She said the traditional “floating products” method is very inexpensive, involving a pan and strainer, and may be more comprehensive in that it can determine whether a miscarriage is finished.
She also wonders whether in the current legal climate of abortion laws, the test could be used not only to prove that an abortion didn’t happen but used as evidence the opposite way to criminalize abortion.
It’s unfortunate that we are evaluating new technology as ‘could this cause more harm than good?’ But I think it would be wrong not to recognize the long history of criminalization of abortion as well as the current reproductive health and policy climate,” Dr. Thaxton said.
Coauthors Amir Mor, MD, PhD, and Hugh Taylor, MD, hold U.S. patent rights related to the methods described in this article. Coauthor David Seifer, MD, received payment from the Women’s Integrated Network and Rutgers Medical School. In this article the authors describe off-label use of the ROM Plus test kit, produced by Laborie USA. This device was used to test vaginal blood for the presence of embryonic or fetal products. The other authors did not report any potential conflicts of interest. Dr. Thaxton reports no relevant financial relationships.
A rapid test strip can accurately identify embryonic or fetal tissue in vaginal blood at the bedside, say authors of a paper published in Obstetrics and Gynecology.
The strip, called the ROM Plus test, can detect alpha-fetoprotein (AFP) and insulin-like growth factor–binding protein 1 (IGFBP-1) to identify the presence of the tissue, researchers say.
A positive test could help diagnose miscarriage and rule out ectopic pregnancy.
The ROM Plus test was originally created for diagnosing rupture of amniotic membranes and has been approved by the Food and Drug Administration. This study describes an off-label use of the test.
Lead author Michelle Volovsky, MD, of the department of obstetrics and gynecology at Maimonides Medical Center in Brooklyn, N.Y., said that in the current legal climate for abortion care, the test could have an additional use for women having vaginal bleeding.
“This test could be used as evidence to confirm that a miscarriage has occurred, and hence, a D&C (dilation and curettage) procedure in that case is not an induced abortion of a viable pregnancy,” Dr. Volovsky said.
Women in study
Three groups of reproductive-age women (totaling 90) were included in the study.
One was a negative control group consisting of nonpregnant women undergoing D&C or experiencing vaginal bleeding (n = 23). The positive control group of women had confirmed intrauterine pregnancy undergoing D&C (n = 31), and the third group was a study group of pregnant women with first-trimester bleeding (n = 36). Twelve women in the study group had confirmed ectopic pregnancies.
High sensitivity and specificity
Overall, 47 women had confirmed embryonic or fetal tissue in vaginal or uterine blood samples. The test strip was accurately positive in 45 of those 47 cases for a test sensitivity of 95.7%. The other 43 had confirmed absence of embryonic or fetal tissue in their vaginal or uterine blood samples.
The test had high specificity as well. “In the absence of embryonic or fetal tissue, such as vaginal blood sampled in cases of ectopic pregnancy, threatened or complete miscarriage, or nonpregnant individuals, the test strip had a specificity of 97.7% for obtaining a negative result,” the authors wrote.
The researchers noted that the high sensitivity and specificity were seen as all tests were performed in real-time, common clinical scenarios encountered in a high-volume, urban ob.gyn. unit.
First-trimester bleeding can be common
First-trimester bleeding occurs in 20%-40% of pregnancies and results in almost 500,000 emergency department visits in the United States every year, the authors wrote.
The most common causes include threatened miscarriage, but more serious etiologies include ectopic pregnancy.
Because criteria often are not met for ectopic pregnancy, many women are told they have a pregnancy of unknown location, which can lead to extensive and expensive follow-up.
“The current study was designed to offer a simple diagnostic alternative that does not require the use of an automated laboratory analyzer,” the authors wrote.
The test could be used by patients with confirmed intrauterine pregnancies at home – a highly desirable feature for people hesitant to come into medical offices or who live in remote areas, they noted.
Questions about test’s use
Lauren Thaxton, MD, assistant professor in the department of women’s health at the University of Texas at Austin, told this publication she sees the ease of use by patients at home as the biggest benefit of the test strips.
She said she’s not sure they would add much benefit otherwise because “we already have a pretty great way of identifying pregnancy tissue by floating the products of conception.”
She said the traditional “floating products” method is very inexpensive, involving a pan and strainer, and may be more comprehensive in that it can determine whether a miscarriage is finished.
She also wonders whether in the current legal climate of abortion laws, the test could be used not only to prove that an abortion didn’t happen but used as evidence the opposite way to criminalize abortion.
It’s unfortunate that we are evaluating new technology as ‘could this cause more harm than good?’ But I think it would be wrong not to recognize the long history of criminalization of abortion as well as the current reproductive health and policy climate,” Dr. Thaxton said.
Coauthors Amir Mor, MD, PhD, and Hugh Taylor, MD, hold U.S. patent rights related to the methods described in this article. Coauthor David Seifer, MD, received payment from the Women’s Integrated Network and Rutgers Medical School. In this article the authors describe off-label use of the ROM Plus test kit, produced by Laborie USA. This device was used to test vaginal blood for the presence of embryonic or fetal products. The other authors did not report any potential conflicts of interest. Dr. Thaxton reports no relevant financial relationships.
FROM OBSTETRICS AND GYNECOLOGY
First-line AFib ablation cuts risk of progression vs. drug therapy
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
CHICAGO – Early ablation of atrial fibrillation (AFib) reduces the risk of progression, compared with antiarrhythmic therapies, according to results of a multicenter, randomized trial called PROGRESSIVE-AF.
Over 36 months of follow-up, the trial linked early ablation with a reduced risk of persistent AFib (1.9% vs. 7.4%), and in addition, those in the ablation group were less likely to have recurrent atrial tachyarrhythmias of any kind (56.5% vs. 77.2%), reported Jason G. Andrade, MD, at the American Heart Association scientific sessions.
Serving as a long-term extension of the EARLY-AF trial published almost 2 years ago, this trial expands evidence that progressive AFib can be attenuated, a concept that has been debated.
“Can early AFib ablation stop progression?” asked Carina Blomström-Lindqvist, MD, PhD. The invited discussant for the PROGRESSION-AF trial, Dr. Blomström-Lundqvist concluded, “here is another set of data that suggests it can.”
By another set of data, Dr. Blomström-Lindqvist was referring to a previously published multinational study called ATTEST In this study, which involved 29 sites worldwide and compared radiofrequency ablation to antiarrhythmic drug therapy, early ablation also produced a lower risk of persistent AFib at the end of 3 years (2.4% vs. 17.5%; P = .0009).
In the previously published open-label EARLY-AF trial, 303 patients with paroxysmal, untreated AFib were randomized to cryoballoon ablation or antiarrhythmic drugs. The primary endpoint was the first documented recurrence of an atrial tachyarrhythmia between 91 and 365 days. The lower rate following ablation (42.9% vs. 67.8%) represented a more than 50% reduction in risk (hazard ratio, 0.48; P < .001) relative to antiarrhythmic therapy.
In PROGRESSIVE-AF, the same 303 patients were monitored continuously for an additional 24 months with an implanted cardiac monitor programmed with an AFib-detection algorithm. The data from the monitor were obtained daily. Over the final 2 years of the study, office visits were conducted every 6 months.
Tachyarrhythmias represent primary endpoint
In addition to persistent AFib, defined as lasting ≥ 7 days or lasting 48 hours to 7 days but requiring cardioversion for termination, patients in PROGRESSIVE-AF were also monitored for recurrent atrial tachyarrhythmias, AFib burden, quality of life (QOL), and health care utilization, and safety.
The average age was roughly 58 years. Although more than one-third had hypertension, most had no other comorbidities. The authors emphasized that the study population overall was relatively young and healthy.
Those randomized to antiarrhythmic therapy in EARLY-AF/PROGRESSIVE-AF received commonly prescribed therapies titrated to maximally tolerated doses using standardized protocols. At the start of EARLY-AF, flecainide, taken by 65% of patients, was the most commonly used agent, followed by sotalol, propafenone, dronedarone, and amiodarone.
At the end of PROGRESSIVE-AF, the order of the most common therapies did not change relative to EARLY-AF, but only 49% of patients were taking flecainide and 31% were no longer taking any antiarrhythmic therapy.
At the end of 3 years of follow-up in EARLY-AF/PROGRESSIVE-AF, the difference in persistent AFib represented a 75% reduction in favor of early ablation (HR, 0.25; 95% confidence interval, 0.09-0.70).
In those treated with ablation relative to those treated with antiarrhythmic therapy, the lower rate of atrial tachyarrhythmia lasting more than 7 days (1.9% vs. 6.0%) represented a 70% risk reduction (HR, 0.30; 95% CI 0.10-0.93). The protection from cardioversion for atrial tachyarrhythmia lasting between 2 and 7 days in duration (0.6% vs. 4.7%) translated into an 86% relative reduction (HR, 0.14; 95% CI, 0.02-0.85).
The impact on QOL for those randomized to ablation, which was measured with both AFib-specific and generic measures, was meaningful to patients, according to Dr. Andrade, director of the Cardiac Electrophysiology Laboratory, Vancouver General Hospital.
For example, the mean difference in the AF Quality of Life Survey (AFEQT), was 8.0 at 1 year and 7.4 at 3 years in favor of ablation. A change of 5 points in this score is considered to be a clinically meaningful difference, according to Dr. Andrade.
Numerically, the relative risk of emergency room visits and cardioversion were lower in the ablation group, but the differences did not reach statistical significance. However, the lower hazard ratio for hospitalization was significant (HR, 0.31; 95% CI, 0.15-0.66), supporting a reduction in consumption of health care resources.
Ablation found safer than drugs
The rate of adverse events of any kind (11.0% vs. 23.5%) and serious adverse events (4.5% vs. 10.1%) were lower in the ablation group.
There were no differences in major adverse cardiovascular events observed in this period of follow-up, but Dr. Andrade pointed out that follow-up was not long enough to expect differences in these events.
Impressed by the magnitude of the reduction in persistent AFib in a population of relatively young and healthy patients considered to be at a low risk of AFib progression, Dr. Blomström-Lindqvist, a professor of cardiology at the Institution of Medical Science, Uppsala, Sweden, indicated that the data support early ablation as a means to reduce risk of this outcome.
However, she did caution that progressive AFib was observed in a relatively small proportion of patients managed with antiarrhythmic therapy at 3 years, an outcome relevant when discussing treatment options with patients.
The results were published in New England Journal of Medicine simultaneously with Dr. Andrade’s presentation.
Dr. Andrade reports financial relationships with Bayer, Bayliss, Biosense, Bristol-Myers Squibb, Medtronic and Servier. The trial, funded largely by the Canadian government and Canadian professional societies, received additional funding from Bayliss and Medtronic. Dr. Blomström-Lundqvist reports financial relationships with Bayer, Boston Scientific, Cathprint, Medtronic, and Sanofi.
AT AHA 2022